The neuropeptide galanin is widely distributed within the
|
|
|
- Linda O’Neal’
- 5 years ago
- Views:
Transcription
1 Transgenic overexpression of galanin in the dorsal root ganglia modulates pain-related behavior Fiona E. Holmes*, Andrea Bacon*, Robert J. P. Pope*, Penny A. Vanderplank*, Niall C. H. Kerr*, Madhu Sukumaran, Vassilis Pachnis, and David Wynick* *University Research Centre for Neuroendocrinology, University of Bristol, Marlborough Street, Bristol BS2 8HW, United Kingdom; and Division of Developmental Neurobiology, National Institute for Medical Research, London NW7 1AA, United Kingdom Edited by Tomas Hökfelt, Karolinska Institutet, Stockholm, Sweden, and approved March 10, 2003 (received for review November 21, 2002) The neuropeptide galanin is expressed in the dorsal root ganglia (DRG) and spinal cord and is thought to be involved in the modulation of pain processing. However, its mechanisms of action are complex and poorly understood, as both facilitatory and inhibitory effects have been described. To understand further the role played by galanin in nociception, we have generated two transgenic lines that overexpress galanin in specific populations of primary afferent DRG neurons in either an inducible or constitutive manner. In the first line, a previously defined enhancer region from the galanin locus was used to target galanin to the DRG (Gal-OE). Transgene expression recapitulates the spatial endogenous galanin distribution pattern in DRG neurons and markedly overexpresses the peptide in the DRG after nerve injury but not in the uninjured state. In the second line, an enhancer region of the c-ret gene was used to constitutively and ectopically target galanin overexpression to the DRG (Ret-OE). The expression of this second transgene does not alter significantly after nerve injury. Here, we report that intact Ret-OE, but not Gal-OE, animals have significantly elevated mechanical and thermal thresholds. After nerve damage, using a spared nerve-injury model, mechanical allodynia is attenuated markedly in both the Gal-OE and Ret-OE mice compared with WT controls. These results support an inhibitory role for galanin in the modulation of nociception both in intact animals and in neuropathic pain states. The neuropeptide galanin is widely distributed within the somatosensory system, where it has been implicated in the modulation of nociception (reviewed in refs. 1 3). In the adult dorsal root ganglia (DRG), galanin is expressed at relatively low levels in 5% of neurons that are predominantly small-diameter C fiber type (4). Expression of the peptide also is detected in the primary afferent terminals of the dorsal spinal cord and in subsets of dorsal horn interneurons (5). Galanin mrna and peptide levels markedly increase in the DRG after axotomy and in several neuropathic pain models of sciatic nerve injury (4, 6 9). After axotomy, galanin is expressed in 40 50% of all DRG neurons (4, 10). In the dorsal horn, axotomy-induced galanin immunoreactivity increases in the afferent terminals, with a concurrent enhancement of synaptic release of the peptide (11). The role of galanin in pain signaling is complex (reviewed in refs. 2, 12, and 13). Electrophysiological and behavioral studies in the intact, uninjured peripheral nervous system have demonstrated both facilitatory (14 21) and inhibitory (22, 23) effects of exogenously applied galanin on nociception, sometimes in a modality-specific manner (14, 16, 20, 21), tending toward inhibition at higher doses. A number of groups have used rodent models of neuropathic pain to study the role of galanin in the modulation of chronic pain behavior. The oldest and least understood pain model involves the scoring of autotomy behavior (self-mutilation of the denervated limb) after sciatic nerve axotomy (24). More recently, several neuropathic pain models have been developed in which the sciatic nerve is only partially denervated, including chronic constriction injury (the Bennett model; ref. 25), partial nerve ligation (the Seltzer model; ref. 26), spinal nerve ligation (the Chung model; ref. 27), photochemically induced nerve ischemia (28, 29), and, most recently, spared nerve injury (SNI) (30). Pain behavior in these models is analogous to that observed in a number of human neuropathic pain conditions, including allodynia, hyperalgesia, and expansion of the receptive field. Electrophysiological experiments on axotomized (23, 31), chronic constriction injury (32), and spinal nerve ligation (15) animals have shown that intrathecal administration of galanin reduces spinal cord excitability. Furthermore, intrathecal administration of the galanin antagonist M35 (31) significantly potentiates the frequency and severity of axotomy-induced autotomy behavior (33). Similarly, several studies have shown that intrathecal galanin significantly reduces allodynia induced by chronic constriction injury (6, 34, 35). In addition, Eaton et al. (36) have shown that a spinal cord implant of genetically modified cells that secrete galanin significantly reduces allodynia after chronic constriction injury. In summary, the precise role played by galanin in the intact peripheral nervous system remains unclear, with both facilitatory and inhibitory effects described. After nerve injury, when levels of endogenous galanin are high, the general consensus is that the peptide plays a predominantly inhibitory function in spinal cord transmission and acts to reduce neuropathic pain behavior. To study further the role played by galanin in the adaptive response of the nervous system to injury, we previously generated and characterized mice carrying a targeted deletion of the galanin gene (16, 37, 38). Uninjured galanin mutant animals have decreased thermal and mechanical thresholds, supporting the proposed inhibitory role for galanin in nociception. Unexpectedly, the galanin mutant mice have a complete absence of the autotomy behavior that normally is seen in WT animals after axotomy, apparently contradicting the previous findings by Verge et al. in rats when using the M35 galanin antagonist (33). Further, we have shown that these mutants have markedly reduced thermal and mechanical allodynia in the partial nerve ligation model when compared with WT controls (16). Most recently, electrophysiological studies have shown that the development of central sensitization is impaired in the mutants; both windup and facilitation of spinal reflexes after conditioning stimulation are attenuated significantly after peripheral nerve injury (39). These data raise the possibility that galanin may play an excitatory role in states of nerve injury, despite the previous body of evidence to the contrary. Here, we describe two lines of transgenic mice. The first line inducibly overexpresses the peptide in the DRG after nerve injury and the second constitutively and ectopically overexpresses galanin in the DRG and spinal cord. Nociceptive behavior in intact transgenic animals and after a SNI model of This paper was submitted directly (Track II) to the PNAS office. Abbreviations: DRG, dorsal root ganglia; SNI, spared nerve injury; IB4, isolectin-b4. To whom correspondence should be addressed. d.wynick@bris.ac.uk PNAS May 13, 2003 vol. 100 no cgi doi pnas
2 neuropathic pain (30) is described and compared with the galanin mutant mice. These results support an inhibitory role for galanin in the modulation of nociception both in intact animals and in neuropathic pain states. Methods Generation of Mice. The generation of galanin null mutant mice (maintained on the inbred 129OlaHsd strain) has been described (38). The mutation results in the deletion of five of the six exons from the galanin gene, including the entire coding region of the peptide. Transgenic mice (CBA B6 strain) that inducibly overexpress galanin in the DRG after nerve injury were generated by using a transgene corresponding to an 20-kb upstream enhancer region of the murine galanin locus and the 4.6-kb galanin-coding region, as described recently (40). Transgenic mice (CBA B6 strain) that constitutively overexpress galanin in the DRG were generated by using the previously described 12-kb flanking regulatory sequences of the murine c-ret gene (41) to drive expression of the 4.6-kb galanin-coding region, inserted at the unique SacII site. In all experiments, mutant and transgenic animals were compared with WT controls on the identical strain background. Surgery. Age-matched adult mice (10 12 weeks old, g) were used in all experiments (n 8 per genotype). Animals were fed standard chow and water ad libitum. All experiments were carried out in accordance with the United Kingdom Animals (Scientific Procedures) Act Mice were anesthetized with Hypnorm (0.315 mg ml fentanyl citrate 10 mg ml fluanisone; Janssen) Hypnovel (5 mg ml midazolam; Roche Applied Sciences) water at a ratio of 1:1:2 at 4 l g. A modification of the spared nerve injury model, originally described by Decosterd and Woolf (30), was used as a model of neuropathic pain. An incision was made in the lateral right hind leg just above the level of the knee, exposing the three terminal branches of the sciatic nerve: the common peroneal, tibial, and sural nerves. The common peroneal and sural nerves were ligated tightly with 7 0 silk and sectioned distal to the ligation removing 2 mm of distal nerve stump. The tibial branch remained untouched during the procedure. The overlying muscle and skin was sutured, and the animals were allowed to recover. In sham-operated animals, the sciatic nerve branches were exposed but not lesioned. Behavioral Testing. In all tests, the examiner was blind to the genotype of the mice. Thermal thresholds were measured according to the method of Hargreaves et al. (42). Animals were habituated to the testing environment for at least 2 h, and testing was performed on two separate occasions before surgery. The hind paws were exposed to a beam of radiant heat through a transparent perspex holding box (Ugo Basile, Varese, Italy), and the latency of withdrawal was recorded automatically. Each hind paw was measured three times, with an interval of at least 5 min between measurements. Mechanical thresholds were measured with a series of calibrated von Frey filaments (Stoelting) from g to a maximum of 3.63 g. Animals were put in perspex enclosures placed on an elevated grid (Ugo Basile) and habituated for at least 2 h before testing. Mechanical sensitivity was assessed on each hind paw, employing the up-down testing paradigm to determine the threshold force required to elicit a withdrawal response to 50% of stimulations (43, 44). Immunohistochemistry. Mice were intracardially perfused with 4% paraformaldehyde PBS, and the spinal columns were removed and postfixed for 4hatroom temperature. The spinal cord and L4 and L5 DRGs were dissected and equilibrated in 20% sucrose overnight at 4 C, embedded in OCT mounting medium, frozen on dry ice, and cryostat-sectioned (16- M sections for DRG and 30- M sections for spinal cord). Sections were blocked and permeabilized in 10% normal donkey serum PBS 0.2% Triton X-100 (PBST) for 1 h at room temperature. Sections then were incubated in rabbit polyclonal antibody to galanin (Affiniti, Nottingham, U.K.) at 1:1,000 in PBST overnight at room temperature, washed 3 10 min in PBS, and incubated in donkey anti-rabbit cy3 (The Jackson Laboratory) at 1:800 for3hatroom temperature. Where appropriate, sections were washed as before and then incubated in biotin-conjugated isolectin-b4 (IB4) at 10 g ml (Sigma) diluted in PBST 0.1 mm CaCl 2, 0.1 mm MgCl 2, and 0.1 mm MnCl 2, followed by extravidin-fitc at 1:100 (Sigma). After washing, sections were mounted in VECTASHIELD (Vector Laboratories). Images were taken by using a Leica fluorescent microscope with RT Color Spot camera and SPOT ADVANCE image-capture system software (Diagnostic Instruments, Sterling Heights, MI). Statistics. Data are presented as the mean SEM. Student s t test was used to analyze the difference in baseline thermal withdrawal thresholds. ANOVAs and Dunnett s or nonparametric Mann Whitney U post hoc tests were used as appropriate to analyze differences between genotypes and at different time points after SNI. A P value of 0.05 was considered to be significant. Results and Discussion WT Mice. Mice were tested for allodynia-like pain behavior before and at 1, 2, 5, 7, 9, 12, and 14 days after SNI. After SNI, all WT mice on both the CBA B6 and 129OlaHsd genetic backgrounds developed marked mechanical allodynia of the lateral surface of the hind paw ipsilateral to the injured nerve, which occurred within 1 day of surgery. The withdrawal threshold decreased from a mean baseline value of g to g (P 0.001) for the CBA B6 animals (Fig. 1) and g to g (P 0.001) for the 129OlaHsd strain (Fig. 2A). Mechanical allodynia increased further by 2 days post-sni and was maintained for the duration of the experiment (Figs. 1B and 2A). Nonoperated controls and sham-operated animals showed no significant change in withdrawal thresholds over the 14-day testing period (results not shown). The animals also were tested for thermal thresholds at 1, 7, and 14 days after SNI. Thermal latencies did not change appreciably from baseline measurements in the 129OlaHsd WT controls (Fig. 2B) or in the CBA B6 WT animals (data not shown). This is consistent with previously published data acquired by using the rat SNI model (30). Because thermal latencies did not significantly change in mice after SNI, we did not measure this modality in subsequent SNI experiments. We have used an adaptation of the existing rat SNI model recently described by S. D. Shields, W. A. Eckert III, and A. I. Basbaum (unpublished work) to induce neuropathic pain behavior in mice. It has several advantages over other existing models: (i) it is technically straightforward to perform, (ii) the degree and time course of the mechanical allodynia are robust and reproducible, (iii) all animals develop allodynia to a similar extent (particularly important when comparing different genotypes), and (iv) the allodynia develops within 1 day and lasts for at least 4 weeks (data not shown). We also studied the effects of the SNI on galanin expression in the DRG and spinal cord. Groups of WT animals from both strain backgrounds were subjected to SNI, and L4 and L5 DRG and lumbar spinal cord tissue was collected at 0, 3, 5, 7, 14, and 21 days after nerve injury. In control, uninjured animals, galanin was expressed only in a few cells in the DRG, as described (4). The number of galanin-positive neurons and the levels of galanin in the neurons in the DRG increased within 3 days of SNI, which NEUROSCIENCE Holmes et al. PNAS May 13, 2003 vol. 100 no
