Case 1. FDA, Legal, and EBAA Medical Standards Panel. Case 1. Case 1 6/25/2012. Samuel B. Barone, M.D. Office of Cellular, Tissue, and Gene Therapies
|
|
|
- Juliana Rice
- 5 years ago
- Views:
Transcription
1 Case 1 FDA, Legal, and EBAA Medical Standards Panel Samuel B. Barone, M.D. Office of Cellular, Tissue, and Gene Therapies Medical Directors Symposium Eye Bank Association of America Annual Meeting 47 y/o female cornea donor died after three-day hospital admission secondary to complications from end stage liver disease (ETOH related) Date of admit: 1/2; Date of death: 1/5 Hospital course included 2 units PRBC Hemodilution algorithm was acceptable Donor non-reactive for all infectious disease testing (RPR, HBsAg, Hep B Core Ab, HCV Ab, HIV I/II, HTLV I/II, NAT Ultrio-HIV/HCV/HBV) June 23, Case 1 Received PRBCs were donated 12/29 all infectious disease testing: non-reactive PRBCs transfused to the cornea donor 3-days after donation (1/3) One cornea was transplanted Case 1 Approximately 3 ½ months later the eye bank was notified that the PRBC blood donor had tested positive for HIV. Blood donor returned for repeat donation approximately 110 days later, testing then indicated the blood donor to be infected with HIV (HIV I/II repeatedly reactive, HIV-1 confirmatory positive, HIV NAT reactive) 3 4 1
2 Case 1 - Question HIV Testing What is the likelihood that the donor was in the window period and was infected with HIV at the time of the recovery? Case 1 - Question HCT/P Deviation A Biological Product Deviation Report was generated to the FDA as required. Would this case be considered a recall by the FDA, since the eye bank was in compliance with Donor Eligibility requirements at the time tissue was distributed? (1) An event that represents a deviation from applicable regulations... or from applicable standards or established specifications that relate to the prevention of communicable disease transmission or HCT/P contamination (2) An unexpected or unforeseeable event that may relate to the transmission or potential transmission of a communicable disease 7 21 CFR (dd) 8 2
3 Reports of HCT/P Deviations (1) You must investigate all HCT/P deviations related to a distributed HCT/P for which you performed a manufacturing step. Reports of HCT/P Deviations cont. (2) You must report any such HCT/P deviation relating to the core CGTP requirements, if the HCT/P deviation occurred in your facility or in a facility that performed a manufacturing step for you under contract, agreement, or other arrangement. Each report must contain a description of the HCT/P deviation, information relevant to the event and the manufacture of the HCT/P involved, and information on all follow-up actions that have been or will be taken in response to the HCT/P deviation (e.g., recalls). 21 CFR (b) 9 21 CFR (b) 10 Reports of HCT/P Deviations cont. Recall (3) You must report each such HCT/P deviation that relates to a core CGTP requirement on Form FDA within 45 days of the discovery of the event... A firm s removal or correction of a marketed product that the Food and Drug Administration considers to be in violation of the laws it administers and against which the agency would initiate legal action, e.g. seizure. Recall does not include a market withdrawal or a stock recovery. 21 CFR (b) CFR 7.3(g) 12 3
4 Relevant Medical Records Market Withdrawal A collection of documents that includes a current donor medical history interview; a current report of the physical assessment of a cadaveric donor or the physical examination of a living donor; and, if available, the following: (1) Laboratory test results (other than results of testing for relevant communicable disease agents required under this subpart); (2) Medical records; (3) Coroner and autopsy reports; and (4) Records or other information received from any source pertaining to risk factors for relevant communicable disease (e.g., social behavior, clinical signs and symptoms of relevant communicable disease, and treatments related to medical conditions suggestive of risk for relevant communicable disease). A firm's removal or correction of a distributed product which involves a minor violation that would not be subject to legal action by the Food and Drug Administration or which involves no violation, e.g., normal stock rotation practices, routine equipment adjustments and repairs, etc. 21 CFR (s) CFR 7.3(j) 14 Quality Program: Information Sharing Ensuring that procedures exist for receiving, investigating, evaluating, and documenting information relating to core CGTP requirements, including complaints, and for sharing any information pertaining to the possible contamination of the HCT/P or the potential for transmission of a communicable disease by the HCT/P with the following: (i) Other establishments that are known to have recovered HCT/Ps from the same donor; Quality Program: Information Sharing cont. (ii) Other establishments that are known to have performed manufacturing steps with respect to the same HCT/P; and (iii) Relating to consignees, in the case of such information received after the HCT/P is made available for distribution, shipped to the consignee, or administered to the recipient, procedures must include provisions for assessing risk and appropriate follow-up, and evaluating the effect this information has on the HCT/P and for the notification of all entities to whom the affected HCT/P was distributed, the quarantine and recall of the HCT/P, and/or reporting to FDA, as necessary. 21 CFR (b)(2) CFR (b)(2) 16 4
5 Case 2 Case 2 - Questions Eye bank received a referral of a 37 y/o male who expired from severe trauma following a MVA. Medical/social history is unremarkable except for a history of Bell s palsy. Referral was declined as history of Bell s palsy what was determined to be an identified risk factor for human transmissible spongiform encephalopathy (TSE). Would you rule out a donor due to a history of Bell's palsy? Would you exclude a donor who was in the acute phase with possible active viral infection? Screening for Transmissible Spongiform Encephalopathy You should determine to be ineligible... (20) Persons who have been diagnosed with dementia or any degenerative or demyelinating disease of the central nervous system or other neurological disease of unknown etiology... Eligibility Determination for Donors Of HCT/Ps Sec. IV.E. 19 Facial Nerve (CN VII) Palsy Supranuclear Brain stem (pons) Tumor Vascular Peripheral Bell s palsy Trauma Peripheral cont. Infection HZV/HSV, HIV, Lyme, syphilis Otitis media/externa Sarcoidosis Tumor Acoustic neuroma Meningioma Guillain-Barre Melkersson-Rosenthal 20 5
6 Requirements Case 3 Agent Required for Screening Testing HIV-1 and -2 All X X Hepatitis B All X X Hepatitis C All X X Syphilis All X X TSE All X WNV All X Sepsis All X Vaccinia (recent smallpox vaccination) All HTLV-I and II Viable, Leukocyte-Rich X X CMV Viable, Leukocyte-Rich X Chlamydia trachomatis Reproductive X X Neisseria gonorrhoeae Reproductive X X 21 CFR (r)/.75/.85 X Eye bank received a call from a transplant surgeon who had been informed by one of their patients of the recent death of another patient who contracted Mad Cow Disease from her corneal transplant". The surgeon provided them the recipient's name, but had no further information. Internet search revealed an obituary with the date of death of 5/28/12. Patient died at home with hospice care following hospitalization in the Neurological unit. 22 Case 3 Database revealed patient had a corneal transplant March Donor tissue came from a shared tissue and eye donor. Review of donor record reveals no relevant medical conditions. Travel history was reported but well within the time frames to not pose an elevated risk for CJD or vcjd. Case 3 - Questions How should the eye bank investigate this case? At what point in the investigation should the eye bank notify the FDA and EBAA?
