Analysis of the effect of tetrahydrobiopterin on PAH gene expression in hepatoma cells
|
|
|
- Stuart Black
- 5 years ago
- Views:
Transcription
1 Analysis of the effect of tetrahydrobiopterin on PAH gene expression in hepatoma cells Cristina Aguado, Belén Pérez, Magdalena Ugarte *, Lourdes R. Desviat Centro de Biología Molecular Severo Ochoa, CSIC-UAM, Universidad Autónoma de Madrid, Cantoblanco, 2849 Madrid, Spain Received 16 January 26; revised 3 February 26; accepted 7 February 26 Available online 17 February 26 Edited by Jesus Avila FEBS Letters 58 (26) Abstract Tetrahydrobiopterin (BH4)-responsive phenylalanine hydroxylase (PAH) deficiency is a recently recognized variant of phenylketonuria, with a probable multifactorial molecular basis. In this study we have investigated the effect of BH4 on PAH gene expression in human hepatoma. Our results show that increased BH4 levels result in an enhancement of PAH activity and PAH protein, due to longer turnover rates, while PAH mrna levels remain unchanged. This was confirmed for mutant PAH proteins (A39V, V388M and Y414C) associated to in vivo BH4 responsiveness, validating previous studies. We can conclude that there is no effect of the cofactor on PAH gene transcription, probably being the chemical chaperone effect of BH4 stabilizing mutant PAH proteins the major underlying mechanism of the response. Ó 26 Federation of European Biochemical Societies. Published by Elsevier B.V. All rights reserved. Keywords: Tetrahydrobiopterin; Phenylketonuria; PAH gene expression; BH4-responsiveness; PKU mutations; Chemical chaperon 1. Introduction Tetrahydrobiopterin ((6R)-L-erythro-5,6,7,8-tetrahydrobiopterin, BH 4 ) is the natural cofactor of phenylalanine hydroxylase (PAH), an hepatic enzyme that catalyzes the hydroxylation of L-Phe to L-Tyr and which is defective in the autosomal recessive disorder phenylketonuria (PKU) [1]. The accumulation of L-Phe in body fluids can be effectively avoided by classical dietary therapy preventing neurological damage. In addition, supplementation with high doses of the cofactor can be an effective therapy for a group of PKU patients [2]. Many studies conducted worldwide have recently demonstrated that there is a high proportion of patients (6 7%), who respond positively to a BH4 loading test [3 5] and several reports have begun to appear proving the efficacy of long-term treatment with BH4 [6 9]. Examination of the BH4-responsive patients genotypes shows a high genetic heterogeneity, although some mutations have been repeatedly associated to the response. In vitro expression data have shown that only a few of these mutations correspond to K M mutants having a slightly decreased affinity * Corresponding author. Fax: address: mugarte@cbm.uam.es (M. Ugarte). Abbreviations: BH4, (6R)-L-erythro-5,6,7,8-tetrahydrobiopterin; PAH, phenylalanine hydroxylase; PKU, phenylketonuria; DAHP, 2,4-diamino-6-hydroxypyrimidine for BH4, and for other mutants stabilization by BH4 was observed [1,11]. In vivo, in the liver of patients with BH4 deficiencies, reduced PAH activity and/or protein have been reported [12,13]. This already suggested that BH4 concentration may be an important regulatory factor in maintaining the steady-state levels of PAH. Similar results were also observed in transgenic mice deficient in cofactor biosynthesis [14,15]. In transgenic mice with a complete or partial deficiency in 6-pyruvoyltetrahydropterin synthase (PTPS), the rate of PAH activity and protein levels increased with BH4 content without affecting PAH mrna levels, apparently ruling out an effect on gene expression [15]. However, for inducible nitric oxide synthase (inos), which also requires BH4 as essential cofactor, a post-transcriptional stabilization of inos mrna by BH4 has been described in several cell types [16,17]. In this study we sought out to analyze the effect of BH4 levels on PAH gene expression in human hepatoma cells, where PAH and the enzymes responsible for the BH4 biosynthetic pathway are naturally expressed. Precursors and inhibitors of the intracellular synthesis pathway, which involve three enzymes (GTP cyclohydrolase I, GTPCH-1, PTPS and sepiapterin reductase, SR) [18,19], were used to modulate BH4 levels. The results of increasing BH4 levels in hepatoma cells discard an effect of the cofactor on PAH gene transcription, and confirm a chaperon-like effect of BH4 lowering PAH degradation rate. The stabilization effect was also demonstrated for three mutant PAH proteins associated to BH4 response and which were tagged with a FLAG epitope and transfected into hepatoma cells for measurement of protein steady state levels in the presence of increased levels of BH4. 2. Materials and methods 2.1. Materials Sepiapterin was obtained from Dr. B. Schircks Laboratories (Jona, Switzerland). Actinomycin D, puromycin, dicumarol and 2,4-diamino- 6-hydroxypyrimidine (DAHP) were from Sigma Aldrich. Cell culture medium and fetal calf serum (FCS) were from Gibco-BRL. Monoclonal antibody anti PAH (PH8) was from Pharmingen. Anti-FLAG M2 antibody was from Sigma. The secondary antibody (antimouse Ig conjugated with horseradish peroxidase) was from Santa Cruz. The Jetpei transfection reagent was obtained from Polyplus transfections Cell culture and transfection Human hepatoma cells Hep3B were cultured under standard conditions in minimum essential medium supplemented with 1% glutamine, 1%FCS and antibiotics. Cells were supplemented with 1 lm sepiapterin or 1 lm dicumarol or 1 mm DAHP for the times indicated. For transient expression of PAH-FLAG constructs, cells were plated in T-75 flasks and transfected with Jetpei following the /$32. Ó 26 Federation of European Biochemical Societies. Published by Elsevier B.V. All rights reserved. doi:1.116/j.febslet
2 1698 C. Aguado et al. / FEBS Letters 58 (26) manufacturer s recommendations. After 5 h incubation the Jetpei DNA mixture was removed and replaced with fresh medium supplemented or not with the various reagents. PAH-FLAG proteins were analyzed after 48 h Determination of intracellular biopterin Hep3B cells treated with the indicated agents were harvested by trypsinization and washed with phosphate-buffered saline. Pellets were resuspended in 1 mm Na-HEPES (ph 7.) and lysed by freeze thawing. After centrifugation to remove cell debris, the supernatant (total cell extract) was used for fluorimetric determination of total bioterin [3] Determination of PAH activity After treatment with the various reagents for 48 h, PAH activity in cell extracts obtained as described above was measured for 3 min at 25 C as conversion of L-Phe to L-Tyr [1] and after elimination of aminoacids using Ultrafree-MC 1 NMWL filters. The amount of L-Tyr formed was measured by HPLC and fluorimetric detection Protein analysis Wild-type PAH protein or transfected PAH-FLAG proteins in Hep3B cells were detected by Western blotting using commercial monoclonal antibodies (anti PAH Ph8 or anti-flag M2, respectively), anti-mouse-ig hoseradish peroxidase conjugate and the enhanced chemiluminiscence detection system ECL (Amersham). For determination of protein turnover, after treatment with the various reagents for 48 h, protein synthesis was stopped by addition of 1 lg/ml puromycin to the culture medium and cells were harvested at defined time points up to 1 h and analyzed by Western blotting. Relative protein amounts were determined by densitometric analysis and expressed as percentage of residual protein mrna analysis Total cellular RNA was isolated using the SV Total RNA Isolation Kit (Promega). For relative PAH mrna quantification, the Taqman Gene Expression assay Hs69359 (Applied Biosystems) was used. RNA was retrotranscribed using the Archive kit and PCR amplified with the Taqman Universal PCR Master Mix, all from Applied Biosystems. The real-time PCR and analysis were performed in an ABIP- RISM 79HT Genetic Analyzer. Amplification efficiency and sample to sample variation were normalised by monitoring glyceraldehyde phosphate dehydrogenase (GAPDH) mrna or 18S rrna Cloning into pflag vector PAH cdna was amplified from pmal-pah expression vector [2] using primers 5 -TTTTGCGGCCGCGATGTCCACTGCGGTCCT- GG-3 and 5 -AAAAGTCGACGGCTTTACTTTATTTTCTGGAG- 3, designed with SalI and NotI restriction sites at their 5 ends, respectively, to allow directed cloning into the pflag-cmv vector (Sigma). The in-frame fusion of amino-terminal FLAG peptide with the PAH protein was confirmed by sequencing performed with BigDye Terminator v.3.1 mix (Applied Biosystems) and analyzed by capillary electrophoresis on an ABI Prism 37 Genetic Analyzer (Applied Biosystems). Mutations were introduced into the wild-type construct using the Quickchange mutagenesis kit (Stratagene) and confirmed by sequencing analysis. 3. Results Human hepatoma cells were used as cellular model to determine the effect of BH4 on PAH gene expression. Sepiapterin was used as precursor of the intracellular BH4 biosynthetic pathway and dicumarol or DAHP as inhibitors of enzymes involved in BH4 synthesis (of SR and GTPCH-1, respectively) to reduce BH4 levels. Pterin analysis in Hep3B cells cultured in standard conditions revealed levels of 15.4 pmol/mg of total biopterin. After 48 h supplementation with 1 lm sepiapterin in the culture medium, biopterin levels increased to 461 ± 1633 pmol/mg, while treatment with dicumarol (1 lm) or DAHP (1 mm) reduced total biopterin to undetectable levels (Fig. 1A). A similar increase or decrease in biopterin levels after supplementation A 6 B biopterin (pmol/mg) PAH activity (nmol Tyr/min.mg) C 12 D 5 PAH mrna (%) PAH protein (%) Fig. 1. Effect of precursors and inhibitors of the BH4 biosynthetic pathway on: (A) biopterin content; (B) PAH activity; (C) PAH mrna and (D) PAH protein in hepatoma cells. After supplementation with sepiapterin (1 lm), dicumarol (1 lm) or DAHP (1 mm) for 48 h, total biopterin, PAH activity, PAH mrna and PAH protein content were measured in cellular extracts as described in Section 2. The data are the means ± S.D. of at least three experiments. SEP, sepiapterin; BH4, precursor; DIC, dicumarol; SR, inhibitor; DAHP, GTPCH-1 inhibitor.
