Microsomal Membranes*
|
|
|
- Lambert Briggs
- 5 years ago
- Views:
Transcription
1 Proceedings of the National Academy of Sciences Vol. 66, No. 2, pp , June 1970 Membrane Structure: Lipid-Protein Interactions in Microsomal Membranes* Benjamin F. Trumpt Sue M. Duttera,t William L. Byrne, and Antti U. Arstila DEPARTMENTS OF PATHOLOGY AND BIOCHEMISTRY, DUKE UNIVERSITY, DURHAM, NORTH CAROLINA Communicated by Henry Lardy, February 26, 1970 Abstract. The relationships of phospholipid to membrane structure and function were examined in hepatic microsomes. Findings indicate that normal microsomal membrane structure is dependent on lipid-protein interactions and that it correlates closely with glucose-6-phosphatase activity. Modification of most phospholipid with phospholipase-c is associated with widening of the membrane which can be reversed following readdition of phospholipid. Introduction. Treatment of rat liver microsomes with phospholipase-c results in inactivation of glucose-6-phosphatase.1 This inactivation can be reversed by addition of a total microsomal lipid fraction, a microsomal phospholipid fraction, mixtures of phospholipid such as Asolectin, or individual phospholipids.' In the present study, we have investigated the relationship of morphologic change in the microsomal membrane to lipid content and to activity of glucose-6-phosphatase, using conditions previously described to show the lipid requirement of this enzyme.1 The results indicate a close correlation between normal membrane ultrastructure and glucose-6-phosphatase activity, demonstrate reversible morphologic changes after treatment of microsomal membranes with phospholipase-c, and show the importance of lipid-protein interactions in membrane structure. These data were recently presented in preliminary form.2 Materials and Methods. Microsomes were prepared from livers of 150 to 200-gm male Holtzman rats which were fasted with free access to water for 48 hr prior to sacrifice. At the time of killing, the livers were immediately removed and immersed in 0.25 Mll sucrose (00-40C) containing 0.01 M Tris (ph 8.0) and 1 mm EDTA. The livers were weighed and homogenized with a Teflon-glass homogenizer in the sucrose- Tris-EDTA solution. Microsomes were isolated at 3.2 X 106g X min1 and the final pellet was resuspended in this solution that, in addition, contained 1 mm dithiothreitol. Glucose-6-phosphatase activity was measured by phosphate release.1 Phospholipase-C treatment of microsomes was performed with a commercial preparation (Sigma) derived from C. welchii. The enzyme was dissolved in 0.01 M Tris buffer, ph , at a concentration of 1 to 3 mg protein/ml. Incubation of microsomes with phospholipase-c was performed in the presence of 2.5 mm calcium chloride and 125 mm Tris (ph 8.0) at 20'C for 30 min. Acetone treatment of microsomes was performed with 10% water in acetone.3 Microsomal lipids were extracted by the method of Folch et al.4 and phospholipid was separated from neutral lipid by elution on a silicic acid column.'. Micellar preparations of phospholipids were prepared by sonic oscillation using a model S-75 Branson Sonifier for 1 to 3 min in 20 mm Tris (ph 8.0) containing 1 mm EDTA. Reconstitution was studied by incubating treated microsomes with a two- to threefold 433
2 434 BIOCHEMISTRY: TRUMP ET AL. PROC. N. A. S. excess of phospholipid; either microsomal phospholipid or Asolectin (a mixture of soybean phosphatides). In some instances, reconstitution was performed in the presence of 1 mm dithiothreitol. For electron microscopy, control and treated microsomes were fixed in suspension by the addition of a sodium cacodylate-buffered (0.1 M) mixture of 4% glutaraldehyde and 4% formaldehyde5 to the microsomal suspension. After fixation in suspension for 30 min at 20'C, the microsomes were collected on Millipore filters (pore size 0.10 u.m) by using vacuum filtration. The Millipore filters were fixed for an additional 60 min and then were washed for 1 to 3 hr in 67 mm sodium cacodylate buffer (ph 7.4), containing 7.5% sucrose, postfixed in 1% s-collidine-buffered osmium tetroxide (ph 7.4), dehydrated in a series of ethanols, and flat embedded in Epon. The specimens were stained en bloc with uranyl acetate and the thin sections were double stained with uranyl acetate and lead citrate. Fragments of the embedded sheets were re-embedded in slots in Epon blocks and sectioned at approximately 300 A in a plane normal to the surface of the Millipore filter. Suspensions of Asolectin or microsomal phospholipid micelles were prepared for electron microscopy in an identical fashion. Glucose-6-phosphatase was demonstrated cytochemically as described in the legend of Figure 3c. Micrographs were taken using an Hitachi HU-11 microscope at a primary magnification of 30,000. Magnification was calibrated with a replica or a diffraction grating, and all lens currents were standardized. The treated microsomes from each experiment were photographed immediately after photography of corresponding control microsomes and without changing the lens parameters. Positive projection prints were made on lantern slide plates at constant magnification. Measurements of membrane thickness were made with a Joyce-Loebl microdensitometer. These were taken in regions showing the trilaminar structure. This trilaminar structure is observed when the section is normal to the membrane surface. Varying the angle of section would be expected to affect the breadth of each peak but not the peak-to-peak measurement. Results. As shown in Table 1, treatment with phospholipase-c resulted in loss of 83% of the original glucose-6-phosphatase activity. This treatment also resulted in the hydrolysis of approximately 70% of microsomal phospholipids that were extracted and analyzed as described.' Incubation of treated microsomes with excess Asolectin or microsomal phospholipids resulted in complete restoration of glucose-6-phosphatase activity; reactivation with phospholipid in the presence of dithiothreitol gave similar results. In a previous publication' it was shown that essentially all microsomal lecithin and sphingomyelin and approximately 60% of phosphatidylethanolamine were cleaved by phospholipase-c treatment. It was also shown that the products of phospholipase-c action themselves have no effect on the observed inactivation or on the reactivation after addition of phospholipid. Acetone-treated microsomes lost 98% of glucose- 6-phosphatase activity with a concomitant loss of 90% of the microsomal phospholipid. Postincubation with Asolectin failed to restore control levels (Table 1). Morphologic studies of untreated microsome fractions revealed a population of vesicular profiles (Fig. ld and e). The vesicles contained a rather sparse flocculent material and occasionally smaller internal vesicles. Contaminating mitochondria, lysosomes, and microbodies were absent. The membrane which limited the vesicles showed a trilaminar appearance. Occasionally, suggestions of striations perpendicular to the laminae were observed. lmlicrodensitometer tracings across the membrane in areas cut normal to the membrane surface yielded two distinct peaks corresponding to the dense laminae separated by a trough (Fig. la). A frequency-distribution plot of peak-to-peak thickness of
3 VOL. 66, 1970 BIOCHEMISTRY: TRUMP ET AL. 435 TABLE 1. Effect of lipid depletion and reconstitution on the glucose-6-phosphatase activity of hepatic microsomes. Glucose-6-phosphatase activity Condition (per cent untreated) (A) Untreated microsomes (100)* (B) Phospholipase C-treated microsomes 17 (C) (B) + Asolectin 119 (D) (B) + microsomal phospholipid 147 (E) (B) + dithiothreitol 19 (F) Acetone-treated microsomes 2 (G) (F) + Asolectin 7 The samples were kept in ice prior to the activity determination for the same interval of time as that which elapsed between preparation of the various samples and fixation. * jmole Pi released/10 min/ml incubation mixture. untreated microsomal membranes is shown in Figure 2. The mean thickness was 50 A, however, there was a small secondary population with a mean thickness of 75 A. Electron micrographs of microsomes incubated for demonstration of glucose-6-phosphatase activity revealed accumulations of lead phosphate reaction product along the dense inner and outer laminae of the membrane (Fig. 3c); the central clear area was free of reaction product. Phospholipase-C-treated microsomes showed general preservation of the vesicular structure (Fig. if), although ribosomes appeared fewer in number. The trilaminar structure of the membranes was well preserved (Fig. if) and in fact appeared more distinct than in control preparations. In addition, small homogeneous densities were often seen eccentrically applied to the surface of the microsomes (Figs. if and 3a). These densities appeared to be continuous with the space between the dense outer and inner laminae, producing a signet ring appearance (Figs. if and 3a). These droplets are assumed to represent the released diglyceride possibly mixed with other components. Additional flocculent material was located between the microsomes. Measurements of the peakto-peak thickness of phospholipase-c-treated microsomes (Fig. lb) revealed a single population with a mean value approximately 1.5 times that of the control preparation (Fig. 2); this population differed significantly (p < 0.01) from the control population. In order to evaluate the possibility that the increase in center-to-center spacing was due to an asymmetric decrease in thickness of the dense layers, individual tracings of the dense layers were measured. It was found that these layers increased rather than decreased in thickness thus precluding the possibility that the increase in center-to-center spacing was related only to a decrease in the dense layers. Examination of microsomes reconstituted with microsomal phospholipid or with Asolectin again revealed a vesicular appearance with preservation of the trilaminar structure (Fig. 1g). Vesicles and the spaces between them appeared relatively empty as compared with the other preparations; the dense eccentric droplets seen in phospholipase-c-treated microsomes were absent. In addition to the microsomes there were complex lamellar membranous profiles (Fig. 3b) identical with those in preparations of purified micelles (Fig. 3d) and, accordingly, these were assumed to represent excess phospholipid which had not been
4 436 BIOCHEMISTRY: TRUMP ET AL. PROC. N. A. S. a 50A b d FIG. 1.-Electron micrographs and microdensitometer tracings of control and treated microsomal membranes. (a)-(c) represent microdensitometer traces across typical membranes in control, phospholipase-treated, and reconstituted microsomes, respectively. The peaks in these tracings represent the dense areas seen within the membrane in electron micrographs as shown in (d)-(g). (a)-(c) are shown at the same magnification; the bar shows 50A at this magnification. (d) and (e) are micrographs of control microsomes. Note the typical trilaminar appearance ol the membrane as indicated by the free arrows in (d) and note the suggestion of periodicity in the membrane structure shown by the apposing arrows in (e). (f) is a micrograph of microsomes treated with phospholipase-c. These membranes are thicker as shown in Fig. (b) and often show the accumulation of homogeneous areas of intermediate density between the inner and outer dense layers (Fig. if, arrow). These are interpreted as accumulations of diglycerides. (g) shows microsomes reconstituted by addition of Asolectin. The double arrow indicates a lamellar profile which probably represents a section of a lipid micelle. All electron micrographs were taken at identical magnification without changing lens parameters. Magnification in (d), (f), and (g) is X195,000. Magnification in (e) is X210,000.
