Localization of P2X and P2Y Receptors in Dorsal Root Ganglia of the Cat
|
|
|
- Marjorie Turner
- 5 years ago
- Views:
Transcription
1 Volume 53(10): , 2005 Journal of Histochemistry & Cytochemistry ARTICLE Localization of P2X and P2Y Receptors in Dorsal Root Ganglia of the Cat Huai-Zhen Ruan, Lori A. Birder, William C. de Groat, Changfeng Tai, James Roppolo, Charles A. Buffington, and Geoffrey Burnstock Autonomic Neuroscience Centre, Royal Free & University College Medical School, London, United Kingdom (H-ZR,GB); Department of Neurobiology, Third Military Medical University, Chongqing, China (H-ZR); University of Pittsburgh School of Medicine, Departments of Medicine (LAB) and Pharmacology (LAB,WCdG,CT,JR), Pittsburgh, Pennsylvania; and Ohio State University Department of Clinical Veterinary Sciences, Columbus Ohio (CAB) SUMMARY The distribution of P2X and P2Y receptor subtypes in upper lumbosacral cat dorsal root ganglia (DRG) has been investigated using immunohistochemistry. Intensity of immunoreactivity for six P2X receptors (P2X 5 receptors were immuno-negative) and the three P2Y receptors examined in cat DRG was in the order of P2Y 2 P2Y 4 P2X 3 P2X 2 P2X 7 P2X 6 P2X 1 P2X 4 P2Y 1. P2X 3, P2Y 2, and P2Y 4 receptor polyclonal antibodies stained 33.8%, 35.3%, and 47.6% of DRG neurons, respectively. Most P2Y 2, P2X 1, P2X 3, P2X 4, and P2X 6 receptor staining was detected in small- and medium-diameter neurons. However, P2Y 4, P2X 2, and P2X 7 staining was present in large- and small-diameter neurons. Doublelabeling immunohistochemistry showed that 90.8%, 32.1%, and 2.4% of P2X 3 receptorpositive neurons coexpressed IB 4, CGRP, and NF200, respectively; whereas 67.4%, 41.3%, and 39.1% of P2Y 4 receptor-positive neurons coexpressed IB 4, CGRP, and NF200, respectively. A total of 18.8%, 16.6%, and 63.5% of P2Y 2 receptor-positive neurons also stained for IB 4, CGRP, and NF200, respectively. Only 30% of DRG neurons in cat were P2X 3 -immunoreactive compared with 90% in rat and in mouse. A further difference was the low expression of P2Y 1 receptors in cat DRG neurons compared with more than 80% of the neurons in rat. Many small-diameter neurons were NF200-positive in cat, again differing from rat and mouse. (J Histochem Cytochem 53: , 2005) Correspondence to: Prof. Geoffrey Burnstock, Autonomic Neuroscience Centre, Royal Free & University College Medical School, Rowland Hill Street, London NW3 2PF, United Kingdom. g.burnstock@ucl.ac.uk Received for publication October 18, 2004; accepted May 4, 2005 [DOI: /jhc.4A ]. KEY WORDS purinoceptors P2X receptor P2Y receptor ATP dorsal root ganglion cat ATP receptors have been classified into two subfamilies, the ion-gated (ionotropic) P2X receptors and the G protein coupled (metabotropic) P2Y receptors (Burnstock and Kennedy 1985; Abbracchio and Burnstock 1994). P2X receptors are highly expressed on both peripheral and central terminals of primary afferent fibers (Vulchanova et al. 1997,1998; Bradbury et al. 1998; Guo et al. 1999; Novakovic et al. 1999; Dunn et al. 2001). Activation of P2X receptors at peripheral sensory nerve endings may initiate sensory impulses, and may be associated with different sensory modalities, including nociceptive and nonnociceptive sensory signals (Cook et al. 1997; Dowd et al. 1998; Burnstock 2000; Tsuda et al. 2000). Further, activation of presynaptic P2X receptors on primary afferent terminals in the spinal cord evoked glutamate release onto dorsal horn neurons (Gu and MacDermott 1997), suggesting a role for ATP in modulating sensory synaptic transmission in the central nervous system. To date, seven distinct P2X subunits (P2X 1 7 ) have been cloned (Chen et al. 1995; Collo et al. 1996; Khakh et al. 2000) and have been found to be expressed in primary sensory neurons (Xiang et al. 1998; Dunn et al. 2001; Ueno et al. 2002). In the rat and mouse, high levels of P2X 3 receptor expression have been detected in a subpopulation of primary afferent neurons, including those of dorsal root ganglia (DRG), trigeminal ganglia, and nodose ganglia (Chen et al. 1995; Cook et al. 1997; Vulchanova et al. 1997,1998; Bradbury et The Histochemical Society, Inc /05/$
2 1274 Ruan, Birder, de Groat, Tai, Roppolo, Buffington, Burnstock al. 1998; Zhong et al. 2001). In addition to P2X 3, low levels of mrna transcripts and proteins for P2X 1,2, P2X 4 6 have been identified in sensory neurons of the rat (Collo et al. 1996; Xiang et al. 1998). So far, eight mammalian P2Y receptors P2Y 1, P2Y 2, P2Y 4, P2Y 6, P2Y 11, P2Y 12 (Hollopeter et al. 2001; Zhang et al. 2001), P2Y 13 (Communi et al. 2001), and the more recent P2Y 14 receptor (Abbracchio et al. 2003) have been cloned and shown to be activated by extracellular nucleotides. These receptors share between 25% and 55% identity (at the amino acid level) and can be distinguished by their specific pharmacological selectivities for both purine and pyrimidine nucleotides (King and Burnstock 2002). Among the eight known P2Y receptors, mrna for P2Y 1 and P2Y 2 is expressed at high levels in rat DRG sensory neurons (Nakamura and Strittmatter 1996; Molliver et al. 2002). Low levels of mrna for P2Y 4 and P2Y 6 have been reported in whole DRG of rat, but neither receptor has been localized to sensory neurons (Sanada et al. 2002). We have also shown recently that P2Y 1, P2Y 2, P2Y 4, and P2Y 6 receptor mrna is expressed in rat sensory ganglia (DRG) and nodose and trigeminal ganglia (Ruan and Burnstock 2003). P2Y 1 receptor polyclonal antibodies stained more than 80% of the sensory neurons, particularly the small-diameter (neurofilament-negative) neurons. In contrast, the P2Y 4 receptor antibody stained more medium- and largediameter (neurofilament-positive) neurons than smalldiameter neurons. The P2Y 1 receptor has been identified on adult rat primary sensory neurons, where it has been implicated in the transduction of mechanical stimuli (Nakamura and Strittmatter 1996) and modulation of the capsaicin receptor, TRPV1 (Tominaga et al. 2001). P2Y 2 receptor activation initiates sustained action potential firing and induces phosphorylation of the transcription factor CREB in isolated sensory neurons that have neurochemical characteristics of nociceptors (Molliver et al. 2002). Furthermore, stimulation of P2Y receptors increases intracellular calcium in isolated sensory neurons and facilitates release of the neuropeptide calcitonin gene-related peptide (CGRP) in vitro (Sanada et al. 2002; Zimmermann et al. 2002). Together, these studies suggest that purines and pyrimidines have multiple actions on sensory neurons mediated by both P2X and P2Y receptors; the challenge remains to delineate which actions have functional importance under various physiological and pathological conditions. The majority of studies of purinergic signaling in mammalian sensory neurons has been carried out in rat and mouse (see Dunn et al. 2001). However, there are no reports of the distribution of the P2X and P2Y receptor protein in DRG of cat, although many important physiological studies have been carried out in the cat sensory nerves (Thor et al. 1989a,b; de Groat 1999; Jänig and Häbler 1999). In previous studies, we have described a painful bladder condition in cats termed feline interstitial cystitis, which is associated with a change in the chemical and electrophysiological properties of sacral sensory neurons (Buffington et al. 1999; de Groat et al. 2001). We also detected changes in the expression of purinergic receptors in the urinary bladder of cats with feline interstitial cystitis (Birder et al. 2004). This study was undertaken to examine the distribution of P2X and P2Y receptors in DRG of normal cats to provide background information for future studies with these receptors in DRG of cats with feline interstitial cystitis. The DRG comprises functionally heterogeneous cell types, and the size (diameter) of their cell bodies closely correlates with their functions; a subpopulation of small-size neurons transmit nociceptive information (Bessou et al. 1971), whereas large-size neurons have been identified as being involved in the transmission of proprioceptive and tactile impulses (Nakamura and Strittmatter 1996). To examine the potential roles of P2 receptor subunits in sensory functions, we determined the subtype expression patterns of the P2X and P2Y receptors in combination with cytochemical markers of sensory neuron populations. We chose to examine the expression of the P2X 3, P2Y 2, and P2Y 4 receptors in relation to the neuropeptide CGRP, as well as binding of the isolectin B 4 from Griffonia simplicifolia type one (IB 4 ). Small-diameter DRG neurons and associated A - and C-fiber afferents are generally considered as nociceptors. Small neurons can be broadly subdivided into two groups based on regulatory growth factors and the presence of neuropeptides; the first group contains, among other peptides, CGRP; the second class possess cell surface glycoconjugates that bind IB 4 (Silverman and Kruger 1990; Alvarez et al. 1991), and because there is limited overlap between the two groups in terms of CGRP and IB 4 staining, are thought to be distinct subgroups of nociceptors. In contrast the antineurofilament antibody neurofilament-200 stains classically defined large neurons (see Bradbury et al. 1998; Wu and Pan 2004). Thus these three markers are useful tools for distinguishing different subsets of sensory neurons. To examine the potential roles of P2 receptor subunits on the function of sensory neurons, the subtype expression pattern of P2 receptors was examined in combination with cytochemical markers of different subsets of sensory neurons. Materials and Methods Animals and Tissue Preparation All procedures were conducted in accordance with Institutional Animal Care and Use Committee policies at each institution [Ohio State University, University of Pittsburgh, and
3 P2X and P2Y Receptors in Cat DRG 1275 Home Office (UK) regulations covering regulated procedures]. Healthy, age-matched cats obtained from commercial suppliers were evaluated at Ohio State University and determined to be free from disease and signs referable to the lower urinary tract. All cats were housed in stainless steel cages at the Ohio State University animal facilities and allowed to acclimatize to their environment for at least 3 months before study. Upper sacral (S 1,S 2 ) DRG were dissected from deeply anaesthetized (induction with 2% halothane and then maintained with -chloralose mg/kg) cats (n 3 of either sex), and after removal of tissue, the animals were sacrificed by an overdose of the anesthesia. The degree of anesthesia was determined to be adequate for surgery by periodically testing for the absence of a withdrawal reflex to a strong pinch of a hind paw and absence of an eye blink reflex to tactile stimulation of the cornea. DRG were fixed in 4% paraformaldehyde in 0.1 M phosphate buffer (ph 7.2) at 4C overnight, and then transferred to 20% sucrose in PBS (overnight at 4C). Thereafter, the tissue blocks were rapidly frozen by immersion in isopentane at 70C for 2 min. Transverse sections through the ganglia (10 m thickness) were cut on a cryostat and thaw-mounted on poly-l-lysine coated slides. Antisera The following primary antisera were used in the current studies: rabbit anti-p2y 1, -P2Y 2, and -P2Y 4 receptor antibodies (Alomone Labs; Jerusalem, Israel, 3 g/ml). The immunogens used for production of polyclonal P2Y 1, P2Y 2, and P2Y 4 antibody were synthetic peptides corresponding to the carboxyl terminal of the cloned rat P2Y 1, P2Y 2, and P2Y 4 receptors, covalently linked to keyhole limpet hemocyanin. The peptide sequences of the P2Y 1, P2Y 2, and P2Y 4 receptors are of amino acid sequence (RALIYKDLDNS- PLRRKS), (KPAYGTTGLPRAKRKSVR), and (HEESISRWADTHQD), respectively. Rabbit anti- P2X 1 P2X 7 receptor antibodies (3 g/ml) were provided by Roche (Palo Alto, CA) (Xiang et al. 1998). Other antibodies used were: mouse anti-neurofilament 200 (NF200, clone N52; Sigma Chemical Co., Poole, UK, 1:400), mouse anti-cgrp (Affiniti Research Products Ltd., Exeter, UK; 1:2000). Biotin-conjugated isolectin B 4 from Griffonia simplicifolia (IB 4 ) was obtained from Sigma (10 g/ml). Immunostaining An indirect immunofluorescence method was used to visualize receptor expression. Briefly, the sections (eight per animal) were washed in PBS (3 5 min) and nonspecific binding was blocked by preincubation were 10% normal horse serum in PBS containing 0.2% Triton X-100 for 30 min. The sections were incubated overnight with the primary antibodies (P2X 1 P2X 7, P2Y 1, P2Y 2, and P2Y 4 ) diluted to 3 g/ml with 10% normal horse serum in PBS containing 0.05% merthiolate and 0.2% Triton X-100. Subsequently, the sections were incubated with Cy3-conjugated donkey anti-rabbit IgG, diluted 1:300 in 1% normal horse serum in PBS containing 0.05% merthiolate for 1 hr at room temperature. Slides were mounted with Citifluor (Citifluor Ltd; London, UK) and examined with fluorescence microscopy. All control experiments were performed using an excess of the appropriate homolog peptide antigen to absorb the primary antibodies and thus confirm a specific immunoreaction or omission of either the primary or secondary antibody from the staining procedure. The sections on which counts of P2X and P2Y receptor positive neurons had been performed were marked and then counterstained with toluidine blue (2.5% in 0.1 M phosphate buffer for 2 min, followed by dehydration through increasing grades of alcohol, cleared in xylene, and cover slipped with DPX mounting medium). This enabled the total neuron numbers to be determined by counting all neurons in the marked sections under bright-field illumination. The number of immunopositive neurons in a marked section was counted and divided by the total number of neurons, to give a percentage. A mean SE mean from the sections was then calculated. Immunofluorescence Double Labeling To demonstrate colocalization of the P2X 3, P2Y 2, and P2Y 4 receptors with a high molecular-weight neurofilament marker (NF200) or CGRP, sections were incubated with P2X 3, P2Y 2, or P2Y 4 antibody overnight and detected with Cy3-conjugated donkey anti-rabbit IgG, then incubated with monoclonal NF200 antibody (1:400; overnight) or monoclonal CGRP antibody (1:2000; overnight) and FITC-conjugated mouse antibody (raised in goat) (Sigma; 1:200, 1 hr). For colocalization with the IB 4 sections were immunostained for the P2X 3, P2Y 2, and P2Y 4 receptor as mentioned previously and then incubated with biotin-conjugated IB 4 (10 g/ml, 16 hr), and streptavidin-fitc (1:200; 1 hr). The sections were washed and mounted in Citifluor. Photomicroscopy Images of immunofluorescence labeling were taken with a Leica DC 200 digital camera (Leica; Heerbrugg, Switzerland) using the automatic exposure setting, attached to a Zeiss Axioplan microscope (Zeiss; Oberkochen, Germany), fitted with the following filters: Cy3, nm excitation, 590 nm; FITC, 470 nm excitation, and 525 nm emission. Images were imported into a graphics package (Adobe Photoshop 5.0; San Jose, CA), where images were not manipulated with the exception of producing multiple images per figure, the addition of text and labeling, and for the double-labeling studies in which the individual images of the single receptors stained green or red are merged to reveal areas of colocalization in yellow/orange. Analysis All analyses were performed at 20 objective magnification. P2X and P2Y receptor expression in ganglia was determined by counting all P2X and P2Y receptor-positive neurons in every sixth section throughout the ganglia (per animal). A total of three individual observers independently judged the slides for positive/negative immunoreactivity. The number of neurons (only counted if a nucleus and cytoplasm was present) immunopositive for each P2 receptor subtype was counted for each section and this number was divided by the total number of neurons per section to give a percentage number of cells immunopositive. The mean percentages SE mean were calculated for each P2 receptor subtype.