3 C. Aguado et al. / FEBS Letters 58 (26) Control Sepiapterin % residual PAH protein DAHP T 1/2 (h) SEPIAPTERIN Control 9,4 DAHP Sepiapterin 21,6 CONTROL DAHP 1, Fig. 2. Analysis of PAH protein turnover in hepatoma cells. After incubation with sepiapterin for 48 h, translation was stopped with puromycin (1 lg/ml) and PAH protein in cells harvested up to 1 h was analysed by Western blot analysis (upper panel). The lower panel shows the representation of the results as a semilogarithmic plot of relative PAH protein obtained after densitometric analysis of Western blot autoradiograms. Shown is the mean of two independent experiments. The inner box shows the estimated half-lives of PAH protein in each condition. % PAH-FLAG protein sepiapterin + sepiapterin wt A39V V388M Y414C Fig. 3. Steady-state levels of wild-type and mutant PAH-FLAG proteins transiently expressed in hepatoma cells in the presence or not of sepiapterin as BH4 precursor. PAH-FLAG proteins were detected in cellular extracts using anti-flag antibodies and quantified by densitometric analysis. The data are the mean ± S.D. of at least three experiments. with sepiapterin or dicumarol/dahp, respectively, was confirmed in all subsequent experiments regarding PAH activity, protein or mrna. Supplementation with sepiapterin produced a 4-fold enhancement in PAH activity in hepatoma cells. Treatment with dicumarol abolished PAH activity completely while DAHP unexpectedly increased it (Fig. 1B). The potentiation of PAH activity with sepiapterin could be due to BH4 playing a regulatory role at the level of protein or mrna expression. To test this hypothesis, we first examined PAH mrna levels in hepatoma cells cultured in standard medium alone or supplemented for 48 h with sepiapterin, dicumarol or DAHP. Quantitation of PAH mrna levels revealed no significant differences between culture conditions, with only a slight reduction in mrna levels in dicumarol treated cells (Fig. 1C). However, direct observation of the cells with dicumarol before harvesting showed generalised cell death which could explain this result. The influence of BH4 on PAH activity observed in sepiapterin treated cells could thus be ascribed to the post-transcriptional level. We next examined PAH protein levels by Western blot analysis, which revealed that in sepiapterin-supplemented cells PAH protein was strongly augmented, correlating with the increase in PAH activity. In dicumarol treated cells no PAH was detectable, while with DAHP, PAH protein levels were increased correlating with the results obtained for PAH activity (Fig. 1D). To determine whether BH4 levels were affecting steady-state protein turnover and taking into account previous evidence of a stabilization effect of BH4 on PAH proteins [1,11,15], we measured PAH protein half-life in hepatoma after treatment with the different compounds. Protein synthesis was stopped with translation inhibitor puromycin (1 lg/ml) and protein amounts were determined by Western blot at defined time points up to 1 h (Fig. 2). PAH protein half-life in hepatoma was estimated at 9.4 h, in line with previous reports in an in vitro cell-free synthesis system [1,11]. Treatment with sepiapterin increased PAH half-life 3-fold, confirming that BH4 may exert a protective effect preventing degradation of PAH protein. To validate the results obtained previously in a cell-free synthesis system [1,11] and to provide a rationale for BH4 responsiveness in PKU, the effect of BH4 on previously studied mutant PAH proteins (A39V, V388M and Y414C) was reassessed in hepatoma. For this purpose, wild-type and mutant PAH constructs tagged with a FLAG epitope were engineered in the expression vector pflag-cmv. The A39V, V388M and Y414C mutations had been shown in an in vitro expression system to be stabilized by BH4 [1,11]. Transfection of the wild-type construct in hepatoma cells resulted in the expression of PAH-FLAG protein detected by Western blot analysis using anti-flag antibody, thus precluding the detection of endogenous PAH. Estimation of PAH-FLAG protein half-life was 9.4 h, the same as that obtained with endogenous PAH, showing that the FLAG peptide does not alter significantly the properties of the PAH protein. Transfection of the mutant constructs A39V, V388M and Y414C revealed that the steady-state amount of protein is somewhat reduced compared to wild-type (Fig. 3), in correlation with what had already been reported by expression analysis in COS cells, indicative of folding defects [2]. Although in this system we have to take into account the presence of endogenous PAH protein which could be forming oligomers with the transfected mutant proteins stabilising them, we sought to find out whether steady-state levels of mutant proteins were increased with sepiapterin. As shown in Fig. 3, in sepiapterin treated cells, the amount of all three mutant proteins is effectively increased although this effect is minimal for wild-type PAH-FLAG protein.
4 17 C. Aguado et al. / FEBS Letters 58 (26) Discussion Oral BH4 supplementation in responsive PKU patients is turning out to be a successful therapy, along with a progressive relaxation or complete withdrawal from dietary therapy [6 9]. A major issue in the understanding of BH4 responsiveness in PKU patients is the knowledge of the disease mechanism once the genetic defect is determined, providing useful information on which to base therapeutic strategies tailored to each patient. Experimental evidence has accumulated suggesting the molecular basis of the response is multifactorial [1,11,15]. A potential effect of BH4 on PAH gene expression was initially put forward, although results in transgenic mice deficient for cofactor biosynthesis do not support this hypothesis [15]. The data obtained in hepatoma concur with the reported observations. The mrna analysis rules out transcriptional regulation or mrna stabilization by BH4, and corroborates that PAH is stabilized by increased BH4 levels, resulting in a net increase in the amount of steady state levels of the enzyme and thus of the activity. It is not uncommon for protein stability to be enhanced by the binding of its cofactor [21 23] and in recombinant PAH, the binding of BH4 was reported to result in reduced proteolysis by trypsin [24]. With the present study, we cannot discard an effect of BH4 on translational regulation as has been described for vitamin B12, cofactor of methionine synthase [25,26]. However, the stabilization of the PAH protein by BH4 alone may account for the increase in steady-state levels of PAH protein observed. The results obtained in hepatoma cells with the inhibitors of the BH4 biosynthetic pathway are not straightforward. Dicumarol and DAHP have been used previously to investigate cytokine-induced nitric oxide production in endothelial and smooth muscle cells [16,17,27]. However, at 1 lm, dicumarol reduced drastically hepatoma cell viability and mrna analysis showed a large variability even for housekeeping genes such as GAPDH or 18S rrna. To circumvent this, we used DAHP which functions as a BH4 mimetic to engage feedback inhibition of GTPCH mediated by the GTPCH feedback regulatory protein (GFRP) [28]. At 1 mm, DAHP results in an unexpected increase in steady-state levels of PAH protein and thus of the relative activity, without affecting mrna levels or protein turnover. This suggests there could be some unforeseen effect maybe on translation which could account for the results. Three mutant proteins (A39V, V388M and Y414C) involved in the in vivo response and previously studied in the cell-free in vitro synthesis system TnT [1,11] have been analyzed in hepatoma, which provides a more physiological milieu correlating with the in vivo situation. The three mutant proteins tagged with a FLAG epitope were found to be stabilized by increased BH4 levels, confirming previous results. Wild-type protein did not increase substantially, similar to what was observed in the TNT system [1,11]. Differences in expression levels between endogenous PAH in hepatoma and PAH synthesized in vitro in the TNT system or in transient expression under a viral promoter may account for these results. The chaperon-like effect of BH4 preventing accelerated degradation is likely not limited to a few PAH proteins, as shown here, but represents a generalised effect on most missense folding PAH mutants explaining the high number of mutations associated to the in vivo response. As it has already been put forward, almost all responsive patients have at least one missense change resulting in a partially active protein and which represent mainly folding defects (excluding a few K M mutants) [29]. The present studies and those performed in transgenic mice [15,3] firmly support the notion that the stabilization of PAH proteins by BH4 is the major mechanism in vivo responsible for BH4 responsiveness in PKU patients. Acknowledgements: The authors thank A. Sánchez for expert technical assistance. This work received support from the Instituto de Salud Carlos III, Ministerio de Sanidad y Consumo (PI5/143, PI5/1413 and PI4571) and from Universidad Autónoma de Madrid-Comunidad Autónoma de Madrid (11/BCB/5). The institutional grant from Fundación Ramón Areces to Centro de Biología Molecular is gratefully acknowledged. References [1] Scriver, C.R. and Kaufman, S. (21) Hyperphenylalaninemia: phenylalanine hydroxylase deficiency in: The Metabolic and Molecular Bases of Inherited Disease (Scriver, C.R., Beaudet, D., Valle, D. and Sly, W.S., Eds.), pp , McGraw-Hill. [2] Blau, N. and Erlandsen, H. (24) The metabolic and molecular bases of tetrahydrobiopterin-responsive phenylalanine hydroxylase deficiency. Mol. Genet. Metab. 82, [3] Desviat, L.R., Perez, B., Belanger-Quintana, A., Castro, M., Aguado, C., Sanchez, A., Garcia, M.J., Martinez-Pardo, M. and Ugarte, M. (24) Tetrahydrobiopterin responsiveness: results of the BH4 loading test in 31 Spanish PKU patients and correlation with their genotype. Mol. Genet. Metab. 