What s New in CIEDs. Keimyung University Dongsan Medical Center Hyoung-Seob Park
|
|
|
- Clement Page
- 5 years ago
- Views:
Transcription
1 What s New in CIEDs Keimyung University Dongsan Medical Center Hyoung-Seob Park
2
3 Transvenous CIEDs Technology: - Highly mature & reliable - Still includes generator, connectors and leads Procedure : - Surgical pocket + Transvenous leads Device issues Pocket - Discomfort, Hematomas, Infections, Cosmetic concerns Leads - Mechanical failures - Infections; Extractions - Mobility restrictions - Incompatibility with MRI
4 History of Leadless Pacing
5 History of Leadless Pacing a: Guiding catheter b: Pusher catheter c: Miniature pacemaker d: Steering arm
6 What s Needed for a Leadless Pacemaker? Catheter-based delivery system High density energy source Low power electronics Novel communication scheme Biocompatible materials Dependable fixation design Retrievability capability
7 Leadless Pacemakers
8 Energy efficient Nanostim Leadless Pacemaker The Nanostim VVIR pacemaker is introduced through the femoral vein into the right ventricle. High-capacity CFx battery Lower resistance due to lack of lead Low-power conductive communication Compatible with Merlin Patient Care System Electrode design is identical to a St. Jude Medical electrode with same steroid elution Designed to prevent dislodgement Double fixation: single turn helix (x 2 pullstrength) plus angled nylon sutures Radiographic indicator to ensure proper number of turns Tethered test mode for perioperative evaluation Designed for retrievability Catheter-based retrieval system
9 Nanostim Leadless Pacemaker
10 The Nanostim Leadless Pacemaker Delivery catheter Delivery System Soft, flexible, deflectable catheter tip designed to minimize complications Tethered feature Integrated protective sleeve 18 F Handle with four functions: Steering the deflectable tip Docking/undocking Rotating the device Releasing tether 18 F introducer
11 The Nanostim Leadless Pacemaker Retrieval System Similar to delivery system Flexible with deflectable tip Integrated protective sleeve 18 F Either single loop or triple loop snare Handle with three functions: Steering the deflectable tip for accurate passage Grabbing and docking the LP Rotating the LP
12 Leadless Pacemaker Implantation Reddy VY et al. Circulation. 2014;129:
13 Leadless Pacemaker Implantation Reddy VY et al. Circulation. 2014;129:
14 Leadless PMK at RV Base
15 Comparison with Conventional System Attribute Lead-based Pacemaker Leadless Pacemaker Implant procedure Surgical pocket + lead (7 F) Percutaneous femoral based delivery (18 F) Implant time minutes minutes 15 X-ray exposure For implanter: Next to the X-ray tube (shorter patient recovery) For implanter: Further away from the source Connections Lead-can connectors None Apparatus in vascular system (chronic) Yes (lead) No (leadless) Apparatus through tricuspid valve (chronic) Yes (lead) No (leadless) System removal Specialization required Removal tools available Longevity (2.5V, 0.4ms, 60 bpm) Accent SR Inductive for lead-based (500 Ω for Accent, 600 Ω for leadless) 100% pacing 11.2 years 75% pacing 11.8 years 50% pacing 12.5 years 25% pacing 13.3 years 100% pacing 9.8 years 75% pacing 11.7 years 50% pacing 14.5 years 25% pacing 18.9 years Battery Replacement Pocket access Femoral access: removal+ new implant Option for another adjacent implant MRI compatibility Conditional image impact MRI conditional status not yet determined Key Benefits for Nanostim LP
