manner to enable rapid recruitment of cells into the tissue. The mucosal vascular addressin (MAdCAM- 1 ) ' and the
|
|
|
- Teresa Robertson
- 5 years ago
- Views:
Transcription
1 Vascular Addressins Are Induced on Islet Vessels during Insulitis in Nonobese Diabetic Mice and Are Involved in Lymphoid Cell Binding to Islet Endothelium Arno Hanninen,** Cariel Taylor,' Philip R. Streeter,11 Leslie S. Stark,11 Jaime M. Sarte,11 Judith A. Shizuru,' Olli Simell,t and Sara A. Michielil *National Public Health Institute; tdepartment of Pediatrics, University of Turku, Turku, Finland; Department of Internal Medicine and IlDepartment of Pathology, Stanford University School ofmedicine, Stanford, California 94305; and 'Department of Veterans Affairs, Centerfor Molecular Biology in Medicine, Palo Alto, California Abstract In the nonobese diabetic (NOD) mouse, lymphocytic and monocytic infiltration of the pancreatic islets leads to beta cell destruction. To investigate the mechanisms by which lymphocytes enter the NOD pancreas, pancreata were immunostained using monoclonal antibodies to a variety of adhesion molecules known to be involved in lymphocyte binding to vascular endothelium, an initial step in the migration of lymphocytes from blood into organized lymphoid and inflamed tissues. These adhesion molecules include: lymphocyte homing receptors involved in tissue-selective binding of lymphocytes to peripheral lymph node (L-selectin) or mucosal lymphoid tissue (LPAM-1, a437- integrin) high-endothelial venules (HEV); and HEV ligands peripheral vascular addressin (PNAd) and mucosal vascular addressin (MAdCAM-1). In NOD pancreata, a4#7 is expressed on most infiltrating cells at all stages of insulitis, whereas L-selectin expression is more pronounced on cells in the islets at later stages. During the development of insulitis, MAdCAM-1 and to a lesser extent PNAd became detectable on vascular endothelium adjacent to and within the inflamed islets. The Stamper-Woodruff in vitro assay was used to examine lymphoid cell binding to such vessels. These functional assays show that both the mucosal (MAdCAM-1 /a47) and the peripheral (PNAd/L-selectin) recognition systems are involved in this binding. Our findings demonstrate that expression of peripheral and mucosal vascular addressins is induced on endothelium in inflamed islets in NOD pancreas, and that these addressins participate in binding lymphoid cells via their homing receptors. This suggests that these adhesion molecules play a role in the pathogenesis of diabetes in these mice by being involved in the migration of lymphocytes from blood into the inflamed pancreas. (J. Clin. Invest : ) Key words: insulin-dependent diabetes * NOD mouse * adhesion molecules * inflammation * insulitis Introduction Extravasation of lymphocytes from the circulation into organized lymphoid tissues and extralymphoid sites is essential in normal immune surveillance as well as in the accumulation of lymphocytes in nonlymphoid tissues during inflammation ( 1- Address correspondence to Dr. Sara A. Michie, Department of Veterans Affairs, Center for Molecular Biology in Medicine, 3801 Miranda Avenue, Palo Alto, CA Receivedfor publication 7 April 1993 and in revisedform 24 June The Journal of Clinical Investigation, Inc. Volume 92, November 1993, ). This extravasation is currently viewed as a multistep process involving recognition and specific interaction of several adhesion molecules on the lymphocyte with their endothelial ligands (6, 7). Such pairing of adhesion molecules is necessary to enable the initial binding of the lymphocyte to the endothelial surface, and at each following step leading to lymphocyte migration through the vessel endothelial cell layer into the tissue. Lymphocyte subpopulations use different but overlapping sets of receptors (7), which in physiologic conditions interact only with their corresponding ligands on endothelial cells. Certain endothelial ligands are normally expressed in a tissue-selective manner so that their interaction with lymphocyte adhesion molecules plays a key role in the tissue-selective homing of lymphocytes (8, 9). Other endothelial ligands can be expressed in a wide variety of normal or inflamed tissues ( 10, 11 ). In such situations, these receptors may be used in a non-organ-specific manner to enable rapid recruitment of cells into the tissue. The mucosal vascular addressin (MAdCAM- 1 ) ' and the peripheral vascular addressin (PNAd) are adhesion molecules preferentially expressed on endothelial cells ofhigh-endothelial venules (HEVs) in mucosal organized lymphoid tissues such as Peyer's patches (PP) or on HEVs of peripheral lymph nodes (PLN), respectively (8, 9). CD54 (ICAM- I) is a non-organspecific adhesion molecule which is constitutively expressed on endothelium at low levels, but is increasingly expressed on endothelial cells and other cell types after activation by inflammatory mediators ( 12). Vascular cell adhesion molecule- 1 (VCAM- 1 ) is also found on activated endothelia in a variety of tissues although some expression has been noted on endothelium from histologically normal tissues ( 13). Antigens expressed on lymphocytes and associated with their homing in a tissue-selective manner to organized lymphoid tissues are known as homing receptors (HR). L-Selectin is a peripheral HR that is essential for lymphocyte homing to PLN but not to mucosal tissues (14). PNAd comprises the surface molecules presenting PLN HEVselective carbohydrate ligands to lymphocyte L-selectin ( 15). The a43.7 integrin, previously termed LPAM- 1 or a4fp, is a mucosal HR that interacts with MAdCAM- 1 ( 16, 17). CDl la/cd 1 8 (LFA- 1) is a lymphocyte receptor for CD54. It is present on all leukocytes and has a non-organ-specific function in lymphocyte-endothelial adhesion ( 18 ). To investigate the possible role of adhesion molecules on lymphocytes and vascular endothelial cells in the migration of 1. Abbreviations used in this paper: HEV, high endothelial venule; HR, homing receptor; ICAM-1, intercellular adhesion molecule-i; MAd- CAM-1, mucosal vascular addressin; MLN, mesenteric lymph node; PLN, peripheral lymph node; PNAd, peripheral vascular addressin; PP, Peyer's patch; VCAM-1, vascular cell adhesion molecule- 1. Vascular Addressins and Homing Receptors in NOD Pancreas 2509
2 lymphocytes from blood to pancreas and subsequently in the formation of islet infiltrates, we followed the expression of these murine adhesion molecules on inflammatory cells and on endothelium in the pancreases of NOD mice from the age when the mononuclear infiltrates are first detected around islets until the age when advanced insulitis and hyperglycemia occur. The functional status of the adhesion molecules found on lymphocytes and endothelial cells in inflamed islets was determined using an in vitro assay of lymphoid cell-endothelial cell binding. Methods Mice. Mice were examined from two NOD colonies. Both colonies are housed under pathogen-free conditions and fed with normal mouse chow. The Turku colony of NOD mice, bred from animals purchased from BomholtgArd, Ry, Denmark, has a cumulative incidence of diabetes by the age of 25 wk of 80% in females and 40% in males. The - incidence of diabetes in the Stanford colony is 85% in females and 45% in males at 25 wk. Age-matched BALB/c and SJL mice were used as controls. Monoclonal antibodies. mabs and MECA-79 (provided by E. Butcher, Stanford University) were used to detect MAd- CAM- I and PNAd, respectively. MECA-79 reacts with a PNAd carbohydrate-associated epitope, and is a sensitive reagent for immunohistologic assessment of HEV PNAd expression. mabs MEL-14 (anti-l-selectin), FD441.8 (anti-cd1 la), and M18/2.a.8 (anti- CD 18) were grown in our laboratory from cells obtained from the American Type Culture Collection (Rockville, MD). ac4f, was detected with mabs specific for the a4 chain (clone R 1-2 provided by I. Weissman (Stanford University) and also purchased from PharMingen, San Diego, CA; PS/2 provided by P. Kincade, University ofoklahoma, Norman, OK) as well as with mabs that react with the 37 chain (722, provided by E. Sikorski and E. Butcher, Stanford University; DATK-32, which specifically reacts only with the (37 chain of a4#7 integrin, provided by D. Andrew, Stanford University). MK2.6 (anti- VCAM-1) was provided by P. Kincade while 3E2 (anti-cd54) was purchased from PharMingen. The following negative control mabs were substituted for species- and isotype-matched anti-hr and antiaddressin mab in in vitro functional assays and in immunohistochemical stainings: (a) (reacts with an epitope on MAdCAM-1 not involved in lymphocyte binding; used as negative control for MECA- 367 in the in vitro assays); (b) IG (reacts with retroviral gp9o; negative control for PS/2 and DATK-32); (c) 9B5 (reacts with human L-selectin; negative control for MEL-14); and (d) OZ-42 (reacts with an antigen in developing cerebellum of mouse; negative control for MECA-79). Immunologic staining of pancreata. Pancreata were taken from NOD and control mice of both sexes at the ages of 3, 4, 5, 6, 8, and wk, and from newly diabetic NOD mice and age-matched controls. The pancreata were snap-frozen in liquid nitrogen and were stored at -70 C. Serial frozen sections were cut, dried, fixed in cold acetone for 5 min, and stained by indirect immunofluorescence. The sections were first incubated with mab, followed by FITC-labeled secondary antibody in 5% normal mouse serum (goat anti-rat IgG; Cappel, Durham, NC; or goat anti-hamster Fab'2, Caltag, South San Francisco, CA for mab 3E2), next with polyclonal guinea pig anti-porcine insulin (Accurate Chem. & Sci. Corp., Westbury, NY), and then with Texas red-conjugated goat anti-guinea pig IgG (Accurate Chem.). All incubations were for 30 min at room temperature followed by three washes in PBS. At least five sections from different levels ofa pancreas were stained with each mab. After staining, sections were mounted in glycerol based medium and viewed by fluorescence microscopy. In some cases, pancreata were immunostained using a three stage immunoperoxidase technique as described, and then were examined by light microscopy (9). In vitro lymphocyte-endothelial cell binding assay. The Stamper- Woodruff in vitro binding assay was performed as described but with slight modifications for this study ( 19, 20). Briefly, 8- and 12-wk-old NOD and BALB/c mice were injected intravenously with 0.2 ml