Anticancer pyrroloquinazoline LBL1 targets nuclear lamins
|
|
|
- Raymond Lane
- 5 years ago
- Views:
Transcription
1 Supporting Information Anticancer pyrroloquinazoline LBL1 targets nuclear lamins Bingbing X. Li, #,* Jingjin Chen, # Bo Chao, # Larry L. David, & Xiangshu Xiao #,* # Program in Chemical Biology, Department of Physiology and Pharmacology, & Department of Biochemistry and Molecular Biology, regon Health & Science University, 3181 SW Sam Jackson Park Rd, Portland, regon 97239, USA Synthetic procedures......s2 Figure S1... S6 Figure S2... S8 Figure S3... S9 Figure S4. S10 Figure S5. S11 Figure S6. S12 References.. S13 S1
2 H H 2 H Cs 2 C 3, propargyl bromide H H 2 LBL1 1 -(1-amino-7-(prop-2-yn-1-yl)-7H-pyrrolo[3,2-f]quinazolin-3-yl)-2-naphthamide (1). To a solution of LBL1 (45 mg, mmol) in DMF (2 ml) was added Cs 2C 3 (83.0 mg, mmol) at rt. After stirring for 30 min, propargyl bromide (12 µl, 0.22 mmol) was added and the resulting mixture was stirred for an additional 4 h. The solid was filtered off and the filtrate was concentrated. The residue was purified by column chromatography on silica gel, eluting with 20:1 EtAc:THF containing 1% DIPEA to give the a gray solid that was further treated with Et 2 (3 ml) and collected by filtration to give the desired compound 1 (8.0 mg, 16%) as a gray solid: mp C. H MR (400 MHz, DMS-d o 1 6) d (s, 1 H), 8.65 (s, 1 H), (m, 5 H), (m, 3 H), 7.45 (d, J = 9.2 Hz, 1 H), 7.34 (d, J = 3.2 Hz, 1 H), 7.28 (brs, 2 H), 5.28 (d, J = 2.4 Hz, 2 H), 3.50 (t, J = 2.4 Hz, 1 H); ESIMS, calcd 392.1, found H 2 H S1 H + S2 H S3 H H 4-Benzoyl--(1-hydroxy-7H-pyrrolo[3,2-f]quinazolin-3-yl)benzamide (S3). A mixture of S2 1 (363 mg, 1.12 mmol) and S1 (250 mg, 0.75 mmol) in dry DMF (5 ml) was stirred at 90 o C for 1 h. Then the solvent was removed and the residue was purified by column chromatography on silica S2
3 gel, eluting with 3:1 EtAc:DCM containing 1% DIPEA to give compound S3, which was washed with DCM (3 ml) to give the desired compound as a yellow solid (80 mg, 26% yields): mp C. H MR (400 MHz, DMS-d o 1 6) d 12.6 (brs, 2 H), 11.7 (s, 1 H), 8.28 (d, J = 8.0 Hz, 2 H), (m, 3 H), 7.78 (d, J = 8.0 Hz, 2 H), (m, 1 H), (m, 3 H), 7.33 (d, J = 8.4 Hz, 1 H), 7.24 (s, 1 H). H H H i) BP, DBU ii) H 3 /MeH H H 2 H S3 2 -(1-amino-7H-pyrrolo[3,2-f]quinazolin-3-yl)-4-benzoylbenzamide (2). To a stirred solution of S3 (50.0 mg, mmol) in dry DMF (3 ml) was added BP (70.4 mg, mmol) and DBU (27.6 µl, mmol). The resultant reaction mixture was stirred for 4 h at 25 C. Then o H 3 (7 in MeH, 0.70 ml, 4.90 mmol) was added. The reaction mixture was stirred at 25 C o for 16 h. The solvent was removed and the residue was purified by column chromatography on silica gel, eluting with 4:1 EtAc:THF containing 1% DIPEA to give a yellow solid, which was further treated with DCM (2 ml) and collected by filtration to give the desired compound 2 (12.0 mg, 25%) as a yellow solid: mp C. H MR (400 MHz, DMS-d o 1 6) d (s, 1 H), (s, 1 H), 8.12 (d, J = 5.2 Hz, 2 H), 7.89 (d, J = 8.8 Hz, 1 H), 7.82 (d, J = 8.0 Hz, 2 H), (m, 2 H), (m, 1 H), (m, 3 H), 7.35 (d, J = 8.4 Hz, 1 H), 7.31 (brs, 1 H), 7.23 (brs, 2 H); ESIMS, calcd 408.1, found S3
4 H 2 H S1 H + F 3 C 8 F 3 C H S4 H H F 3 C H H H i) BP, DBU ii) H 3 /MeH F 3 C H H 2 H S4 3 -(1-hydroxy-7H-pyrrolo[3,2-f]quinazolin-3-yl)-4-(3-(trifluoromethyl)-3H-diazirin-3- yl)benzamide (S4). A mixture of 8 (180 mg, 0.55 mmol) and S1 (84 mg, mmol) in dry DMF (2 ml) was stirred at 90 C for 3 h. Then the solvent was removed and the residue was purified by o column chromatography on silica gel, eluting with 3:1 EtAc:DCM containing 1% DIPEA to give compound S4, which was washed with DCM (3 ml) to give the desired compound as a yellow solid (100 mg, 57% yield): mp >160 C (dec). H MR (400 MHz, DMS-d o 1 6) d 12.6 (brs, 2 H), 11.7 (s, 1 H), 8.23 (d, J = 8.4 Hz, 2 H), 7.87 (d, J = 8.8 Hz, 1 H), 7.61 (t, J = 2.4 Hz, 1 H), 7.42 (d, J = 8.4 Hz, 2 H), 7.31 (d, J = 8.8 Hz, 1 H), 7.22 (brs, 1 H). -(1-Amino-7H-pyrrolo[3,2-f]quinazolin-3-yl)-4-(3-(trifluoromethyl)-3H-diazirin-3- yl)benzamide (3). To a stirred solution of S4 (95.0 mg, 0.23 mmol) in dry DMF (4 ml) was added BP (153 mg, mmol) and DBU (67 µl, 0.46 mmol). The resultant reaction mixture was stirred for 6 h at 25 C. Then H o 3 (7 in MeH, 1.3 ml, 9.1 mmol) was added. The reaction mixture was stirred at 25 C for 16 h. The solvent was removed and the residue was o purified by column chromatography on silica gel, eluting with 20:1 to 4:1 EtAc:THF S4
5 containing 1% DIPEA to give the desired compound 3, which was treated with DCM (2 ml) and collected by filtration to give the desired compound 3 (19.0 mg, 20% yield) as a gray solid (23.0 mg starting material 10 was recovered): mp >200 C (dec). H MR (400 MHz, DMS-d o 1 6) d (s, 1 H), (s, 1 H), 8.08 (d, J = 8.0 Hz, 2 H), 7.89 (d, J = 8.8 Hz, 1 H), 7.60 (s, 1 H), 7.39 (d, J = 8.0 Hz, 2 H), 7.34 (d, J = 8.8 Hz, 1 H), 7.29 (s, 1 H), 7.20 (brs, 2 H); ESIMS, calcd 410.1, found S5
6 S6
7 Figure S1. In vitro cancer cell growth inhibitory activities of LBL1 in the CI-60 cancer cell panel. The GI 50 values (Log 10 transformed) were obtained after incubating the cells with LBL1 for 48 h using sulforhodamine B assay. S7
