EXPRESSION OF FATTY ACID OXIDATION-RELATED GENES IN Acsl4 L -/- PRIMARY HEPATOCYTES. Dennis Lin
|
|
|
- Nancy Jacobs
- 5 years ago
- Views:
Transcription
1 EXPRESSION OF FATTY ACID OXIDATION-RELATED GENES IN Acsl4 L -/- PRIMARY HEPATOCYTES Dennis Lin Senior Honors Thesis Department of Nutrition University of North Carolina at Chapel Hill April 2018 Approved by, Rosalind Coleman Eric L. Klett i
2 Abstract Long-chain acyl-coa synthetases are a family of enzymes responsible for catalyzing the reaction converting long chain fatty acids to fatty acyl-coa. ACSL4, a specific member of this family, plays a role in eicosanoid metabolism and has been found to be associated with liver diseases including non-alcoholic fatty liver disease and hepatocellular carcinoma. In order to study ACSL4 in the context of hepatic lipid metabolism, a murine liver-specific Acsl4 knockout (Acsl4 L -/- ) was developed and characterized in our lab. Based on a previous experiment in our lab, we hypothesized that Acsl4 L -/- would cause a defect in beta adrenergic signaling. In order to test this hypothesis, we conducted experiments to determine the effects of various treatments on the expression of fatty acid oxidation-related genes in both knockout and control hepatocytes. Treatments with fatty acids caused increases in expression of several fatty acid oxidation-related genes in both knockout and control hepatocytes. Hormone treatments did not cause anticipated increases in expression of fatty acid oxidation-related genes in several of the control groups. Because of this we were not able to make conclusions about the effects of the knockout. These results indicated that Acsl4 L -/- did not cause a defect in beta adrenergic signaling. ii
3 Acknowledgements First and foremost, I would like to thank Dr. Rosalind Coleman for allowing me to work in her lab and shaping me into the scientist I am today. I appreciate all the time and patience she invested in me at a point a when I had a lot less knowledge than I do now. I would especially like to thank Dr. Pamela Young, for guiding me every step of the way through my projects and teaching me almost everything I know about lipid research. She was instrumental in helping me grow as a researcher and encouraged me during times of frustration and doubt. I would also like to thank Dr. Eric Klett, Dr. Amanda Suchanek, Dr. Theresa D Aquila, Dr. Shufen Chen, Liyang Zhao, and everyone else in the Coleman lab for giving me advice and support for my experiments. I have grown tremendously during my time in the Coleman lab and would not have been able to do it without all of their support. iii
4 Table of Contents List of Tables... v List of Figures... vi Chapter I. Introduction Liver and Lipid Metabolism ACSL4 Gene and Protein ACSL4 Abnormalities and Human Disease ACSL4 Tissue and Substrate Preferences Study Aims and Hypothesis:... 4 Chapter II. Methods and Materials Primary hepatocyte isolation and preparation Primary hepatocyte hormone treatments Primary hepatocyte lipid treatments Gene expression in ACSL4 L -/- and control primary hepatocytes... 7 Chapter III. Results Oleate and palmitate treatment effects on gene expression Hormone treatment effects on gene expression... 8 Chapter IV. Discussion Adjustments to Methods and Materials Hormone treatment effects on gene expression Fatty acid treatment effects on gene expression Future Directions References iv
5 List of Tables Table 1. Treatment and corresponding vehicle conditions Table 2. RT-PCR Primer Sequences v
6 List of Figures Figure 1. Lipid Treatments Gene Expression (Experiment 1) Figure 2. Lipid Treatments Gene Expression (Experiment 2) Figure 3. Hormone Treatment Gene Expression (Experiment 1) Figure 4. Hormone Treatment Gene Expression (Experiment 2) Figure 5. Hormone Treatments and their Signaling Mechanisms vi
7 Chapter I. Introduction 1.1 Liver and Lipid Metabolism The liver is perhaps the most important organ when it comes to lipid metabolism, acting as the main site for fatty acid (FA) synthesis, fatty acid oxidation, complex lipid synthesis, and very low-density lipoprotein (VLDL) production. The liver acts as a mediator for fat metabolism to meet the energy requirements of the rest of the body. These metabolic pathways are controlled by various enzyme interactions and regulated by the liver under different hormonal and physiological conditions. Lipid metabolism starts with the digestion and absorption of dietary fats. In the mouth, lipids are first broken down by lingual lipases and further processed in the stomach and duodenum where dietary fats are emulsified with help from bile acids secreted from the gallbladder. Lipases secreted from the pancreas further digest lipids into free FA and monoacylglycerol to be absorbed by enterocytes. Within the enterocyte, triacylglerol (TAG) molecules are resynthesized and repackaged along with cholesterol esters (CE), phospholipids (PL), lipoproteins, and fat-soluble vitamins into chylomicron lipoproteins that pass into lacteals that eventually enter circulation. Peripheral tissues hydrolyze TAG from chylomicrons via lipoprotein lipase to obtain FA while remnant chylomicrons and other lipoproteins are taken up by the liver. The liver incorporates the components of remnant lipoproteins particles into various metabolic pathways. Long-chain FAs are activated by the long chain acyl-coa synthetase (ACSL) enzyme family, which converts long chain FAs into long chain fatty acyl-coas and shuttles them into different metabolic pathways. 1 Depending on energy status, activated FAs can enter synthetic pathways such as de novo lipogenesis, TAG synthesis, and phospholipid synthesis. TAG synthesis is initiated by ACSLs, with glycerol-3-phosphate acyltransferase (GPAT) catalyzing the formation of TAG by esterifying glycerol-3-phosphate with acyl-coa. Acetyl-CoA carboxylase (ACC) catalyzes the conversion of acetyl-coa to malonyl-coa and is a ratelimiting enzyme of de novo lipogenesis. ACC activity is inhibited under low energy conditions. Elongation of malonyl-coa for FA synthesis is catalyzed by fatty acid synthetase (FAS). These processes are highly regulated by insulin and glucagon signals that regulate appropriate pathways during fed and fasting conditions respectively. As the major lipid metabolism center of the body, 1
8 the liver also serves as the main distribution center. It exports lipids, TAG, CE, and phospholipids by producing and secreting VLDL for peripheral utilization of lipids and FAs. Hepatic concentrations of lipid and apolipoprotein B100 directly affect VLDL production and secretion. Beta-oxidation is the degradative pathway of FAs activated by ACSLs, which produces acetyl-coa from fatty acyl-coa to be used as energy production via the Krebs cycle and oxidative phosphorylation. The main regulated enzyme of beta-oxidation is carnitine palmitoyltransferase I (CPT-1), which catalyzes the entry of fatty acyl CoA into the mitochondria via conversion to acyl-carnitine. CPT-1 is inhibited by malonyl CoA, which is produced by ACC during de novo lipogenesis. Beta-oxidation is also regulated by the transcription factor peroxisome proliferator-activated receptor alpha (PPARa). PPARa ligand activation induces several genes controlling FA import including CPT-1 and several major enzymes within the beta-oxidation pathway including acyl-coa dehydrogenases ACSL4 Gene and Protein While all enzymes in the ACSL family serve the same enzymatic function, the ACSL isoforms vary in substrate specificity, subcellular location, and potential impact on downstream metabolic functions. With the exception of ACSL4, which is localized to both the plasma membrane and cytosol, most of the ACSL isoforms are membrane associated. 3 Accurate methods for measuring enzymatic activity for individual ACSL isoforms have not been well established due to non-specificity of ACSL inhibitors. It is still unclear how each isoform s distinct structure and subcellular location affect function and activity. This study aims to investigate the effects of a liver-specific ACSL4 knock out (ACSL4 L -/- ) mouse model to study the role of ACSL4 in hepatic lipid metabolism. ACSL4 is a member of the ACS family that is encoded by the Acsl4 gene located on the X-chromosome adjacent to the Col4a5 gene at Xq23 4. The ACSL4 protein contains 711 amino acids with an AMP-dependent synthetase domain and an AMP-binding domain. Alternative splicing produces two possible forms of the ACSL4 isoform. ACSL4 variant 1 is a 75 kda protein located within the cytosol. ACSL4 variant 2 is a 79 kda protein that contains additional hydrophobic amino acids at the N-terminus that contribute to membrane localization in both the outer mitochondrial membrane and the endoplasmic reticulum. 5 2
9 1.3 ACSL4 Abnormalities and Human Disease Sequence abnormalities and sequence deletions surrounding the Acsl4 gene have been associated with several conditions including elliptocytosis, mental retardation, and Alport syndrome. Alport syndrome is an X-linked condition that is characterized by a large deletion of the Xq (including the Acsl4 gene) region of the X-chromosome. Its symptoms include progressive renal failure, hearing loss, and vision loss. High Acsl4 expression in specific regions of the brain, including the cerebellum and hippocampus, and its deficiencies associated with mental retardation suggest that the enzyme may play an important role in brain development. 6 Investigations of Acsl4 expression during mouse development showed high levels of expression in mice embryos and newborn brains. 7 ACSL4 dysregulation has also been studied in the context of hepatic lipid dysfunction associated with non-alcoholic fatty liver disease (NAFLD) and hepatocellular carcinoma. Studies using CLOCK-deficient mouse models found that reduced Acsl4 and Fabp1 expression was associated with reduced steatosis on a high fat diet. 8 This suggests that ACSL4 may play a role in the development of hepatosteatosis and circadian controlled lipid metabolism. In human studies, researchers identified polymorphisms in Acsl4 that were related to liver fat content. Higher hepatic lipid content of NAFLD patients, along with hyperinsulinemia and obesity, were associated with the expression of a rare ACSL4 rs allele. 9 ACSL4 was also found to be involved in hepatocellular carcinoma development. 10 Several human hepatoma cell lines, including Hep3B and HepG2, have exhibited high ACSL4 expression. 11 In patients, ACSL4 has been found to be up-regulated (between 2.3 and 27.5-fold) in hepatocellular carcinoma compared to adjacent non-cancerous tissue. 11 These findings suggest that ACSL4 expression may contribute to dysregulation in hepatic lipid metabolism associated with both cancer and steatosis. 1.4 ACSL4 Tissue and Substrate Preferences As mentioned before, each ACS isoform has a specific FA substrate preference and a unique distribution across tissues in the body. Kinetic analyses have shown that ACSL4 has a preference for polyunsaturated FAs, with lower affinities for saturated and mono, di-, and triunsaturated FAs. The enzyme preferentially utilizes arachidonate and eicosapentaenoate as substrates over the other 8-12 carbon saturated FAs and carbon unsaturated FAs examined in the analyses. 6 ACSL4 is present in the brain and steroidogenic tissues. RNA analyses have 3
