mdlecine, Universite de Montreal, Montreal, Quebec, Canada H3C 1C5
|
|
|
- Delilah Lester
- 5 years ago
- Views:
Transcription
1 J. Phy8iol. (1982), 322, pp With 1 text-figure Printed in Great Britain EXOCRINE PANCREATIC FUNCTION FOLLOWING PROXIMAL SMALL BOWEL RESECTION IN RATS BY MONIQUE D. GELINAS, CLAUDE L. MORIN AND JEAN MORISSET From the Centre de recherche pediatrique and the Detpartement de pediatrie, Service de Gastroenterologie, H6pital Sainte-Justine and Departement de nutrition, Faculte de mdlecine, Universite de Montreal, Montreal, Quebec, Canada H3C 1C5 (Received 8 May 198) SUMMARY 1. In order to assess if proximal enterectomy induces changes in the function of the exocrine pancreas, the exocrine pancreas was studied 1 week, 4 weeks, and 6 months after 5 or 75 % proximal small bowel resection. 2. One week after 5 and 75 % proximal small bowel resections, basal pancreatic bicarbonate outputs, studied by means of an external pancreatic fistula in conscious rats, were increased significantly over control values by 43 and 78 % respectively. Four weeks after a 75 % resection, the bicarbonate output was still significantly higher in resected animals than in sham operated animals. 3. The increase of volume and bicarbonate of the basal pancreatic secretion coincided with a 4-fold increase in plasma secretin concentration 1 week after resection. Both increased pancreatic secretion and plasma secretin concentration were transient. 4. The pancreatic hypersecretion was specifically reversed to control values with an i.p. injection of jejunoileal mucosa homogenate. 5. Serum gastrin and somatostatin values in intestinal mucosa and pancreas were not changed 1 and 4 weeks after enterectomy compared with sham operated animals. 6. The weight of the pancreas and its content of DNA were unaltered by resection. Amylase and chymotrypsinogen per gram pancreatic tissue and per Big DNA were reduced 4 weeks following resections as compared with sham operated rats. After 6 months, chymotrypsinogen appeared further reduced in resected animals. 7. It is concluded that extensive proximal enterectomy in rats produced early, transient and marked increases in basal pancreatic water and bicarbonate secretion and in plasma secretin due to the loss of jejunoileal inhibitorss, and a selective decrease in certain enzymes in pancreatic tissue. INTRODUCTION Proximal intestinal resection induced rapid morphological and functional changes in the gastrointestinal tract (Dowling& Booth, 1967; Nygaard, 1967; Hanson, Osborne & Sharp, 1977; Obertop, Nundy, Malamud & Malt, 1977). Gastric hypersecretion and hyperplasia are well known examples of rapid and transient changes occurring in man, rat and dog following proximal small bowel resection (Osborne, Frederick, Sizer,
2 72 M. D. GELINAS, C. L. MORIN AND J. MORISSET Blair, Cole & Thum, 1966; Caridis, Roberts & Smith, 1969; Seelig, Winborn & Weser, 1977). Gastric hypersecretion could lead to serious clinical problems and could contribute to ulcer formation and diarrhoea sometimes associated with subtotal enterectomy (Osborne et al. 1966; Fielding & Cooke, 197; Buxton, 1974; Weser, 1976). Very few studies have dealt with the influence of small bowel resection on the pancreas. Yet, most of the numerous gut hormones, primarily secretin and cholecystokinin-pancreozymin (CCK-PZ) exert some effects on the exocrine pancreas (Singh & Webster, 1978). It is therefore very likely that enterectomy affects the exocrine pancreas. Baxter (1931) observed a decrease in basal pancreatic secretion of rabbits on the day of 'total' enterectomy. Grossman (1958) noted a marked reduction in basal volume and protein concentration of pancreatic juice in rats two days after 'total' gut resection. Jackson & Linder (1953) reported reduced exocrine pancreatic function correlated with severe atrophy of the pancreas in a young man subjected to extensive small bowel resection 1 year before. However, Lundh & Oldby (1963) noted a normal level of trypsin in the intestinal content of one patient with massive enterectomy and gastric resection. Niessen, Brfigmann, Osswald, Schmidt & Flach (1974) described an increase in volume and bicarbonate of the stimulated pancreatic secretion in two children after subtotal small bowel resection. Similarly, Debas & Yamagishi (1977) reported, in an abstract, that excision of the jejunum and ileum increased the responsiveness of the exocrine pancreas of anaesthetized dogs to exogenous secretin and CCK-PZ, while basal pancreatic secretion was unaltered. However, the same group of researchers (Seal, Debas, Reynolds & Said, 1979) also reported, in another abstract, pancreatic hyposecretion to stimulation by liver extract meal in anaesthetized dogs following small bowel resection. Thus results in the literature span the whole spectrum from pancreatic hyposecretion to hypersecretion. Yet, none of the studies on the pancreas were performed at different time intervals after resection; none were done in rats after proximal enterectomy. The purpose of the present study was to evaluate if proximal small bowel resection done in rats is associated in time with changes of the exocrine pancreas. Changes in pancreatic secretion after intestinal resection are of particular interest because bicarbonate may have a protective role when gastric hypersecretion occurs, and because pancreaticobiliary secretions may be involved in the growth of intestinal mucosa in normal rats (Altmann, 1971) and in enterectomized rats (Weser, Heller & Tawil, 1977; Williamson, Bauer, Ross & Malt, 1978; Hughes, Bates & Dowling, 1978; Gelinas & Morin, 198). The present study suggests that proximal intestinal resection induces transient and marked basal hypersecretion of both plasma secretin and pancreatic bicarbonate outputs due to the loss of jejunoileal inhibitorss. Proximal resection also produces a selective decrease in digestive enzymes of the pancreatic tissue. METHODS Male Sprague-Dawley rats with an initial body weight of 15+-7g (mean+s.e.) were used throughout these experiments. Surgery was performed under pentobarbitone anaesthesia (4-8 mg/1 g body wt i.p.).
3 EXOCRINE PANCREAS AFTER ENTERECTOMY Intestinal surgery At the beginning of each experiment, the rats were subjected either to a sham resection (control groups) or to a proximal intestinal resection (experimental groups). In control rats, division of the jejunum with end-to-end resuture, was carried out at 6 cm distal to the duodenal jejunal junction. In experimental groups, the jejunum and the ileum were measured; removal of either 5 or 75 % of the measured jejunoileum started at 6cm distal to the duodenal jejunal junction with continuity re-established by an end-to-end anastomosis, as described by Lambert (1965). After intestinal surgery, the rats were individually housed and had free access to water and rat chow (Ralston Purina of Canada Ltd.) till the day of pancreatic fistulization or pancreas excision. Pancreatic fistulization In order to collect pure pancreatic juice from conscious rats, short-term external pancreatic fistulae, adapted from Grossman (1958), were installed 1 week, 4 weeks, or 6 months after intestinal surgery. Different groups of rats were used for each period as animals were killed after juice collection, and blood was collected for serum bilirubin analysis. Total serum bilirubin values were found to be within the normal range ( mg/i ml.), with a mean of 57 mg/1 ml. in control and resected animals. Through a mid line abdominal incision, a polyethylene catheter (PE 1 Intramedic, inner diameter -28 mm) was introduced into the uppermost part of the bile duct above a ligature and allowed to drain into the first part of the duodenum to maintain bile flow. A similar tube (25cm long) was pushed for 4mm into the pancreaticobiliary duct immediately above a ligature done at the ductus entry into the duodenum. This catheter was exteriorized through the base of the abdominal incision to collect the pancreatic juice. A silicone tube was introduced into the forestomach to hydrate the rats. The rats were then placed in a Bollman-type cage. A solution of 1% dextrose in -9 % NaCl plus 1 mm-kcl was pumped through the gastrostomy at 1-5 ml./hr till collection time. Measurements of pancreatic secretion Pancreatic juice was collected 2 hr after the fistula was constructed. Basal secretions were measured in microlitres by taking 1 hr collections into a 1 ml. graduated pipette placed 2 in. under the rat and inclined 5 from the horizontal. With a microgasometer (Natelson, 1951), basal bicarbonate concentrations were analysed immediately from the first hour sample collected under light mineral oil. A second lhr basal sample was frozen for protein determination (Lowry, Rosebrough, Farr & Randall, 1951), a convenient measure of enzyme output (De Waele, De Smul, Wissock & Kiekens, 1974; Schneeman & Lyman, 1977). The mean of three 1 hr periods was taken as the basal volume of secretion. Pancreatic secretion was stimulated exqgenously in two subgroups of rats. Forty Crick- Harper-Raper units (CHRU) of secretin and CCK-PZ (Boots Co.) per kg body weight were given within 1 min in two separate i.p. injections. i.p. injections of CCK-PZ have been demonstrated to elicit a response that is more predictable and of a greater magnitude than other routes of administration (Leroy, Morisset & Webster, 1971). Juice was collected for 1 hr immediately following the injections. In another set of experiments, an i.p. injection of jejunoileal mucosa homogenate was given to sham operated and 75 % resected rats. For each injection, mucosa was scraped off 45 cm of the intestine of a normal fasted rat starting 1 cm distal to the duodenal jejunal junction. The mucosa was weighed and homogenized in cold normal saline as described by Ruderman & Nagy (1974); 2-5 ml. of the homogenate (-8 g mucosa) was used for each injection. Pancreatic juice was collected for 2 hr thereafter. The mean of two 1 hr collections was taken to be the basal volume after homogenate injection. A separate control study was done on seven 75 % resected rats with extract of abdominal muscles diluted and injected in the same way as mucosal homogenate. Pre- and post-injection levels of pancreatic flow rate, bicarbonate concentration and output were measured. Pancreas excision Rats with 75% proximal resection and the sham operated rats were killed by stunning and decapitation 4 weeks and 6 months following intestinal surgery. The entire pancreas was excised, trimmed of fat and extraneous tissue, weighed, and homogenized in 4 M-urea as described by Brants & Morisset (1976). Samples of the homogenate (8,ul.) were placed in a SW-5-1 rotor and centrifuged at 43, rev/min for 6 min. Supernatants were immediately collected and diluted in 73
4 74 M. D. GELINAS, C. L. MORIN AND J. MORISSET appropriate buffers for enzyme assays. DNA was measured by the method of Volkin & Cohn (1954) after extraction into 5 N-perchloric acid for 15min at 9TC. Calf thymus DNA was used as standard. Amylase was assayed according to Bernfeld (1955). Lipase activity was measured according to Charbonneau & Morisset (1975) using a ph stat titrimeter (Radiometer). The substrate was prepared as follows: one volume of emulsion (1 volume of 5 % olive oil and 1 volume of 1 % acacia) was mixed with two volumes of NaCl (5 M) and CaCl2 (1 mm); the final ph was set at 9-. Chymotrypsin was determined according to Hummel (1959) and activated by purified bovine trypsin (1 sg/ml.) for 9 min at 4C. All enzyme activities ere expressed in units (1umole product produced per mintue). Protein was determined according to Lowry et al. (1951) with bovine serum albumin as standard. On additional rats, fasted for 4-6 hr, the entire pancreas and total intestinal mucosa were weighed and assayed for somatostatin by the method of Epelbaum, Brazeau, Tsang, Brawer & Martin (1977), 1 and 4 weeks after 5 % small bowel resections, and compared with normal and sham operated rats. Blood was kept for gastrin (Squibb Immunotope New Brunswick, NJ) and secretin determinations by radio-immunoassay. Plasma secretin was assayed according to Tai & Chey (1978). The minimal detectable level was 18pg secretin/ml. plasma, and this was the amount assigned to samples with non-detectable secretin readings. Statistical analysis Results are expressed as means+ standard errors (s.e.). Unless otherwise mentioned, statistical comparisons were made using unpaired Student's t test. When the sample size was relatively small (n < 1), in which case departure from normality easily occurs, a distribution-free method (Mann- Whitney U test) was used in addition to the t test (Colton, 1974). As the t test and the U test gave the same result, only the t test is quoted. RESULTS Fifty per cent proximal gut resection Effects on pancreatic secretion Table 1 shows that compared with sham operated rats, a 5 % proximal resection induced after one week, a slight decrease of body weight, a 26 % increase in basal bicarbonate concentration of the pancreatic juice (P < -1), and a 43% increase in the basal bicarbonate output (P < -1). Differences in the concentration and output of protein of the basal pancreatic secretion were not significant. Secretin and CCK-PZ injections resulted in significant increases over basal values (P <.1, paired t test) in bicarbonate and protein secretion in both sham operated and resected rats. Although basal values differed between sham operated and resected rats, the results obtained following secretin and CCK-PZ injections were similar in both groups of animals. Four weeks after resection, body weights, basal pancreatic flow rate, bicarbonate, and protein concentration and output of sham operated and resected animals were not different. Effects on secretin, gastrin and somatostatin Table 2 shows that 1 week following a 5% proximal gut resection, the mean basal plasma secretin concentration (pg/ml.) was four times higher in resected rats ( ) than in sham operated rats ( ) (P < -1), and much higher than in normal rats (P < 1). However, after 4 weeks, there was no significant difference in basal plasma secretin concentration between resected an sham operated animals. No significant differences were observed between sham operated, resected and normal animals in basal serum gastrin concentration, and in somatostatin contents of both small intestinal mucosa and pancreas 1 or 4 weeks after intestinal surgery.
