Chapter 12, Part 2. Fundamentals of Atmospheric Modeling
|
|
|
- Baldwin Mathews
- 5 years ago
- Views:
Transcription
1 verhead Slides for hapter 12, Part 2 of Fundamentals of Atmospheric Modeling by Mark Z. Jacobson Department of ivil & Environmental Engineering Stanford University Stanford, A January 30, 2002
2 Alkene Reaction With zone Ethene Ethene Ethene molozonide riegee biradical reaction 37% 63% + Formaldehyde riegee biradical + * Formaldehyde Excited riegee biradical (12.89) riegee biradical + N N 2 Formaldehyde (12.90) Excited criegee biradical decomposition 60% + 2 * 21% Excited riegee biradical * Excited formic acid 19% (12.91)
3 Alkene Reaction With zone Propene 7.5% + 3 Formaldehyde Methyl criegee biradical 2 3 Propene Propene molozonide % Formaldehyde % Acetaldehyde 3 * Excited methyl criegee biradical riegee biradical % + * Acetaldehyde Excited criegee biradical (12.92)
4 Alkene Reaction With zone Methylcriegee biradical reaction 3 Methyl criegee biradical + N N 2 3 Acetaldehyde (12.93) Excited methylcriegee biradical decomposition 3 * Excited methyl criegee biradical 3 * Excited acetic acid 16% % % (12.94)
5 Alkene Reaction With Nitrate Ethene --> nitrated organic radicals Ethene + N 3 N 2 Ethyl nitrate radical N Ethylperoxy nitrate radical + N N 2 N 2 Ethoxy nitrate radica (12.95) Propene --> nitrated organic radicals 3 Propene + N 3 N Propyl nitrate radical N Propylperoxy nitrate radical + N N 2 N 3 2 Propoxy nitrate radic (12.96)
6 Aromatic Reaction With ydroxyl Radical Toluene oxidation 8% Benzylperoxy radical 3 2 Benzyl radical 3 Toluene 92% Toluene-hydroxylradical adduct 3 o-ydroxytoluene + 2 o-resol (12.97)
7 Aromatic Reaction With ydroxyl Radical Benzylperoxy radical reaction with N 2 + N N 2 Benzoxy radical 2 Benzaldehyde Benzylperoxy radical + N 2 N Benzyl nitrate (12.98) Toluene-hydroxyl radical adduct reaction 3 + N 3 N 2 Toluene-hydroxyl radical adduct (12.99)
8 Fate of resol resol --> methylphenylperoxy radical and nitrocresol 3 +, Methylphenylperoxy radical o-resol N Methylphenoxy radical N m-nitrocresol (12.100)
9 Terpene Reaction With (1) 16.4% (4) 23.6% Isoprene 3 +, 2 (2) 12.3% (5) 21.2% (3) 12.3% (6) 14.1% Isoprene peroxy radicals (12.101) All six products convert N to N 2
10 Terpene Reaction With Methacrolein production via second product (12.102) N + 2 N Isoprene peroxy radical Methacrolein Formaldehyde Methylvinylketone production via fifth product (12.103) N + 2 N Isoprene peroxy radical Methylvinylketone Formaldehyde
11 Terpene Reaction With zone 2 3 Methylvinylketone + riegee biradical Methacrolein + riegee biradical 2 2 Isoprene 2 3 zonide product + Formaldehyde 3 2 zonide product + Formaldehyde (12.104)
12 Alcohol Reactions ydroxyl radical scavenges methanol (36-hour lifetime) + 85% Formaldehyde 2 15% Methanol Methoxy radical (12.105) ydroxyl radical scavenges ethanol (10-hour lifetime) 5% Ethanol + 90% 2 5% Acetaldehyde Ethoxy radical (12.106)
13 arbon Bond Lumping rganic gases lumped into surrogate groups. PAR (paraffins) -- Single carbon atoms with a single-bond between them LE (olefins) -- Terminal carbon atom pair with a double-bond between the two atoms ALD2 -- Non-terminal carbon atom pairs with a double bond attached to one of the carbons and terminal two-carbon carbonyl groups [-(=)] KET -- Single carbon ketone groups (=) TL (toluene) -- 7-carbon aromatics XYL (m-xylene) -- 8-carbon aromatics ISP (isoprene) -- Terpenes UNR -- Unreactive
14 arbon Bond Lumping (Table 12.7) hemical Name arbon Bond Group hemical Structure Ethane 0.4 PAR +1.6 UNR Ethene 1 ET Formaldehyde 1 FRM hemical Name arbon Bond Group hemical Structure n-butane 4 PAR Trans 2-butene 2 ALD2 Acetaldehyde 1 ALD2 hemical Name arbon Bond Group hemical Structure 2,2,4-Trimethylpentane 8 PAR Propene 1 PAR + 1 LE Propionaldehyde 1 PAR + 1 ALD2 hemical Name arbon Bond Group hemical Structure yclopentane 5 PAR Ethyne 1 PAR + 1 UNR Benzaldehyde 1 ALD2 + 5 UNR 2 3 Toluene 1 TL 3 Ethylbenzene 1 PAR + 1 TL 3 2 m-xylene 1 XYL 3 1,2,3- Trimethylbenzene 1 PAR + 1 XYL
15 Stratospheric hemistry zone mixing ratios stratosphere free troposphere urban air 10 ppmv 40 ppbv ppmv zone production in the stratosphere xygen photolysis 2 + hν λ < 175 nm ( 1 D) + (12.107) 2 + hν < λ < 245 nm (12.108) zone formation M ( 1 D) (12.109) M 3 + M (12.110) zone photodissociation 3 + hν 2 + ( 1 D) λ < 310 nm (12.111) 3 + hν 2 + λ > 310 nm (12.112)
16 zone Destruction by Nx Nitrous oxide reaction: 10% of N 2 destruction N 2 + ( 1 D) 64% 2N 36% N (12.113) Nitrous oxide photolysis: 90% of N 2 destruction (12.114) N 2 + hν N 2 + ( 1 D) λ < 240 nm N catalytically destroys ozone in the upper stratosphere N + 3 N (12.115) N 2 + N + 2 (12.116) (12.117)
17 zone Destruction by x ydroxyl radical formation in stratosphere ( 1 D) (12.118) catalytically destroys ozone in the lower stratosphere (12.119) (12.120) (12.121)
18 Removal reactions Removal of x and Nx (12.122) M N 2 + N 3 (12.123) M 2 + N 2 2 N 2 (12.124) Nitric acid and peroxynitric acid photodissociation is slow Source of Water Vapor (12.125) Methane and carbon monoxide reactions in the stratosphere are similar to those in the free troposphere
19 hlorine Emissions to Stratosphere Table WM (1994) hemical Formula Trade Name hemical Name Percent ontribution to Stratospheric Emissions Anthropogenic Sources F 2 l 2 F-12 Dichlorodifluoromethane 28 Fl 3 F-11 Trichlorofluoromethane 23 l 4 arbon tetrachloride 12 3 l 3 Methyl chloroform 10 Fl 2 F 2 l F Fluorodichloro,2- difluorochloroethane 6 F 2 l F-22 hlorodifluoromethane 3 Natural Sources 3 l --- Methyl chloride 15 l --- ydrochloric acid 3 Total 100%
20 zone Destruction by hlorine Photolysis of chlorinated compounds above 20 km l l F l + hν F + l λ < 250 nm l l (12.126) l l F l + hν F + l λ < 230 nm F F (12.127) l l l l + hν l + l λ < 250 nm l l (12.129) l + hν + l λ < 220 nm (12.130) Methyl chloride scavenging by hydroxyl radical l + l 2 (12.128)
21 zone Destruction by hlorine atalytic ozone destruction by chlorine l + 3 l + 2 (12.131) l + l + 2 (12.132) (12.133) nly 1% of chlorine is typically active as l or l.
