Asthma drug ratios and exacerbations: claims data from universal health coverage systems
|
|
|
- Matthew Horton
- 5 years ago
- Views:
Transcription
1 ORIGINAL ARTICLE ASTHMA Asthma drug ratios and exacerbations: claims data from universal health coverage systems Laurent Laforest 1, Idlir Licaj 1, Gilles Devouassoux 2,Gérard Chatte 3, Jennifer Martin 1 and Eric Van Ganse 1,2 Affiliations: 1 Unité de Pharmacoépidémiologie, CHU-Lyon, Faculté d Odontologie, UMR 5558-LBBE, CNRS, Université Claude Bernard, Lyon, 2 Service de Pneumologie, Hôpital de la Croix Rousse, Hospices Civils de Lyon, Lyon, and 3 Liberal Chest Physician, Caluire, France. Correspondence: E. Van Ganse, Unité de Pharmacoépidémiologie, 11, rue Guillaume Paradin, Lyon, Cedex 08, France. eric.van-ganse@univ-lyon1.fr ABSTRACT In claims data, controller-to-total asthma drug ratios may reflect adequacy of disease management. We verified whether asthma patients with high ratios (o50%) experienced fewer asthmarelated outcomes. Two ratios were studied: that of the inhaled corticosteroids to total asthma drug (ICS/ R03) and that of the inhaled corticosteroids plus leukotriene antagonist receptors-to-total asthma drug (ICS+LTRA/R03). Patients aged years, with o3 respiratory drugs dispensed prescriptions in 2005 were selected from the French national claims data. After excluding null ratios, two groups were defined according to ratio values in 2007: low-ratio group (0%,ratio,50%) and high-ratio group (ratio o50%). For both ratios, asthma-related outcomes and medical-resource utilisation were compared between groups. Of 2162 patients (mean age 27 years and 52% female), patients with non-null ratios were 81% and 85% for ICS/R03 and ICS+LTRA/R03 ratios, respectively. Patients with high ratios were less likely to receive oral corticosteroids than those in the low-ratio group (relative risk 0.79, 95% CI , and 0.80, 95% CI , for ICS/R03 and ICS+LTRA/R03, respectively). High ratio groups also presented fewer asthmarelated hospitalisations. Significant negative correlations were also observed for both ratios, when studied quantitatively, according to patients dispensed level of oral corticosteroids in In claims data, both ICS/R03 and ICS+LTRA/R03 o50% were related to fewer asthma-related outcomes. Ratios should be explored to identify asthma patients at risk of exacerbations. Low ratios can be considered as risk factors of exacerbation whatever the underlying Asthma patients with therapeutic ratios o50% had fewer asthma exacerbation markers, suggesting better control Received: June Accepted after revision: Nov Support statement: This study was supported by a grant from the French National Health Insurance Agency (grant number 11/6/2009/CT/49). Conflict of interest: Disclosures can be found alongside the online version of this article at Copyright ßERS Eur Respir J 2014; 43: DOI: /
2 Introduction Regular use of controller medications remains a key issue in asthma disease management. Observational studies have shown that inconsistent use of controller medications has a direct impact on medical resource utilisation [1, 2]. One promising approach to identifying patients with inconsistent controller use is through claims data, which include exhaustive information on reimbursed medical resource utilisation in the insured population. Using these data to identify patients at risk of insufficient control of chronic diseases, such as asthma, may help improve disease management [3]. Authors have investigated ratios measuring the proportion of dispensed controllers in total asthma therapy as a marker of the quality of care [4, 5]. Studies using these ratios have consistently shown higher levels of asthma-related hospital admissions and emergency room visits for patients with lower ratios [6]. The impact of ratios on oral corticosteroids (OCSs) has been less extensively explored. Furthermore, unlike controller-to-total asthma drug ratio, the inhaled corticosteroid (ICS)-to-total asthma drug ratio (ICS/R03, where R03 codes for asthma therapy according to the Anatomical Therapeutic and Chemical (ATC) classification system) has been poorly studied. It is also unclear whether considering only ICSs or both ICS and leukotriene receptor antagonist (LTRA) agents in the numerator ratio yield concordant results. Lastly, previous studies have relied on data from health management organisations, on specific populations and typically salaried persons with specific processes of care, and there is a need to replicate such studies in other healthcare systems with universal coverage, including patients with a high level of deprivation. We investigated whether low ICS/R03 ratios, which suggest less consistent ICS exposure for a given disease severity, were related to more frequent asthma-related exacerbations and greater overall medical resource utilisation. Parallel investigations were conducted for ICS+LTRA/R03 ratio to verify whether both ratios yielded concordant findings. For each ratio, we also looked for differences in patient characteristics and controller therapy between the high and low ratio groups. Methods Study design and timelines A historical cohort ( ) was obtained from the Permanent Sample of Health Insurance Beneficiaries (EGB), a 1/97th random sample of the French National Claims Data Beneficiaries with individual linkage between ambulatory and hospital care. We selected patients aged years on January 1, 2005, with continuous follow-up between 2005 and 2008, and o3 asthma drugs (R03 code) dispensed in three different quarters during The year 2006 was used to assess patients asthma severity. Severity was approached by the total number of asthma drug classes (including OCSs) dispensed in We conducted analyses in 2007 and 2008 (fig. 1). This observational study was conducted on anonymised claims data, and the National Informatics and Liberty Committee has delivered an overall authorisation to use EGB data for research purposes. Data collected Patient characteristics were age, sex, long-term disease status and free-access-to-care status. Long-term disease status allows severe patients to receive therapy without advancing payment in pharmacies. Freeaccess-to-care status enables patients with socioeconomic difficulties to receive free medical care. Reimbursed therapy included asthma medications, OCSs and antibiotics. Medical contacts (family physicians and respiratory physicians) were counted in the database. We identified hospitalisations with asthma as primary or secondary diagnosis. The primary diagnosis corresponds to the disease that incurred the majority of the resources used during a given hospital stay. ICS/R03 and ICS+LTRA/R03 ratios The ratio of the number of ICS units (whether in fixed combination with long-acting b-agonists (LABAs) or not) to the overall units of asthma drugs (R03 ATC classification) dispensed in 2007 was computed. Asthma drug therapy included ICSs, LABAs, LABA ICS fixed combinations, short-acting b-agonists (SABAs), LTRAs, anticholinergics, anticholinergics SABA fixed combinations, xanthines and cromones (R03 according to ATC classification). Based on this ICS/R03 ratio in 2007, three groups were defined: 0% (no ICS), 0%, ratio,50% (low ratio) and ratio o50% (high ratio). The choice of the 50% threshold was based on previous studies using controller-to-total respiratory drug ratios [4]. The ratio of ICS plus LTRA to total asthma drug (ICS+LTRA/R03), including both LTRA and ICS units in the numerator, was also computed and studied. DOI: /
3 Inclusion period Assessment of asthma severity period Study period Patients aged years 3 reimbursed dispensings of anti-asthma drugs (R03 ATC classification) in at least three different quarters of 2005 Number of dispensed classes of anti-asthma drugs (including oral corticosteroids) Assessment of exposure to ICS (2007) Ratio 1: ICS to total anti-asthma drug ratio Ratio 2: ICS+LTRA to total anti-asthma drug ratio Studied outcomes Markers of asthma exacerbation in 2007 Markers of asthma exacerbation in 2008 FIGURE 1 Study design. ICS: inhaled corticosteroid; R03 ATC: R03 coding for asthma therapy according to the Anatomical Therapeutic and Chemical classification system; LTRA: leukotriene receptor antagonist. Outcome criteria Outcomes were proxies for asthma exacerbations in 2007 and 2008 [7]. The main outcomes were dispensing of the drug classes commonly used to treat asthma exacerbations, i.e. OCSs (percentage of patients with at least one drug dispensed and mean number of prescriptions dispensed). The annual number of visits to a family physician was also investigated. Other criteria were hospitalisations for asthma as primary diagnosis and hospitalisations for asthma as primary or secondary diagnosis. Percentages of patients with at least one hospitalisation, the number of stays per patient and the hospitalisation-related costs were studied in 2007 and Levels of SABAs dispensed were not used as an outcome, as they were included in the denominator of ICS/R03 ratios. Analyses Analyses were conducted in parallel for ICS/R03 and ICS+LTRA/R03 ratios. For both ratios, all analyses were restricted to low- and high-ratio groups (no null-ratio values). First, baseline characteristics and controller therapy dispensed in 2007 were compared between groups. The outcomes were compared between groups in 2007 and in 2008 using standard Chi-squared test (Fisher s exact test when appropriate), ANOVA or Wilcoxon Kruskal Wallis test. Then, multivariate Poisson models were run to compute the risks of receiving OCSs in 2007 (at least one dispensed prescription) for both ratios. It was verified whether patients in the high-ratio group presented lower risks of receiving OCSs in 2007 compared with those in the low-ratio group. Models were adjusted for age, sex, long-term disease status, free-access-to-care status, dispensing of at least one LABA ICS fixed combination in 2007, and ratio group in 2007 (high versus low). Complementary models were also computed with an additional adjustment for the baseline severity variable. The aim was to verify the extent to which differences in outcomes between groups were due to factors other than baseline severity. In the absence of clinical and spirometric data, adjustment for baseline severity was approached by the number of 1380 DOI: /
