Molecular mechanism of filament branching by WASP- Arp2/3 complex: possible models and role of the WH2 domain of WASP proteins
|
|
|
- Jordan Benson
- 5 years ago
- Views:
Transcription
1 Molecular mechanism of filament branching by WASP- Arp2/3 complex: possible models and role of the WH2 domain of WASP proteins 1. Filament branching in vivo and in reconstituted motility assays in vitro In living cells, formation of branched filament arrays is used in various motile processes to produce protrusive, compressive and propulsive forces. Pioneering studies have shown that Arp2/3 complex, which sits at the branched junction, is responsible for filament branching driving lamellipodial extension 1. Various proteins of the WASP family (comprising WASP, N- WASP, WAVE and WASH regulatory complexes, WHAMM and JMY 2 act in a stimulus- responsive fashion, once activated at membranes, to catalyze filament branching. They all use Arp2/3 complex. Reconstituted motility assays have demonstrated that as part of the branching cycle, Arp2/3 first binds to N- WASP coated beads 3. At each catalytic cycle of filament branching, WASP- Arp2/3 complex interacts with a growing filament and Arp2/3 complex is delivered at the branch junction. As filaments grow, the repeated cycle perpetuates the fractal pattern of branched filaments. Evidence for this process has also been obtained in vivo 4, 5. Arp2/3 molecules get incorporated into the branched array at the same rate at which actin is assembled 3, accounting for in vivo observations 1, 6. In electron microscopy studies, newly assembled filaments in the actin tail have been observed with barbed ends attached to Listeria 7, or to ActA- coated beads 8 or to baculovirus 9, consistent with the tethered ratchet model for actin- based motility 10, Biochemical approaches of filament branching by WASP family proteins with Arp2/3 complex The C- terminal constitutively active catalytic domain of all WASP proteins, often called VCA, harbors a WH2 domain (V, sometimes two WH2 domains in tandem repeat) followed by a connector (C ) region and an short acidic (A) sequence. The VCA domain of all WASP family members branches filaments in bulk solution using Arp2/3 complex. The respective roles of the three regions V, C and A, in the branching process have been extensively studied. Branching requires at least V and C, but is most effective with VCA. Hence the WH2 domain (V) is essential for branching. VCA and CA bind Arp2/3 complex with identical affinities (KD = 0.1 µm) and promote the same conformational change of Arp2/3 complex 12, 13. The conformational change in VCA- Arp2/3 complex 14 is linked to a large increase in affinity of Arp2 subunit for ATP 15. Although sufficient for binding to Arp2/3 complex and inducing the same structural change as VCA, CA by itself does not activate Arp2/3 and simply acts as a sequesterer of Arp2/3 complex by competing out VCA 12, 16. Altogether, these results indicate that 1) the structural change promoted by binding of VCA to Arp2/3 complex is insufficient for branching; 2) the WH2 domain is essential for branching without being involved in the binding of Arp2/3 complex. Moreover, the branching efficiency greatly varies from one WASP protein to the other,
2 while all VCAs bind Arp2/3 with very similar affinities 17, 18. The WH2 domain thus plays a crucial role in specifying branching efficiency, independently of Arp2/3 binding 19. Covalent crosslinking experiments suggest that the CA region interacts with several of the seven subunits of Arp2/3 complex, namely Arp2, Arp3, p40 (ARPC1) and p21 (ARPC3) 18, The amphipathic α- helix of the C region remarkably binds the hydrophobic pocket at the barbed face of Arp2, in a similar way to WH2 domains 23, 24. In the presence of G- actin only, VCA binds G- actin via its WH2 domain (KD = 1 µm) in a way that does not affect the binding strength of Arp2/3 to the CA moiety. The VCA- actin complex then participates in barbed- end assembly like profilin- actin 15, 25-27, i.e. VCA dissociates from the barbed end following addition of the VCA- bound actin subunit. The VCA- enhanced ATP binding to Arp2 is not affected by binding of G- actin to the WH2 domain of VCA 15. Fluorescence and size exclusion chromatography studies indicate that both 1:1 VCA- Arp2/3 complex and 1:1:1 actin- VCA- Arp2/3 complexes form in solution in vitro 15, 26, 28, Proposed models for filament branching There is general agreement that filament branching is an autocatalytic reaction, i.e. binding of VCA- Arp2/3 complex to a mother filament is required to initiate a daughter filament that grows at a 70 angle from the growing mother filament 30, 31. The Arp2/3 complex is embedded in the branch junction. Release of WASP from the branch junction terminates the catalytic branching cycle restoring the free enzyme (WASP) and allows daughter branch growth 32. Two different models have been proposed so far: the side- branching model and barbed- end branching model. The proposed side- branching model 33 (for review) posits that WASP proteins first activate the Arp2/3 complex by moving Arp2 by about 30 Å from its position in the isolated state of the complex 34 to a position in which it makes a short pitch connection with Arp3, thus mimicking two consecutive F- actin subunits. The low resolution EM structures of 1:1 WASP- Arp2/3 or VCA- Arp2/3 complex can accommodate this hypothesis 14. The complex of WASP proteins with Arp2/3 then associates with the side of a filament, in the presence of G- actin. The adjacent WH2 domain adjacent to CA, which is essential for branching, binds a G- actin molecule and places it as the third subunit of the daughter filament 35, the first one being Arp3, the second one Arp2. This reaction nucleates the growth of the daughter filament. 3D- reconstructions of the branched junction support this structural organization 35. Notably, this model implicitly posits that the ternary complex of VCA with G- actin and Arp2/3 forms in the presence of filaments, as well as in the presence of G- actin only.
3 The side- branching model faces some internal difficulties. SAXS studies of Arp2/3 in complex with VCA and actin and 3D- reconstructions of the branch junction provide conflicting views of the positions of Arp2 and Arp3 in the daughter filament 23, 35, 36. 3D- reconstructions of VCA or N- WASP in complex with Arp2/3 show that the bound WASP protein occludes the actin filament side- binding site and indicate that VCA is bound to the pointed end of the Arp2- Arp3 heterodimer 14. To resolve these difficulties, it was recently proposed that a second VCA molecule binds Arp2/3 complex to achieve optimum branching, a view supported by the more efficient filament branching of GST- dimerized constructs of VCA 37. This second site, also seen in X- ray crystallographic studies, has a ten- to 100- fold lower affinity 29, 36, 38, which explains why only 1:1 VCA- Arp2/3 and 1:1:1 G- actin- VCA- Arp2/3 complexes have been detected by size- exclusion chromatography 23, 26. Efficient polymerization in branched filaments is monitored in the presence of nm VCA, conditions under which binding of two VCA molecules to Arp2/3 complex may not be significant. Efficient propulsion of N- WASP coated beads takes place at a density of immobilized N- WASP lower than close contact, precluding dimerization. Similarly, the WAVE regulatory complex 39 contains only one WAVE molecule. Whether appropriate dimerization of the WRC allowing binding of two WAVE molecules to a single Arp2/3 complex occurs in vivo is not known. Single molecule imaging of the branching process show that daughter filament nucleation is triggered by the dissociation of a single VCA molecule, initially bound to Arp2/3, from the branch junction 32. An additional difficulty with the side- branching model is that in the physiological context, G- actin is essentially bound to profilin, which is two orders of magnitude more abundant than WH2 domains of activated WASP proteins and has a one order of magnitude higher affinity. Under these conditions, binding of WH2 to G- actin is problematic, while formation of dendritic arrays 40, 41 appears stimulated. The barbed- end branching model was proposed based on evidences for equal length of mother and daughter filaments in EM images and TIRF microscopy video recordings, and for thermodynamic antagonism between branching machineries and proteins targeting barbed ends, including cappers, formins, WH2 domain polymerases and profilin 31, 42, 43. No structural organization of the branched junction at barbed ends was proposed. In physiological solutions containing F- actin and profilin- actin, the barbed end of the mother filament appears as a plausible binding site for the WH2 in the WASP- Arp2/3 complex. In this putative scenario 43, the WH2 domain of VCA would target the terminal actin subunit at the barbed end of a mother filament while the adjacent CA moiety of VCA bound to Arp2/3 complex would simultaneously be positioned at this barbed end to initiate growth of the daughter branch at 70 angle from the mother filament. In this model, the binding of VCA to Arp2/3 complex does not occlude its association to the filament. The CA- Arp2/3 moiety may contribute in transient stabilization of the WH2 domain at the barbed end. Growth of both the mother and daughter filaments may occur from profilin- actin with no constraint, following dissociation of VCA.
