EGFR kinase activity is required for TLR4 signaling and the septic shock response
|
|
|
- Jean Robinson
- 5 years ago
- Views:
Transcription
1 Manuscript EMBOR EGFR kinase activity is required for TLR4 signaling and the septic shock response Saurabh Chattopadhyay, Manoj Veleeparambil, Darshana Poddar, Samar Abdulkhalek, Sudip K. Bandyopadhyay, Volker Fensterl and Ganes C. Sen Corresponding author: Ganes C. Sen, Cleveland Clinic Review timeline: Submission date: 08 March 2015 Editorial Decision: 01 April 2015 Revision received: 12 June 2015 Editorial Decision: 28 July 2015 Revision received: 10 August 2015 Accepted: 17 August 2015 Transaction Report: (Note: With the exception of the correction of typographical or spelling errors that could be a source of ambiguity, letters and reports are not edited. The original formatting of letters and referee reports may not be reflected in this compilation.) Editor: Nonia Pariente 1st Editorial Decision 01 April 2015 Thank you for your submission to EMBO reports. We have now received the three enclosed reports on it. As you will see, although all the referees find the topic of interest and in principle suitable for us, they also consider the study is incomplete and insufficiently conclusive at this stage. Given that all referees provide constructive suggestions on how to make the work more conclusive and insightful, I would like to give you the opportunity to revise your manuscript. If the referee concerns can be adequately addressed, we would be happy to accept your manuscript for publication. However, please note that it is EMBO reports policy to undergo one round of revision only and thus, acceptance of your study will depend on the outcome of the next, final round of peerreview. Revised manuscripts must be submitted within three months of a request for unless a short extension has been discussed with the editor; they will otherwise be treated as new submissions. When preparing your revision, please note that you manuscript will be published in full article format. This means that all materials and methods must be included in the main text, and results and discussion sections kept separate (as you have them). European Molecular Biology Organization 1
2 I look forward to seeing a revised form of your manuscript when it is ready. In the meantime, please contact me if I can be of any assistance. REFEREE REPORTS: Referee #1: In the manuscript "EGFR kinase activity is required for TLR4 signaling and the septic shock response", the authors investigate whether EGFR is important for the transcriptional responses induced by LPS stimulation. The paper is well written and the authors demonstrate that the activity of EGFR is physiologically relevant in that EGFR inhibitors can protect mice against LPS-induced septic shock However, there are a number of problems with interpretation of the biochemical data presented, and alternative explanations for the observations made are not fully considered. Major Comments: 1. Using chemical inhibitors and shrna depletion, the authors show that the kinase activity of EGFR is required for the expression of a subset of LPS-dependent genes. The authors argue that Gefitinb, an EGFR inhibitor, specifically inhibits a subset of LPS-induced genes and does not affect MYD88-dependent genes like TNF. However, Figure 2A suggests that the kinetics of TNF expression are delayed in the presence of Gefitinib (Gf) (i.e, in the absence of Gf TNF expression peaks at 2 hrs while in the presence of Gf TNF peaks at 6 hrs). A more detailed analysis of the kinetics of LPS induced genes may reveal an additional phenotype. Furthermore, there is no discussion about how the expression of IL-1beta, a MYD88-dependent gene, is controlled by EGFR kinase activity. 2. In figure 3, the authors examine the GF-dependent inhibition in MYD88-/- cells. Given the wide body of literature arguing that TNF is a MYD88-dependent gene, I am surprised that LPS still induced a strong TNF response in the absence of MYD88. These responses should be examined side-by-side with matched wild-type cells. 3. Figures 4-6 are the most confusing figures. While the authors nicely show that EGFR is required for both AKT phosphorylation and IRF3-phosphorylation/ nuclear accumulation, the authors attempt to argue that the primary role of EGFR is to activate AKT and subsequently B-catenin. However, the role of EGFR in promoting IRF3 nuclear accumulation cannot be ignored. The data support that AKT activity is required for B-catenin phosphorylation/ nuclear accumulation, but that EGFR may have multiple roles in activating this signaling pathway. 4. The data presented suggests that signaling from the plasma membrane by MYD88 is intact whereas signaling from TRIF on endosomes in blocked. Since EGFR is a known to promote endocytosis, why have the authors not considered the possibility that their inhibitors somehow block LPS-induced TLR4 endocytosis? Ample literature exists to support this idea, as blocking TLR4 endocytosis blocks TRIF signaling, but leaves MYD88 signaling intact. The authors are encouraged to 1) determine if TLR4 endocytosis is influenced by EGFR, and 2) determine if EGFR endocytosis is influenced by LPS treatment. 5. Is EGFR activated in response to LPS? If so, what is the ligand? Minor Points: -Statistics should be performed on transcriptions European Molecular Biology Organization 2
3 Referee #2: This paper suggests a novel form of interaction between the TLR4 and EGFR signaling pathways. The authors provide data that the EGFR tyrosine kinase activity is necessary selectively for the activation of TRIF dependent transcription after stimulation with LPS. EGFR kinase inhibitors such as Gefitinib prevented LPS-induced Akt and IRF3 phosphorylation leading to reduced transcription of IRF-induced genes such as Ifit1 and IFNb. They propose a mechanism by which EGFR signaling is required for PI3K-dependent activation of b-catenin, which in turn is required as a co-activator for IRF-mediated transcription, a finding that they partly had already reported in a previous paper. The authors also provide in vivo relevance for the interaction between TLR4 and EGFR by demonstrating reduced LPS-induced sepsis in mice in the presence of Gefitinib. The findings are interesting and most of the experiments are conclusive. However, the mechanism how TLR 4 and EGFR signaling interact is only poorly clarified. Major points 1) Mechanism of LPS-mediated EGFR transactivation should be better analyzed. The rapid response of IRF phosphorylation and transcriptional induction of IFN target genes indicates that EGFR signaling is ready to be turned on in these cells. This might be explained by EGFR ligands present in the serum of the culture medium. The use of serum free culture conditions and EGFR ligands in combination with LPS could clarify some of these questions. Alternatively, EGFR transactivation could be a possible mechanism. Does LPS stimulation lead to EGFR phosphorylation? If yes, how? Are EGFR ligands or the proteases that cleave them induced by LPS treatment? Is there direct interaction and activation? Despite the fact that Gefitinib is used in a lot of experiments, the authors never show that EGFR is expressed in the cells employed (except for KD experiments), nor do they show inhibition of EGFR phosphorylation after Gefitinib treatment. Additionally the use of another, more specific EGFR inhibitor, like Cetuximab, that inhibits ligand binding, should also be considered to address some of these questions. 2) LPS-mediated TLR4 signaling can occur at the level of the cell membrane (via Myd88 and TIRAP) and at the level of the endosome (via TRIF and TRAM). Only the endosomal TLR4 signaling seems to be affected by the EGFR. Where are the two receptors localized in the studied cells before and after LPS stimulation? The authors might want to explain how and where the interaction/transactivation of TLR4 and EGFR occurs (at the level of membrane or endosome?). 3) For all the results showing RNA expression or cytokine production etc. (Fig. 1, 2, 3, 6 and Suppl Fig S1 and S2) there is no statistical analysis mentioned (no p values etc.) Are the differences observed statistically significant? Minor points 4) In Figure 3, the experiment performed with Tunicamycin provides only indirect evidence for the importance of the TRIF adaptor. The use of TRIF knockout macrophages would be preferable to confirm this finding. 5) The reduction of IRF3 in the nuclear fraction of Gf treated cells (Fig. 5B) does not look convincing and seems like a blotting problem. Referee #3: This MS is a fine extension of published previous work describing, that the TLR3 driven TRIF signalling pathway requires EGFR kinase activity. Because Lps sensing TLR4 activation is critical for sepsis induction, and because Lpps triggers via TLR4 the independently operating MyD88 and TRIF pathway,the authors are able to recapitulate and extend their published TLR3-TRIF-EGFR data in the TLR4-TRIF system.specifically they show that specific EGFR kinase inhibitors block the Tlr4 driven PI3/Akt pathway required for activation of the co-factor beta-catenin,that in turn is needed for phosphorilation of TRIF at Ser 552. The signal pathways adressed have been described/published in other systems. However, that TLR4 uses in Lps driven septic schock EGFR to activate PI3/AKT is novel, and important.the data given are fine, yet somehow incomplete. The authors are urged to adress the question whether ligand activated TLR4 recruits EGFR (Coprecipitation experiments)if so TLR4 might be a target of EGFR. European Molecular Biology Organization 3