22 Removal of Active hlorine Removal of l and l 4 l + 3 l + 2 l l l + 2 (12.134) M l + N 2 l N hlorine monoxide hlorine nitrate (12.135) l hlorine monoxide + 2 l 2 ypochlorous acid (12.136)
23 Removal of Active hlorine l reservoir leaks hν + l λ < 220 nm l + l + 2 l + (12.137) ln 2 reservoir leaks l N + hν l + N λ < 400 nm hlorine nitrate l reservoir leaks Nitrate radical (12.138) l + hν l + λ < 375 nm (12.139)
24 zone Destruction by Bromine 3 Br = methyl bromide (produced biogenically in the oceans and anthropogenically as soil fumigant) Photolysis of methyl above 20 km Br + hν + Br λ < 260 nm (12.140) atalytic ozone destruction by bromine Br + 3 Br + 2 (12.141) Br + Br + 2 (12.142) (12.143)
25 Removal of Active Bromine Removal of Br and Br Br + 2 Br Br + 2 (12.144) M Br + N 2 Br N Bromine monoxide Bromine nitrate (12.145) Br and BrN 2 reservoir leaks Br + Br + 2 (12.146) Br N + hν Br + N λ < 390 nm Bromine nitrate Nitrate radical (12.147)
26 zone Regeneration Figs a, b. Time-evolution of modeled profile of ozone (a) mixing ratio and (b) number concentration at 34 o N latitude, starting with zero ozone our 1 our 6 our 24 Day 5 Day 50 Day 464 Altitude (km) Altitude (km) zone volume mixing ratio (ppmv) zone (10 11 molecules cm -3 )
27 Regeneration Rate of the Global zone Layer Fig hange in ozone column abundance, averaged over the globe, during two global model simulations in which chlorine was present and absent, respectively. In both cases, ozone was initially removed from the model atmosphere. Avgerage global ozone column (Dobson units) No chlorine 250 With chlorine /1 1/7 4/17 7/26 11/4 Day and date of simulation
28 zone ole Growth Table Minimum measured values of ozone column abundances and areal extent of the ozone hole over Antarctic region from Data from NASA Goddard Space Flight enter. The area of the Antarctic is about 13 million km 2 and the area of North America is about 24 million km 2. zone Minima (DU) Size (million km 2 )
29 Polar Stratospheric loud Reactions Type I Polar Stratospheric louds (PSs) nitric acid and water temperature of formation < 195 K diameter µm number concentration 1 partic. cm -3 Type II Polar Stratospheric louds Water ice temperature of formation < 187 K diameter µm number concentration 0.1 partic. cm -3 Reactions on Polar Stratospheric loud Surfaces ln 2 (g) + 2 (a) l(g) + N 3 (a) (12.148) ln 2 (g) + l(a) l 2 (g) + N 3 (a) (12.149) N 2 5 (g) + 2 (a) 2N 3 (a) (12.150) N 2 5 (g) + l(a) ln 2 (g) + N 3 (a) (12.151) l(g) + l(a) l 2 (g) + 2 (a) (12.152)
30 Surface Reaction Rates First-order rate coefficient (s -1 ) k s,q = 1 4 v qγ q a (12.153) Thermal velocity of impinging gas (cm s -1 ) v q = 8k B T πm q 1 2 (12.154) Table Estimated reaction probabilities for the gases in reactions (12.147) - (12.151) on Type I and II PS surfaces. Data from DeMore et al. (1997) and references therein. Reaction Type I PSs Type II PSs ln 2 (g) + 2 (a) ln 2 (g) + l(a) N 2 5 (g) + 2 (a) N 2 5 (g) + l(a) l(g) + l(a)
31 Polar zone Destruction l 2 and l photolysis in early spring l 2 + hν 2l λ < 450 nm (12.164) l + hν l + λ < 375 nm (12.165) hlorine nitrite photolysis in early spring ln 2 + hν l + N 2 λ < 370 nm (12.166) atalytic ozone destruction by dimer mechanism 2 x ( l + 3 l + 2 ) (12.167) l + l M l 2 2 (12.168) l hν l + l λ < 360 nm M l l + 2 (12.170) (12.171)
32 Polar zone Destruction A second catalytic cycle that involves bromine l + 3 l + 2 (12.172) Br + 3 Br + 2 (12.173) Br + l Br + l + 2 (12.174) (12.175)
33 onversion of hlorine Reservoirs to Active hlorine Fig % l, l 37% ln 2 62% l Before PS and photolysis reactions ln 2 l l, l After PS and photolysis reactions
Organic Chemistry. Chapter 23. Hill, Petrucci, McCreary & Perry 4 th. Ed. Alkane to Substituent Group methane CH 4 methyl CH 3
 hapter 23 rganic hemistry ill, Petrucci, Mcreary & Perry 4 th Ed. Alkane to Substituent Group methane 4 methyl 3 ethane 3 3 ethyl 3 2 propane 3 2 3 propyl 3 2 2 isopropyl ( 3 ) 2 or 3 3 butyl 3 2 2 2 butane
hapter 23 rganic hemistry ill, Petrucci, Mcreary & Perry 4 th Ed. Alkane to Substituent Group methane 4 methyl 3 ethane 3 3 ethyl 3 2 propane 3 2 3 propyl 3 2 2 isopropyl ( 3 ) 2 or 3 3 butyl 3 2 2 2 butane
Topic 6 : Structures and Reactions of Hydrocarbons Revised April Alkanes (General formula : C x H 2x+2 )
 6/1 Topic 6 : Structures and Reactions of ydrocarbons Revised April 1995 ydrocarbons all contain arbon and ydrogen only. There are various families of hydrocarbons (subsets of the set of hydrocarbons)
6/1 Topic 6 : Structures and Reactions of ydrocarbons Revised April 1995 ydrocarbons all contain arbon and ydrogen only. There are various families of hydrocarbons (subsets of the set of hydrocarbons)
Supplement of Evaluation of NO + reagent ion chemistry for online measurements of atmospheric volatile organic compounds
 Supplement of Atmos. Meas. Tech., 9, 2909 2925, 2016 http://www.atmos-meas-tech.net/9/2909/2016/ doi:10.5194/amt-9-2909-2016-supplement Author(s) 2016. CC Attribution 3.0 License. Supplement of Evaluation
Supplement of Atmos. Meas. Tech., 9, 2909 2925, 2016 http://www.atmos-meas-tech.net/9/2909/2016/ doi:10.5194/amt-9-2909-2016-supplement Author(s) 2016. CC Attribution 3.0 License. Supplement of Evaluation
H y., are burned in 100 cm 3 of oxygen, which is an excess of oxygen.