8 LBL1 (µm) input pulldown IP: streptavidin LA LC LB1 GAPDH Figure S2. Dose-dependent competition labeling experiment of Figure 3A. The experimental design is exactly the same as that in Figure 3A except that different concentrations of LBL1 were used. S8
9 LA 1 METPS--QRRATR SGAQASSTPLSPTRITRLQEKEDLQELDRLAVYIDRVRSLETEAGLRL 61 LB1 1 MAT ATPVPprmgSRAGGPTTPLSPTRLSRLQEKEELRELDRLAVYIDKVRSLETESALQL 62 LB2 1 MSPPSpgRRREQRrpraaatmATPLP----GRAGGPATPLSPTRLSRLQEKEELRELDRLAHYIDRVRALELEDRLLL 76 LA 62 RITESEEVVSREVSGIKAAYEAELGDARKTLDSVAKERARLQLELSKVREEFKELKARTKKEGDLIAAQARLKDLEALL 141 LB1 63 QVTEREEVRGRELTGLKALYETELADARRALDDTARERAKLQIELGKCKAEHDQLLLYAKKESDLGAQIKLREYEAAL 142 LB2 77 KISEKEEVTTREVSGIKALYESELADARRVLDETARERARLQIEIGKLRAELDEVKSAKKREGELTVAQGRVKDLESLF 156 LA 142 SKEAALSTALSEKRTLEGELHDLRGQVAKLEAALGEAKKQLQDEMLRRVDAERLQTMKEELDFQKIYSEELRETKRR 221 LB1 143 SKDAALATALGDKKSLEGDLEDLKDQIAQLEASLAAAKKQLADETLLKVDLERCQSLTEDLEFRKSMYEEEIETRRK 222 LB2 157 HRSEVELAAALSDKRGLESDVAELRAQLAKAEDGHAVAKKQLEKETLMRVDLERCQSLQEELDFRKSVFEEEVRETRRR 236 LA 222 HETRLVEIDGKQREFESRLADALQELRAQHEDQVEQYKKELEKTYSAKLDARQSAERSLVGAAHEELQQSRIRIDS 301 LB1 223 HETRLVEVDSGRQIEYEYKLAQALHEMREQHDAQVRLYKEELEQTYHAKLEARLSSEMTSTVSAREELMESRMRIES 302 LB2 237 HERRLVEVDSSRQQEYDFKMAQALEELRSQHDEQVRLYKLELEQTYQAKLDSAKLSSDQDKAASAAREELKEARMRLES 316 LA 302 LSAQLSQLQKQLAAKEAKLRDLEDSLARERDTSRRLLAEKEREMAEMRARMQQQLDEYQELLDIKLALDMEIHAYRKLLE 381 LB1 303 LSSQLSLQKESRACLERIQELEDLLAKEKDSRRMLTDKEREMAEIRDQMQQQLDYEQLLDVKLALDMEISAYRKLLE 382 LB2 317 LSYQLSGLQKQASAAEDRIRELEEAMAGERDKFRKMLDAKEQEMTEMRDVMQQQLAEYQELLDVKLALDMEIAYRKLLE 396 LA 382 GEEERL 387 LB1 383 GEEERL 388 LB2 397 GEEERL 402 Identities 60% 59% Figure S3. Sequence alignment of human LA(1-387), LB1(1-388) and LB2(1-402). The identical sequences are highlighted in green. S9
10 A input pulldown IP: streptavidin fragment IB: anti-flag B input pulldown IP: streptavidin fragment IB: anti-flag Figure S4. Representative blots to show that LBL1-P labeled LA(1-387). HEK293T cells were transfected with indicated FLAG-tagged LA fragments. Then the cells were treated with LBL1- P followed by click reaction with a biotin- 3. The lysates were then precipitated with streptavidin-agarose beads. The bound proteins were then analyzed by SDS-PAGE followed by anti-flag blotting analysis. S10
11 LA(1-387) LA(78-387) Coomassie gel Figure S5. LA(1-387) (left) and LA(78-387) were purified to homogeneity from E. coli. The purified proteins were run a 10% SDS-PAGE and then the gels were stained with coomassie blue. S11
12 Figure S6. LBL1 did not change the melting profile of GST. GST (2 µm) was incubated with LBL1 (0 or 5 µm). Then the melting of the samples was analyzed in the same way as in Figure 4E. S12
13 Figure S7. LBL1 induced DSB formation and apoptosis. (A) LBL1 induced phosphorylation of H2AX. MDA-MB-231 cells were treated with LBL1 or doxorubicin (Dox, 0.5 µm) for 24 h. Then the cells were harvested and the cell lysates were prepared for Western blot analysis with indicated antibodies. Dox was used as a positive control. (B-C) LBL1 induced apoptosis in MDA-MB-231 cells. The cells were treated with LBL1 for 5 days. Then the cells were analyzed by flow cytometry after the cells were stained with annexin V and propidium iodide (PI). Quantification of the apoptotic cells (Q1) was presented in (C). S13
14 References: (1) livo, H. F.; Perez-Hernandez,.; Liu, D.; Iruthayanathan, M.; 'Leary, B.; Homan, L. L.; Dillon, J. S., Synthesis and application of a photoaffinity analog of dehydroepiandrosterone (DHEA). Bioorg. Med. Chem. Lett. 2010, 20 (3), S14
15 Protein ID SpectraCounts (w/ LBL1) SpectraCounts (w/o LBL1) LMA_HUMA LMB1_HUMA PYC_HUMA K2C1_HUMA ACTB_HUMA CATD_HUMA PCCA_HUMA TBB5_HUMA K2C5_HUMA DHCR7_HUMA ACTA_HUMA K2C7_HUMA K1C9_HUMA K22E_HUMA K1C14_HUMA TBA1B_HUMA CH60_HUMA TBA1C_HUMA TBB4B_HUMA TBB2A_HUMA TBA1A_HUMA EF1A1_HUMA K2C6C_HUMA AT1A1_HUMA ADT2_HUMA ACTBL_HUMA K2C4_HUMA RA2_HUMA ERG7_HUMA HRPL_HUMA HS90B_HUMA AL3A2_HUMA MCCA_HUMA ECHA_HUMA HRPM_HUMA LMB2_HUMA ATPA_HUMA VDAC2_HUMA GRP78_HUMA AXA2_HUMA VDAC1_HUMA HSP7C_HUMA TBA8_HUMA DHX9_HUMA HRPK_HUMA CLH1_HUMA HRPU_HUMA EPL_HUMA 25 25
16 ADT3_HUMA ADT1_HUMA HRPC_HUMA HS90A_HUMA PM_HUMA FLA_HUMA G3P_HUMA KPYM_HUMA PPIA_HUMA ACT4_HUMA K2C75_HUMA K2C79_HUMA EA_HUMA GRP75_HUMA H4_HUMA K1C13_HUMA SCRB2_HUMA HRPF_HUMA HSP71_HUMA PDIA3_HUMA HRH1_HUMA H2B1J_HUMA ASAH1_HUMA PDIA6_HUMA SGMR1_HUMA DDX17_HUMA LPPRC_HUMA PPT1_HUMA H2B1K_HUMA UCL_HUMA MPCP_HUMA PTBP1_HUMA GLYM_HUMA MDHM_HUMA P5CS_HUMA ILF2_HUMA ILF3_HUMA LAC2_HUMA 6 15 HS71L_HUMA K22_HUMA HYEP_HUMA AT2A2_HUMA TIF1B_HUMA PLEC_HUMA DX39B_HUMA EGFR_HUMA DHC24_HUMA CTL2_HUMA RAB1B_HUMA 19 13
17 H2A1_HUMA AT_HUMA MUTA_HUMA CALX_HUMA ST48_HUMA ACADV_HUMA 8 12 TCPQ_HUMA 8 12 GBLP_HUMA 6 12 VDAC3_HUMA DDX3X_HUMA 9 12 RAB1A_HUMA DDX5_HUMA C03_HUMA ALDA_HUMA 8 11 ATPB_HUMA DPB_HUMA 9 11 EF2_HUMA 6 11 ABCD3_HUMA RAB7A_HUMA SYRC_HUMA 7 11 TERA_HUMA GAAB_HUMA 9 11 SF3B3_HUMA SQRD_HUMA HRPR_HUMA 7 11 H2A1B_HUMA HRH2_HUMA C02_HUMA C12_HUMA K2C72_HUMA K2C73_HUMA AATM_HUMA 8 10 PDIA1_HUMA 9 10 DLDH_HUMA 4 10 H14_HUMA 5 10 PLAK_HUMA 9 10 RL4_HUMA 4 10 _HUMA 6 10 SDHL_HUMA TM205_HUMA TECR_HUMA 9 10 MTCH2_HUMA 5 10 DX39A_HUMA ACT1_HUMA 8 10
Supplementary Figure 1. Chemical structures of activity-based probes (ABPs) and of click reagents used in this study.
