Original Article Detection of immune suppressive neutrophils in peripheral blood samples of cancer patients
|
|
|
- Lorin Pierce
- 5 years ago
- Views:
Transcription
1 Am J Blood Res 2013;3(3): /ISSN: /AJBR Original Article Detection of immune suppressive neutrophils in peripheral blood samples of cancer patients Suyang Hao, Mary Andersen, Hongbo Yu Department of Pathology, UMass Memorial Medical Center and University of Massachusetts Medical School, Worcester, MA, USA Received April 9, 2013; Accepted June 30, 2013; Epub August 19, 2013; Published August 30, 2013 Abstract: Neutrophils provide first-line defense against infections and are potent effectors in innate and adaptive immunity. Recently neutrophils have been shown to play important roles in multiple antitumor reactions. A subset of mature neutrophils in human systemic inflammation has been identified as a unique circulating population of myeloid cells, which is capable of inhibiting T cell responses. These neutrophils show unique immunophenotype (CD11c bright/cd62l dim/cd11b bright/cd16 bright). This study reports detection of mature neutrophils with similar immunophenotype in the peripheral blood samples of cancer patients using flow cytometry analysis. This population of neutrophils is not detected in peripheral blood samples of normal controls. Thus this finding suggests the involvement of mature neutrophils in antitumor immunity. Keywords: Neutrophils, immunosuppressive, cancer, CD62L Introduction Neutrophils are the most abundant leukocytes in the circulating blood. They provide the firstline defense against infection and are potent effectors of inflammation [1]. In addition, neutrophils also are engaged in a complex crosstalk with immune and endothelial cells that bridges innate and adaptive immunity [1, 2]. Recent studies have suggested that neutrophils are active in immunosurveillance against several tumors [3-6]. Even though many aspects of their biology have been thoroughly investigated, a deep insight into the biological role of neutrophils in antitumor reactions is still needed. Neutrophils could be a perfect weapon for the suppression of tumor growth. Recent studies have identified myeloid-derived suppressor cells (MDSC) that are potent suppressors of tumor immunity and therefore a significant impediment to cancer immunotherapy [7]. MDSC accumulate in the blood, lymph nodes, and bone marrow and at tumor sites in most patients and experimental animals with cancer and inhibit both adaptive and innate immunity. MDSC are a heterogeneous family of myeloid cells and they are able to suppress T cell activation. In cancer patients, MDSC are typically CD11b+ CD33+ CD34+ CD14- HLA- DR- and can vary in their expression of CD15 and other markers [8-11]. The variation in MDSC phenotype is consistent with the concept that MDSC are a diverse family of cells that are in various intermediate stages of myeloid cell differentiation [7]. Pillay et al identified a subset of mature neutrophils in human systemic inflammation as a unique circulating population of myeloid cells, which is capable of inhibiting T cell responses [12]. These neutrophils show unique immunophenotype (CD11c bright/cd62l dim/cd11b bright/cd16 bright). These cells are observed in humans in vivo during acute systemic inflammation induced by endotoxin challenge or by severe injury. Local release of hydrogen peroxide from the neutrophils into the immunological synapse between the neutrophils and T cells mediated the suppression of T cell proliferation and required neutrophils expression of the integrin Mac-1. The goal of our study is to evaluate whether the similar subset of neutrophils can be detected in peripheral blood samples of cancer patients, in comparison to normal non-cancer patients.
2 Figure 1. Identification of suppressive neutrophils in peripheral blood samples of cancer patients. A. Neutrophils were selected and gated based on CD45 and side scatter. B. A representative histogram from a normal control. C. A representative histogram from a cancer patient. D. A representative histogram from a non-cancer surgery patient. Materials and methods Peripheral blood samples were retrieved from the hematology laboratory at the Department of Pathology, UMass Memorial Medical Center, Worcester, MA. This study was approved by the UMass Memorial Medical Center Institutional Review Board. Samples and patients Peripheral blood samples were retrieved from 31 cancer patients prior to surgery, radiation, or any systemic chemotherapy. 31 samples from normal healthy patients with routine physical examination were served as controls. There were also 10 samples from patients undergoing surgeries for benign diseases. Flow cytometry analysis For all samples, five-color flow cytometry was performed on Beckman Coulter FC-500 instruments (Beckman Coulter, Inc, Miami, FL) using commercially available reagents. All monoclonal antibodies used in the study were from Beckman Coulter, Inc. (Miami, FL) and they included monoclonal antibodies directed 240 Am J Blood Res 2013;3(3):
3 Figure 2. Comparison of gated neutrophil percentages (A), total white blood cell percentages (B), and absolute counts (C) of suppressive neutrophils in normal controls (represented by ), cancer patients (represented by ), and non-cancer surgery patients (represented by ). against CD45 (clone J. 33, phycoerythrin-texas Red (ECD) conjugated), CD62L (clone DREG56, fluorescein isothiocyanate (FITC) conjugated), CD16 (clone 3GB, phycoerythrin-cyanine7 (PC7) conjugated), CD11b (clone Bear1, phycoerythrin-cyanine5 (PC5) conjugated), and CD11c (clone BU15, phycoerythrin (PE) conjugated). 100 µl aliquot of whole blood was incubated with the appropriate amount of titrated antibodies for 10 minutes at room temperature in the dark. Then 2.0 ml of VersaLyse solution was added to each tube and then incubated in the dark at room temperature for 15 minutes to lyse the red blood cells. Then the tubes were centrifuge at 400 x g (1,400 rpm) for 5 minutes at room temperature and carefully poured off supernatant. Then 1.0 ml of Sheath Fluid was added to the tubes and centrifuged at 400 x g (1,400 rpm) for 5 minutes at room temperature and carefully pour off supernatant. Each cell pellet was then resuspended in 500 µl of Fixative Solution. Store the samples in the refrigerator until they are analyzed on the flow cytometer. The instrument alignment, sensitivities, and spectral compensation were verified daily by standards, calibrators, procedural controls, and normal peripheral blood samples prior to processing of patient samples. For all cases, at least 15,000 cells acquired for each case and analyzed by gating on neutrophils. 241 Am J Blood Res 2013;3(3):
4 Table 1. Cancer patient characteristics in the study Number of patients (n) 31 Mean age in years (range) 62 (46-88) Gender Male 4 Female 27 Cancer diagnosis Colon 1 Breast 5 Pancreatic 2 Lung 1 Melanoma 1 Ovarian 8 Uterine 9 Brain 3 Bladder 1 AJCC clinical cancer stage I/II 18 III/IV 10 Brain cancer 3 Cell counts Total leukocyte counts were counted in whole blood using Beckman Coulter LH750 (Beckman Coulter, Inc, Miami, FL). Using the percentages found by flow cytometry and the absolute leukocyte count, the absolute numbers of circulating neutrophil subsets were calculated. Statistical analysis Data was summarized using means and standard deviations. Student t tests were applied to test for differences between groups (e.g. cancer patients vs. normal controls; non-cancer surgery patients vs. normal controls; cancer patients vs. non-cancer surgery patients). Statistical significance for all comparisons was based on two-sided comparisons with p < Confidence interval is 95%. Data are presented as mean ± standard error of the mean (SEM). Results Normal control samples In order to obtain an accurate and unbiased determination of both percentage and absolute number of subsets of neutrophils in patients, whole blood flow cytometry analysis was performed using a combination of CD45/CD11b/ CD11c/CD16/CD62L. 31 normal control samples were analyzed. The neutrophils were gated on based on CD45 intensity and side scatter (Figure 1A). Then the analysis reveals that most of the neutrophils express bright expression of CD11b, CD11c, CD16 and CD62L. The population of cells with dim expression for CD16 represents eosinophils and it is not included in further measurements. The immune suppressive neutrophils exhibit bright expression of CD11b, CD11c, CD16, but dim expression of CD62L as documented in a previous paper by Pillay et al [12]. This subset of neutrophils suppresses lymphocytes activation. A representative histogram from a normal control is shown in Figure 1B. In this example, this subset of neutrophils comprises 0.3% of gated neutrophils and 0.1% of total white blood cells. The absolute count of this subset of neutrophils is 8/mm 3. In 31 normal control patient samples, the percentages of this subset of neutrophils in gated neutrophils range from 0.3% to 5.1% with mean of 1.5% (standard error of the mean (SEM): 0.17%) (Figure 2A). The percentages of this subset of neutrophils in total white blood cells range from 0.05% to 1.2% with mean of 0.4% (SEM: 0.05%) (Figure 2B). The absolute counts of this subset of neutrophils range from 4/mm 3 to 75/mm 3 with mean of 28.1/mm 3 (SEM: 3.4/mm 3 ) (Figure 2C). Identification of immune suppressive neutrophils in cancer patients Peripheral blood samples were retrieved from 31 cancer patients (mean age 62; range 46-88) with newly diagnosed and histologically confirmed solid malignancies. In accordance with American Joint Committee on Cancer (AJCC) Cancer Staging Manual, seventh edition (2010), there are 18 patients with stage I/II disease and 10 patients with stage III/IV disease. In addition, there are 3 patients with glioblastoma multiforme (GBM) in the brain, which do not have TNM designation. Blood samples collected from patients with cancers were obtained prior to surgery, radiation, or any systemic chemotherapy. Patient characteristics are detailed in Table 1. In cancer patients, freshly drawn whole blood samples were analyzed by flow cytometry analysis using a combination of CD45/CD11b/ CD11c/CD16/CD62L. The neutrophils were 242 Am J Blood Res 2013;3(3):