4 dispensed respiratory drug classes in 2006, including OCSs (0 1, 2 3 and.3) [8]. Similar analyses were also conducted for receiving OCSs in Owing to their low frequency, asthma-related hospitalisations were not studied in multivariate analyses. All analyses were performed on SAS software version 9.3 (SAS Institute Inc., Cary, NC, USA). Additional analyses with quantitative ratios Additional analyses with quantitative ratios were performed for ICS/R03 and ICS+LTRA/R03 ratios. In 2007, both ratios were studied according to the number of dispensed units of OCS in 2007, and then in Univariate and multivariate Poisson regressions were conducted. Multivariate analyses were adjusted for the same cofactors as those in the main analyses. Results Descriptive results Both ratios were computed for 2162 patients (mean age 27 years and 52% female) who met the inclusion criteria, with at least one asthma drug dispensed in The proportions of patients in the no-ics (null ratio), and low- and high-ics ratio groups were 19%, 36% and 45%, respectively. Respective percentages for ICS+LTRA/R03 ratios were 15%, 25% and 59%. After excluding null ratios, analyses were conducted in 1758 and 1827 patients for ICS/R03 and ICS+LTRA/R03 ratios, respectively. Patient characteristics and dispensed controller therapy according to the ICS/R03 and ICS+LTRA/ R03 ratios For both ratios, patients in the low-ratio groups more frequently had free-access-to-care and long-termdisease status (table 1). Conversely, there were no significant differences by either sex or age. Compared with the low-ratio groups, patients in the high-ratio groups received significantly more ICS units, particularly as LABA ICS fixed combinations; received fewer LABAs (not combined); and had fewer primary care consultations in 2007, while they tended to visit more respiratory physicians, although this was not significant (table 1). Patients in the ICS/R03 high-ratio group received fewer LTRAs than those in the ICS/R03 low-ratio group. This was reversed for the ICS+LTRA/R03 ratio. Asthma-related outcomes according to ICS/R03 and ICS+LTRA/R03 ratios Compared with the low-ratio groups, patients in the high-ratio groups had fewer dispensed prescriptions of OCSs in 2007 and in 2008 (table 2). They also experienced fewer asthma-related hospitalisations. Multivariate analyses Patients with a high ratio had a significantly lower risk of receiving OCSs in 2007 compared with those in the low-ratio group. For both ratios, the association persisted in the complementary models, even after adjustment for baseline severity. Similar trends, although less marked, were also observed for receiving OCSs in 2008 (table 3). Additional analyses with quantitative ratios Significant decreases in mean ratios were observed with the number of dispensed units of OCSs in 2007 for both ICS/R03 and ICS+LTRA/R03 ratios. Similar downward trends, although less marked, also appeared with the OCSs dispensed in Results of regression models of ratios with OCS dispensing in 2007 remained significant in multivariate analyses (table 4). Discussion In our sample of 2162 patients, ICS/R03 and ICS+LTRA/R03 ratios were non-null ratios in 81% and 85%, respectively. In those with non-null ratios, the ICS/R03 high-ratio group accounted for 55% of patients. It accounted for 70% of the ICS+LTRA/R03 ratio. Overall, patients in the low-ratio groups experienced more use of OCSs and more asthma-related hospitalisations than those in the high-ratio group (table 2). For both ratios, the risk of receiving OCSs in 2007 was significantly lower in the high-ratio group compared with the low-ratio group, even after adjustment for asthma severity (table 3). The complementary models indicate that the potential difference in asthma severity between groups can explain only in part the lower risks of outcomes in the high ratio group. Likewise, patients with a low ratio experienced more asthmarelated hospitalisations. Additional analyses showed a limited downward variation of quantitative ratios with patients level of dispensed OCSs, although results were statistically significant in univariate analyses (table 4). DOI: /
5 TABLE 1 Patients characteristics according to ICS/R03 drug ratio in 2007 ICS/R03 ICS+LTRA/R03 Low-ratio group # High-ratio p-value Low-ratio group " group + High-ratio group 1 p-value Subjects n Mean age e years Males % Long-term disease status ## % Free-access-to-care status "" % Medical resource utilisation in 2007 o1 visit to a respiratory physician ++ % Mean visits to family physician , Controllers ICS (other than LABA ICS fixed combination) o1 unit % , , Mean number of dispensed units , LABA ICS fixed combination o1 unit % , , Mean number of dispensed units , , Mean number of ICS units (any type) , , LABA (not in LABA ICS fixed combination) o1 unit % , , Mean number of dispensed units , , LTRA o1 unit % , , Mean number of dispensed units , , ICS: inhaled corticosteroid; RO3: Anatomical Therapeutic and Chemical classification system code for asthma therapy; LTRA: leukotriene receptor antagonists. # :0%,ICS/R03,50%; " : ICS/R03 o50%; + :0%,ICS+LTRA/RO3,50%; 1 : ICS+LTRA/RO3o50%; e : age in 2005; ## : patients are dispensed their asthma therapy without advancing the money at the pharmacy; "" : patients with socioeconomic difficulties receive free care; ++ : respiratory physicians with a private consulting room (dispensing resulting from prescriptions from hospital respiratory physicians cannot be identified in the data). Overall, our main findings are in line with previous studies that reported fewer asthma-related hospitalisations and emergency room visits when the controller-to-total asthma drug ratio was o50% [4, 5]. A lower risk of belonging to the high ratio group was found in controller-treated patients with at least two refills of OCSs (OR50.89, 95% CI ) [9]. However, SCHATZ et al. [10] identified a significantly lower risk of receiving OCSs only at a 90% threshold. The exclusion of null -ratios and potential differences in prescribing habits between countries may account for these differences. Indeed, better asthma control and quality of life have been found by SCHATZ et al. [11] in patients with a controller-to-total asthma drug ratio of o50%. In addition to lower exacerbation-related medical resource utilisation during the same calendar year, a high ICS/R03 ratio seems to predict outcomes in the following year, reinforcing a potential role for this marker. However, caution is needed given the more limited differences observed between groups with outcomes measured in 2008 (table 2). Other differences between high- and low-ratio users are noteworthy. The number of medical visits was higher in patients with low ratios, possibly due to more frequent unscheduled visits. As expected, dispensed levels of ICS were higher in the high-ratio groups, although these differences between low- and high-ratio groups were not large (table 1). In addition, patients with high ratios tended to receive more LABA ICS fixed combinations (table 1). This finding has also been observed by BRODER et al. [9]. Patients under LABA ICS fixed combinations are less likely to require SABAs and do not use LABAs, which would decrease the ratio denominator. More regular dispensing of LABA ICS fixed combinations compared with ICS alone has also been observed in claims data [12 14]. However, multivariate models clearly indicated that patients in the high-ratio groups were less likely to receive OCSs, irrespective of concomitant use of LABA ICS fixed combinations. The greater frequency of patients visiting specialists in the high-ratio group is in line with previous studies [9]. A better quality of care provided by respiratory physicians could account for a higher ICS dispensation DOI: /
6 TABLE 2 Asthma-related outcomes according to ICS/R03 and ICS+LTRA/R03 ratios in 2007 ICS/R03 ICS+LTRA/R03 Low-ratio group # High-ratio p-value Low-ratio group " group + High-ratio group 1 p-value Subjects n Oral corticosteroids In 2007 At least one unit dispensed % , , Mean dispensed units e In 2008 At least one unit dispensed % Mean dispensed units e Asthma-related hospitalisations (primary diagnosis) In 2007 Hospitalised % Mean number of stays e In 2008 Hospitalised % Mean number of stays e Asthma-related hospitalisations (primary or secondary diagnosis) In 2007 Hospitalised % Mean number of stays e In 2008 Hospitalised % Mean number of stays e ICS: inhaled corticosteroid; RO3: Anatomical Therapeutic and Chemical classification system code for asthma therapy; LTRA: leukotriene receptor antagonists. # :0%,ICS/R03,50%; " : ICS/R03 o50%; + :0%,ICS+LTRA/RO3,50%; 1 : ICS+LTRA/RO3o50%; e : computed in both users and nonusers. Nonetheless, caution is required, given the low number of private-sector respiratory physicians in our data and nonsignificant results. Patients with free-access-to-care status were more common in low-ratio groups, suggesting that social difficulties may be an obstacle to reaching high ratios. Patients with long-term disease status were also more common among low-ratio users, suggesting potentially more severe asthma in this group with high levels of medical resource utilisation. These points require confirmation in new studies. The ICS/R03 and ICS+LTRA/R03 ratio groups differed as to distribution, using the 50% cut-off value. In particular, patients using both ICS and LTRA were more likely to belong to the ICS/R03 low-ratio group, which was not the case for the ICS+LTRA/R03 ratio. Expectedly, the ICS+LTRA/R03 high-ratio group tended to receive more LTRA than the low-ratio group, while it was the other way round for the ICS/R03 ratio (table 1). Further investigations are needed to better understand the differences between both ratios. Our ICS+LTRA/R03 ratio was close to the defintion of SCHATZ et al. [4], although not identical. For instance, cromones and xanthines were not included in the numerator, while the denominator comprised all LABAs, whether or not as LABA ICS fixed combinations. However, cromones and xanthines are used only marginally. Although there were still LABA users (not combined) at the time of the study (table 1), their proportion has noticeably dropped in France in recent years (personal communication; E. Van Ganse, Unité de Pharmacoépidémiologie, CHU-Lyon, Faculté d Odontologie, Université Claude Bernard, Lyon, France). Nevertheless, despite these differences in definitions, concordant conclusions were obtained with previous studies, indicating a robustness of these markers of quality of care. Besides simplicity of computation, an advantage of using the ratios is the consistency of the benefits observed in the high-ratio groups (o50%) [6], even when the numerator is limited to ICS therapy. The presence of all asthma therapy in the denominator partly accounts for asthma severity, ensuring robustness to an indication bias. DOI: /
