Reduction in Workload and Reporting Time of MRSA Screening with MRSASelect. Medium versus Mannitol-Salt Medium Supplemented with Oxacillin ACCEPTED
|
|
|
- Alban Hamilton
- 5 years ago
- Views:
Transcription
1 JCM Accepts, published online ahead of print on 30 January 2008 J. Clin. Microbiol. doi: /jcm Copyright 2008, American Society for Microbiology and/or the Listed Authors/Institutions. All Rights Reserved. Reduction in Workload and Reporting Time of MRSA Screening with MRSASelect Medium versus Mannitol-Salt Medium Supplemented with Oxacillin Running Title: Workload Reduction of MRSA Screening with MRSASelect *Philippe R.S. Lagacé-Wiens 1,2, Michelle J. Alfa 1,2,3, Kanchana Manickam 2,3, Godfrey K.M Harding 1,2,3. 1. Department of Medical Microbiology and Infectious Diseases, University of Manitoba, Winnipeg, Manitoba, Canada. 2. Clinical Microbiology, Saint-Boniface General Hospital, Winnipeg, Manitoba, Canada. 3. Diagnostic Services of Manitoba, Winnipeg, Manitoba, Canada. *Corresponding author: Clinical Microbiology, Saint-Boniface General Hospital. Winnipeg, Manitoba, Canada. R2H 2A6 Ph. (204) Fax. (204)
2 Abstract: Methicillin resistant Staphylococcus aureus (MRSA) is a significant pathogen in both the nosocomial and community setting and screening for carriers is an important infection control practice in many hospitals. In this retrospective study, we demonstrate that the implementation of an MRSA screening protocol using a selective chromogenic medium (MRSASelect ) reduced the workload for this screening test by 63.7% overall and by 12.6% per specimen and reduced turn-around time for reporting by an average of 1.33 days for all MRSA screening specimens, 1.97 days for MRSA positive specimens and 1.3 days for MRSA negative specimens when compared to standard mannitol salt agar supplemented with 6mg/L oxacillin. 2
3 Introduction Methicillin-resistant Staphylococcus aureus (MRSA) is an important pathogen affecting all areas of hospital practice and is increasingly being recognized as an important pathogen in the community (5). The timely and cost-effective identification and isolation of MRSA carriers in an effort to reduce the intra and inter facility spread on this pathogen remains an important infection control issue in many hospitals (4). The steady increase in prevalence of MRSA combined with increased infection control awareness of MRSA has led to a dramatic increase in microbiology laboratory workload in the form of increased staff, space and material requirements (4, 5). Furthermore, adequate infection control measures require rapid turnaround time and accurate results in order to optimize hospital resources and reduce the spread of MRSA (4, 5). Therefore, MRSA screening methods need to be studied with respect to workload and reporting time as well as accuracy. Selective (eg. mannitol salt agar with oxacillin) media are advocated for MRSA screening (4, 7, 10). These selective and differential media are incubated for 24 to 72 hours and examined for suspect colonies. More rapid alternative methods for screening for MRSA carriers have been developed, including traditional and real-time PCR and chromogenic media which have been shown to be comparable or superior in sensitivity and specificity to traditional selective media such as mannitol salt agars supplemented with antimicrobials (1-3, 6, 9-11). These selective chromogenic media contain colorless proprietary molecules (chromogens) chemically resembling substrates of specific bacterial enzymes but releasing visible dyes when hydrolysed by micro-organisms possessing the required enzymes (9). Chromogens can be tailored enzymes relatively specific to certain organisms, for example, on MRSASelect, only Staphylococcus 3
4 aureus colonies produce a pink color within the recommended incubation period and other Staphylococci are white or colorless. In addition, these media contain multiple antimicrobial agents that serve to inhibit the growth of methicillin susceptible strains of S. aureus and other commensal organisms. The purpose of this study was to determine if a reduction in workload and turn around time could be observed by implementing a protocol using MRSASelect chromogenic medium as a rapid MRSA screening method compared to traditional MSA-based culture. 4
5 42 Methods: Study design: The number of specimens and workload generated by tests performed on the specimens that are processed were tracked automatically by the Delphic Laboratory Information System (Sysmex, Auckland, New Zealand). The workload unit (WLU) is defined as one minute of time and the number of WLUs assigned to a test reflects the actual hands-on time (minutes) needed to complete the test. WLUs are set by the Standards for Management Information Systems in Canadian Health Service Organizations for all common laboratory tests and are based on time studies and are periodically updated to ensure they are accurate and current as new methods are developed. A base number of WLUs are assigned for specimen accessioning, set-up and initial read, and additional tests are deemed extra WLUs. The total WLUs for specific tests (such as MRSA screening) are compiled by the electronic system as additional tests are ordered for suspect organisms. Workload associated with an MRSA screening method based on mannitol-salt agar medium supplemented with 6mg/L oxacillin (MSA/Ox) (Figure 1) was reviewed January June, 2005 using the laboratory information system. In this protocol, specimens were directly inoculated to the medium and examined for suspect colonies after hours incubation. Suspect colonies were confirmed as S. aureus using a 24 hour tube coagulase and methicillin resistance was confirmed after overnight susceptibility testing with the Vitek susceptibility card followed by a commercial MecA latex agglutination test. Negative plates were re-incubated and negative results were reported if plates remained negative after 48 hours. After the implementation of an MRSASelect-based MRSA screening protocol (Figure 1), the workload generated by this new method was tracked for another 6 month period (July 5
6 December, 2006). In this protocol, specimens were directly inoculated to the media and incubated for hours. The growth of any pink colonies on the MRSASelect within this incubation period was reported as MRSA without additional testing. Because the manufacturer of MRSASelect warns that coagulase negative staphylococci may appear pink after prolonged incubation, pink colonies observed after the recommended 24 hour incubation period were confirmed as S. aureus using a 24 hour tube coagulase test. Negative plates that were examined before 18 hours of incubation were re-incubated and suspect colonies later identified were confirmed as S. aureus using a tube coagulase test. Negative results were reported if plates were negative after 24 hours incubation. Turn around time (TAT) was also compared for both protocols. 500 consecutive specimens from the start of each study period were analyzed and reporting times were compared. TAT has been defined as the time from specimen receipt in the laboratory (accessioning) to the time the result was phoned (i.e. positive for MRSA) or to the time the report was printed and sent to the ward (i.e. negative specimens). The accessioning time, phoned report times and final printed report times are automatically recorded by the electronic system, allowing for accurate determination of TAT for MRSA screening regardless of whether they were positive or negative Specimens: Specimens submitted for MRSA surveillance including swabs of the nares, wounds, respiratory, urine, intravascular catheter sites and rectum/stool were included in this study. Diagnostic specimens were not included as these are not directly inoculated on to MRSA screening media. Swabs for MRSA screening were transported in M40 sterile transport systems (Copan Innovations, Corona, CA, USA) to the laboratory where they were plated to media according to the protocol in place at the time. 6
7 90 7
8 Media: The mannitol-salt agar (MSA) supplemented with 6mg/L oxacillin (MSA/Ox) was produced in-house using MSA powder (BD, Oakville, ON, Canada), oxacillin (Sigma, Oakville, ON, Canada) and following manufacturer s instructions. Sheep s blood agar (SBA) was produced in-house with tryptic soy agar powder (BD, Oakville, ON, Canada) and sheep blood (Quad Five, Ryegate, MT, USA). Chromogenic media (MRSASelect ) was purchased from the manufacturer (Biorad, Montreal, Canada). All media were subjected to internal quality control using oxacillin-resistant S. aureus (ATCC 43300) and oxacillin-sensitive S. aureus (ATCC 29213). When specimens were from known MRSA carriers, SBA was co-inoculated with MSA/Ox (under the MSA/Ox protocol) because it increased the sensitivity of MRSA detection in our MRSA positive hospital population (data not shown). Selective enrichment broth (Brain Heart Infusion broth supplemented with ceftizoxime, 5 µg/ml, and aztreonam, 75 µg/ml) (12) was inoculated during the period when MRSASelect for the same population. Our laboratory had previously shown that broth enrichment only improved the sensitivity of direct plating for known MRSA positive patients and that a combined approach using MRSASelect was as sensitive as molecular amplification of MRSA-specific targets from specimens (data not shown). This effect is likely due to common practice of using antimicrobials for MRSA decolonization of patients in our centre Statistical analysis: Student s t-test was used to compare means in the reporting time analysis. 8
