Vascular parkinsonism characteristics, pathogenesis and treatment
|
|
|
- Abner Norman
- 6 years ago
- Views:
Transcription
1 Vascular parkinsonism characteristics, pathogenesis and treatment Amos D. Korczyn Abstract Parkinson disease is a primary degenerative disease of the brain, but parkinsonism can also result from a variety of vascular disorders. Vascular parkinsonism (VP) most frequently presents as lower body parkinsonism, a condition that is accompanied by the development of white matter lesions (WMLs) and lacunes in the brain. Patients with lower body parkinsonism exhibit gait impairment and go on to develop urinary incontinence, abnormal pyramidal responses and cognitive decline. However, WMLs and lacunes are also common observations among elderly individuals who do not have parkinsonism, which causes difficulty in determining the pathogenetic mechanisms that lead to VP. In addition, imaging studies suggest that many pathological and clinical features are common to VP and Binswanger disease, a type of small vessel vascular dementia. This Review summarizes current understanding of the clinical characteristics of VP, as well as knowledge gained from neuroimaging and nuclear imaging of the pathological features of VP. The lack of current treatment options, and the emergence of new therapies such as cerebrospinal fluid drainage, are also discussed. Finally, consideration is given to whether the overlap between VP and Binswanger disease means that these two disorders should be considered as part of the same disease entity. Korczyn, A. D. Nat. Rev. Neurol. advance online publication 28 April 2015; doi: /nrneurol REVIEWS Department of Neurology, Sackler Faculty of Medicine, Tel Aviv University, Ramat Aviv 69978, Israel. amoskor@post.tau.ac.il Introduction Parkinsonism is associated with several aetiologies besides primary neurodegenerative processes, including a variety of vascular mechanisms. 1 The possibility that parkinsonism could result from vascular changes in the brain was suggested in a study published in 1929, which identified a condition that was named in the study as arterio sclerotic parkinsonism; this condition was described as being distinct from idiopathic (sporadic) Parkinson disease (PD). 2 Patients with arteriosclerotic parkinsonism primarily exhibited impairment of gait, although individuals with more-advanced disease displayed additional features (Box 1). The hypothesis that vascular changes in the brain could result in parkinsonism was not widely accepted by the neurological community until other studies proposed similar ideas several decades later. A study published in 1987 suggested that Binswanger disease (also known as subcortical arteriosclerotic encephalopathy), in which thickening and narrowing of arteries in the brain leads to white matter lesions (WMLs) and dementia, was a prominent cause of a gait disorder, referred to as lowerhalf parkinsonism, among elderly individuals. 3 In a subsequent study, the term vascular parkinsonism (VP) was used to describe a clinical phenotype that was primarily characterized by impairment of gait, and was associated with vascular pathogenesis that predominantly affected subcortical white matter. 4 Further evidence for this vascular pathogenesis came from MRI, which identified Competing interests The author declares no competing interests. WMLs in the brains of patients with impairment of gait. 5 Studies of patients with this phenotype also provided the important observation that treatment with levodopa did little to improve their gait. 4,6 Brain lesions with a vascular pathogenesis other than WMLs can also lead to parkinsonian features. One of the first pathological demonstrations of stroke-induced parkinsonism comprised a case of unilateral parkinsonism that developed shortly after an episode of brainstem ischaemia. 7 In contrast to the frequent observation that a nigral lesion results in contralateral parkinsonism, in this case the nigral lesion was ipsilateral to the hemiparkinsonism. Another patient experienced unilateral parkinsonism that followed a contralateral lacunar infarct; 8 such parkinsonism is often a manifestation of midbrain lesions. In other cases, parkinsonian syndromes have developed subacutely and improved spontaneously; brain CT scans demonstrated the existence of basal ganglia infarcts, and led to the conclusion that these individuals had VP, despite no history of stroke and the absence of long-tract changes. 9 Unilateral parkinsonism has also been seen following a contralateral brainstem haemorrhagic stroke. 10 Similar findings have been reported in other studies. 11,12 However, in contrast to the impairment of gait observed with WMLs, patients with a vascular pathogenesis other than WMLs all presented with unilateral disease, and gait impairment was not prominent. Vascular lesions are observed in some patients with idiopathic PD, but whether the occurrence of these lesions is coincidental or a contributing factor to the NATURE REVIEWS NEUROLOGY ADVANCE ONLINE PUBLICATION 1
2 Key points Vascular changes in the brain can manifest as parkinsonism The predominant clinical feature in most cases of vascular parkinsonism (VP) is gait impairment, referred to as lower body parkinsonism As VP advances, patients develop cognitive impairment and incontinence The most prominent pathology observed by neuroimaging in patients with VP involves the white matter of the brain Patients with VP respond only slightly or not at all to dopaminergic medication Similar pathology and clinical features in VP and Binswanger disease suggest that they represent different manifestations along a disease spectrum clinical manifestations is unclear. Evidence suggests that vascular lesions contribute to cognitive impairment in PD. 13 Single lacunes in the basal ganglia seem to be irrelevant to the clinical picture, however, as they are frequently observed in otherwise healthy individuals. 14 Bilateral lesions in the basal ganglia are more likely than single lacunes to coexist with PD, 15 although even in this case, coexistent WMLs could have an important role in the development of parkinsonism. Several nonarteriosclerotic vascular aetiologies have been associated with parkinsonism; however, these aetiologies are all very rare, and white matter brain disease and/or lacunes of arteriosclerotic origin are thought to underlie most cases of VP. 16 The most frequent clinical presentation of VP is lower body parkinsonism. In most cases, this disorder is accompanied by WMLs, lacunar infarcts (either in the white matter or the basal ganglia) and ventricular dilatation. The condition manifests as a slow gait with short steps, which gradually worsens until patients become wheelchair-bound. Patients also frequently go on to develop urinary incontinence, abnormal pyramidal reflex responses and cognitive decline. Parkinsonian features and pyramidal signs in the upper part of the body are minimal. 3 5,16,17 Progressive impairment of gait among elderly individuals has attracted clinical attention for many years. Its aetiology has been largely unknown and, because of the paucity of other symptoms, a variety of terms have been suggested to describe such cases (Box 2). Lower body parkinsonism is common among elderly people, and could be the underlying cause in many cases of progressive impairment of gait. Box 1 Features of VP subtypes The following characteristics were defined by Critchley in Features common to all subtypes of VP Rigidity Masked face Short-stepped gait Additional features associated with more-advanced subtypes of VP Pseudobulbar manifestations Dementia and incontinence Pyramidal signs Cerebellar signs Abbreviation: VP, vascular parkinsonism. Here, VP is reviewed with a focus on lower body parkinsonism as its main manifestation. The clinical and pathological changes that are observed among patients with VP are summarized, and the difficulties in determining the contribution of these changes to the pathogenesis of VP are discussed in light of the fact that some of the pathologies are frequently observed among elderly individuals who do not have parkinsonism. The potential mechanisms through which vascular changes in the brain result in parkinsonism are considered, as is the potential for cerebrospinal fluid (CSF) drainage to offer an effective treatment for VP. Finally, similarities in the pathology of VP and Binswanger disease, and whether or not VP should be considered as a distinct entity, are discussed. Characteristics Regardless of the possible aetiologies and pathogenetic mechanisms of VP, patients with the condition exhibit a number of distinctive characteristics. In most cases, VP manifests as lower body parkinsonism, and several changes are seen in the brain, although these are not necessarily unique to VP. Epidemiology Vascular lesions are infrequent among patients with idiopathic PD and, given the age group that is predominantly affected, those that do occur might be considered a result of age rather than disease. 18,19 Studies of patients with postmortem-confirmed idiopathic PD indicate that vascular lesions occur at a low frequency ( %) in this group, 20 and that VP accounts for 3 5% of all patients with parkinsonism. 21 However, these data are subject to criticism because of the definition of VP used, and a referral bias in the population. For example, a restrictive definition of VP that includes only so-called pure lower body parkinsonism will miss cases in which dementia accompanies the impairment of gait. In contrast to these findings, in the UK Brain Bank clinicopathological study, 24 of 100 patients who had clinically confirmed PD had additional vascular changes that could have contributed to the clinical manifestations. 22 The age of onset of VP is similar to or slightly higher than that of idiopathic PD, 5,23 and the disorder more frequently affects males than it does females. This observation is probably a reflection of the fact that vascular disease is more frequent among men than among women. Clinical features Several clinical features are indicative of VP (Box 3), and the presence of multiple features allows a more definite diagnosis. The predominant manifestation of VP is lower body parkinsonism, which is mainly characterized by impaired gait. Affected individuals have a slow and insecure gait, similar to that observed among patients with PD, although the base (the distance between the feet) is not always as narrow in lower body parkinsonism as it is in idiopathic PD. 17,24 Posture is unstable, and postural responses to maintain balance are poor. 17,25 The legs are spastic rather than rigid, with brisk tendon reflexes (mainly in the knees), and extensor plantar responses are 2 ADVANCE ONLINE PUBLICATION
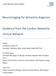