 Supplementary Figure 1. Chemical structures of activity-based probes (ABPs) and of click reagents used in this study. In this study, one fluorophosphonate (FP, 1), three nitrophenol phosphonate probes
Supplementary Figure 1. Chemical structures of activity-based probes (ABPs) and of click reagents used in this study. In this study, one fluorophosphonate (FP, 1), three nitrophenol phosphonate probes
Supporting Information for. Boronic Acid Functionalized Aza-Bodipy (azabdpba) based Fluorescence Optodes for the. analysis of Glucose in Whole Blood
 Supporting Information for Boronic Acid Functionalized Aza-Bodipy (azabdpba) based Fluorescence Optodes for the analysis of Glucose in Whole Blood Yueling Liu, Jingwei Zhu, Yanmei Xu, Yu Qin*, Dechen Jiang*
Supporting Information for Boronic Acid Functionalized Aza-Bodipy (azabdpba) based Fluorescence Optodes for the analysis of Glucose in Whole Blood Yueling Liu, Jingwei Zhu, Yanmei Xu, Yu Qin*, Dechen Jiang*
Supporting Information. for. Access to pyrrolo-pyridines by gold-catalyzed. hydroarylation of pyrroles tethered to terminal alkynes
 Supporting Information for Access to pyrrolo-pyridines by gold-catalyzed hydroarylation of pyrroles tethered to terminal alkynes Elena Borsini 1, Gianluigi Broggini* 1, Andrea Fasana 1, Chiara Baldassarri
Supporting Information for Access to pyrrolo-pyridines by gold-catalyzed hydroarylation of pyrroles tethered to terminal alkynes Elena Borsini 1, Gianluigi Broggini* 1, Andrea Fasana 1, Chiara Baldassarri
Supporting Information for. Use of the Curtius Rearrangement of Acryloyl Azides in the Synthesis of. 3,5-Disubstituted Pyridines: Mechanistic Studies
 Supporting Information for Use of the Curtius Rearrangement of Acryloyl Azides in the Synthesis of 3,5-Disubstituted Pyridines: Mechanistic Studies Ta-Hsien Chuang* a, Yu-Chi Chen b and Someshwar Pola
Supporting Information for Use of the Curtius Rearrangement of Acryloyl Azides in the Synthesis of 3,5-Disubstituted Pyridines: Mechanistic Studies Ta-Hsien Chuang* a, Yu-Chi Chen b and Someshwar Pola
Tel: ; Fax: ;
 Tel.: +98 216 696 9291; Fax: +98 216 696 9291; E-mail: mrasadeghi@pasteur.ac.ir Tel: +98 916 113 7679; Fax: +98 613 333 6380; E-mail: abakhshi_e@ajums.ac.ir A Soluble Chromatin-bound MOI 0 1 5 0 1 5 HDAC2
Tel.: +98 216 696 9291; Fax: +98 216 696 9291; E-mail: mrasadeghi@pasteur.ac.ir Tel: +98 916 113 7679; Fax: +98 613 333 6380; E-mail: abakhshi_e@ajums.ac.ir A Soluble Chromatin-bound MOI 0 1 5 0 1 5 HDAC2
Analysis of fatty acid metabolism using Click-Chemistry and HPLC-MS
 Analysis of fatty acid metabolism using Click-Chemistry and HPLC-MS Alexander J. Pérez and Helge B. Bode -Supporting Information- Contents Experimental section Supplementary figures NMR spectra Page S2
Analysis of fatty acid metabolism using Click-Chemistry and HPLC-MS Alexander J. Pérez and Helge B. Bode -Supporting Information- Contents Experimental section Supplementary figures NMR spectra Page S2
Facile Cu(II) mediated conjugation of thioesters and thioacids to peptides and proteins under mild conditions
 Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2018 Facile Cu(II) mediated conjugation of thioesters and thioacids to peptides
Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2018 Facile Cu(II) mediated conjugation of thioesters and thioacids to peptides
Figure S1. (A) SDS-PAGE separation of GST-fusion proteins purified from E.coli BL21 strain is shown. An equal amount of GST-tag control, LRRK2 LRR
 Figure S1. (A) SDS-PAGE separation of GST-fusion proteins purified from E.coli BL21 strain is shown. An equal amount of GST-tag control, LRRK2 LRR and LRRK2 WD40 GST fusion proteins (5 µg) were loaded
Figure S1. (A) SDS-PAGE separation of GST-fusion proteins purified from E.coli BL21 strain is shown. An equal amount of GST-tag control, LRRK2 LRR and LRRK2 WD40 GST fusion proteins (5 µg) were loaded
Supporting Information
 Supporting Information Synthesis of N-Heteropolycyclic Compounds Including Quinazolinone Skeletons by Using Friedel-Crafts Alkylation Bu Keun Oh, Eun Bi Ko, Jin Wook Han* and Chang Ho Oh* Department of
Supporting Information Synthesis of N-Heteropolycyclic Compounds Including Quinazolinone Skeletons by Using Friedel-Crafts Alkylation Bu Keun Oh, Eun Bi Ko, Jin Wook Han* and Chang Ho Oh* Department of
Use of degradable cationic surfactants with cleavable linkages for enhancing the. chemiluminescence of acridinium ester labels. Supplementary Material
 Use of degradable cationic surfactants with cleavable linkages for enhancing the chemiluminescence of acridinium ester labels Supplementary Material Anand atrajan*and David Wen Siemens Healthcare Diagnostics
Use of degradable cationic surfactants with cleavable linkages for enhancing the chemiluminescence of acridinium ester labels Supplementary Material Anand atrajan*and David Wen Siemens Healthcare Diagnostics
Supporting Information. Copper-catalyzed cascade synthesis of benzimidazoquinazoline derivatives under mild condition
 Supporting Information Copper-catalyzed cascade synthesis of benzimidazoquinazoline derivatives under mild condition Shan Xu, Juyou Lu and Hua Fu* Key Laboratory of Bioorganic Phosphorus Chemistry and
Supporting Information Copper-catalyzed cascade synthesis of benzimidazoquinazoline derivatives under mild condition Shan Xu, Juyou Lu and Hua Fu* Key Laboratory of Bioorganic Phosphorus Chemistry and
Supplementary Materials
 Supplementary Materials Supplementary Materials and Methods Biochemical Methods Methods to assay HMT activities have been previously described (1). In vitro cell assays Proliferation and LCC calculations
Supplementary Materials Supplementary Materials and Methods Biochemical Methods Methods to assay HMT activities have been previously described (1). In vitro cell assays Proliferation and LCC calculations
Supporting Information. Palladium-Catalyzed Formylation of Aryl Iodides with HCOOH as
 Supporting Information Palladium-Catalyzed Formylation of Aryl Iodides with HCOOH as CO Source Guanglong Sun,,, Xue Lv,,, Yinan Zhang, Min Lei,*,, and Lihong Hu*, Jiangsu Key Laboratory for Functional
Supporting Information Palladium-Catalyzed Formylation of Aryl Iodides with HCOOH as CO Source Guanglong Sun,,, Xue Lv,,, Yinan Zhang, Min Lei,*,, and Lihong Hu*, Jiangsu Key Laboratory for Functional
Electronic Supplementary Information
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2015 Electronic Supplementary Information ovel pseudo[2]rotaxanes constructed by selfassembly of dibenzyl
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2015 Electronic Supplementary Information ovel pseudo[2]rotaxanes constructed by selfassembly of dibenzyl
Development of a near-infrared fluorescent probe for monitoring hydrazine in serum and living cells
 Supporting Information for Development of a near-infrared fluorescent probe for monitoring hydrazine in serum and living cells Sasa Zhu, Weiying Lin,* Lin Yuan State Key Laboratory of Chemo/Biosensing
Supporting Information for Development of a near-infrared fluorescent probe for monitoring hydrazine in serum and living cells Sasa Zhu, Weiying Lin,* Lin Yuan State Key Laboratory of Chemo/Biosensing
p-toluenesulfonic Acid-Mediated 1,3-Dipolar Cycloaddition of
 Supporting Information for: p-toluenesulfonic Acid-Mediated 1,3-Dipolar Cycloaddition of Nitroolefins with NaN 3 for Synthesis of 4-Aryl-NH-1,2,3-triazoles Xue-Jing Quan, Zhi-Hui Ren, Yao-Yu Wang, and
Supporting Information for: p-toluenesulfonic Acid-Mediated 1,3-Dipolar Cycloaddition of Nitroolefins with NaN 3 for Synthesis of 4-Aryl-NH-1,2,3-triazoles Xue-Jing Quan, Zhi-Hui Ren, Yao-Yu Wang, and
Supplementary Figure 1. Normal T lymphocyte populations in Dapk -/- mice. (a) Normal thymic development in Dapk -/- mice. Thymocytes from WT and Dapk
 Supplementary Figure 1. Normal T lymphocyte populations in Dapk -/- mice. (a) Normal thymic development in Dapk -/- mice. Thymocytes from WT and Dapk -/- mice were stained for expression of CD4 and CD8.
Supplementary Figure 1. Normal T lymphocyte populations in Dapk -/- mice. (a) Normal thymic development in Dapk -/- mice. Thymocytes from WT and Dapk -/- mice were stained for expression of CD4 and CD8.
