Lesson 9. Measuring Emotional Responses to the Acoustic Startle Reflex: Stimulus Habituation & Sensitization
|
|
|
- Bryce Fisher
- 5 years ago
- Views:
Transcription
1 Measuring Emotional Responses to the Acoustic Startle Reflex: Stimulus Habituation & Sensitization Revised 10 / 29 / 2012 Lesson 9 METHOD: GALVANIC SKIN RESPONSE & THE POLYGRAPH David W. Pittman, Ph.D. Wofford College Modified from: Richard Pflanzer, Ph.D. Associate Professor Indiana University School of Medicine Purdue University School of Science William McMullen Vice President BIOPAC Systems, Inc. BIOPAC Systems, Inc. 42 Aero Camino, Goleta, CA (805) , Fax (805) info@biopac.com Web Site: BIOPAC Systems, Inc.
2 I. INTRODUCTION Electricity flows through an electrical circuit because of a difference in electrical pressure between the beginning and the end of a circuit. Electrical pressure or electromotive force (E) is measured in volts (V). The flow of electricity, called current (I), is measured in amperes (A) or amps for short. As electricity flows through the circuit, resistance to flow occurs. Electrical resistance (R) is measured in ohms ( ). In a simple circuit of direct electrical current, the relationship between the electromotive force causing the electrical current, the resistance to flow of electricity, and the resultant magnitude of the current is described by Ohm s Law. Ohm s Law: I (Amps) = E (Volts) / R (Ohms) If two of the three variables are known, the unknown third variable can be calculated. For example, if voltage and resistance values for a simple circuit are known, the above formula can be used to calculate the value for current; if the values for current and resistance are known, then the formula for computing voltage is E = IR. Ohm s Law implies that if a constant current is applied across a resistance, changes in the resistance will produce a voltage change directly proportional to the resistance change. For example, if a constant current of 1.0 ampere is applied across a resistance of 2.0 ohms, the measured voltage would be 2.0 volts (I = E/R, 1.0 ampere = 2.0 volts/2.0 ohms). If the resistance dropped to 0.5 ohm, the voltage would also fall to 0.5 volt (I = E/R, 1.0 amperes = 0.5 volt/0.5 ohm). In this lesson, you will apply principles of Ohm s Law and record changes in the electrical resistance of the skin. The human skin displays several forms of bioelectric phenomena, especially in areas of the extremities such as the fingers, palms of the hands, and soles of the feet. Galvanic skin resistance (GSR) When a feeble electric current is steadily applied between two electrodes placed about an inch apart, the recorded electrical resistance between them, referred to as the galvanic skin resistance (GSR), varies in accordance with the emotional state of the subject. Galvanic skin potential (GSP) Similarly, if the electrodes are connected to a suitable voltage amplifier, but without any externally applied current, the voltage measured between them, referred to as the galvanic skin potential (GSP), varies with the emotional state of the subject. The combined changes in the GSR and GSP related to the emotion of the subject constitute the galvanic skin response. The physiological basis of the galvanic skin response is a change in autonomic tone, largely sympathetic, occurring in the skin and subcutaneous tissue in response to a change in the affective state of the subject. Changes in peripheral autonomic tone alter sweating and cutaneous blood flow, which in turn change GSR and GSP. For example, if a painful stimulus such as a pinprick is applied to the skin in an area distant to the electrode, the stimulus will reflexively elicit a general phasic sympathetic discharge to sweat glands, increasing secretion. The increase in sweat, although generally small, lowers the electrical resistance of the skin because sweat contains water and electrolytes, both of which increase electrical conductivity of the skin.
3 As in the case of somatic sensory stimuli (e.g., pain, pressure, touch), changes in emotion elicit changes in peripheral autonomic tone and hence the galvanic skin response. A common example is the vasodilation of cutaneous blood vessels of the face (blushing) and increased sweating that often occur in the emotional state of embarrassment. The detection and recording of the galvanic skin response is often combined with the detection and recording of other autonomic-dependent psychophysiological variables such as heart rate, respiratory rate, and blood pressure. The device that detects and records these variables is called a polygraph. Although many people think polygraph is synonymous with lie detector, the literal meaning is many measures (poly - many, graph write). This lesson is a polygraph in the true sense of the word since it uses three types of measures: (a) GSR, (b) respiration, and (c) heart rate. One of the underlying principles involved in using the polygraph as a lie detector is that autonomic nervous system control of heart rate, respiratory rate, blood pressure and flow, and sweating cannot consciously be altered. Another principle is that changes in emotion associated with intentional falsification of answers to carefully selected and worded questions involuntarily and subconsciously alters autonomic output in such a way as to cause recognizable changes in recorded physiological variables. In the experiments that follow, you will record respiration, GSR, and heart rate under various experimental procedures so as to gain a better understanding of polygraphy, its applications, and its limitations. II. EXPERIMENTAL OBJECTIVES 1) To become familiar with procedures for recording the galvanic skin response. 2) To observe and record changes in respiratory rate, heart rate, and skin resistance associated with emotional auditory sensory stimuli. 3) To observe and record changes in respiratory rate, heart rate, and skin resistance associated with habituation and sensitization of the acoustic startle reflex. III. MATERIALS BIOPAC disposable vinyl electrodes (EL503) 3 electrodes per Subject BIOPAC Electrode lead set (SS2L) BIOPAC GSR setup GSR transducer (SS3L) and Electrode gel (GEL1) BIOPAC Respiration transducer (SS5LB or older SS5LA or SS5L) Computer system Biopac Student Lab 4.0 for PC running Windows BIOPAC acquisition unit (MP36 with USB) BIOPAC wall transformer (AC100A) Auditory stimuli (description to be included after data collection is completed)
4 IV. EXPERIMENTAL METHODS For further explanation, use the online support options under the Help Menu. A. SET UP FAST TRACK Set Up 1. You will use the recording computer to record your data and a computer for the stimulus presentation. 2. Make sure the BIOPAC MP36 unit is turned OFF. 3. Plug the transducers in as follows: Respiration (SS5LB) CH 1 Electrode lead set (SS2L) CH 2 GSR (SS3LA or SS57L) CH 3 DETAILED EXPLANATION OF SET UP STEPS The log-in for the laptops is: USER:.\PSYALL password: Psychology1 The log-in for the desktop computers is: USER: PSYNEURO password: Psychology1 BIOPAC MP35 or MP30 Respiratory Transducer (SS5LB or SS5LA or SS5L) plugs into CHannel 1 4. Turn the MP36 Data Acquisition Unit ON. 5. Attach the respiratory transducer (SS5LB) to the Subject (Fig. 9.2). IMPORTANT If using the SS5LA transducer, you must be very careful to not pull or yank on the rubber bow tie portion that contains the sensor element. Electrode lead set (SS2L) plugs into CHannel 2 GSR (SS3LA shown or SS57L lead) plugs into CHannel 3 Fig. 9.1 Equipment Connections Attach the respiratory transducer around the chest below the armpits and above the nipples (Fig. 9.2). The correct tension is critical. The respiratory transducer must be slightly tight at the point of maximal expiration. The respiration transducer can be applied over thin clothing, such as a t-shirt. Fig. 9.2 SS5LB Placement
5 Set up continues 6. Setup the GSR. HINT For a good signal to be picked up, it helps if the subjects have a little sweat on their hands (not a lot, but enough so that their hands are not completely smooth or cold). If subjects wash their hands just prior to the recording or if they have been sitting in a cold room, then they must do something to activate the sweat glands before beginning calibration or recording. If subjects begin with colder hands, the scale will be diminished and the signal will be easily saturated once they warm up during the lesson. SS3LA and GEL1 Fill both cavities of the GSR transducer (SS3L/SS3LA) with gel and attach to the Subject (Fig. 9.4). IMPORTANT You must fill both sensor cavities with electrode gel (GEL1) before attaching to the fingers. You must fill the each cavity of the SS3L/SS3LA GSR transducer with electrode gel to obtain accurate recordings. The SS3L and SS3LA attach to the fingertips in an identical manner (Fig. 9.4) and should be in place for at least five minutes prior to the start of recording. Sensors attach to bottom of fingertips Velcro straps wrap around fingers Fig. 9.4 SS3L/SS3LA attachment and connection The SS3L/SS3LA is typically placed on the index and middle finger of the left hand. Position the transducer so that the sensor is on the bottom of your fingertip (the part without the fingernail) and wrap the Velcro tape around the finger so the transducer fits snugly but not so tight that blood circulation is cut off. It s a fine line between tight and too tight. Set up continues
6 7. Set up the LEAD II recording. a) Place three disposable electrodes (EL503) on the Subject (Fig. 9.5). Place three electrodes at the positions shown (Fig. 9.5). one on right forearm (just above wrist) one on inside right leg (just above ankle bone) one on inside left leg (just above ankle bone) Fig. 9.5 medial surface of right leg, just above the ankle bone. medial surface of left leg, just above the ankle bone. right anterior forearm just above the wrist (same side of arm as palm of hand). b) Attach the electrode lead set (SS2L) to the electrodes (Fig. 9.6). right forearm WHITE lead right leg BLACK lead (ground) left leg RED lead Fig. 9.6 To establish a LEAD II electrode configuration, each of the pinch connectors on the end of the electrode cable must be attached to a specific electrode position. Follow Fig. 9.6 to ensure that you connect each lead (color) to the proper electrode. The pinch connectors will only latch onto the nipple of the electrode from one side of the connector. 8. Start the Biopac Student Lab program. 9. Choose Lesson 9 (L09-Poly-1). 10. Type in your filename. Click OK. END OF SET UP Use your last name and initials as your filename. This ends the Set Up procedure.