3 seen on one or both sides. 5,17,24 Clonus is seen in some patients, and foot movements are considerably slower than those of the upper limbs when affected individuals are sitting. 5,17 Patients also frequently exhibit freezing, 17 when they are temporarily unable to move, but the precise kinematic characteristics of the gait abnormalities in lower body parkinsonism have not been well defined. Gait ignition failure, in which patients experience difficulty in starting to walk, can be another albeit infrequent manifestation. 26 Clinical features other than impairment of gait can be present in lower body parkinsonism. Patients might have abnormal glabellar tap responses, seen as persistent blinking in response to repetitive tapping on the forehead, and other frontal release signs, such as snout or palmomental reflexes, are occasionally present. 25 Performance in Bender s face hand test, in which the patient tries to detect simultaneous touches on the cheek and hand while their eyes are closed, is frequently abnormal. Pseudobulbar symptoms are common, particularly among individuals with advanced disease, and the sense of smell might be lost in rare cases. 27 The upper limbs might also be affected in some patients (though considerably less so than the legs), with brisk tendon reflexes but no tremor. 5,17 The progression of lower body parkinsonism is variable. Onset is acute in some cases, after which symptoms sometimes improve spontaneously, but more commonly remain stable or deteriorate slowly. 9 In most cases, however, onset is insidious, and gait deteriorates slowly, along with gradual development of urinary difficulties and cognitive decline, unless these features are already present. 28 Patients do not report diurnal fluctuations of symptoms, and the condition shows little response to treatment with levodopa or dopamine agonists. 29 Pathology Vascular changes in the brain are the hallmarks of VP. These changes are usually ischaemic (although haemorrhagic changes might occur in rare cases) and affect areas of the brain that are relevant to parkinsonism, including subcortical white matter, basal ganglia, thalamus, and upper brainstem. 29,30 The precise changes in blood vessels have not yet been described in detail, but are expected to consist mainly of vessel wall changes, such as lipohyalinosis, that affect arterioles. To date, only a few pathological studies have focused on patients with VP. In the most detailed of these studies, the brain pathology of patients with VP was compared with that of healthy individuals, patients with idiopathic PD, and patients with Binswanger disease but without parkinsonism. 31 In this study, pallor of the substantia nigra was required for diagnosis of idiopathic PD, but excluded a diagnosis of VP. All patients with VP, as well as those with Binswanger disease but no parkinsonism, exhibited basal ganglia lacunes and extensive changes in the white matter, particularly of the frontal lobes. Similar pathologies were occasionally observed among patients with idiopathic PD, but were much less severe than in patients with VP or Binswanger disease. The white matter changes were accompanied by severe loss of oligodendrocytes. Box 2 Gait disorders in elderly individuals In the absence of aetiological information, various terms have been used to describe progressive gait impairment in the elderly population. Gait apraxia Senile gait High-level gait disorder Psychogenic gait Fear of falling Frontal lobe gait Box 3 Clinical features of vascular parkinsonism Age of onset >70 years Pyramidal signs Pseudobulbar palsy Cognitive impairment Absence of tremor Multiple vascular risk factors, such as hypertension, smoking and diabetes History of stroke Acute onset Rapid progression Poor response to dopaminergic therapy Although pathology is customarily assumed to be the gold standard for diagnosis, this assumption does not necessarily apply to VP, for a variety of reasons. First, no accepted criteria exist for the pathological diagnosis of VP, so autopsy findings cannot be used to corroborate or refute a diagnosis. Furthermore, cases that reach autopsy might not be representative of the general population, and are likely to include unusual cases. Second, MRI might be more sensitive than pathological examination in identifying WMLs, and can be used to quantify the damage (including changes in normal-appearing white matter) because it is conducted while patients are alive, and provides a view of the whole brain rather than the selective sample that is required for examination under the microscope. Microscopic examination is important, however, for identifying the underlying pathological process, such as in CADASIL (cerebral autosomal dominant arteriopathy with subcortical infarcts and leukoencephalopathy), which can manifest as parkinsonism. 32 On rare occasions, other vascular lesions, such as those caused by infection, 33 brain vasculitis 34 or Fabry disease, 35 could be responsible for parkinsonism. Changes detected by brain imaging The precise pathology of VP is unclear, but several changes in brain structure and function are frequently observed in patients with this condition. These changes are detectable with approaches such as MRI and nuclear imaging, which could provide information about the pathogenesis of VP, particularly when complemented by autopsy findings, and may be important indicators of disease progression. Lesions detected by MRI MRI of the brains of patients with lower body parkinsonism typically shows extensive lesions of the periventricular NATURE REVIEWS NEUROLOGY ADVANCE ONLINE PUBLICATION 3
4 Neuroimaging studies of VP to date have mainly used conventional imaging approaches. Advanced imaging techniques, such as diffusion tensor imaging, restingstate functional MRI, examination of normal-appearing white matter, high-field examinations (for example, 7 T MRI) and diffusional kurtosis imaging, could provide further insight into the pathogenesis and manifestations of VP. 42 One challenge in the use of MRI to diagnose VP is that different investigators use different nomenclatures when describing these lesions. Therefore, consensus standards for research into small vessel disease of the brain are urgently needed. 43 Figure 1 Brain MRI in a patient with parkinsonism. The T2 weighted image shows a section of the brain of a 63-yearold male with lower body parkinsonism. Disruption of the periventricular and deep white matter (white and black arrows, respectively) is seen. Permission obtained from Y. Balash, Tel Aviv Sourasky Medical Centre, Tel Aviv, Israel. white matter, the presence of lacunes (mainly in the basal ganglia), and dilatation of the lateral and third cerebral ventricles (Figure 1). 17,28 These changes accumulate over months and years, so quantifying the extent of these changes is of no benefit in establishing the diagnosis. By definition, the presence of vascular lesions in the brain is necessary for the diagnosis of VP, and such a diagnosis following a normal MRI result is unlikely. However, the presence of WMLs or lacunes in the brain is insufficient to make a diagnosis of VP, because these lesions are common among elderly people who are apparently unaffected by parkinsonism. In addition, in patients with idiopathic PD, the presence of these lesions does not correlate with gait abnormalities. 36 Even in elderly individuals with no cognitive impairment, the extent of WMLs corresponds to gait abnormalities and reduced gait velocity. 37,38 One study of individuals aged years who were not disabled and had changes in cerebral white matter showed that abnormal gait parameters correlated strongly with abnormal-appearing white matter on MRI scans, but not with changes in any specific brain region. 39 A subsequent study showed that elderly individuals with severe WMLs were particularly likely to exhibit deterioration of gait. 40 Thus, it seems that diffuse lesions throughout the brain white matter might initiate impairment of gait and cause the cardinal manifestations of VP as they increase in severity. 39 Similar WMLs have also been observed among elderly people with nonspecific cognitive impairments, including those of memory, attention and executiv e functions, perception and c onstruction, and general intelligence. 41 Changes detected by nuclear imaging The use of single-photon emission CT (SPECT) to visual ize presynaptic striatal dopamine transporters typically reveals reduced uptake of dopamine markers in the brains of patients with idiopathic PD compared with healthy individuals. Patients with lower body parkinsonism do not show the same reduction in uptake, as their nigrostriatal dopaminergic pathway is intact (Figure 2). Thus, for cases in which diagnosis is un certain, SPECT can be helpful. 27,44 46 If ischaemic damage affects the stria tum, however, the number of presynaptic dopaminergic terminals could be reduced a situ ation that could also account for a reduced SPECT signal. Thus, when SPECT scans show low signal intensity in the striatum, a combination of SPECT and structural MRI could help to confirm the reason for the reduced signal. Cardiac uptake of 123 I metaiodobenzylguanidine ( 123 I MIBG) into the noradrenergic nerves of the heart is characteristically reduced in idiopathic PD because the sympathetic nervous system is involved in this condition; uptake is less severely impaired or completely unaffected in VP. 27 SPECT can also be used to visualize postsynaptic D2 receptors, and this approach has proved useful in differentiating between idiopathic PD and conditions such as multiple system atrophy and progressive supranuclear palsy However, little evidence exists to suggest that SPECT imaging of D2 receptors is useful in the diagnosis of VP. EEG changes EEG changes in VP consist mainly of nonspecific slowing, similar to those seen in idiopathic PD, though less severe. 38 No pathognomonic changes have been described. Pathogenetic mechanisms Changes in the brain caused by ischaemia can take a variety of forms, which in turn can result in different clinical manifestations. For example, acute midbrain stroke can result in the immediate appearance of hemiparkinsonism that affects the upper and lower limbs and improves following treatment with levodopa. 10 Chronic ischaemic changes that affect the subcortical white matter might result in impairment of gait that progresses slowly and is not improved by dopaminergic medication ADVANCE ONLINE PUBLICATION
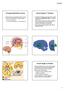
5 a Figure 2 SPECT imaging of the brain in patients with parkinsonism. The radioligand 123 I FP CIT was used to quantify dopamine transporter activity (yellow). a Brain SPECT scan from an 83-year-old woman with lower body parkinsonism, who had good uptake of 123 I FP CIT in the striatum and a poor response to levodopa therapy. b Brain SPECT scan from a 79-year-old woman with right hemiparkinsonism, who demonstrated markedly diminished uptake of 123 I FP CIT in the left striatum and a good response to levodopa therapy. Abbreviation: SPECT, single-photon emission CT. Permission obtained from M. Lorberboym, Department of Nuclear Medicine, Edith Wolfson Medical Centre, Sackler Faculty of Medicine, Tel Aviv University, Israel. b Patients with VP have vascular risk factors, such as hypertension, dyslipidaemia, diabetes mellitus, sleep apnoea and smoking. In such patients, neuroimaging identifies WMLs that are either hypodense on brain CT scans or hyperintense on T2 weighted MRI sequences. 28 Although WMLs are not always caused by vascular changes, 18 the association with vascular risk factors suggests a vascular aetiology. The frequent coexistence of lacunes, mostly in the basal ganglia and thalamus, 28 provides additional support for a vascular pathogenesis. Vascular lesions that affect the basal ganglia or the subcortical and deep white matter might affect the striatothalamocortical loops, leading to gait impairment and other features of lower body parkinsonism. 16,17 The WMLs seen in patients with VP probably result from repeated hypoxic or ischaemic episodes that lead to inflammatory responses in the affected brain areas. This inflammation induces proteases and generation of free radicals, both of which lead to opening of the blood brain barrier, extravasation of noxious elements and consequent myelin breakdown As already mentioned, although WMLs are the most easily demonstrable anatomical lesions in VP, basal ganglia lacunes are also frequently observed in this condition. 38 Both pathologies but particularly WMLs are common among elderly individuals who do not have parkinsonism, especially those who also have hypertension and other vascular risk factors, 38 leading to difficulty in ascertaining which pathology is responsible for the motor dysfunction observed among patients with VP. The basal ganglia are the immediate suspects when discussing parkinsonian symptoms, yet the gait impairment, pyramidal signs and urinary dysfunction observed in VP make frontal WMLs more likely candidates. Bradykinesia and other extrapyramidal features also point to involvement of the basal ganglia thalamocortical circuits. 37 Poor correlation between the information gained from brain imaging and the clinical manifestations of VP adds to the difficulties in determining which pathology is responsible for each of the clinical symptoms. A study published in 2012 that demonstrated disruption of the frontal lobe white matter in patients with VP supports the hypothesis of frontal or prefrontal cortex outflow damage. 54 One study that aimed to determine the location of WMLs that are responsible for abnormalities of gait found an association between WMLs and mild parkinsonian signs in a cohort of 430 patients aged years with intact cognition. 38 A similar study of 148 elderly individuals without dementia identified an additional relationship between poor executive function and WMLs. 55 Voxel-based pathological data confirmed that WMLs were accompanied in many cases by sub cortical lacunes in the basal ganglia and p articularly the thalamus, and that the WMLs and lacunes contributed independently to the parkinsonian signs. Thus, mixed pathology in parkinsonism does not seem to be unusual. 