Neither age at repair nor previous palliation affects outcome in tetralogy of Fallot repair
|
|
|
- Lorena Briggs
- 5 years ago
- Views:
Transcription
1 European Journal of Cardio-Thoracic Surgery 45 (2014) doi: /ejcts/ezt307 Advance Access publication 12 June 2013 ORIGINAL ARTICLE a b c Neither age at repair nor previous palliation affects outcome in tetralogy of Fallot repair Branko Mimic a,b,katel.brown a, Nilesh Oswal a, Jacob Simmonds a, Tain-Yen Hsia a, Victor T. Tsang a, Marc R. De Leval c and Martin Kostolny a, * Cardiothoracic Surgery Department, Great Ormond Street Hospital, London, UK Cardiac Surgery Department, University Children s Hospital, Belgrade, Serbia Cardiothoracic Surgery Department, Harley Street Clinic, London, UK * Corresponding author. Cardiothoracic Surgery Department, Great Ormond Street Hospital for Children, Great Ormond Street, London WC1N 3JH, UK. Tel: ; fax: ; kostom@gosh.nhs.uk (M. Kostolny). Received 18 December 2012; received in revised form 16 April 2013; accepted 29 April 2013 Abstract OBJECTIVES: The study aimed to evaluate the results following complete repair of tetralogy of Fallot (TOF) in relation to age at surgery and to assess the role of palliation in the current era. METHODS: A retrospective review of 251 consecutive patients with TOF repaired between 2003 and 2011 at the Great Ormond Street Hospital was performed. Children were divided into two groups: Group A, younger than 6 months (n = 78) and B, older than 6 months (n = 173). Early clinical outcomes and reoperation/reintervention rates were studied as well as indication for a palliation. RESULTS: There was 1 (0.4%) early and 1 (0.4%) late death after a median follow-up time of 4.5 years. Forty-three patients (17%) underwent repair after initial palliation with inter-stage mortality of 5%. Groups A and B were similar in terms of surgical approach, postoperative complications and length of stay. Significant differences were found in terms of more frequent use of a transannular patch (P = 0.05), longer surgeries (P = 0.02) and a greater proportion of palliated patients (P = 0.002) in older patients. There was no difference in rates of reoperation/reintervention between groups and following both primary and staged repair. Palliated patients were more symptomatic (duct-dependent pulmonary blood flow; P < 0.01, cyanotic spells; P < 0.01), had more extracardiac/genetic anomalies (P < 0.01), coronary anomalies (P = 0.015) and significantly smaller pulmonary annulus, right pulmonary artery (RPA) and left pulmonary artery (LPA) Z-scores (P < 0.01 for all). CONCLUSION: Age at complete repair was not linked to early clinical outcome or reoperation/reintervention rate. Palliative procedures postponed the timing of complete repair, but did not increase the reintervention rate. Keywords: Tetralogy of Fallot Palliation Primary repair INTRODUCTION Although the history of tetralogy of Fallot (TOF) repair extends over six decades, controversy concerning the optimal technique and timing of the corrective surgery still remains. In the current era, primary surgical repair in infants continues to gain increasing acceptance [1, 2]. Proponents of primary neonatal repair in symptomatic patients cite factors such as prevention of time-related end-organ damage from cyanosis, removal of stimulus for right ventricular (RV) hypertrophy and improved lung development to support this policy [3 5]. In addition, this approach avoids the risks of shunt-related complications, namely shunt thrombosis, congestive heart failure, pulmonary hypertension and pulmonary arterial distortion. However, a number of surgeons undertaking a neonatal TOF repair use a trans-ventricular approach [1, 6] and deep hypothermic circulatory arrest [6]. Furthermore, transannular Presented at the 26th Annual Meeting of the European Association for Cardio- Thoracic Surgery, Barcelona, Spain, October patch (TAP) reconstruction of the right ventricular outflow tract (RVOT) was reported to be used more frequently in neonates and young infants [1, 7], with possible deleterious effects of long-term pulmonary regurgitation (PR) [8]. Some have advocated palliating symptomatic neonates to avoid TAP and preserve the pulmonary valve function at the time of complete repair [9, 10]. We have taken the approach of elective primary repair of infants without severe cyanosis around the age of 6 months and staged repair of symptomatic neonates and young infants with unfavourable anatomy. In this study, the relationship between age at corrective surgery and outcome was assessed, and the indications for a staged rather than primary repair were explored. MATERIALS AND METHODS Patient population All patients undergoing either palliation or complete repair of TOF between 1 January 2003 and 1 April 2011 were identified from The Author Published by Oxford University Press on behalf of the European Association for Cardio-Thoracic Surgery. All rights reserved.
2 B. Mimic et al. / European Journal of Cardio-Thoracic Surgery 93 our institution patient database. Data were obtained from medical records and echocardiographic, surgical and catheterization reports. Patients with pulmonary atresia, absent pulmonary valve, major systemic to pulmonary collaterals, double outlet right ventricle with aortic-mitral discontinuity, associated atrioventricular septal defect and those who required concomitant tracheal surgery were excluded. TOF with pulmonary stenosis was diagnosed by trans-thoracic echocardiography and occasionally supplemented by cardiac catheterization to further delineate the anatomy of the pulmonary arteries and/or define the presence of major systemic to pulmonary collaterals. Cardiac catheterization was utilized more often in patients who had previous palliation (49 vs 18%; P = 0.02). Based on age at repair, children were divided into two groups: Group A, younger than 6 months (n = 78, median age 132 days; range , mean weight 5.6 ± 1.0 kg) and Group B, >6 months (n = 173, median age 315 days; range , mean weight 8.4 ± 2.8 kg). Palliation was reserved for symptomatic neonates with sustained cyanosis (saturations <70%), cyanotic spells and ductdependent pulmonary blood flow and those who had unfavourable anatomy such as branch pulmonary artery (PA) hypoplasia or coronary anomalies. Severe non-cardiac anomalies or syndromes might have been a contributing factor in decision-making. This study was registered and approved by the Research & Development office at the Institute of Child Health, London with ethical approval for the use of retrospective, anonymized data. Surgical technique Primary repairs were performed on moderate hypothermia cardiopulmonary bypass (CPB) with aortic and bicaval cannulation and intermittent, antegrade blood cardioplegia. A vent was routinely inserted through the right upper pulmonary vein into the left atrium. The transatrial/transpulmonary approach was the preferred method of repair. The RVOT inspection, and excision/transection of obstructive parietal and septal bundles began through the tricuspid valve. After completion of the infundibular myectomy, the diameters of the RVOT and the pulmonary valve were assessed with Hegar dilators and if found less than the two standard deviations (SDs) for the patient s age and weight [11], the main PA was opened longitudinally. Further RVOT obstruction (RVOTO) relief was performed by additional muscle bundles transection and/or pulmonary valvotomy. If the infundibulum had a long tunnel-like stenosis, a small infundibular incision was performed without incising the pulmonary valve ring. If necessary (Z-score < 3), the pulmonary arteriotomy was extended proximally and a TAP was inserted. Branch pulmonary arterioplasty was performed if indicated either by a separate patch or by extension of main PA (MPA) patch. The ventricular septal defect (VSD) was closed through the tricuspid valve in all but 5 patients. Interatrial communications were routinely closed in 238 (95%) patients. After the coming of CPB, the adequacy of the repair was assessed by measuring the peak RV/left ventricle (LV) pressure ratio in 210 of 251 (84%) patients. An RV/LV pressure ratio of <0.75 was considered as acceptable. Transoesophageal echocardiography was routinely used to assess the repair. If the residual obstruction was demonstrated at the level of the annulus with RV/LV pressure ratio of >0.75, then a TAP was inserted. Residual obstruction at the sub-valvular level was managed by either further muscle bundle resection or ventriculotomy with the placement of infundibular patch. This was required in 31 patients (12%). Outcome was evaluated in March 2012 in terms of patient survival using the survival status recorded in the UK care record service, and the occurrence of any reinterventions including surgical or catheter based was noted since all such procedures are recorded in the database. The follow-up echocardiograms were reviewed on 180/251 (72%) with the mean follow-up time of 2.64 years (range, ). Missing echocardiographic data were those from patients under remote follow-up either abroad or at distant locations in the UK. Statistical analysis Variables are expressed as the mean ± SD or median and range as appropriate. Continuous variables were compared with twotailed, unpaired Student s t-test or Mann Whitney U-test, as applicable. Categorical data were compared with two-tailed Pearson χ 2 test or Fisher s exact test, as applicable. For all analyses, a P-value of <0.05 was considered statistically significant. Freedoms from time-related events (reoperation and reintervention) were analysed using the Kaplan Meier method and for the analysis of differences in the course of survival, the log-rank test was used. Multivariate Cox regression analysis was used to define independent predictors for reoperation and reintervention. RESULTS Demographic and operative results The study included 251 consecutive patients with TOF (138 males, 55%) who underwent complete repair. Extracardiac malformations were found in 37 (15%) patients and a congenital or genetic syndrome was diagnosed in 36 (14%). The median age at repair was 248 days (range, ) and the median weight was 7.2 kg (range ), with the oldest patient having immigrated to the UK shortly before repair was undertaken. Forty-three patients (17%) underwent corrective repair after initial palliation. The demographic and operative data for all patients and the comparison between age groups A and B are depicted in Table 1. The older patients received a TAP more frequently (A: 44 vs B: 57%; P = 0.05) and required longer cross clamp (A: 49.8 ± 19.2 vs B: 55.9 ± 20.6; P = 0.02) and CPB time (A: 96.6 ± 34.5 vs B: ± 35.6; P = 0.03). More patients from age group B had previous palliation (A: 6 vs B: 22%; P = 0.002). The majority in both groups received a right ventriculotomy to further enlarge the RVOT (A: 64 vs B: 72%; P = 0.23). Higher TAP and increased right ventriculotomy rate in patients repaired after 6 months did not result in significantly different RV/LV pressure ratio immediately after correction (A: 0.58 vs B: 0.56; P = 0.14). Different types of PA plasty at the time of complete repair were equally distributed between groups (A: 9 vs B: 15%; P = 0.19). Early outcomes There was 1 (0.4%) early (hospital) death in a child with dysmorphic features who underwent complete repair with a TAP at the age of 19 months and had a major conal branch crossing the RVOT. The patient died of cardiac failure and multiorgan dysfunction on CONGENITAL
