Blümcke et al. 2016, Epilepsy-associated brain tumours in the revised 2016 WHO classification page 1
|
|
|
- Milo Flowers
- 6 years ago
- Views:
Transcription
1 Blümcke et al. 2016, Epilepsy-associated brain tumours in the revised 2016 WHO classification page 1 Low-grade epilepsy-associated neuro-epithelial tumours (LEAT) in the 2016 WHO classification of tumours of the nervous system: what s old, new and blue? Ingmar Blümcke 1, Eleonora Aronica 2,3, Albert Becker 4, Davi d Capper 5, Roland Coras 1, Mrinalini Honavar 6, Thomas S. Jacques 7, Katja Kobow 1, Hajime Miyata 8, Angelika Mühlebner 3,9, Jose Pimentel 10, Figen Söylemezoğlu 11, Maria Thom 12 1 Department of Neuropathology, University Hospital Erlangen, Germany 2 Brain Center Rudolf Magnus, Department of Pathology, University Medical Center Utrecht, The Netherlands 3 Department of (Neuro)Pathology, Academic Medisch Centrum (AMC) and Swammerdam Institute for Life Sciences, Center for Neuroscience, University of Amsterdam; Stichting Epilepsie Instellingen Nederland (SEIN), The Netherlands 4 Department of Neuropathology, University of Bonn Medical Centre, Bonn, Germany 5 Department of Neuropathology, Institute of Pathology, Ruprecht-Karls-University Heidelberg and Clinical Cooperation Unit Neuropathology, German Consortium for Translational Cancer Research (DKTK), German Cancer Research Center (DKFZ), Heidelberg, Germany 6 Department of Anatomic Pathology, Hospital Pedro Hispano, Matosinhos, Portugal 7 Developmental Biology and Cancer Section, UCL Great Ormond Street Institute of Child Health and the Department of Histopathology, Great Ormond Street Hospital for Children, London, United Kingdom 8 Department of Neuropathology, Research Institute for Brain and Blood Vessels Akita, Akita, Japan 9 Department of Pediatrics, Medical University Vienna, Vienna, Austria 10 Laboratory of Neuropathology, Department of Neurology, Hospital de Santa Maria (CHLN), Lisbon, Portugal 11 Department of Pathology, Faculty of Medicine, Hacettepe University, Ankara, Turkey 12 Dept. of Neuropathology, Institute of Neurology, University College London, UK
2 Blümcke et al. 2016, Epilepsy-associated brain tumours in the revised 2016 WHO classification page 2 Abstract Histological classification of brain tumours using the World Health Organization (WHO) system is the gold standard for treatment decision. Dizzying developments in moleculargenetic technology and research have rapidly advanced our current understanding of neurooncology. As a consequence, the WHO has invited their expert panels to revise the current classification system of tumours of the nervous system and to introduce for the first time, a molecular-genetic approach for selected tumour entities, thus setting a new gold standard in histopathology. In May 2016, the revised 4 th Edition of the blue book for brain tumours was released and will have a major impact in stratifying diagnosis and treatment. However, as in previous editions, low grade neuro-epithelial tumours presenting with a mixed neuronal and glial phenotype and early-onset epilepsy as the major neurological symptom (herein designated as low-grade epilepsy-associated neuro-epithelial tumours, LEAT) have not been the subject of significant histological or molecular-genetic innovations, which will be critically reviewed by the Neuropathology Task Force of the International League against Epilepsy (ILAE). The Task Force also proposes a roadmap of how to develop a clinically meaningful and integrated clinico-pathological-genetic classification system for LEAT in the near future. Introduction The WHO series of organ-specific tumour classification systems is the gold standard for histological tumour typing, and compiled in the blue book series. Hence, timely updates are required for each tumour entity to take account of improved knowledge of the underlying pathomechanisms and best patient management as new targets for precision medicine become available. In May 2016, the revised WHO classification of tumours of the central nervous system was released 1, 2 and has already revolutionized the arena of neurooncology. It is for the first time in the blue book series of brain tumours that moleculargenetic information is required for an integrated phenotypic-genotypic diagnosis 3, and this applies to diffusely infiltrating glial and embryonal tumours. Capacity building for moleculargenetic analysis is an ongoing challenge, but many centres around the world have already
3 Blümcke et al. 2016, Epilepsy-associated brain tumours in the revised 2016 WHO classification page 3 implemented this new concept. Increasing availability of immunohistochemical surrogates for molecular genetic alterations 4 will also help in the practical application of the new WHO classification system. It was the consensus of the WHO panel to acknowledge the rapid developments in neuro-oncology since the breakthrough discoveries of 1p/19q co-deletions as diagnostic, prognostic and predictive biomarker in oligodendrogliomas 5-8, followed by discovery of MGMT (O6-methylguanine-DNA methyltransferase) hypermethylation in glioblastomas as a predictive biomarker for treatment response to temozolomide 9, 10, and the discovery of the R132H mutation in the IDH1 gene in diffuse gliomas 11, 12, which can also be detected immunohistochemically 13, 14.The growing number of randomized trials for best treatment of brain tumours, in which patients were stratified by molecular-genetic analysis of microscopically-reviewed surgical tissue, were most helpful to validate clinically meaningful patient cohorts 11, However, this successful multidisciplinary strategy has not been developed nor systematically applied for LEAT, and tumour entities lacking ample evidence for distinct molecular-genetic signatures have fallen behind this rapidly developing process. The everexpanding gulf between Virchow s original concepts of cellular pathology (as a subjective diagnosis based on education and experience) and the evolving era of molecular pathology (as an objective diagnosis obtained from laboratory tests) will further divide the field into tumours with a targeted tumour therapy based on defined molecular alterations and those without. The latter applies in particular to rare low-grade tumour entities. These are difficult to recruit prospectively in sufficiently large series to generate meaningful survival data in reasonable time periods. Indeed, neurodevelopmental tumours with a mixed glial and neuronal phenotype, a benign course, temporal lobe localization, and seizure onset at a young age as a major clinical symptom did not meet current WHO criteria for integrated histological and genetic classification. Such low-grade epilepsy-associated brain tumours (LEAT) cover a large spectrum of neuropathological entities (Table 1), and represent the second largest group of patients submitted to epilepsy surgery 19. The International League against Epilepsy (ILAE) has charged its Commission on Diagnostic Methods and Neuropathology Task Force to address this controversial topic and we will discuss herein both, conceptual benefits of an integrated phenotypic-genotypic approach and also how to bridge the current knowledge gap.
4 Blümcke et al. 2016, Epilepsy-associated brain tumours in the revised 2016 WHO classification page 4 The 2016 WHO classification system: what s new? The 2016 WHO classification includes a number of novel tumour entities, such as the diffuse midline glioma, H3K27M mutated (WHO IV ) with predominantly astrocytic differentiation, occurring mostly in children 20. The presence of a K27M mutation in either histone H3F3A or HIST1H3B/C is mandatory for the diagnosis of this tumour. With respect to epilepsyassociated brain tumours, a gangliocytic tumour was recently reported in a total of 12 adults presenting with temporal lobe epilepsy 21, 22. This lesion is now included in the WHO blue book under the name of multinodular and vacuolating neuronal tumour of the cerebrum. There is no grade assigned yet and it is unclear if it is neoplastic or hamartomatous. It is beyond the scope of this review, however, to systematically list all newly introduced tumour entities. The most important innovation in the new WHO 2016 classification is the panel of molecular genetic tests added to the neuropathologist s armamentarium for studying and classifying brain tumours 2, and which has also helped to discover clinically meaningful new entities 17. The integrated histopathology report should describe, therefore, not only the cellular (microscopic) composition of tumour tissue but also their molecular (genetic) signature 3. As a prominent example, a genetically driven tumour classification applies for all diffuse astrocytic and oligodendroglial tumours. The histological discrimination between astrocytomas, oligodendrogliomas and mixed oligo-astrocytomas has provoked longstanding controversy in past decades in trying to decipher strict histological criteria in both low or high grade subtypes 23. It has been long recognized that the so called clear cell ( oligodendroglial ) component was associated with a better response to chemotherapy 6, yet, interobserver agreement during histological work-up of surgical brain tumour specimens remained poor 24. To solve this ongoing controversy, the WHO now refers to IDH and 1p/19q as molecular-genetic markers to objectively differentiate the spectrum of histopathologically diverse gliomas into clinically meaningful subgroups. Clinical trials confirmed that IDH1 mutated gliomas have a better prognosis than IDH1 wildtype 25 and that 1p/19q co-deletions are strongly associated with favourable outcome in patients with anaplastic gliomas 23, 26. The 2016 WHO classification system: what s old?
5 Blümcke et al. 2016, Epilepsy-associated brain tumours in the revised 2016 WHO classification page 5 Low-grade epilepsy associated neuro-epithelial tumours remain an ever challenging issue for histopathological classification throughout all editions of the WHO blue book, with a remarkable history of newly introduced tumour entities (Table 2). These tumours consist mostly of neuronal and mixed neuronal-glial variants as well as supratentorial low-grade gliomas (see Table 1). As a group, early-onset drug-resistant epilepsy (mean of < 15 years) is often the patient s major or only neurological symptom 19, 27. Another differentiating feature from diffusely infiltrating gliomas are the tumour s frequent localization in the temporal lobe. This group of tumours has been previously termed, and is nowadays recognized as long-term epilepsy-associated tumours (LEAT) 28, with gangliogliomas (GG) and dysembryoplastic neuroepithelial tumours (DNT) as prominent examples. The term LEAT was originally introduced by the Bonn Epilepsy Centre because of a long history of drug-resistant epilepsy (> 2 years) and the epilepsy surgery approach (i.e. intent to treat epilepsy) in all of their patients 28. Meanwhile, the clinical definition for epilepsy and drug resistant epilepsy has changed 29, 30, as has the concept of epilepsy surgery as last treatment resort 31, 32. We suggest, therefore, to change it s definition to low-grade epilepsy-associated neuroepithelial tumours (LEAT). However, this term does not meet the WHO concept for a nosological tumour classification. The same applies for terms such as Epilepsoma or Epileptoma 19, 33. Compared to malignant gliomas, meningiomas or brain metastases, LEAT are rare, representing approximately 2-5% of the entire brain tumour cohort 34. However, they are the second most common lesion in patients submitted to epilepsy surgery. The very broad histological spectrum of these neoplasms is another intriguing observation. This relates to their variable composition of astroglia, oligodendroglia, other clear cells or neurones, inflammatory cellular infiltrates, calcification or protein aggregation, and also to their variable patterns of papillary, rosetted, or nodular growth (Figure 1). Diffuse infiltration with clusters remote from the tumour mass is another frequent observation 19, 27. Associated focal cortical dysplasia has been reported in vastly different frequencies, thereby representing another complex issue in need of clarification 19, 27. Published evidence remains heterogeneous and controversial 19, 27, 35, 36, and the WHO panel could not develop or propose an integrated genotype-phenotype classification and grading system at present. Nevertheless, with recent studies advocating adjuvant chemo-radiation therapy in addition to surgery in low-grade gliomas 16 it is becoming increasingly important that LEAT are clearly distinguishable from IDH1/2-wildtype low-grade gliomas to avoid any hazards of over-