38 WMLs also develop during healthy ageing, and studies that prospectively examined healthy elderly individuals found that age-related WMLs correlated with gait and balance impairment, 38 and predicted the occurrence of depression. 55,56 These findings could indicate that VP is an extreme manifestation of the normal ageing process and is, therefore, very different from other forms of PD. 1 Determination of the factors that drive the progression of white matter changes until they cause neurological manifestations is now important. Another unresolved question is why the function of the upper limbs remains relatively spared as the disease progresses, even when the effect on the lower body is sufficient to render patients wheelchair-bound and incontinent. Treatment The appropriate choice of therapy for VP depends on the pathogenesis. If the lesions affect the nigro striatal pathways, dopaminergic therapy should be beneficial, but such cases are a minority. 57 For most patients, in whom the damage does not affect these pathways, current therapeutic approaches are disappointing. Levodopa, dopamine agonists and other traditional antiparkinsonian drugs are ineffective for VP that is caused by WMLs. 6,58 Subthalamic nucleus stimulation is similarly ineffective, 59 and physical therapy is also of limited value, although well-controlled studies have not been conducted. Therapy that minimizes the vascular risk factors is expected to slow down the rate of decline, but no studies have yet tested this hypothesis. Atherosclerotic brain disease is common among elderly individuals, and can also affect patients with idiopathic PD. These patients with so-called mixed disease are likely to respond to dopaminomimetic therapy, but to a lesser extent than patients with idiopathic PD and no atherosclerotic brain disease. An intriguing therapeutic option that has been suggested for VP is CSF drainage. 60 This approach is often NATURE REVIEWS NEUROLOGY ADVANCE ONLINE PUBLICATION 5
6 Box 4 White matter disease clinical manifestations Lower body parkinsonism Binswanger disease Depression Urinary incontinence Box 5 Risk factors for lower body parkinsonism A number of vascular risk factors for lower body parkinsonism have been identified, including: Hypertension Atherosclerosis Coronary artery disease Smoking Hyperhomocysteinaemia Diabetes mellitus Sleep apnoea Dyslipidaemia used to treat normal pressure hydrocephalus (NPH), a condition with similar symptoms to VP and Binswanger disease. As mentioned above, patients with VP typically have enlarged cerebral ventricles, which are also a symptom of NPH. 3 Impairment of gait is also clinically comparable in VP and NPH. 3,4 Furthermore, WMLs observed in Binswanger disease are remarkably similar to those observed in NPH. 61 Attempts to use CSF drainage to treat patients with VP have produced positive results. 60,62,63 A small doubleblind study published in 2011 supported these findings, although the follow-up period was just 3 months. 64 The use of repeated lumbar puncture or placement of a shunting tube for the treatment of patients with VP remains to be tested in controlled studies with large groups of participants and long follow-up periods. Such studies could determine the effectiveness of these approaches, and also indicate which patients are most or least likely to respond. Other treatment modalities, including repetitive trans cranial magnetic stimulation, have been suggested. One study has shown that this treatment can improve gait in patients with VP, 65 but these results should be considered preliminary. Is VP part of a disease spectrum? Extensive white matter disease of the brain results in disconnection and can affect many cerebral functions, but those that require coordination between many areas are presumably affected to the greatest extent. Such disconnection can result from inflammatory, traumatic, toxic or metabolic disorders, but probably the most common cause in the elderly is ischaemia caused by lesions of penetrating small arterioles. These changes typically progress slowly and result in a variety of clinical manifestations. The most common manifestations are cognitive decline and gait abnormalities, although other syndromes have also been described (Box 4). The existence of WMLs is currently required for a diagnosis of VP, but WMLs are not always associated with motor symptoms. Similar changes to white matter can occur in other conditions that are associated with the same vascular risk factors, so the manifestations of WMLs could be viewed as a spectrum disorder. For example, MRI shows similar WMLs among patients with VP to those seen among patients with Binswanger disease, in which WMLs are associated with dementia. 66 Patients with Binswanger disease also frequently display impaired gait and urinary incontinence, and little attempt has been made to distinguish between WMLs that are responsible for cognitive decline and those that are associated with impairment of gait. Thus, Binswanger disease and lower body parkinsonism have over lapping clinical features, risk factors and neuroimaging manifestations, and could, therefore, represent different manifestations of the same pathology. At advanced stages, patients with either disorder have complex symptoms that include dementia, severe impairment of gait and urinary incontinence, 67 and the initial diagnosis of VP or Binswanger disease often depends on the presentation. Prospective data are lacking, and longterm follow-up studies of these two groups of patients might reveal a common phenotype that combines f eatures of both. The reasons why some patients present with cognitive changes (and are, therefore, diagnosed with Binswanger disease) whereas others have predominant gait problems (and are, therefore, diagnosed with lower body parkinsonism) are unknown. The two conditions cannot c urrently be distinguished on the basis of neuroimaging. As patients with VP typically develop cognitive impairment, and those with Binswanger disease develop gait impairment, considering these changes as a spectrum disorder could be clinically important, as it suggests that, in both cases, patients should be treated to stop the progression of white matter disease by controllin g the risk factors (Box 5). Conclusions VP can occur as a consequence of discrete ischaemic or haemorrhagic strokes, but is most frequently a consequence of brain WMLs and basal ganglia lacunes. The overlap of symptoms and pathology between lower body parkinsonism and Binswanger disease suggests that these conditions belong to a single disease entity. Viewing these diseases as having the same risk factors and reaching the same final condition has important clinical implications. For example, patients should be warned of the likely deteri oration of their condition. Thus, those diagnosed with lower body parkinsonism should be warned not only that their mobility will further decline, but also that cognitive decline is possible, and advance directives should be discussed. Similarly, patients diagnosed with Binswanger disease should be advised to plan for a decline in mobility and to make the necessary adjustments in, for example, their professional life and house fittings. The possibility of depression, either due to lifestyle changes or the WMLs, needs to be considered in both conditions, as drug therapy for depression might be indicated. The pathological features of VP, and its unresponsiveness to treatment with dopaminergic drugs, distinguish 6 ADVANCE ONLINE PUBLICATION
7 this condition from idiopathic PD. Thus, the accurate diagnosis of VP relies on different diagnostic tools from those used in the diagnosis of idiopathic PD, for example, MRI rather than nuclear imaging. The therapeutic implications of an accurate diagnosis are considerable: dopaminergic drugs alleviate the symptoms of idiopathic PD but do not affect disease progression, whereas VP does not respond to dopaminergic drugs, but modification of the disease course could be possible through reduction of vascular risk factors. Studies are now required to determine the most effective approach. Review criteria Articles for inclusion in this Review were identified from a search of PubMed, using the following search terms: vascular parkinsonism, lower body parkinsonism, Parkinson disease + white matter, Parkinson disease + leukoaraiosis and Parkinson disease + magnetic resonance imaging. Papers published in English between 1980 and 2013 were selected for review and full-text versions were obtained when appropriate. Additional references were identified from the publication lists of identified papers. 1. Korczyn, A. D. Parkinson s disease: one disease entity or many? J. Neural Transm. Suppl. 56, (1999). 2. Critchley, M. Arteriosclerotic parkinsonism. Brain 52, (1929). 3. Thompson, P. D. & Marsden, C. D. Gait disorder of subcortical arteriosclerotic encephalopathy: Binswanger s disease. Mov. Disord. 2, 1 8 (1987). 4. FitzGerald, P. M. & Jankovic, J. Lower body parkinsonism: evidence for vascular etiology. Mov. Disord. 4, (1989). 5. Winikates, J. & Jankovic, J. Clinical correlates of vascular parkinsonism. Arch. Neurol. 56, (1999). 6. Parkes, J. D. et al. Parkinson s disease, cerebral arteriosclerosis, and senile dementia. Clinical features and response to levodopa. Q. J. Med. 43, (1974). 7. Hunter, R., Smith, J., Thomson, T. & Dayan, A. D. Hemiparkinsonism with infarction of the ipsilateral substantia nigra. Neuropathol. Appl. Neurobiol. 4, (1978). 8. Lazzarino, L. G., Nicolai, A. & Toppani, D. Subacute parkinsonism from a single lacunar infarct in the basal ganglia. Acta Neurol. (Napoli) 12, (1990). 9. Tolosa, E. S. & Santamaría, J. Parkinsonism and basal ganglia infarcts. Neurology 34, (1984). 10. Harik, S. I., Al-Hinti, J. T., Archer, R. L. & Angtuaco, E. J. Hemiparkinsonism after unilateral traumatic midbrain hemorrhage in a young woman. Neurol. Clin. Pract. 3, 4 7 (2013). 11. Reider-Groswasser, I., Bornstein, N. M. & Korczyn, A. D. Parkinsonism in patients with lacunar infarcts of the basal ganglia. Eur. Neurol. 35, (1995). 12. Ikeda, K,. Kotabe, T., Kanbashi, S. & Kinoshita, M. Parkinsonism in lacunar infarcts of the basal ganglia. Eur. Neurol. 36, (1996). 13. Korczyn, A. D. Vascular contribution to dementia in Parkinson s disease. Neurodegener. Dis. 7, (2010). 14. Inzelberg, R., Bornstein, N. M., Reider, I. & Korczyn, A. D. Basal ganglia lacunes and parkinsonism. Neuroepidemiology 13, (1994). 15. Bhatia, K. P. & Marsden, C. D. The behavioural and motor consequences of focal lesions of the basal ganglia in man. Brain 117, (1994). 16. Balash, Y. & Korczyn, A. D. Vascular parkinsonism. Handb. Clin. Neurol. 84, (2007). 17. Sibon, I., Fenelon, G., Quinn, N. P. & Tison, F. Vascular parkinsonism. J. Neurol. 251, (2004). 18. Leys, D. et al. Could Wallerian degeneration contribute to leuko-araiosis in subjects free of any vascular disorder? J. Neurol. Neurosurg. Psychiatry 54, (1991). 19. Bornstein, N. M., Halevy, G., Treves, T. & Korczyn, A. D. Cerebral atherosclerosis in Parkinsonian patients. Parkinsonism Relat. Disord. 4, (1998). 20. Jellinger, K. A. & Attems, J. Prevalence and impact of vascular and Alzheimer pathologies in Lewy body disease. Acta Neuropathol. 115, (2008). 21. Jellinger, K. A. Prevalence of cerebrovascular lesions in Parkinson s disease. A postmortem study. Acta Neuropathol. 105, (2003). 22. Hughes, A. J., Daniel, S. E., Blankson, S. & Lees, A. J. A clinicopathologic study of 100 cases of Parkinson s disease. Arch. Neurol. 50, (1993). 23. Glass, P. G. et al. The clinical features of pathologically confirmed vascular parkinsonism. J. Neurol. Neurosurg. Psychiatry 83, (2012). 24. Gupta, D. & Kuruvilla, A. Vascular parkinsonism: what makes it different? Postgrad. Med. J. 87, (2011). 25. Okuda, B., Kawabata, K., Tachibana, H., Kamogawa, K. & Okamoto, K. Primitive reflexes distinguish vascular parkinsonism from Parkinson s disease. Clin. Neurol. Neurosurg. 110, (2008). 26. Atchison, P. R., Thompson, P. D., Frackowiak, R. S. & Marsden, C. D. The syndrome of gait ignition failure: a report of six cases. Mov. Disord. 8, (1993). 27. Navarro-Otano, J. et al. 123 I MIBG cardiac uptake, smell identification and 123 I FP CIT SPECT in the differential diagnosis between vascular parkinsonism and Parkinson s disease. Parkinsonism Relat. Disord. 20, (2014). 28. Rektor, I. et al. Vascular pathology in patients with idiopathic Parkinson s disease. Parkinsonism Relat. Disord. 15, (2009). 