4 1st Revision - authors' response 12 June 2015 Reviewers' Comments: Referee #1: In the manuscript "EGFR kinase activity is required for TLR4 signaling and the septic shock response", the authors investigate whether EGFR is important for the transcriptional responses induced by LPS stimulation. The paper is well written and the authors demonstrate that the activity of EGFR is physiologically relevant in that EGFR inhibitors can protect mice against LPS-induced septic shock However, there are a number of problems with interpretation of the biochemical data presented, and alternative explanations for the observations made are not fully considered. Major Comments: 1. Using chemical inhibitors and shrna depletion, the authors show that the kinase activity of EGFR is required for the expression of a subset of LPS-dependent genes. The authors argue that Gefitinb, an EGFR inhibitor, specifically inhibits a subset of LPS-induced genes and does not affect MYD88-dependent genes like TNF. However, Figure 2A suggests that the kinetics of TNF expression are delayed in the presence of Gefitinib (Gf) (i.e, in the absence of Gf TNF expression peaks at 2 hrs while in the presence of Gf TNF peaks at 6 hrs). A more detailed analysis of the kinetics of LPS induced genes may reveal an additional phenotype. Furthermore, there is no discussion about how the expression of IL-1beta, a MYD88-dependent gene, is controlled by EGFR kinase activity. As suggested, a more detailed kinetics of gene induction has now been done (new Figs 1A, 2A). We have now discussed why the induction of some NF-κB-driven genes might be EGFRdependent. 2. In figure 3, the authors examine the GF-dependent inhibition in MYD88-/- cells. Given the wide body of literature arguing that TNF is a MYD88-dependent gene, I am surprised that LPS still induced a strong TNF response in the absence of MYD88. These responses should be examined side-by-side with matched wild-type cells. The side-by-side comparison is now shown in the new Fig S4. It is known that the TRIF branch can also activate NF-κB (Ref#11, Yamamoto et al, Science 2003) 3. Figures 4-6 are the most confusing figures. While the authors nicely show that EGFR is required for both AKT phosphorylation and IRF3-phosphorylation/ nuclear accumulation, the authors attempt to argue that the primary role of EGFR is to activate AKT and subsequently B- catenin. However, the role of EGFR in promoting IRF3 nuclear accumulation cannot be ignored. The data support that AKT activity is required for B-catenin phosphorylation/ nuclear accumulation, but that EGFR may have multiple roles in activating this signaling pathway. We agree and now suggested multiple roles of EGFR in the Discussion. 4. The data presented suggests that signaling from the plasma membrane by MYD88 is intact whereas signaling from TRIF on endosomes in blocked. Since EGFR is a known to promote endocytosis, why have the authors not considered the possibility that their inhibitors somehow block LPS-induced TLR4 endocytosis? Ample literature exists to support this idea, as blocking TLR4 endocytosis blocks TRIF signaling, but leaves MYD88 signaling intact. The authors are encouraged to 1) determine if TLR4 endocytosis is influenced by EGFR, and 2) determine if EGFR endocytosis is influenced by LPS treatment. European Molecular Biology Organization 4
5 We considered this possibility but gefitinib did not inhibit TLR4 endocytosis (New Fig S5). The results with MyD88 KO cells (Fig 3) also suggest that TLR4 internalization was not affected by gefitinib because TNF was strongly induced in its presence. LPS had no effect on EGFR endocytosis. 5. Is EGFR activated in response to LPS? If so, what is the ligand? We could not detect any EGFR activation, as measured by its Tyr phosphorylation, in response to LPS treatment of cells. Minor Points: -Statistics should be performed on transcriptions Statistical analyses of the transcriptional data have now been added Referee #2: This paper suggests a novel form of interaction between the TLR4 and EGFR signaling pathways. The authors provide data that the EGFR tyrosine kinase activity is necessary selectively for the activation of TRIF dependent transcription after stimulation with LPS. EGFR kinase inhibitors such as Gefitinib prevented LPS-induced Akt and IRF3 phosphorylation leading to reduced transcription of IRF-induced genes such as Ifit1 and IFNb. They propose a mechanism by which EGFR signaling is required for PI3K-dependent activation of b-catenin, which in turn is required as a co-activator for IRF-mediated transcription, a finding that they partly had already reported in a previous paper. The authors also provide in vivo relevance for the interaction between TLR4 and EGFR by demonstrating reduced LPS-induced sepsis in mice in the presence of Gefitinib. The findings are interesting and most of the experiments are conclusive. However, the mechanism how TLR 4 and EGFR signaling interact is only poorly clarified. Major points 1) Mechanism of LPS-mediated EGFR transactivation should be better analyzed. The rapid response of IRF phosphorylation and transcriptional induction of IFN target genes indicates that EGFR signaling is ready to be turned on in these cells. This might be explained by EGFR ligands present in the serum of the culture medium. The use of serum free culture conditions and EGFR ligands in combination with LPS could clarify some of these questions. Alternatively, EGFR transactivation could be a possible mechanism. Does LPS stimulation lead to EGFR phosphorylation? If yes, how? Are EGFR ligands or the proteases that cleave them induced by LPS treatment? Is there direct interaction and activation? Despite the fact that Gefitinib is used in a lot of experiments, the authors never show that EGFR is expressed in the cells employed (except for KD experiments), nor do they show inhibition of EGFR phosphorylation after Gefitinib treatment. Additionally the use of another, more specific EGFR inhibitor, like Cetuximab, that inhibits ligand binding, should also be considered to address some of these questions. We did not observe any indication of LPS-mediated transactivation of EGFR, as measured by its phosphorylation. Experiments with serum free culture medium indicate that EGFR internalization promotes IFN gene induction by LPS (new Fig S7). The literature does not suggest any effect of LPS on EGFR ligand synthesis or processing. We have now shown, as suggested, the EGFR expression levels and the effect of gefitinib on EGFR phosphorylation (new Fig S1). As suggested, we have tested Cetuximab, which blocks EGF binding to cell surface EGFR only; it did not inhibit IFN induction by LPS suggesting that intracellular EGFR action promotes TLR4 signaling (new Fig S8). 2) LPS-mediated TLR4 signaling can occur at the level of the cell membrane (via Myd88 and TIRAP) and at the level of the endosome (via TRIF and TRAM). Only the endosomal TLR4 signaling seems to be affected by the EGFR. Where are the two receptors localized in the studied cells before European Molecular Biology Organization 5
6 and after LPS stimulation? The authors might want to explain how and where the interaction/transactivation of TLR4 and EGFR occurs (at the level of membrane or endosome?). We did not observe any physical interaction between EGFR and TLR4 (Fig 4A). LPS treatment triggered internalization of TLR4 and did not affect EGFR location; under our experimental conditions, EGFR was both on plasma and endosomal membranes. We believe that the functional interaction is between endosomal EGFR and endosomal TLR4. 3) For all the results showing RNA expression or cytokine production etc. (Fig. 1, 2, 3, 6 and Suppl Fig S1 and S2) there is no statistical analysis mentioned (no p values etc.) Are the differences observed statistically significant? Statistical analyses have now been included. Minor points 4) In Figure 3, the experiment performed with Tunicamycin provides only indirect evidence for the importance of the TRIF adaptor. The use of TRIF knockout macrophages would be preferable to confirm this finding. The TRIF KO experiment is already in the literature (Ref#25, Shenderov et al J Immunol 2014); we followed their protocol. 5) The reduction of IRF3 in the nuclear fraction of Gf treated cells (Fig. 5B) does not look convincing and seems like a blotting problem. The experiment has been repeated and the new results are shown in Fig 5B. Referee #3: This MS is a fine extension of published previous work describing, that the TLR3 driven TRIF signalling pathway requires EGFR kinase activity. Because Lps sensing TLR4 activation is critical for sepsis induction, and because Lpps triggers via TLR4 the independently operating MyD88 and TRIF pathway, the authors are able to recapitulate and extend their published TLR3-TRIF-EGFR data in the TLR4-TRIF system.specifically they show that specific EGFR kinase inhibitors block the Tlr4 driven PI3/Akt pathway required for activation of the co-factor beta-catenin,that in turn is needed for phosphorilation of TRIF at Ser 552. The signal pathways adressed have been described/published in other systems. However, that TLR4 uses in Lps driven septic schock EGFR to activate PI3/AKT is novel, and important. The data given are fine, yet somehow incomplete. The authors are urged to adress the question whether ligand activated TLR4 recruits EGFR (Coprecipitation experiments)if so TLR4 might be a target of EGFR. As suggested, we have performed the co-immunoprecipitation experiment and the results show that TLR4 and EGFR do not physically interact (Fig 4A). This suggests a more indirect functional interaction. 2nd Editorial Decision 28 July 2015 Thank you for the submission of your revised manuscript to our offices and please accept my apologies for the time we have needed to contact you with a decision on your study. Referees 2 and 3 (referee 1 was not available) have now returned their reports, which as you will see are not in agreement, which has prompted further discussion to make a decision on your study. Below you will find the reports of referees 2 and 3. While the latter is now supportive of publication, the former still has various issues that preclude him/her form supporting publication. On European Molecular Biology Organization 6
