Treatment of cutaneous leishmaniasis among travellers
|
|
|
- Lucas Davidson
- 6 years ago
- Views:
Transcription
1 Journal of Antimicrobial Chemotherapy (2004) 53, DOI: /jac/dkh058 Advance Access publication 16 January 2004 Treatment of cutaneous leishmaniasis among travellers J. Blum 1 *, P. Desjeux 2, E. Schwartz 3,4, B. Beck 1 and C. Hatz 1 1 Swiss Tropical Institute, Socinstrasse 57, 4002 Basel; 2 World Health Organization, CPE/EPH, Avenue Appia 20, 1211 Geneva 27, Switzerland; 3 The Center for Geographic Medicine and Department of Medicine C, The Chaim Sheba Medical Center, Tel Hashomer 52621; 4 Sackler Faculty of Medicine, Tel Aviv University, Tel Aviv, Israel Leishmaniasis is endemic in 88 countries on five continents. There are million cases of cutaneous leishmaniasis reported yearly worldwide. There has been a sharp increase in recorded cases over the last 10 years. Based on geographical distribution, cutaneous leishmaniasis is divided into Old World and New World leishmaniasis. In the past, species could be inferred from geographical setting or determined by performing culture and isoenzyme analysis. The recently developed and now widely available PCR technology allows a rapid diagnosis with determination of most species, and thus enables a species-orientated treatment. While the Old World species mostly cause benign and often self-limiting cutaneous disease, the American species cause a broad spectrum of conditions from benign to severe manifestations, including mucosal involvement. The response to treatment varies according to the species. Therefore, a speciesspecific approach is proposed. Drugs for systemic and topical treatment are presented and discussed with regard to their application, use and adverse effects. Indications for local or systemic treatment are proposed. Drugs under investigation are also mentioned. An overview of published treatment options and a treatment recommendation is given for each of the most important species. The level of evidence of the studies leading to these recommendations is given. Keywords: cutaneous leishmaniasis, travel, systemic treatment, local treatment Introduction The large number of travellers from industrialized countries visiting and increasingly enjoying outdoor activities in endemic areas are at considerable risk of contracting cutaneous leishmaniasis. Most physicians in industrialized countries have little experience with cutaneous leishmaniasis in returning travellers. This often leads to delayed diagnosis and inappropriate management. This review aims to provide recommendations for the rational and effective treatment of patients presenting with cutaneous leishmaniasis of various origins. Evidence-based data on travellers are limited to a few studies, anecdotal reports and investigations among military personnel deployed in endemic areas. 1 3 It is therefore crucial to consider the published experience among patients from endemic areas, even if the immunological background will be different in an important part of the respective study populations. 4 6 Consequently, established treatment schedules can be applied to all patients. Leishmaniasis is endemic in 88 countries on five continents. There are million cases of cutaneous leishmaniasis reported yearly among the local populations. The number of reported cases has increased sharply over the last 10 years. 7 The parasite is transmitted by the bite of various types of phlebotomine sandflies. Based on its geographical distribution, cutaneous leishmaniasis can be divided into Old World (including southern Europe, the Middle East, parts of south-west Asia and Africa) and New World (from southern USA through Latin America to the highlands of Argentina) leishmaniasis. Cutaneous leishmaniasis is caused by various Leishmania species. While Old World species mostly cause benign and often self-limiting cutaneous disease, New World species cause a broad spectrum of conditions from benign to severe manifestations, including mucosal involvement. The clinical spectrum of the disease and its response to treatment vary according to the species. Therefore, a species-specific approach should be considered. 8 The traditional method is to perform an isoenzyme analysis on Leishmania culture to determine the species. Species can often be inferred from geographical setting, thus adding to the accuracy of the diagnosis. In addition, the recently developed PCR technology allows a rapid species-specific diagnosis for most species. 9 The introduction of this novel diagnostic species differentiation using PCR methodology further contributes to the possibility of a targeted, species-orientated treatment. Treatment options: local versus systemic treatment The proposed choice of local or systemic treatment of cutaneous leishmaniasis (Table 1) is guided by the risk of developing mucosal disease. Mucosal leishmaniasis is mainly attributed to Leishmania brasiliensis, but is also described in Leishmania panamensis, 10,11 Leishmania... *Corresponding author. Tel: ; Fax: ; johannes.blum@unibas.ch JAC vol.53 no.2 The British Society for Antimicrobial Chemotherapy 2004; all rights reserved.
2 Table 1. Proposed indications for local and systemic treatment Proposed indications for local treatment lack of risk of developing mucosal lesions Old World cutaneous leishmaniasis L. mexicana cutaneous leishmaniasis small, single lesion absence of lymph node metastasis Proposed indications for systemic treatment presence of mucosal lesion or lymph node metastasis New World cutaneous leishmaniasis except L. mexicana lesions lesions unresponsive to local treatment guyanensis 12 and Leishmania amazonensis. 13 Mucosal lesions were found in Colombia among 19/397 (5%) patients with L. panamensis and among 10/91 (11%) patients with L. brasiliensis infection. 14 The risk of late mucosal disease in cases due to L. brasiliensis has been reported to be 2 10% among untreated cases. 15,16 However, the development of mucosal disease is subject to variation according to place. Because published data do not allow the establishment of the relative risk of mucosal disease for every species, systemic treatment is proposed for all New World species, except for Leishmania mexicana infection, where the risk of developing mucosal leishmaniasis is almost zero. 17 There is evidence that early systemic treatment may prevent mucosal lesions, which are seen more frequently in patients with incomplete or missing antimony treatment. 15,16,18 Although mucosal lesions have been reported in Leishmania donovani 19 infections in Sudan, it is generally accepted to approach Old World cutaneous leishmaniasis with local treatment. Local treatment is applied in patients with a small, single lesion. Based on expert opinion, systemic treatment is used in patients with multiple lesions or large lesions (>5 cm). Systemic treatment is also recommended in patients with metastatic spread and in cutaneous lesions unresponsive to local treatment. Local treatment may be considered in patients for whom systemic treatment is contraindicated, such as those who are pregnant or have cardiac problems Systemic treatment (Table 2) Pentavalent antimonials The pentavalent antimonials meglumine antimonate (85 mg Sb/mL) for intramuscular administration and sodium stibogluconate (100 mg Sb/mL) for intravenous and intramuscular administration have been used for decades for the treatment of New World cutaneous leishmaniasis, and are the gold standard for other new investigational drugs. The biochemical basis for their effectiveness is unknown, but may involve inhibition of ATP synthesis. The drugs exist only in parenteral forms. The dosage is usually given in Sb equivalents (mg/kg/day). The pentavalent antimonials are far from being ideal drugs because of their difficult administration and toxicity. 3,21,22 Treatment schedules and dosages have been debated and changed several times. In the 1980s, the dosage was increased from 10 to 20 mg/kg/day, with an upper limit of 850 mg/day (equivalent to two ampoules of meglumine antimonate). The upper limit of 850 mg Sb/day was abandoned in the early 1990s, 22 since studies indicated a reduced efficacy of lower doses and no higher toxicity was found with the higher doses of the drug. 1 However, the question of the optimal dose has not yet been finally answered. The results of trials in Brazil with lower doses have been published. 23,24 A possible disadvantage of low dosages and/or a short course of treatment is that they could contribute to the appearance of resistance. Pentamidine Pentamidine, an aromatic diamidine, is toxic for a number of protozoa and fungi including Leishmania, Pneumocystis carinii and African trypanosomes. The mechanism of action has not been established. Pentamidine is used as an alternative to the pentavalent antimonials, and is the first line treatment for cutaneous leishmaniasis in French Guyana, where L. guyanensis is responsible for >90% of the cases. 6,25 A study in Colombia (with L. panamensis, L. brasiliensis and L. mexicana) found that a short-course, low-dose regimen of pentamidine isethionate had a similar cure rate (96%) to that of meglumine antimonate (91%), with a similar rate of side effects. 26,27 The short-course, low-dosage regimen of pentamidine in otherwise healthy patients with cutaneous leishmaniasis was better tolerated than the higher dosages applied for Pneumocystis treatment in HIV-positive patients: all adverse effects were reversible and no cases of new diabetes mellitus were found among >2200 patients observed with the low-dose regimen. 6,25,26 However, the higher dosages needed for the treatment of mucosal leishmaniasis (>2000 mg) may cause diabetes mellitus. 28 Blood sugar and glycaemia need to be checked before every injection, because reversible glucosuria and hyperglycaemia have been described after just one dose of pentamidine 200 mg. 29 Imidazoles/triazoles The imidazoles and the structurally related triazoles were introduced as antifungal drugs, but also have an antileishmanial activity. They have the advantage of oral administration and few adverse effects, but are only effective against some species (see species-specific treatment below). Itraconazole has also been used, but data are scarce and controversial. Fluconazole was studied in a randomized, double-blind, placebocontrolled trial in Iran. 30 It was well tolerated and showed promising results in Leishmania major leishmaniasis (see below). Data on other species of leishmaniasis are lacking. Drugs under investigation Miltefosine. Miltefosine, a phosphocholine analogue, showed high in vitro activity against leishmania. The results of an uncontrolled trial in Colombia (Phase I/II) are promising. Using doses of mg/ day for 3 4 weeks, the per protocol cure rate (no parasites after therapy, complete re-epithelialization after 3 months) was 94%. However, a longer follow-up is needed to evaluate the relapse rate. The most common side effects were motion sickness, gastrointestinal complaints, headache and raised liver enzymes. 31 Further controlled studies with various species are needed before miltefosine can be proposed as a routine treatment of cutaneous leishmaniasis. Amphotericin B/liposomal amphotericin B. The antifungal agent amphotericin B desoxycholate is active against Leishmania species, but has the disadvantage of a high incidence of adverse reactions (i.e. hyperpyrexia, severe malaise, hypotension, thrombophlebitis, azotaemia, renal tubular damage, hypokalaemia, anaemia and hepatitis). Several amphotericin B lipid formulations with much lowertoxicities than the free drug have been developed, and have proved to be 159