of the intravascular dye Luconyl blue (40% in saline) (BASF, Ludwigshafen, Germany) and killed 5 min later. NOD pancreata and BALB/c organized lymphoid tissues (PLN and PP) were removed and snap-frozen. Cell suspensions were prepared from BALB/c MLN (mesenteric lymph node) or from one of the following lymphoma cell lines that exhibit tissue-selective binding to HEV in organized lymphoid tissues: 38C1 3, a C3H B cell lymphoma that preferentially binds to PLN over mucosal HEV, using an L-selectin/PNAd recognition system (9, 14, 21 ); or TK 1, an AKR T cell lymphoma which expresses ca437 but not a!41 (VLA-4) and preferentially binds to mucosal over PLN HEV via an a437/madcam-l system (21, 22, 23). To test the ability of mabs that react with lymphocyte HR or vascular addressins to interfere with the binding oflymphoid cells to vascular endothelium in inflamed islets in the NOD pancreas, either lymphoid cells or pancreas tissue sections were preincubated with specific mab, isotype-matched negative control mab, or media immediately prior to the binding assay as described ( 19). Tissue sections were then incubated with 1-3 X 106 lymphoid cells in 0.1 ml DMEM for 30 min at 7 C with mild rotation. Nonadherent cells were decanted, and adherent cells were fixed to tissue sections by 1.5% glutaraldehyde in PBS. Cell binding to vessels was evaluated microscopically under phase or darkfield illumination. Vessels were identified by intravascular localization of Luconyl blue, and their microenvironmental location determined by examination of adjacent sections stained with hematoxylin and eosin or with antiaddressin mabs using an immunoperoxidase technique. For each pancreas, lymphoid cell binding to vessels in or adjacent to islets was counted only if the vessel could be evaluated on serial sections from the assay treated with specific mab, negative control mab, and media. The mean number of cells bound per vessel for each mab was determined for pancreas and organized lymphoid tissues from each mouse. These results for each animal are presented as a percentage of media control. Results Non-organ-specific adhesion molecules on inflammatory cells and endothelium. There was strong uniform expression of CD1 la/cd18 (LFA-1) and CD54 (ICAM-1) by all mononuclear cells that had infiltrated the NOD pancreas. CD54 was also expressed on endothelium of pancreatic vessels in young healthy NOD mice and mice of control strains as well as in prediabetic and diabetic NOD mice. During the development ofdisease in NOD mice, no conspicious increase in intensity of CD54 expression on endothelial cells was observed (data not shown). Endocrine cells themselves were negative for CD54. VCAM- 1 was expressed on endothelium in a subset of pancreatic vessels in BALB/c and NOD mice. In both strains, most (> 97%) of the VCAM- 1 expressing vessels were in the exocrine pancreas. These ranged in appearance from small, flatwalled vessels scattered among the acini to larger veins and arteries adjacent to the interlobular ducts. Occasional small vessels adjacent to uninflamed islets in NOD and BALB/c mice were VCAM- 1 positive as were a few vessels within NOD inflamed islets. Induction of vascular addressins on islet endothelium. There was no expression of PNAd by endothelium in pancreata of control mice (BALB/c and SJL) or in noninflamed pancreata from young, healthy NOD mice. In contrast, a low constitutive expression of MAdCAM- 1 was observed on endothelial cells ofa few small, flat-walled vessels scattered throughout the exocrine pancreas in all mice (Fig. 1 A), but not on endothelium within the islets. During the development of islet infiltrates, MAdCAM-l and PNAd became expressed on islet endothelium. These ad Hdnninen, Taylor, Streeter, Stark, Sarte, Shizuru, Simell, and Michie
3 a be.. b~~~~~~~~~~~~~~..i:.. I fx~~~~r4 'A.1. * 1<3 i C0Ad Fff~~s \wi S;-^Xta4 4A Av * 0 r Of _-~~~~~~~~~~~fi..,a A, - I N s~~~~~~~~~~~. 1 dp.6 vft. X.. Figure 1. Vascular addressin expression in pancreas. (a) MAdCAM-l is expressed by vessels (arrowheads) in the exocrine pancreas of a 60-d-old female BALB/c mouse. Similar expression was noted in exocrine pancreas of all SJL and NOD mice examined (immunoperoxidase stain with mab ; methylene blue counterstain; X300). (b) Early insulitis in 60-d-old female NOD mouse. Two islets (each outlined by arrowheads) with an intervening lymphocyte infiltrate surrounding several MAdCAM-1-expressing vessels (one vessel highlighted by arrow) (immunoperoxidase stain with mab MECA- 367; X150). (c) Section through an islet with severe insulitis from 90-d-old female NOD mouse. The inflamed islet contains several PNAd-expressing vessels, two of which (arrows) show high endothelial morphology (immunoperoxidase with mab MECA-79; x400). dressins, first detected on endothelium within and adjacent to islets in 4-5-wk-old NOD mice, appeared simultaneously with mononuclear cell infiltrates of these islets. As the degree of insulitis became more severe, small vessels expressing these molecules became more numerous in islets (Fig. 1, b and c). In addition, some ofthese vessels developed high endothelial morphology and stained with antiaddressin mab with an intensity similar to that of HEV in organized lymphoid tissues. Without exception, addressin expression within the islets was limited to vessels that were adjacent to or surrounded by inflammatory cells. In addition, MAdCAM- I was expressed in the periphery of some islets with periislet infiltration (periinsulitis), but then its intensity was weak. The overall expression of MAdCAM- 1 was similar to PNAd within infiltrated islets of 12 wk and older NOD mice, with many vessels coexpressing both addressing. In younger NOD mice, however, more islets vessels expressed MAdCAM- 1 than PNAd (Table I). No significant differences in addressin expression were found between pancreata of female and male NOD mice. For example, among the 8-wk-old NOD mice from the Stanford colony (Table I), the percentage of islets expressing MAdCAM- I or PNAd was 40 or 24% in the females, and 44 or 29%, respectively in the males. Expression ofhr on infiltrating lymphocytes. The expression of peripheral (L-selectin) and mucosal (a4,87) HR by lymphocytes in and around inflamed islets was evaluated. The L- selectin peripheral HR was expressed by a minority ofperi- and intraislet lymphocytes present during periinsulitis and early insulitis (Fig. 2 a), whereas strong a4,f7 expression as detected by staining with mabs Rl-2 (anti-a4) and 722 (anti-f37) was evident on the first cells that gathered in periislet locations as well as on lymphocytes that infiltrated the islets throughout all stages of insulitis (Fig. 2, b and c). At more advanced stages of insulitis, L-selectin expression intensified among condensed groups of cells within the inflammatory plaque. Hence, the expression of HR on infiltrating cells shifted from mucosal HR predominance to mixed expression ofboth HR during progression of insulitis (Table IL). In vitro lymphocyte-endothelial cell binding. The Stamper- Woodruffin vitro assay was used to study the specific adhesion pathways mediating interactions between lymphoid cells and vascular endothelium of inflamed islets in the NOD pancreas. Our initial assays used lymphocytes obtained from MLN of young adult BALB/c mice. These lymphocytes bound to endothelium of blood vessels in inflamed islets from 8- and 12-wkold female NOD mice. A large portion of this binding could be blocked with mab directed against MAdCAM- 1 and a smaller portion was blocked with mab against PNAd (Fig. 3 a) (t test, one-sided: P = for versus ; P = for MECA-79 versus OZ-42). Since MLN is known to contain a mixed population of Vascular Addressins and Homing Receptors in NOD Pancreas 2511
4 Table I. Expression of Vascular Addressins on Islet Endothelium ofnod-pancreata during the Development ofinsulitis Islets with Islets with Islets with Age of mice (n) insulitis mucosal addressin peripheral addressin 4wk(1) wk (2) 10±2 7±5 2±3 6 wk (3) 23±10 12±4 5±9 8 wk (5) 25±16 11±10 4±4 8 wk (4)t 47±20 42±13 27±9 12 wk (6) 84±15 56±14 51±26 12 wk (4)t 81±10 74±10 58±12 * In each group, four to six pancreata were examined, but only the data from pancreata with inflammatory changes are included in this table. The mean number of islets per pancreas analyzed for MAd- CAM-l and PNAd expression was 26 (range = and range = 11-59, respectively). * From Stanford NOD colony. All other mice were from Turku NOD colony. lymphocytes, many of which express both L-selectin and a4a7 and thus bind to either PNAd or MAdCAM- 1-expressing vessels, we chose to use lymphoma cell lines with well-characterized tissue-selective binding properties in subsequent assays. This would allow further dissection of the contributions of the peripheral versus mucosal systems in lymphoid cell binding to endothelium in inflamed islets. TK1, which binds well to MAdCAM-l-expressing mucosal HEV via its a4p7 but fails to bind significantly to PLN HEV, bound to many vessels in the inflamed islets of NOD mice. This binding was almost completely inhibited by preincubation of the cells with anti-a4 (mab PS/2) or anti-,37 (mab DATK-32), or by preincubation of the pancreas sections with anti-madcam-l (mab ) (Fig. 3 b). 38C1 3, which binds well to peripheral HEV via L-selectin/PNAd interaction but does not bind significantly to mucosal HEV, also bound to vessels in the inflamed islets. Most of this binding was blocked by mab directed against PNAd (MECA-79) or L-selectin (MEL-14) (Fig. 3 c). Comparison of the sections from the binding assay with adjacent sections stained with MECA-79 or showed that these lymphoma cells bound to vessels a Is ;., 2512 Hanninen, Taylor, Streeter, Stark, Sarte, Shizuru, Simell, and Michie Figure 2. Lymphocyte homing receptor expression in NOD pancreas. (a and b) Semiserial sections of an islet (IS) from 60-d-old male NOD mouse. There is a lymphocytic infiltrate surrounding the islet with a few lymphocytes within the islet. Some of the lymphocytes express L-selectin (one such lymphocyte is identified by arrow in a), while most of the lymphocytes express a4 (b) and f7 (not shown) (immunoperoxidase with mab MEL-14 [a] and R1-2 [b]; 300X). (c) Pancreas from 90-d-old female NOD mouse showing an islet (arrow) completely infiltrated by lymphocytes. The majority of the lymphocytes express f7 (shown) and a4 (not shown) (immunoperoxidase stain with mab 722; X200).