5 VOL. 66, 1970 BIOCHEMISTRY: TRUMP ET AL. 437 Untreated 40 -Phr*sphoapse X A FIG. 2.-Frequency-distribution plot of mem- g 30 brane thicknesses in untreated microsomes, phospholipase-c-treated microsomes, and microsomes reconstituted with Asolectin in the presence or absence = 20 I of dithiothreitol. The statistical significance of these distributions was analyzed by the method of Kol- l0 / mogorov and Smirnov.20 Phospholipase-C-treated \ microsomes were significantly different from untreated microsomes (p < 0.01), whereas microsomes reconstituted in the presence of dithiothreitol did not significantly differ from control values. Micro- L somes reconstituted with Asolectin alone showed evi- 40 dence of secondary larger populations, suggesting that all membranes did not return to normal. These + :1 Recofstvte measurements are based on the pooled results of four 3 R \ OTT different experiments with the following total measurements in each group: untreated, 42; phospho- 20 lipase-c-treated, 23; reconstituted, 39; and reconstituted with dithiothreitol, / I A integrated into the microsomes. Measurements of reconstituted microsomes (Fig. ic) showed a large population similar to untreated preparations (Fig. 2); there were, however, additional populations which represented thicker membranes suggesting a bi- or triphasic population and indicating that some membranes did not return to control thickness. If one looks at the fraction of the population that has a dimension comparable to the major peak in the untreated sample, it appears that reconstitution in the presence of dithiothreitol leads to a more effective reconstitution, although the distribution of values did not differ statistically from values on membranes reconstituted without dithiothreitol. Examination of acetone-treated microsomes revealed complete loss of typical microsomal vesicles. Numerous fine filamentous profiles represented the only remnants of microsomal membranes (Fig. 3e). These dense irregular profiles are assumed to represent mostly residual protein and were often observed to enclose clear spaces which formed a regular pattern along the residual membrane material. The widening of the membrane seen after modification of most phospholipids suggests that changes in protein conformation occur in this condition. We suggest that the membrane widens because hydrophobic bonding with lipids is important for normal protein conformation in microsomal membranes and, when most of the phospholipids are modified, the proteins change in a way which influences the protein-protein interactions. This is in agreement with the concept that membrane proteins have a genetically determined sequence of predominantly hydrophobic amino acids which are dependent on a hydrophobic environment for selection of a final three-dimensional structure.8 These results
6 438 BIOCHEMISTRY: TRUMP ET AL. PROC. N. A. S. + M9 %4~~~~~~~~~~~~~~~~~~~~~~~~~~~. ~ ~ ~ ~ ~ Ik a II 'Jsh3k a,..'-,' i(' t. c 'd t C) FIG. 3.-(a) Electron micrograph showing microsomes treated with phospholipase-c. Note the numerous circular areas of intermediate density which represent accumulations of diglyceride released by the phospholipase treatment. In several areas (free arrows) these densities are seen eccentrically located along the margin of a microsome producing a "signet ring" appearance. In high resolution pictures (Fig. if), these accumulations are seen to be located between the outer and inner dense layers of the microsomal membrane. X48,000. (b) Electron micrograph showing microsomes after reconstitution with Asolectin. Note the micelles of added Asolectin (free arrows) which consist of clusters of concentrically-arranged dense lamellae. X44,000. (e) Control microsomal preparations showing histochemical localization of glucose-6phosphatase activity. Note the accumulation of the dense lead phosphate reaction product along the inner and outer surfaces of the membrane. Glucose-6phosphatase was demonstrated as follows. Microsomes were fixed in 4% sodium cacodylate-buffered paraformaldehyde for 8 mm;- the microsomes were collected on Millipore filters, washed 30 mmn in buffer, and were incubated for 10 mmn in the medium described by Wachstein and Meisel.21 The microsomes were then postfixed in osmium tetroxide as described in the text. X(31,500. (d) Electron micrograph of Asolectin preparations showing the typical micelles. X38,500. (e) High magnification micrograph of microsomal membrane after treatment with acetonewater. Observe that the typical trilaminar structure is not visible; membrane structure has broken down. The membrane is represented by irregular dense profiles (arrows) which are believed to represent residual protein. X 137,500. are in contrast to the study of Lenard and Singer on erythrocyte membranes after treatment with phospholipase C.9 These authors observed no changes in protein conformation as measured by circular dichroism after removal of phospholipids. It should be pointed out, however, that erythrocyte membranes have a higher protein-to-lipid ratio than endoplasmic reticulum and that, in the case of erythrocyte membranes, hydrophobic interactions with lipid may be less important for maintenance of final membrane structure.7
7 VOL. 66, 1970 BIOCHEMISTRY: TRUMP ET AL. 439 It was striking to observe that the trilaminar structure of the microsomal membrane was completely maintained with most phospholipid absent. This is compatible with the assertion that osmium concentration in membranes occurs at hydrophilic interfaces.7 The dense lines in electron micrographs therefore probably reflect interaction with the hydrophilic end of proteins as much as interaction with hydrophilic ends of phospholipids on the unsaturated fatty acids. The results also emphasize the importance of protein for the structural integrity of the microsoinal membrane although at the same time they demonstrate the importance of lipid-protein interactions; if lipids are removed with 10%0 water in acetone, the membrane structure breaks down. This loss of structure after acetone-water treatment is in contrast to results with the inner membrane of mitochondria'0 and, once again, the protein-lipid ratios are significantly higher in mitochondrial inner membranes than they are in endoplasmic reticulum.7 Another striking finding in our experiments was the accumulation of diglyceride within the membrane after treatment with phospholipase-c. Finean and Martonosi noted the appearance of diglyceride droplets in electron micrographs of phospholipase-c-treated muscle microsomes" but did not comment on their ultrastructural location. This accumulation of intramembranous, relatively apolar, lipid suggests, in view of the considerations mentioned above, the presence of two layers of protein. It is presumed that after release of diglyceride, accumulation occurs within the hydrophobic interior of the membrane. This observation gives experimental support to the theoretical model of apolar lipid accumulation within a bilayer recently proposed by Danielli.'2 When excess phospholipid is added, the accumulated diglyceride evidently escapes from the membrane since it does not appear after reconstitution. These studies, along with others,'3 14 emphasize the need to reexamine the bimolecular leaflet hypothesis of membrane structure and emphasize that different structural factors may be important in different types of cellular membranes. In the case of microsomal membranes, our studies emphasize the importance of both lipid and protein to maintain normal fine structure. To be consistent with our data, a macromolecular model for the microsomal membrane must explain the following: (1) phospholipid molecules are readily accessible to or reach the membrane surfaces; (2) the dense lines observed in membranes after postfixation in osmium are present after modification of most phospholipid; (3) the membrane must be capable of reversible changes in dimension related to phospholipid-protein interactions; (4) the apparent accumulation of diglyceride in the interior of the membrane; (5) the presence of dense lines presumably representing protein on each side of the diglyceride; and (6) the trilaminar structure is present when most phospholipid is modified, although in this state the membrane conformation differs from normal. Suich a model might involve a mosaic of repeating protein and lipid subunits,"5 but it is not clear from these results whether the protein mosaic is necessarily continuous. A certain proportion of bilayer might well occur. The important role that changes, in even a single membrane "protomer," can have in determining the over-all architecture of the membrane is emphasized in the hypothesis of Changeux et al.1" Their hy-
8 440 BIOCHEMISTRY: TRUMP ET AL. PROC. N. A. S. pothesis also assists in explaining the observations that various morphologic classes of membranes exist."7-19 These results may also have meaning for studies of membrane synthesis and turnover. They indicate that the potential for normal structure and function can be maintained after phospholipid modifications and expressed when phospholipid is added. The facilitation of reconstitution of structure by dithiothreitol would also suggest a potential role for disulfide interchange, but the in vivo role of these potentialities is unknown. Finally, these results emphasize the possibility that attempts to isolate individual components of membrane systems will be handicapped by the interdependence of membrane components for their normal structure and function. A given component may exhibit its normal structure and specificity irrespective of its environment, but the importance of phospholipid for membrane dimension is a reminder that the typical component in a membrane system, such as the endoplasmic reticulum, would appear to be dependent on direct or indirect interaction with the other components of the membrane. The authors wish to express their appreciation to Dr. Walter Guild for assistance in the microdensitometry, to Mrs. Jessie Calder for assistance in the electron microscopic preparations, and to Bernard Bell for the photographic work. * Supported in part by grants from NIH (AM to B. F. T., F05-TW to A. U. A., GM to W. L. B.), and a grant from the Finnish National Research Council for Medical Sciences to A. U. A. t Requests for reprints may be addressed to Dr. Trump, Department of Pathology, Duke University, Durham, North Carolina. $ Present address: Department of Physiology, Emory University, Atlanta, Georgia. Present address: Department of Biochemistry, University of Tennessee Medical Units, Memphis, Tennessee. 1 Duttera, S. M., W. L. Byrne, and M. C. Ganoza, J. Biol. Chem., 243, 2216 (1968). 2 Trump, B. F., A. U. Arstila, S. M. Duttera, and W. L. Byrne, abstr. in Federation Proc., 28, 403 (1969). 3 Jones, P. D., and S. J. Wakil, J. Biol. Chem., 242, 5267 (1967). 4Folch, J., M. Lees, and G. H. S. Stanley, J. Biol. Chem., 226, 497 (1957). 5 Karnovsky, M. J., J. Cell Biol., 27, 137A (1965). 6 Duttera, S. M., Ph.D. dissertation, Duke University, 1968 (University Microfilms, Inc., Ann Arbor, Michigan). 7Korn, E. D., Federation Proc., 28, 6 (1969). 8 Wallach, D. F., and P. H. Zahler, these PROCEEDINGS, 56, 1552 (1966). 9 Lenard, J., and S. J. Singer, Science, 159, 738 (1968). 10 Fleischer, S., B. F. Fleischer, and W. Stoeckenius, J. Cell Biol., 32, 193 (1967). 11 Finean, J. B., and A. Martonosi, Biochim. Biophys. Acta, 98, 547 (1965). 12 Danielli, J. F., in Formation and Fate of Cell Organelles, ed. K. B. Warren (New York: Academic Press, 1967), p Korn, E. D., Science, 153, 1491 (1966). 14 Benson, A. A., in Membrane Models and the Formation of Biological Membranes, ed. C. Boles and B. A. Pethica (Amsterdam: Wiley Interscience, 1968), p Wallach, D. F. H., and A. Gordon, Federation Proc., 27, 1263 (1968). 16 Changeaux, J. P., J. Thiery, Y. Tung, and C. Kitel, these PROCEEDINGS, 57, 335 (1967). 17 Yamamoto, T., J. Cell Biol., 17, 413 (1963). 18 Sj6strand, F. S., J. Ultrastruct. Res., 9, 561 (1963). 19 Arstila, A. U., and B. F. Trump, Am. J. Path., 53, 687 (1968). 20 Tate, M. W., and R. C. Clelland, Nonparametric and Shortcut Statistics (Danville, Illinois: Interstate Printer and Publisher Inc., 1957), p Wachstein, M., and E. Meisel, J. Histochem. Cytochem., 4, 592 (1956).
