INSTRUCTIONS FOR USE FOR: English
|
|
|
- Tiffany Ferguson
- 5 years ago
- Views:
Transcription
1 INSTRUCTIONS FOR USE FOR: English
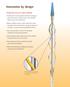
2 INSTRUCTIONS FOR USE FOR: Carefully read all instructions prior to use. Observe all warnings and precautions noted throughout these instructions. Failure to do so may result in complications. DEVICE DESCRIPTION The system consists of an embolic filter device, a delivery catheter, and a retrieval catheter, and is compatible with guide catheters and sheaths having a minimum inner diameter of 0.066". A wire torquing device and peel-away introducer sheath are packaged with the system. The embolic filter device is a nitinol frame with an attached expanded polytetrafluoroethylene (eptfe) filter bag (Figure 1). The filter bag is perforated with pores (nominal pore size of 100 µm). The CBAS Heparin Surface on the GORE Embolic Filter bag consists of stable, covalent, end-point attached heparin of porcine origin. The proximal end of the frame is attached to a PTFE coated guidewire. The distal guidewire has a shapeable, radiopaque floppy tip. The guidewire passes through the distal end of the filter bag. There is rotational freedom between the guidewire and filter components so that when the device is loaded and deployed, the guidewire is free to rotate independent of the filter. There are four radiopaque markers on the filter: three markers on the frame and the radiopaque collar, joining the guidewire to the filter. The filter comes in two sizes to accommodate vessel diameters from 2.5 to 5.5 mm. The frame length and total frame and bag length are shown in Figure 2. The guidewire comes in 185 cm and 300 cm lengths. The delivery catheter and the retrieval catheter each have a working length of 150 cm and a guidewire exit port approximately 30 cm from the distal tip to facilitate single-operator use. The light blue tip of the delivery catheter has an outer diameter of less than or equal to 3.2 Fr (1.1 mm). The proximal end of the delivery catheter is colored blue. The gray tip of the retrieval catheter has an outer diameter of less than or equal to 4.8 Fr (1.6 mm) and contains a radiopaque marker. The proximal end of the retrieval catheter is colored black. FIGURE 1: GORE EMBOLIC FILTER Delivery Catheter Proximal End (blue) Guidewire Guidewire Distal Tip of Delivery Catheter (light blue) Nitinol Filter Bag Olive Floppy Tip (radiopaque) Wire Exit Port Nitinol Markers (3 Places) Filter Bag Collar When deployed from the delivery catheter, the heparin-coated filter bag with perforated holes enables the capture of embolic debris that may be produced during the procedure while maintaining blood perfusion. The frame provides apposition to the vessel wall to enhance debris collection. At the completion of the procedure, the filter is recaptured and removed from the patient using the retrieval catheter. The is supplied sterile and should not be resterilized. FIGURE 2: GORE EMBOLIC FILTER FRAME LENGTH / FRAME AND BAG LENGTH Length and Bag Length Filter Diameter Unconstrained Length * and Bag Length * 5 mm 7 mm 8 mm 9 mm 18 mm 20 mm * When measured in the smallest indicated vessel diameter. CONTENTS One each:, delivery catheter, retrieval catheter, wire torquing device, peel-away introducer. INDICATIONS for use The is indicated for use as a guidewire and embolic protection system to contain and remove embolic material during angioplasty and stenting procedures in carotid arteries with diameters between 2.5 and 5.5 mm. CONTRAINDICATIONS DO NOT USE IN: Patients with known hypersensitivity to heparin, including those patients who have had a previous incident of Heparin-Induced Thrombocytopenia (HIT) type II Patients in whom anti-coagulant or anti-platelet therapy is contraindicated. Patients with tortuous anatomy or disease morphology which would prohibit the safe placement of guide catheters, sheaths, embolic protection systems, or stent systems within the access or target vessels. Patients with known hypersensitivity to nitinol. Patients with unresolved bleeding disorders. WARNINGS Only physicians who have received appropriate training and are familiar with the principles, clinical applications, complications, side effects and hazards commonly associated with carotid artery interventional procedures should use this device. The safety and efficacy of the has not been demonstrated with carotid stent systems other than ACCULINK Carotid Stent, XACT Carotid Stent, PRECISE Carotid Stent, PROTEGE Carotid Stent, and WALLSTENT Carotid Stent as listed in Table 5. 1
3 With any vascular procedure, the possibility of HIT may exist. The incidence of HIT type II is extremely low in vascular patients receiving heparin over a period of several days. If HIT type II is diagnosed, established procedures for the treatment of this condition, including immediate cessation of systemic heparin administration, should be followed. 1,2 If symptoms persist, or the health of the patient appears compromised, alternative pharmaceutical or surgical procedures, including ligation or removal of the device, may be considered at the discretion of the attending physician. The appropriate antiplatelet and anticoagulation therapy should be administered pre- and post-procedure at a dosage deemed appropriate by the physician. Ensure filter is sized properly for target vessel. Severe oversizing of the filter frame can lead to vessel spasm, vessel trauma or frame fracture. Severe undersizing can lead to inadequate vessel apposition and compromised filter efficiency. When introducing the delivery system, ensure that the floppy tip is free within the vessel lumen and is not directed against the vessel wall or advancing between the stent and vessel wall. Failure to do so may result in vessel trauma or the filter becoming trapped between the stent and vessel wall. Provide adequate distance between the radiopaque collar on the filter and the interventional device to avoid the potential for entanglement. Ensure the deployed filter is kept distal to the treatment site and the deployed stent. Do not attempt to pull a deployed filter through the stent or retrieve the filter within the stent. Pulling the filter into the stent area may lead to stent-filter entanglement and / or filter detachment. Avoid excessive movement of the during catheter exchanges especially in a vessel with spasm as this may cause significant vessel damage. If blood flow is significantly reduced, aspirate blood proximal to filter prior to retrieving device. Complete withdrawal of the filter into the retrieval catheter may lead to filter bag tearing or rupture and release of embolic debris. During retrieval, withdraw the retrieval catheter and guidewire together. Pulling on only the retrieval catheter can lead to the filter redeploying from the retrieval catheter. The safety and effectiveness of this device as an embolic filter have not been established in vasculatures outside of the carotid arteries (coronary, cerebral, and peripheral). PRECAUTIONS Confirm the compatibility of the with the intended interventional devices prior to use. Do not use the if the sterile package is compromised or the is damaged. Follow the Directions for Use supplied with all accessories used in conjunction with the. Exercise care in handling the before and during the procedure to reduce the possibility of damaging the filter. The is designed for single use only. Do not resterilize or reuse the. Do not use the after the labeled use by (expiration) date. Do not deliver the with any interventional devices other than the GORE Delivery Catheter. To ensure proper positioning and removal of the filter, the vessel distal to the treatment site should have adequate length and not be excessively tortuous. Do not continue to load device if filter bag is inverted (i.e., folded back on itself) or wrinkled. Stop and pull gently on the distal end of the bag to straighten it prior to completing device loading. Loading an inverted filter bag can lead to excessive loading and deployment forces. Always advance or withdraw the slowly. Never push, withdraw or torque a wire that meets resistance. Resistance may be felt and / or observed under fluoroscopy by noting any buckling of the wire or tip. Determine the cause of the resistance under fluoroscopy and take any necessary remedial action. During delivery, advance the guidewire and the delivery catheter together. Advancing only the guidewire may result in premature filter deployment. Do not rotate the delivery or retrieval catheter excessively in either direction as this may cause the guidewire to wrap around the catheter. Maintain proper guide catheter / sheath support in the common carotid artery throughout the procedure. If guide catheter / sheath access cannot be maintained, the case should be discontinued. Failure to maintain proper support may lead to prolapse of the guide catheter / sheath in the aortic arch, resulting in any of the following: Deployed filter pulled through an undiliated lesion; or Filter-stent entanglement; or Filter fracture and / or detachment; or Stent dislodgement Keep the wire position stable while the filter is deployed. Monitor the target vessel for signs of vasospasm. If evident, treat appropriately. Ensure there is adequate distance between the radiopaque collar on the filter and the distal end of the stent delivery system to achieve the desired stent position. Perform all catheter exchanges cautiously to prevent air embolism or damage to the artery. If excessive debris is collected in the filter and blood flow is obstructed, the filter may be removed and replaced or the procedure may be completed prior to device removal at the physician's discretion. Carefully retrieve the filter through newly deployed stents to ensure the stents are not disrupted. Do not attempt to retrieve filter with the delivery catheter. The presence of heparin on the is not intended to serve as an alternative to the surgeon s chosen intraoperative or postoperative anticoagulation regimens. ADVERSE EVENTS Summary of Clinical Results (G080059) The objective of the Gore EMBOLDEN Clinical Study was to assess the safety and effectiveness of the for embolic protection during carotid artery angioplasty and stenting (CAS) procedures in high surgical risk patients. A comprehensive list of the major adverse events (MAEs) is provided in Table 1. In summary, outcomes in subcategories of the major adverse event end point included death (0.8%), death/major stroke (1.2%), death/any stroke (3.6%), myocardial infarction (MI) (0.4%), and death/stroke/mi (4.0%). The device used in the EMBOLDEN study was improved upon based on feedback received as part of study use. Specifically, the delivery catheter was modified to improve filter deployment characteristics. This change was evaluated in bench testing and this data supported clearance of the improved device. 2
