Lesson 2 ELECTROMYOGRAPHY II Motor unit recruitment Fatigue
|
|
|
- Janel Dalton
- 5 years ago
- Views:
Transcription
1 Physiology Lessons for use with the PC running Windows 98SE, Me, 2000 Pro, XP Pro/Home/Media Lesson 2 ELECTROMYOGRAPHY II Motor unit recruitment Fatigue Manual Revision PL3.7.0 M Richard Pflanzer, Ph.D. Associate Professor Indiana University School of Medicine Purdue University School of Science J.C. Uyehara, Ph.D Biologist BIOPAC Systems, Inc. William McMullen Vice President BIOPAC Systems, Inc. BIOPAC Systems, Inc. 42 Aero Camino, Goleta, CA (805) , Fax (805) info@biopac.com Web: BIOPAC Systems, Inc. 2004
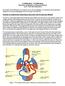
2 Page 2 I. INTRODUCTION Mechanical work, in the physical sense, refers to the application of a force that results in the movement of an object. Skeletal muscle performs mechanical work when the muscle contracts and an object is moved, as in lifting a weight. To lift a weight, your muscles must exert a force great enough to overcome the weight. If you exert less force, then the weight does not move (Fig. 2.1). Physiologically, skeletal muscle is stimulated to contract when the brain or spinal cord activates motor units of the muscle. Motor units are defined as a motoneuron and all of the muscle fibers that the motoneuron innervates. Fig. 2.1 An action potential (AP) in a human motoneuron always causes an action potential in all of the muscle fibers of the motor unit. As a matter of fact, humans generally do not send just one AP at a time down a motoneuron. Instead, a train of APs is sent enough to induce tetany (the sustained fusion of individual muscle twitches) in the muscle fibers of the motor unit. [A discussion of motor units and their control was presented in Lesson 1.] Most human skeletal muscles are composed of hundreds of motor units (Fig. 2.2). When a skeletal muscle is called on to perform mechanical work, the number of motor units in the muscle activated by the brain is proportional to the amount of work to be done by the muscle; the greater the amount of work to be done, the greater the number of motor units activated. Thus, more motor units are simultaneously active when a skeletal muscle lifts 20 kilograms than when the same muscle lifts 5 kilograms. Fig. 2.2 Example of Motor Units The brain determines the number of active motor units required for a muscle to perform a given task by utilizing sensory information from stretch receptors in the muscle and associated tendons and joint capsules. For example, when lifting a bucket of water from the ground, the brain first activates several motor units in the requisite skeletal muscles. If sensory information returning from the muscles indicates the muscles are contracting but not developing adequate power to lift the bucket, the brain activates additional motor units until the sensory information indicates the bucket is being lifted. The sequential activation of motor units to perform a designated task is called motor unit recruitment. Once you have lifted a light object, the brain recruits approximately the same number of motor units to keep the object up, but cycles between different motor units. The muscle fibers consume stored energy available in the muscle, and generate a force by contracting. As the muscle fibers deplete this fuel source, more energy must be created in order to continue contracting. By recruiting different motor units, motor units can relax and replenish their fuel sources.
3 Lesson 2: EMG II Page 3 Skeletal muscles performing acute maximum work or chronic submaximum work of a repetitive nature will eventually fatigue. Fatigue is defined as a decrease in the muscle s ability to generate force. Fatigue is caused by a reversible depletion of the muscle s fuel supply. If the muscle uses its energy sources faster than they can be generated by cellular metabolism, fatigue occurs. During contraction, skeletal muscle cells convert chemical energy into thermal and mechanical energy, and, in the process, produce chemical waste products. Normally the waste products are removed from the muscle by the circulatory system as the blood brings nutrients to the muscle for energy transformation. If certain waste products (metabolites) are not removed at an adequate rate, they will accumulate and chemically interfere with the contractile process, thereby hastening the onset of fatigue. Some accumulated waste products also stimulate pain receptors in surrounding connective tissue and induce cramping of skeletal muscle, a general sign of inadequate blood flow to the muscle. In this lesson, you will examine motor unit recruitment and skeletal muscle fatigue by combining electromyography with dynamometry. When a motor unit is activated, the component muscle fibers generate and conduct their own electrical impulses, which cause the fibers to contract. Although the electrical impulse generated and conducted by each fiber is very weak (less than 100 μvolts), many fibers conducting simultaneously induce voltage differences in the overlying skin which are large enough to be detected by a pair of surface electrodes. The detection, amplification, and recording of changes in skin voltage produced by underlying skeletal muscle contraction is called electromyography, and the recording thus obtained is called an electromyogram (EMG). Power is defined as the amount of work done per unit of time. Dynamometry means the measurement of power (dyno = power, meter = measure), and the graphic record derived from the use of a dynamometer is called a dynagram. In this lesson, the power of contraction of clench muscles will be determined by a hand dynamometer equipped with an electronic transducer for recording. The BIOPAC system will simultaneously record three bands of information: 1) The force you exert on the transducer, 2) The electrical signal produced by the muscle during contraction, and 3) The integrated waveform, which is an indication of the activity levels of the muscle. II. EXPERIMENTAL OBJECTIVES 1) To determine the maximum clench strength for right and left hands and compare differences between male and female. 2) To observe, record, and correlate motor unit recruitment with increased power of skeletal muscle contraction. 3) To record the force produced by clench muscles, EMG, and integrated EMG when inducing fatigue. III. MATERIALS BIOPAC Hand Dynamometer (SS25LA or SS25L) BIOPAC Headphones (OUT1) BIOPAC electrode lead set (SS2L) BIOPAC disposable vinyl electrodes (EL503), 6 electrodes per subject BIOPAC electrode gel (GEL1) and abrasive pad (ELPAD) or Skin cleanser or Alcohol prep Computer system software PC or Mac or greater BIOPAC acquisition unit (MP35 or MP35/MP30) with associated cables BIOPAC wall transformer (AC300 or AC100A)
4 Page 4 IV. EXPERIMENTAL METHODS For further explanation, use the online support options under the Help Menu. A. SET UP FAST TRACK SET UP Detailed Explanation 1. Turn your computer ON. The desktop should appear on the monitor. If it does not appear, ask the laboratory instructor for assistance. 2. Make sure the BIOPAC MP35/MP30 unit is OFF. 3. Plug the equipment in as follows: Hand Dynamometer (SS25LA or SS25L) CH 1 Electrode lead Set (SS2L) CH 3 Headphones (OUT1) back of unit Headphones (BIOPAC OUT1) plug into back of MP35/30 unit 4. Turn ON the BIOPAC MP35/MP30 unit. 5. Attach three electrodes to each forearm (Fig. 2.4). 6. Attach the electrode lead set (SS2L) to the Subject s dominant forearm, following the color code (Fig. 2.4). IMPORTANT Make sure the electrode lead colors match Fig Lead Set BIOPAC SS2L plugs into CHannel 3 Hand Dynamometer (BIOPAC SS25LA or SS25L shown) plugs Fig. 2.3 Equipment Setup Attach three electrodes to each forearm as shown in Fig For optimal electrode adhesion, place electrodes on the skin at least 5 minutes before the start of Calibration. Attach the lead set (SS2L) to the Subject s dominant forearm (Fig. 2.4) for recording Segments 1 and 2 (forearm 1). If the subject is right-handed, the right forearm is generally dominant; if the subject is left-handed, the left forearm is generally dominant. You will switch the lead set to the Subject s non-dominant arm for recording Segments 3 and 4 (Forearm 2). White Lead (-) Red Lead (+) Black Lead (Ground) Set Up continues
5 Lesson 2: EMG II Page 5 Fig. 2.4 Electrode Placement & Lead Attachment 7. Start the BIOPAC Student Lab Program. 8. Choose lesson L02-EMG-2 and click OK. The electrode lead cables are each a different color and each must be attached to a specific electrode position, as shown in Fig The pinch connectors work like a small clothespin and will only latch onto the nipple of the electrode from one side of the connector. 9. Type in a unique filename. No two people can have the same filename, so use a unique identifier, such as the subject s nickname or student ID#. 10. Click OK. This ends the Set Up procedure. END OF SET UP
6 Page 6 B. CALIBRATION The Calibration procedure establishes the hardware s internal parameters (such as gain, offset, and scaling) and is critical for optimum performance. Pay close attention to the Calibration procedure. FAST TRACK CALIBRATION Detailed Explanation of Steps for Calibration 1. Click Calibrate. A pop-up window will tell you to remove any grip force from the hand dynamometer. To do so, set the hand dynamometer down. 2. Set the hand dynamometer down and click OK. Remove your hands from the transducer to ensure there is no force on the transducer. This establishes a zero-force calibration before you continue the calibration sequence. 3. Grasp the BIOPAC hand dynamometer with your hand. SS25LA: Place the short grip bar against the palm, toward the thumb, and wrap your fingers to center the force. SS25L: Grasp as close to the dynagrip crossbar as possible without actually touching the crossbar (Fig. 2.6). Clench with the hand of your dominant forearm. Hand close to bracket but not touching. Dynagrip crossbar IMPORTANT Hold the dynamometer in the same position for all measurements from each arm. Note your hand position for the first segment and try to repeat it for the other segments. SS25LA grip position Fig. 2.5 SS25L grip position 4. Follow the instructions in the successive pop-up windows and click OK when ready for each. 5. When Calibration recording begins, wait about two seconds, then clench the hand dynamometer as hard as possible, and then release. Pop-up windows will guide you through the initial calibration sequence. After you click OK on the third pop-up window, the Calibration recording will begin. The program needs a reading of your maximum clench to perform an auto-calibration. 6. Wait for Calibration to stop. The Calibration procedure will last eight seconds and stop automatically, so let it run its course. 7. Check the Calibration data. After the Calibration recording, the screen should resemble Fig.2.6. If correct, proceed to the data Recording Section. If incorrect, Redo Calibration. Fig. 2.6 END OF CALIBRATION If your recording does not begin with a zero baseline (Subject clenched before two seconds), repeat calibration to match Fig. 2.6.