4 1276 Ruan, Birder, de Groat, Tai, Roppolo, Buffington, Burnstock To calculate percentages of P2X 3, P2Y 2, and P2Y 4 receptor colocalization with cytochemical markers, four randomly selected ganglia sections were chosen for each pair of markers for each animal. For each section, counts were made of the number of positive neurons for P2X 3, P2Y 2, or P2Y 4 receptors, the number of positive neurons for the other marker (NF200, IB 4, or CGRP) and the number of neurons expressing both antigens and percentages were calculated ( SE mean). Results P2X and P2Y Receptor-staining in DRG of Cat The polyclonal antibodies for the seven P2X and three P2Y receptor subtypes labeled different subpopulations of neurons (with the exception of P2X 5 ) (Figure 1) and the number of neurons positive for the different P2 receptor subtypes expressed as a percentage of the total number on neurons counted ( SE mean) are given in Table 1. In control experiments, no signal was observed when the preimmune sera were used. All P2X receptor antibodies, except that for the P2X 5 subtype, labeled neurons of cat DRG. P2X 3 polyclonal antibodies moderately stained 33.8% of the neurons (Table 1; Figure 1C). The expression of P2X 2 and P2X 7 receptors (Figures 1B and 1G) in DRG was lower than that of P2X 3 receptors, but higher than that of P2X 1, P2X 4, or P2X 6 receptors (Table 1; Figures 1A, 1D, and 1F). Most P2X 1, P2X 3, P2X 4, and P2X 6 receptor staining was detected in small- and medium-diameter neurons. However, P2X 2 and P2X 7 polyclonal antibodies stained not only small-diameter neurons, but also large-diameter neurons. No immunoreactivity was observed for P2X 5 receptors (Figure 1E). The immunoreactivity intensity of the six P2X receptors in cat DRG was in the order of P2X 3 P2X 2 P2X 7 P2X 6 P2X 1 P2X 4. Neurons in DRG also expressed P2Y 2 and P2Y 4 receptor subtypes (Table 1; Figure 1). P2Y 2 and P2Y 4 receptor antibodies strongly stained 35.3% and 47.6% Figure 1 Localization of P2X and P2Y receptor immunoreactivity (IR) in cat dorsal root ganglia (DRG). (A) P2X 1 -IR. (B) P2X 2 -IR. (C) P2X 3 -IR. (D) P2X 4 -IR. (E) P2X 5 -IR. (F) P2X 6 -IR. (G) P2X 7 -IR. (H) P2Y 1 -IR. (I) P2Y 2 -IR. (J,K) P2Y 4 -IR. Bar 50 m.
5 P2X and P2Y Receptors in Cat DRG 1277 Table 1 Expression of P2X 1 7 and P2Y 1,2,4 receptor immunoreactivity in cat dorsal root ganglion (DRG) % P2X 1 7 and P2Y 1,2,4 receptor immunopositive cell profiles Receptor P2X 1 P2X 2 P2X 3 P2X 4 P2X 5 DRG n 569/ / / /3012 Receptor P2X 6 P2X 7 P2Y 1 P2Y 2 P2Y 4 DRG n 678/ / / / /2942 n, number of P2X 1 7- or P2Y 1,2,4-positive cell profiles and the total number of cell profiles counted. of the neurons, respectively. The staining was evenly distributed throughout the cytoplasm of these cells and positively stained cells were randomly distributed throughout the ganglia. P2Y 2 receptor staining was localized to the small-diameter neurons (Figure 1I). P2Y 4 antibodies stained not only small-diameter neurons, but also some large-diameter neurons (Figures 1J and 1K). The expression of P2Y 1 receptors was quite low in cat DRG, but did label glial cells of the DRG (Figure 1H). Coexpression of P2X 3, P2Y 2, and P2Y 4 Receptors and Other Neuronal Markers P2X 3, P2Y 2, and P2Y 4 /IB 4. To further identify the sensory neurons that express the most prominent purinergic receptors (P2X 3, P2Y 2, and P2Y 4 ), a number of established markers can be used. IB 4 has also been used as a marker for fluoride-resistant acid phosphatase (FRAP) containing neurons. These are important markers because several lines of evidence suggest that they identify two subpopulations of putative nociceptors in DRG. P2X 3, P2Y 2, and P2Y 4 receptors were expressed in many IB 4 -positive cells, 88.2%, 16.5%, and 87.4%, respectively (Table 2; Figure 2); that is, 90.8%, 18.8%, and 67.4% of P2X 3, P2Y 2, and P2Y 4 receptor-immunoreactive neurons contain IB 4 in cat DRG, respectively. P2X 3, P2Y 2, and P2Y 4 /CGRP. A double immunofluorescence method revealed 32.1%, 16.6%, and 41.3% of neurons that were positive for P2X 3, P2Y 2, and P2Y 4 receptor exhibited CGRP-immunoreactivity, respectively. Conversely, 26.5%, 13.7%, and 46.3% of CGRP-immunoreactive neurons exhibited P2X 3, P2Y 2, and P2Y 4 receptor immunoreactivity, respectively (Table 2; Figure 2). P2X 3, P2Y 2, and P2Y 4 /NF200. NF200 is an anti-neurofilament antibody that has been shown to be a reliable indicator of the myelination state of sensory neurons (Lawson et al., 1984; Lawson and Waddell, 1991). In the rat, it stains the classically defined large-diameter neurons with myelinated axons (Dunn et al. 2001). Costaining for P2X 3, P2Y 2, and P2Y 4 with NF200 was performed to examine the extent to which P2X 3, P2Y 2, and P2Y 4 may be expressed in neurons with myelinated axons. Double immunofluorescence showed that 2.4%, 63.5%, and 39.1% of P2X 3, P2Y 2, and P2Y 4 receptor neuron profiles were also stained for NF200- immunoreactivity, respectively. Conversely, 1.7%, 40.3%, and 28.8% of NF200-immunoreactive neurons coexpressed P2X 3, P2Y 2, and P2Y 4 receptor immunoreactivity, respectively (Table 2; Figure 2). Discussion In this study, we have used immunohistochemistry to study the expression of P2X and P2Y receptors in cat DRG. The results showed that neurons in the DRG express a variety of P2X and P2Y receptor subtypes. Differences in expression level of each receptor subtype exist in different subpopulations of the neurons, which suggests that the diversity of receptor subtypes may be associated with different physiological functions of different neurons. P2X receptors are ligand-gated ion channels activated by extracellular ATP that mediate rapid cation permeability and fast excitatory neurotransmission in both the central and peripheral nervous systems (reviewed by Ralevic and Burnstock 1998). DRG of rat and mouse are well studied in regard to the function of P2X receptors (see Dunn et al. 2000). ATP depolarizes DRG neurons by eliciting fast- and slow-inactivat- Table 2 Colocalization of P2X 3, or P2Y 2 or P2Y 4 receptor immunoreactivity with IB4, or CGRP, or NF200 in cat dorsal root ganglion (DRG) IB 4 -, CGRP-, or NF200-positive neurons expressing P2 receptor positive neurons expressing P2 receptors IB 4 CGRP NF200 IB 4 CGRP NF200 P2X n 877/ /903 22/ / / /1294 P2Y n 189/ / / / / /1625 P2Y n 892/ / / / / /1679 IB 4, isolectin B 4; CGRP, calcitonin gene-related peptide; NF200, high molecular-weight neurofilament; n, number of double-labeled cell profiles and the total number of cell profiles counted, respectively, for each combination of receptor.
