|
|
|
- Madeleine Thompson
- 6 years ago
- Views:
Transcription
1 27027 Tourney Road Valencia, CA Test Updates June 27, 2009 Dear Colleague: Specialty Laboratories is pleased to announce the improvement of our turnaround times on the following Hepatitis assays with the introduction of a new chemiluminescence methodology Hepatitis C Antibody with Reflex to RIBA 2445B Hepatitis C Antibody with Reflex to RIBA + Bands 2446 Hepatitis C Antibody 2448 Hepatitis B Surface Antibody, Quantitative 2450 Hepatitis A Antibody, Total 2451 Hepatitis A Antibody, IgM 2452 Hepatitis B Core Antibody, Total 2453 Hepatitis B Surface Antibody, Qualitative 2454 Hepatitis B Surface Antigen with Reflex to Confirmation 2457 Hepatitis B Core Antibody, IgM 2460 Hepatitis A Antibodies, Total & IgM 2461 Hepatitis B Virus Evaluation 2463 Hepatitis A & B Virus Evaluation 2464 Hepatitis A, B & C Virus Evaluation 2476 Hepatitis B Core Antibody, Total with Reflex to IgM 2477 Hepatitis A Antibody, Total with Reflex to IgM 7492 Hepatitis C Antibody with Reflex RNA DetectR 7493 Hepatitis C Antibody with Reflex to RNA Quantitation 7756 Hepatitis A & B Virus Acute Evaluation 7757 Hepatitis Acute Profile 7758 Hepatitis A, B & C Virus Post Exposure Panel Kidney stone remnant materials will be kept in storage for 21 days affecting the following tests: Stone Analysis, Kidney [4155], Kidney Stone Analysis with Stone Image [4160] and Stone Analysis, Non-Kidney Stones [4161]. We thank you for choosing Specialty and look forward to your continued support. For additional information, please visit our Web site at or contact Client Relations at Respectfully Yours, Christopher Lockhart, M.D. Laboratory Director
2 Test Changes: 2472 Rotavirus Ag Detection Collection Instructions Specimen of choice: fresh stool specimen collected as soon as possible after onset of symptoms (3-5 days). Diapers and rectal swabs for infants are acceptable. Specimens collected in formalin, PVA or SAF are not acceptable. Culturettes with gel are not acceptable. Collect 0.5 to 1 g (ml) fresh stool in a clean, leak proof plastic container (no preservatives). Sample may be stored at 2-8 degrees C for 3 days. For long term storage, freezing at -20 degrees C or lower is recommended. Ship to Specialty Laboratories within 24 hours of collection on dry ice Disaccharidases Collection Instructions Place 5 mg (2 mg) small bowel biopsy in a polypropylene screw cap collection tube and freeze within 2 hours of collection. Keep frozen on dry ice. Store at -50 to -90 degrees C Osteocalcin Specimen/Stability Serum 1.0 (0.5) ml: Refrigerated 72 hours, Frozen 3 Plasma EDTA 1.0 (0.5) ml: Refrigerated 72 hours, Frozen 3 Note: Previous stability: 7560 Treponema pallidum Total Abs, FTA-Abs Quant CPT Code Also Affected DOS Code 7560C 5396 JAK2 V617F Mutation, Ql, w/rfx Exons 12, 13 & MPL W515, S505 CPT Code 83891, 83898, 83902, 83904, If the JAK2 V617F result is negative, then exons 12 and 13 will be performed for an additional charge (CPT codes: 83904x2, 83912). If exons 12 and 13 are negative then MPL W515 and S505 will be performed for an additional charge (83898, 83902, 83904x2, 83912) Hepatitis B Virus Drug Resistance DetectR Regulatory Status RUO (Research Use Only) Always Statement This test(s) was performed using a kit that has not been cleared or approved by the FDA. The analytical performance characteristics of this test have been determined by Specialty Laboratories. This test should not be used for diagnosis without confirmation by other medically established means. 2 of 15
3 8134 Hepatitis B Virus GenotypR Regulatory Status RUO (Research Use Only) Always Statement This test(s) was performed using a kit that has not been cleared or approved by the FDA. The analytical performance characteristics of this test have been determined by Specialty Laboratories. This test should not be used for diagnosis without confirmation by other medically established means Hepatitis B Virus Core/Precore Mutant DetectR Regulatory Status RUO (Research Use Only) Always Statement This test(s) was performed using a kit that has not been cleared or approved by the FDA. The analytical performance characteristics of this test have been determined by Specialty Laboratories. This test should not be used for diagnosis without confirmation by other medically established means Anti-Mullerian Hormone AssessR July 28 Reference Range Male: < 1 year ng/ml 1 6 years ng/ml 7 11 years ng/ml years ng/ml > 17 years ng/ml Female: < 14 years < = 4.0 ng/ml Premenopausal ng/ml Postmenopausal < = 0.3 ng/ml Note This test is not approved for the testing of patient samples from New York State 5686 Aerobic Suscept: Gram Positive MIC Panel Urine/Non-Urine July 28 Components Levofloxacin MIC ADD Ceftriaxone MIC ADD All other components remain the same. 3 of 15
4 2446 Hepatitis C Virus IgG Abs Hepatitis C Antibody Specimen/Stability Serum 1.0 (0.5) ml: Ambient 5 days, Refrigerated 14 days, Frozen 2 Plasma EDTA 1.0 (0.5) ml: Ambient 5 days, Refrigerated 14 days, Plasma Heparin 1.0 (0.5) ml: Ambient 5 days, Refrigerated 14 days, change Component Signal to Cutoff (ADD) Reference Range <1.00 Always Comments When the Signal to Cutoff result is : The patient s sample tests reactive with a low S/CO ratio: > or = 1.0 and < 8.0. The CDC recommends supplemental testing such as RIBA or nucleic acid amplified testing (NAAT) for confirmation (MMWR No. RR-3, 2003). If NAAT is chosen, please submit a new, frozen plasma or serum specimen. When the Signal to Cutoff result is >=8.00: The patient s sample tests reactive with a high S/CO ratio: > or = 8.0. Samples with high S/CO ratios have been shown to repeat as positive using a different methodology 95% of the time or greater. Therefore, additional testing for verification of the result is not recommended (CDC MMWR No. RR-3, 2003) Hepatitis B Virus Surface Antibody Quantitation Hepatitis B Surface Antibody, Quantitation Specimen/Stability Serum 2.0 (1.0) ml: Ambient 5 days, Refrigerated 14 days, Frozen 2 Plasma EDTA 2.0 (1.0) ml: Ambient 5 days, Refrigerated 14 days, Plasma Heparin 2.0 (1.0) ml: Ambient 5 days, Refrigerated 14 days, Note: Plasma ACD no longer accepted, Plasma heparin now accepted; Ambient stability change Reference Range >=10.0 miu/ml Always Comments When the Quantitative result is <10.0 miu/ml: Patient does not have immunity to hepatitis B virus. When the Quantitative result is >=10.0 miu/ml: Patient has immunity to hepatitis B virus. 4 of 15
5 2450 Hepatitis A Virus Total Abs Hepatitis A Antibody, Total Specimen/Stability Serum 1.5 (1.0) ml: Ambient 14 days, Refrigerated 14 days, Frozen 2 Plasma EDTA 1.5 (1.0) ml: Ambient 14 days, Refrigerated 14 days, Plasma Heparin 1.5 (1.0) ml: Ambient 14 days, Refrigerated 14 days, Note: Plasma ACD no longer accepted; Refrigerated stability change, Borderline Also Affected DOS Code Hepatitis A Virus IgM Abs Hepatitis A Antibody, IgM Specimen/Stability Serum 1.5 (1.0) ml: Ambient 14 days, Refrigerated 14 days, Frozen 2 Plasma EDTA 1.5 (1.0) ml: Ambient 14 days, Refrigerated 14 days, Plasma Heparin 1.5 (1.0) ml: Ambient 14 days, Refrigerated 14 days, Note: Plasma ACD no longer accepted; Ambient specimens now accepted; Refrigerated stability change, Borderline Also Affected DOS Code Hepatitis B Virus Core Total Abs Hepatitis B Core Antibody, Total Specimen/Stability Serum 1.5 (1.0) ml: Ambient 5 days, Refrigerated 14 days, Frozen 2 Plasma EDTA 1.5 (1.0) ml: Ambient 5 days, Refrigerated 14 days, Plasma Heparin 1.5 (1.0) ml: Ambient 5 days, Refrigerated 14 days, change 5 of 15