7 Lesson 2: EMG II Page 7 C. RECORDING LESSON DATA FAST TRACK RECORDING Detailed Explanation of Steps for Recording Lesson Data 1. Prepare for the recording. In order to work efficiently, read this entire section so you will know what to do before recording. You will record two segments on each forearm: a. Segments 1 and 3 record Motor unit recruitment. b. Segments 2 and 4 record Fatigue SEGMENT 1 Check the last line of the journal and note the total amount of time available for the recording. Stop each recording segment as soon as possible so you don t waste recording time (time is memory). 2. Note your Assigned Increment level from the journal. Based on your calibrated grip force, the software determines the optimal grid display and force increments. Check the journal and use the indicated increment when directed to increase force throughout the recording. The grid display is set as follows: Force calibration Assigned Increment 0-25 kg 5kg kg 10 kg > 50 kg 20 kg 3. Click Record. After you click Record, the recording will begin and an append marker labeled Forearm 1, Increasing clench force will automatically be inserted. The screen will change to display only the hand dynamometer channel, and a grid using your assigned increment as a division scale will appear so that you can visually review the force level. 4. Clench-Release-Wait and repeat with increasing clench force. Repeat a cycle of Clench-Release-Wait, holding for 2 seconds and waiting for two seconds after releasing before beginning the next cycle. Begin with your Assigned Increment of force (5, 10 or 20 kg) and increase by the Assigned Increment ( , , or for example) for each cycle until maximum clench force is obtained. 5. Click Suspend. The recording should halt, giving you time to review the data and prepare for the next recording segment of Forearm Review the data on the screen. If correct, go to Step 8. If all went well, your data should look similar to Fig The important aspect for you to review is that the data shows multiple peaks (indicating the clench cycles). The data shown is from a Subject who was able to maintain an even force throughout the clench. Your data may be correct even if your peaks are not smooth. Recording continues Fig. 2.7 Motor Unit Recruitment
8 Page 8 If incorrect, go to Step 7. The data would be incorrect if the: a. Suspend was clicked prematurely. b. Instructions were not followed. 7. Click Redo if your data was incorrect and repeat Steps 3-6. Click Redo and repeat Steps 3-6 if necessary. Note that once you click Redo, the data you have just recorded will be erased. SEGMENT 2 8. Click Resume. When you click Resume, the recording will continue and an append marker labeled Forearm 1, Continued clench at maximum force will be automatically inserted. 9. Clench the hand dynamometer with your maximum force. Note this force and try to maintain it. 10. When the maximum clench force displayed on the screen has decreased by more than 50%, click Suspend. 11. Review the data on the screen. Note the maximum clench force so you can determine when the force has decreased by 50% (the maximum force may scroll out of view). Try to maintain the maximum clench force (the forearm will fatigue and the force will decrease). The time to fatigue to 50% of maximal clench force will vary greatly among individuals. When you click Suspend, the recording should halt, giving you time to review the data for segment two. If all went well, your data should look similar to Fig If correct, go Step 13. Fig. 2.8 Fatigue Note that the peak found immediately following the start of the Segment represents the maximal clench force. This example shows the point of fatigue to 50% maximal clench force captured on the same screen, but your maximum force may scroll out of view. You may use the horizontal (time) scroll bar to view your entire recording. If incorrect, go to Step Click Redo if your data was incorrect, and repeat Steps Connect the lead set (SS2L) to the Subject s non-dominant forearm and click OK. The data would be incorrect if: a) You didn t record to the point of 50% maximal clench force. b) Suspend was clicked prematurely. c) The instructions were not followed. Click Redo and have the Subject rest so the arm muscles recover and the fatigue data will be meaningful. When ready repeat Steps Note that once you click Redo, the data you have just recorded will be erased. Connect the lead set (SS2L) to the Subject s non-dominat forearm for the next recording segment. Click OK. Recording continues
9 Lesson 2: EMG II Page 9 SEGMENT To record Forearm 2, connect to the electrodes that are attached (per Set Up Step 5) to the Subject s non-dominant forearm. IMPORTANT Make sure the electrode lead colors match Fig In Segments 3 and 4, you will record Forearm 2, the nondominant forearm, following the same procedure you used for Forearm 1. Disconnect the lead set (SS2L) from the electrodes on Forearm 1 and connect to the electrodes on Forearm 2 per Fig. 2.9 If you have not already attached electrodes to Forearm 2, refer to Set Up Step 5 for proper placement. White Lead (-) Red Lead (+) Black Lead (Ground) 14. Note your Assigned Increment level from the journal. Fig. 2.9 Electrode lead attachment Based on your calibrated grip force, the software determines the optimal grid display and force increments. Check the journal and use the indicated increment when directed to increase force throughout the recording. 15. Click Resume. When you click Resume, the recording will continue and an append marker labeled Forearm 2, Increasing Clench Force will be automatically inserted. Note that the hand dynamometer channel grid uses your assigned increment as a division scale so that you can visually review the force level. 16. Clench-Release-Wait and repeat with increasing clench force. Repeat a cycle of Clench-Release-Wait, holding for 2 seconds and waiting for two seconds after releasing before beginning the next cycle. Begin with your Assigned Increment of force (5, 10 or 20 kg) and increase by the Assigned Increment ( , , or for example) for each cycle until maximum clench force is obtained. 17. Click Suspend. The recording should halt, giving you time to review the data and prepare for the next recording segment. 18. Review the data on the screen. If correct, go to Step 21. If incorrect, go to Step Click Redo if your data was incorrect and repeat Steps If all went well, your data should look similar to Fig The data would be incorrect if the: a. Suspend was clicked prematurely. b. Instructions were not followed. Click Redo and repeat Steps if necessary. Note that once you click Redo, the data you have just recorded will be erased. Recording continues
10 Page 10 SEGMENT Click Resume. When you click Resume, the recording will continue and an append marker labeled Forearm 2, Continued clench at maximum force will be automatically inserted. 21. Clench the hand dynamometer with your maximum force. Note this force and try to maintain it. Note the maximum clench force so you can determine when the force has decreased by 50% (the maximum force may scroll out of view). Try to maintain the maximum clench force (the forearm will fatigue and the force will decrease). 22. When the maximum clench force displayed on the screen has decreased by more than 50%, click Suspend. 23. Review the data on the screen. The time to fatigue to 50% of maximal clench force will vary greatly among individuals. When you click Suspend, the recording should halt, giving you time to review the data from the last recording segment. If all went well, your data should look similar to Fig If correct, go Step 26. If incorrect, go to Step 25. The data would be incorrect if: a) You didn t record to the point of 50% maximal clench force. b) Suspend was clicked prematurely. c) The instructions were not followed. 24. Click Redo if your data was incorrect, and repeat Steps Click Redo and have the Subject rest so the arm muscles recover and the fatigue data will be meaningful. When ready, repeat Steps Note that once you click Redo, the data you have just recorded will be erased. 25. Click Stop. When you click Stop, a dialog box is generated, asking if you are finished with both Forearm recordings. This is simply one last chance to confirm that you don t need to redo the last recording segment. Click Yes to end data recording and automatically save the data. Click No to return to a display with a Redo option. 26. If you want to listen to the EMG signal, go to Step 28. or Listening to the EMG is optional. Listening to the EMG can be a valuable tool in detecting muscle abnormalities, and is performed here for general interest. To skip listening and end the recording, go to Step Subject puts on the headphones. Note: When you click Listen in the next step, the volume through the headphones may be very loud due to system feedback. The volume cannot be adjusted, but you can position the headphones slightly off the ear to reduce the sound. 28. Click Listen. Recording continues
11 Lesson 2: EMG II Page As you listen, try a variety of Clench- Release Wait cycles and watch how the data changes on the screen. You will hear the EMG signal through the headphones as it is being displayed on the screen. The screen will display three channels: CH 1 Force, CH 3 EMG, and CH 40 Integrated EMG. The data on the screen will not be saved. The signal will run until you click Stop. If others in your lab group would like to hear the EMG signal, pass the headphones around before clicking Stop. 30. Click Stop. This will end listening to the EMG. 31. To listen again, click Redo. Another person may want to listen to the EMG. 32. Click Done. A pop-up window with four options will appear. Make your choice, and continue as directed. 33. Remove the electrodes from each forearm. END OF RECORDING If choosing the Record from another subject option: a) Attach electrodes per Set Up Step 5 and continue the entire lesson from Set Up Step 8. b) Each person will need to use a unique file name. Remove the electrode cable pinch connectors, and peel off the electrodes. Throw out the electrodes (BIOPAC electrodes are not reusable). Wash the electrode gel residue from the skin, using soap and water. The electrodes may leave a slight ring on the skin for a few hours, which is quite normal.
