The Tumor Suppressor DAP Kinase Is a Target of RSK-Mediated Survival Signaling
|
|
|
- Rudolph Curtis
- 5 years ago
- Views:
Transcription
1 Current Biology, Vol. 15, , October 11, 2005, 2005 Elsevier Ltd All rights reserved. DOI /j.cub The Tumor Suppressor DAP Kinase Is a Target of RSK-Mediated Survival Signaling Rana Anjum, 1 Philippe P. Roux, 1 Bryan A. Ballif, 1,2 Steven P. Gygi, 1,2 and John Blenis 1, * 1 Department of Cell Biology 2 Taplin Biological Mass Spectrometry Facility Harvard Medical School Boston, Massachusetts Summary The viability of vertebrate cells depends on a complex signaling interplay between survival factors and celldeath effectors. Subtle changes in the equilibrium between these regulators can result in abnormal cell proliferation or cell death, leading to various pathological manifestations [1, 2]. Death-associated protein kinase (DAPK) is a multidomain calcium/calmodulin (CaM)-dependent Ser/Thr protein kinase with an important role in apoptosis regulation and tumor suppression [3 6]. The molecular signaling mechanisms regulating this kinase, however, remain unclear. Here, we show that DAPK is phosphorylated upon activation of the Ras-extracellular signal-regulated kinase (ERK) pathway [7]. This correlates with the suppression of the apoptotic activity of DAPK. We demonstrate that DAPK is a novel target of p90 ribosomal S6 kinases (RSK) 1 and 2, downstream effectors of ERK1/2 [8 11]. Using mass spectrometry, we identified Ser-289 as a novel phosphorylation site in DAPK, which is regulated by RSK. Mutation of Ser- 289 to alanine results in a DAPK mutant with enhanced apoptotic activity, whereas the phosphomimetic mutation (Ser289Glu) attenuates its apoptotic activity. Our results suggest that RSK-mediated phosphorylation of DAPK is a unique mechanism for suppressing the proapoptotic function of this death kinase in healthy cells as well as Ras/Raf-transformed cells. Results and Discussion In HEK293E cells, overexpression of DAPK results in extensive apoptotic cell death by 24 hr posttransfection (Figure 1A, compare bar 1 to bar 5). We sought to determine whether activation of cell-survival pathways in DAPK-expressing cells would provide protection against cell death [7, 12]. The Ras-MAPK (mitogen-activated protein kinase) pathway has been shown to promote cell-survival signaling and is the major mechanism for protection against cell death in certain cell types [7]. Interestingly, we found that treatment of cells with the phorbol ester PMA, which activates the Ras-MAPK pathway but not the PI3K-Akt/PKB pathway, antagonized the death-inducing activity of DAPK, leading to increased survival of DAPK-expressing cells (Figure 1A). In contrast, when cells were pretreated with the MEK1/2/5 inhibitor U0126, there was enhanced cell *Correspondence: john_blenis@hms.harvard.edu death and PMA did not attenuate the apoptotic effects of DAPK (Figure 1A). We considered the possibility that DAPK was inhibited upon phosphorylation by kinases downstream of MEK in this cascade. Because RSK is a potential cell-survival kinase in this cascade [7], we used an antibody that recognizes the consensus phosphorylation motif, RXRXXpS/T, which recognizes substrates of several basophilic kinases within the AGC family such as RSK, as well as Akt and ribosomal S6 kinase 1 (S6K1) [13 15]. Using this antibody, we found that PMA stimulated basophilic site phosphorylation of endogenous (see Figure S1A in the Supplemental Data available with this article online) and overexpressed DAPK (Figure 1B). Importantly, we also found that phosphorylation of DAPK, similar to the death-protecting effect of PMA, was completely inhibited by U0126. These results indicated that activation of the Ras-MAPK cascade induced MEK-dependent phosphorylation of DAPK, suggesting that this cascade may inhibit apoptosis by directly regulating DAPK phosphorylation. To further characterize the pathways leading to DAPK phosphorylation, we stimulated cells with different agonists and analyzed them for DAPK phosphorylation. Stimulation of cells with either PMA or EGF led to robust activation of the ERK-MAPK pathway and DAPK phosphorylation, whereas incubation of cells with insulin, a PI3-kinase agonist, or with anisomycin, a p38-mapk agonist, did not alter DAPK phosphorylation (Figure 1C). Consistent with these results, PMA-induced phosphorylation of DAPK was completely inhibited by U0126 and BIM (bisindolylmaleimide I), indicating that PMA-stimulated DAPK phosphorylation requires MEK and protein kinase C (PKC), respectively. Furthermore, treatment of cells with wortmannin or rapamycin did not alter DAPK phosphorylation, again demonstrating that the PI3-kinase and mtor pathways are not necessary for these effects (Figure S1B). These data suggests that basophilic kinases regulated by the Ras- MAPK pathway are likely involved in DAPK phosphorylation stimulated by PMA and exclude the involvement of other basophilic kinases including Akt, MSK, and S6K. To further establish the link between Ras signaling and DAPK phosphorylation, we transfected activated versions of Ras (Ras61L), B-Raf (ER- B-Raf), and MEK (MEK-DD) in cells overexpressing DAPK [16, 17]. These oncogenic signaling proteins potently stimulated growth-factor- and phorbol-ester-independent phosphorylation of DAPK (Figures 1D and 1E), suggesting that activation of the Ras-MAPK pathway is sufficient to induce DAPK phosphorylation. Finally, we also observed a reduction in DAPK phosphorylation by PMA by partially knocking down endogenous ERK1/2 (Figure S1C). The Ras-MAPK pathway activates the RSK family of proteins in response to growth factors and phorbol esters [8]. Activation of RSK coincides with oncogenic transformation [18], stimulation of G0/G1 transition [19 21], and differentiation of PC12 cells [10]. To determine whether RSK could directly phosphorylate DAPK, we
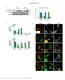
2 DAP Kinase Is a Target of RSK 1763 Figure 1. Inhibition of Cell Death and Phosphorylation of DAPK by the Activation of Ras-MAPK Pathway (A) HEK293E cells were transfected with either the vector or Flag-DAPK construct, along with EGFP plasmid (10:1 ratio), serum-starved 10 hr after transfection, and treated with DMSO alone or PMA (50 ng/ml) with or without U0126 (10 M) or UO126 alone. After 24 hr, cells were fixed and stained with activated anti-caspase-3 antibody. To determine the percent of apoptotic cells, we scored 100 GFP-positive cells in six different fields for apoptotis by counting cells positive for caspase-3 staining. All experiments were repeated at least three times, and the results are represented as means ± standard error. (B) HEK293E cells, transfected with either the vector or Flag-DAPK, were serum-starved for 18 hr and then stimulated with 50 ng/ml PMA for further 15 min, either alone or after pretreatment of cells with U0126 (10 M) for 30 min. The lysates were immunoprecipitated with anti-flag antibody, and DAPK phosphorylation on RXRXXS/T motif was analyzed after immunoblotting with Akt-pSub antibody [α-(p)dapk]. The cell lysates were also immunoblotted for total protein levels (α-flag) and ERK1/2 phosphorylation. (C) DAPK-transfected cells were serum-starved and stimulated with either 50 ng/ml PMA (15 min), 50 ng/ml EGF (15 min), 100 nm insulin (30 min), or 2 M anisomycin (30 min). DAPK was immunoprecipitated from the cell lysates and immunoblotted as in (B), for DAPK phosphorylation, total DAPK levels (α-flag), Akt phosphorylation [α-(p)akt (S473)], (P)-p38, and ERK1/2. (D) HEK293E cells were transfected with DAPK and cotransfected with vector, MEK-DD, or activated Ras (Ras61L). The vector-cotransfected cells were stimulated with PMA (50 ng/ml) for 15 min, after pretreatment of cells with 10 M U0126 (30 min), where indicated. Cell extracts were immunoprecipitated and immunoblotted as in (C). (E) NIH3T3 fibroblasts expressing a conditionally active form of B-Raf were transfected with DAPK. Cells were serum-starved for 18 hr and left untreated or treated with 1 M 4-hydroxy tamoxifen (4-HT) for the indicated time periods for the induction of B-Raf [16]. Cells were immunoprecipitated and immunoblotted as in (C). performed an in vitro kinase assay with immunoprecipitated DAPK as substrate. The kinase-inactive DAPK (K42A) was used in this experiment to avoid potential 32 P incorporation through possible DAPK autophosphorylation. DAPK was phosphorylated in vitro by activated RSK1 but not by kinase-inactive RSK1, and RSK-dependent DAPK phosphorylation was inhibited by UO126 (Figure 2A). To test whether RSK can phosphorylate DAPK in vivo, we coexpressed RSK1 with DAPK in HEK293E cells, which resulted in an 8-fold increase in phosphorylation of DAPK upon stimulation with PMA (Figure 2B). Moreover, when coexpressed in cells, kinase-inactive RSK1 did not increase DAPK phosphorylation (Figure 2B), suggesting that RSK kinase activity is necessary for phosphorylation of DAPK in cells. There was no effect on DAPK phosphorylation when cells were coexpressed with either Akt or MSK2 (Figure S2A). We also observed that both RSK1 and RSK2 were able to phosphorylate DAPK in HEK293E cells (Figure S2B). Interestingly, a constitutively active form of RSK1 (myrrsk1) [8, 22], when coexpressed with DAPK, strongly induced DAPK phosphorylation (Figure 2C). RSK has been shown to be associated with most of its physiological targets [8]. In the present study, we also observed a stimulation-independent interaction of RSK1 with DAPK (Figure 2D).
