Update on the serrated pathway to colorectal carcinoma
|
|
|
- Raymond Carr
- 5 years ago
- Views:
Transcription
1 Human Pathology (2010) xx, xxx xxx Progress in pathology Update on the serrated pathway to colorectal carcinoma Dale C. Snover MD Department of Pathology, Fairview Southdale Hospital, Hibbing, MN 55746, USA Department of Laboratory Medicine and Pathology, The University of Minnesota Medical School, Edina, MN 55435, USA Received 7 May 2010; revised 11 June 2010; accepted 16 June 2010 Keywords: Colon adenocarcinoma; Serrated pathway; Sessile serrated adenoma; Traditional serrated adenoma; Hyperplastic polyp Summary Adenocarcinoma of the large intestine can no longer be considered one disease but rather a family of diseases with different precursor lesions, different molecular pathways, and different end-stage carcinomas with varying prognoses. Approximately 60% of colorectal carcinomas arise from conventional adenomas via the suppressor pathway leading to microsatellite stable carcinomas. These carcinomas represent the pathway that has been the target of screening and prevention programs to date. However, approximately 35% of carcinomas arise along the serrated pathway developing from the precursor lesion known as the sessile serrated adenoma (also referred to as the sessile serrated polyp). Sessile serrated adenomas/polyps lead to carcinomas with extensive CpG island promoter methylation (CpG island methylated phenotype positive carcinomas), which can be either microsatellite instable high or microsatellite stable. The remaining 5% of carcinomas arise from conventional adenomas in patients with germ line mutations of mismatch repair genes (Lynch syndrome), leading to CpG island methylated phenotype negative microsatellite instable carcinomas. Carcinomas arising from sessile serrated adenomas/polyps are not prevented by removing conventional adenomas and hence may be missed in routine screening programs. In addition, a subset of these lesions may potentially progress rapidly to carcinoma; hence, it is likely that these lesions will require a different screening strategy from that used for conventional adenomas. This article reviews the various pathways to colorectal carcinoma with emphasis on the serrated pathway and evaluates the implications of this pathway for colorectal carcinomas screening programs Published by Elsevier Inc. 1. Introduction Colorectal carcinoma is a major health issue in the United States, representing the second most commonly fatal malignancy after lung. Although it is often assumed that almost all colorectal carcinomas arise from conventional adenomas via the suppressor pathway initiated with a mutation of the APC gene (the Fearon-Vogelstein model), it is now clear that this pathway accounts for only approximately 60% of colon carcinoma [1,2]. Most of the Department of Pathology, Fairview Southdale Hospital. address: snoverd@umn.edu. remaining 40% is accounted for by the more recently described serrated pathway leading to CpG island methylated phenotype (CIMP+) carcinoma (approximately 35%), with the remaining 5% arising via the mutator pathway in Lynch syndrome [2]. Trying to understand the multiplicity of pathways is confused by a plethora of overlapping ways to describe these pathways and cancers. Pathways have been defined by the presumed initiating factor (ie, suppressor [chromosome instability] versus mutator [microsatellite instability] pathways) and by the presumed precursor lesions (ie, conventional adenoma-carcinoma sequence versus the serrated pathway). A commonly utilized subdivision into microsatellite stable (MSS) carcinomas (which represent both suppressor pathway carcinomas arising either sporadically or /$ see front matter 2010 Published by Elsevier Inc. doi: /j.humpath
2 2 D. C. Snover in familial adenomatous polyposis as well as approximately 60% of CIMP+ carcinomas arising along the serrated pathway) and microsatellite instable high (MSI-H) carcinomas (by convention usually referred to as MSI carcinoma without the high or -H descriptor), which represent all carcinomas in Lynch syndrome arising via the mutator pathway, as well as approximately 40% of sporadic CIMP+ carcinomas, is useful for detecting patients with Lynch syndrome but still represents a phenotypic end stage of differing pathways. All of these different descriptions overlap to some degree. The conventional APC initiated adenomacarcinoma pathway leads to FAP and sporadic CIMP colon cancers arising via the suppressor pathway as well as cancers in Lynch syndrome arising along the mutator pathway. The alternate serrated pathway leads to CIMP+ carcinomas either by a mutator pathway (leading to sporadic MSI+CIMP+ carcinomas) or a pathway (details of which are still unknown) that leads to MSS CIMP+ carcinomas. In addition, there remain probable other pathways to colorectal carcinomas that have not yet been well worked out, including MSI-low (MIS-L) carcinomas and carcinomas associated with mutation of the MYH gene, both of which may have some relationship to the serrated pathway. These various pathways and their overlaps are summarized in Fig. 1. Current thinking regarding colorectal pathways was recently summarized in review by the late Jeremy Jass, and this article is recommended reading for anyone wishing to understand the multiple pathways to colorectal carcinoma [2]. Because most screening and intervention programs for colon carcinoma have been directed toward the elimination of conventional adenomas, it would appear that at best a 65% reduction in colorectal carcinoma can be expected by our current approach. Eliminating the other 35% will require a better understanding of the serrated pathway and precursor lesions of the serrated pathway, mainly the lesion known as the sessile serrated adenoma (SSA; AKA sessile serrated polyp) and the less common traditional serrated adenoma (TSA). This review will focus on these lesions and current controversies related to them. 2. Molecular mechanisms of carcinomas development via the serrated pathway Developing a management scheme for dealing with serrated lesions requires an understanding of the underlying mechanism of molecular carcinogenesis. The basic Fig. 1 Schematic represent several overlapping ways to describe the development of colorectal carcinoma. The red circles represent mechanisms based on putative initiating factors (the suppressor and mutator pathways). The blue circles represent mechanisms based on the precursor lesion (the conventional adenoma-carcinoma sequence and serrated pathways). The yellow circles represent currently poorly characterized pathways. (Key: FAP = familial adenomatous polyposis; CIMP+ = CpG island methylator phenotype positive; CIMP- = CpG island methylator phenotype negative; MSI-H = high degree of microsatellite instability; MSI-L = low degree of microsatellite instability; MSS = microsatellite stable; TSA = traditional serrated adenoma.)
3 Serrated pathway to colorectal carcinoma mechanism for the suppressor pathway to colon carcinoma begins with a mutation of the APC tumor suppressor gene, which allows unregulated proliferation of epithelial cells with the formation of the a lesion conventionally known as an adenoma (these APC-related adenomas will be referred to as conventional adenomas throughout this review; they are commonly subdivided into tubular, tubulovillous, and villous types, and with these designations, the term conventional is understood and need not be applied in daily practice). This proliferative population then undergoes additional mutations, presumably in a relatively random order, with the eventual progression of a small subset of conventional adenomas to carcinoma with intermediate stages showing increasing degrees of dysplasia. With the exception of conventional adenomas arising in Lynch syndrome, which appear to progress rapidly to carcinoma, the most conventional adenomas never progress and those that do require many years (probably 10 or more) to develop into malignancy. Hence, screening programs periodically removing conventional adenomas can be very successful in preventing MSS carcinomas arising via the suppressor pathway. The mechanism of the most common carcinomas arising via the serrated pathway appears to begin with an activating mutation of the BRAF gene, a gene which when mutated inhibits normal apoptosis of colonic epithelial cells. Lesions bearing the BRAF mutation develop into serrated lesions that are mainly either microvesicular hyperplastic polyps (MVHP) or sessile serrated adenomas/polyps (SSA/P, see terminology, below) [3]. These lesions are prone to methylation of the CpG island promoter regions resulting in the epigenetic silencing of a number of genes. It is presumed that the specific genes silenced may be random events. The specific gene silenced determines what happens with a lesion. The most well characterized epigenetic silencing in these lesions is that of the hmlh1 gene, which is the gene silenced in sporadic MSI carcinomas. The hmlh1 gene is one of the mismatch repair genes, and when silencing occurs, these lesions become MSI and are prone to the development of additional mutations at a rapid rate, similar to what happens to conventional adenomas in patients with Lynch syndrome. Although in most (although unfortunately not all) lesions it appears that methylation of hmlh1 3 Fig. 2 Representation of the serrated pathway to MSI-H carcinoma. This sequential pathway involves slow and rapid steps. The origin of SSA/P remains debatable. It is possible that SSA/P arises directly from normal mucosa or SSA/P might develop from a preexisting MVHP; hence, the arrows for these steps are dotted.
4 4 D. C. Snover is a late event, occurring in all likelihood many years after the lesion initially develops, once hmlh1 is inactivated, there is rapid development of cytological dysplasia (see terminology, below) followed by potentially rapid development of frank malignant transformation. The rate of progression of these lesions has an important impact on screening strategies, discussed below. Carcinomas arising via this pathway are CIMP+MSI carcinomas. The specific molecular events leading to the development of CIMP+MSS carcinomas are not nearly as well characterized as that for CIMP+MSI carcinomas but presumably involve epigenetic silencing of other genes leading to carcinoma via as yet undefined pathways. A simplified diagram of the serrated pathway to CIMP+MSI carcinoma is shown in Fig. 2. There may be several more serrated pathways not related to the BRAF gene mutation. Several types of serrated polyps, including goblet cell rich hyperplastic polyps (GCHP) and TSA, are mutated at the Kras gene rather than the BRAF gene (4,5). There is currently no known direct role of GCHP in carcinogenesis; however, it is clear that TSA may develop into carcinoma, although there has been some controversy as to the type of carcinoma. It has been suggested that MSI-L carcinomas may develop in these lesions after methylation of the MGMT gene or possibly secondary to partial methylation of hmlh1. Finally, there is a recent report of multiple serrated polyps in patients with MYH polyposis associated with Kras mutations; however, this remains to be confirmed and the molecular mechanisms evaluated [6]. 3. Precursor lesions of the serrated pathway As recently as 2003, all of the various serrated lesions were considered hyperplastic or metaplastic polyps, and their malignant potential was not well recognized, but it is now generally recognized that these lesions represent a complex family of lesions differing in their specific underlying mutation and degree of methylation [3-5,7-11]. As yet unknown factors lead to alterations in the location of the proliferative zone as well as differences in the anchoring of crypts within the lesions, leading to a wide range of histological appearances [10]. It is mainly architectural alterations resulting from these abnormalities of proliferation that allow distinction of the different categories of polyps. A summary of the current classification of serrated polyps Table 1 Classification of serrated lesions of the large intestine HP Microvesicular type (MVHP) Goblet cell rich type (GCHP) Mucin poor type (MPHP) SSA/P Without cytological dysplasia With cytological dysplasia TSA Without conventional dysplasia With conventional dysplasia (based on the fourth edition of the World Health Organization [WHO] blue book to be published in 2010 [11]) is shown in Table 1. A diagrammatic representation of the proliferative zone abnormalities and resultant architectural changes of the various serrated lesions is shown in Fig. 3. Although there is now general consensus on the importance of the serrated lesions in the development of carcinomas, and the general categories of lesions are accepted, challenges remain. Included among these are issues related to terminology, diagnostic criteria, and natural history, which affects management decisions. 4. Terminology and diagnostic criteria Details of the histological appearance of these lesions have been reviewed extensively elsewhere [7-11] and will not be repeated here except to provide a brief review and introduction into current issues. Although the terminology is not perfect because these lesions are not truly hyperplastic or metaplastic, the term hyperplastic polyp (HP) has been retained for the most indolent form of serrated lesions [11]. These lesions are characterized by variably prominent serrations occurring in crypts that are generally straight and demonstrate more or less normal localization of the proliferative zone to the base of the crypts (Figs. 3A and 4). HPs are subdivided based on the mucin content, with those demonstrating exclusively goblet cell mucin being termed GCHP, those with small droplet (microvesicular) mucin either alone or intermixed with goblet cells being called MVHP, and those with no mucin being designated mucin poor HPs (MPHP). GCHPs Fig. 3 Graphic representation of the putative sequence for the development of serrated lesions based on the location of the proliferative zone. All 3 lesions begin with normal mucosa in which the proliferative zone (indicated by the thick line) is located at the lower third of the crypt, and cells mature toward the lumen (arrow). The double thin line represents the muscularis mucosae. A, HP. The proliferative zone remains in the lower portion of the crypt, occupying up to half of the length of the crypt. Cells continue to mature toward the surface (arrow), but because of delayed apoptosis, the cells tend to pile up, leading to serrations. B, SSA/P. The proliferative zone is located at various locations throughout the crypt length. This allows maturing cells to move toward the lumen but also toward the base of the crypt (arrow), which leads to distorted and dilated crypts since the muscularis mucosae blocks the downward growth of the crypts. C, TSA. The proliferative zone is also variably located, but small ectopic crypts develop from the side of the original crypt. These miniature crypts create a complex growth pattern. (adapted from Reference [10]).