12 Page 12 V. DATA ANALYSIS FAST TRACK DATA ANALYSIS 1. Enter the Review Saved Data mode and choose the correct file. Note Channel Number (CH) designations: Channel Displays CH 1 CH 3 CH 40 Force EMG (raw data Integrated EMG Analysis of Increasing Clench Force 2. Setup your display for optimal viewing of Segment 1 data. 3. Read the journal and note your force increment in the Data Report. A 4. Set up the measurement boxes as follows: Channel Measurement CH 1 mean CH 3 p-p CH 40 mean Data Analysis continues Detailed Explanation of Data Analysis Steps Enter Review Saved Data from the Lessons menu. For the first part of the analysis, use the data labeled Increasing clench force recorded in Segment 1 for Forearm 1 and Segment 3 for Forearm 2. Note your findings in Table 2.1. For the second part of the analysis, use the data labeled Continued clench at maximum force recorded in Segment 2 for Forearm 1 and Segment 4 for Forearm 2. Note your findings in Table 2.2. Fig The first data segment was the recording before the first marker. See Fig The following tools help you adjust the data window: Autoscale horizontal Horizontal(Time) Scroll Bar Autoscale waveforms Vertical (Amplitude) Scroll Bar Zoom Tool Zoom Previous The Journal summary shows the force increment used in your recordings. The grid divisions should use the same increment. Note this increment in Table 2.1 in the second column, Force (kg) Increments for Peak #1. For subsequent peaks, add the increment (i.e., 5, 10, 15 kg or 10, 20, 30 kg). The measurement boxes are above the marker region in the data window. Each measurement has three sections: channel number, measurement type, and result. The first two sections are pulldown menus that are activated when you click them. The following is a brief description of these specific measurements. mean: displays the average value in the selected area. p-p: finds the maximum value in the selected area and subtracts the minimum value found in the selected area. The selected area is the area selected by the I-Beam tool (including the endpoints).
13 Lesson 2: EMG II Page Use the I-Beam cursor to select an area on the plateau phase of the first clench (Fig. 2.11). A 6. Repeat Step 5 on the plateau of each successive clench. A 7. Scroll to Segment 3 and set up your display for optimal viewing. 8. Repeat Steps 5 and 6 for Segment 3. Analysis of Continued Clench 9. Scroll to Segment 2 and set up your display for optimal viewing. 10. Set up the measurement boxes as follows: Channel CH 1 CH 40 Measurement value delta T 11. Use the I-Beam cursor to select a point of maximal clench force immediately following the start of the segment (Fig. 2.12). B Fig Plateau of first clench selected Segment 3 begins at the third append marker, labeled Forearm 2, Increasing clench force. Enter your measurement findings in Table 2.1. Segment 2 begins at the second append marker, labeled Forearm 1, Continued clench at maximum force. The following is a brief description of the specific measurements not previously covered. value: displays the amplitude value for the channel at the point selected by the I-beam cursor. If a single point is selected, the value is for that point, if an area is selected, the value is the endpoint of the selected area. delta T: displays the amount of time in the selected segment (the difference in time between the endpoints of the selected area). 12. Calculate 50% of the maximum clench force from Step 11. B Data Analysis continues Fig The point selected should represent the maximal clench force at the start of Segment 2 (continuous maximal clench), as shown in Fig You will need this number to complete Step 14.
14 Page Find the point of 50% maximum clench force by using the I-beam cursor and leave the cursor at this point. 14. Select the area from the point of 50% clench force back to the point of maximal clench force by using the I- beam cursor and dragging. (Fig. 2.13). Note the time to fatigue (CH 40 delta T) measurement. B 15. Scroll to Segment 4 and set up your display for optimal viewing. 16. Repeat Steps 11 to 14 for Segment 4. Make an eyeball approximation of the point that is 50% down from the maximal clench point. Then, use the I-beam cursor to click points near this region, noting the value displayed in the measurement box, until you are on a point within 5% of the maximal clench force. Leave the cursor at this point. One way to select the area is as follows: The cursor should be flashing on the point of 50% maximal clench force. Hold down the mouse button and drag to the left of this point until you reach the point of maximal clench force, then release the mouse button. Fig showing area max-50% Segment 4 begins at the fourth append marker, labeled Forearm 2, Continued clench at maximum force. Enter your measurement findings in Table Save or print the data file. You may save the data to a floppy drive, save notes that are 18. Exit the program. in the journal, or print the data file. END OF DATA ANALYSIS END OF LESSON 2 Complete the Lesson 2 Data Report that follows.
15 Lesson 2: EMG II Page 15 DATA REPORT ELECTROMYOGRAPHY II Motor unit recruitment and Fatigue Student s Name: Lab Section: Date: I. Data and Calculations Motor Unit Recruitment Subject Profile Name Age Gender: Male / Female Height Weight Dominant forearm: Right / Left A. Complete Table 2.1 using Segment 1 and Segment 3 data. In the "Force (kg) Increments" column, note the force increment assigned for your recording under Peak #1; the increment was pasted to the Journal and should be noted below from Data Analysis Step 2. For subsequent peaks, add the increment (i.e., 5, 10, 15 or 10, 20, 30). You may not need nine peaks to reach max. Table 2.1 Segment 1 Data Seg. 1 Forearm 1 (Dominant) Seg. 2 Forearm 2 Force Raw Int. Force at Raw Int. Peak # Force (kg) Increments Assigned 1 kg 2 kg 3 kg 4 kg 5 kg 6 kg 7 kg 8 kg 9 kg at Peak [CH 1] mean (kg) EMG [CH 3] p-p (mv) EMG [CH 40] mean (mv) Peak [CH 1] mean (kg) EMG [CH 3] p-p (mv) EMG [CH 40] mean (mv)
16 Page 16 Fatigue B. Complete Table 2.2 using Segment 2 and Segment 4 data. Table 2.2 Segment 2 data Seg. 2 Forearm 1 (Dominant) Seg. 4 Forearm 2 Maximum Clench Force 50% of max clench force Time to Fatigue Maximum Clench Force 50% of max clench force Time to fatigue CH 1 value calculate CH 40 delta T* CH 1 value calculate CH 40 delta T* *Note: You do not need to indicate the delta T (time to fatigue) polarity. The polarity of the delta T measurement reflects the direction the "I-beam" cursor was dragged to select the data. Data selected left to right will have a positive ("+") polarity, while data selected right to left will have a negative ("-") polarity. II. Questions C. Is the strength of your right arm different than your left arm? Yes No D. Is there a difference in the absolute values of force generated by males and females in your class? Yes No What might explain any difference? E. When holding an object, does the number of motor units remain the same? Are the same motor units used for the duration of holding the object? F. As you fatigue, the force exerted by your muscles decreases. What physiological processes explain the decline in strength?
17 Lesson 2: EMG II Page 17 G. Define Motor unit H. Define Motor unit recruitment I. Define Fatigue J. Define EMG K. Define Dynamometry End of Lesson 2 Data Report
Richard Pflanzer, Ph.D. J.C. Uyehara, Ph.D. William McMullen David Pittman, Ph.D. Manual Revision PL3.7.0 M3.0.6 Modified by David Pittman
 ELECTROMYOGRAPHY (EMG) Laboratory Motor unit recruitment & Fatigue Comparison of dominant versus non-dominant arm using a within subject repeated measures statistical analysis Written by: Richard Pflanzer,
ELECTROMYOGRAPHY (EMG) Laboratory Motor unit recruitment & Fatigue Comparison of dominant versus non-dominant arm using a within subject repeated measures statistical analysis Written by: Richard Pflanzer,
Lesson 1 ELECTROMYOGRAPHY I Standard and Integrated EMG
 Physiology Lessons for use with the PC running Windows 98SE, Me, 2000 Pro, XP Pro/Home/Media Lesson 1 ELECTROMYOGRAPHY I Standard and Integrated EMG Manual Revision PL3.7.0 080706 Richard Pflanzer, Ph.D.
Physiology Lessons for use with the PC running Windows 98SE, Me, 2000 Pro, XP Pro/Home/Media Lesson 1 ELECTROMYOGRAPHY I Standard and Integrated EMG Manual Revision PL3.7.0 080706 Richard Pflanzer, Ph.D.
