Essential Requirement for c-kit and Common Chain in Thymocyte Development Cannot be Overruled by Enforced Expression of Bcl-2
|
|
|
- Marjorie Franklin
- 5 years ago
- Views:
Transcription
1 Published Online: 18 June, 2001 Supp Info: Downloaded from jem.rupress.org on October 9, 2018 Brief Definitive Report Essential Requirement for c-kit and Common Chain in Thymocyte Development Cannot be Overruled by Enforced Expression of Bcl-2 By Hans-Reimer Rodewald,* Claudia Waskow,* and Corinne Haller From the *Department for Immunology, University of Ulm, D Ulm, Germany; and the Basel Institute for Immunology, CH-4005 Basel, Switzerland Abstract The thymus in mice lacking both the receptor tyrosine kinase c-kit and the common cytokine receptor chain ( c ) is alymphoid because these receptors provide essential signals at the earliest stages of thymocyte development. The signals transduced by these receptors potentially regulate proliferation, survival, or differentiation, but the contribution of each receptor to distinct intracellular signaling cascades is only poorly defined. Here, we have examined whether enforced expression of Bcl-2 can rescue thymocyte development in c-kit and c single or double mutant mice. A bcl-2 transgene (E-bcl-2-25; expressed in the T cell lineage) was introduced into (a) c-kit and c wild-type (c-kit c bcl ), (b) c-kit deficient (c-kit c bcl ), (c) c -deficient (c-kit c bcl ), or (d) c-kit and c double-deficient mice (c-kit c bcl ). The bcl-2 transgene was functionally active in wild-type and c-kit or c single mutants, as it promoted survival of ex vivo isolated thymocytes, including pro-t cells. In vivo, however, transgenic Bcl-2 did not release T cell precursors from their phenotypic block and failed to increase progenitor or total thymocyte cellularity in c-kit or c single or double mutants. These data argue strongly against a role for Bcl-2 as a key mediator in signaling pathways linked to cytokine and growth factor receptors driving early thymocyte development. Key words: growth factors c-kit common cytokine receptor chain bcl-2 T cell development Introduction Prothymocytes colonizing the thymus are very rare cells. This small pool of thymic immigrants undergoes in the order of 100 fold (unpublished data) expansion before rearrangement of the TCR chain. This expansion is mediated by two key growth/cytokine receptors expressed on prothymocytes, the tyrosine kinase c-kit and common cytokine receptor chain ( c ) IL-7R chain complexes. The ligands binding to these receptors, stem cell factor (SCF) and IL-7, are provided by the epithelial thymic stroma (for reviews, see references 1 3). In mutants lacking c-kit and c together, but not in single c-kit or c - deficient mice, the thymus is alymphoid, and TCR-, -, and - rearrangements are essentially undetectable (4, 5). Thus, c-kit and c provide essential, synergistic signals in pro-t cells (for a review, see reference 2). Synergy between growth factor and cytokine signaling is a common theme in many hematopoietic cells, including stem cells Address correspondence to H.R. Rodewald, Department for Immunology, University of Ulm, Albert-Einstein-Allee 11, D Ulm, Germany. Phone: ; Fax: ; hans-reimer.rodewald@medizin.uni-ulm.de (see, for example, reference 6). Due to the complete lack of thymocytes, c-kit and c double mutant mice may provide a useful model in which individual signaling pathways can be selectively rescued in vivo. At present, the downstream signaling pathways of each receptor are, at least in primary thymocytes, only poorly defined. Expression of the prototypic antiapoptotic protein Bcl-2 is developmentally regulated in thymocytes. Bcl-2 expression is high in immature CD3 CD4 CD8 (triple negative [TN]) and mature CD3 CD4 CD8 or CD3 CD4 CD8 (single positive [SP]), and is downmodulated in intermediate CD3 low CD4 CD8 (double positive [DP]) thymocytes (7, 8). Bcl-2 expression is reduced in thymocytes from mice lacking IL-7R (9), c (8), or IL-7 (10), and is upregulated after exposure of thymocytes from IL-7 deficient mice to IL-7 in vitro (1, 10). Hence, signaling via IL-7R/ c can upregulate Bcl-2 expression in thymocytes. Thymocyte development in bcl-2 deficient mice is normal until early adult life (4 wk of age) followed by a strongly increased sensitivity to apoptotic stimuli affecting, among other cell types, all stages of thymocytes and peripheral lymphocytes (11 13). Similarly, fetal liver derived T cell 1431 J. Exp. Med. The Rockefeller University Press /2001/06/1431/07 $5.00 Volume 193, Number 12, June 18,
2 development is more Bcl-2 independent than bone marrow derived T cell development (13). Collectively, Bcl-2 can act as an antiapoptotic factor in thymocytes, in particular during adult life. As early T cell development is cytokine/growth factor dependent, it is of interest whether growth factor withdrawal induced thymocyte death can be prevented by Bcl-2 expression. There are conflicting data on the role of Bcl-2 for the rescue of T cell development is the absence of IL-7R/ c signaling. Transgene-driven overexpression of Bcl-2 was reported to rescue T cell development in IL-7R chain (9, 14) or c (8, 15) mutant mice (for a review, see reference 1). In contrast, other groups failed to observe a Bcl-2 mediated rescue of c -deficient thymocytes (16, 17; for a review, see reference 2). SCF stimulation can provide antiapoptotic signals in hematopoietic cells. This survival signal may be relayed via activation of phosphatidylinositol 3 (PI-3) kinase and Akt kinase (for a review, see reference 18), but c-kit induced survival may also involve Bcl-2 expression (19). Given that c-kit c mice are completely devoid of thymocytes, we reasoned that these mutants would be a very sensitive model to reveal potential, even minor, effects of Bcl-2 downstream of c-kit and c. In this paper, we show that the bcl-2 transgene was functionally active, and efficiently promoted survival of thymocytes, including the earliest T cell progenitors ex vivo. In vivo, however, transgenic bcl-2 expression failed to release T cell progenitors, both in terms of phenotype and cell numbers, from their developmental block in all mutants tested. As our data, using postnatal day 5 old mutant mice, did not confirm other reports analyzing adult c -deficient bcl-2 transgenic mice (1, 8, 15), we extended our studies to large numbers of adult c -deficient mice with or without the bcl-2 transgene. Notably, thymus cell numbers in adult c mutants without the bcl-2 transgene showed considerable heterogeneity (ranging over two logs), an observation pointing at a possible role for modifier genes in the genetic background of the c mutants. In line with our analysis of neonatal c mutants, enforced expression of Bcl-2 also failed to rescue T cell development in adult c -deficient mice. Our study, therefore, challenges the current view that enforced expression of Bcl-2 can rescue defects in thymocyte development caused by lack of c, and suggests that gene products other than Bcl-2 might modulate signaling pathways linked to cytokine (IL-7) and growth factor (SCF) receptors in early thymocyte development in vivo. Materials and Methods Mice. WB-kit W/ (20) (SLC), c (21), and E-bcl-2-25 (22) (The Jackson Laboratory) were maintained under specific pathogen free-conditions. C-kit and c single and double mutant mice were bred as described (4), and crossed to E-bcl-2-25 mice. Experiments involving c-kit mutant mice were performed on postnatal day 5 old mice, as white spotting locus (W/W) mice die within 10 d after birth (23). W mutant mice were phenotyped by white skin color. Experiments on c mutants were done either on postnatal day 5, or on adult 6 12-wk-old mice. c mutant mice were genotyped by PCR on genomic DNA as described (4). E-bcl-2-25 mice (22) were genotyped by PCR on genomic DNA using oligonucleotides amplifying the human bcl-2 gene (5 oligo: 5-GGTCATGTGTGTGGAGAGCGTCA-3) linked to the SV40 polya site (3 oligo: 5-GTTTCAGGTTCAGGG- GGAGGT-3) yielding a diagnostic DNA fragment of 1 kb. In addition, in some experiments, thymocytes were typed by Western blotting using anti human Bcl-2 specific antibodies (Santa Cruz Biotechnology, Inc.). Monoclonal Antibodies and Flow Cytometry. The following monoclonal antibodies were used for flow cytometric analyses: Anti- lineage antibodies were 2C11 (CD3), H (CD4), (CD8) (both from GIBCO BRL), M1/70.15 (Mac-1; Caltag), Gr-1 (granulocytes), TER119 (red blood cells), DX-5 (NK lineage), B220 (B cells; all PE-labeled), biotinylated ACK-4 (c-kit), FITC-labeled 3C7 (CD25), Cy-Chrome labeled Pgp-1 (CD44; all from BD PharMingen, unless otherwise indicated); streptavidin-allophycocyanin (APC) (Molecular Probes) was used as second step reagent. For FACS analysis, thymocytes were stained with monoclonal antibodies at 5 10 g/ml in PBS/5% FCS for min on ice and washed once in PBS/5% FCS. Cell sorting and FACS analysis were done on FACStar and FACSCalibur, respectively (Becton Dickinson). Fluorescence data are displayed as dot plots using CELLQuest software (Becton Dickinson). Ex Vivo Survival Assay. Total or cell sorter purified thymocyte subpopulations were placed in normal cell culture medium (DMEM plus 10% FCS plus 1 mm pyruvate plus 2 mm glutamine plus M -mercaptoethanol) and cultured without additional growth factors. At time points indicated in Fig. 1, absolute numbers (or percentage of the starting population) of viable cells were determined by cell counting and trypan blue exclusion in a Neubauer counting chamber. Results and Discussion The E-bcl-2-25 Transgene Promotes Survival of c-kit and c Mutant Thymocytes Ex Vivo. To analyze whether enforced expression of the bcl-2 transgene could promote survival of thymocyte populations in c-kit or c mutant mice, the Ebcl-2-25 mouse line was crossed to c-kit and c single and double mutants. In the E-bcl-2-25 mouse line, transgene expression is T lineage specific (22) with an early onset of transgene expression at the CD3 CD4 CD8 stage of development (24). Functional transgenic Bcl-2 expression can be monitored by extended survival times of primary thymocytes isolated ex vivo (24). To test whether the E-bcl transgene was functional in these growth factor receptor mutants, total thymocytes from each strain were cultured in normal cell culture medium, and, at various times after initiation of the culture, numbers of viable cells were determined (Fig. 1, A C). The results from c and c-kit wild-type mice demonstrated, as expected, that cell death of transgenic thymocytes was delayed by about 2 d when compared with nontransgenic cells cultured in parallel (Fig. 1 A). Bcl-2 also mediated in vitro survival of thymocytes from c (Fig. 1 B) or c-kit (Fig. 1 C) single mutant mice. Due to the complete absence of thymocytes (see below), the corresponding analysis could not be performed in c-kit c mice Bcl-2 Does Not Substitute for c-kit and c Signaling in Thymocytes
