The role of the Wolffian ducts in the formation of the sinus vagina: an organ culture study
|
|
|
- Kerry Turner
- 6 years ago
- Views:
Transcription
1 /. Embryol. exp. Morph. 73, , Printed in Great Britain The Company of Biologists 1982 The role of the Wolffian ducts in the formation of the sinus vagina: an organ culture study By GABRIELE BOK AND ULRICH DREWS 1 From the Anatomisches Institut Universitdt Tubingen SUMMARY In mammals formation of a sinus vagina is inhibited in the male by endogenous testosterone from the embryonic testes. To answer the question which morphogenetic events during formation of the vagina are influenced by testosterone, we explanted genital tracts of mouse embryos in the indifferent stage of development in organ culture. Half of the explants were treated with testosterone and therefore developed in male direction. The other half was kept without testosterone and developed constitutively in female direction. Since the antimiiller factor was not present, in both types of cultures the Miillerian ducts were preserved. During female development the Miillerian ducts fused with the dorsolaterally apposed caudal segments of the Wolffian ducts. Thus the caudal segments of the Wolffian ducts were incorporated in the vaginal plate, while cranially the Wolffian ducts degenerated as expected. During male development fusion between Miillerian and Wolffian ducts did not occur. Under the influence of testosterone the respective caudal segments of the Wolffian ducts were surrounded by dense mesenchyme and further male differentiation took place. We conclude that the 'sinus protrusions' or 'sinovaginal bulbs' observed during development of the vagina, are in fact the caudal segments of the Wolffian ducts. They serve as a link between Miillerian ducts and urogenital sinus. Formation of a sinus vagina is prevented by testosterone simply by induction of male development in this area. INTRODUCTION The development of the vagina in man and other mammals has been studied extensively and repeatedly since the beginning of descriptive embryology. The descriptions are governed by the controversy about the derivation of the vagina. The various theories comprised derivation from the Miillerian ducts, from the Wolffian ducts, from the urogenital sinus, or from combinations of these structures. Today, in general, it is held that the upper part of the vagina is a derivative of the fused Miillerian ducts (Miillerian vagina), whereas the lower part is a derivative of the urogenital sinus (sinus vagina) (Forsberg, 1963; Jost, 1971; O'Rahilly, 1977). In our present organ culture study we observed that in the mouse embryo the lower segments of the Wolffian ducts were incorporated in the vaginal plate. As will be shown below, this observation also has a bearing on the interpretation of 1 Author's address: Anatomisches Institut, Universitat Tubingen, Osterbergstr. 3, D-7400 Tubingen, Germany.
2 276 G. BOK AND U. DREWS vagina development in the human. It will be discussed in the light of the formerly widely held view of a contribution of the Wolffian ducts to the vagina. We conclude from our observation that the 'sinovaginal bulbs' or 'sinus protrusions' described in man and other mammals, are in fact the caudal segments of the Wolffian ducts. In general terms the vagina in mammals develops as follows: in the indifferent stage of the genital ducts, the fused Mullerian ducts contact the wall of the urogenital sinus by means of a solid epithelial tip which merges with the epithelium of the dorsal wall of the sinus and bulges into its lumen. The solid epithelial structure is known as the Mullerian tubercle. Laterally and slightly caudally from the Mullerian tubercle, the Wolffian ducts enter the urogenital sinus. In contrast to the Mullerian ducts, the Wolffian ducts open freely into the sinus. During female development the mouth of the Wolffian ducts is transformed into an epithelial pocket carrying the degenerating Wolffian ducts at its cranial end. The bilateral pockets proliferate and form two solid epithelial bulbs, the so-called sinovaginal bulbs, extending as dorsolateral projections from the urogenital sinus. Cranially the bulbs fuse with the already fused Mullerian ducts. The Mullerian tubercle disappears or becomes incorporated into the solid vaginal plate which forms by fusion of the two bulbs with each other. Later on in the solid vaginal plate a lumen forms by necrosis of the central cells. In the mouse the sinovaginal bulbs are represented by open duct-like extensions of the urogenital sinus and were called 'sinus protrusions' by Forsberg (1963). ' The endocrine regulation of vaginal development is as follows: in the male the antimiillerian factor produced in the testes induces regression of the Mullerian ducts and thus prevents formation of a Mullerian vagina (Josso, 1973). The development of a sinus vagina is inhibited by testosterone. The latter notion is based on the following observations: exogenous androgens administered during pregnancy, inhibit the formation of a sinus vagina (Raynaud, 1942,1950). Treatment with antiandrogens leads to development of a sinus vagina in male embryos (Neumann et al. 1975). In the syndrome of testicular feminization (Tfm) in the mouse, characterized by general androgen insensitivity, the Mullerian vagina is inhibited whereas a sinus vagina is formed (Lyon & Hawkes, 1970). In our study we were interested in the morphogenetic mechanism leading to the formation of a sinus vagina and in the relationship of this process to the abnormal course of the male genital ducts in intersexes. In intersexes produced by sex reversal of Tfm heterozygotes the Wolffian ducts and the excretory ducts of the vesicular glands run down behind the sinus vagina and open into the urethra together with the vagina at the level of the penile bulb (Drews, 1975). The present organ culture experiment was designed to show the effect of testosterone on the formation of the sinus vagina. Embryonic mouse genital tracts were explanted in the indifferent stage of development. Half of the cultures were treated with testosterone and therefore developed in male direction.
3 Wolffian ducts and formation of sinus* vagina 277 The other half were kept without testosterone and developed constitutively in female direction. Since the testes were not included in the explants, the antimiiller-factor was not present. Correspondingly, the Miillerian ducts were maintained and developed equally well in both types of cultures. In explants kept without testosterone, the Miillerian ducts fused with the dorsolaterally apposed Wolffian ducts and thus allowed vaginal development to proceed. In explants kept with testosterone, no fusion occurred. Formation of a sinus vagina was prevented by preservation of the integrity and further differentiation of the Wolffian ducts. In addition to genital tracts from normal mouse embryos, we also explanted genital tracts from female embryos heterozygous for testicular feminization (X^/X) in order to observe the expression of the inherent mosaic of androgensensitive X + cells and androgen-insensitive X Tfm cells. In one explant the expression of the mosaic was observed: female development characterized by fusion of Wolffian and Miillerian ducts, alternated with male development represented by non-fusion and maintenance of the integrity of the Wolffian duct. MATERIALS AND METHODS Female NMRI mice obtained from a local commercial breeder or females from our own T/m-mouse colony were mated to NMRI males. Embryos were recovered on days 15,16 and 17, the day of plug detection being day 1. The sex of the embryos was determined by inspection of the gonads. The genital tracts were excised in Tyrode solution. Specimens were cleared ventrally and laterally from adhering tissues to render them as transparent as possible. They were trimmed by cutting off the upper parts of the genital ducts some distance above the bladder. Then the stem of sinus was cut at the level of the symphysis. The bladder itself was removed. The genital tracts were explanted with the dorsal or the lateral side up. The live explants were photographed daily through a dissecting microscope with a stereoadapter. The culture medium used was Dulbecco's MEM with 10 % horse serum, 10 % chick embryo extract and addition of Pen-Strep (50 units/ml). In part of the organ cultures, 10~ 6 M-testosterone was added to the medium (see Table 1). Instead of millipore filters used in earlier studies, a piece of a 1 % agar layer was used as support for the explants at the medium-air interface. The incubator was gased with 5 % CO 2. At the end of the culture period of up to 8 days, explants were fixed in Bouin's solution, oriented with stained agar blocks according to Arnolds (1978), and embedded in Paraplast. Serial sections were prepared and stained with haematoxylin-eosin and HOP A (Haematoxylin, Orange G, Phosphomolybdenic acid, Aniline blue) after Tonutti (Tonutti et al. 1960). The reconstructions were compiled from serial sections with the Perspectomat
4 278 G. BOK AND U. DREWS genital ducts sinus ridges if*. fused Miillerian ducts m*...f elongation into sinus pulsating urethra Fig. 1. Stereophotographs of a living explant from day 15 of pregnancy kept without testosterone, on the first day (A), on the third day (B) and on the fifth day (C) in culture (Exp. No. 13, Table 1). Female development. The outlines of the genital ducts include Wolffian and Miillerian ducts. Fusion and secondary elongation of the Miillerian ducts is visible on day 3 in culture. Downgrowth of the vaginal anlage is evident, particularly when compared to male development shown in Fig. 2. The lower part of the urethra begins to contract rhythmically after a culture period corresponding to day 17 of pregnancy. The explant lies with the dorsal side up on the agar. X44.
5 Wolffian ducts and formation of sinus vagina 279 genital ducts sinus ridges 2A fused Miillerian ducts B ureteric cysts prostate buds pulsating urethra Fig. 2. Stereophotographs of a living explant from day 15 of pregnancy kept with testosterone on the first (A), on the third (B), and on the fifth day (C) in culture. Male development. The Miillerian ducts are preserved and fusion is visible as in female development. Elongation and downgrowth do not occur. Enlargement and thickening of the sinusridgesis followed by outgrowth of multiple prostatic buds and appearance of vesicular glands. The explant lies with the dorsal side up on the agar. x44.