4 TABLE 1: MAJOR ADVERSE EVENTS Number of Subjects 250 Subjects With One or More Major Adverse Events 10 (4.0%) Death 2 (0.8%) Myocardial Infarction 1 (0.4%) Q-Wave MI 0 (0.0%) Non Q-Wave MI 1 (0.4%) Stroke 7 (2.8%) Major Stroke 1 (0.4%) Ischemic Stroke 1 (0.4%) Ipsilateral 1 (0.4%) Non-Ipsilateral 0 (0.0%) Hemorrhagic Stroke 0 (0.0%) Ipsilateral 0 (0.0%) Non-Ipsilateral 0 (0.0%) Minor Stroke 6 (2.4%) Ischemic Stroke 6 (2.4%) Ipsilateral 5 (2.0%) Non-Ipsilateral 1 (0.4%) Hemorrhagic Stroke 0 (0.0%) Ipsilateral 0 (0.0%) Non-Ipsilateral 0 (0.0%) The adverse events (serious and non-serious) that were reported during the Gore EMBOLDEN Clinical Study are summarized in Table 2. Adverse events (AEs) were defined as any unfavorable and unintended sign, symptom, or disease temporally associated with the use of the, whether or not considered related to the device. TABLE 2: SUMMARY OF ADVERSE EVENTS Severity of Adverse Event Non-Serious Serious Overall Subjects with One or More Adverse Events 136 (54.4%) 56 (22.4%) 147 (58.8%) Hypotension 38 (15.2%) 14 (5.6%) 51 (20.4%) Anemia 14 (5.6%) 6 (2.4%) 19 (7.6%) Hypertension 16 (6.4%) 2 (0.8%) 18 (7.2%) Bradycardia 10 (4.0%) 5 (2.0%) 15 (6.0%) Bleeding 11 (4.4%) 1 (0.4%) 12 (4.8%) Headache 11 (4.4%) 2 (0.8%) 12 (4.8%) Nausea 12 (4.8%) - 12 (4.8%) Access site pain 8 (3.2%) - 8 (3.2%) Angina/coronary ischemia 5 (2.0%) 3 (1.2%) 8 (3.2%) Back pain 8 (3.2%) - 8 (3.2%) Cerebral ischemia/transient ischemic attack (TIA) 6 (2.4%) 2 (0.8%) 8 (3.2%) Ecchymosis 8 (3.2%) - 8 (3.2%) Groin hematoma 6 (2.4%) 2 (0.8%) 8 (3.2%) Hematoma 8 (3.2%) - 8 (3.2%) Vessel spasm 7 (2.8%) 1 (0.4%) 8 (3.2%) Numbness 7 (2.8%) - 7 (2.8%) Renal failure/insufficiency 6 (2.4%) 1 (0.4%) 7 (2.8%) Vomiting 7 (2.8%) - 7 (2.8%) Dyspnea 4 (1.6%) 2 (0.8%) 6 (2.4%) 3
5 Severity of Adverse Event Non-Serious Serious Overall Vascular access complication 3 (1.2%) 3 (1.2%) 6 (2.4%) Abdominal pain 5 (2.0%) - 5 (2.0%) Asystole 3 (1.2%) 2 (0.8%) 5 (2.0%) Dizziness 5 (2.0%) - 5 (2.0%) Urinary retention 5 (2.0%) - 5 (2.0%) Visual disturbance 4 (1.6%) 1 (0.4%) 5 (2.0%) Fever 3 (1.2%) 1 (0.4%) 4 (1.6%) Rash 3 (1.2%) 1 (0.4%) 4 (1.6%) Stroke/cerebrovascular accident (CVA) 1 (0.4%) 3 (1.2%) 4 (1.6%) Confusion 3 (1.2%) - 3 (1.2%) Congestive Heart Failure (CHF) - onset or worsening of - 3 (1.2%) 3 (1.2%) Edema 3 (1.2%) - 3 (1.2%) Hypokalemia 3 (1.2%) - 3 (1.2%) Infection at access site 2 (0.8%) 1 (0.4%) 3 (1.2%) Itching 2 (0.8%) 1 (0.4%) 3 (1.2%) Leg pain 3 (1.2%) - 3 (1.2%) Neck pain 3 (1.2%) - 3 (1.2%) Bronchitis 1 (0.4%) 1 (0.4%) 2 (0.8%) COPD, worsening of 1 (0.4%) 1 (0.4%) 2 (0.8%) Elevated WBC 2 (0.8%) - 2 (0.8%) Infection 1 (0.4%) 1 (0.4%) 2 (0.8%) Ischemia/Infarction of tissue/organ - 2 (0.8%) 2 (0.8%) Pseudoaneurysm 1 (0.4%) 1 (0.4%) 2 (0.8%) Retroperitoneal bleed 1 (0.4%) 1 (0.4%) 2 (0.8%) Seizure - 2 (0.8%) 2 (0.8%) Slurred speech 2 (0.8%) - 2 (0.8%) Thrombocytopenia 2 (0.8%) - 2 (0.8%) Altered mental state 1 (0.4%) - 1 (0.4%) Atrial fibrillation - 1 (0.4%) 1 (0.4%) Bacteremia 1 (0.4%) - 1 (0.4%) Blurred vision 1 (0.4%) - 1 (0.4%) Head pain 1 (0.4%) - 1 (0.4%) Hematuria 1 (0.4%) - 1 (0.4%) Hyponatremia 1 (0.4%) - 1 (0.4%) Ischemic stroke - 1 (0.4%) 1 (0.4%) Myocardial infarction (MI) - 1 (0.4%) 1 (0.4%) Non-cardiac chest pain 1 (0.4%) - 1 (0.4%) Respiratory failure - 1 (0.4%) 1 (0.4%) Tachycardia 1 (0.4%) - 1 (0.4%) Thrombocytopenia, heparin-induced 1 (0.4%) - 1 (0.4%) Thrombosis/Occlusion - GEF - 1 (0.4%) 1 (0.4%) Thrombosis/Occlusion - stent 1 (0.4%) - 1 (0.4%) Vessel dissection, perforation or rupture - 1 (0.4%) 1 (0.4%) Nervous System - Other 14 (5.6%) 2 (0.8%) 15 (6.0%) Respiratory, Thoracic & Mediastinal - Other 6 (2.4%) 4 (1.6%) 10 (4.0%) Blood & Lymphatic System - Other 6 (2.4%) 1 (0.4%) 7 (2.8%) Musculoskeletal and Connective Tissue - Other 6 (2.4%) 1 (0.4%) 7 (2.8%) Renal & Urinary - Other 4 (1.6%) 2 (0.8%) 5 (2.0%) Cardiac - Other 1 (0.4%) 3 (1.2%) 4 (1.6%) Gastrointestinal - Other 3 (1.2%) 1 (0.4%) 4 (1.6%) Infection & Infestations - Other 4 (1.6%) - 4 (1.6%) Metabolism & Nutrition - Other 3 (1.2%) 1 (0.4%) 4 (1.6%) Vascular - Other 2 (0.8%) 2 (0.8%) 4 (1.6%) Injury & Procedural - Other 3 (1.2%) - 3 (1.2%) Not Elsewhere Classified - Other 1 (0.4%) 2 (0.8%) 3 (1.2%) Skin & Subcutaneous Tissue - Other 2 (0.8%) - 2 (0.8%) 4
6 POTENTIAL PROCEDURE AND/OR DEVICE RELATED ADVERSE EVENTS: Complications and adverse events can occur when using any embolic protection device in carotid artery stenting procedures. These complications include, but are not limited to: abrupt vessel closure allergic reactions aneurysm angina / coronary ischemia arteriovenous fistula bacteremia or septicemia balloon burst or rupture balloon-associated thrombosis bleeding from anticoagulant or Antiplatelet medications bradycardia / arrhythmia and other conduction disturbances cerebral edema cerebral hemorrhage component damage (e.g., kinked catheters) congestive heart failure death deployment and retrieval failure distal embolization drug reactions embolism (which includes thrombus, plaque, air, device and / or component) emergent / urgent endarterectomy fever fluid overload groin hematoma headache hemorrhage / hematoma hemorrhagic stroke hyperperfusion syndrome hypertension / hypotension infection / sepsis ischemia / infarction of tissue / organ ischemic stroke myocardial infarction pain and tenderness pseudoaneurysm reduced blood flow renal failure / insufficiency restenosis of the stented artery seizure stent deformation stroke or other neurological complications (e.g., paralysis, paraplegia or aphasia) surgery required due to device failure temporary or total occlusion of the artery thromboembolic episodes; thrombophlebitis transient ischemic attacks (TIAs) vascular access complications (e.g., bleeding, vessel damage, pseudoaneurysm and infection) ventricular fibrillation vessel spasm, dissection, rupture, or perforation vessel thrombosis (partial blockage) unstable angina pectoris There were 5 documented cases where, a retrieval catheter other than the one supplied from Gore had to be used because the GORE Retrieval Catheter could not cross the stent. The alternate retrieval catheters used were ANGIOGUARD Retrieval catheter, RX ACCUNET Recovery catheter, and 5Fr Multipurpose catheter. In one of these cases, a TIA was reported as the filter was retrieved. SUMMARY OF CLINICAL RESULTS: The results of this clinical study support the safety and efficacy of the. Objectives The objective of the study was to assess the safety and efficacy of the when used to provide embolic protection during CAS procedures in conjunction with any FDA-approved carotid stent. This study compared the results of CAS with embolic protection using the in high risk surgical subjects to a performance goal derived from carotid stent studies utilizing distal embolic protection. Study Design Prospective, multicenter, non-randomized, single-arm study designed to compare 30-day safety and efficacy of used with FDA-approved carotid stents to an objective performance criteria (OPC) determined from the major adverse event (MAE) rates from prior CAS studies where distal embolic protection devices were used (less than a 12.0% OPC). The primary endpoint for the Gore EMBOLDEN Clinical Study, major adverse events, was a composite of death, stroke, or myocardial infarction (MI) through the 30-day post procedure follow-up. The secondary endpoints for the study were system (device) success, clinical success, access site complications, and neurological events. Study Enrollment Thirty-seven (37) US sites participated in the study and enrolled 250 pivotal subjects between January 2009 and July The data on these pivotal subjects is presented within this document. Patients Studied Eligible patients were diagnosed with carotid stenosis requiring revascularization with anatomic or co-morbid conditions placing them at high-risk for adverse events from CEA. Per NASCET criteria: Symptomatic subjects: >50% stenosis Asymptomatic subjects: >80% stenosis All subjects must have at least one anatomic or co-morbid risk factor placing them at high-risk for adverse events from CEA. The co-morbid and anatomic inclusion criteria for the study are summarized in Table 3. 5
7 TABLE 3: CO-MORBID AND ANATOMIC STUDY INCLUSION CRITERIA Number of Subjects 250 Anatomic Risk 47.2% (118/250) Surgically inaccessible lesions 6.0% (15/250) Post radical head/neck surgery or RT 10.0% (25/250) Spinal immobility of the neck 5.2% (13/250) Presence of tracheostomy stoma 2.4% (6/250) Laryngeal palsy or laryngectomy 2.0% (5/250) Contralateral laryngeal nerve damage 0.4% (1/250) Restenosis after prior CEA 26.8% (67/250) Co-Morbid Risk 64.8% (162/250) Age 80 years 36.4% (91/250) NYHA Class III or IV 3.6% (9/250) COPD with FEV1 < 30% 3.2% (8/250) LVEF < 30% 4.0% (10/250) Uncontrolled diabetes 3.2% (8/250) Unstable angina with EKG changes 0.8% (2/250) MI within 30 days of procedure 0.8% (2/250) Two or more diseased arteries 8.4% (21/250) Planned CABG or valve replacement surgery 1.2% (3/250) Contralateral total occlusion of the ICA 10.4% (26/250) TABLE 4: STUDY DEMOGRAPHICS Number of Subjects 250 Subject Age Mean (Min, Max) 74.7 (51.3, 92.6) Gender Male 61.2% (153/250) Female 38.8% (97/250) Medical History Hypertension 94.4% (236/250) Hyperlipidemia 94.0% (235/250) Diabetes 34.8% (87/250) Respiratory 30.8% (77/250) Current Smoker 20.8% (52/250) History of Stroke, TIA, MI Prior Stroke 18.8% (47/250) Prior MI 25.6% (64/250) Prior TIA 21.6% (54/250) Previous Cardiovascular Interventions Previous CEA 31.6% (79/250) Previous CABG 26.8% (67/250) Previous PCI 31.6% (79/250) Non-Carotid Vascular Disease Coronary 57.2% (143/250) Peripheral 38.8% (97/250) Cerebral 38.8% (97/250) Symptom Status Symptomatic 14.8% (37/250) Asymptomatic 85.2% (213/250) Lesion and Vessel Characteristics Mean Lesion Length (min,max) 23.8 (6.0, 48.5) Mean Minimum Lumen Diameter (MLD) (min, max) 1.6 (0.3, 3.2) Mean % Diameter Stenosis (min, max) 72.6 (44.7, 93.2) 6
8 STUDY RESULTS A summary of the distribution of the stents used in the study is provided in Table 5. Procedural outcomes for the study are summarized in Table 6. Serious adverse events, included hypotension in 5.6% of subjects, anemia in 2.4% of subjects, and bradycardia in 2.0% of subjects. A comprehensive summary of all adverse events is provided in Table 2. The was successfully deployed in 96.4% of subjects and carotid stent deployment was successful in 98.0% of subjects. There were nine (9) technical failures of the. In 6 of the cases an alternate distal filter was used. In the remaining 3 cases the procedure was terminated. The mean filter deployment time was 14 minutes in subjects with successful device deployment. A comprehensive list of the MAEs is provided in Table 1. In summary, outcomes in subcategories of the major adverse event endpoint included death (0.8%), death / major stroke (1.2%), death / any stroke (3.6%), MI (0.4%), and death / stroke / MI (4.0%). An overview of the primary and major adverse event endpoints for the study are summarized in Table 7 and 8. Table 9 summarizes the secondary endpoints for the study. The primary endpoint for the Gore EMBOLDEN Clinical Study met the OPC hypothesis defined for the study (lower than the 12.0% OPC). Subgroup analysis for octogenarians and symptomatic subjects reports MAE rates of 5.4% for both populations. TABLE 5: DISTRIBUTION OF STENTS IMPLANTED Stent Brand Acculink 27.3% (70/256) Precise 35.9% (92/256) Protege 6.6% (17/256) Xact 25.0% (64/256) WallStent 5.1% (13/256) TABLE 6: PROCEDURAL OUTCOMES Number of Subjects 250 Procedure Time (minutes) N = 250 Mean (Std Dev) 50.9 (25.7) Median 45.0 (Min, Max) (16, 153) Total Filter Time (minutes) N = 249 Mean (Std Dev) 13.8 (8.6) Median 11.0 (Min, Max) (0, 63) Total Fluoroscopy Time (minutes) N = 247 Mean (Std Dev) 17.8 (9.3) Median 16.1 (Min, Max) (5.9, 64.5) Hospital Stay (Days) N = 250 Mean (Std Dev) 1.5 (1.5) Median 1.0 (Min, Max) (1, 17) TABLE 7: PRIMARY AND MAJOR ADVERSE EVENT ENDPOINTS Primary Endpoint Intent-To-Treat Population 1 % (n/n) 95% C.I. Overall MAE 4.0% (10/250) (1.9%, 7.2%) Death 0.8% (2/250) (0.1%, 2.9%) Stroke 2.8% (7/250) (1.1%, 5.7%) Death/Any Stroke 3.6% (9/250) (1.7%, 6.7%) Myocardial Infarction (MI) 0.4% (1/250) (0.0%, 2.2%) 1 Intent-To-Treat Population defined as all enrolled subjects TABLE 8: PRIMARY AND MAJOR ADVERSE EVENT ENDPOINTS Per Protocol Population 1 % (n/n) 95% C.I. Primary Endpoint Overall MAE 4.3% (10/234) (2.1%, 7.7%) Death 0.9% (2/234) (0.1%, 3.1%) Stroke 3.0% (7/234) (1.2%, 6.1%) Death/Any Stroke 3.8% (9/234) (1.8%, 7.2%) Myocardial Infarction (MI) 0.4% (1/234) (0.0%, 2.4%) 1 Per Protocol Population defined as enrolled subjects with Device Success and 30-Day follow-up 7