7 B. CALIBRATION The Calibration procedure establishes the hardware s internal parameters (such as gain, offset, and scaling) and is critical for optimum performance. Pay close attention to the Calibration procedure. FAST TRACK Calibration 1. Seat the blindfolded Subject in a comfortable position with hands palm down on the countertop. DETAILED EXPLANATION OF CALIBRATION STEPS The Subject should be seated in a relaxed, comfortable position. The left hand with the GSR electrodes should rest on the lab bench countertop with the palm and fingers placed face down on the countertop. The subject should be wearing the headphones and the blindfold. 2. Click Calibrate. The Calibrate button is in the upper left corner of the screen. 3. Three seconds into the recording, a beep will sound and Subject should inhale and exhale deeply for one cycle, then return to normal breathing. The program needs to see a change in the GSR recording during calibration. 4. Wait for the Calibration to stop. The Calibration will run for 10 seconds and then stop automatically, so let it run its course. 5. Check your calibration data. At the end of the 10-sec calibration recording, your screen should resemble Fig If similar, proceed to the Data Recording section. If different, Redo Calibration. END OF CALIBRATION Figure 9.8 Sample Calibration Data All three recording channels should show some fluctuation. There should be some variation 4-6 seconds into the GSR recording from the deep inhale. If a channel does not show fluctuation, the transducer may not be connected properly or the Subject may not have inhaled deeply enough. IT IS EXTREMELY IMPORTANT THAT YOU CAN SEE SMOOTH MOVEMENT OF THE RESPIRATION CHANNEL (red line) & THAT YOU CAN SEE THE GSR (green line) DURING THE ENTIRE CALIBRATION. IF NOT, THEN REDO THE CALIBRATION AND ADJUST THE RESPIRATION BELT. Click Redo Calibration and repeat the entire calibration sequence until your data resembles the sample data.
8 C. RECORDING LESSON DATA FAST TRACK Recording DETAILED EXPLANATION OF RECORDING STEPS 1. Prepare for the recording. Hints for obtaining optimal data: a) The Subject must stay alert, quiet, and motionless during the 10 minute test. b) The environment must be quiet. c) Sensory input besides the recording must be kept at a minimum since almost any change in the environment may evoke a response. d) Subject should be at his/her resting heart rate in a relaxed mental and physical state, and should not have performed any recent physical or mental exertion. BEGIN RECORDING 2. Click on Record. 3. Begin the test by opening either ASR- Stimulus1 or ASR-Stimulus2 on the stimulus presentation computer. It is important to open the stimulus with the CoolEdit program so you can visually see when the startle and midpoint stimuli are going to occur. You will need to insert markers for each stimulus and the midpoint stimulus. On the stimulus presentation computer, open the audio sample on the desktop ASR-Stimulus1 or ASR-Stimulus2 depending on the assignment of the subject in the pair. It should open in a program called CoolEdit and begin playing. If it does not begin playing there is a play button in the lower left corner of the window. As soon as the sample opens, it will begin playing. You will see a moving cursor on the audio sample. Insert a marker (F9) for each tone. IMPORTANT: There is a change in stimuli at the 5 minute mark. Insert a marker here at minute 5 for both Stimulus1 and Stimulus2. 4. Click Suspend. After returning to baseline following the last tone, press suspend to pause the recording. Make sure that your data is GOOD before quitting the experiment. SEE FIGURE 9.12 on next page for an example of good data. 5. Click Done. A pop-up window with options will appear. Make your choice, and continue as directed. If choosing the Record from another Subject option: a) Attach the sensors per Set Up Steps 5, 6, and 7 and continue the entire lesson from Set Up Step 10. Each person will need to use a unique file name. 6. Remove the sensors and clean-up. END OF RECORDING Remember to disconnect the GSR electrode and wash all of the gel out of the insets before replacing in the recording room
9 V. DATA ANALYSIS FAST TRACK Data Analysis 1. Enter the Review Saved Data mode and choose the correct file. Note Channel Number (CH) designations: Channel Displays CH 41 Heart Rate CH 40 Respiration CH 3 GSR 2. Setup your display window for optimal viewing of the first 5 seconds of the recording. 3. Set up the measurement boxes as follows: Channel Measurement CH 41 mean CH 40 BPM CH 3 p-p CH 3 delta T 4. For each acoustic startle stimulus and the sensitization stimulus at minute 5, you will measure heart rate, respiration, and GSR DETAILED EXPLANATION OF DATA ANALYSIS STEPS Enter Review Saved Data from the Lessons menu. Fig 9.12 The following tools help you adjust the data window: Autoscale horizontal Horizontal (Time) Scroll Bar Autoscale waveforms Vertical (Amplitude) Scroll Bar Zoom Tool Zoom Previous The measurement boxes are above the marker region in the data window. Each measurement has three sections: channel number, measurement type, and value. The first two sections are pulldown menus that are activated when you click on them. The following is a brief description of these specific measurements. mean: displays the average value for the channel for the selected area. BPM: In this lesson, the BPM measurement stands for Breaths Per Minute and calculates the difference in time between the end and beginning of the selected area (same as T), then divides this value into 60 seconds/minute. p-p: displays the amplitude value from the minimum to the maximum point of the selected data. The selected area is the area selected by the I-Beam tool (including the endpoints). Data Analysis continues Fig 9.13 For each measurement, you will need to record a response and a baseline in the GSR Individual Data EXCEL sheet available on the course web site. The Excel sheet will calculate the change from baseline. You should then and paste special values those values into the GROUP data sheet.
10 5. MEASURING RESPIRATION: Beginning with the first stimulus, using the I-Beam cursor, select an area from the start of the first inhale after the stimulus marker indicating the stimulus to the start of the next inhale (Fig. 9.14), and record the respiration rate (BPM) for stimulus 1a. Then, measure the distance between the start of the first inhale before the stimulus to the start of the previous inhale (i.e. a baseline measurement before the stimulus is delivered) and record the respiration rate (BPM) for stimulus 1a baseline. Record these values on your data sheet. The respiration transducer records chest expansion (inhalation) as positive values, and chest deflation (exhalation) as negative values. Therefore, the start of inhalation is recorded as the beginning of the ascending positive waveform. Note: This measurement may be difficult to perform, depending on your data, because small dips in chest expansion can occur within the normal cycle. You must be able to distinguish the small dips from the big dips. Fig 9.14 ABOVE: Example of respiration response measurement BELOW: Example of respiration baseline measurement
11 6. MEASURING GSR & HEART RATE: Also for the first stimulus, using the I- Bean cursor, select the data from the marker indicating stimulus 1 past the top of the GSR response to the stimulus (I would recommend 15s). Record the mean Heart Rate and the p- p GSR values on your data sheet. NOTE THE DELTA T value. You must now select an equivalent length (same delta T) area of the baseline prior to the stimulus and record a baseline Heart Rate and GSR. 7. Repeat Steps 5 & 6 for each of the 6 stimuli before the sensitization stimulus at minute 5, the sensitizations stimulus itself at 5 minutes, and the 6 stimuli following the sensitization stimulus. The Individual GSR Data sheet should automatically calculate your percent change from baseline and link to the cells in the row to copy and paste into the group data sheet. Enter your subject initials, year, sex (1=female, 2=male), and stimulus set (1 or 2) in the yellow highlighted row. Select those cells and in yellow and copy. Go to the GROUP data sheet on GOOGLE DOCS and use the EDIT PASTE VALUES ONLY command to paste your data into the sheet.
