HepG2 Cells Expressing MicroRNA mir-122 Support the Entire Hepatitis C Virus Life Cycle
|
|
|
- Imogen Dickerson
- 6 years ago
- Views:
Transcription
1 JOURNAL OF VIROLOGY, Nov. 2011, p Vol. 85, No X/11/$12.00 doi: /jvi Copyright 2011, American Society for Microbiology. All Rights Reserved. HepG2 Cells Expressing MicroRNA mir-122 Support the Entire Hepatitis C Virus Life Cycle Christopher M. Narbus, 1 Benjamin Israelow, 1 Marion Sourisseau, 1 Maria L. Michta, 1 Sharon E. Hopcraft, 1 Gusti M. Zeiner, 2 and Matthew J. Evans 1 * Department of Microbiology, Mount Sinai School of Medicine, New York, New York 10029, 1 and Department of Microbiology and Immunology, Stanford University School of Medicine, Stanford, California 2 Received 29 July 2011/Accepted 2 September 2011 The liver-specific microrna mir-122 is required for efficient hepatitis C virus (HCV) RNA replication both in cell culture and in vivo. In addition, nonhepatic cells have been rendered more efficient at supporting this stage of the HCV life cycle by mir-122 expression. This study investigated how mir-122 influences HCV replication in the mir-122-deficient HepG2 cell line. Expression of this microrna in HepG2 cells permitted efficient HCV RNA replication and infectious virion production. When a missing HCV receptor is also expressed, these cells efficiently support viral entry and thus the entire HCV life cycle. Hepatitis C virus (HCV), a member of the family Flaviviridae, is associated with more than half of newly diagnosed hepatocellular carcinomas in the United States (1, 31) and is the leading cause of liver transplants worldwide (7). Historically, a lack of model systems to study HCV replication has slowed the progress of HCV research and the development of specific antivirals. Few HCV genomes replicate in culture, and only one efficiently produces infectious particles (23, 39, 42). Most model systems rely on the Huh-7 cell line and derivatives, such as the highly HCV-permissive Huh-7.5 subclone (6). The development of novel cell systems capable of supporting the entire HCV life cycle would allow broader examination of interactions between HCV and host cell biology and would provide essential tools for anti-hcv drug discovery. MicroRNAs (mirnas) are small RNA molecules predicted to downregulate the expression of one-third of human genes by reducing mrna stability and/or translation, depending on the degree of complementarity between the mirna and the mrna target site (2). mir-122, a liver-specific mirna expressed at high levels in hepatocytes, is required to support HCV RNA replication (19). Two mir-122 target sites have been identified in the 5 untranslated region of the HCV genome, and mutation of these sites disrupts HCV RNA replication. Replication is restored by cotransfection with RNA duplexes that produce a mutant mir-122 with complementary changes (16 19, 26). How mir-122 enhances HCV replication is unclear. Although its binding appears to enhance HCV internal ribosome entry site (IRES)-directed translation, this effect is not great enough to account for the difference in RNA replication (16, 17, 40). Furthermore, the normal cellular function of mir-122 in regulating cholesterol and fatty acid biosynthesis is not required for this enhancement (18). Thus, mir-122 has been hypothesized to directly affect HCV RNA replication or stability (17, 19, 26). Although inhibiting mir- * Corresponding author. Mailing address: One Gustave L. Levy Place, Box 1124, New York, NY Phone: (212) Fax: (212) matthew.evans@mssm.edu. These authors contributed equally. Published ahead of print on 14 September has been shown to be an effective therapy in chimpanzees infected with HCV (21), further development of such therapies will require a firmer understanding of how mir-122 promotes HCV replication. The majority of published studies on the role of mir-122 in the HCV life cycle have described research conducted with HCV-permissive Huh-7 cells or derivatives, which express endogenous mir-122 (19). Two studies explored the effect of mir-122 on HCV in divergent cell types and found that mir- 122 enhances intracellular HCV RNA replication 6- to 11-fold in kidney-derived HEK293 cells (8) and 40-fold in mouse embryonic fibroblasts (22). Neither of these studies tested these systems capacities to support the entire HCV life cycle, including early and late events such as cell entry and infectious virion release. We sought to examine how mir-122 affects the HCV life cycle in the hepatocellular carcinoma-derived HepG2 cell line. This cell line does not express endogenous mir-122 (19) and only weakly supports HCV replication (at a level 850-fold lower than that in Huh-7.5 cells), which is insufficient for efficient viral assembly (12, 23). Establishing efficient HCV replication in HepG2 cells would be advantageous as these cells, in contrast to Huh-7 cells, polarize (reviewed in reference 10) and would thus permit the examination of how cell polarity impacts the HCV life cycle. mir-122 expression enhances HCV RNA replication in HepG2 cells. To express mir-122 in HepG2 cells, we transfected these cells with a plasmid carrying the neomycin resistance gene and the green fluorescent protein (GFP) gene with a beta-globin intron insertion containing the human genomic mir-122 precursor sequence (Fig. 1A) (the sequence is available upon request). As mirnas are usually expressed within noncoding or intronic RNA transcribed by RNA polymerase II (3), this context ensures proper processing of mature mir-122. Approximately HepG2 cells seeded on collagen were transfected with 3 g of this plasmid using TransIT-LT1 transfection reagent (Mirus). Cells were selected in medium (Dulbecco s modified Eagle medium [DMEM] with 10% fetal bovine serum [FBS]) containing 400 g/ml G418 (Invitrogen) for several weeks to generate over 1,000 individual colonies, which were pooled and dilution cloned in 96-well plates. Total RNA 12087
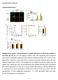
2 12088 NOTES J. VIROL. Downloaded from FIG. 1. mir-122 enhances HCV RNA replication in HepG2 cells. (A) Illustration of the plasmid used to stably express mir-122 in HepG2 cells. Two expression cassettes are shown. The first carries the GFP gene bearing an intron, flanked by splice donor (SD) and splice acceptor (SA) sites, containing the mir-122 genomic locus and precursor sequence. The second carries the neomycin resistance gene (NeoR). HCMV, human cytomegalovirus; SV40, simian virus 40. (B) Northern blots for mir-122, mir-93, and U6. Nucleotide sizes (in bases) are indicated to the left of each blot. (C) Illustration of HCV JFH-1 subgenomic replicon organization. The positions of the GLuc reporter gene, the EMCV IRES, and the HCV nonstructural (NS) protein genes are marked. The locations of the NS5B polymerase active site (GDD) and inactivating mutations (GNN) are also indicated. (D) Quantification of GLuc relative light units (RLU) produced at the indicated hours posttransfection (hpt) by transfection of Huh-7.5 cells, as a positive control, and naïve and mir-122-expressing HepG2 cell clones with either a replication-defective (GNN) or the wild-type (WT) subgenomic HCV replicon. In this and all subsequent luciferase assays, the background value from naïve cell supernatants was RLU (mean standard deviation [SD]). (E) The indicated cell populations were cotransfected with the subgenomic HCV replicon illustrated in panel C and either no RNA (mock), a random-sequence oligonucleotide, or a mir-122-complementary 2 -O-methylated RNA oligonucleotide (mir-122). GLuc activity was assayed at the indicated time points. (F) Diagram of an mirna lentiviral expression construct with an internal cytomegalovirus (CMV) promoter to express transcripts carrying either the blasticidin (BsdR) or puromycin (PuroR) resistance gene followed by the mir-122 genomic locus. LTR, long terminal repeat; Psi, HIV-1 packaging signal; RRE, Rev response element; cppt, central polypurine tract; LTRdelU3, long terminal repeat with a deletion in the U3 region. (G) Naïve HepG2 cells were transduced with lentiviral pseudoparticles bearing the above-mentioned provirus to express either wild-type or mutant (3-4 MT) mir-122. Following drug selection, these cell populations were transfected, in parallel with naïve cells, with GLuc-encoding subgenomic HCV replicon RNA and cells were cultured in either dimethyl sulfoxide (DMSO) or a 6 M concentration, which is 50 times the 50% inhibitory concentration (IC 50 ), of the HCV polymerase inhibitor 2 -C-methyladenosine (2 CMA), to substitute for a GNN control. GLuc activity was assayed at the indicated time points. Results are expressed as means SD (n 3 independent transfections) and are representative of results from multiple experiments. on May 4, 2018 by guest was prepared from expanded single-cell, GFP-positive clones by extraction with TRIzol (Invitrogen). Northern blot analysis using previously described methods (32, 33), with equal quantities of RNA and U6 RNA as a loading control, showed that all clones expressed detectable levels of mir-122 and did not exhibit defects in general mirna processing as determined by blotting for the unrelated mirna mir-93 (Fig. 1B). The capacity of these cells to support HCV RNA replication
3 VOL. 85, 2011 NOTES was tested by transfection with 2 g ofin vitro-transcribed replication-competent (wild-type) and polymerase-defective (GNN) HCV subgenomic replicon RNAs derived from the HCV genotype 2a JFH-1 genome (Fig. 1C) (20) as described previously (5). In these replicons, the HCV IRES directs translation of the Gaussia princeps luciferase protein (GLuc) (37, 38), which was quantified using the Renilla luciferase assay system (Promega) as described previously (30). Although all transfections produced detectable GLuc, the wild-type replicon signal amplified over time, which is indicative of RNA replication (Fig. 1D). Even in naïve HepG2 cells, RNA replication was detectable and statistically significant (compare data for transfections of naïve HepG2 cells with GNN and wild-type replicons at 48 h posttransfection [P ]). At 48 h posttransfection, when replication appears to plateau, mir-122-expressing HepG2 clones supported 50- to 125-fold greater GLuc activity than naïve HepG2 cells (P ). Thus, mir-122 expression enhances HCV subgenomic replicon replication in HepG2 cells. mir-122 levels do not correlate with replication efficiency, suggesting that even low quantities of mir-122 can greatly enhance HCV replication. Notably, the level of GLuc expression from nonreplicating GNN RNAs was higher at all time points in HepG2 cells expressing mir-122 (referred to herein as HepG2 mir-122 cells) than in HepG2 parental cells, suggesting that this mirna enhanced translation from the HCV IRES, HCV RNA stability, or both. The enhancement of HCV RNA replication in HepG2 mir-122 cells was due to expression of this mirna, as cotransfection with a mir-122-specific 2 -O-methylated RNA oligonucleotide antagomir, but not a random-sequence antagomir (19), resulted in a significant reduction in replicon reporter expression (Fig. 1E). This enhancement also required both wild-type mir- 122 and complementary target sites within the HCV genome, as the replication of an HCV replicon with mutant 5 -untranslated-region mir-122 seed sequences, termed p3-4 in a prior publication (19), was not enhanced by wild-type mir-122 and expression of the complementary mutant p3-4 mir-122 sequence did not enhance replication of the wild-type replicon (Fig. 1G). Although expression of the p3-4 mutant mirna did permit replication of the mutant replicon, it was not as efficient as replication with the wild-type pairing. HCV replication in HepG2 cells is limited by transfection efficiency. While mir-122 expression in HepG2 cells enhanced HCV RNA replication, this process was still on average 11-fold less efficient in these cells than in Huh-7.5 cells (Fig. 1D). To examine the frequency of sustained HCV replication, transfected cells were trypsinized and immunostained for HCV NS5A by using the 9E10 mouse monoclonal antibody (23) and a goat anti-mouse Alexa Fluor 647 secondary antibody (Invitrogen). At 72 h posttransfection, 32% of transfected Huh-7.5 cells were NS5A positive, as determined