6 2453 Hepatitis B Virus Surface Total Abs Hepatitis B Surface Antibody, Qualitative Specimen/Stability Serum 2.0 (1.0) ml: Ambient 5 days, Refrigerated 14 days, Frozen 2 Plasma EDTA 2.0 (1.0) ml: Ambient 5 days, Refrigerated 14 days, Plasma Heparin 2.0 (1.0) ml: Ambient 5 days, Refrigerated 14 days, change, Borderline 2454 Hepatitis B Virus Surface Antigen w/reflex Neutralization Hepatitis B Surface Antigen with Reflex to Confirmation Specimen/Stability Serum 4.0 (2.5) ml: Ambient 7 days, Refrigerated 14 days, Frozen 2 Plasma EDTA 4.0 (2.5) ml: Ambient 7 days, Refrigerated 14 days, Plasma Heparin 4.0 (2.5) ml: Ambient 7 days, Refrigerated 14 days, Note: Plasma ACD and Citrate no longer accepted; Ambient, Refrigerated and Frozen Stability change Note Under the guidelines of this new, FDA approved methodology, only certain positive Hepatitis B Surface Antigen test results will reflex to Antibody Neutralization testing for confirmation. Reflex Borderline reactive results will automatically reflex to Hepatitis B Surface Antigen Neutralization for an additional fee. Neutralization results will be reported as Nonreactive, Reactive, Borderline Always Comment When the Neutralization result is Reactive: Hepatitis B Surface Antigen was confirmed as present by the Neutralization assay. When the Neutralization result is Borderline: Suggest repeating the Hepatitis B Surface Antigen assay on a second specimen, if clinically indicated. When the Neutralization result is Nonreactive: Hepatitis B Surface Antigen was not confirmed as present by the Neutralization (confirmation) assay. Prior to informing a patient that he/she is infected with HBsAg, the manufacturer s FDA approved package insert requires that a neutralization assay be used to confirm specific repeatedly reactive HBsAg samples. Because this patient s sample did not confirm, he/she should not be considered positive (reactive) for having a hepatitis B infection. Reactive screening results with nonreactive confirmatory results are most frequently seen in association with biological false reactive samples and repeating the test on a different sample from the same patient may yield identical results (reactive screen/nonreactive confirmation). Also Affected DOS Code of 15
7 2457 Hepatitis B Virus Core IgM Abs Hepatitis B Core Antibody, IgM Specimen/Stability Serum 1.5 (1.0) ml: Ambient 5 days, Refrigerated 14 days, Frozen 2 Plasma EDTA 1.5 (1.0) ml: Ambient 5 days, Refrigerated 14 days, Plasma Heparin 1.5 (1.0) ml: Ambient 5 days, Refrigerated 14 days, change 2476 Hepatitis B Virus Core Total Abs w/reflex IgM Hepatitis B Core Antibody, Total with Reflex to IgM Specimen/Stability Serum 1.5 (1.0) ml: Ambient 5 days, Refrigerated 14 days, Frozen 2 Plasma EDTA 1.5 (1.0) ml: Ambient 5 days, Refrigerated 14 days, Plasma Heparin 1.5 (1.0) ml: Ambient 5 days, Refrigerated 14 days, change Reflex Reactive results will automatically reflex to Hepatitis B Core Antibody, IgM for an additional fee Hepatitis A Virus Total Abs w/reflex IgM Hepatitis A Antibody, Total with Reflex to IgM Specimen/Stability Serum 1.5 (1.0) ml: Ambient 14 days, Refrigerated 14 days, Frozen 2 Plasma EDTA 1.5 (1.0) ml: Ambient 14 days, Refrigerated 14 days, Plasma Heparin 1.5 (1.0) ml: Ambient 14 days, Refrigerated 14 days, Note: Plasma ACD no longer accepted; Refrigerated stability change, Borderline Reflex Reactive results will automatically reflex to Hepatitis A Antibody, IgM for an additional fee. 7 of 15
8 2461 Hepatitis B Virus Evaluation Specimen/Stability Serum 5.0 (4.0) ml: Ambient 5 days, Refrigerated 14 days, Frozen 2 Plasma EDTA 5.0 (4.0) ml: Ambient 5 days, Refrigerated 14 days, Plasma Heparin 5.0 (4.0) ml: Ambient 5 days, Refrigerated 14 days, Note: Plasma EDTA and Heparin are now accepted; Ambient and Refrigerated stability change Changes noted above for the individual components will also apply Hepatitis A, B & C Virus Evaluation Specimen/Stability Serum 6.0 (5.0) ml: Ambient 5 days, Refrigerated 14 days, Frozen 2 Plasma EDTA 6.0 (5.0) ml: Ambient 5 days, Refrigerated 14 days, Plasma Heparin 6.0 (5.0) ml: Ambient 5 days, Refrigerated 14 days, Note: Plasma EDTA and Heparin are now accepted; Ambient now Accepted; Refrigerated stability change Also Affected DOS Code 2463 Changes noted above for the individual components will also apply Hepatitis A & B Virus Acute Evaluation Specimen/Stability Serum 4.0 (3.0) ml: Ambient 5 days, Refrigerated 14 days, Frozen 2 Plasma EDTA 4.0 (3.0) ml: Ambient 5 days, Refrigerated 14 days, Plasma Heparin 4.0 (3.0) ml: Ambient 5 days, Refrigerated 14 days, Note: Plasma ACD no longer accepted; Ambient now accepted; Refrigerated stability change Also Affected DOS Code 7757 Changes noted above for the individual components will also apply Hepatitis A, B & C Virus Post Exposure Panel Specimen/Stability Serum 4.0 (3.0) ml: Ambient 5 days, Refrigerated 14 days, Frozen 2 Plasma EDTA 4.0 (3.0) ml: Ambient 5 days, Refrigerated 14 days, Plasma Heparin 4.0 (3.0) ml: Ambient 5 days, Refrigerated 14 days, Change Changes noted above for the individual components will also apply. 8 of 15
9 2445 Hepatitis C Virus IgG Abs EIA w/reflex RIBA Hepatitis C Antibody with Reflex to RIBA Specimen/Stability Serum 2.0 (1.5) ml: Ambient 5 days, Refrigerated 14 days, Frozen 2 Plasma EDTA 2.0 (1.5) ml: Ambient 5 days, Refrigerated 14 days, Plasma Heparin 2.0 (1.5) ml: Ambient 5 days, Refrigerated 14 days, change Component Signal to Cutoff (ADD) Reference Range <1.00 Reflex Reactive results with a Signal to Cutoff value of will automatically reflex to Hepatitis C Virus Abs Immunoblot [RIBA] for an additional fee. Always Comments When the Signal to Cutoff result is >=8.00: The patient s sample tests reactive with a high S/CO ratio: > or = 8.0. Samples with high S/CO ratios have been shown to repeat as positive using a different methodology 95% of the time or greater. Therefore, additional testing for verification of the result is not recommended (CDC MMWR No. RR-3, 2003). Also Affected DOS Code 2445B 7492 Hepatitis C Virus Abs EIA w/reflex RNA DetectR Hepatitis C Antibody with Reflex to RNA DetectR Specimen #1/Stability Serum 1.5 (1.0) ml: Ambient 5 days, Refrigerated 14 days, Frozen 2 #1 Plasma EDTA 1.5 (1.0) ml: Ambient 5 days, Refrigerated 14 days, Plasma Heparin 1.5 (1.0) ml: Ambient 5 days, Refrigerated 14 days, Change Specimen #2/Stability Plasma EDTA 5.0 (2.0) ml: Frozen 14 days #2 Plasma ACD 5.0 (2.0) ml: Frozen 14 days Plasma PPT Tube 5.0 (2.0) ml: Ambient 72 hours, Refrigerated 72 hours, Frozen 28 days Component Signal to Cutoff (ADD) Reference Range <1.00 Reflex Reactive results with a Signal to Cutoff value of > or = 1.00 will automatically reflex to Hepatitis C Virus RNA DetectR for an additional fee. Always Comments When the Signal to Cutoff result is >=8.00: The patient s sample tests reactive with a high S/CO ratio: > or = 8.0. Samples with high S/CO ratios have been shown to repeat as positive using a different methodology 95% of the time or greater. Therefore, additional testing for verification of the result is not recommended (CDC MMWR No. RR-3, 2003). 9 of 15
10 7493 Hepatitis C Virus IgG Abs [EIA] w/rfx RNA Quantitation PCR Hepatitis C Antibody with Reflex to RNA Quantitation Specimen #1/Stability Serum 1.5 (1.0) ml: Ambient 5 days, Refrigerated 14 days, Frozen 2 #1 Plasma EDTA 1.5 (1.0) ml: Ambient 5 days, Refrigerated 14 days, Plasma Heparin 1.5 (1.0) ml: Ambient 5 days, Refrigerated 14 days, change Specimen #2/Stability Plasma ACD 5.0 (2.0) ml: #2 Plasma EDTA 5.0 (2.0) ml: Plasma PPT Tube 5.0 (2.0) ml: Ambient 72 hours, Refrigerated 72 hours, Frozen 28 days Component Signal to Cutoff (ADD) Reference Range <1.00 Reflex Reactive results with a Signal to Cutoff value of > or = 1.00 will automatically reflex to Hepatitis C Virus RNA Quantitation [PCR] for an additional fee. Always Comments When the Signal to Cutoff result is >=8.00: The patient s sample tests reactive with a high S/CO ratio: > or = 8.0. Samples with high S/CO ratios have been shown to repeat as positive using a different methodology 95% of the time or greater. Therefore, additional testing for verification of the result is not recommended (CDC MMWR No. RR-3, 2003). The CPT Codes provided are based on AMA Guidelines and are for informational purposes only. CPT Coding is the sole responsibility of the billing party. Please direct any questions regarding CPT Coding to the payer being billed. 10 of 15
11 New Referral Tests: The following tests are now available from Quest Diagnostics and may be referred through Specialty Laboratories. S51881 Liver Fibrosis Panel (HepaScore ) (19647X) S51883 NRAS Mutation Analysis (16818) S51882 EGFR Pathway (KRAS with reflex to NRAS, BRAF) (16819) S51887 Iodine, Random Urine (16601) Please call client relations at or visit our website at for ordering information. 11 of 15
12 Discontinued Tests: : S46365 Iodine 24Hr Urine [9549] Recommended replacement: S51886 Iodine, 24-hr Urine [16602] S51500 SensiTrop HIV Co-Receptor Assay Recommended replacement: S51884 HIV-1 Coreceptor Tropism [16710] July 28: 1538 Cardiovascular Disease Risk AssessR Recommended replacement: No replacement 12 of 15