6 Blümcke et al. 2016, Epilepsy-associated brain tumours in the revised 2016 WHO classification page 6 treatment 37. Making the distinction is a challenge for the neuropathologist 37. Improved imaging, leading to earlier surgery before recognition as an long-term epilepsy-associated tumour rather than a tumour presenting with seizures, sometimes piece-meal removal and finally the large variety of histological appearances, may result in an incorrect diagnosis as a diffuse low-grade glioma. The 2016 WHO classification system: what s blue? As histopathological classification systems will be continuously updated and revised, the WHO s current roadmap towards better understanding of carcinogenesis and personalized treatment needs to be adopted to LEAT. It is now generally accepted and also recognized in the 2016 WHO classification, that LEATs lack IDH1 or IDH2 mutations as well as 1p/19q codeletions 11, 36, 38, 39. Published literature on prevalent molecular alterations in LEAT point to the RAS/RAF/MAPK pathway and the PI3K/AKT/mTOR pathway (Figure 2). BRAF V600E mutations were most consistently reported as genetic driver in gangliogliomas (18-56%). It s variable detection also in DNT (0-50%) may reflect, however, the aforementioned difficulties in separating both tumour entities at the microscopic level (see also 40, 49 ). As BRAF V600E mutations were observed also in pleomorphic xanthoastrocytomas 42 and pilocytic astrocytomas 42, 50, their presence cannot be regarded yet as specific for a given tumour entity. In DNT, tyrosine kinase activating FGFR1 gene mutations prevail (58-82%) 40, 49, whereas MYB/MYBL1 alterations were encountered in 87% of angiocentric gliomas 40, another LEAT tumour entity associated with drug-resistant epilepsy and early seizure onset 51. A methylation-based classification from visually selected tumour regions of formalin fixed and paraffin embedded tissue represents a promisingnew option 40, 52-54, but has not been systematically applied and validated for the entire LEAT spectrum. Use of mutationspecific antibodies, i.e. directed against the V600E BRAF mutation 55, may help to further explore the extent of cellular and genetic mosaicism 46, 48, 56. It needs to be shown, however, if such features also contribute to the epileptogenic phenotype. These studies need a careful design based on a validated histopathological classification scheme. In order to further develop this field, the Task Force recommends a specific scenario and environment for randomized controlled trials (RCT; Figure 3), which are largely missing in the
7 Blümcke et al. 2016, Epilepsy-associated brain tumours in the revised 2016 WHO classification page 7 field of LEAT. Given that the majority of these tumours are rare and grow slowly, RCTs will need to include multiple centres to recruit sufficient patient numbers within a reasonable time period. An additional goal should be to develop reference pathology centres and biorepositories of surgical brain specimens and matched blood samples across all different continents to allow for a systematic molecular testing, keeping pace with new technologies or biomarkers as they become available. Reliable assessment of the biological behaviour of a given LEAT and risk for malignant progression (reported as low in general with documented cases of malignant progression 57 ) needs clarification, as the current WHO edition did not specify atypical LEAT variants (WHO II ). The tumour s epileptogenic potential to irritate remote cortical areas or recruit remote cortical networks is another challenging issue in need of clarification. Seizure semiology is determined by a tumour s localization within the brain, i.e. in the mesial or lateral temporal lobe 37. However, satellite tumour cell infiltrates remote from the mass lesion, as described in CD34 immunoreactive gangliogliomas 19, 35, 58, may compromise a successful postsurgical outcome. These questions should be addressed in an RCT setting with systematic genotype-phenotype analysis which also help to clarify the best time period for postsurgical drug withdrawal after complete tumour resection. Although such RCTs do not aim for new interventional treatment targets, they will help to provide evidence for a comprehensive and clinically meaningful clinico-pathological and genetic tumour classification scheme in the near future. ILAE s Task Force for Neuropathology was charged to address this issue and to launch an interdisciplinary agreement study for tumour grading and classification. This Task Force has recently been successful in the implementation of international consensus classification systems for Focal Cortical Dysplasia 59 and Hippocampal Sclerosis 60 in patients with drugresistant epilepsy, and also introducing a collaborative virtual microscopy platform 61. In a first agreement study of 30 tumours randomly selected from a multicentre epilepsy surgery series (German Neuropathology Reference Centre for Epilepsy Surgery in Erlangen), agreement amongst 25 invited colleagues from 12 countries experienced in epilepsy surgery programmes reached only 40% (unpublished data, R. Coras personal communication). Use of immunohistochemical markers to more reliably differentiate histopathological epilepsyassociated neuroepithelial tumours features, i.e. CD34 and MAP2 19, 58, 62, achieved slightly better agreement in the same series of tumour specimens (unpublished data, R. Coras
8 Blümcke et al. 2016, Epilepsy-associated brain tumours in the revised 2016 WHO classification page 8 personal communication). The current situation is reminiscent, therefore, of that in oligodendrogliomas (as discussed above), and we should follow WHO s molecular-genetic vision to achieve a comprehensive and robust classification of LEAT. Conclusion By establishing an integrated phenotype-genotype diagnosis, the new 2016 WHO classification system has introduced a substantial change in our current neuropathological work-up. It represents a paradigm shift, that microscopic inspection alone is not sufficiently reliable to predict the clinical course and treatment response in a given brain tumour. In young patients with epilepsy, a distinct subgroup of brain tumours can be encountered (herein termed low-grade epilepsy-associated neuro-epithelial tumours ). They present with a large and often mixed phenotypic spectrum and are difficult to classify by existing schemes. Also, published series have reported a variable representation of genetic alterations. Randomized clinical trials have not yet been performed to approve clinically meaningful tumour entities. Therefore, the revised WHO classification 2016 contains no histopathological or molecular-genetic advances in relation to this group of tumours. Adapting WHO s vision is mandatory to establish a comprehensive clinico-pathological and genetic classification system for epilepsy-associated neuro-epithelial tumours in the near future. Acknowledgments Members of the ILAE Neuropathology Task Force have been partially supported by the Seventh Framework Programme of the European Commission (DESIRE GA # to IB; EPITARGET GA # to AB, and EA; EPISTOP # to EA). TJ was funded by the Brain Tumour Charity, Children with Cancer, Great Ormond Street Children s Charity and NIHR. References
9 Blümcke et al. 2016, Epilepsy-associated brain tumours in the revised 2016 WHO classification page 9 1. Louis, D.N. et al. WHO Classification of Tumours of the Central Nervous System (Revised 4th edition) (eds. Louis, D.N., Ohgaki, H., Wiestler, O.D. & Cavenee, W.K.) (IARC, Lyon, 2016). 2. Louis, D.N. et al. The 2016 World Health Organization Classification of Tumors of the Central Nervous System: a summary. Acta Neuropathol 131, (2016). 3. Louis, D.N. et al. International Society Of Neuropathology--Haarlem consensus guidelines for nervous system tumor classification and grading. Brain Pathol 24, (2014). 4. Reuss, D.E. et al. ATRX and IDH1-R132H immunohistochemistry with subsequent copy number analysis and IDH sequencing as a basis for an "integrated" diagnostic approach for adult astrocytoma, oligodendroglioma and glioblastoma. Acta Neuropathol 129, (2015). 5. Jenkins, R.B. et al. A t(1;19)(q10;p10) mediates the combined deletions of 1p and 19q and predicts a better prognosis of patients with oligodendroglioma. Cancer Res 66, (2006). 6. Cairncross, J.G. et al. Specific genetic predictors of chemotherapeutic response and survival in patients with anaplastic oligodendrogliomas. J Natl Cancer Inst 90, (1998). 7. Reifenberger, G. et al. in WHO classification of Tumours of the Central Nervous System (revised 4th edition) (eds. Louis, D.N., Ohgaki, H., Wiestler, O.D. & Cavanee, W.K.) (International Agency for Research on Cancer, Lyon, 2016). 8. Reifenberger, J. et al. Molecular-Genetic Analysis of Oligodendroglial Tumors Shows Preferential Allelic Deletions on 19q and 1p. American Journal of Pathology 145, (1994). 9. Stupp, R. et al. Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med 352, (2005). 10. Hegi, M.E. et al. MGMT gene silencing and benefit from temozolomide in glioblastoma. N Engl J Med 352, (2005). 11. Yan, H. et al. IDH1 and IDH2 mutations in gliomas. N Engl J Med 360, (2009). 12. Parsons, D.W. et al. An integrated genomic analysis of human glioblastoma multiforme. Science 321, (2008). 13. Capper, D. et al. Application of mutant IDH1 antibody to differentiate diffuse glioma from nonneoplastic central nervous system lesions and therapy-induced changes. Am J Surg Pathol 34, (2010). 14. Capper, D. et al. Characterization of R132H mutation-specific IDH1 antibody binding in brain tumors. Brain Pathology 20, (2010). 15. Wick, W. et al. NOA-04 randomized phase III trial of sequential radiochemotherapy of anaplastic glioma with procarbazine, lomustine, and vincristine or temozolomide. J Clin Oncol 27, (2009). 16. Buckner, J.C. et al. Radiation plus Procarbazine, CCNU, and Vincristine in Low- Grade Glioma. N Engl J Med 374, (2016). 17. Sturm, D. et al. New Brain Tumor Entities Emerge from Molecular Classification of CNS-PNETs. Cell 164, (2016). 18. Weller, M. et al. MGMT Promoter Methylation Is a Strong Prognostic Biomarker for Benefit from Dose-Intensified Temozolomide Rechallenge in Progressive Glioblastoma: The DIRECTOR Trial. Clin Cancer Res 21, (2015). 19. Blumcke, I., Aronica, E., Urbach, H., Alexopoulos, A. & Gonzalez-Martinez, J.A. A neuropathology-based approach to epilepsy surgery in brain tumors and
10 Blümcke et al. 2016, Epilepsy-associated brain tumours in the revised 2016 WHO classification page 10 proposal for a new terminology use for long-term epilepsy-associated brain tumors. Acta Neuropathol 128, (2014). 20. Castel, D. et al. Histone H3F3A and HIST1H3B K27M mutations define two subgroups of diffuse intrinsic pontine gliomas with different prognosis and phenotypes. Acta Neuropathol 130, (2015). 21. Bodi, I. et al. Two cases of multinodular and vacuolating neuronal tumour. Acta Neuropathol Commun 2, 7 (2014). 22. Huse, J.T. et al. Multinodular and vacuolating neuronal tumors of the cerebrum: 10 cases of a distinctive seizure-associated lesion. Brain Pathol 23, (2013). 23. Reifenberger, G. & Louis, D.N. Oligodendroglioma: toward molecular definitions in diagnostic neuro-oncology. J Neuropathol Exp Neurol 62, (2003). 24. Giannini, C. et al. Oligodendrogliomas: reproducibility and prognostic value of histologic diagnosis and grading. J Neuropathol Exp Neurol 60, (2001). 25. van den Bent, M.J. et al. IDH1 and IDH2 mutations are prognostic but not predictive for outcome in anaplastic oligodendroglial tumors: a report of the European Organization for Research and Treatment of Cancer Brain Tumor Group. Clin Cancer Res 16, (2010). 26. Weller, M. et al. Combined 1p/19q loss in oligodendroglial tumors: predictive or prognostic biomarker? Clin Cancer Res 13, (2007). 27. Thom, M., Blumcke, I. & Aronica, E. Long-term epilepsy-associated tumors. Brain Pathol 22, (2012). 28. Luyken, C. et al. The spectrum of long-term epilepsy-associated tumors: longterm seizure and tumor outcome and neurosurgical aspects. Epilepsia 44, (2003). 29. Fisher, R.S. et al. ILAE official report: a practical clinical definition of epilepsy. Epilepsia 55, (2014). 30. Kwan, P. et al. Definition of drug resistant epilepsy: consensus proposal by the ad hoc Task Force of the ILAE Commission on Therapeutic Strategies. Epilepsia 51, (2010). 31. Blumcke, I. et al. International recommendation for a comprehensive neuropathologic workup of epilepsy surgery brain tissue: A consensus Task Force report from the ILAE Commission on Diagnostic Methods. Epilepsia 57, (2016). 32. Wiebe, S., Blume, W.T., Girvin, J.P. & Eliasziw, M. A randomized, controlled trial of surgery for temporal-lobe epilepsy. N Engl J Med 345, (2001). 33. Japp, A., Gielen, G.H. & Becker, A.J. Recent aspects of classification and epidemiology of epilepsy-associated tumors. Epilepsia 54 Suppl 9, 5-11 (2013). 34. Jemal, A. et al. Cancer statistics, CA Cancer J Clin 56, (2006). 35. Blumcke, I. & Wiestler, O.D. Gangliogliomas: an intriguing tumor entity associated with focal epilepsies. J Neuropathol Exp Neurol 61, (2002). 36. Thom, M. et al. One hundred and one dysembryoplastic neuroepithelial tumors: an adult epilepsy series with immunohistochemical, molecular genetic, and clinical correlations and a review of the literature. J Neuropathol Exp Neurol 70, (2011). 37. Holthausen, H. & Blumcke, I. Epilepsy-associated tumours: what epileptologists should know about neuropathology, terminology, and classification systems. Epileptic Disord (2016). 38. Horbinski, C. et al. Isocitrate dehydrogenase 1 analysis differentiates gangliogliomas from infiltrative gliomas. Brain Pathol 21, (2011).