5 EXOCRINE PANCREAS AFTER ENTERECTOMY 75. C) Qia do a COP -_. -.Q ri;. 2._ q ;._ C; r. c3 s. r_ CL; o. V E o o - * 6 ;. - o 11 M - do o6 t le _t ' C1. 7 em o di.o.. = 1- o> = 4 64 N I t-1 1 if om o~n N r ~ Q Q bc * m en m _ N _ mc -+1+l ow t- I- +- N _ rn N = N g --q V * - o C) s * ++ - q$. (1) (1) e r c 3 g c d - e,, -o ed -..O, = x c: = m l (= = m l w * +-+-+e 4+~ *,- - 4Q (-
6 76 M. D. GELINAS, C. L. MORIN AND J. MORISSET Effects on pancreatic secretion The results of a similar study done 1 week, 4 weeks and 6 months after removal of 75 % of the proximal intestine are compared with those of sham operated rats in Table 3. Body weights were similar in resected and sham operated animals 4 weeks and 6 months following surgery. In the resected groups, 1 week and 4 weeks after surgery, pancreatic bicarbonate outputs were increased significantly by 78 % TABLE 2. Secretin, gastrin and somatostatin levels 1 and 4 weeks after a 5 % proximal intestinal resection Sham Resected Normals (6)t (5) (6) Basal plasma secretin (pg/ml.) 1 week T * 4 weeks t 1-8+ Basal serum gastrin (pg/ml.) 1 week weeks Somatostatin (ng) in total pancreas 1 week weeks Somatostatin (ng) in total mucosa 1 week ±125 4 weeks * P < 1 between sham operated and resected animals 1 week after surgery. t Figures in parentheses indicate the number of animals at each period. T Three values below the detection limit of 1-8 pg/ml. Five values below the detection limit of 1-8 pg/ml. (P < 1) and 5 % (P < 5), respectively. While these differences were due to both an increase in flow rate and bicarbonate concentration in the experimental groups, at 1 week only the bicarbonate concentration was significantly higher (47 %, P < 1), whereas at 4 weeks only the flow rate was statistically increased (27 %, P < -1). Differences in the concentration and output of proteins in the basal secretion at all periods of study were not significant and are not shown in Table 3. At 6 months following surgery the mean values for basal bicarbonate concentration and bicarbonate output were higher by 28 and 44 % respectively in the resected rats; however, those differences were not statistically significant due to a high dispersion of results as reflected by a large standard error. Stimulation with exogenous secretin and CCK-PZ resulted in increases over basal values (P < '1, paired t test) for the studied variables in both sham operated and resected animals: the only exception being in the protein concentration and output of the resected group, where stimulation failed to provide significant increases (results not shown). Fig. 1 demonstrates the significant inhibiting effect of an i.p. injection of a jejunoileal mucosa homogenate on the volume (-3 %, P < 5), bicarbonate concentration (-33 %, P < 1) and bicarbonate output (-54 %, P < 5) in seven 75 % resected rats compared to their own pre-injection values (paired t test). Pre- and
7 EXOCRINE PANCREAS AFTER ENTERECTOMY 77 -*t,i - = (M _r _ > 1-6. & cq cq C1)._._o.- * ) -o o. E. Cv o r- = (M * --4d = cq. co,: Cm m Im = Cb U6 ce C= r N N oz _ = C-oz =: N me m o4 m C14 v 4- *) C)._- a) ^ D:b >, _ m em N en m Cm m em c km v * e Q TD. 4) bl * ca Ca._.=4 z $.4 C. -C (1) (D j U C.) (1) m i 4) F..4 E --ll Ca E - 't - --,= ,. X t- M t- ITZ (1) 4'. C.) (1) m (1) E ---O ce.= tn. m t- llc. Q (1) m (1) $ E ce..-cl X Lm m t- C; C) a P4 X e cue.4., ce Q
8 78 M. D. GELINAS, C. L. MORIN AND J. MORISSET (7) (7) 4 2 *4 (6) 3- ~~~~15 E i2 Z lo1 BHi BHi BHm BHi BHi BHm BHi BHi BHm Volume HCO3 concentration HCO3 output Fig. 1. Effect of an i.p. injection of a jejunoileal mucosa (Hi) or abdominal muscle (Hm) homogenate on basal (B) pancreatic flow rate (volume), HC3- concentration and HCO3- output, 1 week after a 75 % proximal intestinal resection. Results are expressed as means (±s.e.) in sham operated (open bars) and resected rats (hatched bars). Figures in parentheses indicate the number of animals. TABLE 4. Pancreatic tissue 4 weeks after a 75% proximal intestinal resection (experimental group) per 1/sg DNA % change % change from from Mean + S.E. controlt Mean + S.E. controlt Final body weight (g) % -- Pancreas wet weight (mg) T12% Tl9% per g pancreas DNA (mg) % Protein (mg) %** III% Amylase (,smole/min) %* %** Lipase (,umole/min) %** 398& % Chymotrypsinogen (,umole/min) %** %** ** P<.1, * P<*5. t Indicate percentage change in six experimental animals compared with values from six rats taken as 1%. Not significant. post-mucosa injection values were similar in six sham operated animals. The effect of the i.p. injections of the mucosal homogenate was such that there were no longer any differences in pancreatic secretion between sham operated and resected rats. No significant changes were observed in seven other 75 % resected animals following the i.p. injection of a similarly prepared homogenate of abdominal muscles. Biochemical data on pancreatic tissue Pancreatic tissue was obtained 4 weeks (Table 4) and 6 months after 75 % resections; only the results after 4 weeks are shown here. At both periods, body weights, pancreas wet weights and DNA contents of sham operated and resected
9 EXOCRINE PANCREAS AFTER ENTERECTOMY 79 animals did not differ significantly. Decreases in tissue protein and enzyme levels per gram of pancreatic tissue were found in the resected animals in comparison with sham operated animals 4 weeks following resection: protein by 29 % (P < 5), amylase by 34% (P < 1), lipase by 21 % (P < -5) and chymotrypsinogen by 35 % (P < 2). Amylase (P < -1) and chymotrypsinogen (P < 5) contents expressed per 1#tg DNA were also decreased. Six months following resection, only the chymotrypsinogen content was reduced (41 %, P < -5) in the resected group. DISCUSSION This study showed that in rats, 5 and 75 % proximal intestinal resections induced an increase in basal pancreatic bicarbonate output after 1 week, due primarily to higher bicarbonate concentration. This hypersecretion of bicarbonate was accompanied by a 4-fold increase in basal plasma secretin concentration 1 week after intestinal resection. Increases in pancreatic bicarbonate secretion and in plasma secretin were transient, since one month after 5 % resection no differences were seen between sham operated and resected animals. Furthermore, the magnitude and duration of the increase in bicarbonate secretion were related to the extent of the resection being more pronounced and lasting longer in animals with larger resections. Four weeks after a 75 % resection, the bicarbonate output was still significantly higher in resected animals. This study also showed a decrease in certain digestive enzymes of the pancreatic tissue in 75 % resected rats, namely amylase, lipase, and chymotrypsinogen 4 weeks after resection. The tissue chymotrypsinogen level was still much lower 6 months after resection; this finding may explain the resected animals' lack of responsiveness in pancreatic protein secretion after hormonal stimulation. To our knowledge the present findings in partially enterectomized rats have not yet been reported. In comparison with studies measuring basal pancreatic juice in normal conscious rats, values for sham operated rats in the present study were similar in terms of flow rate, bicarbonate and protein concentrations (De Waele et al. 1974; Cavarzan, Teixeira, Sarles, Palasciano, Tiscornia, 1975; Chariot, Roze, LaTour, Souchard, Vaille & Debray, 1976). Some findings of the present study differ from work done by Baxter (1931) in rabbits and Grossman (1958) in rats who showed a marked reduction of the basal pancreatic secretions after gut resection. However, those experiments were performed, either the day of the enterectomy or 2 days later while our experiments were done 1, 4 weeks and 6 months after resection. Furthermore, in these two studies, the entire small bowel was removed, thus eliminating most of the secretin mechanism (Ramirez, Hubel & Clifton, 1966) and CCK-PZ activity (Schneeman & Lyman, 1977). In our study, the duodenum and the first 6 cm of the jejunum were intact. This, as well as the time interval after enterectomy, very likely accounts for the differences in results. In addition to the time interval after enterectomy, the differences observed by Niessen et al. (1974), Debas & Yamagishi (1977) and Seal et al. (1979) could be due to differences in species and state of consciousness, these two factors being known to influence largely basal pancreatic secretions (Chariot et al. 1976). The complex interaction between secretion of the gut, stomach and pancreas are very likely to be disturbed by removal of a part of the small bowel. Alteration of the