FIXATION BY MEANS OF GLUTARALDEHYDE-HYDROGEN PEROXIDE REACTION PRODUCTS
 FIXATION BY MEANS OF GLUTARALDEHYDE-HYDROGEN PEROXIDE REACTION PRODUCTS CAMILLO PERACCHIA and BRANT S. MITTLER. From the Department of Anatomy, Duke University Medical Center, Durham, North Carolina 27706,
FIXATION BY MEANS OF GLUTARALDEHYDE-HYDROGEN PEROXIDE REACTION PRODUCTS CAMILLO PERACCHIA and BRANT S. MITTLER. From the Department of Anatomy, Duke University Medical Center, Durham, North Carolina 27706,
Chapter 1 Membrane Structure and Function
 Chapter 1 Membrane Structure and Function Architecture of Membranes Subcellular fractionation techniques can partially separate and purify several important biological membranes, including the plasma and
Chapter 1 Membrane Structure and Function Architecture of Membranes Subcellular fractionation techniques can partially separate and purify several important biological membranes, including the plasma and
Effect of phospholipase-d on rat kidney mitochondria*
 J. Biosci., Vol. 1, Number 1, March 1979, pp. 75 82. Printed in India. Effect of phospholipase-d on rat kidney mitochondria* S. N. A. ZAIDI, A. C. SHIPSTONE and N. K. GARG Division of Biochemistry, Central
J. Biosci., Vol. 1, Number 1, March 1979, pp. 75 82. Printed in India. Effect of phospholipase-d on rat kidney mitochondria* S. N. A. ZAIDI, A. C. SHIPSTONE and N. K. GARG Division of Biochemistry, Central
LOW-ANGLE X-RAY DIFFRACTION AND ELECTRON-MICROSCOPE STUDIES OF ISOLATED CELL MEMBRANES
 J. Cell Sci. I, 287-296 (1966) 287 Printed in Great Britain LOW-ANGLE X-RAY DIFFRACTION AND ELECTRON-MICROSCOPE STUDIES OF ISOLATED CELL MEMBRANES J. B. FINEAN, R. COLEMAN, W. G. GREEN* AND A. R. LIMBRICK
J. Cell Sci. I, 287-296 (1966) 287 Printed in Great Britain LOW-ANGLE X-RAY DIFFRACTION AND ELECTRON-MICROSCOPE STUDIES OF ISOLATED CELL MEMBRANES J. B. FINEAN, R. COLEMAN, W. G. GREEN* AND A. R. LIMBRICK
Explain the reason for this difference in resolving power.
 1. (a) An electron microscope has a much greater resolving power than an optical microscope. (i) Explain the meaning of the term resolving power. Explain the reason for this difference in resolving power.
1. (a) An electron microscope has a much greater resolving power than an optical microscope. (i) Explain the meaning of the term resolving power. Explain the reason for this difference in resolving power.
Initially, the patients did not receive extra vitamin E except for a very
 EFFECT OF VITAMIN E ON MEMBRANES OF THE INTESTINAL CELL BY I. MOLENAAR, F. A. HOMMES, W. G. BRAAMS, AND H. A. POLMAN CENTER FOR MEDICAL ELECTRON MICROSCOPY AND DEPARTMENT OF PEDIATRICS, UNIVERSITY OF GRONINGEN,
EFFECT OF VITAMIN E ON MEMBRANES OF THE INTESTINAL CELL BY I. MOLENAAR, F. A. HOMMES, W. G. BRAAMS, AND H. A. POLMAN CENTER FOR MEDICAL ELECTRON MICROSCOPY AND DEPARTMENT OF PEDIATRICS, UNIVERSITY OF GRONINGEN,
Intercellular Matrix in Colonies of Candida
 JouRNAL OF BAcTEROLOGY, Sept. 1975, p. 1139-1143 Vol. 123, No. 3 Copyright 0 1975 American Society for Microbiology Printed in U.S.A. ntercellular Matrix in Colonies of Candida K. R. JOSH, J. B. GAVN,*
JouRNAL OF BAcTEROLOGY, Sept. 1975, p. 1139-1143 Vol. 123, No. 3 Copyright 0 1975 American Society for Microbiology Printed in U.S.A. ntercellular Matrix in Colonies of Candida K. R. JOSH, J. B. GAVN,*
ENHANCEMENT OF THE GRANULATION OF ADRFNERGIC STORAGE VESICLES IN DRUG-FREE SOLUTION
 ENHANCEMENT OF THE GRANULATION OF ADRFNERGIC STORAGE VESICLES IN DRUG-FREE SOLUTION TAKASHI IWAYAMA and J. B. FURNESS. From the Department of Zoology, University of Melbourne, Victoria, Australia. Dr.
ENHANCEMENT OF THE GRANULATION OF ADRFNERGIC STORAGE VESICLES IN DRUG-FREE SOLUTION TAKASHI IWAYAMA and J. B. FURNESS. From the Department of Zoology, University of Melbourne, Victoria, Australia. Dr.