by fluorescence-activated cell sorter (FACS) analysis, while naïve and mir-122- expressing HepG2 cell populations exhibited 1.2 and 6.3% NS5A-positive cells, respectively (Fig. 2A). As the HCV proteins in this context are expressed from the encephalomyocarditis virus (EMCV) IRES rather than the HCV IRES, these results suggest that mir-122 enhances HCV RNA replication and not NS5A translation. To more accurately compare the relative capacities of these cells to support HCV replication, cells were cotransfected with HCV subgenomic replicon RNA and, as a transfection control, an in vitro-transcribed, capped, and polyadenylated mrna encoding the firefly luciferase protein (FLuc) constructed identically to one used in a previous study (17). HCV IRES-based GLuc expression results were similar to those described above (Fig. 2B). FLuc activity, quantified using the luciferase assay system (Promega) as described previously (11), was detected in all samples, with peak expression between 4 and 6 h posttransfection (Fig. 2C). At each time point, naïve and mir-122- expressing HepG2 cells had similar FLuc expression levels and thus have comparable transfection efficiencies, which we calculated as the average ratio of FLuc activities at each time point (Fig. 2D). Huh-7.5 cells were more easily transfected than any of the HepG2 cell populations, as FLuc levels in Huh-7.5 cells were 6.5- to 9-fold higher than those in HepG2 cells (Fig. 2D). After normalizing the GLuc values at each time point for transfection efficiency, there was little difference between the abilities of Huh-7.5 and mir-122-expressing HepG2 cells to support HCV RNA replication (Fig. 2E). To directly compare the efficiencies of HCV IRES-dependent translation in the above-mentioned cell types, the GLuc values from GNN replicon transfections were normalized to the respective time point s FLuc values. Huh-7.5 and naïve HepG2 cells exhibited similar abilities to support HCV IRESdependent translation (Fig. 2F). Furthermore, HCV IRESdependent translation was only 1.4- to 2.1-fold more efficient in mir-122-expressing HepG2 cell clones than in the parental HepG2 cell population. Together, these results support the hypothesis that translation is not greatly impacted by mir-122 expression in HepG2 cells (17). HepG2 mir-122 cells can be engineered to support HCV cell entry. With the use of lentiviral pseudoparticles bearing the HCV envelope glycoproteins (HCVpp), HepG2 cells have been shown previously to support HCV cell entry if the missing receptor, CD81, is overexpressed (4, 12, 41). To examine HCV cell entry in mir-122-expressing HepG2 cells, naïve and mir- 122-expressing HepG2 cells were transduced with a lentivirus to stably express the human CD81 protein, according to previously published protocols (30). Transduction efficiency and CD81 cell surface expression were evaluated by staining of nonpermeabilized cells with an antibody reactive to an extracellular CD81 epitope followed by FACS analysis (Fig. 3A) conducted as described previously (12). To determine their capacity to support HCV cell entry, these cells were challenged with HCVpp and, as a positive control, pseudoparticles bearing the vesicular stomatitis virus envelope glycoprotein (VS- VGpp). As shown previously, naïve HepG2 cells are not susceptible to HCVpp infection, in contrast to Huh-7.5 cells, and this deficiency is eliminated by CD81 expression (Fig. 3B). All mir-122-expressing HepG2 clones were similarly susceptible to infection with HCVpp when CD81 was expressed. To gauge the capacity to support HCVcc infection, the effective titer of a single stock of HCVcc for each cell population was determined by limiting dilution assay, the results of which were quantified by NS5A staining as described previously (23). As shown in Fig. 3D, infection of naïve HepG2 cells, expressing neither CD81 nor mir-122, was below the level of detection of this assay. HepG2 cells expressing CD81 but not mir- 122 were 467-fold less infectible with HCVcc than Huh-7.5 cells. The susceptibility to HCVcc infection was increased an-
4 12090 NOTES J. VIROL. FIG. 3. Testing of HCV cell entry capacities. (A) FACS analysis of CD81 expression on the surfaces of the indicated cells. For each cell type, data for isotype control (gray filled) and CD81 JS-81 antibody (unfilled) staining are shown. The y axis represents the percent maximum. (B) Luciferase production from the cell populations indicated in panel A, as well as Huh-7.5 cells as a positive control, 48 h postinfection (hpi) with lentiviral particles bearing the HCV (white bars) and vesicular stomatitis virus (black bars) envelope proteins (means SD of readings from 4 independent infections). (C) Illustration of the Jc1 chimeric HCV genome. The region comprising the core through the end of the NS2 genes corresponding to the amino terminus (light gray) is from the HC-J6 genome, and the remaining NS genes (dark gray) are from the JFH-1 HCV isolate (34). (D) The effective titer of a single preparation of Jc1 HCVcc was determined on the indicated cell populations. The dotted line indicates the level of detection of the assay. Results shown are means SD of readings from 3 independent infections. TCID 50, 50% tissue culture infective dose. other 22- to 77-fold by mir-122 expression in HepG2 cells transduced with CD81, to within 6- to 20-fold that of naïve Huh-7.5 cells. mir-122 expression in HepG2 cells permits efficient infectious HCV release. To test the ability to support infectious HCV assembly and release, we transfected Huh-7.5, HepG2 CD81, and HepG2 CD81/miR-122-expressing cells with fulllength bicistronic HCV RNAs that express the GLuc protein FIG. 2. Transfection efficiency limits HCV replication in HepG2 cells. (A) Example and quantification from three independent transfections (results shown are means SD) by FACS analysis of intracellular NS5A staining within the indicated cell populations 72 h posttransfection with HCV replicons. (B to F) Huh-7.5 cells, as a positive control, and naïve and mir-122-expressing HepG2 cells were cotransfected with GLuc-expressing wild-type and polymerase-defective (GNN) HCV subgenomic replicons and capped FLuc RNA. (B and C) Assays for secreted GLuc (B) and intracellular FLuc (C) were conducted at the indicated time points posttransfection. Results shown are means SD (n 3 independent transfections) and are representative of results from two independent experiments. (D) FLuc values were used to determine transfection efficiencies for the indicated cell populations, calculated as the average ratio of FLuc activities between cell populations at each time point and presented relative to values for Huh-7.5 cells. (E) Graph of GLuc values normalized for relative transfection efficiency to allow comparison of HCV replication efficiencies independently of transfection efficiency. Symbols are as defined in Fig. 3B. (F) Evaluation of HCV IRES activity in the various cell populations, presented as the ratios of GLuc to FLuc measured from each GNN transfection at the indicated time points and normalized to values determined for naïve HepG2 cells. CAP, Cap-dependent translation.
5 VOL. 85, 2011 NOTES FIG. 4. mir-122 enhances replication and infectious virus release in HepG2 cells. (A) Illustration of the bicistronic GLuc-expressing Jc1 chimeric HCV genome. (B) The indicated cells were transfected with full-length HCVcc reporter genomic RNA. To measure RNA replication, reporter activity produced by transfected cells was determined at the indicated time points posttransfection. (C) Filtered supernatants from these transfections were applied to Huh-7.5 cells to determine relative quantities of infectious virus produced by each transfection at the indicated time points. These infections were assayed 48 hpi. (D) The indicated cells were infected with full-length, nonreporter Jc1 HCVcc RNA in parallel. At the indicated time points, supernatants were collected and the quantity of infectious virus release was determined by a TCID 50 assay on Huh-7.5 cells (gray lines; right y axis). At the same time points, portions of the cell populations were passed into fresh plates and analyzed for the presence of HCV by NS5A FACS analysis (dark lines; left y axis). Results shown are means SD of readings from 3 parallel infections. from the HCV IRES (Fig. 4A). Similar to the results of the above-described subgenomic replicon experiments, mir-122 expression enhanced HCV replication in HepG2 cells. These HCV RNAs exhibited more robust replication in the mir-122- expressing HepG2 cells than in naïve HepG2 cells, as the GLuc levels from mir-122-expressing cells were consistently 10- to 36-fold higher (Fig. 4B). Supernatants from these cultures were collected, filtered, and used to infect Huh-7.5 cells to determine the relative amounts of infectious HCVcc released. All cell populations produced infectious virus, and cell populations expressing mir-122 yielded 30- to 71-fold more infectious virus than HepG2 cells not expressing this mirna (Fig. 4C). Thus, mir-122 expression enhanced the complete HCV life cycle in HepG2 cells. To examine the kinetics of both viral spread and virion release, Huh-7.5 and HepG2 CD81/miR-122 cells were infected with nonreporter HCVcc at a low multiplicity of infection (MOI) and the percentage of NS5A-positive cells and the amount of infectious virus release were measured over time. Although virus spread more efficiently within Huh- 7.5 cell cultures, the percentage of NS5A-positive cells did increase in HepG2 CD81/miR-122 cell cultures, and both populations released infectious virus. In this experiment, HCV infection induced overwhelming Huh-7.5 cell death beginning at 5 days postinfection, which makes it difficult to directly compare the amounts of HCVcc produced from the different cell populations. It is unclear why HCVcc did not spread more efficiently within the HepG2 CD81/miR-122 culture. As the results of the above-described experiments suggest that RNA replication is equally efficient in HepG2 CD81/miR-122 cells and Huh-7.5 cells, it is likely that deficiencies in cell entry, perhaps due to cell polarity, or the presence of a more intact interferon response pathway limits infection within HepG2 cells. In summary, we have demonstrated that HepG2 cells support efficient HCV RNA replication and infectious virion release if mir-122 is expressed. The further addition of the HCV entry factor CD81 renders these cells able to support the entire viral life cycle. Our results support the notion that mir-122 is required for HCV RNA replication but does not greatly enhance viral translation. Although a mechanism for this enhanced RNA replication is still not clear, it may occur through stabilization of the HCV genome by shielding of the viral 5-triphosphate from exonucleases or retinoic acid inducible gene I (RIG-I) surveillance, as recently hypothesized by Machlin et al. (26). Our study does not provide evidence that mir- 122 enhances any other stage of the viral life cycle. Although several non-huh-7-based HCV replication systems have been developed, only Huh-6 cells have been shown to be capable of secreting infectious HCV (14). HepG2 cells offer a distinct advantage over these cell lines, as they can grow in a polarized manner that mimics the bile canalicular configuration of hepatocytes in vivo and thus provide a model system that more closely resembles the natural host cell for HCV (reviewed in reference 10). Two essential HCV cell entry factors, claudin-1 (11) and occludin (25, 35), are tight-junction proteins and are not likely to exhibit completely conserved functions in nonpolarized cells. Thus, a polarized-cell model is particularly important for the study of the HCV cell entry process. HepG2 cells have been used in studies of HCV polarized-cell entry using HCVpp (15, 27, 28); however, the scope of such studies is limited by the low titer of these particles and the likelihood that their entry pathways do not perfectly follow those of native HCV. The ability to infect HepG2 cells with HCVcc, which can be efficiently concentrated and purified (13, 24, 29, 36) and is amenable to fluorescent labeling (9), opens the way to conducting detailed analysis of HCV cell entry pathways in polarized cells.
