December Metanephrines, Fractionated, Free, Plasma Plasminogen Activator Inhibitor Type 1 Stool Cultures and Sensitivities
|
|
|
- Liliana Page
- 5 years ago
- Views:
Transcription
1 University of Washington December 2012 TEST change 1,25 Dihydroxy Vitamin D Frequency AFB Blood Cultures Alcohol Screen Anti Deaminated Gliadin, IgG Anti Gliadin Package Anti Mullerian Hormone Package Antiviral Sensitivity, Acyclovir and/or Foscarnet ASO Titer, SRM Calcium, Ionized, Reflexive Panel CBC, Differential & Smear Evaluation Celiac Package CMV, Quantitative by PCR Electrophoresis Tests, with and without Consult Gene Specific Colaris Hemoglobin A2 Quantification by HPLC Immunofixation Tests, with and without Consult Metabolic Panels Collection Reminder CPT New Offline Offline Test, CPT Units, Reference Range, Order Note Stability Test Unit Note New/Changes Send Out In House Reference Range New/Changes Unacceptable Metanephrines, Fractionated, Free, Plasma Plasminogen Activator Inhibitor Type 1 Stool Cultures and Sensitivities Toxic Alcohol Panel Urine Drug Screen Send Out In House Offline, Note Offline New Package Added tests NOTE: NO COURIER SERVICE There will be no courier service and our Reference Lab Services technical support will be closed on Christmas day (Tuesday, December 25th) and New Year s Day (Tuesday, January 1st).
2 1,25 Dihydroxy Vitamin D Weekly on Tuesdays; results next day. VTD125 AFB Culture, Blood Specimen Collection: Blood: Collect in Wampole Isolator 10 tube (call Reference Services at (206) for tube). Disinfect draw site AND tube top with iodine. Do not allow the iodine to pool in the stopper. Allow to dry. Do not overfill or under fill. Whenever possible, inoculate the maximum volume (10 ml) for optimal sensitivity of culture. Unacceptable: Any anticoagulant other than Isolator. Stability: Transport at room temperature to Microbiology Laboratory ASAP. Isolator tube specimens older than 12 hours will be rejected. Specimens must be received in Microbiology laboratory before 10 pm. Specimen Transport: 10 ml (min. 6 ml) blood in Isolator. Daily: 7:00 22:00 [Specimens must be received in Microbiology laboratory before 10 pm.] AFBBC Alcohol Screen Quant. Old CPT: New CPT: 82010, 82055, 84600x2 AS Anti Deaminated Gliadin, IgG On December 1, 2012 celiac testing for Anti Gliadin IgG will be replaced by a superior test, Anti Deaminated Gliadin, IgG. Old AGLIAG New ADGPG Specimen Collection: 3 ml blood in plain (red top) or SST tube. Centrifuge and separate serum. Specimen Transport: 1.0 ml (min 0.5 ml) serum. Freeze. Mon, Wed, Fri Reference Range: 0 19 Units CPT s: Price: $45.76 Anti Gliadin Package On December 1, 2012 the Anti Gliadin order package will go offline. This package ordered two tests which may be ordered separately: Anti Gliadin, IgA; and Anti Gliadin, IgG, which has been replaced by Anti Deaminated Gliadin, IgG. Offline Order : Order Separately: GLIAD AGLIAA, ADGPG Anti Mullerian Hormone Package Extra specimen collection for an Anti Mullerian Hormone study is now discontinued. Package order code RAMHGG will be taken offline. Place orders for this test using RAMHG, instead. Old RAMHGG New RAMHG Antiviral Sensitivity, Acyclovir and/or Foscarnet Testing steps for HSV antiviral sensitivities send out testing have changed and CPTs have been updated. Also, sensitivity testing to Gancyclovir, Pancyclovir and Vidarabine are no longer performed. RVCAV1 (1 st Drug) CPT: 87252, RVCAV2 (2 nd Drug) CPT: 87252, ASO Titer, SRM Old Reporting Units: Todd Units New Reporting Units: IU/mL Old Reference Ranges: 0 5 yrs: <100 Todd Units 6 16 yrs: <200 Todd Units 16 yrs: <100 Todd Units New Reference Ranges: 0 5 yrs: <100 IU/mL 6 16 yrs: <200 IU/mL 16 yrs: <100 IU/mL Old ASO New ASOS 2 UW Laboratory Medicine December 2012
3 Calcium, Ionized, Reflexive Panels Notes: For more information on direct vs. reflexive ionized calcium as well as how to order a direct ionized calcium, see the following website: tests/chemistry/plic Basic Metabolic Panel with Reflexive Ionized Calcium BMPICR Calcium, Ionized, Plasma PLIC Calcium, Ionized, Whole Blood WIC Calcium, Reflexive Ionized ICAR CBC, Differential & Smear Evaluation Stability: The UWMC Laboratory must receive and process specimen within 12 hours of collection. CBD Celiac Package New test Anti Deaminated Gliadin, IgG will replace Anti Gliadin, IgG within the Celiac Package. Old CELIAC New CELAC2 CMV, Quantitative by PCR In an effort to standardize test results for the detection of CMV DNA, the WHO has recently released the 1st International Standard for CMV PCR testing. Our assay had now been standardized with this material and will as of 1 Nov 2012 be resulted in IU/ml instead of the previous copies/ml. This will allow a more accurate comparison of results between laboratories. After this adjustment, the assay has a 95% sensitivity cut-off limit of 20 IU/ml. A conversion factor of 4 copies per 1 IU can be used to relate previous results to the new values. This conversion factor is specific to the method performed in our laboratory, and cannot be applied to methods performed in other laboratories. If you are currently caring for a CMV DNA PCR-positive patient, and have any difficulty with this conversion for serial samples, please let us know. For any additional questions, please call or Keith Jerome (kjerome@u.washington.edu, or Linda Cook, lincook@u.washington.edu, ) Test Order s: CMVQN, CMVLTC, CMVRFX Electrophoresis Tests, with and without Consult As of December 1, 2012, additional billing will be added to all existing electrophoresis panels to reflect pathologist consult, previously included in all panels, which will now be charged. New test order codes have been created to give providers an option to order these panels without consult. Existing Tests with new Pathologist Consult fees added Test Order ELP ELPMCP ELPP UELPG UELPP Name Protein Electrophoresis, Serum Monoclonal Protein Panel, Serum Protein Electrophoresis Reflexive Panel, Serum Protein electrophoresis, Urine Protein Electrophoresis Reflexive Panel, Urine Consult CPT Consult Price $ $ $ $ $28.00 New Tests without Pathologist Consult* * If technologists believe that the interpretation of a specific test result is beyond their scope of practice, the test result will be reported without an interpretation, but with a recommendation to obtain professional consultation. Test Order Name Test Details ELPNR SPEP, No MD Review Same as ELP UELPNR UPEP, No MD Review Same as UELPG Gene Specific Colaris As of December 1, 2012 the Department of Laboratory Medicine will no longer send out testing for Gene Specific Colaris. We now offer ColoSeq Single Gene testing in house. Offline Order : In house Order : RGSCOL CAGENE Hemoglobin A2 Quantification by HPLC Old Reference Ranges: 0 6 mo.: <3.4% >=7mo: % New Reference Ranges: 0 6 mo.: <3.5% >=7mo: % Test Order s: HBA2, THLR5 3
