II. Characterization of the MMP activity that disrupts the DG complex
|
|
|
- Griffin Charles
- 5 years ago
- Views:
Transcription
1 Matrix Metalloproteinase Activity that Disrupts the Dystroglycan Complex: Its Role in the Molecular Pathogenesis of Muscular Dystrophies Kiichiro Matsumura, Ken Arai, Di Zhong, Fumiaki Saito, Hiroko Fukuta-Ohi, Ryuji Maekawa 1, Hiroki Yamada and Teruo Shimizu Department of Neurology and Neuroscience, Teikyo University School of Medicine, Tokyo, Japan, (1) Shionogi Research Laboratories, Shionogi & Co. Ltd., Osaka, Japan Abstract The dystroglycan (DG) complex, composed of two subunits αdg and βdg, interacts with the sarcoglycan complex to form the dystrophin-glycoprotein complex. αdg is a cell surface peripheral membrane protein which binds to the components of the extracellular matrix, while βdg is a type I integral membrane protein which anchors αdg to the cell membrane via the N-terminal extracellular domain. Although defects of the DG gene have not been identified as the primary causes of hereditary diseases in humans, secondary but significant abnormalities of the DG complex have been revealed in a number of muscular dystrophies. In this article, we characterize the matrix metalloproteinase (MMP) activity that disrupts the DG complex by cleaving the extracellular domain of βdg and discuss if this MMP plays a role in the molecular pathogenesis of muscular dystrophies. We also address the therapeutic potential of the drugs that inhibit this MMP activity to decelerate muscle degeneration in these diseases. Key Words: Dystroglycan; Matrix metalloproteinase; Extracellular matrix; Cardiomyopathic hamster; Sarcoglycanopathy. Basic Appl Myol 13 (6): , 2003 I. Introduction: Molecular organization of the dystroglycan (DG) complex The DG complex is a cell membrane-spanning complex composed of two subunits αdg and βdg [13]. In muscle, the DG complex interacts with the sarcoglycan complex to form the dystrophinglycoprotein complex. αdg is a cell surface peripheral membrane protein which binds to the components of the basement membrane, laminin, agrin and perlecan, while βdg is a type I integral membrane protein which anchors αdg to the cell membrane via the N-terminus of the extracellular domain and binds to the cytoskeletal protein dystrophin via the C-terminal cytoplasmic domain [8, 13, 20, 27, 29]. Thus, the DG complex provides a tight link between the extracellular matrix (ECM) and intracellular cytoskeleton via the cell membrane. At present, the role of the DG complex in the assembly and maintenance of the basement membrane remains controversial [7, 11, 15, 30]. These recent findings indicate that the DG complex needs to be disrupted efficiently when tissue remodeling takes place in various conditions and suggest that a specific device may exist for this purpose [31]. As such a device, we have recently identified a matrix metalloproteinase (MMP) activity that cleaves the extracellular domain of βdg [31]. In this paper, we characterize this MMP activity and discuss its role in the molecular pathogenesis of muscular dystrophies. We also address the therapeutic potential of MMP inhibitors to decelerate muscle degeneration in these diseases. II. Characterization of the MMP activity that disrupts the DG complex In skeletal muscle, βdg is detected as a single 43-kDa band (βdg full ) by immunoblot analysis using the monoclonal antibody 43DAG/8D5 against the C- terminal cytoplasmic tail of βdg [1, 31] (fig. 1). 43DAG/8D5 also detects a 30-kDa fragment of βdg (βdg 30 ) in several non-muscle tissues [31]. We clarified the tissue distribution of βdg 30 by immunoblot analysis of various bovine tissues using 43DAG/8D5. βdg 30 was detected in peripheral nerve, smooth muscle, lung and kidney, whereas it was obscure or undetectable in cardiac muscle, skeletal muscle, cerebrum and cerebellum [31]. 299
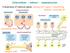
2 Fig. 1. Schematic model of the MMP activity (DG- MMP) that disrupts the DG complex by cleaving the extracellular domain of βdg. To compare the biochemical properties of βdg 30 with βdg full, we performed extraction analysis of the crude membranes of bovine peripheral nerve and rat RT4 schwannoma cells, which express both βdg full and βdg 30 [31]. Although βdg full and βdg 30 were both extracted by 2% Triton X-100, they were not extracted by ph 11 or by 10 mm EDTA [31]. Because βdg is a type I integral membrane protein having a single transmembrane domain, these results indicate that βdg 30 retains this transmembrane domain. Also because βdg 30 is recognized by 43DAG/8D5 directed against the C-terminus of the cytoplasmic domain of βdg, these results indicate that the predicted cleavage site exists in the extracellular domain of βdg and βdg 30 is its C-terminal fragment. To test the hypothesis that MMP may be responsible for the processing of βdg full into βdg 30, we cultured RT4 cells in the presence or absence of N-biphenylsulfonyl-phenylalanine hydroxamic acid (BPHA), which is a highly specific hydroxamate MMP inhibitor [19], harvested the living cells and performed immunoblot analysis using 43DAG/8D5. βdg 30 decreased with increasing concentrations of BPHA [31]. We also tested the effects of 1,10-phenanthroline, which is a transition metal ion chelator and well established as a MMP inhibitor. Because RT4 cells did not grow well in the presence of 1,10-phenanthroline, we instead incubated the total homogenates of harvested RT4 cells in the presence of 1,10-phenanthroline at 37 C for varying time periods. 1,10-Phenanthroline inhibited the proteolysis of βdg in a concentration-dependent manner (Fig. 2). These results indicate that βdg 30 is the processing fragment by MMP and that this MMP is active for the living RT4 cells. We tentatively refer to this MMP activity as DG-MMP in this article. We next tested the effects of another newly developed hydroxamate derivative, (2R)-3-(1H-Indol-3-yl)-2-[4- (2-phenyl)-2H-tetrazol-5-yl] benzenesulfonylamino] propionoc acid (MMI-166), which has a narrower range of inhibitory activity than BPHA [18]. When RT4 cells were cultured in the presence of BPHA or MMI-166, BPHA, but not MMI-166, inhibited the proteolysis of βdg in a concentration-dependent manner (Fig. 3). In addition, BPHA, but not MMI-166, inhibited the Fig. 2. 1,10-Phenanthroline inhibits the processing of βdg by DG-MMP. After 3 days of culture, RT4 cells were harvested, homogenized and then incubated in the presence or absence of 1,10- phenanthroline at 37 C for various time periods. The homogenates were analyzed by immunoblotting using 43DAG/8D5. 1,10- Phenanthroline inhibited the proteolysis of βdg in a concentration-dependent manner. proteolysis of βdg when added to the total homogenates of harvested RT4 cells (not shown). Based on the inhibitory profiles of these reagents [18, 19], we presume that DG-MMP is distinct from MMP-1, MMP- 2/MMP-14 or MMP-9. To clarify if DG-MMP is present in RT4 cells themselves or secreted into the culture medium, RT4 cells were harvested, homogenized and then incubated at 37 C for various times. βdg full decreased and βdg 30 increased with time, indicating that DG-MMP was present in RT4 cells themselves, not secreted into the culture medium [31]. We are currently investigating the possibility that DG- MMP might be a membrane-type MMP. To know if the processing of βdg by DG-MMP affects the integrity of the DG complex, we performed sucrose density gradient sedimentation analysis of RT4 cell membranes. βdg full, but not βdg 30, co-sedimented with αdg (Fig. 4). We isolated the DG complex from the RT4 cell membranes by laminin affinity chromatography. βdg full co-isolated with αdg, which bound to laminin-sepharose directly as a lamininbinding protein [31]. However, βdg 30 did not co-isolate with αdg [31]. We isolated the DG complex from the RT4 cell membranes by wheat germ agglutinin (WGA) affinity chromatography. αdg and βdg full were completely absorbed by WGA-Sepharose and recovered in the eluates [31]. However, βdg 30 was not absorbed and undetectable in the eluates [31]. All together, these results indicate that βdg full, but not βdg 30, is complexed with αdg and thus that the MMP cleavage of βdg into βdg 30 disintegrates the DG complex. This is consistent with the report that the αdg-binding site exists in the N-terminus of the extracellular domain of βdg [27], since βdg 30 is the C-terminal fragment of the cleavage. Because αdg and βdg are responsible for the binding to the ECM and cell membrane respectively, DG-MMP disrupts the link between the ECM and cell membrane via the DG complex. 300
