Local injection of stem cell factor (SCF) improves myocardial homing of systemically delivered c-kit 1 bone marrow-derived stem cells
|
|
|
- Osborn Rodgers
- 5 years ago
- Views:
Transcription
1 Cardiovascular Research (2008) 77, doi: /cvr/cvm027 Local injection of stem cell factor (SCF) improves myocardial homing of systemically delivered c-kit 1 bone marrow-derived stem cells Matthias Lutz 1,MarkRosenberg 1, Fabian Kiessling 2, Volker Eckstein 3,ThomasHeger 1,JuttaKrebs 1, Anthony D. Ho 3,HugoA.Katus 1 and Norbert Frey 1 * 1 Department of Cardiology, Internal Medicine III, University of Heidelberg, INF 410, D Heidelberg, Germany; 2 Department of Medical Physics in Radiology, German Cancer Research Center, INF 280, D Heidelberg, Germany and 3 Department of Hematology, Internal Medicine V, University of Heidelberg, INF 410, D Heidelberg, Germany Received 4 May 2007; accepted 24 September 2007; online publish-ahead-of-print 27 September 2007 Time for primary review 18 days KEYWORDS Stem cells; Homing; Myocardial infarction; Stem Cell Factor (SCF) Aims Recent studies have shown that stem cell therapy may alleviate the detrimental effects of myocardial infarction. Yet, most of these reports observed only modest effects on cardiac function, suggesting that there still is need for improvement before widespread clinical use. One potential approach would be to increase migration of stem cells to the heart. We therefore tested whether local administration of stem cell factor (SCF) improves myocardial homing of intravenously infused lin-/c-kitþ stem cells after myocardial infarction. Methods and results Myocardial infarction was induced in mice via ligation of the left anterior descending artery and 2.5 mg of SCF were injected into the peri-infarct zone. Sham-operated mice and animals with intramyocardial injection of phosphate-buffered saline (PBS) served as controls. Twenty-four hours after myocardial infarction, lin-/c-kitþ stem cells were separated from murine bone marrow by magnetic cell sorting, labelled with the green fluorescent cell tracker CFDA or 111 Indium, and subsequently labelled cells were systemically infused via the tail vein. Another 24 or 72 h later, respectively (i.e. 48 and 96 h after myocardial infarction), hearts were removed and analysed for myocardial homing of stem cells. Green fluorescent stem cells were exclusively detected in the peri-infarct zone of animals having prior SCF treatment. Radioactive measurements revealed that an intramyocardial SCF injection significantly amplified myocardial homing of lin-/c-kitþ stem cells compared to animals with PBS injections ( vs cpm/mg/10 6 cpm, þ60%, P, 0.05) and sham-operated mice without myocardial infarction ( vs cpm/mg/10 6 cpm, þ85%, P, 0.01). Similar results were obtained 72 h after stem cell injection. Conclusion We demonstrate that intramyocardial administration of SCF sustainably directs more lin-/ c-kitþ stem cells to the heart. Future studies will have to show whether higher levels of myocardial SCF (i.e. by virus-mediated gene transfer) can further improve homing of systemically delivered c-kitþ stem cells and thus favourably influence cardiac remodelling following myocardial infarction. 1. Introduction The concept of the heart as a terminally differentiated organ unable to replace its myocytes has long been held in cardiovascular research. Accordingly, it has generally been accepted that the postmitotic heart can only sustain an increased workload by hypertrophy of existing myocytes rather than by addition of newly formed cardiomyocytes. 1 More recently, however, this dogma has been challenged These authors contributed equally to this study * Corresponding author. Tel: þ ; fax: þ address: norbert.frey@med.uni-heidelberg.de, freynorbert@ hotmail.com by the identification of a significant number of Y chromosome positive endothelial and myocardial cells in female allografts after gender-mismatched heart transplantation. 2,3 Similarly, patients with a male-to-female bone marrow transplantation display myocardial repopulation of Y chromosome positive cardiomyocytes. 4 Finally, Jackson et al. 5 were able to show that bone marrow transplanted mice subjected to myocardial infarction reveal cardiomyocytes of exogenous origin, consistent with the hypothesis that bone marrow cells can give rise to cardiomyocytes and, at least in principle, could regenerate the myocardium. Numerous studies have thus subsequently examined a potential therapeutic effect of bone marrow-derived cells Published on behalf of the European Society of Cardiology. All rights reserved. & The Author For permissions please journals.permissions@oxfordjournals.org
2 144 M. Lutz et al. on myocardial function and regeneration after experimental myocardial infarction (reviewed by Murry et al. 6 ). Whereas some of these studies provided evidence for extensive myocardial regeneration after cellular cardiomyoplasty, 7 others found no stem cells that had actually transdifferentiated into cardiomyocytes. 8,9 Yet, regardless of the variable effects on cardiac regeneration, virtually all of these studies found a significant improvement of cardiac contractile function after cellular therapy. Therefore, it is now generally accepted that stem cell therapy can favourably affect cardiac remodelling after myocardial infarction, but the scientific basis of this effect still remains unclear. Two recent studies hypothesized that stem cells may exert their beneficial influence on cardiac repair by supporting neoangiogenesis rather than by transdifferentiation into cardiomyocytes. 10,11 Several pathways and cytokines have been implicated in potentially mediating such an effect, including PI3K/akt, 11 thymosin ß4, 12 hepatocyte growth factor 13 as well as stem cell factor (SCF)/c-kit. 10 The latter pathway is of particular interest, since most of the bone marrowderived stem cells so far utilized for cardiac repair are c-kit positive. C-kit is a protooncogene that maps to the white spotting locus (w) and encodes for a receptor tyrosine kinase. C-kit is highly expressed in adult haematopoietic stem cells and binds to its endogenous ligand SCF, which maps to the steel (Sl) locus. 14 Germline mutations in the Sl or W locus revealed that SCF/c-kit signalling is critical for proliferation, differentiation, and migration of immature cells during spermatogenesis and melanocytogenesis. 15,16 Furthermore, it has been shown that c-kit enriched haematopoietic stem cells have the potential to alleviate postischaemic injury in the myocardium. 7,9,10 Based on these studies, we were asking if increased levels of SCF in the myocardium might direct more bone marrowderived c-kit positive stem cells to the infarcted heart. To test this hypothesis, we induced myocardial infarctions in mice and administered recombinant SCF into the periinfarcted area. Next, c-kit positive stem cells that were either marked by a green fluorescent cell tracker or by radioactive labelling were systemically applied by tail vein puncture. We show here that after pre-treatment with SCF significantly more lineage negative (lin-)/c-kitþ cells can be detected in infarcted mouse hearts, suggesting that the novel approach of local SCF delivery improves myocardial homing of bone marrow-derived stem cells. 2. Methods 2.1 Isolation, purification, and labelling of lin-/ c-kit1 stem cells For extraction of lin-/c-kitþ stem cells, the magnetic-activated cell sorting (MACS)-kit from Miltenyi Biotech was used and processed according to the manufacturer s protocol. Briefly, tibias and femurs of C57BL/6 mice were collected and flushed with phosphatebuffered solution (PBS) containing 2% FCS. To remove cell clumps, crude bone marrow was filtered through a 30 mm nylon mesh (Miltenyi Biotech). For the purpose of depleting mature haematopoietic cells such as T cells, B cells, monocytes/macrophages, granulocytes, erythrocytes as well as their committed precursors, cells were incubated with a cocktail of biotinylated antibodies against a panel of lineage (lin) antigens, including CD5, CD45R (B220), CD11b, Anti-Gr-1 (Ly-6G/C), 7-4, and Ter-119 (Lineage Cell Depletion Kit, Miltenyi Biotech). After addition of anti-biotin microbeads, lin-positive cells were separated using a magnetic column. Enrichment of the lineage negative cells for a subpool of cells expressing the stem cell marker c-kit/cd117 was performed in the same way by application of CD117 microbeads (Miltenyi Biotech). The purity of the separated cells was assessed by fluorescenceactivated cell sorting (FACS) using a phycoerythrin labelled antibody against CD117 (Pharmingen). Integrity and viability of purified lin-/c-kitþ cells was confirmed by propidium iodide (PI) (Sigma) staining. In order to be able to detect systemically delivered stem cells after potential engraftment in the heart in vivo, lin-/c-kitþ cells were labelled with the green fluorescent cell tracker carboxyfluorescein diacetate (CFDA) succinimidyl ester (TefLabs). Briefly, lin-/c-kitþ stem cells were incubated for 30 min at 378C with 5 mg/ml CFDA per 10 6 cells. The staining process was concluded with two washing steps to assure clearance of any unbound CFDA. For intravenous infusion, CFDA-labelled stem cells were collected in serum-free PBS and immediately injected. Radioactive labelling of lin-/c-kitþ cells was performed by incubation of stem cells with 1mCi 111 Indium-oxine ( 111 In) (Tyco Healthcare, Germany) per 10 6 cells for 15 min at room temperature. Subsequently, two washing steps removed any unbound radioactivity. Radioactively labelled lin-/c-kitþ stem cells were again collected in serum-free PBS and injected intravenously via the tail-vein. 2.2 Induction of myocardial infarction and application of lin-/c-kit1 stem cells To induce a myocardial infarction, female C57BL/6 mice at an age of 8 12 weeks were intubated and ventilated with 2% isoflurane (Minivent, HSE). After a left-sided thoracotomy, the left anterior descending (LAD) coronary artery was occluded by a permanent ligation (8 0 suture, Ethicon). Myocardial ischaemia was affirmed by pale decolourisation of the depending myocardium. This operation technique was adapted from the first description by Michael et al. 17 Infarct size was determined in four animals 7 days after the procedure. Scar volume was digitally (Image J) calculated by planimetric analysis of representative haematoxylin-eosin (HE) stained adjacent sections of the infarcted heart. Following this procedure, a total of 2.5 mg recombinant mouse stem cell factor (rscf; Sigma-Aldrich) dissolved in 5 ml PBS was injected into 3 5 sites of the peri-infarct zone using a micro syringe (Hamilton) and a 31G canula. Sham injections were performed with identical volumes of PBS without rscf. After closure of the thorax, anaesthesia was stopped and the mice were weaned from the respirator. Postoperative analgesia was ensured with intraperitoneal injections of Buprenorphin (Essex Pharma, Germany). Twenty-four hours after the surgical procedure labelled (CFDA or 111 Indium) lin-/ c-kitþ cells in 200 ml PBS were intravenously administered via the lateral tail vein. To verify complete intravenous application, we used a 27G canula connected to a short polyethylen-catheter. Reflux of blood indicated a secure intravenous position of the canula and allowed complete infusion of the stem cells. After additional 24 or 72 h, respectively, the animals were sacrificed by a lethal dose of thiopental (ALTANA Pharma, Germany) and the organs were harvested. For quantification of radioactivity, hearts were weighed and subsequently measured in a gamma counter (Perkin Elmer/Packard). Collected data were normalized to organ weight and applied activity (cpm per mg organ weight per 10 6 cpm injected activity cpm/mg/10 6 cpm infused). Biometric data of the animals allocated to three different treatment groups (no myocardial infarction, n ¼ 15; myocardial infarction with PBS-injection, n ¼ 16; myocardial infarction with SCF-injection, n ¼ 16) revealed no significant difference (body weight in g: ; ; and , P ¼ n.s.; heart weight in mg: ; ; respectively, P ¼ n.s.). The investigation conforms to the Guide for the Care and Use of Laboratory Animals published by the US National Institutes of Health (NIH Publication No , revised 1996).