7 TABLE 3 Risks of receiving one or more dispensed prescription of OCSs in case of a high ratio value (o50%) for ICS/R03 (models 1 and 2) and for ICS+LTRA/R03 (models 3 and 4) multivariate models ICS/R03 ICS+LTRA/R03 Model 1 Model 2 Model 3 Model 4 Subjects n Initial multivariate model # Low ratio (0%,ratio,50%) High ratio (ratio o50%) 0.79 ( ) 0.84 ( ) 0.80 ( ) 0.89 ( ) Complementary model with additional adjustment for baseline severity " Low ratio (0%,ratio,50%) High ratio (ratioo50%) 0.89 ( ) 0.92 ( ) 0.85 ( ) 0.94 ( ) Data are presented as relative risk (95% CI). Models 1 and 3: risk of receiving o1 dispensed prescription of oral corticosteroids (OCSs) in 2007; models 2 and 4: risk of receiving o1 dispensed prescription of OCSs in ICS: inhaled corticosteroid; R03: Anatomical Therapeutic and Chemical classification system code for asthma therapy; LTRA: leukotriene receptor antagonist. # : relative risk is adjusted for age (15 29, and years), sex, long-term disease status, free-access-to-care status, o1 visit to a respiratory specialist in 2007 and o1 dispensed presription of long-acting b-agonist ICS fixed combination; " : relative risk is adjusted for the same factors as #, with an additional adjustment for baseline severity as assessed by the number of dispensed respiratory drug classes in 2006, including OCSs (0 1, 2 3 and.3 dispensed prescriptions). Ratios could be used not only in administrative claims databases to identify asthma patients at risk of exacerbations, but could be of interest for identifying such patients in daily medical practice, e.g. from computerised medical records. Some limitations must be acknowledged. Hospitalisations with asthma were uncommon, precluding models with these outcomes. It is noteworthy that our specific asthma-related hospitalisation rate was consistent with those previously reported by the National Health Service in the UK [15]. Although OCSs are TABLE 4 Mean ratio values according to OCS dispensing levels in 2007 and in 2008 OCS units dispensed n o3 Univariate Poisson regression # Multivariate Poisson regression #," OCS units dispensed in 2007 ICS/R03 ratio Number Mean % b , z5-4.88, p, b , z5-2.15, p50.03 ICS+LTRA/R03 ratio Number Mean % b , z5-4.45, p, b , z5-2.86, p OCS units dispensed in 2008 ICS/R03 ratio Number Mean % b , z5-2.69, p b , z5-0.89, p50.37 ICS+LTRA/R03 ratio Number Mean % b , z5-2.32, p50.02 b , z5-1.34, p50.18 OCS: oral corticosteroid; ICS: inhaled corticosteroid; R03: Anatomical Therapeutic and Chemical classification system code for asthma therapy; LTRA: leukotriene receptor antagonist. # : z stands for statistical test-value. " : adjusted for age (15 29, and years), sex, long-term disease status, free-access-to-care status, o1 visit to a respiratory specialist in 2007, o1 dispensing of LABA ICS fixed combination and baseline severity as approached by the number of dispensed respiratory drug classes in 2006, including OCSs (0 1, 2 3 and.3 dispensings) DOI: /
8 recommended in epidemiological studies as outcomes of severe asthma exacerbation [7], they may not be specific for asthma. Additionally, OCSs may be prescribed preventively to patients prior to episodes of exacerbation. However, despite these limitations, significant differences were noted for OCS dispensing levels between ratio groups (tables 3 and 4). Our ratios did not take into account the number of doses per inhaler or the potency of ICS drugs. SCHATZ et al. [10] computed a more elaborate ratio including these variables. However, the basic ratio turned out to be more discriminating for asthma-related outcomes [10]. Severe patients, at higher risk of exacerbations tended to receive more potent ICSs and would more easily qualify as having a higher weighted ratio. Hence, more frequent outcomes would be expected in the high-ratio group when using weighted ratios, which could decrease the difference between low- and high-ratio groups. Additionally, basic ratios are easier to use in practice. Caution is also needed, given the absence of clinical data in claims data. Also, our sample may not be representative of the overall population of asthma patients as it consists of a subgroup of selected patients with regular follow-up, accounting for 1.3% of patients aged years in the EGB data. Less frequently treated asthma patients, potentially at high risk of adverse outcomes, were not included. Finally, to define a high ratio, we used the same cut-off (o50%) as SCHATZ et al. [10], as this threshold has yielded consistent conclusions for various outcomes and facilitates comparisons with previous studies. A simple and unique threshold value, such as 50%, is desirable. Our findings have several implications. Unlike private insurance data, a noticeable advantage of the French claims data lies in their representativeness of the French population, and our data confirm the interest of ratios in non-us healthcare systems. Our findings also support a role for both ICS/R03 and ICS+LTRA/R03 ratios computed from claims data to detect patients potentially at risk of exacerbations. Ratios, independent of the underlying levels of severity/control of the disease and mechanisms, could be used in public health and patient management as a tool to identify patients at higher risk of exacerbation. Due to the paucity of patient characteristics and clinical data in claims databases, it is difficult to further explain differences between low- and high-ratio groups for medical and personal characteristics and, notably, from a socioeconomic point of view. Our ability to identify a higher level of OCS dispensing in the case of low ratio using the 50% threshold does not mean that this value is the most discriminative value for this outcome. The next step of methodological investigations should be the determination of the optimal threshold for OCS dispensing, for both ratios. Another area of research of interest would be the identification of the determinants of such low ratios in a prospective design. More generally, studying the impact of the ratios with complementary levels of drug exposure (prescriptions, dispensings and actual patient use) could shed light on the reasons for inconsistent use of ICS in asthma management. A low ICS/R03 ratio may be due to irregular dispensings of ICS, e.g. as a result of patients incomplete adherence to prescribed therapy or irregular prescription of ICSs by physicians [16, 17]. In conclusion, patients with ICS/R03 ratio or ICS+LTRA/R03 ratio o50% experienced fewer markers of exacerbations, most notably compared with low ICS users, suggesting improved asthma control. Ratios may not only help to identify asthma patients at risk of severe exacerbations from claims data, but they may be of help in clinical practice as a tool to assess the management of asthma. References 1 Suissa S, Ernst P, Kezouh A. Regular use of inhaled corticosteroids and the long term prevention of hospitalisation for asthma. Thorax 2002; 57: Blais L, Ernst P, Boivin JF, et al. Inhaled corticosteroids and the prevention of readmission to hospital for asthma. Am J Respir Crit Care Med 1998; 158: Van Ganse E, van der Linden PD, Leufkens HG, et al. Asthma medications and disease exacerbations: an epidemiological study as a method for asthma surveillance. Eur Respir J 1995; 8: Schatz M, Nakahiro R, Crawford W, et al. Asthma quality-of-care markers using administrative data. Chest 2005; 128: Yong PL, Werner RM. Process quality measures and asthma exacerbations in the medicaid population. J Allergy Clin Immunol 2009; 124: Schatz M, Zeiger RS. Improving asthma outcomes in large populations. J Allergy Clin Immunol 2011; 128: Busse WW, Morgan WJ, Taggart V, et al. Asthma outcomes workshop: overview. J Allergy Clin Immunol 2012; 129: Suppl. 3, S1 S8. 8 Taylor DR, Bateman ED, Boulet LP, et al. A new perspective on concepts of asthma severity and control. Eur Respir J 2008; 32: Broder MS, Gutierrez B, Chang E, et al. Ratio of controller to total asthma medications: determinants of the measure. Am J Manag Care 2010; 16: Schatz M, Zeiger RS, Yang SJ, et al. Relationship of asthma control to asthma exacerbations using surrogate markers within a managed care database. Am J Manag Care 2010; 16: DOI: /
9 11 Schatz M, Zeiger RS, Vollmer WM, et al. The controller-to-total asthma medication ratio is associated with patientcentered as well as utilization outcomes. Chest 2006; 130: Stempel DA, Stoloff SW, Carranza Rosenzweig JR, et al. Adherence to asthma controller medication regimens. Respir Med 2005; 99: Stoloff SW, Stempel DA, Meyer J, et al. Improved refill persistence with fluticasone propionate and salmeterol in a single inhaler compared with other controller therapies. J Allergy Clin Immunol 2004; 113: Marceau C, Lemière C, Berbiche D, et al. Persistence, adherence, and effectiveness of combination therapy among adult patients with asthma. J Allergy Clin Immunol 2006; 118: Cuerq A, Pépin S, Ricordeau P. Remboursement de médicaments antiasthmatiques: une approche de la prévalence et du contrôle de l asthme. [Reimbursement of anti-asthma therapy: an approach to asthma prevalence and level of disease control.] Points de Repère, numéro 24, December Navaratnam P, Jayawant SS, Pedersen CA, et al. Physician adherence to the national asthma prescribing guidelines: evidence from national outpatient survey data in the United States. Ann Allergy Asthma Immunol 2008; 100: Williams LK, Peterson EL, Wells K, et al. Quantifying the proportion of severe asthma exacerbations attributable to inhaled corticosteroid nonadherence. J Allergy Clin Immunol 2011; 128: DOI: /
Adherence to asthma controller medication regimens
 Respiratory Medicine (2005) 99, 1263 1267 Adherence to asthma controller medication regimens D.A. Stempel a,, S.W. Stoloff b, J.R. Carranza Rosenzweig c, R.H. Stanford c, K.L. Ryskina d, A.P. Legorreta
Respiratory Medicine (2005) 99, 1263 1267 Adherence to asthma controller medication regimens D.A. Stempel a,, S.W. Stoloff b, J.R. Carranza Rosenzweig c, R.H. Stanford c, K.L. Ryskina d, A.P. Legorreta
Management of asthma in patients supervised by primary care physicians or by specialists
 Eur Respir J 2006; 27: 42 50 DOI: 10.1183/09031936.06.00035805 CopyrightßERS Journals Ltd 2006 Management of asthma in patients supervised by primary care physicians or by specialists L. Laforest*, E.
Eur Respir J 2006; 27: 42 50 DOI: 10.1183/09031936.06.00035805 CopyrightßERS Journals Ltd 2006 Management of asthma in patients supervised by primary care physicians or by specialists L. Laforest*, E.