11 Blümcke et al. 2016, Epilepsy-associated brain tumours in the revised 2016 WHO classification page Prabowo, A.S. et al. Landscape of chromosomal copy number aberrations in gangliogliomas and dysembryoplastic neuroepithelial tumours. Neuropathol Appl Neurobiol (2015). 40. Qaddoumi, I. et al. Genetic alterations in uncommon low-grade neuroepithelial tumors: BRAF, FGFR1, and MYB mutations occur at high frequency and align with morphology. Acta Neuropathol (2016). 41. Dougherty, M.J. et al. Activating mutations in BRAF characterize a spectrum of pediatric low-grade gliomas. Neuro Oncol 12, (2010). 42. Schindler, G. et al. Analysis of BRAF V600E mutation in 1,320 nervous system tumors reveals high mutation frequencies in pleomorphic xanthoastrocytoma, ganglioglioma and extra-cerebellar pilocytic astrocytoma. Acta Neuropathologica 121, (2011). 43. Ramkissoon, L.A. et al. Genomic analysis of diffuse pediatric low-grade gliomas identifies recurrent oncogenic truncating rearrangements in the transcription factor MYBL1. Proc Natl Acad Sci U S A 110, (2013). 44. Roth, J.J. et al. Diagnostic application of high resolution single nucleotide polymorphism array analysis for children with brain tumors. Cancer Genet 207, (2014). 45. Cruz, G.R. et al. Analysis of KIAA1549-BRAF fusion gene expression and IDH1/IDH2 mutations in low grade pediatric astrocytomas. J Neurooncol 117, (2014). 46. Prabowo, A.S. et al. BRAF V600E mutation is associated with mtor signaling activation in glioneuronal tumors. Brain Pathol 24, (2014). 47. Zhang, J. et al. Whole-genome sequencing identifies genetic alterations in pediatric low-grade gliomas. Nature genetics 45, (2013). 48. Chappe, C. et al. Dysembryoplastic Neuroepithelial Tumors Share with Pleomorphic Xanthoastrocytomas and Gangliogliomas BRAF Mutation and Expression. Brain Pathology 23, (2013). 49. Rivera, B. et al. Germline and somatic FGFR1 abnormalities in dysembryoplastic neuroepithelial tumors. Acta Neuropathol (2016). 50. Korshunov, A. et al. Combined molecular analysis of BRAF and IDH1 distinguishes pilocytic astrocytoma from diffuse astrocytoma. Acta Neuropathologica 118, (2009). 51. Wang, M. et al. Monomorphous angiocentric glioma: a distinctive epileptogenic neoplasm with features of infiltrating astrocytoma and ependymoma. J Neuropathol Exp Neurol 64, (2005). 52. Holsken, A. et al. Adamantinomatous and papillary craniopharyngiomas are characterized by distinct epigenomic as well as mutational and transcriptomic profiles. Acta Neuropathol Commun 4, 20 (2016). 53. Johann, P.D. et al. Atypical Teratoid/Rhabdoid Tumors Are Comprised of Three Epigenetic Subgroups with Distinct Enhancer Landscapes. Cancer Cell 29, (2016). 54. Pajtler, K.W. et al. Molecular Classification of Ependymal Tumors across All CNS Compartments, Histopathological Grades, and Age Groups. Cancer Cell 27, (2015). 55. Capper, D. et al. Assessment of BRAF V600E mutation status by immunohistochemistry with a mutation-specific monoclonal antibody. Acta Neuropathol 122, 11-9 (2011). 56. Koelsche, C. et al. Mutant BRAF V600E protein in ganglioglioma is predominantly expressed by neuronal tumor cells. Acta Neuropathol 125, (2013).
12 Blümcke et al. 2016, Epilepsy-associated brain tumours in the revised 2016 WHO classification page Majores, M. et al. Tumor recurrence and malignant progression of gangliogliomas. Cancer 113, (2008). 58. Blumcke, I. et al. The CD34 epitope is expressed in neoplastic and malformative lesions associated with chronic, focal epilepsies. Acta Neuropathol 97, (1999). 59. Blumcke, I. et al. The clinico-pathological spectrum of Focal Cortical Dysplasias: a consensus classification proposed by an ad hoc Task Force of the ILAE Diagnostic Methods Commission. Epilepsia 52, (2011). 60. Blumcke, I. et al. International consensus classification of hippocampal sclerosis in temporal lobe epilepsy: a Task Force report from the ILAE Commission on Diagnostic Methods. Epilepsia 54, (2013). 61. Coras, R. et al. Good interobserver and intraobserver agreement in the evaluation of the new ILAE classification of focal cortical dysplasias. Epilepsia 53, (2012). 62. Blumcke, I. et al. Distinct expression pattern of microtubule-associated protein-2 in human oligodendrogliomas and glial precursor cells. J Neuropathol Exp Neurol 60, (2001). 63. Schramm, J., Luyken, C., Urbach, H., Fimmers, R. & Blumcke, I. Evidence for a clinically distinct new subtype of grade II astrocytomas in patients with longterm epilepsy. Neurosurgery 55, 340-7; discussion (2004). 64. Blumcke, I., Luyken, C., Urbach, H., Schramm, J. & Wiestler, O.D. An isomorphic subtype of long-term epilepsy-associated astrocytomas associated with benign prognosis. Acta Neuropathol 107, (2004). 65. Zülch, K.J. Histological typing of tumours of the central nervous system (World Health Organization, Geneva, 1979). 66. Kleihues, P., Burger, P.C. & Scheithauer, B.W. (eds.) Histological typing of tumors of the central nervous system (Springer, Berlin, Heidelberg, New York, London, Paris, Tokyo, HongKong, Barcelona, Budapest, 1993). 67. Kleihues, P. & Cavenee, W.K. (eds.) Pathology and genetics of tumors of the nervous system (International Agency for Research on Cancer, Lyon, France, 2000). 68. Louis, D.N., Ohgaki, H., Wiestler, O.D. & Cavenee, W.K. (eds.) WHO classification of tumors of the central nervous system (International Agency for Research on Cancer, Lyon, France, 2007). 69. Agarwal, S. et al. Extraventricular neurocytomas: a morphological and histogenetic consideration. A study of six cases. Pathology 43, (2011). 70. Capper, D. et al. Mutation-specific IDH1 antibody differentiates oligodendrogliomas and oligoastrocytomas from other brain tumors with oligodendroglioma-like morphology. Acta Neuropathol 121, (2011). 71. Horstmann, S. et al. Genetic and expression profiles of cerebellar liponeurocytomas. Brain Pathol 14, (2004). 72. Bridge, J.A. et al. Identification of a novel, recurrent SLC44A1-PRKCA fusion in papillary glioneuronal tumor. Brain Pathol 23, (2013). 73. Solis, O.E. et al. Rosette-forming glioneuronal tumor: a pineal region case with IDH1 and IDH2 mutation analyses and literature review of 43 cases. J Neurooncol 102, (2011). 74. Gessi, M. et al. FGFR1 mutations in Rosette-forming glioneuronal tumors of the fourth ventricle. J Neuropathol Exp Neurol 73, (2014). 75. Gessi, M. et al. Absence of KIAA1549-BRAF fusion in rosette-forming glioneuronal tumors of the fourth ventricle (RGNT). J Neurooncol 110, 21-5 (2012).
13 Blümcke et al. 2016, Epilepsy-associated brain tumours in the revised 2016 WHO classification page Rodriguez, F.J. et al. Disseminated oligodendroglial-like leptomeningeal tumor of childhood: a distinctive clinicopathologic entity. Acta Neuropathol 124, (2012). 77. Rodriguez, F.J. et al. High rate of concurrent BRAF-KIAA1549 gene fusion and 1p deletion in disseminated oligodendroglioma-like leptomeningeal neoplasms (DOLN). Acta Neuropathol 129, (2015). 78. Preuss, M. et al. Disseminated oligodendroglial-like leptomeningeal tumors: preliminary diagnostic and therapeutic results for a novel tumor entity [corrected]. J Neurooncol 124, (2015).
14 Blümcke et al. 2016, Epilepsy-associated brain tumours in the revised 2016 WHO classification page 14 Figure 1: Is it all in one? The histopathological spectrum of nodular growth in low-grade epilepsy-associated neuro-epithelial tumours Legend to Figure 1: Three examples of difficult-to-classify mixed neuronal-glial tumours in the temporal lobe, characterized by areas of nodular tumour growth (arrows in A, C, and E). All patients suffered from intractable epilepsy with seizure onset at young age. A-B: This nodule is composed of unequivocal clusters of ganglion cells (arrowhead in B), not otherwise explicable by anatomical location. C-D: Multinodular intracortical growth consisting of
15 Blümcke et al. 2016, Epilepsy-associated brain tumours in the revised 2016 WHO classification page 15 oligodendroglia-like cells and floating neurones (so-called specific glio-neuronal element, arrowhead in D). E-F: Predominatly astroglial nodule with Rosenthal fibres (arrowhead in F) and protein droplets. All tumours showed other areas of diffuse cell infiltration, with or without aberrant CD34-immunoreactive cell clusters. Other tumours of this patient cohort presented with a different composition of same cytological features. It is an ever challenging question in disease classification whether to lump these variants together as one tumor entity, i.e. epileptoma or specify n+1 new entities, i.e. ganglioglioma vs. DNT vs. composite glio-neuronal tumour? Comprehensive molecular analysis (in a RCT setting, Figure 2) will help to solve this dilemma (see text for further discussion). Scale bar in A = 1mm (applies also to C, E), scale bar in B = 50µm (applies also to D, F).
16 Blümcke et al. 2016, Epilepsy-associated brain tumours in the revised 2016 WHO classification page 16 Figure 2 Legend to Figure 2: Genetic alterations commonly found in LEAT connect 2 major pathways in cell metabolism, the RAS/RAF/MAPK pathway (green) and the PI3K/AKT/mTOR pathway (blue). Upstream in receptor signalling, mutations of FGFR1 have been described in DNT leading to activation of the RAS/RAF/MAPK pathway and the PI3K/AKT/mTOR pathway. BRAF is a component further downstream in the RAS/RAF/MAPK signalling cascade. Activation of this pathway by the V600E mutation leads to phosphrylation of MAPKs, which phosphorylate and regulate activities of partners such as transcriptions factors (CREB, c-fos) for cell proliferation and differentation. These functions made BRAF and MEK1/2 reasonable targets for therapeutic approaches (pink). On the other hand activation is tightly regulated
17 Blümcke et al. 2016, Epilepsy-associated brain tumours in the revised 2016 WHO classification page 17 by substrates of the PI3K/AKT/mTOR signalling cascade. Especially, the inhibiting function of the 2 major complexes TSC and mtor play a profound role in the regulation of autophagy, protein synthesis and cell survival. One of these transcription factors is c-myb/ mybl1 (purple) which is altered in angiocentric gliomas. All genetic alterations described are indicated with a lightening bolt. Abbreviations: - AKT (v-akt murine thymoma viral oncogene homologs 1, 2, 3) (alias: PKB): AKT1 (14q32); AKT2 (19q13); AKT3 (1q44) - DEPTOR (8q24, DEP domain containing MTOR-interacting protein) - FOS (v-fos FBJ murine osteosarcoma viral oncogene homolog) and FOS-like antigen): FOS (14q24); FOSB (19q13); FOSL1 (11q13); FOSL2 (2p23) - GRB2 (17q25) (growth factor receptor-bound protein 2) - INSR (19p13) (insulin receptor)) - MAPK (mitogen-activated protein kinase ): MAPK1 (22q11) (alias: ERK2); MAPK3 (16p11) (alias: ERK1) - MAP2K (mitogen-activated protein kinase kinase 1): MAP2K1 (15q22) (alias: MAPKK1, MEK1); MAP2K2 (19p13) (alias: MAPKK2, MEK2) - MDM2 (12q15) (transformed mouse 3T3 cell double minute 2, p53 binding protein) - MYC (8q24) (v-myc myelocytomatosis viral oncogene homolog (avian) - MTOR (1p36) (mechanistic target of rapamycin) - PI3K (phosphoinositide-3-kinase, catalytic, alpha, beta, delta, gamma polypeptide): PIK3CA (3q26); PIK3CB (3q22); PIK3CD (1p36); PIK3CG (7q22) - PKD1 (14q11) (protein kinase D) (alias: PRKCM (mu)) (Wang et al., 2006) - PTEN (10q23) (phosphatase and tensin homolog deleted on chromosome ten) - RAF (v-raf murine sarcoma viral oncogene homolog): ARAF (Xp11); BRAF (7q34); RAF1 (3p25) - RAS (RAS viral oncogene homolog): HRAS (11p15) (Harvey); KRAS (12p12) (Kirsten); NRAS (1p13) (neuroblastoma) - RHEB (7q36) (ras homolog enriched in brain) - SOS1 (2p21) (son of sevenless homolog 1) - STAT (signal transducer and activator of transcription): STAT1 (2q32); STAT2 (12q13); STAT3 (17q21); STAT4 (2q32); STAT5a (17q11); STAT5b (17q11); STAT6 (12q13)
18 Blümcke et al. 2016, Epilepsy-associated brain tumours in the revised 2016 WHO classification page 18 - TSC1 (9q34) (tuberous Sclerosis 1) - TSC2 (16p13) (tuberous Sclerosis 2)
19 Figure 3 Blümcke et al. 2016, Epilepsy-associated brain tumours in the revised 2016 WHO classification page 19
20 Blümcke et al. 2016, Epilepsy-associated brain tumours in the revised 2016 WHO classification page 20 Legend to Figure 3: A useful and doable scenario for systematic investigations of meaningful clinico-pathological LEAT entities. We believe that the basis for a successfull WHO classification is a systematic genotype-phenotype assessment, validated by randomized and controlled clinical trials. These RCTs can be also used to clarify the benefit of early AED withdrawal after complete tumour resection, but do not primarily aim at novel interventional treatment options (as new drug targets are not validated yet in LEAT).