16 Leadless Trial Arrhythmia/Electrophysiology Permanent Leadless Cardiac Pacing Results of the LEADLESS Trial Vivek Y. Reddy, MD; Reinoud E. Knops, MD; Johannes Sperzel, MD; Marc A. Miller, MD; Jan Petru, MD; Jaroslav Simon, MD; Lucie Sediva, MD; Joris R. de Groot, MD, PhD; Fleur V.Y. Tjong, MD; Peter Jacobson, BS; Alan Ostrosff, MS; Srinivas R. Dukkipati, MD; Jacob S. Koruth, MD; Arthur A.M. Wilde, MD, PhD; Josef Kautzner, MD, PhD; Petr Neuzil, MD, PhD Background Conventional cardiac pacemakers are associated with several potential short- and long-term complications related to either the transvenous lead or subcutaneous pulse generator. We tested the safety and clinical performance of a novel, completely self-contained leadless cardiac pacemaker. Methods and Results The primary safety end point was freedom from complications at 90 days. Secondary performance end points included implant success rate, implant time, and measures of device performance (pacing/sensing thresholds and rate-responsive performance). The mean age of the patient cohort (n=33) was 77±8 years, and 67% of the patients were male (n=22/33). The most common indication for cardiac pacing was permanent atrial fibrillation with atrioventricular block (n=22, 67%). The implant success rate was 97% (n=32). Five patients (15%) required the use of >1 leadless cardiac pacemaker during the procedure. One patient developed right ventricular perforation and cardiac tamponade during the implant procedure, and eventually died as the result of a stroke. The overall complication-free rate was 94% (31/33). After 3 months of follow-up, the measures of pacing performance (sensing, impedance, and pacing threshold) either improved or were stable within the accepted range. Conclusions In a prospective nonrandomized study, a completely self-contained, single-chamber leadless cardiac pacemaker has shown to be safe and feasible. The absence of a transvenous lead and subcutaneous pulse generator could represent a paradigm shift in cardiac pacing. Clinical Trial Registration URL: Unique identifier: NCT (Circulation. 2014;129: ) Key Words: pacemaker, cardiac Nearly new cardiac pacemakers are implanted annually in the United States, and an additional Reddy VY et al. Circulation. 2014;129: accompany these complications of conventional cardiac pacing systems, there are also significant incremental costs associated with each of these untoward outcomes. 5 Although it has
17 Demographics and Procedural Details Reddy VY et al. Circulation. 2014;129:
18 Safety Endpoint 1 Minor Groin Hematoma, no treatment Cardiac Perforation & Tamponade - 70 year-old male with chronic AF, INR 2.1 at implant - Uncomplicated Surgical Repair - But during convalescence (5 days post-op) - Large right-sided stroke (INR = 1.5) - Expired
19 Device Performance Measurements Reddy VY et al. Circulation. 2014;129:
20
21
22 The LEADLESS Pacemaker IDE Study (Leadless II) Evaluate safety and effectiveness of the Nanostim leadless pacemaker in patients indicated for VVIR pacemaker Prospective, single-arm study Estimated Enrollment : 667 patients Primary Safety Endpoint Complication Free Rate (Up to 6 months post implant) Primary Effectiveness Endpoint Pacing Thresholds and R-wave amplitudes at 6 months
23 Micra Transcatheter Pacing System (TPS) 25.9 mm, < 1cc miniaturized VVIR pacemaker (Adapta pacemaker ~10cc) World s smallest, minimally invasive pacing system 10 year longevity Percutaneous access to RV apex via femoral vein Active fixation via 4 self-expanding tines
24 Technical Overview: Pacing Capsule VVIR Volume: 0.75cc / Mass: 2g Programmable Bipolar sensing; 17mm spacing Capture Management Cathode 2.5 mm 2 CapSure Sense Electrical Isolation Parylene Anode 53 mm2 TiN Coated Essential diagnostics (i.e., battery status, pacing threshold, pacing impedance, % paced) Fixation Tines (electrially inactive) Titanium Enclosure 24 mm length 20 Fr Tether / Retrieval Feature 7 yr longevity (100% paced V pacing) Device can be inactivated at the end of battery life MR-conditional-MRI Scans RF communication with programmer and CareLink