 1 (a) Hydrocarbons are compounds which contain hydrogen and carbon only. 10 cm 3 of a gaseous hydrocarbon, C x H y, are burned in 100 cm 3 of oxygen, which is an excess of oxygen. After cooling to room
1 (a) Hydrocarbons are compounds which contain hydrogen and carbon only. 10 cm 3 of a gaseous hydrocarbon, C x H y, are burned in 100 cm 3 of oxygen, which is an excess of oxygen. After cooling to room
Teacher s Tools Chemistry Organic Chemistry: Nomenclature and Isomerism
 1. Hydrocarbons: a) Naming of hydrocarbons is done based on the number of carbons. 1 = meth 6 = hex 2 = eth 7 = hept 3 = prop 8 = oct 4 = but 9 = non 5 = pent 10 = dec b) Alkanes are hydrocarbons without
1. Hydrocarbons: a) Naming of hydrocarbons is done based on the number of carbons. 1 = meth 6 = hex 2 = eth 7 = hept 3 = prop 8 = oct 4 = but 9 = non 5 = pent 10 = dec b) Alkanes are hydrocarbons without
Chapter 4 - Carbon Compounds
 Chapter 4 - Carbon Compounds Carbon compounds organic compounds are tied up with living organisms. So much so, that as we have seen, the presence of methane might be considered an indicator of life. Methane
Chapter 4 - Carbon Compounds Carbon compounds organic compounds are tied up with living organisms. So much so, that as we have seen, the presence of methane might be considered an indicator of life. Methane
ENVIRONMENTAL CHAMBER STUDIES OF ATMOSPHERIC OZONE FORMATION FROM SELECTED BIOGENIC COMPOUNDS
 ENVIRONMENTAL CHAMBER STUDIES OF ATMOSPHERIC OZONE FORMATION FROM SELECTED BIOGENIC COMPOUNDS by WILLIAM P. L. CARTER DONGMIN LUO IRINA MALKINA JOHN PIERCE STATEWIDE AIR POLLUTION RESEARCH CENTER AND COLLEGE
ENVIRONMENTAL CHAMBER STUDIES OF ATMOSPHERIC OZONE FORMATION FROM SELECTED BIOGENIC COMPOUNDS by WILLIAM P. L. CARTER DONGMIN LUO IRINA MALKINA JOHN PIERCE STATEWIDE AIR POLLUTION RESEARCH CENTER AND COLLEGE
BAERLIN2014 stationary measurements and source apportionment at an urban background station in Berlin, Germany
 Supplemental Information for BAERLIN2014 stationary measurements and source apportionment at an urban background station in Berlin, Germany Erika von Schneidemesser, Boris Bonn, Tim M. Butler, Christian
Supplemental Information for BAERLIN2014 stationary measurements and source apportionment at an urban background station in Berlin, Germany Erika von Schneidemesser, Boris Bonn, Tim M. Butler, Christian
1. The compound which reacts fastest with Lucas reagent at room temperature is
 1. The compound which reacts fastest with Lucas reagent at room temperature is (a) butan-1-ol (b) butan-2-ol (c) 2-methylpropan-1-ol (d) 2-methylpropan-2-ol 2. Chlorination of toluene in the presence of
1. The compound which reacts fastest with Lucas reagent at room temperature is (a) butan-1-ol (b) butan-2-ol (c) 2-methylpropan-1-ol (d) 2-methylpropan-2-ol 2. Chlorination of toluene in the presence of
S4 Chemistry National 5
 S4 hemistry National 5 Nature s hemistry Unit ourse Notes- ydrocarbons and onsumer Products Name lass 1 ydrocarbons ydrocarbons can be categorised into family groups known as OMOLOGOUS SERIES. A homologous
S4 hemistry National 5 Nature s hemistry Unit ourse Notes- ydrocarbons and onsumer Products Name lass 1 ydrocarbons ydrocarbons can be categorised into family groups known as OMOLOGOUS SERIES. A homologous
TO-3 BTEX and TPH. Table 13.1a Summary of QC Criteria for TO-3 BTEX Oxygenates
 TO-3 BTEX and TPH Method TO-3 uses cryogenic trapping and a gas chromatograph with a flame ionization detector (FID) to measure volatile organic compounds collected in Summa Canisters or Tedlar Bags. This
TO-3 BTEX and TPH Method TO-3 uses cryogenic trapping and a gas chromatograph with a flame ionization detector (FID) to measure volatile organic compounds collected in Summa Canisters or Tedlar Bags. This
Organic Chemistry. AQA Chemistry topic 7
 rganic hemistry AQA hemistry topic 7 7.1 arbon ompounds as fuels and feedstock rude il rude oil is a finite resource found in rocks. It s the remains of an ancient biomass consisting mainly of plankton
rganic hemistry AQA hemistry topic 7 7.1 arbon ompounds as fuels and feedstock rude il rude oil is a finite resource found in rocks. It s the remains of an ancient biomass consisting mainly of plankton
4. CARBON AND ITS COMPOUND
 . ARBN AND ITS MPUND rganic hemistry- The study of carbon compounds. rganic ompounds- The compounds of carbon (except the oxides of carbon, carbonates, hydro carbonates and carbides) are called organic
. ARBN AND ITS MPUND rganic hemistry- The study of carbon compounds. rganic ompounds- The compounds of carbon (except the oxides of carbon, carbonates, hydro carbonates and carbides) are called organic
large molecules small molecules fuels carbon
 4 10 5 12 6 14 4 2 6 3 8 methane ethane propane butane, pentane & hexane n 2n+2 Saturated hydrocarbons Insoluble in water n 2n Unsaturated hydrocarbons Insoluble in water Burns in plentiful 2 with a clean
4 10 5 12 6 14 4 2 6 3 8 methane ethane propane butane, pentane & hexane n 2n+2 Saturated hydrocarbons Insoluble in water n 2n Unsaturated hydrocarbons Insoluble in water Burns in plentiful 2 with a clean
Radicals. Structure and Stability of Radicals. Radicals are formed from covalent bonds by adding energy in the form of heat (Δ) or light (hν).
 Radicals Chapter 15 A small but significant group of reactions involve radical intermediates. A radical is a reactive intermediate with a single unpaired electron, formed by homolysis of a covalent bond.
Radicals Chapter 15 A small but significant group of reactions involve radical intermediates. A radical is a reactive intermediate with a single unpaired electron, formed by homolysis of a covalent bond.
Uncertainties in isoprene-no x -O 3 chemistry: Implications for surface ozone over the eastern United States PAR TEMP
 Uncertainties in isoprene-no x -O 3 chemistry: Implications for surface ozone over the eastern United States ISOPRENE + NO x O 3 PAR TEMP Leaf Area Arlene M. Fiore Telluride Atmospheric Chemistry Workshop
Uncertainties in isoprene-no x -O 3 chemistry: Implications for surface ozone over the eastern United States ISOPRENE + NO x O 3 PAR TEMP Leaf Area Arlene M. Fiore Telluride Atmospheric Chemistry Workshop
Decreasing wavelength Increasing frequency, energy (& potential damage) UV-C: blocked by ozone UV-B: blocked by ozone UV-A: only blocked
 Ozone Shield a natural process that filters radiati on before it reaches the lower atmosphere. In : Concentration of ozone in this layer is While ozone is made primarily at the equator, there is about
Ozone Shield a natural process that filters radiati on before it reaches the lower atmosphere. In : Concentration of ozone in this layer is While ozone is made primarily at the equator, there is about
Farr High School HIGHER CHEMISTRY. Unit 2 Nature s Chemistry. Question Booklet
 Farr igh School IGER EMISTRY Unit 2 Nature s hemistry Question Booklet 1 ydrocarbons 1. Write the systematic names for a) b) 3 2 3 2 2 3 2 3 3 2. Write the structural formulae and names of 3 isomers of
Farr igh School IGER EMISTRY Unit 2 Nature s hemistry Question Booklet 1 ydrocarbons 1. Write the systematic names for a) b) 3 2 3 2 2 3 2 3 3 2. Write the structural formulae and names of 3 isomers of
Chapter 19-4: Ozone Loss
 Chapter 19-4: Ozone Loss Ozone Shield a natural process that filters ultraviolet (UV) radiation before it reaches the lower atmosphere. In stratosphere: Concentration of ozone in this layer is
Chapter 19-4: Ozone Loss Ozone Shield a natural process that filters ultraviolet (UV) radiation before it reaches the lower atmosphere. In stratosphere: Concentration of ozone in this layer is
Save My Exams! The Home of Revision For more awesome GCSE and A level resources, visit us at Alkenes.
 Save My Exams! The ome of Revision For more awesome GSE and A level resources, visit us at www.savemyexams.co.uk/ Alkenes Question Paper 3 Level IGSE Subject hemistry ExamBoard IE Topic Organic hemistry
Save My Exams! The ome of Revision For more awesome GSE and A level resources, visit us at www.savemyexams.co.uk/ Alkenes Question Paper 3 Level IGSE Subject hemistry ExamBoard IE Topic Organic hemistry
Fuels. 1. Combustion is an example of an exothermic reaction which will give out energy, endothermic reactions are the opposite
 Fuels 1. ombustion is an example of an exothermic reaction which will give out energy, endothermic reactions are the opposite overed ( ) ow well can you do this? 2. The energy given out by a fuel can be
Fuels 1. ombustion is an example of an exothermic reaction which will give out energy, endothermic reactions are the opposite overed ( ) ow well can you do this? 2. The energy given out by a fuel can be
ANSWERS: Isomers. 2) A and F
 NSWERS: Isomers 1) For cis and trans isomers to occur a carbon-carbon double bond must be present as this prevents any rotation about this bond, and the atoms or groups of atoms attached to the two carbon
NSWERS: Isomers 1) For cis and trans isomers to occur a carbon-carbon double bond must be present as this prevents any rotation about this bond, and the atoms or groups of atoms attached to the two carbon
13. ORGANIC CHEMISTRY
 1. ORGANIC EMISTRY III) ALKENES SYNOPSIS Alkenes are unsaturated hydrocarbons. These contain a C =C. They contain two hydrogens less than corresponding alkanes. Double bonded carbon undergoes hybridisation.
1. ORGANIC EMISTRY III) ALKENES SYNOPSIS Alkenes are unsaturated hydrocarbons. These contain a C =C. They contain two hydrogens less than corresponding alkanes. Double bonded carbon undergoes hybridisation.