5 Serrated pathway to colorectal carcinoma 5
6 6 D. C. Snover Fig. 4 Microvesicular hyperplastic polyp. Note that the crypts are relatively straight with narrow bases (40). Also, see Fig. 3A. are found almost exclusively in the left colon and commonly demonstrate Kras mutations, whereas MVHP, which is still predominantly left-sided but with more rightsided lesions than GCHP, tends to be BRAF mutated [4]. MPHP appears to represent a damaged MVHP with reactive change, but this category is rare and has not been well studied. It has been suggested that MVHP may act as a precursor to the more advanced and clearly premalignant SSA/P, in large part because they share the BRAF mutation and a propensity to methylation, although this has not been proven and perhaps is of more theoretical than practical significance. Very rarely will one encounter a MVHP that demonstrates cytological dysplasia resembling conventional adenoma, and hence, it is possible that MVHP may occasionally become methylated at a gene critical for carcinogenesis, as occurs in SSA/P, and hence may progress to malignancy without an intervening SSA/P. Perhaps the most persistent and perplexing issue regarding serrated lesions is the fact that there remains no consensus on the best terminology for the lesion known variably in the literature as sessile serrated adenoma (SSA), sessile serrated polyp (SSP), or serrated polyp with abnormal proliferation. The term SSA was the first term used for this lesion, and a majority of the current literature is written using the term SSA; however, there is a second large group who prefers SSP. The term serrated polyp with abnormal proliferation is rapidly losing favor. The rationale for the use of polyp rather than adenoma for this lesion revolves around a concern that use of adenoma will cause confusion with conventional adenomas arising via the APC mutation pathway, despite the fact that most authors in the field accept that these lesions are functionally premalignant and hence neoplastic. In the practice of this author, who has used the term SSA since 1996, confusion with conventional adenomas has not been an issue with our gastroenterologists, who seem to have no difficulty understanding the difference between SSA, tubular adenoma, tubulovillous adenoma, and villous adenoma once they are familiar with the concept. Many authors believe that SSA is the best term because it reflects the premalignant potential of this lesion in the serrated pathway and is likely to lead to more appropriate management that other more noncommittal terms. Nonetheless, consensus has not been reached, and the upcoming fourth edition of the WHO classification of tumors of the digestive trace will reflect that at the current time either term is acceptable as long as it is recognized that SSA and SSP are referring to the same lesion [11]. For clarity sake, SSA/P will be used throughout this article. Unfortunately, there are still some pathologists who continue to use the term hyperplastic polyp or variant hyperplastic polyp for these lesions, a most unfortunate fact given the considerable management differences between HP and SSA/Ps. The use of the hyperplastic polyp for these lesions should be avoided. SSA/P is characterized mechanistically by movement of the proliferative zone away from its usual location in the base of the crypts, resulting in maturation, which may develop toward the base of the crypts leading to distortion of the crypt architecture, commonly with dilated, L, inverted T, or anchorshaped crypts with mature cells where the proliferative zone normally is located (as evidenced by mucinous differentiation as well as marking for cytokeratin 20 (Figs. 3B and 5) [10]). The crypts still retain their attachment or orientation toward to the muscularis mucosae, however, and are arranged perpendicular to the muscularis mucosae. SSA/P in its earliest stage does not manifest dysplasia resembling conventional adenoma, although with progression toward carcinomas, such dysplasia develops. There has been variation regarding the best term for lesions that combine the features of SSA/P with areas of frank cytological dysplasia that resemble conventional adenomas (Fig. 6). These lesions have in most of the older Fig. 5 SSA/P without cytological dysplasia. Compared to the HP, the overall architecture is distorted with variably shaped crypts and mature cells at the base of the crypts (40). Also, see Fig. 3B.
7 Serrated pathway to colorectal carcinoma Fig. 6 SSA/P with cytological dysplasia. This lesion has characteristics of an SSA/P on the right, but on the left, the epithelium is cytologically dysplastic demonstrating hyperchromatic elongated nuclei with pseudostratification. This may indicate conversion to a MSI state. There is an underlying invasive adenocarcinoma as well (40). literature been called mixed HP-TA, and with the recognition of SSA/P, it turned out that the serrated portion of most of these mixed lesions was in reality SSA/P, leading to the use of the term mixed SSA/P-TA for these lesions [7]. However, it is currently felt that this term is misleading because the cytologically dysplastic part of these polyps is not a conventional TA from a molecular perspective and probably has a more aggressive course than conventional TA. As noted above, conventional TA is a lesion that starts with an APC mutation, acquires addition mutations over time, and eventually, in a small number of cases and generally over the course of 10 years or more, becomes malignant. SSA/P with cytological dysplasia, on the other hand, in some cases (ie, in the case of lesions developing into MSI carcinomas) represents the development of microsatellite instability in the lesion and hence is creating a lesion with a propensity to develop into carcinoma with high frequency and rapidity (see Fig. 2 and discussion above). These lesions are not APC mutated and probably require considerably more aggressive follow-up than conventional adenomas. Hence, SSA/P with cytological dysplasia is the preferred term for this lesion, and the term mixed SSA/P-TA or HP-TA is no longer considered acceptable. The other term that has had some controversy is that of the traditional serrated adenoma (TSA). This lesion is one of several forms of serrated lesions that fit the definition of serrated adenoma proposed in the seminal article by Longacre and Fenoglio-Prieser in 1990 [12]. The definition provided initially for serrated adenoma was that a serrated adenoma was a lesion with dysplasia (and hence adenoma by conventional criteria in use at the time) but which showed an overall serrated configuration. Unfortunately, this definition is overly broad and can be used to describe what are now known to be 3 or possibly 4 different lesions (SSA/P, SSA/P with cytological dysplasia, TSA, and conventional APC adenomas with a serrated growth pattern). Recognizing this, in 2003, it was suggested that the most unique of this group, and the one member of this group which did not have a better name, should be recognized as a separate entity, and the term traditional serrated adenoma was recommended (because it was thought that this lesion was the one member of this group that was most easily recognized by most pathologists, and the one that at the time was probably most commonly being called serrated adenoma, in large part because at the time SSA/P was still being called HP by most pathologists, and SSA/P with cytological dysplasia was being called mixed HP-TA.) [3]. It is of some interest along these lines that a recent reproducibility study identified this lesion as the one with the best kappa statistic of the various serrated lesions, supporting our contention that most pathologists already know how to recognize this lesion [13]. There does continue to be some variation in the diagnosis of TSA, however, in large part because of recent suggestions for expansion of the diagnostic criteria [10]. The original definition of TSA was a very simplistic one, with the most notable feature being the presence of a peculiar type of atypical cell characterized by relatively abundant eosinophilic cytoplasm resembling that of an oncocyte, with elongated nuclei (typically not hyperchromatic) often located in the mid portion of the cell and with little or no pseudostratification and with little proliferative activity as determined by mitotic count or Ki67 immunoreactivity [3,7,10]. This cell was considered by many as being dysplastic and therefore was often equated with the dysplasia of conventional adenomas. Given the lack of proliferative activity (as well as the lack of APC mutations) in these cells, they are not likely to have the same neoplastic potential as conventional dysplasia and are better considered Fig. 7 TSA. This lesion is characterized by an overall somewhat villiform configuration with a very complex overall architecture (40). Also, see Fig. 3C. 7
8 8 D. C. Snover Table 2 Proposal for a semimolecular classification of colorectal polyps Traditional terminology (pre-2003) Recently revised terminology (post-2003) Proposed semimolecular terminology Adenoma Conventional adenoma APC-related neoplastic polyps Tubular adenoma Tubular adenoma Nonserrated neoplastic polyp without villous component Tubulovillous adenoma Tubulovillous adenomas Nonserrated neoplastic polyp with villous component Villous adenoma Villous adenoma Serrated adenoma Hyperplastic polyp Serrated adenomas CIMP-adenomas, BRAF type Hyperplastic polyp SSA/P without cytological dysplasia Sessile serrated neoplastic polyp without cytological dysplasia SSA/P with cytological dysplasia Sessile serrated neoplastic polyp with cytological dysplasia CIMP-adenomas, KRAS type TSA without conventional dysplasia Filiform serrated neoplastic polyp without conventional dysplasia TSA with conventional dysplasia Filiform serrated neoplastic polyp with conventional dysplasia Hyperplastic polyps Hyperplastic polyp, BRAF type Microvesicular HP Microvesicular HP Hyperplastic polyp, Kras type GCHP GCHP Hyperplastic polyp, other Mucin poor HP Mucin poor HP a metaplastic or possibly senescent cell. Although this cell type was original considered a sine qua non for the diagnosis of TSA, it is now apparent that this cell type can be seen in a variety of circumstances including in small areas of otherwise typical SSA/P, in some conventional adenomas (APC-related) and sometimes even in reactive mucosa. An immunohistochemical analysis of proliferation and maturation in serrated lesions has pointed out that a more likely diagnostic abnormality in TSA is the presence of small ectopic crypts created by apparent loss of anchoring of the Fig. 8 TSA with conventional dysplasia. This TSA has cytological dysplasia similar to that seen in conventional adenomas in the right portion of the photo. In this particular case, the dysplasia is high grade with a cribriform pattern of growth. There was invasive carcinoma elsewhere in this polyp (40). crypt bases to the muscularis mucosae [10] (Figs. 3C and 7). These ectopic crypts demonstrated strong CK20 staining in the luminal portions and Ki67 staining in the deeper aspects, hence recapitulating small crypts except that the base of the crypts was free floating in the lamina propria of villi rather than sitting on (or being anchored to ) the muscularis mucosae. We now feel that these ectopic crypts are probably the best defining feature of TSA, although most TSAs will also have the atypical eosinophilic cell noted above. These ectopic crypts are not present in all areas of all TSAs but should be present at least focally. Often TSA will have large areas with the metaplastic eosinophilic cells described above, but using these diagnostic criteria, a TSA can also be composed mainly of goblet cells. Whether these different cell types have any molecular or prognostic implications is also not known. The lesion described as filiform serrated adenoma is probably synonymous with TSA or represents a subset of TSA and may provide less confusing terminology (see Table 2) [14]. Although eosinophilic metaplastic cells often make up a considerable portion of TSAs, atypical cells with many of the morphological attributes of conventional dysplasia (ie, hyperchromasia, pseudostratification and increased proliferative activity) do occur in TSA, probably as part of conversion toward malignancy (Fig. 8). The molecular characteristics of these cells (eg, mutation and methylation status) have not been well characterized, however, and there has been essentially no discussion of the terminology of this converted lesion in the literature. Trying to be analogous to SSA/P, it would probably be appropriate to refer to these lesions as TSA with conventional cytological dysplasia, despite the fact that this is somewhat cumbersome. The specific mechanism of carcinogenesis in TSA is currently not well known and is discussed briefly above under