7 Procedures Adverse Reaction You must establish and maintain procedures appropriate to meet core CGTP requirements for all steps that you perform in the manufacture of HCT/Ps. You must design these procedures to prevent circumstances that increase the risk of the introduction, transmission, or spread of communicable diseases through the use of HCT/Ps. A noxious and unintended response to any HCT/P for which there is a reasonable possibility that the HCT/P caused the response 21 CFR (a) CFR (y) 26 Quality Program: Information Sharing Contact Information Guidance for Industry: CGTP and Additional Requirements for Manufacturers of HCT/Ps (December 2011) Section V. E: With Whom Must an Establishment Share Information Pertaining to the Possible Contamination of or Potential for Transmission of Communicable Disease by an HCT/P? CBER or Consumer Affairs Branch (CAB) ocod@fda.hhs.gov Manufacturers Assistance and Technical Training Branch (MATTB) industry.biologics@fda.gov Follow us on Twitter
TRANSMISSIBLE SPONGIFORM ENCEPHALOPATHIES ADVISORY COMMITTEE MEETING October2010. Issue Summary
 TRANSMISSIBLE SPONGIFORM ENCEPHALOPATHIES ADVISORY COMMITTEE MEETING 28-29October2010 Issue Summary Informational Topic: FDA s Geographic Donor Deferral Policy to Reduce the Possible Risk of Transmission
TRANSMISSIBLE SPONGIFORM ENCEPHALOPATHIES ADVISORY COMMITTEE MEETING 28-29October2010 Issue Summary Informational Topic: FDA s Geographic Donor Deferral Policy to Reduce the Possible Risk of Transmission
TRANSFUSION ASSOCIATED DISEASE, RECALL, OR COMPLICATION INVESTIGATION POLICY I. FATALITIES AND COMPLICATIONS ASSOCIATED WITH TRANSFUSION:
 I. FATALITIES AND COMPLICATIONS ASSOCIATED WITH TRANSFUSION: A. TRANSFUSION RELATED FATALITY: FDA and MEDIC must be notified immediately, and subsequently in writing, when a possible transfusion related
I. FATALITIES AND COMPLICATIONS ASSOCIATED WITH TRANSFUSION: A. TRANSFUSION RELATED FATALITY: FDA and MEDIC must be notified immediately, and subsequently in writing, when a possible transfusion related
A. Anti-HIV-1/2, HIV NAT Lookback: for those identified blood and blood components collected:
 PURPOSE: MATERIALS: To outline a procedure for removing, recalling and/or correcting blood products that do not meet the requirements of the FDA, AABB, or the blood center. Recall/Market Withdrawal Form
PURPOSE: MATERIALS: To outline a procedure for removing, recalling and/or correcting blood products that do not meet the requirements of the FDA, AABB, or the blood center. Recall/Market Withdrawal Form
Guidance for Industry
 Guidance for Industry Advisement for the Use of Gen-Probe Procleix Ultrio Plus Assay for NAT Testing on Cadaveric Donors Release Date: 08/03/14 American Medical Education and Research Association 4757
Guidance for Industry Advisement for the Use of Gen-Probe Procleix Ultrio Plus Assay for NAT Testing on Cadaveric Donors Release Date: 08/03/14 American Medical Education and Research Association 4757
Directed Semen Donor Eligibility Requirements
 Directed Semen Donor Eligibility Requirements Directed (known) reproductive donors using the University Andrology Sperm Banking Program must follow FDA requirements to prevent the transmission of relevant
Directed Semen Donor Eligibility Requirements Directed (known) reproductive donors using the University Andrology Sperm Banking Program must follow FDA requirements to prevent the transmission of relevant
RECALL / EVENT INVESTIGATION & PATIENT LOOK BACK
 RECALL / EVENT INVESTIGATION & PATIENT LOOK BACK Federal, State and AABB requirements for nonconforming product investigation and recipient look back for adverse impact Stacy Ralston MPH, CLS (ASCP), RAC
RECALL / EVENT INVESTIGATION & PATIENT LOOK BACK Federal, State and AABB requirements for nonconforming product investigation and recipient look back for adverse impact Stacy Ralston MPH, CLS (ASCP), RAC
Inspections, Compliance, Enforcement, and Criminal Investigations. Central Texas Regional Blood & Tissue Center 07-Nov-03
 1 of 7 6/10/2009 2:20 PM Inspections, Compliance, Enforcement, and Criminal Investigations Department of Health and Human Services Public Health Service Food and Drug Administration Dallas District 4040
1 of 7 6/10/2009 2:20 PM Inspections, Compliance, Enforcement, and Criminal Investigations Department of Health and Human Services Public Health Service Food and Drug Administration Dallas District 4040
Non-reproductive tissues and cells Recommending authority/ association
 Colour key Minimum requirements as set out in Directive 2004/23/EC More stringent - legy binding More stringent - recommended Not legy binding and not recommended Tested pathogen Donor test/ technique
Colour key Minimum requirements as set out in Directive 2004/23/EC More stringent - legy binding More stringent - recommended Not legy binding and not recommended Tested pathogen Donor test/ technique
[Type here] Version Approval Dates Announcement Date Effective Standards Committee: Jan 2019
![[Type here] Version Approval Dates Announcement Date Effective Standards Committee: Jan 2019 [Type here] Version Approval Dates Announcement Date Effective Standards Committee: Jan 2019](/thumbs/95/123651537.jpg) SECTION D AUTHORIZATION, INFORMED CONSENT, DONOR SCREENING, AND TISSUE RECOVERY, COLLECTION, AND ACQUISITION D1.000 GENERAL POLICIES In addition to the requirements at the series of standards at B1.500,
SECTION D AUTHORIZATION, INFORMED CONSENT, DONOR SCREENING, AND TISSUE RECOVERY, COLLECTION, AND ACQUISITION D1.000 GENERAL POLICIES In addition to the requirements at the series of standards at B1.500,
Question: 1. Are you currently taking an antibiotic?
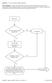 Question: 1. Are you currently taking an antibiotic? Donor Eligibility: A donor with an infection that could cause bacteremia/sepsis or other relevant communicable disease should not donate. The reason
Question: 1. Are you currently taking an antibiotic? Donor Eligibility: A donor with an infection that could cause bacteremia/sepsis or other relevant communicable disease should not donate. The reason
BLOOD COMPONENTS are regulated as
 Managing Recalls and Withdrawals of Blood Components Glenn Ramsey Donor centers are issuing a growing number of recalls and market withdrawals to hospital transfusion services about blood components. More
Managing Recalls and Withdrawals of Blood Components Glenn Ramsey Donor centers are issuing a growing number of recalls and market withdrawals to hospital transfusion services about blood components. More
Non-reproductive tissues and cells
 Colour key Tested pathogen VIRAL Minimum requirements as set out in Directive 2004/23/EC More stringent testing - legy binding on national level More stringent testing - recommended on national level Not
Colour key Tested pathogen VIRAL Minimum requirements as set out in Directive 2004/23/EC More stringent testing - legy binding on national level More stringent testing - recommended on national level Not
2009 Eye Banking Statistical Report Eye Bank Association of America th Street, N.W. Suite 1010 Washington, DC Phone (202) Fax
 2009 Eye Banking Statistical Report Eye Bank Association of America 1015 18th Street, N.W. Suite 1010 Washington, DC 20036 Phone (202) 775-4999 Fax (202) 429-6036 www.restoresight.org Introduction 2009
2009 Eye Banking Statistical Report Eye Bank Association of America 1015 18th Street, N.W. Suite 1010 Washington, DC 20036 Phone (202) 775-4999 Fax (202) 429-6036 www.restoresight.org Introduction 2009
Question: 1. Are you currently taking an antibiotic?
