Developmental Changes in the Expression of Rabbit Left Ventricular Troponin T
|
|
|
- Damon Gilbert
- 5 years ago
- Views:
Transcription
1 742 Developmental Changes in the Expression of Rabbit Left Ventricular Troponin T Page A.W. Anderson, Gudrun E. Moore, and Rashid N. Nassar We examined cardiac troponin T (TnT) isoform expression in rabbit left ventricular myocardium at three different stages of postnatal development. Using sodium dodecyl sulfate gel electrophoresis (PAGE), we resolved five isoforms: TnT,, TnT 2, TnT 3, TnT 4, and TnT s. TnT, had the slowest electrophoretic mobility and TnT 5 the fastest. The predominant isoforms were TnT 2, TnT 3, and TnT 4. The relative amounts of TnT 2, TnT 3, TnT 4, and TnT 5 were examined in myocardium from three age-groups: 3 days (Group 1), days (Group 2), and days (Group 3). The amount of TnT 2 relative to the total amount of TnT (determined by the ratio of the areas under the densitometric curves) decreased significantly (p<0.01) with age from 42 ±4% in Group 1 to 25 ±3% in Group 3. In contrast, the relative amount of TnT 4 increased with age from 23 ± 2% in Group 1 to 33 ± 4% in Group 3 (p<0.01). The relative amounts of the other two isoforms changed biphasically with development: TnT 3 decreased from Group 1 to Group 2 and increased from Group 2 to Group 3. TnT 5, a minor isoform, increased from Group 1 to Group 2 and decreased from Group 2 to Group 3. These developmental changes in troponin T expression may account for some of the maturational changes observed in the physiological and biochemical properties of the myocardium. (Circulation Research 1988;63: ) Troponin T (TnT) binds the troponin complex to tropomyosin and is essential in the interaction of TnT, troponin I (Tnl), and troponin C (TnC), which confers on the myofilaments their sensitivity to calcium. 1 In avian fast skeletal muscle, multiple TnT isoforms are present, and with development, both their expression and the sensitivity of the myofilaments to calcium change. 23 In the mammal, five TnT isoforms have been commonly observed in fast skeletal muscle. 4 This expression, which is believed to occur through differential RNA splicing, 5 is correlated with the sensitivity of the skeletal muscle fibers to calcium. 6 Mammalian ventricular myocardium has recently been found to contain more than one isoform of TnT. Gusev et al 7 found two isoforms in bovine myocardium, while Pearlstone et al, 8 using peptide sequence analysis, found at least two major isoforms in rabbit ventricular myocardium. This peptide analysis also dem- From the Departments of Pediatrics (P.A.W.A.) and Physiology (P.A.W.A. and R.N.N.), Duke University Medical Center, Durham, North Carolina, and the Department of iochemistry (G.E.M.), St. Mary's Hospital Medical School, London, England. Supported in part by National Institutes of Health grants HL-12486, HL-20749, and HL and American Heart Association Grant Address for correspondence: Page A.W. Anderson, MD, Post Office ox 3218, Duke University Medical Center, Durham, NC Received November 4, 1987; accepted May 5, onstrated that rabbit cardiac TnT and rabbit skeletal TnT differed so sufficiently that they most likely have different genes. In a study that assessed the functional effects of the two bovine cardiac TnT isoforms, myofibrillar ATPase activity was found to depend on the isoform of TnT present. 9 In chicken cardiac muscle, TnT expression has been shown to vary with development: one form of TnT mrna was more predominant in early embryonic life, another more predominant in the adult. 10 This is the first study that examines whether, in the mammalian heart, TnT isoform expression changes with development. We found that, like rabbit fast skeletal muscle, 4 rabbit cardiac ventricle expresses five isoforms of TnT. The relative expression of the three major cardiac TnT isoforms was found to change significantly with development. Materials and Methods New Zealand White rabbits were examined at three different postnatal ages: Group 1, 3 days old (n = 8); Group 2, days old (n = 6); and Group 3, days old (n = 5). Each rabbit came from a different litter. The rabbits were anesthetized with sodium pentobarbital (350 mg/kg body weight i.p.). The hearts were removed and washed in Krebs- Henseleit solution (KH) (ph 7.4). The left ventricular free wall was dissected from the heart and placed in a dish cooled on ice.
2 Anderson et al Mammalian Left Ventricular Troponin T Preparation of Samples for Developmental Comparisons Protease inhibitors were placed in all buffers (0.5 /ig/ml antipain, 0.5 /xg/ml chymostatin, 0.5 ^g/ml pepstatin, 2.0 jtg/ml a-macroglobulin, 100 units/ml trasylol, 0.5 mm phenylmethylsulfonyl fluoride, and 2.0 jig/ml leupeptin). Pieces of myocardium, approximately 1 mm3, were cut from the central portion of the free wall and immediately placed in 0.6 ml of skinning solution (75 mm KC2H3O2,15 mm KPO4,5 mm K2EGTA, 5 mm MgCl2, 5 mm K2ATP, 0.5% Triton X-100, and mm MOPS, ph 7) chilled on ice. After 5-10 minutes, the tissue was washed twice in KH and placed in 200 fil of sample buffer (2.0% sodium dodecyl sulfate [SDS], 50 mm Tris [ph 6.8], 20% glycerol, % bromophenol blue, and 1.0% /3-mercaptoethanol). The sample was then frozen at - 80 C until the proteins were examined by SDS polyacrylamide gel electrophoresis (SDSPAGE), which followed the Laemmli approach and used a 3.3% stacking gel and a 1.1% bis-acrylamide concentration.11 Electrophoretic mobility of the left ventricular proteins was examined by allowing them to migrate through an 8% acrylamide separating gel. Standards were run on each gel, including the TnT standard purified from rabbit heart (see below) and canine cardiac actin (kindly provided by Dr. R. John Solaro, University of Illinois at Chicago). Gels were silver-stained and photographed.12 Positive transparencies made from the gels were scanned with an LK Ultroscan laser densitometer (romma, Sweden). The relative amounts of the TnT isoforms and actin were determined by integrating the areas under the densitometric curves. A previous study of the silver stain technique demonstrated that optical density had a linear relation to protein mass from 2 to 70 ng.12 We have found that when excessive loading is avoided, variations in the protein load do not affect the proportions of the isoforms. The amount of each isoform was expressed as a percentage of total TnT. The contribution of TnT, to the total TnT content was not examined because this minor isoform migrated at a rate so close to that of actin that it could not be separated easily from actin on an 8% gel (see Figure 1). The data from the three age-groups were examined statistically using analysis of variance. The arcsine transformation was applied to the data to approximate a normal distribution. Isolation of Thin Filament Proteins Standards were prepared from rabbit ventricles (n= 12, 6 weeks to 3 months of age). All procedures were performed at 4 C to avoid proteolysis.13 TnT and Tnl were prepared (Figure 1) by conventional fractionation.13 This procedure included ammonium sulfate extraction, CM Sephadex chromatography, and DEAE Sephadex chromatography. Tropomyosin (Figure 1) was obtained as a by-product of the ,000 67,000 43,000 _ Actin ]TnT Tm Tnl I A C D TnT, TnT 2 TnT 3 E F Actin TnT* FIGURE 1. The upper panel shows contractile protein standards (troponin T, troponin I, and tropomyosin, 8% polyacrylamide gel) purified from rabbit ventricular myocardium. The five isoforms of troponin T (TnT) are indicated in both upper and lower panels: A, low molecular weight standards obtained from LK, romma, Sweden;, actin; C, troponin T and a small amount of troponin I (Tnl); D, a mixture of actin and troponin T; E, tropomyosin (Tm); F, troponin T and I. Lower panel is a 6% polyacrylamide gel: A, a mixture of actin and troponin T isoforms (TnT,, TnT2, TnT3, TnT4, and TnT5);, troponin T isoforms; C, actin. preparation of troponin13 by dialyzing the pellet from the % ammonium sulfate extraction into a buffer solution containing 100 mm KC1, 50 mm Tris, and 0.1 mm dithiothreitol, ph 8.0. The protein was loaded on a DEAE Sephadex column, equilibrated with buffer and eluted with a gradient of mm KC1. The isolation steps were monitored by SDS-PAGE. The standards were stored at -80 C. Results TnT, purified from rabbit ventricular myocardium, was separated by SDS-PAGE into five isoforms: TnT,, TnT2, TnT3, TnT4> and TnT3 (see Figure 1). These isoforms migrated faster than actin