13 Discontinued Tests: (cont d) Due to the discontinuation of allergy testing at Esoterix Laboratory Services, Inc., the following test codes will no longer be available. In as many cases as possible, an alternative test has been established with testing performed at IBT Laboratories in Lenexa, Kansas. Please note that the new sendout code from Specialty is provided in the list. DISCONTINUED CODE TEST NAME CODE NAME S50118 ALLERGY-CEPHALOSPORIN DRUG IGE S51963 Allergen-Cephalosporin IgE (87510E) S49080 RAST-ALFALFA IGE S51961 Allergen-Alfalfa IgE (59710S) S42068 RAST-ARTICHOKE IGE S51889 Allergen-Artichoke IgE (30610S) S49045 RAST-ASPEN IGE S51958 Allergen-Aspen IgE (64610S) S49317 RAST-ASPERGILLUS AMSTELOIDAMI IGE S51890 Allergen-Aspergillus Amsteloidami IgE (78710E) S46421 RAST-ASPERGILLUS FLAVUS IGE S51934 Allergen-Aspergillus Flavus IgE (191010E) S48811 RAST-ASPERGILLUS NIDULANS IGE S51954 Allergen-Aspergillus Nidulans IgE (190910E) S50915 RAST-ASPERGILLUS TERREUS IGE [M18] S51978 Allergen-Aspergillus Terreus IgE (78110E) S49318 RAST-ASPERGILLUS VERSICOLOR IGE S51891 Allergen-Aspergillus Versicolor IgE (78610A) S41638 RAST-BACCHARIS (GROUNDSEL) IGE S51892 Allergen-Groundsel Tree IgE (67710E) S41746 RAST-BASS BLACK IGE S51893 Allergen-Bass Black Ige (43310S) S43199 RAST-BAYBERRY IGE S51894 Allergen-Bayberry/Waxmyrtle IgE (67510S) S43674 RAST-BEAN PINTO IGE S51895 Allergen-Bean Pinto IgE (56310S) S48540 RAST-CANTALOUPE/MUSKMELON IGE S51947 Allergen-Melon (Cantaloupe, Honeydew, Muskmelon) IgE (54910S) S41182 RAST-CATFISH IGE S51888 Allergen-Catfish IgE (43210S) S46806 RAST-CEDAR MOUNTAIN IGE S51937 Allergen-Cedar Mountain IgE (63010S) S41749 RAST-CHEESE BLEU IGE S51896 Allergen-Cheese Bleu ige (39410E) S42211 RAST-CHEESE PARMESAN IGE S51897 Allergen-Cheese Parmesan IgE (49110S) S42213 RAST-CHEESE PROCESSED AMERICAN IGE S51898 Allergen-Cheese American IgE (31310S) S41748 RAST-CHEESE, SWISS IGE S51899 Allergen-Cheese Swiss IgE (43910S) S50784 RAST-CINNAMON IGG S51975 Allergen-Cinnamon IgG (45920) S50386 RAST-COTTAGE CHEESE IGE S51967 Allergen-Cheese Cottage IgE (34110S) S41487 RAST-CUMIN IGE S51900 Allergen-Cumin Seed IgE (30210S) S50531 RAST-CURVULARIA SPECIFERA [M55] S51972 Allergen-Curvularia Spicifera/Bipolaris IgE (187610E) S43196 RAST-CYPRESS, BALD WHITE IGE S51901 Allergen-Cypress Bald IgE (66010E) S43355 RAST-DOCK, BITTER IGE S51902 Allergen-Dock Bitter IgE (74210E) S48652 RAST-DUCK IGE S51949 Allergen-Duck Meat IgE (40210S) S43072 RAST-ELM, CEDAR (SCRUB) IGE S51904 Allergen-Elm Cedar IgE (163610A) S42058 RAST-ELM-CHINESE IGE S51903 Allergen-Elm Chinese IgE (64210S) S49046 RAST-EPIDERMOPHYTON FLOCCOSUM IGE S51959 Allergen-Epidermophyton Floccosum IgE (77510E) S50367 RAST-ERYTHROMYCIN IGE [C27] S51964 Allergen-Erythromycin IgE (87710E) S46423 RAST-FENNEL, DOG IGE S51935 Allergen-Dog Fennel IgE (74610E) S46937 RAST-FLAXSEED IGE S51940 Allergen-Flaxseed IgE (87910E) S41637 RAST-FLEA MIX IGE S51905 Allergen-Flea IgE (92010E) S47120 RAST-FUSARIUM OXYSPORUM (VASIFEC) IGE S51941 Allergen-Fusarium Oxysporum/Vasinfectum IgE (79110E) S48810 RAST-GREEN ASH IGE S51953 Allergen-Ash Green IgE (64410S) S48728 RAST-HACKBERRY (NORTHERN) IGE S51951 Allergen-Hackberry IgE (67610S) S44076 RAST-HADDOCK IGE S51906 Allergen-Haddock IgE (57210S) S50911 RAST-HELMINTHOSPORIUM INTERSEMINATUM IGE [M33] NEW S51977 Allergen-Helminthosporium Interseminatum IgE (193310E) S48090 RAST-HELMINTHOSPORIUM SATIVUM IGE S51944 Allergen-Helminthospporium Sativum/Drecshiera IgE (79510E) S43071 RAST-HICKORY, WHITE IGE S51907 Allergen-Hickory White IgE (64010E) S46420 RAST-HORSERADISH IGE S51933 Allergen-Horseradish IgE (40710S) 13 of 15
14 Discontinued Tests: (cont d) DISCONTINUED NEW CODE TEST NAME CODE TEST NAME S50369 RAST-HOUSE FLY IGE S51965 Allergen-Fly House IgE (91710E) S50519 RAST-JOHNSON SMUT IGE (G26) S51968 Allergen-Smut Johnson IgE (77910S) S41497 RAST-JUNIPER WESTERN IGE S51908 Allergen-Juniper Western IgE (68310S) S49219 RAST-KARAYA GUM IGE S51962 Allergen-Gum Karaya IgE (88210E) S46462 RAST-LAMB IGE S51936 Allergen-Lamb/Mutton IgE (54310S) S50788 RAST-LEMON (FOOD) IGG S51976 Allergen-Lemon IgG (41020) S43233 RAST-LIMA BEAN IGE S51909 Allergen-Bean Lima IgE (43510E) S43197 RAST-MEXICAN TEA IGE S51910 Allergen-Mexican Tea IgE (72810S) S41989 RAST-NECTARINE IGE S51911 Allergen-Nectarine IgE (47910E) S50527 RAST-OAT SMUT IGE [G28] S51970 Allergen-Smut Oat IgE (79310E) S48567 RAST-OKRA IGE S51948 Allergen-Okra IgE (35310E) S43195 RAST-ORRIS ROOT IGE S51912 Allergen-Orris Root IgE (85910S) S48861 RAST-PAECILOMYCES VARIOTII IGE S51955 Allergen-Paecilomyces Variotii IgE (79010E) S43354 RAST-PARSNIP IGE S51913 Allergen-Parsnip IgE (39510E) S42065 RAST-PEA, BLACK EYED IGE S51914 Allergen-Pea Black-Eyed IgE (41410S) S49319 RAST-PENICILLIUM BREVICOMP IGE S51915 Allergen-Penicillium Brevicompactum IgE (78910E) S43232 RAST-PEPPER, BLACK/WHITE IGE S51916 & S51917 Allergen-Pepper Black IgE (57510S) & Allergen- Pepper White IgE(440105) S46009 RAST-PINE, LOBLOLLY IGE S51932 Allergen-Pine Loblolly IgE (67910E) S47190 RAST-PYRETHRUM IGE S51942 Allergen-Pyrethrum IgE (85810S) S41744 RAST-RABBIT BUSH IGE S51918 Allergen-Rabbit Bush IgE (72010S) S43353 RAST-RADISH IGE S51919 Allergen-Raddish IgE (40610S) S48731 RAST-RAGWEED SOUTHERN IGE S51952 Allergen-Ragweed Southern IgE (189410E) S46635 RAST-RED CEDAR [K26] S51946 Allergen-Cedar Red IgE (68110E) S41379 RAST-RED MAPLE IGE S51920 Allergen-Maple Red IgE (64510S) S41496 RAST-RED OAK IGE S51921 Allergen-Oak Red IgE (163010E) S41495 RAST-RED SNAPPER IGE S51922 Allergen-Red Snapper IgE (46710S) S48713 RAST-RHODOTORULA GLUTINIS IGE S51950 Allergen-Rhodotorula IgE (77010E) S50586 RAST-RHUBARB IGE [F65] S51973 Allergen-Rhubarb IgE (36310E) S49027 RAST-SALTBUSH IGE S51957 Allergen-Saltbush Annual IgE (73910S) S43863 RAST-SHEEP'S WOOL IGE S51923 Allergen-Wool Sheep IgE (87210E) S44248 RAST-SMUT, BERMUDA IGE S51924 Allergen-Smut Bermuda IgE (77810E) S43673 RAST-SQUASH, SUMMER IGE S51925 Allergen-Squash Summer IgE (54710S) S50529 RAST-STEMPHYLIUM SOLANI [M47] S51971 Allergen-Stemphylium Solani IgE (76310S) S50626 RAST-SUGAR CANE IGE [F131] S51974 Allergen-Sugar Cane IgE (42410S) S50370 RAST-SULFAMETHOXAZOL IGE S51966 Allergen-Sulfamethoxazol IgE (87610E) S41276 RAST-TANGERINE IGE S51926 Allergen-Tangerine IgE (142010E) S46814 RAST-TAPIOCA IGE S51939 Allergen-Tapioca IgE (49810S) S48990 RAST-TETRACYCLIN IGE S51956 Allergen-Tetracycline IgE (87810E) S46808 RAST-TREE OF HEAVEN IGE S51938 Allergen-Tree of Heaven IgE (65210E) S51031 RAST-TURNIP IGE [F187] S51979 Allergen-Turnip IgE (41610S) S43356 RAST-VENISON IGE S51927 Allergen-Venison IgE (44710S) S43676 RAST-WALNUT, ENGLISH IGE S51929 Allergen-Walnut English Food IgE (47610S) S44077 RAST-WALNUT,BLACK IGE S51928 Allergen-Walnut Black Food IgE (43710S) S47610 RAST-WATERHEMP, WESTERN IGE S51943 Allergen-Hemp Western Water IgE (72710S) S50523 RAST-WHEAT SMUT IGE (G22) S51969 Allergen-Smut Wheat IgE (79410E) S41275 RAST-WHITE FISH IGE S51930 Allergen-Whitefish IgE (49210E) S48507 RAST-WING SCALE IGE S51945 Allergen-Wingscale IgE (72110S) S42057 RAST-YELLOW DOCK IGE S51931 Allergen-Yellow Dock IgE (71810S) S49079 RAST-YOGURT IGE S51960 Allergen-Yogurt IgE (31510S) 14 of 15
15 Discontinued Tests: (cont d) DISCONTINUED NEW CODE TEST NAME STATUS S50985 CORN POLLEN IGE [G20] Not Available S49021 RAST-ACETYL SALICYLIC ACID Not Available S49486 RAST-CAMEL HAIR/DANDER IGE Not Available S46422 RAST-CEDAR, GIANT WESTERN RED IGE Not Available S46350 RAST-CEDAR, RED (EASTERN) IGE Not Available S41180 RAST-CHERRY BING IGE Not Available S42070 RAST-CHERRY TREE IGE Not Available S50787 RAST-CHERRY, BING IGG Not Available S50119 RAST-CODEINE IGE Not Available S43074 RAST-CYPRESS, MONTEREY IGE Not Available S41158 RAST-DILL MIX IGE Not Available S48732 RAST-GRAIN MILL DUST IGE Not Available S50907 RAST-HELMINTHOSPORIUM MAYDIS IGE (M34) Not Available S49020 RAST-IBUPROFEN IGE Not Available S49151 RAST-IODINE IGE Not Available S41398 RAST-JUNIPER CALIFORNIA IGE Not Available S41499 RAST-JUNIPER ONE SEED IGE Not Available S41498 RAST-JUNIPER PINCHOT IGE Not Available S48508 RAST-LAKE PERCH IGE Not Available S50220 RAST-LIDOCAINE IGE Not Available S50520 RAST-MAPLE SYRUP IGE (F158) Not Available S50060 RAST-MSG IGE Not Available S48712 RAST-MYCOGONE SPP. IGE Not Available S48854 RAST-NEUROSPORA SITOPHILA IGE Not Available S48653 RAST-NIGROSPORA-ORYZAE IGE Not Available S50960 RAST-NOVOCAINE IGE (C22) Not Available S49026 RAST-OAK, CALIFORNIA BLACK IGE Not Available S51105 RAST-PHOMA HERBARUM IGE [M27] Not Available S42069 RAST-POPLAR BALSAM IGE Not Available S48946 RAST-POVERTYWEED IGE Not Available S48899 RAST-RED PEPPER IGE Not Available S41639 RAST-ROSE IGE Not Available S46940 RAST-RYE ITALIAN IGE Not Available S43679 RAST-SAGEBRUSH, COMMON IGE Not Available S48900 RAST-SCORPION IGE Not Available S48714 RAST-SPONDYLOCLADIUM SPP IGE Not Available S46812 RAST-SQUASH, ZUCCHINI IGE Not Available S50782 RAST-SQUASH-SUMMER, IGG Not Available S46965 RAST-TOE MIX IGE Not Available S48449 RAST-WOOD DUST MIX IGE Not Available 15 of 15