5 ~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~... bearing the appropriate, tissue-specific addressin. Some of these vessels had the histologic appearance of HEVs. Discussion MAdCAM- I and PNAd are endothelial adhesion molecules in the mouse that are preferentially expressed on HEV in organized lymphoid tissues where they mediate tissue-selective homing of certain subsets of lymphocytes (8, 9). In this study, we demonstrate induction of these molecules on islet endothelium during diabetic insulitis in NOD mice. This induction is specific and restricted to endothelial cells within and immediately adjacent to the islets. The initial appearance of these vascular addressins on islet endothelium at the age when insulitis is initiated, the specific localization of addressin-expressing vessels to inflamed (but not uninflamed) islets, and the ability of such vessels to bind lymphocytes in an in vitro assay suggest that these molecules are involved in the regulation of lymphocyte influx into the islets. However, we did not observe strong expression of MAdCAM- 1 or PNAd in any islet prior to the a Tissue MAb BALB c PP MECA-79 BALB c PLN 0Z-42 I ~_ NOD Pancreas MECA-79 OZ-42 MLN cell binding to vessels lic of media control) Table II. Expression of a Mucosa-associated (a4a7) and a Peripheral Lymph Node-associated (L-selectin) Homing Receptor on Inflammatory Cells in the Islets ofpancreata*from NOD Mice Age of mice 4 wk 5 wk 6 wk 8 wk 12 wk LPAM-l L-sel * Expression of a4s7 (staining with mabs Rl-2 and 722) and L-selectin (staining with mab MEL- 14) was evaluated from the same pancreata as in Table I using adjacent sections (+ < 33%; ++ = 34-66%; % of infiltrating cells expressing the HR). influx of cells. Neither did we observe PNAd expression on vascular endothelium in human nonlymphoid tissues such as skin or thyroid without a coexisting inflammatory infiltrate (41). These data suggest that some other endothelial adhesion b Tissue BALB c PP NOD Pancreas MAb DATK-32 PS 2 1G DATK-32 PS 2 1G -H II ---I -- T TKI binding to vessels 1 of media control) C Figure 3. Involvement of homing receptors and vascular ad- Tissue MAb dressins in binding of lymphoid cells to endothelium in inflamed islets of NOD pancreata. Each bar represents MECA-79 mean±sd of lymphoid cell binding to endothelium on tissue sections from 12-wk-old NOD pancreata (n = 3) or BALB/c BALB c OZ-42 organized lymphoid tissues (n = 3) with addition of anti-hr PLN MEL-14 or anti-addressin mab (open bars) or class-matched negative control mab (shaded bars). Numbers of vessels evaluated per 915 mouse for each mab treatment ranged from 6 to 17 per pancreas, 7 to 15 per PP, and 17 to 32 per PLN. (a) Pretreatment MECA-79 of tissue sections with (anti-madcam-l) or OZ l MECA-79 (anti-pnad) blocked most of the binding of MLN NOD lymphocytes to HEV in PP and PLN, respectively These an- Pancreas MEL-14 tibodies partially blocked binding of MLN lymphocytes to sss LLvessels in inflamed islets. (b) As previously described, TKl 911[5 cells bound well to HEV in PP but poorly to HEV in PLN l (cells/pln HEV < 6% of cells/pp HEV; data not shown; see reference 21 ). The binding to PP HEV was inhibited by mab 38C13 binding to vessels (% of media control) reactive with either chain of thea4.,7hr (PS/2, DATK-32) or with MAdCAM- I (). Similar to PP HEV, vessels in inflamed islets bound TKl cells via an a4f7/madcam-l mucosal recognition system. (c) As previously reported, 38C13 lymphoma cells bound selectively to PLN but not to PP HEV (in this assay, cells/pp HEV < 8% of cells/pln HEV; data not shown; see reference 21). The lymphoma binding to PLN HEV and vessels in inflamed NOD pancreas could be inhibited by mab MEL-14 (anti-l-selectin) or MECA-79 (anti-pnad). Vascular Addressins and Homing Receptors in NOD Pancreas 2513
6 molecules could be responsible for initializing lymphocytic infiltration into extralymphoid sites during inflammation and that vascular addressins would be expressed only later on as a consequence of subsequent endothelial activation. Even in that case, the present study suggests that vascular addressins are involved in lymphocyte entry into the islets during sustained islet inflammation, which may be necessary in the pathogenesis of insulin-dependent diabetes. However, in an experimental model of arthritis that is induced by an inflammatory chemical (24), the influx of leukocytes begins simultaneously with activation of endothelial cells showing that in vivo, these phenomena are difficult to observe independently of each other. Accordingly, ifexpression ofaddressin molecules on islet-endothelium is increased prior to histologically detectable infiltration of the islets by mononuclear cells, this is a transient stage followed immediately by an influx of inflammatory cells. The events underlying endothelial activation resulting in induction of endothelial adhesion molecules and increased lymphocyte binding to the endothelium are of prime importance in the accumulation of lymphocytes in inflamed tissues. Not surprisingly, endothelial cells can be activated in vitro by inflammatory mediators (25) and in vivo, by injections of cytokines which cause local changes in endothelial morphology with concomitant influx of cells into tissue (26, 27). Within inflamed tissues, these inflammatory mediators can be released by a variety of cells (4, 7). First, they may be released from circulating leukocytes coming into contact with endothelium. Second, they may be secreted from endothelial cells themselves. Third, they may be secreted by cells that are normally present in that tissue, including parenchymal cells and resident leukocytes. In the spontaneous autoimmune syndrome of the NOD mouse that results in the accumulation of inflammatory cells in the pancreatic islets, none of these possibilities can so far be excluded. In this context, it is interesting to examine the experiments of Hutchings et al. (28). By administration of a mab against CD1 lb/cd 1 8 that blocks the adhesion of macrophages and monocytes to endothelium, they not only prevented the development of diabetes in NOD mice after cell transfer but also prevented most ofthe influx of lymphoid cells into islets. This suggests that macrophages may be directly or indirectly involved in the activation of islet endothelium, which then allows the binding and subsequent extravasation of lymphocytes. MAdCAM- 1 was expressed by some vessels in the exocrine pancreas from all NOD, BALB/c, and SJL mice examined, and was the predominant addressin expressed by NOD islet endothelium at early stages of insulitis. This molecule is expressed by endothelium in mucosal tissues such as Peyer's patches and small intestine lamina propria and is the vascular addressin specifically recognized by the a4f7 HR found on lymphocytes that home to mucosal sites ( 17 ). During embryogenesis, the pancreas develops in mesoduodenum, which is derived from the foregut (29). Hence, it is possible that pancreatic endothelium would differentiate during embryogenesis along with the endothelium in mucosal lymphoid tissues. Alternatively, common environmental factors shared between the adult gut and pancreas might be involved in the expression of MAdCAM- 1 by endothelium in both organs. In another study (42), a lymphoid cell line that originated from islet-infiltrates of pancreas from a diabetic human was found to adhere strongly to mucosal HEV, whereas adherence to PLN HEV was much weaker. In the present study, we found that lymphoid cells infiltrating islets expressed a4#7 from early on, whereas the expression of L-selectin was initially low and increased with time. These findings suggest that normal pancreas as well as pancreas with early insulitis are predominantly involved in the mucosal lymphocyte recirculation pathway. Alternatively, the small number ofcells expressing L-selectin among the cells infiltrating the islets in early insulitis may indicate that the cells are activated (30). This would be consistent with the idea that initially, islets contain many activated cells, either as a result of selective homing of activated cells to the islet or activation of resting lymphocytes once they have entered the islets. The subsequent increase in L-selectin-expressing lymphocytes could be the result of selective migration of L-selectin-expressing cells, or could be due to reexpression of L-selectin on previously activated lymphocytes. Although monocytes can account for some of the L-selectin and a4fi7 integrin expression that was observed, our observations must also reflect HR expression by lymphocytes, since most studies have shown lymphocytes predominate in islet infiltrates (31, 32), as was also confirmed by immunohistochemical assessment of leukocytes in pancreata of our mice (data not shown). In the adhesion cascade leading to extravasation, integrin molecules have an essential role in strengthening the initial adhesion that is often mediated by selectins (7, 33). In an inherited disease (leukocyte adhesion deficiency), patients are unable to generate acute inflammatory responses in tissues because of a genetic error in CD18, resulting in severe impairment of leukocyte extravasation (34). Therefore, the uniform expression of this molecule on inflammatory cells in islet-infiltrates is not unexpected. The expression ofcd54 on all inflammatory cells in the NOD pancreas, however, contrasts to that seen in human diabetic pancreas, where the expression of CD54 seems to be restricted mainly to endothelial cells (35). In further contrast to the pancreas from a diabetic human, no conspicuous differences in CD54 expression on endothelial cells in nondiabetic and diabetic pancreata could be observed. This may reflect differences in local levels of cytokines such as IL- 1, TNF, and y-interferon that induce expression of CD54 on many cell types, or differences in inducibility of CD54 expression in different species (36), or simply an observation bias resulting from colocalization of CD54 on inflammatory cells adjacent to endothelium in inflamed islets. CD54 expression on inflammatory cells may be involved in the pathogenesis of insulitis and beta cell destruction in NOD mice, because it may strengthen adhesion during antigen presentation (37), and as a ligand for CD1 8, it may mediate adhesion between inflammatory cells (38). This may allow the cells to firmly adhere to and interact with each other after extravasation. Recently, injection of anti-cd54 mab into young NOD mice was shown to reduce the incidence of diabetes significantly (39). Consistent with another previous study (40), we did not detect CD54 expression on insulin positive cells, suggesting that the effect of this treatment was not conferred by blocking any antigen presenting function of beta cells. In summary, we have shown there is specific induction of mucosal (MAdCAM- 1) and peripheral (PNAd) vascular addressins on vessels in inflamed islets of NOD mice, and accumulation within the islets of lymphocytes which express the a4f7 mucosal and L-selectin peripheral HR. We have used functional assays to demonstrate that both the mucosal (MAd- CAM-1 /a4fl7) and the peripheral (PNAd/L-selectin) recognition systems are involved in lymphocyte binding to vessels in 2514 Hdnninen, Taylor, Streeter, Stark, Sarte, Shizuru, Simell, and Michie
