DOI /j x
|
|
|
- Candice Williams
- 6 years ago
- Views:
Transcription
1 THERAPEUTICS DOI /j x Diffuse cutaneous leishmaniasis responds to miltefosine but then relapses O. Zerpa, M. Ulrich, B. Blanco, M. Polegre, A. Avila, N. Matos,* I. Mendoza, F. Pratlong,à C. Ravelà and J. Convit Institute of Biomedicine, Ministry of Health, Central University of Venezuela, Esq. San Nicolás, San José, Caracas, Venezuela *Regional Service of Public Health Dermatology, Apure, Venezuela Regional Service of Public Health Dermatology, Lara, Venezuela àlaboratoire de Parasitologie, Centre National de Référence des Leishmania and WHO Collaborating Centre on Leishmaniasis, CHU de Montpellier and Université Montpellier 1, Montpellier, France Summary Correspondence Olga Zerpa, Instituto de Biomedicina, Apartado postal 4043, Caracas 1010A, Venezuela. Accepted for publication 14 December 2006 Key words adverse effects, cell-mediated immunity, diffuse cutaneous leishmaniasis, miltefosine Conflicts of interest None declared. Background Diffuse cutaneous leishmaniasis (DCL), although rare, is profoundly incapacitating. At present there is no successful treatment for this progressive protozoan infection, which is associated with the absence of specific cell-mediated immunity (CMI) to Leishmania. This disease shares features with visceral leishmaniasis (VL), including specific CMI inactivity during active disease and a heavy parasitic burden, but VL responds well to treatment. Miltefosine is the first orally administered drug which has shown efficacy in the treatment of VL; it has not been adequately evaluated in the treatment of DCL. Objectives To evaluate the efficacy of miltefosine in the treatment of DCL, using clinical, parasitological, histopathological and immunological criteria. Methods Sixteen patients with DCL were treated with miltefosine, 2Æ0 2Æ5 mgkg )1 daily, for variable periods of time ( days). Patients were hospitalized for the first month and evaluated every 2 weeks until the termination of treatment with routine laboratory chemistry, percentage clinical improvement, presence of parasites in skin smears, growth of parasites in culture medium and in hamsters, histopathological characteristics of the granulomas, adverse side-effects, and reactivity to leishmanin skin test antigen. Further cycles of treatment were given in some of these patients, particularly after suspension of treatment was followed by relapse. Results Patients showed dramatic clinical improvement and reduction in the parasite burden by day 15 after the initiation of treatment, which continued while treatment was maintained. By day 45, 15 patients showed 80 90% clinical improvement. Nevertheless, suspension of treatment was followed by the development of new lesions in all but one patient. Inoculation in hamsters was observed to be the most sensitive technique to detect persisting parasites. Adverse events were very mild. Conclusions Miltefosine produced a dramatic clinical and parasitological response in patients with DCL and improvement continued during drug administration, but with a single exception all patients presented new lesions after suspension of treatment. There was no histological or skin test evidence to suggest the development of CMI during treatment, which may be an indispensable criterion for the evaluation of potentially effective drugs against DCL. Leishmaniasis is an infectious disease caused by flagellated protozoa belonging to the genus Leishmania, transmitted to humans by a phlebotomine insect vector bite. The disease is characterized by a clinical, histopathological and immunological spectrum that depends on the immunological capacity of the host, the species and virulence of the parasite and poorly 1
2 2 Miltefosine in cutaneous leishmaniasis, O. Zerpa et al. defined environmental factors. Clinical manifestations of leishmaniasis include cutaneous, mucosal and visceral lesions. Diffuse cutaneous leishmaniasis (DCL) is a rare form of the disease that is associated with a defective cell-mediated immune response to the Leishmania parasite, 1 allowing its uncontrolled multiplication. This form of leishmaniasis was described in Venezuela by Convit and Lapenta in It occurs as isolated cases in Venezuela, the Dominican Republic, Brazil, Mexico, Bolivia, Colombia, Peru and Africa. 3 In America, DCL is usually caused by parasites of the Leishmania (Leishmania) subgenus, most frequently L. (L.) amazonensis, 3 although cases of DCL produced by L. (Viannia) braziliensis have been reported in immunocompromised individuals. In the Old World, the most frequent cause of DCL is L. aethiopica. DCL usually begins in infancy and is characterized clinically by the appearance of erythematous or skin-coloured papules, plaques or nodules, generally in localized areas. These may be asymmetrical lesions affecting a single limb or symmetrical but limited to upper or lower limbs. 4,5 The lesions usually do not ulcerate unless they are traumatized. When the mucosa is affected, the inflammatory reaction is not severe. The lesions can remain localized for a long period of time with little modification and subsequently disseminate over the body surface in a symmetrical manner. 5 From the immunological point of view, deficiencies in cytokines and accessory signals are associated with parasitespecific anergy. In the epidermis of patients with DCL, Tapia et al. 6 found few CD1a+, CD83+ Langerhans cells, and defective response of keratinocytes in the expression of HLA-DR or intercellular adhesion molecule-1. There is also a deficiency in the production of interleukin (IL)-6 by epidermal cells, which indicates defective production of monokines by antigen-presenting cells. 6,7 The granulomas present in patients with DCL are characterized by a predominantly T-helper 2 response (IL-4, IL-5, IL-10), with a high percentage of naïve T lymphocytes that do not react to the parasite. The leishmanin skin test is negative except in rare cases where it is positive initially and subsequently becomes negative. 4,6,7 The production of antibodies against Leishmania in DCL is elevated, but they do not play a significant protective role. Ulrich et al. have demonstrated that the predominant isotype in the majority of these patients is IgG4, which does not activate the complement cascade. 8,9 There is no effective treatment for DCL. Anti-Leishmania drugs often produce temporary improvement in the clinical manifestations, but this initial activity is inexorably followed by relapse. 3,4 The unremitting course of this disease is often accompanied by severe psychological and social repercussions in those affected. In recent years, success has been reported in the treatment of visceral and cutaneous leishmaniasis with miltefosine, an orally administered medication that was originally developed as an antineoplastic drug. 10 Miltefosine is an alkylphospholipid, an analogue of phosphocholine. Its chemical similarity to the natural phospholipids of cellular membranes suggests that miltefosine probably inhibits transmembrane signals and the synthesis of the cellular membrane. Miltefosine, administered by the oral route at a dose of 100 mg daily for 4 weeks in adults and 2Æ5 mgkg )1 daily in children, has been used in the treatment of visceral leishmaniasis (VL), with a cure rate of 95% and good tolerance in the patients. 10 In American cutaneous leishmaniasis varying rates of cure from 53% to 91% have been reported using a dose of 2Æ5 mgkg )1 daily, depending upon the species of Leishmania causing the infection. 11 Based on previous reports of the efficacy of miltefosine in the treatment of other forms of leishmaniasis, a previous case report showing encouraging results in the treatment of a single case of DCL, 12 and the absence of any definitive treatment for this disease, in this study we have evaluated the therapeutic efficacy of miltefosine in the treatment of DCL in a significantly larger group of patients. Patients and methods Patients Sixteen patients from rural Venezuela, with a diagnosis of DCL confirmed in the Leishmaniasis Clinic of the Institute of Biomedicine, were included in the study. All had been followed for years and had received multiple cycles of treatment with anti-leishmania drugs and immunotherapy with killed Leishmania promastigotes and bacille Calmette-Guérin. The clinical protocol was approved by the Ethical Committee of the Institute. The patients or legal representatives of minors signed informed consent forms before their admission into the study. Treatment A detailed clinical history of each patient was compiled. They were clinically examined, and photographs of lesions were taken, as were biopsies of skin lesions for histopathological study, cultivation of parasites in artificial media and inoculation into hamsters. Skin smears also were taken for parasitological evaluation. Laboratory examinations included full blood count, blood glucose, urea, creatinine, aspartate aminotransferase, alanine aminotransferase, alkaline phosphatase, total and fractionated bilirubin, human immunodeficiency virus and Venereal Disease Research Laboratory test. All of the patients were treated with miltefosine in a dose of 2 2Æ5 mgkg )1 daily and were hospitalized during the first month of treatment. They were evaluated clinically and had weekly laboratory examinations and photographs taken of their lesions. One month after completing treatment in the hospital the patients were released, after verifying that there were no severe secondary effects. Biweekly monitoring until the treatment was terminated included clinical evaluation, laboratory examinations (full blood count, hepatic and renal profiles), skin biopsies for culture, animal inoculation and histopathology, and skin smears. A standard leishmanin test was performed after completion of the first cycle of treatment.
3 Miltefosine in cutaneous leishmaniasis, O. Zerpa et al. 3 Histopathological study Biopsies were processed and stained with haematoxylin and eosin, to verify the presence or absence of parasites and to evaluate the structure of the granulomas. Skin smears were stained with Giemsa to detect parasites. Cultures and inoculation in experimental animals After washing the skin biopsy with phosphate-buffered saline (PBS) containing penicillin 2000 IU ml )1 and gentle maceration in a tissue grinder, 0Æ1 ml of the suspension was seeded in tubes of blood agar base containing 10% defibrinated rabbit blood with a liquid phase of 1% glucose in PBS. Two hamsters were inoculated in the footpad with 0Æ1 ml of each suspension. The cultures and hamsters were observed weekly for the presence of promastigotes and the appearance and persistence of infiltrated lesions, respectively. Identification of Leishmania isolates Identification was based on isoenzymatic and molecular approaches. The isoenzymatic technique was based on starchgel electrophoresis using 13 isoenzyme systems, according to Rioux et al. 13 The 13 enzyme systems investigated were malic enzyme (ME; EC ), isocitrate dehydrogenase (ICD; EC ), 6-phosphogluconate dehydrogenase (PGD; EC ), glucose-6-phosphate dehydrogenase (G6PD; EC ), diaphorase NADH (DIA; EC ), nucleoside purine phosphorylases 1 (NP 1 ; EC ) and 2 (NP 2 ;EC *), glutamate-oxaloacetate transaminases 1 (GOT 1 ; EC ) and 2 (GOT 2 ; EC ), phosphoglucomutase (PGM; EC ), fumarate hydratase (FH; EC ), mannose-phosphate isomerase (MPI; EC ) and glucosephosphate isomerase (GPI; EC ). The molecular approach was based on the sequencing of both strands of a 567-bp region of the RNA polymerase II large subunit gene using primers RP720-D 5 -AAGTACCAGCAGTCCCTCATC-3 and RPOII-FOR 5 -GCAGCCGCACAATGCGCT-3. Sequences were compared with a personal data bank of various Leishmania species. 13 Results The 16 patients comprised 13 males and three females, with a mean age of 27 years. Their disease duration ranged from 1Æ5 to 30 years. All of the patients had received multiple cycles of treatment with pentavalent antimonials, immunotherapy and, in two cases, amphotericin B, with limited improvement and subsequent relapse. The length of treatment with miltefosine was variable, as shown in Table 1. Leishmania amazonensis was identified in 11 cultures, L. mexicana in two, and three were not definitively identified. Before beginning therapy with miltefosine, 13 patients presented papules, plaques and erythematous nodules, symmetrically distributed over the entire body surface (Fig. 1). Three patients presented erythematous plaques asymmetrically located on the face, arms and legs. The leishmanin reaction was negative in all of the patients and the histological sections showed a vacuolated macrophagic granuloma invaded by Table 1 Miltefosine in the treatment of diffuse cutaneous leishmaniasis, first cycle of treatment Patient Age (years) Weight (kg) Daily dose (mg) Day of suspension of treatment Molecular and isoenzymatic identification of Leishmania, MON L. amazonensis, MON L. amazonensis, MON L. amazonensis, MON L. amazonensis, MON L. amazonensis, MON L. amazonensis, MON /50 84 L. mexicana, MON L. amazonensis (contaminated culture of the strain) L. mexicana, MON L. amazonensis, MON L. amazonensis (contaminated culture of the strain) L. amazonensis, MON L. amazonensis, MON days; 190 L. amazonensis, MON 41 50/100 a L. mexicana (contaminated culture of the strain) L. amazonensis, MON 41 a Patient received 50 mg daily for 40 days, then 50 or 100 mg on alternating days.