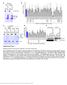
3 Current Biology 1764 Figure 2. RSK Interacts with and Phosphorylates DAPK In Vitro and In Vivo (A) HEK293E cells were transfected with either HA-RSK1 or kinase-inactive RSK1 (RSK1 / ), serum-starved for 18 hr, and treated with PMA (50 ng/ml) for 15 min, with or without pretreatment of cells with U0126 (10 M) for 30 min, and immunoprecipitated with α-ha antibodies. Kinase-inactive DAPK (K42A)-expressing cells were separately lysed, and DAPK was immunoprecipitated with α-flag antibodies. These immunoprecipitates were incubated with RSK immunoprecipitates in a kinase reaction containing [γ- 32 P] ATP for 10 min. The samples were subjected to SDS-PAGE, and the gel was autoradiographed. The lysates were immunoblotted for protein levels, RSK expression, and ERK1/2. (B) Cells were transfected with DAPK and cotransfected with either wild-type RSK1 or the kinase-inactive RSK1 (RSK1 / ). After serum starvation for 18 hr, cells were treated with PMA (50 ng/ml) for 15 min and lysed. DAPK was immunoprecipitated and immunoblotted for phosphorylation on RXRXXS/T motifs [(P)DAPK]. Lysates were immunoblotted for levels of transfected proteins and ERK1/2. (C) HEK293E cells were transfected with DAPK and cotransfected with either wild-type RSK1 or myristoylated RSK1. Cells were serumstarved, stimulated, immunoprecipitated, and immunoblotted as in (B). (D) HEK293E cells were transfected with Flag-DAPK and cotransfected with HA-RSK1. Cells were serum-starved for 18 hr followed by stimulation with PMA (50 ng/ml) for 15 min. The lysates were immunoprecipitated with α-flag antibody and immunoblotted for DAPK phosphorylation. The coimmunoprecipitated RSK was immunoblotted with α-ha antibody. The lysates were also immunoblotted for total protein levels, RSK expression, and ERK1/2. (E) HEK293 cells were transfected with either the mock or RSK1/2 sirna and cotransfected with Flag-DAPK. Cells were starved, stimulated, immunoprecipitated, and immunoblotted as in (D). To further validate the role of RSK1/2 in DAPK phosphorylation, we eliminated RSK1/2 expression by partially knocking down endogenous RSK1/2 with sirna. This resulted in a dramatic decrease in DAPK phosphorylation (Figure 2E), indicating that both RSK1 and RSK2 play a role in regulating DAPK phosphorylation. Taken together, these results demonstrate that DAPK is a specific target of RSK. To identify potential RSK phosphorylation sites in DAPK, tryptic peptides of immunopurified DAPK were subjected to LC-MS/MS/MS analysis with a Fourier transform ion cyclotron resonance (FTICR)/ion trap hybrid mass spectrometer (Figure 3A). One tryptic phosphopeptide, found uniquely from PMA-treated cells, showed phosphorylation at Ser-289, a residue lying in a minimal target site for RSK. The high-mass accuracy measurement of this phosphopeptide in the FTICR cell (0.9 ppm) and the MS 3 spectrum of the precursor ion less a neutral loss of phosphoric acid permitted an unequivocal identification of the phosphorylation site (Figure 3B). The identified phosphorylation site does not lie in a perfect RXRXXS/T consensus sequence reported to be recognized by the anti-rxrxxps/t motif antibody. Therefore, to determine whether this phosphospecific antibody was indeed recognizing DAPK phosphorylated at Ser-289, we generated a phosphorylation-deficient mutant of DAPK (Ser289Ala). Mutation of Ser-289 to alanine almost completely inhibited PMA-induced DAPK phosphorylation (Figure 3C), indicating that Ser- 289 is the predominant site phosphorylated by the activation of Ras-MAPK pathway. Thus, the anti-rxrxxps/ T motif antibody appears to recognize similar, but not identical, basophilic sequences. We also generated point mutations of several other RXRXXS/T sites in DAPK, but we did not see any effect on PMA-induced phosphorylation (data not shown). Moreover, we did not detect these sites in the MS analysis, notwithstanding our conducting of a targeted approach as done for Ser-289. To address the physiological relevance of the RSK-
4 DAP Kinase Is a Target of RSK 1765 Figure 3. Ser-289 Is the Major PMA-Regulated Phosphorylation Site in DAPK (A) HEK293E cells were transfected with Flag-DAPK and treated as shown. A part of the sample was analyzed for DAPK phosphorylation and ERK1/2 levels. The remainder of the sample was subjected to SDS-PAGE and analyzed by liquid-chromatography tandem MS (LC-MS/MS). (B) LC-MS/MS/MS identifies phosphorylation of Ser-289 as an in vivo phosphorylation site on DAPK. MS 1 analysis (left) was performed in the FTICR cell, and the base-peak chromatogram for the entire run is shown above an extracted chromatogram for the m/z range from to encompassing the calculated mass, , for the previously observed phosphopeptide, KA(pS)AVNMEK. The observed mass differed from the calculated mass by less than 1 ppm. Targeted MS 2 (center) of the precursor ion and subsequent MS 3 (right) of the precursor ion less the neutral loss of phosphoric acid produced spectra unambiguously identifying the peptide containing phosphorylated serine 289. (C) HEK293E cells were transfected with either wild-type (WT) or the S289A mutant of DAPK, serum starved, and stimulated with PMA (50 ng/ml) for 15 min. DAPK was immunoprecipitated and immunoblotted for (P)DAPK, DAPK levels, total ERK1/2 levels, and ERK1/2 phosphorylation. mediated phosphorylation of DAPK, we determined whether phosphorylation of DAPK affected its ability to induce cell death. To test this, we transfected cells with wild-type or the phosphorylation-deficient DAPK mutant (Ser289Ala) and monitored their ability to induce cell death by 24 hr. Overexpression of the wild-type DAPK resulted in about 30% cell death. Interestingly, mutation of Ser-289 to alanine induced a dramatic increase in proapoptotic activity of DAPK, resulting in enhanced cell death (Figure 4A). Cells showed about a 50% enhancement in cell death over the wild-type protein. By about 48 hr, most of the cells expressing DAPK- Ser289Ala detached from the substratum. Conversely, mutation of Ser-289 to glutamic acid reduced the proapoptotic activity of DAPK. Ser-289 lies in the calmodulin binding domain (Figure 4B). Binding of calcium-activated calmodulin to the calmodulin binding domain, during the onset of apoptosis, has been shown to alter the conformation of DAPK so that the protein is relieved from autoinhibition [3]. On the basis of this model, it is possible that phosphorylation of Ser-289 in growing cells may hinder the binding of calmodulin so that DAPK retains the autoinhibited conformation. We have not been able to measure significant changes in the catalytic activity of the wild-type or the mutant DAPK proteins when MLC (myosin light chain) was used as a substrate (data not shown). It is possible that Ser289Ala mutation may affect the catalytic activity of DAPK toward specific intracellular substrates, and this may play a role in cell death. Contrary to our findings, a recent report by Chen et al. showed that activation of ERK in DAPK expressing human erythroblastic (D2) cells has a proapoptotic rather than a survival function [23]. These contradictory findings may be attributed in part to the cell-type differences. In addition, it is well established that very potent activation of Ras can lead to cell death [24], although the molecular basis for this remains unclear. Future work will undoubtedly reveal how this molecular switch between survival and death is made by the ERK/RSK/ DAPK protein-kinase module. Our results support a model wherein activation of
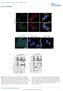
5 Current Biology 1766 Figure 4. Phosphorylation of DAPK on Ser- 289 Antagonizes Its Proapoptotic Activity (A) Graph showing the relative apoptotic activities of the vector, WT DAPK, S289A, and S289E DAPK. HEK293E cells were transfected with the vector, WT DAPK, DAPK S289A, or DAPK S289E along with the EGFP plasmid. The apoptotic cells were quantified as in Figure 1A. All experiments were repeated at least three times, and the results are represented as means ± standard error. The protein levels of WT DAPK and the mutants are shown. (B) Schematic representation of the structure of DAPK, showing various domains and the PMA-regulated RXRXXS/T site, Ser-289. The homology alignment showing conservation of Ser-289 among different mammalian species is also shown. Ras-MAPK pathway negatively regulates the cell-death function of DAPK. In addition to Myt1, DAPK represents the only other kinase known to be regulated downstream of RSK, extending the kinase cascade regulated by the Ras proto-oncoprotein [25]. DAPK also represents the second described tumor suppressor, in addition to TSC2 [15], demonstrated to be antagonized by activated RSK. When Ras is improperly regulated, as in 30% of human cancers, activation of ERK and RSK, as well as subsequent inactivation of DAPK and TSC2, likely contribute to the full tumorigenic phenotype. Phosphorylation of DAPK by RSK may also be the mechanism used to restrain the apoptotic activity