P2X receptors-mediated cytosolic phospholipase A 2 activation in primary afferent sensory neurons contributes to neuropathic pain
|
|
|
- Millicent Hardy
- 5 years ago
- Views:
Transcription
1 Journal of Neurochemistry, 2007, 103, doi: /j x P2X receptors-mediated cytosolic phospholipase A 2 activation in primary afferent sensory neurons contributes to neuropathic pain Makoto Tsuda, 1 Shigeo Hasegawa 1 and Kazuhide Inoue Department of Molecular and System Pharmacology, Graduate School of Pharmaceutical Sciences, Kyushu University, Higashi, Fukuoka, Japan Abstract Activation of P2X 3 and P2X 2/3 receptors (P2X 3 R/P2X 2/3 R), ionotropic ATP receptor subtypes, in primary sensory neurons is involved in neuropathic pain, a debilitating chronic pain that occurs after peripheral nerve injury. However, the underlying mechanisms remain unknown. We investigated the role of cytosolic phospholipase A 2 (cpla 2 ) as a downstream molecule that mediates the P2X 3 R/P2X 2/3 R-dependent neuropathic pain. We found that applying ATP to cultured dorsal root ganglion (DRG) neurons increased the level of Ser505-phosphorylated cpla 2 and caused translocation of Ser505-phosphorylated cpla 2 to the plasma membrane. The ATP-induced cpla 2 activation was inhibited by a selective antagonist of P2X 3 R/P2X 2/3 R and by a selective inhibitor of cpla 2. In the DRG in vivo, the number of cpla 2 -activated neurons was strikingly increased after peripheral nerve injury but not after peripheral inflammation produced by complete Freund s adjuvant. Pharmacological blockade of P2X 3 R/P2X 2/3 R reversed the nerve injury-induced cpla 2 activation in DRG neurons. Moreover, administering the cpla 2 inhibitor near the DRG suppressed nerve injury-induced tactile allodynia, a hallmark of neuropathic pain. Our results suggest that P2X 3 R/P2X 2/3 R-dependent cpla 2 activity in primary sensory neurons is a key event in neuropathic pain and that cpla 2 might be a potential target for treating neuropathic pain. Keywords: cytosolic phospholipase A 2, neuropathic pain, P2X receptor, primary afferent sensory neurons. J. Neurochem. (2007) 103, A growing body of evidence has indicated that P2X receptors, a family of ligand-gated cation channels activated by extracellular ATP, play an important role in generating and modulating pain signaling (Burnstock 2006). Molecular cloning has so far identified seven genes encoding P2X receptor subunits (P2X 1 R P2X 7 R) (Ralevic and Burnstock 1998). In primary afferent sensory neurons, mrna or protein of almost all P2XRs has been detected (Collo et al. 1996; Kobayashi et al. 2005). However, electrophysiological studies investigating P2XR-mediated ATP responses indicated that homomeric P2X 3 R and heteromeric P2X 2/3 R might be the predominant functional P2X receptors in these cells (Rae et al. 1998; Ueno et al. 1999). Studies using mice lacking P2X 3 R or both P2X 2 R and P2X 3 R clearly corroborated these findings (Cockayne et al. 2000, 2005; Souslova et al. 2000). Various attempts to block P2XRs pharmacologically (Jarvis et al. 2001; McGaraughty et al. 2003) or to suppress their expression molecularly (Honore et al. 2002) and genetically (Cockayne et al. 2000; Souslova et al. 2000) did not affect the basal responses to physiological pain (e.g., noxious heat or mechanical stimuli), but rather preferentially suppressed neuropathic pain in response to peripheral nerve injury. Notably, acute systemic administration of the recently developed selective antagonist for P2X 3 and P2X 2/3 R (P2X 3 R/P2X 2/3 R) A , reversed nerve injury-induced abnormal hypersensitivity to innocuous stimuli (tactile allodynia), which is a hallmark of neuropathic pain syndrome (Jarvis et al. 2002; McGaraughty et al. 2005). As A poorly penetrates into the CNS (Wu et al. 2004; Sharp et al. 2006), these observations suggest a prominent role of peripheral P2X 3 R/P2X 2/3 R in primary sensory neurons for tactile allodynia caused by peripheral nerve injury. However, how P2X 3 R/P2X 2/3 R participates in neuropathic pain has remained unknown. Damaging primary sensory neurons causes ongoing discharges originating at the injured site, which are considered to contribute to neuropathic pain (Chen et al. 1999, 2001; Zhou et al. 2001). Applying ATP excites damaged neurons, however, the spontaneous ongoing discharges in damaged dorsal root ganglion (DRG) neurons are not suppressed by P2XR antagonists (Chen et al. 2001; Zhou et al. 2001), thus the role of the ATP-mediated direct neuronal excitation has remained unclear. As P2X 3 R/ Received April 6, 2007; revised manuscript received June 7, 2007; accepted July 2, Address correspondence and reprint requests to Kazuhide Inoue, Department of Molecular and System Pharmacology, Graduate School of Pharmaceutical Sciences, Kyushu University, Maidashi, Higashi, Fukuoka , Japan. inoue@phar.kyushu-u.ac.jp 1 These authors contributed equally to this work. Abbreviations used: CFA, complete Freund s adjuvant; cpla 2, cytosolic phosholipase A 2 ; cpla 2 -i, [N-{(2S,4R)-4-(Biphenyl-2-yl- methyl-isobutyl-amino)-1-[2-(2,4-difluorobenzoyl)-benzoyl]-pyrrolidin-2- ylmethyl}-3-[4-(2,4-dioxothiazolidin-5-ylidinemethyl)-phenyl]-acrylamide; DRG, dorsal root ganglion; IgG, immunoglobulin G; IR, immunoreactivity; P2X 3 R/P2X 2/3 R, P2X 3 and P2X 2/3 receptors; p-cpla 2, Ser505-phosphorylated cpla
2 Cytosolic phospholipase A 2 activation via P2X in neuropathic pain 1409 P2X 2/3 R are permeable to Ca 2+ as well as to Na + and K + (Ueno et al. 1998; Khakh et al. 2001; North 2002), we considered the possibility that stimulating P2X 3 R/P2X 2/3 R may lead to activation of Ca 2+ -dependent biochemical cascades in primary sensory neurons. In the present study, we investigated this possibility by focusing on the role of the Ca 2+ -dependent subclass of the PLA 2 family, namely cytosolic phospholipase A 2 (cpla 2 )/ group IVA PLA 2 (Shimizu et al. 2006). cpla 2 is a crucial enzyme for arachidonic acid and lysophospholipids release and the subsequent generation of phospholipid mediators such as prostaglandins and platelet activating factor (Spencer et al. 1998; Hirabayashi and Shimizu 2000). These mediators have been reported to cause sensitization of primary sensory neurons (Park and Vasko 2005) and to produce allodynic behaviors (Minami et al. 1992, 1994; Morita et al. 2004). It is shown here for the first time that nerve injury induces the activation of cpla 2 in primary afferent sensory neurons by P2X 3 R/P2X 2/3 R stimulation. Moreover, cpla 2 activation significantly contributes to nerve injury-induced tactile allodynia. Therefore, the present study suggests neuronal cpla 2 activation as a key intermediary event of P2X 3 R/P2X 2/3 R- dependent neuropathic pain after peripheral nerve injury. Materials and methods Animals Male Wistar rats ( g) were used. Animals were housed at a temperature of 22 ± 1 C with a 12-h light-dark cycle (light on 8:30 to 20:30), and fed food and water ad libitum. All of the animals used in the present study were obtained, housed, cared for and used in accordance with the guidelines of Kyushu University. Culture of rat primary DRG neurons The lumber DRGs (L1 6 segments) were removed from male Wistar rats and were treated in Dulbecco s modified eagle medium with 20 U/mL papain and 2 mg/ml collagenase type II for 1 h at 37 C. At the end of this treatment the enzyme solution was removed and the DRGs were mechanically dissociated by trituration through a Pasteur pipette in Dulbecco s modified eagle medium. They were suspended in F-12 Nutrient Mixture liquid supplemented with 10% horse serum, 2 mmol/l glutamine, 100 units/ml penicillin, 100 lg/ml streptomycin, 100 ng/ml nerve growth factor (Sigma, St Louis, MO, USA) and 100 ng/ml human glial cell-line derived neurotrophic factor (Promega, Madison, WI, USA). They were plated in 35 mm-diameter tissue culture dishes for western blotting or slide glasses coated with 100 lg/ml poly-l-lysine and 10 lg/ml laminin for immunocytochemistry and maintained in an atmosphere of 5% CO 2 /95% ambient air at 37 C for 72 h. Following incubation, the medium was removed, replaced with fresh medium without horse serum, nerve growth factor and human glial cell-line derived neurotrophic factor, and further cultured at 37 C for an additional 24 h. Reagents were added to DRG neuron cultures and incubated for the indicated time. After these treatments, the medium was removed and the cultures were scraped into ice-cold 125 mmol/l Tris-HCl (ph 7.4) containing 20% glycerol, 4% (w/v) sodium dodecyl sulfate, 0.025% (w/v) bromophenol blue and 5% 2-mercaptoethanol. Protein samples were subjected to polyacrylamide gel electrophoresis and transferred electrophoretically to polyvinylidine difluoride membranes. After blocking, the membranes were incubated with anti-phospho-cpla 2 (anti-p-cpla 2 ) antibody (1 : 1000, Cell Signaling, Beverly, MA, USA) or anti-cpla 2 antibody (1 : 1000, Cell Signaling) and then were incubated with horseradish peroxidase-conjugated anti-rabbit immunoglobulin G (IgG) antibody (1 : 1000, Amersham Biosciences, Buckinghamshire, UK). The blots were detected using a chemiluminescence method (ECL system; Amersham Biosciences). Immunocytochemistry was performed as follows. Immediately after treatment with ATP for 5 min, cells were fixed with 3.7% formaldehyde. After blocking, neurons were incubated with anti-pcpla 2 antibody (1 : 1000) and then were incubated with anti-rabbit IgG-conjugated Alexa Fluor 488 (1 : 1000, Molecular Probes, Eugene, OR, USA), followed by analysis with an LSM510 Imaging System (Zeiss, Oberkochen, Germany). To distinguish among cell size-specific changes, DRG neurons were characterized as small (< 25 lm), medium (25 35 lm), and large (> 35 lm)-sized neurons according to their diameters (Ueno et al. 1999). Neuropathic and inflammatory pain We used the spinal nerve injury model (Kim and Chung 1992) with some modifications (Tsuda et al. 2003): in male Wistar rats a unilateral L5 spinal nerve was tightly ligated and cut just distal to the ligature. For an inflammation pain model, complete Freund s adjuvant (CFA) (0.1 mg/200 ll, Sigma) was injected into the plantar surface of the left hindpaw using a 26-gauge needle. The mechanical allodynia was assessed by using calibrated von Frey filaments ( g) and the paw withdrawal threshold was determined as described previously (Tsuda et al. 2003). Immunohistochemistry Rats were deeply anesthetized by pentobarbital (100 mg/kg, i.p.) and perfused transcardially with 4% paraformaldehyde. DRG sections were removed, post-fixed with the same fixative, and placed in 30% sucrose solution for 24 h at 4 C. The DRG sections (15 lm) were incubated in a blocking solution [3% normal goat serum/0.3% Triton X-100/phosphate-buffered saline(-)] and then with anti-p-cpla 2 antibody (1 : 1000, Cell Signaling). Identification of the type of p-cpla 2 -translocated cells was performed with the following markers: for satellite glia/astrocytes, glial fibrillary acidic protein (1 : 500, Chemicon, Temecula, CA, USA); for neurons, neuronal nuclei (1 : 200, Chemicon) and microtubuleassociated protein 2 (1 : 1000, Chemicon). Neurofilament 200 (1 : 400, Sigma), a marker for myelinated A-fibers, was also used as the primary antibody. Following incubation, the DRG sections were incubated with secondary antibodies (anti-rabbit IgG-conjugated Alexa Fluor 488 or anti-mouse IgG-conjugated Alexa Fluor 546, 1 : 1000, Molecular Probes). The sections were then analyzed by a confocal microscope (LSM510, Zeiss). The number of p-cpla 2 - immunoreactive DRG neurons with translocation was counted in the L5 DRG ipsilateral to the nerve injury. The proportion of the p-cpla 2 -translocated neurons to the total number of DRG neurons was determined in each rat at each time point. For the size-