4 Remarkably, in the barbed- end branching model, the WH2 domain of WASP proteins shares a functional similarity with other WH2 domain barbed end trackers. Corroborating reports indicate that the WH2 of WASP captures barbed ends at least in absence of Arp2/3 complex 44, the WH2 of WAVE1 inhibits barbed end growth at nanomolar concentrations 19, and N- WASP remains processively bound to filament barbed ends 45. These observations add to other evidences for the multifunctionality of WH2 domains and the binding of Spire, VASP and VopF to filament barbed ends via their WH2 domains 46 (for review). In the barbed- end branching model, profilin is expected to compete with the WH2 domain of WASP proteins at barbed ends to inhibit branching, at high concentrations (>10 µm profilin), which accounts for in vitro observations 43, 47. Finally, insertion of the WH2 domain in the filament to find its binding site, as gelsolin, Spire, INF2 or Cordon- Bleu do to sever filaments 48 (for review), may lead to side branching by WASP- Arp2/3. This alternate model includes both barbed- end and side- branching reactions logically, in a way that leads to an identical structural organization of the branch junction. In conclusion, within this model, WASP family proteins appear as new members of the barbed end tracking machineries that control site- directed actin- based motility. The barbed- end branching model satisfactorily accounts for the perpetuation of the polarized, site- directed assembly of the dendritic actin array. Formation of branches that point away from the lamellipodium tip does not happen in this model. Testing the side- and barbed- end branching models requires biochemical and structural data using various mutated Arp2/3 complexes and engineered VCA domains harboring mutations in the WH2 moiety. Structural modeling studies are also required to examine the possibility that the WH2 domain of VCA binds the barbed end of a mother filament to initiate branching. 4. Possible technical issues linked to preferential observation of barbed- end or side- branching of filaments in TIRF microscopy In a number of studies performed on coverslip- immobilized single filaments, only side- branching reactions have been recorded from the side of adsorbed filaments subsequently exposed to G- actin, VCA and Arp2/3 complex Using a different protocol, by initializing branched filament assembly in a test tube before placing the solution in a chamber for TIRF microscopy observation or on a grid for EM observation, both barbed- end branching and side branching events were recorded 53, 31. More recently, barbed- end branching was monitored on single filaments using a protocol that avoids immobilization on the coverslip, thus preserving the freedom of filament barbed ends 43. It has been suggested that differences in coverslip passivation or in filament
5 immobilization, causing more or less restricted motion of filament barbed ends could affect the detection of barbed end branching. So far, the low frequency of side branching has been measured in only two studies 43, 52. The frequency of barbed end branching was measured in only one paper 43. More detailed investigations are required to establish a consensus view of the mechanism of filament branching, a pivotal reaction in a plethora of motile processes. References 1. Svitkina, T.M. & Borisy, G.G. Arp2/3 complex and actin depolymerizing factor/cofilin in dendritic organization and treadmilling of actin filament array in lamellipodia. J Cell Biol 145, (1999). 2. Rotty, J.D., Wu, C. & Bear, J.E. New insights into the regulation and cellular functions of the ARP2/3 complex. Nat Rev Mol Cell Biol 14, 7-12 (2013). 3. Wiesner, S. et al. A biomimetic motility assay provides insight into the mechanism of actin- based motility. J Cell Biol 160, (2003). 4. Millius, A., Watanabe, N. & Weiner, O.D. Diffusion, capture and recycling of SCAR/WAVE and Arp2/3 complexes observed in cells by single- molecule imaging. J Cell Sci 125, (2012). 5. Weisswange, I., Newsome, T.P., Schleich, S. & Way, M. The rate of N- WASP exchange limits the extent of ARP2/3- complex- dependent actin- based motility. Nature 458, (2009). 6. Iwasa, J.H. & Mullins, R.D. Spatial and temporal relationships between actin- filament nucleation, capping, and disassembly. Curr Biol 17, (2007). 7. Zhukarev, V., Ashton, F., Sanger, J.M., Sanger, J.W. & Shuman, H. Organization and structure of actin filament bundles in Listeria- infected cells. Cell Motil Cytoskeleton 30, (1995). 8. Cameron, L.A., Svitkina, T.M., Vignjevic, D., Theriot, J.A. & Borisy, G.G. Dendritic organization of actin comet tails. Curr Biol 11, (2001). 9. Mueller, J. et al. Electron tomography and simulation of baculovirus actin comet tails support a tethered filament model of pathogen propulsion. PLoS Biol 12, e (2014). 10. Demoulin, D., Carlier, M.F., Bibette, J. & Baudry, J. Power transduction of actin filaments ratcheting in vitro against a load. Proc Natl Acad Sci U S A 111, (2014). 11. Mogilner, A. & Oster, G. Force generation by actin polymerization II: the elastic ratchet and tethered filaments. Biophys J 84, (2003). 12. Hufner, K. et al. The verprolin- like central (vc) region of Wiskott- Aldrich syndrome protein induces Arp2/3 complex- dependent actin nucleation. J Biol Chem 276, (2001). 13. Rodnick- Smith, M., Luan, Q., Liu, S.L. & Nolen, B.J. Role and structural mechanism of WASP- triggered conformational changes in branched actin filament nucleation by Arp2/3 complex. Proc Natl Acad Sci U S A (2016). 14. Xu, X.P. et al. Three- dimensional reconstructions of Arp2/3 complex with bound nucleation promoting factors. EMBO J 31, (2012).
6 15. Le Clainche, C., Didry, D., Carlier, M.F. & Pantaloni, D. Activation of Arp2/3 complex by Wiskott- Aldrich Syndrome protein is linked to enhanced binding of ATP to Arp2. J Biol Chem 276, (2001). 16. Rohatgi, R. et al. The interaction between N- WASP and the Arp2/3 complex links Cdc42- dependent signals to actin assembly. Cell 97, (1999). 17. Millard, T.H., Sharp, S.J. & Machesky, L.M. Signalling to actin assembly via the WASP (Wiskott- Aldrich syndrome protein)- family proteins and the Arp2/3 complex. Biochem J 380, 1-17 (2004). 18. Zalevsky, J., Lempert, L., Kranitz, H. & Mullins, R.D. Different WASP family proteins stimulate different Arp2/3 complex- dependent actin- nucleating activities. Curr Biol 11, (2001). 19. Sweeney, M.O., Collins, A., Padrick, S.B. & Goode, B.L. A novel role for WAVE1 in controlling actin network growth rate and architecture. Mol Biol Cell 26, (2015). 20. Balcer, H.I., Daugherty- Clarke, K. & Goode, B.L. The p40/arpc1 subunit of Arp2/3 complex performs multiple essential roles in WASp- regulated actin nucleation. J Biol Chem 285, (2010). 21. Weaver, A.M. et al. Interaction of cortactin and N- WASp with Arp2/3 complex. Curr Biol 12, (2002). 22. Kelly, A.E., Kranitz, H., Dotsch, V. & Mullins, R.D. Actin binding to the central domain of WASP/Scar proteins plays a critical role in the activation of the Arp2/3 complex. J Biol Chem 281, (2006). 23. Boczkowska, M. et al. X- ray scattering study of activated Arp2/3 complex with bound actin- WCA. Structure 16, (2008). 24. Panchal, S.C., Kaiser, D.A., Torres, E., Pollard, T.D. & Rosen, M.K. A conserved amphipathic helix in WASP/Scar proteins is essential for activation of Arp2/3 complex. Nat Struct Biol 10, (2003). 25. Egile, C. et al. Activation of the CDC42 effector N- WASP by the Shigella flexneri IcsA protein promotes actin nucleation by Arp2/3 complex and bacterial actin- based motility. J Cell Biol 146, (1999). 26. Gaucher, J.F. et al. Interactions of isolated C- terminal fragments of neural Wiskott- Aldrich syndrome protein (N- WASP) with actin and Arp2/3 complex. J Biol Chem 287, (2012). 27. Higgs, H.N., Blanchoin, L. & Pollard, T.D. Influence of the C terminus of Wiskott- Aldrich syndrome protein (WASp) and the Arp2/3 complex on actin polymerization. Biochemistry 38, (1999). 28. Marchand, J.B., Kaiser, D.A., Pollard, T.D. & Higgs, H.N. Interaction of WASP/Scar proteins with actin and vertebrate Arp2/3 complex. Nat Cell Biol 3, (2001). 29. Ti, S.C., Jurgenson, C.T., Nolen, B.J. & Pollard, T.D. Structural and biochemical characterization of two binding sites for nucleation- promoting factor WASp- VCA on Arp2/3 complex. Proc Natl Acad Sci U S A 108, E (2011). 30. Machesky, L.M. et al. Scar, a WASp- related protein, activates nucleation of actin filaments by the Arp2/3 complex. Proc Natl Acad Sci U S A 96, (1999). 31. Pantaloni, D., Boujemaa, R., Didry, D., Gounon, P. & Carlier, M.F. The Arp2/3 complex branches filament barbed ends: functional antagonism with capping proteins. Nat Cell Biol 2, (2000). 32. Smith, B.A. et al. Three- color single molecule imaging shows WASP detachment from Arp2/3 complex triggers actin filament branch formation. Elife 2, e01008 (2013).