Lesson 2 EMG 2 Electromyography: Mechanical Work
 Physiology Lessons for use with the Biopac Science Lab MP40 Lesson 2 EMG 2 Electromyography: Mechanical Work PC running Windows XP or Mac OS X 10.3-10.4 Lesson Revision 5.23.2006 BIOPAC Systems, Inc. 42
Physiology Lessons for use with the Biopac Science Lab MP40 Lesson 2 EMG 2 Electromyography: Mechanical Work PC running Windows XP or Mac OS X 10.3-10.4 Lesson Revision 5.23.2006 BIOPAC Systems, Inc. 42
Physiology Lessons for use with the Biopac Student Lab. Lesson 1 ELECTROMYOGRAPHY I Standard and Integrated EMG
 Physiology Lessons for use with the Biopac Student Lab Lesson 1 ELECTROMYOGRAPHY I Standard and Integrated EMG Richard Pflanzer, Ph.D. Associate Professor Emeritus Indiana University School of Medicine
Physiology Lessons for use with the Biopac Student Lab Lesson 1 ELECTROMYOGRAPHY I Standard and Integrated EMG Richard Pflanzer, Ph.D. Associate Professor Emeritus Indiana University School of Medicine
Exercise 5: Muscle Physiology I - Electromyography
 Exercise 5: Muscle Physiology I - Electromyography Readings: Silverthorn, 6 th ed. pg. 410 420 Your brain communicates with your muscles through action potentials on the motor neurons, which are then transmitted
Exercise 5: Muscle Physiology I - Electromyography Readings: Silverthorn, 6 th ed. pg. 410 420 Your brain communicates with your muscles through action potentials on the motor neurons, which are then transmitted
Biopac Student Lab Lesson 5 ELECTROCARDIOGRAPHY (ECG) I Procedure. Rev
 42 Aero Camino, Goleta, CA 93117 www.biopac.com Biopac Student Lab Lesson 5 ELECTROCARDIOGRAPHY (ECG) I Procedure Rev. 07112013 Richard Pflanzer, Ph.D. Associate Professor Emeritus Indiana University School
42 Aero Camino, Goleta, CA 93117 www.biopac.com Biopac Student Lab Lesson 5 ELECTROCARDIOGRAPHY (ECG) I Procedure Rev. 07112013 Richard Pflanzer, Ph.D. Associate Professor Emeritus Indiana University School
Lesson 4 ECG 2 Electrocardiography
 Physiology Lessons for use with the Biopac Science Lab MP40 Lesson 4 ECG 2 Electrocardiography PC running Windows XP or Mac OS X 10.3-10.4 Lesson Revision 1.20.2006 BIOPAC Systems, Inc. 42 Aero Camino,
Physiology Lessons for use with the Biopac Science Lab MP40 Lesson 4 ECG 2 Electrocardiography PC running Windows XP or Mac OS X 10.3-10.4 Lesson Revision 1.20.2006 BIOPAC Systems, Inc. 42 Aero Camino,
Biopac Student Lab Lesson 6 ELECTROCARDIOGRAPHY (ECG) II Analysis Procedure. Rev
 42 Aero Camino, Goleta, CA 93117 www.biopac.com Biopac Student Lab Lesson 6 ELECTROCARDIOGRAPHY (ECG) II Analysis Procedure Rev. 12292017 Richard Pflanzer, Ph.D. Associate Professor Emeritus Indiana University
42 Aero Camino, Goleta, CA 93117 www.biopac.com Biopac Student Lab Lesson 6 ELECTROCARDIOGRAPHY (ECG) II Analysis Procedure Rev. 12292017 Richard Pflanzer, Ph.D. Associate Professor Emeritus Indiana University
Biopac Student Lab Lesson 20 SPINAL CORD REFLEXES Analysis Procedure. Rev
 42 Aero Camino, Goleta, CA 93117 www.biopac.com Biopac Student Lab Lesson 20 SPINAL CORD REFLEXES Analysis Procedure Rev. 12292017 Richard Pflanzer, Ph.D. Associate Professor Emeritus Indiana University
42 Aero Camino, Goleta, CA 93117 www.biopac.com Biopac Student Lab Lesson 20 SPINAL CORD REFLEXES Analysis Procedure Rev. 12292017 Richard Pflanzer, Ph.D. Associate Professor Emeritus Indiana University
Biopac Student Lab Lesson 3 ELECTROENCEPHALOGRAPHY (EEG) I Analysis Procedure. Rev
 42 Aero Camino, Goleta, CA 93117 www.biopac.com Biopac Student Lab Lesson 3 ELECTROENCEPHALOGRAPHY (EEG) I Analysis Procedure Rev. 12112014 Richard Pflanzer, Ph.D. Associate Professor Emeritus Indiana
42 Aero Camino, Goleta, CA 93117 www.biopac.com Biopac Student Lab Lesson 3 ELECTROENCEPHALOGRAPHY (EEG) I Analysis Procedure Rev. 12112014 Richard Pflanzer, Ph.D. Associate Professor Emeritus Indiana
Lesson 10 Aerobic Exercise Physiology
 Physiology Lessons for use with the Biopac Science Lab MP40 Lesson 10 Aerobic Exercise Physiology PC running Windows XP or Mac OS X 10.3-10.4 Lesson Revision 12.19.2005 BIOPAC Systems, Inc. 42 Aero Camino,
Physiology Lessons for use with the Biopac Science Lab MP40 Lesson 10 Aerobic Exercise Physiology PC running Windows XP or Mac OS X 10.3-10.4 Lesson Revision 12.19.2005 BIOPAC Systems, Inc. 42 Aero Camino,
Lesson 5 EEG 1 Electroencephalography: Brain Rhythms
 Physiology Lessons for use with the Biopac Science Lab MP40 PC running Windows XP or Mac OS X 10.3-10.4 Lesson 5 EEG 1 Electroencephalography: Brain Rhythms Lesson Revision 2.23.2006 BIOPAC Systems, Inc.
Physiology Lessons for use with the Biopac Science Lab MP40 PC running Windows XP or Mac OS X 10.3-10.4 Lesson 5 EEG 1 Electroencephalography: Brain Rhythms Lesson Revision 2.23.2006 BIOPAC Systems, Inc.
Biopac Student Lab Lesson 8 RESPIRATORY CYCLE I Analysis Procedure. Rev
 42 Aero Camino, Goleta, CA 93117 www.biopac.com Biopac Student Lab Lesson 8 RESPIRATORY CYCLE I Analysis Procedure Rev. 12292017 Richard Pflanzer, Ph.D. Associate Professor Emeritus Indiana University
42 Aero Camino, Goleta, CA 93117 www.biopac.com Biopac Student Lab Lesson 8 RESPIRATORY CYCLE I Analysis Procedure Rev. 12292017 Richard Pflanzer, Ph.D. Associate Professor Emeritus Indiana University
Overview Active Learning
 : EMG Active Learning Updated 01-06-11 This lesson is based on a presentation given by Dr. Jennifer Lundmark and Ms. Andrea Salmi at the HAPS Conference, Maui, HI, June 2-7, 2001 Overview Active Learning
: EMG Active Learning Updated 01-06-11 This lesson is based on a presentation given by Dr. Jennifer Lundmark and Ms. Andrea Salmi at the HAPS Conference, Maui, HI, June 2-7, 2001 Overview Active Learning
Physiology Lessons for use with the Biopac Student Lab
 Physiology Lessons for use with the Biopac Student Lab Lesson 6 ELECTROCARDIOGRAPHY II Bipolar Leads (Leads I, II, III) Einthoven s Law Mean Electrical Axis on the Frontal Plane Richard Pflanzer, Ph.D.
Physiology Lessons for use with the Biopac Student Lab Lesson 6 ELECTROCARDIOGRAPHY II Bipolar Leads (Leads I, II, III) Einthoven s Law Mean Electrical Axis on the Frontal Plane Richard Pflanzer, Ph.D.
Figure 1 The respiratory cycle
 RESPIRATORY CYCLE Introduction Three primary functions of the respiratory system are to provide oxygen for the body's energy needs, provide an outlet for C0 2 and help maintain the ph of the blood plasma.
RESPIRATORY CYCLE Introduction Three primary functions of the respiratory system are to provide oxygen for the body's energy needs, provide an outlet for C0 2 and help maintain the ph of the blood plasma.
Lesson 5 BIOPAC Systems, Inc. Manual Revision PL3.7.5
 Physiology Lessons for use with the Biopac Student Lab Lesson 5 ELECTROCARDIOGRAPHY I Components of the ECG Richard Pflanzer, Ph.D. Associate Professor Emeritus Indiana University School of Medicine Purdue
Physiology Lessons for use with the Biopac Student Lab Lesson 5 ELECTROCARDIOGRAPHY I Components of the ECG Richard Pflanzer, Ph.D. Associate Professor Emeritus Indiana University School of Medicine Purdue
Lesson 9. Measuring Emotional Responses to the Acoustic Startle Reflex: Stimulus Habituation & Sensitization
 Measuring Emotional Responses to the Acoustic Startle Reflex: Stimulus Habituation & Sensitization Revised 10 / 29 / 2012 Lesson 9 METHOD: GALVANIC SKIN RESPONSE & THE POLYGRAPH David W. Pittman, Ph.D.
Measuring Emotional Responses to the Acoustic Startle Reflex: Stimulus Habituation & Sensitization Revised 10 / 29 / 2012 Lesson 9 METHOD: GALVANIC SKIN RESPONSE & THE POLYGRAPH David W. Pittman, Ph.D.
BSL PRO Lesson H03: Nerve Conduction Velocity: Along the Ulnar Nerve of a Human Subject
 Updated 12-22-03 BSL PRO Lesson H03: Nerve Conduction Velocity: Along the Ulnar Nerve of a Human Subject This PRO lesson describes hardware and software setup of the BSL PRO System to record and measure
Updated 12-22-03 BSL PRO Lesson H03: Nerve Conduction Velocity: Along the Ulnar Nerve of a Human Subject This PRO lesson describes hardware and software setup of the BSL PRO System to record and measure
Exercise 6: Muscle Physiology II Twitch & Summation
 Exercise 6: Muscle Physiology II Twitch & Summation Text Reading: Silverthorn, 5 th ed. 412 419, 425 427; 6 th ed. pg. 410 420 In this exercise, we will investigate the physiology of contraction in the
Exercise 6: Muscle Physiology II Twitch & Summation Text Reading: Silverthorn, 5 th ed. 412 419, 425 427; 6 th ed. pg. 410 420 In this exercise, we will investigate the physiology of contraction in the
Biology 13A Lab #10: Cardiovascular System II ECG & Heart Disease
 Biology 13A Lab #10: Cardiovascular System II ECG & Heart Disease Lab #10 Table of Contents: Expected Learning Outcomes...... 83 Introduction....... 84 Activity 1: Collecting ECG Data..... 85 Activity
Biology 13A Lab #10: Cardiovascular System II ECG & Heart Disease Lab #10 Table of Contents: Expected Learning Outcomes...... 83 Introduction....... 84 Activity 1: Collecting ECG Data..... 85 Activity
Lab 5: Electromyograms (EMGs)
 Lab 5: Electromyograms (EMGs) Overview A motorneuron and all the muscle fibers that it innervates is known as a motor unit. Under normal circumstances, a neuronal action potential activates all of the
Lab 5: Electromyograms (EMGs) Overview A motorneuron and all the muscle fibers that it innervates is known as a motor unit. Under normal circumstances, a neuronal action potential activates all of the
Experiment HH-3: Exercise, the Electrocardiogram, and Peripheral Circulation
 Experiment HH-3: Exercise, the Electrocardiogram, and Peripheral Circulation Background The arterial system functions as a pressure reservoir. Blood enters via the heart and exits through the capillaries.