9 TABLE 9: SECONDARY ENDPOINTS Secondary Endpoints % (n/n) 95% C.I. Device Success 96.4% (241/250) (93.3%, 98.3%) Clinical Success 93.2% (233/250) (89.3%, 96.0%) Access Site Complications 2.4% (6/250) (0.9%, 5.2%) Neurological Events 6.4% (16/250) (3.7%, 10.2%) Device Success defined as successful delivery, deployment and retrieval of the filter. Clinical Success defined as Device Success and the absence of death, emergency endarterectomy, repeat PTA / thrombolysis of target vessel and stroke/mi though hospital discharge. Access Site Complications defined as large hematoma (>5cm or requiring treatment), fistula or pseudoaneurysm formation, retroperitoneal bleeding or need for surgical repair. Neurological Events defined as stroke or TIA / cerebral ischemia through 30 day follow-up. HOW SUPPLIED The is supplied sterile in a single barrier protective sealed pouch. STORAGE AND HANDLING Handle the device with care, and avoid exposure to extreme temperatures and humidity. Store in a cool dry place. MATERIALS REQUIRED A guide catheter or sheath (0.066 minimum I.D.) is required for the An introducer sheath or appropriately sized guide catheter compatible with the vascular anatomy to accommodate the interventional device. Interventional device(s) Heparinized normal saline (sterile) Syringe (ex: 20 cc) DIRECTIONS FOR USE A. Preparation of Filter and Delivery Catheter 1. Prior to insertion of the, the physician should read the Directions for Use. 2. Measure the target vessel diameter where the filter will be deployed using angiography. Refer to Device Sizing table (Table 10) for the appropriate sized filter for the measured reference vessel diameter. TABLE 10: DEVICE SIZING Filter Diameter Unconstrained 5 mm 7 mm Reference Vessel Diameter Range 2.5 mm - 4 mm 4 mm mm Guide Catheter / Sheath min ID 0.066" min ID 0.066" 3. Prior to opening the sterile package, check that the diameter and guidewire length of the filter is correct. 4. Carefully inspect the packaging for damage to the sterile barrier. Do not use the after the use by (expiration) date or if the sterile package is compromised. Peel apart the sterile pouch and remove the components (Figure 3). FIGURE 3: GORE EMBOLIC FILTER AS PACKAGED A. Device / Delivery Catheter in Large Coil Filter Protective Tube Tube Clips Clear Clips B. Retrieval Catheter in Small Coil Black Clips Small Coil with Black Clips Containing the Retrieval Catheter Large Coil with Clear Clips Containing the and Delivery Catheter Rubber Guidewire Clip Proximal End of Retrieval Catheter Peel-away Introducer C. Wire Torquing Device and Peel-away Introducer in Accessory Pouch Wire Torquing Device 5. Using a syringe with heparinized saline, flush the large coil with clear clips containing the and delivery catheter (Figure 3A) to wet the components. 6. Remove the guidewire and delivery catheter from the rubber guidewire clip by rotating the clip 90 away from the guidewire and catheter. Simultaneously advance both until the distal end of the delivery catheter resides outside the coil. 7. Gently remove the protective tube from the tube clips and slide the tube off the filter. 8. Gently remove the filter and delivery catheter from the coil by grasping the distal end of the delivery catheter and withdrawing from the coil. 9. Inspect the system for damage. DO NOT use the if any components are damaged. 8
10 10. Submerge the filter and delivery catheter to wire exit port in heparinized saline. Load device by pulling wire near where it exits the delivery catheter while supporting the distal end of the delivery catheter. Gentle manipulation can be used to remove air bubbles. Pull wire until olive abuts tip of delivery catheter (Figure 4). Do not force the olive into the delivery catheter. If filter bag inverts (i.e., folds back on itself) during loading, stop and pull gently on the distal end of the bag to straighten it prior to completing device loading. 11. For additional flushing, advance device so there is a 1-2 mm gap between the olive and tip of the delivery catheter. Gently insert loaded device (floppy tip first up to the tip of the delivery catheter) into a 20 cc syringe filled with heparinized saline. Providing a seal between the tip of the delivery catheter and wire with fingers, flush 10 cc until fluid emerges from the wire exit port. Ensure floppy tip is protected by not flushing entire contents of syringe. FIGURE 4: LOADED FILTER IN DELIVERY CATHETER Delivery Catheter Proximal End (blue) Guidewire Guidewire Wire Exit Port Delivery Catheter (light blue) Olive Floppy Tip (radiopaque) Collar Markers B. Insertion and Placement 1. Use standard percutaneous techniques. Shape the tip of the guidewire, if desired. 2. Insert the and peel-away introducer (Figure 3C) assembly through a rotating hemostasis valve into an appropriately sized guide catheter or introducer sheath. Once the system, including the wire exit port, is inserted into the guide catheter, remove the peel-away introducer. 3. Advance the delivery catheter and guidewire together through the guide catheter / sheath past the lesion using fluoroscopic guidance. Pushing the delivery catheter and wire together rather than just the wire will prevent premature filter deployment, especially in tortuous anatomy. DO NOT pull on the wire while advancing the system as this may lodge the olive within the delivery catheter. Advance the system cautiously, especially if resistance is encountered. 4. To steer the system to the intended site, consider a two-handed approach, torquing the wire with one hand while advancing the delivery catheter with the other hand. The wire torquing device (Figure 3C) can be used to apply additional torque to the wire to improve steering. Appropriate position is verified when the radiopaque collar (proximal-most marker) of the frame is beyond the lesion (Figure 5). FIGURE 5: APPROPRIATE POSITION OF FILTER PRIOR TO DEPLOYMENT Vessel Lesion Delivery Catheter (light blue) Olive Floppy Tip (radiopaque) Collar Markers 5. Once in position, deploy the filter by maintaining the position of the wire and pulling back on the delivery catheter. 6. Device deployment is complete when the three radiopaque markers on the nitinol frame separate, indicating the frame is released from the delivery catheter and apposing the vessel wall (Figure 6). Note that depending on the fluoroscopy angle, overlap of radiopaque markers may occur. Therefore, it may appear that there are only two radiopaque markers. FIGURE 6: DEPLOYED FILTER Vessel Lesion Filter Bag Olive Floppy Tip (radiopaque) Collar Markers 7. Inject contrast medium to verify the device is in the proper position and flow is present. 8. Remove the delivery catheter and discard. C. During Procedure 1. Use the as a standard guidewire to track the interventional treatment device and other 0.014" compatible guidewire-based interventional devices to the target lesion site. DO NOT attempt to advance catheters and delivery systems beyond the radiopaque collar on the frame. If resistance is felt and the filter is observed moving distally, it is likely that the nonradiopaque tip of the interventional device is in contact with the collar. 2. Contrast media can be injected to visually assess the contents of the filter bag, and to verify that blood flow is not obstructed. 3. Keep the wire position stable while the filter is deployed. DO NOT move the deployed filter, especially in the presence of vasospasm. 4. If excessive debris is collected in the filter and blood flow is obstructed, the may be removed and replaced or the procedure may be completed prior to device removal at the physician's discretion. 5. Complete the procedure making sure the filter remains in the proper position distal to the lesion. D. Retrieval Note: If blood flow is significantly reduced, aspirate blood proximal to filter prior to retrieving filter. 1. Remove all interventional devices from the guide wire. 2. Using a syringe with heparinized saline, flush the small coil with black clips to wet the components. 3. Unclip proximal end of retrieval catheter from rubber clip and remove the retrieval catheter from its coil (black clips) (Figure 3B). 4. Inspect the retrieval catheter for damage. Do not use if any components are damaged. 5. Flush the distal end of the retrieval catheter with heparinized saline. Observe fluid exiting the wire exit port. 6. Route the proximal end of the guidewire through the distal end of the retrieval catheter and out the wire exit port. Handle the tip of the retrieval catheter with care to avoid kinking. Maintain the guidewire position and carefully advance the retrieval 9
11 catheter over the wire to the radiopaque collar of the filter. Slowly advance the retrieval catheter until the frame is collapsed and within the catheter. This is confirmed when the radiopaque frame markers come together to form a single marker and subsequently overlap with the radiopaque marker on the tip of the retrieval catheter (Figure 7). DO NOT use excessive force to obtain marker overlap. Note: If the retrieval catheter has difficulty passing through the stent, a variety of standard interventional techniques to aid in passage of the retrieval catheter may be used. Some options are: Rotate the patient's head to re-orient the anatomy. Change the position of the guiding catheter or sheath to better support the retrieval catheter and filter guidewire. If the stent struts are preventing advancement of the retrieval catheter, post-dilate the stent, or reposition the filter's guidewire. Insert an additional guidewire to straighten the stented area. Consider alternate retrieval catheter techniques. FIGURE 7: APPROPRIATE POSITION OF RETRIEVED DEVICE PRIOR TO REMOVING FROM PATIENT Vessel Pre-procedure Lesion Location Retrieval Catheter (gray) Filter Bag Olive Floppy Tip (radiopaque) Retrieval Catheter Collar Marker Overlapping with Markers 7. If resistance and / or entanglement is encountered with the portion of the filter bag protruding from the retrieval catheter, the filter may be retrieved further into the retrieval catheter at the discretion of the clinician. Additional imaging may be necessary to make the best clinical judgment. DO NOT use excessive force to retrieve the filter bag. Complete withdrawal of the filter into the retrieval catheter may lead to filter bag tearing or rupture and release of embolic debris. 8. Withdraw the entire system slowly through the guide catheter / sheath under fluoroscopic guidance. Withdrawing the retrieval catheter and guidewire together rather than just the retrieval catheter will prevent redeployment of the filter from the retrieval catheter. If there is any resistance when trying to withdraw the filter system into the guide catheter / sheath, pull back the guide catheter / sheath and the filter system together. If there is any resistance when trying to withdraw the filter system into the introducer sheath, withdraw the introducer sheath, guide catheter / sheath and filter system together. REFERENCES 1. Linkins LA, Dans AL, Moores LK, Bona R, Davidson BL, Schulman S, Crowther M; American College of Chest Physicians. Treatment and prevention of heparin-induced thrombocytopenia: antithrombotic therapy and prevention of thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest 2012;141(2)Supplement:e495S-e530S. 2. Warkentin TE. Heparin-coated intravascular devices and heparin-induced thrombocytopenia. In: Warkentin TE, Greinacher A, eds. Heparin-Induced Thrombocytopenia. 5th ed. New York, NY: Informa Healthcare USA; 2012;(20):