3 Figure 1. The bcl-2 transgene (E-bcl-2-25) is functional in T cell progenitors in c-kit and c mutant thymocytes. Nontransgenic (open symbols) or E-bcl-2-25 transgenic (filled symbols) total thymocytes from 5-d-old c-kit c (A) or c-kit c mice (B), or c-kit c mice (C), or cell sorter purified progenitor subsets (CD3 CD4 CD8 [TN]) c-kit CD25 - CD44 (TN 1) (D), or c-kit CD25 CD44 (TN 2) (E) from 5-d-old c-kit c mice were compared for their survival capacity ex vivo. Each symbol represents an individual mouse for each genotype. Thymocytes were placed in normal cell culture medium in vitro, and, at indicated times after initiation of the culture, numbers of viable cells were determined. The E-bcl-2-25 transgene is functional in all of these growth factor receptor mutants, and, importantly, promotes thymocyte survival at those stages, in which mutations in c-kit or c impair thymocyte development (TN 1 and TN 2). Due to the complete absence of thymocytes in the combined absence of c-kit and c, the corresponding analysis could not be performed on c-kit c mice. C-kit and c play essential roles at the most immature stages of T cell development defined by the phenotypes CD25 CD44 (TN 1) and CD25 CD44 (TN 2) (4). In a previous analysis of the E-bcl-2-25 transgenic mice line, functional expression of the bcl-2 transgene was shown for TN 1 cells (24) but c-kit expression was not used to identify pro-t cell within TN 1. The TN 1 population contains c-kit and c-kit cells, and the pro-t cell activity resides only in the c-kit fraction (25). Therefore, we analyzed bcl-2 transgene function in c-kit TN 1 as well as in c-kit TN 2 cells by comparison of the ex vivo survival potential of cell sorter purified progenitors from wild-type and Ebcl-2-25 transgenic mice. The results demonstrate that this bcl-2 transgene clearly promoted survival of both c-kit TN 1 (Fig. 1 D) and c-kit TN 2 (Fig. 1 E) cells in vitro. Thus, the E-bcl-2-25 transgene is functional in all of these growth factor receptor mutants, and, importantly, promotes thymocyte survival at those stages, in which thymocyte development is impaired due to lack of c-kit and c. These data confirm the findings of O Reilly and colleagues (24) on the function of this bcl-2 transgene in TN 2 cells, and extend functional bcl-2 expression to the c-kit (pro-t cell fraction) within the TN 1 subset. Kondo et al. (15) reported that the E-bcl-2-25 transgene was not expressed in the earliest thymic precursors defined as CD3 CD25 c-kit (which would include c-kit TN 1 cells). However, our experiments clearly show that this is not the case, as both c-kit TN 1 and c-kit TN 2 cells from E-bcl-2-25 were markedly protected from growth factor withdrawal induced apoptosis. Collectively, the Ebcl-2-25 transgene is functional at exactly those stages of development which are affected by lack of SCF and IL-7. Enforced Expression of Bcl-2 Does Not Normalize Thymus Cellularity in Postnatal c-kit and c Single and Double Mutant Mice In Vivo. Given that the bcl-2 transgene was functional in the relevant progenitor subsets, we next analyzed the effect of the transgene on thymus cellularity of various growth factor receptor mutants. Mice of eight different genotypes were generated and analyzed: (a) wild-type mice with (c-kit c bcl ) or (b) without (c-kit c bcl ) the transgene, (c) mice deficient for c-kit with (c-kit c bcl ), or (d) without (c-kit c bcl ) the transgene, (e) mice deficient for c with (c-kit c bcl ), or (f) without (ckit c bcl ) the transgene, and, finally, mice deficient for both c-kit and c with (g) (c-kit c bcl ) or (h) without (c-kit c bcl ) the transgene. In Fig. 2, thymocyte numbers are plotted for all individual mice of the indicated genotypes. In total, 121 mice were analyzed (numbers of mice for each group are indicated in Fig. 2). As null mutations in c-kit (c-kit W/W ) are lethal within 10 d after birth, these analyses were done on day 5. Thymocyte numbers are reduced 15-fold in c single and 6-fold in c-kit single mutants when compared with wild-type mice (4). As shown in Fig. 2, and as reported previously (22), the bcl-2 transgene did not increase the total cell number in wild-type mice (Fig. 2 A). Notably, there was no significant (for the statistical evaluation, see the legend to Fig. 2) effect of the bcl-2 transgene on thymocyte cellularity in c single (Fig. 2 B) and c-kit single (Fig. 2 C) mutants. Thus, enforced expression of bcl-2 does not rescue overall thymic cellularity caused by lack of c or c-kit. The thymus anlage in c-kit c mice, lacking lymphoid cells, is composed of disorganized thymic epithelium (26) and thymic dendritic cells, the latter cell type developing independently of these growth factor receptors and any detectable pro-t cell compartment (27). Given that thymocytes are undetectable in the double mutant thymus, this null base line mutant should be very sensitive to uncover even marginal effects of the bcl-2 transgene, i.e., even a partial Bcl-2 mediated rescue should be evident by the reappearance of thymocytes. However, even in bcl-2 transgenic c-kit c mice, thymocytes failed to appear. In the Adult Thymus, the Developmental Block Caused by c Deficiency Is Not Rescued by Transgenic Bcl-2. Given that c-kit null mutations are lethal by postnatal day 10, all experiments involving c-kit single and double mutants, including the relevant control mice (Fig. 2), were performed on 5-dold mice. While these experiments were underway, other investigators reported that bcl-2 transgenes, when bred onto c -deficient mice, rescued T lymphopoiesis, but not B or NK cell development (15). Similarly, bcl-2 transgenes were 1433 Rodewald et al. Brief Definitive Report
4 Figure 2. Failure of transgenic Bcl-2 to normalize thymocyte cellularity in c-kit and c single and double mutant mice in vivo. To asses the effect of transgenic Bcl-2 expression on thymus cellularity in growth factor receptor mutants, mice of eight different genotypes were generated: (A) wild-type mice without (c-kit c bcl ; ) or with (c-kit c bcl ; ) the transgene; (B) mice deficient for c without (c-kit c bcl ; ) or with (c-kit c bcl ; ) the transgene; (C) mice deficient for c-kit without (c-kit c bcl ; ) or with (c-kit c bcl ; ) the transgene; (D) mice deficient for both c-kit and c without (c-kit c bcl ; ) or with (c-kit c bcl ; ) the transgene. All mice were analyzed on postnatal day 5 because c-kit W/W mice die by postnatal day 10. Numbers of mice per group are indicated above each group; below, the mean cell number 1 SD are shown. No significant differences were observed between bcl-2 transgenic and nontransgenic mice (Wilcoxon rank-sum test: A: transgenic vs. nontransgenic, rank-sum normal statistic with correction Z , P value ; B: transgenic vs. nontransgenic, rank-sum normal statistic with correction Z 1.645, P value 0.1; C: transgenic vs. nontransgenic, rank-sum normal statistic with correction Z , P value , alternative hypothesis: true is not equal to 0; in D, no statistical comparison was done because thymocytes were undetectable in both groups of mice). Thymocytes were undetectable in c-kit c mice regardless of the bcl-2 transgene. also reported to rescue thymocytes cellularity in IL-7R deficient mice (9, 14). The discrepancy between lack of rescue of the c deficiency phenotype, in our hands, and rescue by bcl-2 transgenes, reported by others, prompted us to analyze whether a Bcl-2 effect was only evident in adult mice, but not in postnatal mice. Therefore, the impact of the Ebcl-2-25 transgene on thymocyte cellularity in adult c - deficient mice was analyzed. In Fig. 3, thymus cellularity is compared between c and c bcl mice. When thymus cell numbers were analyzed in large numbers (n 25) of c mice, we surprisingly observed a wide range of cell numbers spreading over more than two logs. This data set suggests that c mice fall into three groups: (a) mice with a relatively mild phenotype (10 7 thymocytes), (b) mice with an intermediate phenotype (between 10 6 and 10 7 thymocytes), and (c) mice with the most severe phenotype (between 10 5 and 10 6 thymocytes). Of note, a similar distribution was found in c bcl (n 34) mice. As these mice, but also mice examined by Kondo et al. (15), were on mixed genetic backgrounds (here WB, C57Bl/6, Balb/c, and 129/ ola), it is possible that the penetrance of the c mutation is subject to modifier genes, reminiscent of phenotypic heterogeneity in IL-7R mutants (28). Nevertheless, comparing c versus c bcl mice, both range and mean values were overlapping and not significantly different. Thus, in our hands, a functionally active bcl-2 transgene does not rescue thymus cellularity in adult c -deficient mice. Failure of Transgenic bcl-2 to Release c -deficient Postnatal or Adult Pre-T Cells from Their Phenotypic Block at the CD25 CD44 / Stage. A genetic rescue of a developmental block in the thymus should be evident by increased cell numbers, and by a release from the phenotypic developmental block. For instance, introduction of a TCR chain into SCID or recombination activating gene (RAG) mutant mice releases thymocytes from their block in differentiation at the TN 3 stage, and promotes the development of TN 4 cells and large numbers of CD4 CD8 thymocytes (for a review, see reference 29). In mice lacking c or IL-7R or IL-7, TN thymocyte development is severely affected. The transition from TN 2 (CD44 CD25 ) to TN 3 (CD44 CD25 ) stages is (incompletely) blocked causing increased percentages of TN 2 and decreased percentages of TN 3 cells (30, 31). As a result of this block, TN 4 (CD44 CD25 ) cells are reduced 10-fold in c /IL-7R/ IL-7 mutants (for a review, see reference 2). To test the possibility that such phenotypic release took place without affecting overall cell numbers (Figs. 2 and 3), lineage marker (CD3, CD4, CD8, B220, Ter119, Mac 1, Gr-1, DX5)-negative thymocytes were analyzed by flow cytometry for expression of CD44 versus CD25 (Fig. 4). This analysis included postnatal 5-d-old (Fig. 4, A C) and adult (Fig. 4, D F) mice which had a wild-type (Fig. 4, A and D), c - deficient (Fig. 4, B and E), or c -deficient bcl-2 transgenic (Fig. 4, C and F) genotype. In agreement with previous re Bcl-2 Does Not Substitute for c-kit and c Signaling in Thymocytes
