Barosensory Neurons in the Ventrolateral Medulla in Rabbits and Their Responses to Various Afferent Inputs from Peripheral and Central Sources
|
|
|
- Millicent Robertson
- 6 years ago
- Views:
Transcription
1 Japanese Journal of Physiology, 36, , 1986 Barosensory Neurons in the Ventrolateral Medulla in Rabbits and Their Responses to Various Afferent Inputs from Peripheral and Central Sources Naohito TERUI, Yuka SA1 I, and Mamoru KUMADA Institute of Basic Medical Sciences, The University of Tsukuba, Ibaraki, 305 Japan Abstract In 55 anesthetized and paralyzed adult rabbits, 161 spontaneously active neurons which responded to electrical stimulation of A- fibers of the aortic nerve were found within the ventrolateral medulla (VLM). They were termed barosensory VLM neurons, since the aortic nerve A-fibers were considered to consist exclusively of afferents from arterial baroreceptors. Forty percent of barosensory VLM neurons tested (49/123) were activated antidromically by stimulation of the dorsolateral funiculus indicating that they send descending bulbospinal projections. Spontaneous discharges of barosensory VLM neurons were invariably inhibited by stimulation of aortic nerve A-fibers. Ninety-three percent of 80 neurons tested also responded to stimulation of aortic nerve C-fibers, a mixture of barosensory and nonbarosensory afferents. Natural stimulation of carotid sinus baroreceptors by an intravenous injection of phenylephrine in 19 vagotomized rabbits with aortic nerves disrupted inhibited spontaneous activity of all the 50 barosensory VLM neurons tested. By contrast, pharmacological stimulation of right or left carotid body chemoreceptors by close arterial injection of NaCN into the carotid sinus augmented activity of 93% of barosensory VLM neurons tested (41/44). The neuronal response was always greater to stimulation of chemoreceptors in the contralateral carotid sinus. Seven out of 8 barosensory VLM neurons tested (88%) were orthodromically excited by stimulation of the posterior hypothalamic area. In 74% of the 97 neurons examined in 29 vagotomized animals, a distinct respiratory-related rhythm, locked to that of phrenic nerve activity, was discerned. Thus, spontaneous activity of barosensory VLM neurons is inhibited by afferent inputs from aortic and carotid sinus baroreceptors, but is excited by incoming signals from carotid body chemoreceptors and the posterior hypothalamic area. It is also subject to the influence of the central mechanism generating the respiratory rhythm. Key words : rabbit, barosensory neuron, ventrolateral medulla, aortic Received for publication August 12,
2 1142 N. TERUI, Y. SAEKI, and M. KUMADA nerve, nonbarosensory input. The ventrolateral medulla is a circumscribed region near the ventral surface of the medulla extending approximately between the caudal portion of the motor nucleus of the facial nerve and the rostral third of the inferior olive (DAMPNEY et al., 1982; CAVERSON et al., 1983: Ross et al., 1984a). In cats (TABER,1961; LovICK,1986) and rats (ANDREZIK et al., 1981; LOVICK and HILTON, 1985), it roughly corresponds to the nucleus paragigantocellularis lateralis. In rabbits, it occupies an area coincident with the nucleus reticularis lateralis as termed by MEESEN and OLSZEWSKI (1949). Descending bulbospinal pathways originate from neurons in the ventrolateral medulla and project to the region of the intermediolateral cell column (AMENDT et al., 1979; CAVERSON et al., 1983; Ross et al., 1984b), the major site of sympathetic preganglionic neurons. It has been proposed that neurons in this area play a critical role in the maintenance of the basal level of arterial pressure as well as in the reflex control of it through tonic and reflex control of sympathetic vasomotor discharges (DAMPNEY and MooN, 1980; MCALLEN et al., 1982; REIS et al., 1984; Ross et al., 1984a; GRANATA et al., 1985; TERUI et al., 1986). The purpose of the present study conducted on anesthetized adult rabbits is to delineate the role of neurons in the ventrolateral medulla in integrating barosensory and nonbarosensory afferent inputs from various peripheral and central sources. We first identified spontaneously active units within the ventrolateral medulla which responded to electrical stimulation of A-fibers of the aortic nerve. Since the aortic nerve A-fibers of adult rabbits are considered to consist of afferents from aortic baroreceptors (NEIL et al., 1949; CHALMERS et al., 1967; AARs, 1971; NUMAO et al., 1983), it is possible to activate selectively those neurons involved in the arterial baroreceptor reflex. Such neurons, termed barosensory VLM neurons, were then tested for their descending projections by antidromic activation. The barosensory VLM neurons were further examined for their response to natural, pharmacological or electrical stimulation of various receptors and peripheral and central neural structures. MATERIALS AND METHODS Preparation of animals. Experiments were performed on 55 adult albino rabbits (New Zealand White) of both sexes weighing between 2.7 and 3.7 kg. They were anesthetized with an intravenous administration of urethane (1 g/kg). After insertion of tracheal, arterial and venous cannulas, the animal was paralyzed with pancuronium bromide (initially 2 mg, iv., thereafter mg/h, i.v.) and artificially ventilated by a respiratory pump (Harvard, 661) with a gas mixture of 20% 02 and 80% room air. The end-expiratory CO2 was constantly monitored (Beckman, LB-2) and was maintained at %. Arterial Pco2 was between 25 Japanese Journal of Physiology
3 BAROSENSORY NEURONS IN VENTROLATERAL MEDULLA 1143 and 35 mmhg and arterial pot was above 100 mmhg (measured in 20 animals by Corning gas analyzer 165/2). Body temperature was maintained at C by a thermostatically regulated heating pad connected to a rectal thermistor probe. After preparatory surgical procedures, the animal was fixed to a stereotaxic frame in a prone position with the head tilted forwards at an angle of 45 degrees. The floor of the IVth ventricle was exposed near the obex and covered by a layer of agar jelly (3 /s in saline). A pneumothorax was induced in all the animals in order to assure a stable recording of single unit activity in the brainstem. In 30 rabbits, in which single unit discharges were examined for a respiratory-related rhythm or for their response to a rise in arterial pressure, the vagus nerve was cut in the neck on both sides. Measurement of cardiovascular variables and renal and phrenic nerve activities. Instantaneous and mean arterial pressures and heart rate were monitored continuously in all experiments. Arterial pressure was recorded from the abdominal aorta by a polyethylene catheter inserted through the femoral artery and connected to a transducer (Nihon Kohden, MPU-0.5). Heart rate was computed from the arterial pressure pulse by a tachometer (Nihon Kohden, AT-600G). The left renal nerve was approached retroperitoneally through a left flank incision, and prepared for recording from near the renal artery. The central cut end of the nerve was placed on a pair of bipolar platinum electrodes connected to an amplifier and displayed on an oscilloscope (Tektronix, 5113). The lower and higher cut-off frequencies of the recording system were 100 and 5,000 Hz, respectively. The multifiber renal nerve activity (RNA) was converted into standard pulses by a window discriminator (Nihon Kohden, EN-601J), the threshold of which was set slightly above the noise level. The discharge frequency was counted by a frequency counter with a bin width of 1 s then displayed on, a polygraph. The right phrenic nerve was prepared in the neck, and multifiber phrenic nerve activity was recorded and processed either in the same manner as RNA or by rectifying and then integrating it at a time constant of 50 or 100 ms. Recording of single unit activity and the area explored. Single unit activity of neurons in the left ventrolateral medulla was recorded by micropipettes filled with 0.5 M sodium acetate and pontamine sky blue for marking and mounted in a hydraulic micro-manipulator (Narishige, MO-81). The tip resistance of the electrode was between 2 and 5 megohms. Single unit activity was amplified (Nihon Kohden, MEZ-8201) with reference to a ground plate attached to a neck muscle and was displayed on an oscilloscope. The same signal was simultaneously fed into a channel of a tape recorder (TEAL, 31R) together with RNA, phrenic nerve activity, ECG (lead II), instantaneous arterial pressure and timing pulses of stimulation. The site of unit recording was stained by expelling the potamine sky blue from the electrode by the passage of 20-50µA current (electrode negative) for about 10 min. Stereotaxic coordinates of the point of entry were determined visually with reference to the rostra! margin of the area postrema in the midline, i.e., obex, which was defined as the stereotaxic zero. The area explored for single unit recording Vol. 36, No. 6, 1986
4 1144 N. TERUI, Y. SAEKI, and M. KUMADA extended between 2 and 4 mm to the left of the midsagittal plane, and between 0 and 2.5 mm above the ventral surface of the brainstem. There, spontaneously active single units which responded to electrical stimulation of the right and/or left aortic nerves were systematically searched. When such a unit was identified, it was subsequently tested for responses to one or more of the following maneuvers. (1) Electrical stimulation of the right or left dorsolateral funiculus of the spinal cord for antidromic activation, (2) natural stimulation of arterial baroreceptors by an intravenous injection of phenylephrine, (3) pharmacological stimulation of right and/or left carotid body chemoreceptors by a close arterial injection of sodium cyanide, and (4) electrical stimulation of the posterior hypothalamic area for orthodromic activation of medullary units. When electrical stimulation was involved, the evoked response of the neuronal activity was summed, together with RNA and phrenic nerve activity, over successive sweeps with a data processing computer (Nihon Kohden, ATAC 450) to obtain a peristimulus time histogram. Furthermore, using the same summation technique, spontaneous discharges were examined for their cardiac-related and respiratory-related rhythms. Stimulation of the aortic nerve, arterial baroreceptors, and carotid body chemoreceptors. Both right and left aortic nerves were identified in the neck and prepared for electrical stimulation. The distal cut end of the nerve was placed across a pair of platinum electrodes spaced 2-3 mm apart. Electrical stimuli of the aortic nerve were square-wave pulses of ms duration delivered to the animal from pulse generator (Nihon Kohden, SEN 7103) through an isolation unit. To activate aortic nerve A-fibers selectively, the stimulus intensity was set at V. One to 10 pulses spaced by 10 ms between them were applied to the nerve to elicit the A-fiber reflex. To activate aortic nerve C-fibers as well, the stimulus intensity was set at 7-10 V with the pulse width of 0.5 ms (NUMAO et al., 1983). A single shock was given to the nerve to elicit the reflex response ascribable to C-fibers. Natural stimulation of arterial baroreceptors consisted of an intravenous bolus injection of phenylephrine (40,tg/ml) in a volume of ml of saline over 2s. To stimulate carotid body chemoreceptors, the cranial thyroid artery was isolated on both sides and cannulated with a polyethylene catheter (PE-10). The tip of the catheter was advanced into the common carotid artery until it lay close to the bifurcation. A close intraarterial injection through the right or left thyroid artery of 5-10 µg of sodium cyanide (WAKO, Osaka) in a volume of 0.2 ml of saline over 2 s served as stimulation of carotid body chemoreceptors. Electrical stimulation of the dorsolateral funiculus of the spinal cord and the posterior hypothalamic area. To examine whether or not neurons in the ventrolateral medulla sent descending bulbospinal projections, they were tested for antidromic activation by electrically stimulating the right or left dorsolateral funiculus at the second cervical cord. Unipolar stimulating electrodes were placed in the dorsolateral funiculus of both sides under a direct vision, following a laminectomy at the second cervical cord. The electrode was made from stainless-steel wires (250,tm in diameter) with the tips electrolytically sharpened. It was insulated except Japanese Journal of Physiology