of DAPK in healthy cells. Future studies aimed at resolving the crystal structure of DAPK along with the regulatory calmodulin binding domain would provide insight into the role of regulatory phosphorylation in DAPK function. Supplemental Data Supplemental Data include two figures and Experimental Procedures and are available with this article online at current-biology.com/cgi/content/full/15/19/1762/dc1/. Acknowledgments We thank Ruey-Hwa Chen of National Taiwan University, Taiwan for Flag-DAPK cdna construct and Gregory Hoffman, Jessie Hanrahan, and Andrew Yoon Choo for critical reading of the manuscript. This work was supported by National Institutes of Health Grants CA46595 to J.B. and HG00041 to S.P.G., a postdoctoral fellowship from American Heart Association (AHA) to R.A., and a postdoctoral fellowship from the International Human Frontier Science Program Organization to P.P.R. Received: June 13, 2005 Revised: August 17, 2005 Accepted: August 18, 2005 Published: October 11, 2005 References 1. Thompson, C.B. (1995). Apoptosis in the pathogenesis and treatment of disease. Science 267, Danial, S.S., and Korsmeyer, S.J. (2004). Cell death: Critical control points. Cell 116, Cohen, O., Feinstein, E., and Kimchi, A. (1997). DAP-kinase is a Ca2+/calmodulin-dependent, cytoskeletal-associated protein kinase, with cell death-inducing functions that depend on its catalytic activity. EMBO J. 16, Shohat, G., Spivak-Kroizman, T., Cohen, O., Bialik, S., Shani, G., Berrisi, H., Eisenstein, M., and Kimchi, A. (2001). The proapoptotic function of death-associated protein kinase is controlled by a unique inhibitory autophosphorylation-based mechanism. J. Biol. Chem. 276, Inbal, B., Cohen, O., Polak-Charcon, S., Kopolovic, J., Vadai, E., Eisenbach, L., and Kimchi, A. (1997). DAP kinase links the control of apoptosis to metastasis. Nature 390, Raveh, T., Droguett, G., Horwitz, M.S., DePinho, R.A., and Kimchi, A. (2001). DAP kinase activates a p19arf/p53-mediated apoptotic checkpoint to suppress oncogenic transformation. Nat. Cell Biol. 3, Ballif, B.A., and Blenis, J. (2001). Molecular mechanisms mediating mammalian mitogen-activated protein kinase (MAPK) kinase (MEK)-MAPK cell survival signals. Cell Growth Differ. 12, Roux, P.P., and Blenis, J. (2004). ERK and p38 MAPK-activated protein kinases: A family of protein kinases with diverse biological functions. Microbiol. Mol. Biol. Rev. 68, Blenis, J. (1993). Signal transduction via the MAP kinases: Proceed at your own RSK. Proc. Natl. Acad. Sci. USA 90, Nguyen, T.T., Scimeca, J.C., Filloux, C., Peraldi, P., Carpentier, J.L., and Van Obberghen, E. (1993). Co-regulation of the mitogen-activated protein kinase, extracellular signal-regulated kinase 1, and the 90-kDa ribosomal S6 kinase in PC12 cells. Distinct effects of the neurotrophic factor, nerve growth factor, and the mitogenic factor, epidermal growth factor. J. Biol. Chem. 268, Shimamura, A., Ballif, B.A., Richards, S.A., and Blenis, J. (2000). Rsk1 mediates a MEK-MAP kinase cell survival signal. Curr. Biol. 10, Datta, S.R., Brunet, A., and Greenberg, M.E. (1999). Cellular survival: A play in three Akts. Genes Dev. 13, Manning, B.D., Tee, A.R., Logsdon, M.N., Blenis, J., and Cantley, L.C. (2002). Identification of the tuberous sclerosis complex-2 tumor suppressor gene product tuberin as a target of the phosphoinositide 3-kinase/akt pathway. Mol. Cell 10, Tee, A.R., Anjum, R., and Blenis, J. (2003). Inactivation of the tuberous sclerosis complex-1 and -2 gene products occurs by phosphoinositide 3-kinase/Akt-dependent and -independent phosphorylation of tuberin. J. Biol. Chem. 278, Roux, P.P., Ballif, B.A., Anjum, R., Gygi, S.P., and Blenis, J. (2004). Tumor-promoting phorbol esters and activated Ras inactivate the tuberous sclerosis tumor suppressor complex via p90 ribosomal S6 kinase. Proc. Natl. Acad. Sci. USA 101, Pritchard, C.A., Samuels, M.L., Bosch, E., and McMahon, M. (1995). Conditionally oncogenic forms of the A-Raf and B-Raf protein kinases display different biological and biochemical properties in NIH 3T3 cells. Mol. Cell. Biol. 15, Chou, M.M., and Blenis, J. (1996). The 70 kda S6 kinase com-
6 DAP Kinase Is a Target of RSK 1767 plexes with and is activated by the Rho family G proteins Cdc42 and Rac1. Cell 85, Erikson, E., Stefanovic, D., Blenis, J., Erikson, R.L., and Maller, J.L. (1987). Antibodies to Xenopus egg S6 kinase II recognize S6 kinase from progesterone- and insulin-stimulated Xenopus oocytes and from proliferating chicken embryo fibroblasts. Mol. Cell. Biol. 7, Blenis, J. (1993). Signal transduction via the MAP kinases: Proceed at your own RSK. Proc. Natl. Acad. Sci. USA 90, Chen, R.H., Chung, J., and Blenis, J. (1991). Regulation of pp90rsk phosphorylation and S6 phosphotransferase activity in Swiss 3T3 cells by growth factor-, phorbol ester-, and cyclic AMP-mediated signal transduction. Mol. Cell. Biol. 11, Chen, R.H., and Blenis, J. (1990). Identification of Xenopus S6 protein kinase homologs (pp90rsk) in somatic cells: Phosphorylation and activation during initiation of cell proliferation. Mol. Cell. Biol. 10, Roux, P.P., Richards, S.A., and Blenis, J. (2003). Phosphorylation of p90 ribosomal S6 kinase (RSK) regulates extracellular signal-regulated kinase docking and RSK activity. Mol. Cell. Biol. 23, Chen, C.H., Wang, W.J., Kuo, J.C., Tsai, H.C., Lin, J.R., Chang, Z.F., and Chen, R.H. (2005). Bidirectional signals transduced by DAPK-ERK interaction promote the apoptotic effect of DAPK. EMBO J. 24, Bar-Sagi, D.A. (2001). Ras by any other name. Mol. Cell. Biol. 21, Palmer, A., Gavin, A.C., and Nebreda, A.R. (1998). A link between MAP kinase and p34(cdc2)/cyclin B during oocyte maturation: p90(rsk) phosphorylates and inactivates the p34(cdc2) inhibitory kinase Myt1. EMBO J. 17,
Tuberous sclerosis complex (TSC) is an autosomal dominant
 Tumor-promoting phorbol esters and activated Ras inactivate the tuberous sclerosis tumor suppressor complex via p90 ribosomal S6 kinase Philippe P. Roux*, Bryan A. Ballif*, Rana Anjum*, Steven P. Gygi*,
Tumor-promoting phorbol esters and activated Ras inactivate the tuberous sclerosis tumor suppressor complex via p90 ribosomal S6 kinase Philippe P. Roux*, Bryan A. Ballif*, Rana Anjum*, Steven P. Gygi*,
Communicated by Stephen J. Elledge, Harvard Medical School, Boston, MA, December 8, 2004 (received for review October 21, 2004)
 Quantitative phosphorylation profiling of the ERK p90 ribosomal S6 kinase-signaling cassette and its targets, the tuberous sclerosis tumor suppressors Bryan A. Ballif*, Philippe P. Roux*, Scott A. Gerber*,
Quantitative phosphorylation profiling of the ERK p90 ribosomal S6 kinase-signaling cassette and its targets, the tuberous sclerosis tumor suppressors Bryan A. Ballif*, Philippe P. Roux*, Scott A. Gerber*,
A particular set of insults induces apoptosis (part 1), which, if inhibited, can switch to autophagy. At least in some cellular settings, autophagy se
 A particular set of insults induces apoptosis (part 1), which, if inhibited, can switch to autophagy. At least in some cellular settings, autophagy serves as a defence mechanism that prevents or retards
A particular set of insults induces apoptosis (part 1), which, if inhibited, can switch to autophagy. At least in some cellular settings, autophagy serves as a defence mechanism that prevents or retards
SUPPLEMENTARY INFORMATION
 doi:10.1038/nature10962 Supplementary Figure 1. Expression of AvrAC-FLAG in protoplasts. Total protein extracted from protoplasts described in Fig. 1a was subjected to anti-flag immunoblot to detect AvrAC-FLAG
doi:10.1038/nature10962 Supplementary Figure 1. Expression of AvrAC-FLAG in protoplasts. Total protein extracted from protoplasts described in Fig. 1a was subjected to anti-flag immunoblot to detect AvrAC-FLAG
Cell Signaling part 2
 15 Cell Signaling part 2 Functions of Cell Surface Receptors Other cell surface receptors are directly linked to intracellular enzymes. The largest family of these is the receptor protein tyrosine kinases,
15 Cell Signaling part 2 Functions of Cell Surface Receptors Other cell surface receptors are directly linked to intracellular enzymes. The largest family of these is the receptor protein tyrosine kinases,
Part-4. Cell cycle regulatory protein 5 (Cdk5) A novel target of ERK in Carb induced cell death
 Part-4 Cell cycle regulatory protein 5 (Cdk5) A novel target of ERK in Carb induced cell death 95 1. Introduction The process of replicating DNA and dividing cells can be described as a series of coordinated
Part-4 Cell cycle regulatory protein 5 (Cdk5) A novel target of ERK in Carb induced cell death 95 1. Introduction The process of replicating DNA and dividing cells can be described as a series of coordinated
Supplementary Figure 1. PD-L1 is glycosylated in cancer cells. (a) Western blot analysis of PD-L1 in breast cancer cells. (b) Western blot analysis
 Supplementary Figure 1. PD-L1 is glycosylated in cancer cells. (a) Western blot analysis of PD-L1 in breast cancer cells. (b) Western blot analysis of PD-L1 in ovarian cancer cells. (c) Western blot analysis