7 33. Pollard, T.D. Regulation of actin filament assembly by Arp2/3 complex and formins. Annu Rev Biophys Biomol Struct 36, (2007). 34. Robinson, R.C. et al. Crystal structure of Arp2/3 complex. Science 294, (2001). 35. Rouiller, I. et al. The structural basis of actin filament branching by the Arp2/3 complex. J Cell Biol 180, (2008). 36. Boczkowska, M., Rebowski, G., Kast, D.J. & Dominguez, R. Structural analysis of the transitional state of Arp2/3 complex activation by two actin- bound WCAs. Nat Commun 5, 3308 (2014). 37. Padrick, S.B., Doolittle, L.K., Brautigam, C.A., King, D.S. & Rosen, M.K. Arp2/3 complex is bound and activated by two WASP proteins. Proc Natl Acad Sci U S A 108, E472-9 (2011). 38. Hetrick, B., Han, M.S., Helgeson, L.A. & Nolen, B.J. Small molecules CK- 666 and CK- 869 inhibit actin- related protein 2/3 complex by blocking an activating conformational change. Chem Biol 20, (2013). 39. Chen, Z. et al. Structure and control of the actin regulatory WAVE complex. Nature 468, (2010). 40. Blanchoin, L. et al. Direct observation of dendritic actin filament networks nucleated by Arp2/3 complex and WASP/Scar proteins. Nature 404, (2000). 41. Theriot, J.A., Rosenblatt, J., Portnoy, D.A., Goldschmidt- Clermont, P.J. & Mitchison, T.J. Involvement of profilin in the actin- based motility of L. monocytogenes in cells and in cell- free extracts. Cell 76, (1994). 42. Falet, H. et al. Importance of free actin filament barbed ends for Arp2/3 complex function in platelets and fibroblasts. Proc Natl Acad Sci U S A 99, (2002). 43. Pernier, J., Shekhar, S., Jegou, A., Guichard, B. & Carlier, M.F. Profilin Interaction with Actin Filament Barbed End Controls Dynamic Instability, Capping, Branching, and Motility. Dev Cell 36, (2016). 44. Co, C., Wong, D.T., Gierke, S., Chang, V. & Taunton, J. Mechanism of actin network attachment to moving membranes: barbed end capture by N- WASP WH2 domains. Cell 128, (2007). 45. Khanduja, N. & Kuhn, J.R. Processive acceleration of actin barbed- end assembly by N- WASP. Mol Biol Cell 25, (2014). 46. Carlier, M.F. et al. Control of polarized assembly of actin filaments in cell motility. Cell Mol Life Sci 72, (2015). 47. Suarez, C. et al. Profilin regulates F- actin network homeostasis by favoring formin over Arp2/3 complex. Dev Cell 32, (2015). 48. Carlier, M.F., Pernier, J. & Avvaru, B.S. Control of actin filament dynamics at barbed ends by WH2 domains: from capping to permissive and processive assembly. Cytoskeleton (Hoboken) 70, (2013). 49. Amann, K.J. & Pollard, T.D. Direct real- time observation of actin filament branching mediated by Arp2/3 complex using total internal reflection fluorescence microscopy. Proc Natl Acad Sci U S A 98, (2001). 50. Mahaffy, R.E. & Pollard, T.D. Kinetics of the formation and dissociation of actin filament branches mediated by Arp2/3 complex. Biophys J 91, (2006). 51. Risca, V.I. et al. Actin filament curvature biases branching direction. Proc Natl Acad Sci U S A 109, (2012).
8 52. Smith, B.A., Daugherty- Clarke, K., Goode, B.L. & Gelles, J. Pathway of actin filament branch formation by Arp2/3 complex revealed by single- molecule imaging. Proc Natl Acad Sci U S A 110, (2013). 53. Fujiwara, I., Suetsugu, S., Uemura, S., Takenawa, T. & Ishiwata, S. Visualization and force measurement of branching by Arp2/3 complex and N- WASP in actin filament. Biochem Biophys Res Commun 293, (2002).
The extension of cell protrusions, generated by site-directed
 ATP hydrolysis on actin-related protein 2 3 complex causes debranching of dendritic actin arrays Christophe Le Clainche, Dominique Pantaloni, and Marie-France Carlier* Dynamique du Cytosquelette et Motilité
ATP hydrolysis on actin-related protein 2 3 complex causes debranching of dendritic actin arrays Christophe Le Clainche, Dominique Pantaloni, and Marie-France Carlier* Dynamique du Cytosquelette et Motilité
MCB Topic 19 Regulation of Actin Assembly- Prof. David Rivier
 MCB 252 -Topic 19 Regulation of Actin Assembly- Prof. David Rivier MCB 252 Spring 2017 MCB 252 Cell Biology Topic 19 Regulation of Actin Assembly Reading: Lodish 17.2-17.3, 17.7 MCB 252 Actin Cytoskeleton
MCB 252 -Topic 19 Regulation of Actin Assembly- Prof. David Rivier MCB 252 Spring 2017 MCB 252 Cell Biology Topic 19 Regulation of Actin Assembly Reading: Lodish 17.2-17.3, 17.7 MCB 252 Actin Cytoskeleton
CMB621: Cytoskeleton. Also known as How the cell plays with LEGOs to ensure order, not chaos, is temporally and spatially achieved
 CMB621: Cytoskeleton Also known as How the cell plays with LEGOs to ensure order, not chaos, is temporally and spatially achieved Lecture(s) Overview Lecture 1: What is the cytoskeleton? Membrane interaction
CMB621: Cytoskeleton Also known as How the cell plays with LEGOs to ensure order, not chaos, is temporally and spatially achieved Lecture(s) Overview Lecture 1: What is the cytoskeleton? Membrane interaction
Regulation of Actin Assembly Associated With Protrusion and Adhesion in Cell Migration
 Physiol Rev 88: 489 513, 2008; doi:10.1152/physrev.00021.2007. Regulation of Actin Assembly Associated With Protrusion and Adhesion in Cell Migration CHRISTOPHE LE CLAINCHE AND MARIE-FRANCE CARLIER Laboratoire
Physiol Rev 88: 489 513, 2008; doi:10.1152/physrev.00021.2007. Regulation of Actin Assembly Associated With Protrusion and Adhesion in Cell Migration CHRISTOPHE LE CLAINCHE AND MARIE-FRANCE CARLIER Laboratoire
Cytoskeleton and cell communication
 Cytoskeleton and cell communication Actin monomer has subdomains 1-4. A simplified cartoon is at right. ATP binds, along with Mg ++, within a deep cleft between subdomains 2 & 4. G-actin (globular actin),
Cytoskeleton and cell communication Actin monomer has subdomains 1-4. A simplified cartoon is at right. ATP binds, along with Mg ++, within a deep cleft between subdomains 2 & 4. G-actin (globular actin),
Actin structure. Actin a highly conserved gene. Molecular Cell Biology
 Harvey Lodish Arnold Berk Paul Matsudaira Chris A. Kaiser Monty Krieger Matthew P. Scott Lawrence Zipursky James Darnell Molecular Cell Biology Fifth Edition Chapter 19: Cytoskeleton I: Microfilaments
Harvey Lodish Arnold Berk Paul Matsudaira Chris A. Kaiser Monty Krieger Matthew P. Scott Lawrence Zipursky James Darnell Molecular Cell Biology Fifth Edition Chapter 19: Cytoskeleton I: Microfilaments
Molecular Cell Biology - Problem Drill 20: Cytoskeleton and Cellular Mobility
 Molecular Cell Biology - Problem Drill 20: Cytoskeleton and Cellular Mobility Question No. 1 of 10 1. Which of the following statements about cytoskeletal filaments is correct? Question #1 (A) The Cytoskeleton
Molecular Cell Biology - Problem Drill 20: Cytoskeleton and Cellular Mobility Question No. 1 of 10 1. Which of the following statements about cytoskeletal filaments is correct? Question #1 (A) The Cytoskeleton
cell movement and neuronal migration
 cell movement and neuronal migration Paul Letourneau letou001@umn.edu Chapter 16; The Cytoskeleton; Molecular Biology of the Cell, Alberts et al. 1 Cell migration Cell migration in 3 steps; protrusion,
cell movement and neuronal migration Paul Letourneau letou001@umn.edu Chapter 16; The Cytoskeleton; Molecular Biology of the Cell, Alberts et al. 1 Cell migration Cell migration in 3 steps; protrusion,
Hui Chen, Barbara W. Bernstein and James R. Bamburg
 IBS 25 JANUARY 2000 sequence-specific NA binding by acetylation of the p53 C-terminal domain. Cell 90, 595 606 41 Liu, L. et al. (1999) p53 sites acetylated in vitro by PCAF and p300 are acetylated in
IBS 25 JANUARY 2000 sequence-specific NA binding by acetylation of the p53 C-terminal domain. Cell 90, 595 606 41 Liu, L. et al. (1999) p53 sites acetylated in vitro by PCAF and p300 are acetylated in
Microtubule Forces Kevin Slep
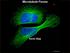 Microtubule Forces Kevin Slep Microtubules are a Dynamic Scaffold Microtubules in red, XMA215 family MT polymerase protein in green Some Microtubule Functions Cell Structure Polarized Motor Track (kinesins
Microtubule Forces Kevin Slep Microtubules are a Dynamic Scaffold Microtubules in red, XMA215 family MT polymerase protein in green Some Microtubule Functions Cell Structure Polarized Motor Track (kinesins
In vitro studies of actin filament and network dynamics R Dyche Mullins and Scott D Hansen
 Available online at www.sciencedirect.com In vitro studies of actin filament and network dynamics R Dyche Mullins and Scott D Hansen Now that many genomes have been sequenced, a central concern of cell
Available online at www.sciencedirect.com In vitro studies of actin filament and network dynamics R Dyche Mullins and Scott D Hansen Now that many genomes have been sequenced, a central concern of cell
EMBO. Arp2/3 complex interactions and actin network turnover in lamellipodia EMBO. open