6 1278 Ruan, Birder, de Groat, Tai, Roppolo, Buffington, Burnstock Figure 2 Colocalization (yellow/orange) of P2X 3, or P2Y 2 or P2Y 4 receptor immunoreactivity (IR) (red) with IB 4, or CGRP or NF200 (green) in the cat dorsal root ganglia (DRG). (A C) P2X 3 -IR and IB 4. (D) P2X 3 -IR and CGRP-IR. (E) P2X 3 -IR and NF200-IR. (F) P2Y 2 -IR and IB 4. (G) P2Y 2 -IR and CGRP-IR. (H) P2Y 2 -IR and NF200-IR. (I) P2Y 4 -IR and IB 4. (J) P2Y 4 -IR and CGRP- IR. (K) P2Y 4 -IR and NF200-IR. Bar 50 m. ing inward currents. The fast-inactivating ATP currents are mediated by homomeric P2X 3 receptors; the slow-desensitizing currents are mediated by heteromeric P2X 2/3 receptors (Bradbury et al. 1998; Burgard et al. 1999). After the cloning of P2X 3 receptors from DRG (Chen et al. 1995; Lewis et al. 1995), more attention has been paid to these ganglia because of the possible involvement of P2X receptors in nociception (Burnstock and Wood 1996; Burnstock 2001). We found that 33% of cat DRG neurons were P2X 3 - immunoreactive with moderate intensity staining. The P2X 3 -immunoreactive neurons were almost exclusively small to medium diameter in size. In a study of P2X 3 -receptor staining of the rat DRG, 90% of the total number of neurons were found to be immunopositive for P2X 3 receptors to a greater or lesser degree, although the small- and medium-diameter neurons were the most intensely stained (Xiang et al. 1998). The percentage of cells that this population represented was not mentioned. In a separate study of P2X 3 receptor immunoreactivity of the rat DGR, a similar percentage of neurons were also stained for P2X 3 receptors
7 P2X and P2Y Receptors in Cat DRG 1279 (Bradbury et al. 1998). In addition to P2X 3 receptors, mrna transcripts and proteins for P2X 1, P2X 2, P2X 4, P2X 5, P2X 6, and P2X 7 receptors have also been detected in rat DRG (Xiang et al. 1998; Ueno et al. 2002). In our present study of cat DRG, immunoreactivity of P2X 1 7 receptor subtypes, except the P2X 5 receptor, was shown to be present. The lack of immunoreactivity to P2X 5 -receptor antibodies might reflect that this antibody does not recognize cat proteins. However, this antibody has been successfully used to visualize P2X 5 receptors in the cat bladder urothelium (Birder et al. 2004), suggesting that the lack of staining for P2X 5 receptors in the DRG is the result of an absence of receptors. ATP, in addition to other purines and pyrimidines, also evokes responses in nociceptors through P2Y metabotropic receptors. The P2Y receptors couple through G proteins to various second messenger pathways mediating slower metabotropic responses. Among the eight known P2Y receptors, mrna for P2Y 1 and P2Y 2 are expressed at high levels in rat DRG neurons (Nakamura and Strittmatter 1996; Molliver et al. 2002). Low levels of mrna for P2Y 4 and P2Y 6 have been reported in whole DRG of rat, but neither receptor has been localized to sensory neurons (Sanada et al. 2002). We have also shown recently that P2Y 1, P2Y 2, P2Y 4, and P2Y 6 receptor mrna is expressed in rat sensory ganglia (DRG, nodose ganglia, and trigeminal ganglia; Ruan and Burnstock 2003). P2Y 1 receptor polyclonal antibodies stained more than 80% of the sensory neurons, particularly the small-diameter (neurofilamentnegative) neurons. The P2Y 4 receptor antibody stained more medium- and large-diameter (neurofilament-positive) neurons than small-diameter neurons. In the present study, P2Y 2 and P2Y 4 receptor polyclonal antibodies stained 35% and 47% of the neurons, respectively, and labeled the neurons very strongly. However, the expression of P2Y 1 receptors was quite low in cat DRG neurons, which differs from the results obtained in rat DRG using in situ hybridization and RT-PCR (Nakamura and Strittmatter 1996; Molliver et al. 2002; Sanada et al. 2002) or immunohistochemistry (Ruan and Burnstock, 2003). To examine the potential roles of P2 receptor subtypes in sensory function, we determined the expression pattern of subtypes in combination with cytochemical markers of sensory neuron populations. We chose to examine the expression of the P2X 3, P2Y 2, and P2Y 4 receptors in relation to CGRP, IB 4, and NF200. In the rat, monkey, and human, information about tissue damage is normally transmitted from the periphery to the spinal cord via the classically defined small dark cells of the DRG, which comprise 70% of all lumbar DRG neurons (Snider and McMahon 1998). These cells predominantly have unmyelinated C-fiber axons. Electrophysiological studies have shown that most of these cells ( 90%) are nociceptive in function (Snider and McMahon 1998). These small cells can be further subdivided into two minimally overlapping groups based on anatomical and functional properties. About one-half of the C-fiber population does not express NGF receptors (McMahon et al. 1994; Averill et al. 1995), but can be identified by a number of markers: they bind the lectin IB 4, express the enzyme TMP, and stain with the antibody LA4 (Bradbury et al. 1998; Petruska et al. 2000). They also express the heat-responsive vanilloid receptor, TRPV1 (Caterina et al. 1997), which implicates them in thermal nociception. The central terminals of these neurons project to lamina IIi of the superficial dorsal horn of the spinal cord, a region where many fine afferents terminate. In vivo studies demonstrated that IB 4 binding sites are selectively expressed by nociceptive afferents (Gerke and Plenderleith 2001). Physiological studies demonstrated that some IB 4 -binding DRG neurons innervate visceral organs (Yoshimura et al. 2003) and therefore may be high-threshold mechanoreceptors or polymodal nociceptors. The other population of C-fiber afferent neurons expresses the NGF receptors trka and p75. Virtually this entire group contains the neuropeptide CGRP, and other neuropeptides, such as substance P, are also found in this population. The central terminals of these neurons project to lamina I/IIo of the superficial dorsal horn of the spinal cord, a region where many fine afferents terminate. The large cells within the cat sensory ganglia were rich in neurofilament. These cells were identified with the antibody neurofilament 200 (NF200). NF200 is a marker of A-fiber sensory neurons in the rat (Perry et al. 1991). A -fibers together with C-fibers are capable of detecting noxious stimuli and initiating pain sensation and are considered as nociceptors (see Wu and Pan 2004). The normal pain threshold in humans is the threshold of A -fiber nociceptors. The large-diameter neurons that are neurofilament-positive are known to possess myelinated axons and to be predominantly responsive to mechanical stimuli (Lawson and Waddell 1991; Perry et al. 1991). As in other mammals, such as the rat, many small-diameter neurons were NF200-positive in the cat DRG. We have found in the current work that 90.8%, 32.1%, and 2.4% of P2X 3 receptor positive neurons coexpress IB 4, CGRP, and NF200, respectively, which are similar to results obtained from the rat DRG (Bradbury et al. 1998; Vulchanova et al. 1998). Because IB 4 has been shown to label almost all FRAP-positive neurons (Wang et al. 1994), and because we found some colocalization of P2X 3 receptors with CGRP, but virtually no P2X 3 receptor colocalization with NF200
8 1280 Ruan, Birder, de Groat, Tai, Roppolo, Buffington, Burnstock neurons, our results indicate that P2X 3 is predominantly expressed by FRAP-containing DRG neurons with unmyelinated axons. This conclusion is supported by the extensive colocalization of P2X 3 and LA4-IR, because LA4 also labels almost all FRAP-positive cells (Dodd and Jessell 1985). It has been proposed that FRAPcontaining and peptide-containing sensory neurons represent two parallel systems for processing of nociceptive information (Hunt and Rossi 1985). A nociceptive role for FRAP-positive neurons is also suggested by the localization of the enzyme activity in nerve fibers in cornea (Silverman and Kruger 1988) and in tooth pulp primary afferents (Fried et al. 1989), because these peripheral targets are thought to be innervated primarily by nociceptors. In contrast to the P2X 3 receptor, the P2Y 4 receptor was detected in 67.4%, 41.3%, and 39.1% of neurons expressing IB 4, CGRP, and NF200, respectively. Thus P2Y 4 receptor expressing neurons are likely to have unmyelinated and myelinated axons and include peptidergic and nonpeptidergic subpopulations. Thus P2Y 4 receptors may modulate the release of the sensory transmitter, CGRP. In addition, the presence of P2Y 4 receptors in peptidergic and nonpeptidergic neurons suggests that peripheral ATP release may excite a broader population of nociceptors in the cat DRG. We have found that 18.8%, 16.6%, and 63.5% of P2Y 2 receptor positive neurons coexpress IB 4, CGRP, and NF200, respectively. These results suggest that most P2Y 2 receptor positive neurons are likely to have myelinated axons and may participate in the conversion of mechanical stimuli into nerve impulses. P2Y receptors are heptahelical G protein coupled receptors, mediating signal transduction via the induction of inositol triphosphate, leading to intracellular Ca 2 release (Strobaek et al. 1996). P2Y receptors may directly mediate purinergic synaptic transmission or may play a modulatory role. Colocalization of P2X 3, P2Y 2, and P2Y 4 receptors was not examined directly in the present study. However, these receptors have been colocalized with various other markers of subpopulations of primary afferent neurons. A total of 88.2% and 87.4% of IB 4 -positive neurons stained for P2X 3 and P2Y 4 receptors, respectively, and 63.5% of P2Y 2 and 39.1% P2Y 4 receptor positive neurons coexpress NF200. Therefore, some overlapping expression between the P2X 3 and P2Y 4, P2Y 2, and P2Y 4 receptors must be occurring. In conclusion, our results suggest that sensory neurons in the cat DRG are likely to respond to ATP and other purines and pyrimidines, not only through ionotropic P2X receptors, but also through metabotropic P2Y receptors. Substantial differences in the distribution of P2 receptor subtypes have been identified in cat DRG, compared with rat and mouse DRG. It will be of interest to clarify the relative physiological and pathophysiological roles of these two receptor family subtypes. Acknowledgments This study was funded by grants from the NIH (RO1 DK-57282) and Roche Palo Alto (LAB). The authors thank Dr. Gillian E. Knight for editorial assistance. Literature Cited Abbracchio MP, Boeynaems JM, Barnard EA, Boyer JL, Kennedy C, Miras-Portugal MT, King BF, et al. (2003) Characterization of the UDP-glucose receptor (re-named here the P2Y 14 receptor) adds diversity to the P2Y receptor family. Trends Pharmacol Sci 24:52 55 Abbracchio MP, Burnstock G (1994) Purinoceptors: are there families of P2X and P2Y purinoceptors? Pharmacol Ther 64: Alvarez FJ, Morris HR, Priestley JV (1991) Sub-populations of smaller diameter trigeminal primary afferent neurons defined by expression of calcitonin gene-related peptide and the cell surface oligosaccharide recognized by monoclonal antibody LA4. J Neurocytol 20: Averill S, McMahon SB, Clary DO, Reichardt LF, Priestley JV (1995) Immunocytochemical localization of trka receptors in chemically identified subgroups of adult rat sensory neurons. Eur J Neurosci 7: Bessou P, Burgess PR, Perl ER, Taylor CB (1971) Dynamic properties of mechanoreceptors with unmyelinated (C) fibers. J Neurophysiol 34: Birder LA, Ruan H-Z, Chopra B, Xiang Z, Barrick S, Buffington CA, Roppolo JR, et al. (2004) Alterations in P2X and P2Y purinergic receptor expression in urinary bladder from normal cats and cats with interstitial cystitis. Am J Physiol Renal Physiol 287: F Bradbury EJ, Burnstock G, McMahon SB (1998) The expression of P2X3 purinoreceptors in sensory neurons: effects of axotomy and glial-derived neurotrophic factor. Mol Cell Neurosci 12: Buffington CA, Chew DJ, Woodworth BE (1999) Interstitial cystitis in humans and cats? Urology 53: Burgard EC, Niforatos W, van Biesen T, Lynch KJ, Touma E, Metzger RE, Kowaluk EA, et al. (1999) P2X receptor-mediated ionic currents in dorsal root ganglion neurons. J Neurophysiol 82: Burnstock G (2000) P2X receptors in sensory neurones. Br J Anaesth 84: Burnstock G (2001) Purine-mediated signalling in pain and visceral perception. Trends Pharmacol Sci 22: Burnstock G, Kennedy C (1985) Is there a basis for distinguishing two types of P2-purinoceptor? Gen Pharmacol 16: Burnstock G, Wood JN (1996) Purinergic receptors: their role in nociception and primary afferent neurotransmission. Curr Opin Neurobiol 6: Caterina MJ, Schumacher MA, Tominage M, Rosen TA, Levine JD, Julius D (1997) The capsaicin receptor: a heat-activated ion channel in the pain pathway. Nature 389: Chen CC, Akopian AN, Sivilotti L, Colquhoun D, Burnstock G, Wood JN (1995) A P2X purinoceptor expressed by a subset of sensory neurons. Nature 377: Collo G, North RA, Kawashima E, Merlo-Pich E, Neidhart S, Surprenant A, Buell G (1996) Cloning of P2X5 and P2X6 receptors and the distribution and properties of an extended family of ATP-gated ion channels. J Neurosci 16: Communi D, Gonzalez NS, Detheux M, Brezillon S, Lannoy V, Parmentier M, Boeynaems JM (2001) Identification of a novel human ADP receptor coupled to Gi. J Biol Chem 44: Cook SP, Vulchanova L, Hargreaves KM, Elde R, McCleskey EW
