The prevalence and severity of chronic asthma have
|
|
|
- Tyler Cunningham
- 5 years ago
- Views:
Transcription
1 An Economic Analysis of Alternative Step-Up Therapies in Asthma Patients Receiving Inhaled Daniel A. Ollendorf, MPH, Alyssa S. Pozniak, BA, Brian W. Bowers, PharmD, Gerry Oster PhD ABSTRACT We analyzed health care claims data to assess the impact of salmeterol xinafoate relative to leukotriene modifiers in asthma patients who were receiving inhaled corticosteroids. Utilization and costs of asthmarelated care were tracked for six months before and 12 months after first receipt of therapy. Use of corticosteroids and short-acting β 2 was lower among salmeterol patients, as were monthly costs of asthma-related care ($101 [±$4] vs $128 [±$9] for leukotriene modifiers). Key Words: salmeterol xinafoate; leukotriene receptor ant; asthma; costs-and-cost-analysis The prevalence and severity of chronic asthma have increased substantially in recent years; between 1980 and 1993, the number of Americans affected by asthma increased more than two-fold, from 6.8 million to over 14 million. 1,2 Mortality caused by asthma also increased by 68% during this period. 3 Exacerbation of asthma symptoms often necessitates the use of rescue medications (e.g., oral steroids, inhaled or oral short-acting β 2 ) as well as physician and/or emergency-room services, all of which can substantially increase costs of care. Prevention of bronchospasm and control of bronchial hyperresponsiveness and inflammation remain the primary goals of drug therapy for chronic asthma. The U.S. National Institutes of Health has stated that inhaled corticosteroids currently represent the most potent and effective anti-inflammatory agents for chronic management of persistent asthma. 4 However, in many patients, optimal control of the disease cannot always be achieved with inhaled corticosteroids alone; accordingly, additional therapy is often warranted. Salmeterol xinafoate (Serevent, GlaxoSmithKline Inc.) and the so-called leukotriene modifiers (montelukast [Singulair], Merck & Co.; zafirlukast [Accolate], AstraZeneca Pharmaceuticals Inc.; and zileuton [Zyflo], Abbott Laboratories Inc.) are agents that are being used increasingly in combination with inhaled corticosteroids in the management of chronic asthma. The safety and efficacy of salmeterol and the leukotriene modifiers have been demonstrated in controlled clinical trials. Salmeterol and zafirlukast also have been compared in a randomized, multicenter, controlled clinical trial. 5 In this study, salmeterol was found to result in significantly greater improvement relative to zafirlukast in pulmonary function, asthma symptoms, Mr. Ollendorf is Senior Analyst and Dr. Oster is Vice President, Policy Analysis Inc. (PAI), in Brookline, Massachusetts. Ms. Pozniak was employed by PAI at the time these analyses were conducted. Dr. Bowers is Senior Health Outcomes Scientist at GlaxoSmithKline Inc., in Research Triangle Park, North Carolina. and supplemental use of the short-acting β 2 agonist albuterol. To date, however, the comparative effects of salmeterol and the leukotriene modifiers on asthma-related health care utilization and costs have not yet been examined. We undertook a study using health care claims data to address this issue. METHODS Overview We conducted a retrospective evaluation of the utilization and costs of asthma-related medications and health care services in patients with chronic asthma who were receiving inhaled corticosteroids and were newly started on salmeterol or a leukotriene modifier, using the claims-processing system of a large New England health insurer. A cohort of plan members with chronic asthma who were receiving inhaled corticosteroids was first identified through a review of all paid pharmacy, professional service, and hospital claims. Among these identified members, patients who were newly started on salmeterol or a leukotriene modifier (i.e., montelukast, zafirlukast, or zileuton) were selected for inclusion in the study sample. The date of the first paid pharmacy claim for one of the study medications was designated the index date. The six-month period prior to the index date and the 12-month period subsequent to this date were designated the pretreatment and follow-up periods, respectively, for each patient in the sample. Utilization and costs of asthma-related care were then compared during the 12-month follow-up period between patients who received salmeterol versus a leukotriene modifier, including the use and associated cost of all asthma-related medications, outpatient services (including physician office, emergency-room, and hospital outpatient visits), and inpatient care. Data Source Data for this study were obtained from the claims-processing system of a large New England health insurer, which includes information on approximately 1.8 million covered lives. Total Vol. 27 No. 3 March 2002 P&T 147
2 billed charges, the amount reimbursed, and the amount of patient copayments were available for all claims. Data available for each paid pharmacy claim included the National Drug Classification (NDC) code, the dispensing date, and the number of therapy-days dispensed. Data available for each paid professional and institutional claim included the date of service, the primary diagnosis (in International Classification of Diseases, 9 th Edition, Clinical Modification [ICD-9-CM] format), and the nature of the service that was rendered (in Health Care Financing Administration Common Procedure Coding System [HCPCS] or Physician s Current Procedural Terminology, 4th Edition [CPT-4] format). Additional data available for each plan member included age, gender, and the start and stop (if applicable) dates of eligibility for plan benefits. Sample Selection The study sample was selected in stepwise fashion. First, all plan members with one or more paid professional-service or institutional claims with a listed diagnosis of asthma (ICD-9-CM 493) between October 1, 1996 and March 31, 1999 were identified. Persons less than 12 years of age were excluded from the sample, consistent with product labeling for most of the medications of interest. Those aged 65 years or older also were excluded because the claims database includes only a limited number of such persons. Next, persons who had at least one pharmacy claim for salmeterol (Serevent, NDC , , , , , , ), montelukast (Singulair, , , , , , ), zafirlukast (Accolate, , ), or zileuton (Zyflo, ) between April 1, 1997 and March 31, 1998 were identified. The date of the earliest observed pharmacy claim during this period was designated the index date ; pretreatment and follow-up periods of six and 12 months, respectively, were then defined for each subject in relation to this date. Patients were assigned to treatment groups (i.e., salmeterol vs. leukotriene modifiers) on the basis of the drug received on the index date. Pharmacy claims were scanned to ensure that all persons selected for inclusion were receiving treatment with an inhaled corticosteroid during the pretreatment and follow-up periods; those who were not were excluded from the study sample. Treatment with inhaled corticosteroids was defined on the basis of one or more and two or more pharmacy claims for such therapy during the pretreatment and follow-up periods, respectively. In addition, to ensure that study therapy was used as an adjunct to (rather than a replacement for) inhaled corticosteroid therapy, the sample was further restricted to those patients who had at least one instance in which an inhaled corticosteroid and one of the study medications were dispensed on the same day. To ensure that treatment received on the index date represented initiation of study therapy as opposed to ongoing care, patients who received any of the study medications during the pretreatment period were excluded from the study sample. Patients with chronic obstructive pulmonary disease (COPD), which was defined on the basis of a listed diagnosis of COPD (ICD-9-CM , ) or receipt of ipratropium bromide, were also excluded. Finally, as is customary in analyses of this nature, patients not continuously enrolled in the health plan over the 18-month period of interest (i.e., six months prior to the index date and 12 months subsequently) were excluded from all analyses, as were those whose health insurance did not include prescription drug coverage. Measures and Analyses Claims were compiled for each study subject for the six months prior to initial receipt of study therapy (pretreatment) and for 12 months subsequently (follow-up). Outcomes of primary interest included the use and associated costs of asthma-related medications and health care services during follow-up. All measures of interest were expressed on a monthly basis. Asthma-related medications were defined to include study therapy (i.e., salmeterol and leukotriene modifiers), inhaled and oral corticosteroids, inhaled and oral short-acting β 2 (e.g., albuterol), xanthine derivatives (e.g., theophylline), and miscellaneous anti-inflammatory agents (e.g., cromolyn sodium). The number of days of therapy with each of these medications was tallied based on the number of days dispensed on each paid pharmacy claim. The numbers of claims for asthma-related physician office, emergency-room, and hospital outpatient visits were tallied, as were the numbers of asthma-related hospitalizations and corresponding days in hospital. Claims were deemed to be asthmarelated based on a listed diagnosis of asthma (ICD-9-CM 493). A direct medical-care cost perspective was employed in all analyses of data. Accordingly, the cost of each claim was estimated by adding the amount reimbursed by the plan and the amount of patient copayment. Utilization and costs of asthma-related care during follow-up were then compared for patients receiving salmeterol or a leukotriene modifier using monthly means and standard errors (SE). Differences in costs were also calculated, along with corresponding 95% confidence intervals. The latter were calculated using techniques of nonparametric bootstrapping, 6 as the distribution of costs was expected to be significantly skewed. In addition to primary analyses of data, secondary analyses of the impact of salmeterol versus leukotriene modifiers alternatively on the costs of respiratory-related and all medical care were conducted. Respiratory-related care was defined on the basis of a relevant listed diagnosis (i.e., ICD-9-CM 466, , , , 493, ). Finally, multivariate analyses (using ordinary least-squares regression) were conducted to control for differences between treatment groups in demographic characteristics and measures of pretreatment utilization. Two models were specified: the dependent variables in these models were the cost of asthmarelated medications and the total cost of asthma-related care during follow-up, respectively. These variables were expressed on a monthly basis and were log-transformed prior to analysis to account for skewness. Explanatory (i.e., independent) variables continued on page P&T March 2002 Vol. 27 No. 3