21 Blümcke et al. 2016, Epilepsy-associated brain tumours in the revised 2016 WHO classification page 21 Table 1: The spectrum of epilepsy associated neuro-epithelial tumours Entity Numbers (%) Onset Duration Age OP ANT I 5 (0.4%) GG I 673 (48.7%) GG II /III 77 (5.6%) ISO I 29 (2.1%) DNT I 256 (18.5%) PA I 81 (5.9%) PXA II 38 (2.7%) OLIGO II /III 97 (7%) ASTRO II /III 110 (8%) Total Legend to Table 1: Summary of tumours collected in adults and children at the German Neuropathology Reference Center for Epilepsy Surgery in Erlangen. More than 80% of tumours present with seizure onset before age of 15 years (upper 6 rows), and localize in 77% to the temporal lobe 19, herein designated as low-grade epilepsy-associated neuroepithelial tumours (LEAT). Early seizure onset and temporal localization separate LEAT from semi-benign and diffusely infiltrating gliomas also encountered in epilepsy surgery series (lower 3 rows). Age at epilepsy onset (mean in years); Epilepsy duration (mean in years); Age at operation (mean in years); ANT angiocentric glioma WHO I ; GG gangliogliomas WHO I - III ; GG II is not specified by the WHO classification, but used as clinical diagnosis ( analogue WHO II ) according to the proposal by Blumcke and Wiestler 35 ; ISO - isomorphic astrocytoma variant (analogous to WHO I 63, 64 ); DNT Dysembryoplastic neuroepithelial tumour WHO I ; PA pilocytic astrocytoma WHO I ; PXA pleomorphic xantoastrocytoma WHO II ; OLIGO oligodendroglioma including mixed glioma WHO II and III ; ASTRO diffuse astrocytoma subtypes WHO II and III.
22 Blümcke et al. 2016, Epilepsy-associated brain tumours in the revised 2016 WHO classification page 22 Table 2: Evolving WHO classification of neuronal and mixed neuronal-glial tumours over years * ** Gangliocytoma Ganglioglioma Anaplastic (malignant) ganglioglioma Dysplastic gangliocytoma of cerebellum (Lhermitte- Duclos) Paraganglioma Composed predominantly of mature ganglion cells Composed of mature ganglion cells and neoplastic glial cells Grades I or II Gangliocytoma or ganglioglioma with anaplasia Grades III or IV Described as a dysplastic variant of Gangliocytoma unchanged unchanged Ganglioglioma with anaplasia in the glial component II Described separately as a tumour-like lesion Identical to extra-adrenal paraganglioma orignating in filum terminale Well differentiated, slowly growing neuroepithelial tumours composed of neoplastic, mature ganglion cells, either alone (GC ), or in combination with neoplastic glial cells (GG or II). Tumours with anaplastic glial component anaplastic GG II or IV Well differentiated, slowly growing neuroepithelial tumours composed of neoplastic, mature ganglion cells, either alone (GC ), or in combination with neoplastic glial cells (GG ), observed in patients with long-term epilepsy. Anaplastic GG, Grade III Criteria for I not established. Discussed in association with Cowden Disease under Familial tumour syndromes Not clear if neoplastic or hamartomatous. If neoplastic, corresponds to unchanged IDH1/2 24, wildtype BRAF V600E mutation in 18-25, % Anaplastic GG, Grade III PTEN (10q23)
23 Blümcke et al. 2016, Epilepsy-associated brain tumours in the revised 2016 WHO classification page 23 Desmoplastic infantile astrocytoma/ ganglioglioma Dysembryoplastic neuroepithelial tumour Central neurocytoma Mixed neuronal-glial neoplasm of infancy with desmoplasia Named as DIG Epilepsy-associated tumour with intracortical location, multinodular architecture and heterogeneous cellular composition Intraventricular tumour composed of uniform round cells with immunohistochemical (SYN) and ultrastructural features of neuronal differentiation Glial neoplasm of infancy with desmoplasia named as DIA Glial and neuronal neoplasm of infancy with desmoplasia named as DIG Simple form with unique glioneuronal element; Complex form with glial nodules in combination with glioneuronal element Reference to nonspecific histological forms (clinical features and imaging need to be considered) Intraventricular tumour composed of uniform round cells with immunohistochemical (SYN) and ultrastructural features of neuronal differentiation Reference to extraventricular examples Upgraded: WHO II Simple form with unique glioneuronal element; Complex form with glial nodules in combination with glioneuronal element Non-specific histological form remains controversial IDH1/2 24, wildtype BRAF V600E mutation in (0-36, %) FGFR1 mutation 40, 49 in 58-82% Intraventricular tumour composed of uniform round cells with immunohistochemical (SYN) and ultrastructural features of neuronal differentiation I Extraventricular Well-circumscribed IDH1/2
24 Blümcke et al. 2016, Epilepsy-associated brain tumours in the revised 2016 WHO classification page 24 neurocytoma Cerebellar liponeurocytoma Papillary glioneuronal tumour Rosette-forming glioneuronal tumour of the 4th ventricle Rare cerebellar neoplasm of adults with advanced neuronal/neurocytic and focal lipomatous differentiation, previously classified as lipomatous medulloblastoma Grades I or II neoplasm composed of uniform round cells with neuronal differentiation, located in brain parenchyma Grade not assigned 69, 70 wildtype I Same definition as in previous edition, but assigned I TP53 missense mutations in 20% 71 Tumour composed of GFAP-positive astrocytes lining hyalinised vascular pseudopapillae and synaptophysin-positive interpapillary collections of sheets of neurocytes, neurons and ganglioid cells SLC44A1-PRKCA fusion 72 Neoplasms of fourth ventricular region composed of a) uniform neurocytes forming rosettes or perivascular pseudorosettes and b) component resembling pilocytic astrocytoma IDH1/IDH2 wildtype 73
25 Blümcke et al. 2016, Epilepsy-associated brain tumours in the revised 2016 WHO classification page 25 FGFR1 mutations 74 No KIAA1549-BRAF fusion or BRAF V600E mutations 75 Multinodular and vacuolating neuronal tumour of the cerebrum Diffuse leptomeningeal glioneuronal tumour Accepted as a pattern of gangliocytoma, previously descibed in patients with epilepsy, predominately in temporal lobe 21, 22 Grade not assigned Predominant and widespread leptomeningeal growth, OLC and neuronal differentiation Deletion 1p in 59% 76, 77, IDH1/2 76, 78 wildtype KIAA1549-BRAF fusion in 75% 77 Grade not assigned
26 Blümcke et al. 2016, Epilepsy-associated brain tumours in the revised 2016 WHO classification page 26 Legend to Table 2: BRAF = v-raf murine sarcoma viral oncogene homolog B, DIA = Desmoplastic infantile astrocytoma, DIG = Desmoplastic infantile ganglioglioma, GC = Gangliocytoma, GG = ganglioglioma, GFAP = glial fibrillary acidic protein, IDH = isocitrate dehydrogenase, MAP2 = microtubule-associated protein 2, NFP = neurofilament protein, NOS = not otherwise specified, NSE = neuron-specific enolase, OLC = oligodendroglial-like cells, PTEN = phosphatase and tensin homolog, SYN = synaptophysin, TSC = tuberous sclerosis complex. * mentioned tumours were classified under Neuronal Tumours along with Ganglioneuroblastoma and Neuroblastoma in the WHO s 1979 classification. ** first appearence of the chapter entitled Neuronal and mixed neuronal-glial tumours, still including Olfactory Neuroblastoma and its variant Olfactory neuroepithelioma.
2017 Diagnostic Slide Session Case 3
 2017 Diagnostic Slide Session Case 3 Andrew Gao, MD Lili-Naz Hazrati, MD, PhD Cynthia Hawkins, MD, PhD Hospital for Sick Children and University of Toronto, Toronto, Canada Disclosures: none Clinical History
2017 Diagnostic Slide Session Case 3 Andrew Gao, MD Lili-Naz Hazrati, MD, PhD Cynthia Hawkins, MD, PhD Hospital for Sick Children and University of Toronto, Toronto, Canada Disclosures: none Clinical History
CNS pathology Third year medical students. Dr Heyam Awad 2018 Lecture 12: CNS tumours 2/3
 CNS pathology Third year medical students Dr Heyam Awad 2018 Lecture 12: CNS tumours 2/3 Pilocytic astrocytoma Relatively benign ( WHO grade 1) Occurs in children and young adults Mostly: in the cerebellum
CNS pathology Third year medical students Dr Heyam Awad 2018 Lecture 12: CNS tumours 2/3 Pilocytic astrocytoma Relatively benign ( WHO grade 1) Occurs in children and young adults Mostly: in the cerebellum
Neuropathology Evening Session: Case 3
 Neuropathology Evening Session: Case 3 Christine E. Fuller, MD Cincinnati Children s Hospital Medical Center Disclosure of Relevant Financial Relationships USCAP requires that all faculty in a position
Neuropathology Evening Session: Case 3 Christine E. Fuller, MD Cincinnati Children s Hospital Medical Center Disclosure of Relevant Financial Relationships USCAP requires that all faculty in a position
Gliomas in the 2016 WHO Classification of CNS Tumors
 Gliomas in the 2016 WHO Classification of CNS Tumors Hindi N Al-Hindi, MD, FCAP Consultant Neuropathologist and Head Section of Anatomic Pathology Department of Pathology and Laboratory Medicine King Faisal
Gliomas in the 2016 WHO Classification of CNS Tumors Hindi N Al-Hindi, MD, FCAP Consultant Neuropathologist and Head Section of Anatomic Pathology Department of Pathology and Laboratory Medicine King Faisal
8 th International Summer School for Neuropathology and Epilepsy Surgery INES July 26-29, 2018 University Hospital, Erlangen Krankenhausstr.
 8 th International Summer School for Neuropathology and Epilepsy Surgery INES 2018 July 26-29, 2018 University Hospital, Erlangen Krankenhausstr. 12 Course Directors: For more information, please contact:
8 th International Summer School for Neuropathology and Epilepsy Surgery INES 2018 July 26-29, 2018 University Hospital, Erlangen Krankenhausstr. 12 Course Directors: For more information, please contact:
CNS TUMORS. D r. Ali Eltayb ( U. of Omdurman. I ). M. Path (U. of Alexandria)
 CNS TUMORS D r. Ali Eltayb ( U. of Omdurman. I ). M. Path (U. of Alexandria) CNS TUMORS The annual incidence of intracranial tumors of the CNS ISmore than intraspinal tumors May be Primary or Secondary
CNS TUMORS D r. Ali Eltayb ( U. of Omdurman. I ). M. Path (U. of Alexandria) CNS TUMORS The annual incidence of intracranial tumors of the CNS ISmore than intraspinal tumors May be Primary or Secondary
Anaplastic Pilocytic Astrocytoma: The fusion of good and bad
 Anaplastic Pilocytic Astrocytoma: The fusion of good and bad Alexandrina Nikova 1, Charalampos-Chrysovalantis Chytoudis-Peroudis 2, Penelope Korkolopoulou 3 and Dimitrios Kanakis 4 Abstract 5 Pilocytic
Anaplastic Pilocytic Astrocytoma: The fusion of good and bad Alexandrina Nikova 1, Charalampos-Chrysovalantis Chytoudis-Peroudis 2, Penelope Korkolopoulou 3 and Dimitrios Kanakis 4 Abstract 5 Pilocytic
Classification of Diffuse Gliomas: Progress, Pearls and Pitfalls. Rob Macaulay Neuropathologist, MCC October 21, 2017
 Classification of Diffuse Gliomas: Progress, Pearls and Pitfalls Rob Macaulay Neuropathologist, MCC October 21, 2017 Objectives Explain why the designation high grade glioma is preferable to GBM for intraoperative
Classification of Diffuse Gliomas: Progress, Pearls and Pitfalls Rob Macaulay Neuropathologist, MCC October 21, 2017 Objectives Explain why the designation high grade glioma is preferable to GBM for intraoperative
Pr D.Figarella-Branger Service d Anatomie Pathologique et de Neuropathologie, La Timone, Marseille UMR 911 Inserm, Université d Aix-Marseille
 Novelties in the WHO 2016 classification of brain tumours Pr D.Figarella-Branger Service d Anatomie Pathologique et de Neuropathologie, La Timone, Marseille UMR 911 Inserm, Université d Aix-Marseille The
Novelties in the WHO 2016 classification of brain tumours Pr D.Figarella-Branger Service d Anatomie Pathologique et de Neuropathologie, La Timone, Marseille UMR 911 Inserm, Université d Aix-Marseille The
In 1988 Dumas-Duport et al. first used
 Copyright 2009, Barrow Neurological Institute Dysembryoplastic Neuroepithelial Tumor: A Review Mark Garrett, MD Jennifer Eschbacher, MD Peter Nakaji, MD Most DNETs are benign, low-grade lesions. However,
Copyright 2009, Barrow Neurological Institute Dysembryoplastic Neuroepithelial Tumor: A Review Mark Garrett, MD Jennifer Eschbacher, MD Peter Nakaji, MD Most DNETs are benign, low-grade lesions. However,
General: Brain tumors are lesions that have mass effect distorting the normal tissue and often result in increased intracranial pressure.