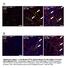
3 Local injection of SCF improves myocardial homing Imunostaining Frozen tissue-blocks were embedded in TissueTec OCT (Sakura) and cut into 7 mm sections, fixed in acetone, and blocked with 2.5% bovine serum albumin (BSA) in PBS for 1 h. As primary antibodies, we used anti-c-kit (C19, Santa Cruz Biotechnology) (1:200), anti- SCF (R&D) (1:200), and anti-sarcomeric alpha-actinin (Sigma) (1:1000), respectively. Antibodies were diluted in blocking solution and incubated for 1 h at room temperature. As secondary antibodies, we utilized Cy3 antibodies against mouse-, rabbit- and goat-igg (Dianova) at a dilution of 1:400. The incubation time was 30 min. Thereafter, slides were washed in PBS, covered with plastic plates, and fixed with mounting medium containing diamidino-2-phenylindol (DAPI) (Vectashield). The sections were examined using a Nikon 80i fluorescence microscope. 2.4 Transthoracic echocardiography Heart function was assessed 2 weeks after stem cell application by echocardiography in anaesthetized mice (2.5% Avertin at 15 ml/g body weight), using a Hewlett Packard Sonos Ultrasound system. The examiner was blinded to the treatment of the mice and obtained three independent M-mode measurements per animal. In the short-axis view, left ventricular end-systolic (LVESD) and left ventricular end-diastolic (LVEDD) chamber diameters and thickness of the interventricular septum and the posterior wall were determined. In order to compare myocardial infarction, left ventricular fractional shortening (FS ¼ [(LVEDD LVESD)/LVEDD] 100) was calculated. 2.5 Statistical analysis Data are presented as mean+sem. The multigroup analysis of the different infarct and sham groups were performed using ANOVA and the Neuman Keuls multiple comparisons post test. P, 0.05 was considered statistically significant. 3. Results 3.1 Magnetic cell sorting yields highly purified lin-/c-kit1 stem cells Lin-/c-kitþ bone marrow cells were isolated from male C57BL/6 mice by magnetic cell sorting, typically resulting in a yield of bone marrow cells per dissected mouse. After the separation process, approximately lin-/c-kitþ stem cells were obtained, corresponding to 1% of the used whole bone marrow cells. FACS analyses were performed to check for purity and viability of the separated stem cells (Figure 1 A C). Figure 1 A shows that a fraction of 1.7% of whole bone marrow cells stains positive for c-kit. Magnetic cell sorting yielded a highly purified cell population with more than 90% of the cells positive for c-kit (Figure 1 B). In addition, more than 95% of the cells displayed no PI labelling, thereby confirming their viability (Figure 1 C). 3.2 Myocardial infarction and stem cell transplantation The study design is outlined in Figure 2. Myocardial infarction was induced under general anaesthesia by direct ligation of the left descending artery (LAD). Determination of the infarct size in a subgroup of four animals revealed scars with a mean volume of cm 3. This corresponded to approximately % of the left ventricular mass (Figure 3 A), similar to previous reports applying this procedure. 17 Shortly after ligation of the LAD, animals were randomly assigned to an injection with 2.5 mg recombinant mouse SCF into the peri-infarct zone (total of 5 ml at 4 5 sites). Control animals received PBS injections. Twentyfour hours later, lin-/c-kitþ cells were labelled with the green fluorescence marker CFDA and subsequently injected into the tail vein by an investigator unaware of the nature of the preceding procedure (SCF vs. PBS). At this timepoint, intramyocardial SCF deposits were still readily detectable by immunostaining (Figure 3 B). 3.3 Lin-/c-kit1 stem cells are detected in stem cell factor-treated mouse hearts To detect intravenously administered lin-/c-kitþ cells in the myocardium, we analysed tissue sections by immunostaining and fluorescence microscopy. Systemically applied stem cells could be differentiated from endogenous cardiac cells using the cell tracker CFDA. One day after application, fluorescent cells were detected in the border zone of the infarction. Interestingly, CFDA-positive cells were typically found in small clusters (Figure 4). Of note, we were only able to detect these cell clusters in hearts with prior SCF treatment. In contrast, in control animals with PBS-injection, no CFDApositive cells could be identified, suggesting that an increase in local SCF abundance facilitated homing of systemically administered c-kitþ stem cells. In addition, the vast majority of green-fluorescent cells also stained positive for c-kit (Figure 5 A C), further supporting the notion that exogenous c-kitþ stem had homed to SCF-treated mouse hearts. On the other hand, we could not detect any cells with c-kit expression without a concomitant CFDA signal, implying a lack of endogenous c-kitþ cells in the mouse heart 24 h post myocardial infarction. In contrast, a recent publication by Fazel et al. 10 reported that 7 days after an experimentally induced myocardial infarction, an increased number of endogenous c-kitþ stem cells from the bone marrow can be found in the heart. This apparent discrepancy might be explained by the earlier time point we used to track transplanted lin-/c-kitþ bone marrow stem cells. Counterstaining of representative tissue-slides of hearts excised 24 and 72 h after stem cell injection with an antibody against alpha-actinin revealed no simultaneous signals for green fluorescent stem cells and the sarcomeric protein alpha-actinin (Figure 5 D F). Therefore, no evidence was found for transdifferentiation of the engrafted cells into cardiomyocytes, at least up to 72 h post transplantation. 3.4 Quantification of radioactively labelled lin-/c-kit1 stem cells reveals a significant improvement of myocardial homing in response to local stem cell factor treatment As described earlier, engrafted c-kitþ stem cells of exogenous origin could be detected in infarcted mouse hearts post local SCF treatment. In order to quantify the improved stem cell homing to the heart, we used radioactive labelling of the lin-/c-kitþ stem cells. For this purpose, c-kitþ stem cells were labelled with the gamma emitter 111 Indium and again systemically administered via tail vein puncture. On the following day, animals were sacrificed and the hearts were removed. Radioactivity of the hearts was measured in a gamma counter. Mice that were treated with intramyocardial injection of SCF (n ¼ 9) after induction of a
4 146 M. Lutz et al. Figure 1 Fluorescence-activated cell sorting (FACS) analysis for characterization of lin-/c-kit positive stem cells: (A) FACS analysis of whole unseparated murine bone marrow cells. Cells were labelled with a c-kit-pe antibody. Quantitative analysis revealed that 1.7% of whole bone marrow cells stained positive for c-kit. (B) After the separation process, 90% of the purified stem cells were c-kit positive; (C) in order to test for viability of the separated stem cells, propidium iodide (PI) staining was performed. Up to 95% of the screened stem cells lack PI -incorporation, thereby confirming viability in the vast majority of purified lin-/c-kitþ stem cells. Figure 2 Cartoon of the study design. Myocardial infarction was induced in mice via ligation of the left anterior descending artery and recombinant mouse stem cell factor (r-mscf) was injected into the peri-infarct zone. Lin-/c-kitþ stem cells (lin-/c-kitþsc) were either labelled with the green fluorescent cell tracker carboxyfluorescein diacetate (CFDA) or 111 Indium ( 111 In). Twenty-four hours later, labelled cells were systemically infused via the tail vein. After another 24 h, the animals were sacrificed and the organs were harvested. Potential engraftment of CFDA positive cell in the myocardium was analysed by immunostaining of tissue sections. In order to quantify myocardial stem cell homing, radioactivity of the hearts was measured in a gamma counter. myocardial infarction revealed a significant 60% increase of emitted radiation compared to controls with a sham-injection of PBS (n ¼ 10) ( vs cpm/mg/10 6 cpm infused, P, 0.05; Figure 6 A). This effect remained statistically significant irrespective of the normalization to applied radioactive activity and/or organ weight (data not shown). When compared to hearts that neither had a myocardial infarction nor a cytokine injection
5 Local injection of SCF improves myocardial homing 147 Figure 3 (A) Myocardial infarction and infarct size: HE staining of a representative slide through the infarcted heart 1 week after permanent ligation of the left anterior descending. At this time, HE-staining and left ventricular thinning clearly delineates the infarct zone from the spared myocardium. Scale bar: 1mm.(B) Intramyocardial deposition of recombinant mouse stem cell factor (SCF): tissue section showing the patchy distribution of recombinant SCF in the myocardium 24 h after direct intramyocardial injection. Recombinant SCF is detected by a polyclonal SCF-antibody. Scalebar: 50 mm. Figure 4 Detection of intramyocardial carboxyfluorescein diacetate (CFDA)- positive cells in infarcted hearts with prior stem cell factor treatment. (A) Clusters of CFDA-positive cells were detected in the peri-infarct zone. (B) Magnified view of a representative CFDA positive cell cluster. (C) Overlay of CFDA positive cells with DAPI. (n ¼ 9), the combination of myocardial infarction and SCF treatment boosted the difference to 85% ( vs cpm/mg/10 6 cpm infused, P, 0.01). Myocardial infarction alone without SCF application resulted in a moderate trend towards increased stem cell homing, but failed to reach statistic significance ( vs cpm/mg/10 6 cpm infused, P. 0.05) (Figure 6 A). To differentiate myocardial engraftment of the injected lin-/ c-kitþ stem cells from a transient phenomenon, a second point of time was chosen according to 111 Indiums half time. Seventy-two hours after stem cell injection animals of the three subgroups were sacrificed and the hearts were removed. Again, hearts of mice that were treated with an intramyocardial injection of SCF (n ¼ 7) showed a significant increase in emitted radiation when compared to animals with myocardial infarction and intramyocardial sham injection (n ¼ 6) or to control animals who neither had an infarction nor an intramyocardial injection (n ¼ 6) ( vs vs cpm/mg/ 10 6 cpm infused; P, 0.05). Myocardial infarction alone without cytokine application only led to a moderate trend towards improved stem cell homing, but again did not reach statistic significance ( vs cpm/mg/10 6 cpm infused; P ¼ n.s.). 4. Discussion While it is still controversial if and to which extent stem cells can actually regenerate infarcted myocardium, several experimental as well as early clinical studies revealed an improved contractile function due to cell therapy, thereby confirming potentially favourable effects of this novel treatment modality. 18 However, in most of these reports, only modest effects on cardiac function were observed, suggesting that there is still a need for significant improvement of stem cell-based therapy before widespread clinical use. One potential approach would be to increase migration and homing of stem cells to the myocardium. We were thus asking if homing of bone marrow-derived stem cells could be enhanced by local delivery of cytokines that could direct these cells to the infarcted heart. C-kit and its endogenous ligand SCF have been shown to be important for mobilization and migration of immature cells. 