4 Immunofixation Tests, with and without Consult On December 1, 2012, additional billing will also be added to all existing immunofixation tests and panels to reflect pathologist consult, previously included in all panels, which will now be charged. New test order codes have been created to give providers an option to order these panels without consult. Existing Tests with new Pathologist Consult fees added Test Order Name Consult CPT Consult Price CIFIX Immunofixation, CSF $28.00 IFIX Immunofixation, $28.00 Serum UIFIXG Immunofixation, Urine $28.00 New Tests without Pathologist Consult* * If technologists believe that the interpretation of a specific test result is beyond their scope of practice, the test result will be reported without an interpretation, but with a recommendation to obtain professional consultation. Test Order CIFXNR IFIXNR UIFXNR Name Immunofixation, CSF, No MD Review Immunofixation, Serum, No MD Review Immunofixation, Urine, No MD Review Test Details Same as CIFIX Same as IFIX Same as UIFIXG Metabolic Panels Unacceptable: Gray top tube or green top with sodium heparin. Note: Green top with lithium heparin is acceptable. Basic Metabolic Panel BMP Basic Metabolic Panel with Reflexive Ionized Calcium BMPICR Comprehensive Metabolic Panel COMP Comprehensive and Hepatic Panel COMPHF Metanephrines, Fractionated, Free, Plasma As of December 1, 2012 the Department of Laboratory Medicine will no longer send out testing for Plasma Free Fractionated Metanephrines. We now offer this testing in house. Offline Order : In house Order : RPLMET PLMET Specimen Collection: 9 ml blood in EDTA (lavender top) tube. Specimen Transport: 2.5 ml (min 0.5 ml) EDTA plasma. Freeze. Weekly Reference Range: Metanephrine: nmol/l Normetanephrine: nmol/l CPT s: Price: $65.52 Plasminogen Activator Inhibitor Type 1 Due to an unavailability of testing reagents and decreased requests from physicians the Coagulation Section of the Department of Laboratory Medicine at the University of Washington Medical Center is moving PAI 1 Activity (Plasminogen Activator Inhibitor Type 1) testing offline immediately. Tests affected are PAI and PAIPAN and FIBSC1. As of October 31st 2012, there are no known laboratories within the US performing this test. However, this may change, as testing reagents become available to the US market at some point in the future. The Coagulation Section of the Department of Laboratory Medicine at the University of Washington Medical Center does offer PAI Antigen testing and recommends using this test as a substitute or screen for abnormal PAI 1 Activity. Test Order s Offline: PAI, PAIPAN, FIBSC1 Stool Cultures and Sensitivities As of November 1, 2012, the Department of Laboratory Medicine will no longer perform individual stool culture testing for Campylobacter, E. coli O157, Yersinia, or the Salmonella, Shigella, Campylobacter and E. coli O157 battery. It is recommended that providers choose the more inclusive stool culture order; Stool C/S and Enteric Battery. Test Order s Offline: STOCC, STOEC, STOYC, STOCEC Test Order Recommended: STOEPC 4
5 Toxic Alcohol Panel Toxic Alcohol Panel is a new test order package that combines Alcohol Screen and Ethylene Glycol testing. Specimen Collection: 6 ml blood in PST (lime green top), EDTA (lavender top), heparin (green top), fluoride/oxalate (grey top), plain (red top) or SST tube. Centrifuge with stopper on, make aliquot quickly as possible and recap aliquot tube immediately. Refrigerate serum/plasma. Specimen Transport: All available (min 3.0 ml) serum/ plasma. Daily Reference Range: Ethyl Alcohol: Negative Methyl Alcohol: Negative Acetone: Negative Isopropanol: Negative Ethylene Glycol: Negative TOXALC CPT: 82010, 82055, 84600x2 Price: $ Urine Drug Screen As of November 1, 2012, additional drugs will be added to the library of drugs detected by the urine drug screen by GCMS method. An updated list of all drugs detected may be viewed at website: web.labmed.washington.edu/tests/chemistry/udd. UDRSCG 5
6 Online Test Guide: Fee Schedule: Call Reference Lab Services at Virology Newsletter: CONTACT US: Tel: web: December 2012 Reference Lab Services University of Washington PO Box Seattle, WA Non-Profit Org. U.S. Postage PAID Seattle, WA Permit No. 62 address service requested
March Immunoglobulins, Total (IgA, IgE, IgG, IgM) Interleukin-6 (IL6) Protein Electrophoresis, CSF. Thyrotropin Receptor Antibody
 University of Washington March 2012 TEST CHANGE Alpha 1-Antitrypsin Anti-IgE Receptor Antibody Anti-Mullerian Hormone Aquaporin-4 Receptor Antibody ColoSeq - Polyposis Complement testing, C3, C4 Everolimus
University of Washington March 2012 TEST CHANGE Alpha 1-Antitrypsin Anti-IgE Receptor Antibody Anti-Mullerian Hormone Aquaporin-4 Receptor Antibody ColoSeq - Polyposis Complement testing, C3, C4 Everolimus
October Courier Service on Holidays There will be no courier service Thursday, Dec. 24th, 2011.
 University of Washington October 2011 TEST CHANGE ACTH Aldosterone BCR/ABL by PCR, Qualitative Chlamydia trachomatis & N.gonorrhoeae, Amp. NA Creutzfeldt-Jakob, CSF Desipramine Doxepine and Nordoxepine,
University of Washington October 2011 TEST CHANGE ACTH Aldosterone BCR/ABL by PCR, Qualitative Chlamydia trachomatis & N.gonorrhoeae, Amp. NA Creutzfeldt-Jakob, CSF Desipramine Doxepine and Nordoxepine,
Reflex Test Protocols
 UCH Clinical Laboratory Reflex Test Protocols Blood Bank Prepare RBCs for Transfusion (aka Crossmatch) N/A No Antibody Screen ordered Antibody Screen Direct Antiglobulin Test (DAT) Polyspecific DAT Positive
UCH Clinical Laboratory Reflex Test Protocols Blood Bank Prepare RBCs for Transfusion (aka Crossmatch) N/A No Antibody Screen ordered Antibody Screen Direct Antiglobulin Test (DAT) Polyspecific DAT Positive
COMPLIANCE. Individual Highlights: Advance Beneficiary Notices Medical Necessity IL Requisition Reflex Test List Panel Test. September 2018.