4 4 Miltefosine in cutaneous leishmaniasis, O. Zerpa et al. Fig 1. Patient 5 on day 0 (left) and during remission after treatment with miltefosine (right). Note prominent nodular lesions on the ears, cheeks and knees and legs before treatment, flattened and essentially without scarring after treatment. numerous lymphoid and plasma cells, with numerous Leishmania amastigotes in the macrophages (Fig. 2). Evolution We developed a clinical scale to evaluate clinical improvement, including elements of inflammation, extension or reduction of affected areas, development of new lesions, and formation of scar tissue. Clinical improvement was estimated to be 30% in 13 patients and 50 65% in three patients after 15 days of treatment. At day 30, 15 patients presented improvement >60%, and one continued with about 30% improvement. At day 45, 15 patients showed 80 90% improvement and one continued to show modest improvement. By day 75, 10 patients showed 100% improvement and in five, improvement was estimated at 80 95% (Table 2). In histological sections, at day 15 we observed evidence of macrophage differentiation, reduced cellularity and fewer parasites. By day 30, the granulomas showed very few parasites (Fig. 3). The granulomas disappeared in the majority of the patients by day 75 and the only observation was nonspecific perivascular inflammatory infiltration. In skin smears, parasites were observed until day 30 in six patients, until day 45 in seven and until day 60 in two. One remained positive until day 190, when treatment was suspended in the absence of a detectable therapeutic response. Growth of parasites in culture medium was observed in 100% of the patients before beginning treatment. Fifteen days after beginning treatment with miltefosine, only three cases gave positive growth in culture medium. Inoculation in hamsters was positive in the 16 patients before therapy was begun and remained positive until day 45 in four. Biopsy material of six patients produced lesions until day 60, one case until day 70, one until day 120 and one until day 190, when treatment was suspended (Table 2). Side-effects were observed in four patients: two complained of nausea and vomiting and reduced the dose without consultation, with improvement in symptoms. One patient presented dizziness on the second day of treatment. On day 20 one patient presented urticaria, that improved with systemic antihistamines. None presented alteration of laboratory parameters during the study. One patient, a 15-year-old boy, died during the course of this study with a clinical diagnosis of pneumonia. He had no haematological, hepatic or renal alterations
5 Miltefosine in cutaneous leishmaniasis, O. Zerpa et al Fig 2. Histological sections before initiation of treatment with miltefosine.the granuloma is composed primarily of cells with large vacuoles containing enormous numbers of amastigotes. Haematoxylin and eosin; original magnification 100 and 400. Table 2 Miltefosine in the treatment of diffuse cutaneous leishmaniasis, first cycle of treatment Patient 100% clinical improvement (day) Parasites detected in smears and biopsies (day) Positive inoculation in hamsters (day) Day of relapse after treatment Reported side-effects None None None None None a Nausea, vomiting Nausea, vomiting Nausea, vomiting None None No, 1 year None No response, 14 b None Patient lost None 14 Only 50% No improvement None improvement c None Nausea, vomiting a New lesions appeared on day 64 of the second cycle of treatment. b Did not respond to a second cycle of treatment. c New lesions during treatment, first cycle. when examined 15 days earlier and exhaustive review of this case has not shown any relationship of this death to DCL pathology nor to treatment with miltefosine. Treatment was suspended in the first nine patients at 3 weeks after the disappearance of lesions and parasitological negativity in smears and histological sections. Nevertheless, new lesions appeared between 30 and 120 days after the suspension of treatment; therefore it was decided to continue treatment for a longer period, 6 months, in the other seven. Twelve patients presented relapse after the first cycle, including those who received 6 months of treatment. A second cycle was administered and five did not respond, with persisting clinical lesions of DCL and presence of parasites in skin smears (Table 3). The period of administration in each cycle was variable; new cycles of treatment were initiated when relapse was detected. Five patients have received a third cycle, with appearance of new lesions when treatment was suspended (Table 4).
6 6 Miltefosine in cutaneous leishmaniasis, O. Zerpa et al. Table 4 Treatment of diffuse cutaneous leishmaniasis with miltefosine, third cycle Patient Age (years) Weight (kg) Daily dose Day of suspension of treatment Reported side-effects None 2 a None None Vomiting None 100 a Patient 2 died during the course of the third cycle of treatment, apparently from pneumonia (under investigation). He had been evaluated by us 15 days before his death and no haematological, hepatic or renal alterations were present. In biopsies obtained from these relapsed patients we observed the presence of granulomas formed by vacuolated macrophages containing numerous parasites. Therefore the second group received additional treatment for 6 months. These patients also presented new lesions 30 days after finishing treatment. The leishmanin reaction remained negative in 15 patients after treatment was terminated; a single patient who has not relapsed had a leishmanin reaction of 13 mm at 1 year after treatment was completed. Discussion 400 Fig 3. Histological sections at 30 days after initiation of treatment with miltefosine. The granulomatous structure is largely resolved and parasites cannot be identified. Haematoxylin and eosin; original magnification 100 and 400. Table 3 Treatment of diffuse cutaneous leishmaniasis with miltefosine, second cycle Patient Age (years) Weight (kg) Daily dose Day of suspension of treatment Reported side-effects None / Nausea None Nausea None None /50 97 Vomiting Nausea, vomiting None None None DCL is a rare form of presentation of leishmaniasis characterized by multiple nodules or nonulcerated plaques that may progress and may eventually cover much of the body surface. This disease manifestation is associated with the absence of manifestations of cell-mediated immunity to the Leishmania parasite. 1 The anti-leishmania treatments used against DCL reduce the initial high parasite burden, but the immune response to the causal agent is not modified, and persisting parasites produce new lesions. In this study we investigated the use of miltefosine in the treatment of DCL because of the success reported in vitro against Leishmania, 14 as well as in therapy of VL in India and of localized cutaneous leishmaniasis (LCL). 11,15 The failure of healing observed in our patients and reported frequently in the literature using conventional anti-leishmania drugs also seemed to justify the trial with miltefosine. The patients treated with miltefosine showed dramatic clinical improvement beginning 15 days after treatment. Nevertheless, complete disappearance of lesions was only observed beginning on day 60, and it was decided to prolong the treatment time in these patients. This observation is in marked contrast with the report of Soto et al. 15 in LCL treated with miltefosine, in which clinical improvement was reported at 4 weeks of treatment. This difference probably reflects the marked difference in the granulomatous structures and components in the two forms of the disease. Cell-mediated hyper-
7 Miltefosine in cutaneous leishmaniasis, O. Zerpa et al. 7 sensitivity, with important cellular components contributing to LCL lesions, would not be expected to respond significantly to miltefosine, which may partially explain the slower response in LCL. In our experience the complete clinical improvement of patients with DCL treated with pentavalent antimonials is only observed after four cycles of treatment (cycles of 20 days with periods of 10 days of rest, in doses recommended by the World Health Organization). Nevertheless, these patients inevitably relapse after chemotherapy. The dose of miltefosine used in this study was 2 2Æ5 mgkg )1 daily. Two patients took lower doses after 15 days because of side-effects. In these patients the clinical improvement was slower and more discrete. This observation coincides with that of Soto et al. in LCL, where the group that received 150 mg daily showed a higher percentage of cure than those who received lower doses. 11 Similar findings were reported in VL, where the group of patients treated with 100 mg daily for 4 weeks presented cure rates of 97% and the group that received 50 mg daily for 6 weeks had a cure rate of 93%. 10 These observations suggest the importance of the use of precise doses in treatment schemes, to avoid therapeutic failures and possible development of resistance to the drug. The reduction in the parasitic burden was related to clinical improvement beginning on day 15 in skin smears and tissue sections. Nevertheless, the more sensitive method of animal inoculation showed the persistence of parasites until day 120 in a patient who showed clinical improvement of 100% at day 75 and disappearance of parasites in smears and histological sections at day 45. This observation clearly demonstrates the persistence of live parasites despite dramatic clinical and parasitological improvement. Histopathologically, the initial granulomas formed by parasite-filled vacuolated macrophages began to show important changes beginning from day 15, with important macrophage differentiation and decrease of the parasite burden. By day 75 the granulomas had completely disappeared, demonstrating the energetic antiparasitic action of this drug. In contrast to what we have observed with other anti-leishmania drugs such as pentavalent antimonials and amphotericin B, in which severe side-effects are reported varying from local manifestations to alterations in cardiovascular, hepatic and renal function, severe side-effects requiring the suspension of therapy were not observed with miltefosine despite the prolonged period of treatment. Laboratory values remained within normal limits during the entire study. The leishmanin reaction remained negative after terminating the treatment, suggesting that the use of miltefosine and profound reduction in the parasite burden was not accompanied by an alteration in the immunological response of the host to Leishmania. This same observation has been reported subsequent to the use of other therapies in DCL. 4 As reported with other drugs used in the treatment of DCL, the appearance of new lesions was observed after terminating treatment with miltefosine in patients treated for days and in those treated for as long as 180 days. Some patients developed lesions during the course of treatment, which clearly suggests that parasite resistance may have developed during this prolonged treatment. The absence of changes in the immune response and the persistence of parasites demonstrated in the inoculation in experimental animals clearly suggest the probability of relapse, and not re-infection, as the cause of new lesions. In this study, miltefosine showed dramatic clinical and parasitological effects during the period of administration of the drug, with few side-effects. The quality of life of these patients was vastly improved and they were able to develop a normal routine of study or work. Nevertheless, apparent development of parasite resistance in some patients and other ethical considerations did not permit indefinite sustained treatment; relapse after suspension of treatment represented a very difficult situation for patients and attending physicians. The absence of immunological reactivity to Leishmania in these patients and those treated with other protocols clearly suggests that treatment of this very challenging pathology must be oriented toward a more intensive search for drugs, immunotherapy or even more complex genetic approaches to correct the profound specific immunological deficiency associated with DCL. Acknowledgments We are grateful to Dr Juergen Engel and Herbert Sindermann from Zentaris Laboratories for their kind donation of the miltefosine for this protocol and to Dr Jean Pierre Dedet, Laboratoire de Parasitologie, Centre National de Référence des Leishmania and WHO Collaborating Centre on Leishmaniasis, CHU de Montpellier and Université Montpellier 1, France, for his collaboration in the identification of the Leishmania strains. We would also like to express our gratitude to Mrs Maria Eugenia Gallinoto for establishing contact with Zentaris Laboratories, as well as to all the staff of the Leishmaniasis Section of our Institute for their continuous help in the development of this protocol. References 1 Convit J, Ulrich M. Antigen-specific immunodeficiency and its relation to the spectrum of American cutaneous leishmaniasis. Biol Res 1993; 26: Convit J, Lapenta P. Sobre un caso de leishmaniasis tegumentaria de forma diseminada. Rev Policlin Caracas 1948; 17: Franke ED, Lucas CM, Tovar AA et al. Diffuse cutaneous leishmaniasis acquired in Peru. Am J Trop Med Hyg 1990; 43: Convit J, Ulrich M, Fernández CT et al. The clinical and immunological spectrum of American cutaneous leishmaniasis. Trans R Soc Trop Med Hyg 1993; 87: Rondón A. Leishmaniasis tegumentaria americana clínica. Dermat Venez 1993; 31 (Suppl. 2): Tapia FJ, Cáceres-Dittmar G, Sanchez MA et al. Adhesion molecules in lesions of American cutaneous leishmaniasis. Exp Dermatol 1994; 3: Castés M, Tapia FJ. Inmunopatologia de la leishmaniasis tegumentaria americana. Acta Cient Venez 1998; 49: Ulrich M. La respuesta humoral en leishmaniasis americana. Dermat Venez 1993; 31 (Suppl. 2):31 3.