3 III. Implications of DG-MMP in the pathogenesis of diseases 1. Cancer invasion/metastasis and infectious diseases The aforementioned findings will have important implications in an array of pathological phenomena. For instance, it has been shown recently that certain carcinoma cell lines express β-dg 30 abundantly [17]. Taken together with our results, carcinoma cells are presumed to employ DG-MMP to disrupt the dystroglycan complex. This will enable carcinoma cells to metastasize and invade other tissues. Interestingly in this respect, BPHA has been developed as a drug to inhibit cancer spread and metastasis [18, 19]. Processing of βdg by DG-MMP may also play a role in the molecular pathogenesis of viral and bacterial infections. It has been shown recently that pathogens such as arena viruses (several strains of lymphocytic choriomeningitis virus and Lassa fever virus) and Mycobacterium leprae bind to the cell surface αdg as an initial step of host cell infection [5, 23, 26]. Therefore, DG-MMP might be a natural defense mechanism against these pathogens, in analogy to matrilysin (MMP-7), which has been shown to play a defensive role against microorganisms in mucosal epithelial cells [16]. 2. Muscular dystrophies Another situation where DG-MMP is implicated is the molecular pathogenesis of hereditary neuromuscular diseases. Over the last 10 years, primary genetic defects have been identified in a number of these diseases. However, the precise molecular pathways by which the primary defects lead to muscle cell degeneration eventually in these diseases have not necessarily been clarified. Studies to elucidate the biological functions and dysfunctions of the proteins which work in close concert with the causative proteins in vivo will be useful in this context. As such, a research on DG processing by DG-MMP could provide us precious clues concerning the molecular pathogenesis of muscular dystrophies caused by the primary defects of the components of the dystrophin-glycoprotein complex and its related proteins, because abnormalities of the DG complex are well known in theses diseases. For instance, defective glycosylation of αdg has been demonstrated in several forms of congenital muscular dystrophies, including Fukuyama-type congenital muscular dystrophy, MDC1C/LGMD2I, muscle-eyebrain disease, Walker-Warburg syndrome and Large myd mice, which are the model animals of congenital muscular dystrophy [2, 3, 9, 10, 14, 21, 32]. These diseases are all caused by the primary defects of the genes encoding glycosyltransferases, which are presumed to disturb glycosylation of αdg. Because glycosylation of αdg is crucial for the binding of laminin [6, 20], its defect is expected to perturb this binding and result in the disruption of the ECM-cell membrane linkage via the DG complex. This scenario is supported by the finding that the antibody against the carbohydrate residues of αdg involved in the binding of laminin induced a dystrophic phenotype in cultured muscle cells [4]. Another intriguing example is sarcoglycanopathy. Having the mutation of the δ-sarcoglycan gene, a genomic deletion including the first exon, cardiomyopathic hamsters are the model animals of sarcoglycanopathy LGMD2F [22, 25]. It is noteworthy that αdg has been shown to be dissociated from βdg and not recovered in the membrane fraction in the muscle of these animals [24, 28]. It has also been shown that αdg is reconstituted into the DG complex when dystrophic changes are corrected by the adenovirus transfer of the δ-sarcoglycan gene [12]. Furthermore, similar observations have been reported in other types of sarcoglycanopathies and their model animals. Based on these observations, we are currently analyzing the activity of DG-MMP in cardiomyopathic hamsters. Preliminary results indicate that DG-MMP is activated in the muscle of these animals (manuscript in preparation). We presume that activation of DG-MMP causes the disruption of the link between the ECM and Fig. 3. BPHA, but not MMI-166, inhibits the processing of βdg by DG-MMP. Various concentrations of BPHA or MMI-166 were added to the culture medium of RT4 cells. After 3 days of culture, cells were harvested, homogenized and analyzed by immunoblotting using 43DAG/8D5. While BPHA inhibited the proteolysis of βdg in a concentration-dependent manner, MMI-166 did not. Fig. 4. βdg full, but not βdg 30, co-sediments with αdg. Sucrose density gradient sedimentation of RT4 cell membranes was performed and analyzed by immunoblotting using 43DAG/8D5. βdg full, but not βdg 30, co-sedimented with αdg. 301
4 Labor and Welfare), [2] Health Sciences Research Grant H12-Brain-017 for Research on Brain Science (Ministry of Health, Labor and Welfare), [3] Research Grants and (Ministry of Education, Sports, Culture, Science and Technology) and [4] a research grant from Toyota Physical & Chemical Research Institute. Address correspondence to: Kiichiro Matsumura, Department of Neurology and Neuroscience, Teikyo University School of Medicine, Tokyo, Japan, Tel: ext Fax: k-matsu@med.teikyo-u.ac.jp References Fig. 5. Hypothetical scheme of the role of DG-MMP in the molecular pathogenesis of muscular dystrophies. DG-MMP is hypothesized to be activated by the initial muscle cell damage in certain muscular dystrophies. This results in the disruption of the ECM-cell membrane linkage via the DG complex, which, in turn, destabilizes the sarcolemma and further augments muscle cell damage in a vicious cycle. cell membrane via the DG complex in the muscle of cardiomyopathic hamsters and this may play an important role in the pathogenesis of muscle degeneration. IV. Therapeutic implications of DG-MMP: do inhibitors of DG-MMP decelerate muscle degeneration in muscular dystrophies? If DG-MMP turns out to be activated in certain muscular dystrophies, this will have significant implications for the molecular pathogenesis of muscle degeneration in these diseases, because the resulting disruption of the ECM-cell membrane linkage via the DG complex is expected to further augment muscle cell damage in a vicious cycle (Fig. 5). Moreover, it will raise the intriguing possibility of a novel pharmacological therapy for these diseases. The MMP inhibitors effective against DG-MMP have been developed as anti-cancer reagents and demonstrated to be not only effective but also safe without serious side effects when administered orally [19]. We are currently planning the oral administration of DG-MMP inhibitors to cardiomyopathic hamsters to see their effects on the progression of muscle degeneration. Acknowledgments. We thank Miki Yamanaka and Yuka Sasayama for their expert technical assistance. This work was supported by [1] Research Grants 13B-3 and 14B-4 for Nervous and Mental Disorders (Ministry of Health, [1] Anderson LVB, Davison K: Multiplex western blotting system for the analysis of muscular dystrophy patients. Am J Pathol 1999; 154: [2] Beltran-Valero De Bernabe D, Currier S, Steinbrecher A, Celli J, Van Beusekom E, Van Der Zwaag B, Kayserili H, Merlini L, Chitayat D, Dobyns WB, Cormand B, Lehesjoki AE, Cruces J, Voit T, Walsh CA, Van Bokhoven H, Brunner HG: Mutations in the O- mannosyltransferase gene POMT1 give rise to the severe neuronal migration disorder Walker- Warburg syndrome. Am J Hum Genet 2002; epub ahead of print. [3] Brockington M, Blake DJ, Prandini P, Brown SC, Torelli S, Benson MA, Ponting CP, Estournet B, Romero NB, Mercuri E, Voit T, Sewry CA, Guicheney P, Muntoni F: Mutations in the fukutin-related protein gene (FKRP) cause a form of congenital muscular dystrophy with secondary laminin α2 deficiency and abnormal glycosylation of α-dystroglycan. Am J Hum Genet 2001; 69: [4] Brown SC, Fassati A, Popplewell L, Page AM, Henry MD, Campbell KP, Dickson G: Dystrophic phenotype induced in vitro by antibody blockade of muscle α-dystroglycanlaminin interaction. J Cell Sci 1999; 112: [5] Cao W, Henry MD, Borrow P, Yamada H, Elder JH, Ravkov EV, Nichol ST, Compans RW, Campbell KP, Oldstone MBA: Identification of α-dystroglycan as a receptor for lymphocytic choriomeningitis virus and Lassa fever virus. Science 1998; 282: [6] Chiba A, Matsumura K, Yamada H, Inazu T, Shimizu T, Kusunoki S, Kanazawa I, Kobata A, Endo T: Structures of sialylated O-linked oligosaccharides of bovine peripheral nerve α- dystroglycan: the role of a novel mannosyl type 302
