HDAC9 Knockout Mice Are Protected From Adipose Tissue Dysfunction and Systemic Metabolic Disease During High- Fat Feeding
|
|
|
- Augusta Wheeler
- 5 years ago
- Views:
Transcription
1 176 Diabetes Volume 63, January 2014 Tapan K. Chatterjee, 1 Joshua E. Basford, 2 Ellen Knoll, 1 Wilson S. Tong, 1 Victor Blanco, 3 Andra L. Blomkalns, 3 Steven Rudich, 4 Alex B. Lentsch, 4 David Y. Hui, 2 and Neal L. Weintraub 1 HDAC9 Knockout Mice Are Protected From Adipose Tissue Dysfunction and Systemic Metabolic Disease During High- Fat Feeding OBESITY STUDIES During chronic caloric excess, adipose tissue expands primarily by enlargement of individual adipocytes, which become stressed with lipid overloading, thereby contributing to obesity-related disease. Although adipose tissue contains numerous preadipocytes, differentiation into functionally competent adipocytes is insufficient to accommodate the chronic caloric excess and prevent adipocyte overloading. We report for the firsttimethatachronichigh-fatdiet(hfd) impairs adipogenic differentiation, leading to accumulation of inefficiently differentiated adipocytes with blunted expression of adipogenic differentiationspecific genes. Preadipocytes from these mice likewise exhibit impaired adipogenic differentiation, and this phenotype persists during in vitro cell culture. HFD-induced impaired adipogenic differentiation is associated with elevated expression of histone deacetylase 9 (HDAC9), an endogenous negative regulator of adipogenic differentiation. Genetic ablation of HDAC9 improves adipogenic differentiation and systemic metabolic state during an HFD, resulting in diminished weight gain, improved glucose tolerance and insulin sensitivity, and reduced hepatosteatosis. Moreover, compared with wild-type mice, HDAC9 knockout mice exhibit upregulated expression of beige adipocyte marker genes, particularly during an HFD, in association with increased energy expenditure and adaptive thermogenesis. These results suggest that targeting HDAC9 may be an effective strategy for combating obesity-related metabolic disease. Diabetes 2014;63: DOI: /db Adipose tissue plays a vital homeostatic role in storing excess calories as triglyceride lipid within adipocytes. Adipocytes also function as endocrine cells, secreting hormones and biologically active molecules that regulate cellular lipid storage capacity, tissue and systemic insulin sensitivity, and metabolic energy balance. During chronic caloric excess, adipose tissue expands primarily by enlargement of individual adipocytes. Over time, these enlarged adipocytes become mechanically stressed with lipid overloading and fail to exhibit proper endocrine function. This contributes to adipose tissue inflammation, ectopic lipid accumulation, decreased 1 Department of Internal Medicine, Division of Cardiovascular Diseases, University of Cincinnati, Cincinnati, OH 2 Department of Pathology, Metabolic Diseases Institute, University of Cincinnati, Cincinnati, OH 3 Department of Emergency Medicine, University of Cincinnati, Cincinnati, OH 4 Department of Surgery, University of Cincinnati, Cincinnati, OH Corresponding author: Tapan K. Chatterjee, tchatterjee@gru.edu. Received 24 July 2013 and accepted 30 September This article contains Supplementary Data online at T.K.C. and N.L.W. are currently affiliated with the Department of Internal Medicine, Vascular Biology Center, Georgia Regents University, Augusta, GA by the American Diabetes Association. See for details.
2 diabetes.diabetesjournals.org Chatterjee and Associates 177 circulating adiponectin levels, glucose intolerance, and insulin resistance. Adipose tissues contain a huge reserve of precursor cells adipose-derived stem cells and committed preadipocytes representing approximately 20 40% of the adipose tissue cellularity (1). It is estimated that daily turnover of adipocytes could be as high as 1 5% under resting conditions, and obesity markedly stimulates preadipocyte replication, particularly in subcutaneous adipose tissues (2,3). However, differentiation of preadipocytes into functionally competent adipocytes is nevertheless insufficient to accommodate the chronic caloric excess and prevent adipocyte overloading. Adipogenic differentiation is a tightly controlled process that involves activation of key transcription factors (PPARg and CEBPa) that induce expression of adipogenic genes (e.g., FABP4 and adiponectin), leading to acquisition of the mature adipocyte phenotype. In addition, epigenetic processes, governed by the actions of chromatin modifying enzymes, including the histone acetyltransferase and histone deacetylase (HDAC) family of proteins, play an important role in adipogenic differentiation (4). We recently reported that histone deacetylase 9 (HDAC9), a class II HDAC, is an endogenous negative regulator of adipogenic differentiation and that downregulation of HDAC9 is necessary for adipogenic differentiation of preadipocytes (5), pointing to a key role of HDAC9 in adipogenic differentiation. In the current study, we report that a chronic high-fat diet (HFD) impairs adipogenic differentiation, promoting accumulation of inefficiently differentiated adipocytes that exhibit diminished expression of adipogenic differentiationspecific genes. We additionally observed that an HFD markedly downregulates beige adipocyte-specific gene expression in white adipose tissues in conjunction with decreased FGF21 expression in this tissue, a known autocrine/ paracrine inducer of beige phenotype in white adipose tissues (6). Preadipocytes from HFD mice exhibit impaired adipogenicdifferentiationinvitro,aphenotypethatpersists despite passage of the cells in culture. This impaired in vitro differentiation is associated with blunted downregulation of HDAC9 expression. Genetic ablation of HDAC9 blocked the deleterious effects of an HFD on adipogenic differentiation, improved insulin sensitivity and glucose tolerance, and prevented ectopic lipid accumulation in the liver. Compared with wild-type mice, HDAC9 knockout mice also exhibit elevated expression of beige adipocyte marker genes in white adipose tissues, concomitant with increased energy expenditure and adaptive thermogenesis. Our study provides evidence that HDAC9 plays a critical role in HFD-induced adipose tissue and metabolic dysfunction, suggesting that targeting HDAC9 could be an effective strategy for combating obesity-related disease. RESEARCH DESIGN AND METHODS Mice HDAC9 knockout mice in the mixed C57BL/6J and 129 background were obtained from Dr. Eric Olson (7). These mice were backcrossed with C57BL/6J mice for seven generations in our laboratories. Subsequently, HDAC9 heterozygous mice were bred to obtain HDAC9 knockout ( 2/2 ) and their wild-type ( +/+ ) littermate controls for use in our experiments. Male HDAC9 knockout mice and their wild-type littermate controls were housed individually and maintained on chow diet after weaning. At 8 weeks of age, these mice were either maintained on chow (Harlan Teklad, LM-485) or switched to an HFD (Research Diet, D12492, with 60% calories from fat) for 12 more weeks. Thereafter, mice were killed, blood was collected via cardiocentesis, and adipose tissues and liver were collected following tissue perfusion with ice-cold saline. All animal studies were conducted using a protocol approved by the Institutional Animal Care and Use Committee of the University of Cincinnati, following appropriate guidelines. Isolation of Preadipocytes, Adipocytes, and In Vitro Adipogenic Differentiation Subcutaneous (inguinal) and epididymal adipose tissues were dissected out, and stromovascular cells and adipocytes were isolated as we described previously by collagenase digestion (5). Preadipocytes were cultured and in vitro differentiated according to the methods as we described previously (5,8). RNA Isolation, Quantitative PCR, Western Blot, and Lipid Droplets Measurement RNA was isolated utilizing a RNeasy lipid mini kit (Qiagen), and quantitative PCR (qpcr) quantification of mrna levels was performed as we described previously (5,8) using a Syber green qpcr kit (Agilent). We selected acidic ribosomal phosphoprotein P0 (Arbp) mrna as a reference for normalization of transcripts under investigation. The choice of Arbp is based on our observation that the levels of this gene in adipose tissues and adipocytes do not change with adipogenic differentiation or with obesity, a finding similar to that reported by Romero et al. (9). The primer sequences used in the qpcr assay are provided in Supplementary Table 1. Western blot analysis and lipid droplet measurement were performed as described previously (5). Mean daily food consumption was determined in chow diet and HFD groups by calculating the amount of consumed food at 24-h intervals for 5 days. These experiments were repeated twice, and the mean daily food consumption was estimated. Body Fat Measurements To assess weight gain, body weights were obtained weekly for mice fed chow or an HFD. Body fat mass was measured in conscious mice fed chow or an HFD for 12 weeks, using 1 H magnetic resonance spectroscopy (EchoMRI-100; Echomedical Systems) as described previously (10).
3 178 HDAC9 in Adipose Tissue Dysfunction Diabetes Volume 63, January 2014 Indirect Calorimetry Mice were allowed to acclimate to respiratory chambers for a period of 2 days. Subsequently, energy expenditure,oxygenconsumption,carbondioxideproduction, and respiratory quotient were measured for 24 h during 12-h light/12-h dark cycles. The volume of oxygen consumed (Vo 2 [ml/(kg 3 h)]) and carbon dioxide produced (Vco 2 [ml/(kg 3 h)]) were measured to calculate energy expenditure (10). Locomotor activity was similarly measured over a 24-h period. Core body temperatures during cold challenge were collected from single housed 12-h fasted mice and were measured rectally with a rodent microprobe and thermometer (Thermalert) at 1 h intervals after placing the mice at 4 C at the beginning of the light cycle. Additional core body temperatures were collected in fed mice at the beginning of the light cycle. Examination of Insulin Levels and Glucose Tolerance For glucose tolerance, glucose levels were measured from tail vein collections immediately prior to and 7.5, 15, 30, 60, 120, and 180 min after an intraperitoneal (IP) injection of glucose at 2 g/kg body weight into mice fasted for 12 h (n = 12 or 8) using glucose strips and repeated measurements. Assessment of pancreatic insulin response to an IP glucose bolus at 2 g/kg body weight involved measuring plasma insulin (Ultra Sensitive Rat Insulin ELISA kit, Crystal Chem, Chicago, IL) in samples collected from tail veins at 0, 7.5, 15, 30, and 60 min after glucose injection in mice fasted for 12 h. Insulin sensitivity was assessed by IP delivery of 0.75 units/kg body weight of porcine insulin and measurement of plasma glucose from tail vein collections at 0, 15, 30, 60, and 90 min after injection in 6-h-fasted mice. Plasma levels of adiponectin, leptin, and resistin were measured in mice after overnight fasting, utilizing commercially available ELISA kits (R&D). Histological Measurement of Adipose Tissue and Liver Tissues were fixedbyimmersioninneutralbuffered formalin (10%), dehydrated in ethanol, and then transferred to xylene solution for embedding in paraffin. Five-micrometer sections were stained with hematoxylin-eosin, and images were analyzed with light microscopy. Brown adipose and hepatic lipid accumulation was examined microscopically after H&E stain. Adipocyte area calculations were determined by measuring the area of greater than 120 cells per 2003 magnification tissue sections using ImageJ version Median adipocyte area was presented as mean 6 SEM. Statistical Analysis Data are expressed as mean 6 SEM. Analysis of variance was accomplished with a one-way ANOVA followed by an unpaired, two-tailed Student t test for P value using Sigma Plot version 11. P values,0.05 were considered statistically significant. Linear regression analysis included R 2 and ANCOVA for the analyses of body weight gain and cumulative energy expenditure data. RESULTS Impaired Adipogenic Differentiation of Preadipocytes From High-Fat Fed Mice The influence of an HFD on adipogenic differentiation was assessed by comparing the ability of preadipocytes from mice fed chow or an HFD to differentiate into mature adipocytes in vitro. Although the number of adipogenic stem/progenitor cells is not reduced by high-fat feeding (3), the potential of these preadipocytes to differentiate into mature adipocytes was markedly blunted. Preadipocytes from chow-fed mice promptly differentiated into mature adipocytes when incubated with adipocyte differentiation medium as expected, with nearly 90% of the cells accumulating lipid droplets in the cytoplasmic compartment (Fig. 1A), accompanied by upregulated expression of adipogenic differentiation-specific genes such as C/EBPa, PPARg, FABP4,andadiponectin(Fig.1B). In contrast, preadipocytes from HFD mice exhibited blunted adipogenic differentiation as evidenced by diminished cytoplasmic lipid droplet accumulation (Fig. 1A) and reduced expression of adipogenic differentiationspecific genes under an identical differentiation protocol (Fig. 1C). The data reported in the current study were obtained using freshly isolated preadipocytes, expanded in culture, and examined after the third passage. Cells at higher passage numbers (6 8) began to show a noticeable decline in their potential for adipogenic differentiation and were thus not extensively studied. Nevertheless, compared with control (chow fed) mice, preadipocytes from HFD mice continued to exhibit reduced adipogenic differentiation despite repeated passaging (5,6) of these cells (data not shown). HFD Perturbs Adipogenic Differentiation via HDAC9 We previously demonstrated that HDAC9 downregulation precedes adipogenic differentiation of preadipocytes both in vitro and in vivo and that overexpression of HDAC9 prevents, while HDAC9 gene deletion accelerates, adipogenic differentiation (5). Together, these data imply that HDAC9 is a negative regulator of adipogenic differentiation, and its downregulation is necessary for activation of the adipogenic differentiation program in preadipocytes (5). This prompted us to determine whether an HFD perturbs adipogenic differentiation through a mechanism involving HDAC9. Basal HDAC9 mrna and protein expression were similar in preadipocytes isolated from chow and HFD mice prior to the onset of differentiation (Fig. 2, day 0). As we previously reported, preadipocytes from chow-fed mice exhibited dramatic downregulation of HDAC9 mrna and protein levels upon induction of adipogenic