5 BAROSENSORY NEURONS IN VENTROLATERAL MEDULLA 1145 Fig. 1. Locations, within the dorsolateral funiculus, of electrical stimulation to activate neurons in the left ventrolateral medulla antidromically. The left panel illustrates evoked response of RNA elicited by electrical stimulation of 4 points, 0.5 mm apart, along a single electrode track penetrating the dorsolateral funiculus at the C2 level. Stimulus consisted of 3 rectangular pulses of 0.2 ms duration at 100 Hz with the intensity of 50 µa. It was applied at the time indicated by arrowheads. Each peristimulus histogram was the sum of 32 successive responses. In this and subsequent figures, the bin width was 5 ms unless otherwise noted. On the right-half of the transverse section of the spinal cord were shown locations of histologically verified sites of effective stimulation in the region of the dorsolateral funiculus for antidromic excitation of barosensory VLM neurons. Although stimulus sites were located on both sides, they were all shown on the right side. Those on the left half of the spinal cord were represented by corresponding sites on the other side. at the very tip which was left bare for 0.3 mm. The stimulus site was determined such that the maximum excitation of RNA was elicited on stimulation. Electrodes were inserted just lateral to the dorsal root entry and mm below the dorsal surface of the spinal cord (Fig. 1). With a fixed pulse duration of 0.2 ms, the stimulus intensity was adjusted times of the thresholds for antidromic activation of the unit ( ma, but mostly below 0.3 ma). Stimulation was monopolar, the anode being a silver plate attached to the neck muscle. That descending bulbospinal fibers were activated by this stimulation was confirmed in 5 rabbits by the unchanged onset latency of the evoked response of RNA before and after a total spinal transection at the first cervical cord. For identification of stimulating sites, they were marked by passing a direct current of 50 µa for 10 s in order to make a small lesion at the electrode tip. Reasons for stimulating the dorsolateral funiculus for antidromic excitation of neurons in the ventrolateral Vol. 36, No. 6, 1986
6 1146 N. TERUI, Y. SAEKI, and M. KUMADA medulla, rather than the intermediolateral cell column as in some studies (CAVERSON et al., 1983; BROWN and GUYENET, 1985), are described in detail in the DIS- CUSSION section. The posterior hypothalamic area was stimulated on the left side by a monopolar electrode, as described above, inserted vertically into the brain through a hole drilled in the frontal bone about 8.5 mm caudal to the bregma about 1 mm lateral to the midline. Referring to the atlas of SAWYER et al. (1954), the stimulus site was determined by the large pressor response (up to 70 mmhg) produced by electrical stimulation with a 10 s train of negative square-wave pulses of 0.2 ms duration at a stimulus frequency of 100 Hz. It was determined post mortem as with the case of the spinal cord. Histological examination. At the end of each experiment, the animal was perfused with saline followed by 10% formaline. The brain and spinal cord were fixed, frozen and sectioned at 50 µm. The sites of dye or a small electrical lesion, produced by electrical current as described above, were identified and related to histological structures before and after staining the section with cresyl violet or neutral red. RESULTS Identification and distribution of barosensory VLM neurons As the result of a systematic survey, within the left ventrolateral medulla (VLM), of spontaneously active neurons which responded to stimulation of aortic nerve A-fibers, 161 barosensory VLM neurons were recorded so far from 55 rabbits (Fig. 2). They were located within the ventrolateral quadrant of the medulla oblongata mostly ventral to the nucleus ambiguus between the levels of the inferior olivary nucleus and the caudal portion of the motor nucleus of the facial nerve (Fig. 3A). Such distribution corresponds very closely to that of the ventrolateral group of the pressor points in the medulla oblongata of rabbits and cats (DAMPNEY et al., 1982; LovicK and HILTON, 1985; MCALLEN, 1986). The spontaneous discharge rate of the 161 neurons, when mean arterial pressure was between 80 and 100 mmhg, was 7.5 ± 6.4 spikes/s (unless otherwise noted, these values represent mean + standard deviation throughout this paper). Antidromic activation of barosensory VLM neurons by stimulation of the spinal cord To examine whether or not barosensory VLM neurons sent descending bulbospinal projections, they were tested for antidromic activation by stimulating the right or left dorsolateral funiculus at the C2 level. Previously established criteria (LIPSKI, 1981), including collision of the evoked spike with a spontaneous spike discharge, a constant latency of the evoked spike and ability to respond to stimulation at high frequencies were used for the assessment for antidromic activation (Fig. 4A, B). Of 123 barosensory VLM neurons tested, 49 (40%) were activated antidromi- Japanese Journal of Physiology
7 BAROSENSORY NEURONS IN VENTROLATERAL MEDULLA 1147 Fig. 2. A barosensory VLM neuron with spontaneous discharges inhibited by stimulation of aortic nerve A-fibers. A: 20 superimposed oscilloscopic records of unit discharges (top) and RNA (bottom) and their responses to aortic nerve stimulation. B and C: peristimulus time histogram of the single unit, illustrated in A, and RNA following stimulation of the left (B) or right (C) aortic nerve. Number of trials was 256. Parameters of the train of 10 pulses for nerve stimulation were 1 V (intensity), 0.2 ms (duration), and 100 Hz (frequency). Stimulus was applied during the period indicated by horizontal bars at the top of each recording. cally. Twenty-six of these units (53%) were excited by stimulating the dorsolateral funiculus ipsilateral to the recording site, whereas 23 units (47%) were excited by stimulation of the contralateral dorsolateral funiculus. Calculated conduction velocities of the descending axons varied between 0.45 and 10.0 m/s. Although it varied over a wide range, there was a tendency of a natural division at the velocity of 1.5 m/s. Eighty-six percent (42/49) had the conduction velocity between 1.5 and 10 m/s, whereas l4 (7/49) had the velocity smaller than 1.5 m/s (Fig. 4C). The spontaneous discharge rate of antidromically activated units and those not activated was, respectively, spikes/s (n = 49) and spikes/s (n = 74). The difference in the two values was statistically insignificant (p >0.1, Student's t-test). There was a preponderance, in the rostral portion of the ventrolateral medulla, of antidromically activated barosensory VLM neurons relative to those not activated (Fig. 3B). To examine this point quantitatively, the barosensory VLM neurons were divided into the following three groups on the basis of the anterior- Vol. 36, No. 6, 1986
8 1148 N. TERUI, Y. SAEKI, and M. KUMADA Fig. 3. A: distribution of 161 barosensory VLM neurons illustrated on 3 representative frontal sections of the medulla extending from 1 to 3 mm rostral to the obex. On the upper 3 sections are shown locations of unit not antidromically activated by stimulation of the dorsolateral funiculus (filled circles) or those not tested for the antidromic response (open circles). On the corresponding lower sections are shown antidromically activated units having the calculated conduction velocity of the bulbospinal axon greater (filled squares) or smaller (asterisks) than 1.5 m/s. B: frequency histogram of distribution of antidromically activated neurons, represented by hatched columns, and that of not-antidromically activated neurons, represented by dotted columns, according to the rostro-caudal coordinate of each unit. The coordinate ranged between 0.5 and 3.5 mm rostral to the obex and was divided into 6 successive segments with a 0.5 mm width. Abbreviations: Cn + Gn, nucleus cuneatus and nucleus gracilis; ION, inferior olivary nucleus; NA, nucleus ambiguus; NmX, dorsal motor nucleus of vagus; NTS, nucleus of solitary tract; NV, spinal nucleus of trigeminal nerve; NXII, nucleus of hypoglossal nerve; Pyr, pyramidal tract; Tr sp V, spinal tract of trigeminal nerve; TS, solitary tract. posterior coordinate of their location along the brainstem. Namely, those located (1) between 3.5 and 2.6mm anterior to the obex (abbreviated as A 3.5 and A 2.6, respectively), (2) between A 2.5 and A 1.6, and (3) between A 1.5 and A 0.6. The ratio of antidromically activated units to not-activated units within the groups (1) through (3) were, respectively, 2.09 (23/11), 0.68 (17/25), and 0.24 (9/38). The ratio was significantly greater within group (1) than in (3) (p <0.01, x2-analysis). It thus follows that the relative distribution of antidromically activated and not-activated barosensory VLM neurons is nonuniform, the former distributed more densely within the rostral portion of the ventrolateral medulla. In short, 40 c of barosensory VLM neurons were confirmed to send descending Japanese Journal of Physiology
9 BAROSENSORY NEURONS IN VENTROLATERAL MEDULLA 1149 Fig. 4. Collision test for antidromic activation of a barosensory VLM neuron. A: response of a unit to stimulation, indicated by an arrow, of the ipsilateral dorsolateral funiculus 9 ms (top tracing) or 7 ms (bottom tracing) after a naturally occurring spike. B: the same unit was stimulated by 2 shocks separated by 2.7 ms (top tracing) or 2.3 ms (bottom tracing). Intensity and duration of the stimulating pulse were, respectively, 300 RA and 0.2 ms. Each record was obtained by superimposing 5 successive tracings. C: frequency histogram of calculated axonal conduction velocities of antidromically activated barosensory VLM neurons excited by stimulation of ipsilateral (open column) or contralateral (hatched column) dorsolateral funiculus at the C2 segment. bulbospinal axons. Such neurons were located preponderantly within the rostral portion of the ventrolateral medulla. Calculated conduction velocities of the descending axons ranged between 0.45 and 10 m/s with a tendency of a natural Vol. 36, No. 6, 1986
10 1150 N. TERUI, Y. SAEKI, and M. KUMADA division at the velocity of 1.5 m/s. Response of barosensory VLM neurons to stimulation of aortic nerve A -fibers When the aortic nerve A-fibers were stimulated by a train of rectangular pulses of 0.2 ms duration at a frequency of 100 Hz, spontaneous activity of the 161 barosensory VLM neurons was diminished without a single exception (Fig. 2). Changing the stimulus frequency between 30 and 300 Hz, while affecting the degree of the response, invariably brought about an inhibition of neuronal activity. The inhibition ascribable to A-fiber afferents was elicited only by tetanic stimulation but not by a single pulse. Although the barosensory VLM neurons were all searched within the left ventrolateral medulla, 93% of them (77/83) responded to both of the individual stimulation of the right or left aortic nerve (Fig. 2B, C). The onset of the inhibition of spontaneous activity following stimulation of the right or left aortic nerve was, respectively, ms (n = 90) and ms (n =148). The difference in the two values was statistically significant (p <0.01; unpaired t-test). Inhibition of spontaneous activity of barosensory VLM neurons always preceded sympathoinhibitory response of left renal nerve activity (RNA) which had the onset latency to stimulation of the right or left aortic nerve of ms (n = 96) and ms (n =148). The difference in the two values was again significant (p <0.01; unpaired t-test). Between the barosensory VLM neurons antidromically activated and not activated, there were no significant differences in the onset latency to stimulation of A-fibers of the left aortic nerve ( ms; n = 44 vs ms; n = 68, p>0.2) or the percentage of neurons activated by both of the individual stimulations of the right or left aortic nerve (94% vs. 93%, p >0.5, both by unpaired t- test). In short, stimulation of aortic nerve A-fibers always resulted in an inhibition of spontaneous activity of barosensory VLM neurons. A vast majority of these neurons, whether sending descending bulbospinal axons or not, received afferent inputs from the aortic nerve of both sides. Response of barosensory VLM neurons to activation of aortic nerve C -fibers The aortic nerve C-fibers of the rabbit are composed of barosensory and nonbarosensory afferent groups. The latter was most likely nociceptive fibers innervating the cardiopulmonary region (NUMAO et al., 1983). The response of barosensory VLM neurons to stimulation of aortic nerve C-fibers was investigated by applying a single pulse to the aortic nerve. Although stimulation of C-fibers inevitably co-activated A-fibers, we found that the neuronal as well as sympathetic, responses due to C-fiber activation were elicited by a single pulse stimulation of the aortic nerve, whereas the responses attributable to A-fibers were produced only by tetanic stimulation. Ninety-three percent (74/80) of barosensory VLM neurons responded to single Japanese Journal of Physiology
11 BAROSENSORY NEURONS IN VENTROLATERAL MEDULLA 1151 Fig. 5. Three representative patterns of responses of barosensory VLM neurons to stimulation of aortic nerve C-fibers. A single shock of 0.5 ms duration and the intensity of 10 V (in A and B) or 7 V (in C) was given to the left (in A and B) or right (in C) aortic nerve at the time indicated by an arrow. In A through C, each tracing represents the response of a single unit (top) or RNA (bottom) obtained by superimposing 64 (in A) or 128 (in B and C) successive trials. According to the criteria described in the text, the neuronal response was classified as I (A), E1-I (B) or I-E2 type (C). _ pulse stimulation of either right or left aortic nerve. A comparable figure (93 c 27/29) was obtained with respect to antidromically activated units. The response to aortic nerve C-fiber stimulation gave rise to a response consisting of various Vol. 36, No. 6, 1986