5 oligosaccharide in the binding with laminin. J Biol Chem 1997; 272: [7] Colognato H, Winkelmann DA, Yurchenco PD: Laminin polymerization induces a receptorcytoskeleton network. J Cell Biol 1999; 145: [8] Ervasti JM, Campbell KP: Membrane organization of the dystrophin-glycoprotein complex. Cell 1991; 66: [9] Grewal PK, Holzfeind PJ, Bittner RE, Hewitt JE: Mutant glycosyltransferase and altered glycosylation of alpha-dystroglycan in the myodystrophy mouse. Nat Genet 2001; 28: [10] Hayashi YK, Ogawa M, Tagawa K, Noguchi S, Ishihara T, Nonaka I, Arahata K: Selective deficiency of α-dystroglycan in Fukuyama-type congenital muscular dystrophy. Neurology 2001; 57: [11] Henry MD, Campbell KP: A role for dystroglycan in basement membrane assembly. Cell 1998; 95: [12] Holt KH, Lim LE, Straub V, Venske DP, Duclos F, Anderson RD, Davidson BL, Campbell KP: Functional rescue of the sarcoglycan complex in the BIO 14.6 hamster using δ-sarcoglycan gene transfer. Molec Cell 1998; 1: [13] Ibraghimov-Beskrovnaya O, Ervasti JM, Leveille CJ, Slaughter CA, Sernett SW, Campbell KP: Primary structure of dystrophin-associated glycoproteins linking dystrophin to the extracellular matrix. Nature 1992; 355: [14] Kano H, Kobayashi K, Herrmann R, Tachikawa M, Manya H, Nishino I, Nonaka I, Straub V, Talim B, Voit T, Topaloglu H, Endo T, Yoshikawa H, Toda T: Deficiency of α- dystroglycan in muscle-eye-brain disease. Biochem Biophys Res Commun 2002; 291: [15] Li S, Harrison D, Carbonetto S, Fassler R, Smyth N, Edgar D, Yurchenco PD. Matrix assembly, regulation, and survival functions of laminin and its receptors in embryonic stem cell differentiation. J Cell Biol 2002; 157: [16] López-Boado YS, Wilson CL, Hooper LV, Gordon JI, Hultgren SJ, Parks WC: Bacterial exposure induces and activates matrilysin in mucosal epithelial cells. J Cell Biol 2000; 148: [17] Losasso C, Tommaso FD, Sgambato A, Ardito R, Cittandini A, Giardina B, Petrucci TC, Brancaccio A: Anomalous dystroglycan in carcinoma cell lines. FEBS Lett 2000; 484: [18] Maekawa R, Maki H, Wada T, Yoshida H, Nishida-Nishimoto K, Okamoto H, Matsumoto Y, Tsuzuki H, Yoshioka T: Anti-metastatic efficacy and safety of MMI-166, a selective matrix metalloproteinase inhibitor. Clin Exp Metastasis 2000; 18: [19] Maekawa R, Maki H, Yoshida H, Hojo K, Tanaka H, Wada T, Uchida N, Takeda Y, Kasai H, Okamoto H, Tsuzuki H, Kambayashi Y, Watanabe F, Kawada K, Toda K, Ohtani M, Sugita K, Yoshioka T: Correlation of antiangiogenic and antitumor efficacy of N- biphenylo sulfonyl-phenlalanine hydroxiamic acid (BPHA), an orally-active, selective matrix metalloproteinase inhibitor. Cancer Res 1999; 59: [20] Matsumura K, Saito F, Yamada H, Hase A, Sunada Y, Shimizu T: Sarcoglycan complex: a muscular supporter of dystroglycan-dystrophin interplay? Cell Molec Biol 1999; 45: [21] Michele DE, Barresi R, Kanagawa M, Saito F, Cohn RD, Satz JS, Dollar J, Nishino I, Kelley RI, Somer H, Straub V, Mathews KD, Moore SA, Campbell KP: Post-translational disruption of dystroglycan ligand interactions in congenital muscular dystrophies. Nature 2002; 418: [22] Nigro V, Okazaki Y, Belsito A, Piluso G, Matsuda Y, Politano L, Nigro G, Ventura C, Abbondanza C, Molinari AM, Acampora D, Nishimura M, Hayashizaki Y, Puca GA: Identification of the Syrian hamster cardiomyopathy gene. Hum Molec Genet 1997; 6: [23] Rambukkana A, Yamada H, Zanazzi G, Mathus T, Salzer JL, Yurchenco PD, Campbell KP, Fischetti VA: Role of α-dystroglycan as a Schwann cell receptor for Mycobacterium leprae. Science 1998; 282: [24] Roberds SL, Ervasti JM, Anderson RD, Ohlendieck K, Kahl SD, Zoloto D, Campbell KP: Disruption of the dystrophin-glycoprotein complex in the cardiomyopathic hamster. J Biol Chem 1993; 268: [25] Sakamoto A, Ono K, Abe M, Jasmin G, Eki T, Murakami Y, Masaki T, Toyo-oka T, Hanaoka F: Both hypertrophic and dilated cardiomyopathies are caused by mutation of the same gene, δ- sarcoglycan, in hamster: an animal model of disrupted dystrophin-associated glycoprotein complex. Proc Natl Acad Sci USA 1997; 94: [26] Sevilla N, Kunz S, Holz A, Lewicki H, Homann D, Yamada H, Campbell KP, de La Torre JC, Oldstone MB: Immunosuppression and resultant viral persistence by specific viral targeting of dendritic cells. J Exp Med 2000; 192: [27] Stasio ED, Sciandra F, Maras B, Tommaqso FD, Petrucci TC, Giardina B, Brancaccio A: 303
6 Structural and functional analysis of the N- terminal extracellular region of β-dystroglycan. Biochem Biophys Res Commun 1999; 206: [28] Straub V, Duclos F, Venzke DP, Lee JC, Cutshall S, Leveille CJ, Campbell KP: Molecular pathognesis of muscle degeneration in the δ- sarcoglycan-deficient hamster. Am J Pathol 1998; 153: [29] Suzuki A, Yoshida M, Hayashi K, Mizuno Y, Hagiwara Y, Ozawa E: Molecular organization at the glycoprotein-complex-binding-site of dystrophin. Three dystrophin-associated proteins bind directly to the carboxy-terminal portion of dystrophin. Eur J Biochem 1994; 220: [30] Williamson RA, Henry MD, Daniels KJ, Hrstka RF, Lee JC, Sunada Y, Ibraghimov-Beskrovnaya O, Campbell KP: Dystroglycan is essential for early embryonic development: disruption of Reichert's membrane in Dag1-null mice. Hum Molec Genet 1997; 6: [31] Yamada H, Saito F, Fukuta-Ohi H, Zhong D, Hase A, Arai K, Okuyama A, Maekawa R, Shimizu T, Matsumura K: Processing of β- dystroglycan by matrix metalloproteinase disrupts the link between the extracellular matrix and cell membrane via the dystroglycan complex. Hum Molec Genet 2001; 10: [32] Yoshida A, Kobayashi K, Manya H, Taniguchi K, Kano H, Mizuno M, Inazu T, Mitsuhashi H, Takahashi S, Takeuchi M, Herrmann R, Straub V, Talim B, Voit T, Topaloglu H, Toda T, Endo T: Muscular dystrophy and neuronal migration disorder caused by mutations in a glycosyltransferase, POMGnT1. Dev Cell 2001; 1:
JBC Papers in Press. Published on February 24, 2009 as Manuscript C
 JBC Papers in Press. Published on February 24, 2009 as Manuscript C900007200 The latest version is at http://www.jbc.org/cgi/doi/10.1074/jbc.c900007200 LARGE repression in epithelium-derived cancer LOSS
JBC Papers in Press. Published on February 24, 2009 as Manuscript C900007200 The latest version is at http://www.jbc.org/cgi/doi/10.1074/jbc.c900007200 LARGE repression in epithelium-derived cancer LOSS
T he hypoglycosylation of a-dystroglycan is a new disease
 1of5 ELECTRONIC LETTER Mutations in the FKRP gene can cause muscle-eye-brain disease and Walker Warburg syndrome D Beltran-Valero de Bernabé, T Voit, C Longman, A Steinbrecher, V Straub, Y Yuva, R Herrmann,
1of5 ELECTRONIC LETTER Mutations in the FKRP gene can cause muscle-eye-brain disease and Walker Warburg syndrome D Beltran-Valero de Bernabé, T Voit, C Longman, A Steinbrecher, V Straub, Y Yuva, R Herrmann,
Three Muscular Dystrophies: Loss of Cytoskeleton-Extracellular Matrix Linkage
 Cell, Vol. 80, 675-679, March 10, 1995, Copyright 1995 by Cell Press Three Muscular Dystrophies: Loss of Cytoskeleton-Extracellular Matrix Linkage Review Kevin P. Campbell Howard Hughes Medical Institute
Cell, Vol. 80, 675-679, March 10, 1995, Copyright 1995 by Cell Press Three Muscular Dystrophies: Loss of Cytoskeleton-Extracellular Matrix Linkage Review Kevin P. Campbell Howard Hughes Medical Institute
LARGE expression in different types of muscular dystrophies other than dystroglycanopathy
 Balci-Hayta et al. BMC Neurology (2018) 18:207 https://doi.org/10.1186/s12883-018-1207-0 RESEARCH ARTICLE LARGE expression in different types of muscular dystrophies other than dystroglycanopathy Burcu
Balci-Hayta et al. BMC Neurology (2018) 18:207 https://doi.org/10.1186/s12883-018-1207-0 RESEARCH ARTICLE LARGE expression in different types of muscular dystrophies other than dystroglycanopathy Burcu
A novel POMT2 mutation causes mild congenital muscular dystrophy with normal brain MRI
 Brain & Development 31 (2009) 465 468 Case report A novel POMT2 mutation causes mild congenital muscular dystrophy with normal brain MRI Terumi Murakami a,b, Yukiko K. Hayashi a, *, Megumu Ogawa a, Satoru
Brain & Development 31 (2009) 465 468 Case report A novel POMT2 mutation causes mild congenital muscular dystrophy with normal brain MRI Terumi Murakami a,b, Yukiko K. Hayashi a, *, Megumu Ogawa a, Satoru
A Dystroglycan Mutation Associated with Limb-Girdle Muscular Dystrophy
 T h e n e w e ngl a nd j o u r na l o f m e dic i n e brief report A Dystroglycan Mutation Associated with Limb-Girdle Muscular Dystrophy Yuji Hara, Ph.D., Burcu Balci-Hayta, Ph.D., Takako Yoshida-Moriguchi,
T h e n e w e ngl a nd j o u r na l o f m e dic i n e brief report A Dystroglycan Mutation Associated with Limb-Girdle Muscular Dystrophy Yuji Hara, Ph.D., Burcu Balci-Hayta, Ph.D., Takako Yoshida-Moriguchi,
Peripheral nerve dystroglycan: its function and potential role in the molecular pathogenesis of neuromuscular diseases
 Y. Fukuyama, M. Osawa and K. Saito (Eds.), Congenital Muscular Dyrfrophies O 1997 Elsevier Science B.V. All rights reserved CHAPTER 22 Peripheral nerve dystroglycan: its function and potential role in
Y. Fukuyama, M. Osawa and K. Saito (Eds.), Congenital Muscular Dyrfrophies O 1997 Elsevier Science B.V. All rights reserved CHAPTER 22 Peripheral nerve dystroglycan: its function and potential role in
Muscular Dystrophy. Biol 405 Molecular Medicine
 Muscular Dystrophy Biol 405 Molecular Medicine Duchenne muscular dystrophy Duchenne muscular dystrophy is a neuromuscular disease that occurs in ~ 1/3,500 male births. The disease causes developmental
Muscular Dystrophy Biol 405 Molecular Medicine Duchenne muscular dystrophy Duchenne muscular dystrophy is a neuromuscular disease that occurs in ~ 1/3,500 male births. The disease causes developmental
Fukutin Gene Mutations Cause Dilated Cardiomyopathy with Minimal Muscle Weakness