83, [4] Muntau, A.C., Roschinger, W., Habich, M., Demmelmair, H., Hoffmann, B., Sommerhoff, C.P. and Roscher, A.A. (22) Tetrahydrobiopterin as an alternative treatment for mild phenylketonuria. New Engl. J. Med. 347, [5] Spaapen, L.J. and Estela Rubio-Gozalbo, M. (23) Tetrahydrobiopterin-responsive phenylalanine hydroxylase deficiency, state of the art. Mol. Genet. Metab. 78, [6] Shintaku, H., Kure, S., Ohura, T., Okano, Y., Ohwada, M., Sugiyama, N., Sakura, N., Yoshida, I., Yoshino, M., Matsubara, Y., Suzuki, K., Aoki, K. and Kitagawa, T. (24) Long-term treatment and diagnosis of tetrahydrobiopterin-responsive hyperphenylalaninemia with a mutant phenylalanine hydroxylase gene. Pediatr. Res. 55, [7] Trefz, F.K., Scheible, D., Frauendienst-Egger, G., Korall, H. and Blau, N. (25) Long-term treatment of patients with mild and classical phenylketonuria by tetrahydrobiopterin. Mol. Genet. Metab. [8] Belanger-Quintana, A., Garcia, M.J., Castro, M., Desviat, L.R., Perez, B., Mejia, B., Ugarte, M. and Martinez-Pardo, M. (25) Spanish BH4-responsive phenylalanine hydroxylase-deficient patients: evolution of seven patients on long-term treatment with tetrahydrobiopterin. Mol. Genet. Metab. 86, S61 S66. [9] Hennermann, J.B., Buhrer, C., Blau, N., Vetter, B. and Monch, E. (25) Long-term treatment with tetrahydrobiopterin increases phenylalanine tolerance in children with severe phenotype of phenylketonuria. Mol. Genet. Metab. 86, S86 S9. [1] Pey, A.L., Perez, B., Desviat, L.R., Martinez, M.A., Aguado, C., Erlandsen, H., Gamez, A., Stevens, R.C., Thorolfsson, M., Ugarte, M. and Martinez, A. (24) Mechanisms underlying responsiveness to tetrahydrobiopterin in mild phenylketonuria mutations. Hum. Mutat. 24, [11] Erlandsen, H., Pey, A.L., Gamez, A., Perez, B., Desviat, L.R., Aguado, C., Koch, R., Surendran, S., Tyring, S., Matalon, R., Scriver, C.R., Ugarte, M., Martinez, A. and Stevens, R.C. (24) Correction of kinetic and stability defects by tetrahydrobiopterin in phenylketonuria patients with certain phenylalanine hydroxylase mutations. Proc. Natl. Acad. Sci. USA 11, [12] Dhondt, J.L. (1991) Strategy for the screening of tetrahydrobiopterin deficiency among hyperphenylalaninaemic patients: 15- years experience. J. Inherit. Metab. Dis. 14, [13] Kaufman, S., Holtzman, N.A., Milstien, S., Butler, L.J. and Krumholz, A. (1975) Phenylketonuria due to a deficiency of
5 C. Aguado et al. / FEBS Letters 58 (26) dihydropteridine reductase. New Engl. J. Med. 293, [14] Hyland, K., Gunasekera, R.S., Engle, T. and Arnold, L.A. (1996) Tetrahydrobiopterin and biogenic amine metabolism in the hph-1 mouse. J. Neurochem. 67, [15] Thony, B., Ding, Z. and Martinez, A. (24) Tetrahydrobiopterin protects phenylalanine hydroxylase activity in vivo: implications for tetrahydrobiopterin-responsive hyperphenylalaninemia. FEBS Lett. 577, [16] Saura, M., Perez-Sala, D., Canada, F.J. and Lamas, S. (1996) Role of tetrahydrobiopterin availability in the regulation of nitricoxide synthase expression in human mesangial cells. J. Biol. Chem. 271, [17] Linscheid, P., Schaffner, A. and Schoedon, G. (1998) Modulation of inducible nitric oxide synthase mrna stability by tetrahydrobiopterin in vascular smooth muscle cells. Biochem. Biophys. Res. Commun. 243, [18] Blau, N., Thony, B., Cotton, G.H. and Hyland, K. (21) Disorders of tetrahydrobioterin and related biogenic amines in: The Metabolic and Molecular Basis of Inherited Disease (Scriver, A.L., Beaudet, A.L., Sly, W.S., Valle, D., Childs, B. and Vogelstein, B., Eds.), pp , McGraw-Hill, New York. [19] Thony, B., Auerbach, G. and Blau, N. (2) Tetrahydrobiopterin biosynthesis, regeneration and functions. Biochem. J. 347 (Pt 1), [2] Pey, A.L., Desviat, L.R., Gamez, A., Ugarte, M. and Perez, B. (23) Phenylketonuria: genotype phenotype correlations based on expression analysis of structural and functional mutations in PAH. Hum. Mutat. 21, [21] Schnell, J.R., Dyson, H.J. and Wright, P.E. (24) Effect of cofactor binding and loop conformation on side chain methyl dynamics in dihydrofolate reductase. Biochemistry 43, [22] Bettati, S., Benci, S., Campanini, B., Raboni, S., Chirico, G., Beretta, S., Schnackerz, K.D., Hazlett, T.L., Gratton, E. and Mozzarelli, A. (2) Role of pyridoxal 5 -phosphate in the structural stabilization of O-acetylserine sulfhydrylase. J. Biol. Chem. 275, [23] Edmondson, D.E. and Newton-Vinson, P. (21) The covalent FAD of monoamine oxidase: structural and functional role and mechanism of the flavinylation reaction. Antioxid. Redox. Signal. 3, [24] Solstad, T., Stokka, A.J., Andersen, O.A. and Flatmark, T. (23) Studies on the regulatory properties of the pterin cofactor and dopamine bound at the active site of human phenylalanine hydroxylase. Eur. J. Biochem. 27, [25] Oltean, S. and Banerjee, R. (23) Nutritional modulation of gene expression and homocysteine utilization by vitamin B12. J. Biol. Chem. 278, [26] Oltean, S. and Banerjee, R. (25) A B12-responsive internal ribosome entry site (IRES) element in human methionine synthase. J. Biol. Chem. 28, [27] Schoedon, G., Blau, N., Schneemann, M., Flury, G. and Schaffner, A. (1994) Nitric oxide production depends on preceding tetrahydrobiopterin synthesis by endothelial cells: selective suppression of induced nitric oxide production by sepiapterin reductase inhibitors. Biochem. Biophys. Res. Commun. 199, [28] Kolinsky, M.A. and Gross, S.S. (24) The mechanism of potent GTP cyclohydrolase I inhibition by 2,4-diamino-6-hydroxypyrimidine: requirement of the GTP cyclohydrolase I feedback regulatory protein. J. Biol. Chem. 279, [29] Perez, B., Desviat, L.R., Gomez-Puertas, P., Martinez, A., Stevens, R.C. and Ugarte, M. (25) Kinetic and stability analysis of PKU mutations identified in BH4-responsive patients. Mol. Genet. Metab. 86, S11 S16. [3] Scavelli, R., Ding, Z., Blau, N., Haavik, J., Martinez, A. and Thony, B. (25) Stimulation of hepatic phenylalanine hydroxylase activity but not Pah-mRNA expression upon oral loading of tetrahydrobiopterin in normal mice. Mol. Genet. Metab. 86, S153 S155.
Extended tetrahydrobiopterin loading test in the diagnosis of cofactor-responsive phenylketonuria: A pilot study
 Molecular Genetics and Metabolism 86 (2005) S1 S5 www.elsevier.com/locate/ymgme Extended tetrahydrobiopterin loading test in the diagnosis of cofactor-responsive phenylketonuria: A pilot study Betina Fiege
Molecular Genetics and Metabolism 86 (2005) S1 S5 www.elsevier.com/locate/ymgme Extended tetrahydrobiopterin loading test in the diagnosis of cofactor-responsive phenylketonuria: A pilot study Betina Fiege
Tetrahydrobiopterin responsiveness: results of the BH 4 loading test in 31 Spanish PKU patients and correlation with their genotype
 Molecular Genetics and Metabolism 83 (2004) 157 162 www.elsevier.com/locate/ymgme Tetrahydrobiopterin responsiveness: results of the BH 4 loading test in 31 Spanish PKU patients and correlation with their
Molecular Genetics and Metabolism 83 (2004) 157 162 www.elsevier.com/locate/ymgme Tetrahydrobiopterin responsiveness: results of the BH 4 loading test in 31 Spanish PKU patients and correlation with their
Kinetic and stability analysis of PKU mutations identiwed in BH 4 -responsive patients
 Molecular Genetics and Metabolism 86 (2005) S11 S16 Minireview www.elsevier.com/locate/ymgme Kinetic and stability analysis of PKU mutations identiwed in BH 4 -responsive patients Belén Pérez a, Lourdes
Molecular Genetics and Metabolism 86 (2005) S11 S16 Minireview www.elsevier.com/locate/ymgme Kinetic and stability analysis of PKU mutations identiwed in BH 4 -responsive patients Belén Pérez a, Lourdes
In vitro expression analysis of R68G and R68S mutations in phenylalanine hydroxylase gene
 Vol. 47 No. 2/2000 365 369 QUARTERLY In vitro expression analysis of R68G and R68S mutations in phenylalanine hydroxylase gene Cezary ekanowski 1, Belen Perez 2, Lourdes R. Desviat 2, Wojciech Wiszniewski
Vol. 47 No. 2/2000 365 369 QUARTERLY In vitro expression analysis of R68G and R68S mutations in phenylalanine hydroxylase gene Cezary ekanowski 1, Belen Perez 2, Lourdes R. Desviat 2, Wojciech Wiszniewski
Phenylketonuria (PKU) the Biochemical Basis. Biol 405 Molecular Medicine
 Phenylketonuria (PKU) the Biochemical Basis Biol 405 Molecular Medicine PKU a history In 1934 Følling identified a clinical condition - imbecillitas phenylpyruvica. Mental retardation associated with this
Phenylketonuria (PKU) the Biochemical Basis Biol 405 Molecular Medicine PKU a history In 1934 Følling identified a clinical condition - imbecillitas phenylpyruvica. Mental retardation associated with this
Tetrahydrobiopterin responsiveness in patients with phenylketonuria
 Clinical Biochemistry 37 (2004) 1083 1090 Tetrahydrobiopterin responsiveness in patients with phenylketonuria Belén Pérez-Dueñas a, Maria Antonia Vilaseca b, *, Anna Mas c, Nilo Lambruschini d, Rafael
Clinical Biochemistry 37 (2004) 1083 1090 Tetrahydrobiopterin responsiveness in patients with phenylketonuria Belén Pérez-Dueñas a, Maria Antonia Vilaseca b, *, Anna Mas c, Nilo Lambruschini d, Rafael
Excerpt from J. Mol. Biol. (2002) 320, :
 Excerpt from J. Mol. Biol. (2002) 320, 1095 1108: Crystal Structure of the Ternary Complex of the Catalytic Domain of Human Phenylalanine Hydroxylase with Tetrahydrobiopterin and 3-(2-Thienyl)-L-alanine,
Excerpt from J. Mol. Biol. (2002) 320, 1095 1108: Crystal Structure of the Ternary Complex of the Catalytic Domain of Human Phenylalanine Hydroxylase with Tetrahydrobiopterin and 3-(2-Thienyl)-L-alanine,
Background and proposal for a pilot EQA scheme for pterins
 Background and proposal for a pilot EQA scheme for pterins enad Blau Division of Inborn Metabolic Diseases University Children's Hospital Heidelberg Germany Tetrahydrobiopterin (BH 4 ) Brimstone butterfly
Background and proposal for a pilot EQA scheme for pterins enad Blau Division of Inborn Metabolic Diseases University Children's Hospital Heidelberg Germany Tetrahydrobiopterin (BH 4 ) Brimstone butterfly
The neonatal tetrahydrobiopterin loading test in phenylketonuria: what is the predictive value?