6 12092 NOTES J. VIROL. We express our gratitude to Peter Sarnow, Kara Norman, and Rohit Jangra for helpful discussions and reagents, Charles Rice for supplying pseudoparticles and HCV plasmids and Huh-7.5 and HepG2 cells, and Timothy Tellinghuisen for the NS5A 9E10 antibody. We also thank Benjamin tenoever, Jillian Shapiro, and Carina Storrs for helpful discussions, technical assistance, and assistance in writing the manuscript. This work was supported by a grant from the National Institutes of Health/National Institute of Allergy and Infectious Diseases (no. R00 AI077800). Benjamin Israelow, Maria L. Michta, and Sharon E. Hopcraft were supported in part by a Public Health Service Institutional Research Training Award (AI07647). Marion Sourisseau was supported in part by the Robin Chemers Neustein Postdoctoral Fellowship award. Matthew Evans is supported in part by the Pew Charitable Funds. REFERENCES 1. Alter, M. J., et al The prevalence of hepatitis C virus infection in the United States, 1988 through N. Engl. J. Med. 341: Ambros, V The functions of animal micrornas. Nature 431: Bartel, D. P MicroRNAs: genomics, biogenesis, mechanism, and function. Cell 116: Bartosch, B., et al Cell entry of hepatitis C virus requires a set of co-receptors that include the CD81 tetraspanin and the SR-B1 scavenger receptor. J. Biol. Chem. 278: Blight, K. J., A. A. Kolykhalov, and C. M. Rice Efficient initiation of HCV RNA replication in cell culture. Science 290: Blight, K. J., J. A. McKeating, and C. M. Rice Highly permissive cell lines for hepatitis C virus genomic and subgenomic RNA replication. J. Virol. 76: Brown, Jr., R. S Hepatitis C and liver transplantation. Nature 436: Chang, J., et al Liver-specific microrna mir-122 enhances the replication of hepatitis C virus in nonhepatic cells. J. Virol. 82: Coller, K. E., et al RNA interference and single particle tracking analysis of hepatitis C virus endocytosis. PLoS Pathog. 5:e Decaens, C., M. Durand, B. Grosse, and D. Cassio Which in vitro models could be best used to study hepatocyte polarity? Biol. Cell 100: Evans, M. J., et al Claudin-1 is a hepatitis C virus co-receptor required for a late step in entry. Nature 446: Flint, M., et al Diverse CD81 proteins support hepatitis C virus infection. J. Virol. 80: Gastaminza, P., et al Ultrastructural and biophysical characterization of hepatitis C virus particles produced in cell culture. J. Virol. 84: Haid, S., M. P. Windisch, R. Bartenschlager, and T. Pietschmann Mouse-specific residues of claudin-1 limit hepatitis C virus genotype 2a infection in a human hepatocyte cell line. J. Virol. 84: Harris, H. J., et al Claudin association with CD81 defines hepatitis C virus entry. J. Biol. Chem. 285: Henke, J. I., et al microrna-122 stimulates translation of hepatitis C virus RNA. EMBO J. 27: Jangra, R. K., M. Yi, and S. M. Lemon Regulation of hepatitis C virus translation and infectious virus production by the microrna mir-122. J. Virol. 84: Jopling, C. L., S. Schutz, and P. Sarnow Position-dependent function for a tandem microrna mir-122-binding site located in the hepatitis C virus RNA genome. Cell Host Microbe 4: Jopling, C. L., M. Yi, A. M. Lancaster, S. M. Lemon, and P. Sarnow Modulation of hepatitis C virus RNA abundance by a liver-specific microrna. Science 309: Kato, T., et al Efficient replication of the genotype 2a hepatitis C virus subgenomic replicon. Gastroenterology 125: Lanford, R. E., et al Therapeutic silencing of microrna-122 in primates with chronic hepatitis C virus infection. Science 327: Lin, L. T., et al Replication of subgenomic hepatitis C virus replicons in mouse fibroblasts is facilitated by deletion of interferon regulatory factor 3 and expression of liver-specific microrna 122. J. Virol. 84: Lindenbach, B. D., et al Complete replication of hepatitis C virus in cell culture. Science 309: Lindenbach, B. D., et al Cell culture-grown hepatitis C virus is infectious in vivo and can be recultured in vitro. Proc. Natl. Acad. Sci. U. S. A. 103: Liu, S., et al Tight junction proteins claudin-1 and occludin control hepatitis C virus entry and are downregulated during infection to prevent superinfection. J. Virol. 83: Machlin, E. S., P. Sarnow, and S. M. Sagan Masking the 5 terminal nucleotides of the hepatitis C virus genome by an unconventional microrna-target RNA complex. Proc. Natl. Acad. Sci. U. S. A. 108: Mee, C. J., et al Hepatitis C virus infection reduces hepatocellular polarity in a vascular endothelial growth factor-dependent manner. Gastroenterology 138: Mee, C. J., et al Polarization restricts hepatitis C virus entry into HepG2 hepatoma cells. J. Virol. 83: Merz, A., et al Biochemical and morphological properties of hepatitis C virus particles and determination of their lipidome. J. Biol. Chem. 286: Michta, M. L., et al Species-specific regions of occludin required by hepatitis C virus for cell entry. J. Virol. 84: Montalto, G., et al Epidemiology, risk factors, and natural history of hepatocellular carcinoma. Ann. N. Y. Acad. Sci. 963: Pall, G. S., and A. J. Hamilton Improved northern blot method for enhanced detection of small RNA. Nat. Protoc. 3: Perez, J. T., et al Influenza A virus-generated small RNAs regulate the switch from transcription to replication. Proc. Natl. Acad. Sci. U. S. A. 107: Pietschmann, T., et al Construction and characterization of infectious intragenotypic and intergenotypic hepatitis C virus chimeras. Proc. Natl. Acad. Sci. U. S. A. 103: Ploss, A., et al Human occludin is a hepatitis C virus entry factor required for infection of mouse cells. Nature 457: Prentoe, J., and J. Bukh Hepatitis C virus expressing flag-tagged envelope protein 2 has unaltered infectivity and density, is specifically neutralized by flag antibodies and can be purified by affinity chromatography. Virology 409: Tannous, B. A., D. E. Kim, J. L. Fernandez, R. Weissleder, and X. O. Breakefield Codon-optimized Gaussia luciferase cdna for mammalian gene expression in culture and in vivo. Mol. Ther. 11: Verhaegent, M., and T. K. Christopoulos Recombinant Gaussia luciferase. Overexpression, purification, and analytical application of a bioluminescent reporter for DNA hybridization. Anal. Chem. 74: Wakita, T., et al Production of infectious hepatitis C virus in tissue culture from a cloned viral genome. Nat. Med. 11: Wilson, J. A., C. Zhang, A. Huys, and C. D. Richardson Human Ago2 is required for efficient microrna 122 regulation of hepatitis C virus RNA accumulation and translation. J. Virol. 85: Zhang, J., et al CD81 is required for hepatitis C virus glycoproteinmediated viral infection. J. Virol. 78: Zhong, J., et al Robust hepatitis C virus infection in vitro. Proc. Natl. Acad. Sci. U. S. A. 102:
~Lentivirus production~
 ~Lentivirus production~ May 30, 2008 RNAi core R&D group member Lentivirus Production Session Lentivirus!!! Is it health threatening to lab technician? What s so good about this RNAi library? How to produce
~Lentivirus production~ May 30, 2008 RNAi core R&D group member Lentivirus Production Session Lentivirus!!! Is it health threatening to lab technician? What s so good about this RNAi library? How to produce
Soft Agar Assay. For each cell pool, 100,000 cells were resuspended in 0.35% (w/v)
 SUPPLEMENTARY MATERIAL AND METHODS Soft Agar Assay. For each cell pool, 100,000 cells were resuspended in 0.35% (w/v) top agar (LONZA, SeaKem LE Agarose cat.5004) and plated onto 0.5% (w/v) basal agar.
SUPPLEMENTARY MATERIAL AND METHODS Soft Agar Assay. For each cell pool, 100,000 cells were resuspended in 0.35% (w/v) top agar (LONZA, SeaKem LE Agarose cat.5004) and plated onto 0.5% (w/v) basal agar.