3 Fig. 1. Responses of WT (CBA B6)- and galanin-overexpressing lines to mechanical stimuli. Presurgery 50% withdrawal thresholds are denoted as day 0. (A) Pre-SNI thresholds were not significantly different between genotypes. After SNI, CBA B6 WT controls developed robust allodynia (Mann Whitney U test; P at day 1). The Gal-OE initially developed allodynia, but thresholds returned to presurgery measurements by day 7 post-sni. *, WT 0 group compared with WT groups at each time point; $, Gal-OE 0 group compared with Gal-OE groups at each time point; &&, comparison between Gal-OE groups at days 5 and 7. ** and *** denote P 0.01 and P 0.001, respectively. ## denotes to P $ and $$ denote P 0.05 and P 0.01, respectively. (B) Pre-SNI thresholds were significantly greater in Ret-OE mice (Mann Whitney U test; P 0.01). After SNI, mechanical withdrawal thresholds were reduced in Ret-OEs, although thresholds remained significantly higher than WT controls for the entire duration of the experiment. *, WT 0 group compared with WT groups at each time point; #, WT compared with Ret-OE at each time point; $, Ret-OE 0 group compared with Ret-OE groups at each time point. * and ** denote P 0.05 and P 0.01, respectively. # and ## denote P 0.05 and P 0.01, respectively. $ and $$ denote P 0.05 and P 0.01, respectively. increased further by 7 days after surgery (Fig. 3), and was maintained for the duration of the experiment (data not shown). Increased galanin immunoreactivity also was seen in the ipsilateral (right side) dorsal horn (Fig. 4). Gal-OE Mice. To obtain an expression cassette to inducibly target various biologically active molecules to the adult DRG, we have used transgenesis to map and characterize the enhancer regions of the murine galanin gene. We have defined a 20-kb transgene that fully recapitulates the endogenous expression pattern of galanin in both the adult intact DRG and in adult DRG and spinal cord after axotomy (A.B., N.C.H.K., F.E.H., and D.W., unpublished work). We now have used this 20-kb galanin enhancer to drive expression of the murine galanin gene in transgenic mice. We have shown recently that these transgenic Fig. 2. Responses of WT (129OlaHsd) and galanin mutant mice to mechanical and thermal stimuli. (A) Presurgery 50% withdrawal thresholds are denoted as day 0. WT animals developed robust allodynia within 1 day of SNI (Mann Whitney U test; P 0.01), and this was maintained for the duration of the experiment. Mutants have significantly lower pre-sni mechanical withdrawal thresholds than WT controls (Mann Whitney U test; P 0.01) and fail to develop allodynia after SNI. (B) Time course of thermal hyperalgesia after SNI. Presurgery withdrawal thresholds are denoted as day 0. Mutants had significantly lower pre-sni thermal withdrawal thresholds than WT controls (Dunnett s P 0.05). There was no significant change in thresholds for either genotype after SNI. *, WT 0 group compared with WT groups at each time point; #, WT compared with knockout at each time point. ** and *** denote P 0.01 and P 0.001, respectively. # and ## denote P 0.05 and P 0.01, respectively. animals have 4-fold-higher levels of galanin in the DRG 1 week after axotomy (measured by RIA) compared with injured WT controls but have normal peptide levels in the intact, uninjured DRG (40). Consistent with this, immunohistochemical staining for galanin in the intact DRG was unchanged, and no significant differences were observed between Gal-OE and matched WT controls in either mechanical or thermal thresholds (Fig. 1A and ref. 40). Somewhat higher levels of galanin staining were observed in the dorsal horn of the intact Gal-OE spinal cord than in the intact WT controls (Fig. 4). These data would imply that there may be increased galanin synthesis in the intact DRG of Gal-OE animals that is transported and stored in the afferent terminals of the dorsal horn. Presumably, the relatively minor increase in galanin expression in the dorsal horn is not sufficient to perturb nociceptive thresholds in intact Gal-OE animals. After SNI, Gal-OE animals developed some degree of me cgi doi pnas Holmes et al.
4 Fig. 3. Time course of immunohistochemical localization of galanin in WT, Gal-OE, and Ret-OE DRG before (time 0) and at 3, 5, and 7 days post-sni. In uninjured WT animals, galanin was expressed in a few neurons in the DRG and was up-regulated after SNI. No differences were noted in DRG staining between uninjured WT and Gal-OE animals. After SNI, DRG galanin immunoreactivity was greater in the Gal-OE than in SNI-injured WT controls at all time points. In the uninjured Ret-OE, galanin was expressed in various neurons. A majority of galaninergic neurons colocalized with IB4, although it is clear that a substantial number of large-diameter neurons that are positive for galanin are negative for IB4 binding (Lower). (Bar 100 M.) chanical allodynia by day 1 after surgery (Fig. 1A) with a withdrawal threshold of g, although this was a significantly greater threshold than the WT controls on the same strain background at this time point (P 0.05). By day 7 post-sni, the withdrawal thresholds returned to presurgery values ( g) and remained at those values for the rest of the experiment (Fig. 1A). Immunohistochemistry for galanin in the DRG ipsilateral to the lesion (right side) of the Gal-OE animals demonstrated that after SNI, galanin expression increased to a maximum, by day 7 after injury (Fig. 3). Stronger neuronal immunoreactivity in the DRG was observed in the SNI-operated Gal-OE animals compared with SNI-operated WT animals at all time points studied (Fig. 3). Galanin expression also was studied in the spinal cord, demonstrating that by day 7 after surgery there is a pronounced up-regulation in the dorsal horn in both WT and Gal-OE animals that is greater in the Gal-OE animals (Fig. 4). The reduction in mechanical thresholds observed in the Gal-OE after SNI may be related to the increasing levels of galanin in the DRG observed between days 3, 5, and 7 (Fig. 3). Further, it is possible that galanin overexpression in other tissues (as yet unknown) such as the autonomic nervous system also may contribute to the observed pain phenotype. These new data further support the hypothesis that galanin indeed does play an inhibitory role in pain processing after nerve injury, compatible with the previous published pharmacological data. Ret-OE Mice. In addition to the Gal-OE mice, we have also generated transgenic animals by using the previously described 12-kb c-ret enhancer region (41) to drive the expression of the murine galanin gene (Ret-OE). This enhancer region recently has been demonstrated to target the IB4-binding neurons in the DRG (41), and expression of the transgene does not alter after axotomy (F.E.H., D.W., and V.P., unpublished data), consistent with the previously reported expression of c-ret, a component of the GDNF receptor (45, 46). Withdrawal thresholds to mechanical and thermal stimulation were measured before SNI. Ret-OE mice had significantly higher baseline mechanical ( g compared with g, P 0.001; Fig. 1B) and thermal ( s compared with s, P 0.05) thresholds than corresponding WT controls. Two of the eight Ret-OE mice tested failed to respond to the highest von Frey filament used, so a maximum value of 3.63 g was assigned to them. These findings are analogous to those by Blakeman et al. (47), who described transgenic mice that ectopically overexpress galanin under the control of the platelet-derived growth factor (PDGF ) promoter (although the precise neuronal distribution of galanin in these PDGF -OE animals has not been documented). These mice are also hypoalgesic to thermal but not mechanical stimulation in the intact state. Thus, both the Ret-OE and PDGF -OE transgenic lines imply an antinociceptive role for galanin in intact animals, albeit in a modality-specific manner. Immunohistochemistry demonstrates that intact Ret-OE animals abundantly expressed galanin in DRG neurons of various sizes, the majority of which colocalize with IB4 (Fig. 3). However, the neurons that expressed the highest levels of galanin were of large diameter and did not bind IB4. These data are consistent with that reported previously, demonstrating that 21% of c-ret-positive neurons in the DRG do not bind IB4 and are predominantly large-diameter cells (45). Galanin expression in the intact Ret-OE spinal cord appears to extend deeper into lamina II than in WT controls (Fig. 4), consistent with its localization in the c-ret neurons, which mostly terminate in inner lamina II (45). IB4-negative and IB4-positive neurons appear to be functionally distinct in that they have particular neurochemical phenotypes and corresponding electrophysiological characteristics that are relevant to the detection and processing of nociceptive stimuli (48 50). Furthermore, IB4- negative and IB4-positive nociceptors terminate in distinct regions of the superficial dorsal horn: IB4-negative afferents project mostly into lamina I and outer lamina II, whereas IB4-positive afferents terminate mostly in inner lamina II (45). The differing nociceptive behavioral phenotypes in the PDGF -OE and Ret-OE lines therefore may reflect differences in the neuronal populations that ectopically express galanin and its precise site of synaptic release within the dorsal horn as well as its potential pre- and postsynaptic targets. Alternatively, because our previous data have demonstrated that galanin plays a role in the developmental survival of DRG neurons (37), it is possible that the sustained overexpression of galanin in the DRG may result in developmental compensatory changes that could contribute to the observed phenotypic changes in pain behavior in the adult transgenic animals of either line. After SNI, the Ret-OE animals had reduced mechanical withdrawal thresholds when compared with baseline measurements from day 2 onward. At 14 days post-sni, the threshold was g (Fig. 1B), which represents a decrease of 73% compared with a 79% decrease in the WT controls at the same time point. It should be noted, however, that at all time points tested after SNI, the mechanical thresholds in the Ret-OE animals remained higher than in the intact WT animals (Fig. 1B), and it is therefore debatable as to whether the reduction in threshold values after SNI can be classified as allodynia in the Ret-OE animals. Galanin expression in the DRG of the Ret-OE animals did not significantly alter after SNI. The increase that was observed in NEUROSCIENCE Holmes et al. PNAS May 13, 2003 vol. 100 no
5 Fig. 4. Immunolocalization of galanin in dorsal spinal cord before and 7 days post-sni. Galanin was present in primary afferent fibers projecting into lamina I and II of the dorsal horn. Immunoreactivity was increased after SNI ipsilateral to the injury side (Right). Some increase in dorsal horn staining was observed in the intact Gal-OE, which was increased further after SNI. In the Ret-OE, galanin immunoreactivity was stronger and extended further, into inner lamina II. Staining was increased after SNI ipsilateral to the injury side, which represents the increase in endogenous galanin expression. (Bar 100 M.) the dorsal horn staining, ipsilateral to the side of injury (Fig. 3), is due to the injury-induced up-regulation of the endogenous galanin gene. Galanin Mutant Mice. Baseline withdrawal thresholds to noxious mechanical and thermal stimuli were measured and compared with WT controls. The mutant mice were significantly more sensitive to both modalities compared with WT controls, with mechanical withdrawal thresholds of g and g (P 0.001; Fig. 2A) and thermal withdrawal latencies of and s (P 0.05; Fig. 2B) for WT and mutants, respectively. These results are in accordance with those reported previously by Kerr et al. (16) and are once again consistent with the proposed inhibitory role played by galanin in the intact animal. As predicted from our previous published data when using the partial nerve ligation model, the galanin mutant animals failed to develop significant mechanical allodynia after SNI on any of the test days (Fig. 2A). The withdrawal thresholds changed by a maximum of 0.13 g at 5 days post-sni (which was not significantly different from the mutant baseline threshold) compared with 0.94 g in the WT controls at this time point. It is clear that the markedly diminished neuropathic pain behavior and decreased electrophysiological responses (39) in the galanin mutant animals are contrary to (i) that observed in the Gal-OE and Ret-OE animals and (ii) the majority of the existing pharmacological data. Further, the deficits in the mutant animals are present by the first day after the nerve lesion in the partial nerve ligation and SNI chronic pain models we have studied, whereas if the phenotype was due to the absence of galanin in the adult, then differences would be expected to occur 3 5 days after injury, which is the time course for the upregulation of the endogenous galanin gene after injury. To explain the above disparities, we hypothesized that the chronic absence of galanin throughout sensory neuron development may cause the loss of a subset of neurons that is important for injury-related pain behavior. Consistent with this, we have demonstrated previously a 13% reduction in the total number of neurons in the adult DRG of galanin mutant animals (37). These deficits were associated with a 2.8-fold increase in the number of apoptotic cells at postnatal day 3 (37). For each of the above transgenic and knockout lines, the effects of galanin on the modulation of pain behavior are mediated via the activation of three G protein-coupled receptor subtypes, designated GALR1, GALR2, and GALR3 (for review, see ref. 51). Binding of galanin to GALR1 has been shown to inhibit adenylyl cyclase (52). Activation of GALR2 stimulates both phospholipase C and ERK (53, 54). These studies raise the possibility that the receptor subtypes function in divergent molecular cascades in the DRG and may play differing roles in cgi doi pnas Holmes et al.