ALCOHOLS, ETHERS, PHENOLS, AND THIOLS
 C22 09/17/2013 11:27:34 Page 319 APTER 22 ALCLS, ETHERS, PHENLS, AND THILS SLUTINS T REVIEW QUESTINS 1. The question allows great freedom of choice. These shown here are very simple examples of each type.
C22 09/17/2013 11:27:34 Page 319 APTER 22 ALCLS, ETHERS, PHENLS, AND THILS SLUTINS T REVIEW QUESTINS 1. The question allows great freedom of choice. These shown here are very simple examples of each type.
6. HALOCARBONS :IN SITU STATION :FLASK STATION 90N 60N 30N 30S 60S 90S
 6. HALOCARBONS :IN SITU STATION :FLASK STATION 90N 60N 30N EQ 30S 60S 90S 120W 60W 0 60E 120E 180 (a) CFC-11 Monthly Data 120 140 160 180 200 220 240 260 280 300 AMY236N00 LMP635N00 KSN233N00 (b) CFC-12
6. HALOCARBONS :IN SITU STATION :FLASK STATION 90N 60N 30N EQ 30S 60S 90S 120W 60W 0 60E 120E 180 (a) CFC-11 Monthly Data 120 140 160 180 200 220 240 260 280 300 AMY236N00 LMP635N00 KSN233N00 (b) CFC-12
Alkenes. Question Paper 1. Chemistry (0620/0971) Cambridge International Examinations (CIE) Topic. Organic chemistry Sub-Topic. Alkenes.
 or more awesome resources, visit us at www.savemyexams.co.uk/ lkenes Question Paper 1 Level IGSE Subject hemistry (0620/0971) Exam oard ambridge International Examinations (IE) Topic Organic chemistry
or more awesome resources, visit us at www.savemyexams.co.uk/ lkenes Question Paper 1 Level IGSE Subject hemistry (0620/0971) Exam oard ambridge International Examinations (IE) Topic Organic chemistry
CHAPTER4 ANSWERS. Multiple Choice Questions. Short Answer Questions. 1. (b) 2. (d) 3. (a) 4. (c) 5. (c) 6. (b) 7. (a) 8. (b)
 CHAPTER4 ANSWERS Multiple Choice Questions 1. (b) 2. (d) 3. (a) 4. (c) 5. (c) 6. (b) 7. (a) 8. (b) 9. (a) 10. (d) 11. (a) 12. (d) 13. (b) 14. (a) 15. (c) 16. (c) 17. (c) 18. (d) 19. (c) 20. (a) 21. (b)
CHAPTER4 ANSWERS Multiple Choice Questions 1. (b) 2. (d) 3. (a) 4. (c) 5. (c) 6. (b) 7. (a) 8. (b) 9. (a) 10. (d) 11. (a) 12. (d) 13. (b) 14. (a) 15. (c) 16. (c) 17. (c) 18. (d) 19. (c) 20. (a) 21. (b)
Solution CHEMISTRY (B) (CH3O)2CH COOH & HCHO. (A) Sulphur (B) Phosphorus (C) Magnesium (D) Chlorine
 CHEMISTRY 31. H H OH H OH H OH H OCH 3 OCH 3 A & B are HIO 4 A B ( 2H2O) (A) HCHO & HCOCH(OCH3)2 (C) (CH3O)2C = O and COOH COOH (B) (CH3O)2CH COOH & HCHO (D) HCOOH & HCOCH(OCH3)2 32. In vulcanization of
CHEMISTRY 31. H H OH H OH H OH H OCH 3 OCH 3 A & B are HIO 4 A B ( 2H2O) (A) HCHO & HCOCH(OCH3)2 (C) (CH3O)2C = O and COOH COOH (B) (CH3O)2CH COOH & HCHO (D) HCOOH & HCOCH(OCH3)2 32. In vulcanization of
Alkenes and Alkynes: Structure and Nomenclature
 hapter 3 3 : Structure and Nomenclature APTER SUMMARY 3.1 Introduction to Alkenes are hydrocarbons in which there is at least one carbon-carbon double bond; alkynes have at least one carbon-carbon triple
hapter 3 3 : Structure and Nomenclature APTER SUMMARY 3.1 Introduction to Alkenes are hydrocarbons in which there is at least one carbon-carbon double bond; alkynes have at least one carbon-carbon triple
Chapter 8 Lecture Reactions of Alkenes
 Organic Chemistry, 9 th Edition L. G. Wade, Jr. Chapter 8 Lecture Reactions of Alkenes 2017 Pearson Education, Inc. Catalytic Hydrogenation of Alkenes Hydrogen (H 2 ) can be added across the double bond
Organic Chemistry, 9 th Edition L. G. Wade, Jr. Chapter 8 Lecture Reactions of Alkenes 2017 Pearson Education, Inc. Catalytic Hydrogenation of Alkenes Hydrogen (H 2 ) can be added across the double bond
Carbon s unique bonding pattern arises from the hybridization of the electrons.
 Unit 8 Neptune, the 8 th planet of our solar system Organic Chemistry Organic: compound containing carbon, excluding oxides and carbonates Carbon is an allotrope, meaning it has different bonding patterns.
Unit 8 Neptune, the 8 th planet of our solar system Organic Chemistry Organic: compound containing carbon, excluding oxides and carbonates Carbon is an allotrope, meaning it has different bonding patterns.
CfE Higher Chemistry Homework. Unit 2: Natures Chemistry. The Chemistry of Cooking and Oxidation of Food. 1. Which of the following is an aldehyde?
 CfE Higher Chemistry Homework Unit 2: Natures Chemistry The Chemistry of Cooking and Oxidation of Food 1. Which of the following is an aldehyde? 2. Which is true of a compound with the following formula?
CfE Higher Chemistry Homework Unit 2: Natures Chemistry The Chemistry of Cooking and Oxidation of Food 1. Which of the following is an aldehyde? 2. Which is true of a compound with the following formula?
Chapter 12 Alkenes & Alkynes. Organic and BioChem
 hapter 12 Alkenes & Alkynes Organic and Biohem Section 12.1 Introduction Unsaturated ydrocarbons ontain one or more carbon-carbon double or triple bonds 3 6? 2 2? Three lasses of Unsaturated ydrocarbons
hapter 12 Alkenes & Alkynes Organic and Biohem Section 12.1 Introduction Unsaturated ydrocarbons ontain one or more carbon-carbon double or triple bonds 3 6? 2 2? Three lasses of Unsaturated ydrocarbons
Part 1: Imaging Spectroscopy: From air quality investigations to healthcare applications
 Part 1: Imaging Spectroscopy: From air quality investigations to healthcare applications Part 2: Chemical Ionisation Reaction Mass Spectrometry (CIR-MS) for Breath Analysis Research 12 th March 2009 Roland
Part 1: Imaging Spectroscopy: From air quality investigations to healthcare applications Part 2: Chemical Ionisation Reaction Mass Spectrometry (CIR-MS) for Breath Analysis Research 12 th March 2009 Roland
Chapter 05 Structure and Preparation of Alkenes: Elimination Reactions part 01
 hapter 05 Structure and Preparation of Alkenes: Elimination Reactions part 01 EM 341: Spring 2012 Prof. Greg ook 2012 Gregory R ook Alkenes ompounds that contain double bonds. Ethylene (Ethene) 2 2012
hapter 05 Structure and Preparation of Alkenes: Elimination Reactions part 01 EM 341: Spring 2012 Prof. Greg ook 2012 Gregory R ook Alkenes ompounds that contain double bonds. Ethylene (Ethene) 2 2012
Unsaturated Hydrocarbons
 Interchapter G Unsaturated ydrocarbons FPO aption TK. G. unsaturated ydrocarbons G1 In this Interchapter, we shall continue our introduction to organic chemistry by discussing unsaturated hydrocarbons.
Interchapter G Unsaturated ydrocarbons FPO aption TK. G. unsaturated ydrocarbons G1 In this Interchapter, we shall continue our introduction to organic chemistry by discussing unsaturated hydrocarbons.