9 114 Results: Reduction in workload: The workload for MRSA screening was retrospectively evaluated for two-different six month periods. Total workload went down from 168,950 to 61,295 WLUs (-63.72%). The reduction was most evident in extra workload units (slide agglutination, tube coagulase, catalase tests, antimicrobial disk diffusion and subculturing) which were reduced from to 3102 WLUs (-90.90%). Specimen number was also less in the second period (from 13,276 to 5,513). Nevertheless, there was a workload reduction of 1.61 WLU/specimen (-12.63% per specimen) which was also primarily due to a reduction in extra workload units from 2.57WLU/specimen to 0.56WLU/specimen ( %.). The anticipated impact for a large clinical laboratory such as ours that processes 11,000 MRSA screening specimens per year tending to a population with approximately 5% MRSA carriage would be approximately 300 hours of technologist time. Reporting time: Reporting times for both study periods are summarized in Table 1. Overall, reporting time for MRSA screening specimens went from 3 days with the MSA/Ox protocol to 1.66 days with the MRSASelect protocol (-44.7%, p < ). The reduction in TAT was relatively larger in MRSA positive specimens (3.53 days to 1.56 days, -55.8%) than for MRSA negative specimens (2.96 days to 1.66 days, 43.9%) but statistically significant reductions in TAT were observed in both positive and negative specimens (p < ). Reporting time was significantly improved for all specimen types (wounds, nares, rectal, respiratory and intravascular line specimens). Urine specimens were too few for statistical analysis. The number of MRSA positive screening 9
10 specimens in the 500 specimens selected from each study period was not significantly different (30/500 (6%) during the MSA/Ox protocol and 20/500 (4.0%) during the MRSASelect protocol, p = 0.60). The broth enrichment identified 5 (20%) of the positive MRSA cases that would have been missed if direct plating to MRSASelect alone had been used to screen previously positive patients. Using sub-group analysis of our data, a broth enrichment protocol under the MRSASelect protocol would increase turn-around time from 1.56 to 2.12 days. However, overall turn-around time would still be improved over an MSA/Ox direct plating protocol (2.12 days vs days, p < 0.001). 10
11 146 Discussion: Sensitivity and specificity of MRSASelect medium (97.3% and 99.8%) has already been studied and compared to MSA/Ox (80.2% and 79.0%) for MRSA screening (9). Although MRSASelect compares favorably or is superior to other solid screening media in terms of sensitivity and specificity, some authors have questioned the screening sensitivity of direct plating on chromogenic media and have demonstrated that pre- incubating in enrichment broth increased detection by 16-24% in several chromogenic media, but the post-enrichment sensitivity of MRSASelect was not analyzed (8). In our hands, parallel testing of both media with broth enrichment and direct PCR in our laboratory for validation purposes corroborated published data but similar sensitivity rates with and without broth enrichment for routine screening, improved sensitivity only being seen in known MRSA positive patients. Although the sensitivity and specificity of chromogenic MRSA screening media has been well studied, the use of chromogenic media for streamlining workflow and rapid result reporting has not been well studied. Our data showed that the protocol using MRSASelect medium requires less additional tests, subcultures and confirmatory tests than the protocol based on MSA/Ox. This resulted in a significant reduction in workload, both overall and per specimen plated. This workload reduction was shown to be primarily due to a reduction in extra workload, (subcultures catalase, coagulase etc.). Furthermore, due to the distinct color difference between S. aureus (pink) and coagulase negative staphylococci (white) on MRSASelect, our data demonstrated there was less frequent need to subculture and repeat susceptibility or identification tests. While the elimination of confirmatory tests (eg. meca PCR or PBP2 latex agglutination) might be expected to result in a greater 11
12 number of false positive MRSA screening tests when using the MRSASelect protocol, parallel runs in our laboratory using latex agglutination confirmatory testing under both protocols revealed that the chromogenic medium did not produce any false positives (data not shown). The high specificity of the MRSASelect medium has also been previously described ( %) and confirmatory testing is not required according to the manufacturer. This recommendation, based on high specificity, is also supported by a number of independent studies (1, 8, 9). Since confirmatory testing is not apparently required with the MRSASelect protocol, workload reduction is expected to be even greater where prevalence of MRSA carriage is higher than in our centre (~6%). Reporting time for both positive and negative MRSA screening specimens was significantly reduced with MRSASelect. Our reporting time of 1.66 days for MRSASelect was similar to those published by others(8). The reduction in TAT was most significant for MRSA positive specimens, which suggests that hospitals with higher rates of MRSA would benefit most from adopting a similar protocol. For negative specimens, the reduction was primarily due to the MRSASelect medium providing comparable specificity results to the MSA/Ox medium after hours of incubation instead of the 48 hours of incubation required for MSA/Ox before a negative result can be issued. For MRSA positive specimens, the reduced TAT was partly due to a reduction in subculturing and repeating susceptibility testing for coagulase negative staphylococci that were inadvertently inoculated to the Vitek susceptibility card (occurred when coagulase negative Staphylococcus was growing on MSA/Ox but not for MRSASelect ). Furthermore, the high specificity of MRSASelect reported in the literature as well as in our own hands effectively eliminated the need for confirmatory 12
13 testing using meca PCR or PBP2 latex agglutination. This resulted in a further reduction of TAT for positive specimens. Another possibility for the observed reduction in reporting time was a change in the provincial infection control guidelines that occurred between the two study periods that eliminated the need for routine screening of stool and urine for MRSA. Fewer specimens contaminated with endogenous flora (such as stool) may have reduced the overall processing time. To evaluate this we analyzed reporting time by specimen type and determined that reporting time was significantly reduced for all specimen types. Therefore, it is unlikely that the specimen source was a major confounder of our findings. Although pre-manufactured chromogenic media for MRSA screening are more costly than routine media produced in-house, the significant reduction in workload, the ability to report MRSA based on pink color at hours without confirmatory tests and the reduced TAT resulted in the use of MRSASelect being a cost-effective alternative for screening MRSA in our laboratory. Indeed, the MRSA screening workload was reduced whereby the work that required two full-time technologists to complete using MSA/Ox could now be handled by one full-time technologist using MRSASelect. However, not all hospital laboratories would benefit from this or a similar protocol. Hospitals not required to or choosing not to screen for MRSA would clearly not benefit. Furthermore, screening is not cost effective in areas where MRSA prevalence is so high as to make contact isolation of carriers prohibitively expensive. Lastly, although subgroup analysis of our data revealed that universal broth-enrichment with subsequent use of MRSASelect agar still provided a reduction in TAT over direct plating on MSA/Ox, laboratories that choose enhanced screening with universal broth-enrichment may not 13
14 observe as significant a reduction in turn-around times. Nevertheless, there is increasing government pressure in both Canada and the United States to increase efforts to screen and identify MRSA carriers in hospital. This is evidenced by recent provincial and state mandates recommending enhanced screening for MRSA. Our findings suggest that where screening for MRSA using solid media is desired or mandated, MRSASelect may be superior to MSA/Ox for routine screening as it has faster TAT and lower labor requirement. Part of the data in this publication was presented in abstract format at the 2007 AMMI/CACMID general meeting in Halifax, Canada. 14