3 Table 2. Drugs and follow-up for treatment of cutaneous leishmaniasis Drug Adverse effect Management/follow-up Pentavalent antimonials Pentamidine cardiac toxicity with reversible ECG alterations is seen in 30 60%: repolarization alterations affecting T wave and ST segment, prolongation of the corrected QT interval; fatal arrhythmias have not been documented with the usual dose of 20 mg Sb/kg 22,76,77 hepatotoxicity seen in 50%; reversible haematotoxicity (anaemia, leucopenia, thrombopenia) 79 hyperamylasaemia tended to occur very early in therapy and to decline despite continued treatment with antimonials subjective complaints: musculoskeletal symptoms, headache, gastrointestinal complaints, pain at the injection site rare complications: glomerulonephritis, acute renal failure, 81 peripheral nephritis, 82 exfoliate dermatitis, herpes zoster 83 aseptic abscess (accidental contact of pentamidine with the subcutaneous tissue) diabetes, hypoglycaemia, proteinuria hypotension 6,26 subjective complaints: myalgia, nausea and gustative abnormalities, headache, pain at the injection side, abdominal pain 6 ECG checks 1 2 every week; interruption of treatment if: (a) significant arrhythmias; (b) QTc longer than 0.5 s (QTc longer than 0.45 s: monitoring/dose reduction); (c) concave ST segment transaminases weekly; treatment interruption if transaminases >5 the upper limit of normal value (ULN) 78 haemoglobin, leucocytes and platelets weekly amylase daily during the first week, then twice weekly; treatment interruption if serum amylase levels became >4 the ULN or lipase levels >15 the ULN, regardless of symptoms; therapy can be resumed, once these values tend significantly towards normal 3,80 weekly examination of urine, creatinine pentamidine has to be injected slowly and strictly intramuscular with a long needle (50 mm) fasting glycaemia and urine for proteinuria and gylcosuria have to be checked before every injection and 3 weeks and 2 months after the last injection 84 blood pressure and heart rate have to be measured before and after the injection (every 15 min for 1 h) 84 Ketoconazole hepatotoxicity reversible, usually mild 54 transaminases weekly; treatment interruption if transaminases >5 ULN diminution of testosterone values (70%), but without reversible, no controls needed diminution of libido or beard growth 54 subjective complaints: abdominal pain, headache, nausea, fever and malaise 54 Fluconazole hepatotoxicity transaminases; treatment interruption if transaminases >5 ULN allergic skin reactions haematotoxicity (anaemia, leucopenia, thrombopenia) haemoglobin, leucocytes and platelets subjective complaints: headache, gastrointestinal complaints useful in the treatment of visceral leishmaniasis. Based on currently available data, liposomal amphotericin B has been insufficiently studied with regard to formulation and dosage to assess its efficacy in cutaneous leishmaniasis. Allopurinol. Allopurinol, an analogue of hypoxanthine, is generally not effective in the absence of pentavalent antimony However, a combination of allopurinol (20 mg/kg/day for 15 days) and stibogluconate (20 mg/kg/day for 15 days) was more effective in L. panamensis (cure rate 71 74%) than stibogluconate alone at the same dose (cure rate 36 39%). 36,37 Drugs influencing the immune system. Immunomodulators represent an interesting possibility for improving the cure rates of pentavalent antimonials, but the lack of solid data does not allow respective recommendations at present. Other drugs. Other drugs, such as rifampicin, 38 dapsone 39 and oral zinc sulphate, 40 have been tested for leishmaniasis treatment. Some are promising, but the results require confirmation before they can be recommended for treatment. 2. Local treatment Physical methods Cutaneous leishmaniasis has been treated in patients of all ages with a wide range of physical methods, including cauterization, surgical excision, cryotherapy and the application of local heat. Cryotherapy 160
4 is performed by repeated topical applications of liquid nitrogen with a cotton-tipped applicator or a cotton swab with moderate pressure to the lesion, up to 2 mm outside the lesion margin. The freezing time per application is s. The procedure is repeated two or three times at short intervals, resulting in a total time of s. Adequate application is reflected in the whitening of the skin at 2 3 mm outside the margins of the lesion The usual post-freeze pattern is some oedema and blistering of the lesion itself for 2 3 days, followed by crusting and formation of an eschar. 41,42 Uncontrolled studies in Old World leishmaniasis, which did not mention the species, gave cures rates of close to 100% after one to three sessions of cryotherapy in Egypt (30/30 patients with clinical and parasitological cure within 4 5 weeks), 41 Jordan (effective and significant clinical response in 214/215 patients within an undetermined time) 43 and Israel (complete clinical healing in 40 lesions in 14 patients within 3 8 weeks). 42 However, in a comparative, non-randomized study from Turkey, the cure rates (complete healing and disappearance of all clinical features) after one or two sessions of cryotherapy were 77% (46/60) after 1 month and 73% (44/60) after 3 months, compared with 85% after 1 and 3 months following intralesional sodium stibogluconate. 44 In an uncontrolled study in Iraq, two sessions with local heat (55 C over 5 min) provided by an infrared lamp and focused on the lesion gave a cure of 177/178 lesions (no species determination performed). 45 However, practical experience with local heat from an infrared lamp shows that healing of the lesion was almost invariably accompanied by a heat-induced skin bulla. Ointment containing 15% paromomycin Topical formulations offer significant advantages over systemic therapy, such as ease of administration, fewer adverse effects and cost effectiveness. However, percutaneous absorption is hampered by the intact skin, mainly in the stratum corneum. Topical applications may be applied to open lesions that have lost their stratum corneum barrier property, but are less successful in lesions where absorption is hindered by epithelial thickening. Paromomycin, an aminoglycoside antibiotic (identical to aminosidine) has been used systemically against both visceral and cutaneous leishmaniasis. As an ointment for topical use, it has been tested in different formulations, either with methylbenzethonium in white soft paraffin, or with urea and white soft paraffin. The combination of paromomycin with methylbenzethonium appears to be more effective than the combination with urea, but it causes more local inflammatory reactions. 46 With New World leishmaniasis, however, experts are hesitant to treat cutaneous lesions topically because of the risk of mucosal disease, although this ointment was reported to have much better cure rates than placebo in two studies. 47,48 Local infiltration with pentavalent antimony Local infiltration of lesions with pentavalent antimony produces the maximum concentration in the lesions and has few systemic side effects, but does not reach metastatic infections. The basic aim is to fill the infected part of the dermis with pentavalent antimony. This means carefully infiltrating the area around the lesion, including the base of the lesion. A fine gauge (25G) needle is used to inject the drug under pressure as the needle advances. Injection into the dermis is difficult, as the tissue space is small. The drug must not be injected Figure 1. Procedure for intralesional treatment with pentavalent antimony (from reference 25). Reproduced with permission from WHO. into the subcutaneous tissue, where it is rapidly absorbed and does not reach the site of infection. The infiltration is performed in a V-shaped pattern, advancing the needle into the base of the lesion (see Figure 1). The solution is injected under the edges of the lesion and the entire lesion until the surface has blanched. Treatment should be given every 5 7 days, a total of two to five times. If the lesion is not healing after five treatments, it should be reviewed in 1 month, when a decision about reverting to systemic treatment should be made. 49 Intralesional infiltration is painful and requires some experience to perform. 50 Species-specific therapy (Table 3) (A) New World leishmaniasis L. mexicana: local treatment. Strains of L. mexicana isolated from Belizean patients were found to be highly susceptible to paromomycin sulphate both in vitro and in animal studies. 51 An ointment with 15% paromomycin/12% methylbenzethonium chloride applied twice daily over 20 days was used in Guatemala and in Belize, where L. brasiliensis and L. mexicana are endemic. In Guatemala, the final clinical response rate at the 12 months follow-up examination was higher in the treatment group (31/35, 88.6%) than in the placebo group (13/33, 39.4%) (P 0.001). 47 In Belize, the cure rate of 53 patients was 74%. 52 L. mexicana: systemic treatment. Ketoconazole (600 mg daily for 28 days) was compared with sodium stibogluconate in patients with cutaneous leishmaniasis in Guatemala. The outcome was related to the species. Whereas ketoconazole had a higher cure rate for L. mexicana (8/9 versus 4/7), the response rate in L. brasiliensis was much lower (7/23 versus 24/25). 53 Considering the virtually non-existant risk of mucosal leishmaniasis with L. mexicana infection, 15% paromomycin/12% methylbenzethonium ointment is recommended as the first-line treatment. Where it is not available, infiltration with glucantime may be an easily accessible option, although no firm data have been published. Systemic 161
5 treatment with ketoconazole is another documented option. The choice depends on the clinical aspect of the lesion (see above). L. panamensis: local treatment. The 1 year cure rate with a 15% paromomycin/12% methylbenzethonium chloride ointment was 85% in 52 Ecuadorian patients, compared with 9% in a non-randomized untreated control group. 48 Nevertheless, experts are reluctant to apply local treatment because of the risk of mucosal leishmaniasis. L. panamensis: systemic treatment. (i) Ketoconazole. In a randomized clinical trial, ketoconazole (600 mg daily for 28 days) had a similar efficacy (16/21, 76%) to sodium stibogluconate (20 mg/kg/day with upper limit of 850 mg, resulting in 13 mg/kg/day) (13/19, 68%), but a much better efficacy than placebo (0/11). 54 (ii) Pentavalent antimonials. The response to antimonials is dose dependent: patients treated with mg/kg/day 1,54 had cure rates of only 68 72%, whereas patients treated with 20 mg/kg/day for 20 days had a % cure rate. 1 The optimal duration of treatment with the dose of 20 mg/kg/day is still debated. Whereas some authors regard a 10 day course of treatment to be sufficient, 2 and equal to a 20 day regimen, 5 other studies indicate a clear positive correlation between treatment duration and efficacy: in one study the cure rates were 20% for 3 days, 53% for 7 days and 84% for 20 days treatment duration. 55 In Guatemala, a surprisingly insufficient response (36 39%) to a 15 day course of pentavalent antimonials could be improved to 71 74% by the addition of allopurinol (20 mg/kg/day/15 days, given in four divided doses). 36,37 This combination was not compared with the usual 20 day course of stibogluconate. However, the addition of allopurinol to stibogluconate provided no clinical benefit in patients with mucosal leishmaniasis. 56 Considering the oral application and the lower rate of side effects with ketoconazole, it can be recommended as the first choice for uncomplicated lesions (i.e. not multiple, not long-lasting lesions, with no sign of mucosal involvement), especially when a close clinical follow-up and patient compliance are guaranteed. Pentavalent antimonials are used if the response to ketoconazole is not satisfactory. Either a 20 day course or the combination with allopurinol is recommended. L. guyanensis. There are only a few, non-randomized trials concerning the optimal treatment of L. guyanensis. The cure rate (6 months) of antimonials (20 mg Sb/kg/day for 20 days) was significantly lower in patients infected with L. guyanensis (26.3%) than in patients infected with L. brasiliensis (50.8%; P = 0.003). 57 In French Guyana, where L. guyanensis is responsible for >90% of cutaneous leishmaniasis, a short-course regimen of pentamidine is the first-line treatment. The cure rates are dose dependent (600 mg, 73%; 900 mg, 90%; 8 mg/kg/day, 90%). A total dose of 1200 mg was proposed. 25 Two injections with 4 mg/kg/day had a cure rate of 89%. Patients with satellite papules or more than three lesions were at a relatively high risk of not being cured. A second treatment with the same dosage had a cure rate of 80%. 6 These doses were in the same range as the dosages proposed by Soto in Columbia (i.e. pentamidine isethionate given in four injections of each 3 mg/kg/day every other day). 26,27 Considering the poor response to pentavalent antimonials and the good experience in French Guyana, pentamidine is recommended as the first choice, in a dosage of four injections containing 3 mg/kg/day every other day. Local treatment is not recommended due to a lack of sound data. L. brasiliensis. Because of the high risk of late mucosal disease in infections with L. brasiliensis, systemic treatment with pentavalent antimonials (Sb 20 mg/kg/day for 20 days) is the gold standard. 3,15,16,22,58 The cure rates found in various studies range from low (50%) 57,59 to excellent (96 100%). 2,21,53 Different study sites and strains as well as different patient selection may contribute to these varying findings. 57 Studies with lower doses gave controversial results. In Guatemala, a shorter course with only 15 mg/kg/day over 14 days gave a final cure rate of 64%, 60 whereas studies from the state of Rio de Janeiro (Brazil) indicated that lower dosages gave similar results to higher doses. Oliveira-Neto et al. 24 compared a low-dose regimen (Sb 5 mg/kg/day for 30 days) with the conventional dosage of Sb 20 mg/kg/day. The cure rates were similar in the two groups (10/12 versus 9/11) after 30 days, with a lower toxicity in the lower dosage group. 