 Fukutin Gene Mutations Cause Dilated Cardiomyopathy with Minimal Muscle Weakness Terumi Murakami, MD, 1,2 Yukiko K. Hayashi MD, PhD, 1 Satoru Noguchi, PhD 1,2 Megumu Ogawa, BS 1 Ikuya Nonaka, MD, PhD,
Fukutin Gene Mutations Cause Dilated Cardiomyopathy with Minimal Muscle Weakness Terumi Murakami, MD, 1,2 Yukiko K. Hayashi MD, PhD, 1 Satoru Noguchi, PhD 1,2 Megumu Ogawa, BS 1 Ikuya Nonaka, MD, PhD,
The genetic and molecular basis of muscular dystrophy: roles of cell matrix linkage in the pathogenesis
 J Hum Genet (2006) 51:915 926 DOI 10.1007/s10038-006-0056-7 MINIREVIEW The genetic and molecular basis of muscular dystrophy: roles of cell matrix linkage in the pathogenesis Motoi Kanagawa Æ Tatsushi
J Hum Genet (2006) 51:915 926 DOI 10.1007/s10038-006-0056-7 MINIREVIEW The genetic and molecular basis of muscular dystrophy: roles of cell matrix linkage in the pathogenesis Motoi Kanagawa Æ Tatsushi
Author Manuscript Faculty of Biology and Medicine Publication
 Serveur Académique Lausannois SERVAL serval.unil.ch Author Manuscript Faculty of Biology and Medicine Publication This paper has been peer-reviewed but dos not include the final publisher proof-corrections
Serveur Académique Lausannois SERVAL serval.unil.ch Author Manuscript Faculty of Biology and Medicine Publication This paper has been peer-reviewed but dos not include the final publisher proof-corrections
Mechanisms of Disease: congenital muscular dystrophies glycosylation takes center stage
 Mechanisms of Disease: congenital muscular dystrophies glycosylation takes center stage Paul T Martin SUMMARY Recent studies have defined a group of muscular dystrophies, now termed the dystroglycanopathies,
Mechanisms of Disease: congenital muscular dystrophies glycosylation takes center stage Paul T Martin SUMMARY Recent studies have defined a group of muscular dystrophies, now termed the dystroglycanopathies,
READ ORPHA.NET WEBSITE ABOUT BETA-SARCOGLYOCANOPATHY LIMB-GIRDLE MUSCULAR DYSTROPHIES
 READ ORPHA.NET WEBSITE ABOUT BETA-SARCOGLYOCANOPATHY LIMB-GIRDLE MUSCULAR DYSTROPHIES (LGMD) Limb-girdle muscular dystrophies (LGMD) are a heterogeneous group of genetically determined disorders with a
READ ORPHA.NET WEBSITE ABOUT BETA-SARCOGLYOCANOPATHY LIMB-GIRDLE MUSCULAR DYSTROPHIES (LGMD) Limb-girdle muscular dystrophies (LGMD) are a heterogeneous group of genetically determined disorders with a
In cardiac muscle, the dystrophin glycoprotein complex includes
 Dissociation of Sarcoglycans and the Dystrophin Carboxyl Terminus From the Sarcolemma in Enteroviral Cardiomyopathy Gil-Hwan Lee,* Cornel Badorff,* Kirk U. Knowlton Abstract Enteroviral infection can cause
Dissociation of Sarcoglycans and the Dystrophin Carboxyl Terminus From the Sarcolemma in Enteroviral Cardiomyopathy Gil-Hwan Lee,* Cornel Badorff,* Kirk U. Knowlton Abstract Enteroviral infection can cause
Progress in human genetics has identified a number. -Sarcoglycan Deficiency Leads to Muscle Membrane Defects and Apoptosis Independent of Dystrophin
 -Sarcoglycan Deficiency Leads to Muscle Membrane Defects and Apoptosis Independent of Dystrophin Andrew A. Hack,* Chantal T. Ly, Fang Jiang, Cynthia J. Clendenin, Kirsten S. Sigrist, Robert L. Wollmann,
-Sarcoglycan Deficiency Leads to Muscle Membrane Defects and Apoptosis Independent of Dystrophin Andrew A. Hack,* Chantal T. Ly, Fang Jiang, Cynthia J. Clendenin, Kirsten S. Sigrist, Robert L. Wollmann,
Aberrant neuromuscular junctions and delayed terminal muscle fiber maturation
 HMG Advance Access published March 10, 2006 Aberrant neuromuscular junctions and delayed terminal muscle fiber maturation in α-dystroglycanopathies Mariko Taniguchi 1, Hiroki Kurahashi 2, Satoru Noguchi
HMG Advance Access published March 10, 2006 Aberrant neuromuscular junctions and delayed terminal muscle fiber maturation in α-dystroglycanopathies Mariko Taniguchi 1, Hiroki Kurahashi 2, Satoru Noguchi
The role of dystroglycan in the prostate epithelium and prostate cancer
 University of Iowa Iowa Research Online Theses and Dissertations Fall 2011 The role of dystroglycan in the prostate epithelium and prostate cancer Alison K. Esser University of Iowa Copyright 2011 Alison
University of Iowa Iowa Research Online Theses and Dissertations Fall 2011 The role of dystroglycan in the prostate epithelium and prostate cancer Alison K. Esser University of Iowa Copyright 2011 Alison
K. P. Loss of sarcolemma nnos in sarcoglycandeficient
 Loss of sarcolemma nnos in sarcoglycan-deficient muscle RACHELLE H. CROSBIE 1,2, RITA BARRESI 1, AND KEVIN P. CAMPBELL 1 Howard Hughes Medical Institute, Department of Physiology and Biophysics, Department
Loss of sarcolemma nnos in sarcoglycan-deficient muscle RACHELLE H. CROSBIE 1,2, RITA BARRESI 1, AND KEVIN P. CAMPBELL 1 Howard Hughes Medical Institute, Department of Physiology and Biophysics, Department
Dystrophin-glycoprotein complex: molecular organization and critical roles in skeletal muscle
 Dystrophin-glycoprotein complex: molecular organization and critical roles in skeletal muscle Yoshihide Sunada and Kevin P. Campbell Howard Hughes Medical Institute, Department of Physiology and Biophysics,
Dystrophin-glycoprotein complex: molecular organization and critical roles in skeletal muscle Yoshihide Sunada and Kevin P. Campbell Howard Hughes Medical Institute, Department of Physiology and Biophysics,
stem cell products Basement Membrane Matrix Products Rat Mesenchymal Stem Cell Growth and Differentiation Products
 stem cell products Basement Membrane Matrix Products Rat Mesenchymal Stem Cell Growth and Differentiation Products Stem Cell Qualified Extracellular Matrix Proteins Stem cell research requires the finest
stem cell products Basement Membrane Matrix Products Rat Mesenchymal Stem Cell Growth and Differentiation Products Stem Cell Qualified Extracellular Matrix Proteins Stem cell research requires the finest
Dystrophy Patients Lacking COOH-terminal Domains of Dystrophin
 Deficiency of Dystrophin-associated Proteins in Duchenne Muscular Dystrophy Patients Lacking COOH-terminal Domains of Dystrophin Kiichiro Matsumura, * Fernando M. S. Tome,t Victor lonasescu, James M. Ervasti,
Deficiency of Dystrophin-associated Proteins in Duchenne Muscular Dystrophy Patients Lacking COOH-terminal Domains of Dystrophin Kiichiro Matsumura, * Fernando M. S. Tome,t Victor lonasescu, James M. Ervasti,
Dubowitz Neuromuscular Centre, Institute of Child Health and Great Ormond Street Hospital for Children, UCL, London, UK. 2
 Brain Pathology ISSN 1015-6305 RESEARCH ARTICLE A Comparative Study of a-dystroglycan Glycosylation in Dystroglycanopathies Suggests that the Hypoglycosylation of a-dystroglycan Does Not Consistently Correlate
Brain Pathology ISSN 1015-6305 RESEARCH ARTICLE A Comparative Study of a-dystroglycan Glycosylation in Dystroglycanopathies Suggests that the Hypoglycosylation of a-dystroglycan Does Not Consistently Correlate
INTRODUCTION. # 2002 Oxford University Press Human Molecular Genetics, 2002, Vol. 11, No
 # 2002 Oxford University Press Human Molecular Genetics, 2002, Vol. 11, No. 21 2673 2687 Skeletal, cardiac and tongue muscle pathology, defective retinal transmission, and neuronal migration defects in
# 2002 Oxford University Press Human Molecular Genetics, 2002, Vol. 11, No. 21 2673 2687 Skeletal, cardiac and tongue muscle pathology, defective retinal transmission, and neuronal migration defects in
Congenital muscular dystrophies: New aspects of an expanding group of disorders
 Congenital muscular dystrophies: New aspects of an expanding group of disorders Matthew T. Lisia, Ronald D. Cohn To cite this version: Matthew T. Lisia, Ronald D. Cohn. Congenital muscular dystrophies:
Congenital muscular dystrophies: New aspects of an expanding group of disorders Matthew T. Lisia, Ronald D. Cohn To cite this version: Matthew T. Lisia, Ronald D. Cohn. Congenital muscular dystrophies:
Muscle Protein Alterations in LGMD2I Patients With Different Mutations in the Fukutin-related Protein Gene
 Volume 56(11): 995 1001, 2008 Journal of Histochemistry & Cytochemistry http://www.jhc.org ARTICLE Muscle Protein Alterations in LGMD2I Patients With Different Mutations in the Fukutin-related Protein
Volume 56(11): 995 1001, 2008 Journal of Histochemistry & Cytochemistry http://www.jhc.org ARTICLE Muscle Protein Alterations in LGMD2I Patients With Different Mutations in the Fukutin-related Protein
Altered membrane proteins and permeability correlate with cardiac dysfunction in cardiomyopathic hamsters
 Am J Physiol Heart Circ Physiol 278: H1362 H1370, 2000. Altered membrane proteins and permeability correlate with cardiac dysfunction in cardiomyopathic hamsters YASUHIRO IKEDA, 1 MARYANN MARTONE, 2 YUSU
Am J Physiol Heart Circ Physiol 278: H1362 H1370, 2000. Altered membrane proteins and permeability correlate with cardiac dysfunction in cardiomyopathic hamsters YASUHIRO IKEDA, 1 MARYANN MARTONE, 2 YUSU
