Testicular interstitial fluid as a monitor for changes in the intratesticular environment in the rat
|
|
|
- Mark Joseph
- 5 years ago
- Views:
Transcription
1 Testicular interstitial fluid as a monitor for changes in the intratesticular environment in the rat R. M. Sharpe and Irene Cooper M.R.C. Reproductive Biology Unit, Centre for Reproductive Biology, 37 Chalmers Street, Edinburgh EH3 9EW, U.K. Summary. The use of testicular interstitial fluid (IF) collected from the rat testis has been validated as (1) an index of the total extracellular extratubular fluid volume of the testis (which reflects the permeability of testicular capillaries) and (2) as a means of measuring changes in the interstitial hormonal environment. The former was tested by comparing the albumin 'space' with the volume of recovered IF in the same testes from control and bilaterally cryptorchid rats sampled at 0\p=n-\40h after injection of hcg. Although the volume of IF recovered was on average only 50% of the albumin 'space', both measures increased in parallel after hcg injection and were always closely correlated (P < 0\m=.\001)over a 4- to 5-fold range. The volume of recovered IF increased with age in parallel with increase in testicular weight, and the testosterone concentration in IF paralleled changes in peripheral serum, increasing from 45 to 80 days of age and then declining. After injection of 25 \g=m\g bovine LH, testosterone levels in IF, spermatic venous (SV) and peripheral venous (PV) blood increased up to 10-fold by 1 h and returned to control levels over the next 11 h. Testosterone levels in IF were always considerably higher than those in SV blood, but this difference was not constant. Subcutaneous injection of rats with an LH-RH agonist resulted in parallel increases in the serum levels of LH and in the IF and PV levels of testosterone. However, at 6 h there was an 'LH-independent' secondary increase in testosterone levels which was associated with an increase in IF volume, reflecting an increase in capillary wall permeability and hence increased transport of LH into the testis. Introduction One of the major difficulties encountered in the study of male reproductive physiology is the measurement of intratesticular hormonal changes. To date, practically all in-vivo studies of testicular function have relied on measurements made in fluids draining from the testis (spermatic vein blood, lymph and rete testis fluid), although the composition of these fluids does not necessarily reflect the qualitative or quantitative composition of fluids within the testis. This is likely to be particularly true for hormones or factors which are produced and act within the testis, e.g. testosterone. In the rat testis there are large interstitial sinusoids filled with interstitial fluid, which bathes and separates the Leydig cells and seminiferous tubules (Fawcett, Neaves & Flores, 1973; Clark, 1976). This fluid is therefore likely to provide a reliable indication of intratesticular (extratubular) hormone concentrations. Testicular interstitial fluid can be collected in relatively large quantities ( µ /testis in an adult rat) by using extremely simple techniques (Sharpe, 1980,1981), and its /83/ $02-00/ Journals of Reproduction & Fertility Ltd
2 composition, including its testosterone concentration, has been reported previously (Sharpe, 1979). In addition, it has been shown that injection of rats with human chorionic gonadotrophin (hcg) results in a large increase in the volume of interstitial fluid within the testis (Sharpe, 1979, 1980, 1981), because of an increased permeability of testicular capillaries (Setchell & Sharpe, 1981). This change is a crucial factor in regulating the energy and uptake of hcg (LH) in the testis (Sharpe, 1981, 1983). The collection of testicular interstitial fluid is therefore of considerable potential value, both as an indicator of changes in capillary wall permeability (i.e. by measurement of interstitial fluid volume) and as a means of assessing changes in the interstitial hormonal environment of the testis. However, before this potential can be realized it has to be shown (1) that the measurement of testicular interstitial fluid volume provides a valid index of the total extracellular (extratubular) fluid volume, and (2) that changes in the hormone content of interstitial fluid can be shown to relate meaningfully to known physiological changes. The latter is particularly important as the collection of interstitial fluid takes place in vitro over a h period at 4 C, yet the hormonal content of the fluid is taken to reflect that existing in vivo at the time of removal of the testis. The present paper reports on the validation of these techniques. Materials and Methods Animals and treatments. Sprague-Dawley rats from our own colony were used for all experiments and were housed under conventional conditions. In two initial experiments the testicular albumin space (or volume of distribution) was compared with the volume of recovered testicular interstitial fluid in individual animals, the experiments being designed so that a wide range of values for these measures were obtained. In Exp. 1, normal intact rats aged 80 days were injected subcutaneously with 0-5 or 45 i.u. hcg (Chorulon: Intervet, Cambridge, U.K.) together with 125I-labelled bovine serum albumin (BSA; approx. 4 IO6 c.p.m.) in a volume of 0-2 ml 0-9% (w/v) NaCl containing 0-2% unlabelled BSA (fraction V : Sigma, St Louis, U.S.A.). Groups of 3 rats were killed at 8,16,24 or 40 h after injection. A further group of 6 rats served as non-hormone-treated controls and were injected subcutaneously with ' 25I-labelled BSA and killed 8 h later. Identical procedures were used in Exp. 2 but only the higher of the two doses of hcg, together with ' 25I-labelled BSA, was injected into normal intact male rats and into rats made bilaterally cryptorchid 5 weeks earlier at 80 days of age, as described previously (de Kretser, Sharpe & Swanston, 1979; Sharpe, 1983). Groups of 3 intact and 3 cryptorchid rats were then killed at 8, 16, 24 and 40 h after injection; control animals were treated as described in Exp. 1. Animals were killed with C02, blood was collected from the trunk and the paired testes were dissected out and subjected to the procedures described below. In Exp. 3, interstitial fluid collected from the testis by 2 different methods was compared. Rats aged 69 days were injected subcutaneously with 0-1 ml 0-9% (w/v) NaCl containing 0-2% BSA or with 1-5 µg ovine LH (NIAMDD-oLH-S23) in the same vehicle. Animals were killed 2 h later and their testes subjected to the procedures described below. Comparison of testosterone levels in interstitial fluid and serum. Three experiments were performed to assess the relationship between levels of testosterone in serum and interstitial fluid in different situations. In Exp. 4, groups of 5 untreated rats aged 25,45,60,80 or 100 days were killed, blood was collected for hormone analysis and the paired testes were dissected out and weighed individually before interstitial fluid was collected as described below. In Exp. 5, rats aged 75 days were injected subcutaneously with 25 µg bovine LH (NIH-LH-B4) in 0-1 ml 0-9% (w/v) NaCl containing 0-2% BSA. Groups of 3 rats were anaesthetized at intervals after this treatment by intraperitoneal injection of pentobarbitone sodium (Sagatal: May & Baker, Dagenham, U.K.), followed by an intracardiac injection of 100 i.u. heparin (Pularin; Duncan, Flockart & Co., London). An abdominal incision was then made and the right spermatic cord exposed and dissected sufficiently to permit a small piece of tinfoil to be inserted below the cord onto which
3 blood could collect. Spermatic vein blood was then collected by cutting either a dorsally descending branch of the spermatic vein (when this was present) or a superficial vein in the spermatic cord itself. Cuts were made with microdissection scissors and care was taken to ensure that the testis remained undisturbed in the scrotum throughout these procedures. When sufficient ( ml) spermatic vein blood had been collected, the animals were immediately killed, peripheral blood was collected from the trunk and the testes were dissected out and testicular interstitial fluid collected as described below. Six rats injected subcutaneously with saline (9 g NaCl/1) served as controls and these were killed in two groups at 2 and 40 h after injection and subjected to the above procedures. In Exp. 6, rats aged 55 days were injected subcutaneously with 5 ng LH-RH agonist (D-Ser-tbu6,des-Gly-NH210) LH-RH ethylamide; Hoechst, A.G., West Germany) in 0-5 ml 0-9% (w/v) NaCl containing 5 mg gelatin (Sigma)/ml and 2-5 mg BSA/ml. Groups of 4 such rats were killed at, 1,2,4,6 and 16 h after injection and trunk blood and testicular interstitial fluid were collected for hormone analysis. Four rats injected with the vehicle alone and killed 4 h after injection served as controls. Collection of testicular interstitial fluid. Interstitial fluid was collected from individual testes essentially as described previously (Sharpe, 1981,1983). Immediately after removal of the testis the caudal end of the testicular capsule was incised carefully and the testis was placed upright in an mm polystyrene tube such that the testis was suspended 1-2 cm above the tube bottom. Fluid was then allowed to percolate from the testis into the tube bottom over the next h at 4 C. The testis was then removed and the tubes centrifuged for 5 min at 1000 g to precipitate any contaminating erythrocytes and the interstitial fluid volume was measured by aspiration in 20 µ amounts. The fluid was then diluted with 10 volumes of 001 M-phosphate-buffered saline (ph 7-5) containing 0-2% BSA and stored at 20 C before assay of testosterone. In animals injected with 125I-labelled BSA the same procedures were followed except that before collection of interstitial fluid the radioactivity in each testis was determined by counting for 1 min in a gamma counter, and In the same the level of radioactivity in the recovered interstitial fluid was measured similarly. animals the radioactivity in 1-5 ml serum was also measured and ranged from 21 to 73 x 103 c.p.m./ml, depending on the time after injection. In Exp. 3, interstitial fluid prepared as described above was compared with fluid collected over a shorter time period. The right testis from each animal was processed as described above whilst the left testis was decapsulated carefully and placed in a 0-5 ml serum vial for 45 min at room temperature. The small amounts of fluid which had collected around the bottom of the testis were then aspirated and centrifuged for 5 min at \000g; the supernatant was aspirated in 5 µ amounts and processed as described for interstitial fluid. Measurement of the testicular albumin space. The albumin space within the testis provides a measure of the extratubular extracellular fluid volume (Setchell, 1978; Setehell & Sharpe, 1981) because albumin does not penetrate appreciably into the seminiferous tubules (Setchell, Hinton, Jacks & Davies, 1976). In the present studies the albumin space was determined by comparing the c.p.m. 125I-labelled BSA per testis with the c.p.m. 125I-labelled BSA per µ serum in individual animals ; division of the former by the latter yields the albumin space in µ per testis (Setchell & Sharpe, 1981). This measurement is only accurate when the levels of ' 25I-labelled BSA in serum and testicular extracellular fluid (i.e. interstitial fluid) have equilibrated and this takes 4-6 h after injection but is then maintained for 40 h or longer (Setchell & Sharpe, 1981 ; Sharpe, 1981, 1983). Therefore, in the present studies the albumin space was measured no earlier than 8 h after injection of ' 25I-labelled BSA, and confirmation that equilibration had occurred was obtained by comparing the concentration of labelled BSA in serum and testicular interstitial fluid in individual animals. The ratio of these values always fell within the range For each rat the albumin space and recovered interstitial fluid volume per left and right testis was measured, and the mean of these values was used in all subsequent calculations and comparisons. Radioiodination. BSA (fraction V, Sigma) was labelled with 125I using lactoperoxidase
4 (Miyachi, Vaitukaitis, Nieschlag & Lipsett, 1972) to a specific activity of between 43 and 62 µ /µ&. Hormone measurements. Serum levels of LH were measured by radioimmunoassay (Fraser & Sandow, 1977) using NIAMDD reagents, and results are expressed in terms of the LH-RP-1 standard. Testosterone concentrations in serum and interstitial fluid were measured by radio immunoassay (Corker & Davidson, 1978). Serum was extracted with 10 volumes of hexane : diethyl ether (4:1, v/v) before assay whilst interstitial fluid was assayed unextracted after appropriate dilution. It has been established previously (Sharpe, 1979) that unextracted and extracted interstitial fluid gave comparable testosterone values in the assay, and this was confirmed in the present study when samples from animals of different ages (Table 1) were assayed (1) direct, (2) after extraction and (3) after boiling for 10 min followed by extraction. The last method was performed to ensure that all protein-bound testosterone was freed for measurement in the RIA. Values obtained by each of the 3 methods showed strong mutual correlation (r > 0-99, 25, = < 0-001) and direct comparison using analysis of variance and paired / test revealed no significant (P > 0-1) difference between the methods. In addition to the above procedures, serum and interstitial fluid samples from the 25-day-old rats were extracted and assayed before and after alumina column chromatography (Furuyama, Mayes & Nugent, 1970) to remove interference from non-specific serum factors which occurs at this age. This Chromatographie step had no significant (P > 0-05) effect on the levels of testosterone measured in interstitial fluid, but more than halved (P < 0-01) the levels of testosterone measured in serum. Statistical analysis. The effects of treatment were analysed using 2-factor analysis of variance (with replication) and Student's t test, whilst the correlation between measurements of interstitial fluid volume (Exps 1 & 2) were assessed using the correlation coefficient. Comparison of testosterone concentrations in fluid from the left and right testes of individual animals (Exp. 3) was made using 2-factor analysis of variance. Results Comparison of the testicular albumin space and recovered interstitialfluid volume Experiment 1. In normal intact male rats, injection of a low dose (0-5 i.u.) of hcg caused a small but significant (P < 005) increase 8 h later in the albumin space and volume of recovered interstitial fluid, although at later times this difference was not evident (Text-fig. 1). In contrast, injection of a high dose (45 i.u.) of hcg caused a large (P < 0-001) and sustained (8-24 h) increase in the albumin space and interstitial fluid volume. Although the patterns of change in the fluid volume and albumin space in the testis were very similar, values obtained for the former were only 41-60% of values obtained for the albumin space in the same testis (Text-fig. 1). Experiment 2. The patterns of change in albumin space and recovered interstitial fluid volume of the testis in control and bilaterally cryptorchid rats over 40 h after injection of 45 i.u. hcg (Textfig. 1) were virtually identical, with values increasing significantly (P < 001) at 8-24 h in control rats and at h ( < 0-001) in bilaterally cryptorchid rats. As in Exp. 1, values obtained for the volume of recovered interstitial fluid from testes of controls rats were only 39-58% of the corre sponding albumin space. In bilaterally cryptorchid rats the recovered fluid volume more closely approximated (51-83%) the testicular albumin space and in these animals the albumin space and interstitial fluid volume were significantly (P < 0-001) greater than in testes from control rats at all times other than at 8 h after injection (Text-fig. 1). Although the recovered interstitial fluid volume was always less than the measured albumin space in testes from all animals in the 4 groups shown in Text-fig. 1, the values for these two measurements were always closely correlated (P < 0-001), both within each treatment group and overall in the two experiments (Text-fig. 2).