3 744 Circulation Research Vol 63, No 4, October , ,000 < Myosin r i _ Actin ]TnT 39, A C D E F G H I J K L M N O P Q R FIGURE 2. An 8% potyacrylamide gel contrasting the left ventricular proteins at the three different stages of development. Each lane contains the proteins from a different heart: three-day-old, lanes, E, I, L, and P; 21- to 22-day-oldgroup, lanes C, F, J, M, and Q; 99- to 109-day-old group, lanes D, G, K, N, andr. Remaining lanes: A, pierce low molecular weight standard; H and O, mixture of actin and purified rabbit cardiac troponin T (TnT). and slower than tropomyosin and Tnl (Figure 1), with TnT, having the slowest mobility and TnT5 the fastest. TnT2, TnT3, and TnT4 were the predominant forms in the purified standard and in the ventricular myocardium. With the molecular weight standards as a reference (Figure 2), Mr of the TnT isoforms ranged around 39,000 daltons. TnT Isoform Expression During Development At the three stages of development examined, the left ventricle contained proteins with electrophoretic mobilities similar to purified TnT standard (Figure 2). The isoforms TnT2, TnT3, TnT4, and TnT3 were readily identified at each age. The changes with development of the major isoforms, TnT2, TnT3, and TnT4, are evident in Figure 2. The densitometric scans of three representative lanes (Figure 3) further illustrate these changes. The amounts of TnT2, TnT3, TnT4, and TnTj, expressed as percentages of the total TnT content, changed with development (see Figure 4 and Table 1). TnT2 decreased, TnT3 decreased then increased, TnT4 increased, and TnT3 increased then decreased with age. Analysis of variance showed significant F values across development (p<0.002). Paired comparisons using Tukey's HSD test showed that differences between all pairs of means were significant at the 0.05 level. The following differences between pairs were significant at the 0.01 level: TnT2, groups 1 and 2, groups 1 and 3, groups 2 and 3; TnT3, groups 1 and 3, groups 2 and 3; and TnT4, groups 1 and 2, groups 1 and 3, groups 2 and 3; TnT5, groups 1 and 2. Discussion Our results provide the first evidence that expression of mammalian cardiac TnT, an essential molecule in the control of contraction and relaxation, changes significantly with maturation. In rabbit fast skeletal muscle, where five TnT isoforms are commonly found,4 the sensitivity of the myofilaments to calcium was found to be related to the TnT isoform content of the single fiber.6 Thus, it is reasonable to suspect that the developmental changes in cardiac TnT isoform expression observed in this study could similarly result in changes in the sensitivity of the cardiac myofilaments to calcium. Such a change would contribute to the developmental changes in myocardial contractility.14 Our results strongly support ourfindingfiveisoforms of TnT in rabbit cardiac muscle. We used conventional procedures for cardiac TnT purification13 in this study to resolve the five isoforms. Pearlstone et al,8 using a modified purification procedure, found at least two major isoforms of TnT in rabbit myocardium. Our observation of five TnT isoforms in rabbit myocardium is consistent with the expression of multiple isoforms reported in studies of other striated muscles. These studies reported five or more TnT isoforms in rabbit and chicken fast skeletal muscle,2-5 two TnT isoforms in bovine cardiac muscle, 79 and two TnT mrna in chicken cardiac muscle.10
4 Anderson et al MnmmaHan Left Ventricular Troponln T 745, Actin TnT 4 TnT. FIGURE 3. Comparison of rabbit left ventricular troponin Tisoforms (TnT 2, TnT 3, TnT 4, and TnT s ) at three ages. Densitometric scans of three lanes of an 8% potyacrylamide gel in the region to which actin and the TnT isoforms migrated: Upper panel, 3-day-old; middle panel, 21-day-old; lower panel, 99-day-old. Developmental changes in proportions of TnT isoforms for all hearts are illustrated graphically in Figure 4 and presented in Table 1. In a separate, ongoing immunological and molecular investigation of the TnT isoforms of rabbit myocardium, we have raised a polyclonal antibody to the TnT isoform TnT 4. Immunoblots demonstrate that this antibody recognizes the five bands characterized as TnT isoforms in this report. This study will be the subject of another communication. We have performed two-dimensional gel electrophoresis on rabbit ventricular myocardium in a separate study 15 and reported five protein spots that comigrated with the purified TnT, each exhibiting two isoelectric points. That study and the present one provide an M r estimate for the TnT isoforms between 38,500 and 39,500 daltons. The two bovine cardiac TnT isoforms have a comparable M r, as estimated by gel electrophoresis. 7 The M r of rabbit cardiac TnT obtained from amino acid sequence is 32,881 daltons. 8 The difference between the M r obtained from the amino acid sequence and that estimated using gel electrophoresis is similar to the difference observed for the M r of skeletal muscle TnT using these two approaches. 16 The developmental differences in TnT expression observed in this study most likely reflect changes in the composition of the thin filament rather than in a cytosolic pool of TnT, composed of several isoform types available for rapid insertion into the thin filaments, for example, in response to a given physiological stimulus. In fact, since the myocardium was detergent skinned and washed before being placed in the SDS sample buffer, a hypothetical pool of unbound TnT would have been lost during membrane extraction, leaving behind only the isoforms contained in the thin filaments. Developmental changes in mammalian cardiac TnT isoform expression could explain the effects of maturation on myofibrillar ATPase activity described by others. Solaro et al 17 found that at ph 7.0, the myofibrillar ATPase activity was the same in the adult and the perinatal canine myocardium; on the other hand, at low ph, the ATPase activity was decreased in the adult but not in the perinatal myocardium. (A resistance to acidosis would be advantageous for survival during the perinatal period when episodes of acidosis are common.) Since myosin ATPase was the same at the two ages studied and TnC had the same properties, Solaro et al concluded that changes in the isoform expression of other thin filament regulatory proteins must occur to explain these differences. They have also found
5 746 Circulation Research Vol 63, No 4, October 1988 GROUP I 50 GROUP II I GROUP Mil a T11T2 TnTj TnT, mi- FIGURE 4. The proportions of troponin T isoforms TnT 2, TnT }, TnT 4, and TnT s found in rabbit left ventricular myocardium at the three stages of development: speckled bar, 3-day-old (n=8); open bar, 21- to 22-day-old (n=6); and striped bar, 99- to 109-day-old (n=5). The vertical lines represent two standard deviations about the mean. recently that the myofibrils from 4-day-old rabbit heart bind less calcium than myofibrils from the 22-day-old and adult heart. 18 PAGE analysis indicated that this maturational increase was not related to TnC, but was likely to be caused by differences in the distribution of TnT isoforms. 18 The developmental regulation of TnT isoform expression could account for the differences in myofibrillar ATPase and in myofibril calcium binding. The maturational increase in cardiac contractility 14 could be related to the changes in TnT isoform expression. These changes during mammalian development warrant studies that attempt to correlate the changes in the composition of myofilament proteins TnT. n <5 to the sensitivity of the myofilaments to calcium. If TnT expression changes in response to the maturational increase in myocardial workload, thus altering the physiological properties of the myofilaments, it is tempting to speculate that a change in TnT isoform expression due to a genetic lesion may cause certain forms of cardiomyopathy. 19 Acknowledgments The authors thank Elizabeth Fair, Annette Oakeley, and Elizabeth Smith for their help in acquiring and analyzing the data and in the preparation of the manuscript. The authors thank Dr. runa Pegoraro- rylawski for her advice and her preparation of the TALE 1. Proportions of Troponin T z, T 3, T 4, and T 5 Present in Left Ventricular Myocardium at the Three Stages of Development Troponin T 2 Troponin T 3 Troponin T 4 Troponin T 3 Group 1 [3 days old, n = 8] Group 2 [21-22 days old, n = 6] Group 3 [ days old, n=s] 42 ±4% 38±6% 25±3% 30±4% 27 ±4% 35 ±4% 23 ±2% 29±4% 33 ±4% 5±2% 8±2% 7±2%