 September 2018 COMPLIANCE Dear Physician: Inova Laboratories (IL) is proud to serve the Northern Virginia community as the only full-service reference laboratory. Each year we disclose information about
September 2018 COMPLIANCE Dear Physician: Inova Laboratories (IL) is proud to serve the Northern Virginia community as the only full-service reference laboratory. Each year we disclose information about
Aerobic Wound Culture and Stain
 Aerobic Wound Culture and Stain Order Name: C WOUN RTS Test Number: 6000153 Revision Date: 03/27/2014 Aerobic Wound Culture and Stain Culture Preferred 1 ml Tissue Sterile Screwtop Container Room Temperature
Aerobic Wound Culture and Stain Order Name: C WOUN RTS Test Number: 6000153 Revision Date: 03/27/2014 Aerobic Wound Culture and Stain Culture Preferred 1 ml Tissue Sterile Screwtop Container Room Temperature
Reflex Test Protocols
 Blood Bank Prepare RBCs for Transfusion (aka Crossmatch) N/A No Antibody Screen ordered Antibody Screen Direct Antiglobulin Test (DAT) Polyspecific DAT Positive polyspecific DAT IgG DAT, C3 DAT Fetal Cell
Blood Bank Prepare RBCs for Transfusion (aka Crossmatch) N/A No Antibody Screen ordered Antibody Screen Direct Antiglobulin Test (DAT) Polyspecific DAT Positive polyspecific DAT IgG DAT, C3 DAT Fetal Cell
Interpath Laboratory, Inc. Test File Update
 As an Interpath customer who receives electronic results or sends electronic orders you may need to be notified when we update our Service Manual. Although we try to keep these changes to a minimum, laboratory
As an Interpath customer who receives electronic results or sends electronic orders you may need to be notified when we update our Service Manual. Although we try to keep these changes to a minimum, laboratory
Anti-FceR (CU Index) Antibody
 Anti-FceR (CU Index) Antibody Order Name: ANTI-FCER Test Number: 5587500 REV DATE:5/31/2007 Anti-FceR (CU Index) Antibody CU Index Preferred 2 ml (1mL) Serum Clot Activator SST (Red/Gray or Tiger Top)
Anti-FceR (CU Index) Antibody Order Name: ANTI-FCER Test Number: 5587500 REV DATE:5/31/2007 Anti-FceR (CU Index) Antibody CU Index Preferred 2 ml (1mL) Serum Clot Activator SST (Red/Gray or Tiger Top)
Basic Metabolic Panel
 Basic Metabolic Panel Order Name: CHEM 8 Test Number: 2028100 REV DATE:2/5/2008 Glucose Urea Nitrogen, Blood (BUN) Creatinine Sodium Potassium Serum/Plasma Chloride Bicarbonate Calcium Anion Gap Calculated
Basic Metabolic Panel Order Name: CHEM 8 Test Number: 2028100 REV DATE:2/5/2008 Glucose Urea Nitrogen, Blood (BUN) Creatinine Sodium Potassium Serum/Plasma Chloride Bicarbonate Calcium Anion Gap Calculated
March Molecular diagnostics. Update distribution changes. Billing and compliance. Hematology. Immunology. Referral testing
 March 2016 Update distribution changes Billing and compliance Compliance Drug Analysis CPT change Hematology Hemoglobin A1c, Screening Reference Range Change Immunology Testing schedule changes Vitamin
March 2016 Update distribution changes Billing and compliance Compliance Drug Analysis CPT change Hematology Hemoglobin A1c, Screening Reference Range Change Immunology Testing schedule changes Vitamin
Reflex Test Protocols
 University of Colorado Hospital Clinical Laboratory Reflex Test Protocols Blood Bank Prepare RBCs for Transfusion (aka Crossmatch) N/A No Antibody Screen ordered Antibody Screen Direct Antiglobulin Test
University of Colorado Hospital Clinical Laboratory Reflex Test Protocols Blood Bank Prepare RBCs for Transfusion (aka Crossmatch) N/A No Antibody Screen ordered Antibody Screen Direct Antiglobulin Test
IU Health Pathology Laboratory. DOS Updates
 IU Health Pathology Laboratory News Bulletin February 23, 2012 DOS Updates Apolipoprotein B QN Ship refrigerated plasma instead of frozen; Frozen plasma is acceptable however. Myoglobin, Serum Discontinued
IU Health Pathology Laboratory News Bulletin February 23, 2012 DOS Updates Apolipoprotein B QN Ship refrigerated plasma instead of frozen; Frozen plasma is acceptable however. Myoglobin, Serum Discontinued
Tests that have had changes to the method/ CPT code, units of measurement, scope of analysis, reference comments, or specimen requirements.
 Immediate Action In our continuing effort to provide you with the highest quality toxicology laboratory services available, we have compiled important changes regarding a number of tests we perform. Listed
Immediate Action In our continuing effort to provide you with the highest quality toxicology laboratory services available, we have compiled important changes regarding a number of tests we perform. Listed
12/10/ Immediate Action, Quest Diagnostics Nichols Institute, Valencia
 NEW TESTS Please Note: Not all test codes assigned to each assay are listed in the table of contents. Please refer to the complete listing on the page numbers indicated. Test Code Test Name Effective Date
NEW TESTS Please Note: Not all test codes assigned to each assay are listed in the table of contents. Please refer to the complete listing on the page numbers indicated. Test Code Test Name Effective Date
Alkaline Phosphatase, Bone Specific
 Alkaline Phosphatase, Bone Specific Order Name: ALK P BONE Test Number: 3656500 REV DATE:11/5/2010 Alkaline Phosphatase, Bone Specific IMMENZ Preferred 1 ml (0.3) Serum Clot Activator SST (Red/Gray or
Alkaline Phosphatase, Bone Specific Order Name: ALK P BONE Test Number: 3656500 REV DATE:11/5/2010 Alkaline Phosphatase, Bone Specific IMMENZ Preferred 1 ml (0.3) Serum Clot Activator SST (Red/Gray or
Acetylcholine Receptor Binding Antibody
 Acetylcholine Receptor Binding Antibody Order Name: ACETY BND Test Number: 5500010 REV DATE:9/17/2008 Acetylcholine Receptor Binding Antibody RIA Preferred 1 ml (0.5) Serum Clot Activator (Red Top, No-Gel)
Acetylcholine Receptor Binding Antibody Order Name: ACETY BND Test Number: 5500010 REV DATE:9/17/2008 Acetylcholine Receptor Binding Antibody RIA Preferred 1 ml (0.5) Serum Clot Activator (Red Top, No-Gel)
HML Update. Summer 2009 Volume 15, No. 3. Keeping Our Clients Informed Questions or Comments: Call
 HML Update Summer 2009 Volume 15, No. 3 Keeping Our Clients Informed Questions or Comments: Call 651-232-3500 Inside: Panel Changes...1-2 New Panels...2-3 H1N1... 3 Note of Appreciation... 3 BD Vacutainer
HML Update Summer 2009 Volume 15, No. 3 Keeping Our Clients Informed Questions or Comments: Call 651-232-3500 Inside: Panel Changes...1-2 New Panels...2-3 H1N1... 3 Note of Appreciation... 3 BD Vacutainer
 27027 Tourney Road Valencia, CA 91355 800 421 7110 www.specialtylabs.com Test Updates September 25, 2009 Dear Colleague: Specialty Laboratories is pleased to announce a new assay, PT and PTT-LA Mixing
27027 Tourney Road Valencia, CA 91355 800 421 7110 www.specialtylabs.com Test Updates September 25, 2009 Dear Colleague: Specialty Laboratories is pleased to announce a new assay, PT and PTT-LA Mixing
PENN STATE MILTON S. HERSHEY MEDICAL CENTER TABLE OF CRITICAL LAB VALUES CURRENT AS OF: 1/2015
 PENN STATE MILTON S. HERSHEY MEDICAL CENTER TABLE OF CRITICAL LAB VALUES CURRENT AS OF: 1/2015 Section: Chemistry For inpatients, once 3 consecutive critical values for the same analyte (test result) have
PENN STATE MILTON S. HERSHEY MEDICAL CENTER TABLE OF CRITICAL LAB VALUES CURRENT AS OF: 1/2015 Section: Chemistry For inpatients, once 3 consecutive critical values for the same analyte (test result) have
Activated Partial Thromboplastin Time (aptt)
 Activated Partial Thromboplastin Time (aptt) Order Name: PTT Test Number: 1500050 REV DATE:12/26/2008 Activated Partial Thromboplastin Time (aptt) CLOT Preferred 2.7 ml Whole Blood Sodium Citrate 3.2%
Activated Partial Thromboplastin Time (aptt) Order Name: PTT Test Number: 1500050 REV DATE:12/26/2008 Activated Partial Thromboplastin Time (aptt) CLOT Preferred 2.7 ml Whole Blood Sodium Citrate 3.2%
Review the following alphabetical listing of our lab test catalog for each of our offered tests. Each page has at least the following elements:
 Site: Fremont Rideout Health Group Laboratory Services Policy and Procedure Creation Date: 08/17/2009 Subject/Title: FRHG Laboratories' Catalog of Tests Q-S Document Owner: Corson, Karen Pagination: Page
Site: Fremont Rideout Health Group Laboratory Services Policy and Procedure Creation Date: 08/17/2009 Subject/Title: FRHG Laboratories' Catalog of Tests Q-S Document Owner: Corson, Karen Pagination: Page
Specimen Type Specimen Container Transport Environment Preferred 2mL(0.5) Serum Clot Activator SST (Red/Gray or Tiger Top) Refrigerated
 Aldolase Order Name: ALDOLASE Test Number: 3600150 Aldolase Enz Preferred 2mL(0.5) Serum Clot Activator SST (Red/Gray or Tiger Top) Refrigerated Special Hemolyzed specimens are not acceptable. Allow specimen
Aldolase Order Name: ALDOLASE Test Number: 3600150 Aldolase Enz Preferred 2mL(0.5) Serum Clot Activator SST (Red/Gray or Tiger Top) Refrigerated Special Hemolyzed specimens are not acceptable. Allow specimen
Alaska Native Medical Center Anchorage, AK
 ANMC Lab Test Requirements Key: Room Temp (20-25C), Refrigerated (2-8C), (-15 to -25C), Hr (Hours), D (Days), W (Weeks), Mo (Months), Yr (Years). Basic Processing Instructions: Centrifuge all blood specimens
ANMC Lab Test Requirements Key: Room Temp (20-25C), Refrigerated (2-8C), (-15 to -25C), Hr (Hours), D (Days), W (Weeks), Mo (Months), Yr (Years). Basic Processing Instructions: Centrifuge all blood specimens
Tests that have had changes to the method/ CPT code, units of measurement, scope of analysis, reference comments, or specimen requirements.