Organic photosynthetic reactions
 rganic photosynthetic reactions Singlet oxygen 2 " excited state! excited state 3 2 ground state atomic orbitals molecular orbitals atomic orbitals 5 rganic photosynthetic reactions Singlet oxygen lifetime
rganic photosynthetic reactions Singlet oxygen 2 " excited state! excited state 3 2 ground state atomic orbitals molecular orbitals atomic orbitals 5 rganic photosynthetic reactions Singlet oxygen lifetime
Characterization of isoprene and its oxidation products at a remote subtropical forested mountain site, South China
 Characterization of isoprene and its oxidation products at a remote subtropical forested mountain site, South China Daocheng Gong, Hao Wang, Boguang Wang *, etc. Institute for Environmental and Climate
Characterization of isoprene and its oxidation products at a remote subtropical forested mountain site, South China Daocheng Gong, Hao Wang, Boguang Wang *, etc. Institute for Environmental and Climate
Oxidizing Alcohols. Questions. Prediction. Analysis. Safety Precautions. Materials. Conclusions. Procedure. 74 MHR Unit 1 Organic Chemistry
 xidizing Alcohols SKILL FUS Predicting Performing and recording Analyzing and interpreting Acidified potassium permanganate solution, KMn 4(aq), acts as an oxidizing agent when it comes in contact with
xidizing Alcohols SKILL FUS Predicting Performing and recording Analyzing and interpreting Acidified potassium permanganate solution, KMn 4(aq), acts as an oxidizing agent when it comes in contact with
CH 3 CH 2 CH 2 CH 2 OH
 1 The alcohols form a homologous series. The first member is methanol and the fourth is butanol. 3 O methanol 3 2 2 2 O butanol (a) Give two general characteristics of a homologous series. (ii) alculate
1 The alcohols form a homologous series. The first member is methanol and the fourth is butanol. 3 O methanol 3 2 2 2 O butanol (a) Give two general characteristics of a homologous series. (ii) alculate
Chapter 2 MASS SPECTROMETRY
 Chapter 2 MASS SPECTMETY The mass spectrometer is an instrument that utilizes a variety of methods to ionize organic and biological compounds into simple cations and cationic species containing one unpaired
Chapter 2 MASS SPECTMETY The mass spectrometer is an instrument that utilizes a variety of methods to ionize organic and biological compounds into simple cations and cationic species containing one unpaired
Oregon State University
 H 223 Worksheet 9 Notes Oregon State University 1. Draw a primary alcohol and name it. OH 1-propanol Note: A primary alcohol has the form RH 2 OH; a secondary alcohol has the form R 2 H OH; and a tertiary
H 223 Worksheet 9 Notes Oregon State University 1. Draw a primary alcohol and name it. OH 1-propanol Note: A primary alcohol has the form RH 2 OH; a secondary alcohol has the form R 2 H OH; and a tertiary
EPA TO-15 QC Criteria
 EPA TO-15 QC Criteria Method TO-15 is used for measuring volatile organic compounds collected in SUMMA canisters by Full Scan GC/MS. The difference between TO-14 and TO-15 is the method of water management.
EPA TO-15 QC Criteria Method TO-15 is used for measuring volatile organic compounds collected in SUMMA canisters by Full Scan GC/MS. The difference between TO-14 and TO-15 is the method of water management.
Chapterwise Previous year Qs
 221 ALCOHAL PHENOLS AND ETHERS 1. Which one is formed when sodium phenoxide is heated with ethyl iodide? [1988] Phenetole Ethyl phenyl alcohol Phenol None of these Ans: 2. Lucas reagent is [1988] Conc.
221 ALCOHAL PHENOLS AND ETHERS 1. Which one is formed when sodium phenoxide is heated with ethyl iodide? [1988] Phenetole Ethyl phenyl alcohol Phenol None of these Ans: 2. Lucas reagent is [1988] Conc.
UNSATURATED HYDROCARBONS
 hemistry 52 hapter 20 UNSATURATED YDROARBONS The unsaturated hydrocarbons consist of three families of homologous compounds that contain multiple bonds between carbon atoms. Alkenes contain carbon carbon
hemistry 52 hapter 20 UNSATURATED YDROARBONS The unsaturated hydrocarbons consist of three families of homologous compounds that contain multiple bonds between carbon atoms. Alkenes contain carbon carbon
Advanced oxidation processes for the removal of [bmim][sal] third generation ionic liquid: Effect of water matrices and intermediates identification
![Advanced oxidation processes for the removal of [bmim][sal] third generation ionic liquid: Effect of water matrices and intermediates identification Advanced oxidation processes for the removal of [bmim][sal] third generation ionic liquid: Effect of water matrices and intermediates identification](/thumbs/96/128474283.jpg) Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2016 Electronic Supplementary Information Advanced oxidation processes for the removal of [bmim][sal]
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2016 Electronic Supplementary Information Advanced oxidation processes for the removal of [bmim][sal]
Carbon and the Molecular Diversity of Life. Chapter 4
 Carbon and the Molecular Diversity of Life Chapter 4 CARBON Carbon is has ability to form large and complex, molecules Aspirin molecular formula? A triglyceride Organic chemistry is study of carbon compounds
Carbon and the Molecular Diversity of Life Chapter 4 CARBON Carbon is has ability to form large and complex, molecules Aspirin molecular formula? A triglyceride Organic chemistry is study of carbon compounds
13. Carboxylic Acids (text )
 2009, Department of Chemistry, The University of Western ntario 13.1 13. Carboxylic Acids (text 14.1 14.9) A. Structure and Nomenclature The carboxylic acid functional group results from the connection
2009, Department of Chemistry, The University of Western ntario 13.1 13. Carboxylic Acids (text 14.1 14.9) A. Structure and Nomenclature The carboxylic acid functional group results from the connection
Outline of Presentation
 Indoor Secondary Organic Aerosol Formation and Health Effects Annette C. Rohr, Sc.D. October 24, 2006 Health Effects of Organic Aerosols Workshop Palo Alto, CA Outline of Presentation Background Indoor
Indoor Secondary Organic Aerosol Formation and Health Effects Annette C. Rohr, Sc.D. October 24, 2006 Health Effects of Organic Aerosols Workshop Palo Alto, CA Outline of Presentation Background Indoor
INVESTIGATION OF CHLORINATED METHANES TREATABILITY USING ACTIVATED SODIUM PERSULFATE
 Preprint: Proceedings of the First International Conference on Environmental Science and Technology (2005) INVESTIGATION OF CHLORINATED METHANES TREATABILITY USING ACTIVATED SODIUM PERSULFATE Duane K.
Preprint: Proceedings of the First International Conference on Environmental Science and Technology (2005) INVESTIGATION OF CHLORINATED METHANES TREATABILITY USING ACTIVATED SODIUM PERSULFATE Duane K.
Alkenes. Isomerism in the alkenes
 Alkenes Alkenes are a family of hydrocarbons (compounds containing carbon and hydrogen only) containing a carbon-carbon double bond. The first two are: ethene 2 4 propene 3 6 You can work out the formula
Alkenes Alkenes are a family of hydrocarbons (compounds containing carbon and hydrogen only) containing a carbon-carbon double bond. The first two are: ethene 2 4 propene 3 6 You can work out the formula
Nature s Chemistry. Fragrances and Skin Care
 St Andrew s and St Bride s High School CfE Higher Chemistry Nature s Chemistry Page 1 of 12 Essential Oils Essential oils are concentrated extracts of the volatile, non-water soluble (hydrophobic) aroma
St Andrew s and St Bride s High School CfE Higher Chemistry Nature s Chemistry Page 1 of 12 Essential Oils Essential oils are concentrated extracts of the volatile, non-water soluble (hydrophobic) aroma
Level 3 Chemistry, 2007
 Level 3 hemistry, 2007 Annotated answers to this organic paper. Q1 QUESTIN NE Give the proper name that gives the structure a unique name (a) Give the systematic IUPA names for the following molecules
Level 3 hemistry, 2007 Annotated answers to this organic paper. Q1 QUESTIN NE Give the proper name that gives the structure a unique name (a) Give the systematic IUPA names for the following molecules
Calibration Guide. 301IRFS / S301D2 with VA301EM
 Calibration Guide 301IRFS / S301D2 with VA301EM 301IRFS / S301D2 with VA301EM Calibration Procedure 1. Warm-Up Period Calibration Handbook Turn the unit on for a minimum of fifteen (15) minutes. Check
Calibration Guide 301IRFS / S301D2 with VA301EM 301IRFS / S301D2 with VA301EM Calibration Procedure 1. Warm-Up Period Calibration Handbook Turn the unit on for a minimum of fifteen (15) minutes. Check
Chemicals Based on Ethylene
 Chemicals Based on Ethylene Ethylene is sometimes known as the king of petrochemicals because more commercial chemicals are produced from ethylene than from any other intermediate. This unique position
Chemicals Based on Ethylene Ethylene is sometimes known as the king of petrochemicals because more commercial chemicals are produced from ethylene than from any other intermediate. This unique position
CH [2] (ii) Give the structural formula of another hydrocarbon which is isomeric with the above.