4 diabetes.diabetesjournals.org Chatterjee and Associates 179 Figure 1 High-fat feeding impairs in vitro adipogenic differentiation of preadipocytes from mouse subcutaneous adipose tissue. A: Lipid droplet accumulation during in vitro adipogenic differentiation of preadipocytes from subcutaneous adipose depot of chow- and HFD-fed mice. Neutral lipids were labeled with oil red-o and visualized by light microscopy following 7 days of differentiation. Oil red-o stained neutral lipids were extracted from cells following 3 and 7 days of differentiation, quantified spectrophotometrically, and normalized against cellular protein levels (right panel). Values are mean 6 SEM of three experiments (*P < 0.05). B: Induction of adipogenic differentiationspecific genes C/EBPa, PPARg, FABP4, and adiponectin following 7-day in vitro differentiation of subcutaneous preadipocytes from chow-fed mice (*P < 0.005). C: Relative mrna levels of adipogenic differentiation-specific genes in 7-day in vitro differentiated preadipocytes from chow- and HFD-fed mice. mrna levels were quantified by qpcr and normalized to Arbp. Values represent mean 6 SEM of fold increase from chow diet using preadipocytes from three different isolates (*P < 0.005). CD, chow diet. differentiation (Fig. 2). Under identical conditions, preadipocytes from HFD mice exhibited blunted downregulation of HDAC9 mrna and protein levels (Fig. 2). HDAC9 is constitutively a nuclear protein in preadipocytes from both chow- and HFD-fed mice, and the induction of adipogenic differentiation downregulates nuclear HDAC9 protein levels only in preadipocytes from the chow-fed mice (data not shown), a finding consistent with our earlier report (5). These findingssuggestthataberranthdac9expressionin preadipocytes from HFD mice may contribute to blunted adipogenic differentiation of these cells. HDAC9 Gene Deletion Improves Adipogenic Differentiation in HFD-Fed Mice The relationship between an HFD, impaired adipogenic differentiation, and HDAC9 was explored by comparing the differentiation potential of preadipocytes isolated from wild-type mice versus HDAC9 knockout mice after feeding an HFD for 12 weeks. The preadipocytes isolated from HFD wild-type and HDAC9 knockout mice exhibited similar growth rates, morphologic appearance, and preadipocyte-specific Pref1 gene expression (data not shown). However, when induced to undergo adipogenic differentiation, preadipocytes from HDAC9 knockout
5 180 HDAC9 in Adipose Tissue Dysfunction Diabetes Volume 63, January 2014 Figure 2 Impaired in vitro adipogenic differentiation parallels elevated HDAC9 expression. HDAC9 mrna (A) and protein (B) expression were quantified by qpcr and Western blot analyses, respectively, on the indicated days of differentiation. Values represent percentage of change from preadipocytes (0 day) and are expressed as mean 6 SEM of three different experiments. C: Western blot images were analyzed utilizing ImageJ software and expressed as percentage of 0 day levels in each condition after normalization to corresponding glyceraldehyde-3-phosphate dehydrogenase values. *P < CD, chow diet; GAPDH, glyceraldehyde-3-phosphate dehydrogenase. mice fed an HFD showed significant lipid accumulation and elevated expression of adipogenic differentiationspecific genes (C/EBPa, PPARg, FABP4, and adiponectin) at levels similar to those observed during differentiation of preadipocytes from chow-fed mice and in marked contrast to the results observed in preadipocytes from HFD wild-type mice (Fig. 3). To verify that these in vitro findings are relevant to adipogenic differentiation in vivo, we examined adipocytes freshly isolated from adipose tissues by collagenase digestion. HDAC9 mrna and protein levels were significantly higher in freshly isolated adipocytes and adipose tissues from HFD wild-type mice compared with chow-fed mice (Fig. 4A). Importantly, freshly isolated adipocytes from HFD wild-type mice exhibited blunted expression of adipocyte-differentiationspecific genes C/EBPa, PPARg, and adiponectin, which was abrogated by HDAC9 gene deletion (Fig. 4B). HDAC9 gene deletion similarly prevented blunted adipogenic gene expression in adipose tissues from both subcutaneous and visceral (epididymal) depots of HFD mice (Supplementary Fig. 1). Taken together, these findings suggest that HDAC9 expression contributes to HFD-induced impaired adipogenic differentiation. HDAC9 Gene Deletion Prevents HFD-Induced Obesity We next examined the effects of HDAC9 gene deletion on HFD-induced obesity. Our initial survey demonstrated that chow-fed HDAC9 knockout mice displayed a slight but statistically insignificant trend of lower body weight ( g) compared with chow-fed wild-type mice ( g). Upon high-fat feeding, HDAC9 knockout mice gained significantly less weight compared with wildtype control mice (Fig. 5A and B). However, HDAC9 knockout and wild-type control mice exhibited similar locomotor activities (Fig. 5C) and consumed comparable amounts of food (Fig. 5D). Nuclear magnetic resonance evaluation of whole body adiposity demonstrated reduced fat mass both in chow- and HFD-fed HDAC9 knockout mice (Fig. 5E), and subcutaneous and epididymal adipose tissue weights were lower in HDAC9 knockout mice compared with wild-type mice (Fig. 5F and G). Histological analysis of the subcutaneous adipose tissues showed that adipocytes in HDAC9 knockout mice were smaller under HFD-fed conditions compared with their wild-type littermate controls (Fig. 5H and I) and that multilocular adipocytes were abundant in chow-fed HDAC9 knockout mice (Fig. 5I), a finding reminiscent of beige phenotypic changes of white adipose tissue (11). HDAC9 Gene Deletion Improves Glucose Tolerance, Insulin Sensitivity, and Lipid Accumulation in Liver Comparison of glucose tolerance between wild-type and HDAC9 knockout mice demonstrated that HDAC9 knockout mice were significantly more glucose tolerant following both chow diet (Fig. 6A) and an HFD (Fig. 6B). The improved glucose tolerance in HDAC9 knockout mice was not due to enhanced insulin secretion, as similar plasma insulin levels were observed in both
6 diabetes.diabetesjournals.org Chatterjee and Associates 181 Figure 3 HDAC9 gene deletion improves adipogenic differentiation of preadipocytes from subcutaneous adipose tissues of HFD mice. Adipogenic gene expression (A) and lipid droplet accumulation (B) were determined as described in Fig. 1. Values represent mean 6 SEM from four experiments. *P < groups of chow-fed mice (Fig. 6C), while plasma insulin levels were significantly lower in HFD-fed HDAC9 knockout mice as compared with wild-type mice (Fig. 6D). Insulin injection into chow-fed wild-type and HDAC9 knockout mice induced a rapid decrease in plasma glucose levels, consistent with preserved insulin sensitivity (Fig. 6E). Importantly, after an HFD, insulin sensitivity was improved in HDAC9 knockout mice as compared with wild-type mice (Fig. 6F). Future studies are needed to determine the relative effects of HDAC9 gene deletion on insulin sensitivity of specific target cells, including adipocytes. Under these experimental conditions, an HFD did not significantly affect plasma adiponectin (data not shown) or lipid profile, except for total cholesterol levels (Supplementary Table 2). However, an HFD upregulated plasma proinflammatory adipokines leptin (Fig. 6G) and resistin (Fig. 6H) in wild-type mice, which was attenuated in HDAC9 knockout mice. Because adipose tissue dysfunction promotes ectopic lipid deposition and accumulation in nonadipose tissues, we assessed lipid accumulation in the liver following an HFD. An HFD precipitated dramatic lipid accumulation in the livers of wild-type mice, which was completely prevented by HDAC9 gene deletion (Fig. 6I). Additionally, liver mass was significantly lower in HDAC9 knockout mice compared with wild-type mice (Fig. 6J). These results provide evidence that HDAC9 gene deletion is also protective against HFD-induced hepatosteatosis. HDAC9 Regulates Energy Expenditure and Adaptive Thermogenesis: Effects on Brown Adipose Tissue Our data indicated that HDAC9 gene deletion results in reduced fat mass and improved metabolic state despite no differences in locomotor activity or food consumption. Subsequently, we determined that HDAC9 knockout mice exhibited elevated energy expenditure (Fig. 7A) and oxygen consumption (Fig. 7B) compared with their wild-type littermates under chow-fed conditions. No differences were detected in respiratory quotient between genotypes (data not shown). In addition, as compared with wild-type mice, HDAC9 knockout mice exhibited a higher basal body temperature and were protected against hypothermia following cold challenge (Fig. 7C), implying improved adaptive thermogenic capacity. The above observations raised the possibility that HDAC9 gene deletion might affect the amount and/or quality of thermogenic brown adipose tissue. HDAC9 is expressed in brown adipose tissue, and HDAC9 knockout mice surprisingly exhibited nearly 50% less brown adipose tissue mass compared with similarly maintained wild-type mice (Fig. 7D). Interestingly, histology demonstrated reduced lipid accumulation in brown adipose tissue of HDAC9 knockout mice fed an HFD as compared with wild-type mice (Fig. 7E). Thus we next investigated the impact of an HFD on the phenotype of brown adipose tissue. Unlike the dramatic changes observed in white adipose tissues (Fig. 1C), brown adipose tissue was
7 182 HDAC9 in Adipose Tissue Dysfunction Diabetes Volume 63, January 2014 Figure 4 HDAC9 gene deletion prevents accumulation of inefficiently differentiated adipocytes in subcutaneous adipose tissue of HFD mice. A: Adipocytes (in vivo differentiated) were isolated from subcutaneous adipose depots of chow- and HFD-fed mice; HDAC9 mrna levels in these cells were analyzed by qpcr. HDAC9 protein levels were determined in epididymal adipose tissue of chow- and HFD-fed mice by Western blot analysis. For mrna expression, values represent mean 6 SEM from four groups of mice (*P < 0.001), while ImageJ analysis of Western blot data were normalized to corresponding glyceraldehyde-3-phosphate dehydrogenase values and represent mean 6 SEM from three groups of mice. B: Effects of an HFD on mrna expression of adipogenic differentiation-specific genes C/EBPa, PPARg, and adiponectin in freshly isolated adipocytes (in vivo differentiated) from subcutaneous adipose tissues of wild-type ( +/+ ) and HDAC9 knockout ( 2/2 ) mice. Values are mean 6 SEM of four experiments. *P < 0.05; CD, chow diet; GAPDH, glyceraldehyde- 3-phosphate dehydrogenase. largely unaffected by an HFD, as demonstrated by qualitatively minor changes in the expression of adipogenic differentiation-specific or thermogenic genes (Fig. 7F), findings similar to those of Fitzgibbons et al. (12). Moreover, expression of UCP1 in brown adipose tissue was not upregulated in HDAC9 knockout mice as compared with wild-type mice in the setting of an HFD (Fig. 7F). Taken together, these findings suggest that alterations in the phenotype of brown adipose tissue consequent to HDAC9 gene deletion are unlikely to account for the marked differences in lean body mass, energy expenditure, adaptive thermogenesis, and metabolic profile following an HFD. Effects of an HFD on Beige Adipocyte Gene Expression: Impact of HDAC9 Gene Deletion Emerging evidence indicates that a specific class of brown-like adipocytes (beige adipocytes), with thermogenic characteristics, accumulate in the white adipose depots of mice (13). Beige adipocytes are reported to be involved in regulation of adaptive thermogenesis and energy expenditure, body fat accumulation, and metabolic homeostasis (14 18). To investigate the potential role of HDAC9 in regulating beige adipocytes, we first compared the levels of beige-specific genes in adipocytes from subcutaneous adipose tissues of chow-fed wild-type and HDAC9 knockout mice. Expression of beige adipocyte marker genes PRDM16, UCP1, CIDEA, and PGC1a was significantly higher (Fig. 8A), and clusters of multiloculated beige adipocytes were more readily apparent (Fig. 5I) in HDAC9 knockout mice as compared with wild-type mice. Importantly, an HFD dramatically decreased expression of beige adipocyte marker genes in wild-type mice, which was strongly abrogated by HDAC9 gene deletion (Fig. 8A). By comparison, the effects of an HFD and HDAC9 gene deletion on beige adipocyte marker genes in adipocytes from epididymal adipose tissue were not significant (Fig. 8B). These findings pertaining to differential expression of beige adipocytespecific genes in subcutaneous adipose tissues are consistent with the leaner adipose tissue mass, increased energy expenditure, and enhanced adaptive thermogenesis observed in HDAC9 knockout mice. Because adipose tissue derived fibroblast growth factor 21 (FGF21) was shown previously to function as an autocrine/paracrine inducer of thermogenic gene expression and beige phenotype in white adipose tissues (6), we examined FGF21 expression in subcutaneous adipocytes of chow- versus HFD-fed mice. An HFD profoundly reduced FGF21 levels in subcutaneous adipocytes of wild-type but not HDAC9 knockout mice (Fig. 8C), suggesting that preserved FGF21 expression, leading to increase in beige adipocytes, may, in part, mediate the beneficial effects of HDAC9 gene deletion on HFD-induced adipose tissue dysfunction. An HFD, however, did not influence FGF21 levels in epididymal adipocytes.