9 Serrated pathway to colorectal carcinoma molecular mechanisms of carcinomas development via the serrated pathway. 5. The role of SSA/P in carcinogenesis and its effect on management of these lesions As noted in the introduction, it is now generally recognized that colon cancer comprises a family of diseases with various molecular pathways. There is now no question that SSA/P is the immediate precursor lesion to sporadic CIMP+MSI carcinomas. SSA/P is likely the precursor lesion to CIMP+MSS carcinomas as well. Combined, CIMP+MSI and CIMP+MSS carcinomas account for approximately 35% of all colorectal carcinoma; hence, these are very important lesions to recognize and deal with if one wants to reduce the risk of colon carcinoma through screening. Unfortunately, we are still somewhat impaired in our understanding of the natural history of serrated lesions because of a lack of longitudinal studies, and current recommendations for management are based in large part on our understanding of the molecular mechanisms of the pathway and on observational studies including recent data on the frequency of serrated pathway associated carcinomas presenting as interval cancers in screening programs. One important difference in screening for serrated lesions (as opposed to screening for conventional adenomas) results from the fact that the CIMP+MSI pathway may be a rapidly progressive pathway, much more rapid than carcinogenesis via the conventional APC pathway, as described above. Recent analysis of interval carcinomas occurring in screening populations has indicated that these cancers are disproportionally MSI [15]. One possible reason for this is that some of these lesions may become malignant very rapidly, with their entire lifespan fitting in the 5 or 10 years between screening examinations. Another potential reason that should not be overlooked, however, is that SSA/P is a very subtle lesion and may be difficult to identify on endoscopic examination, especially for an endoscopist who is not experienced with recognizing the subtle changes that might indicate the presence of a lesion (especially adherent stool or mucin that must be washed off to visualize the lesion). Therefore, interval MSI cancers might also occur because SSA/Ps are being missed. It turns out that interval cancers are not only disproportionally CIMP+MSI, but they are also disproportionally CIMP +MSS [16]. Because there is no a priori reason to suspect that CIMP+MSS development has the same potential rapid progression as seen with CIMP+MSI carcinoma, this finding would bolster the theory that some of the issue related to interval cancers involves failure to identify the underlying lesion. Regardless of the reason, this apparent enhancement of interval cancers with CIMP+ carcinomas arising via the serrated pathway has significant implications on the appropriate interval for rescreening patients with a known SSA/P. Our past recommendations had suggested that the most worrisome lesions of this group, and the lesion with the most pressing need for close surveillance, were the SSA/Ps with cytological dysplasia, since these lesions have the potential to rapidly progress to carcinoma [7,8]. We still maintain that any lesion with this histology should be removed entirely by whatever means necessary and that the patient undergo repeat endoscopy no longer than a year after initial excision (if the endoscopist is comfortable that the lesion has been entirely removed) or sooner if there is any question about adequacy of excision. TSAs, either with or without changes of conventional dysplasia, are probably best treated as conventional adenomas because they do not lead to the development of MSI carcinomas, and hence, there is no reason to suspect that they might be more aggressive than other lesions. In addition, because they tend to occur in the rectosigmoid region and are usually well-circumscribed protruding lesions, surveillance and removal tend to be easier. The real management issue is for SSA/P without cytological dysplasia because these are the most common lesions and are the ones that are most difficult to visualize and remove. Our original recommendations were relatively leisurely regarding this group, with recommendations to remove the lesion if possible or, if the lesion was not easily resected via the endoscopy, to do surveillance at annual intervals to liberally biopsy the lesion and look for cytological dysplasia that would then accelerate the need to remove the lesion [7,8]. If the lesion was completely removed, then we suggested reversion to standard adenoma surveillance intervals. Given the recent data regarding CIMP+ and interval cancers, however, this may not be an adequate strategy. Personal observations by myself and others (Kenneth Batts, MD, personal communications) would indicate that often patients with one SSA/P will harbor others, either synchronously or metachronously. This seems especially true for large lesions found in the right colon. When the criteria for serrated polyposis (formerly known as hyperplastic polyposis ) are met ([1] at least 5 serrated polyps proximal to the sigmoid colon with 2 or more of them larger than 10 mm, [2] any number of serrated polyps proximal to the sigmoid colon in an individual who has a first-degree relative with serrated polyposis, or [3] more than 20 serrated polyps of any size, but distributed throughout the colon ), the situation is more defined, and close surveillance with possible colectomy is considered [11]. However, the criteria for serrated polyposis are at this point still entirely arbitrary, and therefore, it is possible if not probable that the finding of multiple SSA/Ps of any size or location may be adequate to recommend more frequent surveillance. As noted above, the issue with SSA/P may be 2-fold potential rapid progression to carcinoma and incomplete removal. To prevent interval cancers, therefore, shortened interval surveillance may be appropriate. 9
10 10 D. C. Snover Reasonable risk factors suggesting the need for short interval reendoscopy would include size of lesions (lesions smaller than 1 cm may not be as important), location of lesions (right sided lesions may be more important), and age (older patients being more prone to SSA/P as well as to sporadic MSI carcinomas), in addition to number of lesions. A reasonable strategy, and one we are currently recommending, would be to repeat surveillance at 1 year for anyone with 2 or more completely removed SSA/Ps greater than 1 cm and determine future screening based on these findings. If there is any significant risk that a lesion was not completely removed, repeat endoscopy at a shorter interval may well be indicated. If additional SSA/Ps are identified at 1 year, annual surveillance (at least until there is a negative colonoscopy) would seem reasonable. If screening at 1 year is clear of lesions, then extension of the interval to 2 to 3 years would seem appropriate. 6. Proposed solutions to ongoing issues As noted above, perhaps the more important issues regarding serrated lesions involve appropriate management, although terminology remains an issue as well. The management issue is amenable to further clinicopathological studies focusing on natural history, not only for the more common SSA/P but also for TSA, since we know the least about that lesion. Additional molecular characterization of specific genetic changes resulting in CIMP+MSS carcinomas and for carcinomas arising in TSA will also be valuable in this regard, since this information may help determine the best surveillance and prevention strategies. Regarding the terminology issues, at the current time, there appears to be a stalemate between the proponents of SSA and those for SSP. Much of this problem exists because of the traditional use of the term adenoma not as a generic family of lesions (ie, polyps with premalignant potential) but rather for a specific subset of adenomas, that is, adenomas predominantly arising along the suppressor pathway initiated with APC mutations. Perhaps it is time for gastrointestinal pathologists to consider reclassification of adenomas on a semimolecular basis, realizing that it is not practical to perform molecular testing on all lesions removed as adenomas. We could, however, alter our terminology to indicate the likely molecular aspects of adenomas, something that would point out the potential molecular difference and would at a minimum have an educational effect if not a direct management effect. It might also be advisable to eliminate the term adenoma from terminology given the long history of using this word only for precursor lesions to non-cimp carcinomas (ie, APCrelated carcinomas arising via the suppressor pathway and MSI carcinomas arising via MMR mutations in Lynch syndrome); however, finding a convenient shorthand substitute will be difficult. Nonetheless, as a starting point, a suggestion for a semimolecular classification is proposed in Table 2. References [1] Fearon ER, Vogelstein B. A genetic model for colorectal tumorigenesis. Cell 1990;61: [2] Jass JR. Classification of colorectal cancer based on correlation of clinical, morphological and molecular features. Histopathology 2007;50: [3] Torlakovic E, Skovland E, Snover DC, et al. Morphologic reappraisal of serrated colorectal polyps. Am J Surg Pathol 2003;27: [4] O'Brien MJ, Yang S, Mack C, et al. Comparison of microsatellite instability, CpG island methylation phenotype, BRAF and KRAS status in serrated polyps and traditional adenomas indicates separate pathways to distinct colorectal carcinoma end points. Am J Surg Pathol 2006;30: [5] Yachida S, Mudali S, Martin SA, Montgomery EA, Iacobuzio-Donahue CA. Beta-catenin nuclear labeling is a common feature of sessile serrated adenomas and correlates with early neoplastic progression after BRAF activation. Am J Surg Pathol 2009;33: [6] Boparai KS, Dekker E, Van Eeden S, et al. Hyperplastic polyps and sessile serrated adenomas as a phenotypic expression of MYHassociated polyposis. Gastroenterology 2008;135: [7] Snover DC, Jass JR, Fenoglio-Preiser C, Batts KP. Serrated polyps of the large intestine: a morphologic and molecular review of an evolving concept. Am J Clin Pathol 2005;124: [8] Snover DC. Serrated polyps of the large intestine. Sem Diag Pathol 2005;22: [9] East JE, Saunders BP, Jass JR. Sporadic and syndromic hyperplastic polyps and serrated adenomas of the colon: classification, molecular genetics, natural history, and clinical management. Gastroenterol Clin North Am 2008;37: [10] Torlakovic EE, Gomez JD, Driman DK, et al. Sessile serrated adenoma (SSA) vs. traditional serrated adenoma (TSA). Am J Surg Pathol 2008;32:21-9. [11] Snover DC, Ahnen DJ, Burt RW, Odze RD. Serrated polyps of the colon and rectum and serrated ( hyperplastic ) polyposis. In: Bozman FT, Carneiro F, Hruban RH, Theise N, editors. WHO classification of tumours. Pathology and genetics. Tumours of the digestive system. Berlin: Springer-Verlag (4th ed. [in press]). [12] Longacre TA, Fenoglio-Preiser CF. Mixed hyperplastic adenomatous polyps/serrated adenomas. A distinct form of colorectal neoplasia. Am J Surg Pathol 1990;14: [13] Farris AB, Misdraji J, Srivastava A, et al. Sessile serrated adenoma: challenging discrimination from other serrated colonic polyps. Am J Surg Pathol 2008;32:30-5. [14] Yantiss RK, Oh KY, Chen YT, et al. Filiform serrated adenomas: a clinicopathologic and immunophenotypic study of 18 cases. Am J Surg Pathol 2007;31: [15] Sawhney MS, Farrar WD, Gudiseva S, et al. Microsatellite instability in interval colon cancers. Gastroenterology 2006;131: [16] Arain MA, Sawhney M, Sheikh S, et al. CIMP status of interval colon cancers: another piece to the puzzle. Am J Gastroenterol 2010;105:
Serrated Colorectal Polyps New Challenges to Old Dogma. Kenneth Batts, M.D. Abbott Northwestern Hospital Minneapolis, MN
 Serrated Colorectal Polyps New Challenges to Old Dogma Kenneth Batts, M.D. Abbott Northwestern Hospital Minneapolis, MN A Sneak Preview.... This was in the good old days: Adenomas HPPs Mixed Polyps A Sneak
Serrated Colorectal Polyps New Challenges to Old Dogma Kenneth Batts, M.D. Abbott Northwestern Hospital Minneapolis, MN A Sneak Preview.... This was in the good old days: Adenomas HPPs Mixed Polyps A Sneak
A WHO update on Serrated Polyps
 A WHO update on Serrated Polyps Arzu Ensari, MD, PhD Department of Pathology Ankara University Medical School Am J Gastroenterol. 2010 Nov 2. [Epub ahead of The Clinical Significance of Serrated Polyps.