 Anjema et al. Orphanet Journal of Rare Diseases (2016) 11:10 DOI 10.1186/s13023-016-0394-2 RESEARCH Open Access The neonatal tetrahydrobiopterin loading test in phenylketonuria: what is the predictive
Anjema et al. Orphanet Journal of Rare Diseases (2016) 11:10 DOI 10.1186/s13023-016-0394-2 RESEARCH Open Access The neonatal tetrahydrobiopterin loading test in phenylketonuria: what is the predictive
Analysis of Neurotransmitters. Simon Heales
 Analysis of Neurotransmitters Simon Heales HealeS@gosh.nhs.uk Tyrosine O 2 L-Dopa Dopamine HVA Tryptophan 5-HTP PLP Serotonin 5-HIAA Phenylalanine Tyrosine BH4 qbh2 BH2 CSF Sample Requirements Tube 1 Tube
Analysis of Neurotransmitters Simon Heales HealeS@gosh.nhs.uk Tyrosine O 2 L-Dopa Dopamine HVA Tryptophan 5-HTP PLP Serotonin 5-HIAA Phenylalanine Tyrosine BH4 qbh2 BH2 CSF Sample Requirements Tube 1 Tube
INVESTIGATION THE PREVALENCE OF MUTATIONS IVS 10 AND R158Q IN A NUMBER OF IRANIAN PATIENTS WITH PKU
 : 293-297 ISSN: 2277 4998 INVESTIGATION THE PREVALENCE OF MUTATIONS IVS 10 AND R158Q IN A NUMBER OF IRANIAN PATIENTS WITH PKU SHIRIN JAHANBAZI, FATEMEHKESHAVARZI* Department of Biology, Sanandaj Branch,
: 293-297 ISSN: 2277 4998 INVESTIGATION THE PREVALENCE OF MUTATIONS IVS 10 AND R158Q IN A NUMBER OF IRANIAN PATIENTS WITH PKU SHIRIN JAHANBAZI, FATEMEHKESHAVARZI* Department of Biology, Sanandaj Branch,
PKU, KUVAN, and Your Child Making smart choices for PKU treatment
 PKU, KUVAN, and Your Child Making smart choices for PKU treatment An educational brochure brought to you by BioMarin Pharmaceutical Inc. + = Low Low- diet Indication KUVAN (sapropterin dihydrochloride)
PKU, KUVAN, and Your Child Making smart choices for PKU treatment An educational brochure brought to you by BioMarin Pharmaceutical Inc. + = Low Low- diet Indication KUVAN (sapropterin dihydrochloride)
The New England Journal of Medicine TETRAHYDROBIOPTERIN AS AN ALTERNATIVE TREATMENT FOR MILD PHENYLKETONURIA
 TETRAHYDROBIOPTERIN AS AN ALTERNATIVE TREATMENT FOR MILD PHENYLKETONURIA ANIA C. MUNTAU, M.D., WULF RÖSCHINGER, M.D., MATTHIAS HABICH, HANS DEMMELMAIR, PH.D., BJÖRN HOFFMANN, CHRISTIAN P. SOMMERHOFF, M.D.,
TETRAHYDROBIOPTERIN AS AN ALTERNATIVE TREATMENT FOR MILD PHENYLKETONURIA ANIA C. MUNTAU, M.D., WULF RÖSCHINGER, M.D., MATTHIAS HABICH, HANS DEMMELMAIR, PH.D., BJÖRN HOFFMANN, CHRISTIAN P. SOMMERHOFF, M.D.,
New treatment options in PKU and future role of dietary treatment
 New treatment options in PKU and future role of dietary treatment Michael Staudigl (physician), Katharina Dokoupil (nutritionist), Esther M. Maier (physician, head of department) Dr. von Hauner Children
New treatment options in PKU and future role of dietary treatment Michael Staudigl (physician), Katharina Dokoupil (nutritionist), Esther M. Maier (physician, head of department) Dr. von Hauner Children
Recent Studies of Phenylketonuria By: Jennifer Gastelum Dr. Koni Stone Copyright 2014
 Recent Studies of Phenylketonuria By: Jennifer Gastelum Dr. Koni Stone Copyright 2014 Phenylketonuria (PKU) is an autosomal recessive genetic disorder that causes an accumulation of toxic metabolites in
Recent Studies of Phenylketonuria By: Jennifer Gastelum Dr. Koni Stone Copyright 2014 Phenylketonuria (PKU) is an autosomal recessive genetic disorder that causes an accumulation of toxic metabolites in
Identification and functional analysis of. mutations of the phenylalanine. hydroxylase gene in patients with. phenylketonuria
 Identification and functional analysis of mutations of the phenylalanine hydroxylase gene in patients with phenylketonuria Sang-Wun Kim Department of Medicine The Graduate School, Yonsei University Identification
Identification and functional analysis of mutations of the phenylalanine hydroxylase gene in patients with phenylketonuria Sang-Wun Kim Department of Medicine The Graduate School, Yonsei University Identification
Mechanisms Underlying Responsiveness to Tetrahydrobiopterin in Mild Phenylketonuria Mutations
 HUMAN MUTATION 24:388^399 (2004) RESEARCH ARTICLE Mechanisms Underlying Responsiveness to Tetrahydrobiopterin in Mild Phenylketonuria Mutations Angel L. Pey, 1 Belén Pérez, 1 Lourdes R. Desviat, 1 M a
HUMAN MUTATION 24:388^399 (2004) RESEARCH ARTICLE Mechanisms Underlying Responsiveness to Tetrahydrobiopterin in Mild Phenylketonuria Mutations Angel L. Pey, 1 Belén Pérez, 1 Lourdes R. Desviat, 1 M a
Phenylketonuria Jonathan Baghdadi and Evan Marlin
 Phenylketonuria Jonathan Baghdadi and Evan Marlin Hyperphenylalaninemia (HPA) was first connected to certain types of mental retardation in 1934 by Asjborn Folling. Just a few years later it was understood
Phenylketonuria Jonathan Baghdadi and Evan Marlin Hyperphenylalaninemia (HPA) was first connected to certain types of mental retardation in 1934 by Asjborn Folling. Just a few years later it was understood
University of Zurich. Zurich Open Repository and Archive
 University of Zurich Zurich Open Repository and Archive Winterthurerstr. 190 CH-8057 Zurich http://www.zora.uzh.ch Year: 2011 The interplay between genotype, metabolic state and cofactor treatment governs
University of Zurich Zurich Open Repository and Archive Winterthurerstr. 190 CH-8057 Zurich http://www.zora.uzh.ch Year: 2011 The interplay between genotype, metabolic state and cofactor treatment governs
Prerequisites Amino acid synthesis and degradation pathways. Integration of amino acid metabolic pathways with carbohydrate metabolic pathways.