Asthma Population Management: Identifying Persistent Asthma, Defining High Risk Asthma, and Measuring Quality of Asthma Care
 Asthma Population Management: Identifying Persistent Asthma, Defining High Risk Asthma, and Measuring Quality of Asthma Care Michael Schatz, MD, MS Allergy Department Kaiser Permanente, San Diego, CA Constructs
Asthma Population Management: Identifying Persistent Asthma, Defining High Risk Asthma, and Measuring Quality of Asthma Care Michael Schatz, MD, MS Allergy Department Kaiser Permanente, San Diego, CA Constructs
Asthma represents a significant burden to the healthcare system.
 n clinical n Predicting Asthma Outcomes in Commercially Insured and Medicaid Populations Richard H. Stanford, PharmD, MS; Manan B. Shah, PharmD, PhD; Anna O. D Souza, BPharm, PhD; and Michael Schatz, MD,
n clinical n Predicting Asthma Outcomes in Commercially Insured and Medicaid Populations Richard H. Stanford, PharmD, MS; Manan B. Shah, PharmD, PhD; Anna O. D Souza, BPharm, PhD; and Michael Schatz, MD,
Four of 10 patients with asthma suffer moderate REVIEW DUAL-CONTROLLER REGIMENS II: OBSERVATIONAL DATA. Michael S. Blaiss, MD ABSTRACT
 DUAL-CONTROLLER REGIMENS II: OBSERVATIONAL DATA Michael S. Blaiss, MD ABSTRACT The differences between clinical trials and clinical practice often create difficulty for generalizing results of controlled
DUAL-CONTROLLER REGIMENS II: OBSERVATIONAL DATA Michael S. Blaiss, MD ABSTRACT The differences between clinical trials and clinical practice often create difficulty for generalizing results of controlled
The National Asthma Education and Prevention Program s
 Long-Acting b-agonist Among Children and Adults With Asthma Elizabeth A. Wasilevich, PhD, MPH; Sarah J. Clark, MPH; Lisa M. Cohn, MS; and Kevin J. Dombkowski, DrPH Managed Care & Healthcare Communications,
Long-Acting b-agonist Among Children and Adults With Asthma Elizabeth A. Wasilevich, PhD, MPH; Sarah J. Clark, MPH; Lisa M. Cohn, MS; and Kevin J. Dombkowski, DrPH Managed Care & Healthcare Communications,
Asthma is a common chronic medical condition that is associated
 Relationship of Asthma Control to Asthma Exacerbations Using Surrogate Markers Within a Managed Care Database Michael Schatz, MD, MS; Robert S. Zeiger, MD, PhD; Su-Jau T. Yang, PhD; Wansu Chen, MS; William
Relationship of Asthma Control to Asthma Exacerbations Using Surrogate Markers Within a Managed Care Database Michael Schatz, MD, MS; Robert S. Zeiger, MD, PhD; Su-Jau T. Yang, PhD; Wansu Chen, MS; William
The prevalence and severity of chronic asthma have
 An Economic Analysis of Alternative Step-Up Therapies in Asthma Patients Receiving Inhaled Daniel A. Ollendorf, MPH, Alyssa S. Pozniak, BA, Brian W. Bowers, PharmD, Gerry Oster PhD ABSTRACT We analyzed
An Economic Analysis of Alternative Step-Up Therapies in Asthma Patients Receiving Inhaled Daniel A. Ollendorf, MPH, Alyssa S. Pozniak, BA, Brian W. Bowers, PharmD, Gerry Oster PhD ABSTRACT We analyzed
Health care education, delivery, and quality. Influence of patients characteristics and disease management on asthma control
 Influence of patients characteristics and disease management on asthma control Laurent Laforest, MD, PhD, a Eric Van Ganse, MD, PhD, a Gilles Devouassoux, MD, PhD, b Jean Bousquet, MD, PhD, c Stephanie
Influence of patients characteristics and disease management on asthma control Laurent Laforest, MD, PhD, a Eric Van Ganse, MD, PhD, a Gilles Devouassoux, MD, PhD, b Jean Bousquet, MD, PhD, c Stephanie
Comprehensive Research Plan: Inhaled corticosteroids + long-acting beta agonists (ICS+LABA) for the treatment of asthma
 Comprehensive Research Plan: Inhaled corticosteroids + long-acting beta agonists (ICS+LABA) for the treatment of asthma Pharmacoepidemiology Unit July 10, 2014 30 Bond Street, Toronto ON, M5B 1W8 www.odprn.ca
Comprehensive Research Plan: Inhaled corticosteroids + long-acting beta agonists (ICS+LABA) for the treatment of asthma Pharmacoepidemiology Unit July 10, 2014 30 Bond Street, Toronto ON, M5B 1W8 www.odprn.ca
SUPPLEMENTAL MATERIALS FOR:
 SUPPLEMENTAL MATERIALS FOR: Dupouy J, Palmaro A, Fatséas M, et al. Mortality associated with time in and out of buprenorphine treatment in French office-based general practice: a 7-year cohort study. Ann
SUPPLEMENTAL MATERIALS FOR: Dupouy J, Palmaro A, Fatséas M, et al. Mortality associated with time in and out of buprenorphine treatment in French office-based general practice: a 7-year cohort study. Ann
T he accurate classification of asthma severity and control is a
 581 ASTHMA Development and validation of database indexes of asthma severity and Faranak Firoozi, Catherine Lemière, Marie-France Beauchesne, Amélie Forget, Lucie Blais... See end of article for authors
581 ASTHMA Development and validation of database indexes of asthma severity and Faranak Firoozi, Catherine Lemière, Marie-France Beauchesne, Amélie Forget, Lucie Blais... See end of article for authors
ASTRO-LAB AN FP7-SUPPORTED PROJECT TO INVESTIGATE THE SAFETY OF THERAPY IN ASTHMA, WITH A FOCUS ON ADHERENCE
 ASTRO-LAB AN FP7-SUPPORTED PROJECT TO INVESTIGATE THE SAFETY OF THERAPY IN ASTHMA, WITH A FOCUS ON ADHERENCE Eric VAN GANSE, Laurent LAFOREST, Idlir LICAJ Pharmacoepidemiology, CHU-Lyon, France ASSESSMENT
ASTRO-LAB AN FP7-SUPPORTED PROJECT TO INVESTIGATE THE SAFETY OF THERAPY IN ASTHMA, WITH A FOCUS ON ADHERENCE Eric VAN GANSE, Laurent LAFOREST, Idlir LICAJ Pharmacoepidemiology, CHU-Lyon, France ASSESSMENT
Comparison of asthma costs in patients starting fluticasone propionate compared to patients starting montelukast
 RESPIRATORY MEDICINE (2001) 95, 227 234 doi:10.1053/rmed.2000.1027, available online at http://www.idealibrary.com on Comparison of asthma costs in patients starting fluticasone propionate compared to
RESPIRATORY MEDICINE (2001) 95, 227 234 doi:10.1053/rmed.2000.1027, available online at http://www.idealibrary.com on Comparison of asthma costs in patients starting fluticasone propionate compared to
The Safety of Asthma and Allergy Medications in Pregnancy: New Horizons
 The Safety of Asthma and Allergy Medications in Pregnancy: New Horizons Jennifer Namazy, MD Division of Allergy Scripps Clinic, San Diego Michael Schatz, MD, MS Department of Allergy Kaiser Permanente,
The Safety of Asthma and Allergy Medications in Pregnancy: New Horizons Jennifer Namazy, MD Division of Allergy Scripps Clinic, San Diego Michael Schatz, MD, MS Department of Allergy Kaiser Permanente,
Title: Real-life use of inhaled corticosteroids in COPD patients vs. GOLD proposals: a paradigm shift in GOLD 2011?
 ERJ Express. Published on October 31, 2013 as doi: 10.1183/09031936.00162313 Letter to the Editor Title: Real-life use of inhaled corticosteroids in COPD patients vs. GOLD proposals: a paradigm shift in
ERJ Express. Published on October 31, 2013 as doi: 10.1183/09031936.00162313 Letter to the Editor Title: Real-life use of inhaled corticosteroids in COPD patients vs. GOLD proposals: a paradigm shift in
Friday and weekend hospital stays: effects on mortality
 ERJ Express. Published on May 14, 2014 as doi: 10.1183/09031936.00007714 ORIGINAL ARTICLE IN PRESS CORRECTED PROOF Friday and weekend hospital stays: effects on mortality Samy Suissa 1,2, Sophie Dell Aniello
ERJ Express. Published on May 14, 2014 as doi: 10.1183/09031936.00007714 ORIGINAL ARTICLE IN PRESS CORRECTED PROOF Friday and weekend hospital stays: effects on mortality Samy Suissa 1,2, Sophie Dell Aniello
Long Term Care Formulary RS -29
 RESTRICTED USE Asthma/COPD Management 1 of 6 PROTOCOL: Asthma Glossary of Medication Acronyms: SABA: short-acting beta agonist (e.g. salbutamol) SABD: short-acting bronchodilator (e.g. ipratropium or SABA)
RESTRICTED USE Asthma/COPD Management 1 of 6 PROTOCOL: Asthma Glossary of Medication Acronyms: SABA: short-acting beta agonist (e.g. salbutamol) SABD: short-acting bronchodilator (e.g. ipratropium or SABA)
Effects on Resource Utilization of Adding Salmeterol in Combination or Separately to Inhaled Corticosteroids
 RESEARCH Effects on Resource Utilization of Adding Salmeterol in Combination or Separately to Inhaled Corticosteroids JAMES CHAN, PharmD, PhD; RITA L. HUI, PharmD, MS; and MICHELE M. SPENCE, PhD ABSTRACT
RESEARCH Effects on Resource Utilization of Adding Salmeterol in Combination or Separately to Inhaled Corticosteroids JAMES CHAN, PharmD, PhD; RITA L. HUI, PharmD, MS; and MICHELE M. SPENCE, PhD ABSTRACT
Surveillance report Published: 6 April 2016 nice.org.uk. NICE All rights reserved.
 Surveillance report 2016 Chronic obstructive pulmonary disease in over 16s: diagnosis and management (2010) NICE guideline CG101 Surveillance report Published: 6 April 2016 nice.org.uk NICE 2016. All rights
Surveillance report 2016 Chronic obstructive pulmonary disease in over 16s: diagnosis and management (2010) NICE guideline CG101 Surveillance report Published: 6 April 2016 nice.org.uk NICE 2016. All rights
BUKSTEIN, MD; ALLAN LUSKIN, MD; SHIVA G. SAJJAN,
 RESEARCH Administrative Claims Analysis of Asthma-Related Health Care Utilization for Patients Who Received Inhaled Corticosteroids With Either Montelukast or Salmeterol as Combination Therapy FELICIA
RESEARCH Administrative Claims Analysis of Asthma-Related Health Care Utilization for Patients Who Received Inhaled Corticosteroids With Either Montelukast or Salmeterol as Combination Therapy FELICIA
Setting The setting was the community. The economic study was carried out in New Jersey, USA.