6 Anderson et al Mammalian Left Ventricular Troponin T 747 standards from rabbit ventricles. The authors are grateful to Dr. R. John Solaro and to Pat Walker who provided the actin standard and helpful information. References 1. Ebashi S: Ca 2+ and the contractile proteins. J Mol Cell Cardiol 19S4;\6: Imai H, Hirai S, Hirono H, Hirabayashi T: Many isoforms of fast muscle troponin T from chicken legs. J iochem 1986; 99: Reiser PJ, Greaser ML, Moss RL: Tension/pCa characteristics and regulatory proteins of single fibers from chicken neonatal and adult fast and slow skeletal muscles (abstract). iophys J 1987;51:222a 4. riggs MM, Lin JJ-C, Schachat FH: The extent of aminoterminal heterogeneity in rabbit fast skeletal muscle troponin T. J Muscle Res Cell Motil 1987;8: reitbart RE, Nguyen HT, Medford RM, Destree AT, Mahdavi V, Nadal-Ginard : Intricate combinatorial patterns of exon splicing generate multiple regulated troponin T isoforms from a single gene. Cell 1985;41: Schachat FH, Diamond MS, randt PW: Effect of different troponin T-tropomyosin combinations on thin filament activation. J Mol iol 1987;198: Gusev N, arskaya NV, Verin AD, Duzhenkova IV, Khuchua ZA, Zheltova AO: Some properties of cardiac troponin T structure. iochem J 1983^13: Pearlstone JR, Carpenter MR, Smillie L: Amino-acid sequence of rabbit cardiac troponin T. J iol Chem 1986; 261: Tobacman LS, Lee R: Isolation and functional comparison of bovine cardiac troponin T isoforms. J iol Chem 1987; 262: Cooper TA, Ordahl CP: A single cardiac troponin T gene generates embryonic and adult isoforms via developmentally regulated alternate splicing. J iol Chem 1985^60: Laemmli UK: Cleavage of structural proteins during the assembly of the head of bacteriophage T<. Nature 1970; 227: Giulian GG, Moss RL, Greaser M: Improved methodology for analysis and quantitation of proteins on one-dimensional silver-stained slab gels. Anal iochem 1983;129: Potter JD: Preparation of troponin and its subunits, in Frederiksen DW, Cunningham LW (eds): Methods in Enzymology. New York, Academic Press, 1982, pp Nassar R, Reedy MC, Anderson PAW: Developmental changes in the ultrastructure and sarcomere shortening of the isolated rabbit ventricular myocyte. Circ Res 1987; 61: Nassar R, Moore GE, Anderson PAW: Troponin T isoform expression in single physiologically intact ventricular myocytes (abstract). iophys J 1987,51:329a 16. Pearlstone JR, Carpenter MR, Johnson P, Smillie L: Aminoacid sequence of tropomyosin-binding component of rabbit skeletal muscle troponin. Proc Natl Acad Sci USA 1976; 73: Solaro RJ, Kumar P, lanchard EM, Martin AF: Differential effects of ph on calcium activation of myofilaments of adult and perinatal dog hearts. Evidence for developmental differences in thin filament regulation. Circ Res 1986^8: McAuliffe JJ, Lieberman D, Solaro RJ: Ca 2+ binding to troponin C in rabbit cardiac myofibrils during development (abstract). iophys J 1988,53:583a 19. Allen PD, Anderson PAW: Changes in the expression of myocardial troponin T (TnT) isoforms in human heart failure (abstract). J Mol Cell Cardiol 1987;19(suppl IV):S.34 KEYWORDS: troponin T myofilaments calcium sensitivity
Force-pCa Relation and Troponin T Isoforms
 1470 Force-pCa Relation and Troponin T Isoforms of Rabbit Myocardium Rashid Nassar, Nadia N Malouf, Michael B Kelly, Annette E Oakeley, and Page AW Anderson We have previously reported the existence of
1470 Force-pCa Relation and Troponin T Isoforms of Rabbit Myocardium Rashid Nassar, Nadia N Malouf, Michael B Kelly, Annette E Oakeley, and Page AW Anderson We have previously reported the existence of
Thin Filament Changes during in Vivo Rat Heart Development
 003 1-399819 1/3003-0232$03.00/0 PEDIATRIC RESEARCH Copyright O 199 1 International Pediatric Research Foundation, Inc. Vol. 30, No. 3, 1991 Printed in (I. S. A. Thin Filament Changes during in Vivo Rat
003 1-399819 1/3003-0232$03.00/0 PEDIATRIC RESEARCH Copyright O 199 1 International Pediatric Research Foundation, Inc. Vol. 30, No. 3, 1991 Printed in (I. S. A. Thin Filament Changes during in Vivo Rat
SUPPLEMENTARY MATERIAL
 SUPPLEMENTARY MATERIAL Purification and biochemical properties of SDS-stable low molecular weight alkaline serine protease from Citrullus Colocynthis Muhammad Bashir Khan, 1,3 Hidayatullah khan, 2 Muhammad
SUPPLEMENTARY MATERIAL Purification and biochemical properties of SDS-stable low molecular weight alkaline serine protease from Citrullus Colocynthis Muhammad Bashir Khan, 1,3 Hidayatullah khan, 2 Muhammad
Calcium-Induced Fragmentation of Skeletal Muscle Nebulin Filaments'
 J. Biochem. 112, 775-779 (1992) Calcium-Induced Fragmentation of Skeletal Muscle Nebulin Filaments' Ryuichi Tatsumi and Koui Takahashi Meat Science Laboratory, Department of Animal Science, Faculty of
J. Biochem. 112, 775-779 (1992) Calcium-Induced Fragmentation of Skeletal Muscle Nebulin Filaments' Ryuichi Tatsumi and Koui Takahashi Meat Science Laboratory, Department of Animal Science, Faculty of
BIOCHEMICAL INVESTIGATIONS IN THE DIAGNOSTICS OF CARDIOVASCULAR DISORDERS. As. MARUSHCHAK M.I.
 BIOCHEMICAL INVESTIGATIONS IN THE DIAGNOSTICS OF CARDIOVASCULAR DISORDERS As. MARUSHCHAK M.I. Heart attack symptoms Acute MI Measurement of cardiac enzyme levels Measure cardiac enzyme levels at regular
BIOCHEMICAL INVESTIGATIONS IN THE DIAGNOSTICS OF CARDIOVASCULAR DISORDERS As. MARUSHCHAK M.I. Heart attack symptoms Acute MI Measurement of cardiac enzyme levels Measure cardiac enzyme levels at regular
Muscle Cells & Muscle Fiber Contractions. Packet #8
 Muscle Cells & Muscle Fiber Contractions Packet #8 Skeletal muscle is attached to bones and is responsible for movement. Introduction Introduction II Skeletal muscle is composed of bundles of muscle fibers
Muscle Cells & Muscle Fiber Contractions Packet #8 Skeletal muscle is attached to bones and is responsible for movement. Introduction Introduction II Skeletal muscle is composed of bundles of muscle fibers
BIOCHEMICAL EXAMINATION OF ACUTE MYOCARDIAL INFARCTION. Written by Lenka Fialová, translated by Jan Pláteník
 BIOCHEMICAL EXAMINATION OF ACUTE MYOCARDIAL INFARCTION 1 Structure of heart muscle Written by Lenka Fialová, translated by Jan Pláteník Heart muscle (myocardium) is a particular form of striated muscle,
BIOCHEMICAL EXAMINATION OF ACUTE MYOCARDIAL INFARCTION 1 Structure of heart muscle Written by Lenka Fialová, translated by Jan Pláteník Heart muscle (myocardium) is a particular form of striated muscle,
Troponin T isoforms alter the tolerance of transgenic mouse cardiac muscle to acidosis. Thomas M. Nosek, Marco A. Brotto 1, Jian-Ping Jin *,2
 ABB Archives of Biochemistry and Biophysics 430 (2004) 178 184 www.elsevier.com/locate/yabbi Troponin T isoforms alter the tolerance of transgenic mouse cardiac muscle to acidosis Thomas M. Nosek, Marco
ABB Archives of Biochemistry and Biophysics 430 (2004) 178 184 www.elsevier.com/locate/yabbi Troponin T isoforms alter the tolerance of transgenic mouse cardiac muscle to acidosis Thomas M. Nosek, Marco
Muscle Tissue. Xie Fenfen. Department of Histology and Embryology School of Basic Medicine Anhui Medical University
 Muscle Tissue Xie Fenfen Email:xff2005024@126.com Department of Histology and Embryology School of Basic Medicine Key points The structural differences (LM) of 3 types of muscle fibers Molecular structure
Muscle Tissue Xie Fenfen Email:xff2005024@126.com Department of Histology and Embryology School of Basic Medicine Key points The structural differences (LM) of 3 types of muscle fibers Molecular structure
Functional analysis of human cardiac troponin by the in vitro motility assay: comparison of adult, foetal and failing hearts
 Cardiovascular Research 43 (1999) 884 891 www.elsevier.com/ locate/ cardiores www.elsevier.nl/ locate/ cardiores Functional analysis of human cardiac troponin by the in vitro motility assay: comparison
Cardiovascular Research 43 (1999) 884 891 www.elsevier.com/ locate/ cardiores www.elsevier.nl/ locate/ cardiores Functional analysis of human cardiac troponin by the in vitro motility assay: comparison
Muscles and Animal Movement
 Muscles and Animal Movement Evolution of Muscle and Movement Animals are the only multicellular organisms that actively move. Movement is due to muscle cells (motor proteins) Muscle proteins have homologues
Muscles and Animal Movement Evolution of Muscle and Movement Animals are the only multicellular organisms that actively move. Movement is due to muscle cells (motor proteins) Muscle proteins have homologues
Chapter PURIFICATION OF ALKALINE PROTEASES
 Chapter PURIFICATION OF ALKALINE PROTEASES E /xtracellular alkaline proteases produced by Bacillus sp. K 25 and bacillus pumilus K 242, were purified and the homogeneity was examined by electrophoresis.
Chapter PURIFICATION OF ALKALINE PROTEASES E /xtracellular alkaline proteases produced by Bacillus sp. K 25 and bacillus pumilus K 242, were purified and the homogeneity was examined by electrophoresis.