6 neuronal function in the DRG. In situ hybridization studies have shown that GALR1-expressing neurons in the DRG are larger than those that express GALR2, whereas only 20% of neurons express both receptor subtypes (55 57). GALR3 expression has not been demonstrated convincingly in the DRG or dorsal horn (58). In situ hybridization studies also have shown that high levels of GALR1 expression are detected within the interneurons of the superficial dorsal horn, whereas GALR2 is detected only at low levels throughout the dorsal horn (55). The DRG expression of both GALR1 and GALR2 is down-regulated markedly after axotomy (56, 57). Thus, the complement of galanin receptor subtypes both within particular populations of nociceptors and in the neurons in the dorsal horn with which they synapse is clearly crucial to the effects of galanin on nociceptive behavior. Recent studies have indicated that activation of GALR2 may be the principal receptor subtype that mediates the facilitatory effect of galanin in the intact animal, whereas GALR1 is predominantly responsible for the inhibitory actions of galanin in states of nerve injury (3, 17). Axotomy (or any of the neuropathic pain models) markedly increases endogenous galanin synthesis in the DRG, which then is transported to the terminal afferents of the primary sensory neurons. The enhanced release of galanin may predominantly activate GALR1, which is expressed postsynaptically on the dorsal horn interneurons, because presynaptic expression of GALR1 and GALR2 is down-regulated markedly after injury (56, 57). It is also important to note that in both the overexpressing and mutant transgenic lines, it is possible that the chronic alterations in the levels of galanin in the DRG may modify the expression of one or more of the galanin receptor subtype(s) in the DRG and or spinal cord. To date, we have failed to detect specific immunohistochemical staining in the mouse DRG or cord when using antisera raised against rat GALR1 or GALR2 (data not shown); thus, we have been unable to perform expression studies at the protein level in the DRG or spinal cord that would allow us to test the above hypothesis. In summary, we have characterized two transgenic lines that either inducibly overexpress galanin after nerve injury, with a distribution that recapitulates that observed endogenously in WT animals (Gal-OE), or constitutively and ectopically overexpress galanin in both intact and nerve-injured animals (Ret-OE). Behavioral analysis of these transgenic lines and the existing galanin mutants coupled with the previous published data when using PDGF -OE animals supports the hypothesis that galanin indeed does play an inhibitory role in pain processing in the intact animal and in states of nerve injury. This work was supported by the Medical Research Council and the Wellcome Trust. 1. Bartfai, T., Hökfelt, T. & Langel, U. (1993) Crit. Rev. Neurobiol. 7, Xu, X. J., Hökfelt, T., Bartfai, T. & Wiesenfeld-Hallin, Z. (2000) Neuropeptides 34, Liu, H. & Hökfelt, T. (2002) Trends Pharmacol. Sci. 23, Hökfelt, T., Wiesenfeld-Hallin, Z., Villar, M. & Melander, T. (1987) Neurosci. Lett. 83, Skofitsch, G. & Jacobowitz, D. M. (1985) Brain Res. Bull. 15, Hao, J. X., Shi, T. J., Xu, I. S., Kaupilla, T., Xu, X. J., Hökfelt, T., Bartfai, T. & Wiesenfeld-Hallin, Z. (1999) Eur. J. Neurosci. 11, Ma, W. & Bisby, M. A. (1999) Neurosci. Lett. 262, Shi, T. J., Cui, J. G., Meyerson, B. A., Linderoth, B. & Hökfelt, T. (1999) Neuroscience 93, Ma, W. & Bisby, M. A. (1997) Neuroscience 79, Hökfelt, T., Zhang, X. & Wiesenfeld-Hallin, Z. (1994) Trends Neurosci. 17, Colvin, L. A., Mark, M. A. & Duggan, A. W. (1997) Brain Res. 766, Kerr, B. J., Wynick, D., Thompson, S. W. & McMahon, S. B. (2000) Prog. Brain Res. 129, Wiesenfeld-Hallin, Z. & Xu, X. J. (1998) Ann. N.Y. Acad. Sci. 863, Cridland, R. A. & Henry, J. L. (1988) Neuropeptides 11, Flatters, S. J., Fox, A. J. & Dickenson, A. H. (2002) Pain 98, Kerr, B. J., Cafferty, W. B., Gupta, Y. K., Bacon, A., Wynick, D., McMahon, S. B. & Thompson, S. W. (2000) Eur. J. Neurosci. 12, Liu, H. X., Brumovsky, P., Schmidt, R., Brown, W., Payza, K., Hodzic, L., Pou, C., Godbout, C. & Hökfelt, T. (2001) Proc. Natl. Acad. Sci. USA 98, Puttick, R. M., Pinnock, R. D. & Woodruff, G. N. (1994) Eur. J. Pharmacol. 254, Reeve, A. J., Walker, K., Urban, L. & Fox, A. (2000) Neurosci. Lett. 295, Wiesenfeld-Hallin, Z., Villar, M. J. & Hökfelt, T. (1988) Exp. Brain Res. 71, Kuraishi, Y., Kawamura, M., Yamaguchi, T., Houtani, T., Kawabata, S., Futaki, S., Fujii, N. & Satoh, M. (1991) Pain 44, Post, C., Alari, L. & Hökfelt, T. (1988) Acta Physiol. Scand. 132, Wiesenfeld-Hallin, Z., Xu, X. J., Villar, M. J. & Hökfelt, T. (1989) Neurosci. Lett. 105, Wall, P. D., Scadding, J. W. & Tomkiewicz, M. M. (1979) Pain 6, Bennett, G. J. & Xie, Y. K. (1988) Pain 33, Seltzer, Z., Dubner, R. & Shir, Y. (1990) Pain 43, Kim, S. H. & Chung, J. M. (1992) Pain 50, Gazelius, B., Cui, J. G., Svensson, M., Meyerson, B. & Linderoth, B. (1996) NeuroReport 7, Kupers, R., Yu, W., Persson, J. K., Xu, X. J. & Wiesenfeld-Hallin, Z. (1998) Pain 76, Decosterd, I. & Woolf, C. J. (2000) Pain 87, Wiesenfeld-Hallin, Z., Xu, X. J., Langel, U., Bedecs, K., Hökfelt, T. & Bartfai, T. (1992) Proc. Natl. Acad. Sci. USA 89, Xu, S., Zhang, Y., Lundeberg, T. & Yu, L. (2000) Regul. Pept. 95, Verge, V. M., Xu, X. J., Langel, U., Hökfelt, T., Wiesenfeld-Hallin, Z. & Bartfai, T. (1993) Neurosci. Lett. 149, Liu, H. & Hökfelt, T. (2000) Brain Res. 886, Yu, L. C., Lundeberg, S., An, H., Wang, F. X. & Lundeberg, T. (1999) Life Sci. 64, Eaton, M. J., Karmally, S., Martinez, M. A., Plunkett, J. A., Lopez, T. & Cejas, P. J. (1999) J. Periphery Nerv. Syst. 4, Holmes, F. E., Mahoney, S., King, V. R., Bacon, A., Kerr, N. C., Pachnis, V., Curtis, R., Priestley, J. V. & Wynick, D. (2000) Proc. Natl. Acad. Sci. USA 97, Wynick, D., Small, C. J., Bacon, A., Holmes, F. E., Norman, M., Ormandy, C. J., Kilic, E., Kerr, N. C., Ghatei, M., Talamantes, F., et al. (1998) Proc. Natl. Acad. Sci. USA 95, Kerr, B. J., Thompson, S. W., Wynick, D. & McMahon, S. B. (2001) Neuro- Report 12, Bacon, A., Holmes, F. E., Small, C. J., Ghatei, M., Mahoney, S., Bloom, S. R. & Wynick, D. (2002) NeuroReport 13, Sukumaran, M., Waxman, S. G., Wood, J. N. & Pachnis, V. (2001) Dev. Dyn. 222, Hargreaves, K., Dubner, R., Brown, F., Flores, C. & Joris, J. (1988) Pain 32, Chaplan, S. R., Bach, F. W., Pogrel, J. W., Chung, J. M. & Yaksh, T. L. (1994) J. Neurosci. Methods 53, Dixon, W. J. (1980) Annu. Rev. Pharmacol. Toxicol. 20, Bennett, D. L., Michael, G. J., Ramachandran, N., Munson, J. B., Averill, S., Yan, Q., McMahon, S. B. & Priestley, J. V. (1998) J. Neurosci. 18, Bennett, D. L., Boucher, T. J., Armanini, M. P., Poulsen, K. T., Michael, G. J., Priestley, J. V., Phillips, H. S., McMahon, S. B. & Shelton, D. L. (2000) J. Neurosci. 20, Blakeman, K. H., Holmberg, K., Hao, J. X., Xu, X. J., Kahl, U., Lendahl, U., Bartfai, T., Wiesenfeld-Hallin, Z. & Hökfelt, T. (2001) NeuroReport 12, Drew, L. J., Wood, J. N. & Cesare, P. (2002) J. Neurosci. 22, RC Gerke, M. B. & Plenderleith, M. B. (2001) Brain Res. 911, Stucky, C. L. & Lewin, G. R. (1999) J. Neurosci. 19, Branchek, T. A., Smith, K. E., Gerald, C. & Walker, M. W. (2000) Trends Pharmacol. Sci. 21, Habert-Ortoli, E., Amiranoff, B., Loquet, I., Laburthe, M. & Mayaux, J. F. (1994) Proc. Natl. Acad. Sci. USA 91, Fathi, Z., Cunningham, A. M., Iben, L. G., Battaglino, P. B., Ward, S. A., Nichol, K. A., Pine, K. A., Wang, J., Goldstein, M. E., Iismaa, T. P., et al. (1997) Brain Res. Mol. Brain Res. 51, Wittau, N., Grosse, R., Kalkbrenner, F., Gohla, A., Schultz, G. & Gudermann, T. (2000) Oncogene 19, O Donnell, D., Ahmad, S., Wahlestedt, C. & Walker, P. (1999) J. Comp. Neurol. 409, Sten Shi, T. J., Zhang, X., Holmberg, K., Xu, Z. Q. & Hökfelt, T. (1997) Neurosci. Lett. 237, Xu, Z. Q., Shi, T. J., Landry, M. & Hökfelt, T. (1996) NeuroReport 20, Mennicken, F., Hoffert, C., Pelletier, M., Ahmad, S. & O Donnell, D. (2003) J. Chem. Neuroanat. 24, NEUROSCIENCE Holmes et al. PNAS May 13, 2003 vol. 100 no
Galanin is a 29-aa (30 aa in humans) neuropeptide (1). It has
 Receptor subtype-specific pronociceptive and analgesic actions of galanin in the spinal cord: Selective actions via GalR1 and GalR2 receptors Hong-Xiang Liu*, Pablo Brumovsky*, Ralf Schmidt, William Brown,
Receptor subtype-specific pronociceptive and analgesic actions of galanin in the spinal cord: Selective actions via GalR1 and GalR2 receptors Hong-Xiang Liu*, Pablo Brumovsky*, Ralf Schmidt, William Brown,