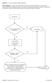 Question: 1. Are you currently taking an antibiotic? Donor Eligibility: A donor with an infection that could cause bacteremia/sepsis or other relevant communicable disease should not donate. The reason
Question: 1. Are you currently taking an antibiotic? Donor Eligibility: A donor with an infection that could cause bacteremia/sepsis or other relevant communicable disease should not donate. The reason
Guidance for Industry
 Guidance for Industry Use of Nucleic Acid Tests on Pooled and Individual Samples from Donors of Whole Blood and Blood Components (including Source Plasma and Source Leukocytes) to Adequately and Appropriately
Guidance for Industry Use of Nucleic Acid Tests on Pooled and Individual Samples from Donors of Whole Blood and Blood Components (including Source Plasma and Source Leukocytes) to Adequately and Appropriately
Non-reproductive tissues and cells Recommending authority/ association
 Colour key Minimum requirements as set out in Directive 2004/23/EC More stringent - legally binding More stringent - recommended Not legally binding and not recommended Non-reproductive tissues and cells
Colour key Minimum requirements as set out in Directive 2004/23/EC More stringent - legally binding More stringent - recommended Not legally binding and not recommended Non-reproductive tissues and cells
Non-reproductive tissues and cells
 Colour key Minimum requirements as set out in Directive 2004/23/EC More stringent testing - legally binding on national level More stringent testing - recommended on national level Not legally binding
Colour key Minimum requirements as set out in Directive 2004/23/EC More stringent testing - legally binding on national level More stringent testing - recommended on national level Not legally binding
Non-reproductive tissues and cells
 Ministry of Health: Institute for Transplantation and Biomedicine / Colour key VIRAL HIV 1 and HIV 2 Hepatitis B Minimum requirements as set out in Directive 2004/23/EC More stringent - legy binding More
Ministry of Health: Institute for Transplantation and Biomedicine / Colour key VIRAL HIV 1 and HIV 2 Hepatitis B Minimum requirements as set out in Directive 2004/23/EC More stringent - legy binding More
2014/LSIF/PD/025 Malaysia s Approach to Testing Strategies
 2014/LSIF/PD/025 Malaysia s Approach to Testing Strategies Submitted by: Malaysia Policy Dialogue and Workshop on Attaining a Safe and Sustainable Blood Supply Chain Manila, Philippines 30 September 1
2014/LSIF/PD/025 Malaysia s Approach to Testing Strategies Submitted by: Malaysia Policy Dialogue and Workshop on Attaining a Safe and Sustainable Blood Supply Chain Manila, Philippines 30 September 1
Non-reproductive tissues and cells
 Colour key Minimum requirements as set out in Directive 2004/23/EC More stringent testing - legally binding on national level More stringent testing - recommended on national level Not legally binding
Colour key Minimum requirements as set out in Directive 2004/23/EC More stringent testing - legally binding on national level More stringent testing - recommended on national level Not legally binding
Non-reproductive tissues and cells
 Colour key Minimum requirements as set out in Directive 2004/23/EC More stringent - legy binding on national level More stringent - recommended on national level Not legy binding and not recommended on
Colour key Minimum requirements as set out in Directive 2004/23/EC More stringent - legy binding on national level More stringent - recommended on national level Not legy binding and not recommended on
CJD (Creutzfeldt-Jakob disease)
 CJD - lyodura and the risk of exposure during healthcare - frequently asked questions We would like to reassure all our patients that tight regulations govern all infection control processes at Addenbrooke's,
CJD - lyodura and the risk of exposure during healthcare - frequently asked questions We would like to reassure all our patients that tight regulations govern all infection control processes at Addenbrooke's,
Non-reproductive tissues and cells Recommending authority/ association
 Colour key Minimum requirements as set out in Directive 2004/23/EC More stringent testing - legy binding on national level More stringent testing - recomd on national level Not legy binding and not recomd
Colour key Minimum requirements as set out in Directive 2004/23/EC More stringent testing - legy binding on national level More stringent testing - recomd on national level Not legy binding and not recomd
Guidance for Industry
 Guidance for Industry Informed Consent Recommendations for Source Plasma Donors Participating in Plasmapheresis and Immunization Programs Additional copies of this guidance are available from the Office
Guidance for Industry Informed Consent Recommendations for Source Plasma Donors Participating in Plasmapheresis and Immunization Programs Additional copies of this guidance are available from the Office
Guidance for Industry
 Guidance for Industry Adequate and Appropriate Donor Screening Tests for Hepatitis B; Hepatitis B Surface Antigen (HBsAg) Assays Used to Test Donors of Whole Blood and Blood Components, Including Source
Guidance for Industry Adequate and Appropriate Donor Screening Tests for Hepatitis B; Hepatitis B Surface Antigen (HBsAg) Assays Used to Test Donors of Whole Blood and Blood Components, Including Source
Guidance for Industry
 Guidance for Industry Lookback for Hepatitis C Virus (HCV): Product Quarantine, Consignee Notification, Further Testing, Product Disposition, and Notification of Transfusion Recipients Based on Donor Test
Guidance for Industry Lookback for Hepatitis C Virus (HCV): Product Quarantine, Consignee Notification, Further Testing, Product Disposition, and Notification of Transfusion Recipients Based on Donor Test
Process or performance improvement is often discussed
 Process Improvement in Transfusion Medicine Ongoing efforts to decrease costs in the clinical laboratory make continuous process improvement especially important in difficult economic times. Process improvement
Process Improvement in Transfusion Medicine Ongoing efforts to decrease costs in the clinical laboratory make continuous process improvement especially important in difficult economic times. Process improvement
Organ and Tissue Donation Emergency Department Orientation. Calgary Health Trust May, 2015
 Organ and Tissue Donation Emergency Department Orientation Calgary Health Trust May, 2015 Fact Vs. Fiction 2 Southern Alberta Organ & Tissue Donation Program Southern Alberta Organ Donation Program HOPE