 27027 Tourney Road Valencia, CA 91355 800 421 7110 www.specialtylabs.com Test Updates November 26, 2008 Dear Colleague: We are pleased to announce the availability of two new assays for an expanded evaluation
27027 Tourney Road Valencia, CA 91355 800 421 7110 www.specialtylabs.com Test Updates November 26, 2008 Dear Colleague: We are pleased to announce the availability of two new assays for an expanded evaluation
 27027 Tourney Road Valencia, CA 91355 800 421 7110 www.specialtylabs.com Test Updates July 27, 2009 Dear Colleague: As we prepare for the upcoming flu season, Specialty Laboratories clients should be aware
27027 Tourney Road Valencia, CA 91355 800 421 7110 www.specialtylabs.com Test Updates July 27, 2009 Dear Colleague: As we prepare for the upcoming flu season, Specialty Laboratories clients should be aware
 27027 Tourney Road Valencia, CA 91355 800 421 7110 www.specialtylabs.com Test Updates February 27, 2009 Dear Colleague: Specialty Laboratories is pleased to announce the introduction of an advanced kidney
27027 Tourney Road Valencia, CA 91355 800 421 7110 www.specialtylabs.com Test Updates February 27, 2009 Dear Colleague: Specialty Laboratories is pleased to announce the introduction of an advanced kidney
 27027 Tourney Road Valencia, CA 91355 800 421 7110 www.specialtylabs.com Test Updates September 25, 2009 Dear Colleague: Specialty Laboratories is pleased to announce a new assay, PT and PTT-LA Mixing
27027 Tourney Road Valencia, CA 91355 800 421 7110 www.specialtylabs.com Test Updates September 25, 2009 Dear Colleague: Specialty Laboratories is pleased to announce a new assay, PT and PTT-LA Mixing
July Monthly Update, Quest Diagnostics Nichols Institute, Valencia
 NEW TESTS Please Note: Not all test codes assigned to each assay are listed in the table of contents. Please refer to the complete listing on the page numbers indicated. Test Name Effective Date Page #
NEW TESTS Please Note: Not all test codes assigned to each assay are listed in the table of contents. Please refer to the complete listing on the page numbers indicated. Test Name Effective Date Page #
 27027 Tourney Road Valencia, CA 91355 800 421 7110 www.specialtylabs.com Test Updates May 27, 2010 Dear Colleague: Our May communication to you brings very exciting news of a new test offering available
27027 Tourney Road Valencia, CA 91355 800 421 7110 www.specialtylabs.com Test Updates May 27, 2010 Dear Colleague: Our May communication to you brings very exciting news of a new test offering available
All abnormal results for this test will automatically have the DNA test reflexed at an additional cost.
 2211 Michigan Avenue Santa Monica, CA 90404 800 421 4449 Dear Colleague: May-June 2004 REVISED 6-15-04 At Specialty we continue to focus on working with you to reduce the problems that can result in tests
2211 Michigan Avenue Santa Monica, CA 90404 800 421 4449 Dear Colleague: May-June 2004 REVISED 6-15-04 At Specialty we continue to focus on working with you to reduce the problems that can result in tests
Allergy IgE Allergy Test Sensitivity and Specificity 6/23/17
 Allergy IgE Allergy Test Sensitivity and Specificity 6/23/17 1 Sensitivity and Specificity Benchmarking Goal Test pooled human sera with known positive and negative reactivity to determine Allergy sensitivity
Allergy IgE Allergy Test Sensitivity and Specificity 6/23/17 1 Sensitivity and Specificity Benchmarking Goal Test pooled human sera with known positive and negative reactivity to determine Allergy sensitivity
Test Catalog Updates August 3, 2016 Updates noted in Red
 s Updates noted in Red BLOD1424 Allergen Food Basil No page Specimen Requirement 0.5 ml (0.3 ml minimum) serum. Send Stability: RMT - NA REFT - 14 days Frozen - 14 days BLOD0248 Allergen Food Green Bean
s Updates noted in Red BLOD1424 Allergen Food Basil No page Specimen Requirement 0.5 ml (0.3 ml minimum) serum. Send Stability: RMT - NA REFT - 14 days Frozen - 14 days BLOD0248 Allergen Food Green Bean
Proposed Revision or Revocation of Maximum Residue Limits for Discontinued Agricultural Pest Control Products: Update 2
 Proposed Maximum Residue Limit PMRL2018-44 Proposed Revision or Revocation of Maximum Residue Limits for Discontinued Agricultural Pest Control Products: Update 2 (publié aussi en français) 8 November
Proposed Maximum Residue Limit PMRL2018-44 Proposed Revision or Revocation of Maximum Residue Limits for Discontinued Agricultural Pest Control Products: Update 2 (publié aussi en français) 8 November
New Test Announcements
 27027 Tourney Road Valencia, CA 91355 800 421 7110 www.specialtylabs.com New Test Announcements Dear Colleague: We are pleased to announce many new test offerings this month. January 25, 2007 In oncology,
27027 Tourney Road Valencia, CA 91355 800 421 7110 www.specialtylabs.com New Test Announcements Dear Colleague: We are pleased to announce many new test offerings this month. January 25, 2007 In oncology,
 27027 Tourney Road Valencia, CA 91355 800 421 7110 www.specialtylabs.com Test Updates October 28, 2009 Dear Colleague: Specialty Laboratories is pleased to announce the January availability of two trace
27027 Tourney Road Valencia, CA 91355 800 421 7110 www.specialtylabs.com Test Updates October 28, 2009 Dear Colleague: Specialty Laboratories is pleased to announce the January availability of two trace
12/10/ Immediate Action, Quest Diagnostics Nichols Institute, Valencia
 NEW TESTS Please Note: Not all test codes assigned to each assay are listed in the table of contents. Please refer to the complete listing on the page numbers indicated. Test Code Test Name Effective Date
NEW TESTS Please Note: Not all test codes assigned to each assay are listed in the table of contents. Please refer to the complete listing on the page numbers indicated. Test Code Test Name Effective Date
 27027 Tourney Road Valencia, CA 91355 800 421 7110 www.specialtylabs.com Test Updates May 21, 2007 Dear Colleague: As part of our continuing efforts to improve testing turnaround times, quality, and service,
27027 Tourney Road Valencia, CA 91355 800 421 7110 www.specialtylabs.com Test Updates May 21, 2007 Dear Colleague: As part of our continuing efforts to improve testing turnaround times, quality, and service,
CMS will not implement the new tier codes for Medicare/Medicaid claims for calendar year 2012.
 January 1, 2012 Re: 2012 AMA CPT Code Changes Dear Valued Client: The American Medical Association (AMA) has made Current Procedural Terminology (CPT) code changes to the 2012 edition of the CPT coding
January 1, 2012 Re: 2012 AMA CPT Code Changes Dear Valued Client: The American Medical Association (AMA) has made Current Procedural Terminology (CPT) code changes to the 2012 edition of the CPT coding
Client Announcement: February 28, 2018 Continuation of transitioning tests from Mayo to ARUP or internally at Fairview Effective Date
 HealthEast Medical Laboratory Client Announcement: February 28, 2018 Continuation of transitioning tests from Mayo to ARUP or internally at Fairview Effective Date 3-6-2018 As noted in previous communications,
HealthEast Medical Laboratory Client Announcement: February 28, 2018 Continuation of transitioning tests from Mayo to ARUP or internally at Fairview Effective Date 3-6-2018 As noted in previous communications,
Please note that our serum opiates EIA screen assay is not appropriate for therapeutic drug monitoring of Oxycodone.