Thiol-Activated gem-dithiols: A New Class of Controllable. Hydrogen Sulfide (H 2 S) Donors
 Thiol-Activated gem-dithiols: A New Class of Controllable Hydrogen Sulfide (H 2 S) Donors Yu Zhao, Jianming Kang, Chung-Min Park, Powell E. Bagdon, Bo Peng, and Ming Xian * Department of Chemistry, Washington
Thiol-Activated gem-dithiols: A New Class of Controllable Hydrogen Sulfide (H 2 S) Donors Yu Zhao, Jianming Kang, Chung-Min Park, Powell E. Bagdon, Bo Peng, and Ming Xian * Department of Chemistry, Washington
Supporting Information
 Supporting Information Wiley-VCH 2008 69451 Weinheim, Germany Enantioselective Rhodium-catalyzed Addition of Arylboronic Acids to α-ketoesters Hai-Feng Duan, Jian-Hua Xie, Xiang-Chen Qiao, Li-Xin Wang,
Supporting Information Wiley-VCH 2008 69451 Weinheim, Germany Enantioselective Rhodium-catalyzed Addition of Arylboronic Acids to α-ketoesters Hai-Feng Duan, Jian-Hua Xie, Xiang-Chen Qiao, Li-Xin Wang,
Supplemental Material
 Supplemental Material General Methods Unless otherwise indicated, all anhydrous solvents were commercially obtained and stored under nitrogen. Reactions were performed under an atmosphere of dry nitrogen
Supplemental Material General Methods Unless otherwise indicated, all anhydrous solvents were commercially obtained and stored under nitrogen. Reactions were performed under an atmosphere of dry nitrogen
Supporting Information
 Supporting Information Wiley-VCH 2008 69451 Weinheim, Germany Chemoselective Peptide Cyclization via Induced Traceless Staudinger Ligation Rolf Kleineweischede, Christian P.R. Hackenberger* Institute for
Supporting Information Wiley-VCH 2008 69451 Weinheim, Germany Chemoselective Peptide Cyclization via Induced Traceless Staudinger Ligation Rolf Kleineweischede, Christian P.R. Hackenberger* Institute for
Supporting Materials. Experimental Section. internal standard TMS (0 ppm). The peak patterns are indicated as follows: s, singlet; d,
 CuBr-Catalyzed Efficient Alkynylation of sp 3 C-H Bonds Adjacent to a itrogen Atom Zhiping Li and Chao-Jun Li* Department of Chemistry, McGill University, 801 Sherbrooke St. West, Montreal, Quebec H3A
CuBr-Catalyzed Efficient Alkynylation of sp 3 C-H Bonds Adjacent to a itrogen Atom Zhiping Li and Chao-Jun Li* Department of Chemistry, McGill University, 801 Sherbrooke St. West, Montreal, Quebec H3A
An Orthogonal Array Optimization of Lipid-like Nanoparticles for. mrna Delivery in Vivo
 Supporting Information An rthogonal Array ptimization of Lipid-like Nanoparticles for mrna Delivery in Vivo Bin Li, Xiao Luo, Binbin Deng, Junfeng Wang, David W. McComb, Yimin Shi, Karin M.L. Gaensler,
Supporting Information An rthogonal Array ptimization of Lipid-like Nanoparticles for mrna Delivery in Vivo Bin Li, Xiao Luo, Binbin Deng, Junfeng Wang, David W. McComb, Yimin Shi, Karin M.L. Gaensler,
Supporting Information. Efficient copper-catalyzed Michael addition of acrylic derivatives with primary alcohols in the presence of base
 Supporting Information Efficient copper-catalyzed Michael addition of acrylic derivatives with primary alcohols in the presence of base Feng Wang, a Haijun Yang, b Hua Fu, b,c * and Zhichao Pei a * a College
Supporting Information Efficient copper-catalyzed Michael addition of acrylic derivatives with primary alcohols in the presence of base Feng Wang, a Haijun Yang, b Hua Fu, b,c * and Zhichao Pei a * a College
NHC-catalyzed cleavage of vicinal diketones and. triketones followed by insertion of enones and
 Supporting Information for NHC-catalyzed cleavage of vicinal diketones and triketones followed by insertion of enones and ynones Ken Takaki*, Makoto Hino, Akira Ohno, Kimihiro Komeyama, Hiroto Yoshida
Supporting Information for NHC-catalyzed cleavage of vicinal diketones and triketones followed by insertion of enones and ynones Ken Takaki*, Makoto Hino, Akira Ohno, Kimihiro Komeyama, Hiroto Yoshida
Supporting information for. Synthesis of phenothiazines from cyclohexanones and. 2-aminobenzenethiols under transition-metal-free conditions
 Supporting information for Synthesis of phenothiazines from cyclohexanones and 2-aminobenzenethiols under transition-metal-free conditions Yunfeng Liao, a Pengcheng Jiang, a Shanping Chen, a Fuhong Xiao,
Supporting information for Synthesis of phenothiazines from cyclohexanones and 2-aminobenzenethiols under transition-metal-free conditions Yunfeng Liao, a Pengcheng Jiang, a Shanping Chen, a Fuhong Xiao,
Allenylphosphine oxides as simple scaffolds for. phosphinoylindoles and phosphinoylisocoumarins
 Supporting Information for Allenylphosphine oxides as simple scaffolds for phosphinoylindoles and phosphinoylisocoumarins G. Gangadhararao, Ramesh Kotikalapudi, M. Nagarjuna Reddy and K. C. Kumara Swamy*
Supporting Information for Allenylphosphine oxides as simple scaffolds for phosphinoylindoles and phosphinoylisocoumarins G. Gangadhararao, Ramesh Kotikalapudi, M. Nagarjuna Reddy and K. C. Kumara Swamy*
ph Switchable and Fluorescent Ratiometric Squarylium Indocyanine Dyes as Extremely Alkaline Sensors
 ph Switchable and Fluorescent Ratiometric Squarylium Indocyanine Dyes as Extremely Alkaline Sensors Jie Li, Chendong Ji, Wantai Yang, Meizhen Yin* State Key Laboratory of Chemical Resource Engineering,
ph Switchable and Fluorescent Ratiometric Squarylium Indocyanine Dyes as Extremely Alkaline Sensors Jie Li, Chendong Ji, Wantai Yang, Meizhen Yin* State Key Laboratory of Chemical Resource Engineering,
Synthesis and Blastocyst Implantation Inhibition Potential of Lupeol Derivatives in Female Mice
 Supporting Information Rec. Nat. Prod. 9:4 (2015) 561-566 Synthesis and Blastocyst Implantation Inhibition Potential of Lupeol Derivatives in Female Mice Anita Mahapatra 1*, Purvi Shah 1, Mehul Jivrajani
Supporting Information Rec. Nat. Prod. 9:4 (2015) 561-566 Synthesis and Blastocyst Implantation Inhibition Potential of Lupeol Derivatives in Female Mice Anita Mahapatra 1*, Purvi Shah 1, Mehul Jivrajani
Graduate School of Nutritional and Environmental Sciences, University of Shizuoka,
 Identification of Indole Alkaloid Structural Units Important for Stimulus Selective TRPM8 Inhibition: SAR Study of Naturally Occurring Iboga Derivatives Yuko Terada,, Mariko Kitajima,, Fuyumi Taguchi,
Identification of Indole Alkaloid Structural Units Important for Stimulus Selective TRPM8 Inhibition: SAR Study of Naturally Occurring Iboga Derivatives Yuko Terada,, Mariko Kitajima,, Fuyumi Taguchi,
Organic Semiconducting Nanoparticles as Efficient Photoacoustic Agents for Lightening Early Thrombus and
 Supporting Information rganic Semiconducting anoparticles as Efficient Photoacoustic Agents for Lightening Early Thrombus and Monitoring Thrombolysis in Living Mice Cao Cui,, Zhen Yang,, Xiang Hu, Jinjun
Supporting Information rganic Semiconducting anoparticles as Efficient Photoacoustic Agents for Lightening Early Thrombus and Monitoring Thrombolysis in Living Mice Cao Cui,, Zhen Yang,, Xiang Hu, Jinjun
Supporting Information
 Supporting Information Developing novel activity-based fluorescent probes that target different classes of proteases Qing Zhu, Aparna Girish, Souvik Chattopadhaya and Shao Q Yao * Departments of Chemistry
Supporting Information Developing novel activity-based fluorescent probes that target different classes of proteases Qing Zhu, Aparna Girish, Souvik Chattopadhaya and Shao Q Yao * Departments of Chemistry
Supporting Information
 Investigation of self-immolative linkers in the design of hydrogen peroxide metalloprotein inhibitors Jody L. Major Jourden, Kevin B. Daniel, and Seth M. Cohen* Department of Chemistry and Biochemistry,
Investigation of self-immolative linkers in the design of hydrogen peroxide metalloprotein inhibitors Jody L. Major Jourden, Kevin B. Daniel, and Seth M. Cohen* Department of Chemistry and Biochemistry,
Global profiling of dynamic protein palmitoylation
 Nature Methods Global profiling of dynamic protein palmitoylation Brent R Martin, Chu Wang, Alexander Adibekian, Sarah E Tully & Benjamin F Cravatt Supplementary Figure 1 Supplementary Figure 2 Supplementary
Nature Methods Global profiling of dynamic protein palmitoylation Brent R Martin, Chu Wang, Alexander Adibekian, Sarah E Tully & Benjamin F Cravatt Supplementary Figure 1 Supplementary Figure 2 Supplementary
Supplemental Information Supplemental information contains 6 figures and Supplemental Experimental Procedures.
 Supplemental Information Supplemental information contains 6 figures and Supplemental Experimental Procedures. Supplemental Figure Legends Fig. S1. HTS of Vpr inhibitors in the RIKE PDepo, related to Fig.
Supplemental Information Supplemental information contains 6 figures and Supplemental Experimental Procedures. Supplemental Figure Legends Fig. S1. HTS of Vpr inhibitors in the RIKE PDepo, related to Fig.