5 gated on based on CD45 intensity and side scatter. Then the analysis reveals that some neutrophils express bright expression of CD11b, CD11c, CD16 and CD62L. But there is increased number of immune suppressive neutrophils, which is characterized by bright expression of CD11b, CD11c, CD16, but dim expression of CD62L. A representative example from a cancer patient is showed in Figure 1C. In this example, this subset of neutrophils comprises 87.7% of gated neutrophils and 10.5% of total white blood cells. The absolute count of this subset of neutrophils is 891/mm 3. In 31 cancer patient samples, the percentages of this subset of neutrophils in gated neutrophils range from 0.5% to 87.7% with mean of 26.8% (SEM: 3.95%) (Figure 2A). The percentages of this subset of neutrophils in total white blood cells range from 0.4% to 49.9% with mean of 10.3% (SEM: 2.36%) (Figure 2B). The absolute counts of this subset of neutrophils range from 49/mm 3 to 9189/mm 3 with mean of 1312/mm 3 (SEM: 328.3/mm 3 ) (Figure 2C). This finding is statistically significant when compared to normal controls in regards to neutrophil percentage (p < ) (Figure 2). However, the cancer stages do not seem to correlate with the number of immune suppressive neutrophils detected in peripheral blood samples. Identification of immune suppressive neutrophils in non-cancer surgery patients There were also 10 samples from patients undergoing surgeries for benign diseases, which include uterine leiomyomata, ovarian cystic benign neoplasms, and brain abscess. There is also increased number of immune suppressive neutrophils, which is characterized by bright expression of CD11b, CD11c, CD16, but dim expression of CD62L. A representative example is demonstrated in Figure 1D. In this example, this subset of neutrophils comprises 7.4% of gated neutrophils and 2.6% of total white blood cells. The absolute count of this subset of neutrophils is 73/mm 3. In 10 noncancer surgery patient samples, the percentage of this subset of neutrophils in gated neutrophils range from 1.2% to 39.1% with mean of 18.0% (SEM: 3.92%) (Figure 2A). The percentages of this subset of neutrophils in total white blood cells range from 0.1% to 20.4% with mean of 10.3% (SEM: 2.36%) (Figure 2B). The absolute counts of this subset of neutrophils range from 17/mm 3 to 2299/mm 3 with mean of 559.8/mm 3 (SEM: 222.1/mm 3 ) (Figure 2C). This finding is statistically significant when compared to normal controls (Figure 2). However, when compared to cancer patients, the difference is not statistically significant. Discussion Pillay et al has reported the identification of a mature neutrophil subset with immune suppressive function, which suppresses T cell activation in a Mac-1-dependent manner [12]. In their study, this subset of neutrophils exhibit unique immunphenotype of CD11c bright/ CD62L dim/cd11b bright/cd16 bright. They have shown that this unique subset of neutrophils was detected in patients suffering systemic inflammation. They also demonstrated that these neutrophils displayed regulatory properties. In various murine models of cancer, sepsis, and transplantation, neutrophils were found to be part of MDSCs. This heterogeneous group of cells, suppressing lymphocyte activation, consists mainly of immature neutrophils and monocytes. Here our study detects the same neutrophil subset in peripheral blood samples from cancer patients. Our work is the first to our knowledge to show a distinct neutrophil phenotype CD11c bright/cd62l dim/cd11b bright/cd16 bright in cancer patients. The percentage and absolute number of this subset of neutrophils is statistically significantly increased in cancer patients when compared to normal controls. There is also increased number of neutrophils in non-cancer surgery patients when compared to normal controls. The percentages and absolute numbers of neutrophils seem to be increased in cancer patients when compared to non-cancer surgery patients, but they are not statistically significant. MDSCs producing high arginase have been found in renal cell carcinoma patients [9]. In human with metastatic cancer disease, arginase I-mediated suppression of lymphocytes was reported [9, 13]. Arginase I depletes the microenvironment of L-arginine, which is essential for various T cell functions. Despite the fact that neutrophil-t cell interactions have been described in lymphoid organs in mice [14, 15], it is still unknown whether human neutrophils 243 Am J Blood Res 2013;3(3):
6 enter lymphoid organs and come into close contact with T cells. In many acute and chronic inflammatory conditions, however, neutrophils and T cells are present in the tissues. As neutrophilic inflammation frequently results in tissue damage, neutrophil-induced inhibition of T cell proliferation might be essential to limit T cell activation and, thereby, maintain tolerance in these inflammatory conditions. The exact role for neutrophils in immune tolerance in humans remains to be examined; however, it is clear that neutrophil association with solid tumors is detrimental for disease outcome, possibly through suppressing immunogenic antitumor responses. Our data also corroborated with previous work by Diaz-Montero CM et al, which reported that circulating MDSC levels in cancer patients of all stages are significantly higher when compared to normal volunteers [16]. They found that percentages and absolute numbers of MDSC increased with clinical cancer stage and extensive tumor burden. Their study provided evidence that MDSC is a clinically important mediator of tumor-mediated immune suppression in patients with solid tumors. Abnormal accumulation of MDSC is an important mechanism of T cell unresponsiveness in cancer patients. Future cancer vaccine trials should therefore take into account circulating MDSC in patients as their presence may significantly decrease their clinical effectiveness. Pharmacologic strategies to reduce levels of MDSC in patients should be tested in future clinical trials. However, it is unknown whether reduction of MDSC in either number or partial reversal of their immunosuppressive properties is possible in cancer patients and whether this would significantly delay tumor progression. This may be a clinically viable strategy to significantly improve the efficacy of immunotherapy in cancer patients. In conclusion, we provide evidence of identification of a unique subset of neutrophils in peripheral blood samples from cancer patients, which have been shown to have immune suppressive capacity. Understanding of the functions of this neutrophil subset in human cancers might provide novel therapeutic strategies to target immune suppression in inflammation and cancer. Disclosure of conflict of interest None. Address correspondence to: Dr. Hongbo Yu, Department of Pathology, UMass Memorial Medical Center, Three Biotech, One Innovation Drive, Worcester, MA 01605, USA. hongbo.yu@ umassmemorial.org References [1] Di Carlo E, Forni G, Lollini P, Colombo MP, Modesti A, Musiani P. The intriguing role of polymorphonuclear neutrophils in antitumor reactions. Blood 2001; 97: [2] Colombo MP, Modesti A, Parmiani G, Forni G. Local cytokine availability elicits tumor rejection and systemic immunity through granulocyte-t-lymphocyte cross-talk. Cancer Res 1992; 52: [3] Lichtenstein A: Granulocytes as possible effectors of tumor immunity. Human Cancer Immunology. In: Oettgen HF, editor. Philadelphia, PA: Saunders 1990; pp: [4] Midorikawa Y, Yamashita T, Sendo F. Modulation of the immune response to transplanted tumors in rats by selective depletion of neutrophils in vivo using a monoclonal antibody: abrogation of specific transplantation resistance to chemical carcinogen-induced syngeneic tumors by selective depletion of neutrophils in vivo. Cancer Res 1990; 50: [5] Matsumoto Y, Saiki I, Murata J, Okuyama H, Tamura M, Azuma I. Recombinant human granulocyte colony-stimulating factor inhibits the metastasis of hematogenous and non-hematogenous tumors in mice. Int J Cancer 1991; 49: [6] Colombo MP, Ferrari G, Stoppacciaro A, Parenza M, Rodolfo M, Mavilio F, Parmiani G. Granulocyte colony-stimulating factor gene transfer suppresses tumorigenicity of a murine adenocarcinoma in vivo. J Exp Med 1991; 173: [7] Ostrand-Rosenberg S and Sinha P. Myeloidderived suppressor cells: linking inflammation and cancer. J Immunol 2009; 182: [8] Filipazzi P, Valenti R, Huber V, Pilla L, Canese P, Iero M, Castelli C, Mariani L, Parmiani G, Rivoltini L. Identification of a new subset of myeloid suppressor cells in peripheral blood of melanoma patients with modulation by a granulocyte-macrophage colony-stimulation factorbased antitumor vaccine. J Clin Oncol 2007; 25: Am J Blood Res 2013;3(3):