Ultrastructure of Mycoplasmatales Virus laidlawii x
 J. gen. Virol. (1972), I6, 215-22I Printed in Great Britain 2I 5 Ultrastructure of Mycoplasmatales Virus laidlawii x By JUDY BRUCE, R. N. GOURLAY, AND D. J. GARWES R. HULL* Agricultural Research Council,
J. gen. Virol. (1972), I6, 215-22I Printed in Great Britain 2I 5 Ultrastructure of Mycoplasmatales Virus laidlawii x By JUDY BRUCE, R. N. GOURLAY, AND D. J. GARWES R. HULL* Agricultural Research Council,
Cell Overview. Hanan Jafar BDS.MSc.PhD
 Cell Overview Hanan Jafar BDS.MSc.PhD THE CELL is made of: 1- Nucleus 2- Cell Membrane 3- Cytoplasm THE CELL Formed of: 1. Nuclear envelope 2. Chromatin 3. Nucleolus 4. Nucleoplasm (nuclear matrix) NUCLEUS
Cell Overview Hanan Jafar BDS.MSc.PhD THE CELL is made of: 1- Nucleus 2- Cell Membrane 3- Cytoplasm THE CELL Formed of: 1. Nuclear envelope 2. Chromatin 3. Nucleolus 4. Nucleoplasm (nuclear matrix) NUCLEUS
Silver-Impregnation of the Golgi Complex in Epididymal Epithelial Cells of Mice
 CELL STRUCTURE AND FUNCTION 8, 339-346 (1984) C by Japan Society for Cell Biology Silver-Impregnation of the Golgi Complex in Epididymal Epithelial Cells of Mice Ikuo Yamaoka, Sumie Katsuta and Yoshimi
CELL STRUCTURE AND FUNCTION 8, 339-346 (1984) C by Japan Society for Cell Biology Silver-Impregnation of the Golgi Complex in Epididymal Epithelial Cells of Mice Ikuo Yamaoka, Sumie Katsuta and Yoshimi
POLLEN-WALL PROTEINS: ELECTRON- MICROSCOPIC LOCALIZATION OF ACID PHOSPHATASE IN THE INTINE OF CROCUS VERNUS
 J. Cell Sci. 8, 727-733 (197O 727 Printed in Great Britain POLLEN-WALL PROTEINS: ELECTRON- MICROSCOPIC LOCALIZATION OF ACID PHOSPHATASE IN THE INTINE OF CROCUS VERNUS R.B. KNOX* AND J. HESLOP-HARRISONf
J. Cell Sci. 8, 727-733 (197O 727 Printed in Great Britain POLLEN-WALL PROTEINS: ELECTRON- MICROSCOPIC LOCALIZATION OF ACID PHOSPHATASE IN THE INTINE OF CROCUS VERNUS R.B. KNOX* AND J. HESLOP-HARRISONf
1. (a) (i) Ability to distinguish points (close together); 1 (ii) Electrons have a shorter wavelength; 1
 1. (a) (i) Ability to distinguish points (close together); 1 Electrons have a shorter wavelength; 1 (b) (i) Golgi / nucleus / mitochondrion / endoplasmic reticulum / chromosome / larger ribosomes; R Membrane
1. (a) (i) Ability to distinguish points (close together); 1 Electrons have a shorter wavelength; 1 (b) (i) Golgi / nucleus / mitochondrion / endoplasmic reticulum / chromosome / larger ribosomes; R Membrane
THE PREPARATION AND ULTRASTRUCTURE OF AVIAN ERYTHROCYTE NUCLEAR ENVELOPE ENCLOSED BY THE PLASMA MEMBRANE
 J. Cell Sci. 34, 81-90 (1978) 8l Printed in Great Britain Company of Biologists Limited igj8 THE PREPARATION AND ULTRASTRUCTURE OF AVIAN ERYTHROCYTE NUCLEAR ENVELOPE ENCLOSED BY THE PLASMA MEMBRANE JAMES
J. Cell Sci. 34, 81-90 (1978) 8l Printed in Great Britain Company of Biologists Limited igj8 THE PREPARATION AND ULTRASTRUCTURE OF AVIAN ERYTHROCYTE NUCLEAR ENVELOPE ENCLOSED BY THE PLASMA MEMBRANE JAMES
PMT. Contains ribosomes attached to the endoplasmic reticulum. Genetic material consists of linear chromosomes. Diameter of the cell is 1 µm
 1. (a) Complete each box in the table, which compares a prokaryotic and a eukaryotic cell, with a tick if the statement is correct or a cross if it is incorrect. Prokaryotic cell Eukaryotic cell Contains
1. (a) Complete each box in the table, which compares a prokaryotic and a eukaryotic cell, with a tick if the statement is correct or a cross if it is incorrect. Prokaryotic cell Eukaryotic cell Contains
the structure of their ducts has been
 Tza JOURNAL 0? INVEa'riGATrVN DEBMATOLOOT Copyright t 1966 by The Williams & Wilkins Co. Vol. 46, No. I Printed in U.S.A. AN ELECTRON MICROSCOPIC STUDY OF THE ADULT HUMAN APOCRINE DUCT* KEN HASHIMOTO,
Tza JOURNAL 0? INVEa'riGATrVN DEBMATOLOOT Copyright t 1966 by The Williams & Wilkins Co. Vol. 46, No. I Printed in U.S.A. AN ELECTRON MICROSCOPIC STUDY OF THE ADULT HUMAN APOCRINE DUCT* KEN HASHIMOTO,
New aspect of hepatic nuclear glycogenosis
 J. clin. Path. (1968), 21, 19 New aspect of hepatic nuclear glycogenosis in diabetes1 F. CARAMIA, F. G. GHERGO, C. BRANCIARI, AND G. MENGHINI From the Institute of General Pathology, University of Rome,
J. clin. Path. (1968), 21, 19 New aspect of hepatic nuclear glycogenosis in diabetes1 F. CARAMIA, F. G. GHERGO, C. BRANCIARI, AND G. MENGHINI From the Institute of General Pathology, University of Rome,
Cell Structure. Present in animal cell. Present in plant cell. Organelle. Function. strength, resist pressure created when water enters
 Cell Structure Though eukaryotic cells contain many organelles, it is important to know which are in plant cells, which are in animal cells and what their functions are. Organelle Present in plant cell
Cell Structure Though eukaryotic cells contain many organelles, it is important to know which are in plant cells, which are in animal cells and what their functions are. Organelle Present in plant cell
A COMPARISON OF MEMBRANE FRACTURE FACES OF FIXED AND UNFIXED GLYCERINATED TISSUE
 J. Cell Set. 21, 437-448 (1976) 43-7 Printed in Great Britain A COMPARISON OF MEMBRANE FRACTURE FACES OF FIXED AND UNFIXED GLYCERINATED TISSUE A. S. BREATHNACH, M. GROSS, B. MARTIN AND C. STOLINSKI Department
J. Cell Set. 21, 437-448 (1976) 43-7 Printed in Great Britain A COMPARISON OF MEMBRANE FRACTURE FACES OF FIXED AND UNFIXED GLYCERINATED TISSUE A. S. BREATHNACH, M. GROSS, B. MARTIN AND C. STOLINSKI Department
psittaci by Silver-Methenamine Staining and
 JOURNAL OF BACTERIOLOGY, July 1972, p. 267-271 Copyright 1972 American Society for Microbiology Vol. 111, No. 1 Printed in U.S.A. Location of Polysaccharide on Chlamydia psittaci by Silver-Methenamine
JOURNAL OF BACTERIOLOGY, July 1972, p. 267-271 Copyright 1972 American Society for Microbiology Vol. 111, No. 1 Printed in U.S.A. Location of Polysaccharide on Chlamydia psittaci by Silver-Methenamine
Ultrastructure of Connective Tissue Cells of Giant African Snails Achatina fulica (Bowdich)
 Kasetsart J. (Nat. Sci.) 36 : 285-290 (2002) Ultrastructure of Connective Tissue Cells of Giant African Snails Achatina fulica (Bowdich) Viyada Seehabutr ABSTRACT The connective tissue sheath of cerebral
Kasetsart J. (Nat. Sci.) 36 : 285-290 (2002) Ultrastructure of Connective Tissue Cells of Giant African Snails Achatina fulica (Bowdich) Viyada Seehabutr ABSTRACT The connective tissue sheath of cerebral
Electron Microscopy of Small Cells: Mycoplasma hominis
 JOURNAL of BAcTRiowOY, Dc. 1969, p. 1402-1408 Copyright 0 1969 American Society for Microbiology Vol. 100, No. 3 Printed In U.S.A. NOTES Electron Microscopy of Small Cells: Mycoplasma hominis JACK MANILOFF
JOURNAL of BAcTRiowOY, Dc. 1969, p. 1402-1408 Copyright 0 1969 American Society for Microbiology Vol. 100, No. 3 Printed In U.S.A. NOTES Electron Microscopy of Small Cells: Mycoplasma hominis JACK MANILOFF
AN ELECTRON-MICROSCOPIC STUDY OF THE STARCH-CONTAINING PLASTIDS IN THE FERN TODEA BARBARA
 J. Cell Sci. 4, 211-221 (1969) 211 Printed in Great Britain AN ELECTRON-MICROSCOPIC STUDY OF THE STARCH-CONTAINING PLASTIDS IN THE FERN TODEA BARBARA H. M. SMITH* AND D. S. SMITHf Department of Biology,
J. Cell Sci. 4, 211-221 (1969) 211 Printed in Great Britain AN ELECTRON-MICROSCOPIC STUDY OF THE STARCH-CONTAINING PLASTIDS IN THE FERN TODEA BARBARA H. M. SMITH* AND D. S. SMITHf Department of Biology,
FIXATION OF TISSUES MODULE 5.1 INTRODUCTION OBJECTIVES 5.2 AIMS OF FIXATION 5.3 PRINCIPLE OF FIXATION. Notes
 MODULE Fixation of Tissues 5 FIXATION OF TISSUES 5.1 INTRODUCTION It is a process by which the cells or tissues are fixed in chemical and partly physical state so that they can withstand subsequent treatment
MODULE Fixation of Tissues 5 FIXATION OF TISSUES 5.1 INTRODUCTION It is a process by which the cells or tissues are fixed in chemical and partly physical state so that they can withstand subsequent treatment
FIRST MIDTERM EXAMINATION
 FIRST MIDTERM EXAMINATION 1. True or false: because enzymes are produced by living organisms and because they allow chemical reactions to occur that would not otherwise occur, enzymes represent an exception
FIRST MIDTERM EXAMINATION 1. True or false: because enzymes are produced by living organisms and because they allow chemical reactions to occur that would not otherwise occur, enzymes represent an exception
QUESTIONSHEET 1. The diagram shows some of the cell structures involved in the secretion of an extracellular enzyme. C D
 QUESTIONSHEET 1 The diagram shows some of the cell structures involved in the secretion of an extracellular enzyme. C D A (a) Identify A,, C, and D. A:... :... C:... D:... [4] (b) Outline the role of each
QUESTIONSHEET 1 The diagram shows some of the cell structures involved in the secretion of an extracellular enzyme. C D A (a) Identify A,, C, and D. A:... :... C:... D:... [4] (b) Outline the role of each
Basophilic. Basophilic structures are stained by basic dyes: Mnemonic: Basophilic = Blue
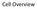 Cell Overview Basophilic Basophilic structures are stained by basic dyes: Basic dyes are positive Basophilic structures are negative (ex. DNA, RNA, ribosomes, RER) Mnemonic: Basophilic = Blue Acidophilic
Cell Overview Basophilic Basophilic structures are stained by basic dyes: Basic dyes are positive Basophilic structures are negative (ex. DNA, RNA, ribosomes, RER) Mnemonic: Basophilic = Blue Acidophilic
Practice Exam 2 MCBII
 1. Which feature is true for signal sequences and for stop transfer transmembrane domains (4 pts)? A. They are both 20 hydrophobic amino acids long. B. They are both found at the N-terminus of the protein.