 1 Lecture Objectives Know the histologic features of the most common tumors of the CNS. Know the differences in behavior of the different tumor types. Be aware of the treatment modalities in the various
1 Lecture Objectives Know the histologic features of the most common tumors of the CNS. Know the differences in behavior of the different tumor types. Be aware of the treatment modalities in the various
The New WHO Classification and the Role of Integrated Molecular Profiling in the Diagnosis of Malignant Gliomas
 The New WHO Classification and the Role of Integrated Molecular Profiling in the Diagnosis of Malignant Gliomas Stefan Prokop, MD Neuropathology Fellow Hospital of the University of Pennsylvania Background
The New WHO Classification and the Role of Integrated Molecular Profiling in the Diagnosis of Malignant Gliomas Stefan Prokop, MD Neuropathology Fellow Hospital of the University of Pennsylvania Background
Case #3. USCAP Neuropathology Evening Seminar/Companion Meeting
 Case #3 USCAP Neuropathology Evening Seminar/Companion Meeting Clinical History A 71-year year-old man presented with a 4-4 week history of word finding difficulty. An initial screening head CT followed
Case #3 USCAP Neuropathology Evening Seminar/Companion Meeting Clinical History A 71-year year-old man presented with a 4-4 week history of word finding difficulty. An initial screening head CT followed
I have no conflicts of interest in relation to this presentation. Vogel FS & Burger PC 3/28/2016
 IF THIS IS NOT GLIOBLASTOMA, THEN WHAT IS IT? Murat Gokden, MD Department of Pathology/Neuropathology University of Arkansas for Medical Sciences Little Rock, AR mgokden@uams.edu I have no conflicts of
IF THIS IS NOT GLIOBLASTOMA, THEN WHAT IS IT? Murat Gokden, MD Department of Pathology/Neuropathology University of Arkansas for Medical Sciences Little Rock, AR mgokden@uams.edu I have no conflicts of
21/03/2017. Disclosure. Practice Changing Articles in Neuro Oncology for 2016/17. Gliomas. Objectives. Gliomas. No conflicts to declare
 Practice Changing Articles in Neuro Oncology for 2016/17 Disclosure No conflicts to declare Frances Cusano, BScPharm, ACPR April 21, 2017 Objectives Gliomas To describe the patient selection, methodology
Practice Changing Articles in Neuro Oncology for 2016/17 Disclosure No conflicts to declare Frances Cusano, BScPharm, ACPR April 21, 2017 Objectives Gliomas To describe the patient selection, methodology
Case Presentation: USCAP Jason T. Huse, MD, PhD Assistant Member Department of Pathology Memorial Sloan Kettering Cancer Center
 Case Presentation: USCAP 2016 Jason T. Huse, MD, PhD Assistant Member Department of Pathology Memorial Sloan Kettering Cancer Center Case History 53 year old female with a long standing history of migraines
Case Presentation: USCAP 2016 Jason T. Huse, MD, PhD Assistant Member Department of Pathology Memorial Sloan Kettering Cancer Center Case History 53 year old female with a long standing history of migraines
Morphological features and genetic alterations
 Morphological features and genetic alterations Tutor : Audrey Rousseau Caget Lise: Université d Angers Iorio Vittoria: Seconda Università degli studi di Napoli Manaila Roxana: Iuliu Hatieganu University
Morphological features and genetic alterations Tutor : Audrey Rousseau Caget Lise: Université d Angers Iorio Vittoria: Seconda Università degli studi di Napoli Manaila Roxana: Iuliu Hatieganu University
Tumors of the Nervous System
 Tumors of the Nervous System Peter Canoll MD. PhD. What I want to cover What are the most common types of brain tumors? Who gets them? How do they present? What do they look like? How do they behave? 1
Tumors of the Nervous System Peter Canoll MD. PhD. What I want to cover What are the most common types of brain tumors? Who gets them? How do they present? What do they look like? How do they behave? 1
Clinical significance of genetic analysis in glioblastoma treatment
 Clinical significance of genetic analysis in glioblastoma treatment Department of Neurosurgery, Graduate School of Medical Sciences, Kyushu University, Fukuoka, Japan Koji Yoshimoto Can we get prognostic
Clinical significance of genetic analysis in glioblastoma treatment Department of Neurosurgery, Graduate School of Medical Sciences, Kyushu University, Fukuoka, Japan Koji Yoshimoto Can we get prognostic
Examining large groups of cancer patients to identify ways of predicting which therapies cancers might respond to.
 Stratified Medicine Examining large groups of cancer patients to identify ways of predicting which therapies cancers might respond to. Looking in detail at cancer cells and their genetic make up. Permit
Stratified Medicine Examining large groups of cancer patients to identify ways of predicting which therapies cancers might respond to. Looking in detail at cancer cells and their genetic make up. Permit
MOLECULAR DIAGNOSTICS OF GLIOMAS
 MOLECULAR DIAGNOSTICS OF GLIOMAS Arie Perry, M.D. Director, Neuropathology Division DIFFUSE GLIOMAS Cell types Astrocytomas (A) Oligodendrogliomas (O) Mixed oligoastrocytoma (MOA) Three WHO grades: II,
MOLECULAR DIAGNOSTICS OF GLIOMAS Arie Perry, M.D. Director, Neuropathology Division DIFFUSE GLIOMAS Cell types Astrocytomas (A) Oligodendrogliomas (O) Mixed oligoastrocytoma (MOA) Three WHO grades: II,
Precision medicine for gliomas
 Precision medicine for YAZMIN ODIA, MD MS LEAD PHYSICIAN OF MEDICAL NEURO-ONCOLOGY DISCLOSURES Novocure: Advisory Board for Optune in No other financial conflicts of interest Glioma OVERVIEW INFILTRATIVE,
Precision medicine for YAZMIN ODIA, MD MS LEAD PHYSICIAN OF MEDICAL NEURO-ONCOLOGY DISCLOSURES Novocure: Advisory Board for Optune in No other financial conflicts of interest Glioma OVERVIEW INFILTRATIVE,
What yield in the last decade about Molecular Diagnostics in Neuro
 What yield in the last decade about Molecular Diagnostics in Neuro Oncology? Raphael Salles S.Medeiros Neuropathologist at HC FMUSP Clinical Research Project Manager at Oncology department at Hospital
What yield in the last decade about Molecular Diagnostics in Neuro Oncology? Raphael Salles S.Medeiros Neuropathologist at HC FMUSP Clinical Research Project Manager at Oncology department at Hospital
Review: Diagnostic, prognostic and predictive relevance of molecular markers in gliomas
 Review: Diagnostic, prognostic and predictive relevance of molecular markers in gliomas Sebastian Brandner MD FRCPath 1 and Andreas von Deimling 2 MD 1) Division of Neuropathology, The National Hospital
Review: Diagnostic, prognostic and predictive relevance of molecular markers in gliomas Sebastian Brandner MD FRCPath 1 and Andreas von Deimling 2 MD 1) Division of Neuropathology, The National Hospital
Pathologic Analysis of CNS Surgical Specimens
 2015 Kenneth M. Earle Memorial Neuropathology Review Pathologic Analysis of CNS Surgical Specimens Peter C. Burger, MD Interdisciplinary Quality Control Familiarity with entities Use of diagnostic algorithm
2015 Kenneth M. Earle Memorial Neuropathology Review Pathologic Analysis of CNS Surgical Specimens Peter C. Burger, MD Interdisciplinary Quality Control Familiarity with entities Use of diagnostic algorithm
Tumors of the Central Nervous System
 Tumors of the Central Nervous System 1 Financial Disclosures I have NO SIGNIFICANT FINANCIAL, GENERAL, OR OBLIGATION INTERESTS TO REPORT Introduction General: Brain tumors are lesions that have mass effect
Tumors of the Central Nervous System 1 Financial Disclosures I have NO SIGNIFICANT FINANCIAL, GENERAL, OR OBLIGATION INTERESTS TO REPORT Introduction General: Brain tumors are lesions that have mass effect
WHO 2016 CNS Tumor Classification Update. DISCLOSURES (Arie Perry, MD) PATTERN RECOGNITION. Arie Perry, M.D. Director, Neuropathology
 WHO 2016 CNS Tumor Classification Update Arie Perry, M.D. Director, Neuropathology DISCLOSURES (Arie Perry, MD) I have no financial relationships to disclose. - and - I will not discuss off label use or
WHO 2016 CNS Tumor Classification Update Arie Perry, M.D. Director, Neuropathology DISCLOSURES (Arie Perry, MD) I have no financial relationships to disclose. - and - I will not discuss off label use or
R1601 Essential Immunohistochemical and Molecular Markers for General CNS Glial Tumors
 October 22, 2018 12:00-1:00 PM Background The World Health Organization Classification of tumors of the Central Nervous System has recently been revised. There is now greater emphasis on molecular phenotype
October 22, 2018 12:00-1:00 PM Background The World Health Organization Classification of tumors of the Central Nervous System has recently been revised. There is now greater emphasis on molecular phenotype
SUPPLEMENTARY INFORMATION
 VOLUME: 1 ARTICLE NUMBER: 0027 In the format provided by the authors and unedited. Rapid intraoperative histology of unprocessed surgical specimens via fibre-laser-based stimulated Raman scattering microscopy
VOLUME: 1 ARTICLE NUMBER: 0027 In the format provided by the authors and unedited. Rapid intraoperative histology of unprocessed surgical specimens via fibre-laser-based stimulated Raman scattering microscopy
Peter Canoll MD. PhD.
 Tumors of the Nervous System Peter Canoll MD. PhD. What I want to cover What are the most common types of brain tumors? Who gets them? How do they ypresent? What do they look like? How do they behave?
Tumors of the Nervous System Peter Canoll MD. PhD. What I want to cover What are the most common types of brain tumors? Who gets them? How do they ypresent? What do they look like? How do they behave?
Childhood Brain and Spinal Cord Tumors Treatment Overview (PDQ )
 1 di 8 04/03/2017 07.31 NCBI Bookshelf. A service of the National Library of Medicine, National Institutes of Health. PDQ Cancer Information Summaries [Internet]. Bethesda (MD): National Cancer Institute
1 di 8 04/03/2017 07.31 NCBI Bookshelf. A service of the National Library of Medicine, National Institutes of Health. PDQ Cancer Information Summaries [Internet]. Bethesda (MD): National Cancer Institute
Corporate Medical Policy
 Corporate Medical Policy Analysis of MGMT Promoter Methylation in Malignant Gliomas File Name: Origination: Last CAP Review: Next CAP Review: Last Review: analysis_of_mgmt_promoter_methylation_in_malignant_gliomas
Corporate Medical Policy Analysis of MGMT Promoter Methylation in Malignant Gliomas File Name: Origination: Last CAP Review: Next CAP Review: Last Review: analysis_of_mgmt_promoter_methylation_in_malignant_gliomas
Chapter 1 Introduction
 Chapter 1 Introduction Men think epilepsy divine, merely because they do not understand it. But if they called everything divine which they do not understand, why, there would be no end to divine things.