Organ and Tissue Donation Emergency Department Orientation Calgary Health Trust May, 2015 Fact Vs. Fiction 2 Southern Alberta Organ & Tissue Donation Program Southern Alberta Organ Donation Program HOPE
Department of Health and Human Services
 Friday, August 24, 2007 Part III Department of Health and Human Services Food and Drug Administration 2 CFR Parts 606 and 60 Current Good Manufacturing Practice for Blood and Blood Components; Notification
Friday, August 24, 2007 Part III Department of Health and Human Services Food and Drug Administration 2 CFR Parts 606 and 60 Current Good Manufacturing Practice for Blood and Blood Components; Notification
Report on the Transplantation Transmission Sentinel Network (TTSN)
 Report on the Transplantation Transmission Sentinel Network (TTSN) CATB Session - AATB s 31st Annual Meeting, Boston September 15, 2007 Scott Brubaker, CTBS AATB Representative, TTSN Advisory Group TTSN
Report on the Transplantation Transmission Sentinel Network (TTSN) CATB Session - AATB s 31st Annual Meeting, Boston September 15, 2007 Scott Brubaker, CTBS AATB Representative, TTSN Advisory Group TTSN
MEDICAL ADVISORY. July 18, 2017
 MEDICAL ADVISORY July 18, 2017 The following changes were made at the June 16, 2017 meeting of the Medical Advisory Board, and will become effective immediately. B1.100 Eye Bank Inspection The Accreditation
MEDICAL ADVISORY July 18, 2017 The following changes were made at the June 16, 2017 meeting of the Medical Advisory Board, and will become effective immediately. B1.100 Eye Bank Inspection The Accreditation
CERTIFIED MAIL RETURN RECEIPT REQUESTED. Esther Hernandez President United States Blood Bank, Inc NW 95th Avenue Miami, Florida
 DEPARTMENT OF HEALTH AND HUMAN SERVICES Food and Drug Administration Silver Spring MD 20993 CERTIFIED MAIL RETURN RECEIPT REQUESTED Esther Hernandez President United States Blood Bank, Inc. 2400 NW 95th
DEPARTMENT OF HEALTH AND HUMAN SERVICES Food and Drug Administration Silver Spring MD 20993 CERTIFIED MAIL RETURN RECEIPT REQUESTED Esther Hernandez President United States Blood Bank, Inc. 2400 NW 95th
User Instructions Allogeneic HPC, Apheresis and HPC, Marrow Donor History Questionnaire. Table of Contents
 User Instructions Allogeneic HPC, Apheresis and HPC, Marrow Donor History Questionnaire Table of Contents Purpose Introduction Capture Questions Methods of Administration DHQ Materials - Structure and
User Instructions Allogeneic HPC, Apheresis and HPC, Marrow Donor History Questionnaire Table of Contents Purpose Introduction Capture Questions Methods of Administration DHQ Materials - Structure and
Guidance for Industry
 Guidance for Industry Revised Recommendations for Donor and Product Management Based on Screening Tests for Syphilis DRAFT GUIDANCE This document is being distributed for comment purposes only. Submit
Guidance for Industry Revised Recommendations for Donor and Product Management Based on Screening Tests for Syphilis DRAFT GUIDANCE This document is being distributed for comment purposes only. Submit
Hot Topics in Tissue Safety
 Hot Topics in Tissue Safety 37 th Annual AATB Meeting National Harbor, Maryland 03 October 2013 Scott A. Brubaker, CTBS Chief Policy Officer PHS Guideline for Reducing Human Immunodeficiency Virus, Hepatitis
Hot Topics in Tissue Safety 37 th Annual AATB Meeting National Harbor, Maryland 03 October 2013 Scott A. Brubaker, CTBS Chief Policy Officer PHS Guideline for Reducing Human Immunodeficiency Virus, Hepatitis
USER INSTRUCTIONS HPC, Cord Blood Donor History Questionnaire. Table of Contents
 Page 1 of 9 USER INSTRUCTIONS HPC, Cord Blood Donor History Questionnaire Table of Contents Purpose Introduction Capture Questions Methods of Administration DHQ Materials Structure and Content Reformatting
Page 1 of 9 USER INSTRUCTIONS HPC, Cord Blood Donor History Questionnaire Table of Contents Purpose Introduction Capture Questions Methods of Administration DHQ Materials Structure and Content Reformatting
Human tissues and cells annual report 2011
 Human tissues and cells annual report 2011 2012 Human tissues and cells Annual report Danish Health and Medicines Authority, 2012. This publication can be freely cited with a clear indication of source.
Human tissues and cells annual report 2011 2012 Human tissues and cells Annual report Danish Health and Medicines Authority, 2012. This publication can be freely cited with a clear indication of source.
Non-reproductive tissues and cells
 Colour key Minimum requirements as set out in Directive 2004/23/EC and its technical Directives (particularly 2006/17/EC) More stringent -legally binding, applies for all donations and all donor profiles
Colour key Minimum requirements as set out in Directive 2004/23/EC and its technical Directives (particularly 2006/17/EC) More stringent -legally binding, applies for all donations and all donor profiles
Reporting from Council of Europe member states on the collection, testing and use of blood and blood components in Europe The 2006 Survey
 Reporting from Council of Europe member states on the collection, testing and use of blood and blood components in Europe The 2006 Survey This questionnaire consists of three sections: A. Collection and
Reporting from Council of Europe member states on the collection, testing and use of blood and blood components in Europe The 2006 Survey This questionnaire consists of three sections: A. Collection and
Current Infectious Disease Screening for the Live Organ Donor
 2013 Public Health and Safety Guidelines for reducing HIV, HBV and HCV Transmission Though Organ Transplantation: Implications for Counseling and Disclosure Dianne LaPointe Rudow DNP, ANP-BC, CCTC Director
2013 Public Health and Safety Guidelines for reducing HIV, HBV and HCV Transmission Though Organ Transplantation: Implications for Counseling and Disclosure Dianne LaPointe Rudow DNP, ANP-BC, CCTC Director
LIVE INTERACTIVE YOUR DESKTOP. Food Recall Process. Cecilia M. Wolyniak Food and Drug Administration Office of Enforcement
 LIVE INTERACTIVE LEARNING @ YOUR DESKTOP Food Recall Process Cecilia M. Wolyniak Food and Drug Administration Office of Enforcement Wednesday, December 9, 2009 LEGAL ISSUES Code of Federal Regulations
LIVE INTERACTIVE LEARNING @ YOUR DESKTOP Food Recall Process Cecilia M. Wolyniak Food and Drug Administration Office of Enforcement Wednesday, December 9, 2009 LEGAL ISSUES Code of Federal Regulations
Non-reproductive tissues and cells Recommending authority/ association