 The EMBO Journal (2008) 27, 982 992 & 2008 European Molecular Biology Organization Some Rights Reserved 0261-4189/08 www.embojournal.org Arp2/3 complex interactions and actin network turnover in lamellipodia
The EMBO Journal (2008) 27, 982 992 & 2008 European Molecular Biology Organization Some Rights Reserved 0261-4189/08 www.embojournal.org Arp2/3 complex interactions and actin network turnover in lamellipodia
GMFβ CONTROLS BRANCHED ACTIN CONTENT AND LAMELLIPODIAL RETRACTION IN FIBROBLASTS. Elizabeth M. Haynes. Chapel Hill 2015
 GMFβ CONTROLS BRANCHED ACTIN CONTENT AND LAMELLIPODIAL RETRACTION IN FIBROBLASTS Elizabeth M. Haynes A dissertation submitted to the faculty of the University of North Carolina at Chapel Hill in partial
GMFβ CONTROLS BRANCHED ACTIN CONTENT AND LAMELLIPODIAL RETRACTION IN FIBROBLASTS Elizabeth M. Haynes A dissertation submitted to the faculty of the University of North Carolina at Chapel Hill in partial
Actin-based motility of intracellular pathogens Edith Gouin 1, Matthew D Welch 2 and Pascale Cossart 1
 Actin-based motility of intracellular pathogens Edith Gouin 1, Matthew D Welch 2 and Pascale Cossart 1 The actin cytoskeleton is harnessed by several pathogenic bacteria that are capable of entering into
Actin-based motility of intracellular pathogens Edith Gouin 1, Matthew D Welch 2 and Pascale Cossart 1 The actin cytoskeleton is harnessed by several pathogenic bacteria that are capable of entering into
Introduction to Cofilin and its Regulation of Actin Dynamics
 Article ID: ISSN 2046-1690 Introduction to Cofilin and its Regulation of Actin Dynamics Author(s):Mr. Kuen Yeow Chin Corresponding Author: Mr. Kuen Yeow Chin, MSc, Department of Cell and Developmental
Article ID: ISSN 2046-1690 Introduction to Cofilin and its Regulation of Actin Dynamics Author(s):Mr. Kuen Yeow Chin Corresponding Author: Mr. Kuen Yeow Chin, MSc, Department of Cell and Developmental
Molecular Cell Biology 5068 In Class Exam 1 September 29, Please print your name:
 Molecular Cell Biology 5068 In Class Exam 1 September 29, 2015 Exam Number: Please print your name: Instructions: Please write only on these pages, in the spaces allotted and not on the back. Write your
Molecular Cell Biology 5068 In Class Exam 1 September 29, 2015 Exam Number: Please print your name: Instructions: Please write only on these pages, in the spaces allotted and not on the back. Write your
ZYXIN-VASP INTERACTIONS ALTER ACTIN REGULATORY ACTIVITY IN ZYXIN-VASP COMPLEXES
 CELLULAR & MOLECULAR BIOLOGY LETTERS http://www.cmbl.org.pl Received: 20 June 2012 Volume 18 (2013) pp 1-10 Final form accepted: 12 October 2012 DOI: 10.2478/s11658-012-0035-2 Published online: 17 October
CELLULAR & MOLECULAR BIOLOGY LETTERS http://www.cmbl.org.pl Received: 20 June 2012 Volume 18 (2013) pp 1-10 Final form accepted: 12 October 2012 DOI: 10.2478/s11658-012-0035-2 Published online: 17 October
Physiol Rev VOL 94 JANUARY ACTIN DYNAMICS, ARCHITECTURE, AND MECHANICS. ii) iv) iii) Cortex crosslinked networks
 Physiol Rev 94: 235 263, 2014 doi:10.1152/physrev.00018.2013 ACTIN DYNAMICS, ARCHITECTURE, AND MECHANICS IN CELL MOTILITY Laurent Blanchoin, Rajaa Boujemaa-Paterski, Cécile Sykes, and Julie Plastino Laboratoire
Physiol Rev 94: 235 263, 2014 doi:10.1152/physrev.00018.2013 ACTIN DYNAMICS, ARCHITECTURE, AND MECHANICS IN CELL MOTILITY Laurent Blanchoin, Rajaa Boujemaa-Paterski, Cécile Sykes, and Julie Plastino Laboratoire
Cell Cycle, Mitosis, and Microtubules. LS1A Final Exam Review Friday 1/12/07. Processes occurring during cell cycle
 Cell Cycle, Mitosis, and Microtubules LS1A Final Exam Review Friday 1/12/07 Processes occurring during cell cycle Replicate chromosomes Segregate chromosomes Cell divides Cell grows Cell Growth 1 The standard
Cell Cycle, Mitosis, and Microtubules LS1A Final Exam Review Friday 1/12/07 Processes occurring during cell cycle Replicate chromosomes Segregate chromosomes Cell divides Cell grows Cell Growth 1 The standard
The many faces of actin: matching assembly factors with cellular structures
 The many faces of actin: matching assembly factors with cellular structures Ekta Seth Chhabra and Henry N. Higgs Actin filaments are major components of at least 15 distinct structures in metazoan cells.
The many faces of actin: matching assembly factors with cellular structures Ekta Seth Chhabra and Henry N. Higgs Actin filaments are major components of at least 15 distinct structures in metazoan cells.
Supplementary Materials. High affinity binding of phosphatidylinositol-4-phosphate. by Legionella pneumophila DrrA
 Supplementary Materials High affinity binding of phosphatidylinositol-4-phosphate by Legionella pneumophila DrrA Running title: Molecular basis of PtdIns(4)P-binding by DrrA Stefan Schoebel, Wulf Blankenfeldt,
Supplementary Materials High affinity binding of phosphatidylinositol-4-phosphate by Legionella pneumophila DrrA Running title: Molecular basis of PtdIns(4)P-binding by DrrA Stefan Schoebel, Wulf Blankenfeldt,
Actin Disassembly by Cofilin, Coronin, and Aip1 Occurs in Bursts and Is Inhibited by Barbed-end Cappers
 Actin Disassembly by Cofilin, Coronin, and Aip1 Occurs in Bursts and Is Inhibited by Barbed-end Cappers The Harvard community has made this article openly available. Please share how this access benefits
Actin Disassembly by Cofilin, Coronin, and Aip1 Occurs in Bursts and Is Inhibited by Barbed-end Cappers The Harvard community has made this article openly available. Please share how this access benefits
Depolymerization of Actin Filaments by Profilin: Effects of. Profilin on Capping Protein Function
 JBC Papers in Press. Published on May 2, 2003 as Manuscript M302796200 Depolymerization of Actin Filaments by Profilin: Effects of Profilin on Capping Protein Function Michael R. Bubb 1,2 *, Elena G. Yarmola
JBC Papers in Press. Published on May 2, 2003 as Manuscript M302796200 Depolymerization of Actin Filaments by Profilin: Effects of Profilin on Capping Protein Function Michael R. Bubb 1,2 *, Elena G. Yarmola
Conformational changes in subdomain- of G-actin upon polymerization into F-actin and upon binding myosin subfragment-l
 Volume 316, number 2, 186190 FEBS 12036 0 1993 Federation of European Biochemical Societies 00145793/93/$6.00 January 1993 Conformational changes in subdomain- of G-actin upon polymerization into F-actin
Volume 316, number 2, 186190 FEBS 12036 0 1993 Federation of European Biochemical Societies 00145793/93/$6.00 January 1993 Conformational changes in subdomain- of G-actin upon polymerization into F-actin
Lysosomes and endocytic pathways 9/27/2012 Phyllis Hanson
 Lysosomes and endocytic pathways 9/27/2012 Phyllis Hanson General principles Properties of lysosomes Delivery of enzymes to lysosomes Endocytic uptake clathrin, others Endocytic pathways recycling vs.
Lysosomes and endocytic pathways 9/27/2012 Phyllis Hanson General principles Properties of lysosomes Delivery of enzymes to lysosomes Endocytic uptake clathrin, others Endocytic pathways recycling vs.
Xenopus Actin-interacting Protein 1 (XAip1) Enhances Cofilin Fragmentation of Filaments by Capping Filament Ends*
 THE JOURNAL OF BIOLOGICAL CHEMISTRY Vol. 277, No. 45, Issue of November 8, pp. 43011 43016, 2002 2002 by The American Society for Biochemistry and Molecular Biology, Inc. Printed in U.S.A. Xenopus Actin-interacting
THE JOURNAL OF BIOLOGICAL CHEMISTRY Vol. 277, No. 45, Issue of November 8, pp. 43011 43016, 2002 2002 by The American Society for Biochemistry and Molecular Biology, Inc. Printed in U.S.A. Xenopus Actin-interacting
Molecular Cell Biology 5068 In Class Exam 1 October 3, 2013
 Molecular Cell Biology 5068 In Class Exam 1 October 3, 2013 Exam Number: Please print your name: Instructions: Please write only on these pages, in the spaces allotted and not on the back. Write your number
Molecular Cell Biology 5068 In Class Exam 1 October 3, 2013 Exam Number: Please print your name: Instructions: Please write only on these pages, in the spaces allotted and not on the back. Write your number
The cytoskeleton and cell movement. (Actin microfilaments)
 The cytoskeleton and cell movement (Actin microfilaments) What is the cytoskeleton? A dynamic network of protein filaments extending throughout the cytoplasm Three types: microfilaments (actin), microtubules
The cytoskeleton and cell movement (Actin microfilaments) What is the cytoskeleton? A dynamic network of protein filaments extending throughout the cytoplasm Three types: microfilaments (actin), microtubules
Mechanisms of alternative splicing regulation
 Mechanisms of alternative splicing regulation The number of mechanisms that are known to be involved in splicing regulation approximates the number of splicing decisions that have been analyzed in detail.
Mechanisms of alternative splicing regulation The number of mechanisms that are known to be involved in splicing regulation approximates the number of splicing decisions that have been analyzed in detail.