12 1152 N. TERUI, Y. SAEKI, and M. KUMADA temporal sequences of inhibitory and excitatory components which was not always the same as that of simultaneously recorded RNA (Fig. 5). When the right or left aortic nerve was stimulated individually, the response pattern of the same barosensory VLM neuron was either the same (35% =12/34) or different (65% = 22/34). Thus, the following numbers were based on the pooled data on the response to right or left aortic nerve stimulation. In 91 of 114 tests performed on 80 units (80%), activation of C-fibers resulted in an inhibition of spontaneous activity. The inhibitory response, however, was often preceded (42%) or followed (34%) by short-lasting excitatory components, which were referred to as early (E1) and late (E2) excitatory components as to their temporal relationship to the inhibitory response (I component; Fig. SB, C). The onset latencies of E1, E2, and I components were, respectively, (n = 48), (n = 39), and ms (n = 91). Responses of barosensory VLM neurons were then classified according to their sequences of excitatory and inhibitory components. The majority of the responses (71% = 81/114) were classified as one of the following three types; the I component only (referred to as I type; 24.6%), E1 and I components (E1-I type; 23.7%), or I and E2 components (I-E2 type; 22.8%). Thirteen cases out of 114 (11.4%) exhibited E1 and/or E2 components only. No apparent difference was found with respect to the response of barosensory VLM neurons antidromically activated and not activated. In short, most barosensory VLM neurons receive afferent inputs from both A- and C-fibers of the aortic nerve. The response to C-fiber activation was primarily an inhibition of spontaneous activity, although often accompanied by a short-lasting excitation. Response of barosensory VLM neurons to a rise in arterial pressure As already mentioned, excitation of aortic nerve C-fibers, which contain nonbarosensory afferents as well, sometimes elicited an excitatory component in the evoked neuronal response. To examine whether or not C-fiber afferents from arterial baroreceptors inhibit spontaneous activity of barosensory VLM neurons, mean arterial pressure was raised above the threshold of C-fibers (about 95 mmhg; JONES and THOREN, 1977) in 19 vagotomized rabbits by a bolus intravenous injection of phenylephrine. It should be noted that in these animal preparations the aortic and vagus nerve were cut on both sides. Thus, the neuronal response to a rise in arterial pressure is derived from carotid sinus baroreceptors. The test was performed on 50 barosensory VLM neurons of which 14 were antidromically activated. Raising mean arterial pressure above 95 mmhg invariably caused an inhibition of activity of all the barosensory VLM neurons tested. In 40 out of 50 neurons (80%), spontaneous firing was totally suppressed for more than 1 s. With respect to antidromically activated units, the corresponding figures were 10 out of 14 units (71%). This pressure-induced inhibition of neuronal activity was observed even in those barosensory VLM neurons which exhibited an excitatory component in the evoked response to C-fiber activation. These results indicate that Japanese Journal of Physiology
13 BAROSENSORY NEURONS IN VENTROLATERAL MEDULLA 1153 Fig. 6. Response of two barosensory VLM neurons to stimulation of left (left panels) or right (right panels) carotid chemoreceptors by a close arterial injection of NaCN (5 µg) in 0.2 ml of saline at the time indicated by an arrow. Each set of recordings consists, from top to bottom, unit activity, RNA, integrated phrenic nerve activity and arterial pressure. A: a single unit that increased its discharge rate to chemoreceptor stimulation of both sides. It was accompanied by an enhancement of phrenic nerve activity (IPNA) and a pressor response (AP). B: another unit from the same animal that exhibited an increase in the discharge rate in response to stimulation of the right (i.e., contralateral) carotid chemoreceptors. Only a decrease in neuronal activity which followed an increase in RNA was elicited by stimulation of left carotid chemoreceptors. afferents from arterial baroreceptors, whether medullated or nonmedullated, uniformly cause an inhibition of spontaneous activity of barosensory VLM neurons. They also demonstrate convergence of the afferent signals from aortic and carotid sinus baroreceptors onto these neurons. Vol. 36, No. 6, 1986
14 1154 N. TERUI, Y. SAEKI, and M. KUMADA Response of barosensory VLM neurons to stimulation of carotid body chemoreceptors The response to stimulation of carotid body chemoreceptors was investigated in 43 barosensory VLM neurons of which 9 were antidromically activated. Right and left carotid body chemoreceptors were individually stimulated by a close arterial injection of sodium cyanide (5-10 µg in 0.2 ml of saline) through a catheter placed in each thyroid artery. In 41 out of these 44 neurons (93%), the neuronal response to arterial chemoreceptor stimulation consisted of an immediate increase in the discharge rate (an early response) followed by a secondary decrease in it (a late response; Fig. 6). The early response occurred prior to the rise in arterial pressure and was accompanied by an enhancement of phrenic and renal nerve activities. It was thus a direct consequence of chemoreceptor stimulation. By contrast, the late response, which occurred subsequent to the pressor response and was accompanied by a decrease in RNA, was a result of the arterial baroreceptor reflex caused by a rise in arterial pressure. In the remaining 3 barosensory VLM neurons, only the late response was elicited. The response to stimulation of each of the right and left carotid body chemoreceptors were compared in 26 barosensory VLM neurons of which 6 were antidromically activated. The neuronal response was greater to stimulation of the right (i.e., contralateral to the recording site) carotid body chemoreceptors, although responses of RNA, phrenic nerve activity and arterial pressure were comparable (Fig. 6). In fact, the peak values of systolic arterial pressure following stimulation of the right and left chemoreceptors were and mmhg (n = 26), respectively. Furthermore, 8 out of 26 neurons responded exclusively to stimulation of right carotid body chemoreceptors, whereas none of the neurons responded exclusively to stimulation of left chemoreceptors. Thus, barosensory VLM neurons receive afferent signals not only from arterial baroreceptors but also from arterial chemoreceptors. The effect of carotid body chemoreceptor stimulation on neuronal activity is excitatory and, furthermore, dominant onto neurons located within the contralateral side of the ventrolateral medulla. Responses of barosensory VLM neurons to stimulation of the posterior hypothalamic area In 5 rabbits, the poterior hypothalamic area, as defined by SAWYER et al. (1954) in rabbits, was electrically stimulated at moderate intensities (less than 200 /2A) to test the orthodromic response of barosensory VLM neurons. The stimulus site was distributed within the peri-fornical area ventromedial to the mammillothalamic tract (Fig. 7). Stimulation of the posterior hypothalamic area by a train of three pulses (100 Hz, 0.2 ms pulse duration) resulted in an excitation of spontaneous discharges of 7 barosensory VLM neurons out of 8. None of the 8 neurons were antidromically activated. The onset latency of the excitatory response was 55.0 ± 15.0 ms with the Japanese Journal of Physiology
15 BAROSENSORY NEURONS IN VENTROLATERAL MEDULLA 1155 Fig. 7. Responses of a barosensory VLM neuron and RNA to stimulation of the posterior hypothalamic area. A: peristimulus time histogram averaged over 256 successive trials in response to stimulation of the left posterior hypothalamic area by 3 pulses given at the time indicated by arrowheads. Stimulus consisted of rectangular pulses of 0.2 ms duration and 200 j A intensity separated by 10 ms. Histologically identified stimulus sites (filled circles) in the hypothalamus from 5 rabbits are shown in B. Abbreviations: IC, internal capsule; FX, fornix; MT, mammillothalamic tract; OT optic tract. neuron and ms with the renal nerve. Thus, above 90% of barosensory VLM neurons responded orthodromically to stimulation of the posterior hypothalamic area. Spontaneous activity of the neurons, if responsive, was invariably increased. In connection with onset-latencies of the excitatory response, to hypothalamic stimulation, of barosensory VLM neurons and of the renal nerve, the difference in the two values calculated on individual neurons was ms (n = 7). When the aortic nerve A-fibers were stimulated in place of the posterior hypothalamic area, the corresponding differences was ms (n = 7). Thus, the sympathoexcitatory response to stimulation of the hypothalamus may be partly mediated by descending pathways that bypass the ventrolateral medulla. Cardiac-related and respiratory-related rhythms in spontaneous discharges of barosensory VLM neurons Sympathetic nerve discharges are synchronized into bursts locked to the cardiac and respiratory cycles. The cardiac-related rhythm is a consequence of the arterial baroreceptor reflex (GEBBER and BARMAN, 1980). On the other hand, the mechanism responsible for the respiratory-related rhythm in tonic sympathetic Vol. 36, No. 6, 1986
16 1156 N. TERUI, Y. SAEKI, and M. KUMADA Fig. 8. Cardiac-related (A) and respiratory-related (B) rhythms observed simultaneously in a barosensory VLM neuron. Tracings consist, from top to bottom, of unit activity, RNA, arterial pressure and electrocardiogram (in A) or phrenic nerve activity (in B). Records are sum of 256 trials triggered by the R-wave (in A) or 128 trials triggered by phrenic nerve activity (in B). Bin width was 5 ms in A and 20 ms in B. discharges, including the site of modulation exerted by the central neuronal mechanism generating the respiratory rhythm or the respiratory central pattern generator (EULER, 1983) on the sympathetic nervous system, is undecided (KOEPCHEN, 1983). The cardiac-related rhythm was examined by superimposing spontaneous discharges of the barosensory VLM neurons, over 128 successive sweeps, triggered by the R-wave of ECG. A distinct cardiac-related rhythm was discerned in 138 out of 161 neurons (86%) (Fig. 8A). Among 49 antidromically activated barosensory VLM neurons, 46 (94%) exhibited that rhythm. As the aortic and vagus nerves were already disrupted on both sides, the result provides another evidence for a convergence of afferent signals from carotid sinus and aortic baroreceptors at or prior to the ventrolateral medulla along the reflex pathway. There was a time lag of ms (n =138) from the bottom of the discharge rate of the neurons to that of the renal nerve (Fig. 8A). Subsequently, in order to see whether or not respiratory-related rhythm already occurred at the barosensory VLM neurons, spontaneous discharges of these neurons were correlated with phrenic nerve activity. In 29 bilaterally vagotomized and paralyzed rabbits under artificial respiration (50-60 cycle/s), spontaneous activ- Japanese Journal of Physiology
17 BAROSENSORY NEURONS IN VENTROLATERAL MEDULLA 1157 ity of barosensory VLM neurons was superimposed over 128 successive sweeps triggered by phrenic nerve activity. If summed neuronal activity exhibited a distinct periodicity linked to that of respiratory volleys in phrenic nerve activity, those neurons were considered to have the respiratory-related rhythm in spontaneous activity. At the time of recording neuronal discharges, the period of respiratory volleys in phrenic nerve activity ranged between 550 and 1,940 ms (1,176.9 ± ms; n=72). Of 97 barosensory VLM neurons tested, 72 (74%) showed a distinct respiratory-related rhythm. As to antidromically activated units, 85% (22/26) of them showed such a rhythm (Fig. 8B). In 70 of the 72 units with the respiratoryrelated rhythm (97%), the discharge rate increased gradually during the midinspiratory phase, reached the maximum between the late-inspiratory and earlyexpiratory phases, and then diminished during the rest of the expiratory phase. This pattern of rhythm in unit discharges was followed, with the lag time of ms (n = 69), by that in RNA. In 2 exceptional units, the peak of the neuronal activity occurred during the early-inspiratory phase. The lag time of the respiratory-related barosensory VLM neurons from the peak of phrenic nerve activity to that of unit discharges was ms (n = 70; range 0 to 390 ms). Thus, the majority of respiratory-related barosensory VLM neurons in vagotomized animals had the discharge property of the inspiratory-expiratory phasespanning type. It has been suggested that respiratory-related rhythm in discharges of barosensory VLM neurons in rats is a result of respiratory changes in arterial pressure which affected activity of arterial baroreceptors (BROWN and GUYENET, 1985). In rabbits, however, this was not the case, since the rhythm of phrenic nerve activity in vagotomized animal preparations was unrelated to that of artificial respiration which mechanically caused fluctuations in arterial pressure of less than 5 mmhg (Fig. 8B). Thus, the respiratory central pattern generator influences, either directly or indirectly, activity of barosensory VLM neurons within the medulla and contributes to generation of the respiratory-related rhythm in sympathetic nerve activity. DISCUSSION The present study confirms previous results obtained from electrophysiological studies in cats (CAVERSON et al., 1983, 1984) that demonstrated participation of neurons in the ventrolateral medulla in the arterial baroreceptor reflex. It also provides definitive evidence for the role of neurons in the ventrolateral medulla in the arterial baroreceptor reflex in rabbits, since previous studies in this animal species all involved electrical or chemical lesions and stimulations which nondiscriminatingly affected cardiovascular as well as noncardiovascular neurons alike (DAMPNEY and MOON, 1980; DAMPNEY et al., 1982; GOODCHILD et al., 1984). In some fundamental respects, however, our results are at odds with those in Vol. 36, No. 6, 1986