Lesson 1 ELECTROMYOGRAPHY I Standard and Integrated EMG
 Physiology Lessons for use with the PC running Windows 98SE, Me, 2000 Pro, XP Pro/Home/Media Lesson 1 ELECTROMYOGRAPHY I Standard and Integrated EMG Manual Revision PL3.7.0 080706 Richard Pflanzer, Ph.D.
Physiology Lessons for use with the PC running Windows 98SE, Me, 2000 Pro, XP Pro/Home/Media Lesson 1 ELECTROMYOGRAPHY I Standard and Integrated EMG Manual Revision PL3.7.0 080706 Richard Pflanzer, Ph.D.
Biopac Student Lab Lesson 5 ELECTROCARDIOGRAPHY (ECG) I Procedure. Rev
 42 Aero Camino, Goleta, CA 93117 www.biopac.com Biopac Student Lab Lesson 5 ELECTROCARDIOGRAPHY (ECG) I Procedure Rev. 07112013 Richard Pflanzer, Ph.D. Associate Professor Emeritus Indiana University School
42 Aero Camino, Goleta, CA 93117 www.biopac.com Biopac Student Lab Lesson 5 ELECTROCARDIOGRAPHY (ECG) I Procedure Rev. 07112013 Richard Pflanzer, Ph.D. Associate Professor Emeritus Indiana University School
Figure 1 The respiratory cycle
 RESPIRATORY CYCLE Introduction Three primary functions of the respiratory system are to provide oxygen for the body's energy needs, provide an outlet for C0 2 and help maintain the ph of the blood plasma.
RESPIRATORY CYCLE Introduction Three primary functions of the respiratory system are to provide oxygen for the body's energy needs, provide an outlet for C0 2 and help maintain the ph of the blood plasma.
Physiology Lessons for use with the Biopac Student Lab. Lesson 1 ELECTROMYOGRAPHY I Standard and Integrated EMG
 Physiology Lessons for use with the Biopac Student Lab Lesson 1 ELECTROMYOGRAPHY I Standard and Integrated EMG Richard Pflanzer, Ph.D. Associate Professor Emeritus Indiana University School of Medicine
Physiology Lessons for use with the Biopac Student Lab Lesson 1 ELECTROMYOGRAPHY I Standard and Integrated EMG Richard Pflanzer, Ph.D. Associate Professor Emeritus Indiana University School of Medicine
Lesson 2 ELECTROMYOGRAPHY II Motor unit recruitment Fatigue
 Physiology Lessons for use with the PC running Windows 98SE, Me, 2000 Pro, XP Pro/Home/Media Lesson 2 ELECTROMYOGRAPHY II Motor unit recruitment Fatigue Manual Revision PL3.7.0 M3.0.6 080706 Richard Pflanzer,
Physiology Lessons for use with the PC running Windows 98SE, Me, 2000 Pro, XP Pro/Home/Media Lesson 2 ELECTROMYOGRAPHY II Motor unit recruitment Fatigue Manual Revision PL3.7.0 M3.0.6 080706 Richard Pflanzer,
Richard Pflanzer, Ph.D. J.C. Uyehara, Ph.D. William McMullen David Pittman, Ph.D. Manual Revision PL3.7.0 M3.0.6 Modified by David Pittman
 ELECTROMYOGRAPHY (EMG) Laboratory Motor unit recruitment & Fatigue Comparison of dominant versus non-dominant arm using a within subject repeated measures statistical analysis Written by: Richard Pflanzer,
ELECTROMYOGRAPHY (EMG) Laboratory Motor unit recruitment & Fatigue Comparison of dominant versus non-dominant arm using a within subject repeated measures statistical analysis Written by: Richard Pflanzer,
Biopac Student Lab Lesson 8 RESPIRATORY CYCLE I Analysis Procedure. Rev
 42 Aero Camino, Goleta, CA 93117 www.biopac.com Biopac Student Lab Lesson 8 RESPIRATORY CYCLE I Analysis Procedure Rev. 12292017 Richard Pflanzer, Ph.D. Associate Professor Emeritus Indiana University
42 Aero Camino, Goleta, CA 93117 www.biopac.com Biopac Student Lab Lesson 8 RESPIRATORY CYCLE I Analysis Procedure Rev. 12292017 Richard Pflanzer, Ph.D. Associate Professor Emeritus Indiana University
Lesson 2 EMG 2 Electromyography: Mechanical Work
 Physiology Lessons for use with the Biopac Science Lab MP40 Lesson 2 EMG 2 Electromyography: Mechanical Work PC running Windows XP or Mac OS X 10.3-10.4 Lesson Revision 5.23.2006 BIOPAC Systems, Inc. 42
Physiology Lessons for use with the Biopac Science Lab MP40 Lesson 2 EMG 2 Electromyography: Mechanical Work PC running Windows XP or Mac OS X 10.3-10.4 Lesson Revision 5.23.2006 BIOPAC Systems, Inc. 42
Biology 13A Lab #10: Cardiovascular System II ECG & Heart Disease
 Biology 13A Lab #10: Cardiovascular System II ECG & Heart Disease Lab #10 Table of Contents: Expected Learning Outcomes...... 83 Introduction....... 84 Activity 1: Collecting ECG Data..... 85 Activity
Biology 13A Lab #10: Cardiovascular System II ECG & Heart Disease Lab #10 Table of Contents: Expected Learning Outcomes...... 83 Introduction....... 84 Activity 1: Collecting ECG Data..... 85 Activity
Biopac Student Lab Lesson 6 ELECTROCARDIOGRAPHY (ECG) II Analysis Procedure. Rev
 42 Aero Camino, Goleta, CA 93117 www.biopac.com Biopac Student Lab Lesson 6 ELECTROCARDIOGRAPHY (ECG) II Analysis Procedure Rev. 12292017 Richard Pflanzer, Ph.D. Associate Professor Emeritus Indiana University
42 Aero Camino, Goleta, CA 93117 www.biopac.com Biopac Student Lab Lesson 6 ELECTROCARDIOGRAPHY (ECG) II Analysis Procedure Rev. 12292017 Richard Pflanzer, Ph.D. Associate Professor Emeritus Indiana University
Experiment HH-3: Exercise, the Electrocardiogram, and Peripheral Circulation
 Experiment HH-3: Exercise, the Electrocardiogram, and Peripheral Circulation Background The arterial system functions as a pressure reservoir. Blood enters via the heart and exits through the capillaries.
Experiment HH-3: Exercise, the Electrocardiogram, and Peripheral Circulation Background The arterial system functions as a pressure reservoir. Blood enters via the heart and exits through the capillaries.
Lesson 4 ECG 2 Electrocardiography
 Physiology Lessons for use with the Biopac Science Lab MP40 Lesson 4 ECG 2 Electrocardiography PC running Windows XP or Mac OS X 10.3-10.4 Lesson Revision 1.20.2006 BIOPAC Systems, Inc. 42 Aero Camino,
Physiology Lessons for use with the Biopac Science Lab MP40 Lesson 4 ECG 2 Electrocardiography PC running Windows XP or Mac OS X 10.3-10.4 Lesson Revision 1.20.2006 BIOPAC Systems, Inc. 42 Aero Camino,
Physiology Lessons for use with the Biopac Student Lab
 Physiology Lessons for use with the Biopac Student Lab Lesson 6 ELECTROCARDIOGRAPHY II Bipolar Leads (Leads I, II, III) Einthoven s Law Mean Electrical Axis on the Frontal Plane Richard Pflanzer, Ph.D.