8 8 Miltefosine in cutaneous leishmaniasis, O. Zerpa et al. 9 Ulrich M, Rodríguez V, Centeno M, Convit J. Differing antibody IgG isotypes in the polar forms of leprosy and cutaneous leishmaniasis characterized by antigen-specific T cell anergy. Clin Exp Immunol 1995; 100: Jha TK, Shyam S, Thakur CP et al. Miltefosine, an oral agent for treatment of Indian visceral leishmaniasis. N Engl J Med 2002; 347: Soto J, Arana BA, Toledo J et al. Miltefosine for New World cutaneous leishmaniasis. Clin Infect Dis 2004; 38: Zerpa O, Blanco B, Kannee C et al. Treatment of diffuse cutaneous leishmaniasis with miltefosine: a case report. Int J Dermatol 2006; 45: Rioux JA, Lanotte G, Serres E et al. Taxonomy of Leishmania. Use of isoenzymes. Suggestions for a new classification. Ann Parasitol Hum Comp 1990; 65: Lux H, Heise N, Klenner T et al. Ether-lipid (alkyl-phospholipid) metabolism and the mechanism of action of etherlipid and analogues in Leishmania. Mol Biochem Parasitol 2000; 111: Soto J, Toledo J, Gutiérrez P et al. Treatment of American cutaneous leishmaniasis with miltefosine, an oral agent. Clin Infect Dis 2001; 33: Gilman AG, Rall TW, Nies AS et al. (eds). Goodman and Gilman s The Pharmacological Basis of Therapeutics, 9th edn. New York, NY: Pergamon Press, Berman JD. Treatment of New World cutaneous and mucosal leishmaniasis. Clin Dermatol 1996; 14: Veiga JPR, Wolff ER, Sampaio RN, Marsden PD. Renal tubular dysfunction in patients with mucocutaneous leishmaniasis treated with pentavalent antimonials. Lancet 1983; ii:569.
Oral miltefosine for Indian post-kala-azar dermal leishmaniasis: a randomised trial
 Tropical Medicine and International Health doi:10.1111/tmi.12015 volume 18 no 1 pp 96 100 january 2013 Oral miltefosine for Indian post-kala-azar dermal leishmaniasis: a randomised trial Shyam Sundar 1,
Tropical Medicine and International Health doi:10.1111/tmi.12015 volume 18 no 1 pp 96 100 january 2013 Oral miltefosine for Indian post-kala-azar dermal leishmaniasis: a randomised trial Shyam Sundar 1,
Impavido. (miltefosine) New Product Slideshow
 Impavido (miltefosine) New Product Slideshow Introduction Brand name: Impavido Generic name: Miltefosine Pharmacological class: Antileishmanial agent Strength and Formulation: 50mg; hard gel capsules Manufacturer:
Impavido (miltefosine) New Product Slideshow Introduction Brand name: Impavido Generic name: Miltefosine Pharmacological class: Antileishmanial agent Strength and Formulation: 50mg; hard gel capsules Manufacturer:
Welcome to the Jungle! Dr Aileen Oon, 2017 Microbiology Registrar
 Welcome to the Jungle! Dr Aileen Oon, 2017 Microbiology Registrar AA 55M presented with sores on left olecranon and umbilical area Umbilical sores present for 3 weeks Left olecranon lesions for 1 week
Welcome to the Jungle! Dr Aileen Oon, 2017 Microbiology Registrar AA 55M presented with sores on left olecranon and umbilical area Umbilical sores present for 3 weeks Left olecranon lesions for 1 week
The Most Common Parasitic Infections In Yemen. Medical Parasitology
 The Most Common Parasitic Infections In Yemen Medical Parasitology ﻓﺎﯾز اﻟﺧوﻻﻧﻲ / د 2 : is a vector-borne disease that transmitted by sandflies and caused by obligate intracellular protozoa of the genus
The Most Common Parasitic Infections In Yemen Medical Parasitology ﻓﺎﯾز اﻟﺧوﻻﻧﻲ / د 2 : is a vector-borne disease that transmitted by sandflies and caused by obligate intracellular protozoa of the genus
Treatment of Cutaneous Leishmaniasis with Allopurinol and Stibogluconate
 165 Treatment of Cutaneous Leishmaniasis with Allopurinol and S. Martinez, M. Gonzalez, and M. E. Vernaza From the Department of Internal Medicine, University of Cauca, Popayan, and the University Hospital
165 Treatment of Cutaneous Leishmaniasis with Allopurinol and S. Martinez, M. Gonzalez, and M. E. Vernaza From the Department of Internal Medicine, University of Cauca, Popayan, and the University Hospital
Short-Course Paromomycin Treatment of Visceral Leishmaniasis in India: 14-Day vs 21-Day Treatment
 MAJOR ARTICLE Short-Course Paromomycin Treatment of Visceral Leishmaniasis in India: 14-Day vs 21-Day Treatment Shyam Sundar, Neha Agrawal, Rakesh Arora, Dipti Agarwal, Madhukar Rai, and Jaya Chakravarty
MAJOR ARTICLE Short-Course Paromomycin Treatment of Visceral Leishmaniasis in India: 14-Day vs 21-Day Treatment Shyam Sundar, Neha Agrawal, Rakesh Arora, Dipti Agarwal, Madhukar Rai, and Jaya Chakravarty
Sodium Stibogluconate treatment for cutaneous leishmaniasis: A clinical study of 43 cases from the north of Jordan
 Sodium Stibogluconate treatment for cutaneous leishmaniasis: A clinical study of 43 cases from the north of Jordan Mamoun Mohammad Al-Athamneh Hiathem Qasem Abu Al-haija Ra ed Smadi Ayman S. Qaqaa Heba
Sodium Stibogluconate treatment for cutaneous leishmaniasis: A clinical study of 43 cases from the north of Jordan Mamoun Mohammad Al-Athamneh Hiathem Qasem Abu Al-haija Ra ed Smadi Ayman S. Qaqaa Heba
Laboratory diagnosis of Blood and tissue flagellates
 Laboratory diagnosis of Blood and tissue flagellates (Leishmania and trypanosma) Sarah Alharbi Clinical Laboratory department Collage of Applied Medical Sciences King Saud University Leishmania and trypanosma:
Laboratory diagnosis of Blood and tissue flagellates (Leishmania and trypanosma) Sarah Alharbi Clinical Laboratory department Collage of Applied Medical Sciences King Saud University Leishmania and trypanosma:
New insights on leishmaniasis in immunosuppressive conditions
 New insights on leishmaniasis in immunosuppressive conditions Javier Moreno Immunoparasitology Unit WHO Collaborative Center for Leishmaniasis Centro Nacional de Microbiología INSTITUTO DE SALUD CARLOS
New insights on leishmaniasis in immunosuppressive conditions Javier Moreno Immunoparasitology Unit WHO Collaborative Center for Leishmaniasis Centro Nacional de Microbiología INSTITUTO DE SALUD CARLOS
Efficacy of Miltefosine in the Treatment of Visceral Leishmaniasis in India After a Decade of Use
 MAJOR ARTICLE Efficacy of Miltefosine in the Treatment of Visceral Leishmaniasis in India After a Decade of Use Shyam Sundar, 1,a Anup Singh, 1,a Madhukar Rai, 1 Vijay K. Prajapati, 1 Avinash K. Singh,
MAJOR ARTICLE Efficacy of Miltefosine in the Treatment of Visceral Leishmaniasis in India After a Decade of Use Shyam Sundar, 1,a Anup Singh, 1,a Madhukar Rai, 1 Vijay K. Prajapati, 1 Avinash K. Singh,
Miltefosine for New World Cutaneous Leishmaniasis
 MAJOR ARTICLE for New World Cutaneous sis J. Soto, 1 B. A. Arana, 4 J. Toledo, 1 N. Rizzo, 4 J. C. Vega, 2 A. Diaz, 4 M. Luz, 1 P. Gutierrez, 1 M. Arboleda, 3 J. D. Berman, 6,a K. Junge, 5 J. Engel, 5
MAJOR ARTICLE for New World Cutaneous sis J. Soto, 1 B. A. Arana, 4 J. Toledo, 1 N. Rizzo, 4 J. C. Vega, 2 A. Diaz, 4 M. Luz, 1 P. Gutierrez, 1 M. Arboleda, 3 J. D. Berman, 6,a K. Junge, 5 J. Engel, 5
~Trichinella Spiralis:
 Musculoskeletal System **Today we are going to talk about the parasites that affect the musculoskeletal system ~Trichinella Spiralis: It s a small nematode that measures to about 2-3mm in length. In general
Musculoskeletal System **Today we are going to talk about the parasites that affect the musculoskeletal system ~Trichinella Spiralis: It s a small nematode that measures to about 2-3mm in length. In general
Review Cutaneous Manifestations of Human and Murine Leishmaniasis
 Review Cutaneous Manifestations of Human and Murine Leishmaniasis Breanna M. Scorza 1, Edgar M. Carvalho 2,3 and Mary E. Wilson 1,4,5,6, * 1 Interdisciplinary Graduate Program in Immunology, University