3 94 B. Mimic et al. / European Journal of Cardio-Thoracic Surgery Table 1: Demographic and operative data and comparison between Groups A and B Variable analysed Group A (n = 78) Group B (n = 173) Overall (n = 251) P-value Preoperative data Male/female 45/33 93/80 138/ χ 2 Age (months), median (range) 4.4 ( ) 10.5 ( ) 8.3 ( ) <0.01 MW Weight (kg), mean ± SD 5.6 ± ± ± 2.7 <0.01 T Right aortic arch, n (%) 11 (14) 20 (12) 31 (12) 0.57 χ 2 Associated cardiac anomalies, n (%) 9 (12) 27 (16) 36 (14) 0.39 χ 2 Associated non-cardiac anomalies, n (%) 9 (12) 26 (15) 37 (15) 0.39 χ 2 Syndromes, n (%) 8 (10) 28 (16) 36 (14) 0.21 χ 2 All coronary anomalies, n (%) 5 (6) 17 (10) 22 (9) 0.47 χ 2 Previous palliation, n (%) 5 (6) 38 (22) 43 (17) χ 2 Operative data (complete repair) TAP plasty, n (%) 34 (44) 98 (57) 132 (53) 0.05 χ 2 Non-TAP with ventriculotomy, n (%) 16 (21) 27 (15) 43 (17) 0.34 χ 2 Non-TAP without ventriculotomy, n (%) 28 (35) 48 (28) 76 (30) 0.25 χ 2 Branch pulmonary arterioplasty, n (%) 7 (9) 26 (15) 33 (13) 0.19 χ 2 Bypass time (min), mean ± SD 96.6 ± ± ± MW AXCT (min), mean ± SD 49.8 ± ± ± MW RV/LV pressure ratio, mean ± SD 0.58 ± ± ± MW TAP: transannular patch; AXCT: aortic cross-clamp time; RV/LV: right ventricle/left ventricle; MW: Mann Whitney U-test; χ 2 : Pearson χ 2 test; F: Fisher s exact test; T: Student s t-test. Bold figures indicate statistically significant differences. Table 2: Postoperative data following complete repair and comparison between groups Variable analysed Group A (n = 78) Group B (n = 173) Overall (n = 251) P-value Number of patients (%) Number of patients (%) Number of patients (%) Hours intubated, median (range) 22 (0 520) 23 (0 714) 23 (0 714) 0.63 MW ICU stay, median hours (range) 68 (22 713) 51 (0 9682) 55.5 ( ) 0.09 MW Early reoperation/reintervention 1 (1) 8 (5) 9 (4) 0.28 F Early death 0 (0) 1 (0.6) 1 (0.4) 1.00 F Delay sternal closure 4 (5) 5 (3) 9 (4) 0.46 F Major bleeding required re-exploration 2 (3) 11 (6) 13 (5) 0.35 F Pleural drainage 4 (5) 9 (5) 13 (5) 0.97 F ECMO 2 (3) 2 (1) 4 (2) 0.59 F Peritoneal dialysis 14 (18) 28 (16) 42 (17) 0.73 χ 2 JET 12 (15) 27 (16) 39 (15) 0.79 χ 2 Amiodaron 10 (13) 22 (13) 32 (13) 0.98 χ 2 Temporary pacing 14 (18) 39 (23) 53 (21) 0.29 χ 2 LCOS 8 (10) 11 (6) 19 (8) 0.28 χ 2 Sepsis 1 (1) 4 (2) 5 (2) 1.00 F Chylothorax 4 (5) 6 (3) 10 (4) 0.51 F NO 2 (3) 6 (3) 8 (3) 1.00 F NEC 1 (1) 1 (0.6) 2 (1) 0.52 F Brain infarct 1 (1) 0 (0) 1 (0.4) 0.31 F PM implantation 0 (0) 2 (1) 2 (1) 1.00 F Diaphragm plication 1 (1) 2 (1) 3 (1) 1.00 F ECMO: extracorporeal membrane oxygenation; JET: junctional ectopic tachycardia; LCOS: low cardiac output syndrome; NO: nitric oxide; NEC: necrotizing enterocolitis; PM: pace maker; MW: Mann Whitney U-test; F: Fisher s exact test; χ 2 : Pearson χ 2 test. the sixth postoperative day. The entire cohort had a median duration of mechanical ventilator support of 23 h (range, 0 714), and a median postoperative intensive care unit (ICU) stay of 55.5 h (range, ), with no observed differences between Groups A and B. Postoperative complications (Table 2) were also similar between Groups A and B. Nine of 251 patients required at total of 11 early reoperations and 1 reintervention in the catheter lab during the same admission. Six patients had reintervention(s) for a residual RVOTO or branch PA stenosis, 1 patient had a residual VSD, 1 had a repair of ruptured tricuspid valve chordae and 1 had a closure of residual patent ductus arteriosus. Late outcomes There was 1 (0.4%) late (after discharge) death in an ex-premature baby with DiGeorge syndrome who underwent primary repair with a TAP at the age of 15 months with prolonged ICU stay (8
4 B. Mimic et al. / European Journal of Cardio-Thoracic Surgery 95 Table 3: Indication for and type of late reoperation and reintervention Reoperations Number of reoperations Reinterventions Number of reinterventions RVOTO relief with muscle bundles resection or TAP plasty 3 Balloon dilatation of LPA and RPA stenosis 4 RV-PA conduit insertion for residual RVOTO and/or pulmonary arteries 9 Balloon dilatation of LPA stenosis 3 stenosis RV-PA conduit insertion for free PR and RV dilatation 3 Balloon dilatation of RPA stenosis 0 RV-PA conduit replacement for conduit obstruction 2 Balloon dilatation of RVOT 3 Aortopexy 1 Balloon dilatation of RPA stent 2 Vascular ring repair 1 Balloon dilatation of LPA stent 4 Balloon dilatation of Hanckok conduit 1 stenosis Stenting of RPA and LPA stenosis 2 Stenting of LPA stenosis 7 Stenting of RPA stenosis 2 ASD device closure 1 PA band dilatation 1 Total RVOTO: right ventricle outflow tract obstruction; RV: right ventricle; PR: pulmonary regurgitation; TAP: transannular patch; RV-PA: right ventricle to pulmonary artery; RPA: right pulmonary artery; LPA: left pulmonary artery; PA: pulmonary artery. days). The echocardiography before discharge showed grossly impaired RV function and the child died at home after a sudden event 18 months postoperatively. Twenty-nine patients underwent 19 late reoperations and 30 late catheter based reinterventions after a median time of 18 (range, 1 61) and 10 months (range, ), respectively. Indications for and types of surgical and catheter reintervention are given in Table 3. Both 5-year freedom from reoperation and freedom from any reoperation and reintervention were not significantly different between the 2 patient groups (Fig. 1). Univariate Cox regression models for the hazard of a first reintervention are given in Table 4: age group, palliations and congenital syndromes did not increase the risk of reintervention. Univariate analysis indicated the hazard was greater with TAP (P = 0.04), a second run of bypass (P =0.03) and arterioplasty of branch pulmonary arteries at the time of complete repair (P < 0.01). A Cox multiple regression model indicated that only pulmonary arterioplasty was significant when all factors were considered together (P < 0.01). At a median echocardiographic follow-up of 2.6 years, the mean peak RVOT velocity in Groups A and B was similar at 2.23 ± 0.74 and 2.23 ± 0.73 m/s, respectively (P = 0.846). The mean value of PR grade (coded as follows: none-0, mild-1; moderate-2; severe-3; free-4) was estimated as severe or free in 50% of patients. TAP was associated with significantly more severe PR: 75% of these had severe or free vs 24% of those without TAP, P < Despite finding older patients to be more likely to receive a TAP, the proportion with severe PR was not significantly greater in Group B than A (severe or free PR 54 vs 43%, respectively; P = 0.33). All but 14 patients had either normal or mildly impaired RV function at the last follow-up; however, there was no significant difference between Groups A and B in terms of RV function. The palliated group During the study period, 47 patients underwent palliation initially, of whom 43 underwent complete repair (2 patients died interstage and 2 were awaiting repair at the time of study closure). Figure 1: Actuarial freedom from any late reoperation and reintervention and comparison between age groups. Eight of these 43 had at least one shunt palliation performed at another institution. There were 35 surgical and 4 interventional palliations performed at our institution (Table 5). For shunt palliation, a sternotomy was the preferred approach (29/34 shunts). At the shunt operation, 3 of 34 had a patch enlargement of the proximal LPA. Indications for palliation were one or a combination of following factors: sustained cyanosis (<70%), cyanotic spells, ductdependent pulmonary blood flow, hypoplastic branch pulmonary arteries (right pulmonary artery (RPA) and/or left pulmonary artery (LPA) Z-score < 2), weight <3 kg, prematurity and anomalous coronary (LAD/RCA) crossing RVOT. The median age at palliation was 27 days (range, 1 296), median weight 3.3 kg (range, ). Thirty-seven of 39 (95%) had two and more indications for palliation. Three of 4 patients who underwent shunt palliation due to major coronary anomaly (LAD/RCA) crossing the RVOT were >3 months old at the time of palliation. A comparison CONGENITAL
5 96 B. Mimic et al. / European Journal of Cardio-Thoracic Surgery Table 4: Cox regression model for the hazard of a first reintervention of any type Univariate Cox regression Hazard ratio for first reintervention = surgery + catheter (95% CI) P-value Group A vs B 0.87 (0.44, 1.75) 0.70 Palliated vs non-palliated 1.41 (0.64, 3.11) 0.38 Syndromic vs non-palliated 1.43 (0.62, 3.25) 0.40 TAP 2.12 (1.04, 4.33) 0.04 Monocusp valve 0.67 (0.09, 4.95) 0.70 Pulmonary arterioplasty 7.14 (3.69, 13.81) <0.001 CPB second run 2.35 (1.07, 5.17) 0.03 RVOT patch 0.47 (0.14, 1.53) 0.21 MPA patch 0.76 (0.32, 1.84) 0.55 Multiple Cox regression, including the three significant factors CPB second run 1.54 (0.68, 3.45) 0.30 TAP 1.27 (0.60, 3.44) 0.53 Pulmonary arterioplasty 6.09 (2.96, 12.54) <0.001 TAP: transannular patch; RVOT: right ventricle outflow tract; PA: pulmonary artery; CPB: cardiopulmonary bypass. Bold figures indicate risk factors for a reintervention. Table 5: Palliative procedures performed at our institution Palliation type No of patients Age (days) at palliation, median (range) Surgical Right modified BT shunt 24 Left modified BT shunt 4 Right modified BT shunt + LPA 3 27(1 296) plasty Central systemic to pulmonary 3 shunt RVOT patch 1 61 Overall surgical palliation (1 296) Interventional Balloon pulmonary valvotomy 2 9 (4 14) Stenting of RVOT (13 58) Overall interventional palliation (4 58) BT: Blalock Taussig; LPA: left pulmonary artery; RVOT: right ventricle outflow tract. between patients who underwent primary and staged repair at our institution is given in Table 6. Patients who required staged repair were more symptomatic (duct-dependent pulmonary blood flow 29 vs 0%; P < 0.01, cyanotic spells 34 10%; P < 0.01) and had significantly more extracardiac anomalies, syndromes (43 vs 20%; P < 0.01), and coronary anomalies (20 vs 7%; P = 0.015). Furthermore, they represent the subset of children with less favourable anatomy characterized by significantly smaller pulmonary annulus, RPA and LPA Z-scores (P < 0.01 for all). A palliative procedure increased the age at complete repair, with median time from palliation to repair of 375 days (range ). Only 5 palliated patients had complete repair before 6 months of age. Although palliated patients had more TAP (83 vs 48%; P < 0.01) at the time of corrective surgery than those who underwent primary repair, they did not experience a significantly higher reoperation/reintervention rate (Fig. 2). There was 1 early shunt-related death and 1 late death in a child with VACTERL association and severely hypoplastic pulmonary arteries who died of a non-cardiac reason 1 year after palliation. There were five major complications after surgical palliation. One right shunt occlusion and infection, which required taking down of the shunt followed by left shunt insertion, extracorporeal membrane oxygenation (ECMO) support and stenting of the RPA. There were 3 mediastinal explorations due to major bleeding and 1 cardiac arrest. DISCUSSION This study reviews a single-centre experience with the surgical treatment of TOF over the most recent era ( ). Inclusion of patients for study was based on a diagnosis of TOF rather than the operative procedure, in order to optimally elucidate important mortalities and morbidities at all stages, and thus reflect on our practice. During the study period, elective primary repair was considered for all patients 6 months of age. Staged repair was reserved for symptomatic neonates with sustained cyanosis and duct-dependent pulmonary blood flow. Unfavourable coronary anatomy, hypoplastic branch pulmonary arteries, associated extracardiac anomalies and genetic syndromes also affected our decision in favour of initial palliation. Optimal age at repair The elective repair of an asymptomatic infant with TOF has been proposed as optimally timed between 3 and 12 months of age. It has been shown that infants undergoing primary repair before 3 months of age experienced greater morbidity than older infants; more frequent need for peritoneal drainage, longer times to extubation and longer stays in the ICU [12, 13]. Bove et al. [14] have shown that decreasing the age at repair, at least to the age of 3 months, does not affect the early clinical outcome. Furthermore, they proposed complete repair between 3 and 6 months of age, irrespective of symptomatic status or previous palliation. They believe this age range enhances the opportunity of relieving the hypoplastic RVOT with minimal transannular incision, with the risk of revision being required later, but highlight the long-term protective effect of restrictive physiology on RV function. These conclusions are in keeping with our study, which demonstrated that children undergoing primary repair before the age of 6 months had equivalent mid-term results in terms of recurrent RVOTO and RV function. Furthermore, younger age at repair was not linked to any increased perioperative morbidity measure, nor was it linked to the risk of reinterventions. Whether repair at a younger age increases the need for the use of a TAP remains uncertain [12, 15, 16]. The higher incidence of TAP in the older patients in our series might be explained by progressive right ventricle hypertrophy and reduced PA development leading to more aggressive RVOTO relief at the time of complete repair. However, it is more likely that this finding is the result of our policy to correct the anatomically and physiologically favourable patients while palliating the others. The fact that palliation postponed complete repair with a median inter-stage period of 375 days (all but 5 palliated patients underwent complete repair