10 8 M. D. GELINAS, C. L. MORIN AND J. MORISSET gut nervous system may be responsible for the observed increase in volume and bicarbonate of the basal pancreatic secretion (Fleischer & Kasper, 1974); however, our experiment with mucosal homogenate injection argues against this explanation. Furthermore, hyperplasia or hypertrophy of the pancreas could not account for the results since no significant increases in pancreas weights, cell number (DNA/g pancreas), or total protein per cell (protein/,sg DNA) in the pancreatic tissue were found at different times after enterectomy. In addition, gastrin does not mediate the observed changes since no differences in serum gastrin were observed after resection; this has been previously reported (Oscarson, Veen, Williamson, Ross & Malt, 1977). It is tempting to speculate that following intestinal resection, the increase in volume and bicarbonate of the basal pancreatic secretion is induced by increased secretin concentration as observed, which is due to the loss of a jejunoileal inhibitors) and/or to gastric hypersecretion that could itself be secondary to the loss of jejunoileal inhibitors) (Osborne etal. 1966; Ruderman& Nagy, 1974). The inhibitory phenomenon observed in resected animals after mucosa homogenate injection, but not after muscle injection, shows that the results are due to some specific factor(s) present in the jejunoileal mucosa rather than to a non specific factor present in all tissues. Furthermore, the inhibitory phenomenon appears to be saturated in sham operated animals and suggests the presence of a limited number of receptor sites for a hormonal inhibitor controlling basal pancreatic secretion. Since somatostatin is present in the gastrointestinal mucosa and pancreas (Rufener, Dubois, Malaisse-Lagae & Orci, 1975), and has been found to inhibit basal pancreatic exocrine secretion in conscious rats (Chariot et al. 1978), we analysed the somatostatin content of the whole small bowel mucosa and pancreas at different times after resection. However, no difference was found between transacted and resected animals in either tissue, showing that the early adaptive growth that occurs in the gut mucosa after resection (Hanson et al. 1977; Obertop et al. 1977), restores rapidly the level of somatostatin in the gut and that a deficit in somatostatin is unlikely to explain the observed results. Although we do not know which mucosal factor is involved in the increased levels of secretin and basal pancreatic secretion following resection, the relationship between the magnitude of resection and the response in pancreatic secretion supports the existence ofmucosal inhibitorss. Furthermore, the adaptive intestinal hyperplasia and hypertrophy following proximal resection in rats may account for the transience of the pancreatic hypersecretion and hypersecretinemia, the inhibitors) being gradually synthesized within the proliferative response. Our observations are consistent with Debas & Yamagishi's hypothesis (1977), that resection of the jejunoileum removes an entero-pancreotone. Pancreotone, the polypeptide material extracted from ileal and colonic mucosa of pigs and cats (Harper, Hood, Mushens & Smy, 1979) may be the inhibitor of pancreatic secretion lost with resection, since part of the ileum was removed in our experiments. The observed hypersecretion in secretin, volume and bicarbonate of the basal pancreatic secretion may serve as useful adaptive mechanisms to counterbalance the gastric hypersecretion associated with extensive resection, and possibly to influence the adaptation of the remaining gut. This study was supported by the Medical Research Council of Canada (MA-332) and by the Fondation Justine Lacoste-Beaubien. The authors wish to express their gratitude to Dr W. Y. Chey for plasma secretin determinations,
11 EXOCRINE PANCREAS AFTER ENTERECTOMY 81 to Dr Paul Brazeau and Dr Robert Benoit for somatostatin assays and to Dr Jean Leveille for serum gastrin analyses. REFERENCES ALTMANN, G. G. (1971). Influence of bile and pancreatic secretions on the size of the intestinal villi in the rat. Am. J. Anat. 132, BAXTER, S. G. (1931). Continuous pancreatic secretion in the rabbit. Am. J. Phy8iol. %, BERNFELD, P. (1955). Amylases a & ft. Method in Enzymology, ed. COLOWICK, S. P. & KAPLAN, N. O., pp New York: Academic Press. BRANTS, F. & MORISSET, J. (1976). Trophic effect of cholecystokinin-pancreozymin on pancreatic acinar cells from rats of different ages. Proc. Soc. exp. Biol. Med. 153, BUXTON, B. (1974). Small bowel resection and gastric acid hypersecretion. Gut 15, CARIDIS, D. T., ROBERTS, M. & SMITH, G. (1969). The effect of small bowel resection on gastric acid secretion in the rat. Surgery, St-Loui8 65, CAVARZAN, A., TEIXEIRA, A. S., SARLES, H., PALASCIANO, G. & TISCORNIA,. (1975). Action of intragastric ethanol on the pancreatic secretion of conscious rats. Dige8tion 13, CHARBONNEAU, R. & MORISSET, J. (1975). Conditions optimales pour le dosage de la lipase pancreatique. Forum international francophone de Recherche8 en Gatroenterologie, p Sherbrooke: R. Prince. CHARIOT, J., RozE, C. LATOUR, J., SOUCHARD, M., VAILLE, C. & DEBRAY, C. (1976). La secretion pancreatique chez le rat. Pathol. Biol. (Pari8) 24, CHARIOT, J., Roz*, C., VAILLE, C. & DEBRAY, C. (1978). Effects of somatostatin on the external secretion of the pancreas of the rat. Gastroenterology 75, COLTON, T. (1974). Statistics in Medicine, chap. 4 and 7. Boston: Little, Brown. DEBAS, H. T. & YAMAGISHI, T. (1977). Enteric inhibition of pancreatic secretion: evidence for entero-pancreotone. Gatroenterology 72, 145. DE WAELE, B., DE SMUL, A., WISSOCK, P. & KIEKENS, R. (1974). La secr6tion pancreatique chez le rat. Influence de l'intervention chirurgicale, de la narcose, de l'hypothermie et de la derivation du suc gastrique ou du suc pancreatique. Biol. & Ga8tro-enterol. 7, DOWLING, R. H. & BOOTH, C. C. (1967). Structural and functional changes following small intestinal resection in the rat. Clin. Sci. 32, EPELBAUM, J., BRAZEAU, P., TSANG, D., BRAWER, J. & MARTIN, J. B. (1977). Subcellular distribution of radioimmunoassayable somatostatin in rat brain. Brain Res. 126, FIELDING, J. F. & COOKE, W. T. (197). Peptic ulceration in Crohn's disease (regional enteritis). Gut 11, FLEIsOHER, K. & KASPER, H. (1974). Exocrine pancreatic function in uraemic rats. Acta hepat. (Hamburg) 21, GWLINAS, M. D. & MORIN, C. L. (198). Effects ofbile and pancreatic secretions on intestinal mucosa after proximal small bowel resection in rats. Can. J. Phy8iol. & Pharmac. 58, GROSSMAN, M. I. (1958). Pancreatic secretion in the rat. Am. J. Phy8iol. 194, HANSON, W. R., OSBORNE, J. W. & SHARP, J. C. (1977). Compensation by the residual intestine after intestinal resection in the rat. II. Influence of post-operative time interval. Gastroenterology 72, HARPER, A. A., HOOD, A. J. C., MUSHENS, J. & SMY, J. R. (1979). Pancreotone, an inhibitor of pancreatic secretion in extracts of ileal and colonic mucosa. J. Physiol. 292, HUGHES, C. A., BATES, T. & DOWLING, R. (1978). Cholecystokinin and secretin prevent the intestinal mucosal hypoplasia of total parenteral nutrition in the dog Ga8troenterology 75, HUMMEL, B. C. (1959). A modified spectrophotometric determination of chymotrypsin, trypsin and thrombin. Can. J. Biochem. Phy8iol. 37, JACKSON, W. P. U. & LINDER, G. C. (1953). The influence of malnutrition on the pancreas. Report of a case following small gut resection. Metabolism 2, LAMBERT, R. (1965). Surgery of the Dige8tive Sy8tem in the Rat. Springfield, Ill.: C. C. Thomas. LOWRY,. H., ROSEBROUGH, N. J., FARR, A. L. & RANDALL, R. J. (1951). Protein measurement with the Folin phenol reagent. J. biol. Chem. 193, LEROY, J., MORISSET, J. A. & WEBSTER, P. D. (1971). Dose-related response of pancreatic synthesis and secretion to cholecystokinin-pancreozymin. J. Lab. clin. Med. 78,
12 82 M. D. GELINAS, C. L. MORIN AND J. MORISSET LUNDH, G. & OLDBY, N. (1963). Pancreatic secretion after massive resection of the small intestine. Acta chir. scand. 126, NATELSON, S. (1951). Routine use of ultramicro methods in the clinical laboratory. Am. J. clin. Path. 21, NIESSEN, K. H., BRtGMANN, G., OSSWALD, P., SCHMIDT, K. & FLACH, A. (1974). Studies of pancreas adaptation in infants and children after subtotal small bowel resection. Klin. Wschr. 52, NYGAARD, K. (1967). Resection of the small intestine in rats. III. Morphological changes in the intestinal tract. Acta chir. scand. 133, OBERTOP, H., NUNDY, S., MALAMUD, D. & MALT, R. A. (1977). Onset of cell proliferation in the shortened gut. Rapid hyperplasia after jejunal resection. Gastroenterology 72, OSBORNE, M. P., FREDERICK, P. L., SIZER, J. S., BLAIR, D., COLE, P. & THUM, W. (1966). Mechanism of gastric hypersecretion following massive intestinal resection. Ann. Surg. 164, OSCARSON, J. A., VEEN, H. F., WILLIAMSON, R. C. N., Ross, J. S. & MALT, R. A. (1977). Compensatory postresectional hyperplasia and starvation atrophy in small bowel: dissociation from endogenous gastrin levels. GCsatroenterology 72, RAMIREZ, J., HUBEL, K. A. & CLIFTON, J. A. (1966). Intestinal factors affecting pancreatic exocrine secretion in the rat. Am. J. Physiol. 211, R RUDERMAN, R. L. & NAGY, E. (1974). Intestinal control of gastric acid secretion in the rat. Ann. Surg. 179, RUFENER, C., DUBOIS, M. P., MALAISSE-LAGAE, F. & ORCI, L. (1975). Immunofluorescent reactivity to anti-somatostatin in the gastrointestinal mucosa of the dog. Diabetologia 11, SCHNEEMAN, B.. & LYMAN, R. L. (1977). Distribution of cholecystokinin activity in the rat small intestine. Digestion 15, SEAL, A. M., DEBAS, H. T., REYNOLDS, C. & SAID, S. I. (1979). Gastric and pancreatic hyposecretion following massive small bowel resection. Gastroenterology 76, SEELIG, L. L., WINBORN, W. B. & WESER, E. (1977). Effect of small bowel resection on the gastric mucosa in the rat. Gastroenterology 72, SINGH, M. & WEBSTER, P. D. (1978). Neurohormonal control ofpancreatic secretion. Calstroenterology 74, TAI, H. H. & CHEY, W. Y. (1978). Rapid extraction of secretin from plasma by XAD-2 resin and its application in the radioimmunoassay of secretin. Analyt. Biochem. 87, VOLKIN, E. & COHN, W. E. (1954). Estimation of nucleic acids. Analyt. Biochem. 1, WESER, E. (1976). The management of patients after small bowel resection. Gastroenterology 71, WESER, E., HELLER, R. & TAWIL, T. (1977). Stimulation of mucosal growth in the rat ileum by bile and pancreatic secretions after jejunal resection. Gastroenterology 73, WILLIAMSON, R. C. N., BAUER, F. L. R., ROSS, J. S. & MALT, R. A. (1978). Contributions of bile and pancreatic juice to cell proliferation in ileal mucosa. Surgery, St. Louis 83,
Cooke, Nahrwold and Grossman, 1967]. In the present experiments, attempts. Wales, 2033, Australia.