 Asthma rescue and allergy medication use among asthmatic children with prior allergy prescriptions who initiated asthma controller therapy Luskin A, Bukstein D, Kocevar V S, Yin D D Record Status This
Asthma rescue and allergy medication use among asthmatic children with prior allergy prescriptions who initiated asthma controller therapy Luskin A, Bukstein D, Kocevar V S, Yin D D Record Status This
Assessment of the safety of long-acting β 2 -agonists in routine asthma care: the ASTRO-LAB protocol
 www.nature.com/npjpcrm All rights reserved 2055-1010/15 PROTOCOL OPEN Assessment of the safety of long-acting β 2 -agonists in routine asthma care: the ASTRO-LAB protocol Eric Van Ganse 1,2, Nathalie Texier
www.nature.com/npjpcrm All rights reserved 2055-1010/15 PROTOCOL OPEN Assessment of the safety of long-acting β 2 -agonists in routine asthma care: the ASTRO-LAB protocol Eric Van Ganse 1,2, Nathalie Texier
Ethnic disparities in asthma treatment and outcomes in children aged under 15 years in New Zealand: analysis of national databases
 Prim Care Respir J 2013; 22(3): 312-318 RESEARCH PAPER Ethnic disparities in asthma treatment and outcomes in children aged under 15 years in New Zealand: analysis of national databases Todd D Gillies
Prim Care Respir J 2013; 22(3): 312-318 RESEARCH PAPER Ethnic disparities in asthma treatment and outcomes in children aged under 15 years in New Zealand: analysis of national databases Todd D Gillies
Optum Research Database Proportion of asthma patients with prescription fills for rescue and controller medications by year,
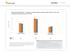 ASTHMA CHART 1 Optum Research Database Proportion of asthma patients with prescription fills for rescue and controller medications by year, 215-216 7 6 5 4 3 2 1 1 Controller medication 1 Rescue medication
ASTHMA CHART 1 Optum Research Database Proportion of asthma patients with prescription fills for rescue and controller medications by year, 215-216 7 6 5 4 3 2 1 1 Controller medication 1 Rescue medication
I nhaled corticosteroids are the treatment of choice in the
 880 ORIGINAL ARTICLE Regular use of inhaled corticosteroids and the long term prevention of hospitalisation for asthma S Suissa, P Ernst, A Kezouh... See end of article for authors affiliations... Correspondence
880 ORIGINAL ARTICLE Regular use of inhaled corticosteroids and the long term prevention of hospitalisation for asthma S Suissa, P Ernst, A Kezouh... See end of article for authors affiliations... Correspondence
Process quality measures and asthma exacerbations in the Medicaid population
 Process quality measures and asthma exacerbations in the Medicaid population Pierre L. Yong, MD, MPH, a,b,c,d,e and Rachel M. Werner, MD, PhD a,d Philadelphia, Pa Background: Asthma quality assessment
Process quality measures and asthma exacerbations in the Medicaid population Pierre L. Yong, MD, MPH, a,b,c,d,e and Rachel M. Werner, MD, PhD a,d Philadelphia, Pa Background: Asthma quality assessment
Randomized controlled trial of adherence with single or combination inhaled corticosteroid/long-acting b-agonist inhaler therapy in asthma
 Randomized controlled trial of adherence with single or combination inhaled corticosteroid/long-acting b-agonist inhaler therapy in asthma Kyle Perrin, MBChB, FRACP, a,b Mathew Williams, Dip Ex Sci, a
Randomized controlled trial of adherence with single or combination inhaled corticosteroid/long-acting b-agonist inhaler therapy in asthma Kyle Perrin, MBChB, FRACP, a,b Mathew Williams, Dip Ex Sci, a
Meta-analysis of the risk of mortality with salmeterol and the effect of concomitant inhaled corticosteroid therapy
 c Additional details are published online only at http:// thorax.bmj.com/content/vol65/ issue1 1 University of Otago Wellington, Wellington, New Zealand; 2 Capital & Coast District Health Board, Wellington,
c Additional details are published online only at http:// thorax.bmj.com/content/vol65/ issue1 1 University of Otago Wellington, Wellington, New Zealand; 2 Capital & Coast District Health Board, Wellington,
Evaluation of Asthma Management in Middle EAst North Africa Adult population
 STUDY REPORT SUMMARY Evaluation of Asthma Management in Middle EAst North Africa Adult population Descriptive study on the management of asthma in an asthmatic Middle East Africa adult population Background/Rationale:
STUDY REPORT SUMMARY Evaluation of Asthma Management in Middle EAst North Africa Adult population Descriptive study on the management of asthma in an asthmatic Middle East Africa adult population Background/Rationale:
Outcomes: Initially, our primary definitions of pneumonia was severe pneumonia, where the subject was hospitalized
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
Drug prescriptions (Pharm) Exposure (36/48 months)
 ANNEX SECTION PART A - Study design: Figure 1 overview of the study design Drug prescriptions (Pharm) 2006-2010 Exposure (36/48 months) flexible time windows (e.g. 90 days) time index date (hospital discharge)
ANNEX SECTION PART A - Study design: Figure 1 overview of the study design Drug prescriptions (Pharm) 2006-2010 Exposure (36/48 months) flexible time windows (e.g. 90 days) time index date (hospital discharge)
Online supplementary material
 Online supplementary material Add-on long-acting β2-agonist (LABA) in a separate inhaler as asthma step-up therapy versus increased dose of inhaled corticosteroid (ICS) or ICS/LABA combination inhaler
Online supplementary material Add-on long-acting β2-agonist (LABA) in a separate inhaler as asthma step-up therapy versus increased dose of inhaled corticosteroid (ICS) or ICS/LABA combination inhaler
Asthma and Adherence to Inhaled Corticosteroids: Current Status and Future Perspectives
 Asthma and Adherence to Inhaled Corticosteroids: Current Status and Future Perspectives Camilla Boslev Bårnes MD and Charlotte Suppli Ulrik MD DMSc Introduction Methods Review of the Literature Adherence
Asthma and Adherence to Inhaled Corticosteroids: Current Status and Future Perspectives Camilla Boslev Bårnes MD and Charlotte Suppli Ulrik MD DMSc Introduction Methods Review of the Literature Adherence
#1 cause of school absenteeism in children 13 million missed days annually
 Asthma Update 2013 Jennifer W. McCallister, MD, FACP, FCCP Associate Professor Pulmonary & Critical Care Medicine The Ohio State University Wexner Medical Center Disclosures None 2 Objectives Review burden
Asthma Update 2013 Jennifer W. McCallister, MD, FACP, FCCP Associate Professor Pulmonary & Critical Care Medicine The Ohio State University Wexner Medical Center Disclosures None 2 Objectives Review burden
Global Initiative for Asthma (GINA) What s new in GINA 2017?
 Global Initiative for Asthma (GINA) GINA Global Strategy for Asthma Management and Prevention Asthma-COPD overlap The word syndrome has been removed from the previous term asthma-copd overlap syndrome
Global Initiative for Asthma (GINA) GINA Global Strategy for Asthma Management and Prevention Asthma-COPD overlap The word syndrome has been removed from the previous term asthma-copd overlap syndrome
Dr Christopher Worsnop
 Dr Christopher Worsnop Respiratory & Sleep Physician Austin Hospital, Melbourne Supported by: Top Tips in Modern Asthma Management Dr Christopher Worsnop Rotorua GPCME Meeting June 2013 Speaker declaration
Dr Christopher Worsnop Respiratory & Sleep Physician Austin Hospital, Melbourne Supported by: Top Tips in Modern Asthma Management Dr Christopher Worsnop Rotorua GPCME Meeting June 2013 Speaker declaration
Breakfast Session Prof Neil Barnes Professor of Respiratory Medicine London Chest Hospital & The Royal London Hospital United Kingdom
 Breakfast Session Prof Neil Barnes Professor of Respiratory Medicine London Chest Hospital & The Royal London Hospital United Kingdom 2 BEYOND SYMPTOMS ADDRESSING FUTURE RISK IN ASTHMA South GP CME 2013,
Breakfast Session Prof Neil Barnes Professor of Respiratory Medicine London Chest Hospital & The Royal London Hospital United Kingdom 2 BEYOND SYMPTOMS ADDRESSING FUTURE RISK IN ASTHMA South GP CME 2013,
April 10 th, Bond Street, Toronto ON, M5B 1W8
 Comprehensive Research Plan: Inhaled long-acting muscarinic antagonists (LAMAs; long-acting anticholinergics) for the treatment of chronic obstructive pulmonary disease (COPD) April 10 th, 2014 30 Bond
Comprehensive Research Plan: Inhaled long-acting muscarinic antagonists (LAMAs; long-acting anticholinergics) for the treatment of chronic obstructive pulmonary disease (COPD) April 10 th, 2014 30 Bond
INHALED AND NASAL CORTICOSTEROID USE AND THE RISK OF FRACTURE. Online data supplement.