12 DEFINITIONS Authorised Representative in the European Community Batch Code Catalogue Number Caution CAUTION: USA Federal Law restricts the sale, distribution, or use of this device to, by, or on the order of a physician. Consult Instructions for Use Date of Manufacture 2 STERILIZE Do Not Resterilize Do Not Reuse Do Not Use if Package is Damaged Keep Dry Manufacturer Serial Number Use By Sterile Sterilized using Ethylene Oxide Store in a Cool Place Delivery Catheter Delivery Catheter Filter Diameter Unconstrained Guiding Catheter Length Reference Vessel Diameter Range Retrieval Catheter Retrieval Catheter 11
13 Manufacturer W.L. Gore & Asssociates, Inc North Fourth Street Flagstaff, Arizona UNITED STATES Order Information: Tel.: Tel.: Technical Information: Tel.: Tel.: For international contact and additional product information, visit GORE and designs are trademarks of W. L. Gore & Associates. CBAS is a trademark of Carmeda AB, a wholly owned subsidiary of W. L. Gore & Associates, Inc. ACCULINK and XACT are trademarks of Abbott Cardiovascular Systems, Inc. PRECISE is a trademark of Cordis Corporation. PROTEGE is a trademark of Covidien Ltd. WALLSTENT is a trademark of AMS Medivent S.A , , 2014, 2016 W. L. Gore & Associates, Inc. APRIL 2016 Printed on recyclable paper
DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service
 M AY. 6. 2011 10:37 A M F D A - C D R H - O D E - P M O N O. 4147 P. 1 DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration 10903 New Hampshire Avenue Document Control
M AY. 6. 2011 10:37 A M F D A - C D R H - O D E - P M O N O. 4147 P. 1 DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration 10903 New Hampshire Avenue Document Control
1 Description. 2 Indications. 3 Warnings ASPIRATION CATHETER
 Page 1 of 5 ASPIRATION CATHETER Carefully read all instructions prior to use, observe all warnings and precautions noted throughout these instructions. Failure to do so may result in complications. STERILE.
Page 1 of 5 ASPIRATION CATHETER Carefully read all instructions prior to use, observe all warnings and precautions noted throughout these instructions. Failure to do so may result in complications. STERILE.
AXS Catalyst Distal Access Catheter
 AXS Catalyst Distal Access Catheter Directions for Use 2 AXS Catalyst Distal Access Catheter ONLY Caution: Federal Law (USA) restricts this device to sale by or on the order of a physician. warning Contents
AXS Catalyst Distal Access Catheter Directions for Use 2 AXS Catalyst Distal Access Catheter ONLY Caution: Federal Law (USA) restricts this device to sale by or on the order of a physician. warning Contents
The SplitWire Percutaneous Transluminal Angioplasty Scoring Device. Instructions for Use
 The SplitWire Percutaneous Transluminal Angioplasty Scoring Device Instructions for Use Contents Contains one (1) SplitWire device. Sterile. Sterilized with ethylene oxide gas. Radiopaque. For single use
The SplitWire Percutaneous Transluminal Angioplasty Scoring Device Instructions for Use Contents Contains one (1) SplitWire device. Sterile. Sterilized with ethylene oxide gas. Radiopaque. For single use
ER REBOA Catheter. Instructions for Use
 ER REBOA Catheter Instructions for Use Prytime Medical Devices, Inc. 229 N. Main Street Boerne, TX 78006, USA feedback@prytimemedical.com www.prytimemedical.com US 1 210 340 0116 U.S. and Foreign Patents
ER REBOA Catheter Instructions for Use Prytime Medical Devices, Inc. 229 N. Main Street Boerne, TX 78006, USA feedback@prytimemedical.com www.prytimemedical.com US 1 210 340 0116 U.S. and Foreign Patents
NON-COMPLIANT PTCA RAPID EXCHANGE DILATATION CATHETER
 Page 1 of 5 NON-COMPLIANT PTCA RAPID EXCHANGE DILATATION CATHETER STERILE. SINGLE USE ONLY. Sterilized with ethylene oxide gas. Non pyrogenic. Do not resterilize. Do not use opened or damaged packages.
Page 1 of 5 NON-COMPLIANT PTCA RAPID EXCHANGE DILATATION CATHETER STERILE. SINGLE USE ONLY. Sterilized with ethylene oxide gas. Non pyrogenic. Do not resterilize. Do not use opened or damaged packages.
We Catch. What Others Miss
 We Catch What Others Miss GORE Embolic Filter Diamond Frame Circumferential Filter Attachment eptfe Filter Media with 100-Micron Pores and Hydrophilic Heparin Coating Diamond Frame designed to enhance
We Catch What Others Miss GORE Embolic Filter Diamond Frame Circumferential Filter Attachment eptfe Filter Media with 100-Micron Pores and Hydrophilic Heparin Coating Diamond Frame designed to enhance
RECOMMENDED INSTRUCTIONS FOR USE
 Rapid Exchange PTCA Dilatation Catheter RECOMMENDED INSTRUCTIONS FOR USE Available in diameters 1.25mm to 4.5mm and in lengths 09mm to 40mm Caution: This device should be used only by physicians trained
Rapid Exchange PTCA Dilatation Catheter RECOMMENDED INSTRUCTIONS FOR USE Available in diameters 1.25mm to 4.5mm and in lengths 09mm to 40mm Caution: This device should be used only by physicians trained
Instructions for Use Cordis PRECISE PRO Rx Nitinol Stent System
 100000017144.3 100000017144.3 Instructions for Use Cordis PRECISE PRO Rx Nitinol Stent System Figure 1.065" (1.65 mm) OR.078" (1.98 mm) 16 4 13 10 11 15 8 3 14.062" (1.57 mm) OR.065" (1.65 mm).038" (0.97
100000017144.3 100000017144.3 Instructions for Use Cordis PRECISE PRO Rx Nitinol Stent System Figure 1.065" (1.65 mm) OR.078" (1.98 mm) 16 4 13 10 11 15 8 3 14.062" (1.57 mm) OR.065" (1.65 mm).038" (0.97
INSTRUCTIONS FOR USE FOR:
 INSTRUCTIONS FOR USE FOR: en English cs Čeština da Dansk nl Nederlands fi Suomi fr Français de Deutsch el Ελληνικά hu Magyar it Italiano lt Lietuvių no Norsk pl Polski pt Português es Español sv Svenska
INSTRUCTIONS FOR USE FOR: en English cs Čeština da Dansk nl Nederlands fi Suomi fr Français de Deutsch el Ελληνικά hu Magyar it Italiano lt Lietuvių no Norsk pl Polski pt Português es Español sv Svenska
1 Comparison of Warfarin and Aspirin for Symptomatic Intracranial Arterial Stenosis. N Engl J Med 352;13, March 31, 2005
 The risk of ischemic stroke in patients with Intracranial Atherosclerotic Disease (ICAD) ranges from 7 to 24%. 1,2 Developed specifically for the treatment of ICAD, the Wingspan Stent System and Gateway
The risk of ischemic stroke in patients with Intracranial Atherosclerotic Disease (ICAD) ranges from 7 to 24%. 1,2 Developed specifically for the treatment of ICAD, the Wingspan Stent System and Gateway
Instructions for Use ANGIOGUARD XP Emboli Capture Guidewire System
 10257072-3 Instructions for Use ANGIOGUARD XP Emboli Capture Guidewire System 1 2 This page has been This page has been 3 CAUTION: Federal (USA) law restricts this device to sale by or on the order of
10257072-3 Instructions for Use ANGIOGUARD XP Emboli Capture Guidewire System 1 2 This page has been This page has been 3 CAUTION: Federal (USA) law restricts this device to sale by or on the order of
PTA Balloon Dilatation Catheter
 PTA Balloon Dilatation Catheter March 2016 1/6(3214-0) Catalogue number Serial number Lot number Use by Do not reuse Do not resterilize Do not use if package is damaged Consult instructions for use Keep
PTA Balloon Dilatation Catheter March 2016 1/6(3214-0) Catalogue number Serial number Lot number Use by Do not reuse Do not resterilize Do not use if package is damaged Consult instructions for use Keep
Assurant Cobalt Iliac BALLOON EXPANDABLE STENT SYSTEM
 Assurant Cobalt Iliac BALLOON EXPANDABLE STENT SYSTEM Innovating for life. CONFORMABILIT Y 6 F S H E AT H C O M PAT I B I L I T Y THE ASSURANT COBALT ILIAC STENT, WITH ITS UNIQUE COBALT CHROMIUM MODULAR
Assurant Cobalt Iliac BALLOON EXPANDABLE STENT SYSTEM Innovating for life. CONFORMABILIT Y 6 F S H E AT H C O M PAT I B I L I T Y THE ASSURANT COBALT ILIAC STENT, WITH ITS UNIQUE COBALT CHROMIUM MODULAR
Single Pass MCA Revascularization with Trevo
 Single Pass MCA Revascularization with Trevo By Concentric Medical ProVueTM Retriever Case Reported By Sascha Prothmann, MD Klinikum rechts der Isar, Munich, Germany Images contained herein courtesy of
Single Pass MCA Revascularization with Trevo By Concentric Medical ProVueTM Retriever Case Reported By Sascha Prothmann, MD Klinikum rechts der Isar, Munich, Germany Images contained herein courtesy of
Understanding aneurysms and flow diversion treatment
 Surpass Streamline Flow Diverter See package insert for complete indications, contraindications, warnings and instructions for use. INTENDED USE / INDICATIONS FOR USE The Surpass Streamline Flow Diverter
Surpass Streamline Flow Diverter See package insert for complete indications, contraindications, warnings and instructions for use. INTENDED USE / INDICATIONS FOR USE The Surpass Streamline Flow Diverter
Directions For Use. All directions should be read before use
 Directions For Use All directions should be read before use DEVICE DESCRIPTION: The CLEANER.XT Rotational Thrombectomy System is a percutaneous, 6Fr catheter based system (single piece construction) that
Directions For Use All directions should be read before use DEVICE DESCRIPTION: The CLEANER.XT Rotational Thrombectomy System is a percutaneous, 6Fr catheter based system (single piece construction) that
PTA Balloon Dilatation Catheter
 PTA Balloon Dilatation Catheter 1/6(3211-0) February 2016 TIS-488-06272017 Catalogue number Serial number Lot number Use by Do not reuse Do not resterilize Do not use if package is damaged Consult instructions
PTA Balloon Dilatation Catheter 1/6(3211-0) February 2016 TIS-488-06272017 Catalogue number Serial number Lot number Use by Do not reuse Do not resterilize Do not use if package is damaged Consult instructions
Peripheral Intravascular Lithotripsy (IVL) Catheter Instructions for Use (IFU)
 Peripheral Intravascular Lithotripsy (IVL) Catheter Instructions for Use (IFU) For use with the Shockwave Medical, Inc. IVL Generator and Connector Cable Indication for Use The Shockwave Medical IVL System
Peripheral Intravascular Lithotripsy (IVL) Catheter Instructions for Use (IFU) For use with the Shockwave Medical, Inc. IVL Generator and Connector Cable Indication for Use The Shockwave Medical IVL System
DURABLE. CONSISTENT. SAFE. IN.PACT Admiral Drug-Coated Balloon
 DURABLE. CONSISTENT. SAFE. Drug-Coated Balloon THE SCIENCE BEHIND THE OUTCOMES DCB has proven, long-term durable outcomes across multiple clinical trials, as well as across complex patient and lesion types.