Experiment HH-3: Exercise, the Electrocardiogram, and Peripheral Circulation Background The arterial system functions as a pressure reservoir. Blood enters via the heart and exits through the capillaries.
BC1002 Module 1. Biol BC1002, Spring semester 2005, Module 1, Lab 1-1
 BC1002 Module 1 Biol BC1002, Spring semester 2005, Module 1, Lab 1-1 MODULE 1: LAB 1 MUSCLE PHYSIOLOGY Spring semester 2005, Week of January 24-28. Learning objectives to learn the microscopic structure
BC1002 Module 1 Biol BC1002, Spring semester 2005, Module 1, Lab 1-1 MODULE 1: LAB 1 MUSCLE PHYSIOLOGY Spring semester 2005, Week of January 24-28. Learning objectives to learn the microscopic structure
Overview. Page 1 of 9. Impedance Cardiography
 Updated 05.14.10 BSL PRO Lesson H21: Impedance Cardiography Data collected from a subject using the referenced set-up procedure. Note that dz/dt maximum is determined on a cycle by cycle basis from the
Updated 05.14.10 BSL PRO Lesson H21: Impedance Cardiography Data collected from a subject using the referenced set-up procedure. Note that dz/dt maximum is determined on a cycle by cycle basis from the
Raw BP and Eight Hemodynamic Measurements Displayed in Real Time
 APPLICATION NOTE 42 Aero Camino, Goleta, CA 93117 Tel (805) 685-0066 Fax (805) 685-0067 info@biopac.com www.biopac.com Updated 01.11.2016 Application Note 115: Hemodynamic Measurements Raw BP and Eight
APPLICATION NOTE 42 Aero Camino, Goleta, CA 93117 Tel (805) 685-0066 Fax (805) 685-0067 info@biopac.com www.biopac.com Updated 01.11.2016 Application Note 115: Hemodynamic Measurements Raw BP and Eight
Evaluation copy. EMG and Muscle Fatigue. Computer
 EMG and Muscle Fatigue Computer 18 Voluntary muscle contraction is the result of communication between the brain and individual muscle fibers of the musculoskeletal system. A thought is transformed into
EMG and Muscle Fatigue Computer 18 Voluntary muscle contraction is the result of communication between the brain and individual muscle fibers of the musculoskeletal system. A thought is transformed into
Experiment HM-11: Electromyograms (EMG) for Paired Arm Wrestling
 Experiment HM-11: Electromyograms (EMG) for Paired Arm Wrestling Background The movement of parts of the body is accomplished through a system of levers composed of skeletal muscles and bones. In a lever,
Experiment HM-11: Electromyograms (EMG) for Paired Arm Wrestling Background The movement of parts of the body is accomplished through a system of levers composed of skeletal muscles and bones. In a lever,
1724 Lab: Frog Skeletal Muscle Physiology (Marieb Exercise 16A) Marieb/iWorx / Ziser, 2002
 1724 Lab: Frog Skeletal Muscle Physiology (Marieb Exercise 16A) Marieb/iWorx / Ziser, 2002 I. Introduction. Read the introductory material in your lab manual Marieb Ex 16A: Skeletal Muscle Physiology Frog
1724 Lab: Frog Skeletal Muscle Physiology (Marieb Exercise 16A) Marieb/iWorx / Ziser, 2002 I. Introduction. Read the introductory material in your lab manual Marieb Ex 16A: Skeletal Muscle Physiology Frog
Cardiovascular Effects of Exercise. Background Cardiac function
 Cardiovascular Effects of Exercise In this experiment, you will record an electrocardiogram, or ECG, and finger pulse from a healthy volunteer. You will then compare the ECG and pulse recordings when the
Cardiovascular Effects of Exercise In this experiment, you will record an electrocardiogram, or ECG, and finger pulse from a healthy volunteer. You will then compare the ECG and pulse recordings when the
ELECTROMYOGRAM ANALYSIS OF MUSCLE FUNCTION INTRODUCTION
 ELECTROMYOGRAM ANALYSIS OF MUSCLE FUNCTION STANDARDS: 3.3.10.B - Explain cell functions and processes in terms of chemical reactions and energy changes. 3.3.12.B - Evaluate relationships between structure
ELECTROMYOGRAM ANALYSIS OF MUSCLE FUNCTION STANDARDS: 3.3.10.B - Explain cell functions and processes in terms of chemical reactions and energy changes. 3.3.12.B - Evaluate relationships between structure
iworx Sample Lab Experiment HP-7: Hypothesis-driven Biofeedback Lab/Research Study
 Experiment HP-7: Hypothesis-driven Biofeedback Lab/Research Study What is Biofeedback? Biofeedback is a technique that people can use to learn to control their body's physiological functions. The definition
Experiment HP-7: Hypothesis-driven Biofeedback Lab/Research Study What is Biofeedback? Biofeedback is a technique that people can use to learn to control their body's physiological functions. The definition
iworx Sample Lab Experiment HC-3: Pulse Wave Velocity
 Experiment HC-3: Pulse Wave Velocity Exercise 1: The Pulse Wave Velocity at Rest Aim: To measure the pulse wave velocity in a resting individual. Procedure 1. Click on the Record button, located on the
Experiment HC-3: Pulse Wave Velocity Exercise 1: The Pulse Wave Velocity at Rest Aim: To measure the pulse wave velocity in a resting individual. Procedure 1. Click on the Record button, located on the
SS11LA Pneumotach Transducer
 BIOPAC Systems, Inc. 42 Aero Camino Goleta, CA 93117 Ph (805) 685-0066 Fax (805) 685-0067 www.biopac.com info@biopac.com SS11LA Pneumotach Transducer The SS11LA can be used to measure respiratory flow
BIOPAC Systems, Inc. 42 Aero Camino Goleta, CA 93117 Ph (805) 685-0066 Fax (805) 685-0067 www.biopac.com info@biopac.com SS11LA Pneumotach Transducer The SS11LA can be used to measure respiratory flow
Experiment HH-3: Exercise, the Electrocardiogram, and Peripheral Circulation
 Experiment HH-3: Exercise, the Electrocardiogram, and Peripheral Circulation Exercise 1: The ECG and the Pulse in a Resting Subject Aim: To measure and correlate the ECG and the pulse in a resting individual.
Experiment HH-3: Exercise, the Electrocardiogram, and Peripheral Circulation Exercise 1: The ECG and the Pulse in a Resting Subject Aim: To measure and correlate the ECG and the pulse in a resting individual.
Experiment HH-2: The Electrocardiogram and Heart Sounds
 Experiment HH-2: The Electrocardiogram and Heart Sounds Exercise 1: The ECG in a Resting Subject Aim: To measure the ECG in a resting individual. Procedure 1. Click on the Record button, located on the
Experiment HH-2: The Electrocardiogram and Heart Sounds Exercise 1: The ECG in a Resting Subject Aim: To measure the ECG in a resting individual. Procedure 1. Click on the Record button, located on the
Skeletal muscles are composed of hundreds to thousands of individual cells,
 2 E X E R C I S E Skeletal Muscle Physiology O B J E C T I V E S 1. To define these terms used in describing muscle physiology: multiple motor unit summation, maximal stimulus, treppe, wave summation,
2 E X E R C I S E Skeletal Muscle Physiology O B J E C T I V E S 1. To define these terms used in describing muscle physiology: multiple motor unit summation, maximal stimulus, treppe, wave summation,
Warning: Avoid putting the tip of the syringe needle too deeply into the hole to avoid damage to the heart tissue.
 Experiment AM-12: Crayfish Electrocardiogram (ECG) Exercise 1: The Crayfish ECG and Heart Rate Aim: To record the electrical trace (ECG) produced by the contraction of a resting heart, and to determine
Experiment AM-12: Crayfish Electrocardiogram (ECG) Exercise 1: The Crayfish ECG and Heart Rate Aim: To record the electrical trace (ECG) produced by the contraction of a resting heart, and to determine
iworx Sample Lab Experiment HH-4: The Six-Lead Electrocardiogram
 Experiment HH-4: The Six-Lead Electrocardiogram Background The cardiac cycle involves a sequential contraction of the atria and the ventricles. These contractions are triggered by the coordinated electrical
Experiment HH-4: The Six-Lead Electrocardiogram Background The cardiac cycle involves a sequential contraction of the atria and the ventricles. These contractions are triggered by the coordinated electrical
BIO 360: Vertebrate Physiology Lab 8b: Electrical activity of muscular contractions
 Lab report DUE 3/19 Muscle tissues maintain electrical imbalances, or potentials, across cell membranes by concentrating positive or negative charges on opposite sides of those membranes. These potentials
Lab report DUE 3/19 Muscle tissues maintain electrical imbalances, or potentials, across cell membranes by concentrating positive or negative charges on opposite sides of those membranes. These potentials
Experiment HM-7: Electromyogram (EMG) Activity in Antagonistic Muscles and Range of Motion
 Experiment HM-7: Electromyogram (EMG) Activity in Antagonistic Muscles and Range of Motion Exercise 1: Antagonistic Muscles in Forearm Aim: To study the EMG activity in muscles that work in opposition
Experiment HM-7: Electromyogram (EMG) Activity in Antagonistic Muscles and Range of Motion Exercise 1: Antagonistic Muscles in Forearm Aim: To study the EMG activity in muscles that work in opposition
Humans make voluntary decisions to talk, walk, stand up, or sit down. The
 2 E X E R C I S E Skeletal Muscle Physiology O B J E C T I V E S 1. To define motor unit, twitch, latent period, contraction phase, relaxation phase, threshold, summation, tetanus, fatigue, isometric contraction,
2 E X E R C I S E Skeletal Muscle Physiology O B J E C T I V E S 1. To define motor unit, twitch, latent period, contraction phase, relaxation phase, threshold, summation, tetanus, fatigue, isometric contraction,
Lesson 16 BLOOD PRESSURE
 Physiology Lessons for use with the Lesson 16 BLOOD PRESSURE Indirect measurement Ventricular Systole & Diastole Korotkoff sounds Mean Arterial pressure Manual Revision 3.7.3 061808 Richard Pflanzer, Ph.D.