Mechanical sensitization of cutaneous sensory fibers in the spared nerve injury mouse model
 Smith et al. Molecular Pain 2013, 9:61 MOLECULAR PAIN SHORT REPORT Open Access Mechanical sensitization of cutaneous sensory fibers in the spared nerve injury mouse model Amanda K Smith, Crystal L O Hara
Smith et al. Molecular Pain 2013, 9:61 MOLECULAR PAIN SHORT REPORT Open Access Mechanical sensitization of cutaneous sensory fibers in the spared nerve injury mouse model Amanda K Smith, Crystal L O Hara
Sensory coding and somatosensory system
 Sensory coding and somatosensory system Sensation and perception Perception is the internal construction of sensation. Perception depends on the individual experience. Three common steps in all senses
Sensory coding and somatosensory system Sensation and perception Perception is the internal construction of sensation. Perception depends on the individual experience. Three common steps in all senses
A role for uninjured afferents in neuropathic pain
 Acta Physiologica Sinica, October 25, 2008, 60 (5): 605-609 http://www.actaps.com.cn 605 Review A role for uninjured afferents in neuropathic pain Richard A. Meyer 1,2,3,*, Matthias Ringkamp 1 Departments
Acta Physiologica Sinica, October 25, 2008, 60 (5): 605-609 http://www.actaps.com.cn 605 Review A role for uninjured afferents in neuropathic pain Richard A. Meyer 1,2,3,*, Matthias Ringkamp 1 Departments
Enhanced formalin nociceptive responses following L5 nerve ligation in the rat reveals neuropathy-induced inflammatory hyperalgesia
 University of Kentucky From the SelectedWorks of Renee R. Donahue 2001 Enhanced formalin nociceptive responses following L5 nerve ligation in the rat reveals neuropathy-induced inflammatory hyperalgesia
University of Kentucky From the SelectedWorks of Renee R. Donahue 2001 Enhanced formalin nociceptive responses following L5 nerve ligation in the rat reveals neuropathy-induced inflammatory hyperalgesia
Centrally Mediated Anti-Hyperalgesic and Anti-Allodynic Effect of Tolperisone in Spared Nerve Injury Model of Neuropathic Pain
 Centrally Mediated Anti-Hyperalgesic and Anti-Allodynic Effect of Tolperisone in Spared Nerve Injury Model of Neuropathic Pain Keyur S Patel *, Punam D Sachdeva Department of Pharmacology, A.R.College
Centrally Mediated Anti-Hyperalgesic and Anti-Allodynic Effect of Tolperisone in Spared Nerve Injury Model of Neuropathic Pain Keyur S Patel *, Punam D Sachdeva Department of Pharmacology, A.R.College
Original Article Upregulation of neuregulin-1 reverses signs of neuropathic pain in rats
 Int J Clin Exp Pathol 2014;7(9):5916-5921 www.ijcep.com /ISSN:1936-2625/IJCEP0001263 Original Article Upregulation of neuregulin-1 reverses signs of neuropathic pain in rats Guojun Wang 1,2*, Dawei Dai
Int J Clin Exp Pathol 2014;7(9):5916-5921 www.ijcep.com /ISSN:1936-2625/IJCEP0001263 Original Article Upregulation of neuregulin-1 reverses signs of neuropathic pain in rats Guojun Wang 1,2*, Dawei Dai
Opioid Receptor Trafficking in Pain States
 Encyclopedia of Pain Springer-Verlag Berlin Heidelberg 2007 10.1007/978-3-540-29805-2_2962 Robert F. Schmidt and William D. Willis Opioid Receptor Trafficking in Pain States Catherine M. Cahill 3, Anne
Encyclopedia of Pain Springer-Verlag Berlin Heidelberg 2007 10.1007/978-3-540-29805-2_2962 Robert F. Schmidt and William D. Willis Opioid Receptor Trafficking in Pain States Catherine M. Cahill 3, Anne
The development of the nociceptive responses in neurokinin-1 receptor knockout mice
 SOMATENSORY SYSTEMS, PAIN The development of the nociceptive responses in neurokinin-1 receptor knockout mice Tamara E. King, 1,CA Mark J. S. Heath, 2 Pierre Debs, 3 Marion B. E. Davis, 2 Rene Hen 4 and
SOMATENSORY SYSTEMS, PAIN The development of the nociceptive responses in neurokinin-1 receptor knockout mice Tamara E. King, 1,CA Mark J. S. Heath, 2 Pierre Debs, 3 Marion B. E. Davis, 2 Rene Hen 4 and
Pharmacology of Pain Transmission and Modulation
 Pharmacology of Pain Transmission and Modulation 2 Jürg Schliessbach and Konrad Maurer Nociceptive Nerve Fibers Pain is transmitted to the central nervous system via thinly myelinated Aδ and unmyelinated
Pharmacology of Pain Transmission and Modulation 2 Jürg Schliessbach and Konrad Maurer Nociceptive Nerve Fibers Pain is transmitted to the central nervous system via thinly myelinated Aδ and unmyelinated
Galanin Receptor 1 Deletion Exacerbates Hippocampal Neuronal Loss After Systemic Kainate Administration in Mice.
 Current Literature In Basic Science Galanin Receptors Modulate Seizures Galanin Receptor 1 Deletion Exacerbates Hippocampal Neuronal Loss After Systemic Kainate Administration in Mice. Schauwecker PE.
Current Literature In Basic Science Galanin Receptors Modulate Seizures Galanin Receptor 1 Deletion Exacerbates Hippocampal Neuronal Loss After Systemic Kainate Administration in Mice. Schauwecker PE.
A subpopulation of nociceptors specifically linked to itch
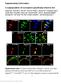 Supplementary Information A subpopulation of nociceptors specifically linked to itch Liang Han 1, Chao Ma 3,4, Qin Liu 1,2, Hao-Jui Weng 1,2, Yiyuan Cui 5, Zongxiang Tang 1,2, Yushin Kim 1, Hong Nie 4,
Supplementary Information A subpopulation of nociceptors specifically linked to itch Liang Han 1, Chao Ma 3,4, Qin Liu 1,2, Hao-Jui Weng 1,2, Yiyuan Cui 5, Zongxiang Tang 1,2, Yushin Kim 1, Hong Nie 4,
GABA B Receptor Agonist Baclofen Has Non-Specific Antinociceptive Effect in the Model of Peripheral Neuropathy in the Rat
 Physiol. Res. 3: 31-3, GABA B Receptor Agonist Baclofen Has Non-Specific Antinociceptive Effect in the Model of Peripheral Neuropathy in the Rat M. FRANĚK, Š. VACULÍN, R. ROKYTA Department of Normal, Pathological
Physiol. Res. 3: 31-3, GABA B Receptor Agonist Baclofen Has Non-Specific Antinociceptive Effect in the Model of Peripheral Neuropathy in the Rat M. FRANĚK, Š. VACULÍN, R. ROKYTA Department of Normal, Pathological
Introduction to some interesting research questions: Molecular biology of the primary afferent nociceptor
 Introduction to some interesting research questions: Molecular biology of the primary afferent nociceptor NOCICEPTORS ARE NOT IDENTICAL PEPTIDE SubP/CGRP Trk A NON-PEPTIDE IB4 P2X 3 c-ret Snider and McMahon
Introduction to some interesting research questions: Molecular biology of the primary afferent nociceptor NOCICEPTORS ARE NOT IDENTICAL PEPTIDE SubP/CGRP Trk A NON-PEPTIDE IB4 P2X 3 c-ret Snider and McMahon
NMDA-Receptor Antagonists and Opioid Receptor Interactions as Related to Analgesia and Tolerance
 Vol. 19 No. 1(Suppl.) January 2000 Journal of Pain and Symptom Management S7 Proceedings Supplement NDMA-Receptor Antagonists: Evolving Role in Analgesia NMDA-Receptor Antagonists and Opioid Receptor Interactions
Vol. 19 No. 1(Suppl.) January 2000 Journal of Pain and Symptom Management S7 Proceedings Supplement NDMA-Receptor Antagonists: Evolving Role in Analgesia NMDA-Receptor Antagonists and Opioid Receptor Interactions
The Egyptian Journal of Hospital Medicine (January 2018) Vol. 70 (12), Page
 The Egyptian Journal of Hospital Medicine (January 2018) Vol. 70 (12), Page 2172-2177 Blockage of HCN Channels with ZD7288 Attenuates Mechanical Hypersensitivity in Rats Model of Diabetic Neuropathy Hussain
The Egyptian Journal of Hospital Medicine (January 2018) Vol. 70 (12), Page 2172-2177 Blockage of HCN Channels with ZD7288 Attenuates Mechanical Hypersensitivity in Rats Model of Diabetic Neuropathy Hussain
Peripheral nerve injury alters excitatory synaptic transmission in lamina II of the rat dorsal horn
 J Physiol (2003), 548.1, pp. 131 138 DOI: 10.1113/jphysiol.2002.036186 The Physiological Society 2003 www.jphysiol.org Peripheral nerve injury alters excitatory synaptic transmission in lamina II of the
J Physiol (2003), 548.1, pp. 131 138 DOI: 10.1113/jphysiol.2002.036186 The Physiological Society 2003 www.jphysiol.org Peripheral nerve injury alters excitatory synaptic transmission in lamina II of the
Focal Lysolecithin-Induced Demyelination of Peripheral Afferents Results in Neuropathic Pain Behavior That Is Attenuated by Cannabinoids
 The Journal of Neuroscience, April 15, 2003 23(8):3221 3233 3221 Focal Lysolecithin-Induced Demyelination of Peripheral Afferents Results in Neuropathic Pain Behavior That Is Attenuated by Cannabinoids
The Journal of Neuroscience, April 15, 2003 23(8):3221 3233 3221 Focal Lysolecithin-Induced Demyelination of Peripheral Afferents Results in Neuropathic Pain Behavior That Is Attenuated by Cannabinoids
Rapamycin Suppresses Astrocytic and Microglial Activation and Reduced Development of Neuropathic Pain after Spinal Cord Injury in Mice.