18 1158 N. TERUI, Y. SAEKI, and M. KUMADA cats as reported by CAVERSON et al. (1983, 1984). First, in cats, activation of arterial baroreceptors by an injection of phenylephrine either increased (50% =11/22) or decreased (5O =11 /22) the discharge rate of "ventrolateral medullary neurons" that projected their axons to the region of the intermediolateral cell column of the spinal cord. In rabbits, activation of arterial barosensory afferents either by an injection of phenylephrine or electrical stimulation of the aortic nerve A-fibers invariably diminished the spontaneous discharge rate of barosensory VLM neurons tested, some of which sent descending axons to the spinal cord. Secondly, in cats, activation of carotid chemoreceptors by sodium cyanide resulted in an increase (59 c =13/22), decrease (23 = 5/22) or no change (18% _ 4/22) in spontaneous activity of ventromedullary neurons which also responded to baroreceptor activation. In rabbits, however, the majority of barosensory VLM neurons (93%=40/43) was excited by stimulation of carotid chemoreceptors, although a few neurons (7% = 3/43) were unaffected by it. And finally, in cats, conduction velocities of descending spinal axons of ventrolateral medullary neurons ranged between 4.9 and 60.7 m/s and were never less than 1.5 m/s, i.e., those of nonmyelinated fibers. By contrast, in rabbits, there were a group of barosensory VLM neurons which had the conduction velocity of their spinal axons less than 1.5 m/s. In view of a very similar distribution of neurons examined within the ventrolateral medulla in cats and rabbits, such drastic differences in results are quite puzzling, unless one attributes them to the species differences. There are, however, at least three major differences in experimental methods that might have caused these discrepancies in results. First, to activate arterial barosensory afferents, CAVERSON et al. (1983, 1984) used both electrical stimulation of the carotid sinus and aortic nerves and an injection of phenylephrine. As has been shown (FIDONE and SATO, 1969; DEGROAT and LALLEY, 1974), carotid sinus and aortic nerves of cats contain both barosensory and chemosensory afferents. Since afferents of both modalities are composed of A- and C-fiber groups, it is impossible to activate afferent fibers of either modality selectively by electrical stimulation. As mentioned earlier, the aortic nerve A-fibers of adult rabbits, which we stimulated to activate barosensory VLM neurons, are considered to consist of barosensory afferents. Nevertheless, it is hard to explain why natural stimulation of arterial baroreceptors by raising arterial pressure always inhibited spontaneous activity of barosensory VLM neurons in rabbits, but excited about half the ventrolateral medullary neurons in cats. Secondly, the neurons they examined were mostly but not always spontaneously active. Therefore, barosensory VLM neurons without spontaneous activity in rabbits might have been excited by baroreceptor stimulation. However, even spontaneously active neurons in the cat's ventrolateral medulla responded differentially to stimulation of arterial baroreceptors. They interpreted the opposite effect of baroreceptor stimulation on ventrolateral medullary neurons to mean that these neurons originated bulbospinal sympatho-excitatory and sympatho-inhibitory pathways. However, recording of synaptic activity in the sympathetic preganglionic Japanese Journal of Physiology
19 BAROSENSORY NEURONS IN VENTROLATERAL MEDULLA 1159 neurons in cats with intact baroreceptor nerves revealed that spontaneously occurring synaptic activity consisted predominantly of excitatory post-synaptic potentials whereas inhibitory post-synaptic potentials were rarely observed (DEMBOWSKY et al., 1985). Furthermore, in cats, microinjections of D,L-homocysteic acid to stimulate neurons in the nucleus paragigantocellularis lateralis which lay caudal to the facial nucleus and adjacent to the rostral third of the inferior alive evoked a rise in arterial pressure and vasoconstrictions (LovicK and HILTON, 1985; MCALLEN,1986). Ross et al. (1984a) demonstrated that the descending bulbospinal pathway originating from the rostral ventrolateral medulla of the rat was sympatho-excitatory. Our results rather than those by CAVERSON et al. (1983, 1984) fit in with these findings. Another important difference in experimental methods is that barosensory VLM neurons in the present study were examined for their bulbospinal projections by stimulating the dorsolateral funiculus at the upper cervical cord rather than the region of the intermediolateral cell column of the thoracic cord. Admittedly, there is no guarantee that antidromically activated barosensory VLM neurons actually innervate sympathetic preganglionic neurons directly or via interneurons. However, the same argument holds even if the region of the intermediolateral cell column is electrically stimulated, since stimulation may excite both passing fibers and, as a result of current spread, cells outside the region. Justifications for stimulating the dorsolateral funiculus at the second cervical segment are as follows. (1) In various animal species including rabbits as shown in this study, there are prominent descending sympatho-excitatory pathways passing through the dorsolateral funiculus of the cervical cord (BARMAN, 1984). (2) Pressor and sympatho-excitatory points in the ventrolateral medulla in rabbits correspond very closely to the location of spinally projecting cells (BLESSING et al., 1981; DAMPNEY et al., 1982). Since sympatho-excitatory response involves sympathetic preganglionic neurons, it follows that neurons in the ventrolateral medulla project, directly or via spinal interneurons, to sympathetic preganglionic neurons. (3) When descending the sympatho-excitatory pathways were tested for their antidromic activation of neurons in the ventrolateral medulla, such neurons were mostly activated by stimulation of the dorsolateral funiculus, although some were activated by stimulation of the ventrolateral quadrant of the spinal cord (LovICK, 1985). (4) These facts collectively lead us to conclude that some, or perhaps most, ventrolateral medullary neurons send descending sympatho-excitatory axons through the dorsolateral funiculus, either directly or via interneurons, to the sympathetic preganglionic neurons. (5) Finally, and most importantly, in order to identify barosensory VLM neurons with slow conducting bulbospinal axons, the stimulus site had to be close enough to the recording site. Otherwise, a naturally occurring orthodromic spike will more likely collide with an antidromically evoked spike somewhere along the axon. Consequently, the neuron in question could be erroneously identified as not antidromically activated. This might underlie absence of slow conducting bulbospinal axons in the cat, since stimulation was applied at Vol. 36, No. 6, 1986
20 1160 N. TERUI, Y. SAEKI, and M. KUMADA the level of the second thoracic cord (CAVERSON et al., 1983, 1984). In connection with the species difference, BROWN and GUYENET (1985) described "cardiovascular neurons" in the rat's rostra! ventrolateral medulla which behaved very similarly to the barosensory VLM neurons in the rabbit. These neurons, all of which exhibited spontaneous activity, were immediately and completely suppressed by a rapid rise of arterial pressure by a bolus injection of norepinephrine. More than half of the 138 cardiovascular neurons sent descending axons which were classified into fast and slow conducting fibers with a division of the velocity appearing at 1.5 m/s. Furthermore, some cardiovascular neurons were excited by hypothalamic stimulation. Our results are in line with observations by KANNAN and YAMASHITA (1984) in rats. Namely, about half of the spontaneously active neurons in the ventrolateral medulla which sent efferent projections to the paraventricular nucleus of the hypothalamus were inhibited but never excited by baroreceptor activation induced by an intravenous bolus injection of phenylephrine. Despite some fundamental differences in results obtained from cats and rabbits, we extend previous findings in that spontaneous activity of barosensory VLM neurons is inhibited by inputs from aortic and carotid sinus baroreceptors mediated by barosensory A- and C-fibers and is increased by the afferent signal from carotid body chemoreceptors, the excitatory effect being greater from receptors in the contralateral side. The latter finding may underlie the observation by KUBIN et al. (1985) that splitting the medulla in the cat, through disruption of abundant neuronal connections across the midsagittal plane, greatly attenuated the carotid body chemoreceptor-sympathetic reflex. Furthermore, nonbarosensory afferents among aortic nerve C-fibers (NUMAO et al., 1983) affect barosensory VLM neurons. There is abundant convergence of afferent signals from baro- and chemoreceptors, both ipsilateral and contralateral, onto a single barosensory VLM neuron. Another major finding of the present study is demonstration of the respiratoryrelated rhythm in spontaneous activity of barosensory VLM neurons. This rhythm is not a consequence of the arterial baroreceptor reflex, since it is locked to phrenic nerve activity but not to the minute changes in arterial pressure generated by artificial respiration. The respiratory-related rhythm of neurons preceded by about 170 ms that of RNA. This result demonstrates an intramedullary coupling between the respiratory central pattern generator and barosensory VLM neurons. Such coupling probably underlies the observation by HUKUHARA (1984) in rabbits that respiratory-related volleys of RNA usually occurred during the late inspiratory and early and middle expiratory phases, when the period of phrenic volleys was under 2 s as in the present study. On the basis of the fact that primary afferent fibers of the aortic nerve project predominantly to the intermediate portion of the nucleus tractus solitarii (NTS) in rabbits (WALLACH and LOEWY,1980; KALIA,1981), we can estimate the conduction time and velocity from the NTS to the ventrolateral medulla. The conduction time Japanese Journal of Physiology
to Regulation of the Brain Vessels
 Short Communication Japanese Journal of Physiology, 34,193-197,1984 The Relevance of Cardio-pulmonary-vascular Reflex to Regulation of the Brain Vessels Masatsugu NAKAI and Koichi OGINO Department of Cardiovascular
Short Communication Japanese Journal of Physiology, 34,193-197,1984 The Relevance of Cardio-pulmonary-vascular Reflex to Regulation of the Brain Vessels Masatsugu NAKAI and Koichi OGINO Department of Cardiovascular
Regulation of respiration
 Regulation of respiration Breathing is controlled by the central neuronal network to meet the metabolic demands of the body Neural regulation Chemical regulation Respiratory center Definition: A collection
Regulation of respiration Breathing is controlled by the central neuronal network to meet the metabolic demands of the body Neural regulation Chemical regulation Respiratory center Definition: A collection
Department of Neurology/Division of Anatomical Sciences
 Spinal Cord I Lecture Outline and Objectives CNS/Head and Neck Sequence TOPIC: FACULTY: THE SPINAL CORD AND SPINAL NERVES, Part I Department of Neurology/Division of Anatomical Sciences LECTURE: Monday,
Spinal Cord I Lecture Outline and Objectives CNS/Head and Neck Sequence TOPIC: FACULTY: THE SPINAL CORD AND SPINAL NERVES, Part I Department of Neurology/Division of Anatomical Sciences LECTURE: Monday,
DEVELOPMENT OF BRAIN
 Ahmed Fathalla OBJECTIVES At the end of the lecture, students should: List the components of brain stem. Describe the site of brain stem. Describe the relations between components of brain stem & their
Ahmed Fathalla OBJECTIVES At the end of the lecture, students should: List the components of brain stem. Describe the site of brain stem. Describe the relations between components of brain stem & their
A CENTRAL NORADRENERGIC MECHANISM RESPONSIBLE FOR MODULATION OF THE ARTERIAL BARORECEPTOR REFLEX IN CATS
 www.kopfinstruments.com A CENTRAL NORADRENERGIC MECHANISM RESPONSIBLE FOR MODULATION OF THE ARTERIAL BARORECEPTOR REFLEX IN CATS V. S. EREMEEV, Ph.D. R. S. KHRUSTALEVA, Ph.D. V. A. TSYRLIN, Ph.D. Yu. I.
www.kopfinstruments.com A CENTRAL NORADRENERGIC MECHANISM RESPONSIBLE FOR MODULATION OF THE ARTERIAL BARORECEPTOR REFLEX IN CATS V. S. EREMEEV, Ph.D. R. S. KHRUSTALEVA, Ph.D. V. A. TSYRLIN, Ph.D. Yu. I.