7 balance, we will not ask that you address all of them. However, a few do need to be addressed in a last round of minor revision before your study can be accepted for publication. Specifically, for a successful revision, we will require that: - you discuss the caveats of using HEK293 cells instead of macrophages/myeloid cells for some of the experiments - address point 7 from referee 2's report regarding supplementary figure 5, which needs to be strengthened (more cells shown, findings quantified, etc) - discuss previous literature supporting a role for EGFR in the induction of cytokines - provide a model summarizing the overall message of the study In addition, from an editorial point of view, some things also need to be taken care of: -please ensure that all relevant figures and supplementary figures have been generated according to proper statistical analysis procedures, and all figure legends include information on the number of independent experiments measured, the type of error bars used (SD, SE,...), statistical test applied to the data and exact P values considered significant. In figures 1-SF3, only information regarding the number of experiments is included. SF4-8 are missing all statistical information. We look forward to receiving a final version of your study as soon as it is ready, and always within the next four weeks. REFEREE REPORTS: Referee #2: This reviewer is not satisfied with the revision of the paper since many of the questions asked have not been adequately addressed (see point 1 of the previous revision). Little has been done towards a better mechanistic understanding of the pathways proposed in particular the elucidation of the mechanism how TLR 4 and EGFR signaling interact is still unclear. The observation that in serum starved medium LPS does not induce IFNb (Sup Fig 7A) points towards the need of EGFR ligands to induce EGFR and the suggested downstream pathways and target genes. However, this is still poorly examined. More mechanistic experiments could have been done around this finding. EGFR activation and the inhibitory effect of Gefitinib should have been shown in myeloid cells and not in HEK293 cells (Supl. Fig 1B). This is not what the reviewer asked for. This data are important to strengthen the evidence that the effects seen by EGFR inhibitors in myeloid cells are due to EGFR kinase inhibition. Moreover, this reviewer does not understand why the Co-IPs were performed in HEK293 cells which are not object of this study. These experiments should be performed in macrophages and/or myeloid cells used for the other studies in the paper. Signaling in HEK 293 cells might be completely different than in macrophages. None of the Western Blots in Fig. 4 includes EGFR and phospho-egfr analysis, which would also demonstrate to which extend EGFR phosphorylation is inhibited by Gefitinib is respect to Akt inhibition etc. Experiments with TRIF deficient BMDM should have been included similar to what performed in Fig. 3 with MyD88-/- cells. In Supl. Fig 4 the authors show how TNF production in Myd88 -/- cells compares to wild-type cells. European Molecular Biology Organization 7
8 This should be shown also for the other genes analyzed in Myd88-/- BMDM, which are shown in Fig. 3 such as IL1 and IFNs etc. In non-permeabilized cells it seems that there is more TLR4 signal on the cell surface after Gefitinib treatment? Is this a representative image (only one single cell is shown)? Moreover, the signal should be quantified before drawing conclusions. The same applies to permeabilized cells. The in vivo sepsis experiment is convincing. However, what about the expression of the other cytokines (analyzed in vitro) after in vivo LPS treatment in the presence of Gefitinib? Moreover, there have been several papers in the literature reporting EGFR requirement for the induction of cytokines. For example for IL-6 in macrophages there have been two papers published in J. Immunol in 2013 and Nat. Cell Biol which have not been mentioned by the authors. A model should be included that summarizes the major findings. Referee #3: This MS has been adaequately revised in view of the crticism raised by the reviewers.the revised version strongly favours the novel and unexpected interpretation, that EGFR kinase activity is required for TLR4 medited activation of the PI3k/Akt pathway, that in turn activates beta-catenin (an obligatory co-activator of IRF's). Since the authors provide compelling aruments/data that extracellular EGFR appears not to be involved, the authors conclude that a not yet understood "functional interaction of endodosomal EGFR and TLR4" comes into play.in other words, the data clearly show an essential role of EGFR kinase activity in TLR-4 driven type 1Interferon induction, yet the final answer of the mechanism is still lacking. Nevertheless, as it stands this well written MS provides novel and valid informations on TLR-4 mediated signalling. 2nd Revision - authors' response 10 August 2015 Thank you for editing our manuscript. We have revised it further following your instructions. The highlights of our revisions are as follows. The reasons and the caveats of using HEK293 cells have been discussed Fig S5 has been expanded and data quantified (new Fig S5B) Previous reports on EGFR and cytokine induction have been discussed and cited A working model has been provided and discussed (new Fig 7F) The missing statistical information has been provided Many original Western blot images have been deposited Uncompressed images have been provided for Figs 1E, 4D (new blot), 4E, 5D and 6C. Figs 5A and 5C have been replaced with less contrasted images I hope that the paper is now acceptable for publication. 3rd Editorial Decision 17 August 2015 I am very pleased to accept your manuscript for publication in the next available issue of EMBO reports. Thank you for your contribution to EMBO reports and congratulations on a successful publication. Please consider us again in the future for your most exciting work. European Molecular Biology Organization 8
NFIL3/E4BP4 controls Type 2 T helper cell cytokine expression
 Manuscript EMBO-2010-75657 NFIL3/E4BP4 controls Type 2 T helper cell cytokine expression Masaki Kashiwada, Suzanne L. Cassel, John D. Colgan and Paul B. Rothman Corresponding author: Paul Rothman, University
Manuscript EMBO-2010-75657 NFIL3/E4BP4 controls Type 2 T helper cell cytokine expression Masaki Kashiwada, Suzanne L. Cassel, John D. Colgan and Paul B. Rothman Corresponding author: Paul Rothman, University
The unfolded protein response is shaped by the NMD pathway
 Manuscript EMBOR-2014-39696 The unfolded protein response is shaped by the NMD pathway Rachid Karam, Chih-Hong Lou, Heike Kroeger, Lulu Huang, Jonathan H. Lin and Miles F. Wilkinson Corresponding author:
Manuscript EMBOR-2014-39696 The unfolded protein response is shaped by the NMD pathway Rachid Karam, Chih-Hong Lou, Heike Kroeger, Lulu Huang, Jonathan H. Lin and Miles F. Wilkinson Corresponding author:
Src-dependent autophagic degradation of Ret in FAKsignaling defective cancer cells
 Manuscript EMBOR-2011-35678 Src-dependent autophagic degradation of Ret in FAKsignaling defective cancer cells Emma Sandilands, Bryan Serrels, Simon Wilkinson and Margaret C Frame Corresponding author:
Manuscript EMBOR-2011-35678 Src-dependent autophagic degradation of Ret in FAKsignaling defective cancer cells Emma Sandilands, Bryan Serrels, Simon Wilkinson and Margaret C Frame Corresponding author:
Intracellular MHC class II molecules promote TLR-triggered innate. immune responses by maintaining Btk activation
 Intracellular MHC class II molecules promote TLR-triggered innate immune responses by maintaining Btk activation Xingguang Liu, Zhenzhen Zhan, Dong Li, Li Xu, Feng Ma, Peng Zhang, Hangping Yao and Xuetao
Intracellular MHC class II molecules promote TLR-triggered innate immune responses by maintaining Btk activation Xingguang Liu, Zhenzhen Zhan, Dong Li, Li Xu, Feng Ma, Peng Zhang, Hangping Yao and Xuetao
Myelin suppresses axon regeneration by PIR-B/SHPmediated inhibition of Trk activity
 Manuscript EMBO-2010-76298 Myelin suppresses axon regeneration by PIR-B/SHPmediated inhibition of Trk activity Yuki Fujita, Shota Endo, Toshiyuki Takai and Toshihide Yamashita Corresponding author: Toshihide
Manuscript EMBO-2010-76298 Myelin suppresses axon regeneration by PIR-B/SHPmediated inhibition of Trk activity Yuki Fujita, Shota Endo, Toshiyuki Takai and Toshihide Yamashita Corresponding author: Toshihide
GENETIC ANALYSIS OF RAS SIGNALING PATHWAYS IN CELL PROLIFERATION, MIGRATION AND SURVIVAL
 Manuscript EMBO-2009-72496 GENETIC ANALYSIS OF RAS SIGNALING PATHWAYS IN CELL PROLIFERATION, MIGRATION AND SURVIVAL Matthias Drosten, Alma Dhawahir, Eleanor Sum, Jelena Urosevic, Carmen Lechuga, Luis Esteban,
Manuscript EMBO-2009-72496 GENETIC ANALYSIS OF RAS SIGNALING PATHWAYS IN CELL PROLIFERATION, MIGRATION AND SURVIVAL Matthias Drosten, Alma Dhawahir, Eleanor Sum, Jelena Urosevic, Carmen Lechuga, Luis Esteban,
EGFR kinase activity is required for TLR4 signaling and the septic shock response
 rticle EGFR kinase activity is required for TLR4 signaling and the septic shock response Saurabh Chattopadhyay, Manoj Veleeparambil, Darshana Poddar, Samar bdulkhalek, Sudip K andyopadhyay, Volker Fensterl
rticle EGFR kinase activity is required for TLR4 signaling and the septic shock response Saurabh Chattopadhyay, Manoj Veleeparambil, Darshana Poddar, Samar bdulkhalek, Sudip K andyopadhyay, Volker Fensterl
Foxm1 Transcription Factor is Required for Lung Fibrosis and Epithelial to Mesenchymal Transition.
 Manuscript EMBO-2012-82682 Foxm1 Transcription Factor is Required for Lung Fibrosis and Epithelial to Mesenchymal Transition. David Balli, Vladimir Ustiyan, Yufang Zhang, I-Ching Wang, Alex J. Masino,