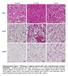
8 diabetes.diabetesjournals.org Chatterjee and Associates 183 Figure 5 HDAC9 gene deletion attenuates the gain in body weight, adiposity, and adipocyte enlargement induced by an HFD. A: Growth curves of male wild-type (filled circles) and HDAC9 knockout mice (open circles) fed an HFD (*P < ). Slope of wild-type versus HDAC9 knockout growth curves. B: Mean weekly body weight gain of wild-type ( +/+ ) and HDAC9 knockout ( 2/2 ) mice fed an HFD (*P < 0.001). C: Cumulative locomotor activities over a 24-h period of wild-type ( +/+ ) and knockout mice ( 2/2 ) on chow. D: Mean daily food consumption in wild-type mice fed chow (open circles) and an HFD (closed circles) versus comparably fed HDAC9 knockout mice (open and closed triangles, respectively). E: Whole body adiposity measured by nuclear magnetic resonance in chow and HFD mice. Black bars represent wild-type while gray bars represent HDAC9 knockout mice (*P < 0.05). F: Subcutaneous and (G) epididymal adipose tissue weights of wild-type (black bars) and HDAC9 knockout (gray bars) mice maintained on chow or an HFD for 12 weeks. Values are mean 6 SEM from six mice (#P < 0.05 compared with wild-type CD mice; *P < 0.05 compared with wild-type HFD mice). H: Median adipocyte cross-sectional areas were determined from adipose tissues collected from mice fed chow or an HFD for 12 weeks. Data represent mean 6 SEM of median adipocyte areas from n = 6 per group (*P < 0.05). I: Microscopic images of H&E stained subcutaneous adipose tissues from wild-type ( +/+ ) and HDAC9 knockout ( 2/2 ) mice maintained on chow or an HFD for 12 weeks. Note the presence of multilocular adipocytes (arrows) within the inset. CD, chow diet; SQ, subcutaneous; Epi, epididymal. DISCUSSION We previously demonstrated that HDAC9 is a negative regulator of adipogenic differentiation; expression of HDAC9 falls abruptly at the onset of differentiation of preadipocytes, thereby permitting activation of the adipogenic differentiation program (5). We now report that this regulatory mechanism is disrupted by an HFD, leading to blunted HDAC9 downregulation and impaired adipogenic differentiation. HDAC9 gene deletion prevents impaired adipogenesis in vitro and accumulation of improperly differentiated adipocytes in vivo while reducing ectopic lipid accumulation in the liver and improving systemic insulin sensitivity and glucose tolerance. Additionally, HDAC9 knockout mice exhibit increased energy expenditure and are protected from the hypothermic effects of fasting and cold challenge. Moreover, HDAC9 gene deletion promotes elevated basal expression of beige adipocyte marker genes in white adipose tissue and prevents their downregulation in response to an HFD. HDAC9 gene deletion also prevents HFD-induced downregulation of FGF21, a known autocrine/paracrine inducer of beige phenotype,inwhiteadiposetissues(6).ourfindings thus provide novel insight into the role of HDAC9 in
9 184 HDAC9 in Adipose Tissue Dysfunction Diabetes Volume 63, January 2014 Figure 6 HDAC9 gene deletion improves glucose tolerance, insulin sensitivity, and lipid accumulation in liver. Glucose tolerance of mice fed chow (A) oranhfd(b) for 12 weeks and glucose-stimulated insulin secretion (C, D) were determined following an IP injection of a glucose solution (2 g/kg body weight). Closed symbols represent wild-type ( +/+ ) and open symbols represent HDAC9 knockout ( 2/2 ) mice. Values represent mean 6 SEM from 6 10 mice in each group (note that baseline insulin levels were subtracted in C; *P < 0.05 compared with comparably fed wild-type mice). Systemic insulin sensitivity was assessed measuring plasma glucose concentrations after an IP injection of insulin (0.75 units/kg) in chow- (E) and HFD-fed (F) mice following a 6-h fast. Values represent mean 6 SEM from six mice in each group (*P < 0.05 compared with wild-type HFD). Plasma leptin (G) and resistin (H) levels were assayed in wild-type and HDAC9 knockout mice maintained on chow and an HFD. Values are mean 6 SEM from six mice in each group (*P < 0.05 compared with chow; #P < 0.05 compared with HFD fed wild-type mice). I: Representative histology of hemotoxylin and eosin stained paraffin-embedded liver tissue sections from HFD-fed wild-type and HDAC9 knockout mice. J: Liver mass of wild-type and HDAC9 knockout mice fed chow and an HFD (*P < 0.05). Values are mean 6 SEM from 4 6 mice in each group. AUC, area under the curve; CD, chow diet. regulating adipose tissue function during diet-induced obesity. The mechanisms whereby an HFD disrupts HDAC9 downregulation and adipogenic differentiation of preadipocytes remain to be determined. An HFD does not negatively influence adipogenic precursor cell abundance in adipose tissues (3), implying that sufficient numbers of precursor cells are available to support adipogenic differentiation. Moreover, HDAC9 levels in preadipocytes from chow and HFD mice were similar prior to the onset of adipogenic differentiation. However, unlike preadipocytes from chow-fed mice, preadipocytes from HFD mice failed to sufficiently downregulate HDAC9 expression, which appears necessary for adipogenic differentiation (5). Considering that the cells in our study were isolated and propagated through in vitro culture with multiple rounds of cell division, the impaired adipocyte differentiation is independent of the impact of an HFD on the in vivo milieu of adipose tissues (e.g., hypoxia, neuronal input, and hormonal influences).ourresults,therefore,suggest that an HFD impairs adipocyte differentiation through cell autonomous and heritable (epigenetic) mechanisms involving dysregulated HDAC9 expression in these cells. The precise nature of these epigenetic regulatory mechanisms, and whether they are reversible
10 diabetes.diabetesjournals.org Chatterjee and Associates 185 Figure 7 HDAC9 knockout mice exhibit increased energy expenditure, oxygen consumption, and resistance to cold challenge. A: Energy expenditure (24 h) was determined in wild-type (black line) and HDAC9 knockout (red line) mice maintained on chow. The thick black region in the x-axis represents dark period (12 h). B: O 2 consumption was quantified in wild-type ( +/+ ) and HDAC9 knockout ( 2/2 ) mice maintained on chow. C: Body temperature was measured under basal conditions and following cold challenge. D: Brown adipose tissue (interscapular) depots were dissected and weighed in wild-type and HDAC9 knockout mice fed a chow diet. E: Histology (H&E staining) shows lipid accumulation in brown adipose tissue following an HFD in wild-type and HDAC9 knockout mice. F: Expression of adipogenic (PPARg and FABP4) and brown adipose tissue (PRDM16, CIDEA, PGC1a, and UCP1) marker genes in brown (interscapular) adipose tissue following an HFD and chow was quantified in wild-type and HDAC9 knockout mice. *P < 0.05; CD, chow diet; KO, knockout. with prolonged discontinuation of an HFD, remains to be determined. Our data also suggest that endogenous HDAC9 directlycontributestotheadversesystemicmetabolic consequences of an HFD. Thus HDAC9 knockout mice were leaner, less glucose intolerant, and more insulin sensitive than their wild-type littermates despite similar food consumption and locomotor activity. HDAC9 is expressed in islet b-cells, and HDAC9 gene deletion in the pancreas was reported to enhance b-cell mass (19). Although these observations suggested that HDAC9 inactivation potentially could confer metabolic benefits by increasing insulin secretion, our data indicate that differences in insulin sensitivity, and not secretion, are responsible for improved glucose tolerance in HDAC9 knockout mice (Fig. 6). Rather, our datasuggestthatthemetabolicbenefits of HDAC9 gene deletion in a chronic HFD are potentially the result of increased thermogenic energy expenditure related to upregulated beige adipocyte gene expression. Thus beige-specific genes in subcutaneous adipose tissues are downregulated during an HFD in wild-type mice,inparallelwithincreasedbodymass,expanded white adipose tissue depots, systemic insulin resistance, and glucose intolerance, all of which were markedly attenuated in HDAC9 knockout mice. By contrast, while HDAC9 gene deletion led to reduced brown tissue adipose mass during an HFD, it had little
11 186 HDAC9 in Adipose Tissue Dysfunction Diabetes Volume 63, January 2014 Figure 8 HDAC9 gene deletion prevents beige adipocyte marker genes9 downregulation in subcutaneous adipocytes of HFD-fed mice. Expression of beige adipocyte marker genes in subcutaneous (A) and epididymal (B) adipocytes of wild-type and HDAC9 knockout mice fed chow and an HFD was determined by RT-qPCR as described in Fig. 1 (*P < 0.05). Compared with subcutaneous, epididymal adipocytes express nearly 100-fold lower UCP1 mrna levels in wild-type mice under chow-fed conditions. C: FGF21 mrna levels in subcutaneous and epididymal adipocytes of wild-type and HDAC9 knockout mice on chow and an HFD (*P < 0.001). Values are mean 6 SEM of 4 6 mice in each experimental condition. Compared with subcutaneous, epididymal adipocytes express nearly 10-fold lower FGF21 mrna levels in wild-type mice under chow-fed conditions. CD, chow diet; SQ, subcutaneous; Epi, epididymal. impact on thermogenic brown adipose gene expression. Whether the reduced brown adipose tissue mass is a compensatory response to increased beige adipocytes, or vice versa, is presently unclear (15). Further investigations will be required to establish with certainty whether increased beige adipocyte gene expression mediates the improved metabolic phenotype of HDAC9 knockout mice. Nevertheless, these findings raise the intriguing possibility that downregulation of beige adipocytes contributes to the deleterious metabolic effects of an HFD, a process that could potentially be ablated by therapeutically targeting HDAC9. Precisely how HDAC9 regulates beige adipocytes remains to be determined; however, we observed that FGF21 expression was dramatically reduced in subcutaneous adipocytes of HFD-fed wild-type, but not of HDAC9 knockout, mice. FGF21 plays a major role in inducing beige adipocyte gene expression, including UCP1, leading to upregulation of thermogenesis (6,20). ThefactthatHDAC9genedeletionpreventsHFDinduced FGF21 downregulation implies that endogenous HDAC9 could be a negative regulator of FGF21. Further studies will be required to determine the potentially complex underlying mechanisms of regulation of FGF21 by HDAC9 during an HFD. Since beige adipocytes are inversely related to obesity in humans (14,21,22), our findings support the notion that targeting HDAC9 could be a fruitful approach to treating obesity-related disease. Funding. This study was supported by National Institutes of Health grants HL and HL (to N.L.W.). Duality of Interest. No potential conflicts of interest relevant to this article were reported. Author Contributions. T.K.C., D.Y.H., and N.L.W. researched data; contributed to the discussion; and wrote, reviewed, and edited the manuscript. J.E.B. researched data, contributed to the discussion, and wrote the manuscript. E.K. and W.S.T. researched data. V.B. and A.B.L. researched data and contributed to the discussion. A.L.B. and S.R. contributed to the discussion and reviewed and edited the manuscript. T.K.C. is the guarantor of this work and, as such, had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