3 continued from page 148 in these models included age, age squared, gender, the number of days of therapy (per month) during pretreatment with inhaled and oral corticosteroids, as well as inhaled and oral short-acting β 2, the number (per month) of asthmarelated physician, emergency-room, and hospital outpatient visits and hospitalizations during pretreatment, and the total monthly cost of asthma-related care during pretreatment. Adjusted mean costs were then calculated; costs were retransformed to consistent estimates of their natural values using Duan s smearing estimator. 7 All analyses were performed with the Statistical Analysis System (SAS) Version RESULTS Economic Analysis of Step-Up Therapies in Asthma Table 1 Demographics and Other Characteristics Salmeterol+ICS LTM+ICS Characteristic (n=259) (n=106) P Age (mean, SE) 42.3 (0.8) 43.8 (1.2) * Gender (% female) Monthly pretreatment use of asthma-related medications (therapy-days) (mean, SE): Oral 1.07 (0.20) 1.64 (0.35) Inhaled (0.46) (0.83) Oral 0.55 (0.19) 1.75 (0.57) Inhaled (0.64) (1.17) Xanthine derivatives 2.65 (0.47) 4.22 (0.89) agents 1.04 (0.25) 1.05 (0.38) Monthly pretreatment use of asthma-related health care services (mean, SE) Physician office visits 0.23 (0.02) 0.30 (0.05) Emergency-room visits 0.08 (0.01) 0.05 (0.01) Hospital outpatient visits 0.04 (0.01) 0.02 (0.01) Hospital admissions 0.01 (0.00) 0.01 (0.01) Monthly total pretreatment cost of asthma-related care ($) (mean, SE) $84 ($6) $101 ($10) ICS= inhaled corticosteroids; LTM= leukotriene modifiers; * t-test; chi-square test; Wilcoxon rank-sum test. Patient Characteristics A total of 365 patients met all study entry criteria (259 and 106 for salmeterol and leukotriene modifiers, respectively). The groups were similar with respect to age, gender, and most measures of pretreatment utilization (Table 1). Mean (±SE) monthly pretreatment use of inhaled corticosteroids, however, was significantly lower among patients who received salmeterol (10.50 [±0.46] vs [±0.83] therapy-days for leukotriene modifiers, P=0.036), as was the use of oral short-acting β 2 (0.55 [±0.19] vs [±0.57] therapy-days respectively, P=0.024). Monthly costs of asthma-related care (i.e., medications and health care services) also were significantly lower among salmeterol patients ($84 [±$6] vs. $101 [±$10] for leukotriene modifiers, P=0.022). There was some crossover receipt of study therapy during follow-up. Approximately 20% of leukotriene modifier patients received salmeterol, while 11% and 12% of salmeterol patients received montelukast and zafirlukast, respectively (Table 2). Use of zileuton was relatively infrequent in both groups. Approximately 65% of patients had one or more physician office visits during follow-up; emergency-room services were required by one-third of patients. Hospitalization for asthma was relatively infrequent. Utilization of Asthma-Related Medications and Services On a monthly basis, the use of inhaled short-acting β 2 was substantially lower during follow-up among patients who received salmeterol (7.51 [±0.47] vs [±0.99] therapy-days for leukotriene modifiers) (Table 3); the use of oral short-acting β 2 and oral corticosteroids also was somewhat lower among these patients. The number of physician office visits per month was nominally lower among patients receiving salmeterol (0.194 [±0.022] vs [±0.035] for leukotriene modifiers); conversely, the number of emergency-room visits was higher among these patients (0.051 [±0.007] vs [±0.010] for salmeterol and leukotriene modifiers, respectively). The use of other hospital outpatient and inpatient services did not differ materially between the two treatment groups. Costs of Asthma-Related Care The costs of asthma-related medications and health care services are presented in Table 4. Monthly costs of study therapy, inhaled corticosteroids, and rescue medications (i.e., oral steroids, inhaled and oral short-acting β 2 ) were lower among patients receiving salmeterol; the total mean monthly cost of asthma-related medications was approximately $20 lower among these patients ($79 [±$3] vs. $98 [±$5] for salmeterol and leukotriene modifiers respectively; 95% confidence interval [CI] for difference: -$33, -$7). Monthly costs of asthma-related outpatient care ($19 [±$2] vs. $23 [±$4] for salmeterol and leukotriene modifiers, respectively) and inpatient care ($4 [±$2] vs. $7 [±$5], respectively) were slightly lower among salmeterol patients. Total monthly costs of asthma-related care were 20% lower among salmeterol patients ($101 [±$4] vs. $128 [±$9] for leukotriene modifiers; 95% CI for difference: -$51, -$4); this difference was primarily manifested in differences in the costs of asthma-related medications. Multivariate Analyses After multivariate adjustment, mean monthly costs of asthmarelated medications and total costs of asthma-related care were essentially identical to those reported in univariate analyses of data. Adjusted mean monthly costs of asthma-related medications were $75 and $95 for salmeterol and leukotriene modifier patients, respectively; total monthly costs of asthmarelated care were $99 and $122, respectively. Vol. 27 No. 3 March 2002 P&T 151
4 Table 2 Number (Percentage) of Patients Receiving Asthma-related Medications and Health Care Services During Follow-up Type of Medication/ Salmeterol+ICS LTM+ICS Service (n=259) (n=106) n (%) Medications Salmeterol 259 (100) 21 (19.8) Montelukast 31 (12) 16 (15.1) Zafirlukast 29 (11.2) 99 (93.4) Zileuton 3 (1.2) 6 (5.7) Oral 84 (32.4) 41 (38.7) Inhaled 259 (100) 106 (100) Oral 12 (4.6) 10 (9.4) Inhaled 214 (82.6) 93 (87.7) Xanthine derivatives 33 (12.7) 14 (13.2) agents 24 (9.3) 9 (8.5) Outpatient Physician office visits 162 (62.5) 83 (78.3) Emergency-room visits 80 (30.9) 25 (23.6) Hospital outpatient visits 27 (10.4) 11 (10.4) Hospitalization 6 (2.3) 2 (1.9) ICS= inhaled corticosteroids; LTM= leukotriene modifiers. Table 3 Use of Asthma-related Medications and Health Care Services During Follow-up Type of Medication/ Salmeterol+ICS LTM+ICS Service (n=259) (n=106) Mean per Month (SE) Medications (therapy-days) Salmeterol (0.47) 1.65 (0.46) Montelukast 0.94 (0.21) 1.72 (0.54) Zafirlukast 1.23 (0.28) (1.02) Zileuton 0.10 (0.08) 0.47 (0.22) Oral 0.73 (0.16) 1.36 (0.30) Inhaled (0.44) (0.74) Oral 0.47 (0.18) 2.05 (0.68) Inhaled 7.51 (0.47) (0.99) Xanthine derivatives 2.64 (0.48) 3.24 (0.87) agents 1.07 (0.26) 0.66 (0.28) Outpatient Physician office visits (0.022) (0.035) Emergency-room visits (0.007) (0.010) Hospital outpatient visits (0.003) (0.004) Hospitalizations (0.002) (0.004) Days in hospital (0.004) (0.008) ICS= inhaled corticosteroids; LTM= leukotriene modifiers. DISCUSSION Using health care claims data from a large New England insurer, we examined the economic impact of salmeterol versus leukotriene modifiers among patients aged 13 to 65 years with chronic asthma who were receiving inhaled corticosteroids. Health care claims were compiled for six months prior to their first receipt of study therapy (pretreatment) and for 12 months subsequently (follow-up). Utilization and costs of care were then compared during follow-up between patients receiving salmeterol or a leukotriene modifier. The use of inhaled short-acting β 2 was substantially lower among patients receiving salmeterol; their use of other rescue medications (i.e., oral β 2, oral steroids) was also lower. The total monthly costs of asthma-related care were 20% lower among salmeterol patients, a difference that was primarily caused by the lower costs of asthma-related medications. The cost savings with salmeterol would be estimated to total over $300 per patient annually. We acknowledge several limitations of our study. First, as with most nonexperimental research, we cannot rule out the possibility that our results simply reflect the effects of selection bias (e.g., differences in disease severity between salmeterol and leukotriene modifier patients) rather than those of salmeterol therapy per se. Indeed, pretreatment use of inhaled corticosteroids was significantly lower among patients receiving salmeterol, as was the total cost of asthma-related care. Although our primary findings were unchanged when we controlled for selected demographic and pretreatment differences between groups, other differences might have remained for which we did not adequately control. We identified patients using inhaled corticosteroids based on the presence of one or more and two or more relevant claims during pretreatment and follow-up, respectively; attempts to use more restrictive criteria would have resulted in large reductions in sample size. Although we included patients in our sample with infrequent use of corticosteroids, there is precedent for our methods. The results of a Canadian study of prescription claims suggest that only about 7.5% of persons using inhaled corticosteroids would meet criteria for regular use (i.e., prescriptions filled at least every 90 days). 9 In another study of the effects of inhaled corticosteroid use on asthma-related hospitalization, using data from a large managed-care organization, the mean annual number of prescriptions for inhaled corticosteroids was estimated to be Treatment groups were defined on the basis of one or more claims for study therapy; our sample therefore included patients with infrequent use of study medications over 12 months of follow-up. To examine this issue, we also conducted analyses of the monthly costs of asthma-related care alter- 152 P&T March 2002 Vol. 27 No. 3