Supplementary Figure 1.TRIM33 binds β-catenin in the nucleus. a & b, Co-IP of endogenous TRIM33 with β-catenin in HT-29 cells (a) and HEK 293T cells
 Supplementary Figure 1.TRIM33 binds β-catenin in the nucleus. a & b, Co-IP of endogenous TRIM33 with β-catenin in HT-29 cells (a) and HEK 293T cells (b). TRIM33 was immunoprecipitated, and the amount of
Supplementary Figure 1.TRIM33 binds β-catenin in the nucleus. a & b, Co-IP of endogenous TRIM33 with β-catenin in HT-29 cells (a) and HEK 293T cells (b). TRIM33 was immunoprecipitated, and the amount of
Masatoshi Shibuya,Takahisa Sato, Masaki Tomizawa, and Yoshiharu Iwabuchi* Department of Organic Chemistry, Graduate School of Pharmaceutical Sciences,
 Oxoammonium ion/naclo 2 : An Expedient, Catalytic System for One-pot Oxidation of Primary Alcohols to Carboxylic Acid with Broad Substrate Applicability Masatoshi Shibuya,Takahisa Sato, Masaki Tomizawa,
Oxoammonium ion/naclo 2 : An Expedient, Catalytic System for One-pot Oxidation of Primary Alcohols to Carboxylic Acid with Broad Substrate Applicability Masatoshi Shibuya,Takahisa Sato, Masaki Tomizawa,
Figure S1. Sorting nexin 9 (SNX9) specifically binds psmad3 and not psmad 1/5/8. Lysates from AKR-2B cells untreated (-) or stimulated (+) for 45 min
 Figure S1. Sorting nexin 9 (SNX9) specifically binds psmad3 and not psmad 1/5/8. Lysates from AKR2B cells untreated () or stimulated () for 45 min with 5 ng/ml TGFβ or 10 ng/ml BMP4 were incubated with
Figure S1. Sorting nexin 9 (SNX9) specifically binds psmad3 and not psmad 1/5/8. Lysates from AKR2B cells untreated () or stimulated () for 45 min with 5 ng/ml TGFβ or 10 ng/ml BMP4 were incubated with
Nature Methods: doi: /nmeth Supplementary Figure 1
 Supplementary Figure 1 Subtiligase-catalyzed ligations with ubiquitin thioesters and 10-mer biotinylated peptides. (a) General scheme for ligations between ubiquitin thioesters and 10-mer, biotinylated
Supplementary Figure 1 Subtiligase-catalyzed ligations with ubiquitin thioesters and 10-mer biotinylated peptides. (a) General scheme for ligations between ubiquitin thioesters and 10-mer, biotinylated
Orvinols with Mixed Kappa/Mu Opioid Receptor Agonist Activity
 Supporting Information Orvinols with Mixed Kappa/Mu Opioid Receptor Agonist Activity Greedy, Benjamin M.; Bradbury, Faye.; Thomas, Mark P.; Grivas, Konstantinos; Cami-Kobeci, Gerta; Archambeau, Ashley.;
Supporting Information Orvinols with Mixed Kappa/Mu Opioid Receptor Agonist Activity Greedy, Benjamin M.; Bradbury, Faye.; Thomas, Mark P.; Grivas, Konstantinos; Cami-Kobeci, Gerta; Archambeau, Ashley.;
Supplementary Information
 Supplementary Information Targeted Disruption of the EZH2/EED Complex Inhibits EZH2- dependent Cancer Woojin Kim 1,2,3, Gregory H. Bird 2,3,4, Tobias Neff 5, Guoji Guo 1,2,3, Marc A. Kerenyi 1,2,3, Loren
Supplementary Information Targeted Disruption of the EZH2/EED Complex Inhibits EZH2- dependent Cancer Woojin Kim 1,2,3, Gregory H. Bird 2,3,4, Tobias Neff 5, Guoji Guo 1,2,3, Marc A. Kerenyi 1,2,3, Loren
Manganese powder promoted highly efficient and selective synthesis of fullerene mono- and biscycloadducts at room temperature
 Supplementary Information Manganese powder promoted highly efficient and selective synthesis of fullerene mono- and biscycloadducts at room temperature Weili Si 1, Xuan Zhang 1, Shirong Lu 1, Takeshi Yasuda
Supplementary Information Manganese powder promoted highly efficient and selective synthesis of fullerene mono- and biscycloadducts at room temperature Weili Si 1, Xuan Zhang 1, Shirong Lu 1, Takeshi Yasuda
Novel D-erythro N-Octanoyl Sphingosine Analogs As Chemo- and Endocrine. Resistant Breast Cancer Therapeutics
 Page 11 of 32 Cancer Chemotherapy and Pharmacology Novel D-erythro N-Octanoyl Sphingosine Analogs As Chemo- and Endocrine Resistant Breast Cancer Therapeutics James W. Antoon, Jiawang Liu, Adharsh P. Ponnapakkam,
Page 11 of 32 Cancer Chemotherapy and Pharmacology Novel D-erythro N-Octanoyl Sphingosine Analogs As Chemo- and Endocrine Resistant Breast Cancer Therapeutics James W. Antoon, Jiawang Liu, Adharsh P. Ponnapakkam,
Thermal shift binding experiments were carried out using Thermofluor 384 ELS system. Protein
 Supplementary Methods Thermal shift assays Thermal shift binding experiments were carried out using Thermofluor 384 ELS system. Protein unfolding was examined by monitoring the fluorescence of ANS (1-anilinonaphthalene-8-
Supplementary Methods Thermal shift assays Thermal shift binding experiments were carried out using Thermofluor 384 ELS system. Protein unfolding was examined by monitoring the fluorescence of ANS (1-anilinonaphthalene-8-
Supplementary Figure 1
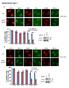 Supplementary Figure 1 a γ-h2ax MDC1 RNF8 FK2 BRCA1 U2OS Cells sgrna-1 ** 60 sgrna 40 20 0 % positive Cells (>5 foci per cell) b ** 80 sgrna sgrna γ-h2ax MDC1 γ-h2ax RNF8 FK2 MDC1 BRCA1 RNF8 FK2 BRCA1
Supplementary Figure 1 a γ-h2ax MDC1 RNF8 FK2 BRCA1 U2OS Cells sgrna-1 ** 60 sgrna 40 20 0 % positive Cells (>5 foci per cell) b ** 80 sgrna sgrna γ-h2ax MDC1 γ-h2ax RNF8 FK2 MDC1 BRCA1 RNF8 FK2 BRCA1
An Unusual Glycosylation Product from a Partially Protected Fucosyl Donor. under Silver Triflate activation conditions. Supporting information
 An Unusual Glycosylation Product from a Partially Protected Fucosyl Donor under Silver Triflate activation conditions Robin Daly a and Eoin M. Scanlan* a e-mail: eoin.scanlan@tcd.ie a Trinity Biomedical
An Unusual Glycosylation Product from a Partially Protected Fucosyl Donor under Silver Triflate activation conditions Robin Daly a and Eoin M. Scanlan* a e-mail: eoin.scanlan@tcd.ie a Trinity Biomedical
ELECTRONIC SUPPLEMENTARY INFORMATION
 ELECTRIC SUPPLEMETARY IFRMATI Searching for new cell-penetrating agents: hybrid cyclobutane-proline γ, γ peptides. Esther Gorrea, a Daniel Carbajo, b,c Raquel Gutiérrez-Abad, a na Illa, a Vicenç Branchadell,
ELECTRIC SUPPLEMETARY IFRMATI Searching for new cell-penetrating agents: hybrid cyclobutane-proline γ, γ peptides. Esther Gorrea, a Daniel Carbajo, b,c Raquel Gutiérrez-Abad, a na Illa, a Vicenç Branchadell,
Supporting Information
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2018 Supporting Information Facile Three-Step Synthesis and Photophysical Properties of [8]-, [9]-,
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2018 Supporting Information Facile Three-Step Synthesis and Photophysical Properties of [8]-, [9]-,
Supporting Information. Recyclable hypervalent-iodine-mediated solid-phase peptide
 Supporting Information Recyclable hypervalent-iodine-mediated solid-phase peptide synthesis and cyclic peptide synthesis Dan Liu, Ya-Li Guo, Jin Qu and Chi Zhang* for Address: State Key Laboratory of Elemento-Organic
Supporting Information Recyclable hypervalent-iodine-mediated solid-phase peptide synthesis and cyclic peptide synthesis Dan Liu, Ya-Li Guo, Jin Qu and Chi Zhang* for Address: State Key Laboratory of Elemento-Organic
Solid Phase Peptide Synthesis (SPPS) and Solid Phase. Fragment Coupling (SPFC) Mediated by Isonitriles
 Solid Phase Peptide Synthesis (SPPS) and Solid Phase Fragment Coupling (SPFC) Mediated by Isonitriles Ting Wang a and Samuel J. Danishefsky a,b,* alaboratory for Bioorganic Chemistry, Sloan- Kettering
Solid Phase Peptide Synthesis (SPPS) and Solid Phase Fragment Coupling (SPFC) Mediated by Isonitriles Ting Wang a and Samuel J. Danishefsky a,b,* alaboratory for Bioorganic Chemistry, Sloan- Kettering
Supporting Information Synthesis of 2-Aminobenzonitriles through Nitrosation Reaction and Sequential Iron(III)-Catalyzed C C Bond Cleavage of 2-Arylin
 Supporting Information Synthesis of 2-Aminobenzonitriles through Nitrosation Reaction and Sequential Iron(III)-Catalyzed C C Bond Cleavage of 2-Arylindoles Wei-Li Chen, Si-Yi Wu, Xue-Ling Mo, Liu-Xu Wei,
Supporting Information Synthesis of 2-Aminobenzonitriles through Nitrosation Reaction and Sequential Iron(III)-Catalyzed C C Bond Cleavage of 2-Arylindoles Wei-Li Chen, Si-Yi Wu, Xue-Ling Mo, Liu-Xu Wei,
Electronic Supplementary Information
 Electronic Supplementary Information A Novel and Facile Zn-mediated Intramolecular Five-membered Cyclization of β-tetraarylporphyrin Radicals from β-bromotetraarylporphyrins Dong-Mei Shen, Chao Liu, Qing-Yun
Electronic Supplementary Information A Novel and Facile Zn-mediated Intramolecular Five-membered Cyclization of β-tetraarylporphyrin Radicals from β-bromotetraarylporphyrins Dong-Mei Shen, Chao Liu, Qing-Yun
Supplementary Figure 1. MAT IIα is Acetylated at Lysine 81.