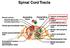 Spinal Cord Tracts DESCENDING SPINAL TRACTS: Are concerned with somatic motor function, modification of ms. tone, visceral innervation, segmental reflexes. Main tracts arise form cerebral cortex and others
Spinal Cord Tracts DESCENDING SPINAL TRACTS: Are concerned with somatic motor function, modification of ms. tone, visceral innervation, segmental reflexes. Main tracts arise form cerebral cortex and others
Motor tracts Both pyramidal tracts and extrapyramidal both starts from cortex: Area 4 Area 6 Area 312 Pyramidal: mainly from area 4 Extrapyramidal:
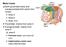 Motor tracts Both pyramidal tracts and extrapyramidal both starts from cortex: Area 4 Area 6 Area 312 Pyramidal: mainly from area 4 Extrapyramidal: mainly from area 6 area 6 Premotorarea: uses external
Motor tracts Both pyramidal tracts and extrapyramidal both starts from cortex: Area 4 Area 6 Area 312 Pyramidal: mainly from area 4 Extrapyramidal: mainly from area 6 area 6 Premotorarea: uses external
Influence of the hypothalamic paraventricular nucleus on cardiovascular neurones in the rostral ventrolateral medullaoftherat
 Keywords: Paraventricular nucleus, Ventrolateral medulla, Neurone 7347 Journal of Physiology (1998), 513.2, pp. 521 530 521 Influence of the hypothalamic paraventricular nucleus on cardiovascular neurones
Keywords: Paraventricular nucleus, Ventrolateral medulla, Neurone 7347 Journal of Physiology (1998), 513.2, pp. 521 530 521 Influence of the hypothalamic paraventricular nucleus on cardiovascular neurones
By Dr. Saeed Vohra & Dr. Sanaa Alshaarawy
 By Dr. Saeed Vohra & Dr. Sanaa Alshaarawy 1 By the end of the lecture, students will be able to : Distinguish the internal structure of the components of the brain stem in different levels and the specific
By Dr. Saeed Vohra & Dr. Sanaa Alshaarawy 1 By the end of the lecture, students will be able to : Distinguish the internal structure of the components of the brain stem in different levels and the specific
211MDS Pain theories
 211MDS Pain theories Definition In 1986, the International Association for the Study of Pain (IASP) defined pain as a sensory and emotional experience associated with real or potential injuries, or described
211MDS Pain theories Definition In 1986, the International Association for the Study of Pain (IASP) defined pain as a sensory and emotional experience associated with real or potential injuries, or described
susceptibility of either the axons in the dorsal and ventral roots, or the intramedullary
 213 J. Physiol. (31958) I40, 2I3-2I9 THE SITE OF ACTION OF PROCAINE ON THE ISOLATED SPINAL CORD OF THE FROG BY M. HARMEL AND J. L. MALCOLM From the Department of Physiology, State University of New York,
213 J. Physiol. (31958) I40, 2I3-2I9 THE SITE OF ACTION OF PROCAINE ON THE ISOLATED SPINAL CORD OF THE FROG BY M. HARMEL AND J. L. MALCOLM From the Department of Physiology, State University of New York,
J. Physiol. (I956) I33,
 232 J. Physiol. (I956) I33, 232-242 A STUDY OF THE EFFECT OF THE PATTERN OF ELECTRICAL STIMULATION OF THE AORTIC NERVE ON THE REFLEX DEPRESSOR RESPONSES By W. W. DOUGLAS, J. M. RITCHIE AND W. SCHAUMANN*
232 J. Physiol. (I956) I33, 232-242 A STUDY OF THE EFFECT OF THE PATTERN OF ELECTRICAL STIMULATION OF THE AORTIC NERVE ON THE REFLEX DEPRESSOR RESPONSES By W. W. DOUGLAS, J. M. RITCHIE AND W. SCHAUMANN*
Nervous System C H A P T E R 2
 Nervous System C H A P T E R 2 Input Output Neuron 3 Nerve cell Allows information to travel throughout the body to various destinations Receptive Segment Cell Body Dendrites: receive message Myelin sheath
Nervous System C H A P T E R 2 Input Output Neuron 3 Nerve cell Allows information to travel throughout the body to various destinations Receptive Segment Cell Body Dendrites: receive message Myelin sheath
I: To describe the pyramidal and extrapyramidal tracts. II: To discuss the functions of the descending tracts.
 Descending Tracts I: To describe the pyramidal and extrapyramidal tracts. II: To discuss the functions of the descending tracts. III: To define the upper and the lower motor neurons. 1. The corticonuclear
Descending Tracts I: To describe the pyramidal and extrapyramidal tracts. II: To discuss the functions of the descending tracts. III: To define the upper and the lower motor neurons. 1. The corticonuclear
Human Anatomy. Spinal Cord and Spinal Nerves
 Human Anatomy Spinal Cord and Spinal Nerves 1 The Spinal Cord Link between the brain and the body. Exhibits some functional independence from the brain. The spinal cord and spinal nerves serve two functions:
Human Anatomy Spinal Cord and Spinal Nerves 1 The Spinal Cord Link between the brain and the body. Exhibits some functional independence from the brain. The spinal cord and spinal nerves serve two functions:
A Cardiocardiac Sympathovagal Reflex in the Cat
 A Cardiocardiac Sympathovagal Reflex in the Cat By Peter J. Schwartz, Massimo Pagani, Federico Lombardi, Alberto Malliani, and Arthur M. Brown ABSTRACT The reflex changes in single cardiac vagal efferent
A Cardiocardiac Sympathovagal Reflex in the Cat By Peter J. Schwartz, Massimo Pagani, Federico Lombardi, Alberto Malliani, and Arthur M. Brown ABSTRACT The reflex changes in single cardiac vagal efferent
The Nervous System: Sensory and Motor Tracts of the Spinal Cord
 15 The Nervous System: Sensory and Motor Tracts of the Spinal Cord PowerPoint Lecture Presentations prepared by Steven Bassett Southeast Community College Lincoln, Nebraska Introduction Millions of sensory
15 The Nervous System: Sensory and Motor Tracts of the Spinal Cord PowerPoint Lecture Presentations prepared by Steven Bassett Southeast Community College Lincoln, Nebraska Introduction Millions of sensory
Chapter 9. Nervous System
 Chapter 9 Nervous System Central Nervous System (CNS) vs. Peripheral Nervous System(PNS) CNS Brain Spinal cord PNS Peripheral nerves connecting CNS to the body Cranial nerves Spinal nerves Neurons transmit
Chapter 9 Nervous System Central Nervous System (CNS) vs. Peripheral Nervous System(PNS) CNS Brain Spinal cord PNS Peripheral nerves connecting CNS to the body Cranial nerves Spinal nerves Neurons transmit
Lecture 4 The BRAINSTEM Medulla Oblongata
 Lecture 4 The BRAINSTEM Medulla Oblongata Introduction to brainstem 1- Medulla oblongata 2- Pons 3- Midbrain - - - occupies the posterior cranial fossa of the skull. connects the narrow spinal cord
Lecture 4 The BRAINSTEM Medulla Oblongata Introduction to brainstem 1- Medulla oblongata 2- Pons 3- Midbrain - - - occupies the posterior cranial fossa of the skull. connects the narrow spinal cord
Auditory and Vestibular Systems
 Auditory and Vestibular Systems Objective To learn the functional organization of the auditory and vestibular systems To understand how one can use changes in auditory function following injury to localize
Auditory and Vestibular Systems Objective To learn the functional organization of the auditory and vestibular systems To understand how one can use changes in auditory function following injury to localize
UNIT ACTIVITY IN THE MEDULLA OBLONGATA OF FISHES
 [218] UNIT ACTIVITY IN THE MEDULLA OBLONGATA OF FISHES BY S. WOLDRING AND M. N. J. DIRKEN Physiological Institute, Groningen, Netherlands (Received 17 July 1950) (With Plate 2 and one Text-figure) Adrian
[218] UNIT ACTIVITY IN THE MEDULLA OBLONGATA OF FISHES BY S. WOLDRING AND M. N. J. DIRKEN Physiological Institute, Groningen, Netherlands (Received 17 July 1950) (With Plate 2 and one Text-figure) Adrian
Different adrenal sympathetic preganglionic neurons regulate epinephrine and norepinephrine secretion
 Am J Physiol Regulatory Integrative Comp Physiol 279: R1763 R1775, 2000. Different adrenal sympathetic preganglionic neurons regulate epinephrine and norepinephrine secretion SHAUN F. MORRISON AND WEI-HUA
Am J Physiol Regulatory Integrative Comp Physiol 279: R1763 R1775, 2000. Different adrenal sympathetic preganglionic neurons regulate epinephrine and norepinephrine secretion SHAUN F. MORRISON AND WEI-HUA
Axon Nerve impulse. Axoplasm Receptor. Axomembrane Stimuli. Schwann cell Effector. Myelin Cell body
 Nervous System Review 1. Explain a reflex arc. 2. Know the structure, function and location of a sensory neuron, interneuron, and motor neuron 3. What is (a) Neuron Axon Nerve impulse Axoplasm Receptor
Nervous System Review 1. Explain a reflex arc. 2. Know the structure, function and location of a sensory neuron, interneuron, and motor neuron 3. What is (a) Neuron Axon Nerve impulse Axoplasm Receptor
bradycardia was abolished by vagotomy or atropine ( mg kg-' i.v.).
 J. Physiol (1987), 389, pp. 23-35 23 With 5 text-figures Printed in Great Britain DIFFERENTIAL CONTROL OF CARDIAC AND VASOMOTOR ACTIVITY BY NEURONES IN NUCLEUS PARAGIGANTOCELLULARIS LATERALIS IN THE CAT
J. Physiol (1987), 389, pp. 23-35 23 With 5 text-figures Printed in Great Britain DIFFERENTIAL CONTROL OF CARDIAC AND VASOMOTOR ACTIVITY BY NEURONES IN NUCLEUS PARAGIGANTOCELLULARIS LATERALIS IN THE CAT
Neural Integration I: Sensory Pathways and the Somatic Nervous System
 15 Neural Integration I: Sensory Pathways and the Somatic Nervous System PowerPoint Lecture Presentations prepared by Jason LaPres Lone Star College North Harris An Introduction to Sensory Pathways and
15 Neural Integration I: Sensory Pathways and the Somatic Nervous System PowerPoint Lecture Presentations prepared by Jason LaPres Lone Star College North Harris An Introduction to Sensory Pathways and
Feed-forward and feedback regulation of bladder contractility by Barrington s nucleus in cats
 J Physiol 557.1 (2004) pp 287 305 287 Feed-forward and feedback regulation of bladder contractility by Barrington s nucleus in cats Mitsuyoshi Sasaki Department of Physiology, Tokyo Medical University,
J Physiol 557.1 (2004) pp 287 305 287 Feed-forward and feedback regulation of bladder contractility by Barrington s nucleus in cats Mitsuyoshi Sasaki Department of Physiology, Tokyo Medical University,
Medical Neuroscience Tutorial
 Pain Pathways Medical Neuroscience Tutorial Pain Pathways MAP TO NEUROSCIENCE CORE CONCEPTS 1 NCC1. The brain is the body's most complex organ. NCC3. Genetically determined circuits are the foundation
Pain Pathways Medical Neuroscience Tutorial Pain Pathways MAP TO NEUROSCIENCE CORE CONCEPTS 1 NCC1. The brain is the body's most complex organ. NCC3. Genetically determined circuits are the foundation
Human Anatomy and Physiology - Problem Drill 11: Neural Tissue & The Nervous System
 Human Anatomy and Physiology - Problem Drill 11: Neural Tissue & The Nervous System Question No. 1 of 10 The human body contains different types of tissue. The tissue is formed into organs and organ systems.