15 Acknowledgements The skilled technical assistance of Pat DeGagné, Barbara J. Sheard, and Jan Schmidt, as well as the other technologists in the Microbiology lab at the St. Boniface General Hospital site is acknowledged. 15
16 Cherkaoui, A., G. Renzi, P. Francois, and J. Schrenzel Comparison of four chromogenic media for culture-based screening of meticillin-resistant Staphylococcus aureus. J Med Microbiol 56: de San, N., O. Denis, M. F. Gasasira, R. De Mendonca, C. Nonhoff, and M. J. Struelens Controlled evaluation of the IDI-MRSA assay for detection of colonization by methicillin-resistant Staphylococcus aureus in diverse mucocutaneous specimens. J Clin Microbiol 45: Flayhart, D., J. F. Hindler, D. A. Bruckner, G. Hall, R. K. Shrestha, S. A. Vogel, S. S. Richter, W. Howard, R. Walther, and K. C. Carroll Multicenter evaluation of BBL CHROMagar MRSA medium for direct detection of methicillin-resistant Staphylococcus aureus from surveillance cultures of the anterior nares. J Clin Microbiol 43: Harbarth, S Control of endemic methicillin-resistant Staphylococcus aureus-recent advances and future challenges. Clin Microbiol Infect 12: Kluytmans-Vandenbergh, M. F., and J. A. Kluytmans Community- acquired methicillin-resistant Staphylococcus aureus: current perspectives. Clin Microbiol Infect 12 Suppl 1: Kluytmans, J., A. Van Griethuysen, P. Willemse, and P. Van Keulen Performance of CHROMagar selective medium and oxacillin resistance screening agar base for identifying Staphylococcus aureus and detecting methicillin resistance. J Clin Microbiol 40: Miller, M., and M. Bell Epidemiology and Infection Control Microbiology, p In H. Isenberg (ed.), Clinical Microbiology Procedures Handbook, vol. 3. ASM Press, Washington, DC. 16
17 Nahimana, I., P. Francioli, and D. S. Blanc Evaluation of three chromogenic media (MRSA-ID, MRSA-Select and CHROMagar MRSA) and ORSAB for surveillance cultures of methicillin-resistant Staphylococcus aureus. Clin Microbiol Infect 12: Stoakes, L., R. Reyes, J. Daniel, G. Lennox, M. A. John, R. Lannigan, and Z. Hussain Prospective comparison of a new chromogenic medium, MRSASelect, to CHROMagar MRSA and mannitol-salt medium supplemented with oxacillin or cefoxitin for detection of methicillin-resistant Staphylococcus aureus. J Clin Microbiol 44: Velasco, D., M. del Mar Tomas, M. Cartelle, A. Beceiro, A. Perez, F. Molina, R. Moure, R. Villanueva, and G. Bou Evaluation of different methods for detecting methicillin (oxacillin) resistance in Staphylococcus aureus. J Antimicrob Chemother 55: Wagenvoort, J. H., M. F. van de Cruijs, C. T. Meuwissen, J. M. Gronenschild, and E. I. De Brauwer Comparison of an enrichment broth- enhanced commercial PCR procedure versus bacteriological culture for separating non-colonized from suspected or colonized MRSA individuals. Eur J Clin Microbiol Infect Dis 26: Wertheim, H., H. A. Verbrugh, C. van Pelt, P. de Man, A. van Belkum, and M. C. Vos Improved detection of methicillin-resistant Staphylococcus aureus using phenyl mannitol broth containing aztreonam and ceftizoxime. J Clin Microbiol 39:
18 FIGURE 1: Comparison of process flow for MSA/Ox and MRSASelect protocols. Specimens from previously known MRSA carriers were also concomitantly plated to non-selective media (sheep blood agar) in the MSA/Ox protocol or inoculated into enrichment broth in the MRSASelect protocol (broth only worked-up if direct plated specimens negative at 24 hours). 18
19 TABLE 1: Turn-around time for final reporting results of MRSA screening specimens by specimen type. 286 MRSA positive MRSA negative All specimens MRSASelect (n=500) MSA/Ox (n=500) p value n Mean TAT (days) n Mean TAT (days) < < < Nares < Wound < Rectal IV catheter site < Respiratory Urine * 287 *Insufficient data for statistical analysis. 19
Veerle Compernolle, Gerda Verschraegen, and Geert Claeys* Department of Microbiology, Ghent University Hospital, Ghent, Belgium
 JOURNAL OF CLINICAL MICROBIOLOGY, Jan. 2007, p. 154 158 Vol. 45, No. 1 0095-1137/07/$08.00 0 doi:10.1128/jcm.01115-06 Copyright 2007, American Society for Microbiology. All Rights Reserved. Combined Use
JOURNAL OF CLINICAL MICROBIOLOGY, Jan. 2007, p. 154 158 Vol. 45, No. 1 0095-1137/07/$08.00 0 doi:10.1128/jcm.01115-06 Copyright 2007, American Society for Microbiology. All Rights Reserved. Combined Use
Ueli von Ah, Dieter Wirz, and A. U. Daniels*
 JOURNAL OF CLINICAL MICROBIOLOGY, June 2008, p. 2083 2087 Vol. 46, No. 6 0095-1137/08/$08.00 0 doi:10.1128/jcm.00611-08 Copyright 2008, American Society for Microbiology. All Rights Reserved. Rapid Differentiation
JOURNAL OF CLINICAL MICROBIOLOGY, June 2008, p. 2083 2087 Vol. 46, No. 6 0095-1137/08/$08.00 0 doi:10.1128/jcm.00611-08 Copyright 2008, American Society for Microbiology. All Rights Reserved. Rapid Differentiation
(multidrug-resistant Pseudomonas aeruginosa; MDRP)
 220 2009 (multidrug-resistant Pseudomonas aeruginosa; MDRP) 21 4 1 21 10 4 amikacin (AMK), imipenem/cilastatin (IPM), ciprofloxacin (CPFX) multidrug-resistant Pseudomonas aeruginosa (MDRP) CHROMagar TM
220 2009 (multidrug-resistant Pseudomonas aeruginosa; MDRP) 21 4 1 21 10 4 amikacin (AMK), imipenem/cilastatin (IPM), ciprofloxacin (CPFX) multidrug-resistant Pseudomonas aeruginosa (MDRP) CHROMagar TM
Comparison of flocked and rayon swabs for detection of nasal carriage of Staphylococcus aureus amongst pathology staff
 JCM Accepts, published online ahead of print on 26 May 2010 J. Clin. Microbiol. doi:10.1128/jcm.01617-09 Copyright 2010, American Society for Microbiology and/or the Listed Authors/Institutions. All Rights
JCM Accepts, published online ahead of print on 26 May 2010 J. Clin. Microbiol. doi:10.1128/jcm.01617-09 Copyright 2010, American Society for Microbiology and/or the Listed Authors/Institutions. All Rights
During High-Volume Clinical Use ACCEPTED. Healthcare, Evanston, IL, Northwestern University s Feinberg School of Medicine, Chicago,
 JCM Accepts, published online ahead of print on 11 July 2007 J. Clin. Microbiol. doi:10.1128/jcm.00670-07 Copyright 2007, American Society for Microbiology and/or the Listed Authors/Institutions. All Rights
JCM Accepts, published online ahead of print on 11 July 2007 J. Clin. Microbiol. doi:10.1128/jcm.00670-07 Copyright 2007, American Society for Microbiology and/or the Listed Authors/Institutions. All Rights
GBS Screening, diagnosis and clinically relevant resistance
 GBS Screening, diagnosis and clinically relevant resistance Pierrette Melin National Reference Centre for GBS Medical Microbiology, University Hospital of Liege 1 Background Evolution of culture methods
GBS Screening, diagnosis and clinically relevant resistance Pierrette Melin National Reference Centre for GBS Medical Microbiology, University Hospital of Liege 1 Background Evolution of culture methods
OUTLINE Laboratory Detection and Reporting of Streptococcus agalactiae
 OUTLINE Laboratory Detection and Reporting of Streptococcus agalactiae I. Importance of prenatal screening strategies II. Past approaches Erik Munson Clinical Microbiology Wheaton Franciscan Laboratory
OUTLINE Laboratory Detection and Reporting of Streptococcus agalactiae I. Importance of prenatal screening strategies II. Past approaches Erik Munson Clinical Microbiology Wheaton Franciscan Laboratory
Laboratory Detection and Reporting of Streptococcus agalactiae
 Laboratory Detection and Reporting of Streptococcus agalactiae Erik Munson Clinical Microbiology Wheaton Franciscan Laboratory Milwaukee, Wisconsin The presenter states no conflict of interest and has
Laboratory Detection and Reporting of Streptococcus agalactiae Erik Munson Clinical Microbiology Wheaton Franciscan Laboratory Milwaukee, Wisconsin The presenter states no conflict of interest and has
Frequency of Nasal Carriage of Staphylococcus Aureus and Its Antimicrobial Resistance Pattern in Patients on Hemodialysis
 Dialysis Frequency of Nasal Carriage of Staphylococcus Aureus and Its Antimicrobial Resistance Pattern in Patients on Hemodialysis Roya Ghasemian, 1 Narges Najafi, 1 Atieh Makhlough, 2 Mohammad Khademloo
Dialysis Frequency of Nasal Carriage of Staphylococcus Aureus and Its Antimicrobial Resistance Pattern in Patients on Hemodialysis Roya Ghasemian, 1 Narges Najafi, 1 Atieh Makhlough, 2 Mohammad Khademloo
Discussion points CLSI M100 S19 Update. #1 format of tables has changed. #2 non susceptible category
 Discussion points 2009 CLSI M100 S19 Update Nebraska Public Health Laboratory Changes most important to routine antimicrobial susceptibility testing. Documents available Janet Hindler discussion slide
Discussion points 2009 CLSI M100 S19 Update Nebraska Public Health Laboratory Changes most important to routine antimicrobial susceptibility testing. Documents available Janet Hindler discussion slide