 IP: Flag a Mascot PTM Modified Mass Error Position Gene Names Score Score Sequence m/z [ppm] 81 MAT2A;AMS2;MATA2 35.6 137.28 _AAVDYQK(ac)VVR_ 595.83-2.28 b Pre-immu After-immu Flag- WT K81R WT K81R / Flag
IP: Flag a Mascot PTM Modified Mass Error Position Gene Names Score Score Sequence m/z [ppm] 81 MAT2A;AMS2;MATA2 35.6 137.28 _AAVDYQK(ac)VVR_ 595.83-2.28 b Pre-immu After-immu Flag- WT K81R WT K81R / Flag
Organic and biochemical synthesis of monolignol biosynthetic pathway intermediates
 Jie Liu 2012-2-8 Organic and biochemical synthesis of monolignol biosynthetic pathway intermediates 1. Organic synthesis of 5-hydroxyferulic acid Malonic acid 3, 4-Dihydroxy-5-methoxy-benzaldehyde 0.1
Jie Liu 2012-2-8 Organic and biochemical synthesis of monolignol biosynthetic pathway intermediates 1. Organic synthesis of 5-hydroxyferulic acid Malonic acid 3, 4-Dihydroxy-5-methoxy-benzaldehyde 0.1
A Hierarchy of Aryloxide Deprotection by Boron Tribromide. Supporting Information
 A Hierarchy of Aryloxide Deprotection by Boron Tribromide Sreenivas Punna, Stéphane Meunier and M. G. Finn* Department of Chemistry and The Skaggs Institute for Chemical Biology, The Scripps Research Institute,
A Hierarchy of Aryloxide Deprotection by Boron Tribromide Sreenivas Punna, Stéphane Meunier and M. G. Finn* Department of Chemistry and The Skaggs Institute for Chemical Biology, The Scripps Research Institute,
Supporting Information. Ruthenium(II)-Catalyzed C H Alkynylation of Weakly-Coordinating Benzoic Acids. Ruhuai Mei, Shou-Kun Zhang, and Lutz Ackermann*
 Supporting Information Ruthenium(II)-Catalyzed C H Alkynylation of Weakly-Coordinating Benzoic Acids Ruhuai Mei, Shou-Kun Zhang, and Lutz Ackermann* Institut für Organische und Biomolekulare Chemie, Georg-August-Universität
Supporting Information Ruthenium(II)-Catalyzed C H Alkynylation of Weakly-Coordinating Benzoic Acids Ruhuai Mei, Shou-Kun Zhang, and Lutz Ackermann* Institut für Organische und Biomolekulare Chemie, Georg-August-Universität
Scheme S1. Synthesis of glycose-amino ligand.
 Scheme S1. Synthesis of glycose-amino ligand. 5-Chloro-1-pentyl-2,3,4,6-tetra-O-acetyl-ß-D-glucopyranoside S2 To a solution of penta-o-acetyl-ß-d-glucopyranoside S1 (3.0 g, 7.69 mmol) and 5-chloropentan-1-ol
Scheme S1. Synthesis of glycose-amino ligand. 5-Chloro-1-pentyl-2,3,4,6-tetra-O-acetyl-ß-D-glucopyranoside S2 To a solution of penta-o-acetyl-ß-d-glucopyranoside S1 (3.0 g, 7.69 mmol) and 5-chloropentan-1-ol
Supporting Information. Use of Potassium. -Trifluoroborato Amides in Suzuki-Miyaura. Cross-Coupling Reactions
 Supporting Information Use of Potassium -Trifluoroborato Amides in Suzuki-Miyaura Cross-Coupling Reactions Gary A. Molander* and Ludivine Jean-Gérard Roy and Diana Vagelos Laboratories, Department of Chemistry,
Supporting Information Use of Potassium -Trifluoroborato Amides in Suzuki-Miyaura Cross-Coupling Reactions Gary A. Molander* and Ludivine Jean-Gérard Roy and Diana Vagelos Laboratories, Department of Chemistry,
Rameshwar Prasad Pandit and Yong Rok Lee * School of Chemical Engineering, Yeungnam University, Gyeongsan , Korea
 Electronic Supplementary Material (ESI) for rganic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2014 Novel ne-pot Synthesis of Diverse γ,δ-unsaturated β-ketoesters by Thermal
Electronic Supplementary Material (ESI) for rganic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2014 Novel ne-pot Synthesis of Diverse γ,δ-unsaturated β-ketoesters by Thermal
Fluorescent probes for detecting monoamine oxidase activity and cell imaging
 Fluorescent probes for detecting monoamine oxidase activity and cell imaging Xuefeng Li, Huatang Zhang, Yusheng Xie, Yi Hu, Hongyan Sun *, Qing Zhu * Supporting Information Table of Contents 1. General
Fluorescent probes for detecting monoamine oxidase activity and cell imaging Xuefeng Li, Huatang Zhang, Yusheng Xie, Yi Hu, Hongyan Sun *, Qing Zhu * Supporting Information Table of Contents 1. General
Supporting Information
 HYDRAmers: Design, synthesis and characterization of different generation novel Hydra-like dendrons based on multifunctionalized adamantane Giuseppe Lamanna,* Julie Russier, Cécilia Ménard-Moyon, and Alberto
HYDRAmers: Design, synthesis and characterization of different generation novel Hydra-like dendrons based on multifunctionalized adamantane Giuseppe Lamanna,* Julie Russier, Cécilia Ménard-Moyon, and Alberto
Bile acids initiate cholestatic liver injury by triggering a hepatic specific inflammatory response. Supplementary Results
 Bile acids initiate cholestatic liver injury by triggering a hepatic specific inflammatory response Shi-Ying Cai 1, Xinshou Ouyang 1, Yonglin Chen 1, Carol J. Soroka 1, Juxian Wang 2, Albert Mennone 1,
Bile acids initiate cholestatic liver injury by triggering a hepatic specific inflammatory response Shi-Ying Cai 1, Xinshou Ouyang 1, Yonglin Chen 1, Carol J. Soroka 1, Juxian Wang 2, Albert Mennone 1,
Pt IV azido complexes undergo copper-free click reactions with alkynes
 Electronic Supplementary Material (ESI) for Dalton Transactions. This journal is The Royal Society of Chemistry 208 Pt IV azido complexes undergo copper-free click reactions with alkynes Electronic Supplementary
Electronic Supplementary Material (ESI) for Dalton Transactions. This journal is The Royal Society of Chemistry 208 Pt IV azido complexes undergo copper-free click reactions with alkynes Electronic Supplementary
Nature Biotechnology: doi: /nbt.2354
 a b c Summed up peptide ion signals (arb. u.) 1E+03 INSR 1E+02 1E+01 1E+00-10 - 5 0 5 10 Summed up peptide ion signals (arb. u.) 1E+03 INSR 1E+02 1E+01 1E+00-10 -5 0 5 10 1E+03 1E+02 1E+01 INSR 1E+00-10
a b c Summed up peptide ion signals (arb. u.) 1E+03 INSR 1E+02 1E+01 1E+00-10 - 5 0 5 10 Summed up peptide ion signals (arb. u.) 1E+03 INSR 1E+02 1E+01 1E+00-10 -5 0 5 10 1E+03 1E+02 1E+01 INSR 1E+00-10
Supporting Information. Radical fluorination powered expedient synthesis of 3 fluorobicyclo[1.1.1]pentan 1 amine
![Supporting Information. Radical fluorination powered expedient synthesis of 3 fluorobicyclo[1.1.1]pentan 1 amine Supporting Information. Radical fluorination powered expedient synthesis of 3 fluorobicyclo[1.1.1]pentan 1 amine](/thumbs/94/121628201.jpg) Electronic Supplementary Material (ESI) for rganic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2015 Supporting Information Radical fluorination powered expedient synthesis
Electronic Supplementary Material (ESI) for rganic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2015 Supporting Information Radical fluorination powered expedient synthesis
Supplementary Notes. HTS compatible FRET-based conformational sensors clarify membrane receptor activation
 Supplementary otes TS compatible FRET-based conformational sensors clarify membrane receptor activation Pauline Scholler 1,2,3,4, David Moreno-Delgado 1,2,3, athalie Guillet-Lecat 1,2,3, Etienne Doumazane
Supplementary otes TS compatible FRET-based conformational sensors clarify membrane receptor activation Pauline Scholler 1,2,3,4, David Moreno-Delgado 1,2,3, athalie Guillet-Lecat 1,2,3, Etienne Doumazane
Lewis acid-catalyzed regioselective synthesis of chiral α-fluoroalkyl amines via asymmetric addition of silyl dienolates to fluorinated sulfinylimines
 Supporting Information for Lewis acid-catalyzed regioselective synthesis of chiral α-fluoroalkyl amines via asymmetric addition of silyl dienolates to fluorinated sulfinylimines Yingle Liu a, Jiawang Liu
Supporting Information for Lewis acid-catalyzed regioselective synthesis of chiral α-fluoroalkyl amines via asymmetric addition of silyl dienolates to fluorinated sulfinylimines Yingle Liu a, Jiawang Liu
Supporting Information
 Supporting Information Cobalt-Catalyzed Carbonylation of C(sp 2 )-H Bonds with Azodicarboxylate as the Carbonyl Source Jiabin Ni,, Jie Li,,š Zhoulong Fan,, and Ao Zhang *,,,š CAS Key Laboratory of Receptor
Supporting Information Cobalt-Catalyzed Carbonylation of C(sp 2 )-H Bonds with Azodicarboxylate as the Carbonyl Source Jiabin Ni,, Jie Li,,š Zhoulong Fan,, and Ao Zhang *,,,š CAS Key Laboratory of Receptor
Toxins 2016, 8, 222; doi: /toxins
 S1 of S8 Supplementary Materials: Mapping Protein Protein Interactions of the Resistance Related Bacterial Zeta Toxin Epsilon Antitoxin Complex (ε2ζ2) with High Affinity Peptide Ligands Using Fluorescence
S1 of S8 Supplementary Materials: Mapping Protein Protein Interactions of the Resistance Related Bacterial Zeta Toxin Epsilon Antitoxin Complex (ε2ζ2) with High Affinity Peptide Ligands Using Fluorescence
Regioective Halogenation of 2-Substituted-1,2,3-Triazole via sp 2 C-H Activation
 Regioective Halogenation of 2-Substituted-1,2,3-Triazole via sp 2 C-H Activation Qingshan Tian, Xianmin Chen, Wei Liu, Zechao Wang, Suping Shi, Chunxiang Kuang,* Department of Chemistry, Tongji University,
Regioective Halogenation of 2-Substituted-1,2,3-Triazole via sp 2 C-H Activation Qingshan Tian, Xianmin Chen, Wei Liu, Zechao Wang, Suping Shi, Chunxiang Kuang,* Department of Chemistry, Tongji University,
Microwave heating in peptide side chain modification via sulfhydryl reaction
 Microwave heating in peptide side chain modification via sulfhydryl reaction E. Calce and S. De Luca* Institute of Biostructures and Bioimaging, National Research Council, 80134 Naples, Italy stefania.deluca@cnr.it
Microwave heating in peptide side chain modification via sulfhydryl reaction E. Calce and S. De Luca* Institute of Biostructures and Bioimaging, National Research Council, 80134 Naples, Italy stefania.deluca@cnr.it
The Annexin V Apoptosis Assay
 The Annexin V Apoptosis Assay Development of the Annexin V Apoptosis Assay: 1990 Andree at al. found that a protein, Vascular Anticoagulant α, bound to phospholipid bilayers in a calcium dependent manner.