6 B. Mimic et al. / European Journal of Cardio-Thoracic Surgery 97 Table 6: Comparison between patients that underwent primary and staged repair (palliated patients that referred from other institutions were excluded; Z-scores for palliated patients were collected before palliation) Variable analysed Staged repair (n = 35) Primary repair (n = 208) P-value Preoperative data Spells, n (%) 12 (34) 21 (10) <0.01 (χ 2 ) DAP dependant pulmonary circulation, n (%) 10 (29) 0 (0) <0.01 (F) Prematurity, n (%) 8 (23) 24 (12) 0.07 (χ 2 ) Extracardiac anomalies/syndromes, n (%) 15 (43) 42 (20) <0.01 (χ 2 ) All coronary anomalies, n (%) 7 (20) 15 (7) (χ 2 ) (LAD from RCA/RCA from LCA) (3/1) (2/0) Pulmonary annulus Z-score, mean ± SD 4.34 ± ± 3.01 <0.01 (T) RPA Z-score, mean ± SD 2.27 ± ± 1.24 <0.01 (T) LPA Z-score, mean ± SD 2.09 ± ± 1.58 <0.01 (T) Catheterization prior complete repair, n (%) 21 (60) 32 (15) <0.01 (χ 2 ) Operative data (complete repair) TAP, n (%) 29 (83) 99 (48) <0.01 (χ 2 ) Bypass time (min), mean ± SD ± ± (MW) AXCT (min), mean ± SD 54.9 ± ± (MW) Branch PA plasty, n (%) 8 (23) 24 (12) 0.07 (χ 2 ) Follow-up data Reoperation and/or reintervention, n (%) 6 (17) 23 (11) 0.45 (χ 2 ) DAP: ductus arteriosus persistens; LAD: left anterior descending; RCA: right coronary artery; TAP: transannular patch; AXCT: aortic cross-clamp time; RPA: right pulmonary artery; LPA: left pulmonary artery; MW: Mann Whitney U-test; χ 2 : Pearson χ 2 test; F: Fisher s exact test.; T: Student s t-test. Bold figures indicate statistically significant differences. Figure 2: Actuarial freedom from any late reoperation/reintervention and comparison between primary and staged repair. after the age of 6 months) and that palliated patients need significantly more TAP than those undergoing primary repair (83 vs 48%) suggest that the severity of RVOTO, rather than age at repair in isolation, determines the frequency with which TAP is used. This finding correlates with other studies that have suggested that age at repair does not influence the incidence of use of TAP [17] and that palliated patients required more TAP [9]. Significantly longer cross clamp and CPB times in the older age group in our series might be the result of similar factors. Staged vs primary repair The management of the symptomatic neonates and young infants under 3 months of age is more controversial. Options for these children include surgical or interventional palliation or early primary repair. Kanter et al. [18] have recently shown an early mortality of 1/17 in the shunted group and no mortality among 20 primary repaired neonates. In the same study, the actuarial freedom from death or reoperation at 5 years was 69% for the primary repaired and 77% for the shunted neonates. A multiinstitutional report from 2002 [19] showed that for patients 3 months of age or less, the mortality for a shunt vs a repair was similar (6.2 vs 8.0%). Knott-Craig et al. [20] demonstrated a relatively high mortality rate of 11% for staged repair, whereas Sfyridis et al.[21] showed zero operative and inter-stage mortality in shunted patients. Our overall inter-stage mortality of 5% (2/39) is consistent with other reports showing relatively low mortality rates with staged repair of TOF [21, 22]. Furthermore, our strategy is in accordance with authors who advocate an individualized approach rather than primary repair in all TOF patients [7, 10, 13]. The different types of surgical and interventional palliation utilized in our institution express an effort to offer an optimal palliation to each patient, based on their clinical condition, weight and underlying anatomy. Symptomatic neonates with branch pulmonary arteries of relatively reasonable size (>3 mm) were more likely to receive right or left shunt palliation, whereas those with severely hypoplastic pulmonary arteries underwent either a central systemic to pulmonary shunt or RVOT patch. A RVOT stent has recently become a possible alternative for symptomatic neonates with critical PV stenosis [23]. Reoperation and reintervention Freedom from surgical or catheter reintervention after TOF repair in early infancy in published series [16, 24, 25] ranges from 79 to 96% at 20 years. Bove et al. [14] demonstrated a 3-year freedom from reoperation for residual or recurrent RVOTO of 86%. Tamesberger et al. [1] has shown that the majority of CONGENITAL
7 98 B. Mimic et al. / European Journal of Cardio-Thoracic Surgery reintervention (75%) is due to stenotic LPA. The 5-year freedom from reoperation and reintervention for any cause of 86% (equivalent between palliated and primary repair groups, and unrelated to age at repair) in our series appears comparable to other published data [13, 16, 21]. Twenty of 30 (66%) reinterventions in our series occurred because of a LPA stenosis, which contributed as an indication for reoperation or reintervention in 21 of 29 patients. Furthermore, branch pulmonary arterioplasty at the time of complete repair regardless of the technique employed was the strongest predictor of reintervention and reoperation in our experience. Correction with a TAP did not influence late reoperation/reintervention rate in our series. This correlates with the study by Alexiou et al. [16], which demonstrated that the use of a TAP is not a significant factor for any further operation and intervention for pulmonary valve replacement (PVR). Only three patients received an RV-PA conduit for free PR associated with significant RV dilatation. On the other hand, a relatively high proportion of PVR in our patients undergoing reoperation (12/15) might be a result of RVOTO at multiple levels (from infundibulum to proximal branch pulmonary arteries), usually combined with free PR in children who had received a TAP. However, further follow-up is mandatory to assess effect of TAP on long-term RV function and the necessity for PVR. Limitations This study is limited by its retrospective nature. Echocardiographic follow-up data are incomplete with relatively short median followup time. Patients were divided into two groups with a cut-off age at repair of 6 months being rather arbitrary. Since the palliated group represents the subset of patients with less favourable anatomy and physiology often associated with extracardiac or coronary anomalies, we are not able to compare the outcome of palliation and early primary repair. Consequently, we do not know what the outcome would have been if we had done repair instead of palliation for these high-risk patients. Our strategy and conclusions Currently, for symptomatic patients, primary repair is considered in the absence of coronary anomalies, small pulmonary arteries and major extracardiac problems irrespective of age. For asymptomatic patients, a repair is undertaken ideally between 4 and 6 months of age. Age at complete repair was not linked to early clinical outcomes or reoperation/reintervention rate. Palliation postponed the timing of complete repair, and palliated patients were more likely to receive a TAP, however, this was unrelated to the reintervention rate. Our strategy to selectively palliate patients results in as good an outcome as primary repair. Primary repair at an age <6 months gave good mid-term results in patients with favourable anatomy. It is possible to operate earlier and leave staged repair for the subset of symptomatic patients with the most unfavourable anatomy. Conflict of interest: none declared. REFERENCES [1] Tamesberger MI, Lechner E, Mair R, Hofer A, Sames-Dolzer E, Tulzer G. Early Primary Repair of Tetralogy of Fallot in Neonates and Infants Less Than Four Months Of Age. Ann Thorac Surg 2008;86: [2] Pigula FA, Khalil PN, Mayer JE, del Nido PJ, Jonas RA. Repair of tetralogy of Fallot in neonates and young infants. Circulation 1999;100:II [3] Rabinovitch M, Herrera-deLeon V, Castaneda AR, Reid L. Growth and development of the pulmonary vascular bed in patients with tetralogy of Fallot with or without pulmonary atresia. Circulation 1981;64: [4] Derby CD, Pizarro C. Routine primary repair of tetralogy of Fallot in the neonate. Expert Rev Cardiovasc Ther 2005;3: [5] Walsh EP, Rockenmacher S, Keane JF, Hougen TJ, Lock JE, Castaneda AR. Late results in patients with tetralogy of Fallot repaired during infancy. Circulation 1988;77: [6] Hirsch JC, Mosca RS, Bove EL. Complete repair of tetralogy of Fallot in the neonate: results in the modern era. Ann Surg 2000;232: [7] Pozzi M, Dipesh BT, Kitchiner D, Arnold R. Tetralogy of Fallot: what operation, at which age. Eur J Cardiothorac Surg 2000;17: [8] Norgard G, Gatzoulis MA, Moraes F, Lincoln C, Shore DF, Shinebourne EA et al. Relationship between type of outflow tract repair and postoperative right ventricle diastolic physiology in tetralogy of Fallot. Implication for long-term outcome. Circulation 1996;94: [9] Stewart RD, Backer CL, Young L, Mavroudis C. Tetralogy of Fallot: Results of a Pulmonary Valve-Sparing Strategy. Ann Thorac Surg 2005;80: [10] Fraser CD Jr, McKenzie ED, Cooley DA. Tetralogy of Fallot: surgical management individualized to the patient. Ann Thorac Surg 2001;71: [11] Rowlatt JF, Rimoldi HJ, Lev M. The quantitative anatomy of the normal child s heart. Pediatr Clin North Am 1963;10: [12] Van Arsdell GS, Maharaj GS, Tom J, Rao VK, Coles JG, Freedom RM et al. What is the optimal age for repair of tetralogy of Fallot? Circulation 2000; 102 (19 suppl 3):III [13] Ooi A, Moorjani N, Baliulis G, Keeton BR, Salmon AP, Monro JL et al. Medium term outcome for infant repair in tetralogy of Fallot: Indicators for timing of surgery. Eur J Cardiothorac Surg 2006;30: [14] Bové T, François K, Van De Kerckhove K, Panzer J, De Groote K, De Wolf D et al. Assessment of a right-ventricular infundibulum-sparing approach in transatrial-transpulmonary repair of tetralogy of Fallot. Eur J Cardiothorac Surg 2012;41: [15] Vobecky SJ, Williams WG, Trusler GA, Coles JG, Rebeyka IM, Smallhorn J et al. Survival analysis of infants under 18 months presenting with tetralogy of Fallot. Ann Thorac Surg 1993;56: [16] Alexiou C, Chen Q, Galogavrou M, Gnanapragasam J, Salmon AP, Keeton BR et al. Repair of tetralogy of Fallot in infancy with a transventricular or a transatrial approach. Eur J Cardiothorac Surg 2002;22: [17] Parry AJ, McElhinney DB, Kung GC, Reddy VM, Brook MM, Hanley FL. Elective primary repair of acyanotic tetralogy of Fallot in early infancy: overall outcome and impact on the pulmonary valve. J Am Coll Cardiol 2000;36: [18] Kanter KR, Kogon BE, Kirshbom PM, Carlock PR. Symptomatic Neonatal Tetralogy of Fallot: Repair or Shunt? Ann Thorac Surg 2010;89: [19] Mulder TJ, Pyles LA, Stolfi A, Pickoff AS, Moller JH. A multicenter analysis of the choice of initial surgical procedure in tetralogy of Fallot. Pediatr Cardiol 2002;23: [20] Knott-Craig CJ, Elkins RC, Lane MM, Holz J, McCue C, Ward KE. A 26-year experience with surgical management of tetralogy of Fallot: risk analysis for mortality or late reintervention. Ann Thorac Surg 1998;66: [21] Sfyridis PG, Kirvassilis GV, Papagiannis JK, Avramidis DP, Ieromonachos CGZavaropoulos PN et al. Preservation of right ventricular structure and function following transatrial-transpulmonary repair of tetralogy of Fallot. Eur J Cardiothorac Surg 2012;0:1 7. [22] Gladman G, McCrindle BW, Williams WG, Freedom RM, Benson LN. The modified Blalock-Taussig shunt: clinical impact and morbidity in Fallot s tetralogy in the current era. J Thorac Cardiovasc Surg 1997;114: [23] Dohlen G, Chaturvedi RR, Benson LN, Ozawa A, Van Arsdell GS, Fruitman DS et al. Stenting of the right ventricle outflow tract in the symptomatic infant with tetralogy of Fallot. Heart 2009;95: [24] Bacha EA, Scheule AM, Zurakowski D, Erickson LC, Hung J, Lang P et al. Long-term results after early primary repair of tetralogy of Fallot. J Thorac Cardiovasc Surg 2001;122: [25] Cobanoglu A, Schultz JM. Total correction of tetralogy of Fallot in the first year of life: late results. Ann Thorac Surg 2002;74:133 8.