 Case 30 Phenylketonuria Focus concept The characteristics of phenylalanine hydroxylase, the enzyme missing in persons afflicted with the genetic disorder phenylketonuria (PKU), are examined. Prerequisites
Case 30 Phenylketonuria Focus concept The characteristics of phenylalanine hydroxylase, the enzyme missing in persons afflicted with the genetic disorder phenylketonuria (PKU), are examined. Prerequisites
Phenylketonuria and Its Variants
 ANNALS OF CLINICAL AND LABORATORY SCIENCE, Vol. 7, No. 2 Copyright 1977, Institute for Clinical Science Phenylketonuria and Its Variants SEYMOUR KAUFMAN, Ph.D. and SHELDON MILSTIEN Ph.D. Laboratory of
ANNALS OF CLINICAL AND LABORATORY SCIENCE, Vol. 7, No. 2 Copyright 1977, Institute for Clinical Science Phenylketonuria and Its Variants SEYMOUR KAUFMAN, Ph.D. and SHELDON MILSTIEN Ph.D. Laboratory of
Genetic and phenotypic aspects of phenylalanine hydroxylase deficiency in Spain: molecular survey by regions
 European Journal of Human Genetics (1999) 7, 386 392 t 1999 Stockton Press All rights reserved 1018 4813/99 $12.00 http://www.stockton-press.co.uk/ejhg ARTICLE Genetic and phenotypic aspects of phenylalanine
European Journal of Human Genetics (1999) 7, 386 392 t 1999 Stockton Press All rights reserved 1018 4813/99 $12.00 http://www.stockton-press.co.uk/ejhg ARTICLE Genetic and phenotypic aspects of phenylalanine
Supplementary Fig. 1. GPRC5A post-transcriptionally down-regulates EGFR expression. (a) Plot of the changes in steady state mrna levels versus
 Supplementary Fig. 1. GPRC5A post-transcriptionally down-regulates EGFR expression. (a) Plot of the changes in steady state mrna levels versus changes in corresponding proteins between wild type and Gprc5a-/-
Supplementary Fig. 1. GPRC5A post-transcriptionally down-regulates EGFR expression. (a) Plot of the changes in steady state mrna levels versus changes in corresponding proteins between wild type and Gprc5a-/-
Construction of a hepatocellular carcinoma cell line that stably expresses stathmin with a Ser25 phosphorylation site mutation
 Construction of a hepatocellular carcinoma cell line that stably expresses stathmin with a Ser25 phosphorylation site mutation J. Du 1, Z.H. Tao 2, J. Li 2, Y.K. Liu 3 and L. Gan 2 1 Department of Chemistry,
Construction of a hepatocellular carcinoma cell line that stably expresses stathmin with a Ser25 phosphorylation site mutation J. Du 1, Z.H. Tao 2, J. Li 2, Y.K. Liu 3 and L. Gan 2 1 Department of Chemistry,
Supplementary Information Titles Journal: Nature Medicine
 Supplementary Information Titles Journal: Nature Medicine Article Title: Corresponding Author: Supplementary Item & Number Supplementary Fig.1 Fig.2 Fig.3 Fig.4 Fig.5 Fig.6 Fig.7 Fig.8 Fig.9 Fig. Fig.11
Supplementary Information Titles Journal: Nature Medicine Article Title: Corresponding Author: Supplementary Item & Number Supplementary Fig.1 Fig.2 Fig.3 Fig.4 Fig.5 Fig.6 Fig.7 Fig.8 Fig.9 Fig. Fig.11
Plasma tetrahydrobiopterin and its pharmacokinetic following oral administration
 Molecular Genetics and Metabolism 81 (24) 45 51 www.elsevier.com/locate/ymgme Plasma tetrahydrobiopterin and its pharmacokinetic following oral administration Betina Fiege, a,1 Diana Ballhausen, b Lucja
Molecular Genetics and Metabolism 81 (24) 45 51 www.elsevier.com/locate/ymgme Plasma tetrahydrobiopterin and its pharmacokinetic following oral administration Betina Fiege, a,1 Diana Ballhausen, b Lucja
Author's personal copy
 Molecular Genetics and Metabolism 10 (2011) 116 121 Contents lists available at ScienceDirect Molecular Genetics and Metabolism journal homepage: www.elsevier.com/locate/ymgme Molecular genetics and impact
Molecular Genetics and Metabolism 10 (2011) 116 121 Contents lists available at ScienceDirect Molecular Genetics and Metabolism journal homepage: www.elsevier.com/locate/ymgme Molecular genetics and impact
Materials and Methods , The two-hybrid principle.
 The enzymatic activity of an unknown protein which cleaves the phosphodiester bond between the tyrosine residue of a viral protein and the 5 terminus of the picornavirus RNA Introduction Every day there
The enzymatic activity of an unknown protein which cleaves the phosphodiester bond between the tyrosine residue of a viral protein and the 5 terminus of the picornavirus RNA Introduction Every day there
The molecular basis of phenylketonuria in Koreans
 J Hum Genet (2004) 49:617 621 DOI 10.1007/s10038-004-0197-5 ORIGINAL ARTICLE Dong Hwan Lee Æ Soo Kyung Koo Æ Kwang-Soo Lee Young-Joo Yeon Æ Hyun-Jeong Oh Æ Sang-Wun Kim Sook-Jin Lee Æ Sung-Soo Kim Æ Jong-Eun
J Hum Genet (2004) 49:617 621 DOI 10.1007/s10038-004-0197-5 ORIGINAL ARTICLE Dong Hwan Lee Æ Soo Kyung Koo Æ Kwang-Soo Lee Young-Joo Yeon Æ Hyun-Jeong Oh Æ Sang-Wun Kim Sook-Jin Lee Æ Sung-Soo Kim Æ Jong-Eun
number Done by Corrected by Doctor
 number 33 Done by Omar Sami Corrected by Waseem abu obeida Doctor Diala Too late for second guessing, too late to go back to sleep 1 P age Just try defying gravity This sheet will be divided to two parts,
number 33 Done by Omar Sami Corrected by Waseem abu obeida Doctor Diala Too late for second guessing, too late to go back to sleep 1 P age Just try defying gravity This sheet will be divided to two parts,
Neurotransmitter Disorders.
 Neurotransmitter Disorders Simon.heales@gosh.nhs.uk Chemical Neurotransmission Neurotransmitters Substances that upon release from nerve terminals, act on receptor sites at postsynaptic membranes to produce
Neurotransmitter Disorders Simon.heales@gosh.nhs.uk Chemical Neurotransmission Neurotransmitters Substances that upon release from nerve terminals, act on receptor sites at postsynaptic membranes to produce
Supplementary Data 1. Alanine substitutions and position variants of APNCYGNIPL. Applied in
 Supplementary Data 1. Alanine substitutions and position variants of APNCYGNIPL. Applied in Supplementary Fig. 2 Substitution Sequence Position variant Sequence original APNCYGNIPL original APNCYGNIPL
Supplementary Data 1. Alanine substitutions and position variants of APNCYGNIPL. Applied in Supplementary Fig. 2 Substitution Sequence Position variant Sequence original APNCYGNIPL original APNCYGNIPL
Sapropterin dihydrochloride treatment in Turkish hyperphenylalaninemic patients under age four
 The Turkish Journal of Pediatrics 2015; 57: 213-218 Original Sapropterin dihydrochloride treatment in Turkish hyperphenylalaninemic patients under age four Özlem Ünal 1, Hülya Gökmen-Özel 2, Turgay Coşkun
The Turkish Journal of Pediatrics 2015; 57: 213-218 Original Sapropterin dihydrochloride treatment in Turkish hyperphenylalaninemic patients under age four Özlem Ünal 1, Hülya Gökmen-Özel 2, Turgay Coşkun
Supplementary Appendix
 Supplementary Appendix This appendix has been provided by the authors to give readers additional information about their work. Supplement to: Choi YL, Soda M, Yamashita Y, et al. EML4-ALK mutations in
Supplementary Appendix This appendix has been provided by the authors to give readers additional information about their work. Supplement to: Choi YL, Soda M, Yamashita Y, et al. EML4-ALK mutations in
AMERICAN ACADEMY OF PEDIATRICS. New Developments in Hyperphenylalaninemia. Committee on Nutrition
 AMERICAN ACADEMY OF PEDIATRICS Committee on Nutrition New Developments in Hyperphenylalaninemia In recent years it has become apparent that hyperphenylalaninemia in newborn infants may be caused by a variety
AMERICAN ACADEMY OF PEDIATRICS Committee on Nutrition New Developments in Hyperphenylalaninemia In recent years it has become apparent that hyperphenylalaninemia in newborn infants may be caused by a variety
Supplementary information
 Supplementary information Human Cytomegalovirus MicroRNA mir-us4-1 Inhibits CD8 + T Cell Response by Targeting ERAP1 Sungchul Kim, Sanghyun Lee, Jinwook Shin, Youngkyun Kim, Irini Evnouchidou, Donghyun
Supplementary information Human Cytomegalovirus MicroRNA mir-us4-1 Inhibits CD8 + T Cell Response by Targeting ERAP1 Sungchul Kim, Sanghyun Lee, Jinwook Shin, Youngkyun Kim, Irini Evnouchidou, Donghyun
Supplementary Material
 Supplementary Material Nuclear import of purified HIV-1 Integrase. Integrase remains associated to the RTC throughout the infection process until provirus integration occurs and is therefore one likely
Supplementary Material Nuclear import of purified HIV-1 Integrase. Integrase remains associated to the RTC throughout the infection process until provirus integration occurs and is therefore one likely
Figure S1 Time-dependent down-modulation of HER3 by EZN No Treatment. EZN-3920, 2 μm. Time, h
 Figure S1 Time-dependent down-modulation of HER3 by EZN-392 HE ER3 mrna A, %Contr rol 12 No Treatment EZN-392, 2 μm 1 8 6 4 2 2 8 24 Time, h Figure S2. Specific target down-modulation by HER3 (EZN-392)
Figure S1 Time-dependent down-modulation of HER3 by EZN-392 HE ER3 mrna A, %Contr rol 12 No Treatment EZN-392, 2 μm 1 8 6 4 2 2 8 24 Time, h Figure S2. Specific target down-modulation by HER3 (EZN-392)
Luminescent platforms for monitoring changes in the solubility of amylin and huntingtin in living cells
 Electronic Supplementary Material (ESI) for Molecular BioSystems. This journal is The Royal Society of Chemistry 2016 Contents Supporting Information Luminescent platforms for monitoring changes in the
Electronic Supplementary Material (ESI) for Molecular BioSystems. This journal is The Royal Society of Chemistry 2016 Contents Supporting Information Luminescent platforms for monitoring changes in the
HEK293FT cells were transiently transfected with reporters, N3-ICD construct and
 Supplementary Information Luciferase reporter assay HEK293FT cells were transiently transfected with reporters, N3-ICD construct and increased amounts of wild type or kinase inactive EGFR. Transfections
Supplementary Information Luciferase reporter assay HEK293FT cells were transiently transfected with reporters, N3-ICD construct and increased amounts of wild type or kinase inactive EGFR. Transfections
Islet viability assay and Glucose Stimulated Insulin Secretion assay RT-PCR and Western Blot
 Islet viability assay and Glucose Stimulated Insulin Secretion assay Islet cell viability was determined by colorimetric (3-(4,5-dimethylthiazol-2-yl)-2,5- diphenyltetrazolium bromide assay using CellTiter