A WHO update on Serrated Polyps Arzu Ensari, MD, PhD Department of Pathology Ankara University Medical School Am J Gastroenterol. 2010 Nov 2. [Epub ahead of The Clinical Significance of Serrated Polyps.
General Session 7: Controversies in Screening and Surveillance in Colorectal Cancer
 General Session 7: Controversies in Screening and Surveillance in Colorectal Cancer Complexities of Pathological Assessment: Serrated Polyps/Adenomas Carolyn Compton, MD, PhD Professor of Life Sciences,
General Session 7: Controversies in Screening and Surveillance in Colorectal Cancer Complexities of Pathological Assessment: Serrated Polyps/Adenomas Carolyn Compton, MD, PhD Professor of Life Sciences,
Hyperplastische Polyps Innocent bystanders?
 Hyperplastische Polyps Innocent bystanders?? K. Geboes P th l i h O tl dk d Pathologische Ontleedkunde, KULeuven Content Historical Classification Relation Hyperplastic polyps carcinoma The concept cept
Hyperplastische Polyps Innocent bystanders?? K. Geboes P th l i h O tl dk d Pathologische Ontleedkunde, KULeuven Content Historical Classification Relation Hyperplastic polyps carcinoma The concept cept
Serrated Lesions in the Bowel Cancer Screening Programme
 Serrated Lesions in the Bowel Cancer Screening Programme Mark Arends Cambridge & Edinburgh Serrated Lesions of Large Bowel 1. Hyperplastic polyp 2. Serrated adenoma 3. Mixed polyp 4. Sessile serrated lesion
Serrated Lesions in the Bowel Cancer Screening Programme Mark Arends Cambridge & Edinburgh Serrated Lesions of Large Bowel 1. Hyperplastic polyp 2. Serrated adenoma 3. Mixed polyp 4. Sessile serrated lesion
Synchronous and Subsequent Lesions of Serrated Adenomas and Tubular Adenomas of the Colorectum
 Tsumura T, et al 1 Synchronous and Subsequent Lesions of Serrated Adenomas and Tubular Adenomas of the Colorectum T. Tsumura a T. Hiyama d S. Tanaka b M. Yoshihara d K. Arihiro c K. Chayama a Departments
Tsumura T, et al 1 Synchronous and Subsequent Lesions of Serrated Adenomas and Tubular Adenomas of the Colorectum T. Tsumura a T. Hiyama d S. Tanaka b M. Yoshihara d K. Arihiro c K. Chayama a Departments
Sessile Serrated Polyps
 Årsmøtet i Den norske Patologforening 2014 Sessile Serrated Polyps Tor J. Eide Oslo Universitetssykehus The term serrated include a group of lesions with a sawtoothlike appearance of the crypts and the
Årsmøtet i Den norske Patologforening 2014 Sessile Serrated Polyps Tor J. Eide Oslo Universitetssykehus The term serrated include a group of lesions with a sawtoothlike appearance of the crypts and the
Serrated Polyps and a Classification of Colorectal Cancer
 Serrated Polyps and a Classification of Colorectal Cancer Ian Chandler June 2011 Structure Serrated polyps and cancer Molecular biology The Jass classification The familiar but oversimplified Vogelsteingram
Serrated Polyps and a Classification of Colorectal Cancer Ian Chandler June 2011 Structure Serrated polyps and cancer Molecular biology The Jass classification The familiar but oversimplified Vogelsteingram
Serrated Polyps, Part 2: Their Mechanisms and Management Ryan C. Romano, DO
 Polyps, Part 2: Their Mechanisms and Management Ryan C. Romano, DO In the prelude to this article ( Polyps Part I: Their Confusing History) we discussed the evolution of colorectal serrated polyp classification,
Polyps, Part 2: Their Mechanisms and Management Ryan C. Romano, DO In the prelude to this article ( Polyps Part I: Their Confusing History) we discussed the evolution of colorectal serrated polyp classification,
Arzu Ensari, MD, PhD Department of Pathology Ankara University Medical School
 Precursors of Colorectal Carcinoma Arzu Ensari, MD, PhD Department of Pathology Ankara University Medical School Hyperplastic polyp Adenomatous polyp Colorectal carcinoma IBD-associated (1-2%) Sporadic
Precursors of Colorectal Carcinoma Arzu Ensari, MD, PhD Department of Pathology Ankara University Medical School Hyperplastic polyp Adenomatous polyp Colorectal carcinoma IBD-associated (1-2%) Sporadic
Update on Colonic Serrated (and Conventional) Adenomatous Polyps
 Update on Colonic Serrated (and Conventional) Adenomatous Polyps Maui, HI 2018 Robert D. Odze, MD, FRCPC Chief, Division of GI Pathology Professor of Pathology Brigham and Women s Hospital Harvard Medical
Update on Colonic Serrated (and Conventional) Adenomatous Polyps Maui, HI 2018 Robert D. Odze, MD, FRCPC Chief, Division of GI Pathology Professor of Pathology Brigham and Women s Hospital Harvard Medical
Colonic Polyp. Najmeh Aletaha. MD
 Colonic Polyp Najmeh Aletaha. MD 1 Polyps & classification 2 Colorectal cancer risk factors 3 Pathogenesis 4 Surveillance polyp of the colon refers to a protuberance into the lumen above the surrounding
Colonic Polyp Najmeh Aletaha. MD 1 Polyps & classification 2 Colorectal cancer risk factors 3 Pathogenesis 4 Surveillance polyp of the colon refers to a protuberance into the lumen above the surrounding
Pathology of serrated colorectal lesions
 Correspondence to Dr Adrian C Bateman, Department of Cellular Pathology, University Hospital Southampton NHS Foundation Trust, MP002, Level E, South Block, Southampton General Hospital, Tremona Road, Southampton
Correspondence to Dr Adrian C Bateman, Department of Cellular Pathology, University Hospital Southampton NHS Foundation Trust, MP002, Level E, South Block, Southampton General Hospital, Tremona Road, Southampton
Alberta Colorectal Cancer Screening Program (ACRCSP) Post Polypectomy Surveillance Guidelines
 Alberta Colorectal Cancer Screening Program (ACRCSP) Post Polypectomy Surveillance Guidelines June 2013 ACRCSP Post Polypectomy Surveillance Guidelines - 2 TABLE OF CONTENTS Background... 3 Terms, Definitions
Alberta Colorectal Cancer Screening Program (ACRCSP) Post Polypectomy Surveillance Guidelines June 2013 ACRCSP Post Polypectomy Surveillance Guidelines - 2 TABLE OF CONTENTS Background... 3 Terms, Definitions
Beyond the APC era Alternative pathways to CRC. Jeremy R Jass McGill University
 Beyond the APC era Alternative pathways to CRC Jeremy R Jass McGill University Outline Limitations of APC model KRAS and serrated polyps CRC and CpG island methylation Serrated pathway to CRC Fusion pathways
Beyond the APC era Alternative pathways to CRC Jeremy R Jass McGill University Outline Limitations of APC model KRAS and serrated polyps CRC and CpG island methylation Serrated pathway to CRC Fusion pathways
Sessile serrated polyps: Cancer risk and appropriate surveillance
 REVIEW CME CREDIT EDUCATIONAL OBJECTIVE: Readers will understand the role of the recently recognized serrated neoplasia pathway in the development of colorectal cancer ROHIT MAKKAR, MD St. Michael s Hospital,
REVIEW CME CREDIT EDUCATIONAL OBJECTIVE: Readers will understand the role of the recently recognized serrated neoplasia pathway in the development of colorectal cancer ROHIT MAKKAR, MD St. Michael s Hospital,
Inflammatory bowel disease (IBD) patients have a higher risk of
 Serrated Colorectal Lesions in Patients With Inflammatory Bowel Disease Alyssa M. Parian, MD, and Mark G. Lazarev, MD Dr Parian and Dr Lazarev are assistant professors of medicine at Johns Hopkins University
Serrated Colorectal Lesions in Patients With Inflammatory Bowel Disease Alyssa M. Parian, MD, and Mark G. Lazarev, MD Dr Parian and Dr Lazarev are assistant professors of medicine at Johns Hopkins University
Sessile serrated adenomas: prevalence of dysplasia and carcinoma in 2139 patients
 Caris Research Institute, Irving, Texas, USA Correspondence to Richard H Lash, MD, Caris Research Institute, 6655 MacArthur Blvd, Irving, TX 75039, USA; rlash@carisdx.com Accepted 11 March 2010 Published
Caris Research Institute, Irving, Texas, USA Correspondence to Richard H Lash, MD, Caris Research Institute, 6655 MacArthur Blvd, Irving, TX 75039, USA; rlash@carisdx.com Accepted 11 March 2010 Published
Disclaimer: I belong to the speakers bureau of the American Serrated Society, often referred to as the ASS
 An Exposé on Serrated Lesions of the Colorectum How do you know it is a hyperplastic polyp and not something else? How do you know it is something else and not a hyperplastic polyp? Disclaimer: I belong
An Exposé on Serrated Lesions of the Colorectum How do you know it is a hyperplastic polyp and not something else? How do you know it is something else and not a hyperplastic polyp? Disclaimer: I belong
Development of Carcinoma Pathways
 The Construction of Genetic Pathway to Colorectal Cancer Moriah Wright, MD Clinical Fellow in Colorectal Surgery Creighton University School of Medicine Management of Colon and Diseases February 23, 2019
The Construction of Genetic Pathway to Colorectal Cancer Moriah Wright, MD Clinical Fellow in Colorectal Surgery Creighton University School of Medicine Management of Colon and Diseases February 23, 2019
Serrated architecture in colonic mucosa means having
 A Historical Perspective and Exposé on Serrated Polyps of the Colorectum This article offers a historical perspective on the discovery of 3 types of serrated colorectal polyps recognized in the past 60
A Historical Perspective and Exposé on Serrated Polyps of the Colorectum This article offers a historical perspective on the discovery of 3 types of serrated colorectal polyps recognized in the past 60
The Clinical Significance of Serrated Polyps
 CLINICAL AND SYSTEMATIC S nature publishing group 229 CME The Clinical Significance of Serrated Polyps Christopher S. Huang, MD 1, Fr anc is A. Far raye, M D, M S c 1, Sh i Yang, M D 2 and Michael J. O
CLINICAL AND SYSTEMATIC S nature publishing group 229 CME The Clinical Significance of Serrated Polyps Christopher S. Huang, MD 1, Fr anc is A. Far raye, M D, M S c 1, Sh i Yang, M D 2 and Michael J. O
Large Colorectal Adenomas An Approach to Pathologic Evaluation
 Anatomic Pathology / LARGE COLORECTAL ADENOMAS AND PATHOLOGIC EVALUATION Large Colorectal Adenomas An Approach to Pathologic Evaluation Elizabeth D. Euscher, MD, 1 Theodore H. Niemann, MD, 1 Joel G. Lucas,
Anatomic Pathology / LARGE COLORECTAL ADENOMAS AND PATHOLOGIC EVALUATION Large Colorectal Adenomas An Approach to Pathologic Evaluation Elizabeth D. Euscher, MD, 1 Theodore H. Niemann, MD, 1 Joel G. Lucas,
Colorectal adenocarcinoma leading cancer in developed countries In US, annual deaths due to colorectal adenocarcinoma 57,000.