![Cooke, Nahrwold and Grossman, 1967]. In the present experiments, attempts. Wales, 2033, Australia. Cooke, Nahrwold and Grossman, 1967]. In the present experiments, attempts. Wales, 2033, Australia.](/thumbs/96/128693653.jpg) Quarterly Journal of Experimental Phyeiology (1973) 58, 335-343 BASAL AND POSTPRANDIAL PANCREATIC SECRETION IN RATS. By H. M. SiHw and T. J. HEATH. From the School of Physiology and Pharmacology, The University
Quarterly Journal of Experimental Phyeiology (1973) 58, 335-343 BASAL AND POSTPRANDIAL PANCREATIC SECRETION IN RATS. By H. M. SiHw and T. J. HEATH. From the School of Physiology and Pharmacology, The University
Hyperplasia of the exocrine pancreas after small bowel resection in the rat*
 Hyperplasia of the exocrine pancreas after small bowel resection in the rat* P HAEGEL, C STOCK,t J MARESCAUX, B PETIT, AND J F GRENIER From the INSERM, Unit 61, Biophysiopathology of the Intestine, Strasbourg,
Hyperplasia of the exocrine pancreas after small bowel resection in the rat* P HAEGEL, C STOCK,t J MARESCAUX, B PETIT, AND J F GRENIER From the INSERM, Unit 61, Biophysiopathology of the Intestine, Strasbourg,
Diversion of bile and pancreatic juices from the duodenum to the jejunum has
 GASTROENTEROLOGY Copyright 1969 by The Williams & Wilkins Co. Vol. 56, No.4 Printed in U.S.A. EFFECT OF EXCLUSION, ACIDIFICATION, AND EXCISION OF THE DUODENUM ON GASTRIC ACID SECRETION AND THE PRODUCTION
GASTROENTEROLOGY Copyright 1969 by The Williams & Wilkins Co. Vol. 56, No.4 Printed in U.S.A. EFFECT OF EXCLUSION, ACIDIFICATION, AND EXCISION OF THE DUODENUM ON GASTRIC ACID SECRETION AND THE PRODUCTION
Effect of acid infusion into various levels of the intestine on gastric and pancreatic secretion in the cat
 Gut, 1969, 10, 749-753 Effect of acid infusion into various levels of the intestine on gastric and pancreatic secretion in the cat S. J. KONTUREK, J. DUBIEL, AND B. GABRY9 From the Department of Medicine,
Gut, 1969, 10, 749-753 Effect of acid infusion into various levels of the intestine on gastric and pancreatic secretion in the cat S. J. KONTUREK, J. DUBIEL, AND B. GABRY9 From the Department of Medicine,
SIMULTANEOUS MEASUREMENT OF THE PANCREATIC AND BILIARY RESPONSE TO CCK AND SECRETIN
 GASTROENTEROLOGY 70:403-407, 1976 Copyright 1976 by The Williams & Wilkins Co. Vol. 70, No. 3 Printed in U.S.A. SIMULTANEOUS MEASUREMENT OF THE PANCREATIC AND BILIARY RESPONSE TO CCK AND SECRETIN Primate
GASTROENTEROLOGY 70:403-407, 1976 Copyright 1976 by The Williams & Wilkins Co. Vol. 70, No. 3 Printed in U.S.A. SIMULTANEOUS MEASUREMENT OF THE PANCREATIC AND BILIARY RESPONSE TO CCK AND SECRETIN Primate
EFFECT OF HORMONES ON PANCREATIC MACROMOLECULAR TRANSPORT
 GASTROENTEROLOGY 68: 1536-1542, 1975 Copyright 1975 by The Williams & Wilkins Co. Vol. 68, No.6 Printed in U.S.A. EFFECT OF HORMONES ON PANCREATIC MACROMOLECULAR TRANSPORT MANJIT SiNGH, M.D., F.R.C.P.
GASTROENTEROLOGY 68: 1536-1542, 1975 Copyright 1975 by The Williams & Wilkins Co. Vol. 68, No.6 Printed in U.S.A. EFFECT OF HORMONES ON PANCREATIC MACROMOLECULAR TRANSPORT MANJIT SiNGH, M.D., F.R.C.P.
DIRECT AND INDIRECT EFFECTS OF DEXTROSE AND AMINO ACIDS ON GUT MASS
 GASTROENTEROLOGY 72:76-71, 1977 Copyright 1977 by the American Gastroenterological Association Vol. 72, No.4, Part 1 Printed in U.S.A. DRECT AND NDRECT EFFECTS OF DEXTROSE AND AMNO ACDS ON GUT MASS MONROE
GASTROENTEROLOGY 72:76-71, 1977 Copyright 1977 by the American Gastroenterological Association Vol. 72, No.4, Part 1 Printed in U.S.A. DRECT AND NDRECT EFFECTS OF DEXTROSE AND AMNO ACDS ON GUT MASS MONROE
DIGESTIVE. CHAPTER 17 Lecture: Part 1 Part 2 BIO 212: ANATOMY & PHYSIOLOGY II
 BIO 212: ANATOMY & PHYSIOLOGY II CHAPTER 17 Lecture: DIGESTIVE Part 1 Part 2 Dr. Lawrence G. Altman www.lawrencegaltman.com Some illustrations are courtesy of McGraw-Hill. SMALL INTESTINE DUODENUM > JEJUNUM
BIO 212: ANATOMY & PHYSIOLOGY II CHAPTER 17 Lecture: DIGESTIVE Part 1 Part 2 Dr. Lawrence G. Altman www.lawrencegaltman.com Some illustrations are courtesy of McGraw-Hill. SMALL INTESTINE DUODENUM > JEJUNUM
(Received 6 August 1979)
 J. Phyoiol. (1980), 303, pp. 33-41 33 With 2 text-figurew Printed in Great Britain PARALLEL SECRETION OF ENZYMES BY THE RABBIT PANCREAS BY E. L. GILLILAND* AND G. GLAZER From the Academic Surgical Unit,
J. Phyoiol. (1980), 303, pp. 33-41 33 With 2 text-figurew Printed in Great Britain PARALLEL SECRETION OF ENZYMES BY THE RABBIT PANCREAS BY E. L. GILLILAND* AND G. GLAZER From the Academic Surgical Unit,
EFFECTS OF EXOGENOUSLY ADDED SHORT-CHAIN FATTY ACIDS ON PANCREATIC EXOCRINE SECRETION IN DOMESTIC RABBIT
 EFFECTS OF EXOGENOUSLY ADDED SHORT-CHAIN FATTY ACIDS ON PANCREATIC EXOCRINE SECRETION IN DOMESTIC RABBIT DOJANA N 1., POP A 2., PAPUC C 3. 1 Department of Animal Physiology, Faculty of Veterinary Medicine,
EFFECTS OF EXOGENOUSLY ADDED SHORT-CHAIN FATTY ACIDS ON PANCREATIC EXOCRINE SECRETION IN DOMESTIC RABBIT DOJANA N 1., POP A 2., PAPUC C 3. 1 Department of Animal Physiology, Faculty of Veterinary Medicine,
Chapter 26 The Digestive System
 Chapter 26 The Digestive System Digestive System Gastroenterology is the study of the stomach and intestine. Digestion Catabolism Absorption Anabolism The actions of the digestive system are controlled
Chapter 26 The Digestive System Digestive System Gastroenterology is the study of the stomach and intestine. Digestion Catabolism Absorption Anabolism The actions of the digestive system are controlled
Ali Yaghi. Yaseen Fatayer. M.Khatatbeh
 6 Ali Yaghi Yaseen Fatayer M.Khatatbeh P a g e 1 pancreatic secretions note: The pancreas has endocrine (secretions are released toward the blood) and exocrine(secretions are released through the canalicular
6 Ali Yaghi Yaseen Fatayer M.Khatatbeh P a g e 1 pancreatic secretions note: The pancreas has endocrine (secretions are released toward the blood) and exocrine(secretions are released through the canalicular
Understandings, Applications & Skills
 D.2 Digestion Understandings, Applications & Skills Statement D.2.U1 Nervous and hormonal mechanisms control the secretion of digestive juices. D.2.U2 Exocrine glands secrete to the surface of the body
D.2 Digestion Understandings, Applications & Skills Statement D.2.U1 Nervous and hormonal mechanisms control the secretion of digestive juices. D.2.U2 Exocrine glands secrete to the surface of the body
Includes mouth, pharynx, esophagus, stomach, small intestine, large intestine, rectum, anus. Salivary glands, liver, gallbladder, pancreas
 Chapter 14 The Digestive System and Nutrition Digestive System Brings Nutrients Into the Body The digestive system includes Gastrointestinal (GI) tract (hollow tube) Lumen: space within this tube Includes
Chapter 14 The Digestive System and Nutrition Digestive System Brings Nutrients Into the Body The digestive system includes Gastrointestinal (GI) tract (hollow tube) Lumen: space within this tube Includes
Causes of pancreatic insufficiency. Eugen Dumitru
 Causes of pancreatic insufficiency Eugen Dumitru Pancreatic Exocrine Insufficiency (PEI) 1. The Concept 2. The Causes 3. The Consequences Pancreatic Exocrine Insufficiency (PEI) 1. The Concept 2. The Causes
Causes of pancreatic insufficiency Eugen Dumitru Pancreatic Exocrine Insufficiency (PEI) 1. The Concept 2. The Causes 3. The Consequences Pancreatic Exocrine Insufficiency (PEI) 1. The Concept 2. The Causes
THE DEPENDENCE OF EXOCRINE PANCREATIC SECRETION ON INSULIN IN SHEEP
 Quarterly Journal of Experimental Physiology (1984) 69, 35-39 3 5 Printed in Great Britain THE DEPENDENCE OF EXOCRINE PANCREATIC SECRETION ON INSULIN IN SHEEP STEFAN PIERZYNOWSKI AND W. BAREJ The Institute
Quarterly Journal of Experimental Physiology (1984) 69, 35-39 3 5 Printed in Great Britain THE DEPENDENCE OF EXOCRINE PANCREATIC SECRETION ON INSULIN IN SHEEP STEFAN PIERZYNOWSKI AND W. BAREJ The Institute
EFFECT OF VAGOTOMY ON PANCREATIC SECRETION STIMULATED BY ENDOGENOUS AND EXOGENOUS SECRETIN
 GASTROENTEROLOGY Copyright,. 1971 by The Williams & Wilkins Co. Vol. 60, No. 3 P>-inted in U. S. A. EFFECT OF VAGOTOMY ON PANCREATIC SECRETION STIMULATED BY ENDOGENOUS AND EXOGENOUS SECRETIN HARRIS J.
GASTROENTEROLOGY Copyright,. 1971 by The Williams & Wilkins Co. Vol. 60, No. 3 P>-inted in U. S. A. EFFECT OF VAGOTOMY ON PANCREATIC SECRETION STIMULATED BY ENDOGENOUS AND EXOGENOUS SECRETIN HARRIS J.
Chapter 14: The Digestive System
 Chapter 14: The Digestive System Digestive system consists of Muscular tube (digestive tract) alimentary canal Accessory organs teeth, tongue, glandular organs 6 essential activities 1. 2. 3. 4. 5. 6.
Chapter 14: The Digestive System Digestive system consists of Muscular tube (digestive tract) alimentary canal Accessory organs teeth, tongue, glandular organs 6 essential activities 1. 2. 3. 4. 5. 6.