7 [9] Zea AH, Rodriguez PC, Atkins MB, Hernandez C, Signoretti S, Zabaleta J, McDermott D, Quiceno D, Youmans A, O Neill A, Mier J, Ochoa AC. Arginase-producing myeloid suppressor cells in renal cell carcinoma patient: a mechanism of tumor evasion. Cancer Res 2005; 65: [10] Mirza N, Fishman M, Fricke I, Dunn M, Neuger AM, Frost TJ, Lush RM, Antonia S, Gabrilovich DI. All-trans-retinoic acid improves differentiation of myeloid cells and immune response in cancer patients. Cancer Res 2006; 66: [11] Srivastava MK, Bosch JJ, Thompson JA, Ksander BR, Edelman MJ, Ostrand-Rosenberg S. Lung cancer patients CD4+ T cells are activated in vitro by MHC II cell-based vaccines despite the presence of myeloid-derived suppressor cells. Cancer Immunol Immunother 2008; 57: [12] Pillay J, Kamp VM, van Hoffen E, Visser T, Tak T, Lammers J, Ulfman LH, Leenen LP, Pickkers P, Koenderman L. A subset of neutrophils in human systemic inflammation inhibits T cell responses through Mac-1. J Clin Invest 2012; 122: [13] Rodriguez PC, Ernstoff MS, Hernandez C, Atkins M, Zabaleta J, Sierra R, Ochoa AC. Arginase I-producing myeloid-derived suppressor cells in renal cell carcinoma are a subpopulation of activated granulocytes. Cancer Res 2009; 69: [14] Abadie V, Badell E, Douillard P, Ensergueix D, Leenen PJ, Tanguy M, Fiette L, Saeland S, Gicquel B, Winter N. Neutrophils rapidly migrate via lymphatics after Mycobacterium bovis BCG intradermal vaccination and shuttle live bacilli to the draining lymph nodes. Blood 2005; 106: [15] Maletto BA, Ropolo AS, Alignani DO, Liscovsky MV, Ranocchia RP, Moron VG, Pistoresi-Palencia MC. Presence of neutrophil-bearing antigen in lymphoid organs of immune mice. Blood 2006; 108: [16] Diaz-Montero CM, Salem ML, Nishimura MI, Garrett-Mayer E, Cole DJ, Montero AJ. Increased circulating myeloid-derived suppressor cells correlate with clinical cancer stage, metastatic tumor burden, and doxorubicin-cyclophosphamide chemotherapy. Cancer Immunol Immunother 2009; 58: Am J Blood Res 2013;3(3):
Myeloid-derived suppressor cell
 JJIAO 30 4 : 271 278, 2012 Myeloid-derived suppressor cell 1 1 Myeloid-derived suppressor cells MDSC MDSC T MDSC MDSC MDSC MDSC MDSC myeloid-derived suppressor cells MDSC MDSC, myeloid-derived suppressor
JJIAO 30 4 : 271 278, 2012 Myeloid-derived suppressor cell 1 1 Myeloid-derived suppressor cells MDSC MDSC T MDSC MDSC MDSC MDSC MDSC myeloid-derived suppressor cells MDSC MDSC, myeloid-derived suppressor
Naive, memory and regulatory T lymphocytes populations analysis
 Naive, memory and regulatory T lymphocytes populations analysis Jaen Olivier, PhD ojaen@beckmancoulter.com Cellular Analysis application specialist Beckman Coulter France Introduction Flow cytometric analysis
Naive, memory and regulatory T lymphocytes populations analysis Jaen Olivier, PhD ojaen@beckmancoulter.com Cellular Analysis application specialist Beckman Coulter France Introduction Flow cytometric analysis
The Journal of International Medical Research 2011; 39:
 The Journal of International Medical Research 211; 39: 1381 1391 Increased Circulating Immunosuppressive CD14 + HLA-DR /low Cells Correlate with Clinical Cancer Stage and Pathological Grade in Patients
The Journal of International Medical Research 211; 39: 1381 1391 Increased Circulating Immunosuppressive CD14 + HLA-DR /low Cells Correlate with Clinical Cancer Stage and Pathological Grade in Patients
COURSE: Medical Microbiology, PAMB 650/720 - Fall 2008 Lecture 16
 COURSE: Medical Microbiology, PAMB 650/720 - Fall 2008 Lecture 16 Tumor Immunology M. Nagarkatti Teaching Objectives: Introduction to Cancer Immunology Know the antigens expressed by cancer cells Understand
COURSE: Medical Microbiology, PAMB 650/720 - Fall 2008 Lecture 16 Tumor Immunology M. Nagarkatti Teaching Objectives: Introduction to Cancer Immunology Know the antigens expressed by cancer cells Understand
CB-1158 Inhibits the Immuno-oncology Target Arginase and Causes an Immune Mediated Anti-Tumor Response
 CB-1158 Inhibits the Immuno-oncology Target Arginase and Causes an Immune Mediated Anti-Tumor Response Francesco Parlati, Ph.D. Calithera Biosciences South San Francisco, CA Arginine Depletion by Arginase
CB-1158 Inhibits the Immuno-oncology Target Arginase and Causes an Immune Mediated Anti-Tumor Response Francesco Parlati, Ph.D. Calithera Biosciences South San Francisco, CA Arginine Depletion by Arginase
Letter to Editor Tissue micro arrays for immunohistochemical detection of inflammatory infiltrates in renal cell carcinoma
 Int J Clin Exp Med 2014;7(4):1175-1179 www.ijcem.com /ISSN:1940-5901/IJCEM0000102 Letter to Editor Tissue micro arrays for immunohistochemical detection of inflammatory infiltrates in renal cell carcinoma
Int J Clin Exp Med 2014;7(4):1175-1179 www.ijcem.com /ISSN:1940-5901/IJCEM0000102 Letter to Editor Tissue micro arrays for immunohistochemical detection of inflammatory infiltrates in renal cell carcinoma
New evidences. Biomarkers to explore immunoparalysis: what future for immunostimulation? B. François CHU Limoges (France)
 New evidences Biomarkers to explore immunoparalysis: what future for immunostimulation? B. François CHU Limoges (France) Background Hotchkiss, NEJM 2003 New understanding of response in sepsis PICS Persistent
New evidences Biomarkers to explore immunoparalysis: what future for immunostimulation? B. François CHU Limoges (France) Background Hotchkiss, NEJM 2003 New understanding of response in sepsis PICS Persistent
Application Information Bulletin: Human NK Cells Phenotypic characterizing of human Natural Killer (NK) cell populations in peripheral blood
 Application Information Bulletin: Human NK Cells Phenotypic characterizing of human Natural Killer (NK) cell populations in peripheral blood Christopher A Fraker, Ph.D., University of Miami - Miami, Florida
Application Information Bulletin: Human NK Cells Phenotypic characterizing of human Natural Killer (NK) cell populations in peripheral blood Christopher A Fraker, Ph.D., University of Miami - Miami, Florida
The Immune System. A macrophage. ! Functions of the Immune System. ! Types of Immune Responses. ! Organization of the Immune System
 The Immune System! Functions of the Immune System! Types of Immune Responses! Organization of the Immune System! Innate Defense Mechanisms! Acquired Defense Mechanisms! Applied Immunology A macrophage
The Immune System! Functions of the Immune System! Types of Immune Responses! Organization of the Immune System! Innate Defense Mechanisms! Acquired Defense Mechanisms! Applied Immunology A macrophage
General Overview of Immunology. Kimberly S. Schluns, Ph.D. Associate Professor Department of Immunology UT MD Anderson Cancer Center
 General Overview of Immunology Kimberly S. Schluns, Ph.D. Associate Professor Department of Immunology UT MD Anderson Cancer Center Objectives Describe differences between innate and adaptive immune responses
General Overview of Immunology Kimberly S. Schluns, Ph.D. Associate Professor Department of Immunology UT MD Anderson Cancer Center Objectives Describe differences between innate and adaptive immune responses
Reprogramming Tumor Associated Dendritic Cells for Immunotherapy
 Reprogramming Tumor Associated Dendritic Cells for Immunotherapy Edgar Engleman, M.D. Professor of Pathology and Medicine Stanford University Disclosures: Founder of Dendreon, a biotechnology company that
Reprogramming Tumor Associated Dendritic Cells for Immunotherapy Edgar Engleman, M.D. Professor of Pathology and Medicine Stanford University Disclosures: Founder of Dendreon, a biotechnology company that
Tumor Immunology. Wirsma Arif Harahap Surgical Oncology Consultant
 Tumor Immunology Wirsma Arif Harahap Surgical Oncology Consultant 1) Immune responses that develop to cancer cells 2) Escape of cancer cells 3) Therapies: clinical and experimental Cancer cells can be
Tumor Immunology Wirsma Arif Harahap Surgical Oncology Consultant 1) Immune responses that develop to cancer cells 2) Escape of cancer cells 3) Therapies: clinical and experimental Cancer cells can be
Immunology Lecture 4. Clinical Relevance of the Immune System
 Immunology Lecture 4 The Well Patient: How innate and adaptive immune responses maintain health - 13, pg 169-181, 191-195. Immune Deficiency - 15 Autoimmunity - 16 Transplantation - 17, pg 260-270 Tumor
Immunology Lecture 4 The Well Patient: How innate and adaptive immune responses maintain health - 13, pg 169-181, 191-195. Immune Deficiency - 15 Autoimmunity - 16 Transplantation - 17, pg 260-270 Tumor
Allergy and Immunology Review Corner: Chapter 19 of Immunology IV: Clinical Applications in Health and Disease, by Joseph A. Bellanti, MD.
 Allergy and Immunology Review Corner: Chapter 19 of Immunology IV: Clinical Applications in Health and Disease, by Joseph A. Bellanti, MD. Chapter 19: Tolerance, Autoimmunity, and Autoinflammation Prepared
Allergy and Immunology Review Corner: Chapter 19 of Immunology IV: Clinical Applications in Health and Disease, by Joseph A. Bellanti, MD. Chapter 19: Tolerance, Autoimmunity, and Autoinflammation Prepared
Manipulating the Tumor Environment
 Manipulating the Tumor Environment Vincenzo Bronte Verona University Hospital vincenzo.bronte@univr.it Escape from immune control can be viewed as one of the «Hallmarks of Cancer» D. Hanahan and R. A.