Manuscript EMBO-2012-82682 Foxm1 Transcription Factor is Required for Lung Fibrosis and Epithelial to Mesenchymal Transition. David Balli, Vladimir Ustiyan, Yufang Zhang, I-Ching Wang, Alex J. Masino,
Title:Role of LPAR3, PKC and EGFR in LPA-induced cell migration in oral squamous carcinoma cells
 Author's response to reviews Title:Role of LPAR3, PKC and EGFR in LPA-induced cell migration in oral squamous carcinoma cells Authors: Ingvild J Brusevold (i.j.brusevold@medisin.uio.no) Ingun H Tveteraas
Author's response to reviews Title:Role of LPAR3, PKC and EGFR in LPA-induced cell migration in oral squamous carcinoma cells Authors: Ingvild J Brusevold (i.j.brusevold@medisin.uio.no) Ingun H Tveteraas
Targeted proteomics reveals strain-specific changes in the mouse insulin and central metabolic pathways after sustained high-fat diet
 Molecular Systems Biology Peer Review Process File Targeted proteomics reveals strain-specific changes in the mouse insulin and central metabolic pathways after sustained high-fat diet Eduard Sabidó, Yibo
Molecular Systems Biology Peer Review Process File Targeted proteomics reveals strain-specific changes in the mouse insulin and central metabolic pathways after sustained high-fat diet Eduard Sabidó, Yibo
Tankyrase 1 regulates centrosome function by controlling CPAP stability
 Manuscript EMBOR-2011-35655 Tankyrase 1 regulates centrosome function by controlling CPAP stability Mi Kyung Kim, Charles Dudognon and Susan Smith Corresponding author: Susan Smith, The Skirball Institute
Manuscript EMBOR-2011-35655 Tankyrase 1 regulates centrosome function by controlling CPAP stability Mi Kyung Kim, Charles Dudognon and Susan Smith Corresponding author: Susan Smith, The Skirball Institute
Fibroadipogenic progenitors mediate the ability of HDAC inhibitors to promote regeneration in dystrophic muscles of young, but not old mdx mice
 Fibroadipogenic progenitors mediate the ability of HDAC inhibitors to promote regeneration in dystrophic muscles of young, but not old mdx mice Mozzetta C., Consalvi S., Saccone V., Tierney M., Diamantini
Fibroadipogenic progenitors mediate the ability of HDAC inhibitors to promote regeneration in dystrophic muscles of young, but not old mdx mice Mozzetta C., Consalvi S., Saccone V., Tierney M., Diamantini
Insulin-like growth factor-1 stimulates regulatory T cells and suppresses autoimmune disease
 Insulin-like growth factor-1 stimulates regulatory T cells and suppresses autoimmune disease Daniel Bilbao, Luisa Luciani, Bjarki Johannesson, Agnieszka Piszczek & Nadia Rosenthal Corresponding author:
Insulin-like growth factor-1 stimulates regulatory T cells and suppresses autoimmune disease Daniel Bilbao, Luisa Luciani, Bjarki Johannesson, Agnieszka Piszczek & Nadia Rosenthal Corresponding author:
Ubiquitination and deubiquitination of NP protein regulates influenza A virus RNA replication
 Manuscript EMBO-2010-74756 Ubiquitination and deubiquitination of NP protein regulates influenza A virus RNA replication Tsai-Ling Liao, Chung-Yi Wu, Wen-Chi Su, King-Song Jeng and Michael Lai Corresponding
Manuscript EMBO-2010-74756 Ubiquitination and deubiquitination of NP protein regulates influenza A virus RNA replication Tsai-Ling Liao, Chung-Yi Wu, Wen-Chi Su, King-Song Jeng and Michael Lai Corresponding
Physiological release of endogenous tau is stimulated by neuronal activity
 Manuscript EMBOR-2013-37101 Physiological release of endogenous tau is stimulated by neuronal activity Emma C Phillips, Dawn HW Lau, Wendy Noble, Diane P Hanger and Amy M Pooler Corresponding author: Amy
Manuscript EMBOR-2013-37101 Physiological release of endogenous tau is stimulated by neuronal activity Emma C Phillips, Dawn HW Lau, Wendy Noble, Diane P Hanger and Amy M Pooler Corresponding author: Amy
TD-BF01: Innate immunity to microorganisms
 TD-BF01: Innate immunity to microorganisms I. Toll receptors (adapted from Takeuchi, O. et al. (1999) Immunity 11:443; Kawai, T. et al. (1999) Immunity 11:115; Hemmi, H. et al. (2000) Nature 408:740; Muzio,
TD-BF01: Innate immunity to microorganisms I. Toll receptors (adapted from Takeuchi, O. et al. (1999) Immunity 11:443; Kawai, T. et al. (1999) Immunity 11:115; Hemmi, H. et al. (2000) Nature 408:740; Muzio,
Supplementary Figure 1
 Supplementary Figure 1 Constitutive EGFR signaling does not activate canonical EGFR signals (a) U251EGFRInd cells with or without tetracycline exposure (24h, 1µg/ml) were treated with EGF for 15 minutes
Supplementary Figure 1 Constitutive EGFR signaling does not activate canonical EGFR signals (a) U251EGFRInd cells with or without tetracycline exposure (24h, 1µg/ml) were treated with EGF for 15 minutes
Toll-like Receptor Signaling
 Toll-like Receptor Signaling 1 Professor of Medicine University of Massachusetts Medical School, Worcester, MA, USA Why do we need innate immunity? Pathogens multiply very fast We literally swim in viruses
Toll-like Receptor Signaling 1 Professor of Medicine University of Massachusetts Medical School, Worcester, MA, USA Why do we need innate immunity? Pathogens multiply very fast We literally swim in viruses
S1a S1b S1c. S1d. S1f S1g S1h SUPPLEMENTARY FIGURE 1. - si sc Il17rd Il17ra bp. rig/s IL-17RD (ng) -100 IL-17RD
 SUPPLEMENTARY FIGURE 1 0 20 50 80 100 IL-17RD (ng) S1a S1b S1c IL-17RD β-actin kda S1d - si sc Il17rd Il17ra rig/s15-574 - 458-361 bp S1f S1g S1h S1i S1j Supplementary Figure 1. Knockdown of IL-17RD enhances
SUPPLEMENTARY FIGURE 1 0 20 50 80 100 IL-17RD (ng) S1a S1b S1c IL-17RD β-actin kda S1d - si sc Il17rd Il17ra rig/s15-574 - 458-361 bp S1f S1g S1h S1i S1j Supplementary Figure 1. Knockdown of IL-17RD enhances
RNA:DNA hybrids are a novel molecular pattern sensed by TLR9
 Manuscript EMBO-2013-86117 RNA:DNA hybrids are a novel molecular pattern sensed by TLR9 Rachel E Rigby, Lauren M Webb, Karen J MacKenzie, Yue Li, Andrea Leitch, Martin AM Reijns, Rachel J Lundie, Ailsa
Manuscript EMBO-2013-86117 RNA:DNA hybrids are a novel molecular pattern sensed by TLR9 Rachel E Rigby, Lauren M Webb, Karen J MacKenzie, Yue Li, Andrea Leitch, Martin AM Reijns, Rachel J Lundie, Ailsa
Xenopus Paraxial Protocadherin inhibits Wnt/β -catenin signalling via Casein Kinase 2β
 Manuscript EMBOR-2011-35178 Xenopus Paraxial Protocadherin inhibits Wnt/β -catenin signalling via Casein Kinase 2β Anja Kietzmann, Yingqun Wang, Dominik Weber and Herbert Steinbeisser Corresponding author:
Manuscript EMBOR-2011-35178 Xenopus Paraxial Protocadherin inhibits Wnt/β -catenin signalling via Casein Kinase 2β Anja Kietzmann, Yingqun Wang, Dominik Weber and Herbert Steinbeisser Corresponding author:
Toll-like Receptors (TLRs): Biology, Pathology and Therapeutics
 Toll-like Receptors (TLRs): Biology, Pathology and Therapeutics Dr Sarah Sasson SydPATH Registrar 23 rd June 2014 TLRs: Introduction Discovered in 1990s Recognise conserved structures in pathogens Rely
Toll-like Receptors (TLRs): Biology, Pathology and Therapeutics Dr Sarah Sasson SydPATH Registrar 23 rd June 2014 TLRs: Introduction Discovered in 1990s Recognise conserved structures in pathogens Rely
RAW264.7 cells stably expressing control shrna (Con) or GSK3b-specific shrna (sh-
 1 a b Supplementary Figure 1. Effects of GSK3b knockdown on poly I:C-induced cytokine production. RAW264.7 cells stably expressing control shrna (Con) or GSK3b-specific shrna (sh- GSK3b) were stimulated
1 a b Supplementary Figure 1. Effects of GSK3b knockdown on poly I:C-induced cytokine production. RAW264.7 cells stably expressing control shrna (Con) or GSK3b-specific shrna (sh- GSK3b) were stimulated
A transient reversal of mirna-mediated repression controls macrophage activation
 Manuscript EMBOR-2013-37763 A transient reversal of mirna-mediated repression controls macrophage activation Anup Mazumder, Mainak Bose, Abhijit Chakraborty, Saikat Chakrabarti and Suvendra N. Bhattacharyya
Manuscript EMBOR-2013-37763 A transient reversal of mirna-mediated repression controls macrophage activation Anup Mazumder, Mainak Bose, Abhijit Chakraborty, Saikat Chakrabarti and Suvendra N. Bhattacharyya
The RNA binding protein Arrest (Bruno) regulates alternative splicing to enable myofibril maturation in Drosophila flight muscle
 Manuscript EMBOR-2014-39791 The RNA binding protein Arrest (Bruno) regulates alternative splicing to enable myofibril maturation in Drosophila flight muscle Maria L. Spletter, Christiane Barz, Assa Yeroslaviz,
Manuscript EMBOR-2014-39791 The RNA binding protein Arrest (Bruno) regulates alternative splicing to enable myofibril maturation in Drosophila flight muscle Maria L. Spletter, Christiane Barz, Assa Yeroslaviz,
EGF receptor transactivation is crucial for cholinergic MAP kinase signaling in human keratinocytes
 1st Electronic Conference on Molecular Science EGF receptor transactivation is crucial for cholinergic MAP kinase signaling in human keratinocytes Wymke Ockenga, Sina Kühne, Antje Banning and Ritva Tikkanen
1st Electronic Conference on Molecular Science EGF receptor transactivation is crucial for cholinergic MAP kinase signaling in human keratinocytes Wymke Ockenga, Sina Kühne, Antje Banning and Ritva Tikkanen
Opposing activities of the Ras and Hippo pathways converge on regulation of YAP protein turnover
 The EMBO Journal Peer Review Process File - EMBO-2014-89385 Manuscript EMBO-2014-89385 Opposing activities of the Ras and Hippo pathways converge on regulation of YAP protein turnover Xin Hong, Hung Thanh
The EMBO Journal Peer Review Process File - EMBO-2014-89385 Manuscript EMBO-2014-89385 Opposing activities of the Ras and Hippo pathways converge on regulation of YAP protein turnover Xin Hong, Hung Thanh
Whole genome sequencing identifies zoonotic transmission of MRSA isolates with the novel meca homologue mecc
 Whole genome sequencing identifies zoonotic transmission of MRSA isolates with the novel meca homologue mecc Ewan M. Harrison, Gavin K. Paterson, Matthew T.G. Holden, Jesper Larsen, Marc Stegger, Anders