Chapter 15 Gastrointestinal System
 Chapter 15 Gastrointestinal System Dr. LL Wang E-mail: wanglinlin@zju.edu.cn Rm 608, Block B, Research Building, School of Medicine, Zijingang Campus Pancreatic Secretion The exocrine cells in the pancreas
Chapter 15 Gastrointestinal System Dr. LL Wang E-mail: wanglinlin@zju.edu.cn Rm 608, Block B, Research Building, School of Medicine, Zijingang Campus Pancreatic Secretion The exocrine cells in the pancreas
rabbit, 45 min for dog) and more slowly for dehydrocholic acid (25- decrease, questioning the mechanism by which bile acids increase bile
 J. Physiol. (1972), 224, pp. 259-269 259 With 6 text-ftgure8 Printed in Great Britain SPECIES DIFFERENCES IN THE CHOLERETIC RESPONSE TO BILE SALTS BY CURTIS D. KLAASSEN From the Clinical Pharmacology and
J. Physiol. (1972), 224, pp. 259-269 259 With 6 text-ftgure8 Printed in Great Britain SPECIES DIFFERENCES IN THE CHOLERETIC RESPONSE TO BILE SALTS BY CURTIS D. KLAASSEN From the Clinical Pharmacology and
Physiology Unit 4 DIGESTIVE PHYSIOLOGY
 Physiology Unit 4 DIGESTIVE PHYSIOLOGY In Physiology Today Functions Motility Ingestion Mastication Deglutition Peristalsis Secretion 7 liters/day! Exocrine/endocrine Digestion Absorption Digestion of
Physiology Unit 4 DIGESTIVE PHYSIOLOGY In Physiology Today Functions Motility Ingestion Mastication Deglutition Peristalsis Secretion 7 liters/day! Exocrine/endocrine Digestion Absorption Digestion of
MECHANISM BY WHICH FAT IN THE UPPER SMALL INTESTINE INHIBITS GASTRIC ACID
 GASTROENTEROLOGY Copyright 1969 by The Williams & Wilkins Co. Vol. 56, No.3 Printea in U.S.A. MECHANISM BY WHICH FAT IN THE UPPER SMALL INTESTINE INHIBITS GASTRIC ACID H. T. DEBAS, M.D., B. S. BEDI, M.B.,
GASTROENTEROLOGY Copyright 1969 by The Williams & Wilkins Co. Vol. 56, No.3 Printea in U.S.A. MECHANISM BY WHICH FAT IN THE UPPER SMALL INTESTINE INHIBITS GASTRIC ACID H. T. DEBAS, M.D., B. S. BEDI, M.B.,
Pancreas Fox Chapter 18 part 2 (also Chapter 19.3 & 19.4)
 Vert Phys PCB3743 Pancreas Fox Chapter 18 part 2 (also Chapter 19.3 & 19.4) T. Houpt, Ph.D. Anatomy of Digestive System Peristalsis Stomach and Acid Secretion Liver and Bile Secretion Pancreas and pancreatic
Vert Phys PCB3743 Pancreas Fox Chapter 18 part 2 (also Chapter 19.3 & 19.4) T. Houpt, Ph.D. Anatomy of Digestive System Peristalsis Stomach and Acid Secretion Liver and Bile Secretion Pancreas and pancreatic
Gastrointestinal Anatomy and Physiology. Bio 219 Napa Valley College Dr. Adam Ross
 Gastrointestinal Anatomy and Physiology Bio 219 Napa Valley College Dr. Adam Ross Functions of digestive system Digestion Breakdown of food (chemically) using enzymes, acid, and water Absorption Nutrients,
Gastrointestinal Anatomy and Physiology Bio 219 Napa Valley College Dr. Adam Ross Functions of digestive system Digestion Breakdown of food (chemically) using enzymes, acid, and water Absorption Nutrients,
NOTES: The Digestive System (Ch 14, part 2)
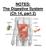 NOTES: The Digestive System (Ch 14, part 2) PANCREAS Structure of the pancreas: The pancreas produces PANCREATIC JUICE that is then secreted into a pancreatic duct. The PANCREATIC DUCT leads to the The
NOTES: The Digestive System (Ch 14, part 2) PANCREAS Structure of the pancreas: The pancreas produces PANCREATIC JUICE that is then secreted into a pancreatic duct. The PANCREATIC DUCT leads to the The
Section Coordinator: Jerome W. Breslin, PhD, Assistant Professor of Physiology, MEB 7208, ,
 IDP Biological Systems Gastrointestinal System Section Coordinator: Jerome W. Breslin, PhD, Assistant Professor of Physiology, MEB 7208, 504-568-2669, jbresl@lsuhsc.edu Overall Learning Objectives 1. Characterize
IDP Biological Systems Gastrointestinal System Section Coordinator: Jerome W. Breslin, PhD, Assistant Professor of Physiology, MEB 7208, 504-568-2669, jbresl@lsuhsc.edu Overall Learning Objectives 1. Characterize
[Jorpes, 1968] has shown that these hormones have a common
![[Jorpes, 1968] has shown that these hormones have a common [Jorpes, 1968] has shown that these hormones have a common](/thumbs/95/124914414.jpg) Q. Jl exp. Phy8iol. (1971) 56, 113-122 THE INFLUENCE OF CHRONIC ADMINISTRATION OF PENTA- GASTRIN ON THE RAT PANCREAS. By P. D. MAYSTON and J. A. BARROWMAN. From the Department of Physiology, The London
Q. Jl exp. Phy8iol. (1971) 56, 113-122 THE INFLUENCE OF CHRONIC ADMINISTRATION OF PENTA- GASTRIN ON THE RAT PANCREAS. By P. D. MAYSTON and J. A. BARROWMAN. From the Department of Physiology, The London
University of Buea. Faculty of Health Sciences. Programme in Medicine
 Faculty of Health Sciences University of Buea Wednesday, 28 th January 2009 Time: 8 00-10 00 Programme in Medicine MED 303 (Gastrointestinal Physiology) EXAMS (2008-2009) Identify the letter of the choice
Faculty of Health Sciences University of Buea Wednesday, 28 th January 2009 Time: 8 00-10 00 Programme in Medicine MED 303 (Gastrointestinal Physiology) EXAMS (2008-2009) Identify the letter of the choice
Sphincters heartburn diaphragm The Stomach gastric glands pepsin, chyme The Small Intestine 1-Digestion Is Completed in the Small Intestine duodenum
 Sphincters are muscles that encircle tubes and act as valves. The tubes close when the sphincters contract and they open when the sphincters relax. When food or saliva is swallowed, the sphincter relaxes
Sphincters are muscles that encircle tubes and act as valves. The tubes close when the sphincters contract and they open when the sphincters relax. When food or saliva is swallowed, the sphincter relaxes
PHYSIOLOGY OF THE DIGESTIVE SYSTEM
 Student Name CHAPTER 26 PHYSIOLOGY OF THE DIGESTIVE SYSTEM D igestion is the process of breaking down complex nutrients into simpler units suitable for absorption. It involves two major processes: mechanical
Student Name CHAPTER 26 PHYSIOLOGY OF THE DIGESTIVE SYSTEM D igestion is the process of breaking down complex nutrients into simpler units suitable for absorption. It involves two major processes: mechanical
Station 1. Identify (= name) the spaces or structures labeled 1 9.
 Station 1. Identify (= name) the spaces or structures labeled 1 9. 1 9 8 2 7 3 4 5 6 Station 2. Identify/name the parts or structures labeled 10 20. 10. Is this the right or left lung? 15 13 14 16 11 12
Station 1. Identify (= name) the spaces or structures labeled 1 9. 1 9 8 2 7 3 4 5 6 Station 2. Identify/name the parts or structures labeled 10 20. 10. Is this the right or left lung? 15 13 14 16 11 12
Biology 12 Unit 5 Pretest
 Biology Unit 5 Pretest Name: Date: Block:. Chewing aids digestion by A. stimulating release of bile B. increasing surface area of food C. breaking up polymers into monomers D. stimulating water absorption.
Biology Unit 5 Pretest Name: Date: Block:. Chewing aids digestion by A. stimulating release of bile B. increasing surface area of food C. breaking up polymers into monomers D. stimulating water absorption.
GASTROENTEROLOGY. Official Publication of the American Gastroenterological Association. COPYRIGHT 1974 THE \VILLlAMS & WILKINS Co.
 GASTROENTEROLOGY Official Publication of the American Gastroenterological Association COPYRIGHT 1974 THE \VILLlAMS & WILKINS Co. Vol. 66 February 1974 Number 2 HYPERSECRETION OF GASTRIN ASSOCIATED WITH
GASTROENTEROLOGY Official Publication of the American Gastroenterological Association COPYRIGHT 1974 THE \VILLlAMS & WILKINS Co. Vol. 66 February 1974 Number 2 HYPERSECRETION OF GASTRIN ASSOCIATED WITH
Symposium on diagnosis of pancreatic disease
 Gut, 1975, 16, 648-663 Symposium on diagnosis of pancreatic disease Pancreatic function tests: T. SCRATCHERD The physiological background From the Department ofphysiology, The University of Sheffield,
Gut, 1975, 16, 648-663 Symposium on diagnosis of pancreatic disease Pancreatic function tests: T. SCRATCHERD The physiological background From the Department ofphysiology, The University of Sheffield,
THE INHIBITORY EFFECT OF STILBOESTROL ON GASTRIC SECRETION IN CATS
 Brit. J. Pharmacol. (1950), 5, 3S9. THE INHIBITORY EFFECT OF STILBOESTROL ON GASTRIC SECRETION IN CATS BY K. N. OJHA* AND D. R. WOOD From the Department of Pharmacology and Therapeutics, University of
Brit. J. Pharmacol. (1950), 5, 3S9. THE INHIBITORY EFFECT OF STILBOESTROL ON GASTRIC SECRETION IN CATS BY K. N. OJHA* AND D. R. WOOD From the Department of Pharmacology and Therapeutics, University of
Digestive Lecture Test Questions Set 4
 Digestive Lecture Test Questions Set 4 1. Which of the following is not associated directly with the small intestine: a. villi b. circular folds c. microvilli d. haustrae e. secretin 2. The largest (longest)
Digestive Lecture Test Questions Set 4 1. Which of the following is not associated directly with the small intestine: a. villi b. circular folds c. microvilli d. haustrae e. secretin 2. The largest (longest)
OPERATIVE TREATMENT OF ULCER DISEASE
 Página 1 de 8 Copyright 2001 Lippincott Williams & Wilkins Greenfield, Lazar J., Mulholland, Michael W., Oldham, Keith T., Zelenock, Gerald B., Lillemoe, Keith D. Surgery: Scientific Principles & Practice,
Página 1 de 8 Copyright 2001 Lippincott Williams & Wilkins Greenfield, Lazar J., Mulholland, Michael W., Oldham, Keith T., Zelenock, Gerald B., Lillemoe, Keith D. Surgery: Scientific Principles & Practice,
1-It is to prevent back flow of fecal content from colon into small intestine.
 Function of the ileocecal valve: 1-It is to prevent back flow of fecal content from colon into small intestine. 2-The wall of the ileum for several centimeters preceding valve has a thickened muscular
Function of the ileocecal valve: 1-It is to prevent back flow of fecal content from colon into small intestine. 2-The wall of the ileum for several centimeters preceding valve has a thickened muscular
to food and histamine
 Gut, 97,, 53-57 Maximal acid response of Pavlov pouches to food and histamine A. MARVIN BROOKS AND MORTON I. GROSSMAN From the Veterans Administration Center and UCLA School of Medicine, Departments of
Gut, 97,, 53-57 Maximal acid response of Pavlov pouches to food and histamine A. MARVIN BROOKS AND MORTON I. GROSSMAN From the Veterans Administration Center and UCLA School of Medicine, Departments of
STUDIES OF SMALL BOWEL ADAPTATION AFTER INTESTINAL RESECTION IN THE RAT
 GASTROENTEROLOGY Copyright @ 1971 by The Williams & Wilkins Co. Vol. 60, No.1 Printed in U. S. A. STUDIES OF SMALL BOWEL ADAPTATION AFTER INTESTINAL RESECTION IN THE RAT ELLIOT WESER, M.D., AND MARY H.