16,19,20 We thus hypothesized that local delivery of SCF would improve migration of lin-/c-kitþ stem cells to the infarcted heart. Here we demonstrate that direct injection of recombinant SCF into the peri-infarct zone indeed directed significantly more lin-/c-kitþ stem cells of exogenous origin to the infarcted heart compared to control mice without SCF injection. Quantification of myocardial homing was performed by radioactive labelling of haematopoietic stem cells with 111 Indium. In order to exclude a transient phenomenon, measurements were conducted at two different time points (24 and 72 h), revealing a sustained engraftment of systemically injected stem cells in the myocardium. Due to the rapid radioactive decay of 111 Indium, later time points could not be assessed. To the best of our knowledge, this is the first report to show that c-kit positive stem cells applied through a peripheral vein can be attracted to the heart by local injection of a cytokine. What could be the mechanism of this finding? Homing of undifferentiated cells to a peripheral organ consists of a two-step process that begins with binding of stem cells to adhesive molecules in the vasculature followed by local chemotaxis to the site of engraftment. Myocardial accumulation of SCF could serve as an adhesive factor for c-kit positive cells by a direct receptor-ligand interaction in the microcirculation of the heart. Furthermore, a gradient for c-kit positive stem cells could be created by local injection of SCF, thereby potentially facilitating their chemotactic attraction. In line with this notion, we observed exogenous bone marrow-derived stem cells only in the infarct border zone close to the sites of SCF injection. Also, these cells were typically found in small clusters adjacent to small blood vessels, consistent with transmigration from the vasculature. As a result, we noted a significantly higher number of c-kit positive stem cells in the heart after cytokine treatment. A similar observation has been made by Sun et al., 19 who assessed the potential of SCF to direct c-kitþ neural stem cells to injured brain tissue: recombinant SCF strongly induced migration of stem cells through activation of c-kit both in vitro and in vivo. Likewise, Kunisada et al. 16 reported a transgenic mouse model in which overexpression of SCF in keratinocytes led to extensive migration of c-kit positive melanocyte precursors to the skin. Taken together, these findings as well as our data suggest that SCF can act as a potent chemoattractant for c-kit positive cells in vivo. Yet, other mechanisms may also contribute to the observed improvement of myocardial homing of lin-/ c-kitþ cells upon SCF treatment. In this regard, it has been shown that the SCF/c-kit interaction promotes cell
6 148 M. Lutz et al. Figure 5 Co-staining of carboxyfluorescein diacetate (CFDA)-positive cells for c-kit and alpha-actinin reveals maintenance of an undifferentiated phenotype. (A C) Green-fluorescent CFDA-labelled cells (A) also stain positive for c-kit (B) merged with nuclear DAPI staining, indicating that the cells kept their undifferentiated state (C). Note the typical perivascular clustering of systemically delivered exogenous c-kit positive cells. (D F) Green-fluorescent CFDA-labelled cells (D) are negative for alpha-actinin (E) merged with DAPI staining, suggesting that lin-/c-kitþ stem cells did not transdifferentiate into cardiomyocytes(f). Scalebar: 50 mm. Figure 6 Stem cell factor (SCF) pre-treatment enhances myocardial homing of systemically delivered radioactively labelled stem cells. (A) Mice treated with an intramyocardial injection of recombinant stem cell factor (rscf) (n ¼ 9) after induction of myocardial infarction (MI) revealed a significant 60% increase of emitted radiation compared to controls with a sham-injection of PBS (n ¼ 10) ( vs cpm/mg/10 6 cpm infused, P, 0.05) 24 h post infusion. When compared to hearts that neither had a myocardial infarction nor a cytokine injection (n ¼ 9), the combination of myocardial infarction and SCF treatment increased the difference to 85% ( vs cpm/mg/10 6 cpm infused, P, 0.01). Myocardial infarction alone without SCF application resulted in a moderate trend towards increased stem cell homing, but failed to reach statistic significance ( vs cpm/mg/10 6 cpm infused, P. 0.05). Data are presented as meanþsem. (B) Seventy-two hours after stem cell application similar results were obtained. Hearts of mice that were treated with an intramyocardial injection of SCF (n ¼ 7) still showed a significant increase in emitted radiation when compared to animals with myocardial infarction and intramyocardial sham injection (n ¼ 6) or to control animals who neither had an infarction nor an intramyocardial injection (n ¼ 6) ( vs vs cpm/mg/10 6 cpm infused; P, 0.05). Myocardial infarction alone without cytokine application only led to a moderate trend towards improved stem cell homing, but failed to reach statistic significance ( vs cpm/mg/10 6 cpm infused; P ¼ n.s.). Data are presented as meanþsem. survival by inhibition of apoptosis in multiple cell types, including haematopoietic progenitor cells. For example, activation of the receptor tyrosine kinase c-kit by SCF in neurons led to phosphorylation of the antiapoptotic kinase Akt via PI3K and thus conferred a neuroprotective effect. 21 Therefore, it is possible that our observation of an increased number of c-kit positive cells can at least in part be attributed to improved survival of these cells via activation of antiapoptotic pathways. Regardless of the underlying mechanism, there is accumulating evidence that an increased number of c-kit positive cells improve healing and remodelling of the infarcted heart. First, Orlic et al. 7 demonstrated that direct transplantation of c-kit positive bone marrow cells into the border zone of infarcted mouse myocardium led to a significant improvement of contractile function. While Balsam et al. 9 in a very similar experiment could not confirm actual transdifferentiation of the transplanted cells into
7 Local injection of SCF improves myocardial homing 149 cardiomyocytes, they nevertheless observed a significant increase in fractional shortening of cell-treated hearts. More recently, Fazel et al. 10 provided a potential explanation for these advantageous effects even in the absence of newly formed cardiomyocytes: mice with compound heterozygosity for two inactivating mutations of the c-kit receptor displayed a marked reduction in contractile function as well as survival post myocardial infarction compared to wild-type mice. Conversely, transplantation of wild-type, GFP-labelled bone marrow cells rescued the cardiomyopathic phenotype. Mechanistically, these authors could show that the beneficial effect of c-kitþ cells in the heart depends on their ability to create a pro-angiogenic milieu in the infarct border zone with increased expression of VEGF. Interestingly, neither transdifferentiation of c-kitþ bone marrow cells into cardiomyocytes was required for the therapeutic effect, nor the permanent engraftment of these cells in the infarcted myocardium, since very few GFP-positive cells could still be detected 28 days post transplantation. 10 In our experimental model, intramyocardial SCF application led to a patchy and localized distribution pattern. While this approach resulted in considerably higher stem cell numbers in the heart, only a small absolute amount (approximately 400 cells of the initially injected c-kit positive cells) homed to the heart. It was therefore not surprising that increased myocardial homing did not translate into a functional benefit in our experimental set-up, as revealed by echocardiography at 14 days (Supplementary material online, Figure S1). What are the potential clinical implications of these findings? Several investigators already reported small-scale clinical trials in which (usually unfractionated) autologous bone marrow cells were infused into the coronary arteries of patients with a recent myocardial infarction. While some of these studies observed a modest improvement of the ejection fraction in the treatment group, 22,23 there is still no evidence to suggest that the transplanted cells transdifferentiated to cardiomyocytes. This has led to the hypothesis that bone marrow cells infused into the coronaries rather provide cytokines and growth factors which in turn favourably modulate the cardiac adaptation to myocardial infarction. 11,24 While it seems likely that several such factors will be identified, we believe that SCF could be such a molecule which facilitates homing of c-kit positive stem cells to the heart and thereby potentially mediates a therapeutic effect. Conclusion In summary, we provide the proof of concept that enrichment of SCF in the infarcted heart results in significantly increased homing of systemically applied lin-/c-kitþ bone marrow-derived stem cells. Future experiments will have to show if higher levels of SCF homogenously distributed throughout the myocardium (i.e. by virus-mediated gene transfer or transgenesis) can further enhance migration of systemically delivered c-kitþ stem cells to the injured heart. Finally, it will be critical to test if the SCF-mediated enrichment of c-kit positive cells also translates into improved contractile function and thus a favourable effect on cardiac remodelling post myocardial infarction. Supplementary material Supplementary material is available at Cardiovascular Research online. Acknowledgements We want to express our gratitude to Ulrike Engel and Christian Ackermann from the Nikon Imaging Center at the University of Heidelberg, as well as Mrs Dorothea Mews-Zeides from the media centre of the University of Heidelberg for providing excellent technical assistance. Conflict of interest: none declared. Funding This work has been supported by internal funds of the University of Heidelberg. References 1. MacLellan WR, Schneider MD. Genetic dissection of cardiac control pathways. Annu Rev Physiol 2000;62: Quaini F, Urbanek K, Beltrami AP, Finato N, Beltrami C, Nadal-Ginard B et al. Chimerism of the transplanted heart. NEJM 2002;346: Minami E, Laflamme MA, Saffitz JE, Murry CE. Extracardiac progenitor cells repopulate most major cell types in the transplanted human heart. Circulation 2005;112: Deb A, Wang S, Skelding KA, Miller D, Simper D, Caplice NM. Bone marrow-derived cardiomyocytes are present in adult human heart: a study of gender-mismatched bone marrow transplantation patients. Circulation 2003;107: Jackson K, Majka SM, Wang H, Pocius J, Hartley CJ, Majesky MW et al. Regeneration of ischemic cardiac muscle and vascular endothelium by adult stem cells. J Clin Invest 2001;107: Murry CE, Reinecke H, Pabon LM. Regeneration gaps: observations on stem cells and cardiac repair. J Am Coll Cardiol 2006;47: Orlic D, Kajstura J, Chimenti S, Jakoniuk I, Anderson SM, Li B et al. Bone marrow cells regenerate infarcted myocardium. Nature 2001;410: Murry CE, Soonpaa MH, Reinecke H, Nakajima H, Rubart M, Pasumarthi KBS et al. Haematopoietic stem cells do not transdifferentiate into cardiac myocytes in myocardial infarcts. Nature 2004;428: Balsam LB, Wagers AJ, Christensen JL, Kofidis T, Weissman IL, Robbins RC. Haematopoietic stem cells adopt mature haematopoietic fates in ischaemic myocardium. Nature 2004;428: Fazel S, Cimini M, Chen L, Li S, Angoulvant D, Fedak P et al. Cardioprotective c-kitþ cells are from the bone marrow and regulate the myocardial balance of angiogenic cytokines. J Clin Invest 2006;116: Gnecchi M, He H, Noiseaux N, Liang OD, Zhang L, Morello F et al. Evidence supporting paracrine hypothesis for Akt-modified mesenchymal stem cell-mediated cardiac protection and functional improvement. FASEB J 2006;20: Smart N, Risebro CA, Melville AA, Moses K, Schwartz RJ, Chien KR et al. Thymosin ß4 induces adult epicardial progenitor mobilization and neovascularization. Nature 2007;445: Duan HF, Wu CT, Wu DL, Lu Y, Liu HJ, Ha XQ et al. Treatment of myocardial ischemia with bone marrow derived mesenchymal stem cells overexpressing hepatocyte growth factor. Mol Ther 2003;8: Chabot B, Stephenson DA, Chapman VM, Besmer P, Bernstein A. The proto-oncogene c-kit encoding a transmembrane tyrosine kinase receptor maps to the mouse W locus. Nature 1988;335: Ohta H, Yomogida K, Dohmae K, Nishimune Y. Regulation of proliferation and differentiation in spermatogonial stem cells: the role of c-kit and its ligand SCF. Development 2000;127: Kunisada T, Yoshida H, Yamazaki H, Miyamoto A, Hemmi H, Nishimura E et al. Transgene expression of steel factor in the basal layer of epidermis promotes survival, proliferation, differentiation and migration of melanocyte precursors. Development 1998;125:
8 150 M. Lutz et al. 17. Michael LH, Entman ML, Heartley CJ, Youker KA, Zhu J, Hall SR et al. Myocardial ischemia and reperfusion: a murine model. Am J Phys 1995;269: H2147 H Fukada K, Yuasa S. Stem cells as a source of regenerative cardiomyocytes. Circ Res 2006;98: Sun L, Lee J, Fine HA. Neuronally expressed stem cell factor induces neural stem cell migration to areas of brain injury. J Clin Invest 2004; 113: Orlic D, Kajstura J, Chimenti S, Limana F, Jakoniuk I, Quaini F et al. Mobilized bone marrow cells repair the infarcted heart, improving function and survival. PNAS 2001;98: Dhandapani KM, Wade FM, Wakade C, Mahesh VB, Brann DW. Neuroprotection by stem cell factor in rat cortical neurons involves AKT and NFkB. J Neurochem 2005;95: Wollert KC, Meyer GP, Lotz J, Ringes-Lichtenberg S, Lippolt P, Breidenbach C et al. Intracoronary autologous bone-marrow cell transfer after myocardial infarction: the BOOST randomised controlled clinical trial. Lancet 2004;364: Schachinger V, Erbs S, Elsasser A, Haberbosch W, Hambrecht R, Holschermann H et al. Intracoronary Bone Marrow-Derived Progenitor Cells in acute myocardial infarction. N Engl J Med 2006;355: Rosenzweig A. Cardiac Cell Therapy Mixed Results from Mixed Cells. N Engl J Med 2006;355:
Paracrine Mechanisms in Adult Stem Cell Signaling and Therapy
 Paracrine Mechanisms in Adult Stem Cell Signaling and Therapy Massimiliano Gnecchi, Zhiping Zhang, Aiguo Ni, Victor J. Dzau Circulation Research 2008 Nov 21;103(11):1204-19 Introduction(1) After AMI all
Paracrine Mechanisms in Adult Stem Cell Signaling and Therapy Massimiliano Gnecchi, Zhiping Zhang, Aiguo Ni, Victor J. Dzau Circulation Research 2008 Nov 21;103(11):1204-19 Introduction(1) After AMI all
VEGFR2-Mediated Vascular Dilation as a Mechanism of VEGF-Induced Anemia and Bone Marrow Cell Mobilization
 Cell Reports, Volume 9 Supplemental Information VEGFR2-Mediated Vascular Dilation as a Mechanism of VEGF-Induced Anemia and Bone Marrow Cell Mobilization Sharon Lim, Yin Zhang, Danfang Zhang, Fang Chen,
Cell Reports, Volume 9 Supplemental Information VEGFR2-Mediated Vascular Dilation as a Mechanism of VEGF-Induced Anemia and Bone Marrow Cell Mobilization Sharon Lim, Yin Zhang, Danfang Zhang, Fang Chen,
Resident cardiac stem cells: how to find and use them
 Resident cardiac stem cells: how to find and use them G. Hasenfuß Cardiology and Pneumology Heart Research Center Göttingen Georg-August-University Göttingen Definition: Stem cell Selfrenewal Stem cell
Resident cardiac stem cells: how to find and use them G. Hasenfuß Cardiology and Pneumology Heart Research Center Göttingen Georg-August-University Göttingen Definition: Stem cell Selfrenewal Stem cell
ENDOGENOUS CARDIAC STEM CELLS IN THE REGENERATION OF ACUTE AND CHRONIC ISCHEMIC MYOCARDIUM
 ENDOGENOUS CARDIAC STEM CELLS IN THE REGENERATION OF ACUTE AND CHRONIC ISCHEMIC MYOCARDIUM Bernardo Nadal-Ginard, M.D., Ph.D. New York Medical College Angioplasty Summit 2004, Seoul 04/29/04 MYOCARDIAL
ENDOGENOUS CARDIAC STEM CELLS IN THE REGENERATION OF ACUTE AND CHRONIC ISCHEMIC MYOCARDIUM Bernardo Nadal-Ginard, M.D., Ph.D. New York Medical College Angioplasty Summit 2004, Seoul 04/29/04 MYOCARDIAL
SUPPLEMENTARY INFORMATION
 doi:10.1038/nature10188 Supplementary Figure 1. Embryonic epicardial genes are down-regulated from midgestation stages and barely detectable post-natally. Real time qrt-pcr revealed a significant down-regulation
doi:10.1038/nature10188 Supplementary Figure 1. Embryonic epicardial genes are down-regulated from midgestation stages and barely detectable post-natally. Real time qrt-pcr revealed a significant down-regulation
Stem Cells. Keith Channon. Department of Cardiovascular Medicine University of Oxford John Radcliffe Hospital, Oxford
 Stem Cells Keith Channon Department of Cardiovascular Medicine University of Oxford John Radcliffe Hospital, Oxford Adult Stem Cells Unique cells that are capable of self-renewal Have the ability to differentiate
Stem Cells Keith Channon Department of Cardiovascular Medicine University of Oxford John Radcliffe Hospital, Oxford Adult Stem Cells Unique cells that are capable of self-renewal Have the ability to differentiate
Pretargeting and Bioorthogonal Click Chemistry-Mediated Endogenous Stem Cell Homing for Heart Repair
 Pretargeting and Bioorthogonal Click Chemistry-Mediated Endogenous Stem Cell Homing for Heart Repair Mouse Model of Myocardial Infarction (MI) All animal work was compliant with the Institutional Animal
Pretargeting and Bioorthogonal Click Chemistry-Mediated Endogenous Stem Cell Homing for Heart Repair Mouse Model of Myocardial Infarction (MI) All animal work was compliant with the Institutional Animal
Evaluation of directed and random motility in microslides Assessment of leukocyte adhesion in flow chambers
 Evaluation of directed and random motility in microslides Motility experiments in IBIDI microslides, image acquisition and processing were performed as described. PMN, which ended up in an angle < 180
Evaluation of directed and random motility in microslides Motility experiments in IBIDI microslides, image acquisition and processing were performed as described. PMN, which ended up in an angle < 180
Cardiac Myocytes are Recruited by Bone Marrow-Derived Cells in Intact Murine Heart
 Kobe J. Med. Sci., Vol. 48, No. 6, pp. 161-166, 2002 Cardiac Myocytes are Recruited by Bone Marrow-Derived Cells in Intact Murine Heart SEIMI SATOMI-KOBAYASHI 1, SEINOSUKE KAWASHIMA 1*, TSUYOSHI SAKODA
Kobe J. Med. Sci., Vol. 48, No. 6, pp. 161-166, 2002 Cardiac Myocytes are Recruited by Bone Marrow-Derived Cells in Intact Murine Heart SEIMI SATOMI-KOBAYASHI 1, SEINOSUKE KAWASHIMA 1*, TSUYOSHI SAKODA
Therapeutic Potential of Human Umbilical Cord Derived Stem Cells in a Rat Myocardial Infarction Model
 Therapeutic Potential of Human Umbilical Cord Derived Stem Cells in a Rat Myocardial Infarction Model Kai Hong Wu, MD, PhD,* Bin Zhou, PhD,* Cun Tao Yu, MD, Bin Cui, MD, Shi Hong Lu, BS, Zhong Chao Han,
Therapeutic Potential of Human Umbilical Cord Derived Stem Cells in a Rat Myocardial Infarction Model Kai Hong Wu, MD, PhD,* Bin Zhou, PhD,* Cun Tao Yu, MD, Bin Cui, MD, Shi Hong Lu, BS, Zhong Chao Han,
Supplementary Material
 Supplementary Material Induction of myocardial infarction Mice were anesthetized by intraperitoneal injection of pentobarbital (7 mg/kg). In the supine position, endotracheal intubation was performed.
Supplementary Material Induction of myocardial infarction Mice were anesthetized by intraperitoneal injection of pentobarbital (7 mg/kg). In the supine position, endotracheal intubation was performed.
Cell therapy: enhancing the therapeutic potential of cardiac progenitors for delivery post myocardial infarction. Rita Alonaizan
 Cell therapy: enhancing the therapeutic potential of cardiac progenitors for delivery post myocardial infarction Rita Alonaizan Department of Physiology, Anatomy & Genetics St Catherine s College Supervisor:
Cell therapy: enhancing the therapeutic potential of cardiac progenitors for delivery post myocardial infarction Rita Alonaizan Department of Physiology, Anatomy & Genetics St Catherine s College Supervisor:
Cardiac Gene Therapy: Beyond the Mouse. David M Kaye Heart Failure Research Group Baker IDI, Melbourne, AUSTRALIA
 Cardiac Gene Therapy: Beyond the Mouse David M Kaye Heart Failure Research Group Baker IDI, Melbourne, AUSTRALIA Presenter Disclosure Information FINANCIAL DISCLOSURE: Equity: Osprey Medical Grants/Research
Cardiac Gene Therapy: Beyond the Mouse David M Kaye Heart Failure Research Group Baker IDI, Melbourne, AUSTRALIA Presenter Disclosure Information FINANCIAL DISCLOSURE: Equity: Osprey Medical Grants/Research
Multimodality Imaging in Cardiac Stem Cell Research
 Multimodality Imaging in Cardiac Stem Cell Research IL SUK SOHN, MD, PhD Department of Cardiology Kyung Hee University Hospital at Gangdong Kyung Hee University School of Medicine, Seoul, Korea Stem Cell
Multimodality Imaging in Cardiac Stem Cell Research IL SUK SOHN, MD, PhD Department of Cardiology Kyung Hee University Hospital at Gangdong Kyung Hee University School of Medicine, Seoul, Korea Stem Cell
Mesenchymal Stem Cells to Repair Vascular Damage after Chemotherapy: Past, Present and Future
 Mesenchymal Stem Cells to Repair Vascular Damage after Chemotherapy: Past, Present and Future Cell Therapy 2014 Las Vegas, NV, USA Sulaiman Al-Hashmi, PhD Sultan Qaboos University Oman What are MSCs? Stem
Mesenchymal Stem Cells to Repair Vascular Damage after Chemotherapy: Past, Present and Future Cell Therapy 2014 Las Vegas, NV, USA Sulaiman Al-Hashmi, PhD Sultan Qaboos University Oman What are MSCs? Stem
Journal Club Semmler Lorenz
 Beer et al. 2015 - Analysis of the Secretome of Apoptotic Peripheral Blood Mononuclear Cells: Impact of Released Proteins and Exosomes for Tissue Regeneration Journal Club 13.11.2017 1 Introduction to
Beer et al. 2015 - Analysis of the Secretome of Apoptotic Peripheral Blood Mononuclear Cells: Impact of Released Proteins and Exosomes for Tissue Regeneration Journal Club 13.11.2017 1 Introduction to
Suppl Video: Tumor cells (green) and monocytes (white) are seeded on a confluent endothelial
 Supplementary Information Häuselmann et al. Monocyte induction of E-selectin-mediated endothelial activation releases VE-cadherin junctions to promote tumor cell extravasation in the metastasis cascade
Supplementary Information Häuselmann et al. Monocyte induction of E-selectin-mediated endothelial activation releases VE-cadherin junctions to promote tumor cell extravasation in the metastasis cascade
c Ischemia (30 min) Reperfusion (8 w) Supplementary Figure bp 300 bp Ischemia (30 min) Reperfusion (4 h) Dox 20 mg/kg i.p.
