BAK multimerization for apoptosis, but not bid binding, is inhibited by negatively charged residue in the BAK hydrophobic groove
|
|
|
- Arleen Cooper
- 6 years ago
- Views:
Transcription
1 Azad and Storey Molecular Cancer 2013, 12:65 SHORT COMMUNICATION Open Access BAK multimerization for apoptosis, but not bid binding, is inhibited by negatively charged residue in the BAK hydrophobic groove Abul Azad and Alan Storey * Abstract Background: BCL-2 family proteins BAK and BAX orchestrate outer mitochondrial membrane permeabilization (MOMP) during apoptosis by forming pores in the membrane to release apoptogenic factors that commits a cell to death. BAK and BAX therefore function as a point of no return in the apoptotic cascade. BAK activation is a multistep process involving conformational changes, mediated by BH3-only proteins or p53, which lead eventually to oligomerization and pore formation. Further, recent reports show that BAK activation is also linked to and dependent upon dephosphorylation of both tyrosine and serine residues. Findings: We hypothesized that phosphorylation of BAK at tyrosine residue 110 (Y110) was functionally important during the BAK activation process. BAK/BAX double knockout HCT116 cells expressing a phosphor-mimetic BAK mutant (BAK Y110E), showed impaired dimerization and multimerization capacity when treated with either UV irradiation or etoposide when compared to cells reconstituted to express wild-type BAK. The Y110E mutant also showed decreased release of cytochrome c from isolated mitochondria challenged with tbid protein, resulting in a failure to activate caspase 3. Interestingly, co-immunoprecipitation experiments suggest that a negative charge at this residue may be important for the recruitment of Bid to BAK, but conversely that this also impairs BAK:BAK interactions. Conclusion: These findings implicate dephosphorylation of Y110 as having an important mechanistic role in BAK activation, and underscores how post-translational modifications are intimately linked and coupled to the proteinprotein interactions required for BAK activation during apoptosis. Keywords: Apoptosis, Mitochondria, BCL-2, BAK, BH3, Cytochrome c, Tyrosine Phosphorylation, DNA damage Findings The abrogation of apoptotic responses is a common feature of almost all cancer cells. Whether or not a cell undergoes apoptosis in response to stress depends on responses following the receipt of signals generated by cellular damage. The mitochondrial apoptotic pathway is regulated by BCL-2 proteins that can be divided into different pro- and anti-apoptotic groups depending on their structure and function [1]. Pro-apoptotic BH3-only proteins, such as Bid, Bim and Noxa, are required for the activation of multi-domain BCL-2 effector proteins BAK and BAX. BAK and BAX constitute a pre-requisite * Correspondence: astorey62@gmail.com Department of Oncology, Weatherall Institute of Molecular Medicine, University of Oxford, Oxford OX3 9DS, UK gateway for mitochondrial apoptosis to proceed, indeed cells lacking both BAK and BAX fail to undergo mitochondrial apoptosis [2]. The BAK responds to a myriad of death signals and plays a key role in executing mitochondrial apoptosis, effecting MOMP through oligomerization of the protein that releases apoptogenic factors including cytochrome c, that in turn lead to downstream activation of caspase 3. BAK activation is complex, involving a number of intra-molecular conformational changes leading to dimerization followed by oligomerization [2-4]. An early event during BAK activation is a conformational change towards to N-terminus of the protein [5]. This is followed by exposure of the BAK BH3 domain that then inserts into a hydrophobic groove on another BAK molecule leading to dimer formation [6]. Resultant homodimers then can form higher order structures via interaction 2013 Azad and Storey; licensee BioMed Central Ltd. This is an Open Access article distributed under the terms of the Creative Commons Attribution License ( which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
2 Azad and Storey Molecular Cancer 2013, 12:65 Page 2 of 5 between α6:α6 helices [7]. BAK N-terminal conformational change can be brought about by one of two mechanisms: first, by the transient binding of BH3-only proteins (such as tbid) [8] to the BAK hydrophobic groove [2,9], or alternatively by the binding of p53 to residues near the BAK N-terminus [10-12]. Binding of BH3 proteins such as tbid to the BAK hydrophobic groove occurs with high affinity, but is necessarily transient as this same region of BAK is also required to nucleate BAK multimerization. Recently a model of BAK activation that tries to take into account the differing affinities of BH3 proteins for both pro- and anti-apoptotic BCL-2 proteins has been proposed [13]. In healthy, otherwise undamaged cells, BAK is present almost exclusively in a very heavily phosphorylated form. We recently demonstrated that BAK activation for apoptosis induction is closely linked to, and indeed dependent upon, specific dephosphorylation events on the protein [14]. The initial event in the BAK activation process is dephosphorylation at tyrosine 108 (Y108), an obligatory step that is required to permit conformational change by BH3 or p53 proteins [14,15]. Further, we found that a subsequent PP2A-mediated dephosphorylation of BAK at serine 117 (S117) was required both for BH3 proteins to gain access to the BAK hydrophobic groove, and permit BAK dimerization via BAK-BH3:BAK-groove interactions [16]. During our investigations into the role of phosphorylation in regulating BAK activation, we reported in mass spectrometry analysis that BAK was also phosphorylated at residue Y110 [14]. Mutation of this residue to mimic either the dephosphorylated or phosphorylated forms of the protein (BAK mutants Y110F and Y110E respectively) did not affect the ability of BAK to undergo N-terminal conformational change [14]. However, modelling using PyMol based on BAK structure 2IMS, suggested that phosphorylation at Y110 might impinge upon the ability of BH3 proteins to bind BAK and may also inhibit BAK multimerization, as the Y110 side chain, like that of S117, may occlude access to the hydrophobic groove (Figure 1A). To test this idea, we performed multimerization assays using sub-cellular fractions enriched in mitochondria from HCT116 bak / bax / cells that had been reconstituted to express either wild-type (WT) or mutant BAK proteins (HCT-BAK cells). Cross-linking reactions were performed with the sulfhydryl-to-sulfhydryl crosslinker BMH (1, 6- bismaleimidohexane, Pierce) on sub-cellular fractions enriched in mitochondria, then analysed by western blotting to detected BAK multimers, as previously described [14,16]. Following DNA damage by UV, BAK dimers, trimers and higher-order complexes were observed with WT BAK and the Y110F mutant, but multimer formation was impaired severely by the Y110E mutation (Figure 1B, upper panel). As we have noted previously, BAK crosslinking with BMH can be problematic with the formation of a faster migrating intra-molecularly cross-linked monomeric BAK protein (M x ) and generation of non-specific dimer bands without any DNA damage. A further crosslinking experiment a different sulfhydryl crosslinker, BMOE (Bismaleimidoethane), that generates only BAK dimers and at the same time minimizes the detection of both intra-molecularly cross-linked BAK monomers and non-specific dimer forms [16], supported the previous experiments with dimers being detected only when WT or the Y110F mutant was used (Figure 1B, lower panel). When the experiment was repeated using etoposide as the apoptotic inducer in place of UV, again dimers were readily detected in cells expressing the WT and Y110F proteins, but were not generated by the Y110E mutant (Figure 1C). The stronger dimer band present generated by the Y110F mutant compared to WT we suppose may be due to the mutant forming dimers more readily if the WT protein was not fully dephosphorylated following UV damage. The failure of the BAK Y110E mutant to dimerize or multimerize we reasoned may be due to interference with the binding of BH3 proteins, as observed previously for the S117E phosphor-mimic [16]. We therefore tested the ability of the BH3-activator protein tbid to release cytochrome c from mitochondrial preparations using a previously established method [16,17]. When purified tbid protein was incubated with mitochondrial preparations from cells expressing either the WT or Y110F BAK proteins, cytochrome c was readily released into the supernatant (Figure 2A). Consistent with the multimerization assays, tbid was unable to release cytochrome c from mitochondrial preparations from the BAK-Y110E mutant cells compared to the HCT116-BAK or HCT116- BAKY110F cells (Figure 2A). The small quantity of cytochrome c that was detected in the supernatant fraction derived from the Y110E mutant might be due to the mutant being very inefficient at releasing cytochrome c since the substituted amino acid may not correctly mimic a phosphor-tyrosine residue, however we have noted that processing of the samples leads to a degree of leakiness of the mitochondrial preparations where mitochondria expressing BAK mutants we find to be more fragile compared to cells expressing WT BAK, as previously noted [16]. Likewise, the Y110F mutant showed amounts of cytochrome c retained in the pellet fraction decreased somewhat when mitochondrial preparations were treated with increasing amounts of tbid, but by comparison cytochrome c was readily detected in the supernatant fraction even in the absence of tbid, and did not increase to the same extent as observed for the WT BAK protein. To investigate further whether the cytochrome c release was likely to be functionally significant and bypass potential problems of mitochondrial fragility, we performed caspase 3 activation
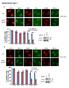