1. Which feature is true for signal sequences and for stop transfer transmembrane domains (4 pts)? A. They are both 20 hydrophobic amino acids long. B. They are both found at the N-terminus of the protein.
This week s topic will be: Evidence for the Fluid Mosaic Model. Developing theories, testing hypotheses and techniques for visualizing cells
 Tutorials, while not mandatory, will allow you to improve your final grade in this course. Thank you for your attendance to date. These notes are not a substitute for the discussions that we will have
Tutorials, while not mandatory, will allow you to improve your final grade in this course. Thank you for your attendance to date. These notes are not a substitute for the discussions that we will have
Cell Structure and Function
 Household pin w/ bactera Cell Structure and Function Chapter 4 Same bacteria on pinhead Fig. 4-1c, p.50 Review: Ionic Bonds Na has 11p and 10e making it (+) Cl has 18e and 17 p making it (-) The attraction
Household pin w/ bactera Cell Structure and Function Chapter 4 Same bacteria on pinhead Fig. 4-1c, p.50 Review: Ionic Bonds Na has 11p and 10e making it (+) Cl has 18e and 17 p making it (-) The attraction
Some Observations on the Fine Structure of the Goblet Cells. Special Reference to the Well-Developed Agranular Endoplasmic Reticulum
 Okajimas Folia Anat. Jpn., 58(4-6) : 583-594, March 1982 Some Observations on the Fine Structure of the Goblet Cells in the Nasal Respiratory Epithelium of the Rat, with Special Reference to the Well-Developed
Okajimas Folia Anat. Jpn., 58(4-6) : 583-594, March 1982 Some Observations on the Fine Structure of the Goblet Cells in the Nasal Respiratory Epithelium of the Rat, with Special Reference to the Well-Developed
LIPID-CYTOCHROME c MEMBRANE
 THE STRUCTURE OF A LIPID-CYTOCHROME c MEMBRANE ALLEN B. BLAUROCK From the Cardiovascular Research Institute, San Francisco, California 94122 University of California, ABsrRAcr In the preceding paper a
THE STRUCTURE OF A LIPID-CYTOCHROME c MEMBRANE ALLEN B. BLAUROCK From the Cardiovascular Research Institute, San Francisco, California 94122 University of California, ABsrRAcr In the preceding paper a
Cell Cell
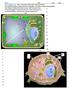 Go to cellsalive.com. Select Interactive Cell Models: Plant and Animal. Fill in the information on Plant and Animal Organelles, then Click on Start the Animation Select Plant or Animal Cell below the box.
Go to cellsalive.com. Select Interactive Cell Models: Plant and Animal. Fill in the information on Plant and Animal Organelles, then Click on Start the Animation Select Plant or Animal Cell below the box.
From Atoms to Cells: Fundamental Building Blocks. Models of atoms. A chemical connection
 From Atoms to Cells: A chemical connection Fundamental Building Blocks Matter - all materials that occupy space & have mass Matter is composed of atoms Atom simplest form of matter not divisible into simpler
From Atoms to Cells: A chemical connection Fundamental Building Blocks Matter - all materials that occupy space & have mass Matter is composed of atoms Atom simplest form of matter not divisible into simpler
Questions in Cell Biology
 Name: Questions in Cell Biology Directions: The following questions are taken from previous IB Final Papers on the subject of cell biology. Answer all questions. This will serve as a study guide for the
Name: Questions in Cell Biology Directions: The following questions are taken from previous IB Final Papers on the subject of cell biology. Answer all questions. This will serve as a study guide for the
Yara shwabkeh. Osama Alkhader. Heba Kalbouneh
 2 Yara shwabkeh Osama Alkhader Heba Kalbouneh CELL OVERVIEW -Note ; the important thing is to know how the organelles appear under the microscope - the stains we usually use in Histology are composed of
2 Yara shwabkeh Osama Alkhader Heba Kalbouneh CELL OVERVIEW -Note ; the important thing is to know how the organelles appear under the microscope - the stains we usually use in Histology are composed of
ACERTAIN degree of correlation between
 Electron Microscopy of Human Plasma Lipoprotein Separated by Ultracentrifugation By D. E. BEISCHER, PH.D. The particle size distribution of the Sf 30 "class" of serum lipoprotein was determined by the
Electron Microscopy of Human Plasma Lipoprotein Separated by Ultracentrifugation By D. E. BEISCHER, PH.D. The particle size distribution of the Sf 30 "class" of serum lipoprotein was determined by the
Ferritin-Conjugated Antibodies Used for Labeling of Organelles Involved
 Proc. Nat. Acad. Sci. USA Vol. 71, No. 5, pp. 2033-2037, May 1974 Ferritin-Conjugated Antibodies Used for Labeling of Organelles Involved in the Cellular Synthesis and Transport of Procollagen (ferritin-antibody
Proc. Nat. Acad. Sci. USA Vol. 71, No. 5, pp. 2033-2037, May 1974 Ferritin-Conjugated Antibodies Used for Labeling of Organelles Involved in the Cellular Synthesis and Transport of Procollagen (ferritin-antibody
ON THE PRESENCE OF A CILIATED COLUMNAR EPITHELIAL CELL TYPE WITHIN THE BOVINE CERVICAL MUCOSA 1
 ON THE PRESENCE OF A CILIATED COLUMNAR EPITHELIAL CELL TYPE WITHIN THE BOVINE CERVICAL MUCOSA 1 R. I. Wordinger, 2 J. B. Ramsey, I. F. Dickey and I. R. Hill, Jr. Clemson University, Clemson, South Carolina
ON THE PRESENCE OF A CILIATED COLUMNAR EPITHELIAL CELL TYPE WITHIN THE BOVINE CERVICAL MUCOSA 1 R. I. Wordinger, 2 J. B. Ramsey, I. F. Dickey and I. R. Hill, Jr. Clemson University, Clemson, South Carolina
Electron Microscope Studies of HeLa Cells Infected with Herpes Virus
 244 STOKER, M. G. P., SMITH, K. M. & Ross, R. W. (1958). J. gen. Microbiol. 19,244-249 Electron Microscope Studies of HeLa Cells Infected with Herpes Virus BY M: G. P. STOKER, K. M. SMITH AND R. W. ROSS
244 STOKER, M. G. P., SMITH, K. M. & Ross, R. W. (1958). J. gen. Microbiol. 19,244-249 Electron Microscope Studies of HeLa Cells Infected with Herpes Virus BY M: G. P. STOKER, K. M. SMITH AND R. W. ROSS
triplelayered unit for the plasma cell membrane component; two such Model parameters are assigned to three varieties of peripheral nerve
 STRUCTURAL PARAMETERS OF NERVE MYELIN* BY C. R. WORTHINGTON DEPARTMENT OF PHYSICS AND BIOPHYSICS RESEARCH DIVISION, UNIVERSITY OF MICHIGAN, ANN ARBOR Communicated by J. L. Oncley, April 7, 1969 Abstract.-Low-angle
STRUCTURAL PARAMETERS OF NERVE MYELIN* BY C. R. WORTHINGTON DEPARTMENT OF PHYSICS AND BIOPHYSICS RESEARCH DIVISION, UNIVERSITY OF MICHIGAN, ANN ARBOR Communicated by J. L. Oncley, April 7, 1969 Abstract.-Low-angle
INVESTIGATIVE OPHTHALMOLOGY. Corneal and conjunctival changes in dysproteinemia
 August 1969 Volume 8, Number 4 INVESTIGATIVE OPHTHALMOLOGY Corneal and conjunctival changes in dysproteinemia 7?. M. H. Pinkerton and David M. Robertson A case of dysproteinemia with corneal and conjunctival
August 1969 Volume 8, Number 4 INVESTIGATIVE OPHTHALMOLOGY Corneal and conjunctival changes in dysproteinemia 7?. M. H. Pinkerton and David M. Robertson A case of dysproteinemia with corneal and conjunctival
AMERICAN NATIONAL SCHOOL General Certificate of Education Advanced Level
 MERIN NTIONL SHOOL General ertificate of Education dvanced Level IOLOGY 9700/01 Paper 1 Multiple hoice lass 1 dditional Materials: Multiple hoice nswer Sheet Soft clean eraser Soft pencil (type or H is
MERIN NTIONL SHOOL General ertificate of Education dvanced Level IOLOGY 9700/01 Paper 1 Multiple hoice lass 1 dditional Materials: Multiple hoice nswer Sheet Soft clean eraser Soft pencil (type or H is
MORPHOLOGIC CHARACTERISTICS OF THE CHEMICALLY INDUCED ACROSOME REACTION IN HUMAN SPERMATOZOA*
 FERTILITY AND STERILITY Copyright 1979 The American Fertility Society Vol. 32, No.1, July 1979 Printed in U.SA. MORPHOLOGIC CHARACTERISTICS OF THE CHEMICALLY INDUCED ACROSOME REACTION IN HUMAN SPERMATOZOA*
FERTILITY AND STERILITY Copyright 1979 The American Fertility Society Vol. 32, No.1, July 1979 Printed in U.SA. MORPHOLOGIC CHARACTERISTICS OF THE CHEMICALLY INDUCED ACROSOME REACTION IN HUMAN SPERMATOZOA*
High resolution structural evidence suggests the Sarcoplasmic Reticulum forms microdomains with Acidic Stores (lyososomes) in the heart.