7 inflamed islets. This is the first demonstration of induction of functional MAdCAM- 1 and PNAd in an extralymphoid site of chronic inflammation. Our results suggest that these vascular addressins and lymphocyte HR, which normally govern lymphocyte traffic to mucosal and peripheral lymphoid organs, are involved in lymphocyte traffic to NOD pancreas. Thus, these adhesion molecules may be expected to play an important role in the development of diabetes in these mice. Acknowledaments We wish to thank D. Andrew, P. Bolt, E. Butcher, B. Englehardt, C. G. Fathman, and S. Jalkanen for thoughtful review of this manuscript. This work was supported by grants from the Foundation for Diabetes Research (Finland), the Pediatric Research Foundation, Turku University Foundation, Orion Corporation Research Foundation, the Department of Veterans Affairs, the National Institutes of Health (Al , DK-39959, GM-37734) and Juvenile Diabetes Foundation International. References 1. Gowans, J. L., and E. J. Knight The route of recirculation of lymphocytes in the rat. Proc. R. Soc. Lond. B. Biol. Sci. 159: Yednock, T. A., and S. D. Rosen Lymphocyte homing. Adv. Immunol. 44: Jalkanen, S., and E. C. Butcher In vitro analysis of the homing properties ofhuman lymphocytes: developmental regulation of functional receptors for high-endothelial venules. Blood. 66: Pober, J. S., and R. S. Cotran The role of endothelial cells in inflammation. Transplantation (Baltimore). 50: Osborn, L Leukocyte adhesion to endothelium in inflammation. Cell. 62: Stoolman, L. M Adhesion molecules controlling lymphocyte migration. Cell. 56: Butcher, E. C Leukocyte-endothelial cell recognition: three (or more) steps to specificity and diversity. Cell. 67: Streeter, P. R., E. L. Berg, B. T. N. Rouse, R. F. Bargatze, and E. C. Butcher A tissue-specific endothelial cell molecule involved in lymphocyte homing. Nature (Lond.). 331: Streeter, P. R., B. T. N. Rouse, and E. C. Butcher Immunohistologic and functional characterization of a vascular addressin involved in lymphocyte homing into peripheral lymph nodes. J. Cell Biol. 107: Spertini, O., F. W. Luscinskas, G. S. Kansas, J. M. Munro, J. D. Griffin, M. A. Gimbrone, and T. F. Tedder Leukocyte adhesion molecule-l ( LAM-I, L-selectin) interacts with an inducible endothelial cell ligand to support leukocyte adhesion. J. Immunol. 147: Elices, M. J., L. Osborn, Y. Takada, C. Crouse, S. Luhowskyj, M. E. Hemler, and R. R. Lobb VCAM- I on activated endothelium interacts with the leukocyte integrin VLA-4 at a site distinct from the VLA-4/fibronectin binding site. Cell. 60: Springer, T. A Adhesion receptors of the immune system. Nature (Lond.). 346: Page, C., M. Rose, M. Yacoub, and R. Pigott Antigenic heterogeneity of vascular endothelium. Am. J. Pathol. 141: Gallatin, W. M., I. L. Weissman, and E. C. Butcher A cell surface molecule involved in organ-specific homing of lymphocytes. Nature (Lond.). 304: Lasky, L. A., M. S. Singer, D. Dowbenko, Y. Imai, W. J. Henzel, C. Grimley, C. Fennie, N. Gillett, S. R. Watson, and S. D. Rosen An endothelial ligand for L-selectin is a novel mucin-like molecule. Cell. 69: Holzmann, B., B. W. McIntyre, and I. L. Weissman Identification of a murine Peyer's patch-specific lymphocyte homing receptor as an integrin molecule with an a chain homologous to human VLA-4a. Cell. 56: Berlin, C., E. L. Berg, M. J. Briskin, D. P. Andrew, P. J. Kilshaw, B. Holzmann, I. L. Weissman, A. Hamann, and E. C. Butcher a4,f7 integrin mediates lymphocyte binding to the mucosal vascular addressin MAdCAM-1. Cell. 74: Hamann, A., D. Jablonski-Westrich, A. Duijvestijn, E. C. Butcher, H. Baisch, R. Harder, and H. G. Thiele Evidence for an accessory role of LFA- 1 in lymphocyte-high endothelium interaction during homing. J. Immunol. 140: Stamper, H. B., Jr., and J. J. Woodruff Lymphocyte homing into lymph nodes. In vitro demonstration of the selective affinity of recirculating lymphocytes for high-endothelial venules. J. Exp. Med. 144: Butcher, E. C., R. G. Scollay, and I. L. Weissman Lymphocyte adherence to high-endothelial venules: characterization of a modified in vitro assay, and examination of the binding of syngeneic and allogeneic lymphocyte populations. J. Immunol. 123: Bargatze, R. F., P. R. Streeter, and E. C. Butcher Expression of low levels of peripheral lymph-node associated vascular addressin in mucosal lymphoid tissues: possible relevance to the dissemination of passaged AKR lymphomas. J. Cell. Biochem. 42: Butcher, E. C., R. G. Scollay, and I. L. Weissman Organ specificity of lymphocyte interaction with organ-specific determinants on high-endothelial venules. Eur. J. Immunol. 10: Ruegg, C., A. A. Postigo, E. E. Sikorski, E. C. Butcher, R. Pytela, and D. J. Erle Role of integrin a487/a4fp in lymphocyte adherence to fibronectin and VCAM-I and in homotypic cell clustering. J. Cell Biol. 117: Graham, P. C., and S. L. Shaman Peroxidase arthritis. II. Lymphoid cell-endothelial interactions during a developing immunologic inflammatory response. Am. J. Pathol. 69: Luscinskas, F. W., A. F. Brock, M. A. Arnaout, and M. A. Gimbrone Endothelial leukocyte adhesion molecule-l-dependent and leukocyte (CD1 1 /CD18)-dependent mechanisms contribute to polymorphonuclear leukocyte adhesion to cytokine-activated human vascular endothelium. J. Immunol. 142: Colditz, I., R. Zwahlen, B. Dewald, and M. Baggiolini In vivo inflammatory activity of neutrophil activating factor, a novel chemotactic peptide derived from human monocytes. Am. J. Pathol. 134: Munro, J. M., J. S. Pober, and R. S. Cotran Tumor necrosis factor and interferon gamma induce distinct patterns of endothelial activation and associated leukocyte accumulation in skin of Pabio anubis. Am. J. Pathol. 135: Hutchings, P., H. Rosen, L. O'Reilly, E. Simpson, S. Gordon, and A. Cooke Transfer of diabetes in mice prevented by blockade of adhesionpromoting receptor on macrophages. Nature (Lond.). 348: Hamilton, W. J., and R. M. H. McMinn Digestive system. In Textbook of Human Anatomy. W. J. Hamilton, editor. Macmillan Press, Ltd., London Basingstoke Jung, T. M., W. M. Gallatin, I. L. Weissman, and M. 0. Dailey Down-regulation of homing receptors after T cell activation. J. Immunol. 141: Miyazaki, A., T. Hanafusa, K. Yamada, J. Miyagawa, H. Fujino-Kurihara, H. Nakajima, K. Nonaka, and S. Tauri Predominance oft lymphocytes in pancreatic islets and spleen of prediabetic non-obese diabetic (NOD) mice: a longitudinal study. Clin. Exp. Immunol. 60: Shimizu, J., S. Ikehara, J. Toki, H. Ohtsuki, R. Yasumizu, and Y. Hamashima Immunohistochemical and ultrastructural studies on beta cell destruction in NOD mouse, an animal model for Type 1 diabetes mellitus. Acta Histochem. Cytochem. 20: Hynes, R Integrins: versatility, modulation and signaling in cell adhesion. Cell. 89: Kishimoto, T. K., R. S. Larson, A. L. Corbi, M. L. Dustin, D. E. Staunton, and T. A. Springer The leukocyte integrins. Adv. Immunol. 46: Hanninen, A., S. Jalkanen, M. Salmi, S. Toikkanen, G. Nikolakaros, and 0. Simell Macrophages, T cell receptor usage and endothelial cell activation in the pancreas at the onset of insulin-dependent diabetes mellitus. J. Clin. Invest. 90: Dustin, M. L., D. E. Staunton, and T. A. Springer Supergene families meet in the immune system. Immunol. Today. 9: Altmann, D. M., N. Hogg, J. Trowsdale, and D. Wilkinson. Cotransfection of ICAM- I and HLA DR reconstitutes human antigen presenting cell function in mouse L cells. Nature (Lond.). 338: Rothlein, R., M. L. Dustin, S. D. Marlin, and T. A. Springer A human intercellular adhesion molecule (ICAM-1) distinct from LFA-1. J. Immunol. 137: Amano, K., R. Yoneda, Y. Tominaga, Y. Hirao, T. Taki, and K. Yokono Association of ICAM-l and LFA- I molecules in the development of diabetes in NOD mice. Diabetes. 41:38a. (Abstr.) 40. O'Reilly, L. A., P. R. Hutchings, P. R. Crocker, E. Simpson, T. Lund, D. Kioussis, F. Takei, J. Baird, and A. Cooke Characterization of pancreatic islet cell infiltrates in NOD mice: effect of cell transfer and transgene expression. Eur. J. Immunol. 21: Michie, S. A., P. R. Streeter, P. A. Bolt, E. C. Butcher, and L. J. Picker The human peripheral lymph node vascular addressin: an inducible endothelial antigen involved in lymphocyte homing. Am J. Pathol. In press. 42. Hanninen. A. M. Salmi, 0. Simell, and S. Jalkanen Endothelial cell-binding properties of lymphocytes infiltrated into human diabetic pancreas. Implications for the pathogenesis of IDDM. Diabetes. In press. Vascular Addressins and Homing Receptors in NOD Pancreas 2515
Sharon S. Evans, Ph.D. Dept. Immunology, RPCI Jan. 29 & Feb 3, 2015
 Adhesion Molecules And Lymphocyte Homing Sharon S. Evans, Ph.D. Dept. Immunology, RPCI Jan. 29 & Feb 3, 2015 Outline General introduction to lymphocyte trafficking homeostatic recirculation vs active recruitment.
Adhesion Molecules And Lymphocyte Homing Sharon S. Evans, Ph.D. Dept. Immunology, RPCI Jan. 29 & Feb 3, 2015 Outline General introduction to lymphocyte trafficking homeostatic recirculation vs active recruitment.
Innate immune cells---- one time migration preprogrammed homing properties
 Innate immune cells---- one time migration preprogrammed homing properties neutrophils---acute inflammation monocytes---subacute/chronic inflammation eosinophils---parasitic or allergic inflammation natural
Innate immune cells---- one time migration preprogrammed homing properties neutrophils---acute inflammation monocytes---subacute/chronic inflammation eosinophils---parasitic or allergic inflammation natural
The Immune Response in Time and Space
 The Immune Response in Time and Space Chapters 14 & 4 Sharon S. Evans, Ph.D. Department of Immunology 845-3421 sharon.evans@roswellpark.org September 18 & 23, 2014 Inflammation Inflammation Complex response
The Immune Response in Time and Space Chapters 14 & 4 Sharon S. Evans, Ph.D. Department of Immunology 845-3421 sharon.evans@roswellpark.org September 18 & 23, 2014 Inflammation Inflammation Complex response
Chapter 3, Part A (Pages 37-45): Leukocyte Migration into Tissues
 Allergy and Immunology Review Corner: Chapter 3, Part A (pages 37-45) of Cellular and Molecular Immunology (Seventh Edition), by Abul K. Abbas, Andrew H. Lichtman and Shiv Pillai. Chapter 3, Part A (Pages
Allergy and Immunology Review Corner: Chapter 3, Part A (pages 37-45) of Cellular and Molecular Immunology (Seventh Edition), by Abul K. Abbas, Andrew H. Lichtman and Shiv Pillai. Chapter 3, Part A (Pages
inflamed extralymphoid tissues
 7 Clin Pathol 1996;49:721-727 Departmnent of Histopathology, University College London Medical School, London J M Munro Division of Nephrology and Department of Medicine, Children's Hospital, the Departmnent
7 Clin Pathol 1996;49:721-727 Departmnent of Histopathology, University College London Medical School, London J M Munro Division of Nephrology and Department of Medicine, Children's Hospital, the Departmnent
The recruitment of leukocytes and plasma proteins from the blood to sites of infection and tissue injury is called inflammation
 The migration of a particular type of leukocyte into a restricted type of tissue, or a tissue with an ongoing infection or injury, is often called leukocyte homing, and the general process of leukocyte
The migration of a particular type of leukocyte into a restricted type of tissue, or a tissue with an ongoing infection or injury, is often called leukocyte homing, and the general process of leukocyte
Leukocyte Rolling Velocities and Migration Are Optimized by Cooperative L-Selectin and Intercellular Adhesion Molecule-1 Functions
 This information is current as of November 24, 2018. Leukocyte Rolling Velocities and Migration Are Optimized by Cooperative L-Selectin and Intercellular Adhesion Molecule-1 Functions Takafumi Kadono,
This information is current as of November 24, 2018. Leukocyte Rolling Velocities and Migration Are Optimized by Cooperative L-Selectin and Intercellular Adhesion Molecule-1 Functions Takafumi Kadono,
Genetics. Environment. You Are Only 10% Human. Pathogenesis of IBD. Advances in the Pathogenesis of IBD: Genetics Leads to Function IBD
 Advances in the Pathogenesis of IBD: Genetics Leads to Function Pathogenesis of IBD Environmental Factors Microbes Scott Plevy, MD Associate Professor of Medicine, Microbiology & Immunology UNC School
Advances in the Pathogenesis of IBD: Genetics Leads to Function Pathogenesis of IBD Environmental Factors Microbes Scott Plevy, MD Associate Professor of Medicine, Microbiology & Immunology UNC School
LECTURE 12: MUCOSAL IMMUNITY GUT STRUCTURE
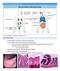 LECTURE 12: MUCOSAL IMMUNITY GUT STRUCTURE - Small intestine in humans is around 3-4 metres long - Internal surface of the small intestines are lined by villi o Villi are composed of absorptive cells (epithelial/enterocytes)
LECTURE 12: MUCOSAL IMMUNITY GUT STRUCTURE - Small intestine in humans is around 3-4 metres long - Internal surface of the small intestines are lined by villi o Villi are composed of absorptive cells (epithelial/enterocytes)
Molecular and Cellular Basis of Immune Protection of Mucosal Surfaces
 Molecular and Cellular Basis of Immune Protection of Mucosal Surfaces Department of Biologic & Materials Sciences School of Dentistry University of Michigan Ann Arbor, Michigan 48109-1078 1 Image quality
Molecular and Cellular Basis of Immune Protection of Mucosal Surfaces Department of Biologic & Materials Sciences School of Dentistry University of Michigan Ann Arbor, Michigan 48109-1078 1 Image quality