Manipulating the Tumor Environment Vincenzo Bronte Verona University Hospital vincenzo.bronte@univr.it Escape from immune control can be viewed as one of the «Hallmarks of Cancer» D. Hanahan and R. A.
Novel RCC Targets from Immuno-Oncology and Antibody-Drug Conjugates
 Novel RCC Targets from Immuno-Oncology and Antibody-Drug Conjugates Christopher Turner, MD Vice President, Clinical Science 04 November 2016 Uveal Melanoma Celldex Pipeline CANDIDATE INDICATION Preclinical
Novel RCC Targets from Immuno-Oncology and Antibody-Drug Conjugates Christopher Turner, MD Vice President, Clinical Science 04 November 2016 Uveal Melanoma Celldex Pipeline CANDIDATE INDICATION Preclinical
Advances in Cancer Immunotherapy
 Advances in Cancer Immunotherapy Immunology 101 for the Non-Immunologist Arnold H. Zea, PhD azea@lsuhsc.edu Disclosures No relevant financial relationships to disclose This presentation does not contain
Advances in Cancer Immunotherapy Immunology 101 for the Non-Immunologist Arnold H. Zea, PhD azea@lsuhsc.edu Disclosures No relevant financial relationships to disclose This presentation does not contain
Phagocytosis of FITC labelled opsonized and non-opsonized E. coli bacteria by monocytes and granulocytes in a whole blood assay
 Phagocytosis of FITC labelled opsonized and non-opsonized E. coli bacteria by monocytes and granulocytes in a whole blood assay APPLICATION NOTE Author: Andreas Spittler, MD, Associate Professor for Pathophysiology
Phagocytosis of FITC labelled opsonized and non-opsonized E. coli bacteria by monocytes and granulocytes in a whole blood assay APPLICATION NOTE Author: Andreas Spittler, MD, Associate Professor for Pathophysiology
1. Background and Significance Currently there are no efficacious therapies for renal cell carcinoma (RCC). Despite recent
 1. Background and Significance Currently there are no efficacious therapies for renal cell carcinoma (RCC). Despite recent advances in development of protective immune-based therapies for RCC, the prognosis
1. Background and Significance Currently there are no efficacious therapies for renal cell carcinoma (RCC). Despite recent advances in development of protective immune-based therapies for RCC, the prognosis
Copyright. Tocagen Inc. Lead Product Candidate: Toca 511 & Toca FC Preclinical Overview
 Lead Product Candidate: Toca 511 & Toca FC Preclinical Overview Toca 511, delivers CD prodrug activator gene selectively to cancer cells Regulatory genes Structural RRV genes CD gene Regulatory genes Toca
Lead Product Candidate: Toca 511 & Toca FC Preclinical Overview Toca 511, delivers CD prodrug activator gene selectively to cancer cells Regulatory genes Structural RRV genes CD gene Regulatory genes Toca
Supplementary Figure 1. Characterization of basophils after reconstitution of SCID mice
 Supplementary figure legends Supplementary Figure 1. Characterization of after reconstitution of SCID mice with CD4 + CD62L + T cells. (A-C) SCID mice (n = 6 / group) were reconstituted with 2 x 1 6 CD4
Supplementary figure legends Supplementary Figure 1. Characterization of after reconstitution of SCID mice with CD4 + CD62L + T cells. (A-C) SCID mice (n = 6 / group) were reconstituted with 2 x 1 6 CD4
Targeting tumour associated macrophages in anti-cancer therapies. Annamaria Gal Seminar Series on Drug Discovery Budapest 5 January 2018
 Targeting tumour associated macrophages in anti-cancer therapies Annamaria Gal Seminar Series on Drug Discovery Budapest 5 January 2018 Macrophages: Professional phagocytes of the myeloid lineage APC,
Targeting tumour associated macrophages in anti-cancer therapies Annamaria Gal Seminar Series on Drug Discovery Budapest 5 January 2018 Macrophages: Professional phagocytes of the myeloid lineage APC,
DURACLONE RE THERE ARE EVENTS YOU CANNOT AFFORD TO MISS
 DURACLONE RE THERE ARE EVENTS YOU CANNOT AFFORD TO MISS Your clinical research trial companion For Reseach Use Only - Not for use in Diagnostic procedures THERE ARE EVENTS YOU CANNOT AFFORD TO MISS Rare
DURACLONE RE THERE ARE EVENTS YOU CANNOT AFFORD TO MISS Your clinical research trial companion For Reseach Use Only - Not for use in Diagnostic procedures THERE ARE EVENTS YOU CANNOT AFFORD TO MISS Rare
DCVax Knutson Northwest Biotherapeutics Inc. INT BIOTECH CONV & EXHIB 2002 Suppl June 09-12
 DCVax Knutson 407 447090 Immunotherapy for androgen Independent prostate cancer: Results of a phase I trial with a tumor vaccine of autologous dendritic cells loaded with r-psma (DCVax"' Prostate). Assikis
DCVax Knutson 407 447090 Immunotherapy for androgen Independent prostate cancer: Results of a phase I trial with a tumor vaccine of autologous dendritic cells loaded with r-psma (DCVax"' Prostate). Assikis
Engineered Immune Cells for Cancer Therapy : Current Status and Prospects
 When Engineering Meets Immunology Engineered Immune Cells for Cancer Therapy : Current Status and Prospects Yong Taik Lim, Ph.D. Nanomedical Systems Laboratory (http://www.nanomedicalsystems.org) SKKU
When Engineering Meets Immunology Engineered Immune Cells for Cancer Therapy : Current Status and Prospects Yong Taik Lim, Ph.D. Nanomedical Systems Laboratory (http://www.nanomedicalsystems.org) SKKU
Regulation of anti-tumor immunity through migration of immune cell subsets within the tumor microenvironment Thomas F. Gajewski, M.D., Ph.D.
 Regulation of anti-tumor immunity through migration of immune cell subsets within the tumor microenvironment Thomas F. Gajewski, M.D., Ph.D. Professor, Departments of Pathology and Medicine Program Leader,
Regulation of anti-tumor immunity through migration of immune cell subsets within the tumor microenvironment Thomas F. Gajewski, M.D., Ph.D. Professor, Departments of Pathology and Medicine Program Leader,
Chapter 1. Chapter 1 Concepts. MCMP422 Immunology and Biologics Immunology is important personally and professionally!
 MCMP422 Immunology and Biologics Immunology is important personally and professionally! Learn the language - use the glossary and index RNR - Reading, Note taking, Reviewing All materials in Chapters 1-3
MCMP422 Immunology and Biologics Immunology is important personally and professionally! Learn the language - use the glossary and index RNR - Reading, Note taking, Reviewing All materials in Chapters 1-3
Effector mechanisms of cell-mediated immunity: Properties of effector, memory and regulatory T cells
 ICI Basic Immunology course Effector mechanisms of cell-mediated immunity: Properties of effector, memory and regulatory T cells Abul K. Abbas, MD UCSF Stages in the development of T cell responses: induction
ICI Basic Immunology course Effector mechanisms of cell-mediated immunity: Properties of effector, memory and regulatory T cells Abul K. Abbas, MD UCSF Stages in the development of T cell responses: induction
Emerging Concepts of Cancer Immunotherapy
 Emerging Concepts of Cancer Immunotherapy Jeffrey Schlom, Ph.D. Laboratory of Tumor Immunology and Biology (LTIB) Center for Cancer Research National Cancer Institute, NIH Immune Cell Infiltrate in Primary
Emerging Concepts of Cancer Immunotherapy Jeffrey Schlom, Ph.D. Laboratory of Tumor Immunology and Biology (LTIB) Center for Cancer Research National Cancer Institute, NIH Immune Cell Infiltrate in Primary
Surgery-induced monocytic myeloid-derived suppressor cells expand regulatory T cells in lung cancer
 /, 2017, Vol. 8, (No. 10), pp: 17050-17058 Surgery-induced monocytic myeloid-derived suppressor cells expand regulatory T cells in lung cancer Jun Wang 1,*, Liu Yang 2,*, Lu Yu 3, Yi-Yin Wang 2, Rui Chen
/, 2017, Vol. 8, (No. 10), pp: 17050-17058 Surgery-induced monocytic myeloid-derived suppressor cells expand regulatory T cells in lung cancer Jun Wang 1,*, Liu Yang 2,*, Lu Yu 3, Yi-Yin Wang 2, Rui Chen
a Beckman Coulter Life Sciences: White Paper
 a Beckman Coulter Life Sciences: White Paper An 8-color DuraClone IM panel for detection of Human blood dendritic cells by flow cytometry Nathalie Dupas 1, Snehita Sattiraju 2, Neha Girish 2, Murthy Pendyala
a Beckman Coulter Life Sciences: White Paper An 8-color DuraClone IM panel for detection of Human blood dendritic cells by flow cytometry Nathalie Dupas 1, Snehita Sattiraju 2, Neha Girish 2, Murthy Pendyala
Establishing a Pure Lymphocyte Gate for Subset Analysis by Flow Cytometry
 Cytornetry (Communications in Clinical Cytometry) 26:172-177 (1996) Establishing a Pure Lymphocyte Gate for Subset nalysis by Flow Cytometry Joseph M. Homtinovich, Sara D. Sparks, and Karen P. Mann Clinical
Cytornetry (Communications in Clinical Cytometry) 26:172-177 (1996) Establishing a Pure Lymphocyte Gate for Subset nalysis by Flow Cytometry Joseph M. Homtinovich, Sara D. Sparks, and Karen P. Mann Clinical
ACTIVATION OF T LYMPHOCYTES AND CELL MEDIATED IMMUNITY
 ACTIVATION OF T LYMPHOCYTES AND CELL MEDIATED IMMUNITY The recognition of specific antigen by naïve T cell induces its own activation and effector phases. T helper cells recognize peptide antigens through
ACTIVATION OF T LYMPHOCYTES AND CELL MEDIATED IMMUNITY The recognition of specific antigen by naïve T cell induces its own activation and effector phases. T helper cells recognize peptide antigens through