Physiology Lessons for use with the Lesson 16 BLOOD PRESSURE Indirect measurement Ventricular Systole & Diastole Korotkoff sounds Mean Arterial pressure Manual Revision 3.7.3 061808 Richard Pflanzer, Ph.D.
Experiment HP-28: Control of EEG and Biofeedback Lab/Research Study
 Experiment HP-28: Control of EEG and Biofeedback Lab/Research Study Equipment Required PC or Mac Computer IXTA, USB cable and power supply iwire-b3g cable and EEG leads Reusable Gold-Cup EEG Electrodes
Experiment HP-28: Control of EEG and Biofeedback Lab/Research Study Equipment Required PC or Mac Computer IXTA, USB cable and power supply iwire-b3g cable and EEG leads Reusable Gold-Cup EEG Electrodes
Biology 325 Fall 2003 Human anatomy and physiology
 SKELETAL MUSCLE PROPERTIES Background : In this experiment, you will investigate the physiological properties of skeletal muscle from the isolated amphibian gastrocnemius. You will examine skeletal muscle
SKELETAL MUSCLE PROPERTIES Background : In this experiment, you will investigate the physiological properties of skeletal muscle from the isolated amphibian gastrocnemius. You will examine skeletal muscle
Lab #3: Electrocardiogram (ECG / EKG)
 Lab #3: Electrocardiogram (ECG / EKG) An introduction to the recording and analysis of cardiac activity Introduction The beating of the heart is triggered by an electrical signal from the pacemaker. The
Lab #3: Electrocardiogram (ECG / EKG) An introduction to the recording and analysis of cardiac activity Introduction The beating of the heart is triggered by an electrical signal from the pacemaker. The
Electromyography II Laboratory (Hand Dynamometer Transducer)
 (Hand Dynamometer Transducer) Introduction As described in the Electromyography I laboratory session, electromyography (EMG) is an electrical signal that can be recorded with electrodes placed on the surface
(Hand Dynamometer Transducer) Introduction As described in the Electromyography I laboratory session, electromyography (EMG) is an electrical signal that can be recorded with electrodes placed on the surface
Grip Strength and Muscle Fatigue JB19
 Grip Strength and Muscle Fatigue JB19 Skeletal muscle is composed of bundles of individual muscle fibers (see Figure 1) and has unique properties which allow it to respond to stimuli by contracting. Individual
Grip Strength and Muscle Fatigue JB19 Skeletal muscle is composed of bundles of individual muscle fibers (see Figure 1) and has unique properties which allow it to respond to stimuli by contracting. Individual
Muscle Function Analysis
 Muscle Function Analysis LabQuest 15 Muscle tissues maintain electrical imbalances, or potentials, across cell membranes by concentrating positive or negative charges on opposite sides of those membranes.
Muscle Function Analysis LabQuest 15 Muscle tissues maintain electrical imbalances, or potentials, across cell membranes by concentrating positive or negative charges on opposite sides of those membranes.
Sanako Lab 100 STS USER GUIDE
 Sanako Lab 100 STS USER GUIDE Copyright 2002-2015 SANAKO Corporation. All rights reserved. Microsoft is a registered trademark. Microsoft Windows XP, Windows Vista and Windows 7 are trademarks of Microsoft
Sanako Lab 100 STS USER GUIDE Copyright 2002-2015 SANAKO Corporation. All rights reserved. Microsoft is a registered trademark. Microsoft Windows XP, Windows Vista and Windows 7 are trademarks of Microsoft
CAMOSUN COLLEGE BIOLOGY 144 (2010) LABS
 LAB 8: CARDIOVASCULAR PHYSIOLOGY PART 1. HEART SOUNDS AND PULSE DETERMINATIONS Introduction Two distinct sounds can be heard during each cardiac cycle. These sounds are commonly described as lub and dup
LAB 8: CARDIOVASCULAR PHYSIOLOGY PART 1. HEART SOUNDS AND PULSE DETERMINATIONS Introduction Two distinct sounds can be heard during each cardiac cycle. These sounds are commonly described as lub and dup
Labs #7 and #8: Vertebrate Skeletal Muscle
 Labs #7 and #8: Vertebrate Skeletal Muscle In this experiment, you will investigate the physiological properties of skeletal muscle from the isolated toad gastrocnemius. Concepts to understand include
Labs #7 and #8: Vertebrate Skeletal Muscle In this experiment, you will investigate the physiological properties of skeletal muscle from the isolated toad gastrocnemius. Concepts to understand include
Electromyography Lab
 Electromyography Lab By: Chelsea L. Giles Instructor: Professor Delon Washo-Krupps Experiment Conducted On: February 15, 2015 Lab Report Submitted: February 23, 2015 Title: Electromyography Lab Introduction:
Electromyography Lab By: Chelsea L. Giles Instructor: Professor Delon Washo-Krupps Experiment Conducted On: February 15, 2015 Lab Report Submitted: February 23, 2015 Title: Electromyography Lab Introduction:
#AH145 - TSD101B Respiratory Effort Transducer
 BIOPAC Systems, Inc. 42 Aero Camino Goleta, CA 93117 Ph (805) 685-0066 Fax (805) 685-0067 www.biopac.com info@biopac.com #AH145 - TSD101B Respiratory Effort Transducer The TSD101B can be used to record
BIOPAC Systems, Inc. 42 Aero Camino Goleta, CA 93117 Ph (805) 685-0066 Fax (805) 685-0067 www.biopac.com info@biopac.com #AH145 - TSD101B Respiratory Effort Transducer The TSD101B can be used to record
Exercise 1: Eye-Hand Reaction Times Aim: To measure the reaction time of a subject to a visual cue when responding with the hand.
 Experiment HN-4: Hand vs. Foot Reactions Exercise 1: Eye-Hand Reaction Times Aim: To measure the reaction time of a subject to a visual cue when responding with the hand. Procedure 1. Instruct the subject
Experiment HN-4: Hand vs. Foot Reactions Exercise 1: Eye-Hand Reaction Times Aim: To measure the reaction time of a subject to a visual cue when responding with the hand. Procedure 1. Instruct the subject
Exercise 2: Effects of Cold Temperature Aim: To record changes in heart rate after the heart is bathed in cold Ringer s solution.
 Experiment AM-3: Heart Muscle Warning: The heart preparation used in this experiment is functional for a limited period of time. If the muscle is bathed periodically in Ringer s solution, it will work
Experiment AM-3: Heart Muscle Warning: The heart preparation used in this experiment is functional for a limited period of time. If the muscle is bathed periodically in Ringer s solution, it will work
Cardiovascular Physiology
 Cardiovascular Physiology The mammalian heart is a pump that pushes blood around the body and is made of four chambers: right and left atria and right and left ventricles. The two atria act as collecting
Cardiovascular Physiology The mammalian heart is a pump that pushes blood around the body and is made of four chambers: right and left atria and right and left ventricles. The two atria act as collecting
#4 Cardiovascular I The Heart & EKG
 #4 Cardiovascular I Objectives: Identify a list of human heart structures using a virtual human dissection Dissect a sheep heart to identify external and internal structures Identify a list of human heart
#4 Cardiovascular I Objectives: Identify a list of human heart structures using a virtual human dissection Dissect a sheep heart to identify external and internal structures Identify a list of human heart
Lab #3: Electrocardiogram (ECG / EKG)
 Lab #3: Electrocardiogram (ECG / EKG) An introduction to the recording and analysis of cardiac activity Introduction The beating of the heart is triggered by an electrical signal from the pacemaker. The
Lab #3: Electrocardiogram (ECG / EKG) An introduction to the recording and analysis of cardiac activity Introduction The beating of the heart is triggered by an electrical signal from the pacemaker. The
iworx Sample Lab Experiment AN-5: Cockroach Leg Mechanoreceptors
 Experiment AN-5: Cockroach Leg Mechanoreceptors Exercise 1: Chordotonal Organs Aim: To explore the basic characteristics of the chordotonal organs, their response to direction and intensity of leg movement,
Experiment AN-5: Cockroach Leg Mechanoreceptors Exercise 1: Chordotonal Organs Aim: To explore the basic characteristics of the chordotonal organs, their response to direction and intensity of leg movement,
Experiment HE-12: Targeted Exercise with Wireless Electrocardiogram (ECG)
 Experiment HE-12: Targeted Exercise with Wireless Electrocardiogram (ECG) Get Your Cardio On Preparations for this experiment need to be reviewed with the students in advance. There will be exercise routines
Experiment HE-12: Targeted Exercise with Wireless Electrocardiogram (ECG) Get Your Cardio On Preparations for this experiment need to be reviewed with the students in advance. There will be exercise routines
Hanwell Instruments Ltd. Instruction Manual
 Hanwell Instruments Ltd Instruction Manual Document Title RL5000 Sensors - User Guide Document No. IM4177 Issue No. 3 Hanwell Instruments Ltd 12 Mead Business Centre Mead Lane Hertford SG13 7BJ UNITED
Hanwell Instruments Ltd Instruction Manual Document Title RL5000 Sensors - User Guide Document No. IM4177 Issue No. 3 Hanwell Instruments Ltd 12 Mead Business Centre Mead Lane Hertford SG13 7BJ UNITED
Teacher s Guide. Physiology Lessons for use with the Biopac Science Lab MP40
 Physiology Lessons for use with the Biopac Science Lab MP40 Teacher s Guide Overview 2 About Electrodes 3 The Body Electric 6 Waveforms 7 National Science Standards 19 Lesson 1 Lesson 2 Lesson 3 Lesson
Physiology Lessons for use with the Biopac Science Lab MP40 Teacher s Guide Overview 2 About Electrodes 3 The Body Electric 6 Waveforms 7 National Science Standards 19 Lesson 1 Lesson 2 Lesson 3 Lesson
Evaluation copy. Grip Strength and Muscle Fatigue. Computer
 Grip Strength and Muscle Fatigue Computer 17 Skeletal muscle is composed of bundles of individual muscle fibers (see Figure 1) and has unique properties which allow it to respond to stimuli by contracting.