 Colonic Neoplasia Remotti Colorectal adenocarcinoma leading cancer in developed countries In US, annual incidence of colorectal adenocarcinoma 150,000. In US, annual deaths due to colorectal adenocarcinoma
Colonic Neoplasia Remotti Colorectal adenocarcinoma leading cancer in developed countries In US, annual incidence of colorectal adenocarcinoma 150,000. In US, annual deaths due to colorectal adenocarcinoma
Sessile Serrated Polyps: An Important Route to Colorectal Cancer
 Focused Review 1585 Sessile Serrated Polyps: An Important Route to Colorectal Cancer Matthew F. Kalady, MD Abstract The serrated neoplastic pathway accounts for approximately 30% of colorectal cancers.
Focused Review 1585 Sessile Serrated Polyps: An Important Route to Colorectal Cancer Matthew F. Kalady, MD Abstract The serrated neoplastic pathway accounts for approximately 30% of colorectal cancers.
Gastroenterology, Hepatology & Digestive Disorders
 Research Article Gastroenterology, Hepatology & Digestive Disorders Investigating the Prevalence and Progression of Serrated Polyps Tampa VA Experience Shreya Narayanan MD 1*, Brijesh B. Patel MD 2, David
Research Article Gastroenterology, Hepatology & Digestive Disorders Investigating the Prevalence and Progression of Serrated Polyps Tampa VA Experience Shreya Narayanan MD 1*, Brijesh B. Patel MD 2, David
WEO CRC SC Meeting. Barcelona, Spain October 23, 2015
 WEO CRC SC Meeting Barcelona, Spain October 23, 2015 Identification of serrated polyposis syndrome in the context of population-based CRC screening programs Evelien Dekker Academic Medical Center Amsterdam,
WEO CRC SC Meeting Barcelona, Spain October 23, 2015 Identification of serrated polyposis syndrome in the context of population-based CRC screening programs Evelien Dekker Academic Medical Center Amsterdam,
Molecular features of colorectal polyps presenting Kudo s type II mucosal crypt pattern: are they based on the same mechanism of tumorigenesis?
 E171 Molecular features of colorectal polyps presenting Kudo s type II mucosal crypt pattern: are they based on the same mechanism of tumorigenesis? Authors Kensuke Shinmura 1, Kazuo Konishi 1, Toshiko
E171 Molecular features of colorectal polyps presenting Kudo s type II mucosal crypt pattern: are they based on the same mechanism of tumorigenesis? Authors Kensuke Shinmura 1, Kazuo Konishi 1, Toshiko
Cancer emerging from the recurrence of sessile serrated adenoma/polyp resected endoscopically 5 years ago
 Japanese Journal of Clinical Oncology, 2016, 46(1) 89 95 doi: 10.1093/jjco/hyv154 Advance Access Publication Date: 3 November 2015 Case Report Case Report Cancer emerging from the recurrence of sessile
Japanese Journal of Clinical Oncology, 2016, 46(1) 89 95 doi: 10.1093/jjco/hyv154 Advance Access Publication Date: 3 November 2015 Case Report Case Report Cancer emerging from the recurrence of sessile
General Surgery Grand Grounds
 General Surgery Grand Grounds University of Colorado Health Sciences Center Case Presentation December 24, 2009 Adam Lackey, PGY-5 J.L. - 2111609 27 YO female with chief complaint of abdominal pain. PMHx:
General Surgery Grand Grounds University of Colorado Health Sciences Center Case Presentation December 24, 2009 Adam Lackey, PGY-5 J.L. - 2111609 27 YO female with chief complaint of abdominal pain. PMHx:
A Significant Number of Sessile Serrated Adenomas Might Not Be Accurately Diagnosed in Daily Practice
 Gut and Liver, Vol. 4, No. 4, December 2010, pp. 498-502 original article A Significant Number of Sessile Serrated Adenomas Might Not Be Accurately Diagnosed in Daily Practice Soo Woong Kim*, Jae Myung
Gut and Liver, Vol. 4, No. 4, December 2010, pp. 498-502 original article A Significant Number of Sessile Serrated Adenomas Might Not Be Accurately Diagnosed in Daily Practice Soo Woong Kim*, Jae Myung
Serrated polyps of the colorectum: is sessile serrated adenoma distinguishable from hyperplastic polyp in a daily practice?
 Virchows Arch (2007) 450:613 618 DOI 10.1007/s00428-007-0413-8 ORIGINAL ARTICLE Serrated polyps of the colorectum: is sessile serrated adenoma distinguishable from hyperplastic polyp in a daily practice?
Virchows Arch (2007) 450:613 618 DOI 10.1007/s00428-007-0413-8 ORIGINAL ARTICLE Serrated polyps of the colorectum: is sessile serrated adenoma distinguishable from hyperplastic polyp in a daily practice?
Douglas K. Rex, MD Indiana University Hospital Indianapolis, IN
 Serrated Adenomas: What do they mean and what to do about them? Douglas K. Rex, MD Indiana University Hospital Indianapolis, IN Colorectal Cancer Molecular Basis Pathway Frequency Genes MSI Precursor Speed
Serrated Adenomas: What do they mean and what to do about them? Douglas K. Rex, MD Indiana University Hospital Indianapolis, IN Colorectal Cancer Molecular Basis Pathway Frequency Genes MSI Precursor Speed
Arthur Purdy Stout Society of Surgical Pathologists Companion Meeting. Microsatellite Instability and Serrated Adenomas in Common Practice
 Arthur Purdy Stout Society of Surgical Pathologists Companion Meeting Microsatellite Instability and Serrated Adenomas in Common Practice United States and Canadian Academy of Pathology Annual Meeting
Arthur Purdy Stout Society of Surgical Pathologists Companion Meeting Microsatellite Instability and Serrated Adenomas in Common Practice United States and Canadian Academy of Pathology Annual Meeting
SOME OBSERVATIONS ON COLORECTAL POLYPS; ONLY CONCERNS THOSE WHO HAVE A COLON.