 INHALED AND NASAL CORTICOSTEROID USE AND THE RISK OF FRACTURE. Online data supplement. Samy Suissa, PhD, Marc Baltzan, MD, MSc, Richard Kremer, MD, Pierre Ernst, MD, MSc 26 METHODS Source of data We used
INHALED AND NASAL CORTICOSTEROID USE AND THE RISK OF FRACTURE. Online data supplement. Samy Suissa, PhD, Marc Baltzan, MD, MSc, Richard Kremer, MD, Pierre Ernst, MD, MSc 26 METHODS Source of data We used
Patients with chronic obstructive pulmonary
 HEALTH ECONOMICS Delivery of Ipratropium and Albuterol Combination Therapy for Chronic Obstructive Pulmonary Disease: Effectiveness of a Two-in-one Inhaler Versus Separate Inhalers Elizabeth Chrischilles,
HEALTH ECONOMICS Delivery of Ipratropium and Albuterol Combination Therapy for Chronic Obstructive Pulmonary Disease: Effectiveness of a Two-in-one Inhaler Versus Separate Inhalers Elizabeth Chrischilles,
One-year claims analysis comparing inhaled fluticasone propionate with zafirlukast for the treatment of asthma
 One-year claims analysis comparing inhaled fluticasone propionate with zafirlukast for the treatment of asthma David A. Stempel, MD, a Jay W. Meyer, PhD, b Richard H. Stanford, PharmD, MS, c and Steve
One-year claims analysis comparing inhaled fluticasone propionate with zafirlukast for the treatment of asthma David A. Stempel, MD, a Jay W. Meyer, PhD, b Richard H. Stanford, PharmD, MS, c and Steve
Policy Evaluation: Step Therapy Prior Authorization of Combination Inhaled Corticosteroid / Long-Acting Beta-Agonists
 Drug Use Research & Management Program OHA Division of Medical Assistance Programs 500 Summer Street NE, E35; Salem, OR 97301-1079 Phone 503-947-5220 Fax 503-947-1119 Policy Evaluation: Step Therapy Prior
Drug Use Research & Management Program OHA Division of Medical Assistance Programs 500 Summer Street NE, E35; Salem, OR 97301-1079 Phone 503-947-5220 Fax 503-947-1119 Policy Evaluation: Step Therapy Prior
NG80. Asthma: diagnosis, monitoring and chronic asthma management (NG80)
 Asthma: diagnosis, monitoring and chronic asthma management (NG80) NG80 NICE has checked the use of its content in this product and the sponsor has had no influence on the content of this booklet. NICE
Asthma: diagnosis, monitoring and chronic asthma management (NG80) NG80 NICE has checked the use of its content in this product and the sponsor has had no influence on the content of this booklet. NICE
Research in Real Life
 Research in Real Life Study 1: Exploratory study - identifying the benefits of pmdi versus Diskus for delivering fluticasone/salmeterol combination therapy in patients with chronic obstructive pulmonary
Research in Real Life Study 1: Exploratory study - identifying the benefits of pmdi versus Diskus for delivering fluticasone/salmeterol combination therapy in patients with chronic obstructive pulmonary
Learning the Asthma Guidelines by Case Studies
 Learning the Asthma Guidelines by Case Studies Timothy Craig, DO Professor of Medicine and Pediatrics Distinguished Educator Penn State University Hershey Medical Center Objectives 1. Learn the Asthma
Learning the Asthma Guidelines by Case Studies Timothy Craig, DO Professor of Medicine and Pediatrics Distinguished Educator Penn State University Hershey Medical Center Objectives 1. Learn the Asthma
CHAPTER 5. Weekly self-monitoring and treatment adjustment benefit patients with partly controlled and uncontrolled asthma
 CHAPTER 5 Weekly self-monitoring and treatment adjustment benefit patients with partly controlled and uncontrolled asthma Victor van der Meer, Henk F. van Stel, Moira J. Bakker, Albert C. Roldaan, Willem
CHAPTER 5 Weekly self-monitoring and treatment adjustment benefit patients with partly controlled and uncontrolled asthma Victor van der Meer, Henk F. van Stel, Moira J. Bakker, Albert C. Roldaan, Willem
Interventions to improve adherence to inhaled steroids for asthma. Respiratory department
 Interventions to improve adherence to inhaled steroids for asthma Respiratory department Content Overview Research References Overview Asthma is a chronic breathing condition that affects more than 300
Interventions to improve adherence to inhaled steroids for asthma Respiratory department Content Overview Research References Overview Asthma is a chronic breathing condition that affects more than 300
Research in real life ltd study protocol for Teva Pharmaceutical Industries Ltd.
 Research in real life ltd study protocol for Teva Pharmaceutical Industries Ltd. The use of a spacer in the delivery of large (Fluticasone Propionate) and small particle FP and Qvar Spacer vs Non-Spacer
Research in real life ltd study protocol for Teva Pharmaceutical Industries Ltd. The use of a spacer in the delivery of large (Fluticasone Propionate) and small particle FP and Qvar Spacer vs Non-Spacer
Safety of β2-agonists in asthma
 Safety of β2-agonists in asthma Sanjeeva Dissanayake IPAC-RS/UF Orlando Inhalation Conference March 20, 2014 Overview Origin of concerns Mechanistic hypotheses Large scale clinical datasets Interpretation
Safety of β2-agonists in asthma Sanjeeva Dissanayake IPAC-RS/UF Orlando Inhalation Conference March 20, 2014 Overview Origin of concerns Mechanistic hypotheses Large scale clinical datasets Interpretation
TORCH: Salmeterol and Fluticasone Propionate and Survival in COPD
 TORCH: and Propionate and Survival in COPD April 19, 2007 Justin Lee Pharmacy Resident University Health Network Outline Overview of COPD Pathophysiology Pharmacological Treatment Overview of the TORCH
TORCH: and Propionate and Survival in COPD April 19, 2007 Justin Lee Pharmacy Resident University Health Network Outline Overview of COPD Pathophysiology Pharmacological Treatment Overview of the TORCH
THIS TALK STATINS REDUCE: Immortal time bias Immeasurable time bias Time-window bias TIME-RELATED BIASES IN PHARMACOEPIDEMIOLOGY
 TIME-RELATED BIASES IN PHARMACOEPIDEMIOLOGY Samy Suissa McGill University and Jewish General Hospital Montreal, Canada ISPE mid-year meeting, Miami April 22, 2012 STATINS REDUCE: JAMA 2000: Fracture rate
TIME-RELATED BIASES IN PHARMACOEPIDEMIOLOGY Samy Suissa McGill University and Jewish General Hospital Montreal, Canada ISPE mid-year meeting, Miami April 22, 2012 STATINS REDUCE: JAMA 2000: Fracture rate
BMJ Open. Healthcare utilization by pregnant asthma patients in South Korea: a cohort study using a nationwide claims data
 Healthcare utilization by pregnant asthma patients in South Korea: a cohort study using a nationwide claims data Journal: Manuscript ID: bmjopen-0-00 Article Type: Research Date Submitted by the Author:
Healthcare utilization by pregnant asthma patients in South Korea: a cohort study using a nationwide claims data Journal: Manuscript ID: bmjopen-0-00 Article Type: Research Date Submitted by the Author:
GINA. At-A-Glance Asthma Management Reference. for adults, adolescents and children 6 11 years. Updated 2017
 GINA At-A-Glance Asthma Management Reference for adults, adolescents and children 6 11 years Updated 2017 This resource should be used in conjunction with the Global Strategy for Asthma Management and
GINA At-A-Glance Asthma Management Reference for adults, adolescents and children 6 11 years Updated 2017 This resource should be used in conjunction with the Global Strategy for Asthma Management and
Title: Correlates of quality of life of pre-obese and obese patients: a pharmacy-based cross-sectional survey
 Author's response to reviews Title: Correlates of quality of life of pre-obese and obese patients: a pharmacy-based cross-sectional survey Authors: Laurent Laforest (laurent.laforest@chu-lyon.fr) Eric
Author's response to reviews Title: Correlates of quality of life of pre-obese and obese patients: a pharmacy-based cross-sectional survey Authors: Laurent Laforest (laurent.laforest@chu-lyon.fr) Eric
Research Article Effect of Pregnancy on Quantitative Medication Use and Relation to Exacerbations in Asthma
 Hindawi BioMed Research International Volume 2017, Article ID 8276190, 15 pages https://doi.org/10.1155/2017/8276190 Research Article Effect of Pregnancy on Quantitative Medication Use and Relation to
Hindawi BioMed Research International Volume 2017, Article ID 8276190, 15 pages https://doi.org/10.1155/2017/8276190 Research Article Effect of Pregnancy on Quantitative Medication Use and Relation to
Putting NICE guidance into practice. Resource impact report: Asthma: diagnosis, monitoring and chronic asthma management (NG80)
 Putting NICE guidance into practice Resource impact report: Asthma: diagnosis, monitoring and chronic asthma management (NG80) Published: November 2017 Summary This report focuses on the recommendations
Putting NICE guidance into practice Resource impact report: Asthma: diagnosis, monitoring and chronic asthma management (NG80) Published: November 2017 Summary This report focuses on the recommendations
CME Workshop. Q&A on Asthma Control. What is asthma control? Naomi s asthma. How is asthma control assessed?
 Focus on CME at the University of British Columbia CME Workshop Q&A on Control Louis-Philippe Boulet, MD, FRCPC Presented at the World Organization, and at UBC s 4th Annual Advances in Respiratory and
Focus on CME at the University of British Columbia CME Workshop Q&A on Control Louis-Philippe Boulet, MD, FRCPC Presented at the World Organization, and at UBC s 4th Annual Advances in Respiratory and
Are young adults with asthma treated sufficiently with inhaled steroids? A population-based study of prescription data from 1991 and 1994
 Br J Clin Pharmacol 1996; 41: 285 289 Are young adults with asthma treated sufficiently with inhaled steroids? A population-based study of prescription data from 1991 and 1994 DAVID GAIST1, JESPER HALLAS1,
Br J Clin Pharmacol 1996; 41: 285 289 Are young adults with asthma treated sufficiently with inhaled steroids? A population-based study of prescription data from 1991 and 1994 DAVID GAIST1, JESPER HALLAS1,
Improving asthma outcomes in large populations
 Current perspectives Improving asthma outcomes in large populations Michael Schatz, MD, MS, and Robert S. Zeiger, MD, PhD San Diego, Calif This article summarizes our experience using administrative, survey,
Current perspectives Improving asthma outcomes in large populations Michael Schatz, MD, MS, and Robert S. Zeiger, MD, PhD San Diego, Calif This article summarizes our experience using administrative, survey,
Estimated date of first patient enrolled Q III/IV Estimated date of last patient completed Q3 2015
 PROTOCOL SYNOPSIS A 26 week, randomized, double-blind, parallel-group, active controlled, multicenter, multinational safety study evaluating the risk of serious asthma-related events during treatment with
PROTOCOL SYNOPSIS A 26 week, randomized, double-blind, parallel-group, active controlled, multicenter, multinational safety study evaluating the risk of serious asthma-related events during treatment with
National Trends in Ambulatory Asthma Treatment,
 National Trends in Ambulatory Asthma Treatment, 1997 2009 Ashley Higashi, MPH 1, Shu Zhu, MPH 1, Randall S. Stafford, MD, PhD 2, and G. Caleb Alexander, MD, MS 1,3,4,5 1 Section of General Internal Medicine,
National Trends in Ambulatory Asthma Treatment, 1997 2009 Ashley Higashi, MPH 1, Shu Zhu, MPH 1, Randall S. Stafford, MD, PhD 2, and G. Caleb Alexander, MD, MS 1,3,4,5 1 Section of General Internal Medicine,
(Asthma) Diagnosis, monitoring and chronic asthma management
 Dubai Standards of Care 2018 (Asthma) Diagnosis, monitoring and chronic asthma management Preface Asthma is one of the most common problem dealt with in daily practice. In Dubai, the management of chronic
Dubai Standards of Care 2018 (Asthma) Diagnosis, monitoring and chronic asthma management Preface Asthma is one of the most common problem dealt with in daily practice. In Dubai, the management of chronic
Predicting, Preventing and Managing Asthma Exacerbations. Heather Zar Department of Paediatrics & Child Health University of Cape Town South Africa
 Predicting, Preventing and Managing Asthma Exacerbations Heather Zar Department of Paediatrics & Child Health University of Cape Town South Africa Asthma exacerbations Predicting exacerbation recognising
Predicting, Preventing and Managing Asthma Exacerbations Heather Zar Department of Paediatrics & Child Health University of Cape Town South Africa Asthma exacerbations Predicting exacerbation recognising
New TLD Indication: Asthma RELIEF-1 Study. Nick ten Hacken, MD Study Co-Principal Investigator
 New TLD Indication: Asthma RELIEF-1 Study Nick ten Hacken, MD Study Co-Principal Investigator The Burden of Asthma WW Adult Asthma Prevalence 230.5 Million Mild/Intermittent 161.9 Million (70%) Moderate
New TLD Indication: Asthma RELIEF-1 Study Nick ten Hacken, MD Study Co-Principal Investigator The Burden of Asthma WW Adult Asthma Prevalence 230.5 Million Mild/Intermittent 161.9 Million (70%) Moderate
Do We Need Biologics in Pediatric Asthma Management?