Human Anatomy and Physiology - Problem Drill 11: Neural Tissue & The Nervous System Question No. 1 of 10 The human body contains different types of tissue. The tissue is formed into organs and organ systems.
Arterial Blood Supply
 Arterial Blood Supply Brain is supplied by pairs of internal carotid artery and vertebral artery. The four arteries lie within the subarachnoid space Their branches anastomose on the inferior surface of
Arterial Blood Supply Brain is supplied by pairs of internal carotid artery and vertebral artery. The four arteries lie within the subarachnoid space Their branches anastomose on the inferior surface of
Nervous Systems: Diversity & Functional Organization
 Nervous Systems: Diversity & Functional Organization Diversity of Neural Signaling The diversity of neuron structure and function allows neurons to play many roles. 3 basic function of all neurons: Receive
Nervous Systems: Diversity & Functional Organization Diversity of Neural Signaling The diversity of neuron structure and function allows neurons to play many roles. 3 basic function of all neurons: Receive
Pain classifications slow and fast
 Pain classifications slow and fast Fast Pain Slow Pain Sharp, pricking (Aδ) fiber Short latency Well localized Short duration Dull, burning (C) fiber Slower onset Diffuse Long duration Less emotional Emotional,
Pain classifications slow and fast Fast Pain Slow Pain Sharp, pricking (Aδ) fiber Short latency Well localized Short duration Dull, burning (C) fiber Slower onset Diffuse Long duration Less emotional Emotional,
SUPPLEMENTARY INFORMATION. Supplementary Figure 1
 SUPPLEMENTARY INFORMATION Supplementary Figure 1 The supralinear events evoked in CA3 pyramidal cells fulfill the criteria for NMDA spikes, exhibiting a threshold, sensitivity to NMDAR blockade, and all-or-none
SUPPLEMENTARY INFORMATION Supplementary Figure 1 The supralinear events evoked in CA3 pyramidal cells fulfill the criteria for NMDA spikes, exhibiting a threshold, sensitivity to NMDAR blockade, and all-or-none
Hypothalamus. To learn how the brain regulates neuroendocrine secretions NTA Ch 14, pgs Key Figs: 14-3; 14-4,
 Hypothalamus Objectives To learn the general organization of the hypothalamus and the functions of the major nuclei NTA Ch 14, pgs. 419-422 Key Figs: 14-2, 14-3 To learn how the brain regulates neuroendocrine
Hypothalamus Objectives To learn the general organization of the hypothalamus and the functions of the major nuclei NTA Ch 14, pgs. 419-422 Key Figs: 14-2, 14-3 To learn how the brain regulates neuroendocrine
anaesthetized cat: role of the nucleus tractus solitarii
 3858 Journal of Physiology (1995), 487.3, pp.739-749 739 Hypothalamic modulation of laryngeal reflexes in the anaesthetized cat: role of the nucleus tractus solitarii M. S. Dawid-Milner, L. Silva-Carvalho,
3858 Journal of Physiology (1995), 487.3, pp.739-749 739 Hypothalamic modulation of laryngeal reflexes in the anaesthetized cat: role of the nucleus tractus solitarii M. S. Dawid-Milner, L. Silva-Carvalho,
Cardiovascular Physiology IV.
 Cardiovascular Physiology IV. 48. Short-term control mechanisms of arterial blood pressure. 49. Long-term control of arterial blood pressure. Ferenc Domoki, November 14 2017. Challenges/expectations Blood
Cardiovascular Physiology IV. 48. Short-term control mechanisms of arterial blood pressure. 49. Long-term control of arterial blood pressure. Ferenc Domoki, November 14 2017. Challenges/expectations Blood
EE 791 Lecture 2 Jan 19, 2015
 EE 791 Lecture 2 Jan 19, 2015 Action Potential Conduction And Neural Organization EE 791-Lecture 2 1 Core-conductor model: In the core-conductor model we approximate an axon or a segment of a dendrite
EE 791 Lecture 2 Jan 19, 2015 Action Potential Conduction And Neural Organization EE 791-Lecture 2 1 Core-conductor model: In the core-conductor model we approximate an axon or a segment of a dendrite
Hole s Human Anatomy and Physiology Tenth Edition. Chapter 10
 PowerPoint Lecture Outlines to accompany Hole s Human Anatomy and Physiology Tenth Edition Shier Butler Lewis Chapter 10 Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or
PowerPoint Lecture Outlines to accompany Hole s Human Anatomy and Physiology Tenth Edition Shier Butler Lewis Chapter 10 Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or
Chapter 17 Nervous System
 Chapter 17 Nervous System 1 The Nervous System Two Anatomical Divisions Central Nervous System (CNS) Brain and Spinal Cord Peripheral Nervous System (PNS) Two Types of Cells Neurons Transmit nerve impulses
Chapter 17 Nervous System 1 The Nervous System Two Anatomical Divisions Central Nervous System (CNS) Brain and Spinal Cord Peripheral Nervous System (PNS) Two Types of Cells Neurons Transmit nerve impulses
Brainstem. By Dr. Bhushan R. Kavimandan
 Brainstem By Dr. Bhushan R. Kavimandan Development Ventricles in brainstem Mesencephalon cerebral aqueduct Metencephalon 4 th ventricle Mylencephalon 4 th ventricle Corpus callosum Posterior commissure
Brainstem By Dr. Bhushan R. Kavimandan Development Ventricles in brainstem Mesencephalon cerebral aqueduct Metencephalon 4 th ventricle Mylencephalon 4 th ventricle Corpus callosum Posterior commissure
High Threshold Aortic Baroreceptors Afferents in the Sympathetic Nerve of Monkey. Biswanath KoLEY, Pratima PAL, and Juthika KoLEY
 Japanese Journal of Physiology, 39, 145-153, 1989 High Threshold Aortic Baroreceptors Afferents in the Sympathetic Nerve of Monkey Biswanath KoLEY, Pratima PAL, and Juthika KoLEY Electrophysiology Unit,
Japanese Journal of Physiology, 39, 145-153, 1989 High Threshold Aortic Baroreceptors Afferents in the Sympathetic Nerve of Monkey Biswanath KoLEY, Pratima PAL, and Juthika KoLEY Electrophysiology Unit,
The Central Auditory System
 THE AUDITORY SYSTEM Each auditory nerve sends information to the cochlear nucleus. The Central Auditory System From there, projections diverge to many different pathways. The Central Auditory System There
THE AUDITORY SYSTEM Each auditory nerve sends information to the cochlear nucleus. The Central Auditory System From there, projections diverge to many different pathways. The Central Auditory System There
Autonomic Nervous System (the visceral motor system) Steven McLoon Department of Neuroscience University of Minnesota
 Autonomic Nervous System (the visceral motor system) Steven McLoon Department of Neuroscience University of Minnesota 1 Course News Coffee Hour Monday, Nov 6, 9:00-10:00am Surdyk s Café in Northrop Auditorium
Autonomic Nervous System (the visceral motor system) Steven McLoon Department of Neuroscience University of Minnesota 1 Course News Coffee Hour Monday, Nov 6, 9:00-10:00am Surdyk s Café in Northrop Auditorium
Lecture VIII. The Spinal Cord, Reflexes and Brain Pathways!
 Reflexes and Brain Bio 3411! Monday!! 1! Readings! NEUROSCIENCE 5 th ed: Review Chapter 1 pp. 11-21;!!Read Chapter 9 pp. 189-194, 198! THE BRAIN ATLAS 3 rd ed:! Read pp. 4-17 on class web site! Look at
Reflexes and Brain Bio 3411! Monday!! 1! Readings! NEUROSCIENCE 5 th ed: Review Chapter 1 pp. 11-21;!!Read Chapter 9 pp. 189-194, 198! THE BRAIN ATLAS 3 rd ed:! Read pp. 4-17 on class web site! Look at
Internal Organisation of the Brainstem
 Internal Organisation of the Brainstem Major tracts and nuclei of the brainstem (Notes) The brainstem is the major pathway for tracts and houses major nuclei, that contain sensory, motor and autonomics
Internal Organisation of the Brainstem Major tracts and nuclei of the brainstem (Notes) The brainstem is the major pathway for tracts and houses major nuclei, that contain sensory, motor and autonomics
(Received 5 November 1963) rabbit were 65 and 80 mm Hg, respectively. The mean arterial blood
 J. Phy8iol. (1964), 174, pp. 136-171 163 With 5 text-figure8 Printed in Great Britain AORTIC BARORCPTOR THRSHOLD AND SNSITIVITY IN RABBITS AT DIFFRNT AGS BY C. M. BLOOR* From the Nuffield Institute for
J. Phy8iol. (1964), 174, pp. 136-171 163 With 5 text-figure8 Printed in Great Britain AORTIC BARORCPTOR THRSHOLD AND SNSITIVITY IN RABBITS AT DIFFRNT AGS BY C. M. BLOOR* From the Nuffield Institute for
Blood pressure. Formation of the blood pressure: Blood pressure. Formation of the blood pressure 5/1/12
 Blood pressure Blood pressure Dr Badri Paudel www.badripaudel.com Ø Blood pressure means the force exerted by the blood against the vessel wall Ø ( or the force exerted by the blood against any unit area
Blood pressure Blood pressure Dr Badri Paudel www.badripaudel.com Ø Blood pressure means the force exerted by the blood against the vessel wall Ø ( or the force exerted by the blood against any unit area
Business. Midterm #1 is Monday, study hard!