8 B. Mimic et al. / European Journal of Cardio-Thoracic Surgery 99 APPENDIX. CONFERENCE DISCUSSION Dr G. Sarris (Athens, Greece): The authors report their entire last eight years experience with surgery for tetralogy of Fallot including all patients, palliations as well as repairs, and achieved truly excellent results with an early mortality of 0.4%, which is in keeping with the best results that are published in the literature. There were, however, some reoperations, including 11 early reoperations during the same hospitalization and a not-insignificant rate of late reoperations for the right ventricular outflow tract. Of the patients who were palliated, 35 had their palliation at the authors institution, and only eight were referred there with a previous palliative procedure. Therefore, of the 78 patients who comprised the early repair group, only 5% had a prior palliative procedure. But of the 173 patients who comprised the late group, 20% had shunts at the authors institution. One could therefore say that the group of patients who had later repair was burdened by these patients who were deemed to be unfavourable enough in terms of anatomy and clinical situation that they were shunted. And conversely, the group of patients who were subjected to early repair was relieved of the higher risk patients because those high-risk patients were shunted and therefore had their repair postponed. The authors question was is there a relationship between age at surgery and outcome? and they concluded that age at repair does not seem to affect clinical outcome. My comment is that this conclusion is not true for all of this group of patients, but only for the subgroup of patients who do not need early palliative help. Therefore, following this comment, I have two questions. The first one is: based on your analysis of this experience, have you changed your policy of selective shunting? And do you still offer, therefore, selective shunting to your patients based on these criteria that you have outlined? Dr Mimic: It is a quite difficult question. I think that we have changed our policy a little bit recently. So definitely there is still a group of patients who require some kind of palliation, actually different types of palliation. We offered in our institution, actually expressed our wish to offer the best possible palliation to the child regarding underlying anatomy, clinical condition, and weight at that time. Probably still there is still room for shunt palliation and also other types of palliation, but we think that the preferable age for complete repair is between four and six months of age. Dr Sarris: My follow-up question, then, pertains to the situation of a child with tetralogy, a newborn who might present to your unit without symptoms and without any of the listed indications for shunting. What would your recommendation for repair be for these patients? Would it be neonatal repair? Dr Mimic:Definitely we still do not do early neonatal repair in asymptomatic patients. Probably the best age, as I already mentioned, is between four and six months of age. But in symptomatic neonates with unfavourable anatomy, most likely we would offer a palliation. CONGENITAL
Surgical options for tetralogy of Fallot
 Surgical options for tetralogy of Fallot Serban Stoica FRCS(CTh) MD ACHD study day, 19 September 2017 Anatomy Physiology Children Adults Complications Follow up Anatomy Etienne Fallot (1850-1911) VSD Overriding
Surgical options for tetralogy of Fallot Serban Stoica FRCS(CTh) MD ACHD study day, 19 September 2017 Anatomy Physiology Children Adults Complications Follow up Anatomy Etienne Fallot (1850-1911) VSD Overriding
Surgical Treatment for Double Outlet Right Ventricle. Masakazu Nakao Consultant, Paediatric Cardiothoracic Surgery
 for Double Outlet Right Ventricle Masakazu Nakao Consultant, Paediatric Cardiothoracic Surgery 1 History Intraventricular tunnel (Kawashima) First repair of Taussig-Bing anomaly (Kirklin) Taussig-Bing
for Double Outlet Right Ventricle Masakazu Nakao Consultant, Paediatric Cardiothoracic Surgery 1 History Intraventricular tunnel (Kawashima) First repair of Taussig-Bing anomaly (Kirklin) Taussig-Bing
Surgical Management of TOF in Adults. Dr Flora Tsang Associate Consultant Department of Cardiothoracic Surgery Queen Mary Hospital
 Surgical Management of TOF in Adults Dr Flora Tsang Associate Consultant Department of Cardiothoracic Surgery Queen Mary Hospital Tetralogy of Fallot (TOF) in Adults Most common cyanotic congenital heart
Surgical Management of TOF in Adults Dr Flora Tsang Associate Consultant Department of Cardiothoracic Surgery Queen Mary Hospital Tetralogy of Fallot (TOF) in Adults Most common cyanotic congenital heart
MRI (AND CT) FOR REPAIRED TETRALOGY OF FALLOT
 MRI (AND CT) FOR REPAIRED TETRALOGY OF FALLOT Linda B Haramati MD, MS Departments of Radiology and Medicine Bronx, New York OUTLINE Pathogenesis Variants Initial surgical treatments Basic MR protocols
MRI (AND CT) FOR REPAIRED TETRALOGY OF FALLOT Linda B Haramati MD, MS Departments of Radiology and Medicine Bronx, New York OUTLINE Pathogenesis Variants Initial surgical treatments Basic MR protocols
Introduction. Study Design. Background. Operative Procedure-I
 Risk Factors for Mortality After the Norwood Procedure Using Right Ventricle to Pulmonary Artery Shunt Ann Thorac Surg 2009;87:178 86 86 Addressor: R1 胡祐寧 2009/3/4 AM7:30 SICU 討論室 Introduction Hypoplastic
Risk Factors for Mortality After the Norwood Procedure Using Right Ventricle to Pulmonary Artery Shunt Ann Thorac Surg 2009;87:178 86 86 Addressor: R1 胡祐寧 2009/3/4 AM7:30 SICU 討論室 Introduction Hypoplastic
Departments of Paediatrics and Cardiology, Mater Dei Hospital, Malta
 IMAGES in PAEDIATRIC CARDIOLOGY Bugeja J, Grech V, DeGiovanni JV. Right ventricular outflow tract stenting effective palliation for Fallot s tetralogy. Departments of Paediatrics and Cardiology, Mater
IMAGES in PAEDIATRIC CARDIOLOGY Bugeja J, Grech V, DeGiovanni JV. Right ventricular outflow tract stenting effective palliation for Fallot s tetralogy. Departments of Paediatrics and Cardiology, Mater
Deok Young Choi, Gil Hospital, Gachon University NEONATES WITH EBSTEIN S ANOMALY: PROBLEMS AND SOLUTION
 Deok Young Choi, Gil Hospital, Gachon University NEONATES WITH EBSTEIN S ANOMALY: PROBLEMS AND SOLUTION Carpentier classification Chauvaud S, Carpentier A. Multimedia Manual of Cardiothoracic Surgery 2007
Deok Young Choi, Gil Hospital, Gachon University NEONATES WITH EBSTEIN S ANOMALY: PROBLEMS AND SOLUTION Carpentier classification Chauvaud S, Carpentier A. Multimedia Manual of Cardiothoracic Surgery 2007
Perimembranous VSD: When Do We Ask For A Surgical Closure? LI Xin. Department of Cardiothoracic Surgery Queen Mary Hospital Hong Kong
 Perimembranous VSD: When Do We Ask For A Surgical Closure? LI Xin Department of Cardiothoracic Surgery Queen Mary Hospital Hong Kong Classification (by Kirklin) I. Subarterial (10%) Outlet, conal, supracristal,
Perimembranous VSD: When Do We Ask For A Surgical Closure? LI Xin Department of Cardiothoracic Surgery Queen Mary Hospital Hong Kong Classification (by Kirklin) I. Subarterial (10%) Outlet, conal, supracristal,
In 1980, Bex and associates 1 first introduced the initial
 Technique of Aortic Translocation for the Management of Transposition of the Great Arteries with a Ventricular Septal Defect and Pulmonary Stenosis Victor O. Morell, MD, and Peter D. Wearden, MD, PhD In
Technique of Aortic Translocation for the Management of Transposition of the Great Arteries with a Ventricular Septal Defect and Pulmonary Stenosis Victor O. Morell, MD, and Peter D. Wearden, MD, PhD In
Appendix A.1: Tier 1 Surgical Procedure Terms and Definitions
 Appendix A.1: Tier 1 Surgical Procedure Terms and Definitions Tier 1 surgeries AV Canal Atrioventricular Septal Repair, Complete Repair of complete AV canal (AVSD) using one- or two-patch or other technique,
Appendix A.1: Tier 1 Surgical Procedure Terms and Definitions Tier 1 surgeries AV Canal Atrioventricular Septal Repair, Complete Repair of complete AV canal (AVSD) using one- or two-patch or other technique,
Journal of the American College of Cardiology Vol. 36, No. 7, by the American College of Cardiology ISSN /00/$20.
 Journal of the American College of Cardiology Vol. 36, No. 7, 2000 2000 by the American College of Cardiology ISSN 0735-1097/00/$20.00 Published by Elsevier Science Inc. PII S0735-1097(00)00989-X Elective
Journal of the American College of Cardiology Vol. 36, No. 7, 2000 2000 by the American College of Cardiology ISSN 0735-1097/00/$20.00 Published by Elsevier Science Inc. PII S0735-1097(00)00989-X Elective
Cover Page. The handle holds various files of this Leiden University dissertation.
 Cover Page The handle http://hdl.handle.net/1887/19123 holds various files of this Leiden University dissertation. Author: Hoohenkerk, Gerard Joannes Franciscus Title: Surgical correction of atrioventricular
Cover Page The handle http://hdl.handle.net/1887/19123 holds various files of this Leiden University dissertation. Author: Hoohenkerk, Gerard Joannes Franciscus Title: Surgical correction of atrioventricular
Inter-surgeon variability in long-term outcomes after transatrial repair of tetralogy of Fallot: 25 years experience with 675 patients.