9 P2X and P2Y Receptors in Cat DRG 1281 (1997) Distinct ATP receptors on pain-sensing and stretch-sensing neurons. Nature 387: de Groat WC (1999) Neural control of the urinary bladder and sexual organs. In Mathias CJ, Bannister R, eds. Autonomic Failure. 4th ed. Oxford, Oxford Medical Publications, de Groat WC, Fraser MO, Yoshiyama M, Smerin S, Tai C, Chancellor MB, Yoshimura N, et al. (2001) Neural control of the urethra. Scand J Urol Nephrol Suppl 207: Dodd J, Jessell TM (1985) Lactoseries carbohydrates specify subsets of dorsal root ganglion neurons projecting to the superficial dorsal horn of rat spinal cord. J Neurosci 5: Dowd E, McQueen DS, Chessell IP, Humphrey PP (1998) P2X receptor-mediated excitation of nociceptive afferents in the normal and arthritic rat knee joint. Br J Pharmacol 125: Dunn PM, Liu M, Zhong Y, King B, Burnstock G (2000) Diinosine pentaphosphate: an antagonist which discriminates between recombinant P2X 3 and P2X 2/3 receptors and between two P2X receptors in rat sensory neurones. Br J Pharmacol 130: Dunn PM, Zhong Y, Burnstock G (2001) P2X receptors in peripheral neurones. Prog Neurobiol 65: Fried K, Arvidsson J, Robertson B, Brodin E, Theodorsson E (1989) Combined retrograde tracing and enzyme/immunohistochemistry of trigeminal ganglion cell bodies innervating tooth pulps in the rat. Neuroscience 33: Gerke MB, Plenderleith MB (2001) Binding sites for the plant lectin Bandera simplicifolia I-isolectin B(4) are expressed by nociceptive primary sensory neurones. Brain Res 911: Gu JG, MacDermott AB (1997) Activation of ATP P2X receptors elicits glutamate release from sensory neuron synapses. Nature 389: Guo A, Vulchanova L, Wang J, Li X, Elde R (1999) Immunocytochemical localization of the vanilloid receptor 1 (VR1): relationship to neuropeptides, the P2X3 purinoceptor and IB4 binding sites. Eur J Neurosci 11: Hollopeter G, Jantzen HM, Vincent D, Li G, England L, Ramakrishnan V, Yang RB, et al. (2001) Identification of the platelet ADP receptor targeted by antithrombotic drugs. Nature 409: Hunt SP, Rossi J (1985) Peptide- and non-peptide-containing unmyelinated primary afferents: the parallel processing of nociceptive information. Phil Trans R Soc Lond B308: Jänig W, Häbler H-J (1999) Organization of the autonomic nervous system: structure and function. In Appenzeller O, ed. Handbook of Clinical Neurology. Vol. 74 (30): The Autonomic Nervous System. Part 1. Normal Functions. Netherlands, Elsevier, 1 52 Khakh BS, Barnard EA, Burnstock G, Kennedy C, King BF, North RA, Séguéla P, et al. (2000) P2X receptor-channels. In Girdlestone D, ed. The IUPHAR Compendium of Receptor Characterization and Classification. London, IUPHAR Media Ltd, King BF, Burnstock G (2002) Purinergic receptors. In: Pangalos M, Davies C, eds. Understanding G Protein-Coupled Receptors and Their Role in the CNS. Oxford University Press, Oxford, Lawson SN, Harper AA, Harper EI, Garson JA, Anderton BH (1984) A monoclonal antibody against neurofilament protein specifically labels a subpopulation of rat sensory neurones. J Comp Neurol 228: Lawson SN, Waddell PJ (1991) Soma neurofilament immunoreactivity is related to cell size and fibre conduction velocity in rat primary sensory neurons. J Physiol (Lond) 435:41 63 Lewis C, Neidhart S, Holy C, North RA, Buell G, Surprenant A (1995) Coexpression of P2X 2 and P2X 3 receptor subunits can account for ATP-gated currents in sensory neurons. Nature 377: McMahon SB, Armanini MP, Ling LH, Phillips HS (1994) Expression and coexpression of Trk receptors in subpopulations of adult primary sensory neurons projecting to identified peripheral targets. Neuron 12: Molliver DC, Cook SP, Carlsten JA, Wright DE, McCleskey EW (2002) ATP and UTP excite sensory neurons and induce CREB phosphorylation through the metabotropic receptor, P2Y2. Eur J Neurosci 16: Nakamura F, Strittmatter SM (1996) P2Y1 purinergic receptors in sensory neurons: contribution to touch-induced impulse generation. Proc Natl Acad Sci USA 93: Novakovic SD, Kassotakis LC, Oglesby IB, Smith JAM, Eglen RM, Ford APDW, Hunter JC (1999) Immunocytochemical localization of P2X 3 purinoceptors in sensory neurons in naive rats and following neuropathic injury. Pain 80: Perry MJ, Lawson SN, Robertson J (1991) Neurofilament immunoreactivity in populations of rat primary afferent neurons: a quantitative study of phosphorylated and non-phosphorylated subunits. J Neurocytol 20: Petruska JC, Cooper BY, Gu JG, Rau KK, Johnson RD (2000) Distribution of P2X1, P2X2, and P2X3 receptor subunits in rat primary afferents: relation to population markers and specific cell types. J Chem Neuroanat 20: Ralevic V, Burnstock G (1998) Receptors for purines and pyrimidines. Pharmacol Rev 50: Ruan HZ, Burnstock G (2003) Localisation of P2Y 1 and P2Y 4 receptors in dorsal root, nodose and trigeminal ganglia of the rat. Histochem Cell Biol 120: Sanada M, Yasuda H, Omatsu-Kanbe M, Sango K, Isono T, Matsuura H, Kikkawa R (2002) Increase in intracellular Ca 2 and calcitonin gene-related peptide release through metabotropic P2Y receptors in rat dorsal root ganglion neurons. Neuroscience 111: Silverman JD, Kruger L (1988) Lectin and neuropeptide labeling of separate populations of dorsal rat testis and cornea whole-mount preparations. Somatos Res 5: Silverman JD, Kruger L (1990) Selective neuronal glycoconjugate expression in sensory and autonomic ganglia: relation of lectin reactivity to peptide and enzyme markers. J Neurocytol 19: Snider WD, McMahon SB (1998) Tackling pain at the source: new insights into nociceptors. Neuron 20: Strobaek D, Olesen SP, Christophersen P, Dissing S (1996) P2- purinoceptor-mediated formation of inositol phosphates and intracellular Ca 2 transients in human coronary artery smooth muscle cells. Br J Pharmacol 118: Thor KB, Blais DP, de Groat WC (1989a) Behavioural analysis of the postnatal development micturition in kittens. Dev Brain Res 46: Thor KB, Morgan C, Nadelhaft I, Houston M, de Groat WC (1989b) Organization of afferent and efferent pathways in the pudendal nerve of the female cat. J Comp Neurol 288: Tominaga M, Wada M, Masu M (2001) Potentiation of capsaicin receptor activity by metabotropic ATP receptors as a possible mechanism for ATP-evoked pain and hyperalgesia. Proc Natl Acad Sci USA 98: Tsuda M, Koizumi S, Kita A, Shigemoto Y, Ueno S, Inoue K (2000) Mechanical allodynia caused by intraplantar injection of P2X receptor agonist in rats: involvement of heteromeric P2X2/3 receptor signaling in capsaicin-insensitive primary afferent neurons. J Neurosci 20:1 5 Ueno S, Yamada H, Moriyama T, Honda K, Takano Y, Kamiya H, Katsuragi T (2002) Measurement of dorsal root ganglion P2X mrna by SYBR Green fluorescence. Brain Res Prog 10: Vulchanova L, Arvidsson U, Riedl M, Wang J, Buell G, Surprenant A, North RA, et al. (1997) Immunocytochemical study of the P2X 2 and P2X 3 receptor subunits in rat and monkey sensory neurons and their central terminals. Neuropharmacology 36: Vulchanova L, Riedl MS, Shuster SJ, Stone LS, Hargreaves KM, Buell G, Surprenant A, et al. (1998) P2X 3 is expressed by DRG neurons that terminate in inner lamina II. Eur J Neurosci 10: Wang H, Rivero-Melian C, Robertson B, Grant G (1994) Transganglionic transport and binding of the isolectin B4 from Griffonia simplicifolia I in rat primary sensory neurons. Neuroscience 62:
10 1282 Ruan, Birder, de Groat, Tai, Roppolo, Buffington, Burnstock Wu ZZ, Pan HL (2004) Tetrodotoxin-sensitive and -resistant Na channel currents in subsets of small sensory neurons of rats. Brain Res 1029: Xiang Z, Bo X, Burnstock G (1998) Localization of ATP-gated P2X receptor immunoreactivity in rat sensory and sympathetic ganglia. Neurosci Lett 256: Yoshimura N, Seki S, Erickson KA, Erickson VL, Hancellor MB, de Groat WC (2003) Histological and electrical properties of rat dorsal root ganglion neurons innervating the lower urinary tract. J Neurosci 23: Zhang FL, Luo L, Gustafson E, Lachowicz J, Smith M, Qiao X, Liu YH, et al. (2001) ADP is the cognate ligand for the orphan G protein-coupled receptor SP1999. J Biol Chem 276: Zhong Y, Dunn PM, Bardini M, Ford AP, Cockayne DA, Burnstock G (2001) Changes in P2X receptor responses of sensory neurons from P2X 3 -deficient mice. Eur J Neurosci 14: Zimmermann K, Reeh PW, Averbeck B (2002) ATP can enhance the proton-induced CGRP release through P2Y receptors and secondary PGE(2) release in isolated rat dura mater. Pain 97:
Sensory coding and somatosensory system
 Sensory coding and somatosensory system Sensation and perception Perception is the internal construction of sensation. Perception depends on the individual experience. Three common steps in all senses
Sensory coding and somatosensory system Sensation and perception Perception is the internal construction of sensation. Perception depends on the individual experience. Three common steps in all senses
Pharmacology of Pain Transmission and Modulation
 Pharmacology of Pain Transmission and Modulation 2 Jürg Schliessbach and Konrad Maurer Nociceptive Nerve Fibers Pain is transmitted to the central nervous system via thinly myelinated Aδ and unmyelinated
Pharmacology of Pain Transmission and Modulation 2 Jürg Schliessbach and Konrad Maurer Nociceptive Nerve Fibers Pain is transmitted to the central nervous system via thinly myelinated Aδ and unmyelinated
Alterations in P2X and P2Y purinergic receptor expression in urinary bladder from normal cats and cats with interstitial cystitis
 Am J Physiol Renal Physiol 287: F1084 F1091, 2004. First published July 13, 2004; doi:10.1152/ajprenal.00118.2004. Alterations in P2X and P2Y purinergic receptor expression in urinary bladder from normal
Am J Physiol Renal Physiol 287: F1084 F1091, 2004. First published July 13, 2004; doi:10.1152/ajprenal.00118.2004. Alterations in P2X and P2Y purinergic receptor expression in urinary bladder from normal
211MDS Pain theories
 211MDS Pain theories Definition In 1986, the International Association for the Study of Pain (IASP) defined pain as a sensory and emotional experience associated with real or potential injuries, or described
211MDS Pain theories Definition In 1986, the International Association for the Study of Pain (IASP) defined pain as a sensory and emotional experience associated with real or potential injuries, or described
Pathophysiology of Pain
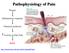 Pathophysiology of Pain Wound Inflammatory response Chemical mediators Activity in Pain Path PAIN http://neuroscience.uth.tmc.edu/s2/chapter08.html Chris Cohan, Ph.D. Dept. of Pathology/Anat Sci University
Pathophysiology of Pain Wound Inflammatory response Chemical mediators Activity in Pain Path PAIN http://neuroscience.uth.tmc.edu/s2/chapter08.html Chris Cohan, Ph.D. Dept. of Pathology/Anat Sci University
The Nervous System. Dr. ZHANG Xiong Dept. of Physiology ZJU School of Medicine.