 Immediate Action In our continuing effort to provide you with the highest quality toxicology laboratory services available, we have compiled important changes regarding a number of tests we perform. Listed
Immediate Action In our continuing effort to provide you with the highest quality toxicology laboratory services available, we have compiled important changes regarding a number of tests we perform. Listed
June Monthly Update, Quest Diagnostics Nichols Institute, Valencia
 June 2013 - Monthly Update, NEW TESTS Please Note: Not all test codes assigned to each assay are listed in the table of contents. Please refer to the complete listing on the page numbers indicated. Test
June 2013 - Monthly Update, NEW TESTS Please Note: Not all test codes assigned to each assay are listed in the table of contents. Please refer to the complete listing on the page numbers indicated. Test
1. Purpose 1.1. To define testing locations, schedule and order priority for each test performed in the core laboratory.
 Department Of Pathology GEN.1017.03 Integrated Test Schedule Version# 5 Department Specimen Processing POLICY NO. 1938 PAGE NO. 1 OF 6 Printed copies are for reference only. Please refer to the electronic
Department Of Pathology GEN.1017.03 Integrated Test Schedule Version# 5 Department Specimen Processing POLICY NO. 1938 PAGE NO. 1 OF 6 Printed copies are for reference only. Please refer to the electronic
H. PYLORI AB, IGG HPYL
 H. PYLORI AB, IGG HPYL Specimen Required: 3 ml blood, Serum gel tube Analytical Time: 1 day CPT Code: 86677 HAPTOGLOBIN HAP Specimen Required: 1.5 ml blood, gel tube Reference Range: 30-200 mg/dl CPT Code:
H. PYLORI AB, IGG HPYL Specimen Required: 3 ml blood, Serum gel tube Analytical Time: 1 day CPT Code: 86677 HAPTOGLOBIN HAP Specimen Required: 1.5 ml blood, gel tube Reference Range: 30-200 mg/dl CPT Code:
Tests that have had changes to the method/ CPT code, units of measurement, scope of analysis, reference comments, or specimen requirements.
 In our continuing effort to provide you with the highest quality toxicology laboratory services available, we have compiled important changes regarding a number of tests we perform. Listed below are the
In our continuing effort to provide you with the highest quality toxicology laboratory services available, we have compiled important changes regarding a number of tests we perform. Listed below are the
COLLECTION TUBES FOR PHLEBOTOMY
 COLLECTION TUBES FOR PHLEBOTOMY Red Top None Blood clots, and the serum is separated by centrifugation Chemistries, Immunology and Serology, Blood Bank (Crossmatch) Gold Top None Serum separator tube (SST)
COLLECTION TUBES FOR PHLEBOTOMY Red Top None Blood clots, and the serum is separated by centrifugation Chemistries, Immunology and Serology, Blood Bank (Crossmatch) Gold Top None Serum separator tube (SST)
Patient Preparation Unique patient preparation requirements are listed under each test in the Test Directory.
 Specimen Collection Lab results are only as good as the specimen provided. Patient preparation, venipuncture technique, specimen handling, and transportation can all affect the quantity of results. Health
Specimen Collection Lab results are only as good as the specimen provided. Patient preparation, venipuncture technique, specimen handling, and transportation can all affect the quantity of results. Health
TEST REQUEST INFORMATION- VIROLOGY
 Chlamydia/ Gonorrhea Nucleic Acid Amplification VC75 Qualitative Nucleic Acid Amplification SPECIMEN: Genital swab or first catch urine CONTAINER:GEN-PROBE APTIMA 2 Combo swab transport tube or urine transport
Chlamydia/ Gonorrhea Nucleic Acid Amplification VC75 Qualitative Nucleic Acid Amplification SPECIMEN: Genital swab or first catch urine CONTAINER:GEN-PROBE APTIMA 2 Combo swab transport tube or urine transport
Reference Range: Age/sex dependent; appropriate normals provided with result Analytical Time: 4 hours Day(s) Test Set Up: Daily CPT Code: 84100
 Parathyroid Hormone See PTH PARIETAL CELL ANTIBODY APCAB; PARIETAL; PCA Specimen Required: 2.5 ml blood, gel tube Analytical Time: 3-5 days CPT Code: 86255 PCP SCREEN URINE PCPQU Specimen Required: 50
Parathyroid Hormone See PTH PARIETAL CELL ANTIBODY APCAB; PARIETAL; PCA Specimen Required: 2.5 ml blood, gel tube Analytical Time: 3-5 days CPT Code: 86255 PCP SCREEN URINE PCPQU Specimen Required: 50
March Monthly Update, Quest Diagnostics Nichols Institute, Valencia
 NEW TESTS Please Note: Not all test codes assigned to each assay are listed in the table of contents. Please refer to the complete listing on the page numbers indicated. Test Code Test Name Effective Date
NEW TESTS Please Note: Not all test codes assigned to each assay are listed in the table of contents. Please refer to the complete listing on the page numbers indicated. Test Code Test Name Effective Date
10/7/ Immediate Action, Quest Diagnostics Nichols Institute, Valencia
 REDIRECTS Please Not all test codes assigned to each assay are listed in the table of contents. Please refer to the complete listing on the page numbers indicated. Test Code Test Name Effective Date Page
REDIRECTS Please Not all test codes assigned to each assay are listed in the table of contents. Please refer to the complete listing on the page numbers indicated. Test Code Test Name Effective Date Page
Current September 2017
 1 Lab Service PRICE 87077 PATHOGEN ID 6 87088 PRESUMP ID, URINE CULTUR 6 87140 HSV CULTURE TYPING 13 87186 MIC (MIN INHIB CONC) 7 AMYLASE, SERUM 3 ANA SCREEN/RFX TITER AND DSDNA 4 ANA SCREEN/RFX TITER/COMPREHEN
1 Lab Service PRICE 87077 PATHOGEN ID 6 87088 PRESUMP ID, URINE CULTUR 6 87140 HSV CULTURE TYPING 13 87186 MIC (MIN INHIB CONC) 7 AMYLASE, SERUM 3 ANA SCREEN/RFX TITER AND DSDNA 4 ANA SCREEN/RFX TITER/COMPREHEN
HANDLING GENERAL INFORMATION TEST REQUESTS TYPES OF TESTS TEST REQUEST FORM
 SECTION 3: SAMPLE COLLECTION & HANDLING GENERAL INFORMATION SAMPLE INTRODUCTION COLLECTION & HANDLING GENERAL results. INFORMATION The quality of laboratory test results depends greatly on the quality
SECTION 3: SAMPLE COLLECTION & HANDLING GENERAL INFORMATION SAMPLE INTRODUCTION COLLECTION & HANDLING GENERAL results. INFORMATION The quality of laboratory test results depends greatly on the quality
All abnormal results for this test will automatically have the DNA test reflexed at an additional cost.