29. Foltynie, T., Barker, R. & Brayne, C. Vascular parkinsonism: a review of the precision and frequency of the diagnosis. Neuroepidemiology 21, 1 7 (2002). 30. Zijlmans, J. C. et al. EEG findings in patients with vascular parkinsonism. Acta Neurol. Scand. 98, (1998). 31. Yamanouchi, H. & Nagura, H. Neurological signs and frontal white matter lesions in vascular parkinsonism. A clinicopathologic study. Stroke 28, (1997). 32. Wegner, F. et al. Vascular parkinsonism in a CADASIL case with an intact nigrostriatal dopaminergic system. J. Neurol. 254, (2007). 33. Carr, J. Parkinsonism secondary to neurosyphilis. Mov. Disord. 24, 1407 (2009). 34. Fabiani, G., Teive, H. A., Germiniani, F. M., Sá, D. S. & Werneck, L. C. Reversible parkinsonian syndrome in systemic and brain vasculitis. Mov. Disord. 17, (2002). 35. Buechner, S., De Cristofaro, M. T., Ramat, S. & Borsini, W. Parkinsonism and Anderson Fabry s disease: a case report. Mov. Disord. 21, (2006). 36. Antonini, A. et al. The relationship between cerebral vascular disease and parkinsonism: The VADO study. Parkinsonism Relat. Disord. 18, (2012). 37. Bohnen, N. I. & Albin, R. L. White matter lesions in Parkinson disease. Nat. Rev. Neurol. 7, (2011). 38. Baezner, H. et al. Association of gait and balance disorders with age-related white matter changes: the LADIS study. Neurology 70, (2008). 39. de Laat, K. F. et al. Cerebral white matter lesions and lacunar infarcts contribute to the presence of mild parkinsonian signs. Stroke 43, (2012). 40. Kreisel, S. H. et al. Deterioration of gait and balance over time: the effects of age-related white matter change the LADIS study. Cerebrovasc. Dis. 35, (2013). 41. Kloppenborg, R. P., Nederkoorn, P. J., Geerlings, M. I. & van den Berg, E. Presence and progression of white matter hyperintensities and cognition: a meta-analysis. Neurology 82, (2014). 42. van Norden, A. G. et al. Causes and consequences of cerebral small vessel disease. The RUN DMC study: a prospective cohort study. Study rationale and protocol. BMC Neurol. 11, 29 (2011). 43. Wardlaw, J. M. et al. Neuroimaging standards for research into small vessel disease and its contribution to ageing and neurodegeneration. Lancet Neurol. 12, (2013). 44. Lorberboym, M., Djaldetti, R., Melamed, E., Sadeh, M. & Lampl, Y. 123 I FP CIT SPECT imaging of dopamine transporters in patients with cerebrovascular disease and clinical diagnosis of vascular parkinsonism. J. Nucl. Med. 45, (2004). 45. Tzen, K. Y., Lu, C. S., Yen, T. C., Wey, S. P. & Ting, G. Differential diagnosis of Parkinson s disease and vascular parkinsonism by 99m Tc TRODAT 1. J. Nucl. Med. 42, (2001). 46. Brigo, F., Matinella, A., Erro, R. & Tinazzi, M. [¹²³I]FP-CIT SPECT (DaTSCAN) may be a useful tool to differentiate between Parkinson s disease and vascular or drug-induced parkinsonisms: a meta-analysis. Eur. J. Neurol. 21, 1369 e90 (2014). 47. Jakobson Mo, S. et al. Pre- and postsynaptic dopamine SPECT in idiopathic Parkinsonian diseases: a follow-up study. Biomed. Res. Int. 2013, (2013). 48. Oyanagi, C. et al. Comparison of striatal dopamine D2 receptors in Parkinson s disease and progressive supranuclear palsy patients using 123 I iodobenzofuran single-photon emission computed tomography. J. Neuroimaging 12, (2002). NATURE REVIEWS NEUROLOGY ADVANCE ONLINE PUBLICATION 7
8 49. Kim, Y. J. et al. Combination of dopamine transporter and D2 receptor SPECT in the diagnostic evaluation of PD, MSA, and PSP. Mov. Disord. 17, (2002). 50. Benítez-Rivero, S. et al. Clinical features and 123 I FP CIT SPECT imaging in vascular parkinsonism and Parkinson s disease. J. Neurol. Neurosurg. Psychiatry 84, (2013). 51. Jellinger, K. A. Vascular parkinsonism neuropathological findings. Acta Neurol. Scand. 105, (2002). 52. Auriel, E. et al. Clinical, radiological and pathological correlates of leukoaraiosis. Acta Neurol. Scand. 123, (2011). 53. Rosenberg, G. A., Bjerke, M. & Wallin, A. Multimodal markers of inflammation in the subcortical ischemic vascular disease type of vascular cognitive impairment. Stroke 45, (2014). 54. Wang, H. C., Hsu, J. L. & Leemans, A. Diffusion tensor imaging of vascular parkinsonism: structural changes in cerebral white matter and the association with clinical severity. Arch. Neurol. 69, (2012). 55. Murray., M. E. et al. Functional impact of white matter hyperintensities in cognitively normal elderly subjects. Arch. Neurol. 67, (2010). 56. Teodorczuk, A. et al. Relationship between baseline white-matter changes and development of late-life depressive symptoms: 3 year results from the LADIS study. Psychol. Med. 40, (2010). 57. Ling, M. J., Aggarwal, A. & Morris, J. G. Doparesponsive parkinsonism secondary to right temporal lobe haemorrhage. Mov. Disord. 17, (2002). 58. Zijlmans, J. C., Daniel, S. E., Hughes, A. J., Révész, T. & Lees, A. J. Clinicopathological investigation of vascular parkinsonism, including clinical criteria for diagnosis. Mov. Disord. 19, (2004). 59. Krack, P. et al. Ineffective subthalamic nucleus stimulation in levodopa-resistant postischemic parkinsonism. Neurology 54, (2000). 60. Espay, A. J., Narayan, R. K., Duker, A. P., Barrett, E. T. Jr & de Courten-Myers, G. Lowerbody parkinsonism: reconsidering the threshold for external lumbar drainage. Nat. Clin. Pract. Neurol. 4, (2008). 61. Tullberg, M. et al. White matter changes in normal pressure hydrocephalus and Binswanger disease: specificity, predictive value and correlations to axonal degeneration and demyelination. Acta Neurol. Scand. 105, (2002). 62. Akiguchi, I. et al. Shunt-responsive parkinsonism and reversible white matter lesions in patients with idiopathic NPH. J. Neurol. 255, (2008). 63. Ondo, W. G., Chan, L. L. & Levy, J. K. Vascular parkinsonism: clinical correlates predicting motor improvement after lumbar puncture. Mov. Disord. 17, (2002). 64. Tisell, M. et al. Shunt surgery in patients with hydrocephalus and white matter changes. J. Neurosurg. 114, (2011). 65. Yip, C. W. et al. A prospective pilot study of repetitive transcranial magnetic stimulation for gait dysfunction in vascular parkinsonism. Clin. Neurol. Neurosurg. 115, (2013). 66. Caplan, L. R. & Schoene, W. C. Clinical features of subcortical arteriosclerotic encephalopathy (Binswanger disease). Neurology 28, (1978). 67. Caplan, L. R. Binswanger disease revisited. Neurology 45, (1995). Acknowledgements I am grateful to Yacov Balash (Tel Aviv Sourasky Medical Centre, Tel-Aviv, Israel) for supplying Figure 1, and to Mordechai Lorberboym (Edith Wolfson Medical Centre, Sackler Faculty of Medicine, Tel Aviv University, Israel) for supplying Figure 2. 8 ADVANCE ONLINE PUBLICATION
Introduction, use of imaging and current guidelines. John O Brien Professor of Old Age Psychiatry University of Cambridge
 Introduction, use of imaging and current guidelines John O Brien Professor of Old Age Psychiatry University of Cambridge Why do we undertake brain imaging in AD and other dementias? Exclude other causes
Introduction, use of imaging and current guidelines John O Brien Professor of Old Age Psychiatry University of Cambridge Why do we undertake brain imaging in AD and other dementias? Exclude other causes
Transcranial sonography in movement disorders
 Transcranial sonography in movement disorders Uwe Walter 1st Residential Training of the European Society of Neurosonology and Cerebral Hemodynamics September 7-12, 2008 Bertinoro, Italy Department of
Transcranial sonography in movement disorders Uwe Walter 1st Residential Training of the European Society of Neurosonology and Cerebral Hemodynamics September 7-12, 2008 Bertinoro, Italy Department of
DIFFERENTIAL DIAGNOSIS SARAH MARRINAN
 Parkinson s Academy Registrar Masterclass Sheffield DIFFERENTIAL DIAGNOSIS SARAH MARRINAN 17 th September 2014 Objectives Importance of age in diagnosis Diagnostic challenges Brain Bank criteria Differential
Parkinson s Academy Registrar Masterclass Sheffield DIFFERENTIAL DIAGNOSIS SARAH MARRINAN 17 th September 2014 Objectives Importance of age in diagnosis Diagnostic challenges Brain Bank criteria Differential
Vascular parkinsonism an important cause of parkinsonism in older people
 B. Thanvi et al. Age and Ageing 2005; 34: 114 119 doi:10.1093/ageing/afi025 Age and Ageing Vol. 34 No. 2 British Geriatrics Society 2005; all rights reserved Vascular parkinsonism an important cause of
B. Thanvi et al. Age and Ageing 2005; 34: 114 119 doi:10.1093/ageing/afi025 Age and Ageing Vol. 34 No. 2 British Geriatrics Society 2005; all rights reserved Vascular parkinsonism an important cause of
III./3.1. Movement disorders with akinetic rigid symptoms
 III./3.1. Movement disorders with akinetic rigid symptoms III./3.1.1. Parkinson s disease Parkinson s disease (PD) is the second most common neurodegenerative disorder worldwide after Alzheimer s disease.
III./3.1. Movement disorders with akinetic rigid symptoms III./3.1.1. Parkinson s disease Parkinson s disease (PD) is the second most common neurodegenerative disorder worldwide after Alzheimer s disease.
Parkinson s Disease in the Elderly A Physicians perspective. Dr John Coyle
 Parkinson s Disease in the Elderly A Physicians perspective Dr John Coyle Overview Introduction Epidemiology and aetiology Pathogenesis Diagnosis and clinical features Treatment Psychological issues/ non
Parkinson s Disease in the Elderly A Physicians perspective Dr John Coyle Overview Introduction Epidemiology and aetiology Pathogenesis Diagnosis and clinical features Treatment Psychological issues/ non
Evolution of a concept: Apraxia/higher level gait disorder. ataxia v. apraxia gait = limb apraxia. low, middle, high gait disturbance levels
 Case #1 81-year-old woman Gait Imbalance: Two Unusual Cases in Older Patients February 2008: 3 years of gradually progressive gait imbalance no vertigo, dizziness or paresthesias etiology unclear on examination
Case #1 81-year-old woman Gait Imbalance: Two Unusual Cases in Older Patients February 2008: 3 years of gradually progressive gait imbalance no vertigo, dizziness or paresthesias etiology unclear on examination
Pathogenesis of Degenerative Diseases and Dementias. D r. Ali Eltayb ( U. of Omdurman. I ). M. Path (U. of Alexandria)
 Pathogenesis of Degenerative Diseases and Dementias D r. Ali Eltayb ( U. of Omdurman. I ). M. Path (U. of Alexandria) Dementias Defined: as the development of memory impairment and other cognitive deficits
Pathogenesis of Degenerative Diseases and Dementias D r. Ali Eltayb ( U. of Omdurman. I ). M. Path (U. of Alexandria) Dementias Defined: as the development of memory impairment and other cognitive deficits
Imaging biomarkers for Parkinson s disease
 3 rd Congress of the European Academy of Neurology Amsterdam, The Netherlands, June 24 27, 2017 Teaching Course 6 MDS-ES/EAN: Neuroimaging in movement disorders - Level 2 Imaging biomarkers for Parkinson
3 rd Congress of the European Academy of Neurology Amsterdam, The Netherlands, June 24 27, 2017 Teaching Course 6 MDS-ES/EAN: Neuroimaging in movement disorders - Level 2 Imaging biomarkers for Parkinson
Overview. Overview. Parkinson s disease. Secondary Parkinsonism. Parkinsonism: Motor symptoms associated with impairment in basal ganglia circuits
 Overview Overview Parkinsonism: Motor symptoms associated with impairment in basal ganglia circuits The differential diagnosis of Parkinson s disease Primary vs. Secondary Parkinsonism Proteinopathies:
Overview Overview Parkinsonism: Motor symptoms associated with impairment in basal ganglia circuits The differential diagnosis of Parkinson s disease Primary vs. Secondary Parkinsonism Proteinopathies:
Update on functional brain imaging in Movement Disorders
 Update on functional brain imaging in Movement Disorders Mario Masellis, MSc, MD, FRCPC, PhD Assistant Professor & Clinician-Scientist Sunnybrook Health Sciences Centre University of Toronto 53 rd CNSF
Update on functional brain imaging in Movement Disorders Mario Masellis, MSc, MD, FRCPC, PhD Assistant Professor & Clinician-Scientist Sunnybrook Health Sciences Centre University of Toronto 53 rd CNSF
MULTI SYSTEM ATROPHY: REPORT OF TWO CASES Dipu Bhuyan 1, Rohit Kr. Chandak 2, Pankaj Kr. Patel 3, Sushant Agarwal 4, Debjanee Phukan 5
 MULTI SYSTEM ATROPHY: REPORT OF TWO CASES Dipu Bhuyan 1, Rohit Kr. Chandak 2, Pankaj Kr. Patel 3, Sushant Agarwal 4, Debjanee Phukan 5 HOW TO CITE THIS ARTICLE: Dipu Bhuyan, Rohit Kr. Chandak, Pankaj Kr.
MULTI SYSTEM ATROPHY: REPORT OF TWO CASES Dipu Bhuyan 1, Rohit Kr. Chandak 2, Pankaj Kr. Patel 3, Sushant Agarwal 4, Debjanee Phukan 5 HOW TO CITE THIS ARTICLE: Dipu Bhuyan, Rohit Kr. Chandak, Pankaj Kr.