10 revealed the presence of Acsl4 transcripts in the adrenal cortex, brain, lung, ovaries, testes, and liver. 6 Because of its preference for polyunsaturated FAs, ACSL4 may play an important role in eicosanoid metabolism. Arachidonate has potential to generate inflammatory products as a precursor to several clinically important eicosanoids and metabolites of eicosanoids. Cyclooxygenases metabolize arachidonate to prostaglandins and thromboxanes that mediate inflammation and platelet aggregation. To prevent excessive eicosanoid synthesis, free arachidonate is converted to arachidonoyl-coa by ACS enzymes and re-esterified into phospholipids. 1.5 Study Aims and Hypothesis: Liver-specific ACSL4 knock out mice (ACSL4 L -/- ) have been developed to further investigate the role of ACSL4 in hepatic lipid metabolism. Under fasting and ketogenic conditions, ACSL4 L -/- mice and control mice do not exhibit differences in ACS activity, serum TAG, or serum glucose. Ongoing studies have suggested that ACSL4 L -/- may affect transcriptional regulation of beta-oxidation by disrupting adrenergic signaling. Hepatic lipid metabolism is tightly controlled by hormone responses. Glucagon and catecholamines, such as norepinephrine, stimulate G-protein coupled receptors and initiate signaling cascades that regulate both glucose and lipid homeostasis via transcriptional regulation. Stimulation of adrenergic and glucagon receptors increases production of camp and activates protein kinase A (PKA). PKA phosphorylates transcription factor CREB, which induces PGC-1α gene expression. Acting as a coactivator, PGC-1α is recruited by transcription factors such as PPARα that bind to the promoter regions of FA oxidation enzyme genes to increase expression. 12 A previous study tested the effects of norepinephrine treatments on cultured primary hepatocytes from both control and ACSL4 L -/- mice. Results showed lowered gene expression of beta-oxidation genes among ACSL4 L -/- hepatocytes after treatment compared to control. Treatments with PKA activator 8-Br-cAMP (a long-acting derivative of camp) induced similar levels of gene expression of beta-oxidation genes in both ACSL4 L -/- and control hepatocytes suggesting a possible role of ACSL4 in adrenergic signaling pathway before PKA activation. Oleic acid, but not long chain saturated or polyunsaturated FAs, has been found to stimulate camp production and PKA activity. PKA phosphorylates SIRT1, which then deacetylates and activates PGC-1α to subsequently increase expression of beta-oxidation genes. 13 4
11 Therefore, our study aimed to examine the effects of Acsl4 deletion on the expression of FA oxidation genes after treatments with several hormones and metabolites that promote oxidation and to validate the findings of the previous study. We hypothesize that after treatments that activate the adrenergic receptor, ACSL4 L -/- hepatocytes will exhibit lower FA oxidation gene expression compared to control. We also hypothesize that after oleic acid treatment, ACSL4 L -/- hepatocytes will exhibit greater FA oxidation gene expression compared to control. Lastly, we hypothesize that after treatments that directly elevate intracellular camp levels, ACSL4 L -/- and control hepatocytes will have similar FA oxidation gene expression. 5
12 Chapter II. Methods and Materials 2.1 Primary hepatocyte isolation and preparation Primary hepatocytes were isolated from the livers of 3-month-old ACSL4 L -/- mice and floxed control littermates. Because Acsl4 is X-linked, we used male knockout mice for our experiments. Hepatocytes were plated at 4 X 10 5 cells per well in 6-well plates with William s E. media (5% serum). After two hours, William s E. media was replaced with low glucose DMEM (1g/L glucose, 10% fetal bovine serum, penicillin-streptomycin). Before treatments, primary hepatocytes of both genotypes were serum starved in low glucose DMEM (5mM glucose, 1mM sodium pyruvate, 1mM L-Glutamine, serum free) media using 1 ml per well for 4 hours. 2.2 Primary hepatocyte hormone treatments All treatments were prepared using the same glucose (5mM) DMEM media at 1 ml per well. Hormone solutions were added to three wells of each plate and the corresponding vehicle control solutions were added to the remaining three wells of each plate. The norepinephrine solution was prepared at 10µM from a 10mM norepinephrine stock (dissolved in water) diluted in media. The 8-Br-cAMP treatment solution was prepared at 1 mm from an 80mM stock (dissolved in water) diluted in media. The forskolin treatment solution was prepared at 40 µm from a 4mM stock (dissolved in DMSO) diluted in media. The glucagon treatment solution was prepared at 100mM from a 1mg/mL stock (dissolved in phosphate buffered saline) diluted in media. All vehicle control solutions were prepared by diluting the corresponding vehicle (Table 1) in media by the same dilution factor used for diluting the stock hormone solution. After incubating the hepatocytes for 1 hour, the cells were washed twice with chilled PBS and frozen at -80 C. 2.3 Primary hepatocyte lipid treatments All treatments were prepared using the same glucose (5mM) DMEM media at 1 ml per well. Lipid solutions were added to three wells of each plate and the corresponding vehicle control solutions were added to the remaining three wells of each plate. The oleate treatment was prepared at 500µM from a 100mM stock (in 50% ethanol) diluted in media. The palmitate treatment was prepared at 500µM from a 200mM stock (in 50% ethanol) diluted in media. Both 6
13 stock solutions were first diluted with fatty acid free bovine serum albumin (1:25 for oleate and 1:50 for palmitate) and allowed to complex at 37 C for 1 hour. Carnitine and serum free media was added to both solutions for final concentrations of 1mM carnitine and 500µM of FA. After treating the primary hepatocytes for 3.5 hours, the cells were washed twice with chilled PBS and stored at -80 C. 2.4 Gene expression in ACSL4 L -/- and control primary hepatocytes Before testing expression of FA oxidation genes, the liver-specific knockout of Acsl4 was confirmed. RNA was extracted from both ACSL4 L -/- hepatocytes and littermate control hepatocytes using phenol chloroform extraction (Invitrogen TRIzol reagent). RNA concentration was measured by spectrophotometry and cdna was made by reverse transcription (iscript cdna synthesis kit) using 1µg of RNA. The mrna levels of Acsl4 were quantified with real time PCR using ACSL4 and β-actin primers (Table 1) to determine the relative expression of Acsl4 compared to Actin (Biorad itaq universal SYBR green supermix). Acsl4 expression in ACSL4 L -/- mice was less than 1% of control littermates, confirming the knockout. The same process was carried out to determine the gene expression levels of the other genes of interest using the corresponding forward and reverse primers (Table 2) and normalizing to Actin expression. For each data set generated, cycle threshold values were normalized to Actin and treatment control values. 7
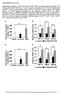
14 Chapter III. Results 3.1 Oleate and palmitate treatment effects on gene expression To determine whether ACSL4 L -/- disrupts oleate s signaling effects on the expression of beta-oxidation genes, ACSL4 L -/- and control hepatocytes were treated with oleate and palmitate. Two experiments were conducted using a different set of hepatocytes. RT-PCR results from Experiment 1 showed increased Cpt1a, Acox1, Pgc1a, and Ppara expression (relative to Actin) following both oleate and palmitate treatments (Figure 1). These increases in gene expression occurred in both ACSL4 L -/- and control hepatocytes. Experiment 2 results, with palmitate treatment, showed increased Cpt1a, Acox1, Ppara and Creb expression in control hepatocytes along with increased Acox1 and decrease Ppara expression in ACSL4 L -/- hepatocytes (Figure 2). Experiment 2 results, with oleate treatment, showed increased Creb expression and decreased Pgc-1α and Pparα expression in control hepatocytes. In ACSL4 L -/- hepatocytes, oleate treatment resulted in increased Creb and Acox1 expression and decreased Pgc-1α and Pparα expression. 3.2 Hormone treatment effects on gene expression To investigate whether ACSL4 L -/- disrupts adrenergic signaling effects on beta oxidation gene expression, ACSL4 L -/- and control hepatocytes were treated with norepinephrine, forskolin, glucagon, and 8-Br-cAMP. All of these hormone treatments increase expression of fatty acid oxidation-related genes by increasing intracellular camp (Figure 5). RT-PCR results from Experiment 1 showed increased Pgc1a, Cpt1a, and Ppara expression following norepinephrine treatment only in control hepatocytes (Figure 3A). With 8-Br-cAMP treatment, Pgc1a expression increased only in ACSL4 L -/- hepatocytes. There was also lowered Cpt1a and Ppara expression among all ACSL4 L -/- hepatocytes in the 8-Br-cAMP study compared to control (Figure 3B). After forskolin treatment, Ppar expression decreased only in ACSL4 L -/- hepatocytes (Figure 3C). After glucagon treatment, Pepck expression increased only in control hepatocytes (Figure 3C). There was also lowered Pdk4 expression in ACSL4 L -/- hepatocytes compared to control following glucagon treatment. After correcting the defects of Experiment 1, the experiment was repeated. Experiment 2 results showed different patterns of gene expression after treating hepatocytes with 8
15 norepinephrine and 8-Br-cAMP treatments. Unlike in Experiment 1, norepinephrine treatment increased Pgc1a expression in both ACSL4 L -/- and control hepatocytes and increased Cpt1a expression in only ACSL4 L -/- hepatocytes. Cpt1a and Ppara expression decreased among control hepatocytes following norepinephrine treatment (Figure 4A). Treatment with 8-Br-cAMP only affected Pgc1a expression, with increased expression in both control and ACSL4 L -/- hepatocytes (Figure 4B). Following forskolin treatment, only Pgc1a expression increased in control hepatocytes while only Cpt1a expression decreased in ACSL4 L -/- hepatocytes (Figure 4C). Following glucagon treatment, both Pepck and G6pase expression increased in control hepatocytes (Figure 4D). Unlike in Experiment 1, G6pase expression also increased in ACSL4 L - /- hepatocytes following glucagon treatment. 9