3 1410 M. Tsuda et al. distribution histogram data, measurement of the cross-sectional areas of p-cpla 2 -translocated neurons was made by using an LSM Image Browser (Zeiss) and only neurons with clearly visible nuclei were used for the quantification. To distinguish among cell sizespecific changes, DRG neurons were characterized as small (< 600 lm 2 ), medium ( lm 2 ), and large (> 1200 lm 2 )- sized neurons according to their cross-sectional areas (Obata et al. 2004). Drug treatment Rats were implanted with catheters for intrathecal injection according to the method described previously (Yaksh et al. 1980). Under isoflurane anesthesia, a sterile 32 gauge intrathecal catheter (ReCathCo) was inserted through the atlanto-occipital membrane and to the L4 or L5 DRG and externalized through the skin (Ji et al. 2002). After the experiments, we confirmed that the tip of the catheter was positioned near the L5 DRG. Rats were injected intrathecally with [N-{(2S,4R)-4-(Biphenyl-2-ylmethyl-isobutyl-amino)-1-[2-(2,4-difluorobenzoyl)-benzoyl]-pyrrolidin-2-ylmethyl}-3-[4-(2,4-dioxothiazolidin-5-ylidinemethyl)-phenyl]-acrylamide (cpla 2 -i, 2.5 nmol/ 10 ll, Calbiochem, San Diego, CA, USA) using a 25 ll Hamilton syringe with a 30-gauge needle once a day from day 0 (just before the nerve injury) to day 13. The paw withdrawal threshold was tested h after the injection of cpla 2 -i at 1, 3, 7 and 14 days post-injury. After the test on day 14, to examine the level of p-cpla 2 in injured DRG neurons in cpla 2 -i- and vehicle-treated groups using immunohistochemistry, the L5 DRG ipsilateral to the nerve injury was removed. For the experiment in which the effect of a single administration of cpla 2 -i on the established allodynia was examined on day 7 after nerve injury, behavioral test was performed immediately before and 45 min after the injection of cpla 2 -i (2.5 nmol/10 ll). A (100 lmol/kg, Sigma) was injected intravenously with a 26-gauge needle 14 days after the nerve injury. To examine the level and translocation of p-cpla 2 in the injured DRG neurons in the A and vehicle-treated groups, the rats were fixed 90 min after injection, and the L5 DRG ipsilateral to the nerve injury was removed and used for immunohistochemistry. Statistical analysis Statistical analyses of the results were made with Student s t test, Student s paired t test or the Mann Whitney U test. Results P2X 3 R/P2X 2/3 R-dependent activation of cpla 2 in cultured primary sensory neurons Full activation of cpla 2 requires two events: phosphorylation of serine residues (including serine505) and translocation of cpla 2 for accessing membrane phospholipids (Hirabayashi and Shimizu 2000). To investigate cpla 2 activation in DRG neurons, we applied ATP to these cells and assessed the level of cpla 2 phosphorylation by western blot analyses using an antibody that recognized Ser505- phosphorylated cpla 2 (p-cpla 2 ) (Su et al. 2004). We found that ATP (10 and 50 lmol/l) caused an increase in the level of p-cpla 2, with maximal effect at 10 lmol/l ATP (Fig. 1a). The bands corresponding to cpla 2 and p-cpla 2 were detected at about 110 kda, which were identical to their molecular weight (Clark et al. 1990; Nemenoff et al. 1993). The level of p-cpla 2 began to increase 3 min after the application of ATP and peaked at 5 min (Fig. 1b). The ATP-induced cpla 2 phosphorylation was prevented by A , a potent and selective antagonist for P2X 3 R/ P2X 2/3 R (Jarvis et al. 2002) (Fig. 1c) as well as by suramin, PPADS and TNP-ATP (data not illustrated). Applying a,bmethylene ATP, an agonist of P2X 1 - and P2X 3 -containing P2XRs (North and Surprenant 2000), also increased p-cpla 2 levels (Fig. 1d). When DRG neurons were pre-treated with the Ca 2+ chelator EGTA (5 and 10 mmol/l) prior to applying ATP, the ATP-induced cpla 2 phosphorylation was significantly inhibited (Fig. 1e). The increase of p-cpla 2 by ATP (Fig. 1g) and by a,b-methylene ATP (data not illustrated) was abolished by cpla 2 -i, a selective inhibitor for cpla 2 (Seno et al. 2000), showing the specificity of the used antibody. Immuocytochemical studies corroborated the western blotting results. Applying ATP caused an increase in p-cpla 2 -immunoreactivity (IR) at the vicinity of the plasma membrane of DRG neurons (Fig. 1f). The cell-size of p-cpla 2 -translocated DRG neurons ranged from small to large (small: 47/90, medium: 86/108 and large: 8/8). These results thus indicate that ATP activates cpla 2 via P2X 3 R/ P2X 2/3 R in cultured DRG neurons. Peripheral nerve injury induces cpla 2 activation in primary sensory neurons in vivo Recent evidence has suggested that P2X 3 R/P2X 2/3 R in DRG neurons play an important role in neuropathic pain (Tsuda et al. 2000; Barclay et al. 2002; Honore et al. 2002; Jarvis et al. 2002), predicting the activation of cpla 2 in DRG neurons under this condition. To determine the possible involvement of cpla 2 in DRG neurons, we first performed immunofluorescent analysis in DRG of rats that had been unilaterally injured in the fifth lumbar (L5) spinal nerve, a model of neuropathic pain (Kim and Chung 1992). Injury to the L5 nerve caused an increase of p-cpla 2 -IR in ipsilateral L5 DRG neurons (Fig. 2a). On the subcellular level, p- cpla 2 -IR did not accumulate around the nuclei (immunostained with the neuronal nuclear marker, neuronal nuclei) (Fig. 2b d), but rather accumulated at the edges of the area immunostained with the neuronal marker microtubule-associated protein 2 (Fig. 2e g). Accumulated p-cpla 2 -IR was never observed in glial fibrillary acidic protein-positive satellite glia/astrocytes (Fig. 2h j) in the contralateral L5 DRG (Fig. 2a) or either side of the non-injured L4 DRG (data not illustrated). In agreement with our in vitro data, cpla 2 -i treatment (see below) markedly decreased p-cpla 2 - IR, indicating again the specificity of p-cpla 2 -IR shown here. The intraplantar injection of CFA is a model of
4 Cytosolic phospholipase A 2 activation via P2X in neuropathic pain 1411 (a) ATP (µmol/l) for 5 min (b) (c) (µmol/l) ATP (µmol/l) for 5 min (µmol/l) (d) (e) (f) (µmol/l) (mmol/l) (g) (µmol/l) (µmol/l) (mmol/l) Fig. 1 Cytosolic phosholipase A 2 (cpla 2 ) is activated via stimulating P2X 3 and P2X 2/3 receptors in rat primary dorsal root ganglion (DRG) neurons. (a g) Western blot (a e, g) and immunocytochemical (f) analyses of Ser505-phosphorylated cpla 2 (p-cpla 2 ) protein after applying ATP or its analog to rat primary DRG neurons. The primary DRG culture was incubated with ATP (1 50 lmol/l) or a,b-methylene ATP (abmeatp; 100 lmol/l) for 1 10 min. A , cpla 2 -i or EGTA was added to the neurons 10 or 30 min before the application of ATP. The total cpla 2 protein loaded on each lane was also detected as loading controls. The bar graphs show the relative values of p-cpla 2 protein induction with respect to control, after normalizing for the cpla 2 protein levels. *p < 0.05, **p < 0.01 compared with the control group. #p < 0.05 compared with the ATP-stimulated group. (f) Increase in the level of p-cpla 2 immunofluorescence and translocation of p-cpla 2 to the plasma membrane in neurons 5 min after the application of 10 lmol/l ATP compared with control. Scale bar, 20 lm. Similar results were observed in each of three experiments. inflammatory pain (Barclay et al. 2002). Although CFA produced a significant decrease in the paw withdrawal threshold (day 0: 14.4 ± 0.6 g, day 7: 2.0 ± 0.3 g, p < 0.001) after 7 days, we did not observe any change in the level and the distribution of p-cpla 2 -IR in both the L5 (Fig. 2k) and L4 DRG (data not illustrated). These results indicate that peripheral nerve injury but not persistent peripheral inflammation causes activation of cpla 2 in DRG neurons. To determine the cell-size distribution of DRG neurons with p-cpla 2 -translocation, we measured the cross-sectional areas of DRG neurons with clearly visible nuclei. Translocated p-cpla 2 was observed in medium- to large-sized neurons as well as in small-sized neurons in the ipsilateral L5 DRG (Fig. 2l). The percentage of p-cpla 2 - translocated DRG neurons for each cell-size, however, was quite different: a much higher proportion was seen in the large-sized neurons than in the small-sized DRG neurons (Fig. 2l). Large-sized cells positive for p-cpla 2 -IR were double-labeled with the marker of myelinated afferent neurons neurofilament 200 (Obata et al. 2004) (Fig. 2m o), suggesting that cpla 2 is predominantly activated myelinated DRG neurons ipsilateral to the injury. P2X 3 R/P2X 2/3 R are involved in nerve injury-induced cpla 2 activation Administration of A (100 lmol/kg, i.v.) markedly suppressed both the level and translocation of p-cpla 2 -IR in injured DRG neurons within 90 min (Fig. 3a) and moreover significantly reduced the number of DRG neurons showing translocated p-cpla 2 in response to nerve injury (p < 0.001, Fig. 3b). In addition, A administration also reduced tactile allodynia in rats as previously demonstrated (Jarvis et al. 2002) (data not illustrated). These data thus strongly indicate that the activation of cpla 2 in damaged DRG neurons following peripheral nerve injury might result from P2X 3 R/P2X 2/3 R stimulation. Inhibition of cpla 2 activation suppresses nerve injury-induced tactile allodynia To determine functional relevance of cpla 2 activation in neuropathic pain, we counted the number of cells with activated cpla 2 in the L5 DRG at various time points subsequent to the nerve injury. The number of cells was increased as early as day 3 (p < 0.001) and the peak was observed on day 14 (p < 0.001) (Fig. 4). The number of
5 1412 M. Tsuda et al. (a) (k) (b) (c) (d) (l) ( ) (e) (f) (g) (h) (i) (j) (m) (n) (o) Fig. 2 Cytosolic phosholipase A 2 (cpla 2 ) is activated in dorsal root ganglion (DRG) neurons after injury to the L5 spinal nerve. (a) Visualization of the Ser505-phosphorylated cpla 2 (p-cpla 2 ) protein detected by p-cpla 2 antibody in the L5 DRG by immunofluorescence analysis with a confocal microscope. Photomicrographs showing the p-cpla 2 immunofluorescence in the L5 DRG 14 days after the nerve injury. (b j) Double immunofluorescence labeling of p-cpla 2 with neuronal nuclei (NeuN) (b d) and microtubule-associated protein 2 (MAP2) (e g), markers of neurons; glial fibrillary acidic protein (GFAP) (h j), a marker of satellite glia/astrocytes in the ipsilateral L5 DRG 14 days after nerve injury. (k) Immunohistochemical analysis of p-cpla 2 protein in the L5 DRG 7 days after injection of complete Freund s adjuvant into the plantar surface of the hindpaw, an inflammatory pain model. (l) Size-distribution histogram of p-cpla 2 - translocated neurons (bar graph) and the percentage of p-cpla 2 - translocated DRG neurons relative to the total number of neurons (line graph) in the ipsilateral L5 DRG 14 days after nerve injury. A crosssectional area of about 650 neurons with clearly visible nuclei was measured. Open bars, total DRG neurons; filled bars, p-cpla 2 - translocated neurons (p-cpla 2 neurons). (m o) Double immunofluorescence labeling of p-cpla 2 with neurofilament 200 (NF200), a marker of myelinated A-fibers, in the ipsilateral L5 DRG 14 days after the nerve injury. Scale bars, 50 lm. neurons in the contralateral L5 DRG was not different from that in naïve rats (data not illustrated). The time-course of the change and the bilateral difference in p-cpla 2 -translocated cells in the DRG were matched closely with the emergence of tactile allodynia (Fig. 4). Furthermore, we injected the selective cpla 2 inhibitor cpla 2 -i through a catheter whose tip was positioned near the L5 DRG (Ji et al. 2002) and examined its effect on the development of tactile allodynia after nerve injury. Rats were treated with cpla 2 -i (2.5 nmol/ 10 ll, n = 5) or vehicle (5% dimethyl sulfoxide 10 ll, n = 5) once a day for 14 days. Vehicle-treated rats displayed a marked decrease in the paw withdrawal threshold following nerve injury (p < 0.001) (Fig. 5a). In contrast, rats treated with cpla 2 -i showed only a slight, but not significant, decrease in the threshold after nerve injury. The significant difference in the threshold between cpla 2 -i and vehicletreated rats was observed on days 3, 7 and 14 after the injury (day 3: p < 0.001, day 7 and 14: p < 0.01) (Fig. 5a). In addition, on day 14 the level of p-cpla 2 -IR in the ipsilateral DRG of cpla 2 -i-treated rats was much lower than in vehicle-treated rats (Fig. 5c). Furthermore, a single administration of cpla 2 -i (2.5 nmol/10 ll, n = 8) near the DRG 7 days after nerve injury also significantly suppressed the expression of tactile allodynia within 45 min (p < 0.001, Fig. 5b). Alteration in motor behavior after cpla 2 -i treatment was not observed (data not shown). Nor did cpla 2 -i affect the paw withdrawal threshold on the side contralateral to the nerve injury: at 14 days the threshold of nerve-injured rats treated with vehicle was 13.1 ± 1.2 g compared with 13.5 ± 1.0 g at 14 days of cpla 2 -i-treated rats (p > 0.1). These results together indicate that inhibiting cpla 2 activation by cpla 2 -i prevents the development and reduces the expression of tactile allodynia caused by nerve injury. Discussion In the present study, we provide the first evidence that cpla 2 is activated in DRG neurons after nerve injury and that the activation of cpla 2 is a key intermediary event of P2X 3 R/ P2X 2/3 R-dependent tactile allodynia, a major behavioral