3 Azad and Storey Molecular Cancer 2013, 12:65 Page 3 of 5 Figure 1 Effects of mutation of tyrosine 110 (Y110) on the ability of BAK to form multimers following DNA damage. (A) PyMol generated model of BAK structure using PBD file 2IMS. Locations on the α4 helix of Y108 (red), together with S117 (blue) and Y110 (red) where the side chains of these residues face the hydrophobic surface groove (green), are indicated. Both the BH3 domain and BH1 encompassing α2-α5 helices (residue ) constitutes the hydrophobic surface groove. (B) BAK multimerization following DNA damage was analysed in HCT116-BAK cells as described in [16]. HCT116 DKO cells were reconstituted with the WT BAK, Y110F or Y110E BAK proteins. Mitochondria were isolated from cells expressing WT, Y110F and Y110E in ±UV 10mJ/cm 2 for 8 hrs. 100 μg of mitochondria were cross-linked with either BMH (top panel) or BMOE (bottom panel). BAK was detected by western blot using a rabbit anti BAK monoclonal (abcam, Y164). The input was the 5% of mitochondrial extract used in the cross-linking studies to ensure equal loading (middle panel). Non cross-linked BAK runs as a monomer (M) and also as an intra-molecularly linked monomer (Mx). BAK dimers (D) and trimers (T) and higher order structures are indicated. Image is representative of 3 independent experiments. (C) Similar multimerization experiment to (B) was performed with HCT116 cells expressing WT BAK, Y110F or Y110E proteins ± 50 μm etoposide treatment for 8 hrs. Mitochondrial proteins were crosslinked with BMOE as described previously and detected as above. Note that BMOE generates only dimer forms of BAK. assays using a FITC-conjugated antibody that recognizes only activated caspase 3 using FACS on intact cells. This revealed that UV irradiation resulted in an increase in caspase 3 activation in BAK WT or Y110F cells, but in marked contrast no caspase 3 activation was detected in UV-irradiated Y110E cells, findings in-line with and supporting the cytochrome c release experiment (Figure 2B). We conclude that the Y110E mutation interferes with the ability of BAK to form dimers and multimers, and that this results in the failure to release cytochrome c and activate caspase 3. In previous studies, we found that neither the BAK Y110F nor Y110E mutations had any effect on BAK being able to undergo the N-terminal conformational change associated with an early step in BAK activation, indicating that the mutations did not perturb the overall BAK structure [14]. Since either p53 or BH3 proteins bring about BAK conformational change by different routes [16], we next asked whether the BAK mutants recruited p53 to the mitochondria as efficiently as the WT protein. The p53 binding site on BAK is located near the N-terminus of the protein involving residues E24/E25 [12,16], and p53 recruitment to the mitochondria is dependent on BAK expression [10,18,19]. Following DNA damage by UV irradiation, we found that p53 was readily recruited to the mitochondria as was detected by western blot in mitochondrial extracts of cells expressing the WT or either of the two mutant BAK Y110F or Y110E (Figure 3A). As the Bid BH3 sequence interacts with the BAK hydrophobic groove, we next examined whether Bid was able to Co-IP BAK
4 Azad and Storey Molecular Cancer 2013, 12:65 Page 4 of 5 Figure 2 Cytochrome c release and caspase3 activation in BAK mutant cells. (A) Cytochrome c release assays were performed essentially as described [15,16]. Mitochondria were isolated from HCT116-WT BAK (top panel), HCT1116-Y110F (middle panel) and HCT116-Y110E (bottom panel) cells and incubated with recombinant tbid (1 ng/μl, R&D). After incubation at 37 C for 30 min, mitochondria were separated into pellet and supernatant fractions by centrifugation. Cytochrome c levels were analysed by western blotting with anti-cytochrome c antibody (BD Pharmingen) in both pellet (indicating retention in mitochondria) and supernatant (sup; released from mitochondria) fractions. In all cytochrome c release assays, an equal amount of isolated mitochondria not treated with tbid was used as input. (B) Activation of caspase 3 was analysed by FACS using an antibody that specifically recognises only cleaved (therefore active) form of caspase 3 in HCT116 cells expressing WT BAK, BAK-Y110F or BAK-Y110E mutants treated with ± 10mJ/cm 2 UV. Data are the mean percentage of 3 independent experiments, ±S.E.M. following UV irradiation. As expected, Bid was able to pull down BAK protein following UV irradiation from whole cell extracts of cells expressing either the WT or Y110F mutant BAK proteins (Figure 3B). Somewhat surprisingly, Bid was able to pull down more BAK from the Y110E cells when compared to cells expressing either WT or the Y110F mutant (Figure 3B). To explore further the role of a negative charge at Y110 on the recruitment of Bid to BAK, we performed similar IP/western blot experiments using mitochondrial enriched preparations. We found that similar levels of Bid were able to be immunoprecipitated in the three BAK cell lines expressing either WT, Y110F or Y110E mutants, irrespective of whether the cells has been treated with UV or not (Figure 3C). As before, WT or Y110F BAK proteins were only co-precipitated with Bid following UV damage, and Figure 3 Effects of BAK mutations on p53 mitochondrial translocation and ability to bind Bid. (A) Translocation of p53 into mitochondria after DNA damage. Mitochondria were isolated from cells expressing WT BAK, BAK-Y110F or BAK-Y110E mutant proteins following treatment with ± 10mJ/cm 2 UV. p53 was detected by western blot using the DO1 antibody (upper panel). Expression levels of the BAK proteins (detected as above) are also shown (lower panel). (B) Total cell extracts were prepared from HCT116 cells expressing WT BAK, Y110F or Y110E mutants following treatment with ± 10mJ/cm 2 UV with extraction buffer (20 mm Tris HCl ph7.4, 135 mm NaCl, 1.5 mm MgCl2, 1 mm EGTA, 10% glycerol, protease and phosphatase inhibitor cocktail containing 1% CHAPS). Immunoprecipitations were performed with anti Bid antibody (Cell Signalling) and BAK was detected by immunoblotting with rabbit anti-bak (Y164, abcam). The input was 5% of the extracts used in immunoprecipitation reactions. Here and in panel (C) below, the non-uv treated WT cell extract was used for the IgG control immunoprecipitation. (C) Mitochondrial-enriched sub-cellular fractions were prepared from cells expressing WT or BAK mutants ± UV treatment as outlined in Figure 1. Immunoprecipitations for Bid and detection of BAK by western blotting were as in (B) above. The input was 5% of the extracts used in immunoprecipitation reactions. that a greater amount of the Y110E BAK mutant we again found was readily pulled down by Co-IP with Bid following UV treatment (Figure 3C). However, in these mitochondrial enriched subcellular fractions we were also now able to detect BAK that had been pulled down by the Bid antibody even in the absence of UV treatment. Together these findings provides the first evidence
5 Azad and Storey Molecular Cancer 2013, 12:65 Page 5 of 5 that a negative charge located in the BAK hydrophobic surface groove may be a factor important in the recruitment of BH3 proteins such as Bid to BAK. Blockade of the BAK hydrophobic groove by phosphorylation of S117, or an S117E mutant, each impair binding of either BH3 peptides or proteins to BAK. Located further along the same helix as S117 but also facing the hydrophobic BAK surface pocket, we now identify a role for Y110 in the BAK activation process. Taken together, our results identify for the first time a novel and important bi-functional role for posttranslational modifications at Y110. Binding of BH3 proteins to BAK has been reported to occur with high affinity yet the interaction must be transient [20], as the same hydrophobic surface interface is required to nucleate BAK dimerization. We recently reported that phosphorylation of BAK at S117 blocks access of BH3 sequences of Bid and BAK to the hydrophobic pocket on BAK. In contrast, these findings suggest that while the Y110E mutant interferes with the ability to form stable BAK-BH3:BAK-groove interactions for dimerization, a negative charge at this site may also be a positive factor that is involved in the recruitment of BH3-containing proteins, such as Bid, to BAK. This implies that dephosphorylation of S117 must occur prior to dephosphorylation of Y110 in order to permit Bid recruitment, however the exact timing of these events and whether binding of other BH3-containing BCL-2 family proteins is also affected by this modification remains to be established. Findings presented here further underscore the importance of post-translational modifications in regulating different steps in BAK activation, and may reveal new avenues to either potentiate or inhibit BAK activity through modulation of the phosphorylation status of the protein. Competing interests The authors state that they have no competing interests. Authors contributions AA and AS designed the experiments; AA performed all experimental procedures; AA and AS analysed data and generated images, and prepared and approved the final version of the manuscript. Acknowledgements This work was supported by funding from Cancer Research UK. We also thank R. Youle for the gift of the HCT116 bax / bak / double knockout cells. Received: 24 January 2013 Accepted: 5 June 2013 Published: 19 June 2013 References 1. Adams JM, Cory S: The Bcl-2 apoptotic switch in cancer development and therapy. Oncogene 2007, 26: Wei MC, Zong WX, Cheng EH, Lindsten T, Panoutsakopoulou V, Ross AJ, Roth KA, MacGregor GR, Thompson CB, Korsmeyer SJ: Proapoptotic BAX and BAK: a requisite gateway to mitochondrial dysfunction and death. Science 2001, 292: van Van Delft MF, Huang DC: How the Bcl-2 family of proteins interact to regulate apoptosis. Cell Res 2006, 16: Youle RJ, Strasser A: The BCL-2 protein family: opposing activities that mediate cell death. Nat Rev Mol Cell Biol 2008, 9: Griffiths GJ, Dubrez L, Morgan CP, Jones NA, Whitehouse J, Corfe BM, Dive C, Hickman JA: Cell damage-induced conformational changes of the pro-apoptotic protein Bak in vivo precede the onset of apoptosis. JCellBiol1999, 144: Dewson G, Kratina T, Sim HW, Puthalakath H, Adams JM, Colman PM, Kluck RM: To trigger apoptosis, Bak exposes its BH3 domain and homodimerizes via BH3:groove interactions. Mol Cell 2008, 30: Dewson G, Kratina T, Czabotar P, Day CL, Adams JM, Kluck RM: Bak activation for apoptosis involves oligomerization of dimers via their alpha6 helices. Mol Cell 2009, 36: Kim H, Rafiuddin-Shah M, Tu HC, Jeffers JR, Zambetti GP, Hsieh JJ, Cheng EH: Hierarchical regulation of mitochondrion-dependent apoptosis by BCL-2 subfamilies. Nat Cell Biol 2006, 8: Korsmeyer SJ, Wei MC, Saito M, Weiler S, Oh KJ, Schlesinger PH: Proapoptotic cascade activates BID, which oligomerizes BAK or BAX into pores that result in the release of cytochrome c. Cell Death Differ 2000, 7: Leu JI, Dumont P, Hafey M, Murphy ME, George DL: Mitochondrial p53 activates Bak and causes disruption of a Bak-Mcl1 complex. Nat Cell Biol 2004, 6: Mihara M, Erster S, Zaika A, Petrenko O, Chittenden T, Pancoska P, Moll UM: p53 has a direct apoptogenic role at the mitochondria. Mol Cell 2003, 11: Pietsch EC, Perchiniak E, Canutescu AA, Wang G, Dunbrack RL, Murphy ME: Oligomerization of BAK by p53 utilizes conserved residues of the p53 DNA binding domain. J Biol Chem 2008, 283: Llambi F, Moldoveanu T, Tait SW, Bouchier-Hayes L, Temirov J, McCormick LL, Dillon CP, Green DR: A unified model of mammalian BCL-2 protein family interactions at the mitochondria. Mol Cell 2011, 44: Fox JL, Ismail F, Azad A, Ternette N, Leverrier S, Edelmann MJ, Kessler BM, Leigh IM, Jackson S, Storey A: Tyrosine dephosphorylation is required for Bak activation in apoptosis. EMBO J 2010, 29: Fox J, Azad A, Ismail F, Storey A: "Licensed to kill": tyrosine dephosphorylation and Bak activation. Cell Cycle 2011, 10: Azad A, Fox J, Leverrier S, Storey A: Blockade of the BAK hydrophobic groove by inhibitory phosphorylation regulates commitment to apoptosis. PLoS One 2012, 7:e Waterhouse NJ, Trapani JA: A new quantitative assay for cytochrome c release in apoptotic cells. Cell Death Differ 2003, 10: Moll UM, Wolff S, Speidel D, Deppert W: Transcription-independent proapoptotic functions of p53. Curr Opin Cell Biol 2005, 17: Leverrier S, Bergamaschi D, Ghali L, Ola A, Warnes G, Akgul B, Blight K, Garcia-Escudero R, Penna A, Eddaoudi A, Storey A: Role of HPV E6 proteins in preventing UVB-induced release of pro-apoptotic factors from the mitochondria. Apoptosis 2007, 12: Dai H, Smith A, Meng XW, Schneider PA, Pang YP, Kaufmann SH: Transient binding of an activator BH3 domain to the Bak BH3-binding groove initiates Bak oligomerization. J Cell Biol 2011, 194: doi: / Cite this article as: Azad and Storey: BAK multimerization for apoptosis, but not bid binding, is inhibited by negatively charged residue in the BAK hydrophobic groove. Molecular Cancer :65. Submit your next manuscript to BioMed Central and take full advantage of: Convenient online submission Thorough peer review No space constraints or color figure charges Immediate publication on acceptance Inclusion in PubMed, CAS, Scopus and Google Scholar Research which is freely available for redistribution Submit your manuscript at
Programmed Cell Death (apoptosis)
 Programmed Cell Death (apoptosis) Stereotypic death process includes: membrane blebbing nuclear fragmentation chromatin condensation and DNA framentation loss of mitochondrial integrity and release of
Programmed Cell Death (apoptosis) Stereotypic death process includes: membrane blebbing nuclear fragmentation chromatin condensation and DNA framentation loss of mitochondrial integrity and release of
Table S1. New colony formation 7 days after stimulation with doxo and VCR in JURKAT cells
 Table S1. New colony formation 7 days after stimulation with and in JURKAT cells drug co + number of colonies 7±14 4±7 48±11 JURKAT cells were stimulated and analyzed as in Table 1. Drug concentrations
Table S1. New colony formation 7 days after stimulation with and in JURKAT cells drug co + number of colonies 7±14 4±7 48±11 JURKAT cells were stimulated and analyzed as in Table 1. Drug concentrations
Degradation of the Proapoptotic Proteins Bik, Puma, and Bim with Bcl-2 Domain 3 Homology in Chlamydia trachomatis-infected Cells
 INFECTION AND IMMUNITY, Mar. 2005, p. 1861 1864 Vol. 73, No. 3 0019-9567/05/$08.00 0 doi:10.1128/iai.73.3.1861 1864.2005 Copyright 2005, American Society for Microbiology. All Rights Reserved. Degradation
INFECTION AND IMMUNITY, Mar. 2005, p. 1861 1864 Vol. 73, No. 3 0019-9567/05/$08.00 0 doi:10.1128/iai.73.3.1861 1864.2005 Copyright 2005, American Society for Microbiology. All Rights Reserved. Degradation
Supplementary Information
 Supplementary Information Significance of p53 dynamics in regulating apoptosis in response to ionizing radiation, and polypharmacological strategies Bing Liu 1,, Divesh Bhatt 1,, Zoltan N. Oltvai 2, Joel
Supplementary Information Significance of p53 dynamics in regulating apoptosis in response to ionizing radiation, and polypharmacological strategies Bing Liu 1,, Divesh Bhatt 1,, Zoltan N. Oltvai 2, Joel
supplementary information
 DOI: 10.1038/ncb1875 Figure S1 (a) The 79 surgical specimens from NSCLC patients were analysed by immunohistochemistry with an anti-p53 antibody and control serum (data not shown). The normal bronchi served
DOI: 10.1038/ncb1875 Figure S1 (a) The 79 surgical specimens from NSCLC patients were analysed by immunohistochemistry with an anti-p53 antibody and control serum (data not shown). The normal bronchi served
Supplementary Figures
 Supplementary Figures Figure S1. Validation of kinase regulators of ONC201 sensitivity. Validation and screen results for changes in cell viability associated with the combination of ONC201 treatment (1
Supplementary Figures Figure S1. Validation of kinase regulators of ONC201 sensitivity. Validation and screen results for changes in cell viability associated with the combination of ONC201 treatment (1
Interplay of Bcl-2 Proteins Decides the Life or Death Fate
 The Open Cell Signaling Journal, 2011, 3, 3-8 3 Interplay of Bcl-2 Proteins Decides the Life or Death Fate Grant Dewson * Open Access Walter and Eliza Hall Institute of Medical Research, Melbourne, Victoria
The Open Cell Signaling Journal, 2011, 3, 3-8 3 Interplay of Bcl-2 Proteins Decides the Life or Death Fate Grant Dewson * Open Access Walter and Eliza Hall Institute of Medical Research, Melbourne, Victoria
Bcl-2 changes conformation to inhibit Bax oligomerization
 The EMBO Journal (26) 25, 2287 2296 & 26 European Molecular Biology Organization All Rights Reserved 261-4189/6 www.embojournal.org changes conformation to inhibit oligomerization THE EMBO JOURNAL Paulina
The EMBO Journal (26) 25, 2287 2296 & 26 European Molecular Biology Organization All Rights Reserved 261-4189/6 www.embojournal.org changes conformation to inhibit oligomerization THE EMBO JOURNAL Paulina
Apoptosis Chapter 9. Neelu Yadav PhD
 Apoptosis Chapter 9 Neelu Yadav PhD Neelu.Yadav@Roswellpark.org 1 Apoptosis: Lecture outline Apoptosis a programmed cell death pathway in normal homeostasis Core Apoptosis cascade is conserved Compare
Apoptosis Chapter 9 Neelu Yadav PhD Neelu.Yadav@Roswellpark.org 1 Apoptosis: Lecture outline Apoptosis a programmed cell death pathway in normal homeostasis Core Apoptosis cascade is conserved Compare
ab65311 Cytochrome c Releasing Apoptosis Assay Kit
 ab65311 Cytochrome c Releasing Apoptosis Assay Kit Instructions for Use For the rapid, sensitive and accurate detection of Cytochrome c translocation from Mitochondria into Cytosol during Apoptosis in
ab65311 Cytochrome c Releasing Apoptosis Assay Kit Instructions for Use For the rapid, sensitive and accurate detection of Cytochrome c translocation from Mitochondria into Cytosol during Apoptosis in
Silibinin i activates p53-caspase-2 pathway and causes caspase-mediated cleavage of Cip1/p21 in apoptosis
 Silibinin i activates p53-caspase-2 pathway and causes caspase-mediated cleavage of Cip1/p21 in apoptosis induction in bladder transitional-cell papilloma RT4 cells: Evidence for a regulatory loop between
Silibinin i activates p53-caspase-2 pathway and causes caspase-mediated cleavage of Cip1/p21 in apoptosis induction in bladder transitional-cell papilloma RT4 cells: Evidence for a regulatory loop between
Mitochondria in apoptosis. Jean-Claude Martinou, MD, Ph.D Department of cell biology University of Geneva Geneva, Switzerland
 Mitochondria in apoptosis Jean-Claude Martinou, MD, Ph.D Department of cell biology University of Geneva Geneva, Switzerland The dual role of mitochondria in life and death QuickTime and a TIFF (Uncompressed)
Mitochondria in apoptosis Jean-Claude Martinou, MD, Ph.D Department of cell biology University of Geneva Geneva, Switzerland The dual role of mitochondria in life and death QuickTime and a TIFF (Uncompressed)
Problem Set 8 Key 1 of 8
 7.06 2003 Problem Set 8 Key 1 of 8 7.06 2003 Problem Set 8 Key 1. As a bright MD/PhD, you are interested in questions about the control of cell number in the body. Recently, you've seen three patients
7.06 2003 Problem Set 8 Key 1 of 8 7.06 2003 Problem Set 8 Key 1. As a bright MD/PhD, you are interested in questions about the control of cell number in the body. Recently, you've seen three patients
SUPPLEMENTARY INFORMATION
 Figure S1 Induction of non-apoptotic death of SV40-transformed and primary DKO MEFs, and DKO thymocytes. (A-F) STS-induced non-apoptotic death of DKO MEF. (A, B) Reduced viability of DKO MEFs after exposure
Figure S1 Induction of non-apoptotic death of SV40-transformed and primary DKO MEFs, and DKO thymocytes. (A-F) STS-induced non-apoptotic death of DKO MEF. (A, B) Reduced viability of DKO MEFs after exposure
a b G75 G60 Sw-2 Sw-1 Supplementary Figure 1. Structure predictions by I-TASSER Server.