16 Chapter IV. Discussion 4.1 Adjustments to Methods and Materials Experiment 1 produced gene expression data that were inconsistent with the expected treatment effects. The data also exhibited high variability between technical replicates within treatment groups. This was likely due to the poor quality of the extracted RNA and reagents used for treatments. Further analysis of A260/A230 ratios revealed poor RNA purity of Experiment 1 extracted RNA. Prior to performing Experiment 2 with new cells, several adjustments were made to improve experimental conditions. In order to improve the quality and purity of the extracted RNA, the extraction was performed with smaller batches of cells at a time to prevent thawing/degradation that would disturb expression patterns. RNA was also eluted using spin columns to improve purity and yield for extraction. RNAase free micro-volume tubes were also used in place of standard micro-volume tubes during cdna synthesis. New reagents were purchased for norepinephrine, 8-Br-cAMP, and glucagon treatments prior to the second experiment to replace expired reagents used for the first experiment. 4.2 Hormone treatment effects on gene expression The main objective of this study was to validate the findings of a previous experiment in our lab that suggested a possible role for ACSL4 in beta adrenergic signaling. While Experiment 2 was conducted using higher quality RNA resulting in less apparent variability, the hormone treatments did not induce the gene expression patterns that were expected in control hepatocytes. All hormone treatments used for this study should have induced a similar response in control cells by raising intracellular levels of camp. Subsequent PKA activation results in CREB phosphorylation and downstream induction of PGC1a to upregulate genes associated with fatty acid oxidation and gluconeogenesis. Norepinephrine interacts with beta adrenergic GPCRs and causes the alpha subunit to activate adenylyl cyclase to convert ATP to camp. Glucagon works similarly to increase camp production by interacting with a different GPCR. Forskolin directly interacts with adenylyl cyclase at its catalytic domain to increase camp production. 8-Br-cAMP is a lipophilic synthetic analog that can enter cells, is resistant to cellular phosphodiesterases, and can directly activate PKA. 10
17 While all of these hormones have different mechanisms of action, they were expected to elicit similar increases in the expression of fatty acid oxidation genes in control hepatocytes. Not only did all of the treatments have inconsistent effects on increasing fatty acid oxidation gene expression, norepinephrine treatment actually caused a decrease in Cpt1a and Ppara expression in control hepatocytes. The Acsl4 L -/- hepatocytes did exhibit different expression patterns compared to control, however results were inconclusive due to the inconsistent effects observed in the control samples. Overall, the hormone treatments did not produce the anticipated effects and did not validate the findings of our previous study. Because of this, we were unable to form conclusions about whether the liver specific ACSL4 knockout decreased beta adrenergic signaling or other exogenous mechanisms of FA oxidation gene regulation. 4.3 Fatty acid treatment effects on gene expression This study also explored the role of fatty acids as intracellular signaling molecules to modulate fatty acid oxidation. Oleate increases intracellular levels of camp and expression of fatty acid oxidation genes in C2C12 myotube cells. 14 While the mechanism behind how oleate elicits this cellular response is unknown, this finding illustrates the potential for therapeutic manipulation of intracellular lipid signaling to alter fatty acid oxidation through transcriptional regulation. Similar to the results from hormone treatment experiments, the gene expression changes following the lipid treatments in our study were not consistent with the findings of the published study. Oleate treatments were expected to increase expression of fatty acid oxidation genes while palmitate was not expected to affect expression. In control hepatocytes palmitate treatment increased gene expression of all genes of interest except Pgc-1a while oleate treatment only increased Creb expression. Similar to what was observed in the hormone treatment experiment, the ACSL4 knockout seemed to alter the cellular responses to treatments. Because the published study observed this signaling effect of oleate in C2C12 myotubes, it is possible that there may be a different effect in primary hepatocytes. This experiment must be repeated in order to draw conclusions on how different long chain fatty acids modulate fatty acid oxidation in hepatocytes and how the absence of ACSL4 alters this process. 11
18 4.4 Future Directions In an attempt to validate the findings of the previous study, most of the results from the hormone treatment experiment were inconclusive. Several steps can be taken to optimize treatment conditions to improve consistency of gene expression changes. Due to different mechanisms of action, each treatment could induce changes in gene expression at different rates. Time course experiments would allow us to optimize hormone treatment times for our study and better determine the effects of the ACSL4 knock out on gene expression. Concentration of the hormone treatments were based on the previous study and prior literature but could also be optimized in the context of our experiment. The efficacy of each treatment could also be validated by using immunoassays to measure intracellular camp concentrations. To further explore how each individual treatment or ACSL4 knockout affected gene expression, covalent modifications of CREB, PGC-1a, and SIRT-1 could be measured using western blot. We also plan to repeat the fatty acid treatment experiments to determine the role of fatty acid signaling in modulating gene expression in primary hepatocytes. 12
19 8 Cpt1a Oleate and Palmitate Treatment Creb Acox1 Pgc1a Ppara Relative mrna (B-actin) Oleate OA WT OA KO PA WT PA KO OA WT OA KO PA WT PA KO OA WT OA KO PA WT PA KO OA WT OA KO PA WT PA KO OA WT OA KO PA WT PA KO Palmitate Oleate Palmitate Oleate Palmitate Oleate Palmitate Oleate Palmitate Figure 1. Lipid Treatments Gene Expression (Experiment 1) Relative gene expression (Actin) of Cpt1a, Creb, Acox1, Pgc1a, and Ppara in ACSL4 L -/- and WT hepatocytes following 3.5-hour oleate or palmitate treatment. Oleate and Palmitate Treatment 2.5 Cpt1a Creb Acox1 Pgc1a Ppara Relative mrna (B-actin) OA WT Oleate OA KO PA WT PA KO OA WT OA KO PA WT PA KO OA WT OA KO PA WT PA KO OA WT OA KO PA WT PA KO OA WT OA KO PA WT PA KO Palmitate Oleate Palmitate Oleate Palmitate Oleate Palmitate Oleate Palmitate Figure 2. Lipid Treatments Gene Expression (Experiment 2) Relative gene expression (Actin) of Cpt1a, Creb, Acox1, Pgc1a, and Ppara in ACSL4 L -/- and WT hepatocytes following 3.5-hour oleate or palmitate treatment. 13
20 Figure 3. Hormone Treatment Gene Expression (Experiment 1) Relative gene expression (Actin) of Pgc1a, Cpt1a, and Ppara following 1-hour (A) norepinephrine, (B) 8-Br-cAMP, and (C) forskolin treatments. Relative expression of Pepck, Pdk4, and G6pase following 1 hour (D) glucagon treatment 14
21 Figure 4. Hormone Treatment Gene Expression (Experiment 2) Relative gene expression (Actin) of Pgc1a, Cpt1a, and Ppara following 1-hour (A) norepinephrine, (B) 8-Br-cAMP, and (C) forskolin treatments. Relative expression of Pepck, Pdk4, and G6pase following 1-hour (D) glucagon treatment 15
22 Figure 5. Hormone Treatments and their Signaling Mechanisms 16
23 Treatment Vehicle Concentration Time (Hr) Norepinephrine H2O 10µM 1 8-Br-cAMP H2O 1mM 1 Forskolin DMSO 40µM 1 Glucagon PBS 100mM 1 Oleate BSA 500µM 3.5 Palmitate BSA 500µM 3.5 Table 1. Treatment and Corresponding Vehicle Conditions 17
24 Target Direction Sequence Acsl4 Forward ACAGCCTTGGAACCCGGGAGA Reverse CCCAATGCAGTGAGGCCACTTCC β-actin Forward GGCTCCTAGCACCATGAAGA Reverse GAAAGGGTGTAAAACGCAGC Pgc-1α Forward CAACATGCTCAAGCCAAACCAACA Reverse CGCTCAATAGTCTTGTTCTCAAATGGG Pparα Forward CCTCAGGGTACCACTACGGAGTT Reverse TCGCCGAAAGAAGCCCTTA Cpt1a Forward CAACTATTATGCCATGGATTTTGTGCTT Reverse CGATACATGATCATGGCGTGAACG Creb Forward TCAGCCGGGTACTACCATTC Reverse TTCAGCAGGCTGTGTAGGAA Acox1 Forward GGTGGACCTCTGTCTTGTTCA Reverse AAACCTTCAGGCCCAAGTGAG Pepck Forward TCAACACCGACCTCCCTTAC Reverse CATTGTGCCGCTATCTCAAA G6pase Forward TCTGTCCCGGATCTACCTTG Reverse TGGCAAAGGGTGTAGTGTCA Pdk4 Forward CCGCTGTCCATGAAGCA Reverse GCAGAAAAGCAAAGGACGTT Table 2. RT-PCR Primer Sequences 18
25 References 1. Nguyen, P. et al. Liver lipid metabolism. J. Anim. Physiol. Anim. Nutr. (Berl). 92, (2008). 2. Rakhshandehroo, M., Knoch, B., Müller, M. & Kersten, S. Peroxisome proliferatoractivated receptor alpha target genes. PPAR Res. 2010, (2010). 3. Küch, E. M. et al. Differentially localized acyl-coa synthetase 4 isoenzymes mediate the metabolic channeling of fatty acids towards phosphatidylinositol. Biochim. Biophys. Acta - Mol. Cell Biol. Lipids 1841, (2014). 4. Piccini, M. et al. FACL4, a New Gene Encoding Long-Chain Acyl-CoA Synthetase 4, Is Deleted in a Family with Alport Syndrome, Elliptocytosis, and Mental Retardation. Genomics 47, (1998). 5. Piccini, M. et al. FACL4, a New Gene Encoding Long-Chain Acyl-CoA Synthetase 4, Is Deleted in a Family with Alport Syndrome, Elliptocytosis, and Mental Retardation. Genomics 47, (1998). 6. Kang, M. J. et al. A novel arachidonate-preferring acyl-coa synthetase is present in steroidogenic cells of the rat adrenal, ovary, and testis. Proc. Natl. Acad. Sci. U. S. A. 94, (1997). 7. Cao, Y., Murphy, K. J., McIntyre, T. M., Zimmerman, G. A. & Prescott, S. M. Expression of fatty acid-coa ligase 4 during development and in brain. FEBS Lett. 467, (2000). 8. Kudo, T., Tamagawa, T., Kawashima, M., Mito, N. & Shibata, S. Attenuating Effect of Clock Mutation on Triglyceride Contents in the ICR Mouse Liver under a High-Fat Diet. J. Biol. Rhythms 22, (2007). 9. Kotronen, A. et al. Genetic variation in the ADIPOR2 gene is associated with liver fat content and its surrogate markers in three independent cohorts. Eur. J. Endocrinol. 160, (2009). 10. Sung, Y. K. et al. Fatty acid-coa ligase 4 is overexpressed in human hepatocellular carcinoma. Cancer Sci. 94, (2003). 11. Liang, Y.-C. et al. Involvement of fatty acid-coa ligase 4 in hepatocellular carcinoma growth: roles of cyclic AMP and p38 mitogen-activated protein kinase. World J. 19
26 Gastroenterol. 11, (2005). 12. Puigserver, P. & Spiegelman, B. M. Peroxisome Proliferator-Activated Receptor-γ Coactivator 1α (PGC-1α): Transcriptional Coactivator and Metabolic Regulator. Endocr. Rev. 24, (2003). 13. Lim, J.-H. et al. Oleic acid stimulates complete oxidation of fatty acids through protein kinase A-dependent activation of SIRT1-PGC1α complex. J. Biol. Chem. 288, (2013). 14. Lim, J.-H. et al. Oleic acid stimulates complete oxidation of fatty acids through protein kinase A-dependent activation of SIRT1-PGC1α complex. J. Biol. Chem. 288, (2013). 20
Supplementary Figure 1.