5 Figure 3. Failure of transgenic bcl-2 to rescue thymus cellularity in adult c -deficient mice in vivo. To analyze the effect of transgenic bcl-2 expression on thymus cellularity in adult mice, animals of four different genotypes were generated: (A) wild-type mice without (c-kit c bcl ; ) or with (c-kit c bcl ; ) the transgene, and (B) mice deficient for c without (c-kit c bcl ; ) or with (c-kit c bcl ; ) the transgene. Null mutations in c-kit (W/W) are lethal by postnatal day 10 precluding an analysis of adult c-kit deficient mice. Numbers of mice per group are indicated above each group; below, the mean thymocyte cell number 1 SD are shown. Although the range of cell numbers per thymus was very large (over two logs), no differences were observed between bcl-2 transgenic and nontransgenic mice. ports (30, 31), data shown in Fig. 4 confirmed that the transition from TN 2 to TN 3 was inhibited, and that TN 4 cells were almost absent in postnatal ( c : 16% TN 2, 63% TN 3, and 14% TN 4, versus c : 42% TN 2, 36% TN 3, and 1% TN 4), and in adult ( c : 11% TN 2, 63% TN 3, and 21% TN 4, versus c : 40% TN 2, 57% TN 3, and 1% TN 4) c mice. A release from the c block should be detectable by decreased percentages of TN 2, increased percentages of TN 3, and, most visibly, by rescue of the lacking TN 4 compartment. Notably, the pattern of TN subsets was unaltered comparing c and c bcl mice. This holds true for both young (Fig. 4, compare B versus C) and adult (Fig. 4, compare E versus F) mice. In addition to the overall reduction in thymocyte cellularity, cell numbers of lineage marker negative thymocytes are also reduced in c mice (for a review, see reference 2). To analyze lin cell numbers in these mutant mice, CD4 CD8 (DN) thymocytes were prepared (by CD4 and CD8 antibody binding and C lysis), and the resulting populations were stained with the lineage-mix antibodies (see above). In postnatal c mice, the DN population contained 43% lin cells (which split into TN 1 to 4 subsets as Figure 4. Failure of transgenic bcl-2 to release postnatal or adult pre-t cells from their c dependence at the CD44 / CD25 stage. Lineage marker (CD3, CD4, CD8, B220, Ter119, Mac 1 Gr-1, DX5) negative thymocytes from postnatal day 5 (A C) or adult (D F) c wild-type (A and D), c (B and E), or c bcl (C and F) mice were analyzed by flow cytometry for expression of CD44 vs. CD25. Thymocytes develop from TN 1 (CD44 CD25 ) via TN 2 (CD44 CD25 ) and TN 3 (CD44 CD25 ) to TN 4 (CD44 CD25 ). In c mice, thymocyte development is incompletely blocked at the transition from TN 2 to TN 3 causing a strong reduction in TN 4 cells (14% [postnatal] or 21% [adult] TN 4 in c wildtype vs. 1% [for both postnatal or adult] TN 4 in c ). Enforced expression of Bcl-2 does not release postnatal or adult thymic precursors from the developmental block caused by lack of c (compare B vs. C, and E vs. F). shown in Fig. 4). In contrast, in postnatal c mice, the DN population contained only 27% lin cells, demonstrating an overall reduction in thymic lin cellularity of 30-fold (15-fold reduction in overall thymocyte cell numbers and 2-fold reduction of lin cells). The proportion of thymic lin cells among DN thymocytes was strongly reduced in adult mice compared with young mice (young c mice: 43% lin cells; adult c mice: 7.8% lin cells). Of note, the proportion of thymic lin cells among DN thymocytes was not increased by Bcl-2 (postnatal mice: c 43%; c 27%; c bcl 25% lin cells; adult mice: c 7.8%; c 4.4%; c bcl 3.1% lin cells). Collectively, enforced expression of Bcl-2 failed to rescue the phenotypic block in c mice, and did not increase the lin thymic progenitor pool. This finding is consistent with the observed inability of Bcl-2 to rescue overall thymus cell numbers. In two previous reports suggesting that transgenic bcl-2 can rescue T cell development in c mice (8, 15), the phenotype of TN subsets, according to CD44 versus CD25 staining on lineage-negative thymocytes, had not been investigated. Kondo et al. analyzed progenitor populations 1435 Rodewald et al. Brief Definitive Report
6 for expression of c-kit versus CD25 gated on CD3 cells, an analysis which may have obscured phenotype and distribution within TN subsets because many other nonprogenitor cells types such as NK cell or myeloid cells are included in the CD3 cells. Clearly, failure of a Bcl-2 mediated rescue of the strongest c phenotype, i.e., restoration of TN 4 cells, cannot be detected by this type of analysis. Phenotypic and quantitative analysis of the TN 1 to 4 subsets in c mice now demonstrates that enforced expression of Bcl-2 does not rescue the c - mutant phenotype. Growth Factor and Cytokine Receptor mediated Signals Are Essential at the First Stages of Thymocyte Development: A Role for Bcl-2? Thymocyte development can be separated into three major stages: (a) growth factor and cytokine-driven expansion of rare colonizing T cell progenitors; (b) selection of thymocytes for further development on the basis of productive TCR chain/pre-tcr complexes (-selection); and (c) / TCR based repertoire selection. In this first expansion, c-kit and c act in synergy, i.e., each receptor mutant alone is permissive for T cell development but mice lacking both receptors lack all thymocytes. The molecular mechanisms underlying this signaling synergy in thymocytes are unknown. Clearly, one or both of these receptors may induce an intracellular antiapoptotic signal. In this paper, we have analyzed bcl-2 transgenic young c-kit and c single and double mutants, as well as adult c mutants. Enforced expression of Bcl-2 (a) failed to increase thymocyte cell numbers, and (b) TN 2/TN 3 cells were not released from their block in differentiation. To account for possible variability within each group of mice, large numbers of animals were analyzed, both for young and adult mice. Consistent with our finding that Bcl-2 expression cannot overcome defects in c-kit thymocytes, Domen and Weissman showed that development of mast cells, a c-kit dependent lineage, was not rescued by expression of transgenic bcl-2 in viable c-kit mutants (W 41 /W 41 ) (6). In stem cells, Bcl-2 has been suggested to synergize with c-kit signaling in preventing cell death but here, as in early thymocytes (this study), Bcl-2 does not act downstream of c-kit (6). The effect of bcl-2 transgenes on the thymus in c -deficient mice, has been studied by us (this paper) and others (8, 15), and the results are clearly conflicting. Kondo et al., crossed the very same bcl-2 transgenic line (E-bcl-2-25) into c -deficient mice, and reported partial rescue of thymocyte cell numbers ( [ c ] vs [ c E-bcl-2-25]). Although these authors observed almost no effect of the bcl-2 transgene on the precursor pool (defined as CD3 c-kit ), the increase in cell numbers was somewhat stronger in more mature thymocytes (defined as CD69 cells). We cannot reproduce the findings by Kondo et al. on overall cellularity, and have no evidence that bcl-2 expression can release c -deficient thymocytes from their phenotypic block (Fig. 4). This discrepancy is not easily explained. One caveat in this type of genetic rescue experiment by crossing mouse lines is based on the fact that c deficiency appears to be sensitive to modifier genes, and mice used in our study, but also mice used by others, were on mixed genetic backgrounds (only three backcrosses to B6 [15], or two backcrosses to B6 or Balb/c [8]). As we observed a massive heterogeneity of cell numbers ranging over two logs which became evident only after analysis of large numbers of mice (Fig. 3), such heterogeneity might account for the observed discrepancies. A detailed analysis of the developmental block in c -mutants with mild versus intermediate versus severe reduction in thymus cell numbers could give insights into the nature of any modifier gene(s). Enforced Bcl-2 expression may tilt death versus differentiation choices constantly towards survival along various intra- and extrathymic T cell differentiation stages, causing nonspecific accumulation of thymocytes. Nevertheless, we conclude that a Bcl-2 mediated survival signal is insufficient to substitute for the stage-specific signals provided by c-kit and c in prothymocyte development. We thank Drs. H.J. Fehling, U. Grawunder (Ulm), J.P. DiSanto (Paris), and H. Spits (Amsterdam) for discussions, and Dr. Louise Ryan (Dana-Farber Cancer Institute, Boston, MA) for statistical evaluations. The Basel Institute for Immunology was founded and supported by F. Hoffmann-La Roche (Basel, Switzerland). H.R. Rodewald was supported by the Deutsche Forschungsgemeinschaft (SFB-497- B5), and by Landesforschungschwerpunkt Baden-Württemberg. Submitted: 26 February 2001 Revised: 25 April 2001 Accepted: 27 April 2001 References 1. Akashi, K., M. Kondo, and I.L. Weissman Role of interleukin-7 in T-cell development from hematopoietic stem cells. Immunol. Rev. 165: DiSanto, J.P., and H.-R. Rodewald In vivo roles of receptor tyrosine kinases and cytokine receptors in thymocyte development. Curr. Opin. Immunol. 10: Haks, M.C., M.A. Oosterwegel, B. Blom, H.M. Spits, and A.M. Kruisbeek Cell-fate decisions in early T cell development: regulation by cytokine receptors and the pre- TCR. Semin. Immunol. 11: Rodewald, H.-R., M. Ogawa, C. Haller, W. Waskow, and J.P. DiSanto Pro-thymocyte expansion by c-kit and the common cytokine receptor chain is essential for repertoire formation. Immunity. 6: Rodewald, H.-R., and C. Haller Antigen receptor junctional diversity in growth factor receptor deficient mice. Dev. Comp. Immunol. 22: Domen, J., and I.L. Weissman Hematopoietic stem cells need two signals to prevent apoptosis; BCL-2 can provide one of these, Kitl/c-Kit signaling the other. J. Exp. Med. 192: Moore, N.C., G. Anderson, G.T. Williams, J.J. Owen, and E.J. Jenkinson Developmental regulation of bcl-2 expression in the thymus. Immunology. 81: Nakajima, H., and W.J. Leonard Role of Bcl-2 in alpha beta T cell development in mice deficient in the common cytokine receptor gamma-chain: the requirement for Bcl-2 differs depending on the TCR/MHC affinity. J. Immunol. 162: Akashi, K., M. Kondo, U. von Freeden-Jeffry, R. Murray, 1436 Bcl-2 Does Not Substitute for c-kit and c Signaling in Thymocytes