Immunogenic and Functional Organization of Hepatitis C Virus (HCV) Glycoprotein E2 on Infectious HCV Virions
 JOURNAL OF VIROLOGY, Jan. 2007, p. 1043 1047 Vol. 81, No. 2 0022-538X/07/$08.00 0 doi:10.1128/jvi.01710-06 Copyright 2007, American Society for Microbiology. All Rights Reserved. Immunogenic and Functional
JOURNAL OF VIROLOGY, Jan. 2007, p. 1043 1047 Vol. 81, No. 2 0022-538X/07/$08.00 0 doi:10.1128/jvi.01710-06 Copyright 2007, American Society for Microbiology. All Rights Reserved. Immunogenic and Functional
DNA context and promoter activity affect gene expression in lentiviral vectors
 ACTA BIOMED 2008; 79: 192-196 Mattioli 1885 O R I G I N A L A R T I C L E DNA context and promoter activity affect gene expression in lentiviral vectors Gensheng Mao 1, Francesco Marotta 2, Jia Yu 3, Liang
ACTA BIOMED 2008; 79: 192-196 Mattioli 1885 O R I G I N A L A R T I C L E DNA context and promoter activity affect gene expression in lentiviral vectors Gensheng Mao 1, Francesco Marotta 2, Jia Yu 3, Liang
Permissivity of primary hepatocytes and hepatoma cell lines to support hepatitis C virus infection
 Journal of General Virology (215), 96, 1369 1373 DOI 1.199/vir..85 Short Communication Correspondence Jane A. McKeating j.a.mckeating@bham.ac.uk Permissivity of primary hepatocytes and hepatoma cell lines
Journal of General Virology (215), 96, 1369 1373 DOI 1.199/vir..85 Short Communication Correspondence Jane A. McKeating j.a.mckeating@bham.ac.uk Permissivity of primary hepatocytes and hepatoma cell lines
Viral Vectors In The Research Laboratory: Just How Safe Are They? Dawn P. Wooley, Ph.D., SM(NRM), RBP, CBSP
 Viral Vectors In The Research Laboratory: Just How Safe Are They? Dawn P. Wooley, Ph.D., SM(NRM), RBP, CBSP 1 Learning Objectives Recognize hazards associated with viral vectors in research and animal
Viral Vectors In The Research Laboratory: Just How Safe Are They? Dawn P. Wooley, Ph.D., SM(NRM), RBP, CBSP 1 Learning Objectives Recognize hazards associated with viral vectors in research and animal
Certificate of Analysis
 Certificate of Analysis Catalog No. Amount Lot Number 631987 10 μg Specified on product label. Product Information plvx-ef1α-ires-mcherry is a bicistronic lentiviral expression vector that can be used
Certificate of Analysis Catalog No. Amount Lot Number 631987 10 μg Specified on product label. Product Information plvx-ef1α-ires-mcherry is a bicistronic lentiviral expression vector that can be used
Supplementary information
 Supplementary information Human Cytomegalovirus MicroRNA mir-us4-1 Inhibits CD8 + T Cell Response by Targeting ERAP1 Sungchul Kim, Sanghyun Lee, Jinwook Shin, Youngkyun Kim, Irini Evnouchidou, Donghyun
Supplementary information Human Cytomegalovirus MicroRNA mir-us4-1 Inhibits CD8 + T Cell Response by Targeting ERAP1 Sungchul Kim, Sanghyun Lee, Jinwook Shin, Youngkyun Kim, Irini Evnouchidou, Donghyun
Hepatitis C in Australia:
 Hepatitis C in Australia: Epidemiology and Clinical Presentation (and a bit of virology ) A/Prof Mark Douglas Hepatitis C - Distribution Te and Jensen 2010 Clin Liver Dis Hepatitis C Epidemiology Estimated
Hepatitis C in Australia: Epidemiology and Clinical Presentation (and a bit of virology ) A/Prof Mark Douglas Hepatitis C - Distribution Te and Jensen 2010 Clin Liver Dis Hepatitis C Epidemiology Estimated
Supplementary Figure 1. SC35M polymerase activity in the presence of Bat or SC35M NP encoded from the phw2000 rescue plasmid.
 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 Supplementary Figure 1. SC35M polymerase activity in the presence of Bat or SC35M NP encoded from the phw2000 rescue plasmid. HEK293T
1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 Supplementary Figure 1. SC35M polymerase activity in the presence of Bat or SC35M NP encoded from the phw2000 rescue plasmid. HEK293T
sirna a powerful tool to unravel hepatitis C virus-host interactions within the infectious life cycle
 Author manuscript, published in "Journal of Hepatology 2008;48(3):523-5" DOI : 10.1016/j.jhep.2007.12.007 Journal of Hepatology: Journal Club sirna a powerful tool to unravel hepatitis C virus-host interactions
Author manuscript, published in "Journal of Hepatology 2008;48(3):523-5" DOI : 10.1016/j.jhep.2007.12.007 Journal of Hepatology: Journal Club sirna a powerful tool to unravel hepatitis C virus-host interactions
Supplementary Information. Novel lentiviral vectors with mutated reverse transcriptase for mrna delivery of TALE nucleases
 Supplementary Information Novel lentiviral vectors with mutated reverse transcriptase for mrna delivery of TALE nucleases Ulrike Mock 1, Kristoffer Riecken 1, Belinda Berdien 1, Waseem Qasim 2, Emma Chan
Supplementary Information Novel lentiviral vectors with mutated reverse transcriptase for mrna delivery of TALE nucleases Ulrike Mock 1, Kristoffer Riecken 1, Belinda Berdien 1, Waseem Qasim 2, Emma Chan
Hepatitis C virus (HCV) is a highly variable, Isolate-Dependent Use of Claudins for Cell Entry by Hepatitis C Virus VIRAL HEPATITIS
 VIRAL HEPATITIS Isolate-Dependent Use of Claudins for Cell Entry by Hepatitis C Virus Sibylle Haid, 1 Christina Grethe, 1 Michael T. Dill, 2 Markus Heim, 2 Lars Kaderali, 3 and Thomas Pietschmann 1 Hepatitis
VIRAL HEPATITIS Isolate-Dependent Use of Claudins for Cell Entry by Hepatitis C Virus Sibylle Haid, 1 Christina Grethe, 1 Michael T. Dill, 2 Markus Heim, 2 Lars Kaderali, 3 and Thomas Pietschmann 1 Hepatitis
VIROLOGY. Engineering Viral Genomes: Retrovirus Vectors
 VIROLOGY Engineering Viral Genomes: Retrovirus Vectors Viral vectors Retrovirus replicative cycle Most mammalian retroviruses use trna PRO, trna Lys3, trna Lys1,2 The partially unfolded trna is annealed
VIROLOGY Engineering Viral Genomes: Retrovirus Vectors Viral vectors Retrovirus replicative cycle Most mammalian retroviruses use trna PRO, trna Lys3, trna Lys1,2 The partially unfolded trna is annealed
Hepatitis C virus (HCV) infection is a major public health burden
 Identification of Alpha Interferon-Induced Envelope Mutations of Hepatitis C Virus In Vitro Associated with Increased Viral Fitness and Interferon Resistance Stéphanie B. N. Serre, a Henrik B. Krarup,
Identification of Alpha Interferon-Induced Envelope Mutations of Hepatitis C Virus In Vitro Associated with Increased Viral Fitness and Interferon Resistance Stéphanie B. N. Serre, a Henrik B. Krarup,
Supplementary Information. Supplementary Figure 1
 Supplementary Information Supplementary Figure 1 1 Supplementary Figure 1. Functional assay of the hcas9-2a-mcherry construct (a) Gene correction of a mutant EGFP reporter cell line mediated by hcas9 or
Supplementary Information Supplementary Figure 1 1 Supplementary Figure 1. Functional assay of the hcas9-2a-mcherry construct (a) Gene correction of a mutant EGFP reporter cell line mediated by hcas9 or
Introduction retroposon
 17.1 - Introduction A retrovirus is an RNA virus able to convert its sequence into DNA by reverse transcription A retroposon (retrotransposon) is a transposon that mobilizes via an RNA form; the DNA element
17.1 - Introduction A retrovirus is an RNA virus able to convert its sequence into DNA by reverse transcription A retroposon (retrotransposon) is a transposon that mobilizes via an RNA form; the DNA element
Dynamic Interaction of Stress Granule, DDX3X and IKK-α Mediates Multiple Functions in
 Dynamic Interaction of Stress Granule, and Mediates Multiple Functions in Hepatitis C Virus Infection Véronique Pène, Qisheng Li#, Catherine Sodroski, Ching-Sheng Hsu, T. Jake Liang# Liver Diseases Branch,
Dynamic Interaction of Stress Granule, and Mediates Multiple Functions in Hepatitis C Virus Infection Véronique Pène, Qisheng Li#, Catherine Sodroski, Ching-Sheng Hsu, T. Jake Liang# Liver Diseases Branch,
MicroRNA sponges: competitive inhibitors of small RNAs in mammalian cells
 MicroRNA sponges: competitive inhibitors of small RNAs in mammalian cells Margaret S Ebert, Joel R Neilson & Phillip A Sharp Supplementary figures and text: Supplementary Figure 1. Effect of sponges on
MicroRNA sponges: competitive inhibitors of small RNAs in mammalian cells Margaret S Ebert, Joel R Neilson & Phillip A Sharp Supplementary figures and text: Supplementary Figure 1. Effect of sponges on
Supplemental Materials and Methods Plasmids and viruses Quantitative Reverse Transcription PCR Generation of molecular standard for quantitative PCR
 Supplemental Materials and Methods Plasmids and viruses To generate pseudotyped viruses, the previously described recombinant plasmids pnl4-3-δnef-gfp or pnl4-3-δ6-drgfp and a vector expressing HIV-1 X4
Supplemental Materials and Methods Plasmids and viruses To generate pseudotyped viruses, the previously described recombinant plasmids pnl4-3-δnef-gfp or pnl4-3-δ6-drgfp and a vector expressing HIV-1 X4
Domain III of NS5A contributes to both RNA replication and assembly of hepatitis C virus particles
 Journal of General Virology (2009), 90, 1329 1334 DOI 10.1099/vir.0.009332-0 Short Communication Domain III of NS5A contributes to both RNA replication and assembly of hepatitis C virus particles Mair
Journal of General Virology (2009), 90, 1329 1334 DOI 10.1099/vir.0.009332-0 Short Communication Domain III of NS5A contributes to both RNA replication and assembly of hepatitis C virus particles Mair
This is a pre- or post-print of an article published in Steinmann, E., Pietschmann, T. Cell culture systems for hepatitis C virus (2013) Current
 This is a pre- or post-print of an article published in Steinmann, E., Pietschmann, T. Cell culture systems for hepatitis C virus (2013) Current Topics in Microbiology and Immunology, 369, pp. 17-48. Chapter
This is a pre- or post-print of an article published in Steinmann, E., Pietschmann, T. Cell culture systems for hepatitis C virus (2013) Current Topics in Microbiology and Immunology, 369, pp. 17-48. Chapter
Identification of IFN-alpha Induced Envelope Mutations of Hepatitis C Virus In Vitro Associated
 JVI Accepts, published online ahead of print on 18 September 2013 J. Virol. doi:10.1128/jvi.00901-13 Copyright 2013, American Society for Microbiology. All Rights Reserved. 1 2 Serre et al., September
JVI Accepts, published online ahead of print on 18 September 2013 J. Virol. doi:10.1128/jvi.00901-13 Copyright 2013, American Society for Microbiology. All Rights Reserved. 1 2 Serre et al., September
7.012 Quiz 3 Answers
 MIT Biology Department 7.012: Introductory Biology - Fall 2004 Instructors: Professor Eric Lander, Professor Robert A. Weinberg, Dr. Claudette Gardel Friday 11/12/04 7.012 Quiz 3 Answers A > 85 B 72-84
MIT Biology Department 7.012: Introductory Biology - Fall 2004 Instructors: Professor Eric Lander, Professor Robert A. Weinberg, Dr. Claudette Gardel Friday 11/12/04 7.012 Quiz 3 Answers A > 85 B 72-84
Certificate of Analysis
 Certificate of Analysis plvx-ef1α-ires-puro Vector Table of Contents Product Information... 1 Description... 2 Location of Features... 3 Additional Information... 3 Quality Control Data... 4 Catalog No.