(Nonobese Diabetic) and Control Mice
 Developmental Immunology, 1994, Vol 3, pp. 273-282 Reprints available directly from the publisher Photocopying permitted by license only (C) 1994 Harwood Academic Publishers GmbH Printed in Singapore Strain-Dependent
Developmental Immunology, 1994, Vol 3, pp. 273-282 Reprints available directly from the publisher Photocopying permitted by license only (C) 1994 Harwood Academic Publishers GmbH Printed in Singapore Strain-Dependent
Macrophages, T Cell Receptor Usage, and Endothelial Cell Activation
 Macrophages, T Cell Receptor Usage, and Endothelial Cell Activation in the Pancreas at the Onset of Insulin-dependent Diabetes Mellitus Amo Hanninen, * Sirpa Jalkanen,'11 Marko Salmi,t11 Sakari Toikkanen,I
Macrophages, T Cell Receptor Usage, and Endothelial Cell Activation in the Pancreas at the Onset of Insulin-dependent Diabetes Mellitus Amo Hanninen, * Sirpa Jalkanen,'11 Marko Salmi,t11 Sakari Toikkanen,I
Putting it Together. Stephen Canfield Secondary Lymphoid System. Tonsil Anterior Cervical LN s
 Putting it Together Stephen Canfield smc12@columbia.edu Secondary Lymphoid System Tonsil Anterior Cervical LN s Axillary LN s Mediastinal/Retroperitoneal LN s Thoracic Duct Appendix Spleen Inguinal LN
Putting it Together Stephen Canfield smc12@columbia.edu Secondary Lymphoid System Tonsil Anterior Cervical LN s Axillary LN s Mediastinal/Retroperitoneal LN s Thoracic Duct Appendix Spleen Inguinal LN
Cell Adhesion and Migration IV. Lymphocyte trafficking in the intestine and liver *
 themes Cell Adhesion and Migration IV. Lymphocyte trafficking in the intestine and liver * M. SALMI, 1 D. ADAMS, 2 AND S. JALKANEN 1 1 MediCity Research Laboratories, University of Turku and National Public
themes Cell Adhesion and Migration IV. Lymphocyte trafficking in the intestine and liver * M. SALMI, 1 D. ADAMS, 2 AND S. JALKANEN 1 1 MediCity Research Laboratories, University of Turku and National Public
PBS Class #2 Introduction to the Immune System part II Suggested reading: Abbas, pgs , 27-30
 PBS 803 - Class #2 Introduction to the Immune System part II Suggested reading: Abbas, pgs. 15-25, 27-30 Learning Objectives Compare and contrast the maturation of B and T lymphocytes Compare and contrast
PBS 803 - Class #2 Introduction to the Immune System part II Suggested reading: Abbas, pgs. 15-25, 27-30 Learning Objectives Compare and contrast the maturation of B and T lymphocytes Compare and contrast
and a carbohydrate-based ligand (12, 12a) expressed by the EC of PN HEV cells. L-Selectin is a member of the "selectin"
 Rapid Publication A Lymphocyte Homing Receptor (L-Selectin) Mediates the In Vitro Attachment of Lymphocytes to Myelinated Tracts of the Central Nervous System Kun Huang, Joyce S. Geoffroy, Mark S. Singer,
Rapid Publication A Lymphocyte Homing Receptor (L-Selectin) Mediates the In Vitro Attachment of Lymphocytes to Myelinated Tracts of the Central Nervous System Kun Huang, Joyce S. Geoffroy, Mark S. Singer,
Homing and adhesion molecules in autoimmune gastritis
 Homing and adhesion molecules in autoimmune gastritis Simon P. Barrett, Amy Riordon, Ban Hock Toh, Paul A. Gleeson, and Ian R. van Driel Department of Pathology and Immunology, Monash University Medical
Homing and adhesion molecules in autoimmune gastritis Simon P. Barrett, Amy Riordon, Ban Hock Toh, Paul A. Gleeson, and Ian R. van Driel Department of Pathology and Immunology, Monash University Medical
Supplementary Materials. for Garmy-Susini, et al, Integrin 4 1 signaling is required for lymphangiogenesis and tumor metastasis
 Supplementary Materials for Garmy-Susini, et al, Integrin 4 1 signaling is required for lymphangiogenesis and tumor metastasis 1 Supplementary Figure Legends Supplementary Figure 1: Integrin expression
Supplementary Materials for Garmy-Susini, et al, Integrin 4 1 signaling is required for lymphangiogenesis and tumor metastasis 1 Supplementary Figure Legends Supplementary Figure 1: Integrin expression
Supplementary Figure 1. Generation of knockin mice expressing L-selectinN138G. (a) Schematics of the Sellg allele (top), the targeting vector, the
 Supplementary Figure 1. Generation of knockin mice expressing L-selectinN138G. (a) Schematics of the Sellg allele (top), the targeting vector, the targeted allele in ES cells, and the mutant allele in
Supplementary Figure 1. Generation of knockin mice expressing L-selectinN138G. (a) Schematics of the Sellg allele (top), the targeting vector, the targeted allele in ES cells, and the mutant allele in
4. TEXTBOOK: ABUL K. ABBAS. ANDREW H. LICHTMAN. CELLULAR AND MOLECULAR IMMUNOLOGY. 5 TH EDITION. Chapter 2. pg
 LECTURE: 03 Title: CELLS INVOLVED IN THE IMMUNE RESPONSE LEARNING OBJECTIVES: The student should be able to: Identify the organs where the process of the blood formation occurs. Identify the main cell
LECTURE: 03 Title: CELLS INVOLVED IN THE IMMUNE RESPONSE LEARNING OBJECTIVES: The student should be able to: Identify the organs where the process of the blood formation occurs. Identify the main cell
Antigen Presentation and T Lymphocyte Activation. Abul K. Abbas UCSF. FOCiS
 1 Antigen Presentation and T Lymphocyte Activation Abul K. Abbas UCSF FOCiS 2 Lecture outline Dendritic cells and antigen presentation The role of the MHC T cell activation Costimulation, the B7:CD28 family
1 Antigen Presentation and T Lymphocyte Activation Abul K. Abbas UCSF FOCiS 2 Lecture outline Dendritic cells and antigen presentation The role of the MHC T cell activation Costimulation, the B7:CD28 family
Date of preparation: 06/ IBD/
 Mechanisms Mechanisms of of Disease Disease: The The Science of of IBD IBD 64002 Date of preparation: 06/11 2013 IBD/2013-11-01 Table of Contents IBD Background and Disease Management Etiology and Pathophysiology
Mechanisms Mechanisms of of Disease Disease: The The Science of of IBD IBD 64002 Date of preparation: 06/11 2013 IBD/2013-11-01 Table of Contents IBD Background and Disease Management Etiology and Pathophysiology
Impaired Mucosal Immunity in L-Selectin-Deficient Mice Orally Immunized
 This information is current as of October 22, 2018. Impaired Mucosal Immunity in L-Selectin-Deficient Mice Orally Immunized with a Salmonella Vaccine Vector David W. Pascual, Michelle D. White, Trina Larson
This information is current as of October 22, 2018. Impaired Mucosal Immunity in L-Selectin-Deficient Mice Orally Immunized with a Salmonella Vaccine Vector David W. Pascual, Michelle D. White, Trina Larson
The Development of Lymphocytes: B Cell Development in the Bone Marrow & Peripheral Lymphoid Tissue Deborah A. Lebman, Ph.D.
 The Development of Lymphocytes: B Cell Development in the Bone Marrow & Peripheral Lymphoid Tissue Deborah A. Lebman, Ph.D. OBJECTIVES 1. To understand how ordered Ig gene rearrangements lead to the development
The Development of Lymphocytes: B Cell Development in the Bone Marrow & Peripheral Lymphoid Tissue Deborah A. Lebman, Ph.D. OBJECTIVES 1. To understand how ordered Ig gene rearrangements lead to the development
ACTIVATION OF T LYMPHOCYTES AND CELL MEDIATED IMMUNITY
 ACTIVATION OF T LYMPHOCYTES AND CELL MEDIATED IMMUNITY The recognition of specific antigen by naïve T cell induces its own activation and effector phases. T helper cells recognize peptide antigens through
ACTIVATION OF T LYMPHOCYTES AND CELL MEDIATED IMMUNITY The recognition of specific antigen by naïve T cell induces its own activation and effector phases. T helper cells recognize peptide antigens through
Entry of Naive CD4 T Cells into Peripheral Lymph Nodes Requires L-Selectin
 Bffef De~ni~'ve Report Entry of Naive CD4 T Cells into Peripheral Lymph Nodes Requires L-Selectin By Linda M. Bradley,* Susan R. Watson,r and Susan L. Swain* From the *Department of Biology and the University
Bffef De~ni~'ve Report Entry of Naive CD4 T Cells into Peripheral Lymph Nodes Requires L-Selectin By Linda M. Bradley,* Susan R. Watson,r and Susan L. Swain* From the *Department of Biology and the University
Lecture 9: T-cell Mediated Immunity
 Lecture 9: T-cell Mediated Immunity Questions to Consider How do T cells know where to go? Questions to Consider How do T cells know where to go? How does antigen get targeted to a T cell expressing the
Lecture 9: T-cell Mediated Immunity Questions to Consider How do T cells know where to go? Questions to Consider How do T cells know where to go? How does antigen get targeted to a T cell expressing the
MACROPHAGE "MONOCYTES" SURFACE RECEPTORS
 LECTURE: 13 Title: MACROPHAGE "MONOCYTES" SURFACE RECEPTORS LEARNING OBJECTIVES: The student should be able to: Describe the blood monocytes (size, and shape of nucleus). Enumerate some of the monocytes
LECTURE: 13 Title: MACROPHAGE "MONOCYTES" SURFACE RECEPTORS LEARNING OBJECTIVES: The student should be able to: Describe the blood monocytes (size, and shape of nucleus). Enumerate some of the monocytes
Hidenobu Kanda, Rebecca Newton, Russell Klein, Yuka Morita, Michael D. Gunn & Steven D. Rosen
 Autotaxin, a lysophosphatidic acid-producing ectoenzyme, promotes lymphocyte entry into secondary lymphoid organs Hidenou Kanda, Reecca Newton, Russell Klein, Yuka Morita, Michael D. Gunn & Steven D. Rosen
Autotaxin, a lysophosphatidic acid-producing ectoenzyme, promotes lymphocyte entry into secondary lymphoid organs Hidenou Kanda, Reecca Newton, Russell Klein, Yuka Morita, Michael D. Gunn & Steven D. Rosen
Immunology Lecture 4. Clinical Relevance of the Immune System
 Immunology Lecture 4 The Well Patient: How innate and adaptive immune responses maintain health - 13, pg 169-181, 191-195. Immune Deficiency - 15 Autoimmunity - 16 Transplantation - 17, pg 260-270 Tumor
Immunology Lecture 4 The Well Patient: How innate and adaptive immune responses maintain health - 13, pg 169-181, 191-195. Immune Deficiency - 15 Autoimmunity - 16 Transplantation - 17, pg 260-270 Tumor
Effector T Cells and
 1 Effector T Cells and Cytokines Andrew Lichtman, MD PhD Brigham and Women's Hospital Harvard Medical School 2 Lecture outline Cytokines Subsets of CD4+ T cells: definitions, functions, development New
1 Effector T Cells and Cytokines Andrew Lichtman, MD PhD Brigham and Women's Hospital Harvard Medical School 2 Lecture outline Cytokines Subsets of CD4+ T cells: definitions, functions, development New
T Cell Activation, Costimulation and Regulation
 1 T Cell Activation, Costimulation and Regulation Abul K. Abbas, MD University of California San Francisco 2 Lecture outline T cell antigen recognition and activation Costimulation, the B7:CD28 family
1 T Cell Activation, Costimulation and Regulation Abul K. Abbas, MD University of California San Francisco 2 Lecture outline T cell antigen recognition and activation Costimulation, the B7:CD28 family
Homing and Inflammation!
 Homing and Inflammation! Micro 204. Molecular and Cellular Immunology! Lecturer: Jason Cyster! jason.cyster@ucsf.edu! How do cells migrate from blood into tissue?! the four step model! role of selectins,
Homing and Inflammation! Micro 204. Molecular and Cellular Immunology! Lecturer: Jason Cyster! jason.cyster@ucsf.edu! How do cells migrate from blood into tissue?! the four step model! role of selectins,
Lymphoid architecture & Leukocyte recirculation. Thursday Jan 26th, 2017
 Lymphoid architecture & Leukocyte recirculation Thursday Jan 26th, 2017 Topics The life of immune cells Where are they born? Where are they educated? Where do they function? How do they get there? The
Lymphoid architecture & Leukocyte recirculation Thursday Jan 26th, 2017 Topics The life of immune cells Where are they born? Where are they educated? Where do they function? How do they get there? The
Immune response. This overview figure summarizes simply how our body responds to foreign molecules that enter to it.
 Immune response This overview figure summarizes simply how our body responds to foreign molecules that enter to it. It s highly recommended to watch Dr Najeeb s lecture that s titled T Helper cells and
Immune response This overview figure summarizes simply how our body responds to foreign molecules that enter to it. It s highly recommended to watch Dr Najeeb s lecture that s titled T Helper cells and
Monocytes must also leave the circulation in response to inflammatory signals, and neutrophils and other granulocytes must be able to do so as well.