 a b G75 2 2 G60 Sw-2 Sw-1 Supplementary Figure 1. Structure predictions by I-TASSER Server. a. Overlay of top 10 models generated by I-TASSER illustrates the potential effect of 7 amino acid insertion
a b G75 2 2 G60 Sw-2 Sw-1 Supplementary Figure 1. Structure predictions by I-TASSER Server. a. Overlay of top 10 models generated by I-TASSER illustrates the potential effect of 7 amino acid insertion
Bioluminescence Resonance Energy Transfer (BRET)-based studies of receptor dynamics in living cells with Berthold s Mithras
 Bioluminescence Resonance Energy Transfer (BRET)-based studies of receptor dynamics in living cells with Berthold s Mithras Tarik Issad, Ralf Jockers and Stefano Marullo 1 Because they play a pivotal role
Bioluminescence Resonance Energy Transfer (BRET)-based studies of receptor dynamics in living cells with Berthold s Mithras Tarik Issad, Ralf Jockers and Stefano Marullo 1 Because they play a pivotal role
#19 Apoptosis Chapter 9. Neelu Yadav PhD
 #19 Apoptosis Chapter 9 Neelu Yadav PhD Neelu.Yadav@Roswellpark.org Why cells decide to die? - Stress, harmful, not needed - Completed its life span Death stimulation or Stress Cell Survival Death Functions
#19 Apoptosis Chapter 9 Neelu Yadav PhD Neelu.Yadav@Roswellpark.org Why cells decide to die? - Stress, harmful, not needed - Completed its life span Death stimulation or Stress Cell Survival Death Functions
Part-4. Cell cycle regulatory protein 5 (Cdk5) A novel target of ERK in Carb induced cell death
 Part-4 Cell cycle regulatory protein 5 (Cdk5) A novel target of ERK in Carb induced cell death 95 1. Introduction The process of replicating DNA and dividing cells can be described as a series of coordinated
Part-4 Cell cycle regulatory protein 5 (Cdk5) A novel target of ERK in Carb induced cell death 95 1. Introduction The process of replicating DNA and dividing cells can be described as a series of coordinated
Phosphorylation switches Bax from promoting to inhibiting apoptosis thereby increasing drug resistance
 Article Phosphorylation switches Bax from promoting to inhibiting apoptosis thereby increasing drug resistance Justin Kale 1,, Ozgur Kutuk 2,, Glauber Costa Brito 3, Tallulah S Andrews 4, Brian Leber 5,
Article Phosphorylation switches Bax from promoting to inhibiting apoptosis thereby increasing drug resistance Justin Kale 1,, Ozgur Kutuk 2,, Glauber Costa Brito 3, Tallulah S Andrews 4, Brian Leber 5,
Cell Biology Lecture 9 Notes Basic Principles of cell signaling and GPCR system
 Cell Biology Lecture 9 Notes Basic Principles of cell signaling and GPCR system Basic Elements of cell signaling: Signal or signaling molecule (ligand, first messenger) o Small molecules (epinephrine,
Cell Biology Lecture 9 Notes Basic Principles of cell signaling and GPCR system Basic Elements of cell signaling: Signal or signaling molecule (ligand, first messenger) o Small molecules (epinephrine,
Molecular biology :- Cancer genetics lecture 11
 Molecular biology :- Cancer genetics lecture 11 -We have talked about 2 group of genes that is involved in cellular transformation : proto-oncogenes and tumour suppressor genes, and it isn t enough to
Molecular biology :- Cancer genetics lecture 11 -We have talked about 2 group of genes that is involved in cellular transformation : proto-oncogenes and tumour suppressor genes, and it isn t enough to
Effects of Second Messengers
 Effects of Second Messengers Inositol trisphosphate Diacylglycerol Opens Calcium Channels Binding to IP 3 -gated Channel Cooperative binding Activates Protein Kinase C is required Phosphorylation of many
Effects of Second Messengers Inositol trisphosphate Diacylglycerol Opens Calcium Channels Binding to IP 3 -gated Channel Cooperative binding Activates Protein Kinase C is required Phosphorylation of many
p53 and Apoptosis: Master Guardian and Executioner Part 2
 p53 and Apoptosis: Master Guardian and Executioner Part 2 p14arf in human cells is a antagonist of Mdm2. The expression of ARF causes a rapid increase in p53 levels, so what would you suggest?.. The enemy
p53 and Apoptosis: Master Guardian and Executioner Part 2 p14arf in human cells is a antagonist of Mdm2. The expression of ARF causes a rapid increase in p53 levels, so what would you suggest?.. The enemy
Chapter 11: Enzyme Catalysis
 Chapter 11: Enzyme Catalysis Matching A) high B) deprotonated C) protonated D) least resistance E) motion F) rate-determining G) leaving group H) short peptides I) amino acid J) low K) coenzymes L) concerted
Chapter 11: Enzyme Catalysis Matching A) high B) deprotonated C) protonated D) least resistance E) motion F) rate-determining G) leaving group H) short peptides I) amino acid J) low K) coenzymes L) concerted
The discovery of Bcl-2
 The discovery of Bcl-2 Bcl-2: B-cell lymphoma 2 The pro-survival subfamily of Bcl-2 protein family Cloning of Bcl-2 as the oncogene which is deregulated at t(14;18) lymphomas Pioneer works by Tsujimoto
The discovery of Bcl-2 Bcl-2: B-cell lymphoma 2 The pro-survival subfamily of Bcl-2 protein family Cloning of Bcl-2 as the oncogene which is deregulated at t(14;18) lymphomas Pioneer works by Tsujimoto
Apoptotic Pathways in Mammals Dr. Douglas R. Green
 Apoptotic Pathways in Mammals Douglas R. Green 1 Apoptosis A form of cell death that is defined morphologically, and features a number of biochemical events Programmed cell death Cell death that occurs
Apoptotic Pathways in Mammals Douglas R. Green 1 Apoptosis A form of cell death that is defined morphologically, and features a number of biochemical events Programmed cell death Cell death that occurs
SUPPLEMENTARY INFORMATION
 Supplementary Discussion The cell cycle machinery and the DNA damage response network are highly interconnected and co-regulated in assuring faithful duplication and partition of genetic materials into
Supplementary Discussion The cell cycle machinery and the DNA damage response network are highly interconnected and co-regulated in assuring faithful duplication and partition of genetic materials into
Discoveries and Controversies in BCL-2 Proteins Mediated Apoptosis. Janet Zheng, Ariele Viacava Follis, Richard W. Kriwacki*, and Tudor Moldoveanu*
 Received Date : 21-Jun-2015 Revised Date : 30-Aug-2015 Accepted Date : 23-Sep-2015 Article type : Minireview Discoveries and Controversies in BCL-2 Proteins Mediated Apoptosis Janet Zheng, Ariele Viacava
Received Date : 21-Jun-2015 Revised Date : 30-Aug-2015 Accepted Date : 23-Sep-2015 Article type : Minireview Discoveries and Controversies in BCL-2 Proteins Mediated Apoptosis Janet Zheng, Ariele Viacava
ERK1/2/MAPK pathway-dependent regulation of the telomeric factor TRF2
 ERK1/2/MAPK pathway-dependent regulation of the telomeric factor TRF2 SUPPLEMENTARY FIGURES AND TABLE Supplementary Figure S1: Conservation of the D domain throughout evolution. Alignment of TRF2 sequences
ERK1/2/MAPK pathway-dependent regulation of the telomeric factor TRF2 SUPPLEMENTARY FIGURES AND TABLE Supplementary Figure S1: Conservation of the D domain throughout evolution. Alignment of TRF2 sequences
Supporting Information. Structure-based Lead Optimization and Biological Evaluation of BAX Direct Activators as Novel Potential Anticancer Agents
 Supporting Information Structure-based Lead Optimization and Biological Evaluation of BAX Direct Activators as Novel Potential Anticancer Agents Mariano Stornaiuolo, 1 * Giuseppe La Regina, 2 Sara Passacantilli,
Supporting Information Structure-based Lead Optimization and Biological Evaluation of BAX Direct Activators as Novel Potential Anticancer Agents Mariano Stornaiuolo, 1 * Giuseppe La Regina, 2 Sara Passacantilli,
Apoptosis Oncogenes. Srbová Martina
 Apoptosis Oncogenes Srbová Martina Cell Cycle Control point Cyclin B Cdk1 Cyclin D Cdk4 Cdk6 Cyclin A Cdk2 Cyclin E Cdk2 Cyclin-dependent kinase (Cdk) have to bind a cyclin to become active Regulation
Apoptosis Oncogenes Srbová Martina Cell Cycle Control point Cyclin B Cdk1 Cyclin D Cdk4 Cdk6 Cyclin A Cdk2 Cyclin E Cdk2 Cyclin-dependent kinase (Cdk) have to bind a cyclin to become active Regulation
Enzyme-coupled Receptors. Cell-surface receptors 1. Ion-channel-coupled receptors 2. G-protein-coupled receptors 3. Enzyme-coupled receptors
 Enzyme-coupled Receptors Cell-surface receptors 1. Ion-channel-coupled receptors 2. G-protein-coupled receptors 3. Enzyme-coupled receptors Cell-surface receptors allow a flow of ions across the plasma
Enzyme-coupled Receptors Cell-surface receptors 1. Ion-channel-coupled receptors 2. G-protein-coupled receptors 3. Enzyme-coupled receptors Cell-surface receptors allow a flow of ions across the plasma
Determination of the temporal pattern and importance of BALF1 expression in Epstein-Barr viral infection
 Determination of the temporal pattern and importance of BALF1 expression in Epstein-Barr viral infection Melissa Mihelidakis May 6, 2004 7.340 Research Proposal Introduction Apoptosis, or programmed cell
Determination of the temporal pattern and importance of BALF1 expression in Epstein-Barr viral infection Melissa Mihelidakis May 6, 2004 7.340 Research Proposal Introduction Apoptosis, or programmed cell
Cell death and the mitochondria: therapeutic targeting of the BCL-2 family-driven pathway
 British Journal of Pharmacology DOI:10.1111/bph.12431 www.brjpharmacol.org Themed Issue: Mitochondrial Pharmacology: Energy, Injury & Beyond REVIEW Cell death and the mitochondria: therapeutic targeting
British Journal of Pharmacology DOI:10.1111/bph.12431 www.brjpharmacol.org Themed Issue: Mitochondrial Pharmacology: Energy, Injury & Beyond REVIEW Cell death and the mitochondria: therapeutic targeting
KEY CONCEPT QUESTIONS IN SIGNAL TRANSDUCTION
 Signal Transduction - Part 2 Key Concepts - Receptor tyrosine kinases control cell metabolism and proliferation Growth factor signaling through Ras Mutated cell signaling genes in cancer cells are called
Signal Transduction - Part 2 Key Concepts - Receptor tyrosine kinases control cell metabolism and proliferation Growth factor signaling through Ras Mutated cell signaling genes in cancer cells are called
BIO360 Fall 2013 Quiz 1
 BIO360 Fall 2013 Quiz 1 1. Examine the diagram below. There are two homologous copies of chromosome one and the allele of YFG carried on the light gray chromosome has undergone a loss-of-function mutation.