Supplementary Figure 1. PD-L1 is glycosylated in cancer cells. (a) Western blot analysis of PD-L1 in breast cancer cells. (b) Western blot analysis of PD-L1 in ovarian cancer cells. (c) Western blot analysis
Growth and Differentiation Phosphorylation Sampler Kit
 Growth and Differentiation Phosphorylation Sampler Kit E 0 5 1 0 1 4 Kits Includes Cat. Quantity Application Reactivity Source Akt (Phospho-Ser473) E011054-1 50μg/50μl IHC, WB Human, Mouse, Rat Rabbit
Growth and Differentiation Phosphorylation Sampler Kit E 0 5 1 0 1 4 Kits Includes Cat. Quantity Application Reactivity Source Akt (Phospho-Ser473) E011054-1 50μg/50μl IHC, WB Human, Mouse, Rat Rabbit
SUPPLEMENTARY INFORMATION
 Supplementary Table 1. Cell sphingolipids and S1P bound to endogenous TRAF2. Sphingolipid Cell pmol/mg TRAF2 immunoprecipitate pmol/mg Sphingomyelin 4200 ± 250 Not detected Monohexosylceramide 311 ± 18
Supplementary Table 1. Cell sphingolipids and S1P bound to endogenous TRAF2. Sphingolipid Cell pmol/mg TRAF2 immunoprecipitate pmol/mg Sphingomyelin 4200 ± 250 Not detected Monohexosylceramide 311 ± 18
Phospho-AKT Sampler Kit
 Phospho-AKT Sampler Kit E 0 5 1 0 0 3 Kits Includes Cat. Quantity Application Reactivity Source Akt (Ab-473) Antibody E021054-1 50μg/50μl IHC, WB Human, Mouse, Rat Rabbit Akt (Phospho-Ser473) Antibody
Phospho-AKT Sampler Kit E 0 5 1 0 0 3 Kits Includes Cat. Quantity Application Reactivity Source Akt (Ab-473) Antibody E021054-1 50μg/50μl IHC, WB Human, Mouse, Rat Rabbit Akt (Phospho-Ser473) Antibody
KEY CONCEPT QUESTIONS IN SIGNAL TRANSDUCTION
 Signal Transduction - Part 2 Key Concepts - Receptor tyrosine kinases control cell metabolism and proliferation Growth factor signaling through Ras Mutated cell signaling genes in cancer cells are called
Signal Transduction - Part 2 Key Concepts - Receptor tyrosine kinases control cell metabolism and proliferation Growth factor signaling through Ras Mutated cell signaling genes in cancer cells are called
MEK1 Assay Kit 1 Catalog # Lot # 16875
 MEK1 Assay Kit 1 Kit Components Assay Dilution Buffer (ADB), Catalog # 20-108. Three vials, each containing 1.0ml of assay dilution buffer (20mM MOPS, ph 7.2, 25mM ß-glycerol phosphate, 5mM EGTA, 1mM sodium
MEK1 Assay Kit 1 Kit Components Assay Dilution Buffer (ADB), Catalog # 20-108. Three vials, each containing 1.0ml of assay dilution buffer (20mM MOPS, ph 7.2, 25mM ß-glycerol phosphate, 5mM EGTA, 1mM sodium
The PI3K/AKT axis. Dr. Lucio Crinò Medical Oncology Division Azienda Ospedaliera-Perugia. Introduction
 The PI3K/AKT axis Dr. Lucio Crinò Medical Oncology Division Azienda Ospedaliera-Perugia Introduction Phosphoinositide 3-kinase (PI3K) pathway are a family of lipid kinases discovered in 1980s. They have
The PI3K/AKT axis Dr. Lucio Crinò Medical Oncology Division Azienda Ospedaliera-Perugia Introduction Phosphoinositide 3-kinase (PI3K) pathway are a family of lipid kinases discovered in 1980s. They have
Nature Structural & Molecular Biology: doi: /nsmb.3218
 Supplementary Figure 1 Endogenous EGFR trafficking and responses depend on biased ligands. (a) Lysates from HeLa cells stimulated for 2 min. with increasing concentration of ligands were immunoblotted
Supplementary Figure 1 Endogenous EGFR trafficking and responses depend on biased ligands. (a) Lysates from HeLa cells stimulated for 2 min. with increasing concentration of ligands were immunoblotted
G-Protein Signaling. Introduction to intracellular signaling. Dr. SARRAY Sameh, Ph.D
 G-Protein Signaling Introduction to intracellular signaling Dr. SARRAY Sameh, Ph.D Cell signaling Cells communicate via extracellular signaling molecules (Hormones, growth factors and neurotransmitters
G-Protein Signaling Introduction to intracellular signaling Dr. SARRAY Sameh, Ph.D Cell signaling Cells communicate via extracellular signaling molecules (Hormones, growth factors and neurotransmitters
Enzyme-coupled Receptors. Cell-surface receptors 1. Ion-channel-coupled receptors 2. G-protein-coupled receptors 3. Enzyme-coupled receptors
 Enzyme-coupled Receptors Cell-surface receptors 1. Ion-channel-coupled receptors 2. G-protein-coupled receptors 3. Enzyme-coupled receptors Cell-surface receptors allow a flow of ions across the plasma
Enzyme-coupled Receptors Cell-surface receptors 1. Ion-channel-coupled receptors 2. G-protein-coupled receptors 3. Enzyme-coupled receptors Cell-surface receptors allow a flow of ions across the plasma
RAS Genes. The ras superfamily of genes encodes small GTP binding proteins that are responsible for the regulation of many cellular processes.
 ۱ RAS Genes The ras superfamily of genes encodes small GTP binding proteins that are responsible for the regulation of many cellular processes. Oncogenic ras genes in human cells include H ras, N ras,
۱ RAS Genes The ras superfamily of genes encodes small GTP binding proteins that are responsible for the regulation of many cellular processes. Oncogenic ras genes in human cells include H ras, N ras,
Predictive PP1Ca binding region in BIG3 : 1,228 1,232aa (-KAVSF-) HEK293T cells *** *** *** KPL-3C cells - E E2 treatment time (h)
 Relative expression ERE-luciferase activity activity (pmole/min) activity (pmole/min) activity (pmole/min) activity (pmole/min) MCF-7 KPL-3C ZR--1 BT-474 T47D HCC15 KPL-1 HBC4 activity (pmole/min) a d
Relative expression ERE-luciferase activity activity (pmole/min) activity (pmole/min) activity (pmole/min) activity (pmole/min) MCF-7 KPL-3C ZR--1 BT-474 T47D HCC15 KPL-1 HBC4 activity (pmole/min) a d
PREPARED FOR: U.S. Army Medical Research and Materiel Command Fort Detrick, Maryland
 AD Award Number: W81XWH-09-1-0279 TITLE: Regulation of mtor by Nutrients PRINCIPAL INVESTIGATOR: Kun-Liang Guan CONTRACTING ORGANIZATION: University of San Diego La Jolla, CA 92093 REPORT DATE: July 2010
AD Award Number: W81XWH-09-1-0279 TITLE: Regulation of mtor by Nutrients PRINCIPAL INVESTIGATOR: Kun-Liang Guan CONTRACTING ORGANIZATION: University of San Diego La Jolla, CA 92093 REPORT DATE: July 2010
Protein kinases are enzymes that add a phosphate group to proteins according to the. ATP + protein OH > Protein OPO 3 + ADP
 Protein kinase Protein kinases are enzymes that add a phosphate group to proteins according to the following equation: 2 ATP + protein OH > Protein OPO 3 + ADP ATP represents adenosine trisphosphate, ADP
Protein kinase Protein kinases are enzymes that add a phosphate group to proteins according to the following equation: 2 ATP + protein OH > Protein OPO 3 + ADP ATP represents adenosine trisphosphate, ADP
Diabetes Mellitus and Breast Cancer
 Masur K, Thévenod F, Zänker KS (eds): Diabetes and Cancer. Epidemiological Evidence and Molecular Links. Front Diabetes. Basel, Karger, 2008, vol 19, pp 97 113 Diabetes Mellitus and Breast Cancer Ido Wolf
Masur K, Thévenod F, Zänker KS (eds): Diabetes and Cancer. Epidemiological Evidence and Molecular Links. Front Diabetes. Basel, Karger, 2008, vol 19, pp 97 113 Diabetes Mellitus and Breast Cancer Ido Wolf
Biol403 MAP kinase signalling
 Biol403 MAP kinase signalling The mitogen activated protein kinase (MAPK) pathway is a signalling cascade activated by a diverse range of effectors. The cascade regulates many cellular activities including
Biol403 MAP kinase signalling The mitogen activated protein kinase (MAPK) pathway is a signalling cascade activated by a diverse range of effectors. The cascade regulates many cellular activities including
Cellular Signaling Pathways. Signaling Overview
 Cellular Signaling Pathways Signaling Overview Signaling steps Synthesis and release of signaling molecules (ligands) by the signaling cell. Transport of the signal to the target cell Detection of the
Cellular Signaling Pathways Signaling Overview Signaling steps Synthesis and release of signaling molecules (ligands) by the signaling cell. Transport of the signal to the target cell Detection of the
Supplemental material for Hernandez et al. Dicoumarol downregulates human PTTG1/Securin mrna expression. through inhibition of Hsp90
 Supplemental material for Hernandez et al. Dicoumarol downregulates human PTTG1/Securin mrna expression through inhibition of Hsp90 Dicoumarol-Sepharose co-precipitation. Hsp90 inhibitors can co-precipitate
Supplemental material for Hernandez et al. Dicoumarol downregulates human PTTG1/Securin mrna expression through inhibition of Hsp90 Dicoumarol-Sepharose co-precipitation. Hsp90 inhibitors can co-precipitate
Supplementary Figure 1. Normal T lymphocyte populations in Dapk -/- mice. (a) Normal thymic development in Dapk -/- mice. Thymocytes from WT and Dapk
 Supplementary Figure 1. Normal T lymphocyte populations in Dapk -/- mice. (a) Normal thymic development in Dapk -/- mice. Thymocytes from WT and Dapk -/- mice were stained for expression of CD4 and CD8.
Supplementary Figure 1. Normal T lymphocyte populations in Dapk -/- mice. (a) Normal thymic development in Dapk -/- mice. Thymocytes from WT and Dapk -/- mice were stained for expression of CD4 and CD8.