25 Technical Overview: Delivery System Non-OTW; 24F introducer sheath to RA Distal delivery cup to hold device; 22F 16F proximal shaft Fixed shape proximal curve Off-plane distal articulation 105cm working length (femoral) Tether Distal delivery cup to hold device; 22F Implant Tether Facilitates fixation verification (Tug Test) Mechanism for re-capture of PLP Device Removable Medical Grade Suture material: PET with PTFE coating (braided)
26 Micra Transcatheter Pacing Study ClinicalTrials.gov Identifier : NCT Single arm, multi-center global clinical trial Estimated Enrollment : 780 patients Study Start Date : November 2013 Estimated Study Completion Date : June 2018 At ~50 centers Primary Outcomes at 6 months : - Safety : Major complication-free rate - Efficacy : Low and stable pacing capture thresholds
27 The S-ICD System
28 Design Goals of Subcutaneous ICD Therapy To avoid both the short- and long-term complications associated with transvenous leads To defibrillate with more uniform voltage gradients, reducing myocardial damage To sense activation across the whole heart, improving accuracy for arrhythmia detection. To provide an option for patient sub-populations for which TV-ICD is not ideal To reduce risk of lead failure in young and active patients
29 Effective defibrillation without transvenous leads The S-ICD System: Entirely subcutaneous Does not require leads in the heart, leaving the vasculature untouched Placed strictly by anatomical landmarks, removing the need for fluoroscopy at implant Sophisticated algorithms provide performance equal to transvenous ICDs 1 1) Burke M, et al. Safety and Efficacy of a Subcutaneous Implantable-Defibrillator (S-ICD System US IDE Study). Late-Breaking Abstract Session. HRS Gold M, et al. Head-to-Head Comparison of Arrhythmia Discrimination Performance of Subcutaneous and Transvenous ICD Arrhythmia Detection Algorithms: The START Study. Journal of Cardiovascular Electrophysiology; Vol 23:4( )
30 The S-ICD System Provides effective defibrillation for ventricular tachyarrhythmias No risk of vascular injury Low risk of systemic infection Preserves Venous access Avoids risks associated with endovascular lead extraction Fluoroscopy not required
31 The S-ICD System 80 joule (delivered) biphasic shock 10 seconds charge time to 80J 5.1 year projected longevity 30 seconds post-shock pacing Single electrode connection Full featured episode storage
32
33 Four Configulation of a S-ICD Bardy GH et al. N Engl J Med. 2010;363:36-44.
34 Location of the Components of a S-ICD Bardy GH et al. N Engl J Med. 2010;363:36-44.
35 S-ICD System Components SQ-RX TM Pulse Generator Q-GUIDE Electrode Insertion Tool Single use tool 36cm total length 3mm shaft diameter Volume: 69 cc Weight: 145 grams Thickness: 15.7 mm Energy: 80J (delivered) Waveform: Biphasic Q-TRAK TM Electrode Multistrand cable-core design No hollow core, no inner coils Durable polyurethane insulator Designed to withstand cardiopulmonary resusci tation (CPR) forces Q-TECH Tablet Programmer AC powered/battery backup Wanded RF telemetry Wireless printing Micro SD card
36 Implant Procedure
37 Ideal Device Placement
38 Rhythm Detection Technology Three far-field sensing vectors Primary, Secondary, Alternate Automatic or manual vector selection Morphologically rich signal similar to a surface ECG Sense electrodes positioned away from large muscle groups Maximum flexibility to solve sensing issues non invasively ALTERNATE Sense vector reprogramming
39 Episodes Up to 5 shocks per 80J Up to 128 seconds of S-ECG storage per episode Storage of up to 44 episode Therapy Delivery Adaptive Shock Polarity System remembers the polarity of the last successful shock and automatically selects this shock polarity for the first shock of an episode Shock Vector Encompasses the entire left chest Tolerant of a wide variety of cardiac sizes/ orientation/hypertrophy