Chapter 7: Membranes
 Chapter 7: Membranes Roles of Biological Membranes The Lipid Bilayer and the Fluid Mosaic Model Transport and Transfer Across Cell Membranes Specialized contacts (junctions) between cells What are the
Chapter 7: Membranes Roles of Biological Membranes The Lipid Bilayer and the Fluid Mosaic Model Transport and Transfer Across Cell Membranes Specialized contacts (junctions) between cells What are the
Signal Transduction Cascades
 Signal Transduction Cascades Contents of this page: Kinases & phosphatases Protein Kinase A (camp-dependent protein kinase) G-protein signal cascade Structure of G-proteins Small GTP-binding proteins,
Signal Transduction Cascades Contents of this page: Kinases & phosphatases Protein Kinase A (camp-dependent protein kinase) G-protein signal cascade Structure of G-proteins Small GTP-binding proteins,
Cell motility: the integrating role of the plasma membrane
 Eur Biophys J (2011) 40:1013 1027 DOI 10.1007/s00249-011-0741-0 REVIEW Cell motility: the integrating role of the plasma membrane Kinneret Keren Received: 23 July 2011 / Accepted: 24 July 2011 / Published
Eur Biophys J (2011) 40:1013 1027 DOI 10.1007/s00249-011-0741-0 REVIEW Cell motility: the integrating role of the plasma membrane Kinneret Keren Received: 23 July 2011 / Accepted: 24 July 2011 / Published
Cell Motility. Junfeng Ji ( 纪俊峰 ), PhD
 Cell Motility Junfeng Ji ( 纪俊峰 ), PhD Professor Laboratory of Somatic Reprogramming and Stem Cell Aging Center for Stem Cell and Tissue Engineering School of Medicine Zhejiang University Email: jijunfeng@zju.edu.cnedu
Cell Motility Junfeng Ji ( 纪俊峰 ), PhD Professor Laboratory of Somatic Reprogramming and Stem Cell Aging Center for Stem Cell and Tissue Engineering School of Medicine Zhejiang University Email: jijunfeng@zju.edu.cnedu
MOLECULAR CELL BIOLOGY
 1 Lodish Berk Kaiser Krieger scott Bretscher Ploegh Matsudaira MOLECULAR CELL BIOLOGY SEVENTH EDITION CHAPTER 13 Moving Proteins into Membranes and Organelles Copyright 2013 by W. H. Freeman and Company
1 Lodish Berk Kaiser Krieger scott Bretscher Ploegh Matsudaira MOLECULAR CELL BIOLOGY SEVENTH EDITION CHAPTER 13 Moving Proteins into Membranes and Organelles Copyright 2013 by W. H. Freeman and Company
Supplementary Table 1: Motor-clutch gene mrna expression Myosin Motors
 Supplementary Table 1: Motor-clutch gene mrna expression Myosin Motors Symbol Description Expression Rank MYL6 myosin, light chain 6, alkali, smooth muscle and non-muscle 11903 79 MYH9 myosin, heavy chain
Supplementary Table 1: Motor-clutch gene mrna expression Myosin Motors Symbol Description Expression Rank MYL6 myosin, light chain 6, alkali, smooth muscle and non-muscle 11903 79 MYH9 myosin, heavy chain
Mechanism of Actin Filament Turnover by Severing and Nucleation at Different Concentrations of ADF/Cofilin
 Molecular Cell 24, 13 23, October 6, 2006 ª2006 Elsevier Inc. DOI 10.1016/j.molcel.2006.08.006 Mechanism of Actin Filament Turnover by Severing and Nucleation at Different Concentrations of ADF/Cofilin
Molecular Cell 24, 13 23, October 6, 2006 ª2006 Elsevier Inc. DOI 10.1016/j.molcel.2006.08.006 Mechanism of Actin Filament Turnover by Severing and Nucleation at Different Concentrations of ADF/Cofilin
The Amyloid Precursor Protein Has a Flexible Transmembrane Domain and Binds Cholesterol
 The Amyloid Precursor Protein Has a Flexible Transmembrane Domain and Binds Cholesterol Science 336, 1171 (2013) Coach Prof. : Dr. Chung-I Chang Sit-in Prof.: Dr. Wei Yuan Yang Presenter: Han-Ying Wu Date:
The Amyloid Precursor Protein Has a Flexible Transmembrane Domain and Binds Cholesterol Science 336, 1171 (2013) Coach Prof. : Dr. Chung-I Chang Sit-in Prof.: Dr. Wei Yuan Yang Presenter: Han-Ying Wu Date:
Muscle Dr. Ted Milner (KIN 416)
 Muscle Dr. Ted Milner (KIN 416) Muscles are biological motors which actively generate force and produce movement through the process of contraction. The molecular mechanism responsible for muscle contraction
Muscle Dr. Ted Milner (KIN 416) Muscles are biological motors which actively generate force and produce movement through the process of contraction. The molecular mechanism responsible for muscle contraction
Effects of Second Messengers
 Effects of Second Messengers Inositol trisphosphate Diacylglycerol Opens Calcium Channels Binding to IP 3 -gated Channel Cooperative binding Activates Protein Kinase C is required Phosphorylation of many
Effects of Second Messengers Inositol trisphosphate Diacylglycerol Opens Calcium Channels Binding to IP 3 -gated Channel Cooperative binding Activates Protein Kinase C is required Phosphorylation of many
Supplementary Figures
 Supplementary Figures Supplementary Figure 1. (a) Uncropped version of Fig. 2a. RM indicates that the translation was done in the absence of rough mcirosomes. (b) LepB construct containing the GGPG-L6RL6-
Supplementary Figures Supplementary Figure 1. (a) Uncropped version of Fig. 2a. RM indicates that the translation was done in the absence of rough mcirosomes. (b) LepB construct containing the GGPG-L6RL6-
Supplementary Figure S1 Black silicon and dragonfly wing nanotopography.
 Supplementary Figure S1 Black silicon and dragonfly wing nanotopography. Representative low-magnification scanning electron micrographs of a) Black silicon (bsi) and b) Diplacodes bipunctata dragonfly
Supplementary Figure S1 Black silicon and dragonfly wing nanotopography. Representative low-magnification scanning electron micrographs of a) Black silicon (bsi) and b) Diplacodes bipunctata dragonfly
PAPER No. : 16, Bioorganic and biophysical chemistry MODULE No. : 22, Mechanism of enzyme catalyst reaction (I) Chymotrypsin
 Subject Paper No and Title 16 Bio-organic and Biophysical Module No and Title 22 Mechanism of Enzyme Catalyzed reactions I Module Tag CHE_P16_M22 Chymotrypsin TABLE OF CONTENTS 1. Learning outcomes 2.
Subject Paper No and Title 16 Bio-organic and Biophysical Module No and Title 22 Mechanism of Enzyme Catalyzed reactions I Module Tag CHE_P16_M22 Chymotrypsin TABLE OF CONTENTS 1. Learning outcomes 2.
Contraction, Locomotion (Ch 16), Shelden
 Contraction, Locomotion (Ch 16), Shelden 1 Molec Biol Cell 5 th ed. Supplemental Videos Actin-dependent ATP hydrolysis by myosin generates motile force The Myosin ATPase activity cycle 2 ATP binding releases
Contraction, Locomotion (Ch 16), Shelden 1 Molec Biol Cell 5 th ed. Supplemental Videos Actin-dependent ATP hydrolysis by myosin generates motile force The Myosin ATPase activity cycle 2 ATP binding releases
CELLS. Cells. Basic unit of life (except virus)
 Basic unit of life (except virus) CELLS Prokaryotic, w/o nucleus, bacteria Eukaryotic, w/ nucleus Various cell types specialized for particular function. Differentiation. Over 200 human cell types 56%
Basic unit of life (except virus) CELLS Prokaryotic, w/o nucleus, bacteria Eukaryotic, w/ nucleus Various cell types specialized for particular function. Differentiation. Over 200 human cell types 56%
Lecture Series 5 Cellular Membranes
 Lecture Series 5 Cellular Membranes Cellular Membranes A. Membrane Composition and Structure B. Animal Cell Adhesion C. Passive Processes of Membrane Transport D. Active Transport E. Endocytosis and Exocytosis
Lecture Series 5 Cellular Membranes Cellular Membranes A. Membrane Composition and Structure B. Animal Cell Adhesion C. Passive Processes of Membrane Transport D. Active Transport E. Endocytosis and Exocytosis
CELL BIOLOGY - CLUTCH CH CELL JUNCTIONS AND TISSUES.