12 diabetes.diabetesjournals.org Chatterjee and Associates 187 References 1. Hauner H. Secretory factors from human adipose tissue and their functional role. Proc Nutr Soc 2005;64: Rigamonti A, Brennand K, Lau F, Cowan CA. Rapid cellular turnover in adipose tissue. PLoS One 2011;6:e Joe AW, Yi L, Even Y, Vogl AW, Rossi FM. Depot-specific differences in adipogenic progenitor abundance and proliferative response to high-fat diet. Stem Cells 2009;27: Pinnick KE, Karpe F. DNA methylation of genes in adipose tissue. Proc Nutr Soc 2011;70: Chatterjee TK, Idelman G, Blanco V, et al. Histone deacetylase 9 is a negative regulator of adipogenic differentiation. J Biol Chem 2011;286: Fisher FM, Kleiner S, Douris N, et al. FGF21 regulates PGC-1a and browning of white adipose tissues in adaptive thermogenesis. Genes Dev 2012;26: Zhang CL, McKinsey TA, Chang S, Antos CL, Hill JA, Olson EN. Class II histone deacetylases act as signal-responsive repressors of cardiac hypertrophy. Cell 2002;110: Chatterjee TK, Stoll LL, Denning GM, et al. Proinflammatory phenotype of perivascular adipocytes: influence of high-fat feeding. Circ Res 2009;104: Romero MdelM, Grasa MdelM, Esteve M, Fernández-López JA, Alemany M. Semiquantitative RT-PCR measurement of gene expression in rat tissues including a correction for varying cell size and number. Nutr Metab (Lond) 2007;4: Hofmann SM, Zhou L, Perez-Tilve D, et al. Adipocyte LDL receptor-related protein-1 expression modulates postprandial lipid transport and glucose homeostasis in mice. J Clin Invest 2007;117: Seale P, Conroe HM, Estall J, et al. Prdm16 determines the thermogenic program of subcutaneous white adipose tissue in mice. J Clin Invest 2011; 121: Fitzgibbons TP, Kogan S, Aouadi M, Hendricks GM, Straubhaar J, Czech MP. Similarity of mouse perivascular and brown adipose tissues and their resistance to diet-induced inflammation. Am J Physiol Heart Circ Physiol 2011;301:H1425 H Wu J, Boström P, Sparks LM, et al. Beige adipocytes are a distinct type of thermogenic fat cell in mouse and human. Cell 2012;150: Wu J, Cohen P, Spiegelman BM. Adaptive thermogenesis in adipocytes: is beige the new brown? Genes Dev 2013;27: Schulz TJ, Huang P, Huang TL, et al. Brown-fat paucity due to impaired BMP signalling induces compensatory browning of white fat. Nature 2013; 495: Auffret J, Viengchareun S, Carré N, et al. Beige differentiation of adipose depots in mice lacking prolactin receptor protects against high-fat-dietinduced obesity. FASEB J 2012;26: Qian SW, Tang Y, Li X, et al. BMP4-mediated brown fat-like changes in white adipose tissue alter glucose and energy homeostasis. Proc Natl Acad Sci USA 2013;110:E798 E Ye L, Wu J, Cohen P, et al. Fat cells directly sense temperature to activate thermogenesis. Proc Natl Acad Sci U S A 2013;110: Lenoir O, Flosseau K, Ma FX, et al. Specific control of pancreatic endocrine b- and d-cell mass by class IIa histone deacetylases HDAC4, HDAC5, and HDAC9. Diabetes 2011;60: Lee P, Werner CD, Kebebew E, Celi FS. Functional thermogenic beige adipogenesis is inducible in human neck fat. Int J Obes (Lond). 21 May [Epub ahead of print] 21. Cypess AM, Lehman S, Williams G, et al. Identification and importance of brown adipose tissue in adult humans. Genes Dev 2013;27: Sharp LZ, Shinoda K, Ohno H, et al. Human BAT possesses molecular signatures that resemble beige/brite cells. PLoS One 2012;7:e49452
GPR120 *** * * Liver BAT iwat ewat mwat Ileum Colon. UCP1 mrna ***
 a GPR120 GPR120 mrna/ppia mrna Arbitrary Units 150 100 50 Liver BAT iwat ewat mwat Ileum Colon b UCP1 mrna Fold induction 20 15 10 5 - camp camp SB202190 - - - H89 - - - - - GW7647 Supplementary Figure
a GPR120 GPR120 mrna/ppia mrna Arbitrary Units 150 100 50 Liver BAT iwat ewat mwat Ileum Colon b UCP1 mrna Fold induction 20 15 10 5 - camp camp SB202190 - - - H89 - - - - - GW7647 Supplementary Figure
Males- Western Diet WT KO Age (wks) Females- Western Diet WT KO Age (wks)
 Relative Arv1 mrna Adrenal 33.48 +/- 6.2 Skeletal Muscle 22.4 +/- 4.93 Liver 6.41 +/- 1.48 Heart 5.1 +/- 2.3 Brain 4.98 +/- 2.11 Ovary 4.68 +/- 2.21 Kidney 3.98 +/-.39 Lung 2.15 +/-.6 Inguinal Subcutaneous
Relative Arv1 mrna Adrenal 33.48 +/- 6.2 Skeletal Muscle 22.4 +/- 4.93 Liver 6.41 +/- 1.48 Heart 5.1 +/- 2.3 Brain 4.98 +/- 2.11 Ovary 4.68 +/- 2.21 Kidney 3.98 +/-.39 Lung 2.15 +/-.6 Inguinal Subcutaneous
SUPPLEMENTARY INFORMATION
 doi: 1.138/nature7221 Brown fat selective genes 12 1 Control Q-RT-PCR (% of Control) 8 6 4 2 Ntrk3 Cox7a1 Cox8b Cox5b ATPase b2 ATPase f1a1 Sirt3 ERRα Elovl3/Cig3 PPARα Zic1 Supplementary Figure S1. stimulates
doi: 1.138/nature7221 Brown fat selective genes 12 1 Control Q-RT-PCR (% of Control) 8 6 4 2 Ntrk3 Cox7a1 Cox8b Cox5b ATPase b2 ATPase f1a1 Sirt3 ERRα Elovl3/Cig3 PPARα Zic1 Supplementary Figure S1. stimulates
SUPPLEMENTARY INFORMATION
 doi:10.1038/nature12652 Supplementary Figure 1. PRDM16 interacts with endogenous EHMT1 in brown adipocytes. Immunoprecipitation of PRDM16 complex by flag antibody (M2) followed by Western blot analysis
doi:10.1038/nature12652 Supplementary Figure 1. PRDM16 interacts with endogenous EHMT1 in brown adipocytes. Immunoprecipitation of PRDM16 complex by flag antibody (M2) followed by Western blot analysis
Supplementary Figure 1. DNA methylation of the adiponectin promoter R1, Pparg2, and Tnfa promoter in adipocytes is not affected by obesity.
 Supplementary Figure 1. DNA methylation of the adiponectin promoter R1, Pparg2, and Tnfa promoter in adipocytes is not affected by obesity. (a) Relative amounts of adiponectin, Ppar 2, C/ebp, and Tnf mrna
Supplementary Figure 1. DNA methylation of the adiponectin promoter R1, Pparg2, and Tnfa promoter in adipocytes is not affected by obesity. (a) Relative amounts of adiponectin, Ppar 2, C/ebp, and Tnf mrna
Supplementary Figure 1. DJ-1 modulates ROS concentration in mouse skeletal muscle.
 Supplementary Figure 1. DJ-1 modulates ROS concentration in mouse skeletal muscle. (a) mrna levels of Dj1 measured by quantitative RT-PCR in soleus, gastrocnemius (Gastroc.) and extensor digitorum longus
Supplementary Figure 1. DJ-1 modulates ROS concentration in mouse skeletal muscle. (a) mrna levels of Dj1 measured by quantitative RT-PCR in soleus, gastrocnemius (Gastroc.) and extensor digitorum longus
control kda ATGL ATGLi HSL 82 GAPDH * ** *** WT/cTg WT/cTg ATGLi AKO/cTg AKO/cTg ATGLi WT/cTg WT/cTg ATGLi AKO/cTg AKO/cTg ATGLi iwat gwat ibat
 body weight (g) tissue weights (mg) ATGL protein expression (relative to GAPDH) HSL protein expression (relative to GAPDH) ### # # kda ATGL 55 HSL 82 GAPDH 37 2.5 2. 1.5 1..5 2. 1.5 1..5.. Supplementary
body weight (g) tissue weights (mg) ATGL protein expression (relative to GAPDH) HSL protein expression (relative to GAPDH) ### # # kda ATGL 55 HSL 82 GAPDH 37 2.5 2. 1.5 1..5 2. 1.5 1..5.. Supplementary
BEIGE AND BROWN FAT: BASIC BIOLOGY AND NOVEL THERAPEUTICS Dr. Carl Ascoli
 BEIGE AND BROWN FAT: BASIC BIOLOGY AND NOVEL THERAPEUTICS Dr. Carl Ascoli Symposium Co-Chairs: Bruce M. Spiegelman (Harvard/Dana Farber) and Sven Enerbäck (U.Gothenburg) April 17-23, 2015 Snowbird Resort,
BEIGE AND BROWN FAT: BASIC BIOLOGY AND NOVEL THERAPEUTICS Dr. Carl Ascoli Symposium Co-Chairs: Bruce M. Spiegelman (Harvard/Dana Farber) and Sven Enerbäck (U.Gothenburg) April 17-23, 2015 Snowbird Resort,
Supplementary Materials for
 www.sciencesignaling.org/cgi/content/full/8/407/ra127/dc1 Supplementary Materials for Loss of FTO in adipose tissue decreases Angptl4 translation and alters triglyceride metabolism Chao-Yung Wang,* Shian-Sen
www.sciencesignaling.org/cgi/content/full/8/407/ra127/dc1 Supplementary Materials for Loss of FTO in adipose tissue decreases Angptl4 translation and alters triglyceride metabolism Chao-Yung Wang,* Shian-Sen
A synergistic anti-obesity effect by a combination of capsinoids and cold temperature through the promotion of beige adipocyte biogenesis
 A synergistic anti-obesity effect by a combination of capsinoids and cold temperature through the promotion of beige adipocyte biogenesis Kana Ohyama, 1,2 Yoshihito Nogusa, 1 Kosaku Shinoda, 2 Katsuya
A synergistic anti-obesity effect by a combination of capsinoids and cold temperature through the promotion of beige adipocyte biogenesis Kana Ohyama, 1,2 Yoshihito Nogusa, 1 Kosaku Shinoda, 2 Katsuya
General Laboratory methods Plasma analysis: Gene Expression Analysis: Immunoblot analysis: Immunohistochemistry:
 General Laboratory methods Plasma analysis: Plasma insulin (Mercodia, Sweden), leptin (duoset, R&D Systems Europe, Abingdon, United Kingdom), IL-6, TNFα and adiponectin levels (Quantikine kits, R&D Systems
General Laboratory methods Plasma analysis: Plasma insulin (Mercodia, Sweden), leptin (duoset, R&D Systems Europe, Abingdon, United Kingdom), IL-6, TNFα and adiponectin levels (Quantikine kits, R&D Systems
1.5 ASK1KO fed. fasted 16 hrs w/o water. Fed. 4th. 4th WT ASK1KO N=29, 11(WT), ,5(ASK1KO) ASK1KO ASK1KO **** Time [h]
![1.5 ASK1KO fed. fasted 16 hrs w/o water. Fed. 4th. 4th WT ASK1KO N=29, 11(WT), ,5(ASK1KO) ASK1KO ASK1KO **** Time [h] 1.5 ASK1KO fed. fasted 16 hrs w/o water. Fed. 4th. 4th WT ASK1KO N=29, 11(WT), ,5(ASK1KO) ASK1KO ASK1KO **** Time [h]](/thumbs/87/97258189.jpg) 7: 13: 19: 1: 7: 151117 a 151117 4th 4th b c RQ.95 KO.9.85.8.75.7 light dark light dark.65 7: 19: 7: 19: 7: Means ± SEM, N=6 RQ 1..9.8.7.6.6 KO CL (-) CL (+) ibat weight ratio (/body weight) [%].5.4.3.2.1
7: 13: 19: 1: 7: 151117 a 151117 4th 4th b c RQ.95 KO.9.85.8.75.7 light dark light dark.65 7: 19: 7: 19: 7: Means ± SEM, N=6 RQ 1..9.8.7.6.6 KO CL (-) CL (+) ibat weight ratio (/body weight) [%].5.4.3.2.1
Supplemental Information Supplementary Table 1. Tph1+/+ Tph1 / Analyte Supplementary Table 2. Tissue Vehicle LP value
 Supplemental Information Supplementary Table. Urinary and adipose tissue catecholamines in Tph +/+ and Tph / mice fed a high fat diet for weeks. Tph +/+ Tph / Analyte ewat ibat ewat ibat Urine (ng/ml)
Supplemental Information Supplementary Table. Urinary and adipose tissue catecholamines in Tph +/+ and Tph / mice fed a high fat diet for weeks. Tph +/+ Tph / Analyte ewat ibat ewat ibat Urine (ng/ml)
* This work was supported, in whole or in part, by National Institutes of Health Grants HL076684, HL62984 (to N. L. W.), and ES (to S. H.