Whole genome sequencing identifies zoonotic transmission of MRSA isolates with the novel meca homologue mecc Ewan M. Harrison, Gavin K. Paterson, Matthew T.G. Holden, Jesper Larsen, Marc Stegger, Anders
Non-acidic activation of pain-related Acid-Sensing Ion Channel 3 by lipids
 Manuscript EMBO-2015-92335 Non-acidic activation of pain-related Acid-Sensing Ion Channel 3 by lipids Sébastien Marra, Romain Ferru-Clément, Véronique Breuil, Anne Delaunay, Marine Christin, Valérie Friend,
Manuscript EMBO-2015-92335 Non-acidic activation of pain-related Acid-Sensing Ion Channel 3 by lipids Sébastien Marra, Romain Ferru-Clément, Véronique Breuil, Anne Delaunay, Marine Christin, Valérie Friend,
FGF22 signaling regulates synapse formation during postinjury remodeling of the spinal cord
 Manuscript EMBO-2014-90578 FGF22 signaling regulates synapse formation during postinjury remodeling of the spinal cord Anne Jacobi, Kristina Loy, Anja M Schmalz, Mikael Hellsten, Hisashi Umemori, Martin
Manuscript EMBO-2014-90578 FGF22 signaling regulates synapse formation during postinjury remodeling of the spinal cord Anne Jacobi, Kristina Loy, Anja M Schmalz, Mikael Hellsten, Hisashi Umemori, Martin
Reviewers' comments: Reviewer #1 (Remarks to the Author):
 Reviewers' comments: Reviewer #1 (Remarks to the Author): In this manuscript, Song et al. identified FBXW7 as a new positive regulator for RIG-Itriggered type I IFN signaling pathway. The authors observed
Reviewers' comments: Reviewer #1 (Remarks to the Author): In this manuscript, Song et al. identified FBXW7 as a new positive regulator for RIG-Itriggered type I IFN signaling pathway. The authors observed
Alcoholic hepatitis is a drug-induced disorder
 Alcoholic hepatitis is a drug-induced disorder Gyongyi Szabo, MD, PhD Professor of Medicine University of Massachusetts Medical School Source: 2 Sobernation.com Clinical Progression of ALD Mortality Acute
Alcoholic hepatitis is a drug-induced disorder Gyongyi Szabo, MD, PhD Professor of Medicine University of Massachusetts Medical School Source: 2 Sobernation.com Clinical Progression of ALD Mortality Acute
PKCλ Is Critical in AMPA Receptor Phosphorylation and Synaptic Incorporation during LTP
 Manuscript EMBO-2012-82900 PKCλ Is Critical in AMPA Receptor Phosphorylation and Synaptic Incorporation during LTP Si-Qiang Ren, Jing-Zhi Yan, Xiao-Yan Zhang, Yun-Fei Bu, Wei-Wei Pan, Wen Yao, Tian Tian
Manuscript EMBO-2012-82900 PKCλ Is Critical in AMPA Receptor Phosphorylation and Synaptic Incorporation during LTP Si-Qiang Ren, Jing-Zhi Yan, Xiao-Yan Zhang, Yun-Fei Bu, Wei-Wei Pan, Wen Yao, Tian Tian
Antithetical NFATc1-Sox2 and p53-mir200 signaling networks govern pancreatic cancer cell plasticity
 The EMBO Journal Peer Review Process File - EMBO-2014-89574 Manuscript EMBO-2014-89574 Antithetical NFATc1-Sox2 and p53-mir200 signaling networks govern pancreatic cancer cell plasticity Shiv K. Singh,
The EMBO Journal Peer Review Process File - EMBO-2014-89574 Manuscript EMBO-2014-89574 Antithetical NFATc1-Sox2 and p53-mir200 signaling networks govern pancreatic cancer cell plasticity Shiv K. Singh,
5-HT 6 Receptor Recruitment of mtor as a Mechanism for Perturbed Cognition in Schizophrenia
 Manuscript EMM-2012-01410 5-HT 6 Receptor Recruitment of mtor as a Mechanism for Perturbed Cognition in Schizophrenia Julie Meffre, Séverine Chaumont-Dubel, Clotilde Mannoury la Cour, Florence Loiseau,
Manuscript EMM-2012-01410 5-HT 6 Receptor Recruitment of mtor as a Mechanism for Perturbed Cognition in Schizophrenia Julie Meffre, Séverine Chaumont-Dubel, Clotilde Mannoury la Cour, Florence Loiseau,
REVIEWERS' COMMENTS: Reviewer #1 (Remarks to the Author):
 Editorial Note: this manuscript has been previously reviewed at another journal that is not operating a transparent peer review scheme. This document only contains reviewer comments and rebuttal letters
Editorial Note: this manuscript has been previously reviewed at another journal that is not operating a transparent peer review scheme. This document only contains reviewer comments and rebuttal letters
Nuclear Export of Histone Deacetylase 7 During Thymic Selection is required for Immune Self-tolerance
 The EMBO Journal Peer Review Process File - EMBO-2012-80891 Manuscript EMBO-2012-80891 Nuclear Export of Histone Deacetylase 7 During Thymic Selection is required for Immune Self-tolerance Herbert G Kasler,
The EMBO Journal Peer Review Process File - EMBO-2012-80891 Manuscript EMBO-2012-80891 Nuclear Export of Histone Deacetylase 7 During Thymic Selection is required for Immune Self-tolerance Herbert G Kasler,
CKIP-1 couples Smurf1 ubiquitin ligase with Rpt6 subunit of proteasome to promote substrate degradation
 Manuscript EMBO-2012-36312 CKIP-1 couples Smurf1 ubiquitin ligase with Rpt6 subunit of proteasome to promote substrate degradation Yifang Wang, Jing Nie, Yiwu Wang, Luo Zhang, Guichun Xing, Ping Xie, Fuchu
Manuscript EMBO-2012-36312 CKIP-1 couples Smurf1 ubiquitin ligase with Rpt6 subunit of proteasome to promote substrate degradation Yifang Wang, Jing Nie, Yiwu Wang, Luo Zhang, Guichun Xing, Ping Xie, Fuchu
ROMANIAN ACADEMY INSTITUTE OF CELLULAR BIOLOGY AND PATHOLOGY NICOLAE SIMIONESCU. PhD THESIS Summary
 ROMANIAN ACADEMY INSTITUTE OF CELLULAR BIOLOGY AND PATHOLOGY NICOLAE SIMIONESCU PhD THESIS Summary INVOLVEMENT OF ALARMIN HMGB1 IN INFLAMMATORY PROCESSES ASSOCIATED WITH VASCULAR DYSFUNCTION IN HYPERLIPIDEMIA
ROMANIAN ACADEMY INSTITUTE OF CELLULAR BIOLOGY AND PATHOLOGY NICOLAE SIMIONESCU PhD THESIS Summary INVOLVEMENT OF ALARMIN HMGB1 IN INFLAMMATORY PROCESSES ASSOCIATED WITH VASCULAR DYSFUNCTION IN HYPERLIPIDEMIA
Peer review correspondence
 Highly polarized Th17 cells induce EAE via a T-bet-independent mechanism Heather M. Grifka-Walk, Stephen J. Lalor and Benjamin M. Segal Corresponding author: Benjamin Segal, Holtom-Garrett Program in Neuroimmunology
Highly polarized Th17 cells induce EAE via a T-bet-independent mechanism Heather M. Grifka-Walk, Stephen J. Lalor and Benjamin M. Segal Corresponding author: Benjamin Segal, Holtom-Garrett Program in Neuroimmunology
Tubby and tubby-like protein 1 are new MerTK ligands for phagocytosis
 Manuscript EMBO-2010-75649 Tubby and tubby-like protein 1 are new MerTK ligands for phagocytosis Nora B. Caberoy, Yixiong Zhou and Wei Li Corresponding author: Wei Li, University of Miami School of Medicine
Manuscript EMBO-2010-75649 Tubby and tubby-like protein 1 are new MerTK ligands for phagocytosis Nora B. Caberoy, Yixiong Zhou and Wei Li Corresponding author: Wei Li, University of Miami School of Medicine
1. TLR. TLR Toll-like receptors. Toll Toll-like receptor, TLR TLR TLR TLR. type I TLR TLR. Toll
 54pp.145 152 2004 1. TLR T B TLR Toll-like receptors TLR TLR I IFN TLR T B B T Toll NF- B 1996 565-0871 3-1 TEL 06-6879-8303 FAX 06-6879-8305 E-mail uemattsu@biken.osaka-u.ac.jp Toll Toll-like receptor,
54pp.145 152 2004 1. TLR T B TLR Toll-like receptors TLR TLR I IFN TLR T B B T Toll NF- B 1996 565-0871 3-1 TEL 06-6879-8303 FAX 06-6879-8305 E-mail uemattsu@biken.osaka-u.ac.jp Toll Toll-like receptor,
SOS1 and Ras regulate epithelial tight junction formation in the human airway through EMP1
 Manuscript EMBOR-2014-39218 SOS1 and Ras regulate epithelial tight junction formation in the human airway through EMP1 Joanne Durgan, Guangbo Tao, Matthew S. Walters, Oliver Florey, Anja Schmidt, Vanessa
Manuscript EMBOR-2014-39218 SOS1 and Ras regulate epithelial tight junction formation in the human airway through EMP1 Joanne Durgan, Guangbo Tao, Matthew S. Walters, Oliver Florey, Anja Schmidt, Vanessa
A novel fragile X syndrome mutation reveals a conserved role for the carboxy-terminus in FMRP localization and function
 A novel fragile X syndrome mutation reveals a conserved role for the carboxy-terminus in FMRP localization and function Zeynep Okray, Celine E.F. de Esch, Hilde Van Esch, Koen Devriendt, Annelies Claeys,
A novel fragile X syndrome mutation reveals a conserved role for the carboxy-terminus in FMRP localization and function Zeynep Okray, Celine E.F. de Esch, Hilde Van Esch, Koen Devriendt, Annelies Claeys,
Inhibitors of mitochondrial Kv1.3 channels induce Bax/Bakindependent death of cancer cells
 Manuscript EMM-2011-01039 Inhibitors of mitochondrial Kv1.3 channels induce Bax/Bakindependent death of cancer cells Luigi Leanza, Brian Henry, Nicola Sassi, Mario Zoratti, K. George Chandy, Erich Gulbins
Manuscript EMM-2011-01039 Inhibitors of mitochondrial Kv1.3 channels induce Bax/Bakindependent death of cancer cells Luigi Leanza, Brian Henry, Nicola Sassi, Mario Zoratti, K. George Chandy, Erich Gulbins
Discrete domains of gene expression in germinal layers distinguish the development of gyrencephaly
 Manuscript EMBO-2015-91176 Discrete domains of gene expression in germinal layers distinguish the development of gyrencephaly Camino De Juan Romero, Carl Bruder, Ugo Tomasello, Jose Miguel Sanz-Anquela
Manuscript EMBO-2015-91176 Discrete domains of gene expression in germinal layers distinguish the development of gyrencephaly Camino De Juan Romero, Carl Bruder, Ugo Tomasello, Jose Miguel Sanz-Anquela
Combinations of genetic mutations in the adult neural stem cell compartment determine brain tumour phenotypes
 Manuscript EMBO-2009-71812 Combinations of genetic mutations in the adult neural stem cell compartment determine brain tumour phenotypes Thomas S. Jacques, Alexander Swales, Monika J. Brzozowski, Nico
Manuscript EMBO-2009-71812 Combinations of genetic mutations in the adult neural stem cell compartment determine brain tumour phenotypes Thomas S. Jacques, Alexander Swales, Monika J. Brzozowski, Nico
Supplementary Figure 1. Normal T lymphocyte populations in Dapk -/- mice. (a) Normal thymic development in Dapk -/- mice. Thymocytes from WT and Dapk
 Supplementary Figure 1. Normal T lymphocyte populations in Dapk -/- mice. (a) Normal thymic development in Dapk -/- mice. Thymocytes from WT and Dapk -/- mice were stained for expression of CD4 and CD8.