 Supplementary Figure 1. FGF21 does not exert direct effects on hepatic glucose production. The liver explants from C57BL/6J mice (A, B) or primary rat hepatocytes (C, D) were incubated with rmfgf21 (2
Supplementary Figure 1. FGF21 does not exert direct effects on hepatic glucose production. The liver explants from C57BL/6J mice (A, B) or primary rat hepatocytes (C, D) were incubated with rmfgf21 (2
BCM 221 LECTURES OJEMEKELE O.
 BCM 221 LECTURES BY OJEMEKELE O. OUTLINE INTRODUCTION TO LIPID CHEMISTRY STORAGE OF ENERGY IN ADIPOCYTES MOBILIZATION OF ENERGY STORES IN ADIPOCYTES KETONE BODIES AND KETOSIS PYRUVATE DEHYDROGENASE COMPLEX
BCM 221 LECTURES BY OJEMEKELE O. OUTLINE INTRODUCTION TO LIPID CHEMISTRY STORAGE OF ENERGY IN ADIPOCYTES MOBILIZATION OF ENERGY STORES IN ADIPOCYTES KETONE BODIES AND KETOSIS PYRUVATE DEHYDROGENASE COMPLEX
Supplemental Table 1 Primer sequences (mouse) used for real-time qrt-pcr studies
 Supplemental Table 1 Primer sequences (mouse) used for real-time qrt-pcr studies Gene symbol Forward primer Reverse primer ACC1 5'-TGAGGAGGACCGCATTTATC 5'-GCATGGAATGGCAGTAAGGT ACLY 5'-GACACCATCTGTGATCTTG
Supplemental Table 1 Primer sequences (mouse) used for real-time qrt-pcr studies Gene symbol Forward primer Reverse primer ACC1 5'-TGAGGAGGACCGCATTTATC 5'-GCATGGAATGGCAGTAAGGT ACLY 5'-GACACCATCTGTGATCTTG
LIPID METABOLISM. Sri Widia A Jusman Department of Biochemistry & Molecular Biology FMUI
 LIPID METABOLISM Sri Widia A Jusman Department of Biochemistry & Molecular Biology FMUI Lipid metabolism is concerned mainly with fatty acids cholesterol Source of fatty acids from dietary fat de novo
LIPID METABOLISM Sri Widia A Jusman Department of Biochemistry & Molecular Biology FMUI Lipid metabolism is concerned mainly with fatty acids cholesterol Source of fatty acids from dietary fat de novo
CHM333 LECTURE 34: 11/30 12/2/09 FALL 2009 Professor Christine Hrycyna
 Lipid Metabolism β-oxidation FA Acetyl-CoA Triacylglycerols (TAGs) and glycogen are the two major forms of stored energy in vertebrates Glycogen can supply ATP for muscle contraction for less than an hour
Lipid Metabolism β-oxidation FA Acetyl-CoA Triacylglycerols (TAGs) and glycogen are the two major forms of stored energy in vertebrates Glycogen can supply ATP for muscle contraction for less than an hour
BIOL2171 ANU TCA CYCLE
 TCA CYCLE IMPORTANCE: Oxidation of 2C Acetyl Co-A 2CO 2 + 3NADH + FADH 2 (8e-s donated to O 2 in the ETC) + GTP (energy) + Heat OVERVIEW: Occurs In the mitochondrion matrix. 1. the acetyl portion of acetyl-coa
TCA CYCLE IMPORTANCE: Oxidation of 2C Acetyl Co-A 2CO 2 + 3NADH + FADH 2 (8e-s donated to O 2 in the ETC) + GTP (energy) + Heat OVERVIEW: Occurs In the mitochondrion matrix. 1. the acetyl portion of acetyl-coa
Oxidation of Long Chain Fatty Acids
 Oxidation of Long Chain Fatty Acids Dr NC Bird Oxidation of long chain fatty acids is the primary source of energy supply in man and animals. Hibernating animals utilise fat stores to maintain body heat,
Oxidation of Long Chain Fatty Acids Dr NC Bird Oxidation of long chain fatty acids is the primary source of energy supply in man and animals. Hibernating animals utilise fat stores to maintain body heat,
number Done by Corrected by Doctor Faisal Al-Khatibe
 number 24 Done by Mohammed tarabieh Corrected by Doctor Faisal Al-Khatibe 1 P a g e *Please look over the previous sheet about fatty acid synthesis **Oxidation(degradation) of fatty acids, occurs in the
number 24 Done by Mohammed tarabieh Corrected by Doctor Faisal Al-Khatibe 1 P a g e *Please look over the previous sheet about fatty acid synthesis **Oxidation(degradation) of fatty acids, occurs in the
Lipids digestion and absorption, Biochemistry II
 Lipids digestion and absorption, blood plasma lipids, lipoproteins Biochemistry II Lecture 1 2008 (J.S.) Triacylglycerols (as well as free fatty acids and both free and esterified cholesterol) are very
Lipids digestion and absorption, blood plasma lipids, lipoproteins Biochemistry II Lecture 1 2008 (J.S.) Triacylglycerols (as well as free fatty acids and both free and esterified cholesterol) are very
Chapter 26 Biochemistry 5th edition. phospholipids. Sphingolipids. Cholesterol. db=books&itool=toolbar
 http://www.ncbi.nlm.nih.gov/sites/entrez? db=books&itool=toolbar 1 The surface of a soap bubble is a bilayer formed by detergent molecules 2 Chapter 26 Biochemistry 5th edition phospholipids Sphingolipids
http://www.ncbi.nlm.nih.gov/sites/entrez? db=books&itool=toolbar 1 The surface of a soap bubble is a bilayer formed by detergent molecules 2 Chapter 26 Biochemistry 5th edition phospholipids Sphingolipids
Biochemistry: A Short Course
 Tymoczko Berg Stryer Biochemistry: A Short Course Second Edition CHAPTER 28 Fatty Acid Synthesis 2013 W. H. Freeman and Company Chapter 28 Outline 1. The first stage of fatty acid synthesis is transfer
Tymoczko Berg Stryer Biochemistry: A Short Course Second Edition CHAPTER 28 Fatty Acid Synthesis 2013 W. H. Freeman and Company Chapter 28 Outline 1. The first stage of fatty acid synthesis is transfer
23.1 Lipid Metabolism in Animals. Chapter 23. Micelles Lipid Metabolism in. Animals. Overview of Digestion Lipid Metabolism in
 Denniston Topping Caret Copyright! The McGraw-Hill Companies, Inc. Permission required for reproduction or display. Chapter 23 Fatty Acid Metabolism Triglycerides (Tgl) are emulsified into fat droplets
Denniston Topping Caret Copyright! The McGraw-Hill Companies, Inc. Permission required for reproduction or display. Chapter 23 Fatty Acid Metabolism Triglycerides (Tgl) are emulsified into fat droplets
OVERVIEW M ET AB OL IS M OF FR EE FA TT Y AC ID S
 LIPOLYSIS LIPOLYSIS OVERVIEW CATABOLISM OF FREE FATTY ACIDS Nonesterified fatty acids Source:- (a) breakdown of TAG in adipose tissue (b) action of Lipoprotein lipase on plasma TAG Combined with Albumin
LIPOLYSIS LIPOLYSIS OVERVIEW CATABOLISM OF FREE FATTY ACIDS Nonesterified fatty acids Source:- (a) breakdown of TAG in adipose tissue (b) action of Lipoprotein lipase on plasma TAG Combined with Albumin
Lipid Metabolism. Remember fats?? Triacylglycerols - major form of energy storage in animals
 Remember fats?? Triacylglycerols - major form of energy storage in animals Your energy reserves: ~0.5% carbs (glycogen + glucose) ~15% protein (muscle, last resort) ~85% fat Why use fat for energy? 1 gram
Remember fats?? Triacylglycerols - major form of energy storage in animals Your energy reserves: ~0.5% carbs (glycogen + glucose) ~15% protein (muscle, last resort) ~85% fat Why use fat for energy? 1 gram
Summary of fatty acid synthesis
 Lipid Metabolism, part 2 1 Summary of fatty acid synthesis 8 acetyl CoA + 14 NADPH + 14 H+ + 7 ATP palmitic acid (16:0) + 8 CoA + 14 NADP + + 7 ADP + 7 Pi + 7 H20 1. The major suppliers of NADPH for fatty
Lipid Metabolism, part 2 1 Summary of fatty acid synthesis 8 acetyl CoA + 14 NADPH + 14 H+ + 7 ATP palmitic acid (16:0) + 8 CoA + 14 NADP + + 7 ADP + 7 Pi + 7 H20 1. The major suppliers of NADPH for fatty
Unit IV Problem 3 Biochemistry: Cholesterol Metabolism and Lipoproteins
 Unit IV Problem 3 Biochemistry: Cholesterol Metabolism and Lipoproteins - Cholesterol: It is a sterol which is found in all eukaryotic cells and contains an oxygen (as a hydroxyl group OH) on Carbon number
Unit IV Problem 3 Biochemistry: Cholesterol Metabolism and Lipoproteins - Cholesterol: It is a sterol which is found in all eukaryotic cells and contains an oxygen (as a hydroxyl group OH) on Carbon number
Biochemistry: A Short Course
 Tymoczko Berg Stryer Biochemistry: A Short Course Second Edition CHAPTER 27 Fatty Acid Degradation Dietary Lipid (Triacylglycerol) Metabolism - In the small intestine, fat particles are coated with bile
Tymoczko Berg Stryer Biochemistry: A Short Course Second Edition CHAPTER 27 Fatty Acid Degradation Dietary Lipid (Triacylglycerol) Metabolism - In the small intestine, fat particles are coated with bile
Roles of Lipids. principal form of stored energy major constituents of cell membranes vitamins messengers intra and extracellular
 Roles of Lipids principal form of stored energy major constituents of cell membranes vitamins messengers intra and extracellular = Oxidation of fatty acids Central energy-yielding pathway in animals. O
Roles of Lipids principal form of stored energy major constituents of cell membranes vitamins messengers intra and extracellular = Oxidation of fatty acids Central energy-yielding pathway in animals. O
Fatty acids synthesis
 Fatty acids synthesis The synthesis start from Acetyl COA the first step requires ATP + reducing power NADPH! even though the oxidation and synthesis are different pathways but from chemical part of view
Fatty acids synthesis The synthesis start from Acetyl COA the first step requires ATP + reducing power NADPH! even though the oxidation and synthesis are different pathways but from chemical part of view
Dietary Lipid Metabolism
 Dietary Lipid Metabolism Presented by Dr. Mohammad Saadeh The requirements for the Pharmaceutical Biochemistry II Philadelphia University Faculty of pharmacy OVERVIEW Lipids are a heterogeneous group.