The New England Journal of Medicine MUTATIONS IN THE SARCOGLYCAN GENES IN PATIENTS WITH MYOPATHY
 MUTATIONS IN THE SARCOGLYCAN GENES IN PATIENTS WITH MYOPATHY DAVID J. DUGGAN, B.S., J. RAFAEL GOROSPE, M.D., PH.D., MARINA FANIN, M.S., ERIC P. HOFFMAN, PH.D., A CORRADO ANGELINI, M.D. ABSTRACT Background
MUTATIONS IN THE SARCOGLYCAN GENES IN PATIENTS WITH MYOPATHY DAVID J. DUGGAN, B.S., J. RAFAEL GOROSPE, M.D., PH.D., MARINA FANIN, M.S., ERIC P. HOFFMAN, PH.D., A CORRADO ANGELINI, M.D. ABSTRACT Background
Effects and regulation of dystroglycan glycosylation in cancer
 University of Iowa Iowa Research Online Theses and Dissertations Spring 2015 Effects and regulation of dystroglycan glycosylation in cancer Michael Raymond Miller University of Iowa Copyright 2015 Michael
University of Iowa Iowa Research Online Theses and Dissertations Spring 2015 Effects and regulation of dystroglycan glycosylation in cancer Michael Raymond Miller University of Iowa Copyright 2015 Michael
The dystrophin glycoprotein complex (DGC)1 consists
 Molecular Organization of Sarcoglycan Complex in Mouse Myotubes in Culture Yiu-mo Chan,* Carsten G. Bönnemann,* Hart G.W. Lidov, and Louis M. Kunkel* *Howard Hughes Medical Institute, Division of Genetics,
Molecular Organization of Sarcoglycan Complex in Mouse Myotubes in Culture Yiu-mo Chan,* Carsten G. Bönnemann,* Hart G.W. Lidov, and Louis M. Kunkel* *Howard Hughes Medical Institute, Division of Genetics,
The addition of sugar moiety determines the blood group
 The addition of sugar moiety determines the blood group Sugars attached to glycoproteins and glycolipids on the surfaces of red blood cells determine the blood group termed A, B, and O. The A and B antigens
The addition of sugar moiety determines the blood group Sugars attached to glycoproteins and glycolipids on the surfaces of red blood cells determine the blood group termed A, B, and O. The A and B antigens
JCB. A stoichiometric complex of neurexins and dystroglycan in brain. Article
 JCB Published Online: 16 July, 2001 Supp Info: http://doi.org/10.1083/jcb.200105003 Downloaded from jcb.rupress.org on August 26, 2018 Article A stoichiometric complex of neurexins and dystroglycan in
JCB Published Online: 16 July, 2001 Supp Info: http://doi.org/10.1083/jcb.200105003 Downloaded from jcb.rupress.org on August 26, 2018 Article A stoichiometric complex of neurexins and dystroglycan in
The recruitment of leukocytes and plasma proteins from the blood to sites of infection and tissue injury is called inflammation
 The migration of a particular type of leukocyte into a restricted type of tissue, or a tissue with an ongoing infection or injury, is often called leukocyte homing, and the general process of leukocyte
The migration of a particular type of leukocyte into a restricted type of tissue, or a tissue with an ongoing infection or injury, is often called leukocyte homing, and the general process of leukocyte
Rama Abbady. Odai Bani-Monia. Diala Abu-Hassan
 5 Rama Abbady Odai Bani-Monia Diala Abu-Hassan Lipid Rafts Lipid rafts are aggregates (accumulations) of sphingolipids. They re semisolid clusters (10-200 nm) of cholesterol and sphingolipids (sphingomyelin
5 Rama Abbady Odai Bani-Monia Diala Abu-Hassan Lipid Rafts Lipid rafts are aggregates (accumulations) of sphingolipids. They re semisolid clusters (10-200 nm) of cholesterol and sphingolipids (sphingomyelin
Refining genotype ^phenotype correlations in muscular dystrophies with defective glycosylation of dystroglycan
 Brain Advance Access published September 18, 2007 doi:10.1093/brain/awm212 Brain (2007)Page1of11 Refining genotype ^phenotype correlations in muscular dystrophies with defective glycosylation of dystroglycan
Brain Advance Access published September 18, 2007 doi:10.1093/brain/awm212 Brain (2007)Page1of11 Refining genotype ^phenotype correlations in muscular dystrophies with defective glycosylation of dystroglycan
Disruption of heart sarcoglycan complex and severe cardiomyopathy caused by β sarcoglycan mutations
 102 Department of Neuromuscular Diseases, Istituto Nazionale Neurologico C Besta, Via Celoria 11, 20133 Milano, Italy R Barresi C Di Blasi T Negri R Brugnoni S Daniel F Cornelio L Morandi M Mora Department
102 Department of Neuromuscular Diseases, Istituto Nazionale Neurologico C Besta, Via Celoria 11, 20133 Milano, Italy R Barresi C Di Blasi T Negri R Brugnoni S Daniel F Cornelio L Morandi M Mora Department
Distribution of Dystroglycan in Normal Adult Mouse Tissues
 Volume 46(4): 449 457, 1998 The Journal of Histochemistry & Cytochemistry http://www.jhc.org ARTICLE Distribution of Dystroglycan in Normal Adult Mouse Tissues Madeleine Durbeej, Michael D. Henry, Maria
Volume 46(4): 449 457, 1998 The Journal of Histochemistry & Cytochemistry http://www.jhc.org ARTICLE Distribution of Dystroglycan in Normal Adult Mouse Tissues Madeleine Durbeej, Michael D. Henry, Maria
Major Histocompatibility Complex (MHC) and T Cell Receptors
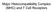 Major Histocompatibility Complex (MHC) and T Cell Receptors Historical Background Genes in the MHC were first identified as being important genes in rejection of transplanted tissues Genes within the MHC
Major Histocompatibility Complex (MHC) and T Cell Receptors Historical Background Genes in the MHC were first identified as being important genes in rejection of transplanted tissues Genes within the MHC
Skeletal Muscle and the Molecular Basis of Contraction. Lanny Shulman, O.D., Ph.D. University of Houston College of Optometry
 Skeletal Muscle and the Molecular Basis of Contraction Lanny Shulman, O.D., Ph.D. University of Houston College of Optometry Like neurons, all muscle cells can be excited chemically, electrically, and
Skeletal Muscle and the Molecular Basis of Contraction Lanny Shulman, O.D., Ph.D. University of Houston College of Optometry Like neurons, all muscle cells can be excited chemically, electrically, and
Ethnically Diverse Causes of Walker-Warburg Syndrome (WWS): FCMD Mutations Are a More Common Cause of WWS Outside of the Middle East
 HUMAN MUTATION Mutation in Brief #1025, 29:E231-E241, (2008) Online MUTATION IN BRIEF Ethnically Diverse Causes of Walker-Warburg Syndrome (WWS): FCMD Mutations Are a More Common Cause of WWS Outside of
HUMAN MUTATION Mutation in Brief #1025, 29:E231-E241, (2008) Online MUTATION IN BRIEF Ethnically Diverse Causes of Walker-Warburg Syndrome (WWS): FCMD Mutations Are a More Common Cause of WWS Outside of
REVIEW Glycobiology of neuromuscular disorders
 Glycobiology vol. 13 no. 8 pp. 67R±75R, 2003 DOI: 10.1093/glycob/cwg077 REVIEW Glycobiology of neuromuscular disorders Paul T. Martin 1,2 and Hudson H. Freeze 3 2 Department of Neuroscience, Glycobiology
Glycobiology vol. 13 no. 8 pp. 67R±75R, 2003 DOI: 10.1093/glycob/cwg077 REVIEW Glycobiology of neuromuscular disorders Paul T. Martin 1,2 and Hudson H. Freeze 3 2 Department of Neuroscience, Glycobiology
The functional role of dystrophin in the heart: implications for inherited and non-inherited heart disease. Matthew Scott Barnabei
 The functional role of dystrophin in the heart: implications for inherited and non-inherited heart disease by Matthew Scott Barnabei A dissertation submitted in partial fulfillment of the requirements
The functional role of dystrophin in the heart: implications for inherited and non-inherited heart disease by Matthew Scott Barnabei A dissertation submitted in partial fulfillment of the requirements
1. TLR. TLR Toll-like receptors. Toll Toll-like receptor, TLR TLR TLR TLR. type I TLR TLR. Toll
 54pp.145 152 2004 1. TLR T B TLR Toll-like receptors TLR TLR I IFN TLR T B B T Toll NF- B 1996 565-0871 3-1 TEL 06-6879-8303 FAX 06-6879-8305 E-mail uemattsu@biken.osaka-u.ac.jp Toll Toll-like receptor,
54pp.145 152 2004 1. TLR T B TLR Toll-like receptors TLR TLR I IFN TLR T B B T Toll NF- B 1996 565-0871 3-1 TEL 06-6879-8303 FAX 06-6879-8305 E-mail uemattsu@biken.osaka-u.ac.jp Toll Toll-like receptor,
Glycoproteomic characterization of recombinant mouse α-dystroglycan
 Glycobiology vol. 22 no. 5 pp. 662 675, 2012 doi:10.1093/glycob/cws002 Advance Access publication on January 11, 2012 Glycoproteomic characterization of recombinant mouse α-dystroglycan Rebecca Harrison
Glycobiology vol. 22 no. 5 pp. 662 675, 2012 doi:10.1093/glycob/cws002 Advance Access publication on January 11, 2012 Glycoproteomic characterization of recombinant mouse α-dystroglycan Rebecca Harrison
Problem Set 8 Key 1 of 8
 7.06 2003 Problem Set 8 Key 1 of 8 7.06 2003 Problem Set 8 Key 1. As a bright MD/PhD, you are interested in questions about the control of cell number in the body. Recently, you've seen three patients
7.06 2003 Problem Set 8 Key 1 of 8 7.06 2003 Problem Set 8 Key 1. As a bright MD/PhD, you are interested in questions about the control of cell number in the body. Recently, you've seen three patients
J Jpn Coll Angiol, 2009, 49: collagen disease, genetic polymorphism, MRL mice, recombinant inbred strains, Cd72. MRL/Mp-lpr/lpr MRL/ lpr