24 Oliveira-Neto et al. 23 treated 156 patients with the lower dose; 84% were cured and did not develop mucosal disease or relapse during an observation period of 5 10 years. However, the authors concluded that although this low-dose regimen is adequate for this particular endemic region, it should not be applied without confirmation in other endemic settings. The diversity of L. brasiliensis susceptibility to pentavalent antimonials means that the chance of a favourable treatment response can not be evaluated in the individual traveller. Therefore, the current recommendation to give pentavalent antimonials at the dosage of 20 mg/kg/day for 20 days is maintained. 22,58 Poor clinical response in some studies raises the question of antimonial resistance; however, there is no published evidence to date to confirm true resistance in cutaneous leishmaniasis. Other species. Data on the treatment of other species of New World cutaneous leishmaniasis like L. amazonensis, Leishmania venezuelensis or Leishmania peruviana are scarce. Therefore, the current recommendation of pentavalent antimonials at the dosage of 20 mg/kg/day for 20 days is still valid. 22,50,58 (B) Old World leishmaniasis L. major. The risk of metastatic lesions, including mucosal leishmaniasis, is almost zero, except for L. donovani in Sudan, and local treatment is often used. An ointment containing 15% paromomycin and 12% methylbenzethonium chloride (applied twice daily for 10 days) was more effective than placebo in L. major cutaneous leishmaniasis (cure 29/ 39 versus 4/15). 61 The parasitological cure rate for the same ointment was 72% (48/67) after 10 days. After an additional 20 days, the rate was 87% (58/67). 62 Apart from local inflammation and pain, the ointment was well tolerated. The use of 15% paromomycin/12% urea ointment in two randomized, placebo-controlled studies on L. major in Iran 63 and Tunisia 64 could not demonstrate any clinical benefit, even though there was a significant parasitological response at day 15 in the Iran study. Local infiltration with antimonials (sodium stibogluconate 100 mg/ml ml/lesion or meglumine antimonate ml/ lesion) was studied in Saudi Arabia in regions where L. major and Leishmania tropica are endemic, and where lesions of acute leishmaniasis often heal spontaneously within 7 12 months. Two to 15 infiltrations (from all sides, until the lesion has blanched) were needed to achieve cure rates of 72 99% In a randomized, double-blind, placebo-controlled trial in Iran, fluconazole 200 mg daily for 6 weeks showed promising results. Healing of lesions was complete for 63/80 (79%) at the 3 months follow-up in the fluconazole group, but only in 22/65 (34%) in the 162
6 Table 3. Treatment by species Species Drug Dosage Level of evidence a L. mexicana local: ointment: 15% paromomycin plus 12% twice daily for 20 days B 47,52 methylbenzethonium chloride ketoconazole 600 mg daily for 28 days B 53 L. panamensis ketoconazole 600 mg daily for 28 days A 54 pentavalent antimonials 20 mg Sb/kg/day for 20 days A 1,2 pentavalent antimonials and in addition allopurinol 20 mg Sb/kg/day for 15 days A 36,37 20 mg/kg/day given in four doses for 15 days L. guyanensis pentamidine isethionate four injections of 3 mg/kg/day every other day L. brasiliensis pentavalent antimonials 20 mg Sb/kg/day for 20 days A 22,53,58 L. major 15% paromomycin/12% methylbenzethonium twice daily for days A 61,62 chloride ointment localized heat or cryotherapy two sessions with localized heat (55 C C 45 for 5 min) two to three sessions of topical application of liquid nitrogen C local infiltration with antimonials sodium stibogluconate, meglumine antimonate A complete blanching of lesion has to be achieved upper limit 5 ml per infiltration and 20 mg Sb/kg once or twice weekly one to five infiltrations fluconazole 200 mg daily for 6 weeks A 30 L. tropica/l. infantum local infiltration with antimonials sodium stibogluconate, meglumine D antimonate complete blanching of lesion has to be achieved upper limit 5 ml per infiltration and 20 mg Sb/kg once to twice weekly one to five infiltrations 15% paromomycin/12% methylbenzethonium twice daily for days D chloride ointment cryotherapy or localized heat two to three sessions of topical application of C 41 43, D liquid nitrogen; two sessions with localized heat (55 C for 5 min) pentavalent antimonials 20 mg Sb/kg/day for days D fluconazole 200 mg daily for 6 weeks D C 6,25 a Level of evidence: recommendation grade. (A) Randomized, controlled trial in representative collective. (B) Randomized, controlled trial in partially representative (small patient number, different species included) collective. Cohort trial or case control study in representative collective. (C) Cohort trial or case control study in partially representative collective, series of cases in representative collective. (D) Series of cases in partially representative (small patient number, different species included) collective, informal expert opinion, other information. placebo group. 30 Ketoconazole is less well studied, but appeared to be effective in L. major in Israel. The cure rate was 70% after mg daily for 4 6 weeks. 68,69 Pentavalent antimonials were efficient in six patients with a dose of 20 mg/kg/day over days, 2 and are an alternative in cases of treatment failure. An ointment containing 15% paromomycin and 12% methylbenzethonium chloride, intralesional pentavalent antimonials and thermotherapy (see above) are possible first choices in this usually self-limiting disease, although these different treatment options have rarely been tested against one another. The choice between them depends on the experience of the treating physician and the availability of the method. If systemic treatment is indicated, fluconazole or ketoconazole is recommended. L. tropica/leishmania infantum. There are no randomized, doubleblind, placebo-controlled clinical studies for treatment of these species. 163
7 According to estimates made by the WHO, 90% of L. tropica lesions in Pakistan could be healed by intralesional pentavalent antimonials. 49 This treatment gave cure rates of 72 99% in Saudi Arabia, where L. tropica and L. major are endemic (see above) However, recent experience in Israel, using PCR for speciesspecific diagnosis, 70 showed recurrent failure of local paromomycin and intralesional sodium stibogluconate treatment against L. tropica, and a good response to 10 days of systemic sodium stibogluconate treatment (E. Schwartz, unpublished results). In studies using ketoconazole, L. tropica appeared to be less responsive than L. major. 68 In India, a 10 week course of ketoconazole 400 mg/day was ineffective in patients with L. tropica lesions. 71,72 Anecdotal reports have shown a therapeutic response of L. infantum lesions to intralesionally injected N-methylglucamine. 73 Thermotherapy, intralesional pentavalent antimonials and ointments are possible first choices in this usually self-limiting disease. Again, the choice depends on the experience of the treating physician and the availability of the method. The role of fluconazole in the treatment of L. infantum and L. tropica has not yet been established. Clinical experience has shown that pentavalent antimonials are effective in severe infections with big or multiple lesions on the face or over the joints. 2 Management issues: clinical follow-up and treatment failure Cutaneous leishmaniasis lesions may demonstrate only a partial clinical response after 3 4 weeks, and may not completely heal until several weeks after completion of treatment. Therefore, patients should be re-evaluated 4 6 weeks after the completion of treatment. In patients with New World cutaneous leishmaniasis, management is more complicated, since in most cases systemic treatment is warranted. The treatment of choice is pentavalent antimonials, which require meticulous follow-up because of the potential for adverse events (see Table 2). Thus, in many centres these patients are treated in hospital, although some authors have had good experience with ambulatory care. 21 The most common and worrisome laboratory abnormalities are elevated liver enzymes and asymptomatic hyperamylasaemia. We usually continue treatment if liver enzymes are below five times the normal range and amylase less than five times the normal range (Table 2). Interestingly, by decreasing the dose or interrupting treatment for several days, the abnormal values go back towards normal, and after reinstitution of the drug, the abnormal laboratory findings may not recur. 74 Relapse may occur several months after complete cure. It is important to distinguish between clinical and parasitological healing. Despite clinical healing of a lesion, PCR can remain positive several years later. 75 References 1. Ballou, W. R., McClain, J. B., Gordon, D. M. et al. (1987). Safety and efficacy of high-dose sodium stibogluconate therapy of American cutaneous leishmaniasis. Lancet ii, Wortmann, G., Miller, R. S., Oster, C. et al. (2002). A randomized, double-blind study of the efficacy of a 10- or 20-day course of sodium stibogluconate for treatment of cutaneous leishmaniasis in United States military personnel. Clinical Infectious Diseases 35, Aronson, N. E., Wortmann, G. W., Johnson, S. C. et al. (1998). Safety and efficacy of intravenous sodium stibogluconate in the treatment of leishmaniasis: recent U.S. military experience. Clinical Infectious Diseases 27, Gutierrez, Y., Salinas, G. H., Palma, G. et al. (1991). Correlation between histopathology, immune response, clinical presentation, and evolution in Leishmania braziliensis infection. American Journal of Tropical Medicine and Hygiene 45, Palacios, R., Osorio, L. E., Grajalew, L. F. et al. (2001). Treatment failure in children in a randomized clinical trial with 10 and 20 days of meglumine antimonate for cutaneous leishmaniasis due to Leishmania viannia species. American Journal of Tropical Medicine and Hygiene 64, Nacher, M., Carme, B., Sainte, M. D. et al. (2001). Influence of clinical presentation on the efficacy of a short course of pentamidine in the treatment of cutaneous leishmaniasis in French Guiana. Annals of Tropical Medicine and Parasitology 95, Desjeux, P. (2001). Worldwide increasing risk factors for leishmaniasis. Medical Microbiology and Immunology 190, Blum, J., Hatz, C. & Junghanss, T. (1994). The therapy of cutaneous and mucocutaneous leishmaniasis. Deutsche Medizinische Wochenschrift 119, Marfurt, J., Niederweiser, I., Makia, N. D. et al. (2003). Diagnostic genotyping of Old and New World Leishmania species by PCR-RFLP. Diagnostic Microbiology and Infectious Disease 46, Osorio, L. E., Castillo, C. M. & Ochoa, M. T. (1998). Mucosal leishmaniasis due to Leishmania (Viannia) panamensis in Colombia: clinical characteristics. American Journal of Tropical Medicine and Hygiene 59, Saenz, R. E., Paz, H. M., de Rodriguez, G. C. et al. (1989). Mucocutaneous leishmaniasis in Panama. Etiologic agent, epidemiologic and clinical aspects. Revista Medica de Panama 14, Santrich, C., Segura, I., Arias, A. L. et al. (1990). Mucosal disease caused by Leishmania braziliensis guyanensis. American Journal of Tropical Medicine and Hygiene 42, Lucas, C. M., Franke, E. D., Cachay, M. I. et al. (1998). Geographic distribution and clinical description of leishmaniasis cases in Peru. American Journal of Tropical Medicine and Hygiene 59, Saravia, N. G., Segura, I., Holguin, A. F. et al. (1998). Epidemiologic, genetic, and clinical associations among phenotypically distinct populations of Leishmania (Viannia) in Colombia. American Journal of Tropical Medicine and Hygiene 59, Marsden, P. D. (1986). Mucosal leishmaniasis ( espundia Escomel, 1911). Transactions of the Royal Society of Tropical Medicine and Hygiene 80, Jones, T. C., Johnson, W. D., Jr, Barretto, A. C. et al. (1987). Epidemiology of American cutaneous leishmaniasis due to Leishmania braziliensis braziliensis. Journal of Infectious Diseases 156, Andrade-Narvaez, F. J., Vargas-Gonzalez, A., Canto-Lara, S. B. et al. (2001). Clinical picture of cutaneous leishmaniases due to Leishmania (Leishmania) mexicana in the Yucatan peninsula, Mexico. Memorias do Instituto Oswaldo Cruz 96, Blum, J., Junghanss, T. & Hatz, C. (1994). Erroneous tracks in the diagnosis of cutaneous and mucocutaneous leishmaniasis. Schweizerische Rundschau fur Medizin Praxis 83, El Hassan, A. M. & Zijlstra, E. E. (2001). Leishmaniasis in Sudan. Mucosal leishmaniasis. Transactions of the Royal Society of Tropical Medicine and Hygiene 95, Suppl. 1, S Oliveira-Neto, M. P., Schubach, A., Mattos, M. et al. (1997). Intralesional therapy of American cutaneous leishmaniasis with pentavalent antimony in Rio de Janeiro, Brazil an area of Leishmania (V.) braziliensis transmission. International Journal of Dermatology 36, Seaton, R. A., Morrison, J., Man, I. et al. (1999). Out-patient parenteral antimicrobial therapy a viable option for the management of cutaneous leishmaniasis. Quarterly Journal of Medicine 92, Herwaldt, B. L. & Berman, J. D. (1992). Recommendations for treating leishmaniasis with sodium stibogluconate (Pentostam) and 164