5 Table 4 Costs of Asthma-related Medications and Health Care Services During Follow-up Type of Salmeterol+ LTM+ Difference ([Sal- Medication/ ICS ICS meterol+ics] Service (n=259) (n=106) [LTM+ICS]) Mean per Month (SE), $ Estimate (95% CI) Medications Salmeterol 28 (1) 3 (1) 25 (21, 27) Montelukast 2 (0) 4 (1) -2 (-5, 1) Zafirlukast 2 (0) 31 (2) -29 (-33, -25) Zileuton 0 (0) 1 (0) -1 (-2, 0) Oral 0 (0) 0 (0) 0 (0, 0) Inhaled 35 (2) 39 (3) -5 (-13, 3) Oral 1 (0) 3 (1) -2 (-4, 0) Inhaled 7 (1) 12 (1) -5 (-8, -2) Xanthine derivatives 2 (0) 3 (1) -1 (-4, 1) Miscellaneous agents 2 (1) 1 (1) 1 (-1, 3) Total medication 79 (3) 98 (5) -20 (-33, -7) Outpatient care Physician office visits 12 (1) 16 (2) -5 (-11, 1) Emergency-room visits 6 (1) 6 (2) 0 (-6, 6) Hospital outpatient visits 1 (0) 1 (0) 0 (-1, 1) Total outpatient care 19 (2) 23 (4) -4 (-14, 5) Inpatient Care 4 (2) 7 (5) -3 (-16, 7) Total Asthma-Related Care 101 (4) 128 (9) -26 (-51, -4) NOTE: sum of component costs might not equal total because of rounding. ICS= inhaled corticosteroids; LTM= leukotriene modifiers; CI= confidence intervals. natively among patients with two or more (n=339) and four or more (n=276) paid claims for study therapy. Findings were similar to those reported herein. The number of patients who received salmeterol and one or more leukotriene modifiers during follow-up (and vice versa) was not inconsequential. Inclusion of these crossover patients in the study had the potential to modify the effects we observed. To address this, we replicated our analyses excluding crossover patients. A total of 289 (79.2%) of the 365 patients in our original sample met this criterion (n=203 and 86 for salmeterol and leukotriene modifiers, respectively). The difference in mean monthly costs of asthma-related care was similar to that observed in our original analyses ($89 [±$4] vs. $116 [±$9] for salmeterol and leukotriene modifiers, respectively). The use of asthma-related medications was assessed based on information on the numbers of paid prescription claims and corresponding therapy-days dispensed. These data, therefore, reflect the amount of medication purchased by patients, not that which was actually taken. The accuracy of this method of measuring adherence to prescribed medication varies considerably by disease and medication Our estimates might also be subject to errors in the coding of claims. Finally, although our findings were based on a broad cross section of patients and health plans from a large insurer, our results might not be generalizable to any given setting (e.g., an inner-city health clinic); we also excluded patients aged 65 years or older, as the database includes only a small number of such persons. Despite these limitations, we believe that our study has important implications. To the best of our knowledge, it is the first to examine the economic impact of salmeterol versus leukotriene modifiers as add-on therapy to inhaled corticosteroids. Our findings suggest that in a real world clinical setting, the use of salmeterol in combination with inhaled corticosteroids offers meaningful cost savings and reductions in the need for rescue medication compared to treatment with leukotriene modifiers plus inhaled corticosteroids. Our study supports results from randomized controlled trials that have demonstrated the benefits of salmeterol relative to leukotriene modifiers. 5 Acknowledgements We wish to thank John Edelsberg, MD, MPH for his contributions to this study. We also thank Erin Richardson and Sheila Ercolini for their assistance in the preparation of this manuscript. REFERENCES 1. Centers for Disease Control and Prevention. Surveillance for asthma United States, MMWR 1998;47(SS-1): Centers for Disease Control and Prevention. Asthma mortality and hospitalization among children and young adults United States, MMWR 1996;45(17): Halfon N, Newacheck P. Childhood asthma and poverty: Differential impacts and utilization of health services. Pediatrics 1993;91(1): National Heart, Lung, and Blood Institute. Expert Panel Report 2: Guidelines for the Diagnosis and Management of Asthma. Bethesda, MD: National Institutes of Health, April Publication no Busse W, Nelson H, Wolfe J, Kalberg C, Yancey SW, Rickard KA. Comparison of inhaled salmeterol with oral zafirlukast in patients with asthma. J Allergy Clin Immunol 1999;103: Chaudhary MA, Stearns SC. Estimating confidence intervals for costeffectiveness ratios: An example from a clinical trial. Statistics in Medicine 1996;15: Duan N. Smearing estimate: A nonparametric retransformation method. Journal of the American Statistical Association 1983;78(383): SAS Institute Inc., SAS/STAT User s Guide, Version 6, Fourth ed. Vol 1&2, Cary, NC: SAS Institute Inc., Blais L, Ernst P, Boivin JF, Suissa S. Inhaled corticosteroids and the prevention of readmission to hospital for asthma. Am J Respir Crit Care Med 1998;158: Donahue JG, Weiss ST, Livingston JM, Goetsch MA, Geineder DK, Platt R. Inhaled steroids and the risk of hospitalization for asthma. JAMA 1997;277: Christensen DB, Williams B, Goldberg HI, Martin DP, Engelberg R, LoGerfo JP. Comparison of prescription and medical records in reflecting patient antihypertensive drug therapy. Ann Pharmacother 1994;28: Rizzo JA, Simons WR. Variations in compliance among hypertensive patients by drug class: Implications for health care costs. Clinical Therapeutics 1997;19(6): Grymonpre RE, Didur CD, Montgomery PR, Sitar DS. Pill count, self-report, and pharmacy claims data to measure medication adherence in the elderly. Ann Pharmacother 1998;32: Funding for this research was provided by GlaxoSmithKline Inc., Research Triangle Park, North Carolina. Vol. 27 No. 3 March 2002 P&T 153
Comparison of asthma costs in patients starting fluticasone propionate compared to patients starting montelukast
 RESPIRATORY MEDICINE (2001) 95, 227 234 doi:10.1053/rmed.2000.1027, available online at http://www.idealibrary.com on Comparison of asthma costs in patients starting fluticasone propionate compared to
RESPIRATORY MEDICINE (2001) 95, 227 234 doi:10.1053/rmed.2000.1027, available online at http://www.idealibrary.com on Comparison of asthma costs in patients starting fluticasone propionate compared to
Four of 10 patients with asthma suffer moderate REVIEW DUAL-CONTROLLER REGIMENS II: OBSERVATIONAL DATA. Michael S. Blaiss, MD ABSTRACT
 DUAL-CONTROLLER REGIMENS II: OBSERVATIONAL DATA Michael S. Blaiss, MD ABSTRACT The differences between clinical trials and clinical practice often create difficulty for generalizing results of controlled
DUAL-CONTROLLER REGIMENS II: OBSERVATIONAL DATA Michael S. Blaiss, MD ABSTRACT The differences between clinical trials and clinical practice often create difficulty for generalizing results of controlled
Adherence to asthma controller medication regimens
 Respiratory Medicine (2005) 99, 1263 1267 Adherence to asthma controller medication regimens D.A. Stempel a,, S.W. Stoloff b, J.R. Carranza Rosenzweig c, R.H. Stanford c, K.L. Ryskina d, A.P. Legorreta
Respiratory Medicine (2005) 99, 1263 1267 Adherence to asthma controller medication regimens D.A. Stempel a,, S.W. Stoloff b, J.R. Carranza Rosenzweig c, R.H. Stanford c, K.L. Ryskina d, A.P. Legorreta
Pharmacy Medical Policy Asthma and Chronic Obstructive Pulmonary Disease Medication Management
 Pharmacy Medical Policy Asthma and Chronic Obstructive Pulmonary Disease Medication Management Table of Contents Policy: Commercial Information Pertaining to All Policies Endnotes Policy: Medicare References
Pharmacy Medical Policy Asthma and Chronic Obstructive Pulmonary Disease Medication Management Table of Contents Policy: Commercial Information Pertaining to All Policies Endnotes Policy: Medicare References
The National Asthma Education and Prevention Program s
 Long-Acting b-agonist Among Children and Adults With Asthma Elizabeth A. Wasilevich, PhD, MPH; Sarah J. Clark, MPH; Lisa M. Cohn, MS; and Kevin J. Dombkowski, DrPH Managed Care & Healthcare Communications,
Long-Acting b-agonist Among Children and Adults With Asthma Elizabeth A. Wasilevich, PhD, MPH; Sarah J. Clark, MPH; Lisa M. Cohn, MS; and Kevin J. Dombkowski, DrPH Managed Care & Healthcare Communications,
One-year claims analysis comparing inhaled fluticasone propionate with zafirlukast for the treatment of asthma
 One-year claims analysis comparing inhaled fluticasone propionate with zafirlukast for the treatment of asthma David A. Stempel, MD, a Jay W. Meyer, PhD, b Richard H. Stanford, PharmD, MS, c and Steve
One-year claims analysis comparing inhaled fluticasone propionate with zafirlukast for the treatment of asthma David A. Stempel, MD, a Jay W. Meyer, PhD, b Richard H. Stanford, PharmD, MS, c and Steve
Current Asthma Management: Opportunities for a Nutrition-Based Intervention
 Current Asthma Management: Opportunities for a Nutrition-Based Intervention Stanley J. Szefler, MD Approximately 22 million Americans, including 6 million children, have asthma. It is one of the most prevalent
Current Asthma Management: Opportunities for a Nutrition-Based Intervention Stanley J. Szefler, MD Approximately 22 million Americans, including 6 million children, have asthma. It is one of the most prevalent
Quality ID #444 (NQF 1799): Medication Management for People with Asthma National Quality Strategy Domain: Efficiency and Cost Reduction
 Quality ID #444 (NQF 1799): Medication Management for People with Asthma National Quality Strategy Domain: Efficiency and Cost Reduction 2018 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY MEASURE TYPE:
Quality ID #444 (NQF 1799): Medication Management for People with Asthma National Quality Strategy Domain: Efficiency and Cost Reduction 2018 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY MEASURE TYPE:
Zhao Y Y et al. Ann Intern Med 2012;156:
 Zhao Y Y et al. Ann Intern Med 2012;156:560-569 Introduction Fibrates are commonly prescribed to treat dyslipidemia An increase in serum creatinine level after use has been observed in randomized, placebocontrolled
Zhao Y Y et al. Ann Intern Med 2012;156:560-569 Introduction Fibrates are commonly prescribed to treat dyslipidemia An increase in serum creatinine level after use has been observed in randomized, placebocontrolled
Setting The setting was the community. The economic study was carried out in New Jersey, USA.