BIO360 Fall 2013 Quiz 1 1. Examine the diagram below. There are two homologous copies of chromosome one and the allele of YFG carried on the light gray chromosome has undergone a loss-of-function mutation.
A new score predicting the survival of patients with spinal cord compression from myeloma
 Douglas et al. BMC Cancer 2012, 12:425 RESEARCH ARTICLE Open Access A new score predicting the survival of patients with spinal cord compression from myeloma Sarah Douglas 1, Steven E Schild 2 and Dirk
Douglas et al. BMC Cancer 2012, 12:425 RESEARCH ARTICLE Open Access A new score predicting the survival of patients with spinal cord compression from myeloma Sarah Douglas 1, Steven E Schild 2 and Dirk
Apoptosis-based Therapies: Mechanisms and Applications
 Apoptosis-based Therapies: Mechanisms and Applications Perspective Bcl-2 family members, potential usage of BH3 domains as drug targets Bcl-2/xL inhibitors - Antisense, inhibitors of protein-protein interactions,
Apoptosis-based Therapies: Mechanisms and Applications Perspective Bcl-2 family members, potential usage of BH3 domains as drug targets Bcl-2/xL inhibitors - Antisense, inhibitors of protein-protein interactions,
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION doi:10.1038/nature11429 S1a 6 7 8 9 Nlrc4 allele S1b Nlrc4 +/+ Nlrc4 +/F Nlrc4 F/F 9 Targeting construct 422 bp 273 bp FRT-neo-gb-PGK-FRT 3x.STOP S1c Nlrc4 +/+ Nlrc4 F/F casp1
SUPPLEMENTARY INFORMATION doi:10.1038/nature11429 S1a 6 7 8 9 Nlrc4 allele S1b Nlrc4 +/+ Nlrc4 +/F Nlrc4 F/F 9 Targeting construct 422 bp 273 bp FRT-neo-gb-PGK-FRT 3x.STOP S1c Nlrc4 +/+ Nlrc4 F/F casp1
Signaling Apoptosis. Scott André Oakes, M.D. Dept. of Pathology Univ. of Calif-San Francisco. Cyt c Release BAX/BAK. Apoptosome Formation
 Signaling Apoptosis Cyt c Release BAX/BAK Apoptosome Formation Caspase Activation Scott André Oakes, M.D. Dept. of Pathology Univ. of Calif-San Francisco Why do we need cell death? Sculpt Organs Control
Signaling Apoptosis Cyt c Release BAX/BAK Apoptosome Formation Caspase Activation Scott André Oakes, M.D. Dept. of Pathology Univ. of Calif-San Francisco Why do we need cell death? Sculpt Organs Control
Introduction to pathology lecture 5/ Cell injury apoptosis. Dr H Awad 2017/18
 Introduction to pathology lecture 5/ Cell injury apoptosis Dr H Awad 2017/18 Apoptosis = programmed cell death = cell suicide= individual cell death Apoptosis cell death induced by a tightly regulated
Introduction to pathology lecture 5/ Cell injury apoptosis Dr H Awad 2017/18 Apoptosis = programmed cell death = cell suicide= individual cell death Apoptosis cell death induced by a tightly regulated
ARV Mode of Action. Mode of Action. Mode of Action NRTI. Immunopaedia.org.za
 ARV Mode of Action Mode of Action Mode of Action - NRTI Mode of Action - NNRTI Mode of Action - Protease Inhibitors Mode of Action - Integrase inhibitor Mode of Action - Entry Inhibitors Mode of Action
ARV Mode of Action Mode of Action Mode of Action - NRTI Mode of Action - NNRTI Mode of Action - Protease Inhibitors Mode of Action - Integrase inhibitor Mode of Action - Entry Inhibitors Mode of Action
APPLICATION NOTE 1850 Millrace Drive, Suite 3A Eugene, Oregon
 APPLICATION NOTE 185 Millrace Drive, Suite 3A Eugene, Oregon 973 In-Cell ELISA (ICE) Assay Platform Monitoring apoptosis in cells: a high-throughput, quantitative cellbased assay. Rev. Introduction: Apoptosis:
APPLICATION NOTE 185 Millrace Drive, Suite 3A Eugene, Oregon 973 In-Cell ELISA (ICE) Assay Platform Monitoring apoptosis in cells: a high-throughput, quantitative cellbased assay. Rev. Introduction: Apoptosis:
Modulating the Bcl-2 Family of Apoptosis Suppressors for Potential Therapeutic Benefit in Cancer
 Modulating the Bcl-2 Family of Apoptosis Suppressors for Potential Therapeutic Benefit in Cancer Gordon C. Shore and Jean Viallet Members of the BCL-2 family of proteins regulate and execute many cell
Modulating the Bcl-2 Family of Apoptosis Suppressors for Potential Therapeutic Benefit in Cancer Gordon C. Shore and Jean Viallet Members of the BCL-2 family of proteins regulate and execute many cell
Mitochondria/Cytosol Fractionation Kit
 ab65320 Mitochondria/Cytosol Fractionation Kit Instructions for Use For the rapid, sensitive and accurate isolation of Mitochondrial and Cytosolic fractions from living cells. This product is for research
ab65320 Mitochondria/Cytosol Fractionation Kit Instructions for Use For the rapid, sensitive and accurate isolation of Mitochondrial and Cytosolic fractions from living cells. This product is for research
Supplementary Figure S1. Venn diagram analysis of mrna microarray data and mirna target analysis. (a) Western blot analysis of T lymphoblasts (CLS)
 Supplementary Figure S1. Venn diagram analysis of mrna microarray data and mirna target analysis. (a) Western blot analysis of T lymphoblasts (CLS) and their exosomes (EXO) in resting (REST) and activated
Supplementary Figure S1. Venn diagram analysis of mrna microarray data and mirna target analysis. (a) Western blot analysis of T lymphoblasts (CLS) and their exosomes (EXO) in resting (REST) and activated
Apoptotic cell signaling in cancer progression and therapyw
 Integrative Biology Dynamic Article Links Cite this: Integr. Biol., 2011, 3, 279 296 www.rsc.org/ibiology REVIEW ARTICLE Apoptotic cell signaling in cancer progression and therapyw Jessica Plati, a Octavian
Integrative Biology Dynamic Article Links Cite this: Integr. Biol., 2011, 3, 279 296 www.rsc.org/ibiology REVIEW ARTICLE Apoptotic cell signaling in cancer progression and therapyw Jessica Plati, a Octavian
Myelin suppresses axon regeneration by PIR-B/SHPmediated inhibition of Trk activity
 Manuscript EMBO-2010-76298 Myelin suppresses axon regeneration by PIR-B/SHPmediated inhibition of Trk activity Yuki Fujita, Shota Endo, Toshiyuki Takai and Toshihide Yamashita Corresponding author: Toshihide
Manuscript EMBO-2010-76298 Myelin suppresses axon regeneration by PIR-B/SHPmediated inhibition of Trk activity Yuki Fujita, Shota Endo, Toshiyuki Takai and Toshihide Yamashita Corresponding author: Toshihide
Pro-apoptotic signalling through Toll-like receptor 3 involves TRIF-dependent
 Pro-apoptotic signalling through Toll-like receptor 3 involves TRIF-dependent activation of caspase-8 and is under the control of inhibitor of apoptosis proteins in melanoma cells Arnim Weber, Zofia Kirejczyk,
Pro-apoptotic signalling through Toll-like receptor 3 involves TRIF-dependent activation of caspase-8 and is under the control of inhibitor of apoptosis proteins in melanoma cells Arnim Weber, Zofia Kirejczyk,
BIK BIM NOXA PUMA MCL-1. p53
 HT116 cells A IK IM NOXA PUMA ML-1 p53 48 24 48 24 48 24 48 24 48 24 48 24 48 24 48 24 48 Procaspase 3 PARP leaved Product 12 8 4 24 hr 48 hr Figure S1. HT116 cell death by different proteasome inhibitors.