 Inter-surgeon variability in long-term outcomes after transatrial repair of tetralogy of Fallot: 25 years experience with 675 patients. Y d'udekem, JC Galati, IE Konstantinov, MMH Cheung, CP Brizard Royal
Inter-surgeon variability in long-term outcomes after transatrial repair of tetralogy of Fallot: 25 years experience with 675 patients. Y d'udekem, JC Galati, IE Konstantinov, MMH Cheung, CP Brizard Royal
Tetralogy of Fallot (TOF) with atrioventricular (AV)
 Tetralogy of Fallot with Atrioventricular Canal Defect: Two Patch Repair Sitaram M. Emani, MD, and Pedro J. del Nido, MD Tetralogy of Fallot (TOF) with atrioventricular (AV) canal defect is classified
Tetralogy of Fallot with Atrioventricular Canal Defect: Two Patch Repair Sitaram M. Emani, MD, and Pedro J. del Nido, MD Tetralogy of Fallot (TOF) with atrioventricular (AV) canal defect is classified
Tetralogy of Fallot (TOF) with absent pulmonary valve
 Repair of Tetralogy of Fallot with Absent Pulmonary Valve Syndrome Karl F. Welke, MD, and Ross M. Ungerleider, MD, MBA Tetralogy of Fallot (TOF) with absent pulmonary valve syndrome (APVS) occurs in 5%
Repair of Tetralogy of Fallot with Absent Pulmonary Valve Syndrome Karl F. Welke, MD, and Ross M. Ungerleider, MD, MBA Tetralogy of Fallot (TOF) with absent pulmonary valve syndrome (APVS) occurs in 5%
Since first successfully performed by Jatene et al, the
 Long-Term Predictors of Aortic Root Dilation and Aortic Regurgitation After Arterial Switch Operation Marcy L. Schwartz, MD; Kimberlee Gauvreau, ScD; Pedro del Nido, MD; John E. Mayer, MD; Steven D. Colan,
Long-Term Predictors of Aortic Root Dilation and Aortic Regurgitation After Arterial Switch Operation Marcy L. Schwartz, MD; Kimberlee Gauvreau, ScD; Pedro del Nido, MD; John E. Mayer, MD; Steven D. Colan,
SURGICAL TREATMENT AND OUTCOME OF CONGENITAL HEART DISEASE
 SURGICAL TREATMENT AND OUTCOME OF CONGENITAL HEART DISEASE Mr. W. Brawn Birmingham Children s Hospital. Aims of surgery The aim of surgery in congenital heart disease is to correct or palliate the heart
SURGICAL TREATMENT AND OUTCOME OF CONGENITAL HEART DISEASE Mr. W. Brawn Birmingham Children s Hospital. Aims of surgery The aim of surgery in congenital heart disease is to correct or palliate the heart
The modified Konno procedure, or subaortic ventriculoplasty,
 Modified Konno Procedure for Left Ventricular Outflow Tract Obstruction David P. Bichell, MD The modified Konno procedure, or subaortic ventriculoplasty, first described by Cooley and Garrett in1986, 1
Modified Konno Procedure for Left Ventricular Outflow Tract Obstruction David P. Bichell, MD The modified Konno procedure, or subaortic ventriculoplasty, first described by Cooley and Garrett in1986, 1
Surgical Repair of Ventricular Septal Defect; Contemporary Results and Risk Factors for a Complicated Course
 Pediatr Cardiol (2017) 38:264 270 DOI 10.1007/s00246-016-1508-2 ORIGINAL ARTICLE Surgical Repair of Ventricular Septal Defect; Contemporary Results and Risk Factors for a Complicated Course Maartje Schipper
Pediatr Cardiol (2017) 38:264 270 DOI 10.1007/s00246-016-1508-2 ORIGINAL ARTICLE Surgical Repair of Ventricular Septal Defect; Contemporary Results and Risk Factors for a Complicated Course Maartje Schipper
Congenital Heart Defects
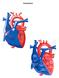 Normal Heart Congenital Heart Defects 1. Patent Ductus Arteriosus The ductus arteriosus connects the main pulmonary artery to the aorta. In utero, it allows the blood leaving the right ventricle to bypass
Normal Heart Congenital Heart Defects 1. Patent Ductus Arteriosus The ductus arteriosus connects the main pulmonary artery to the aorta. In utero, it allows the blood leaving the right ventricle to bypass
Early and Late Results of Total Correction of Tetralogy of Fallot
 ORIGINAL REPORT Early and Late Results of Total Correction of Tetralogy of Fallot Khosrow Hashemzadeh 1 and Shahryar Hashemzadeh 2 1 Department of Cardiovascular Surgery, Cardiovascular Research Center,
ORIGINAL REPORT Early and Late Results of Total Correction of Tetralogy of Fallot Khosrow Hashemzadeh 1 and Shahryar Hashemzadeh 2 1 Department of Cardiovascular Surgery, Cardiovascular Research Center,
Cardiac Catheterization Cases Primary Cardiac Diagnoses Facility 12 month period from to PRIMARY DIAGNOSES (one per patient)
 PRIMARY DIAGNOSES (one per patient) Septal Defects ASD (Atrial Septal Defect) PFO (Patent Foramen Ovale) ASD, Secundum ASD, Sinus venosus ASD, Coronary sinus ASD, Common atrium (single atrium) VSD (Ventricular
PRIMARY DIAGNOSES (one per patient) Septal Defects ASD (Atrial Septal Defect) PFO (Patent Foramen Ovale) ASD, Secundum ASD, Sinus venosus ASD, Coronary sinus ASD, Common atrium (single atrium) VSD (Ventricular
Perioperative Management of DORV Case
 Perioperative Management of DORV Case James P. Spaeth, MD Department of Anesthesia Cincinnati Children s Hospital Medical Center University of Cincinnati Objectives: 1. Discuss considerations regarding
Perioperative Management of DORV Case James P. Spaeth, MD Department of Anesthesia Cincinnati Children s Hospital Medical Center University of Cincinnati Objectives: 1. Discuss considerations regarding
Outline. Congenital Heart Disease. Special Considerations for Special Populations: Congenital Heart Disease
 Special Considerations for Special Populations: Congenital Heart Disease Valerie Bosco, FNP, EdD Alison Knauth Meadows, MD, PhD University of California San Francisco Adult Congenital Heart Program Outline
Special Considerations for Special Populations: Congenital Heart Disease Valerie Bosco, FNP, EdD Alison Knauth Meadows, MD, PhD University of California San Francisco Adult Congenital Heart Program Outline
The first report of the Society of Thoracic Surgeons
 REPORT The Society of Thoracic Surgeons National Congenital Heart Surgery Database Report: Analysis of the First Harvest (1994 1997) Constantine Mavroudis, MD, Melanie Gevitz, BA, W. Steves Ring, MD, Charles
REPORT The Society of Thoracic Surgeons National Congenital Heart Surgery Database Report: Analysis of the First Harvest (1994 1997) Constantine Mavroudis, MD, Melanie Gevitz, BA, W. Steves Ring, MD, Charles
The need for right ventricular outflow tract reconstruction
 Polytetrafluoroethylene Bicuspid Pulmonary Valve Implantation James A. Quintessenza, MD The need for right ventricular outflow tract reconstruction and pulmonary valve replacement is increasing for many
Polytetrafluoroethylene Bicuspid Pulmonary Valve Implantation James A. Quintessenza, MD The need for right ventricular outflow tract reconstruction and pulmonary valve replacement is increasing for many
Determination of TOF Characteristics in a Tertiary Care Centre of Bangladesh
 46 BANGLADESH J CHILD HEALTH 2010; VOL 34 (3): 86-91 Determination of TOF Characteristics in a Tertiary Care Centre of Bangladesh JAHANGIR KABIR 1, REZOANA RIMA 2, SOOFIA KHATOON 3, REZAUL HASSAN 4, AMM
46 BANGLADESH J CHILD HEALTH 2010; VOL 34 (3): 86-91 Determination of TOF Characteristics in a Tertiary Care Centre of Bangladesh JAHANGIR KABIR 1, REZOANA RIMA 2, SOOFIA KHATOON 3, REZAUL HASSAN 4, AMM
Although most patients with Ebstein s anomaly live
 Management of Neonatal Ebstein s Anomaly Christopher J. Knott-Craig, MD, FACS Although most patients with Ebstein s anomaly live through infancy, those who present clinically as neonates are a distinct
Management of Neonatal Ebstein s Anomaly Christopher J. Knott-Craig, MD, FACS Although most patients with Ebstein s anomaly live through infancy, those who present clinically as neonates are a distinct
East and Central African Journal of Surgery Volume 12 Number 2 November /December 2007
 23 Modified Blalock-Taussig Shunt in Palliative Cardiac Surgery E.V. Ussiri 1, E.T.M. Nyawawa 1, U. Mpoki 2, E.R. Lugazia 2, G.C. Mannam 3, L.R. Sajja 4. S. Sompali 4 1 Specialist Surgeon, Cardiothoracic
23 Modified Blalock-Taussig Shunt in Palliative Cardiac Surgery E.V. Ussiri 1, E.T.M. Nyawawa 1, U. Mpoki 2, E.R. Lugazia 2, G.C. Mannam 3, L.R. Sajja 4. S. Sompali 4 1 Specialist Surgeon, Cardiothoracic
가천의대길병원소아심장과최덕영 PA C IVS THE EVALUATION AND PRINCIPLES OF TREATMENT STRATEGY
 가천의대길병원소아심장과최덕영 PA C IVS THE EVALUATION AND PRINCIPLES OF TREATMENT STRATEGY PA c IVS (not only pulmonary valve disease) Edwards JE. Pathologic Alteration of the right heart. In: Konstam MA, Isner M, eds.
가천의대길병원소아심장과최덕영 PA C IVS THE EVALUATION AND PRINCIPLES OF TREATMENT STRATEGY PA c IVS (not only pulmonary valve disease) Edwards JE. Pathologic Alteration of the right heart. In: Konstam MA, Isner M, eds.