6 280 G. BOK AND U. DREWS P 40 of F. Forster, Schaffhausen, Switzerland. The drawing apparatus transforms into parallel perspective the outlines of epithelial structures drawn from the microscope with a drawing mirror. It allows to generate directly a threedimensional picture by superposition of the outlines in the calculated distance. RESULTS The following description is based on the list of cultures in Table 1. As indicated, the genital tracts were explanted on days 15,16 or 17 of pregnancy, the day of plug detection being day 1. The culture period is given in days corresponding to days of pregnancy. At the time of explantation, the testis and the ovary were clearly distinguishable by inspection in the dissecting microscope. Correspondingly, explants from embryos with ovaries were designated as XX and those from embryos with testes as XY. In each experimental series, half of the explants were cultured with testosterone and the other half without testosterone, so that treated and untreated explants from the same litter could be compared directly. Male embryos (XY) were always included in the group with testosterone. Comparable untreated and treated explants were fixed for histology in daily intervals covering the entire culture period. For the sake of clarity we will separately describe the morphology of the explants in the indifferent stage at the beginning of the culture period, then the female development of explants cultured without testosterone, the male development induced by testosterone, and finally the observations in Tfm heterozygotes. The description refers to stereophotographs of the live explants taken in daily intervals, to the serial sections, and to the representative graphic reconstructions thereof. Indifferent stage In the mouse the Miillerian ducts reach the urogenital sinus at day 14 of pregnancy. In the respective area beneath the bladder, the sinus is typically U shaped in cross section with two sinus ridges or dorsal urethral folds facing the rectum (Forsberg, 1963). The Wolffian ducts enter the sinus ridges dorsally, whereas the Miillerian ducts contact the sinus ridges slightly cranially and medially from the latter. In this region it is not before day 16 that a sex difference becomes visible. After excision of the genital tract from the embryo on day 15 or 16 of pregnancy, the area of contact between Wolffian and Miillerian ducts with the urogenital sinus appeared as a characteristic rhombic figure when viewed dorsally in dark-field transmission illumination through the dissecting microscope. The lower crura were formed by the sinus ridges, whereas the upper crura corresponded to the superposed epithelial outlines of both genital ducts. Several hours after explantation the explants flattened and the tissue again became translucent. Fig. 1A and 2B show explants of day 15 photographed at the day of
7 Wolffian ducts and formation of sinus vagina 281 explantation. Both were explanted with the dorsal side up. The rhombus formed by the sinus ridges and the genital ducts can be recognized. Individual Wolffian and Mullerian ducts however cannot be designated with certainty. Both explants represent the indifferent stage from which female development starts off in cultures without testosterone (Fig. 1) and male development in cultures with testosterone (Fig. 2). To visualize the exact position of the ducts at the beginning of the culture period, three explants of day 15 and two of day 16 were fixed and processed for histology on the same day after they had been oriented on the agar and grown for about 5 h (Table 1). Figure 3 shows a specimen of day 16 explanted with the dorsal side up. In the centre, two closely apposed Mullerian ducts and two dorsolaterally located Wolffian ducts can be seen just before entering the sinus. Probably due to damage during cutting and handling, some cells in the mesenchyme undergo necrosis whereas in the epithelium of the ducts mitotic figures are visible. Figure 7 shows a graphic reconstruction of a specimen explanted at day 16 of pregnancy and fixed for histology the same day. The ducts are still in the indifferent stage of development. The specimen is comparable to those of day 15. The reconstruction shows only the upper half of the rhombus visible in the living explants (compare Fig. 1A and 2A). The upper crura of the rhombus are formed by the superposed Wolffian of Mullerian ducts. Development without testosterone During female development in vivo, the Mullerian ducts fuse in the midline and the Wolffian ducts degenerate. Degeneration of the Wolffian ducts starts on day 16 of pregnancy midway between the gonads and the point of contact with the sinus and proceeds cranially and caudally up to day 19. Our cultures kept without testosterone developed constitutively in the female direction. Although only the lower ends of the genital ducts were included in the explants, fusion of the Mullerian ducts and degeneration of the Wolffian ducts followed the same time schedule as in vivo. In the living explants, fusion and enlargement of the Mullerian ducts was clearly visible after 2 days in culture (Fig. IB). The fused lower segments of the Mullerian ducts appeared as a bell-shaped central thickening in the upper half of the rhombus visible in the preceding indifferent stage. With further development two crura emerged from the lower base of the bell-shaped structure connecting the latter with the lateral sinus ridges (Fig. IB and C). The crura represented the Mullerian ducts elongating caudally to fuse with the lower segments of the Wolffian ducts. A clear outline of the Wolffian ducts was not visible in the living explants; they were hidden in the pronounced bell-shaped structure of the fused Mullerian ducts. From the histology of the explants described below, it became clear that of the caudal straight portions of the Wolffian ducts included in the explants, the lower two thirds remained intact and finally fused with the Mullerian ducts.
8 Table 1. List of explants and time schedule of cultivation The explants described belong to four experiments conducted on days 15, 16 and 17 of pregnancy. Explants from embryos with ovaries are designated as XX and those from embryos with testes as XY or Tfm/Y when carrying testicular feminization. They are listed on the day of interruption and fixation for histology. The time schedule shows that each day is represented by comparables cultures kept without testosterone (0T) and with testosterone (+T). Exp. No 7 Tfm/+ 9 (1) XX(3) 0T XX(1 Tfm/YCl XY(2' XX(1) 0T Exp. No 13 NMRI 9 (1) XY(5) +T XY(2) +T Exp. No 6 Tfm/+ 9 (4) XX(2) 0T XX(2) 0T XX(2) +T XX(2) 0T XX(2) +T XX(2) 0T XX(2) +T XX(1) 0T XX(1) +T Exp. No 3 Tfm/+ 9 (4) days Culture period corresponding to days of pregnancy r/m/y(l) 0T (2) +T XX(6) 0T Tfm/Yh) 0T XY(1) +T to oo O w o D W
9 Wolffian ducts and formation of sinus vagina 283 Fig. 3. Histology of an explant from day 16 of pregnancy after 5 h in culture. Indifferent stage of development. The closely apposed genital ducts and a fragment of the urogenital sinus are visible. Wd = Wolffian duct; Md = Miillerian duct; ugs = urogenital sinus. x95. The lower half of the rhombus seen in the living explants and representing the sinus ridges, was transformed into a cup-like structure into which the fused Miillerian ducts sank down (Fig. IB and C). When oriented in a lateral position on the agar (not shown) the cup of the sinus ridges carrying the genital ducts bulged dorsally like a backpack at the neck of the urethra. This characteristic configuration appears also in vivo during female development. In the living explants, development could be followed easily up to day 19 (Fig. 1C). On prolonged culture periods as in Exp. No. 13, Table 1, the explants became opaque and clumsy in outline although the histological structure was still well preserved. On day 17 in most of the explants the caudal part of the urethra started to contract rhythmically. The contractions finally pulled the urethra into an abnormal ventrally bent position (Fig. 1C and 2C). The contractions were independent of testosterone and have already been described by Cunha (1973). They are, however, not generated by a smooth muscle layer as stated by Cunha, but by the cross-striated urethral muscle of the mouse (Thiedemann & Drews, 1980). The characteristic features of the explants grown without testosterone were secondary elongation of the fused Miillerian ducts, preservation of the caudal segments of the Wolffian ducts, and finally longitudinal fusion of Wolffian and Miillerian ducts. The fusion of Wolffian and Miillerian ducts is demonstrated in the serial sections of a day-19 explant after 3 days in culture (Fig. 5). Cranially the degenerating remnants of the Wolffian ducts are found in a dorsolateral position attached to the enlarged and fused Miillerian ducts (Fig. 5A). More caudally, the Miillerian ducts segregate again (Fig. 5B). The Wolffian ducts gradually enlarge and the cuboidal epithelium with signs of degeneration is replaced by high columnar epithelium showing many mitoses. Concomitantly, the Wolffian epithelium acquires the characteristic appearance of Miillerian
10 284 G. BOK AND U. DREWS % N W-:; ':i;^^.^^. r-,. Fig. 4. Histology of an explant from day 16 of pregnancy after 1 day in culture with testosterone. (A) Level of fused Mullerian ducts. (B) On the left side of the explant Wolffian and Mullerian ducts fuse with each other thus indicating the presence of a X r / m /X + mosaic. (C) Entrance of the ducts into the urogenital sinus. The explant was oriented in a lateral position. The corresponding graphic reconstruction is shown in Fig. 8. x95. fmd = fused Mullerian ducts; Md = Mullerian duct; Wd = Wolffian duct; ugs = urogenital sinus.
11 Wolffian ducts and formation of sinus vagina 285 epithelium and after fusion with the latter, the contribution of Wolffian epithelium to the vagina can be hardly identified except by location (Fig. 5B and C). The fused Wolffian and Miillerian ducts merge with the solid sinus ridges. Thereby the original free opening of both Wolffian and Miillerian ducts into the urogenital sinus is lost (Fig. 5D). Female development of the explants is visualized by the reconstructions in Figs 7-10, which were all prepared in the same scale from serial sections of specimens of experiment No. 6 (Table 1). Figure 7 shows a specimen in the indifferent stage of day 16 fixed on the day of explantation. The specimen of Fig. 8 was treated with testosterone and expressed the phenotype of a Tfm heterozygote. In this context it may be used to demonstrate fusion and enlargement of the Miillerian ducts at day 17 which occurred likewise in treated and untreated cultures. Figure 9 shows an explant of day 18. In this case major asymmetries developed probably due to inadequate orientation on the agar. At the left side the contact between the lower end of the Wolffian duct and the left Miillerian duct is not established, whereas on the right side fusion occurred regularly. The cranial portions of the Wolffian ducts have already regressed. Figure 10 is the reconstruction of the female specimen of day 19, the serial sections of which are shown in Fig. 5A-D. In the Wolffian ducts two parts can be distinguished. In the degenerating upper parts the ducts are small and taper off cranially. The lower parts are enlarged and fuse over their whole length with the elongating roots of the Miillerian ducts. The fused ducts of both sides merge with the dorsal sinus ridges, which have developed into prominent 'buttocks' at the dorsal aspect of the urethra (compare the living explants in Fig. 1C). Development with testosterone The development of the explants treated with testosterone corresponded to male development in vivo with one important exception: the antimiillerian factor was missing and therefore the Miillerian ducts were maintained and developed as in the female. This fact was constitutive for our experimental design. It allowed close comparison of the male and the female phenotype by using the Miillerian ducts as a landmark common to both types of explants. On the other hand, differences observed in the development of the sinus vagina were restricted to effects of testosterone. In general, cultures treated with testosterone exhibited a better overall growth (compare Fig. 1 and 2). Mesenchymal and epithelial tissues appeared more healthy. Necrotic areas in the centre of the cultures were less frequently observed and were less extended. Even the fused Miillerian structures seemed to profit from trophic influences exerted by testosterone. After 2 days in culture, corresponding to day 17 of pregnancy (Fig. 2B), the central bell-shaped outline of the fused Miillerian ducts was clearly visible as in female development. The sinus ridges also transformed into a cup-like structure. The projection of the combined sinus ridges dorsally and laterally can easily be EMB73
12 286 G. BOK AND U. DREWS Figs 5 & 6
13 Wolffian ducts and formation of sinus vagina 287 seen in the stereophotographs of Fig. 2B and C. However, formation of segregating crura and caudal elongation of the Miillerian ducts did not occur. In contrast to the development without testosterone, the fused Miillerian ducts did not move down between the sinus ridges. Instead, at the upper rim of the cup formed by the ridges, a number of epithelial buds appeared which were well developed after 4 days in culture (Fig. 2C). The buds represented the vesicular glands arising from the base of the Wolffian ducts and the prostate glands formed by the adjacent sinus epithelium. The histology of the explants cultured with testosterone is shown in Fig. 6A-D. The respective explant stems from experiment No. 6, day 19, and is directly comparable with the explant of Fig. 5A-D which stems from the same litter and the same day the only difference being treatment with testosterone. During male development the Wolffian ducts were maintained throughout the whole length of the specimens in about the same size as the Miillerian ducts. In contrast to female development no fusion occurred between Wolffian and Miillerian ducts. After day 17, the Wolffian epithelium showed signs of stimulated growth with enlargement of the ducts and appearance of many mitotic figures. On day 18 the buds of the vesicular glands appeared as thickening and doubling of the epithelial lining (Fig. 6B, C). On day 19, the vesicular glands formed flat cap-like protrusions at the dorsomedial aspects of the Wolffian ducts. In Fig. 6B and C and the corresponding reconstruction Fig. 11 the buds are still included in the Wolffian epithelium and extend over the whole length of the lower segments of the ducts. The explant of experiment 13 (Table 1) shown in Fig. 13 and in the stereophotographs of Fig. 2C is slightly further developed. The tips of the vesicular glands have now separated from the Wolffian ducts. Without testosterone the Miillerian ducts elongated caudally by fusion with the dorsolaterally located Wolffian ducts. With testosterone this fusion did not occur. By the enlarging Wolffian structures the Miillerian ducts were pushed together and restricted to the original point of contact with the sinus. In cross sections they formed a characteristic triangular roof over the dorsal aspect of the urethra (Fig. 6A, B), wedged in between the expanding Wolffian ducts and the vesicular glands. Besides the changes in the epithelial ducts we observed also Fig. 5. Histology of an explant of day 16 of pregnancy grown for 3 days in culture without testosterone. Female development. (A) Fused Mullerian ducts with dorsolaterally attached degenerating Wolffian ducts (on the right the specimen was damaged after fixation). (B) The Mullerian ducts segregate again. The caudal portions of the Wolffian ducts are well preserved. (C) Wolffian and Mullerian ducts fuse. (D) The combined ducts merge with the sinus ridges. The graphic reconstruction of the explant is shown in Fig. 10. Abbreviations as in Fig. 4. x95. Fig 6. Serial sections of an explant of day 16 of pregnancy grown for 3 days in culture with testosterone. Male development. (A) Wolffian and fused Mullerian ducts show stimulated growth. (B) and (C) On the dorsal aspect of the Wolffian ducts the buds of the vesicular glands appear. The Mullerian ducts form a triangular roof at their original point of contact with the urogenital sinus. (D) From the sinus ridges (sr) prostate buds arise. The graphic reconstruction of the explant is shown in Fig. 11. X95.