HT116 cells A IK IM NOXA PUMA ML-1 p53 48 24 48 24 48 24 48 24 48 24 48 24 48 24 48 24 48 Procaspase 3 PARP leaved Product 12 8 4 24 hr 48 hr Figure S1. HT116 cell death by different proteasome inhibitors.
Early cell death (FGF) B No RunX transcription factor produced Yes No differentiation
 Solution Key - Practice Questions Question 1 a) A recent publication has shown that the fat stem cells (FSC) can act as bone stem cells to repair cavities in the skull, when transplanted into immuno-compromised
Solution Key - Practice Questions Question 1 a) A recent publication has shown that the fat stem cells (FSC) can act as bone stem cells to repair cavities in the skull, when transplanted into immuno-compromised
Signal-Transduction Cascades - 2. The Phosphoinositide Cascade
 Signal-Transduction Cascades - 2 The Phosphoinositide Cascade Calcium ion as a second messenger Tyrosine kinase and receptor dimerization scribd.com Faisal Khatib JU The Phosphoinositide Cascade Used by
Signal-Transduction Cascades - 2 The Phosphoinositide Cascade Calcium ion as a second messenger Tyrosine kinase and receptor dimerization scribd.com Faisal Khatib JU The Phosphoinositide Cascade Used by
Mammalian Membrane Protein Extraction Kit
 Mammalian Membrane Protein Extraction Kit Catalog number: AR0155 Boster s Mammalian Membrane Protein Extraction Kit is a simple, rapid and reproducible method to prepare cellular protein fractions highly
Mammalian Membrane Protein Extraction Kit Catalog number: AR0155 Boster s Mammalian Membrane Protein Extraction Kit is a simple, rapid and reproducible method to prepare cellular protein fractions highly
Type of file: PDF Title of file for HTML: Supplementary Information Description: Supplementary Figures
 Type of file: PDF Title of file for HTML: Supplementary Information Description: Supplementary Figures Type of file: MOV Title of file for HTML: Supplementary Movie 1 Description: NLRP3 is moving along
Type of file: PDF Title of file for HTML: Supplementary Information Description: Supplementary Figures Type of file: MOV Title of file for HTML: Supplementary Movie 1 Description: NLRP3 is moving along
Part I Molecular Cell Biology
 1 Part I Molecular Cell Biology RNA Regulation: Advances in Molecular Biology and Medicine, First Edition. Edited by Robert A. Meyers. 2014 Wiley-VCH Verlag GmbH & Co. KGaA. Published 2014 by Wiley-VCH
1 Part I Molecular Cell Biology RNA Regulation: Advances in Molecular Biology and Medicine, First Edition. Edited by Robert A. Meyers. 2014 Wiley-VCH Verlag GmbH & Co. KGaA. Published 2014 by Wiley-VCH
G-Protein Signaling. Introduction to intracellular signaling. Dr. SARRAY Sameh, Ph.D
 G-Protein Signaling Introduction to intracellular signaling Dr. SARRAY Sameh, Ph.D Cell signaling Cells communicate via extracellular signaling molecules (Hormones, growth factors and neurotransmitters
G-Protein Signaling Introduction to intracellular signaling Dr. SARRAY Sameh, Ph.D Cell signaling Cells communicate via extracellular signaling molecules (Hormones, growth factors and neurotransmitters
BCL-2 Proteins and Outer Mitochondrial Membrane Permeabilization. AP Research STEM. Word Count: 4226
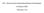 BCL-2 Proteins and Outer Mitochondrial Membrane Permeabilization AP Research STEM Word Count: 4226 BCL-2 PROTEINS AND OUTER MITOCHONDRIAL MEMBRANE PERMEABILIZATION 1 Abstract This paper is a review of
BCL-2 Proteins and Outer Mitochondrial Membrane Permeabilization AP Research STEM Word Count: 4226 BCL-2 PROTEINS AND OUTER MITOCHONDRIAL MEMBRANE PERMEABILIZATION 1 Abstract This paper is a review of
Signal Transduction Cascades
 Signal Transduction Cascades Contents of this page: Kinases & phosphatases Protein Kinase A (camp-dependent protein kinase) G-protein signal cascade Structure of G-proteins Small GTP-binding proteins,
Signal Transduction Cascades Contents of this page: Kinases & phosphatases Protein Kinase A (camp-dependent protein kinase) G-protein signal cascade Structure of G-proteins Small GTP-binding proteins,
AperTO - Archivio Istituzionale Open Access dell'università di Torino
 AperTO - Archivio Istituzionale Open Access dell'università di Torino From the nucleus to the mitochondria and backthe odyssey of a multitask STAT3 This is the author's manuscript Original Citation: From
AperTO - Archivio Istituzionale Open Access dell'università di Torino From the nucleus to the mitochondria and backthe odyssey of a multitask STAT3 This is the author's manuscript Original Citation: From
The Immunoassay Guide to Successful Mass Spectrometry. Orr Sharpe Robinson Lab SUMS User Meeting October 29, 2013
 The Immunoassay Guide to Successful Mass Spectrometry Orr Sharpe Robinson Lab SUMS User Meeting October 29, 2013 What is it? Hey! Look at that! Something is reacting in here! I just wish I knew what it
The Immunoassay Guide to Successful Mass Spectrometry Orr Sharpe Robinson Lab SUMS User Meeting October 29, 2013 What is it? Hey! Look at that! Something is reacting in here! I just wish I knew what it
SUPPLEMENTARY INFORMATION
 Supplementary Table 1. Cell sphingolipids and S1P bound to endogenous TRAF2. Sphingolipid Cell pmol/mg TRAF2 immunoprecipitate pmol/mg Sphingomyelin 4200 ± 250 Not detected Monohexosylceramide 311 ± 18
Supplementary Table 1. Cell sphingolipids and S1P bound to endogenous TRAF2. Sphingolipid Cell pmol/mg TRAF2 immunoprecipitate pmol/mg Sphingomyelin 4200 ± 250 Not detected Monohexosylceramide 311 ± 18
Cbl ubiquitin ligase: Lord of the RINGs
 Cbl ubiquitin ligase: Lord of the RINGs Not just quite interesting - really interesting! A cell must be able to degrade proteins when their activity is no longer required. Many eukaryotic proteins are
Cbl ubiquitin ligase: Lord of the RINGs Not just quite interesting - really interesting! A cell must be able to degrade proteins when their activity is no longer required. Many eukaryotic proteins are
CELLULAR & MOLECULAR BIOLOGY LETTERS
 CELLULAR & MOLECULAR BIOLOGY LETTERS http://www.cmbl.org.pl Received: 17 May 2012 Volume 18 (2013) pp 58-74 Final form accepted: 31 October 2012 DOI: 10.2478/s11658-012-0039-y Published online: 16 November
CELLULAR & MOLECULAR BIOLOGY LETTERS http://www.cmbl.org.pl Received: 17 May 2012 Volume 18 (2013) pp 58-74 Final form accepted: 31 October 2012 DOI: 10.2478/s11658-012-0039-y Published online: 16 November
(B D) Three views of the final refined 2Fo-Fc electron density map of the Vpr (red)-ung2 (green) interacting region, contoured at 1.4σ.