The Annexin V Apoptosis Assay Development of the Annexin V Apoptosis Assay: 1990 Andree at al. found that a protein, Vascular Anticoagulant α, bound to phospholipid bilayers in a calcium dependent manner.
Divergent Construction of Pyrazoles via Michael Addition of N-Aryl Hydrazones to 1,2-Diaza-1,3-dienes
 Divergent Construction of Pyrazoles via Michael Addition of N-Aryl Hydrazones to 1,2-Diaza-1,3-dienes Serena Mantenuto, Fabio Mantellini, Gianfranco Favi,* and Orazio A. Attanasi Department of Biomolecular
Divergent Construction of Pyrazoles via Michael Addition of N-Aryl Hydrazones to 1,2-Diaza-1,3-dienes Serena Mantenuto, Fabio Mantellini, Gianfranco Favi,* and Orazio A. Attanasi Department of Biomolecular
EXPERIMENTAL PROCEDURES, OPTIMIZATION STUDIES, CHARACTERIZATION DATA and NMR SPECTRA
 Supporting Information for Chemical Communications: S1 SUPPRTING INFRMATIN FR CHEM. CMMUN. Direct, facile synthesis of N-acyl-α-amino amides from α-keto esters and ammonia Rukundo Ntaganda, a Tamara Milovic,
Supporting Information for Chemical Communications: S1 SUPPRTING INFRMATIN FR CHEM. CMMUN. Direct, facile synthesis of N-acyl-α-amino amides from α-keto esters and ammonia Rukundo Ntaganda, a Tamara Milovic,
mm C3a. 1 mm C3a Time (s) C5a. C3a. Blank. 10 mm Time (s) Time (s)
 125 I-C5a (cpm) Fluorescnece Em 520nm a 4000 3000 2000 1000 c 0 5000 4000 3000 2000 Blank C5a C3a 6 0.3 mm C3a 7 9 10 11 12 13 15 16 0.3 mm C5a 0 300 600 900 1200 Time (s) 17 Fluorescnece Em 520nm Fluorescnece
125 I-C5a (cpm) Fluorescnece Em 520nm a 4000 3000 2000 1000 c 0 5000 4000 3000 2000 Blank C5a C3a 6 0.3 mm C3a 7 9 10 11 12 13 15 16 0.3 mm C5a 0 300 600 900 1200 Time (s) 17 Fluorescnece Em 520nm Fluorescnece
Ruthenium-Catalyzed C H Oxygenation on Aryl Weinreb Amides
 Supporting Information Ruthenium-Catalyzed C H xygenation on Aryl Weinreb Amides Fanzhi Yang and Lutz Ackermann* Institut für rganische und Biomolekulare Chemie Georg-August-Universität Tammannstrasse
Supporting Information Ruthenium-Catalyzed C H xygenation on Aryl Weinreb Amides Fanzhi Yang and Lutz Ackermann* Institut für rganische und Biomolekulare Chemie Georg-August-Universität Tammannstrasse
Supplemental Information. The Hedgehog Pathway Effector Smoothened. Exhibits Signaling Competency in the Absence. of Ciliary Accumulation
 Chemistry & Biology, Volume 21 Supplemental Information The edgehog Pathway Effector Exhibits Signaling Competency in the Absence of Ciliary Accumulation Chih-Wei Fan, Baozhi Chen, Irene Franco, Jianming
Chemistry & Biology, Volume 21 Supplemental Information The edgehog Pathway Effector Exhibits Signaling Competency in the Absence of Ciliary Accumulation Chih-Wei Fan, Baozhi Chen, Irene Franco, Jianming
Supplementary Figure S1 Supplementary Figure S2
 Supplementary Figure S A) The blots shown in Figure B were qualified by using Gel-Pro analyzer software (Rockville, MD, USA). The ratio of LC3II/LC3I to actin was then calculated. The data are represented
Supplementary Figure S A) The blots shown in Figure B were qualified by using Gel-Pro analyzer software (Rockville, MD, USA). The ratio of LC3II/LC3I to actin was then calculated. The data are represented
Dual-site Controlled and Lysosome-targeted ICT-PET-FRET. Fluorescent Probe for Monitoring ph Changes in Living Cells
 Supporting information for Dual-site Controlled and Lysosome-targeted ICT-PET-FRET Fluorescent Probe for Monitoring ph Changes in Living Cells Baoli Dong, Xuezhen Song, Chao Wang, Xiuqi Kong, Yonghe Tang
Supporting information for Dual-site Controlled and Lysosome-targeted ICT-PET-FRET Fluorescent Probe for Monitoring ph Changes in Living Cells Baoli Dong, Xuezhen Song, Chao Wang, Xiuqi Kong, Yonghe Tang
Efficient Metal-Free Pathway to Vinyl Thioesters with Calcium Carbide as the Acetylene Source
 Electronic Supplementary Material (ESI) for Green Chemistry. This journal is The Royal Society of Chemistry 2015 Supporting Information Efficient Metal-Free Pathway to Vinyl Thioesters with Calcium Carbide
Electronic Supplementary Material (ESI) for Green Chemistry. This journal is The Royal Society of Chemistry 2015 Supporting Information Efficient Metal-Free Pathway to Vinyl Thioesters with Calcium Carbide
Highly efficient hydrazination of conjugated nitroalkenes via imidazole or DMAP mediated Morita-Baylis-Hillman reaction
 Experimental Data and ptimization Tables For Highly efficient hydrazination of conjugated nitroalkenes via imidazole or DMAP mediated Morita-Baylis-Hillman reaction M. Dadwal, S. M. Mobin, I... amboothiri
Experimental Data and ptimization Tables For Highly efficient hydrazination of conjugated nitroalkenes via imidazole or DMAP mediated Morita-Baylis-Hillman reaction M. Dadwal, S. M. Mobin, I... amboothiri
A Novel Synthesis of Arylpyrrolo[1,2-a]pyrazinone Derivatives
![A Novel Synthesis of Arylpyrrolo[1,2-a]pyrazinone Derivatives A Novel Synthesis of Arylpyrrolo[1,2-a]pyrazinone Derivatives](/thumbs/77/74538875.jpg) Molecules 2004, 9, 574-582 molecules ISS 1420-049 http://www.mdpi.org A ovel Synthesis of ylpyrrolo[1,2-a]pyrazinone Derivatives Fei Wang*, Jiawei Wang and Shoufang Zhang School of Pharmaceutical Engineering,
Molecules 2004, 9, 574-582 molecules ISS 1420-049 http://www.mdpi.org A ovel Synthesis of ylpyrrolo[1,2-a]pyrazinone Derivatives Fei Wang*, Jiawei Wang and Shoufang Zhang School of Pharmaceutical Engineering,
Supporting information for
 Supporting information for A Coordination Gelator that Shows a Reversible Chromatic Change and a Sol-Gel Phase Transition Behavior upon xidative / Reductive Stimuli Shin-ichiro Kawano, orifumi Fujita,
Supporting information for A Coordination Gelator that Shows a Reversible Chromatic Change and a Sol-Gel Phase Transition Behavior upon xidative / Reductive Stimuli Shin-ichiro Kawano, orifumi Fujita,
Supplementary Information
 Supplementary Information Figure S1. Int6 gene silencing efficiency. (A) Western Blot analysis of Int6 expression at different times after sirna transfection. Int6 expression is strongly silenced in Int6
Supplementary Information Figure S1. Int6 gene silencing efficiency. (A) Western Blot analysis of Int6 expression at different times after sirna transfection. Int6 expression is strongly silenced in Int6
Development of a Cell-penetrating Peptide that Exhibits Responsive. Changes in its Secondary Structure in the Cellular Environment
 Development of a Cell-penetrating Peptide that Exhibits Responsive Changes in its Secondary Structure in the Cellular Environment iroko Yamashita, 1 Takuma Kato, 2 Makoto ba, 2 Takashi Misawa, 1 Takayuki
Development of a Cell-penetrating Peptide that Exhibits Responsive Changes in its Secondary Structure in the Cellular Environment iroko Yamashita, 1 Takuma Kato, 2 Makoto ba, 2 Takashi Misawa, 1 Takayuki
CDI Mediated Monoacylation of Symmetrical Diamines and Selective Acylation of Primary Amines of Unsymmetrical Diamines
 Supporting information: CDI Mediated Monoacylation of Symmetrical Diamines and Selective Acylation of Primary Amines of Unsymmetrical Diamines Sanjeev K. Verma*, Ramarao Ghorpade, Ajay Pratap and M. P.