5 240-1 Alb. 100H () Hours Text-fig. 1. Comparison of the volume of recovered testicular interstitial fluid (IF; solid lines) with the measured testicular albumin space (broken lines) in (a) normal adult rats injected with 0-5 i.u. (, O) or 45 i.u. hcg (^, and (b) in normal adult rats (^, ) or adult rats made bilaterally cryptorchid 6 weeks previously (, ) both of which were injected with 45 i.u. hcg. Each point is the mean ± s.d. for 3 or 4 animals and full details are given in the text. 300 A* Recovered IF vol. (µ ) Text-fig. 2. Correlation between the volume of recovered testicular interstitial fluid (IF) and the measured testicular albumin space in individual animals. The data are derived from the experiments illustrated in Text-fig. 1.
6 Testosterone concentrations in interstitial fluid collected by two methods Testosterone levels in testicular fluid collected over 45 min from the decapsulated left testes of rats injected with saline or 1-5 µg LH were 368 ± 288 and 1083 ±111 ng/ml, respectively (mean ± s.d., = 4). The comparable values obtained for interstitial fluid collected over 16 h from the right testis using our standard procedure were 517 ± 176 and 1433 ± 234 ng/ml, respectively. Overall, the difference between values obtained by the two methods approached statistical significance (0-05 < < 0-2). Only very small quantities (5-10 µ ) of fluid could be collected from decapsulated testes and, because of the damage to some seminiferous tubules that this procedure inevitably causes, it is likely that interstitial fluid collected by decapsulation will have a higher contamination with seminiferous tubule/rete testis fluid than that collected by our standard procedure; this probably accounts for the difference in mean testosterone values obtained for fluid collected by the two methods. Age-related changes in the volume of interstitialfluid and its testosterone concentration With increase in age (Exp. 4), the recovered volume of interstitial fluid increased in parallel with testicular weight (Table 1). The mean concentration of testosterone in interstitial fluid decreased slightly from 25 to 45 days of age, then increased 3- to 4-fold to reach peak values at days of age before falling to lower values at 100 days of age. These changes were more or less paralleled by changes in the serum concentration of testosterone, although the ratio of the testosterone concentration in interstitial fluid to that in serum was at its highest at 25 days of age and then fell significantly (P < 0-05) by 45 and 60 days of age before rising again slightly (Table 1). Table 1. Age-related changes in testicular interstitial fluid (IF) volume and its testosterone concentration in relation to testicular weight and peripheral serum levels of testosterone (mean ± s.d., 5) = Days of age Testicular wt (mg) 326 ± ± ± ± IF vol. (µ /testis) 8 ± 4 43 ± ± ± ± 9 Testosterone cone, (ng/ml) IF 245 ± ± ± ± ± 61 Serum 1-6 ± ± ± ± ± 0-8 Ratio IF:serum 193 ± ±19 90 ± ± ± 37 Testosterone concentrations in interstitial fluid and serum after injection of LH In rats injected with 25 µg LH (Exp. 5), the levels of testosterone in interstitial fluid, spermatic venous blood and peripheral venous blood changed in a similar manner with time. Testosterone values rose 7- to 10-fold in the first hour after injection and then returned gradually to control levels over the next 11 h (Text-fig. 3). However, despite the obvious similarity in the pattern of response closer inspection revealed significant differences in the relative change in testosterone values in the 3 compartments. The ratio of the testosterone concentration in interstitial fluid to that in spermatic venous blood did not remain constant after LH injection, but decreased significantly (P < ) with respect to control values in the initial 4 h and then increased at 8-30 h after injection, this increase being relatively slight (P < 0-05) at 8-16 h but of considerable magnitude (P < 0001) at 20 and 30 h. At 20 and 30 h, testosterone levels in spermatic venous blood fell significantly (P < 0-02) below the level in control animals, whilst in the same rats testosterone concentrations in
7 LH F control SV control PV control -i Hours Text-fig. 3. Temporal changes in the level of testosterone in testicular interstitial fluid (IF), spermatic venous blood (SV) and peripheral venous blood (PV) after injection of adult rats with 25 µg LH. The testosterone concentrations (a) are shown on a logarithmic scale whereas the relative concentration of testosterone in IF to that in SV (b) is shown on an arithmetic scale. Each point is the mean ± s.d. for 3 animals except for the control values (broken lines) which are the mean ± s.d. for 6 animals. In (a), asterisks indicate where values fell significantly (P < 0-02) below control values; in (b) asterisks indicate where values were significantly (P < P < 0-001) lower or higher than the corresponding control values. interstitial fluid remained within the control range (Text-fig. 3). Similarly, when the ratio of the testosterone concentration in interstitial fluid to that in peripheral venous blood was compared, differences comparable to those illustrated for interstitial fluid/spermatic venous blood were observed whilst the spermatic/peripheral venous blood ratio of testosterone showed no significant (P > 0-05) variation with time after injection (data not shown). In this study, the recovered interstitial fluid volume was increased significantly (P < 0-001) at 4 h after injection of LH (102 ± 8 µ /testis, compared with 62 ± 8 µ /testis in controls; mean ± s.d.), returned to control levels briefly at 12 h, was increased again (P < 0-05) at h after injection (16 h: 79 ± 8 µ /testis; 20 h: 98 ± 29 µ /testis) and returned to control levels at h. Testosterone concentrations in interstitial fluid and serum after injection of LH-RH agonist Injection of 5 ng LH-RH agonist (Exp. 6) caused the expected increase in the serum concentrations of LH, which peaked at 2 h and returned to control levels by 6 h after injection. This
8 increase in LH was accompanied by a 10-fold increase in the values of testosterone in interstitial fluid and peripheral serum (Text-fig. 4). The change in testosterone levels followed the changes in serum LH levels up to 4 h after injection, but at 6 h there was a secondary increase (P < 0-001) in the concentrations of testosterone in interstitial fluid and serum and even by 16 h testosterone levels increase in were still raised significantly (P < 0-05) above control levels. This secondary testosterone values occurred in the absence of any increase in the serum concentrations of LH, but was associated with a significant (P < 0-01) increase in the testicular interstitial fluid volume, which was first evident at 6 h and persisted until 16 h after injection (Text-fig. 4). Text-fig. 4. Temporal changes in testicular interstitial fluid (IF) volume (hatched area) in relation to the serum levels of LH (a) and to the serum and IF levels of testosterone (b), after injection of adult rats with 5 ng LH-RH agonist. Each point is the mean ± s.d. for 4 rats. Discussion To reach their testicular targets, all blood-borne hormones and nutrients must first pass from the capillaries into testicular interstitial fluid, and this is also the medium by which the Leydig cells and seminiferous tubules communicate (Fawcett et al., 1973 ; Clark, 1976). The hormonal composition of interstitial fluid provides the most accurate indication of hormone levels surrounding the Leydig cells and seminiferous tubules, whilst the volume of interstitial fluid reflects changes in the permeability of testicular capillaries (Setchell & Sharpe, 1981). The present study provides strong
9 evidence that testicular interstitial fluid which is collected by allowing fluid to drain from the incised testis offers an extremely simple means of obtaining reliable information on both of these features. The method used for the collection of interstitial fluid is unlikely to result in 100% recovery of extracellular extratubular fluid within the testis, if only because the lymphatic sinusoids from which it must drain are discontinuous, convoluted and looped (Clark, 1976), and so pockets of interstitial fluid will remain. However, it is reasonable to expect that the volume offluid recovered will vary in proportion to the total volume of interstitial fluid, and this was assessed by comparing the total interstitial fluid volume (i.e. the albumin space) with the volume of recovered fluid. Albumin was used as a marker because it does not penetrate into the seminiferous tubules (Setchell et al., 1976) or into cells (see Setchell, 1978; Setchell, Laurie & Fritz, 1980), and it therefore provides an accurate assessment of the extracellular extratubular fluid volume within the testis (Setchell & Sharpe, 1981 ). When the testicular extracellular fluid volume was varied over a 4- to 5-fold range (Exps 1 & 2), the volume of recovered interstitial fluid provided an accurate reflection of changes in the total extra cellular fluid volume (albumin space), despite the fact that the actual values for these two measures differed by a factor of about 2. The estimation of the testicular albumin space confirmed that the recovered interstitial fluid represents the interstitial fluid which bathes the outside of the seminiferous tubules rather than the fluid present within the tubule lumen. Previous studies had demonstrated this on the grounds of the composition of interstitial fluid (Sharpe, 1979), but the present finding that the ratio of the concentration of 125I-labelled BSA in serum and interstitial fluid at equilibrium never fell below 0-93 reaffirms that contamination of the recovered interstitial fluid with seminiferous tubule fluid (which will contain negligible labelled albumin) was minimal. Contamination of interstitial fluid with serum from testicular blood must also be minimal in view of the minute total blood volume contained within the testis (Setchell & Sharpe, 1981). The results of Exps 3-6 demonstrate that the testosterone concentration in recovered testicular interstitial fluid probably provides a reliable indication of the testosterone concentration around the Leydig cells and seminiferous tubules. Testosterone concentrations in interstitial fluid were comparable to those reported by others who obtained interstitial fluid by micropuncture of the testis (Comhaire & Vermeulen, 1976), and the pattern of change in levels broadly paralleled those in the vasculature. In particular, the observation that changes in testosterone levels in interstitial fluid over 30- or 60-min periods closely followed those in spermatic venous or peripheral blood after stimulation with LH suggests that, despite the prolonged period over which the interstitial fluid was collected (16 h at 4 C), its testosterone concentration appeared to reflect