 2211 Michigan Avenue Santa Monica, CA 90404 800 421 4449 Dear Colleague: October, 2004 Last month, in anticipation of cold and flu season, we introduced the Respiratory Virus DetectR (Hexaplex ) with Influenza
2211 Michigan Avenue Santa Monica, CA 90404 800 421 4449 Dear Colleague: October, 2004 Last month, in anticipation of cold and flu season, we introduced the Respiratory Virus DetectR (Hexaplex ) with Influenza
March Monthly Update, Quest Diagnostics Nichols Institute, Valencia
 NEW TESTS Please Note: Not all test codes assigned to each assay are listed in the table of contents. Please refer to the complete listing on the page numbers indicated. Test Code Test Name Effective Date
NEW TESTS Please Note: Not all test codes assigned to each assay are listed in the table of contents. Please refer to the complete listing on the page numbers indicated. Test Code Test Name Effective Date
H. PYLORI AB, IGG HPYL
 H. PYLORI AB, IGG HPYL Specimen Required: 3 ml blood, Serum gel tube Analytical Time: 1 day CPT Code: 86677 HAPTOGLOBIN HAP Specimen Required: 1.5 ml blood, gel tube Reference Range: 30-200 mg/dl CPT Code:
H. PYLORI AB, IGG HPYL Specimen Required: 3 ml blood, Serum gel tube Analytical Time: 1 day CPT Code: 86677 HAPTOGLOBIN HAP Specimen Required: 1.5 ml blood, gel tube Reference Range: 30-200 mg/dl CPT Code:
Case History Number One. Case History Two. Selection and Preparation of Allergen Vaccines (Extracts) Single Allergen Associated With Allergic Disease
 Selection and Preparation of Allergen Vaccines (Extracts) Richard F. Lockey, M.D. Division of Allergy and Immunology Department of Internal Medicine University of South Florida College of Medicine and
Selection and Preparation of Allergen Vaccines (Extracts) Richard F. Lockey, M.D. Division of Allergy and Immunology Department of Internal Medicine University of South Florida College of Medicine and
Allergen, Drugs, Amoxicillin IgE IGES1 Allergen, Drugs, Ampicillin IGES1 Allergen, Drugs, Penicillin G (major) IGES1
 Allergen, Drugs, Amoxicillin IgE 0055421 IGES1 Allergen, Drugs, Ampicillin 0055422 IGES1 Allergen, Drugs, Penicillin G (major) 0099628 IGES1 Allergen, Drugs, Penicillin V (minor) 0099629 IGES1 Allergen,
Allergen, Drugs, Amoxicillin IgE 0055421 IGES1 Allergen, Drugs, Ampicillin 0055422 IGES1 Allergen, Drugs, Penicillin G (major) 0099628 IGES1 Allergen, Drugs, Penicillin V (minor) 0099629 IGES1 Allergen,
REAGENTS AND MATERIALS This test kit contains sufficient wells and reagents to assay the serum of 3 patients for antibodies to 90 different foods.
 INTENDED USE This kit is for measuring the relative amount of food-specific IgG antibody in human serum. The values obtained must always be correlated with the clinical presentation, since elevation of
INTENDED USE This kit is for measuring the relative amount of food-specific IgG antibody in human serum. The values obtained must always be correlated with the clinical presentation, since elevation of
Within 14 days at 2-8⁰C. DO NOT FREEZE WHOLE BLOOD Micro Typing Systems 50 µl plasma
 Test Name *Minimum Collection Tubes Sample Stability ABO/Rh Blood Typing 50 µl cells EDTA whole blood (lavender-top) Within 14 days at 2-8⁰C. DO NOT FREEZE WHOLE BLOOD Micro Typing Systems 50 µl plasma
Test Name *Minimum Collection Tubes Sample Stability ABO/Rh Blood Typing 50 µl cells EDTA whole blood (lavender-top) Within 14 days at 2-8⁰C. DO NOT FREEZE WHOLE BLOOD Micro Typing Systems 50 µl plasma
Human Allergen Specific IgE ELISA Kits
 Intended Use Human Allergen Specific IgE ELISA Kits These kits are in vitro assays for the qualitative or quantitative detection of allergen-specific IgE antibodies in human serum. They are intended for
Intended Use Human Allergen Specific IgE ELISA Kits These kits are in vitro assays for the qualitative or quantitative detection of allergen-specific IgE antibodies in human serum. They are intended for
5701 Aerobic Stool Culture, Routine Component Method Reference Range. Organism/Pathogen CULTURE Not detected. Echinococcus IgG EIA Not detected
 2211 Michigan Avenue Santa Monica, CA 90404 800-421-4449 Coming SOON Coronavirus (SARS) RNA by PCR Specialty has a TaqMan assay for the detection of the SARS coronavirus directly from patient respiratory
2211 Michigan Avenue Santa Monica, CA 90404 800-421-4449 Coming SOON Coronavirus (SARS) RNA by PCR Specialty has a TaqMan assay for the detection of the SARS coronavirus directly from patient respiratory
March Immunoglobulins, Total (IgA, IgE, IgG, IgM) Interleukin-6 (IL6) Protein Electrophoresis, CSF. Thyrotropin Receptor Antibody
 University of Washington March 2012 TEST CHANGE Alpha 1-Antitrypsin Anti-IgE Receptor Antibody Anti-Mullerian Hormone Aquaporin-4 Receptor Antibody ColoSeq - Polyposis Complement testing, C3, C4 Everolimus
University of Washington March 2012 TEST CHANGE Alpha 1-Antitrypsin Anti-IgE Receptor Antibody Anti-Mullerian Hormone Aquaporin-4 Receptor Antibody ColoSeq - Polyposis Complement testing, C3, C4 Everolimus
TEST REQUEST INFORMATION- VIROLOGY
 Chlamydia/ Gonorrhea Nucleic Acid Amplification VC75 Qualitative Nucleic Acid Amplification SPECIMEN: Genital swab or first catch urine CONTAINER:GEN-PROBE APTIMA 2 Combo swab transport tube or urine transport
Chlamydia/ Gonorrhea Nucleic Acid Amplification VC75 Qualitative Nucleic Acid Amplification SPECIMEN: Genital swab or first catch urine CONTAINER:GEN-PROBE APTIMA 2 Combo swab transport tube or urine transport
June Monthly Update, Quest Diagnostics Nichols Institute, Valencia
 June 2013 - Monthly Update, NEW TESTS Please Note: Not all test codes assigned to each assay are listed in the table of contents. Please refer to the complete listing on the page numbers indicated. Test
June 2013 - Monthly Update, NEW TESTS Please Note: Not all test codes assigned to each assay are listed in the table of contents. Please refer to the complete listing on the page numbers indicated. Test
Allergy Seasons. weather and other environmental factors.
 3 Allergy Seasons American Academy of Allergy & Asthma Just like a weather report, an allergy report is a daily ritual across the country. But, if you think an allergy report is limited to certain times
3 Allergy Seasons American Academy of Allergy & Asthma Just like a weather report, an allergy report is a daily ritual across the country. But, if you think an allergy report is limited to certain times
2018 HIV and HCV Diagnostic Testing Survey
 2018 HIV and HCV Diagnostic Testing Survey This survey is designed to capture the 2017 HIV and HCV testing practices in state and local public health laboratories (PHL). The results of the survey will
2018 HIV and HCV Diagnostic Testing Survey This survey is designed to capture the 2017 HIV and HCV testing practices in state and local public health laboratories (PHL). The results of the survey will
588G: Dietary Antigen Testing: Sensitivity and Complement 1/5. Dietary Antigen Exposure by Food Group
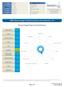 PATIENT NAME: CLINIC: DOB: SAMPLE DATE: RECEIVE DATE: REPORT DATE: R //2 /28/27 /29/27 /3/27 Dunwoody Labs 9 Dunwoody Park Suite 2 Dunwoody, GA 3338 USA Phone: 6787366374 Fax: 776747 Nine Dunwoody Park,
PATIENT NAME: CLINIC: DOB: SAMPLE DATE: RECEIVE DATE: REPORT DATE: R //2 /28/27 /29/27 /3/27 Dunwoody Labs 9 Dunwoody Park Suite 2 Dunwoody, GA 3338 USA Phone: 6787366374 Fax: 776747 Nine Dunwoody Park,
My Plate Healthy Eating 1
 My Plate Healthy Eating 1 Learn more about healthy eating! Go to www.choosemyplate.gov Finding out how many calories YOU need for a day is a first step in managing your weight. Exercise is the key to any
My Plate Healthy Eating 1 Learn more about healthy eating! Go to www.choosemyplate.gov Finding out how many calories YOU need for a day is a first step in managing your weight. Exercise is the key to any
Allergy Testing ALLERGY TESTING. 0.3 ml Serum, Red-Top Tube, no additive (Preferred), Serum separator tube (Acceptable) PER ALLERGEN; Minimum 0.
 ALLERGY TESTING Specimen Requirements: 0.3 ml Serum, Red-Top Tube, no additive (Preferred), Serum separator tube (Acceptable) PER ALLERGEN; Minimum 0.15 ml Transport temperature: Room temperature Stabilities:
ALLERGY TESTING Specimen Requirements: 0.3 ml Serum, Red-Top Tube, no additive (Preferred), Serum separator tube (Acceptable) PER ALLERGEN; Minimum 0.15 ml Transport temperature: Room temperature Stabilities:
Milan Analytica AG Baslerstrasse Rheinfelden
 Milan Analytica AG Baslerstrasse 15 4310 Rheinfelden www.milananalytica.ch Product list valid from 01.09.2012 Please ask for evaluation samples under Product Article number Circulating immune complexes
Milan Analytica AG Baslerstrasse 15 4310 Rheinfelden www.milananalytica.ch Product list valid from 01.09.2012 Please ask for evaluation samples under Product Article number Circulating immune complexes
THE COMPLETE ALLERGY SERVICE ALLERGIES & YOUR PET IMPROVE YOUR PET S QUALITY OF LIFE INNOVATION IN ANIMAL HEALTHCARE
 THE COMPLETE ALLERGY SERVICE ALLERGIES & YOUR PET IMPROVE YOUR PET S QUALITY OF LIFE INNOVATION IN ANIMAL HEALTHCARE WHAT IS AN ALLERGY? An allergy is a reaction of the immune system to a common substance
THE COMPLETE ALLERGY SERVICE ALLERGIES & YOUR PET IMPROVE YOUR PET S QUALITY OF LIFE INNOVATION IN ANIMAL HEALTHCARE WHAT IS AN ALLERGY? An allergy is a reaction of the immune system to a common substance
October Monthly Update, Quest Diagnostics Nichols Institute, Valencia
 ANNOUNCEMENTS Please Note: Not all test codes assigned to each assay are listed in the table of contents. Please refer to the complete listing on the page numbers indicated. Test Name Page # Reactivated
ANNOUNCEMENTS Please Note: Not all test codes assigned to each assay are listed in the table of contents. Please refer to the complete listing on the page numbers indicated. Test Name Page # Reactivated
588-Complete Dietary Antigen Testing
 18716_REPORT_COMPETE DUNWOODY ABS 9 Dunwoody Park, Suite 121 Dunwoody, GA 3338 P: 678-736-6374 F: 77-674-171 Email: info@dunwoodylabs.com www.dunwoodylabs.com PATIENT INFO NAME: SAMPE PATIENT REQUISITION
18716_REPORT_COMPETE DUNWOODY ABS 9 Dunwoody Park, Suite 121 Dunwoody, GA 3338 P: 678-736-6374 F: 77-674-171 Email: info@dunwoodylabs.com www.dunwoodylabs.com PATIENT INFO NAME: SAMPE PATIENT REQUISITION
Test Definition: FMCPS Meningoencephalitis Comprehensive Panel (Serum)
 Reporting Title: Meningoencephalitis Comp Panel, S Performing Location: Focus Diagnostics, Specimen Requirements: Draw blood in a plain, red-top tube(s). (Serum gel tube is acceptable.) Spin down and send
Reporting Title: Meningoencephalitis Comp Panel, S Performing Location: Focus Diagnostics, Specimen Requirements: Draw blood in a plain, red-top tube(s). (Serum gel tube is acceptable.) Spin down and send
IgG Food Antibody Assessment (Serum)
 IgG Food Antibody Assessment (Serum) Patient: DOB: Sex: MRN: Order Number: Completed: Received: Collected: IgG Food Antibody Results Dairy Vegetables Fish/Shellfish Nuts and Grains Casein Cheddar cheese
IgG Food Antibody Assessment (Serum) Patient: DOB: Sex: MRN: Order Number: Completed: Received: Collected: IgG Food Antibody Results Dairy Vegetables Fish/Shellfish Nuts and Grains Casein Cheddar cheese
COMPLIANCE. Individual Highlights: Advance Beneficiary Notices Medical Necessity IL Requisition Reflex Test List Panel Test. September 2018.