The Rastelli procedure has been traditionally used for repair
 En-bloc Rotation of the Truncus Arteriosus A Technique for Complete Anatomic Repair of Transposition of the Great Arteries/Ventricular Septal Defect/Left Ventricular Outflow Tract Obstruction or Double
En-bloc Rotation of the Truncus Arteriosus A Technique for Complete Anatomic Repair of Transposition of the Great Arteries/Ventricular Septal Defect/Left Ventricular Outflow Tract Obstruction or Double
Neonatal Repair of Truncus Arteriosus: Continuing Improvement in Outcomes
 ORIGINAL ARTICLES: CARDIOVASCULAR Neonatal Repair of Truncus Arteriosus: Continuing Improvement in Outcomes LeNardo D. Thompson, MD, Doff B. McElhinney, MD, V. Mohan Reddy, MD, Ed Petrossian, MD, Norman
ORIGINAL ARTICLES: CARDIOVASCULAR Neonatal Repair of Truncus Arteriosus: Continuing Improvement in Outcomes LeNardo D. Thompson, MD, Doff B. McElhinney, MD, V. Mohan Reddy, MD, Ed Petrossian, MD, Norman
3/14/2011 MANAGEMENT OF NEWBORNS CARDIAC INTENSIVE CARE CONFERENCE FOR HEALTH PROFESSIONALS IRVINE, CA. MARCH 7, 2011 WITH HEART DEFECTS
 CONFERENCE FOR HEALTH PROFESSIONALS IRVINE, CA. MARCH 7, 2011 MANAGEMENT OF NEWBORNS WITH HEART DEFECTS A NTHONY C. CHANG, MD, MBA, MPH M E D I C AL D I RE C T OR, HEART I N S T I T U T E C H I LDRE N
CONFERENCE FOR HEALTH PROFESSIONALS IRVINE, CA. MARCH 7, 2011 MANAGEMENT OF NEWBORNS WITH HEART DEFECTS A NTHONY C. CHANG, MD, MBA, MPH M E D I C AL D I RE C T OR, HEART I N S T I T U T E C H I LDRE N
Indications for the Brock operation in current
 Thorax (1973), 28, 1. Indications for the Brock operation in current treatment of tetralogy of Fallot H. R. MATTHEWS and R. H. R. BELSEY Department of Thoracic Surgery, Frenchay Hospital, Bristol It is
Thorax (1973), 28, 1. Indications for the Brock operation in current treatment of tetralogy of Fallot H. R. MATTHEWS and R. H. R. BELSEY Department of Thoracic Surgery, Frenchay Hospital, Bristol It is
Pulmonary Valve Replacement
 Pulmonary Valve Replacement Christian Kreutzer MD Hospital Nacional Alejandro Posadas Hospital Universitario Austral (No disclosures) Scope of the problem. CHD with PS or PA require a RVOT procedure. Tetralogy
Pulmonary Valve Replacement Christian Kreutzer MD Hospital Nacional Alejandro Posadas Hospital Universitario Austral (No disclosures) Scope of the problem. CHD with PS or PA require a RVOT procedure. Tetralogy
LEFT VENTRICULAR OUTFLOW OBSTRUCTION WITH A VSD: OPTIONS FOR SURGICAL MANAGEMENT
 LEFT VENTRICULAR OUTFLOW OBSTRUCTION WITH A VSD: OPTIONS FOR SURGICAL MANAGEMENT 10-13 March 2017 Ritz Carlton, Riyadh, Saudi Arabia Zohair AlHalees, MD Consultant, Cardiac Surgery Heart Centre LEFT VENTRICULAR
LEFT VENTRICULAR OUTFLOW OBSTRUCTION WITH A VSD: OPTIONS FOR SURGICAL MANAGEMENT 10-13 March 2017 Ritz Carlton, Riyadh, Saudi Arabia Zohair AlHalees, MD Consultant, Cardiac Surgery Heart Centre LEFT VENTRICULAR
T who has survived first-stage palliative surgical management
 Intermediate Procedures After First-Stage Norwood Operation Facilitate Subsequent Repair Richard A. Jonas, MD Department of Cardiac Surgery, Children s Hospital, Boston, Massachusetts Actuarial analysis
Intermediate Procedures After First-Stage Norwood Operation Facilitate Subsequent Repair Richard A. Jonas, MD Department of Cardiac Surgery, Children s Hospital, Boston, Massachusetts Actuarial analysis
Recent technical advances and increasing experience
 Pediatric Open Heart Operations Without Diagnostic Cardiac Catheterization Jean-Pierre Pfammatter, MD, Pascal A. Berdat, MD, Thierry P. Carrel, MD, and Franco P. Stocker, MD Division of Pediatric Cardiology,
Pediatric Open Heart Operations Without Diagnostic Cardiac Catheterization Jean-Pierre Pfammatter, MD, Pascal A. Berdat, MD, Thierry P. Carrel, MD, and Franco P. Stocker, MD Division of Pediatric Cardiology,
Repair of tetralogy of Fallot in infancy with a transventricular or a transatrial approach q
 European Journal of Cardio-thoracic Surgery 22 (2002) 174 183 www.elsevier.com/locate/ejcts Repair of tetralogy of Fallot in infancy with a transventricular or a transatrial approach q Christos Alexiou
European Journal of Cardio-thoracic Surgery 22 (2002) 174 183 www.elsevier.com/locate/ejcts Repair of tetralogy of Fallot in infancy with a transventricular or a transatrial approach q Christos Alexiou
Surgery for Congenital Heart Disease
 The influence of perioperative factors on outcomes in children aged less than 18 months after repair of tetralogy of Fallot Elisabeth I. van Dongen, MD a Angelique G. Glansdorp, MD a Reinout J. Mildner,
The influence of perioperative factors on outcomes in children aged less than 18 months after repair of tetralogy of Fallot Elisabeth I. van Dongen, MD a Angelique G. Glansdorp, MD a Reinout J. Mildner,
Tetralogy of Fallot (TOF) with absent pulmonary valve syndrome (APV) is
 Chen et al Surgery for Congenital Heart Disease Superior outcomes for repair in infants and neonates with tetralogy of Fallot with absent pulmonary valve syndrome Jonathan M. Chen, MD, a Julie S. Glickstein,
Chen et al Surgery for Congenital Heart Disease Superior outcomes for repair in infants and neonates with tetralogy of Fallot with absent pulmonary valve syndrome Jonathan M. Chen, MD, a Julie S. Glickstein,
The pulmonary valve is the most common heart valve
 Biologic versus Mechanical Valve Replacement of the Pulmonary Valve After Multiple Reconstructions of the RVOT Tract S. Adil Husain, MD, and John Brown, MD Indiana University School of Medicine, Department
Biologic versus Mechanical Valve Replacement of the Pulmonary Valve After Multiple Reconstructions of the RVOT Tract S. Adil Husain, MD, and John Brown, MD Indiana University School of Medicine, Department
Absent Pulmonary Valve Syndrome
 Absent Pulmonary Valve Syndrome Fact sheet on Absent Pulmonary Valve Syndrome In this condition, which has some similarities to Fallot's Tetralogy, there is a VSD with narrowing at the pulmonary valve.
Absent Pulmonary Valve Syndrome Fact sheet on Absent Pulmonary Valve Syndrome In this condition, which has some similarities to Fallot's Tetralogy, there is a VSD with narrowing at the pulmonary valve.
Adult Congenital Heart Disease T S U N ` A M I!
 Adult Congenital Heart Disease T S U N ` A M I! Erwin Oechslin, MD, FRCPC, FESC Director, Congenital Cardiac Centre for Adults University Health Network Peter Munk Cardiac Centre / Toronto General Hospital
Adult Congenital Heart Disease T S U N ` A M I! Erwin Oechslin, MD, FRCPC, FESC Director, Congenital Cardiac Centre for Adults University Health Network Peter Munk Cardiac Centre / Toronto General Hospital
Hemodynamic assessment after palliative surgery
 THERAPY AND PREVENTION CONGENITAL HEART DISEASE Hemodynamic assessment after palliative surgery for hypoplastic left heart syndrome PETER LANG, M.D., AND WILLIAM I. NORWOOD, M.D., PH.D. ABSTRACT Ten patients
THERAPY AND PREVENTION CONGENITAL HEART DISEASE Hemodynamic assessment after palliative surgery for hypoplastic left heart syndrome PETER LANG, M.D., AND WILLIAM I. NORWOOD, M.D., PH.D. ABSTRACT Ten patients
Since 1954, when Lillehei completed the first successful
 Current Surgical Management of Tetralogy of Fallot Vaughn A. Starnes, MD, Giovanni Battista Luciani, MD, David A. Latter, MD, and Michael 1. Griffin, MD Division of Cardiothoracic Surgery, University of
Current Surgical Management of Tetralogy of Fallot Vaughn A. Starnes, MD, Giovanni Battista Luciani, MD, David A. Latter, MD, and Michael 1. Griffin, MD Division of Cardiothoracic Surgery, University of
Repair or Replacement
 Surgical intervention post MitraClip Device: Repair or Replacement Saudi Heart Association, February 21-24 Rüdiger Lange, MD, PhD Nicolo Piazza, MD, FRCPC, FESC German Heart Center, Munich, Germany Division
Surgical intervention post MitraClip Device: Repair or Replacement Saudi Heart Association, February 21-24 Rüdiger Lange, MD, PhD Nicolo Piazza, MD, FRCPC, FESC German Heart Center, Munich, Germany Division
Repair of Complete Atrioventricular Septal Defects Single Patch Technique
 Repair of Complete Atrioventricular Septal Defects Single Patch Technique Fred A. Crawford, Jr., MD The first repair of a complete atrioventricular septal defect was performed in 1954 by Lillehei using
Repair of Complete Atrioventricular Septal Defects Single Patch Technique Fred A. Crawford, Jr., MD The first repair of a complete atrioventricular septal defect was performed in 1954 by Lillehei using
Children with Single Ventricle Physiology: The Possibilities
 Children with Single Ventricle Physiology: The Possibilities William I. Douglas, M.D. Pediatric Cardiovascular Surgery Children s Memorial Hermann Hospital The University of Texas Health Science Center
Children with Single Ventricle Physiology: The Possibilities William I. Douglas, M.D. Pediatric Cardiovascular Surgery Children s Memorial Hermann Hospital The University of Texas Health Science Center
An audit of surgical repair of Tetralogy of Fallot in an African tertiary care centre
 SURGICAL REPAIR OF TETRALOGY OF FALLOT An audit of surgical repair of Tetralogy of Fallot in an African tertiary care centre Deliwe P. Ngwezi *, Katharina Vanderdonck #, Solomon E. Levin # and Antoinette
SURGICAL REPAIR OF TETRALOGY OF FALLOT An audit of surgical repair of Tetralogy of Fallot in an African tertiary care centre Deliwe P. Ngwezi *, Katharina Vanderdonck #, Solomon E. Levin # and Antoinette
Candice Silversides, MD Toronto Congenital Cardiac Centre for Adults University of Toronto Toronto, Canada
 PVR Following Repair of TOF Now? When? Candice Silversides, MD Toronto Congenital Cardiac Centre for Adults University of Toronto Toronto, Canada Late Complications after TOF repair Repair will be necessary
PVR Following Repair of TOF Now? When? Candice Silversides, MD Toronto Congenital Cardiac Centre for Adults University of Toronto Toronto, Canada Late Complications after TOF repair Repair will be necessary
The Double Switch Using Bidirectional Glenn and Hemi-Mustard. Frank Hanley
 The Double Switch Using Bidirectional Glenn and Hemi-Mustard Frank Hanley No relationships to disclose CCTGA Interesting Points for Discussion What to do when. associated defects must be addressed surgically:
The Double Switch Using Bidirectional Glenn and Hemi-Mustard Frank Hanley No relationships to disclose CCTGA Interesting Points for Discussion What to do when. associated defects must be addressed surgically:
Pediatric Echocardiography Examination Content Outline
 Pediatric Echocardiography Examination Content Outline (Outline Summary) # Domain Subdomain Percentage 1 Anatomy and Physiology Normal Anatomy and Physiology 10% 2 Abnormal Pathology and Pathophysiology
Pediatric Echocardiography Examination Content Outline (Outline Summary) # Domain Subdomain Percentage 1 Anatomy and Physiology Normal Anatomy and Physiology 10% 2 Abnormal Pathology and Pathophysiology
Notes by Sandra Dankwa 2009 HF- Heart Failure DS- Down Syndrome IE- Infective Endocarditis ET- Exercise Tolerance. Small VSD Symptoms -asymptomatic
 Congenital Heart Disease: Notes. Condition Pathology PC Ix Rx Ventricular septal defect (VSD) L R shuntsdefect anywhere in the ventricle, usually perimembranous (next to the tricuspid valve) 30% 1)small
Congenital Heart Disease: Notes. Condition Pathology PC Ix Rx Ventricular septal defect (VSD) L R shuntsdefect anywhere in the ventricle, usually perimembranous (next to the tricuspid valve) 30% 1)small
Intraoperative Stents to Rehabilitate Severely Stenotic Pulmonary Vessels
 Intraoperative Stents to Rehabilitate Severely Stenotic Pulmonary Vessels Ross M. Ungerleider, MD, Troy A. Johnston, MD, Martin P. O Laughlin, MD, James J. Jaggers, MD, and Peter R. Gaskin, MD Division
Intraoperative Stents to Rehabilitate Severely Stenotic Pulmonary Vessels Ross M. Ungerleider, MD, Troy A. Johnston, MD, Martin P. O Laughlin, MD, James J. Jaggers, MD, and Peter R. Gaskin, MD Division
Surgery For Ebstein Anomaly
 Surgery For Ebstein Anomaly Christian Pizarro, MD Chief, Pediatric Cardiothoracic Surgery Director, Nemours Cardiac Center Alfred I. dupont Hospital for Children Professor of Surgery and Pediatrics Sidney
Surgery For Ebstein Anomaly Christian Pizarro, MD Chief, Pediatric Cardiothoracic Surgery Director, Nemours Cardiac Center Alfred I. dupont Hospital for Children Professor of Surgery and Pediatrics Sidney
Tetralogy of Fallot Latest data in risk stratification and replacement of pulmonic valve
 Tetralogy of Fallot Latest data in risk stratification and replacement of pulmonic valve Alexandra A Frogoudaki Adult Congenital Heart Clinic Second Cardiology Department ATTIKON University Hospital No
Tetralogy of Fallot Latest data in risk stratification and replacement of pulmonic valve Alexandra A Frogoudaki Adult Congenital Heart Clinic Second Cardiology Department ATTIKON University Hospital No
ECHOCARDIOGRAPHIC APPROACH TO CONGENITAL HEART DISEASE: THE UNOPERATED ADULT
 ECHOCARDIOGRAPHIC APPROACH TO CONGENITAL HEART DISEASE: THE UNOPERATED ADULT Karen Stout, MD, FACC Divisions of Cardiology University of Washington Medical Center Seattle Children s Hospital NO DISCLOSURES
ECHOCARDIOGRAPHIC APPROACH TO CONGENITAL HEART DISEASE: THE UNOPERATED ADULT Karen Stout, MD, FACC Divisions of Cardiology University of Washington Medical Center Seattle Children s Hospital NO DISCLOSURES
describes 28 years of experience in the surgical management of tetralogy of Fallot with subarterial VSD at Tenri Hospital in Japan.