levels at the time of death. reinforced by our finding that testosterone concentrations in testicular This conclusion is extracellular fluid obtained within 45 min of death were similar to those in interstitial fluid collected over 16 h from the contralateral testis. This study is the first to compare interstitial fluid and blood levels of testosterone and several aspects of the results are of physiological significance. The constantly higher levels of testosterone in interstitial fluid than those in spermatic venous blood indicate that testosterone values in the latter underestimate actual levels within the testis. Nor was there a constant difference or ratio between testosterone levels in interstitial fluid and spermatic venous blood (Text-fig. 3); when interstitial fluid levels of testosterone were very high, a relatively large proportion of this testosterone was secreted into the bloodstream but when interstitial fluid levels of testosterone were little testosterone was secreted into the near normal (i.e. similar to levels in control rats), relatively bloodstream. The latter finding may indicate the presence of a mechanism for conserving intra testicular testosterone concentrations, particularly because at h after LH injection blood levels of testosterone fell significantly below control levels whilst the interstitial fluid concentration of testosterone remained within the control range. Further evidence that the proportion of testicular testosterone secreted into the bloodstream is not constant was provided by the observation that the ratio of the testosterone concentration in interstitial fluid to that in serum was
10 highest in rats of 25 days of age and decreased significantly to lower levels at later ages. This change is perhaps related to the altered distribution of testicular capillaries which occurs during early puberty (Kormano, 1967). The importance of testicular interstitial fluid as a regulator of hormone transport into the testis (Sharpe, 1981, 1983) is also illustrated in the present studies. When LH-RH agonist was injected subcutaneously to stimulate LH and hence testosterone secretion (Exp. 6), rising serum levels of LH were paralleled by rising levels of testosterone in interstitial fluid and peripheral serum up to 2 h, but between 4 and 6 h, when serum levels of LH fell below those in control animals, testosterone values in both compartments showed a secondary increase (Text-fig. 4), a change almost identical to that in immature rats injected with LH-RH agonist (Sharpe & Fraser, 1980a). Two possible causes of this apparent 'LH-independent' increase in testosterone secretion are that it is due to (1) a direct effect of the LH-RH agonist on the Leydig cell, or (2) the associated increase in interstitial fluid volume (Text-fig. 4). Although the former option is consistent with the initial stimulatory effects of LH-RH agonist on Leydig cell testosterone secretion, the magnitude of the secondary increase in testosterone and its timing make this possibility less likely (see Sharpe, Doogan & Cooper, 1982; Sharpe & Harmar, 1983). In contrast, the increase in interstitial fluid volume between 4 and 6 h after injection of LH-RH agonist, which occurred presumably in response to the raised levels of LH (Setchell & Sharpe, 1981), would have increased the transport of LH from the bloodstream into interstitial fluid and hence to the Leydig cells (Sharpe, 1981, 1983). Therefore, although in this experiment the serum levels of LH remained at or below control levels between 6 and 16 h, the amount of LH reaching the Leydig cells would have been above normal due to the increase in capillary wall permeability. This re-emphasizes the potential importance of capillary wall permeability changes as a means of locally regulating gonadotrophin entry into the testis. This study has demonstrated that the collection of interstitial fluid from the rat testis can provide a reliable guide to the total volume of testicular interstitial fluid (and thus to the permeability of testicular capillaries) and to the composition of the fluid that normally surrounds the Leydig cells and seminiferous tubules. It provides a simple means of sampling the intra testicular environment and has obvious applications in the measurement of factors such as inhibin and androgen-binding protein, which are secreted within the testis, and hormones such as testosterone, oestradiol, 'testicular LH-RH' (Sharpe & Fraser, 1980b) and endorphins (Dong, Phillips, Halmi, Krieger & Bardin, 1982), all of which are believed to be of importance as intra testicular communicators or regulators. We thank Dr J. Sandow and Hoechst, A.G., for the gift of LH-RH agonist and NIAMDD, U.S.A., for the gift of bovine and ovine LH and materials for LH radioimmunoassay. References Clark, R.V. (1976) Three dimensional organization of testicular interstitial tissue and lymphatic space in the rat. Anat. Ree. 184, Comhaire, F.H. & Vermeulen, A. (1976) Testosterone concentration in the fluids of seminiferous tubules, the interstitium and the rete testis of the rat. J. Endocr. 70, Corker, C.S. & Davidson, D.W. (1978) Radioimmuno assay of testosterone in various biological fluids without chromatography. J. Steroid Biochem. 9, de Kretser, D.M., Sharpe, R.M. & Swanston, I.A. (1979) Alterations in steroidogenesis and hcg binding in the cryptorchid rat testis. Endocrinology 105, Dong, T.S., Phillips, D.M., Halmi, N., Krieger, D. & Bardin, C.W. (1982) ß-endorphin is present in the male reproductive tract of five species. Biol. Reprod. 27, Fawcett, D.W., Neaves, W.B. & Flores, M.N. (1973) Comparative observations on intertubular lympha tics and the organization of the interstitial tissue of the mammalian testis. Biol. Reprod. 9, Fraser, H.M. & Sandow, J. (1977) Gonadotrophin release by a highly active analogue of LHRH in rats immunized against LHRH. J. Endocr. 74, Furuyama, S., Mayes, D.M. & Nugent, C.A. (1970) A radioimmunoassay for plasma testosterone. Steroids 16, Kormano, M. (1967) An angiographie study of the testicular vasculature in the postnatal rat. Z. Anat. Entw. Gesch. 126,
11 Miyachi, Y., Vaitukaitis, J.L., Nieschlag, E. & Lipsett, M.B. (1972) Enzymatic radioiodination of gonadotropins. J. clin. Endocr. Metab. 34, Setchell, B.P. (1978) The Mammalian Testis, pp Paul Elek, London. Setchell, B.P. & Sharpe, R.M. (1981) The effect of injected human chorionic gonadotrophin on capillary permeability, extracellular fluid volume and the flow of lymph and blood in the testes of rats. J. Endocr. 91, Setchell, B.P., Hinton, B.T., Jacks, F. & Davies, R.V. (1976) The restricted penetration of iodinated rat FSH and LH into the seminiferous tubules of the rat testis. Molec. cell. Endocr. 6, Setchell, B.P., Laurie, M.S. & Fritz, LB. (1980) The development of the function of the blood testis barrier in rats and mice. In Testicular Development, Structure and Function, pp Eds E. Steinberger & A. Steinberger. Raven Press, New York. Sharpe, R.M. (1979) Gonadotrophin-induced accumula tion of 'interstitial fluid' in the rat testis. J. Reprod. Fert. 55, Sharpe, R.M. (1980) Temporal relationship between interstitial fluid accumulation and changes in gonadotropin receptor numbers and steroidogenesis in the rat testis. Biol. Reprod. 22, Sharpe, R.M. (1981) The importance of testicular interstitial fluid in the transport of injected hcg to the Leydig cells. Int. J. Androl. 4, Sharpe, R.M. (1983) Impaired gonadotrophin uptake in vivo by the cryptorchid rat testis. J. Reprod. Fert. 67, Sharpe, R.M. & Fraser, H.M. (1980a) Inhibition of maturational changes in Leydig cell function by treatment of rats with an agonist of LH-RH. J. Reprod. Fert. 60, Sharpe, R.M. & Fraser, H.M. (1980b) HCG stimulation of testicular LHRH-like activity. Nature, Lond. 287, Sharpe, R.M. & Harmar, A.J. (1983) The nature and biological actions of'testicular LHRH'. In Hormones and Cell Regulation, vol. 7, pp Eds J. E. Dumont, J. Nunez & R. M. Dentón. Elsevier Biomedicai Press, Amsterdam Sharpe, R.M., Doogan, D.G. & Cooper, I. (1982) Stimulation of Leydig cell testosterone secretion in vitro and in vivo in hypophysectomized rats by an agonist of luteinizing hormone releasing hormone. Biochem. biophys. Res. Commun. 106, Received 30 December 1982
(Received 9th January 1974)
 RELEASE OF LH AND FSH IN THE NORMAL INTACT RAM BY SYNTHETIC LH-RF AND THE EFFECT OF PRETREATMENT WITH TESTOSTERONE PROPIONATE C. R. N. HOPKINSON, H. C. PANT and R. J. FITZPATRICK Department of Veterinary
RELEASE OF LH AND FSH IN THE NORMAL INTACT RAM BY SYNTHETIC LH-RF AND THE EFFECT OF PRETREATMENT WITH TESTOSTERONE PROPIONATE C. R. N. HOPKINSON, H. C. PANT and R. J. FITZPATRICK Department of Veterinary
$ To whom all correspondence should be addressed. Anita H. Payne,+ Kar-Lit Wong, and Margarita M. Vega
 THE JOURNAL OF B~OLOCICAL CHEMISTRY Vul. 255. No. 15. Issue of August 10. pp. 7118-7122, 1980 Prmted cn (I S. A. Differential Effects of Single and Repeated Administrations of Gonadotropins on Luteinizing
THE JOURNAL OF B~OLOCICAL CHEMISTRY Vul. 255. No. 15. Issue of August 10. pp. 7118-7122, 1980 Prmted cn (I S. A. Differential Effects of Single and Repeated Administrations of Gonadotropins on Luteinizing
different ratios of PMSG and HCG on the occurrence of follicular haemorrhage THE induction of ovulation with PMSG and HCG in the rat has been studied
 Q. Jl exp. Physiol. (1968) 53, 129-135 THE INDUCTION OF OVULATION IN IMMATURE RATS TREATED WITH PREGNANT MARE'S SERUM GONADOTROPHIN AND HUMAN CHORIONIC GONADOTROPHIN. By S. F. LUNN and E. T. BELL. From
Q. Jl exp. Physiol. (1968) 53, 129-135 THE INDUCTION OF OVULATION IN IMMATURE RATS TREATED WITH PREGNANT MARE'S SERUM GONADOTROPHIN AND HUMAN CHORIONIC GONADOTROPHIN. By S. F. LUNN and E. T. BELL. From
PURIFICATION AND ACTION SITES OF A FOLLICLE STIMULATING HORMONE INHIBITOR FROM BOVINE FOLLICULAR FLUID t
 PURIFICATION AND ACTION SITES OF A FOLLICLE STIMULATING HORMONE INHIBITOR FROM BOVINE FOLLICULAR FLUID t E. Sato, T. Ishibashi and A. Iritani Kyoto university 2, Kyoto 606, Japan Summary The purification
PURIFICATION AND ACTION SITES OF A FOLLICLE STIMULATING HORMONE INHIBITOR FROM BOVINE FOLLICULAR FLUID t E. Sato, T. Ishibashi and A. Iritani Kyoto university 2, Kyoto 606, Japan Summary The purification
Effects of Growth Hormone on Testicular Testosterone Production In Vitro in Transgenic Mice
 Southern Illinois University Carbondale OpenSIUC Honors Theses University Honors Program 12-1989 Effects of Growth Hormone on Testicular Testosterone Production In Vitro in Transgenic Mice Angela Susan
Southern Illinois University Carbondale OpenSIUC Honors Theses University Honors Program 12-1989 Effects of Growth Hormone on Testicular Testosterone Production In Vitro in Transgenic Mice Angela Susan
Pande, Dasgupta & Kar, 1967). certainly no greater than in testicular venous blood, and may have been even less.