 Asthma rescue and allergy medication use among asthmatic children with prior allergy prescriptions who initiated asthma controller therapy Luskin A, Bukstein D, Kocevar V S, Yin D D Record Status This
Asthma rescue and allergy medication use among asthmatic children with prior allergy prescriptions who initiated asthma controller therapy Luskin A, Bukstein D, Kocevar V S, Yin D D Record Status This
Dual-controller therapy, or combinations REVIEW DUAL-CONTROLLER REGIMENS I: DATA FROM RANDOMIZED, CONTROLLED CLINICAL TRIALS.
 DUAL-CONTROLLER REGIMENS I: DATA FROM RANDOMIZED, CONTROLLED CLINICAL TRIALS Samy Suissa, PhD ABSTRACT Dual-controller therapy, or combinations of 2 or more pharmacotherapies with complementary mechanisms
DUAL-CONTROLLER REGIMENS I: DATA FROM RANDOMIZED, CONTROLLED CLINICAL TRIALS Samy Suissa, PhD ABSTRACT Dual-controller therapy, or combinations of 2 or more pharmacotherapies with complementary mechanisms
The economic burden of asthma in US children: Estimates from the National Medical Expenditure Survey
 The economic burden of asthma in US children: Estimates from the National Medical Expenditure Survey Paula Lozano, MD, MPH, a Sean D. Sullivan, PhD, b David H. Smith, MHA, PhD, b and Kevin B. Weiss, MD,
The economic burden of asthma in US children: Estimates from the National Medical Expenditure Survey Paula Lozano, MD, MPH, a Sean D. Sullivan, PhD, b David H. Smith, MHA, PhD, b and Kevin B. Weiss, MD,
Dual-Controller Asthma Therapy: Rationale and Clinical Benefits
 B/1 Dual-Controller Asthma Therapy: Rationale and Clinical Benefits MODULE B The 1997 National Heart, Lung, and Blood Institute (NHLBI) Expert Panel guidelines on asthma management recommend a 4-step approach
B/1 Dual-Controller Asthma Therapy: Rationale and Clinical Benefits MODULE B The 1997 National Heart, Lung, and Blood Institute (NHLBI) Expert Panel guidelines on asthma management recommend a 4-step approach
The New England Journal of Medicine LOW-DOSE INHALED CORTICOSTEROIDS AND THE PREVENTION OF DEATH FROM ASTHMA
 LOW-DOSE INHALED CORTICOSTEROIDS AND THE PREVENTION OF DEATH FROM ASTHMA SAMY SUISSA, PH.D., PIERRE ERNST, M.D., SERGE BENAYOUN, M.SC., MARC BALTZAN, M.D., AND BING CAI, M.SC. ABSTRACT Background Although
LOW-DOSE INHALED CORTICOSTEROIDS AND THE PREVENTION OF DEATH FROM ASTHMA SAMY SUISSA, PH.D., PIERRE ERNST, M.D., SERGE BENAYOUN, M.SC., MARC BALTZAN, M.D., AND BING CAI, M.SC. ABSTRACT Background Although
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Nucala) Reference Number: CP.PHAR.200 Effective Date: 04.01.16 Last Review Date: 02.18 Line of Business: Commercial, Medicaid Coding Implications Revision Log See Important Reminder at
Clinical Policy: (Nucala) Reference Number: CP.PHAR.200 Effective Date: 04.01.16 Last Review Date: 02.18 Line of Business: Commercial, Medicaid Coding Implications Revision Log See Important Reminder at
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Fasenra) Reference Number: CP.PHAR.## Effective Date: 01.16.18 Last Review Date: 05.18 Line of Business: Commercial, Medicaid Revision Log See Important Reminder at the end of this policy
Clinical Policy: (Fasenra) Reference Number: CP.PHAR.## Effective Date: 01.16.18 Last Review Date: 05.18 Line of Business: Commercial, Medicaid Revision Log See Important Reminder at the end of this policy
In 2002, it was reported that 72 of 1000
 REPORTS Aligning Patient Care and Asthma Treatment Guidelines Eric Cannon, PharmD Abstract This article describes how the National Asthma Education and Prevention Program Guidelines for the Diagnosis and
REPORTS Aligning Patient Care and Asthma Treatment Guidelines Eric Cannon, PharmD Abstract This article describes how the National Asthma Education and Prevention Program Guidelines for the Diagnosis and
II: Moderate Worsening airflow limitations Dyspnea on exertion, cough, and sputum production; patient usually seeks medical
 Table 3.1. Classification of COPD Severity Stage Pulmonary Function Test Findings Symptoms I: Mild Mild airflow limitations +/ Chronic cough and sputum production; patient unaware of abnormal FEV 1 80%
Table 3.1. Classification of COPD Severity Stage Pulmonary Function Test Findings Symptoms I: Mild Mild airflow limitations +/ Chronic cough and sputum production; patient unaware of abnormal FEV 1 80%
The 3 components of evidence. Economic Analysis of Asthma Practices ...PRESENTATIONS... Based on a presentation by David A.
 ...PRESENTATIONS... Economic Analysis of Asthma Practices Based on a presentation by David A. Stempel, MD Presentation Summary When deciding on treatment for patients with asthma, clinicians should consider
...PRESENTATIONS... Economic Analysis of Asthma Practices Based on a presentation by David A. Stempel, MD Presentation Summary When deciding on treatment for patients with asthma, clinicians should consider
Asthma Population Management: Identifying Persistent Asthma, Defining High Risk Asthma, and Measuring Quality of Asthma Care
 Asthma Population Management: Identifying Persistent Asthma, Defining High Risk Asthma, and Measuring Quality of Asthma Care Michael Schatz, MD, MS Allergy Department Kaiser Permanente, San Diego, CA Constructs
Asthma Population Management: Identifying Persistent Asthma, Defining High Risk Asthma, and Measuring Quality of Asthma Care Michael Schatz, MD, MS Allergy Department Kaiser Permanente, San Diego, CA Constructs
Learning the Asthma Guidelines by Case Studies
 Learning the Asthma Guidelines by Case Studies Timothy Craig, DO Professor of Medicine and Pediatrics Distinguished Educator Penn State University Hershey Medical Center Objectives 1. Learn the Asthma
Learning the Asthma Guidelines by Case Studies Timothy Craig, DO Professor of Medicine and Pediatrics Distinguished Educator Penn State University Hershey Medical Center Objectives 1. Learn the Asthma
The clinical quality measures as selected by the Clinical Management subcommittee for 2016 for the adult population are:
 For 2016 the Clinical Integration Program is moving its clinical quality measures from the Verisk Healthcare Quality and Risk Measures to the National Committee for Quality Assurance HEDIS based measures.
For 2016 the Clinical Integration Program is moving its clinical quality measures from the Verisk Healthcare Quality and Risk Measures to the National Committee for Quality Assurance HEDIS based measures.
Asthma is a highly prevalent and costly
 Asthma Treatment Guidelines: How Do We Measure Up? Robert P. Navarro, PharmD Abstract The use of clinical guidelines for the management of asthma can help improve patient outcomes and control costs. This
Asthma Treatment Guidelines: How Do We Measure Up? Robert P. Navarro, PharmD Abstract The use of clinical guidelines for the management of asthma can help improve patient outcomes and control costs. This
Patients with chronic obstructive pulmonary
 HEALTH ECONOMICS Delivery of Ipratropium and Albuterol Combination Therapy for Chronic Obstructive Pulmonary Disease: Effectiveness of a Two-in-one Inhaler Versus Separate Inhalers Elizabeth Chrischilles,
HEALTH ECONOMICS Delivery of Ipratropium and Albuterol Combination Therapy for Chronic Obstructive Pulmonary Disease: Effectiveness of a Two-in-one Inhaler Versus Separate Inhalers Elizabeth Chrischilles,
Clinical trial efficacy: What does it really tell you?
 Clinical trial efficacy: What does it really tell you? Joseph Spahn, MD Denver, Colo The primary goal of most clinical trials is an evaluation of the efficacy of the drug being evaluated. Therefore, it
Clinical trial efficacy: What does it really tell you? Joseph Spahn, MD Denver, Colo The primary goal of most clinical trials is an evaluation of the efficacy of the drug being evaluated. Therefore, it
Q: Should patients with mild asthma
 1-MINUTE CONSULT CME CREDIT EDUCATIONAL OBJECTIVE: Readers will consider prescribing inhaled corticosteroids to their patients who have mild persistent asthma brief answers to specific clinical questions
1-MINUTE CONSULT CME CREDIT EDUCATIONAL OBJECTIVE: Readers will consider prescribing inhaled corticosteroids to their patients who have mild persistent asthma brief answers to specific clinical questions
Inhaled bronchodilators relax constricted airways and treat the noisy part of asthma: coughing, wheezing, choking and shortness of breath.