Megan S. Lim MD PhD. Translating Mass Spectrometry-Based Proteomics of Malignant Lymphoma into Clinical Application
 Translating Mass Spectrometry-Based Proteomics of Malignant Lymphoma into Clinical Application Megan S. Lim MD PhD FRCPC Department of Pathology, University of Michigan, Ann Arbor, MI Proteomics is a multi-faceted
Translating Mass Spectrometry-Based Proteomics of Malignant Lymphoma into Clinical Application Megan S. Lim MD PhD FRCPC Department of Pathology, University of Michigan, Ann Arbor, MI Proteomics is a multi-faceted
William C. Comb, Jessica E. Hutti, Patricia Cogswell, Lewis C. Cantley, and Albert S. Baldwin
 Molecular Cell, Volume 45 Supplemental Information p85 SH2 Domain Phosphorylation by IKK Promotes Feedback Inhibition of PI3K and Akt in Response to Cellular Starvation William C. Comb, Jessica E. Hutti,
Molecular Cell, Volume 45 Supplemental Information p85 SH2 Domain Phosphorylation by IKK Promotes Feedback Inhibition of PI3K and Akt in Response to Cellular Starvation William C. Comb, Jessica E. Hutti,
m 6 A mrna methylation regulates AKT activity to promote the proliferation and tumorigenicity of endometrial cancer
 SUPPLEMENTARY INFORMATION Articles https://doi.org/10.1038/s41556-018-0174-4 In the format provided by the authors and unedited. m 6 A mrna methylation regulates AKT activity to promote the proliferation
SUPPLEMENTARY INFORMATION Articles https://doi.org/10.1038/s41556-018-0174-4 In the format provided by the authors and unedited. m 6 A mrna methylation regulates AKT activity to promote the proliferation
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION doi:1.138/nature9814 a A SHARPIN FL B SHARPIN ΔNZF C SHARPIN T38L, F39V b His-SHARPIN FL -1xUb -2xUb -4xUb α-his c Linear 4xUb -SHARPIN FL -SHARPIN TF_LV -SHARPINΔNZF -SHARPIN
SUPPLEMENTARY INFORMATION doi:1.138/nature9814 a A SHARPIN FL B SHARPIN ΔNZF C SHARPIN T38L, F39V b His-SHARPIN FL -1xUb -2xUb -4xUb α-his c Linear 4xUb -SHARPIN FL -SHARPIN TF_LV -SHARPINΔNZF -SHARPIN
Principles of Genetics and Molecular Biology
 Cell signaling Dr. Diala Abu-Hassan, DDS, PhD School of Medicine Dr.abuhassand@gmail.com Principles of Genetics and Molecular Biology www.cs.montana.edu Modes of cell signaling Direct interaction of a
Cell signaling Dr. Diala Abu-Hassan, DDS, PhD School of Medicine Dr.abuhassand@gmail.com Principles of Genetics and Molecular Biology www.cs.montana.edu Modes of cell signaling Direct interaction of a
Cell cycle, signaling to cell cycle, and molecular basis of oncogenesis
 Cell cycle, signaling to cell cycle, and molecular basis of oncogenesis MUDr. Jiří Vachtenheim, CSc. CELL CYCLE - SUMMARY Basic terminology: Cyclins conserved proteins with homologous regions; their cellular
Cell cycle, signaling to cell cycle, and molecular basis of oncogenesis MUDr. Jiří Vachtenheim, CSc. CELL CYCLE - SUMMARY Basic terminology: Cyclins conserved proteins with homologous regions; their cellular
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION doi:10.1038/nature11429 S1a 6 7 8 9 Nlrc4 allele S1b Nlrc4 +/+ Nlrc4 +/F Nlrc4 F/F 9 Targeting construct 422 bp 273 bp FRT-neo-gb-PGK-FRT 3x.STOP S1c Nlrc4 +/+ Nlrc4 F/F casp1
SUPPLEMENTARY INFORMATION doi:10.1038/nature11429 S1a 6 7 8 9 Nlrc4 allele S1b Nlrc4 +/+ Nlrc4 +/F Nlrc4 F/F 9 Targeting construct 422 bp 273 bp FRT-neo-gb-PGK-FRT 3x.STOP S1c Nlrc4 +/+ Nlrc4 F/F casp1
The Tissue Engineer s Toolkit
 The Tissue Engineer s Toolkit Stimuli Detection and Response Ken Webb, Ph. D. Assistant Professor Dept. of Bioengineering Clemson University Environmental Stimulus-Cellular Response Environmental Stimuli
The Tissue Engineer s Toolkit Stimuli Detection and Response Ken Webb, Ph. D. Assistant Professor Dept. of Bioengineering Clemson University Environmental Stimulus-Cellular Response Environmental Stimuli
Signal Transduction I
 Signal Transduction I Prof. Tianhua Zhou Department of Cell Biology Zhejiang University School of Medicine Office hours by appointment tzhou@zju.edu.cn Signal transduction: Key contents for signal transduction:
Signal Transduction I Prof. Tianhua Zhou Department of Cell Biology Zhejiang University School of Medicine Office hours by appointment tzhou@zju.edu.cn Signal transduction: Key contents for signal transduction:
The elements of G protein-coupled receptor systems
 The elements of G protein-coupled receptor systems Prostaglandines Sphingosine 1-phosphate a receptor that contains 7 membrane-spanning domains a coupled trimeric G protein which functions as a switch
The elements of G protein-coupled receptor systems Prostaglandines Sphingosine 1-phosphate a receptor that contains 7 membrane-spanning domains a coupled trimeric G protein which functions as a switch
Supplementary Figure 1 CD4 + T cells from PKC-θ null mice are defective in NF-κB activation during T cell receptor signaling. CD4 + T cells were
 Supplementary Figure 1 CD4 + T cells from PKC-θ null mice are defective in NF-κB activation during T cell receptor signaling. CD4 + T cells were isolated from wild type (PKC-θ- WT) or PKC-θ null (PKC-θ-KO)
Supplementary Figure 1 CD4 + T cells from PKC-θ null mice are defective in NF-κB activation during T cell receptor signaling. CD4 + T cells were isolated from wild type (PKC-θ- WT) or PKC-θ null (PKC-θ-KO)
Supplementary Materials
 Supplementary Materials Figure S1. MTT Cell viability assay. To measure the cytotoxic potential of the oxidative treatment, the MTT [3-(4,5-dimethylthiazol- 2-yl)-2,5-diphenyl tetrazolium bromide] assay
Supplementary Materials Figure S1. MTT Cell viability assay. To measure the cytotoxic potential of the oxidative treatment, the MTT [3-(4,5-dimethylthiazol- 2-yl)-2,5-diphenyl tetrazolium bromide] assay
a b G75 G60 Sw-2 Sw-1 Supplementary Figure 1. Structure predictions by I-TASSER Server.
 a b G75 2 2 G60 Sw-2 Sw-1 Supplementary Figure 1. Structure predictions by I-TASSER Server. a. Overlay of top 10 models generated by I-TASSER illustrates the potential effect of 7 amino acid insertion
a b G75 2 2 G60 Sw-2 Sw-1 Supplementary Figure 1. Structure predictions by I-TASSER Server. a. Overlay of top 10 models generated by I-TASSER illustrates the potential effect of 7 amino acid insertion
Cyclin I activates Cdk5 and regulates expression of Bcl-2 and Bcl-XL in postmitotic mouse cells
 Research article Cyclin I activates Cdk5 and regulates expression of Bcl-2 and Bcl-XL in postmitotic mouse cells Paul T. Brinkkoetter, 1 Paul Olivier, 2 Jimmy S. Wu, 1 Scott Henderson, 1 Ronald D. Krofft,
Research article Cyclin I activates Cdk5 and regulates expression of Bcl-2 and Bcl-XL in postmitotic mouse cells Paul T. Brinkkoetter, 1 Paul Olivier, 2 Jimmy S. Wu, 1 Scott Henderson, 1 Ronald D. Krofft,
Intracellular MHC class II molecules promote TLR-triggered innate. immune responses by maintaining Btk activation
 Intracellular MHC class II molecules promote TLR-triggered innate immune responses by maintaining Btk activation Xingguang Liu, Zhenzhen Zhan, Dong Li, Li Xu, Feng Ma, Peng Zhang, Hangping Yao and Xuetao
Intracellular MHC class II molecules promote TLR-triggered innate immune responses by maintaining Btk activation Xingguang Liu, Zhenzhen Zhan, Dong Li, Li Xu, Feng Ma, Peng Zhang, Hangping Yao and Xuetao
SUPPLEMENTARY INFORMATION
 DOI: 10.1038/ncb3311 A B TSC2 -/- MEFs C Rapa Hours WCL 0 6 12 24 36 pakt.s473 AKT ps6k S6K CM IGF-1 Recipient WCL - + - + - + pigf-1r IGF-1R pakt ps6 AKT D 1 st SILAC 2 nd SILAC E GAPDH FGF21 ALKPGVIQILGVK
DOI: 10.1038/ncb3311 A B TSC2 -/- MEFs C Rapa Hours WCL 0 6 12 24 36 pakt.s473 AKT ps6k S6K CM IGF-1 Recipient WCL - + - + - + pigf-1r IGF-1R pakt ps6 AKT D 1 st SILAC 2 nd SILAC E GAPDH FGF21 ALKPGVIQILGVK
SUPPLEMENTARY INFORMATION
 Supplementary Figures Supplementary Figure S1. Binding of full-length OGT and deletion mutants to PIP strips (Echelon Biosciences). Supplementary Figure S2. Binding of the OGT (919-1036) fragments with
Supplementary Figures Supplementary Figure S1. Binding of full-length OGT and deletion mutants to PIP strips (Echelon Biosciences). Supplementary Figure S2. Binding of the OGT (919-1036) fragments with
Receptor mediated Signal Transduction
 Receptor mediated Signal Transduction G-protein-linked receptors adenylyl cyclase camp PKA Organization of receptor protein-tyrosine kinases From G.M. Cooper, The Cell. A molecular approach, 2004, third
Receptor mediated Signal Transduction G-protein-linked receptors adenylyl cyclase camp PKA Organization of receptor protein-tyrosine kinases From G.M. Cooper, The Cell. A molecular approach, 2004, third
Supplementary Material