7 and I.L. Weissman Bcl-2 rescues T lymphopoiesis in interleukin-7 receptor-deficient mice. Cell. 89: von Freeden-Jeffry, U., N. Solvason, M. Howard, and R. Murray The earliest T lineage-committed cells depend on IL-7 for Bcl-2 expression and normal cell cycle progression. Immunity Veis, D.J., C.M. Sorenson, J.R. Shutter, and S.J. Korsmeyer Bcl-2-deficient mice demonstrate fulminant lymphoid apoptosis, polycystic kidneys, and hypopigmented hair. Cell. 75: Nakayama, K., I. Negishi, K. Kuida, H. Sawa, and D.Y. Loh Targeted disruption of Bcl-2 alpha beta in mice: occurrence of gray hair, polycystic kidney disease, and lymphocytopenia. Proc. Natl. Acad. Sci. USA. 91: Matsuzaki, Y., K. Nakayama, T. Tomita, M. Isoda, D.Y. Loh, and H. Nakauchi Role of bcl-2 in the development of lymphoid cells from the hematopoietic stem cell. Blood. 89: Maraskovsky, E., L.A. O Reilly, M. Teepe, L.M. Corcoran, J.J. Peschon, and A. Strasser Bcl-2 can rescue T lymphocyte development in interleukin-7 receptor-deficient mice but not in mutant rag-1 / mice. Cell. 89: Kondo, M., K. Akashi, J. Domen, K. Sugamura, and I.L. Weissman Bcl-2 rescues T lymphopoiesis, but not B or NK cell development, in common gamma chain-deficient mice. Immunity. 7: Blom, B., H. Spits, and P. Krimpenfort The role of the common gamma chain of the IL-2, IL-4, IL-7 and IL-15 receptors in development of lymphocytes. Constitutive expression of bcl-2 does not rescue the developmental defects in gamma common deficient mice. In Cytokines and Growth Factors in Blood Transfusion. C.T. Smit Sibinga, P.C. Das, and B. Löwenberg, editors. B. Kluwer Academic Publishers, Dordrecht, Netherlands Jacobs, H., P. Krimpenfort, M. Haks, J. Allen, B. Blom, C. Demolliere, A. Kruisbeek, H. Spits, and A. Berns PIM1 reconstitutes thymus cellularity in interleukin 7- and common gamma chain-mutant mice and permits thymocyte maturation in Rag- but not CD3gamma-deficient mice. J. Exp. Med. 190: Linnekin, D Early signaling pathways activated by c-kit in hematopoietic cells. Int. J. Biochem. Cell Biol. 31: Carson, W.E., S. Haldar, R.A. Baiocchi, C.M. Croce, and M.A. Caligiuri The c-kit ligand suppresses apoptosis of human natural killer cells through the upregulation of bcl-2. Proc. Natl. Acad. Sci. USA. 91: Russell, E Hereditary anemias of the mouse: a review for geneticists. Adv. Genet. 20: DiSanto, J.P., W. Müller, D. Guy-Grand, A. Fischer, and K. Rajewsky Lymphoid development in mice with a targeted deletion of the interleukin 2 receptor gamma chain. Proc. Natl. Acad. Sci. USA. 92: Strasser, A., A.W. Harris, and S. Cory bcl-2 transgene inhibits T cell death and perturbs thymic self-censorship. Cell. 67: Russell, E.S., and F.A. Lawson Selection and inbreeding for longevity of a lethal type. J. Hered. 50: O Reilly, L., A. Harris, and A. Strasser bcl-2 transgene expression promotes survival and reduces proliferation of CD3 CD4 CD8 T cell progenitors. Int. Immunol. 9: Godfrey, D.I., A. Zlotnik, and T. Suda Phenotypic and functional characterisation of c-kit expression during intrathymic T cell development. J. Immunol. 149: Rodewald, H.-R., and H.J. Fehling Molecular and cellular events in early thymocyte development. Adv. Immunol. 69: Rodewald, H.R., T. Brocker, and C. Haller Developmental dissociation of thymic dendritic cell and thymocyte lineages revealed in growth factor receptor mutant mice. Proc. Natl. Acad. Sci. USA. 96: Peschon, J.J., P.J. Morrissey, K.H. Grabstein, F.J. Ramsdell, E. Maraskovsky, B.C. Gliniak, L.S. Park, S.F. Siegler, D.E. Williams, C.B. Ware, et al Early lymphocyte expansion is severely impaired in interleukin 7 receptor deficient mice. J. Exp. Med. 180: Kisielow, P., and H. von Boehmer Development and selection of T cells: facts and puzzles. Adv. Immunol. 58: Moore, T.A., U. von-freeden-jeffry, R. Murray, and A. Zlotnik Inhibition of gamma delta T cell development and early thymocyte maturation in IL-7 / mice. J. Immunol. 157: He, Y.W., H. Nakajima, W.J. Leonhard, B. Adkins, and T.R. Malek The common -chain of cytokine receptors regulates intrathymic T cell development at multiple stages. J. Immunol. 158: Rodewald et al. Brief Definitive Report
Development of B and T lymphocytes
 Development of B and T lymphocytes What will we discuss today? B-cell development T-cell development B- cell development overview Stem cell In periphery Pro-B cell Pre-B cell Immature B cell Mature B cell
Development of B and T lymphocytes What will we discuss today? B-cell development T-cell development B- cell development overview Stem cell In periphery Pro-B cell Pre-B cell Immature B cell Mature B cell
Thymic dendritic cells (DCs) reside in the medulla and at the
 Developmental dissociation of thymic dendritic cell and thymocyte lineages revealed in growth factor receptor mutant mice Hans-Reimer Rodewald*, Thomas Brocker, and Corinne Haller Basel Institute for Immunology,
Developmental dissociation of thymic dendritic cell and thymocyte lineages revealed in growth factor receptor mutant mice Hans-Reimer Rodewald*, Thomas Brocker, and Corinne Haller Basel Institute for Immunology,
Recommended reading: Abbas et al. 5th edition, chapters 7 and 10; Janeway and Travers, 5th edition, chapter 7.
 Harvard-MIT Division of Health Sciences and Technology HST.176: Cellular and Molecular Immunology Course Director: Dr. Shiv Pillai 10/05/05; 11 AM Shiv Pillai T Lymphocyte Development Recommended reading:
Harvard-MIT Division of Health Sciences and Technology HST.176: Cellular and Molecular Immunology Course Director: Dr. Shiv Pillai 10/05/05; 11 AM Shiv Pillai T Lymphocyte Development Recommended reading:
T cell development October 28, Dan Stetson
 T cell development October 28, 2016 Dan Stetson stetson@uw.edu 441 Lecture #13 Slide 1 of 29 Three lectures on T cells (Chapters 8, 9) Part 1 (Today): T cell development in the thymus Chapter 8, pages
T cell development October 28, 2016 Dan Stetson stetson@uw.edu 441 Lecture #13 Slide 1 of 29 Three lectures on T cells (Chapters 8, 9) Part 1 (Today): T cell development in the thymus Chapter 8, pages
Introduc)on to Immunology. T Cell Development
 Introduc)on to Immunology T Cell Development Adolfo Ferrando af2196@columbia.edu Thymic stroma cells provide the microenvironment for T cell development. T cells develop from progenitors that are derived
Introduc)on to Immunology T Cell Development Adolfo Ferrando af2196@columbia.edu Thymic stroma cells provide the microenvironment for T cell development. T cells develop from progenitors that are derived
T cell maturation. T-cell Maturation. What allows T cell maturation?
 T-cell Maturation What allows T cell maturation? Direct contact with thymic epithelial cells Influence of thymic hormones Growth factors (cytokines, CSF) T cell maturation T cell progenitor DN DP SP 2ry
T-cell Maturation What allows T cell maturation? Direct contact with thymic epithelial cells Influence of thymic hormones Growth factors (cytokines, CSF) T cell maturation T cell progenitor DN DP SP 2ry
T Cell Development. Xuefang Cao, MD, PhD. November 3, 2015
 T Cell Development Xuefang Cao, MD, PhD November 3, 2015 Thymocytes in the cortex of the thymus Early thymocytes development Positive and negative selection Lineage commitment Exit from the thymus and
T Cell Development Xuefang Cao, MD, PhD November 3, 2015 Thymocytes in the cortex of the thymus Early thymocytes development Positive and negative selection Lineage commitment Exit from the thymus and
Supplementary Table; Supplementary Figures and legends S1-S21; Supplementary Materials and Methods
 Silva et al. PTEN posttranslational inactivation and hyperactivation of the PI3K/Akt pathway sustain primary T cell leukemia viability Supplementary Table; Supplementary Figures and legends S1-S21; Supplementary
Silva et al. PTEN posttranslational inactivation and hyperactivation of the PI3K/Akt pathway sustain primary T cell leukemia viability Supplementary Table; Supplementary Figures and legends S1-S21; Supplementary
T Cell Differentiation
 T Cell Differentiation Ned Braunstein, MD MHC control of Immune Responsiveness: Concept Whether or not an individual makes an immune response to a particular antigen depends on what MHC alleles an individual
T Cell Differentiation Ned Braunstein, MD MHC control of Immune Responsiveness: Concept Whether or not an individual makes an immune response to a particular antigen depends on what MHC alleles an individual
Supplementary Materials for
 www.sciencesignaling.org/cgi/content/full/3/114/ra23/dc1 Supplementary Materials for Regulation of Zap70 Expression During Thymocyte Development Enables Temporal Separation of CD4 and CD8 Repertoire Selection
www.sciencesignaling.org/cgi/content/full/3/114/ra23/dc1 Supplementary Materials for Regulation of Zap70 Expression During Thymocyte Development Enables Temporal Separation of CD4 and CD8 Repertoire Selection
Development of Natural Killer Cells from Lymphohematopoietic Progenitors of Murine Fetal Liver
 Development of Natural Killer Cells from Lymphohematopoietic Progenitors of Murine Fetal Liver YUKHI AIBA, MAKIO OGAWA Ralph H. Johnson Department of Veterans Affairs Medical Center, Department of Medicine,
Development of Natural Killer Cells from Lymphohematopoietic Progenitors of Murine Fetal Liver YUKHI AIBA, MAKIO OGAWA Ralph H. Johnson Department of Veterans Affairs Medical Center, Department of Medicine,
Supplementary Figure 1. Characterization of basophils after reconstitution of SCID mice
 Supplementary figure legends Supplementary Figure 1. Characterization of after reconstitution of SCID mice with CD4 + CD62L + T cells. (A-C) SCID mice (n = 6 / group) were reconstituted with 2 x 1 6 CD4
Supplementary figure legends Supplementary Figure 1. Characterization of after reconstitution of SCID mice with CD4 + CD62L + T cells. (A-C) SCID mice (n = 6 / group) were reconstituted with 2 x 1 6 CD4
5/1/13. The proportion of thymus that produces T cells decreases with age. The cellular organization of the thymus
 T cell precursors migrate from the bone marrow via the blood to the thymus to mature 1 2 The cellular organization of the thymus The proportion of thymus that produces T cells decreases with age 3 4 1
T cell precursors migrate from the bone marrow via the blood to the thymus to mature 1 2 The cellular organization of the thymus The proportion of thymus that produces T cells decreases with age 3 4 1
Overview B cell development T cell development
 Topics Overview B cell development T cell development Lymphocyte development overview (Cont) Receptor diversity is produced by gene rearrangement and is random Includes specificities that will bind to
Topics Overview B cell development T cell development Lymphocyte development overview (Cont) Receptor diversity is produced by gene rearrangement and is random Includes specificities that will bind to
Out of the IgM B cells that develop daily in the
 Published Online: 5 July, 1999 Supp Info: http://doi.org/10.1084/jem.190.1.75 Downloaded from jem.rupress.org on May 9, 2018 B Cell Development in the Spleen Takes Place in Discrete Steps and Is Determined
Published Online: 5 July, 1999 Supp Info: http://doi.org/10.1084/jem.190.1.75 Downloaded from jem.rupress.org on May 9, 2018 B Cell Development in the Spleen Takes Place in Discrete Steps and Is Determined
The development of T cells in the thymus
 T cells rearrange their receptors in the thymus whereas B cells do so in the bone marrow. The development of T cells in the thymus The lobular/cellular organization of the thymus Immature cells are called
T cells rearrange their receptors in the thymus whereas B cells do so in the bone marrow. The development of T cells in the thymus The lobular/cellular organization of the thymus Immature cells are called
Figure S1. Generation of inducible PTEN deficient mice and the BMMCs (A) B6.129 Pten loxp/loxp mice were mated with B6.
 Figure S1. Generation of inducible PTEN deficient mice and the BMMCs (A) B6.129 Pten loxp/loxp mice were mated with B6.129-Gt(ROSA)26Sor tm1(cre/ert2)tyj /J mice. To induce deletion of the Pten locus,
Figure S1. Generation of inducible PTEN deficient mice and the BMMCs (A) B6.129 Pten loxp/loxp mice were mated with B6.129-Gt(ROSA)26Sor tm1(cre/ert2)tyj /J mice. To induce deletion of the Pten locus,
Supplementary Figure S1. Flow cytometric analysis of the expression of Thy1 in NH cells. Flow cytometric analysis of the expression of T1/ST2 and
 Supplementary Figure S1. Flow cytometric analysis of the expression of Thy1 in NH cells. Flow cytometric analysis of the expression of T1/ST2 and Thy1 in NH cells derived from the lungs of naïve mice.