6 Cytosolic phospholipase A 2 activation via P2X in neuropathic pain 1413 (a) (b) (a) (b) Fig. 3 Cytosolic phosholipase A 2 (cpla 2 ) is activated via stimulating P2X 3 and P2X 2/3 receptors in dorsal root ganglion (DRG) neurons after nerve injury. (a) Photomicrographs showing the Ser505-phosphorylated cpla 2 (p-cpla 2 ) immunofluorescence in the ipsilateral L5 DRG of vehicle- and A treated rats 14 days after the nerve injury. Rats were administered A (100 lmol/kg) or vehicle (PBS) intravenously 90 min before fixation. Scale bar, 50 lm. (b) The number of p-cpla 2 -translocated DRG neurons in the ipsilateral L5 DRG 90 min after administering A About 630 neuron profiles were counted in eight to ten randomly chosen sections from three to four rats in each group. Results are percentages (Mean ± SEM) of p-cpla 2 -translocated DRG neurons (p-cpla 2 neurons) relative to the total number of neurons. ***p < compared with the vehicletreated group. Fig. 4 The time-course of the change in phospho-cytosolic phosholipase A 2 (cpla 2 )-translocated dorsal root ganglion neurons was correlated with the emergence of tactile allodynia. Time course of change in paw withdrawal threshold (Mean ± SEM: bar graph) and the percentages of p-cpla 2 -translocated neurons relative to the total number of neurons in the L5 dorsal root ganglion ipsilateral to the nerve injury (Mean ± SEM: line graph). The paw withdrawal threshold of tactile stimulation to the ipsilateral hindpaw was examined using von Frey filaments neuron profiles were counted in eight to twelve randomly chosen sections from three to four rats at each time point. ***p < 0.001, ###p < compared with pre-injury baseline (Pre). consequence of nerve injury. ATP-dependent phosphorylation of cpla 2 and its translocation in primary cultured DRG neurons was inhibited either by the selective P2X 3 R/P2X 2/3 R antagonist A or by the Ca 2+ chelator EGTA, demonstrating that ATP activates cpla 2 via P2X 3 R/P2X 2/ 3R expressed on DRG neurons in a Ca 2+ -dependent manner. Interestingly, neither the level of the phosphorylated cpla 2 nor its localization was altered in DRG neurons in an animal model of chronic inflammatory pain. It thus seems likely that (c) Fig. 5 Selective cytosolic phosholipase A 2 (cpla 2 ) inhibitor suppresses tactile allodynia and cpla 2 activation caused by injury to the L5 spinal nerve. (a) Effect of a selective cpla 2 inhibitor, [N-{(2S,4R)- 4-(Biphenyl-2-ylmethyl-isobutyl-amino)-1-[2-(2,4-difluorobenzoyl)-benzoyl]-pyrrolidin-2-ylmethyl}-3-[4-(2,4-dioxothiazolidin-5-ylidinemethyl) -phenyl]-acrylamide (cpla 2 -i), on the development of nerve injuryinduced tactile allodynia. cpla 2 -i (2.5 nmol, n = 5) or vehicle (5% DMSO in PBS, n = 5) was administered near the dorsal root ganglion once a day for 14 days. Results are Mean ± SEM of the paw withdrawal threshold. Filled squares, cpla 2 -i; open squares, vehicle. ***p < compared with the threshold on day 0. ##p < 0.01, ###p < compared with the threshold of the vehicle-treated group. (b) Effect of a single administration of cpla 2 -i (2.5 nmol, n = 8) on the decrease in paw withdrawal threshold (Mean ± SEM) 7 days after nerve injury. ***p < compared with pre-injury baseline (Pre). ###p < compared with the threshold on day 7. (c) Photomicrographs showing the p-cpla 2 immunofluorescence in the ipsilateral L5 dorsal root ganglion of vehicle-treated rats and cpla 2 -i-treated rats 14 days after the nerve injury. Scale bar, 50 lm. cpla 2 activation is specifically associated with nerve injury. This notion is consistent with our data showing that cpla 2 was only activated in L5 DRG neurons, but not in L4 DRG neurons, of L5 spinal nerve injured rats. We also found that cpla 2 activation in nerve-injured rats was reversed by acute administration of A , suggesting that the cpla 2 activation depends upon ongoing signaling of ATP-activated P2X 3 R/P2X 2/3 R. Putative sources of ATP could be Schwann cells (Liu and Bennett 2003), satellite glia (Martin 1992), or injured nerves (Franke and Illes 2006). Our present behavioral study reveals the functional relevance of cpla 2 in neuropathic pain: a selective inhibition of cpla 2 activation in DRG neurons significantly suppressed the development and expression of tactile allodynia. Basal sensitivity to mechanical stimuli and motor function were not affected by this treatment. The time-dependent activation of cpla 2 in damaged DRG neurons paralleled the emergence of tactile allodynia, providing substantial support for its contribution
7 1414 M. Tsuda et al. to neuropathic pain. L5 dorsal rhizotomy reduces tactile allodynia following injury of the L5 spinal nerve, indicating an important role of damaged DRG neurons in neuropathic pain (Yoon et al. 1996; Obata et al. 2004). Our data now indicate that P2X 3 R/P2X 2/3 R-dependent activation of cpla 2 in these cells is important for the development and expression of tactile allodynia after peripheral nerve injury. Our present results showing that cpla 2 activation was observed mainly in medium- to large-sized DRG neurons seem to be partly inconsistent with previous observations that P2X 3 R is expressed mainly in small- to medium-sized DRG neurons (Bradbury et al. 1998; Vulchanova et al. 1998; Novakovic et al. 1999; Ramer et al. 2001; Kobayashi et al. 2005). However, several studies confirmed that large-sized DRG neurons also expressed P2X 3 R at both protein and mrna albeit at low levels (Novakovic et al. 1999; Ramer et al. 2001; Kage et al. 2002; Kobayashi et al. 2005). Interestingly, a recent work has demonstrated that nerve damage decreased P2X 3 R protein expression in small-sized DRG neurons, but not in large-sized DRG neurons (Kage et al. 2002). Furthermore, in healthy DRG only 20% of the neurons express P2X 2 R (Kim et al. 2003; Kobayashi et al. 2005), but the number of P2X 2 R-expressing neurons increased to 73% after peripheral nerve injury including large-sized neurons (Kim et al. 2003). These findings and the data presented here suggest that following peripheral nerve injury, functional P2X 3 R and/or P2X 2/3 R may be expressed in medium- to large-sized DRG neurons to activate cpla 2. This is supported by previous data showing that in response to nerve injury ATP-evoked firing is observed ectopically in injured Ab-primary afferent fibers (Chen et al. 1999, 2001; Zhou et al. 2001). Alternatively, it is possible that ATP- P2X 3 R/P2X 2/3 R signaling may indirectly cause cpla 2 activation in large-sized DRG neurons through an unknown neuron-to-neuron communication mechanism in the DRG. The subcellular targeting of cpla 2 is important for accessing membrane lipids to release arachidonic acid (Spencer et al. 1998; Hirabayashi and Shimizu 2000). In the present in vitro and in vivo studies we found the cpla 2 translocation to the plasma membrane in DRG neurons. These results seem to be unique, as almost all the previous in vitro data have shown that cpla 2 translocates to the cytoplasmic membrane of the perinuclear region (Schievella et al. 1995; Evans et al. 2001; Stahelin et al. 2003). The translocation to the cytoplasmic membrane might be because of the binding specificity of its C2 domain to phosphatidylcholine that is abundant at the membranes of the perinuclear region (Stahelin et al. 2003). Considering the fact that the cpla 2 translocation to the perinuclear region was demonstrated in non-neuronal cells (Schievella et al. 1995; Evans et al. 2001; Stahelin et al. 2003), it is possible that the lipid component in damaged neuronal cells could be somewhat different from that in non-neuronal cells. Alternatively, from a recent work showing that physical association of cpla 2 with components of the NADPH oxidase system results in translocation to the plasma membrane (Shmelzer et al. 2003), additional signals and/or proteins may have roles in the P2X 3 R/P2X 2/3 R-mediated cpla 2 translocation to the plasma membrane of DRG neurons after nerve injury. Putative candidate signaling molecules downstream of cpla 2 activation could be prostaglandins (Minami et al. 1992, 1994), platelet activating factor (Morita et al. 2004) or lysophosphatidic acid (Inoue et al. 2004), all of which have been reported to produce tactile allodynia. Phospholipid products mediated by cpla 2 activation may be secreted from DRG neurons and, in turn, increase the excitation of DRG neurons. The expression of many receptors for lipid mediators in DRG neurons (Park and Vasko 2005) supports this notion. Alternatively, cpla 2 -mediated products may also affect the functions of neighboring cells including sympathetic nerve terminals that sprout into the DRG and form abnormal connections to DRG neurons after nerve injury (McLachlan et al. 1993; Chung et al. 1996). Several phospholipids have been implicated as retrograde substances that affect neurotransmission at pre-synapses in the nervous system (Chen and Bazan 2005). Thus, cpla 2 -mediated products might enhance the neuronal connection between sympathetic terminals and DRG neurons and thereby increase the sympathetic nerve-dependent excitation of sensory neurons. Studies in our laboratory determining cpla 2 -mediated products and their roles in neuropathic pain are currently underway. In summary, the present study shows for the first time that stimulating P2X 3 R/P2X 2/3 R activates cpla 2 in primary afferent sensory neurons and identifies cpla 2 activity as a key event in the pathogenesis of neuropathic pain. In our behavioral study, cpla 2 -i suppressed neuropathic allodynia without any substantial deficit in the basal sensitivity. Thus, interfering with the cpla 2 activity may also be a potential therapeutic target for treating neuropathic pain. Acknowledgements We thank Dr Knut Biber for critical reading and suggestions. This work was supported by grants from the Ministry of Education, Culture, Sports, Science and Technology of Japan (to MT, KI). References Barclay J., Patel S., Dorn G. et al. (2002) Functional downregulation of P2X 3 receptor subunit in rat sensory neurons reveals a significant role in chronic neuropathic and inflammatory pain. J. Neurosci. 22, Bradbury E. J., Burnstock G. and McMahon S. B. (1998) The expression of P2X 3 purinoreceptors in sensory neurons: effects of axotomy and glial-derived neurotrophic factor. Mol. Cell. Neurosci. 12, Burnstock G. (2006) Purinergic P2 receptors as targets for novel analgesics. Pharmacol. Ther. 110,