 Do We Need Biologics in Pediatric Asthma Management? Ting Fan LEUNG, MBChB, MD, FRCPCH, FAAAAI Professor and Chairman Department of Paediatrics The Chinese University of Hong Kong Asthma and Allergy by
Do We Need Biologics in Pediatric Asthma Management? Ting Fan LEUNG, MBChB, MD, FRCPCH, FAAAAI Professor and Chairman Department of Paediatrics The Chinese University of Hong Kong Asthma and Allergy by
Asthma Among Minnesota Health Care Program Beneficiaries
 Asthma Among Minnesota Health Care Program Beneficiaries A JOINT REPORT FROM THE MINNESOTA DEPARTMENT OF HEALTH AND THE MINNESOTA DEPARTMENT OF HUMAN SERVICES November 2018 Asthma Among Minnesota Health
Asthma Among Minnesota Health Care Program Beneficiaries A JOINT REPORT FROM THE MINNESOTA DEPARTMENT OF HEALTH AND THE MINNESOTA DEPARTMENT OF HUMAN SERVICES November 2018 Asthma Among Minnesota Health
Kirthi Gunasekera MD Respiratory Physician National Hospital of Sri Lanka Colombo,
 Kirthi Gunasekera MD Respiratory Physician National Hospital of Sri Lanka Colombo, BRONCHODILATORS: Beta Adrenoreceptor Agonists Actions Adrenoreceptor agonists have many of the same actions as epinephrine/adrenaline,
Kirthi Gunasekera MD Respiratory Physician National Hospital of Sri Lanka Colombo, BRONCHODILATORS: Beta Adrenoreceptor Agonists Actions Adrenoreceptor agonists have many of the same actions as epinephrine/adrenaline,
The Acute & Maintenance Treatment of Asthma via Aerosolized Medications
 The Acute & Maintenance Treatment of Asthma via Aerosolized Medications Douglas S. Gardenhire, EdD, RRT-NPS, FAARC Associate Professor and Chairman Department of Respiratory Therapy Objectives Define Asthma.
The Acute & Maintenance Treatment of Asthma via Aerosolized Medications Douglas S. Gardenhire, EdD, RRT-NPS, FAARC Associate Professor and Chairman Department of Respiratory Therapy Objectives Define Asthma.
Clinical efficacy of montelukast in anti-inflammatory treatment of asthma and allergic rhinitis
 Clinical efficacy of montelukast in anti-inflammatory treatment of asthma and allergic rhinitis Kim Hyun Hee, MD, PhD. Dept. of Pediatrics The Catholic University of Korea College of Medicine Achieving
Clinical efficacy of montelukast in anti-inflammatory treatment of asthma and allergic rhinitis Kim Hyun Hee, MD, PhD. Dept. of Pediatrics The Catholic University of Korea College of Medicine Achieving
Study Exposures, Outcomes:
 GSK Medicine: Coreg IR, Coreg CR, and InnoPran Study No.: WWE111944/WEUSRTP3149 Title: A nested case-control study of the association between Coreg IR and Coreg CR and hypersensitivity reactions: anaphylactic
GSK Medicine: Coreg IR, Coreg CR, and InnoPran Study No.: WWE111944/WEUSRTP3149 Title: A nested case-control study of the association between Coreg IR and Coreg CR and hypersensitivity reactions: anaphylactic
Original Article. ARTICLE INFO Article history: Received 9 September 2017 Received in revised form 19 November 2017 Accepted 20 November 2017
 Original Article Pharm Sci Asia 2018; 45 (1), 13-21 DOI : 10.29090/psa.2018.01.013 Investigation on medication use and factors associated with levels of asthma control among asthmatic outpatients at University
Original Article Pharm Sci Asia 2018; 45 (1), 13-21 DOI : 10.29090/psa.2018.01.013 Investigation on medication use and factors associated with levels of asthma control among asthmatic outpatients at University
Methods to Diagnose Adherence Status
 Methods to Diagnose Adherence Status March 4, 2014 Andrew G Weinstein MD Associate Clinical Professor Pediatrics Jefferson Medical College President, Adherence Management Systems Disclosures President,
Methods to Diagnose Adherence Status March 4, 2014 Andrew G Weinstein MD Associate Clinical Professor Pediatrics Jefferson Medical College President, Adherence Management Systems Disclosures President,
GSK Medicine: salmeterol, Salmeterol+Fluticasone proprionate, fluticasone propionate, beclomethasone
 GSK Medicine: salmeterol, Salmeterol+Fluticasone proprionate, fluticasone propionate, beclomethasone Study No.: WWE111984/WEUSRTP2640/EPI40528 Title: The Asthma Death Case Control Study (ADCCS): Association
GSK Medicine: salmeterol, Salmeterol+Fluticasone proprionate, fluticasone propionate, beclomethasone Study No.: WWE111984/WEUSRTP2640/EPI40528 Title: The Asthma Death Case Control Study (ADCCS): Association
Asthma is a highly prevalent and costly
 Asthma Treatment Guidelines: How Do We Measure Up? Robert P. Navarro, PharmD Abstract The use of clinical guidelines for the management of asthma can help improve patient outcomes and control costs. This
Asthma Treatment Guidelines: How Do We Measure Up? Robert P. Navarro, PharmD Abstract The use of clinical guidelines for the management of asthma can help improve patient outcomes and control costs. This
Methodological Framework and Tools for Cross-National comparisons of Drug Utilization Data
 Methodological Framework and Tools for Cross-National comparisons of Drug Utilization Data Vander Stichele RH, MD, PhD Heymans Institute of Pharmacology, Ghent University, Belgium Summer School EACPT August
Methodological Framework and Tools for Cross-National comparisons of Drug Utilization Data Vander Stichele RH, MD, PhD Heymans Institute of Pharmacology, Ghent University, Belgium Summer School EACPT August
Global Initiative for Asthma (GINA) What s new in GINA 2016?
 Global Initiative for Asthma (GINA) What s new in GINA 2016? GINA Global Strategy for Asthma Management and Prevention GINA: A Brief History Established in 1993 Collaboration between NHLBI and WHO Multiple
Global Initiative for Asthma (GINA) What s new in GINA 2016? GINA Global Strategy for Asthma Management and Prevention GINA: A Brief History Established in 1993 Collaboration between NHLBI and WHO Multiple
The distribution of COPD in UK general practice using the new GOLD classification
 ORIGINAL ARTICLE COPD The distribution of COPD in UK general practice using the new GOLD classification John Haughney 1, Kevin Gruffydd-Jones 2, June Roberts 3, Amanda J. Lee 4, Alison Hardwell 5 and Lorcan
ORIGINAL ARTICLE COPD The distribution of COPD in UK general practice using the new GOLD classification John Haughney 1, Kevin Gruffydd-Jones 2, June Roberts 3, Amanda J. Lee 4, Alison Hardwell 5 and Lorcan
THE USE OF LONG-ACTING β 2 -AGONISTS AS MONOTHERAPY IN CHILDREN AND ADULTS
 THE USE OF LONG-ACTING β 2 -AGONISTS AS MONOTHERAPY IN CHILDREN AND ADULTS Pat G Camp 1,2, Tingting Zhang 3, M Anne Smith 3, Bruce C Carleton 3 1 UBC James Hogg Research Centre and the Institute for Heart
THE USE OF LONG-ACTING β 2 -AGONISTS AS MONOTHERAPY IN CHILDREN AND ADULTS Pat G Camp 1,2, Tingting Zhang 3, M Anne Smith 3, Bruce C Carleton 3 1 UBC James Hogg Research Centre and the Institute for Heart
Case-Compare Impact Report
 Case-Compare Impact Report October 8, 20 For CME Activity: Developed through an independent educational grant from Genentech: Moderate to Severe Persistent Asthma: A Case-Based Panel Discussion (March
Case-Compare Impact Report October 8, 20 For CME Activity: Developed through an independent educational grant from Genentech: Moderate to Severe Persistent Asthma: A Case-Based Panel Discussion (March
LAMA Products for the Treatment of COPD
 FINAL REPORT LAMA Products for the Treatment of COPD Pharmacoepidemiology Unit: Censored Final Report Tara Gomes, Andrea Gershon, Matthew Stanbrook, Ximena Camacho, Diana Martins, Samantha Singh, Zhan
FINAL REPORT LAMA Products for the Treatment of COPD Pharmacoepidemiology Unit: Censored Final Report Tara Gomes, Andrea Gershon, Matthew Stanbrook, Ximena Camacho, Diana Martins, Samantha Singh, Zhan
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Fasenra) Reference Number: CP.PHAR.## Effective Date: 01.16.18 Last Review Date: 05.18 Line of Business: Commercial, Medicaid Revision Log See Important Reminder at the end of this policy
Clinical Policy: (Fasenra) Reference Number: CP.PHAR.## Effective Date: 01.16.18 Last Review Date: 05.18 Line of Business: Commercial, Medicaid Revision Log See Important Reminder at the end of this policy
Australian Asthma Handbook. Key table and figures Version 1.2
 Australian Asthma Handbook Key table and figures Version 1.2 DIAGNOSIS OF ASTHMA Figure. Steps in the diagnosis of asthma in adults Table. Findings that increase or decrease the probability of asthma in
Australian Asthma Handbook Key table and figures Version 1.2 DIAGNOSIS OF ASTHMA Figure. Steps in the diagnosis of asthma in adults Table. Findings that increase or decrease the probability of asthma in
aclidinium 322 micrograms inhalation powder (Eklira Genuair ) SMC No. (810/12) Almirall S.A.