Dietary Lipid Metabolism Presented by Dr. Mohammad Saadeh The requirements for the Pharmaceutical Biochemistry II Philadelphia University Faculty of pharmacy OVERVIEW Lipids are a heterogeneous group.
ANSC/NUTR 618 LIPIDS & LIPID METABOLISM. Triacylglycerol and Fatty Acid Metabolism
 ANSC/NUTR 618 LIPIDS & LIPID METABOLISM II. Triacylglycerol synthesis A. Overall pathway Glycerol-3-phosphate + 3 Fatty acyl-coa à Triacylglycerol + 3 CoASH B. Enzymes 1. Acyl-CoA synthase 2. Glycerol-phosphate
ANSC/NUTR 618 LIPIDS & LIPID METABOLISM II. Triacylglycerol synthesis A. Overall pathway Glycerol-3-phosphate + 3 Fatty acyl-coa à Triacylglycerol + 3 CoASH B. Enzymes 1. Acyl-CoA synthase 2. Glycerol-phosphate
MILK BIOSYNTHESIS PART 3: FAT
 MILK BIOSYNTHESIS PART 3: FAT KEY ENZYMES (FROM ALL BIOSYNTHESIS LECTURES) FDPase = fructose diphosphatase Citrate lyase Isocitrate dehydrogenase Fatty acid synthetase Acetyl CoA carboxylase Fatty acyl
MILK BIOSYNTHESIS PART 3: FAT KEY ENZYMES (FROM ALL BIOSYNTHESIS LECTURES) FDPase = fructose diphosphatase Citrate lyase Isocitrate dehydrogenase Fatty acid synthetase Acetyl CoA carboxylase Fatty acyl
Lecture 36. Key Concepts. Overview of lipid metabolism. Reactions of fatty acid oxidation. Energy yield from fatty acid oxidation
 Lecture 36 Lipid Metabolism 1 Fatty Acid Oxidation Ketone Bodies Key Concepts Overview of lipid metabolism Reactions of fatty acid oxidation Energy yield from fatty acid oxidation Formation of ketone bodies
Lecture 36 Lipid Metabolism 1 Fatty Acid Oxidation Ketone Bodies Key Concepts Overview of lipid metabolism Reactions of fatty acid oxidation Energy yield from fatty acid oxidation Formation of ketone bodies
BIOL212 Biochemistry of Disease. Metabolic Disorders - Obesity
 BIOL212 Biochemistry of Disease Metabolic Disorders - Obesity Obesity Approx. 23% of adults are obese in the U.K. The number of obese children has tripled in 20 years. 10% of six year olds are obese, rising
BIOL212 Biochemistry of Disease Metabolic Disorders - Obesity Obesity Approx. 23% of adults are obese in the U.K. The number of obese children has tripled in 20 years. 10% of six year olds are obese, rising
THE GLUCOSE-FATTY ACID-KETONE BODY CYCLE Role of ketone bodies as respiratory substrates and metabolic signals
 Br. J. Anaesth. (1981), 53, 131 THE GLUCOSE-FATTY ACID-KETONE BODY CYCLE Role of ketone bodies as respiratory substrates and metabolic signals J. C. STANLEY In this paper, the glucose-fatty acid cycle
Br. J. Anaesth. (1981), 53, 131 THE GLUCOSE-FATTY ACID-KETONE BODY CYCLE Role of ketone bodies as respiratory substrates and metabolic signals J. C. STANLEY In this paper, the glucose-fatty acid cycle
Fatty Acid and Triacylglycerol Metabolism 1
 Fatty Acid and Triacylglycerol Metabolism 1 Mobilization of stored fats and oxidation of fatty acids Lippincott s Chapter 16 What is the first lecture about What is triacylglycerol Fatty acids structure
Fatty Acid and Triacylglycerol Metabolism 1 Mobilization of stored fats and oxidation of fatty acids Lippincott s Chapter 16 What is the first lecture about What is triacylglycerol Fatty acids structure
Intermediary metabolism. Eva Samcová
 Intermediary metabolism Eva Samcová Metabolic roles of tissues Four major tissues play a dominant role in fuel metabolism : liver, adipose, muscle, and brain. These tissues do not function in isolation.
Intermediary metabolism Eva Samcová Metabolic roles of tissues Four major tissues play a dominant role in fuel metabolism : liver, adipose, muscle, and brain. These tissues do not function in isolation.
Biosynthesis of Fatty Acids. By Dr.QUTAIBA A. QASIM
 Biosynthesis of Fatty Acids By Dr.QUTAIBA A. QASIM Fatty Acids Definition Fatty acids are comprised of hydrocarbon chains terminating with carboxylic acid groups. Fatty acids and their associated derivatives
Biosynthesis of Fatty Acids By Dr.QUTAIBA A. QASIM Fatty Acids Definition Fatty acids are comprised of hydrocarbon chains terminating with carboxylic acid groups. Fatty acids and their associated derivatives
Fatty Acid and Triacylglycerol Metabolism 1
 Fatty Acid and Triacylglycerol Metabolism 1 Mobilization of stored fats and oxidation of fatty acids Lippincott s Chapter 16 What is the first lecture about What is triacylglycerol Fatty acids structure
Fatty Acid and Triacylglycerol Metabolism 1 Mobilization of stored fats and oxidation of fatty acids Lippincott s Chapter 16 What is the first lecture about What is triacylglycerol Fatty acids structure
Lipid Metabolism * OpenStax
 OpenStax-CNX module: m46462 1 Lipid Metabolism * OpenStax This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License 3.0 By the end of this section, you will be able
OpenStax-CNX module: m46462 1 Lipid Metabolism * OpenStax This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License 3.0 By the end of this section, you will be able
Gene Polymorphisms and Carbohydrate Diets. James M. Ntambi Ph.D
 Gene Polymorphisms and Carbohydrate Diets James M. Ntambi Ph.D Fatty Acids that Flux into Tissue Lipids are from Dietary Sources or are Made De novo from Glucose or Fructose Glucose Fructose Acetyl-CoA
Gene Polymorphisms and Carbohydrate Diets James M. Ntambi Ph.D Fatty Acids that Flux into Tissue Lipids are from Dietary Sources or are Made De novo from Glucose or Fructose Glucose Fructose Acetyl-CoA
Metabolism of acylglycerols and sphingolipids. Martina Srbová
 Metabolism of acylglycerols and sphingolipids Martina Srbová Types of glycerolipids and sphingolipids 1. Triacylglycerols function as energy reserves adipose tissue (storage of triacylglycerol), lipoproteins
Metabolism of acylglycerols and sphingolipids Martina Srbová Types of glycerolipids and sphingolipids 1. Triacylglycerols function as energy reserves adipose tissue (storage of triacylglycerol), lipoproteins
Synthesis of Fatty Acids and Triacylglycerol
 Synthesis of Fatty Acids and Triacylglycerol Lippincott s Chapter 16 Fatty Acid Synthesis Mainly in the Liver Requires Carbon Source: Acetyl CoA Reducing Power: NADPH 8 CH 3 COO C 15 H 33 COO Energy Input:
Synthesis of Fatty Acids and Triacylglycerol Lippincott s Chapter 16 Fatty Acid Synthesis Mainly in the Liver Requires Carbon Source: Acetyl CoA Reducing Power: NADPH 8 CH 3 COO C 15 H 33 COO Energy Input:
Bio 366: Biological Chemistry II Test #1, 100 points (7 pages)
 Bio 366: Biological Chemistry II Test #1, 100 points (7 pages) READ THIS: Take a numbered test and sit in the seat with that number on it. Remove the numbered sticker from the desk, and stick it on the
Bio 366: Biological Chemistry II Test #1, 100 points (7 pages) READ THIS: Take a numbered test and sit in the seat with that number on it. Remove the numbered sticker from the desk, and stick it on the
Chapter 16 - Lipid Metabolism
 Chapter 16 - Lipid Metabolism Fatty acids have four major physiologic roles in the cell: Building blocks of phospholipids and glycolipids Added onto proteins to create lipoproteins, which targets them
Chapter 16 - Lipid Metabolism Fatty acids have four major physiologic roles in the cell: Building blocks of phospholipids and glycolipids Added onto proteins to create lipoproteins, which targets them
Metabolism of cardiac muscle. Dr. Mamoun Ahram Cardiovascular system, 2013
 Metabolism of cardiac muscle Dr. Mamoun Ahram Cardiovascular system, 2013 References This lecture Mark s Basic Medical Biochemistry, 4 th ed., p. 890-891 Hand-out Why is this topic important? Heart failure
Metabolism of cardiac muscle Dr. Mamoun Ahram Cardiovascular system, 2013 References This lecture Mark s Basic Medical Biochemistry, 4 th ed., p. 890-891 Hand-out Why is this topic important? Heart failure
LIPID METABOLISM
 LIPID METABOLISM LIPOGENESIS LIPOGENESIS LIPOGENESIS FATTY ACID SYNTHESIS DE NOVO FFA in the blood come from :- (a) Dietary fat (b) Dietary carbohydrate/protein in excess of need FA TAG Site of synthesis:-
LIPID METABOLISM LIPOGENESIS LIPOGENESIS LIPOGENESIS FATTY ACID SYNTHESIS DE NOVO FFA in the blood come from :- (a) Dietary fat (b) Dietary carbohydrate/protein in excess of need FA TAG Site of synthesis:-
CHY2026: General Biochemistry. Lipid Metabolism
 CHY2026: General Biochemistry Lipid Metabolism Lipid Digestion Lipid Metabolism Fats (triglycerides) are high metabolic energy molecules Fats yield 9.3 kcal of energy (carbohydrates and proteins 4.1 kcal)
CHY2026: General Biochemistry Lipid Metabolism Lipid Digestion Lipid Metabolism Fats (triglycerides) are high metabolic energy molecules Fats yield 9.3 kcal of energy (carbohydrates and proteins 4.1 kcal)
Lipid Metabolism. Catabolism Overview
 Lipid Metabolism Pratt & Cornely, Chapter 17 Catabolism Overview Lipids as a fuel source from diet Beta oxidation Mechanism ATP production Ketone bodies as fuel 1 High energy More reduced Little water