 Journal of PhysiOoloqy (1988). 407. pp. 363-372 363 W} ith I tpxt-fiyure 1'rinted in (;freat Britaini THE COMPOSITION OF EXTRACELLULAR INTERSTITIAL FLUID COLLECTED WITH A PUSH-PULL CANNULA FROM THE TESTES
Journal of PhysiOoloqy (1988). 407. pp. 363-372 363 W} ith I tpxt-fiyure 1'rinted in (;freat Britaini THE COMPOSITION OF EXTRACELLULAR INTERSTITIAL FLUID COLLECTED WITH A PUSH-PULL CANNULA FROM THE TESTES
Department of Obstetrics and Gynecology, School of Medicine, Chiba University, Chiba
 Endocrinol. Japon. 1971, 18 (6), 477 `485 Radioimmunoassays for Rat Follicle Stimulating and Luteinizing Hormones KATSUYOSHI SEKI, MITSUNORI SEKI, TERUFUMI YOSHIHARA AND HIDEYASU MAEDA Department of Obstetrics
Endocrinol. Japon. 1971, 18 (6), 477 `485 Radioimmunoassays for Rat Follicle Stimulating and Luteinizing Hormones KATSUYOSHI SEKI, MITSUNORI SEKI, TERUFUMI YOSHIHARA AND HIDEYASU MAEDA Department of Obstetrics
CHANGES IN LEVELS OF FOLLICLE STIMULATING HORMONE AND LUTEINIZING HORMONE IN THE BOVINE PITUITARY GLAND AT OVULATION
 CHANGES IN LEVELS OF FOLLICLE STIMULATING HORMONE AND LUTEINIZING HORMONE IN THE BOVINE PITUITARY GLAND AT OVULATION A. M. RAKHA and H. A. ROBERTSON The Division of Agricultural Biochemistry, Department
CHANGES IN LEVELS OF FOLLICLE STIMULATING HORMONE AND LUTEINIZING HORMONE IN THE BOVINE PITUITARY GLAND AT OVULATION A. M. RAKHA and H. A. ROBERTSON The Division of Agricultural Biochemistry, Department
MASANORI YAMAMOTO, HATSUKI HIBI and KOJI MIYAKE. Department of Urology, Nagoya University School of Medicine, Nagoya, Japan ABSTRACT
 Nagoya J. Med. Sci. 57. 133-142, 1994 EFFECT OF ALBUMIN ON PROLUMINAL MOVEMENT OF 3H-ANDROGEN INTO SEMINIFEROUS AND EPIDIDYMAL TUBULES AND ANDROGEN BINDING IN THE INTERSTITIUM OF THE TESTIS AND EPIDIDYMIS
Nagoya J. Med. Sci. 57. 133-142, 1994 EFFECT OF ALBUMIN ON PROLUMINAL MOVEMENT OF 3H-ANDROGEN INTO SEMINIFEROUS AND EPIDIDYMAL TUBULES AND ANDROGEN BINDING IN THE INTERSTITIUM OF THE TESTIS AND EPIDIDYMIS
Control of testicular vasomotion by testosterone and tubular factors in rats
 Control of testicular vasomotion by testosterone and tubular factors in rats O. Colin, A. Bergh, J-E. Damber and A. Widmark Departments of ^Anatomy, 2Pathology, and 3Urology and Andrology, 4Physiology,
Control of testicular vasomotion by testosterone and tubular factors in rats O. Colin, A. Bergh, J-E. Damber and A. Widmark Departments of ^Anatomy, 2Pathology, and 3Urology and Andrology, 4Physiology,
GARY S. KLEDZIK LIONEL CUSAN CLAUDE AUCLAIR, PH.D. PAUL A. KELLY, PH.D. FERNAND LABRIE, M.D., PH.D.*
 FERTILITY AND STERILITY Copyright 1978 The American Fertility Society Vol. 3, No.3, September 1978 Printed in U S.A. INHIBITION OF OVARIAN LUTEINIZING HORMONE (LH) AND FOLLICLE-STIMULATING HORMONE RECEPTOR
FERTILITY AND STERILITY Copyright 1978 The American Fertility Society Vol. 3, No.3, September 1978 Printed in U S.A. INHIBITION OF OVARIAN LUTEINIZING HORMONE (LH) AND FOLLICLE-STIMULATING HORMONE RECEPTOR
Effects and interactions of LH and LHRH agonist on testicular morphology and function in hypophysectomized rats
 Effects and interactions of LH and LHRH agonist on testicular morphology and function in hypophysectomized rats J. B. Kerr and R. M. Sharpe M.R.C. Reproductive Biology Unit, Centrefor Reproductive Biology,
Effects and interactions of LH and LHRH agonist on testicular morphology and function in hypophysectomized rats J. B. Kerr and R. M. Sharpe M.R.C. Reproductive Biology Unit, Centrefor Reproductive Biology,
Mechanisms of precocious puberty induced in male rats by
 Mechanisms of precocious puberty induced in male rats by pituitary grafts R. Aguilar, C. Bellido, J. E. S\l=a'\nchez-Criadoand E. Aguilar Department of Physiology, Faculty of Medicine, Cordoba University,
Mechanisms of precocious puberty induced in male rats by pituitary grafts R. Aguilar, C. Bellido, J. E. S\l=a'\nchez-Criadoand E. Aguilar Department of Physiology, Faculty of Medicine, Cordoba University,
Plasma levels of gonadotropin releasing hormone during menstrual cycle of Macaca radiata
 J. Biosci., Vol. 10, Number 4, December 1986, pp. 423-428. Printed in India. Plasma levels of gonadotropin releasing hormone during menstrual cycle of Macaca radiata N. MATHIALAGAN and A. JAGANNADHA RAO
J. Biosci., Vol. 10, Number 4, December 1986, pp. 423-428. Printed in India. Plasma levels of gonadotropin releasing hormone during menstrual cycle of Macaca radiata N. MATHIALAGAN and A. JAGANNADHA RAO
binding in cryptorchid rats
 Androgen secretion and characteristics of testicular hcg binding in cryptorchid rats B. D. Schanbacher Roman L. Hruska U.S. Meat Animal Research Center, Science and Education Administration- Agricultural
Androgen secretion and characteristics of testicular hcg binding in cryptorchid rats B. D. Schanbacher Roman L. Hruska U.S. Meat Animal Research Center, Science and Education Administration- Agricultural
ESUR SCROTAL AND PENILE IMAGING WORKING GROUP MULTIMODALITY IMAGING APPROACH TO SCROTAL AND PENILE PATHOLOGIES 2ND ESUR TEACHING COURSE
 ESUR SCROTAL AND PENILE IMAGING WORKING GROUP MULTIMODALITY IMAGING APPROACH TO SCROTAL AND PENILE PATHOLOGIES 2ND ESUR TEACHING COURSE NORMAL ANATOMY OF THE SCROTUM MICHAEL NOMIKOS M.D. F.E.B.U. UROLOGICAL
ESUR SCROTAL AND PENILE IMAGING WORKING GROUP MULTIMODALITY IMAGING APPROACH TO SCROTAL AND PENILE PATHOLOGIES 2ND ESUR TEACHING COURSE NORMAL ANATOMY OF THE SCROTUM MICHAEL NOMIKOS M.D. F.E.B.U. UROLOGICAL
INFLUENCE OF NEONATAL CASTRATION OR NEONATAL ANTI-GONADOTROPIN TREATMENT ON FERTILITY, PHALLUS DEVELOPMENT, AND MALE SEXUAL BEHAVIOR IN THE MOUSE*
 FERTILITY AND STERILITY Copyright 1975 The American Fertility Society Vol. 26, No.9. September 1975 Printed in U.SA. INFLUENCE OF NEONATAL CASTRATION OR NEONATAL ANTI-GONADOTROPIN TREATMENT ON FERTILITY,
FERTILITY AND STERILITY Copyright 1975 The American Fertility Society Vol. 26, No.9. September 1975 Printed in U.SA. INFLUENCE OF NEONATAL CASTRATION OR NEONATAL ANTI-GONADOTROPIN TREATMENT ON FERTILITY,
10.7 The Reproductive Hormones
 10.7 The Reproductive Hormones December 10, 2013. Website survey?? QUESTION: Who is more complicated: men or women? The Female Reproductive System ovaries: produce gametes (eggs) produce estrogen (steroid
10.7 The Reproductive Hormones December 10, 2013. Website survey?? QUESTION: Who is more complicated: men or women? The Female Reproductive System ovaries: produce gametes (eggs) produce estrogen (steroid
Male Reproductive Structures I. Overview A. Main functions: 1. Produce a haploid male gamete (sperm) 2. Deposit sperm in the female so fertilization
 Male Reproductive Structures I. Overview A. Main functions: 1. Produce a haploid male gamete (sperm) 2. Deposit sperm in the female so fertilization may occur! A. Scrotum 1. Muscular pouch that holds the
Male Reproductive Structures I. Overview A. Main functions: 1. Produce a haploid male gamete (sperm) 2. Deposit sperm in the female so fertilization may occur! A. Scrotum 1. Muscular pouch that holds the
Many studies have been published concerning the in vitro conversion of testosterone
 Testosterone gradient across the tubular wall F. H. COMHAIRE A. VERMEULEN Section of Endocrinology of the Department of Internal Medicine, State University, Akademisch Ziekenhuis, De Pintelaan, 135 B 9000
Testosterone gradient across the tubular wall F. H. COMHAIRE A. VERMEULEN Section of Endocrinology of the Department of Internal Medicine, State University, Akademisch Ziekenhuis, De Pintelaan, 135 B 9000
(Received 22 July 1957) It is now generally accepted that the unequal distribution of ions between cells
 190 J. Physiol. (I958) I40, I90-200 THE EFFECT OF ALTERATIONS OF PLASMA SODIUM ON THE SODIUM AND POTASSIUM CONTENT OF MUSCLE IN THE RAT By F. 0. DOSEKUN AND D. MENDEL From the Department of Physiology,
190 J. Physiol. (I958) I40, I90-200 THE EFFECT OF ALTERATIONS OF PLASMA SODIUM ON THE SODIUM AND POTASSIUM CONTENT OF MUSCLE IN THE RAT By F. 0. DOSEKUN AND D. MENDEL From the Department of Physiology,
Male Reproductive System
 21-1 21-2 Reproductive System Male Reproductive System Genital Tract In males the testes, held outside the body in the scrotum (optimum temp of about 35 0 C), produce sperm. Sperm mature in coiled tubes
21-1 21-2 Reproductive System Male Reproductive System Genital Tract In males the testes, held outside the body in the scrotum (optimum temp of about 35 0 C), produce sperm. Sperm mature in coiled tubes
(Haltmeyer & Eik-Neis, 1969; Katongole, Naftolin & Short, 1971). rutting behaviour and a rise in the male hierarchy (Lincoln, Youngson &
 THE RELATIONSHIP BETWEEN SOCIAL STATUS AND REPRODUCTIVE ACTIVITY IN MALE IMPALA, AEPYCEROS MELAMPUS P. S. BRAMLEY and W. B. NEAVES Departments of Animal Physiology and Anatomy, University of Nairobi, P.O.