 September 2018 COMPLIANCE Dear Physician: Inova Laboratories (IL) is proud to serve the Northern Virginia community as the only full-service reference laboratory. Each year we disclose information about
September 2018 COMPLIANCE Dear Physician: Inova Laboratories (IL) is proud to serve the Northern Virginia community as the only full-service reference laboratory. Each year we disclose information about
FDA Reentry Guidance
 FDA Reentry Guidance T. cruzi - Chagas, Hepatitis C and HIV Wednesday 6/6/18 Doug Denyer O Dina Hurlburt, SBB(ASCP)CM Outline Impact to Clients Review of FDA Guidance Documents Reentry Request Process
FDA Reentry Guidance T. cruzi - Chagas, Hepatitis C and HIV Wednesday 6/6/18 Doug Denyer O Dina Hurlburt, SBB(ASCP)CM Outline Impact to Clients Review of FDA Guidance Documents Reentry Request Process
November Monthly Update, Quest Diagnostics Nichols Institute, Valencia
 NEW TESTS Please Note: Not all test codes assigned to each assay are listed in the table of contents. Please refer to the complete listing on the page numbers indicated. Test Code Test Name Effective Date
NEW TESTS Please Note: Not all test codes assigned to each assay are listed in the table of contents. Please refer to the complete listing on the page numbers indicated. Test Code Test Name Effective Date
THE STAR
 THE STAR 210-521-6886 Fall Allergies The beginning of fall means the return of cooler weather. From our favorite pumpkin-spiced latte, to the changing color of the leaves there are tons of things to love
THE STAR 210-521-6886 Fall Allergies The beginning of fall means the return of cooler weather. From our favorite pumpkin-spiced latte, to the changing color of the leaves there are tons of things to love
Allergology product list 2018
 Milan Analytica AG Baslerstrasse 15 4310 Rheinfelden Switzerland www.milananalytica.ch Allergology product list 2018 Please ask for more information under Product Article number Circulating immune complexes
Milan Analytica AG Baslerstrasse 15 4310 Rheinfelden Switzerland www.milananalytica.ch Allergology product list 2018 Please ask for more information under Product Article number Circulating immune complexes
Medizinische Labordiagnostika AG. EUROIMMUN EUROLINE profiles for allergy diagnostics Europe. Indicator and CCD. Sweet vernal grass Orchard grass
 EUROIMMUN Europe Food Indicator and Allergy profile Allergens in groups e6 e82 es4 Guinea pig Rabbit Cage bird mix 1 g1 g3 Sweet vernal grass Orchard grass marker Inhalation 2 (Order no. DP 3110-1601-1
EUROIMMUN Europe Food Indicator and Allergy profile Allergens in groups e6 e82 es4 Guinea pig Rabbit Cage bird mix 1 g1 g3 Sweet vernal grass Orchard grass marker Inhalation 2 (Order no. DP 3110-1601-1
 27027 Tourney Road Valencia, CA 91355 800 421 7110 www.specialtylabs.com Test Updates November 25, 2010 Dear Colleague: This client letter contains many changes to our reference ranges in the area of chemistry
27027 Tourney Road Valencia, CA 91355 800 421 7110 www.specialtylabs.com Test Updates November 25, 2010 Dear Colleague: This client letter contains many changes to our reference ranges in the area of chemistry
IMMEDIATE HOT LINE: Effective January 6, 2014
 MEDICARE COVERAGE OF LABORATORY TESTING Please remember when ordering laboratory tests that are billed to Medicare/Medicaid or other federally funded programs, the following requirements apply: 1. Only
MEDICARE COVERAGE OF LABORATORY TESTING Please remember when ordering laboratory tests that are billed to Medicare/Medicaid or other federally funded programs, the following requirements apply: 1. Only
LAWS, STANDARDS & REGULATIONS
 Appendixes APPENDIX 1: LAWS, STANDARDS & REGULATIONS The United States enjoys one of the safest food supplies in the world. The laws and regulations required to achieve that safe food supply are lengthy
Appendixes APPENDIX 1: LAWS, STANDARDS & REGULATIONS The United States enjoys one of the safest food supplies in the world. The laws and regulations required to achieve that safe food supply are lengthy
It is believed that a meal plan that includes low FODMAPs also may help ease symptoms from other health conditions, such as:
 If you have digestive issues, this could be why. You don't have to have IBS (Irritable Bowel Syndrome) or Colitis (a chronically inflammed colon), to experience some of these symptoms. FODMAP The acronym
If you have digestive issues, this could be why. You don't have to have IBS (Irritable Bowel Syndrome) or Colitis (a chronically inflammed colon), to experience some of these symptoms. FODMAP The acronym
You have to eat to lose fat!
 Guide to Losing Fat HASfit's healthy meal was made for you. You need an easy to use system to eat healthy and get lean, but not feel starved all day. We designed this system to be easy for you to integrate
Guide to Losing Fat HASfit's healthy meal was made for you. You need an easy to use system to eat healthy and get lean, but not feel starved all day. We designed this system to be easy for you to integrate
March Monthly Update, Quest Diagnostics Nichols Institute, Valencia
 NEW TESTS Please Note: Not all test codes assigned to each assay are listed in the table of contents. Please refer to the complete listing on the page numbers indicated. Test Name Effective Date Page #
NEW TESTS Please Note: Not all test codes assigned to each assay are listed in the table of contents. Please refer to the complete listing on the page numbers indicated. Test Name Effective Date Page #
Potassium and Your CKD Diet
 A TO Z HEALTH GUIDE Potassium and Your CKD Diet What is potassium and why is it important to you? Potassium is a mineral found in many of the foods you eat. It plays a role in keeping your heartbeat regular
A TO Z HEALTH GUIDE Potassium and Your CKD Diet What is potassium and why is it important to you? Potassium is a mineral found in many of the foods you eat. It plays a role in keeping your heartbeat regular
October Courier Service on Holidays There will be no courier service Thursday, Dec. 24th, 2011.
 University of Washington October 2011 TEST CHANGE ACTH Aldosterone BCR/ABL by PCR, Qualitative Chlamydia trachomatis & N.gonorrhoeae, Amp. NA Creutzfeldt-Jakob, CSF Desipramine Doxepine and Nordoxepine,
University of Washington October 2011 TEST CHANGE ACTH Aldosterone BCR/ABL by PCR, Qualitative Chlamydia trachomatis & N.gonorrhoeae, Amp. NA Creutzfeldt-Jakob, CSF Desipramine Doxepine and Nordoxepine,
LAST NAME FIRST NAME MIDDLE NAME DATE OF BIRTH GENDER PHYSICIAN ID. TESTNAME PATIENT Female For doctor's reference
 FINAL REPORT DATE: ACCESSION ID: 05-04-2017 16:24 SPECIMEN COLLECTED: 11-03-2016 1611040000 SPECIMEN RECEIVED: 11-04-2016 00:00 LAST NAME FIRST NAME MIDDLE NAME DATE OF BIRTH GENDER PHYSICIAN ID TESTNAME
FINAL REPORT DATE: ACCESSION ID: 05-04-2017 16:24 SPECIMEN COLLECTED: 11-03-2016 1611040000 SPECIMEN RECEIVED: 11-04-2016 00:00 LAST NAME FIRST NAME MIDDLE NAME DATE OF BIRTH GENDER PHYSICIAN ID TESTNAME
My Diabetic Meal Plan during Pregnancy
 My Diabetic Meal Plan during Pregnancy When you have diabetes and are pregnant, you need to eat small meals and s throughout the day to help control your blood sugar. This also helps you get in enough
My Diabetic Meal Plan during Pregnancy When you have diabetes and are pregnant, you need to eat small meals and s throughout the day to help control your blood sugar. This also helps you get in enough
MYFITNESSBUDDY. Healthy Living Guide Part 1 ENTER CLIENT
 MYFITNESSBUDDY Healthy Living Guide Part 1 ENTER CLIENT 2012 NEW YEAR RESOLUTION COMPANY NAME HERE Happy New Year! 2012 is here and if you are reading this You have made the decision to embark on Healthy
MYFITNESSBUDDY Healthy Living Guide Part 1 ENTER CLIENT 2012 NEW YEAR RESOLUTION COMPANY NAME HERE Happy New Year! 2012 is here and if you are reading this You have made the decision to embark on Healthy
New Test ANNOUNCEMENT
 March 2003 W New Test ANNOUNCEMENT A Mayo Reference Services Publication Pediatric Allergy Screen
March 2003 W New Test ANNOUNCEMENT A Mayo Reference Services Publication Pediatric Allergy Screen
Business Unit: Quest Diagnostics Nichols Institute, Valencia. June 2011 Update Dear Colleague,
 Volume 2 Apr/2011 Business Unit: June 2011 Update Dear Colleague, This letter begins with a thank you for your very prompt response to our recent request to visit our website and update your contact information.
Volume 2 Apr/2011 Business Unit: June 2011 Update Dear Colleague, This letter begins with a thank you for your very prompt response to our recent request to visit our website and update your contact information.