DURABLE. CONSISTENT. SAFE. Drug-Coated Balloon THE SCIENCE BEHIND THE OUTCOMES DCB has proven, long-term durable outcomes across multiple clinical trials, as well as across complex patient and lesion types.
CLARIVEIN INFUSION CATHETER
 CLARIVEIN INFUSION CATHETER General Product Description Overview The ClariVein Infusion Catheter (ClariVein -IC) is an infusion catheter system designed to introduce physician-specified medicaments into
CLARIVEIN INFUSION CATHETER General Product Description Overview The ClariVein Infusion Catheter (ClariVein -IC) is an infusion catheter system designed to introduce physician-specified medicaments into
System.
 System www.penumbrainc.com POD System Case Examples 45 cm of POD Packing Coil in 1.5 to 3 mm diameter dilating vessel Bronchial Artery Embolization Dr. Amit Kakkar and Dr. Aksim Rivera, Bronx, NY Inflow:
System www.penumbrainc.com POD System Case Examples 45 cm of POD Packing Coil in 1.5 to 3 mm diameter dilating vessel Bronchial Artery Embolization Dr. Amit Kakkar and Dr. Aksim Rivera, Bronx, NY Inflow:
Instructions for Use 1
 Instructions for Use 1 Instructions for Use IMPORTANT! Please read before use. Description The Sniper is a dual lumen catheter comprised of an inner guidewire/infusion lumen and an outer, annular lumen
Instructions for Use 1 Instructions for Use IMPORTANT! Please read before use. Description The Sniper is a dual lumen catheter comprised of an inner guidewire/infusion lumen and an outer, annular lumen
Quick Reference Guide
 Quick Reference Guide Indications for Use The AFX Endovascular AAA System is indicated for endovascular treatment in patients with AAA. The devices are indicated for patients with suitable aneurysm morphology
Quick Reference Guide Indications for Use The AFX Endovascular AAA System is indicated for endovascular treatment in patients with AAA. The devices are indicated for patients with suitable aneurysm morphology
Instructions for Use Reprocessed LASSO Circular Mapping Diagnostic Electrophysiology (EP) Catheter
 Instructions for Use Reprocessed LASSO Circular Mapping Diagnostic Electrophysiology (EP) Catheter Caution: Federal (USA) law restricts this device to sale by or on the order of a physician. DEVICE DESCRIPTION
Instructions for Use Reprocessed LASSO Circular Mapping Diagnostic Electrophysiology (EP) Catheter Caution: Federal (USA) law restricts this device to sale by or on the order of a physician. DEVICE DESCRIPTION
Talent Abdominal Stent Graft
 Talent Abdominal with THE Xcelerant Hydro Delivery System Expanding the Indications for EVAR Treat More Patients Short Necks The Talent Abdominal is the only FDA-approved device for proximal aortic neck
Talent Abdominal with THE Xcelerant Hydro Delivery System Expanding the Indications for EVAR Treat More Patients Short Necks The Talent Abdominal is the only FDA-approved device for proximal aortic neck
REBEL. Platinum Chromium Coronary Stent System. Patient Information Guide
 REBEL Patient Information Guide REBEL PATIENT INFORMATION GUIDE You have recently had a REBEL bare metal stent implanted in the coronary arteries of your heart. The following information is important for
REBEL Patient Information Guide REBEL PATIENT INFORMATION GUIDE You have recently had a REBEL bare metal stent implanted in the coronary arteries of your heart. The following information is important for
CAROTID ARTERY ANGIOPLASTY
 CAROTID ARTERY ANGIOPLASTY Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Medical Coverage Guideline
CAROTID ARTERY ANGIOPLASTY Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Medical Coverage Guideline
Introduction What Causes Peripheral Vascular Disease? How Do Doctors Treat Peripheral Vascular Disease?... 9
 Patient Information Table of Contents Introduction... 3 What is Peripheral Vascular Disease?... 5 What Are Some of the Symptoms of Peripheral Vascular Disease?... 7 What Causes Peripheral Vascular Disease?...
Patient Information Table of Contents Introduction... 3 What is Peripheral Vascular Disease?... 5 What Are Some of the Symptoms of Peripheral Vascular Disease?... 7 What Causes Peripheral Vascular Disease?...
Ruby Coil. Large Volume Detachable Coils
 Ruby Coil Ruby Case Examples 38 mm Hepatic Artery Aneurysm Pulmonary AVM 2 Coils Dr. James Benenati Miami Cardiac and Vascular Institute, FL Y90 Embolization Type 2 Endoleak Dr. J Moskovitz Florida Hospital,
Ruby Coil Ruby Case Examples 38 mm Hepatic Artery Aneurysm Pulmonary AVM 2 Coils Dr. James Benenati Miami Cardiac and Vascular Institute, FL Y90 Embolization Type 2 Endoleak Dr. J Moskovitz Florida Hospital,
Balloon in Balloon (BIB )
 Balloon in Balloon (BIB ) Stent Placement Catheter INSTRUCTIONS FOR USE CAUTION: FEDERAL (USA) LAW RESTRICTS THIS DEVICE TO SALE BY OR ON THE ORDER OF A PHYSICIAN. Read all instructions prior to use. Distributed
Balloon in Balloon (BIB ) Stent Placement Catheter INSTRUCTIONS FOR USE CAUTION: FEDERAL (USA) LAW RESTRICTS THIS DEVICE TO SALE BY OR ON THE ORDER OF A PHYSICIAN. Read all instructions prior to use. Distributed
Introduction 3. What is Peripheral Vascular Disease? 5. What Are Some of the Symptoms of Peripheral Vascular Disease? 6
 Patient Information Table of Contents Introduction 3 What is Peripheral Vascular Disease? 5 What Are Some of the Symptoms of Peripheral Vascular Disease? 6 What Causes Peripheral Vascular Disease? 7 How
Patient Information Table of Contents Introduction 3 What is Peripheral Vascular Disease? 5 What Are Some of the Symptoms of Peripheral Vascular Disease? 6 What Causes Peripheral Vascular Disease? 7 How
Advanced Innovation for Exceptional Performance
 Advanced Innovation for Exceptional Performance longest BALLOON LENGTHS ON THE MARKET Bard Peripheral Vascular Ultraverse 014 300 mm Boston Scientific Coyote 220 mm Cordis SLEEK RX 220 mm Aviator Plus
Advanced Innovation for Exceptional Performance longest BALLOON LENGTHS ON THE MARKET Bard Peripheral Vascular Ultraverse 014 300 mm Boston Scientific Coyote 220 mm Cordis SLEEK RX 220 mm Aviator Plus
Radux StandTall Instructions for Use Sheath Extender and Securement Clasp
 Radux StandTall Instructions for Use Sheath Extender and Securement Clasp USA Caution Federal (USA) law restricts this device to sale by or on the order of a health care professional. Caution The Radux
Radux StandTall Instructions for Use Sheath Extender and Securement Clasp USA Caution Federal (USA) law restricts this device to sale by or on the order of a health care professional. Caution The Radux
Figure 1: Revolution TM Peripheral Atherectomy System Diagram. Table 1: Revolution TM Peripheral Atherectomy System Specifications Minimum Burr
 Instructions For Use All instructions should be read before use CAUTION Investigational Device. Limited by Federal (or United States) law to investigational use. DEVICE DESCRIPTION: The Revolution TM Peripheral
Instructions For Use All instructions should be read before use CAUTION Investigational Device. Limited by Federal (or United States) law to investigational use. DEVICE DESCRIPTION: The Revolution TM Peripheral
Peel-Apart Percutaneous Introducer Kits for
 Bard Access Systems Peel-Apart Percutaneous Introducer Kits for Table of Contents Contents Page Bard Implanted Ports Hickman*, Leonard*, Broviac*, Tenckhoff*, and Groshong* Catheters Introduction....................................
Bard Access Systems Peel-Apart Percutaneous Introducer Kits for Table of Contents Contents Page Bard Implanted Ports Hickman*, Leonard*, Broviac*, Tenckhoff*, and Groshong* Catheters Introduction....................................