GASTROENTEROLOGY Copyright @ 1971 by The Williams & Wilkins Co. Vol. 60, No.1 Printed in U. S. A. STUDIES OF SMALL BOWEL ADAPTATION AFTER INTESTINAL RESECTION IN THE RAT ELLIOT WESER, M.D., AND MARY H.
Gut hormones in gastrointestinal disease
 J. clin. Path., 33, Suppl. (Ass. Clin. Path.), 8, 76-84 Gut hormones in gastrointestinal disease H. S. BESTERMAN From the Department of Medicine, Royal Postgraduate Medical School, Du Cane Road, London
J. clin. Path., 33, Suppl. (Ass. Clin. Path.), 8, 76-84 Gut hormones in gastrointestinal disease H. S. BESTERMAN From the Department of Medicine, Royal Postgraduate Medical School, Du Cane Road, London
ESPEN Congress The Hague 2017
 ESPEN Congress The Hague 2017 Meeting nutritional needs of acute care patients Feeding acute pancreatitis patients J. Luttikhold (NL) FEEDING ACUTE PANCREATITIS PATIENTS Joanna Luttikhold, MD PhD Registrar
ESPEN Congress The Hague 2017 Meeting nutritional needs of acute care patients Feeding acute pancreatitis patients J. Luttikhold (NL) FEEDING ACUTE PANCREATITIS PATIENTS Joanna Luttikhold, MD PhD Registrar
Overview of digestion or, gut reactions - to food
 Key concepts in Digestion. Indigestion module Overview of digestion or, gut reactions - to food Prof. Barry Campbell Gastroenterology Cellular & Molecular Physiology e-mail: bjcampbl@liv.ac.uk http://pcwww.liv.ac.uk/~bjcampbl
Key concepts in Digestion. Indigestion module Overview of digestion or, gut reactions - to food Prof. Barry Campbell Gastroenterology Cellular & Molecular Physiology e-mail: bjcampbl@liv.ac.uk http://pcwww.liv.ac.uk/~bjcampbl
THE SIGNIFICANCE OF HORMONES, BILE SALTS, AND FEEDING IN THE REGULATION OF BILE AND OTHER DIGESTIVE SECRETIONS IN THE RAT
 THE SIGNIFICANCE OF HORMONES, BILE SALTS, AND FEEDING IN THE REGULATION OF BILE AND OTHER DIGESTIVE SECRETIONS IN THE RAT By H. M. SHA w* and T. HEATH* [Manuscript received 16 July 1971] Abstract Conscious
THE SIGNIFICANCE OF HORMONES, BILE SALTS, AND FEEDING IN THE REGULATION OF BILE AND OTHER DIGESTIVE SECRETIONS IN THE RAT By H. M. SHA w* and T. HEATH* [Manuscript received 16 July 1971] Abstract Conscious
The physiology of gastrointestinal system 3.
 The physiology of gastrointestinal system 3. Stomach, pancreas, bile LO.: 64,65,66 Prof. Gyula Sáry While studying functions of the gastrointestinal system, Pavlov described the conditional response, a
The physiology of gastrointestinal system 3. Stomach, pancreas, bile LO.: 64,65,66 Prof. Gyula Sáry While studying functions of the gastrointestinal system, Pavlov described the conditional response, a
Overview of digestion or, gut reactions - to food
 1 Key concepts in Digestion. Indigestion module Overview of digestion or, gut reactions to food Prof. Barry Campbell Gastroenterology Cellular & Molecular Physiology email: bjcampbl@liv.ac.uk http://pcwww.liv.ac.uk/~bjcampbl
1 Key concepts in Digestion. Indigestion module Overview of digestion or, gut reactions to food Prof. Barry Campbell Gastroenterology Cellular & Molecular Physiology email: bjcampbl@liv.ac.uk http://pcwww.liv.ac.uk/~bjcampbl
Soft palate elevates, closing off the nasopharynx. Hard palate Tongue Bolus Epiglottis. Glottis Larynx moves up and forward.
 The Cephalic Phase Chemical and mechanical digestion begins in the mouth Saliva is an exocrine secretion Salivary secretion is under autonomic control Softens and lubricates food Chemical digestion: salivary
The Cephalic Phase Chemical and mechanical digestion begins in the mouth Saliva is an exocrine secretion Salivary secretion is under autonomic control Softens and lubricates food Chemical digestion: salivary
Digestive System. Part A Multiple Choice. 1. Which of the following is NOT a digestive enzyme? A. Pepsin. B. Ptyalin. C. Gastrin. D. Trypsin.
 Digestive System Part A Multiple Choice 1. Which of the following is NOT a digestive enzyme? A. Pepsin. B. Ptyalin. C. Gastrin. D. Trypsin. 2. The presence of large numbers of mitochondria in the cells
Digestive System Part A Multiple Choice 1. Which of the following is NOT a digestive enzyme? A. Pepsin. B. Ptyalin. C. Gastrin. D. Trypsin. 2. The presence of large numbers of mitochondria in the cells
The physiology of gastrointestinal system 3.
 The physiology of gastrointestinal system 3. Stomach, pancreas, bile Dr. Gabriella Kékesi The mechanism and regulation of gastric juice secretion (Lo.) 64. Secretory cells in stomach Composition and role
The physiology of gastrointestinal system 3. Stomach, pancreas, bile Dr. Gabriella Kékesi The mechanism and regulation of gastric juice secretion (Lo.) 64. Secretory cells in stomach Composition and role
diabetes mellitus and chronic pancreatitis
 Gut, 1966, 7, 277 Exocrine and endocrine pancreatic function in diabetes mellitus and chronic pancreatitis N. PETERS1, A. P. DCK, C. N. HALES, D. H. ORRELL, AND MARTN SARNER1 From Addenbrooke's Hospital
Gut, 1966, 7, 277 Exocrine and endocrine pancreatic function in diabetes mellitus and chronic pancreatitis N. PETERS1, A. P. DCK, C. N. HALES, D. H. ORRELL, AND MARTN SARNER1 From Addenbrooke's Hospital
The Digestive System. What is the advantage of a one-way gut? If you swallow something, is it really inside you?
 The Digestive System What is the advantage of a one-way gut?! If you swallow something, is it really inside you? Functions and Processes of the Digestive System: Move nutrients, water, electrolytes from
The Digestive System What is the advantage of a one-way gut?! If you swallow something, is it really inside you? Functions and Processes of the Digestive System: Move nutrients, water, electrolytes from
Nutrition. Autotrophs. plants, some protists & bacteria producers
 Nutrition Autotrophs plants, some protists & bacteria producers Nutrition Heterotrophs animals, fungi, some protists & bacteria consumers Animal Nutrition Most obtain food by ingestion take in their food
Nutrition Autotrophs plants, some protists & bacteria producers Nutrition Heterotrophs animals, fungi, some protists & bacteria consumers Animal Nutrition Most obtain food by ingestion take in their food
DIGESTION SBI 3C: NOVEMBER 2010
 DIGESTION SBI 3C: NOVEMBER 2010 DIAGRAM OF DIGESTIVE SYSTEM: Mouth Esophagus Liver Gallbladder Large Intestine Appendix Stomach Pancreas Small Intestine Rectum Anus STAGES OF DIGESTION: 1. INGESTION Taking
DIGESTION SBI 3C: NOVEMBER 2010 DIAGRAM OF DIGESTIVE SYSTEM: Mouth Esophagus Liver Gallbladder Large Intestine Appendix Stomach Pancreas Small Intestine Rectum Anus STAGES OF DIGESTION: 1. INGESTION Taking
Digestive System 7/15/2015. Outline Digestive System. Digestive System
 Digestive System Biology 105 Lecture 18 Chapter 15 Outline Digestive System I. Functions II. Layers of the GI tract III. Major parts: mouth, pharynx, esophagus, stomach, small intestine, large intestine,
Digestive System Biology 105 Lecture 18 Chapter 15 Outline Digestive System I. Functions II. Layers of the GI tract III. Major parts: mouth, pharynx, esophagus, stomach, small intestine, large intestine,
Digestive System Module 6: Accessory Organs in Digestion: The Liver, Pancreas, and Gallbladder
 Connexions module: m49293 1 Digestive System Module 6: Accessory Organs in Digestion: The Liver, Pancreas, and Gallbladder Donna Browne Based on Accessory Organs in Digestion: The Liver, Pancreas, and
Connexions module: m49293 1 Digestive System Module 6: Accessory Organs in Digestion: The Liver, Pancreas, and Gallbladder Donna Browne Based on Accessory Organs in Digestion: The Liver, Pancreas, and
Intestinal phase of gastric secretion in patients with
 Gut, 1978, 19, 321-326 Intestinal phase of gastric secretion in patients with duodenal ulcer S. J. KONTUREK1, N. KWIECIEN, W. OBTULOWICZ, E. SITO, AND J. OLEKSY From the Institute ofphysiology, Medical
Gut, 1978, 19, 321-326 Intestinal phase of gastric secretion in patients with duodenal ulcer S. J. KONTUREK1, N. KWIECIEN, W. OBTULOWICZ, E. SITO, AND J. OLEKSY From the Institute ofphysiology, Medical
Digestive System. Digestive System. Digestion is the process of reducing food to small molecules that can be absorbed into the body.
 Digestive System Digestion is the process of reducing food to small molecules that can be absorbed into the body. 2 Types of Digestion Mechanical digestion physical breakdown of food into small particles
Digestive System Digestion is the process of reducing food to small molecules that can be absorbed into the body. 2 Types of Digestion Mechanical digestion physical breakdown of food into small particles
s. J. RUNE, M.D., AND F. W. HENRIKSEN, M.D.
 GASTROENTEROLOGY Copyright 1969 by The Williams & Wilkins Co. Vol. 56, No.4 Printed in U.S.A. CARBON DOXDE TENSONS N TlE PROXMAL PART OF THE CANNE GASTRONTESTNAL TRACT s. J. RUNE, M.D., AND F. W. HENRKSEN,
GASTROENTEROLOGY Copyright 1969 by The Williams & Wilkins Co. Vol. 56, No.4 Printed in U.S.A. CARBON DOXDE TENSONS N TlE PROXMAL PART OF THE CANNE GASTRONTESTNAL TRACT s. J. RUNE, M.D., AND F. W. HENRKSEN,
Physiology 12. Overview. The Gastrointestinal Tract. Germann Ch 19
 Physiology 12 The Gastrointestinal Tract Germann Ch 19 Overview 1 Basic functions of the GI tract Digestion Secretion Absorption Motility Basic functions of the GI tract Digestion: : Dissolving and breaking
Physiology 12 The Gastrointestinal Tract Germann Ch 19 Overview 1 Basic functions of the GI tract Digestion Secretion Absorption Motility Basic functions of the GI tract Digestion: : Dissolving and breaking
Gastrointestinal Physiology. Secretion
 Gastrointestinal Physiology Secretion Fig. 24.26 Functions Provided by secretory glands which serve 2 functions: - Digestive enzymes. - Lubrication and protection of the mucosa. Types of secretory structures
Gastrointestinal Physiology Secretion Fig. 24.26 Functions Provided by secretory glands which serve 2 functions: - Digestive enzymes. - Lubrication and protection of the mucosa. Types of secretory structures
NURSE-UP DIGESTIVE SYSTEM AKA G.I. SYSTEM
 NURSE-UP DIGESTIVE SYSTEM AKA G.I. SYSTEM The digestive system is used for breaking down food into nutrients which then pass into the circulatory system and are taken to where they are needed in the body.