SKELETAL MUSCLE CHARACTERISTICS
 THE MUSCULAR SYSTEM SKELETAL MUSCLE CHARACTERISTICS Most are attached by tendons to bones Cells are multinucleate Striated have visible banding Voluntary subject to conscious control Cells are surrounded
THE MUSCULAR SYSTEM SKELETAL MUSCLE CHARACTERISTICS Most are attached by tendons to bones Cells are multinucleate Striated have visible banding Voluntary subject to conscious control Cells are surrounded
Skeletal Muscle and the Molecular Basis of Contraction. Lanny Shulman, O.D., Ph.D. University of Houston College of Optometry
 Skeletal Muscle and the Molecular Basis of Contraction Lanny Shulman, O.D., Ph.D. University of Houston College of Optometry Like neurons, all muscle cells can be excited chemically, electrically, and
Skeletal Muscle and the Molecular Basis of Contraction Lanny Shulman, O.D., Ph.D. University of Houston College of Optometry Like neurons, all muscle cells can be excited chemically, electrically, and
Cardiac Troponin Testing and Chest Pain Patients: Exploring the Shades of Gray
 Cardiac Troponin Testing and Chest Pain Patients: Exploring the Shades of Gray Nichole Korpi-Steiner, PhD, DABCC, FACB University of North Carolina Chapel Hill, NC Learning Objectives Describe the acute
Cardiac Troponin Testing and Chest Pain Patients: Exploring the Shades of Gray Nichole Korpi-Steiner, PhD, DABCC, FACB University of North Carolina Chapel Hill, NC Learning Objectives Describe the acute
Mechanism of Muscular Contraction
 ~ Sorin2:er Jack A. Rail Mechanism of Muscular Contraction Contents 1 Setting the Stage: Myosin, Actin, Actomyosin and ATP... 1.1 Introduction... 1 1.2 Muscle Structure as Observed by Nineteenth Century
~ Sorin2:er Jack A. Rail Mechanism of Muscular Contraction Contents 1 Setting the Stage: Myosin, Actin, Actomyosin and ATP... 1.1 Introduction... 1 1.2 Muscle Structure as Observed by Nineteenth Century
Protein MultiColor Stable, Low Range
 Product Name: DynaMarker Protein MultiColor Stable, Low Range Code No: DM670L Lot No: ******* Size: 200 μl x 3 (DM670 x 3) (120 mini-gel lanes) Storage: 4 C Stability: 12 months at 4 C Storage Buffer:
Product Name: DynaMarker Protein MultiColor Stable, Low Range Code No: DM670L Lot No: ******* Size: 200 μl x 3 (DM670 x 3) (120 mini-gel lanes) Storage: 4 C Stability: 12 months at 4 C Storage Buffer:
CHAPTER 6 2/9/2016. Learning Objectives List the four traits that all muscle types have in common.
 Learning Objectives List the four traits that all muscle types have in common. CHAPTER 6 The Muscular System Demonstrate and explain the use of antagonistic muscle pairs. Describe the attachment of muscle
Learning Objectives List the four traits that all muscle types have in common. CHAPTER 6 The Muscular System Demonstrate and explain the use of antagonistic muscle pairs. Describe the attachment of muscle
Vmax from 1[37 to 3 94 ML/s. These results indicate that a much greater diversity. Laboratory. University of Wisconsin, Madison, WI 53706, U.S.A.
 Journal of Physiology (1988). 406. pp. 85-98 85 IJ'ith 6 text-fiyroes Pon1tedl in (bgealt B?rit(ain VARIATIONS IN CONTRACTILE PROPERTIES OF RABBIT SINGLE MUSCLE FIBRES IN RELATION TO TROPONIN T ISOFORMS
Journal of Physiology (1988). 406. pp. 85-98 85 IJ'ith 6 text-fiyroes Pon1tedl in (bgealt B?rit(ain VARIATIONS IN CONTRACTILE PROPERTIES OF RABBIT SINGLE MUSCLE FIBRES IN RELATION TO TROPONIN T ISOFORMS
Medical Biology. Dr. Khalida Ibrahim
 Dr. Khalida Ibrahim Medical Biology MUSCLE TISSUE 1. Muscle tissue is characterized by its well-developed properties of contraction. 2. Muscle is responsible for the movements of the body and the various
Dr. Khalida Ibrahim Medical Biology MUSCLE TISSUE 1. Muscle tissue is characterized by its well-developed properties of contraction. 2. Muscle is responsible for the movements of the body and the various
Microanatomy of Muscles. Anatomy & Physiology Class
 Microanatomy of Muscles Anatomy & Physiology Class Three Main Muscle Types Objectives: By the end of this presentation you will have the information to: 1. 2. 3. 4. 5. 6. Describe the 3 main types of muscles.
Microanatomy of Muscles Anatomy & Physiology Class Three Main Muscle Types Objectives: By the end of this presentation you will have the information to: 1. 2. 3. 4. 5. 6. Describe the 3 main types of muscles.
5. What component of the sarcomere is not attached to the Z line?
 Model 2: Anatomy of a Sarcomere 1. Label the thick filament and the thin filament in Model 2. 2. How many sarcomeres are shown in Model 2? 3. Using Model 2, based on the locations of thick and thin filaments,
Model 2: Anatomy of a Sarcomere 1. Label the thick filament and the thin filament in Model 2. 2. How many sarcomeres are shown in Model 2? 3. Using Model 2, based on the locations of thick and thin filaments,
Concept 50.5: The physical interaction of protein filaments is required for muscle function
 Concept 50.5: The physical interaction of protein filaments is required for muscle function Muscle activity is a response to input from the nervous system The action of a muscle is always to contract Vertebrate
Concept 50.5: The physical interaction of protein filaments is required for muscle function Muscle activity is a response to input from the nervous system The action of a muscle is always to contract Vertebrate
Chapter 7 The Muscular System. Mosby items and derived items 2012 by Mosby, Inc., an affiliate of Elsevier Inc. 1
 Chapter 7 The Muscular System Mosby items and derived items 2012 by Mosby, Inc., an affiliate of Elsevier Inc. 1 INTRODUCTION A. Muscular tissue enables the body and its parts to move 1. Three types of
Chapter 7 The Muscular System Mosby items and derived items 2012 by Mosby, Inc., an affiliate of Elsevier Inc. 1 INTRODUCTION A. Muscular tissue enables the body and its parts to move 1. Three types of
Outline. Bio 105: Muscular System. Muscular System. Types of Muscles. Smooth Muscle. Cardiac Muscle 4/6/2016
 Outline Bio 105: Muscular System Lecture 11 Chapter 6 Characteristics of muscles 3 types of muscles Functions of muscles Structure of skeletal muscles Mechanics of muscle contraction Energy sources for
Outline Bio 105: Muscular System Lecture 11 Chapter 6 Characteristics of muscles 3 types of muscles Functions of muscles Structure of skeletal muscles Mechanics of muscle contraction Energy sources for
Ch. 6: Contraction of Skeletal Muscle Physiological Anatomy of Skeletal Muscle
 Ch. 6: Contraction of Skeletal Muscle 40% skeletal muscle + 10% smooth and cardiac muscle Ch. 7: Excitation of Skeletal Muscle Ch. 9: Contraction and Excitation of Smooth Muscle Physiological Anatomy of
Ch. 6: Contraction of Skeletal Muscle 40% skeletal muscle + 10% smooth and cardiac muscle Ch. 7: Excitation of Skeletal Muscle Ch. 9: Contraction and Excitation of Smooth Muscle Physiological Anatomy of
Isolation and Functional Comparison of Bovine Cardiac Troponin T Isoforms*
 THE JOURNAL OF BIOLOGICAL CHEMISTRY Vol. 262, No. 9, Issue of March 25, pp. 4059-4064,1987 Printed in USA. Isolation and Functional Comparison of Bovine Cardiac Troponin T Isoforms* (Received for publication,
THE JOURNAL OF BIOLOGICAL CHEMISTRY Vol. 262, No. 9, Issue of March 25, pp. 4059-4064,1987 Printed in USA. Isolation and Functional Comparison of Bovine Cardiac Troponin T Isoforms* (Received for publication,
SCS MOLECULAR WEIGHT MARKERS 2,500-17,000 Caltons
 LECTROPHORES/S Revised November 1992 SCS MOLECULAR WEIGHT MARKERS 2,500-17,000 Caltons I NTRODUCTION Electrophoresis in polyacrylamide gels in the presence of sodium dodecyl sulfate (SDS), an anionic detergent,
LECTROPHORES/S Revised November 1992 SCS MOLECULAR WEIGHT MARKERS 2,500-17,000 Caltons I NTRODUCTION Electrophoresis in polyacrylamide gels in the presence of sodium dodecyl sulfate (SDS), an anionic detergent,
DOWNLOAD PDF STRUCTURE AND REGULATION OF CARDIAC AND SKELETAL MUSCLE THIN FILAMENTS
 Chapter 1 : ACTC1 - Wikipedia LARRY S. TOBACMAN STRUCTURE AND REGULATION OF CARDIAC AND The biological production of force and movement can be understood only when it is. Structure[ edit ] There are three
Chapter 1 : ACTC1 - Wikipedia LARRY S. TOBACMAN STRUCTURE AND REGULATION OF CARDIAC AND The biological production of force and movement can be understood only when it is. Structure[ edit ] There are three
Supplementary material: Materials and suppliers
 Supplementary material: Materials and suppliers Electrophoresis consumables including tris-glycine, acrylamide, SDS buffer and Coomassie Brilliant Blue G-2 dye (CBB) were purchased from Ameresco (Solon,
Supplementary material: Materials and suppliers Electrophoresis consumables including tris-glycine, acrylamide, SDS buffer and Coomassie Brilliant Blue G-2 dye (CBB) were purchased from Ameresco (Solon,