 2211 Michigan Avenue Santa Monica, CA 90404 800 421 4449 Dear Colleague: May-June 2004 REVISED 6-15-04 At Specialty we continue to focus on working with you to reduce the problems that can result in tests
2211 Michigan Avenue Santa Monica, CA 90404 800 421 4449 Dear Colleague: May-June 2004 REVISED 6-15-04 At Specialty we continue to focus on working with you to reduce the problems that can result in tests
February Billing and Compliance. Chemistry/Immunology. Help Us Help You. Microbiology. Referral Testing. Supplies. Website
 February 2014 Billing and Compliance ICD 10 update It s coming! Chemistry/Immunology Hepatitis B Antibody reporting change Help Us Help You Completion of Allina Health Laboratory manual request forms Unscheduled
February 2014 Billing and Compliance ICD 10 update It s coming! Chemistry/Immunology Hepatitis B Antibody reporting change Help Us Help You Completion of Allina Health Laboratory manual request forms Unscheduled
Business Unit: Quest Diagnostics Nichols Institute, Valencia. June 2011 Update Dear Colleague,
 Volume 2 Apr/2011 Business Unit: June 2011 Update Dear Colleague, This letter begins with a thank you for your very prompt response to our recent request to visit our website and update your contact information.
Volume 2 Apr/2011 Business Unit: June 2011 Update Dear Colleague, This letter begins with a thank you for your very prompt response to our recent request to visit our website and update your contact information.
January Billing and compliance. Chemistry. Help us help you. Referral testing. Additional CPT code changes announced for 2015
 January 2015 Billing and compliance Additional CPT code changes announced for 2015 Chemistry CA 27.29 conversion to CA 15-3 nearing completion Vitamin B12 and LD, Total specimen change Timed Urine Protein
January 2015 Billing and compliance Additional CPT code changes announced for 2015 Chemistry CA 27.29 conversion to CA 15-3 nearing completion Vitamin B12 and LD, Total specimen change Timed Urine Protein
IMMEDIATE HOT LINE: Effective January 6, 2014
 MEDICARE COVERAGE OF LABORATORY TESTING Please remember when ordering laboratory tests that are billed to Medicare/Medicaid or other federally funded programs, the following requirements apply: 1. Only
MEDICARE COVERAGE OF LABORATORY TESTING Please remember when ordering laboratory tests that are billed to Medicare/Medicaid or other federally funded programs, the following requirements apply: 1. Only
November Monthly Update, Quest Diagnostics Nichols Institute, Valencia
 NEW TESTS Please Note: Not all test codes assigned to each assay are listed in the table of contents. Please refer to the complete listing on the page numbers indicated. Test Code Test Name Effective Date
NEW TESTS Please Note: Not all test codes assigned to each assay are listed in the table of contents. Please refer to the complete listing on the page numbers indicated. Test Code Test Name Effective Date
REFLEX TESTING LIST March 2012
 Aeromonas/Plesiomonas Positive findings Organism ID & Susceptibility April 2011 Culture *AFB Culture and Stain Positive findings Organism ID & Susceptibility Nov. 1999 *AFB Culture Only Positive findings
Aeromonas/Plesiomonas Positive findings Organism ID & Susceptibility April 2011 Culture *AFB Culture and Stain Positive findings Organism ID & Susceptibility Nov. 1999 *AFB Culture Only Positive findings
 27027 Tourney Road Valencia, CA 91355 800 421 7110 www.specialtylabs.com Test Updates November 26, 2008 Dear Colleague: We are pleased to announce the availability of two new assays for an expanded evaluation
27027 Tourney Road Valencia, CA 91355 800 421 7110 www.specialtylabs.com Test Updates November 26, 2008 Dear Colleague: We are pleased to announce the availability of two new assays for an expanded evaluation
October Billing and compliance. Coagulation. Immunohistochemistry. Immunology. Referral testing ICD 10
 October 2015 Billing and compliance ICD 10 Coagulation Processing, transport and stability changes for multiple coagulation tests Immunohistochemistry IHC stain updates Immunology Lyme Western Blot testing
October 2015 Billing and compliance ICD 10 Coagulation Processing, transport and stability changes for multiple coagulation tests Immunohistochemistry IHC stain updates Immunology Lyme Western Blot testing
April 28, RE: Immediate Action test change notification. Dear Valued Client:
 April 28, 2010 RE: Immediate Action test change notification Dear Valued Client: The following pages detail important information regarding a test requiring database modifications. Please be advised that
April 28, 2010 RE: Immediate Action test change notification Dear Valued Client: The following pages detail important information regarding a test requiring database modifications. Please be advised that
 27027 Tourney Road Valencia, CA 91355 800 421 7110 www.specialtylabs.com Test Updates July 27, 2009 Dear Colleague: As we prepare for the upcoming flu season, Specialty Laboratories clients should be aware
27027 Tourney Road Valencia, CA 91355 800 421 7110 www.specialtylabs.com Test Updates July 27, 2009 Dear Colleague: As we prepare for the upcoming flu season, Specialty Laboratories clients should be aware
If a critical result is obtained for a clinic patient after the clinic has closed, we will notify the individual on-call for the clinic.
 UCSF Clinical Laboratories Critical ('Panic') and Subcritical Values Policy (Last updated 11/3/17): In collaboration with the clinical services, the following results have been determined to represent
UCSF Clinical Laboratories Critical ('Panic') and Subcritical Values Policy (Last updated 11/3/17): In collaboration with the clinical services, the following results have been determined to represent
Sample Selection- Vignettes
 Sample Selection- Vignettes Rangaraj Selvarangan, BVSc, PhD, D(ABMM) Professor, UMKC School of Medicine Director, Microbiology, Virology and Molecular Infectious Diseases Laboratory Director, Laboratory
Sample Selection- Vignettes Rangaraj Selvarangan, BVSc, PhD, D(ABMM) Professor, UMKC School of Medicine Director, Microbiology, Virology and Molecular Infectious Diseases Laboratory Director, Laboratory
MICROBIOLOGY SPECIMEN COLLECTION MANUAL
 Lee Memorial Health System Lee County, FL CLINICAL LABORATORY MICROBIOLOGY SPECIMEN COLLECTION MANUAL ACID FAST CULTURE Specimen Type see Specimen Chart ACID FAST STAIN see Specimen Chart Acid Fast stain
Lee Memorial Health System Lee County, FL CLINICAL LABORATORY MICROBIOLOGY SPECIMEN COLLECTION MANUAL ACID FAST CULTURE Specimen Type see Specimen Chart ACID FAST STAIN see Specimen Chart Acid Fast stain
Dynacare Laboratories Test Changes
 CHROMIUM CHROMIUM CHROM CHROM 82495 82495 3508250 3508250 Plasma/ Whole blood 3.0 ml 3.0 ml Royal Blue Top (EDTA) Tube Royal Blue Top (EDTA) Tube Separate plasma/serum within 45 minutes of collection,
CHROMIUM CHROMIUM CHROM CHROM 82495 82495 3508250 3508250 Plasma/ Whole blood 3.0 ml 3.0 ml Royal Blue Top (EDTA) Tube Royal Blue Top (EDTA) Tube Separate plasma/serum within 45 minutes of collection,
Please note that our serum opiates EIA screen assay is not appropriate for therapeutic drug monitoring of Oxycodone.