BASIC MECHANISM OF TUMOR IMMUNE SUPPRESSION
 BASIC MECHANISM OF TUMOR IMMUNE SUPPRESSION Zihai Li, M.D., Ph.D. Leader, Cancer Immunology Program Hollings Cancer Center Medical University of South Carolina (MUSC) DISCLOSURE GOALS Understand that immune
BASIC MECHANISM OF TUMOR IMMUNE SUPPRESSION Zihai Li, M.D., Ph.D. Leader, Cancer Immunology Program Hollings Cancer Center Medical University of South Carolina (MUSC) DISCLOSURE GOALS Understand that immune
NKTR-255: Accessing The Immunotherapeutic Potential Of IL-15 for NK Cell Therapies
 NKTR-255: Accessing The Immunotherapeutic Potential Of IL-15 for NK Cell Therapies Saul Kivimäe Senior Scientist, Research Biology Nektar Therapeutics NK Cell-Based Cancer Immunotherapy, September 26-27,
NKTR-255: Accessing The Immunotherapeutic Potential Of IL-15 for NK Cell Therapies Saul Kivimäe Senior Scientist, Research Biology Nektar Therapeutics NK Cell-Based Cancer Immunotherapy, September 26-27,
Darwinian selection and Newtonian physics wrapped up in systems biology
 Darwinian selection and Newtonian physics wrapped up in systems biology Concept published in 1957* by Macfarland Burnet (1960 Nobel Laureate for the theory of induced immune tolerance, leading to solid
Darwinian selection and Newtonian physics wrapped up in systems biology Concept published in 1957* by Macfarland Burnet (1960 Nobel Laureate for the theory of induced immune tolerance, leading to solid
MACROPHAGE "MONOCYTES" SURFACE RECEPTORS
 LECTURE: 13 Title: MACROPHAGE "MONOCYTES" SURFACE RECEPTORS LEARNING OBJECTIVES: The student should be able to: Describe the blood monocytes (size, and shape of nucleus). Enumerate some of the monocytes
LECTURE: 13 Title: MACROPHAGE "MONOCYTES" SURFACE RECEPTORS LEARNING OBJECTIVES: The student should be able to: Describe the blood monocytes (size, and shape of nucleus). Enumerate some of the monocytes
Monocyte subsets in health and disease. Marion Frankenberger
 Monocyte subsets in health and disease Marion Frankenberger main cellular components: Leukocytes Erythrocytes Composition of whole blood Monocytes belong to the cellular components of peripheral blood
Monocyte subsets in health and disease Marion Frankenberger main cellular components: Leukocytes Erythrocytes Composition of whole blood Monocytes belong to the cellular components of peripheral blood
Human Progenitor Cell Enumeration by Flow Cytometry Practical Considerations
 Human Progenitor Cell Enumeration by Flow Cytometry Practical Considerations Bruce Briggs, M.S., S.I. (A.S.C.P.) OSU Medical Center Clinical Flow Cytometry Objectives Conceptual overview: How the Flow
Human Progenitor Cell Enumeration by Flow Cytometry Practical Considerations Bruce Briggs, M.S., S.I. (A.S.C.P.) OSU Medical Center Clinical Flow Cytometry Objectives Conceptual overview: How the Flow
Measuring Dendritic Cells
 Measuring Dendritic Cells A.D. Donnenberg, V.S. Donnenberg UNIVERSITY of PITTSBURGH CANCER INSTITUTE CCS Longbeach 10_04 Measuring DC Rare event detection The basics of DC measurement Applications Cancer
Measuring Dendritic Cells A.D. Donnenberg, V.S. Donnenberg UNIVERSITY of PITTSBURGH CANCER INSTITUTE CCS Longbeach 10_04 Measuring DC Rare event detection The basics of DC measurement Applications Cancer
Getting Beyond the Flags: Quantitative assessment of immature granulocyte (IG) populations may improve the assessment of sepsis and inflammation.
 Getting Beyond the Flags: Quantitative assessment of immature granulocyte (IG) populations may improve the assessment of sepsis and inflammation. Sysmex America White Paper One Nelson C. White Parkway,
Getting Beyond the Flags: Quantitative assessment of immature granulocyte (IG) populations may improve the assessment of sepsis and inflammation. Sysmex America White Paper One Nelson C. White Parkway,
Fluorochrome Panel 1 Panel 2 Panel 3 Panel 4 Panel 5 CTLA-4 CTLA-4 CD15 CD3 FITC. Bio) PD-1 (MIH4, BD) ICOS (C398.4A, Biolegend) PD-L1 (MIH1, BD)
 Additional file : Table S. Antibodies used for panel stain to identify peripheral immune cell subsets. Panel : PD- signaling; Panel : CD + T cells, CD + T cells, B cells; Panel : Tregs; Panel :, -T, cdc,
Additional file : Table S. Antibodies used for panel stain to identify peripheral immune cell subsets. Panel : PD- signaling; Panel : CD + T cells, CD + T cells, B cells; Panel : Tregs; Panel :, -T, cdc,
Role of microenvironment and tumor interactions in melanoma progression with special regard to the prognostic significance of immune cell infiltrate
 Role of microenvironment and tumor interactions in melanoma progression with special regard to the prognostic significance of immune cell infiltrate PhD thesis Dr. Anita Mohos Doctoral School of Pathological
Role of microenvironment and tumor interactions in melanoma progression with special regard to the prognostic significance of immune cell infiltrate PhD thesis Dr. Anita Mohos Doctoral School of Pathological
DURACLONE IF BE CERTAIN ABOUT THE RESPONSE. l res. a il n c n. For Research Use Only - Not for use in Diagnostic procedures
 DURACLONE IF earch tria l res lc a om ic il n c n nio pa Yo ur BE CERTAIN ABOUT THE RESPONSE For Research Use Only - Not for use in Diagnostic procedures BE CERTAIN ABOUT THE RESPONSE The sensitive and
DURACLONE IF earch tria l res lc a om ic il n c n nio pa Yo ur BE CERTAIN ABOUT THE RESPONSE For Research Use Only - Not for use in Diagnostic procedures BE CERTAIN ABOUT THE RESPONSE The sensitive and
Question 1. Kupffer cells, microglial cells and osteoclasts are all examples of what type of immune system cell?
 Abbas Chapter 2: Sarah Spriet February 8, 2015 Question 1. Kupffer cells, microglial cells and osteoclasts are all examples of what type of immune system cell? a. Dendritic cells b. Macrophages c. Monocytes
Abbas Chapter 2: Sarah Spriet February 8, 2015 Question 1. Kupffer cells, microglial cells and osteoclasts are all examples of what type of immune system cell? a. Dendritic cells b. Macrophages c. Monocytes
NKTR-255: Accessing IL-15 Therapeutic Potential through Robust and Sustained Engagement of Innate and Adaptive Immunity
 NKTR-255: Accessing IL-15 Therapeutic Potential through Robust and Sustained Engagement of Innate and Adaptive Immunity Peiwen Kuo Scientist, Research Biology Nektar Therapeutics August 31 st, 2018 Emerging
NKTR-255: Accessing IL-15 Therapeutic Potential through Robust and Sustained Engagement of Innate and Adaptive Immunity Peiwen Kuo Scientist, Research Biology Nektar Therapeutics August 31 st, 2018 Emerging
Effector T Cells and
 1 Effector T Cells and Cytokines Andrew Lichtman, MD PhD Brigham and Women's Hospital Harvard Medical School 2 Lecture outline Cytokines Subsets of CD4+ T cells: definitions, functions, development New
1 Effector T Cells and Cytokines Andrew Lichtman, MD PhD Brigham and Women's Hospital Harvard Medical School 2 Lecture outline Cytokines Subsets of CD4+ T cells: definitions, functions, development New
Flow cytometry leukocyte differential : a critical appraisal
 Flow cytometry leukocyte differential : a critical appraisal Francis Lacombe Flow cytometry department University Hospital of Bordeaux, Pessac, France francis.lacombe@chu-bordeaux.fr 2008 HORIBA ABX, All
Flow cytometry leukocyte differential : a critical appraisal Francis Lacombe Flow cytometry department University Hospital of Bordeaux, Pessac, France francis.lacombe@chu-bordeaux.fr 2008 HORIBA ABX, All
Initiation and regulation of effector T cell responses in the prostate
 University of Iowa Iowa Research Online Theses and Dissertations 2011 Initiation and regulation of effector T cell responses in the prostate Jessica M. Haverkamp University of Iowa Copyright 2011 Jessica
University of Iowa Iowa Research Online Theses and Dissertations 2011 Initiation and regulation of effector T cell responses in the prostate Jessica M. Haverkamp University of Iowa Copyright 2011 Jessica
Bihong Zhao, M.D, Ph.D Department of Pathology
 Bihong Zhao, M.D, Ph.D Department of Pathology 04-28-2009 Is tumor self or non-self? How are tumor antigens generated? What are they? How does immune system respond? Introduction Tumor Antigens/Categories
Bihong Zhao, M.D, Ph.D Department of Pathology 04-28-2009 Is tumor self or non-self? How are tumor antigens generated? What are they? How does immune system respond? Introduction Tumor Antigens/Categories
Ex vivo Human Antigen-specific T Cell Proliferation and Degranulation Willemijn Hobo 1, Wieger Norde 1 and Harry Dolstra 2*
 Ex vivo Human Antigen-specific T Cell Proliferation and Degranulation Willemijn Hobo 1, Wieger Norde 1 and Harry Dolstra 2* 1 Department of Laboratory Medicine - Laboratory of Hematology, Radboud University
Ex vivo Human Antigen-specific T Cell Proliferation and Degranulation Willemijn Hobo 1, Wieger Norde 1 and Harry Dolstra 2* 1 Department of Laboratory Medicine - Laboratory of Hematology, Radboud University
Nature Immunology: doi: /ni Supplementary Figure 1. Examples of staining for each antibody used for the mass cytometry analysis.