 High resolution structural evidence suggests the Sarcoplasmic Reticulum forms microdomains with Acidic Stores (lyososomes) in the heart. Daniel Aston, Rebecca A. Capel, Kerrie L. Ford, Helen C. Christian,
High resolution structural evidence suggests the Sarcoplasmic Reticulum forms microdomains with Acidic Stores (lyososomes) in the heart. Daniel Aston, Rebecca A. Capel, Kerrie L. Ford, Helen C. Christian,
Thursday, October 16 th
 Thursday, October 16 th Good morning. Those of you needing to take the Enzymes and Energy Quiz will start very soon. Students who took the quiz Wednesday: Please QUIETLY work on the chapter 6 reading guide.
Thursday, October 16 th Good morning. Those of you needing to take the Enzymes and Energy Quiz will start very soon. Students who took the quiz Wednesday: Please QUIETLY work on the chapter 6 reading guide.
STUDIES OF THE HUMAN UNFERTILIZED TUBAL OVUM*t
 FERTILITY AND STERILITY Copyright @ 1973 by The Williams & Wilkins Co. Vol. 24, No.8, August 1973 Printed in U.S.A. STUDIES OF THE HUMAN UNFERTILIZED TUBAL OVUM*t C. NORIEGA, M.D., AND C. OBERTI, M.D.
FERTILITY AND STERILITY Copyright @ 1973 by The Williams & Wilkins Co. Vol. 24, No.8, August 1973 Printed in U.S.A. STUDIES OF THE HUMAN UNFERTILIZED TUBAL OVUM*t C. NORIEGA, M.D., AND C. OBERTI, M.D.
Lecture 15. Membrane Proteins I
 Lecture 15 Membrane Proteins I Introduction What are membrane proteins and where do they exist? Proteins consist of three main classes which are classified as globular, fibrous and membrane proteins. A
Lecture 15 Membrane Proteins I Introduction What are membrane proteins and where do they exist? Proteins consist of three main classes which are classified as globular, fibrous and membrane proteins. A
The Cytoplasm Li Shulei Department of Histology & Embryology
 The Cytoplasm Li Shulei lishulei@tom.com Department of Histology & Embryology Cell components Cytoplasm Plasma membrane Organelles Cytoplasmic deposits Cytoskeleton Cytosol ( Matrix ) Nucleus Plasma membrane
The Cytoplasm Li Shulei lishulei@tom.com Department of Histology & Embryology Cell components Cytoplasm Plasma membrane Organelles Cytoplasmic deposits Cytoskeleton Cytosol ( Matrix ) Nucleus Plasma membrane
Participation of Endogenous Fatty Acids in Ca 2+ Release Activation from Mitochondria
 Gen. Physiol. Biophys. (1985), 4, 549 556 549 Participation of Endogenous Fatty Acids in Ca 2+ Release Activation from Mitochondria B. I. MEDVEDEV, E. P. SEVERINA, V. G. GOGVADZE, E. A. CHUKHLOVA and Yu.
Gen. Physiol. Biophys. (1985), 4, 549 556 549 Participation of Endogenous Fatty Acids in Ca 2+ Release Activation from Mitochondria B. I. MEDVEDEV, E. P. SEVERINA, V. G. GOGVADZE, E. A. CHUKHLOVA and Yu.
SBI3U7 Cell Structure & Organelles. 2.2 Prokaryotic Cells 2.3 Eukaryotic Cells
 SBI3U7 Cell Structure & Organelles 2.2 Prokaryotic Cells 2.3 Eukaryotic Cells No nucleus Prokaryotic Cells No membrane bound organelles Has a nucleus Eukaryotic Cells Membrane bound organelles Unicellular
SBI3U7 Cell Structure & Organelles 2.2 Prokaryotic Cells 2.3 Eukaryotic Cells No nucleus Prokaryotic Cells No membrane bound organelles Has a nucleus Eukaryotic Cells Membrane bound organelles Unicellular
Chapter 1 Plasma membranes
 1 of 5 TEXTBOOK ANSWERS Chapter 1 Plasma membranes Recap 1.1 1 The plasma membrane: keeps internal contents of the cell confined to one area keeps out foreign molecules that damage or destroy the cell
1 of 5 TEXTBOOK ANSWERS Chapter 1 Plasma membranes Recap 1.1 1 The plasma membrane: keeps internal contents of the cell confined to one area keeps out foreign molecules that damage or destroy the cell
Cell are made up of organelles. An ORGANELLE is a specialized subunit within a cell that has a specific function.
 Plant and Animal Cells The Cell Theory All living things are made up of one or more cells. All cells come from other cells. Organization of Living Things Cell are made up of organelles. An ORGANELLE is
Plant and Animal Cells The Cell Theory All living things are made up of one or more cells. All cells come from other cells. Organization of Living Things Cell are made up of organelles. An ORGANELLE is
Cell Membrane Structure (1.3) IB Diploma Biology
 Cell Membrane Structure (1.3) IB Diploma Biology Essential idea: The structure of biological membranes makes them fluid and dynamic http://www.flickr.com/photos/edsweeney/6346198056/ 1.3.1 Phospholipids
Cell Membrane Structure (1.3) IB Diploma Biology Essential idea: The structure of biological membranes makes them fluid and dynamic http://www.flickr.com/photos/edsweeney/6346198056/ 1.3.1 Phospholipids
Classification of Lipids
 Classification of Lipids Neutral Lipids Amphipathic Lipids Amphipathic Lipids Most cell-membrane lipids are one of two main classes of amphipathic hydrolyzable lipids. Glycerophospholipids (phosphoglycerides):
Classification of Lipids Neutral Lipids Amphipathic Lipids Amphipathic Lipids Most cell-membrane lipids are one of two main classes of amphipathic hydrolyzable lipids. Glycerophospholipids (phosphoglycerides):
CELL PART OF THE DAY. Chapter 7: Cell Structure and Function
 CELL PART OF THE DAY Chapter 7: Cell Structure and Function Cell Membrane Cell membranes are composed of two phospholipid layers. Cell membrane is flexible, not rigid The cell membrane has two major functions.
CELL PART OF THE DAY Chapter 7: Cell Structure and Function Cell Membrane Cell membranes are composed of two phospholipid layers. Cell membrane is flexible, not rigid The cell membrane has two major functions.
Introduction. Biochemistry: It is the chemistry of living things (matters).
 Introduction Biochemistry: It is the chemistry of living things (matters). Biochemistry provides fundamental understanding of the molecular basis for the function and malfunction of living things. Biochemistry
Introduction Biochemistry: It is the chemistry of living things (matters). Biochemistry provides fundamental understanding of the molecular basis for the function and malfunction of living things. Biochemistry
Determination of the Distribution of Cilia on the Surface of the Mantle of Cypraea caputserpentis utilizing Scanning Electron Microscopy
 Determination of the Distribution of Cilia on the Surface of the Mantle of Cypraea caputserpentis utilizing Scanning Electron Microscopy DURATION September 10, 1990- May 7, 1991 Tracie A. Yokoi Advisor
Determination of the Distribution of Cilia on the Surface of the Mantle of Cypraea caputserpentis utilizing Scanning Electron Microscopy DURATION September 10, 1990- May 7, 1991 Tracie A. Yokoi Advisor
SEASONAL CHANGES OF AVOCADO LIPIDS DURING FRUIT DEVELOPMENT AND STORAGE
 California Avocado Society 1968 Yearbook 52: 102-108 SEASONAL CHANGES OF AVOCADO LIPIDS DURING FRUIT DEVELOPMENT AND STORAGE Yoshio Kikuta Present address: Department of Botany, Faculty of Agriculture,
California Avocado Society 1968 Yearbook 52: 102-108 SEASONAL CHANGES OF AVOCADO LIPIDS DURING FRUIT DEVELOPMENT AND STORAGE Yoshio Kikuta Present address: Department of Botany, Faculty of Agriculture,
Discipline Courses-I. Semester-I. Paper II: Plant Cell Biology. Unit-II. Lesson: Cell Membrane Structure. Lesson Developer: Rina Majumdar
 Discipline Courses-I Semester-I Paper II: Plant Cell Biology Unit-II Lesson: Cell Membrane Structure Lesson Developer: Rina Majumdar College/Department: Maitreyi College, University of Delhi Institute
Discipline Courses-I Semester-I Paper II: Plant Cell Biology Unit-II Lesson: Cell Membrane Structure Lesson Developer: Rina Majumdar College/Department: Maitreyi College, University of Delhi Institute
Organelles. copyright cmassengale 1
 Organelles copyright cmassengale 1 Organelles Very small (Microscopic) Perform various functions for a cell Found in the cytoplasm May or may not be membrane-bound 2 Animal Cell Organelles Nucleolus Nucleus
Organelles copyright cmassengale 1 Organelles Very small (Microscopic) Perform various functions for a cell Found in the cytoplasm May or may not be membrane-bound 2 Animal Cell Organelles Nucleolus Nucleus
History of the Cell. History of the Cell 10/24/2013. Unit 3: Cellular Structure and Function. Robert Hooke (1665) Robert Hooke (1665)
 Unit 3: Cellular Structure and Function Mr. Hulse BVHS 2013-2014 Unit 3: Learning Targets 1-9 History of the Cell Robert Hooke (1665) 1 st person to see a cell Observed a piece of cork using a microscope
Unit 3: Cellular Structure and Function Mr. Hulse BVHS 2013-2014 Unit 3: Learning Targets 1-9 History of the Cell Robert Hooke (1665) 1 st person to see a cell Observed a piece of cork using a microscope