 2211 Michigan Avenue Santa Monica, CA 90404 800 421 4449 Dear Colleague: October, 2004 Last month, in anticipation of cold and flu season, we introduced the Respiratory Virus DetectR (Hexaplex ) with Influenza
2211 Michigan Avenue Santa Monica, CA 90404 800 421 4449 Dear Colleague: October, 2004 Last month, in anticipation of cold and flu season, we introduced the Respiratory Virus DetectR (Hexaplex ) with Influenza
Ward Manual. PHC Diagnostic Virology & Reference Laboratory
 PHC Diagnostic Virology & Reference Laboratory I. General Information The SPH Virology Laboratory is located at St. Paul s Hospital 1081 Burrard Street, Room 625, Burrard Building, Vancouver, B.C. V6Z
PHC Diagnostic Virology & Reference Laboratory I. General Information The SPH Virology Laboratory is located at St. Paul s Hospital 1081 Burrard Street, Room 625, Burrard Building, Vancouver, B.C. V6Z
Interpath Laboratory, Inc. Test File Update
 As an Interpath customer who receives electronic results or sends electronic orders you may need to be notified when we update our Service Manual. Although we try to keep these changes to a minimum, laboratory
As an Interpath customer who receives electronic results or sends electronic orders you may need to be notified when we update our Service Manual. Although we try to keep these changes to a minimum, laboratory
Pathology and Laboratory
 Pathology and Laboratory CPT CPT copyright 2011 American Medical Association. All rights reserved. Fee schedules, relative value units, conversion factors and/or related components are not assigned by
Pathology and Laboratory CPT CPT copyright 2011 American Medical Association. All rights reserved. Fee schedules, relative value units, conversion factors and/or related components are not assigned by
July Monthly Update, Quest Diagnostics Nichols Institute, Valencia
 NEW TESTS Please Note: Not all test codes assigned to each assay are listed in the table of contents. Please refer to the complete listing on the page numbers indicated. Test Name Effective Date Page #
NEW TESTS Please Note: Not all test codes assigned to each assay are listed in the table of contents. Please refer to the complete listing on the page numbers indicated. Test Name Effective Date Page #
The Medical Laboratory
 The Medical Laboratory Purpose of a Laboratory testing Record an individuals state of health Satisfy employment, Insurance or legal requirements Research and clinical trials Detect asymptomatic conditions
The Medical Laboratory Purpose of a Laboratory testing Record an individuals state of health Satisfy employment, Insurance or legal requirements Research and clinical trials Detect asymptomatic conditions
History of the Vacutainer Tube. Coagulant Blood Tests
 History of the Vacutainer Tube The different tubes are known as a vacutainer and were developed by Joseph Kleiner in 1947. The vacutainer is currently being manufactured by Becton, Dickinson and Company
History of the Vacutainer Tube The different tubes are known as a vacutainer and were developed by Joseph Kleiner in 1947. The vacutainer is currently being manufactured by Becton, Dickinson and Company
St. Louis University Forensic Toxicology Laboratory
 St. Louis University Forensic Toxicology Laboratory Specimen Collection Guidelines Postmortem Forensic Toxicology Sample Collection Guidelines Scope The provides comprehensive analysis of general unknowns
St. Louis University Forensic Toxicology Laboratory Specimen Collection Guidelines Postmortem Forensic Toxicology Sample Collection Guidelines Scope The provides comprehensive analysis of general unknowns
Atypical Pneumonia Antibodies
 Atypical Pneumonia Antibodies Order Name: ATYP PNEUM Test Number: 5564200 REV DATE:1/17/2011 Adenovirus IgG and IgM Antibodies Chlamydia pneumoniae IgM Antibody Chlamydia pneumoniae IgG Antibody Influenza
Atypical Pneumonia Antibodies Order Name: ATYP PNEUM Test Number: 5564200 REV DATE:1/17/2011 Adenovirus IgG and IgM Antibodies Chlamydia pneumoniae IgM Antibody Chlamydia pneumoniae IgG Antibody Influenza
Cleveland Clinic Laboratories
 Cleveland Clinic Laboratories Technical Update February 2016 Cleveland Clinic Laboratories is dedicated to keeping you updated and informed about recent testing changes. That s why we are happy to provide
Cleveland Clinic Laboratories Technical Update February 2016 Cleveland Clinic Laboratories is dedicated to keeping you updated and informed about recent testing changes. That s why we are happy to provide
35.0. ug/ml Trough. 8.0 Carbamazepine 15.0 ug/ml Digoxin 2.5 ng/ml Gentamicin Peak Random Trough
 The following are a list by department of Critical (Panic) Values: CHEMISTRY TEST LESS THAN GREATER THAN UNITS Glucose 0-7days 40 200 mg/dl Glucose > 7 days 50 400 mg/dl Glucose, CSF 25 mg/dl Sodium 121
The following are a list by department of Critical (Panic) Values: CHEMISTRY TEST LESS THAN GREATER THAN UNITS Glucose 0-7days 40 200 mg/dl Glucose > 7 days 50 400 mg/dl Glucose, CSF 25 mg/dl Sodium 121
Applicable To Employees of the Gundersen Boscobel Area Hospital Laboratory and Gundersen Palmer Lutheran Hospital and Clinics laboratories.
 Subject Calcium C311 Index Number Lab-8810 Section Laboratory Subsection Regional/Affiliates Category Departmental Contact Judy Dayton Last Revised 4/11/2017 References Required document for Laboratory
Subject Calcium C311 Index Number Lab-8810 Section Laboratory Subsection Regional/Affiliates Category Departmental Contact Judy Dayton Last Revised 4/11/2017 References Required document for Laboratory
Clinical Microbiology CLS 2019 Clinical Microbiology Laboratory Scheme Application Form
 Laboratory Scheme Application Form complete all sections below and return to LGC Standards Proficiency Testing by email, fax or post. Returning customer Lab ID: Purchase order no.: (compulsory) Round Despatch
Laboratory Scheme Application Form complete all sections below and return to LGC Standards Proficiency Testing by email, fax or post. Returning customer Lab ID: Purchase order no.: (compulsory) Round Despatch
Specimen Collection. Urine Vacutainers. Laboratory Services September 2018
 1 Specimen Collection Urine Vacutainers Laboratory Services September 2018 Agenda Before you begin Collection Labeling Packaging & Transport Resources Click on the links above to be taken directly to that
1 Specimen Collection Urine Vacutainers Laboratory Services September 2018 Agenda Before you begin Collection Labeling Packaging & Transport Resources Click on the links above to be taken directly to that
Test Definition: HBELC Hemoglobin Electrophoresis Cascade, Blood
 Reporting Title: HGB Electrophoresis Cascade Performing Location: Rochester Advisory Information: Alpha-thalassemias with only 1 or 2 alpha-globin gene deletions are not recognized by this testing protocol.
Reporting Title: HGB Electrophoresis Cascade Performing Location: Rochester Advisory Information: Alpha-thalassemias with only 1 or 2 alpha-globin gene deletions are not recognized by this testing protocol.