Corporate Medical Policy
 Corporate Medical Policy Dopamine Transporter Imaging with Single Photon Emission File Name: Origination: Last CAP Review: Next CAP Review: Last Review: dopamine_transporter_imaging_with_single_photon_emission_computed_tomography
Corporate Medical Policy Dopamine Transporter Imaging with Single Photon Emission File Name: Origination: Last CAP Review: Next CAP Review: Last Review: dopamine_transporter_imaging_with_single_photon_emission_computed_tomography
Vascular Dementia. Laura Pedelty, PhD MD The University of Illinois at Chicago and Jesse Brown VA Medical Center
 Vascular Dementia Laura Pedelty, PhD MD The University of Illinois at Chicago and Jesse Brown VA Medical Center none Disclosures Objectives To review the definition of Vascular Cognitive Impairment (VCI);
Vascular Dementia Laura Pedelty, PhD MD The University of Illinois at Chicago and Jesse Brown VA Medical Center none Disclosures Objectives To review the definition of Vascular Cognitive Impairment (VCI);
Evaluation of Parkinson s Patients and Primary Care Providers
 Evaluation of Parkinson s Patients and Primary Care Providers 2018 Movement Disorders Half Day Symposium Elise Anderson MD Medical Co-Director, PBSI Movement Disorders 6/28/2018 1 Disclosures GE Speaker,
Evaluation of Parkinson s Patients and Primary Care Providers 2018 Movement Disorders Half Day Symposium Elise Anderson MD Medical Co-Director, PBSI Movement Disorders 6/28/2018 1 Disclosures GE Speaker,
Dementia Update. October 1, 2013 Dylan Wint, M.D. Cleveland Clinic Lou Ruvo Center for Brain Health Las Vegas, Nevada
 Dementia Update October 1, 2013 Dylan Wint, M.D. Cleveland Clinic Lou Ruvo Center for Brain Health Las Vegas, Nevada Outline New concepts in Alzheimer disease Biomarkers and in vivo diagnosis Future trends
Dementia Update October 1, 2013 Dylan Wint, M.D. Cleveland Clinic Lou Ruvo Center for Brain Health Las Vegas, Nevada Outline New concepts in Alzheimer disease Biomarkers and in vivo diagnosis Future trends
Parkinson s Disease. Prevalence. Mark S. Baron, M.D. Cardinal Features. Clinical Characteristics. Not Just a Movement Disorder
 Prevalence Parkinson s Disease Mark S. Baron, M.D. Associate Professor of Neurology Movement Disorders Section VCU School of Medicine Common disorder Approaching 1% by 65 yrs of age, 2% by 80 yrs of age
Prevalence Parkinson s Disease Mark S. Baron, M.D. Associate Professor of Neurology Movement Disorders Section VCU School of Medicine Common disorder Approaching 1% by 65 yrs of age, 2% by 80 yrs of age
Differential Diagnosis of Hypokinetic Movement Disorders
 Differential Diagnosis of Hypokinetic Movement Disorders Dr Donald Grosset Consultant Neurologist - Honorary Professor Institute of Neurological Sciences - Glasgow University Hypokinetic Parkinson's Disease
Differential Diagnosis of Hypokinetic Movement Disorders Dr Donald Grosset Consultant Neurologist - Honorary Professor Institute of Neurological Sciences - Glasgow University Hypokinetic Parkinson's Disease
Neurodegenerative Disease. April 12, Cunningham. Department of Neurosciences
 Neurodegenerative Disease April 12, 2017 Cunningham Department of Neurosciences NEURODEGENERATIVE DISEASE Any of a group of hereditary and sporadic conditions characterized by progressive dysfunction,
Neurodegenerative Disease April 12, 2017 Cunningham Department of Neurosciences NEURODEGENERATIVE DISEASE Any of a group of hereditary and sporadic conditions characterized by progressive dysfunction,
FILE / PERIVENTRICULAR MICROVASCULAR ISCHEMIC CHANGES EBOOK
 07 June, 2018 FILE / PERIVENTRICULAR MICROVASCULAR ISCHEMIC CHANGES EBOOK Document Filetype: PDF 365.11 KB 0 FILE / PERIVENTRICULAR MICROVASCULAR ISCHEMIC CHANGES EBOOK I recently had a MRI last week with
07 June, 2018 FILE / PERIVENTRICULAR MICROVASCULAR ISCHEMIC CHANGES EBOOK Document Filetype: PDF 365.11 KB 0 FILE / PERIVENTRICULAR MICROVASCULAR ISCHEMIC CHANGES EBOOK I recently had a MRI last week with
Patient with vertigo, dizziness and depression
 Clinical Case - Test Yourself Neuro/Head and Neck Radiology Patient with vertigo, dizziness and depression Michael Mantatzis, Paraskevi Argyropoulou, Panos Prassopoulos Radiology Department, Democritus
Clinical Case - Test Yourself Neuro/Head and Neck Radiology Patient with vertigo, dizziness and depression Michael Mantatzis, Paraskevi Argyropoulou, Panos Prassopoulos Radiology Department, Democritus
brain MRI for neuropsychiatrists: what do you need to know
 brain MRI for neuropsychiatrists: what do you need to know Christoforos Stoupis, MD, PhD Department of Radiology, Spital Maennedorf, Zurich & Inselspital, University of Bern, Switzerland c.stoupis@spitalmaennedorf.ch
brain MRI for neuropsychiatrists: what do you need to know Christoforos Stoupis, MD, PhD Department of Radiology, Spital Maennedorf, Zurich & Inselspital, University of Bern, Switzerland c.stoupis@spitalmaennedorf.ch
Parts of the motor circuits
 MOVEMENT DISORDERS Parts of the motor circuits cortical centers: there are centers in all the cortical lobes subcortical centers: caudate nucleus putamen pallidum subthalamical nucleus (Luys) nucleus ruber
MOVEMENT DISORDERS Parts of the motor circuits cortical centers: there are centers in all the cortical lobes subcortical centers: caudate nucleus putamen pallidum subthalamical nucleus (Luys) nucleus ruber
Brain imaging for the diagnosis of people with suspected dementia
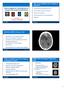 Why do we undertake brain imaging in dementia? Brain imaging for the diagnosis of people with suspected dementia Not just because guidelines tell us to! Exclude other causes for dementia Help confirm diagnosis
Why do we undertake brain imaging in dementia? Brain imaging for the diagnosis of people with suspected dementia Not just because guidelines tell us to! Exclude other causes for dementia Help confirm diagnosis
Parkinson Disease. Lorraine Kalia, MD, PhD, FRCPC. Presented by: Ontario s Geriatric Steering Committee
 Parkinson Disease Lorraine Kalia, MD, PhD, FRCPC Key Learnings Parkinson Disease (L. Kalia) Key Learnings Parkinson disease is the most common but not the only cause of parkinsonism Parkinson disease is
Parkinson Disease Lorraine Kalia, MD, PhD, FRCPC Key Learnings Parkinson Disease (L. Kalia) Key Learnings Parkinson disease is the most common but not the only cause of parkinsonism Parkinson disease is
Dementia and Healthy Ageing : is the pathology any different?
 Dementia and Healthy Ageing : is the pathology any different? Professor David Mann, Professor of Neuropathology, University of Manchester, Hope Hospital, Salford DEMENTIA Loss of connectivity within association
Dementia and Healthy Ageing : is the pathology any different? Professor David Mann, Professor of Neuropathology, University of Manchester, Hope Hospital, Salford DEMENTIA Loss of connectivity within association
United Council for Neurologic Subspecialties Geriatric Neurology Written Examination Content Outline
 United Council for Neurologic Subspecialties Geriatric Neurology Written Examination Content Outline REV 3/24/09 The UCNS Geriatric Neurology examination was established to determine the level of competence
United Council for Neurologic Subspecialties Geriatric Neurology Written Examination Content Outline REV 3/24/09 The UCNS Geriatric Neurology examination was established to determine the level of competence
Non Alzheimer Dementias
 Non Alzheimer Dementias Randolph B Schiffer Department of Neuropsychiatry and Behavioral Science Texas Tech University Health Sciences Center 9/11/2007 Statement of Financial Disclosure Randolph B Schiffer,,
Non Alzheimer Dementias Randolph B Schiffer Department of Neuropsychiatry and Behavioral Science Texas Tech University Health Sciences Center 9/11/2007 Statement of Financial Disclosure Randolph B Schiffer,,
Delirium & Dementia. Nicholas J. Silvestri, MD
 Delirium & Dementia Nicholas J. Silvestri, MD Outline Delirium vs. Dementia Neural pathways relating to consciousness Encephalopathy Stupor Coma Dementia Delirium vs. Dementia Delirium Abrupt onset Lasts
Delirium & Dementia Nicholas J. Silvestri, MD Outline Delirium vs. Dementia Neural pathways relating to consciousness Encephalopathy Stupor Coma Dementia Delirium vs. Dementia Delirium Abrupt onset Lasts
Parkinson s Disease. Sirilak yimcharoen
 Parkinson s Disease Sirilak yimcharoen EPIDEMIOLOGY ~1% of people over 55 years Age range 35 85 years peak age of onset is in the early 60s ~5% of cases characterized by an earlier age of onset (typically
Parkinson s Disease Sirilak yimcharoen EPIDEMIOLOGY ~1% of people over 55 years Age range 35 85 years peak age of onset is in the early 60s ~5% of cases characterized by an earlier age of onset (typically
Acute stroke. Ischaemic stroke. Characteristics. Temporal classification. Clinical features. Interpretation of Emergency Head CT
 Ischaemic stroke Characteristics Stroke is the third most common cause of death in the UK, and the leading cause of disability. 80% of strokes are ischaemic Large vessel occlusive atheromatous disease
Ischaemic stroke Characteristics Stroke is the third most common cause of death in the UK, and the leading cause of disability. 80% of strokes are ischaemic Large vessel occlusive atheromatous disease
The motor regulator. 1) Basal ganglia/nucleus
 The motor regulator 1) Basal ganglia/nucleus Neural structures involved in the control of movement Basal Ganglia - Components of the basal ganglia - Function of the basal ganglia - Connection and circuits
The motor regulator 1) Basal ganglia/nucleus Neural structures involved in the control of movement Basal Ganglia - Components of the basal ganglia - Function of the basal ganglia - Connection and circuits
EMERGING TREATMENTS FOR PARKINSON S DISEASE
 EMERGING TREATMENTS FOR PARKINSON S DISEASE Katerina Markopoulou, MD, PhD Director Neurodegenerative Diseases Program Department of Neurology NorthShore University HealthSystem Clinical Assistant Professor