 Colour key Minimum requirements as set out in Directive 2004/23/EC More stringent - legy binding More stringent - recommended Not legy binding and not recommended Non-reproductive tissues and cells VIRAL
Colour key Minimum requirements as set out in Directive 2004/23/EC More stringent - legy binding More stringent - recommended Not legy binding and not recommended Non-reproductive tissues and cells VIRAL
DEPARTMENT OF HEALTH AND SENIOR SERVICES PO BOX 360 TRENTON, N.J
 JON S. CORZINE Governor DEPARTMENT OF HEALTH AND SENIOR SERVICES PO BOX 360 TRENTON, N.J. 08625-0360 www.nj.gov/health HEATHER HOWARD Commissioner TO: FROM: Public Health Council Heather Howard Commissioner
JON S. CORZINE Governor DEPARTMENT OF HEALTH AND SENIOR SERVICES PO BOX 360 TRENTON, N.J. 08625-0360 www.nj.gov/health HEATHER HOWARD Commissioner TO: FROM: Public Health Council Heather Howard Commissioner
Outbreak Investigation Guidance for Vectorborne Diseases
 COMMUNICABLE DISEASE OUTBREAK MANUAL New Jersey s Public Health Response APPENDIX T3: EXTENDED GUIDANCE Outbreak Investigation Guidance for Vectorborne Diseases As per N.J.A.C. 8:57, viruses that are transmitted
COMMUNICABLE DISEASE OUTBREAK MANUAL New Jersey s Public Health Response APPENDIX T3: EXTENDED GUIDANCE Outbreak Investigation Guidance for Vectorborne Diseases As per N.J.A.C. 8:57, viruses that are transmitted
The New Regulations - Special IVD Issues
 The New Regulations - Special IVD Issues Dirk Stynen, Ph. D. President - Principal Consultant Qarad Geel, Belgium RMD Brussels October 2018 The IVD Regulation 2017/746 October 29, 2018 www.qarad.com 2
The New Regulations - Special IVD Issues Dirk Stynen, Ph. D. President - Principal Consultant Qarad Geel, Belgium RMD Brussels October 2018 The IVD Regulation 2017/746 October 29, 2018 www.qarad.com 2
The TTSN - A Collaborative Biovigilance System
 The TTSN - A Collaborative Biovigilance System 5th World Congress on Tissue Banking 12th International Conference of the APASTB Kuala Lumpur, Malaysia June 4, 2008 Scott A. Brubaker, CTBS American Association
The TTSN - A Collaborative Biovigilance System 5th World Congress on Tissue Banking 12th International Conference of the APASTB Kuala Lumpur, Malaysia June 4, 2008 Scott A. Brubaker, CTBS American Association
The DRAI (Donors >12 yrs old) versus Requirements
 Q# or Pg # Pg 1 Question or section as it appears on the DRAI questionnaire (Donors >12 yrs old) 5-7-12 Donor Name, Person Interviewed (name, relationship), Contact Information (phone, address, city, state,
Q# or Pg # Pg 1 Question or section as it appears on the DRAI questionnaire (Donors >12 yrs old) 5-7-12 Donor Name, Person Interviewed (name, relationship), Contact Information (phone, address, city, state,
QSEAL Recovered Plasma Specification
 QSEAL Recovered Plasma Specification Background The is part of a series of standards that comprise the Plasma Protein Therapeutics Association (PPTA) QSEAL Standards Program. PPTA's Voluntary Standards
QSEAL Recovered Plasma Specification Background The is part of a series of standards that comprise the Plasma Protein Therapeutics Association (PPTA) QSEAL Standards Program. PPTA's Voluntary Standards
Blood Donor Counselling
 Blood Donor Counselling A presentation for the Nepal Red Cross Society Blood Transfusion Service Dr Che Kit Lin On behalf of GAP and Hong Kong Red Cross 26 th August 2014 Purpose and Outcomes of Workshop
Blood Donor Counselling A presentation for the Nepal Red Cross Society Blood Transfusion Service Dr Che Kit Lin On behalf of GAP and Hong Kong Red Cross 26 th August 2014 Purpose and Outcomes of Workshop
User Instructions Allogeneic HPC, Apheresis and HPC, Marrow Donor History Questionnaire. Table of Contents
 User Instructions Allogeneic HPC, Apheresis and HPC, Marrow Donor History Questionnaire Table of Contents Purpose Introduction Methods of Administration DHQ Materials - Structure and Content Reformatting
User Instructions Allogeneic HPC, Apheresis and HPC, Marrow Donor History Questionnaire Table of Contents Purpose Introduction Methods of Administration DHQ Materials - Structure and Content Reformatting
2006 Guidelines for gamete and embryo donation
 2006 Guidelines for gamete and embryo donation The Practice Committee of the American Society for Reproductive Medicine and the Practice Committee of the Society for Assisted Reproductive Technology Birmingham,
2006 Guidelines for gamete and embryo donation The Practice Committee of the American Society for Reproductive Medicine and the Practice Committee of the Society for Assisted Reproductive Technology Birmingham,
[Submitted Electronically]
![[Submitted Electronically] [Submitted Electronically]](/thumbs/82/85246007.jpg) [Submitted Electronically] Dr. Matthew J. Kuehnert, Director Office of Blood, Organ, and Other Tissue Safety Division of Healthcare Quality Promotion National Center for Emerging and Zoonotic Infectious
[Submitted Electronically] Dr. Matthew J. Kuehnert, Director Office of Blood, Organ, and Other Tissue Safety Division of Healthcare Quality Promotion National Center for Emerging and Zoonotic Infectious
Relevant Communicable Diseases in HCT/Ps
 Relevant Communicable Diseases in HCT/Ps Phyllis I. Warkentin, MD Professor of Pathology and Pediatrics University of Nebraska Medical Center FACT Chief Medical Officer Relevant Communicable Diseases in
Relevant Communicable Diseases in HCT/Ps Phyllis I. Warkentin, MD Professor of Pathology and Pediatrics University of Nebraska Medical Center FACT Chief Medical Officer Relevant Communicable Diseases in
Guidance Document for Source Establishments Reporting Adverse Reactions to Human Cells, Tissues and Organs
 Guidance Document for Source Establishments Reporting Adverse Reactions to Human Cells, Tissues and Organs (Effective Date: 2010-05-25) Canada Vigilance Adverse Reaction Monitoring Program and Database,
Guidance Document for Source Establishments Reporting Adverse Reactions to Human Cells, Tissues and Organs (Effective Date: 2010-05-25) Canada Vigilance Adverse Reaction Monitoring Program and Database,
Organ Procurement and Transplantation Network
 OPTN Organ Procurement and Transplantation Network POLICIES This document provides the policy language approved by the OPTN/UNOS Board at its meeting in June 2015 as part of the Operations and Safety Committee
OPTN Organ Procurement and Transplantation Network POLICIES This document provides the policy language approved by the OPTN/UNOS Board at its meeting in June 2015 as part of the Operations and Safety Committee