Recommendations for Celiac Disease Testing. IgA & ttg IgA
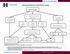 Recommendations for Celiac Disease Testing IgA & ttg IgA IgA Normal ttg IgA Negative IgA >10 mg/dl, but less than age matched range IgA
Recommendations for Celiac Disease Testing IgA & ttg IgA IgA Normal ttg IgA Negative IgA >10 mg/dl, but less than age matched range IgA
TEST LIST SAMPLE REQUIREMENT. 1 ml serum None
 ALBUMIN TEST NAME ALKALINE PHOSPHATASE ALLERGY PROFILE, FOOD 30 allergens ALLERGY PROFILE, INHALANT 30 Allergens ALT AMYLASE ANA ANTI- TG ANTI-GLIADIN IGG ANTI-GLIADIN IGA ANTI-HBS ANTI-HCV ANTI-TPO APOLIPOPROTEIN
ALBUMIN TEST NAME ALKALINE PHOSPHATASE ALLERGY PROFILE, FOOD 30 allergens ALLERGY PROFILE, INHALANT 30 Allergens ALT AMYLASE ANA ANTI- TG ANTI-GLIADIN IGG ANTI-GLIADIN IGA ANTI-HBS ANTI-HCV ANTI-TPO APOLIPOPROTEIN
Test Definition: FNMR2 NMR LipoProfile w/ir Markers
 Reporting Title: Performing Location: Labcorp Burlington Specimen Requirements: Submit only one of the following specimens: Serum Draw blood in a plain red-top tube(s). (Serum gel tube is not acceptable.)
Reporting Title: Performing Location: Labcorp Burlington Specimen Requirements: Submit only one of the following specimens: Serum Draw blood in a plain red-top tube(s). (Serum gel tube is not acceptable.)
Specimen Collection Requirements
 The following is a job aid listing the specimen collection requirements for laboratory testing at Colchester East Hants Health Center. Specimens must be accompanied by the Patient Information Form G09.
The following is a job aid listing the specimen collection requirements for laboratory testing at Colchester East Hants Health Center. Specimens must be accompanied by the Patient Information Form G09.
 27027 Tourney Road Valencia, CA 91355 800 421 7110 www.specialtylabs.com Test Updates October 28, 2009 Dear Colleague: Specialty Laboratories is pleased to announce the January availability of two trace
27027 Tourney Road Valencia, CA 91355 800 421 7110 www.specialtylabs.com Test Updates October 28, 2009 Dear Colleague: Specialty Laboratories is pleased to announce the January availability of two trace
Test Bulletin. Save time, money, resources, and precious blood by NOT collecting extra specimens just in case
 Test Bulletin October 2018 Save time, money, resources, and precious blood by NOT collecting extra specimens just in case ACL Laboratories has seen an increase in the number of specimens submitted without
Test Bulletin October 2018 Save time, money, resources, and precious blood by NOT collecting extra specimens just in case ACL Laboratories has seen an increase in the number of specimens submitted without
Interpath Laboratory, Inc. Test File Update
 As an Interpath customer who receives electronic results or sends electronic orders you may need to be notified when we update our Service Manual. Although we try to keep these changes to a minimum, laboratory
As an Interpath customer who receives electronic results or sends electronic orders you may need to be notified when we update our Service Manual. Although we try to keep these changes to a minimum, laboratory
Minimum Whole Blood Volumes for Microcollection Tubes for Neonates, Pediatrics, Patients less than 45 kg (100 lb) and Difficult Collections
 The following table outlines the minimum whole blood volume that must be drawn into a microcollection tube (unless otherwise stated) for an individual test on a neonate, pediatric, patient weighing less
The following table outlines the minimum whole blood volume that must be drawn into a microcollection tube (unless otherwise stated) for an individual test on a neonate, pediatric, patient weighing less
Pilot studies with reference specimens
 Pilot studies with reference specimens HUPO: Plasma Proteome Project Workshop July 16-17, 17, 2003 Daniel W. Chan, Ph.D.,DABCC Professor of Pathology, Oncology, Radiology & Urology Director of Clinical
Pilot studies with reference specimens HUPO: Plasma Proteome Project Workshop July 16-17, 17, 2003 Daniel W. Chan, Ph.D.,DABCC Professor of Pathology, Oncology, Radiology & Urology Director of Clinical
TELEFAX. Toxoplasma Serology Laboratory (TSL) DATE: TO: FAX: FROM:
 TELEFAX DATE: TO: FAX: FROM: Toxoplasma Serology Laboratory RE: SPECIMEN REQUIREMENTS AND TESTING INFORMATION (effective 1/15/16) THIS TRANSMISSION CONSISTS OF 16 PAGES INCLUDING THIS COVER PAGE. IF THERE
TELEFAX DATE: TO: FAX: FROM: Toxoplasma Serology Laboratory RE: SPECIMEN REQUIREMENTS AND TESTING INFORMATION (effective 1/15/16) THIS TRANSMISSION CONSISTS OF 16 PAGES INCLUDING THIS COVER PAGE. IF THERE
Lab Director: Russell P. Tracy, Ph.D. Coordinator: Elaine Cornell Lab/Project Manager: Rebekah Boyle, M.S. Repository Manager: Sarah Nightingale
 Presented by: Jessica Rooney Colchester Research Facility Laboratory for Clinical Biochemistry Research (LCBR) University of Vermont Department of Pathology and Laboratory Medicine Lab Director: Russell
Presented by: Jessica Rooney Colchester Research Facility Laboratory for Clinical Biochemistry Research (LCBR) University of Vermont Department of Pathology and Laboratory Medicine Lab Director: Russell
LANE RMC TEST DICTIONARY For Lab testing not listed below - contact
 LANE RMC TEST DICTIONARY For Lab testing not listed below - contact 658-4320 Revised 03/2017 TEST NAME SPECIMEN VOLUME CONTAINER SPECIAL INSTRUCTIONS Acetone Plasma or Serum 1ml Green Gel or SST Green
LANE RMC TEST DICTIONARY For Lab testing not listed below - contact 658-4320 Revised 03/2017 TEST NAME SPECIMEN VOLUME CONTAINER SPECIAL INSTRUCTIONS Acetone Plasma or Serum 1ml Green Gel or SST Green
Collection (Specimen Source Required on all tests) Sputum: >5 ml required. First morning specimen preferred.
 Type Acid Fast (Mycobacteria) Sputum: >5 ml required. First morning specimen preferred. For blood, sodium heparin tube preferred. Lithium heparin acceptable. Do not centrifuge.. delay. Swabs are not appropriate
Type Acid Fast (Mycobacteria) Sputum: >5 ml required. First morning specimen preferred. For blood, sodium heparin tube preferred. Lithium heparin acceptable. Do not centrifuge.. delay. Swabs are not appropriate
October Monthly Update, Quest Diagnostics Nichols Institute, Valencia
 ANNOUNCEMENTS Please Note: Not all test codes assigned to each assay are listed in the table of contents. Please refer to the complete listing on the page numbers indicated. Test Name Page # Reactivated
ANNOUNCEMENTS Please Note: Not all test codes assigned to each assay are listed in the table of contents. Please refer to the complete listing on the page numbers indicated. Test Name Page # Reactivated
SERVICE SCHEDULE, FEES & GUIDELINES FOR SAMPLE SHIPMENT
 SERVICE SCHEDULE, FEES & GUIDELINES FOR SAMPLE SHIPMENT ACYLCARNITINE PROFILE: PLASMA/SERUM, PKU CARD, WHOLE BLOOD Amount of sample: Plasma (heparinized is preferred, EDTA acceptable) or serum: 0.5 ml
SERVICE SCHEDULE, FEES & GUIDELINES FOR SAMPLE SHIPMENT ACYLCARNITINE PROFILE: PLASMA/SERUM, PKU CARD, WHOLE BLOOD Amount of sample: Plasma (heparinized is preferred, EDTA acceptable) or serum: 0.5 ml
HOSPITALS IN-COMMON LABORATORY INC.