EMERGING TREATMENTS FOR PARKINSON S DISEASE Katerina Markopoulou, MD, PhD Director Neurodegenerative Diseases Program Department of Neurology NorthShore University HealthSystem Clinical Assistant Professor
Subthalamic Nucleus Deep Brain Stimulation (STN-DBS)
 Subthalamic Nucleus Deep Brain Stimulation (STN-DBS) A Neurosurgical Treatment for Parkinson s Disease Parkinson s Disease Parkinson s disease is a common neurodegenerative disorder that affects about
Subthalamic Nucleus Deep Brain Stimulation (STN-DBS) A Neurosurgical Treatment for Parkinson s Disease Parkinson s Disease Parkinson s disease is a common neurodegenerative disorder that affects about
PSYC& 100: Biological Psychology (Lilienfeld Chap 3) 1
 PSYC& 100: Biological Psychology (Lilienfeld Chap 3) 1 1 What is a neuron? 2 Name and describe the functions of the three main parts of the neuron. 3 What do glial cells do? 4 Describe the three basic
PSYC& 100: Biological Psychology (Lilienfeld Chap 3) 1 1 What is a neuron? 2 Name and describe the functions of the three main parts of the neuron. 3 What do glial cells do? 4 Describe the three basic
Basal ganglia Sujata Sofat, class of 2009
 Basal ganglia Sujata Sofat, class of 2009 Basal ganglia Objectives Describe the function of the Basal Ganglia in movement Define the BG components and their locations Describe the motor loop of the BG
Basal ganglia Sujata Sofat, class of 2009 Basal ganglia Objectives Describe the function of the Basal Ganglia in movement Define the BG components and their locations Describe the motor loop of the BG
Laura Tormoehlen, M.D. Neurology and EM-Toxicology Indiana University
 Laura Tormoehlen, M.D. Neurology and EM-Toxicology Indiana University Disclosures! No conflicts of interest to disclose Neuroimaging 101! Plain films! Computed tomography " Angiography " Perfusion! Magnetic
Laura Tormoehlen, M.D. Neurology and EM-Toxicology Indiana University Disclosures! No conflicts of interest to disclose Neuroimaging 101! Plain films! Computed tomography " Angiography " Perfusion! Magnetic
Pietro Cortelli. IRCCS Istituto delle Scienze Neurologiche di Bologna DIBINEM, Alma Mater Studiorum - Università di Bologna
 Pietro Cortelli IRCCS Istituto delle Scienze Neurologiche di Bologna DIBINEM, Alma Mater Studiorum - Università di Bologna HYSTORY 1900 description of OPCA (Dejerine, Thomas) 1960 description of Shy-Drager
Pietro Cortelli IRCCS Istituto delle Scienze Neurologiche di Bologna DIBINEM, Alma Mater Studiorum - Università di Bologna HYSTORY 1900 description of OPCA (Dejerine, Thomas) 1960 description of Shy-Drager
Clinical Diagnosis. Step 1: Dementia or not? Diagnostic criteria for dementia (DSM-IV)
 Step 1: Dementia or not? Diagnostic criteria for dementia (DSM-IV) A. The development of multiple cognitive deficits manifested by both 1 and 2 1 1. Memory impairment 2. One (or more) of the following
Step 1: Dementia or not? Diagnostic criteria for dementia (DSM-IV) A. The development of multiple cognitive deficits manifested by both 1 and 2 1 1. Memory impairment 2. One (or more) of the following
Cerebral small vessel disease
 Cerebral small vessel disease What is it? What are the clinical syndromes? How do we diagnose it? What is the pathophysiology? New insights from genetics? Possible therapies? Small Vessel disease Changes
Cerebral small vessel disease What is it? What are the clinical syndromes? How do we diagnose it? What is the pathophysiology? New insights from genetics? Possible therapies? Small Vessel disease Changes
A. General features of the basal ganglia, one of our 3 major motor control centers:
 Reading: Waxman pp. 141-146 are not very helpful! Computer Resources: HyperBrain, Chapter 12 Dental Neuroanatomy Suzanne S. Stensaas, Ph.D. March 1, 2012 THE BASAL GANGLIA Objectives: 1. What are the main
Reading: Waxman pp. 141-146 are not very helpful! Computer Resources: HyperBrain, Chapter 12 Dental Neuroanatomy Suzanne S. Stensaas, Ph.D. March 1, 2012 THE BASAL GANGLIA Objectives: 1. What are the main
FDG-PET e parkinsonismi
 Parkinsonismi FDG-PET e parkinsonismi Valentina Berti Dipartimento di Scienze Biomediche, Sperimentali e Cliniche Sez. Medicina Nucleare Università degli Studi di Firenze History 140 PubMed: FDG AND parkinsonism
Parkinsonismi FDG-PET e parkinsonismi Valentina Berti Dipartimento di Scienze Biomediche, Sperimentali e Cliniche Sez. Medicina Nucleare Università degli Studi di Firenze History 140 PubMed: FDG AND parkinsonism
The Parkinson s You Can t See
 The Parkinson s You Can t See We principally see the motor phenomena of Parkinson's disease, but is there an early stage without visible features? Might this provide a window for disease-modifying therapy?
The Parkinson s You Can t See We principally see the motor phenomena of Parkinson's disease, but is there an early stage without visible features? Might this provide a window for disease-modifying therapy?
Neuroimaging for dementia diagnosis. Guidance from the London Dementia Clinical Network
 Neuroimaging for dementia diagnosis Guidance from the London Dementia Clinical Network Authors Dr Stephen Orleans-Foli Consultant Psychiatrist, West London Mental Health NHS Trust Dr Jeremy Isaacs Consultant
Neuroimaging for dementia diagnosis Guidance from the London Dementia Clinical Network Authors Dr Stephen Orleans-Foli Consultant Psychiatrist, West London Mental Health NHS Trust Dr Jeremy Isaacs Consultant
What if it s not Alzheimer s? Update on Lewy body dementia and frontotemporal dementia
 What if it s not Alzheimer s? Update on Lewy body dementia and frontotemporal dementia Dementia: broad term for any acquired brain condition impairing mental function such that ADLs are impaired. Includes:
What if it s not Alzheimer s? Update on Lewy body dementia and frontotemporal dementia Dementia: broad term for any acquired brain condition impairing mental function such that ADLs are impaired. Includes:
A. General features of the basal ganglia, one of our 3 major motor control centers:
 Reading: Waxman pp. 141-146 are not very helpful! Computer Resources: HyperBrain, Chapter 12 Dental Neuroanatomy Suzanne S. Stensaas, Ph.D. April 22, 2010 THE BASAL GANGLIA Objectives: 1. What are the
Reading: Waxman pp. 141-146 are not very helpful! Computer Resources: HyperBrain, Chapter 12 Dental Neuroanatomy Suzanne S. Stensaas, Ph.D. April 22, 2010 THE BASAL GANGLIA Objectives: 1. What are the
La neurosonologia. Ecografia cerebrale e nuove applicazioni nelle malattie neurodegenerative. Nelle patologie degenerative e vascolari cerebrali
 La neurosonologia Nelle patologie degenerative e vascolari cerebrali Andrea Pilotto Ecografia cerebrale e nuove applicazioni nelle malattie neurodegenerative Prof. Daniela Berg Department of Neurodegeneration
La neurosonologia Nelle patologie degenerative e vascolari cerebrali Andrea Pilotto Ecografia cerebrale e nuove applicazioni nelle malattie neurodegenerative Prof. Daniela Berg Department of Neurodegeneration
Movement Disorders. Psychology 372 Physiological Psychology. Background. Myasthenia Gravis. Many Types
 Background Movement Disorders Psychology 372 Physiological Psychology Steven E. Meier, Ph.D. Listen to the audio lecture while viewing these slides Early Studies Found some patients with progressive weakness
Background Movement Disorders Psychology 372 Physiological Psychology Steven E. Meier, Ph.D. Listen to the audio lecture while viewing these slides Early Studies Found some patients with progressive weakness
Cheyenne 11/28 Neurological Disorders II. Transmissible Spongiform Encephalopathy
 Cheyenne 11/28 Neurological Disorders II Transmissible Spongiform Encephalopathy -E.g Bovine4 Spongiform Encephalopathy (BSE= mad cow disease), Creutzfeldt-Jakob disease, scrapie (animal only) -Sporadic:
Cheyenne 11/28 Neurological Disorders II Transmissible Spongiform Encephalopathy -E.g Bovine4 Spongiform Encephalopathy (BSE= mad cow disease), Creutzfeldt-Jakob disease, scrapie (animal only) -Sporadic:
Dopamine Transporter Imaging with Single Photon Emission Computed Tomography
 Last Review Status/Date: September 2014 Page: 1 of 14 Photon Emission Computed Tomography Description Dopamine transporter imaging with single photon emission computed tomography (DAT-SPECT) is being evaluated
Last Review Status/Date: September 2014 Page: 1 of 14 Photon Emission Computed Tomography Description Dopamine transporter imaging with single photon emission computed tomography (DAT-SPECT) is being evaluated
The central nervous system
 Sectc.qxd 29/06/99 09:42 Page 81 Section C The central nervous system CNS haemorrhage Subarachnoid haemorrhage Cerebral infarction Brain atrophy Ring enhancing lesions MRI of the pituitary Multiple sclerosis
Sectc.qxd 29/06/99 09:42 Page 81 Section C The central nervous system CNS haemorrhage Subarachnoid haemorrhage Cerebral infarction Brain atrophy Ring enhancing lesions MRI of the pituitary Multiple sclerosis
17 th WORKSHOP ON NEUROOTOLOGY. Dementia & Imbalance. DR. ATRI CHATTERJEE Assistant Professor. Neurology VMMC & SafdarJung Hospital New Delhi
 17 th WORKSHOP ON NEUROOTOLOGY Dementia & Imbalance DR. ATRI CHATTERJEE Assistant Professor. Neurology VMMC & SafdarJung Hospital New Delhi ?Association Dementia Imbalance?Causation balance: An even distribution
17 th WORKSHOP ON NEUROOTOLOGY Dementia & Imbalance DR. ATRI CHATTERJEE Assistant Professor. Neurology VMMC & SafdarJung Hospital New Delhi ?Association Dementia Imbalance?Causation balance: An even distribution
Parkinsonism in corticobasal syndrome may not be primarily due to presynaptic dopaminergic deficiency
 Neurology Asia 2015; 20(1) : 23 27 Parkinsonism in corticobasal syndrome may not be primarily due to presynaptic dopaminergic deficiency 1 Ji Young Yun MD, 2 Jong-Min Kim MD, PhD, 3 Han-Joon Kim MD PhD,
Neurology Asia 2015; 20(1) : 23 27 Parkinsonism in corticobasal syndrome may not be primarily due to presynaptic dopaminergic deficiency 1 Ji Young Yun MD, 2 Jong-Min Kim MD, PhD, 3 Han-Joon Kim MD PhD,
How to Diagnose Early (Prodromal) Lewy Body Dementia. Ian McKeith MD, FRCPsych, F Med Sci.