 Supplementary Figure 1 Overall structure of the DDB1 DCAF1 Vpr UNG2 complex. (A) The final refined 2Fo-Fc electron density map, contoured at 1.4σ of Vpr, illustrating well-defined side chains. (B D) Three
Supplementary Figure 1 Overall structure of the DDB1 DCAF1 Vpr UNG2 complex. (A) The final refined 2Fo-Fc electron density map, contoured at 1.4σ of Vpr, illustrating well-defined side chains. (B D) Three
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION doi:1.138/nature9814 a A SHARPIN FL B SHARPIN ΔNZF C SHARPIN T38L, F39V b His-SHARPIN FL -1xUb -2xUb -4xUb α-his c Linear 4xUb -SHARPIN FL -SHARPIN TF_LV -SHARPINΔNZF -SHARPIN
SUPPLEMENTARY INFORMATION doi:1.138/nature9814 a A SHARPIN FL B SHARPIN ΔNZF C SHARPIN T38L, F39V b His-SHARPIN FL -1xUb -2xUb -4xUb α-his c Linear 4xUb -SHARPIN FL -SHARPIN TF_LV -SHARPINΔNZF -SHARPIN
Nature Immunology: doi: /ni.3866
 Nature Immunology: doi:10.1038/ni.3866 Supplementary Figure 1 The effect of TIPE2 on chemotaxis. a, The expression of TIPE2 in dhl-60c, dhl-60t, TIPE2-expressing and 15/16Q-expressing dhl-60t neutrophils
Nature Immunology: doi:10.1038/ni.3866 Supplementary Figure 1 The effect of TIPE2 on chemotaxis. a, The expression of TIPE2 in dhl-60c, dhl-60t, TIPE2-expressing and 15/16Q-expressing dhl-60t neutrophils
Crystallization-grade After D After V3 cocktail. Time (s) Time (s) Time (s) Time (s) Time (s) Time (s)
 Ligand Type Name 6 Crystallization-grade After 447-52D After V3 cocktail Receptor CD4 Resonance Units 5 1 5 1 5 1 Broadly neutralizing antibodies 2G12 VRC26.9 Resonance Units Resonance Units 3 1 15 1 5
Ligand Type Name 6 Crystallization-grade After 447-52D After V3 cocktail Receptor CD4 Resonance Units 5 1 5 1 5 1 Broadly neutralizing antibodies 2G12 VRC26.9 Resonance Units Resonance Units 3 1 15 1 5
MEK1 Assay Kit 1 Catalog # Lot # 16875
 MEK1 Assay Kit 1 Kit Components Assay Dilution Buffer (ADB), Catalog # 20-108. Three vials, each containing 1.0ml of assay dilution buffer (20mM MOPS, ph 7.2, 25mM ß-glycerol phosphate, 5mM EGTA, 1mM sodium
MEK1 Assay Kit 1 Kit Components Assay Dilution Buffer (ADB), Catalog # 20-108. Three vials, each containing 1.0ml of assay dilution buffer (20mM MOPS, ph 7.2, 25mM ß-glycerol phosphate, 5mM EGTA, 1mM sodium
Supplementary Figure 1. Normal T lymphocyte populations in Dapk -/- mice. (a) Normal thymic development in Dapk -/- mice. Thymocytes from WT and Dapk
 Supplementary Figure 1. Normal T lymphocyte populations in Dapk -/- mice. (a) Normal thymic development in Dapk -/- mice. Thymocytes from WT and Dapk -/- mice were stained for expression of CD4 and CD8.
Supplementary Figure 1. Normal T lymphocyte populations in Dapk -/- mice. (a) Normal thymic development in Dapk -/- mice. Thymocytes from WT and Dapk -/- mice were stained for expression of CD4 and CD8.
Chapter 2 The Bcl-2 Family Proteins
 Chapter 2 The Bcl-2 Family Proteins Wen-Xing Ding and Xiao-Ming Yin Abstract The Bcl-2 family proteins are a group of evolutionarily conserved molecules that regulate apoptosis mainly at the site of mitochondria.
Chapter 2 The Bcl-2 Family Proteins Wen-Xing Ding and Xiao-Ming Yin Abstract The Bcl-2 family proteins are a group of evolutionarily conserved molecules that regulate apoptosis mainly at the site of mitochondria.
Under the Radar Screen: How Bugs Trick Our Immune Defenses
 Under the Radar Screen: How Bugs Trick Our Immune Defenses Session 8: Apoptosis Marie-Eve Paquet and Gijsbert Grotenbreg Whitehead Institute for Biomedical Research Myxoma virus Poxvirus Infects rabbits
Under the Radar Screen: How Bugs Trick Our Immune Defenses Session 8: Apoptosis Marie-Eve Paquet and Gijsbert Grotenbreg Whitehead Institute for Biomedical Research Myxoma virus Poxvirus Infects rabbits
NIH Public Access Author Manuscript Nature. Author manuscript; available in PMC 2010 January 31.
 NIH Public Access Author Manuscript Published in final edited form as: Nature. 2009 April 30; 458(7242): 1127. doi:10.1038/nature07986. Cytoplasmic Functions of the Tumor Suppressor p53 Douglas R. Green
NIH Public Access Author Manuscript Published in final edited form as: Nature. 2009 April 30; 458(7242): 1127. doi:10.1038/nature07986. Cytoplasmic Functions of the Tumor Suppressor p53 Douglas R. Green
An Epstein-Barr virus-encoded microrna targets PUMA to promote host cell survival
 An Epstein-Barr virus-encoded microrna targets to promote host cell survival The Journal of Experimental Medicine 205(11): 2551-2560, 2008. 1 Elizabeth Yee-Wai Choy, Kam-Leung Siu, Kin-Hang Kok, Raymond
An Epstein-Barr virus-encoded microrna targets to promote host cell survival The Journal of Experimental Medicine 205(11): 2551-2560, 2008. 1 Elizabeth Yee-Wai Choy, Kam-Leung Siu, Kin-Hang Kok, Raymond
Work-flow: protein sample preparation Precipitation methods Removal of interfering substances Specific examples:
 Dr. Sanjeeva Srivastava IIT Bombay Work-flow: protein sample preparation Precipitation methods Removal of interfering substances Specific examples: Sample preparation for serum proteome analysis Sample
Dr. Sanjeeva Srivastava IIT Bombay Work-flow: protein sample preparation Precipitation methods Removal of interfering substances Specific examples: Sample preparation for serum proteome analysis Sample
Tala Saleh. Ahmad Attari. Mamoun Ahram
 23 Tala Saleh Ahmad Attari Minna Mushtaha Mamoun Ahram In the previous lecture, we discussed the mechanisms of regulating enzymes through inhibitors. Now, we will start this lecture by discussing regulation
23 Tala Saleh Ahmad Attari Minna Mushtaha Mamoun Ahram In the previous lecture, we discussed the mechanisms of regulating enzymes through inhibitors. Now, we will start this lecture by discussing regulation
Sodium selenite induces apoptosis in colon cancer cells via Bax-dependent mitochondrial pathway
 European Review for Medical and Pharmacological Sciences Sodium selenite induces apoptosis in colon cancer cells via Bax-dependent mitochondrial pathway Z. LI 1,2, J. MENG 3, T.-J. XU 4, X.-Y. QIN 5, X.-D.
European Review for Medical and Pharmacological Sciences Sodium selenite induces apoptosis in colon cancer cells via Bax-dependent mitochondrial pathway Z. LI 1,2, J. MENG 3, T.-J. XU 4, X.-Y. QIN 5, X.-D.
Bio 401 Sp2014. Multiple Choice (Circle the letter corresponding to the correct answer)
 MIDTERM EXAM KEY (60 pts) You should have 5 pages. Please put your name on every page. You will have 70 minutes to complete the exam. You may begin working as soon as you receive the exam. NOTE: the RED
MIDTERM EXAM KEY (60 pts) You should have 5 pages. Please put your name on every page. You will have 70 minutes to complete the exam. You may begin working as soon as you receive the exam. NOTE: the RED
Phenylketonuria (PKU) Structure of Phenylalanine Hydroxylase. Biol 405 Molecular Medicine
 Phenylketonuria (PKU) Structure of Phenylalanine Hydroxylase Biol 405 Molecular Medicine 1998 Crystal structure of phenylalanine hydroxylase solved. The polypeptide consists of three regions: Regulatory
Phenylketonuria (PKU) Structure of Phenylalanine Hydroxylase Biol 405 Molecular Medicine 1998 Crystal structure of phenylalanine hydroxylase solved. The polypeptide consists of three regions: Regulatory
Transcription-independent pro-apoptotic functions of p53 Ute M Moll 1, Sonja Wolff 1, Daniel Speidel 2 and Wolfgang Deppert 2
 Transcription-independent pro-apoptotic functions of p53 Ute M Moll 1, Sonja Wolff 1, Daniel Speidel 2 and Wolfgang Deppert 2 Induction of apoptosis is one of the central activities by which p53 exerts
Transcription-independent pro-apoptotic functions of p53 Ute M Moll 1, Sonja Wolff 1, Daniel Speidel 2 and Wolfgang Deppert 2 Induction of apoptosis is one of the central activities by which p53 exerts
Signal Transduction: G-Protein Coupled Receptors
 Signal Transduction: G-Protein Coupled Receptors Federle, M. (2017). Lectures 4-5: Signal Transduction parts 1&2: nuclear receptors and GPCRs. Lecture presented at PHAR 423 Lecture in UIC College of Pharmacy,
Signal Transduction: G-Protein Coupled Receptors Federle, M. (2017). Lectures 4-5: Signal Transduction parts 1&2: nuclear receptors and GPCRs. Lecture presented at PHAR 423 Lecture in UIC College of Pharmacy,
Supplemental Data Macrophage Migration Inhibitory Factor MIF Interferes with the Rb-E2F Pathway
 Supplemental Data Macrophage Migration Inhibitory Factor MIF Interferes with the Rb-E2F Pathway S1 Oleksi Petrenko and Ute M. Moll Figure S1. MIF-Deficient Cells Have Reduced Transforming Ability (A) Soft
Supplemental Data Macrophage Migration Inhibitory Factor MIF Interferes with the Rb-E2F Pathway S1 Oleksi Petrenko and Ute M. Moll Figure S1. MIF-Deficient Cells Have Reduced Transforming Ability (A) Soft
Supplementary Figure 1
 Supplementary Figure 1 a γ-h2ax MDC1 RNF8 FK2 BRCA1 U2OS Cells sgrna-1 ** 60 sgrna 40 20 0 % positive Cells (>5 foci per cell) b ** 80 sgrna sgrna γ-h2ax MDC1 γ-h2ax RNF8 FK2 MDC1 BRCA1 RNF8 FK2 BRCA1
Supplementary Figure 1 a γ-h2ax MDC1 RNF8 FK2 BRCA1 U2OS Cells sgrna-1 ** 60 sgrna 40 20 0 % positive Cells (>5 foci per cell) b ** 80 sgrna sgrna γ-h2ax MDC1 γ-h2ax RNF8 FK2 MDC1 BRCA1 RNF8 FK2 BRCA1
William C. Comb, Jessica E. Hutti, Patricia Cogswell, Lewis C. Cantley, and Albert S. Baldwin
 Molecular Cell, Volume 45 Supplemental Information p85 SH2 Domain Phosphorylation by IKK Promotes Feedback Inhibition of PI3K and Akt in Response to Cellular Starvation William C. Comb, Jessica E. Hutti,
Molecular Cell, Volume 45 Supplemental Information p85 SH2 Domain Phosphorylation by IKK Promotes Feedback Inhibition of PI3K and Akt in Response to Cellular Starvation William C. Comb, Jessica E. Hutti,
How the Bcl 2 family of proteins interact to regulate apoptosis
 REVIEW Mark F van Delft and David CS Huang Cell Research (2006)16: 203-213 2006 IBCB, SIBS, CAS All rights reserved 1001-0602/06 $ 30.00 203 www.nature.com/cr How the Bcl 2 family of proteins interact
REVIEW Mark F van Delft and David CS Huang Cell Research (2006)16: 203-213 2006 IBCB, SIBS, CAS All rights reserved 1001-0602/06 $ 30.00 203 www.nature.com/cr How the Bcl 2 family of proteins interact
Propagation of the Signal
 OpenStax-CNX module: m44452 1 Propagation of the Signal OpenStax College This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License 3.0 By the end of this section,
OpenStax-CNX module: m44452 1 Propagation of the Signal OpenStax College This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License 3.0 By the end of this section,
Genetic information flows from mrna to protein through the process of translation
 Genetic information flows from mrn to protein through the process of translation TYPES OF RN (RIBONUCLEIC CID) RN s job - protein synthesis (assembly of amino acids into proteins) Three main types: 1.