40 Therapy Delivery Post-Shock Pacing Transthoracic pacing Delivered for up to 30 seconds post-shock Demand based 50 ppm using 200 ma
41
42 Patient Distribution Weiss R et al. Circulation. 2013;128:
43 Effectiveness and Safety Endpoints Effectiveness Endpoints Met 100% conversion of induced arrhythmias in evaluable patients Safety Endpoints Met 99% Free from S-ICD TM System complications Both endpoints met even under worst case sensitivity analysis Spontaneous VF/VT Episodes Complications 119 events in 21 patients 100% converted with 80J or spontaneousl y converted 92% first shock conversion efficacy 4.4% perioperative complication rate 4 explant for infection (first 1/3 of pts) No systemic infection or endocarditis No arrhythmic deaths Weiss R et al. Circulation. 2013;128:
44 Complication Free Rate Weiss R et al. Circulation. 2013;128:
45
46 EFFORTLESS Registry 472 patients enrolled between Feb 2011 and April % primary prevention typical for EU - 49 ± 18 years (range 9 88) younger demographic day mean follow-up (range days) 232 patients enrolled in QOL sub-study (not yet published) Lambiase PD et al. Eur Heart J. 2014;35:
47 EFFORTLESS Registry Lambiase PD et al. Eur Heart J. 2014;35:
48 Inappropriate Therapy IAS & Programming Reasons for IAS p = % of patients received dual-zone programming; with an inappropriate shock rate of 6.4%. Only 9 study patients (2%) experienced inappropriate shocks after initial interventions (re-programming, exercise tests, medication changes). Only one VF/SVT discrimination error in the conditional shock zone.
49 Appropriate Shock for Clinical Event of VF
50 CONTROVERSIES IN ARRHYTHMIA AND ELECTROPHYSIOLOGY Who Should Receive the Subcutaneous Implanted Defibrillator? The Subcutaneous Implantable Cardioverter Defibrillator (ICD) Should Be Considered in all ICD Patients Who Do Not Require Pacing Jeanne E. Poole, MD; Michael R. Gold, MD, PhD The evolution of implantable cardioverter defibrillator (ICD) technology for the past 3 decades has been nothing short of explosive, incorporating progressively transvenous leads, multizone programming, dual chamber antibradycardia pacing, antitachycardia pacing (ATP), sophisticated single- and dual-chamber discrimination algorithms, cardiac resynchronization therapy (CRT), and programmable options numbering into the thousands. As a consequence, ICDs have been used to treat patients with a variety of clinical needs, including those with a known history of ventricular tachycardia (VT), survivors of out of hospital cardiac arrest, patients with requirements for pacing or resynchronization with concomitant indications for an ICD, and patients who do not fit within these categories but are at risk for sudden cardiac death (SCD). (VF). In fact, more complex therapies are not always better for patient outcomes. The Dual Chamber and VVI Implantable Defibrillator (DAVID) trial showed that indiscriminate right ventricular pacing is associated with increased risk of heart failure hospitalization and death, and more recent analyses continue to question the overuse of dual chamber devices. 1,2 Similarly, CRT may be associated with more heart failure hospitalization among patients without QRS prolongation. 3 The landmark prospective randomized trials showed a mortality benefit of ICDs for both primary and secondary prevention of SCD. The early secondary prevention trials, such as Antiarrhythmics versus Implantable Defibrillator (AVID) and Canadian Implantable Defibrillator Study (CIDS), used both epicardial and transvenous lead systems, with devices capable of limited programming with little or no pacing. 4,5 Poole JE et al. Circ Arrhythm Electrophysiol. 2013;6:
51 Thanks for your attention!!
New generations pacemakers and ICDs: an update
 Advances in Cardiac Arrhythmias and Great Innovations in Cardiology XXVII Giornate Cardiologiche Torinesi New generations pacemakers and ICDs: an update Prof. Fiorenzo Gaita, MD Division of Cardiology
Advances in Cardiac Arrhythmias and Great Innovations in Cardiology XXVII Giornate Cardiologiche Torinesi New generations pacemakers and ICDs: an update Prof. Fiorenzo Gaita, MD Division of Cardiology
NOVEL DEVICE TECHNOLOGIES
 NOVEL DEVICE TECHNOLOGIES Leadless Pacemakers and Subcutaneous ICDs Do the Benefits Outweigh MRI Incompatibility? Disclosures None Background PPMs and ICDs are very effective therapy for treating bradyarrhythmias
NOVEL DEVICE TECHNOLOGIES Leadless Pacemakers and Subcutaneous ICDs Do the Benefits Outweigh MRI Incompatibility? Disclosures None Background PPMs and ICDs are very effective therapy for treating bradyarrhythmias
INNOVATIONS IN DEVICE THERAPY:
 INNOVATIONS IN DEVICE THERAPY: Subcutaneous ICDs, Leadless Pacemakers, CRT Indications David J Wilber MD Loyola University Medical Center Disclosures: ACC Foundation: Consultant; Biosense / Webster: Consultant,
INNOVATIONS IN DEVICE THERAPY: Subcutaneous ICDs, Leadless Pacemakers, CRT Indications David J Wilber MD Loyola University Medical Center Disclosures: ACC Foundation: Consultant; Biosense / Webster: Consultant,
Lead Selection and Subcutaneous ICD Considerations. The Future of CIEDs
 Lead Selection and Subcutaneous ICD Considerations The Future of CIEDs February, 2013 Steven P. Kutalek, MD Director, Cardiac Electrophysiology Drexel University College of Medicine New Technologies 2012
Lead Selection and Subcutaneous ICD Considerations The Future of CIEDs February, 2013 Steven P. Kutalek, MD Director, Cardiac Electrophysiology Drexel University College of Medicine New Technologies 2012
Leadless Pacing. Osama Diab Assistant Prof. of Cardiology Ain Shams University Egypt
 Leadless Pacing Osama Diab Assistant Prof. of Cardiology Ain Shams University Egypt The weakest link in Pacemaker system the lead. The more the leads the more the complications Dislodgement Fracture Insulation
Leadless Pacing Osama Diab Assistant Prof. of Cardiology Ain Shams University Egypt The weakest link in Pacemaker system the lead. The more the leads the more the complications Dislodgement Fracture Insulation
Update on Device Innovation (S-ICD, Wearable, Leadless)
 Update on Device Innovation (S-ICD, Wearable, Leadless) C. W. Israel Dept. of Medicine - Cardiology Evangelical Hospital Bielefeld J. W. Goethe University Frankfurt Carsten.Israel@em.uni-frankfurt.de Conflicts
Update on Device Innovation (S-ICD, Wearable, Leadless) C. W. Israel Dept. of Medicine - Cardiology Evangelical Hospital Bielefeld J. W. Goethe University Frankfurt Carsten.Israel@em.uni-frankfurt.de Conflicts
ICD: Basics, Programming and Trouble-shooting
 ICD: Basics, Programming and Trouble-shooting Amir AbdelWahab, MD Electrophysiology and Pacing Service Cardiology Department Cairo University Feb 2013 Evolution of ICD Technology ICD Evolution Indications
ICD: Basics, Programming and Trouble-shooting Amir AbdelWahab, MD Electrophysiology and Pacing Service Cardiology Department Cairo University Feb 2013 Evolution of ICD Technology ICD Evolution Indications
MEET MICRA. Micra TM ACTUAL SIZE. Transcatheter Pacing System
 MEET MICRA ACTUAL SIZE Micra TM Transcatheter Pacing System MEET MICRA The world s smallest pacemaker 1 MINIATURIZED. 93% smaller than modern-day pacemakers 6 Completely self contained within the heart,
MEET MICRA ACTUAL SIZE Micra TM Transcatheter Pacing System MEET MICRA The world s smallest pacemaker 1 MINIATURIZED. 93% smaller than modern-day pacemakers 6 Completely self contained within the heart,
MEET MICRA. Micra TM ACTUAL SIZE. Transcatheter Pacing System