THE RELATIONSHIP BETWEEN SOCIAL STATUS AND REPRODUCTIVE ACTIVITY IN MALE IMPALA, AEPYCEROS MELAMPUS P. S. BRAMLEY and W. B. NEAVES Departments of Animal Physiology and Anatomy, University of Nairobi, P.O.
A. K. EYONG AND V. B. BRAIDE
 GLOBAL JOURNAL OF MEDICAL SCIENCES VOL 8, NO. 1&2, 2009: 23-30 COPYRIGHT BACHUDO SCIENCE CO. LTD PRINTED IN NIGERIA. ISSN 1596-2911 www.globaljournalseries.com; Email: info@globaljournalseries.com EFFECTS
GLOBAL JOURNAL OF MEDICAL SCIENCES VOL 8, NO. 1&2, 2009: 23-30 COPYRIGHT BACHUDO SCIENCE CO. LTD PRINTED IN NIGERIA. ISSN 1596-2911 www.globaljournalseries.com; Email: info@globaljournalseries.com EFFECTS
androgens (including testosterone) has been demonstrated in Sertoli cells (Grootegoed Effects of FSH and testosterone on Sertoli cells
 Effects of FSH and testosterone on Sertoli cells and spermatocytes from rat testis F. F. G. ROMMERTS J. A. GROOTEGOED, H. J. VAN DER MOLEN Department of Biochemistry (Division of Chemical Endocrinology),
Effects of FSH and testosterone on Sertoli cells and spermatocytes from rat testis F. F. G. ROMMERTS J. A. GROOTEGOED, H. J. VAN DER MOLEN Department of Biochemistry (Division of Chemical Endocrinology),
Hormones of brain-testicular axis
 (Hormone Function) Hormones of brain-testicular axis anterior pituitary drives changes during puberty controlled by GnRH from hypothalamus begins to secrete FSH, LH LH targets interstitial endocrinocytes
(Hormone Function) Hormones of brain-testicular axis anterior pituitary drives changes during puberty controlled by GnRH from hypothalamus begins to secrete FSH, LH LH targets interstitial endocrinocytes
Testicular relapse in acute lymphoblastic leukaemia: Studies with an experimental mouse model
 Br. J. Cancer (1984), 49, 73-78 Testicular relapse in acute lymphoblastic leukaemia: Studies with an experimental mouse model H. Jackson, N.C. Jackson, M. Bock & M. Lendon' Department of Pharmacology,
Br. J. Cancer (1984), 49, 73-78 Testicular relapse in acute lymphoblastic leukaemia: Studies with an experimental mouse model H. Jackson, N.C. Jackson, M. Bock & M. Lendon' Department of Pharmacology,
Effect of cadmium chloride on the serum levels of follicle stimulating hormone (FSH), luteinizing hormone (LH) and androgens in the adult male rat
 Proc. Indian Acad. Sci., Vol. 87 B, (Experimental Biology-3), No.7, July 1978, pp. 161-167, @ printed in India Effect of cadmium chloride on the serum levels of follicle stimulating hormone (FSH), luteinizing
Proc. Indian Acad. Sci., Vol. 87 B, (Experimental Biology-3), No.7, July 1978, pp. 161-167, @ printed in India Effect of cadmium chloride on the serum levels of follicle stimulating hormone (FSH), luteinizing
Effect of a Single Injection of Human Chorionic Gonadotropin (hcg) on Testicular Hormones and Gonadotropins in the Thoroughbred Stallion
 NOTE Effect of a Single Injection of Human Chorionic Gonadotropin (hcg) on Testicular Hormones and Gonadotropins in the Thoroughbred Stallion Nobuo TSUNODA 1, Qiang WENG 2, 3, Shunichi NAGATA 4, Hiroyuki
NOTE Effect of a Single Injection of Human Chorionic Gonadotropin (hcg) on Testicular Hormones and Gonadotropins in the Thoroughbred Stallion Nobuo TSUNODA 1, Qiang WENG 2, 3, Shunichi NAGATA 4, Hiroyuki
The Effects of Selective Withdrawal of FSH or LH. on Spermatogenesis in the Immature Rat
 BIOLOGY OF REPRODUCTION 14,489-494(1976) The Effects of Selective Withdrawal of FSH or LH on Spermatogenesis in the Immature Rat H. G. MADHWA RAJ and MARTIN DYM Departments of Obstetrics and Gynaecology
BIOLOGY OF REPRODUCTION 14,489-494(1976) The Effects of Selective Withdrawal of FSH or LH on Spermatogenesis in the Immature Rat H. G. MADHWA RAJ and MARTIN DYM Departments of Obstetrics and Gynaecology
Cambridge CB2 3EG. ['25I]L-thyroxine. Experiments were performed after 24 hr had elapsed.
![Cambridge CB2 3EG. ['25I]L-thyroxine. Experiments were performed after 24 hr had elapsed. Cambridge CB2 3EG. ['25I]L-thyroxine. Experiments were performed after 24 hr had elapsed.](/thumbs/95/125304458.jpg) J. Physiol. (1971), 212, pp. 447-454 447 With 2 text-ftgurea Printed in Great Britain AN EXAMINATION OF THE EXTENT OF REVERSIBILITY OF THYROXINE BINDING WITHIN THE THYROXINE DISTRIBUTION SPACE IN THE RABBIT
J. Physiol. (1971), 212, pp. 447-454 447 With 2 text-ftgurea Printed in Great Britain AN EXAMINATION OF THE EXTENT OF REVERSIBILITY OF THYROXINE BINDING WITHIN THE THYROXINE DISTRIBUTION SPACE IN THE RABBIT
GARY S. KLEDZIK, PH.D. LIONEL CUSAN CLAUDE AUCLAIR, PH.D. PAUL A. KELLY, PH.D. FERNAND LABRIE, M.D., PH.D.*
 , FERTILITY AND STERILITY Copyright" 19 The American Fertility Society Vol. 29, No.5, May 19 Printed in U.S.A. INHIBITORY EFFECT OF A LUTEINIZING HORMONE (LH)-RELEASING HORMONE AGONIST ON RAT OVARIAN LH
, FERTILITY AND STERILITY Copyright" 19 The American Fertility Society Vol. 29, No.5, May 19 Printed in U.S.A. INHIBITORY EFFECT OF A LUTEINIZING HORMONE (LH)-RELEASING HORMONE AGONIST ON RAT OVARIAN LH
gonadotrophin-induced disruption of spermatogenesis
 Macrophage activation enhances the human chorionic gonadotrophin-induced disruption of spermatogenesis in the rat J. B. Kerr and R. M. Sharpe Department of Anatomy, Monash University, Clayton, Melbourne,
Macrophage activation enhances the human chorionic gonadotrophin-induced disruption of spermatogenesis in the rat J. B. Kerr and R. M. Sharpe Department of Anatomy, Monash University, Clayton, Melbourne,
12/3/12. Managing Bull Development to Optimize Fertility Rearing bulls for fertility
 Managing Bull Development to Optimize Fertility Rearing bulls for fertility Effect of post weaning nutrition (after normal calf hood nutrition) - testis size - age at puberty - semen quality Effect of
Managing Bull Development to Optimize Fertility Rearing bulls for fertility Effect of post weaning nutrition (after normal calf hood nutrition) - testis size - age at puberty - semen quality Effect of
Histology of Male Reproductive system (1)
 Histology of Male Reproductive system (1) Prof. Dr. Malak A. Al-yawer Learning Objectives At the end of this lecture, the medical student will be able to: State the organization of the testis Define seminiferous
Histology of Male Reproductive system (1) Prof. Dr. Malak A. Al-yawer Learning Objectives At the end of this lecture, the medical student will be able to: State the organization of the testis Define seminiferous
Gonadotropin Regulation of Rat Ovarian Lysosomes" Existence of a Hormone Specific Dual Control Mechanism
 Bit)science Reports, Vol. 8, No. 3, 1988 Gonadotropin Regulation of Rat Ovarian Lysosomes" Existence of a Hormone Specific Dual Control Mechanism N. Dhanasekaran 1,2 and N. R. Moudgal Received March 24,
Bit)science Reports, Vol. 8, No. 3, 1988 Gonadotropin Regulation of Rat Ovarian Lysosomes" Existence of a Hormone Specific Dual Control Mechanism N. Dhanasekaran 1,2 and N. R. Moudgal Received March 24,
Chapter 28: REPRODUCTIVE SYSTEM: MALE
 Chapter 28: REPRODUCTIVE SYSTEM: MALE I. FUNCTIONAL ANATOMY (Fig. 28.1) A. Testes: glands which produce male gametes, as well as glands producing testosterone 2. Seminiferous tubules (Fig.28.3; 28.5) a.
Chapter 28: REPRODUCTIVE SYSTEM: MALE I. FUNCTIONAL ANATOMY (Fig. 28.1) A. Testes: glands which produce male gametes, as well as glands producing testosterone 2. Seminiferous tubules (Fig.28.3; 28.5) a.