Supplementary Figure 1. Normal T lymphocyte populations in Dapk -/- mice. (a) Normal thymic development in Dapk -/- mice. Thymocytes from WT and Dapk -/- mice were stained for expression of CD4 and CD8.
The wound inflammatory response exacerbates growth of pre-neoplastic cells and progression to cancer
 The EMBO Journal Peer Review Process File - EMBO-2014-90147 Manuscript EMBO-2014-90147 The wound inflammatory response exacerbates growth of pre-neoplastic cells and progression to cancer Nicole Antonio,
The EMBO Journal Peer Review Process File - EMBO-2014-90147 Manuscript EMBO-2014-90147 The wound inflammatory response exacerbates growth of pre-neoplastic cells and progression to cancer Nicole Antonio,
Follow this and additional works at: Part of the Medicine and Health Sciences Commons
 Washington University School of Medicine Digital Commons@Becker Open Access Publications 2009 Induction of IFN-beta and the innate antiviral response in myeloid cells occurs through an IPS-1-dependent
Washington University School of Medicine Digital Commons@Becker Open Access Publications 2009 Induction of IFN-beta and the innate antiviral response in myeloid cells occurs through an IPS-1-dependent
Phospholipase C γ Prof. Graham Carpenter
 Graham Carpenter, h.d. rofessor of Biochemistry Cornelia Crooke Department of Biochemistry Vanderbilt University School of Medicine, Nashville, TN 1 Receptor Tyrosine Kinases GF Extracellular M Intracellular
Graham Carpenter, h.d. rofessor of Biochemistry Cornelia Crooke Department of Biochemistry Vanderbilt University School of Medicine, Nashville, TN 1 Receptor Tyrosine Kinases GF Extracellular M Intracellular
Innate Immunity. Chapter 3. Connection Between Innate and Adaptive Immunity. Know Differences and Provide Examples. Antimicrobial peptide psoriasin
 Chapter Know Differences and Provide Examples Innate Immunity kin and Epithelial Barriers Antimicrobial peptide psoriasin -Activity against Gram (-) E. coli Connection Between Innate and Adaptive Immunity
Chapter Know Differences and Provide Examples Innate Immunity kin and Epithelial Barriers Antimicrobial peptide psoriasin -Activity against Gram (-) E. coli Connection Between Innate and Adaptive Immunity
Reviewers' comments: Reviewer #1 (Remarks to the Author):
 Reviewers' comments: Reviewer #1 (Remarks to the Author): The manuscript by Wu et al describes critical role of RNA binding protein CUGBP1 in the development of TGF-beta-mediated liver fibrosis. The activation
Reviewers' comments: Reviewer #1 (Remarks to the Author): The manuscript by Wu et al describes critical role of RNA binding protein CUGBP1 in the development of TGF-beta-mediated liver fibrosis. The activation
Supplementary Figure 1. PD-L1 is glycosylated in cancer cells. (a) Western blot analysis of PD-L1 in breast cancer cells. (b) Western blot analysis
 Supplementary Figure 1. PD-L1 is glycosylated in cancer cells. (a) Western blot analysis of PD-L1 in breast cancer cells. (b) Western blot analysis of PD-L1 in ovarian cancer cells. (c) Western blot analysis
Supplementary Figure 1. PD-L1 is glycosylated in cancer cells. (a) Western blot analysis of PD-L1 in breast cancer cells. (b) Western blot analysis of PD-L1 in ovarian cancer cells. (c) Western blot analysis
Controlled induction of human pancreatic progenitors produces functional beta-like cells in vitro.
 Manuscript EMBO-2015-91058 Controlled induction of human pancreatic progenitors produces functional beta-like cells in vitro. Holger A Russ, Audrey V Parent, Jennifer J Ringler, Thomas G Hennings, Gopika
Manuscript EMBO-2015-91058 Controlled induction of human pancreatic progenitors produces functional beta-like cells in vitro. Holger A Russ, Audrey V Parent, Jennifer J Ringler, Thomas G Hennings, Gopika
GW182 proteins cause PABP dissociation from silenced mirna targets in the absence of deadenylation
 Manuscript EMBO-2012-83275 GW182 proteins cause PABP dissociation from silenced mirna targets in the absence of deadenylation Latifa Zekri, Duygu Kuzuoğlu-Öztürk and Elisa Izaurralde Corresponding author:
Manuscript EMBO-2012-83275 GW182 proteins cause PABP dissociation from silenced mirna targets in the absence of deadenylation Latifa Zekri, Duygu Kuzuoğlu-Öztürk and Elisa Izaurralde Corresponding author:
Somatically Mutated ABL1 is an Actionable and Essential NSCLC Survival Gene
 Somatically Mutated ABL1 is an Actionable and Essential NSCLC Survival Gene Ewelina Testoni, Natalie L. Stephenson, Pedro Torres-Ayuso, Anna A. Marusiak, Eleanor W. Trotter, Andrew Hudson, Cassandra L.
Somatically Mutated ABL1 is an Actionable and Essential NSCLC Survival Gene Ewelina Testoni, Natalie L. Stephenson, Pedro Torres-Ayuso, Anna A. Marusiak, Eleanor W. Trotter, Andrew Hudson, Cassandra L.
Supplementary Information
 Supplementary Information mediates STAT3 activation at retromer-positive structures to promote colitis and colitis-associated carcinogenesis Zhang et al. a b d e g h Rel. Luc. Act. Rel. mrna Rel. mrna
Supplementary Information mediates STAT3 activation at retromer-positive structures to promote colitis and colitis-associated carcinogenesis Zhang et al. a b d e g h Rel. Luc. Act. Rel. mrna Rel. mrna
Anchored Phosphatases Modulate Glucose Homeostasis
 Manuscript EMBO-2012-82507 Anchored Phosphatases Modulate Glucose Homeostasis Simon Hinke, Manuel Navedo, Allison Ulman, Jennifer Whiting, Patrick Nygren, Geng Tian, Antonio Jimenez-Caliani, Lorene Langeberg,
Manuscript EMBO-2012-82507 Anchored Phosphatases Modulate Glucose Homeostasis Simon Hinke, Manuel Navedo, Allison Ulman, Jennifer Whiting, Patrick Nygren, Geng Tian, Antonio Jimenez-Caliani, Lorene Langeberg,
Innate immunity. Abul K. Abbas University of California San Francisco. FOCiS
 1 Innate immunity Abul K. Abbas University of California San Francisco FOCiS 2 Lecture outline Components of innate immunity Recognition of microbes and dead cells Toll Like Receptors NOD Like Receptors/Inflammasome
1 Innate immunity Abul K. Abbas University of California San Francisco FOCiS 2 Lecture outline Components of innate immunity Recognition of microbes and dead cells Toll Like Receptors NOD Like Receptors/Inflammasome
Innate immune regulation of T-helper (Th) cell homeostasis in the intestine
 Innate immune regulation of T-helper (Th) cell homeostasis in the intestine Masayuki Fukata, MD, Ph.D. Research Scientist II Division of Gastroenterology, Department of Medicine, F. Widjaja Foundation,
Innate immune regulation of T-helper (Th) cell homeostasis in the intestine Masayuki Fukata, MD, Ph.D. Research Scientist II Division of Gastroenterology, Department of Medicine, F. Widjaja Foundation,
Title: Oral administration of PPC enhances antigen-specific CD8+ T cell responses while reducing IgE levels in sensitized mice.
 Author's response to reviews Title: Oral administration of PPC enhances antigen-specific CD8+ T cell responses while reducing IgE levels in sensitized mice. Authors: Mike Burrows (mburrows@tampabayresearch.org)
Author's response to reviews Title: Oral administration of PPC enhances antigen-specific CD8+ T cell responses while reducing IgE levels in sensitized mice. Authors: Mike Burrows (mburrows@tampabayresearch.org)
PKR is required for macrophage apoptosis after activation of TLR-4. By Christina, Ania and Xiaofu
 PKR is required for macrophage apoptosis after activation of TLR-4 By Christina, Ania and Xiaofu Introduction Macrophage PAMP Bacteria LPS Gram -ve LTA Gram +ve activation Clearance Macrophage PAMP Bacteria
PKR is required for macrophage apoptosis after activation of TLR-4 By Christina, Ania and Xiaofu Introduction Macrophage PAMP Bacteria LPS Gram -ve LTA Gram +ve activation Clearance Macrophage PAMP Bacteria
Intracellular MHC class II molecules promote TLR-triggered innate immune responses by maintaining activation of the kinase Btk
 Intracellular MHC class II molecules promote TLR-triggered innate immune responses by maintaining activation of the kinase Btk Xingguang Liu 1, Zhenzhen Zhan 1, Dong Li, Li Xu 1, Feng Ma, Peng Zhang 1,
Intracellular MHC class II molecules promote TLR-triggered innate immune responses by maintaining activation of the kinase Btk Xingguang Liu 1, Zhenzhen Zhan 1, Dong Li, Li Xu 1, Feng Ma, Peng Zhang 1,
Minor point: In Table S1 there seems to be a typo - multiplicity of the data for the Sm3+ structure 134.8?