 SOME OBSERVATIONS ON COLORECTAL POLYPS; ONLY CONCERNS THOSE WHO HAVE A COLON. Dr Castleman Harvard In 1951 Dr. Benjamin Castleman succeeded Dr. Mallory as Chief of Pathology and Editor of the Case Records
SOME OBSERVATIONS ON COLORECTAL POLYPS; ONLY CONCERNS THOSE WHO HAVE A COLON. Dr Castleman Harvard In 1951 Dr. Benjamin Castleman succeeded Dr. Mallory as Chief of Pathology and Editor of the Case Records
Imaging Evaluation of Polyps. CT Colonography: Sessile Adenoma. Polyps, DALMs & Megacolon Objectives
 Polyps, DALMs & Megacolon: Pathology and Imaging of the Colon and Rectum Angela D. Levy and Leslie H. Sobin Washington, DC Drs. Levy and Sobin have indicated that they have no relationships which, in the
Polyps, DALMs & Megacolon: Pathology and Imaging of the Colon and Rectum Angela D. Levy and Leslie H. Sobin Washington, DC Drs. Levy and Sobin have indicated that they have no relationships which, in the
Colon Cancer and Hereditary Cancer Syndromes
 Colon Cancer and Hereditary Cancer Syndromes Gisela Keller Institute of Pathology Technische Universität München gisela.keller@lrz.tum.de Colon Cancer and Hereditary Cancer Syndromes epidemiology models
Colon Cancer and Hereditary Cancer Syndromes Gisela Keller Institute of Pathology Technische Universität München gisela.keller@lrz.tum.de Colon Cancer and Hereditary Cancer Syndromes epidemiology models
Review Article Serrated Polyposis: An Enigmatic Model of Colorectal Cancer Predisposition
 SAGE-Hindawi Access to Research Pathology Research International Volume 2011, Article ID 157073, 13 pages doi:10.4061/2011/157073 Review Article Serrated Polyposis: An Enigmatic Model of Colorectal Cancer
SAGE-Hindawi Access to Research Pathology Research International Volume 2011, Article ID 157073, 13 pages doi:10.4061/2011/157073 Review Article Serrated Polyposis: An Enigmatic Model of Colorectal Cancer
6 semanas de embarazo. Tubulovillous adenoma with dysplasia icd 10. Inicio / Embarazo / 6 semanas de embarazo
 Inicio / Embarazo / 6 semanas de embarazo 6 semanas de embarazo Tubulovillous adenoma with dysplasia icd 10 Free, official coding info for 2018 ICD-10-CM D13.2 - includes detailed rules, notes, synonyms,
Inicio / Embarazo / 6 semanas de embarazo 6 semanas de embarazo Tubulovillous adenoma with dysplasia icd 10 Free, official coding info for 2018 ICD-10-CM D13.2 - includes detailed rules, notes, synonyms,
PERSPECTIVES IN CLINICAL GASTROENTEROLOGY AND HEPATOLOGY
 CLINICAL GASTROENTEROLOGY AND HEPATOLOGY 2013;11:760 767 PERSPECTIVES IN CLINICAL GASTROENTEROLOGY AND HEPATOLOGY Serrated Colon Polyps as Precursors to Colorectal Cancer SETH SWEETSER,* THOMAS C. SMYRK,
CLINICAL GASTROENTEROLOGY AND HEPATOLOGY 2013;11:760 767 PERSPECTIVES IN CLINICAL GASTROENTEROLOGY AND HEPATOLOGY Serrated Colon Polyps as Precursors to Colorectal Cancer SETH SWEETSER,* THOMAS C. SMYRK,
5/2/2018. Low Grade Dysplasia of GI Tract. High Grade Dysplasia of GI Tract. Dysplasia in Gastrointestinal Tract: Practical Pearls and Issues
 Dysplasia in Gastrointestinal Tract: Practical Pearls and Issues Arief Suriawinata, M.D. Professor of Pathology and Laboratory Medicine Geisel School of Medicine at Dartmouth Department of Pathology and
Dysplasia in Gastrointestinal Tract: Practical Pearls and Issues Arief Suriawinata, M.D. Professor of Pathology and Laboratory Medicine Geisel School of Medicine at Dartmouth Department of Pathology and
Frequency of coexistent carcinoma in sessile serrated adenoma/polyps and traditional serrated adenomas removed by endoscopic resection
 THIEME E451 Frequency of coexistent carcinoma in sessile serrated adenoma/polyps and traditional serrated adenomas removed by endoscopic resection Authors Hirotsugu Saiki 1, Tsutomu Nishida 1, Masashi
THIEME E451 Frequency of coexistent carcinoma in sessile serrated adenoma/polyps and traditional serrated adenomas removed by endoscopic resection Authors Hirotsugu Saiki 1, Tsutomu Nishida 1, Masashi
The Natural History of Right-Sided Lesions
 The Natural History of Right-Sided Lesions Jasper L.A. Vleugels Dept of Gastroenterology and Hepatology, Academic Medical Center, Amsterdam, the Netherlands. None Disclosures Agenda Is there evidence that
The Natural History of Right-Sided Lesions Jasper L.A. Vleugels Dept of Gastroenterology and Hepatology, Academic Medical Center, Amsterdam, the Netherlands. None Disclosures Agenda Is there evidence that
Quality ID #343: Screening Colonoscopy Adenoma Detection Rate National Quality Strategy Domain: Effective Clinical Care
 Quality ID #343: Screening Colonoscopy Adenoma Detection Rate National Quality Strategy Domain: Effective Clinical Care 2018 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY MEASURE TYPE: Outcome DESCRIPTION:
Quality ID #343: Screening Colonoscopy Adenoma Detection Rate National Quality Strategy Domain: Effective Clinical Care 2018 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY MEASURE TYPE: Outcome DESCRIPTION:
colorectal cancer Colorectal cancer hereditary sporadic Familial 1/12/2018
 colorectal cancer Adenocarcinoma of the colon and rectum is the third most common site of new cancer cases and deaths in men (following prostate and lung or bronchus cancer) and women (following breast
colorectal cancer Adenocarcinoma of the colon and rectum is the third most common site of new cancer cases and deaths in men (following prostate and lung or bronchus cancer) and women (following breast
SERRATED POLYPS OF THE COLORECTUM: HISTOLOGICAL
 POL J PATHOL 2010; 1: 8-22 SERRATED POLYPS OF THE COLORECTUM: HISTOLOGICAL CLASSIFICATION AND CLINICAL SIGNIFICANCE JANINA ORŁOWSKA Histopathology Laboratory of the Department of Gastroenterology and Hepatology,
POL J PATHOL 2010; 1: 8-22 SERRATED POLYPS OF THE COLORECTUM: HISTOLOGICAL CLASSIFICATION AND CLINICAL SIGNIFICANCE JANINA ORŁOWSKA Histopathology Laboratory of the Department of Gastroenterology and Hepatology,
In current practice in surgical pathology, colorectal polyps
 Colorectal Polyps With Extensive Absorptive Enterocyte Differentiation Histologically Distinct Variant of Hyperplastic Polyps Hidejiro Yokoo, MD; M. Irtaza Usman, Bsc(Hons); Susan Wheaton, MD; Patricia
Colorectal Polyps With Extensive Absorptive Enterocyte Differentiation Histologically Distinct Variant of Hyperplastic Polyps Hidejiro Yokoo, MD; M. Irtaza Usman, Bsc(Hons); Susan Wheaton, MD; Patricia
Endoscopic discrimination of sessile serrated adenomas from other serrated lesions
 ONCOLOGY LETTERS 2: 785-789, 2011 Endoscopic discrimination of sessile serrated adenomas from other serrated lesions SHIN HSEGW 1, KEIICHI MITSUYM 1, HIROSHI KWNO 1, KEIKO RIT 1, YSUHIKO MEYM 1, YOSHITO
ONCOLOGY LETTERS 2: 785-789, 2011 Endoscopic discrimination of sessile serrated adenomas from other serrated lesions SHIN HSEGW 1, KEIICHI MITSUYM 1, HIROSHI KWNO 1, KEIKO RIT 1, YSUHIKO MEYM 1, YOSHITO
The Importance of Complete Colonoscopy and Exploration of the Cecal Region
 The Importance of Complete Colonoscopy and Exploration of the Cecal Region Kuangi Fu, Takahiro Fujii, Takahisa Matsuda, and Yutaka Saito 2 2.1 The Importance of a Complete Colonoscopy Ever since case-control
The Importance of Complete Colonoscopy and Exploration of the Cecal Region Kuangi Fu, Takahiro Fujii, Takahisa Matsuda, and Yutaka Saito 2 2.1 The Importance of a Complete Colonoscopy Ever since case-control
Patologia sistematica V Gastroenterologia Prof. Stefano Fiorucci. Colon polyps. Colorectal cancer
 Patologia sistematica V Gastroenterologia Prof. Stefano Fiorucci Colon polyps Colorectal cancer Harrison s Principles of Internal Medicine 18 Ed. 2012 Colorectal cancer 70% Colorectal cancer CRC and colon
Patologia sistematica V Gastroenterologia Prof. Stefano Fiorucci Colon polyps Colorectal cancer Harrison s Principles of Internal Medicine 18 Ed. 2012 Colorectal cancer 70% Colorectal cancer CRC and colon
Serrated Colorectal Polyps and the Serrated Neoplasia Pathway
 Serrated Colorectal Polyps and the Serrated Neoplasia Pathway An update on the pathology and clinical significance Mark Be(ngton Envoi Specialist Pathologists Overview 1. A framework of what is included
Serrated Colorectal Polyps and the Serrated Neoplasia Pathway An update on the pathology and clinical significance Mark Be(ngton Envoi Specialist Pathologists Overview 1. A framework of what is included
Neoplastic Colon Polyps. Joyce Au SUNY Downstate Grand Rounds, October 18, 2012
 Neoplastic Colon Polyps Joyce Au SUNY Downstate Grand Rounds, October 18, 2012 CASE 55M with Hepatitis C, COPD (FEV1=45%), s/p vasectomy, knee surgery Meds: albuterol, flunisolide, mometasone, tiotropium
Neoplastic Colon Polyps Joyce Au SUNY Downstate Grand Rounds, October 18, 2012 CASE 55M with Hepatitis C, COPD (FEV1=45%), s/p vasectomy, knee surgery Meds: albuterol, flunisolide, mometasone, tiotropium
Carol A. Burke, MD, FACG
 Updated Guidelines for CRC C Screening and Surveillance Carol A. Burke MD, FACG, FASGE, FACP Cleveland Clinic, Cleveland, OH Gastroenterology t 2012;143:844 143 Gut 2010;59:666 1 Caveat for all Recommendations
Updated Guidelines for CRC C Screening and Surveillance Carol A. Burke MD, FACG, FASGE, FACP Cleveland Clinic, Cleveland, OH Gastroenterology t 2012;143:844 143 Gut 2010;59:666 1 Caveat for all Recommendations
Polypectomy and Local Resections of the Colorectum Structured Pathology Reporting Proforma
 Polypectomy and Local Resections of the Colorectum Structured Pathology Reporting Proforma Mandatory questions (i.e. protocol standards) are in bold (e.g. S1.03). Family name Given name(s) Date of birth
Polypectomy and Local Resections of the Colorectum Structured Pathology Reporting Proforma Mandatory questions (i.e. protocol standards) are in bold (e.g. S1.03). Family name Given name(s) Date of birth
Colon Cancer Screening & Surveillance. Amit Patel, MD PGY-4 GI Fellow
 Colon Cancer Screening & Surveillance Amit Patel, MD PGY-4 GI Fellow Epidemiology CRC incidence and mortality rates vary markedly around the world. Globally, CRC is the third most commonly diagnosed cancer
Colon Cancer Screening & Surveillance Amit Patel, MD PGY-4 GI Fellow Epidemiology CRC incidence and mortality rates vary markedly around the world. Globally, CRC is the third most commonly diagnosed cancer
Update on staging colorectal carcinoma, the 8 th edition AJCC. General overview of staging. When is staging required? 11/1/2017
 Update on staging colorectal carcinoma, the 8 th edition AJCC Dale C. Snover, MD November 3, 2017 General overview of staging Reason for uniform staging Requirements to use AJCC manual and/or CAP protocols
Update on staging colorectal carcinoma, the 8 th edition AJCC Dale C. Snover, MD November 3, 2017 General overview of staging Reason for uniform staging Requirements to use AJCC manual and/or CAP protocols
Title: Serrated polyposis syndrome associated with long-standing inflammatory bowel disease
 Title: Serrated polyposis syndrome associated with long-standing inflammatory bowel disease Authors: Jesús Castro, Miriam Cuatrecasas, Francesc Balaguer, Elena Ricart, María Pellisé DOI: 10.17235/reed.2017.5068/2017
Title: Serrated polyposis syndrome associated with long-standing inflammatory bowel disease Authors: Jesús Castro, Miriam Cuatrecasas, Francesc Balaguer, Elena Ricart, María Pellisé DOI: 10.17235/reed.2017.5068/2017
INTRODUCTION.