 Inhaled bronchodilators relax constricted airways and treat the noisy part of asthma: coughing, wheezing, choking and shortness of breath. AccuNeb inhalation 0.021% solution: 0.63mg/3mL 3-4 times solution
Inhaled bronchodilators relax constricted airways and treat the noisy part of asthma: coughing, wheezing, choking and shortness of breath. AccuNeb inhalation 0.021% solution: 0.63mg/3mL 3-4 times solution
Outcomes: Initially, our primary definitions of pneumonia was severe pneumonia, where the subject was hospitalized
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
DESCRIPTION: Percentage of patients aged 5 years and older with a diagnosis of persistent asthma who were prescribed long-term control medication
 Measure #53 (NQF 0047): Asthma: Pharmacologic Therapy for Persistent Asthma - Ambulatory Care Setting National Quality Strategy Domain: Effective Clinical Care 2016 PQRS OPTIONS FOR INDIVIDUAL MEASURES:
Measure #53 (NQF 0047): Asthma: Pharmacologic Therapy for Persistent Asthma - Ambulatory Care Setting National Quality Strategy Domain: Effective Clinical Care 2016 PQRS OPTIONS FOR INDIVIDUAL MEASURES:
Cataracts are a major public health problem,
 Eur Respir J 2006; 27: 1168 1174 DOI: 10.1183/09031936.06.00043005 CopyrightßERS Journals Ltd 2006 Low-dose inhaled and nasal corticosteroid use and the risk of cataracts P. Ernst*,#, M. Baltzan*,",+,
Eur Respir J 2006; 27: 1168 1174 DOI: 10.1183/09031936.06.00043005 CopyrightßERS Journals Ltd 2006 Low-dose inhaled and nasal corticosteroid use and the risk of cataracts P. Ernst*,#, M. Baltzan*,",+,
I nhaled corticosteroids are the treatment of choice in the
 880 ORIGINAL ARTICLE Regular use of inhaled corticosteroids and the long term prevention of hospitalisation for asthma S Suissa, P Ernst, A Kezouh... See end of article for authors affiliations... Correspondence
880 ORIGINAL ARTICLE Regular use of inhaled corticosteroids and the long term prevention of hospitalisation for asthma S Suissa, P Ernst, A Kezouh... See end of article for authors affiliations... Correspondence
PATIENT-IMPACT SCORECARD
 UNDERSTANDING THE GSC PATIENT-IMPACT SCORECARD What is the GSC Patient-Impact Scorecard? The GSC Patient-Impact Scorecard shows pharmacy performance scores for GSC patients during the period indicated.
UNDERSTANDING THE GSC PATIENT-IMPACT SCORECARD What is the GSC Patient-Impact Scorecard? The GSC Patient-Impact Scorecard shows pharmacy performance scores for GSC patients during the period indicated.
Effects on Resource Utilization of Adding Salmeterol in Combination or Separately to Inhaled Corticosteroids
 RESEARCH Effects on Resource Utilization of Adding Salmeterol in Combination or Separately to Inhaled Corticosteroids JAMES CHAN, PharmD, PhD; RITA L. HUI, PharmD, MS; and MICHELE M. SPENCE, PhD ABSTRACT
RESEARCH Effects on Resource Utilization of Adding Salmeterol in Combination or Separately to Inhaled Corticosteroids JAMES CHAN, PharmD, PhD; RITA L. HUI, PharmD, MS; and MICHELE M. SPENCE, PhD ABSTRACT
Asthma in Pediatric Patients. DanThuy Dao, D.O., FAAP. Disclosures. None
 Asthma in Pediatric Patients DanThuy Dao, D.O., FAAP Disclosures None Objectives 1. Discuss the evaluation and management of asthma in a pediatric patient 2. Accurately assess asthma severity and level
Asthma in Pediatric Patients DanThuy Dao, D.O., FAAP Disclosures None Objectives 1. Discuss the evaluation and management of asthma in a pediatric patient 2. Accurately assess asthma severity and level
Montelukast vs. Inhaled Low-Dose Budesonide as Monotherapy in the Treatment of Mild Persistent Asthma: A Randomized Double Blind Controlled Trial
 Montelukast vs. Inhaled Low-Dose Budesonide as Monotherapy in the Treatment of Mild Persistent Asthma: A Randomized Double Blind Controlled Trial by Vikram Kumar, a P. Ramesh, a Rakesh Lodha, a R. M. Pandey,
Montelukast vs. Inhaled Low-Dose Budesonide as Monotherapy in the Treatment of Mild Persistent Asthma: A Randomized Double Blind Controlled Trial by Vikram Kumar, a P. Ramesh, a Rakesh Lodha, a R. M. Pandey,
Methods to Diagnose Adherence Status
 Methods to Diagnose Adherence Status March 4, 2014 Andrew G Weinstein MD Associate Clinical Professor Pediatrics Jefferson Medical College President, Adherence Management Systems Disclosures President,
Methods to Diagnose Adherence Status March 4, 2014 Andrew G Weinstein MD Associate Clinical Professor Pediatrics Jefferson Medical College President, Adherence Management Systems Disclosures President,
Asthma drug ratios and exacerbations: claims data from universal health coverage systems
 ORIGINAL ARTICLE ASTHMA Asthma drug ratios and exacerbations: claims data from universal health coverage systems Laurent Laforest 1, Idlir Licaj 1, Gilles Devouassoux 2,Gérard Chatte 3, Jennifer Martin
ORIGINAL ARTICLE ASTHMA Asthma drug ratios and exacerbations: claims data from universal health coverage systems Laurent Laforest 1, Idlir Licaj 1, Gilles Devouassoux 2,Gérard Chatte 3, Jennifer Martin
METHODS RESULTS. Supported by funding from Ortho-McNeil Janssen Scientific Affairs, LLC
 PREDICTORS OF MEDICATION ADHERENCE AMONG PATIENTS WITH SCHIZOPHRENIC DISORDERS TREATED WITH TYPICAL AND ATYPICAL ANTIPSYCHOTICS IN A LARGE STATE MEDICAID PROGRAM S.P. Lee 1 ; K. Lang 2 ; J. Jackel 2 ;
PREDICTORS OF MEDICATION ADHERENCE AMONG PATIENTS WITH SCHIZOPHRENIC DISORDERS TREATED WITH TYPICAL AND ATYPICAL ANTIPSYCHOTICS IN A LARGE STATE MEDICAID PROGRAM S.P. Lee 1 ; K. Lang 2 ; J. Jackel 2 ;
Friday and weekend hospital stays: effects on mortality
 ERJ Express. Published on May 14, 2014 as doi: 10.1183/09031936.00007714 ORIGINAL ARTICLE IN PRESS CORRECTED PROOF Friday and weekend hospital stays: effects on mortality Samy Suissa 1,2, Sophie Dell Aniello
ERJ Express. Published on May 14, 2014 as doi: 10.1183/09031936.00007714 ORIGINAL ARTICLE IN PRESS CORRECTED PROOF Friday and weekend hospital stays: effects on mortality Samy Suissa 1,2, Sophie Dell Aniello
A randomized controlled trial of a pediatric asthma outreach program
 A randomized controlled trial of a pediatric asthma outreach program Dirk K. Greineder, MD, PhD, a,b Kathleen C. Loane, RN, c and Paula Parks, RN, NP a Boston, Mass Background: Previous studies have shown
A randomized controlled trial of a pediatric asthma outreach program Dirk K. Greineder, MD, PhD, a,b Kathleen C. Loane, RN, c and Paula Parks, RN, NP a Boston, Mass Background: Previous studies have shown
Clinical Policy: Omalizumab (Xolair) Reference Number: ERX.SPA.141 Effective Date: Last Review Date: 08.17
 Clinical Policy: (Xolair) Reference Number: ERX.SPA.141 Effective Date: 03.01.14 Last Review Date: 08.17 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
Clinical Policy: (Xolair) Reference Number: ERX.SPA.141 Effective Date: 03.01.14 Last Review Date: 08.17 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
Chronic obstructive pulmonary disease (COPD) is characterized
 RESEARCH Impact of COPD Exacerbation Frequency on Costs for a Managed Care Population Anand A. Dalal, PhD, MBA; Jeetvan Patel, MS; Anna D Souza, BPharm, PhD; Eileen Farrelly, MPH; Saurabh Nagar, MS; and
RESEARCH Impact of COPD Exacerbation Frequency on Costs for a Managed Care Population Anand A. Dalal, PhD, MBA; Jeetvan Patel, MS; Anna D Souza, BPharm, PhD; Eileen Farrelly, MPH; Saurabh Nagar, MS; and
Comprehensive Research Plan: Inhaled corticosteroids + long-acting beta agonists (ICS+LABA) for the treatment of asthma
 Comprehensive Research Plan: Inhaled corticosteroids + long-acting beta agonists (ICS+LABA) for the treatment of asthma Pharmacoepidemiology Unit July 10, 2014 30 Bond Street, Toronto ON, M5B 1W8 www.odprn.ca
Comprehensive Research Plan: Inhaled corticosteroids + long-acting beta agonists (ICS+LABA) for the treatment of asthma Pharmacoepidemiology Unit July 10, 2014 30 Bond Street, Toronto ON, M5B 1W8 www.odprn.ca
HEALTH CARE EXPENDITURES ASSOCIATED WITH PERSISTENT EMERGENCY DEPARTMENT USE: A MULTI-STATE ANALYSIS OF MEDICAID BENEFICIARIES
 HEALTH CARE EXPENDITURES ASSOCIATED WITH PERSISTENT EMERGENCY DEPARTMENT USE: A MULTI-STATE ANALYSIS OF MEDICAID BENEFICIARIES Presented by Parul Agarwal, PhD MPH 1,2 Thomas K Bias, PhD 3 Usha Sambamoorthi,
HEALTH CARE EXPENDITURES ASSOCIATED WITH PERSISTENT EMERGENCY DEPARTMENT USE: A MULTI-STATE ANALYSIS OF MEDICAID BENEFICIARIES Presented by Parul Agarwal, PhD MPH 1,2 Thomas K Bias, PhD 3 Usha Sambamoorthi,
BUKSTEIN, MD; ALLAN LUSKIN, MD; SHIVA G. SAJJAN,
 RESEARCH Administrative Claims Analysis of Asthma-Related Health Care Utilization for Patients Who Received Inhaled Corticosteroids With Either Montelukast or Salmeterol as Combination Therapy FELICIA
RESEARCH Administrative Claims Analysis of Asthma-Related Health Care Utilization for Patients Who Received Inhaled Corticosteroids With Either Montelukast or Salmeterol as Combination Therapy FELICIA
Combination Therapy with Inhaled Long-Acting
 Combination Therapy with Inhaled Long-Acting 2 -Agonists and Inhaled Corticosteroids: A Paradigm Shift in Asthma Management Stuart Stoloff, M.D., Kim Poinsett-Holmes, Pharm.D., and Paul M. Dorinsky, M.D.