 Immunology Dr. John J. Haddad Chapter 15 Leukocyte Migration and inflammation Lymphocytes must recirculate in the body in search of antigens. Since antigens are brought to lymph nodes and the spleen, the
Immunology Dr. John J. Haddad Chapter 15 Leukocyte Migration and inflammation Lymphocytes must recirculate in the body in search of antigens. Since antigens are brought to lymph nodes and the spleen, the
Test Bank for Basic Immunology Functions and Disorders of the Immune System 4th Edition by Abbas
 Test Bank for Basic Immunology Functions and Disorders of the Immune System 4th Edition by Abbas Chapter 04: Antigen Recognition in the Adaptive Immune System Test Bank MULTIPLE CHOICE 1. Most T lymphocytes
Test Bank for Basic Immunology Functions and Disorders of the Immune System 4th Edition by Abbas Chapter 04: Antigen Recognition in the Adaptive Immune System Test Bank MULTIPLE CHOICE 1. Most T lymphocytes
A Molecular Basis for the Compartmentalization of Effector B Cell Responses. Abstract. Introduction
 Differential Expression of Tissue-specific Adhesion Molecules on Human Circulating Antibody-forming Cells After Systemic, Enteric, and Nasal Immunizations A Molecular Basis for the Compartmentalization
Differential Expression of Tissue-specific Adhesion Molecules on Human Circulating Antibody-forming Cells After Systemic, Enteric, and Nasal Immunizations A Molecular Basis for the Compartmentalization
INTRODUCTION. Sharada Kommajosyula,* Shiva Reddy, Kristina Nitschke, Jagat R. Kanwar,* Muralidhar Karanam, and Geoffrey W.
 Leukocytes infiltrating the pancreatic islets of nonobese diabetic mice are transformed into inactive exiles by combinational anti-cell adhesion therapy Sharada Kommajosyula,* Shiva Reddy, Kristina Nitschke,
Leukocytes infiltrating the pancreatic islets of nonobese diabetic mice are transformed into inactive exiles by combinational anti-cell adhesion therapy Sharada Kommajosyula,* Shiva Reddy, Kristina Nitschke,
Review Questions: Janeway s Immunobiology 8th Edition by Kenneth Murphy
 Review Questions: Janeway s Immunobiology 8th Edition by Kenneth Murphy Chapter 11 (pages 429-460): Dynamics of Adaptive Immunity prepared by Kelly von Elten, Walter Reed National Military Medical Center,
Review Questions: Janeway s Immunobiology 8th Edition by Kenneth Murphy Chapter 11 (pages 429-460): Dynamics of Adaptive Immunity prepared by Kelly von Elten, Walter Reed National Military Medical Center,
TCR, MHC and coreceptors
 Cooperation In Immune Responses Antigen processing how peptides get into MHC Antigen processing involves the intracellular proteolytic generation of MHC binding proteins Protein antigens may be processed
Cooperation In Immune Responses Antigen processing how peptides get into MHC Antigen processing involves the intracellular proteolytic generation of MHC binding proteins Protein antigens may be processed
Lymphocyte migration across high endothelium is associated with increases in 4 1 integrin (VLA-4) affinity
 Journal of Cell Science 104, 1049-1059 (1993) Printed in Great Britain The Company of Biologists Limited 1993 1049 Lymphocyte migration across high endothelium is associated with increases in 4 1 integrin
Journal of Cell Science 104, 1049-1059 (1993) Printed in Great Britain The Company of Biologists Limited 1993 1049 Lymphocyte migration across high endothelium is associated with increases in 4 1 integrin
Homing and Inflammation!
 Homing and Inflammation! Micro 204. Molecular and Cellular Immunology! Lecturer: Jason Cyster! jason.cyster@ucsf.edu! How do cells migrate from blood into tissue?! the four step model! role of selectins,
Homing and Inflammation! Micro 204. Molecular and Cellular Immunology! Lecturer: Jason Cyster! jason.cyster@ucsf.edu! How do cells migrate from blood into tissue?! the four step model! role of selectins,
T-cell activation T cells migrate to secondary lymphoid tissues where they interact with antigen, antigen-presenting cells, and other lymphocytes:
 Interactions between innate immunity & adaptive immunity What happens to T cells after they leave the thymus? Naïve T cells exit the thymus and enter the bloodstream. If they remain in the bloodstream,
Interactions between innate immunity & adaptive immunity What happens to T cells after they leave the thymus? Naïve T cells exit the thymus and enter the bloodstream. If they remain in the bloodstream,
T-cell activation T cells migrate to secondary lymphoid tissues where they interact with antigen, antigen-presenting cells, and other lymphocytes:
 Interactions between innate immunity & adaptive immunity What happens to T cells after they leave the thymus? Naïve T cells exit the thymus and enter the bloodstream. If they remain in the bloodstream,
Interactions between innate immunity & adaptive immunity What happens to T cells after they leave the thymus? Naïve T cells exit the thymus and enter the bloodstream. If they remain in the bloodstream,
Immunology 2017: Lecture 12 handout. Secondary lymphoid organs. Dr H Awad
 Immunology 2017: Lecture 12 handout Secondary lymphoid organs Dr H Awad INTRODUCTION So far we discussed the cells of the immune system and how they recognize their antigens and get stimulated. The number
Immunology 2017: Lecture 12 handout Secondary lymphoid organs Dr H Awad INTRODUCTION So far we discussed the cells of the immune system and how they recognize their antigens and get stimulated. The number
FOR OPTIMAL GUT HEALTH KEMIN.COM/GUTHEALTH
 FOR OPTIMAL GUT HEALTH KEMIN.COM/GUTHEALTH ALETA A SOURCE OF 1,3-BETA GLUCANS Aleta is highly bioavailable, offering a concentration greater than 5% of 1,3-beta glucans. Aleta provides a consistent response
FOR OPTIMAL GUT HEALTH KEMIN.COM/GUTHEALTH ALETA A SOURCE OF 1,3-BETA GLUCANS Aleta is highly bioavailable, offering a concentration greater than 5% of 1,3-beta glucans. Aleta provides a consistent response
Attribution: University of Michigan Medical School, Department of Microbiology and Immunology
 Attribution: University of Michigan Medical School, Department of Microbiology and Immunology License: Unless otherwise noted, this material is made available under the terms of the Creative Commons Attribution
Attribution: University of Michigan Medical School, Department of Microbiology and Immunology License: Unless otherwise noted, this material is made available under the terms of the Creative Commons Attribution
Immunology - Problem Drill 04: Structure and Functions of the Immune System
 Immunology - Problem Drill 04: Structure and Functions of the Immune System Question No. 1 of 10 1. Which one of the following is non-encapsulated and less organized secondary lymphoid organ? Question
Immunology - Problem Drill 04: Structure and Functions of the Immune System Question No. 1 of 10 1. Which one of the following is non-encapsulated and less organized secondary lymphoid organ? Question
What is Autoimmunity?
 Autoimmunity What is Autoimmunity? Robert Beatty MCB150 Autoimmunity is an immune response to self antigens that results in disease. The immune response to self is a result of a breakdown in immune tolerance.
Autoimmunity What is Autoimmunity? Robert Beatty MCB150 Autoimmunity is an immune response to self antigens that results in disease. The immune response to self is a result of a breakdown in immune tolerance.
What is Autoimmunity?
 Autoimmunity What is Autoimmunity? Robert Beatty MCB150 Autoimmunity is an immune response to self antigens that results in disease. The immune response to self is a result of a breakdown in immune tolerance.
Autoimmunity What is Autoimmunity? Robert Beatty MCB150 Autoimmunity is an immune response to self antigens that results in disease. The immune response to self is a result of a breakdown in immune tolerance.
Suppl Video: Tumor cells (green) and monocytes (white) are seeded on a confluent endothelial
 Supplementary Information Häuselmann et al. Monocyte induction of E-selectin-mediated endothelial activation releases VE-cadherin junctions to promote tumor cell extravasation in the metastasis cascade
Supplementary Information Häuselmann et al. Monocyte induction of E-selectin-mediated endothelial activation releases VE-cadherin junctions to promote tumor cell extravasation in the metastasis cascade
T-LYMPHOCYTE INTERACTIONS WITH ENDOTHELIUM AND EXTRACELLULAR MATRIX
 T-LYMPHOCYTE INTERACTIONS WITH ENDOTHELIUM AND EXTRACELLULAR MATRIX Stephen W. Hunt III Division of Immunopathology, Warner-Lambert/Parke-Davis Pharmaceutical Research Division, Ann Arbor, Ml 48105 Estelle
T-LYMPHOCYTE INTERACTIONS WITH ENDOTHELIUM AND EXTRACELLULAR MATRIX Stephen W. Hunt III Division of Immunopathology, Warner-Lambert/Parke-Davis Pharmaceutical Research Division, Ann Arbor, Ml 48105 Estelle
SUPPLEMENTARY FIGURE 1
 SUPPLEMENTARY FIGURE 1 A LN Cell count (1 ) 1 3 1 CD+ 1 1 CDL lo CD hi 1 CD+FoxP3+ 1 1 1 7 3 3 3 % of cells 9 7 7 % of cells CD+ 3 1 % of cells CDL lo CD hi 1 1 % of CD+ cells CD+FoxP3+ 3 1 % of CD+ T
SUPPLEMENTARY FIGURE 1 A LN Cell count (1 ) 1 3 1 CD+ 1 1 CDL lo CD hi 1 CD+FoxP3+ 1 1 1 7 3 3 3 % of cells 9 7 7 % of cells CD+ 3 1 % of cells CDL lo CD hi 1 1 % of CD+ cells CD+FoxP3+ 3 1 % of CD+ T
Lymphoid System: cells of the immune system. Answer Sheet
 Lymphoid System: cells of the immune system Answer Sheet Q1 Which areas of the lymph node have most CD3 staining? A1 Most CD3 staining is present in the paracortex (T cell areas). This is towards the outside
Lymphoid System: cells of the immune system Answer Sheet Q1 Which areas of the lymph node have most CD3 staining? A1 Most CD3 staining is present in the paracortex (T cell areas). This is towards the outside
IMMUNE CELL SURFACE RECEPTORS AND THEIR FUNCTIONS
 LECTURE: 07 Title: IMMUNE CELL SURFACE RECEPTORS AND THEIR FUNCTIONS LEARNING OBJECTIVES: The student should be able to: The chemical nature of the cellular surface receptors. Define the location of the
LECTURE: 07 Title: IMMUNE CELL SURFACE RECEPTORS AND THEIR FUNCTIONS LEARNING OBJECTIVES: The student should be able to: The chemical nature of the cellular surface receptors. Define the location of the
Mucosal Immunology Sophomore Dental and Optometry Microbiology Section I: Immunology. Robin Lorenz
 Mucosal Immunology Sophomore Dental and Optometry Microbiology Section I: Immunology Robin Lorenz rlorenz@uab.edu Why do we Need to Understand How the Mucosal Immune System Works? The mucosa is the major
Mucosal Immunology Sophomore Dental and Optometry Microbiology Section I: Immunology Robin Lorenz rlorenz@uab.edu Why do we Need to Understand How the Mucosal Immune System Works? The mucosa is the major
Cytokines modulate the functional activities of individual cells and tissues both under normal and pathologic conditions Interleukins,
 Cytokines http://highered.mcgraw-hill.com/sites/0072507470/student_view0/chapter22/animation the_immune_response.html Cytokines modulate the functional activities of individual cells and tissues both under
Cytokines http://highered.mcgraw-hill.com/sites/0072507470/student_view0/chapter22/animation the_immune_response.html Cytokines modulate the functional activities of individual cells and tissues both under
Effector mechanisms of cell-mediated immunity: Properties of effector, memory and regulatory T cells
 ICI Basic Immunology course Effector mechanisms of cell-mediated immunity: Properties of effector, memory and regulatory T cells Abul K. Abbas, MD UCSF Stages in the development of T cell responses: induction