Supplementary Figure S1. Flow cytometric analysis of the expression of Thy1 in NH cells. Flow cytometric analysis of the expression of T1/ST2 and Thy1 in NH cells derived from the lungs of naïve mice.
SUPPLEMENTARY INFORMATION
 Complete but curtailed T-cell response to very-low-affinity antigen Dietmar Zehn, Sarah Y. Lee & Michael J. Bevan Supp. Fig. 1: TCR chain usage among endogenous K b /Ova reactive T cells. C57BL/6 mice
Complete but curtailed T-cell response to very-low-affinity antigen Dietmar Zehn, Sarah Y. Lee & Michael J. Bevan Supp. Fig. 1: TCR chain usage among endogenous K b /Ova reactive T cells. C57BL/6 mice
During development of T cells in the thymus most
 Role of Different T Cell Receptors in the Development of Pre T Cells By Jan Buer,* Iannis Aifantis,* James P. DiSanto, Hans Joerg Fehling, and Harald von Boehmer* From the *Institut Necker, Institut National
Role of Different T Cell Receptors in the Development of Pre T Cells By Jan Buer,* Iannis Aifantis,* James P. DiSanto, Hans Joerg Fehling, and Harald von Boehmer* From the *Institut Necker, Institut National
T Cell Development II: Positive and Negative Selection
 T Cell Development II: Positive and Negative Selection 8 88 The two phases of thymic development: - production of T cell receptors for antigen, by rearrangement of the TCR genes CD4 - selection of T cells
T Cell Development II: Positive and Negative Selection 8 88 The two phases of thymic development: - production of T cell receptors for antigen, by rearrangement of the TCR genes CD4 - selection of T cells
Evidence That versus T Cell Fate Determination Is Initiated Independently of T Cell Receptor Signaling
 Evidence That versus T Cell Fate Determination Is Initiated Independently of T Cell Receptor Signaling By Joonsoo Kang, Ariane Volkmann,* and David H. Raulet* From the *Department of Molecular and Cellular
Evidence That versus T Cell Fate Determination Is Initiated Independently of T Cell Receptor Signaling By Joonsoo Kang, Ariane Volkmann,* and David H. Raulet* From the *Department of Molecular and Cellular
Haematopoietic stem cells
 Haematopoietic stem cells Neil P. Rodrigues, DPhil NIH Centre for Biomedical Research Excellence in Stem Cell Biology Boston University School of Medicine neil.rodrigues@imm.ox.ac.uk Haematopoiesis: An
Haematopoietic stem cells Neil P. Rodrigues, DPhil NIH Centre for Biomedical Research Excellence in Stem Cell Biology Boston University School of Medicine neil.rodrigues@imm.ox.ac.uk Haematopoiesis: An
The Adaptive Immune Response. T-cells
 The Adaptive Immune Response T-cells T Lymphocytes T lymphocytes develop from precursors in the thymus. Mature T cells are found in the blood, where they constitute 60% to 70% of lymphocytes, and in T-cell
The Adaptive Immune Response T-cells T Lymphocytes T lymphocytes develop from precursors in the thymus. Mature T cells are found in the blood, where they constitute 60% to 70% of lymphocytes, and in T-cell
What determines the CD4:CD8 T cell ratio in the immune system?
 What determines the CD4:CD8 T cell ratio in the immune system? Insights from genetic and mathematical modelling of thymocyte development Benedict Seddon 1 Cell of the immune system 2 Maintaining homeostasis
What determines the CD4:CD8 T cell ratio in the immune system? Insights from genetic and mathematical modelling of thymocyte development Benedict Seddon 1 Cell of the immune system 2 Maintaining homeostasis
Attribution: University of Michigan Medical School, Department of Microbiology and Immunology
 Attribution: University of Michigan Medical School, Department of Microbiology and Immunology License: Unless otherwise noted, this material is made available under the terms of the Creative Commons Attribution
Attribution: University of Michigan Medical School, Department of Microbiology and Immunology License: Unless otherwise noted, this material is made available under the terms of the Creative Commons Attribution
pro-b large pre-b small pre-b CCCP (µm) Rag1 -/- ;33.C9HCki
 a TMRM FI (Median) b TMRM FI (Median) c 20 15 10 5 0 8 6 4 2 0 pro-b large pre-b small pre-b 0 10 20 30 40 50 60 70 80 90 100 TMRM (nm) pro-b large pre-b small pre-b 0 1 2 4 8 16 32 64 128 256 CCCP (mm)
a TMRM FI (Median) b TMRM FI (Median) c 20 15 10 5 0 8 6 4 2 0 pro-b large pre-b small pre-b 0 10 20 30 40 50 60 70 80 90 100 TMRM (nm) pro-b large pre-b small pre-b 0 1 2 4 8 16 32 64 128 256 CCCP (mm)
Supplementary Material
 Supplementary Material Taghon, Yui, and Rothenberg: Mast cell diversion of T-lineage precursor cells by the essential T-lineage transcription factor GATA Supplemental Table Supplemental Figures 1-6 Supplemental
Supplementary Material Taghon, Yui, and Rothenberg: Mast cell diversion of T-lineage precursor cells by the essential T-lineage transcription factor GATA Supplemental Table Supplemental Figures 1-6 Supplemental
BCR-ABL - LSK BCR-ABL + LKS - (%)
 Marker Clone BCR-ABL + LSK (%) BCR-ABL + LKS - (%) BCR-ABL - LSK (%) P value vs. BCR-ABL + LKS - P value vs. BCR-ABL - LSK CD2 RM2-5 12.9 ± 3.6 36.7 ± 6.5 19.3 ± 2.4 0.01 0.10 CD5 53-7.3 13.9 ± 3.2 20.8
Marker Clone BCR-ABL + LSK (%) BCR-ABL + LKS - (%) BCR-ABL - LSK (%) P value vs. BCR-ABL + LKS - P value vs. BCR-ABL - LSK CD2 RM2-5 12.9 ± 3.6 36.7 ± 6.5 19.3 ± 2.4 0.01 0.10 CD5 53-7.3 13.9 ± 3.2 20.8
The Bcl-2 regulated apoptotic pathway is critical for the
 The Journal of Immunology Defects in the Bcl-2 Regulated Apoptotic Pathway Lead to Preferential Increase of CD25 low Foxp3 + Anergic CD4 + T Cells Yifan Zhan,*, Yuxia Zhang,* Daniel Gray,* Emma M. Carrington,*
The Journal of Immunology Defects in the Bcl-2 Regulated Apoptotic Pathway Lead to Preferential Increase of CD25 low Foxp3 + Anergic CD4 + T Cells Yifan Zhan,*, Yuxia Zhang,* Daniel Gray,* Emma M. Carrington,*
Supporting Information Table of Contents
 Supporting Information Table of Contents Supporting Information Figure 1 Page 2 Supporting Information Figure 2 Page 4 Supporting Information Figure 3 Page 5 Supporting Information Figure 4 Page 6 Supporting
Supporting Information Table of Contents Supporting Information Figure 1 Page 2 Supporting Information Figure 2 Page 4 Supporting Information Figure 3 Page 5 Supporting Information Figure 4 Page 6 Supporting
The Thymus as The Primary Site of T-cell Production
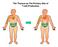 The Thymus as The Primary Site of T-cell Production Thymus Histology Lobulated organ with outer cortex and inner medulla C M Ordered Microenvironments Support T-cell Development CD4-CD8- precursors CD4+CD8+
The Thymus as The Primary Site of T-cell Production Thymus Histology Lobulated organ with outer cortex and inner medulla C M Ordered Microenvironments Support T-cell Development CD4-CD8- precursors CD4+CD8+
Immune tolerance and the prevention of autoimmune diseases
 The Journal of Immunology Deficiency of the Src Homology Region 2 Domain-Containing Phosphatase 1 (SHP-1) Causes Enrichment of CD4 CD25 Regulatory T Cells 1 Jennifer D. Carter,* Gina M. Calabrese,* Makoto
The Journal of Immunology Deficiency of the Src Homology Region 2 Domain-Containing Phosphatase 1 (SHP-1) Causes Enrichment of CD4 CD25 Regulatory T Cells 1 Jennifer D. Carter,* Gina M. Calabrese,* Makoto
MYRIAM CAPONE*, RICHARD D. HOCKETT, JR., AND ALBERT ZLOTNIK* MATERIALS AND METHODS
 Proc. Natl. Acad. Sci. USA Vol. 95, pp. 12522 12527, October 1998 Immunology Kinetics of T cell receptor,, and rearrangements during adult thymic development: T cell receptor rearrangements are present
Proc. Natl. Acad. Sci. USA Vol. 95, pp. 12522 12527, October 1998 Immunology Kinetics of T cell receptor,, and rearrangements during adult thymic development: T cell receptor rearrangements are present
MATERIALS AND METHODS. Neutralizing antibodies specific to mouse Dll1, Dll4, J1 and J2 were prepared as described. 1,2 All
 MATERIALS AND METHODS Antibodies (Abs), flow cytometry analysis and cell lines Neutralizing antibodies specific to mouse Dll1, Dll4, J1 and J2 were prepared as described. 1,2 All other antibodies used
MATERIALS AND METHODS Antibodies (Abs), flow cytometry analysis and cell lines Neutralizing antibodies specific to mouse Dll1, Dll4, J1 and J2 were prepared as described. 1,2 All other antibodies used
sequences of a styx mutant reveals a T to A transversion in the donor splice site of intron 5
 sfigure 1 Styx mutant mice recapitulate the phenotype of SHIP -/- mice. (A) Analysis of the genomic sequences of a styx mutant reveals a T to A transversion in the donor splice site of intron 5 (GTAAC
sfigure 1 Styx mutant mice recapitulate the phenotype of SHIP -/- mice. (A) Analysis of the genomic sequences of a styx mutant reveals a T to A transversion in the donor splice site of intron 5 (GTAAC
Comprehensive evaluation of human immune system reconstitution in NSG. and NSG -SGM3 mouse models toward the development of a novel ONCO-HU
 Comprehensive evaluation of human immune system reconstitution in NSG and NSG -SGM3 mouse models toward the development of a novel ONCO-HU xenograft model Aaron Middlebrook, 1 Eileen Snowden, 2 Warren
Comprehensive evaluation of human immune system reconstitution in NSG and NSG -SGM3 mouse models toward the development of a novel ONCO-HU xenograft model Aaron Middlebrook, 1 Eileen Snowden, 2 Warren
The Differential Staging of Murine Thymic Lymphoma Cell Lines, Scid.adh, R1.1 and EL-4
 The Differential Staging of Murine Thymic Lymphoma Cell Lines, Scid.adh, R1.1 and EL-4 Jong Seok Chae 1, Hae-jung Kim 1, Weon Seo Park 1, Youngmee Bae 1 and Kyeong Cheon Jung 2 1 Department of Pathology,
The Differential Staging of Murine Thymic Lymphoma Cell Lines, Scid.adh, R1.1 and EL-4 Jong Seok Chae 1, Hae-jung Kim 1, Weon Seo Park 1, Youngmee Bae 1 and Kyeong Cheon Jung 2 1 Department of Pathology,
Lecture outline. Immunological tolerance and immune regulation. Central and peripheral tolerance. Inhibitory receptors of T cells. Regulatory T cells
 1 Immunological tolerance and immune regulation Abul K. Abbas UCSF 2 Lecture outline Central and peripheral tolerance Inhibitory receptors of T cells Regulatory T cells 1 The immunological equilibrium:
1 Immunological tolerance and immune regulation Abul K. Abbas UCSF 2 Lecture outline Central and peripheral tolerance Inhibitory receptors of T cells Regulatory T cells 1 The immunological equilibrium:
Supplementary Figure 1 Cytokine receptors on developing thymocytes that can potentially signal Runx3d expression.