14 288 G. BOK AND U. DREWS ugs ugs XX indifferent day 16 Fig. 7. Graphic reconstruction of an explant of day 16 in the indifferent stage fixed at the day of explantation. The reconstructed epithelial structures correspond to the upper half of the rhombus seen in the living explants of Figs 1A and 2A. The reconstructions Figs 7-11 are taken from experiment No. 6 (Table 1) and drawn to the same scale. Md = Miillerian duct; Wd = Wolffian duct; ugs = urogenital sinus; sr = sinus ridges. X T f m /x + + testosterone day 17 Fig. 8. Graphic reconstruction of an explant from day 16 of pregnancy cultured for one day with testosterone. Intersex development. The area of fusion between the left Wolffian and Miillerian duct indicates the presence of androgen insensitive X^cells in this region. Same explant as in Fig. 4. fmd Wd fused Md + Wd XX day 18 Wd Fig. 9. Graphic reconstruction of an explant from day 16 of pregnancy kept for 2 days in culture without testosterone. Female development. Due to inadequate orientation on the agar the contact between the left Wolffian and Miillerian duct is not established, whereas on the right side fusion of the ducts occurred regularly.
15 fmd Wolffian ducts and formation of sinus vagina degen. Wd XX day 19 Fig. 10. Graphic reconstruction of an explant from day 16 of pregnancy kept for 3 days in culture without testosterone. Female development. The fused Miillerian ducts segregate again and elongate caudally. The lower ends of the Wolffian ducts fuse longitudinally with the Miillerian ducts, while the upper parts degenerate. Same explant as in Fig. 5. Wd fmd fmd Wd XX + testosterone day 19 Fig. 11. Graphic reconstruction of an explant from day 16 of pregnancy kept for 3 days in culture with testosterone. Male development. Fusion between Wolffian and Miillerian ducts does not occur. The Wolffian ducts are well preserved and the appearance of vesicular glands is indicated by a dorsal thickening of the epithelium. Same explant as in Fig. 6.
16 290 G. BOK AND U. DREWS Fig. 12. Section of an explant from day 15 of pregnancy cultured for 4 days without testosterone. (Experiment No. 13, Table 1). Below the entrance of the genital ducts lateral furrows divide the urogenital sinus (ugs) into an urethral and a vaginal part (vag). X59. Fig. 13. Similar explant as in Fig. 12 cultured with testosterone. Appearance of vesicular glands (sv) and prostate buds (pr). Same explant as in Fig. 2C. x59. differences in the behaviour of the mesenchyme. In explants kept with testosterone densely packed circular mesenchyme appeared around the Wolffian ducts which was not present in explants cultured without testosterone (compare Fig. 5Cand6C). In the reconstructions male development can be followed starting out from the indifferent stage of Fig. 7 through the stage with fused Miillerian ducts as in Fig. 8, to the fully expressed male phenotype in Fig. 11. Although Fig. 8 represents a Tfm heterozygote as described below, it can be used to visualize the general development of Wolffian and Miillerian ducts on day 17. Comparison of Fig. 11
17 Wolffian ducts and formation of sinus vagina 291 with the corresponding female stage in Fig. 10 demostrates the enlargement of the Wolffian ducts, the lack of fusion with the Miillerian ducts, and the compression and retention of the Miillerian ducts at their original point of contact with the urogenital sinus. Influence of Tfm in the heterozygous state As indicated in Table 1 in experiments No. 3, No. 6 and No. 7, the mothers were heterozygous for Tfm (testicular feminization). Therefore, embryos with testes were either normal male (XY) or Tfm embryos (Tfm/Y). On day 15 of pregnancy, normal XY and Tfm embryos were distinguished by co-culture of testis and Wolffian body as described in Hannappel etal. (1981). On day 16 male embryos were discarded. On day 17, Tfm embryos were identified by direct inspection of the Wolffian body. The Tfm mutation of the mouse is characterized by complete androgen insensitivity. Since Tfm is X linked, heterozygotes are X-inactivation mosaics with respect to androgen sensitivity. The mosaic of androgen-sensitive X + cells and androgen-insensitive X Tfm cells is, however, only expressed in androgendependent male development. Without testosterone both cell types follow constitutively the female pathway of development. Experiment No. 6, Table 1, was especially designed to show the effects of the mosaic in the development of the genital tract. Only XX embryos with ovaries were used. Because of the segregation of the maternal X chromosomes, about half of the embryos were expected to be heterozygous for Tfm. Embryos with ovaries thus were either normal females or females heterozygous for 77m. It was not possible to tell the Tfm heterozygotes in advance. Without testosterone genital tracts from Tfm heterozygotes develop as normal females. Therefore they were carried on as XX. Only in explants cultured with testosterone was there a chance to identify a Tfm heterozygote by the expression of the X- inactivation mosaic. The explants were examined according to the criteria for morphogenetic behaviour characteristic for development without or with testosterone as derived from the explants of experiments No. 7 and No. 13 (Table 1). After all in only one explant fixed on day 17, were there clear indications that we were dealing with a Tfm heterozygote. The explant is documented in the reconstruction of Fig. 8 and in the sections of Fig. 4. On the left hand Wolffian and Miillerian ducts fused with one another as in female development. However, as can be seen in the reconstruction of Fig. 8, fusion was restricted to a short segment of the ducts. Above and below this area and on the right side of the explant the ducts separated as in male development. This observation indicated that in the left Wolffian duct an area with androgen-insensitive cells was present in the epithelium or even more likely in the Wolffian mesenchyme. In this area, fusion with the Miillerian ducts could not be prevented by testosterone. Since expression of the mosaic was observed in only one specimen, this finding cannot be generalized. Further experiments are in progress.
18 292 G. BOK AND U. DREWS DISCUSSION The nowadays generally accepted view of the development of the vagina is based on the work of Koff (1933). He described the development of the vagina in human embryos ranging from 11 mm to 169 mm. On the basis of graphic representations drawn to scale from serial sections and from wax-plate reconstructions, he concluded that the vaginal plate is formed by proliferation of two epithelial pockets arising from the sinus epithelium. He coined the term 'sinovaginal bulbs' which is now widely used in text books. According to the description of Koff (1933), the entire sinus vagina is derived from the sinovaginal bulbs and is only of sinus origin in so far as the bulbs evaginated from the sinus. His interpretation implies that the vaginal plate arises by upgrowth of the sinovaginal bulbs. On weighing the arguments, however, as done by O'Rahilly (1977) in his comprehensive review on the development of the human vagina, one realizes that other interpretations have not been definitely excluded. In particular, this holds true for the formerly widely held opinion that the vaginal plate grows down to the perineum and that the Wolffian ducts contribute to its formation. The epithelial pockets which form the sinovaginal bulbs are continuous with the remnants of the Wolffian ducts. Therefore, the first investigators obviously took it forgranted that the epithelial pockets were identical to the lower parts of the Wolffian ducts. Correspondingly, Hart (1901) called the structures 'Wolffian bulbs' and assumed, that the lower third of the vagina down to the hymen was of Wolffian duct origin. With our own observations in mind, statements such as that of Hart (1901) appear quite convincing: 'In two previous communications I discussed the questions of the origin of the hymen and vagina. I there attempted to show that the lower ends of the Wolffian ducts enter into the formation of the former, and that the latter was Miillerian in origin only in its upper two-thirds, the lower third being formed by blended urogenital sinus and Wolffian ducts' (p. 330). The quotation seems to indicate that the role of the Wolffian ducts in vagina development was a clear fact from the beginning. However this is not the case. A closer look at the evidence presented by Hart shows that it consists of a combination of scanty observations in human embryos with intelligent interpretations of adult female anatomy and far-stretched reasoning in comparative embryology by combining ideas on gastrulation and germ-layer theories with formation of the vagina of marsupials. Mijsberg (1924) used the same terms ('Wolff sche Blaschen' and 'Wolff'sche Hocker'). He was convinced that only a small segment of the vaginal anlage was of Wolffian origin located between the upper-miillerian-derived and the lowersinus-derived portions. In our eyes, his statement is also amazingly true: 'The meaning of the fusion of Wolffian and Miillerian ducts at their lower ends which in the human leads to the situation that the Miillerian ducts are connected to the sinus via the Wolffian ducts, is hard to understand. Also in other species a similar fusion occurs' (p. 746). With our knowledge on the endocrine regulation of sex
19 Wolffian ducts and formation of sinus vagina 293 organ development, we are tempted to add: incorporation of Wolffian duct tissue in the vaginal plate makes sense in respect to hormonal regulation. It allows for the switch off of constitutive vaginal development in the male by simply inducing male development in the respective duct segments via testosterone. In his monograph on the derivation and differentiation of the vaginal epithelium Forsberg (1963), in principle, followed Koff (1933). In his description of vaginal development in the mouse and other mammals he used the term 'sinus protrusions' instead of sinovaginal bulbs. However, in his concluding remarks on the histochemical characterization of the epithelium of Miillerian and Wolffian ducts and of the urogenital sinus in human embryos he states: 'After taking into consideration the circumstances that may have influenced the enzyme activity in the different epithelia, it must be pointed out that the distribution of acid phosphatase, esterase, and arylsulphatase, possibly also leucine aminopeptidase, favours the view that the vaginal plate is a Wolffian derivate; particularly as the morphological investigations also suggest this. Another possibility is that the Wolffian epithelium induces a cranial growth of the sinus epithelium, thereby changing its character' (p. 113). From the above eclectic citations and even more from the comprehensive discussion of the topic by O'Rahilly (1977) it appears that the question of derivation of the vagina cannot be settled solely by classical methods of descriptive embryology. The same is true for the closely related controversy of upgrowth of sinovaginal bulbs versus downgrowth of Wolffian epithelium. Our in vitro approach had the advantage that morphogenetic movements were directly visible in the living explants. Direct comparison of constitutive female and testosterone-induced male development allowed the unequivocal follow up of the fate of the structures in question. The characteristic behaviour of Miillerian and Wolffian ducts during female or male development is visualized in Fig. 14, which is based on the reconstructions of the explants of day 19. The location of the ducts and their histological appearance in cultures without testosterone corresponded to the in vivo situation in the female mouse embryo as described by Forsberg (1963). However, our observations show that the 'sinus protrusions' of Forsberg are in fact identical to the lower segments of the Wolffian ducts. Witschi (1970) assumed that in the human embryo the 'sino-vaginal bulbs' of Koff (1933) were identical to the lower segments of the Wolffian ducts. He reexamined the embryos used by Koff and concluded, that the vaginal plate arises by downgrowth behind the urogenital sinus, and that the Wolffian ducts take part in its formation. In the drawing of Koff's mm human embryo re-interpreted by Witschi and reproduced by O'Rahilly (fig. 2, p. 129 in O'Rahilly (1977)), the location and pattern of coalescence of Miillerian ducts and Wolffian ducts are identical to that exhibited by our day-19 specimen without testosterone (Fig. 14A). In his fig. 3 (p. 130) O'Rahilly (1977) compares as equivalent the 'classical
20 294 G. BOK AND U. DREWS XX day 19 without testosterone with testosterone Fig. 14. Female and male development drawn after the reconstructions of Figs 10 and 11. Without testosterone the Miillerian ducts elongate caudally and fuse with the Wolffian ducts. Thus the starting point for the vaginal plate is formed which elongates further by separation from the urogenital sinus. With testosterone fusion of Wolffian and Miillerian ducts is prevented by male differentiation of the respective Wolffian duct segments. The formation of a sinus vagina is thus inhibited. view' of Koff assuming upgrowth of the vaginal plate with Witschi's view of vaginal downgrowth and incorporation of Wolffian ducts. Our experiment demonstrates that Witschi's view was correct. We thank Dr E. Heywinkel-Hannappel for help in preparing the explants and the stereophotographs and Mrs A. Show-Klett for technical assistance. Supported by the Deutsche Forschungsgemeinschaft (Dr 94/4). The contribution of G. Bok will be submitted as inaugural dissertation at the Fakultat Theoretische Medizin der Eberhard-Karls-Universitat Tubingen. REFERENCES ARNOLDS, W. J. A. (1978). Oriented embedding of small objects in agar-paraffin, with reference marks for serial section reconstruction. Stain Techn 53, CUNHA, G. R. (1973). Peristaltic contractions of the murine urogenital sinus. Anat. Rec. 177, DREWS, U. (1975). Direct and mediated effects of testosterone: the development of intersexes in sex reversed mosaic mice, heterozygous for testicular feminization. Anat. Embryol. 146, FORSBERG, J. -G. (1963). Derivation and Differentiation of the Vaginal Epithelium. Institute of Anatomy, Lund: PhD thesis. HANNAPPEL, E. & DREWS, U. (1981). Stimulation of growth by testosterone via the mesenchyme. Recombination of tissues from Tfm and wild-type preputial gland anlagen of mouse embryo. Cell Tissue Res. 221, HART, D. BERRY (1901). A contribution to the morphology of the human urogenital tract. /. Anat. Physioi, Paris 35, Josso, N. (1973). In vitro synthesis of Mullerian-inhibiting hormone by seminiferous tubules isolated from the calf foetal testis. Endocrinology 93, JOST, A. (1971). Embryonic sexual differentiation (morphology, physiology, abnormalities).