Physiology Lessons for use with the Biopac Student Lab Lesson 6 ELECTROCARDIOGRAPHY II Bipolar Leads (Leads I, II, III) Einthoven s Law Mean Electrical Axis on the Frontal Plane Richard Pflanzer, Ph.D.
Biopac Student Lab Lesson 3 ELECTROENCEPHALOGRAPHY (EEG) I Analysis Procedure. Rev
 42 Aero Camino, Goleta, CA 93117 www.biopac.com Biopac Student Lab Lesson 3 ELECTROENCEPHALOGRAPHY (EEG) I Analysis Procedure Rev. 12112014 Richard Pflanzer, Ph.D. Associate Professor Emeritus Indiana
42 Aero Camino, Goleta, CA 93117 www.biopac.com Biopac Student Lab Lesson 3 ELECTROENCEPHALOGRAPHY (EEG) I Analysis Procedure Rev. 12112014 Richard Pflanzer, Ph.D. Associate Professor Emeritus Indiana
Biopac Student Lab Lesson 20 SPINAL CORD REFLEXES Analysis Procedure. Rev
 42 Aero Camino, Goleta, CA 93117 www.biopac.com Biopac Student Lab Lesson 20 SPINAL CORD REFLEXES Analysis Procedure Rev. 12292017 Richard Pflanzer, Ph.D. Associate Professor Emeritus Indiana University
42 Aero Camino, Goleta, CA 93117 www.biopac.com Biopac Student Lab Lesson 20 SPINAL CORD REFLEXES Analysis Procedure Rev. 12292017 Richard Pflanzer, Ph.D. Associate Professor Emeritus Indiana University
Exercise 5: Muscle Physiology I - Electromyography
 Exercise 5: Muscle Physiology I - Electromyography Readings: Silverthorn, 6 th ed. pg. 410 420 Your brain communicates with your muscles through action potentials on the motor neurons, which are then transmitted
Exercise 5: Muscle Physiology I - Electromyography Readings: Silverthorn, 6 th ed. pg. 410 420 Your brain communicates with your muscles through action potentials on the motor neurons, which are then transmitted
iworx Sample Lab Experiment HP-7: Hypothesis-driven Biofeedback Lab/Research Study
 Experiment HP-7: Hypothesis-driven Biofeedback Lab/Research Study What is Biofeedback? Biofeedback is a technique that people can use to learn to control their body's physiological functions. The definition
Experiment HP-7: Hypothesis-driven Biofeedback Lab/Research Study What is Biofeedback? Biofeedback is a technique that people can use to learn to control their body's physiological functions. The definition
POLYGRAPH BIOFEEDBACK
 POLYGRAPH BIOFEEDBACK GALVANIC SKIN RESPONSE (GSR) THE POLYGRAPH Polygraph many measures (many poly, graphein to write) - recording at the same time the changes of several biological parameters (usually
POLYGRAPH BIOFEEDBACK GALVANIC SKIN RESPONSE (GSR) THE POLYGRAPH Polygraph many measures (many poly, graphein to write) - recording at the same time the changes of several biological parameters (usually
Cardiovascular Effects of Exercise. Background Cardiac function
 Cardiovascular Effects of Exercise In this experiment, you will record an electrocardiogram, or ECG, and finger pulse from a healthy volunteer. You will then compare the ECG and pulse recordings when the
Cardiovascular Effects of Exercise In this experiment, you will record an electrocardiogram, or ECG, and finger pulse from a healthy volunteer. You will then compare the ECG and pulse recordings when the
Lesson 5 BIOPAC Systems, Inc. Manual Revision PL3.7.5
 Physiology Lessons for use with the Biopac Student Lab Lesson 5 ELECTROCARDIOGRAPHY I Components of the ECG Richard Pflanzer, Ph.D. Associate Professor Emeritus Indiana University School of Medicine Purdue
Physiology Lessons for use with the Biopac Student Lab Lesson 5 ELECTROCARDIOGRAPHY I Components of the ECG Richard Pflanzer, Ph.D. Associate Professor Emeritus Indiana University School of Medicine Purdue
#AH145 - TSD101B Respiratory Effort Transducer
 BIOPAC Systems, Inc. 42 Aero Camino Goleta, CA 93117 Ph (805) 685-0066 Fax (805) 685-0067 www.biopac.com info@biopac.com #AH145 - TSD101B Respiratory Effort Transducer The TSD101B can be used to record
BIOPAC Systems, Inc. 42 Aero Camino Goleta, CA 93117 Ph (805) 685-0066 Fax (805) 685-0067 www.biopac.com info@biopac.com #AH145 - TSD101B Respiratory Effort Transducer The TSD101B can be used to record
Exercise 6: Muscle Physiology II Twitch & Summation
 Exercise 6: Muscle Physiology II Twitch & Summation Text Reading: Silverthorn, 5 th ed. 412 419, 425 427; 6 th ed. pg. 410 420 In this exercise, we will investigate the physiology of contraction in the
Exercise 6: Muscle Physiology II Twitch & Summation Text Reading: Silverthorn, 5 th ed. 412 419, 425 427; 6 th ed. pg. 410 420 In this exercise, we will investigate the physiology of contraction in the
Overview. Page 1 of 9. Impedance Cardiography
 Updated 05.14.10 BSL PRO Lesson H21: Impedance Cardiography Data collected from a subject using the referenced set-up procedure. Note that dz/dt maximum is determined on a cycle by cycle basis from the
Updated 05.14.10 BSL PRO Lesson H21: Impedance Cardiography Data collected from a subject using the referenced set-up procedure. Note that dz/dt maximum is determined on a cycle by cycle basis from the
Lesson 5 EEG 1 Electroencephalography: Brain Rhythms
 Physiology Lessons for use with the Biopac Science Lab MP40 PC running Windows XP or Mac OS X 10.3-10.4 Lesson 5 EEG 1 Electroencephalography: Brain Rhythms Lesson Revision 2.23.2006 BIOPAC Systems, Inc.
Physiology Lessons for use with the Biopac Science Lab MP40 PC running Windows XP or Mac OS X 10.3-10.4 Lesson 5 EEG 1 Electroencephalography: Brain Rhythms Lesson Revision 2.23.2006 BIOPAC Systems, Inc.
Lesson 10 Aerobic Exercise Physiology
 Physiology Lessons for use with the Biopac Science Lab MP40 Lesson 10 Aerobic Exercise Physiology PC running Windows XP or Mac OS X 10.3-10.4 Lesson Revision 12.19.2005 BIOPAC Systems, Inc. 42 Aero Camino,
Physiology Lessons for use with the Biopac Science Lab MP40 Lesson 10 Aerobic Exercise Physiology PC running Windows XP or Mac OS X 10.3-10.4 Lesson Revision 12.19.2005 BIOPAC Systems, Inc. 42 Aero Camino,
MEDIUM-FLOW PNEUMOTACH TRANSDUCER
 MEDIUM-FLOW PNEUMOTACH TRANSDUCER SS11LA for MP3x and MP45 System TSD117 & TSD117-MRI for MP150/MP100 System RX117 Replacement Airflow Head See also: AFT series of accessories for airflow and gas analysis
MEDIUM-FLOW PNEUMOTACH TRANSDUCER SS11LA for MP3x and MP45 System TSD117 & TSD117-MRI for MP150/MP100 System RX117 Replacement Airflow Head See also: AFT series of accessories for airflow and gas analysis
Experiment HH-3: Exercise, the Electrocardiogram, and Peripheral Circulation
 Experiment HH-3: Exercise, the Electrocardiogram, and Peripheral Circulation Exercise 1: The ECG and the Pulse in a Resting Subject Aim: To measure and correlate the ECG and the pulse in a resting individual.
Experiment HH-3: Exercise, the Electrocardiogram, and Peripheral Circulation Exercise 1: The ECG and the Pulse in a Resting Subject Aim: To measure and correlate the ECG and the pulse in a resting individual.