Maximizing Cornea and Tissue Donation through Specimen Quality
 Maximizing Cornea and Tissue Donation through Specimen Quality Robert W. Bresler, Sydney D. Gastreich, Elias G. Koulouriotis, Linda S. Martin, Susan Diane Brockmeier, Chak-Sum Ho, PhD Abstract Purpose:
Maximizing Cornea and Tissue Donation through Specimen Quality Robert W. Bresler, Sydney D. Gastreich, Elias G. Koulouriotis, Linda S. Martin, Susan Diane Brockmeier, Chak-Sum Ho, PhD Abstract Purpose:
Standards for OPOs (In addition to Section 59A-1.005)
 Standards for OPOs (In addition to Section 59A-1.005) (20) Each OPO shall comply with 42 CFR Part 485, 1994, and make the records relating to the federal standards available upon request to surveyors for
Standards for OPOs (In addition to Section 59A-1.005) (20) Each OPO shall comply with 42 CFR Part 485, 1994, and make the records relating to the federal standards available upon request to surveyors for
Structure and duties of Blood service
 NATIONAL CENTER FOR TRANSFUSION MEDICINE Prevalence of infectious diseases among Mongolian blood donors Namjil Erdenebayar General director of The National Center for Transfusion Medicine, Mongolia NCTM,
NATIONAL CENTER FOR TRANSFUSION MEDICINE Prevalence of infectious diseases among Mongolian blood donors Namjil Erdenebayar General director of The National Center for Transfusion Medicine, Mongolia NCTM,
Department of Health and Human Services
 Tuesday, May 25, 2004 Part II Department of Health and Human Services Food and Drug Administration 21 CFR Parts 210, 211, 820, and 1271 Eligibility Determination for Donors of Human Cells, Tissues, and
Tuesday, May 25, 2004 Part II Department of Health and Human Services Food and Drug Administration 21 CFR Parts 210, 211, 820, and 1271 Eligibility Determination for Donors of Human Cells, Tissues, and
Uniform DRAI - Donor >12 yrs old Requirements Crosswalk
 Q# or Pg # Pg 1 Question or section as it appears on the Uniform DRAI - Donor >12 yrs old 9-10-14 Donor Name, Person Interviewed (name, relationship), Contact Information (phone, address, city, state,
Q# or Pg # Pg 1 Question or section as it appears on the Uniform DRAI - Donor >12 yrs old 9-10-14 Donor Name, Person Interviewed (name, relationship), Contact Information (phone, address, city, state,
15. Procuring, processing and transporting gametes and
 15. Procuring, processing and transporting gametes and embryos Version 6.0 On this page: : Extracts from the HFE Act Directions HFEA guidance: Documented procedures: general Patient selection and procurement
15. Procuring, processing and transporting gametes and embryos Version 6.0 On this page: : Extracts from the HFE Act Directions HFEA guidance: Documented procedures: general Patient selection and procurement
17. Storage of gametes and embryos
 17. Storage of gametes and embryos This guidance note contains: Mandatory requirements Extracts from the HFE Act 1990 (as amended) Extracts from licence conditions Reference to relevant HFEA Directions
17. Storage of gametes and embryos This guidance note contains: Mandatory requirements Extracts from the HFE Act 1990 (as amended) Extracts from licence conditions Reference to relevant HFEA Directions
Confirmatory Testing Algorithm. Routine Donor Testing Algorithm
 Confirmatory Testing Algorithm Blood Confirmatory ABO/Rh Beckman Coulter PK-ABO/Rh Immucor NEO ABO/Rh Immucor NEO ABO/Rh Immucor Manual ABO/Rh Typing Immucor Manual ABO/Rh Typing Discrepancy Resolution
Confirmatory Testing Algorithm Blood Confirmatory ABO/Rh Beckman Coulter PK-ABO/Rh Immucor NEO ABO/Rh Immucor NEO ABO/Rh Immucor Manual ABO/Rh Typing Immucor Manual ABO/Rh Typing Discrepancy Resolution
Public Health, Infections and Transplantation
 Public Health, Infections and Transplantation Dr Kerry Chant Chief Health Officer & Deputy Director General Population and Public Health NSW Ministry of Health May2014 Public Health Infection and Transplantation
Public Health, Infections and Transplantation Dr Kerry Chant Chief Health Officer & Deputy Director General Population and Public Health NSW Ministry of Health May2014 Public Health Infection and Transplantation
Student Health Requirements Master of Arts, Biomedical Sciences Program
 Student Health Requirements Master of Arts, Biomedical Sciences Program Hepatitis B Policy All Biomedical sciences program students, just as physicians in practice, are required to be current with required
Student Health Requirements Master of Arts, Biomedical Sciences Program Hepatitis B Policy All Biomedical sciences program students, just as physicians in practice, are required to be current with required
AFDO Conference June 9, Mercedes Mota, Director Medical Device, Surgical Care Johnson & Johnson Quality & Compliance Worldwide
 AFDO Conference June 9, 2009 Mercedes Mota, Director Medical Device, Surgical Care Johnson & Johnson Quality & Compliance Worldwide Government Agencies 483 observations A violation of the FD&C Act involving
AFDO Conference June 9, 2009 Mercedes Mota, Director Medical Device, Surgical Care Johnson & Johnson Quality & Compliance Worldwide Government Agencies 483 observations A violation of the FD&C Act involving
Within 14 days at 2-8⁰C. DO NOT FREEZE WHOLE BLOOD Micro Typing Systems 50 µl plasma
 Test Name *Minimum Collection Tubes Sample Stability ABO/Rh Blood Typing 50 µl cells EDTA whole blood (lavender-top) Within 14 days at 2-8⁰C. DO NOT FREEZE WHOLE BLOOD Micro Typing Systems 50 µl plasma
Test Name *Minimum Collection Tubes Sample Stability ABO/Rh Blood Typing 50 µl cells EDTA whole blood (lavender-top) Within 14 days at 2-8⁰C. DO NOT FREEZE WHOLE BLOOD Micro Typing Systems 50 µl plasma
Guide to the preparation, use and quality assurance of blood components
 Contents Foreword...3 Members of the European Committee (Partial Agreement) on Blood Transfusion... 8 Members of the GTS working group... 22 Members of the TS066 working group... 30 Recommendation No.
Contents Foreword...3 Members of the European Committee (Partial Agreement) on Blood Transfusion... 8 Members of the GTS working group... 22 Members of the TS066 working group... 30 Recommendation No.
Database on Blood Safety 2009 in ECO Member States
 Database on Blood Safety 2009 in ECO Member States Section 1: Administrative Information Information provided by: 1.1 Name: 1.2 Title: 1.3 Position: 1.4 Organization: 1.5 Address: 1.6 Country: 1.7 Tel.
Database on Blood Safety 2009 in ECO Member States Section 1: Administrative Information Information provided by: 1.1 Name: 1.2 Title: 1.3 Position: 1.4 Organization: 1.5 Address: 1.6 Country: 1.7 Tel.