Supporting information: CDI Mediated Monoacylation of Symmetrical Diamines and Selective Acylation of Primary Amines of Unsymmetrical Diamines Sanjeev K. Verma*, Ramarao Ghorpade, Ajay Pratap and M. P.
SUPPORTING INFORMATION. Transition metal-promoted synthesis of 2-aryl/heteroaryl-thioquinazoline: C-S
 1 SUPPORTING INFORMATION Transition metal-promoted synthesis of 2-aryl/heteroaryl-thioquinazoline: C-S Bond formation by Chan-Lam Cross-Coupling Reaction SATYA KARUNA PULAKHANDAM a, NARESH KUMAR KATARI
1 SUPPORTING INFORMATION Transition metal-promoted synthesis of 2-aryl/heteroaryl-thioquinazoline: C-S Bond formation by Chan-Lam Cross-Coupling Reaction SATYA KARUNA PULAKHANDAM a, NARESH KUMAR KATARI
Supplementary Information
 Supplementary Information Ruthenium(IV) porphyrin catalyzed phosphoramidation of aldehyde with phosphoryl azide as nitrene source Wenbo Xiao, Cong-Ying Zhou and Chi-Ming Che* Department of Chemistry, State
Supplementary Information Ruthenium(IV) porphyrin catalyzed phosphoramidation of aldehyde with phosphoryl azide as nitrene source Wenbo Xiao, Cong-Ying Zhou and Chi-Ming Che* Department of Chemistry, State
Direct Aerobic Carbonylation of C(sp 2 )-H and C(sp 3 )-H Bonds through Ni/Cu Synergistic Catalysis with DMF as the Carbonyl Source
 Direct Aerobic Carbonylation of C(sp 2 )-H and C(sp 3 )-H Bonds through Ni/Cu Synergistic Catalysis with DMF as the Carbonyl Source Xuesong Wu, Yan Zhao, and Haibo Ge* Table of Contents General Information...
Direct Aerobic Carbonylation of C(sp 2 )-H and C(sp 3 )-H Bonds through Ni/Cu Synergistic Catalysis with DMF as the Carbonyl Source Xuesong Wu, Yan Zhao, and Haibo Ge* Table of Contents General Information...
Supplementary Information
 Supplementary Information Supplementary Figure 1. CD4 + T cell activation and lack of apoptosis after crosslinking with anti-cd3 + anti-cd28 + anti-cd160. (a) Flow cytometry of anti-cd160 (5D.10A11) binding
Supplementary Information Supplementary Figure 1. CD4 + T cell activation and lack of apoptosis after crosslinking with anti-cd3 + anti-cd28 + anti-cd160. (a) Flow cytometry of anti-cd160 (5D.10A11) binding
Supplementary Figure S I: Effects of D4F on body weight and serum lipids in apoe -/- mice.
 Supplementary Figures: Supplementary Figure S I: Effects of D4F on body weight and serum lipids in apoe -/- mice. Male apoe -/- mice were fed a high-fat diet for 8 weeks, and given PBS (model group) or
Supplementary Figures: Supplementary Figure S I: Effects of D4F on body weight and serum lipids in apoe -/- mice. Male apoe -/- mice were fed a high-fat diet for 8 weeks, and given PBS (model group) or
Synthetic chemistry-led creation of a difluorinated biaryl ether non-nucleoside reverse transcriptase inhibitor
 upplementary Material for rganic & Biomolecular Chemistry ynthetic chemistry-led creation of a difluorinated biaryl ether non-nucleoside reverse transcriptase inhibitor Lyn. Jones* Amy Randall, scar Barba
upplementary Material for rganic & Biomolecular Chemistry ynthetic chemistry-led creation of a difluorinated biaryl ether non-nucleoside reverse transcriptase inhibitor Lyn. Jones* Amy Randall, scar Barba
Eur. J. Org. Chem WILEY-VCH Verlag GmbH & Co. KGaA, Weinheim, 2009 ISSN X SUPPORTING INFORMATION
 Eur. J. rg. Chem. 2009 WILEY-VC Verlag Gmb & Co. KGaA, 69451 Weinheim, 2009 ISS 1434 193X SUPPRTIG IFRMATI Title: ew GM1 Ganglioside Derivatives for Selective Single and Double Labelling of the atural
Eur. J. rg. Chem. 2009 WILEY-VC Verlag Gmb & Co. KGaA, 69451 Weinheim, 2009 ISS 1434 193X SUPPRTIG IFRMATI Title: ew GM1 Ganglioside Derivatives for Selective Single and Double Labelling of the atural
hexahistidine tagged GRP78 devoid of the KDEL motif (GRP78-His) on SDS-PAGE. This
 SUPPLEMENTAL FIGURE LEGEND Fig. S1. Generation and characterization of. (A) Coomassie staining of soluble hexahistidine tagged GRP78 devoid of the KDEL motif (GRP78-His) on SDS-PAGE. This protein was expressed
SUPPLEMENTAL FIGURE LEGEND Fig. S1. Generation and characterization of. (A) Coomassie staining of soluble hexahistidine tagged GRP78 devoid of the KDEL motif (GRP78-His) on SDS-PAGE. This protein was expressed
Supplementary Materials Contents
 Supplementary Materials Contents Supporting information... S1 1. General Information & Materials... S2 2. General Procedure for ptimization of Amidation of Aryl Bromides with Copper/,-Dimethylglycine Catalytic
Supplementary Materials Contents Supporting information... S1 1. General Information & Materials... S2 2. General Procedure for ptimization of Amidation of Aryl Bromides with Copper/,-Dimethylglycine Catalytic
Supporting Information for
 Supporting Information for Ligand-directed chemistry of AMPA receptors confers live-cell fluorescent biosensors under natural habitat Shigeki Kiyonaka*, Seiji Sakamoto, Sho Wakayama, Yuma Morikawa, Muneo
Supporting Information for Ligand-directed chemistry of AMPA receptors confers live-cell fluorescent biosensors under natural habitat Shigeki Kiyonaka*, Seiji Sakamoto, Sho Wakayama, Yuma Morikawa, Muneo
Nitro-Grela-type complexes containing iodides. robust and selective catalysts for olefin metathesis
 Supporting Information for Nitro-Grela-type complexes containing iodides robust and selective catalysts for olefin metathesis under challenging conditions. Andrzej Tracz, 1,2 Mateusz Matczak, 1 Katarzyna
Supporting Information for Nitro-Grela-type complexes containing iodides robust and selective catalysts for olefin metathesis under challenging conditions. Andrzej Tracz, 1,2 Mateusz Matczak, 1 Katarzyna
Electronic Supplementary Information
 Electronic Supplementary Information Cholate-Derived Amphiphilic Molecular Baskets as Glucose Transporters across Lipid Membranes Hongkwan Cho and Yan Zhao* Department of Chemistry, Iowa State University,
Electronic Supplementary Information Cholate-Derived Amphiphilic Molecular Baskets as Glucose Transporters across Lipid Membranes Hongkwan Cho and Yan Zhao* Department of Chemistry, Iowa State University,
Organic Letters. Synthesis of Oxygen-Free [2]Rotaxanes: Recognition of Diarylguanidinium Ions by Tetraazacyclophanes. and Sheng-Hsien Chiu*
![Organic Letters. Synthesis of Oxygen-Free [2]Rotaxanes: Recognition of Diarylguanidinium Ions by Tetraazacyclophanes. and Sheng-Hsien Chiu* Organic Letters. Synthesis of Oxygen-Free [2]Rotaxanes: Recognition of Diarylguanidinium Ions by Tetraazacyclophanes. and Sheng-Hsien Chiu*](/thumbs/93/111782529.jpg) Organic Letters Synthesis of Oxygen-Free [2]Rotaxanes: Recognition of Diarylguanidinium Ions by Tetraazacyclophanes Yu-Hsuan Chang, Yong-Jay Lee, Chien-Chen Lai, Yi-Hung Liu, Shie-Ming Peng, and Sheng-Hsien
Organic Letters Synthesis of Oxygen-Free [2]Rotaxanes: Recognition of Diarylguanidinium Ions by Tetraazacyclophanes Yu-Hsuan Chang, Yong-Jay Lee, Chien-Chen Lai, Yi-Hung Liu, Shie-Ming Peng, and Sheng-Hsien
Nature Immunology: doi: /ni.3866
 Nature Immunology: doi:10.1038/ni.3866 Supplementary Figure 1 The effect of TIPE2 on chemotaxis. a, The expression of TIPE2 in dhl-60c, dhl-60t, TIPE2-expressing and 15/16Q-expressing dhl-60t neutrophils
Nature Immunology: doi:10.1038/ni.3866 Supplementary Figure 1 The effect of TIPE2 on chemotaxis. a, The expression of TIPE2 in dhl-60c, dhl-60t, TIPE2-expressing and 15/16Q-expressing dhl-60t neutrophils