 The Nervous System Dr. ZHANG Xiong Dept. of Physiology ZJU School of Medicine Http://10.10.10.151 Part 1. Summary of the nervous system The Nervous System Central Nervous System Brain + Spinal Cord Peripheral
The Nervous System Dr. ZHANG Xiong Dept. of Physiology ZJU School of Medicine Http://10.10.10.151 Part 1. Summary of the nervous system The Nervous System Central Nervous System Brain + Spinal Cord Peripheral
浙江大学医学院基础医学整合课程 各论 III. The Nervous System. Dr. ZHANG Xiong Dept. of Physiology ZJU School of Medicine
 The Nervous System Dr. ZHANG Xiong Dept. of Physiology ZJU School of Medicine xiongzhang@zju.edu.cn http://10.202.77.12/ 1 Part 1. Summary of the nervous system 2 The Nervous System Central Nervous System
The Nervous System Dr. ZHANG Xiong Dept. of Physiology ZJU School of Medicine xiongzhang@zju.edu.cn http://10.202.77.12/ 1 Part 1. Summary of the nervous system 2 The Nervous System Central Nervous System
Somatic Sensation (MCB160 Lecture by Mu-ming Poo, Friday March 9, 2007)
 Somatic Sensation (MCB160 Lecture by Mu-ming Poo, Friday March 9, 2007) Introduction Adrian s work on sensory coding Spinal cord and dorsal root ganglia Four somatic sense modalities Touch Mechanoreceptors
Somatic Sensation (MCB160 Lecture by Mu-ming Poo, Friday March 9, 2007) Introduction Adrian s work on sensory coding Spinal cord and dorsal root ganglia Four somatic sense modalities Touch Mechanoreceptors
Introduction to some interesting research questions: Molecular biology of the primary afferent nociceptor
 Introduction to some interesting research questions: Molecular biology of the primary afferent nociceptor NOCICEPTORS ARE NOT IDENTICAL PEPTIDE SubP/CGRP Trk A NON-PEPTIDE IB4 P2X 3 c-ret Snider and McMahon
Introduction to some interesting research questions: Molecular biology of the primary afferent nociceptor NOCICEPTORS ARE NOT IDENTICAL PEPTIDE SubP/CGRP Trk A NON-PEPTIDE IB4 P2X 3 c-ret Snider and McMahon
BLADDER AND CUTANEOUS SENSORY NEURONS OF THE RAT EXPRESS DIFFERENT FUNCTIONAL P2X RECEPTORS
 Neuroscience 120 (2003) 667 675 BLADDER AND CUTANEOUS SENSORY NEURONS OF THE RAT EXPRESS DIFFERENT FUNCTIONAL P2X RECEPTORS Y. ZHONG, a * 1 A. S. BANNING, a D. A. COCKAYNE, b A. P. D. W. FORD, b G. BURNSTOCK
Neuroscience 120 (2003) 667 675 BLADDER AND CUTANEOUS SENSORY NEURONS OF THE RAT EXPRESS DIFFERENT FUNCTIONAL P2X RECEPTORS Y. ZHONG, a * 1 A. S. BANNING, a D. A. COCKAYNE, b A. P. D. W. FORD, b G. BURNSTOCK
Chapter 12 Nervous Tissue. Copyright 2009 John Wiley & Sons, Inc. 1
 Chapter 12 Nervous Tissue Copyright 2009 John Wiley & Sons, Inc. 1 Terms to Know CNS PNS Afferent division Efferent division Somatic nervous system Autonomic nervous system Sympathetic nervous system Parasympathetic
Chapter 12 Nervous Tissue Copyright 2009 John Wiley & Sons, Inc. 1 Terms to Know CNS PNS Afferent division Efferent division Somatic nervous system Autonomic nervous system Sympathetic nervous system Parasympathetic
MOLECULAR AND CELLULAR NEUROSCIENCE
 MOLECULAR AND CELLULAR NEUROSCIENCE BMP-218 November 4, 2014 DIVISIONS OF THE NERVOUS SYSTEM The nervous system is composed of two primary divisions: 1. CNS - Central Nervous System (Brain + Spinal Cord)
MOLECULAR AND CELLULAR NEUROSCIENCE BMP-218 November 4, 2014 DIVISIONS OF THE NERVOUS SYSTEM The nervous system is composed of two primary divisions: 1. CNS - Central Nervous System (Brain + Spinal Cord)
P2X receptors in peripheral neurons
 Progress in Neurobiology 65 (2001) 107 134 www.elsevier.com/locate/pneurobio P2X receptors in peripheral neurons Philip M. Dunn, Yu Zhong, Geoffrey Burnstock * Autonomic Neuroscience Institute, Royal Free
Progress in Neurobiology 65 (2001) 107 134 www.elsevier.com/locate/pneurobio P2X receptors in peripheral neurons Philip M. Dunn, Yu Zhong, Geoffrey Burnstock * Autonomic Neuroscience Institute, Royal Free
What is pain?: An unpleasant sensation. What is an unpleasant sensation?: Pain. - Aristotle.
 What is pain?: An unpleasant sensation. What is an unpleasant sensation?: Pain. - Aristotle. Nociception The detection of tissue damage or impending tissue damage, but There can be tissue damage without
What is pain?: An unpleasant sensation. What is an unpleasant sensation?: Pain. - Aristotle. Nociception The detection of tissue damage or impending tissue damage, but There can be tissue damage without
Product Datasheet. Vanilloid R1/TRPV1 Antibody NB Unit Size: 0.05 ml. Aliquot and store at -20C or -80C. Avoid freeze-thaw cycles.
 Product Datasheet Vanilloid R1/TRPV1 Antibody NB100-1617 Unit Size: 0.05 ml Aliquot and store at -20C or -80C. Avoid freeze-thaw cycles. Publications: 15 Protocols, Publications, Related Products, Reviews,
Product Datasheet Vanilloid R1/TRPV1 Antibody NB100-1617 Unit Size: 0.05 ml Aliquot and store at -20C or -80C. Avoid freeze-thaw cycles. Publications: 15 Protocols, Publications, Related Products, Reviews,
Mechanism of Pain Production
 Mechanism of Pain Production Pain conducting nerve fibers are small myelinated (A-delta) or unmyelinated nerve fibers (C-fibers). Cell bodies are in the dorsal root ganglia (DRG) or sensory ganglia of
Mechanism of Pain Production Pain conducting nerve fibers are small myelinated (A-delta) or unmyelinated nerve fibers (C-fibers). Cell bodies are in the dorsal root ganglia (DRG) or sensory ganglia of
Module H NERVOUS SYSTEM
 Module H NERVOUS SYSTEM Topic from General functions of the nervous system Organization of the nervous system from both anatomical & functional perspectives Gross & microscopic anatomy of nervous tissue
Module H NERVOUS SYSTEM Topic from General functions of the nervous system Organization of the nervous system from both anatomical & functional perspectives Gross & microscopic anatomy of nervous tissue
Introduction ORIGINAL PAPER. Zhenghua Xiang Æ Geoffrey Burnstock
 Histochem Cell Biol (2006) 125: 327 336 DOI 10.1007/s00418-005-0071-3 ORIGINAL PAPER Zhenghua Xiang Æ Geoffrey Burnstock Distribution of P2Y 6 and P2Y 12 receptor: their colocalization with calbindin,
Histochem Cell Biol (2006) 125: 327 336 DOI 10.1007/s00418-005-0071-3 ORIGINAL PAPER Zhenghua Xiang Æ Geoffrey Burnstock Distribution of P2Y 6 and P2Y 12 receptor: their colocalization with calbindin,
LOCALIZATION OF SUBSTANCE P-IMMUNOREACTIVITY IN THE DEVELOPING HUMAN URINARY BLADDER
 ACTA HISTOCHEM. CYTOCHEM. Vol. 21, No. 2, 1988 LOCALIZATION OF SUBSTANCE P-IMMUNOREACTIVITY IN THE DEVELOPING HUMAN URINARY BLADDER SHASHI WADHWA AND VEENA BIJLANI Department of Anatomy, All-India Institute
ACTA HISTOCHEM. CYTOCHEM. Vol. 21, No. 2, 1988 LOCALIZATION OF SUBSTANCE P-IMMUNOREACTIVITY IN THE DEVELOPING HUMAN URINARY BLADDER SHASHI WADHWA AND VEENA BIJLANI Department of Anatomy, All-India Institute
Nervous System. Master controlling and communicating system of the body. Secrete chemicals called neurotransmitters
 Nervous System Master controlling and communicating system of the body Interacts with the endocrine system to control and coordinate the body s responses to changes in its environment, as well as growth,
Nervous System Master controlling and communicating system of the body Interacts with the endocrine system to control and coordinate the body s responses to changes in its environment, as well as growth,
Expression of the transient receptor potential channels TRPV1, TRPA1 and TRPM8 in mouse trigeminal primary afferent neurons innervating the dura
 Washington University School of Medicine Digital Commons@Becker Open Access Publications 212 Expression of the transient receptor potential channels TRPV1, TRPA1 and TRPM8 in mouse trigeminal primary afferent
Washington University School of Medicine Digital Commons@Becker Open Access Publications 212 Expression of the transient receptor potential channels TRPV1, TRPA1 and TRPM8 in mouse trigeminal primary afferent
A role for uninjured afferents in neuropathic pain
 Acta Physiologica Sinica, October 25, 2008, 60 (5): 605-609 http://www.actaps.com.cn 605 Review A role for uninjured afferents in neuropathic pain Richard A. Meyer 1,2,3,*, Matthias Ringkamp 1 Departments
Acta Physiologica Sinica, October 25, 2008, 60 (5): 605-609 http://www.actaps.com.cn 605 Review A role for uninjured afferents in neuropathic pain Richard A. Meyer 1,2,3,*, Matthias Ringkamp 1 Departments
Pain. Pain. Pain: One definition. Pain: One definition. Pain: One definition. Pain: One definition. Psyc 2906: Sensation--Introduction 9/27/2006
 Pain Pain Pain: One Definition Classic Paths A new Theory Pain and Drugs According to the international Association for the Study (Merskey & Bogduk, 1994), Pain is an unpleasant sensory and emotional experience
Pain Pain Pain: One Definition Classic Paths A new Theory Pain and Drugs According to the international Association for the Study (Merskey & Bogduk, 1994), Pain is an unpleasant sensory and emotional experience
Chapter 11: Nervous System and Nervous Tissue
 Chapter 11: Nervous System and Nervous Tissue I. Functions and divisions of the nervous system A. Sensory input: monitor changes in internal and external environment B. Integrations: make decisions about
Chapter 11: Nervous System and Nervous Tissue I. Functions and divisions of the nervous system A. Sensory input: monitor changes in internal and external environment B. Integrations: make decisions about
Pain and impulse conduction
 1 Pain and impulse conduction L.H.D.J. Booij According to the World Health Organisation pain is defined as an unpleasant sensation that occurs from imminent tissue damage. From a physiological perspective,
1 Pain and impulse conduction L.H.D.J. Booij According to the World Health Organisation pain is defined as an unpleasant sensation that occurs from imminent tissue damage. From a physiological perspective,
Neural Control of Lower Urinary Tract Function. William C. de Groat University of Pittsburgh Medical School
 Neural Control of Lower Urinary Tract Function William C. de Groat University of Pittsburgh Medical School Disclosures Current funding: NIH Grants, DK093424, DK-091253, DK-094905, DK-090006. Other financial
Neural Control of Lower Urinary Tract Function William C. de Groat University of Pittsburgh Medical School Disclosures Current funding: NIH Grants, DK093424, DK-091253, DK-094905, DK-090006. Other financial
Chapter 11 Introduction to the Nervous System and Nervous Tissue Chapter Outline
 Chapter 11 Introduction to the Nervous System and Nervous Tissue Chapter Outline Module 11.1 Overview of the Nervous System (Figures 11.1-11.3) A. The nervous system controls our perception and experience
Chapter 11 Introduction to the Nervous System and Nervous Tissue Chapter Outline Module 11.1 Overview of the Nervous System (Figures 11.1-11.3) A. The nervous system controls our perception and experience
Receptors and Neurotransmitters: It Sounds Greek to Me. Agenda. What We Know About Pain 9/7/2012
 Receptors and Neurotransmitters: It Sounds Greek to Me Cathy Carlson, PhD, RN Northern Illinois University Agenda We will be going through this lecture on basic pain physiology using analogies, mnemonics,
Receptors and Neurotransmitters: It Sounds Greek to Me Cathy Carlson, PhD, RN Northern Illinois University Agenda We will be going through this lecture on basic pain physiology using analogies, mnemonics,
What it Takes to be a Pain
 What it Takes to be a Pain Pain Pathways and the Neurophysiology of pain Dennis S. Pacl, MD, FACP, FAChPM Austin Palliative Care/ Hospice Austin A Definition of Pain complex constellation of unpleasant
What it Takes to be a Pain Pain Pathways and the Neurophysiology of pain Dennis S. Pacl, MD, FACP, FAChPM Austin Palliative Care/ Hospice Austin A Definition of Pain complex constellation of unpleasant
number Done by Corrected by Doctor
 number 13 Done by Tamara Wahbeh Corrected by Doctor Omar Shaheen In this sheet the following concepts will be covered: 1. Divisions of the nervous system 2. Anatomy of the ANS. 3. ANS innervations. 4.
number 13 Done by Tamara Wahbeh Corrected by Doctor Omar Shaheen In this sheet the following concepts will be covered: 1. Divisions of the nervous system 2. Anatomy of the ANS. 3. ANS innervations. 4.