Table 1: Dietary Sources of Omega-3 and Omega-3 Fatty Acids. Fatty Acid. Food Sources. (i) Omega-6 Types. LA, linoleic acid (18:2 n-6)
 As for saturated and monounsaturated fatty acids, the omega-6 and omega-3 polyunsaturated fatty acids (PUFA) are chemically linked to fat structures known as triglycerides in the various foods and oils
As for saturated and monounsaturated fatty acids, the omega-6 and omega-3 polyunsaturated fatty acids (PUFA) are chemically linked to fat structures known as triglycerides in the various foods and oils
The pages that follow contain information critical to protecting the health of your patients and the citizens of Colorado.
 Health Alert Network Tri-County Health Department Serving Adams, Arapahoe and Douglas Counties Phone 303/220-9200 Fax 303/741-4173 www.tchd.org Follow us on Twitter @TCHDHealth and @TCHDEmergency John
Health Alert Network Tri-County Health Department Serving Adams, Arapahoe and Douglas Counties Phone 303/220-9200 Fax 303/741-4173 www.tchd.org Follow us on Twitter @TCHDHealth and @TCHDEmergency John
Interpath Laboratory, Inc. Test File Update
 As an Interpath customer who receives electronic results or sends electronic orders you may need to be notified when we update our Service Manual. Although we try to keep these changes to a minimum, laboratory
As an Interpath customer who receives electronic results or sends electronic orders you may need to be notified when we update our Service Manual. Although we try to keep these changes to a minimum, laboratory
Interpath Laboratory, Inc. Test File Update
 As an Interpath customer who receives electronic results or sends electronic orders you may need to be notified when we update our Service Manual. Although we try to keep these changes to a minimum, laboratory
As an Interpath customer who receives electronic results or sends electronic orders you may need to be notified when we update our Service Manual. Although we try to keep these changes to a minimum, laboratory
Review the following alphabetical listing of our lab test catalog for each of our offered tests. Each page has at least the following elements:
 Site: Fremont Rideout Health Group Laboratory Services Policy and Procedure Creation Date: 08/17/2009 Subject/Title: FRHG Laboratories' Catalog of Tests Q-S Document Owner: Corson, Karen Pagination: Page
Site: Fremont Rideout Health Group Laboratory Services Policy and Procedure Creation Date: 08/17/2009 Subject/Title: FRHG Laboratories' Catalog of Tests Q-S Document Owner: Corson, Karen Pagination: Page
Allergy Testing and Allergy Immunotherapy
 All Panels and individual tests below are billed by CPT Code 86003 or contain a component that bills 86003 Test Name Allergic Bronchopulmonary Aspergillosis Panel II Allergy Panel 1 Allergy Panel 2 Allergy
All Panels and individual tests below are billed by CPT Code 86003 or contain a component that bills 86003 Test Name Allergic Bronchopulmonary Aspergillosis Panel II Allergy Panel 1 Allergy Panel 2 Allergy
Documentation, Codebook, and Frequencies
 2 Documentation, Codebook, and Frequencies Laboratory Component: Hepatitis B: core antibody, surface antibody and surface antigen; Hepatitis C: confirmed antibody; Hepatitis D antibody Survey Years: 2003
2 Documentation, Codebook, and Frequencies Laboratory Component: Hepatitis B: core antibody, surface antibody and surface antigen; Hepatitis C: confirmed antibody; Hepatitis D antibody Survey Years: 2003
January Billing and compliance. Chemistry. Help us help you. Referral testing. Additional CPT code changes announced for 2015
 January 2015 Billing and compliance Additional CPT code changes announced for 2015 Chemistry CA 27.29 conversion to CA 15-3 nearing completion Vitamin B12 and LD, Total specimen change Timed Urine Protein
January 2015 Billing and compliance Additional CPT code changes announced for 2015 Chemistry CA 27.29 conversion to CA 15-3 nearing completion Vitamin B12 and LD, Total specimen change Timed Urine Protein
Prevalence of Fungal Allergy in Patients with Allergic Rhinosinusitis
 Prevalence of Fungal Allergy in Patients with Allergic Rhinosinusitis Kyle Kennedy, M.D. Karen Calhoun,, M.D. June 5, 1999 Hypothesis Subset of patients with signs and symptoms of allergic rhinosinusitis
Prevalence of Fungal Allergy in Patients with Allergic Rhinosinusitis Kyle Kennedy, M.D. Karen Calhoun,, M.D. June 5, 1999 Hypothesis Subset of patients with signs and symptoms of allergic rhinosinusitis
INTERPATH LABORATORY, INC. TEST UPDATES
 EFFECTIVE DATE: February 16, 2016 INTERPATH LABORATORY, INC. TEST UPDATES Please take note of the following test updates and make the appropriate changes in your Interpath Service Manual. Feel free to
EFFECTIVE DATE: February 16, 2016 INTERPATH LABORATORY, INC. TEST UPDATES Please take note of the following test updates and make the appropriate changes in your Interpath Service Manual. Feel free to
March Molecular diagnostics. Update distribution changes. Billing and compliance. Hematology. Immunology. Referral testing
 March 2016 Update distribution changes Billing and compliance Compliance Drug Analysis CPT change Hematology Hemoglobin A1c, Screening Reference Range Change Immunology Testing schedule changes Vitamin
March 2016 Update distribution changes Billing and compliance Compliance Drug Analysis CPT change Hematology Hemoglobin A1c, Screening Reference Range Change Immunology Testing schedule changes Vitamin
HML Update. Summer 2009 Volume 15, No. 3. Keeping Our Clients Informed Questions or Comments: Call
 HML Update Summer 2009 Volume 15, No. 3 Keeping Our Clients Informed Questions or Comments: Call 651-232-3500 Inside: Panel Changes...1-2 New Panels...2-3 H1N1... 3 Note of Appreciation... 3 BD Vacutainer
HML Update Summer 2009 Volume 15, No. 3 Keeping Our Clients Informed Questions or Comments: Call 651-232-3500 Inside: Panel Changes...1-2 New Panels...2-3 H1N1... 3 Note of Appreciation... 3 BD Vacutainer
ASSAYS TO SUPPORT. HIV Treatment Decisions
 ASSAYS TO SUPPORT HIV Treatment Decisions Human Immunodeficiency Virus (HIV) HIV and AIDS remain a persistent problem for the United States and countries around the world. Efforts to identify HIV-positive
ASSAYS TO SUPPORT HIV Treatment Decisions Human Immunodeficiency Virus (HIV) HIV and AIDS remain a persistent problem for the United States and countries around the world. Efforts to identify HIV-positive
10/7/ Immediate Action, Quest Diagnostics Nichols Institute, Valencia
 REDIRECTS Please Not all test codes assigned to each assay are listed in the table of contents. Please refer to the complete listing on the page numbers indicated. Test Code Test Name Effective Date Page
REDIRECTS Please Not all test codes assigned to each assay are listed in the table of contents. Please refer to the complete listing on the page numbers indicated. Test Code Test Name Effective Date Page
Maximizing Cornea and Tissue Donation through Specimen Quality
 Maximizing Cornea and Tissue Donation through Specimen Quality Robert W. Bresler, Sydney D. Gastreich, Elias G. Koulouriotis, Linda S. Martin, Susan Diane Brockmeier, Chak-Sum Ho, PhD Abstract Purpose:
Maximizing Cornea and Tissue Donation through Specimen Quality Robert W. Bresler, Sydney D. Gastreich, Elias G. Koulouriotis, Linda S. Martin, Susan Diane Brockmeier, Chak-Sum Ho, PhD Abstract Purpose:
Acetylcholine Receptor Binding Antibody
 Acetylcholine Receptor Binding Antibody Order Name: ACETY BND Test Number: 5500010 REV DATE:9/17/2008 Acetylcholine Receptor Binding Antibody RIA Preferred 1 ml (0.5) Serum Clot Activator (Red Top, No-Gel)
Acetylcholine Receptor Binding Antibody Order Name: ACETY BND Test Number: 5500010 REV DATE:9/17/2008 Acetylcholine Receptor Binding Antibody RIA Preferred 1 ml (0.5) Serum Clot Activator (Red Top, No-Gel)
May Monthly Update, Quest Diagnostics Nichols Institute, Valencia
 NEW TESTS Please Note: Not all test codes assigned to each assay are listed in the table of contents. Please refer to the complete listing on the page numbers indicated. Test Code Test Name Effective Date
NEW TESTS Please Note: Not all test codes assigned to each assay are listed in the table of contents. Please refer to the complete listing on the page numbers indicated. Test Code Test Name Effective Date
TO BE RESCINDED 2
 ACTION: Original DATE: 12/03/2015 1:06 PM 5160-4-19 Allergy services. TO BE RESCINDED (A) Allergy testing. (1) Providers of physician services may be reimbursed for the performance and evaluation of allergy
ACTION: Original DATE: 12/03/2015 1:06 PM 5160-4-19 Allergy services. TO BE RESCINDED (A) Allergy testing. (1) Providers of physician services may be reimbursed for the performance and evaluation of allergy
Integrating HIV Screening Into
 MaxiMizing Third ParTy reimbursement for hiv TesTing Integrating HIV Screening Into Title X Services IntroductIon HIV screening services are a core family planning service, and all individuals aged 13-64
MaxiMizing Third ParTy reimbursement for hiv TesTing Integrating HIV Screening Into Title X Services IntroductIon HIV screening services are a core family planning service, and all individuals aged 13-64
Technical Bulletin No. 104b
 CPAL Central Pennsylvania Alliance Laboratory Technical Bulletin No. 104b June 22, 2016 Update to Guidelines for Diagnosing HIV Infection CPAL s Testing Algorithm for Diagnostic HIV Testing HIV Geenius
CPAL Central Pennsylvania Alliance Laboratory Technical Bulletin No. 104b June 22, 2016 Update to Guidelines for Diagnosing HIV Infection CPAL s Testing Algorithm for Diagnostic HIV Testing HIV Geenius
25* or higher Underweight. 240 mg/dl and above High (More than twice the risk as desirable level.) OK, but higher is better
 Biometrics Screening Biometric Health Indicators The charts below provide a summary of the ranges for each of the biometric screening tests. Be sure to check with your doctor if your results are outside
Biometrics Screening Biometric Health Indicators The charts below provide a summary of the ranges for each of the biometric screening tests. Be sure to check with your doctor if your results are outside
CDIA TM Rubella IgG/IgM Rapid Test Kit
 CDIA TM Rubella IgG/IgM Rapid Test Kit Cat.No: DTSJZ024 Lot. No. (See product label) Intended Use The CDIA TM Rubella IgG/IgM Rapid Test Kit is a rapid chromatographic immunoassay for the qualitative detection
CDIA TM Rubella IgG/IgM Rapid Test Kit Cat.No: DTSJZ024 Lot. No. (See product label) Intended Use The CDIA TM Rubella IgG/IgM Rapid Test Kit is a rapid chromatographic immunoassay for the qualitative detection
Low FODMAP Diet. OK, but what are FODMAPs and who should avoid them?