Lipid Metabolism Pratt & Cornely, Chapter 17 Catabolism Overview Lipids as a fuel source from diet Beta oxidation Mechanism ATP production Ketone bodies as fuel 1 High energy More reduced Little water
Nafith Abu Tarboush DDS, MSc, PhD
 Nafith Abu Tarboush DDS, MSc, PhD natarboush@ju.edu.jo www.facebook.com/natarboush Lipids (cholesterol, cholesterol esters, phospholipids & triacylglycerols) combined with proteins (apolipoprotein) in
Nafith Abu Tarboush DDS, MSc, PhD natarboush@ju.edu.jo www.facebook.com/natarboush Lipids (cholesterol, cholesterol esters, phospholipids & triacylglycerols) combined with proteins (apolipoprotein) in
Synthesis of Fatty Acids and Triacylglycerol
 Fatty Acid Synthesis Synthesis of Fatty Acids and Triacylglycerol Requires Carbon Source: Reducing Power: NADPH Energy Input: ATP Why Energy? Why Energy? Fatty Acid Fatty Acid + n(atp) ΔG o : -ve Fatty
Fatty Acid Synthesis Synthesis of Fatty Acids and Triacylglycerol Requires Carbon Source: Reducing Power: NADPH Energy Input: ATP Why Energy? Why Energy? Fatty Acid Fatty Acid + n(atp) ΔG o : -ve Fatty
Chapter 22, Fatty Acid Metabolism CH 3 (CH 2 ) 14 CO 2 R C C O2 CH 2 OH O R. Lipase + 3 H 2 O
 hapter 22, Fatty Acid Metabolism Pages: 603-613 I. Introduction - Fatty acids have 4 major physiological roles: - omponents of phospholipids and glycolipids (membranes) - Attachment to Proteins targets
hapter 22, Fatty Acid Metabolism Pages: 603-613 I. Introduction - Fatty acids have 4 major physiological roles: - omponents of phospholipids and glycolipids (membranes) - Attachment to Proteins targets
Integration Of Metabolism
 Integration Of Metabolism Metabolism Consist of Highly Interconnected Pathways The basic strategy of catabolic metabolism is to form ATP, NADPH, and building blocks for biosyntheses. 1. ATP is the universal
Integration Of Metabolism Metabolism Consist of Highly Interconnected Pathways The basic strategy of catabolic metabolism is to form ATP, NADPH, and building blocks for biosyntheses. 1. ATP is the universal
Leen Alsahele. Razan Al-zoubi ... Faisal
 25 Leen Alsahele Razan Al-zoubi... Faisal last time we started talking about regulation of fatty acid synthesis and degradation *regulation of fatty acid synthesis by: 1- regulation of acetyl CoA carboxylase
25 Leen Alsahele Razan Al-zoubi... Faisal last time we started talking about regulation of fatty acid synthesis and degradation *regulation of fatty acid synthesis by: 1- regulation of acetyl CoA carboxylase
Medical Biochemistry and Molecular Biology department
 Medical Biochemistry and Molecular Biology department Cardiac Fuels [Sources of energy for the Cardiac muscle] Intended learning outcomes of the lecture: By the end of this lecture you would be able to:-
Medical Biochemistry and Molecular Biology department Cardiac Fuels [Sources of energy for the Cardiac muscle] Intended learning outcomes of the lecture: By the end of this lecture you would be able to:-
Companion to Biosynthesis of Ketones & Cholesterols, Regulation of Lipid Metabolism Lecture Notes
 Companion to Biosynthesis of Ketones & Cholesterols, Regulation of Lipid Metabolism Lecture Notes The major site of acetoacetate and 3-hydorxybutyrate production is in the liver. 3-hydorxybutyrate is the
Companion to Biosynthesis of Ketones & Cholesterols, Regulation of Lipid Metabolism Lecture Notes The major site of acetoacetate and 3-hydorxybutyrate production is in the liver. 3-hydorxybutyrate is the
Biochemistry - I SPRING Mondays and Wednesdays 9:30-10:45 AM (MR-1307) Lectures Based on Profs. Kevin Gardner & Reza Khayat
 Biochemistry - I Mondays and Wednesdays 9:30-10:45 AM (MR-1307) SPRING 2017 Lectures 21-22 Based on Profs. Kevin Gardner & Reza Khayat 1 Outline Vertebrate processing of dietary lipids Mobilization of
Biochemistry - I Mondays and Wednesdays 9:30-10:45 AM (MR-1307) SPRING 2017 Lectures 21-22 Based on Profs. Kevin Gardner & Reza Khayat 1 Outline Vertebrate processing of dietary lipids Mobilization of
Modifications of Pyruvate Handling in Health and Disease Prof. Mary Sugden
 Modifications of Handling Modifications of Handling Centre for Diabetes and Metabolic Medicine Institute of Cell and Molecular Science Barts and the London School of Medicine and Dentistry 1 Potential
Modifications of Handling Modifications of Handling Centre for Diabetes and Metabolic Medicine Institute of Cell and Molecular Science Barts and the London School of Medicine and Dentistry 1 Potential
6. How Are Fatty Acids Produced? 7. How Are Acylglycerols and Compound Lipids Produced? 8. How Is Cholesterol Produced?
 Lipid Metabolism Learning bjectives 1 How Are Lipids Involved in the Generationand Storage of Energy? 2 How Are Lipids Catabolized? 3 What Is the Energy Yield from the xidation of Fatty Acids? 4 How Are
Lipid Metabolism Learning bjectives 1 How Are Lipids Involved in the Generationand Storage of Energy? 2 How Are Lipids Catabolized? 3 What Is the Energy Yield from the xidation of Fatty Acids? 4 How Are
Biosynthesis of Fatty Acids
 Biosynthesis of Fatty Acids Fatty acid biosynthesis takes place in the cytosol rather than the mitochondria and requires a different activation mechanism and different enzymes and coenzymes than fatty
Biosynthesis of Fatty Acids Fatty acid biosynthesis takes place in the cytosol rather than the mitochondria and requires a different activation mechanism and different enzymes and coenzymes than fatty
ANSC/NUTR 618 Lipids & Lipid Metabolism
 I. Overall concepts A. Definitions ANC/NUTR 618 Lipids & Lipid Metabolism 1. De novo synthesis = synthesis from non-fatty acid precursors a. Carbohydrate precursors (glucose, lactate, and pyruvate) b.
I. Overall concepts A. Definitions ANC/NUTR 618 Lipids & Lipid Metabolism 1. De novo synthesis = synthesis from non-fatty acid precursors a. Carbohydrate precursors (glucose, lactate, and pyruvate) b.
Dietary α-linolenic acid-rich flaxseed oil prevents against alcoholic hepatic steatosis
 Dietary α-linolenic acid-rich flaxseed oil prevents against alcoholic hepatic steatosis via ameliorating lipid homeostasis at adipose tissue-liver axis in mice Meng Wang a, Xiao-Jing Zhang a, Kun Feng
Dietary α-linolenic acid-rich flaxseed oil prevents against alcoholic hepatic steatosis via ameliorating lipid homeostasis at adipose tissue-liver axis in mice Meng Wang a, Xiao-Jing Zhang a, Kun Feng
Fatty acid breakdown
 Fatty acids contain a long hydrocarbon chain and a terminal carboxylate group. Most contain between 14 and 24 carbon atoms. The chains may be saturated or contain double bonds. The complete oxidation of
Fatty acids contain a long hydrocarbon chain and a terminal carboxylate group. Most contain between 14 and 24 carbon atoms. The chains may be saturated or contain double bonds. The complete oxidation of
Voet Biochemistry 3e John Wiley & Sons, Inc.
 * * Voet Biochemistry 3e Lipid Metabolism Part I: (Chap. 25, sec.1-3) Glucose C 6 H 12 O 6 + 6 O 2 6 CO 2 + 6 H 2 O G o = -2823 kj/mol Fats (palmitic acid) C 16 H 32 O 2 + 23 O 2 16 CO 2 + 16 H 2 O G o
* * Voet Biochemistry 3e Lipid Metabolism Part I: (Chap. 25, sec.1-3) Glucose C 6 H 12 O 6 + 6 O 2 6 CO 2 + 6 H 2 O G o = -2823 kj/mol Fats (palmitic acid) C 16 H 32 O 2 + 23 O 2 16 CO 2 + 16 H 2 O G o
Integrative Metabolism: Significance
 Integrative Metabolism: Significance Energy Containing Nutrients Carbohydrates Fats Proteins Catabolism Energy Depleted End Products H 2 O NH 3 ADP + Pi NAD + NADP + FAD + Pi NADH+H + NADPH+H + FADH2 Cell
Integrative Metabolism: Significance Energy Containing Nutrients Carbohydrates Fats Proteins Catabolism Energy Depleted End Products H 2 O NH 3 ADP + Pi NAD + NADP + FAD + Pi NADH+H + NADPH+H + FADH2 Cell
Biosynthesis of Triacylglycerides (TG) in liver. Mobilization of stored fat and oxidation of fatty acids
 Biosynthesis of Triacylglycerides (TG) in liver Mobilization of stored fat and oxidation of fatty acids Activation of hormone sensitive lipase This enzyme is activated when phosphorylated (3,5 cyclic AMPdependent
Biosynthesis of Triacylglycerides (TG) in liver Mobilization of stored fat and oxidation of fatty acids Activation of hormone sensitive lipase This enzyme is activated when phosphorylated (3,5 cyclic AMPdependent
number Done by Corrected by Doctor Nayef Karadsheh
 number 11 Done by حسام أبو عوض Corrected by Moayyad Al-Shafei Doctor Nayef Karadsheh 1 P a g e General Regulatory Aspects in Metabolism: We can divide all pathways in metabolism to catabolicand anabolic.
number 11 Done by حسام أبو عوض Corrected by Moayyad Al-Shafei Doctor Nayef Karadsheh 1 P a g e General Regulatory Aspects in Metabolism: We can divide all pathways in metabolism to catabolicand anabolic.
BIOSYNTHESIS OF FATTY ACIDS. doc. Ing. Zenóbia Chavková, CSc.