21 Wolffian ducts and formation of sinus vagina 295 In: Hermaphroditism, Genital Anomalies and Related Endocrine Disorders. 2nd edition (eds H. W. Jones Jr. & W. W. Scott), p. 16. Baltimore: The Williams and Wilkins Co. KOFF, A. K. (1933). Development of the vagina in the human foetus. Contr. Embryol. No. 140, Carnegie Inst. 24, LYON, M. F. & HAWKES, S. G. (1970). X-linked gene for testicular feminization in the mouse. Nature 227, MIJSBERG, W. A. (1924). Uber die Entwicklung der Vagina, des Hymen und des Sinus urogenitalis beim Menschen. Z. Anat. EntwGesch. 74, NEUMANN, F. ET AL. (1975). The role of androgens in sexual differentiation of mammals. In Intersexuality in the Animal Kingdom, (ed. R. Reinboth), pp Berlin, Heidelberg, New York: Springer-Verlag. O'RAHILLY, R. (1977). The development of the vagina in the human. In: Morphogenesis and Malformations of the Genital System. Birth defects original article series 13 (2) (eds Richard J. Blandau & Daniel Bergsma), pp New York. RAYNAUD, A. (1942). D6veloppement des Glandes Annexes du Tractus Genital de la Souris. C. r. Sianc. Soc. Biol. 136, RAYNAUD, A. (1950). Recherches Experimental sur le Developpment de L'Appareil Genital et le Fonctionnement des Glandes Endocrines des Foetus de Souris et de Mulot. Archs. Anat. microsc. Morph. exp. 39, THIEDEMANN, K. -U. & DREWS, U. (1980). Nuclei in testicular feminization (Tfm) are not activated by intact testosterone receptor complexes: a morphometric study in striated urethral muscle of mosaic mice. Cell Tissue Res. 212, TONUTTI, E., WELLER, O., SCHUCHARDT, E. & HEINKE, E. (1960). Die Mdnnliche Keimdruse, p.175. Stuttgart: Georg Thieme Verlag, S. WITSCHI, E. (1970). Development and differentiation of the uterus. In: Prenatal Life, (ed. H. C. Mack), pp Detroit: Wayne State University Press. (Accepted 28 September 1982)
22
Growth pattern of the sex ducts in foetal mouse hermaphrodites
 /. Embryol. exp. Morph. 73, 59-68, 1983 59 Printed in Great Britain The Company of Biologists Limited 1983 Growth pattern of the sex ducts in foetal mouse hermaphrodites By C. YDING ANDERSEN 1, A. G. BYSKOV
/. Embryol. exp. Morph. 73, 59-68, 1983 59 Printed in Great Britain The Company of Biologists Limited 1983 Growth pattern of the sex ducts in foetal mouse hermaphrodites By C. YDING ANDERSEN 1, A. G. BYSKOV
under its influence, male development occurs; in its absence, female development is established.
 Sex differentiation is a complex process that involves many genes, including some that are autosomal. The key to sexual dimorphism is the Y chromosome, which contains the testis determining gene called
Sex differentiation is a complex process that involves many genes, including some that are autosomal. The key to sexual dimorphism is the Y chromosome, which contains the testis determining gene called
Development of the Genital System
 Development of the Genital System Professor Alfred Cuschieri Department of Anatomy University of Malta The mesonephros develops primitive nephrotomes draining into a mesonephric duct nephrotome mesonephric
Development of the Genital System Professor Alfred Cuschieri Department of Anatomy University of Malta The mesonephros develops primitive nephrotomes draining into a mesonephric duct nephrotome mesonephric
Normal and Abnormal Development of the Genital Tract. Dr.Raghad Abdul-Halim
 Normal and Abnormal Development of the Genital Tract Dr.Raghad Abdul-Halim objectives: Revision of embryology. Clinical presentation, investigations and clinical significance of most common developmental
Normal and Abnormal Development of the Genital Tract Dr.Raghad Abdul-Halim objectives: Revision of embryology. Clinical presentation, investigations and clinical significance of most common developmental
Experimental contributions to the study of the embryology of the vagina
 Human Reproduction Vol.21, No.6 pp. 1623 1628, 2006 Advance Access publication February 13, 2006. doi:10.1093/humrep/del031 Experimental contributions to the study of the embryology of the vagina M.L.Sánchez-Ferrer
Human Reproduction Vol.21, No.6 pp. 1623 1628, 2006 Advance Access publication February 13, 2006. doi:10.1093/humrep/del031 Experimental contributions to the study of the embryology of the vagina M.L.Sánchez-Ferrer
Sexual differentiation:
 Abnormal Development of Female Genitalia Dr. Maryam Fetal development of gonads, external genitalia, Mullerian ducts and Wolffian ducts can be disrupted at a variety of points, leading to a wide range
Abnormal Development of Female Genitalia Dr. Maryam Fetal development of gonads, external genitalia, Mullerian ducts and Wolffian ducts can be disrupted at a variety of points, leading to a wide range
Developmental Changes of Müllerian and Wolffian Ducts in Domestic Cat Fetuses
 Exp. Anim. 58(1), 41 45, 2009 Note Developmental Changes of Müllerian and Wolffian Ducts in Domestic Cat Fetuses Tomo INOMATA 1), Hiroyoshi NINOMIYA 1), Katsuyasu SAKITA 1), Naomi KASHIWAZAKI 2), Junya
Exp. Anim. 58(1), 41 45, 2009 Note Developmental Changes of Müllerian and Wolffian Ducts in Domestic Cat Fetuses Tomo INOMATA 1), Hiroyoshi NINOMIYA 1), Katsuyasu SAKITA 1), Naomi KASHIWAZAKI 2), Junya
W.S. O University of Hong Kong
 W.S. O University of Hong Kong Development of the Genital System 1. Sexual differentiation 2. Differentiation of the gonads a. Germ cells extragonadal in origin b. Genital ridge intermediate mesoderm consisting
W.S. O University of Hong Kong Development of the Genital System 1. Sexual differentiation 2. Differentiation of the gonads a. Germ cells extragonadal in origin b. Genital ridge intermediate mesoderm consisting
Development of the urogenital system
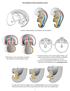 Development of the urogenital system Location of the pronephros, mesonephros and metanephros Differentiation of the intermedierm mesoderm into nephrotome and mesonephric tubules Connection between aorta
Development of the urogenital system Location of the pronephros, mesonephros and metanephros Differentiation of the intermedierm mesoderm into nephrotome and mesonephric tubules Connection between aorta
EMBRYOLOGIC BASIS OF FEMALE CONGENITAL TRACT MALFORMATIONS
 EMBRYOLOGIC BASIS OF FEMALE CONGENITAL TRACT MALFORMATIONS Prof. Pedro Acién, MD, FRCOG Maribel Acién, MD San Juan University Hospital/ Miguel Hernández University Campus of San Juan. Alicante. Spain Instituto
EMBRYOLOGIC BASIS OF FEMALE CONGENITAL TRACT MALFORMATIONS Prof. Pedro Acién, MD, FRCOG Maribel Acién, MD San Juan University Hospital/ Miguel Hernández University Campus of San Juan. Alicante. Spain Instituto
(Received 16th October 1969)
 EFFECT OF AN ANTI-ANDROGEN ON THE DIFFERENTIATION OF THE INTERNAL GENITAL ORGANS IN DOGS H. STEINBECK, F. NEUMANN and W. ELGER Main Laboratory of Schering AG, Berlin, West Germany (Received 16th October
EFFECT OF AN ANTI-ANDROGEN ON THE DIFFERENTIATION OF THE INTERNAL GENITAL ORGANS IN DOGS H. STEINBECK, F. NEUMANN and W. ELGER Main Laboratory of Schering AG, Berlin, West Germany (Received 16th October
11. SEXUAL DIFFERENTIATION. Germinal cells, gonocytes. Indifferent stage INDIFFERENT STAGE
 11. SEXUAL DIFFERENTIATION INDIFFERENT STAGE Early in pregnancy, (within 10-15 % of the pregnancy s expected length) a genital ridge is formed in the sides of the embryonic tissue, ventral to the mesonephros
11. SEXUAL DIFFERENTIATION INDIFFERENT STAGE Early in pregnancy, (within 10-15 % of the pregnancy s expected length) a genital ridge is formed in the sides of the embryonic tissue, ventral to the mesonephros
Mesenchymal control over elongating and branching morphogenesis in salivary gland development
 J. Embryol. exp. Morph. Vol. 66, pp. 9-, 98 9 Printed in Great Britain Company of Biologists Limited 98 Mesenchymal control over elongating and branching morphogenesis in salivary gland development ByHIROYUKI
J. Embryol. exp. Morph. Vol. 66, pp. 9-, 98 9 Printed in Great Britain Company of Biologists Limited 98 Mesenchymal control over elongating and branching morphogenesis in salivary gland development ByHIROYUKI
Sex Determination and Development of Reproductive Organs
 Sex Determination and Development of Reproductive Organs Sex determination The SRY + gene is necessary and probably sufficient for testis development The earliest sexual difference appears in the gonad
Sex Determination and Development of Reproductive Organs Sex determination The SRY + gene is necessary and probably sufficient for testis development The earliest sexual difference appears in the gonad
Let s Talk About Hormones!