Male reproduction. Cross section of Human Testis ผศ.ดร.พญ.ส ว ฒณ ค ปต ว ฒ ภาคว ชาสร รว ทยา คณะแพทยศาสตร ศ ร ราชพยาบาล 1. Aims
 Aims Male reproduction Male reproductive structure Spermatogenesis ส ว ฒณ ค ปต ว ฒ ห อง 216 โทร: 7578 Hypothalamo-pituitary-testicular axis Male sex hormone action Male reproductive structure Male reproductive
Aims Male reproduction Male reproductive structure Spermatogenesis ส ว ฒณ ค ปต ว ฒ ห อง 216 โทร: 7578 Hypothalamo-pituitary-testicular axis Male sex hormone action Male reproductive structure Male reproductive
Fertility Diagnostics
 Fertility Diagnostics Fertility hormones measured on PATHFAST For internal use only Diagnostics PATHFAST Chemiluminescence-immuno-analyzer 1 Content: page 1. Fertility hormones - general aspects 1.1 Reproductive
Fertility Diagnostics Fertility hormones measured on PATHFAST For internal use only Diagnostics PATHFAST Chemiluminescence-immuno-analyzer 1 Content: page 1. Fertility hormones - general aspects 1.1 Reproductive
MALE REPRODUCTIVE SYSTEM
 MALE REPRODUCTIVE SYSTEM 1. The male reproductive system is made up of the following structures, EXCEPT: a. prostate; b. testicle; c. spermatic ducts; d. vestibular bulbs; e. seminal vesicles. 2.The testicle:
MALE REPRODUCTIVE SYSTEM 1. The male reproductive system is made up of the following structures, EXCEPT: a. prostate; b. testicle; c. spermatic ducts; d. vestibular bulbs; e. seminal vesicles. 2.The testicle:
Significance of carbohydrate residues in human chorionic gonadotropin (hcg) for expression of biological activity
 Pure & Appl. Chem., Vol. 62, No. 7, pp. 1427-1432,1990. Printed in Great Britain. @ 1990 IUPAC Significance of carbohydrate residues in human chorionic gonadotropin (hcg) for expression of biological activity
Pure & Appl. Chem., Vol. 62, No. 7, pp. 1427-1432,1990. Printed in Great Britain. @ 1990 IUPAC Significance of carbohydrate residues in human chorionic gonadotropin (hcg) for expression of biological activity
injected daily were {Received 7th August 1972) Summary. Groups of rats ovariectomized 5 weeks previously
 FFCTS OF XOGNOUS OSTRADIOL AND PROGSTRON ON SRUM LVLS OF PROSTAGLANDIN F AND LUTINIZING HORMON IN CHRONICALLY OVARICTOMIZD RATS S. K. SAKSNA, R. STL and M. J. K. HARPR Worcester Foundation for xperimental
FFCTS OF XOGNOUS OSTRADIOL AND PROGSTRON ON SRUM LVLS OF PROSTAGLANDIN F AND LUTINIZING HORMON IN CHRONICALLY OVARICTOMIZD RATS S. K. SAKSNA, R. STL and M. J. K. HARPR Worcester Foundation for xperimental
Changes in FSH and the pulsatile secretion of LH during the delay in oestrus induced by treatment of ewes
 Changes in FSH and the pulsatile secretion of LH during the delay in oestrus induced by treatment of ewes with bovine follicular fluid A. S. McNeilly M.R.C. Reproductive Biology Unit, University of Edinburgh
Changes in FSH and the pulsatile secretion of LH during the delay in oestrus induced by treatment of ewes with bovine follicular fluid A. S. McNeilly M.R.C. Reproductive Biology Unit, University of Edinburgh
Reproduction. AMH Anti-Müllerian Hormone. Analyte Information
 Reproduction AMH Anti-Müllerian Hormone Analyte Information - 1-2011-01-11 AMH Anti-Müllerian Hormone Introduction Anti-Müllerian Hormone (AMH) is a glycoprotein dimer composed of two 72 kda monomers 1.
Reproduction AMH Anti-Müllerian Hormone Analyte Information - 1-2011-01-11 AMH Anti-Müllerian Hormone Introduction Anti-Müllerian Hormone (AMH) is a glycoprotein dimer composed of two 72 kda monomers 1.
Male Reproductive System Dr. Gary Mumaugh
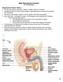 Male Reproductive System Dr. Gary Mumaugh Reproductive System Basics Primary sex organs (gonads) testes in males, ovaries in females Gonads produce sex cells called gametes (gametes means spouses) and
Male Reproductive System Dr. Gary Mumaugh Reproductive System Basics Primary sex organs (gonads) testes in males, ovaries in females Gonads produce sex cells called gametes (gametes means spouses) and
Leydig cell number and function in the adult cynomolgus
 Leydig cell number and function in the adult cynomolgus monkey (Macaca fascicularis) is increased by daily hcg treatment but not by daily FSH treatment K. J. Teerds, F. F. G. Rommerts, H. J. G. van de
Leydig cell number and function in the adult cynomolgus monkey (Macaca fascicularis) is increased by daily hcg treatment but not by daily FSH treatment K. J. Teerds, F. F. G. Rommerts, H. J. G. van de
Chapter 14 Reproduction Review Assignment
 Date: Mark: _/45 Chapter 14 Reproduction Review Assignment Multiple Choice Identify the choice that best completes the statement or answers the question. 1. Use the diagram above to answer the next question.
Date: Mark: _/45 Chapter 14 Reproduction Review Assignment Multiple Choice Identify the choice that best completes the statement or answers the question. 1. Use the diagram above to answer the next question.
The Third Department of Internal Medicine, University of Tokyo Faculty of Medicine, Hongo, Tokyo 113
 Endocrinol. Japon. 1974, 21 (2), 115 ` 119 A Radioimmunoassay for Serum Dehydroepiandrosterone HISAHIKO SEKIHARA, TOHRU YAMAJI, NAKAAKI OHSAWA AND HIROSHI IBAYASHI * The Third Department of Internal Medicine,
Endocrinol. Japon. 1974, 21 (2), 115 ` 119 A Radioimmunoassay for Serum Dehydroepiandrosterone HISAHIKO SEKIHARA, TOHRU YAMAJI, NAKAAKI OHSAWA AND HIROSHI IBAYASHI * The Third Department of Internal Medicine,
FOLLICLE-STIMULATING HORMONE CONTENT OF THE PITUITARY GLAND BEFORE IMPLANTATION IN THE MOUSE AND RAT
 FOLLICLE-STIMULATING HORMONE CONTENT OF THE PITUITARY GLAND BEFORE IMPLANTATION IN THE MOUSE AND RAT B. M. BINDON Department of Veterinary Physiology, University of Sydney, Sydney 2006, Australia (Received
FOLLICLE-STIMULATING HORMONE CONTENT OF THE PITUITARY GLAND BEFORE IMPLANTATION IN THE MOUSE AND RAT B. M. BINDON Department of Veterinary Physiology, University of Sydney, Sydney 2006, Australia (Received
(FITC) or rhodamine blue isothiocyanate (RBITC) for use in mixed egg-transfer experiments. Both FITC and RBITC bind to the zona pellucida
 THE LABELLING OF LIVING RABBIT OVA WITH FLUORESCENT DYES J. W. OVERSTREET Department of Anatomy and International Institute for the Study of Human Reproduction, Columbia University, College of Physicians
THE LABELLING OF LIVING RABBIT OVA WITH FLUORESCENT DYES J. W. OVERSTREET Department of Anatomy and International Institute for the Study of Human Reproduction, Columbia University, College of Physicians
(Received 22nd July 1974)
 THE VENOUS DRAINAGE OF THE ACCESSORY REPRODUCTIVE ORGANS OF THE RAT WITH SPECIAL REFERENCE TO PROSTATIC METABOLISM M. H. LEWIS and D. B. MOFFAT Anatomy Department, University College, Cardiff (Received
THE VENOUS DRAINAGE OF THE ACCESSORY REPRODUCTIVE ORGANS OF THE RAT WITH SPECIAL REFERENCE TO PROSTATIC METABOLISM M. H. LEWIS and D. B. MOFFAT Anatomy Department, University College, Cardiff (Received
Reproductive Endocrinology. Isabel Hwang Department of Physiology Faculty of Medicine University of Hong Kong Hong Kong May2007
 Reproductive Endocrinology Isabel Hwang Department of Physiology Faculty of Medicine University of Hong Kong Hong Kong May2007 isabelss@hkucc.hku.hk A 3-hormone chain of command controls reproduction with
Reproductive Endocrinology Isabel Hwang Department of Physiology Faculty of Medicine University of Hong Kong Hong Kong May2007 isabelss@hkucc.hku.hk A 3-hormone chain of command controls reproduction with
Cholecystokinin antagonist, proglumide, stimulates growth hormone release in the rat
 J. Biosci., Vol. 15, Number 1, March 1990, pp. 17 21. Printed in India. Cholecystokinin antagonist, proglumide, stimulates growth hormone release in the rat E. VIJAYAN* and S. M. McCANN Department of Physiology,
J. Biosci., Vol. 15, Number 1, March 1990, pp. 17 21. Printed in India. Cholecystokinin antagonist, proglumide, stimulates growth hormone release in the rat E. VIJAYAN* and S. M. McCANN Department of Physiology,
Long term follow-up study of 60 cases
 INTERNATIONAL JOURNALOFANDROLOGY 8 (1985) 177-185 Department of Endocrinology', Clinical Research Unit for the Male2, NataonalResearch Institute for Family Planning, Beijing and Depaltment of Urology3,
INTERNATIONAL JOURNALOFANDROLOGY 8 (1985) 177-185 Department of Endocrinology', Clinical Research Unit for the Male2, NataonalResearch Institute for Family Planning, Beijing and Depaltment of Urology3,
Pulsatile gonadotrophin releasing hormone versus gonadotrophin treatment of hypothalamic hypogonadism in males
 Human Reproduction vol.8 Suppl.2 pp. 175-179, 1993 Pulsatile gonadotrophin releasing hormone versus gonadotrophin treatment of hypothalamic hypogonadism in males Jochen Schopohl Medizinische Klinik, Klinikum
Human Reproduction vol.8 Suppl.2 pp. 175-179, 1993 Pulsatile gonadotrophin releasing hormone versus gonadotrophin treatment of hypothalamic hypogonadism in males Jochen Schopohl Medizinische Klinik, Klinikum
Epididymal proteins mimic the androgenic effect on
 Epididymal proteins mimic the androgenic effect on zona pellucida recognition by immature hamster Patricia S. Cuasnic\l=u'\,Fernanda Gonz\l=a'\lezEcheverr\l=i'\a,Alejandra Piazza, Lucrecia Pi\l=n~\eiroand
Epididymal proteins mimic the androgenic effect on zona pellucida recognition by immature hamster Patricia S. Cuasnic\l=u'\,Fernanda Gonz\l=a'\lezEcheverr\l=i'\a,Alejandra Piazza, Lucrecia Pi\l=n~\eiroand
THE EFFECTS OF LIGATION OF CAUDA EPIDIDYMIDIS ON THE DOG TESTIS
 Copyright 1974 The American Fertility Society FERTILITY AND STERILITY Vol. 25, No.3, March, 1974 Printed in U.S.A. THE EFFECTS OF LIGATION OF CAUDA EPIDIDYMIDIS ON THE DOG TESTIS A. M. VARE, M.B.B.S.,
Copyright 1974 The American Fertility Society FERTILITY AND STERILITY Vol. 25, No.3, March, 1974 Printed in U.S.A. THE EFFECTS OF LIGATION OF CAUDA EPIDIDYMIDIS ON THE DOG TESTIS A. M. VARE, M.B.B.S.,
Male Reproductive System. Dr Maan Al-Abbasi PhD, MSc, MBChB, MD
 Male Reproductive System Dr Maan Al-Abbasi PhD, MSc, MBChB, MD Learning Objectives 1. Describe the General Anatomy of the Male Reproductive System 2. Identify the structures that are related to the prostate.
Male Reproductive System Dr Maan Al-Abbasi PhD, MSc, MBChB, MD Learning Objectives 1. Describe the General Anatomy of the Male Reproductive System 2. Identify the structures that are related to the prostate.