 Immunoglobulin E Ab., Single Allergen: Allergens are now searchable and viewable in the web test manual database but are not listed individually in the printable pdf-format documents. Trace Metals: Trace
Immunoglobulin E Ab., Single Allergen: Allergens are now searchable and viewable in the web test manual database but are not listed individually in the printable pdf-format documents. Trace Metals: Trace
Sample Selection, Collection, Transport: Issues & Challenges
 Sample Selection, Collection, Transport: Issues & Challenges Jim Dunn, PhD, D(ABMM) Director, Medical Microbiology and Virology Texas Children s Hospital LEARNING OBJECTIVES 1. Identify factors to consider
Sample Selection, Collection, Transport: Issues & Challenges Jim Dunn, PhD, D(ABMM) Director, Medical Microbiology and Virology Texas Children s Hospital LEARNING OBJECTIVES 1. Identify factors to consider
CRITICAL VALUES, SIGNIFICANT FINDINGS LIST and STAT RESULTS
 Page 1 of 6 Affected Sites: X Enterprise Chandler Good Samaritan CRITICAL VALUES: s are defined as an analytic result that suggests a clinical condition that may be lifethreatening and may require immediate
Page 1 of 6 Affected Sites: X Enterprise Chandler Good Samaritan CRITICAL VALUES: s are defined as an analytic result that suggests a clinical condition that may be lifethreatening and may require immediate
The information contained here may be very important to your practice. Please take a moment to review this document.
 September 2017 Dear Healthcare Provider, The information contained here may be very important to your practice. Please take a moment to review this document. TEST BULLETIN T-SPOT TB TESTING Please see
September 2017 Dear Healthcare Provider, The information contained here may be very important to your practice. Please take a moment to review this document. TEST BULLETIN T-SPOT TB TESTING Please see
Please contact Customer Service with any questions. Ph: option 5
 Sarasota Memorial Laboratory Services is pleased to announce that we are now performing the following testing in house: Immature Platelet Fraction (IPF) The Immature Platelet Fraction (IPF) is used to
Sarasota Memorial Laboratory Services is pleased to announce that we are now performing the following testing in house: Immature Platelet Fraction (IPF) The Immature Platelet Fraction (IPF) is used to
INTERPATH LABORATORY, INC. TEST UPDATES
 EFFECTIVE DATE: February 16, 2016 INTERPATH LABORATORY, INC. TEST UPDATES Please take note of the following test updates and make the appropriate changes in your Interpath Service Manual. Feel free to
EFFECTIVE DATE: February 16, 2016 INTERPATH LABORATORY, INC. TEST UPDATES Please take note of the following test updates and make the appropriate changes in your Interpath Service Manual. Feel free to
Specimen Collection Requirements
 The following is a job aid listing the specimen collection requirements for laboratory testing at Colchester East Hants Health Center. Specimens must be accompanied by the Patient Information Form G09.
The following is a job aid listing the specimen collection requirements for laboratory testing at Colchester East Hants Health Center. Specimens must be accompanied by the Patient Information Form G09.
Technical Bulletin No. 172
 CPAL Central Pennsylvania Alliance Laboratory QuantiFERON -TB Gold Plus Assay Contact: J Matthew Groeller, MPA(HCM), MT(ASCP), 717-851-4516 Operations Manager, Clinical Pathology, CPAL Jennifer Thebo,
CPAL Central Pennsylvania Alliance Laboratory QuantiFERON -TB Gold Plus Assay Contact: J Matthew Groeller, MPA(HCM), MT(ASCP), 717-851-4516 Operations Manager, Clinical Pathology, CPAL Jennifer Thebo,
LIST OF TESTS WITH COLLECTION INFORMATION. Form Page 1 of 19
 ALL TESTS Tests listed are the most commonly ordered tests. For further information on tests not listed call the laboratory, look for the test in the computer or look at Lab Corp Manual ACTH (Adenocorticotropic
ALL TESTS Tests listed are the most commonly ordered tests. For further information on tests not listed call the laboratory, look for the test in the computer or look at Lab Corp Manual ACTH (Adenocorticotropic
2019 Reference Manual Updates
 2019 Reference Manual Updates (January 2019 to date) The enclosed Client Grams include updated and supplemental information to our most recently published Reference Manual (2019) and are intended to be
2019 Reference Manual Updates (January 2019 to date) The enclosed Client Grams include updated and supplemental information to our most recently published Reference Manual (2019) and are intended to be
Clinical Chemistry (CHE 221)
 Clinical Chemistry (CHE 221) Experiment # 14 Blood Alcohol Determination by Gas Chromatography and by Reaction with Alcohol Dehydrogenase Name Date Performed Date Submitted Partners Name(s) Partners Name(s)
Clinical Chemistry (CHE 221) Experiment # 14 Blood Alcohol Determination by Gas Chromatography and by Reaction with Alcohol Dehydrogenase Name Date Performed Date Submitted Partners Name(s) Partners Name(s)
Color tube for vitamin d
 Color tube for vitamin d Gogamz Menu What's New and Beneficial About Broccoli. Since our recommended cooking method for broccoli has always been Quick Steaming, we are excited to report on numerous. Feb
Color tube for vitamin d Gogamz Menu What's New and Beneficial About Broccoli. Since our recommended cooking method for broccoli has always been Quick Steaming, we are excited to report on numerous. Feb
Standard Operating Procedure
 Standard Operating Procedure 1. Definitions Section: Laboratory Version No. 1.2 Initials: AD Title: 2.0 Testing Algorithm Revision Date: 01 Sep 2011 1.1 SOP: Standard Operating Procedure 1.2 CRF: Case
Standard Operating Procedure 1. Definitions Section: Laboratory Version No. 1.2 Initials: AD Title: 2.0 Testing Algorithm Revision Date: 01 Sep 2011 1.1 SOP: Standard Operating Procedure 1.2 CRF: Case
2014 Notice to Physicians
 2014 Notice to Physicians August 20, 2014 Dear Physician, will only pay for tests that are deemed medically necessary. These regulations impact physicians who order the tests as well as the laboratories
2014 Notice to Physicians August 20, 2014 Dear Physician, will only pay for tests that are deemed medically necessary. These regulations impact physicians who order the tests as well as the laboratories
In-Common Laboratories
 Stability Information: Specimen stability information has been added or revised for the following tests: Vitamin B6, Dehydroepiandrosterone, Thyroid Antibodies (Thyroid Peroxidase Ab & Thyroglobulin Ab)
Stability Information: Specimen stability information has been added or revised for the following tests: Vitamin B6, Dehydroepiandrosterone, Thyroid Antibodies (Thyroid Peroxidase Ab & Thyroglobulin Ab)
Tests that have had changes to the method/ CPT code, units of measurement, scope of analysis, reference comments, or specimen requirements.
 Updates In our continuing effort to provide you with the highest quality toxicology laboratory services available, we have compiled important changes regarding a number of tests we perform. Listed below
Updates In our continuing effort to provide you with the highest quality toxicology laboratory services available, we have compiled important changes regarding a number of tests we perform. Listed below
Lab Guide 2019 Metabolic Section Lab Guide
 Lab Guide 2019 Metabolic Section Lab Guide Quantitative Amino acids Plasma Plasma. Container/Tube: Preferred EDTA, Place immediately in ice. Acceptable: lithium heparin, sodium heparin. Patient preparation:
Lab Guide 2019 Metabolic Section Lab Guide Quantitative Amino acids Plasma Plasma. Container/Tube: Preferred EDTA, Place immediately in ice. Acceptable: lithium heparin, sodium heparin. Patient preparation:
C-1-Q IMMUNE COMPLEX Immune Complex, C-1-Q Binding Assay
 C-1-Q IMMUNE COMPLEX Immune Complex, C-1-Q Binding Assay 5 ml blood, gel tube Special Instructions: Avoid hemolysis. Transfer serum to plastic vial immediately after centrifugation and freeze. Less than
C-1-Q IMMUNE COMPLEX Immune Complex, C-1-Q Binding Assay 5 ml blood, gel tube Special Instructions: Avoid hemolysis. Transfer serum to plastic vial immediately after centrifugation and freeze. Less than