Comparison of the BD MAX MRSA Assay and the BD GeneOhm MRSA. Achromopeptidase Assay for the Detection of Methicillin-Resistant Staphylococcus aureus
 JCM Accepts, published online ahead of print on 18 July 2012 J. Clin. Microbiol. doi:10.1128/jcm.01496-12 Copyright 2012, American Society for Microbiology. All Rights Reserved. 1 2 3 Comparison of the
JCM Accepts, published online ahead of print on 18 July 2012 J. Clin. Microbiol. doi:10.1128/jcm.01496-12 Copyright 2012, American Society for Microbiology. All Rights Reserved. 1 2 3 Comparison of the
Mycobacteria Diagnostic Testing in Manitoba. Dr. Michelle Alfa Medical Director, DSM Clin Micro Discipline
 Mycobacteria Diagnostic Testing in Manitoba Dr. Michelle Alfa Medical Director, DSM Clin Micro Discipline Acknowlegements: Assunta Rendina: Charge Tech HSC Lab Joyce Wolf & Dr. Meenu Sharma: NML Dr. Kanchana
Mycobacteria Diagnostic Testing in Manitoba Dr. Michelle Alfa Medical Director, DSM Clin Micro Discipline Acknowlegements: Assunta Rendina: Charge Tech HSC Lab Joyce Wolf & Dr. Meenu Sharma: NML Dr. Kanchana
Online Ordering Available. GBS Detect
 Online Ordering Available GBS Detect Fast New color change method for cultivation and identification of beta-hemolytic strains of Group B Streptococcus! Strep B Carrot Broth kit is a color change method
Online Ordering Available GBS Detect Fast New color change method for cultivation and identification of beta-hemolytic strains of Group B Streptococcus! Strep B Carrot Broth kit is a color change method
Rifampin Resistance. Charlottesville, Virginia i0w organisms in Trypticase soy broth (BBL Microbiology
 ANTIMICROBIAL AGENTS AND CHEMOTHERAPY, Apr. 1980, p. 658-662 0066-4804/80/04-0658/05$02.00/0 Vol. 17, No. 14 Treatment of Experimental Staphylococcal Infections: Effect of Rifampin Alone and in Combination
ANTIMICROBIAL AGENTS AND CHEMOTHERAPY, Apr. 1980, p. 658-662 0066-4804/80/04-0658/05$02.00/0 Vol. 17, No. 14 Treatment of Experimental Staphylococcal Infections: Effect of Rifampin Alone and in Combination
VRE. Is There Validity to VRE Admission Screening? Dr. Michelle Alfa, Diagnos>c Services Manitoba A Webber Training Teleclass. Overview of Session:
 Dr. Michelle Alfa, Ph.D., FCCM Medical Director, Clinical Microbiology Discipline, Diagnostic Services Manitoba Hosted by Paul Webber paul@webbertraining.com December 19, 2013 Session Rated PG99: Scenes
Dr. Michelle Alfa, Ph.D., FCCM Medical Director, Clinical Microbiology Discipline, Diagnostic Services Manitoba Hosted by Paul Webber paul@webbertraining.com December 19, 2013 Session Rated PG99: Scenes
CONSIDERATIONS IN UTI DETECTION AND POTENTIAL IMPACT ON ANTIBIOTIC STEWARDSHIP
 CONSIDERATIONS IN UTI DETECTION AND POTENTIAL IMPACT ON ANTIBIOTIC STEWARDSHIP ERIN H. GRAF, PHD, D(ABMM) Director, Infectious Disease Diagnostics Laboratory Assistant Professor, Clinical Pathology and
CONSIDERATIONS IN UTI DETECTION AND POTENTIAL IMPACT ON ANTIBIOTIC STEWARDSHIP ERIN H. GRAF, PHD, D(ABMM) Director, Infectious Disease Diagnostics Laboratory Assistant Professor, Clinical Pathology and
Biological Consulting Services
 Biological Consulting Services of North Florida/ Inc. May 13, 2009 Aphex BioCleanse Systems, Inc. Dear Sirs, We have completed antimicrobial efficacy study on the supplied Multi-Purpose Solution. The testing
Biological Consulting Services of North Florida/ Inc. May 13, 2009 Aphex BioCleanse Systems, Inc. Dear Sirs, We have completed antimicrobial efficacy study on the supplied Multi-Purpose Solution. The testing
RAPID REDUCTION OF STAPHYLOCOCCUS AUREUS POPULATIONS ON STAINLESS STEEL SURFACES BY ZEOLITE CERAMIC COATINGS CONTAINING SILVER AND ZINC IONS
 RAPID REDUCTION OF STAPHYLOCOCCUS AUREUS POPULATIONS ON STAINLESS STEEL SURFACES BY ZEOLITE CERAMIC COATINGS CONTAINING SILVER AND ZINC IONS INE 2 (CAR HEADLINE 2) By: K. R. Bright C. P. Gerba P. A. Rusin
RAPID REDUCTION OF STAPHYLOCOCCUS AUREUS POPULATIONS ON STAINLESS STEEL SURFACES BY ZEOLITE CERAMIC COATINGS CONTAINING SILVER AND ZINC IONS INE 2 (CAR HEADLINE 2) By: K. R. Bright C. P. Gerba P. A. Rusin
BD BBL CHROMagar Staph aureus
 INSTRUCTIONS FOR USE READY-TO-USE PLATED MEDIA PA-257074.06 Rev.: Sep 2011 BD BBL CHROMagar Staph aureus INTENDED USE BBL CHROMagar Staph aureus is a selective medium for the isolation, enumeration and
INSTRUCTIONS FOR USE READY-TO-USE PLATED MEDIA PA-257074.06 Rev.: Sep 2011 BD BBL CHROMagar Staph aureus INTENDED USE BBL CHROMagar Staph aureus is a selective medium for the isolation, enumeration and
Received 3 November 2010/Returned for modification 26 December 2010/Accepted 1 February 2011
 JOURNAL OF CLINICAL MICROBIOLOGY, Apr. 2011, p. 1240 1244 Vol. 49, No. 4 0095-1137/11/$12.00 doi:10.1128/jcm.02220-10 Copyright 2011, American Society for Microbiology. All Rights Reserved. Genotypic and
JOURNAL OF CLINICAL MICROBIOLOGY, Apr. 2011, p. 1240 1244 Vol. 49, No. 4 0095-1137/11/$12.00 doi:10.1128/jcm.02220-10 Copyright 2011, American Society for Microbiology. All Rights Reserved. Genotypic and
Mt. San Antonio College Microbiology 22 Lab Schedule for Spring 2018 Mon/Weds. Split Lab Sections ONLY
 Mt. San Antonio College Microbiology 22 Lab Schedule for Spring 2018 Mon/ Split Lab Sections ONLY Wk 1 Feb. 26 Orientation with Introductions & Safety Rules/Regulations Feb. 28 Orientation with Pathogen
Mt. San Antonio College Microbiology 22 Lab Schedule for Spring 2018 Mon/ Split Lab Sections ONLY Wk 1 Feb. 26 Orientation with Introductions & Safety Rules/Regulations Feb. 28 Orientation with Pathogen
Mt. San Antonio College Microbiology 22 Lab Schedule for Spring 2018 Tues/Thurs. Split Lab Sections ONLY
 Mt. San Antonio College Microbiology 22 Lab Schedule for Spring 2018 Tues/ Split Lab Sections ONLY Wk 1 Feb. 27 Orientation with Introductions & Safety Rules/Regulations March 1 Orientation with Pathogen
Mt. San Antonio College Microbiology 22 Lab Schedule for Spring 2018 Tues/ Split Lab Sections ONLY Wk 1 Feb. 27 Orientation with Introductions & Safety Rules/Regulations March 1 Orientation with Pathogen
Title: Rapid PCR Tests for MRSA in Hospitalized Patients: Diagnostic Accuracy, Clinical and Cost-Effectiveness
 Title: Rapid PCR Tests for MRSA in Hospitalized Patients: Diagnostic Accuracy, Clinical and Cost-Effectiveness Date: 23 Oct 2007 Context and Policy Issues: Staphylococcus aureus (SA), a gram positive bacterium,
Title: Rapid PCR Tests for MRSA in Hospitalized Patients: Diagnostic Accuracy, Clinical and Cost-Effectiveness Date: 23 Oct 2007 Context and Policy Issues: Staphylococcus aureus (SA), a gram positive bacterium,
Received 4 September 2008/Returned for modification 24 October 2008/Accepted 25 December 2008
 JOURNAL OF CLINICAL MICROBIOLOGY, Mar. 2009, p. 758 764 Vol. 47, No. 3 0095-1137/09/$08.00 0 doi:10.1128/jcm.01714-08 Copyright 2009, American Society for Microbiology. All Rights Reserved. Multicenter
JOURNAL OF CLINICAL MICROBIOLOGY, Mar. 2009, p. 758 764 Vol. 47, No. 3 0095-1137/09/$08.00 0 doi:10.1128/jcm.01714-08 Copyright 2009, American Society for Microbiology. All Rights Reserved. Multicenter
320 MBIO Microbial Diagnosis. Aljawharah F. Alabbad Noorah A. Alkubaisi 2017
 320 MBIO Microbial Diagnosis Aljawharah F. Alabbad Noorah A. Alkubaisi 2017 Pathogens of the Urinary tract The urinary system is composed of organs that regulate the chemical composition and volume of
320 MBIO Microbial Diagnosis Aljawharah F. Alabbad Noorah A. Alkubaisi 2017 Pathogens of the Urinary tract The urinary system is composed of organs that regulate the chemical composition and volume of
ENG MYCO WELL D- ONE REV. 1.UN 29/09/2016 REF. MS01283 REF. MS01321 (COMPLETE KIT)
 ENG MYCO WELL D- ONE MYCO WELL D-ONE System for the presumptive identification and antimicrobial susceptibility test of urogenital mycoplasmas, Gardnerella vaginalis, Trichomonas vaginalis, Candida albicans
ENG MYCO WELL D- ONE MYCO WELL D-ONE System for the presumptive identification and antimicrobial susceptibility test of urogenital mycoplasmas, Gardnerella vaginalis, Trichomonas vaginalis, Candida albicans
Mt. San Antonio College Microbiology 22 Lab Schedule for Fall 2017 Tues/Thurs. Split Lab Sections ONLY
 Mt. San Antonio College Microbiology 22 Lab Schedule for Fall 2017 Tues/ Split Lab Sections ONLY Wk 1 Aug. 29 Orientation with Introductions & Safety Rules/Regulations Aug. 31 Orientation with Pathogen
Mt. San Antonio College Microbiology 22 Lab Schedule for Fall 2017 Tues/ Split Lab Sections ONLY Wk 1 Aug. 29 Orientation with Introductions & Safety Rules/Regulations Aug. 31 Orientation with Pathogen
Alberta Provincial Laboratory for Public Health, Edmonton, Alberta, Canada 1 ;
 JCM Accepts, published online ahead of print on 21 September 2011 J. Clin. Microbiol. doi:10.1128/jcm.05211-11 Copyright 2011, American Society for Microbiology and/or the Listed Authors/Institutions.
JCM Accepts, published online ahead of print on 21 September 2011 J. Clin. Microbiol. doi:10.1128/jcm.05211-11 Copyright 2011, American Society for Microbiology and/or the Listed Authors/Institutions.