8 review of pertinent clinical studies. American Journal of Tropical Medicine and Hygiene 46, Oliveira-Neto, M. P., Schubach, A., Mattos, M. et al. (1997). A lowdose antimony treatment in 159 patients with American cutaneous leishmaniasis: extensive follow-up studies (up to 10 years). American Journal of Tropical Medicine and Hygiene 57, Oliveira-Neto, M. P., Schubach, A., Mattos, M. et al. (1997). Treatment of American cutaneous leishmaniasis: a comparison between low dosage (5 mg/kg/day) and high dosage (20 mg/kg/day) antimony regimens. Pathologie Biologie 45, Pradinaud, R. (1994). Le traitement de la leishmaniose tégumentaire par la pentamidine en Guyane française. Medecine Tropicale 54, Soto-Mancipe, J., Grogl, M. & Berman, J. D. (1993). Evaluation of pentamidine for the treatment of cutaneous leishmaniasis in Colombia. Clinical Infectious Diseases 16, Soto, J., Buffet, P., Grogl, M. et al. (1994). Successful treatment of Colombian cutaneous leishmaniasis with four injections of pentamidine. American Journal of Tropical Medicine and Hygiene 50, Amato, V., Amato, J., Nicodemo, A. et al. (1998). Treatment of mucocutaneous leishmaniasis with pentamidine isothionate. Annales de Dermatologie et de Venereologie 125, Tomkins, A. & Bryceson, A. (1972). Ocular leishmaniasis and pentamidine diabetes. Transactions of the Royal Society of Tropical Medicine and Hygiene 66, Alrajhi, A. A., Ibrahim, E. A., De Vol, E. B. et al. (2002). Fluconazole for the treatment of cutaneous leishmaniasis caused by Leishmania major. New England Journal of Medicine 346, Soto, J., Toledo, J., Gutierrez, P. et al. (2001). Treatment of American cutaneous leishmaniasis with miltefosine, an oral agent. Clinical Infectious Diseases 33, E Saenz, R. E., Paz, H. M., Johnson, C. M. et al. (1989). Treatment of American cutaneous leishmaniasis with orally administered allopurinol riboside. Journal of Infectious Diseases 160, Guderian, R. H., Chico, M. E., Rogers, M. D. et al. (1991). Placebo controlled treatment of Ecuadorian cutaneous leishmaniasis. American Journal of Tropical Medicine and Hygiene 45, Esfandiarpour, I. & Alavi, A. (2002). Evaluating the efficacy of allopurinol and meglumine antimoniate (Glucantime) in the treatment of cutaneous leishmaniasis. International Journal of Dermatology 41, D Oliveira, J. A., Machado, P. R. & Carvalho, E. M. (1997). Evaluating the efficacy of allopurinol for the treatment of cutaneous leishmaniasis. International Journal of Dermatology 36, Martinez, S. & Marr, J. J. (1992). Allopurinol in the treatment of American cutaneous leishmaniasis. New England Journal of Medicine 326, Martinez, S., Gonzalez, M. & Vernaza, M. E. (1997). Treatment of cutaneous leishmaniasis with allopurinol and stibogluconate. Clinical Infectious Diseases 24, Kochar, D. K., Aseri, S., Sharma, B. V. et al. (2000). The role of rifampicin in the management of cutaneous leishmaniasis. Quarterly Journal of Medicine 93, Dogra, J. (1991). A double-blind study on the efficacy of oral dapsone in cutaneous leishmaniasis. Transactions of the Royal Society of Tropical Medicine and Hygiene 85, Sharquie, K. E., Najim, R. A., Farjou, I. B. et al. (2001). Oral zinc sulphate in the treatment of acute cutaneous leishmaniasis. Clinical and Experimental Dermatology 26, Bassiouny, A., El Meshad, M., Talaat, M. et al. (1982). Cryosurgery in cutaneous leishmaniasis. British Journal of Dermatology 107, Leibovici, V. & Aram, H. (1986). Cryotherapy in acute cutaneous leishmaniasis. International Journal of Dermatology 25, al Majali, O., Routh, H. B., Abuloham, O. et al. (1997). A 2-year study of liquid nitrogen therapy in cutaneous leishmaniasis. International Journal of Dermatology 36, Gurei, M. S., Tatli, N., Ozbilge, H. et al. (2000). Efficacy of cryotherapy and intralesional pentostam in treatment of cutaneous leishmaniasis. Journal of the Egyptian Society of Parasitology 30, Junaid, A. J. (1986). Treatment of cutaneous leishmaniasis with infrared heat. International Journal of Dermatology 25, Bryceson, A. D., Murphy, A. & Moody, A. H. (1994). Treatment of Old World cutaneous leishmaniasis with aminosidine ointment: results of an open study in London. Transactions of the Royal Society of Tropical Medicine and Hygiene 88, Arana, B. A., Mendoza, C. E., Rizzo, N. R. et al. (2001). Randomized, controlled, double-blind trial of topical treatment of cutaneous leishmaniasis with paromomycin plus methylbenzethonium chloride ointment in Guatemala. American Journal of Tropical Medicine and Hygiene 65, Krause, G. & Kroeger, A. (1994). Topical treatment of American cutaneous leishmaniasis with paramomycin and methylbenzethonium chloride: a clinical study under field conditions in Ecuador. Transactions of the Royal Society of Tropical Medicine and Hygiene 88, Munir, M., Mohammed, K. & Babkerhyl, M. (2002). Guidelines for the Treatment and Prevention of Cutaneous Leishmaniasis in Pakistan. Ministry of Health Pakistan; WHO, Health Net International. 50. Berman, J. D. (1997). Human leishmaniasis: clinical, diagnostic, and chemotherapeutic developments in the last 10 years. Clinical Infectious Diseases 24, el On, J., Cawich, F., Evans, D. A. et al. (1993). Topical treatment of cutaneous leishmaniasis in Belize: in vitro and in vivo studies with Leishmania mexicana. International Journal for Parasitology 23, Weinrauch, L., Cawich, F., Craig, P. et al. (1993). Topical treatment of New World cutaneous leishmaniasis in Belize: a clinical study. Journal of the American Academy of Dermatology 29, Navin, T. R., Arana, B. A., Arana, F. E. et al. (1992). Placebocontrolled clinical trial of sodium stibogluconate (Pentostam) versus ketoconazole for treating cutaneous leishmaniasis in Guatemala. Journal of Infectious Diseases 165, Saenz, R. E., Paz, H. & Berman, J. D. (1990). Efficacy of ketoconazole against Leishmania braziliensis panamensis cutaneous leishmaniasis. American Journal of Medicine 89, Soto, J., Fuya, P., Herrera, R. et al. (1998). Topical paromomycin/ methylbenzethonium chloride plus parenteral meglumine antimonate as treatment for American cutaneous leishmaniasis: controlled study. Clinical Infectious Diseases 26, Llanos-Cuentas, A., Echevarria, J., Cruz, M. et al. (1997). Efficacy of sodium stibogluconate alone and in combination with allopurinol for treatment of mucocutaneous leishmaniasis. Clinical Infectious Diseases 25, Romero, G. A., Guerra, M. V., Paes, M. G. et al. (2001). Comparison of cutaneous leishmaniasis due to Leishmania (Viannia) braziliensis and L. (V.) guyanensis in Brazil: therapeutic response to meglumine antimoniate. American Journal of Tropical Medicine and Hygiene 65, Herwaldt, B. L. (1999). Leishmaniasis. Lancet 354, Almeida, R., D Oliveira, A., Jr, Machado, P. et al. (1999). Randomized, double-blind study of stibogluconate plus human granulocyte macrophage colony-stimulating factor versus stibogluconate alone in the treatment of cutaneous leishmaniasis. Journal of Infectious Diseases 180, Navin, T. R., Arana, B. A., Arana, F. E. et al. (1990). Placebocontrolled clinical trial of meglumine antimonate (glucantime) vs. localized controlled heat in the treatment of cutaneous leishmaniasis in Guatemala. American Journal of Tropical Medicine and Hygiene 42, el On, J., Halevy, S., Grunwald, M. H. et al. (1992). Topical treatment of Old World cutaneous leishmaniasis caused by Leishmania major: a double-blind control study. Journal of the American Academy of Dermatology 27, el On, J., Livshin, R., Paz, Z. E. et al. (1985). Topical treatment of cutaneous leishmaniasis. Journal of Investigative Dermatology 291,
9 63. Asilian, A., Jalayer, T., Whitworth, J. A. et al. (1995). A randomized, placebo-controlled trial of a two-week regimen of aminosidine (paromomycin) ointment for treatment of cutaneous leishmaniasis in Iran. American Journal of Tropical Medicine and Hygiene 53, Ben Salah, A., Zakraoui, H., Zaatour, A. et al. (1995). A randomized, placebo-controlled trial in Tunisia treating cutaneous leishmaniasis with paromomycin ointment. American Journal of Tropical Medicine and Hygiene 53, Alkhawajah, A. M., Larbi, E., al Gindan, Y. et al. (1997). Treatment of cutaneous leishmaniasis with antimony: intramuscular versus intralesional administration. Annals of Tropical Medicine and Parasitology 91, Faris, R. M., Jarallah, J. S., Khoja, T. A. et al. (1993). Intralesional treatment of cutaneous leishmaniasis with sodium stibogluconate antimony. International Journal of Dermatology 32, Tallab, T. M., Bahamdam, K. A., Mirdad, S. et al. (1996). Cutaneous leishmaniasis: schedules for intralesional treatment with sodium stibogluconate. International Journal of Dermatology 35, Weinrauch, L., Livshin, R. & el On, J. (1987). Ketoconazole in cutaneous leishmaniasis. British Journal of Dermatology 117, Weinrauch, L., Livshin, R., Even-Paz, Z. et al. (1983). Efficacy of ketoconazole in cutaneous leishmaniasis. Archives of Dermatological Research 275, Anders, G., Eisenberger, C. L., Jonas, F. et al. (2002). Distinguishing Leishmania tropica and Leishmania major in the Middle East using the polymerase chain reaction with kinetoplast DNA-specific primers. Transactions of the Royal Society of Tropical Medicine and Hygiene 96, Suppl. 1, S Singh, S., Singh, R. & Sundar, S. (1995). Failure of ketoconazole in oriental sore in India. Journal of Chemotherapy 7, Suppl. 4, Singh,S., Singh, R. & Sundar, S. (1995). Failure of ketoconazole treatment in cutaneous leishmaniasis. International Journal of Dermatology 34, Del Giudice, P., Marty, P., Lacour, J. P. et al. (1998). Cutaneous leishmaniasis due to Leishmania infantum. Case reports and literature review. Archives of Dermatology 134, Scope, A., Trau, H., Andreas, G. et al. (2003). Experience with New World cutaneous leishmaniasis in Israeli travelers. Journal of the American Academy of Dermatology 49, Schubach, A., Haddad, F., Oliveira-Neto, M. P. et al. (1998). Detection of Leishmania DNA by polymerase chain reaction in scars of treated human patients. Journal of Infectious Diseases 178, Antezana, G., Zeballos, R., Mendoza, C. et al. (1992). Electrocardiographic alterations during treatment of mucocutaneous leishmaniasis with meglumine antimoniate and allopurinol. Transactions of the Royal Society of Tropical Medicine and Hygiene 86, Ribeiro, A. L., Drummond, J. B., Volpini, A. C. et al. (1999). Electrocardiographic changes during low-dose, short-term therapy of cutaneous leishmaniasis with the pentavalent antimonial meglumine. Brazilian Journal of Medical and Biological Research 32, Hepburn, N. C., Siddique, I., Howie, A. F. et al. (1993). Hepatotoxicity of sodium stibogluconate in leishmaniasis. Lancet 342, Hepburn, N. C. (1993). Thrombocytopenia complicating sodium stibogluconate therapy for cutaneous leishmaniasis. Transactions of the Royal Society of Tropical Medicine and Hygiene 87, Gasser, R. A., Jr, Magill, A. J., Oster, C. N. et al. (1994). Pancreatitis induced by pentavalent antimonial agents during treatment of leishmaniasis. Clinical Infectious Diseases 18, Rodrigues, M. L., Costa, R. S., Souza, C. S. et al. (1999). Nephrotoxicity attributed to meglumine antimoniate (Glucantime) in the treatment of generalized cutaneous leishmaniasis. Revista do Instituto de Medicina Tropical de Sao Paulo 41, Brummitt, C. F., Porter, J. A. & Herwaldt, B. L. (1996). Reversible peripheral neuropathy associated with sodium stibogluconate therapy for American cutaneous leishmaniasis. Clinical Infectious Diseases 22, Wortmann, G. W., Aronson, N. E., Byrd, J. C. et al. (1998). Herpes zoster and lymphopenia associated with sodium stibogluconate therapy for cutaneous leishmaniasis. Clinical Infectious Diseases 27, Hellier, I., Dereure, O., Tournillac, I. et al. (2000). Treatment of Old World cutaneous leishmaniasis by pentamidine isethionate. An open study of 11 patients. Dermatology 200,
LeishMan Recommendations for Treatment of Cutaneous and Mucosal Leishmaniasis in Travelers, 2014
 I S T M 116 REVIEW LeishMan Recommendations for Treatment of Cutaneous and Mucosal Leishmaniasis in Travelers, 2014 Johannes Blum, MD, Pierre Buffet, MD, Leo Visser, MD, # Gundel Harms, MD, Mark S. Bailey,
I S T M 116 REVIEW LeishMan Recommendations for Treatment of Cutaneous and Mucosal Leishmaniasis in Travelers, 2014 Johannes Blum, MD, Pierre Buffet, MD, Leo Visser, MD, # Gundel Harms, MD, Mark S. Bailey,
Cutaneous Leishmaniasis in a Chinese man returning from the Amazon forest in Brazil
 Hong Kong J. Dermatol. Venereol. (2013) 21, 79-83 Case Report Cutaneous Leishmaniasis in a Chinese man returning from the Amazon forest in Brazil JTHT Yu and SY Wong A 60-year-old man presented with ulcerated
Hong Kong J. Dermatol. Venereol. (2013) 21, 79-83 Case Report Cutaneous Leishmaniasis in a Chinese man returning from the Amazon forest in Brazil JTHT Yu and SY Wong A 60-year-old man presented with ulcerated
Acta Dermatovenerol Croat 2008;16(2):60-64 CLINICAL ARTICLE
 2008;16(2):60-64 CLINICAL ARTICLE Clinical Efficacy of Intramuscular Meglumine Antimoniate Alone and in Combination with Intralesional Meglumine Antimoniate in the Treatment of Old World Cutaneous Leishmaniasis