Review Cutaneous Manifestations of Human and Murine Leishmaniasis Breanna M. Scorza 1, Edgar M. Carvalho 2,3 and Mary E. Wilson 1,4,5,6, * 1 Interdisciplinary Graduate Program in Immunology, University
Cutaneous leishmaniasis: advances in disease pathogenesis, diagnostics and therapeutics
 Clinical dermatology Review article CED Clinical and Experimental Dermatology Cutaneous leishmaniasis: advances in disease pathogenesis, diagnostics and therapeutics M. Ameen Royal Free Hospital, Royal
Clinical dermatology Review article CED Clinical and Experimental Dermatology Cutaneous leishmaniasis: advances in disease pathogenesis, diagnostics and therapeutics M. Ameen Royal Free Hospital, Royal
HAEMOFLAGELLATES. Dr. Anuluck Junkum Department of Parasitology Faculty of Medicine
 HAEMOFLAGELLATES Dr. Anuluck Junkum Department of Parasitology Faculty of Medicine Objective Can describe the morphology, life cycle, pathology, diagnosis and prevention of Leishmania spp. and Trypanosoma
HAEMOFLAGELLATES Dr. Anuluck Junkum Department of Parasitology Faculty of Medicine Objective Can describe the morphology, life cycle, pathology, diagnosis and prevention of Leishmania spp. and Trypanosoma
Cutaneous Leishmaniasis in a Chinese man returning from the Amazon forest in Brazil
 Hong Kong J. Dermatol. Venereol. (2013) 21, 79-83 Case Report Cutaneous Leishmaniasis in a Chinese man returning from the Amazon forest in Brazil JTHT Yu and SY Wong A 60-year-old man presented with ulcerated
Hong Kong J. Dermatol. Venereol. (2013) 21, 79-83 Case Report Cutaneous Leishmaniasis in a Chinese man returning from the Amazon forest in Brazil JTHT Yu and SY Wong A 60-year-old man presented with ulcerated
Original Article Comparison of fine needle aspiration with biopsy in the diagnosis of cutaneous leishmaniasis Ikramullah Khan, Rifat Yasmin
 Original Article Comparison of fine needle aspiration with biopsy in the diagnosis of cutaneous leishmaniasis Ikramullah Khan, Rifat Yasmin Department of Dermatology, Pakistan Institute of Medical Sciences,
Original Article Comparison of fine needle aspiration with biopsy in the diagnosis of cutaneous leishmaniasis Ikramullah Khan, Rifat Yasmin Department of Dermatology, Pakistan Institute of Medical Sciences,
Leishmaniasis: Challenges for Vaccine Development
 Leishmaniasis: Challenges for Vaccine Development Steven G. Reed Infectious Disease Research Institute Seattle IDRI Goals: Leishmaniasis To improve existing vaccines for use in therapy and prevention.
Leishmaniasis: Challenges for Vaccine Development Steven G. Reed Infectious Disease Research Institute Seattle IDRI Goals: Leishmaniasis To improve existing vaccines for use in therapy and prevention.
Leishmaniasis, Kala Azar(The Black Fever)
 Leishmaniasis, Kala Azar(The Black Fever) By Lawrence Hall Etiologic agent Protist obligate intracellular parasite, Transmission Vectors Phylum: Euglenozoa (genus Leishmania) Over 21 species that infect
Leishmaniasis, Kala Azar(The Black Fever) By Lawrence Hall Etiologic agent Protist obligate intracellular parasite, Transmission Vectors Phylum: Euglenozoa (genus Leishmania) Over 21 species that infect
Single-Dose Liposomal Amphotericin B in the Treatment of Visceral Leishmaniasis in India: A Multicenter Study
 MAJOR ARTICLE Single-Dose Liposomal Amphotericin B in the Treatment of Visceral Leishmaniasis in India: A Multicenter Study S. Sundar, 1 T. K. Jha, 2 C. P. Thakur, 3 M. Mishra, 4 V. P. Singh, 1 and R.
MAJOR ARTICLE Single-Dose Liposomal Amphotericin B in the Treatment of Visceral Leishmaniasis in India: A Multicenter Study S. Sundar, 1 T. K. Jha, 2 C. P. Thakur, 3 M. Mishra, 4 V. P. Singh, 1 and R.
The New England Journal of Medicine
 The New England Journal of Medicine Copyright 22 by the Massachusetts Medical Society VOLUME 347 N OVEMBER 28, 22 NUMBER 22 ORAL MILTEFOSINE FOR INDIAN VISCERAL LEISHMANIASIS SHYAM SUNDAR, M.D., T.K. JHA,
The New England Journal of Medicine Copyright 22 by the Massachusetts Medical Society VOLUME 347 N OVEMBER 28, 22 NUMBER 22 ORAL MILTEFOSINE FOR INDIAN VISCERAL LEISHMANIASIS SHYAM SUNDAR, M.D., T.K. JHA,
Leishmaniasis. By Joseph Knight, PA-C. 2. Explain the differences in the reasons leishmaniasis is spreading in Afghanistan and India.
 Leishmaniasis By Joseph Knight, PA-C Objectives 1. Identify the two types of leishmaniasis. 2. Explain the differences in the reasons leishmaniasis is spreading in Afghanistan and India. 3. Discuss how
Leishmaniasis By Joseph Knight, PA-C Objectives 1. Identify the two types of leishmaniasis. 2. Explain the differences in the reasons leishmaniasis is spreading in Afghanistan and India. 3. Discuss how
Post Kala-azar Dermal Leishmaniasis (PKDL) from the field to the cellular and the subcellular levels
 Post Kala-azar Dermal Leishmaniasis (PKDL) from the field to the cellular and the subcellular levels A M EL Hassan Institute of Endemic Diseases University of Khartoum Introduction PKDL is a VL related
Post Kala-azar Dermal Leishmaniasis (PKDL) from the field to the cellular and the subcellular levels A M EL Hassan Institute of Endemic Diseases University of Khartoum Introduction PKDL is a VL related
Research Article Enhancement of a T H 1 Immune Response in Amphotericin B-Treated Mucocutaneous Leishmaniasis
 Biomedicine and Biotechnology Volume 27, Article ID 9641, 4 pages doi:1.1155/27/9641 Research Article Enhancement of a T H 1 Immune Response in Amphotericin B-Treated Mucocutaneous Leishmaniasis Washington
Biomedicine and Biotechnology Volume 27, Article ID 9641, 4 pages doi:1.1155/27/9641 Research Article Enhancement of a T H 1 Immune Response in Amphotericin B-Treated Mucocutaneous Leishmaniasis Washington
BIO Parasitology Spring 2009
 BIO 475 - Parasitology Spring 2009 Stephen M. Shuster Northern Arizona University http://www4.nau.edu/isopod Lecture 5 Discovery of the Disease In 1924 the Kala-Azar Commission noted that the distribution
BIO 475 - Parasitology Spring 2009 Stephen M. Shuster Northern Arizona University http://www4.nau.edu/isopod Lecture 5 Discovery of the Disease In 1924 the Kala-Azar Commission noted that the distribution
Cutaneous leishmaniasis in an overseas Filipino worker who responded favorably to oral itraconazole
 CSE REPORT Cutaneous leishmaniasis in an overseas Filipino worker who responded favorably to oral itraconazole udi, MD 1, ilza ellamy R. Limcangco, MD, FPDS 2, Johannes F, Dayrit, MD, FPDS 2,3 Introduction:
CSE REPORT Cutaneous leishmaniasis in an overseas Filipino worker who responded favorably to oral itraconazole udi, MD 1, ilza ellamy R. Limcangco, MD, FPDS 2, Johannes F, Dayrit, MD, FPDS 2,3 Introduction:
Research Article Efficacy and Safety of Paromomycin in Treatment of Post-Kala-Azar Dermal Leishmaniasis
 ISRN Parasitology, Article ID 548010, 4 pages http://dx.doi.org/10.1155/2014/548010 Research Article Efficacy and Safety of Paromomycin in Treatment of Post-Kala-Azar Dermal Leishmaniasis Shyam Sundar,
ISRN Parasitology, Article ID 548010, 4 pages http://dx.doi.org/10.1155/2014/548010 Research Article Efficacy and Safety of Paromomycin in Treatment of Post-Kala-Azar Dermal Leishmaniasis Shyam Sundar,
TOWARD AN APPROACH FOR CUTANEOUS LEISHMANIA TREATMENT. Mohammed Wael Daboul
 Original Articles DOI: 10.7241/ourd.20131.09 Source of Support: Nil Competing Interests: None TOWARD AN APPROACH FOR CUTANEOUS LEISHMANIA TREATMENT Mohammed Wael Daboul Laboratory Medicine Specialist,
Original Articles DOI: 10.7241/ourd.20131.09 Source of Support: Nil Competing Interests: None TOWARD AN APPROACH FOR CUTANEOUS LEISHMANIA TREATMENT Mohammed Wael Daboul Laboratory Medicine Specialist,
Series Editors Samuel J. Black, University of Massachusetts, Amherst, MA, US.A. J. Richard Seed, University of North Carolina, Chapel Hill, NC, US.A.