8 Cytosolic phospholipase A 2 activation via P2X in neuropathic pain 1415 Chen C. and Bazan N. G. (2005) Lipid signaling: sleep, synaptic plasticity, and neuroprotection. Prostaglandins Other Lipid Mediat 77, Chen Y., Shu Y. and Zhao Z. (1999) Ectopic purinergic sensitivity develops at sites of chronic nerve constriction injury in rat. Neuroreport. 10, Chen Y., Zhang Y. H. and Zhao Z. Q. (2001) Novel purinergic sensitivity develops in injured sensory axons following sciatic nerve transection in rat. Brain Res. 911, Chung K., Lee B. H., Yoon Y. W. and Chung J. M. (1996) Sympathetic sprouting in the dorsal root ganglia of the injured peripheral nerve in a rat neuropathic pain model. J. Comp. Neurol. 376, Clark J. D., Milona N. and Knopf J. L. (1990) Purification of a 110- kilodalton cytosolic phospholipase A 2 from the human monocytic cell line U937. Proc. Natl Acad. Sci. USA 87, Cockayne D. A., Hamilton S. G., Zhu Q. M. et al. (2000) Urinary bladder hyporeflexia and reduced pain-related behaviour in P2X 3 - deficient mice. Nature 407, Cockayne D. A., Dunn P. M., Zhong Y. et al. (2005) P2X 2 knockout mice and P2X 2 /P2X 3 double knockout mice reveal a role for the P2X 2 receptor subunit in mediating multiple sensory effects of ATP. J. Physiol. 567, Collo G., North R. A., Kawashima E., Merlo-Pich E., Neidhart S., Surprenant A. and Buell G. (1996) Cloning of P2X 5 and P2X 6 receptors and the distribution and properties of an extended family of ATP-gated ion channels. J. Neurosci. 16, Evans J. H., Spencer D. M., Zweifach A. and Leslie C. C. (2001) Intracellular calcium signals regulating cytosolic phospholipase A 2 translocation to internal membranes. J. Biol. Chem. 276, Franke H. and Illes P. (2006) Involvement of P2 receptors in the growth and survival of neurons in the CNS. Pharmacol. Ther. 109, Hirabayashi T. and Shimizu T. (2000) Localization and regulation of cytosolic phospholipase A 2. Biochim. Biophys. Acta 1488, Honore P., Kage K., Mikusa J., Watt A. T., Johnston J. F., Wyatt J. R., Faltynek C. R., Jarvis M. F. and Lynch K. (2002) Analgesic profile of intrathecal P2X 3 antisense oligonucleotide treatment in chronic inflammatory and neuropathic pain states in rats. Pain 99, Inoue M., Rashid M. H., Fujita R., Contos J. J., Chun J. and Ueda H. (2004) Initiation of neuropathic pain requires lysophosphatidic acid receptor signaling. Nat. Med. 10, Jarvis M. F., Wismer C. T., Schweitzer E., Yu H., van Biesen T., Lynch K. J., Burgard E. C. and Kowaluk E. A. (2001) Modulation of BzATP and formalin induced nociception: attenuation by the P2X receptor antagonist, TNP-ATP and enhancement by the P2X 3 allosteric modulator, cibacron blue. Br. J. Pharmacol. 132, Jarvis M. F., Burgard E. C., McGaraughty S. et al. (2002) A , a novel potent and selective non-nucleotide antagonist of P2X 3 and P2X 2/3 receptors, reduces chronic inflammatory and neuropathic pain in the rat. Proc. Natl Acad. Sci. USA 99, Ji R. R., Samad T. A., Jin S. X., Schmoll R. and Woolf C. J. (2002) p38 MAPK activation by NGF in primary sensory neurons after inflammation increases TRPV1 levels and maintains heat hyperalgesia. Neuron 36, Kage K., Niforatos W., Zhu C. Z., Lynch K. J., Honore P. and Jarvis M. F. (2002) Alteration of dorsal root ganglion P2X 3 receptor expression and function following spinal nerve ligation in the rat. Exp. Brain Res. 147, Khakh B. S., Burnstock G., Kennedy C., King B. F., North R. A., Seguela P., Voigt M. and Humphrey P. P. (2001) International union of pharmacology. XXIV. Current status of the nomenclature and properties of P2X receptors and their subunits. Pharmacol. Rev. 53, Kim S. H. and Chung J. M. (1992) An experimental model for peripheral neuropathy produced by segmental spinal nerve ligation in the rat. Pain 50, Kim C., Chung J. M. and Chung K. (2003) Changes in the gene expression of six subtypes of P2X receptors in rat dorsal root ganglion after spinal nerve ligation. Neurosci. Lett. 337, Kobayashi K., Fukuoka T., Yamanaka H., Dai Y., Obata K., Tokunaga A. and Noguchi K. (2005) Differential expression patterns of mrnas for P2X receptor subunits in neurochemically characterized dorsal root ganglion neurons in the rat. J. Comp. Neurol. 481, Liu G. J. and Bennett M. R. (2003) ATP secretion from nerve trunks and Schwann cells mediated by glutamate. Neuroreport 14, Martin D. L. (1992) Synthesis and release of neuroactive substances by glial cells. Glia 5, McGaraughty S., Wismer C. T., Zhu C. Z., Mikusa J., Honore P., Chu K. L., Lee C. H., Faltynek C. R. and Jarvis M. F. (2003) Effects of A , a novel and selective P2X 3 /P2X 2/3 receptor antagonist, on neuropathic, inflammatory and chemogenic nociception following intrathecal and intraplantar administration. Br. J. Pharmacol. 140, McGaraughty S., Honore P., Wismer C. T., Mikusa J., Zhu C. Z., McDonald H. A., Bianchi B., Faltynek C. R. and Jarvis M. F. (2005) Endogenous opioid mechanisms partially mediate P2X 3 / P2X 2/3 -related antinociception in rat models of inflammatory and chemogenic pain but not neuropathic pain. Br. J. Pharmacol. 146, McLachlan E. M., Janig W., Devor M. and Michaelis M. (1993) Peripheral nerve injury triggers noradrenergic sprouting within dorsal root ganglia. Nature 363, Minami T., Uda R., Horiguchi S., Ito S., Hyodo M. and Hayaishi O. (1992) Allodynia evoked by intrathecal administration of prostaglandin F 2 alpha to conscious mice. Pain 50, Minami T., Uda R., Horiguchi S., Ito S., Hyodo M. and Hayaishi O. (1994) Allodynia evoked by intrathecal administration of prostaglandin E 2 to conscious mice. Pain 57, Morita K., Morioka N., Abdin J., Kitayama S., Nakata Y. and Dohi T. (2004) Development of tactile allodynia and thermal hyperalgesia by intrathecally administered platelet-activating factor in mice. Pain 111, Nemenoff R. A., Winitz S., Qian N. X., Van Putten V., Johnson G. L. and Heasley L. E. (1993) Phosphorylation and activation of a high molecular weight form of phospholipase A 2 by p42 microtubuleassociated protein 2 kinase and protein kinase C. J. Biol. Chem. 268, North R. A. (2002) Molecular physiology of P2X receptors. Physiol. Rev. 82, North R. A. and Surprenant A. (2000) Pharmacology of cloned P2X receptors. Annu. Rev. Pharmacol. Toxicol. 40, Novakovic S. D., Kassotakis L. C., Oglesby I. B., Smith J. A., Eglen R. M., Ford A. P. and Hunter J. C. (1999) Immunocytochemical localization of P2X 3 purinoceptors in sensory neurons in naive rats and following neuropathic injury. Pain 80, Obata K., Yamanaka H., Kobayashi K., Dai Y., Mizushima T., Katsura H., Fukuoka T., Tokunaga A. and Noguchi K. (2004) Role of mitogen-activated protein kinase activation in injured and intact primary afferent neurons for mechanical and heat hypersensitivity after spinal nerve ligation. J. Neurosci. 24, Park K. A. and Vasko M. R. (2005) Lipid mediators of sensitivity in sensory neurons. Trends Pharmacol. Sci. 26,
9 1416 M. Tsuda et al. Rae M. G., Rowan E. G. and Kennedy C. (1998) Pharmacological properties of P2X 3 -receptors present in neurones of the rat dorsal root ganglia. Br. J. Pharmacol. 124, Ralevic V. and Burnstock G. (1998) Receptors for purines and pyrimidines. Pharmacol. Rev. 50, Ramer M. S., Bradbury E. J. and McMahon S. B. (2001) Nerve growth factor induces P2X 3 expression in sensory neurons. J. Neurochem. 77, Schievella A. R., Regier M. K., Smith W. L. and Lin L. L. (1995) Calcium-mediated translocation of cytosolic phospholipase A 2 to the nuclear envelope and endoplasmic reticulum. J. Biol. Chem. 270, Seno K., Okuno T., Nishi K. et al. (2000) Pyrrolidine inhibitors of human cytosolic phospholipase A 2. J. Med. Chem. 43, Sharp C. J., Reeve A. J., Collins S. D., Martindale J. C., Summerfield S. G., Sargent B. S., Bate S. T. and Chessell I. P. (2006) Investigation into the role of P2X 3 /P2X 2/3 receptors in neuropathic pain following chronic constriction injury in the rat: an electrophysiological study. Br. J. Pharmacol. 148, Shimizu T., Ohto T. and Kita Y. (2006) Cytosolic phospholipase A 2 : biochemical properties and physiological roles. IUBMB Life 58, Shmelzer Z., Haddad N., Admon E., Pessach I., Leto T. L., Eitan-Hazan Z., Hershfinkel M. and Levy R. (2003) Unique targeting of cytosolic phospholipase A 2 to plasma membranes mediated by the NADPH oxidase in phagocytes. J. Cell Biol. 162, Souslova V., Cesare P., Ding Y. et al. (2000) Warm-coding deficits and aberrant inflammatory pain in mice lacking P2X 3 receptors. Nature 407, Spencer A. G., Woods J. W., Arakawa T., Singer I. I. and Smith W. L. (1998) Subcellular localization of prostaglandin endoperoxide H synthases-1 and -2 by immunoelectron microscopy. J. Biol. Chem. 273, Stahelin R. V., Rafter J. D., Das S. and Cho W. (2003) The molecular basis of differential subcellular localization of C2 domains of protein kinase C-alpha and group IVa cytosolic phospholipase A 2. J. Biol. Chem. 278, Su H., McClarty G., Dong F., Hatch G. M., Pan Z. K. and Zhong G. (2004) Activation of Raf/MEK/ERK/cPLA 2 signaling pathway is essential for chlamydial acquisition of host glycerophospholipids. J. Biol. Chem. 279, Tsuda M., Koizumi S., Kita A., Shigemoto Y., Ueno S. and Inoue K. (2000) Mechanical allodynia caused by intraplantar injection of P2X receptor agonist in rats: involvement of heteromeric P2X 2/3 receptor signaling in capsaicin-insensitive primary afferent neurons. J. Neurosci. 20, RC90. Tsuda M., Shigemoto-Mogami Y., Koizumi S., Mizokoshi A., Kohsaka S., Salter M. W. and Inoue K. (2003) P2X 4 receptors induced in spinal microglia gate tactile allodynia after nerve injury. Nature 424, Ueno S., Koizumi S. and Inoue K. (1998) Characterization of Ca 2+ influx through recombinant P2X receptor in C6BU-1 cells. Br. J. Pharmacol. 124, Ueno S., Tsuda M., Iwanaga T. and Inoue K. (1999) Cell type-specific ATP-activated responses in rat dorsal root ganglion neurons. Br. J. Pharmacol. 126, Vulchanova L., Riedl M. S., Shuster S. J., Stone L. S., Hargreaves K. M., Buell G., Surprenant A., North R. A. and Elde R. (1998) P2X 3 is expressed by DRG neurons that terminate in inner lamina II. Eur. J. Neurosci. 10, Wu G., Whiteside G. T., Lee G., Nolan S., Niosi M., Pearson M. S. and Ilyin V. I. (2004) A , a selective P2X 3 /P2X 2/3 receptor antagonist, reverses inflammatory mechanical hyperalgesia through action at peripheral receptors in rats. Eur. J. Pharmacol. 504, Yaksh T. L., Jessell T. M., Gamse R., Mudge A. W. and Leeman S. E. (1980) Intrathecal morphine inhibits substance P release from mammalian spinal cord in vivo. Nature 286, Yoon Y. W., Na H. S. and Chung J. M. (1996) Contributions of injured and intact afferents to neuropathic pain in an experimental rat model. Pain 64, Zhou J., Chung K. and Chung J. M. (2001) Development of purinergic sensitivity in sensory neurons after peripheral nerve injury in the rat. Brain Res. 915,
A role for uninjured afferents in neuropathic pain
 Acta Physiologica Sinica, October 25, 2008, 60 (5): 605-609 http://www.actaps.com.cn 605 Review A role for uninjured afferents in neuropathic pain Richard A. Meyer 1,2,3,*, Matthias Ringkamp 1 Departments
Acta Physiologica Sinica, October 25, 2008, 60 (5): 605-609 http://www.actaps.com.cn 605 Review A role for uninjured afferents in neuropathic pain Richard A. Meyer 1,2,3,*, Matthias Ringkamp 1 Departments
Introduction to some interesting research questions: Molecular biology of the primary afferent nociceptor
 Introduction to some interesting research questions: Molecular biology of the primary afferent nociceptor NOCICEPTORS ARE NOT IDENTICAL PEPTIDE SubP/CGRP Trk A NON-PEPTIDE IB4 P2X 3 c-ret Snider and McMahon
Introduction to some interesting research questions: Molecular biology of the primary afferent nociceptor NOCICEPTORS ARE NOT IDENTICAL PEPTIDE SubP/CGRP Trk A NON-PEPTIDE IB4 P2X 3 c-ret Snider and McMahon
NIH Public Access Author Manuscript J Neuropathic Pain Symptom Palliation. Author manuscript; available in PMC 2007 March 26.
 NIH Public Access Author Manuscript Published in final edited form as: J Neuropathic Pain Symptom Palliation. 2005 ; 1(1): 19 23. Sympathetic Fiber Sprouting in Chronically Compressed Dorsal Root Ganglia
NIH Public Access Author Manuscript Published in final edited form as: J Neuropathic Pain Symptom Palliation. 2005 ; 1(1): 19 23. Sympathetic Fiber Sprouting in Chronically Compressed Dorsal Root Ganglia
ATP receptors in pain sensation: Involvement of spinal microglia and P2X 4 receptors
 Purinergic Signalling (2005) 1: 95 100 # Springer 2005 Review ATP receptors in pain sensation: Involvement of spinal microglia and P2X 4 receptors Kazuhide Inoue 1,2, Makoto Tsuda 1 & Schuichi Koizumi
Purinergic Signalling (2005) 1: 95 100 # Springer 2005 Review ATP receptors in pain sensation: Involvement of spinal microglia and P2X 4 receptors Kazuhide Inoue 1,2, Makoto Tsuda 1 & Schuichi Koizumi
Potential for delta opioid receptor agonists as analgesics in chronic pain therapy
 Potential for delta opioid receptor agonists as analgesics in chronic pain therapy David Kendall & Bengt von Mentzer; PharmNovo AB/UK Alex Conibear & Eamonn Kelly, University of Bristol Junaid Asghar,
Potential for delta opioid receptor agonists as analgesics in chronic pain therapy David Kendall & Bengt von Mentzer; PharmNovo AB/UK Alex Conibear & Eamonn Kelly, University of Bristol Junaid Asghar,
P2X 3 receptors and peripheral pain mechanisms
 J Physiol 554.2 pp 301 308 301 SYMPOSIUM REPORT P2X 3 receptors and peripheral pain mechanisms R. Alan North Institute of Molecular Physiology, University of Sheffield, Western Bank, Sheffield S10 2TN,
J Physiol 554.2 pp 301 308 301 SYMPOSIUM REPORT P2X 3 receptors and peripheral pain mechanisms R. Alan North Institute of Molecular Physiology, University of Sheffield, Western Bank, Sheffield S10 2TN,
Special Issue on Pain and Itch
 Special Issue on Pain and Itch Title: Recent Progress in Understanding the Mechanisms of Pain and Itch Guest Editor of the Special Issue: Ru-Rong Ji, PhD Chronic pain is a major health problem world-wide.