 Low FODMAP Diet FODMAP? What does that stand for? Fermentable Oligosaccharides (oligo few, saccharide sugar ) Disaccharides ( two sugars ) Monosaccharides ( one sugar ) And Polyols (these are sugar alcohols)
Low FODMAP Diet FODMAP? What does that stand for? Fermentable Oligosaccharides (oligo few, saccharide sugar ) Disaccharides ( two sugars ) Monosaccharides ( one sugar ) And Polyols (these are sugar alcohols)
Aerobic Wound Culture and Stain
 Aerobic Wound Culture and Stain Order Name: C WOUN RTS Test Number: 6000153 Revision Date: 03/27/2014 Aerobic Wound Culture and Stain Culture Preferred 1 ml Tissue Sterile Screwtop Container Room Temperature
Aerobic Wound Culture and Stain Order Name: C WOUN RTS Test Number: 6000153 Revision Date: 03/27/2014 Aerobic Wound Culture and Stain Culture Preferred 1 ml Tissue Sterile Screwtop Container Room Temperature
Case Studies, or Verification Vignettes
 Case Studies, or Verification Vignettes 1 Vignette #1 Change from One Automated AST to Another Your lab is changing from one FDA-cleared automated AST to another Is a verification study required? 2 Yes
Case Studies, or Verification Vignettes 1 Vignette #1 Change from One Automated AST to Another Your lab is changing from one FDA-cleared automated AST to another Is a verification study required? 2 Yes
Adventures in Discordance- HIV Testing
 Anne Gaynor, PhD On behalf of Monica M. Parker, PhD Director, Bloodborne Viruses Laboratory Wadsworth Center, NYSDOH Adventures in Discordance- HIV Testing HIV Laboratory Testing Algorithm HIV-1/2 antigen/antibody
Anne Gaynor, PhD On behalf of Monica M. Parker, PhD Director, Bloodborne Viruses Laboratory Wadsworth Center, NYSDOH Adventures in Discordance- HIV Testing HIV Laboratory Testing Algorithm HIV-1/2 antigen/antibody
February Monthly Update, Quest Diagnostics Nichols Institute, Valencia
 NEW TESTS Please Note: Not all test codes assigned to each assay are listed in the table of contents. Please refer to the complete listing on the page numbers indicated. Test Code Test Name Effective Date
NEW TESTS Please Note: Not all test codes assigned to each assay are listed in the table of contents. Please refer to the complete listing on the page numbers indicated. Test Code Test Name Effective Date
Acetylcholine Receptor Modulating Antibody
 Acetylcholine Receptor Modulating Antibody Order Name: ACETY MOD Test Number: 5516500 TEST COMPONENTS REV DATE: 09/25/2012 Acetylcholine Receptor Modulating Antibody SEMI-FC Preferred 1 ml (0.5) Serum
Acetylcholine Receptor Modulating Antibody Order Name: ACETY MOD Test Number: 5516500 TEST COMPONENTS REV DATE: 09/25/2012 Acetylcholine Receptor Modulating Antibody SEMI-FC Preferred 1 ml (0.5) Serum
 27027 Tour ney Road Valencia, CA 91355 8 0 0 4 2 1 7 1 1 0 www.specialtylabs.com Test Updates December 25, 2010 Dear Colleague: Please review the following specific test listings for important information
27027 Tour ney Road Valencia, CA 91355 8 0 0 4 2 1 7 1 1 0 www.specialtylabs.com Test Updates December 25, 2010 Dear Colleague: Please review the following specific test listings for important information
Client will make 2 specific goals to decrease her potassium intake. Client will make 1 specific goal to decrease her fluid retention.
 Title: Renal Diet Target Audience: Renal Dialysis Patients LESSON PLAN Name: Heidi Washburn Terminal Objective Client will be able to plan an appropriate renal dialysis diet for him/herself. Domain Cognitive
Title: Renal Diet Target Audience: Renal Dialysis Patients LESSON PLAN Name: Heidi Washburn Terminal Objective Client will be able to plan an appropriate renal dialysis diet for him/herself. Domain Cognitive
6/11/15. BACTERIAL STDs IN A POST- HIV WORLD. Learning Objectives. How big a problem are STIs in the U.S.?
 BACTERIAL STDs IN A POST- HIV WORLD Tracey Graney, PhD, MT(ASCP) Monroe Community College Learning Objectives Describe the epidemiology and incidence of bacterial STDs in the U.S. Describe current detection
BACTERIAL STDs IN A POST- HIV WORLD Tracey Graney, PhD, MT(ASCP) Monroe Community College Learning Objectives Describe the epidemiology and incidence of bacterial STDs in the U.S. Describe current detection
 www.mydiabeticdiet.eu The Keto diet has exploded in popularity as an effective way of losing weight, balancing blood sugar and reducing insulin resistance for type 2 diabetics In fact, many people have
www.mydiabeticdiet.eu The Keto diet has exploded in popularity as an effective way of losing weight, balancing blood sugar and reducing insulin resistance for type 2 diabetics In fact, many people have
Sample Report Exclusively in the UK and Ireland Contact: Regenerus Laboratories T: +44 (0) E:
 Sample Report Exclusively in the UK and Ireland Contact: Regenerus Laboratories T: + () 7 87 E: info@regeneruslabs.com Patient Information Sheet: Name: Jon Doe Example Date Drawn: //6 Date of Birth: //966
Sample Report Exclusively in the UK and Ireland Contact: Regenerus Laboratories T: + () 7 87 E: info@regeneruslabs.com Patient Information Sheet: Name: Jon Doe Example Date Drawn: //6 Date of Birth: //966
The Alphabet Soup of Viral Hepatitis Testing
 The Alphabet Soup of Viral Hepatitis Testing August 18, 2011 Patricia Slev, PhD, DABCC Medical Director, Serologic Hepatitis and Retrovirus Laboratory, ARUP Laboratories Assistant Professor of Pathology,
The Alphabet Soup of Viral Hepatitis Testing August 18, 2011 Patricia Slev, PhD, DABCC Medical Director, Serologic Hepatitis and Retrovirus Laboratory, ARUP Laboratories Assistant Professor of Pathology,
Principles of the DASH Diet
 DASH Diet Lower your blood pressure by changing your eating habits. The DASH diet is based on findings from the "Dietary Approaches to Stop Hypertension" clinical study that found that high blood pressure
DASH Diet Lower your blood pressure by changing your eating habits. The DASH diet is based on findings from the "Dietary Approaches to Stop Hypertension" clinical study that found that high blood pressure
APHL/CDC HIV Demonstration Project Demonstration Project for HIV Nucleic Acid Testing (NAT) Referral Updated: January 2018
 APHL/CDC HIV Demonstration Project Demonstration Project for HIV Nucleic Acid Testing (NAT) Referral Updated: January 2018 Shipping and Submission Instructions Thank you for participating as a submitting
APHL/CDC HIV Demonstration Project Demonstration Project for HIV Nucleic Acid Testing (NAT) Referral Updated: January 2018 Shipping and Submission Instructions Thank you for participating as a submitting
Welcome to KPAUL Mastery Coaching. Personal Power Program. Kpaul Journal. Belongs To:
 Welcome to KPAUL Mastery Coaching Personal Power Program Kpaul Journal Belongs To: Copyright 2017 Kent Paul All Rights Reserved Kpaul Media LLC recommends using this Kpaul Journal. For more information
Welcome to KPAUL Mastery Coaching Personal Power Program Kpaul Journal Belongs To: Copyright 2017 Kent Paul All Rights Reserved Kpaul Media LLC recommends using this Kpaul Journal. For more information
HEALTH TIPS FOR THE MONTH OF OCTOBER 2017 HEALTHY EATING IS IN YOUR MIND Continuous
 HEALTH TIPS FOR THE MONTH OF OCTOBER 2017 HEALTHY EATING IS IN YOUR MIND Continuous 8. Add calcium for strong bones Calcium is one of the key nutrients that your body needs in order to stay strong and
HEALTH TIPS FOR THE MONTH OF OCTOBER 2017 HEALTHY EATING IS IN YOUR MIND Continuous 8. Add calcium for strong bones Calcium is one of the key nutrients that your body needs in order to stay strong and
Viral Hepatitis Diagnosis and Management
 Viral Hepatitis Diagnosis and Management CLINICAL BACKGROUND Viral hepatitis is a relatively common disease (25 per 100,000 individuals in the United States) caused by a diverse group of hepatotropic agents
Viral Hepatitis Diagnosis and Management CLINICAL BACKGROUND Viral hepatitis is a relatively common disease (25 per 100,000 individuals in the United States) caused by a diverse group of hepatotropic agents
Guidelines for the Laboratory Detection of Chlamydia trachomatis, Neisseria gonorrhoeae and Treponema pallidum Testing
 Guidelines for the Laboratory Detection of Chlamydia trachomatis, Neisseria gonorrhoeae and Treponema pallidum Testing Recommendations from the an expert consultation meeting held at CDC January 13-15,
Guidelines for the Laboratory Detection of Chlamydia trachomatis, Neisseria gonorrhoeae and Treponema pallidum Testing Recommendations from the an expert consultation meeting held at CDC January 13-15,
Dear Healthcare Provider, The information contained here may be very important to your practice. Please take a moment to review this document.
 February 2018 Dear Healthcare Provider, The information contained here may be very important to your practice. Please take a moment to review this document. TEST BULLETIN CHLAMYDIA/GONORRHEA SPECIMEN COLLECTION
February 2018 Dear Healthcare Provider, The information contained here may be very important to your practice. Please take a moment to review this document. TEST BULLETIN CHLAMYDIA/GONORRHEA SPECIMEN COLLECTION