Guidance Document for Investigating and Reporting Adverse Reactions to the EBAA. Version 3
 Guidance Document for Investigating and Reporting Adverse Reactions to the EBAA Version 3 July 2017 Contents INTRODUCTION... 2 GENERAL GUIDANCE FOR INVESTIGATING ADVERSE REACTIONS... 3 Imputability Level
Guidance Document for Investigating and Reporting Adverse Reactions to the EBAA Version 3 July 2017 Contents INTRODUCTION... 2 GENERAL GUIDANCE FOR INVESTIGATING ADVERSE REACTIONS... 3 Imputability Level
Objectives 9/23/2014. Why is Organ Donation Important? Conflict of interest-none
 Objectives Understand the role of the UW OTD Discuss the need for organ donation From referral to recovery: An overview of the phases of the organ donation process Discuss the importance of the provider
Objectives Understand the role of the UW OTD Discuss the need for organ donation From referral to recovery: An overview of the phases of the organ donation process Discuss the importance of the provider
2.0 MINIMUM PROCUREMENT STANDARDS FOR AN ORGAN PROCUREMENT ORGANIZATION (OPO)
 2.0 MINIMUM PROCUREMENT STANDARDS FOR AN ORGAN PROCUREMENT ORGANIZATION (OPO) In order to maximize the gift of donation and optimize recipient outcomes and safety, the Organ Procurement Organization (OPO)
2.0 MINIMUM PROCUREMENT STANDARDS FOR AN ORGAN PROCUREMENT ORGANIZATION (OPO) In order to maximize the gift of donation and optimize recipient outcomes and safety, the Organ Procurement Organization (OPO)
Appendix A: Disease-Specific Chapters
 Ministry of Health and Long-Term Care Infectious Diseases Protocol Appendix A: Disease-Specific Chapters Chapter: Creutzfeldt-Jakob Disease, all types Revised March 2017 Creutzfeldt-Jakob Disease, all
Ministry of Health and Long-Term Care Infectious Diseases Protocol Appendix A: Disease-Specific Chapters Chapter: Creutzfeldt-Jakob Disease, all types Revised March 2017 Creutzfeldt-Jakob Disease, all
Objectives. Nebraska Organ Recovery 2/16/2015. The Role of Hospice and Palliative Care in Organ and Tissue Donation
 The Role of Hospice and Palliative Care in Organ and Tissue Donation Objectives Summarize the organ and tissue donation process. Identify the role of hospice and palliative care staff in the organ and
The Role of Hospice and Palliative Care in Organ and Tissue Donation Objectives Summarize the organ and tissue donation process. Identify the role of hospice and palliative care staff in the organ and
June 8, Division of Dockets Management (HFA 305) Food and Drug Administration 5630 Fishers Lane, Rm Rockville, MD 20852
 June 8, 2018 Division of Dockets Management (HFA 305) Food and Drug Administration 5630 Fishers Lane, Rm. 1061 Rockville, MD 20852 Submitted via http://www.regulations.gov Re: Docket No. FDA 2016 D 0545,
June 8, 2018 Division of Dockets Management (HFA 305) Food and Drug Administration 5630 Fishers Lane, Rm. 1061 Rockville, MD 20852 Submitted via http://www.regulations.gov Re: Docket No. FDA 2016 D 0545,
CORD BLOOD TRANSPLANTATION STUDY CBU EXCLUSION AND QUARANTINE RELEASE FORM
 (Any answer of Yes indicates exclusion) 1. Informed Consent and Maternal/Infant Demographics a. Lack of confirmation of signed and witnessed informed consent 1 Yes 2 No Is the following information missing
(Any answer of Yes indicates exclusion) 1. Informed Consent and Maternal/Infant Demographics a. Lack of confirmation of signed and witnessed informed consent 1 Yes 2 No Is the following information missing
Compliance Program Guidance Manual. Chapter 42 - Blood and Blood Products
 Compliance Program Guidance Manual Chapter 42 - Blood and Blood Products Inspection of Licensed and Unlicensed Blood Banks, Brokers, Reference Laboratories, and Contractors- 7342.001 Implementation Date:
Compliance Program Guidance Manual Chapter 42 - Blood and Blood Products Inspection of Licensed and Unlicensed Blood Banks, Brokers, Reference Laboratories, and Contractors- 7342.001 Implementation Date:
Guidance for Industry
 Reprinted from FDA s website by EAS Consulting Group, LLC Guidance for Industry Recommendations for Screening, Testing, and Management of Blood Donors and Blood and Blood Components Based on Screening
Reprinted from FDA s website by EAS Consulting Group, LLC Guidance for Industry Recommendations for Screening, Testing, and Management of Blood Donors and Blood and Blood Components Based on Screening
Understanding IVF Processes in Surrogacy
 Melvin H. Thornton II MD Medical Director CT Fertility Understanding IVF Processes in Surrogacy The Basics Surrogacy involves multiple parties IVF CLINIC Egg donors screening and matching* Medical process
Melvin H. Thornton II MD Medical Director CT Fertility Understanding IVF Processes in Surrogacy The Basics Surrogacy involves multiple parties IVF CLINIC Egg donors screening and matching* Medical process
Revisions to Labeling Requirements for Blood and Blood Components, Including Source Plasma
 This document is scheduled to be published in the Federal Register on 01/03/2012 and available online at http://federalregister.gov/a/2011-33554, and on FDsys.gov 4160-01-P DEPARTMENT OF HEALTH AND HUMAN
This document is scheduled to be published in the Federal Register on 01/03/2012 and available online at http://federalregister.gov/a/2011-33554, and on FDsys.gov 4160-01-P DEPARTMENT OF HEALTH AND HUMAN
Patient Name: MRN: DOB: Treatment Location:
 Page 1 of 5 I. TO (Required) This Section is required to be completed by all patients who undergo kidney transplant surgery. I hereby consent to and authorize Dr. and his/her assistant(s), including supervised
Page 1 of 5 I. TO (Required) This Section is required to be completed by all patients who undergo kidney transplant surgery. I hereby consent to and authorize Dr. and his/her assistant(s), including supervised
The testing of Donated Blood and Components at NHSBT
 The testing of Donated Blood and Components at NHSBT NHSBT is responsible for collecting all donated blood and platelets in England, then processing into components before issuing to client hospitals.
The testing of Donated Blood and Components at NHSBT NHSBT is responsible for collecting all donated blood and platelets in England, then processing into components before issuing to client hospitals.
Mahoning County Public Health. Epidemiology Response Annex
 Mahoning County Public Health Epidemiology Response Annex Created: May 2006 Updated: February 2015 Mahoning County Public Health Epidemiology Response Annex Table of Contents Epidemiology Response Document
Mahoning County Public Health Epidemiology Response Annex Created: May 2006 Updated: February 2015 Mahoning County Public Health Epidemiology Response Annex Table of Contents Epidemiology Response Document
FDA Reentry Guidance
 FDA Reentry Guidance T. cruzi - Chagas, Hepatitis C and HIV Wednesday 6/6/18 Doug Denyer O Dina Hurlburt, SBB(ASCP)CM Outline Impact to Clients Review of FDA Guidance Documents Reentry Request Process
FDA Reentry Guidance T. cruzi - Chagas, Hepatitis C and HIV Wednesday 6/6/18 Doug Denyer O Dina Hurlburt, SBB(ASCP)CM Outline Impact to Clients Review of FDA Guidance Documents Reentry Request Process
Coding for Preventive Services A Guide for HIV Providers
 Coding for Preventive Services A Guide for HIV Providers Jessie Murphy, MPH and Michelle Cataldo, LCSW, April 2016 Implementation of the Patient Protection and Affordable Care Act and other regulatory
Coding for Preventive Services A Guide for HIV Providers Jessie Murphy, MPH and Michelle Cataldo, LCSW, April 2016 Implementation of the Patient Protection and Affordable Care Act and other regulatory