Special Issue on Pain and Itch Title: Recent Progress in Understanding the Mechanisms of Pain and Itch Guest Editor of the Special Issue: Ru-Rong Ji, PhD Chronic pain is a major health problem world-wide.
A subpopulation of nociceptors specifically linked to itch
 Supplementary Information A subpopulation of nociceptors specifically linked to itch Liang Han 1, Chao Ma 3,4, Qin Liu 1,2, Hao-Jui Weng 1,2, Yiyuan Cui 5, Zongxiang Tang 1,2, Yushin Kim 1, Hong Nie 4,
Supplementary Information A subpopulation of nociceptors specifically linked to itch Liang Han 1, Chao Ma 3,4, Qin Liu 1,2, Hao-Jui Weng 1,2, Yiyuan Cui 5, Zongxiang Tang 1,2, Yushin Kim 1, Hong Nie 4,
Pathophysiology of Pain
 Pathophysiology of Pain Wound Inflammatory response Chemical mediators Activity in Pain Path PAIN http://neuroscience.uth.tmc.edu/s2/chapter08.html Chris Cohan, Ph.D. Dept. of Pathology/Anat Sci University
Pathophysiology of Pain Wound Inflammatory response Chemical mediators Activity in Pain Path PAIN http://neuroscience.uth.tmc.edu/s2/chapter08.html Chris Cohan, Ph.D. Dept. of Pathology/Anat Sci University
Enhanced formalin nociceptive responses following L5 nerve ligation in the rat reveals neuropathy-induced inflammatory hyperalgesia
 University of Kentucky From the SelectedWorks of Renee R. Donahue 2001 Enhanced formalin nociceptive responses following L5 nerve ligation in the rat reveals neuropathy-induced inflammatory hyperalgesia
University of Kentucky From the SelectedWorks of Renee R. Donahue 2001 Enhanced formalin nociceptive responses following L5 nerve ligation in the rat reveals neuropathy-induced inflammatory hyperalgesia
Repeated intra-articular injections of acidic saline produce long-lasting joint pain and. widespread hyperalgesia
 Repeated intra-articular injections of acidic saline produce long-lasting joint pain and widespread hyperalgesia N. Sugimura 1, M. Ikeuchi 1 *, M. Izumi 1, T. Kawano 2, K. Aso 1, T. Kato 1, T. Ushida 3,
Repeated intra-articular injections of acidic saline produce long-lasting joint pain and widespread hyperalgesia N. Sugimura 1, M. Ikeuchi 1 *, M. Izumi 1, T. Kawano 2, K. Aso 1, T. Kato 1, T. Ushida 3,
Risk Factors That Predispose Patients to Orofacial Pain
 Risk Factors That Predispose Patients to Orofacial Pain Paul Durham, PhD Distinguished Professor of Cell Biology Director Center for Biomedical & Life Sciences Missouri State University Cluster Headache
Risk Factors That Predispose Patients to Orofacial Pain Paul Durham, PhD Distinguished Professor of Cell Biology Director Center for Biomedical & Life Sciences Missouri State University Cluster Headache
Involvement of phosphatase and tensin homolog deleted from chromosome 10 in rodent model of neuropathic pain
 Huang et al. Journal of Neuroinflammation (2015) 12:59 DOI 10.1186/s12974-015-0280-1 JOURNAL OF NEUROINFLAMMATION RESEARCH Open Access Involvement of phosphatase and tensin homolog deleted from chromosome
Huang et al. Journal of Neuroinflammation (2015) 12:59 DOI 10.1186/s12974-015-0280-1 JOURNAL OF NEUROINFLAMMATION RESEARCH Open Access Involvement of phosphatase and tensin homolog deleted from chromosome
Diabetic Complications Consortium
 Diabetic Complications Consortium Application Title: Cathepsin S inhibition and diabetic neuropathy Principal Investigator: Nigel A Calcutt 1. Project Accomplishments: We investigated the efficacy of cathepsin
Diabetic Complications Consortium Application Title: Cathepsin S inhibition and diabetic neuropathy Principal Investigator: Nigel A Calcutt 1. Project Accomplishments: We investigated the efficacy of cathepsin
The Egyptian Journal of Hospital Medicine (January 2018) Vol. 70 (12), Page
 The Egyptian Journal of Hospital Medicine (January 2018) Vol. 70 (12), Page 2172-2177 Blockage of HCN Channels with ZD7288 Attenuates Mechanical Hypersensitivity in Rats Model of Diabetic Neuropathy Hussain
The Egyptian Journal of Hospital Medicine (January 2018) Vol. 70 (12), Page 2172-2177 Blockage of HCN Channels with ZD7288 Attenuates Mechanical Hypersensitivity in Rats Model of Diabetic Neuropathy Hussain
P2X3 receptors are present on sensory afferent neurons. Nociceptive Responses in the Uninjured State
 The Journal of Pain, Vol 8, No 7 (July), 2007: pp 556-562 Available online at www.sciencedirect.com 1 -Adrenergic Receptors Augment P2X 3 Receptor Mediated Nociceptive Responses in the Uninjured State
The Journal of Pain, Vol 8, No 7 (July), 2007: pp 556-562 Available online at www.sciencedirect.com 1 -Adrenergic Receptors Augment P2X 3 Receptor Mediated Nociceptive Responses in the Uninjured State
Charlie Taylor, PhD CpTaylor Consulting Chelsea, MI, USA
 Contribution of Calcium Channel α 2 δ Binding Sites to the Pharmacology of Gabapentin and Pregabalin Charlie Taylor, PhD CpTaylor Consulting Chelsea, MI, USA Disclosure Information Charlie Taylor, PhD
Contribution of Calcium Channel α 2 δ Binding Sites to the Pharmacology of Gabapentin and Pregabalin Charlie Taylor, PhD CpTaylor Consulting Chelsea, MI, USA Disclosure Information Charlie Taylor, PhD
Rapamycin Suppresses Astrocytic and Microglial Activation and Reduced Development of Neuropathic Pain after Spinal Cord Injury in Mice.
 Rapamycin Suppresses Astrocytic and Microglial Activation and Reduced Development of Neuropathic Pain after Spinal Cord Injury in Mice. Satoshi Tateda, M.D., Haruo Kanno, M.D., Ph.D., Hiroshi Ozawa, M.D.,
Rapamycin Suppresses Astrocytic and Microglial Activation and Reduced Development of Neuropathic Pain after Spinal Cord Injury in Mice. Satoshi Tateda, M.D., Haruo Kanno, M.D., Ph.D., Hiroshi Ozawa, M.D.,
MOLECULAR AND CELLULAR NEUROSCIENCE
 MOLECULAR AND CELLULAR NEUROSCIENCE BMP-218 November 4, 2014 DIVISIONS OF THE NERVOUS SYSTEM The nervous system is composed of two primary divisions: 1. CNS - Central Nervous System (Brain + Spinal Cord)
MOLECULAR AND CELLULAR NEUROSCIENCE BMP-218 November 4, 2014 DIVISIONS OF THE NERVOUS SYSTEM The nervous system is composed of two primary divisions: 1. CNS - Central Nervous System (Brain + Spinal Cord)
Opioid Receptor Trafficking in Pain States
 Encyclopedia of Pain Springer-Verlag Berlin Heidelberg 2007 10.1007/978-3-540-29805-2_2962 Robert F. Schmidt and William D. Willis Opioid Receptor Trafficking in Pain States Catherine M. Cahill 3, Anne
Encyclopedia of Pain Springer-Verlag Berlin Heidelberg 2007 10.1007/978-3-540-29805-2_2962 Robert F. Schmidt and William D. Willis Opioid Receptor Trafficking in Pain States Catherine M. Cahill 3, Anne
211MDS Pain theories
 211MDS Pain theories Definition In 1986, the International Association for the Study of Pain (IASP) defined pain as a sensory and emotional experience associated with real or potential injuries, or described
211MDS Pain theories Definition In 1986, the International Association for the Study of Pain (IASP) defined pain as a sensory and emotional experience associated with real or potential injuries, or described
Chapter 7 Nerve tissue 1 Liu Jiamei
 Chapter 7 Nerve tissue 1 Liu Jiamei General description: nerve tissue nerve cells (neurons): show numerous long processes receive the stimulation make contact with each other, conduct the nerve impulse
Chapter 7 Nerve tissue 1 Liu Jiamei General description: nerve tissue nerve cells (neurons): show numerous long processes receive the stimulation make contact with each other, conduct the nerve impulse
Involvement of Locus Coeruleus Noradrenergic Neurons in Supraspinal Antinociception by, -Methylene-ATP in Rats
 J Pharmacol Sci 94, 153 160 (2004) Journal of Pharmacological Sciences 2004 The Japanese Pharmacological Society Full Paper Involvement of Locus Coeruleus Noradrenergic Neurons in Supraspinal Antinociception
J Pharmacol Sci 94, 153 160 (2004) Journal of Pharmacological Sciences 2004 The Japanese Pharmacological Society Full Paper Involvement of Locus Coeruleus Noradrenergic Neurons in Supraspinal Antinociception
Abbott s TRPV1 Antagonist Program
 Abbott s TRPV1 Antagonist Program Connie R. Faltynek, Ph.D. Sr. Director, Neuroscience and Pain Discovery American Pain Society Annual Meeting Basic Science Dinner Symposium May 9, 2008 Early History of
Abbott s TRPV1 Antagonist Program Connie R. Faltynek, Ph.D. Sr. Director, Neuroscience and Pain Discovery American Pain Society Annual Meeting Basic Science Dinner Symposium May 9, 2008 Early History of
What Cell Make Up the Brain and Spinal Cord
 What Cell Make Up the Brain and Spinal Cord Jennifer LaVail, Ph.D. (http://anatomy.ucsf.edu/pages/lavaillab/index.html) What kinds of cells are these?" Neuron?" Epithelial cell?" Glial cell?" What makes
What Cell Make Up the Brain and Spinal Cord Jennifer LaVail, Ph.D. (http://anatomy.ucsf.edu/pages/lavaillab/index.html) What kinds of cells are these?" Neuron?" Epithelial cell?" Glial cell?" What makes
Functional Downregulation of P2X 3 Receptor Subunit in Rat Sensory Neurons Reveals a Significant Role in Chronic Neuropathic and Inflammatory Pain
 The Journal of Neuroscience, September 15, 2002, 22(18):8139 8147 Functional Downregulation of P2X 3 Receptor Subunit in Rat Sensory Neurons Reveals a Significant Role in Chronic Neuropathic and Inflammatory
The Journal of Neuroscience, September 15, 2002, 22(18):8139 8147 Functional Downregulation of P2X 3 Receptor Subunit in Rat Sensory Neurons Reveals a Significant Role in Chronic Neuropathic and Inflammatory
Seizure: the clinical manifestation of an abnormal and excessive excitation and synchronization of a population of cortical
 Are There Sharing Mechanisms of Epilepsy, Migraine and Neuropathic Pain? Chin-Wei Huang, MD, PhD Department of Neurology, NCKUH Basic mechanisms underlying seizures and epilepsy Seizure: the clinical manifestation
Are There Sharing Mechanisms of Epilepsy, Migraine and Neuropathic Pain? Chin-Wei Huang, MD, PhD Department of Neurology, NCKUH Basic mechanisms underlying seizures and epilepsy Seizure: the clinical manifestation
Original Article Upregulation of neuregulin-1 reverses signs of neuropathic pain in rats
 Int J Clin Exp Pathol 2014;7(9):5916-5921 www.ijcep.com /ISSN:1936-2625/IJCEP0001263 Original Article Upregulation of neuregulin-1 reverses signs of neuropathic pain in rats Guojun Wang 1,2*, Dawei Dai
Int J Clin Exp Pathol 2014;7(9):5916-5921 www.ijcep.com /ISSN:1936-2625/IJCEP0001263 Original Article Upregulation of neuregulin-1 reverses signs of neuropathic pain in rats Guojun Wang 1,2*, Dawei Dai
Electrical Stimulation Control Nerve Regeneration via the p38 Mitogen-activated Protein Kinase and CREB
 Electrical Stimulation Control Nerve Regeneration via the p38 Mitogen-activated Protein Kinase and CREB Kenji Kawamura, Yoshio Kano. Kibi International University, Takahashi-city, Japan. Disclosures: K.