Certificate of Analysis plvx-ef1α-ires-puro Vector Table of Contents Product Information... 1 Description... 2 Location of Features... 3 Additional Information... 3 Quality Control Data... 4 Catalog No.
Received 5 November 2010/Accepted 6 December 2010
 JOURNAL OF VIROLOGY, Mar. 2011, p. 2138 2147 Vol. 85, No. 5 0022-538X/11/$12.00 doi:10.1128/jvi.02313-10 Copyright 2011, American Society for Microbiology. All Rights Reserved. Production of Hepatitis
JOURNAL OF VIROLOGY, Mar. 2011, p. 2138 2147 Vol. 85, No. 5 0022-538X/11/$12.00 doi:10.1128/jvi.02313-10 Copyright 2011, American Society for Microbiology. All Rights Reserved. Production of Hepatitis
Supplementary information. MARCH8 inhibits HIV-1 infection by reducing virion incorporation of envelope glycoproteins
 Supplementary information inhibits HIV-1 infection by reducing virion incorporation of envelope glycoproteins Takuya Tada, Yanzhao Zhang, Takayoshi Koyama, Minoru Tobiume, Yasuko Tsunetsugu-Yokota, Shoji
Supplementary information inhibits HIV-1 infection by reducing virion incorporation of envelope glycoproteins Takuya Tada, Yanzhao Zhang, Takayoshi Koyama, Minoru Tobiume, Yasuko Tsunetsugu-Yokota, Shoji
Regulation of Infectious Genotype 1a Hepatitis C Virus Production. by Domain III of NS5A
 JVI Accepts, published online ahead of print on April 0 J. Virol. doi:0./jvi.0-0 Copyright 0, American Society for Microbiology and/or the Listed Authors/Institutions. All Rights Reserved. JVI0-0 Version
JVI Accepts, published online ahead of print on April 0 J. Virol. doi:0./jvi.0-0 Copyright 0, American Society for Microbiology and/or the Listed Authors/Institutions. All Rights Reserved. JVI0-0 Version
Essential Role of Cyclophilin A for Hepatitis C Virus Replication and Virus Production and Possible Link to Polyprotein Cleavage Kinetics
 Essential Role of Cyclophilin A for Hepatitis C Virus Replication and Virus Production and Possible Link to Polyprotein Cleavage Kinetics Artur Kaul 1, Sarah Stauffer 1., Carola Berger 1., Thomas Pertel
Essential Role of Cyclophilin A for Hepatitis C Virus Replication and Virus Production and Possible Link to Polyprotein Cleavage Kinetics Artur Kaul 1, Sarah Stauffer 1., Carola Berger 1., Thomas Pertel
Translation. Host Cell Shutoff 1) Initiation of eukaryotic translation involves many initiation factors
 Translation Questions? 1) How does poliovirus shutoff eukaryotic translation? 2) If eukaryotic messages are not translated how can poliovirus get its message translated? Host Cell Shutoff 1) Initiation
Translation Questions? 1) How does poliovirus shutoff eukaryotic translation? 2) If eukaryotic messages are not translated how can poliovirus get its message translated? Host Cell Shutoff 1) Initiation
Essential Role of Domain III of Nonstructural Protein 5A for Hepatitis C Virus Infectious Particle Assembly
 Essential Role of Domain III of Nonstructural Protein 5A for Hepatitis C Virus Infectious Particle Assembly Nicole Appel. a, Margarita Zayas., Sven Miller b, Jacomine Krijnse-Locker a, Torsten Schaller
Essential Role of Domain III of Nonstructural Protein 5A for Hepatitis C Virus Infectious Particle Assembly Nicole Appel. a, Margarita Zayas., Sven Miller b, Jacomine Krijnse-Locker a, Torsten Schaller
Bi 8 Lecture 17. interference. Ellen Rothenberg 1 March 2016
 Bi 8 Lecture 17 REGulation by RNA interference Ellen Rothenberg 1 March 2016 Protein is not the only regulatory molecule affecting gene expression: RNA itself can be negative regulator RNA does not need
Bi 8 Lecture 17 REGulation by RNA interference Ellen Rothenberg 1 March 2016 Protein is not the only regulatory molecule affecting gene expression: RNA itself can be negative regulator RNA does not need
Hepatitis B Antiviral Drug Development Multi-Marker Screening Assay
 Hepatitis B Antiviral Drug Development Multi-Marker Screening Assay Background ImQuest BioSciences has developed and qualified a single-plate method to expedite the screening of antiviral agents against
Hepatitis B Antiviral Drug Development Multi-Marker Screening Assay Background ImQuest BioSciences has developed and qualified a single-plate method to expedite the screening of antiviral agents against
Pre-made Lentiviral Particles for Fluorescent Proteins
 Pre-made Lentiviral Particles for Fluorescent Proteins Catalog# Product Name Amounts Fluorescent proteins expressed under sucmv promoter: LVP001 LVP001-PBS LVP002 LVP002-PBS LVP011 LVP011-PBS LVP012 LVP012-PBS
Pre-made Lentiviral Particles for Fluorescent Proteins Catalog# Product Name Amounts Fluorescent proteins expressed under sucmv promoter: LVP001 LVP001-PBS LVP002 LVP002-PBS LVP011 LVP011-PBS LVP012 LVP012-PBS
Nature Medicine: doi: /nm.4322
 1 2 3 4 5 6 7 8 9 10 11 Supplementary Figure 1. Predicted RNA structure of 3 UTR and sequence alignment of deleted nucleotides. (a) Predicted RNA secondary structure of ZIKV 3 UTR. The stem-loop structure
1 2 3 4 5 6 7 8 9 10 11 Supplementary Figure 1. Predicted RNA structure of 3 UTR and sequence alignment of deleted nucleotides. (a) Predicted RNA secondary structure of ZIKV 3 UTR. The stem-loop structure
Production of Infectious Hepatitis C Virus of Various Genotypes in Cell Cultures
 JOURNAL OF VIROLOGY, May 2007, p. 4405 4411 Vol. 81, No. 9 0022-538X/07/$08.00 0 doi:10.1128/jvi.02334-06 Copyright 2007, American Society for Microbiology. All Rights Reserved. Production of Infectious
JOURNAL OF VIROLOGY, May 2007, p. 4405 4411 Vol. 81, No. 9 0022-538X/07/$08.00 0 doi:10.1128/jvi.02334-06 Copyright 2007, American Society for Microbiology. All Rights Reserved. Production of Infectious
Circular RNAs (circrnas) act a stable mirna sponges
 Circular RNAs (circrnas) act a stable mirna sponges cernas compete for mirnas Ancestal mrna (+3 UTR) Pseudogene RNA (+3 UTR homolgy region) The model holds true for all RNAs that share a mirna binding
Circular RNAs (circrnas) act a stable mirna sponges cernas compete for mirnas Ancestal mrna (+3 UTR) Pseudogene RNA (+3 UTR homolgy region) The model holds true for all RNAs that share a mirna binding
Determinants of the Hepatitis C Virus Nonstructural Protein 2 Protease Domain Required for Production of Infectious Virus
 JOURNAL OF VIROLOGY, Dec. 2009, p. 12702 12713 Vol. 83, No. 24 0022-538X/09/$12.00 doi:10.1128/jvi.01184-09 Copyright 2009, American Society for Microbiology. All Rights Reserved. Determinants of the Hepatitis
JOURNAL OF VIROLOGY, Dec. 2009, p. 12702 12713 Vol. 83, No. 24 0022-538X/09/$12.00 doi:10.1128/jvi.01184-09 Copyright 2009, American Society for Microbiology. All Rights Reserved. Determinants of the Hepatitis
Identification of Mutation(s) in. Associated with Neutralization Resistance. Miah Blomquist
 Identification of Mutation(s) in the HIV 1 gp41 Subunit Associated with Neutralization Resistance Miah Blomquist What is HIV 1? HIV-1 is an epidemic that affects over 34 million people worldwide. HIV-1
Identification of Mutation(s) in the HIV 1 gp41 Subunit Associated with Neutralization Resistance Miah Blomquist What is HIV 1? HIV-1 is an epidemic that affects over 34 million people worldwide. HIV-1
Ready-to-use Lentiviral Particles for intracelular labeling
 Ready-to-use Lentiviral Particles for intracelular labeling (LocLight TM Living cell imaging lentivirus for sub-cellular localization) LocLight TM cell organelle labeling lentivirus are provided as 200ul/per
Ready-to-use Lentiviral Particles for intracelular labeling (LocLight TM Living cell imaging lentivirus for sub-cellular localization) LocLight TM cell organelle labeling lentivirus are provided as 200ul/per
MicroRNA and Male Infertility: A Potential for Diagnosis
 Review Article MicroRNA and Male Infertility: A Potential for Diagnosis * Abstract MicroRNAs (mirnas) are small non-coding single stranded RNA molecules that are physiologically produced in eukaryotic
Review Article MicroRNA and Male Infertility: A Potential for Diagnosis * Abstract MicroRNAs (mirnas) are small non-coding single stranded RNA molecules that are physiologically produced in eukaryotic
Polyomaviridae. Spring
 Polyomaviridae Spring 2002 331 Antibody Prevalence for BK & JC Viruses Spring 2002 332 Polyoma Viruses General characteristics Papovaviridae: PA - papilloma; PO - polyoma; VA - vacuolating agent a. 45nm
Polyomaviridae Spring 2002 331 Antibody Prevalence for BK & JC Viruses Spring 2002 332 Polyoma Viruses General characteristics Papovaviridae: PA - papilloma; PO - polyoma; VA - vacuolating agent a. 45nm
Recombinant Protein Expression Retroviral system
 Recombinant Protein Expression Retroviral system Viruses Contains genome DNA or RNA Genome encased in a protein coat or capsid. Some viruses have membrane covering protein coat enveloped virus Ø Essential
Recombinant Protein Expression Retroviral system Viruses Contains genome DNA or RNA Genome encased in a protein coat or capsid. Some viruses have membrane covering protein coat enveloped virus Ø Essential
Jumpstart your research with ViraPower Lentiviral Expression Systems
 ViraPower Lentiviral Expression Systems Jumpstart your research with ViraPower Lentiviral Expression Systems With ViraPower Lentiviral Systems you can: Efficiently transduce both dividing and non-dividing