 Supplementary Figure 1 Cytokine receptors on developing thymocytes that can potentially signal Runx3d expression. (a) Characterization of c-independent SP8 cells. Stainings for maturation markers (top)
Supplementary Figure 1 Cytokine receptors on developing thymocytes that can potentially signal Runx3d expression. (a) Characterization of c-independent SP8 cells. Stainings for maturation markers (top)
Nature Immunology: doi: /ni Supplementary Figure 1. Huwe1 has high expression in HSCs and is necessary for quiescence.
 Supplementary Figure 1 Huwe1 has high expression in HSCs and is necessary for quiescence. (a) Heat map visualizing expression of genes with a known function in ubiquitin-mediated proteolysis (KEGG: Ubiquitin
Supplementary Figure 1 Huwe1 has high expression in HSCs and is necessary for quiescence. (a) Heat map visualizing expression of genes with a known function in ubiquitin-mediated proteolysis (KEGG: Ubiquitin
Microbiology 204. Background Slides on T Cell Development For Preparation for Flipped Classroom setting. Art Weiss.
 Microbiology 204 Background Slides on T Cell Development For Preparation for Flipped Classroom setting Art Weiss October 23, 2015 Thymic Lobule Structure From: Immunobiology, Janeway, et al., 5th edition
Microbiology 204 Background Slides on T Cell Development For Preparation for Flipped Classroom setting Art Weiss October 23, 2015 Thymic Lobule Structure From: Immunobiology, Janeway, et al., 5th edition
IL-7. Alternative names. Discovery. Structure. Hergen Spits* SUMMARY BACKGROUND
 IL-7 Hergen Spits* Division of Immunology, Netherlands Cancer Institute, Plesmanlaan 121, 1066 CX Amsterdam, The Netherlands * corresponding author tel: 31-20-5122063, fax: 31-20-5122057, e-mail: hergen@nki.nl
IL-7 Hergen Spits* Division of Immunology, Netherlands Cancer Institute, Plesmanlaan 121, 1066 CX Amsterdam, The Netherlands * corresponding author tel: 31-20-5122063, fax: 31-20-5122057, e-mail: hergen@nki.nl
0.0 All T-lymph B-lymph Sarcomas Carcinomas Germ cell. Tumor type
 Fig S1 A B Tumors per mouse 1.2 1.0 0.8 0.6 0.4 0.2 0.0 All T-lymph B-lymph Sarcomas Carcinomas Germ cell Tumor type -/- (n = 46) Q/- (n = 76) Q/Q (n = 31) Tumors per mouse 1.2 1.0 0.8 0.6 0.4 0.2 0.0
Fig S1 A B Tumors per mouse 1.2 1.0 0.8 0.6 0.4 0.2 0.0 All T-lymph B-lymph Sarcomas Carcinomas Germ cell Tumor type -/- (n = 46) Q/- (n = 76) Q/Q (n = 31) Tumors per mouse 1.2 1.0 0.8 0.6 0.4 0.2 0.0
The human thymus is the central lymphoid organ that provides
 Reevaluation of T Cell Receptor Excision Circles as a Measure of Human Recent Thymic Emigrants 1 Ping Ye and Denise E. Kirschner 2 The human thymus exports newly generated T cells to the periphery. As
Reevaluation of T Cell Receptor Excision Circles as a Measure of Human Recent Thymic Emigrants 1 Ping Ye and Denise E. Kirschner 2 The human thymus exports newly generated T cells to the periphery. As
Natural Killer Cells: Development, Diversity, and Applications to Human Disease Dr. Michael A. Caligiuri
 Natural Killer Cells: Development, Diversity, November 26, 2008 The Ohio State University Comprehensive Cancer Center The James Cancer Hospital and Solove Research Institute Columbus, Ohio, USA 1 Human
Natural Killer Cells: Development, Diversity, November 26, 2008 The Ohio State University Comprehensive Cancer Center The James Cancer Hospital and Solove Research Institute Columbus, Ohio, USA 1 Human
CD34+ Cells: A Comparison of Stem and Progenitor Cells in Cord Blood, Peripheral Blood, and the Bone Marrow
 White Paper September 2016 CD34+ Cells: A Comparison of Stem and Progenitor Cells in Cord Blood, Peripheral Blood, and the Bone Marrow Lily C. Trajman, PhD Introduction: Hematopoietic Stem Cells (HSCs)
White Paper September 2016 CD34+ Cells: A Comparison of Stem and Progenitor Cells in Cord Blood, Peripheral Blood, and the Bone Marrow Lily C. Trajman, PhD Introduction: Hematopoietic Stem Cells (HSCs)
SUPPLEMENTARY INFORMATION
 doi: 1.138/nature775 4 O.D. (595-655) 3 1 -ζ no antibody isotype ctrl Plated Soluble 1F6 397 7H11 Supplementary Figure 1 Soluble and plated anti- Abs induce -! signalling. B3Z cells stably expressing!
doi: 1.138/nature775 4 O.D. (595-655) 3 1 -ζ no antibody isotype ctrl Plated Soluble 1F6 397 7H11 Supplementary Figure 1 Soluble and plated anti- Abs induce -! signalling. B3Z cells stably expressing!
Scott Abrams, Ph.D. Professor of Oncology, x4375 Kuby Immunology SEVENTH EDITION
 Scott Abrams, Ph.D. Professor of Oncology, x4375 scott.abrams@roswellpark.org Kuby Immunology SEVENTH EDITION CHAPTER 13 Effector Responses: Cell- and Antibody-Mediated Immunity Copyright 2013 by W. H.
Scott Abrams, Ph.D. Professor of Oncology, x4375 scott.abrams@roswellpark.org Kuby Immunology SEVENTH EDITION CHAPTER 13 Effector Responses: Cell- and Antibody-Mediated Immunity Copyright 2013 by W. H.
T-cell activation T cells migrate to secondary lymphoid tissues where they interact with antigen, antigen-presenting cells, and other lymphocytes:
 Interactions between innate immunity & adaptive immunity What happens to T cells after they leave the thymus? Naïve T cells exit the thymus and enter the bloodstream. If they remain in the bloodstream,
Interactions between innate immunity & adaptive immunity What happens to T cells after they leave the thymus? Naïve T cells exit the thymus and enter the bloodstream. If they remain in the bloodstream,
T-cell activation T cells migrate to secondary lymphoid tissues where they interact with antigen, antigen-presenting cells, and other lymphocytes:
 Interactions between innate immunity & adaptive immunity What happens to T cells after they leave the thymus? Naïve T cells exit the thymus and enter the bloodstream. If they remain in the bloodstream,
Interactions between innate immunity & adaptive immunity What happens to T cells after they leave the thymus? Naïve T cells exit the thymus and enter the bloodstream. If they remain in the bloodstream,
Although the thymus is the predominant source of T cells in
 Identification of an early T cell progenitor for a pathway of T cell maturation in the bone marrow Sussan Dejbakhsh-Jones and Samuel Strober* Division of Immunology and Rheumatology, Department of Medicine,
Identification of an early T cell progenitor for a pathway of T cell maturation in the bone marrow Sussan Dejbakhsh-Jones and Samuel Strober* Division of Immunology and Rheumatology, Department of Medicine,
Ig light chain rearrangement: Rescue pathway
 B Cell Development Ig light chain rearrangement: Rescue pathway There is only a 1:3 chance of the join between the V and J region being in frame Vk Jk Ck Non-productive Rearrangement Light chain has a
B Cell Development Ig light chain rearrangement: Rescue pathway There is only a 1:3 chance of the join between the V and J region being in frame Vk Jk Ck Non-productive Rearrangement Light chain has a
Nature Immunology: doi: /ni Supplementary Figure 1. DNA-methylation machinery is essential for silencing of Cd4 in cytotoxic T cells.
 Supplementary Figure 1 DNA-methylation machinery is essential for silencing of Cd4 in cytotoxic T cells. (a) Scheme for the retroviral shrna screen. (b) Histogram showing CD4 expression (MFI) in WT cytotoxic
Supplementary Figure 1 DNA-methylation machinery is essential for silencing of Cd4 in cytotoxic T cells. (a) Scheme for the retroviral shrna screen. (b) Histogram showing CD4 expression (MFI) in WT cytotoxic
SUPPLEMENTARY INFORMATION
 doi:10.1038/nature10134 Supplementary Figure 1. Anti-inflammatory activity of sfc. a, Autoantibody immune complexes crosslink activating Fc receptors, promoting activation of macrophages, and WWW.NATURE.COM/NATURE
doi:10.1038/nature10134 Supplementary Figure 1. Anti-inflammatory activity of sfc. a, Autoantibody immune complexes crosslink activating Fc receptors, promoting activation of macrophages, and WWW.NATURE.COM/NATURE
Ikaros Null Mice Display Defects in T Cell Selection and CD4 versus CD8 Lineage Decisions
 This information is current as of February 23, 2013. Ikaros Null Mice Display Defects in T Cell Selection and CD4 versus CD8 Lineage Decisions Julie A. Urban and Susan Winandy J Immunol 2004; 173:4470-4478;
This information is current as of February 23, 2013. Ikaros Null Mice Display Defects in T Cell Selection and CD4 versus CD8 Lineage Decisions Julie A. Urban and Susan Winandy J Immunol 2004; 173:4470-4478;
Defensive mechanisms include :
 Acquired Immunity Defensive mechanisms include : 1) Innate immunity (Natural or Non specific) 2) Acquired immunity (Adaptive or Specific) Cell-mediated immunity Humoral immunity Two mechanisms 1) Humoral
Acquired Immunity Defensive mechanisms include : 1) Innate immunity (Natural or Non specific) 2) Acquired immunity (Adaptive or Specific) Cell-mediated immunity Humoral immunity Two mechanisms 1) Humoral
Question 1. Kupffer cells, microglial cells and osteoclasts are all examples of what type of immune system cell?