 a Marker Ripk3 +/ 5 bp 3 bp b Ischemia (3 min) Reperfusion (4 h) d 2 mg/kg i.p. 1 w 5 w Sacrifice for IF size A subset for echocardiography and morphological analysis c Ischemia (3 min) Reperfusion (8
a Marker Ripk3 +/ 5 bp 3 bp b Ischemia (3 min) Reperfusion (4 h) d 2 mg/kg i.p. 1 w 5 w Sacrifice for IF size A subset for echocardiography and morphological analysis c Ischemia (3 min) Reperfusion (8
Meeting Report. From December 8 to 11, 2012 at Atlanta, GA, U.S.A
 Meeting Report Affiliation Department of Transfusion Medicine and Cell Therapy Name Hisayuki Yao Name of the meeting Period and venue Type of your presentation Title of your presentation The 54 th Annual
Meeting Report Affiliation Department of Transfusion Medicine and Cell Therapy Name Hisayuki Yao Name of the meeting Period and venue Type of your presentation Title of your presentation The 54 th Annual
Supplementary Figure 1. Confocal immunofluorescence showing mitochondrial translocation of Drp1. Cardiomyocytes treated with H 2 O 2 were prestained
 Supplementary Figure 1. Confocal immunofluorescence showing mitochondrial translocation of Drp1. Cardiomyocytes treated with H 2 O 2 were prestained with MitoTracker (red), then were immunostained with
Supplementary Figure 1. Confocal immunofluorescence showing mitochondrial translocation of Drp1. Cardiomyocytes treated with H 2 O 2 were prestained with MitoTracker (red), then were immunostained with
Devices are So Old School: The New World of Myocardial Regeneration
 Devices are So Old School: The New World of Myocardial Regeneration Todd K. Rosengart, M.D. Professor and Chairman DeBakey-Bard Chair of Surgery Michael E. DeBakey Department of Surgery Professor, Texas
Devices are So Old School: The New World of Myocardial Regeneration Todd K. Rosengart, M.D. Professor and Chairman DeBakey-Bard Chair of Surgery Michael E. DeBakey Department of Surgery Professor, Texas
Supporting Information. Calculation of the relative contributions of myocyte proliferation, stem cell. Supporting Information Fig 1 (page 9)
 Supporting Information Table of contents Calculation of the relative contributions of myocyte proliferation, stem cell differentiation and cardioprotection (page 2) Supporting Information Fig 1 (page 9)
Supporting Information Table of contents Calculation of the relative contributions of myocyte proliferation, stem cell differentiation and cardioprotection (page 2) Supporting Information Fig 1 (page 9)
Research progress of adult stem cells and clinical applications
 18 4 2006 8 Chinese Bulletin of Life Sciences Vol. 18, No. 4 Aug., 2006 1004-0374(2006)04-0328-05 100850 Q831 A Research progress of adult stem cells and clinical applications XI Jia-Fei, WANG Yun-Fang,
18 4 2006 8 Chinese Bulletin of Life Sciences Vol. 18, No. 4 Aug., 2006 1004-0374(2006)04-0328-05 100850 Q831 A Research progress of adult stem cells and clinical applications XI Jia-Fei, WANG Yun-Fang,
Cardioprotective c-kit + cells are from the bone marrow and regulate the myocardial balance of angiogenic cytokines
 Related Commentary, page 1838 Research article Cardioprotective c-kit + cells are from the bone marrow and regulate the myocardial balance of angiogenic cytokines Shafie Fazel, 1 Massimo Cimini, 1 Liwen
Related Commentary, page 1838 Research article Cardioprotective c-kit + cells are from the bone marrow and regulate the myocardial balance of angiogenic cytokines Shafie Fazel, 1 Massimo Cimini, 1 Liwen
International Graduate Research Programme in Cardiovascular Science
 1 International Graduate Research Programme in Cardiovascular Science This work has been supported by the European Community s Sixth Framework Programme under grant agreement n LSHM-CT-2005-01883 EUGeneHeart.
1 International Graduate Research Programme in Cardiovascular Science This work has been supported by the European Community s Sixth Framework Programme under grant agreement n LSHM-CT-2005-01883 EUGeneHeart.
Supplemental Figure I
 Supplemental Figure I Kl ( mmol/l)-induced Force orta M (mn) 1 (mn) 1 Supplemental Figure I. Kl-induced contractions. and, Kl ( mmol/l)-induced contractions of the aorta () and those of mesenteric arteries
Supplemental Figure I Kl ( mmol/l)-induced Force orta M (mn) 1 (mn) 1 Supplemental Figure I. Kl-induced contractions. and, Kl ( mmol/l)-induced contractions of the aorta () and those of mesenteric arteries
European Society of Cardiology Congress DONOR AGE NEGATIVELY INFLUENCES THE CYTOPROTECTIVE PARACRINE EFFECTS EXERTED BY HUMAN MESENCHYMAL STEM CELLS
 European Society of Cardiology Congress 28 Aug - 01 Sep 2009, Stockholm - Sweden DONOR AGE NEGATIVELY INFLUENCES THE CYTOPROTECTIVE PARACRINE EFFECTS EXERTED BY HUMAN MESENCHYMAL STEM CELLS Massimiliano
European Society of Cardiology Congress 28 Aug - 01 Sep 2009, Stockholm - Sweden DONOR AGE NEGATIVELY INFLUENCES THE CYTOPROTECTIVE PARACRINE EFFECTS EXERTED BY HUMAN MESENCHYMAL STEM CELLS Massimiliano
Dr. Alexander Lyon Senior Lecturer and Consultant Cardiologist Clinical Lead in Cardio-Oncology Royal Brompton Hospital, London UK
 Advanced heart failure - devices, mechanical circulatory support and cardiac transplantation Monday 30 January 2017 Stem cell and gene therapies for heart failure Dr. Alexander Lyon Senior Lecturer and
Advanced heart failure - devices, mechanical circulatory support and cardiac transplantation Monday 30 January 2017 Stem cell and gene therapies for heart failure Dr. Alexander Lyon Senior Lecturer and
Cardiac repair with thymosin 4 and cardiac reprogramming factors
 Ann. N.Y. Acad. Sci. ISSN 0077-8923 ANNALS OF THE NEW YORK ACADEMY OF SCIENCES Issue: Thymosins in Health and Disease Cardiac repair with thymosin 4 and cardiac reprogramming factors Deepak Srivastava,
Ann. N.Y. Acad. Sci. ISSN 0077-8923 ANNALS OF THE NEW YORK ACADEMY OF SCIENCES Issue: Thymosins in Health and Disease Cardiac repair with thymosin 4 and cardiac reprogramming factors Deepak Srivastava,
Advanced Multi-Layer Speckle Strain Permits Transmural Myocardial Function Analysis in Health and Disease:
 Advanced Multi-Layer Speckle Strain Permits Transmural Myocardial Function Analysis in Health and Disease: Clinical Case Examples Jeffrey C. Hill, BS, RDCS Echocardiography Laboratory, University of Massachusetts
Advanced Multi-Layer Speckle Strain Permits Transmural Myocardial Function Analysis in Health and Disease: Clinical Case Examples Jeffrey C. Hill, BS, RDCS Echocardiography Laboratory, University of Massachusetts
Comparison of Young and Old Cardiac Telocytes Using Atomic Force Microscopy
 Comparison of Young and Old Cardiac Telocytes Using Atomic Force Microscopy Jiali Luo 1, 2, 3, 4, a, Shanshan Feng 1, 2, 3, 4, b 1Key Laboratory of Regenerative Medicine, Ministry of Education, Jinan University,
Comparison of Young and Old Cardiac Telocytes Using Atomic Force Microscopy Jiali Luo 1, 2, 3, 4, a, Shanshan Feng 1, 2, 3, 4, b 1Key Laboratory of Regenerative Medicine, Ministry of Education, Jinan University,
DOWNLOAD PDF CARDIAC REMODELING AND CELL DEATH IN HEART FAILURE
 Chapter 1 : The fibrosis-cell death axis in heart failure Remodeling may be defined as changes in the morphology, structure, and function of the heart related to alterations in loading conditions and/or
Chapter 1 : The fibrosis-cell death axis in heart failure Remodeling may be defined as changes in the morphology, structure, and function of the heart related to alterations in loading conditions and/or
Promoting Fracture Healing Through Systemic or Local Administration of Allogeneic Mesenchymal Stem Cells
 Promoting Fracture Healing Through Systemic or Local Administration of Allogeneic Mesenchymal Stem Cells Gang Li Dept. of Orthopaedics and Traumatology School of Biomedical Sciences, The Chinese University
Promoting Fracture Healing Through Systemic or Local Administration of Allogeneic Mesenchymal Stem Cells Gang Li Dept. of Orthopaedics and Traumatology School of Biomedical Sciences, The Chinese University
Supplementary material page 1/10
 Supplementary Figure 1. Metoprolol administration during ongoing AMI reduces MVO in STEMI patients (a, b) Complete representative CMR exams (short-axis covering the entire left ventricle (LV) from base
Supplementary Figure 1. Metoprolol administration during ongoing AMI reduces MVO in STEMI patients (a, b) Complete representative CMR exams (short-axis covering the entire left ventricle (LV) from base
Endogenous TNFα orchestrates the trafficking of neutrophils into and within lymphatic vessels during acute inflammation
 SUPPLEMENTARY INFORMATION Endogenous TNFα orchestrates the trafficking of neutrophils into and within lymphatic vessels during acute inflammation Samantha Arokiasamy 1,2, Christian Zakian 1, Jessica Dilliway
SUPPLEMENTARY INFORMATION Endogenous TNFα orchestrates the trafficking of neutrophils into and within lymphatic vessels during acute inflammation Samantha Arokiasamy 1,2, Christian Zakian 1, Jessica Dilliway
Protocol. Progenitor Cell Therapy for the Treatment of Damaged Myocardium due to Ischemia
 (20218) Medical Benefit Effective Date: 01/01/11 Next Review Date: 07/18 Preauthorization No Review Dates: 09/10, 07/11, 07/12, 07/13, 07/14, 07/15, 07/16, 07/17 This protocol considers this test or procedure
(20218) Medical Benefit Effective Date: 01/01/11 Next Review Date: 07/18 Preauthorization No Review Dates: 09/10, 07/11, 07/12, 07/13, 07/14, 07/15, 07/16, 07/17 This protocol considers this test or procedure
SUPPLEMENTARY INFORMATION
 doi:10.1038/nature12652 Supplementary Figure 1. PRDM16 interacts with endogenous EHMT1 in brown adipocytes. Immunoprecipitation of PRDM16 complex by flag antibody (M2) followed by Western blot analysis
doi:10.1038/nature12652 Supplementary Figure 1. PRDM16 interacts with endogenous EHMT1 in brown adipocytes. Immunoprecipitation of PRDM16 complex by flag antibody (M2) followed by Western blot analysis
Cardiac Regeneration. Piero Anversa, MD, Annarosa Leri, MD, Jan Kajstura, PHD Valhalla, New York THE HEART AS A POST-MITOTIC ORGAN: THE CONTROVERSY
 Journal of the American College of Cardiology Vol. 47, No. 9, 2006 2006 by the American College of Cardiology Foundation ISSN 0735-1097/06/$32.00 Published by Elsevier Inc. doi:10.1016/j.jacc.2006.02.003
Journal of the American College of Cardiology Vol. 47, No. 9, 2006 2006 by the American College of Cardiology Foundation ISSN 0735-1097/06/$32.00 Published by Elsevier Inc. doi:10.1016/j.jacc.2006.02.003
SUPPLEMENTARY FIG. S2. Representative counting fields used in quantification of the in vitro neural differentiation of pattern of dnscs.