 Rapamycin Suppresses Astrocytic and Microglial Activation and Reduced Development of Neuropathic Pain after Spinal Cord Injury in Mice. Satoshi Tateda, M.D., Haruo Kanno, M.D., Ph.D., Hiroshi Ozawa, M.D.,
Rapamycin Suppresses Astrocytic and Microglial Activation and Reduced Development of Neuropathic Pain after Spinal Cord Injury in Mice. Satoshi Tateda, M.D., Haruo Kanno, M.D., Ph.D., Hiroshi Ozawa, M.D.,
NIH Public Access Author Manuscript J Neuropathic Pain Symptom Palliation. Author manuscript; available in PMC 2007 March 26.
 NIH Public Access Author Manuscript Published in final edited form as: J Neuropathic Pain Symptom Palliation. 2005 ; 1(1): 19 23. Sympathetic Fiber Sprouting in Chronically Compressed Dorsal Root Ganglia
NIH Public Access Author Manuscript Published in final edited form as: J Neuropathic Pain Symptom Palliation. 2005 ; 1(1): 19 23. Sympathetic Fiber Sprouting in Chronically Compressed Dorsal Root Ganglia
Evaluation of Gabapentin and S-( )-3-Isobutylgaba in a Rat Model of Postoperative Pain
 0022-3565/97/2823-1242$03.00/0 THE JOURNAL OF PHARMACOLOGY AND EXPERIMENTAL THERAPEUTICS Vol. 282, No. 3 Copyright 1997 by The American Society for Pharmacology and Experimental Therapeutics Printed in
0022-3565/97/2823-1242$03.00/0 THE JOURNAL OF PHARMACOLOGY AND EXPERIMENTAL THERAPEUTICS Vol. 282, No. 3 Copyright 1997 by The American Society for Pharmacology and Experimental Therapeutics Printed in
EFFECTS OF Pueraria mirifica ON NEUROPATHIC PAIN IN RATS
 EFFECTS OF Pueraria mirifica ON NEUROPATHIC PAIN IN RATS Kamolchanok Tanchotikul 1, Quankamon Dejativongse Na Ayudhya 1*, Orawan Piyaboon 1 and Supin Chompoopong 2 1 Department of Biology, Mahidol Wittayanusorn
EFFECTS OF Pueraria mirifica ON NEUROPATHIC PAIN IN RATS Kamolchanok Tanchotikul 1, Quankamon Dejativongse Na Ayudhya 1*, Orawan Piyaboon 1 and Supin Chompoopong 2 1 Department of Biology, Mahidol Wittayanusorn
Published online October 10, 2016
 Published online October 10, 2016 reviewed by Jim C. Eisenach, Wake Forest University; Ronald Lindsay, Zebra Biologics; Remi Quirion, McGill University; and Tony L. Yaksh, University of California, San
Published online October 10, 2016 reviewed by Jim C. Eisenach, Wake Forest University; Ronald Lindsay, Zebra Biologics; Remi Quirion, McGill University; and Tony L. Yaksh, University of California, San
Glycine-gated ion channels Converging mechanism and therapeutic potentials
 Glycine-gated ion channels Converging mechanism and therapeutic potentials Li Zhang Laboratory for Integrative Neuroscience, National Institute on Alcohol Abuse and Alcoholism, National Institutes of Health,
Glycine-gated ion channels Converging mechanism and therapeutic potentials Li Zhang Laboratory for Integrative Neuroscience, National Institute on Alcohol Abuse and Alcoholism, National Institutes of Health,
International Conference on Biological Sciences and Technology (BST 2016)
 International Conference on Biological Sciences and Technology (BST 2016) Analgesic Contrast of Diverse Administrations of Botulinum Neurotoxin A (BoTN/A) Using Rat Neuropathic Pain Model Qiong-Fang YANG1,3,
International Conference on Biological Sciences and Technology (BST 2016) Analgesic Contrast of Diverse Administrations of Botulinum Neurotoxin A (BoTN/A) Using Rat Neuropathic Pain Model Qiong-Fang YANG1,3,
Pathophysiology of Pain
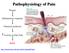 Pathophysiology of Pain Wound Inflammatory response Chemical mediators Activity in Pain Path PAIN http://neuroscience.uth.tmc.edu/s2/chapter08.html Chris Cohan, Ph.D. Dept. of Pathology/Anat Sci University
Pathophysiology of Pain Wound Inflammatory response Chemical mediators Activity in Pain Path PAIN http://neuroscience.uth.tmc.edu/s2/chapter08.html Chris Cohan, Ph.D. Dept. of Pathology/Anat Sci University
New high affinity peptide antagonists to the spinal galanin
 BrNM Journal of Pharmacology (1995) 116, 276-28 B 1995 Stockton Press All rights reserved 7-1188/95 $12. S New high affinity peptide antagonists to the spinal galanin receptor Xiao-Jun Xu, 'Zsuzsanna Wiesenfeld-Hallin,
BrNM Journal of Pharmacology (1995) 116, 276-28 B 1995 Stockton Press All rights reserved 7-1188/95 $12. S New high affinity peptide antagonists to the spinal galanin receptor Xiao-Jun Xu, 'Zsuzsanna Wiesenfeld-Hallin,
Seizure: the clinical manifestation of an abnormal and excessive excitation and synchronization of a population of cortical
 Are There Sharing Mechanisms of Epilepsy, Migraine and Neuropathic Pain? Chin-Wei Huang, MD, PhD Department of Neurology, NCKUH Basic mechanisms underlying seizures and epilepsy Seizure: the clinical manifestation
Are There Sharing Mechanisms of Epilepsy, Migraine and Neuropathic Pain? Chin-Wei Huang, MD, PhD Department of Neurology, NCKUH Basic mechanisms underlying seizures and epilepsy Seizure: the clinical manifestation
TMC9 as a novel mechanosensitive ion channel
 TMC9 as a novel mechanosensitive ion channel Mechanical forces play numerous roles in physiology. When an object contacts our skin, it exerts a force that is encoded as touch or pain depending on its intensity.
TMC9 as a novel mechanosensitive ion channel Mechanical forces play numerous roles in physiology. When an object contacts our skin, it exerts a force that is encoded as touch or pain depending on its intensity.
Introduction ORIGINAL ARTICLE. Seiji Ohtori Kazuhisa Takahashi Hideshige Moriya
 Eur Spine J (2003) 12 :211 215 DOI 10.1007/s00586-002-0506-7 ORIGINAL ARTICLE Seiji Ohtori Kazuhisa Takahashi Hideshige Moriya Calcitonin gene-related peptide immunoreactive DRG neurons innervating the
Eur Spine J (2003) 12 :211 215 DOI 10.1007/s00586-002-0506-7 ORIGINAL ARTICLE Seiji Ohtori Kazuhisa Takahashi Hideshige Moriya Calcitonin gene-related peptide immunoreactive DRG neurons innervating the
Potential for delta opioid receptor agonists as analgesics in chronic pain therapy
 Potential for delta opioid receptor agonists as analgesics in chronic pain therapy David Kendall & Bengt von Mentzer; PharmNovo AB/UK Alex Conibear & Eamonn Kelly, University of Bristol Junaid Asghar,
Potential for delta opioid receptor agonists as analgesics in chronic pain therapy David Kendall & Bengt von Mentzer; PharmNovo AB/UK Alex Conibear & Eamonn Kelly, University of Bristol Junaid Asghar,
Charlie Taylor, PhD CpTaylor Consulting Chelsea, MI, USA
 Contribution of Calcium Channel α 2 δ Binding Sites to the Pharmacology of Gabapentin and Pregabalin Charlie Taylor, PhD CpTaylor Consulting Chelsea, MI, USA Disclosure Information Charlie Taylor, PhD
Contribution of Calcium Channel α 2 δ Binding Sites to the Pharmacology of Gabapentin and Pregabalin Charlie Taylor, PhD CpTaylor Consulting Chelsea, MI, USA Disclosure Information Charlie Taylor, PhD
BRIEF COMMUNICATIONS American Neurological Association Published by Wiley-Liss, Inc., through Wiley Subscription Services
 BRIEF COMMUNICATIONS Gene Transfer of Glutamic Acid Decarboxylase Reduces Neuropathic Pain Shuanglin Hao, MD, PhD, 1 Marina Mata, MD, 1 Darren Wolfe, PhD, 2 Joseph C. Glorioso, PhD, 2 and David J. Fink,
BRIEF COMMUNICATIONS Gene Transfer of Glutamic Acid Decarboxylase Reduces Neuropathic Pain Shuanglin Hao, MD, PhD, 1 Marina Mata, MD, 1 Darren Wolfe, PhD, 2 Joseph C. Glorioso, PhD, 2 and David J. Fink,
Pain Pathways. Dr Sameer Gupta Consultant in Anaesthesia and Pain Management, NGH
 Pain Pathways Dr Sameer Gupta Consultant in Anaesthesia and Pain Management, NGH Objective To give you a simplistic and basic concepts of pain pathways to help understand the complex issue of pain Pain
Pain Pathways Dr Sameer Gupta Consultant in Anaesthesia and Pain Management, NGH Objective To give you a simplistic and basic concepts of pain pathways to help understand the complex issue of pain Pain
211MDS Pain theories
 211MDS Pain theories Definition In 1986, the International Association for the Study of Pain (IASP) defined pain as a sensory and emotional experience associated with real or potential injuries, or described
211MDS Pain theories Definition In 1986, the International Association for the Study of Pain (IASP) defined pain as a sensory and emotional experience associated with real or potential injuries, or described
What Does the Mechanism of Spinal Cord Stimulation Tell Us about Complex Regional Pain Syndrome?pme_
 Pain Medicine 2010; 11: 1278 1283 Wiley Periodicals, Inc. What Does the Mechanism of Spinal Cord Stimulation Tell Us about Complex Regional Pain Syndrome?pme_915 1278..1283 Joshua P. Prager, MD, MS Center
Pain Medicine 2010; 11: 1278 1283 Wiley Periodicals, Inc. What Does the Mechanism of Spinal Cord Stimulation Tell Us about Complex Regional Pain Syndrome?pme_915 1278..1283 Joshua P. Prager, MD, MS Center
Somatic Sensation (MCB160 Lecture by Mu-ming Poo, Friday March 9, 2007)
 Somatic Sensation (MCB160 Lecture by Mu-ming Poo, Friday March 9, 2007) Introduction Adrian s work on sensory coding Spinal cord and dorsal root ganglia Four somatic sense modalities Touch Mechanoreceptors
Somatic Sensation (MCB160 Lecture by Mu-ming Poo, Friday March 9, 2007) Introduction Adrian s work on sensory coding Spinal cord and dorsal root ganglia Four somatic sense modalities Touch Mechanoreceptors