 Let s Talk About Hormones! This lesson was created by Serena Reves and Nichelle Penney, with materials from the BCTF and The Pride Education Network. Hormones are responsible for the regulation of many
Let s Talk About Hormones! This lesson was created by Serena Reves and Nichelle Penney, with materials from the BCTF and The Pride Education Network. Hormones are responsible for the regulation of many
10. Development of genital system. Gonads. Genital ducts. External genitalia.
 10. Development of genital system. Gonads. Genital ducts. External genitalia. Gonads, genital ducts and the external genital organs initially pass through an indifferent period of development, which is
10. Development of genital system. Gonads. Genital ducts. External genitalia. Gonads, genital ducts and the external genital organs initially pass through an indifferent period of development, which is
Influence of a temporary embryonic testis graft on the regression of Miillerian ducts in female chick embryo
 J. Embryol exp. Morph. Vol. 67, pp. 81-87, 1982 g \ Printed in Great Britain Company of Biologists Limited 1982 Influence of a temporary embryonic testis graft on the regression of Miillerian ducts in
J. Embryol exp. Morph. Vol. 67, pp. 81-87, 1982 g \ Printed in Great Britain Company of Biologists Limited 1982 Influence of a temporary embryonic testis graft on the regression of Miillerian ducts in
DEVELOPMENT OF THE ADRENAL GLAND; TESTES AND MESONEPHRIC DUCT. Reading Assignment: The Developing Human, Clinically Oriented Embryology pp
 Developmental Anatomy Steven M. Hill, Ph.D. 9/16/13 DEVELOPMENT OF THE ADRENAL GLAND; TESTES AND MESONEPHRIC DUCT Reading Assignment: The Developing Human, Clinically Oriented Embryology pp. 264-273. Objectives:
Developmental Anatomy Steven M. Hill, Ph.D. 9/16/13 DEVELOPMENT OF THE ADRENAL GLAND; TESTES AND MESONEPHRIC DUCT Reading Assignment: The Developing Human, Clinically Oriented Embryology pp. 264-273. Objectives:
IN SUMMARY HST 071 NORMAL & ABNORMAL SEXUAL DIFFERENTIATION Fetal Sex Differentiation Postnatal Diagnosis and Management of Intersex Abnormalities
 Harvard-MIT Division of Health Sciences and Technology HST.071: Human Reproductive Biology Course Director: Professor Henry Klapholz IN SUMMARY HST 071 Title: Fetal Sex Differentiation Postnatal Diagnosis
Harvard-MIT Division of Health Sciences and Technology HST.071: Human Reproductive Biology Course Director: Professor Henry Klapholz IN SUMMARY HST 071 Title: Fetal Sex Differentiation Postnatal Diagnosis
Chapter 7 DEVELOPMENT AND SEX DETERMINATION
 Chapter 7 DEVELOPMENT AND SEX DETERMINATION Chapter Summary The male and female reproductive systems produce the sperm and eggs, and promote their meeting and fusion, which results in a fertilized egg.
Chapter 7 DEVELOPMENT AND SEX DETERMINATION Chapter Summary The male and female reproductive systems produce the sperm and eggs, and promote their meeting and fusion, which results in a fertilized egg.
Sexual differentiation is sequential process:
 Genital lsystem J. H. Lue Sexual differentiation is sequential process:.genetic (chromosomal) sex -- determined at fertilization.gonad sex -- is differentiated after 7th week.phenotypic sex -- under normal
Genital lsystem J. H. Lue Sexual differentiation is sequential process:.genetic (chromosomal) sex -- determined at fertilization.gonad sex -- is differentiated after 7th week.phenotypic sex -- under normal
612.6I7.5:612.6I6.I. different, but most of them appear to be satisfactory from a qualitative
 442 612.6I7.5:612.6I6.I SIZE CHANGES IN THE SEMINAL VESICLES OF THE MOUSE DURING DEVELOPMENT AND AFTER CASTRATION. BY RUTH DEANESLY AND A. S. PARKES'. (From the National Institute for Medical Research,
442 612.6I7.5:612.6I6.I SIZE CHANGES IN THE SEMINAL VESICLES OF THE MOUSE DURING DEVELOPMENT AND AFTER CASTRATION. BY RUTH DEANESLY AND A. S. PARKES'. (From the National Institute for Medical Research,
Animal Reproduction. Hypothalamic-pituitary-gonadal axis. # lectures for cumulative test # 01 book 01
 Animal Reproduction JP Advis DVM, Ph.D. Bartlett Hall, Animal Sciences, Cook, (732) 932-9240, advis@aesop.rutgers.edu 05 Course website: rci.rutgers.edu/~advis Material to be covered: About lecture Meetings
Animal Reproduction JP Advis DVM, Ph.D. Bartlett Hall, Animal Sciences, Cook, (732) 932-9240, advis@aesop.rutgers.edu 05 Course website: rci.rutgers.edu/~advis Material to be covered: About lecture Meetings
Experimental Neoplastic Formation in Embryonic Chick Brains
 Experimental Neoplastic Formation in Embryonic Chick Brains by BENGT KALLEN 1 From the Tornblad Institute of Comparative Embryology, Lund WITH TWO PLATES IN mammalian teratology, a malformation consisting
Experimental Neoplastic Formation in Embryonic Chick Brains by BENGT KALLEN 1 From the Tornblad Institute of Comparative Embryology, Lund WITH TWO PLATES IN mammalian teratology, a malformation consisting
SISTEMA REPRODUCTOR (LA IDEA FIJA) Copyright 2004 Pearson Education, Inc., publishing as Benjamin Cummings
 SISTEMA REPRODUCTOR (LA IDEA FIJA) How male and female reproductive systems differentiate The reproductive organs and how they work How gametes are produced and fertilized Pregnancy, stages of development,
SISTEMA REPRODUCTOR (LA IDEA FIJA) How male and female reproductive systems differentiate The reproductive organs and how they work How gametes are produced and fertilized Pregnancy, stages of development,
- production of two types of gametes -- fused at fertilization to form zygote
 Male reproductive system I. Sexual reproduction -- overview - production of two types of gametes -- fused at fertilization to form zygote - promotes genetic variety among members of a species -- each offspring
Male reproductive system I. Sexual reproduction -- overview - production of two types of gametes -- fused at fertilization to form zygote - promotes genetic variety among members of a species -- each offspring
REPRODUCCIÓN. La idea fija. Copyright 2004 Pearson Education, Inc., publishing as Benjamin Cummings
 REPRODUCCIÓN La idea fija How male and female reproductive systems differentiate The reproductive organs and how they work How gametes are produced and fertilized Pregnancy, stages of development, birth
REPRODUCCIÓN La idea fija How male and female reproductive systems differentiate The reproductive organs and how they work How gametes are produced and fertilized Pregnancy, stages of development, birth
Sexual Differentiation Fall 2008 Bios 90
 Sexual Differentiation Fall 2008 Bios 90 Jennifer Swann, Professor Dept Biol Sci, Lehigh University Asexual reproduction Budding : offspring develop as a growth on the body of the parent jellyfishes, echinoderms,
Sexual Differentiation Fall 2008 Bios 90 Jennifer Swann, Professor Dept Biol Sci, Lehigh University Asexual reproduction Budding : offspring develop as a growth on the body of the parent jellyfishes, echinoderms,
Urinary System. ectoderm. notochord. mesonephric tubules. Nephrogenic Cord (left)
 Urinary System NOTE: Urine proion requires an increased capillary surface area (glomeruli), epithelial s to collect plasma filtrate and extract desirable constituents, and a system to convey urine away
Urinary System NOTE: Urine proion requires an increased capillary surface area (glomeruli), epithelial s to collect plasma filtrate and extract desirable constituents, and a system to convey urine away
Bios 90/95. Jennifer Swann, PhD
 Sexual Differentiation Fall 2007 Bios 90/95 Jennifer Swann, PhD Dept Biol Sci, Lehigh University Why have sexes? What determines sex? Environment Genetics Hormones What causes these differences? The true
Sexual Differentiation Fall 2007 Bios 90/95 Jennifer Swann, PhD Dept Biol Sci, Lehigh University Why have sexes? What determines sex? Environment Genetics Hormones What causes these differences? The true
To General Embryology Dr: Azza Zaki
 Introduction To General Embryology The Human Development is a continuous process that begins when an ovum from a female is fertilized by a sperm from a male. Cell division, growth and differentiation transform
Introduction To General Embryology The Human Development is a continuous process that begins when an ovum from a female is fertilized by a sperm from a male. Cell division, growth and differentiation transform
Development of the Urinary System
 Development of the Urinary System Lecture Objectives Understand the development of the kidney and related organs of the urinary system. Define the pronephrons, mesonephrons and metanephrons. Understand
Development of the Urinary System Lecture Objectives Understand the development of the kidney and related organs of the urinary system. Define the pronephrons, mesonephrons and metanephrons. Understand
Urinary system development. Male ( ) and Female ( ) Reproductive Systems Development
 Urinary system development Male ( ) and Female ( ) Reproductive Systems Development Urogenital system develops from mesodermal uro-genital ridge (intermediate mesoderm) development of male and female genital
Urinary system development Male ( ) and Female ( ) Reproductive Systems Development Urogenital system develops from mesodermal uro-genital ridge (intermediate mesoderm) development of male and female genital
Urogenital System. PUMC Dept. of Anat. Hist. & Embry. 钱晓菁 XIAO-JING QIAN Dept. of Anatomy, Histology & Embryology Peking Union Medical College
 Urogenital System 钱晓菁 XIAO-JING QIAN Dept. of Anatomy, Histology & Embryology Peking Union Medical College intermediate mesoderm urogenital ridge mesonephric ridge genital ridge I. Urinary System 1. kidney
Urogenital System 钱晓菁 XIAO-JING QIAN Dept. of Anatomy, Histology & Embryology Peking Union Medical College intermediate mesoderm urogenital ridge mesonephric ridge genital ridge I. Urinary System 1. kidney
Tissues. Tissues - Overview. Bio 101 Laboratory 3. Epithelial Tissues and Integument
 Bio 101 Laboratory 3 Epithelial Tissues and Integument 1 Tissues Tissues to be examined under the microscope Epithelial Tissue Integument Connective Tissue **We will be doing muscle and nervous tissues
Bio 101 Laboratory 3 Epithelial Tissues and Integument 1 Tissues Tissues to be examined under the microscope Epithelial Tissue Integument Connective Tissue **We will be doing muscle and nervous tissues
Embryology /organogenesis/ Week 4 Development and teratology of reproductive system.