Islet viability assay and Glucose Stimulated Insulin Secretion assay Islet cell viability was determined by colorimetric (3-(4,5-dimethylthiazol-2-yl)-2,5- diphenyltetrazolium bromide assay using CellTiter
Molecular Biology (BIOL 4320) Exam #2 May 3, 2004
 Molecular Biology (BIOL 4320) Exam #2 May 3, 2004 Name SS# This exam is worth a total of 100 points. The number of points each question is worth is shown in parentheses after the question number. Good
Molecular Biology (BIOL 4320) Exam #2 May 3, 2004 Name SS# This exam is worth a total of 100 points. The number of points each question is worth is shown in parentheses after the question number. Good
Revision Log. See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Kuvan) Reference Number: CP. PHAR.43 Effective Date: 02.01.10 Last Review Date: 02.18 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
Clinical Policy: (Kuvan) Reference Number: CP. PHAR.43 Effective Date: 02.01.10 Last Review Date: 02.18 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
Amino acid Catabolism
 Enzymatic digestion of dietary proteins in gastrointestinal-tract. Amino acid Catabolism Amino acids: 1. There are 20 different amino acid, they are monomeric constituents of proteins 2. They act as precursors
Enzymatic digestion of dietary proteins in gastrointestinal-tract. Amino acid Catabolism Amino acids: 1. There are 20 different amino acid, they are monomeric constituents of proteins 2. They act as precursors
The What, Why and How of Large Neutral Amino Acids
 The What, Why and How of Large Neutral Amino Acids Kathryn Moseley, MS, RD Assistant Professor of Pediatrics USC/Keck School of Medicine LAC+USC Medical Center December 5 th, 2017 The opinions reflected
The What, Why and How of Large Neutral Amino Acids Kathryn Moseley, MS, RD Assistant Professor of Pediatrics USC/Keck School of Medicine LAC+USC Medical Center December 5 th, 2017 The opinions reflected
General Laboratory methods Plasma analysis: Gene Expression Analysis: Immunoblot analysis: Immunohistochemistry:
 General Laboratory methods Plasma analysis: Plasma insulin (Mercodia, Sweden), leptin (duoset, R&D Systems Europe, Abingdon, United Kingdom), IL-6, TNFα and adiponectin levels (Quantikine kits, R&D Systems
General Laboratory methods Plasma analysis: Plasma insulin (Mercodia, Sweden), leptin (duoset, R&D Systems Europe, Abingdon, United Kingdom), IL-6, TNFα and adiponectin levels (Quantikine kits, R&D Systems
UNCORRECTED PROOF ORIGINAL ARTICLE. Steven F Dobrowolski 1, K Borski 2, CE Ellingson 1, R Koch 3, HL Levy 4 and EW Naylor 5
 (29), 5 & 29 The Japan Society of Human Genetics All rights reserved 434-56/9 $32. www.nature.com/jhg ORIGINAL ARTICLE A limited spectrum of phenylalanine hydroxylase mutations is observed in phenylketonuria
(29), 5 & 29 The Japan Society of Human Genetics All rights reserved 434-56/9 $32. www.nature.com/jhg ORIGINAL ARTICLE A limited spectrum of phenylalanine hydroxylase mutations is observed in phenylketonuria
Phosphate buffered saline (PBS) for washing the cells TE buffer (nuclease-free) ph 7.5 for use with the PrimePCR Reverse Transcription Control Assay
 Catalog # Description 172-5080 SingleShot Cell Lysis Kit, 100 x 50 µl reactions 172-5081 SingleShot Cell Lysis Kit, 500 x 50 µl reactions For research purposes only. Introduction The SingleShot Cell Lysis
Catalog # Description 172-5080 SingleShot Cell Lysis Kit, 100 x 50 µl reactions 172-5081 SingleShot Cell Lysis Kit, 500 x 50 µl reactions For research purposes only. Introduction The SingleShot Cell Lysis
Supplementary Figure 1.TRIM33 binds β-catenin in the nucleus. a & b, Co-IP of endogenous TRIM33 with β-catenin in HT-29 cells (a) and HEK 293T cells
 Supplementary Figure 1.TRIM33 binds β-catenin in the nucleus. a & b, Co-IP of endogenous TRIM33 with β-catenin in HT-29 cells (a) and HEK 293T cells (b). TRIM33 was immunoprecipitated, and the amount of
Supplementary Figure 1.TRIM33 binds β-catenin in the nucleus. a & b, Co-IP of endogenous TRIM33 with β-catenin in HT-29 cells (a) and HEK 293T cells (b). TRIM33 was immunoprecipitated, and the amount of
MicroRNA sponges: competitive inhibitors of small RNAs in mammalian cells
 MicroRNA sponges: competitive inhibitors of small RNAs in mammalian cells Margaret S Ebert, Joel R Neilson & Phillip A Sharp Supplementary figures and text: Supplementary Figure 1. Effect of sponges on
MicroRNA sponges: competitive inhibitors of small RNAs in mammalian cells Margaret S Ebert, Joel R Neilson & Phillip A Sharp Supplementary figures and text: Supplementary Figure 1. Effect of sponges on
SUPPLEMENTARY INFORMATION. Supplementary Figures S1-S9. Supplementary Methods
 SUPPLEMENTARY INFORMATION SUMO1 modification of PTEN regulates tumorigenesis by controlling its association with the plasma membrane Jian Huang 1,2#, Jie Yan 1,2#, Jian Zhang 3#, Shiguo Zhu 1, Yanli Wang
SUPPLEMENTARY INFORMATION SUMO1 modification of PTEN regulates tumorigenesis by controlling its association with the plasma membrane Jian Huang 1,2#, Jie Yan 1,2#, Jian Zhang 3#, Shiguo Zhu 1, Yanli Wang
Phenylketonuria (PKU) is an inherited metabolic disorder involving a group of genetic mutations that reduce the ability
 Tetrahydrobiopterin Therapy for Phenylketonuria in Infants and Young Children Barbara K. Burton, MD, Darius J. Adams, MD, Dorothy K. Grange, MD, John I. Malone, MD, Elaina Jurecki, MS, RD, Heather Bausell,
Tetrahydrobiopterin Therapy for Phenylketonuria in Infants and Young Children Barbara K. Burton, MD, Darius J. Adams, MD, Dorothy K. Grange, MD, John I. Malone, MD, Elaina Jurecki, MS, RD, Heather Bausell,
BIOL 158: BIOLOGICAL CHEMISTRY II
 BIOL 158: BIOLOGICAL CHEMISTRY II Lecture 5: Vitamins and Coenzymes Lecturer: Christopher Larbie, PhD Introduction Cofactors bind to the active site and assist in the reaction mechanism Apoenzyme is an
BIOL 158: BIOLOGICAL CHEMISTRY II Lecture 5: Vitamins and Coenzymes Lecturer: Christopher Larbie, PhD Introduction Cofactors bind to the active site and assist in the reaction mechanism Apoenzyme is an
Supplemental Materials and Methods Plasmids and viruses Quantitative Reverse Transcription PCR Generation of molecular standard for quantitative PCR
 Supplemental Materials and Methods Plasmids and viruses To generate pseudotyped viruses, the previously described recombinant plasmids pnl4-3-δnef-gfp or pnl4-3-δ6-drgfp and a vector expressing HIV-1 X4
Supplemental Materials and Methods Plasmids and viruses To generate pseudotyped viruses, the previously described recombinant plasmids pnl4-3-δnef-gfp or pnl4-3-δ6-drgfp and a vector expressing HIV-1 X4
Supplementary Figure 1
 Supplementary Figure 1 Asymmetrical function of 5p and 3p arms of mir-181 and mir-30 families and mir-142 and mir-154. (a) Control experiments using mirna sensor vector and empty pri-mirna overexpression
Supplementary Figure 1 Asymmetrical function of 5p and 3p arms of mir-181 and mir-30 families and mir-142 and mir-154. (a) Control experiments using mirna sensor vector and empty pri-mirna overexpression
Efficacy and safety of BH4 before the age of 4 years in patients with mild phenylketonuria
 DOI 10.1007/s10545-012-9464-3 ORIGINAL ARTICLE Efficacy and safety of BH4 before the age of 4 years in patients with mild phenylketonuria Oriane Leuret & Magalie Barth & Alice Kuster & Didier Eyer & Loïc
DOI 10.1007/s10545-012-9464-3 ORIGINAL ARTICLE Efficacy and safety of BH4 before the age of 4 years in patients with mild phenylketonuria Oriane Leuret & Magalie Barth & Alice Kuster & Didier Eyer & Loïc
PROTEIN METABOLISM: SPECIFIC WAYS OF AMINO ACIDS CATABOLISM AND SYNTHESIS
 PROTEIN METABOLISM: SPECIFIC WAYS OF AMINO ACIDS CATABOLISM AND SYNTHESIS SPECIFIC WAYS OF AMINO ACID CATABOLISM After removing of amino group the carbon skeletons of amino acids are transformed into metabolic
PROTEIN METABOLISM: SPECIFIC WAYS OF AMINO ACIDS CATABOLISM AND SYNTHESIS SPECIFIC WAYS OF AMINO ACID CATABOLISM After removing of amino group the carbon skeletons of amino acids are transformed into metabolic
Genetics of monoamine neurotransmitter disorders
 Review Article Genetics of monoamine neurotransmitter disorders Wai-Kwan Siu Kowloon West Cluster Laboratory Genetic Service, Department of Pathology, Princess Margaret Hospital, Hong Kong SAR, China Correspondence
Review Article Genetics of monoamine neurotransmitter disorders Wai-Kwan Siu Kowloon West Cluster Laboratory Genetic Service, Department of Pathology, Princess Margaret Hospital, Hong Kong SAR, China Correspondence
Molecular Genetics and Metabolism
 Molecular Genetics and Metabolism 96 (2009) 158 163 Contents lists available at ScienceDirect Molecular Genetics and Metabolism journal homepage: www.elsevier.com/locate/ymgme Minireview Optimizing the
Molecular Genetics and Metabolism 96 (2009) 158 163 Contents lists available at ScienceDirect Molecular Genetics and Metabolism journal homepage: www.elsevier.com/locate/ymgme Minireview Optimizing the
A model of human phenylalanine metabolism in normal subjects and in phenylketonuric patients
 Proc. Natl. Acad. Sci. USA Vol. 96, pp. 3160 3164, March 1999 Medical Sciences A model of human phenylalanine metabolism in normal subjects and in phenylketonuric patients SEYMOUR KAUFMAN* Laboratory of
Proc. Natl. Acad. Sci. USA Vol. 96, pp. 3160 3164, March 1999 Medical Sciences A model of human phenylalanine metabolism in normal subjects and in phenylketonuric patients SEYMOUR KAUFMAN* Laboratory of
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION FOR Liver X Receptor α mediates hepatic triglyceride accumulation through upregulation of G0/G1 Switch Gene 2 (G0S2) expression I: SUPPLEMENTARY METHODS II: SUPPLEMENTARY FIGURES
SUPPLEMENTARY INFORMATION FOR Liver X Receptor α mediates hepatic triglyceride accumulation through upregulation of G0/G1 Switch Gene 2 (G0S2) expression I: SUPPLEMENTARY METHODS II: SUPPLEMENTARY FIGURES
Pre-mRNA has introns The splicing complex recognizes semiconserved sequences
 Adding a 5 cap Lecture 4 mrna splicing and protein synthesis Another day in the life of a gene. Pre-mRNA has introns The splicing complex recognizes semiconserved sequences Introns are removed by a process