Materials and Methods
 Anesthesiology 2003; 98:203 8 2003 American Society of Anesthesiologists, Inc. Lippincott Williams & Wilkins, Inc. Intrathecal Lidocaine Reverses Tactile Allodynia Caused by Nerve Injuries and Potentiates
Anesthesiology 2003; 98:203 8 2003 American Society of Anesthesiologists, Inc. Lippincott Williams & Wilkins, Inc. Intrathecal Lidocaine Reverses Tactile Allodynia Caused by Nerve Injuries and Potentiates
Biomechanics of Pain: Dynamics of the Neuromatrix
 Biomechanics of Pain: Dynamics of the Neuromatrix Partap S. Khalsa, D.C., Ph.D. Department of Biomedical Engineering The Neuromatrix From: Melzack R (1999) Pain Suppl 6:S121-6. NIOSH STAR Symposium May
Biomechanics of Pain: Dynamics of the Neuromatrix Partap S. Khalsa, D.C., Ph.D. Department of Biomedical Engineering The Neuromatrix From: Melzack R (1999) Pain Suppl 6:S121-6. NIOSH STAR Symposium May
Supplementary Table 1. List of primers used in this study
 Supplementary Table 1. List of primers used in this study Gene Forward primer Reverse primer Rat Met 5 -aggtcgcttcatgcaggt-3 5 -tccggagacacaggatgg-3 Rat Runx1 5 -cctccttgaaccactccact-3 5 -ctggatctgcctggcatc-3
Supplementary Table 1. List of primers used in this study Gene Forward primer Reverse primer Rat Met 5 -aggtcgcttcatgcaggt-3 5 -tccggagacacaggatgg-3 Rat Runx1 5 -cctccttgaaccactccact-3 5 -ctggatctgcctggcatc-3
Research Article Contralateral Metabolic Activation Related to Plastic Changes in the Spinal Cord after Peripheral Nerve Injury in Rats
 Neural Plasticity Volume 2015, Article ID 438319, 6 pages http://dx.doi.org/10.1155/2015/438319 Research Article Contralateral Metabolic Activation Related to Plastic Changes in the Spinal Cord after Peripheral
Neural Plasticity Volume 2015, Article ID 438319, 6 pages http://dx.doi.org/10.1155/2015/438319 Research Article Contralateral Metabolic Activation Related to Plastic Changes in the Spinal Cord after Peripheral
Receptors and Neurotransmitters: It Sounds Greek to Me. Agenda. What We Know About Pain 9/7/2012
 Receptors and Neurotransmitters: It Sounds Greek to Me Cathy Carlson, PhD, RN Northern Illinois University Agenda We will be going through this lecture on basic pain physiology using analogies, mnemonics,
Receptors and Neurotransmitters: It Sounds Greek to Me Cathy Carlson, PhD, RN Northern Illinois University Agenda We will be going through this lecture on basic pain physiology using analogies, mnemonics,
THE EFFECT OF VOLUNATARY EXERCISE ON NEUROPATHIC PAIN. Kevin L. Farmer
 THE EFFECT OF VOLUNATARY EXERCISE ON NEUROPATHIC PAIN BY Kevin L. Farmer Submitted to the graduate degree program in Rehabilitation Science and the Graduate Faculty of the University of Kansas Medical
THE EFFECT OF VOLUNATARY EXERCISE ON NEUROPATHIC PAIN BY Kevin L. Farmer Submitted to the graduate degree program in Rehabilitation Science and the Graduate Faculty of the University of Kansas Medical
Analgesia Mediated by the TRPM8 Cold Receptor in Chronic Neuropathic Pain
 Current Biology 16, 1591 1605, August 22, 2006 ª2006 Elsevier Ltd All rights reserved DOI 10.1016/j.cub.2006.07.061 Analgesia Mediated by the TRPM8 Cold Receptor in Chronic Neuropathic Pain Article Clare
Current Biology 16, 1591 1605, August 22, 2006 ª2006 Elsevier Ltd All rights reserved DOI 10.1016/j.cub.2006.07.061 Analgesia Mediated by the TRPM8 Cold Receptor in Chronic Neuropathic Pain Article Clare
Research Article Effects of Electroacupuncture Treatment on Bone Cancer Pain Model with Morphine Tolerance
 Evidence-Based Complementary and Alternative Medicine Volume 2016, Article ID 8028474, 5 pages http://dx.doi.org/10.1155/2016/8028474 Research Article Effects of Electroacupuncture Treatment on Bone Cancer
Evidence-Based Complementary and Alternative Medicine Volume 2016, Article ID 8028474, 5 pages http://dx.doi.org/10.1155/2016/8028474 Research Article Effects of Electroacupuncture Treatment on Bone Cancer
Recent advances in study on the role of NPY in pain modulation
 19 5 2007 10 Chinese Bulletin of Life Sciences Vol. 19, No. 5 Oct., 2007 1004-0374(2007)05-0551-06 Y ( 710061) Y (NPY) NPY NPY Y1 Y2 NPY NPY Y1 Y2 Y Q426 R338 A Recent advances in study on the role of
19 5 2007 10 Chinese Bulletin of Life Sciences Vol. 19, No. 5 Oct., 2007 1004-0374(2007)05-0551-06 Y ( 710061) Y (NPY) NPY NPY Y1 Y2 NPY NPY Y1 Y2 Y Q426 R338 A Recent advances in study on the role of
EDUCATION M.D., Peking Union Medical College, Beijing, China, 1999 B.S., Beijing University, College of Life Science, Beijing, China, 1994
 CHAO MA, M.D. Yale University School of Medicine, Department of Anesthesiology, 333 Cedar Street, TMP3, New Haven, CT 06510, USA. Phone: 203-785-3522 (O), 203-606-7959 (C), Fax: 203-737-1528, Email: chao.ma@yale.edu
CHAO MA, M.D. Yale University School of Medicine, Department of Anesthesiology, 333 Cedar Street, TMP3, New Haven, CT 06510, USA. Phone: 203-785-3522 (O), 203-606-7959 (C), Fax: 203-737-1528, Email: chao.ma@yale.edu
Reflexes. Handout on The Basic Reflex Arc and Stretch and Tendon Reflexes. -55 mv -70 mv EPSP. By Noel Ways
 Reflexes Handout on The Basic Reflex Arc and Stretch and Tendon Reflexes By Noel Ways Basic Reflex Arch 2. : s are always unipolar and will conduct and impulse to a control center. In this case the control
Reflexes Handout on The Basic Reflex Arc and Stretch and Tendon Reflexes By Noel Ways Basic Reflex Arch 2. : s are always unipolar and will conduct and impulse to a control center. In this case the control
Involvement of phosphatase and tensin homolog deleted from chromosome 10 in rodent model of neuropathic pain
 Huang et al. Journal of Neuroinflammation (2015) 12:59 DOI 10.1186/s12974-015-0280-1 JOURNAL OF NEUROINFLAMMATION RESEARCH Open Access Involvement of phosphatase and tensin homolog deleted from chromosome
Huang et al. Journal of Neuroinflammation (2015) 12:59 DOI 10.1186/s12974-015-0280-1 JOURNAL OF NEUROINFLAMMATION RESEARCH Open Access Involvement of phosphatase and tensin homolog deleted from chromosome
Structural basis for the role of inhibition in facilitating adult brain plasticity
 Structural basis for the role of inhibition in facilitating adult brain plasticity Jerry L. Chen, Walter C. Lin, Jae Won Cha, Peter T. So, Yoshiyuki Kubota & Elly Nedivi SUPPLEMENTARY FIGURES 1-6 a b M
Structural basis for the role of inhibition in facilitating adult brain plasticity Jerry L. Chen, Walter C. Lin, Jae Won Cha, Peter T. So, Yoshiyuki Kubota & Elly Nedivi SUPPLEMENTARY FIGURES 1-6 a b M
The Nervous System S P I N A L R E F L E X E S
 The Nervous System S P I N A L R E F L E X E S Reflexes Rapid, involuntary, predictable motor response to a stimulus Spinal Reflexes Spinal somatic reflexes Integration center is in the spinal cord Effectors
The Nervous System S P I N A L R E F L E X E S Reflexes Rapid, involuntary, predictable motor response to a stimulus Spinal Reflexes Spinal somatic reflexes Integration center is in the spinal cord Effectors
Anesthesiology, V 100, No 4, Apr American Society of Anesthesiologists, Inc. Lippincott Williams & Wilkins, Inc.
 Anesthesiology 2004; 100:912 21 2004 American Society of Anesthesiologists, Inc. Lippincott Williams & Wilkins, Inc. Nerve Injury Induces a Tonic Bilateral -Opioid Receptor mediated Inhibitory Effect on
Anesthesiology 2004; 100:912 21 2004 American Society of Anesthesiologists, Inc. Lippincott Williams & Wilkins, Inc. Nerve Injury Induces a Tonic Bilateral -Opioid Receptor mediated Inhibitory Effect on
Pain Mechanisms. Prof Michael G Irwin MD, FRCA, FANZCA FHKAM Head Department of Anaesthesiology University of Hong Kong. The Somatosensory System
 ain Mechanisms rof Michael G Irwin MD, FRCA, FANZCA FHKAM Head Department of Anaesthesiology University of Hong Kong The Somatosensory System Frontal cortex Descending pathway eriaqueductal gray matter
ain Mechanisms rof Michael G Irwin MD, FRCA, FANZCA FHKAM Head Department of Anaesthesiology University of Hong Kong The Somatosensory System Frontal cortex Descending pathway eriaqueductal gray matter
Effects of Palonosetron, a 5-HT3 Receptor Antagonist, on Mechanical Allodynia in a Rat Model of Postoperative Pain
 Original Article Korean J Pain 2013 April; Vol. 26, No. 2: 125-129 pissn 2005-9159 eissn 2093-0569 http://dx.doi.org/10.3344/kjp.2013.26.2.125 Effects of Palonosetron, a 5-HT3 Receptor Antagonist, on Mechanical
Original Article Korean J Pain 2013 April; Vol. 26, No. 2: 125-129 pissn 2005-9159 eissn 2093-0569 http://dx.doi.org/10.3344/kjp.2013.26.2.125 Effects of Palonosetron, a 5-HT3 Receptor Antagonist, on Mechanical
Received 26 April 2004; revised 21 August 2004; accepted 20 September 2004 Available online 19 November 2004
 Experimental Neurology 191 (2005) 128 136 www.elsevier.com/locate/yexnr Ectopic discharges from injured nerve fibers are highly correlated with tactile allodynia only in early, but not late, stage in rats
Experimental Neurology 191 (2005) 128 136 www.elsevier.com/locate/yexnr Ectopic discharges from injured nerve fibers are highly correlated with tactile allodynia only in early, but not late, stage in rats
Pain classifications slow and fast