Response to cryptorchidism of the testis and epididymis
 Response to cryptorchidism of the testis and epididymis of the opossum (Didelphis virginiana) Jane N. Scott, H. Ira Fritz and F. Nagy Departments ofanatomy and Biological Chemistry, Wright State University
Response to cryptorchidism of the testis and epididymis of the opossum (Didelphis virginiana) Jane N. Scott, H. Ira Fritz and F. Nagy Departments ofanatomy and Biological Chemistry, Wright State University
9.4 Regulating the Reproductive System
 9.4 Regulating the Reproductive System The Reproductive System to unite a single reproductive cell from a female with a single reproductive cell from a male Both male and female reproductive systems include
9.4 Regulating the Reproductive System The Reproductive System to unite a single reproductive cell from a female with a single reproductive cell from a male Both male and female reproductive systems include
HCG (human chorionic gonadotropin); Novarel Pregnyl (chorionic gonadotropin); Ovidrel (choriogonadotropin alfa)
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.08.09 Subject: HCG Page: 1 of 5 Last Review Date: June 19, 2015 HCG Powder, Novarel, Pregnyl, Ovidrel
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.08.09 Subject: HCG Page: 1 of 5 Last Review Date: June 19, 2015 HCG Powder, Novarel, Pregnyl, Ovidrel
Ovarian Function in the Preovulatory Rabbit.2
 BOLOGY OF REPRODUCTON 17, 34-38 (1977) Ovarian Function in the Preovulatory Rabbit.2 CHUNG H. WU, LUS BLASCO, GEORGE L. FLCKNGER and GEORGE MKHAL Endocrine Section, Department of Obstetrics and Gynecology,
BOLOGY OF REPRODUCTON 17, 34-38 (1977) Ovarian Function in the Preovulatory Rabbit.2 CHUNG H. WU, LUS BLASCO, GEORGE L. FLCKNGER and GEORGE MKHAL Endocrine Section, Department of Obstetrics and Gynecology,
might be due to a direct action on the thyroid, like that of the thiouracil
 288 J. Physiol. (1953) I20, 288-297 COMPARISON OF THE EFFECTS OF THIOURACIL, THY- ROXINE AND CORTISONE ON THE THYROID FUNCTION OF RABBITS BY N. B. MYANT* From the Department of Clinical Research, University
288 J. Physiol. (1953) I20, 288-297 COMPARISON OF THE EFFECTS OF THIOURACIL, THY- ROXINE AND CORTISONE ON THE THYROID FUNCTION OF RABBITS BY N. B. MYANT* From the Department of Clinical Research, University
Phlebotomy and Basic Blood Analysis
 Topic 2: Phlebotomy and Basic Blood Analysis Laboratory Manual Section 09 HPHE 6720 Dr. Cheatham Outline What is phlebotomy Review of Universal Precautions Phlebotomy Techniques Fingerprick Venipuncture
Topic 2: Phlebotomy and Basic Blood Analysis Laboratory Manual Section 09 HPHE 6720 Dr. Cheatham Outline What is phlebotomy Review of Universal Precautions Phlebotomy Techniques Fingerprick Venipuncture
Reproductive FSH. Analyte Information
 Reproductive FSH Analyte Information 1 Follicle-stimulating hormone Introduction Follicle-stimulating hormone (FSH, also known as follitropin) is a glycoprotein hormone secreted by the anterior pituitary
Reproductive FSH Analyte Information 1 Follicle-stimulating hormone Introduction Follicle-stimulating hormone (FSH, also known as follitropin) is a glycoprotein hormone secreted by the anterior pituitary
Model Answer. M.Sc. Zoology (First Semester) Examination Paper LZT 103 (Endocrinology)
 Model Answer M.Sc. Zoology (First Semester) Examination-2013 Paper LZT 103 (Endocrinology) Section A 1. (i) d (ii) b (iii) b (iv) c (v) c (vi) a (vii) c (viii) a (ix) d (x) b Section B Q.2 Answer Hormonal
Model Answer M.Sc. Zoology (First Semester) Examination-2013 Paper LZT 103 (Endocrinology) Section A 1. (i) d (ii) b (iii) b (iv) c (v) c (vi) a (vii) c (viii) a (ix) d (x) b Section B Q.2 Answer Hormonal
Motility and eosin uptake of formaldehyde-treated ram
 Motility and eosin uptake of formaldehyde-treated ram spermatozoa O. A. Osinowo, J. O. Bale, E. O. Oyedipe and L. O. Eduvie Department ofanimal Reproduction, National Animal Production Research Institute,
Motility and eosin uptake of formaldehyde-treated ram spermatozoa O. A. Osinowo, J. O. Bale, E. O. Oyedipe and L. O. Eduvie Department ofanimal Reproduction, National Animal Production Research Institute,
LIE ASSAY OF GONADOTROPIN in human blood is one of the most important
 Changes in Human Serum FSH Levels During the Normal Menstrual Cycle MASAO IGARASHI, M.D., JUNJI KAMIOKA, M.D., YOICHI EHARA, M.D., and SEIICHI MATSUMOTO, M.D. LIE ASSAY OF GONADOTROPIN in human blood is
Changes in Human Serum FSH Levels During the Normal Menstrual Cycle MASAO IGARASHI, M.D., JUNJI KAMIOKA, M.D., YOICHI EHARA, M.D., and SEIICHI MATSUMOTO, M.D. LIE ASSAY OF GONADOTROPIN in human blood is
Growth pattern of the sex ducts in foetal mouse hermaphrodites
 /. Embryol. exp. Morph. 73, 59-68, 1983 59 Printed in Great Britain The Company of Biologists Limited 1983 Growth pattern of the sex ducts in foetal mouse hermaphrodites By C. YDING ANDERSEN 1, A. G. BYSKOV
/. Embryol. exp. Morph. 73, 59-68, 1983 59 Printed in Great Britain The Company of Biologists Limited 1983 Growth pattern of the sex ducts in foetal mouse hermaphrodites By C. YDING ANDERSEN 1, A. G. BYSKOV
Male reproductive system The physiology of sexual act
 Male reproductive system The physiology of sexual act Gabriella Kékesi 65. The development and physiology of the male reproductive system. The physiology of the sexual act Define chromosomal, gonadal and
Male reproductive system The physiology of sexual act Gabriella Kékesi 65. The development and physiology of the male reproductive system. The physiology of the sexual act Define chromosomal, gonadal and
Leydig cell hypofunction resulting in male pseudohermaphroditism*
 FERmlTY AND STERILlTY Copyright" 1982 The American Fertility Society Vol. 37, No.6, May 1982 Printed in U.S.A. Leydig cell hypofunction resulting in male pseudohermaphroditism* Peter A. Lee, M.D., Ph.D.t+1I
FERmlTY AND STERILlTY Copyright" 1982 The American Fertility Society Vol. 37, No.6, May 1982 Printed in U.S.A. Leydig cell hypofunction resulting in male pseudohermaphroditism* Peter A. Lee, M.D., Ph.D.t+1I
C-Peptide I and II (Rat) ELISA
 C-Peptide I and II (Rat) ELISA For the quantitative determination of rat C-peptide in plasma, serum, urine, and cell culture supernatant Please read carefully due to Critical Changes, e.g., blot plate
C-Peptide I and II (Rat) ELISA For the quantitative determination of rat C-peptide in plasma, serum, urine, and cell culture supernatant Please read carefully due to Critical Changes, e.g., blot plate
DAX1, testes development role 7, 8 DFFRY, spermatogenesis role 49 DMRT genes, male sex differentiation role 15
 Subject Index N-Acetylcysteine, sperm quality effects 71 Ambiguous genitalia, origins 1, 2 Anti-Müllerian hormone function 13 receptors 13 Sertoli cell secretion 10, 38 Apoptosis assays in testes 73, 74
Subject Index N-Acetylcysteine, sperm quality effects 71 Ambiguous genitalia, origins 1, 2 Anti-Müllerian hormone function 13 receptors 13 Sertoli cell secretion 10, 38 Apoptosis assays in testes 73, 74
ANDROGEN BIOSYNTHESIS IN EXPERIMENTAL CRYPTORCHIDISM*
 FERTILITY AND STERILITY Copyright ~ 1975 The American Fertility Society Vol. 26, No.7, July 1975 Printed in U.S A. ANDROGEN BIOSYNTHESIS IN EXPERIMENTAL CRYPTORCHIDISM* JUAN CARLOS HOSCHOIAN, PH.D., AND
FERTILITY AND STERILITY Copyright ~ 1975 The American Fertility Society Vol. 26, No.7, July 1975 Printed in U.S A. ANDROGEN BIOSYNTHESIS IN EXPERIMENTAL CRYPTORCHIDISM* JUAN CARLOS HOSCHOIAN, PH.D., AND
Effect of Resistin on Granulosa and Theca Cell Function in Cattle
 1 Effect of Resistin on Granulosa and Theca Cell Function in Cattle D.V. Lagaly, P.Y. Aad, L.B. Hulsey, J.A. Grado-Ahuir and L.J. Spicer Story in Brief Resistin is an adipokine that has not been extensively
1 Effect of Resistin on Granulosa and Theca Cell Function in Cattle D.V. Lagaly, P.Y. Aad, L.B. Hulsey, J.A. Grado-Ahuir and L.J. Spicer Story in Brief Resistin is an adipokine that has not been extensively
Outline. Male Reproductive System Testes and Sperm Hormonal Regulation
 Outline Male Reproductive System Testes and Sperm Hormonal Regulation Female Reproductive System Genital Tract Hormonal Levels Uterine Cycle Fertilization and Pregnancy Control of Reproduction Infertility
Outline Male Reproductive System Testes and Sperm Hormonal Regulation Female Reproductive System Genital Tract Hormonal Levels Uterine Cycle Fertilization and Pregnancy Control of Reproduction Infertility
Primary sex organs (gonads): testes and ovaries. Accessory reproductive organs: ducts, glands, and external genitalia
 Male Reproductive System Primary sex organs (gonads): testes and ovaries Produce sex cells (gametes) Secrete steroid sex hormones Androgens (males) Estrogens and progesterone (females) Accessory reproductive
Male Reproductive System Primary sex organs (gonads): testes and ovaries Produce sex cells (gametes) Secrete steroid sex hormones Androgens (males) Estrogens and progesterone (females) Accessory reproductive
Male Anatomy. testes, genetically determined in mammals - testis releases hormones that then control the development of secondary sex characteristics
 Male Anatomy Male Anatomy Primary Organ testes, genetically determined in mammals - testis releases hormones that then control the development of secondary sex characteristics 1) Secondary Organs internal
Male Anatomy Male Anatomy Primary Organ testes, genetically determined in mammals - testis releases hormones that then control the development of secondary sex characteristics 1) Secondary Organs internal
Hormonal Control of Male Sexual Function
 Hormonal Control of Male Sexual Function A majority of the control of sexual functions in the male (and the female) begins with secretions of gonadotropin-releasing hormone (GnRH) by the hypothalamus.
Hormonal Control of Male Sexual Function A majority of the control of sexual functions in the male (and the female) begins with secretions of gonadotropin-releasing hormone (GnRH) by the hypothalamus.
Corticosteroids affect the testicular androgen production in male common carp, Cyprinus carpio L.