 Supplementary Figure 1 Examples of staining for each antibody used for the mass cytometry analysis. To illustrate the functionality of each antibody probe, representative plots illustrating the expected
Supplementary Figure 1 Examples of staining for each antibody used for the mass cytometry analysis. To illustrate the functionality of each antibody probe, representative plots illustrating the expected
Animal tissue-specific biomolecules influence on rats with cyclophosphamide-induced immunosuppression
 Animal tissue-specific biomolecules influence on rats with cyclophosphamide-induced immunosuppression Natural immunostimulators obtained over Sus scrofa tissue extraction with the use of water with modified
Animal tissue-specific biomolecules influence on rats with cyclophosphamide-induced immunosuppression Natural immunostimulators obtained over Sus scrofa tissue extraction with the use of water with modified
Hematopoiesis. Hematopoiesis. Hematopoiesis
 Chapter. Cells and Organs of the Immune System Hematopoiesis Hematopoiesis- formation and development of WBC and RBC bone marrow. Hematopoietic stem cell- give rise to any blood cells (constant number,
Chapter. Cells and Organs of the Immune System Hematopoiesis Hematopoiesis- formation and development of WBC and RBC bone marrow. Hematopoietic stem cell- give rise to any blood cells (constant number,
Licia Rivoltini, MD Unit of Immunotherapy of Human Tumors
 Licia Rivoltini, MD Unit of Immunotherapy of Human Tumors The complex network of anti-tumor immunity Innate immunity First line defense Tumor cell Adaptive immunity Specificity & memory Kidd et al., Nature
Licia Rivoltini, MD Unit of Immunotherapy of Human Tumors The complex network of anti-tumor immunity Innate immunity First line defense Tumor cell Adaptive immunity Specificity & memory Kidd et al., Nature
Tumor Microenvironment and Immune Suppression
 Tumor Microenvironment and Immune Suppression Hassane M. Zarour,, MD Department of Medicine, Division of Hematology-Oncology, University of Pittsburgh Cancer Institute Hallmarks of Cancer: The Next Generation
Tumor Microenvironment and Immune Suppression Hassane M. Zarour,, MD Department of Medicine, Division of Hematology-Oncology, University of Pittsburgh Cancer Institute Hallmarks of Cancer: The Next Generation
PREPARED FOR: U.S. Army Medical Research and Materiel Command Fort Detrick, Maryland Approved for public release; distribution unlimited
 AD (Leave blank) Award Number: W81XWH-07-1-0345 TITLE: Second-Generation Therapeutic DNA Lymphoma Vaccines PRINCIPAL INVESTIGATOR: Larry W. Kwak, M.D., Ph.D. CONTRACTING ORGANIZATION: University of Texas
AD (Leave blank) Award Number: W81XWH-07-1-0345 TITLE: Second-Generation Therapeutic DNA Lymphoma Vaccines PRINCIPAL INVESTIGATOR: Larry W. Kwak, M.D., Ph.D. CONTRACTING ORGANIZATION: University of Texas
Comprehensive evaluation of human immune system reconstitution in NSG. and NSG -SGM3 mouse models toward the development of a novel ONCO-HU
 Comprehensive evaluation of human immune system reconstitution in NSG and NSG -SGM3 mouse models toward the development of a novel ONCO-HU xenograft model Aaron Middlebrook, 1 Eileen Snowden, 2 Warren
Comprehensive evaluation of human immune system reconstitution in NSG and NSG -SGM3 mouse models toward the development of a novel ONCO-HU xenograft model Aaron Middlebrook, 1 Eileen Snowden, 2 Warren
Vaccine Therapy for Cancer
 Vaccine Therapy for Cancer Lawrence N Shulman, MD Chief Medical Officer Senior Vice President for Medical Affairs Chief, Division of General Oncology Dana-Farber Cancer Institute Disclosures for Lawrence
Vaccine Therapy for Cancer Lawrence N Shulman, MD Chief Medical Officer Senior Vice President for Medical Affairs Chief, Division of General Oncology Dana-Farber Cancer Institute Disclosures for Lawrence
The Immune System. Innate. Adaptive. - skin, mucosal barriers - complement - neutrophils, NK cells, mast cells, basophils, eosinophils
 Objectives - explain the rationale behind cellular adoptive immunotherapy - describe methods of improving cellular adoptive immunotherapy - identify mechanisms of tumor escape from cellular adoptive immunotherapy
Objectives - explain the rationale behind cellular adoptive immunotherapy - describe methods of improving cellular adoptive immunotherapy - identify mechanisms of tumor escape from cellular adoptive immunotherapy
Suppl Video: Tumor cells (green) and monocytes (white) are seeded on a confluent endothelial
 Supplementary Information Häuselmann et al. Monocyte induction of E-selectin-mediated endothelial activation releases VE-cadherin junctions to promote tumor cell extravasation in the metastasis cascade
Supplementary Information Häuselmann et al. Monocyte induction of E-selectin-mediated endothelial activation releases VE-cadherin junctions to promote tumor cell extravasation in the metastasis cascade
Connections Between Autoimmune Disease & Cancer
 February 6, 2018 Connections Between Autoimmune Disease & Cancer Dr. Reetesh Bose, PGY2 Supervisor: Dr. Jennifer Beecker, MD, CCFP(EM), FRCPC, DABD, FAAD University of Ottawa, Division of Dermatology Definitions
February 6, 2018 Connections Between Autoimmune Disease & Cancer Dr. Reetesh Bose, PGY2 Supervisor: Dr. Jennifer Beecker, MD, CCFP(EM), FRCPC, DABD, FAAD University of Ottawa, Division of Dermatology Definitions
Additional file 6. Summary of experiments characterizing development of adaptive immune response
 Additional file 6. Summary of experiments characterizing development of adaptive immune response Tests performed to evaluate the development of adaptive immunity in patients are summarized in Table 1.
Additional file 6. Summary of experiments characterizing development of adaptive immune response Tests performed to evaluate the development of adaptive immunity in patients are summarized in Table 1.