 How to Diagnose Early (Prodromal) Lewy Body Dementia Ian McKeith MD, FRCPsych, F Med Sci. Parkinson s Disease Lewy Body Disease Time PD Dementia Lewy Body Dementias Dementia with Lewy Bodies (DLB) Diagnostic
How to Diagnose Early (Prodromal) Lewy Body Dementia Ian McKeith MD, FRCPsych, F Med Sci. Parkinson s Disease Lewy Body Disease Time PD Dementia Lewy Body Dementias Dementia with Lewy Bodies (DLB) Diagnostic
Clinical Features and Treatment of Parkinson s Disease
 Clinical Features and Treatment of Parkinson s Disease Richard Camicioli, MD, FRCPC Cognitive and Movement Disorders Department of Medicine University of Alberta 1 Objectives To review the diagnosis and
Clinical Features and Treatment of Parkinson s Disease Richard Camicioli, MD, FRCPC Cognitive and Movement Disorders Department of Medicine University of Alberta 1 Objectives To review the diagnosis and
Diagnosis and management of non-alzheimer dementias. Melissa Yu, M.D. Department of Neurology
 Diagnosis and management of non-alzheimer dementias Melissa Yu, M.D. Department of Neurology AGENDA Introduction When to think of alternate diagnoses Other forms of dementia Other reasons for confusion
Diagnosis and management of non-alzheimer dementias Melissa Yu, M.D. Department of Neurology AGENDA Introduction When to think of alternate diagnoses Other forms of dementia Other reasons for confusion
Objectives. RAIN Difficult Diagnosis 2014: A 75 year old woman with falls. Case History: First visit. Case History: First Visit
 Objectives RAIN Difficult Diagnosis 2014: A 75 year old woman with falls Alexandra Nelson MD, PhD UCSF Memory and Aging Center/Gladstone Institute of Neurological Disease Recognize important clinical features
Objectives RAIN Difficult Diagnosis 2014: A 75 year old woman with falls Alexandra Nelson MD, PhD UCSF Memory and Aging Center/Gladstone Institute of Neurological Disease Recognize important clinical features
DISORDERS OF THE MOTOR SYSTEM. Jeanette J. Norden, Ph.D. Professor Emerita Vanderbilt University School of Medicine
 DISORDERS OF THE MOTOR SYSTEM Jeanette J. Norden, Ph.D. Professor Emerita Vanderbilt University School of Medicine THE MOTOR SYSTEM To understand disorders of the motor system, we need to review how a
DISORDERS OF THE MOTOR SYSTEM Jeanette J. Norden, Ph.D. Professor Emerita Vanderbilt University School of Medicine THE MOTOR SYSTEM To understand disorders of the motor system, we need to review how a
T he main symptoms of idiopathic Parkinson s disease
 1211 PAPER How useful is [ 123 I]b-CIT SPECT in clinical practice? J Eerola, P J Tienari, S Kaakkola, P Nikkinen, J Launes... See end of article for authors affiliations... Correspondence to: Dr Johanna
1211 PAPER How useful is [ 123 I]b-CIT SPECT in clinical practice? J Eerola, P J Tienari, S Kaakkola, P Nikkinen, J Launes... See end of article for authors affiliations... Correspondence to: Dr Johanna
MUSCULOSKELETAL AND NEUROLOGICAL DISORDERS
 MUSCULOSKELETAL AND NEUROLOGICAL DISORDERS There are a wide variety of Neurologic and Musculoskeletal disorders which can impact driving safety. Impairment may be the result of altered muscular, skeletal,
MUSCULOSKELETAL AND NEUROLOGICAL DISORDERS There are a wide variety of Neurologic and Musculoskeletal disorders which can impact driving safety. Impairment may be the result of altered muscular, skeletal,
Dementia. Stephen S. Flitman, MD Medical Director 21st Century Neurology
 Dementia Stephen S. Flitman, MD Medical Director 21st Century Neurology www.neurozone.org Dementia is a syndrome Progressive memory loss, plus Progressive loss of one or more cognitive functions: Language
Dementia Stephen S. Flitman, MD Medical Director 21st Century Neurology www.neurozone.org Dementia is a syndrome Progressive memory loss, plus Progressive loss of one or more cognitive functions: Language
Falling and Forgetting
 Falling and Forgetting Dr TC Chan Dr LW Chu Inter-hosipital Geriatric Meeting 29-5-2009 Case 1 Mr A 86/M ADL-I Walk unaided Live with family Non smoker non drinker Good past health Attend A&E 10/2008 due
Falling and Forgetting Dr TC Chan Dr LW Chu Inter-hosipital Geriatric Meeting 29-5-2009 Case 1 Mr A 86/M ADL-I Walk unaided Live with family Non smoker non drinker Good past health Attend A&E 10/2008 due
Large artery: an important target for cerebral small vessel diseases
 Review Article Page 1 of 5 Large artery: an important target for cerebral small vessel diseases Wei-Hai Xu Department of Neurology, Peking Union Medical College Hospital, Chinese Academy of Medical Sciences
Review Article Page 1 of 5 Large artery: an important target for cerebral small vessel diseases Wei-Hai Xu Department of Neurology, Peking Union Medical College Hospital, Chinese Academy of Medical Sciences
Extrapyramidal Motor System. Basal Ganglia or Striatum. Basal Ganglia or Striatum 3/3/2010
 Extrapyramidal Motor System Basal Ganglia or Striatum Descending extrapyramidal paths receive input from other parts of motor system: From the cerebellum From the basal ganglia or corpus striatum Caudate
Extrapyramidal Motor System Basal Ganglia or Striatum Descending extrapyramidal paths receive input from other parts of motor system: From the cerebellum From the basal ganglia or corpus striatum Caudate
Dopamine Transporter Imaging With Single-Photon Emission Computed. Tomography
 Dopamine Transporter Imaging With Single-Photon Emission Computed Tomography Policy Number: 6.01.54 Last Review: 9/2017 Origination: 9/2015 Next Review: 9/2018 Policy Blue Cross and Blue Shield of Kansas
Dopamine Transporter Imaging With Single-Photon Emission Computed Tomography Policy Number: 6.01.54 Last Review: 9/2017 Origination: 9/2015 Next Review: 9/2018 Policy Blue Cross and Blue Shield of Kansas
Parkinson s Disease Initial Clinical and Diagnostic Evaluation. J. Timothy Greenamyre, MD, PhD
 Parkinson s Disease Initial Clinical and Diagnostic Evaluation J. Timothy Greenamyre, MD, PhD Involuntary tremulous motion, with lessened muscular power, in parts not in action and even when supported
Parkinson s Disease Initial Clinical and Diagnostic Evaluation J. Timothy Greenamyre, MD, PhD Involuntary tremulous motion, with lessened muscular power, in parts not in action and even when supported
Functional Distinctions
 Functional Distinctions FUNCTION COMPONENT DEFICITS Start Basal Ganglia Spontaneous Movements Move UMN/LMN Cerebral Cortex Brainstem, Spinal cord Roots/peripheral nerves Plan Cerebellum Ataxia Adjust Cerebellum
Functional Distinctions FUNCTION COMPONENT DEFICITS Start Basal Ganglia Spontaneous Movements Move UMN/LMN Cerebral Cortex Brainstem, Spinal cord Roots/peripheral nerves Plan Cerebellum Ataxia Adjust Cerebellum
Dementia. Assessing Brain Damage. Mental Status Examination
 Dementia Assessing Brain Damage Mental status examination Information about current behavior and thought including orientation to reality, memory, and ability to follow instructions Neuropsychological
Dementia Assessing Brain Damage Mental status examination Information about current behavior and thought including orientation to reality, memory, and ability to follow instructions Neuropsychological
ORIGINAL CONTRIBUTION. The Natural History of the Syndrome of Primary Progressive Freezing Gait
 ORIGINAL CONTRIBUTION The Natural History of the Syndrome of Primary Progressive Freezing Gait Stewart A. Factor, DO; Danna L. Jennings, MD; Eric S. Molho, MD; Kenneth L. Marek, MD Background: Primary
ORIGINAL CONTRIBUTION The Natural History of the Syndrome of Primary Progressive Freezing Gait Stewart A. Factor, DO; Danna L. Jennings, MD; Eric S. Molho, MD; Kenneth L. Marek, MD Background: Primary
Basal ganglia motor circuit
 Parkinson s Disease Basal ganglia motor circuit 1 Direct pathway (gas pedal) 2 Indirect pathway (brake) To release or augment the tonic inhibition of GPi on thalamus Direct pathway There is a tonic inhibition
Parkinson s Disease Basal ganglia motor circuit 1 Direct pathway (gas pedal) 2 Indirect pathway (brake) To release or augment the tonic inhibition of GPi on thalamus Direct pathway There is a tonic inhibition
MRI of Pathological Aging Brain
 MRI of Pathological Aging Brain Yukio Miki Department of Radiology, Osaka City University A variety of pathological changes occur in the brain with aging, and many of these changes can be identified by
MRI of Pathological Aging Brain Yukio Miki Department of Radiology, Osaka City University A variety of pathological changes occur in the brain with aging, and many of these changes can be identified by
Visualization and simulated animations of pathology and symptoms of Parkinson s disease
 Visualization and simulated animations of pathology and symptoms of Parkinson s disease Prof. Yifan HAN Email: bctycan@ust.hk 1. Introduction 2. Biochemistry of Parkinson s disease 3. Course Design 4.
Visualization and simulated animations of pathology and symptoms of Parkinson s disease Prof. Yifan HAN Email: bctycan@ust.hk 1. Introduction 2. Biochemistry of Parkinson s disease 3. Course Design 4.
Common Forms of Dementia Handout Package
 Common Forms of Dementia Handout Package Common Forms of Dementia 1 Learning Objectives As a result of working through this module, you should be better able to: 1. Describe clinical features of 4 major
Common Forms of Dementia Handout Package Common Forms of Dementia 1 Learning Objectives As a result of working through this module, you should be better able to: 1. Describe clinical features of 4 major
HYPERTENSIVE ENCEPHALOPATHY
 HYPERTENSIVE ENCEPHALOPATHY Reversible posterior leukoencephalopathy syndrome Cause Renal disease Pheochromocytoma Disseminated vasculitis Eclampsia Acute toxemia Medications & illicit drugs (cocaine)
HYPERTENSIVE ENCEPHALOPATHY Reversible posterior leukoencephalopathy syndrome Cause Renal disease Pheochromocytoma Disseminated vasculitis Eclampsia Acute toxemia Medications & illicit drugs (cocaine)
Brain tissue and white matter lesion volume analysis in diabetes mellitus type 2
 Brain tissue and white matter lesion volume analysis in diabetes mellitus type 2 C. Jongen J. van der Grond L.J. Kappelle G.J. Biessels M.A. Viergever J.P.W. Pluim On behalf of the Utrecht Diabetic Encephalopathy
Brain tissue and white matter lesion volume analysis in diabetes mellitus type 2 C. Jongen J. van der Grond L.J. Kappelle G.J. Biessels M.A. Viergever J.P.W. Pluim On behalf of the Utrecht Diabetic Encephalopathy
Central nervous system
 Central nervous system By Dr. Mohsen Dashti Clinical Medicine & Pathology 316 7 th Lecture Lecture outline Review of structure & function. Symptoms, signs & tests. Specific diseases. Review of structure
Central nervous system By Dr. Mohsen Dashti Clinical Medicine & Pathology 316 7 th Lecture Lecture outline Review of structure & function. Symptoms, signs & tests. Specific diseases. Review of structure
Movement Disorders: A Brief Overview
 Movement Disorders: A Brief Overview Albert Hung, MD, PhD Massachusetts General Hospital Harvard Medical School August 17, 2006 Cardinal Features of Parkinsonism Tremor Rigidity Bradykinesia Postural imbalance
Movement Disorders: A Brief Overview Albert Hung, MD, PhD Massachusetts General Hospital Harvard Medical School August 17, 2006 Cardinal Features of Parkinsonism Tremor Rigidity Bradykinesia Postural imbalance
1 MS Lesions in T2-Weighted Images
 1 MS Lesions in T2-Weighted Images M.A. Sahraian, E.-W. Radue 1.1 Introduction Multiple hyperintense lesions on T2- and PDweighted sequences are the characteristic magnetic resonance imaging (MRI) appearance
1 MS Lesions in T2-Weighted Images M.A. Sahraian, E.-W. Radue 1.1 Introduction Multiple hyperintense lesions on T2- and PDweighted sequences are the characteristic magnetic resonance imaging (MRI) appearance
Stroke Parkinsonism and Parkinson s Disease AAIM
 NEUROLOGICAL PROBLEMS IN THE ELDERLY, Stroke Parkinsonism and Parkinson s Disease AAIM October 17, 2012 Robert Lund, MD STROKE Elderly Stroke Etiologies 05/10/2012 3 Highest Incidence for Stroke in Ages
NEUROLOGICAL PROBLEMS IN THE ELDERLY, Stroke Parkinsonism and Parkinson s Disease AAIM October 17, 2012 Robert Lund, MD STROKE Elderly Stroke Etiologies 05/10/2012 3 Highest Incidence for Stroke in Ages
ORIGINAL CONTRIBUTION. Transcranial Brain Sonography Findings in Discriminating Between Parkinsonism and Idiopathic Parkinson Disease
 ORIGINAL CONTRIBUTION Transcranial Brain Sonography Findings in Discriminating Between Parkinsonism and Idiopathic Parkinson Disease Uwe Walter, MD; Dirk Dressler, MD; omas Probst, MD; Alexander Wolters,
ORIGINAL CONTRIBUTION Transcranial Brain Sonography Findings in Discriminating Between Parkinsonism and Idiopathic Parkinson Disease Uwe Walter, MD; Dirk Dressler, MD; omas Probst, MD; Alexander Wolters,
Dopamine Transporter Imaging With Single-Photon Emission Computed. Tomography
 Dopamine Transporter Imaging With Single-Photon Emission Computed Tomography Policy Number: 6.01.54 Last Review: 9/2018 Origination: 9/2015 Next Review: 9/2019 Policy Blue Cross and Blue Shield of Kansas
Dopamine Transporter Imaging With Single-Photon Emission Computed Tomography Policy Number: 6.01.54 Last Review: 9/2018 Origination: 9/2015 Next Review: 9/2019 Policy Blue Cross and Blue Shield of Kansas
Views and Reviews. [ 123 I]FP-CIT (DaTscan) SPECT Brain Imaging in Patients with Suspected Parkinsonian Syndromes ABSTRACT
![Views and Reviews. [ 123 I]FP-CIT (DaTscan) SPECT Brain Imaging in Patients with Suspected Parkinsonian Syndromes ABSTRACT Views and Reviews. [ 123 I]FP-CIT (DaTscan) SPECT Brain Imaging in Patients with Suspected Parkinsonian Syndromes ABSTRACT](/thumbs/76/73233066.jpg) Views and Reviews [ 123 I]FP-CIT (DaTscan) SPECT Brain Imaging in Patients with Suspected Parkinsonian Syndromes Robert A. Hauser, MD, Donald G. Grosset, MD From the Departments of Neurology, Molecular
Views and Reviews [ 123 I]FP-CIT (DaTscan) SPECT Brain Imaging in Patients with Suspected Parkinsonian Syndromes Robert A. Hauser, MD, Donald G. Grosset, MD From the Departments of Neurology, Molecular
Downloaded from Binswanger s Disease. Binswanger s Disease
 Medrech ISSN No. 2394-3971 Review Article Binswanger s Disease Ramoju Kishore Kumar 1*, N. Sri Ram 2, I. Sree Harsha 3. 1,3 Pharm. D 3 rd Year, Holy Mary College of Pharmacy, 2 Associate professor, Holy
Medrech ISSN No. 2394-3971 Review Article Binswanger s Disease Ramoju Kishore Kumar 1*, N. Sri Ram 2, I. Sree Harsha 3. 1,3 Pharm. D 3 rd Year, Holy Mary College of Pharmacy, 2 Associate professor, Holy
Early Clinical Features of Parkinson s Disease and Related Disorders. Dr. Alastair Noyce
 1 Specialist Registrar in Neurology, London Deanery Parkinson s UK Doctoral Research Fellow Project lead for PREDICT-PD Declarations Salary: Parkinson's UK, Barts and the London NHS Trust Grants: Parkinson's
1 Specialist Registrar in Neurology, London Deanery Parkinson s UK Doctoral Research Fellow Project lead for PREDICT-PD Declarations Salary: Parkinson's UK, Barts and the London NHS Trust Grants: Parkinson's
Lecture XIII. Brain Diseases I - Parkinsonism! Brain Diseases I!