Electrical Stimulation Control Nerve Regeneration via the p38 Mitogen-activated Protein Kinase and CREB Kenji Kawamura, Yoshio Kano. Kibi International University, Takahashi-city, Japan. Disclosures: K.
Sigma-1 Receptor Antagonist BD1047 Reduces Allodynia and Spinal ERK Phosphorylation Following Chronic Compression of Dorsal Root Ganglion in Rats
 Korean J Physiol Pharmacol Vol 14: 359-364, December, 2010 DOI: 10.4196/kjpp.2010.14.6.359 Sigma-1 Receptor Antagonist BD1047 Reduces Allodynia and Spinal ERK Phosphorylation Following Chronic Compression
Korean J Physiol Pharmacol Vol 14: 359-364, December, 2010 DOI: 10.4196/kjpp.2010.14.6.359 Sigma-1 Receptor Antagonist BD1047 Reduces Allodynia and Spinal ERK Phosphorylation Following Chronic Compression
Pharmacology of Pain Transmission and Modulation
 Pharmacology of Pain Transmission and Modulation 2 Jürg Schliessbach and Konrad Maurer Nociceptive Nerve Fibers Pain is transmitted to the central nervous system via thinly myelinated Aδ and unmyelinated
Pharmacology of Pain Transmission and Modulation 2 Jürg Schliessbach and Konrad Maurer Nociceptive Nerve Fibers Pain is transmitted to the central nervous system via thinly myelinated Aδ and unmyelinated
Major Structures of the Nervous System. Brain, cranial nerves, spinal cord, spinal nerves, ganglia, enteric plexuses and sensory receptors
 Major Structures of the Nervous System Brain, cranial nerves, spinal cord, spinal nerves, ganglia, enteric plexuses and sensory receptors Nervous System Divisions Central Nervous System (CNS) consists
Major Structures of the Nervous System Brain, cranial nerves, spinal cord, spinal nerves, ganglia, enteric plexuses and sensory receptors Nervous System Divisions Central Nervous System (CNS) consists
NMDA-Receptor Antagonists and Opioid Receptor Interactions as Related to Analgesia and Tolerance
 Vol. 19 No. 1(Suppl.) January 2000 Journal of Pain and Symptom Management S7 Proceedings Supplement NDMA-Receptor Antagonists: Evolving Role in Analgesia NMDA-Receptor Antagonists and Opioid Receptor Interactions
Vol. 19 No. 1(Suppl.) January 2000 Journal of Pain and Symptom Management S7 Proceedings Supplement NDMA-Receptor Antagonists: Evolving Role in Analgesia NMDA-Receptor Antagonists and Opioid Receptor Interactions
Chapter 11 Introduction to the Nervous System and Nervous Tissue Chapter Outline
 Chapter 11 Introduction to the Nervous System and Nervous Tissue Chapter Outline Module 11.1 Overview of the Nervous System (Figures 11.1-11.3) A. The nervous system controls our perception and experience
Chapter 11 Introduction to the Nervous System and Nervous Tissue Chapter Outline Module 11.1 Overview of the Nervous System (Figures 11.1-11.3) A. The nervous system controls our perception and experience
Neurotransmitter Systems II Receptors. Reading: BCP Chapter 6
 Neurotransmitter Systems II Receptors Reading: BCP Chapter 6 Neurotransmitter Systems Normal function of the human brain requires an orderly set of chemical reactions. Some of the most important chemical
Neurotransmitter Systems II Receptors Reading: BCP Chapter 6 Neurotransmitter Systems Normal function of the human brain requires an orderly set of chemical reactions. Some of the most important chemical
Analgesia Mediated by the TRPM8 Cold Receptor in Chronic Neuropathic Pain
 Current Biology 16, 1591 1605, August 22, 2006 ª2006 Elsevier Ltd All rights reserved DOI 10.1016/j.cub.2006.07.061 Analgesia Mediated by the TRPM8 Cold Receptor in Chronic Neuropathic Pain Article Clare
Current Biology 16, 1591 1605, August 22, 2006 ª2006 Elsevier Ltd All rights reserved DOI 10.1016/j.cub.2006.07.061 Analgesia Mediated by the TRPM8 Cold Receptor in Chronic Neuropathic Pain Article Clare
Introduction ORIGINAL ARTICLE. Seiji Ohtori Kazuhisa Takahashi Hideshige Moriya
 Eur Spine J (2003) 12 :211 215 DOI 10.1007/s00586-002-0506-7 ORIGINAL ARTICLE Seiji Ohtori Kazuhisa Takahashi Hideshige Moriya Calcitonin gene-related peptide immunoreactive DRG neurons innervating the
Eur Spine J (2003) 12 :211 215 DOI 10.1007/s00586-002-0506-7 ORIGINAL ARTICLE Seiji Ohtori Kazuhisa Takahashi Hideshige Moriya Calcitonin gene-related peptide immunoreactive DRG neurons innervating the
Spinal Cord Injury Pain. Michael Massey, DO CentraCare Health St Cloud, MN 11/07/2018
 Spinal Cord Injury Pain Michael Massey, DO CentraCare Health St Cloud, MN 11/07/2018 Objectives At the conclusion of this session, participants should be able to: 1. Understand the difference between nociceptive
Spinal Cord Injury Pain Michael Massey, DO CentraCare Health St Cloud, MN 11/07/2018 Objectives At the conclusion of this session, participants should be able to: 1. Understand the difference between nociceptive
The Nervous System. Dr. ZHANG Xiong Dept. of Physiology ZJU School of Medicine.
 The Nervous System Dr. ZHANG Xiong Dept. of Physiology ZJU School of Medicine Http://10.10.10.151 Part 1. Summary of the nervous system The Nervous System Central Nervous System Brain + Spinal Cord Peripheral
The Nervous System Dr. ZHANG Xiong Dept. of Physiology ZJU School of Medicine Http://10.10.10.151 Part 1. Summary of the nervous system The Nervous System Central Nervous System Brain + Spinal Cord Peripheral
Byeol-Rim Kang and Chang-Beohm Ahn Department of Acupuncture and Moxibustion, College of Oriental Medicine Dong-Eui University, Busan , Korea
 The American Journal of Chinese Medicine, Vol. 35, No. 6, 987 993 2007 World Scientific Publishing Company Institute for Advanced Research in Asian Science and Medicine N-Methyl-D-Aspartate Antagonist
The American Journal of Chinese Medicine, Vol. 35, No. 6, 987 993 2007 World Scientific Publishing Company Institute for Advanced Research in Asian Science and Medicine N-Methyl-D-Aspartate Antagonist
BLADDER AND CUTANEOUS SENSORY NEURONS OF THE RAT EXPRESS DIFFERENT FUNCTIONAL P2X RECEPTORS
 Neuroscience 120 (2003) 667 675 BLADDER AND CUTANEOUS SENSORY NEURONS OF THE RAT EXPRESS DIFFERENT FUNCTIONAL P2X RECEPTORS Y. ZHONG, a * 1 A. S. BANNING, a D. A. COCKAYNE, b A. P. D. W. FORD, b G. BURNSTOCK
Neuroscience 120 (2003) 667 675 BLADDER AND CUTANEOUS SENSORY NEURONS OF THE RAT EXPRESS DIFFERENT FUNCTIONAL P2X RECEPTORS Y. ZHONG, a * 1 A. S. BANNING, a D. A. COCKAYNE, b A. P. D. W. FORD, b G. BURNSTOCK
浙江大学医学院基础医学整合课程 各论 III. The Nervous System. Dr. ZHANG Xiong Dept. of Physiology ZJU School of Medicine
 The Nervous System Dr. ZHANG Xiong Dept. of Physiology ZJU School of Medicine xiongzhang@zju.edu.cn http://10.202.77.12/ 1 Part 1. Summary of the nervous system 2 The Nervous System Central Nervous System
The Nervous System Dr. ZHANG Xiong Dept. of Physiology ZJU School of Medicine xiongzhang@zju.edu.cn http://10.202.77.12/ 1 Part 1. Summary of the nervous system 2 The Nervous System Central Nervous System
Department of Orthopaedic Surgery, Tohoku University Graduate School of Medicine, Sendai, Japan, 2
 Low-energy Extracorporeal Shock Wave Therapy Promotes VEGF Expression and Angiogenesis and Improve Locomotor and Sensory Functions after spinal cord injury Kenichiro Yahata 1, Hiroshi Ozawa, M.D., Ph.D.
Low-energy Extracorporeal Shock Wave Therapy Promotes VEGF Expression and Angiogenesis and Improve Locomotor and Sensory Functions after spinal cord injury Kenichiro Yahata 1, Hiroshi Ozawa, M.D., Ph.D.
Changes in P2X receptor responses of sensory neurons from P2X 3 -de cient mice
 European Journal of Neuroscience, Vol. 14, pp. 1784±1792, 2001 ã Federation of European Neuroscience Societies Changes in P2X receptor responses of sensory neurons from P2X 3 -de cient mice Yu Zhong, 1
European Journal of Neuroscience, Vol. 14, pp. 1784±1792, 2001 ã Federation of European Neuroscience Societies Changes in P2X receptor responses of sensory neurons from P2X 3 -de cient mice Yu Zhong, 1
Cannabinoid Agonists Inhibit Neuropathic Pain Induced by Brachial Plexus Avulsion in Mice by Affecting Glial Cells and MAP Kinases
 Cannabinoid Agonists Inhibit Neuropathic Pain Induced by Brachial Plexus Avulsion in Mice by Affecting Glial Cells and MAP Kinases Ana F. Paszcuk 1, Rafael C. Dutra 1, Kathryn A. B. S. da Silva 1, Nara
Cannabinoid Agonists Inhibit Neuropathic Pain Induced by Brachial Plexus Avulsion in Mice by Affecting Glial Cells and MAP Kinases Ana F. Paszcuk 1, Rafael C. Dutra 1, Kathryn A. B. S. da Silva 1, Nara
Preclinical evaluation of pain in endometriosis
 receptor antagonism reduces peripheral and central hyperalgesia in a preclinical mouse model of endometriosis Erin Greaves, Andrew W Horne, Helen Jerina, arta ikolajczak, Lisa Hilferty, Rory itchell, Sue
receptor antagonism reduces peripheral and central hyperalgesia in a preclinical mouse model of endometriosis Erin Greaves, Andrew W Horne, Helen Jerina, arta ikolajczak, Lisa Hilferty, Rory itchell, Sue
April 29, Neurophysiology. Chul-Kyu Park, Ph.D. Assistant Professor Department of Physiology, Graduate School of Medicine, Gachon University,
 April 29, 2016 Neurophysiology Chul-Kyu Park, Ph.D. Assistant Professor Department of Physiology, Graduate School of Medicine, Gachon University, Cells in the brain Neurons glia 1. Astrocytes 2. Microglia
April 29, 2016 Neurophysiology Chul-Kyu Park, Ph.D. Assistant Professor Department of Physiology, Graduate School of Medicine, Gachon University, Cells in the brain Neurons glia 1. Astrocytes 2. Microglia
Rapamycin suppresses astrocytic and microglial activation and reduces development of neuropathic pain after spinal cord injury in mice.
 Rapamycin suppresses astrocytic and microglial activation and reduces development of neuropathic pain after spinal cord injury in mice. Satoshi Tateda, MD, Haruo Kanno, MD, PhD, Hiroshi Ozawa, MD, PhD,
Rapamycin suppresses astrocytic and microglial activation and reduces development of neuropathic pain after spinal cord injury in mice. Satoshi Tateda, MD, Haruo Kanno, MD, PhD, Hiroshi Ozawa, MD, PhD,
Receptor-interacting Protein Kinases Mediate Necroptosis In Neural Tissue Damage After Spinal Cord Injury
 Receptor-interacting Protein Kinases Mediate Necroptosis In Neural Tissue Damage After Spinal Cord Injury Haruo Kanno, M.D., Ph.D., Hiroshi Ozawa, M.D., Ph.D., Satoshi Tateda, M.D., Kenichiro Yahata, M.D.,
Receptor-interacting Protein Kinases Mediate Necroptosis In Neural Tissue Damage After Spinal Cord Injury Haruo Kanno, M.D., Ph.D., Hiroshi Ozawa, M.D., Ph.D., Satoshi Tateda, M.D., Kenichiro Yahata, M.D.,
EDUCATION M.D., Peking Union Medical College, Beijing, China, 1999 B.S., Beijing University, College of Life Science, Beijing, China, 1994
 CHAO MA, M.D. Yale University School of Medicine, Department of Anesthesiology, 333 Cedar Street, TMP3, New Haven, CT 06510, USA. Phone: 203-785-3522 (O), 203-606-7959 (C), Fax: 203-737-1528, Email: chao.ma@yale.edu
CHAO MA, M.D. Yale University School of Medicine, Department of Anesthesiology, 333 Cedar Street, TMP3, New Haven, CT 06510, USA. Phone: 203-785-3522 (O), 203-606-7959 (C), Fax: 203-737-1528, Email: chao.ma@yale.edu
Lidocaine inhibits neurite growth in mouse dorsal root ganglion cells in culture
 Lidocaine inhibits neurite growth in mouse dorsal root ganglion cells in culture 3 Hiromi ~iruma', Hiroshi ~ aru~ama~, Zyun'ici B. Simada, Takashi Katakural, Sumio ~ oka~, Toshifumi ~akenaka~ and Tadashi
Lidocaine inhibits neurite growth in mouse dorsal root ganglion cells in culture 3 Hiromi ~iruma', Hiroshi ~ aru~ama~, Zyun'ici B. Simada, Takashi Katakural, Sumio ~ oka~, Toshifumi ~akenaka~ and Tadashi
TMC9 as a novel mechanosensitive ion channel
 TMC9 as a novel mechanosensitive ion channel Mechanical forces play numerous roles in physiology. When an object contacts our skin, it exerts a force that is encoded as touch or pain depending on its intensity.