BSL PRO Lesson H03: Nerve Conduction Velocity: Along the Ulnar Nerve of a Human Subject
 Updated 12-22-03 BSL PRO Lesson H03: Nerve Conduction Velocity: Along the Ulnar Nerve of a Human Subject This PRO lesson describes hardware and software setup of the BSL PRO System to record and measure
Updated 12-22-03 BSL PRO Lesson H03: Nerve Conduction Velocity: Along the Ulnar Nerve of a Human Subject This PRO lesson describes hardware and software setup of the BSL PRO System to record and measure
Comparing the Functional Performance of BIOPAC Respiratory Belt-Transducers With and Without Repair Tape
 Comparing the Functional Performance of BIOPAC Respiratory Belt-Transducers With and Without Repair Tape Ames A., Berning M., Chreptowicz N., Hackney R., Kelly E., Noffke A., Lokuta AJ.* *Corresponding
Comparing the Functional Performance of BIOPAC Respiratory Belt-Transducers With and Without Repair Tape Ames A., Berning M., Chreptowicz N., Hackney R., Kelly E., Noffke A., Lokuta AJ.* *Corresponding
SS11LA Pneumotach Transducer
 BIOPAC Systems, Inc. 42 Aero Camino Goleta, CA 93117 Ph (805) 685-0066 Fax (805) 685-0067 www.biopac.com info@biopac.com SS11LA Pneumotach Transducer The SS11LA can be used to measure respiratory flow
BIOPAC Systems, Inc. 42 Aero Camino Goleta, CA 93117 Ph (805) 685-0066 Fax (805) 685-0067 www.biopac.com info@biopac.com SS11LA Pneumotach Transducer The SS11LA can be used to measure respiratory flow
Lab 5: Electromyograms (EMGs)
 Lab 5: Electromyograms (EMGs) Overview A motorneuron and all the muscle fibers that it innervates is known as a motor unit. Under normal circumstances, a neuronal action potential activates all of the
Lab 5: Electromyograms (EMGs) Overview A motorneuron and all the muscle fibers that it innervates is known as a motor unit. Under normal circumstances, a neuronal action potential activates all of the
iworx Sample Lab Experiment HC-3: Pulse Wave Velocity
 Experiment HC-3: Pulse Wave Velocity Exercise 1: The Pulse Wave Velocity at Rest Aim: To measure the pulse wave velocity in a resting individual. Procedure 1. Click on the Record button, located on the
Experiment HC-3: Pulse Wave Velocity Exercise 1: The Pulse Wave Velocity at Rest Aim: To measure the pulse wave velocity in a resting individual. Procedure 1. Click on the Record button, located on the
Experiment HM-11: Electromyograms (EMG) for Paired Arm Wrestling
 Experiment HM-11: Electromyograms (EMG) for Paired Arm Wrestling Background The movement of parts of the body is accomplished through a system of levers composed of skeletal muscles and bones. In a lever,
Experiment HM-11: Electromyograms (EMG) for Paired Arm Wrestling Background The movement of parts of the body is accomplished through a system of levers composed of skeletal muscles and bones. In a lever,
CAMOSUN COLLEGE BIOLOGY 144 (2010) LABS
 LAB 8: CARDIOVASCULAR PHYSIOLOGY PART 1. HEART SOUNDS AND PULSE DETERMINATIONS Introduction Two distinct sounds can be heard during each cardiac cycle. These sounds are commonly described as lub and dup
LAB 8: CARDIOVASCULAR PHYSIOLOGY PART 1. HEART SOUNDS AND PULSE DETERMINATIONS Introduction Two distinct sounds can be heard during each cardiac cycle. These sounds are commonly described as lub and dup
Experiment HM-7: Electromyogram (EMG) Activity in Antagonistic Muscles and Range of Motion
 Experiment HM-7: Electromyogram (EMG) Activity in Antagonistic Muscles and Range of Motion Exercise 1: Antagonistic Muscles in Forearm Aim: To study the EMG activity in muscles that work in opposition
Experiment HM-7: Electromyogram (EMG) Activity in Antagonistic Muscles and Range of Motion Exercise 1: Antagonistic Muscles in Forearm Aim: To study the EMG activity in muscles that work in opposition
Raw BP and Eight Hemodynamic Measurements Displayed in Real Time
 APPLICATION NOTE 42 Aero Camino, Goleta, CA 93117 Tel (805) 685-0066 Fax (805) 685-0067 info@biopac.com www.biopac.com Updated 01.11.2016 Application Note 115: Hemodynamic Measurements Raw BP and Eight
APPLICATION NOTE 42 Aero Camino, Goleta, CA 93117 Tel (805) 685-0066 Fax (805) 685-0067 info@biopac.com www.biopac.com Updated 01.11.2016 Application Note 115: Hemodynamic Measurements Raw BP and Eight
Exercise 2: Effects of Cold Temperature Aim: To record changes in heart rate after the heart is bathed in cold Ringer s solution.
 Experiment AM-3: Heart Muscle Warning: The heart preparation used in this experiment is functional for a limited period of time. If the muscle is bathed periodically in Ringer s solution, it will work
Experiment AM-3: Heart Muscle Warning: The heart preparation used in this experiment is functional for a limited period of time. If the muscle is bathed periodically in Ringer s solution, it will work
#7 - Respiratory System
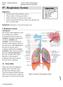 #7 - Respiratory System Objectives: Study the parts of the respiratory system Observe slides of the lung and trachea Perform spirometry to measure lung volumes Define and understand the lung volumes and
#7 - Respiratory System Objectives: Study the parts of the respiratory system Observe slides of the lung and trachea Perform spirometry to measure lung volumes Define and understand the lung volumes and
iworx Sample Lab Experiment AN-5: Cockroach Leg Mechanoreceptors
 Experiment AN-5: Cockroach Leg Mechanoreceptors Exercise 1: Chordotonal Organs Aim: To explore the basic characteristics of the chordotonal organs, their response to direction and intensity of leg movement,
Experiment AN-5: Cockroach Leg Mechanoreceptors Exercise 1: Chordotonal Organs Aim: To explore the basic characteristics of the chordotonal organs, their response to direction and intensity of leg movement,
Experiment HE-12: Targeted Exercise with Wireless Electrocardiogram (ECG)
 Experiment HE-12: Targeted Exercise with Wireless Electrocardiogram (ECG) Get Your Cardio On Preparations for this experiment need to be reviewed with the students in advance. There will be exercise routines
Experiment HE-12: Targeted Exercise with Wireless Electrocardiogram (ECG) Get Your Cardio On Preparations for this experiment need to be reviewed with the students in advance. There will be exercise routines
Experiment HH-2: The Electrocardiogram and Heart Sounds
 Experiment HH-2: The Electrocardiogram and Heart Sounds Exercise 1: The ECG in a Resting Subject Aim: To measure the ECG in a resting individual. Procedure 1. Click on the Record button, located on the
Experiment HH-2: The Electrocardiogram and Heart Sounds Exercise 1: The ECG in a Resting Subject Aim: To measure the ECG in a resting individual. Procedure 1. Click on the Record button, located on the
#4 Cardiovascular I The Heart & EKG
 #4 Cardiovascular I Objectives: Identify a list of human heart structures using a virtual human dissection Dissect a sheep heart to identify external and internal structures Identify a list of human heart
#4 Cardiovascular I Objectives: Identify a list of human heart structures using a virtual human dissection Dissect a sheep heart to identify external and internal structures Identify a list of human heart
1724 Lab: Frog Skeletal Muscle Physiology (Marieb Exercise 16A) Marieb/iWorx / Ziser, 2002
 1724 Lab: Frog Skeletal Muscle Physiology (Marieb Exercise 16A) Marieb/iWorx / Ziser, 2002 I. Introduction. Read the introductory material in your lab manual Marieb Ex 16A: Skeletal Muscle Physiology Frog
1724 Lab: Frog Skeletal Muscle Physiology (Marieb Exercise 16A) Marieb/iWorx / Ziser, 2002 I. Introduction. Read the introductory material in your lab manual Marieb Ex 16A: Skeletal Muscle Physiology Frog
BP-600 Noninvasive Blood Pressure Sensor
 Technical Note BP-600 Overview A person's cardiac output, peripheral vascular resistance, blood pressure, and other cardiovascular parameters change in response to the activities and events taking place
Technical Note BP-600 Overview A person's cardiac output, peripheral vascular resistance, blood pressure, and other cardiovascular parameters change in response to the activities and events taking place
Overview Active Learning
 : EMG Active Learning Updated 01-06-11 This lesson is based on a presentation given by Dr. Jennifer Lundmark and Ms. Andrea Salmi at the HAPS Conference, Maui, HI, June 2-7, 2001 Overview Active Learning
: EMG Active Learning Updated 01-06-11 This lesson is based on a presentation given by Dr. Jennifer Lundmark and Ms. Andrea Salmi at the HAPS Conference, Maui, HI, June 2-7, 2001 Overview Active Learning
Cardiovascular Physiology Laboratory Clive M. Baumgarten, Ph.D.