10/13/11. Cell Theory. Cell Structure
 Cell Structure Grade 12 Biology Cell Theory All organisms are composed of one or more cells. Cells are the smallest living units of all living organisms. Cells arise only by division of a previously existing
Cell Structure Grade 12 Biology Cell Theory All organisms are composed of one or more cells. Cells are the smallest living units of all living organisms. Cells arise only by division of a previously existing
EDUCATIONAL OBJECTIVES
 EDUCATIONAL OBJECTIVES The lectures and reading assignments of BIS 2A are designed to convey a large number of facts and concepts that have evolved from modern studies of living organisms. In order to
EDUCATIONAL OBJECTIVES The lectures and reading assignments of BIS 2A are designed to convey a large number of facts and concepts that have evolved from modern studies of living organisms. In order to
ELECTRON MICROSCOPIC STUDIES ON EQUINE ENCEPHALOSIS VIRUS
 Onderstepoort]. vet. Res. 40 (2), 53-58 (1973) ELECTRON MICROSCOPIC STUDIES ON EQUINE ENCEPHALOSIS VIRUS G. LECATSAS, B. J. ERASMUS and H. J. ELS, Veterinary Research Institute, Onderstepoort ABSTRACT
Onderstepoort]. vet. Res. 40 (2), 53-58 (1973) ELECTRON MICROSCOPIC STUDIES ON EQUINE ENCEPHALOSIS VIRUS G. LECATSAS, B. J. ERASMUS and H. J. ELS, Veterinary Research Institute, Onderstepoort ABSTRACT
AP Biology Cells: Chapters 4 & 5
 AP Biology Cells: Chapters 4 & 5 Multiple Choice Identify the choice that best completes the statement or answers the question. 1. The was the first unifying principle of biology. a. spontaneous generation
AP Biology Cells: Chapters 4 & 5 Multiple Choice Identify the choice that best completes the statement or answers the question. 1. The was the first unifying principle of biology. a. spontaneous generation
ON THE THICKNESS OF THE UNIT MEMBRANE
 Published Online: 1 May, 1963 Supp Info: http://doi.org/10.1083/jcb.17.2.413 Downloaded from jcb.rupress.org on September 1, 2018 ON THE THICKNESS OF THE UNIT MEMBRANE TOSHIYUKI YAMAMOTO, M.D. From the
Published Online: 1 May, 1963 Supp Info: http://doi.org/10.1083/jcb.17.2.413 Downloaded from jcb.rupress.org on September 1, 2018 ON THE THICKNESS OF THE UNIT MEMBRANE TOSHIYUKI YAMAMOTO, M.D. From the
PORE-LIKE STRUCTURES IN BIOLOGICAL MEMBRANES
 J. Cell Sci. 25, 157-161 (1977) 157 Printed in Great Britain PORE-LIKE STRUCTURES IN BIOLOGICAL MEMBRANES L. ORCI, A. PERRELET, FRANCINE MALAISSE-LAGAE AND P. VASSALLI* Institute of Histology and Embryology,
J. Cell Sci. 25, 157-161 (1977) 157 Printed in Great Britain PORE-LIKE STRUCTURES IN BIOLOGICAL MEMBRANES L. ORCI, A. PERRELET, FRANCINE MALAISSE-LAGAE AND P. VASSALLI* Institute of Histology and Embryology,
Fine Structure of the Normal Trigeminal Ganglion in the Cat and Monkey*
 Fine Structure of the Normal Trigeminal Ganglion in the Cat and Monkey* DAVID S. MAXWELL, PH.D. Principal Contributor and Leader of Discussion HE inclusion of animal material m a y be justified as a means
Fine Structure of the Normal Trigeminal Ganglion in the Cat and Monkey* DAVID S. MAXWELL, PH.D. Principal Contributor and Leader of Discussion HE inclusion of animal material m a y be justified as a means
Scanning Electron Microscopy of Thiobacilli
 Arch. Microbiol. 99, 323-329 (1974) 0 by Springer-Verlag 1974 Scanning Electron Microscopy of Thiobacilli Grown on Colloïdal Sulfur J. Baldensperger", L. J. Guarraia**, and W. J. Humphreys*** Department
Arch. Microbiol. 99, 323-329 (1974) 0 by Springer-Verlag 1974 Scanning Electron Microscopy of Thiobacilli Grown on Colloïdal Sulfur J. Baldensperger", L. J. Guarraia**, and W. J. Humphreys*** Department
First to View Cells. copyright cmassengale
 CELL THEORY All living things are made of cells Cells are the basic unit of structure and function in an organism (basic unit of life) Cells come from the reproduction of existing cells (cell division)
CELL THEORY All living things are made of cells Cells are the basic unit of structure and function in an organism (basic unit of life) Cells come from the reproduction of existing cells (cell division)
8/7/18. UNIT 2: Cells Chapter 3: Cell Structure and Function. I. Cell Theory (3.1) A. Early studies led to the development of the cell theory
 8/7/18 UNIT 2: Cells Chapter 3: Cell Structure and Function I. Cell Theory (3.1) A. Early studies led to the development of the cell theory 1. Discovery of Cells a. Robert Hooke (1665)-Used compound microscope
8/7/18 UNIT 2: Cells Chapter 3: Cell Structure and Function I. Cell Theory (3.1) A. Early studies led to the development of the cell theory 1. Discovery of Cells a. Robert Hooke (1665)-Used compound microscope
THE ENZYMATIC PREPARATION OF ISOLATED INTACT PARENCHYMAL CELLS FROM RAT LIVER
 Published Online: 1 December, 1967 Supp Info: http://doi.org/10.1083/jcb.35.3.675 Downloaded from jcb.rupress.org on November 18, 2018 THE ENZYMATIC PREPARATION OF ISOLATED INTACT PARENCHYMAL CELLS FROM
Published Online: 1 December, 1967 Supp Info: http://doi.org/10.1083/jcb.35.3.675 Downloaded from jcb.rupress.org on November 18, 2018 THE ENZYMATIC PREPARATION OF ISOLATED INTACT PARENCHYMAL CELLS FROM
Molecular Cell Biology. Prof. D. Karunagaran. Department of Biotechnology. Indian Institute of Technology Madras
 Molecular Cell Biology Prof. D. Karunagaran Department of Biotechnology Indian Institute of Technology Madras Module 4 Membrane Organization and Transport Across Membranes Lecture 1 Cell Membrane and Transport
Molecular Cell Biology Prof. D. Karunagaran Department of Biotechnology Indian Institute of Technology Madras Module 4 Membrane Organization and Transport Across Membranes Lecture 1 Cell Membrane and Transport
THE PROBABLE ROLE OF PHOSPHATIDYL CHOLINES IN THE TANNIC ACID ENHANCEMENT OF CYTOMEMBRANE
 THE PROBABLE ROLE OF PHOSPHATIDYL CHOLINES IN THE TANNIC ACID ENHANCEMENT OF CYTOMEMBRANE ELECTRON CONTRAST MOSHE KALINA and DANIEL C. PEASE From the Department of Anatomy, School of Medicine, University
THE PROBABLE ROLE OF PHOSPHATIDYL CHOLINES IN THE TANNIC ACID ENHANCEMENT OF CYTOMEMBRANE ELECTRON CONTRAST MOSHE KALINA and DANIEL C. PEASE From the Department of Anatomy, School of Medicine, University
R,;habdomyosarcoma, the most common
 Fine-structural classification of orbital rhabdomyosarcoma Arnold J. Kroll Six cases of orbital rhabdomyosarcoma were studied with the electron microscope. Tumor cells (rhabdomyoblasts) could be classified
Fine-structural classification of orbital rhabdomyosarcoma Arnold J. Kroll Six cases of orbital rhabdomyosarcoma were studied with the electron microscope. Tumor cells (rhabdomyoblasts) could be classified
however, and the present communication is concerned with some of
 THE AGGLUTINATION OF HUMAN ERYTHROCYTES MODIFIED BY TREATMENT WITH NEWCASTLE DISEASE AND INFLUENZA VIRUS' ALFRED L. FLORMAN' Pediatric Service and Division of Bacteriology, The Mount Sinai Hospital, New
THE AGGLUTINATION OF HUMAN ERYTHROCYTES MODIFIED BY TREATMENT WITH NEWCASTLE DISEASE AND INFLUENZA VIRUS' ALFRED L. FLORMAN' Pediatric Service and Division of Bacteriology, The Mount Sinai Hospital, New
Ch7: Membrane Structure & Function
 Ch7: Membrane Structure & Function History 1915 RBC membranes studied found proteins and lipids 1935 membrane mostly phospholipids 2 layers 1950 electron microscopes supported bilayer idea (Sandwich model)
Ch7: Membrane Structure & Function History 1915 RBC membranes studied found proteins and lipids 1935 membrane mostly phospholipids 2 layers 1950 electron microscopes supported bilayer idea (Sandwich model)
Basic Structure of a Cell
 Basic Structure of a Cell 1 Introduction to Cells Cells are the basic units of organisms Cells can only be observed under microscope Basic types of cells: Animal Cell Plant Cell Bacterial Cell 2 Number
Basic Structure of a Cell 1 Introduction to Cells Cells are the basic units of organisms Cells can only be observed under microscope Basic types of cells: Animal Cell Plant Cell Bacterial Cell 2 Number
Membrane biochemistry. Red blood cell ghost Only plasmalemma Size known (7 µm) Gorter & Grendel 1925 Tension on surface Enough lipid for 2 layers
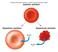 Membrane biochemistry Red blood cell ghost Only plasmalemma Size known (7 µm) Gorter & Grendel 1925 Tension on surface Enough lipid for 2 layers Fig. 6.5 Osmosis water moves passively from high WATER
Membrane biochemistry Red blood cell ghost Only plasmalemma Size known (7 µm) Gorter & Grendel 1925 Tension on surface Enough lipid for 2 layers Fig. 6.5 Osmosis water moves passively from high WATER