Combination Therapy with Inhaled Long-Acting 2 -Agonists and Inhaled Corticosteroids: A Paradigm Shift in Asthma Management Stuart Stoloff, M.D., Kim Poinsett-Holmes, Pharm.D., and Paul M. Dorinsky, M.D.
INHALED AND NASAL CORTICOSTEROID USE AND THE RISK OF FRACTURE. Online data supplement.
 INHALED AND NASAL CORTICOSTEROID USE AND THE RISK OF FRACTURE. Online data supplement. Samy Suissa, PhD, Marc Baltzan, MD, MSc, Richard Kremer, MD, Pierre Ernst, MD, MSc 26 METHODS Source of data We used
INHALED AND NASAL CORTICOSTEROID USE AND THE RISK OF FRACTURE. Online data supplement. Samy Suissa, PhD, Marc Baltzan, MD, MSc, Richard Kremer, MD, Pierre Ernst, MD, MSc 26 METHODS Source of data We used
Reducing COPD Exacerbation Readmissions in a Community-Based Teaching Hospital
 Reducing COPD Exacerbation Readmissions in a Community-Based Teaching Hospital Dawn Waddell, PharmD, BCPS Clinical Pharmacy Manager Lisa Kingdon, PharmD, BCPS Clinical Pharmacy Specialist Dawn Waddell
Reducing COPD Exacerbation Readmissions in a Community-Based Teaching Hospital Dawn Waddell, PharmD, BCPS Clinical Pharmacy Manager Lisa Kingdon, PharmD, BCPS Clinical Pharmacy Specialist Dawn Waddell
Asthma medications: Know your options - MayoClinic.com. Asthma medications: Know your options
 MayoClinic.com reprints This single copy is for your personal, noncommercial use only. For permission to reprint multiple copies or to order presentation-ready copies for distribution, use the reprints
MayoClinic.com reprints This single copy is for your personal, noncommercial use only. For permission to reprint multiple copies or to order presentation-ready copies for distribution, use the reprints
Clinical Quality Measures Summary of Upcoming Enhancements
 Upcoming coding enhancements will impact the logic behind the clinical quality indicators applicable to your practice specialty. Please refer to this grid for a summary of the coding enhancements and some
Upcoming coding enhancements will impact the logic behind the clinical quality indicators applicable to your practice specialty. Please refer to this grid for a summary of the coding enhancements and some
Clinical Policy: Omalizumab (Xolair) Reference Number: ERX.SPA.141 Effective Date:
 Clinical Policy: (Xolair) Reference Number: ERX.SPA.141 Effective Date: 03.01.14 Last Review Date: 02.18 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
Clinical Policy: (Xolair) Reference Number: ERX.SPA.141 Effective Date: 03.01.14 Last Review Date: 02.18 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
The natural history of asthma and early intervention
 The natural history of asthma and early intervention Stanley J. Szefler, MD Denver, Colo The understanding of the natural history of asthma has changed significantly during the last 4 decades, with the
The natural history of asthma and early intervention Stanley J. Szefler, MD Denver, Colo The understanding of the natural history of asthma has changed significantly during the last 4 decades, with the
Controversial Issues in the Management of Childhood Asthma: Insights from NIH Asthma Network Studies
 Controversial Issues in the Management of Childhood Asthma: Insights from NIH Asthma Network Studies Stanley J. Szefler, MD Helen Wohlberg and Herman Lambert Chair in Pharmacokinetics, Head, Pediatric
Controversial Issues in the Management of Childhood Asthma: Insights from NIH Asthma Network Studies Stanley J. Szefler, MD Helen Wohlberg and Herman Lambert Chair in Pharmacokinetics, Head, Pediatric
ONLINE DATA SUPPLEMENT - ASTHMA INTERVENTION PROGRAM PREVENTS READMISSIONS IN HIGH HEALTHCARE UTILIZERS
 R2 (REVISED MANUSCRIPT BLUE 200208-877OC) ONLINE DATA SUPPLEMENT - ASTHMA INTERVENTION PROGRAM PREVENTS READMISSIONS IN HIGH HEALTHCARE UTILIZERS Mario Castro, M.D., M.P.H. Nina A. Zimmermann R.N. Sue
R2 (REVISED MANUSCRIPT BLUE 200208-877OC) ONLINE DATA SUPPLEMENT - ASTHMA INTERVENTION PROGRAM PREVENTS READMISSIONS IN HIGH HEALTHCARE UTILIZERS Mario Castro, M.D., M.P.H. Nina A. Zimmermann R.N. Sue
Corporate Medical Policy
 Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: bronchial_thermoplasty 10/2010 3/2018 3/2019 3/2018 Description of Procedure or Service Bronchial thermoplasty
Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: bronchial_thermoplasty 10/2010 3/2018 3/2019 3/2018 Description of Procedure or Service Bronchial thermoplasty
Strep Test 87070, 87071, 87081, Pharyngitis (CWP)
 Clinical Excellence Measures Use of these codes should be appropriate to the service(s) rendered and follow the billing guidelines. For HEDIS measures the codes are from the NCQA HEDIS specifications and
Clinical Excellence Measures Use of these codes should be appropriate to the service(s) rendered and follow the billing guidelines. For HEDIS measures the codes are from the NCQA HEDIS specifications and
Cost-Motivated Treatment Changes in Commercial Claims:
 Cost-Motivated Treatment Changes in Commercial Claims: Implications for Non- Medical Switching August 2017 THE MORAN COMPANY 1 Cost-Motivated Treatment Changes in Commercial Claims: Implications for Non-Medical
Cost-Motivated Treatment Changes in Commercial Claims: Implications for Non- Medical Switching August 2017 THE MORAN COMPANY 1 Cost-Motivated Treatment Changes in Commercial Claims: Implications for Non-Medical
Medications Affecting The Respiratory System
 Medications Affecting The Respiratory System Overview Asthma is a chronic inflammatory disorder of the airways. It is an intermittent and reversible airflow obstruction that affects the bronchioles. The
Medications Affecting The Respiratory System Overview Asthma is a chronic inflammatory disorder of the airways. It is an intermittent and reversible airflow obstruction that affects the bronchioles. The
ABCBS PCMH Specifications. ARKANSAS BLUE CROSS and BLUE SHIELD An Independent Licensee of the Blue Cross and Blue Shield Association
 ABCBS PCMH 2016 Specifications An Independent Licensee of the Blue Cross and Blue Shield Association Table of Contents I. Terminology 3 II. Transformation Activities 2016 List of Activities 4 3 month 5-9
ABCBS PCMH 2016 Specifications An Independent Licensee of the Blue Cross and Blue Shield Association Table of Contents I. Terminology 3 II. Transformation Activities 2016 List of Activities 4 3 month 5-9
The use of controller medications management of pediatric bronchial asthma -
 Original Research Article The use of controller medications in the management of pediatric bronchial asthma - Dynamic patterns of LTRA Anna Todorova 1*, Antoaneta Tsvetkova 2, Silvia Mihaylova 2 1 Faculty
Original Research Article The use of controller medications in the management of pediatric bronchial asthma - Dynamic patterns of LTRA Anna Todorova 1*, Antoaneta Tsvetkova 2, Silvia Mihaylova 2 1 Faculty
Study Exposures, Outcomes:
 GSK Medicine: Coreg IR, Coreg CR, and InnoPran Study No.: WWE111944/WEUSRTP3149 Title: A nested case-control study of the association between Coreg IR and Coreg CR and hypersensitivity reactions: anaphylactic
GSK Medicine: Coreg IR, Coreg CR, and InnoPran Study No.: WWE111944/WEUSRTP3149 Title: A nested case-control study of the association between Coreg IR and Coreg CR and hypersensitivity reactions: anaphylactic
Type of intervention Treatment. Economic study type Cost-effectiveness analysis.