Chapter 1 Introduction Men think epilepsy divine, merely because they do not understand it. But if they called everything divine which they do not understand, why, there would be no end to divine things.
Radioterapia no Tratamento dos Gliomas de Baixo Grau
 Radioterapia no Tratamento dos Gliomas de Baixo Grau Dr. Luis Souhami University Montreal - Canada Low Grade Gliomas Relatively rare Heterogeneous, slow growing tumors WHO Classification Grade I Pilocytic
Radioterapia no Tratamento dos Gliomas de Baixo Grau Dr. Luis Souhami University Montreal - Canada Low Grade Gliomas Relatively rare Heterogeneous, slow growing tumors WHO Classification Grade I Pilocytic
The 2016 WHO classification of central nervous system tumors: what neurologists need to know
 REVIEW C URRENT OPINION The 2016 WHO classification of central nervous system tumors: what neurologists need to know John C. DeWitt a,, Andreas Mock a,b,c,, and David N. Louis a Purpose of review The 2016
REVIEW C URRENT OPINION The 2016 WHO classification of central nervous system tumors: what neurologists need to know John C. DeWitt a,, Andreas Mock a,b,c,, and David N. Louis a Purpose of review The 2016
Applications of molecular neuro-oncology - a review of diffuse glioma integrated diagnosis and emerging molecular entities
 Wood et al. Diagnostic Pathology (2019) 14:29 https://doi.org/10.1186/s13000-019-0802-8 REVIEW Applications of molecular neuro-oncology - a review of diffuse glioma integrated diagnosis and emerging molecular
Wood et al. Diagnostic Pathology (2019) 14:29 https://doi.org/10.1186/s13000-019-0802-8 REVIEW Applications of molecular neuro-oncology - a review of diffuse glioma integrated diagnosis and emerging molecular
Supplemental Information. Molecular, Pathological, Radiological, and Immune. Profiling of Non-brainstem Pediatric High-Grade
 Cancer Cell, Volume 33 Supplemental Information Molecular, Pathological, Radiological, and Immune Profiling of Non-brainstem Pediatric High-Grade Glioma from the HERBY Phase II Randomized Trial Alan Mackay,
Cancer Cell, Volume 33 Supplemental Information Molecular, Pathological, Radiological, and Immune Profiling of Non-brainstem Pediatric High-Grade Glioma from the HERBY Phase II Randomized Trial Alan Mackay,
5 th International Summer School for Neuropathology and Epilepsy Surgery INES October 6-9, 2016 University Hospital, Erlangen
 5 th International Summer School for Neuropathology and Epilepsy Surgery INES 2016 October 6-9, 2016 University Hospital, Erlangen Course Director: For more information, please contact: Ingmar Blümcke
5 th International Summer School for Neuropathology and Epilepsy Surgery INES 2016 October 6-9, 2016 University Hospital, Erlangen Course Director: For more information, please contact: Ingmar Blümcke
Phase II Pediatric Study With Dabrafenib in Combination With Trametinib in Patients With HGG and LGG
 Find Studies About Studies Submit Studies Resources About Site Phase II Pediatric Study With Dabrafenib in Combination With Trametinib in Patients With HGG and LGG The safety and scientific validity of
Find Studies About Studies Submit Studies Resources About Site Phase II Pediatric Study With Dabrafenib in Combination With Trametinib in Patients With HGG and LGG The safety and scientific validity of
The recently updated World Health Organization (WHO)
 REVIEW ARTICLE The Expanding Family of Glioneuronal Tumors Daniela S. Allende, MD and Richard A. Prayson, MD Abstract: Three new entities have been recently added to the group of glioneuronal tumors in
REVIEW ARTICLE The Expanding Family of Glioneuronal Tumors Daniela S. Allende, MD and Richard A. Prayson, MD Abstract: Three new entities have been recently added to the group of glioneuronal tumors in
WHO 2016 CNS TUMOR CLASSIFICATION UPDATE. Arie Perry, M.D. Director, Neuropathology
 WHO 2016 CNS TUMOR CLASSIFICATION UPDATE Arie Perry, M.D. Director, Neuropathology DISCLOSURES (Arie Perry, MD) I have no financial relationships to disclose. - and - I will not discuss off label use or
WHO 2016 CNS TUMOR CLASSIFICATION UPDATE Arie Perry, M.D. Director, Neuropathology DISCLOSURES (Arie Perry, MD) I have no financial relationships to disclose. - and - I will not discuss off label use or
H3F3A K27M Mutation in Pediatric CNS Tumors. A Marker for Diffuse High-Grade Astrocytomas
 Anatomic Pathology / H3.3 Mutations in Pediatric Diffuse High-Grade Astrocytomas H3F3A K27M Mutation in Pediatric CNS Tumors A Marker for Diffuse High-Grade Astrocytomas Gerrit H. Gielen, MD, 1 Marco Gessi,
Anatomic Pathology / H3.3 Mutations in Pediatric Diffuse High-Grade Astrocytomas H3F3A K27M Mutation in Pediatric CNS Tumors A Marker for Diffuse High-Grade Astrocytomas Gerrit H. Gielen, MD, 1 Marco Gessi,
Histopathological Study and Categorisation of Brain Tumors
 Histopathological Study and Categorisation of Brain Tumors Ruchira Wadhwa 1*, Purvi Patel 2, Hansa Goswami 3 1 Third Year Resident, 2 Assistant Professor, 3 Professor and Head, Department of Pathology,
Histopathological Study and Categorisation of Brain Tumors Ruchira Wadhwa 1*, Purvi Patel 2, Hansa Goswami 3 1 Third Year Resident, 2 Assistant Professor, 3 Professor and Head, Department of Pathology,
Pleomorphic Xanthoastrocytoma
 Pleomorphic Xanthoastrocytoma Christine E. Fuller Keywords Pleomorphic xanthoastrocytoma; Pleomorphic xanthoastrocytoma with anaplastic features 2.1 OVERVIEW Pleomorphic xanthoastrocytoma (PXA) is an uncommon
Pleomorphic Xanthoastrocytoma Christine E. Fuller Keywords Pleomorphic xanthoastrocytoma; Pleomorphic xanthoastrocytoma with anaplastic features 2.1 OVERVIEW Pleomorphic xanthoastrocytoma (PXA) is an uncommon
We are IntechOpen, the first native scientific publisher of Open Access books. International authors and editors. Our authors are among the TOP 1%
 We are IntechOpen, the first native scientific publisher of Open Access books 3,350 108,000 1.7 M Open access books available International authors and editors Downloads Our authors are among the 151 Countries
We are IntechOpen, the first native scientific publisher of Open Access books 3,350 108,000 1.7 M Open access books available International authors and editors Downloads Our authors are among the 151 Countries
Astroblastoma: Radiologic-Pathologic Correlation and Distinction from Ependymoma
 AJNR Am J Neuroradiol 23:243 247, February 2002 Case Report Astroblastoma: Radiologic-Pathologic Correlation and Distinction from Ependymoma John D. Port, Daniel J. Brat, Peter C. Burger, and Martin G.
AJNR Am J Neuroradiol 23:243 247, February 2002 Case Report Astroblastoma: Radiologic-Pathologic Correlation and Distinction from Ependymoma John D. Port, Daniel J. Brat, Peter C. Burger, and Martin G.
Integrating molecular markers into the World Health Organization classification of CNS tumors: a survey of the neuro-oncology community
 Integrating molecular markers into the World Health Organization classification of CNS tumors: a survey of the neuro-oncology community The Harvard community has made this article openly available. Please
Integrating molecular markers into the World Health Organization classification of CNS tumors: a survey of the neuro-oncology community The Harvard community has made this article openly available. Please
Site Specific Coding Rules MALIGNANT CENTRAL NERVOUS SYSTEM TUMORS
 Multiple Primary and Histology Site Specific Coding Rules MALIGNANT CENTRAL NERVOUS SYSTEM TUMORS 1 Prerequisites 2 Completion of Multiple Primary and Histology General Coding Rules 3 There are many ways
Multiple Primary and Histology Site Specific Coding Rules MALIGNANT CENTRAL NERVOUS SYSTEM TUMORS 1 Prerequisites 2 Completion of Multiple Primary and Histology General Coding Rules 3 There are many ways
Announcing cimpact-now: the Consortium to Inform Molecular and Practical Approaches to CNS Tumor Taxonomy
 Zurich Open Repository and Archive University of Zurich Main Library Strickhofstrasse 39 CH-8057 Zurich www.zora.uzh.ch Year: 2017 Announcing cimpact-now: the Consortium to Inform Molecular and Practical
Zurich Open Repository and Archive University of Zurich Main Library Strickhofstrasse 39 CH-8057 Zurich www.zora.uzh.ch Year: 2017 Announcing cimpact-now: the Consortium to Inform Molecular and Practical
Imaging for suspected glioma
 Imaging for suspected glioma 1.1.1 Offer standard structural MRI (defined as T2 weighted, FLAIR, DWI series and T1 pre- and post-contrast volume) as the initial diagnostic test for suspected glioma, unless
Imaging for suspected glioma 1.1.1 Offer standard structural MRI (defined as T2 weighted, FLAIR, DWI series and T1 pre- and post-contrast volume) as the initial diagnostic test for suspected glioma, unless
Case Report Complex Form Variant of Dysembryoplastic Neuroepithelial Tumor of the Cerebellum
 Case Reports in Pathology Volume 2012, Article ID 718651, 4 pages doi:10.1155/2012/718651 Case Report Complex Form Variant of Dysembryoplastic Neuroepithelial Tumor of the Cerebellum Jesús Vaquero, 1,
Case Reports in Pathology Volume 2012, Article ID 718651, 4 pages doi:10.1155/2012/718651 Case Report Complex Form Variant of Dysembryoplastic Neuroepithelial Tumor of the Cerebellum Jesús Vaquero, 1,
Malignant Transformation of a Dysembryoplastic Neuroepithelial Tumor (DNET) Characterized by Genome-Wide Methylation Analysis
 J Neuropathol Exp Neurol Vol. 75, No. 4, April 2016, pp. 358 365 doi: 10.1093/jnen/nlw007 ORIGINAL ARTICLE Malignant Transformation of a Dysembryoplastic Neuroepithelial Tumor (DNET) Characterized by Genome-Wide
J Neuropathol Exp Neurol Vol. 75, No. 4, April 2016, pp. 358 365 doi: 10.1093/jnen/nlw007 ORIGINAL ARTICLE Malignant Transformation of a Dysembryoplastic Neuroepithelial Tumor (DNET) Characterized by Genome-Wide
BRAF V600E Mutation Is Associated with mtor Signaling Activation in Glioneuronal Tumors
 Brain Pathology ISSN 115-635 RESEARCH ARTICLE BRAF V6E Mutation Is Associated with mtor Signaling Activation in Glioneuronal Tumors Avanita S. Prabowo 1 ; Anand M. Iyer 1 ; Tim J. Veersema 3,4 ; Jasper
Brain Pathology ISSN 115-635 RESEARCH ARTICLE BRAF V6E Mutation Is Associated with mtor Signaling Activation in Glioneuronal Tumors Avanita S. Prabowo 1 ; Anand M. Iyer 1 ; Tim J. Veersema 3,4 ; Jasper
Understanding general brain tumor pathology, Part I: The basics. Craig Horbinski, M.D., Ph.D. Department of Pathology University of Kentucky
 Understanding general brain tumor pathology, Part I: The basics Craig Horbinski, M.D., Ph.D. Department of Pathology University of Kentucky plan of attack what IS a pathologist, anyway? what s so special
Understanding general brain tumor pathology, Part I: The basics Craig Horbinski, M.D., Ph.D. Department of Pathology University of Kentucky plan of attack what IS a pathologist, anyway? what s so special
Diagnostic application of SNParrays to brain cancers
 Diagnostic application of SNParrays to brain cancers Adriana Olar 4/17/2018 No disclosures 55 yo M, focal motor seizure T2 T1-post C DIAGNOSIS BRAIN, LEFT FRONTAL LOBE, BIOPSY: - DIFFUSE GLIOMA, OLIGODENDROGLIAL
Diagnostic application of SNParrays to brain cancers Adriana Olar 4/17/2018 No disclosures 55 yo M, focal motor seizure T2 T1-post C DIAGNOSIS BRAIN, LEFT FRONTAL LOBE, BIOPSY: - DIFFUSE GLIOMA, OLIGODENDROGLIAL
CHINESE MEDICAL ASSOCIATION
 Zhu et al. Chinese Neurosurgical Journal (2017) 3:22 DOI 10.1186/s41016-017-0087-2 CHINESE NEUROSURGICAL SOCIETY CASE REPORT CHINESE MEDICAL ASSOCIATION Anaplastic pleomorphic xanthoastrocytoma with disseminated
Zhu et al. Chinese Neurosurgical Journal (2017) 3:22 DOI 10.1186/s41016-017-0087-2 CHINESE NEUROSURGICAL SOCIETY CASE REPORT CHINESE MEDICAL ASSOCIATION Anaplastic pleomorphic xanthoastrocytoma with disseminated