Two Types of Vesicles
 Eur. J. Biochem. 41,37-43 (1974) Two Types of Vesicles from the Erythrocyte-Ghost Membrane Differing in Surface Charge Separation and Characterization by Preparative Free-Flow Electrophoresis Hans-G. HEIDRICH
Eur. J. Biochem. 41,37-43 (1974) Two Types of Vesicles from the Erythrocyte-Ghost Membrane Differing in Surface Charge Separation and Characterization by Preparative Free-Flow Electrophoresis Hans-G. HEIDRICH
The Cell Organelles. Eukaryotic cell. The plasma membrane separates the cell from the environment. Plasma membrane: a cell s boundary
 Eukaryotic cell The Cell Organelles Enclosed by plasma membrane Subdivided into membrane bound compartments - organelles One of the organelles is membrane bound nucleus Cytoplasm contains supporting matrix
Eukaryotic cell The Cell Organelles Enclosed by plasma membrane Subdivided into membrane bound compartments - organelles One of the organelles is membrane bound nucleus Cytoplasm contains supporting matrix
CELLS CONTAINING LANGERHANS GRANULES IN HUMAN LYMPH NODES OF DERMATOPATHIC LYMPHADENOPATHY*
 THS JOURNAL OF INVEBTIOATIVR DERMATOLOGY Copyright 1969 by The Williams & Wilkins Co. Vol. 93, No. 4 Printed in U.S.A. CELLS CONTAINING LANGERHANS GRANULES IN HUMAN LYMPH NODES OF DERMATOPATHIC LYMPHADENOPATHY*
THS JOURNAL OF INVEBTIOATIVR DERMATOLOGY Copyright 1969 by The Williams & Wilkins Co. Vol. 93, No. 4 Printed in U.S.A. CELLS CONTAINING LANGERHANS GRANULES IN HUMAN LYMPH NODES OF DERMATOPATHIC LYMPHADENOPATHY*
CELL MEMBRANES (MAS)
 CELL MEMBRANES (MAS) 1 CELL MEMBRANE area of the cell immediately surrounding the cytoplasm the most conserved structure in living cells. Every living thing on this planet has some type of membrane 2 Anatomy
CELL MEMBRANES (MAS) 1 CELL MEMBRANE area of the cell immediately surrounding the cytoplasm the most conserved structure in living cells. Every living thing on this planet has some type of membrane 2 Anatomy
The Cell and Cellular transport
 Cell theory (1838): The Cell 1. All organisms are composed of one or more cells, and the life processes of metabolism and heredity occur within these cells. 2. Cells are the smallest living things, the
Cell theory (1838): The Cell 1. All organisms are composed of one or more cells, and the life processes of metabolism and heredity occur within these cells. 2. Cells are the smallest living things, the
1.4 Page 1 Cell Membranes S. Preston 1
 AS Unit 1: Basic Biochemistry and Cell Organisation Name: Date: Topic 1.3 Cell Membranes and Transport Page 1 1.3 Cell Membranes and Transport from your syllabus l. Cell Membrane Structure 1. Read and
AS Unit 1: Basic Biochemistry and Cell Organisation Name: Date: Topic 1.3 Cell Membranes and Transport Page 1 1.3 Cell Membranes and Transport from your syllabus l. Cell Membrane Structure 1. Read and
Membranes. Chapter 5. Membrane Structure
 Membranes Chapter 5 Membrane Structure Lipid Bilayer model: - double phospholipid layer - Gorter & Grendel: 1925 Fluid Mosaic model: consist of -phospholipids arranged in a bilayer -globular proteins inserted
Membranes Chapter 5 Membrane Structure Lipid Bilayer model: - double phospholipid layer - Gorter & Grendel: 1925 Fluid Mosaic model: consist of -phospholipids arranged in a bilayer -globular proteins inserted
AN ULTRASTRUCTURA-L STUDY OF THE MEMBRANES OF KERATINIZING WOOL FOLLICLE CELLS
 J. Cell Sci. II, 205-219 (1972) 205 Printed in Great Britain AN ULTRASTRUCTURA-L STUDY OF THE MEMBRANES OF KERATINIZING WOOL FOLLICLE CELLS D.F.G.ORWIN AND R.W.THOMSON Wool Research Organisation of N.Z.
J. Cell Sci. II, 205-219 (1972) 205 Printed in Great Britain AN ULTRASTRUCTURA-L STUDY OF THE MEMBRANES OF KERATINIZING WOOL FOLLICLE CELLS D.F.G.ORWIN AND R.W.THOMSON Wool Research Organisation of N.Z.
Chapter 7. (7-1 and 7-2) A Tour of the Cell
 Chapter 7 (7-1 and 7-2) A Tour of the Cell Microscopes as Windows to the World of Cells Cells were first described in 1665 by Robert Hooke. By the mid-1800s, the accumulation of scientific evidence led
Chapter 7 (7-1 and 7-2) A Tour of the Cell Microscopes as Windows to the World of Cells Cells were first described in 1665 by Robert Hooke. By the mid-1800s, the accumulation of scientific evidence led
IT has been shown (Chou, 1957 a, b) that there are three kinds of lipid
 279 The Ultra-fine Structure of Lipid Globules in the Neurones of Helix aspersa By J. T. Y. CHOU and G. A. MEEK (From the Cytological Laboratory, Department of Zoology; and Department of Human Anatomy,
279 The Ultra-fine Structure of Lipid Globules in the Neurones of Helix aspersa By J. T. Y. CHOU and G. A. MEEK (From the Cytological Laboratory, Department of Zoology; and Department of Human Anatomy,
Chapter 2 Cell. Zhou Li Prof. Dept. of Histology and Embryology
 Chapter 2 Cell Zhou Li Prof. Dept. of Histology and Embryology The inner life of the cell Ⅰ. Plasma membrane (Plasmalemma) 1.1 The structure Unit membrane: inner layer 3-layered structure outer layer mediat
Chapter 2 Cell Zhou Li Prof. Dept. of Histology and Embryology The inner life of the cell Ⅰ. Plasma membrane (Plasmalemma) 1.1 The structure Unit membrane: inner layer 3-layered structure outer layer mediat
Phosphatase Activity of Drosophila Salivary Glands
 Phosphatase Activity of Drosophila Salivary Glands BY W. L. DOYLE (From the Department of Anatomy, University of Chicago) THE presence of alkaline phosphatase in chromosomes has been demonstrated by means
Phosphatase Activity of Drosophila Salivary Glands BY W. L. DOYLE (From the Department of Anatomy, University of Chicago) THE presence of alkaline phosphatase in chromosomes has been demonstrated by means
Lesson Overview. 7.2 Cell Structure
 7.2 Cell Organization The eukaryotic cell can be divided into two major parts: the nucleus and the cytoplasm. The cytoplasm is the fluid portion of the cell outside the nucleus. Prokaryotes do not have
7.2 Cell Organization The eukaryotic cell can be divided into two major parts: the nucleus and the cytoplasm. The cytoplasm is the fluid portion of the cell outside the nucleus. Prokaryotes do not have
THE FORM OF HAEMOGLOBIN IN THE ERYTHROCYTES OF THE COD, GADUS CALLARIAS
 J. Cell Set. 8, 407-412 (1971) 407 Printed in Great Britain THE FORM OF HAEMOGLOBIN IN THE ERYTHROCYTES OF THE COD, GADUS CALLARIAS N.W.THOMAS Department of Anatomy, Marischal College, Aberdeen, Scotland
J. Cell Set. 8, 407-412 (1971) 407 Printed in Great Britain THE FORM OF HAEMOGLOBIN IN THE ERYTHROCYTES OF THE COD, GADUS CALLARIAS N.W.THOMAS Department of Anatomy, Marischal College, Aberdeen, Scotland
(d) are made mainly of lipids and of proteins that lie like thin sheets on the membrane surface
 Which of the following statements is no true? Biological membranes (a) are composed partly of amphipathic lipids (b) have hydrophobic and hydrophilic regions (c) are typically in a fluid state (d) are
Which of the following statements is no true? Biological membranes (a) are composed partly of amphipathic lipids (b) have hydrophobic and hydrophilic regions (c) are typically in a fluid state (d) are
A Tour of the Cell. Ch. 7
 A Tour of the Cell Ch. 7 Cell Theory O All organisms are composed of one or more cells. O The cell is the basic unit of structure and organization of organisms. O All cells come from preexisting cells.
A Tour of the Cell Ch. 7 Cell Theory O All organisms are composed of one or more cells. O The cell is the basic unit of structure and organization of organisms. O All cells come from preexisting cells.
BIOLOGY 111. CHAPTER 3: The Cell: The Fundamental Unit of Life
 BIOLOGY 111 CHAPTER 3: The Cell: The Fundamental Unit of Life The Cell: The Fundamental Unit of Life Learning Outcomes 3.1 Explain the similarities and differences between prokaryotic and eukaryotic cells
BIOLOGY 111 CHAPTER 3: The Cell: The Fundamental Unit of Life The Cell: The Fundamental Unit of Life Learning Outcomes 3.1 Explain the similarities and differences between prokaryotic and eukaryotic cells
I. Fluid Mosaic Model A. Biological membranes are lipid bilayers with associated proteins
 Lecture 6: Membranes and Cell Transport Biological Membranes I. Fluid Mosaic Model A. Biological membranes are lipid bilayers with associated proteins 1. Characteristics a. Phospholipids form bilayers
Lecture 6: Membranes and Cell Transport Biological Membranes I. Fluid Mosaic Model A. Biological membranes are lipid bilayers with associated proteins 1. Characteristics a. Phospholipids form bilayers