 Citation: (2012) 3, e6; doi:10.1038/ctg.2011.5 & 2012 the American College of Gastroenterology All rights reserved 2155-384x/12 www.nature.com/ctg Prevalence of Different Subtypes of Serrated Polyps and
Citation: (2012) 3, e6; doi:10.1038/ctg.2011.5 & 2012 the American College of Gastroenterology All rights reserved 2155-384x/12 www.nature.com/ctg Prevalence of Different Subtypes of Serrated Polyps and
Review Article Serrated Polyps and Their Alternative Pathway to the Colorectal Cancer: A Systematic Review
 Gastroenterology Research and Practice Volume 2015, Article ID 573814, 7 pages http://dx.doi.org/10.1155/2015/573814 Review Article Serrated Polyps and Their Alternative Pathway to the Colorectal Cancer:
Gastroenterology Research and Practice Volume 2015, Article ID 573814, 7 pages http://dx.doi.org/10.1155/2015/573814 Review Article Serrated Polyps and Their Alternative Pathway to the Colorectal Cancer:
Familial and Hereditary Colon Cancer
 Familial and Hereditary Colon Cancer Aasma Shaukat, MD, MPH, FACG, FASGE, FACP GI Section Chief, Minneapolis VAMC Associate Professor, Division of Gastroenterology, Department of Medicine, University of
Familial and Hereditary Colon Cancer Aasma Shaukat, MD, MPH, FACG, FASGE, FACP GI Section Chief, Minneapolis VAMC Associate Professor, Division of Gastroenterology, Department of Medicine, University of
Research Article The CIMP Phenotype in BRAF Mutant Serrated Polyps from a Prospective Colonoscopy Patient Cohort
 Gastroenterology Research and Practice, Article ID 374926, 1 pages http://dx.doi.org/1.1155/214/374926 Research Article The CIMP Phenotype in BRAF Mutant Serrated Polyps from a Prospective Colonoscopy
Gastroenterology Research and Practice, Article ID 374926, 1 pages http://dx.doi.org/1.1155/214/374926 Research Article The CIMP Phenotype in BRAF Mutant Serrated Polyps from a Prospective Colonoscopy
Pathology perspective of colonic polyposis syndromes
 Pathology perspective of colonic polyposis syndromes When are too many polyps too many? David Schaeffer Head and Consultant Pathologist, Department of Pathology and Laboratory Medicine, Vancouver General
Pathology perspective of colonic polyposis syndromes When are too many polyps too many? David Schaeffer Head and Consultant Pathologist, Department of Pathology and Laboratory Medicine, Vancouver General
Luminal Histological Outline and Colonic Adenoma Phenotypes
 Luminal Histological Outline and Colonic Adenoma Phenotypes CARLOS A. RUBIO Gastrointestinal and Liver Pathology Research Laboratory, Department of Pathology, Karolinska Institute and University Hospital,
Luminal Histological Outline and Colonic Adenoma Phenotypes CARLOS A. RUBIO Gastrointestinal and Liver Pathology Research Laboratory, Department of Pathology, Karolinska Institute and University Hospital,
what is the alternative mechanism of histogenesis? Aspects of the morphology of the adenomacarcinoma Morphology of the
 Refer to: Morson B: Polyps and cancer of the large bowel. West J Med 125:93-99, Aug 1976 THE WESTERN Journal of Miedicine Polyps and Cancer of the Large Bowel BASIL MORSON, MD, London MORTALITY STATISTICS
Refer to: Morson B: Polyps and cancer of the large bowel. West J Med 125:93-99, Aug 1976 THE WESTERN Journal of Miedicine Polyps and Cancer of the Large Bowel BASIL MORSON, MD, London MORTALITY STATISTICS
The Detection of Proximal Colon Polyps and Its Importance in Screening Colonoscopy
 ORIGINAL RESEARCH GASTROENTEROLOGY // INTERNAL MEDICINE The Detection of Proximal Colon Polyps and Its Importance in Screening Colonoscopy Răzvan Opaschi 1, Simona Băţagă 1, Ioan Macarie 2, Imola Török
ORIGINAL RESEARCH GASTROENTEROLOGY // INTERNAL MEDICINE The Detection of Proximal Colon Polyps and Its Importance in Screening Colonoscopy Răzvan Opaschi 1, Simona Băţagă 1, Ioan Macarie 2, Imola Török
Quality indicators for colonoscopy and colonoscopist. Mirjana Kalauz Clinical Hospital Center Zagreb
 Quality indicators for colonoscopy and colonoscopist Mirjana Kalauz Clinical Hospital Center Zagreb Why is quality monitoring important in CRC screening programme? Quality adjustment in all endoscopic
Quality indicators for colonoscopy and colonoscopist Mirjana Kalauz Clinical Hospital Center Zagreb Why is quality monitoring important in CRC screening programme? Quality adjustment in all endoscopic
Disclosure Statement. Colorectal Polyps Everything Old Is New Again A Bit of Recent History. Dr Castleman Harvard. Elizabeth Montgomery
 Colorectal Polyps Everything Old Is New Again A Bit of Recent History Elizabeth Montgomery Disclosure Statement Dr. Montgomery reports no relevant financial relationships with commercial interests. Dr
Colorectal Polyps Everything Old Is New Again A Bit of Recent History Elizabeth Montgomery Disclosure Statement Dr. Montgomery reports no relevant financial relationships with commercial interests. Dr
Joseph Misdraji, M.D. GI pathology Unit Massachusetts General Hospital
 Joseph Misdraji, M.D. GI pathology Unit Massachusetts General Hospital jmisdraji@partners.org Adenoma Low-grade appendiceal mucinous neoplasm High-grade appendiceal mucinous neoplasm Adenocarcinoma Serrated
Joseph Misdraji, M.D. GI pathology Unit Massachusetts General Hospital jmisdraji@partners.org Adenoma Low-grade appendiceal mucinous neoplasm High-grade appendiceal mucinous neoplasm Adenocarcinoma Serrated
Colon and Rectum: 2018 Solid Tumor Rules
 2018 SEER Solid Tumor Manual 2018 KCR SPRING TRAINING Colon and Rectum: 2018 Solid Tumor Rules 1 Colon and Rectum Solid Tumor Rules Separate sections for: Introduction Changes from 2007 MP/H rules Equivalent
2018 SEER Solid Tumor Manual 2018 KCR SPRING TRAINING Colon and Rectum: 2018 Solid Tumor Rules 1 Colon and Rectum Solid Tumor Rules Separate sections for: Introduction Changes from 2007 MP/H rules Equivalent
Gastric Polyps. Bible class
 Gastric Polyps Bible class 29.08.2018 Starting my training in gastroenterology, some decades ago, my first chief always told me that colonoscopy may seem technically more challenging but gastroscopy has
Gastric Polyps Bible class 29.08.2018 Starting my training in gastroenterology, some decades ago, my first chief always told me that colonoscopy may seem technically more challenging but gastroscopy has
05/07/2018. Organisation. The English screening programme what is happening? Organisation. Bowel cancer screening in the UK is:
 Organisation The English screening programme what is happening? Phil Quirke Lead Pathologist Bowel Cancer Screening PHE England Bowel Cancer Screening Pathology Committee Started 2006 with roll out 4 devolved
Organisation The English screening programme what is happening? Phil Quirke Lead Pathologist Bowel Cancer Screening PHE England Bowel Cancer Screening Pathology Committee Started 2006 with roll out 4 devolved
2015 Winter School 대장종양성병변의진단과치료. Dong Kyung Chang. Sungkyunkwan University, School of Medicine Samsung Medical Center
 2017 gastroenterology Winter School 77 2015 Winter School 대장종양성병변의진단과치료 Dong Kyung Chang Sungkyunkwan University, School of Medicine Samsung Medical Center Colon Polyps (Epithelial origin) Neoplastic Premalignant
2017 gastroenterology Winter School 77 2015 Winter School 대장종양성병변의진단과치료 Dong Kyung Chang Sungkyunkwan University, School of Medicine Samsung Medical Center Colon Polyps (Epithelial origin) Neoplastic Premalignant
Diagnostic Difficulties Encountered Among Colorectal Polyps
 Diagnostic Difficulties Encountered Among Colorectal Polyps Rhonda K. Yantiss, M.D. Professor of Pathology and Laboratory Medicine Department of Pathology and Laboratory Medicine Weill Cornell Medical
Diagnostic Difficulties Encountered Among Colorectal Polyps Rhonda K. Yantiss, M.D. Professor of Pathology and Laboratory Medicine Department of Pathology and Laboratory Medicine Weill Cornell Medical
The Pathologist s Role in the Diagnosis and Management of Neoplasia in Barrett s Oesophagus Cian Muldoon, St. James s Hospital, Dublin
 The Pathologist s Role in the Diagnosis and Management of Neoplasia in Barrett s Oesophagus Cian Muldoon, St. James s Hospital, Dublin 24.06.15 Norman Barrett Smiles [A brief digression - Chair becoming
The Pathologist s Role in the Diagnosis and Management of Neoplasia in Barrett s Oesophagus Cian Muldoon, St. James s Hospital, Dublin 24.06.15 Norman Barrett Smiles [A brief digression - Chair becoming
B Base excision repair, in MUTYH-associated polyposis and colorectal cancer, BRAF testing, for hereditary colorectal cancer, 696
 Index Note: Page numbers of article titles are in boldface type. A Adenomatous polyposis, familial. See Familial adenomatous polyposis. Anal anastomosis, ileal-pouch, proctocolectomy with, in FAP, 591
Index Note: Page numbers of article titles are in boldface type. A Adenomatous polyposis, familial. See Familial adenomatous polyposis. Anal anastomosis, ileal-pouch, proctocolectomy with, in FAP, 591
Original Article Immunohistochemical expression profiles of BRAF (V600E/VE1) in serrated colon polyps in Turkish population
 Int J Clin Exp Pathol 2017;10(8):8868-8874 www.ijcep.com /ISSN:1936-2625/IJCEP0057472 Original Article Immunohistochemical expression profiles of BRAF (V600E/VE1) in serrated colon polyps in Turkish population
Int J Clin Exp Pathol 2017;10(8):8868-8874 www.ijcep.com /ISSN:1936-2625/IJCEP0057472 Original Article Immunohistochemical expression profiles of BRAF (V600E/VE1) in serrated colon polyps in Turkish population
Index. Note: Page numbers of article titles are in boldface type.