 THE JOURNAL OF BIOLOGICAL CHEMISTRY VOL. 286, NO. 31, pp. 27836 27847, August 5, 2011 Printed in the U.S.A. Histone Deacetylase 9 Is a Negative Regulator of Adipogenic Differentiation * S Received for
THE JOURNAL OF BIOLOGICAL CHEMISTRY VOL. 286, NO. 31, pp. 27836 27847, August 5, 2011 Printed in the U.S.A. Histone Deacetylase 9 Is a Negative Regulator of Adipogenic Differentiation * S Received for
Effects of growth hormone secretagogue receptor agonist and antagonist in nonobese type 2 diabetic MKR mice
 Effects of growth hormone secretagogue receptor agonist and antagonist in nonobese type 2 diabetic MKR mice Rasha Mosa (MBCHC, M.D, PhD candidate) School of Biomedical Sciences University of Queensland
Effects of growth hormone secretagogue receptor agonist and antagonist in nonobese type 2 diabetic MKR mice Rasha Mosa (MBCHC, M.D, PhD candidate) School of Biomedical Sciences University of Queensland
SUPPLEMENTARY INFORMATION
 DOI: 10.1038/ncb2211 a! mir-143! b! mir-103/107! let-7a! mir-144! mir-122a! mir-126-3p! mir-194! mir-27a! mir-30c! Figure S1 Northern blot analysis of mir-143 expression dependent on feeding conditions.
DOI: 10.1038/ncb2211 a! mir-143! b! mir-103/107! let-7a! mir-144! mir-122a! mir-126-3p! mir-194! mir-27a! mir-30c! Figure S1 Northern blot analysis of mir-143 expression dependent on feeding conditions.
Supplemental Information. Increased 4E-BP1 Expression Protects. against Diet-Induced Obesity and Insulin. Resistance in Male Mice
 Cell Reports, Volume 16 Supplemental Information Increased 4E-BP1 Expression Protects against Diet-Induced Obesity and Insulin Resistance in Male Mice Shih-Yin Tsai, Ariana A. Rodriguez, Somasish G. Dastidar,
Cell Reports, Volume 16 Supplemental Information Increased 4E-BP1 Expression Protects against Diet-Induced Obesity and Insulin Resistance in Male Mice Shih-Yin Tsai, Ariana A. Rodriguez, Somasish G. Dastidar,
Figure S1. Body composition, energy homeostasis and substrate utilization in LRH-1 hep+/+ (white bars) and LRH-1 hep-/- (black bars) mice.
 Figure S1. Body composition, energy homeostasis and substrate utilization in LRH-1 hep+/+ (white bars) and LRH-1 hep-/- (black bars) mice. (A) Lean and fat masses, determined by EchoMRI. (B) Food and water
Figure S1. Body composition, energy homeostasis and substrate utilization in LRH-1 hep+/+ (white bars) and LRH-1 hep-/- (black bars) mice. (A) Lean and fat masses, determined by EchoMRI. (B) Food and water
A novel role for vitamin D: modulation of expression and function of the local renin angiotensin system in mouse pancreatic islets
 Diabetologia () 5:77 DOI.7/s5--- SHORT COMMUNICATION A novel role for vitamin D: modulation of expression and function of the local renin angiotensin system in mouse pancreatic islets Q. Cheng & Y. C.
Diabetologia () 5:77 DOI.7/s5--- SHORT COMMUNICATION A novel role for vitamin D: modulation of expression and function of the local renin angiotensin system in mouse pancreatic islets Q. Cheng & Y. C.
Mouse Glu-OC (undercarboxylated osteocalcin) and Gla-OC (carboxylated osteocalcin) levels were
 Supplemental Data Supplemental Materials and Methods Plasma measurements Mouse Glu-OC (undercarboxylated osteocalcin) and Gla-OC (carboxylated osteocalcin) levels were determined using ELISA kits according
Supplemental Data Supplemental Materials and Methods Plasma measurements Mouse Glu-OC (undercarboxylated osteocalcin) and Gla-OC (carboxylated osteocalcin) levels were determined using ELISA kits according
Metabolic ER stress and inflammation in white adipose tissue (WAT) of mice with dietary obesity.
 Supplementary Figure 1 Metabolic ER stress and inflammation in white adipose tissue (WAT) of mice with dietary obesity. Male C57BL/6J mice were fed a normal chow (NC, 10% fat) or a high-fat diet (HFD,
Supplementary Figure 1 Metabolic ER stress and inflammation in white adipose tissue (WAT) of mice with dietary obesity. Male C57BL/6J mice were fed a normal chow (NC, 10% fat) or a high-fat diet (HFD,
Supplementary Figure 1
 Supplementary Figure 1 how HFD how HFD Epi WT p p Hypothalamus p p Inguinal WT T Liver Lean mouse adipocytes p p p p p p Obese mouse adipocytes Kidney Muscle Spleen Heart p p p p p p p p Extracellular
Supplementary Figure 1 how HFD how HFD Epi WT p p Hypothalamus p p Inguinal WT T Liver Lean mouse adipocytes p p p p p p Obese mouse adipocytes Kidney Muscle Spleen Heart p p p p p p p p Extracellular
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION doi:10.1038/nature11464 Supplemental Figure S1. The expression of Vegfb is increased in obese and diabetic mice as compared to lean mice. a-b, Body weight and postprandial blood
SUPPLEMENTARY INFORMATION doi:10.1038/nature11464 Supplemental Figure S1. The expression of Vegfb is increased in obese and diabetic mice as compared to lean mice. a-b, Body weight and postprandial blood
Supplementary Figure 1
 VO (ml kg - min - ) VCO (ml kg - min - ) Respiratory exchange ratio Energy expenditure (cal kg - min - ) Locomotor activity (x count) Body temperature ( C) Relative mrna expression TA Sol EDL PT Heart
VO (ml kg - min - ) VCO (ml kg - min - ) Respiratory exchange ratio Energy expenditure (cal kg - min - ) Locomotor activity (x count) Body temperature ( C) Relative mrna expression TA Sol EDL PT Heart
Supplementary Table 2. Plasma lipid profiles in wild type and mutant female mice submitted to a HFD for 12 weeks wt ERα -/- AF-1 0 AF-2 0
 Supplementary Table 1. List of specific primers used for gene expression analysis. Genes Primer forward Primer reverse Hprt GCAGTACAGCCCCAAAATGG AACAAAGTCTGGCCTGTATCCA Srebp-1c GGAAGCTGTCGGGGTAGCGTC CATGTCTTCAAATGTGCAATCCAT
Supplementary Table 1. List of specific primers used for gene expression analysis. Genes Primer forward Primer reverse Hprt GCAGTACAGCCCCAAAATGG AACAAAGTCTGGCCTGTATCCA Srebp-1c GGAAGCTGTCGGGGTAGCGTC CATGTCTTCAAATGTGCAATCCAT
SUPPLEMENTARY INFORMATION
 -. -. SUPPLEMENTARY INFORMATION DOI: 1.1/ncb86 a WAT-1 WAT- BAT-1 BAT- sk-muscle-1 sk-muscle- mir-133b mir-133a mir-6 mir-378 mir-1 mir-85 mir-378 mir-6a mir-18 mir-133a mir- mir- mir-341 mir-196a mir-17
-. -. SUPPLEMENTARY INFORMATION DOI: 1.1/ncb86 a WAT-1 WAT- BAT-1 BAT- sk-muscle-1 sk-muscle- mir-133b mir-133a mir-6 mir-378 mir-1 mir-85 mir-378 mir-6a mir-18 mir-133a mir- mir- mir-341 mir-196a mir-17
Supplementary Table 1.
 Supplementary Table 1. Expression of genes involved in brown fat differentiation in WAT of db/db mice treated with HDAC inhibitors. Data are expressed as fold change (FC) versus control. symbol FC SAHA
Supplementary Table 1. Expression of genes involved in brown fat differentiation in WAT of db/db mice treated with HDAC inhibitors. Data are expressed as fold change (FC) versus control. symbol FC SAHA
Supplemental Fig. 1. Relative mrna Expression. Relative mrna Expression WT KO WT KO RT 4 0 C
 Supplemental Fig. 1 A 1.5 1..5 Hdac11 (ibat) n=4 n=4 n=4 n=4 n=4 n=4 n=4 n=4 WT KO WT KO WT KO WT KO RT 4 C RT 4 C Supplemental Figure 1. Hdac11 mrna is undetectable in KO adipose tissue. Quantitative
Supplemental Fig. 1 A 1.5 1..5 Hdac11 (ibat) n=4 n=4 n=4 n=4 n=4 n=4 n=4 n=4 WT KO WT KO WT KO WT KO RT 4 C RT 4 C Supplemental Figure 1. Hdac11 mrna is undetectable in KO adipose tissue. Quantitative
ab Adipogenesis Assay Kit (Cell-Based)
 ab133102 Adipogenesis Assay Kit (Cell-Based) Instructions for Use For the study of induction and inhibition of adipogenesis in adherent cells. This product is for research use only and is not intended
ab133102 Adipogenesis Assay Kit (Cell-Based) Instructions for Use For the study of induction and inhibition of adipogenesis in adherent cells. This product is for research use only and is not intended
Supplementary Table 1. Primer Sequences Used for Quantitative Real-Time PCR
 Supplementary Table 1. Primer Sequences Used for Quantitative Real-Time PCR Gene Forward Primer (5-3 ) Reverse Primer (5-3 ) cadl CTTGGGGGCGCGTCT CTGTTCTTTTGTGCCGTTTCG cyl-coenzyme Dehydrogenase, very
Supplementary Table 1. Primer Sequences Used for Quantitative Real-Time PCR Gene Forward Primer (5-3 ) Reverse Primer (5-3 ) cadl CTTGGGGGCGCGTCT CTGTTCTTTTGTGCCGTTTCG cyl-coenzyme Dehydrogenase, very
A microrna-34a/fgf21 Regulatory Axis and Browning of White Fat
 A microrna-34a/fgf21 Regulatory Axis and Browning of White Fat Jongsook Kim Kemper, Ph.D Department of Molecular and Integrative Physiology, University of Illinois at Urbana-Champaign, USA 213 International
A microrna-34a/fgf21 Regulatory Axis and Browning of White Fat Jongsook Kim Kemper, Ph.D Department of Molecular and Integrative Physiology, University of Illinois at Urbana-Champaign, USA 213 International
Supplemental Information. Intermittent Fasting Promotes. White Adipose Browning and Decreases Obesity. by Shaping the Gut Microbiota
 Cell Metabolism, Volume 26 Supplemental Information Intermittent Fasting Promotes White Adipose Browning and Decreases Obesity by Shaping the Gut Microbiota Guolin Li, Cen Xie, Siyu Lu, Robert G. Nichols,
Cell Metabolism, Volume 26 Supplemental Information Intermittent Fasting Promotes White Adipose Browning and Decreases Obesity by Shaping the Gut Microbiota Guolin Li, Cen Xie, Siyu Lu, Robert G. Nichols,
The Epigenetics of Obesity: Individual, Social, and Environmental Influences. K. J. Claycombe, Ph.D.