 LEISHMANIA World Class Parasites VOLUME 4 Volumes in the World Class Parasites book series are written for researchers, students and scholars who enjoy reading about excellent research on problems of global
LEISHMANIA World Class Parasites VOLUME 4 Volumes in the World Class Parasites book series are written for researchers, students and scholars who enjoy reading about excellent research on problems of global
Case Report Development of Cutaneous Leishmaniasis after Leishmania Skin Test
 Case Reports in Medicine Volume 2011, Article ID 631079, 4 pages doi:10.1155/2011/631079 Case Report Development of Cutaneous Leishmaniasis after Leishmania Skin Test Paulo R. Machado, 1, 2 Augusto M.
Case Reports in Medicine Volume 2011, Article ID 631079, 4 pages doi:10.1155/2011/631079 Case Report Development of Cutaneous Leishmaniasis after Leishmania Skin Test Paulo R. Machado, 1, 2 Augusto M.
International Conference on Parasitology August 24-26, 2015 Philadelphia, Pennsylvania, USA
 International Conference on Parasitology August 24-26, 2015 Philadelphia, Pennsylvania, USA SYMPOSIA THE CHALLENGE OF PARASITES AND IMMUNOSUPRESSION: FROM DIAGNOSIS TO TREATMENT from the bench to the bed
International Conference on Parasitology August 24-26, 2015 Philadelphia, Pennsylvania, USA SYMPOSIA THE CHALLENGE OF PARASITES AND IMMUNOSUPRESSION: FROM DIAGNOSIS TO TREATMENT from the bench to the bed
SUMMARY. Cutaneous leishmaniasis with only skin involvement: single to multiple skin ulcers, satellite lesions and nodular lymphangitis.
 SUMMARY Leishmaniasis is a disease affecting predominantly people in the developing countries; 350 million people worldwide are at risk and yearly more than 2 million new cases occur. Leishmaniasis is
SUMMARY Leishmaniasis is a disease affecting predominantly people in the developing countries; 350 million people worldwide are at risk and yearly more than 2 million new cases occur. Leishmaniasis is
Blood Smears Only 6 October Sample Preparation and Quality Control 15B-K
 NEW YORK STATE Parasitology Proficiency Testing Program Blood Smears Only 6 October 5 The purpose of the New York State Proficiency Testing Program in the category of Parasitology - Blood Smears Only is
NEW YORK STATE Parasitology Proficiency Testing Program Blood Smears Only 6 October 5 The purpose of the New York State Proficiency Testing Program in the category of Parasitology - Blood Smears Only is
Leishmaniasis. CDR R.L. Gutierrez Oct 2014
 Leishmaniasis CDR R.L. Gutierrez Oct 2014 Overview Protozoan parasite(s) of tissue and WBCs Many species / Many Syndromes (Cutaneous / Visceral) Pathogen: Location - Old World vs. New World Host: Immune
Leishmaniasis CDR R.L. Gutierrez Oct 2014 Overview Protozoan parasite(s) of tissue and WBCs Many species / Many Syndromes (Cutaneous / Visceral) Pathogen: Location - Old World vs. New World Host: Immune
Antimonial Resistance & Combination therapy in Indian Visceral Leishmaniasis. Shyam Sundar Banaras Hindu University Varanasi, India
 Antimonial Resistance & Combination therapy in Indian Visceral Leishmaniasis Shyam Sundar Banaras Hindu University Varanasi, India History of the Current Epidemic in India & Antimony Resistance 6 Official
Antimonial Resistance & Combination therapy in Indian Visceral Leishmaniasis Shyam Sundar Banaras Hindu University Varanasi, India History of the Current Epidemic in India & Antimony Resistance 6 Official
Acta Dermatovenerol Croat 2008;16(2):60-64 CLINICAL ARTICLE
 2008;16(2):60-64 CLINICAL ARTICLE Clinical Efficacy of Intramuscular Meglumine Antimoniate Alone and in Combination with Intralesional Meglumine Antimoniate in the Treatment of Old World Cutaneous Leishmaniasis
2008;16(2):60-64 CLINICAL ARTICLE Clinical Efficacy of Intramuscular Meglumine Antimoniate Alone and in Combination with Intralesional Meglumine Antimoniate in the Treatment of Old World Cutaneous Leishmaniasis
POST KALA-AZAR DERMAL LEISHMANIASIS WITH ULCERATION ON FOOT: AN ATYPICAL CASE PRESENTATION SUCCESSFULLY TREATED WITH MILTEFOSINE
 American Journal of Infectious Diseases 10 (2): 50-55, 2014 ISSN: 1553-6203 2014 Science Publication doi:10.3844/ajidsp.2014.50.55 Published Online 10 (2) 2014 (http://www.thescipub.com/ajid.toc) POST
American Journal of Infectious Diseases 10 (2): 50-55, 2014 ISSN: 1553-6203 2014 Science Publication doi:10.3844/ajidsp.2014.50.55 Published Online 10 (2) 2014 (http://www.thescipub.com/ajid.toc) POST
Development of Mathematical models predicting the density of vectors: Case of sandflies vectors of leishmaniasis
 Development of Mathematical models predicting the density of vectors: Case of sandflies vectors of leishmaniasis HABIBA MEJHED Département Génie Informatique, Ecole Nationale des sciences appliquées Université
Development of Mathematical models predicting the density of vectors: Case of sandflies vectors of leishmaniasis HABIBA MEJHED Département Génie Informatique, Ecole Nationale des sciences appliquées Université
Cutaneous Leishmaniasis : Global overview
 Cutaneous Leishmaniasis : Global overview Meeting of stakeholders for selected Health R&D Demostration Projects, 7-10 May 2014, WHO, Geneva Dr. Daniel Argaw Dagne, NTD/WHO CSR - DDC AFRO Leishmaniasis
Cutaneous Leishmaniasis : Global overview Meeting of stakeholders for selected Health R&D Demostration Projects, 7-10 May 2014, WHO, Geneva Dr. Daniel Argaw Dagne, NTD/WHO CSR - DDC AFRO Leishmaniasis
Animal models for infectious diseases caused by parasites: Leishmaniasis
 Drug Discovery Today: Disease Models Vol. 1, No. 1 2004 DRUG DISCOVERY TODAY DISEASE MODELS Editors-in-Chief Jan Tornell AstraZeneca, Sweden Denis Noble University of Oxford, UK Infectious diseases Animal
Drug Discovery Today: Disease Models Vol. 1, No. 1 2004 DRUG DISCOVERY TODAY DISEASE MODELS Editors-in-Chief Jan Tornell AstraZeneca, Sweden Denis Noble University of Oxford, UK Infectious diseases Animal
Treatment of Disseminated Leishmaniasis With Liposomal Amphotericin B
 MAJOR ARTICLE Treatment of Disseminated Leishmaniasis With Liposomal Amphotericin B Paulo R. L. Machado, 1,2 Maria Elisa A. Rosa, 1 Luís H. Guimarães, 1,2 Fernanda V. O. Prates, 1 Adriano Queiroz, 1,2
MAJOR ARTICLE Treatment of Disseminated Leishmaniasis With Liposomal Amphotericin B Paulo R. L. Machado, 1,2 Maria Elisa A. Rosa, 1 Luís H. Guimarães, 1,2 Fernanda V. O. Prates, 1 Adriano Queiroz, 1,2
What is Kala-azar? What are Signs & Symptoms of Kala-Azar?
 What is Kala-azar? Kala-azar is a slow progressing indigenous disease caused by a protozoan parasite of genus Leishmania In India Leishmania donovani is the only parasite causing this disease The parasite
What is Kala-azar? Kala-azar is a slow progressing indigenous disease caused by a protozoan parasite of genus Leishmania In India Leishmania donovani is the only parasite causing this disease The parasite
Departments of Dermatology, Venereology and Leprosy and *Microbiology, Indira Gandhi Medical College, Shimla, India.
 Net Study Comparative efþ cacy of intralesional sodium stibogluconate (SSG) alone and its combination with intramuscular SSG to treat localized cutaneous leishmaniasis: Results of a pilot study Ajeet K.
Net Study Comparative efþ cacy of intralesional sodium stibogluconate (SSG) alone and its combination with intramuscular SSG to treat localized cutaneous leishmaniasis: Results of a pilot study Ajeet K.