 Supplementary Data SUPPLEMENTARY FIG. S1. Representative counting fields used in quantification of the in vitro neural differentiation of pattern of anpcs. A panel of lineage-specific markers were used
Supplementary Data SUPPLEMENTARY FIG. S1. Representative counting fields used in quantification of the in vitro neural differentiation of pattern of anpcs. A panel of lineage-specific markers were used
Protection against doxorubicin-induced myocardial dysfunction in mice by cardiac-specific expression of carboxyl terminus of hsp70-interacting protein
 Protection against doxorubicin-induced myocardial dysfunction in mice by cardiac-specific expression of carboxyl terminus of hsp70-interacting protein Lei Wang 1, Tian-Peng Zhang 1, Yuan Zhang 2, Hai-Lian
Protection against doxorubicin-induced myocardial dysfunction in mice by cardiac-specific expression of carboxyl terminus of hsp70-interacting protein Lei Wang 1, Tian-Peng Zhang 1, Yuan Zhang 2, Hai-Lian
Eosinophils are required. for the maintenance of plasma cells in the bone marrow
 Eosinophils are required for the maintenance of plasma cells in the bone marrow Van Trung Chu, Anja Fröhlich, Gudrun Steinhauser, Tobias Scheel, Toralf Roch, Simon Fillatreau, James J. Lee, Max Löhning
Eosinophils are required for the maintenance of plasma cells in the bone marrow Van Trung Chu, Anja Fröhlich, Gudrun Steinhauser, Tobias Scheel, Toralf Roch, Simon Fillatreau, James J. Lee, Max Löhning
Effective Targeting of Quiescent Chronic Myelogenous
 Cancer Cell, Volume 7 Supplemental Information Effective Targeting of Quiescent Chronic Myelogenous Leukemia Stem Cells by Histone Deacetylase Inhibitors in Combination with Imatinib Mesylate Bin Zhang,
Cancer Cell, Volume 7 Supplemental Information Effective Targeting of Quiescent Chronic Myelogenous Leukemia Stem Cells by Histone Deacetylase Inhibitors in Combination with Imatinib Mesylate Bin Zhang,
The concept of regenerative medicine using the body s
 MINI-REVIEW: EXPERT OPINIONS Stem Cell Therapy in Perspective Bodo E. Strauer, MD; Ran Kornowski, MD The concept of regenerative medicine using the body s own stem cells and growth factors to repair tissues
MINI-REVIEW: EXPERT OPINIONS Stem Cell Therapy in Perspective Bodo E. Strauer, MD; Ran Kornowski, MD The concept of regenerative medicine using the body s own stem cells and growth factors to repair tissues
Electron micrograph of phosphotungstanic acid-stained exosomes derived from murine
 1 SUPPLEMENTARY INFORMATION SUPPLEMENTARY FIGURES Supplementary Figure 1. Physical properties of murine DC-derived exosomes. a, Electron micrograph of phosphotungstanic acid-stained exosomes derived from
1 SUPPLEMENTARY INFORMATION SUPPLEMENTARY FIGURES Supplementary Figure 1. Physical properties of murine DC-derived exosomes. a, Electron micrograph of phosphotungstanic acid-stained exosomes derived from
Supplementary Figure 1
 Supplementary Figure 1 The average sigmoid parametric curves of capillary dilation time courses and average time to 50% peak capillary diameter dilation computed from individual capillary responses averaged
Supplementary Figure 1 The average sigmoid parametric curves of capillary dilation time courses and average time to 50% peak capillary diameter dilation computed from individual capillary responses averaged
C57BL/6 Mice are More Appropriate. than BALB/C Mice in Inducing Dilated Cardiomyopathy with Short-Term Doxorubicin Treatment
 Original Article C57BL/6 Mice are More Appropriate Acta Cardiol Sin 2012;28:236 240 Heart Failure & Cardiomyopathy C57BL/6 Mice are More Appropriate than BALB/C Mice in Inducing Dilated Cardiomyopathy
Original Article C57BL/6 Mice are More Appropriate Acta Cardiol Sin 2012;28:236 240 Heart Failure & Cardiomyopathy C57BL/6 Mice are More Appropriate than BALB/C Mice in Inducing Dilated Cardiomyopathy
Cell implantation after myocardial infarction: a 10 years experience from the ICREC laboratory
 Cell implantation after myocardial infarction: a 10 years experience from the ICREC laboratory BANFF-SCT Joint Scientific Meeting 2017 Barcelona, 29 th March Santi Roura, PhD Grup ICREC IGTP HuGTiP (Badalona)
Cell implantation after myocardial infarction: a 10 years experience from the ICREC laboratory BANFF-SCT Joint Scientific Meeting 2017 Barcelona, 29 th March Santi Roura, PhD Grup ICREC IGTP HuGTiP (Badalona)
Citation for published version (APA): Velde, S. V. D. (2006). Stem cell-mediated regeneration of the infarcted heart: inflammation rules? s.n.
 University of Groningen Stem cell-mediated regeneration of the infarcted heart Velde, Susanne van der IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish to
University of Groningen Stem cell-mediated regeneration of the infarcted heart Velde, Susanne van der IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish to
Chronic variable stress activates hematopoietic stem cells
 SUPPLEMENTARY INFORMATION Chronic variable stress activates hematopoietic stem cells Timo Heidt *, Hendrik B. Sager *, Gabriel Courties, Partha Dutta, Yoshiko Iwamoto, Alex Zaltsman, Constantin von zur
SUPPLEMENTARY INFORMATION Chronic variable stress activates hematopoietic stem cells Timo Heidt *, Hendrik B. Sager *, Gabriel Courties, Partha Dutta, Yoshiko Iwamoto, Alex Zaltsman, Constantin von zur
Tcf21 MCM ; R26 mtmg Sham GFP Col 1/3 TAC 8W TAC 2W. Postn MCM ; R26 mtmg Sham GFP Col 1/3 TAC 8W TAC 2W
 A Tcf21 MCM ; R26 mtmg Sham GFP Col 1/3 Tcf21 MCM ; R26 mtmg TAC 2W Tcf21 MCM ; R26 mtmg TAC 8W B Postn MCM ; R26 mtmg Sham GFP Col 1/3 Postn MCM ; R26 mtmg TAC 2W Postn MCM ; R26 mtmg TAC 8W Supplementary
A Tcf21 MCM ; R26 mtmg Sham GFP Col 1/3 Tcf21 MCM ; R26 mtmg TAC 2W Tcf21 MCM ; R26 mtmg TAC 8W B Postn MCM ; R26 mtmg Sham GFP Col 1/3 Postn MCM ; R26 mtmg TAC 2W Postn MCM ; R26 mtmg TAC 8W Supplementary
Supplementary Materials. for Garmy-Susini, et al, Integrin 4 1 signaling is required for lymphangiogenesis and tumor metastasis
 Supplementary Materials for Garmy-Susini, et al, Integrin 4 1 signaling is required for lymphangiogenesis and tumor metastasis 1 Supplementary Figure Legends Supplementary Figure 1: Integrin expression
Supplementary Materials for Garmy-Susini, et al, Integrin 4 1 signaling is required for lymphangiogenesis and tumor metastasis 1 Supplementary Figure Legends Supplementary Figure 1: Integrin expression
Stem cells in the dog heart are self-renewing, clonogenic, and multipotent and regenerate infarcted myocardium, improving cardiac function
 Stem cells in the dog heart are self-renewing, clonogenic, and multipotent and regenerate infarcted myocardium, improving cardiac function Axel Linke*, Patrick Müller*, Daria Nurzynska*, Claudia Casarsa*,
Stem cells in the dog heart are self-renewing, clonogenic, and multipotent and regenerate infarcted myocardium, improving cardiac function Axel Linke*, Patrick Müller*, Daria Nurzynska*, Claudia Casarsa*,
Pearson r = P (one-tailed) = n = 9
 8F4-Specific Lysis, % 1 UPN1 UPN3 8 UPN7 6 Pearson r =.69 UPN2 UPN5 P (one-tailed) =.192 4 UPN8 n = 9 2 UPN9 UPN4 UPN6 5 1 15 2 25 8 8F4, % Max MFI Supplementary Figure S1. AML samples UPN1-UPN9 show variable
8F4-Specific Lysis, % 1 UPN1 UPN3 8 UPN7 6 Pearson r =.69 UPN2 UPN5 P (one-tailed) =.192 4 UPN8 n = 9 2 UPN9 UPN4 UPN6 5 1 15 2 25 8 8F4, % Max MFI Supplementary Figure S1. AML samples UPN1-UPN9 show variable
E. Cervio, P. Danieli, C. Ciuffreda, F. Pisano, M. Roccio, M. Gnecchi. The authors have no financial disclosures to declare
 16 th ISCT Annual Meeting SOLUBLE FACTORS RELEASED BY HUMAN MESENCHYMAL STEM CELLS OF FETAL ORIGIN LEAD TO CARDIOMYOCYTE PROTECTION THROUGH THE INHIBITION OF PRO-APOPTOTIC SIGNALING E. Cervio, P. Danieli,
16 th ISCT Annual Meeting SOLUBLE FACTORS RELEASED BY HUMAN MESENCHYMAL STEM CELLS OF FETAL ORIGIN LEAD TO CARDIOMYOCYTE PROTECTION THROUGH THE INHIBITION OF PRO-APOPTOTIC SIGNALING E. Cervio, P. Danieli,
Myocardial Regeneration and Stem Cell Repair
 Myocardial Regeneration and Stem Cell Repair Annarosa Leri, MD, Jan Kajstura, MD, Piero Anversa, MD, and William H. Frishman, MD Abstract: Recent evidence would suggest that the heart is not a terminally
Myocardial Regeneration and Stem Cell Repair Annarosa Leri, MD, Jan Kajstura, MD, Piero Anversa, MD, and William H. Frishman, MD Abstract: Recent evidence would suggest that the heart is not a terminally
ECM1 controls T H 2 cell egress from lymph nodes through re-expression of S1P 1
 ZH, Li et al, page 1 ECM1 controls T H 2 cell egress from lymph nodes through re-expression of S1P 1 Zhenhu Li 1,4,Yuan Zhang 1,4, Zhiduo Liu 1, Xiaodong Wu 1, Yuhan Zheng 1, Zhiyun Tao 1, Kairui Mao 1,
ZH, Li et al, page 1 ECM1 controls T H 2 cell egress from lymph nodes through re-expression of S1P 1 Zhenhu Li 1,4,Yuan Zhang 1,4, Zhiduo Liu 1, Xiaodong Wu 1, Yuhan Zheng 1, Zhiyun Tao 1, Kairui Mao 1,
Case Report Multimodality Imaging of Chronic Ischemia
 SAGE-Hindawi Access to Research Volume 2011, Article ID 739702, 4 pages doi:10.4061/2011/739702 Case Report Multimodality Imaging of Chronic Ischemia Kiyotake Ishikawa, Dennis Ladage, Kleopatra Rapti,
SAGE-Hindawi Access to Research Volume 2011, Article ID 739702, 4 pages doi:10.4061/2011/739702 Case Report Multimodality Imaging of Chronic Ischemia Kiyotake Ishikawa, Dennis Ladage, Kleopatra Rapti,
Cell Combination Therapy. Disclosures
 Cell Combination Therapy Joshua M. Hare, M.D. Louis Lemberg Professor Senior Associate Dean Chief Science Officer Interdisciplinary Stem Cell Institute The Miller School of Medicine, University of Miami
Cell Combination Therapy Joshua M. Hare, M.D. Louis Lemberg Professor Senior Associate Dean Chief Science Officer Interdisciplinary Stem Cell Institute The Miller School of Medicine, University of Miami
Deposited on: 25 July 2011
 Morton, E., Macrae, I.M., McCabe, C., Brown, S.M. and White, F. (2006) Identification of the growth arrest and DNA damage protein GADD34 in the normal human heart and demonstration of alterations in expression
Morton, E., Macrae, I.M., McCabe, C., Brown, S.M. and White, F. (2006) Identification of the growth arrest and DNA damage protein GADD34 in the normal human heart and demonstration of alterations in expression
Cardiomyocyte progenitor cell-derived exosomes stimulate migration of endothelial cells
 Translational Cardiology Most Recent Points of View J. Cell. Mol. Med. Vol 14, No 5, 2010 pp. 1064-1070 Guest Editor: P. Doevendans Cardiomyocyte progenitor cell-derived exosomes stimulate migration of
Translational Cardiology Most Recent Points of View J. Cell. Mol. Med. Vol 14, No 5, 2010 pp. 1064-1070 Guest Editor: P. Doevendans Cardiomyocyte progenitor cell-derived exosomes stimulate migration of
Supplemental Experimental Procedures
 Cell Stem Cell, Volume 2 Supplemental Data A Temporal Switch from Notch to Wnt Signaling in Muscle Stem Cells Is Necessary for Normal Adult Myogenesis Andrew S. Brack, Irina M. Conboy, Michael J. Conboy,
Cell Stem Cell, Volume 2 Supplemental Data A Temporal Switch from Notch to Wnt Signaling in Muscle Stem Cells Is Necessary for Normal Adult Myogenesis Andrew S. Brack, Irina M. Conboy, Michael J. Conboy,
SUPPLEMENTARY DATA. Supplementary Table 2. Antibodies used for Immunofluoresence. Supplementary Table 3. Real-time PCR primer sequences.