 The Epigenetics of Obesity: Individual, Social, and Environmental Influences K. J. Claycombe, Ph.D. What can happen to our gene(s) that would cause obesity? Modification via Epigenetic alterations C
The Epigenetics of Obesity: Individual, Social, and Environmental Influences K. J. Claycombe, Ph.D. What can happen to our gene(s) that would cause obesity? Modification via Epigenetic alterations C
SUPPLEMENTARY INFORMATION. Supplemental Figure 1. Body weight and blood glucose parameters of chow-diet (CD)
 SUPPLEMENTARY INFORMATION LEGENDS Supplemental Figure. Body weight and blood glucose parameters of chow-diet (CD) fed and high-fat diet (HFD) fed mice. (A) Body weight was measured at the beginning of
SUPPLEMENTARY INFORMATION LEGENDS Supplemental Figure. Body weight and blood glucose parameters of chow-diet (CD) fed and high-fat diet (HFD) fed mice. (A) Body weight was measured at the beginning of
Supplemental Information. Brown Adipogenic Reprogramming. Induced by a Small Molecule
 Cell Reports, Volume 18 Supplemental Information rown dipogenic Reprogramming Induced by a Small Molecule aoming Nie, Tao Nie, Xiaoyan Hui, Ping Gu, Liufeng Mao, Kuai Li, Ran Yuan, Jiashun Zheng, Haixia
Cell Reports, Volume 18 Supplemental Information rown dipogenic Reprogramming Induced by a Small Molecule aoming Nie, Tao Nie, Xiaoyan Hui, Ping Gu, Liufeng Mao, Kuai Li, Ran Yuan, Jiashun Zheng, Haixia
ENHANCEMENT OF BROWN ADIPOSE TISSUE DEVELOPMENT IN VIVO BY A NOVEL INSULIN SENSITIZER
 ENHANCEMENT F BRWN ADIPSE TISSUE DEVELPMENT IN VIV BY A NVEL INSULIN SENSITIZER William G. McDonald 1, Serena L. Cole 1, Brian N. Finck 2, Danielle D. Holewa 1, Angela S. Brightwell-Conrad 1, Charles Mackenzie
ENHANCEMENT F BRWN ADIPSE TISSUE DEVELPMENT IN VIV BY A NVEL INSULIN SENSITIZER William G. McDonald 1, Serena L. Cole 1, Brian N. Finck 2, Danielle D. Holewa 1, Angela S. Brightwell-Conrad 1, Charles Mackenzie
SUPPLEMENTARY DATA. Supplementary Table 1. Primers used in qpcr
 Supplementary Table 1. Primers used in qpcr Gene forward primer (5'-3') reverse primer (5'-3') β-actin AGAGGGAAATCGTGCGTGAC CAATAGTGATGACCTGGCCGT Hif-p4h-2 CTGGGCAACTACAGGATAAAC GCGTCCCAGTCTTTATTTAGATA
Supplementary Table 1. Primers used in qpcr Gene forward primer (5'-3') reverse primer (5'-3') β-actin AGAGGGAAATCGTGCGTGAC CAATAGTGATGACCTGGCCGT Hif-p4h-2 CTGGGCAACTACAGGATAAAC GCGTCCCAGTCTTTATTTAGATA
Oestrogen signalling in white adipose progenitor cells inhibits differentiation into brown adipose and smooth muscle cells
 Received Feb 4 Accepted 8 Sep 4 Published Oct 4 DOI:.8/ncomms696 Oestrogen signalling in white adipose progenitor cells inhibits differentiation into brown adipose and smooth muscle cells Kfir Lapid,,
Received Feb 4 Accepted 8 Sep 4 Published Oct 4 DOI:.8/ncomms696 Oestrogen signalling in white adipose progenitor cells inhibits differentiation into brown adipose and smooth muscle cells Kfir Lapid,,
Control 7 d cold 7 d CL
 Control 7 d cold 7 d ibat iwat gwat Supplementary Figure 1. Histology of adipose tissues after cold or 3-adrenergic receptor stimulation. C57BL/6J wild-type mice were housed at 4 C or injected daily with
Control 7 d cold 7 d ibat iwat gwat Supplementary Figure 1. Histology of adipose tissues after cold or 3-adrenergic receptor stimulation. C57BL/6J wild-type mice were housed at 4 C or injected daily with
SUPPLEMENTARY DATA. Nature Medicine: doi: /nm.4171
 SUPPLEMENTARY DATA Supplementary Figure 1 a b c PF %Change - -4-6 Body weight Lean mass Body fat Tissue weight (g).4.3.2.1. PF GC iwat awat BAT PF d e f g week 2 week 3 NEFA (mmol/l) 1..5. PF phsl (Ser565)
SUPPLEMENTARY DATA Supplementary Figure 1 a b c PF %Change - -4-6 Body weight Lean mass Body fat Tissue weight (g).4.3.2.1. PF GC iwat awat BAT PF d e f g week 2 week 3 NEFA (mmol/l) 1..5. PF phsl (Ser565)
Supplementary Table 1. The primers used for quantitative RT-PCR. Gene name Forward (5 > 3 ) Reverse (5 > 3 )
 770 771 Supplementary Table 1. The primers used for quantitative RT-PCR. Gene name Forward (5 > 3 ) Reverse (5 > 3 ) Human CXCL1 GCGCCCAAACCGAAGTCATA ATGGGGGATGCAGGATTGAG PF4 CCCCACTGCCCAACTGATAG TTCTTGTACAGCGGGGCTTG
770 771 Supplementary Table 1. The primers used for quantitative RT-PCR. Gene name Forward (5 > 3 ) Reverse (5 > 3 ) Human CXCL1 GCGCCCAAACCGAAGTCATA ATGGGGGATGCAGGATTGAG PF4 CCCCACTGCCCAACTGATAG TTCTTGTACAGCGGGGCTTG
Supplementary Fig. 1 eif6 +/- mice show a reduction in white adipose tissue, blood lipids and normal glycogen synthesis. The cohort of the original
 Supplementary Fig. 1 eif6 +/- mice show a reduction in white adipose tissue, blood lipids and normal glycogen synthesis. The cohort of the original phenotypic screening was n=40. For specific tests, the
Supplementary Fig. 1 eif6 +/- mice show a reduction in white adipose tissue, blood lipids and normal glycogen synthesis. The cohort of the original phenotypic screening was n=40. For specific tests, the
Interplay between FGF21 and insulin action in the liver regulates metabolism
 Research article Interplay between FGF21 and insulin action in the liver regulates metabolism Brice Emanuelli, 1 Sara G. Vienberg, 1 Graham Smyth, 1 Christine Cheng, 2 Kristin I. Stanford, 1 Manimozhiyan
Research article Interplay between FGF21 and insulin action in the liver regulates metabolism Brice Emanuelli, 1 Sara G. Vienberg, 1 Graham Smyth, 1 Christine Cheng, 2 Kristin I. Stanford, 1 Manimozhiyan
Supplementary Information
 Supplementary Information GADD34-deficient mice develop obesity, nonalcoholic fatty liver disease, hepatic carcinoma and insulin resistance Naomi Nishio and Ken-ichi Isobe Department of Immunology, Nagoya
Supplementary Information GADD34-deficient mice develop obesity, nonalcoholic fatty liver disease, hepatic carcinoma and insulin resistance Naomi Nishio and Ken-ichi Isobe Department of Immunology, Nagoya
University of California, San Diego La Jolla CA 92093
 AD Award Number: W81XWH-11-1-0131 TITLE: Role of Inflammation and Insulin Resistance in Mouse Models of Breast Cancer PRINCIPAL INVESTIGATOR: Jerrold Olefsky, M.D. CONTRACTING ORGANIZATION: University
AD Award Number: W81XWH-11-1-0131 TITLE: Role of Inflammation and Insulin Resistance in Mouse Models of Breast Cancer PRINCIPAL INVESTIGATOR: Jerrold Olefsky, M.D. CONTRACTING ORGANIZATION: University
A Central Role of MG53 in Metabolic Syndrome. and Type-2 Diabetes
 A Central Role of MG53 in Metabolic Syndrome and Type-2 Diabetes Yan Zhang, Chunmei Cao, Rui-Ping Xiao Institute of Molecular Medicine (IMM) Peking University, Beijing, China Accelerated Aging in China
A Central Role of MG53 in Metabolic Syndrome and Type-2 Diabetes Yan Zhang, Chunmei Cao, Rui-Ping Xiao Institute of Molecular Medicine (IMM) Peking University, Beijing, China Accelerated Aging in China
Molecular Medicine. Brown adipose tissue responds to cold and adrenergic
 Brown adipose tissue responds to cold and adrenergic stimulation by induction of FGF21 Dionysios V. Chartoumpekis, 1, Ioannis G. Habeos, 1, Panos G. Ziros, 1 Agathoklis I. Psyrogiannis, 1 Venetsana E.
Brown adipose tissue responds to cold and adrenergic stimulation by induction of FGF21 Dionysios V. Chartoumpekis, 1, Ioannis G. Habeos, 1, Panos G. Ziros, 1 Agathoklis I. Psyrogiannis, 1 Venetsana E.
Male 30. Female. Body weight (g) Age (weeks) Age (weeks) Atg7 f/f Atg7 ΔCD11c
 ody weight (g) ody weight (g) 34 3 Male 3 27 Female 26 24 22 18 7 9 11 13 15 17 19 21 23 21 18 15 7 9 11 13 15 17 19 21 23 Age (weeks) Age (weeks) Supplementary Figure 1. Lean phenotypes in mice regardless
ody weight (g) ody weight (g) 34 3 Male 3 27 Female 26 24 22 18 7 9 11 13 15 17 19 21 23 21 18 15 7 9 11 13 15 17 19 21 23 Age (weeks) Age (weeks) Supplementary Figure 1. Lean phenotypes in mice regardless
An introduction to the COCVD Metabolic Phenotyping Core
 An introduction to the COCVD Metabolic Phenotyping Core Capabilities and procedures Manager: Wendy S. Katz, Ph.D. University of Kentucky Medical Center Department of Pharmacology 577 Charles T. Wethington
An introduction to the COCVD Metabolic Phenotyping Core Capabilities and procedures Manager: Wendy S. Katz, Ph.D. University of Kentucky Medical Center Department of Pharmacology 577 Charles T. Wethington
Tissue factor-par2 signaling promotes diet-induced obesity and adipose
 Supplementary figures for Tissue factor-par2 signaling promotes diet-induced obesity and adipose inflammation. Leylla Badeanlou 1, Christian Furlan-Freguia 2, Guang Yang 1, Wolfram Ruf 2,3, and Fahumiya
Supplementary figures for Tissue factor-par2 signaling promotes diet-induced obesity and adipose inflammation. Leylla Badeanlou 1, Christian Furlan-Freguia 2, Guang Yang 1, Wolfram Ruf 2,3, and Fahumiya
Up-Regulation of Mitochondrial Activity and Acquirement of Brown Adipose Tissue-Like Property in the White Adipose Tissue of Fsp27 Deficient Mice
 Up-Regulation of Mitochondrial Activity and Acquirement of Brown Adipose Tissue-Like Property in the White Adipose Tissue of Fsp27 Deficient Mice Shen Yon Toh 1,2,3., Jingyi Gong 2., Guoli Du 2., John
Up-Regulation of Mitochondrial Activity and Acquirement of Brown Adipose Tissue-Like Property in the White Adipose Tissue of Fsp27 Deficient Mice Shen Yon Toh 1,2,3., Jingyi Gong 2., Guoli Du 2., John
Supplemental Table 1. Plasma NEFA and liver triglyceride levels in ap2-hif1ako and ap2-hif2ako mice under control and high fat diets.
 Supplemental Table 1. Plasma NEFA and liver triglyceride levels in Hif1aKO and Hif2aKO mice under control and high fat diets. Hif1a (n=6) Hif1aK O (n=6) Hif2a Hif2aK O Hif1a (n=5) Hif1aKO (n=5) Hif2a Hif2aK
Supplemental Table 1. Plasma NEFA and liver triglyceride levels in Hif1aKO and Hif2aKO mice under control and high fat diets. Hif1a (n=6) Hif1aK O (n=6) Hif2a Hif2aK O Hif1a (n=5) Hif1aKO (n=5) Hif2a Hif2aK
DOWNLOAD PDF ADIPOSE TISSUE AND ADIPOKINES IN HEALTH AND DISEASE (NUTRITION AND HEALTH)
 Chapter 1 : Adiposity, Adipokines, and Adiposopathy - Sick Fat Explained Adipose Tissue and Adipokines in Health and Disease, Second Edition is a useful resource for physicians interested in adipose tissue
Chapter 1 : Adiposity, Adipokines, and Adiposopathy - Sick Fat Explained Adipose Tissue and Adipokines in Health and Disease, Second Edition is a useful resource for physicians interested in adipose tissue
Mfn2 deletion in brown adipose tissue protects from insulin resistance and impairs thermogenesis
 Article Mfn2 deletion in brown adipose tissue protects from insulin resistance and impairs thermogenesis Kiana Mahdaviani 1,2, Ilan Y Benador 1,2, Shi Su 1, Raffi A Gharakhanian 1, Linsey Stiles 1,2, Kyle
Article Mfn2 deletion in brown adipose tissue protects from insulin resistance and impairs thermogenesis Kiana Mahdaviani 1,2, Ilan Y Benador 1,2, Shi Su 1, Raffi A Gharakhanian 1, Linsey Stiles 1,2, Kyle
Mice lacking the syndecan-3 gene are resistant to diet-induced obesity
 Research article Mice lacking the syndecan-3 gene are resistant to diet-induced obesity April D. Strader, 1 Ofer Reizes, 2 Stephen C. Woods, 1 Stephen C. Benoit, 1 and Randy J. Seeley 1 1 Department of
Research article Mice lacking the syndecan-3 gene are resistant to diet-induced obesity April D. Strader, 1 Ofer Reizes, 2 Stephen C. Woods, 1 Stephen C. Benoit, 1 and Randy J. Seeley 1 1 Department of
ALT (U/L) (Relative expression) HDL (mm) (Relative expression) ALT (U/L) (Relative expression)
 a DMT mrna () 8 6 r =.96 P =. DMT mrna () 8 6 r =. P =.6 DMT mrna () 8 6 r =.99 P =.6 DMT mrna () 8 6 r =. P =.9 DMT mrna () BMI (kg/m ) 8 6 r =.7 P =.966 DMT mrna () 8 ALT (U/L) 8 6 r = -.66 P =.76 DMT
a DMT mrna () 8 6 r =.96 P =. DMT mrna () 8 6 r =. P =.6 DMT mrna () 8 6 r =.99 P =.6 DMT mrna () 8 6 r =. P =.9 DMT mrna () BMI (kg/m ) 8 6 r =.7 P =.966 DMT mrna () 8 ALT (U/L) 8 6 r = -.66 P =.76 DMT
Metabolic Solutions Development Company, Kalamazoo, USA.