 aclidinium 322 micrograms inhalation powder (Eklira Genuair ) SMC No. (810/12) Almirall S.A. 05 October 2012 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product and
aclidinium 322 micrograms inhalation powder (Eklira Genuair ) SMC No. (810/12) Almirall S.A. 05 October 2012 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product and
Journal of the COPD Foundation. Journal Club. Chronic Obstructive Pulmonary Diseases:
 85 Journal Club Chronic Obstructive Pulmonary Diseases: Journal of the COPD Foundation Journal Club Ron Balkissoon, MD, MSc, DIH, FRCPC 1 Abbreviations: inhaled corticosteroid, ICS; long acting β 2 -agonist,
85 Journal Club Chronic Obstructive Pulmonary Diseases: Journal of the COPD Foundation Journal Club Ron Balkissoon, MD, MSc, DIH, FRCPC 1 Abbreviations: inhaled corticosteroid, ICS; long acting β 2 -agonist,
Healthy People 2010 Asthma Objectives December 2009 Update
 December 2009 Update Healthy People 2010 is a set of national health goals focusing on disease prevention and health promotion to be reached by the year 2010. The following are the Healthy People 2010
December 2009 Update Healthy People 2010 is a set of national health goals focusing on disease prevention and health promotion to be reached by the year 2010. The following are the Healthy People 2010
WEBINAR. Difficult-to-treat and severe asthma: changing the paradigm
 WEBINAR Difficult-to-treat and severe asthma: changing the paradigm A multidisciplinary discussion on new therapies, and how to identify and manage difficult-to-treat and severe asthma DIFFICULT-TO-TREAT
WEBINAR Difficult-to-treat and severe asthma: changing the paradigm A multidisciplinary discussion on new therapies, and how to identify and manage difficult-to-treat and severe asthma DIFFICULT-TO-TREAT
Inhaled Corticosteroid vs. Add-On Long-Acting Beta-Agonist for Step-Up Therapy in Asthma
 Online Data Supplement Inhaled Corticosteroid vs. Add-On Long-Acting Beta-Agonist for Step-Up Therapy in Asthma Elliot Israel, Nicolas Roche, Richard J. Martin, Gene Colice, Paul M. Dorinsky, Dirkje S.
Online Data Supplement Inhaled Corticosteroid vs. Add-On Long-Acting Beta-Agonist for Step-Up Therapy in Asthma Elliot Israel, Nicolas Roche, Richard J. Martin, Gene Colice, Paul M. Dorinsky, Dirkje S.
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Nucala) Reference Number: CP.PHAR.200 Effective Date: 04.01.16 Last Review Date: 02.18 Line of Business: Commercial, Medicaid Coding Implications Revision Log See Important Reminder at
Clinical Policy: (Nucala) Reference Number: CP.PHAR.200 Effective Date: 04.01.16 Last Review Date: 02.18 Line of Business: Commercial, Medicaid Coding Implications Revision Log See Important Reminder at
Improved Transparency in Key Operational Decisions in Real World Evidence
 PharmaSUG 2018 - Paper RW-06 Improved Transparency in Key Operational Decisions in Real World Evidence Rebecca Levin, Irene Cosmatos, Jamie Reifsnyder United BioSource Corp. ABSTRACT The joint International
PharmaSUG 2018 - Paper RW-06 Improved Transparency in Key Operational Decisions in Real World Evidence Rebecca Levin, Irene Cosmatos, Jamie Reifsnyder United BioSource Corp. ABSTRACT The joint International
The New England Journal of Medicine LOW-DOSE INHALED CORTICOSTEROIDS AND THE PREVENTION OF DEATH FROM ASTHMA
 LOW-DOSE INHALED CORTICOSTEROIDS AND THE PREVENTION OF DEATH FROM ASTHMA SAMY SUISSA, PH.D., PIERRE ERNST, M.D., SERGE BENAYOUN, M.SC., MARC BALTZAN, M.D., AND BING CAI, M.SC. ABSTRACT Background Although
LOW-DOSE INHALED CORTICOSTEROIDS AND THE PREVENTION OF DEATH FROM ASTHMA SAMY SUISSA, PH.D., PIERRE ERNST, M.D., SERGE BENAYOUN, M.SC., MARC BALTZAN, M.D., AND BING CAI, M.SC. ABSTRACT Background Although
Dual-Controller Asthma Therapy: Rationale and Clinical Benefits
 B/1 Dual-Controller Asthma Therapy: Rationale and Clinical Benefits MODULE B The 1997 National Heart, Lung, and Blood Institute (NHLBI) Expert Panel guidelines on asthma management recommend a 4-step approach
B/1 Dual-Controller Asthma Therapy: Rationale and Clinical Benefits MODULE B The 1997 National Heart, Lung, and Blood Institute (NHLBI) Expert Panel guidelines on asthma management recommend a 4-step approach
Pharmacoeconomic Analysis of Asthma in Pediatric Patients in Tertiary Care Hospital in Kerala
 Indian Journal of Pharmacy Practice Association of Pharmaceutical Teachers of India Pharmacoeconomic Analysis of Asthma in Pediatric Patients in Tertiary Care Hospital in Kerala Stejin J*, Kishor KV, Nandha
Indian Journal of Pharmacy Practice Association of Pharmaceutical Teachers of India Pharmacoeconomic Analysis of Asthma in Pediatric Patients in Tertiary Care Hospital in Kerala Stejin J*, Kishor KV, Nandha
Medicine OBSERVATIONAL STUDY
 Medicine OBSERVATIONAL STUDY Recurrent Wheezing in Infants A Population-Based Study Manon Belhassen, MSc, Jacques De Blic, MD, PhD, Laurent Laforest, MD, PhD, Valérie Laigle, PharmD, Céline Chanut-Vogel,
Medicine OBSERVATIONAL STUDY Recurrent Wheezing in Infants A Population-Based Study Manon Belhassen, MSc, Jacques De Blic, MD, PhD, Laurent Laforest, MD, PhD, Valérie Laigle, PharmD, Céline Chanut-Vogel,
Rapid Effects of Inhaled Corticosteroids in Acute Asthma Gustavo J. Rodrigo, MD.
 Rapid Effects of Inhaled Corticosteroids in Acute Asthma Gustavo J. Rodrigo, MD WWW.chestjournal.org Background: Current reviews on the use of inhaled corticosteroids (ICS) for acute asthma underestimated
Rapid Effects of Inhaled Corticosteroids in Acute Asthma Gustavo J. Rodrigo, MD WWW.chestjournal.org Background: Current reviews on the use of inhaled corticosteroids (ICS) for acute asthma underestimated
Pulmonary and Critical Care
 Pulmonary and Critical Care 2015-2016 DRAFT REPORT FOR COMMENT April 21, 2016 This report is funded by the Department of Health and Human Services under contract HHSM-500-2012-00009I Task Order HHSM-500-T0000
Pulmonary and Critical Care 2015-2016 DRAFT REPORT FOR COMMENT April 21, 2016 This report is funded by the Department of Health and Human Services under contract HHSM-500-2012-00009I Task Order HHSM-500-T0000
Use of inhaled and oral glucocorticoids, severity of inflammatory disease and risk of hip/femur fracture: a population-based case control study
 Original Article doi: 10.1111/j.1365-2796.2006.01754.x Use of inhaled and oral glucocorticoids, severity of inflammatory disease and risk of hip/femur fracture: a population-based case control study F.
Original Article doi: 10.1111/j.1365-2796.2006.01754.x Use of inhaled and oral glucocorticoids, severity of inflammatory disease and risk of hip/femur fracture: a population-based case control study F.
«Impact of a pharmaceutical intervention to improve adherence of inhaled medication in asthma and COPD patients»
 «Impact of a pharmaceutical intervention to improve adherence of inhaled medication in asthma and COPD patients» Baseline data from a randomized controlled trial 5 th Swiss Health Services Research Symposium,
«Impact of a pharmaceutical intervention to improve adherence of inhaled medication in asthma and COPD patients» Baseline data from a randomized controlled trial 5 th Swiss Health Services Research Symposium,
Current approaches to the diagnosis and treatment of idiopathic pulmonary fibrosis in Europe: the AIR survey
 REVIEW IDIOPATHIC PULMONARY FIBROSIS Current approaches to the diagnosis and treatment of idiopathic pulmonary fibrosis in Europe: the AIR survey Vincent Cottin 1,2 Affiliations: 1 Hospices Civils de Lyon,
REVIEW IDIOPATHIC PULMONARY FIBROSIS Current approaches to the diagnosis and treatment of idiopathic pulmonary fibrosis in Europe: the AIR survey Vincent Cottin 1,2 Affiliations: 1 Hospices Civils de Lyon,
PATIENT-IMPACT SCORECARD
 UNDERSTANDING THE GSC PATIENT-IMPACT SCORECARD What is the GSC Patient-Impact Scorecard? The GSC Patient-Impact Scorecard shows pharmacy performance scores for GSC patients during the period indicated.
UNDERSTANDING THE GSC PATIENT-IMPACT SCORECARD What is the GSC Patient-Impact Scorecard? The GSC Patient-Impact Scorecard shows pharmacy performance scores for GSC patients during the period indicated.