 Supplementary Table 2. Antibodies used for Immunofluoresence. Antibody Dilution Source Goat anti-pdx1 1:100 R&D Systems Rabbit anti-hnf6 1:100 Santa Cruz Biotechnology Mouse anti-nkx6.1 1:200 Developmental
Supplementary Table 2. Antibodies used for Immunofluoresence. Antibody Dilution Source Goat anti-pdx1 1:100 R&D Systems Rabbit anti-hnf6 1:100 Santa Cruz Biotechnology Mouse anti-nkx6.1 1:200 Developmental
Follow-up of the CUPID Gene Therapy Trials
 Follow-up of the CUPID Gene Therapy Trials Roger J. Hajjar, MD Icahn School of Medicine at Mount Sinai New York, NY Pathway to Heart Failure Injury / Damage Mature Dysfunctional Dying New Coronary Disease
Follow-up of the CUPID Gene Therapy Trials Roger J. Hajjar, MD Icahn School of Medicine at Mount Sinai New York, NY Pathway to Heart Failure Injury / Damage Mature Dysfunctional Dying New Coronary Disease
Supplementary Figure S1. Flow cytometric analysis of the expression of Thy1 in NH cells. Flow cytometric analysis of the expression of T1/ST2 and
 Supplementary Figure S1. Flow cytometric analysis of the expression of Thy1 in NH cells. Flow cytometric analysis of the expression of T1/ST2 and Thy1 in NH cells derived from the lungs of naïve mice.
Supplementary Figure S1. Flow cytometric analysis of the expression of Thy1 in NH cells. Flow cytometric analysis of the expression of T1/ST2 and Thy1 in NH cells derived from the lungs of naïve mice.
Alternatively Activated Macrophages Determine the Repair of the Infarcted
 Alternatively Activated Macrophages Determine the Repair of the Infarcted Adult Murine Heart (Shiraishi et al.) List of Supplemental Materials Supplemental Methods Supplemental Figure 1. Cardiac CD206
Alternatively Activated Macrophages Determine the Repair of the Infarcted Adult Murine Heart (Shiraishi et al.) List of Supplemental Materials Supplemental Methods Supplemental Figure 1. Cardiac CD206
Supplemental Methods: Histopathology scoring of individual components of Valentino
 Supplementary Materials Online: Supplemental Methods: Histopathology scoring of individual components of Valentino synovitis grade and Mankin cartilage pathology scale Hemophilic synovitis was graded 0-10
Supplementary Materials Online: Supplemental Methods: Histopathology scoring of individual components of Valentino synovitis grade and Mankin cartilage pathology scale Hemophilic synovitis was graded 0-10
Supporting Information
 Supporting Information lpek et al. 1.173/pnas.1121217 SI Materials and Methods Mice. cell knockout, inos / (Taconic arms), Rag1 /, INγR /, and IL-12p4 / mice (The Jackson Laboratory) were maintained and/or
Supporting Information lpek et al. 1.173/pnas.1121217 SI Materials and Methods Mice. cell knockout, inos / (Taconic arms), Rag1 /, INγR /, and IL-12p4 / mice (The Jackson Laboratory) were maintained and/or
Supplemental Information. Tissue Myeloid Progenitors Differentiate. into Pericytes through TGF-b Signaling. in Developing Skin Vasculature
 Cell Reports, Volume 18 Supplemental Information Tissue Myeloid Progenitors Differentiate into Pericytes through TGF-b Signaling in Developing Skin Vasculature Tomoko Yamazaki, Ani Nalbandian, Yutaka Uchida,
Cell Reports, Volume 18 Supplemental Information Tissue Myeloid Progenitors Differentiate into Pericytes through TGF-b Signaling in Developing Skin Vasculature Tomoko Yamazaki, Ani Nalbandian, Yutaka Uchida,
Blocking antibodies and peptides. Rat anti-mouse PD-1 (29F.1A12, rat IgG2a, k), PD-
 Supplementary Methods Blocking antibodies and peptides. Rat anti-mouse PD-1 (29F.1A12, rat IgG2a, k), PD- L1 (10F.9G2, rat IgG2b, k), and PD-L2 (3.2, mouse IgG1) have been described (24). Anti-CTLA-4 (clone
Supplementary Methods Blocking antibodies and peptides. Rat anti-mouse PD-1 (29F.1A12, rat IgG2a, k), PD- L1 (10F.9G2, rat IgG2b, k), and PD-L2 (3.2, mouse IgG1) have been described (24). Anti-CTLA-4 (clone
CD14 + S100A9 + Monocytic Myeloid-Derived Suppressor Cells and Their Clinical Relevance in Non-Small Cell Lung Cancer
 CD14 + S1A9 + Monocytic Myeloid-Derived Suppressor Cells and Their Clinical Relevance in Non-Small Cell Lung Cancer Po-Hao, Feng M.D., Kang-Yun, Lee, M.D. Ph.D., Ya-Ling Chang, Yao-Fei Chan, Lu- Wei, Kuo,Ting-Yu
CD14 + S1A9 + Monocytic Myeloid-Derived Suppressor Cells and Their Clinical Relevance in Non-Small Cell Lung Cancer Po-Hao, Feng M.D., Kang-Yun, Lee, M.D. Ph.D., Ya-Ling Chang, Yao-Fei Chan, Lu- Wei, Kuo,Ting-Yu
Extending the time window of mammalian heart regeneration by thymosin beta 4
 J. Cell. Mol. Med. Vol 18, No 12, 2014 pp. 2417-2424 Extending the time window of mammalian heart regeneration by thymosin beta 4 Liu Rui #, Nie Yu #, Lian Hong, He Feng, Han Chunyong, Meng Jian, Zheng
J. Cell. Mol. Med. Vol 18, No 12, 2014 pp. 2417-2424 Extending the time window of mammalian heart regeneration by thymosin beta 4 Liu Rui #, Nie Yu #, Lian Hong, He Feng, Han Chunyong, Meng Jian, Zheng
Journal Club WS 2012/13 Stefanie Nickl
 Journal Club WS 2012/13 Stefanie Nickl Background Mesenchymal Stem Cells First isolation from bone marrow 30 ys ago Isolation from: spleen, heart, skeletal muscle, synovium, amniotic fluid, dental pulp,
Journal Club WS 2012/13 Stefanie Nickl Background Mesenchymal Stem Cells First isolation from bone marrow 30 ys ago Isolation from: spleen, heart, skeletal muscle, synovium, amniotic fluid, dental pulp,
Direct ex vivo characterization of human antigen-specific CD154 + CD4 + T cells Rapid antigen-reactive T cell enrichment (Rapid ARTE)
 Direct ex vivo characterization of human antigen-specific CD154 + CD4 + T cells Rapid antigen-reactive T cell enrichment (Rapid ARTE) Introduction Workflow Antigen (ag)-specific T cells play a central
Direct ex vivo characterization of human antigen-specific CD154 + CD4 + T cells Rapid antigen-reactive T cell enrichment (Rapid ARTE) Introduction Workflow Antigen (ag)-specific T cells play a central
Exercise in Adverse Cardiac Remodeling: of Mice and Men
 Exercise in Adverse Cardiac Remodeling: of Mice and Men 17-01-2013 Dirk J Duncker Experimental Cardiology, Cardiology, Thoraxcenter Cardiovascular Research Institute COEUR Erasmus MC, University Medical
Exercise in Adverse Cardiac Remodeling: of Mice and Men 17-01-2013 Dirk J Duncker Experimental Cardiology, Cardiology, Thoraxcenter Cardiovascular Research Institute COEUR Erasmus MC, University Medical
MRI ACS-ben. Tamás Simor MD, PhD, Med Hab. University of Pécs, Heart Institute
 MRI ACS-ben Tamás Simor MD, PhD, Med Hab Time Course of Changes in Infarct Size, Viable Myocardium, and LV Mass After Reperfused and Nonreperfused MI Blue lines denote reperfused myocardial infarction
MRI ACS-ben Tamás Simor MD, PhD, Med Hab Time Course of Changes in Infarct Size, Viable Myocardium, and LV Mass After Reperfused and Nonreperfused MI Blue lines denote reperfused myocardial infarction
Radiologic Assessment of Myocardial Viability
 November 2001 Radiologic Assessment of Myocardial Viability Joshua Moss, Harvard Medical School Year III Patient EF 66yo female with a 3-year history of intermittent chest pain previously relieved by sublingual
November 2001 Radiologic Assessment of Myocardial Viability Joshua Moss, Harvard Medical School Year III Patient EF 66yo female with a 3-year history of intermittent chest pain previously relieved by sublingual
In the name of GOD. Animal models of cardiovascular diseases: myocardial infarction & hypertension
 In the name of GOD Animal models of cardiovascular diseases: myocardial infarction & hypertension 44 Presentation outline: Cardiovascular diseases Acute myocardial infarction Animal models for myocardial
In the name of GOD Animal models of cardiovascular diseases: myocardial infarction & hypertension 44 Presentation outline: Cardiovascular diseases Acute myocardial infarction Animal models for myocardial
CT for Myocardial Characterization of Cardiomyopathy. Byoung Wook Choi, Yonsei University Severance Hospital, Seoul, Korea
 CT for Myocardial Characterization of Cardiomyopathy Byoung Wook Choi, Yonsei University Severance Hospital, Seoul, Korea Cardiomyopathy Elliott P et al. Eur Heart J 2008;29:270-276 The European Society
CT for Myocardial Characterization of Cardiomyopathy Byoung Wook Choi, Yonsei University Severance Hospital, Seoul, Korea Cardiomyopathy Elliott P et al. Eur Heart J 2008;29:270-276 The European Society
Chapter. Department of Cardiology, 2 Department of Nuclear Medicine and the 3
 Chapter 9 Saskia L.M.A. Beeres 1 Jeroen J. Bax 1 Petra Dibbets-Schneider 2 Marcel P.M. Stokkel 2 Willem E. Fibbe 3 Ernst E. van der Wall 1 Martin J. Schalij 1 Douwe E. Atsma 1 1 Department of Cardiology,
Chapter 9 Saskia L.M.A. Beeres 1 Jeroen J. Bax 1 Petra Dibbets-Schneider 2 Marcel P.M. Stokkel 2 Willem E. Fibbe 3 Ernst E. van der Wall 1 Martin J. Schalij 1 Douwe E. Atsma 1 1 Department of Cardiology,
Detection and Assessment of MI: Use of Imaging Methods. Robert O. Bonow, M.D.