2008;16(2):60-64 CLINICAL ARTICLE Clinical Efficacy of Intramuscular Meglumine Antimoniate Alone and in Combination with Intralesional Meglumine Antimoniate in the Treatment of Old World Cutaneous Leishmaniasis
data indicating a 31% efficacy in northwest Colombia. 6
 Am. J. Trop. Med. Hyg., 64(3, 4), 2001, pp. 187 193 Copyright 2001 by The American Society of Tropical Medicine and Hygiene TREATMENT FAILURE IN CHILDREN IN A RANDOMIZED CLINICAL TRIAL WITH 10 AND 20 DAYS
Am. J. Trop. Med. Hyg., 64(3, 4), 2001, pp. 187 193 Copyright 2001 by The American Society of Tropical Medicine and Hygiene TREATMENT FAILURE IN CHILDREN IN A RANDOMIZED CLINICAL TRIAL WITH 10 AND 20 DAYS
Impavido. (miltefosine) New Product Slideshow
 Impavido (miltefosine) New Product Slideshow Introduction Brand name: Impavido Generic name: Miltefosine Pharmacological class: Antileishmanial agent Strength and Formulation: 50mg; hard gel capsules Manufacturer:
Impavido (miltefosine) New Product Slideshow Introduction Brand name: Impavido Generic name: Miltefosine Pharmacological class: Antileishmanial agent Strength and Formulation: 50mg; hard gel capsules Manufacturer:
Miltefosine for New World Cutaneous Leishmaniasis
 MAJOR ARTICLE for New World Cutaneous sis J. Soto, 1 B. A. Arana, 4 J. Toledo, 1 N. Rizzo, 4 J. C. Vega, 2 A. Diaz, 4 M. Luz, 1 P. Gutierrez, 1 M. Arboleda, 3 J. D. Berman, 6,a K. Junge, 5 J. Engel, 5
MAJOR ARTICLE for New World Cutaneous sis J. Soto, 1 B. A. Arana, 4 J. Toledo, 1 N. Rizzo, 4 J. C. Vega, 2 A. Diaz, 4 M. Luz, 1 P. Gutierrez, 1 M. Arboleda, 3 J. D. Berman, 6,a K. Junge, 5 J. Engel, 5
Oral miltefosine for Indian post-kala-azar dermal leishmaniasis: a randomised trial
 Tropical Medicine and International Health doi:10.1111/tmi.12015 volume 18 no 1 pp 96 100 january 2013 Oral miltefosine for Indian post-kala-azar dermal leishmaniasis: a randomised trial Shyam Sundar 1,
Tropical Medicine and International Health doi:10.1111/tmi.12015 volume 18 no 1 pp 96 100 january 2013 Oral miltefosine for Indian post-kala-azar dermal leishmaniasis: a randomised trial Shyam Sundar 1,
HAEMOFLAGELLATES. Dr. Anuluck Junkum Department of Parasitology Faculty of Medicine
 HAEMOFLAGELLATES Dr. Anuluck Junkum Department of Parasitology Faculty of Medicine Objective Can describe the morphology, life cycle, pathology, diagnosis and prevention of Leishmania spp. and Trypanosoma
HAEMOFLAGELLATES Dr. Anuluck Junkum Department of Parasitology Faculty of Medicine Objective Can describe the morphology, life cycle, pathology, diagnosis and prevention of Leishmania spp. and Trypanosoma
Sodium Stibogluconate treatment for cutaneous leishmaniasis: A clinical study of 43 cases from the north of Jordan
 Sodium Stibogluconate treatment for cutaneous leishmaniasis: A clinical study of 43 cases from the north of Jordan Mamoun Mohammad Al-Athamneh Hiathem Qasem Abu Al-haija Ra ed Smadi Ayman S. Qaqaa Heba
Sodium Stibogluconate treatment for cutaneous leishmaniasis: A clinical study of 43 cases from the north of Jordan Mamoun Mohammad Al-Athamneh Hiathem Qasem Abu Al-haija Ra ed Smadi Ayman S. Qaqaa Heba
Therapeutic Options for Visceral Leishmaniasis
 Drugs (2013) 73:1863 1888 DOI 10.1007/s40265-013-0133-0 REVIEW ARTICLE Therapeutic Options for Visceral Leishmaniasis Begoña Monge-Maillo Rogelio López-Vélez Published online: 30 October 2013 Ó The Author(s)
Drugs (2013) 73:1863 1888 DOI 10.1007/s40265-013-0133-0 REVIEW ARTICLE Therapeutic Options for Visceral Leishmaniasis Begoña Monge-Maillo Rogelio López-Vélez Published online: 30 October 2013 Ó The Author(s)
Treatment of Cutaneous Leishmaniasis with Allopurinol and Stibogluconate
 165 Treatment of Cutaneous Leishmaniasis with Allopurinol and S. Martinez, M. Gonzalez, and M. E. Vernaza From the Department of Internal Medicine, University of Cauca, Popayan, and the University Hospital
165 Treatment of Cutaneous Leishmaniasis with Allopurinol and S. Martinez, M. Gonzalez, and M. E. Vernaza From the Department of Internal Medicine, University of Cauca, Popayan, and the University Hospital
World Health Organization Department of Communicable Disease Surveillance and Response
 WHO Report on Global Surveillance of Epidemic-prone Infectious Diseases World Health Organization Department of Communicable Disease Surveillance and Response This document has been downloaded from the
WHO Report on Global Surveillance of Epidemic-prone Infectious Diseases World Health Organization Department of Communicable Disease Surveillance and Response This document has been downloaded from the
Intralesional meglumine antimoniate for the treatment of localised cutaneous leishmaniasis: a retrospective review of a Brazilian referral centre
 512 Mem Inst Oswaldo Cruz, Rio de Janeiro, Vol. 111(8): 512-516, August 2016 Intralesional meglumine antimoniate for the treatment of localised cutaneous leishmaniasis: a retrospective review of a Brazilian
512 Mem Inst Oswaldo Cruz, Rio de Janeiro, Vol. 111(8): 512-516, August 2016 Intralesional meglumine antimoniate for the treatment of localised cutaneous leishmaniasis: a retrospective review of a Brazilian
Original Article Chloroquine in cutaneous leishmaniasis
 Original Article Chloroquine in cutaneous leishmaniasis Ikramullah Khan, Rifat Yasmin, Iqbal Sidiqui Department of Dermatology, Pakistan Institute of Medical Sciences, Islamabad Abstract Background Chloroquine
Original Article Chloroquine in cutaneous leishmaniasis Ikramullah Khan, Rifat Yasmin, Iqbal Sidiqui Department of Dermatology, Pakistan Institute of Medical Sciences, Islamabad Abstract Background Chloroquine
Cutaneous Leishmaniasis : Global overview
 Cutaneous Leishmaniasis : Global overview Meeting of stakeholders for selected Health R&D Demostration Projects, 7-10 May 2014, WHO, Geneva Dr. Daniel Argaw Dagne, NTD/WHO CSR - DDC AFRO Leishmaniasis
Cutaneous Leishmaniasis : Global overview Meeting of stakeholders for selected Health R&D Demostration Projects, 7-10 May 2014, WHO, Geneva Dr. Daniel Argaw Dagne, NTD/WHO CSR - DDC AFRO Leishmaniasis
Leishmaniasis. CDR R.L. Gutierrez Oct 2014
 Leishmaniasis CDR R.L. Gutierrez Oct 2014 Overview Protozoan parasite(s) of tissue and WBCs Many species / Many Syndromes (Cutaneous / Visceral) Pathogen: Location - Old World vs. New World Host: Immune
Leishmaniasis CDR R.L. Gutierrez Oct 2014 Overview Protozoan parasite(s) of tissue and WBCs Many species / Many Syndromes (Cutaneous / Visceral) Pathogen: Location - Old World vs. New World Host: Immune
Leishmaniasis WRAIR- GEIS 'Operational Clinical Infectious Disease' Course
 Leishmaniasis WRAIR- GEIS 'Operational Clinical Infectious Disease' Course UNCLASSIFIED Acknowledgments LTC James E. Moon, MD Chief, Sleep Trials Branch Walter Reed Army Institute of Research CDR Ramiro
Leishmaniasis WRAIR- GEIS 'Operational Clinical Infectious Disease' Course UNCLASSIFIED Acknowledgments LTC James E. Moon, MD Chief, Sleep Trials Branch Walter Reed Army Institute of Research CDR Ramiro
Cutaneous leishmaniasis: advances in disease pathogenesis, diagnostics and therapeutics
 Clinical dermatology Review article CED Clinical and Experimental Dermatology Cutaneous leishmaniasis: advances in disease pathogenesis, diagnostics and therapeutics M. Ameen Royal Free Hospital, Royal
Clinical dermatology Review article CED Clinical and Experimental Dermatology Cutaneous leishmaniasis: advances in disease pathogenesis, diagnostics and therapeutics M. Ameen Royal Free Hospital, Royal
COMPARISON OF MEGLUMINE ANTIMONIATE AND PENTAMIDINE FOR PERUVIAN CUTANEOUS LEISHMANIASIS
 Am. J. Trop. Med. Hyg., 72(2), 2005, pp. 133 137 Copyright 2005 by The American Society of Tropical Medicine and Hygiene COMPARISON OF MEGLUMINE ANTIMONIATE AND PENTAMIDINE FOR PERUVIAN CUTANEOUS LEISHMANIASIS
Am. J. Trop. Med. Hyg., 72(2), 2005, pp. 133 137 Copyright 2005 by The American Society of Tropical Medicine and Hygiene COMPARISON OF MEGLUMINE ANTIMONIATE AND PENTAMIDINE FOR PERUVIAN CUTANEOUS LEISHMANIASIS
Welcome to the Jungle! Dr Aileen Oon, 2017 Microbiology Registrar
 Welcome to the Jungle! Dr Aileen Oon, 2017 Microbiology Registrar AA 55M presented with sores on left olecranon and umbilical area Umbilical sores present for 3 weeks Left olecranon lesions for 1 week
Welcome to the Jungle! Dr Aileen Oon, 2017 Microbiology Registrar AA 55M presented with sores on left olecranon and umbilical area Umbilical sores present for 3 weeks Left olecranon lesions for 1 week
Leishmaniasis, Kala Azar(The Black Fever)
 Leishmaniasis, Kala Azar(The Black Fever) By Lawrence Hall Etiologic agent Protist obligate intracellular parasite, Transmission Vectors Phylum: Euglenozoa (genus Leishmania) Over 21 species that infect
Leishmaniasis, Kala Azar(The Black Fever) By Lawrence Hall Etiologic agent Protist obligate intracellular parasite, Transmission Vectors Phylum: Euglenozoa (genus Leishmania) Over 21 species that infect
Leishmaniasis. MAJ Kris Paolino September 2014
 Leishmaniasis MAJ Kris Paolino September 2014 Thanks to COL (Ret) Kent Kester MAJ Leyi Lin http://www.niaid.nih.gov/topics/leishmaniasis History Sir William Boog Leishman (1865-1926) Matriculated at the
Leishmaniasis MAJ Kris Paolino September 2014 Thanks to COL (Ret) Kent Kester MAJ Leyi Lin http://www.niaid.nih.gov/topics/leishmaniasis History Sir William Boog Leishman (1865-1926) Matriculated at the
Recommendations for Coping with Leishmaniasis: A Review of Control Strategies. Centro de Convenções de Reboças Red Room 17: 00h
 Recommendations for Coping with Leishmaniasis: A Review of Control Strategies Centro de Convenções de Reboças 08.04.2014 Red Room 17: 00h Leishmaniasis - a Global Problem Visceral 2012 300 000 cases 20,000
Recommendations for Coping with Leishmaniasis: A Review of Control Strategies Centro de Convenções de Reboças 08.04.2014 Red Room 17: 00h Leishmaniasis - a Global Problem Visceral 2012 300 000 cases 20,000
SUMMARY. Cutaneous leishmaniasis with only skin involvement: single to multiple skin ulcers, satellite lesions and nodular lymphangitis.
 SUMMARY Leishmaniasis is a disease affecting predominantly people in the developing countries; 350 million people worldwide are at risk and yearly more than 2 million new cases occur. Leishmaniasis is
SUMMARY Leishmaniasis is a disease affecting predominantly people in the developing countries; 350 million people worldwide are at risk and yearly more than 2 million new cases occur. Leishmaniasis is
Summary of Cases & Epidemiology Aspects of Leishmaniasis in Thailand
 Summary of Cases & Epidemiology Aspects of Leishmaniasis in Thailand Sukmee T. 1, Mungthin M. 2, Apiwathanasorn C. 3, Leelayoova S. 2 1 Department of Microbiology, Phramongkutklao College of Medicine 2
Summary of Cases & Epidemiology Aspects of Leishmaniasis in Thailand Sukmee T. 1, Mungthin M. 2, Apiwathanasorn C. 3, Leelayoova S. 2 1 Department of Microbiology, Phramongkutklao College of Medicine 2
ISPUB.COM. Cutaneous Leishmaniasis in Iran. C Yazdi, M Narmani, B Sadri INTRODUCTION CASE 1 CASE 2
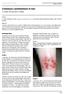 ISPUB.COM The Internet Journal of Infectious Diseases Volume 3 Number 1 Cutaneous Leishmaniasis in Iran C Yazdi, M Narmani, B Sadri Citation C Yazdi, M Narmani, B Sadri. Cutaneous Leishmaniasis in Iran.
ISPUB.COM The Internet Journal of Infectious Diseases Volume 3 Number 1 Cutaneous Leishmaniasis in Iran C Yazdi, M Narmani, B Sadri Citation C Yazdi, M Narmani, B Sadri. Cutaneous Leishmaniasis in Iran.
CONTROLE DAS LEISHMANIOSES O QUE FALTA FAZER? Centro de Convenções de Reboças Red Room 17: 00h
 CONTROLE DAS LEISHMANIOSES O QUE FALTA FAZER? Centro de Convenções de Reboças 08.04.2014 Red Room 17: 00h Leishmaniasis - a Global Problem Visceral 2012 300 000 cases 20,000 deaths (6.7%) 310 million at
CONTROLE DAS LEISHMANIOSES O QUE FALTA FAZER? Centro de Convenções de Reboças 08.04.2014 Red Room 17: 00h Leishmaniasis - a Global Problem Visceral 2012 300 000 cases 20,000 deaths (6.7%) 310 million at
Research Article Efficacy and Safety of Paromomycin in Treatment of Post-Kala-Azar Dermal Leishmaniasis
 ISRN Parasitology, Article ID 548010, 4 pages http://dx.doi.org/10.1155/2014/548010 Research Article Efficacy and Safety of Paromomycin in Treatment of Post-Kala-Azar Dermal Leishmaniasis Shyam Sundar,
ISRN Parasitology, Article ID 548010, 4 pages http://dx.doi.org/10.1155/2014/548010 Research Article Efficacy and Safety of Paromomycin in Treatment of Post-Kala-Azar Dermal Leishmaniasis Shyam Sundar,
Downloaded from:
 Wise, ES; Armstrong, MS; Watson, J; Lockwood, DN (2012) Monitoring toxicity associated with parenteral sodium stibogluconate in the day-case management of returned travellers with New World cutaneous leishmaniasis
Wise, ES; Armstrong, MS; Watson, J; Lockwood, DN (2012) Monitoring toxicity associated with parenteral sodium stibogluconate in the day-case management of returned travellers with New World cutaneous leishmaniasis
Leishmaniasis. By Joseph Knight, PA-C. 2. Explain the differences in the reasons leishmaniasis is spreading in Afghanistan and India.