Grip Strength and Muscle Fatigue Computer 17 Skeletal muscle is composed of bundles of individual muscle fibers (see Figure 1) and has unique properties which allow it to respond to stimuli by contracting.
FT-302 Force Transducer
 Technical Note FT-302 LabScribe is a trademark of 2015 Overview The FT-302 is a high-sensitivity dual-range research grade force transducer designed to measure forces in the 0.005 to 10 gram and 0 to 100
Technical Note FT-302 LabScribe is a trademark of 2015 Overview The FT-302 is a high-sensitivity dual-range research grade force transducer designed to measure forces in the 0.005 to 10 gram and 0 to 100
Bio-logic AuDX PRO FLEX Screening tympanometry and audiometry Quick start guide
 AuDX PRO FLEX Screening tympanometry and audiometry Quick start guide Doc. No.7-26-1374-EN/00 Part No.7-26-13704-EN Natus Medical Denmark ApS. +45 45 75 55 55. otoinfo@natus.com Natus Medical Incorporated.
AuDX PRO FLEX Screening tympanometry and audiometry Quick start guide Doc. No.7-26-1374-EN/00 Part No.7-26-13704-EN Natus Medical Denmark ApS. +45 45 75 55 55. otoinfo@natus.com Natus Medical Incorporated.
MEDIUM-FLOW PNEUMOTACH TRANSDUCER
 MEDIUM-FLOW PNEUMOTACH TRANSDUCER SS11LA for MP3x and MP45 System TSD117 & TSD117-MRI for MP150/MP100 System RX117 Replacement Airflow Head See also: AFT series of accessories for airflow and gas analysis
MEDIUM-FLOW PNEUMOTACH TRANSDUCER SS11LA for MP3x and MP45 System TSD117 & TSD117-MRI for MP150/MP100 System RX117 Replacement Airflow Head See also: AFT series of accessories for airflow and gas analysis
Comparing the Functional Performance of BIOPAC Respiratory Belt-Transducers With and Without Repair Tape
 Comparing the Functional Performance of BIOPAC Respiratory Belt-Transducers With and Without Repair Tape Ames A., Berning M., Chreptowicz N., Hackney R., Kelly E., Noffke A., Lokuta AJ.* *Corresponding
Comparing the Functional Performance of BIOPAC Respiratory Belt-Transducers With and Without Repair Tape Ames A., Berning M., Chreptowicz N., Hackney R., Kelly E., Noffke A., Lokuta AJ.* *Corresponding
SANAKO Lab 100 STS USER GUIDE
 SANAKO Lab 100 STS USER GUIDE Copyright 2008 SANAKO Corporation. All rights reserved. Microsoft is a registered trademark. Microsoft Windows 2000 and Windows XP are trademarks of Microsoft Corporation.
SANAKO Lab 100 STS USER GUIDE Copyright 2008 SANAKO Corporation. All rights reserved. Microsoft is a registered trademark. Microsoft Windows 2000 and Windows XP are trademarks of Microsoft Corporation.
The following describes general use of ELIOS device
 General use of the device The following describes general use of ELIOS device 1. Introduction of graphical user interface Presence indicator of SD card Charging indicator General Informations Screen title
General use of the device The following describes general use of ELIOS device 1. Introduction of graphical user interface Presence indicator of SD card Charging indicator General Informations Screen title
Lab 4: Introduction to Physiological Measurements - Cardiovascular
 Lab 4: Introduction to Physiological Measurements - Cardiovascular INTRODUCTION: This lab will demonstrate cardiovascular measurements by creating an ECG with instruments used in previous labs. Students
Lab 4: Introduction to Physiological Measurements - Cardiovascular INTRODUCTION: This lab will demonstrate cardiovascular measurements by creating an ECG with instruments used in previous labs. Students
version User s Guide nnnnnnnnnnnnnnnnnnnnnn AUTOMATIC POULTRY SCALES BAT2 Lite
 version 1.02.0 User s Guide nnnnnnnnnnnnnnnnnnnnnn AUTOMATIC POULTRY SCALES BAT2 Lite 1. INTRODUCTION... 2 1.1. Scales Description... 2 1.2. Basic Technical Parameters... 2 1.3. Factory Setup of the Scales...
version 1.02.0 User s Guide nnnnnnnnnnnnnnnnnnnnnn AUTOMATIC POULTRY SCALES BAT2 Lite 1. INTRODUCTION... 2 1.1. Scales Description... 2 1.2. Basic Technical Parameters... 2 1.3. Factory Setup of the Scales...
Step-by-Step RECD Guide
 Precision Audiometric Instruments www.medrx-usa.com Step-by-Step RECD Guide The RECD task involves 4 steps: 1 - Complete Calibration of the Speakers and Probe Tube 2 - Measure an Ear Response 3 - Perform
Precision Audiometric Instruments www.medrx-usa.com Step-by-Step RECD Guide The RECD task involves 4 steps: 1 - Complete Calibration of the Speakers and Probe Tube 2 - Measure an Ear Response 3 - Perform
VMA Demo Unit. Introduction. This document provides information on how to set up and operate the VMA Demo Unit. Figure 1: VMA Demo Unit
 Application Specific Controllers Technical Manual 636.3 VMA Controller Section Technical Bulletin Issue Date 0199 VMA Demo Unit Introduction This document provides information on how to set up and operate
Application Specific Controllers Technical Manual 636.3 VMA Controller Section Technical Bulletin Issue Date 0199 VMA Demo Unit Introduction This document provides information on how to set up and operate
Grip Strength and Muscle Fatigue
 Anatomy & Physiology Foothill High School Grip Strength and Muscle Fatigue Skeletal muscle is composed of bundles of individual muscle fibers (see Figure 1) and has unique properties which allow it to
Anatomy & Physiology Foothill High School Grip Strength and Muscle Fatigue Skeletal muscle is composed of bundles of individual muscle fibers (see Figure 1) and has unique properties which allow it to
Adventure 4 Plus Service Manual
 Adventure 4 Plus Service Manual 1 TABLE OF CONTENTS CHAPTER 1: SERIAL NUMBER LOCATION...3 CHAPTER 2: PREVENTATIVE MAINTENANCE 2.1 Preventative Maintenance. 4 2.2 Tension and Centering the Running Belt....6
Adventure 4 Plus Service Manual 1 TABLE OF CONTENTS CHAPTER 1: SERIAL NUMBER LOCATION...3 CHAPTER 2: PREVENTATIVE MAINTENANCE 2.1 Preventative Maintenance. 4 2.2 Tension and Centering the Running Belt....6
RELEASED. Clearing your active insulin
 To clear all your settings: 1. Make sure the pump is not connected to your body. 2. Go to the Manage Settings screen. Menu > Utilities > Manage Settings 3. Simultaneously press and hold and until the Manage
To clear all your settings: 1. Make sure the pump is not connected to your body. 2. Go to the Manage Settings screen. Menu > Utilities > Manage Settings 3. Simultaneously press and hold and until the Manage
The Gaze Cueing Paradigm with Eye Tracking Background Set-up Lab
 iworx Physiology Lab Experiment Experiment HP-17 The Gaze Cueing Paradigm with Eye Tracking Background Set-up Lab Note: The lab presented here is intended for evaluation purposes only. iworx users should
iworx Physiology Lab Experiment Experiment HP-17 The Gaze Cueing Paradigm with Eye Tracking Background Set-up Lab Note: The lab presented here is intended for evaluation purposes only. iworx users should
Introduction to EMG. Figure 1
 Experiment 13 An electromyogram, or EMG, is a graphical recording of electrical activity within muscles. Activation of muscles by nerves results in changes in ion flow across cell membranes, which generates
Experiment 13 An electromyogram, or EMG, is a graphical recording of electrical activity within muscles. Activation of muscles by nerves results in changes in ion flow across cell membranes, which generates
BP-600 Noninvasive Blood Pressure Sensor
 Technical Note BP-600 Overview A person's cardiac output, peripheral vascular resistance, blood pressure, and other cardiovascular parameters change in response to the activities and events taking place
Technical Note BP-600 Overview A person's cardiac output, peripheral vascular resistance, blood pressure, and other cardiovascular parameters change in response to the activities and events taking place
Adventure 3 Plus Service Manual
 Adventure 3 Plus Service Manual 1 TABLE OF CONTENTS CHAPTER 1: SERIAL NUMBER LOCATION...3 CHAPTER 2: PREVENTATIVE MAINTENANCE 2.1 Preventative Maintenance. 4 2.2 Tension and Centering the Running Belt....6
Adventure 3 Plus Service Manual 1 TABLE OF CONTENTS CHAPTER 1: SERIAL NUMBER LOCATION...3 CHAPTER 2: PREVENTATIVE MAINTENANCE 2.1 Preventative Maintenance. 4 2.2 Tension and Centering the Running Belt....6
Practical Guidelines For the Enraf-Nonius Commercial Training Myomed 632
 Practical Guidelines For the Enraf-Nonius Commercial Training Myomed 632 Copyright: & Exclusive Sales and Service: Enraf-Nonius B.V. P.O. Box 12080 3004 GB ROTTERDAM The Netherlands Tel: +31 (0)10 20 30
Practical Guidelines For the Enraf-Nonius Commercial Training Myomed 632 Copyright: & Exclusive Sales and Service: Enraf-Nonius B.V. P.O. Box 12080 3004 GB ROTTERDAM The Netherlands Tel: +31 (0)10 20 30
Experiment HP-13: The Gaze Cue Paradigm
 Experiment HP-13: The Gaze Cue Paradigm Background During almost all social interactions, people s eyes convey information about their direction of attention as well as their emotional and mental status.