ICI Basic Immunology course Effector mechanisms of cell-mediated immunity: Properties of effector, memory and regulatory T cells Abul K. Abbas, MD UCSF Stages in the development of T cell responses: induction
Lymphoid Organs and Lymphocyte Trafficking. Dr. Issa Abu-Dayyeh
 Lymphoid Organs and Lymphocyte Trafficking Dr. Issa Abu-Dayyeh Invader recognition Where does invader recognition take place?? Secondary lymphoid organs: Lymph nodes Spleen Mucosal-associated lymphoid
Lymphoid Organs and Lymphocyte Trafficking Dr. Issa Abu-Dayyeh Invader recognition Where does invader recognition take place?? Secondary lymphoid organs: Lymph nodes Spleen Mucosal-associated lymphoid
Chapter 2 (pages 22 33): Cells and Tissues of the Immune System. Prepared by Kristen Dazy, MD, Scripps Clinic Medical Group
 Allergy and Immunology Review Corner: Cellular and Molecular Immunology, 8th Edition By Abul K. Abbas, MBBS; Andrew H. H. Lichtman, MD, PhD; and Shiv Pillai, MBBS, PhD. Chapter 2 (pages 22 33): Cells and
Allergy and Immunology Review Corner: Cellular and Molecular Immunology, 8th Edition By Abul K. Abbas, MBBS; Andrew H. H. Lichtman, MD, PhD; and Shiv Pillai, MBBS, PhD. Chapter 2 (pages 22 33): Cells and
Endogenous TNFα orchestrates the trafficking of neutrophils into and within lymphatic vessels during acute inflammation
 SUPPLEMENTARY INFORMATION Endogenous TNFα orchestrates the trafficking of neutrophils into and within lymphatic vessels during acute inflammation Samantha Arokiasamy 1,2, Christian Zakian 1, Jessica Dilliway
SUPPLEMENTARY INFORMATION Endogenous TNFα orchestrates the trafficking of neutrophils into and within lymphatic vessels during acute inflammation Samantha Arokiasamy 1,2, Christian Zakian 1, Jessica Dilliway
Lymphocyte homing to the gut: attraction, adhesion, and commitment
 Marko Salmi Sirpa Jalkanen Lymphocyte homing to the gut: attraction, adhesion, and commitment Authors address Marko Salmi, Sirpa Jalkanen National Public Health Institute Turku, MediCity Research Laboratory,
Marko Salmi Sirpa Jalkanen Lymphocyte homing to the gut: attraction, adhesion, and commitment Authors address Marko Salmi, Sirpa Jalkanen National Public Health Institute Turku, MediCity Research Laboratory,
?Who binds to it. ? Who binds these inflammatory proteins RAGE SUGAR-FREE GLYCOBIOLOGY INFLAMMATION. BASIC SCIENCE?sugar chain structure
 SUGAR-FREE GLYCOBIOLOGY BASIC SCIENCE?sugar chain structure Unusual Sugar Chain It -- is a carboxylate Make lots of It Make an It antibody Inflammatory proteins?who binds to it? Who binds these inflammatory
SUGAR-FREE GLYCOBIOLOGY BASIC SCIENCE?sugar chain structure Unusual Sugar Chain It -- is a carboxylate Make lots of It Make an It antibody Inflammatory proteins?who binds to it? Who binds these inflammatory
Neutrophils contribute to fracture healing by synthesizing fibronectin+ extracellular matrix rapidly after injury
 Neutrophils contribute to fracture healing by synthesizing fibronectin+ extracellular matrix rapidly after injury Bastian OW, Koenderman L, Alblas J, Leenen LPH, Blokhuis TJ. Neutrophils contribute to
Neutrophils contribute to fracture healing by synthesizing fibronectin+ extracellular matrix rapidly after injury Bastian OW, Koenderman L, Alblas J, Leenen LPH, Blokhuis TJ. Neutrophils contribute to
Cytokines, adhesion molecules and apoptosis markers. A comprehensive product line for human and veterinary ELISAs
 Cytokines, adhesion molecules and apoptosis markers A comprehensive product line for human and veterinary ELISAs IBL International s cytokine product line... is extremely comprehensive. The assays are
Cytokines, adhesion molecules and apoptosis markers A comprehensive product line for human and veterinary ELISAs IBL International s cytokine product line... is extremely comprehensive. The assays are
Lymphoid tissue. 1. Central Lymphoid tissue. - The central lymphoid tissue (also known as primary) is composed of bone morrow and thymus.
 1. Central Lymphoid tissue Lymphoid tissue - The central lymphoid tissue (also known as primary) is composed of bone morrow and thymus. Bone Morrow - The major site of hematopoiesis in humans. - Hematopoiesis
1. Central Lymphoid tissue Lymphoid tissue - The central lymphoid tissue (also known as primary) is composed of bone morrow and thymus. Bone Morrow - The major site of hematopoiesis in humans. - Hematopoiesis
Är diabetes mellitus en autoimmun sjukdom? Olle Korsgren
 Är diabetes mellitus en autoimmun sjukdom? Olle Korsgren Type 1 Diabetes is currently regarded as a T cell mediated autoimmune disease, a notion expressed in over 50 000 scientific publications. Acute
Är diabetes mellitus en autoimmun sjukdom? Olle Korsgren Type 1 Diabetes is currently regarded as a T cell mediated autoimmune disease, a notion expressed in over 50 000 scientific publications. Acute
immunity produced by an encounter with an antigen; provides immunologic memory. active immunity clumping of (foreign) cells; induced by crosslinking
 active immunity agglutination allografts immunity produced by an encounter with an antigen; provides immunologic memory. clumping of (foreign) cells; induced by crosslinking of antigenantibody complexes.
active immunity agglutination allografts immunity produced by an encounter with an antigen; provides immunologic memory. clumping of (foreign) cells; induced by crosslinking of antigenantibody complexes.
Mon, Wed, Fri 11:00 AM-12:00 PM. Owen, Judy, Jenni Punt, and Sharon Stranford Kuby-Immunology, 7th. Edition. W.H. Freeman and Co., New York.
 Course Title: Course Number: Immunology Biol-341/541 Semester: Fall 2013 Location: HS 268 Time: Instructor: 8:00-9:30 AM Tue/Thur Dr. Colleen M. McDermott Office: Nursing Ed 101 (424-1217) E-mail*: mcdermot@uwosh.edu
Course Title: Course Number: Immunology Biol-341/541 Semester: Fall 2013 Location: HS 268 Time: Instructor: 8:00-9:30 AM Tue/Thur Dr. Colleen M. McDermott Office: Nursing Ed 101 (424-1217) E-mail*: mcdermot@uwosh.edu
Differential Recruitment of T- and IgA B-lymphocytes in the Developing Mammary Gland in Relation to Homing Receptors and Vascular Addressins
 Volume 47(12): 1581 1592, 1999 The Journal of Histochemistry & Cytochemistry http://www.jhc.org ARTICLE Differential Recruitment of T- and IgA B-lymphocytes in the Developing Mammary Gland in Relation
Volume 47(12): 1581 1592, 1999 The Journal of Histochemistry & Cytochemistry http://www.jhc.org ARTICLE Differential Recruitment of T- and IgA B-lymphocytes in the Developing Mammary Gland in Relation
Cell-mediated response (what type of cell is activated and what gets destroyed?)
 The Immune System Reading Guide (Chapter 43) Name Per 1. The immune response in animals can be divided into innate immunity and adaptive immunity. As an overview, complete this figure indicating the divisions
The Immune System Reading Guide (Chapter 43) Name Per 1. The immune response in animals can be divided into innate immunity and adaptive immunity. As an overview, complete this figure indicating the divisions
Anti-DC-SIGN/CD209 murine monoclonal antibodies
 Anti-DC-SIGN/CD209 murine monoclonal antibodies DC-SIGN (DC Specific, ICAM-3 Grabbing, Nonintegrin) / CD209 and L-SIGN (liver/lymph node-specific ICAM-3-grabbing nonintegrin CD299/ DC-SIGNR (DC-SIGN-related
Anti-DC-SIGN/CD209 murine monoclonal antibodies DC-SIGN (DC Specific, ICAM-3 Grabbing, Nonintegrin) / CD209 and L-SIGN (liver/lymph node-specific ICAM-3-grabbing nonintegrin CD299/ DC-SIGNR (DC-SIGN-related
Effects of biological response modifiers in psoriasis and psoriatic arthritis Goedkoop, A.Y.
 UvA-DARE (Digital Academic Repository) Effects of biological response modifiers in psoriasis and psoriatic arthritis Goedkoop, A.Y. Link to publication Citation for published version (APA): Goedkoop, A.
UvA-DARE (Digital Academic Repository) Effects of biological response modifiers in psoriasis and psoriatic arthritis Goedkoop, A.Y. Link to publication Citation for published version (APA): Goedkoop, A.
Principles of Adaptive Immunity
 Principles of Adaptive Immunity Chapter 3 Parham Hans de Haard 17 th of May 2010 Agenda Recognition molecules of adaptive immune system Features adaptive immune system Immunoglobulins and T-cell receptors
Principles of Adaptive Immunity Chapter 3 Parham Hans de Haard 17 th of May 2010 Agenda Recognition molecules of adaptive immune system Features adaptive immune system Immunoglobulins and T-cell receptors
NEUTROPHIL, BASOPHIL, EOSINOPHIL, AND PLATELETS SURFACE RECEPTORS
 LECTURE: 15 Title NEUTROPHIL, BASOPHIL, EOSINOPHIL, AND PLATELETS SURFACE RECEPTORS LEARNING OBJECTIVES: The student should be able to: Determine the relative percentages in blood for the various types
LECTURE: 15 Title NEUTROPHIL, BASOPHIL, EOSINOPHIL, AND PLATELETS SURFACE RECEPTORS LEARNING OBJECTIVES: The student should be able to: Determine the relative percentages in blood for the various types
The Immune System: The Mind Body Connection. Presented by Margaret Kemeny, Ph.D. Department of Psychiatry, University of California, San Francisco
 The Immune System: The Mind Body Connection Presented by Margaret Kemeny, Ph.D. Department of Psychiatry, University of California, San Francisco Psychoneuroimmunology Investigation of the bidirectional
The Immune System: The Mind Body Connection Presented by Margaret Kemeny, Ph.D. Department of Psychiatry, University of California, San Francisco Psychoneuroimmunology Investigation of the bidirectional
CHAPTER 9 BIOLOGY OF THE T LYMPHOCYTE
 CHAPTER 9 BIOLOGY OF THE T LYMPHOCYTE Coico, R., Sunshine, G., (2009) Immunology : a short course, 6 th Ed., Wiley-Blackwell 1 CHAPTER 9 : Biology of The T Lymphocytes 1. 2. 3. 4. 5. 6. 7. Introduction
CHAPTER 9 BIOLOGY OF THE T LYMPHOCYTE Coico, R., Sunshine, G., (2009) Immunology : a short course, 6 th Ed., Wiley-Blackwell 1 CHAPTER 9 : Biology of The T Lymphocytes 1. 2. 3. 4. 5. 6. 7. Introduction
Homing of Mucosal Leukocytes to Joints
 Homing of Mucosal Leukocytes to Joints Distinct Endothelial Ligands in Synovium Mediate Leukocyte-subtype Specific Adhesion Marko Salmi,* Pertti Rajala, and Sirpa Jalkanen* *National Public Health Institute,
Homing of Mucosal Leukocytes to Joints Distinct Endothelial Ligands in Synovium Mediate Leukocyte-subtype Specific Adhesion Marko Salmi,* Pertti Rajala, and Sirpa Jalkanen* *National Public Health Institute,
CD8 cytotoxic T-cell clone rapidly transfers autoimmune diabetes in very young NOD and MHC class I-compatible scid mice
 Diabetologia (1997) 40: 1044 1052 Springer-Verlag 1997 CD8 cytotoxic T-cell clone rapidly transfers autoimmune diabetes in very young NOD and MHC class I-compatible scid mice R. Yoneda, K. Yokono, M. Nagata,
Diabetologia (1997) 40: 1044 1052 Springer-Verlag 1997 CD8 cytotoxic T-cell clone rapidly transfers autoimmune diabetes in very young NOD and MHC class I-compatible scid mice R. Yoneda, K. Yokono, M. Nagata,
Rapid Flow Cytometry Method for Quantitation of LFA-1-Adhesive T Cells
 CLINICAL AND VACCINE IMMUNOLOGY, Mar. 2006, p. 403 408 Vol. 13, No. 3 1556-6811/06/$08.00 0 doi:10.1128/cvi.13.3.403 408.2006 Rapid Flow Cytometry Method for Quantitation of LFA-1-Adhesive T Cells Brian
CLINICAL AND VACCINE IMMUNOLOGY, Mar. 2006, p. 403 408 Vol. 13, No. 3 1556-6811/06/$08.00 0 doi:10.1128/cvi.13.3.403 408.2006 Rapid Flow Cytometry Method for Quantitation of LFA-1-Adhesive T Cells Brian
LIST OF ORGANS FOR HISTOPATHOLOGICAL ANALYSIS:!! Neural!!!!!!Respiratory:! Brain : Cerebrum,!!! Lungs and trachea! Olfactory, Cerebellum!!!!Other:!