 Leishmaniasis By Joseph Knight, PA-C Objectives 1. Identify the two types of leishmaniasis. 2. Explain the differences in the reasons leishmaniasis is spreading in Afghanistan and India. 3. Discuss how
Leishmaniasis By Joseph Knight, PA-C Objectives 1. Identify the two types of leishmaniasis. 2. Explain the differences in the reasons leishmaniasis is spreading in Afghanistan and India. 3. Discuss how
Control of leishmaniasis
 SIXTIETH WORLD HEALTH ASSEMBLY A60/10 Provisional agenda item 12.3 22 March 2007 Control of leishmaniasis Report by the Secretariat BACKGROUND 1. Leishmaniasis is endemic in 88 countries in the world and
SIXTIETH WORLD HEALTH ASSEMBLY A60/10 Provisional agenda item 12.3 22 March 2007 Control of leishmaniasis Report by the Secretariat BACKGROUND 1. Leishmaniasis is endemic in 88 countries in the world and
Short-Course Paromomycin Treatment of Visceral Leishmaniasis in India: 14-Day vs 21-Day Treatment
 MAJOR ARTICLE Short-Course Paromomycin Treatment of Visceral Leishmaniasis in India: 14-Day vs 21-Day Treatment Shyam Sundar, Neha Agrawal, Rakesh Arora, Dipti Agarwal, Madhukar Rai, and Jaya Chakravarty
MAJOR ARTICLE Short-Course Paromomycin Treatment of Visceral Leishmaniasis in India: 14-Day vs 21-Day Treatment Shyam Sundar, Neha Agrawal, Rakesh Arora, Dipti Agarwal, Madhukar Rai, and Jaya Chakravarty
Interleukin-10 and Interferon-γ Levels in Patients with Cutaneous Leishmaniasis Treated with Cryotherapy
 IJMS Vol 42, No 5, September 2017 Brief Report Interleukin-10 and Interferon-γ Levels in Patients with Cutaneous Leishmaniasis Treated with Cryotherapy Farhad Handjani 1,2, MD; Saeed Reza Yousef 1,2, MD;
IJMS Vol 42, No 5, September 2017 Brief Report Interleukin-10 and Interferon-γ Levels in Patients with Cutaneous Leishmaniasis Treated with Cryotherapy Farhad Handjani 1,2, MD; Saeed Reza Yousef 1,2, MD;
Authors Chappuis, François; Alirol, Emilie; Worku, Dagemlidet T; Mueller, Yolanda; Ritmeijer, Koert
 MSF Field Research High mortality among older patients treated with pentavalent antimonials for visceral leishmaniasis in East Africa and rationale for switch to liposomal amphotericin B Item type Article
MSF Field Research High mortality among older patients treated with pentavalent antimonials for visceral leishmaniasis in East Africa and rationale for switch to liposomal amphotericin B Item type Article
Current therapeutics for visceral leischmaniasis. F.Amri
 Current therapeutics for visceral leischmaniasis F.Amri Department of pediatrics Kairouan Intoduction The visceral leischmaniasis is an anthropozoonose caused by a protozoar, the Leishmania infantum The
Current therapeutics for visceral leischmaniasis F.Amri Department of pediatrics Kairouan Intoduction The visceral leischmaniasis is an anthropozoonose caused by a protozoar, the Leishmania infantum The
Antimonial Resistance & Combination therapy in Indian Visceral Leishmaniasis. Shyam Sundar Banaras Hindu University Varanasi, India
 Antimonial Resistance & Combination therapy in Indian Visceral Leishmaniasis Shyam Sundar Banaras Hindu University Varanasi, India History of the Current Epidemic in India & Antimony Resistance 6 Official
Antimonial Resistance & Combination therapy in Indian Visceral Leishmaniasis Shyam Sundar Banaras Hindu University Varanasi, India History of the Current Epidemic in India & Antimony Resistance 6 Official
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT 2 QUALITATIVE AND QUANTITATIVE COMPOSITION
 SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Pentostam Injection. 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Sodium Stibogluconate equivalent to 100 mg pentavalent antimony in each
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Pentostam Injection. 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Sodium Stibogluconate equivalent to 100 mg pentavalent antimony in each
Cutaneous leishmaniasis in an overseas Filipino worker who responded favorably to oral itraconazole
 CSE REPORT Cutaneous leishmaniasis in an overseas Filipino worker who responded favorably to oral itraconazole udi, MD 1, ilza ellamy R. Limcangco, MD, FPDS 2, Johannes F, Dayrit, MD, FPDS 2,3 Introduction:
CSE REPORT Cutaneous leishmaniasis in an overseas Filipino worker who responded favorably to oral itraconazole udi, MD 1, ilza ellamy R. Limcangco, MD, FPDS 2, Johannes F, Dayrit, MD, FPDS 2,3 Introduction:
EVALUATION OF MILTEFOSINE AGAINST LEISHMANIA MAJOR (MRHO/IR/75/ER): IN VITRO AND IN VIVO STUDIES
 ORIGINAL REPORT EVALUATION OF MILTEFOSINE AGAINST LEISHMANIA MAJOR (MRHO/IR/75/ER): IN VITRO AND IN VIVO STUDIES J. Esmaeili 1, M. Mohebali 1*, G. H. Edrissian 1, S. M. Rezayat 2, M. Ghazi-Khansari 2 and
ORIGINAL REPORT EVALUATION OF MILTEFOSINE AGAINST LEISHMANIA MAJOR (MRHO/IR/75/ER): IN VITRO AND IN VIVO STUDIES J. Esmaeili 1, M. Mohebali 1*, G. H. Edrissian 1, S. M. Rezayat 2, M. Ghazi-Khansari 2 and
Randomized, Double-Blinded, Phase 2 Trial of WR 279,396 (Paromomycin and Gentamicin) for Cutaneous Leishmaniasis in Panama
 Am. J. Trop. Med. Hyg., 89(3), 2013, pp. 557 563 doi:10.4269/ajtmh.12-0736 Copyright 2013 by The American Society of Tropical Medicine and Hygiene Randomized, Double-Blinded, Phase 2 Trial of WR 279,396
Am. J. Trop. Med. Hyg., 89(3), 2013, pp. 557 563 doi:10.4269/ajtmh.12-0736 Copyright 2013 by The American Society of Tropical Medicine and Hygiene Randomized, Double-Blinded, Phase 2 Trial of WR 279,396
Control of leishmaniasis
 EXECUTIVE BOARD EB118/4 118th Session 11 May 2006 Provisional agenda item 5.1 Control of leishmaniasis Report by the Secretariat BACKGROUND 1. Leishmaniasis is endemic in 88 countries in the world and
EXECUTIVE BOARD EB118/4 118th Session 11 May 2006 Provisional agenda item 5.1 Control of leishmaniasis Report by the Secretariat BACKGROUND 1. Leishmaniasis is endemic in 88 countries in the world and
Efficacy of pentavalent antimoniate intralesional infiltration therapy for cutaneous leishmaniasis: A systematic review
 COLLECTION REVIEW Efficacy of pentavalent antimoniate intralesional infiltration therapy for cutaneous leishmaniasis: A systematic review Nayara Castelano Brito, Ana Rabello, Gláucia Fernandes Cota* Pesquisas
COLLECTION REVIEW Efficacy of pentavalent antimoniate intralesional infiltration therapy for cutaneous leishmaniasis: A systematic review Nayara Castelano Brito, Ana Rabello, Gláucia Fernandes Cota* Pesquisas
Mucosal leishmaniasis: the experience of a Brazilian referral center
 Rev Soc Bras Med Trop 5(3):38-33, May-June, 08 doi: 0.590/0037-88-0478-07 Major Article Mucosal leishmaniasis: the experience of a Brazilian referral center Mariana Junqueira Pedras [],[], Janaína de Pina
Rev Soc Bras Med Trop 5(3):38-33, May-June, 08 doi: 0.590/0037-88-0478-07 Major Article Mucosal leishmaniasis: the experience of a Brazilian referral center Mariana Junqueira Pedras [],[], Janaína de Pina
Drug resistance in Indian visceral leishmaniasis
 Tropical Medicine and International Health volume6no11pp849±854november2001 Drug resistance in Indian visceral leishmaniasis Shyam Sundar Kala-azar Medical Research Centre, Banaras Hindu University, Varanasi,
Tropical Medicine and International Health volume6no11pp849±854november2001 Drug resistance in Indian visceral leishmaniasis Shyam Sundar Kala-azar Medical Research Centre, Banaras Hindu University, Varanasi,
CUTANEOUS LEISHMANIASIS
 CUTANEOUS LEISHMANIASIS Why are you neglecting me? A WHO initiative to control Cutaneous Leishmaniasis in selected Old World areas This document has been produced as the result of a WHO Informal Consultative
CUTANEOUS LEISHMANIASIS Why are you neglecting me? A WHO initiative to control Cutaneous Leishmaniasis in selected Old World areas This document has been produced as the result of a WHO Informal Consultative
WRAIR- GEIS 'OPERATIONAL CLINICAL INFECTIOUS DISEASE' COURSE
 Leishmaniasis WRAIR- GEIS 'OPERATIONAL CLINICAL INFECTIOUS DISEASE' COURSE The opinions or assertions contained herein are the private views of the author, and are not to be construed as official, or as
Leishmaniasis WRAIR- GEIS 'OPERATIONAL CLINICAL INFECTIOUS DISEASE' COURSE The opinions or assertions contained herein are the private views of the author, and are not to be construed as official, or as
WMLN Case Study. Necrotizing Palate Biopsy Specimen. Youngmi Kim Sr. Microbiologist TB Lab & Parasitology Lab MS, M(ASCP)
 WMLN Case Study Necrotizing Palate Biopsy Specimen Youngmi Kim Sr. Microbiologist TB Lab & Parasitology Lab MS, M(ASCP) 44 year old male from Honduras Immigrated to US 12 years ago to WI 4 years ago Seen
WMLN Case Study Necrotizing Palate Biopsy Specimen Youngmi Kim Sr. Microbiologist TB Lab & Parasitology Lab MS, M(ASCP) 44 year old male from Honduras Immigrated to US 12 years ago to WI 4 years ago Seen
Treatment of Disseminated Leishmaniasis With Liposomal Amphotericin B
 MAJOR ARTICLE Treatment of Disseminated Leishmaniasis With Liposomal Amphotericin B Paulo R. L. Machado, 1,2 Maria Elisa A. Rosa, 1 Luís H. Guimarães, 1,2 Fernanda V. O. Prates, 1 Adriano Queiroz, 1,2
MAJOR ARTICLE Treatment of Disseminated Leishmaniasis With Liposomal Amphotericin B Paulo R. L. Machado, 1,2 Maria Elisa A. Rosa, 1 Luís H. Guimarães, 1,2 Fernanda V. O. Prates, 1 Adriano Queiroz, 1,2
VISERAL LEISHMANIASI S (KALA-AZAR)
 VISERAL LEISHMANIASI S (KALA-AZAR) :OUTLINES DEFINITION. EPIDEMIOLOGY. PARASITE & VECTOR. PATHOLOGY CLINICAL & LIFE CYCLE. PICTURE. COMPLICATIONS. DIAGNOSIS. INVESTIGATIONS. MANAGEMENT TREATMENT S CONTROL.
VISERAL LEISHMANIASI S (KALA-AZAR) :OUTLINES DEFINITION. EPIDEMIOLOGY. PARASITE & VECTOR. PATHOLOGY CLINICAL & LIFE CYCLE. PICTURE. COMPLICATIONS. DIAGNOSIS. INVESTIGATIONS. MANAGEMENT TREATMENT S CONTROL.
CLINCOPATHOLOGICAL CASE
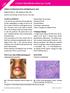 CLINCOPATHOLOGICAL CASE Solitary erythematous firm noduloplaque on nose Hassab El-Naby H., MD, Mosbeh Al Sadat, MD Department of Dermatology, Al-Azhar University, Cairo, Egypt. CLINICAL FINDINGS A 36-year-old
CLINCOPATHOLOGICAL CASE Solitary erythematous firm noduloplaque on nose Hassab El-Naby H., MD, Mosbeh Al Sadat, MD Department of Dermatology, Al-Azhar University, Cairo, Egypt. CLINICAL FINDINGS A 36-year-old
I nfectious diseases steal the headlines on a
 649 REVIEW Leishmaniasis Tonio V Piscopo, Charles Mallia Azzopardi... Epidemiology, disease patterns, immunology, diagnosis, treatment and control measures of are described. Various issues relating to
649 REVIEW Leishmaniasis Tonio V Piscopo, Charles Mallia Azzopardi... Epidemiology, disease patterns, immunology, diagnosis, treatment and control measures of are described. Various issues relating to
by author Drug Therapy in African Visceral Leishmaniasis 28th ECCMID Conference, Madrid, Spain 21 April 2018
 Drug Therapy in African Visceral Leishmaniasis 28th ECCMID Conference, Madrid, Spain 21 April 2018 Dr. Monique Wasunna, Director, DNDi Africa Regional Office Presentation Outline 1 2 4 5 Introduction leishmaniasis
Drug Therapy in African Visceral Leishmaniasis 28th ECCMID Conference, Madrid, Spain 21 April 2018 Dr. Monique Wasunna, Director, DNDi Africa Regional Office Presentation Outline 1 2 4 5 Introduction leishmaniasis
Kala-Azar- Treatment Update
 JOURNAL OF ADVANCES IN MEDICINE (JAM) DOI: 10.5958/2319-4324.2016.00002.X REVIEW PAPER Kala-Azar- Treatment Update Anup Singh Associate Professor, Department of Medicine, Institute of Medical Sciences,
JOURNAL OF ADVANCES IN MEDICINE (JAM) DOI: 10.5958/2319-4324.2016.00002.X REVIEW PAPER Kala-Azar- Treatment Update Anup Singh Associate Professor, Department of Medicine, Institute of Medical Sciences,
Leishmaniasis: A forgotten disease among neglected people
 ISPUB.COM The Internet Journal of Health Volume 11 Number 2 Leishmaniasis: A forgotten disease among neglected people Y Homsi, G Makdisi Citation Y Homsi, G Makdisi. Leishmaniasis: A forgotten disease
ISPUB.COM The Internet Journal of Health Volume 11 Number 2 Leishmaniasis: A forgotten disease among neglected people Y Homsi, G Makdisi Citation Y Homsi, G Makdisi. Leishmaniasis: A forgotten disease
BIO Parasitology Spring 2009
 BIO 475 - Parasitology Spring 2009 Stephen M. Shuster Northern Arizona University http://www4.nau.edu/isopod Lecture 5 Discovery of the Disease In 1924 the Kala-Azar Commission noted that the distribution
BIO 475 - Parasitology Spring 2009 Stephen M. Shuster Northern Arizona University http://www4.nau.edu/isopod Lecture 5 Discovery of the Disease In 1924 the Kala-Azar Commission noted that the distribution
Intralesional Infiltration with Meglumine Antimoniate for the Treatment of Leishmaniasis Recidiva Cutis in Ecuador
 In order to provide our readers with timely access to new content, papers accepted by the American Journal of Tropical Medicine and Hygiene are posted online ahead of print publication. Papers that have
In order to provide our readers with timely access to new content, papers accepted by the American Journal of Tropical Medicine and Hygiene are posted online ahead of print publication. Papers that have
Malaria prevention: source = The Sanford Guide to Antimicrobial Therapy. Gilbert et al.