 BIOSYNTHESIS OF FATTY ACIDS doc. Ing. Zenóbia Chavková, CSc. The pathway for the of FAs is not the reversal of the oxidation pathway Both pathways are separated within different cellular compartments In
BIOSYNTHESIS OF FATTY ACIDS doc. Ing. Zenóbia Chavková, CSc. The pathway for the of FAs is not the reversal of the oxidation pathway Both pathways are separated within different cellular compartments In
Lecture 15. Signal Transduction Pathways - Introduction
 Lecture 15 Signal Transduction Pathways - Introduction So far.. Regulation of mrna synthesis Regulation of rrna synthesis Regulation of trna & 5S rrna synthesis Regulation of gene expression by signals
Lecture 15 Signal Transduction Pathways - Introduction So far.. Regulation of mrna synthesis Regulation of rrna synthesis Regulation of trna & 5S rrna synthesis Regulation of gene expression by signals
Chapter 10. Introduction to Nutrition and Metabolism, 3 rd edition David A Bender Taylor & Francis Ltd, London 2002
 Chapter 10 Introduction to Nutrition and Metabolism, 3 rd edition David A Bender Taylor & Francis Ltd, London 2002 Chapter 10: Integration and Control of Metabolism Press the space bar or click the mouse
Chapter 10 Introduction to Nutrition and Metabolism, 3 rd edition David A Bender Taylor & Francis Ltd, London 2002 Chapter 10: Integration and Control of Metabolism Press the space bar or click the mouse
The molecule that serves as the major source of readily available body fuel is: a. fat. b. glucose. c. acetyl CoA. d. cellulose.
 The molecule that serves as the major source of readily available body fuel is: a. fat. b. glucose. c. acetyl CoA. d. cellulose. Dietary fats are important because: a. they keep blood pressure normal.
The molecule that serves as the major source of readily available body fuel is: a. fat. b. glucose. c. acetyl CoA. d. cellulose. Dietary fats are important because: a. they keep blood pressure normal.
2-more complex molecules (fatty acyl esters) as triacylglycerols.
 ** Fatty acids exist in two forms:- 1-free fatty acids (unesterified) 2-more complex molecules (fatty acyl esters) as triacylglycerols. ** most tissues might use fatty acids as source of energy during
** Fatty acids exist in two forms:- 1-free fatty acids (unesterified) 2-more complex molecules (fatty acyl esters) as triacylglycerols. ** most tissues might use fatty acids as source of energy during
Moh Tarek + Suhayb. Tamara Al-Azzeh + Asmaa Aljeelani ... Faisal
 28 Moh Tarek + Suhayb Tamara Al-Azzeh + Asmaa Aljeelani... Faisal Digestion of dietary lipids Lipid digestion and absorption are complex processes. They involve soluble enzymes, substrates with different
28 Moh Tarek + Suhayb Tamara Al-Azzeh + Asmaa Aljeelani... Faisal Digestion of dietary lipids Lipid digestion and absorption are complex processes. They involve soluble enzymes, substrates with different
number Done by Corrected by Doctor Nayef Karadsheh
 number 13 Done by Asma Karameh Corrected by Saad hayek Doctor Nayef Karadsheh Gluconeogenesis This lecture covers gluconeogenesis with aspects of: 1) Introduction to glucose distribution through tissues.
number 13 Done by Asma Karameh Corrected by Saad hayek Doctor Nayef Karadsheh Gluconeogenesis This lecture covers gluconeogenesis with aspects of: 1) Introduction to glucose distribution through tissues.
Lehninger 5 th ed. Chapter 17
 Lehninger 5 th ed. Chapter 17 December 26, 2010 Prof. Shimon Schuldiner Email: Shimon.Schuldiner@huji.ac.il Phone: 6585992 CHAPTER 17 Fatty Acid Catabolism Key topics: How fats are digested in animals
Lehninger 5 th ed. Chapter 17 December 26, 2010 Prof. Shimon Schuldiner Email: Shimon.Schuldiner@huji.ac.il Phone: 6585992 CHAPTER 17 Fatty Acid Catabolism Key topics: How fats are digested in animals
Fatty Acid Degradation. Catabolism Overview. TAG and FA 11/11/2015. Chapter 27, Stryer Short Course. Lipids as a fuel source diet Beta oxidation
 Fatty Acid Degradation Chapter 27, Stryer Short Course Catabolism verview Lipids as a fuel source diet Beta oxidation saturated Unsaturated dd chain Ketone bodies as fuel Physiology High energy More reduced
Fatty Acid Degradation Chapter 27, Stryer Short Course Catabolism verview Lipids as a fuel source diet Beta oxidation saturated Unsaturated dd chain Ketone bodies as fuel Physiology High energy More reduced
Energy metabolism - the overview
 Energy metabolism - the overview Josef Fontana EC - 40 Overview of the lecture Important terms of the energy metabolism The overview of the energy metabolism The main pathways of the energy metabolism
Energy metabolism - the overview Josef Fontana EC - 40 Overview of the lecture Important terms of the energy metabolism The overview of the energy metabolism The main pathways of the energy metabolism
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION FOR Liver X Receptor α mediates hepatic triglyceride accumulation through upregulation of G0/G1 Switch Gene 2 (G0S2) expression I: SUPPLEMENTARY METHODS II: SUPPLEMENTARY FIGURES
SUPPLEMENTARY INFORMATION FOR Liver X Receptor α mediates hepatic triglyceride accumulation through upregulation of G0/G1 Switch Gene 2 (G0S2) expression I: SUPPLEMENTARY METHODS II: SUPPLEMENTARY FIGURES
Moh Tarek. Razi Kittaneh. Jaqen H ghar
 14 Moh Tarek Razi Kittaneh Jaqen H ghar Naif Karadsheh Gluconeogenesis is making glucose from non-carbohydrates precursors. Although Gluconeogenesis looks like Glycolysis in many steps, it is not the simple
14 Moh Tarek Razi Kittaneh Jaqen H ghar Naif Karadsheh Gluconeogenesis is making glucose from non-carbohydrates precursors. Although Gluconeogenesis looks like Glycolysis in many steps, it is not the simple
Physiology Unit 1 CELL SIGNALING: CHEMICAL MESSENGERS AND SIGNAL TRANSDUCTION PATHWAYS
 Physiology Unit 1 CELL SIGNALING: CHEMICAL MESSENGERS AND SIGNAL TRANSDUCTION PATHWAYS In Physiology Today Cell Communication Homeostatic mechanisms maintain a normal balance of the body s internal environment
Physiology Unit 1 CELL SIGNALING: CHEMICAL MESSENGERS AND SIGNAL TRANSDUCTION PATHWAYS In Physiology Today Cell Communication Homeostatic mechanisms maintain a normal balance of the body s internal environment
Biochemistry Sheet 27 Fatty Acid Synthesis Dr. Faisal Khatib
 Page1 بسم رلاهللا On Thursday, we discussed the synthesis of fatty acids and its regulation. We also went on to talk about the synthesis of Triacylglycerol (TAG). Last time, we started talking about the
Page1 بسم رلاهللا On Thursday, we discussed the synthesis of fatty acids and its regulation. We also went on to talk about the synthesis of Triacylglycerol (TAG). Last time, we started talking about the
Plasma lipoproteins & atherosclerosis by. Prof.Dr. Maha M. Sallam
 Biochemistry Department Plasma lipoproteins & atherosclerosis by Prof.Dr. Maha M. Sallam 1 1. Recognize structures,types and role of lipoproteins in blood (Chylomicrons, VLDL, LDL and HDL). 2. Explain
Biochemistry Department Plasma lipoproteins & atherosclerosis by Prof.Dr. Maha M. Sallam 1 1. Recognize structures,types and role of lipoproteins in blood (Chylomicrons, VLDL, LDL and HDL). 2. Explain
number Done by Corrected by Doctor
 number 20 Done by Corrected by Rana Ghassan Doctor Only 4 questions in the mid-term exam are based on the 4 lectures to be given by Dr Faisal. Dr Faisal will give us 10 lectures, the first 4 are included
number 20 Done by Corrected by Rana Ghassan Doctor Only 4 questions in the mid-term exam are based on the 4 lectures to be given by Dr Faisal. Dr Faisal will give us 10 lectures, the first 4 are included
Supporting Information Table of content
 Supporting Information Table of content Supporting Information Fig. S1 Supporting Information Fig. S2 Supporting Information Fig. S3 Supporting Information Fig. S4 Supporting Information Fig. S5 Supporting
Supporting Information Table of content Supporting Information Fig. S1 Supporting Information Fig. S2 Supporting Information Fig. S3 Supporting Information Fig. S4 Supporting Information Fig. S5 Supporting
Final Review Sessions. 3/16 (FRI) 126 Wellman (4-6 6 pm) 3/19 (MON) 1309 Surge 3 (4-6 6 pm) Office Hours
 Final Review Sessions 3/16 (FRI) 126 Wellman (4-6 6 pm) 3/19 (MON) 1309 Surge 3 (4-6 6 pm) Office ours 3/14 (WED) 9:30 11:30 am (Rebecca) 3/16 (FRI) 9-11 am (Abel) Final ESSENTIALS Posted Lecture 20 ormonal
Final Review Sessions 3/16 (FRI) 126 Wellman (4-6 6 pm) 3/19 (MON) 1309 Surge 3 (4-6 6 pm) Office ours 3/14 (WED) 9:30 11:30 am (Rebecca) 3/16 (FRI) 9-11 am (Abel) Final ESSENTIALS Posted Lecture 20 ormonal
Ahmad Ulnar. Faisal Nimri ... Dr.Faisal
 24 Ahmad Ulnar Faisal Nimri... Dr.Faisal Fatty Acid Synthesis - Occurs mainly in the Liver (to store excess carbohydrates as triacylglycerols(fat)) and in lactating mammary glands (for the production of
24 Ahmad Ulnar Faisal Nimri... Dr.Faisal Fatty Acid Synthesis - Occurs mainly in the Liver (to store excess carbohydrates as triacylglycerols(fat)) and in lactating mammary glands (for the production of
Implications of mitochondrial skeletal muscle metabolism on diabetes and obesity before and after weight loss
 GG2 Implications of mitochondrial skeletal muscle metabolism on diabetes and obesity before and after weight loss Dr Giacomo Gastaldi CHRU Montpellier Folie 1 GG2 19.10.2009 GG_PC; 12.10.2009 Plan Introduction
GG2 Implications of mitochondrial skeletal muscle metabolism on diabetes and obesity before and after weight loss Dr Giacomo Gastaldi CHRU Montpellier Folie 1 GG2 19.10.2009 GG_PC; 12.10.2009 Plan Introduction
Principles of Genetics and Molecular Biology
 Cell signaling Dr. Diala Abu-Hassan, DDS, PhD School of Medicine Dr.abuhassand@gmail.com Principles of Genetics and Molecular Biology www.cs.montana.edu Modes of cell signaling Direct interaction of a