ViraPower Lentiviral Expression Systems Jumpstart your research with ViraPower Lentiviral Expression Systems With ViraPower Lentiviral Systems you can: Efficiently transduce both dividing and non-dividing
Globally, an estimated 160 million people are
 Incorporation of Primary Patient-Derived Glycoproteins Into Authentic Infectious Hepatitis C Virus Particles Juliane Doerrbecker, 1 Martina Friesland, 1 Nina Riebesehl, 1 Corinne Ginkel, 1 Patrick Behrendt,
Incorporation of Primary Patient-Derived Glycoproteins Into Authentic Infectious Hepatitis C Virus Particles Juliane Doerrbecker, 1 Martina Friesland, 1 Nina Riebesehl, 1 Corinne Ginkel, 1 Patrick Behrendt,
NOTES. Nonhepatic Cell Lines HeLa and 293 Support Efficient Replication of the Hepatitis C Virus Genotype 2a Subgenomic Replicon
 JOURNAL OF VIROLOGY, Jan. 2005, p. 592 596 Vol. 79, No. 1 0022-538X/05/$08.00 0 doi:10.1128/jvi.79.1.592 596.2005 Copyright 2005, American Society for Microbiology. All Rights Reserved. NOTES Nonhepatic
JOURNAL OF VIROLOGY, Jan. 2005, p. 592 596 Vol. 79, No. 1 0022-538X/05/$08.00 0 doi:10.1128/jvi.79.1.592 596.2005 Copyright 2005, American Society for Microbiology. All Rights Reserved. NOTES Nonhepatic
Animal Models for the Study of Hepatitis C Virus Infection and Related Liver Disease
 GASTROENTEROLOGY 2012;142:1279 1287 Animal Models for the Study of Hepatitis C Virus Infection and Related Liver Disease Jens Bukh Copenhagen Hepatitis C Program (CO-HEP), Department of Infectious Diseases
GASTROENTEROLOGY 2012;142:1279 1287 Animal Models for the Study of Hepatitis C Virus Infection and Related Liver Disease Jens Bukh Copenhagen Hepatitis C Program (CO-HEP), Department of Infectious Diseases
CRISPR/Cas9 cleavage of viral DNA efficiently suppresses hepatitis B virus
 0 0 CRISPR/Cas cleavage of viral DNA efficiently suppresses hepatitis B virus Authors: Vyas Ramanan,, Amir Shlomai,, David B.T. Cox,,,, Robert E. Schwartz,,, Eleftherios Michailidis, Ankit Bhatta, David
0 0 CRISPR/Cas cleavage of viral DNA efficiently suppresses hepatitis B virus Authors: Vyas Ramanan,, Amir Shlomai,, David B.T. Cox,,,, Robert E. Schwartz,,, Eleftherios Michailidis, Ankit Bhatta, David
Correspondence should be addressed to Shilin Li; and Limin Chen; limin chen
 Mediators of Inflammation Volume 25, Article ID 58989, 5 pages http://dx.doi.org/.55/25/58989 Research Article Vitamin D Potentiates the Inhibitory Effect of MicroRNA-3a in Hepatitis C Virus Replication
Mediators of Inflammation Volume 25, Article ID 58989, 5 pages http://dx.doi.org/.55/25/58989 Research Article Vitamin D Potentiates the Inhibitory Effect of MicroRNA-3a in Hepatitis C Virus Replication
Inhibition of HIV-1 Integration in Ex Vivo-Infected CD4 T Cells from Elite Controllers
 JOURNAL OF VIROLOGY, Sept. 2011, p. 9646 9650 Vol. 85, No. 18 0022-538X/11/$12.00 doi:10.1128/jvi.05327-11 Copyright 2011, American Society for Microbiology. All Rights Reserved. Inhibition of HIV-1 Integration
JOURNAL OF VIROLOGY, Sept. 2011, p. 9646 9650 Vol. 85, No. 18 0022-538X/11/$12.00 doi:10.1128/jvi.05327-11 Copyright 2011, American Society for Microbiology. All Rights Reserved. Inhibition of HIV-1 Integration
Supplementary Materials for
 www.sciencesignaling.org/cgi/content/full/8/406/ra126/dc1 Supplementary Materials for The microrna mir-485 targets host and influenza virus transcripts to regulate antiviral immunity and restrict viral
www.sciencesignaling.org/cgi/content/full/8/406/ra126/dc1 Supplementary Materials for The microrna mir-485 targets host and influenza virus transcripts to regulate antiviral immunity and restrict viral
Plasmid-Driven Formation of Influenza Virus-Like Particles
 JOURNAL OF VIROLOGY, Jan. 2000, p. 547 551 Vol. 74, No. 1 0022-538X/00/$04.00 0 Copyright 2000, American Society for Microbiology. All Rights Reserved. Plasmid-Driven Formation of Influenza Virus-Like
JOURNAL OF VIROLOGY, Jan. 2000, p. 547 551 Vol. 74, No. 1 0022-538X/00/$04.00 0 Copyright 2000, American Society for Microbiology. All Rights Reserved. Plasmid-Driven Formation of Influenza Virus-Like
Materials and Methods , The two-hybrid principle.
 The enzymatic activity of an unknown protein which cleaves the phosphodiester bond between the tyrosine residue of a viral protein and the 5 terminus of the picornavirus RNA Introduction Every day there
The enzymatic activity of an unknown protein which cleaves the phosphodiester bond between the tyrosine residue of a viral protein and the 5 terminus of the picornavirus RNA Introduction Every day there
Tel: ; Fax: ;
 Tel.: +98 216 696 9291; Fax: +98 216 696 9291; E-mail: mrasadeghi@pasteur.ac.ir Tel: +98 916 113 7679; Fax: +98 613 333 6380; E-mail: abakhshi_e@ajums.ac.ir A Soluble Chromatin-bound MOI 0 1 5 0 1 5 HDAC2
Tel.: +98 216 696 9291; Fax: +98 216 696 9291; E-mail: mrasadeghi@pasteur.ac.ir Tel: +98 916 113 7679; Fax: +98 613 333 6380; E-mail: abakhshi_e@ajums.ac.ir A Soluble Chromatin-bound MOI 0 1 5 0 1 5 HDAC2
Doctoral Degree Program in Marine Biotechnology, College of Marine Sciences, Doctoral Degree Program in Marine Biotechnology, Academia Sinica, Taipei,
 Cyclooxygenase 2 facilitates dengue virus replication and serves as a potential target for developing antiviral agents Chun-Kuang Lin 1,2, Chin-Kai Tseng 3,4, Yu-Hsuan Wu 3,4, Chih-Chuang Liaw 1,5, Chun-
Cyclooxygenase 2 facilitates dengue virus replication and serves as a potential target for developing antiviral agents Chun-Kuang Lin 1,2, Chin-Kai Tseng 3,4, Yu-Hsuan Wu 3,4, Chih-Chuang Liaw 1,5, Chun-
Supplementary Figures
 Supplementary Figures Supplementary Figure 1 Characterization of stable expression of GlucB and sshbira in the CT26 cell line (a) Live cell imaging of stable CT26 cells expressing green fluorescent protein
Supplementary Figures Supplementary Figure 1 Characterization of stable expression of GlucB and sshbira in the CT26 cell line (a) Live cell imaging of stable CT26 cells expressing green fluorescent protein
Supplemental Figure S1. Expression of Cirbp mrna in mouse tissues and NIH3T3 cells.
 SUPPLEMENTAL FIGURE AND TABLE LEGENDS Supplemental Figure S1. Expression of Cirbp mrna in mouse tissues and NIH3T3 cells. A) Cirbp mrna expression levels in various mouse tissues collected around the clock
SUPPLEMENTAL FIGURE AND TABLE LEGENDS Supplemental Figure S1. Expression of Cirbp mrna in mouse tissues and NIH3T3 cells. A) Cirbp mrna expression levels in various mouse tissues collected around the clock
7.012 Problem Set 6 Solutions
 Name Section 7.012 Problem Set 6 Solutions Question 1 The viral family Orthomyxoviridae contains the influenza A, B and C viruses. These viruses have a (-)ss RNA genome surrounded by a capsid composed
Name Section 7.012 Problem Set 6 Solutions Question 1 The viral family Orthomyxoviridae contains the influenza A, B and C viruses. These viruses have a (-)ss RNA genome surrounded by a capsid composed
Effects of Mutations of the Initiation Nucleotides on Hepatitis C Virus RNA Replication in the Cell
 JOURNAL OF VIROLOGY, Apr. 2004, p. 3633 3643 Vol. 78, No. 7 0022-538X/04/$08.00 0 DOI: 10.1128/JVI.78.7.3633 3643.2004 Copyright 2004, American Society for Microbiology. All Rights Reserved. Effects of
JOURNAL OF VIROLOGY, Apr. 2004, p. 3633 3643 Vol. 78, No. 7 0022-538X/04/$08.00 0 DOI: 10.1128/JVI.78.7.3633 3643.2004 Copyright 2004, American Society for Microbiology. All Rights Reserved. Effects of
Prentoe et al., November 1, Hypervariable Region 1 Deletion and Required Adaptive Envelope Mutations Confer Decreased
 JVI Accepts, published online ahead of print on 20 November 2013 J. Virol. doi:10.1128/jvi.02017-13 Copyright 2013, American Society for Microbiology. All Rights Reserved. 1 Prentoe et al., November 1,
JVI Accepts, published online ahead of print on 20 November 2013 J. Virol. doi:10.1128/jvi.02017-13 Copyright 2013, American Society for Microbiology. All Rights Reserved. 1 Prentoe et al., November 1,
Introduction to the Impact of Resistance in Hepatitis C
 Introduction to the Impact of Resistance in Hepatitis C Sponsored by AbbVie 2/1/2017 Presented by Sammy Saab, MD, MPH, FACG, AGAF, FAASLD February 1 st, 2017 1 AbbVie disclosures This is an Abbvie sponsored
Introduction to the Impact of Resistance in Hepatitis C Sponsored by AbbVie 2/1/2017 Presented by Sammy Saab, MD, MPH, FACG, AGAF, FAASLD February 1 st, 2017 1 AbbVie disclosures This is an Abbvie sponsored
Conditional and reversible disruption of essential herpesvirus protein functions
 nature methods Conditional and reversible disruption of essential herpesvirus protein functions Mandy Glaß, Andreas Busche, Karen Wagner, Martin Messerle & Eva Maria Borst Supplementary figures and text:
nature methods Conditional and reversible disruption of essential herpesvirus protein functions Mandy Glaß, Andreas Busche, Karen Wagner, Martin Messerle & Eva Maria Borst Supplementary figures and text:
Fayth K. Yoshimura, Ph.D. September 7, of 7 HIV - BASIC PROPERTIES
 1 of 7 I. Viral Origin. A. Retrovirus - animal lentiviruses. HIV - BASIC PROPERTIES 1. HIV is a member of the Retrovirus family and more specifically it is a member of the Lentivirus genus of this family.