 Online publication June 24, 2009 1, 2 1 J Jpn Coll Angiol, 2009, 49: 11 16 collagen disease, genetic polymorphism, MRL mice, recombinant inbred strains, Cd72 SNPs case-control study MHC Fcγ NO MRL/Mp-lpr/lpr
Online publication June 24, 2009 1, 2 1 J Jpn Coll Angiol, 2009, 49: 11 16 collagen disease, genetic polymorphism, MRL mice, recombinant inbred strains, Cd72 SNPs case-control study MHC Fcγ NO MRL/Mp-lpr/lpr
Female Carrier of Duchenne Muscular Dystrophy Presenting With Secondary Dilated Cardiomyopathy : A Case Report
 J Cardiol 2001 ; 38: 35 40 Duchenne 1 Female Carrier of Duchenne Muscular Dystrophy Presenting With Secondary Dilated Cardiomyopathy : A Case Report Shigeki Kiyoaki Toru Kenji Kojiro HIRAMATSU, MD MAEKAWA,
J Cardiol 2001 ; 38: 35 40 Duchenne 1 Female Carrier of Duchenne Muscular Dystrophy Presenting With Secondary Dilated Cardiomyopathy : A Case Report Shigeki Kiyoaki Toru Kenji Kojiro HIRAMATSU, MD MAEKAWA,
18 (2), DOI: /bjmg
 18 (2), 2015 71-76 DOI: 10.1515/bjmg-2015-0088 CASE REPORT SARCOLEMMAL DEFICIENCY OF SARCOGLYCAN COMPLEX IN AN 18-MONTH-OLD TURKISH BOY WITH A LARGE DELETION IN THE BETA SARCOGLYCAN GENE Diniz G 1,*, Tekgul
18 (2), 2015 71-76 DOI: 10.1515/bjmg-2015-0088 CASE REPORT SARCOLEMMAL DEFICIENCY OF SARCOGLYCAN COMPLEX IN AN 18-MONTH-OLD TURKISH BOY WITH A LARGE DELETION IN THE BETA SARCOGLYCAN GENE Diniz G 1,*, Tekgul
Introduction. Overview
 Congenital muscular dystrophies Emma Clement MD ( Dr. Clement of Great Ormond Street Children's Hospital has no relevant financial relationships to disclose. ) Heinz Jungbluth MD PhD ( Dr. Jungbluth of
Congenital muscular dystrophies Emma Clement MD ( Dr. Clement of Great Ormond Street Children's Hospital has no relevant financial relationships to disclose. ) Heinz Jungbluth MD PhD ( Dr. Jungbluth of
Molecular mechanisms of muscular dystrophies: old and new players
 MECHANISMS OF DISEASE Molecular mechanisms of muscular dystrophies: old and new players Kay E Davies* and Kristen J Nowak Abstract The study of the muscle cell in the muscular dystrophies (MDs) has shown
MECHANISMS OF DISEASE Molecular mechanisms of muscular dystrophies: old and new players Kay E Davies* and Kristen J Nowak Abstract The study of the muscle cell in the muscular dystrophies (MDs) has shown
Charlie Taylor, PhD CpTaylor Consulting Chelsea, MI, USA
 Contribution of Calcium Channel α 2 δ Binding Sites to the Pharmacology of Gabapentin and Pregabalin Charlie Taylor, PhD CpTaylor Consulting Chelsea, MI, USA Disclosure Information Charlie Taylor, PhD
Contribution of Calcium Channel α 2 δ Binding Sites to the Pharmacology of Gabapentin and Pregabalin Charlie Taylor, PhD CpTaylor Consulting Chelsea, MI, USA Disclosure Information Charlie Taylor, PhD
CARDIAC muscle is commonly affected in muscular
 362 THE NEW ENGLAND JOURNAL OF MEDICINE Feb. 8, 1996 BRIEF REPORT: DEFICIENCY OF A DYSTROPHIN-ASSOCIATED GLYCOPROTEIN (ADHALIN) IN A PATIENT WITH MUSCULAR DYSTROPHY AND CARDIOMYOPATHY RICARDO FADIC, M.D.,
362 THE NEW ENGLAND JOURNAL OF MEDICINE Feb. 8, 1996 BRIEF REPORT: DEFICIENCY OF A DYSTROPHIN-ASSOCIATED GLYCOPROTEIN (ADHALIN) IN A PATIENT WITH MUSCULAR DYSTROPHY AND CARDIOMYOPATHY RICARDO FADIC, M.D.,
Mutations in several components of the dystrophin glycoprotein. Animal Models for Muscular Dystrophy Show Different Patterns of Sarcolemmal Disruption
 Animal Models for Muscular Dystrophy Show Different Patterns of Sarcolemmal Disruption Volker Straub,* Jill A. Rafael, Jeffrey S. Chamberlain, and Kevin P. Campbell* Department of *Physiology and Biophysics
Animal Models for Muscular Dystrophy Show Different Patterns of Sarcolemmal Disruption Volker Straub,* Jill A. Rafael, Jeffrey S. Chamberlain, and Kevin P. Campbell* Department of *Physiology and Biophysics
A rare case of muscular dystrophy with POMT2 and FKRP gene mutation. Present by : Ghasem Khazaei Supervisor :Dr Mina Mohammadi Sarband
 A rare case of muscular dystrophy with POMT2 and FKRP gene mutation Present by : Ghasem Khazaei Supervisor :Dr Mina Mohammadi Sarband Index : Congenital muscular dystrophy (CMD) Dystroglycanopathies Walker-Warburg
A rare case of muscular dystrophy with POMT2 and FKRP gene mutation Present by : Ghasem Khazaei Supervisor :Dr Mina Mohammadi Sarband Index : Congenital muscular dystrophy (CMD) Dystroglycanopathies Walker-Warburg
T-cell activation T cells migrate to secondary lymphoid tissues where they interact with antigen, antigen-presenting cells, and other lymphocytes:
 Interactions between innate immunity & adaptive immunity What happens to T cells after they leave the thymus? Naïve T cells exit the thymus and enter the bloodstream. If they remain in the bloodstream,
Interactions between innate immunity & adaptive immunity What happens to T cells after they leave the thymus? Naïve T cells exit the thymus and enter the bloodstream. If they remain in the bloodstream,
T-cell activation T cells migrate to secondary lymphoid tissues where they interact with antigen, antigen-presenting cells, and other lymphocytes:
 Interactions between innate immunity & adaptive immunity What happens to T cells after they leave the thymus? Naïve T cells exit the thymus and enter the bloodstream. If they remain in the bloodstream,
Interactions between innate immunity & adaptive immunity What happens to T cells after they leave the thymus? Naïve T cells exit the thymus and enter the bloodstream. If they remain in the bloodstream,
Animal Models for Genetic Neuromuscular Diseases
 J Mol Neurosci (2008) 34:241 248 DOI 10.1007/s12031-007-9023-9 Animal Models for Genetic Neuromuscular Diseases Mariz Vainzof & Danielle Ayub-Guerrieri & Paula C. G. Onofre & Poliana C. M. Martins & Vanessa
J Mol Neurosci (2008) 34:241 248 DOI 10.1007/s12031-007-9023-9 Animal Models for Genetic Neuromuscular Diseases Mariz Vainzof & Danielle Ayub-Guerrieri & Paula C. G. Onofre & Poliana C. M. Martins & Vanessa
MUSCLE DISEASE ANTIBODIES NOVOCASTRA ADVANCING MUSCLE DISEASE DIAGNOSIS, MANAGEMENT AND RESEARCH RESULTS YOU CAN RELY ON
 MUSCLE DISEASE ANTIBODIES ADVANCING MUSCLE DISEASE DIAGNOSIS, MANAGEMENT AND RESEARCH NOVOCASTRA RESULTS YOU CAN RELY ON Novocastra Muscle Disease Antibodies The Novocastra muscle disease portfolio comprises
MUSCLE DISEASE ANTIBODIES ADVANCING MUSCLE DISEASE DIAGNOSIS, MANAGEMENT AND RESEARCH NOVOCASTRA RESULTS YOU CAN RELY ON Novocastra Muscle Disease Antibodies The Novocastra muscle disease portfolio comprises
Replication Defective Enterovirus Infections: Implications for Type I Diabetes
 Replication Defective Enterovirus Infections: Implications for Type I Diabetes N. M. Chapman Department of Pathology & Microbiology University of Nebraska Medical Center Enterovirus Genome and 2 Capsid
Replication Defective Enterovirus Infections: Implications for Type I Diabetes N. M. Chapman Department of Pathology & Microbiology University of Nebraska Medical Center Enterovirus Genome and 2 Capsid
Chapter Skeletal Muscle Structure and Function
 Chapter 10.2 Skeletal Muscle Structure and Function Introduction to Muscle Physiology Movement is a fundamental characteristic of all living things All muscle cells (skeletal, cardiac, and smooth) are
Chapter 10.2 Skeletal Muscle Structure and Function Introduction to Muscle Physiology Movement is a fundamental characteristic of all living things All muscle cells (skeletal, cardiac, and smooth) are
Basic Immunology. Lecture 5 th and 6 th Recognition by MHC. Antigen presentation and MHC restriction
 Basic Immunology Lecture 5 th and 6 th Recognition by MHC. Antigen presentation and MHC restriction Molecular structure of MHC, subclasses, genetics, functions. Antigen presentation and MHC restriction.
Basic Immunology Lecture 5 th and 6 th Recognition by MHC. Antigen presentation and MHC restriction Molecular structure of MHC, subclasses, genetics, functions. Antigen presentation and MHC restriction.
Congenital Muscular Dystrophy: Hospital Based Study in Egyptian Pediatric Patients
 World Journal of Medical Sciences 10 (4): 503-507, 2014 ISSN 1817-3055 IDOSI Publications, 2014 DOI: 10.5829/idosi.wjms.2014.10.4.953 Congenital Muscular Dystrophy: Hospital Based Study in Egyptian Pediatric
World Journal of Medical Sciences 10 (4): 503-507, 2014 ISSN 1817-3055 IDOSI Publications, 2014 DOI: 10.5829/idosi.wjms.2014.10.4.953 Congenital Muscular Dystrophy: Hospital Based Study in Egyptian Pediatric
The Angiopoietin Axis in Cancer
 Ang2 Ang1 The Angiopoietin Axis in Cancer Tie2 An Overview: The Angiopoietin Axis Plays an Essential Role in the Regulation of Tumor Angiogenesis Growth of a tumor beyond a limiting size is dependent upon
Ang2 Ang1 The Angiopoietin Axis in Cancer Tie2 An Overview: The Angiopoietin Axis Plays an Essential Role in the Regulation of Tumor Angiogenesis Growth of a tumor beyond a limiting size is dependent upon
Materials and Methods , The two-hybrid principle.