 Cardiovascular Physiology Laboratory Clive M. Baumgarten, Ph.D. To pump blood, the heart has a rhythmical sequence of both electrical and mechanical events, the cardiac cycle. The electrical activity,
Cardiovascular Physiology Laboratory Clive M. Baumgarten, Ph.D. To pump blood, the heart has a rhythmical sequence of both electrical and mechanical events, the cardiac cycle. The electrical activity,
iworx Sample Lab Experiment HP-1: The Galvanic Skin Response (GSR) and Emotion
 Experiment HP-1: The Galvanic Skin Response (GSR) and Emotion What is a GSR? The Galvanic Skin Response (GSR) is one of several electrodermal responses (EDRs). EDRs are changes in the electrical properties
Experiment HP-1: The Galvanic Skin Response (GSR) and Emotion What is a GSR? The Galvanic Skin Response (GSR) is one of several electrodermal responses (EDRs). EDRs are changes in the electrical properties
Posner s Attention Test
 iworx Physiology Lab Experiment Experiment HP-18 Posner s Attention Test Background Setup Lab Note: The lab presented here is intended for evaluation purposes only. iworx users should refer to the User
iworx Physiology Lab Experiment Experiment HP-18 Posner s Attention Test Background Setup Lab Note: The lab presented here is intended for evaluation purposes only. iworx users should refer to the User
Angular Measurements with BIOPAC Goniometers & Torsiometers
 APPLICATION NOTES 42 Aero Camino, Goleta, CA 93117 Tel (805) 685-0066 Fax (805) 685-0067 info@biopac.com support@.biopac.com Application Note 140 Angular Measurements with BIOPAC Goniometers & Torsiometers
APPLICATION NOTES 42 Aero Camino, Goleta, CA 93117 Tel (805) 685-0066 Fax (805) 685-0067 info@biopac.com support@.biopac.com Application Note 140 Angular Measurements with BIOPAC Goniometers & Torsiometers
Lesson 1: Types of ECG s
 Electrocardiography Lesson 1: Types of ECG s Objectives: Distinguish between a single-channel ECG machine and a multi-channel machine. Distinguish between a manual ECG machine and an automatic ECG machine.
Electrocardiography Lesson 1: Types of ECG s Objectives: Distinguish between a single-channel ECG machine and a multi-channel machine. Distinguish between a manual ECG machine and an automatic ECG machine.
LABORATORY INVESTIGATION
 LABORATORY INVESTIGATION Recording Electrocardiograms The taking of an electrocardiogram is an almost universal part of any complete physical examination. From the ECG record of the electrical activity
LABORATORY INVESTIGATION Recording Electrocardiograms The taking of an electrocardiogram is an almost universal part of any complete physical examination. From the ECG record of the electrical activity
Red Light - Green Light
 iworx Physiology Lab Experiment Experiment HN-9 Red Light - Green Light Background Setup Lab Note: The lab presented here is intended for evaluation purposes only. iworx users should refer to the User
iworx Physiology Lab Experiment Experiment HN-9 Red Light - Green Light Background Setup Lab Note: The lab presented here is intended for evaluation purposes only. iworx users should refer to the User
BC1002 Module 1. Biol BC1002, Spring semester 2005, Module 1, Lab 1-1
 BC1002 Module 1 Biol BC1002, Spring semester 2005, Module 1, Lab 1-1 MODULE 1: LAB 1 MUSCLE PHYSIOLOGY Spring semester 2005, Week of January 24-28. Learning objectives to learn the microscopic structure
BC1002 Module 1 Biol BC1002, Spring semester 2005, Module 1, Lab 1-1 MODULE 1: LAB 1 MUSCLE PHYSIOLOGY Spring semester 2005, Week of January 24-28. Learning objectives to learn the microscopic structure
FT-302 Force Transducer
 Technical Note FT-302 LabScribe is a trademark of 2015 Overview The FT-302 is a high-sensitivity dual-range research grade force transducer designed to measure forces in the 0.005 to 10 gram and 0 to 100
Technical Note FT-302 LabScribe is a trademark of 2015 Overview The FT-302 is a high-sensitivity dual-range research grade force transducer designed to measure forces in the 0.005 to 10 gram and 0 to 100
The Effect of Sympathetic Nervous System Stimulation on Skeletal Muscle Reflex Response Time
 1 The Effect of Sympathetic Nervous System Stimulation on Skeletal Muscle Reflex Response Time Anna Batley, Rebeca Cerda, William Dyke, Celena Ho, Abigail Moeller University of Wisconsin-Madison Physiology
1 The Effect of Sympathetic Nervous System Stimulation on Skeletal Muscle Reflex Response Time Anna Batley, Rebeca Cerda, William Dyke, Celena Ho, Abigail Moeller University of Wisconsin-Madison Physiology
How to install the Wahoo RPM Cadence on your bike: How to install the Wahoo RPM Cadence using the Shoe Clip Mount:
 Setup This setup guide is intended to assist with installing your Wahoo RPM Cadence sensor on your bike or shoe and pairing to the Wahoo Fitness app. How to install the Wahoo RPM Cadence on your bike:
Setup This setup guide is intended to assist with installing your Wahoo RPM Cadence sensor on your bike or shoe and pairing to the Wahoo Fitness app. How to install the Wahoo RPM Cadence on your bike:
Breathing and pulmonary function
 EXPERIMENTAL PHYSIOLOGY EXPERIMENT 5 Breathing and pulmonary function Ying-ying Chen, PhD Dept. of Physiology, Zhejiang University School of Medicine bchenyy@zju.edu.cn Breathing Exercise 1: Tests of pulmonary
EXPERIMENTAL PHYSIOLOGY EXPERIMENT 5 Breathing and pulmonary function Ying-ying Chen, PhD Dept. of Physiology, Zhejiang University School of Medicine bchenyy@zju.edu.cn Breathing Exercise 1: Tests of pulmonary
Experiment HP-28: Control of EEG and Biofeedback Lab/Research Study
 Experiment HP-28: Control of EEG and Biofeedback Lab/Research Study Equipment Required PC or Mac Computer IXTA, USB cable and power supply iwire-b3g cable and EEG leads Reusable Gold-Cup EEG Electrodes
Experiment HP-28: Control of EEG and Biofeedback Lab/Research Study Equipment Required PC or Mac Computer IXTA, USB cable and power supply iwire-b3g cable and EEG leads Reusable Gold-Cup EEG Electrodes
The Gaze Cueing Paradigm with Eye Tracking Background Set-up Lab
 iworx Physiology Lab Experiment Experiment HP-17 The Gaze Cueing Paradigm with Eye Tracking Background Set-up Lab Note: The lab presented here is intended for evaluation purposes only. iworx users should
iworx Physiology Lab Experiment Experiment HP-17 The Gaze Cueing Paradigm with Eye Tracking Background Set-up Lab Note: The lab presented here is intended for evaluation purposes only. iworx users should
Warning: Avoid putting the tip of the syringe needle too deeply into the hole to avoid damage to the heart tissue.
 Experiment AM-12: Crayfish Electrocardiogram (ECG) Exercise 1: The Crayfish ECG and Heart Rate Aim: To record the electrical trace (ECG) produced by the contraction of a resting heart, and to determine
Experiment AM-12: Crayfish Electrocardiogram (ECG) Exercise 1: The Crayfish ECG and Heart Rate Aim: To record the electrical trace (ECG) produced by the contraction of a resting heart, and to determine
Cardiovascular Physiology
 Cardiovascular Physiology The mammalian heart is a pump that pushes blood around the body and is made of four chambers: right and left atria and right and left ventricles. The two atria act as collecting
Cardiovascular Physiology The mammalian heart is a pump that pushes blood around the body and is made of four chambers: right and left atria and right and left ventricles. The two atria act as collecting
Lesson 16 BLOOD PRESSURE
 Physiology Lessons for use with the Lesson 16 BLOOD PRESSURE Indirect measurement Ventricular Systole & Diastole Korotkoff sounds Mean Arterial pressure Manual Revision 3.7.3 061808 Richard Pflanzer, Ph.D.