1 of 7 I. Viral Origin. A. Retrovirus - animal lentiviruses. HIV - BASIC PROPERTIES 1. HIV is a member of the Retrovirus family and more specifically it is a member of the Lentivirus genus of this family.
Nature Medicine: doi: /nm.2109
 HIV 1 Infects Multipotent Progenitor Cells Causing Cell Death and Establishing Latent Cellular Reservoirs Christoph C. Carter, Adewunmi Onafuwa Nuga, Lucy A. M c Namara, James Riddell IV, Dale Bixby, Michael
HIV 1 Infects Multipotent Progenitor Cells Causing Cell Death and Establishing Latent Cellular Reservoirs Christoph C. Carter, Adewunmi Onafuwa Nuga, Lucy A. M c Namara, James Riddell IV, Dale Bixby, Michael
Hepatitis C Virus and Antiviral Drug Resistance
 Gut and Liver, Vol. 10, No. 6, November 2016, pp. 890-895 Review Hepatitis C Virus and Antiviral Drug Resistance Seungtaek Kim, Kwang-Hyub Han, and Sang Hoon Ahn Institute of Gastroenterology, Department
Gut and Liver, Vol. 10, No. 6, November 2016, pp. 890-895 Review Hepatitis C Virus and Antiviral Drug Resistance Seungtaek Kim, Kwang-Hyub Han, and Sang Hoon Ahn Institute of Gastroenterology, Department
SUPPLEMENTARY INFORMATION
 sirna pool: Control Tetherin -HA-GFP HA-Tetherin -Tubulin Supplementary Figure S1. Knockdown of HA-tagged tetherin expression by tetherin specific sirnas. HeLa cells were cotransfected with plasmids expressing
sirna pool: Control Tetherin -HA-GFP HA-Tetherin -Tubulin Supplementary Figure S1. Knockdown of HA-tagged tetherin expression by tetherin specific sirnas. HeLa cells were cotransfected with plasmids expressing
Supplementary information
 Supplementary information Exosomes mediate the cell-to-cell transmission of interferon alpha-induced antiviral activity Jianhua Li, Kuancheng Liu, Yang Liu, Yan Xu, Fei Zhang, Huijuan Yang, Jiangxia Liu,
Supplementary information Exosomes mediate the cell-to-cell transmission of interferon alpha-induced antiviral activity Jianhua Li, Kuancheng Liu, Yang Liu, Yan Xu, Fei Zhang, Huijuan Yang, Jiangxia Liu,
Manish Sagar, 1,2 Xueling Wu, 2 Sandra Lee, 3 and Julie Overbaugh 2 *
 JOURNAL OF VIROLOGY, Oct. 2006, p. 9586 9598 Vol. 80, No. 19 0022-538X/06/$08.00 0 doi:10.1128/jvi.00141-06 Copyright 2006, American Society for Microbiology. All Rights Reserved. Human Immunodeficiency
JOURNAL OF VIROLOGY, Oct. 2006, p. 9586 9598 Vol. 80, No. 19 0022-538X/06/$08.00 0 doi:10.1128/jvi.00141-06 Copyright 2006, American Society for Microbiology. All Rights Reserved. Human Immunodeficiency
Hepatitis C virus (HCV) is a global public health problem,
 Molecular Clones of Hepatitis C Virus: Applications to Animal Models Michael Gale, Jr., and Michael R. Beard Abstract Hepatitis C virus (HCV) is a global public health problem, with approximately 3% of
Molecular Clones of Hepatitis C Virus: Applications to Animal Models Michael Gale, Jr., and Michael R. Beard Abstract Hepatitis C virus (HCV) is a global public health problem, with approximately 3% of
Supplementary Figure 1
 Supplementary Figure 1 Asymmetrical function of 5p and 3p arms of mir-181 and mir-30 families and mir-142 and mir-154. (a) Control experiments using mirna sensor vector and empty pri-mirna overexpression
Supplementary Figure 1 Asymmetrical function of 5p and 3p arms of mir-181 and mir-30 families and mir-142 and mir-154. (a) Control experiments using mirna sensor vector and empty pri-mirna overexpression
Disclosures. No conflict of interest. van der Ree, AASLD
 A Single Dose of, a GalNAc-Conjugated Oligonucleotide Targeting mir-122, Results in Undetectable HCV RNA Levels in Chronic Hepatitis C Patients at Week 28 of Follow-up M.H. van der Ree 1, J.M.L. de Vree
A Single Dose of, a GalNAc-Conjugated Oligonucleotide Targeting mir-122, Results in Undetectable HCV RNA Levels in Chronic Hepatitis C Patients at Week 28 of Follow-up M.H. van der Ree 1, J.M.L. de Vree
CDC website:
 Hepatitis B virus CDC website: http://www.cdc.gov/ncidod/diseases/hepatitis/slideset/hep_b/slide_1.htm Relevance Key Features es of Hepatitis t B Virus 250 million people infected worldwide. In areas of
Hepatitis B virus CDC website: http://www.cdc.gov/ncidod/diseases/hepatitis/slideset/hep_b/slide_1.htm Relevance Key Features es of Hepatitis t B Virus 250 million people infected worldwide. In areas of
Fine Mapping of a cis-acting Sequence Element in Yellow Fever Virus RNA That Is Required for RNA Replication and Cyclization
 JOURNAL OF VIROLOGY, Feb. 2003, p. 2265 2270 Vol. 77, No. 3 0022-538X/03/$08.00 0 DOI: 10.1128/JVI.77.3.2265 2270.2003 Copyright 2003, American Society for Microbiology. All Rights Reserved. Fine Mapping
JOURNAL OF VIROLOGY, Feb. 2003, p. 2265 2270 Vol. 77, No. 3 0022-538X/03/$08.00 0 DOI: 10.1128/JVI.77.3.2265 2270.2003 Copyright 2003, American Society for Microbiology. All Rights Reserved. Fine Mapping
Supplementary Fig. 1. Delivery of mirnas via Red Fluorescent Protein.
 prfp-vector RFP Exon1 Intron RFP Exon2 prfp-mir-124 mir-93/124 RFP Exon1 Intron RFP Exon2 Untransfected prfp-vector prfp-mir-93 prfp-mir-124 Supplementary Fig. 1. Delivery of mirnas via Red Fluorescent
prfp-vector RFP Exon1 Intron RFP Exon2 prfp-mir-124 mir-93/124 RFP Exon1 Intron RFP Exon2 Untransfected prfp-vector prfp-mir-93 prfp-mir-124 Supplementary Fig. 1. Delivery of mirnas via Red Fluorescent
Pre-made Reporter Lentivirus for NF-κB Signal Pathway
 Pre-made Reporter for NF-κB Signal Pathway Cat# Product Name Amounts LVP965-P or: LVP965-P-PBS NFKB-GFP (Puro) LVP966-P or: LVP966-P-PBS NFKB-RFP (Puro) LVP967-P or: LVP967-P-PBS NFKB-Luc (Puro) LVP968-P
Pre-made Reporter for NF-κB Signal Pathway Cat# Product Name Amounts LVP965-P or: LVP965-P-PBS NFKB-GFP (Puro) LVP966-P or: LVP966-P-PBS NFKB-RFP (Puro) LVP967-P or: LVP967-P-PBS NFKB-Luc (Puro) LVP968-P
Chapter 2. Investigation into mir-346 Regulation of the nachr α5 Subunit
 15 Chapter 2 Investigation into mir-346 Regulation of the nachr α5 Subunit MicroRNA s (mirnas) are small (< 25 base pairs), single stranded, non-coding RNAs that regulate gene expression at the post transcriptional
15 Chapter 2 Investigation into mir-346 Regulation of the nachr α5 Subunit MicroRNA s (mirnas) are small (< 25 base pairs), single stranded, non-coding RNAs that regulate gene expression at the post transcriptional
Bioluminescence Resonance Energy Transfer (BRET)-based studies of receptor dynamics in living cells with Berthold s Mithras
 Bioluminescence Resonance Energy Transfer (BRET)-based studies of receptor dynamics in living cells with Berthold s Mithras Tarik Issad, Ralf Jockers and Stefano Marullo 1 Because they play a pivotal role
Bioluminescence Resonance Energy Transfer (BRET)-based studies of receptor dynamics in living cells with Berthold s Mithras Tarik Issad, Ralf Jockers and Stefano Marullo 1 Because they play a pivotal role
VIRAL TITER COUNTS. The best methods of measuring infectious lentiviral titer
 VIRAL TITER COUNTS The best methods of measuring infectious lentiviral titer FLUORESCENCE CYCLES qpcr of Viral RNA SUMMARY Viral vectors are now routinely used for gene transduction in a wide variety of
VIRAL TITER COUNTS The best methods of measuring infectious lentiviral titer FLUORESCENCE CYCLES qpcr of Viral RNA SUMMARY Viral vectors are now routinely used for gene transduction in a wide variety of
Rama Nada. - Malik
 - 2 - Rama Nada - - Malik 1 P a g e We talked about HAV in the previous lecture, now we ll continue the remaining types.. Hepatitis E It s similar to virus that infect swine, so its most likely infect
- 2 - Rama Nada - - Malik 1 P a g e We talked about HAV in the previous lecture, now we ll continue the remaining types.. Hepatitis E It s similar to virus that infect swine, so its most likely infect
JVI Accepts, published online ahead of print on 23 March 2011 J. Virol. doi: /jvi
 JVI Accepts, published online ahead of print on 23 March 2011 J. Virol. doi:10.1128/jvi.02116-10 Copyright 2011, American Society for Microbiology and/or the Listed Authors/Institutions. All Rights Reserved.
JVI Accepts, published online ahead of print on 23 March 2011 J. Virol. doi:10.1128/jvi.02116-10 Copyright 2011, American Society for Microbiology and/or the Listed Authors/Institutions. All Rights Reserved.