READ BEFORE USING. Contents:
 READ BEFORE USING The Liveyon ReGen product is derived from voluntarily donated human umbilical cord. ReGen can only be sold to, and used by licensed physicians. Read this entire package insert carefully
READ BEFORE USING The Liveyon ReGen product is derived from voluntarily donated human umbilical cord. ReGen can only be sold to, and used by licensed physicians. Read this entire package insert carefully
Impact of Testing Strategies to Reduce Transmission Risk for HBV. Ravi Reddy, M Vermeulen South African National Blood Service (SANBS) 29 July 2013
 Impact of Testing Strategies to Reduce Transmission Risk for HBV Ravi Reddy, M Vermeulen South African National Blood Service (SANBS) 29 July 2013 Overview of SANBS SANBS is a private not for profit company
Impact of Testing Strategies to Reduce Transmission Risk for HBV Ravi Reddy, M Vermeulen South African National Blood Service (SANBS) 29 July 2013 Overview of SANBS SANBS is a private not for profit company
The 2008 Guidelines for Gamete and Embryo Donation
 2008 Guidelines for gamete and embryo donation: a Practice Committee report Practice Committee of the American Society for Reproductive Medicine, and the Practice Committee of the Society for Assisted
2008 Guidelines for gamete and embryo donation: a Practice Committee report Practice Committee of the American Society for Reproductive Medicine, and the Practice Committee of the Society for Assisted
patients with blood borne viruses Controlled Document Number: Version Number: 4 Controlled Document Sponsor: Controlled Document Lead:
 CONTROLLED DOCUMENT Procedure for the management of patients with blood borne viruses CATEGORY: CLASSIFICATION: PURPOSE Controlled Document Number: Version Number: 4 Controlled Document Sponsor: Controlled
CONTROLLED DOCUMENT Procedure for the management of patients with blood borne viruses CATEGORY: CLASSIFICATION: PURPOSE Controlled Document Number: Version Number: 4 Controlled Document Sponsor: Controlled
INSTRUCTIONS FOR BLOOD BANK INSPECTION CHECKLIST AND REPORT CMS-282
 Note to Surveyors: INSTRUCTIONS FOR BLOOD BANK INSPECTION CHECKLIST AND REPORT CMS-282 This form consists of portions of the FDA-2609. These portions are the only parts of the FDA-2609 that have been approved
Note to Surveyors: INSTRUCTIONS FOR BLOOD BANK INSPECTION CHECKLIST AND REPORT CMS-282 This form consists of portions of the FDA-2609. These portions are the only parts of the FDA-2609 that have been approved
TGA regulation of local manufacturers of plasma derivatives CSL Behring Australia s overview
 IPFA Workshop Session 7: REGULATORY CONSIDERATIONS TGA regulation of local manufacturers of plasma derivatives CSL Behring Australia s overview Mary Lavithis, CSL Behring Australia Presented at: IPFA Asia
IPFA Workshop Session 7: REGULATORY CONSIDERATIONS TGA regulation of local manufacturers of plasma derivatives CSL Behring Australia s overview Mary Lavithis, CSL Behring Australia Presented at: IPFA Asia
The Gift of Life. Organ Donation and its Challenges Presented by Jessica Roth, RN, BSN, CPTC
 The Gift of Life Organ Donation and its Challenges Presented by Jessica Roth, RN, BSN, CPTC Nebraska Organ Recovery System (NORS) Federally designated as an organ procurement organization (OPO) Tasked
The Gift of Life Organ Donation and its Challenges Presented by Jessica Roth, RN, BSN, CPTC Nebraska Organ Recovery System (NORS) Federally designated as an organ procurement organization (OPO) Tasked
DHQ Flowcharts v2.0 eff. February 2016
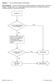 Question: 1. Are you feeling healthy and well today? Donor Eligibility: A person should be free of infectious diseases, including colds, on the day of donation. A person who is not in good health should
Question: 1. Are you feeling healthy and well today? Donor Eligibility: A person should be free of infectious diseases, including colds, on the day of donation. A person who is not in good health should
December 20, Dear Dr. Frieden:
 December 20, 2011 The Honorable Thomas Frieden, MD, MPH Director, Centers for Disease Control and Prevention 1600 Clifton Road, NE. Mailstop A-07 Atlanta, GA 30329 Ref: [Docket No. CDC-2011-0011]; Public
December 20, 2011 The Honorable Thomas Frieden, MD, MPH Director, Centers for Disease Control and Prevention 1600 Clifton Road, NE. Mailstop A-07 Atlanta, GA 30329 Ref: [Docket No. CDC-2011-0011]; Public
Maintaining a Safe and Adequate Blood Supply in the Event of Pandemic Influenza. Guidelines for National Blood Transfusion Services
 Maintaining a Safe and Adequate Blood Supply in the Event of Pandemic Influenza Guidelines for National Blood Transfusion Services 19 May 2006 1 Rationale Current global concern that an occurrence of pandemic
Maintaining a Safe and Adequate Blood Supply in the Event of Pandemic Influenza Guidelines for National Blood Transfusion Services 19 May 2006 1 Rationale Current global concern that an occurrence of pandemic
Juventas SHARING LIFE CONSENT FOR PLASMA INFUSION
 Juventas SHARING LIFE Print Name Date of Birth CONSENT FOR PLASMA INFUSION To the Patient or Guardian of the Patient: The purpose of this document is to provide written information as of (month and day),
Juventas SHARING LIFE Print Name Date of Birth CONSENT FOR PLASMA INFUSION To the Patient or Guardian of the Patient: The purpose of this document is to provide written information as of (month and day),
HEALTH SCREENING QUESTIONNAIRE. Work-up. Donor ID EdgeCell #:
 HEALTH SCREENING QUESTIONNAIRE Héma-Québec Registre des Donneurs de Cellules Souches 4045 Côte-vertu, St-Laurent, QC, Canada, H4R 2W7 Tél : + 514-832-1031 Fax : + 514-832-0266 www.hema-quebec.qc.ca CT
HEALTH SCREENING QUESTIONNAIRE Héma-Québec Registre des Donneurs de Cellules Souches 4045 Côte-vertu, St-Laurent, QC, Canada, H4R 2W7 Tél : + 514-832-1031 Fax : + 514-832-0266 www.hema-quebec.qc.ca CT
HEPATITIS SCREENING IN THE LITHUANIAN BLOOD ESTABLISHMENTS. Joana Bikulčienė Director of National blood center
 HEPATITIS SCREENING IN THE LITHUANIAN BLOOD ESTABLISHMENTS Joana Bikulčienė Director of National blood center j.bikulciene@kraujodonoryste.lt Organizational structure of Lithuanian blood service 1 specialized
HEPATITIS SCREENING IN THE LITHUANIAN BLOOD ESTABLISHMENTS Joana Bikulčienė Director of National blood center j.bikulciene@kraujodonoryste.lt Organizational structure of Lithuanian blood service 1 specialized
TSDR Pharmacy Inc. dba brandmd Skin Care 11/9/17
 TSDR Pharmacy Inc. dba brandmd Skin Care 11/9/17 Division of Pharmaceutical Quality Operations IV 19701 Fairchild, Irvine, CA 92612-2506 Telephone: 949-608-2900 Fax: 949-608-4417 WARNING LETTER VIA SIGNATURE
TSDR Pharmacy Inc. dba brandmd Skin Care 11/9/17 Division of Pharmaceutical Quality Operations IV 19701 Fairchild, Irvine, CA 92612-2506 Telephone: 949-608-2900 Fax: 949-608-4417 WARNING LETTER VIA SIGNATURE
FORM TITLE: Risk Behavior Assessment. 1. Are you a male who has had sex with another male in the preceding five years?
 RELESE DTE: 12/07/2012 EFFECTIVE DTE: 12/13/2012 1. re you a male who has had sex with another male in the preceding five years? 2. Have you injected drugs for non-medical reason in the preceding five
RELESE DTE: 12/07/2012 EFFECTIVE DTE: 12/13/2012 1. re you a male who has had sex with another male in the preceding five years? 2. Have you injected drugs for non-medical reason in the preceding five