Adding a 5 cap Lecture 4 mrna splicing and protein synthesis Another day in the life of a gene. Pre-mRNA has introns The splicing complex recognizes semiconserved sequences Introns are removed by a process
TETRAHYDROBIOPTERIN AND GTP CYCLOHYDROLASE I IN A RAT MODEL OF ENDOTOXIC SHOCK: RELATION TO NITRIC OXIDE SYNTHESIS
 Experimental Physiology (1996), 81, 665-671 Printed in Great Britain TETRAHYDROBIOPTERIN AND GTP CYCLOHYDROLASE I IN A RAT MODEL OF ENDOTOXIC SHOCK: RELATION TO NITRIC OXIDE SYNTHESIS Y. HATTORI, N. NAKANISHI*,
Experimental Physiology (1996), 81, 665-671 Printed in Great Britain TETRAHYDROBIOPTERIN AND GTP CYCLOHYDROLASE I IN A RAT MODEL OF ENDOTOXIC SHOCK: RELATION TO NITRIC OXIDE SYNTHESIS Y. HATTORI, N. NAKANISHI*,
Tel: ; Fax: ;
 Tel.: +98 216 696 9291; Fax: +98 216 696 9291; E-mail: mrasadeghi@pasteur.ac.ir Tel: +98 916 113 7679; Fax: +98 613 333 6380; E-mail: abakhshi_e@ajums.ac.ir A Soluble Chromatin-bound MOI 0 1 5 0 1 5 HDAC2
Tel.: +98 216 696 9291; Fax: +98 216 696 9291; E-mail: mrasadeghi@pasteur.ac.ir Tel: +98 916 113 7679; Fax: +98 613 333 6380; E-mail: abakhshi_e@ajums.ac.ir A Soluble Chromatin-bound MOI 0 1 5 0 1 5 HDAC2
Merck KGaA, Darmstadt, Germany, Receives EU-Approval to Extend Kuvan Use to Children with PKU Below 4 Years of Age
 Your Contact News Release Gangolf Schrimpf +49 6151 72-9591 Investor Relations +49 6151 72-3321 July 20, 2015 Merck KGaA, Darmstadt, Germany, Receives EU-Approval to Extend Kuvan Use to Children with PKU
Your Contact News Release Gangolf Schrimpf +49 6151 72-9591 Investor Relations +49 6151 72-3321 July 20, 2015 Merck KGaA, Darmstadt, Germany, Receives EU-Approval to Extend Kuvan Use to Children with PKU
Tetrahydrobiopterin in Cardiovascular Health and Disease
 ANTIOXIDANTS & REDOX SIGNALING Volume 20, Number 18, 2014 ª Mary Ann Liebert, Inc. DOI: 10.1089/ars.2013.5566 COMPREHENSIVE INVITED REVIEW Tetrahydrobiopterin in Cardiovascular Health and Disease Jennifer
ANTIOXIDANTS & REDOX SIGNALING Volume 20, Number 18, 2014 ª Mary Ann Liebert, Inc. DOI: 10.1089/ars.2013.5566 COMPREHENSIVE INVITED REVIEW Tetrahydrobiopterin in Cardiovascular Health and Disease Jennifer
Cell isolation. Spleen and lymph nodes (axillary, inguinal) were removed from mice
 Supplementary Methods: Cell isolation. Spleen and lymph nodes (axillary, inguinal) were removed from mice and gently meshed in DMEM containing 10% FBS to prepare for single cell suspensions. CD4 + CD25
Supplementary Methods: Cell isolation. Spleen and lymph nodes (axillary, inguinal) were removed from mice and gently meshed in DMEM containing 10% FBS to prepare for single cell suspensions. CD4 + CD25
Supplementary data Supplementary Figure 1 Supplementary Figure 2
 Supplementary data Supplementary Figure 1 SPHK1 sirna increases RANKL-induced osteoclastogenesis in RAW264.7 cell culture. (A) RAW264.7 cells were transfected with oligocassettes containing SPHK1 sirna
Supplementary data Supplementary Figure 1 SPHK1 sirna increases RANKL-induced osteoclastogenesis in RAW264.7 cell culture. (A) RAW264.7 cells were transfected with oligocassettes containing SPHK1 sirna
Protocol for Gene Transfection & Western Blotting
 The schedule and the manual of basic techniques for cell culture Advanced Protocol for Gene Transfection & Western Blotting Schedule Day 1 26/07/2008 Transfection Day 3 28/07/2008 Cell lysis Immunoprecipitation
The schedule and the manual of basic techniques for cell culture Advanced Protocol for Gene Transfection & Western Blotting Schedule Day 1 26/07/2008 Transfection Day 3 28/07/2008 Cell lysis Immunoprecipitation
Supplementary appendix
 Supplementary appendix This appendix formed part of the original submission and has been peer reviewed. We post it as supplied by the authors. Supplement to: van Spronsen FJ, van Wegberg AMJ, Ahring K,
Supplementary appendix This appendix formed part of the original submission and has been peer reviewed. We post it as supplied by the authors. Supplement to: van Spronsen FJ, van Wegberg AMJ, Ahring K,
Structural Studies on Phenylalanine Hydroxylase and Implications Toward Understanding and Treating Phenylketonuria
 Structural Studies on Phenylalanine Hydroxylase and Implications Toward Understanding and Treating Phenylketonuria Heidi Erlandsen, DrSci; Marianne G. Patch, PhD; Alejandra Gamez, PhD; Mary Straub; and
Structural Studies on Phenylalanine Hydroxylase and Implications Toward Understanding and Treating Phenylketonuria Heidi Erlandsen, DrSci; Marianne G. Patch, PhD; Alejandra Gamez, PhD; Mary Straub; and
Data Sheet PD-1 / NFAT Reporter - Jurkat Cell Line Catalog #: 60535
 Data Sheet PD-1 / NFAT Reporter - Jurkat Cell Line Catalog #: 60535 Product Description Recombinant Jurkat T cell expressing firefly luciferase gene under the control of NFAT response elements with constitutive
Data Sheet PD-1 / NFAT Reporter - Jurkat Cell Line Catalog #: 60535 Product Description Recombinant Jurkat T cell expressing firefly luciferase gene under the control of NFAT response elements with constitutive
MCB130 Midterm. GSI s Name:
 1. Peroxisomes are small, membrane-enclosed organelles that function in the degradation of fatty acids and in the degradation of H 2 O 2. Peroxisomes are not part of the secretory pathway and peroxisomal
1. Peroxisomes are small, membrane-enclosed organelles that function in the degradation of fatty acids and in the degradation of H 2 O 2. Peroxisomes are not part of the secretory pathway and peroxisomal
Molecular Genetics and Metabolism
 Molecular Genetics and Metabolism 101 (2010) 110 114 Contents lists available at ScienceDirect Molecular Genetics and Metabolism journal homepage: www.elsevier.com/locate/ymgme Sapropterin therapy increases
Molecular Genetics and Metabolism 101 (2010) 110 114 Contents lists available at ScienceDirect Molecular Genetics and Metabolism journal homepage: www.elsevier.com/locate/ymgme Sapropterin therapy increases
Introduction. R Christensen 1, S Kolvraa 1, RM Blaese 2 and TG Jensen 1 1
 INHERITED DISEASE (2000) 7, 1971 1978 2000 Macmillan Publishers Ltd All rights reserved 0969-7128/00 $15.00 www.nature.com/gt RESEARCH ARTICLE Development of a skin-based metabolic sink for phenylalanine
INHERITED DISEASE (2000) 7, 1971 1978 2000 Macmillan Publishers Ltd All rights reserved 0969-7128/00 $15.00 www.nature.com/gt RESEARCH ARTICLE Development of a skin-based metabolic sink for phenylalanine
Mapping the Ligand-binding Site on a GPCR Using Genetically-encoded Photocrosslinkers
 Mapping the Ligand-binding Site on a GPCR Using Genetically-encoded Photocrosslinkers Amy Grunbeck, Thomas Huber, Pallavi Sachdev, Thomas P. Sakmar Laboratory of Molecular Biology and Biochemistry, The
Mapping the Ligand-binding Site on a GPCR Using Genetically-encoded Photocrosslinkers Amy Grunbeck, Thomas Huber, Pallavi Sachdev, Thomas P. Sakmar Laboratory of Molecular Biology and Biochemistry, The
Mitochondrial Trifunctional Protein (TFP) Protein Quantity Microplate Assay Kit
 PROTOCOL Mitochondrial Trifunctional Protein (TFP) Protein Quantity Microplate Assay Kit DESCRIPTION Mitochondrial Trifunctional Protein (TFP) Protein Quantity Microplate Assay Kit Sufficient materials
PROTOCOL Mitochondrial Trifunctional Protein (TFP) Protein Quantity Microplate Assay Kit DESCRIPTION Mitochondrial Trifunctional Protein (TFP) Protein Quantity Microplate Assay Kit Sufficient materials
L6 GLUT4myc Cell Growth Protocol
 L6 GLUT4myc Cell Growth Protocol Background: Parental L6 cells selected for high fusion (2, 3) were stably transfected with a rat GLUT4 cdna carrying a myc epitope (recognized by the commercially available
L6 GLUT4myc Cell Growth Protocol Background: Parental L6 cells selected for high fusion (2, 3) were stably transfected with a rat GLUT4 cdna carrying a myc epitope (recognized by the commercially available
The Schedule and the Manual of Basic Techniques for Cell Culture
 The Schedule and the Manual of Basic Techniques for Cell Culture 1 Materials Calcium Phosphate Transfection Kit: Invitrogen Cat.No.K2780-01 Falcon tube (Cat No.35-2054:12 x 75 mm, 5 ml tube) Cell: 293
The Schedule and the Manual of Basic Techniques for Cell Culture 1 Materials Calcium Phosphate Transfection Kit: Invitrogen Cat.No.K2780-01 Falcon tube (Cat No.35-2054:12 x 75 mm, 5 ml tube) Cell: 293
Kit for assay of thioredoxin
 FkTRX-02-V2 Kit for assay of thioredoxin The thioredoxin system is the major protein disulfide reductase in cells and comprises thioredoxin, thioredoxin reductase and NADPH (1). Thioredoxin systems are
FkTRX-02-V2 Kit for assay of thioredoxin The thioredoxin system is the major protein disulfide reductase in cells and comprises thioredoxin, thioredoxin reductase and NADPH (1). Thioredoxin systems are
Disclosure. I have no disclosures to mention
 Disclosure I have no disclosures to mention Liberalization of the Dietary Restrictions for PKU Patients: Large Neutral Amino Acids (LNAA) Supplementation in PKU Mice Dr. Eddy A. van der Zee, University
Disclosure I have no disclosures to mention Liberalization of the Dietary Restrictions for PKU Patients: Large Neutral Amino Acids (LNAA) Supplementation in PKU Mice Dr. Eddy A. van der Zee, University
Chapter 7 Conclusions
 VII-1 Chapter 7 Conclusions VII-2 The development of cell-based therapies ranging from well-established practices such as bone marrow transplant to next-generation strategies such as adoptive T-cell therapy
VII-1 Chapter 7 Conclusions VII-2 The development of cell-based therapies ranging from well-established practices such as bone marrow transplant to next-generation strategies such as adoptive T-cell therapy
HCC1937 is the HCC1937-pcDNA3 cell line, which was derived from a breast cancer with a mutation
 SUPPLEMENTARY INFORMATION Materials and Methods Human cell lines and culture conditions HCC1937 is the HCC1937-pcDNA3 cell line, which was derived from a breast cancer with a mutation in exon 20 of BRCA1
SUPPLEMENTARY INFORMATION Materials and Methods Human cell lines and culture conditions HCC1937 is the HCC1937-pcDNA3 cell line, which was derived from a breast cancer with a mutation in exon 20 of BRCA1
Functional analysis and in vitro correction. splicing FAH mutations causing tyrosinemia type I.