 !! www.clutchprep.com CONCEPT: CELL-CELL ADHESION Cells must be able to bind and interact with nearby cells in order to have functional and strong tissues Cells can in two main ways - Homophilic interactions
!! www.clutchprep.com CONCEPT: CELL-CELL ADHESION Cells must be able to bind and interact with nearby cells in order to have functional and strong tissues Cells can in two main ways - Homophilic interactions
A. Membrane Composition and Structure. B. Animal Cell Adhesion. C. Passive Processes of Membrane Transport. D. Active Transport
 Cellular Membranes A. Membrane Composition and Structure Lecture Series 5 Cellular Membranes B. Animal Cell Adhesion E. Endocytosis and Exocytosis A. Membrane Composition and Structure The Fluid Mosaic
Cellular Membranes A. Membrane Composition and Structure Lecture Series 5 Cellular Membranes B. Animal Cell Adhesion E. Endocytosis and Exocytosis A. Membrane Composition and Structure The Fluid Mosaic
Actin binding proteins
 Cell Adhesion & Migration ISSN: 1933-6918 (Print) 1933-6926 (Online) Journal homepage: http://www.tandfonline.com/loi/kcam20 Actin binding proteins Stephane R. Gross To cite this article: Stephane R. Gross
Cell Adhesion & Migration ISSN: 1933-6918 (Print) 1933-6926 (Online) Journal homepage: http://www.tandfonline.com/loi/kcam20 Actin binding proteins Stephane R. Gross To cite this article: Stephane R. Gross
Lecture Series 4 Cellular Membranes
 Lecture Series 4 Cellular Membranes Reading Assignments Read Chapter 11 Membrane Structure Review Chapter 12 Membrane Transport Review Chapter 15 regarding Endocytosis and Exocytosis Read Chapter 20 (Cell
Lecture Series 4 Cellular Membranes Reading Assignments Read Chapter 11 Membrane Structure Review Chapter 12 Membrane Transport Review Chapter 15 regarding Endocytosis and Exocytosis Read Chapter 20 (Cell
Statin inhibition of HMG-CoA reductase: a 3-dimensional view
 Atherosclerosis Supplements 4 (2003) 3/8 www.elsevier.com/locate/atherosclerosis Statin inhibition of HMG-CoA reductase: a 3-dimensional view Eva Istvan * Department of Molecular Microbiology, Howard Hughes
Atherosclerosis Supplements 4 (2003) 3/8 www.elsevier.com/locate/atherosclerosis Statin inhibition of HMG-CoA reductase: a 3-dimensional view Eva Istvan * Department of Molecular Microbiology, Howard Hughes
Identification of cofilin, coronin, Rac and capz in actin tails using a Listeria affinity approach
 Journal of Cell Science 111, 2877-2884 (1998) Printed in Great Britain The Company of Biologists Limited 1998 JCS9821 2877 Identification of cofilin, coronin, Rac and capz in actin tails using a Listeria
Journal of Cell Science 111, 2877-2884 (1998) Printed in Great Britain The Company of Biologists Limited 1998 JCS9821 2877 Identification of cofilin, coronin, Rac and capz in actin tails using a Listeria
Lecture Series 4 Cellular Membranes. Reading Assignments. Selective and Semi-permeable Barriers
 Lecture Series 4 Cellular Membranes Reading Assignments Read Chapter 11 Membrane Structure Review Chapter 12 Membrane Transport Review Chapter 15 regarding Endocytosis and Exocytosis Read Chapter 20 (Cell
Lecture Series 4 Cellular Membranes Reading Assignments Read Chapter 11 Membrane Structure Review Chapter 12 Membrane Transport Review Chapter 15 regarding Endocytosis and Exocytosis Read Chapter 20 (Cell
Cell Biology Lecture 9 Notes Basic Principles of cell signaling and GPCR system
 Cell Biology Lecture 9 Notes Basic Principles of cell signaling and GPCR system Basic Elements of cell signaling: Signal or signaling molecule (ligand, first messenger) o Small molecules (epinephrine,
Cell Biology Lecture 9 Notes Basic Principles of cell signaling and GPCR system Basic Elements of cell signaling: Signal or signaling molecule (ligand, first messenger) o Small molecules (epinephrine,
2, 6, 3, 7, 8, 9, 10, 5
 A novel regulatory element in cardiac muscle troponin T. Striated muscle contraction is regulated by the position of tropomyosin on actin. That position, in turn, is regulated by the troponin complex (troponin
A novel regulatory element in cardiac muscle troponin T. Striated muscle contraction is regulated by the position of tropomyosin on actin. That position, in turn, is regulated by the troponin complex (troponin
L I F E S C I E N C E S
 1a L I F E S C I E N C E S 5 -UUA AUA UUC GAA AGC UGC AUC GAA AAC UGU GAA UCA-3 5 -TTA ATA TTC GAA AGC TGC ATC GAA AAC TGT GAA TCA-3 3 -AAT TAT AAG CTT TCG ACG TAG CTT TTG ACA CTT AGT-5 OCTOBER 31, 2006
1a L I F E S C I E N C E S 5 -UUA AUA UUC GAA AGC UGC AUC GAA AAC UGU GAA UCA-3 5 -TTA ATA TTC GAA AGC TGC ATC GAA AAC TGT GAA TCA-3 3 -AAT TAT AAG CTT TCG ACG TAG CTT TTG ACA CTT AGT-5 OCTOBER 31, 2006
Lecture 15. Signal Transduction Pathways - Introduction
 Lecture 15 Signal Transduction Pathways - Introduction So far.. Regulation of mrna synthesis Regulation of rrna synthesis Regulation of trna & 5S rrna synthesis Regulation of gene expression by signals
Lecture 15 Signal Transduction Pathways - Introduction So far.. Regulation of mrna synthesis Regulation of rrna synthesis Regulation of trna & 5S rrna synthesis Regulation of gene expression by signals
Enzymes Part III: regulation II. Dr. Mamoun Ahram Summer, 2017
 Enzymes Part III: regulation II Dr. Mamoun Ahram Summer, 2017 Advantage This is a major mechanism for rapid and transient regulation of enzyme activity. A most common mechanism is enzyme phosphorylation
Enzymes Part III: regulation II Dr. Mamoun Ahram Summer, 2017 Advantage This is a major mechanism for rapid and transient regulation of enzyme activity. A most common mechanism is enzyme phosphorylation
WIP: more than a WASp-interacting protein
 Review WIP: more than a WASp-interacting protein Sophia Fried, 1 Omri Matalon, 1 Elad Noy, 1 and Mira Barda-Saad 2 The Mina and Everard Goodman Faculty of Life Sciences, Bar-Ilan University, Ramat-Gan,
Review WIP: more than a WASp-interacting protein Sophia Fried, 1 Omri Matalon, 1 Elad Noy, 1 and Mira Barda-Saad 2 The Mina and Everard Goodman Faculty of Life Sciences, Bar-Ilan University, Ramat-Gan,
ATP-independent reversal of a membrane protein aggregate by a chloroplast SRP
 ATP-independent reversal of a membrane protein aggregate by a chloroplast SRP Peera Jaru-Ampornpan 1, Kuang Shen 1,3, Vinh Q. Lam 1,3, Mona Ali 2, Sebastian Doniach 2, Tony Z. Jia 1, Shu-ou Shan 1 Supplementary
ATP-independent reversal of a membrane protein aggregate by a chloroplast SRP Peera Jaru-Ampornpan 1, Kuang Shen 1,3, Vinh Q. Lam 1,3, Mona Ali 2, Sebastian Doniach 2, Tony Z. Jia 1, Shu-ou Shan 1 Supplementary
Actin cytoskeletal dynamics in T lymphocyte activation and migration
 Actin cytoskeletal dynamics in T lymphocyte activation and migration Yvonne Samstag, Sybille M. Eibert, Martin Klemke, and Guido H. Wabnitz Institute for Immunology, Ruprecht-Karls-University, Heidelberg,
Actin cytoskeletal dynamics in T lymphocyte activation and migration Yvonne Samstag, Sybille M. Eibert, Martin Klemke, and Guido H. Wabnitz Institute for Immunology, Ruprecht-Karls-University, Heidelberg,
Lecture Series 4 Cellular Membranes
 Lecture Series 4 Cellular Membranes Reading Assignments Read Chapter 11 Membrane Structure Review Chapter 21 pages 709-717 717 (Animal( Cell Adhesion) Review Chapter 12 Membrane Transport Review Chapter
Lecture Series 4 Cellular Membranes Reading Assignments Read Chapter 11 Membrane Structure Review Chapter 21 pages 709-717 717 (Animal( Cell Adhesion) Review Chapter 12 Membrane Transport Review Chapter
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION doi:10.1038/nature22394 Supplementary Table 1 Observed intermolecular interactions within the GLP-1:GLP-1R TMD interface. Superscripts refer to the Wootten residue numbering system
SUPPLEMENTARY INFORMATION doi:10.1038/nature22394 Supplementary Table 1 Observed intermolecular interactions within the GLP-1:GLP-1R TMD interface. Superscripts refer to the Wootten residue numbering system
Molecular Graphics Perspective of Protein Structure and Function
 Molecular Graphics Perspective of Protein Structure and Function VMD Highlights > 20,000 registered Users Platforms: Unix (16 builds) Windows MacOS X Display of large biomolecules and simulation trajectories
Molecular Graphics Perspective of Protein Structure and Function VMD Highlights > 20,000 registered Users Platforms: Unix (16 builds) Windows MacOS X Display of large biomolecules and simulation trajectories
Rama Abbady. Odai Bani-Monia. Diala Abu-Hassan
 5 Rama Abbady Odai Bani-Monia Diala Abu-Hassan Lipid Rafts Lipid rafts are aggregates (accumulations) of sphingolipids. They re semisolid clusters (10-200 nm) of cholesterol and sphingolipids (sphingomyelin