Cell signaling Dr. Diala Abu-Hassan, DDS, PhD School of Medicine Dr.abuhassand@gmail.com Principles of Genetics and Molecular Biology www.cs.montana.edu Modes of cell signaling Direct interaction of a
BCH 4054 Spring 2001 Chapter 24 Lecture Notes
 BCH 4054 Spring 2001 Chapter 24 Lecture Notes 1 Chapter 24 Fatty Acid Catabolism 2 Fatty Acids as Energy Source Triglycerides yield 37 kj/g dry weight Protein 17 kj/g Glycogen 16 kj/g (even less wet weight)
BCH 4054 Spring 2001 Chapter 24 Lecture Notes 1 Chapter 24 Fatty Acid Catabolism 2 Fatty Acids as Energy Source Triglycerides yield 37 kj/g dry weight Protein 17 kj/g Glycogen 16 kj/g (even less wet weight)
Metabolic integration and Regulation
 Metabolic integration and Regulation 109700: Graduate Biochemistry Trimester 2/2016 Assistant Prof. Dr. Panida Khunkaewla kpanida@sut.ac.th School of Chemistry Suranaree University of Technology 1 Overview
Metabolic integration and Regulation 109700: Graduate Biochemistry Trimester 2/2016 Assistant Prof. Dr. Panida Khunkaewla kpanida@sut.ac.th School of Chemistry Suranaree University of Technology 1 Overview
Cholesterol metabolism. Function Biosynthesis Transport in the organism Hypercholesterolemia
 Cholesterol metabolism Function Biosynthesis Transport in the organism Hypercholesterolemia - component of all cell membranes - precursor of bile acids steroid hormones vitamin D Cholesterol Sources: dietary
Cholesterol metabolism Function Biosynthesis Transport in the organism Hypercholesterolemia - component of all cell membranes - precursor of bile acids steroid hormones vitamin D Cholesterol Sources: dietary
OVERVIEW OF ENERGY AND METABOLISM
 Biochemistry 5. Bio-Energetics & ATP 5.1) ADP, ATP and Cellular Respiration OVERVIEW OF ENERGY AND METABOLISM 1. The food we eat, (carbohydrates/ glucose /sugar, lipids/fat, proteins), are our only source
Biochemistry 5. Bio-Energetics & ATP 5.1) ADP, ATP and Cellular Respiration OVERVIEW OF ENERGY AND METABOLISM 1. The food we eat, (carbohydrates/ glucose /sugar, lipids/fat, proteins), are our only source
GENERAL CHARACTERISTICS OF THE ENDOCRINE SYSTEM FIGURE 17.1
 GENERAL CHARACTERISTICS OF THE ENDOCRINE SYSTEM FIGURE 17.1 1. The endocrine system consists of glands that secrete chemical signals, called hormones, into the blood. In addition, other organs and cells
GENERAL CHARACTERISTICS OF THE ENDOCRINE SYSTEM FIGURE 17.1 1. The endocrine system consists of glands that secrete chemical signals, called hormones, into the blood. In addition, other organs and cells
Signal Transduction Cascades
 Signal Transduction Cascades Contents of this page: Kinases & phosphatases Protein Kinase A (camp-dependent protein kinase) G-protein signal cascade Structure of G-proteins Small GTP-binding proteins,
Signal Transduction Cascades Contents of this page: Kinases & phosphatases Protein Kinase A (camp-dependent protein kinase) G-protein signal cascade Structure of G-proteins Small GTP-binding proteins,
Cell Signaling (part 1)
 15 Cell Signaling (part 1) Introduction Bacteria and unicellular eukaryotes respond to environmental signals and to signaling molecules secreted by other cells for mating and other communication. In multicellular
15 Cell Signaling (part 1) Introduction Bacteria and unicellular eukaryotes respond to environmental signals and to signaling molecules secreted by other cells for mating and other communication. In multicellular
The role of apolipoprotein D in lipid metabolism and metabolic syndrome
 UQÀM: Department of Biological Sciences The role of apolipoprotein D in lipid metabolism and metabolic syndrome Catherine Mounier PhD Apolipoprotein D Apolipoprotéine D (apod) Secreted protein Associated
UQÀM: Department of Biological Sciences The role of apolipoprotein D in lipid metabolism and metabolic syndrome Catherine Mounier PhD Apolipoprotein D Apolipoprotéine D (apod) Secreted protein Associated
Roles of Lipids in the body
 Roles of Lipids in the body 1- Energy storage : Lipids contain a lot of calories in a small space. Since Lipids are generally insoluble in polar substances such as water, they are stored in special ways
Roles of Lipids in the body 1- Energy storage : Lipids contain a lot of calories in a small space. Since Lipids are generally insoluble in polar substances such as water, they are stored in special ways
Chemistry 1120 Exam 4 Study Guide
 Chemistry 1120 Exam 4 Study Guide Chapter 12 12.1 Identify and differentiate between macronutrients (lipids, amino acids and saccharides) and micronutrients (vitamins and minerals). Master Tutor Section
Chemistry 1120 Exam 4 Study Guide Chapter 12 12.1 Identify and differentiate between macronutrients (lipids, amino acids and saccharides) and micronutrients (vitamins and minerals). Master Tutor Section
Tala Saleh. Razi Kittaneh ... Nayef Karadsheh
 Tala Saleh Razi Kittaneh... Nayef Karadsheh β-oxidation of Fatty Acids The oxidation of fatty acids occurs in 3 steps: Step 1: Activation of the Fatty acid FA + HS-CoA + ATP FA-CoA + AMP + PPi - The fatty
Tala Saleh Razi Kittaneh... Nayef Karadsheh β-oxidation of Fatty Acids The oxidation of fatty acids occurs in 3 steps: Step 1: Activation of the Fatty acid FA + HS-CoA + ATP FA-CoA + AMP + PPi - The fatty
Glucose. Glucose. Insulin Action. Introduction to Hormonal Regulation of Fuel Metabolism
 Glucose Introduction to Hormonal Regulation of Fuel Metabolism Fasting level 3.5-5 mmol (1 mmol = 18 mg/dl) Postprandial 6-10 mmol Amount of glucose in circulation is dependent on: Absorption from the
Glucose Introduction to Hormonal Regulation of Fuel Metabolism Fasting level 3.5-5 mmol (1 mmol = 18 mg/dl) Postprandial 6-10 mmol Amount of glucose in circulation is dependent on: Absorption from the
Niacin Metabolism: Effects on Cholesterol
 Niacin Metabolism: Effects on Cholesterol By Julianne R. Edwards For Dr. William R. Proulx, PhD, RD Associate Professor of Nutrition and Dietetics In partial fulfillments for the requirements of NUTR342
Niacin Metabolism: Effects on Cholesterol By Julianne R. Edwards For Dr. William R. Proulx, PhD, RD Associate Professor of Nutrition and Dietetics In partial fulfillments for the requirements of NUTR342
Digestion and transport of TAG by plasma lipoproteins
 Digestion and transport of TAG by plasma lipoproteins Lipoproteins are multimolecular complexes of lipids and proteins, they are not macromolecules They transport lipids in the plasma because lipids are
Digestion and transport of TAG by plasma lipoproteins Lipoproteins are multimolecular complexes of lipids and proteins, they are not macromolecules They transport lipids in the plasma because lipids are
Biochemistry 7/11/ Bio-Energetics & ATP. 5.1) ADP, ATP and Cellular Respiration OVERVIEW OF ENERGY AND METABOLISM
 Biochemistry 5. Bio-Energetics & ATP 5.1) ADP, ATP and Cellular Respiration Prof. Dr. Klaus Heese OVERVIEW OF ENERGY AND METABOLISM 1. The food we eat, (carbohydrates/ glucose /sugar, lipids/fat, proteins),
Biochemistry 5. Bio-Energetics & ATP 5.1) ADP, ATP and Cellular Respiration Prof. Dr. Klaus Heese OVERVIEW OF ENERGY AND METABOLISM 1. The food we eat, (carbohydrates/ glucose /sugar, lipids/fat, proteins),
Lipid metabolism. Degradation and biosynthesis of fatty acids Ketone bodies
 Lipid metabolism Degradation and biosynthesis of fatty acids Ketone bodies Fatty acids (FA) primary fuel molecules in the fat category main use is for long-term energy storage high level of energy storage:
Lipid metabolism Degradation and biosynthesis of fatty acids Ketone bodies Fatty acids (FA) primary fuel molecules in the fat category main use is for long-term energy storage high level of energy storage:
Multiple choice: Circle the best answer on this exam. There are 12 multiple choice questions, each question is worth 3 points.
 CHEM 4420 Exam 4 Spring 2015 Dr. Stone Page 1 of 6 Name Use complete sentences when requested. There are 120 possible points on this exam. Therefore there are 20 bonus points. Multiple choice: Circle the
CHEM 4420 Exam 4 Spring 2015 Dr. Stone Page 1 of 6 Name Use complete sentences when requested. There are 120 possible points on this exam. Therefore there are 20 bonus points. Multiple choice: Circle the
Physiology Unit 1 METABOLISM OF LIPIDS AND PROTEINS
 Physiology Unit 1 METABOLISM OF LIPIDS AND PROTEINS Alternate Fuel Sources When glucose levels are low Proteins and Triglycerides will be metabolized Tissues will use different fuel sources depending on:
Physiology Unit 1 METABOLISM OF LIPIDS AND PROTEINS Alternate Fuel Sources When glucose levels are low Proteins and Triglycerides will be metabolized Tissues will use different fuel sources depending on:
The citric acid cycle Sitruunahappokierto Citronsyracykeln
 The citric acid cycle Sitruunahappokierto Citronsyracykeln Ove Eriksson BLL/Biokemia ove.eriksson@helsinki.fi Metabolome: The complete set of small-molecule metabolites to be found in a cell or an organism.
The citric acid cycle Sitruunahappokierto Citronsyracykeln Ove Eriksson BLL/Biokemia ove.eriksson@helsinki.fi Metabolome: The complete set of small-molecule metabolites to be found in a cell or an organism.
Regulating Hepatic Cellular Cholesterol
 Under circumstances of cholesterol deficiency, Sterol Regulatory Element Binding Proteins (SREBPs) via binding to DNA nuclear response elements set off genomic production of proteins and enzymes that induce
Under circumstances of cholesterol deficiency, Sterol Regulatory Element Binding Proteins (SREBPs) via binding to DNA nuclear response elements set off genomic production of proteins and enzymes that induce