![CH [2] (ii) Give the structural formula of another hydrocarbon which is isomeric with the above. CH [2] (ii) Give the structural formula of another hydrocarbon which is isomeric with the above.](/thumbs/75/72821632.jpg) 1 The alkenes are unsaturated hydrocarbons. They form a homologous series, the members of which have the same chemical properties. They undergo addition reactions and are easily oxidised. (a) The following
1 The alkenes are unsaturated hydrocarbons. They form a homologous series, the members of which have the same chemical properties. They undergo addition reactions and are easily oxidised. (a) The following
Homologous Series. Learning Outcomes :
 omologous Series N5 Learning Outcomes : Revision of homologous series including alkanes, alkenes, cycloalkanes, amines Reactions of alkenes including halogenoalkanes Systematic naming of hydrocarbons,
omologous Series N5 Learning Outcomes : Revision of homologous series including alkanes, alkenes, cycloalkanes, amines Reactions of alkenes including halogenoalkanes Systematic naming of hydrocarbons,
Fundamentals of Organic Chemistry CHEM 109 For Students of Health Colleges Credit hrs.: (2+1)
 Fundamentals of Organic Chemistry CHEM 109 For Students of Health Colleges Credit hrs.: (2+1) King Saud University College of Science, Chemistry Department CHEM 109 CHAPTER 7. CARBOXYLIC ACIDS AND THEIR
Fundamentals of Organic Chemistry CHEM 109 For Students of Health Colleges Credit hrs.: (2+1) King Saud University College of Science, Chemistry Department CHEM 109 CHAPTER 7. CARBOXYLIC ACIDS AND THEIR
This is an addition reaction. (Other types of reaction have been substitution and elimination). Addition reactions are typically exothermic.
 Reactions of Alkenes Since bonds are stronger than bonds, double bonds tend to react to convert the double bond into bonds + X-Y X Y This is an addition reaction. (Other types of reaction have been substitution
Reactions of Alkenes Since bonds are stronger than bonds, double bonds tend to react to convert the double bond into bonds + X-Y X Y This is an addition reaction. (Other types of reaction have been substitution
National 5 Unit Two : Nature s Chemistry
 National 5 Unit Two : Nature s Chemistry Fuels A fuel is a chemical which burns, giving off energy. Combustion is a reaction of a substance with oxygen giving off energy. The test for oxygen is it relights
National 5 Unit Two : Nature s Chemistry Fuels A fuel is a chemical which burns, giving off energy. Combustion is a reaction of a substance with oxygen giving off energy. The test for oxygen is it relights
Name the ester produced when methanol and pentanoic acid react. methyl pentanoate. Name the type of reaction used to make an ester
 1 Name the ester produced when methanol and pentanoic acid react methyl pentanoate 2 Name the type of reaction used to make an ester condensation reaction 3 Name the by-product of the reaction used to
1 Name the ester produced when methanol and pentanoic acid react methyl pentanoate 2 Name the type of reaction used to make an ester condensation reaction 3 Name the by-product of the reaction used to
Recent Changes in Eastern U.S. Forests: Implications for Air Quality
 Recent Changes in Eastern U.S. Forests: Implications for Air Quality Arlene M. Fiore Seminar at the Center for Sustainability and the Global Environment (SAGE) University of Wisconsin-Madison December
Recent Changes in Eastern U.S. Forests: Implications for Air Quality Arlene M. Fiore Seminar at the Center for Sustainability and the Global Environment (SAGE) University of Wisconsin-Madison December
Evaluation of a New Ionic Liquid Stationary Phase with PEG Like Selectivity
 Evaluation of a New Ionic Liquid Stationary Phase with PEG Like Selectivity L.M.Sidisky, G. A. Baney, J.L. Desorcie, D.L. Shollenberger, G. Serrano, and K.K. Stenerson Supelco, Div. of Sigma-Aldrich Bellefonte,
Evaluation of a New Ionic Liquid Stationary Phase with PEG Like Selectivity L.M.Sidisky, G. A. Baney, J.L. Desorcie, D.L. Shollenberger, G. Serrano, and K.K. Stenerson Supelco, Div. of Sigma-Aldrich Bellefonte,
DAV CENTENARY PUBLIC SCHOOL, PASCHIM ENCLAVE, NEW DELHI
 1How will you bring about the following conversions? (i) Ethanol to but-1-yne Ethane to bromoethene (iii) Propene to 1-nitropropane (iv) Toluene to benzyl alcohol (v) Propene to propyne (vi) Ethanol to
1How will you bring about the following conversions? (i) Ethanol to but-1-yne Ethane to bromoethene (iii) Propene to 1-nitropropane (iv) Toluene to benzyl alcohol (v) Propene to propyne (vi) Ethanol to
Assessing and Improving the Isoprene Oxidation Mechanism
 CRC Report No. A-97 Assessing and Improving the Isoprene Oxidation Mechanism Executive Summary May 2016 COORDINATING RESEARCH COUNCIL, INC. 5755 NORTH POINT PARKWAY SUITE 265 ALPHARETTA, GA 30022 The Coordinating
CRC Report No. A-97 Assessing and Improving the Isoprene Oxidation Mechanism Executive Summary May 2016 COORDINATING RESEARCH COUNCIL, INC. 5755 NORTH POINT PARKWAY SUITE 265 ALPHARETTA, GA 30022 The Coordinating
Chapter 20 Carboxylic Acids. Introduction
 hapter 20 arboxylic Acids Introduction arbonyl (-=) and hydroxyl (-H) on the same carbon is carboxyl group. arboxyl group is usually written -H or 2 H. Aliphatic acids have an alkyl group bonded to -H.
hapter 20 arboxylic Acids Introduction arbonyl (-=) and hydroxyl (-H) on the same carbon is carboxyl group. arboxyl group is usually written -H or 2 H. Aliphatic acids have an alkyl group bonded to -H.