Changes in P2X receptor responses of sensory neurons from P2X 3 -de cient mice
 European Journal of Neuroscience, Vol. 14, pp. 1784±1792, 2001 ã Federation of European Neuroscience Societies Changes in P2X receptor responses of sensory neurons from P2X 3 -de cient mice Yu Zhong, 1
European Journal of Neuroscience, Vol. 14, pp. 1784±1792, 2001 ã Federation of European Neuroscience Societies Changes in P2X receptor responses of sensory neurons from P2X 3 -de cient mice Yu Zhong, 1
Risk Factors That Predispose Patients to Orofacial Pain
 Risk Factors That Predispose Patients to Orofacial Pain Paul Durham, PhD Distinguished Professor of Cell Biology Director Center for Biomedical & Life Sciences Missouri State University Cluster Headache
Risk Factors That Predispose Patients to Orofacial Pain Paul Durham, PhD Distinguished Professor of Cell Biology Director Center for Biomedical & Life Sciences Missouri State University Cluster Headache
CHAPTER 10 THE SOMATOSENSORY SYSTEM
 CHAPTER 10 THE SOMATOSENSORY SYSTEM 10.1. SOMATOSENSORY MODALITIES "Somatosensory" is really a catch-all term to designate senses other than vision, hearing, balance, taste and smell. Receptors that could
CHAPTER 10 THE SOMATOSENSORY SYSTEM 10.1. SOMATOSENSORY MODALITIES "Somatosensory" is really a catch-all term to designate senses other than vision, hearing, balance, taste and smell. Receptors that could
P2X 3 receptors and peripheral pain mechanisms
 J Physiol 554.2 pp 301 308 301 SYMPOSIUM REPORT P2X 3 receptors and peripheral pain mechanisms R. Alan North Institute of Molecular Physiology, University of Sheffield, Western Bank, Sheffield S10 2TN,
J Physiol 554.2 pp 301 308 301 SYMPOSIUM REPORT P2X 3 receptors and peripheral pain mechanisms R. Alan North Institute of Molecular Physiology, University of Sheffield, Western Bank, Sheffield S10 2TN,
Overactive Bladder; OAB. Aδ silent C. Adenosine triphosphate; ATP SD g n Key words
 25-32 2009 SD 200-270g n 28 4 1 2 3 4 PE-50 4-5 0.25 60 C Key words acupuncture stimulation overactive bladder rat bladder irritation C C fiber Received November 17, 2008; Accepted January 9, 2009 I Overactive
25-32 2009 SD 200-270g n 28 4 1 2 3 4 PE-50 4-5 0.25 60 C Key words acupuncture stimulation overactive bladder rat bladder irritation C C fiber Received November 17, 2008; Accepted January 9, 2009 I Overactive
Introduction to Neurobiology
 Biology 240 General Zoology Introduction to Neurobiology Nervous System functions: communication of information via nerve signals integration and processing of information control of physiological and
Biology 240 General Zoology Introduction to Neurobiology Nervous System functions: communication of information via nerve signals integration and processing of information control of physiological and
The Nervous System: Neural Tissue Pearson Education, Inc.
 13 The Nervous System: Neural Tissue Introduction Nervous System Characteristics Controls and adjust the activity of the body Provides swift but brief responses The nervous system includes: Central Nervous
13 The Nervous System: Neural Tissue Introduction Nervous System Characteristics Controls and adjust the activity of the body Provides swift but brief responses The nervous system includes: Central Nervous
Inflammation-Induced Airway Hypersensitivity: From Ion Channels to Patients
 Inflammation-Induced Airway Hypersensitivity: From Ion Channels to Patients Lu-Yuan Lee, Ph.D. Airway Sensory Neurobiology Laboratory Department of Physiology University of Kentucky Medical Center BACKGROUND
Inflammation-Induced Airway Hypersensitivity: From Ion Channels to Patients Lu-Yuan Lee, Ph.D. Airway Sensory Neurobiology Laboratory Department of Physiology University of Kentucky Medical Center BACKGROUND
Department of Neurology/Division of Anatomical Sciences
 Spinal Cord I Lecture Outline and Objectives CNS/Head and Neck Sequence TOPIC: FACULTY: THE SPINAL CORD AND SPINAL NERVES, Part I Department of Neurology/Division of Anatomical Sciences LECTURE: Monday,
Spinal Cord I Lecture Outline and Objectives CNS/Head and Neck Sequence TOPIC: FACULTY: THE SPINAL CORD AND SPINAL NERVES, Part I Department of Neurology/Division of Anatomical Sciences LECTURE: Monday,
TMC9 as a novel mechanosensitive ion channel
 TMC9 as a novel mechanosensitive ion channel Mechanical forces play numerous roles in physiology. When an object contacts our skin, it exerts a force that is encoded as touch or pain depending on its intensity.
TMC9 as a novel mechanosensitive ion channel Mechanical forces play numerous roles in physiology. When an object contacts our skin, it exerts a force that is encoded as touch or pain depending on its intensity.
Autonomic Nervous System. Lanny Shulman, O.D., Ph.D. University of Houston College of Optometry
 Autonomic Nervous System Lanny Shulman, O.D., Ph.D. University of Houston College of Optometry Peripheral Nervous System A. Sensory Somatic Nervous System B. Autonomic Nervous System 1. Sympathetic Nervous
Autonomic Nervous System Lanny Shulman, O.D., Ph.D. University of Houston College of Optometry Peripheral Nervous System A. Sensory Somatic Nervous System B. Autonomic Nervous System 1. Sympathetic Nervous
W11: Functional Pain Syndromes: Bidirectional Influences in Visceral Nociceptive Processing Aims of course/workshop
 W11: Functional Pain Syndromes: Bidirectional Influences in Visceral Nociceptive Processing Workshop Chair: Matthew Fraser, United States 27 August 2013 09:00-10:30 Start End Topic Speakers 09:00 09:25
W11: Functional Pain Syndromes: Bidirectional Influences in Visceral Nociceptive Processing Workshop Chair: Matthew Fraser, United States 27 August 2013 09:00-10:30 Start End Topic Speakers 09:00 09:25
Seizure: the clinical manifestation of an abnormal and excessive excitation and synchronization of a population of cortical
 Are There Sharing Mechanisms of Epilepsy, Migraine and Neuropathic Pain? Chin-Wei Huang, MD, PhD Department of Neurology, NCKUH Basic mechanisms underlying seizures and epilepsy Seizure: the clinical manifestation
Are There Sharing Mechanisms of Epilepsy, Migraine and Neuropathic Pain? Chin-Wei Huang, MD, PhD Department of Neurology, NCKUH Basic mechanisms underlying seizures and epilepsy Seizure: the clinical manifestation
Neurophysiology scripts. Slide 2
 Neurophysiology scripts Slide 2 Nervous system and Endocrine system both maintain homeostasis in the body. Nervous system by nerve impulse and Endocrine system by hormones. Since the nerve impulse is an
Neurophysiology scripts Slide 2 Nervous system and Endocrine system both maintain homeostasis in the body. Nervous system by nerve impulse and Endocrine system by hormones. Since the nerve impulse is an
A subpopulation of nociceptors specifically linked to itch
 Supplementary Information A subpopulation of nociceptors specifically linked to itch Liang Han 1, Chao Ma 3,4, Qin Liu 1,2, Hao-Jui Weng 1,2, Yiyuan Cui 5, Zongxiang Tang 1,2, Yushin Kim 1, Hong Nie 4,
Supplementary Information A subpopulation of nociceptors specifically linked to itch Liang Han 1, Chao Ma 3,4, Qin Liu 1,2, Hao-Jui Weng 1,2, Yiyuan Cui 5, Zongxiang Tang 1,2, Yushin Kim 1, Hong Nie 4,
NEURONS Chapter Neurons: specialized cells of the nervous system 2. Nerves: bundles of neuron axons 3. Nervous systems
 NEURONS Chapter 12 Figure 12.1 Neuronal and hormonal signaling both convey information over long distances 1. Nervous system A. nervous tissue B. conducts electrical impulses C. rapid communication 2.
NEURONS Chapter 12 Figure 12.1 Neuronal and hormonal signaling both convey information over long distances 1. Nervous system A. nervous tissue B. conducts electrical impulses C. rapid communication 2.
Somatosensory modalities!
 Somatosensory modalities! The somatosensory system codes five major sensory modalities:! 1. Discriminative touch! 2. Proprioception (body position and motion)! 3. Nociception (pain and itch)! 4. Temperature!
Somatosensory modalities! The somatosensory system codes five major sensory modalities:! 1. Discriminative touch! 2. Proprioception (body position and motion)! 3. Nociception (pain and itch)! 4. Temperature!