 Cost-effectiveness of salmeterol/fluticasone propionate combination product 50/250 micro g twice daily and budesonide 800 micro g twice daily in the treatment of adults and adolescents with asthma Lundback
Cost-effectiveness of salmeterol/fluticasone propionate combination product 50/250 micro g twice daily and budesonide 800 micro g twice daily in the treatment of adults and adolescents with asthma Lundback
ASSOCIATION OF ASTHMA CONTROL WITH HEALTH CARE UTILIZATION A PROSPECTIVE EVALUATION
 Online Supplement for: ASSOCIATION OF ASTHMA CONTROL WITH HEALTH CARE UTILIZATION A PROSPECTIVE EVALUATION METHODS Population The Northwest Region of Kaiser Permanente (KPNW) is a large, federally qualified,
Online Supplement for: ASSOCIATION OF ASTHMA CONTROL WITH HEALTH CARE UTILIZATION A PROSPECTIVE EVALUATION METHODS Population The Northwest Region of Kaiser Permanente (KPNW) is a large, federally qualified,
GSK Medicine: Study Number: Title: Rationale: Study Period: Objectives: Indication: Study Investigators/Centers: Research Methods: Data Source
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
Chronic Obstructive Pulmonary Disease (COPD) Clinical Guideline
 Chronic Obstructive Pulmonary Disease (COPD) Clinical These clinical guidelines are designed to assist clinicians by providing an analytical framework for the evaluation and treatment of patients. They
Chronic Obstructive Pulmonary Disease (COPD) Clinical These clinical guidelines are designed to assist clinicians by providing an analytical framework for the evaluation and treatment of patients. They
Drug prescriptions (Pharm) Exposure (36/48 months)
 ANNEX SECTION PART A - Study design: Figure 1 overview of the study design Drug prescriptions (Pharm) 2006-2010 Exposure (36/48 months) flexible time windows (e.g. 90 days) time index date (hospital discharge)
ANNEX SECTION PART A - Study design: Figure 1 overview of the study design Drug prescriptions (Pharm) 2006-2010 Exposure (36/48 months) flexible time windows (e.g. 90 days) time index date (hospital discharge)
Research in Real Life
 Research in Real Life Study 1: Exploratory study - identifying the benefits of pmdi versus Diskus for delivering fluticasone/salmeterol combination therapy in patients with chronic obstructive pulmonary
Research in Real Life Study 1: Exploratory study - identifying the benefits of pmdi versus Diskus for delivering fluticasone/salmeterol combination therapy in patients with chronic obstructive pulmonary
Title Page. Title Behavioral Influences on Controller Inhaler Use for Persistent Asthma in a Patient-Centered Medical Home
 Title Page Title Behavioral Influences on Controller Inhaler Use for Persistent Asthma in a Patient-Centered Medical Home Authors Sue J. Lee a, Kathleen J. Pincus a, PharmD, BCPS, Adrienne A. Williams,
Title Page Title Behavioral Influences on Controller Inhaler Use for Persistent Asthma in a Patient-Centered Medical Home Authors Sue J. Lee a, Kathleen J. Pincus a, PharmD, BCPS, Adrienne A. Williams,
Low Dose Inhaled Corticosteroid in Asthma & Prevention of Acute Coronary Syndrome MMJ 2010; 9(1):57 62
 Low Dose Inhaled Corticosteroid in Asthma & Prevention of Acute Coronary Syndrome MMJ 2010; 9(1):57 62 Abdul Hameed Al Qaseer*, Shaheed Al Hilli** *C.A.B.M., Ass. Professor, Department of Medicine, Al
Low Dose Inhaled Corticosteroid in Asthma & Prevention of Acute Coronary Syndrome MMJ 2010; 9(1):57 62 Abdul Hameed Al Qaseer*, Shaheed Al Hilli** *C.A.B.M., Ass. Professor, Department of Medicine, Al
During the last decade, disease management has. Asthma Disease Management: Regression to the Mean or Better? MANAGERIAL
 Asthma Disease Management: Regression to the Mean or Better? David Tinkelman, MD; and Steve Wilson, MA Objectives: To assess the effectiveness of disease management as an adjunct to treatment for chronic
Asthma Disease Management: Regression to the Mean or Better? David Tinkelman, MD; and Steve Wilson, MA Objectives: To assess the effectiveness of disease management as an adjunct to treatment for chronic
Kirthi Gunasekera MD Respiratory Physician National Hospital of Sri Lanka Colombo,
 Kirthi Gunasekera MD Respiratory Physician National Hospital of Sri Lanka Colombo, BRONCHODILATORS: Beta Adrenoreceptor Agonists Actions Adrenoreceptor agonists have many of the same actions as epinephrine/adrenaline,
Kirthi Gunasekera MD Respiratory Physician National Hospital of Sri Lanka Colombo, BRONCHODILATORS: Beta Adrenoreceptor Agonists Actions Adrenoreceptor agonists have many of the same actions as epinephrine/adrenaline,
Policy Evaluation: Step Therapy Prior Authorization of Combination Inhaled Corticosteroid / Long-Acting Beta-Agonists
 Drug Use Research & Management Program OHA Division of Medical Assistance Programs 500 Summer Street NE, E35; Salem, OR 97301-1079 Phone 503-947-5220 Fax 503-947-1119 Policy Evaluation: Step Therapy Prior
Drug Use Research & Management Program OHA Division of Medical Assistance Programs 500 Summer Street NE, E35; Salem, OR 97301-1079 Phone 503-947-5220 Fax 503-947-1119 Policy Evaluation: Step Therapy Prior
Asthma is a common chronic medical condition that is associated
 Relationship of Asthma Control to Asthma Exacerbations Using Surrogate Markers Within a Managed Care Database Michael Schatz, MD, MS; Robert S. Zeiger, MD, PhD; Su-Jau T. Yang, PhD; Wansu Chen, MS; William
Relationship of Asthma Control to Asthma Exacerbations Using Surrogate Markers Within a Managed Care Database Michael Schatz, MD, MS; Robert S. Zeiger, MD, PhD; Su-Jau T. Yang, PhD; Wansu Chen, MS; William
Asthma Management Updates: A Focus on Long-acting Muscarinic Antagonists and Intermittent Inhaled Corticosteroid Dosing
 Asthma Management Updates: A Focus on Long-acting Muscarinic Antagonists and Intermittent Inhaled Corticosteroid Dosing Diana M. Sobieraj, PharmD, BCPS Assistant Professor University of Connecticut School
Asthma Management Updates: A Focus on Long-acting Muscarinic Antagonists and Intermittent Inhaled Corticosteroid Dosing Diana M. Sobieraj, PharmD, BCPS Assistant Professor University of Connecticut School
Inhaler Confusion. Today s Speaker Dr. Randall Brown. Director of Asthma Programs 6/7/2016. Dr. Randall Brown March 31, 2016
 + Inhaler Confusion Dr. Randall Brown March 31, 2016 + Today s Speaker Dr. Randall Brown Director of Asthma Programs Center for Managing Chronic Disease University of Michigan 1 ASTHMA ESSENTIALS IN PRIMARY
+ Inhaler Confusion Dr. Randall Brown March 31, 2016 + Today s Speaker Dr. Randall Brown Director of Asthma Programs Center for Managing Chronic Disease University of Michigan 1 ASTHMA ESSENTIALS IN PRIMARY
Louisiana Medicaid. Provider Update. Volume 27, Issue 3 May/June UNISYS Health Information Management Acquired. by Molina Healthcare
 Louisiana Medicaid Provider Update Volume 27, Issue 3 May/June 2010 UNISYS Health Information Management Acquired by Molina Healthcare Effective May 1, 2010, Molina Healthcare purchased the Health Information
Louisiana Medicaid Provider Update Volume 27, Issue 3 May/June 2010 UNISYS Health Information Management Acquired by Molina Healthcare Effective May 1, 2010, Molina Healthcare purchased the Health Information
Shoroq Mahmoud Altawalbeh. Submitted to the Graduate Faculty of. the School of Pharmacy in partial fulfillment. of the requirements for the degree of
 ADDITION OF LEUKOTRIENE RECEPTOR ANTAGONISTS VERSUS LONG ACTING BETA AGONISTS TO INHALED CORTICOSTEROID THERAPY FOR ASTHMA TREATMENT IN OLDER ADULTS: A RETROSPECTIVE DATA ANALYSIS OF EFFECTIVENESS, SAFETY
ADDITION OF LEUKOTRIENE RECEPTOR ANTAGONISTS VERSUS LONG ACTING BETA AGONISTS TO INHALED CORTICOSTEROID THERAPY FOR ASTHMA TREATMENT IN OLDER ADULTS: A RETROSPECTIVE DATA ANALYSIS OF EFFECTIVENESS, SAFETY
Short-acting, inhaled 2-adrenergic receptor agonists
 CLINICAL STUDIES Inhaled Beta-2 Adrenergic Receptor Agonists and Primary Cardiac Arrest Rozenn N. Lemaitre, PhD, David S. Siscovick, MD, PhD, Bruce M. Psaty, MD, PhD, Rachel M. Pearce, MS, Trivellore E.
CLINICAL STUDIES Inhaled Beta-2 Adrenergic Receptor Agonists and Primary Cardiac Arrest Rozenn N. Lemaitre, PhD, David S. Siscovick, MD, PhD, Bruce M. Psaty, MD, PhD, Rachel M. Pearce, MS, Trivellore E.
Jae Jin An, Ph.D. Michael B. Nichol, Ph.D.
 IMPACT OF MULTIPLE MEDICATION COMPLIANCE ON CARDIOVASCULAR OUTCOMES IN PATIENTS WITH TYPE II DIABETES AND COMORBID HYPERTENSION CONTROLLING FOR ENDOGENEITY BIAS Jae Jin An, Ph.D. Michael B. Nichol, Ph.D.
IMPACT OF MULTIPLE MEDICATION COMPLIANCE ON CARDIOVASCULAR OUTCOMES IN PATIENTS WITH TYPE II DIABETES AND COMORBID HYPERTENSION CONTROLLING FOR ENDOGENEITY BIAS Jae Jin An, Ph.D. Michael B. Nichol, Ph.D.