EVALUATION OF MILTEFOSINE AGAINST LEISHMANIA MAJOR (MRHO/IR/75/ER): IN VITRO AND IN VIVO STUDIES
 ORIGINAL REPORT EVALUATION OF MILTEFOSINE AGAINST LEISHMANIA MAJOR (MRHO/IR/75/ER): IN VITRO AND IN VIVO STUDIES J. Esmaeili 1, M. Mohebali 1*, G. H. Edrissian 1, S. M. Rezayat 2, M. Ghazi-Khansari 2 and
ORIGINAL REPORT EVALUATION OF MILTEFOSINE AGAINST LEISHMANIA MAJOR (MRHO/IR/75/ER): IN VITRO AND IN VIVO STUDIES J. Esmaeili 1, M. Mohebali 1*, G. H. Edrissian 1, S. M. Rezayat 2, M. Ghazi-Khansari 2 and
Leishmania donovani is the agent of cutaneous leishmaniasis in Sri Lanka
 1 Leishmania donovani is the agent of cutaneous leishmaniasis in Sri Lanka HVYD Siriwardana,* HA Noyes, NJ Beeching,* ML Chance,* ND Karunaweera, and PA Bates* *Liverpool School of Tropical Medicine, Liverpool,
1 Leishmania donovani is the agent of cutaneous leishmaniasis in Sri Lanka HVYD Siriwardana,* HA Noyes, NJ Beeching,* ML Chance,* ND Karunaweera, and PA Bates* *Liverpool School of Tropical Medicine, Liverpool,
CLINCOPATHOLOGICAL CASE
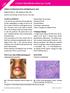 CLINCOPATHOLOGICAL CASE Solitary erythematous firm noduloplaque on nose Hassab El-Naby H., MD, Mosbeh Al Sadat, MD Department of Dermatology, Al-Azhar University, Cairo, Egypt. CLINICAL FINDINGS A 36-year-old
CLINCOPATHOLOGICAL CASE Solitary erythematous firm noduloplaque on nose Hassab El-Naby H., MD, Mosbeh Al Sadat, MD Department of Dermatology, Al-Azhar University, Cairo, Egypt. CLINICAL FINDINGS A 36-year-old
Laboratory investigation of Cutaneous Leishmaniasis in Karachi
 Laboratory investigation of Cutaneous Leishmaniasis in Karachi Pages with reference to book, From 248 To 250 G.M. Rajpar, M.A. Khan, A. Hafiz ( Department of Microbiology, Jinnah Postgraduate Medical Centre,
Laboratory investigation of Cutaneous Leishmaniasis in Karachi Pages with reference to book, From 248 To 250 G.M. Rajpar, M.A. Khan, A. Hafiz ( Department of Microbiology, Jinnah Postgraduate Medical Centre,
Immunology Lecture 4. Clinical Relevance of the Immune System
 Immunology Lecture 4 The Well Patient: How innate and adaptive immune responses maintain health - 13, pg 169-181, 191-195. Immune Deficiency - 15 Autoimmunity - 16 Transplantation - 17, pg 260-270 Tumor
Immunology Lecture 4 The Well Patient: How innate and adaptive immune responses maintain health - 13, pg 169-181, 191-195. Immune Deficiency - 15 Autoimmunity - 16 Transplantation - 17, pg 260-270 Tumor
Original Article Chloroquine in cutaneous leishmaniasis
 Original Article Chloroquine in cutaneous leishmaniasis Ikramullah Khan, Rifat Yasmin, Iqbal Sidiqui Department of Dermatology, Pakistan Institute of Medical Sciences, Islamabad Abstract Background Chloroquine
Original Article Chloroquine in cutaneous leishmaniasis Ikramullah Khan, Rifat Yasmin, Iqbal Sidiqui Department of Dermatology, Pakistan Institute of Medical Sciences, Islamabad Abstract Background Chloroquine
Visceral leishmaniasis: an endemic disease with global impact
 Visceral leishmaniasis: an endemic disease with global impact Professor Olivier Lortholary, MD, PhD Department of Infectious and Tropical diseases Hôpital Necker-Enfants Malades Université Paris Descartes
Visceral leishmaniasis: an endemic disease with global impact Professor Olivier Lortholary, MD, PhD Department of Infectious and Tropical diseases Hôpital Necker-Enfants Malades Université Paris Descartes
Leishmaniasis: A forgotten disease among neglected people
 ISPUB.COM The Internet Journal of Health Volume 11 Number 2 Leishmaniasis: A forgotten disease among neglected people Y Homsi, G Makdisi Citation Y Homsi, G Makdisi. Leishmaniasis: A forgotten disease
ISPUB.COM The Internet Journal of Health Volume 11 Number 2 Leishmaniasis: A forgotten disease among neglected people Y Homsi, G Makdisi Citation Y Homsi, G Makdisi. Leishmaniasis: A forgotten disease
Chapter 24 The Immune System
 Chapter 24 The Immune System The Immune System Layered defense system The skin and chemical barriers The innate and adaptive immune systems Immunity The body s ability to recognize and destroy specific
Chapter 24 The Immune System The Immune System Layered defense system The skin and chemical barriers The innate and adaptive immune systems Immunity The body s ability to recognize and destroy specific
COURSE: Medical Microbiology, PAMB 650/720 - Fall 2008 Lecture 16
 COURSE: Medical Microbiology, PAMB 650/720 - Fall 2008 Lecture 16 Tumor Immunology M. Nagarkatti Teaching Objectives: Introduction to Cancer Immunology Know the antigens expressed by cancer cells Understand
COURSE: Medical Microbiology, PAMB 650/720 - Fall 2008 Lecture 16 Tumor Immunology M. Nagarkatti Teaching Objectives: Introduction to Cancer Immunology Know the antigens expressed by cancer cells Understand
2.Trichomonas vaginalis
 2.Trichomonas vaginalis 1. Pathogenic to human &causes vaginitis (trichomoniasis). 2. troph. Is round or pear like in shape, contains 4-6 flagella, all originating from anterior end & only one extend posteriorly.
2.Trichomonas vaginalis 1. Pathogenic to human &causes vaginitis (trichomoniasis). 2. troph. Is round or pear like in shape, contains 4-6 flagella, all originating from anterior end & only one extend posteriorly.
CHRONIC INFLAMMATION
 CHRONIC INFLAMMATION Chronic inflammation is an inflammatory response of prolonged duration often for months, years or even indefinitely. Its prolonged course is proved by persistence of the causative
CHRONIC INFLAMMATION Chronic inflammation is an inflammatory response of prolonged duration often for months, years or even indefinitely. Its prolonged course is proved by persistence of the causative
A histopathological study of cutaneous leishmaniasis in Sri Lanka
 A histopathological study of cutaneous leishmaniasis in Sri Lanka C H P Herath 1, N V I Ratnatunga 2, R Waduge 3, P Ratnayake 3, C N Ratnatunga 4, S Ramadasa 2 (Index words: cutaneous leishmaniasis, histopathology,
A histopathological study of cutaneous leishmaniasis in Sri Lanka C H P Herath 1, N V I Ratnatunga 2, R Waduge 3, P Ratnayake 3, C N Ratnatunga 4, S Ramadasa 2 (Index words: cutaneous leishmaniasis, histopathology,
DENDRITIC CELLS ACTIVATED BY CpG MOTIFS ARE POTENT INDUCERS OF A TH1 IMMUNE RESPONSE THAT PROTECTS MICE AGAINST LEISHMANIASIS
 DENDRITIC CELLS ACTIVATED BY CpG MOTIFS ARE POTENT INDUCERS OF A TH1 IMMUNE RESPONSE THAT PROTECTS MICE AGAINST LEISHMANIASIS Dissertation zur Erlangung des naturwissenschaftlichen Doktorgrades der Bayerischen
DENDRITIC CELLS ACTIVATED BY CpG MOTIFS ARE POTENT INDUCERS OF A TH1 IMMUNE RESPONSE THAT PROTECTS MICE AGAINST LEISHMANIASIS Dissertation zur Erlangung des naturwissenschaftlichen Doktorgrades der Bayerischen
Erythema gyratumrepens-like eruption in a patient with epidermolysisbullosaacquisita associated with ulcerative colitis
 Erythema gyratumrepens-like eruption in a patient with epidermolysisbullosaacquisita associated with ulcerative colitis A. España C. Sitaru* M. Pretel L. Aguado J. Jimenez# Department of Dermatology, University
Erythema gyratumrepens-like eruption in a patient with epidermolysisbullosaacquisita associated with ulcerative colitis A. España C. Sitaru* M. Pretel L. Aguado J. Jimenez# Department of Dermatology, University
Nanosilver in the treatment of localized cutaneous leishmaniasis caused by Leishmania major (MRHO/IR/75/ER): an in vitro and in vivo study
 DARU Vol. 17, No. 4 2009 285 Nanosilver in the treatment of localized cutaneous leishmaniasis caused by Leishmania major (MRHO/IR/75/ER): an in vitro and in vivo study *1 Mohebali M., 2 Rezayat M.M., 3
DARU Vol. 17, No. 4 2009 285 Nanosilver in the treatment of localized cutaneous leishmaniasis caused by Leishmania major (MRHO/IR/75/ER): an in vitro and in vivo study *1 Mohebali M., 2 Rezayat M.M., 3
GENUS: LEISHMANIA. Under the genus Leishmania, there are 2 subgenus: SPECIES PARASITIC IN MEN. Under subgenus Leishmania, there are following species:
 GENUS: LEISHMANIA Species parasitic in man: Under the genus Leishmania, there are 2 subgenus: 1. Leishmania 2. Viannia SPECIES PARASITIC IN MEN Under subgenus Leishmania, there are following species: LEISHMANIA
GENUS: LEISHMANIA Species parasitic in man: Under the genus Leishmania, there are 2 subgenus: 1. Leishmania 2. Viannia SPECIES PARASITIC IN MEN Under subgenus Leishmania, there are following species: LEISHMANIA
Necrotizing and suppurative lymphadenitis in Leishmania
 Tropical Medicine and International Health VOLUME I NO. 2 PP 243-250 APRIL 1996 Necrotizing and suppurative lymphadenitis in Leishmania major infections A. Gaafar r, A. Ismai13, A. Y. El Kadaro4, Elsir
Tropical Medicine and International Health VOLUME I NO. 2 PP 243-250 APRIL 1996 Necrotizing and suppurative lymphadenitis in Leishmania major infections A. Gaafar r, A. Ismai13, A. Y. El Kadaro4, Elsir
Leishmaniasis WRAIR- GEIS 'Operational Clinical Infectious Disease' Course
 Leishmaniasis WRAIR- GEIS 'Operational Clinical Infectious Disease' Course UNCLASSIFIED Acknowledgments LTC James E. Moon, MD Chief, Sleep Trials Branch Walter Reed Army Institute of Research CDR Ramiro
Leishmaniasis WRAIR- GEIS 'Operational Clinical Infectious Disease' Course UNCLASSIFIED Acknowledgments LTC James E. Moon, MD Chief, Sleep Trials Branch Walter Reed Army Institute of Research CDR Ramiro
CONTROLE DAS LEISHMANIOSES O QUE FALTA FAZER? Centro de Convenções de Reboças Red Room 17: 00h
 CONTROLE DAS LEISHMANIOSES O QUE FALTA FAZER? Centro de Convenções de Reboças 08.04.2014 Red Room 17: 00h Leishmaniasis - a Global Problem Visceral 2012 300 000 cases 20,000 deaths (6.7%) 310 million at
CONTROLE DAS LEISHMANIOSES O QUE FALTA FAZER? Centro de Convenções de Reboças 08.04.2014 Red Room 17: 00h Leishmaniasis - a Global Problem Visceral 2012 300 000 cases 20,000 deaths (6.7%) 310 million at
VISERAL LEISHMANIASI S (KALA-AZAR)
 VISERAL LEISHMANIASI S (KALA-AZAR) :OUTLINES DEFINITION. EPIDEMIOLOGY. PARASITE & VECTOR. PATHOLOGY CLINICAL & LIFE CYCLE. PICTURE. COMPLICATIONS. DIAGNOSIS. INVESTIGATIONS. MANAGEMENT TREATMENT S CONTROL.