 Index Note: Page numbers of article titles are in boldface type. A Abdominal surgery prior as factor in laparoscopic colorectal surgery, 554 555 Abscess(es) CRC presenting as, 539 540 Adenocarcinoma of
Index Note: Page numbers of article titles are in boldface type. A Abdominal surgery prior as factor in laparoscopic colorectal surgery, 554 555 Abscess(es) CRC presenting as, 539 540 Adenocarcinoma of
GOBLET CELL CARCINOID. Hanlin L. Wang, MD, PhD University of California Los Angeles
 GOBLET CELL CARCINOID Hanlin L. Wang, MD, PhD University of California Los Angeles Disclosure of Relevant Financial Relationships USCAP requires that all planners (Education Committee) in a position to
GOBLET CELL CARCINOID Hanlin L. Wang, MD, PhD University of California Los Angeles Disclosure of Relevant Financial Relationships USCAP requires that all planners (Education Committee) in a position to
GOBLET CELL CARCINOID
 GOBLET CELL CARCINOID Hanlin L. Wang, MD, PhD University of California Los Angeles Disclosure of Relevant Financial Relationships USCAP requires that all planners (Education Committee) in a position to
GOBLET CELL CARCINOID Hanlin L. Wang, MD, PhD University of California Los Angeles Disclosure of Relevant Financial Relationships USCAP requires that all planners (Education Committee) in a position to
Genetic testing all you need to know
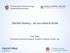 Genetic testing all you need to know Sue Clark Consultant Colorectal Surgeon, St Mark s Hospital, London, UK. Colorectal cancer Familial 33% Polyposis syndromes
Genetic testing all you need to know Sue Clark Consultant Colorectal Surgeon, St Mark s Hospital, London, UK. Colorectal cancer Familial 33% Polyposis syndromes
Surveying the Colon; Polyps and Advances in Polypectomy
 Surveying the Colon; Polyps and Advances in Polypectomy Educational Objectives Identify classifications of polyps Describe several types of polyps Verbalize rationale for polypectomy Identify risk factors
Surveying the Colon; Polyps and Advances in Polypectomy Educational Objectives Identify classifications of polyps Describe several types of polyps Verbalize rationale for polypectomy Identify risk factors
UvA-DARE (Digital Academic Repository) Serrated polyps of the colon and rectum Hazewinkel, Y. Link to publication
 UvA-DARE (Digital Academic Repository) Serrated polyps of the colon and rectum Hazewinkel, Y. Link to publication Citation for published version (APA): Hazewinkel, Y. (2014). Serrated polyps of the colon
UvA-DARE (Digital Academic Repository) Serrated polyps of the colon and rectum Hazewinkel, Y. Link to publication Citation for published version (APA): Hazewinkel, Y. (2014). Serrated polyps of the colon
Pathology Today American Society for Clinical Pathology Annual Meeting Workshop No: 1340 Gastrointestinal Pathology: New Approaches to Old Problems
 Pathology Today American Society for Clinical Pathology Annual Meeting Workshop No: 1340 Gastrointestinal Pathology: New Approaches to Old Problems ASCP Annual Meeting/WASPaLM XXVI World Congress Las Vegas,
Pathology Today American Society for Clinical Pathology Annual Meeting Workshop No: 1340 Gastrointestinal Pathology: New Approaches to Old Problems ASCP Annual Meeting/WASPaLM XXVI World Congress Las Vegas,
Colorectal Neoplasia. Dr. Smita Devani MBChB, MRCP. Consultant Physician and Gastroenterologist Aga Khan University Hospital, Nairobi
 Colorectal Neoplasia Dr. Smita Devani MBChB, MRCP Consultant Physician and Gastroenterologist Aga Khan University Hospital, Nairobi Case History BT, 69yr male Caucasian History of rectal bleeding No change
Colorectal Neoplasia Dr. Smita Devani MBChB, MRCP Consultant Physician and Gastroenterologist Aga Khan University Hospital, Nairobi Case History BT, 69yr male Caucasian History of rectal bleeding No change
2019 COLLECTION TYPE: MIPS CLINICAL QUALITY MEASURES (CQMS) MEASURE TYPE: Outcome High Priority
 Quality ID #343: Screening Colonoscopy Adenoma Detection Rate National Quality Strategy Domain: Effective Clinical Care Meaningful Measure Area: Preventive Care 2019 COLLECTION TYPE: MIPS CLINICAL QUALITY
Quality ID #343: Screening Colonoscopy Adenoma Detection Rate National Quality Strategy Domain: Effective Clinical Care Meaningful Measure Area: Preventive Care 2019 COLLECTION TYPE: MIPS CLINICAL QUALITY
Guidelines for Colonoscopy Surveillance After Screening and Polypectomy: A Consensus Update by the US Multi-Society Task Force on Colorectal Cancer
 Guidelines for Colonoscopy Surveillance After Screening and Polypectomy: A Consensus Update by the US Multi-Society Task Force on Colorectal Cancer David A. Lieberman, 1 Douglas K. Rex, 2 Sidney J. Winawer,
Guidelines for Colonoscopy Surveillance After Screening and Polypectomy: A Consensus Update by the US Multi-Society Task Force on Colorectal Cancer David A. Lieberman, 1 Douglas K. Rex, 2 Sidney J. Winawer,
Epithelial Columnar Breast Lesions: Histopathology and Molecular Markers
 29th Annual International Conference Advances in the Application of Monoclonal Antibodies in Clinical Oncology and Symposium on Cancer Stem Cells 25 th -27t h June, 2012, Mykonos, Greece Epithelial Columnar
29th Annual International Conference Advances in the Application of Monoclonal Antibodies in Clinical Oncology and Symposium on Cancer Stem Cells 25 th -27t h June, 2012, Mykonos, Greece Epithelial Columnar
Measure #343: Screening Colonoscopy Adenoma Detection Rate National Quality Strategy Domain: Effective Clincal Care
 Measure #343: Screening Colonoscopy Adenoma Detection Rate National Quality Strategy Domain: Effective Clincal Care 2016 PQRS OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY DESCRIPTION: The percentage
Measure #343: Screening Colonoscopy Adenoma Detection Rate National Quality Strategy Domain: Effective Clincal Care 2016 PQRS OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY DESCRIPTION: The percentage
Expert panel observations
 Expert panel observations Professor Neil A Shepherd Gloucester and Cheltenham, UK Gloucestershire Cellular Pathology Laboratory Three big issues in BCSP pathology serrated pathology & what do we do about
Expert panel observations Professor Neil A Shepherd Gloucester and Cheltenham, UK Gloucestershire Cellular Pathology Laboratory Three big issues in BCSP pathology serrated pathology & what do we do about
D uring the past few years, it has become
 682 REVIEW My approach to serrated polyps of the colorectum T Higuchi, J R Jass... Hyperplastic polyps of the colorectum are heterogeneous lesions, a subset of which is now regarded as the precursor of
682 REVIEW My approach to serrated polyps of the colorectum T Higuchi, J R Jass... Hyperplastic polyps of the colorectum are heterogeneous lesions, a subset of which is now regarded as the precursor of
Yukari Fujimori 1, Takahiro Fujimori 1, Johji Imura 2*, Tamotsu Sugai 3, Takashi Yao 4, Ryo Wada 5, Yoichi Ajioka 6 and Yasuo Ohkura 7
 Fujimori et al. Diagnostic Pathology 2012, 7:59 RESEARCH Open Access An assessment of the diagnostic criteria for sessile serrated adenoma/polyps: SSA/Ps using image processing software analysis for Ki67
Fujimori et al. Diagnostic Pathology 2012, 7:59 RESEARCH Open Access An assessment of the diagnostic criteria for sessile serrated adenoma/polyps: SSA/Ps using image processing software analysis for Ki67
Select problems in cystic pancreatic lesions
 Disclosure Select problems in cystic pancreatic lesions Five Prime Therapeutics shareholder Adicet Bio shareholder Bristol-Meyer Squibb advisory board grace.kim@ucsf.edu Pancreatic cystic lesions Intraductal
Disclosure Select problems in cystic pancreatic lesions Five Prime Therapeutics shareholder Adicet Bio shareholder Bristol-Meyer Squibb advisory board grace.kim@ucsf.edu Pancreatic cystic lesions Intraductal
Case history: Figure 1. H&E, 5x. Figure 2. H&E, 20x.
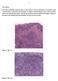 1 Case history: A 49 year-old female presented with a 5 year history of chronic anal fissure. The patient s past medical history is otherwise unremarkable. On digital rectal examination there was a very
1 Case history: A 49 year-old female presented with a 5 year history of chronic anal fissure. The patient s past medical history is otherwise unremarkable. On digital rectal examination there was a very
Clinicopathological Study of Serrated Polyps of the Colorectum, with Special Reference to Maspin Expression
 Showa Univ J Med Sci 24 4, 285 292, December 2012 Original Clinicopathological Study of Serrated Polyps of the Colorectum, with Special Reference to Maspin Expression Yasuo OCHIAI 1 2, Nobuyuki OHIKE 1,
Showa Univ J Med Sci 24 4, 285 292, December 2012 Original Clinicopathological Study of Serrated Polyps of the Colorectum, with Special Reference to Maspin Expression Yasuo OCHIAI 1 2, Nobuyuki OHIKE 1,
Familial and Hereditary Colon Cancer
 Familial and Hereditary Colon Cancer Aasma Shaukat, MD, MPH, FACG, FASGE, FACP GI Section Chief, Minneapolis VAMC Associate Professor, Division of Gastroenterology, Department of Medicine, University of
Familial and Hereditary Colon Cancer Aasma Shaukat, MD, MPH, FACG, FASGE, FACP GI Section Chief, Minneapolis VAMC Associate Professor, Division of Gastroenterology, Department of Medicine, University of
Disclosure slide I Member of advisory board for AMGEN, ROCHE, BOEHRINGER I Speaker honoraria from FALK Pharma, Pfizer, Lilly and ROCHE I Third party f
 Early (and not so early) colorectal cancer: The pathologist s point of view Daniela E. Aust, Institute for Pathology, University Hospital Dresden, Germany Disclosure slide I Member of advisory board for
Early (and not so early) colorectal cancer: The pathologist s point of view Daniela E. Aust, Institute for Pathology, University Hospital Dresden, Germany Disclosure slide I Member of advisory board for
Early (and not so early) colorectal cancer: The pathologist s point of view
 Early (and not so early) colorectal cancer: The pathologist s point of view Daniela E. Aust, Institute for Pathology, University Hospital Dresden, Germany Disclosure slide I Member of advisory board for
Early (and not so early) colorectal cancer: The pathologist s point of view Daniela E. Aust, Institute for Pathology, University Hospital Dresden, Germany Disclosure slide I Member of advisory board for
COLON AND RECTUM SOLID TUMOR RULES ABSTRACTORS TRAINING
 COLON AND RECTUM SOLID TUMOR RULES ABSTRACTORS TRAINING COLON AND RECTUM SOLID TUMOR RULES Separate sections for: Introduction Changes from 2007 MP/H rules Equivalent Terms Terms that are NOT Equivalent
COLON AND RECTUM SOLID TUMOR RULES ABSTRACTORS TRAINING COLON AND RECTUM SOLID TUMOR RULES Separate sections for: Introduction Changes from 2007 MP/H rules Equivalent Terms Terms that are NOT Equivalent
Pathology in Slovenian CRC screening programme:
 Pathology in Slovenian CRC screening programme: Findings, organisation and quality assurance Snježana Frković Grazio University Medical Center Ljubljana, Slovenia Slovenia s population: 2 million Incidence
Pathology in Slovenian CRC screening programme: Findings, organisation and quality assurance Snježana Frković Grazio University Medical Center Ljubljana, Slovenia Slovenia s population: 2 million Incidence
Neoplasia 2018 Lecture 2. Dr Heyam Awad MD, FRCPath
 Neoplasia 2018 Lecture 2 Dr Heyam Awad MD, FRCPath ILOS 1. List the differences between benign and malignant tumors. 2. Recognize the histological features of malignancy. 3. Define dysplasia and understand
Neoplasia 2018 Lecture 2 Dr Heyam Awad MD, FRCPath ILOS 1. List the differences between benign and malignant tumors. 2. Recognize the histological features of malignancy. 3. Define dysplasia and understand