 Pain classifications slow and fast Fast Pain Slow Pain Sharp, pricking (Aδ) fiber Short latency Well localized Short duration Dull, burning (C) fiber Slower onset Diffuse Long duration Less emotional Emotional,
Pain classifications slow and fast Fast Pain Slow Pain Sharp, pricking (Aδ) fiber Short latency Well localized Short duration Dull, burning (C) fiber Slower onset Diffuse Long duration Less emotional Emotional,
Basolateral Amygdala Lesion Inhibits the Development of Pain Chronicity in Neuropathic Pain Rats
 Basolateral Amygdala Lesion Inhibits the Development of Pain Chronicity in Neuropathic Pain Rats Zheng Li 1,3., Jing Wang 2., Lin Chen 1,3, Meng Zhang 1,3, You Wan 1,3 * 1 Neuroscience Research Institute,
Basolateral Amygdala Lesion Inhibits the Development of Pain Chronicity in Neuropathic Pain Rats Zheng Li 1,3., Jing Wang 2., Lin Chen 1,3, Meng Zhang 1,3, You Wan 1,3 * 1 Neuroscience Research Institute,
Role of CB1 and CB2 cannabinoid receptors in the development of joint pain. induced by monosodium iodoacetate
 *Manuscript 1 1 1 1 1 1 1 Role of CB1 and CB cannabinoid receptors in the development of joint pain induced by monosodium iodoacetate Carmen La Porta 1*, Simona Andreea Bura 1*, Auxiliadora Aracil-Fernández,
*Manuscript 1 1 1 1 1 1 1 Role of CB1 and CB cannabinoid receptors in the development of joint pain induced by monosodium iodoacetate Carmen La Porta 1*, Simona Andreea Bura 1*, Auxiliadora Aracil-Fernández,
PAIN MANAGEMENT in the CANINE PATIENT
 PAIN MANAGEMENT in the CANINE PATIENT Laurie Edge-Hughes, BScPT, MAnimSt (Animal Physio), CAFCI, CCRT Part 1: Laurie Edge-Hughes, BScPT, MAnimSt (Animal Physio), CAFCI, CCRT 1 Pain is the most common reason
PAIN MANAGEMENT in the CANINE PATIENT Laurie Edge-Hughes, BScPT, MAnimSt (Animal Physio), CAFCI, CCRT Part 1: Laurie Edge-Hughes, BScPT, MAnimSt (Animal Physio), CAFCI, CCRT 1 Pain is the most common reason
MANAGING CHRONIC PAIN
 George Hardas MANAGING CHRONIC PAIN The guide to understanding chronic pain and how to manage it. George Hardas MMed (UNSW) MScMed (Syd) MChiro (Macq) BSc (Syd) Grad Cert Pain Management (Syd) Cognitive
George Hardas MANAGING CHRONIC PAIN The guide to understanding chronic pain and how to manage it. George Hardas MMed (UNSW) MScMed (Syd) MChiro (Macq) BSc (Syd) Grad Cert Pain Management (Syd) Cognitive
SUPPLEMENTARY INFORMATION
 METHODS Mice and rats. Behavioural experiments were performed in male and female 7 12 week old mice or in male 7 10 week old Wistar rats. Permissions for the animal experiments have been obtained from
METHODS Mice and rats. Behavioural experiments were performed in male and female 7 12 week old mice or in male 7 10 week old Wistar rats. Permissions for the animal experiments have been obtained from
Victor Chaban, Ph.D., MSCR
 Hormonal Modulation of Pain : Estrogen Modulation of Visceral Nociception Victor Chaban, Ph.D., MSCR Department of Internal Medicine Charles R. Drew University of Medicine and Science Department of Medicine
Hormonal Modulation of Pain : Estrogen Modulation of Visceral Nociception Victor Chaban, Ph.D., MSCR Department of Internal Medicine Charles R. Drew University of Medicine and Science Department of Medicine
Analgesic effects of the COX-2 inhibitor parecoxib on surgical pain through suppression of spinal ERK signaling
 EXPERIMENTAL AND THERAPEUTIC MEDICINE 6: 275-279, 2013 Analgesic effects of the COX-2 inhibitor parecoxib on surgical pain through suppression of spinal ERK signaling YA-JING GUO 1, XU-DAN SHI 2, DI FU
EXPERIMENTAL AND THERAPEUTIC MEDICINE 6: 275-279, 2013 Analgesic effects of the COX-2 inhibitor parecoxib on surgical pain through suppression of spinal ERK signaling YA-JING GUO 1, XU-DAN SHI 2, DI FU
Changes in long2term syna ptic plasticity in the spinal dorsal horn of neuropathic pa in rats
 226 ( ) JOURNAL OF PEKIN G UNIVERSITY( HEAL TH SCIENCES) Vol. 35 No. 3 J une 2003,,, (, 100083) [ ] / ; ; ; / [ ] :, (central sensitization) :Sprague2Dawley 5/ 6 (L5/ L6), C2, C2 L TP : (1), ( 100 Hz,
226 ( ) JOURNAL OF PEKIN G UNIVERSITY( HEAL TH SCIENCES) Vol. 35 No. 3 J une 2003,,, (, 100083) [ ] / ; ; ; / [ ] :, (central sensitization) :Sprague2Dawley 5/ 6 (L5/ L6), C2, C2 L TP : (1), ( 100 Hz,
CHAPTER 10 THE SOMATOSENSORY SYSTEM
 CHAPTER 10 THE SOMATOSENSORY SYSTEM 10.1. SOMATOSENSORY MODALITIES "Somatosensory" is really a catch-all term to designate senses other than vision, hearing, balance, taste and smell. Receptors that could
CHAPTER 10 THE SOMATOSENSORY SYSTEM 10.1. SOMATOSENSORY MODALITIES "Somatosensory" is really a catch-all term to designate senses other than vision, hearing, balance, taste and smell. Receptors that could
Repeated intra-articular injections of acidic saline produce long-lasting joint pain and. widespread hyperalgesia
 Repeated intra-articular injections of acidic saline produce long-lasting joint pain and widespread hyperalgesia N. Sugimura 1, M. Ikeuchi 1 *, M. Izumi 1, T. Kawano 2, K. Aso 1, T. Kato 1, T. Ushida 3,
Repeated intra-articular injections of acidic saline produce long-lasting joint pain and widespread hyperalgesia N. Sugimura 1, M. Ikeuchi 1 *, M. Izumi 1, T. Kawano 2, K. Aso 1, T. Kato 1, T. Ushida 3,
Possible Involvement of Brain-Derived Neurotrophic Factor in Analgesic Effects of 4-Methylcatechol on Neuropathic Pain
 49 Bull Yamaguchi Med School 57 3-4 :49-56, 2010 Possible Involvement of Brain-Derived Neurotrophic Factor in Analgesic Effects of 4-Methylcatechol on Neuropathic Pain Kozo Ishikawa 2 Department of Emergency
49 Bull Yamaguchi Med School 57 3-4 :49-56, 2010 Possible Involvement of Brain-Derived Neurotrophic Factor in Analgesic Effects of 4-Methylcatechol on Neuropathic Pain Kozo Ishikawa 2 Department of Emergency
Clarke's Column Neurons as the Focus of a Corticospinal Corollary Circuit. Supplementary Information. Adam W. Hantman and Thomas M.
 Clarke's Column Neurons as the Focus of a Corticospinal Corollary Circuit Supplementary Information Adam W. Hantman and Thomas M. Jessell Supplementary Results Characterizing the origin of primary
Clarke's Column Neurons as the Focus of a Corticospinal Corollary Circuit Supplementary Information Adam W. Hantman and Thomas M. Jessell Supplementary Results Characterizing the origin of primary
Module H NERVOUS SYSTEM
 Module H NERVOUS SYSTEM Topic from General functions of the nervous system Organization of the nervous system from both anatomical & functional perspectives Gross & microscopic anatomy of nervous tissue
Module H NERVOUS SYSTEM Topic from General functions of the nervous system Organization of the nervous system from both anatomical & functional perspectives Gross & microscopic anatomy of nervous tissue
Spinal morphine but not ziconotide or gabapentin analgesia is affected by alternative splicing of voltage-gated calcium channel CaV2.
 City University of New York (CUNY) CUNY Academic Works Publications and Research City College of New York December 13 Spinal morphine but not ziconotide or gabapentin analgesia is affected by alternative
City University of New York (CUNY) CUNY Academic Works Publications and Research City College of New York December 13 Spinal morphine but not ziconotide or gabapentin analgesia is affected by alternative
effect on the upregulation of these cell surface markers. The mean peak fluorescence intensity
 SUPPLEMENTARY FIGURE 1 Supplementary Figure 1 ASIC1 disruption or blockade does not effect in vitro and in vivo antigen-presenting cell activation. (a) Flow cytometric analysis of cell surface molecules
SUPPLEMENTARY FIGURE 1 Supplementary Figure 1 ASIC1 disruption or blockade does not effect in vitro and in vivo antigen-presenting cell activation. (a) Flow cytometric analysis of cell surface molecules
Proceedings of the World Small Animal Veterinary Association Sydney, Australia 2007
 Proceedings of the World Small Animal Sydney, Australia 2007 Hosted by: Next WSAVA Congress REDUCING THE PAIN FACTOR AN UPDATE ON PERI-OPERATIVE ANALGESIA Sandra Forysth, BVSc DipACVA Institute of Veterinary,
Proceedings of the World Small Animal Sydney, Australia 2007 Hosted by: Next WSAVA Congress REDUCING THE PAIN FACTOR AN UPDATE ON PERI-OPERATIVE ANALGESIA Sandra Forysth, BVSc DipACVA Institute of Veterinary,
Pain. Pain. Pain: One definition. Pain: One definition. Pain: One definition. Pain: One definition. Psyc 2906: Sensation--Introduction 9/27/2006
 Pain Pain Pain: One Definition Classic Paths A new Theory Pain and Drugs According to the international Association for the Study (Merskey & Bogduk, 1994), Pain is an unpleasant sensory and emotional experience
Pain Pain Pain: One Definition Classic Paths A new Theory Pain and Drugs According to the international Association for the Study (Merskey & Bogduk, 1994), Pain is an unpleasant sensory and emotional experience
Special Issue on Pain and Itch
 Special Issue on Pain and Itch Title: Recent Progress in Understanding the Mechanisms of Pain and Itch Guest Editor of the Special Issue: Ru-Rong Ji, PhD Chronic pain is a major health problem world-wide.
Special Issue on Pain and Itch Title: Recent Progress in Understanding the Mechanisms of Pain and Itch Guest Editor of the Special Issue: Ru-Rong Ji, PhD Chronic pain is a major health problem world-wide.