 MEET MICRA ACTUAL SIZE Micra TM Transcatheter Pacing System MEET MICRA The world s smallest pacemaker 1 MINIATURIZED. 93% smaller than modern-day pacemakers 7 Completely self contained within the heart,
MEET MICRA ACTUAL SIZE Micra TM Transcatheter Pacing System MEET MICRA The world s smallest pacemaker 1 MINIATURIZED. 93% smaller than modern-day pacemakers 7 Completely self contained within the heart,
Pediatric pacemakers & ICDs:
 Pediatric pacemakers & ICDs: perioperative management Manchula Navaratnam Clinical Assistant Professor LPCH, Stanford SPA 2016 Conflict of interest: none Objectives Indications in pediatrics Components
Pediatric pacemakers & ICDs: perioperative management Manchula Navaratnam Clinical Assistant Professor LPCH, Stanford SPA 2016 Conflict of interest: none Objectives Indications in pediatrics Components
Essentials of Pacemakers and ICD s. Rajesh Banker, MD, MPH
 Essentials of Pacemakers and ICD s Rajesh Banker, MD, MPH Pacemakers have 4 basic functions: Stimulate cardiac depolarization Sense intrinsic cardiac function Respond to increased metabolic demand by providing
Essentials of Pacemakers and ICD s Rajesh Banker, MD, MPH Pacemakers have 4 basic functions: Stimulate cardiac depolarization Sense intrinsic cardiac function Respond to increased metabolic demand by providing
Supplemental Material
 Supplemental Material 1 Table S1. Codes for Patient Selection Cohort Codes Primary PM CPT: 33206 or 33207 or 33208 (without 33225) ICD-9 proc: 37.81, 37.82, 37.83 Primary ICD Replacement PM Replacement
Supplemental Material 1 Table S1. Codes for Patient Selection Cohort Codes Primary PM CPT: 33206 or 33207 or 33208 (without 33225) ICD-9 proc: 37.81, 37.82, 37.83 Primary ICD Replacement PM Replacement
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE INTERVENTIONAL PROCEDURES PROGRAMME Interventional procedure overview of a leadless cardiac pacemaker implantation for bradyarrhythmias Bradyarrhythmias
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE INTERVENTIONAL PROCEDURES PROGRAMME Interventional procedure overview of a leadless cardiac pacemaker implantation for bradyarrhythmias Bradyarrhythmias
Subcutaneous implantable cardioverter-defibrillator (S-ICD)
 NEW DRUGS AND TECHNOLOGIES IN CARDIOLOGY Cardiology Journal 2011, Vol. 18, No. 3, pp. 326 331 Copyright 2011 Via Medica ISSN 1897 5593 Subcutaneous implantable cardioverter-defibrillator (S-ICD) S. Suave
NEW DRUGS AND TECHNOLOGIES IN CARDIOLOGY Cardiology Journal 2011, Vol. 18, No. 3, pp. 326 331 Copyright 2011 Via Medica ISSN 1897 5593 Subcutaneous implantable cardioverter-defibrillator (S-ICD) S. Suave
Subcutaneous implantable cardioverter defibrillator insertion for preventing sudden cardiac death
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Interventional procedure consultation document Subcutaneous implantable cardioverter defibrillator insertion for preventing sudden cardiac death A subcutaneous
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Interventional procedure consultation document Subcutaneous implantable cardioverter defibrillator insertion for preventing sudden cardiac death A subcutaneous
Leadless pacing: Technical challenges and concerns. Panos Vardas President Elect of the ESC, Prof. of Cardiology, Heraklion University Hospital
 Leadless pacing: Technical challenges and concerns Panos Vardas President Elect of the ESC, Prof. of Cardiology, Heraklion University Hospital Modest consultancy fees/honoraria from Bayer Boehringer Ingelheim
Leadless pacing: Technical challenges and concerns Panos Vardas President Elect of the ESC, Prof. of Cardiology, Heraklion University Hospital Modest consultancy fees/honoraria from Bayer Boehringer Ingelheim
News der kardialen Device - Therapie! Christian Binggeli! Rhythmologie Klinik St. Anna, Luzern! HerzGefässZentrum Zürich, KlinikImPark, Zürich!