 EARLY AND LATE RESULTS OF REPAIR OF TETRALOGY OF FALLOT WITH SUBARTERIAL VENTRICULAR SEPTAL DEFECT A comparative evaluation of tetralogy with perimembranous ventricular septal defect Between November 1966
EARLY AND LATE RESULTS OF REPAIR OF TETRALOGY OF FALLOT WITH SUBARTERIAL VENTRICULAR SEPTAL DEFECT A comparative evaluation of tetralogy with perimembranous ventricular septal defect Between November 1966
Congenital heart disease: When to act and what to do?
 Leading Article Congenital heart disease: When to act and what to do? Duminda Samarasinghe 1 Sri Lanka Journal of Child Health, 2010; 39: 39-43 (Key words: Congenital heart disease) Congenital heart disease
Leading Article Congenital heart disease: When to act and what to do? Duminda Samarasinghe 1 Sri Lanka Journal of Child Health, 2010; 39: 39-43 (Key words: Congenital heart disease) Congenital heart disease
The application of autologous pulmonary artery in surgical correction of complicated aortic arch anomaly
 Original Article The application of autologous pulmonary artery in surgical correction of complicated aortic arch anomaly Shusheng Wen, Jianzheng Cen, Jimei Chen, Gang Xu, Biaochuan He, Yun Teng, Jian
Original Article The application of autologous pulmonary artery in surgical correction of complicated aortic arch anomaly Shusheng Wen, Jianzheng Cen, Jimei Chen, Gang Xu, Biaochuan He, Yun Teng, Jian
5.8 Congenital Heart Disease
 5.8 Congenital Heart Disease Congenital heart diseases (CHD) refer to structural or functional heart diseases, which are present at birth. Some of these lesions may be discovered later. prevalence of Chd
5.8 Congenital Heart Disease Congenital heart diseases (CHD) refer to structural or functional heart diseases, which are present at birth. Some of these lesions may be discovered later. prevalence of Chd
Surgical Treatment for Atrioventricular Septal Defect. Masakazu Nakao Consultant, Paediatric Cardiothoracic Surgery
 Surgical Treatment for Atrioventricular Septal Defect Masakazu Nakao Consultant, Paediatric Cardiothoracic Surgery 1 History Rastelli classification (Rastelli) Pulmonary artery banding (Muller & Dammann)
Surgical Treatment for Atrioventricular Septal Defect Masakazu Nakao Consultant, Paediatric Cardiothoracic Surgery 1 History Rastelli classification (Rastelli) Pulmonary artery banding (Muller & Dammann)
Congenital Heart Disease II: The Repaired Adult
 Congenital Heart Disease II: The Repaired Adult Doreen DeFaria Yeh, MD FACC Assistant Professor, Harvard Medical School MGH Adult Congenital Heart Disease Program Echocardiography Section, no disclosures
Congenital Heart Disease II: The Repaired Adult Doreen DeFaria Yeh, MD FACC Assistant Professor, Harvard Medical School MGH Adult Congenital Heart Disease Program Echocardiography Section, no disclosures
Long-term results (22 years) of the Ross Operation a single institutional experience
 Long-term results (22 years) of the Ross Operation a single institutional experience Authors: Costa FDA, Schnorr GM, Veloso M,Calixto A, Colatusso D, Balbi EM, Torres R, Ferreira ADA, Colatusso C Department
Long-term results (22 years) of the Ross Operation a single institutional experience Authors: Costa FDA, Schnorr GM, Veloso M,Calixto A, Colatusso D, Balbi EM, Torres R, Ferreira ADA, Colatusso C Department
Outcome after repair of atrioventricular septal defect with tetralogy of Fallot
 CONGENITAL HEART DISEASE Outcome after repair of atrioventricular septal defect with tetralogy of Fallot Jeffrey H. Shuhaiber, MD, a Barbara Robinson, MD, a Kimberlee Gauvreau, ScD, b Roger Breitbart,
CONGENITAL HEART DISEASE Outcome after repair of atrioventricular septal defect with tetralogy of Fallot Jeffrey H. Shuhaiber, MD, a Barbara Robinson, MD, a Kimberlee Gauvreau, ScD, b Roger Breitbart,
Accepted Manuscript. The Left atrioventricular valve: The Achilles Heel of incomplete endocardial cushion defects. Meena Nathan, MD, MPH
 Accepted Manuscript The Left atrioventricular valve: The Achilles Heel of incomplete endocardial cushion defects Meena Nathan, MD, MPH PII: S0022-5223(18)32898-8 DOI: https://doi.org/10.1016/j.jtcvs.2018.10.120
Accepted Manuscript The Left atrioventricular valve: The Achilles Heel of incomplete endocardial cushion defects Meena Nathan, MD, MPH PII: S0022-5223(18)32898-8 DOI: https://doi.org/10.1016/j.jtcvs.2018.10.120
DIAGNOSIS, MANAGEMENT AND OUTCOME OF HEART DISEASE IN SUDANESE PATIENTS
 434 E AST AFRICAN MEDICAL JOURNAL September 2007 East African Medical Journal Vol. 84 No. 9 September 2007 DIAGNOSIS, MANAGEMENT AND OUTCOME OF CONGENITAL HEART DISEASE IN SUDANESE PATIENTS K.M.A. Sulafa,
434 E AST AFRICAN MEDICAL JOURNAL September 2007 East African Medical Journal Vol. 84 No. 9 September 2007 DIAGNOSIS, MANAGEMENT AND OUTCOME OF CONGENITAL HEART DISEASE IN SUDANESE PATIENTS K.M.A. Sulafa,
Congenital Heart Disease An Approach for Simple and Complex Anomalies
 Congenital Heart Disease An Approach for Simple and Complex Anomalies Michael D. Pettersen, MD Director, Echocardiography Rocky Mountain Hospital for Children Denver, CO None Disclosures 1 ASCeXAM Contains
Congenital Heart Disease An Approach for Simple and Complex Anomalies Michael D. Pettersen, MD Director, Echocardiography Rocky Mountain Hospital for Children Denver, CO None Disclosures 1 ASCeXAM Contains
Results of reparative surgery for tetralogy of Fallot: data from the European Association for Cardio-Thoracic Surgery Congenital Database
 European Journal of Cardio-Thoracic Surgery 42 (2012) 766 774 doi:10.1093/ejcts/ezs478 ORIGINAL ARTICLE Results of reparative surgery for tetralogy of Fallot: data from the European Association for Cardio-Thoracic
European Journal of Cardio-Thoracic Surgery 42 (2012) 766 774 doi:10.1093/ejcts/ezs478 ORIGINAL ARTICLE Results of reparative surgery for tetralogy of Fallot: data from the European Association for Cardio-Thoracic
An anterior aortoventriculoplasty, known as the Konno-
 The Konno-Rastan Procedure for Anterior Aortic Annular Enlargement Mark E. Roeser, MD An anterior aortoventriculoplasty, known as the Konno-Rastan procedure, is a useful tool for the cardiac surgeon. Originally,
The Konno-Rastan Procedure for Anterior Aortic Annular Enlargement Mark E. Roeser, MD An anterior aortoventriculoplasty, known as the Konno-Rastan procedure, is a useful tool for the cardiac surgeon. Originally,
PULMONARY ARTERY STENTING AFfER TOTAL SURGICAL CORRECTION OF RIGHT VENTRICULAR OUTFLOW TRACT OBSTRUCTIVE LESIONS
 Articles 5 PULMONARY ARTERY STENTING AFfER TOTAL SURGICAL CORRECTION OF RIGHT VENTRICULAR OUTFLOW TRACT OBSTRUCTIVE LESIONS SAMEH ARAB, MD; ERIC ROSENTHAL, MD, MRCP; SHAKEEL QURESHI, MB, MRCP; MICHAEL
Articles 5 PULMONARY ARTERY STENTING AFfER TOTAL SURGICAL CORRECTION OF RIGHT VENTRICULAR OUTFLOW TRACT OBSTRUCTIVE LESIONS SAMEH ARAB, MD; ERIC ROSENTHAL, MD, MRCP; SHAKEEL QURESHI, MB, MRCP; MICHAEL
Research Presentation June 23, Nimish Muni Resident Internal Medicine
 Research Presentation June 23, 2009 Nimish Muni Resident Internal Medicine Research Question In adult patients with repaired Tetralogy of Fallot, how does Echocardiography compare to MRI in evaluating
Research Presentation June 23, 2009 Nimish Muni Resident Internal Medicine Research Question In adult patients with repaired Tetralogy of Fallot, how does Echocardiography compare to MRI in evaluating
The background of the Cardiac Sonographer Network News masthead is a diagnostic image:
 Number 5 Welcome Number 5 Welcome to the newsletter created just for you: sonographers who perform pediatric echocardiograms in primarily adult echo labs. Each issue features tips on echocardiography of
Number 5 Welcome Number 5 Welcome to the newsletter created just for you: sonographers who perform pediatric echocardiograms in primarily adult echo labs. Each issue features tips on echocardiography of
Adults with Congenital Heart Disease. Michael E. McConnell MD, Wendy Book MD Teresa Lyle RN NNP
 Adults with Congenital Heart Disease Michael E. McConnell MD, Wendy Book MD Teresa Lyle RN NNP Outline History of CHD Statistics Specific lesions (TOF, TGA, Single ventricle) Erythrocytosis Pregnancy History
Adults with Congenital Heart Disease Michael E. McConnell MD, Wendy Book MD Teresa Lyle RN NNP Outline History of CHD Statistics Specific lesions (TOF, TGA, Single ventricle) Erythrocytosis Pregnancy History
Mid-term Result of One and One Half Ventricular Repair in a Patient with Pulmonary Atresia and Intact Ventricular Septum
 Mid-term Result of One and One Half Ventricular Repair in a Patient with Pulmonary Atresia and Intact Ventricular Septum Kagami MIYAJI, MD, Akira FURUSE, MD, Toshiya OHTSUKA, MD, and Motoaki KAWAUCHI,
Mid-term Result of One and One Half Ventricular Repair in a Patient with Pulmonary Atresia and Intact Ventricular Septum Kagami MIYAJI, MD, Akira FURUSE, MD, Toshiya OHTSUKA, MD, and Motoaki KAWAUCHI,
Debate: Should Ductal Stent Implantation be Considered for All Newborn Infants with Reduced Pulmonary Blood Flow?_Pros
 Debate: Should Ductal Stent Implantation be Considered for All Newborn Infants with Reduced Pulmonary Blood Flow?_Pros Mazeni Alwi Institut Jantung Negara Kuala Lumpur, Malaysia 5 th Asia Pacific Congenital
Debate: Should Ductal Stent Implantation be Considered for All Newborn Infants with Reduced Pulmonary Blood Flow?_Pros Mazeni Alwi Institut Jantung Negara Kuala Lumpur, Malaysia 5 th Asia Pacific Congenital
Coronary Artery from the Wrong Sinus of Valsalva: A Physiologic Repair Strategy
 Coronary Artery from the Wrong Sinus of Valsalva: A Physiologic Repair Strategy Tom R. Karl, MS, MD he most commonly reported coronary artery malformation leading to sudden death in children and young
Coronary Artery from the Wrong Sinus of Valsalva: A Physiologic Repair Strategy Tom R. Karl, MS, MD he most commonly reported coronary artery malformation leading to sudden death in children and young
Survival Rates of Children with Congenital Heart Disease continue to improve.