 Abbas Chapter 2: Sarah Spriet February 8, 2015 Question 1. Kupffer cells, microglial cells and osteoclasts are all examples of what type of immune system cell? a. Dendritic cells b. Macrophages c. Monocytes
Abbas Chapter 2: Sarah Spriet February 8, 2015 Question 1. Kupffer cells, microglial cells and osteoclasts are all examples of what type of immune system cell? a. Dendritic cells b. Macrophages c. Monocytes
EML Erythroid and Neutrophil Differentiation Protocols Cristina Pina 1*, Cristina Fugazza 2 and Tariq Enver 3
 EML Erythroid and Neutrophil Differentiation Protocols Cristina Pina 1*, Cristina Fugazza 2 and Tariq Enver 3 1 Department of Haematology, University of Cambridge, Cambridge, UK; 2 Dipartimento de Biotecnologie
EML Erythroid and Neutrophil Differentiation Protocols Cristina Pina 1*, Cristina Fugazza 2 and Tariq Enver 3 1 Department of Haematology, University of Cambridge, Cambridge, UK; 2 Dipartimento de Biotecnologie
Immunology Lecture 4. Clinical Relevance of the Immune System
 Immunology Lecture 4 The Well Patient: How innate and adaptive immune responses maintain health - 13, pg 169-181, 191-195. Immune Deficiency - 15 Autoimmunity - 16 Transplantation - 17, pg 260-270 Tumor
Immunology Lecture 4 The Well Patient: How innate and adaptive immune responses maintain health - 13, pg 169-181, 191-195. Immune Deficiency - 15 Autoimmunity - 16 Transplantation - 17, pg 260-270 Tumor
Unexpectedly late expression of intracellular CD3ε and TCR γδ proteins during adult thymus development
 International Immunology, Vol. 11, No. 10, pp. 1641 1650 1999 The Japanese Society for Immunology Unexpectedly late expression of intracellular CD3ε and TCR γδ proteins during adult thymus development
International Immunology, Vol. 11, No. 10, pp. 1641 1650 1999 The Japanese Society for Immunology Unexpectedly late expression of intracellular CD3ε and TCR γδ proteins during adult thymus development
Adaptive immune responses: T cell-mediated immunity
 MICR2209 Adaptive immune responses: T cell-mediated immunity Dr Allison Imrie allison.imrie@uwa.edu.au 1 Synopsis: In this lecture we will discuss the T-cell mediated immune response, how it is activated,
MICR2209 Adaptive immune responses: T cell-mediated immunity Dr Allison Imrie allison.imrie@uwa.edu.au 1 Synopsis: In this lecture we will discuss the T-cell mediated immune response, how it is activated,
Follicular Lymphoma. ced3 APOPTOSIS. *In the nematode Caenorhabditis elegans 131 of the organism's 1031 cells die during development.
 Harvard-MIT Division of Health Sciences and Technology HST.176: Cellular and Molecular Immunology Course Director: Dr. Shiv Pillai Follicular Lymphoma 1. Characterized by t(14:18) translocation 2. Ig heavy
Harvard-MIT Division of Health Sciences and Technology HST.176: Cellular and Molecular Immunology Course Director: Dr. Shiv Pillai Follicular Lymphoma 1. Characterized by t(14:18) translocation 2. Ig heavy
Eosinophils are required. for the maintenance of plasma cells in the bone marrow
 Eosinophils are required for the maintenance of plasma cells in the bone marrow Van Trung Chu, Anja Fröhlich, Gudrun Steinhauser, Tobias Scheel, Toralf Roch, Simon Fillatreau, James J. Lee, Max Löhning
Eosinophils are required for the maintenance of plasma cells in the bone marrow Van Trung Chu, Anja Fröhlich, Gudrun Steinhauser, Tobias Scheel, Toralf Roch, Simon Fillatreau, James J. Lee, Max Löhning
Supplementary Information
 Supplementary Information Supplementary Figure 1! a! b! Nfatc1!! Nfatc1"! P1! P2! pa1! pa2! ex1! ex2! exons 3-9! ex1! ex11!!" #" Nfatc1A!!" Nfatc1B! #"!" Nfatc1C! #" DN1! DN2! DN1!!A! #A!!B! #B!!C! #C!!A!
Supplementary Information Supplementary Figure 1! a! b! Nfatc1!! Nfatc1"! P1! P2! pa1! pa2! ex1! ex2! exons 3-9! ex1! ex11!!" #" Nfatc1A!!" Nfatc1B! #"!" Nfatc1C! #" DN1! DN2! DN1!!A! #A!!B! #B!!C! #C!!A!
Nuclear Export of Histone Deacetylase 7 During Thymic Selection is required for Immune Self-tolerance
 The EMBO Journal Peer Review Process File - EMBO-2012-80891 Manuscript EMBO-2012-80891 Nuclear Export of Histone Deacetylase 7 During Thymic Selection is required for Immune Self-tolerance Herbert G Kasler,
The EMBO Journal Peer Review Process File - EMBO-2012-80891 Manuscript EMBO-2012-80891 Nuclear Export of Histone Deacetylase 7 During Thymic Selection is required for Immune Self-tolerance Herbert G Kasler,
By James R. Carlyle,* Alison M. Michie,* Caren Furlonger, Toru Nakano, Michael J. Lenardo, Christopher J. Paige,* and Juan Carlos Zúñiga-Pflücker*
 Published Online: 21 July, 1997 Supp Info: http://doi.org/10.1084/jem.186.2.173 Downloaded from jem.rupress.org on September 2, 2018 Identification of a Novel Developmental Stage Marking Lineage Commitment
Published Online: 21 July, 1997 Supp Info: http://doi.org/10.1084/jem.186.2.173 Downloaded from jem.rupress.org on September 2, 2018 Identification of a Novel Developmental Stage Marking Lineage Commitment
Blocking antibodies and peptides. Rat anti-mouse PD-1 (29F.1A12, rat IgG2a, k), PD-
 Supplementary Methods Blocking antibodies and peptides. Rat anti-mouse PD-1 (29F.1A12, rat IgG2a, k), PD- L1 (10F.9G2, rat IgG2b, k), and PD-L2 (3.2, mouse IgG1) have been described (24). Anti-CTLA-4 (clone
Supplementary Methods Blocking antibodies and peptides. Rat anti-mouse PD-1 (29F.1A12, rat IgG2a, k), PD- L1 (10F.9G2, rat IgG2b, k), and PD-L2 (3.2, mouse IgG1) have been described (24). Anti-CTLA-4 (clone
The g c Family of Cytokines Prof. Warren J. Leonard M.D.
 The Family of Cytokines Chief, Laboratory of Molecular Immunology Director, Immunology Center National Heart, Lung, and Blood Institute National Institutes of Health Department of Health and Human Services
The Family of Cytokines Chief, Laboratory of Molecular Immunology Director, Immunology Center National Heart, Lung, and Blood Institute National Institutes of Health Department of Health and Human Services
Introduction. Introduction. Lymphocyte development (maturation)
 Introduction Abbas Chapter 8: Lymphocyte Development and the Rearrangement and Expression of Antigen Receptor Genes Christina Ciaccio, MD Children s Mercy Hospital January 5, 2009 Lymphocyte development
Introduction Abbas Chapter 8: Lymphocyte Development and the Rearrangement and Expression of Antigen Receptor Genes Christina Ciaccio, MD Children s Mercy Hospital January 5, 2009 Lymphocyte development
Early Thymocyte Development Is Regulated by Modulation of E2A Protein Activity
 Published Online: 10 September, 2001 Supp Info: http://doi.org/10.1084/jem.194.6.733 Downloaded from jem.rupress.org on January 15, 2019 Early Thymocyte Development Is Regulated by Modulation of E2A Protein
Published Online: 10 September, 2001 Supp Info: http://doi.org/10.1084/jem.194.6.733 Downloaded from jem.rupress.org on January 15, 2019 Early Thymocyte Development Is Regulated by Modulation of E2A Protein
Nature Immunology: doi: /ni Supplementary Figure 1. Gene expression profile of CD4 + T cells and CTL responses in Bcl6-deficient mice.
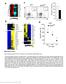 Supplementary Figure 1 Gene expression profile of CD4 + T cells and CTL responses in Bcl6-deficient mice. (a) Gene expression profile in the resting CD4 + T cells were analyzed by an Affymetrix microarray
Supplementary Figure 1 Gene expression profile of CD4 + T cells and CTL responses in Bcl6-deficient mice. (a) Gene expression profile in the resting CD4 + T cells were analyzed by an Affymetrix microarray
Tolerance 2. Regulatory T cells; why tolerance fails. Abul K. Abbas UCSF. FOCiS
 1 Tolerance 2. Regulatory T cells; why tolerance fails Abul K. Abbas UCSF FOCiS 2 Lecture outline Regulatory T cells: functions and clinical relevance Pathogenesis of autoimmunity: why selftolerance fails
1 Tolerance 2. Regulatory T cells; why tolerance fails Abul K. Abbas UCSF FOCiS 2 Lecture outline Regulatory T cells: functions and clinical relevance Pathogenesis of autoimmunity: why selftolerance fails
Tolerance, autoimmunity and the pathogenesis of immunemediated inflammatory diseases. Abul K. Abbas UCSF
 Tolerance, autoimmunity and the pathogenesis of immunemediated inflammatory diseases Abul K. Abbas UCSF Balancing lymphocyte activation and control Activation Effector T cells Tolerance Regulatory T cells
Tolerance, autoimmunity and the pathogenesis of immunemediated inflammatory diseases Abul K. Abbas UCSF Balancing lymphocyte activation and control Activation Effector T cells Tolerance Regulatory T cells
1. Overview of Adaptive Immunity
 Chapter 17A: Adaptive Immunity Part I 1. Overview of Adaptive Immunity 2. T and B Cell Production 3. Antigens & Antigen Presentation 4. Helper T cells 1. Overview of Adaptive Immunity The Nature of Adaptive
Chapter 17A: Adaptive Immunity Part I 1. Overview of Adaptive Immunity 2. T and B Cell Production 3. Antigens & Antigen Presentation 4. Helper T cells 1. Overview of Adaptive Immunity The Nature of Adaptive
The Development of Lymphocytes: B Cell Development in the Bone Marrow & Peripheral Lymphoid Tissue Deborah A. Lebman, Ph.D.