NURSE-UP DIGESTIVE SYSTEM AKA G.I. SYSTEM The digestive system is used for breaking down food into nutrients which then pass into the circulatory system and are taken to where they are needed in the body.
pancreaticobiliary diversion in rats. An effect of raised CCK plasma concentrations
 Gut, 1987, 28, SI, 269-273 Exocrine pancreatic nodules after longterm pancreaticobiliary diversion in rats. An effect of raised CCK plasma concentrations B M MIAZZA, S WIDGREN, J A CHAYVIALLE, T NICOLET,
Gut, 1987, 28, SI, 269-273 Exocrine pancreatic nodules after longterm pancreaticobiliary diversion in rats. An effect of raised CCK plasma concentrations B M MIAZZA, S WIDGREN, J A CHAYVIALLE, T NICOLET,
Bio& 242 Unit 1 / Lecture 4
 Bio& 242 Unit 1 / Lecture 4 system: Gastric hormones GASTRIN: Secretion: By enteroendocrine (G) in gastric pits of the mucosa. Stimulus: Stomach distention and acid ph of chyme causes Gastrin. Action:
Bio& 242 Unit 1 / Lecture 4 system: Gastric hormones GASTRIN: Secretion: By enteroendocrine (G) in gastric pits of the mucosa. Stimulus: Stomach distention and acid ph of chyme causes Gastrin. Action:
6.2.1 Exocrine pancreatic insufficiency
 6.2.1 Exocrine pancreatic insufficiency Authors: Jean Louis Frossard, Alain Sauty 1. INTRODUCTION Exocrine pancreatic insufficiency is a biological and clinical condition that is characterized by a progressive
6.2.1 Exocrine pancreatic insufficiency Authors: Jean Louis Frossard, Alain Sauty 1. INTRODUCTION Exocrine pancreatic insufficiency is a biological and clinical condition that is characterized by a progressive
Insulin Stimulation test
 Insulin Stimulation test Insulin hypogycemia is a potent stimulus of acid secretion. When blood sugar is < 50.0mg/dl (2.8mmol/L) vagus is stimulated by hypoglycemia. This test is best limited to those
Insulin Stimulation test Insulin hypogycemia is a potent stimulus of acid secretion. When blood sugar is < 50.0mg/dl (2.8mmol/L) vagus is stimulated by hypoglycemia. This test is best limited to those
Kinin forming and destroying activities in human bile and mucous membranes of the biliary tract
 Br. J. Pharmac. (1969), 37, 172-177. Kinin forming and destroying activities in human bile and mucous membranes of the biliary tract H. M. NIELSEN Surgical Department L, Aarhus Municipal Hospital, University
Br. J. Pharmac. (1969), 37, 172-177. Kinin forming and destroying activities in human bile and mucous membranes of the biliary tract H. M. NIELSEN Surgical Department L, Aarhus Municipal Hospital, University
An overview of the digestive system. mouth pharynx esophagus stomach small intestine large intestine rectum anus
 An overview of the digestive system mouth pharynx esophagus stomach small intestine large intestine rectum anus Why GIT? What are the main steps in the digestive process? Ingestion intake of food via the
An overview of the digestive system mouth pharynx esophagus stomach small intestine large intestine rectum anus Why GIT? What are the main steps in the digestive process? Ingestion intake of food via the
BPK 312 Nutrition for Fitness & Sport. Lecture 2. Digestion & Absorption of Food Nutrients
 BPK 312 Nutrition for Fitness & Sport Lecture 2 Digestion & Absorption of Food Nutrients 1. Overview of digestion & absorption of nutrients 2. Functional anatomy of the gastrointestinal (GI) tract 3. Digestion
BPK 312 Nutrition for Fitness & Sport Lecture 2 Digestion & Absorption of Food Nutrients 1. Overview of digestion & absorption of nutrients 2. Functional anatomy of the gastrointestinal (GI) tract 3. Digestion
Energy, Chemical Reactions and Enzymes
 Phosphorylation Hydrolysis Energy, Chemical Reactions and Enzymes Chapter 2 (selections) What is Energy? Energy is the capacity to do work Potential Energy Kinetic Energy Chemical Bond Energy Like a rechargeable
Phosphorylation Hydrolysis Energy, Chemical Reactions and Enzymes Chapter 2 (selections) What is Energy? Energy is the capacity to do work Potential Energy Kinetic Energy Chemical Bond Energy Like a rechargeable
The Digestive System. Chapter 25
 The Digestive System Chapter 25 Introduction Structure of the digestive system A tube that extends from mouth to anus Accessory organs are attached Functions include Ingestion Movement Digestion Absorption
The Digestive System Chapter 25 Introduction Structure of the digestive system A tube that extends from mouth to anus Accessory organs are attached Functions include Ingestion Movement Digestion Absorption
Biology 20: Digestive System Did you get it? Questions and Answers
 Biology 20: Digestive System Did you get it? Questions and Answers Mouth 1. What is the purpose of chewing? a. break food into smaller pieces so there is a greater SA for chemicals to work on. 2. Salivary
Biology 20: Digestive System Did you get it? Questions and Answers Mouth 1. What is the purpose of chewing? a. break food into smaller pieces so there is a greater SA for chemicals to work on. 2. Salivary
Effect of Ileo-Jejunal Intestinal Adaptation
 Tohoku J. exp. Med., 1987, 151, 419-428 Effect of Ileo-Jejunal Intestinal Adaptation Colectomy in Dogs Transposition after Total on IWAO SASAKI, TAKASHI TUCHIYA, HIROO NAITO, YUJI FUNAYAMA, MORIHIKO TODA,
Tohoku J. exp. Med., 1987, 151, 419-428 Effect of Ileo-Jejunal Intestinal Adaptation Colectomy in Dogs Transposition after Total on IWAO SASAKI, TAKASHI TUCHIYA, HIROO NAITO, YUJI FUNAYAMA, MORIHIKO TODA,
INFLUENCE OF INTRAJEJUNAL GLUCOSE ON PANCREATIC EXOCRINE FUNCTION IN MAN
 GASTROENTEROLOGY Copyright @ 1971 by The Williams & Wilkins Co. Vol. 60, No.5 Printed in U.S.A. INFLUENCE OF INTRAJEJUNAL GLUCOSE ON PANCREATIC EXOCRINE FUNCTION IN MAN WALTER P, DYCK, M.D. Department
GASTROENTEROLOGY Copyright @ 1971 by The Williams & Wilkins Co. Vol. 60, No.5 Printed in U.S.A. INFLUENCE OF INTRAJEJUNAL GLUCOSE ON PANCREATIC EXOCRINE FUNCTION IN MAN WALTER P, DYCK, M.D. Department
Gastric, intestinal and colonic absorption of metoprolol in
 Br. J. clin. Pharmac. (1985), 19, 85S-89S Gastric, intestinal and colonic absorption of metoprolol in the rat J. DOMENECH', M. ALBA', J. M. MORERA', R. OBACH' & J. M. PLA DELFINA2 'Department of Pharmaceutics,
Br. J. clin. Pharmac. (1985), 19, 85S-89S Gastric, intestinal and colonic absorption of metoprolol in the rat J. DOMENECH', M. ALBA', J. M. MORERA', R. OBACH' & J. M. PLA DELFINA2 'Department of Pharmaceutics,
1) Four main feeding mechanisms of animals a) Suspension feeders i) (1) Humpback whales b) Substrate feeders i)
 1 AP Biology March 2008 Digestion Chapter 41 Homeostatic mechanisms manage an animal s energy budget. 1) Four main feeding mechanisms of animals Suspension feeders (1) Humpback whales Substrate feeders
1 AP Biology March 2008 Digestion Chapter 41 Homeostatic mechanisms manage an animal s energy budget. 1) Four main feeding mechanisms of animals Suspension feeders (1) Humpback whales Substrate feeders
(GH-RIH), which were shown to inhibit the release of CCK induced by. (OP-CCK) as well as a small rise in bicarbonate output attaining a peak
 J. Phyaiol. (1976), 257, pp. 663-672 663 With 5 text-ftgure8 Printed in Great Britain EFFECT OF BOMBESIN AND RELATED PEPTIDES ON THE RELEASE AND ACTION OF INTESTINAL HORMONES ON PANCREATIC SECRETION By
J. Phyaiol. (1976), 257, pp. 663-672 663 With 5 text-ftgure8 Printed in Great Britain EFFECT OF BOMBESIN AND RELATED PEPTIDES ON THE RELEASE AND ACTION OF INTESTINAL HORMONES ON PANCREATIC SECRETION By
BIO 139 ANATOMY AND PHYSIOLOGY II THE DIGESTIVE SYSTEM
 BIO 139 ANATOMY AND PHYSIOLOGY II THE DIGESTIVE SYSTEM MARY CATHERINE FLATH, Ph.D. DIGESTIVE SYSTEM ORGANS MOUTH PHARYNX ESOPHAGUS STOMACH SMALL INTESTINE LARGE INTESTINE SALIVARY GLANDS PANCREAS LIVER
BIO 139 ANATOMY AND PHYSIOLOGY II THE DIGESTIVE SYSTEM MARY CATHERINE FLATH, Ph.D. DIGESTIVE SYSTEM ORGANS MOUTH PHARYNX ESOPHAGUS STOMACH SMALL INTESTINE LARGE INTESTINE SALIVARY GLANDS PANCREAS LIVER
NCERT MULTIPLE CHOICE QUESTIONS
 91 CHAPTER 16 DIGESTION AND ABSORPTION MULTIPLE CHOICE QUESTIONS 1. Select what is not true of intestinal villi among followings a. They possess microvilli b. They increase the surface area c. They are
91 CHAPTER 16 DIGESTION AND ABSORPTION MULTIPLE CHOICE QUESTIONS 1. Select what is not true of intestinal villi among followings a. They possess microvilli b. They increase the surface area c. They are
Chapter 20 The Digestive System Exam Study Questions
 Chapter 20 The Digestive System Exam Study Questions 20.1 Overview of GI Processes 1. Describe the functions of digestive system. 2. List and define the four GI Processes: 20.2 Functional Anatomy of the
Chapter 20 The Digestive System Exam Study Questions 20.1 Overview of GI Processes 1. Describe the functions of digestive system. 2. List and define the four GI Processes: 20.2 Functional Anatomy of the
Serum gastrin and gastric acid responses to meals at various ph levels in man
 Gut, 1974, 15, 526-530 Serum gastrin and gastric acid responses to meals at various ph levels in man S. J. KONTURK,1 J. BIRNAT, AND J. OLKSY From the Institute ofphysiology, Medical Academy, Krak6w, Poland,
Gut, 1974, 15, 526-530 Serum gastrin and gastric acid responses to meals at various ph levels in man S. J. KONTURK,1 J. BIRNAT, AND J. OLKSY From the Institute ofphysiology, Medical Academy, Krak6w, Poland,
The Small Intestine. The pyloric sphincter at the bottom of the stomach opens, squirting small amounts of food into your small intestine.