TMC9 as a novel mechanosensitive ion channel Mechanical forces play numerous roles in physiology. When an object contacts our skin, it exerts a force that is encoded as touch or pain depending on its intensity.
Victor Chaban, Ph.D., MSCR
 Hormonal Modulation of Pain : Estrogen Modulation of Visceral Nociception Victor Chaban, Ph.D., MSCR Department of Internal Medicine Charles R. Drew University of Medicine and Science Department of Medicine
Hormonal Modulation of Pain : Estrogen Modulation of Visceral Nociception Victor Chaban, Ph.D., MSCR Department of Internal Medicine Charles R. Drew University of Medicine and Science Department of Medicine
Published online October 10, 2016
 Published online October 10, 2016 reviewed by Jim C. Eisenach, Wake Forest University; Ronald Lindsay, Zebra Biologics; Remi Quirion, McGill University; and Tony L. Yaksh, University of California, San
Published online October 10, 2016 reviewed by Jim C. Eisenach, Wake Forest University; Ronald Lindsay, Zebra Biologics; Remi Quirion, McGill University; and Tony L. Yaksh, University of California, San
What it Takes to be a Pain
 What it Takes to be a Pain Pain Pathways and the Neurophysiology of pain Dennis S. Pacl, MD, FACP, FAChPM Austin Palliative Care/ Hospice Austin A Definition of Pain complex constellation of unpleasant
What it Takes to be a Pain Pain Pathways and the Neurophysiology of pain Dennis S. Pacl, MD, FACP, FAChPM Austin Palliative Care/ Hospice Austin A Definition of Pain complex constellation of unpleasant
Somatic Sensation (MCB160 Lecture by Mu-ming Poo, Friday March 9, 2007)
 Somatic Sensation (MCB160 Lecture by Mu-ming Poo, Friday March 9, 2007) Introduction Adrian s work on sensory coding Spinal cord and dorsal root ganglia Four somatic sense modalities Touch Mechanoreceptors
Somatic Sensation (MCB160 Lecture by Mu-ming Poo, Friday March 9, 2007) Introduction Adrian s work on sensory coding Spinal cord and dorsal root ganglia Four somatic sense modalities Touch Mechanoreceptors
Adenosine A1 receptors mediate local anti-nociceptive effects of acupuncture
 Adenosine A1 receptors mediate local anti-nociceptive effects of acupuncture Nanna Goldman *, Michael Chen *, Takumi Fujita *, Qiwu Xu, Weiguo Peng, Wei Liu, Tina K. Jensen, Yong Pei, Fushun Wang, Xiaoning
Adenosine A1 receptors mediate local anti-nociceptive effects of acupuncture Nanna Goldman *, Michael Chen *, Takumi Fujita *, Qiwu Xu, Weiguo Peng, Wei Liu, Tina K. Jensen, Yong Pei, Fushun Wang, Xiaoning
10.1: Introduction. Cell types in neural tissue: Neurons Neuroglial cells (also known as neuroglia, glia, and glial cells) Dendrites.
 10.1: Introduction Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display. Cell types in neural tissue: Neurons Neuroglial cells (also known as neuroglia, glia, and glial
10.1: Introduction Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display. Cell types in neural tissue: Neurons Neuroglial cells (also known as neuroglia, glia, and glial
TRPA1 induced in sensory neurons contributes to cold hyperalgesia after inflammation and nerve injury
 Research article TRPA1 induced in sensory neurons contributes to cold hyperalgesia after inflammation and nerve injury Koichi Obata, 1 Hirokazu Katsura, 1 Toshiyuki Mizushima, 1 Hiroki Yamanaka, 1 Kimiko
Research article TRPA1 induced in sensory neurons contributes to cold hyperalgesia after inflammation and nerve injury Koichi Obata, 1 Hirokazu Katsura, 1 Toshiyuki Mizushima, 1 Hiroki Yamanaka, 1 Kimiko
Received 26 April 2004; revised 21 August 2004; accepted 20 September 2004 Available online 19 November 2004
 Experimental Neurology 191 (2005) 128 136 www.elsevier.com/locate/yexnr Ectopic discharges from injured nerve fibers are highly correlated with tactile allodynia only in early, but not late, stage in rats
Experimental Neurology 191 (2005) 128 136 www.elsevier.com/locate/yexnr Ectopic discharges from injured nerve fibers are highly correlated with tactile allodynia only in early, but not late, stage in rats
Pain Mechanisms. Prof Michael G Irwin MD, FRCA, FANZCA FHKAM Head Department of Anaesthesiology University of Hong Kong. The Somatosensory System
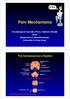 ain Mechanisms rof Michael G Irwin MD, FRCA, FANZCA FHKAM Head Department of Anaesthesiology University of Hong Kong The Somatosensory System Frontal cortex Descending pathway eriaqueductal gray matter
ain Mechanisms rof Michael G Irwin MD, FRCA, FANZCA FHKAM Head Department of Anaesthesiology University of Hong Kong The Somatosensory System Frontal cortex Descending pathway eriaqueductal gray matter
SUPPLEMENTARY INFORMATION
 doi: 10.1038/nature06994 A phosphatase cascade by which rewarding stimuli control nucleosomal response A. Stipanovich*, E. Valjent*, M. Matamales*, A. Nishi, J.H. Ahn, M. Maroteaux, J. Bertran-Gonzalez,
doi: 10.1038/nature06994 A phosphatase cascade by which rewarding stimuli control nucleosomal response A. Stipanovich*, E. Valjent*, M. Matamales*, A. Nishi, J.H. Ahn, M. Maroteaux, J. Bertran-Gonzalez,
Supplementary Information
 Supplementary Information Supplement References 1 Caterina MJ, Leffler A, Malmberg AB et al. Impaired nociception and pain sensation in mice lacking the capsaicin receptor. Science 2000; 288:306-313. 2
Supplementary Information Supplement References 1 Caterina MJ, Leffler A, Malmberg AB et al. Impaired nociception and pain sensation in mice lacking the capsaicin receptor. Science 2000; 288:306-313. 2
Participations of Glias and Immune Cells in Neuropathic Pain
 Article ID: WMC003728 ISSN 2046-1690 Participations of Glias and Immune Cells in Neuropathic Pain Corresponding Author: Dr. Akio Hiura, Associate Professor, Oral Histology, School of Dentistry, University
Article ID: WMC003728 ISSN 2046-1690 Participations of Glias and Immune Cells in Neuropathic Pain Corresponding Author: Dr. Akio Hiura, Associate Professor, Oral Histology, School of Dentistry, University
TFEB-mediated increase in peripheral lysosomes regulates. Store Operated Calcium Entry
 TFEB-mediated increase in peripheral lysosomes regulates Store Operated Calcium Entry Luigi Sbano, Massimo Bonora, Saverio Marchi, Federica Baldassari, Diego L. Medina, Andrea Ballabio, Carlotta Giorgi
TFEB-mediated increase in peripheral lysosomes regulates Store Operated Calcium Entry Luigi Sbano, Massimo Bonora, Saverio Marchi, Federica Baldassari, Diego L. Medina, Andrea Ballabio, Carlotta Giorgi
SUPPLEMENTARY INFORMATION
 Supplementary Figures Supplementary Figure S1. Binding of full-length OGT and deletion mutants to PIP strips (Echelon Biosciences). Supplementary Figure S2. Binding of the OGT (919-1036) fragments with
Supplementary Figures Supplementary Figure S1. Binding of full-length OGT and deletion mutants to PIP strips (Echelon Biosciences). Supplementary Figure S2. Binding of the OGT (919-1036) fragments with
Product Datasheet. Vanilloid R1/TRPV1 Antibody NB Unit Size: 0.05 ml. Aliquot and store at -20C or -80C. Avoid freeze-thaw cycles.
 Product Datasheet Vanilloid R1/TRPV1 Antibody NB100-1617 Unit Size: 0.05 ml Aliquot and store at -20C or -80C. Avoid freeze-thaw cycles. Publications: 15 Protocols, Publications, Related Products, Reviews,
Product Datasheet Vanilloid R1/TRPV1 Antibody NB100-1617 Unit Size: 0.05 ml Aliquot and store at -20C or -80C. Avoid freeze-thaw cycles. Publications: 15 Protocols, Publications, Related Products, Reviews,
Neuronal plasma membrane
 ORGANELLES ORGANELLES Neuronal plasma membrane The neuronal plasma membrane contains several local domains with unique properties Presynaptic terminal Endoplasmic Reticulum In neurons the Nissl bodies
ORGANELLES ORGANELLES Neuronal plasma membrane The neuronal plasma membrane contains several local domains with unique properties Presynaptic terminal Endoplasmic Reticulum In neurons the Nissl bodies
Negative Effect of High Calcium Levels on Schwann Cell Survival
 Neurophysiology, Vol. 44, No. 4, September, 2012 Negative Effect of High Calcium Levels on Schwann Cell Survival J.-G. Yan, 1 M. Agresti, 1 L.-L. Zhang 1, H. S. Matloub, 1 and J. R. Sanger 1 Received April
Neurophysiology, Vol. 44, No. 4, September, 2012 Negative Effect of High Calcium Levels on Schwann Cell Survival J.-G. Yan, 1 M. Agresti, 1 L.-L. Zhang 1, H. S. Matloub, 1 and J. R. Sanger 1 Received April
Brian Kahan, D.O. FAAPMR, DABPM, DAOCRM, FIPP Center for Pain Medicine and Physiatric Rehabilitation 2002 Medical Parkway Suite 150 Annapolis, MD
 Brian Kahan, D.O. FAAPMR, DABPM, DAOCRM, FIPP Center for Pain Medicine and Physiatric Rehabilitation 2002 Medical Parkway Suite 150 Annapolis, MD 1630 Main Street Suite 215 Chester, MD 410-571-9000 www.4-no-pain.com
Brian Kahan, D.O. FAAPMR, DABPM, DAOCRM, FIPP Center for Pain Medicine and Physiatric Rehabilitation 2002 Medical Parkway Suite 150 Annapolis, MD 1630 Main Street Suite 215 Chester, MD 410-571-9000 www.4-no-pain.com
Sensory coding and somatosensory system
 Sensory coding and somatosensory system Sensation and perception Perception is the internal construction of sensation. Perception depends on the individual experience. Three common steps in all senses
Sensory coding and somatosensory system Sensation and perception Perception is the internal construction of sensation. Perception depends on the individual experience. Three common steps in all senses
Brain, Behavior, and Immunity
 Brain, Behavior, and Immunity xxx (2010) xxx xxx Contents lists available at ScienceDirect Brain, Behavior, and Immunity journal homepage: www.elsevier.com/locate/ybrbi Involvement of microglial P2X7 receptors
Brain, Behavior, and Immunity xxx (2010) xxx xxx Contents lists available at ScienceDirect Brain, Behavior, and Immunity journal homepage: www.elsevier.com/locate/ybrbi Involvement of microglial P2X7 receptors
Centrally Mediated Anti-Hyperalgesic and Anti-Allodynic Effect of Tolperisone in Spared Nerve Injury Model of Neuropathic Pain
 Centrally Mediated Anti-Hyperalgesic and Anti-Allodynic Effect of Tolperisone in Spared Nerve Injury Model of Neuropathic Pain Keyur S Patel *, Punam D Sachdeva Department of Pharmacology, A.R.College
Centrally Mediated Anti-Hyperalgesic and Anti-Allodynic Effect of Tolperisone in Spared Nerve Injury Model of Neuropathic Pain Keyur S Patel *, Punam D Sachdeva Department of Pharmacology, A.R.College
Spinal Glia and Proinflammatory Cytokines Mediate Mirror-Image Neuropathic Pain in Rats
 1026 The Journal of Neuroscience, February 1, 2003 23(3):1026 1040 Spinal Glia and Proinflammatory Cytokines Mediate Mirror-Image Neuropathic Pain in Rats Erin D. Milligan, 1 Carin Twining, 1 Marucia Chacur,
1026 The Journal of Neuroscience, February 1, 2003 23(3):1026 1040 Spinal Glia and Proinflammatory Cytokines Mediate Mirror-Image Neuropathic Pain in Rats Erin D. Milligan, 1 Carin Twining, 1 Marucia Chacur,
Synapse Formation. Steven McLoon Department of Neuroscience University of Minnesota
 Synapse Formation Steven McLoon Department of Neuroscience University of Minnesota 1 Course News Midterm Exam Monday, Nov 13 9:30-11:30am Bring a #2 pencil!! 2 Course News Lecture schedule: Mon (Oct 31)
Synapse Formation Steven McLoon Department of Neuroscience University of Minnesota 1 Course News Midterm Exam Monday, Nov 13 9:30-11:30am Bring a #2 pencil!! 2 Course News Lecture schedule: Mon (Oct 31)
Analgesic effects of the COX-2 inhibitor parecoxib on surgical pain through suppression of spinal ERK signaling
 EXPERIMENTAL AND THERAPEUTIC MEDICINE 6: 275-279, 2013 Analgesic effects of the COX-2 inhibitor parecoxib on surgical pain through suppression of spinal ERK signaling YA-JING GUO 1, XU-DAN SHI 2, DI FU
EXPERIMENTAL AND THERAPEUTIC MEDICINE 6: 275-279, 2013 Analgesic effects of the COX-2 inhibitor parecoxib on surgical pain through suppression of spinal ERK signaling YA-JING GUO 1, XU-DAN SHI 2, DI FU
Glycine-gated ion channels Converging mechanism and therapeutic potentials
 Glycine-gated ion channels Converging mechanism and therapeutic potentials Li Zhang Laboratory for Integrative Neuroscience, National Institute on Alcohol Abuse and Alcoholism, National Institutes of Health,
Glycine-gated ion channels Converging mechanism and therapeutic potentials Li Zhang Laboratory for Integrative Neuroscience, National Institute on Alcohol Abuse and Alcoholism, National Institutes of Health,
Nature Neuroscience: doi: /nn.2275
 Supplementary Figure S1. The presence of MeCP2 in enriched primary glial cultures from rat or mouse brains is not neuronal. Western blot analysis of protein extracts from (a) rat glial and neuronal cultures.