 Business Optional midterm review Tuesday 5-6pm Bring your Physio EX CD to lab this week Homework #6 and 7 due in lab this week Additional respiratory questions need to be completed for HW #7 Midterm #1
Business Optional midterm review Tuesday 5-6pm Bring your Physio EX CD to lab this week Homework #6 and 7 due in lab this week Additional respiratory questions need to be completed for HW #7 Midterm #1
Theme 2: Cellular mechanisms in the Cochlear Nucleus
 Theme 2: Cellular mechanisms in the Cochlear Nucleus The Cochlear Nucleus (CN) presents a unique opportunity for quantitatively studying input-output transformations by neurons because it gives rise to
Theme 2: Cellular mechanisms in the Cochlear Nucleus The Cochlear Nucleus (CN) presents a unique opportunity for quantitatively studying input-output transformations by neurons because it gives rise to
POSTSYNAPTIC INHIBITION OF CRAYFISH TONIC FLEXOR MOTOR NEURONES BY ESCAPE COMMANDS
 J. exp. Biol. (1980), 85, 343-347 343 With a figures Printed in Great Britain POSTSYNAPTIC INHIBITION OF CRAYFISH TONIC FLEXOR MOTOR NEURONES BY ESCAPE COMMANDS BY J. Y. KUWADA, G. HAGIWARA AND J. J. WINE
J. exp. Biol. (1980), 85, 343-347 343 With a figures Printed in Great Britain POSTSYNAPTIC INHIBITION OF CRAYFISH TONIC FLEXOR MOTOR NEURONES BY ESCAPE COMMANDS BY J. Y. KUWADA, G. HAGIWARA AND J. J. WINE
Vestibular inputs to premotor respiratory interneurons in the feline C 1 -C 2 spinal cord. Adam Robert Anker. BS, University of Pittsburgh, 2003
 Vestibular inputs to premotor respiratory interneurons in the feline C 1 -C 2 spinal cord by Adam Robert Anker BS, University of Pittsburgh, 2003 Submitted to the Graduate Faculty of The Department of
Vestibular inputs to premotor respiratory interneurons in the feline C 1 -C 2 spinal cord by Adam Robert Anker BS, University of Pittsburgh, 2003 Submitted to the Graduate Faculty of The Department of
Primary Functions. Monitor changes. Integrate input. Initiate a response. External / internal. Process, interpret, make decisions, store information
 NERVOUS SYSTEM Monitor changes External / internal Integrate input Primary Functions Process, interpret, make decisions, store information Initiate a response E.g., movement, hormone release, stimulate/inhibit
NERVOUS SYSTEM Monitor changes External / internal Integrate input Primary Functions Process, interpret, make decisions, store information Initiate a response E.g., movement, hormone release, stimulate/inhibit
gallamine triethiodide (Flaxedil), the animals were artificially
 Proc. Natl. cad. Sci. US Vol. 75, No. 10, pp. 5239-5243, October 1978 Physiological Sciences Nature of differential sympathetic discharges in chemoreceptor reflexes (laterality in sympathetic action/cardiac
Proc. Natl. cad. Sci. US Vol. 75, No. 10, pp. 5239-5243, October 1978 Physiological Sciences Nature of differential sympathetic discharges in chemoreceptor reflexes (laterality in sympathetic action/cardiac
lecture #2 Done by : Tyma'a Al-zaben
 lecture #2 Done by : Tyma'a Al-zaben ** Hello SERTONIN! note:: the slide included within the sheet but make sure back to slide for pictures in the previous lecture we talk about ascending tract and its
lecture #2 Done by : Tyma'a Al-zaben ** Hello SERTONIN! note:: the slide included within the sheet but make sure back to slide for pictures in the previous lecture we talk about ascending tract and its
Human Anatomy - Problem Drill 11: The Spinal Cord and Spinal Nerves
 Human Anatomy - Problem Drill 11: The Spinal Cord and Spinal Nerves Question No. 1 of 10 Instructions: (1) Read the problem statement and answer choices carefully, (2) Work the problems on paper as needed,
Human Anatomy - Problem Drill 11: The Spinal Cord and Spinal Nerves Question No. 1 of 10 Instructions: (1) Read the problem statement and answer choices carefully, (2) Work the problems on paper as needed,
Introduction to Physiological Psychology
 Introduction to Physiological Psychology Review Kim Sweeney ksweeney@cogsci.ucsd.edu www.cogsci.ucsd.edu/~ksweeney/psy260.html Today n Discuss Final Paper Proposal (due 3/10) n General Review 1 The article
Introduction to Physiological Psychology Review Kim Sweeney ksweeney@cogsci.ucsd.edu www.cogsci.ucsd.edu/~ksweeney/psy260.html Today n Discuss Final Paper Proposal (due 3/10) n General Review 1 The article
REGULATION OF CARDIOVASCULAR SYSTEM
 REGULATION OF CARDIOVASCULAR SYSTEM Jonas Addae Medical Sciences, UWI REGULATION OF CARDIOVASCULAR SYSTEM Intrinsic Coupling of cardiac and vascular functions - Autoregulation of vessel diameter Extrinsic
REGULATION OF CARDIOVASCULAR SYSTEM Jonas Addae Medical Sciences, UWI REGULATION OF CARDIOVASCULAR SYSTEM Intrinsic Coupling of cardiac and vascular functions - Autoregulation of vessel diameter Extrinsic
Human Anatomy. Autonomic Nervous System
 Human Anatomy Autonomic Nervous System 1 Autonomic Nervous System ANS complex system of nerves controls involuntary actions. Works with the somatic nervous system (SNS) regulates body organs maintains
Human Anatomy Autonomic Nervous System 1 Autonomic Nervous System ANS complex system of nerves controls involuntary actions. Works with the somatic nervous system (SNS) regulates body organs maintains
Lecturer. Prof. Dr. Ali K. Al-Shalchy MBChB/ FIBMS/ MRCS/ FRCS 2014
 Lecturer Prof. Dr. Ali K. Al-Shalchy MBChB/ FIBMS/ MRCS/ FRCS 2014 Dorsal root: The dorsal root carries both myelinated and unmyelinated afferent fibers to the spinal cord. Posterior gray column: Long
Lecturer Prof. Dr. Ali K. Al-Shalchy MBChB/ FIBMS/ MRCS/ FRCS 2014 Dorsal root: The dorsal root carries both myelinated and unmyelinated afferent fibers to the spinal cord. Posterior gray column: Long
Table of Contents: Chapter 1 The organization of the spinal cord Charles Watson and Gulgun Kayalioglu
 Table of Contents: Chapter 1 The organization of the spinal cord Charles Watson and Gulgun Kayalioglu The gross anatomy of the spinal cord Spinal cord segments Spinal nerves Spinal cord gray and white
Table of Contents: Chapter 1 The organization of the spinal cord Charles Watson and Gulgun Kayalioglu The gross anatomy of the spinal cord Spinal cord segments Spinal nerves Spinal cord gray and white
Neural Basis of Motor Control
 Neural Basis of Motor Control Central Nervous System Skeletal muscles are controlled by the CNS which consists of the brain and spinal cord. Determines which muscles will contract When How fast To what
Neural Basis of Motor Control Central Nervous System Skeletal muscles are controlled by the CNS which consists of the brain and spinal cord. Determines which muscles will contract When How fast To what
Laboratory Manual for Comparative Anatomy and Physiology Figure 15.1 Transparency Master 114
 Neuron Capillary Astrocyte Microglial cell Neuron Fluid-filled cavity Process of oligodendrocyte Ependymal cells Brain or spinal cord tissue Myelin sheath Nerve fibers Figure 15.1 Transparency Master 114
Neuron Capillary Astrocyte Microglial cell Neuron Fluid-filled cavity Process of oligodendrocyte Ependymal cells Brain or spinal cord tissue Myelin sheath Nerve fibers Figure 15.1 Transparency Master 114
b. The groove between the two crests is called 2. The neural folds move toward each other & the fuse to create a
 Chapter 13: Brain and Cranial Nerves I. Development of the CNS A. The CNS begins as a flat plate called the B. The process proceeds as: 1. The lateral sides of the become elevated as waves called a. The
Chapter 13: Brain and Cranial Nerves I. Development of the CNS A. The CNS begins as a flat plate called the B. The process proceeds as: 1. The lateral sides of the become elevated as waves called a. The
Unit VIII Problem 3 Neuroanatomy: Brain Stem, Cranial Nerves and Scalp
 Unit VIII Problem 3 Neuroanatomy: Brain Stem, Cranial Nerves and Scalp - Brain stem: It is connected to the cerebellum and cerebral hemispheres. Rostral end of brain stem: diencephalon is the area which
Unit VIII Problem 3 Neuroanatomy: Brain Stem, Cranial Nerves and Scalp - Brain stem: It is connected to the cerebellum and cerebral hemispheres. Rostral end of brain stem: diencephalon is the area which
Nervous System. Student Learning Objectives:
 Nervous System Student Learning Objectives: Identify the primary parts of the neuron Identify the major structures of the central nervous system Identify the major structures of the peripheral nervous
Nervous System Student Learning Objectives: Identify the primary parts of the neuron Identify the major structures of the central nervous system Identify the major structures of the peripheral nervous
Reticular Formation George R. Leichnetz, Ph.D.
 Reticular Formation George R. Leichnetz, Ph.D. OBJECTIVES 1. To understand the anatomical and functional organization of the brainstem reticular formation into three general regions: median (raphe), medial
Reticular Formation George R. Leichnetz, Ph.D. OBJECTIVES 1. To understand the anatomical and functional organization of the brainstem reticular formation into three general regions: median (raphe), medial
Biological Bases of Behavior. 3: Structure of the Nervous System
 Biological Bases of Behavior 3: Structure of the Nervous System Neuroanatomy Terms The neuraxis is an imaginary line drawn through the spinal cord up to the front of the brain Anatomical directions are
Biological Bases of Behavior 3: Structure of the Nervous System Neuroanatomy Terms The neuraxis is an imaginary line drawn through the spinal cord up to the front of the brain Anatomical directions are
Characteristics of Rat Aortic Baroreceptors with Nonmedullated Afferent Nerve Fibers
 231 Characteristics of Rat Aortic Baroreceptors with Nonmedullated Afferent Nerve Fibers PETER THOREN, WILLIAM R. SAUM, AND ARTHUR M. BROWN SUMMARY Nervous activity was recorded from nonmedullated afferent
231 Characteristics of Rat Aortic Baroreceptors with Nonmedullated Afferent Nerve Fibers PETER THOREN, WILLIAM R. SAUM, AND ARTHUR M. BROWN SUMMARY Nervous activity was recorded from nonmedullated afferent
Birmingham B15 2TJ. myelinated primary afferent fibres of the sinus nerve terminated within. carotid sinus. (Received 6 November 1974)
 J. Physiol. (1975), 251, pp. 61-78 61 With 8 text-ftgure8 Printed in Great Britain THE SINUS NERVE AND BARORECEPTOR INPUT TO THE MEDULLA OF THE CAT By J. LIPSKI,* R. M. McALLEN AND K. M. SPYER From the
J. Physiol. (1975), 251, pp. 61-78 61 With 8 text-ftgure8 Printed in Great Britain THE SINUS NERVE AND BARORECEPTOR INPUT TO THE MEDULLA OF THE CAT By J. LIPSKI,* R. M. McALLEN AND K. M. SPYER From the
Volume expansion does not activate neuronal projections from the NTS or depressor VLM to the RVLM
 Volume expansion does not activate neuronal projections from the NTS or depressor VLM to the RVLM ANTHONY D. SHAFTON, ANDREW RYAN, BARRY MCGRATH, AND EMILIO BADOER Department of Medicine, Monash Medical
Volume expansion does not activate neuronal projections from the NTS or depressor VLM to the RVLM ANTHONY D. SHAFTON, ANDREW RYAN, BARRY MCGRATH, AND EMILIO BADOER Department of Medicine, Monash Medical
Anatomy & Physiology Central Nervous System Worksheet
 1. What are the two parts of the CNS? 2. What are the four functions of the CNS Anatomy & Physiology Central Nervous System Worksheet 3. What are the four functions of the meninges? (p430) 4. Starting
1. What are the two parts of the CNS? 2. What are the four functions of the CNS Anatomy & Physiology Central Nervous System Worksheet 3. What are the four functions of the meninges? (p430) 4. Starting
Supplementary Figure 1
 Supplementary Figure 1 Miniature microdrive, spike sorting and sleep stage detection. a, A movable recording probe with 8-tetrodes (32-channels). It weighs ~1g. b, A mouse implanted with 8 tetrodes in
Supplementary Figure 1 Miniature microdrive, spike sorting and sleep stage detection. a, A movable recording probe with 8-tetrodes (32-channels). It weighs ~1g. b, A mouse implanted with 8 tetrodes in
Microcircuitry coordination of cortical motor information in self-initiation of voluntary movements
 Y. Isomura et al. 1 Microcircuitry coordination of cortical motor information in self-initiation of voluntary movements Yoshikazu Isomura, Rie Harukuni, Takashi Takekawa, Hidenori Aizawa & Tomoki Fukai
Y. Isomura et al. 1 Microcircuitry coordination of cortical motor information in self-initiation of voluntary movements Yoshikazu Isomura, Rie Harukuni, Takashi Takekawa, Hidenori Aizawa & Tomoki Fukai
Chapter 11: Nervous System and Nervous Tissue
 Chapter 11: Nervous System and Nervous Tissue I. Functions and divisions of the nervous system A. Sensory input: monitor changes in internal and external environment B. Integrations: make decisions about
Chapter 11: Nervous System and Nervous Tissue I. Functions and divisions of the nervous system A. Sensory input: monitor changes in internal and external environment B. Integrations: make decisions about
Integrative Synaptic Mechanisms in the Caudal Ganglion of the Crayfish
 Integrative Synaptic Mechanisms in the Caudal Ganglion of the Crayfish JAMES B. PRESTON and DONALD KENNEDY ABSTRACT A study of activity recorded with intracellular micropipettes was undertaken in the caudal
Integrative Synaptic Mechanisms in the Caudal Ganglion of the Crayfish JAMES B. PRESTON and DONALD KENNEDY ABSTRACT A study of activity recorded with intracellular micropipettes was undertaken in the caudal
Nature Neuroscience: doi: /nn Supplementary Figure 1. Splenic atrophy and leucopenia caused by T3 SCI.