Genetic information flows from mrn to protein through the process of translation TYPES OF RN (RIBONUCLEIC CID) RN s job - protein synthesis (assembly of amino acids into proteins) Three main types: 1.
Cancer Translational Medicine
 CTM Review Cancer Translational Medicine Cancer Transl Med 2016;2(1):7 20 doi: 10.4103/2395-3977.177558 BCL2 Family, Mitochondrial Apoptosis, and Beyond Haiming Dai 1, X. Wei Meng 2, Scott H. Kaufmann
CTM Review Cancer Translational Medicine Cancer Transl Med 2016;2(1):7 20 doi: 10.4103/2395-3977.177558 BCL2 Family, Mitochondrial Apoptosis, and Beyond Haiming Dai 1, X. Wei Meng 2, Scott H. Kaufmann
AACR 101st Annual Meeting 2010, Washington D.C. Experimental and Molecular Therapeutics Section 29; Abstract #3855
 Investigation of the Growth Inhibitory Activity of the MEK Inhibitor ARRY-162 in Combination with Everolimus in a Variety of KRas and PI3K Pathway Mutant Cancers Brian Tunquist, Tyler Risom, Debbie Anderson,
Investigation of the Growth Inhibitory Activity of the MEK Inhibitor ARRY-162 in Combination with Everolimus in a Variety of KRas and PI3K Pathway Mutant Cancers Brian Tunquist, Tyler Risom, Debbie Anderson,
Recombinant Protein Expression Retroviral system
 Recombinant Protein Expression Retroviral system Viruses Contains genome DNA or RNA Genome encased in a protein coat or capsid. Some viruses have membrane covering protein coat enveloped virus Ø Essential
Recombinant Protein Expression Retroviral system Viruses Contains genome DNA or RNA Genome encased in a protein coat or capsid. Some viruses have membrane covering protein coat enveloped virus Ø Essential
Complexity DNA. Genome RNA. Transcriptome. Protein. Proteome. Metabolites. Metabolome
 DNA Genome Complexity RNA Transcriptome Systems Biology Linking all the components of a cell in a quantitative and temporal manner Protein Proteome Metabolites Metabolome Where are the functional elements?
DNA Genome Complexity RNA Transcriptome Systems Biology Linking all the components of a cell in a quantitative and temporal manner Protein Proteome Metabolites Metabolome Where are the functional elements?
The Biochemistry of apoptosis
 The Biochemistry of apoptosis 1 1 The apoptosis is composed of multiple biochemical events 2 2 Biochemical, cellular, and molecular events in Apoptosis 1. Membrane blebbing; phosphatidyl serine exposure
The Biochemistry of apoptosis 1 1 The apoptosis is composed of multiple biochemical events 2 2 Biochemical, cellular, and molecular events in Apoptosis 1. Membrane blebbing; phosphatidyl serine exposure
The validity of the diagnosis of inflammatory arthritis in a large population-based primary care database
 Nielen et al. BMC Family Practice 2013, 14:79 RESEARCH ARTICLE Open Access The validity of the diagnosis of inflammatory arthritis in a large population-based primary care database Markus MJ Nielen 1*,
Nielen et al. BMC Family Practice 2013, 14:79 RESEARCH ARTICLE Open Access The validity of the diagnosis of inflammatory arthritis in a large population-based primary care database Markus MJ Nielen 1*,
Membrane Binding by tbid Initiates an Ordered Series of Events Culminating in Membrane Permeabilization by Bax
 Membrane Binding by tbid Initiates an Ordered Series of Events Culminating in Membrane Permeabilization by Bax Jonathan F. Lovell, 1 Lieven P. Billen, 1 Scott Bindner, 1 Aisha Shamas-Din, 1 Cecile Fradin,
Membrane Binding by tbid Initiates an Ordered Series of Events Culminating in Membrane Permeabilization by Bax Jonathan F. Lovell, 1 Lieven P. Billen, 1 Scott Bindner, 1 Aisha Shamas-Din, 1 Cecile Fradin,
The functional investigation of the interaction between TATA-associated factor 3 (TAF3) and p53 protein
 THESIS BOOK The functional investigation of the interaction between TATA-associated factor 3 (TAF3) and p53 protein Orsolya Buzás-Bereczki Supervisors: Dr. Éva Bálint Dr. Imre Miklós Boros University of
THESIS BOOK The functional investigation of the interaction between TATA-associated factor 3 (TAF3) and p53 protein Orsolya Buzás-Bereczki Supervisors: Dr. Éva Bálint Dr. Imre Miklós Boros University of
MCB*4010 Midterm Exam / Winter 2008
 MCB*4010 Midterm Exam / Winter 2008 Name: ID: Instructions: Answer all 4 questions. The number of marks for each question indicates how many points you need to provide. Write your answers in point form,
MCB*4010 Midterm Exam / Winter 2008 Name: ID: Instructions: Answer all 4 questions. The number of marks for each question indicates how many points you need to provide. Write your answers in point form,
a surface permeabilized
 a surface permeabilized RAW 64.7 P388D1 J774 b CD11b + Ly-6G - Blood Monocytes WT Supplementary Figure 1. Cell surface expression on macrophages and DCs. (a) RAW64.7, P388D1, and J774 cells were subjected
a surface permeabilized RAW 64.7 P388D1 J774 b CD11b + Ly-6G - Blood Monocytes WT Supplementary Figure 1. Cell surface expression on macrophages and DCs. (a) RAW64.7, P388D1, and J774 cells were subjected
Cell Communication. Chapter 11. Biology Eighth Edition Neil Campbell and Jane Reece. PowerPoint Lecture Presentations for
 Chapter 11 Cell Communication PowerPoint Lecture Presentations for Biology Eighth Edition Neil Campbell and Jane Reece Lectures by Chris Romero, updated by Erin Barley with contributions from Joan Sharp
Chapter 11 Cell Communication PowerPoint Lecture Presentations for Biology Eighth Edition Neil Campbell and Jane Reece Lectures by Chris Romero, updated by Erin Barley with contributions from Joan Sharp
Interacts Directly with and Negatively Regulates Pro-apoptotic Bax*
 THE JOURNAL OF BIOLOGICAL CHEMISTRY Vol. 278, No. 3, Issue of January 17, pp. 2058 2065, 2003 2003 by The American Society for Biochemistry and Molecular Biology, Inc. Printed in U.S.A. 14-3-3 Interacts
THE JOURNAL OF BIOLOGICAL CHEMISTRY Vol. 278, No. 3, Issue of January 17, pp. 2058 2065, 2003 2003 by The American Society for Biochemistry and Molecular Biology, Inc. Printed in U.S.A. 14-3-3 Interacts
T cell maturation. T-cell Maturation. What allows T cell maturation?
 T-cell Maturation What allows T cell maturation? Direct contact with thymic epithelial cells Influence of thymic hormones Growth factors (cytokines, CSF) T cell maturation T cell progenitor DN DP SP 2ry
T-cell Maturation What allows T cell maturation? Direct contact with thymic epithelial cells Influence of thymic hormones Growth factors (cytokines, CSF) T cell maturation T cell progenitor DN DP SP 2ry
T H E J O U R N A L O F C E L L B I O L O G Y
 T H E J O U R N A L O F C E L L B I O L O G Y Supplemental material Krenn et al., http://www.jcb.org/cgi/content/full/jcb.201110013/dc1 Figure S1. Levels of expressed proteins and demonstration that C-terminal
T H E J O U R N A L O F C E L L B I O L O G Y Supplemental material Krenn et al., http://www.jcb.org/cgi/content/full/jcb.201110013/dc1 Figure S1. Levels of expressed proteins and demonstration that C-terminal