Supplementary Figure S1. The presence of MeCP2 in enriched primary glial cultures from rat or mouse brains is not neuronal. Western blot analysis of protein extracts from (a) rat glial and neuronal cultures.
Analgesics, pain and tolerance: The St John s discussion
 EDITORIAL Analgesics, pain and tolerance: The St John s discussion Four of the articles in the present issue of Pain Research & Management discuss preclinical issues relevant to understanding the changes
EDITORIAL Analgesics, pain and tolerance: The St John s discussion Four of the articles in the present issue of Pain Research & Management discuss preclinical issues relevant to understanding the changes
Chapter 12 Nervous Tissue. Copyright 2009 John Wiley & Sons, Inc. 1
 Chapter 12 Nervous Tissue Copyright 2009 John Wiley & Sons, Inc. 1 Terms to Know CNS PNS Afferent division Efferent division Somatic nervous system Autonomic nervous system Sympathetic nervous system Parasympathetic
Chapter 12 Nervous Tissue Copyright 2009 John Wiley & Sons, Inc. 1 Terms to Know CNS PNS Afferent division Efferent division Somatic nervous system Autonomic nervous system Sympathetic nervous system Parasympathetic
Modulation of TRP channels by resolvins in mouse and human
 July 9, 2015, Ion Channel Retreat, Vancouver Ion Channel and Pain Targets Modulation of TRP channels by resolvins in mouse and human Ru-Rong Ji, PhD Pain Research Division Department of Anesthesiology
July 9, 2015, Ion Channel Retreat, Vancouver Ion Channel and Pain Targets Modulation of TRP channels by resolvins in mouse and human Ru-Rong Ji, PhD Pain Research Division Department of Anesthesiology
Local Anesthetics. Xiaoping Du Room E417 MSB Department of Pharmacology Phone (312) ;
 Local Anesthetics Xiaoping Du Room E417 MSB Department of Pharmacology Phone (312)355 0237; Email: xdu@uic.edu Summary: Local anesthetics are drugs used to prevent or relieve pain in the specific regions
Local Anesthetics Xiaoping Du Room E417 MSB Department of Pharmacology Phone (312)355 0237; Email: xdu@uic.edu Summary: Local anesthetics are drugs used to prevent or relieve pain in the specific regions
P2 receptors and chronic pain
 Purinergic Signalling (2007) 3:135 144 DOI 10.1007/s11302-006-9045-8 REVIEW P2 receptors and chronic pain Kazuhide Inoue Received: 8 January 2006 / Accepted: 31 January 2006 / Published online: 19 January
Purinergic Signalling (2007) 3:135 144 DOI 10.1007/s11302-006-9045-8 REVIEW P2 receptors and chronic pain Kazuhide Inoue Received: 8 January 2006 / Accepted: 31 January 2006 / Published online: 19 January
Supporting Information
 ATP from synaptic terminals and astrocytes regulates NMDA receptors and synaptic plasticity through PSD- 95 multi- protein complex U.Lalo, O.Palygin, A.Verkhratsky, S.G.N. Grant and Y. Pankratov Supporting
ATP from synaptic terminals and astrocytes regulates NMDA receptors and synaptic plasticity through PSD- 95 multi- protein complex U.Lalo, O.Palygin, A.Verkhratsky, S.G.N. Grant and Y. Pankratov Supporting
San Francisco Chronicle, June 2001
 PAIN San Francisco Chronicle, June 2001 CONGENITAL INSENSITIVITY TO PAIN PAIN IS A SUBJECTIVE EXPERIENCE: It is not a stimulus MAJOR FEATURES OF THE PAIN EXPERIENCE: Sensory discriminative Affective (emotional)
PAIN San Francisco Chronicle, June 2001 CONGENITAL INSENSITIVITY TO PAIN PAIN IS A SUBJECTIVE EXPERIENCE: It is not a stimulus MAJOR FEATURES OF THE PAIN EXPERIENCE: Sensory discriminative Affective (emotional)
Mechanism of Pain Production
 Mechanism of Pain Production Pain conducting nerve fibers are small myelinated (A-delta) or unmyelinated nerve fibers (C-fibers). Cell bodies are in the dorsal root ganglia (DRG) or sensory ganglia of
Mechanism of Pain Production Pain conducting nerve fibers are small myelinated (A-delta) or unmyelinated nerve fibers (C-fibers). Cell bodies are in the dorsal root ganglia (DRG) or sensory ganglia of
Mechanical sensitization of cutaneous sensory fibers in the spared nerve injury mouse model
 Smith et al. Molecular Pain 2013, 9:61 MOLECULAR PAIN SHORT REPORT Open Access Mechanical sensitization of cutaneous sensory fibers in the spared nerve injury mouse model Amanda K Smith, Crystal L O Hara
Smith et al. Molecular Pain 2013, 9:61 MOLECULAR PAIN SHORT REPORT Open Access Mechanical sensitization of cutaneous sensory fibers in the spared nerve injury mouse model Amanda K Smith, Crystal L O Hara
Distinct roles of matrix metalloproteases in the early- and late-phase development of neuropathic pain
 Distinct roles of matrix metalloproteases in the early- and late-phase development of neuropathic pain Yasuhiko Kawasaki,, Zhen-Zhong Xu,, Xiaoying Wang,, Jong Yeon Park, Zhi-Ye Zhuang, Ping-Heng Tan,
Distinct roles of matrix metalloproteases in the early- and late-phase development of neuropathic pain Yasuhiko Kawasaki,, Zhen-Zhong Xu,, Xiaoying Wang,, Jong Yeon Park, Zhi-Ye Zhuang, Ping-Heng Tan,
Pain Pathways. Dr Sameer Gupta Consultant in Anaesthesia and Pain Management, NGH
 Pain Pathways Dr Sameer Gupta Consultant in Anaesthesia and Pain Management, NGH Objective To give you a simplistic and basic concepts of pain pathways to help understand the complex issue of pain Pain
Pain Pathways Dr Sameer Gupta Consultant in Anaesthesia and Pain Management, NGH Objective To give you a simplistic and basic concepts of pain pathways to help understand the complex issue of pain Pain
doi: /brain/awr345 Brain 2012: 135;
 doi:10.1093/brain/awr345 Brain 2012: 135; 359 375 359 BRAIN A JOURNAL OF NEUROLOGY Reduced conduction failure of the main axon of polymodal nociceptive C-fibres contributes to painful diabetic neuropathy
doi:10.1093/brain/awr345 Brain 2012: 135; 359 375 359 BRAIN A JOURNAL OF NEUROLOGY Reduced conduction failure of the main axon of polymodal nociceptive C-fibres contributes to painful diabetic neuropathy
BIOH111. o Cell Module o Tissue Module o Integumentary system o Skeletal system o Muscle system o Nervous system o Endocrine system
 BIOH111 o Cell Module o Tissue Module o Integumentary system o Skeletal system o Muscle system o Nervous system o Endocrine system Endeavour College of Natural Health endeavour.edu.au 1 TEXTBOOK AND REQUIRED/RECOMMENDED
BIOH111 o Cell Module o Tissue Module o Integumentary system o Skeletal system o Muscle system o Nervous system o Endocrine system Endeavour College of Natural Health endeavour.edu.au 1 TEXTBOOK AND REQUIRED/RECOMMENDED
Neurobiology. Cells of the nervous system
 Neurobiology Cells of the nervous system Anthony Heape 2010 1 The nervous system Central nervous system (CNS) Peripheral nervous system (PNS) 2 Enteric nervous system (digestive tract, gall bladder and
Neurobiology Cells of the nervous system Anthony Heape 2010 1 The nervous system Central nervous system (CNS) Peripheral nervous system (PNS) 2 Enteric nervous system (digestive tract, gall bladder and
Synaptic plasticityhippocampus. Neur 8790 Topics in Neuroscience: Neuroplasticity. Outline. Synaptic plasticity hypothesis
 Synaptic plasticityhippocampus Neur 8790 Topics in Neuroscience: Neuroplasticity Outline Synaptic plasticity hypothesis Long term potentiation in the hippocampus How it s measured What it looks like Mechanisms
Synaptic plasticityhippocampus Neur 8790 Topics in Neuroscience: Neuroplasticity Outline Synaptic plasticity hypothesis Long term potentiation in the hippocampus How it s measured What it looks like Mechanisms
Chapter 7. The Nervous System
 Chapter 7 The Nervous System General overview of the nervous system functions Sensory input (info travels in along afferent pathways) Integration (information is processed) Sensory neurons Spinal cord
Chapter 7 The Nervous System General overview of the nervous system functions Sensory input (info travels in along afferent pathways) Integration (information is processed) Sensory neurons Spinal cord
What Does the Mechanism of Spinal Cord Stimulation Tell Us about Complex Regional Pain Syndrome?pme_
 Pain Medicine 2010; 11: 1278 1283 Wiley Periodicals, Inc. What Does the Mechanism of Spinal Cord Stimulation Tell Us about Complex Regional Pain Syndrome?pme_915 1278..1283 Joshua P. Prager, MD, MS Center
Pain Medicine 2010; 11: 1278 1283 Wiley Periodicals, Inc. What Does the Mechanism of Spinal Cord Stimulation Tell Us about Complex Regional Pain Syndrome?pme_915 1278..1283 Joshua P. Prager, MD, MS Center
Biomechanics of Pain: Dynamics of the Neuromatrix
 Biomechanics of Pain: Dynamics of the Neuromatrix Partap S. Khalsa, D.C., Ph.D. Department of Biomedical Engineering The Neuromatrix From: Melzack R (1999) Pain Suppl 6:S121-6. NIOSH STAR Symposium May
Biomechanics of Pain: Dynamics of the Neuromatrix Partap S. Khalsa, D.C., Ph.D. Department of Biomedical Engineering The Neuromatrix From: Melzack R (1999) Pain Suppl 6:S121-6. NIOSH STAR Symposium May
Central and peripheral sites of action for CB 2 receptor mediated analgesic activity in chronic inflammatory and neuropathic pain models in rats
 8.. British Journal of Pharmacology DOI:./j.76-8..6.x www.brjpharmacol.org RESEARCH PAPERbph_6 Central and peripheral sites of action for CB receptor mediated analgesic activity in chronic inflammatory
8.. British Journal of Pharmacology DOI:./j.76-8..6.x www.brjpharmacol.org RESEARCH PAPERbph_6 Central and peripheral sites of action for CB receptor mediated analgesic activity in chronic inflammatory
Chapter 11: Functional Organization of Nervous Tissue
 Chapter 11: Functional Organization of Nervous Tissue I. Functions of the Nervous System A. List and describe the five major nervous system functions: 1. 2. 3. 4. 5. II. Divisions of the Nervous System
Chapter 11: Functional Organization of Nervous Tissue I. Functions of the Nervous System A. List and describe the five major nervous system functions: 1. 2. 3. 4. 5. II. Divisions of the Nervous System