ASTHMA IN THE PEDIATRIC POPULATION
 ASTHMA IN THE PEDIATRIC POPULATION SEARCH Rotation 2 August 23, 2010 Objectives Define asthma as a chronic disease Discuss the morbidity of asthma in pediatrics Discuss a few things that a health center
ASTHMA IN THE PEDIATRIC POPULATION SEARCH Rotation 2 August 23, 2010 Objectives Define asthma as a chronic disease Discuss the morbidity of asthma in pediatrics Discuss a few things that a health center
Burden of asthma with elevated blood eosinophil levels
 Casciano et al. BMC Pulmonary Medicine (2016) 16:100 DOI 10.1186/s12890-016-0263-8 RESEARCH ARTICLE Burden of asthma with elevated blood eosinophil levels Julian Casciano 1*, Jerry A. Krishnan 2, Mary
Casciano et al. BMC Pulmonary Medicine (2016) 16:100 DOI 10.1186/s12890-016-0263-8 RESEARCH ARTICLE Burden of asthma with elevated blood eosinophil levels Julian Casciano 1*, Jerry A. Krishnan 2, Mary
Asthma in Pregnancy. Asthma. Chronic Airway Inflammation. Objective Measures of Airflow. Peak exp. flow rate (PEFR)
 Chronic Airway Inflammation Asthma in Pregnancy Robin Field, MD Maternal Fetal Medicine Kaiser Permanente San Francisco Asthma Chronic airway inflammation increased airway responsiveness to a variety of
Chronic Airway Inflammation Asthma in Pregnancy Robin Field, MD Maternal Fetal Medicine Kaiser Permanente San Francisco Asthma Chronic airway inflammation increased airway responsiveness to a variety of
Optum Research Database Proportion of asthma patients with prescription fills for rescue and controller medications by year,
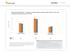 ASTHMA CHART 1 Optum Research Database Proportion of asthma patients with prescription fills for rescue and controller medications by year, 215-216 7 6 5 4 3 2 1 1 Controller medication 1 Rescue medication
ASTHMA CHART 1 Optum Research Database Proportion of asthma patients with prescription fills for rescue and controller medications by year, 215-216 7 6 5 4 3 2 1 1 Controller medication 1 Rescue medication
LCD L B-type Natriuretic Peptide (BNP) Assays
 LCD L30559 - B-type Natriuretic Peptide (BNP) Assays Contractor Information Contractor Name: Novitas Solutions, Inc. Contractor Number(s): 12501, 12502, 12101, 12102, 12201, 12202, 12301, 12302, 12401,
LCD L30559 - B-type Natriuretic Peptide (BNP) Assays Contractor Information Contractor Name: Novitas Solutions, Inc. Contractor Number(s): 12501, 12502, 12101, 12102, 12201, 12202, 12301, 12302, 12401,
Asthma represents a significant burden to the healthcare system.
 n clinical n Predicting Asthma Outcomes in Commercially Insured and Medicaid Populations Richard H. Stanford, PharmD, MS; Manan B. Shah, PharmD, PhD; Anna O. D Souza, BPharm, PhD; and Michael Schatz, MD,
n clinical n Predicting Asthma Outcomes in Commercially Insured and Medicaid Populations Richard H. Stanford, PharmD, MS; Manan B. Shah, PharmD, PhD; Anna O. D Souza, BPharm, PhD; and Michael Schatz, MD,
SUMMARY THIS IS A PRINTED COPY OF AN ELECTRONIC DOCUMENT. PLEASE CHECK ITS VALIDITY BEFORE USE.
 i SUMMARY ZENECA PHARMACEUTICALS FINISHED PRODUCT: ACTIVE INGREDIENT: ACCOLATE zafirlukast (ZD9188) Trial title (number): A Dose-ranging, Safety and Efficacy Trial with Zafirlukast (ACCOLATE ) in the Treatment
i SUMMARY ZENECA PHARMACEUTICALS FINISHED PRODUCT: ACTIVE INGREDIENT: ACCOLATE zafirlukast (ZD9188) Trial title (number): A Dose-ranging, Safety and Efficacy Trial with Zafirlukast (ACCOLATE ) in the Treatment
12/18/2017. Disclosures. Asthma Management Updates: A Focus on Long-acting Muscarinic Antagonists and Intermittent Inhaled Corticosteroid Dosing
 Asthma Management Updates: A Focus on Long-acting Muscarinic Antagonists and Intermittent Inhaled Corticosteroid Dosing Diana M. Sobieraj, PharmD, BCPS Assistant Professor University of Connecticut School
Asthma Management Updates: A Focus on Long-acting Muscarinic Antagonists and Intermittent Inhaled Corticosteroid Dosing Diana M. Sobieraj, PharmD, BCPS Assistant Professor University of Connecticut School
NG80. Asthma: diagnosis, monitoring and chronic asthma management (NG80)
 Asthma: diagnosis, monitoring and chronic asthma management (NG80) NG80 NICE has checked the use of its content in this product and the sponsor has had no influence on the content of this booklet. NICE
Asthma: diagnosis, monitoring and chronic asthma management (NG80) NG80 NICE has checked the use of its content in this product and the sponsor has had no influence on the content of this booklet. NICE
Section II: Detailed Measure Specifications
 Section II: Detailed Measure Specifications Provide sufficient detail to describe how a measure would be calculated from the recommended data sources, uploading a separate document (+ Upload attachment)
Section II: Detailed Measure Specifications Provide sufficient detail to describe how a measure would be calculated from the recommended data sources, uploading a separate document (+ Upload attachment)
Economic Burden in Direct Costs of Concomitant Chronic Obstructive Pulmonary Disease and Asthma in a Medicare Advantage Population
 RESEARCH Economic Burden in Direct Costs of Concomitant Chronic Obstructive Pulmonary Disease Christopher M. Blanchette, PhD; Benjamin Gutierrez, PhD; Caron Ory, RN, MSN; Eunice Chang, PhD; and Manabu
RESEARCH Economic Burden in Direct Costs of Concomitant Chronic Obstructive Pulmonary Disease Christopher M. Blanchette, PhD; Benjamin Gutierrez, PhD; Caron Ory, RN, MSN; Eunice Chang, PhD; and Manabu
Asthma. Definition. Symptoms
 Asthma Definition Asthma is a condition in which your airways narrow and swell and produce extra mucus. This can make breathing difficult and trigger coughing, wheezing and shortness of breath. For some
Asthma Definition Asthma is a condition in which your airways narrow and swell and produce extra mucus. This can make breathing difficult and trigger coughing, wheezing and shortness of breath. For some
Asthma Update A/Prof. John Abisheganaden. Senior Consultant, Dept Of Respiratory & Crit Care Medicine Tan Tock Seng Hospital
 Asthma Update - 2013 A/Prof. John Abisheganaden Senior Consultant, Dept Of Respiratory & Crit Care Medicine Tan Tock Seng Hospital Asthma A complex syndrome Multifaceted disease Heterogeneous Genetic and
Asthma Update - 2013 A/Prof. John Abisheganaden Senior Consultant, Dept Of Respiratory & Crit Care Medicine Tan Tock Seng Hospital Asthma A complex syndrome Multifaceted disease Heterogeneous Genetic and
Bridges to Excellence Chronic Obstructive Pulmonary Disease Care Recognition Program Guide
 Bridges to Excellence Chronic Obstructive Pulmonary Disease Care Recognition Program Guide Altarum Bridges to Excellence 3520 Green Court, Suite 300 Ann Arbor, MI 48105 bte@altarum.org www.bridgestoexcellence.org
Bridges to Excellence Chronic Obstructive Pulmonary Disease Care Recognition Program Guide Altarum Bridges to Excellence 3520 Green Court, Suite 300 Ann Arbor, MI 48105 bte@altarum.org www.bridgestoexcellence.org
Biologic Agents in the treatment of Severe Asthma
 Biologic Agents in the treatment of Severe Asthma Daniel L Maxwell, D.O., FACOI, FAASM Clinical Assistant Professor of Medicine Michigan State University College of Osteopathic Medicine College of Human
Biologic Agents in the treatment of Severe Asthma Daniel L Maxwell, D.O., FACOI, FAASM Clinical Assistant Professor of Medicine Michigan State University College of Osteopathic Medicine College of Human
Provider Respiratory Inservice
 Provider Respiratory Inservice 2 Welcome Opening Remarks We will cover: Definition of Asthma & COPD Evidence based guidelines for diagnosis, evaluation, and management of asthma Evidence based guidelines
Provider Respiratory Inservice 2 Welcome Opening Remarks We will cover: Definition of Asthma & COPD Evidence based guidelines for diagnosis, evaluation, and management of asthma Evidence based guidelines
GSK Medicine: salmeterol, Salmeterol+Fluticasone proprionate, fluticasone propionate, beclomethasone
 GSK Medicine: salmeterol, Salmeterol+Fluticasone proprionate, fluticasone propionate, beclomethasone Study No.: WWE111984/WEUSRTP2640/EPI40528 Title: The Asthma Death Case Control Study (ADCCS): Association
GSK Medicine: salmeterol, Salmeterol+Fluticasone proprionate, fluticasone propionate, beclomethasone Study No.: WWE111984/WEUSRTP2640/EPI40528 Title: The Asthma Death Case Control Study (ADCCS): Association
Respiratory Subcommittee of PTAC meeting held 5 February (minutes for web publishing)
 Respiratory Subcommittee of PTAC meeting held 5 February 2010 (minutes for web publishing) Respiratory Subcommittee minutes are published in accordance with the Terms of Reference for the Pharmacology
Respiratory Subcommittee of PTAC meeting held 5 February 2010 (minutes for web publishing) Respiratory Subcommittee minutes are published in accordance with the Terms of Reference for the Pharmacology
The adult therapeutic substitution protocol follows:
 Therapeutic Substitution of Albuterol - Implemented 1/15/2013 After years of evaluation, comparative studies have not consistently demonstrated clinical or safety advantages of levalbuterol over racemic
Therapeutic Substitution of Albuterol - Implemented 1/15/2013 After years of evaluation, comparative studies have not consistently demonstrated clinical or safety advantages of levalbuterol over racemic
COPD. Breathing Made Easier
 COPD Breathing Made Easier Catherine E. Cooke, PharmD, BCPS, PAHM Independent Consultant, PosiHleath Clinical Associate Professor, University of Maryland School of Pharmacy This program has been brought
COPD Breathing Made Easier Catherine E. Cooke, PharmD, BCPS, PAHM Independent Consultant, PosiHleath Clinical Associate Professor, University of Maryland School of Pharmacy This program has been brought