 Malaria prevention: Agent Adult dose a Contraindications Application Notes Atovaquoneproguanil 250/100mg x1/day Insufficient data in pregnancy Prophylaxis for areas with CQ resistance, 2 expensive with
Malaria prevention: Agent Adult dose a Contraindications Application Notes Atovaquoneproguanil 250/100mg x1/day Insufficient data in pregnancy Prophylaxis for areas with CQ resistance, 2 expensive with
Leishmaniasis impact and treatment access
 REVIEW 10.1111/j.1469-0691.2011.03635.x Leishmaniasis impact and treatment access M. den Boer, D. Argaw, J. Jannin and J. Alvar Leishmaniasis Control Programme, WHO/IDM. Av. Appia, Geneva, Switzerland
REVIEW 10.1111/j.1469-0691.2011.03635.x Leishmaniasis impact and treatment access M. den Boer, D. Argaw, J. Jannin and J. Alvar Leishmaniasis Control Programme, WHO/IDM. Av. Appia, Geneva, Switzerland
References for Application for inclusion of paromomycin in the WHO Model List of Essential Medicines
 6 (a) Visceral leishmaniasis disease burden. Desjeux P. Leishmaniasis: current situation and new perspectives. Comp Immunol Microbiol Infect Dis 2004:27:305-18. Sundar S, Murray HW. Availability of miltefosine
6 (a) Visceral leishmaniasis disease burden. Desjeux P. Leishmaniasis: current situation and new perspectives. Comp Immunol Microbiol Infect Dis 2004:27:305-18. Sundar S, Murray HW. Availability of miltefosine
Leishmaniasis chemotherapy challenges and opportunities
 REVIEW 10.1111/j.1469-0691.2011.03630.x Leishmaniasis chemotherapy challenges and opportunities S. L. Croft 1 and P. Olliaro 2,3 1) Faculty of Infectious and Tropical Diseases, London School of Hygiene
REVIEW 10.1111/j.1469-0691.2011.03630.x Leishmaniasis chemotherapy challenges and opportunities S. L. Croft 1 and P. Olliaro 2,3 1) Faculty of Infectious and Tropical Diseases, London School of Hygiene
New insights on leishmaniasis in immunosuppressive conditions
 New insights on leishmaniasis in immunosuppressive conditions Javier Moreno Immunoparasitology Unit WHO Collaborative Center for Leishmaniasis Centro Nacional de Microbiología INSTITUTO DE SALUD CARLOS
New insights on leishmaniasis in immunosuppressive conditions Javier Moreno Immunoparasitology Unit WHO Collaborative Center for Leishmaniasis Centro Nacional de Microbiología INSTITUTO DE SALUD CARLOS
Neglected Diseases (NDs) Landscape in Brazil and South America
 Frontiers in Science on Neglected Diseases 13 th November 2014 Neglected Diseases (NDs) Landscape in Brazil and South America Jeffrey Shaw São Paulo University Biomedical Sciences Institute jeffreyj@usp.br
Frontiers in Science on Neglected Diseases 13 th November 2014 Neglected Diseases (NDs) Landscape in Brazil and South America Jeffrey Shaw São Paulo University Biomedical Sciences Institute jeffreyj@usp.br
Cutaneous Leishmaniasis in an Immigrant Saudi Worker: A Case Report
 J HEALTH POPUL NUTR 2014 Jun;32(2):372-376 ISSN 1606-0997 $ 5.00+0.20 INTERNATIONAL CENTRE FOR DIARRHOEAL DISEASE RESEARCH, BANGLADESH CASE STUDY Cutaneous Leishmaniasis in an Immigrant Saudi Worker: A
J HEALTH POPUL NUTR 2014 Jun;32(2):372-376 ISSN 1606-0997 $ 5.00+0.20 INTERNATIONAL CENTRE FOR DIARRHOEAL DISEASE RESEARCH, BANGLADESH CASE STUDY Cutaneous Leishmaniasis in an Immigrant Saudi Worker: A
~Trichinella Spiralis:
 Musculoskeletal System **Today we are going to talk about the parasites that affect the musculoskeletal system ~Trichinella Spiralis: It s a small nematode that measures to about 2-3mm in length. In general
Musculoskeletal System **Today we are going to talk about the parasites that affect the musculoskeletal system ~Trichinella Spiralis: It s a small nematode that measures to about 2-3mm in length. In general
Lupoid Cutaneous Leishmaniasis: a Case Report
 Dermatol Ther (2011) 1(2):36-41. DOI 10.1007/s13555-011-0008-9 CASE REPORT Lupoid Cutaneous Leishmaniasis: a Case Report Aida Khaled Samia Goucha Sonia Trabelsi Rachida Zermani Bécima Fazaa To view enhanced
Dermatol Ther (2011) 1(2):36-41. DOI 10.1007/s13555-011-0008-9 CASE REPORT Lupoid Cutaneous Leishmaniasis: a Case Report Aida Khaled Samia Goucha Sonia Trabelsi Rachida Zermani Bécima Fazaa To view enhanced
The Most Common Parasitic Infections In Yemen. Medical Parasitology
 The Most Common Parasitic Infections In Yemen Medical Parasitology ﻓﺎﯾز اﻟﺧوﻻﻧﻲ / د 2 : is a vector-borne disease that transmitted by sandflies and caused by obligate intracellular protozoa of the genus
The Most Common Parasitic Infections In Yemen Medical Parasitology ﻓﺎﯾز اﻟﺧوﻻﻧﻲ / د 2 : is a vector-borne disease that transmitted by sandflies and caused by obligate intracellular protozoa of the genus
PHYTOTHERAPY: AN INNOVATIVE PERSPECTIVE IN LEISHMANIASIS TREATMENT
 PHYTOTHERAPY: AN INNOVATIVE PERSPECTIVE IN LEISHMANIASIS TREATMENT LUIZ FILIPE GONÇALVES DE OLIVEIRA Fundação Oswaldo Cruz Instituto Oswaldo Cruz Laboratório de Biologia Molecular e Doenças Endêmicas Leishmaniases
PHYTOTHERAPY: AN INNOVATIVE PERSPECTIVE IN LEISHMANIASIS TREATMENT LUIZ FILIPE GONÇALVES DE OLIVEIRA Fundação Oswaldo Cruz Instituto Oswaldo Cruz Laboratório de Biologia Molecular e Doenças Endêmicas Leishmaniases
Case Report Development of Cutaneous Leishmaniasis after Leishmania Skin Test
 Case Reports in Medicine Volume 2011, Article ID 631079, 4 pages doi:10.1155/2011/631079 Case Report Development of Cutaneous Leishmaniasis after Leishmania Skin Test Paulo R. Machado, 1, 2 Augusto M.
Case Reports in Medicine Volume 2011, Article ID 631079, 4 pages doi:10.1155/2011/631079 Case Report Development of Cutaneous Leishmaniasis after Leishmania Skin Test Paulo R. Machado, 1, 2 Augusto M.
KYAMC Journal Vol. 3, No.-1, June Evaluation of Adverse Effects of Sodium Stibogluconate in the Treatment of Visceral Leishmaniasis
 Original Article Evaluation of Adverse Effects of Sodium Stibogluconate in the Treatment of Visceral Leishmaniasis Dr. Md. Abdus Sattar Miah 1, Dr. Md. Abdus Salam 2, Dr. Md. Azizul Haque 3, Dr. Md. Azizul
Original Article Evaluation of Adverse Effects of Sodium Stibogluconate in the Treatment of Visceral Leishmaniasis Dr. Md. Abdus Sattar Miah 1, Dr. Md. Abdus Salam 2, Dr. Md. Azizul Haque 3, Dr. Md. Azizul
Paclitaxel and Trastuzumab Breast Cancer
 Systemic Anti Cancer Treatment Protocol Paclitaxel and Trastuzumab Breast Cancer PROTOCOL REF: MPHAPTRBR (Version No: 1.0) Approved for use in: HER2 positive breast cancer. For adjuvant use in T1 or T2
Systemic Anti Cancer Treatment Protocol Paclitaxel and Trastuzumab Breast Cancer PROTOCOL REF: MPHAPTRBR (Version No: 1.0) Approved for use in: HER2 positive breast cancer. For adjuvant use in T1 or T2
Eastern Mediterranean Health Journal, Vol. 9, No. 4,
 Eastern Mediterranean Health Journal, Vol. 9, No. 4, 2003 837 Antimony-resistant Leishmania donovani in eastern Sudan: incidence and in vitro correlation M.G. Abdo, 1 W.M. Elamin, 1 E.A.G. Khalil 1 and
Eastern Mediterranean Health Journal, Vol. 9, No. 4, 2003 837 Antimony-resistant Leishmania donovani in eastern Sudan: incidence and in vitro correlation M.G. Abdo, 1 W.M. Elamin, 1 E.A.G. Khalil 1 and
A Mathematical model of cutaneous leishmaniasis
 Rochester Institute of Technology RIT Scholar Works Theses Thesis/Dissertation Collections 6-1-29 A Mathematical model of cutaneous leishmaniasis Karthik Bathena Follow this and additional works at: http://scholarworks.rit.edu/theses
Rochester Institute of Technology RIT Scholar Works Theses Thesis/Dissertation Collections 6-1-29 A Mathematical model of cutaneous leishmaniasis Karthik Bathena Follow this and additional works at: http://scholarworks.rit.edu/theses
Children with Visceral Leishmaniasis Presented to Omdurman Emergency Hospital for Children
 Original Article Children with Visceral Leishmaniasis Presented to Omdurman Emergency Hospital for Children Elfakey Walyeldinl, Ahmed Muawial, Mohamed Abdurrahman2 and Suwar M 03 Department of Paediatrics
Original Article Children with Visceral Leishmaniasis Presented to Omdurman Emergency Hospital for Children Elfakey Walyeldinl, Ahmed Muawial, Mohamed Abdurrahman2 and Suwar M 03 Department of Paediatrics
Application for Inclusion of MILTEFOSINE on WHO Model List of Essential Medicines:
 Application for Inclusion of MILTEFOSINE on WHO Model List of Essential Medicines: Comments re Feb 2011 reviews of the v 2010 application February 23, 2011 1. The application and its review In v 2010,
Application for Inclusion of MILTEFOSINE on WHO Model List of Essential Medicines: Comments re Feb 2011 reviews of the v 2010 application February 23, 2011 1. The application and its review In v 2010,
Single-Dose Liposomal Amphotericin B in the Treatment of Visceral Leishmaniasis in India: A Multicenter Study
 MAJOR ARTICLE Single-Dose Liposomal Amphotericin B in the Treatment of Visceral Leishmaniasis in India: A Multicenter Study S. Sundar, 1 T. K. Jha, 2 C. P. Thakur, 3 M. Mishra, 4 V. P. Singh, 1 and R.
MAJOR ARTICLE Single-Dose Liposomal Amphotericin B in the Treatment of Visceral Leishmaniasis in India: A Multicenter Study S. Sundar, 1 T. K. Jha, 2 C. P. Thakur, 3 M. Mishra, 4 V. P. Singh, 1 and R.