PTCA Dilatation Catheter. Instructions for Use
 PTCA Dilatation Catheter Instructions for Use Rx ONLY Caution: Federal law restricts this device to sale by or on the order of a physician STERILE. Sterilized with ethylene oxide gas Distributed by Medinol
PTCA Dilatation Catheter Instructions for Use Rx ONLY Caution: Federal law restricts this device to sale by or on the order of a physician STERILE. Sterilized with ethylene oxide gas Distributed by Medinol
Peter A. Soukas, M.D., FACC, FSVM, FSCAI, RPVI
 Peter A. Soukas, M.D., FACC, FSVM, FSCAI, RPVI Director, Peripheral Vascular Interventional Laboratory Director, Vascular & Endovascular Medicine Fellowship Program Assistant Professor of Medicine The
Peter A. Soukas, M.D., FACC, FSVM, FSCAI, RPVI Director, Peripheral Vascular Interventional Laboratory Director, Vascular & Endovascular Medicine Fellowship Program Assistant Professor of Medicine The
PTCA Dilatation Catheter. Instructions for Use
 PTCA Dilatation Catheter Instructions for Use Rx ONLY Caution: Federal law restricts this device to sale by or on the order of a physician STERILE. Sterilized with ethylene oxide gas Distributed by Medinol
PTCA Dilatation Catheter Instructions for Use Rx ONLY Caution: Federal law restricts this device to sale by or on the order of a physician STERILE. Sterilized with ethylene oxide gas Distributed by Medinol
Carotid Artery Stenting
 Carotid Artery Stenting JESSICA MITCHELL, ACNP CENTRAL ILLINOIS RADIOLOGICAL ASSOCIATES External Carotid Artery (ECA) can easily be identified from Internal Carotid Artery (ICA) by noticing the branches.
Carotid Artery Stenting JESSICA MITCHELL, ACNP CENTRAL ILLINOIS RADIOLOGICAL ASSOCIATES External Carotid Artery (ECA) can easily be identified from Internal Carotid Artery (ICA) by noticing the branches.
Balloon in Balloon (BIB )
 in (BIB ) Dilatation Catheter INSTRUCTIONS FOR USE CAUTION: FEDERAL (USA) LAW RESTRICTS THIS DEVICE TO SALE BY OR ON THE ORDER OF A PHYSICIAN. Read all instructions prior to use. Distributed by: B. Braun
in (BIB ) Dilatation Catheter INSTRUCTIONS FOR USE CAUTION: FEDERAL (USA) LAW RESTRICTS THIS DEVICE TO SALE BY OR ON THE ORDER OF A PHYSICIAN. Read all instructions prior to use. Distributed by: B. Braun
Neuroform Microdelivery Stent System
 Neuroform Microdelivery Stent System Patient Information Booklet TABLE OF CONTENTS What is the Purpose of This Booklet?...2 What is a Wide Neck, Intracranial Aneurysm?...2 What is the Treatment for Wide
Neuroform Microdelivery Stent System Patient Information Booklet TABLE OF CONTENTS What is the Purpose of This Booklet?...2 What is a Wide Neck, Intracranial Aneurysm?...2 What is the Treatment for Wide
Carotid Artery Stenting
 Carotid Artery Stenting Natural history of the carotid stenosis Asymptomatic 80% carotid stenosis - 6% risk of stroke / year Symptomatic carotid stenosis have 10% risk of CVA at one year and 40% at 5 years
Carotid Artery Stenting Natural history of the carotid stenosis Asymptomatic 80% carotid stenosis - 6% risk of stroke / year Symptomatic carotid stenosis have 10% risk of CVA at one year and 40% at 5 years
INSTRUCTIONS FOR USE
 PERICARDIUM COVERED STENT (PCS) INSTRUCTIONS FOR USE STERILE - Ethylene Oxide Sterilized. For single use only. Do not re-sterilize. THE CLINICAL TECHNIQUES AND PROCEDURES DESCRIBED HERE DO NOT REPRESENT
PERICARDIUM COVERED STENT (PCS) INSTRUCTIONS FOR USE STERILE - Ethylene Oxide Sterilized. For single use only. Do not re-sterilize. THE CLINICAL TECHNIQUES AND PROCEDURES DESCRIBED HERE DO NOT REPRESENT
Innovation by design. Technology that sets a new standard
 Innovation by design Technology that sets a new standard Flexible nitinol scoring element with three rectangular spiral struts works in tandem with a semi-compliant balloon to score the target lesion Balloon
Innovation by design Technology that sets a new standard Flexible nitinol scoring element with three rectangular spiral struts works in tandem with a semi-compliant balloon to score the target lesion Balloon
Chapter 4 Section 9.1
 Surgery Chapter 4 Section 9.1 Issue Date: August 26, 1985 Authority: 32 CFR 199.4(c)(2) and (c)(3) 1.0 CPT 1 PROCEDURE CODES 33010-33130, 33140, 33141, 33361-33369, 33200-37186, 37195-37785, 92950-93272,
Surgery Chapter 4 Section 9.1 Issue Date: August 26, 1985 Authority: 32 CFR 199.4(c)(2) and (c)(3) 1.0 CPT 1 PROCEDURE CODES 33010-33130, 33140, 33141, 33361-33369, 33200-37186, 37195-37785, 92950-93272,
FROM THE EVERYDAY TO THE EXTRAORDINARY
 FROM THE EVERYDAY TO THE EXTRAORDINARY Created with the collaboration of more than 250 physicians around the world, ENDURANT empowers you to create stronger outcomes for more patients, including those
FROM THE EVERYDAY TO THE EXTRAORDINARY Created with the collaboration of more than 250 physicians around the world, ENDURANT empowers you to create stronger outcomes for more patients, including those
Chapter 4 Section 9.1
 Surgery Chapter 4 Section 9.1 Issue Date: August 26, 1985 Authority: 32 CFR 199.4(c)(2) and (c)(3) 1.0 CPT 1 PROCEDURE CODES 33010-33130, 33140, 33141, 33200-37186, 37195-37785, 92950-93272, 93303-93581,
Surgery Chapter 4 Section 9.1 Issue Date: August 26, 1985 Authority: 32 CFR 199.4(c)(2) and (c)(3) 1.0 CPT 1 PROCEDURE CODES 33010-33130, 33140, 33141, 33200-37186, 37195-37785, 92950-93272, 93303-93581,
Cardiva Catalyst III INSTRUCTIONS FOR USE
 Cardiva Catalyst III INSTRUCTIONS FOR USE IFU 2469 Rev. G CAUTION Federal (USA) law restricts this device to sale by or on the order of a physician. For single use only GRAPHICAL SYMBOLS ON THE CARDIVA
Cardiva Catalyst III INSTRUCTIONS FOR USE IFU 2469 Rev. G CAUTION Federal (USA) law restricts this device to sale by or on the order of a physician. For single use only GRAPHICAL SYMBOLS ON THE CARDIVA
Bifurcated system Proximal suprarenal stent Modular (aortic main body and two iliac legs) Full thickness woven polyester graft material Fully
 Physician Training Bifurcated system Proximal suprarenal stent Modular (aortic main body and two iliac legs) Full thickness woven polyester graft material Fully supported by self-expanding z-stents H&L-B
Physician Training Bifurcated system Proximal suprarenal stent Modular (aortic main body and two iliac legs) Full thickness woven polyester graft material Fully supported by self-expanding z-stents H&L-B
Directions For Use. All directions should be read before use. Page 1 of 8
 Directions For Use All directions should be read before use Page 1 of 8 WARNING: For single use only. Do not reuse, reprocess or re-sterilize. Reuse, reprocessing or re-sterilization may compromise the
Directions For Use All directions should be read before use Page 1 of 8 WARNING: For single use only. Do not reuse, reprocess or re-sterilize. Reuse, reprocessing or re-sterilization may compromise the
Access More Patients. Customize Each Seal.
 Access More. Customize Each Seal. The Least Invasive Path Towards Proven Patency ULTRA LOW PROFILE TO EASE ADVANCEMENT The flexible, ultra-low 12F ID Ovation ix delivery system enables you to navigate
Access More. Customize Each Seal. The Least Invasive Path Towards Proven Patency ULTRA LOW PROFILE TO EASE ADVANCEMENT The flexible, ultra-low 12F ID Ovation ix delivery system enables you to navigate
Phoenix Atherectomy System Product Overview /LB
 Phoenix Atherectomy System Product Overview Phoenix Atherectomy System- The next generation of atherectomy The first hybrid atherectomy system available Combines the benefits of existing atherectomy systems
Phoenix Atherectomy System Product Overview Phoenix Atherectomy System- The next generation of atherectomy The first hybrid atherectomy system available Combines the benefits of existing atherectomy systems
ASAHI Corsair INSTRUCTIONS FOR USE
 ASAHI Corsair INSTRUCTIONS FOR USE SYMBOLS Legal manufacturer Do not use if package is damaged GC Minimum GC I.D. REF Catalogue number Caution, consult accompanying documents Maximum injection pressure
ASAHI Corsair INSTRUCTIONS FOR USE SYMBOLS Legal manufacturer Do not use if package is damaged GC Minimum GC I.D. REF Catalogue number Caution, consult accompanying documents Maximum injection pressure
Issam D. Moussa, MD. Professor of Medicine Mayo Clinic College of Medicine Chair, Division of Cardiovascular Diseases Mayo Clinic Jacksonville, FL
 Carotid Technologies and Protection Issam D. Moussa, MD Professor of Medicine Mayo Clinic College of Medicine Chair, Division of Cardiovascular Diseases Mayo Clinic Jacksonville, FL Disclosure Statement
Carotid Technologies and Protection Issam D. Moussa, MD Professor of Medicine Mayo Clinic College of Medicine Chair, Division of Cardiovascular Diseases Mayo Clinic Jacksonville, FL Disclosure Statement
COIL SYSTEM ORDERING INFORMATION
 EMBOLIZATION SYSTEM RUBY COIL SYSTEM FRAME with Ruby Standard FILL with Ruby Soft Now with 3 & 40 mm sizes! SYSTEM ANCHOR with Now designed to embolize 3 4 mm vessels! PACK with Coil Now with WAVE Shape
EMBOLIZATION SYSTEM RUBY COIL SYSTEM FRAME with Ruby Standard FILL with Ruby Soft Now with 3 & 40 mm sizes! SYSTEM ANCHOR with Now designed to embolize 3 4 mm vessels! PACK with Coil Now with WAVE Shape
Z-MED II BALLOON AORTIC AND PULMONIC VALVULOPLASTY CATHETER. Instructions for Use
 Z-MED II BALLOON AORTIC AND PULMONIC VALVULOPLASTY CATHETER Instructions for Use CAUTION: Federal (USA) Law restricts this device to sale by or on the order of a physician. Manufactured for: B. Braun Interventional
Z-MED II BALLOON AORTIC AND PULMONIC VALVULOPLASTY CATHETER Instructions for Use CAUTION: Federal (USA) Law restricts this device to sale by or on the order of a physician. Manufactured for: B. Braun Interventional
GORE EXCLUDER AAA Endoprosthesis ANNUAL CLINICAL UPDATE OCTOBER Section I Clinical experience. Section II Worldwide commercial experience
 GORE EXCLUDER AAA Endoprosthesis ANNUAL CLINICAL UPDATE OCTOBER 2018 Abstract This annual clinical update provides a review of the ongoing experience with the GORE EXCLUDER AAA Endoprosthesis used in the
GORE EXCLUDER AAA Endoprosthesis ANNUAL CLINICAL UPDATE OCTOBER 2018 Abstract This annual clinical update provides a review of the ongoing experience with the GORE EXCLUDER AAA Endoprosthesis used in the
Document No. BMB/IFU/40 Rev No. & Date 00 & 15/11/2017 Issue No & Date 01 & 15/11/2017
 Central Venous Catheter Device Description Multi-lumen catheters incorporate separate, non-communicating vascular access lumens within a single catheter body. Minipunctur Access Sets And Trays: Used for
Central Venous Catheter Device Description Multi-lumen catheters incorporate separate, non-communicating vascular access lumens within a single catheter body. Minipunctur Access Sets And Trays: Used for
Caution: Federal (U.S.A.) law restricts this device to sale by or on the order of a physician.