Shiv Pillai Ragon Institute, Massachusetts General Hospital Harvard Medical School
 CTLs, Natural Killers and NKTs 1 Shiv Pillai Ragon Institute, Massachusetts General Hospital Harvard Medical School CTL inducing tumor apoptosis 3 Lecture outline CD8 + Cytotoxic T lymphocytes (CTL) Activation/differentiation
CTLs, Natural Killers and NKTs 1 Shiv Pillai Ragon Institute, Massachusetts General Hospital Harvard Medical School CTL inducing tumor apoptosis 3 Lecture outline CD8 + Cytotoxic T lymphocytes (CTL) Activation/differentiation
Supplementary Figure 1. Double-staining immunofluorescence analysis of invasive colon and breast cancers. Specimens from invasive ductal breast
 Supplementary Figure 1. Double-staining immunofluorescence analysis of invasive colon and breast cancers. Specimens from invasive ductal breast carcinoma (a) and colon adenocarcinoma (b) were staining
Supplementary Figure 1. Double-staining immunofluorescence analysis of invasive colon and breast cancers. Specimens from invasive ductal breast carcinoma (a) and colon adenocarcinoma (b) were staining
The spectrum of flow cytometry of the bone marrow
 The spectrum of flow cytometry of the bone marrow Anna Porwit Lund University Faculty of Medicine Dept. of Clinical Sciences Div. Oncology and Pathology anna.porwit@med.lu.se Disclosure of speaker s interests
The spectrum of flow cytometry of the bone marrow Anna Porwit Lund University Faculty of Medicine Dept. of Clinical Sciences Div. Oncology and Pathology anna.porwit@med.lu.se Disclosure of speaker s interests
IMMUNOLOGY. Source, Isolate, Culture, And Analyze Immune Cells. Scientists Helping Scientists
 IMMUNOLOGY Source, Isolate, Culture, And Analyze Immune Cells Scientists Helping Scientists WWW.STEMCELL.COM TABLE OF CONTENTS Tools For Your Immunology Research 4 Primary Cells: It All Starts with The
IMMUNOLOGY Source, Isolate, Culture, And Analyze Immune Cells Scientists Helping Scientists WWW.STEMCELL.COM TABLE OF CONTENTS Tools For Your Immunology Research 4 Primary Cells: It All Starts with The
SPECIFIC AIMS. II year (1st semester)
 II year (1st semester) Scientific Field IMMUNOLOGY AND IMMUNOPATHOLOGY TUTOR ECTS MALISAN F. COORDINATOR MED/04 Immunology and Immunopathology Malisan Florence 5 MED/04 Immunology and Immunopathology Testi
II year (1st semester) Scientific Field IMMUNOLOGY AND IMMUNOPATHOLOGY TUTOR ECTS MALISAN F. COORDINATOR MED/04 Immunology and Immunopathology Malisan Florence 5 MED/04 Immunology and Immunopathology Testi
Part III Innate and Adaptive Immune Cells: General Introduction
 Innate and Adaptive Immune Cells: General Introduction Iván López-Expósito As an organ specialized in food digestion and nutrient absorption, the intestinal mucosa presents a huge surface area (almost
Innate and Adaptive Immune Cells: General Introduction Iván López-Expósito As an organ specialized in food digestion and nutrient absorption, the intestinal mucosa presents a huge surface area (almost
Posters and Presentations
 Posters and Presentations June 2017: American Society of Clinical Oncology (ASCO) Annual - Preliminary Correlative Analysis of PD-L1 expression from the SUNRISE Study. View April 2017: American Association
Posters and Presentations June 2017: American Society of Clinical Oncology (ASCO) Annual - Preliminary Correlative Analysis of PD-L1 expression from the SUNRISE Study. View April 2017: American Association
Flow Cytometric Applications for Immunotherapy of Cancer
 Flow Cytometric Applications for Immunotherapy of Cancer Bone Marrow Transplantation and Cellular Therapy Program and Divisions of Neuro-oncology and Neurosurgery University of Alabama at Birmingham Basic
Flow Cytometric Applications for Immunotherapy of Cancer Bone Marrow Transplantation and Cellular Therapy Program and Divisions of Neuro-oncology and Neurosurgery University of Alabama at Birmingham Basic
J Clin Oncol 26: by American Society of Clinical Oncology INTRODUCTION
 VOLUME 26 NUMBER 19 JULY 1 28 JOURNAL OF CLINICAL ONCOLOGY O R I G I N A L R E P O R T Phenotypic and Functional Analysis of Dendritic Cells and Clinical Outcome in Patients With High-Risk Melanoma Treated
VOLUME 26 NUMBER 19 JULY 1 28 JOURNAL OF CLINICAL ONCOLOGY O R I G I N A L R E P O R T Phenotypic and Functional Analysis of Dendritic Cells and Clinical Outcome in Patients With High-Risk Melanoma Treated
Citation Acta medica Nagasakiensia. 1991, 36
 NAOSITE: Nagasaki University's Ac Title Author(s) Induction of Killer Activity in Per Chemotherapy with Methotrexate, Vin (M-VAC) Taniguchi, Keisuke Citation Acta medica Nagasakiensia. 1991, 36 Issue Date
NAOSITE: Nagasaki University's Ac Title Author(s) Induction of Killer Activity in Per Chemotherapy with Methotrexate, Vin (M-VAC) Taniguchi, Keisuke Citation Acta medica Nagasakiensia. 1991, 36 Issue Date
A Versatile Tool to Study Immune Checkpoint Therapeutics: Syngeneic Tumor Mouse Models in vivo
 The Solution Provider for Drug Discovery in Oncology A Versatile Tool to Study Immune Checkpoint Therapeutics: Syngeneic Tumor Mouse Models in vivo Holger Weber - 1 - Drug discovery platform for oncology
The Solution Provider for Drug Discovery in Oncology A Versatile Tool to Study Immune Checkpoint Therapeutics: Syngeneic Tumor Mouse Models in vivo Holger Weber - 1 - Drug discovery platform for oncology
George A. Dominguez 1, John Roop 1, Alexander Polo 1, Anthony Campisi 1, Dmitry I. Gabrilovich 2, and Amit Kumar 1
 Using artificial intelligence to distinguish subjects with prostate cancer (PCa) from benign prostate hyperplasia (BPH) through immunophenotyping of MDSCs and lymphocyte cell populations George A. Dominguez
Using artificial intelligence to distinguish subjects with prostate cancer (PCa) from benign prostate hyperplasia (BPH) through immunophenotyping of MDSCs and lymphocyte cell populations George A. Dominguez
CANCER IMMUNOTHERAPY IN VETERINARY ONCOLOGY
 CANCER IMMUNOTHERAPY IN VETERINARY ONCOLOGY Matt Dowling, DVM, MS, Practice limited to Oncology Staff Oncologist VCA Northwest Veterinary Specialists Overview Cancer and the immune system Introduction
CANCER IMMUNOTHERAPY IN VETERINARY ONCOLOGY Matt Dowling, DVM, MS, Practice limited to Oncology Staff Oncologist VCA Northwest Veterinary Specialists Overview Cancer and the immune system Introduction
Microbiology 204: Cellular and Molecular Immunology
 Microbiology 204: Cellular and Molecular Immunology Class meets MWF 1:00-2:30PM (*exceptions: no class Fri Sept 23, Fri Oct 14, Nov 11, or Wed Nov 23) Lectures are open to auditors and will be live-streamed
Microbiology 204: Cellular and Molecular Immunology Class meets MWF 1:00-2:30PM (*exceptions: no class Fri Sept 23, Fri Oct 14, Nov 11, or Wed Nov 23) Lectures are open to auditors and will be live-streamed
VISTA, a novel immune checkpoint protein ligand that suppresses anti-tumor tumor T cell responses. Li Wang. Dartmouth Medical School
 VISTA, a novel immune checkpoint protein ligand that suppresses anti-tumor tumor T cell responses Li Wang Dartmouth Medical School The B7 Immunoglobulin Super-Family immune regulators APC T cell Co-stimulatory:
VISTA, a novel immune checkpoint protein ligand that suppresses anti-tumor tumor T cell responses Li Wang Dartmouth Medical School The B7 Immunoglobulin Super-Family immune regulators APC T cell Co-stimulatory:
CHAPTER 3 LABORATORY PROCEDURES
 CHAPTER 3 LABORATORY PROCEDURES CHAPTER 3 LABORATORY PROCEDURES 3.1 HLA TYPING Molecular HLA typing will be performed for all donor cord blood units and patients in the three reference laboratories identified
CHAPTER 3 LABORATORY PROCEDURES CHAPTER 3 LABORATORY PROCEDURES 3.1 HLA TYPING Molecular HLA typing will be performed for all donor cord blood units and patients in the three reference laboratories identified
The immune response against cancer
 The immune response against cancer Maries van den Broek Institute of Experimental Immunology vandenbroek@immunology.uzh.ch The immune system Main cells of the immune system Dendritic cell Monocyte Macrophage
The immune response against cancer Maries van den Broek Institute of Experimental Immunology vandenbroek@immunology.uzh.ch The immune system Main cells of the immune system Dendritic cell Monocyte Macrophage
Nano Immuno Chemotherapy to treat cancers
 Nano Immuno Chemotherapy to treat cancers Dr. Giovanna Lollo, MiNT, France Dr. Ilaria Marigo, IOV, Italy University of Angers MiNt: Micro et Nanomedicines Biomimetiques France Instituto Oncologico Veneto
Nano Immuno Chemotherapy to treat cancers Dr. Giovanna Lollo, MiNT, France Dr. Ilaria Marigo, IOV, Italy University of Angers MiNt: Micro et Nanomedicines Biomimetiques France Instituto Oncologico Veneto
T Lymphocyte Activation and Costimulation. FOCiS. Lecture outline
 1 T Lymphocyte Activation and Costimulation Abul K. Abbas, MD UCSF FOCiS 2 Lecture outline T cell activation Costimulation, the B7:CD28 family Inhibitory receptors of T cells Targeting costimulators for
1 T Lymphocyte Activation and Costimulation Abul K. Abbas, MD UCSF FOCiS 2 Lecture outline T cell activation Costimulation, the B7:CD28 family Inhibitory receptors of T cells Targeting costimulators for
VUmc Basispresentatie
 Clinical diagnostic cytometry Gerrit J Schuurhuis Dept of Hematology VU University Medical Center Amsterdam, Netherlands Use of immunophenotyping at diagnosis to trace residual disease after therapy 1.
Clinical diagnostic cytometry Gerrit J Schuurhuis Dept of Hematology VU University Medical Center Amsterdam, Netherlands Use of immunophenotyping at diagnosis to trace residual disease after therapy 1.