 The enzymatic activity of an unknown protein which cleaves the phosphodiester bond between the tyrosine residue of a viral protein and the 5 terminus of the picornavirus RNA Introduction Every day there
The enzymatic activity of an unknown protein which cleaves the phosphodiester bond between the tyrosine residue of a viral protein and the 5 terminus of the picornavirus RNA Introduction Every day there
Role of non-raft cholesterol in lymphocytic choriomeningitis virus infection via a-dystroglycan
 Journal of General Virology (06), 87, 673 678 DOI 10.1099/vir.0.81444-0 Short Communication Correspondence Salvatore Carbonetto sal.carbonetto@mcgill.ca Role of non-raft cholesterol in lymphocytic choriomeningitis
Journal of General Virology (06), 87, 673 678 DOI 10.1099/vir.0.81444-0 Short Communication Correspondence Salvatore Carbonetto sal.carbonetto@mcgill.ca Role of non-raft cholesterol in lymphocytic choriomeningitis
Protein Trafficking in the Secretory and Endocytic Pathways
 Protein Trafficking in the Secretory and Endocytic Pathways The compartmentalization of eukaryotic cells has considerable functional advantages for the cell, but requires elaborate mechanisms to ensure
Protein Trafficking in the Secretory and Endocytic Pathways The compartmentalization of eukaryotic cells has considerable functional advantages for the cell, but requires elaborate mechanisms to ensure
Intercellular indirect communication
 Intercellular indirect communication transmission of chemical signals: sending cell signal transmitting tissue hormone medium receiving cell hormone intercellular fluid blood neurocrine neurotransmitter
Intercellular indirect communication transmission of chemical signals: sending cell signal transmitting tissue hormone medium receiving cell hormone intercellular fluid blood neurocrine neurotransmitter
News Release. Identification of the cause of chronic active Epstein Barr virus (EBV) infection and the mechanism by which EBV causes cancer
 News Release Title Identification of the cause of chronic active Epstein Barr virus (EBV) infection and the mechanism by which EBV causes cancer Key Points A research group in Japan, led by Dr. Hiroshi
News Release Title Identification of the cause of chronic active Epstein Barr virus (EBV) infection and the mechanism by which EBV causes cancer Key Points A research group in Japan, led by Dr. Hiroshi
Significance of the MHC
 CHAPTER 8 Major Histocompatibility Complex (MHC) What is is MHC? HLA H-2 Minor histocompatibility antigens Peter Gorer & George Sneell (1940) Significance of the MHC role in immune response role in organ
CHAPTER 8 Major Histocompatibility Complex (MHC) What is is MHC? HLA H-2 Minor histocompatibility antigens Peter Gorer & George Sneell (1940) Significance of the MHC role in immune response role in organ
Dr Mahmood S Choudhery, PhD, Postdoc (USA) Assistant Professor Tissue Engineering and Regenerative Medicine King Edward Medical University/Mayo
 Integration of Cells into Tissues Dr Mahmood S Choudhery, PhD, Postdoc (USA) Assistant Professor Tissue Engineering and Regenerative Medicine King Edward Medical University/Mayo Hospital Lahore 1. How
Integration of Cells into Tissues Dr Mahmood S Choudhery, PhD, Postdoc (USA) Assistant Professor Tissue Engineering and Regenerative Medicine King Edward Medical University/Mayo Hospital Lahore 1. How
Matrix metalloproteinases
 Matrix metalloproteinases,, 1. 2. 2-1. Domain 2-2. TIMPs 2-3. MMPs, 3. 4. 1. Collagens, proteoglycans (Extracellular matrix, ECM),,. matrixins matrix metalloproteinases (MMPs) [1, 2]. MMPs,, cytokines
Matrix metalloproteinases,, 1. 2. 2-1. Domain 2-2. TIMPs 2-3. MMPs, 3. 4. 1. Collagens, proteoglycans (Extracellular matrix, ECM),,. matrixins matrix metalloproteinases (MMPs) [1, 2]. MMPs,, cytokines
Cells & Tissues. Chapter 3
 Cells & Tissues Chapter 3 Cell Theory Cell is structural and functional unit of life Activity of an organism is dependent upon its cells Principle of Complementarity functions of cells are dependent upon
Cells & Tissues Chapter 3 Cell Theory Cell is structural and functional unit of life Activity of an organism is dependent upon its cells Principle of Complementarity functions of cells are dependent upon
Key words: tumor imaging, radioiodinated monoclonal antibody, colorectal cancer
 Key words: tumor imaging, radioiodinated monoclonal antibody, colorectal cancer Table 1 Serological analysis of the expression of H-15 antigen in established cancer cell lines and in normal cells. MoAb
Key words: tumor imaging, radioiodinated monoclonal antibody, colorectal cancer Table 1 Serological analysis of the expression of H-15 antigen in established cancer cell lines and in normal cells. MoAb
Iowa Wellstone Center Muscle Tissue and Cell Culture Repository
 Iowa Wellstone Center Muscle Tissue and Cell Culture Repository Steven A. Moore, M.D., Ph.D. The University of Iowa Department of Pathology and Iowa Wellstone Muscular Dystrophy Cooperative Research Center
Iowa Wellstone Center Muscle Tissue and Cell Culture Repository Steven A. Moore, M.D., Ph.D. The University of Iowa Department of Pathology and Iowa Wellstone Muscular Dystrophy Cooperative Research Center
Overview of methodology, tools and reagents for evaluating cell proliferation and invasion using multicellular tumor spheroids.
 The Next Step in the Evolution of 3D Culture: Utilizing Extracellular Matrix to Enhance Multicellular Tumor Spheroid Models for Proliferation and Invasion Overview of methodology, tools and reagents for
The Next Step in the Evolution of 3D Culture: Utilizing Extracellular Matrix to Enhance Multicellular Tumor Spheroid Models for Proliferation and Invasion Overview of methodology, tools and reagents for
Immunohistochemical Study of Dystrophin Associated Glycoproteins in Limb-girdle Muscular Dystrophies
 Dystrophin Immunohistochemical Study of Dystrophin Associated Glycoproteins in Limb-girdle Muscular Dystrophies NSC 89-2314-B-002-111 88 8 1 89 7 31 ( Peroxidase -AntiPeroxidase Immnofluorescence) Abstract
Dystrophin Immunohistochemical Study of Dystrophin Associated Glycoproteins in Limb-girdle Muscular Dystrophies NSC 89-2314-B-002-111 88 8 1 89 7 31 ( Peroxidase -AntiPeroxidase Immnofluorescence) Abstract
Sarcoglycan Subcomplex Expression in Normal Human Smooth Muscle
 Volume 55(8): 831 843, 2007 Journal of Histochemistry & Cytochemistry http://www.jhc.org ARTICLE Sarcoglycan Subcomplex Expression in Normal Human Smooth Muscle Giuseppe Anastasi, Giuseppina Cutroneo,
Volume 55(8): 831 843, 2007 Journal of Histochemistry & Cytochemistry http://www.jhc.org ARTICLE Sarcoglycan Subcomplex Expression in Normal Human Smooth Muscle Giuseppe Anastasi, Giuseppina Cutroneo,
L imb-girdle muscular dystrophies (LGMD) and congenital
 1of7 ONLINE MUTATION REPORT FKRP (826C.A) frequently causes limb-girdle muscular dystrophy in German patients M C Walter, J A Petersen, R Stucka, D Fischer, R Schröder, M Vorgerd, A Schroers, H Schreiber,
1of7 ONLINE MUTATION REPORT FKRP (826C.A) frequently causes limb-girdle muscular dystrophy in German patients M C Walter, J A Petersen, R Stucka, D Fischer, R Schröder, M Vorgerd, A Schroers, H Schreiber,
Using a proteomic approach to identify proteasome interacting proteins in mammalian
 Supplementary Discussion Using a proteomic approach to identify proteasome interacting proteins in mammalian cells, we describe in the present study a novel chaperone complex that plays a key role in the
Supplementary Discussion Using a proteomic approach to identify proteasome interacting proteins in mammalian cells, we describe in the present study a novel chaperone complex that plays a key role in the
silent epidemic,. (WHO),
 Tel: 02-740-8686; E-mail: hhbkim@snu.ac.kr silent epidemic,. (WHO),. 5 3, 1. 50 70. 50%, 25%, 20% (12~35%). 2.8% 0.7% 4. ( ). bone remodeling (osteoblast), (osteoclast),.. 3~4.. 70% (osteocyte) (bone lining
Tel: 02-740-8686; E-mail: hhbkim@snu.ac.kr silent epidemic,. (WHO),. 5 3, 1. 50 70. 50%, 25%, 20% (12~35%). 2.8% 0.7% 4. ( ). bone remodeling (osteoblast), (osteoclast),.. 3~4.. 70% (osteocyte) (bone lining
The Childhood Muscular Dystrophies: Diseases Sharing a Common Pathogenesis of Membrane Instability
 Carrell-Krusen Symposium Invited Lecture The Childhood Muscular Dystrophies: Diseases Sharing a Common Pathogenesis of Membrane Instability Jerry R. Mendell, MD; Zarife Sahenk, MD; Thomas W. Prior, PhD
Carrell-Krusen Symposium Invited Lecture The Childhood Muscular Dystrophies: Diseases Sharing a Common Pathogenesis of Membrane Instability Jerry R. Mendell, MD; Zarife Sahenk, MD; Thomas W. Prior, PhD
Cell Membranes. Dr. Diala Abu-Hassan School of Medicine Cell and Molecular Biology
 Cell Membranes Dr. Diala Abu-Hassan School of Medicine Dr.abuhassand@gmail.com Cell and Molecular Biology Organelles 2Dr. Diala Abu-Hassan Membrane proteins Major components of cells Nucleic acids DNA
Cell Membranes Dr. Diala Abu-Hassan School of Medicine Dr.abuhassand@gmail.com Cell and Molecular Biology Organelles 2Dr. Diala Abu-Hassan Membrane proteins Major components of cells Nucleic acids DNA
Macrophage Activation & Cytokine Release. Dendritic Cells & Antigen Presentation. Neutrophils & Innate Defense
 Macrophage Activation & Cytokine Release Dendritic Cells & Antigen Presentation Neutrophils & Innate Defense Neutrophils Polymorphonuclear cells (PMNs) are recruited to the site of infection where they
Macrophage Activation & Cytokine Release Dendritic Cells & Antigen Presentation Neutrophils & Innate Defense Neutrophils Polymorphonuclear cells (PMNs) are recruited to the site of infection where they
Vascular Basement Membrane Components in Angiogenesis An Act of Balance
 Directions in Science TheScientificWorldJOURNAL (2008) 8, 1246 1249 ISSN 1537-744X; DOI 10.1100/tsw.2008.155 Vascular Basement Membrane Components in Angiogenesis An Act of Balance Lars Jakobsson 1, *
Directions in Science TheScientificWorldJOURNAL (2008) 8, 1246 1249 ISSN 1537-744X; DOI 10.1100/tsw.2008.155 Vascular Basement Membrane Components in Angiogenesis An Act of Balance Lars Jakobsson 1, *
Influenza A virus hemagglutinin and neuraminidase act as novel motile machinery. Tatsuya Sakai, Shin I. Nishimura, Tadasuke Naito, and Mineki Saito
 Supplementary Information for: Influenza A virus hemagglutinin and neuraminidase act as novel motile machinery Tatsuya Sakai, Shin I. Nishimura, Tadasuke Naito, and Mineki Saito Supplementary Methods Supplementary
Supplementary Information for: Influenza A virus hemagglutinin and neuraminidase act as novel motile machinery Tatsuya Sakai, Shin I. Nishimura, Tadasuke Naito, and Mineki Saito Supplementary Methods Supplementary
Skeletal, cardiac, and respiratory muscle function and histopathology in the P448Lneo mouse model of FKRP-deficient muscular dystrophy
 Yu et al. Skeletal Muscle (2018) 8:13 https://doi.org/10.1186/s13395-018-0158-x RESEARCH Open Access Skeletal, cardiac, and respiratory muscle function and histopathology in the P448Lneo mouse model of
Yu et al. Skeletal Muscle (2018) 8:13 https://doi.org/10.1186/s13395-018-0158-x RESEARCH Open Access Skeletal, cardiac, and respiratory muscle function and histopathology in the P448Lneo mouse model of
Cell matrix interactions in muscle disease
 Journal of Pathology J Pathol 2012; 226: 200 218 Published online in Wiley Online Library (wileyonlinelibrary.com) DOI: 10.1002/path.3020 INVITED REVIEW Cell matrix interactions in muscle disease Virginie
Journal of Pathology J Pathol 2012; 226: 200 218 Published online in Wiley Online Library (wileyonlinelibrary.com) DOI: 10.1002/path.3020 INVITED REVIEW Cell matrix interactions in muscle disease Virginie
General information. Cell mediated immunity. 455 LSA, Tuesday 11 to noon. Anytime after class.