ACCEPTED. Comparison of disk diffusion and agar dilution methods for erythromycin and
 AAC Accepts, published online ahead of print on January 00 Antimicrob. Agents Chemother. doi:./aac.000-0 Copyright 00, American Society for Microbiology and/or the Listed Authors/Institutions. All Rights
AAC Accepts, published online ahead of print on January 00 Antimicrob. Agents Chemother. doi:./aac.000-0 Copyright 00, American Society for Microbiology and/or the Listed Authors/Institutions. All Rights
Prevalence of Methicillin Resistant Staphylococcus aureus on computer mice on the campus of Southern Adventist University
 Southern Adventist Univeristy KnowledgeExchange@Southern Senior Research Projects Southern Scholars 4-9-2011 Prevalence of Methicillin Resistant Staphylococcus aureus on computer mice on the campus of
Southern Adventist Univeristy KnowledgeExchange@Southern Senior Research Projects Southern Scholars 4-9-2011 Prevalence of Methicillin Resistant Staphylococcus aureus on computer mice on the campus of
Development of a phenotypic method for fecal carriage detection of OXA-48-producing
 JCM Accepts, published online ahead of print on 11 May 2011 J. Clin. Microbiol. doi:10.1128/jcm.00055-11 Copyright 2011, American Society for Microbiology and/or the Listed Authors/Institutions. All Rights
JCM Accepts, published online ahead of print on 11 May 2011 J. Clin. Microbiol. doi:10.1128/jcm.00055-11 Copyright 2011, American Society for Microbiology and/or the Listed Authors/Institutions. All Rights
Guidance on screening and confirmation of carbapenem resistant Enterobacteriacae (CRE) December 12, 2011
 Guidance on screening and confirmation of carbapenem resistant Enterobacteriacae (CRE) December 12, 2011 Objectives: To discuss the guidelines for detection of CRE in the laboratory setting. To review
Guidance on screening and confirmation of carbapenem resistant Enterobacteriacae (CRE) December 12, 2011 Objectives: To discuss the guidelines for detection of CRE in the laboratory setting. To review
Staphylococci. Gram stain: gram positive cocci arranged in clusters.
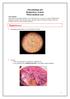 Microbiology lab Respiratory system Third medical year Lab contents: Gram positive bacteria (Staphylococcus and Streptococcus spp), two types of filamentous fungi (Aspergillus and Penicillium spp), and
Microbiology lab Respiratory system Third medical year Lab contents: Gram positive bacteria (Staphylococcus and Streptococcus spp), two types of filamentous fungi (Aspergillus and Penicillium spp), and
EDUCATIONAL COMMENTARY THROAT CULTURES LEARNING OUTCOMES. Upon completion of this exercise, the participant should be able to:
 EDUCATIONAL COMMENTARY THROAT CULTURES LEARNING OUTCOMES Upon completion of this exercise, the participant should be able to: distinguish three types of hemolysis produced by bacterial colonies. discuss
EDUCATIONAL COMMENTARY THROAT CULTURES LEARNING OUTCOMES Upon completion of this exercise, the participant should be able to: distinguish three types of hemolysis produced by bacterial colonies. discuss
Prevalence of and risk factors for MRSA colonization in HIV-positive outpatients in Singapore
 Kyaw et al. AIDS Research and Therapy 2012, 9:33 RESEARCH Open Access Prevalence of and risk factors for MRSA colonization in HIV-positive outpatients in Singapore Win Mar Kyaw 1*, Linda Kay Lee 1, Wong
Kyaw et al. AIDS Research and Therapy 2012, 9:33 RESEARCH Open Access Prevalence of and risk factors for MRSA colonization in HIV-positive outpatients in Singapore Win Mar Kyaw 1*, Linda Kay Lee 1, Wong
Validation of the MALDI-TOF for the Identification of Neisseria gonorrhoeae
 Proposal Validation of the MALDI-TOF for the Identification of Neisseria gonorrhoeae Laboratory Director Sandip H. Shah, Ph.D. 517-335-8063 517-335-8051 (fax) ShahS@Michigan.gov Acting Director, Division
Proposal Validation of the MALDI-TOF for the Identification of Neisseria gonorrhoeae Laboratory Director Sandip H. Shah, Ph.D. 517-335-8063 517-335-8051 (fax) ShahS@Michigan.gov Acting Director, Division
Michael Hombach, 1 * Gaby E. Pfyffer, 1 Malgorzata Roos, 2 and Katja Lucke 1
 JOURNAL OF CLINICAL MICROBIOLOGY, Nov. 2010, p. 3882 3887 Vol. 48, No. 11 0095-1137/10/$12.00 doi:10.1128/jcm.00670-10 Copyright 2010, American Society for Microbiology. All Rights Reserved. Detection
JOURNAL OF CLINICAL MICROBIOLOGY, Nov. 2010, p. 3882 3887 Vol. 48, No. 11 0095-1137/10/$12.00 doi:10.1128/jcm.00670-10 Copyright 2010, American Society for Microbiology. All Rights Reserved. Detection
Labquality External Quality Assesment Programmes General Bacteriology 1 1/2010
 Labquality External Quality Assesment Programmes General Bacteriology 1 1/2010 Photos and text: Markku Koskela, M.D., Ph.D. Clinical microbiology specialist Oulu, Finland Sample 1/2010 Pus from an infected
Labquality External Quality Assesment Programmes General Bacteriology 1 1/2010 Photos and text: Markku Koskela, M.D., Ph.D. Clinical microbiology specialist Oulu, Finland Sample 1/2010 Pus from an infected
The Challenge of Managing Staphylococcus aureus Bacteremia
 The Challenge of Managing Staphylococcus aureus Bacteremia M A R G A R E T G R A Y B S P F C S H P C L I N I C A L P R A C T I C E M A N A G E R N O R T H / I D P H A R M A C I S T A L B E R T A H E A
The Challenge of Managing Staphylococcus aureus Bacteremia M A R G A R E T G R A Y B S P F C S H P C L I N I C A L P R A C T I C E M A N A G E R N O R T H / I D P H A R M A C I S T A L B E R T A H E A
Clinical Failure of Vancomycin Treatment of Staphylococcus aureus Infection in a Tertiary Care Hospital in Southern Brazil
 224 BJID 2003; 7 (June) Clinical Failure of Vancomycin Treatment of Staphylococcus aureus Infection in a Tertiary Care Hospital in Southern Brazil Larissa Lutz, Adão Machado, Nadia Kuplich and Afonso Luís
224 BJID 2003; 7 (June) Clinical Failure of Vancomycin Treatment of Staphylococcus aureus Infection in a Tertiary Care Hospital in Southern Brazil Larissa Lutz, Adão Machado, Nadia Kuplich and Afonso Luís
Overview of Mycobacterial Culture, Identification, and Drug Susceptibility Testing
 Overview of Mycobacterial Culture, Identification, and Drug Susceptibility Testing 1. Essentials for the Mycobacteriology Laboratory: Promoting Quality Practices 1.1 Overview: Mycobacterial Culture, Identification,
Overview of Mycobacterial Culture, Identification, and Drug Susceptibility Testing 1. Essentials for the Mycobacteriology Laboratory: Promoting Quality Practices 1.1 Overview: Mycobacterial Culture, Identification,
1430 West McCoy Lane Santa Maria, CA p:
 091217TR HardyCHROM BluEcoli 1 HardyCHROM Candida 2 HardyCHROM ECC 3 HardyCHROM ESBL 4 HardyCHROM Listeria 5 HardyCHROM MRSA 6 HardyCHROM O157 7 HardyCHROM Salmonella 8 HardyCHROM SS NoPRO 9 HardyCHROM
091217TR HardyCHROM BluEcoli 1 HardyCHROM Candida 2 HardyCHROM ECC 3 HardyCHROM ESBL 4 HardyCHROM Listeria 5 HardyCHROM MRSA 6 HardyCHROM O157 7 HardyCHROM Salmonella 8 HardyCHROM SS NoPRO 9 HardyCHROM
Synergy of beta-lactams with vancomycin against methicillin-resistant. Staphylococcus aureus: correlation of the disk diffusion and the
 JCM Accepted Manuscript Posted Online 16 December 2015 J. Clin. Microbiol. doi:10.1128/jcm.01779-15 Copyright 2015, American Society for Microbiology. All Rights Reserved. 1 2 3 Synergy of beta-lactams
JCM Accepted Manuscript Posted Online 16 December 2015 J. Clin. Microbiol. doi:10.1128/jcm.01779-15 Copyright 2015, American Society for Microbiology. All Rights Reserved. 1 2 3 Synergy of beta-lactams
AN IMMUNOASSAY TEST FOR THE QUALITATIVE DETECTION OF STREP A ANTIGEN IN THROAT SWAB SPECIMENS
 CLARITY Strep A Dipsticks FOR LABORATORY AND PROFESSIONAL USE AN IMMUNOASSAY TEST FOR THE QUALITATIVE DETECTION OF STREP A ANTIGEN IN THROAT SWAB SPECIMENS CLIA COMPLEXITY: Waived CLARITY Strep A Dipsticks:
CLARITY Strep A Dipsticks FOR LABORATORY AND PROFESSIONAL USE AN IMMUNOASSAY TEST FOR THE QUALITATIVE DETECTION OF STREP A ANTIGEN IN THROAT SWAB SPECIMENS CLIA COMPLEXITY: Waived CLARITY Strep A Dipsticks:
SUPPLEMENTAL TESTING. Tan Thean Yen
 SUPPLEMETAL TESTG Tan Thean Yen To Supplement Definition: add as a supplement to what seems insufficient "supplement your diet" Why supplement? urrent methods don t work well Additional information provided
SUPPLEMETAL TESTG Tan Thean Yen To Supplement Definition: add as a supplement to what seems insufficient "supplement your diet" Why supplement? urrent methods don t work well Additional information provided