 Embryology /organogenesis/ Week 4 Development and teratology of reproductive system. Male or female sex is determined by spermatozoon Y in the moment of fertilization SRY gene, on the short arm of the
Embryology /organogenesis/ Week 4 Development and teratology of reproductive system. Male or female sex is determined by spermatozoon Y in the moment of fertilization SRY gene, on the short arm of the
Chapter 16: Steroid Hormones (Lecture 17)
 Chapter 16: Steroid Hormones (Lecture 17) A) 21 or fewer carbon atoms B) Precursor: 27 carbon cholesterol C) major classes of steroid hormones 1) progestagens a) progesterone- prepares lining of uterus
Chapter 16: Steroid Hormones (Lecture 17) A) 21 or fewer carbon atoms B) Precursor: 27 carbon cholesterol C) major classes of steroid hormones 1) progestagens a) progesterone- prepares lining of uterus
Human Anatomy Unit 3 REPRODUCTIVE SYSTEM
 Human Anatomy Unit 3 REPRODUCTIVE SYSTEM In Anatomy Today Male Reproductive System Gonads = testes primary organ responsible for sperm production development/maintenan ce of secondary sex characteristics
Human Anatomy Unit 3 REPRODUCTIVE SYSTEM In Anatomy Today Male Reproductive System Gonads = testes primary organ responsible for sperm production development/maintenan ce of secondary sex characteristics
Remember from the first year embryology Trilaminar disc has 3 layers: ectoderm, mesoderm, and endoderm
 Development of face Remember from the first year embryology Trilaminar disc has 3 layers: ectoderm, mesoderm, and endoderm The ectoderm forms the neural groove, then tube The neural tube lies in the mesoderm
Development of face Remember from the first year embryology Trilaminar disc has 3 layers: ectoderm, mesoderm, and endoderm The ectoderm forms the neural groove, then tube The neural tube lies in the mesoderm
Development of teeth. 5.DM - Pedo
 Development of teeth 5.DM - Pedo Tooth development process of continuous changes in predetermined order starts from dental lamina A band of ectodermal cells growing from the epithelium of the embryonic
Development of teeth 5.DM - Pedo Tooth development process of continuous changes in predetermined order starts from dental lamina A band of ectodermal cells growing from the epithelium of the embryonic
Obstetrics Content Outline Obstetrics - Fetal Abnormalities
 Obstetrics Content Outline Obstetrics - Fetal Abnormalities Effective February 2007 10 16% renal agenesis complete absence of the kidneys occurs when ureteric buds fail to develop Or degenerate before
Obstetrics Content Outline Obstetrics - Fetal Abnormalities Effective February 2007 10 16% renal agenesis complete absence of the kidneys occurs when ureteric buds fail to develop Or degenerate before
DEFINITION: Masculinization of external genitalia in patients with normal 46XX karyotype.
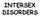 INTERSEX DISORDERS DEFINITION: Masculinization of external genitalia in patients with normal 46XX karyotype. - Degree of masculinization variable: - mild clitoromegaly - complete fusion of labia folds
INTERSEX DISORDERS DEFINITION: Masculinization of external genitalia in patients with normal 46XX karyotype. - Degree of masculinization variable: - mild clitoromegaly - complete fusion of labia folds
Midgut. Over its entire length the midgut is supplied by the superior mesenteric artery
 Gi Embryology 3 Midgut the midgut is suspended from the dorsal abdominal wall by a short mesentery and communicates with the yolk sac by way of the vitelline duct or yolk stalk Over its entire length the
Gi Embryology 3 Midgut the midgut is suspended from the dorsal abdominal wall by a short mesentery and communicates with the yolk sac by way of the vitelline duct or yolk stalk Over its entire length the
CHARA CTERS* nearly sterile. As in other similar combinations the sex glands are reduced ZOOLOGY: E. WITSCHI
 ZOOLOGY: E. WITSCHI VOL. 23, 1937 35 STIMULATIVE AND INHIBITIVE INDUCTION IN THE DEVELOPMENT OF PRIMARY AND SECONDARY SEX CHARA CTERS* By EMIL WITSCHI DEPARTMENT OF ZOOLOGY, STATE UNIVERSITY OF IOWA Read
ZOOLOGY: E. WITSCHI VOL. 23, 1937 35 STIMULATIVE AND INHIBITIVE INDUCTION IN THE DEVELOPMENT OF PRIMARY AND SECONDARY SEX CHARA CTERS* By EMIL WITSCHI DEPARTMENT OF ZOOLOGY, STATE UNIVERSITY OF IOWA Read
1. Both asexual and sexual reproduction occur in the animal kingdom
 1. Both asexual and sexual reproduction occur in the animal kingdom Asexual reproduction involves the formation of individuals whose genes all come from one parent. There is no fusion of sperm and egg.
1. Both asexual and sexual reproduction occur in the animal kingdom Asexual reproduction involves the formation of individuals whose genes all come from one parent. There is no fusion of sperm and egg.
SUMMARY. Keywords: quail, Coturnix japonica, morphology, ovary, oviduct, neurotrophins, immunohistochemistry
 SUMMARY Keywords: quail, Coturnix japonica, morphology, ovary, oviduct, neurotrophins, immunohistochemistry Studies on the development of biological systems have expanded using animal models, always to
SUMMARY Keywords: quail, Coturnix japonica, morphology, ovary, oviduct, neurotrophins, immunohistochemistry Studies on the development of biological systems have expanded using animal models, always to
Class VIII Chapter 10 Reaching the Age of Adolescence Science
 Question 1: What is the term used for secretions of endocrine glands responsible for changes taking place in the body? Hormones are chemical substances which are secreted by endocrine glands. They are
Question 1: What is the term used for secretions of endocrine glands responsible for changes taking place in the body? Hormones are chemical substances which are secreted by endocrine glands. They are
Animal Science 434 Reproductive Physiology"
 Animal Science 434 Reproductive Physiology" Embryogenesis of the Pituitary and Sexual Development: Part A Development of the Pituitary Gland" Infundibulum" Brain" Rathke s Pouch" Stomodeum" Germ Cell Migration"
Animal Science 434 Reproductive Physiology" Embryogenesis of the Pituitary and Sexual Development: Part A Development of the Pituitary Gland" Infundibulum" Brain" Rathke s Pouch" Stomodeum" Germ Cell Migration"
Mohammad Sha ban. Basheq Jehad. Hamzah Nakhleh
 11 Mohammad Sha ban Basheq Jehad Hamzah Nakhleh Physiology of the reproductive system In physiology, we are concerned with the mechanisms in which the system functions, and how the system responds to different
11 Mohammad Sha ban Basheq Jehad Hamzah Nakhleh Physiology of the reproductive system In physiology, we are concerned with the mechanisms in which the system functions, and how the system responds to different
Copyright 2003 Pearson Education, Inc. publishing as Benjamin Cummings. Dr. Nabil Khouri
 Dr. Nabil Khouri Objectives: General objectives: - to identify the kidney s structures, function and location - to analyze the relationship between microscopic structure and function Specific objectives:
Dr. Nabil Khouri Objectives: General objectives: - to identify the kidney s structures, function and location - to analyze the relationship between microscopic structure and function Specific objectives:
Development of the female Reproductive System. Dr. Susheela Rani
 Development of the female Reproductive System Dr. Susheela Rani Genital System Gonads Internal genitals External genitals Determining sex chronology of events Genetic sex Determined at fertilization Gonadal
Development of the female Reproductive System Dr. Susheela Rani Genital System Gonads Internal genitals External genitals Determining sex chronology of events Genetic sex Determined at fertilization Gonadal
FIGURE 54 represents the urogenital apparatus
 CHAPTER VI THE UROGENITAL ORGANS FIGURE 54 represents the urogenital apparatus of a thirty-inch female specimen of Alligator mississippiensis. Figure 55 shows the corresponding organs of a male A lucius;
CHAPTER VI THE UROGENITAL ORGANS FIGURE 54 represents the urogenital apparatus of a thirty-inch female specimen of Alligator mississippiensis. Figure 55 shows the corresponding organs of a male A lucius;
FLASH CARDS. Kalat s Book Chapter 11 Alphabetical
 FLASH CARDS www.biologicalpsych.com Kalat s Book Chapter 11 Alphabetical alpha-fetoprotein alpha-fetoprotein Alpha-Fetal Protein (AFP) or alpha-1- fetoprotein. During a prenatal sensitive period, estradiol
FLASH CARDS www.biologicalpsych.com Kalat s Book Chapter 11 Alphabetical alpha-fetoprotein alpha-fetoprotein Alpha-Fetal Protein (AFP) or alpha-1- fetoprotein. During a prenatal sensitive period, estradiol
STRUCTURE AND FUNCTION OF THE FEMALE REPRODUCTIVE SYSTEM
 Unit 7B STRUCTURE AND FUNCTION OF THE FEMALE REPRODUCTIVE SYSTEM LEARNING OBJECTIVES 1. Learn the structures of the female reproductive tract. 2. Learn the functions of the female reproductive tract. 3.
Unit 7B STRUCTURE AND FUNCTION OF THE FEMALE REPRODUCTIVE SYSTEM LEARNING OBJECTIVES 1. Learn the structures of the female reproductive tract. 2. Learn the functions of the female reproductive tract. 3.