VISERAL LEISHMANIASI S (KALA-AZAR) :OUTLINES DEFINITION. EPIDEMIOLOGY. PARASITE & VECTOR. PATHOLOGY CLINICAL & LIFE CYCLE. PICTURE. COMPLICATIONS. DIAGNOSIS. INVESTIGATIONS. MANAGEMENT TREATMENT S CONTROL.
Blood Smears Only 07 February Sample Preparation and Quality Control 12B A
 NEW YORK STATE Parasitology Proficiency Testing Program Blood Smears Only 07 February 2012 The purpose of the New York State Proficiency Testing Program in the category of Parasitology Blood Smears Only
NEW YORK STATE Parasitology Proficiency Testing Program Blood Smears Only 07 February 2012 The purpose of the New York State Proficiency Testing Program in the category of Parasitology Blood Smears Only
University Journal of Medicine and Medical Specialities
 University Journal of Medicine and Medical Specialities Volume 1 Issue 1 2015 ATYPICAL PRESENTATION OF LEPROMATOUS LEPROSY LOCALISED TO BILATERAL FLANKS ANANDAN V, RAJESH HN Stanley Medical College Abstract:
University Journal of Medicine and Medical Specialities Volume 1 Issue 1 2015 ATYPICAL PRESENTATION OF LEPROMATOUS LEPROSY LOCALISED TO BILATERAL FLANKS ANANDAN V, RAJESH HN Stanley Medical College Abstract:
SOD1 Plasma Level as a Biomarker for Therapeutic Failure in Cutaneous Leishmaniasis
 BRIEF REPORT SOD1 Plasma Level as a Biomarker for Therapeutic Failure in Cutaneous Leishmaniasis Ricardo Khouri, 1,2,4 Gilvaneia Silva Santos, 1 George Soares, 1 Jackson M. Costa, 1 Aldina Barral, 1,2,3
BRIEF REPORT SOD1 Plasma Level as a Biomarker for Therapeutic Failure in Cutaneous Leishmaniasis Ricardo Khouri, 1,2,4 Gilvaneia Silva Santos, 1 George Soares, 1 Jackson M. Costa, 1 Aldina Barral, 1,2,3
WMLN Case Study. Necrotizing Palate Biopsy Specimen. Youngmi Kim Sr. Microbiologist TB Lab & Parasitology Lab MS, M(ASCP)
 WMLN Case Study Necrotizing Palate Biopsy Specimen Youngmi Kim Sr. Microbiologist TB Lab & Parasitology Lab MS, M(ASCP) 44 year old male from Honduras Immigrated to US 12 years ago to WI 4 years ago Seen
WMLN Case Study Necrotizing Palate Biopsy Specimen Youngmi Kim Sr. Microbiologist TB Lab & Parasitology Lab MS, M(ASCP) 44 year old male from Honduras Immigrated to US 12 years ago to WI 4 years ago Seen
199 (M. A. Bioproducts, Walkersville, Md.). placed on 22-mm2 cover slips and incubated at 37 C in
 INFECTION AND IMMUNITY, June 1981, p. 1249-1253 0019-9567/81/061249-05$02.00/0 Vol. 32, No. 3 Interaction of Leishmania donovani Promastigotes with Human Monocyte-Derived Macrophages: Parasite Entry, Intracellular
INFECTION AND IMMUNITY, June 1981, p. 1249-1253 0019-9567/81/061249-05$02.00/0 Vol. 32, No. 3 Interaction of Leishmania donovani Promastigotes with Human Monocyte-Derived Macrophages: Parasite Entry, Intracellular
PDF of Trial CTRI Website URL -
 Clinical Trial Details (PDF Generation Date :- Fri, 04 Jan 2019 20:47:10 GMT) CTRI Number Last Modified On 25/04/2017 Post Graduate Thesis Type of Trial Type of Study Study Design Public Title of Study
Clinical Trial Details (PDF Generation Date :- Fri, 04 Jan 2019 20:47:10 GMT) CTRI Number Last Modified On 25/04/2017 Post Graduate Thesis Type of Trial Type of Study Study Design Public Title of Study
Cutaneous Leishmaniasis in an Immigrant Saudi Worker: A Case Report
 J HEALTH POPUL NUTR 2014 Jun;32(2):372-376 ISSN 1606-0997 $ 5.00+0.20 INTERNATIONAL CENTRE FOR DIARRHOEAL DISEASE RESEARCH, BANGLADESH CASE STUDY Cutaneous Leishmaniasis in an Immigrant Saudi Worker: A
J HEALTH POPUL NUTR 2014 Jun;32(2):372-376 ISSN 1606-0997 $ 5.00+0.20 INTERNATIONAL CENTRE FOR DIARRHOEAL DISEASE RESEARCH, BANGLADESH CASE STUDY Cutaneous Leishmaniasis in an Immigrant Saudi Worker: A
Immunological Aspects of Parasitic Diseases in Immunocompromised Individuals. Taniawati Supali. Department of Parasitology
 Immunological Aspects of Parasitic Diseases in Immunocompromised Individuals Taniawati Supali Department of Parasitology 1 Defense mechanism in human Th17 (? ) Acute Chronic Th1 Th 2 Intracellular Treg
Immunological Aspects of Parasitic Diseases in Immunocompromised Individuals Taniawati Supali Department of Parasitology 1 Defense mechanism in human Th17 (? ) Acute Chronic Th1 Th 2 Intracellular Treg
Leishmaniasis. MAJ Kris Paolino September 2014
 Leishmaniasis MAJ Kris Paolino September 2014 Thanks to COL (Ret) Kent Kester MAJ Leyi Lin http://www.niaid.nih.gov/topics/leishmaniasis History Sir William Boog Leishman (1865-1926) Matriculated at the
Leishmaniasis MAJ Kris Paolino September 2014 Thanks to COL (Ret) Kent Kester MAJ Leyi Lin http://www.niaid.nih.gov/topics/leishmaniasis History Sir William Boog Leishman (1865-1926) Matriculated at the
Egyptian Dermatology Online Journal Vol. 6 No 1: 14, June 2010
 Wells Syndrome H. Gammaz, H. Amer, A. Adly and S. Mahmoud Egyptian Dermatology Online Journal 6 (1): 14 Al-Haud Al-Marsoud Hospital, Cairo, Egypt e-mail: hananderma@hotmail.com Submitted: April 15, 2010
Wells Syndrome H. Gammaz, H. Amer, A. Adly and S. Mahmoud Egyptian Dermatology Online Journal 6 (1): 14 Al-Haud Al-Marsoud Hospital, Cairo, Egypt e-mail: hananderma@hotmail.com Submitted: April 15, 2010
Intralesional meglumine antimoniate for the treatment of localised cutaneous leishmaniasis: a retrospective review of a Brazilian referral centre
 512 Mem Inst Oswaldo Cruz, Rio de Janeiro, Vol. 111(8): 512-516, August 2016 Intralesional meglumine antimoniate for the treatment of localised cutaneous leishmaniasis: a retrospective review of a Brazilian
512 Mem Inst Oswaldo Cruz, Rio de Janeiro, Vol. 111(8): 512-516, August 2016 Intralesional meglumine antimoniate for the treatment of localised cutaneous leishmaniasis: a retrospective review of a Brazilian
A Novel Noninvasive Method for Diagnosis of Visceral Leishmaniasis by. rk39 Test in Sputum Samples
 JCM Accepts, published online ahead of print on 0 June 00 J. Clin. Microbiol. doi:0./jcm.00-0 Copyright 00, American Society for Microbiology and/or the Listed Authors/Institutions. All Rights Reserved.
JCM Accepts, published online ahead of print on 0 June 00 J. Clin. Microbiol. doi:0./jcm.00-0 Copyright 00, American Society for Microbiology and/or the Listed Authors/Institutions. All Rights Reserved.
Received 13 June 2010/Returned for modification 30 July 2010/Accepted 12 August 2010
 ANTIMICROBIAL AGENTS AND CHEMOTHERAPY, Nov. 2010, p. 4699 4704 Vol. 54, No. 11 0066-4804/10/$12.00 doi:10.1128/aac.00809-10 Copyright 2010, American Society for Microbiology. All Rights Reserved. Reductions
ANTIMICROBIAL AGENTS AND CHEMOTHERAPY, Nov. 2010, p. 4699 4704 Vol. 54, No. 11 0066-4804/10/$12.00 doi:10.1128/aac.00809-10 Copyright 2010, American Society for Microbiology. All Rights Reserved. Reductions
Tumor Immunology. Tumor (latin) = swelling
 Tumor Immunology Tumor (latin) = swelling benign tumor malignant tumor Tumor immunology : the study of the types of antigens that are expressed by tumors how the immune system recognizes and responds to
Tumor Immunology Tumor (latin) = swelling benign tumor malignant tumor Tumor immunology : the study of the types of antigens that are expressed by tumors how the immune system recognizes and responds to
New Insights into Diagnosing Leishmaniasis
 New Insights into Diagnosing Leishmaniasis Eric Zini Snow meeting, 13 March 2009 Climate Variability and Visceral Leishmaniasis in Europe WHO/TDR, Jan. 2008 Late Eighties Maroli et al., Trop Med Int Health
New Insights into Diagnosing Leishmaniasis Eric Zini Snow meeting, 13 March 2009 Climate Variability and Visceral Leishmaniasis in Europe WHO/TDR, Jan. 2008 Late Eighties Maroli et al., Trop Med Int Health
1. Specificity: specific activity for each type of pathogens. Immunity is directed against a particular pathogen or foreign substance.