The organization of skeletal muscles. Excitation contraction coupling. Whole Skeletal Muscles contractions. Muscle Energetics
 Muscle and Movement The organization of skeletal muscles Excitation contraction coupling Whole Skeletal Muscles contractions Muscle Energetics The molecular bases of movement Muscular cells use molecular
Muscle and Movement The organization of skeletal muscles Excitation contraction coupling Whole Skeletal Muscles contractions Muscle Energetics The molecular bases of movement Muscular cells use molecular
Muscle Tissue- 3 Types
 AN INTRODUCTION TO MUSCLE TISSUE Muscle Tissue- 3 Types Skeletal muscle (focus on these) Cardiac muscle Smooth muscle FUNCTIONS OF SKELETAL MUSCLES Produce movement of the skeleton Maintain posture and
AN INTRODUCTION TO MUSCLE TISSUE Muscle Tissue- 3 Types Skeletal muscle (focus on these) Cardiac muscle Smooth muscle FUNCTIONS OF SKELETAL MUSCLES Produce movement of the skeleton Maintain posture and
ANSC/FSTC 607 Biochemistry and Physiology of Muscle as a Food PRIMARY, SECONDARY, AND TERTIARY MYOTUBES
 ANSC/FSTC 607 Biochemistry and Physiology of Muscle as a Food PRIMARY, SECONDARY, AND TERTIARY MYOTUBES I. Satellite Cells A. Proliferative, myoblastic cells that lie in invaginations in the sarcolemma
ANSC/FSTC 607 Biochemistry and Physiology of Muscle as a Food PRIMARY, SECONDARY, AND TERTIARY MYOTUBES I. Satellite Cells A. Proliferative, myoblastic cells that lie in invaginations in the sarcolemma
Muscles Muscles are effectors which enable movement to be carried out
 Muscles 13.8 Muscles are effectors which enable movement to be carried out Muscle Is responsible for almost all the movements in animals 3 types Cardiac muscle Smo oth muscle Skeletal mus cle (aka striped
Muscles 13.8 Muscles are effectors which enable movement to be carried out Muscle Is responsible for almost all the movements in animals 3 types Cardiac muscle Smo oth muscle Skeletal mus cle (aka striped
MUSCLE TISSUE (MUSCLE PHYSIOLOGY) PART I: MUSCLE STRUCTURE
 PART I: MUSCLE STRUCTURE Muscle Tissue A primary tissue type, divided into: skeletal muscle cardiac muscle smooth muscle Functions of Skeletal Muscles Produce skeletal movement Maintain body position Support
PART I: MUSCLE STRUCTURE Muscle Tissue A primary tissue type, divided into: skeletal muscle cardiac muscle smooth muscle Functions of Skeletal Muscles Produce skeletal movement Maintain body position Support
Chapter Skeletal Muscle Structure and Function
 Chapter 10.2 Skeletal Muscle Structure and Function Introduction to Muscle Physiology Movement is a fundamental characteristic of all living things All muscle cells (skeletal, cardiac, and smooth) are
Chapter 10.2 Skeletal Muscle Structure and Function Introduction to Muscle Physiology Movement is a fundamental characteristic of all living things All muscle cells (skeletal, cardiac, and smooth) are
Chapter 10 -Muscle Tissue
 Chapter 10 -Muscle Tissue Muscles: 1. Overview of Muscle Tissue A. Review 5 functions of muscle tissue. B. Review the 5 properties of muscle tissue. WHICH do they share with nervous tissue? (2, plus the
Chapter 10 -Muscle Tissue Muscles: 1. Overview of Muscle Tissue A. Review 5 functions of muscle tissue. B. Review the 5 properties of muscle tissue. WHICH do they share with nervous tissue? (2, plus the
Skeletal Muscle : Structure
 1 Skeletal Muscle : Structure Dr.Viral I. Champaneri, MD Assistant Professor Department of Physiology 2 Learning objectives 1. Gross anatomy of the skeletal muscle 2. Myofilaments & their molecular structure
1 Skeletal Muscle : Structure Dr.Viral I. Champaneri, MD Assistant Professor Department of Physiology 2 Learning objectives 1. Gross anatomy of the skeletal muscle 2. Myofilaments & their molecular structure
Skeletal muscle in the light of its structure
 Mechanism of contraction of Skeletal muscle in the light of its structure By Dr. Mudassar Ali Roomi (MBBS, M. Phil) Muscle Tissue Skeletal Muscle Cardiac Muscle Smooth Muscle Skeletal Muscle Long cylindrical
Mechanism of contraction of Skeletal muscle in the light of its structure By Dr. Mudassar Ali Roomi (MBBS, M. Phil) Muscle Tissue Skeletal Muscle Cardiac Muscle Smooth Muscle Skeletal Muscle Long cylindrical
Muscle Dr. Ted Milner (KIN 416)
 Muscle Dr. Ted Milner (KIN 416) Muscles are biological motors which actively generate force and produce movement through the process of contraction. The molecular mechanism responsible for muscle contraction
Muscle Dr. Ted Milner (KIN 416) Muscles are biological motors which actively generate force and produce movement through the process of contraction. The molecular mechanism responsible for muscle contraction
Physiology sheet #2. The heart composed of 3 layers that line its lumen and cover it from out side, these layers are :
 Physiology sheet #2 * We will talk in this lecture about cardiac muscle physiology, the mechanism and the energy sources of their contraction and intracellular calcium homeostasis. # Slide 4 : The heart
Physiology sheet #2 * We will talk in this lecture about cardiac muscle physiology, the mechanism and the energy sources of their contraction and intracellular calcium homeostasis. # Slide 4 : The heart
Isolation and Structural Characterization of Cap-Binding Proteins from Poliovirus-Infected HeLa Cells
 JOURNAL OF VIROLOGY, May 1985. p. 515-524 0022-538X/85/050515-10$02.00/0 Copyright C 1985, American Society for Microbiology Vol. 54, No. 2 Isolation and Structural Characterization of Cap-Binding Proteins
JOURNAL OF VIROLOGY, May 1985. p. 515-524 0022-538X/85/050515-10$02.00/0 Copyright C 1985, American Society for Microbiology Vol. 54, No. 2 Isolation and Structural Characterization of Cap-Binding Proteins
ANSC (FSTC) 607 Physiology and Biochemistry of Muscle as a Food MUSCLE CONTRACTION
 I. Basic model of muscle contraction A. Overall ANSC (FSTC) 607 Physiology and Biochemistry of Muscle as a Food MUSCLE CONTRACTION 1. Calcium is released from sarcoplasmic reticulum. 2. Myosin globular
I. Basic model of muscle contraction A. Overall ANSC (FSTC) 607 Physiology and Biochemistry of Muscle as a Food MUSCLE CONTRACTION 1. Calcium is released from sarcoplasmic reticulum. 2. Myosin globular
2, 6, 3, 7, 8, 9, 10, 5
 A novel regulatory element in cardiac muscle troponin T. Striated muscle contraction is regulated by the position of tropomyosin on actin. That position, in turn, is regulated by the troponin complex (troponin
A novel regulatory element in cardiac muscle troponin T. Striated muscle contraction is regulated by the position of tropomyosin on actin. That position, in turn, is regulated by the troponin complex (troponin
Although it is a cliché to speak of the need for heart to
 It Takes Heart to Win: What Makes the Heart Powerful? Kerry S. McDonald and Todd J. Herron Department of Physiology, University of Missouri, Columbia, Missouri 65212 The pumping action of the heart varies
It Takes Heart to Win: What Makes the Heart Powerful? Kerry S. McDonald and Todd J. Herron Department of Physiology, University of Missouri, Columbia, Missouri 65212 The pumping action of the heart varies
Mechanics of Cardiac Tissue
 Bioengineering 6000: System Physiology I Mechanics of Cardiac Tissue PD Dr.-Ing. Frank B. Sachse Overview Recapitulation Excitation Propagation II Force Development Experimental Studies Anatomy Cross Bridge
Bioengineering 6000: System Physiology I Mechanics of Cardiac Tissue PD Dr.-Ing. Frank B. Sachse Overview Recapitulation Excitation Propagation II Force Development Experimental Studies Anatomy Cross Bridge
Regulatory Proteins from Dorsal Muscle of the Carp
 J. Biochent. 82, 931-938 (1977) Regulatory Proteins from Dorsal Muscle of the Carp Kunihiko KONNO,* Ken-ichi ARAI,* and Shizuo WATANABE** *The Department of Food Science, Faculty of Fisheries, Hokkaido
J. Biochent. 82, 931-938 (1977) Regulatory Proteins from Dorsal Muscle of the Carp Kunihiko KONNO,* Ken-ichi ARAI,* and Shizuo WATANABE** *The Department of Food Science, Faculty of Fisheries, Hokkaido
Reconstitution of Neutral Amino Acid Transport From Partially Purified Membrane Components From Ehrlich Ascites Tumor Cells
 Journal of Supramolecular Structure 7:481-487 (1977) Molecular Aspects of Membrane Transport 5 1 1-5 17 Reconstitution of Neutral Amino Acid Transport From Partially Purified Membrane Components From Ehrlich
Journal of Supramolecular Structure 7:481-487 (1977) Molecular Aspects of Membrane Transport 5 1 1-5 17 Reconstitution of Neutral Amino Acid Transport From Partially Purified Membrane Components From Ehrlich
Page 1. Introduction Skeletal muscle cells have unique characteristics which allow for body movement.