Performance of the BD GeneOhm MRSA Achromopeptidase (ACP) Extraction for Real-time PCR Detection of Methicillin-resistant
 JCM Accepts, published online ahead of print on 20 April 2011 J. Clin. Microbiol. doi:10.1128/jcm.02431-10 Copyright 2011, American Society for Microbiology and/or the Listed Authors/Institutions. All
JCM Accepts, published online ahead of print on 20 April 2011 J. Clin. Microbiol. doi:10.1128/jcm.02431-10 Copyright 2011, American Society for Microbiology and/or the Listed Authors/Institutions. All
IP Lab Webinar 8/23/2012
 2 What Infection Preventionists need to know about the Laboratory Anne Maher, MS, M(ASCP), CIC Richard VanEnk PhD, CIC 1 Objectives Describe what the laboratory can do for you; common laboratory tests
2 What Infection Preventionists need to know about the Laboratory Anne Maher, MS, M(ASCP), CIC Richard VanEnk PhD, CIC 1 Objectives Describe what the laboratory can do for you; common laboratory tests
Sensitivity of Surveillance Testing for Multidrug-Resistant Gram-Negative Bacteria in the
 JCM Accepts, published online ahead of print on 20 August 2014 J. Clin. Microbiol. doi:10.1128/jcm.02369-14 Copyright 2014, American Society for Microbiology. All Rights Reserved. 1 2 Sensitivity of Surveillance
JCM Accepts, published online ahead of print on 20 August 2014 J. Clin. Microbiol. doi:10.1128/jcm.02369-14 Copyright 2014, American Society for Microbiology. All Rights Reserved. 1 2 Sensitivity of Surveillance
Anne Beall BS, MT, Solutions Consultant, biomérieux, Durham, NC
 Anne Beall BS, MT, Solutions Consultant, biomérieux, Durham, NC List 5 of the most common problems in microbiology laboratories seen today Discovering those problems in your laboratory Gathering data Possible
Anne Beall BS, MT, Solutions Consultant, biomérieux, Durham, NC List 5 of the most common problems in microbiology laboratories seen today Discovering those problems in your laboratory Gathering data Possible
Weds. Date. Aug. 26. Sept. 2
 Mt.SanAntonioCollege Microbiology 22 Lab Schedule for Fall 2015 Mon./ Split Lab Sections ONLY Wk. Mon. 1 Aug. 24 Orientation with Introductions & Safety Rules/Regulations 2 Aug. 31 Exercise #1: The Microscope
Mt.SanAntonioCollege Microbiology 22 Lab Schedule for Fall 2015 Mon./ Split Lab Sections ONLY Wk. Mon. 1 Aug. 24 Orientation with Introductions & Safety Rules/Regulations 2 Aug. 31 Exercise #1: The Microscope
Infectious Disease Diagnostics in the Geisinger Health System
 10X Essentials Infectious Disease Diagnostics in the Geisinger Health System NEW: Rapid GeneXpert PCR NEW Test Codes: MRSAP, MRSA Screen PCR CDIFP, C. difficile/ Epi PCR EVP, Enterovirus PCR Specimens
10X Essentials Infectious Disease Diagnostics in the Geisinger Health System NEW: Rapid GeneXpert PCR NEW Test Codes: MRSAP, MRSA Screen PCR CDIFP, C. difficile/ Epi PCR EVP, Enterovirus PCR Specimens
Laboratory Evaluation of the BD MAX TM MRSA Assay
 JCM Accepts, published online ahead of print on 14 May 2014 J. Clin. Microbiol. doi:10.1128/jcm.00237-14 Copyright 2014, American Society for Microbiology. All Rights Reserved. 1 Laboratory Evaluation
JCM Accepts, published online ahead of print on 14 May 2014 J. Clin. Microbiol. doi:10.1128/jcm.00237-14 Copyright 2014, American Society for Microbiology. All Rights Reserved. 1 Laboratory Evaluation
TM SS Hardy tr
 TM 013114tr You don t dress like they did in 1968... so why are you still culturing like they did in 1968? The year was 1968, and bell bottoms and tie dye were all the rage. Also in 1968, Hektoen Enteric
TM 013114tr You don t dress like they did in 1968... so why are you still culturing like they did in 1968? The year was 1968, and bell bottoms and tie dye were all the rage. Also in 1968, Hektoen Enteric
Enrichment culture of CSF is of limited value in the diagnosis of neonatal meningitis
 Enrichment culture of CSF is of limited value in the diagnosis of neonatal S. H. Chaudhry, D. Wagstaff, Anupam Gupta, I. C. Bowler, D. P. Webster To cite this version: S. H. Chaudhry, D. Wagstaff, Anupam
Enrichment culture of CSF is of limited value in the diagnosis of neonatal S. H. Chaudhry, D. Wagstaff, Anupam Gupta, I. C. Bowler, D. P. Webster To cite this version: S. H. Chaudhry, D. Wagstaff, Anupam
Comparison of Different Phenotypic Approaches to Screen and Detect mecc-harboring Methicillin-Resistant Staphylococcus aureus
 Edinburgh Research Explorer Comparison of Different Phenotypic Approaches to Screen and Detect mecc-harboring Methicillin-Resistant Staphylococcus aureus Citation for published version: Kriegeskorte, A,
Edinburgh Research Explorer Comparison of Different Phenotypic Approaches to Screen and Detect mecc-harboring Methicillin-Resistant Staphylococcus aureus Citation for published version: Kriegeskorte, A,
THE CARRIER STATE: METHICILLIN - RESISTANT STAPHYLOCOCCUS AUREUS A HOSPITAL STUDY "SCREENING OF HOSPITAL PERSONNEL" FOR NASAL CARRIAGE OF STAPH AUREUS
 THE CARRIER STATE: METHICILLIN - RESISTANT STAPHYLOCOCCUS AUREUS A HOSPITAL STUDY "SCREENING OF HOSPITAL PERSONNEL" FOR NASAL CARRIAGE OF STAPH AUREUS Abstract Pages with reference to book, From 35 To
THE CARRIER STATE: METHICILLIN - RESISTANT STAPHYLOCOCCUS AUREUS A HOSPITAL STUDY "SCREENING OF HOSPITAL PERSONNEL" FOR NASAL CARRIAGE OF STAPH AUREUS Abstract Pages with reference to book, From 35 To
Educational Workshops 2016
 Educational Workshops 2016 Keynote CPE Screening We are grateful to Dr Andrew Dodgson, Consultant Microbiologist, Public Health England and Central Manchester Hospitals NHS Foundation Trust Terminology
Educational Workshops 2016 Keynote CPE Screening We are grateful to Dr Andrew Dodgson, Consultant Microbiologist, Public Health England and Central Manchester Hospitals NHS Foundation Trust Terminology
Received 21 October 2005/Returned for modification 23 December 2005/Accepted 21 May 2006
 JOURNAL OF CLINICAL MICROBIOLOGY, Aug. 2006, p. 2904 2908 Vol. 44, No. 8 0095-1137/06/$08.00 0 doi:10.1128/jcm.02211-05 Copyright 2006, American Society for Microbiology. All Rights Reserved. Concurrent
JOURNAL OF CLINICAL MICROBIOLOGY, Aug. 2006, p. 2904 2908 Vol. 44, No. 8 0095-1137/06/$08.00 0 doi:10.1128/jcm.02211-05 Copyright 2006, American Society for Microbiology. All Rights Reserved. Concurrent
An algorithm based on one or two nasal samples is accurate to identify persistent nasal carriers of Staphylococcus aureus
 ORIGINAL ARTICLE BACTERIOLOGY An algorithm based on one or two nasal samples is accurate to identify persistent nasal carriers of Staphylococcus aureus P. O. Verhoeven 1,2, F. Grattard 1,2, A. Carricajo
ORIGINAL ARTICLE BACTERIOLOGY An algorithm based on one or two nasal samples is accurate to identify persistent nasal carriers of Staphylococcus aureus P. O. Verhoeven 1,2, F. Grattard 1,2, A. Carricajo
Multi-clonal origin of macrolide-resistant Mycoplasma pneumoniae isolates. determined by multiple-locus variable-number tandem-repeat analysis
 JCM Accepts, published online ahead of print on 30 May 2012 J. Clin. Microbiol. doi:10.1128/jcm.00678-12 Copyright 2012, American Society for Microbiology. All Rights Reserved. 1 2 Multi-clonal origin
JCM Accepts, published online ahead of print on 30 May 2012 J. Clin. Microbiol. doi:10.1128/jcm.00678-12 Copyright 2012, American Society for Microbiology. All Rights Reserved. 1 2 Multi-clonal origin
Labquality External Quality Assessment Programmes General Bacteriology 1 3/2013
 Labquality External Quality Assessment Programmes General Bacteriology 1 3/2013 Photos and text: Markku Koskela, M.D., Ph.D. Clinical microbiology specialist Oulu, Finland Sample 21/2013 Maxillary sinus
Labquality External Quality Assessment Programmes General Bacteriology 1 3/2013 Photos and text: Markku Koskela, M.D., Ph.D. Clinical microbiology specialist Oulu, Finland Sample 21/2013 Maxillary sinus
Receipt within 1 day of specimen collection. Report AFB b smear result within 1 day from receipt of specimen
 Recommendation Promote rapid delivery of specimens to the laboratory Use fluorescent acid-fast staining and promptly transmit results by phone, FAX, or electronically Identify growth as acid-fast and use
Recommendation Promote rapid delivery of specimens to the laboratory Use fluorescent acid-fast staining and promptly transmit results by phone, FAX, or electronically Identify growth as acid-fast and use
Retrospective and Prospective Verification of the Cepheid Xpert Flu Assay
 JCM Accepts, published online ahead of print on 20 July 2011 J. Clin. Microbiol. doi:10.1128/jcm.01162-11 Copyright 2011, American Society for Microbiology and/or the Listed Authors/Institutions. All Rights