Iranian J Parasitol: Vol. 8, No.3, July -Sep 2013, pp Iranian J Parasitol. Open access Journal at ijpa.tums.ac.ir
 Iranian J Parasitol: Vol. 8, No.3, July -Sep 2013, pp.396-401 Iranian J Parasitol Tehran University of Medical Sciences Publication http:// tums.ac.ir Open access Journal at http:// ijpa.tums.ac.ir Iranian
Iranian J Parasitol: Vol. 8, No.3, July -Sep 2013, pp.396-401 Iranian J Parasitol Tehran University of Medical Sciences Publication http:// tums.ac.ir Open access Journal at http:// ijpa.tums.ac.ir Iranian
Case Report Localised Leishmaniasis of Oral Mucosa: Report of an Unusual Clinicopathological Entity
 Case Reports in Dentistry, Article ID 753149, 5 pages http://dx.doi.org/10.1155/2014/753149 Case Report Localised Leishmaniasis of Oral Mucosa: Report of an Unusual Clinicopathological Entity Deepak Passi,
Case Reports in Dentistry, Article ID 753149, 5 pages http://dx.doi.org/10.1155/2014/753149 Case Report Localised Leishmaniasis of Oral Mucosa: Report of an Unusual Clinicopathological Entity Deepak Passi,
Visceral Leishmaniasis combination therapies
 Best Science for the Most Neglected From patient needs to implementation of new treatments Visceral Leishmaniasis combination therapies Shyam Sundar Institute of Medical Sciences Banaras Hindu University
Best Science for the Most Neglected From patient needs to implementation of new treatments Visceral Leishmaniasis combination therapies Shyam Sundar Institute of Medical Sciences Banaras Hindu University
Viral Hepatitis. Background
 Viral Hepatitis Background Hepatitis or inflammation of the liver can be caused by infectious and noninfectious problems. Infectious etiologies include viruses, bacteria, fungi and parasites. Noninfectious
Viral Hepatitis Background Hepatitis or inflammation of the liver can be caused by infectious and noninfectious problems. Infectious etiologies include viruses, bacteria, fungi and parasites. Noninfectious
Product Information BROWN SNAKE ANTIVENOM AUST R 74897
 Product Information APPROVED NAME BROWN SNAKE ANTIVENOM AUST R 74897 DESCRIPTION BROWN SNAKE ANTIVENOM is prepared from the plasma of horses immunised with the venom of the brown snake (Pseudonaja textilis).
Product Information APPROVED NAME BROWN SNAKE ANTIVENOM AUST R 74897 DESCRIPTION BROWN SNAKE ANTIVENOM is prepared from the plasma of horses immunised with the venom of the brown snake (Pseudonaja textilis).
of America (IDSA) and the American Society of Tropical
 Clinical Infectious Diseases IDSA GUIDELINE Diagnosis and Treatment of Leishmaniasis: Clinical Practice Guidelines by the Infectious Diseases Society of America (IDSA) and the American Society of Tropical
Clinical Infectious Diseases IDSA GUIDELINE Diagnosis and Treatment of Leishmaniasis: Clinical Practice Guidelines by the Infectious Diseases Society of America (IDSA) and the American Society of Tropical
The feature of papers and citation analysis of eleven journals in tropical medicine indexed by Science Citation Index Expanded
 Mem Inst Oswaldo Cruz, Rio de Janeiro, Vol. 100(7): 805-810, November 2005 805 The feature of papers and citation analysis of eleven journals in tropical medicine indexed by Science Citation Index Expanded
Mem Inst Oswaldo Cruz, Rio de Janeiro, Vol. 100(7): 805-810, November 2005 805 The feature of papers and citation analysis of eleven journals in tropical medicine indexed by Science Citation Index Expanded
Post Travel Fever. Dr. Eyal Leshem. Center for Geographic Medicine Sheba Medical Center Tel Hashomer, Israel
 Post Travel Fever Dr. Eyal Leshem Center for Geographic Medicine Sheba Medical Center Tel Hashomer, Israel Introduction Fever in returned traveler: Trivial vs. life threatening infections Tropical and
Post Travel Fever Dr. Eyal Leshem Center for Geographic Medicine Sheba Medical Center Tel Hashomer, Israel Introduction Fever in returned traveler: Trivial vs. life threatening infections Tropical and
Leishmaniasis. caused by Leishmania spp. 利什曼原蟲 transmitted by Phlebotomine flies (Sandflies 白蛉 )
 Leishmaniasis caused by Leishmania spp. 利什曼原蟲 transmitted by Phlebotomine flies (Sandflies 白蛉 ) Cutaneous Leishmaniasis Mucocutaneous Leishmaniasis Visceral Leishmaniasis About 12 million people are currently
Leishmaniasis caused by Leishmania spp. 利什曼原蟲 transmitted by Phlebotomine flies (Sandflies 白蛉 ) Cutaneous Leishmaniasis Mucocutaneous Leishmaniasis Visceral Leishmaniasis About 12 million people are currently
Leishmaniasis. caused by Leishmania spp. 利什曼原蟲 transmitted by Phlebotomine flies (Sandflies 白蛉 ) 皮膚型黏膜型內臟型. Cutaneous Leishmaniasis
 Leishmaniasis caused by Leishmania spp. 利什曼原蟲 transmitted by Phlebotomine flies (Sandflies 白蛉 ) 皮膚型黏膜型內臟型 Cutaneous Leishmaniasis Mucocutaneous Leishmaniasis Visceral Leishmaniasis About 12 million people
Leishmaniasis caused by Leishmania spp. 利什曼原蟲 transmitted by Phlebotomine flies (Sandflies 白蛉 ) 皮膚型黏膜型內臟型 Cutaneous Leishmaniasis Mucocutaneous Leishmaniasis Visceral Leishmaniasis About 12 million people
Research Article Enhancement of a T H 1 Immune Response in Amphotericin B-Treated Mucocutaneous Leishmaniasis
 Biomedicine and Biotechnology Volume 27, Article ID 9641, 4 pages doi:1.1155/27/9641 Research Article Enhancement of a T H 1 Immune Response in Amphotericin B-Treated Mucocutaneous Leishmaniasis Washington
Biomedicine and Biotechnology Volume 27, Article ID 9641, 4 pages doi:1.1155/27/9641 Research Article Enhancement of a T H 1 Immune Response in Amphotericin B-Treated Mucocutaneous Leishmaniasis Washington
Departments of Dermatology, Venereology and Leprosy and *Microbiology, Indira Gandhi Medical College, Shimla, India.
 Net Study Comparative efþ cacy of intralesional sodium stibogluconate (SSG) alone and its combination with intramuscular SSG to treat localized cutaneous leishmaniasis: Results of a pilot study Ajeet K.
Net Study Comparative efþ cacy of intralesional sodium stibogluconate (SSG) alone and its combination with intramuscular SSG to treat localized cutaneous leishmaniasis: Results of a pilot study Ajeet K.
Topical Par o mo my cin with or without Gentamicin for Cutaneous Leish ma ni a sis
 T h e n e w e ngl a nd j o u r na l o f m e dic i n e original article Topical Par o mo my cin with or without Gentamicin for Cutaneous Leish ma ni a sis Afif Ben Salah, M.D., Nathalie Ben Messaoud, M.D.,
T h e n e w e ngl a nd j o u r na l o f m e dic i n e original article Topical Par o mo my cin with or without Gentamicin for Cutaneous Leish ma ni a sis Afif Ben Salah, M.D., Nathalie Ben Messaoud, M.D.,
Elimination of VL in the Indian subcontinent is it achievable?
 Elimination of VL in the Indian subcontinent is it achievable? P Das Rajendra Memorial Research Institute, Patna Jorge Alvar DNDi, Geneva Bhawna Sharma DNDi, India Leishmaniasis 350 million at risk worldwide
Elimination of VL in the Indian subcontinent is it achievable? P Das Rajendra Memorial Research Institute, Patna Jorge Alvar DNDi, Geneva Bhawna Sharma DNDi, India Leishmaniasis 350 million at risk worldwide
Nanosilver in the treatment of localized cutaneous leishmaniasis caused by Leishmania major (MRHO/IR/75/ER): an in vitro and in vivo study
 DARU Vol. 17, No. 4 2009 285 Nanosilver in the treatment of localized cutaneous leishmaniasis caused by Leishmania major (MRHO/IR/75/ER): an in vitro and in vivo study *1 Mohebali M., 2 Rezayat M.M., 3
DARU Vol. 17, No. 4 2009 285 Nanosilver in the treatment of localized cutaneous leishmaniasis caused by Leishmania major (MRHO/IR/75/ER): an in vitro and in vivo study *1 Mohebali M., 2 Rezayat M.M., 3
Phlebotomus argentipes. Amphotericin B has long been recognized as a powerful anti-kala-azar drug
 A STUDY OF EFFICACY AND ADVERSE DRUG REACTIONS OF CONVENTIONAL AMPHOTERICIN B IN THE TREATMENT OF KALA-AZAR Rajesh Kumar Pandey 1, S. N. Singh 2, Bharat Kumar 3, Kashif Shahnawaz 4 HOW TO CITE THIS ARTICLE:
A STUDY OF EFFICACY AND ADVERSE DRUG REACTIONS OF CONVENTIONAL AMPHOTERICIN B IN THE TREATMENT OF KALA-AZAR Rajesh Kumar Pandey 1, S. N. Singh 2, Bharat Kumar 3, Kashif Shahnawaz 4 HOW TO CITE THIS ARTICLE:
DTA3/COFUND Programme Research Project Proforma
 Page 1 General Information Project Code Partner University Faculty/School/Department/Research Centres First supervisor Please provide name and weblink Second supervisor Please provide name and weblink
Page 1 General Information Project Code Partner University Faculty/School/Department/Research Centres First supervisor Please provide name and weblink Second supervisor Please provide name and weblink
Fluconazole in the Treatment of Cutaneous Leishmaniasis Caused by Leishmania braziliensis: A Randomized Controlled Trial
 Clinical Infectious Diseases MAJOR ARTICLE Fluconazole in the Treatment of Cutaneous Leishmaniasis Caused by Leishmania braziliensis: A Randomized Controlled Trial Fernanda V. de O. Prates, 1 Mayra E.
Clinical Infectious Diseases MAJOR ARTICLE Fluconazole in the Treatment of Cutaneous Leishmaniasis Caused by Leishmania braziliensis: A Randomized Controlled Trial Fernanda V. de O. Prates, 1 Mayra E.
Azathioprine toxicity criteria and severity descriptors for the listing of biological agents for rheumatoid arthritis on the PBS
 Azathioprine toxicity criteria and severity descriptors for the listing of biological agents for rheumatoid arthritis on the PBS Only valid for adult patients Azathioprine must be at a dose of at least
Azathioprine toxicity criteria and severity descriptors for the listing of biological agents for rheumatoid arthritis on the PBS Only valid for adult patients Azathioprine must be at a dose of at least
Tropical medicine rounds
 Oxford, IJD International 0011-9059 Blackwell 41 UK Science, Journal Ltd 2002 of Dermatology Tropical medicine rounds Cutaneous André Báfica, Leishmaniasis Fabiano Oliveira, Luiz A. R. Freitas, Eliane
Oxford, IJD International 0011-9059 Blackwell 41 UK Science, Journal Ltd 2002 of Dermatology Tropical medicine rounds Cutaneous André Báfica, Leishmaniasis Fabiano Oliveira, Luiz A. R. Freitas, Eliane
Alberta Health Public Health Notifiable Disease Management Guidelines January 2013
 January 2013 Rickettsial Infections Revision Dates Case Definition Reporting Requirements Remainder of the Guideline (i.e., Etiology to References sections inclusive) January 2013 January 2013 December
January 2013 Rickettsial Infections Revision Dates Case Definition Reporting Requirements Remainder of the Guideline (i.e., Etiology to References sections inclusive) January 2013 January 2013 December
2.Trichomonas vaginalis
 2.Trichomonas vaginalis 1. Pathogenic to human &causes vaginitis (trichomoniasis). 2. troph. Is round or pear like in shape, contains 4-6 flagella, all originating from anterior end & only one extend posteriorly.
2.Trichomonas vaginalis 1. Pathogenic to human &causes vaginitis (trichomoniasis). 2. troph. Is round or pear like in shape, contains 4-6 flagella, all originating from anterior end & only one extend posteriorly.
NEW ZEALAND DATA SHEET
 NEW ZEALAND DATA SHEET 1.PRODUCT NAME PENTACARINAT 300mg Injection, powder 2.QUALITATIVE AND QUANTITATIVE COMPOSITION Pentamidine Isetionate B.P. 300mg 3.PHARMACEUTICAL FORM Powder for reconstitution.
NEW ZEALAND DATA SHEET 1.PRODUCT NAME PENTACARINAT 300mg Injection, powder 2.QUALITATIVE AND QUANTITATIVE COMPOSITION Pentamidine Isetionate B.P. 300mg 3.PHARMACEUTICAL FORM Powder for reconstitution.
Carboplatin / Gemcitabine Gynaecological Cancer
 Systemic Anti Cancer Treatment Protocol Carboplatin / Gemcitabine Gynaecological Cancer PROCTOCOL REF: MPHAGYNCAG (Version No: 1.0) Approved for use in: Recurrent/metastatic endometrial carcinoma Previously
Systemic Anti Cancer Treatment Protocol Carboplatin / Gemcitabine Gynaecological Cancer PROCTOCOL REF: MPHAGYNCAG (Version No: 1.0) Approved for use in: Recurrent/metastatic endometrial carcinoma Previously