 DOROTHY RADFORD Survival Rates of Children with Congenital Heart Disease continue to improve. 1940-20% 1960-40% 1980-70% 2010->90% Percentage of children with CHD reaching age of 18 years 1938 First Patent
DOROTHY RADFORD Survival Rates of Children with Congenital Heart Disease continue to improve. 1940-20% 1960-40% 1980-70% 2010->90% Percentage of children with CHD reaching age of 18 years 1938 First Patent
TGA, VSD, and LVOTO. Cheul Lee, MD. Department of Thoracic and Cardiovascular Surgery Sejong General Hospital
 Surgical Management of TGA, VSD, and LVOTO Cheul Lee, MD Department of Thoracic and Cardiovascular Surgery Sejong General Hospital TGA, VSD, and LVOTO Incidence : 0.7% of all CHD 20% of TGA with VSD 4%
Surgical Management of TGA, VSD, and LVOTO Cheul Lee, MD Department of Thoracic and Cardiovascular Surgery Sejong General Hospital TGA, VSD, and LVOTO Incidence : 0.7% of all CHD 20% of TGA with VSD 4%
Outcomes of Mitral Valve Repair for Mitral Regurgitation Due to Degenerative Disease
 Outcomes of Mitral Valve Repair for Mitral Regurgitation Due to Degenerative Disease TIRONE E. DAVID, MD ; SEMIN THORAC CARDIOVASC SURG 19:116-120c 2007 ELSEVIER INC. PRESENTED BY INTERN 許士盟 Mitral valve
Outcomes of Mitral Valve Repair for Mitral Regurgitation Due to Degenerative Disease TIRONE E. DAVID, MD ; SEMIN THORAC CARDIOVASC SURG 19:116-120c 2007 ELSEVIER INC. PRESENTED BY INTERN 許士盟 Mitral valve
Down Syndrome Medical Interest Group Friday, 12 June Cardiac Surgery in patients with Down Syndrome
 Down Syndrome Medical Interest Group Friday, 12 June 2015 Cardiac Surgery in patients with Down Syndrome Mr. Attilio Lotto, FRCS CTh Congenital Cardiac Surgeon Cardiac surgery in patients with Down syndrome
Down Syndrome Medical Interest Group Friday, 12 June 2015 Cardiac Surgery in patients with Down Syndrome Mr. Attilio Lotto, FRCS CTh Congenital Cardiac Surgeon Cardiac surgery in patients with Down syndrome
AORTIC COARCTATION. Synonyms: - Coarctation of the aorta
 AORTIC COARCTATION Synonyms: - Coarctation of the aorta Definition: Aortic coarctation is a congenital narrowing of the aorta, usually located after the left subclavian artery, near the ductus or the ligamentum
AORTIC COARCTATION Synonyms: - Coarctation of the aorta Definition: Aortic coarctation is a congenital narrowing of the aorta, usually located after the left subclavian artery, near the ductus or the ligamentum
Anatomy & Physiology
 1 Anatomy & Physiology Heart is divided into four chambers, two atrias & two ventricles. Atrioventricular valves (tricuspid & mitral) separate the atria from ventricles. they open & close to control flow
1 Anatomy & Physiology Heart is divided into four chambers, two atrias & two ventricles. Atrioventricular valves (tricuspid & mitral) separate the atria from ventricles. they open & close to control flow
Management of a Patient after the Bidirectional Glenn
 Management of a Patient after the Bidirectional Glenn Melissa B. Jones MSN, APRN, CPNP-AC CICU Nurse Practitioner Children s National Health System Washington, DC No Disclosures Objectives qbriefly describe
Management of a Patient after the Bidirectional Glenn Melissa B. Jones MSN, APRN, CPNP-AC CICU Nurse Practitioner Children s National Health System Washington, DC No Disclosures Objectives qbriefly describe
Indication and Technique of Total Correction of Tetralogy of Fallot in 228 Patients
 Indication and Technique of Total Correction of Tetralogy of Fallot in 228 Patients Qingyu Wu, MD Department of Cardiac Surgery, Cardiovascular Institute and Fu Wai Hospital, Chinese Academy of Medical
Indication and Technique of Total Correction of Tetralogy of Fallot in 228 Patients Qingyu Wu, MD Department of Cardiac Surgery, Cardiovascular Institute and Fu Wai Hospital, Chinese Academy of Medical
The Chest X-ray for Cardiologists
 Mayo Clinic & British Cardiovascular Society at the Royal College of Physicians, London : 21-23-October 2013 Cases-Controversies-Updates 2013 The Chest X-ray for Cardiologists Michael Rubens Royal Brompton
Mayo Clinic & British Cardiovascular Society at the Royal College of Physicians, London : 21-23-October 2013 Cases-Controversies-Updates 2013 The Chest X-ray for Cardiologists Michael Rubens Royal Brompton
Morbidity and Mortality Risk Factors in Adults With Congenital Heart Disease Undergoing Cardiac Reoperations
 ORIGINAL ARTICLES: SURGERY: The Annals of Thoracic Surgery CME Program is located online at http://cme.ctsnetjournals.org. To take the CME activity related to this article, you must have either an STS
ORIGINAL ARTICLES: SURGERY: The Annals of Thoracic Surgery CME Program is located online at http://cme.ctsnetjournals.org. To take the CME activity related to this article, you must have either an STS
Heart and Lungs. LUNG Coronal section demonstrates relationship of pulmonary parenchyma to heart and chest wall.
 Heart and Lungs Normal Sonographic Anatomy THORAX Axial and coronal sections demonstrate integrity of thorax, fetal breathing movements, and overall size and shape. LUNG Coronal section demonstrates relationship
Heart and Lungs Normal Sonographic Anatomy THORAX Axial and coronal sections demonstrate integrity of thorax, fetal breathing movements, and overall size and shape. LUNG Coronal section demonstrates relationship
Cardiac MRI in ACHD What We. ACHD Patients
 Cardiac MRI in ACHD What We Have Learned to Apply to ACHD Patients Faris Al Mousily, MBChB, FAAC, FACC Consultant, Pediatric Cardiology, KFSH&RC/Jeddah Adjunct Faculty, Division of Pediatric Cardiology
Cardiac MRI in ACHD What We Have Learned to Apply to ACHD Patients Faris Al Mousily, MBChB, FAAC, FACC Consultant, Pediatric Cardiology, KFSH&RC/Jeddah Adjunct Faculty, Division of Pediatric Cardiology
Hybrid Stage I Palliation / Bilateral PAB
 Hybrid Stage I Palliation / Bilateral PAB Jeong-Jun Park Dept. of Thoracic & Cardiovascular Surgery Asan Medical Center, University of Ulsan CASE 1 week old neonate with HLHS GA 38 weeks Birth weight 3.0Kg
Hybrid Stage I Palliation / Bilateral PAB Jeong-Jun Park Dept. of Thoracic & Cardiovascular Surgery Asan Medical Center, University of Ulsan CASE 1 week old neonate with HLHS GA 38 weeks Birth weight 3.0Kg
Repair of very severe tricuspid regurgitation following detachment of the tricuspid valve
 OPEN ACCESS Images in cardiology Repair of very severe tricuspid regurgitation following detachment of the tricuspid valve Ahmed Mahgoub 1, Hassan Kamel 2, Walid Simry 1, Hatem Hosny 1, * 1 Aswan Heart
OPEN ACCESS Images in cardiology Repair of very severe tricuspid regurgitation following detachment of the tricuspid valve Ahmed Mahgoub 1, Hassan Kamel 2, Walid Simry 1, Hatem Hosny 1, * 1 Aswan Heart
Congenital pulmonary vein (PV) stenosis with anatomically
 Pulmonary Vein Stenosis With Normal Connection: Associated Cardiac Abnormalities and Variable Outcome John P. Breinholt, BS, John A. Hawkins, MD, LuAnn Minich, MD, Lloyd Y. Tani, MD, Garth S. Orsmond,
Pulmonary Vein Stenosis With Normal Connection: Associated Cardiac Abnormalities and Variable Outcome John P. Breinholt, BS, John A. Hawkins, MD, LuAnn Minich, MD, Lloyd Y. Tani, MD, Garth S. Orsmond,
Index. cardiology.theclinics.com. Note: Page numbers of article titles are in boldface type.
 Index Note: Page numbers of article titles are in boldface type. A ACHD. See Adult congenital heart disease (ACHD) Adult congenital heart disease (ACHD), 503 512 across life span prevalence of, 504 506
Index Note: Page numbers of article titles are in boldface type. A ACHD. See Adult congenital heart disease (ACHD) Adult congenital heart disease (ACHD), 503 512 across life span prevalence of, 504 506
IMAGES. in PAEDIATRIC CARDIOLOGY. Abstract
 IMAGES in PAEDIATRIC CARDIOLOGY Images Paediatr Cardiol. 2008 Apr-Jun; 10(2): 11 17. PMCID: PMC3232589 Transcatheter closure of symptomatic aortopulmonary window in an infant F Pillekamp, 1 T Hannes, 1
IMAGES in PAEDIATRIC CARDIOLOGY Images Paediatr Cardiol. 2008 Apr-Jun; 10(2): 11 17. PMCID: PMC3232589 Transcatheter closure of symptomatic aortopulmonary window in an infant F Pillekamp, 1 T Hannes, 1
RESULTS OF 102 CASES OF COMPLETE REPAIR OF CONGENITAL HEART DEFECTS IN PATIENTS WEIGHING 700 TO 2500 GRAMS
 RESULTS OF 102 CASES OF COMPLETE REPAIR OF CONGENITAL HEART DEFECTS IN PATIENTS WEIGHING 700 TO 2500 GRAMS V. Mohan Reddy, MD a Doff B. McElhinney, MD a Theresa Sagrado, BA a Andrew J. Parry, MD a David
RESULTS OF 102 CASES OF COMPLETE REPAIR OF CONGENITAL HEART DEFECTS IN PATIENTS WEIGHING 700 TO 2500 GRAMS V. Mohan Reddy, MD a Doff B. McElhinney, MD a Theresa Sagrado, BA a Andrew J. Parry, MD a David
For Personal Use. Copyright HMP 2013
 12-00415 Case Report J INVASIVE CARDIOL 2013;25(4):E69-E71 A Concert in the Heart. Bilateral Melody Valve Implantation in the Branch Pulmonary Arteries Nicola Maschietto, MD, PhD and Ornella Milanesi,
12-00415 Case Report J INVASIVE CARDIOL 2013;25(4):E69-E71 A Concert in the Heart. Bilateral Melody Valve Implantation in the Branch Pulmonary Arteries Nicola Maschietto, MD, PhD and Ornella Milanesi,
Cover Page. The handle holds various files of this Leiden University dissertation.
 Cover Page The handle http://hdl.handle.net/1887/19123 holds various files of this Leiden University dissertation. Author: Hoohenkerk, Gerard Joannes Franciscus Title: Surgical correction of atrioventricular
Cover Page The handle http://hdl.handle.net/1887/19123 holds various files of this Leiden University dissertation. Author: Hoohenkerk, Gerard Joannes Franciscus Title: Surgical correction of atrioventricular
The complications of cardiac surgery:
 The complications of cardiac surgery: a walk on the Dark Side? Prof Rik De Decker Red Cross Children s Hospital CME Nov/Dec 2011 http://www.cmej.org.za Why should you care? You are about to leave your
The complications of cardiac surgery: a walk on the Dark Side? Prof Rik De Decker Red Cross Children s Hospital CME Nov/Dec 2011 http://www.cmej.org.za Why should you care? You are about to leave your
Fetal Tetralogy of Fallot
 36 Fetal Tetralogy of Fallot E.D. Bespalova, R.M. Gasanova, O.A.Pitirimova National Scientific and Practical Center of Cardiovascular Surgery, Moscow Elena D. Bespalova, MD Professor, Director Rena M,
36 Fetal Tetralogy of Fallot E.D. Bespalova, R.M. Gasanova, O.A.Pitirimova National Scientific and Practical Center of Cardiovascular Surgery, Moscow Elena D. Bespalova, MD Professor, Director Rena M,