18 Urinary system. 19 Male reproductive system. Female reproductive system. Blok 11: Genital and Urinary Tract Diseases
 Blok 11: Genital and Urinary Tract Diseases 18 Urinary System 19 Male Genital System 20 Female Genital System 18 Urinary system You should be able to: 1. Describe the structures and associated functions
Blok 11: Genital and Urinary Tract Diseases 18 Urinary System 19 Male Genital System 20 Female Genital System 18 Urinary system You should be able to: 1. Describe the structures and associated functions
Biology of Reproduction-Biol 326
 Biology of Reproduction-Biol 326 READ ALL INSTRUCTIONS CAREFULLY. ANSWER ALL THE QUESTIONS ON THE ANSWER SHEET. THE ANSWER ON THE ANSWER SHEET IS YOUR OFFICIAL ANSWER REGARDLESS OF WHAT YOU MARK ON THE
Biology of Reproduction-Biol 326 READ ALL INSTRUCTIONS CAREFULLY. ANSWER ALL THE QUESTIONS ON THE ANSWER SHEET. THE ANSWER ON THE ANSWER SHEET IS YOUR OFFICIAL ANSWER REGARDLESS OF WHAT YOU MARK ON THE
Outline. Male Reproductive System Testes and Sperm Hormonal Regulation
 Outline Male Reproductive System Testes and Sperm Hormonal Regulation Female Reproductive System Genital Tract Hormonal Levels Uterine Cycle Fertilization and Pregnancy Control of Reproduction Infertility
Outline Male Reproductive System Testes and Sperm Hormonal Regulation Female Reproductive System Genital Tract Hormonal Levels Uterine Cycle Fertilization and Pregnancy Control of Reproduction Infertility
Bio Section IV Late Development. Development of the Tetrapod Limb. Pattern Formation. Skeletal pattern formation. Gilbert 9e Chapter 13
 Bio 127 - Section IV Late Development Development of the Tetrapod Limb Gilbert 9e Chapter 13 Pattern Formation Limbs show an amazing aspect of development Similarities: Architectural symmetry in all four
Bio 127 - Section IV Late Development Development of the Tetrapod Limb Gilbert 9e Chapter 13 Pattern Formation Limbs show an amazing aspect of development Similarities: Architectural symmetry in all four
Biology 224 Human Anatomy and Physiology II Week 9; Lecture 2; Wednesday Stuart Sumida. Development and Structure, of the Reproductive System
 Biology 224 Human Anatomy and Physiology II Week 9; Lecture 2; Wednesday Stuart Sumida Development and Structure, of the Reproductive System Don t forget the relationships of the structures of the layers
Biology 224 Human Anatomy and Physiology II Week 9; Lecture 2; Wednesday Stuart Sumida Development and Structure, of the Reproductive System Don t forget the relationships of the structures of the layers
PERINEAL HYPOSPADIAS IN A CROSS BREED DOG: A CASE REPORT
 Indo-Am. J. Agric. & Vet. Sci., 2014 M Gokulakrishnan ISSN 2321 9602 and L Nagarajan, www.iajavs.com 2014 Vol. 2, No. 3, September 2014 2014 Meghana Publications. All Rights Reserved Case Report PERINEAL
Indo-Am. J. Agric. & Vet. Sci., 2014 M Gokulakrishnan ISSN 2321 9602 and L Nagarajan, www.iajavs.com 2014 Vol. 2, No. 3, September 2014 2014 Meghana Publications. All Rights Reserved Case Report PERINEAL
Morphogenesis by dissociated immature rat testicular cells in primary culture
 /. Einhryol. exp. Morph. Vol. 44, pp. 297-302, 1978 297 Printed in Great Britain (p Company of Biologists Limited 1978 SHORT PAPER Morphogenesis by dissociated immature rat testicular cells in primary
/. Einhryol. exp. Morph. Vol. 44, pp. 297-302, 1978 297 Printed in Great Britain (p Company of Biologists Limited 1978 SHORT PAPER Morphogenesis by dissociated immature rat testicular cells in primary
Testes (male gonads) -Produce sperm -Produce sex hormones -Found in a sac called the scrotum -Suspended outside of the body cavity for temperature
 REPRODUCTION Testes (male gonads) -Produce sperm -Produce sex hormones -Found in a sac called the scrotum -Suspended outside of the body cavity for temperature reduction -Testes wall made of fibrous connective
REPRODUCTION Testes (male gonads) -Produce sperm -Produce sex hormones -Found in a sac called the scrotum -Suspended outside of the body cavity for temperature reduction -Testes wall made of fibrous connective
Biology of Reproduction- Zool 346 Exam 2
 Biology of Reproduction- Zool 346 Exam 2 ANSWER ALL THE QUESTIONS ON THE ANSWER SHEET. THE ANSWER ON THE ANSWER SHEET IS YOUR OFFICIAL ANSWER. Some critical words are boldfaced. This exam is 7 pages long.
Biology of Reproduction- Zool 346 Exam 2 ANSWER ALL THE QUESTIONS ON THE ANSWER SHEET. THE ANSWER ON THE ANSWER SHEET IS YOUR OFFICIAL ANSWER. Some critical words are boldfaced. This exam is 7 pages long.
Urinary System Laboratory
 Urinary System Laboratory 1 Adrenal gland Organs of The Urinary System Renal artery and vein Kidney Ureter Urinary bladder Figure 26.1 2 Urethra Functions of the urinary system organs: Urethra expels urine
Urinary System Laboratory 1 Adrenal gland Organs of The Urinary System Renal artery and vein Kidney Ureter Urinary bladder Figure 26.1 2 Urethra Functions of the urinary system organs: Urethra expels urine
Efferent Ducts and Epididymis
 increase) the secretion of each of the androgen regulated proteins. Regulation of spermatogenesis is therefore an extremely complex cascade of cell-cell interactions with the Leydig cells supporting germ
increase) the secretion of each of the androgen regulated proteins. Regulation of spermatogenesis is therefore an extremely complex cascade of cell-cell interactions with the Leydig cells supporting germ
Histology Urinary system
 Histology Urinary system Urinary system Composed of two kidneys, two ureters, the urinary bladder, and the urethra, the urinary system plays a critical role in: 1- Blood filtration,(filtration of cellular
Histology Urinary system Urinary system Composed of two kidneys, two ureters, the urinary bladder, and the urethra, the urinary system plays a critical role in: 1- Blood filtration,(filtration of cellular
LABORATORY EXERCISES FOR MALE REPRODUCTIVE SYSTEM
 LABORATORY EXERCISES FOR MALE REPRODUCTIVE SYSTEM Slide #101 (1096). Testis, rat. sustentacular ( Sertoli ) cells Nuclei of Sustentacular cells Leydig cells Spermatogonia Spermatocytes Spermatids pale
LABORATORY EXERCISES FOR MALE REPRODUCTIVE SYSTEM Slide #101 (1096). Testis, rat. sustentacular ( Sertoli ) cells Nuclei of Sustentacular cells Leydig cells Spermatogonia Spermatocytes Spermatids pale
A classification of epithelial tissues
 A classification of epithelial tissues Ramray Bhat Molecular Reproduction Development and Genetics ramray@iisc.ac.in Textbooks for my portion Molecular Biology of the Cell (Bruce Alberts) 6 th Edition
A classification of epithelial tissues Ramray Bhat Molecular Reproduction Development and Genetics ramray@iisc.ac.in Textbooks for my portion Molecular Biology of the Cell (Bruce Alberts) 6 th Edition
Guide to Small Animal Reproductive Imaging using the Vevo 770
 Guide to Small Animal Reproductive Imaging using the Vevo 770 Course Objectives: After completion of this module, the participant will be able to accomplish the following: Recognize reproductive female
Guide to Small Animal Reproductive Imaging using the Vevo 770 Course Objectives: After completion of this module, the participant will be able to accomplish the following: Recognize reproductive female
Sex differentiation of germ cells in the teleost, Oryzias latipes, during normal embryonic development
 J. Embryol. exp. Morph. Vol. 2, 2, pp. -, 2 Printed in Great Britain Sex differentiation of germ cells in the teleost, Oryzias latipes, during normal embryonic development ByNORIYUKI SATOH AND NOBUO EGAMI
J. Embryol. exp. Morph. Vol. 2, 2, pp. -, 2 Printed in Great Britain Sex differentiation of germ cells in the teleost, Oryzias latipes, during normal embryonic development ByNORIYUKI SATOH AND NOBUO EGAMI
BIOL 2402 Reproductive Systems!
 Dr. Chris Doumen! Female Reproductive Anatomy BIOL 2402 Reproductive Systems! Establishing the Ovarian Cycle During childhood, until puberty Ovaries grow and secrete small amounts of estrogens Estrogen
Dr. Chris Doumen! Female Reproductive Anatomy BIOL 2402 Reproductive Systems! Establishing the Ovarian Cycle During childhood, until puberty Ovaries grow and secrete small amounts of estrogens Estrogen
Lecture 15 (Nov 16 th ): Hormones and Sexual Behavior Lecture Outline. 4) Gender Phenotype : Organizing Effects of Sex Hormones in Utero and Anomalies
 Lecture 15 (Nov 16 th ): Hormones and Sexual Behavior Lecture Outline 1) Organs / Glands / Hormonal Communication 2) Sex Hormones: Male vs. Female 3) Genetic Gender (XX, XY) 4) Gender Phenotype : Organizing
Lecture 15 (Nov 16 th ): Hormones and Sexual Behavior Lecture Outline 1) Organs / Glands / Hormonal Communication 2) Sex Hormones: Male vs. Female 3) Genetic Gender (XX, XY) 4) Gender Phenotype : Organizing
PHYSIOLOGY AND PATHOLOGY OF SEXUAL DIFFERENTIATION
 PHYSIOLOGY AND PATHOLOGY OF SEXUAL DIFFERENTIATION Prof. Dr med. Jolanta Słowikowska-Hilczer Department of Andrology and Reproductive Endocrinology Medical University of Łódź, Poland Sexual determination
PHYSIOLOGY AND PATHOLOGY OF SEXUAL DIFFERENTIATION Prof. Dr med. Jolanta Słowikowska-Hilczer Department of Andrology and Reproductive Endocrinology Medical University of Łódź, Poland Sexual determination
Bio 322 Human Anatomy Objectives for the laboratory exercise Female Reproductive System
 Bio 322 Human Anatomy Objectives for the laboratory exercise Female Reproductive System Required reading before beginning this lab: Saladin, KS: Human Anatomy 5 th ed (2017) Chapter 26 For this lab you
Bio 322 Human Anatomy Objectives for the laboratory exercise Female Reproductive System Required reading before beginning this lab: Saladin, KS: Human Anatomy 5 th ed (2017) Chapter 26 For this lab you
The Effect of Cortisone on Cell Proliferation and Migration in Peripheral Nerves undergoing Wallerian degeneration
 The Effect of Cortisone on Cell Proliferation and Migration in Peripheral Nerves undergoing Wallerian by G. A. THOMAS 1 From the Department of Anatomy, Guy's Hospital Medical School, London INTRODUCTION
The Effect of Cortisone on Cell Proliferation and Migration in Peripheral Nerves undergoing Wallerian by G. A. THOMAS 1 From the Department of Anatomy, Guy's Hospital Medical School, London INTRODUCTION
Effect of the T-mutation on histogenesis of the mouse embryo under the testis capsule
 /. Embryol. exp. Morph. Vol. 5, pp. -3, 79 Printed in Great Britain Company of Biologists Limited 79 Effect of the T-mutation on histogenesis of the mouse embryo under the testis capsule By H. FUJIMOTO
/. Embryol. exp. Morph. Vol. 5, pp. -3, 79 Printed in Great Britain Company of Biologists Limited 79 Effect of the T-mutation on histogenesis of the mouse embryo under the testis capsule By H. FUJIMOTO
Anatomy and Physiology Tissue Review
 Anatomy and Physiology Tissue Review OVERVIEW Histology practicals can be rough, especially when access to slides is limited to the lab period. This resource provides an opportunity to learn or review
Anatomy and Physiology Tissue Review OVERVIEW Histology practicals can be rough, especially when access to slides is limited to the lab period. This resource provides an opportunity to learn or review
Three-dimensional structure of the human small intestinal mucosa in health and disease
 Gut, 1969, 10, 6-12 Three-dimensional structure of the human small intestinal mucosa in health and disease C. A. LOEHRY AND B. CREAMER From the Gastrointestinal Laboratory, St Thomas' Hospital, London
Gut, 1969, 10, 6-12 Three-dimensional structure of the human small intestinal mucosa in health and disease C. A. LOEHRY AND B. CREAMER From the Gastrointestinal Laboratory, St Thomas' Hospital, London
Identification in rats of a programming window for reproductive tract masculinization, disruption of which leads to hypospadias and cryptorchidism
 Research article Identification in rats of a programming window for reproductive tract masculinization, disruption of which leads to hypospadias and cryptorchidism Michelle Welsh, Philippa T.K. Saunders,
Research article Identification in rats of a programming window for reproductive tract masculinization, disruption of which leads to hypospadias and cryptorchidism Michelle Welsh, Philippa T.K. Saunders,
4.05 Remember the structures of the reproductive system
 4.05 Remember the structures of the reproductive system 4.05 Remember the structures of the reproductive system Essential question What are the structures of the reproductive system? 2 Structures of the
4.05 Remember the structures of the reproductive system 4.05 Remember the structures of the reproductive system Essential question What are the structures of the reproductive system? 2 Structures of the
Urinary Anatomy. Lab 40. Kidneys. Nephrons. Renal Corpuscle
 Urinary Anatomy Lab 40. Urinary Anatomy and Kidney Dissection Kidneys: filters blood, produces urine Ureters: convey urine to bladder Bladder: holding tank Urethra: carries urine to the outside for elimination
Urinary Anatomy Lab 40. Urinary Anatomy and Kidney Dissection Kidneys: filters blood, produces urine Ureters: convey urine to bladder Bladder: holding tank Urethra: carries urine to the outside for elimination