 Supplementary Material The Androgen Receptor is a negative regulator of eif4e Phosphorylation at S209: Implications for the use of mtor inhibitors in advanced prostate cancer Supplementary Figures Supplemental
Supplementary Material The Androgen Receptor is a negative regulator of eif4e Phosphorylation at S209: Implications for the use of mtor inhibitors in advanced prostate cancer Supplementary Figures Supplemental
Lecture: CHAPTER 13 Signal Transduction Pathways
 Lecture: 10 17 2016 CHAPTER 13 Signal Transduction Pathways Chapter 13 Outline Signal transduction cascades have many components in common: 1. Release of a primary message as a response to a physiological
Lecture: 10 17 2016 CHAPTER 13 Signal Transduction Pathways Chapter 13 Outline Signal transduction cascades have many components in common: 1. Release of a primary message as a response to a physiological
Supplementary Figure 1
 Supplementary Figure 1 Supplementary Figure 1. Neither the activation nor suppression of the MAPK pathway affects the ASK1/Vif interaction. (a, b) HEK293 cells were cotransfected with plasmids encoding
Supplementary Figure 1 Supplementary Figure 1. Neither the activation nor suppression of the MAPK pathway affects the ASK1/Vif interaction. (a, b) HEK293 cells were cotransfected with plasmids encoding
Regulation of cell function by intracellular signaling
 Regulation of cell function by intracellular signaling Objectives: Regulation principle Allosteric and covalent mechanisms, Popular second messengers, Protein kinases, Kinase cascade and interaction. regulation
Regulation of cell function by intracellular signaling Objectives: Regulation principle Allosteric and covalent mechanisms, Popular second messengers, Protein kinases, Kinase cascade and interaction. regulation
1. Activated receptor tyrosine kinases (RTKs) phosphorylates themselves
 Enzyme-coupled receptors Transmembrane proteins Ligand-binding domain on the outer surface Cytoplasmic domain acts as an enzyme itself or forms a complex with enzyme 1. Activated receptor tyrosine kinases
Enzyme-coupled receptors Transmembrane proteins Ligand-binding domain on the outer surface Cytoplasmic domain acts as an enzyme itself or forms a complex with enzyme 1. Activated receptor tyrosine kinases
SUPPLEMENTARY INFORMATION
 DOI: 10.1038/ncb3076 Supplementary Figure 1 btrcp targets Cep68 for degradation during mitosis. a) Cep68 immunofluorescence in interphase and metaphase. U-2OS cells were transfected with control sirna
DOI: 10.1038/ncb3076 Supplementary Figure 1 btrcp targets Cep68 for degradation during mitosis. a) Cep68 immunofluorescence in interphase and metaphase. U-2OS cells were transfected with control sirna
Supplementary Materials for
 www.sciencesignaling.org/cgi/content/full/6/278/rs11/dc1 Supplementary Materials for In Vivo Phosphoproteomics Analysis Reveals the Cardiac Targets of β-adrenergic Receptor Signaling Alicia Lundby,* Martin
www.sciencesignaling.org/cgi/content/full/6/278/rs11/dc1 Supplementary Materials for In Vivo Phosphoproteomics Analysis Reveals the Cardiac Targets of β-adrenergic Receptor Signaling Alicia Lundby,* Martin
Signaling. Dr. Sujata Persad Katz Group Centre for Pharmacy & Health research
 Signaling Dr. Sujata Persad 3-020 Katz Group Centre for Pharmacy & Health research E-mail:sujata.persad@ualberta.ca 1 Growth Factor Receptors and Other Signaling Pathways What we will cover today: How
Signaling Dr. Sujata Persad 3-020 Katz Group Centre for Pharmacy & Health research E-mail:sujata.persad@ualberta.ca 1 Growth Factor Receptors and Other Signaling Pathways What we will cover today: How
EGF receptor transactivation is crucial for cholinergic MAP kinase signaling in human keratinocytes
 1st Electronic Conference on Molecular Science EGF receptor transactivation is crucial for cholinergic MAP kinase signaling in human keratinocytes Wymke Ockenga, Sina Kühne, Antje Banning and Ritva Tikkanen
1st Electronic Conference on Molecular Science EGF receptor transactivation is crucial for cholinergic MAP kinase signaling in human keratinocytes Wymke Ockenga, Sina Kühne, Antje Banning and Ritva Tikkanen
Identification of non-ser/thr-pro consensus motifs for Cdk1 and their roles in mitotic regulation of C2H2 zinc finger proteins and Ect2
 SUPPLEMENTARY INFORMATION Identification of non-ser/thr-pro consensus motifs for Cdk1 and their roles in mitotic regulation of C2H2 zinc finger proteins and Ect2 Kazuhiro Suzuki, Kosuke Sako, Kazuhiro
SUPPLEMENTARY INFORMATION Identification of non-ser/thr-pro consensus motifs for Cdk1 and their roles in mitotic regulation of C2H2 zinc finger proteins and Ect2 Kazuhiro Suzuki, Kosuke Sako, Kazuhiro
Supporting Information. FADD regulates NF-кB activation and promotes ubiquitination of cflip L to induce. apoptosis
 1 2 Supporting Information 3 4 5 FADD regulates NF-кB activation and promotes ubiquitination of cflip L to induce apoptosis 6 7 Kishu Ranjan and Chandramani Pathak* 8 9 Department of Cell Biology, School
1 2 Supporting Information 3 4 5 FADD regulates NF-кB activation and promotes ubiquitination of cflip L to induce apoptosis 6 7 Kishu Ranjan and Chandramani Pathak* 8 9 Department of Cell Biology, School
Synthesis and Biological Evaluation of Protein Kinase D Inhibitors
 Synthesis and Biological Evaluation of Protein Kinase D Inhibitors Celeste Alverez Topic Seminar October 26, 2013 Celeste Alverez @ Wipf Group 10/26/2013 1 Protein Kinase D (PKD) A novel family of serine/threonine
Synthesis and Biological Evaluation of Protein Kinase D Inhibitors Celeste Alverez Topic Seminar October 26, 2013 Celeste Alverez @ Wipf Group 10/26/2013 1 Protein Kinase D (PKD) A novel family of serine/threonine
Egr-1 regulates RTA transcription through a cooperative involvement of transcriptional regulators
 /, 2017, Vol. 8, (No. 53), pp: 91425-91444 Egr-1 regulates RTA transcription through a cooperative involvement of transcriptional regulators Roni Sarkar 1 and Subhash C. Verma 1 1 Department of Microbiology
/, 2017, Vol. 8, (No. 53), pp: 91425-91444 Egr-1 regulates RTA transcription through a cooperative involvement of transcriptional regulators Roni Sarkar 1 and Subhash C. Verma 1 1 Department of Microbiology
Nature Structural and Molecular Biology: doi: /nsmb Supplementary Figure 1
 Supplementary Figure 1 Mutational analysis of the SA2-Scc1 interaction in vitro and in human cells. (a) Autoradiograph (top) and Coomassie stained gel (bottom) of 35 S-labeled Myc-SA2 proteins (input)
Supplementary Figure 1 Mutational analysis of the SA2-Scc1 interaction in vitro and in human cells. (a) Autoradiograph (top) and Coomassie stained gel (bottom) of 35 S-labeled Myc-SA2 proteins (input)
Supplementary Figure 1.TRIM33 binds β-catenin in the nucleus. a & b, Co-IP of endogenous TRIM33 with β-catenin in HT-29 cells (a) and HEK 293T cells
 Supplementary Figure 1.TRIM33 binds β-catenin in the nucleus. a & b, Co-IP of endogenous TRIM33 with β-catenin in HT-29 cells (a) and HEK 293T cells (b). TRIM33 was immunoprecipitated, and the amount of
Supplementary Figure 1.TRIM33 binds β-catenin in the nucleus. a & b, Co-IP of endogenous TRIM33 with β-catenin in HT-29 cells (a) and HEK 293T cells (b). TRIM33 was immunoprecipitated, and the amount of
Supplemental Data Macrophage Migration Inhibitory Factor MIF Interferes with the Rb-E2F Pathway
 Supplemental Data Macrophage Migration Inhibitory Factor MIF Interferes with the Rb-E2F Pathway S1 Oleksi Petrenko and Ute M. Moll Figure S1. MIF-Deficient Cells Have Reduced Transforming Ability (A) Soft
Supplemental Data Macrophage Migration Inhibitory Factor MIF Interferes with the Rb-E2F Pathway S1 Oleksi Petrenko and Ute M. Moll Figure S1. MIF-Deficient Cells Have Reduced Transforming Ability (A) Soft
Introduction: 年 Fas signal-mediated apoptosis. PI3K/Akt
 Fas-ligand (CD95-L; Fas-L) Fas (CD95) Fas (apoptosis) 年 了 不 度 Fas Fas-L 力 不 Fas/Fas-L T IL-10Fas/Fas-L 不 年 Fas signal-mediated apoptosis 度降 不 不 力 U-118, HeLa, A549, Huh-7 MCF-7, HepG2. PI3K/Akt FasPI3K/Akt
Fas-ligand (CD95-L; Fas-L) Fas (CD95) Fas (apoptosis) 年 了 不 度 Fas Fas-L 力 不 Fas/Fas-L T IL-10Fas/Fas-L 不 年 Fas signal-mediated apoptosis 度降 不 不 力 U-118, HeLa, A549, Huh-7 MCF-7, HepG2. PI3K/Akt FasPI3K/Akt
Electrical Stimulation Control Nerve Regeneration via the p38 Mitogen-activated Protein Kinase and CREB
 Electrical Stimulation Control Nerve Regeneration via the p38 Mitogen-activated Protein Kinase and CREB Kenji Kawamura, Yoshio Kano. Kibi International University, Takahashi-city, Japan. Disclosures: K.
Electrical Stimulation Control Nerve Regeneration via the p38 Mitogen-activated Protein Kinase and CREB Kenji Kawamura, Yoshio Kano. Kibi International University, Takahashi-city, Japan. Disclosures: K.