 Supplementary Figure 1 Splenic atrophy and leucopenia caused by T3 SCI. (a) Gross anatomy of representative spleens from control and T3 SCI mice at 28 days post-injury. (b and c) Hematoxylin and eosin
Supplementary Figure 1 Splenic atrophy and leucopenia caused by T3 SCI. (a) Gross anatomy of representative spleens from control and T3 SCI mice at 28 days post-injury. (b and c) Hematoxylin and eosin
Chapter 7. The Nervous System: Structure and Control of Movement
 Chapter 7 The Nervous System: Structure and Control of Movement Objectives Discuss the general organization of the nervous system Describe the structure & function of a nerve Draw and label the pathways
Chapter 7 The Nervous System: Structure and Control of Movement Objectives Discuss the general organization of the nervous system Describe the structure & function of a nerve Draw and label the pathways
Chapter 7. Objectives
 Chapter 7 The Nervous System: Structure and Control of Movement Objectives Discuss the general organization of the nervous system Describe the structure & function of a nerve Draw and label the pathways
Chapter 7 The Nervous System: Structure and Control of Movement Objectives Discuss the general organization of the nervous system Describe the structure & function of a nerve Draw and label the pathways
Chapter 16. APR Enhanced Lecture Slides
 Chapter 16 APR Enhanced Lecture Slides See separate PowerPoint slides for all figures and tables pre-inserted into PowerPoint without notes and animations. Copyright The McGraw-Hill Companies, Inc. Permission
Chapter 16 APR Enhanced Lecture Slides See separate PowerPoint slides for all figures and tables pre-inserted into PowerPoint without notes and animations. Copyright The McGraw-Hill Companies, Inc. Permission
Hole s Human Anatomy and Physiology Eleventh Edition. Chapter 10
 PowerPoint Lecture Outlines to accompany Hole s Human Anatomy and Physiology Eleventh Edition Shier Butler Lewis Chapter 10 Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction
PowerPoint Lecture Outlines to accompany Hole s Human Anatomy and Physiology Eleventh Edition Shier Butler Lewis Chapter 10 Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction
General Sensory Pathways of the Face Area, Taste Pathways and Hearing Pathways
 General Sensory Pathways of the Face Area, Taste Pathways and Hearing Pathways Lecture Objectives Describe pathways for general sensations (pain, temperature, touch and proprioception) from the face area.
General Sensory Pathways of the Face Area, Taste Pathways and Hearing Pathways Lecture Objectives Describe pathways for general sensations (pain, temperature, touch and proprioception) from the face area.
Nature Methods: doi: /nmeth Supplementary Figure 1. Activity in turtle dorsal cortex is sparse.
 Supplementary Figure 1 Activity in turtle dorsal cortex is sparse. a. Probability distribution of firing rates across the population (notice log scale) in our data. The range of firing rates is wide but
Supplementary Figure 1 Activity in turtle dorsal cortex is sparse. a. Probability distribution of firing rates across the population (notice log scale) in our data. The range of firing rates is wide but
ParasymPathetic Nervous system. Done by : Zaid Al-Ghnaneem
 ParasymPathetic Nervous system Done by : Zaid Al-Ghnaneem In this lecture we are going to discuss Parasympathetic, in the last lecture we took sympathetic and one of the objectives of last lecture was
ParasymPathetic Nervous system Done by : Zaid Al-Ghnaneem In this lecture we are going to discuss Parasympathetic, in the last lecture we took sympathetic and one of the objectives of last lecture was
Spinal Interneurons. Control of Movement
 Control of Movement Spinal Interneurons Proprioceptive afferents have a variety of termination patterns in the spinal cord. This can be seen by filling physiologically-identified fibers with HRP, so their
Control of Movement Spinal Interneurons Proprioceptive afferents have a variety of termination patterns in the spinal cord. This can be seen by filling physiologically-identified fibers with HRP, so their
Chapter 11 Introduction to the Nervous System and Nervous Tissue Chapter Outline
 Chapter 11 Introduction to the Nervous System and Nervous Tissue Chapter Outline Module 11.1 Overview of the Nervous System (Figures 11.1-11.3) A. The nervous system controls our perception and experience
Chapter 11 Introduction to the Nervous System and Nervous Tissue Chapter Outline Module 11.1 Overview of the Nervous System (Figures 11.1-11.3) A. The nervous system controls our perception and experience
(Received February 6, 1934.)
 218 6I2.327:6I2.826 THE EFFECTS OF HYPOTHALAMIC STIMULATION ON GASTRIC MOTILITY. BY J. BEATTIE AND D. SHE E HAN (Rockefeller Research Fellow). (From the Department of Anatomy, McGill University, Montreal.)
218 6I2.327:6I2.826 THE EFFECTS OF HYPOTHALAMIC STIMULATION ON GASTRIC MOTILITY. BY J. BEATTIE AND D. SHE E HAN (Rockefeller Research Fellow). (From the Department of Anatomy, McGill University, Montreal.)
Chapter 20 (2) The Heart
 Chapter 20 (2) The Heart ----------------------------------------------------------------------------------------------------------------------------------------- Describe the component and function of
Chapter 20 (2) The Heart ----------------------------------------------------------------------------------------------------------------------------------------- Describe the component and function of
Chapter 3. Structure and Function of the Nervous System. Copyright (c) Allyn and Bacon 2004
 Chapter 3 Structure and Function of the Nervous System 1 Basic Features of the Nervous System Neuraxis: An imaginary line drawn through the center of the length of the central nervous system, from the
Chapter 3 Structure and Function of the Nervous System 1 Basic Features of the Nervous System Neuraxis: An imaginary line drawn through the center of the length of the central nervous system, from the
-Ensherah Mokheemer. -Amani Nofal. -Loai Alzghoul
 -1 -Ensherah Mokheemer -Amani Nofal -Loai Alzghoul 1 P a g e Today we will start talking about the physiology of the nervous system and we will mainly focus on the Central Nervous System. Introduction:
-1 -Ensherah Mokheemer -Amani Nofal -Loai Alzghoul 1 P a g e Today we will start talking about the physiology of the nervous system and we will mainly focus on the Central Nervous System. Introduction:
(Received 10 April 1956)
 446 J. Physiol. (I956) I33, 446-455 A COMPARISON OF FLEXOR AND EXTENSOR REFLEXES OF MUSCULAR ORIGIN BY M. G. F. FUORTES AND D. H. HUBEL From the Department ofneurophysiology, Walter Reed Army Institute
446 J. Physiol. (I956) I33, 446-455 A COMPARISON OF FLEXOR AND EXTENSOR REFLEXES OF MUSCULAR ORIGIN BY M. G. F. FUORTES AND D. H. HUBEL From the Department ofneurophysiology, Walter Reed Army Institute
Autonomic Nervous System, Visceral Sensation and Visceral Reflexes Jeff Dupree, Ph.D.
 Autonomic Nervous System, Visceral Sensation and Visceral Reflexes Jeff Dupree, Ph.D. OBJECTIVES After studying the material of this lecture, the student should know the: 1. basic divisions of the autonomic
Autonomic Nervous System, Visceral Sensation and Visceral Reflexes Jeff Dupree, Ph.D. OBJECTIVES After studying the material of this lecture, the student should know the: 1. basic divisions of the autonomic
Shlgejl MATSUMOTO. First Department of Oral and Maxillofacial Surgery, Niigata University School of Dentistry, Niigata, 951 Japan
 Japanese Journal of Physiology, 37, 359-368, 1987 Effects of Temporal Trachea-Occlusion at the End of Expiration on Internal Intercostal Muscle Activity in the Rabbit Shlgejl MATSUMOTO First Department
Japanese Journal of Physiology, 37, 359-368, 1987 Effects of Temporal Trachea-Occlusion at the End of Expiration on Internal Intercostal Muscle Activity in the Rabbit Shlgejl MATSUMOTO First Department
Cellular Bioelectricity
 ELEC ENG 3BB3: Cellular Bioelectricity Notes for Lecture 24 Thursday, March 6, 2014 8. NEURAL ELECTROPHYSIOLOGY We will look at: Structure of the nervous system Sensory transducers and neurons Neural coding
ELEC ENG 3BB3: Cellular Bioelectricity Notes for Lecture 24 Thursday, March 6, 2014 8. NEURAL ELECTROPHYSIOLOGY We will look at: Structure of the nervous system Sensory transducers and neurons Neural coding
Nervous System. The Peripheral Nervous System Agenda Review of CNS v. PNS PNS Basics Cranial Nerves Spinal Nerves Reflexes Pathways
 Nervous System Agenda Review of CNS v. PNS PNS Basics Cranial Nerves Spinal Nerves Sensory Motor Review of CNS v. PNS Central nervous system (CNS) Brain Spinal cord Peripheral nervous system (PNS) All
Nervous System Agenda Review of CNS v. PNS PNS Basics Cranial Nerves Spinal Nerves Sensory Motor Review of CNS v. PNS Central nervous system (CNS) Brain Spinal cord Peripheral nervous system (PNS) All
PHY3111 Mid-Semester Test Study. Lecture 2: The hierarchical organisation of vision
 PHY3111 Mid-Semester Test Study Lecture 2: The hierarchical organisation of vision 1. Explain what a hierarchically organised neural system is, in terms of physiological response properties of its neurones.
PHY3111 Mid-Semester Test Study Lecture 2: The hierarchical organisation of vision 1. Explain what a hierarchically organised neural system is, in terms of physiological response properties of its neurones.
Experimental Physiology
 Exp Physiol 99.11 (214) pp 1453 1466 1453 Research Paper Research Paper Intrinsic chemosensitivity of rostral ventrolateral medullary sympathetic premotor neurons in the in situ arterially perfused preparation
Exp Physiol 99.11 (214) pp 1453 1466 1453 Research Paper Research Paper Intrinsic chemosensitivity of rostral ventrolateral medullary sympathetic premotor neurons in the in situ arterially perfused preparation
closely resembling that following an antidromic impulse [Eccles and
 185 6I2.833. 96 REFLEX INTERRUPTIONS OF RHYTHMIC DISCHARGE. By E. C. HOFF, H. E. HOFF AND D. SHEEHAN1. (New Haven, Conn.) (From the Laboratory of Physiology, Yale University School of Medicine.) (Received
185 6I2.833. 96 REFLEX INTERRUPTIONS OF RHYTHMIC DISCHARGE. By E. C. HOFF, H. E. HOFF AND D. SHEEHAN1. (New Haven, Conn.) (From the Laboratory of Physiology, Yale University School of Medicine.) (Received
PART I. Disorders of the Heart Rhythm: Basic Principles
 PART I Disorders of the Heart Rhythm: Basic Principles FET01.indd 1 1/11/06 9:53:05 AM FET01.indd 2 1/11/06 9:53:06 AM CHAPTER 1 The Cardiac Electrical System The heart spontaneously generates electrical
PART I Disorders of the Heart Rhythm: Basic Principles FET01.indd 1 1/11/06 9:53:05 AM FET01.indd 2 1/11/06 9:53:06 AM CHAPTER 1 The Cardiac Electrical System The heart spontaneously generates electrical
The Nervous System PART A
 7 The Nervous System PART A PowerPoint Lecture Slide Presentation by Jerry L. Cook, Sam Houston University ESSENTIALS OF HUMAN ANATOMY & PHYSIOLOGY EIGHTH EDITION ELAINE N. MARIEB Structural Classification
7 The Nervous System PART A PowerPoint Lecture Slide Presentation by Jerry L. Cook, Sam Houston University ESSENTIALS OF HUMAN ANATOMY & PHYSIOLOGY EIGHTH EDITION ELAINE N. MARIEB Structural Classification
Skin types: hairy and glabrous (e.g. back vs. palm of hand)
 Lecture 19 revised 03/10 The Somatic Sensory System Skin- the largest sensory organ we have Also protects from evaporation, infection. Skin types: hairy and glabrous (e.g. back vs. palm of hand) 2 major
Lecture 19 revised 03/10 The Somatic Sensory System Skin- the largest sensory organ we have Also protects from evaporation, infection. Skin types: hairy and glabrous (e.g. back vs. palm of hand) 2 major