 Lecture XIII. Brain Diseases I - Parkinsonism! Bio 3411! Wednesday!! Lecture XIII. Brain Diseases - I.! 1! Brain Diseases I! NEUROSCIENCE 5 th ed! Page!!Figure!!Feature! 408 18.9 A!!Substantia Nigra in
Lecture XIII. Brain Diseases I - Parkinsonism! Bio 3411! Wednesday!! Lecture XIII. Brain Diseases - I.! 1! Brain Diseases I! NEUROSCIENCE 5 th ed! Page!!Figure!!Feature! 408 18.9 A!!Substantia Nigra in
CN V! touch! pain! Touch! P/T!
 CN V! touch! pain! Touch! P/T! Visual Pathways! L! R! B! A! C! D! LT! E! F! RT! G! hypothalamospinal! and! ALS! Vestibular Pathways! 1. Posture/Balance!!falling! 2. Head Position! 3. Eye-Head Movements
CN V! touch! pain! Touch! P/T! Visual Pathways! L! R! B! A! C! D! LT! E! F! RT! G! hypothalamospinal! and! ALS! Vestibular Pathways! 1. Posture/Balance!!falling! 2. Head Position! 3. Eye-Head Movements
Parkinsonism or Parkinson s Disease I. Symptoms: Main disorder of movement. Named after, an English physician who described the then known, in 1817.
 Parkinsonism or Parkinson s Disease I. Symptoms: Main disorder of movement. Named after, an English physician who described the then known, in 1817. Four (4) hallmark clinical signs: 1) Tremor: (Note -
Parkinsonism or Parkinson s Disease I. Symptoms: Main disorder of movement. Named after, an English physician who described the then known, in 1817. Four (4) hallmark clinical signs: 1) Tremor: (Note -
NIH Public Access Author Manuscript Mov Disord. Author manuscript; available in PMC 2009 May 18.
 NIH Public Access Author Manuscript Published in final edited form as: Mov Disord. 2008 August 15; 23(11): 1602 1605. doi:10.1002/mds.22161. Emergence of Parkinsons Disease in Essential Tremor: A Study
NIH Public Access Author Manuscript Published in final edited form as: Mov Disord. 2008 August 15; 23(11): 1602 1605. doi:10.1002/mds.22161. Emergence of Parkinsons Disease in Essential Tremor: A Study
CT and MR Imaging in Young Stroke Patients
 CT and MR Imaging in Young Stroke Patients Ashfaq A. Razzaq,Behram A. Khan,Shahid Baig ( Department of Neurology, Aga Khan University Hospital, Karachi. ) Abstract Pages with reference to book, From 66
CT and MR Imaging in Young Stroke Patients Ashfaq A. Razzaq,Behram A. Khan,Shahid Baig ( Department of Neurology, Aga Khan University Hospital, Karachi. ) Abstract Pages with reference to book, From 66
University of Bristol - Explore Bristol Research
 Skrobot, O. A., O'Brien, J., Black, S., Chen, C., DeCarli, C., Erkinjuntti, T.,... Kehoe, P. G. (2016). The vascular impairment of cognition classification consensus study. Alzheimer's and Dementia. DOI:
Skrobot, O. A., O'Brien, J., Black, S., Chen, C., DeCarli, C., Erkinjuntti, T.,... Kehoe, P. G. (2016). The vascular impairment of cognition classification consensus study. Alzheimer's and Dementia. DOI:
Parkinson s Disease Update
 Parkinson s Disease Update Elise Anderson MD Providence Center for Parkinson s Disease October 26, 2017 11/6/2017 1 Disclosures GE Speaker, DaTSCAN 11/6/2017 2 Outline PD diagnosis Motor and nonmotor symptoms
Parkinson s Disease Update Elise Anderson MD Providence Center for Parkinson s Disease October 26, 2017 11/6/2017 1 Disclosures GE Speaker, DaTSCAN 11/6/2017 2 Outline PD diagnosis Motor and nonmotor symptoms
COGNITIVE SCIENCE 107A. Motor Systems: Basal Ganglia. Jaime A. Pineda, Ph.D.
 COGNITIVE SCIENCE 107A Motor Systems: Basal Ganglia Jaime A. Pineda, Ph.D. Two major descending s Pyramidal vs. extrapyramidal Motor cortex Pyramidal system Pathway for voluntary movement Most fibers originate
COGNITIVE SCIENCE 107A Motor Systems: Basal Ganglia Jaime A. Pineda, Ph.D. Two major descending s Pyramidal vs. extrapyramidal Motor cortex Pyramidal system Pathway for voluntary movement Most fibers originate
Final Report. Title of Project: Quantifying and measuring cortical reorganisation and excitability with post-stroke Wii-based Movement Therapy
 Final Report Author: Dr Penelope McNulty Qualification: PhD Institution: Neuroscience Research Australia Date: 26 th August, 2015 Title of Project: Quantifying and measuring cortical reorganisation and
Final Report Author: Dr Penelope McNulty Qualification: PhD Institution: Neuroscience Research Australia Date: 26 th August, 2015 Title of Project: Quantifying and measuring cortical reorganisation and
Multiple choice questions: ANSWERS
 Multiple choice questions: ANSWERS Chapter 1. Redefining Parkinson s disease 1. Common non-motor features that precede the motor findings in Parkinson s disease (PD) include all of the following except?
Multiple choice questions: ANSWERS Chapter 1. Redefining Parkinson s disease 1. Common non-motor features that precede the motor findings in Parkinson s disease (PD) include all of the following except?
Središnja medicinska knjižnica
 Središnja medicinska knjižnica Adamec I., Klepac N., Milivojević I., Radić B., Habek M. (2012) Sick sinus syndrome and orthostatic hypotension in Parkinson's disease. Acta Neurologica Belgica, 112 (3).
Središnja medicinska knjižnica Adamec I., Klepac N., Milivojević I., Radić B., Habek M. (2012) Sick sinus syndrome and orthostatic hypotension in Parkinson's disease. Acta Neurologica Belgica, 112 (3).
First described by James Parkinson in his classic 1817 monograph, "An Essay on the Shaking Palsy"
 Parkinson's Disease First described by James Parkinson in his classic 1817 monograph, "An Essay on the Shaking Palsy" Parkinson s disease (PD) is a neurological disorder characterized by a progressive
Parkinson's Disease First described by James Parkinson in his classic 1817 monograph, "An Essay on the Shaking Palsy" Parkinson s disease (PD) is a neurological disorder characterized by a progressive
DISORDERS OF THE NERVOUS SYSTEM
 DISORDERS OF THE NERVOUS SYSTEM Bell Work What s your reaction time? Go to this website and check it out: https://www.justpark.com/creative/reaction-timetest/ Read the following brief article and summarize
DISORDERS OF THE NERVOUS SYSTEM Bell Work What s your reaction time? Go to this website and check it out: https://www.justpark.com/creative/reaction-timetest/ Read the following brief article and summarize
DRUG TREATMENT OF PARKINSON S DISEASE. Mr. D.Raju, M.pharm, Lecturer
 DRUG TREATMENT OF PARKINSON S DISEASE Mr. D.Raju, M.pharm, Lecturer PARKINSON S DISEASE (parkinsonism) is a neurodegenerative disorder which affects t h e b a s a l g a n g l i a - and is associated with
DRUG TREATMENT OF PARKINSON S DISEASE Mr. D.Raju, M.pharm, Lecturer PARKINSON S DISEASE (parkinsonism) is a neurodegenerative disorder which affects t h e b a s a l g a n g l i a - and is associated with
Introduction to Dementia: Diagnosis & Evaluation. Created in March 2005 Duration: about 15 minutes
 Introduction to Dementia: Diagnosis & Evaluation Created in March 2005 Duration: about 15 minutes Axel Juan, MD The Geriatrics Institute axel.juan@med.va.gov 305-575-3388 Credits Principal medical contributor:
Introduction to Dementia: Diagnosis & Evaluation Created in March 2005 Duration: about 15 minutes Axel Juan, MD The Geriatrics Institute axel.juan@med.va.gov 305-575-3388 Credits Principal medical contributor:
Amyotrophic lateral sclerosis (ALS) is a progressive neurodegenerative
 ORIGINAL RESEARCH E. Matsusue S. Sugihara S. Fujii T. Kinoshita T. Nakano E. Ohama T. Ogawa Cerebral Cortical and White Matter Lesions in Amyotrophic Lateral Sclerosis with Dementia: Correlation with MR
ORIGINAL RESEARCH E. Matsusue S. Sugihara S. Fujii T. Kinoshita T. Nakano E. Ohama T. Ogawa Cerebral Cortical and White Matter Lesions in Amyotrophic Lateral Sclerosis with Dementia: Correlation with MR