A Hepatocyte Growth Factor Receptor (Met) Insulin Receptor hybrid governs hepatic glucose metabolism SUPPLEMENTARY FIGURES, LEGENDS AND METHODS
 A Hepatocyte Growth Factor Receptor (Met) Insulin Receptor hybrid governs hepatic glucose metabolism Arlee Fafalios, Jihong Ma, Xinping Tan, John Stoops, Jianhua Luo, Marie C. DeFrances and Reza Zarnegar
A Hepatocyte Growth Factor Receptor (Met) Insulin Receptor hybrid governs hepatic glucose metabolism Arlee Fafalios, Jihong Ma, Xinping Tan, John Stoops, Jianhua Luo, Marie C. DeFrances and Reza Zarnegar
INTERACTION DRUG BODY
 INTERACTION DRUG BODY What the drug does to the body What the body does to the drug Receptors - intracellular receptors - membrane receptors - Channel receptors - G protein-coupled receptors - Tyrosine-kinase
INTERACTION DRUG BODY What the drug does to the body What the body does to the drug Receptors - intracellular receptors - membrane receptors - Channel receptors - G protein-coupled receptors - Tyrosine-kinase
Bioluminescence Resonance Energy Transfer (BRET)-based studies of receptor dynamics in living cells with Berthold s Mithras
 Bioluminescence Resonance Energy Transfer (BRET)-based studies of receptor dynamics in living cells with Berthold s Mithras Tarik Issad, Ralf Jockers and Stefano Marullo 1 Because they play a pivotal role
Bioluminescence Resonance Energy Transfer (BRET)-based studies of receptor dynamics in living cells with Berthold s Mithras Tarik Issad, Ralf Jockers and Stefano Marullo 1 Because they play a pivotal role
FGL2 A new biomarker for cancer in a simple blood test
 FGL2 A new biomarker for cancer in a simple blood test WHO IS FGL2 Human gene (chromosome 7) is 7 kb long, 2 exons, monomer protein 70 KD, tetramer in solution. Fibrinogen-like protein 2 (Fgl2), a member
FGL2 A new biomarker for cancer in a simple blood test WHO IS FGL2 Human gene (chromosome 7) is 7 kb long, 2 exons, monomer protein 70 KD, tetramer in solution. Fibrinogen-like protein 2 (Fgl2), a member
Supplementary Figure 1. Basal level EGFR across a panel of ESCC lines. Immunoblots demonstrate the expression of phosphorylated and total EGFR as
 Supplementary Figure 1. Basal level EGFR across a panel of ESCC lines. Immunoblots demonstrate the expression of phosphorylated and total EGFR as well as their downstream effectors across a panel of ESCC
Supplementary Figure 1. Basal level EGFR across a panel of ESCC lines. Immunoblots demonstrate the expression of phosphorylated and total EGFR as well as their downstream effectors across a panel of ESCC
Early cell death (FGF) B No RunX transcription factor produced Yes No differentiation
 Solution Key - Practice Questions Question 1 a) A recent publication has shown that the fat stem cells (FSC) can act as bone stem cells to repair cavities in the skull, when transplanted into immuno-compromised
Solution Key - Practice Questions Question 1 a) A recent publication has shown that the fat stem cells (FSC) can act as bone stem cells to repair cavities in the skull, when transplanted into immuno-compromised
Prostaglandin E 2 reduces radiation-induced epithelial apoptosis through a mechanism involving AKT activation and bax translocation
 Research article Prostaglandin E 2 reduces radiation-induced epithelial apoptosis through a mechanism involving AKT activation and bax translocation Teresa G. Tessner, Filipe Muhale, Terrence E. Riehl,
Research article Prostaglandin E 2 reduces radiation-induced epithelial apoptosis through a mechanism involving AKT activation and bax translocation Teresa G. Tessner, Filipe Muhale, Terrence E. Riehl,
Propagation of the Signal
 OpenStax-CNX module: m44452 1 Propagation of the Signal OpenStax College This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License 3.0 By the end of this section,
OpenStax-CNX module: m44452 1 Propagation of the Signal OpenStax College This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License 3.0 By the end of this section,
Chapter 15: Signal transduction
 Chapter 15: Signal transduction Know the terminology: Enzyme-linked receptor, G-protein linked receptor, nuclear hormone receptor, G-protein, adaptor protein, scaffolding protein, SH2 domain, MAPK, Ras,
Chapter 15: Signal transduction Know the terminology: Enzyme-linked receptor, G-protein linked receptor, nuclear hormone receptor, G-protein, adaptor protein, scaffolding protein, SH2 domain, MAPK, Ras,
Molecular Cell Biology - Problem Drill 19: Cell Signaling Pathways and Gene Expression
 Molecular Cell Biology - Problem Drill 19: Cell Signaling Pathways and Gene Expression Question No. 1 of 10 1. Which statement about cell signaling is correct? Question #1 (A) Cell signaling involves receiving
Molecular Cell Biology - Problem Drill 19: Cell Signaling Pathways and Gene Expression Question No. 1 of 10 1. Which statement about cell signaling is correct? Question #1 (A) Cell signaling involves receiving
SUPPLEMENTARY INFORMATION
 DOI:.38/ncb2822 a MTC02 FAO cells EEA1 b +/+ MEFs /DAPI -/- MEFs /DAPI -/- MEFs //DAPI c HEK 293 cells WCE N M C P AKT TBC1D7 Lamin A/C EEA1 VDAC d HeLa cells WCE N M C P AKT Lamin A/C EEA1 VDAC Figure
DOI:.38/ncb2822 a MTC02 FAO cells EEA1 b +/+ MEFs /DAPI -/- MEFs /DAPI -/- MEFs //DAPI c HEK 293 cells WCE N M C P AKT TBC1D7 Lamin A/C EEA1 VDAC d HeLa cells WCE N M C P AKT Lamin A/C EEA1 VDAC Figure
Computational Biology I LSM5191
 Computational Biology I LSM5191 Aylwin Ng, D.Phil Lecture 6 Notes: Control Systems in Gene Expression Pulling it all together: coordinated control of transcriptional regulatory molecules Simple Control:
Computational Biology I LSM5191 Aylwin Ng, D.Phil Lecture 6 Notes: Control Systems in Gene Expression Pulling it all together: coordinated control of transcriptional regulatory molecules Simple Control:
Supplementary Materials for
 www.sciencesignaling.org/cgi/content/full/8/398/rs12/dc1 Supplementary Materials for Quantitative phosphoproteomics reveals new roles for the protein phosphatase PP6 in mitotic cells Scott F. Rusin, Kate
www.sciencesignaling.org/cgi/content/full/8/398/rs12/dc1 Supplementary Materials for Quantitative phosphoproteomics reveals new roles for the protein phosphatase PP6 in mitotic cells Scott F. Rusin, Kate
Nature Immunology doi: /ni.3268
 Supplementary Figure 1 Loss of Mst1 and Mst2 increases susceptibility to bacterial sepsis. (a) H&E staining of colon and kidney sections from wild type and Mst1 -/- Mst2 fl/fl Vav-Cre mice. Scale bar,
Supplementary Figure 1 Loss of Mst1 and Mst2 increases susceptibility to bacterial sepsis. (a) H&E staining of colon and kidney sections from wild type and Mst1 -/- Mst2 fl/fl Vav-Cre mice. Scale bar,
WHY TARGETTING SIGNALLING PATHWAYS?
 WHY TARGETTING SIGNALLING PATHWAYS? Cancer cells are particularly sensitive to stress therefore sensitive to inhibition of their hyper activated signaling proteins the re instatement of lost tumor suppressors.
WHY TARGETTING SIGNALLING PATHWAYS? Cancer cells are particularly sensitive to stress therefore sensitive to inhibition of their hyper activated signaling proteins the re instatement of lost tumor suppressors.
A Novel mtor-regulated Phosphorylation Site in Elongation Factor 2 Kinase Modulates the Activity of the Kinase and Its Binding to Calmodulin
 REFERENCES CONTENT ALERTS A Novel mtor-regulated Phosphorylation Site in Elongation Factor 2 Kinase Modulates the Activity of the Kinase and Its Binding to Calmodulin Gareth J. Browne and Christopher G.
REFERENCES CONTENT ALERTS A Novel mtor-regulated Phosphorylation Site in Elongation Factor 2 Kinase Modulates the Activity of the Kinase and Its Binding to Calmodulin Gareth J. Browne and Christopher G.
 Many Forms of Cell-Cell Communication Regulate Tissue Function and Phenotype Physiological Functions of Gap Junctions Homeostasis buffering/sharing of ions, nutrients, and water Metabolic support nutrient
Many Forms of Cell-Cell Communication Regulate Tissue Function and Phenotype Physiological Functions of Gap Junctions Homeostasis buffering/sharing of ions, nutrients, and water Metabolic support nutrient
Gankyrin plays an essential role in Ras-induced tumorigenesis through regulation of the RhoA/ROCK pathway in mammalian cells
 Research article Gankyrin plays an essential role in Ras-induced tumorigenesis through regulation of the RhoA/ROCK pathway in mammalian cells Jiang-Hong Man, Bing Liang, Yue-Xi Gu, Tao Zhou, Ai-Ling Li,
Research article Gankyrin plays an essential role in Ras-induced tumorigenesis through regulation of the RhoA/ROCK pathway in mammalian cells Jiang-Hong Man, Bing Liang, Yue-Xi Gu, Tao Zhou, Ai-Ling Li,
T H E J O U R N A L O F C E L L B I O L O G Y
 Supplemental material Jewell et al., http://www.jcb.org/cgi/content/full/jcb.201007176/dc1 T H E J O U R N A L O F C E L L B I O L O G Y Figure S1. IR Munc18c association is independent of IRS-1. (A and
Supplemental material Jewell et al., http://www.jcb.org/cgi/content/full/jcb.201007176/dc1 T H E J O U R N A L O F C E L L B I O L O G Y Figure S1. IR Munc18c association is independent of IRS-1. (A and
(a) Schematic diagram of the FS mutation of UVRAG in exon 8 containing the highly instable
 Supplementary Figure 1. Frameshift (FS) mutation in UVRAG. (a) Schematic diagram of the FS mutation of UVRAG in exon 8 containing the highly instable A 10 DNA repeat, generating a premature stop codon
Supplementary Figure 1. Frameshift (FS) mutation in UVRAG. (a) Schematic diagram of the FS mutation of UVRAG in exon 8 containing the highly instable A 10 DNA repeat, generating a premature stop codon
Calcium-Mediated Apoptosis and Apoptotic Sensitization in Prostate Cancer. CONTRACTING ORGANIZATION: M.D. Anderson Cancer Center Houston, TX 77030
 AD Award Number: DAMD17-01-1-0070 TITLE: Calcium-Mediated Apoptosis and Apoptotic Sensitization in Prostate Cancer PRINCIPAL INVESTIGATOR: Nicholas J. Donato, Ph.D. CONTRACTING ORGANIZATION: M.D. Anderson
AD Award Number: DAMD17-01-1-0070 TITLE: Calcium-Mediated Apoptosis and Apoptotic Sensitization in Prostate Cancer PRINCIPAL INVESTIGATOR: Nicholas J. Donato, Ph.D. CONTRACTING ORGANIZATION: M.D. Anderson
Cell signaling. How do cells receive and respond to signals from their surroundings?