 Page 1 of 5 Reprocessed by Instructions for Use Reprocessed Ultrasound Catheters Reprocessed Device for Single Use Caution: Federal (U.S.A.) law restricts this device to sale by or on the order of a physician.
Page 1 of 5 Reprocessed by Instructions for Use Reprocessed Ultrasound Catheters Reprocessed Device for Single Use Caution: Federal (U.S.A.) law restricts this device to sale by or on the order of a physician.
INSTRUCTIONS FOR USE FOR:
 INSTRUCTIONS FOR USE FOR: en English INSTRUCTIONS FOR USE GORE ENFORM PREPERITONEAL BIOMATERIAL Carefully read all instructions prior to use. Observe all instructions, warnings, and precautions noted throughout.
INSTRUCTIONS FOR USE FOR: en English INSTRUCTIONS FOR USE GORE ENFORM PREPERITONEAL BIOMATERIAL Carefully read all instructions prior to use. Observe all instructions, warnings, and precautions noted throughout.
Current Non-Surgical Cardiac Interventions. By Pam Bayles, RN, BSN
 Current Non-Surgical Cardiac Interventions By Pam Bayles, RN, BSN Balloon Angioplasty & DES A balloon-tipped catheter was first used in 1964 to treat a cause of atherosclerotic disease in a patient s leg
Current Non-Surgical Cardiac Interventions By Pam Bayles, RN, BSN Balloon Angioplasty & DES A balloon-tipped catheter was first used in 1964 to treat a cause of atherosclerotic disease in a patient s leg
Sustained Release. Superior Results.
 ELUVIA Drug-Eluting Vascular Stent System Sustained Release. Superior Results. Superiority determined in Post Hoc Superiority Analysis. 12-Month Primary Patency rate of 86.8% in the Eluvia arm (n=309)
ELUVIA Drug-Eluting Vascular Stent System Sustained Release. Superior Results. Superiority determined in Post Hoc Superiority Analysis. 12-Month Primary Patency rate of 86.8% in the Eluvia arm (n=309)
INSTRUCTIONS FOR USE FOR: English
 INSTRUCTIONS FOR USE FOR: en English fr INSTRUCTIONS FOR USE FOR: GORE PROPATEN Vascular Graft I. INDICATIONS FOR USE II. III. IV. CONTRAINDICATIONS US CLINICAL EXPERIENCE PACKAGE HANDLING V. TECHNICAL
INSTRUCTIONS FOR USE FOR: en English fr INSTRUCTIONS FOR USE FOR: GORE PROPATEN Vascular Graft I. INDICATIONS FOR USE II. III. IV. CONTRAINDICATIONS US CLINICAL EXPERIENCE PACKAGE HANDLING V. TECHNICAL
Initial placement 20FR Guidewire PEG kit REORDER NO:
 Initial placement 20FR Guidewire PEG kit REORDER NO: 00710802 INSTRUCTIONS FOR USE 1 of 5 These products have been manufactured not to include latex. Intended Use: The Initial placement 20FR Guidewire
Initial placement 20FR Guidewire PEG kit REORDER NO: 00710802 INSTRUCTIONS FOR USE 1 of 5 These products have been manufactured not to include latex. Intended Use: The Initial placement 20FR Guidewire
Initial placement 24FR Pull PEG kit REORDER NO:
 Initial placement 24FR Pull PEG kit REORDER NO: 00710805 INSTRUCTIONS FOR USE 1 of 5 These products have been manufactured not to include latex. Intended Use: The Initial placement 24FR Pull PEG kit is
Initial placement 24FR Pull PEG kit REORDER NO: 00710805 INSTRUCTIONS FOR USE 1 of 5 These products have been manufactured not to include latex. Intended Use: The Initial placement 24FR Pull PEG kit is
Pocket Reference Guide For (CTO) Technologies
 to receive our latest news and key activities. Pocket Reference Guide For (CTO) Technologies LinkedIn page Follow us on CORDIS EMEA Table of Contents Essential prescribing information for both FRONTRUNNER
to receive our latest news and key activities. Pocket Reference Guide For (CTO) Technologies LinkedIn page Follow us on CORDIS EMEA Table of Contents Essential prescribing information for both FRONTRUNNER
New Trials in Progress: ACT 1. Jon Matsumura, MD Cannes, France June 28, 2008
 New Trials in Progress: ACT 1 Jon Matsumura, MD Cannes, France June 28, 2008 Faculty Disclosure I disclose the following financial relationships: Consultant, CAS training director, and/or research grants
New Trials in Progress: ACT 1 Jon Matsumura, MD Cannes, France June 28, 2008 Faculty Disclosure I disclose the following financial relationships: Consultant, CAS training director, and/or research grants
Zenith Renu AAA Converter Graft. Device Description Planning and Sizing Deployment Sequence Patient Follow-Up
 Zenith Renu AAA Converter Graft Device Description Planning and Sizing Deployment Sequence Patient Follow-Up Device description: Device indications The Zenith Renu AAA Converter Graft with Z-Trak Introduction
Zenith Renu AAA Converter Graft Device Description Planning and Sizing Deployment Sequence Patient Follow-Up Device description: Device indications The Zenith Renu AAA Converter Graft with Z-Trak Introduction
ULTRA ICE PLUS ULTRASOUND IMAGING CATHETER. Know where you are. See what you want to avoid.
 ULTRA ICE PLUS ULTRASOUND IMAGING CATHETER Know where you are. See what you want to avoid. ULTRA ICE PLUS for Transseptal Puncture Know where you are. See what you want to avoid. Fluoroscopy alone provides
ULTRA ICE PLUS ULTRASOUND IMAGING CATHETER Know where you are. See what you want to avoid. ULTRA ICE PLUS for Transseptal Puncture Know where you are. See what you want to avoid. Fluoroscopy alone provides
CAREFULLY READ ALL INSTRUCTIONS PRIOR TO USE
 CAREFULLY READ ALL INSTRUCTIONS PRIOR TO USE INDICATIONS FOR USE The LATERA Absorbable Nasal Implant is indicated for supporting upper and lower lateral nasal cartilage. CAUTION: Federal law restricts
CAREFULLY READ ALL INSTRUCTIONS PRIOR TO USE INDICATIONS FOR USE The LATERA Absorbable Nasal Implant is indicated for supporting upper and lower lateral nasal cartilage. CAUTION: Federal law restricts
Finally, the Control You Need to Deliver Accurate Treatment
 Finally, the Control You Need to Deliver Accurate Treatment LIFESTREAM Covered Stent. has joined BD When you reach for a balloon expandable stent, you require accuracy. The LIFESTREAM Balloon Expandable
Finally, the Control You Need to Deliver Accurate Treatment LIFESTREAM Covered Stent. has joined BD When you reach for a balloon expandable stent, you require accuracy. The LIFESTREAM Balloon Expandable
PERFORMANCE YOU CAN TRUST. EverFlex Self-expanding Peripheral Stent with Entrust Delivery System
 PERFORMANCE YOU CAN TRUST EverFlex Self-expanding Peripheral Stent with Entrust Delivery System The Entrust Delivery System is designed to provide improved patient outcomes and procedural efficiency. The
PERFORMANCE YOU CAN TRUST EverFlex Self-expanding Peripheral Stent with Entrust Delivery System The Entrust Delivery System is designed to provide improved patient outcomes and procedural efficiency. The
VERNACULAR Trial & Clinical Experience with the VENOVO Venous Stent
 Stephen Black, MD VERNACULAR Trial & Clinical Experience with the VENOVO Venous Stent 1 Speaker Disclaimers The speakers presentation today is on behalf of Bard Peripheral Vascular, Inc. Any discussion
Stephen Black, MD VERNACULAR Trial & Clinical Experience with the VENOVO Venous Stent 1 Speaker Disclaimers The speakers presentation today is on behalf of Bard Peripheral Vascular, Inc. Any discussion
Chapter 4 Section 9.1
 Surgery Chapter 4 Section 9.1 Issue Date: August 26, 1985 Authority: 32 CFR 199.4(c)(2) and (c)(3) 1.0 CPT 1 PROCEDURE CODES 33010-33130, 33140, 33141, 33361-33369, 33200-37186, 37195-37785, 92950-93272,
Surgery Chapter 4 Section 9.1 Issue Date: August 26, 1985 Authority: 32 CFR 199.4(c)(2) and (c)(3) 1.0 CPT 1 PROCEDURE CODES 33010-33130, 33140, 33141, 33361-33369, 33200-37186, 37195-37785, 92950-93272,
History of the Powerlink System Design and Clinical Results. Edward B. Diethrich Arizona Heart Hospital Phoenix, AZ
 History of the Powerlink System Design and Clinical Results Edward B. Diethrich Arizona Heart Hospital Phoenix, AZ Powerlink System: Unibody-Bifurcated Design Long Main Body Low-Porosity Proprietary eptfe
History of the Powerlink System Design and Clinical Results Edward B. Diethrich Arizona Heart Hospital Phoenix, AZ Powerlink System: Unibody-Bifurcated Design Long Main Body Low-Porosity Proprietary eptfe
PrOspective multicenter study of carotid artery stenting Usinng mer Stent OCEANUS study!!!