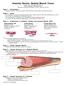 Anatomy Review: Skeletal Muscle Tissue Graphics are used with permission of: adam.com (http://www.adam.com/) Benjamin Cummings Publishing Co (http://www.awl.com/bc) Page 1. Introduction Skeletal muscle
Anatomy Review: Skeletal Muscle Tissue Graphics are used with permission of: adam.com (http://www.adam.com/) Benjamin Cummings Publishing Co (http://www.awl.com/bc) Page 1. Introduction Skeletal muscle
The diagram shows stained muscle fibres in a section taken from a muscle
 Q1.Slow and fast skeletal muscles both contain slow and fast muscle fibres but in different proportions. The proportion can be determined by observing stained sections of muscle under a microscope. The
Q1.Slow and fast skeletal muscles both contain slow and fast muscle fibres but in different proportions. The proportion can be determined by observing stained sections of muscle under a microscope. The
Troponin Mutation Caused Diastolic Dysfunction and Experimental Treatment in Transgenic Mice with Cardiomyopathy
 DOI 10.7603/s40782-014-0017-6 GSTF Journal of Advances in Medical Research (JAMR) Vol.1 No.2, October Troponin Mutation Caused Diastolic Dysfunction and Experimental Treatment in Transgenic Mice with Cardiomyopathy
DOI 10.7603/s40782-014-0017-6 GSTF Journal of Advances in Medical Research (JAMR) Vol.1 No.2, October Troponin Mutation Caused Diastolic Dysfunction and Experimental Treatment in Transgenic Mice with Cardiomyopathy
The origin of passive force enhancement in skeletal muscle
 Am J Physiol Cell Physiol 294: C74 C78, 2008. First published October 10, 2007; doi:10.1152/ajpcell.00218.2007. The origin of passive force enhancement in skeletal muscle V. Joumaa, 1 D. E. Rassier, 2
Am J Physiol Cell Physiol 294: C74 C78, 2008. First published October 10, 2007; doi:10.1152/ajpcell.00218.2007. The origin of passive force enhancement in skeletal muscle V. Joumaa, 1 D. E. Rassier, 2
Muscles & Motor Locomotion Why Do We Need All That ATP?
 Muscles & Motor Locomotion Why Do We Need All That ATP? 2006-2007 Animal Locomotion What are the advantages of locomotion? sessile motile Lots of ways to get around Lots of ways to get around mollusk mammal
Muscles & Motor Locomotion Why Do We Need All That ATP? 2006-2007 Animal Locomotion What are the advantages of locomotion? sessile motile Lots of ways to get around Lots of ways to get around mollusk mammal
Topics Covered. General muscle structure non-muscle components, macro-structure, contractile elements, membrane components.
 1 Topics Covered General muscle structure non-muscle components, macro-structure, contractile elements, membrane components. Contractile function Contractile and regulatory proteins, force production.
1 Topics Covered General muscle structure non-muscle components, macro-structure, contractile elements, membrane components. Contractile function Contractile and regulatory proteins, force production.
BIL 256 Cell and Molecular Biology Lab Spring, Tissue-Specific Isoenzymes
 BIL 256 Cell and Molecular Biology Lab Spring, 2007 Background Information Tissue-Specific Isoenzymes A. BIOCHEMISTRY The basic pattern of glucose oxidation is outlined in Figure 3-1. Glucose is split
BIL 256 Cell and Molecular Biology Lab Spring, 2007 Background Information Tissue-Specific Isoenzymes A. BIOCHEMISTRY The basic pattern of glucose oxidation is outlined in Figure 3-1. Glucose is split
Class XI Chapter 20 Locomotion and Movement Biology
 Question 1: Draw the diagram of a sarcomere of skeletal muscle showing different regions. The diagrammatic representation of a sarcomere is as follows: Question 2: Define sliding filament theory of muscle
Question 1: Draw the diagram of a sarcomere of skeletal muscle showing different regions. The diagrammatic representation of a sarcomere is as follows: Question 2: Define sliding filament theory of muscle
Lecture 9A. Muscle structure. Outline
 Lecture 9A Muscle structure Outline Smooth, skeletal, and cardiac muscle tissues Structure and function of skeletal muscle cells. Sarcomeres structure and contraction Actin-myosin interaction and sliding
Lecture 9A Muscle structure Outline Smooth, skeletal, and cardiac muscle tissues Structure and function of skeletal muscle cells. Sarcomeres structure and contraction Actin-myosin interaction and sliding
Animal Locomotion. Lots of ways to get around. What are the advantages of locomotion? Why Do We Need All That ATP?
 Animal Locomotion What are the advantages of locomotion? Motor Mechanisms sessile (Muscles & Motor Locomotion) motile Why Do We Need All That ATP? 2006-2007 mollusk mammal bird reptile We saw how some
Animal Locomotion What are the advantages of locomotion? Motor Mechanisms sessile (Muscles & Motor Locomotion) motile Why Do We Need All That ATP? 2006-2007 mollusk mammal bird reptile We saw how some
About This Chapter. Skeletal muscle Mechanics of body movement Smooth muscle Cardiac muscle Pearson Education, Inc.
 About This Chapter Skeletal muscle Mechanics of body movement Smooth muscle Cardiac muscle Skeletal Muscle Usually attached to bones by tendons Origin: closest to the trunk or to more stationary bone Insertion:
About This Chapter Skeletal muscle Mechanics of body movement Smooth muscle Cardiac muscle Skeletal Muscle Usually attached to bones by tendons Origin: closest to the trunk or to more stationary bone Insertion:
Protocol for Gene Transfection & Western Blotting
 The schedule and the manual of basic techniques for cell culture Advanced Protocol for Gene Transfection & Western Blotting Schedule Day 1 26/07/2008 Transfection Day 3 28/07/2008 Cell lysis Immunoprecipitation
The schedule and the manual of basic techniques for cell culture Advanced Protocol for Gene Transfection & Western Blotting Schedule Day 1 26/07/2008 Transfection Day 3 28/07/2008 Cell lysis Immunoprecipitation
Hole s Human Anatomy and Physiology Eleventh Edition. Mrs. Hummer. Chapter 9 Muscular System
 Hole s Human Anatomy and Physiology Eleventh Edition Mrs. Hummer Chapter 9 Muscular System 1 Chapter 9 Muscular System Skeletal Muscle usually attached to bones under conscious control striated Three Types
Hole s Human Anatomy and Physiology Eleventh Edition Mrs. Hummer Chapter 9 Muscular System 1 Chapter 9 Muscular System Skeletal Muscle usually attached to bones under conscious control striated Three Types
Connective tissue MUSCLE TISSUE
 Connective tissue MUSCLE TISSUE Part 1 General features of MT Develop from mesoderm Many cells, less intercellular matrix Function contraction (shortening) Skeletal (striated, voluntary) Types of MT Cardiac
Connective tissue MUSCLE TISSUE Part 1 General features of MT Develop from mesoderm Many cells, less intercellular matrix Function contraction (shortening) Skeletal (striated, voluntary) Types of MT Cardiac
Muscle Contraction and (Microscopic) Anatomy. Some copyright issues but I won t tell if you
 Muscle Contraction and (Microscopic) Anatomy Some copyright issues but I won t tell if you don t What is a contraction? It is so important to visualise the fact that muscles work their magic by simply
Muscle Contraction and (Microscopic) Anatomy Some copyright issues but I won t tell if you don t What is a contraction? It is so important to visualise the fact that muscles work their magic by simply
The Muscular System PART A
 6 The Muscular System PART A PowerPoint Lecture Slide Presentation by Jerry L. Cook, Sam Houston University ESSENTIALS OF HUMAN ANATOMY & PHYSIOLOGY EIGHTH EDITION ELAINE N. MARIEB The Muscular System
6 The Muscular System PART A PowerPoint Lecture Slide Presentation by Jerry L. Cook, Sam Houston University ESSENTIALS OF HUMAN ANATOMY & PHYSIOLOGY EIGHTH EDITION ELAINE N. MARIEB The Muscular System
Titin isoform variance and length dependence of activation in skinned bovine cardiac muscle
 J Physiol (2003), 553.1, pp. 147 154 DOI: 10.1113/jphysiol.2003.049759 The Physiological Society 2003 www.jphysiol.org Titin isoform variance and length dependence of activation in skinned bovine cardiac
J Physiol (2003), 553.1, pp. 147 154 DOI: 10.1113/jphysiol.2003.049759 The Physiological Society 2003 www.jphysiol.org Titin isoform variance and length dependence of activation in skinned bovine cardiac
Chapter 50. You re on your own for: Sensory Reception Mechanoreceptors Gravity, Hearing and Equilibrium. Chemoreception taste and smell
 1 Sensory and Motor Mechanisms 2 Chapter 50 You re on your own for: Sensory Reception Mechanoreceptors Gravity, Hearing and Equilibrium Chemoreception taste and smell Photoreceptors vision It s interesting.
1 Sensory and Motor Mechanisms 2 Chapter 50 You re on your own for: Sensory Reception Mechanoreceptors Gravity, Hearing and Equilibrium Chemoreception taste and smell Photoreceptors vision It s interesting.