 The Development of Lymphocytes: B Cell Development in the Bone Marrow & Peripheral Lymphoid Tissue Deborah A. Lebman, Ph.D. OBJECTIVES 1. To understand how ordered Ig gene rearrangements lead to the development
The Development of Lymphocytes: B Cell Development in the Bone Marrow & Peripheral Lymphoid Tissue Deborah A. Lebman, Ph.D. OBJECTIVES 1. To understand how ordered Ig gene rearrangements lead to the development
Stochastic component to development of class I major histocompatibility complex-specific T cells
 Proc. Natl. Acad. Sci. USA Vol. 91, pp. 220-224, January 1994 Immunology Stochastic component to development of class I major histocompatibility complex-specific T cells ANDREA ITANO*, DIMITIUS KIOUSSISt,
Proc. Natl. Acad. Sci. USA Vol. 91, pp. 220-224, January 1994 Immunology Stochastic component to development of class I major histocompatibility complex-specific T cells ANDREA ITANO*, DIMITIUS KIOUSSISt,
T Cell Receptor & T Cell Development
 T Cell Receptor & T Cell Development Questions for the next 2 lectures: How do you generate a diverse T cell population with functional TCR rearrangements? How do you generate a T cell population that
T Cell Receptor & T Cell Development Questions for the next 2 lectures: How do you generate a diverse T cell population with functional TCR rearrangements? How do you generate a T cell population that
Prepared by: Dr.Mansour Al-Yazji
 C L L CLL Prepared by: Abd El-Hakeem Abd El-Rahman Abu Naser Ahmed Khamis Abu Warda Ahmed Mohammed Abu Ghaben Bassel Ziad Abu Warda Nedal Mostafa El-Nahhal Dr.Mansour Al-Yazji LEUKEMIA Leukemia is a form
C L L CLL Prepared by: Abd El-Hakeem Abd El-Rahman Abu Naser Ahmed Khamis Abu Warda Ahmed Mohammed Abu Ghaben Bassel Ziad Abu Warda Nedal Mostafa El-Nahhal Dr.Mansour Al-Yazji LEUKEMIA Leukemia is a form
Introduction TRANSPLANTATION. Defu Zeng, Petra Hoffmann, Fengshuo Lan, Philip Huie, John Higgins, and Samuel Strober
 TRANSPLANTATION Unique patterns of surface receptors, cytokine secretion, and immune functions distinguish T cells in the bone marrow from those in the periphery: impact on allogeneic bone marrow transplantation
TRANSPLANTATION Unique patterns of surface receptors, cytokine secretion, and immune functions distinguish T cells in the bone marrow from those in the periphery: impact on allogeneic bone marrow transplantation
Problem Set 8 Key 1 of 8
 7.06 2003 Problem Set 8 Key 1 of 8 7.06 2003 Problem Set 8 Key 1. As a bright MD/PhD, you are interested in questions about the control of cell number in the body. Recently, you've seen three patients
7.06 2003 Problem Set 8 Key 1 of 8 7.06 2003 Problem Set 8 Key 1. As a bright MD/PhD, you are interested in questions about the control of cell number in the body. Recently, you've seen three patients
Supplementary Figure 1. Example of gating strategy
 Supplementary Figure 1. Example of gating strategy Legend Supplementary Figure 1: First, gating is performed to include only single cells (singlets) (A) and CD3+ cells (B). After gating on the lymphocyte
Supplementary Figure 1. Example of gating strategy Legend Supplementary Figure 1: First, gating is performed to include only single cells (singlets) (A) and CD3+ cells (B). After gating on the lymphocyte
of whole cell cultures in U-bottomed wells of a 96-well plate are shown. 2
 Supplementary online material Supplementary figure legends Supplementary Figure 1 Exposure to T reg cells causes loss of T resp cells in co-cultures. T resp cells were stimulated with CD3+CD28 alone or
Supplementary online material Supplementary figure legends Supplementary Figure 1 Exposure to T reg cells causes loss of T resp cells in co-cultures. T resp cells were stimulated with CD3+CD28 alone or
VUmc Basispresentatie
 Clinical diagnostic cytometry Gerrit J Schuurhuis Dept of Hematology VU University Medical Center Amsterdam, Netherlands Use of immunophenotyping at diagnosis to trace residual disease after therapy 1.
Clinical diagnostic cytometry Gerrit J Schuurhuis Dept of Hematology VU University Medical Center Amsterdam, Netherlands Use of immunophenotyping at diagnosis to trace residual disease after therapy 1.
Effector mechanisms of cell-mediated immunity: Properties of effector, memory and regulatory T cells
 ICI Basic Immunology course Effector mechanisms of cell-mediated immunity: Properties of effector, memory and regulatory T cells Abul K. Abbas, MD UCSF Stages in the development of T cell responses: induction
ICI Basic Immunology course Effector mechanisms of cell-mediated immunity: Properties of effector, memory and regulatory T cells Abul K. Abbas, MD UCSF Stages in the development of T cell responses: induction
Goals. Goals. From Bench to Cageside: Immunology of Humanizing Mice
 From Bench to Cageside: Immunology of Humanizing Mice Zachary T Freeman Goals Understand relevant immunology related to humanized mice Understand three main components that contribute to formation of humanized
From Bench to Cageside: Immunology of Humanizing Mice Zachary T Freeman Goals Understand relevant immunology related to humanized mice Understand three main components that contribute to formation of humanized
From Bench to Cageside: Immunology of Humanizing Mice. Zachary T Freeman
 From Bench to Cageside: Immunology of Humanizing Mice Zachary T Freeman Goals Understand relevant immunology related to humanized mice Understand three main components that contribute to formation of humanized
From Bench to Cageside: Immunology of Humanizing Mice Zachary T Freeman Goals Understand relevant immunology related to humanized mice Understand three main components that contribute to formation of humanized
Helminth worm, Schistosomiasis Trypanosomes, sleeping sickness Pneumocystis carinii. Ringworm fungus HIV Influenza
 Helminth worm, Schistosomiasis Trypanosomes, sleeping sickness Pneumocystis carinii Ringworm fungus HIV Influenza Candida Staph aureus Mycobacterium tuberculosis Listeria Salmonella Streptococcus Levels
Helminth worm, Schistosomiasis Trypanosomes, sleeping sickness Pneumocystis carinii Ringworm fungus HIV Influenza Candida Staph aureus Mycobacterium tuberculosis Listeria Salmonella Streptococcus Levels
Additional file 6. Summary of experiments characterizing development of adaptive immune response
 Additional file 6. Summary of experiments characterizing development of adaptive immune response Tests performed to evaluate the development of adaptive immunity in patients are summarized in Table 1.
Additional file 6. Summary of experiments characterizing development of adaptive immune response Tests performed to evaluate the development of adaptive immunity in patients are summarized in Table 1.
Supplementary Figure 1. Successful excision of genes from WBM lysates and
 Supplementary Information: Supplementary Figure 1. Successful excision of genes from WBM lysates and survival of mice with different genotypes. (a) The proper excision of Pten, p110α, p110α and p110δ was
Supplementary Information: Supplementary Figure 1. Successful excision of genes from WBM lysates and survival of mice with different genotypes. (a) The proper excision of Pten, p110α, p110α and p110δ was
CHAPTER 9 BIOLOGY OF THE T LYMPHOCYTE
 CHAPTER 9 BIOLOGY OF THE T LYMPHOCYTE Coico, R., Sunshine, G., (2009) Immunology : a short course, 6 th Ed., Wiley-Blackwell 1 CHAPTER 9 : Biology of The T Lymphocytes 1. 2. 3. 4. 5. 6. 7. Introduction
CHAPTER 9 BIOLOGY OF THE T LYMPHOCYTE Coico, R., Sunshine, G., (2009) Immunology : a short course, 6 th Ed., Wiley-Blackwell 1 CHAPTER 9 : Biology of The T Lymphocytes 1. 2. 3. 4. 5. 6. 7. Introduction
Generation of ST2-GFP reporter mice and characterization of ILC1 cells following infection
 Supplementary Figure 1 Generation of ST2-GFP reporter mice and characterization of ILC1 cells following infection with influenza virus. (a) ST2-GFP reporter mice were generated as described in Methods.
Supplementary Figure 1 Generation of ST2-GFP reporter mice and characterization of ILC1 cells following infection with influenza virus. (a) ST2-GFP reporter mice were generated as described in Methods.
Supplementary Figure S1. PTPN2 levels are not altered in proliferating CD8+ T cells. Lymph node (LN) CD8+ T cells from C57BL/6 mice were stained with
 Supplementary Figure S1. PTPN2 levels are not altered in proliferating CD8+ T cells. Lymph node (LN) CD8+ T cells from C57BL/6 mice were stained with CFSE and stimulated with plate-bound α-cd3ε (10µg/ml)
Supplementary Figure S1. PTPN2 levels are not altered in proliferating CD8+ T cells. Lymph node (LN) CD8+ T cells from C57BL/6 mice were stained with CFSE and stimulated with plate-bound α-cd3ε (10µg/ml)
Scientific report: Delineating cellular stages and regulation of human NK cell development to improve NK cell-based therapy for cancer (Dnr )
 Scientific report: Delineating cellular stages and regulation of human NK cell development to improve NK cell-based therapy for cancer (Dnr 130259) The main goal of this project focuses on establishing
Scientific report: Delineating cellular stages and regulation of human NK cell development to improve NK cell-based therapy for cancer (Dnr 130259) The main goal of this project focuses on establishing
Increased IL-12 induced STAT-4 signaling in CD8 T cells. from aged mice
 Increased IL-2 induced STAT-4 signaling in CD8 T cells from aged mice Erin Rottinghaus * Abstract: Aging is associated with poor immune function leading to increased susceptibility to infectious diseases
Increased IL-2 induced STAT-4 signaling in CD8 T cells from aged mice Erin Rottinghaus * Abstract: Aging is associated with poor immune function leading to increased susceptibility to infectious diseases
Chapter 11. B cell generation, Activation, and Differentiation. Pro-B cells. - B cells mature in the bone marrow.
 Chapter B cell generation, Activation, and Differentiation - B cells mature in the bone marrow. - B cells proceed through a number of distinct maturational stages: ) Pro-B cell ) Pre-B cell ) Immature
Chapter B cell generation, Activation, and Differentiation - B cells mature in the bone marrow. - B cells proceed through a number of distinct maturational stages: ) Pro-B cell ) Pre-B cell ) Immature
Immune Regulation and Tolerance
 Immune Regulation and Tolerance Immunoregulation: A balance between activation and suppression of effector cells to achieve an efficient immune response without damaging the host. Activation (immunity)
Immune Regulation and Tolerance Immunoregulation: A balance between activation and suppression of effector cells to achieve an efficient immune response without damaging the host. Activation (immunity)
A Slfn2 mutation causes lymphoid and myeloid immunodeficiency due to loss of immune cell quiescence
 Supplementary Information A Slfn mutation causes lymphoid and myeloid immunodeficiency due to loss of immune cell quiescence Michael Berger, Philippe Kres, Karine Crozat, Xiaohong Li, Ben A. Croker, Owen
Supplementary Information A Slfn mutation causes lymphoid and myeloid immunodeficiency due to loss of immune cell quiescence Michael Berger, Philippe Kres, Karine Crozat, Xiaohong Li, Ben A. Croker, Owen
Interferon γ regulates idiopathic pneumonia syndrome, a. Th17 + CD4 + T-cell-mediated GvH disease
 Interferon γ regulates idiopathic pneumonia syndrome, a Th17 + CD4 + T-cell-mediated GvH disease Nora Mauermann, Julia Burian, Christophe von Garnier, Stefan Dirnhofer, Davide Germano, Christine Schuett,
Interferon γ regulates idiopathic pneumonia syndrome, a Th17 + CD4 + T-cell-mediated GvH disease Nora Mauermann, Julia Burian, Christophe von Garnier, Stefan Dirnhofer, Davide Germano, Christine Schuett,
Cover Page. The handle holds various files of this Leiden University dissertation.
 Cover Page The handle http://hdl.handle.net/1887/23854 holds various files of this Leiden University dissertation. Author: Marel, Sander van der Title: Gene and cell therapy based treatment strategies
Cover Page The handle http://hdl.handle.net/1887/23854 holds various files of this Leiden University dissertation. Author: Marel, Sander van der Title: Gene and cell therapy based treatment strategies