Experiment HP-13: The Gaze Cue Paradigm Background During almost all social interactions, people s eyes convey information about their direction of attention as well as their emotional and mental status.
Red Light - Green Light
 iworx Physiology Lab Experiment Experiment HN-9 Red Light - Green Light Background Setup Lab Note: The lab presented here is intended for evaluation purposes only. iworx users should refer to the User
iworx Physiology Lab Experiment Experiment HN-9 Red Light - Green Light Background Setup Lab Note: The lab presented here is intended for evaluation purposes only. iworx users should refer to the User
Figure HN-5-L1: The Sequence menu containing the preprogramming light sequences used in this experiment.
 HN-5: Visual Reflexes and Color Stimulation Exercise 1: Reaction Time and Single Color Visual Cues Aim: To measure the reaction time of a subject to a visual cue. Procedure 1. Instruct the subject to:
HN-5: Visual Reflexes and Color Stimulation Exercise 1: Reaction Time and Single Color Visual Cues Aim: To measure the reaction time of a subject to a visual cue. Procedure 1. Instruct the subject to:
SpikerBox Neural Engineering Workshop
 SpikerBox Neural Engineering Workshop A Workshop Curriculum for Grades 9-12 Developed as a two-day, two hours/day workshop Developed by UW Graduate Students: Stephanie Seeman, Bethany Kondiles, and Katherine
SpikerBox Neural Engineering Workshop A Workshop Curriculum for Grades 9-12 Developed as a two-day, two hours/day workshop Developed by UW Graduate Students: Stephanie Seeman, Bethany Kondiles, and Katherine
Cardiovascular Physiology Laboratory Clive M. Baumgarten, Ph.D.
 Cardiovascular Physiology Laboratory Clive M. Baumgarten, Ph.D. To pump blood, the heart has a rhythmical sequence of both electrical and mechanical events, the cardiac cycle. The electrical activity,
Cardiovascular Physiology Laboratory Clive M. Baumgarten, Ph.D. To pump blood, the heart has a rhythmical sequence of both electrical and mechanical events, the cardiac cycle. The electrical activity,
Posner s Attention Test
 iworx Physiology Lab Experiment Experiment HP-18 Posner s Attention Test Background Setup Lab Note: The lab presented here is intended for evaluation purposes only. iworx users should refer to the User
iworx Physiology Lab Experiment Experiment HP-18 Posner s Attention Test Background Setup Lab Note: The lab presented here is intended for evaluation purposes only. iworx users should refer to the User
841T. Adventure4 Adventure5 TREADMILL OWNER S MANUAL
 841T Adventure4 Adventure5 TREADMILL OWNER S MANUAL Read the treadmill guide before using this owner s manual. ASSEMBLY WARNING There are several areas during the assembly process that special attention
841T Adventure4 Adventure5 TREADMILL OWNER S MANUAL Read the treadmill guide before using this owner s manual. ASSEMBLY WARNING There are several areas during the assembly process that special attention
LABORATORY INVESTIGATION
 LABORATORY INVESTIGATION Recording Electrocardiograms The taking of an electrocardiogram is an almost universal part of any complete physical examination. From the ECG record of the electrical activity
LABORATORY INVESTIGATION Recording Electrocardiograms The taking of an electrocardiogram is an almost universal part of any complete physical examination. From the ECG record of the electrical activity
QUICK START GUIDE TYMPANOMETRY AND AUDIOMETRY FOR SENTIERO DESKTOP DEVICES
 DEVICE CONNECTORS On/off button Headphones or second transducer Power supply Air pump USB cable Label printer Patient response Probe button Make sure all your appropriate transducers are connected (Tymp
DEVICE CONNECTORS On/off button Headphones or second transducer Power supply Air pump USB cable Label printer Patient response Probe button Make sure all your appropriate transducers are connected (Tymp
Experiment B-44 Muscle Strength
 1 Experiment B-44 Muscle Strength Objectives To learn about the skeletal muscles. To find correlations between dominant, gender and more to grip strength. Modules and Sensors PC + NeuLog application USB-200
1 Experiment B-44 Muscle Strength Objectives To learn about the skeletal muscles. To find correlations between dominant, gender and more to grip strength. Modules and Sensors PC + NeuLog application USB-200
ECE ECE PRINCIPLES OF BIOMEDICAL SYSTEMS & DEVICES LAB 1 - ELECTROCARDIOGRAM
 ECE0909.404.01 ECE 0909.504.03 PRINCIPLES OF BIOMEDICAL SYSTEMS & DEVICES LAB 1 - ELECTROCARDIOGRAM The purpose of this laboratory is to introduce you to electrocardiogram, its acquisition and interpretation.
ECE0909.404.01 ECE 0909.504.03 PRINCIPLES OF BIOMEDICAL SYSTEMS & DEVICES LAB 1 - ELECTROCARDIOGRAM The purpose of this laboratory is to introduce you to electrocardiogram, its acquisition and interpretation.
Iso-inertial dynamometer
 Iso-inertial dynamometer A. Equipment set up B. Data acquisition and export C. Training mode 1 P a g e A. Equipment set up 1. Connect all the devices as depicted below: 2. Launch the software by clicking
Iso-inertial dynamometer A. Equipment set up B. Data acquisition and export C. Training mode 1 P a g e A. Equipment set up 1. Connect all the devices as depicted below: 2. Launch the software by clicking
Adventure 5 Plus Service Manual
 Adventure 5 Plus Service Manual 1 TABLE OF CONTENTS CHAPTER 1: SERIAL NUMBER LOCATION...3 CHAPTER 2: PREVENTATIVE MAINTENANCE 2.1 Preventative Maintenance. 4 2.2 Tension and Centering the Running Belt....6
Adventure 5 Plus Service Manual 1 TABLE OF CONTENTS CHAPTER 1: SERIAL NUMBER LOCATION...3 CHAPTER 2: PREVENTATIVE MAINTENANCE 2.1 Preventative Maintenance. 4 2.2 Tension and Centering the Running Belt....6
HUMAN ANATOMY AND PHYSIOLOGY
 HUMAN ANATOMY AND PHYSIOLOGY NAME Detection of heart sounds. Clean the ear pieces of the stethoscope before using. The ear pieces should be pointing slightly forward when inserted into the ears because
HUMAN ANATOMY AND PHYSIOLOGY NAME Detection of heart sounds. Clean the ear pieces of the stethoscope before using. The ear pieces should be pointing slightly forward when inserted into the ears because
Emotional State Recognition via Physiological Measurement and Processing
 APPLICATION NOTE 42 Aero Camino, Goleta, CA 93117 Tel (805) 685-0066 Fax (805) 685-0067 info@biopac.com www.biopac.com Application Note 276: Emotional State Recognition via Physiological Measurement and
APPLICATION NOTE 42 Aero Camino, Goleta, CA 93117 Tel (805) 685-0066 Fax (805) 685-0067 info@biopac.com www.biopac.com Application Note 276: Emotional State Recognition via Physiological Measurement and
Evolve 3 & 5 Service Manual
 Evolve 3 & 5 Service Manual 1 Product Browse 2 Contents CHAPTER 1: SERIAL NUMBER LOCATION... 5 CHAPTER 2: CONSOLE INSTRUCTIONS 2.1 Console Overview... 6 2.1.1 Evolve 3 Console Overview... 6 2.1.2 Evolve
Evolve 3 & 5 Service Manual 1 Product Browse 2 Contents CHAPTER 1: SERIAL NUMBER LOCATION... 5 CHAPTER 2: CONSOLE INSTRUCTIONS 2.1 Console Overview... 6 2.1.1 Evolve 3 Console Overview... 6 2.1.2 Evolve