 Clin Genet 2014: 86: 167 171 Printed in Singapore. All rights reserved Short Report 2013 John Wiley & Sons A/S. Published by John Wiley & Sons Ltd CLINICAL GENETICS doi: 10.1111/cge.12243 Functional analysis
Clin Genet 2014: 86: 167 171 Printed in Singapore. All rights reserved Short Report 2013 John Wiley & Sons A/S. Published by John Wiley & Sons Ltd CLINICAL GENETICS doi: 10.1111/cge.12243 Functional analysis
Lecture 10 - Protein Turnover and Amino Acid Catabolism
 Lecture 10 - Protein Turnover and Amino Acid Catabolism Chem 454: Regulatory Mechanisms in Biochemistry University of Wisconsin-Eau Claire 1 Introduction 2 Proteins are degraded into amino acids. Protein
Lecture 10 - Protein Turnover and Amino Acid Catabolism Chem 454: Regulatory Mechanisms in Biochemistry University of Wisconsin-Eau Claire 1 Introduction 2 Proteins are degraded into amino acids. Protein
Table S1. Sequence of human and mouse primers used for RT-qPCR measurements.
 Table S1. Sequence of human and mouse primers used for RT-qPCR measurements. Ca9, carbonic anhydrase IX; Ndrg1, N-myc downstream regulated gene 1; L28, ribosomal protein L28; Hif1a, hypoxia inducible factor
Table S1. Sequence of human and mouse primers used for RT-qPCR measurements. Ca9, carbonic anhydrase IX; Ndrg1, N-myc downstream regulated gene 1; L28, ribosomal protein L28; Hif1a, hypoxia inducible factor
Summary comments: An essential pattern employed by all living things: DNA ö RNA ö Protein
 Summary comments: An essential pattern employed by all living things: DNA ö RNA ö Protein DNA is the information storage molecule. RNA in an intermediate in information flow. Proteins do most of the jobs
Summary comments: An essential pattern employed by all living things: DNA ö RNA ö Protein DNA is the information storage molecule. RNA in an intermediate in information flow. Proteins do most of the jobs
MOLECULAR CELL BIOLOGY
 1 Lodish Berk Kaiser Krieger scott Bretscher Ploegh Matsudaira MOLECULAR CELL BIOLOGY SEVENTH EDITION CHAPTER 13 Moving Proteins into Membranes and Organelles Copyright 2013 by W. H. Freeman and Company
1 Lodish Berk Kaiser Krieger scott Bretscher Ploegh Matsudaira MOLECULAR CELL BIOLOGY SEVENTH EDITION CHAPTER 13 Moving Proteins into Membranes and Organelles Copyright 2013 by W. H. Freeman and Company
Quantification of phenylalanine hydroxylase activity by isotope-dilution liquid chromatography-electrospray ionization tandem mass spectrometry
 Zurich Open Repository and Archive University of Zurich Main Library Strickhofstrasse 39 CH-8057 Zurich www.zora.uzh.ch Year: 2012 Quantification of phenylalanine hydroxylase activity by isotope-dilution
Zurich Open Repository and Archive University of Zurich Main Library Strickhofstrasse 39 CH-8057 Zurich www.zora.uzh.ch Year: 2012 Quantification of phenylalanine hydroxylase activity by isotope-dilution
HA Convention 2016 Master course How to Handle Abnormal Newborn Metabolic Screening Results Causes, Management and Follow up
 HA Convention 2016 Master course How to Handle Abnormal Newborn Metabolic Screening Results Causes, Management and Follow up Dr. Josephine Chong Clinical Professional Consultant Centre of Inborn Errors
HA Convention 2016 Master course How to Handle Abnormal Newborn Metabolic Screening Results Causes, Management and Follow up Dr. Josephine Chong Clinical Professional Consultant Centre of Inborn Errors
Chemistry 107 Exam 4 Study Guide
 Chemistry 107 Exam 4 Study Guide Chapter 10 10.1 Recognize that enzyme catalyze reactions by lowering activation energies. Know the definition of a catalyst. Differentiate between absolute, relative and
Chemistry 107 Exam 4 Study Guide Chapter 10 10.1 Recognize that enzyme catalyze reactions by lowering activation energies. Know the definition of a catalyst. Differentiate between absolute, relative and
Key knowledge base & conceptual questions
 Key knowledge base & conceptual questions Why is it said the pentose phosphate pathway is the major source of reducing power? What are the differences, in structure and in function, between NADH and NADPH?
Key knowledge base & conceptual questions Why is it said the pentose phosphate pathway is the major source of reducing power? What are the differences, in structure and in function, between NADH and NADPH?
20S Proteasome Activity Assay Kit
 20S Proteasome Activity Assay Kit For 100 Assays Cat. No. APT280 FOR RESEARCH USE ONLY NOT FOR USE IN DIAGNOSTIC PROCEDURES USA & Canada Phone: +1(800) 437-7500 Fax: +1 (951) 676-9209 Europe +44 (0) 23
20S Proteasome Activity Assay Kit For 100 Assays Cat. No. APT280 FOR RESEARCH USE ONLY NOT FOR USE IN DIAGNOSTIC PROCEDURES USA & Canada Phone: +1(800) 437-7500 Fax: +1 (951) 676-9209 Europe +44 (0) 23
Data Sheet PD-1 / NFAT Reporter - Jurkat Cell Line Catalog #: 60535
 Data Sheet PD-1 / NFAT Reporter - Jurkat Cell Line Catalog #: 60535 Product Description Recombinant Jurkat T cell expressing firefly luciferase gene under the control of NFAT response elements with constitutive
Data Sheet PD-1 / NFAT Reporter - Jurkat Cell Line Catalog #: 60535 Product Description Recombinant Jurkat T cell expressing firefly luciferase gene under the control of NFAT response elements with constitutive
Supplementary Figure 1. Normal T lymphocyte populations in Dapk -/- mice. (a) Normal thymic development in Dapk -/- mice. Thymocytes from WT and Dapk
 Supplementary Figure 1. Normal T lymphocyte populations in Dapk -/- mice. (a) Normal thymic development in Dapk -/- mice. Thymocytes from WT and Dapk -/- mice were stained for expression of CD4 and CD8.
Supplementary Figure 1. Normal T lymphocyte populations in Dapk -/- mice. (a) Normal thymic development in Dapk -/- mice. Thymocytes from WT and Dapk -/- mice were stained for expression of CD4 and CD8.
Suppl. Figure 1. T 3 induces autophagic flux in hepatic cells. (A) RFP-GFP-LC3 transfected HepG2/TRα cells were visualized and cells were quantified
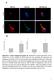 Suppl. Figure 1. T 3 induces autophagic flux in hepatic cells. (A) RFP-GFP-LC3 transfected HepG2/TRα cells were visualized and cells were quantified for RFP-LC3 puncta (red dots) representing both autolysosomes
Suppl. Figure 1. T 3 induces autophagic flux in hepatic cells. (A) RFP-GFP-LC3 transfected HepG2/TRα cells were visualized and cells were quantified for RFP-LC3 puncta (red dots) representing both autolysosomes
Electron micrograph of phosphotungstanic acid-stained exosomes derived from murine
 1 SUPPLEMENTARY INFORMATION SUPPLEMENTARY FIGURES Supplementary Figure 1. Physical properties of murine DC-derived exosomes. a, Electron micrograph of phosphotungstanic acid-stained exosomes derived from
1 SUPPLEMENTARY INFORMATION SUPPLEMENTARY FIGURES Supplementary Figure 1. Physical properties of murine DC-derived exosomes. a, Electron micrograph of phosphotungstanic acid-stained exosomes derived from