 PrOspective multicenter study of carotid artery stenting Usinng mer Stent OCEANUS study!!! Prof Piotr Pieniazek MD PhD Jagiellonian University Institute of Cardiology, John Paul II Hospital Krakow, Poland
PrOspective multicenter study of carotid artery stenting Usinng mer Stent OCEANUS study!!! Prof Piotr Pieniazek MD PhD Jagiellonian University Institute of Cardiology, John Paul II Hospital Krakow, Poland
MAXIMIZE RADIAL SOLUTIONS TO PERIPHERAL CHALLENGES
 MAXIMIZE RADIAL SOLUTIONS TO PERIPHERAL CHALLENGES PUSHING BOUNDARIES Terumo Interventional Systems is committed to your success with innovative procedural solutions and ongoing support for your most challenging
MAXIMIZE RADIAL SOLUTIONS TO PERIPHERAL CHALLENGES PUSHING BOUNDARIES Terumo Interventional Systems is committed to your success with innovative procedural solutions and ongoing support for your most challenging
William A. Gray MD System Chief of Cardiovascular Services, Main Line Health President, Lankenau Heart Institute Wynnewood, PA USA
 William A. Gray MD System Chief of Cardiovascular Services, President, Wynnewood, PA USA What are the possible causes of stroke in CAS? Operator error Technique (balloon sizing, wire misadventure, EPD
William A. Gray MD System Chief of Cardiovascular Services, President, Wynnewood, PA USA What are the possible causes of stroke in CAS? Operator error Technique (balloon sizing, wire misadventure, EPD
Neuroform EZ Stent System
 Neuroform EZ Stent System Directions for Use 2 Neuroform EZ Stent System 1 cm 1.9 cm 135 cm 185 cm ONLY Caution: Federal Law (USA) restricts this device to sale by or on the order of a physician. warning
Neuroform EZ Stent System Directions for Use 2 Neuroform EZ Stent System 1 cm 1.9 cm 135 cm 185 cm ONLY Caution: Federal Law (USA) restricts this device to sale by or on the order of a physician. warning
MICHAEL R. JAFF, DO MASSACHUSETTS, UNITED STATES. Medtronic Further. Together
 DRUG-COATED BALL0ON TREATMENT FOR PATIENTS WITH INTERMITTENT CLAUDICATION: INSIGHTS FROM THE IN.PACT GLOBAL FULL CLINICAL COHORT MICHAEL R. JAFF, DO MASSACHUSETTS, UNITED STATES Medtronic Further. Together
DRUG-COATED BALL0ON TREATMENT FOR PATIENTS WITH INTERMITTENT CLAUDICATION: INSIGHTS FROM THE IN.PACT GLOBAL FULL CLINICAL COHORT MICHAEL R. JAFF, DO MASSACHUSETTS, UNITED STATES Medtronic Further. Together
Carotid Artery Stent: Is it ready for prime time?
 2010 CATH LAB SYMPOSIUM Carotid Artery Stent: Is it ready for prime time? Luis F. Tami, MD, FACC, FSCAI Interventional Cardiology and Vascular Medicine Memorial Regional Hospital August 2010 CAE and CAS
2010 CATH LAB SYMPOSIUM Carotid Artery Stent: Is it ready for prime time? Luis F. Tami, MD, FACC, FSCAI Interventional Cardiology and Vascular Medicine Memorial Regional Hospital August 2010 CAE and CAS
SCAFFOLD Study Gore PTFE mesh-covered stent preclinical and clinical data so far. Peter A. Schneider, MD Kaiser Foundation Hospital Honolulu, Hawaii
 SCAFFOLD Study Gore PTFE mesh-covered stent preclinical and clinical data so far Peter A. Schneider, MD Kaiser Foundation Hospital Honolulu, Hawaii Disclosure Peter A. Schneider... I have the following
SCAFFOLD Study Gore PTFE mesh-covered stent preclinical and clinical data so far Peter A. Schneider, MD Kaiser Foundation Hospital Honolulu, Hawaii Disclosure Peter A. Schneider... I have the following
DISRUPT PAD. (( Data Summary )) DISRUPT PAD Data Summary SPL Rev. B 2016 Shockwave Medical Inc. All rights reserved.
 DISRUPT PAD (( Data Summary )) DISRUPT PAD Data Summary SPL 60971 Rev. B 1 Summary of the key findings from the DISRUPT PAD Study 99% of femoropopliteal lesions treated were moderately or severely calcified.
DISRUPT PAD (( Data Summary )) DISRUPT PAD Data Summary SPL 60971 Rev. B 1 Summary of the key findings from the DISRUPT PAD Study 99% of femoropopliteal lesions treated were moderately or severely calcified.
Azur CX 35 Peripheral Coil System (Detachable) Instructions for Use
 Azur CX 35 Peripheral Coil System (Detachable) Instructions for Use DEVICE DESCRIPTION The Detachable Azur CX 35 Peripheral Coil System (Azur system) consists of a coil implant attached to a delivery system.
Azur CX 35 Peripheral Coil System (Detachable) Instructions for Use DEVICE DESCRIPTION The Detachable Azur CX 35 Peripheral Coil System (Azur system) consists of a coil implant attached to a delivery system.
Stellarex Drug-coated 0.035" Angioplasty Balloon. Featuring EnduraCoat Technology. The Clear DCB Choice
 Stellarex Drug-coated 0.035" Angioplasty Balloon Featuring EnduraCoat Technology The Clear DCB Choice Top-tier outcomes with the lowest therapeutic drug dose in all patients common to complex 89.0 % patency
Stellarex Drug-coated 0.035" Angioplasty Balloon Featuring EnduraCoat Technology The Clear DCB Choice Top-tier outcomes with the lowest therapeutic drug dose in all patients common to complex 89.0 % patency
Single Use Curlew TM Multiple Biopsy Forceps
 Single Use Curlew TM Multiple Biopsy Forceps 13 SPECIMEN WITH METAL STORAGE CYLINDER With In Situ Fixation, Batch or Single Specimen Collection US Patents 5,782,747; 5,980,468; 6,071,248; foreign patents
Single Use Curlew TM Multiple Biopsy Forceps 13 SPECIMEN WITH METAL STORAGE CYLINDER With In Situ Fixation, Batch or Single Specimen Collection US Patents 5,782,747; 5,980,468; 6,071,248; foreign patents
Introducing a New Treatment Method for Brain Aneurysms
 Pipeline Embolization Device Introducing a New Treatment Method for Brain Aneurysms UNDERSTANDING ANEURYSMS What is a brain aneurysm? An aneurysm is an outpouching in an artery caused by weakness in the
Pipeline Embolization Device Introducing a New Treatment Method for Brain Aneurysms UNDERSTANDING ANEURYSMS What is a brain aneurysm? An aneurysm is an outpouching in an artery caused by weakness in the
CAROTID ARTERY ANGIOPLASTY
 CAROTID ARTERY ANGIOPLASTY Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services, procedures, medical devices and
CAROTID ARTERY ANGIOPLASTY Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services, procedures, medical devices and
Vascu-PICC WITH CUFF PERIPHERALLY INSERTED CENTRAL VEIN ACCESS CATHETER INSTRUCTIONS FOR USE
 Vascu-PICC WITH CUFF PERIPHERALLY INSERTED CENTRAL VEIN ACCESS CATHETER INSTRUCTIONS FOR USE INDICATIONS FOR USE: The Vascu-PICC with cuff Peripherally Inserted Central Vein Catheters are designed for
Vascu-PICC WITH CUFF PERIPHERALLY INSERTED CENTRAL VEIN ACCESS CATHETER INSTRUCTIONS FOR USE INDICATIONS FOR USE: The Vascu-PICC with cuff Peripherally Inserted Central Vein Catheters are designed for
Emerge. PTCA Dilatation Catheter ONLY. monorail. over-the-wire
 90990229-01 Emerge monorail over-the-wire PTCA Dilatation Catheter Table of Contents 2014-09 < EN > warning...1 device description...1 Table 1. Emerge Balloon Coatings...1 Contents...1 Intended use/indications
90990229-01 Emerge monorail over-the-wire PTCA Dilatation Catheter Table of Contents 2014-09 < EN > warning...1 device description...1 Table 1. Emerge Balloon Coatings...1 Contents...1 Intended use/indications
Excimer Laser for Coronary Intervention: Case Study RADIAL APPROACH: CORONARY LASER ATHERECTOMY FOR CTO OF THE LAD FOLLOWED BY PTCA NO STENTING
 Excimer Laser for Coronary Intervention: Case Study RADIAL APPROACH: CORONARY LASER ATHERECTOMY FOR CTO OF THE LAD FOLLOWED BY PTCA NO STENTING 1 2013 Spectranetics. All Rights Reserved. Approved for External
Excimer Laser for Coronary Intervention: Case Study RADIAL APPROACH: CORONARY LASER ATHERECTOMY FOR CTO OF THE LAD FOLLOWED BY PTCA NO STENTING 1 2013 Spectranetics. All Rights Reserved. Approved for External
Acute dissections of the descending thoracic aorta (Debakey
 Endovascular Treatment of Acute Descending Thoracic Aortic Dissections Nimesh D. Desai, MD, PhD, and Joseph E. Bavaria, MD Acute dissections of the descending thoracic aorta (Debakey type III or Stanford
Endovascular Treatment of Acute Descending Thoracic Aortic Dissections Nimesh D. Desai, MD, PhD, and Joseph E. Bavaria, MD Acute dissections of the descending thoracic aorta (Debakey type III or Stanford
MULTILAYER FLOW MODULATOR (MFM ) FOR PERIPHERAL ANEURYSMS TREATMENT (LOWER LIMBS, VISCERALS AND SUBCLAVIAN)
 0459 MULTILAYER FLOW MODULATOR (MFM ) FOR PERIPHERAL ANEURYSMS TREATMENT (LOWER LIMBS, VISCERALS AND SUBCLAVIAN) WITH ITS ANGIOGRAPHIC CONTROL DELIVERY SYSTEM INSTRUCTIONS FOR USE (I.F.U.) English Français
0459 MULTILAYER FLOW MODULATOR (MFM ) FOR PERIPHERAL ANEURYSMS TREATMENT (LOWER LIMBS, VISCERALS AND SUBCLAVIAN) WITH ITS ANGIOGRAPHIC CONTROL DELIVERY SYSTEM INSTRUCTIONS FOR USE (I.F.U.) English Français
Desara TV and Desara Blue TV
 Desara TV and Desara Blue TV Sling for Female Stress Urinary Incontinence Instructions For Use D I Prescription Use only Do not reuse Sterilized using ethylene oxide Available Electronically M Manufactured
Desara TV and Desara Blue TV Sling for Female Stress Urinary Incontinence Instructions For Use D I Prescription Use only Do not reuse Sterilized using ethylene oxide Available Electronically M Manufactured
TIPS & TRICKS for optimal SILK placement
 I. Antiplatelet Premedication: The correctly chosen premedication is essential. PLAVIX (minimum 600mg) should ideally be administered during eight days prior to procedure (75mg per day). However, a loading
I. Antiplatelet Premedication: The correctly chosen premedication is essential. PLAVIX (minimum 600mg) should ideally be administered during eight days prior to procedure (75mg per day). However, a loading
Challenges. 1. Sizing. 2. Proximal landing zone 3. Distal landing zone 4. Access vessels 5. Spinal cord ischemia 6. Endoleak
 Disclosure I have the following potential conflicts of interest to report: Consulting: Medtronic, Gore Employment in industry Stockholder of a healthcare company Owner of a healthcare company Other(s)
Disclosure I have the following potential conflicts of interest to report: Consulting: Medtronic, Gore Employment in industry Stockholder of a healthcare company Owner of a healthcare company Other(s)
Carotid Artery Stenting
 Carotid Artery Stenting Woong Chol Kang M.D. Gil Medical Center, Gachon University of Medicine and Science, Incheon, Korea Carotid Stenosis and Stroke ~25% of stroke is due to carotid disease, the reminder
Carotid Artery Stenting Woong Chol Kang M.D. Gil Medical Center, Gachon University of Medicine and Science, Incheon, Korea Carotid Stenosis and Stroke ~25% of stroke is due to carotid disease, the reminder
Device Preparation (all steps to be performed per standard interventional technique)
 sertera Biopsy Device Instructions for Use Please read all information carefully. Failure to properly follow the instructions may lead to unintended consequences. Important: This package insert is designed
sertera Biopsy Device Instructions for Use Please read all information carefully. Failure to properly follow the instructions may lead to unintended consequences. Important: This package insert is designed