Blocking the expression of the hepatitis B virus S gene in hepatocellular carcinoma cell lines with an anti-gene locked nucleic acid in vitro
 Blocking the expression of the hepatitis B virus S gene in hepatocellular carcinoma cell lines with an anti-gene locked nucleic acid in vitro Y.-B. Deng, H.-J. Qin, Y.-H. Luo, Z.-R. Liang and J.-J. Zou
Blocking the expression of the hepatitis B virus S gene in hepatocellular carcinoma cell lines with an anti-gene locked nucleic acid in vitro Y.-B. Deng, H.-J. Qin, Y.-H. Luo, Z.-R. Liang and J.-J. Zou
Regulation of cell signaling cascades by influenza A virus
 The Second International Symposium on Optimization and Systems Biology (OSB 08) Lijiang, China, October 31 November 3, 2008 Copyright 2008 ORSC & APORC, pp. 389 394 Regulation of cell signaling cascades
The Second International Symposium on Optimization and Systems Biology (OSB 08) Lijiang, China, October 31 November 3, 2008 Copyright 2008 ORSC & APORC, pp. 389 394 Regulation of cell signaling cascades
Virion Genome Genes and proteins Viruses and hosts Diseases Distinctive characteristics
 Hepadnaviruses Virion Genome Genes and proteins Viruses and hosts Diseases Distinctive characteristics Hepatitis viruses A group of unrelated pathogens termed hepatitis viruses cause the vast majority
Hepadnaviruses Virion Genome Genes and proteins Viruses and hosts Diseases Distinctive characteristics Hepatitis viruses A group of unrelated pathogens termed hepatitis viruses cause the vast majority
Supplementary Figure 1. AdipoR1 silencing and overexpression controls. (a) Representative blots (upper and lower panels) showing the AdipoR1 protein
 Supplementary Figure 1. AdipoR1 silencing and overexpression controls. (a) Representative blots (upper and lower panels) showing the AdipoR1 protein content relative to GAPDH in two independent experiments.
Supplementary Figure 1. AdipoR1 silencing and overexpression controls. (a) Representative blots (upper and lower panels) showing the AdipoR1 protein content relative to GAPDH in two independent experiments.
HIV-1 Virus-like Particle Budding Assay Nathan H Vande Burgt, Luis J Cocka * and Paul Bates
 HIV-1 Virus-like Particle Budding Assay Nathan H Vande Burgt, Luis J Cocka * and Paul Bates Department of Microbiology, Perelman School of Medicine at the University of Pennsylvania, Philadelphia, USA
HIV-1 Virus-like Particle Budding Assay Nathan H Vande Burgt, Luis J Cocka * and Paul Bates Department of Microbiology, Perelman School of Medicine at the University of Pennsylvania, Philadelphia, USA
Supplementary Information
 Supplementary Information HBV maintains electrostatic homeostasis by modulating negative charges from phosphoserine and encapsidated nucleic acids Authors: Pei-Yi Su 1,2,3, Ching-Jen Yang 2, Tien-Hua Chu
Supplementary Information HBV maintains electrostatic homeostasis by modulating negative charges from phosphoserine and encapsidated nucleic acids Authors: Pei-Yi Su 1,2,3, Ching-Jen Yang 2, Tien-Hua Chu
Hepatitis C virus (HCV) is an enveloped,
 Human Serum Leads to Differentiation of Human Hepatoma Cells, Restoration of Very-Low-Density Lipoprotein Secretion, and a 1000-Fold Increase in HCV Japanese Fulminant Hepatitis Type 1 Titers Rineke H.G.
Human Serum Leads to Differentiation of Human Hepatoma Cells, Restoration of Very-Low-Density Lipoprotein Secretion, and a 1000-Fold Increase in HCV Japanese Fulminant Hepatitis Type 1 Titers Rineke H.G.
Worldwide more than 130 million people are
 RAPID COMMUNICATION Cyclosporine A Inhibits Hepatitis C Virus Nonstructural Protein 2 Through Cyclophilin A Sandra Ciesek, 1,2 Eike Steinmann, 2 Heiner Wedemeyer, 1 Michael P. Manns, 1 Johann Neyts, 3
RAPID COMMUNICATION Cyclosporine A Inhibits Hepatitis C Virus Nonstructural Protein 2 Through Cyclophilin A Sandra Ciesek, 1,2 Eike Steinmann, 2 Heiner Wedemeyer, 1 Michael P. Manns, 1 Johann Neyts, 3
Pre-made Reporter Lentivirus for JAK-STAT Signaling Pathway
 Pre-made Reporter for JAK-STAT Signaling Pathway Cat# Product Name Amounts LVP937-P or: LVP937-P-PBS ISRE-GFP (Puro) LVP938-P or: LVP938-P-PBS ISRE-RFP (Puro) LVP939-P or: LVP939-P-PBS ISRE-Luc (Puro)
Pre-made Reporter for JAK-STAT Signaling Pathway Cat# Product Name Amounts LVP937-P or: LVP937-P-PBS ISRE-GFP (Puro) LVP938-P or: LVP938-P-PBS ISRE-RFP (Puro) LVP939-P or: LVP939-P-PBS ISRE-Luc (Puro)
Cellular MicroRNA and P Bodies Modulate Host-HIV-1 Interactions. 指導教授 : 張麗冠博士 演講者 : 黃柄翰 Date: 2009/10/19
 Cellular MicroRNA and P Bodies Modulate Host-HIV-1 Interactions 指導教授 : 張麗冠博士 演講者 : 黃柄翰 Date: 2009/10/19 1 MicroRNA biogenesis 1. Pri mirna: primary mirna 2. Drosha: RNaseIII 3. DCR 1: Dicer 1 RNaseIII
Cellular MicroRNA and P Bodies Modulate Host-HIV-1 Interactions 指導教授 : 張麗冠博士 演講者 : 黃柄翰 Date: 2009/10/19 1 MicroRNA biogenesis 1. Pri mirna: primary mirna 2. Drosha: RNaseIII 3. DCR 1: Dicer 1 RNaseIII
Coronaviruses cause acute, mild upper respiratory infection (common cold).
 Coronaviruses David A. J. Tyrrell Steven H. Myint GENERAL CONCEPTS Clinical Presentation Coronaviruses cause acute, mild upper respiratory infection (common cold). Structure Spherical or pleomorphic enveloped
Coronaviruses David A. J. Tyrrell Steven H. Myint GENERAL CONCEPTS Clinical Presentation Coronaviruses cause acute, mild upper respiratory infection (common cold). Structure Spherical or pleomorphic enveloped
Technical Bulletin No. 162
 CPAL Central Pennsylvania Alliance Laboratory Technical Bulletin No. 162 cobas 6800 HCV Viral Load Assay - New Platform - June 1, 2017 Contact: Heather Habig, MLS (ASCP) CM, MB CM, 717-851-1422 Operations
CPAL Central Pennsylvania Alliance Laboratory Technical Bulletin No. 162 cobas 6800 HCV Viral Load Assay - New Platform - June 1, 2017 Contact: Heather Habig, MLS (ASCP) CM, MB CM, 717-851-1422 Operations
Satiya Wati,* Peng Li, Christopher J. Burrell, and Jillian M. Carr
 JOURNAL OF VIROLOGY, Sept. 2007, p. 10161 10171 Vol. 81, No. 18 0022-538X/07/$08.00 0 doi:10.1128/jvi.00313-07 Copyright 2007, American Society for Microbiology. All Rights Reserved. Dengue Virus (DV)
JOURNAL OF VIROLOGY, Sept. 2007, p. 10161 10171 Vol. 81, No. 18 0022-538X/07/$08.00 0 doi:10.1128/jvi.00313-07 Copyright 2007, American Society for Microbiology. All Rights Reserved. Dengue Virus (DV)
Reverse Genetics of RNA Viruses
 Reverse Genetics of RNA Viruses Reverse Genetics (RG) he creation of a virus with a fulllength copy of the viral genome he most powerful tool in modern virology RG of RNA viruses Generation or recovery
Reverse Genetics of RNA Viruses Reverse Genetics (RG) he creation of a virus with a fulllength copy of the viral genome he most powerful tool in modern virology RG of RNA viruses Generation or recovery
RNA interference induced hepatotoxicity results from loss of the first synthesized isoform of microrna-122 in mice
 SUPPLEMENTARY INFORMATION RNA interference induced hepatotoxicity results from loss of the first synthesized isoform of microrna-122 in mice Paul N Valdmanis, Shuo Gu, Kirk Chu, Lan Jin, Feijie Zhang,
SUPPLEMENTARY INFORMATION RNA interference induced hepatotoxicity results from loss of the first synthesized isoform of microrna-122 in mice Paul N Valdmanis, Shuo Gu, Kirk Chu, Lan Jin, Feijie Zhang,
Ralf Bartenschlager, Charles Rice, and Michael Sofia are honored with the 2016 Lasker~DeBakey Clinical Medical Research Award
 The Journal of Clinical Investigation NEWS Ralf Bartenschlager, Charles Rice, and Michael Sofia are honored with the 2016 Lasker~DeBakey Clinical Medical Research Award Hepatitis C virus (HCV) infects
The Journal of Clinical Investigation NEWS Ralf Bartenschlager, Charles Rice, and Michael Sofia are honored with the 2016 Lasker~DeBakey Clinical Medical Research Award Hepatitis C virus (HCV) infects
Received 3 September 2002/Accepted 15 January 2003
 JOURNAL OF VIROLOGY, Apr. 2003, p. 4646 4657 Vol. 77, No. 8 0022-538X/03/$08.00 0 DOI: 10.1128/JVI.77.8.4646 4657.2003 Copyright 2003, American Society for Microbiology. All Rights Reserved. Ability of
JOURNAL OF VIROLOGY, Apr. 2003, p. 4646 4657 Vol. 77, No. 8 0022-538X/03/$08.00 0 DOI: 10.1128/JVI.77.8.4646 4657.2003 Copyright 2003, American Society for Microbiology. All Rights Reserved. Ability of
Hepatitis C Virus and Cytokine Responses
 Hepatitis C Virus and Cytokine Responses Eui-Cheol Shin, M.D., Ph.D. Laboratory of Immunology & Infectious Diseases (LIID), Graduate School of Medical Science & Engineering (GSMSE), KAIST Daejeon, Korea
Hepatitis C Virus and Cytokine Responses Eui-Cheol Shin, M.D., Ph.D. Laboratory of Immunology & Infectious Diseases (LIID), Graduate School of Medical Science & Engineering (GSMSE), KAIST Daejeon, Korea
Life Sciences 1A Midterm Exam 2. November 13, 2006
 Name: TF: Section Time Life Sciences 1A Midterm Exam 2 November 13, 2006 Please write legibly in the space provided below each question. You may not use calculators on this exam. We prefer that you use
Name: TF: Section Time Life Sciences 1A Midterm Exam 2 November 13, 2006 Please write legibly in the space provided below each question. You may not use calculators on this exam. We prefer that you use