JCM Accepts, published online ahead of print on 20 July 2011 J. Clin. Microbiol. doi:10.1128/jcm.01162-11 Copyright 2011, American Society for Microbiology and/or the Listed Authors/Institutions. All Rights
GUIDE TO INFECTION CONTROL IN THE HOSPITAL. Carbapenem-resistant Enterobacteriaceae
 GUIDE TO INFECTION CONTROL IN THE HOSPITAL CHAPTER 47: Carbapenem-resistant Enterobacteriaceae Authors E-B Kruse, MD H. Wisplinghoff, MD Chapter Editor Michelle Doll, MD, MPH) Topic Outline Key Issue Known
GUIDE TO INFECTION CONTROL IN THE HOSPITAL CHAPTER 47: Carbapenem-resistant Enterobacteriaceae Authors E-B Kruse, MD H. Wisplinghoff, MD Chapter Editor Michelle Doll, MD, MPH) Topic Outline Key Issue Known
Validation of Vitek version 7.01 software for testing staphylococci against vancomycin
 Diagnostic Microbiology and Infectious Disease 43 (2002) 135 140 www.elsevier.com/locate/diagmicrobio Validation of Vitek version 7.01 software for testing staphylococci against vancomycin P.M. Raney a,
Diagnostic Microbiology and Infectious Disease 43 (2002) 135 140 www.elsevier.com/locate/diagmicrobio Validation of Vitek version 7.01 software for testing staphylococci against vancomycin P.M. Raney a,
EUCAST Frequently Asked Questions. by author. Rafael Cantón Hospital Universitario Ramón y Cajal, Madrid, Spain EUCAST Clinical Data Coordinator
 EUCAST Frequently Asked Questions Rafael Cantón Hospital Universitario Ramón y Cajal, Madrid, Spain EUCAST Clinical Data Coordinator Erika Matuschek EUCAST Development Laboratory, Växjö Sweden Monday 24
EUCAST Frequently Asked Questions Rafael Cantón Hospital Universitario Ramón y Cajal, Madrid, Spain EUCAST Clinical Data Coordinator Erika Matuschek EUCAST Development Laboratory, Växjö Sweden Monday 24
Staphylococci. What s to be Covered. Clinical Scenario #1
 Staphylococci Micrococcus, which, when limited in its extent and activity, causes acute suppurative inflammation (phlegmon), produces, when more extensive and intense in its action on the human system,
Staphylococci Micrococcus, which, when limited in its extent and activity, causes acute suppurative inflammation (phlegmon), produces, when more extensive and intense in its action on the human system,
Received 30 March 2005; returned 16 June 2005; revised 8 September 2005; accepted 12 September 2005
 Journal of Antimicrobial Chemotherapy (2005) 56, 1047 1052 doi:10.1093/jac/dki362 Advance Access publication 20 October 2005 Evaluation of PPI-0903M (T91825), a novel cephalosporin: bactericidal activity,
Journal of Antimicrobial Chemotherapy (2005) 56, 1047 1052 doi:10.1093/jac/dki362 Advance Access publication 20 October 2005 Evaluation of PPI-0903M (T91825), a novel cephalosporin: bactericidal activity,
What s to be Covered. Microbiology of staphylococci Epidemiology of S. aureus infections Pathogenesis of S. aureus infections
 Staphylococci Micrococcus, which, when limited in its extent and activity, causes acute suppurative inflammation (phlegmon), produces, when more extensive and intense in its action on the human system,
Staphylococci Micrococcus, which, when limited in its extent and activity, causes acute suppurative inflammation (phlegmon), produces, when more extensive and intense in its action on the human system,
D. A. Leigh and G. Joy. Department of Microbiology, Wycombe General Hospital, High Wycombe, Bucks HP11 2TT, UK
 Journal of Antimicrobial Chemotherapy (13) 31, 0-17 Treatment of familial staphylococcal infection comparison of mnpirocin nasal ointment and chlorhexidine/neomycin (aseptin) cream in eradication of nasal
Journal of Antimicrobial Chemotherapy (13) 31, 0-17 Treatment of familial staphylococcal infection comparison of mnpirocin nasal ointment and chlorhexidine/neomycin (aseptin) cream in eradication of nasal
Evaluation of methicillin-resistant Staphylococcus aureus (MRSA) colonization in pigs and people that work with pigs in Ontario Veterinary College
 Evaluation of methicillin-resistant Staphylococcus aureus (MRSA) colonization in pigs and people that work with pigs in Ontario Veterinary College Final Report September 2007 This research has been possible
Evaluation of methicillin-resistant Staphylococcus aureus (MRSA) colonization in pigs and people that work with pigs in Ontario Veterinary College Final Report September 2007 This research has been possible
Pseudomonas aeruginosa
 JOURNAL OF CLINICAL MICROBIOLOGY, July 1983, p. 16-164 95-1137/83/716-5$2./ Copyright C) 1983, American Society for Microbiology Vol. 18, No. 1 A Three-Year Study of Nosocomial Infections Associated with
JOURNAL OF CLINICAL MICROBIOLOGY, July 1983, p. 16-164 95-1137/83/716-5$2./ Copyright C) 1983, American Society for Microbiology Vol. 18, No. 1 A Three-Year Study of Nosocomial Infections Associated with
A.F. GENITAL SYSTEM. ITEMS NECESSARY BUT NOT INCLUDED IN THE KIT A.F. GENITAL SYSTEM Reagent (ref ) Mycoplasma Transport Broth (ref.
 A.F. GENITAL SYSTEM ENGLISH System for detection, count and susceptibility test of pathogenic urogenital microorganisms DESCRIPTION A.F. GENITAL SYSTEM is a 24-well system containing desiccated biochemical
A.F. GENITAL SYSTEM ENGLISH System for detection, count and susceptibility test of pathogenic urogenital microorganisms DESCRIPTION A.F. GENITAL SYSTEM is a 24-well system containing desiccated biochemical
Latex Agglutination Kits
 Latex Agglutination Kits 062916tr Headquarters A Culture of Service 1430 West McCoy Lane Santa Maria, CA 93455 800.266.2222 : phone Sales@HardyDiagnostics.com www.hardydiagnostics.com Distribution Centers
Latex Agglutination Kits 062916tr Headquarters A Culture of Service 1430 West McCoy Lane Santa Maria, CA 93455 800.266.2222 : phone Sales@HardyDiagnostics.com www.hardydiagnostics.com Distribution Centers
Rapid-VIDITEST. Strep A Card. One step Strep A Card for the detection of Group A Streptococcal antigen from throat swabs or culture.
 Rapid-VIDITEST Strep A Card One step Strep A Card for the detection of Group A Streptococcal antigen from throat swabs or culture. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II 365, Vestec,
Rapid-VIDITEST Strep A Card One step Strep A Card for the detection of Group A Streptococcal antigen from throat swabs or culture. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II 365, Vestec,
Performance of Various Testing Methodologies for Detection of Heteroresistant Vancomycin-Intermediate Staphylococcus aureus in Bloodstream Isolates
 JOURNAL OF CLINICAL MICROBIOLOGY, Apr. 2011, p. 1489 1494 Vol. 49, No. 4 0095-1137/11/$12.00 doi:10.1128/jcm.02302-10 Copyright 2011, American Society for Microbiology. All Rights Reserved. Performance
JOURNAL OF CLINICAL MICROBIOLOGY, Apr. 2011, p. 1489 1494 Vol. 49, No. 4 0095-1137/11/$12.00 doi:10.1128/jcm.02302-10 Copyright 2011, American Society for Microbiology. All Rights Reserved. Performance
International Journal of Pharma and Bio Sciences
 Research Article Microbiology International Journal of Pharma and Bio Sciences ISSN 0975-6299 DETECTION OF MRSA FROM NASAL CARRIER AMONG DIABETIC FOOT ULCER PATIENTS IN TERTIARY CARE HOSPITAL PUDUCHERRY
Research Article Microbiology International Journal of Pharma and Bio Sciences ISSN 0975-6299 DETECTION OF MRSA FROM NASAL CARRIER AMONG DIABETIC FOOT ULCER PATIENTS IN TERTIARY CARE HOSPITAL PUDUCHERRY
Redefine Performance. BD Veritor. System Revolutionizes Testing at the Point of Care. Fast. Streamlined Workflow Requires minimal hands-on time
 CLIA WAIVED Redefine Performance System BD Veritor System Revolutionizes Testing at the Point of Care Accurate The first CLIA-waived Digital Immunoassay (DIA), a new category of diagnostic tests where
CLIA WAIVED Redefine Performance System BD Veritor System Revolutionizes Testing at the Point of Care Accurate The first CLIA-waived Digital Immunoassay (DIA), a new category of diagnostic tests where
The Clinical Significance of Blood Cultures. Presented BY; Cindy Winfrey, MSN, RN, CIC, DON- LTC TM, VA- BC TM
 The Clinical Significance of Blood Cultures Presented BY; Cindy Winfrey, MSN, RN, CIC, DON- LTC TM, VA- BC TM OVERVIEW Blood cultures are considered an important laboratory tool used to diagnose serious
The Clinical Significance of Blood Cultures Presented BY; Cindy Winfrey, MSN, RN, CIC, DON- LTC TM, VA- BC TM OVERVIEW Blood cultures are considered an important laboratory tool used to diagnose serious
Rapid-VIDITEST. Strep A Blister. One step Strep A Blister for the detection of Group A Streptococcal antigen from throat swabs or culture.