 General information Cell mediated immunity 455 LSA, Tuesday 11 to noon Anytime after class T-cell precursors Thymus Naive T-cells (CD8 or CD4) email: lcoscoy@berkeley.edu edu Use MCB150 as subject line
General information Cell mediated immunity 455 LSA, Tuesday 11 to noon Anytime after class T-cell precursors Thymus Naive T-cells (CD8 or CD4) email: lcoscoy@berkeley.edu edu Use MCB150 as subject line
Structure and Function of Antigen Recognition Molecules
 MICR2209 Structure and Function of Antigen Recognition Molecules Dr Allison Imrie allison.imrie@uwa.edu.au 1 Synopsis: In this lecture we will examine the major receptors used by cells of the innate and
MICR2209 Structure and Function of Antigen Recognition Molecules Dr Allison Imrie allison.imrie@uwa.edu.au 1 Synopsis: In this lecture we will examine the major receptors used by cells of the innate and
Matriglycan: a novel polysaccharide that links dystroglycan to the basement membrane
 Glycobiology, 2015, vol. 25, no. 7, 702 713 doi: 10.1093/glycob/cwv021 Advance Access Publication Date: 16 April 2015 Review Review Matriglycan: a novel polysaccharide that links dystroglycan to the basement
Glycobiology, 2015, vol. 25, no. 7, 702 713 doi: 10.1093/glycob/cwv021 Advance Access Publication Date: 16 April 2015 Review Review Matriglycan: a novel polysaccharide that links dystroglycan to the basement
Anonymous MIT students MK and NM Natalie Kuldell May 2009
 Anonymous MIT students MK and NM Natalie Kuldell 20.020 13 May 2009 Technical Document: oncocures Problem: Dynamic Metastasis Imaging Our project has been designed with the hope of creating a way to detect
Anonymous MIT students MK and NM Natalie Kuldell 20.020 13 May 2009 Technical Document: oncocures Problem: Dynamic Metastasis Imaging Our project has been designed with the hope of creating a way to detect
Lecture 3: Cells and Tissues. Bio 219 Dr. Adam Ross
 Lecture 3: Cells and Tissues Bio 219 Dr. Adam Ross Cell Physiology Cell Physiology Brief review of organelles Should be mostly review Cell surrounded by plasma membrane Lipid bilayer Also surrounds organelles
Lecture 3: Cells and Tissues Bio 219 Dr. Adam Ross Cell Physiology Cell Physiology Brief review of organelles Should be mostly review Cell surrounded by plasma membrane Lipid bilayer Also surrounds organelles
Seminar. Muscular dystrophies
 Muscular dystrophies Eugenio Mercuri, Francesco Muntoni Muscular dystrophies are a heterogeneous group of inherited disorders that share similar clinical features and dystrophic changes on muscle biopsy.
Muscular dystrophies Eugenio Mercuri, Francesco Muntoni Muscular dystrophies are a heterogeneous group of inherited disorders that share similar clinical features and dystrophic changes on muscle biopsy.
There are approximately 30,000 proteasomes in a typical human cell Each proteasome is approximately 700 kda in size The proteasome is made up of 3
 Proteasomes Proteasomes Proteasomes are responsible for degrading proteins that have been damaged, assembled improperly, or that are of no profitable use to the cell. The unwanted protein is literally
Proteasomes Proteasomes Proteasomes are responsible for degrading proteins that have been damaged, assembled improperly, or that are of no profitable use to the cell. The unwanted protein is literally
Chapter 6. Antigen Presentation to T lymphocytes
 Chapter 6 Antigen Presentation to T lymphocytes Generation of T-cell Receptor Ligands T cells only recognize Ags displayed on cell surfaces These Ags may be derived from pathogens that replicate within
Chapter 6 Antigen Presentation to T lymphocytes Generation of T-cell Receptor Ligands T cells only recognize Ags displayed on cell surfaces These Ags may be derived from pathogens that replicate within
Asymptomatic carriers for homozygous novel mutations in the FKRP gene: the other end of the spectrum
 (2003) 11, 923 930 & 2003 Nature Publishing Group All rights reserved 1018-4813/03 $25.00 www.nature.com/ejhg ARTICLE Asymptomatic carriers for homozygous novel mutations in the FKRP gene: the other end
(2003) 11, 923 930 & 2003 Nature Publishing Group All rights reserved 1018-4813/03 $25.00 www.nature.com/ejhg ARTICLE Asymptomatic carriers for homozygous novel mutations in the FKRP gene: the other end
Research Article: Histology and Cell Biology Preliminary study on sarcoglycan sub-complex in rat cerebral and cerebellar cortex
 IJAE Vol. 117, n. 1: 54-64, 2012 Italian Journal of Anatomy and Embryology Research Article: Histology and Cell Biology Preliminary study on sarcoglycan sub-complex in rat cerebral and cerebellar cortex
IJAE Vol. 117, n. 1: 54-64, 2012 Italian Journal of Anatomy and Embryology Research Article: Histology and Cell Biology Preliminary study on sarcoglycan sub-complex in rat cerebral and cerebellar cortex
HIV INFECTION: An Overview
 HIV INFECTION: An Overview UNIVERSITY OF PAPUA NEW GUINEA SCHOOL OF MEDICINE AND HEALTH SCIENCES DIVISION OF BASIC MEDICAL SCIENCES DISCIPLINE OF BIOCHEMISTRY & MOLECULAR BIOLOGY PBL MBBS II SEMINAR VJ
HIV INFECTION: An Overview UNIVERSITY OF PAPUA NEW GUINEA SCHOOL OF MEDICINE AND HEALTH SCIENCES DIVISION OF BASIC MEDICAL SCIENCES DISCIPLINE OF BIOCHEMISTRY & MOLECULAR BIOLOGY PBL MBBS II SEMINAR VJ
The Adaptive Immune Response: T lymphocytes and Their Functional Types *
 OpenStax-CNX module: m46560 1 The Adaptive Immune Response: T lymphocytes and Their Functional Types * OpenStax This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution
OpenStax-CNX module: m46560 1 The Adaptive Immune Response: T lymphocytes and Their Functional Types * OpenStax This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution
Medical Virology Immunology. Dr. Sameer Naji, MB, BCh, PhD (UK) Head of Basic Medical Sciences Dept. Faculty of Medicine The Hashemite University
 Medical Virology Immunology Dr. Sameer Naji, MB, BCh, PhD (UK) Head of Basic Medical Sciences Dept. Faculty of Medicine The Hashemite University Human blood cells Phases of immune responses Microbe Naïve
Medical Virology Immunology Dr. Sameer Naji, MB, BCh, PhD (UK) Head of Basic Medical Sciences Dept. Faculty of Medicine The Hashemite University Human blood cells Phases of immune responses Microbe Naïve
This is a repository copy of A role for dystroglycan in the pathophysiology of acute leukemic cells..
 This is a repository copy of A role for dystroglycan in the pathophysiology of acute leukemic cells.. White Rose Research Online URL for this paper: http://eprints.whiterose.ac.uk/117598/ Version: Accepted
This is a repository copy of A role for dystroglycan in the pathophysiology of acute leukemic cells.. White Rose Research Online URL for this paper: http://eprints.whiterose.ac.uk/117598/ Version: Accepted