 L13: Acquired or adaptive (specific) immunity The resistance, which absent at the time of first exposure to a pathogen, but develops after being exposed to the pathogen is called acquired immunity. It
L13: Acquired or adaptive (specific) immunity The resistance, which absent at the time of first exposure to a pathogen, but develops after being exposed to the pathogen is called acquired immunity. It
Visceral Leishmaniasis combination therapies
 Best Science for the Most Neglected From patient needs to implementation of new treatments Visceral Leishmaniasis combination therapies Shyam Sundar Institute of Medical Sciences Banaras Hindu University
Best Science for the Most Neglected From patient needs to implementation of new treatments Visceral Leishmaniasis combination therapies Shyam Sundar Institute of Medical Sciences Banaras Hindu University
SMe. Me NITROIMIDAZOLES FOR VISCERAL LEISHMANIASIS FEXINIDAZOLE AND VL-2098 SHYAM SUNDAR
 SMe O 2 CH 2 O Me ITROIMIDAZOLES FOR VISCERAL LEISHMAIASIS FEXIIDAZOLE AD VL-2098 SHYAM SUDAR Target Product Profile for a CE Optimal Target Profile Target Label VL and PKDL VL Spp All species L. donavani
SMe O 2 CH 2 O Me ITROIMIDAZOLES FOR VISCERAL LEISHMAIASIS FEXIIDAZOLE AD VL-2098 SHYAM SUDAR Target Product Profile for a CE Optimal Target Profile Target Label VL and PKDL VL Spp All species L. donavani
Chapter 23 Immunity Exam Study Questions
 Chapter 23 Immunity Exam Study Questions 1. Define 1) Immunity 2) Neutrophils 3) Macrophage 4) Epitopes 5) Interferon 6) Complement system 7) Histamine 8) Mast cells 9) Antigen 10) Antigens receptors 11)
Chapter 23 Immunity Exam Study Questions 1. Define 1) Immunity 2) Neutrophils 3) Macrophage 4) Epitopes 5) Interferon 6) Complement system 7) Histamine 8) Mast cells 9) Antigen 10) Antigens receptors 11)
Question 1. Kupffer cells, microglial cells and osteoclasts are all examples of what type of immune system cell?
 Abbas Chapter 2: Sarah Spriet February 8, 2015 Question 1. Kupffer cells, microglial cells and osteoclasts are all examples of what type of immune system cell? a. Dendritic cells b. Macrophages c. Monocytes
Abbas Chapter 2: Sarah Spriet February 8, 2015 Question 1. Kupffer cells, microglial cells and osteoclasts are all examples of what type of immune system cell? a. Dendritic cells b. Macrophages c. Monocytes
Disseminated cutaneous BCG infection following BCG immunotherapy in patients with lepromatous leprosy
 Lepr Rev (2015) 86, 180 185 CASE REPORT Disseminated cutaneous BCG infection following BCG immunotherapy in patients with lepromatous leprosy GEETI KHULLAR*, TARUN NARANG*, KUSUM SHARMA**, UMA NAHAR SAIKIA***
Lepr Rev (2015) 86, 180 185 CASE REPORT Disseminated cutaneous BCG infection following BCG immunotherapy in patients with lepromatous leprosy GEETI KHULLAR*, TARUN NARANG*, KUSUM SHARMA**, UMA NAHAR SAIKIA***
Recommendations for Coping with Leishmaniasis: A Review of Control Strategies. Centro de Convenções de Reboças Red Room 17: 00h
 Recommendations for Coping with Leishmaniasis: A Review of Control Strategies Centro de Convenções de Reboças 08.04.2014 Red Room 17: 00h Leishmaniasis - a Global Problem Visceral 2012 300 000 cases 20,000
Recommendations for Coping with Leishmaniasis: A Review of Control Strategies Centro de Convenções de Reboças 08.04.2014 Red Room 17: 00h Leishmaniasis - a Global Problem Visceral 2012 300 000 cases 20,000
Scenario of Histoid Hansen at a Tertiary Care Hospital in South India
 Indian J Lepr 2018, 90 : 235-240 Hind Kusht Nivaran Sangh, New Delhi http://www.ijl.org.in Case Series Scenario of Histoid Hansen at a Tertiary Care Hospital in South India 1 2 3 R Subha, M Ananthi, V
Indian J Lepr 2018, 90 : 235-240 Hind Kusht Nivaran Sangh, New Delhi http://www.ijl.org.in Case Series Scenario of Histoid Hansen at a Tertiary Care Hospital in South India 1 2 3 R Subha, M Ananthi, V
Cutaneous Leishmania
 DISCLOSURE OF RELEVANT RELATIONSHIPS WITH INDUSTRY Cutaneous Leishmania Ibrahim Khalifeh, MD I do not have any relevant relationships with industry. Cutaneous Leishmania: Is it a Legend? Ibrahim Khalifeh,
DISCLOSURE OF RELEVANT RELATIONSHIPS WITH INDUSTRY Cutaneous Leishmania Ibrahim Khalifeh, MD I do not have any relevant relationships with industry. Cutaneous Leishmania: Is it a Legend? Ibrahim Khalifeh,
Summary of Cases & Epidemiology Aspects of Leishmaniasis in Thailand
 Summary of Cases & Epidemiology Aspects of Leishmaniasis in Thailand Sukmee T. 1, Mungthin M. 2, Apiwathanasorn C. 3, Leelayoova S. 2 1 Department of Microbiology, Phramongkutklao College of Medicine 2
Summary of Cases & Epidemiology Aspects of Leishmaniasis in Thailand Sukmee T. 1, Mungthin M. 2, Apiwathanasorn C. 3, Leelayoova S. 2 1 Department of Microbiology, Phramongkutklao College of Medicine 2
Spectrum of clinical presentations
 Spectrum of clinical presentations Case History A 7-day-old male patient born full-term via uncomplicated vaginal delivery was seen for multiple erythematous red-brown purpuric lesions that were present
Spectrum of clinical presentations Case History A 7-day-old male patient born full-term via uncomplicated vaginal delivery was seen for multiple erythematous red-brown purpuric lesions that were present
Medical Bacteriology- Lecture 10. Mycobacterium. Actinomycetes. Nocardia
 Medical Bacteriology- Lecture 10 Mycobacterium Actinomycetes Nocardia 1 Mycobacterium Characteristics - Large, very weakly gram positive rods - Obligate aerobes, related to Actinomycetes - Catalase positive
Medical Bacteriology- Lecture 10 Mycobacterium Actinomycetes Nocardia 1 Mycobacterium Characteristics - Large, very weakly gram positive rods - Obligate aerobes, related to Actinomycetes - Catalase positive
Shigella Pathogenesis and Vaccine Development
 Shigella Pathogenesis and Vaccine Development Ryan Ranallo, Ph.D. Department of Enteric Infections Division of Communicable Diseases and Immunology Walter Reed Army Institute of Research Causes of Travelers
Shigella Pathogenesis and Vaccine Development Ryan Ranallo, Ph.D. Department of Enteric Infections Division of Communicable Diseases and Immunology Walter Reed Army Institute of Research Causes of Travelers
H erpes simplex virus infection of the
 Herpes simplex keratitis An experimental study Samuel J. Kimura, Victor Diaz-Bonnet, and Masao Okumoto The incidence of complicated herpes simplex keratitis appears to have increased and the important
Herpes simplex keratitis An experimental study Samuel J. Kimura, Victor Diaz-Bonnet, and Masao Okumoto The incidence of complicated herpes simplex keratitis appears to have increased and the important
Leishmaniaand Leishmaniasis
 Leishmaniaand Leishmaniasis Methodenseminar SS2014 IRMA SCHABUSSOVA, PhD Institute of Specific Prophylaxis and Tropical Medicine; Medical University Vienna Kinderspitalgasse 15, A-1090 Vienna, Austria
Leishmaniaand Leishmaniasis Methodenseminar SS2014 IRMA SCHABUSSOVA, PhD Institute of Specific Prophylaxis and Tropical Medicine; Medical University Vienna Kinderspitalgasse 15, A-1090 Vienna, Austria
Clinical and immunopathological aspects of disseminated cutaneous leishmaniasis
 Acta Tropica, 56( 1994)315-325 0 1994 Elsevier Science B.V. All rights reserved 0001-706X/94/$07.00 315 ACTROP 00364 Clinical and immunopathological aspects of disseminated cutaneous leishmaniasis E.M.
Acta Tropica, 56( 1994)315-325 0 1994 Elsevier Science B.V. All rights reserved 0001-706X/94/$07.00 315 ACTROP 00364 Clinical and immunopathological aspects of disseminated cutaneous leishmaniasis E.M.
العصوي الوعاي ي الورام = angiomatosis Bacillary
 1 / 7 BACILLARY ANGIOMATOSIS Epidemiology BA is most commonly seen in patients with acquired immunodeficiency syndrome (AIDS) and a CD4 count less than 50 cells/mm 3, with an incidence of 1.2 cases per
1 / 7 BACILLARY ANGIOMATOSIS Epidemiology BA is most commonly seen in patients with acquired immunodeficiency syndrome (AIDS) and a CD4 count less than 50 cells/mm 3, with an incidence of 1.2 cases per
Therapeutic Options for Visceral Leishmaniasis
 Drugs (2013) 73:1863 1888 DOI 10.1007/s40265-013-0133-0 REVIEW ARTICLE Therapeutic Options for Visceral Leishmaniasis Begoña Monge-Maillo Rogelio López-Vélez Published online: 30 October 2013 Ó The Author(s)
Drugs (2013) 73:1863 1888 DOI 10.1007/s40265-013-0133-0 REVIEW ARTICLE Therapeutic Options for Visceral Leishmaniasis Begoña Monge-Maillo Rogelio López-Vélez Published online: 30 October 2013 Ó The Author(s)
Post-kala-azar Dermal Leishmaniasis Associated With AIDS*
 BJID 2003; 7 (June) 229 Post-kala-azar Dermal Leishmaniasis Associated With AIDS* Achiléa Bittencourt, Nancy Silva, Andréa Straatmann, Victor Luiz Correia Nunes, Ivonise Follador and Roberto Badaró Universitary
BJID 2003; 7 (June) 229 Post-kala-azar Dermal Leishmaniasis Associated With AIDS* Achiléa Bittencourt, Nancy Silva, Andréa Straatmann, Victor Luiz Correia Nunes, Ivonise Follador and Roberto Badaró Universitary
Cell-Mediated Immune Response in Indian Kala Azar and
 INFECTION AND IMMUNITY, Nov. 1983, p. 702-707 Vol. 42, No. 2 0019-9567/83/110702-06$02.00/0 Copyright 1983, American Society for Microbiology Cell-Mediated Immune Response in Indian Kala Azar and Post-Kala
INFECTION AND IMMUNITY, Nov. 1983, p. 702-707 Vol. 42, No. 2 0019-9567/83/110702-06$02.00/0 Copyright 1983, American Society for Microbiology Cell-Mediated Immune Response in Indian Kala Azar and Post-Kala
Degos Disease: A Case Report and Review of Literature
 Degos Disease: A Case Report and Review of Literature Monira waked Egyptian Dermatology Online Journal 4 (1): 5, June 2008 Al Houd Al Marsod Hospital Submitted for publication: May 25 th, 2008 Accepted
Degos Disease: A Case Report and Review of Literature Monira waked Egyptian Dermatology Online Journal 4 (1): 5, June 2008 Al Houd Al Marsod Hospital Submitted for publication: May 25 th, 2008 Accepted