 PEER REVIEW FILE Reviewers' comments: Reviewer #1 (Remarks to the Author): Finogenova and colleagues investigated aspects of the structural organization of the Nuclear Exosome Targeting (NEXT) complex,
PEER REVIEW FILE Reviewers' comments: Reviewer #1 (Remarks to the Author): Finogenova and colleagues investigated aspects of the structural organization of the Nuclear Exosome Targeting (NEXT) complex,
Involvement of NMDAR2A tyrosine phosphorylation in depression-related behavior
 Manuscript EMBO-2009-71034 Involvement of NMDAR2A tyrosine phosphorylation in depression-related behavior Sachiko Taniguchi, Asami Tanimura, Yuji Kiyama, Tohru Tezuka, Ayako M. Watabe, Norikazu Katayama,
Manuscript EMBO-2009-71034 Involvement of NMDAR2A tyrosine phosphorylation in depression-related behavior Sachiko Taniguchi, Asami Tanimura, Yuji Kiyama, Tohru Tezuka, Ayako M. Watabe, Norikazu Katayama,
TLR9 protects non-immune cells from stress by modulating mitochondrial ATP synthesis through the inhibition of SERCA2
 Manuscript EMBOR-2013-37945 TLR9 protects non-immune cells from stress by modulating mitochondrial ATP synthesis through the inhibition of SERCA2 Yasunori Shintani, Hannes C.A. Drexler, Hidetaka Kioka,
Manuscript EMBOR-2013-37945 TLR9 protects non-immune cells from stress by modulating mitochondrial ATP synthesis through the inhibition of SERCA2 Yasunori Shintani, Hannes C.A. Drexler, Hidetaka Kioka,
Title:Inhibitory effects of astaxanthin on azoxymethane-induced colonic preneoplastic lesions in C57/BL/KsJ-db/db mice
 Author's response to reviews Title:Inhibitory effects of astaxanthin on azoxymethane-induced colonic preneoplastic lesions in C57/BL/KsJ-db/db mice Authors: Takahiro Kochi (kottii924@yahoo.co.jp) Masahito
Author's response to reviews Title:Inhibitory effects of astaxanthin on azoxymethane-induced colonic preneoplastic lesions in C57/BL/KsJ-db/db mice Authors: Takahiro Kochi (kottii924@yahoo.co.jp) Masahito
Supplementary Figure 1.TRIM33 binds β-catenin in the nucleus. a & b, Co-IP of endogenous TRIM33 with β-catenin in HT-29 cells (a) and HEK 293T cells
 Supplementary Figure 1.TRIM33 binds β-catenin in the nucleus. a & b, Co-IP of endogenous TRIM33 with β-catenin in HT-29 cells (a) and HEK 293T cells (b). TRIM33 was immunoprecipitated, and the amount of
Supplementary Figure 1.TRIM33 binds β-catenin in the nucleus. a & b, Co-IP of endogenous TRIM33 with β-catenin in HT-29 cells (a) and HEK 293T cells (b). TRIM33 was immunoprecipitated, and the amount of
Splicing Factor HnRNP H Drives an Oncogenic Splicing Switch in Gliomas
 Manuscript EMBO-2011-77697 Splicing Factor HnRNP H Drives an Oncogenic Splicing Switch in Gliomas Clare LeFave, Massimo Squatrito, Sandra Vorlova, Gina Rocco, Cameron Brennan, Eric Holland, Ying-Xian Pan,
Manuscript EMBO-2011-77697 Splicing Factor HnRNP H Drives an Oncogenic Splicing Switch in Gliomas Clare LeFave, Massimo Squatrito, Sandra Vorlova, Gina Rocco, Cameron Brennan, Eric Holland, Ying-Xian Pan,
William C. Comb, Jessica E. Hutti, Patricia Cogswell, Lewis C. Cantley, and Albert S. Baldwin
 Molecular Cell, Volume 45 Supplemental Information p85 SH2 Domain Phosphorylation by IKK Promotes Feedback Inhibition of PI3K and Akt in Response to Cellular Starvation William C. Comb, Jessica E. Hutti,
Molecular Cell, Volume 45 Supplemental Information p85 SH2 Domain Phosphorylation by IKK Promotes Feedback Inhibition of PI3K and Akt in Response to Cellular Starvation William C. Comb, Jessica E. Hutti,
Integrin CD11b negatively regulates TLR-triggered inflammatory responses by. activating Syk and promoting MyD88 and TRIF degradation via cbl-b
 Integrin CD11b negatively regulates TLR-triggered inflammatory responses by activating Syk and promoting MyD88 and TRIF degradation via cbl-b Chaofeng Han, Jing Jin, Sheng Xu, Haibo Liu, Nan Li, and Xuetao
Integrin CD11b negatively regulates TLR-triggered inflammatory responses by activating Syk and promoting MyD88 and TRIF degradation via cbl-b Chaofeng Han, Jing Jin, Sheng Xu, Haibo Liu, Nan Li, and Xuetao
Innate Immunity & Inflammation
 Innate Immunity & Inflammation The innate immune system is an evolutionally conserved mechanism that provides an early and effective response against invading microbial pathogens. It relies on a limited
Innate Immunity & Inflammation The innate immune system is an evolutionally conserved mechanism that provides an early and effective response against invading microbial pathogens. It relies on a limited
Newly Recognized Components of the Innate Immune System
 Newly Recognized Components of the Innate Immune System NOD Proteins: Intracellular Peptidoglycan Sensors NOD-1 NOD-2 Nod Protein LRR; Ligand Recognition CARD RICK I-κB p50 p65 NF-κB Polymorphisms in Nod-2
Newly Recognized Components of the Innate Immune System NOD Proteins: Intracellular Peptidoglycan Sensors NOD-1 NOD-2 Nod Protein LRR; Ligand Recognition CARD RICK I-κB p50 p65 NF-κB Polymorphisms in Nod-2
Better tools. Better research!
 TLR Signaling Pathways in Immunity & Beyond October 17, 2012 Better tools. Better research! www.novusbio.com TLR Signaling in Immunity & Beyond The Big Question: How to tease apart complex TLR pathways?
TLR Signaling Pathways in Immunity & Beyond October 17, 2012 Better tools. Better research! www.novusbio.com TLR Signaling in Immunity & Beyond The Big Question: How to tease apart complex TLR pathways?
Supplementary Figure 1
 Supplementary Figure 1 YAP negatively regulates IFN- signaling. (a) Immunoblot analysis of Yap knockdown efficiency with sh-yap (#1 to #4 independent constructs) in Raw264.7 cells. (b) IFN- -Luc and PRDs
Supplementary Figure 1 YAP negatively regulates IFN- signaling. (a) Immunoblot analysis of Yap knockdown efficiency with sh-yap (#1 to #4 independent constructs) in Raw264.7 cells. (b) IFN- -Luc and PRDs
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION doi:1.138/nature9814 a A SHARPIN FL B SHARPIN ΔNZF C SHARPIN T38L, F39V b His-SHARPIN FL -1xUb -2xUb -4xUb α-his c Linear 4xUb -SHARPIN FL -SHARPIN TF_LV -SHARPINΔNZF -SHARPIN
SUPPLEMENTARY INFORMATION doi:1.138/nature9814 a A SHARPIN FL B SHARPIN ΔNZF C SHARPIN T38L, F39V b His-SHARPIN FL -1xUb -2xUb -4xUb α-his c Linear 4xUb -SHARPIN FL -SHARPIN TF_LV -SHARPINΔNZF -SHARPIN
A genome-scale screen reveals context-dependent ovarian cancer sensitivity to mirna overexpression
 A genome-scale screen reveals context-dependent ovarian cancer sensitivity to mirna overexpression Benjamin B. Shields, Chad V. Pecot, Hua Gao, Elizabeth McMillan, Malia Potts, Christa Nagel, Scott Purinton,
A genome-scale screen reveals context-dependent ovarian cancer sensitivity to mirna overexpression Benjamin B. Shields, Chad V. Pecot, Hua Gao, Elizabeth McMillan, Malia Potts, Christa Nagel, Scott Purinton,
Type of file: PDF Title of file for HTML: Supplementary Information Description: Supplementary Figures
 Type of file: PDF Title of file for HTML: Supplementary Information Description: Supplementary Figures Type of file: MOV Title of file for HTML: Supplementary Movie 1 Description: NLRP3 is moving along
Type of file: PDF Title of file for HTML: Supplementary Information Description: Supplementary Figures Type of file: MOV Title of file for HTML: Supplementary Movie 1 Description: NLRP3 is moving along
Supplementary Materials for
 www.sciencesignaling.org/cgi/content/full/4/199/ra75/dc1 Supplementary Materials for Signaling by the Matrix Proteoglycan Decorin Controls Inflammation and Cancer Through PDCD4 and MicroRNA-21 Rosetta
www.sciencesignaling.org/cgi/content/full/4/199/ra75/dc1 Supplementary Materials for Signaling by the Matrix Proteoglycan Decorin Controls Inflammation and Cancer Through PDCD4 and MicroRNA-21 Rosetta
Reviewers' comments: Reviewer #1 (Remarks to the Author):
 Reviewers' comments: Reviewer #1 (Remarks to the Author): The authors have presented data demonstrating activation of AKT as a common resistance mechanism in EGFR mutation positive, EGFR TKI resistant
Reviewers' comments: Reviewer #1 (Remarks to the Author): The authors have presented data demonstrating activation of AKT as a common resistance mechanism in EGFR mutation positive, EGFR TKI resistant
Bidirectional integrative regulation of Cav1.2 calcium channel by microrna mir-103: role in pain.