HiPer Western Blotting Teaching Kit
 HiPer Western Blotting Teaching Kit Product Code: HTI009 Number of experiments that can be performed: 5/20 Duration of Experiment: ~ 2 days Day 1: 6-8 hours (SDS- PAGE and Electroblotting) Day 2: 3 hours
HiPer Western Blotting Teaching Kit Product Code: HTI009 Number of experiments that can be performed: 5/20 Duration of Experiment: ~ 2 days Day 1: 6-8 hours (SDS- PAGE and Electroblotting) Day 2: 3 hours
Skeletal Muscle Contraction and ATP Demand
 Skeletal Muscle Contraction and ATP Demand Anatomy & Structure Contraction Cycling Calcium Regulation Types of Contractions Force, Power, and Contraction Velocity Epimysium - separates fascia and muscle
Skeletal Muscle Contraction and ATP Demand Anatomy & Structure Contraction Cycling Calcium Regulation Types of Contractions Force, Power, and Contraction Velocity Epimysium - separates fascia and muscle
DISTRIBUTION OF ISOENZYMES OF THE GLYCOGENOLYTIC CASCADE IN DIFFERENT TYPES OF MUSCLE FIBRE
 Volume 67, number 1 FEBS LETTERS August 1976 DISTRIBUTION OF ISOENZYMES OF THE GLYCOGENOLYTIC CASCADE IN DIFFERENT TYPES OF MUSCLE FIBRE Ann BURCHELL, Patricia T. W. COHEN and Philip COHEN Department of
Volume 67, number 1 FEBS LETTERS August 1976 DISTRIBUTION OF ISOENZYMES OF THE GLYCOGENOLYTIC CASCADE IN DIFFERENT TYPES OF MUSCLE FIBRE Ann BURCHELL, Patricia T. W. COHEN and Philip COHEN Department of
Troponin-Tropomyosin Abnormalities in Hamster Cardiomyopathy
 Troponin-Tropomyosin Abnormalities in Hamster Cardiomyopathy Ashwani Malhotra and James Scheuer With the technical assistance of Kirit Patel and M. Cecilia Lopez Division of Cardiology, Department ofmedicine,
Troponin-Tropomyosin Abnormalities in Hamster Cardiomyopathy Ashwani Malhotra and James Scheuer With the technical assistance of Kirit Patel and M. Cecilia Lopez Division of Cardiology, Department ofmedicine,
CLASS SET Unit 4: The Muscular System STUDY GUIDE
 NPHS Anatomy & Physiology Questions to answer: 1) List three functions of the muscular system. 1) movement 2) thermogenesis (generates heat) 3) posture & body/joint support CLASS SET Unit 4: The Muscular
NPHS Anatomy & Physiology Questions to answer: 1) List three functions of the muscular system. 1) movement 2) thermogenesis (generates heat) 3) posture & body/joint support CLASS SET Unit 4: The Muscular
Elisabeth Huff Lonergan, PhD Steven M. Lonergan, PhD Mark J. Anderson, PhD
 Elisabeth Huff Lonergan, PhD Steven M. Lonergan, PhD Mark J. Anderson, PhD The complexity of tenderness. Differences in tenderness due to postmortem aging is difficult to predict and manage Structure is
Elisabeth Huff Lonergan, PhD Steven M. Lonergan, PhD Mark J. Anderson, PhD The complexity of tenderness. Differences in tenderness due to postmortem aging is difficult to predict and manage Structure is
MUSCULAR TISSUE. Dr. Gary Mumaugh
 MUSCULAR TISSUE Dr. Gary Mumaugh MUSCLE OVERVIEW The three types of muscle tissue are skeletal, cardiac, and smooth These types differ in structure, location, function, and means of activation FUNCTIONAL
MUSCULAR TISSUE Dr. Gary Mumaugh MUSCLE OVERVIEW The three types of muscle tissue are skeletal, cardiac, and smooth These types differ in structure, location, function, and means of activation FUNCTIONAL
BIOLOGY - CLUTCH CH.49 - MUSCLE SYSTEMS.
 !! www.clutchprep.com BIOLOGY - CLUTCH Muscle system organ system that includes skeletal, cardiac, and smooth muscle Muscle tissue capable of contracting through the interaction of actin and myosin proteins
!! www.clutchprep.com BIOLOGY - CLUTCH Muscle system organ system that includes skeletal, cardiac, and smooth muscle Muscle tissue capable of contracting through the interaction of actin and myosin proteins
Use of double- stranded DNA mini- circles to characterize the covalent topoisomerase- DNA complex
 SUPPLEMENTARY DATA Use of double- stranded DNA mini- circles to characterize the covalent topoisomerase- DNA complex Armêl Millet 1, François Strauss 1 and Emmanuelle Delagoutte 1 1 Structure et Instabilité
SUPPLEMENTARY DATA Use of double- stranded DNA mini- circles to characterize the covalent topoisomerase- DNA complex Armêl Millet 1, François Strauss 1 and Emmanuelle Delagoutte 1 1 Structure et Instabilité
OF LIGHT CHAINS OF CARDIAC MYOSIN ISOZYMES: ATRIAL AND VENTRICULAR MYOSINS
 CROSS-HYBRIDIZATION OF LIGHT CHAINS OF CARDIAC MYOSIN ISOZYMES: ATRIAL AND VENTRICULAR MYOSINS Gabor HOLLGSI*, Sudhir SRIVASTAVA** and Joan WIKMAN-COFFELT University of California, San Francisco Cardiovascular
CROSS-HYBRIDIZATION OF LIGHT CHAINS OF CARDIAC MYOSIN ISOZYMES: ATRIAL AND VENTRICULAR MYOSINS Gabor HOLLGSI*, Sudhir SRIVASTAVA** and Joan WIKMAN-COFFELT University of California, San Francisco Cardiovascular
POSTDOCTORAL TRAINING
 2 Curriculum Vitae JULIAN E. STELZER, PhD CONTACT DETAILS Phone: 216-368-8636 julian.stelzer@case.edu EDUCATION 1992-96 McGill University, Montréal, Canada BS, Physiology. 1996-98 University of Saskatchewan,
2 Curriculum Vitae JULIAN E. STELZER, PhD CONTACT DETAILS Phone: 216-368-8636 julian.stelzer@case.edu EDUCATION 1992-96 McGill University, Montréal, Canada BS, Physiology. 1996-98 University of Saskatchewan,
AP Biology. Animal Locomotion. Muscles & Motor Locomotion. Why Do We Need All That ATP? Lots of ways to get around. Muscle
 Muscles & Motor Locomotion Animal Locomotion What are the advantages of locomotion? sessile motile Why Do We Need All That? 2006-2007 Lots of ways to get around Lots of ways to get around mollusk mammal
Muscles & Motor Locomotion Animal Locomotion What are the advantages of locomotion? sessile motile Why Do We Need All That? 2006-2007 Lots of ways to get around Lots of ways to get around mollusk mammal
Product Information. Instruction for Use. Troponin I ELISA kit Catalog Number: EA Storage Temperature: 2 8 C
 9620 Medical Center Dr., Suite 200, Rockville, MD 20850 Phone: 1.888.267.4436 Fax: 301-340-9254 Email: techsupport@origene.com Web: www.origene.com Product Information Troponin I ELISA kit Catalog Number:
9620 Medical Center Dr., Suite 200, Rockville, MD 20850 Phone: 1.888.267.4436 Fax: 301-340-9254 Email: techsupport@origene.com Web: www.origene.com Product Information Troponin I ELISA kit Catalog Number:
Western Immunoblotting Preparation of Samples:
 Western Immunoblotting Preparation of Samples: Total Protein Extraction from Culture Cells: Take off the medium Wash culture with 1 x PBS 1 ml hot Cell-lysis Solution into T75 flask Scrap out the cells
Western Immunoblotting Preparation of Samples: Total Protein Extraction from Culture Cells: Take off the medium Wash culture with 1 x PBS 1 ml hot Cell-lysis Solution into T75 flask Scrap out the cells
The Sliding Filament Theory
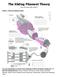 The Sliding Filament Theory Model 1: Muscle Histology Review How do muscle cells contract? Use your knowledge of muscle tissue histology to fill in the blanks numbered 1-11 with the following terms: Fasicle,
The Sliding Filament Theory Model 1: Muscle Histology Review How do muscle cells contract? Use your knowledge of muscle tissue histology to fill in the blanks numbered 1-11 with the following terms: Fasicle,
HPLC '88. Poster Presentation. Isolation of Thymosin B4 from Thymosin Fraction 5 by Reverse Phase HPLC
 Essentials in HPLC '88 Poster Presentation Isolation of Thymosin B4 from Thymosin Fraction 5 by Reverse Phase HPLC M. Badamchian, M.P. Strickler, M.J. Stone, A.L. Goldstein for Waters.bioresearchThe absolute,
Essentials in HPLC '88 Poster Presentation Isolation of Thymosin B4 from Thymosin Fraction 5 by Reverse Phase HPLC M. Badamchian, M.P. Strickler, M.J. Stone, A.L. Goldstein for Waters.bioresearchThe absolute,
The Muscular System. Muscle tissue is one of the 4 tissue types in vertebrates Muscle
 The Muscular System The Muscular System Muscle tissue is one of the 4 tissue types in vertebrates Muscle The Muscular System Muscle tissue is one of the 4 tissue types in vertebrates Muscle Nervous The
The Muscular System The Muscular System Muscle tissue is one of the 4 tissue types in vertebrates Muscle The Muscular System Muscle tissue is one of the 4 tissue types in vertebrates Muscle Nervous The
SERUM LIPOPROTEIN ESTIMATION BY POLYACRYL AMIDE GEL DISC ELECTROPHORESIS
 Nagoya J. med. Sci. 37: 23-27, 1974 SERUM LIPOPROTEIN ESTIMATION BY POLYACRYL AMIDE GEL DISC ELECTROPHORESIS FUMIHIKO OHYA, TAKAHIKO OHYA and assistant, Miss KAZUMI TODA 2nd Department of lnternal Medicine,
Nagoya J. med. Sci. 37: 23-27, 1974 SERUM LIPOPROTEIN ESTIMATION BY POLYACRYL AMIDE GEL DISC ELECTROPHORESIS FUMIHIKO OHYA, TAKAHIKO OHYA and assistant, Miss KAZUMI TODA 2nd Department of lnternal Medicine,
Smooth Cardiac Skeletal Location Around tubes Heart tissue attached to skeleton Moves stuff thru Heart beat pumps Moves body parts
 Biology 067 - Muscular system A. Type of muscles: Smooth Cardiac Skeletal Location Around tubes Heart tissue attached to skeleton Function Moves stuff thru Heart beat pumps Moves body parts tubes blood
Biology 067 - Muscular system A. Type of muscles: Smooth Cardiac Skeletal Location Around tubes Heart tissue attached to skeleton Function Moves stuff thru Heart beat pumps Moves body parts tubes blood
Muscular System. Human A & P
 Muscular System Human A & P There are 3 types of muscle tissue: A. Skeletal B. Smooth C. Cardiac The essential function of a muscle is contraction, or shortening, and are responsible for essentially all
Muscular System Human A & P There are 3 types of muscle tissue: A. Skeletal B. Smooth C. Cardiac The essential function of a muscle is contraction, or shortening, and are responsible for essentially all
Chapter 10 Muscle Tissue and Physiology Chapter Outline
 Chapter 10 Muscle Tissue and Physiology Chapter Outline Module 10.1 Overview of muscle tissue (Figures 10.1 10.2) A. Types of Muscle Tissue (Figure 10.1) 1. The three types of cells in muscle tissue are,,
Chapter 10 Muscle Tissue and Physiology Chapter Outline Module 10.1 Overview of muscle tissue (Figures 10.1 10.2) A. Types of Muscle Tissue (Figure 10.1) 1. The three types of cells in muscle tissue are,,
Challenging Issues in Cardiac Biomarker interpretation. F. Nikaeen. MD interventional Cardiologist
 Challenging Issues in Cardiac Biomarker interpretation F. Nikaeen. MD interventional Cardiologist Biomarkers Types of Troponin Troponin C Binds calcium Troponin I Binds actin Troponin T Binds tropomyosin
Challenging Issues in Cardiac Biomarker interpretation F. Nikaeen. MD interventional Cardiologist Biomarkers Types of Troponin Troponin C Binds calcium Troponin I Binds actin Troponin T Binds tropomyosin
Ch 12: Muscles sarcolemma, t-tubules, sarcoplasmic reticulum, myofibrils, myofilaments, sarcomere...