Intracellular signalling cascades associated with TRP channels
 Gerald Thiel Intracellular signalling cascades associated with TRP channels Current state of investigations and potential applications Saarland University, Germany Campus Saarbrücken Department of Medical
Gerald Thiel Intracellular signalling cascades associated with TRP channels Current state of investigations and potential applications Saarland University, Germany Campus Saarbrücken Department of Medical
Major Structures of the Nervous System. Brain, cranial nerves, spinal cord, spinal nerves, ganglia, enteric plexuses and sensory receptors
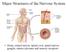 Major Structures of the Nervous System Brain, cranial nerves, spinal cord, spinal nerves, ganglia, enteric plexuses and sensory receptors Nervous System Divisions Central Nervous System (CNS) consists
Major Structures of the Nervous System Brain, cranial nerves, spinal cord, spinal nerves, ganglia, enteric plexuses and sensory receptors Nervous System Divisions Central Nervous System (CNS) consists
The Nervous System. Nervous System Functions 1. gather sensory input 2. integration- process and interpret sensory input 3. cause motor output
 The Nervous System Nervous System Functions 1. gather sensory input 2. integration- process and interpret sensory input 3. cause motor output The Nervous System 2 Parts of the Nervous System 1. central
The Nervous System Nervous System Functions 1. gather sensory input 2. integration- process and interpret sensory input 3. cause motor output The Nervous System 2 Parts of the Nervous System 1. central
Biomechanics of Pain: Dynamics of the Neuromatrix
 Biomechanics of Pain: Dynamics of the Neuromatrix Partap S. Khalsa, D.C., Ph.D. Department of Biomedical Engineering The Neuromatrix From: Melzack R (1999) Pain Suppl 6:S121-6. NIOSH STAR Symposium May
Biomechanics of Pain: Dynamics of the Neuromatrix Partap S. Khalsa, D.C., Ph.D. Department of Biomedical Engineering The Neuromatrix From: Melzack R (1999) Pain Suppl 6:S121-6. NIOSH STAR Symposium May
Charlie Taylor, PhD CpTaylor Consulting Chelsea, MI, USA
 Contribution of Calcium Channel α 2 δ Binding Sites to the Pharmacology of Gabapentin and Pregabalin Charlie Taylor, PhD CpTaylor Consulting Chelsea, MI, USA Disclosure Information Charlie Taylor, PhD
Contribution of Calcium Channel α 2 δ Binding Sites to the Pharmacology of Gabapentin and Pregabalin Charlie Taylor, PhD CpTaylor Consulting Chelsea, MI, USA Disclosure Information Charlie Taylor, PhD
EE 791 Lecture 2 Jan 19, 2015
 EE 791 Lecture 2 Jan 19, 2015 Action Potential Conduction And Neural Organization EE 791-Lecture 2 1 Core-conductor model: In the core-conductor model we approximate an axon or a segment of a dendrite
EE 791 Lecture 2 Jan 19, 2015 Action Potential Conduction And Neural Organization EE 791-Lecture 2 1 Core-conductor model: In the core-conductor model we approximate an axon or a segment of a dendrite
Omar Ismail. Dana Almanzalji. Faisal Mohammad
 11 Omar Ismail Dana Almanzalji Faisal Mohammad Neuronal classification: Neurons are responsible for transmitting the action potential to the brain. The speed at which the action potential is transmitted
11 Omar Ismail Dana Almanzalji Faisal Mohammad Neuronal classification: Neurons are responsible for transmitting the action potential to the brain. The speed at which the action potential is transmitted
Histological and Electrical Properties of Rat Dorsal Root Ganglion Neurons Innervating the Lower Urinary Tract
 The Journal of Neuroscience, May 15, 2003 23(10):4355 4361 4355 Histological and Electrical Properties of Rat Dorsal Root Ganglion Neurons Innervating the Lower Urinary Tract Naoki Yoshimura, 1,2 Satoshi
The Journal of Neuroscience, May 15, 2003 23(10):4355 4361 4355 Histological and Electrical Properties of Rat Dorsal Root Ganglion Neurons Innervating the Lower Urinary Tract Naoki Yoshimura, 1,2 Satoshi
Victor Chaban, Ph.D., MSCR
 Hormonal Modulation of Pain : Estrogen Modulation of Visceral Nociception Victor Chaban, Ph.D., MSCR Department of Internal Medicine Charles R. Drew University of Medicine and Science Department of Medicine
Hormonal Modulation of Pain : Estrogen Modulation of Visceral Nociception Victor Chaban, Ph.D., MSCR Department of Internal Medicine Charles R. Drew University of Medicine and Science Department of Medicine
Somatosensation. Recording somatosensory responses. Receptive field response to pressure
 Somatosensation Mechanoreceptors that respond to touch/pressure on the surface of the body. Sensory nerve responds propotional to pressure 4 types of mechanoreceptors: Meissner corpuscles & Merkel discs
Somatosensation Mechanoreceptors that respond to touch/pressure on the surface of the body. Sensory nerve responds propotional to pressure 4 types of mechanoreceptors: Meissner corpuscles & Merkel discs
16. which is not synthesised in postganglionic sympathetic neurons a. L-dopa b. DA c. NA d. A e. ACh
 NERVOUS SYSTEM 1. Visual pathways a. Have P cells that are associated with colour b. Utilize the primary colours, red, yellow and blue c. Have simple cells which respond to all light stimuli d. Pass through
NERVOUS SYSTEM 1. Visual pathways a. Have P cells that are associated with colour b. Utilize the primary colours, red, yellow and blue c. Have simple cells which respond to all light stimuli d. Pass through
Lesson 33. Objectives: References: Chapter 16: Reading for Next Lesson: Chapter 16:
 Lesson 33 Lesson Outline: Nervous System Structure and Function Neuronal Tissue Supporting Cells Neurons Nerves Functional Classification of Neuronal Tissue Organization of the Nervous System Peripheral
Lesson 33 Lesson Outline: Nervous System Structure and Function Neuronal Tissue Supporting Cells Neurons Nerves Functional Classification of Neuronal Tissue Organization of the Nervous System Peripheral
Pain Mechanisms. Prof Michael G Irwin MD, FRCA, FANZCA FHKAM Head Department of Anaesthesiology University of Hong Kong. The Somatosensory System
 ain Mechanisms rof Michael G Irwin MD, FRCA, FANZCA FHKAM Head Department of Anaesthesiology University of Hong Kong The Somatosensory System Frontal cortex Descending pathway eriaqueductal gray matter
ain Mechanisms rof Michael G Irwin MD, FRCA, FANZCA FHKAM Head Department of Anaesthesiology University of Hong Kong The Somatosensory System Frontal cortex Descending pathway eriaqueductal gray matter
10.1: Introduction. Cell types in neural tissue: Neurons Neuroglial cells (also known as neuroglia, glia, and glial cells) Dendrites.
 10.1: Introduction Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display. Cell types in neural tissue: Neurons Neuroglial cells (also known as neuroglia, glia, and glial
10.1: Introduction Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display. Cell types in neural tissue: Neurons Neuroglial cells (also known as neuroglia, glia, and glial
Fundamentals of the Nervous System and Nervous Tissue: Part C
 PowerPoint Lecture Slides prepared by Janice Meeking, Mount Royal College C H A P T E R 11 Fundamentals of the Nervous System and Nervous Tissue: Part C Warm Up What is a neurotransmitter? What is the
PowerPoint Lecture Slides prepared by Janice Meeking, Mount Royal College C H A P T E R 11 Fundamentals of the Nervous System and Nervous Tissue: Part C Warm Up What is a neurotransmitter? What is the
Neurotransmitter Systems II Receptors. Reading: BCP Chapter 6
 Neurotransmitter Systems II Receptors Reading: BCP Chapter 6 Neurotransmitter Systems Normal function of the human brain requires an orderly set of chemical reactions. Some of the most important chemical
Neurotransmitter Systems II Receptors Reading: BCP Chapter 6 Neurotransmitter Systems Normal function of the human brain requires an orderly set of chemical reactions. Some of the most important chemical
Repeated intra-articular injections of acidic saline produce long-lasting joint pain and. widespread hyperalgesia
 Repeated intra-articular injections of acidic saline produce long-lasting joint pain and widespread hyperalgesia N. Sugimura 1, M. Ikeuchi 1 *, M. Izumi 1, T. Kawano 2, K. Aso 1, T. Kato 1, T. Ushida 3,
Repeated intra-articular injections of acidic saline produce long-lasting joint pain and widespread hyperalgesia N. Sugimura 1, M. Ikeuchi 1 *, M. Izumi 1, T. Kawano 2, K. Aso 1, T. Kato 1, T. Ushida 3,
University of Groningen. Neuronal control of micturition Kuipers, Rutger
 University of Groningen Neuronal control of micturition Kuipers, Rutger IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish to cite from it. Please check the
University of Groningen Neuronal control of micturition Kuipers, Rutger IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish to cite from it. Please check the
Chapter Six Review Sections 1 and 2
 NAME PER DATE Chapter Six Review Sections 1 and 2 Matching: 1. afferent nerves 2. autonomic nervous system 3. cell body 4. central nervous system (CNS) 5. dendrites 6. efferent nerves 7. myelin sheath
NAME PER DATE Chapter Six Review Sections 1 and 2 Matching: 1. afferent nerves 2. autonomic nervous system 3. cell body 4. central nervous system (CNS) 5. dendrites 6. efferent nerves 7. myelin sheath
Chapter 17 Nervous System
 Chapter 17 Nervous System 1 The Nervous System Two Anatomical Divisions Central Nervous System (CNS) Brain and Spinal Cord Peripheral Nervous System (PNS) Two Types of Cells Neurons Transmit nerve impulses
Chapter 17 Nervous System 1 The Nervous System Two Anatomical Divisions Central Nervous System (CNS) Brain and Spinal Cord Peripheral Nervous System (PNS) Two Types of Cells Neurons Transmit nerve impulses
Drugs Affecting The Autonomic Nervous System(ANS)
 Drugs Affecting The Autonomic Nervous System(ANS) ANS Pharmacology Lecture 1 Dr. Hiwa K. Saaed College of Pharmacy, University of Sulaimani 2018-2019 AUTOMATIC NERVOUS SYSTEM (ANS) The ANS is the major
Drugs Affecting The Autonomic Nervous System(ANS) ANS Pharmacology Lecture 1 Dr. Hiwa K. Saaed College of Pharmacy, University of Sulaimani 2018-2019 AUTOMATIC NERVOUS SYSTEM (ANS) The ANS is the major
What Cell Make Up the Brain and Spinal Cord
 What Cell Make Up the Brain and Spinal Cord Jennifer LaVail, Ph.D. (http://anatomy.ucsf.edu/pages/lavaillab/index.html) What kinds of cells are these?" Neuron?" Epithelial cell?" Glial cell?" What makes
What Cell Make Up the Brain and Spinal Cord Jennifer LaVail, Ph.D. (http://anatomy.ucsf.edu/pages/lavaillab/index.html) What kinds of cells are these?" Neuron?" Epithelial cell?" Glial cell?" What makes
SYNAPTIC COMMUNICATION
 BASICS OF NEUROBIOLOGY SYNAPTIC COMMUNICATION ZSOLT LIPOSITS 1 NERVE ENDINGS II. Interneuronal communication 2 INTERNEURONAL COMMUNICATION I. ELECTRONIC SYNAPSE GAP JUNCTION II. CHEMICAL SYNAPSE SYNAPSES
BASICS OF NEUROBIOLOGY SYNAPTIC COMMUNICATION ZSOLT LIPOSITS 1 NERVE ENDINGS II. Interneuronal communication 2 INTERNEURONAL COMMUNICATION I. ELECTRONIC SYNAPSE GAP JUNCTION II. CHEMICAL SYNAPSE SYNAPSES
Brian Kahan, D.O. FAAPMR, DABPM, DAOCRM, FIPP Center for Pain Medicine and Physiatric Rehabilitation 2002 Medical Parkway Suite 150 Annapolis, MD
 Brian Kahan, D.O. FAAPMR, DABPM, DAOCRM, FIPP Center for Pain Medicine and Physiatric Rehabilitation 2002 Medical Parkway Suite 150 Annapolis, MD 1630 Main Street Suite 215 Chester, MD 410-571-9000 www.4-no-pain.com
Brian Kahan, D.O. FAAPMR, DABPM, DAOCRM, FIPP Center for Pain Medicine and Physiatric Rehabilitation 2002 Medical Parkway Suite 150 Annapolis, MD 1630 Main Street Suite 215 Chester, MD 410-571-9000 www.4-no-pain.com
Introduction to Nervous Tissue
 Introduction to Nervous Tissue Nervous Tissue Controls and integrates all body activities within limits that maintain life Three basic functions 1. sensing changes with sensory receptors 2. interpreting
Introduction to Nervous Tissue Nervous Tissue Controls and integrates all body activities within limits that maintain life Three basic functions 1. sensing changes with sensory receptors 2. interpreting
Proceedings of the World Small Animal Veterinary Association Sydney, Australia 2007
 Proceedings of the World Small Animal Sydney, Australia 2007 Hosted by: Next WSAVA Congress REDUCING THE PAIN FACTOR AN UPDATE ON PERI-OPERATIVE ANALGESIA Sandra Forysth, BVSc DipACVA Institute of Veterinary,
Proceedings of the World Small Animal Sydney, Australia 2007 Hosted by: Next WSAVA Congress REDUCING THE PAIN FACTOR AN UPDATE ON PERI-OPERATIVE ANALGESIA Sandra Forysth, BVSc DipACVA Institute of Veterinary,
Somatosensory System. Steven McLoon Department of Neuroscience University of Minnesota
 Somatosensory System Steven McLoon Department of Neuroscience University of Minnesota 1 Course News Dr. Riedl s review session this week: Tuesday (Oct 10) 4-5pm in MCB 3-146B 2 Sensory Systems Sensory
Somatosensory System Steven McLoon Department of Neuroscience University of Minnesota 1 Course News Dr. Riedl s review session this week: Tuesday (Oct 10) 4-5pm in MCB 3-146B 2 Sensory Systems Sensory
EDUCATION M.D., Peking Union Medical College, Beijing, China, 1999 B.S., Beijing University, College of Life Science, Beijing, China, 1994
 CHAO MA, M.D. Yale University School of Medicine, Department of Anesthesiology, 333 Cedar Street, TMP3, New Haven, CT 06510, USA. Phone: 203-785-3522 (O), 203-606-7959 (C), Fax: 203-737-1528, Email: chao.ma@yale.edu
CHAO MA, M.D. Yale University School of Medicine, Department of Anesthesiology, 333 Cedar Street, TMP3, New Haven, CT 06510, USA. Phone: 203-785-3522 (O), 203-606-7959 (C), Fax: 203-737-1528, Email: chao.ma@yale.edu
Pathophysiology of Pain. Ramon Go MD Assistant Professor Anesthesiology and Pain medicine NYP-CUMC
 Pathophysiology of Pain Ramon Go MD Assistant Professor Anesthesiology and Pain medicine NYP-CUMC Learning Objectives Anatomic pathway of nociception Discuss the multiple target sites of pharmacological
Pathophysiology of Pain Ramon Go MD Assistant Professor Anesthesiology and Pain medicine NYP-CUMC Learning Objectives Anatomic pathway of nociception Discuss the multiple target sites of pharmacological
What are the 6 types of neuroglia and their functions?!