Rapid Flow Cytometry Method for Quantitation of LFA-1-Adhesive T Cells
 CLINICAL AND VACCINE IMMUNOLOGY, Mar. 2006, p. 403 408 Vol. 13, No. 3 1556-6811/06/$08.00 0 doi:10.1128/cvi.13.3.403 408.2006 Rapid Flow Cytometry Method for Quantitation of LFA-1-Adhesive T Cells Brian
CLINICAL AND VACCINE IMMUNOLOGY, Mar. 2006, p. 403 408 Vol. 13, No. 3 1556-6811/06/$08.00 0 doi:10.1128/cvi.13.3.403 408.2006 Rapid Flow Cytometry Method for Quantitation of LFA-1-Adhesive T Cells Brian
Tumor Associated Macrophages as a Novel Target for Cancer Therapy
 Tumor mass Tumor Associated Macrophage Tumor Associated Macrophages as a Novel Target for Cancer Therapy This booklet contains forward-looking statements that are based on Amgen s current expectations
Tumor mass Tumor Associated Macrophage Tumor Associated Macrophages as a Novel Target for Cancer Therapy This booklet contains forward-looking statements that are based on Amgen s current expectations
Scott Abrams, Ph.D. Professor of Oncology, x4375 Kuby Immunology SEVENTH EDITION
 Scott Abrams, Ph.D. Professor of Oncology, x4375 scott.abrams@roswellpark.org Kuby Immunology SEVENTH EDITION CHAPTER 13 Effector Responses: Cell- and Antibody-Mediated Immunity Copyright 2013 by W. H.
Scott Abrams, Ph.D. Professor of Oncology, x4375 scott.abrams@roswellpark.org Kuby Immunology SEVENTH EDITION CHAPTER 13 Effector Responses: Cell- and Antibody-Mediated Immunity Copyright 2013 by W. H.
Effective activity of cytokine-induced killer cells against autologous metastatic melanoma including cells with stemness features
 Effective activity of cytokine-induced killer cells against autologous metastatic melanoma including cells with stemness features Loretta Gammaitoni, Lidia Giraudo, Valeria Leuci, et al. Clin Cancer Res
Effective activity of cytokine-induced killer cells against autologous metastatic melanoma including cells with stemness features Loretta Gammaitoni, Lidia Giraudo, Valeria Leuci, et al. Clin Cancer Res
Immune Checkpoints in the Tumor Environment:
 Immune Checkpoints in the Tumor Environment: Novel Targets and the Clinical Promise of Combined Immunotherapies Monday, 28 October 2013 07.00 08.00 (AM) Parkside Ballroom B, Convention Centre Level 1 Phosphatidylserine
Immune Checkpoints in the Tumor Environment: Novel Targets and the Clinical Promise of Combined Immunotherapies Monday, 28 October 2013 07.00 08.00 (AM) Parkside Ballroom B, Convention Centre Level 1 Phosphatidylserine
Gladstone Institutes, University of California (UCSF), San Francisco, USA
 Fluorescence-linked Antigen Quantification (FLAQ) Assay for Fast Quantification of HIV-1 p24 Gag Marianne Gesner, Mekhala Maiti, Robert Grant and Marielle Cavrois * Gladstone Institutes, University of
Fluorescence-linked Antigen Quantification (FLAQ) Assay for Fast Quantification of HIV-1 p24 Gag Marianne Gesner, Mekhala Maiti, Robert Grant and Marielle Cavrois * Gladstone Institutes, University of
Adenovirus engineered human dendritic cell vaccine induces natural killer cell chemotaxis
 Adenovirus engineered human dendritic cell vaccine induces natural killer cell chemotaxis via CXCL8/IL 8 and CXCL10/IP 10 chemokines Lazar Vujanović, Ph.D. Research Instructor P.I. Lisa H. Butterfield,
Adenovirus engineered human dendritic cell vaccine induces natural killer cell chemotaxis via CXCL8/IL 8 and CXCL10/IP 10 chemokines Lazar Vujanović, Ph.D. Research Instructor P.I. Lisa H. Butterfield,
Technical Bulletin No. 157
 CPAL Central Pennsylvania Alliance Laboratory Technical Bulletin No. 157 January 11, 2017 Flow Cytometry Lymphocyte Subset Analysis Testing Platform Change Change Effective Date: February 6, 2017 Methods
CPAL Central Pennsylvania Alliance Laboratory Technical Bulletin No. 157 January 11, 2017 Flow Cytometry Lymphocyte Subset Analysis Testing Platform Change Change Effective Date: February 6, 2017 Methods
CONTRACTING ORGANIZATION: Johns Hopkins University School of Medicine Baltimore, MD 21205
 AD Award Number: DAMD7---7 TITLE: Development of Artificial Antigen Presenting Cells for Prostate Cancer Immunotherapy PRINCIPAL INVESTIGATOR: Jonathan P. Schneck, M.D., Ph.D. Mathias Oelke, Ph.D. CONTRACTING
AD Award Number: DAMD7---7 TITLE: Development of Artificial Antigen Presenting Cells for Prostate Cancer Immunotherapy PRINCIPAL INVESTIGATOR: Jonathan P. Schneck, M.D., Ph.D. Mathias Oelke, Ph.D. CONTRACTING
NK cell flow cytometric assay In vivo DC viability and migration assay
 NK cell flow cytometric assay 6 NK cells were purified, by negative selection with the NK Cell Isolation Kit (Miltenyi iotec), from spleen and lymph nodes of 6 RAG1KO mice, injected the day before with
NK cell flow cytometric assay 6 NK cells were purified, by negative selection with the NK Cell Isolation Kit (Miltenyi iotec), from spleen and lymph nodes of 6 RAG1KO mice, injected the day before with
In vitro human regulatory T cell expansion
 - 1 - Human CD4 + CD25 + CD127 dim/- regulatory T cell Workflow isolation, in vitro expansion and analysis In vitro human regulatory T cell expansion Introduction Regulatory T (Treg) cells are a subpopulation
- 1 - Human CD4 + CD25 + CD127 dim/- regulatory T cell Workflow isolation, in vitro expansion and analysis In vitro human regulatory T cell expansion Introduction Regulatory T (Treg) cells are a subpopulation
LAG-3: Validation Of Next Generation Checkpoint Pathways
 LAG-3: Validation Of Next Generation Checkpoint Pathways Frédéric Triebel, CO/CMO Immune Checkpoint Modulation & Combination Therapies April 13, 2016 London, UK. 1 AX:PRR; NADAQ:PBMD Notice: Forward Looking
LAG-3: Validation Of Next Generation Checkpoint Pathways Frédéric Triebel, CO/CMO Immune Checkpoint Modulation & Combination Therapies April 13, 2016 London, UK. 1 AX:PRR; NADAQ:PBMD Notice: Forward Looking
immunity defenses invertebrates vertebrates chapter 48 Animal defenses --
 defenses Animal defenses -- immunity chapter 48 invertebrates coelomocytes, amoebocytes, hemocytes sponges, cnidarians, etc. annelids basophilic amoebocytes, acidophilic granulocytes arthropod immune systems
defenses Animal defenses -- immunity chapter 48 invertebrates coelomocytes, amoebocytes, hemocytes sponges, cnidarians, etc. annelids basophilic amoebocytes, acidophilic granulocytes arthropod immune systems
Blocking antibodies and peptides. Rat anti-mouse PD-1 (29F.1A12, rat IgG2a, k), PD-
 Supplementary Methods Blocking antibodies and peptides. Rat anti-mouse PD-1 (29F.1A12, rat IgG2a, k), PD- L1 (10F.9G2, rat IgG2b, k), and PD-L2 (3.2, mouse IgG1) have been described (24). Anti-CTLA-4 (clone
Supplementary Methods Blocking antibodies and peptides. Rat anti-mouse PD-1 (29F.1A12, rat IgG2a, k), PD- L1 (10F.9G2, rat IgG2b, k), and PD-L2 (3.2, mouse IgG1) have been described (24). Anti-CTLA-4 (clone
REPROGRAMING IMMUNITY IN RENAL CELL CARCINOMA
 REPROGRAMING IMMUNITY IN RENAL CELL CARCINOMA RETHINKING TYROSINE KINASE INHIBITORS Dr. L.M. Antón Aparicio. Complejo Universitario de La Coruña INTRODUCTION Angiogenesis, which is regulated by a fine
REPROGRAMING IMMUNITY IN RENAL CELL CARCINOMA RETHINKING TYROSINE KINASE INHIBITORS Dr. L.M. Antón Aparicio. Complejo Universitario de La Coruña INTRODUCTION Angiogenesis, which is regulated by a fine
ACTIVATION AND EFFECTOR FUNCTIONS OF CELL-MEDIATED IMMUNITY AND NK CELLS. Choompone Sakonwasun, MD (Hons), FRCPT
 ACTIVATION AND EFFECTOR FUNCTIONS OF CELL-MEDIATED IMMUNITY AND NK CELLS Choompone Sakonwasun, MD (Hons), FRCPT Types of Adaptive Immunity Types of T Cell-mediated Immune Reactions CTLs = cytotoxic T lymphocytes
ACTIVATION AND EFFECTOR FUNCTIONS OF CELL-MEDIATED IMMUNITY AND NK CELLS Choompone Sakonwasun, MD (Hons), FRCPT Types of Adaptive Immunity Types of T Cell-mediated Immune Reactions CTLs = cytotoxic T lymphocytes
CD40L TCR IL-12 TLR-L
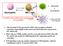 CD40L B cells plasmacells Neutrophils TCR inkt cells IL-12 Ab production Can inkt cells modulate the cytokine profile of neutrophils? TLR-L CD4+ and CD8+ T cell responses 1. The invariant TCR expressed
CD40L B cells plasmacells Neutrophils TCR inkt cells IL-12 Ab production Can inkt cells modulate the cytokine profile of neutrophils? TLR-L CD4+ and CD8+ T cell responses 1. The invariant TCR expressed