 Rapid-VIDITEST Strep A Blister One step Strep A Blister for the detection of Group A Streptococcal antigen from throat swabs or culture. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II
Rapid-VIDITEST Strep A Blister One step Strep A Blister for the detection of Group A Streptococcal antigen from throat swabs or culture. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II
Phenotypic Detection of Methicillin Resistance in Staphylococcus aureus by Disk Diffusion Testing and Etest on Mueller-Hinton Agar
 JOURNAL OF CLINICAL MICROBIOLOGY, Dec. 2006, p. 4395 4399 Vol. 44, No. 12 0095-1137/06/$08.00 0 doi:10.1128/jcm.01411-06 Copyright 2006, American Society for Microbiology. All Rights Reserved. Phenotypic
JOURNAL OF CLINICAL MICROBIOLOGY, Dec. 2006, p. 4395 4399 Vol. 44, No. 12 0095-1137/06/$08.00 0 doi:10.1128/jcm.01411-06 Copyright 2006, American Society for Microbiology. All Rights Reserved. Phenotypic
Routine endotracheal cultures for the prediction of sepsis in ventilated babies
 Archives of Disease in Childhood, 1989, 64, 34-38 Routine endotracheal cultures for the prediction of sepsis in ventilated babies T A SLAGLE, E M BIFANO, J W WOLF, AND S J GROSS Department of Pediatrics,
Archives of Disease in Childhood, 1989, 64, 34-38 Routine endotracheal cultures for the prediction of sepsis in ventilated babies T A SLAGLE, E M BIFANO, J W WOLF, AND S J GROSS Department of Pediatrics,
Stool bench. Cultures: SARAH
 Stool bench The bacteria found in stool are representative of the bacteria that are present in the digestive system (gastrointestinal tract). Certain bacteria and fungi called normal flora inhabit everyone's
Stool bench The bacteria found in stool are representative of the bacteria that are present in the digestive system (gastrointestinal tract). Certain bacteria and fungi called normal flora inhabit everyone's
Prevalence of Streptococcus pneumoniae in Respiratory Samples 1. from Patients with Tracheostomy in a Long-Term Care Facility 2
 JCM Accepts, published online ahead of print on 25 July 2012 J. Clin. Microbiol. doi:10.1128/jcm.00763-12 Copyright 2012, American Society for Microbiology. All Rights Reserved. 1 Prevalence of Streptococcus
JCM Accepts, published online ahead of print on 25 July 2012 J. Clin. Microbiol. doi:10.1128/jcm.00763-12 Copyright 2012, American Society for Microbiology. All Rights Reserved. 1 Prevalence of Streptococcus
In vitro assessment of dual drug combinations to inhibit growth of Neisseria gonorrhoeae
 AAC Accepted Manuscript Posted Online 26 January 2015 Antimicrob. Agents Chemother. doi:10.1128/aac.04127-14 Copyright 2015, American Society for Microbiology. All Rights Reserved. 1 2 In vitro assessment
AAC Accepted Manuscript Posted Online 26 January 2015 Antimicrob. Agents Chemother. doi:10.1128/aac.04127-14 Copyright 2015, American Society for Microbiology. All Rights Reserved. 1 2 In vitro assessment
From the labo to the ICU: Surveillance cultures in daily ICU practice. Pieter Depuydt MD PhD Dept. Intensive Care Ghent University Hospital
 From the labo to the ICU: Surveillance cultures in daily ICU practice Pieter Depuydt MD PhD Dept. Intensive Care Ghent University Hospital Question 1: What is the current practice of surveillance cultures
From the labo to the ICU: Surveillance cultures in daily ICU practice Pieter Depuydt MD PhD Dept. Intensive Care Ghent University Hospital Question 1: What is the current practice of surveillance cultures
LESSON 2.6 WORKBOOK Diagnosing infections, and, what s up your nose?
 Staphylococcus aureus Morphology: The physical form or structure of a microbe.. LESSON 2.6 WORKBOOK Diagnosing infections, and, what s up your nose? Now we have discussed the different requirements that
Staphylococcus aureus Morphology: The physical form or structure of a microbe.. LESSON 2.6 WORKBOOK Diagnosing infections, and, what s up your nose? Now we have discussed the different requirements that
Diagnosis of TB: Laboratory Ken Jost Tuesday April 1, 2014
 TB Nurse Case Management San Antonio, Texas April 1 3, 2014 Diagnosis of TB: Laboratory Ken Jost Tuesday April 1, 2014 Ken Jost, BA has the following disclosures to make: No conflict of interests No relevant
TB Nurse Case Management San Antonio, Texas April 1 3, 2014 Diagnosis of TB: Laboratory Ken Jost Tuesday April 1, 2014 Ken Jost, BA has the following disclosures to make: No conflict of interests No relevant
Microbiology products. Liofilchem Chromatic
 ET Microbiology products M IC R O PL A N www.liofilchem.net Liofilchem Chromatic c h ro m o g e n i c c u l t u re m e d i a LIOFILCHEM s.r.l. Via Scozia, Zona Industriale 64026 Roseto degli Abruzzi (Te)
ET Microbiology products M IC R O PL A N www.liofilchem.net Liofilchem Chromatic c h ro m o g e n i c c u l t u re m e d i a LIOFILCHEM s.r.l. Via Scozia, Zona Industriale 64026 Roseto degli Abruzzi (Te)
Urine bench. John Ferguson Sept 2013
 Urine bench John Ferguson Sept 2013 Overview Specimen collection- separate presentation Urinalysis: protein, blood, white cells, nitrite Microscopy- crystals and casts- separate presentations quantitative
Urine bench John Ferguson Sept 2013 Overview Specimen collection- separate presentation Urinalysis: protein, blood, white cells, nitrite Microscopy- crystals and casts- separate presentations quantitative
JCM Accepts, published online ahead of print on 5 March 2008 J. Clin. Microbiol. doi: /jcm
 JCM Accepts, published online ahead of print on March 00 J. Clin. Microbiol. doi:0./jcm.00-0 Copyright 00, American Society for Microbiology and/or the Listed Authors/Institutions. All Rights Reserved.
JCM Accepts, published online ahead of print on March 00 J. Clin. Microbiol. doi:0./jcm.00-0 Copyright 00, American Society for Microbiology and/or the Listed Authors/Institutions. All Rights Reserved.
Respiratory Pathogen Panel TEM-PCR Test Code:
 Respiratory Pathogen Panel TEM-PCR Test Code: 220000 Tests in this Panel Enterovirus group Human bocavirus Human coronavirus (4 types) Human metapneumovirus Influenza A - Human influenza Influenza A -
Respiratory Pathogen Panel TEM-PCR Test Code: 220000 Tests in this Panel Enterovirus group Human bocavirus Human coronavirus (4 types) Human metapneumovirus Influenza A - Human influenza Influenza A -
HOSPITAL INFECTION CONTROL
 HOSPITAL INFECTION CONTROL Objectives To be able to define hospital acquired infections discuss the sources and routes of transmission of infections in a hospital describe methods of prevention and control
HOSPITAL INFECTION CONTROL Objectives To be able to define hospital acquired infections discuss the sources and routes of transmission of infections in a hospital describe methods of prevention and control
Gram Positive Coccus Staphylococci Dr. Hala Al Daghistani
 Medical bacteriology Gram Positive Coccus Staphylococci Dr. Hala Al Daghistani The Staphylococci are gram-positive spherical cells, nonmotile, usually arranged in grapelike irregular clusters. Some are
Medical bacteriology Gram Positive Coccus Staphylococci Dr. Hala Al Daghistani The Staphylococci are gram-positive spherical cells, nonmotile, usually arranged in grapelike irregular clusters. Some are
PNEUMOCOCCUS VALENT LATEX KIT
 PNEUMOCOCCUS 7-10-13-VALENT LATEX KIT Latex particles coated with pneumococcal antiserum raised in rabbits for in vitro diagnostic use Application The Pneumococcus 7-10-13-valent Latex Kit is a ready to
PNEUMOCOCCUS 7-10-13-VALENT LATEX KIT Latex particles coated with pneumococcal antiserum raised in rabbits for in vitro diagnostic use Application The Pneumococcus 7-10-13-valent Latex Kit is a ready to
Upper Respiratory Infections. Mehreen Arshad, MD Assistant Professor Pediatric Infectious Diseases Duke University
 Upper Respiratory Infections Mehreen Arshad, MD Assistant Professor Pediatric Infectious Diseases Duke University Disclosures None Objectives Know the common age- and season-specific causes of pharyngitis
Upper Respiratory Infections Mehreen Arshad, MD Assistant Professor Pediatric Infectious Diseases Duke University Disclosures None Objectives Know the common age- and season-specific causes of pharyngitis