2. Which male target tissues respond to testosterone, and which require dihydrotestosterone?
 308 PHYSIOLOGY CASES AND PROBLEMS Case 56 Male Pseudohermaphroditism: Sa-Reductase Deficiency Fourteen years ago, Wally and Wanda Garvey, who live in rural North Carolina, had their first child. The baby
308 PHYSIOLOGY CASES AND PROBLEMS Case 56 Male Pseudohermaphroditism: Sa-Reductase Deficiency Fourteen years ago, Wally and Wanda Garvey, who live in rural North Carolina, had their first child. The baby
Comparative Anatomy Urogenital System. Note Set 11 Chapter 15
 Comparative Anatomy Urogenital System Note Set 11 Chapter 15 Urogenital System Ducts of excretory and reproductive systems are intimately associated Figure 14.1: Embryonic and evolutionary development
Comparative Anatomy Urogenital System Note Set 11 Chapter 15 Urogenital System Ducts of excretory and reproductive systems are intimately associated Figure 14.1: Embryonic and evolutionary development
1. Be able to characterize the menstrual cycle from the perspective of the ovary a. Follicular phase b. Luteal phase
 Human Sexuality Exam II Review Material Gametogenesis: Oogenesis 1. Be able to characterize the menstrual cycle from the perspective of the ovary a. Follicular phase b. Luteal phase 2. Know the relative
Human Sexuality Exam II Review Material Gametogenesis: Oogenesis 1. Be able to characterize the menstrual cycle from the perspective of the ovary a. Follicular phase b. Luteal phase 2. Know the relative
Department of Plastic Surgery, University Hospital, Groningen, The Netherlands
 SURGICAL CORRECTION OF FEMALE PSEUDOHERMA- PHRODITISM DUE TO ADRENAL HYPERPLASIA By A. J. C. HUFFSTADT, M.D. Department of Plastic Surgery, University Hospital, Groningen, The Netherlands SINCE the work
SURGICAL CORRECTION OF FEMALE PSEUDOHERMA- PHRODITISM DUE TO ADRENAL HYPERPLASIA By A. J. C. HUFFSTADT, M.D. Department of Plastic Surgery, University Hospital, Groningen, The Netherlands SINCE the work
H I S T O L O G Y O F T H E U R I N A R Y S Y S T E M
 SCPA 602- Anatomical Basis For Pathological Study H I S T O L O G Y O F T H E U R I N A R Y S Y S T E M S O M P H O N G N A R K P I N I T, M. D. D E P A R T M E N T O F P A T H O B I O L O G Y F A C U
SCPA 602- Anatomical Basis For Pathological Study H I S T O L O G Y O F T H E U R I N A R Y S Y S T E M S O M P H O N G N A R K P I N I T, M. D. D E P A R T M E N T O F P A T H O B I O L O G Y F A C U
Formation of Urine: Formation of Urine
 The Urinary outflow tract: monitors and regulates extra-cellular fluids excretes harmful substances in urine, including nitrogenous wastes (urea) returns useful substances to bloodstream maintain balance
The Urinary outflow tract: monitors and regulates extra-cellular fluids excretes harmful substances in urine, including nitrogenous wastes (urea) returns useful substances to bloodstream maintain balance
Chapter 14 The Reproductive System
 Biology 12 Name: Reproductive System Per: Date: Chapter 14 The Reproductive System Complete using BC Biology 12, page 436-467 14. 1 Male Reproductive System pages 440-443 1. Distinguish between gametes
Biology 12 Name: Reproductive System Per: Date: Chapter 14 The Reproductive System Complete using BC Biology 12, page 436-467 14. 1 Male Reproductive System pages 440-443 1. Distinguish between gametes
Urinary System. J. H. Lue. intermediate mesoderm cloaca coelomic epithelium
 Urinary System J. H. Lue Primordium: intermediate mesoderm cloaca coelomic epithelium 1 3w(18d) 3 w 4w (24d) 4w (26d) 2 Intermediate mesoderm 3 Intermediate mesoderm intermediate mesoderm urogenital ridge
Urinary System J. H. Lue Primordium: intermediate mesoderm cloaca coelomic epithelium 1 3w(18d) 3 w 4w (24d) 4w (26d) 2 Intermediate mesoderm 3 Intermediate mesoderm intermediate mesoderm urogenital ridge
PELVIS II: FUNCTION TABOOS (THE VISCERA) Defecation Urination Ejaculation Conception
 PELVIS II: FUNCTION TABOOS (THE VISCERA) Defecation Urination Ejaculation Conception REVIEW OF PELVIS I Pelvic brim, inlet Pelvic outlet True pelvis-- --viscera Tilt forward Mid-sagital views-- --how the
PELVIS II: FUNCTION TABOOS (THE VISCERA) Defecation Urination Ejaculation Conception REVIEW OF PELVIS I Pelvic brim, inlet Pelvic outlet True pelvis-- --viscera Tilt forward Mid-sagital views-- --how the
Male Reproductive System
 Male Reproductive System organs that function in: gamete and hormone production not all in abdominal cavity paired testicles = controlled by LH & FSH duct systems accessory glands Testis: Gross Histology
Male Reproductive System organs that function in: gamete and hormone production not all in abdominal cavity paired testicles = controlled by LH & FSH duct systems accessory glands Testis: Gross Histology
Male Reproductive System
 Male Reproductive System Constitution of male reproductive system Genital gland ----testis Genital ducts epididymis / ductus deferens / urinary duct Accessory sex glands Penis prostate gland Seminal vesicle
Male Reproductive System Constitution of male reproductive system Genital gland ----testis Genital ducts epididymis / ductus deferens / urinary duct Accessory sex glands Penis prostate gland Seminal vesicle
Male Anatomy. testes, genetically determined in mammals - testis releases hormones that then control the development of secondary sex characteristics
 Male Anatomy Male Anatomy Primary Organ testes, genetically determined in mammals - testis releases hormones that then control the development of secondary sex characteristics 1) Secondary Organs internal
Male Anatomy Male Anatomy Primary Organ testes, genetically determined in mammals - testis releases hormones that then control the development of secondary sex characteristics 1) Secondary Organs internal
Development of the Urinary System. 3 Distinct Embryonic Kidney Structures
 Development of the Urinary System Excretory portion of urinary system derived from intermediate mesoderm Week 4: 1 st nephrons/renal corpuscles form Nephrotomes form and develop hollow lumens to form nephric
Development of the Urinary System Excretory portion of urinary system derived from intermediate mesoderm Week 4: 1 st nephrons/renal corpuscles form Nephrotomes form and develop hollow lumens to form nephric
THE sebaceous glands of the rabbit consist of clusters of about ten cells
 79 On the Relationship between Mammary, Sweat, and Sebaceous Glands By D. B. CARLISLE (From the Department of Zoology and Comparative Anatomy, Oxford, and the Plymouth Laboratory of the Marine Biological
79 On the Relationship between Mammary, Sweat, and Sebaceous Glands By D. B. CARLISLE (From the Department of Zoology and Comparative Anatomy, Oxford, and the Plymouth Laboratory of the Marine Biological
.Protein LYONIZATION. The process by which all X chromosomes in excess of one are made genetically inactive and heterochromatic.
 + Electrical field - LYONIZATION Colleen Jackson-Cook, Ph.D, FACMG Sanger Hall, Room 5-7 ccook@mcvh-vcu.edu The process by which all X chromosomes in excess of one are made genetically inactive and heterochromatic.
+ Electrical field - LYONIZATION Colleen Jackson-Cook, Ph.D, FACMG Sanger Hall, Room 5-7 ccook@mcvh-vcu.edu The process by which all X chromosomes in excess of one are made genetically inactive and heterochromatic.
androgen on the seminal vesicles it had neither a blocking effect on the penile
 MORPHOLOGICAL AND BEHAVIOURAL EFFECTS OF AN 'ANTIANDROGEN' IN MALE RATS F. A. BEACH and W. H. WESTBROOK Department of Psychology, University of California, Berkeley, California 94720, U.S.A. (Received
MORPHOLOGICAL AND BEHAVIOURAL EFFECTS OF AN 'ANTIANDROGEN' IN MALE RATS F. A. BEACH and W. H. WESTBROOK Department of Psychology, University of California, Berkeley, California 94720, U.S.A. (Received
Animal Development. Lecture 3. Germ Cells and Sex
 Animal Development Lecture 3 Germ Cells and Sex 1 The ovary of sow. The ovary of mare. The ovary of cow. The ovary of ewe. 2 3 The ovary. A generalized vertebrate ovary. (Wilt and Hake, Ch 2, 2004) 4 The
Animal Development Lecture 3 Germ Cells and Sex 1 The ovary of sow. The ovary of mare. The ovary of cow. The ovary of ewe. 2 3 The ovary. A generalized vertebrate ovary. (Wilt and Hake, Ch 2, 2004) 4 The
Proximal distal sequence of development of the skeletal tissues in the penis of rat and the inductive effect of epithelium
 J. Embryol. exp. Morph. 92, 133-143 (1986) 133 Printed in Great Britain The Company of Biologists Limited 1986 Proximal distal sequence of development of the skeletal tissues in the penis of rat and the
J. Embryol. exp. Morph. 92, 133-143 (1986) 133 Printed in Great Britain The Company of Biologists Limited 1986 Proximal distal sequence of development of the skeletal tissues in the penis of rat and the
Testicular feminization. The features of this syndrome in its typical form are these (Fig. 1): (1) Female bodily configuration.
 Personal Practice Archives of Disease in Childhood, 1970, 45, 595. The XY Female Child C. J. DEWHURST From the Institute of Obstetrics and Gynaecology, Queen Charlotte's Maternity Hospital, and Chelsea
Personal Practice Archives of Disease in Childhood, 1970, 45, 595. The XY Female Child C. J. DEWHURST From the Institute of Obstetrics and Gynaecology, Queen Charlotte's Maternity Hospital, and Chelsea