Disclaimers. Molecular pathology of brain tumors. Some aspects only. Some details are inevitably personal opinions
 Molecular pathology of brain tumors Disclaimers Some aspects only H.K. Ng The Chinese University of Hong Kong Some details are inevitably personal opinions Free ppt : http://www.acp.cuhk.edu.hk/hkng Why
Molecular pathology of brain tumors Disclaimers Some aspects only H.K. Ng The Chinese University of Hong Kong Some details are inevitably personal opinions Free ppt : http://www.acp.cuhk.edu.hk/hkng Why
A clinical perspective on neuropathology and molecular genetics in brain tumors
 A clinical perspective on neuropathology and molecular genetics in brain tumors M.J. van den Bent Erasmus MC Cancer Institute Rotterdam, the Netherlands Disclosures Member speakersbureau: MSD Consultancy:
A clinical perspective on neuropathology and molecular genetics in brain tumors M.J. van den Bent Erasmus MC Cancer Institute Rotterdam, the Netherlands Disclosures Member speakersbureau: MSD Consultancy:
Advances in Brain Tumor Research: Leveraging BIG data for BIG discoveries
 Advances in Brain Tumor Research: Leveraging BIG data for BIG discoveries Jill Barnholtz-Sloan, PhD Associate Professor & Associate Director for Bioinformatics and Translational Informatics jsb42@case.edu
Advances in Brain Tumor Research: Leveraging BIG data for BIG discoveries Jill Barnholtz-Sloan, PhD Associate Professor & Associate Director for Bioinformatics and Translational Informatics jsb42@case.edu
2018 Diagnostic Slide Session Case #8
 2018 Diagnostic Slide Session Case #8 Angela N. Viaene, MacLean P. Nasrallah, and Zissimos Mourelatos Hospital of the University of Pennsylvania AANP June 9, 2018 Disclosures: none Clinical History Healthy,
2018 Diagnostic Slide Session Case #8 Angela N. Viaene, MacLean P. Nasrallah, and Zissimos Mourelatos Hospital of the University of Pennsylvania AANP June 9, 2018 Disclosures: none Clinical History Healthy,
Pediatric Brain Tumors: Updates in Treatment and Care
 Pediatric Brain Tumors: Updates in Treatment and Care Writer Classroom Rishi R. Lulla, MD MS Objectives Introduce the common pediatric brain tumors Discuss current treatment strategies for pediatric brain
Pediatric Brain Tumors: Updates in Treatment and Care Writer Classroom Rishi R. Lulla, MD MS Objectives Introduce the common pediatric brain tumors Discuss current treatment strategies for pediatric brain
Epidemiology and Semiology of Tumor-based Epilepsy December 2, 2012
 Epidemiology and Semiology of Tumor-based Epilepsy December 2, 2012 Charles J. Vecht, MD, PhD Medical Center The Hague SEIN Epilepsy Foundation, The Netherlands CHU Pitié-Salpêtrière, Paris, France American
Epidemiology and Semiology of Tumor-based Epilepsy December 2, 2012 Charles J. Vecht, MD, PhD Medical Center The Hague SEIN Epilepsy Foundation, The Netherlands CHU Pitié-Salpêtrière, Paris, France American
The new WHO 2016 classification of brain tumors what neurosurgeons need to know
 DOI 10.1007/s00701-016-3062-3 REVIEW ARTICLE - BRAIN TUMORS The new WHO 2016 classification of brain tumors what neurosurgeons need to know Rouzbeh Banan 1 & Christian Hartmann 1 Received: 8 July 2016
DOI 10.1007/s00701-016-3062-3 REVIEW ARTICLE - BRAIN TUMORS The new WHO 2016 classification of brain tumors what neurosurgeons need to know Rouzbeh Banan 1 & Christian Hartmann 1 Received: 8 July 2016
SPECIAL SLIDE SEMINAR CASE 3
 SPECIAL SLIDE SEMINAR CASE 3 Tihana Džombeta, MD Leo Pažanin, MD, PhD Department of Pathology, School of Medicine, University of Zagreb Department of Pathology, Clinical Hospital Centre Sestre milosrdnice
SPECIAL SLIDE SEMINAR CASE 3 Tihana Džombeta, MD Leo Pažanin, MD, PhD Department of Pathology, School of Medicine, University of Zagreb Department of Pathology, Clinical Hospital Centre Sestre milosrdnice
Gangliogliomas: A Report of Five Cases
 Case Report Gangliogliomas: A Report of Five Cases Nair V, Suri VS, Tatke M, Saran RK, Malhotra V, Singh D* Departments of Pathology and *Neurosurgery, G. B. Pant Hospital, New Delhi, India. Correspondence
Case Report Gangliogliomas: A Report of Five Cases Nair V, Suri VS, Tatke M, Saran RK, Malhotra V, Singh D* Departments of Pathology and *Neurosurgery, G. B. Pant Hospital, New Delhi, India. Correspondence
MolDX: Chromosome 1p/19q deletion analysis
 MolDX: Chromosome 1p/19q deletion analysis CGS Administrators, LLC Jump to Section... Please Note: This is a Proposed LCD. Proposed LCDs are works in progress and not necessarily a reflection of the current
MolDX: Chromosome 1p/19q deletion analysis CGS Administrators, LLC Jump to Section... Please Note: This is a Proposed LCD. Proposed LCDs are works in progress and not necessarily a reflection of the current
성균관대학교삼성창원병원신경외과학교실신경종양학 김영준. KNS-MT-03 (April 15, 2015)
 성균관대학교삼성창원병원신경외과학교실신경종양학 김영준 INTRODUCTIONS Low grade gliomas (LGG) - heterogeneous group of tumors with astrocytic, oligodendroglial, ependymal, or mixed cellular histology - In adults diffuse, infiltrating
성균관대학교삼성창원병원신경외과학교실신경종양학 김영준 INTRODUCTIONS Low grade gliomas (LGG) - heterogeneous group of tumors with astrocytic, oligodendroglial, ependymal, or mixed cellular histology - In adults diffuse, infiltrating
Genomic analysis of childhood High grade glial (HGG) brain tumors
 Genomic analysis of childhood High grade glial (HGG) brain tumors Linda D Cooley Children s Mercy, Kansas City The Children s Mercy Hospital, 2017 Genomic analysis of childhood High grade glial (HGG) brain
Genomic analysis of childhood High grade glial (HGG) brain tumors Linda D Cooley Children s Mercy, Kansas City The Children s Mercy Hospital, 2017 Genomic analysis of childhood High grade glial (HGG) brain
Brain Tumors. Medulloblastoma. Pilocytic astrocytoma: Ahmed Koriesh, MD. Pathological finding
 NeuroPathology Page 8 Brain Tumors Pathological finding Pseudorosette Rosenthal fibers Rosettes Wet Keratin Psammoma bodies Fried egg Tumor Ependymoma, SEGA Pilocytic astrocytoma Medulloblastoma Craniopharyngioma
NeuroPathology Page 8 Brain Tumors Pathological finding Pseudorosette Rosenthal fibers Rosettes Wet Keratin Psammoma bodies Fried egg Tumor Ependymoma, SEGA Pilocytic astrocytoma Medulloblastoma Craniopharyngioma
Challenges and opportunities for understanding epilepsy and cognitive and behavioral comorbidities. Eleonora Aronica
 Biobanks and Databases -Basis for Translational Research Challenges and opportunities for understanding epilepsy and cognitive and behavioral comorbidities Eleonora Aronica Department of (Neuro)Pathology,
Biobanks and Databases -Basis for Translational Research Challenges and opportunities for understanding epilepsy and cognitive and behavioral comorbidities Eleonora Aronica Department of (Neuro)Pathology,
Systemic Treatment. Third International Neuro-Oncology Course. 23 May 2014
 Low-Grade Astrocytoma of the CNS: Systemic Treatment Third International Neuro-Oncology Course São Paulo, Brazil 23 May 2014 John de Groot, MD Associate Professor, Neuro-Oncology UT MD Anderson Cancer
Low-Grade Astrocytoma of the CNS: Systemic Treatment Third International Neuro-Oncology Course São Paulo, Brazil 23 May 2014 John de Groot, MD Associate Professor, Neuro-Oncology UT MD Anderson Cancer
PROPOSED/DRAFT Local Coverage Determination (LCD): MolDX: Chromosome 1p/19q deletion analysis (DL36483)
 moldx: Chromosome 1p/19q deletion analysis (DL36483) Page 1 of 8 PROPOSED/DRAFT Local Coverage Determination (LCD): MolDX: Chromosome 1p/19q deletion analysis (DL36483) Close Section Navigation
moldx: Chromosome 1p/19q deletion analysis (DL36483) Page 1 of 8 PROPOSED/DRAFT Local Coverage Determination (LCD): MolDX: Chromosome 1p/19q deletion analysis (DL36483) Close Section Navigation
MALIGNANT GLIOMAS: TREATMENT AND CHALLENGES
 MALIGNANT GLIOMAS: TREATMENT AND CHALLENGES DISCLOSURE No conflicts of interest to disclose Patricia Bruns APRN, CNS Givens Brain Tumor Center Abbott Northwestern Hospital October 12, 2018 OBJECTIVES THEN
MALIGNANT GLIOMAS: TREATMENT AND CHALLENGES DISCLOSURE No conflicts of interest to disclose Patricia Bruns APRN, CNS Givens Brain Tumor Center Abbott Northwestern Hospital October 12, 2018 OBJECTIVES THEN
Contemporary Management of Glioblastoma
 Contemporary Management of Glioblastoma Incidence Rates of Primary Brain Tumors Central Brain Tumor Registry of the United States, 1992-1997 100 Number of Cases per 100,000 Population 10 1 0.1 x I x I
Contemporary Management of Glioblastoma Incidence Rates of Primary Brain Tumors Central Brain Tumor Registry of the United States, 1992-1997 100 Number of Cases per 100,000 Population 10 1 0.1 x I x I
Chemotherapy in malignant brain tumors
 Chemotherapy in malignant brain tumors Frank Zimmermann Institut für Radioonkologie Universitätsspital Basel Petersgraben 4 CH 4031 Basel zimmermannf@uhbs.ch Tumor types Neuro-epithelial tumors - Glioblastoma
Chemotherapy in malignant brain tumors Frank Zimmermann Institut für Radioonkologie Universitätsspital Basel Petersgraben 4 CH 4031 Basel zimmermannf@uhbs.ch Tumor types Neuro-epithelial tumors - Glioblastoma
Epilepsy and Brain Tumor. Apasri Lusawat M.D. Pediatric Neurology Prasat Neurological Institute
 Epilepsy and Brain Tumor Apasri Lusawat M.D. Pediatric Neurology Prasat Neurological Institute Outline Overview Pathogenesis : Tumor-> epilepsy Etiology of Epilepsy, age EPILEPSY AND BRAIN TUMORS CNS neoplasms
Epilepsy and Brain Tumor Apasri Lusawat M.D. Pediatric Neurology Prasat Neurological Institute Outline Overview Pathogenesis : Tumor-> epilepsy Etiology of Epilepsy, age EPILEPSY AND BRAIN TUMORS CNS neoplasms
AMERICAN ASSOCIATION OF NEUROPATHOLOGISTS COMPANION SOCIETY MEETING at the 106 th ANNUAL MEETING OF THE USCAP San Antonio, March 4, 2017
 AMERICAN ASSOCIATION OF NEUROPATHOLOGISTS COMPANION SOCIETY MEETING at the 106 th ANNUAL MEETING OF THE USCAP San Antonio, March 4, 2017 SYLLABUS Papillary Tumor of the Pineal Region and the Differential
AMERICAN ASSOCIATION OF NEUROPATHOLOGISTS COMPANION SOCIETY MEETING at the 106 th ANNUAL MEETING OF THE USCAP San Antonio, March 4, 2017 SYLLABUS Papillary Tumor of the Pineal Region and the Differential
SYSTEMIC MANAGEMENT OF PEDIATRIC PRIMARY BRAIN TUMORS
 SYSTEMIC MANAGEMENT OF PEDIATRIC PRIMARY BRAIN TUMORS María E. Echevarría, MD Assistant Professor University of Puerto Rico Medical Sciences Campus DISCLOSURES No disclosures INTRODUCTION Pediatric CNS
SYSTEMIC MANAGEMENT OF PEDIATRIC PRIMARY BRAIN TUMORS María E. Echevarría, MD Assistant Professor University of Puerto Rico Medical Sciences Campus DISCLOSURES No disclosures INTRODUCTION Pediatric CNS
The genetic landscape of ganglioglioma
 Pekmezci et al. Acta Neuropathologica Communications (2018) 6:47 https://doi.org/10.1186/s40478-018-0551-z RESEARCH The genetic landscape of ganglioglioma Open Access Melike Pekmezci 1, Javier E. Villanueva-Meyer
Pekmezci et al. Acta Neuropathologica Communications (2018) 6:47 https://doi.org/10.1186/s40478-018-0551-z RESEARCH The genetic landscape of ganglioglioma Open Access Melike Pekmezci 1, Javier E. Villanueva-Meyer
Patients Treated with Leksell Gamma Knife
 Patients Treated with Leksell Gamma Knife 1968-2016 TREATMENTS REPORTED 2016 BY REGION AND INDICATION INDICATION Asia excl. Europe Latin Middle East & Africa North Grand Total Benign Tumors 12283 9778
Patients Treated with Leksell Gamma Knife 1968-2016 TREATMENTS REPORTED 2016 BY REGION AND INDICATION INDICATION Asia excl. Europe Latin Middle East & Africa North Grand Total Benign Tumors 12283 9778
Case Report Atypical Presentation of Atypical Teratoid Rhabdoid Tumor in a Child
 Case Reports in Oncological Medicine Volume 2013, Article ID 815923, 4 pages http://dx.doi.org/10.1155/2013/815923 Case Report Atypical Presentation of Atypical Teratoid Rhabdoid Tumor in a Child Y. T.