 Warm Up! Take out your 11C Notes What are the 6 types of neuroglia and their functions?! Astrocytes Microglia Ependymal Cells Satellite Cells Schwann Cells Oligodendrocytes Support, brace, & nutrient transfer
Warm Up! Take out your 11C Notes What are the 6 types of neuroglia and their functions?! Astrocytes Microglia Ependymal Cells Satellite Cells Schwann Cells Oligodendrocytes Support, brace, & nutrient transfer
Adenosine 5 0 -triphosphate and its relationship with other mediators that activate pelvic nerve afferent neurons in the rat colorectum
 Purinergic Signalling (2006) 2: 517 526 DOI: 10.1007/s11302-005-5305-2 # Springer 2006 Adenosine 5 0 -triphosphate and its relationship with other mediators that activate pelvic nerve afferent neurons
Purinergic Signalling (2006) 2: 517 526 DOI: 10.1007/s11302-005-5305-2 # Springer 2006 Adenosine 5 0 -triphosphate and its relationship with other mediators that activate pelvic nerve afferent neurons
The Egyptian Journal of Hospital Medicine (January 2018) Vol. 70 (12), Page
 The Egyptian Journal of Hospital Medicine (January 2018) Vol. 70 (12), Page 2172-2177 Blockage of HCN Channels with ZD7288 Attenuates Mechanical Hypersensitivity in Rats Model of Diabetic Neuropathy Hussain
The Egyptian Journal of Hospital Medicine (January 2018) Vol. 70 (12), Page 2172-2177 Blockage of HCN Channels with ZD7288 Attenuates Mechanical Hypersensitivity in Rats Model of Diabetic Neuropathy Hussain
Pain Pathways. Dr Sameer Gupta Consultant in Anaesthesia and Pain Management, NGH
 Pain Pathways Dr Sameer Gupta Consultant in Anaesthesia and Pain Management, NGH Objective To give you a simplistic and basic concepts of pain pathways to help understand the complex issue of pain Pain
Pain Pathways Dr Sameer Gupta Consultant in Anaesthesia and Pain Management, NGH Objective To give you a simplistic and basic concepts of pain pathways to help understand the complex issue of pain Pain
Portions from Chapter 6 CHAPTER 7. The Nervous System: Neurons and Synapses. Chapter 7 Outline. and Supporting Cells
 CHAPTER 7 The Nervous System: Neurons and Synapses Chapter 7 Outline Neurons and Supporting Cells Activity in Axons The Synapse Acetylcholine as a Neurotransmitter Monoamines as Neurotransmitters Other
CHAPTER 7 The Nervous System: Neurons and Synapses Chapter 7 Outline Neurons and Supporting Cells Activity in Axons The Synapse Acetylcholine as a Neurotransmitter Monoamines as Neurotransmitters Other
Neuron types and Neurotransmitters
 Neuron types and Neurotransmitters Faisal I. Mohammed. PhD, MD University of Jordan 1 Transmission of Receptor Information to the Brain the larger the nerve fiber diameter the faster the rate of transmission
Neuron types and Neurotransmitters Faisal I. Mohammed. PhD, MD University of Jordan 1 Transmission of Receptor Information to the Brain the larger the nerve fiber diameter the faster the rate of transmission
Introduction ORIGINAL ARTICLE. Seiji Ohtori Kazuhisa Takahashi Hideshige Moriya
 Eur Spine J (2003) 12 :211 215 DOI 10.1007/s00586-002-0506-7 ORIGINAL ARTICLE Seiji Ohtori Kazuhisa Takahashi Hideshige Moriya Calcitonin gene-related peptide immunoreactive DRG neurons innervating the
Eur Spine J (2003) 12 :211 215 DOI 10.1007/s00586-002-0506-7 ORIGINAL ARTICLE Seiji Ohtori Kazuhisa Takahashi Hideshige Moriya Calcitonin gene-related peptide immunoreactive DRG neurons innervating the
Bi/CNS/NB 150: Neuroscience. November 11, 2015 SOMATOSENSORY SYSTEM. Ralph Adolphs
 Bi/CNS/NB 150: Neuroscience November 11, 2015 SOMATOSENSORY SYSTEM Ralph Adolphs 1 Menu for today Touch -peripheral -central -plasticity Pain 2 Sherrington (1948): senses classified as --teloreceptive
Bi/CNS/NB 150: Neuroscience November 11, 2015 SOMATOSENSORY SYSTEM Ralph Adolphs 1 Menu for today Touch -peripheral -central -plasticity Pain 2 Sherrington (1948): senses classified as --teloreceptive
Eugenol Inhibits ATP-induced P2X Currents in Trigeminal Ganglion Neurons
 Korean J Physiol Pharmacol Vol 12: 315-321, December, 2008 Eugenol Inhibits ATP-induced P2X Currents in Trigeminal Ganglion Neurons Hai Ying Li 1, Byung Ky Lee 1, Joong Soo Kim 1, Sung Jun Jung 2, and
Korean J Physiol Pharmacol Vol 12: 315-321, December, 2008 Eugenol Inhibits ATP-induced P2X Currents in Trigeminal Ganglion Neurons Hai Ying Li 1, Byung Ky Lee 1, Joong Soo Kim 1, Sung Jun Jung 2, and
Cancer-induced bone pain
 Cancer-induced bone pain Common Prevalent in particular cancers: breast (73%), prostate (68%), thyroid (42%), lung (36%), renal (35%), colon (5%) Correlates with an increased morbidity Reduced performance
Cancer-induced bone pain Common Prevalent in particular cancers: breast (73%), prostate (68%), thyroid (42%), lung (36%), renal (35%), colon (5%) Correlates with an increased morbidity Reduced performance
ANATOMY AND PHYSIOLOGY OF NEURONS. AP Biology Chapter 48
 ANATOMY AND PHYSIOLOGY OF NEURONS AP Biology Chapter 48 Objectives Describe the different types of neurons Describe the structure and function of dendrites, axons, a synapse, types of ion channels, and
ANATOMY AND PHYSIOLOGY OF NEURONS AP Biology Chapter 48 Objectives Describe the different types of neurons Describe the structure and function of dendrites, axons, a synapse, types of ion channels, and
Chapter 8 Nervous System
 Chapter 8 Nervous System Two message centers: Functions of these systems: 1. * 2. * Overview of the Nervous System Parts: General Functions: Functions Sensory input: Sensation via nerves Integration: interpretation
Chapter 8 Nervous System Two message centers: Functions of these systems: 1. * 2. * Overview of the Nervous System Parts: General Functions: Functions Sensory input: Sensation via nerves Integration: interpretation
HUMAN MOTOR CONTROL. Emmanuel Guigon
 HUMAN MOTOR CONTROL Emmanuel Guigon Institut des Systèmes Intelligents et de Robotique Université Pierre et Marie Curie CNRS / UMR 7222 Paris, France emmanuel.guigon@upmc.fr e.guigon.free.fr/teaching.html
HUMAN MOTOR CONTROL Emmanuel Guigon Institut des Systèmes Intelligents et de Robotique Université Pierre et Marie Curie CNRS / UMR 7222 Paris, France emmanuel.guigon@upmc.fr e.guigon.free.fr/teaching.html
Clarke's Column Neurons as the Focus of a Corticospinal Corollary Circuit. Supplementary Information. Adam W. Hantman and Thomas M.
 Clarke's Column Neurons as the Focus of a Corticospinal Corollary Circuit Supplementary Information Adam W. Hantman and Thomas M. Jessell Supplementary Results Characterizing the origin of primary
Clarke's Column Neurons as the Focus of a Corticospinal Corollary Circuit Supplementary Information Adam W. Hantman and Thomas M. Jessell Supplementary Results Characterizing the origin of primary
Collin County Community College BIOL Week 5. Nervous System. Nervous System
 Collin County Community College BIOL 2401 Week 5 Nervous System 1 Nervous System The process of homeostasis makes sure that the activities that occur in the body are maintained within normal physiological
Collin County Community College BIOL 2401 Week 5 Nervous System 1 Nervous System The process of homeostasis makes sure that the activities that occur in the body are maintained within normal physiological
Supplementary Figure 1
 Supplementary Figure 1 The average sigmoid parametric curves of capillary dilation time courses and average time to 50% peak capillary diameter dilation computed from individual capillary responses averaged
Supplementary Figure 1 The average sigmoid parametric curves of capillary dilation time courses and average time to 50% peak capillary diameter dilation computed from individual capillary responses averaged
QUIZ/TEST REVIEW NOTES SECTION 7 NEUROPHYSIOLOGY [THE SYNAPSE AND PHARMACOLOGY]
![QUIZ/TEST REVIEW NOTES SECTION 7 NEUROPHYSIOLOGY [THE SYNAPSE AND PHARMACOLOGY] QUIZ/TEST REVIEW NOTES SECTION 7 NEUROPHYSIOLOGY [THE SYNAPSE AND PHARMACOLOGY]](/thumbs/86/94924826.jpg) QUIZ/TEST REVIEW NOTES SECTION 7 NEUROPHYSIOLOGY [THE SYNAPSE AND PHARMACOLOGY] Learning Objectives: Explain how neurons communicate stimulus intensity Explain how action potentials are conducted along
QUIZ/TEST REVIEW NOTES SECTION 7 NEUROPHYSIOLOGY [THE SYNAPSE AND PHARMACOLOGY] Learning Objectives: Explain how neurons communicate stimulus intensity Explain how action potentials are conducted along
Basic Neuroscience. Sally Curtis
 The Physiology of Pain Basic Neuroscience Sally Curtis sac3@soton.ac.uk The behaviour of humans is a result of the actions of nerves. Nerves form the basis of Thoughts, sensations and actions both reflex
The Physiology of Pain Basic Neuroscience Sally Curtis sac3@soton.ac.uk The behaviour of humans is a result of the actions of nerves. Nerves form the basis of Thoughts, sensations and actions both reflex
LECTURE STRUCTURE ASC171 NERVOUS SYSTEM PART 1: BACKGROUND 26/07/2015. Module 5
 LECTURE STRUCTURE PART 1: Background / Introduction PART 2: Structure of the NS, how it operates PART 3: CNS PART 4: PNS Why did the action potential cross the synaptic junction? To get to the other side
LECTURE STRUCTURE PART 1: Background / Introduction PART 2: Structure of the NS, how it operates PART 3: CNS PART 4: PNS Why did the action potential cross the synaptic junction? To get to the other side
Chapter 7. The Nervous System: Structure and Control of Movement
 Chapter 7 The Nervous System: Structure and Control of Movement Objectives Discuss the general organization of the nervous system Describe the structure & function of a nerve Draw and label the pathways
Chapter 7 The Nervous System: Structure and Control of Movement Objectives Discuss the general organization of the nervous system Describe the structure & function of a nerve Draw and label the pathways
Nervous System. 2. Receives information from the environment from CNS to organs and glands. 1. Relays messages, processes info, analyzes data
 Nervous System 1. Relays messages, processes info, analyzes data 2. Receives information from the environment from CNS to organs and glands 3. Transmits impulses from CNS to muscles and glands 4. Transmits
Nervous System 1. Relays messages, processes info, analyzes data 2. Receives information from the environment from CNS to organs and glands 3. Transmits impulses from CNS to muscles and glands 4. Transmits
Lecture VIII. The Spinal Cord, Reflexes and Brain Pathways!
 Reflexes and Brain Bio 3411! Monday!! 1! Readings! NEUROSCIENCE 5 th ed: Review Chapter 1 pp. 11-21;!!Read Chapter 9 pp. 189-194, 198! THE BRAIN ATLAS 3 rd ed:! Read pp. 4-17 on class web site! Look at
Reflexes and Brain Bio 3411! Monday!! 1! Readings! NEUROSCIENCE 5 th ed: Review Chapter 1 pp. 11-21;!!Read Chapter 9 pp. 189-194, 198! THE BRAIN ATLAS 3 rd ed:! Read pp. 4-17 on class web site! Look at
Chapter 7. Objectives
 Chapter 7 The Nervous System: Structure and Control of Movement Objectives Discuss the general organization of the nervous system Describe the structure & function of a nerve Draw and label the pathways
Chapter 7 The Nervous System: Structure and Control of Movement Objectives Discuss the general organization of the nervous system Describe the structure & function of a nerve Draw and label the pathways
Neurons, Synapses, and Signaling
 Neurons, Synapses, and Signaling The Neuron is the functional unit of the nervous system. Neurons are composed of a cell body, which contains the nucleus and organelles; Dendrites which are extensions
Neurons, Synapses, and Signaling The Neuron is the functional unit of the nervous system. Neurons are composed of a cell body, which contains the nucleus and organelles; Dendrites which are extensions