Chapter 11 Introduction to the Nervous System and Nervous Tissue Chapter Outline
 Chapter 11 Introduction to the Nervous System and Nervous Tissue Chapter Outline Module 11.1 Overview of the Nervous System (Figures 11.1-11.3) A. The nervous system controls our perception and experience
Chapter 11 Introduction to the Nervous System and Nervous Tissue Chapter Outline Module 11.1 Overview of the Nervous System (Figures 11.1-11.3) A. The nervous system controls our perception and experience
Neuroscience with Pharmacology 2 Functions and Mechanisms of Reflexes. Prof Richard Ribchester
 Neuroscience with Pharmacology 2 Functions and Mechanisms of Reflexes Prof Richard Ribchester René Descartes Cogito, ergo sum The 21st century still holds many challenges to Neuroscience and Pharmacology
Neuroscience with Pharmacology 2 Functions and Mechanisms of Reflexes Prof Richard Ribchester René Descartes Cogito, ergo sum The 21st century still holds many challenges to Neuroscience and Pharmacology
Variety of muscle responses to tactile stimuli
 Variety of muscle responses to tactile stimuli Julita Czarkowska-Bauch Department of Neurophysiology, Nencki Institute of Experimental Biology, 3 Pasteur St., 02-093 Warsaw, Poland Abstract. Influences
Variety of muscle responses to tactile stimuli Julita Czarkowska-Bauch Department of Neurophysiology, Nencki Institute of Experimental Biology, 3 Pasteur St., 02-093 Warsaw, Poland Abstract. Influences
Yanbing He 1, 2, Shiyuan Xu 1, Junjie Huang 2, Qingjuan Gong 2. NEURAL REGENERATION RESEARCH May 2014,Volume 9,Issue 10
 NEURAL REGENERATION RESEARCH May 14,Volume 9,Issue 1 www.nrronline.org Analgesic effect of intrathecal bumetanide is accompanied by changes in spinal sodium-potassium-chloride co-transporter 1 and potassium-chloride
NEURAL REGENERATION RESEARCH May 14,Volume 9,Issue 1 www.nrronline.org Analgesic effect of intrathecal bumetanide is accompanied by changes in spinal sodium-potassium-chloride co-transporter 1 and potassium-chloride
Department of Neurology/Division of Anatomical Sciences
 Spinal Cord I Lecture Outline and Objectives CNS/Head and Neck Sequence TOPIC: FACULTY: THE SPINAL CORD AND SPINAL NERVES, Part I Department of Neurology/Division of Anatomical Sciences LECTURE: Monday,
Spinal Cord I Lecture Outline and Objectives CNS/Head and Neck Sequence TOPIC: FACULTY: THE SPINAL CORD AND SPINAL NERVES, Part I Department of Neurology/Division of Anatomical Sciences LECTURE: Monday,
NNZ-2566 in Rett Syndrome and Autism Spectrum Disorders Role and Update
 NNZ-2566 in Rett Syndrome and Autism Spectrum Disorders Role and Update 1 Overview The natural growth factor IGF-1 is broken down in the body to IGF-1[1-3] NNZ-2566 is an analogue of IGF-1[1-3] developed
NNZ-2566 in Rett Syndrome and Autism Spectrum Disorders Role and Update 1 Overview The natural growth factor IGF-1 is broken down in the body to IGF-1[1-3] NNZ-2566 is an analogue of IGF-1[1-3] developed
ANAT2010. Concepts of Neuroanatomy (II) S2 2018
 ANAT2010 Concepts of Neuroanatomy (II) S2 2018 Table of Contents Lecture 13: Pain and perception... 3 Lecture 14: Sensory systems and visual pathways... 11 Lecture 15: Techniques in Neuroanatomy I in vivo
ANAT2010 Concepts of Neuroanatomy (II) S2 2018 Table of Contents Lecture 13: Pain and perception... 3 Lecture 14: Sensory systems and visual pathways... 11 Lecture 15: Techniques in Neuroanatomy I in vivo
Brian Kahan, D.O. FAAPMR, DABPM, DAOCRM, FIPP Center for Pain Medicine and Physiatric Rehabilitation 2002 Medical Parkway Suite 150 Annapolis, MD
 Brian Kahan, D.O. FAAPMR, DABPM, DAOCRM, FIPP Center for Pain Medicine and Physiatric Rehabilitation 2002 Medical Parkway Suite 150 Annapolis, MD 1630 Main Street Suite 215 Chester, MD 410-571-9000 www.4-no-pain.com
Brian Kahan, D.O. FAAPMR, DABPM, DAOCRM, FIPP Center for Pain Medicine and Physiatric Rehabilitation 2002 Medical Parkway Suite 150 Annapolis, MD 1630 Main Street Suite 215 Chester, MD 410-571-9000 www.4-no-pain.com
Materials and Methods
 Anesthesiology 2004; 100:1258 62 2004 American Society of Anesthesiologists, Inc. Lippincott Williams & Wilkins, Inc. Spinal Adenosine Receptor Activation Reduces Hypersensitivity after Surgery by a Different
Anesthesiology 2004; 100:1258 62 2004 American Society of Anesthesiologists, Inc. Lippincott Williams & Wilkins, Inc. Spinal Adenosine Receptor Activation Reduces Hypersensitivity after Surgery by a Different
Exercise 1: (5pts) Exercise 2: (5pts) Observe the following diagram and answer the questions below.
 S.E. Biology Exercise 1: (5pts) Observe the following diagram and answer the questions below. 1. Give a suitable title for the given schematic drawing. 2. Indicate the type of the stimulus, the conductor,
S.E. Biology Exercise 1: (5pts) Observe the following diagram and answer the questions below. 1. Give a suitable title for the given schematic drawing. 2. Indicate the type of the stimulus, the conductor,
Yan-Ping Chen, Shao-Rui Chen, and Hui-Lin Pan
 0022-3565/05/3152-696 703$20.00 THE JOURNAL OF PHARMACOLOGY AND EXPERIMENTAL THERAPEUTICS Vol. 315, No. 2 Copyright 2005 by The American Society for Pharmacology and Experimental Therapeutics 91314/3054541
0022-3565/05/3152-696 703$20.00 THE JOURNAL OF PHARMACOLOGY AND EXPERIMENTAL THERAPEUTICS Vol. 315, No. 2 Copyright 2005 by The American Society for Pharmacology and Experimental Therapeutics 91314/3054541
Phenotypic Analysis of Mice Deficient in the Type 2 Galanin Receptor (GALR2)
 MOLECULAR AND CELLULAR BIOLOGY, June 2005, p. 4804 4811 Vol. 25, No. 11 0270-7306/05/$08.00 0 doi:10.1128/mcb.25.11.4804 4811.2005 Copyright 2005, American Society for Microbiology. All Rights Reserved.
MOLECULAR AND CELLULAR BIOLOGY, June 2005, p. 4804 4811 Vol. 25, No. 11 0270-7306/05/$08.00 0 doi:10.1128/mcb.25.11.4804 4811.2005 Copyright 2005, American Society for Microbiology. All Rights Reserved.
Curcumin Attenuates Mechanical and Thermal Hyperalgesia in Chronic Constrictive Injury Model of Neuropathic Pain
 Pain Ther (2014) 3:59 69 DOI 10.1007/s40122-014-0024-4 ORIGINAL RESEARCH Curcumin Attenuates Mechanical and Thermal Hyperalgesia in Chronic Constrictive Injury Model of Neuropathic Pain Yu Xiang Di Cao
Pain Ther (2014) 3:59 69 DOI 10.1007/s40122-014-0024-4 ORIGINAL RESEARCH Curcumin Attenuates Mechanical and Thermal Hyperalgesia in Chronic Constrictive Injury Model of Neuropathic Pain Yu Xiang Di Cao
Diabetic Complications Consortium
 Diabetic Complications Consortium Application Title: Cathepsin S inhibition and diabetic neuropathy Principal Investigator: Nigel A Calcutt 1. Project Accomplishments: We investigated the efficacy of cathepsin
Diabetic Complications Consortium Application Title: Cathepsin S inhibition and diabetic neuropathy Principal Investigator: Nigel A Calcutt 1. Project Accomplishments: We investigated the efficacy of cathepsin
susceptibility of either the axons in the dorsal and ventral roots, or the intramedullary
 213 J. Physiol. (31958) I40, 2I3-2I9 THE SITE OF ACTION OF PROCAINE ON THE ISOLATED SPINAL CORD OF THE FROG BY M. HARMEL AND J. L. MALCOLM From the Department of Physiology, State University of New York,
213 J. Physiol. (31958) I40, 2I3-2I9 THE SITE OF ACTION OF PROCAINE ON THE ISOLATED SPINAL CORD OF THE FROG BY M. HARMEL AND J. L. MALCOLM From the Department of Physiology, State University of New York,
Dissecting out mechanisms responsible for peripheral neuropathic pain: Implications for diagnosis and therapy
 Life Sciences 74 (2004) 2605 2610 www.elsevier.com/locate/lifescie Dissecting out mechanisms responsible for peripheral neuropathic pain: Implications for diagnosis and therapy Clifford J. Woolf* Neural
Life Sciences 74 (2004) 2605 2610 www.elsevier.com/locate/lifescie Dissecting out mechanisms responsible for peripheral neuropathic pain: Implications for diagnosis and therapy Clifford J. Woolf* Neural
Sensory Assessment of Regional Analgesia in Humans
 REVIEW ARTICLE Dennis M. Fisher, M.D., Editor-in-Chief Anesthesiology 2000; 93:1517 30 2000 American Society of Anesthesiologists, Inc. Lippincott Williams & Wilkins, Inc. Sensory Assessment of Regional
REVIEW ARTICLE Dennis M. Fisher, M.D., Editor-in-Chief Anesthesiology 2000; 93:1517 30 2000 American Society of Anesthesiologists, Inc. Lippincott Williams & Wilkins, Inc. Sensory Assessment of Regional
Enhanced hippocampal noradrenaline and serotonin release in galanin-overexpressing mice after repeated forced swimming test
 Enhanced hippocampal noradrenaline and serotonin release in galanin-overexpressing mice after repeated forced swimming test Takashi Yoshitake*, Fu-Hua Wang*, Eugenia Kuteeva*, Kristina Holmberg*, Masatoshi
Enhanced hippocampal noradrenaline and serotonin release in galanin-overexpressing mice after repeated forced swimming test Takashi Yoshitake*, Fu-Hua Wang*, Eugenia Kuteeva*, Kristina Holmberg*, Masatoshi
Mechanism of Pain Production
 Mechanism of Pain Production Pain conducting nerve fibers are small myelinated (A-delta) or unmyelinated nerve fibers (C-fibers). Cell bodies are in the dorsal root ganglia (DRG) or sensory ganglia of
Mechanism of Pain Production Pain conducting nerve fibers are small myelinated (A-delta) or unmyelinated nerve fibers (C-fibers). Cell bodies are in the dorsal root ganglia (DRG) or sensory ganglia of
TNF-a/TNFR1 Signaling Is Required for the Development and Function of Primary Nociceptors
 Article TNF-a/TNFR1 Signaling Is Required for the Development and Function of Primary Nociceptors Michael A. Wheeler, 1,2 Danielle L. Heffner, 1 Suemin Kim, 1 Sarah M. Espy, 1 Anthony J. Spano, 1 Corey
Article TNF-a/TNFR1 Signaling Is Required for the Development and Function of Primary Nociceptors Michael A. Wheeler, 1,2 Danielle L. Heffner, 1 Suemin Kim, 1 Sarah M. Espy, 1 Anthony J. Spano, 1 Corey
Preserved Acute Pain and Reduced Neuropathic Pain in Mice Lacking PKC
 REPORTS cm length of PE-10 tubing (inner diameter, 0.28 mm; outer diameter, 0.61 mm), serving as an inflow cannula, was inserted into the subarachnoid space through an incision in the atlanto-occipital
REPORTS cm length of PE-10 tubing (inner diameter, 0.28 mm; outer diameter, 0.61 mm), serving as an inflow cannula, was inserted into the subarachnoid space through an incision in the atlanto-occipital
Neurotrophic in uences on neuropathic pain
 Neurotrophic in uences on neuropathic pain Stephen B. McMahon and William B. J. Ca erty Centre for Neuroscience Research, King s College London, Guy s Campus, Hodgkin Building, London SE1 1UL, UK Abstract.
Neurotrophic in uences on neuropathic pain Stephen B. McMahon and William B. J. Ca erty Centre for Neuroscience Research, King s College London, Guy s Campus, Hodgkin Building, London SE1 1UL, UK Abstract.
Department of Orthopaedic Surgery, Tohoku University Graduate School of Medicine, Sendai, Japan, 2
 Low-energy Extracorporeal Shock Wave Therapy Promotes VEGF Expression and Angiogenesis and Improve Locomotor and Sensory Functions after spinal cord injury Kenichiro Yahata 1, Hiroshi Ozawa, M.D., Ph.D.
Low-energy Extracorporeal Shock Wave Therapy Promotes VEGF Expression and Angiogenesis and Improve Locomotor and Sensory Functions after spinal cord injury Kenichiro Yahata 1, Hiroshi Ozawa, M.D., Ph.D.
Byeol-Rim Kang and Chang-Beohm Ahn Department of Acupuncture and Moxibustion, College of Oriental Medicine Dong-Eui University, Busan , Korea
 The American Journal of Chinese Medicine, Vol. 35, No. 6, 987 993 2007 World Scientific Publishing Company Institute for Advanced Research in Asian Science and Medicine N-Methyl-D-Aspartate Antagonist
The American Journal of Chinese Medicine, Vol. 35, No. 6, 987 993 2007 World Scientific Publishing Company Institute for Advanced Research in Asian Science and Medicine N-Methyl-D-Aspartate Antagonist