Chapter 3 Structure and Stereochemistry of Alkanes. Classification Review. Alkenes: Structure and Stereochem Slide 3-2
 hapter 3 Structure and Stereochemistry of Alkanes lassification Review Alkenes: Structure and Stereochem Slide 3-2 1 Alkane Structural Formulas All - single bonds Saturated with hydrogens (no pi bonds)
hapter 3 Structure and Stereochemistry of Alkanes lassification Review Alkenes: Structure and Stereochem Slide 3-2 1 Alkane Structural Formulas All - single bonds Saturated with hydrogens (no pi bonds)
Supplement of Secondary organic aerosol formation from the β-pinene+no 3 system: effect of humidity and peroxy radical fate
 Supplement of Atmos. Chem. Phys., 15, 7497 7522, 2015 http://www.atmos-chem-phys.net/15/7497/2015/ doi:10.5194/acp-15-7497-2015-supplement Author(s) 2015. CC Attribution 3.0 License. Supplement of Secondary
Supplement of Atmos. Chem. Phys., 15, 7497 7522, 2015 http://www.atmos-chem-phys.net/15/7497/2015/ doi:10.5194/acp-15-7497-2015-supplement Author(s) 2015. CC Attribution 3.0 License. Supplement of Secondary
Chapter 15 Alcohols, Diols, and Thiols
 Chapter 15 Alcohols, Diols, and Thiols 15.1 Sources of Alcohols Methanol Methanol is an industrial chemical end uses: solvent, antifreeze, fuel principal use: preparation of formaldehyde Methanol Methanol
Chapter 15 Alcohols, Diols, and Thiols 15.1 Sources of Alcohols Methanol Methanol is an industrial chemical end uses: solvent, antifreeze, fuel principal use: preparation of formaldehyde Methanol Methanol
Biogenic chemistry in a region of declining emissions: Insights from the NASA SEAC 4 RS Campaign
 Biogenic chemistry in a region of declining emissions: Insights from the NASA SEAC 4 RS Campaign Jenny A. Fisher University of Wollongong with Daniel Jacob, Katherine Travis, Patrick Kim, Eloise Marais,
Biogenic chemistry in a region of declining emissions: Insights from the NASA SEAC 4 RS Campaign Jenny A. Fisher University of Wollongong with Daniel Jacob, Katherine Travis, Patrick Kim, Eloise Marais,
Observational constraints on the chemistry of isoprene nitrates over the eastern United States
 Click Here for Full Article JOURNAL OF GEOPHYSICAL RESEARCH, VOL. 112,, doi:10.1029/2006jd007747, 2007 Observational constraints on the chemistry of isoprene nitrates over the eastern United States Larry
Click Here for Full Article JOURNAL OF GEOPHYSICAL RESEARCH, VOL. 112,, doi:10.1029/2006jd007747, 2007 Observational constraints on the chemistry of isoprene nitrates over the eastern United States Larry
Current ERPG TM Values (2011)
 Current ERPG TM Values (2011) *Please refer to the key at the end of the table.* Chemical (CAS Number) ERPG-1 ERPG-2 ERPG-3 LEL*** Chemical (CAS Number) ERPG-1 ERPG-2 ERPG-3 LEL*** Acetaldehyde (75-07-0)...10
Current ERPG TM Values (2011) *Please refer to the key at the end of the table.* Chemical (CAS Number) ERPG-1 ERPG-2 ERPG-3 LEL*** Chemical (CAS Number) ERPG-1 ERPG-2 ERPG-3 LEL*** Acetaldehyde (75-07-0)...10
CHEM 263 Notes Nov 5, 2013
 EM 263 Notes Nov 5, 2013 Ketone Naming Example Another naming example involves Gyrinal (shown below), which is an anti-microbial agent from the water boatman beetle (orixa species), and is toxic to fish
EM 263 Notes Nov 5, 2013 Ketone Naming Example Another naming example involves Gyrinal (shown below), which is an anti-microbial agent from the water boatman beetle (orixa species), and is toxic to fish
Vast majority of over 20 million known compounds are based on C: Generally contain C and H + other elements Great variety of compounds
 1 Vast majority of over 20 million known compounds are based on C: Generally contain C and + other elements Great variety of compounds 2 3 Isomers have identical composition but different structures Two
1 Vast majority of over 20 million known compounds are based on C: Generally contain C and + other elements Great variety of compounds 2 3 Isomers have identical composition but different structures Two
11/5/ Oxidation of Alkenes: Cleavage to Carbonyl Compounds. Oxidation of Alkenes: Cleavage to Carbonyl Compounds
 8.8 Oxidation of Alkenes: Cleavage to Carbonyl Compounds Ozone (O 3 ) is useful double-bond cleavage reagent Ozone is generated by passing a stream of oxygen through a highvoltage electrical discharge
8.8 Oxidation of Alkenes: Cleavage to Carbonyl Compounds Ozone (O 3 ) is useful double-bond cleavage reagent Ozone is generated by passing a stream of oxygen through a highvoltage electrical discharge
Carboxylic Acids and Their Derivatives
 arboxylic Acids and Their Derivatives Families ontaining the arbonyl Group Family Y Z Y Z aldehyde or ketone carboxylic acid or -- ester or -- acid halide or -F,-l,-Br,-I acid anhydride or amide or -N
arboxylic Acids and Their Derivatives Families ontaining the arbonyl Group Family Y Z Y Z aldehyde or ketone carboxylic acid or -- ester or -- acid halide or -F,-l,-Br,-I acid anhydride or amide or -N
CHM 424L Organic Laboratory, Dr. Laurie S. Starkey Introduction to Mass Spectrometry
 CM 424L rganic Laboratory, Dr. Laurie S. Starkey Introduction to Mass Spectrometry Mass spectrometry is used to determine a sample's molecular mass and molecular formula. Some structural information can
CM 424L rganic Laboratory, Dr. Laurie S. Starkey Introduction to Mass Spectrometry Mass spectrometry is used to determine a sample's molecular mass and molecular formula. Some structural information can
Alkane C-C single bond (propane) Alkene C=C double bond (propene) Alcohol - OH group (1-propanol) major. minor
 Functional group* and name? Alkane - single bond (propane) *alkanes not really regarded as a functional group Alkene = double bond (propene) Addition of an unsymmetrical reagent to unsymmetrical alkene
Functional group* and name? Alkane - single bond (propane) *alkanes not really regarded as a functional group Alkene = double bond (propene) Addition of an unsymmetrical reagent to unsymmetrical alkene
Understanding Dissolved Gas Analysis of Ester Liquids
 Understanding Dissolved Gas Analysis of Ester Liquids an Updated Review of Gas Generated in Ester Liquid by Stray Gassing, Thermal Decomposition and Electrical Discharge Initially presented at the 216
Understanding Dissolved Gas Analysis of Ester Liquids an Updated Review of Gas Generated in Ester Liquid by Stray Gassing, Thermal Decomposition and Electrical Discharge Initially presented at the 216
Site Specific Conditional Sampler Garfield County, Colorado. VOC Data Summaries. Prepared for
 Site Specific Conditional Sampler Garfield County, Colorado VOC Data Summaries Prepared for Garfield County Public Health 195 West 14 th Street Rifle, Colorado 81650 Prepared by 1901 Sharp Point Dr., Suite
Site Specific Conditional Sampler Garfield County, Colorado VOC Data Summaries Prepared for Garfield County Public Health 195 West 14 th Street Rifle, Colorado 81650 Prepared by 1901 Sharp Point Dr., Suite
Alcohol aldehydes cetones and carboxylic acids
 Alcohol aldehydes cetones and carboxylic acids 1 Classes of organic compounds 2 Alcohols Alcohols are organic compounds containing hydroxyl (-OH) group attached to C atom. In an alcohol, -OH group replaces
Alcohol aldehydes cetones and carboxylic acids 1 Classes of organic compounds 2 Alcohols Alcohols are organic compounds containing hydroxyl (-OH) group attached to C atom. In an alcohol, -OH group replaces
Naming Organic Halide Organic Halide: is a compound that contains one or more halogen atoms (F, Cl, Br, I) as part of its molecular structure.
 Naming Organic Halide Organic Halide: is a compound that contains one or more halogen atoms (F, Cl, Br, I) as part of its molecular structure. Organic halides have many important uses including: fire retardation,
Naming Organic Halide Organic Halide: is a compound that contains one or more halogen atoms (F, Cl, Br, I) as part of its molecular structure. Organic halides have many important uses including: fire retardation,
A. CONFORMATIONAL ISOMERS
 EMISTRY 104 elp Sheet #3 Organic (Part II): ISOMERS hapters 2.9 (structural isomers), 6.5 (geometric isomers), 6.11 (constitutional isomers), 7.2f (optical isomers) Do the topics appropriate for your lecture
EMISTRY 104 elp Sheet #3 Organic (Part II): ISOMERS hapters 2.9 (structural isomers), 6.5 (geometric isomers), 6.11 (constitutional isomers), 7.2f (optical isomers) Do the topics appropriate for your lecture
APPENDIX K FY 2000 REPORT TO CONGRESS ON THE USE OF OZONE DEPLETING SUBSTANCES
 APPENDIX K FY 2000 REPORT TO CONGRESS ON THE USE OF OZONE DEPLETING SUBSTANCES Ozone is a bluish gas that is harmful to breathe. Nearly 90 percent of the Earth's ozone is in the stratosphere, which is
APPENDIX K FY 2000 REPORT TO CONGRESS ON THE USE OF OZONE DEPLETING SUBSTANCES Ozone is a bluish gas that is harmful to breathe. Nearly 90 percent of the Earth's ozone is in the stratosphere, which is
Compatibility Nonwoven Oil And Universal Sorbents
 Compatibility Nonwoven Oil And Universal Sorbents Pads Rolls Pillows Drum Top Covers Socks Flakes ADR Strips Spillosens sorbents rapidly and effectively remove liquid spills and leaks. As a precautionary
Compatibility Nonwoven Oil And Universal Sorbents Pads Rolls Pillows Drum Top Covers Socks Flakes ADR Strips Spillosens sorbents rapidly and effectively remove liquid spills and leaks. As a precautionary
Lab 5: Reactions of Organic Compounds and Qualitative Analysis
 Lab 5: eactions of rganic Compounds and Qualitative Analysis bjectives: - To better understand several chemical reactions. - To identify an unknown chemical by testing its chemical and physical properties.