 LIST OF ORGANS FOR HISTOPATHOLOGICAL ANALYSIS:!! Neural!!!!!!Respiratory:! Brain : Cerebrum,!!! Lungs and trachea! Olfactory, Cerebellum!!!!Other:! Spinal cord and peripheral nerves! Eyes, Inner ear, nasal
LIST OF ORGANS FOR HISTOPATHOLOGICAL ANALYSIS:!! Neural!!!!!!Respiratory:! Brain : Cerebrum,!!! Lungs and trachea! Olfactory, Cerebellum!!!!Other:! Spinal cord and peripheral nerves! Eyes, Inner ear, nasal
Nutrition & Gut Immunity
 Nutrition & Gut Immunity Kenneth A. Kudsk, MD FACS Professor of Surgery University of Wisconsin - Madison Madison, Wisconsin No conflicts of interest to disclose % Survival Survival vs. Malnutrition (after
Nutrition & Gut Immunity Kenneth A. Kudsk, MD FACS Professor of Surgery University of Wisconsin - Madison Madison, Wisconsin No conflicts of interest to disclose % Survival Survival vs. Malnutrition (after
1) Mononuclear phagocytes : 2) Regarding acute inflammation : 3) The epithelioid cells of follicular granulomas are :
 Pathology Second 1) Mononuclear phagocytes : - Are the predominant cells in three day old wounds - Are common in liver, spleen and pancreasd - Produce fibroblast growth factor - Secrete interferon-g -
Pathology Second 1) Mononuclear phagocytes : - Are the predominant cells in three day old wounds - Are common in liver, spleen and pancreasd - Produce fibroblast growth factor - Secrete interferon-g -
Molecules controlling lymphocyte migration to the gut
 148 Gut 1999;45:148 153 Review Molecules controlling lymphocyte migration to the gut Preface Continuous lymphocyte migration to normal gut is a prerequisite for immune homoeostasis in humans. In this review
148 Gut 1999;45:148 153 Review Molecules controlling lymphocyte migration to the gut Preface Continuous lymphocyte migration to normal gut is a prerequisite for immune homoeostasis in humans. In this review
Immune system. Aims. Immune system. Lymphatic organs. Inflammation. Natural immune system. Adaptive immune system
 Aims Immune system Lymphatic organs Inflammation Natural immune system Adaptive immune system Major histocompatibility complex (MHC) Disorders of the immune system 1 2 Immune system Lymphoid organs Immune
Aims Immune system Lymphatic organs Inflammation Natural immune system Adaptive immune system Major histocompatibility complex (MHC) Disorders of the immune system 1 2 Immune system Lymphoid organs Immune
Human Anatomy and Physiology - Problem Drill 20: Immunity and the Lymphatic System
 Human Anatomy and Physiology - Problem Drill 20: Immunity and the Lymphatic System Question No. 1 of 10 The lymphatic system is formed early during human development. Which of the following statements
Human Anatomy and Physiology - Problem Drill 20: Immunity and the Lymphatic System Question No. 1 of 10 The lymphatic system is formed early during human development. Which of the following statements
Immune responses in autoimmune diseases
 Immune responses in autoimmune diseases Erika Jensen-Jarolim Dept. of Pathophysiology Medical University Vienna CCHD Lecture January 24, 2007 Primary immune organs: Bone marrow Thymus Secondary: Lymph
Immune responses in autoimmune diseases Erika Jensen-Jarolim Dept. of Pathophysiology Medical University Vienna CCHD Lecture January 24, 2007 Primary immune organs: Bone marrow Thymus Secondary: Lymph
Medical Virology Immunology. Dr. Sameer Naji, MB, BCh, PhD (UK) Head of Basic Medical Sciences Dept. Faculty of Medicine The Hashemite University
 Medical Virology Immunology Dr. Sameer Naji, MB, BCh, PhD (UK) Head of Basic Medical Sciences Dept. Faculty of Medicine The Hashemite University Human blood cells Phases of immune responses Microbe Naïve
Medical Virology Immunology Dr. Sameer Naji, MB, BCh, PhD (UK) Head of Basic Medical Sciences Dept. Faculty of Medicine The Hashemite University Human blood cells Phases of immune responses Microbe Naïve
Generation of post-germinal centre myeloma plasma B cell.
 Generation of post-germinal centre myeloma. DNA DAMAGE CXCR4 Homing to Lytic lesion activation CD38 CD138 CD56 Phenotypic markers Naive Secondary lymphoid organ Multiple myeloma is a malignancy of s caused
Generation of post-germinal centre myeloma. DNA DAMAGE CXCR4 Homing to Lytic lesion activation CD38 CD138 CD56 Phenotypic markers Naive Secondary lymphoid organ Multiple myeloma is a malignancy of s caused
Microbiology 204: Cellular and Molecular Immunology
 Microbiology 204: Cellular and Molecular Immunology Class meets MWF 1:00-2:30PM (*exceptions: no class Fri Sept 23, Fri Oct 14, Nov 11, or Wed Nov 23) Lectures are open to auditors and will be live-streamed
Microbiology 204: Cellular and Molecular Immunology Class meets MWF 1:00-2:30PM (*exceptions: no class Fri Sept 23, Fri Oct 14, Nov 11, or Wed Nov 23) Lectures are open to auditors and will be live-streamed
Immune response to infection
 Immune response to infection Dr. Sandra Nitsche (Sandra.Nitsche@rub.de ) 20.06.2018 1 Course of acute infection Typical acute infection that is cleared by an adaptive immune reaction 1. invasion of pathogen
Immune response to infection Dr. Sandra Nitsche (Sandra.Nitsche@rub.de ) 20.06.2018 1 Course of acute infection Typical acute infection that is cleared by an adaptive immune reaction 1. invasion of pathogen
Follicular Lymphoma. ced3 APOPTOSIS. *In the nematode Caenorhabditis elegans 131 of the organism's 1031 cells die during development.
 Harvard-MIT Division of Health Sciences and Technology HST.176: Cellular and Molecular Immunology Course Director: Dr. Shiv Pillai Follicular Lymphoma 1. Characterized by t(14:18) translocation 2. Ig heavy
Harvard-MIT Division of Health Sciences and Technology HST.176: Cellular and Molecular Immunology Course Director: Dr. Shiv Pillai Follicular Lymphoma 1. Characterized by t(14:18) translocation 2. Ig heavy
Basic immunology. Lecture 9. Innate immunity: inflammation, leukocyte migration. Péter Engelmann
 Basic immunology Lecture 9. Innate immunity: inflammation, leukocyte migration Péter Engelmann Different levels of the immune response Recognition molecules of innate immunity Initiation of local and systemic
Basic immunology Lecture 9. Innate immunity: inflammation, leukocyte migration Péter Engelmann Different levels of the immune response Recognition molecules of innate immunity Initiation of local and systemic
ACTIVATION AND EFFECTOR FUNCTIONS OF CELL-MEDIATED IMMUNITY AND NK CELLS. Choompone Sakonwasun, MD (Hons), FRCPT
 ACTIVATION AND EFFECTOR FUNCTIONS OF CELL-MEDIATED IMMUNITY AND NK CELLS Choompone Sakonwasun, MD (Hons), FRCPT Types of Adaptive Immunity Types of T Cell-mediated Immune Reactions CTLs = cytotoxic T lymphocytes
ACTIVATION AND EFFECTOR FUNCTIONS OF CELL-MEDIATED IMMUNITY AND NK CELLS Choompone Sakonwasun, MD (Hons), FRCPT Types of Adaptive Immunity Types of T Cell-mediated Immune Reactions CTLs = cytotoxic T lymphocytes
The surveillance of the body for foreign antigens is a. Molecular Mechanisms of Lymphocyte Homing to Peripheral Lymph Nodes
 Molecular Mechanisms of Lymphocyte Homing to Peripheral Lymph Nodes By R. Aaron Warnock,* Sanaz Askari, Eugene C. Butcher,* and Ulrich H. von Andrian From the *Laboratory of Immunology and Vascular Biology,
Molecular Mechanisms of Lymphocyte Homing to Peripheral Lymph Nodes By R. Aaron Warnock,* Sanaz Askari, Eugene C. Butcher,* and Ulrich H. von Andrian From the *Laboratory of Immunology and Vascular Biology,
Integrin 4 7 Antagonists: Activities, Mechanisms of Action and Therapeutic Prospects
 118 Current Immunology Reviews, 2012, 8, 118-134 Integrin 4 7 Antagonists: Activities, Mechanisms of Action and Therapeutic Prospects Dulce Soler-Ferran *,1 and Michael J. Briskin 2 1 Former Affiliation:
118 Current Immunology Reviews, 2012, 8, 118-134 Integrin 4 7 Antagonists: Activities, Mechanisms of Action and Therapeutic Prospects Dulce Soler-Ferran *,1 and Michael J. Briskin 2 1 Former Affiliation:
Question 1. Kupffer cells, microglial cells and osteoclasts are all examples of what type of immune system cell?
 Abbas Chapter 2: Sarah Spriet February 8, 2015 Question 1. Kupffer cells, microglial cells and osteoclasts are all examples of what type of immune system cell? a. Dendritic cells b. Macrophages c. Monocytes
Abbas Chapter 2: Sarah Spriet February 8, 2015 Question 1. Kupffer cells, microglial cells and osteoclasts are all examples of what type of immune system cell? a. Dendritic cells b. Macrophages c. Monocytes
Immunology - Lecture 2 Adaptive Immune System 1
 Immunology - Lecture 2 Adaptive Immune System 1 Book chapters: Molecules of the Adaptive Immunity 6 Adaptive Cells and Organs 7 Generation of Immune Diversity Lymphocyte Antigen Receptors - 8 CD markers
Immunology - Lecture 2 Adaptive Immune System 1 Book chapters: Molecules of the Adaptive Immunity 6 Adaptive Cells and Organs 7 Generation of Immune Diversity Lymphocyte Antigen Receptors - 8 CD markers
T Lymphocyte Activation and Costimulation. FOCiS. Lecture outline
 1 T Lymphocyte Activation and Costimulation Abul K. Abbas, MD UCSF FOCiS 2 Lecture outline T cell activation Costimulation, the B7:CD28 family Inhibitory receptors of T cells Targeting costimulators for
1 T Lymphocyte Activation and Costimulation Abul K. Abbas, MD UCSF FOCiS 2 Lecture outline T cell activation Costimulation, the B7:CD28 family Inhibitory receptors of T cells Targeting costimulators for