Physiology Lessons for use with the Lesson 16 BLOOD PRESSURE Indirect measurement Ventricular Systole & Diastole Korotkoff sounds Mean Arterial pressure Manual Revision 3.7.3 061808 Richard Pflanzer, Ph.D.
iworx Sample Lab Experiment HH-4: The Six-Lead Electrocardiogram
 Experiment HH-4: The Six-Lead Electrocardiogram Background The cardiac cycle involves a sequential contraction of the atria and the ventricles. These contractions are triggered by the coordinated electrical
Experiment HH-4: The Six-Lead Electrocardiogram Background The cardiac cycle involves a sequential contraction of the atria and the ventricles. These contractions are triggered by the coordinated electrical
Experiment HP-13: The Gaze Cue Paradigm
 Experiment HP-13: The Gaze Cue Paradigm Background During almost all social interactions, people s eyes convey information about their direction of attention as well as their emotional and mental status.
Experiment HP-13: The Gaze Cue Paradigm Background During almost all social interactions, people s eyes convey information about their direction of attention as well as their emotional and mental status.
Quick Notes. BioCapture : Acquiring Respiratory Effort data
 BioCapture : Acquiring Respiratory Effort data Respiratory effort, or plethysmography, is the measurement of rhythmic fluctuation of the chest and/or abdomen. There are only a few primary methods of measuring
BioCapture : Acquiring Respiratory Effort data Respiratory effort, or plethysmography, is the measurement of rhythmic fluctuation of the chest and/or abdomen. There are only a few primary methods of measuring
Living with Newton's Laws
 Task #1 - Newton s 1 st Law - This is a pain in the neck Let's suppose you are in your car, waiting at a stop light. Like any good driver, you have your seat belt buckled. (It's the law.) Suddenly, a car
Task #1 - Newton s 1 st Law - This is a pain in the neck Let's suppose you are in your car, waiting at a stop light. Like any good driver, you have your seat belt buckled. (It's the law.) Suddenly, a car
Lab 4: Introduction to Physiological Measurements - Cardiovascular
 Lab 4: Introduction to Physiological Measurements - Cardiovascular INTRODUCTION: This lab will demonstrate cardiovascular measurements by creating an ECG with instruments used in previous labs. Students
Lab 4: Introduction to Physiological Measurements - Cardiovascular INTRODUCTION: This lab will demonstrate cardiovascular measurements by creating an ECG with instruments used in previous labs. Students
Exercise 1: Eye-Hand Reaction Times Aim: To measure the reaction time of a subject to a visual cue when responding with the hand.
 Experiment HN-4: Hand vs. Foot Reactions Exercise 1: Eye-Hand Reaction Times Aim: To measure the reaction time of a subject to a visual cue when responding with the hand. Procedure 1. Instruct the subject
Experiment HN-4: Hand vs. Foot Reactions Exercise 1: Eye-Hand Reaction Times Aim: To measure the reaction time of a subject to a visual cue when responding with the hand. Procedure 1. Instruct the subject
Experiment HC-1: Blood Pressure, Peripheral Circulation, and Body Position
 Experiment HC-1: Blood Pressure, Peripheral Circulation, and Body Position Exercise 1: Blood Pressures from the Left Arm Aim: To determine the systolic and diastolic blood pressures in a reclining subject,
Experiment HC-1: Blood Pressure, Peripheral Circulation, and Body Position Exercise 1: Blood Pressures from the Left Arm Aim: To determine the systolic and diastolic blood pressures in a reclining subject,
Evolve 3 & 5 Service Manual
 Evolve 3 & 5 Service Manual 1 Product Browse 2 Contents CHAPTER 1: SERIAL NUMBER LOCATION... 5 CHAPTER 2: CONSOLE INSTRUCTIONS 2.1 Console Overview... 6 2.1.1 Evolve 3 Console Overview... 6 2.1.2 Evolve
Evolve 3 & 5 Service Manual 1 Product Browse 2 Contents CHAPTER 1: SERIAL NUMBER LOCATION... 5 CHAPTER 2: CONSOLE INSTRUCTIONS 2.1 Console Overview... 6 2.1.1 Evolve 3 Console Overview... 6 2.1.2 Evolve
Stress and its vegetative symptoms can be greatly reduced through biofeedback training, where you intentionally work on lowering your skin response.
 Skin response Skin response, also known as galvanic skin response (GSR), electrodermal response (EDR) or skin conductance (SC) is a measurement method based on bio-electrical properties of the skin. The
Skin response Skin response, also known as galvanic skin response (GSR), electrodermal response (EDR) or skin conductance (SC) is a measurement method based on bio-electrical properties of the skin. The
2.0. Desktop Fitting Guide getting started. Preparation of the hearing instruments
 Desktop Fitting Guide getting started This Guide provides you a detailed introduction to hearing instrument fitting with Phonak Target 2.0. Please also find the [News] in the Phonak Target start screen.
Desktop Fitting Guide getting started This Guide provides you a detailed introduction to hearing instrument fitting with Phonak Target 2.0. Please also find the [News] in the Phonak Target start screen.
Lab #3: Electrocardiogram (ECG / EKG)
 Lab #3: Electrocardiogram (ECG / EKG) An introduction to the recording and analysis of cardiac activity Introduction The beating of the heart is triggered by an electrical signal from the pacemaker. The
Lab #3: Electrocardiogram (ECG / EKG) An introduction to the recording and analysis of cardiac activity Introduction The beating of the heart is triggered by an electrical signal from the pacemaker. The
Bio-logic AuDX PRO FLEX Screening tympanometry and audiometry Quick start guide
 AuDX PRO FLEX Screening tympanometry and audiometry Quick start guide Doc. No.7-26-1374-EN/00 Part No.7-26-13704-EN Natus Medical Denmark ApS. +45 45 75 55 55. otoinfo@natus.com Natus Medical Incorporated.
AuDX PRO FLEX Screening tympanometry and audiometry Quick start guide Doc. No.7-26-1374-EN/00 Part No.7-26-13704-EN Natus Medical Denmark ApS. +45 45 75 55 55. otoinfo@natus.com Natus Medical Incorporated.