 The Small Intestine The pyloric sphincter at the bottom of the stomach opens, squirting small amounts of food into your small intestine. approximately six metres (the longest section of your digestive
The Small Intestine The pyloric sphincter at the bottom of the stomach opens, squirting small amounts of food into your small intestine. approximately six metres (the longest section of your digestive
5. Which component of the duodenal contents entering the stomach causes the most severe changes to gastric mucosa:
 Gastro-intestinal disorders 1. Which are the most common causes of chronic gastritis? 1. Toxic substances 2. Chronic stress 3. Alimentary factors 4. Endogenous noxious stimuli 5. Genetic factors 2. Chronic
Gastro-intestinal disorders 1. Which are the most common causes of chronic gastritis? 1. Toxic substances 2. Chronic stress 3. Alimentary factors 4. Endogenous noxious stimuli 5. Genetic factors 2. Chronic
The gallbladder. Bile secretion:
 The gallbladder is a thin walled green muscular sac on the inferior surface of the liver. The gallbladder stores bile that is not immediately needed for digestion and concentrates it. When the muscular
The gallbladder is a thin walled green muscular sac on the inferior surface of the liver. The gallbladder stores bile that is not immediately needed for digestion and concentrates it. When the muscular
Using a technique by which it is possible to study gastro-intestinal absorption
 531 J. Physiol. (I956) I34, 53I-537 THE ABSORPTION OF GLUCOSE BY THE INTACT RAT BY P. C. REYNELL AND G. H. SPRAY From the Nuffield Department of Clinical Medicine, University of Oxford (Received 30 May
531 J. Physiol. (I956) I34, 53I-537 THE ABSORPTION OF GLUCOSE BY THE INTACT RAT BY P. C. REYNELL AND G. H. SPRAY From the Nuffield Department of Clinical Medicine, University of Oxford (Received 30 May
EFFECT OF SECRETIN AND CHOLECYSTOKININ ON GASTRIC EMPTYING AND GASTRIC SECRETION IN MAN
 GA8TRONTROLOGY Copyright 197 by The Williams & Wilkins Co. Vol. 58, No.6 Fdnted in U.S.A. FFCT OF SCRTIN AND CHOLCYSTOKININ ON GASTRIC MPTYING AND GASTRIC SCRTION IN MAN W. Y. CHY, M.D., S. HITANANT, M.D.,
GA8TRONTROLOGY Copyright 197 by The Williams & Wilkins Co. Vol. 58, No.6 Fdnted in U.S.A. FFCT OF SCRTIN AND CHOLCYSTOKININ ON GASTRIC MPTYING AND GASTRIC SCRTION IN MAN W. Y. CHY, M.D., S. HITANANT, M.D.,
Overview. Physiology 1. The Gastrointestinal Tract. Guyton section XI
 Overview Physiology 1 The Gastrointestinal Tract Guyton section XI Basic functions of the GI tract Digestion Secretion Absorption Motility Basic functions of the GI tract Digestion: : Dissolving and breaking
Overview Physiology 1 The Gastrointestinal Tract Guyton section XI Basic functions of the GI tract Digestion Secretion Absorption Motility Basic functions of the GI tract Digestion: : Dissolving and breaking
Short Bowel Syndrome: Medical management
 Short Bowel Syndrome: Medical management La Sindrome dell'intestino Corto in età pediatrica Brescia 18 marzo 2011 Jon A.Vanderhoof, M.D. Division of Pediatric GI Harvard Medical School Children s Hospital,
Short Bowel Syndrome: Medical management La Sindrome dell'intestino Corto in età pediatrica Brescia 18 marzo 2011 Jon A.Vanderhoof, M.D. Division of Pediatric GI Harvard Medical School Children s Hospital,
PEPSIN SECRETION DURING DAMAGE BY ETHANOL AND SALICYLIC ACID
 GASTROENTEROLOGY Copyriht 1972 by The Williams & Wilkins Co. Vol. 62. No. 3 Printed in U.S. A. PEPSIN SECRETION DURING DAMAGE BY ETHANOL AND SALICYLIC ACID LEONARD R. JOHNSON, PH.D. Department of Physiology
GASTROENTEROLOGY Copyriht 1972 by The Williams & Wilkins Co. Vol. 62. No. 3 Printed in U.S. A. PEPSIN SECRETION DURING DAMAGE BY ETHANOL AND SALICYLIC ACID LEONARD R. JOHNSON, PH.D. Department of Physiology
Experimental Acute Pancreatitis in Mice
 GASTROENTEROLOGY 76:529-534, 1979 Experimental Acute Pancreatitis in Mice Protective Effects of Glucagon T ADAO MANABE, M.D., and MICHAEL L. STEER, M.D. Department of Surgery, Beth Israel Hospital, and
GASTROENTEROLOGY 76:529-534, 1979 Experimental Acute Pancreatitis in Mice Protective Effects of Glucagon T ADAO MANABE, M.D., and MICHAEL L. STEER, M.D. Department of Surgery, Beth Israel Hospital, and
Biology 12 - Digestion Notes
 Biology 12 - Digestion Notes Anatomy Physiology Functions of the Digestive System -------------------------------------------------------------------------------------- food (enzymes, bile, HCl) to assist
Biology 12 - Digestion Notes Anatomy Physiology Functions of the Digestive System -------------------------------------------------------------------------------------- food (enzymes, bile, HCl) to assist
EFFECT OF CARBENOXOLONE ON THE GASTRIC MUCOSAL BARRIER IN MAN AFTER ADMINISTRATION OF TAUROCHOLIC ACID
 GASTROENTEROLOGY 64: 1101-1105, 1973 Copyright 1973 by The Williams & Wilkins Co. Vol. 64 No.6 Printed in U.S.A. EFFECT OF CARBENOXOLONE ON THE GASTRIC MUCOSAL BARRIER IN MAN AFTER ADMINISTRATION OF TAUROCHOLIC
GASTROENTEROLOGY 64: 1101-1105, 1973 Copyright 1973 by The Williams & Wilkins Co. Vol. 64 No.6 Printed in U.S.A. EFFECT OF CARBENOXOLONE ON THE GASTRIC MUCOSAL BARRIER IN MAN AFTER ADMINISTRATION OF TAUROCHOLIC
ALTERED PANCREATIC AND BILIARY FUNCTION AFTER VAGOTOMY AND PYLOROPLASTY
 GASTROENTEROLOGY 66: 22-27, 1974 Copyright 1974 by The Williams & Wilkins Co. Vol. 66, No.1 Printed in U.S.A. ALTERED PANCREATIC AND BILIARY FUNCTION AFTER VAGOTOMY AND PYLOROPLASTY JUAN R. MALAGELADA,
GASTROENTEROLOGY 66: 22-27, 1974 Copyright 1974 by The Williams & Wilkins Co. Vol. 66, No.1 Printed in U.S.A. ALTERED PANCREATIC AND BILIARY FUNCTION AFTER VAGOTOMY AND PYLOROPLASTY JUAN R. MALAGELADA,
Why would fatty foods aggravate the patient s RUQ pain? What effect does cholecystokinin (CCK) have on gastric emptying?
 CASE 28 A 43-year-old woman presents to the emergency department with the acute onset of abdominal pain. Her pain is located to the right upper quadrant (RUQ) and radiates to the right shoulder. She reports
CASE 28 A 43-year-old woman presents to the emergency department with the acute onset of abdominal pain. Her pain is located to the right upper quadrant (RUQ) and radiates to the right shoulder. She reports
2. SECRETIONS OF THE DIGESTIVE TRACT
 2. SECRETIONS OF THE DIGESTIVE TRACT SECRETORY GLANDS AND CELLS The alimentary tract produces a large variety and quantity of substances which contribute to digesting the food, and protecting and regulating
2. SECRETIONS OF THE DIGESTIVE TRACT SECRETORY GLANDS AND CELLS The alimentary tract produces a large variety and quantity of substances which contribute to digesting the food, and protecting and regulating
Nutritional assessments and diagnosis of digestive disorders
 Nutritional assessments and diagnosis of digestive disorders AASER ABDELAZIM Assistant professor of Medical Biochemistry Zagazig University, Egypt University of Bisha, KSA aaserabdelazim@yahoo.com 7 Mal
Nutritional assessments and diagnosis of digestive disorders AASER ABDELAZIM Assistant professor of Medical Biochemistry Zagazig University, Egypt University of Bisha, KSA aaserabdelazim@yahoo.com 7 Mal
Augmentation of Cysteamine and Mepirizole-Induced Lesions in the Rat Duodenum and Stomach by Histamine or Indomethacin
 Augmentation of Cysteamine and Mepirizole-Induced Lesions in the Rat Duodenum and Stomach by Histamine or Indomethacin Hironori TANAKA, Yoshimi KUWAHARA and Susumu OKABE Department of Applied Pharmacology,
Augmentation of Cysteamine and Mepirizole-Induced Lesions in the Rat Duodenum and Stomach by Histamine or Indomethacin Hironori TANAKA, Yoshimi KUWAHARA and Susumu OKABE Department of Applied Pharmacology,
THE DIGESTIVE SYSTEM
 THE DIGESTIVE SYSTEM Composed of two parts: 1. 2. There are 4 main parts of digestion: 1. Ingestion: 2. Digestion: a. Mechanical Digestion: Example: b. Chemical Digestion: Example: 3. Absorption: 4. Egestion:
THE DIGESTIVE SYSTEM Composed of two parts: 1. 2. There are 4 main parts of digestion: 1. Ingestion: 2. Digestion: a. Mechanical Digestion: Example: b. Chemical Digestion: Example: 3. Absorption: 4. Egestion:
2. SECRETIONS OF THE DIGESTIVE TRACT
 2. SECRETIONS OF THE DIGESTIVE TRACT SECRETORY GLANDS AND CELLS The alimentary tract produces a large variety and quantity of substances which contribute to digesting the food, and protecting and regulating
2. SECRETIONS OF THE DIGESTIVE TRACT SECRETORY GLANDS AND CELLS The alimentary tract produces a large variety and quantity of substances which contribute to digesting the food, and protecting and regulating
Gut hormone release after intestinal resection
 Gut, 1982, 23, 854-861 Gut hormone release after intestinal resection H S BESTERMAN,* T E ADRIAN, C N MALLINSON, N D CHRISTOFIDES, D L SARSON, A PERA, L LOMBARDO, R MODIGLIANI, and S R BLOOM From the Department
Gut, 1982, 23, 854-861 Gut hormone release after intestinal resection H S BESTERMAN,* T E ADRIAN, C N MALLINSON, N D CHRISTOFIDES, D L SARSON, A PERA, L LOMBARDO, R MODIGLIANI, and S R BLOOM From the Department
ABDOMEN - GI. Duodenum
 TALA SALEH ABDOMEN - GI Duodenum - Notice the shape of the duodenum, it looks like capital G shape tube which extends from the pyloroduodenal junction to the duodenojejunal junction. - It is 10 inches
TALA SALEH ABDOMEN - GI Duodenum - Notice the shape of the duodenum, it looks like capital G shape tube which extends from the pyloroduodenal junction to the duodenojejunal junction. - It is 10 inches