5 Rama Abbady Odai Bani-Monia Diala Abu-Hassan Lipid Rafts Lipid rafts are aggregates (accumulations) of sphingolipids. They re semisolid clusters (10-200 nm) of cholesterol and sphingolipids (sphingomyelin
Integrin structure and signaling
 Integrin structure and signaling Ramray Bhat ramray@iisc.ac.in 08022932764 History -1 Richard Hynes begins his stint in Michael Stoker s laboratory to look for cell surface changes in PyMT virus transformed
Integrin structure and signaling Ramray Bhat ramray@iisc.ac.in 08022932764 History -1 Richard Hynes begins his stint in Michael Stoker s laboratory to look for cell surface changes in PyMT virus transformed
Mechanism of Muscular Contraction
 ~ Sorin2:er Jack A. Rail Mechanism of Muscular Contraction Contents 1 Setting the Stage: Myosin, Actin, Actomyosin and ATP... 1.1 Introduction... 1 1.2 Muscle Structure as Observed by Nineteenth Century
~ Sorin2:er Jack A. Rail Mechanism of Muscular Contraction Contents 1 Setting the Stage: Myosin, Actin, Actomyosin and ATP... 1.1 Introduction... 1 1.2 Muscle Structure as Observed by Nineteenth Century
PHSI3009 Frontiers in Cellular Physiology 2017
 Overview of PHSI3009 L2 Cell membrane and Principles of cell communication L3 Signalling via G protein-coupled receptor L4 Calcium Signalling L5 Signalling via Growth Factors L6 Signalling via small G-protein
Overview of PHSI3009 L2 Cell membrane and Principles of cell communication L3 Signalling via G protein-coupled receptor L4 Calcium Signalling L5 Signalling via Growth Factors L6 Signalling via small G-protein
SUPPLEMENTARY INFORMATION
 doi:10.1038/nature19102 Supplementary Discussion Benzothiazepine Binding in Ca V Ab Diltiazem and other benzothiazepines inhibit Ca V 1.2 channels in a frequency-dependent manner consistent with pore block
doi:10.1038/nature19102 Supplementary Discussion Benzothiazepine Binding in Ca V Ab Diltiazem and other benzothiazepines inhibit Ca V 1.2 channels in a frequency-dependent manner consistent with pore block
Biochemical Reaction Rate Constant Value Source a s -1 [1]
![Biochemical Reaction Rate Constant Value Source a s -1 [1] Biochemical Reaction Rate Constant Value Source a s -1 [1]](/thumbs/95/123499384.jpg) S1 Table. Kinetic Rate Constants. Biochemical Reaction Rate Constant Value Source a Hydrolysis of C3(H 2 O) k "( ) 8.3 10-7 s -1 [1] Association of Factor B to C3(H 2 O) C3(H 2 O)B Association of Factor
S1 Table. Kinetic Rate Constants. Biochemical Reaction Rate Constant Value Source a Hydrolysis of C3(H 2 O) k "( ) 8.3 10-7 s -1 [1] Association of Factor B to C3(H 2 O) C3(H 2 O)B Association of Factor
October 26, Lecture Readings. Vesicular Trafficking, Secretory Pathway, HIV Assembly and Exit from Cell
 October 26, 2006 Vesicular Trafficking, Secretory Pathway, HIV Assembly and Exit from Cell 1. Secretory pathway a. Formation of coated vesicles b. SNAREs and vesicle targeting 2. Membrane fusion a. SNAREs
October 26, 2006 Vesicular Trafficking, Secretory Pathway, HIV Assembly and Exit from Cell 1. Secretory pathway a. Formation of coated vesicles b. SNAREs and vesicle targeting 2. Membrane fusion a. SNAREs
REGULATION OF ENZYME ACTIVITY. Medical Biochemistry, Lecture 25
 REGULATION OF ENZYME ACTIVITY Medical Biochemistry, Lecture 25 Lecture 25, Outline General properties of enzyme regulation Regulation of enzyme concentrations Allosteric enzymes and feedback inhibition
REGULATION OF ENZYME ACTIVITY Medical Biochemistry, Lecture 25 Lecture 25, Outline General properties of enzyme regulation Regulation of enzyme concentrations Allosteric enzymes and feedback inhibition
I. Fluid Mosaic Model A. Biological membranes are lipid bilayers with associated proteins
 Lecture 6: Membranes and Cell Transport Biological Membranes I. Fluid Mosaic Model A. Biological membranes are lipid bilayers with associated proteins 1. Characteristics a. Phospholipids form bilayers
Lecture 6: Membranes and Cell Transport Biological Membranes I. Fluid Mosaic Model A. Biological membranes are lipid bilayers with associated proteins 1. Characteristics a. Phospholipids form bilayers
Lane: 1. Spectra BR protein ladder 2. PFD 3. TERM 4. 3-way connector 5. 2-way connector
 kda 1 2 3 4 5 26 14 1 7 Lane: 1. Spectra BR protein ladder 2. PFD 3. TERM 4. 3-way connector 5. 2-way connector 5 4 35 25 15 1 Supplementary Figure 1. SDS-PAGE of acterially expressed and purified proteins.
kda 1 2 3 4 5 26 14 1 7 Lane: 1. Spectra BR protein ladder 2. PFD 3. TERM 4. 3-way connector 5. 2-way connector 5 4 35 25 15 1 Supplementary Figure 1. SDS-PAGE of acterially expressed and purified proteins.
BIOL 4374/BCHS 4313 Cell Biology Exam #2 March 22, 2001
 BIOL 4374/BCHS 4313 Cell Biology Exam #2 March 22, 2001 SS# Name This exam is worth a total of 100 points. The number of points each question is worth is shown in parentheses. Good luck! 1. (2) In the
BIOL 4374/BCHS 4313 Cell Biology Exam #2 March 22, 2001 SS# Name This exam is worth a total of 100 points. The number of points each question is worth is shown in parentheses. Good luck! 1. (2) In the
Antigen Recognition by T cells
 Antigen Recognition by T cells TCR only recognize foreign Ags displayed on cell surface These Ags can derive from pathogens, which replicate within cells or from pathogens or their products that cells
Antigen Recognition by T cells TCR only recognize foreign Ags displayed on cell surface These Ags can derive from pathogens, which replicate within cells or from pathogens or their products that cells
Supplementary Material
 Supplementary Material Supplementary Text Text S1. Time distributions of the high FRET efficiency at different concentrations of EF-G.GTP From Fig. 1, the model of ribosomal translocation at non-saturating
Supplementary Material Supplementary Text Text S1. Time distributions of the high FRET efficiency at different concentrations of EF-G.GTP From Fig. 1, the model of ribosomal translocation at non-saturating
Signal-Transduction Cascades - 2. The Phosphoinositide Cascade
 Signal-Transduction Cascades - 2 The Phosphoinositide Cascade Calcium ion as a second messenger Tyrosine kinase and receptor dimerization scribd.com Faisal Khatib JU The Phosphoinositide Cascade Used by
Signal-Transduction Cascades - 2 The Phosphoinositide Cascade Calcium ion as a second messenger Tyrosine kinase and receptor dimerization scribd.com Faisal Khatib JU The Phosphoinositide Cascade Used by
Identification of influential proteins in the classical retinoic acid signaling pathway
 Ghaffari and Petzold Theoretical Biology and Medical Modelling (2018) 15:16 https://doi.org/10.1186/s12976-018-0088-7 RESEARCH Open Access Identification of influential proteins in the classical retinoic
Ghaffari and Petzold Theoretical Biology and Medical Modelling (2018) 15:16 https://doi.org/10.1186/s12976-018-0088-7 RESEARCH Open Access Identification of influential proteins in the classical retinoic
Reactive Oxygen Species Regulate Protrusion Efficiency by Controlling Actin Dynamics
 Reactive Oxygen Species Regulate Protrusion Efficiency by Controlling Actin Dynamics Nicolas Taulet, Violaine D. Delorme-Walker, Céline DerMardirossian* Department of Immunology and Microbial Science,
Reactive Oxygen Species Regulate Protrusion Efficiency by Controlling Actin Dynamics Nicolas Taulet, Violaine D. Delorme-Walker, Céline DerMardirossian* Department of Immunology and Microbial Science,
Skeletal Muscle : Structure
 1 Skeletal Muscle : Structure Dr.Viral I. Champaneri, MD Assistant Professor Department of Physiology 2 Learning objectives 1. Gross anatomy of the skeletal muscle 2. Myofilaments & their molecular structure
1 Skeletal Muscle : Structure Dr.Viral I. Champaneri, MD Assistant Professor Department of Physiology 2 Learning objectives 1. Gross anatomy of the skeletal muscle 2. Myofilaments & their molecular structure
SUPPLEMENTARY INFORMATION
 doi:10.1038/nature14008 Supplementary Figure 1. Sequence alignment of A/little yellow-shouldered bat/guatemala/060/2010 (H17N10) polymerase with that of human strain A/Victoria/3/75(H3N2). The secondary
doi:10.1038/nature14008 Supplementary Figure 1. Sequence alignment of A/little yellow-shouldered bat/guatemala/060/2010 (H17N10) polymerase with that of human strain A/Victoria/3/75(H3N2). The secondary
Chemical Surface Transformation 1
 Chemical Surface Transformation 1 Chemical reactions at Si H surfaces (inorganic and organic) can generate very thin films (sub nm thickness up to µm): inorganic layer formation by: thermal conversion:
Chemical Surface Transformation 1 Chemical reactions at Si H surfaces (inorganic and organic) can generate very thin films (sub nm thickness up to µm): inorganic layer formation by: thermal conversion:
Tala Saleh. Ahmad Attari. Mamoun Ahram