 New Insulin Sensitizers Produce Differentiation of Brown-like Adipose Cells from a Subcutaneous Fat Depot and Increase Secretion of Adiponectin in vitro William G. McDonald, Serena L. Cole, Danielle D.
New Insulin Sensitizers Produce Differentiation of Brown-like Adipose Cells from a Subcutaneous Fat Depot and Increase Secretion of Adiponectin in vitro William G. McDonald, Serena L. Cole, Danielle D.
ΦΛΕΓΜΟΝΗ ΚΑΙ ΔΙΑΒΗΤΗΣ
 ΦΛΕΓΜΟΝΗ ΚΑΙ ΔΙΑΒΗΤΗΣ ΘΩΜΑΣ ΠΑΠΑΔΟΠΟΥΛΟΣ, MD, PHD ΕΠΕΜΒΑΤΙΚΟΣ ΚΑΡΔΙΟΛΟΓΟΣ ΙΑΤΡΙΚΟ ΔΙΑΒΑΛΚΑΝΙΚΟ ΚΕΝΤΡΟ Inflammation as a cause of disease has entered the popular imagination. Diet ( macronutrients )
ΦΛΕΓΜΟΝΗ ΚΑΙ ΔΙΑΒΗΤΗΣ ΘΩΜΑΣ ΠΑΠΑΔΟΠΟΥΛΟΣ, MD, PHD ΕΠΕΜΒΑΤΙΚΟΣ ΚΑΡΔΙΟΛΟΓΟΣ ΙΑΤΡΙΚΟ ΔΙΑΒΑΛΚΑΝΙΚΟ ΚΕΝΤΡΟ Inflammation as a cause of disease has entered the popular imagination. Diet ( macronutrients )
Reviewer #1 (Remarks to the Author)
 Reviewer #1 (Remarks to the Author) The authors provide an interesting data set concerning the effects of an ATGL inhibitor on energy balance and indices of insulin action in mice fed a high fat diet.
Reviewer #1 (Remarks to the Author) The authors provide an interesting data set concerning the effects of an ATGL inhibitor on energy balance and indices of insulin action in mice fed a high fat diet.
Supplementary Figure S1. Effect of Glucose on Energy Balance in WT and KHK A/C KO
 Supplementary Figure S1. Effect of Glucose on Energy Balance in WT and KHK A/C KO Mice. WT mice and KHK-A/C KO mice were provided drinking water containing 10% glucose or tap water with normal chow ad
Supplementary Figure S1. Effect of Glucose on Energy Balance in WT and KHK A/C KO Mice. WT mice and KHK-A/C KO mice were provided drinking water containing 10% glucose or tap water with normal chow ad
Expression of acid base transporters in the kidney collecting duct in Slc2a7 -/-
 Supplemental Material Results. Expression of acid base transporters in the kidney collecting duct in Slc2a7 -/- and Slc2a7 -/- mice. The expression of AE1 in the kidney was examined in Slc26a7 KO mice.
Supplemental Material Results. Expression of acid base transporters in the kidney collecting duct in Slc2a7 -/- and Slc2a7 -/- mice. The expression of AE1 in the kidney was examined in Slc26a7 KO mice.
Id1 Promotes Obesity by Suppressing Brown Adipose Thermogenesis and White Adipose Browning
 Diabetes Volume 66, June 2017 1611 Id1 Promotes Obesity by Suppressing Brown Adipose Thermogenesis and White Adipose Browning Mallikarjun Patil, Bal Krishan Sharma, Sawsan Elattar, Judith Chang, Shweta
Diabetes Volume 66, June 2017 1611 Id1 Promotes Obesity by Suppressing Brown Adipose Thermogenesis and White Adipose Browning Mallikarjun Patil, Bal Krishan Sharma, Sawsan Elattar, Judith Chang, Shweta
SUPPLEMENTARY DATA. Supplementary Table 1. Primer sequences for qrt-pcr
 Supplementary Table 1. Primer sequences for qrt-pcr Gene PRDM16 UCP1 PGC1α Dio2 Elovl3 Cidea Cox8b PPARγ AP2 mttfam CyCs Nampt NRF1 16s-rRNA Hexokinase 2, intron 9 β-actin Primer Sequences 5'-CCA CCA GCG
Supplementary Table 1. Primer sequences for qrt-pcr Gene PRDM16 UCP1 PGC1α Dio2 Elovl3 Cidea Cox8b PPARγ AP2 mttfam CyCs Nampt NRF1 16s-rRNA Hexokinase 2, intron 9 β-actin Primer Sequences 5'-CCA CCA GCG
ANSC/NUTR 601 GENERAL ANIMAL NUTRITION Stearoyl-CoA desaturase, VLDL metabolism, and obesity
 ANSC/NUTR 601 GENERAL ANIMAL NUTRITION Stearoyl-CoA desaturase, VLDL metabolism, and obesity I. Stearoyl coenzyme A desaturase and obesity in rodents A. Stearoyl coenzyme A desaturase (SCD) is the 9 desaturase.
ANSC/NUTR 601 GENERAL ANIMAL NUTRITION Stearoyl-CoA desaturase, VLDL metabolism, and obesity I. Stearoyl coenzyme A desaturase and obesity in rodents A. Stearoyl coenzyme A desaturase (SCD) is the 9 desaturase.
In The Name Of God. In The Name Of. EMRI Modeling Group
 In The Name Of God In The Name Of God EMRI Modeling Group Cells work together in functionally related groups called tissues Types of tissues: Epithelial lining and covering Connective support Muscle movement
In The Name Of God In The Name Of God EMRI Modeling Group Cells work together in functionally related groups called tissues Types of tissues: Epithelial lining and covering Connective support Muscle movement
Supporting Information
 Supporting Information Charalambous et al. 10.1073/pnas.1406119111 SI Experimental Procedures Serum and Tissue Biochemistry. Enzymatic assay kits were used for determination of plasma FFAs (Roche), TAGs
Supporting Information Charalambous et al. 10.1073/pnas.1406119111 SI Experimental Procedures Serum and Tissue Biochemistry. Enzymatic assay kits were used for determination of plasma FFAs (Roche), TAGs
SUPPLEMENTARY INFORMATION
 DOI: 10.1038/ncb3461 In the format provided by the authors and unedited. Supplementary Figure 1 (associated to Figure 1). Cpeb4 gene-targeted mice develop liver steatosis. a, Immunoblot displaying CPEB4
DOI: 10.1038/ncb3461 In the format provided by the authors and unedited. Supplementary Figure 1 (associated to Figure 1). Cpeb4 gene-targeted mice develop liver steatosis. a, Immunoblot displaying CPEB4
Fig. S1. Dose-response effects of acute administration of the β3 adrenoceptor agonists CL316243, BRL37344, ICI215,001, ZD7114, ZD2079 and CGP12177 at
 Fig. S1. Dose-response effects of acute administration of the β3 adrenoceptor agonists CL316243, BRL37344, ICI215,001, ZD7114, ZD2079 and CGP12177 at doses of 0.1, 0.5 and 1 mg/kg on cumulative food intake
Fig. S1. Dose-response effects of acute administration of the β3 adrenoceptor agonists CL316243, BRL37344, ICI215,001, ZD7114, ZD2079 and CGP12177 at doses of 0.1, 0.5 and 1 mg/kg on cumulative food intake
Muscle-specific 4E-BP1 signaling activation improves metabolic parameters during aging and obesity
 Muscle-specific 4E-BP1 signaling activation improves metabolic parameters during aging and obesity Shihyin Tsai,, Albert R. La Spada, Brian K. Kennedy J Clin Invest. 2015;125(8):2952-2964. https://doi.org/10.1172/jci77361.
Muscle-specific 4E-BP1 signaling activation improves metabolic parameters during aging and obesity Shihyin Tsai,, Albert R. La Spada, Brian K. Kennedy J Clin Invest. 2015;125(8):2952-2964. https://doi.org/10.1172/jci77361.
Endocrine Disrupters as Obesogens
 Endocrine Disrupters as Obesogens Is the environment making us fat? Bruce Blumberg, Ph.D. Department of Developmental and Cell Biology Department of Pharmaceutical Sciences Developmental Biology Center
Endocrine Disrupters as Obesogens Is the environment making us fat? Bruce Blumberg, Ph.D. Department of Developmental and Cell Biology Department of Pharmaceutical Sciences Developmental Biology Center
FAT. It s Not All That! A Closer Look at the Two Main Types of Fat in Our Bodies: Visceral and Subcutaneous Fat
 Mary-Kate Perrone Capstone Seminar July 14, 2007 Draft #2 Fat Stats FAT. It s Not All That! A Closer Look at the Two Main Types of Fat in Our Bodies: Visceral and Subcutaneous Fat According to the 2003-2004
Mary-Kate Perrone Capstone Seminar July 14, 2007 Draft #2 Fat Stats FAT. It s Not All That! A Closer Look at the Two Main Types of Fat in Our Bodies: Visceral and Subcutaneous Fat According to the 2003-2004
Supplementary Information Titles Journal: Nature Medicine
 Supplementary Information Titles Journal: Nature Medicine Article Title: Corresponding Author: Supplementary Item & Number Supplementary Fig.1 Fig.2 Fig.3 Fig.4 Fig.5 Fig.6 Fig.7 Fig.8 Fig.9 Fig. Fig.11
Supplementary Information Titles Journal: Nature Medicine Article Title: Corresponding Author: Supplementary Item & Number Supplementary Fig.1 Fig.2 Fig.3 Fig.4 Fig.5 Fig.6 Fig.7 Fig.8 Fig.9 Fig. Fig.11
Nature Structural & Molecular Biology: doi: /nsmb Supplementary Figure 1. Generation and validation of mtef4-knockout mice.
 Supplementary Figure 1 Generation and validation of mtef4-knockout mice. (a) Alignment of EF4 (E. coli) with mouse, yeast and human EF4. (b) Domain structures of mouse mtef4 compared to those of EF4 (E.
Supplementary Figure 1 Generation and validation of mtef4-knockout mice. (a) Alignment of EF4 (E. coli) with mouse, yeast and human EF4. (b) Domain structures of mouse mtef4 compared to those of EF4 (E.