Lab 5: eactions of rganic Compounds and Qualitative Analysis bjectives: - To better understand several chemical reactions. - To identify an unknown chemical by testing its chemical and physical properties.
PRACTICE PROBLEMS CHEMISTRY
 PRATIE PRBLEMS EMISTRY G ame: Batch: Date: 1. Which nitrogen in LSD is most basic: 1 2 3 3 ( 2 5 ) 2 (A) 1 2 () 3 All are equally basic 2. Which of the following statement for 2 = 2, 2 = 3 2 is / are correct.
PRATIE PRBLEMS EMISTRY G ame: Batch: Date: 1. Which nitrogen in LSD is most basic: 1 2 3 3 ( 2 5 ) 2 (A) 1 2 () 3 All are equally basic 2. Which of the following statement for 2 = 2, 2 = 3 2 is / are correct.
Alcohols, Phenols, Ethers And Thiols Lec:3
 Alcohols, Phenols, Ethers And Thiols Lec:3 The word alcohol refers to a class of compounds that contain an group called a hydroxyl or hydroxyl group, bounded to an alkyl group. Alcohols can be viewed as
Alcohols, Phenols, Ethers And Thiols Lec:3 The word alcohol refers to a class of compounds that contain an group called a hydroxyl or hydroxyl group, bounded to an alkyl group. Alcohols can be viewed as
Current ERPG Values (2016)
 Current ERPG Values (2016) *Please refer to the key at the end of the table.* Chemical (CAS Number) ERPG-1 ERPG-2 ERPG-3 LEL*** Acetaldehyde (75-07-0)...10 ppmj 200 ppm 1000 ppm Acetaldehyde (75-07-0)...
Current ERPG Values (2016) *Please refer to the key at the end of the table.* Chemical (CAS Number) ERPG-1 ERPG-2 ERPG-3 LEL*** Acetaldehyde (75-07-0)...10 ppmj 200 ppm 1000 ppm Acetaldehyde (75-07-0)...
Lab 6: Reactions of Organic Compounds and Qualitative Analysis
 Lab 6: eactions of rganic Compounds and Qualitative Analysis bjectives: - To better understand several chemical reactions. - To identify an unknown chemical by testing its chemical and physical properties.
Lab 6: eactions of rganic Compounds and Qualitative Analysis bjectives: - To better understand several chemical reactions. - To identify an unknown chemical by testing its chemical and physical properties.
Firrhill High School CfE Higher Chemistry
 Firrhill High School CfE Higher Chemistry Unit 2 Homework Natures Chemistry 1 1. Write the general formula for the alkanols. Alcohols, Carboxylic Acids and Esters 2. Draw the structural formulae for each
Firrhill High School CfE Higher Chemistry Unit 2 Homework Natures Chemistry 1 1. Write the general formula for the alkanols. Alcohols, Carboxylic Acids and Esters 2. Draw the structural formulae for each
Mass Spectrometry Introduction
 Mass Spectrometry Introduction Chem 744 Spring 2013 What MS is and is not MS is NOT a spectroscopic method. Molecules are not absorbing EM radiation MS is the generation, separation and characterization
Mass Spectrometry Introduction Chem 744 Spring 2013 What MS is and is not MS is NOT a spectroscopic method. Molecules are not absorbing EM radiation MS is the generation, separation and characterization
6/9/2015. Unit 15: Organic Chemistry Lesson 15.2: Substituted Hydrocarbons & Functional Groups
 1-chloropropane 2-methylpropane 1-iodobutane Ethanoic Acid Unit 15: Organic Chemistry Lesson 15.2: Substituted Hydrocarbons & Functional Groups 43 It Ain t Just Hydrocarbons There are all sorts of organic
1-chloropropane 2-methylpropane 1-iodobutane Ethanoic Acid Unit 15: Organic Chemistry Lesson 15.2: Substituted Hydrocarbons & Functional Groups 43 It Ain t Just Hydrocarbons There are all sorts of organic
TWA [2] (mg/m3) [5] (ppm) [6]
![TWA [2] (mg/m3) [5] (ppm) [6] TWA [2] (mg/m3) [5] (ppm) [6]](/thumbs/76/74282081.jpg) (2-Butoxyethoxy) ethanol 112-34-5 67.5 10 101.2 15 - DIR 2006/15/CE (2-Methoxyethoxy)ethanol 111-77-3 50.1 10 - - Skin DIR 2006/15/CE Acetic acid 64-19-7 25 10 - - - DIR 91/322/CE Acetone 67-64-1 1210
(2-Butoxyethoxy) ethanol 112-34-5 67.5 10 101.2 15 - DIR 2006/15/CE (2-Methoxyethoxy)ethanol 111-77-3 50.1 10 - - Skin DIR 2006/15/CE Acetic acid 64-19-7 25 10 - - - DIR 91/322/CE Acetone 67-64-1 1210
Supplement of Formaldehyde production from isoprene oxidation across NOx regimes
 Supplement of Atmos. Chem. Phys., 16, 2597 2610, 2016 http://www.atmos-chem-phys.net/16/2597/2016/ doi:10.5194/acp-16-2597-2016-supplement Author(s) 2016. CC Attribution 3.0 License. Supplement of Formaldehyde
Supplement of Atmos. Chem. Phys., 16, 2597 2610, 2016 http://www.atmos-chem-phys.net/16/2597/2016/ doi:10.5194/acp-16-2597-2016-supplement Author(s) 2016. CC Attribution 3.0 License. Supplement of Formaldehyde
TEST-7/11 th, 12 th & 12 th Passed /Set-P
 III-A/172, Nehru Nagar Ghaziabad. Ph : 9811212090, 9868502091. email : bmcchemistry@gmail.com, website : www.bmcchemistry.com Type : JEE MAIN 2015-16 TEST-7/11 th, 12 th & 12 th Passed /Set-P Topic : rganic
III-A/172, Nehru Nagar Ghaziabad. Ph : 9811212090, 9868502091. email : bmcchemistry@gmail.com, website : www.bmcchemistry.com Type : JEE MAIN 2015-16 TEST-7/11 th, 12 th & 12 th Passed /Set-P Topic : rganic
Diverse Reactions of Alkenes
 Chapter 8- Alkenes: Reactions and Synthesis Ashley Piekarski, Ph.D. Diverse Reactions of Alkenes Alkenes react with many electrophiles to give useful products by addiaon (ocen through special reagents)
Chapter 8- Alkenes: Reactions and Synthesis Ashley Piekarski, Ph.D. Diverse Reactions of Alkenes Alkenes react with many electrophiles to give useful products by addiaon (ocen through special reagents)
Organic Chemistry Part 2
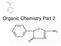 Organic Chemistry Part 2 Benzene Benzene is a special structure C 6 H 6 The carbon-carbon bonds aren t a single or double bond but something in-between Resonance bond CYCLIC HYDROCARBONS Carbon chains
Organic Chemistry Part 2 Benzene Benzene is a special structure C 6 H 6 The carbon-carbon bonds aren t a single or double bond but something in-between Resonance bond CYCLIC HYDROCARBONS Carbon chains
Alehydes, Ketones and Carboxylic Acid
 Alehydes, Ketones and Carboxylic Acid Aldehydes and Ketones: Introduction Aldedydes and ketones are organic compounds that contain carbon-oxygen doule bonds. The general formula for aldehydes is O C R
Alehydes, Ketones and Carboxylic Acid Aldehydes and Ketones: Introduction Aldedydes and ketones are organic compounds that contain carbon-oxygen doule bonds. The general formula for aldehydes is O C R
4/7/2011. Chapter 13 Organic Chemistry. Structural Formulas. 3. Petroleum Products
 Chapter 13 Organic Chemistry 13-1. Carbon Bonds 13-2. Alkanes 13-3. Petroleum Products 13-5. Isomers 13-6. Unsaturated Hydrocarbons 13-7. Benzene 13-9. 13-10. Polymers 13-11. Carbohydrates 13-12. Photosynthesis
Chapter 13 Organic Chemistry 13-1. Carbon Bonds 13-2. Alkanes 13-3. Petroleum Products 13-5. Isomers 13-6. Unsaturated Hydrocarbons 13-7. Benzene 13-9. 13-10. Polymers 13-11. Carbohydrates 13-12. Photosynthesis