 23 Tala Saleh Ahmad Attari Minna Mushtaha Mamoun Ahram In the previous lecture, we discussed the mechanisms of regulating enzymes through inhibitors. Now, we will start this lecture by discussing regulation
23 Tala Saleh Ahmad Attari Minna Mushtaha Mamoun Ahram In the previous lecture, we discussed the mechanisms of regulating enzymes through inhibitors. Now, we will start this lecture by discussing regulation
Biological motion. Motorproteins. Common features of motorproteins. Molecular mechanisms of biological motion. Structural homology
 Biological motion Molecular motion Molecular mechanisms of biological motion. Cellular motion Zsolt Mártonfalvi Bacterial flagellum Body motion Keratocyte moving on surface Motorproteins Mechanoenzymes
Biological motion Molecular motion Molecular mechanisms of biological motion. Cellular motion Zsolt Mártonfalvi Bacterial flagellum Body motion Keratocyte moving on surface Motorproteins Mechanoenzymes
UNIVERSITY OF GUELPH CHEM 4540 ENZYMOLOGY Winter 2005 Quiz #2: March 24, 2005, 11:30 12:50 Instructor: Prof R. Merrill ANSWERS
 UNIVERSITY F GUELPH CHEM 4540 ENZYMLGY Winter 2005 Quiz #2: March 24, 2005, 11:30 12:50 Instructor: Prof R. Merrill ANSWERS Instructions: Time allowed = 80 minutes. Total marks = 30. This quiz represents
UNIVERSITY F GUELPH CHEM 4540 ENZYMLGY Winter 2005 Quiz #2: March 24, 2005, 11:30 12:50 Instructor: Prof R. Merrill ANSWERS Instructions: Time allowed = 80 minutes. Total marks = 30. This quiz represents
Biological Membranes. Lipid Membranes. Bilayer Permeability. Common Features of Biological Membranes. A highly selective permeability barrier
 Biological Membranes Structure Function Composition Physicochemical properties Self-assembly Molecular models Lipid Membranes Receptors, detecting the signals from outside: Light Odorant Taste Chemicals
Biological Membranes Structure Function Composition Physicochemical properties Self-assembly Molecular models Lipid Membranes Receptors, detecting the signals from outside: Light Odorant Taste Chemicals
ARP2/3 COMPLEX HAS A NEUROPROTECTIVE ROLE AND IS REQUIRED FOR MATURE DENDRITIC SPINE HEAD MORPHOLOGY
 ARP2/3 COMPLEX HAS A NEUROPROTECTIVE ROLE AND IS REQUIRED FOR MATURE DENDRITIC SPINE HEAD MORPHOLOGY A DISSERTATION SUBMITTED TO THE FACULTY OF THE GRADUATE SCHOOL OF THE UNIVERSITY OF MINNESOTA BY MARCELA
ARP2/3 COMPLEX HAS A NEUROPROTECTIVE ROLE AND IS REQUIRED FOR MATURE DENDRITIC SPINE HEAD MORPHOLOGY A DISSERTATION SUBMITTED TO THE FACULTY OF THE GRADUATE SCHOOL OF THE UNIVERSITY OF MINNESOTA BY MARCELA
1. Identify and characterize interesting phenomena! 2. Characterization should stimulate some questions/models! 3. Combine biochemistry and genetics
 1. Identify and characterize interesting phenomena! 2. Characterization should stimulate some questions/models! 3. Combine biochemistry and genetics to gain mechanistic insight! 4. Return to step 2, as
1. Identify and characterize interesting phenomena! 2. Characterization should stimulate some questions/models! 3. Combine biochemistry and genetics to gain mechanistic insight! 4. Return to step 2, as
Assembly of the Contractile System In Nonmuscle Cells. Tanya SVITKINA
 Assembly of the Contractile System In Nonmuscle Cells Tanya SVITKINA ACTIN MHC phosphorylation, S100A4 (Mts1) binding MYOSIN II QUASI-SARCOMERIC ORGANIZATION OF STRESS FIBERS Myosin and actin REF-52 fibroblasts
Assembly of the Contractile System In Nonmuscle Cells Tanya SVITKINA ACTIN MHC phosphorylation, S100A4 (Mts1) binding MYOSIN II QUASI-SARCOMERIC ORGANIZATION OF STRESS FIBERS Myosin and actin REF-52 fibroblasts
The actin cytoskeleton in cancer cell motility
 Clin Exp Metastasis (2009) 26:273 287 DOI 10.1007/s10585-008-9174-2 RESEARCH PAPER The actin cytoskeleton in cancer cell motility Michael F. Olson Æ Erik Sahai Received: 19 January 2008 / Accepted: 25
Clin Exp Metastasis (2009) 26:273 287 DOI 10.1007/s10585-008-9174-2 RESEARCH PAPER The actin cytoskeleton in cancer cell motility Michael F. Olson Æ Erik Sahai Received: 19 January 2008 / Accepted: 25
Lipids and Membranes
 Lipids and Membranes Presented by Dr. Mohammad Saadeh The requirements for the Pharmaceutical Biochemistry I Philadelphia University Faculty of pharmacy Biological membranes are composed of lipid bilayers
Lipids and Membranes Presented by Dr. Mohammad Saadeh The requirements for the Pharmaceutical Biochemistry I Philadelphia University Faculty of pharmacy Biological membranes are composed of lipid bilayers
Regulation of the Actin Cytoskeleton-Plasma Membrane Interplay by Phosphoinositides
 Physiol Rev 90: 259 289, 2010; doi:10.1152/physrev.00036.2009. Regulation of the Actin Cytoskeleton-Plasma Membrane Interplay by Phosphoinositides JUHA SAARIKANGAS, HONGXIA ZHAO, AND PEKKA LAPPALAINEN
Physiol Rev 90: 259 289, 2010; doi:10.1152/physrev.00036.2009. Regulation of the Actin Cytoskeleton-Plasma Membrane Interplay by Phosphoinositides JUHA SAARIKANGAS, HONGXIA ZHAO, AND PEKKA LAPPALAINEN
Lipids and Membranes
 Lipids and Membranes Presented by Dr. Mohammad Saadeh The requirements for the Pharmaceutical Biochemistry I Philadelphia University Faculty of pharmacy Membrane transport D. Endocytosis and Exocytosis
Lipids and Membranes Presented by Dr. Mohammad Saadeh The requirements for the Pharmaceutical Biochemistry I Philadelphia University Faculty of pharmacy Membrane transport D. Endocytosis and Exocytosis
Globular proteins Proteins globular fibrous
 Globular proteins Globular proteins Proteins are biochemical compounds consisting of one or more polypeptides typically folded into a globular or fibrous form in a biologically functional way. Globular
Globular proteins Globular proteins Proteins are biochemical compounds consisting of one or more polypeptides typically folded into a globular or fibrous form in a biologically functional way. Globular
Study of different types of ubiquitination
 Study of different types of ubiquitination Rudi Beyaert (rudi.beyaert@irc.vib-ugent.be) VIB UGent Center for Inflammation Research Ghent, Belgium VIB Training Novel Proteomics Tools: Identifying PTMs October
Study of different types of ubiquitination Rudi Beyaert (rudi.beyaert@irc.vib-ugent.be) VIB UGent Center for Inflammation Research Ghent, Belgium VIB Training Novel Proteomics Tools: Identifying PTMs October
Enzyme-coupled Receptors. Cell-surface receptors 1. Ion-channel-coupled receptors 2. G-protein-coupled receptors 3. Enzyme-coupled receptors
 Enzyme-coupled Receptors Cell-surface receptors 1. Ion-channel-coupled receptors 2. G-protein-coupled receptors 3. Enzyme-coupled receptors Cell-surface receptors allow a flow of ions across the plasma
Enzyme-coupled Receptors Cell-surface receptors 1. Ion-channel-coupled receptors 2. G-protein-coupled receptors 3. Enzyme-coupled receptors Cell-surface receptors allow a flow of ions across the plasma
Lecture 15. Membrane Proteins I
 Lecture 15 Membrane Proteins I Introduction What are membrane proteins and where do they exist? Proteins consist of three main classes which are classified as globular, fibrous and membrane proteins. A
Lecture 15 Membrane Proteins I Introduction What are membrane proteins and where do they exist? Proteins consist of three main classes which are classified as globular, fibrous and membrane proteins. A
BIOLOGY 103 Spring 2001 MIDTERM LAB SECTION
 BIOLOGY 103 Spring 2001 MIDTERM NAME KEY LAB SECTION ID# (last four digits of SS#) STUDENT PLEASE READ. Do not put yourself at a disadvantage by revealing the content of this exam to your classmates. Your
BIOLOGY 103 Spring 2001 MIDTERM NAME KEY LAB SECTION ID# (last four digits of SS#) STUDENT PLEASE READ. Do not put yourself at a disadvantage by revealing the content of this exam to your classmates. Your
PRC2 crystal clear. Matthieu Schapira
 PRC2 crystal clear Matthieu Schapira Epigenetic mechanisms control the combination of genes that are switched on and off in any given cell. In turn, this combination, called the transcriptional program,
PRC2 crystal clear Matthieu Schapira Epigenetic mechanisms control the combination of genes that are switched on and off in any given cell. In turn, this combination, called the transcriptional program,
7.61 Eukaryotic Cell Biology: Principles and Practice Aims/Focus of the Course. Cell Biology in Context. M. Krieger and R.
 7.61 Eukaryotic Cell Biology: Principles and Practice 2004 M. Krieger and R. Hynes Aims/Focus of the Course Principles and Approaches of Modern Cell Biology Molecules Cells Tissues Organisms How do we
7.61 Eukaryotic Cell Biology: Principles and Practice 2004 M. Krieger and R. Hynes Aims/Focus of the Course Principles and Approaches of Modern Cell Biology Molecules Cells Tissues Organisms How do we