RNA interference induced hepatotoxicity results from loss of the first synthesized isoform of microrna-122 in mice
 SUPPLEMENTARY INFORMATION RNA interference induced hepatotoxicity results from loss of the first synthesized isoform of microrna-122 in mice Paul N Valdmanis, Shuo Gu, Kirk Chu, Lan Jin, Feijie Zhang,
SUPPLEMENTARY INFORMATION RNA interference induced hepatotoxicity results from loss of the first synthesized isoform of microrna-122 in mice Paul N Valdmanis, Shuo Gu, Kirk Chu, Lan Jin, Feijie Zhang,
(A) PCR primers (arrows) designed to distinguish wild type (P1+P2), targeted (P1+P2) and excised (P1+P3)14-
 1 Supplemental Figure Legends Figure S1. Mammary tumors of ErbB2 KI mice with 14-3-3σ ablation have elevated ErbB2 transcript levels and cell proliferation (A) PCR primers (arrows) designed to distinguish
1 Supplemental Figure Legends Figure S1. Mammary tumors of ErbB2 KI mice with 14-3-3σ ablation have elevated ErbB2 transcript levels and cell proliferation (A) PCR primers (arrows) designed to distinguish
Yiying Zhang, PhD Research Scientist. Research Summary:
 Yiying Zhang, PhD Research Scientist Research Summary: Address: Naomi Berrie Diabetes Center at Columbia University Medical Center Russ Berrie Medical Science Pavilion 1150 St. Nicholas Avenue New York,
Yiying Zhang, PhD Research Scientist Research Summary: Address: Naomi Berrie Diabetes Center at Columbia University Medical Center Russ Berrie Medical Science Pavilion 1150 St. Nicholas Avenue New York,
TBP (H) CACAGTGAATCTTGGTTGTAAACTTGA AAACCGCTTGGGATTATATTCG ANGPTL8 (H) CTGGGCCCTGCCTACCGAGA CCGATGCTGCTGTGCCACCA [1]
![TBP (H) CACAGTGAATCTTGGTTGTAAACTTGA AAACCGCTTGGGATTATATTCG ANGPTL8 (H) CTGGGCCCTGCCTACCGAGA CCGATGCTGCTGTGCCACCA [1] TBP (H) CACAGTGAATCTTGGTTGTAAACTTGA AAACCGCTTGGGATTATATTCG ANGPTL8 (H) CTGGGCCCTGCCTACCGAGA CCGATGCTGCTGTGCCACCA [1]](/thumbs/89/99233308.jpg) ESM Table 1. Immunoblot antibodies. Primary Supplier Dilution Antibody Akt Cell Signaling 1:1000 Technology Phosphorylated Cell Signaling 1:1000 Akt (Ser 473) Technology PKCε Cell Signaling 1:1000 Technology
ESM Table 1. Immunoblot antibodies. Primary Supplier Dilution Antibody Akt Cell Signaling 1:1000 Technology Phosphorylated Cell Signaling 1:1000 Akt (Ser 473) Technology PKCε Cell Signaling 1:1000 Technology
Molecular pathways linking metabolic inflammation and thermogenesis G. Solinas Summary
 Published in "" which should be cited to refer to this work. Molecular pathways linking metabolic inflammation and thermogenesis G. Solinas http://doc.rero.ch Laboratory of Metabolic Stress Biology, Division
Published in "" which should be cited to refer to this work. Molecular pathways linking metabolic inflammation and thermogenesis G. Solinas http://doc.rero.ch Laboratory of Metabolic Stress Biology, Division
Brown Adipose Tissue Responds to Cold and Adrenergic Stimulation by Induction of FGF21
 Brown Adipose Tissue Responds to Cold and Adrenergic Stimulation by Induction of FGF21 Dionysios V Chartoumpekis, 1* Ioannis G Habeos, 1* Panos G Ziros, 1 Agathoklis I Psyrogiannis, 1 Venetsana E Kyriazopoulou,
Brown Adipose Tissue Responds to Cold and Adrenergic Stimulation by Induction of FGF21 Dionysios V Chartoumpekis, 1* Ioannis G Habeos, 1* Panos G Ziros, 1 Agathoklis I Psyrogiannis, 1 Venetsana E Kyriazopoulou,
FSP27 contributes to efficient energy storage in murine white adipocytes by promoting the formation of unilocular lipid droplets
 Research article Related Commentary, page 2693 FSP27 contributes to efficient energy storage in murine white adipocytes by promoting the formation of unilocular lipid droplets Naonobu Nishino, 1 Yoshikazu
Research article Related Commentary, page 2693 FSP27 contributes to efficient energy storage in murine white adipocytes by promoting the formation of unilocular lipid droplets Naonobu Nishino, 1 Yoshikazu
Method of leptin dosing, strain, and group housing influence leptin sensitivity in high-fat-fed weanling mice
 Am J Physiol Regul Integr Comp Physiol 284: R87 R100, 2003; 10.1152/ajpregu.00431.2002. Method of leptin dosing, strain, and group housing influence leptin sensitivity in high-fat-fed weanling mice HEATHER
Am J Physiol Regul Integr Comp Physiol 284: R87 R100, 2003; 10.1152/ajpregu.00431.2002. Method of leptin dosing, strain, and group housing influence leptin sensitivity in high-fat-fed weanling mice HEATHER
Phenotypic characterization of adipose-specific VDR knockout mice
 Phenotypic characterization of adipose-specific VDR knockout mice An honors thesis presented to the Department of Biological Sciences University at Albany, State University of New York in partial fulfillment
Phenotypic characterization of adipose-specific VDR knockout mice An honors thesis presented to the Department of Biological Sciences University at Albany, State University of New York in partial fulfillment
Supplementary Figure 1
 Supplementary Figure 1 a Percent of body weight! (%) 4! 3! 1! Epididymal fat Subcutaneous fat Liver SD Percent of body weight! (%) ** 3! 1! SD Percent of body weight! (%) 6! 4! SD ** b Blood glucose (mg/dl)!
Supplementary Figure 1 a Percent of body weight! (%) 4! 3! 1! Epididymal fat Subcutaneous fat Liver SD Percent of body weight! (%) ** 3! 1! SD Percent of body weight! (%) 6! 4! SD ** b Blood glucose (mg/dl)!
Central injection of fibroblast growth factor 1 induces sustained remission of diabetic hyperglycemia in rodents
 Central injection of fibroblast growth factor 1 induces sustained remission of diabetic hyperglycemia in rodents Jarrad M Scarlett 1,,1, Jennifer M Rojas 1,1, Miles E Matsen 1, Karl J Kaiyala 3, Darko
Central injection of fibroblast growth factor 1 induces sustained remission of diabetic hyperglycemia in rodents Jarrad M Scarlett 1,,1, Jennifer M Rojas 1,1, Miles E Matsen 1, Karl J Kaiyala 3, Darko
Adipose Tissue as an Endocrine Organ. Abdel Moniem Ibrahim, MD Professor of Physiology Cairo University
 Adipose Tissue as an Endocrine Organ Abdel Moniem Ibrahim, MD Professor of Physiology Cairo University Functions of Adipose Tissue Adipose tissue expresses and secretes a variety of bioactive peptides,
Adipose Tissue as an Endocrine Organ Abdel Moniem Ibrahim, MD Professor of Physiology Cairo University Functions of Adipose Tissue Adipose tissue expresses and secretes a variety of bioactive peptides,
Osteopontin mediates obesity-induced adipose tissue macrophage infiltration and insulin resistance in mice
 Research article Osteopontin mediates obesity-induced adipose tissue macrophage infiltration and insulin resistance in mice Takashi Nomiyama, 1 Diego Perez-Tilve, 2 Daisuke Ogawa, 1 Florence Gizard, 1
Research article Osteopontin mediates obesity-induced adipose tissue macrophage infiltration and insulin resistance in mice Takashi Nomiyama, 1 Diego Perez-Tilve, 2 Daisuke Ogawa, 1 Florence Gizard, 1
Analysis of AVP functions via V1a and V1b receptors with knockout mice. Akito Tanoue
 Analysis of AVP functions via V1a and V1b receptors with knockout mice Akito Tanoue Department of Pharmacology, National Research Institute for Child Health and Development Arginine-Vasopressin (AVP) is
Analysis of AVP functions via V1a and V1b receptors with knockout mice Akito Tanoue Department of Pharmacology, National Research Institute for Child Health and Development Arginine-Vasopressin (AVP) is
Lack of TRPV2 impairs thermogenesis in mouse brown adipose tissue
 Manuscript EMBO-2015-40819 Lack of TRPV2 impairs thermogenesis in mouse brown adipose tissue Wuping Sun, Kunitoshi Uchida, Yoshiro Suzuki, Yiming Zhou, Minji Kim, Yasunori Takayama, Nobuyuki Takahashi,
Manuscript EMBO-2015-40819 Lack of TRPV2 impairs thermogenesis in mouse brown adipose tissue Wuping Sun, Kunitoshi Uchida, Yoshiro Suzuki, Yiming Zhou, Minji Kim, Yasunori Takayama, Nobuyuki Takahashi,
Expanded View Figures
 Expanded View Figures A B C D E F G H I J K L Figure EV1. The dysregulated lipid metabolic phenotype of mouse models of metabolic dysfunction is most pronounced in the fasted state. A L Male 12-weeks-old
Expanded View Figures A B C D E F G H I J K L Figure EV1. The dysregulated lipid metabolic phenotype of mouse models of metabolic dysfunction is most pronounced in the fasted state. A L Male 12-weeks-old
Supplementary Figures
 Supplementary Figures Supplementary Figure 1 Characterization of stable expression of GlucB and sshbira in the CT26 cell line (a) Live cell imaging of stable CT26 cells expressing green fluorescent protein
Supplementary Figures Supplementary Figure 1 Characterization of stable expression of GlucB and sshbira in the CT26 cell line (a) Live cell imaging of stable CT26 cells expressing green fluorescent protein
Mesenchymal progenitor cells in intramuscular connective tissue development
 Mesenchymal progenitor cells in intramuscular connective tissue development Min Du, Ph.D. Professor and Endowed Chair Department of Animal Sciences Washington State University Pullman, WA Beef Quality:
Mesenchymal progenitor cells in intramuscular connective tissue development Min Du, Ph.D. Professor and Endowed Chair Department of Animal Sciences Washington State University Pullman, WA Beef Quality:
(a) Significant biological processes (upper panel) and disease biomarkers (lower panel)
 Supplementary Figure 1. Functional enrichment analyses of secretomic proteins. (a) Significant biological processes (upper panel) and disease biomarkers (lower panel) 2 involved by hrab37-mediated secretory
Supplementary Figure 1. Functional enrichment analyses of secretomic proteins. (a) Significant biological processes (upper panel) and disease biomarkers (lower panel) 2 involved by hrab37-mediated secretory
HSN301 Diet and Disease Entire Note Summary
 Topic 1: Metabolic Syndrome Learning Objectives: 1. Define obesity 2. Factors causing obesity 3. Obesity as a risk factor for metabolic syndrome 4. The pathogenesis of metabolic syndrome 5. Treatment of
Topic 1: Metabolic Syndrome Learning Objectives: 1. Define obesity 2. Factors causing obesity 3. Obesity as a risk factor for metabolic syndrome 4. The pathogenesis of metabolic syndrome 5. Treatment of
University of Groningen. Non-alcoholic fatty liver disease Sheedfar, Fareeba
 University of Groningen Non-alcoholic fatty liver disease Sheedfar, Fareeba IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish to cite from it. Please check
University of Groningen Non-alcoholic fatty liver disease Sheedfar, Fareeba IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish to cite from it. Please check
Supplemental Information. FGF19, FGF21, and an FGFR1/b-Klotho-Activating. Antibody Act on the Nervous System. to Regulate Body Weight and Glycemia
 Cell Metabolism, Volume 26 Supplemental Information FGF19, FGF21, and an FGFR1/b-Klotho-Activating Antibody Act on the Nervous System to Regulate Body Weight and Glycemia Tian Lan, Donald A. Morgan, Kamal
Cell Metabolism, Volume 26 Supplemental Information FGF19, FGF21, and an FGFR1/b-Klotho-Activating Antibody Act on the Nervous System to Regulate Body Weight and Glycemia Tian Lan, Donald A. Morgan, Kamal
SUPPLEMENTARY DATA. Supplementary Table 1. Primers used for PCR and qpcr Primer Name
 Supplementary Table. Primers used for PCR and qpcr Primer Name ccession Number Fwd Rev Type of PCR Cre NC_8 GGCGTCTTCCGC GTGCCCCTCGTTTG Standard PCR LoUcp CCGGGCTGTCTCCGCGG GGCTGTTCGCCCGGCC Standard PCR
Supplementary Table. Primers used for PCR and qpcr Primer Name ccession Number Fwd Rev Type of PCR Cre NC_8 GGCGTCTTCCGC GTGCCCCTCGTTTG Standard PCR LoUcp CCGGGCTGTCTCCGCGG GGCTGTTCGCCCGGCC Standard PCR
Fructose Upregulates FGF23 Expression In MC3T3 Pre-osteoblasts
 Fructose Upregulates FGF23 Expression In MC3T3 Pre-osteoblasts Edek A. Williams, B.S.E., Veronique Douard, Ph.D., Joseph M. Lomuti, B.S., Ronaldo Ferraris, Ph.D., J. C. Fritton, Ph.D.. Rutgers University,
Fructose Upregulates FGF23 Expression In MC3T3 Pre-osteoblasts Edek A. Williams, B.S.E., Veronique Douard, Ph.D., Joseph M. Lomuti, B.S., Ronaldo Ferraris, Ph.D., J. C. Fritton, Ph.D.. Rutgers University,
Omega-3 Fatty Acids Mitigate Obesity-induced Osteoarthritis And Accelerate Wound Repair
 Omega-3 Fatty Acids Mitigate Obesity-induced Osteoarthritis And Accelerate Wound Repair Chia-Lung Wu, MS, Deeptee Jain, MD, Jenna McNeill, BS, Dianne Little, BVSc, PhD, John Anderson, MD, Janet Huebner,
Omega-3 Fatty Acids Mitigate Obesity-induced Osteoarthritis And Accelerate Wound Repair Chia-Lung Wu, MS, Deeptee Jain, MD, Jenna McNeill, BS, Dianne Little, BVSc, PhD, John Anderson, MD, Janet Huebner,
Obesity in aging: Hormonal contribution
 Obesity in aging: Hormonal contribution Hormonal issues in obesity and aging Hormonal role in regulation of energy balance Genetic component in hormonal regulation Life style contribution to hormonal changes
Obesity in aging: Hormonal contribution Hormonal issues in obesity and aging Hormonal role in regulation of energy balance Genetic component in hormonal regulation Life style contribution to hormonal changes
BMP6 treatment compensates for the molecular defect and ameliorates hemochromatosis in Hfe knockout mice
 SUPPLEMENTARY MATERIALS BMP6 treatment compensates for the molecular defect and ameliorates hemochromatosis in Hfe knockout mice Elena Corradini, Paul J. Schmidt, Delphine Meynard, Cinzia Garuti, Giuliana
SUPPLEMENTARY MATERIALS BMP6 treatment compensates for the molecular defect and ameliorates hemochromatosis in Hfe knockout mice Elena Corradini, Paul J. Schmidt, Delphine Meynard, Cinzia Garuti, Giuliana