 Ch 12: Muscles Review micro-anatomy of muscle tissue Terminology examples: sarcolemma, t-tubules, sarcoplasmic reticulum, myofibrils, myofilaments, sarcomere... SLOs Differentiate levels of muscle structure:
Ch 12: Muscles Review micro-anatomy of muscle tissue Terminology examples: sarcolemma, t-tubules, sarcoplasmic reticulum, myofibrils, myofilaments, sarcomere... SLOs Differentiate levels of muscle structure:
Identification of NADPH-thioredoxin reductase system
 Proc. Nat. Acad. Sci. USA Vol. 72, No. 11, pp. 4233-4237, November 1975 Biochemistry Identification of NADPH-thioredoxin reductase system in Euglena gracilis* (ribonucleotide reduction) S. MUNAVALLIO,
Proc. Nat. Acad. Sci. USA Vol. 72, No. 11, pp. 4233-4237, November 1975 Biochemistry Identification of NADPH-thioredoxin reductase system in Euglena gracilis* (ribonucleotide reduction) S. MUNAVALLIO,
Biological motion. Motorproteins. Common features of motorproteins. Molecular mechanisms of biological motion. Structural homology
 Biological motion Molecular motion Molecular mechanisms of biological motion. Cellular motion Zsolt Mártonfalvi Bacterial flagellum Body motion Keratocyte moving on surface Motorproteins Mechanoenzymes
Biological motion Molecular motion Molecular mechanisms of biological motion. Cellular motion Zsolt Mártonfalvi Bacterial flagellum Body motion Keratocyte moving on surface Motorproteins Mechanoenzymes
Calcium regulation of troponin and its role in the dynamics of contraction and relaxation
 Page 1 of 11 1 Calcium regulation of troponin and its role in the dynamics of contraction and relaxation Robert Stehle, Bogdan Iorga, and Gabriele Pfitzer Institute of Vegetative Physiology, Faculty of
Page 1 of 11 1 Calcium regulation of troponin and its role in the dynamics of contraction and relaxation Robert Stehle, Bogdan Iorga, and Gabriele Pfitzer Institute of Vegetative Physiology, Faculty of
Calcium Regulation in Squid Mantle and Scallop Adductor Muscles 1
 . Biochem. 89, 581-589 (1981) Calcium Regulation in Squid Mantle and Scallop Adductor Muscles 1 Kunihiko KONNO,* Ken-ichi ARM,* Mikiharu YOSHIDA,** and Shizuo WATANABE*** Department of Food Science, Faculty
. Biochem. 89, 581-589 (1981) Calcium Regulation in Squid Mantle and Scallop Adductor Muscles 1 Kunihiko KONNO,* Ken-ichi ARM,* Mikiharu YOSHIDA,** and Shizuo WATANABE*** Department of Food Science, Faculty
#1 20. physiology. Muscle tissue 30/9/2015. Ahmad Adel Sallal. Mohammad Qudah
 # 20 physiology Muscle tissue Ahmad Adel Sallal 30/9/205 Mohammad Qudah MUSCLES PHYSIOLOGY Awn, welcome to the first physiology lecture in the MSS, I wish you a perfect exams with high grades, and never
# 20 physiology Muscle tissue Ahmad Adel Sallal 30/9/205 Mohammad Qudah MUSCLES PHYSIOLOGY Awn, welcome to the first physiology lecture in the MSS, I wish you a perfect exams with high grades, and never
Structure of the striated muscle general properties
 Structure of the striated muscle general properties Structure of the striated muscle membrane systems 1. Myofibrillum (contractile proteins) 2. Sarcoplasmic reticulum (SR) longitudinal tubule 3. SR terminal
Structure of the striated muscle general properties Structure of the striated muscle membrane systems 1. Myofibrillum (contractile proteins) 2. Sarcoplasmic reticulum (SR) longitudinal tubule 3. SR terminal
Examination Period 3: 2016/17
 Examination Period 3: 2016/17 Module Title Level Time Allowed Introduction to Physiology Four Two hours Instructions to students: Enter your student number not your name on all answer books. Answer all
Examination Period 3: 2016/17 Module Title Level Time Allowed Introduction to Physiology Four Two hours Instructions to students: Enter your student number not your name on all answer books. Answer all
Session 3-Part 2: Skeletal Muscle
 Session 3-Part 2: Skeletal Muscle Course: Introduction to Exercise Science-Level 2 (Exercise Physiology) Presentation Created by Ken Baldwin, M.ED, ACSM-H/FI Copyright EFS Inc. All Rights Reserved. Skeletal
Session 3-Part 2: Skeletal Muscle Course: Introduction to Exercise Science-Level 2 (Exercise Physiology) Presentation Created by Ken Baldwin, M.ED, ACSM-H/FI Copyright EFS Inc. All Rights Reserved. Skeletal
1. Locomotion. 2. Repositioning. 3. Internal movement
 MUSCLE and MOVEMENT Chapters 20, 8, 21 1. Locomotion A. Movement B. 2. Repositioning A. 3. Internal movement A. 1 Muscle Cells 1. Contractile 2. Myocytes 3. Striated A. Skeletal B. Cardiac 4. Smooth 5.
MUSCLE and MOVEMENT Chapters 20, 8, 21 1. Locomotion A. Movement B. 2. Repositioning A. 3. Internal movement A. 1 Muscle Cells 1. Contractile 2. Myocytes 3. Striated A. Skeletal B. Cardiac 4. Smooth 5.
1. Locomotion. 2. Repositioning. 3. Internal movement
 MUSCLE and MOVEMENT Chapters 20, 8, 21 1. Locomotion A. Movement B. 2. Repositioning A. 3. Internal movement A. Muscle Cells 1. Contractile 2. Myocytes 3. Striated A. Skeletal B. Cardiac 4. Smooth 5. Striated
MUSCLE and MOVEMENT Chapters 20, 8, 21 1. Locomotion A. Movement B. 2. Repositioning A. 3. Internal movement A. Muscle Cells 1. Contractile 2. Myocytes 3. Striated A. Skeletal B. Cardiac 4. Smooth 5. Striated
Supplementary Figures
 Supplementary Figures Supplementary Figure 1 Minicircle topoisomer generation. a, Generation of supercoiled topoisomers. Minicircles were nicked with the sequence-specific nicking endonuclease, Nb.BbvCI.
Supplementary Figures Supplementary Figure 1 Minicircle topoisomer generation. a, Generation of supercoiled topoisomers. Minicircles were nicked with the sequence-specific nicking endonuclease, Nb.BbvCI.