 Cell signaling How do cells receive and respond to signals from their surroundings? Prokaryotes and unicellular eukaryotes are largely independent and autonomous. In multicellular organisms there is a
Cell signaling How do cells receive and respond to signals from their surroundings? Prokaryotes and unicellular eukaryotes are largely independent and autonomous. In multicellular organisms there is a
DAP Kinase A Proapoptotic Gene That Functions as a Tumor Suppressor
 Experimental Cell Research 264, 185 192 (2001) doi:10.1006/excr.2000.5134, available online at http://www.idealibrary.com on DAP Kinase A Proapoptotic Gene That Functions as a Tumor Suppressor Tal Raveh
Experimental Cell Research 264, 185 192 (2001) doi:10.1006/excr.2000.5134, available online at http://www.idealibrary.com on DAP Kinase A Proapoptotic Gene That Functions as a Tumor Suppressor Tal Raveh
GENETIC ANALYSIS OF RAS SIGNALING PATHWAYS IN CELL PROLIFERATION, MIGRATION AND SURVIVAL
 Manuscript EMBO-2009-72496 GENETIC ANALYSIS OF RAS SIGNALING PATHWAYS IN CELL PROLIFERATION, MIGRATION AND SURVIVAL Matthias Drosten, Alma Dhawahir, Eleanor Sum, Jelena Urosevic, Carmen Lechuga, Luis Esteban,
Manuscript EMBO-2009-72496 GENETIC ANALYSIS OF RAS SIGNALING PATHWAYS IN CELL PROLIFERATION, MIGRATION AND SURVIVAL Matthias Drosten, Alma Dhawahir, Eleanor Sum, Jelena Urosevic, Carmen Lechuga, Luis Esteban,
4.2 RESULTS AND DISCUSSION
 phosphorylated proteins on treatment with Erlotinib.This chapter describes the finding of the SILAC experiment. 4.2 RESULTS AND DISCUSSION This is the first global report of its kind using dual strategies
phosphorylated proteins on treatment with Erlotinib.This chapter describes the finding of the SILAC experiment. 4.2 RESULTS AND DISCUSSION This is the first global report of its kind using dual strategies
supplementary information
 DOI: 10.1038/ncb1875 Figure S1 (a) The 79 surgical specimens from NSCLC patients were analysed by immunohistochemistry with an anti-p53 antibody and control serum (data not shown). The normal bronchi served
DOI: 10.1038/ncb1875 Figure S1 (a) The 79 surgical specimens from NSCLC patients were analysed by immunohistochemistry with an anti-p53 antibody and control serum (data not shown). The normal bronchi served
Signal Transduction Pathways Involved in Phosphorylation and
 JBC Papers in Press. Published on March 28, 2001 as Manuscript M009047200 Signal Transduction Pathways Involved in Phosphorylation and Activation of p70 S6K Following Exposure to UVA Irradiation* Yiguo
JBC Papers in Press. Published on March 28, 2001 as Manuscript M009047200 Signal Transduction Pathways Involved in Phosphorylation and Activation of p70 S6K Following Exposure to UVA Irradiation* Yiguo
hexahistidine tagged GRP78 devoid of the KDEL motif (GRP78-His) on SDS-PAGE. This
 SUPPLEMENTAL FIGURE LEGEND Fig. S1. Generation and characterization of. (A) Coomassie staining of soluble hexahistidine tagged GRP78 devoid of the KDEL motif (GRP78-His) on SDS-PAGE. This protein was expressed
SUPPLEMENTAL FIGURE LEGEND Fig. S1. Generation and characterization of. (A) Coomassie staining of soluble hexahistidine tagged GRP78 devoid of the KDEL motif (GRP78-His) on SDS-PAGE. This protein was expressed
Control of Cell Proliferation by Peptide Growth Factors. Autocrine Growth Factor Production Causes Malignant Transformation?
 Control of Cell Proliferation by Peptide Growth Factors Autocrine Growth Factor Production Causes Malignant Transformation? Transforming Activities From Condition Media from a Tumor Cell Line Condition
Control of Cell Proliferation by Peptide Growth Factors Autocrine Growth Factor Production Causes Malignant Transformation? Transforming Activities From Condition Media from a Tumor Cell Line Condition
(A) SW480, DLD1, RKO and HCT116 cells were treated with DMSO or XAV939 (5 µm)
 Supplementary Figure Legends Figure S1. Tankyrase inhibition suppresses cell proliferation in an axin/β-catenin independent manner. (A) SW480, DLD1, RKO and HCT116 cells were treated with DMSO or XAV939
Supplementary Figure Legends Figure S1. Tankyrase inhibition suppresses cell proliferation in an axin/β-catenin independent manner. (A) SW480, DLD1, RKO and HCT116 cells were treated with DMSO or XAV939
Properties of Allosteric Enzymes
 Properties of Allosteric Enzymes (1) An allosteric enzyme possesses at least spatially distinct binding sites on the protein molecules the active or the catalytic site and the regulator or the allosteric
Properties of Allosteric Enzymes (1) An allosteric enzyme possesses at least spatially distinct binding sites on the protein molecules the active or the catalytic site and the regulator or the allosteric
Supplementary Information Supplementary Fig. 1. Elevated Usp9x in melanoma and NRAS mutant melanoma cells are dependent on NRAS for 3D growth.
 Supplementary Information Supplementary Fig. 1. Elevated Usp9x in melanoma and NRAS mutant melanoma cells are dependent on NRAS for 3D growth. a. Immunoblot for Usp9x protein in NRAS mutant melanoma cells
Supplementary Information Supplementary Fig. 1. Elevated Usp9x in melanoma and NRAS mutant melanoma cells are dependent on NRAS for 3D growth. a. Immunoblot for Usp9x protein in NRAS mutant melanoma cells
Revision. camp pathway
 االله الرحمن الرحيم بسم Revision camp pathway camp pathway Revision camp pathway Adenylate cyclase Adenylate Cyclase enzyme Adenylate cyclase catalyses the formation of camp from ATP. Stimulation or inhibition
االله الرحمن الرحيم بسم Revision camp pathway camp pathway Revision camp pathway Adenylate cyclase Adenylate Cyclase enzyme Adenylate cyclase catalyses the formation of camp from ATP. Stimulation or inhibition
SUPPLEMENTARY INFORMATION
 Figure S1 Treatment with both Sema6D and Plexin-A1 sirnas induces the phenotype essentially identical to that induced by treatment with Sema6D sirna alone or Plexin-A1 sirna alone. (a,b) The cardiac tube
Figure S1 Treatment with both Sema6D and Plexin-A1 sirnas induces the phenotype essentially identical to that induced by treatment with Sema6D sirna alone or Plexin-A1 sirna alone. (a,b) The cardiac tube
BCR-ABL uncouples canonical JAK2-STAT5 signaling in chronic myeloid
 Supplementary Results BCR-ABL uncouples canonical JAK2-STAT5 signaling in chronic myeloid leukemia Oliver Hantschel*, Wolfgang Warsch*, Eva Eckelhart*, Ines Kaupe, Florian Grebien, Kay-Uwe Wagner, Giulio
Supplementary Results BCR-ABL uncouples canonical JAK2-STAT5 signaling in chronic myeloid leukemia Oliver Hantschel*, Wolfgang Warsch*, Eva Eckelhart*, Ines Kaupe, Florian Grebien, Kay-Uwe Wagner, Giulio
Revisiting post-translational regulation of cell cycle CDKs
 Revisiting post-translational regulation of cell cycle CDKs Cyclin-CDK complexes during cell cycle Cyclin B-CDK1 G0 E2F prb Cyclin A-CDK1 G2 M Cyclin D-CDK4 CDK6 + prb Cyclin A-CDK2 S R G1 Cyclin E-CDK2
Revisiting post-translational regulation of cell cycle CDKs Cyclin-CDK complexes during cell cycle Cyclin B-CDK1 G0 E2F prb Cyclin A-CDK1 G2 M Cyclin D-CDK4 CDK6 + prb Cyclin A-CDK2 S R G1 Cyclin E-CDK2
CONTRACTING ORGANIZATION: Rush University Medical Center Chicago, IL 60612
 AD Award Number: W81XWH-11-1-0302 TITLE: Yin and Yang of Heparanase in breast Tumor Initiation PRINCIPAL INVESTIGATOR: Xiulong Xu, Ph.D. CONTRACTING ORGANIZATION: Rush University Medical Center Chicago,
AD Award Number: W81XWH-11-1-0302 TITLE: Yin and Yang of Heparanase in breast Tumor Initiation PRINCIPAL INVESTIGATOR: Xiulong Xu, Ph.D. CONTRACTING ORGANIZATION: Rush University Medical Center Chicago,
Lecture #27 Lecturer A. N. Koval
 Lecture #27 Lecturer A. N. Koval Hormones Transduce Signals to Affect Homeostatic Mechanisms Koval A. (C), 2011 2 Lipophilic hormones Classifying hormones into hydrophilic and lipophilic molecules indicates
Lecture #27 Lecturer A. N. Koval Hormones Transduce Signals to Affect Homeostatic Mechanisms Koval A. (C), 2011 2 Lipophilic hormones Classifying hormones into hydrophilic and lipophilic molecules indicates
Supplementary Figure 1
 Supplementary Figure 1 YAP negatively regulates IFN- signaling. (a) Immunoblot analysis of Yap knockdown efficiency with sh-yap (#1 to #4 independent constructs) in Raw264.7 cells. (b) IFN- -Luc and PRDs
Supplementary Figure 1 YAP negatively regulates IFN- signaling. (a) Immunoblot analysis of Yap knockdown efficiency with sh-yap (#1 to #4 independent constructs) in Raw264.7 cells. (b) IFN- -Luc and PRDs