 Manuscript EMBO-2011-77953 Bidirectional integrative regulation of Cav1.2 calcium channel by microrna mir-103: role in pain. Alexandre Favereaux, Olivier Thoumine, Rabia Bouali-Benazzouz, Virginie Roques,
Manuscript EMBO-2011-77953 Bidirectional integrative regulation of Cav1.2 calcium channel by microrna mir-103: role in pain. Alexandre Favereaux, Olivier Thoumine, Rabia Bouali-Benazzouz, Virginie Roques,
TRIM5 requires Ube2W to anchor Lys63-linked ubiquitin chains and restrict reverse transcription
 Manuscript EMBO-2014-90361 TRIM5 requires Ube2W to anchor Lys63-linked ubiquitin chains and restrict reverse transcription Adam J Fletcher, Devin E Christensen, Chad Nelson, Choon Ping Tan, Torsten Schaller,
Manuscript EMBO-2014-90361 TRIM5 requires Ube2W to anchor Lys63-linked ubiquitin chains and restrict reverse transcription Adam J Fletcher, Devin E Christensen, Chad Nelson, Choon Ping Tan, Torsten Schaller,
Title: Cytosolic DNA-mediated, STING-dependent pro-inflammatory gene. Fig. S1. STING ligands-mediated signaling response in MEFs. (A) Primary MEFs (1
 1 Supporting Information 2 3 4 Title: Cytosolic DNA-mediated, STING-dependent pro-inflammatory gene induction necessitates canonical NF-κB activation through TBK1 5 6 Authors: Abe et al. 7 8 9 Supporting
1 Supporting Information 2 3 4 Title: Cytosolic DNA-mediated, STING-dependent pro-inflammatory gene induction necessitates canonical NF-κB activation through TBK1 5 6 Authors: Abe et al. 7 8 9 Supporting
Stewart et al. CD36 ligands promote sterile inflammation through assembly of a TLR 4 and 6 heterodimer
 NFκB (fold induction) Stewart et al. ligands promote sterile inflammation through assembly of a TLR 4 and 6 heterodimer a. mrna (fold induction) 5 4 3 2 1 LDL oxldl Gro1a MIP-2 RANTES mrna (fold induction)
NFκB (fold induction) Stewart et al. ligands promote sterile inflammation through assembly of a TLR 4 and 6 heterodimer a. mrna (fold induction) 5 4 3 2 1 LDL oxldl Gro1a MIP-2 RANTES mrna (fold induction)
Supplementary information to: Mechanism of lipopolysaccharide-induced skin edema formation in the mouse
 Supplementary information to: Mechanism of lipopolysaccharide-induced skin edema formation in the mouse Ivan Zanoni*, Renato Ostuni*, Simona Barresi, Marco Di Gioia, Achille Broggi, Barbara Costa, Roberta
Supplementary information to: Mechanism of lipopolysaccharide-induced skin edema formation in the mouse Ivan Zanoni*, Renato Ostuni*, Simona Barresi, Marco Di Gioia, Achille Broggi, Barbara Costa, Roberta
SUPPLEMENTARY INFORMATION
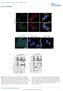 DOI:.38/ncb2822 a MTC02 FAO cells EEA1 b +/+ MEFs /DAPI -/- MEFs /DAPI -/- MEFs //DAPI c HEK 293 cells WCE N M C P AKT TBC1D7 Lamin A/C EEA1 VDAC d HeLa cells WCE N M C P AKT Lamin A/C EEA1 VDAC Figure
DOI:.38/ncb2822 a MTC02 FAO cells EEA1 b +/+ MEFs /DAPI -/- MEFs /DAPI -/- MEFs //DAPI c HEK 293 cells WCE N M C P AKT TBC1D7 Lamin A/C EEA1 VDAC d HeLa cells WCE N M C P AKT Lamin A/C EEA1 VDAC Figure
ACBD3-mediated recruitment of PI4KB to picornavirus RNA replication sites
 Manuscript EMBO-2011-78279 ACBD3-mediated recruitment of PI4KB to picornavirus RNA replication sites Jun Sasaki, Kumiko Ishikawa, Minetaro Arita and Koki Taniguchi Corresponding author: Dr. Jun Sasaki,
Manuscript EMBO-2011-78279 ACBD3-mediated recruitment of PI4KB to picornavirus RNA replication sites Jun Sasaki, Kumiko Ishikawa, Minetaro Arita and Koki Taniguchi Corresponding author: Dr. Jun Sasaki,
Supplemental Information. Cellular Decision Making. by Non-Integrative Processing of TLR Inputs
 Cell Reports, Volume 19 Supplemental Information Cellular Decision Making by Non-Integrative Processing of TLR Inputs Ryan A. Kellogg, Chengzhe Tian, Martin Etzrodt, and Savasx Tay 1. Classifier Description
Cell Reports, Volume 19 Supplemental Information Cellular Decision Making by Non-Integrative Processing of TLR Inputs Ryan A. Kellogg, Chengzhe Tian, Martin Etzrodt, and Savasx Tay 1. Classifier Description
RIP1 Negatively Regulates Basal Autophagic Flux through TFEB to Control Sensitivity to Apoptosis
 Manuscript EMBOR-2014-39496 RIP1 Negatively Regulates Basal Autophagic Flux through TFEB to Control Sensitivity to Apoptosis Tohru Yonekawa, Graciela Gamez, Jihye Kim, Aik Choon Tan, Jackie Thorburn, Jacob
Manuscript EMBOR-2014-39496 RIP1 Negatively Regulates Basal Autophagic Flux through TFEB to Control Sensitivity to Apoptosis Tohru Yonekawa, Graciela Gamez, Jihye Kim, Aik Choon Tan, Jackie Thorburn, Jacob
NOD1 contributes to mouse host defense against Helicobacter pylori via induction of type I IFN and activation of the ISGF3 signaling pathway
 Research article NOD1 contributes to mouse host defense against Helicobacter pylori via induction of type I IFN and activation of the ISGF3 signaling pathway Tomohiro Watanabe, 1,2 Naoki Asano, 1 Stefan
Research article NOD1 contributes to mouse host defense against Helicobacter pylori via induction of type I IFN and activation of the ISGF3 signaling pathway Tomohiro Watanabe, 1,2 Naoki Asano, 1 Stefan
Reviewers' comments: Reviewer #1 (Remarks to the Author):
 Reviewers' comments: Reviewer #1 (Remarks to the Author): In this manuscript, Hasan et al analyse the transcriptional program used by pathogenic Th17 cells raised in the presence of IL-23 as compared to
Reviewers' comments: Reviewer #1 (Remarks to the Author): In this manuscript, Hasan et al analyse the transcriptional program used by pathogenic Th17 cells raised in the presence of IL-23 as compared to
SUPPLEMENTARY INFORMATION
 doi: 1.138/nature89 IFN- (ng ml ) 5 4 3 1 Splenocytes NS IFN- (ng ml ) 6 4 Lymph node cells NS Nfkbiz / Nfkbiz / Nfkbiz / Nfkbiz / IL- (ng ml ) 3 1 Splenocytes IL- (ng ml ) 1 8 6 4 *** ** Lymph node cells
doi: 1.138/nature89 IFN- (ng ml ) 5 4 3 1 Splenocytes NS IFN- (ng ml ) 6 4 Lymph node cells NS Nfkbiz / Nfkbiz / Nfkbiz / Nfkbiz / IL- (ng ml ) 3 1 Splenocytes IL- (ng ml ) 1 8 6 4 *** ** Lymph node cells
SUPPLEMENTARY FIGURES
 SUPPLEMENTARY FIGURES Supplementary Figure 1. (A) Left, western blot analysis of ISGylated proteins in Jurkat T cells treated with 1000U ml -1 IFN for 16h (IFN) or left untreated (CONT); right, western
SUPPLEMENTARY FIGURES Supplementary Figure 1. (A) Left, western blot analysis of ISGylated proteins in Jurkat T cells treated with 1000U ml -1 IFN for 16h (IFN) or left untreated (CONT); right, western
Quantification of early stage lesions for loss of p53 should be shown in the main figures.
 Reviewer #1 (Remarks to the Author): Expert in prostate cancer The manuscript "Clonal dynamics following p53 loss of heterozygosity in Kras-driven cancers" uses a number of novel genetically engineered
Reviewer #1 (Remarks to the Author): Expert in prostate cancer The manuscript "Clonal dynamics following p53 loss of heterozygosity in Kras-driven cancers" uses a number of novel genetically engineered
TOLL-LIKE RECEPTORS AND CYTOKINES IN SEPSIS
 TOLL-LIKE RECEPTORS AND CYTOKINES IN SEPSIS A/PROF WILLIAM SEWELL ST VINCENT S CLINICAL SCHOOL, UNSW SYDPATH, ST VINCENT S HOSPITAL SYDNEY GARVAN INSTITUTE INNATE VERSUS ADAPTIVE IMMUNE RESPONSES INNATE
TOLL-LIKE RECEPTORS AND CYTOKINES IN SEPSIS A/PROF WILLIAM SEWELL ST VINCENT S CLINICAL SCHOOL, UNSW SYDPATH, ST VINCENT S HOSPITAL SYDNEY GARVAN INSTITUTE INNATE VERSUS ADAPTIVE IMMUNE RESPONSES INNATE
Supplementary Figures for TSC1 controls macrophage polarization to prevent inflammatory disorder by Linnan Zhu et al
 Supplementary Figures for TSC1 controls macrophage polarization to prevent inflammatory disorder by Linnan Zhu et al Suppl. Fig. 1 Tissue DN C Proteins kd TSC1-17 TSC 1 loxp bp -48-285 ctin PEMs Neutrophils
Supplementary Figures for TSC1 controls macrophage polarization to prevent inflammatory disorder by Linnan Zhu et al Suppl. Fig. 1 Tissue DN C Proteins kd TSC1-17 TSC 1 loxp bp -48-285 ctin PEMs Neutrophils
Cover Letter. Reviewer 1:
 Cover Letter Michael Yang, M.D., Ph.D. Managing Editor of Cancer Research Frontiers 1188 Willis Ave, #109, Albertson, NY 11507, USA Phone: +1-917-426-1571 http://cancer-research-frontiers.org/ Dear Dr.
Cover Letter Michael Yang, M.D., Ph.D. Managing Editor of Cancer Research Frontiers 1188 Willis Ave, #109, Albertson, NY 11507, USA Phone: +1-917-426-1571 http://cancer-research-frontiers.org/ Dear Dr.
Figure S1. Western blot analysis of clathrin RNA interference in human DCs Human immature DCs were transfected with 100 nm Clathrin SMARTpool or
 Figure S1. Western blot analysis of clathrin RNA interference in human DCs Human immature DCs were transfected with 100 nm Clathrin SMARTpool or control nontargeting sirnas. At 90 hr after transfection,
Figure S1. Western blot analysis of clathrin RNA interference in human DCs Human immature DCs were transfected with 100 nm Clathrin SMARTpool or control nontargeting sirnas. At 90 hr after transfection,
Supplementary Fig. 1 eif6 +/- mice show a reduction in white adipose tissue, blood lipids and normal glycogen synthesis. The cohort of the original
 Supplementary Fig. 1 eif6 +/- mice show a reduction in white adipose tissue, blood lipids and normal glycogen synthesis. The cohort of the original phenotypic screening was n=40. For specific tests, the
Supplementary Fig. 1 eif6 +/- mice show a reduction in white adipose tissue, blood lipids and normal glycogen synthesis. The cohort of the original phenotypic screening was n=40. For specific tests, the