Case Reports in Oncological Medicine Volume 2013, Article ID 815923, 4 pages http://dx.doi.org/10.1155/2013/815923 Case Report Atypical Presentation of Atypical Teratoid Rhabdoid Tumor in a Child Y. T.
LOW GRADE ASTROCYTOMAS
 LOW GRADE ASTROCYTOMAS This article was provided to us by David Schiff, MD, Associate Professor of Neurology, Neurosurgery, and Medicine at University of Virginia, Charlottesville. We appreciate his generous
LOW GRADE ASTROCYTOMAS This article was provided to us by David Schiff, MD, Associate Professor of Neurology, Neurosurgery, and Medicine at University of Virginia, Charlottesville. We appreciate his generous
Anna Maria Buccoliero Department of Biomedicine, Careggi Hospital Florence
 PEDIATRIC RHABDOID MENINGIOMA Anna Maria Buccoliero Department of Biomedicine, Careggi Hospital Florence CLINICAL HISTORY A 3-year-old boy, with a recent history of seizures, was admitted to the Neurosurgery
PEDIATRIC RHABDOID MENINGIOMA Anna Maria Buccoliero Department of Biomedicine, Careggi Hospital Florence CLINICAL HISTORY A 3-year-old boy, with a recent history of seizures, was admitted to the Neurosurgery
CASE OF THE WEEK PROFESSOR YASSER METWALLY
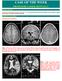 CASE OF THE WEEK PROFESSOR YASSER METWALLY CLINICAL PICTURE CLINICAL PICTURE: CLINICAL PICTURE: A 6 years old male patient presented clinically with intractable complex partial seizure. The child is mentally
CASE OF THE WEEK PROFESSOR YASSER METWALLY CLINICAL PICTURE CLINICAL PICTURE: CLINICAL PICTURE: A 6 years old male patient presented clinically with intractable complex partial seizure. The child is mentally
Dysembryoplastic Neuroepithelial Tumor (DNT): Morphological and Immunohistochemical Features
 JKAU: Med. Sci., Vol. 14 No. 3, pp: 45-54 (2007 A.D. / 1428 A.H.) Dysembryoplastic Neuroepithelial Tumor (DNT): Morphological and Immunohistochemical Features Jaudah A. Al-Maghrabi 1,2 (MD, FRCPC) Department
JKAU: Med. Sci., Vol. 14 No. 3, pp: 45-54 (2007 A.D. / 1428 A.H.) Dysembryoplastic Neuroepithelial Tumor (DNT): Morphological and Immunohistochemical Features Jaudah A. Al-Maghrabi 1,2 (MD, FRCPC) Department
Molecular diagnostics of gliomas: state of the art
 Acta Neuropathol (2010) 120:567 584 DOI 10.1007/s00401-010-0736-4 REVIEW Molecular diagnostics of gliomas: state of the art Markus J. Riemenschneider Judith W. M. Jeuken Pieter Wesseling Guido Reifenberger
Acta Neuropathol (2010) 120:567 584 DOI 10.1007/s00401-010-0736-4 REVIEW Molecular diagnostics of gliomas: state of the art Markus J. Riemenschneider Judith W. M. Jeuken Pieter Wesseling Guido Reifenberger
5-hydroxymethylcytosine loss is associated with poor prognosis for
 5-hydroxymethylcytosine loss is associated with poor prognosis for patients with WHO grade II diffuse astrocytomas Feng Zhang 1,*, Yifan Liu 2, Zhiwen Zhang 1, Jie Li 1, Yi Wan 3, Liying Zhang 1, Yangmei
5-hydroxymethylcytosine loss is associated with poor prognosis for patients with WHO grade II diffuse astrocytomas Feng Zhang 1,*, Yifan Liu 2, Zhiwen Zhang 1, Jie Li 1, Yi Wan 3, Liying Zhang 1, Yangmei
In 2005, Wang and coworkers 1 described a report of 8
 Angiocentric Glioma A Clinicopathologic Review of 5 Tumors With Identification of Associated Cortical Dysplasia Trent Marburger, MD; Richard Prayson, MD N Context. Angiocentric glioma is a rare, epilepsy-associated,
Angiocentric Glioma A Clinicopathologic Review of 5 Tumors With Identification of Associated Cortical Dysplasia Trent Marburger, MD; Richard Prayson, MD N Context. Angiocentric glioma is a rare, epilepsy-associated,
Enterprise Interest None
 Enterprise Interest None Heterogeneous chromosomal profiles in a unique series of DIPG in children and young adults European Congress of Pathology Amsterdam, 6 th September 2017 Charlotte Dufour, Romain
Enterprise Interest None Heterogeneous chromosomal profiles in a unique series of DIPG in children and young adults European Congress of Pathology Amsterdam, 6 th September 2017 Charlotte Dufour, Romain
Pathological assessment of epilepsy surgery brain tissue
 pathologica 2016;108:80-86 Original article Pathological assessment of epilepsy surgery brain tissue G. Marucci 1, M. Giulioni 2 1 Section of Pathology, Bellaria Hospital, Azienda USL IRCCS-ISNB Institute
pathologica 2016;108:80-86 Original article Pathological assessment of epilepsy surgery brain tissue G. Marucci 1, M. Giulioni 2 1 Section of Pathology, Bellaria Hospital, Azienda USL IRCCS-ISNB Institute
Corporate Medical Policy
 Corporate Medical Policy BRAF Gene Variant Testing to Select Melanoma or Glioma Patients File Name: Origination: Last CAP Review: Next CAP Review: Last Review: braf_gene_variant_testing_to_select_melanoma_or_glioma_patients_for_targeted_
Corporate Medical Policy BRAF Gene Variant Testing to Select Melanoma or Glioma Patients File Name: Origination: Last CAP Review: Next CAP Review: Last Review: braf_gene_variant_testing_to_select_melanoma_or_glioma_patients_for_targeted_
How genetic & biochemical alterations in brain tumors contribute to epileptogenesis
 How genetic & biochemical alterations in brain tumors contribute to epileptogenesis December 2 nd, 2012 Joon H. Uhm, MD FRCPC Departments of Neurology & Oncology Mayo Clinic, Rochester, MN American Epilepsy
How genetic & biochemical alterations in brain tumors contribute to epileptogenesis December 2 nd, 2012 Joon H. Uhm, MD FRCPC Departments of Neurology & Oncology Mayo Clinic, Rochester, MN American Epilepsy
1st North American Workshop on Neuropathology and Epilepsy Surgery
 April 27-30, 2017 Tudor Arms DoubleTree Hotel Cleveland, OH Endorsed by April 27-30, 2017 Tudor Arms DoubleTree Hotel Cleveland, OH Workshop Description Reliable neuropathological work-up of epilepsy-associated
April 27-30, 2017 Tudor Arms DoubleTree Hotel Cleveland, OH Endorsed by April 27-30, 2017 Tudor Arms DoubleTree Hotel Cleveland, OH Workshop Description Reliable neuropathological work-up of epilepsy-associated
Title. CitationBrain Tumor Pathology, 34(4): Issue Date Doc URL. Rights. Type. Additional There Information.
 Title Diencephalic pediatric low-grade glioma harboring th sequential biopsy specimens Ishi, Yukitomo; Hatanaka, Kanako C.; Yamaguchi, Shig Author(s) Terasaka, Shunsuke; Houkin, Kiyohiro CitationBrain
Title Diencephalic pediatric low-grade glioma harboring th sequential biopsy specimens Ishi, Yukitomo; Hatanaka, Kanako C.; Yamaguchi, Shig Author(s) Terasaka, Shunsuke; Houkin, Kiyohiro CitationBrain
Carmustine implants and Temozolomide for the treatment of newly diagnosed high grade glioma
 National Institute for Health and Clinical Excellence Health Technology Appraisal Carmustine implants and Temozolomide for the treatment of newly diagnosed high grade glioma Personal statement Conventional
National Institute for Health and Clinical Excellence Health Technology Appraisal Carmustine implants and Temozolomide for the treatment of newly diagnosed high grade glioma Personal statement Conventional
Tumours of the Central Nervous System (CNS) Molecular Information Reporting Guide
 Sponsored by Tumours of the Central Nervous System (CNS) Molecular Information Reporting Guide Family/Last name Given name(s) Date of birth DD MM YYYY Patient identifiers Date of request Accession/Laboratory
Sponsored by Tumours of the Central Nervous System (CNS) Molecular Information Reporting Guide Family/Last name Given name(s) Date of birth DD MM YYYY Patient identifiers Date of request Accession/Laboratory
Five Most Common Problems in Surgical Neuropathology
 Five Most Common Problems in Surgical Neuropathology If the brain were so simple that we could understand it, we would be so simple that we couldn t Emerson Pugh What is your greatest difficulty in neuropathology?
Five Most Common Problems in Surgical Neuropathology If the brain were so simple that we could understand it, we would be so simple that we couldn t Emerson Pugh What is your greatest difficulty in neuropathology?
monotonous, stippled, round, smoothcontoured nuclei and scanty acidophilic or
 monotonous, stippled, round, smoothcontoured nuclei and scanty acidophilic or vacuolated cytoplasm. The cells are surrounded by a loose fibrillary stroma that is traversed by delicate capillaries. Ill
monotonous, stippled, round, smoothcontoured nuclei and scanty acidophilic or vacuolated cytoplasm. The cells are surrounded by a loose fibrillary stroma that is traversed by delicate capillaries. Ill
Karl Kashofer, Phd Institut für Pathologie Medizinische Universität Graz
 Expanding on WHO guideline compliant molecular testing of central nervous system tumors by low density whole genome sequencing. Karl Kashofer, Phd Institut für Pathologie Medizinische Universität Graz
Expanding on WHO guideline compliant molecular testing of central nervous system tumors by low density whole genome sequencing. Karl Kashofer, Phd Institut für Pathologie Medizinische Universität Graz
Biomarkers in Neuro-Ophthalmic Tumors
 Biomarkers in Neuro-Ophthalmic Tumors Fausto J. Rodríguez MD Department of Pathology Johns Hopkins University School of Medicine Disclosure of Relevant Financial Relationships Disclosure of Relevant Financial
Biomarkers in Neuro-Ophthalmic Tumors Fausto J. Rodríguez MD Department of Pathology Johns Hopkins University School of Medicine Disclosure of Relevant Financial Relationships Disclosure of Relevant Financial