 Detection and Assessment of MI: Use of Imaging Methods Robert O. Bonow, M.D. Detection and Assessment of MI: Use of Imaging Methods Robert O. Bonow, M.D. No Relationships to Disclose Expert Consensus Document
Detection and Assessment of MI: Use of Imaging Methods Robert O. Bonow, M.D. Detection and Assessment of MI: Use of Imaging Methods Robert O. Bonow, M.D. No Relationships to Disclose Expert Consensus Document
Mesenchymal Stem Cells and Cancer: Their Interplay
 Mesenchymal Stem Cells and Cancer: Their Interplay Gang Li, MBBS, DPhil (Oxon) Stem Cell and Regeneration Program School of Biomedical Sciences Li Ka Shing Institute of Health Sciences Department of Orthopaedics
Mesenchymal Stem Cells and Cancer: Their Interplay Gang Li, MBBS, DPhil (Oxon) Stem Cell and Regeneration Program School of Biomedical Sciences Li Ka Shing Institute of Health Sciences Department of Orthopaedics
Autologous bone marrow stem cell transplantation,
 Tissue Distribution of F-FDG-Labeled Peripheral Hematopoietic Stem Cells After Intracoronary Administration in Patients with Myocardial Infarction Won Jun Kang 1, Hyun-Jae Kang 2, Hyo-Soo Kim 2, June-Key
Tissue Distribution of F-FDG-Labeled Peripheral Hematopoietic Stem Cells After Intracoronary Administration in Patients with Myocardial Infarction Won Jun Kang 1, Hyun-Jae Kang 2, Hyo-Soo Kim 2, June-Key
The cardiomyocyte cell cycle
 DePauw University Scholarly and Creative Work from DePauw University Biology Faculty publications Biology 2006 The cardiomyocyte cell cycle Pascal J. Lafontant DePauw University, pascallafontant@depauw.edu
DePauw University Scholarly and Creative Work from DePauw University Biology Faculty publications Biology 2006 The cardiomyocyte cell cycle Pascal J. Lafontant DePauw University, pascallafontant@depauw.edu
Supplementary Figure 1. Characterization of basophils after reconstitution of SCID mice
 Supplementary figure legends Supplementary Figure 1. Characterization of after reconstitution of SCID mice with CD4 + CD62L + T cells. (A-C) SCID mice (n = 6 / group) were reconstituted with 2 x 1 6 CD4
Supplementary figure legends Supplementary Figure 1. Characterization of after reconstitution of SCID mice with CD4 + CD62L + T cells. (A-C) SCID mice (n = 6 / group) were reconstituted with 2 x 1 6 CD4
CD34+ Cells: A Comparison of Stem and Progenitor Cells in Cord Blood, Peripheral Blood, and the Bone Marrow
 White Paper September 2016 CD34+ Cells: A Comparison of Stem and Progenitor Cells in Cord Blood, Peripheral Blood, and the Bone Marrow Lily C. Trajman, PhD Introduction: Hematopoietic Stem Cells (HSCs)
White Paper September 2016 CD34+ Cells: A Comparison of Stem and Progenitor Cells in Cord Blood, Peripheral Blood, and the Bone Marrow Lily C. Trajman, PhD Introduction: Hematopoietic Stem Cells (HSCs)
ENGINEERING THREE DIMENSIONAL CARDIOSPHERES FROM PLURIPOTENT STEM CELLS
 ENGINEERING THREE DIMENSIONAL CARDIOSPHERES FROM PLURIPOTENT STEM CELLS A Thesis Presented to The Academic Faculty by Nicole Votaw In Partial Fulfillment of the Requirements for the Degree B.S. in Biomedical
ENGINEERING THREE DIMENSIONAL CARDIOSPHERES FROM PLURIPOTENT STEM CELLS A Thesis Presented to The Academic Faculty by Nicole Votaw In Partial Fulfillment of the Requirements for the Degree B.S. in Biomedical
Progenitor Cell Therapy for the Treatment of Damaged Myocardium due to Ischemia. Original Policy Date
 MP 2.02.14 Progenitor Cell Therapy for the Treatment of Damaged Myocardium due to Ischemia Medical Policy Section Medicine Issue 12:2013 Original Policy Date 12:2013 Last Review Status/Date Reviewed with
MP 2.02.14 Progenitor Cell Therapy for the Treatment of Damaged Myocardium due to Ischemia Medical Policy Section Medicine Issue 12:2013 Original Policy Date 12:2013 Last Review Status/Date Reviewed with
Cardiomyocyte proliferation and progenitor cell recruitment underlie therapeutic regeneration after myocardial infarction in the adult mouse heart
 OPEN ACCESS Cardiomyocyte proliferation and progenitor cell recruitment underlie therapeutic regeneration after myocardial infarction in the adult mouse heart Konstantinos Malliaras, Yiqiang Zhang, Jeffrey
OPEN ACCESS Cardiomyocyte proliferation and progenitor cell recruitment underlie therapeutic regeneration after myocardial infarction in the adult mouse heart Konstantinos Malliaras, Yiqiang Zhang, Jeffrey
Role of Inflammation in Pulmonary Hypertension
 Role of Inflammation in Pulmonary Hypertension K. R. Stenmark University of Colorado Denver, USA Prominent Fibroproliferative Changes are Observed in the Lung Vasculature of Infants With Pulmonary Arterial
Role of Inflammation in Pulmonary Hypertension K. R. Stenmark University of Colorado Denver, USA Prominent Fibroproliferative Changes are Observed in the Lung Vasculature of Infants With Pulmonary Arterial
SUPPLEMENTARY MATERIALS. IL-4 as a Repurposed Biological Drug for Myocardial Infarction through. Augmentation of Reparative Cardiac Macrophages:
 1 SUPPLEMENTARY MATERIALS IL-4 as a Repurposed Biological Drug for Myocardial Infarction through Augmentation of Reparative Cardiac Macrophages: Proof-of-Concept Data in Mice Yusuke Shintani MD PhD, Tomoya
1 SUPPLEMENTARY MATERIALS IL-4 as a Repurposed Biological Drug for Myocardial Infarction through Augmentation of Reparative Cardiac Macrophages: Proof-of-Concept Data in Mice Yusuke Shintani MD PhD, Tomoya
Supplementary Materials for
 www.sciencesignaling.org/cgi/content/full/8/375/ra41/dc1 Supplementary Materials for Actin cytoskeletal remodeling with protrusion formation is essential for heart regeneration in Hippo-deficient mice
www.sciencesignaling.org/cgi/content/full/8/375/ra41/dc1 Supplementary Materials for Actin cytoskeletal remodeling with protrusion formation is essential for heart regeneration in Hippo-deficient mice
SUPPLEMENTARY INFORMATION
 Complete but curtailed T-cell response to very-low-affinity antigen Dietmar Zehn, Sarah Y. Lee & Michael J. Bevan Supp. Fig. 1: TCR chain usage among endogenous K b /Ova reactive T cells. C57BL/6 mice
Complete but curtailed T-cell response to very-low-affinity antigen Dietmar Zehn, Sarah Y. Lee & Michael J. Bevan Supp. Fig. 1: TCR chain usage among endogenous K b /Ova reactive T cells. C57BL/6 mice
SUPPLEMENTARY INFORMATION
 doi:10.1038/nature12215 Supplementary Figure 1. The effects of full and dissociated GR agonists in supporting BFU-E self-renewal divisions. BFU-Es were cultured in self-renewal medium with indicated GR
doi:10.1038/nature12215 Supplementary Figure 1. The effects of full and dissociated GR agonists in supporting BFU-E self-renewal divisions. BFU-Es were cultured in self-renewal medium with indicated GR
Anti-ceramide Antibody Prevents the Radiation GI Syndrome in Mice
 Anti-ceramide Antibody Prevents the Radiation GI Syndrome in Mice Jimmy A. Rotolo 1, Branka Stancevic 1, Jianjun Zhang 1, Guoqiang Hua 1, John Fuller 1, Xianglei Yin 1, Adriana Haimovitz-Friedman 2, Kisu
Anti-ceramide Antibody Prevents the Radiation GI Syndrome in Mice Jimmy A. Rotolo 1, Branka Stancevic 1, Jianjun Zhang 1, Guoqiang Hua 1, John Fuller 1, Xianglei Yin 1, Adriana Haimovitz-Friedman 2, Kisu
(a) Significant biological processes (upper panel) and disease biomarkers (lower panel)
 Supplementary Figure 1. Functional enrichment analyses of secretomic proteins. (a) Significant biological processes (upper panel) and disease biomarkers (lower panel) 2 involved by hrab37-mediated secretory
Supplementary Figure 1. Functional enrichment analyses of secretomic proteins. (a) Significant biological processes (upper panel) and disease biomarkers (lower panel) 2 involved by hrab37-mediated secretory
Ischemic heart disease
 Ischemic heart disease Introduction In > 90% of cases: the cause is: reduced coronary blood flow secondary to: obstructive atherosclerotic vascular disease so most of the time it is called: coronary artery
Ischemic heart disease Introduction In > 90% of cases: the cause is: reduced coronary blood flow secondary to: obstructive atherosclerotic vascular disease so most of the time it is called: coronary artery
SUPPLEMENTAL MATERIAL. Supplementary Methods
 SUPPLEMENTAL MATERIAL Supplementary Methods Culture of cardiomyocytes, fibroblasts and cardiac microvascular endothelial cells The isolation and culturing of neonatal rat ventricular cardiomyocytes was
SUPPLEMENTAL MATERIAL Supplementary Methods Culture of cardiomyocytes, fibroblasts and cardiac microvascular endothelial cells The isolation and culturing of neonatal rat ventricular cardiomyocytes was
SUPPLEMENTARY INFORMATION
 doi:10.1038/nature10134 Supplementary Figure 1. Anti-inflammatory activity of sfc. a, Autoantibody immune complexes crosslink activating Fc receptors, promoting activation of macrophages, and WWW.NATURE.COM/NATURE
doi:10.1038/nature10134 Supplementary Figure 1. Anti-inflammatory activity of sfc. a, Autoantibody immune complexes crosslink activating Fc receptors, promoting activation of macrophages, and WWW.NATURE.COM/NATURE
SUPPLEMENTARY INFORMATION
 a c e doi:10.1038/nature10407 b d f Supplementary Figure 1. SERCA2a complex analysis. (a) Two-dimensional SDS-PAGE gels of SERCA2a complexes. A silver-stained SDSPAGE gel is shown, which reveals a 12 kda
a c e doi:10.1038/nature10407 b d f Supplementary Figure 1. SERCA2a complex analysis. (a) Two-dimensional SDS-PAGE gels of SERCA2a complexes. A silver-stained SDSPAGE gel is shown, which reveals a 12 kda
Strategic Research Development in Stem Cell and Regenerative Medicine in HKU Professor Sum-ping Lee Dean HKU Li Ka Shing Faculty of Medicine
 Strategic Research Development in Stem Cell and Regenerative Medicine in HKU Professor Sum-ping Lee Dean HKU Li Ka Shing Faculty of Medicine Adult stem cells Cells that are capable of self renewal and
Strategic Research Development in Stem Cell and Regenerative Medicine in HKU Professor Sum-ping Lee Dean HKU Li Ka Shing Faculty of Medicine Adult stem cells Cells that are capable of self renewal and