 Chapter 5 Corticosteroids affect the testicular androgen production in male common carp, Cyprinus carpio L. Co-authors: Jan G.D. Lambert, Henk J.Th. Goos submitted inhibits the testicular androgen production
Chapter 5 Corticosteroids affect the testicular androgen production in male common carp, Cyprinus carpio L. Co-authors: Jan G.D. Lambert, Henk J.Th. Goos submitted inhibits the testicular androgen production
Reproductive Hormones
 Reproductive Hormones Male gonads: testes produce male sex cells! sperm Female gonads: ovaries produce female sex cells! ovum The union of male and female sex cells during fertilization produces a zygote
Reproductive Hormones Male gonads: testes produce male sex cells! sperm Female gonads: ovaries produce female sex cells! ovum The union of male and female sex cells during fertilization produces a zygote
The Male Reproductive System
 The Male Reproductive System YONG-MEI CHEN ( 陈咏梅 ) Dept. of Anatomy, Histology & Embryology Peking Union Medical College Tel:69156461 E-mail address: pumc_he@126.com Content Spermatogenesis Spermiogenesis
The Male Reproductive System YONG-MEI CHEN ( 陈咏梅 ) Dept. of Anatomy, Histology & Embryology Peking Union Medical College Tel:69156461 E-mail address: pumc_he@126.com Content Spermatogenesis Spermiogenesis
612.6I7.5:612.6I6.I. different, but most of them appear to be satisfactory from a qualitative
 442 612.6I7.5:612.6I6.I SIZE CHANGES IN THE SEMINAL VESICLES OF THE MOUSE DURING DEVELOPMENT AND AFTER CASTRATION. BY RUTH DEANESLY AND A. S. PARKES'. (From the National Institute for Medical Research,
442 612.6I7.5:612.6I6.I SIZE CHANGES IN THE SEMINAL VESICLES OF THE MOUSE DURING DEVELOPMENT AND AFTER CASTRATION. BY RUTH DEANESLY AND A. S. PARKES'. (From the National Institute for Medical Research,
Stability of thyroxine and triiodothyronine in
 Stability of thyroxine and triiodothyronine in biological fluids LYNN NYE, T. H. YEO, VIVIAN CHAN1, D. GOLDIE2, AND J. From the Department of Chemical Pathology, St Bartholomew's Hospital, London EC] J.
Stability of thyroxine and triiodothyronine in biological fluids LYNN NYE, T. H. YEO, VIVIAN CHAN1, D. GOLDIE2, AND J. From the Department of Chemical Pathology, St Bartholomew's Hospital, London EC] J.
Induced Ovulation in the Mouse and the Measurement of Its Inhibition
 Induced Ovulation in the Mouse and the Measurement of Its Inhibition t' NATESAIER PURSHOTTAM, ~ MARCUS M. MASON, AND GREGORY PINCUS RUNNER has shown that the immature mouse ovary responds by superovulation
Induced Ovulation in the Mouse and the Measurement of Its Inhibition t' NATESAIER PURSHOTTAM, ~ MARCUS M. MASON, AND GREGORY PINCUS RUNNER has shown that the immature mouse ovary responds by superovulation
estimates were made of the normal rate of increase in plasma urea over periods in skin and in plasma, hypertonic sodium chloride solution was
 482 J. Physiol. (I95I) II5, 482-487 THE STTE OF BODY WTER IN THE CT BY M. GRCE EGGLETON From the Department of Physiology, University College, London (Received 5 July 1951) In the course of an investigation
482 J. Physiol. (I95I) II5, 482-487 THE STTE OF BODY WTER IN THE CT BY M. GRCE EGGLETON From the Department of Physiology, University College, London (Received 5 July 1951) In the course of an investigation
Developmental stages of fetal-type Leydig cells in prepubertal rats
 Development 107, 213-220 (1989) Printed in Great Britain The Company of Biologists Limited 1989 213 Developmental stages of fetal-type Leydig cells in prepubertal rats T. KUOPIO 1 *, J. TAPANAINEN 2, L.
Development 107, 213-220 (1989) Printed in Great Britain The Company of Biologists Limited 1989 213 Developmental stages of fetal-type Leydig cells in prepubertal rats T. KUOPIO 1 *, J. TAPANAINEN 2, L.
of Nebraska - Lincoln
 University of Nebraska - Lincoln DigitalCommons@University of Nebraska - Lincoln Roman L. Hruska U.S. Meat Animal Research Center U.S. Department of Agriculture: Agricultural Research Service, Lincoln,
University of Nebraska - Lincoln DigitalCommons@University of Nebraska - Lincoln Roman L. Hruska U.S. Meat Animal Research Center U.S. Department of Agriculture: Agricultural Research Service, Lincoln,
Evidence for a major role of inhibin in the feedback control of FSH in the male rat
 Evidence for a major role of inhibin in the feedback control of FSH in the male rat G. F. Weinbauer, J. M. S. Bartlett, U. Fingscheidt, C. G. Tsonis, D. M. de Kretser and E. Nieschlag fmflx Planck Clinical
Evidence for a major role of inhibin in the feedback control of FSH in the male rat G. F. Weinbauer, J. M. S. Bartlett, U. Fingscheidt, C. G. Tsonis, D. M. de Kretser and E. Nieschlag fmflx Planck Clinical
Superovulation of Beef Heifers with Follicle Stimulating Hormone or Human Menopausal Gonadotropin: Acute Effects on Hormone Secretion
 Superovulation of Beef Heifers with Follicle Stimulating Hormone or Human Menopausal Gonadotropin: Acute Effects on Hormone Secretion A.S. Leaflet R1362 Acacia A. Alcivar, graduate research assistant,
Superovulation of Beef Heifers with Follicle Stimulating Hormone or Human Menopausal Gonadotropin: Acute Effects on Hormone Secretion A.S. Leaflet R1362 Acacia A. Alcivar, graduate research assistant,
Validation of coupled bioassay for inhibin by pituitary cell culture assay
 J. Biosci., Vol. 14, No. 2, June 1989, pp. 79 90. Printed in India. Validation of coupled bioassay for inhibin by pituitary cell culture assay SUSHMA R. DHINGRA, S. DURAISWAMI, NARENDER KUMAR* and LAKSHMI
J. Biosci., Vol. 14, No. 2, June 1989, pp. 79 90. Printed in India. Validation of coupled bioassay for inhibin by pituitary cell culture assay SUSHMA R. DHINGRA, S. DURAISWAMI, NARENDER KUMAR* and LAKSHMI
Chapter 14 The Reproductive System
 Biology 12 Name: Reproductive System Per: Date: Chapter 14 The Reproductive System Complete using BC Biology 12, page 436-467 14. 1 Male Reproductive System pages 440-443 1. Distinguish between gametes
Biology 12 Name: Reproductive System Per: Date: Chapter 14 The Reproductive System Complete using BC Biology 12, page 436-467 14. 1 Male Reproductive System pages 440-443 1. Distinguish between gametes
Male Reproduction Organs. 1. Testes 2. Epididymis 3. Vas deferens 4. Urethra 5. Penis 6. Prostate 7. Seminal vesicles 8. Bulbourethral glands
 Outline Terminology Human Reproduction Biol 105 Lecture Packet 21 Chapter 17 I. Male Reproduction A. Reproductive organs B. Sperm development II. Female Reproduction A. Reproductive organs B. Egg development
Outline Terminology Human Reproduction Biol 105 Lecture Packet 21 Chapter 17 I. Male Reproduction A. Reproductive organs B. Sperm development II. Female Reproduction A. Reproductive organs B. Egg development
MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question.
 BIO 210 Anatomy and Physiology Homework #10: Chs. 26-28 DUE: (see course schedule) Assignments not turned in at the beginning of class will be accepted. MULTIPLE CHOICE. Choose the one alternative that
BIO 210 Anatomy and Physiology Homework #10: Chs. 26-28 DUE: (see course schedule) Assignments not turned in at the beginning of class will be accepted. MULTIPLE CHOICE. Choose the one alternative that
Refer to the figure below, a diagram of a renal tubule, to answer the following questions.
 1. The digestion and utilization of which nutrient creates the greatest need for osmoregulation by the kidneys? a. protein b. starch c. fat d. oil e. cellulose 2. Which of the following is true of urea?
1. The digestion and utilization of which nutrient creates the greatest need for osmoregulation by the kidneys? a. protein b. starch c. fat d. oil e. cellulose 2. Which of the following is true of urea?
- production of two types of gametes -- fused at fertilization to form zygote
 Male reproductive system I. Sexual reproduction -- overview - production of two types of gametes -- fused at fertilization to form zygote - promotes genetic variety among members of a species -- each offspring
Male reproductive system I. Sexual reproduction -- overview - production of two types of gametes -- fused at fertilization to form zygote - promotes genetic variety among members of a species -- each offspring
Yutaka; SAKAMOTO, Hiromi. Citation 泌尿器科紀要 (1986), 32(10):
 Title46XX male; report of case OKUYAMA, Akihiko; KONDO, Nobuyuki; Author(s) NAKAMURA, Masahiro; SONODA, Takao; Yutaka; SAKAMOTO, Hiromi Citation 泌尿器科紀要 (1986), 32(10): 1539-1542 Issue Date 1986-10 URL
Title46XX male; report of case OKUYAMA, Akihiko; KONDO, Nobuyuki; Author(s) NAKAMURA, Masahiro; SONODA, Takao; Yutaka; SAKAMOTO, Hiromi Citation 泌尿器科紀要 (1986), 32(10): 1539-1542 Issue Date 1986-10 URL
published by Hektoen and SchuIhof (1), and since the appearance
 ON THE INNERVATION AND SECRETORY PATH OF THE THYROID GLAND. BY C. S. HICKS (Beit Memorial Fellow). (From the Biochemistry Laboratories, Cambridge, and Physiology Department, University of Adelaide.) IN
ON THE INNERVATION AND SECRETORY PATH OF THE THYROID GLAND. BY C. S. HICKS (Beit Memorial Fellow). (From the Biochemistry Laboratories, Cambridge, and Physiology Department, University of Adelaide.) IN
NROSCI/BIOSC 1070 and MSNBIO 2070 September 11, 2017 Control Mechanisms 2: Endocrine Control
 NROSCI/BIOSC 1070 and MSNBIO 2070 September 11, 2017 Control Mechanisms 2: Endocrine Control Hormones are chemical messengers that are secreted into the blood by endocrine cells or specialized neurons.
NROSCI/BIOSC 1070 and MSNBIO 2070 September 11, 2017 Control Mechanisms 2: Endocrine Control Hormones are chemical messengers that are secreted into the blood by endocrine cells or specialized neurons.
The effect of thyroid activity on adult rat spermatogenesis
 The effect of thyroid activity on adult rat spermatogenesis Ai, J. 1* ; Zarifkar, A. 2 ; Takhshid, M. A. 3 ; Alavi, J. 1 and Moradzadeh, M. 2 1 Department of Anatomical Sciences, School of Medicine, University
The effect of thyroid activity on adult rat spermatogenesis Ai, J. 1* ; Zarifkar, A. 2 ; Takhshid, M. A. 3 ; Alavi, J. 1 and Moradzadeh, M. 2 1 Department of Anatomical Sciences, School of Medicine, University