Adventure 4 Plus Service Manual
 Adventure 4 Plus Service Manual 1 TABLE OF CONTENTS CHAPTER 1: SERIAL NUMBER LOCATION...3 CHAPTER 2: PREVENTATIVE MAINTENANCE 2.1 Preventative Maintenance. 4 2.2 Tension and Centering the Running Belt....6
Adventure 4 Plus Service Manual 1 TABLE OF CONTENTS CHAPTER 1: SERIAL NUMBER LOCATION...3 CHAPTER 2: PREVENTATIVE MAINTENANCE 2.1 Preventative Maintenance. 4 2.2 Tension and Centering the Running Belt....6
Thank you very much for agreeing to participate in the Ontario Sleep Health Study
 Thank you very much for agreeing to participate in the Ontario Sleep Health Study By participating in this study, you are playing a key role in helping us better understand the impact of our genes on our
Thank you very much for agreeing to participate in the Ontario Sleep Health Study By participating in this study, you are playing a key role in helping us better understand the impact of our genes on our
The following describes general use of ELIOS device
 General use of the device The following describes general use of ELIOS device 1. Introduction of graphical user interface Presence indicator of SD card Charging indicator General Informations Screen title
General use of the device The following describes general use of ELIOS device 1. Introduction of graphical user interface Presence indicator of SD card Charging indicator General Informations Screen title
QUICK START GUIDE TYMPANOMETRY AND AUDIOMETRY FOR SENTIERO DESKTOP DEVICES
 DEVICE CONNECTORS On/off button Headphones or second transducer Power supply Air pump USB cable Label printer Patient response Probe button Make sure all your appropriate transducers are connected (Tymp
DEVICE CONNECTORS On/off button Headphones or second transducer Power supply Air pump USB cable Label printer Patient response Probe button Make sure all your appropriate transducers are connected (Tymp
Fujitsu LifeBook T Series TabletPC Voluntary Product Accessibility Template
 Fujitsu LifeBook T Series TabletPC Voluntary Product Accessibility Template 1194.21 Software Applications and Operating Systems* (a) When software is designed to run on a system that This product family
Fujitsu LifeBook T Series TabletPC Voluntary Product Accessibility Template 1194.21 Software Applications and Operating Systems* (a) When software is designed to run on a system that This product family
Thank you very much for agreeing to participate in the Ontario Sleep and Brain Health Study
 Thank you very much for agreeing to participate in the Ontario Sleep and Brain Health Study By participating in this study, you will play a key role in helping us better understand the links between sleep
Thank you very much for agreeing to participate in the Ontario Sleep and Brain Health Study By participating in this study, you will play a key role in helping us better understand the links between sleep
Blood Pressure Laboratory
 Introduction The blood that circulates throughout the body maintains a flow and pressure. The nervous system can change the flow and pressure based on the particular needs at a given time. For example,
Introduction The blood that circulates throughout the body maintains a flow and pressure. The nervous system can change the flow and pressure based on the particular needs at a given time. For example,
Lab #3: Electrocardiogram (ECG / EKG)
 Lab #3: Electrocardiogram (ECG / EKG) An introduction to the recording and analysis of cardiac activity Introduction The beating of the heart is triggered by an electrical signal from the pacemaker. The
Lab #3: Electrocardiogram (ECG / EKG) An introduction to the recording and analysis of cardiac activity Introduction The beating of the heart is triggered by an electrical signal from the pacemaker. The
Adventure 5 Plus Service Manual
 Adventure 5 Plus Service Manual 1 TABLE OF CONTENTS CHAPTER 1: SERIAL NUMBER LOCATION...3 CHAPTER 2: PREVENTATIVE MAINTENANCE 2.1 Preventative Maintenance. 4 2.2 Tension and Centering the Running Belt....6
Adventure 5 Plus Service Manual 1 TABLE OF CONTENTS CHAPTER 1: SERIAL NUMBER LOCATION...3 CHAPTER 2: PREVENTATIVE MAINTENANCE 2.1 Preventative Maintenance. 4 2.2 Tension and Centering the Running Belt....6
Skeletal muscles are composed of hundreds to thousands of individual cells,
 2 E X E R C I S E Skeletal Muscle Physiology O B J E C T I V E S 1. To define these terms used in describing muscle physiology: multiple motor unit summation, maximal stimulus, treppe, wave summation,
2 E X E R C I S E Skeletal Muscle Physiology O B J E C T I V E S 1. To define these terms used in describing muscle physiology: multiple motor unit summation, maximal stimulus, treppe, wave summation,
N4i Installation Manual. (picture of N4i screen)
 N4i Installation Manual (picture of N4i screen) 1 Table of Contents N4i Computer Parts List... 4 Netpulse Stabilizer Bar Parts List... 6 Hardware List... 7 Tools Needed for Installation... 7 N4i Assembly
N4i Installation Manual (picture of N4i screen) 1 Table of Contents N4i Computer Parts List... 4 Netpulse Stabilizer Bar Parts List... 6 Hardware List... 7 Tools Needed for Installation... 7 N4i Assembly
Metabolic and Thermal Response to Exercise
 iworx Physiology Lab Experiment Experiment HE-1 Metabolic and Thermal Response to Exercise Note: The lab presented here is intended for evaluation purposes only. iworx users should refer to the User Area
iworx Physiology Lab Experiment Experiment HE-1 Metabolic and Thermal Response to Exercise Note: The lab presented here is intended for evaluation purposes only. iworx users should refer to the User Area
Neuro-Audio Version 2010
 ABR PTA ASSR Multi-ASSR OAE TEOAE DPOAE SOAE ECochG MLR P300 Neuro-Audio Version 2010 one device for all audiological tests Auditory brainstem response (ABR)/Brainstem evoked response audiometry (BERA)
ABR PTA ASSR Multi-ASSR OAE TEOAE DPOAE SOAE ECochG MLR P300 Neuro-Audio Version 2010 one device for all audiological tests Auditory brainstem response (ABR)/Brainstem evoked response audiometry (BERA)
Adventure 3 Plus Service Manual
 Adventure 3 Plus Service Manual 1 TABLE OF CONTENTS CHAPTER 1: SERIAL NUMBER LOCATION...3 CHAPTER 2: PREVENTATIVE MAINTENANCE 2.1 Preventative Maintenance. 4 2.2 Tension and Centering the Running Belt....6
Adventure 3 Plus Service Manual 1 TABLE OF CONTENTS CHAPTER 1: SERIAL NUMBER LOCATION...3 CHAPTER 2: PREVENTATIVE MAINTENANCE 2.1 Preventative Maintenance. 4 2.2 Tension and Centering the Running Belt....6
HealthCare International, Inc. PO Box 1509, Langley, WA P: (360) or (800)
 Owner s Manual V2.2 HealthCare International, Inc. PO Box 1509, Langley, WA 98260 www.hcifitness.com sales@hcifitness.com P: (360) 321-7090 or (800) 398-9121 1 Table of Contents Safety Precautions... 3
Owner s Manual V2.2 HealthCare International, Inc. PO Box 1509, Langley, WA 98260 www.hcifitness.com sales@hcifitness.com P: (360) 321-7090 or (800) 398-9121 1 Table of Contents Safety Precautions... 3
BLINDED MODE USERS GUIDE SUPPLEMENT
 BLINDED MODE USERS GUIDE SUPPLEMENT BLINDED MODE user s guide SUPPLEMENT WARNING: The SEVEN PLUS when set to Blinded Mode using the Data Manager Software will not provide real-time continuous glucose readings,
BLINDED MODE USERS GUIDE SUPPLEMENT BLINDED MODE user s guide SUPPLEMENT WARNING: The SEVEN PLUS when set to Blinded Mode using the Data Manager Software will not provide real-time continuous glucose readings,
ELECTROMYOGRAM ANALYSIS OF MUSCLE FUNCTION INTRODUCTION
 ELECTROMYOGRAM ANALYSIS OF MUSCLE FUNCTION STANDARDS: 3.3.10.B - Explain cell functions and processes in terms of chemical reactions and energy changes. 3.3.12.B - Evaluate relationships between structure
ELECTROMYOGRAM ANALYSIS OF MUSCLE FUNCTION STANDARDS: 3.3.10.B - Explain cell functions and processes in terms of chemical reactions and energy changes. 3.3.12.B - Evaluate relationships between structure
New User FAQs. Myzone, Heart Rate Zones & The MZ-3
 New User FAQs Myzone, Heart Rate Zones & The MZ-3 What is Myzone? Myzone is a chest strap and monitoring system that displays heart rate, calories, time and effort to a TV or monitor or to the Myzone App,
New User FAQs Myzone, Heart Rate Zones & The MZ-3 What is Myzone? Myzone is a chest strap and monitoring system that displays heart rate, calories, time and effort to a TV or monitor or to the Myzone App,
Experiment B-18 Heart Rate and Coughing
 1 Experiment B-18 Heart Rate and Coughing Objectives To study the changes in heart rate at rest and after coughing. Modules and Sensors PC + NeuLog application USB-200 module NUL-208 Heart rate & pulse
1 Experiment B-18 Heart Rate and Coughing Objectives To study the changes in heart rate at rest and after coughing. Modules and Sensors PC + NeuLog application USB-200 module NUL-208 Heart rate & pulse
Iso-inertial dynamometer
 Iso-inertial dynamometer A. Equipment set up B. Data acquisition and export C. Training mode 1 P a g e A. Equipment set up 1. Connect all the devices as depicted below: 2. Launch the software by clicking
Iso-inertial dynamometer A. Equipment set up B. Data acquisition and export C. Training mode 1 P a g e A. Equipment set up 1. Connect all the devices as depicted below: 2. Launch the software by clicking
Titronics Range of Motion Hardware & Software Installation
 Titronics Range of Motion Hardware & Software Installation Step 1 Titronics ROM Software Installation Insert the Titronics Flash Drive into a USB port. Open the Flash Drive and click on Titronics.exe Click
Titronics Range of Motion Hardware & Software Installation Step 1 Titronics ROM Software Installation Insert the Titronics Flash Drive into a USB port. Open the Flash Drive and click on Titronics.exe Click