 News der kardialen Device - Therapie! Christian Binggeli! Rhythmologie Klinik St. Anna, Luzern! HerzGefässZentrum Zürich, KlinikImPark, Zürich! Complications after Implantation / Replacement / Upgrade
News der kardialen Device - Therapie! Christian Binggeli! Rhythmologie Klinik St. Anna, Luzern! HerzGefässZentrum Zürich, KlinikImPark, Zürich! Complications after Implantation / Replacement / Upgrade
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE INTERVENTIONAL PROCEDURES PROGRAMME Interventional procedure overview of leadless cardiac pacemaker implantation for bradyarrhythmias Bradyarrhythmias
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE INTERVENTIONAL PROCEDURES PROGRAMME Interventional procedure overview of leadless cardiac pacemaker implantation for bradyarrhythmias Bradyarrhythmias
Leadless pacemakers and subcutaneous ICD s: will we use them for most of our patients? K.-H. Kuck Asklepios Klinik St. Georg Hamburg, Germany
 Leadless pacemakers and subcutaneous ICD s: will we use them for most of our patients? K.-H. Kuck Asklepios Klinik St. Georg Hamburg, Germany Disclosure Statement Research Grants Consultant / Advisory
Leadless pacemakers and subcutaneous ICD s: will we use them for most of our patients? K.-H. Kuck Asklepios Klinik St. Georg Hamburg, Germany Disclosure Statement Research Grants Consultant / Advisory
Leadless pacing. P.E. Vardas Professor of Cardiology University of Crete, Greece
 Leadless pacing P.E. Vardas Professor of Cardiology University of Crete, Greece DISCLOSURES Small teaching fees from: Bayer/J&J, Boeringer, Menarini Small consulting fees from: Medtronic 1957: First External
Leadless pacing P.E. Vardas Professor of Cardiology University of Crete, Greece DISCLOSURES Small teaching fees from: Bayer/J&J, Boeringer, Menarini Small consulting fees from: Medtronic 1957: First External
Device Interrogation- Pacemakers, ICD and Loop Recorders. Dulce Obias-Manno, RN, MHSA, CCDS,CEPS, FHRS Device Clinic Coordinator, MHVI
 Device Interrogation- Pacemakers, ICD and Loop Recorders Dulce Obias-Manno, RN, MHSA, CCDS,CEPS, FHRS Device Clinic Coordinator, MHVI Disclosures Consultant: Medtronic Speaker s Bureau: St. Jude Medical
Device Interrogation- Pacemakers, ICD and Loop Recorders Dulce Obias-Manno, RN, MHSA, CCDS,CEPS, FHRS Device Clinic Coordinator, MHVI Disclosures Consultant: Medtronic Speaker s Bureau: St. Jude Medical
What You Should Know About Subcutaneous and Transvenous ICD
 What You Should Know About Subcutaneous and Transvenous ICD N. A. Mark Estes III MD Professor of Medicine Tufts University School of Medicine Director, New England Cardiac Arrhythmia Center Tufts Medical
What You Should Know About Subcutaneous and Transvenous ICD N. A. Mark Estes III MD Professor of Medicine Tufts University School of Medicine Director, New England Cardiac Arrhythmia Center Tufts Medical
NEIL CISPER TECHNICAL FIELD ENGINEER ICD/CRTD BASICS
 NEIL CISPER TECHNICAL FIELD ENGINEER ICD/CRTD BASICS OBJECTIVES Discuss history of ICDs Review the indications for ICD and CRT therapy Describe basic lead and device technology Discuss different therapies
NEIL CISPER TECHNICAL FIELD ENGINEER ICD/CRTD BASICS OBJECTIVES Discuss history of ICDs Review the indications for ICD and CRT therapy Describe basic lead and device technology Discuss different therapies
Advanced ICD Concepts
 1 4 2 5 7 3 6 8 Advanced ICD Concepts This presentation is provided with the understanding that the slide content must not be altered in any manner as the content is subject to FDA regulations. This presentation
1 4 2 5 7 3 6 8 Advanced ICD Concepts This presentation is provided with the understanding that the slide content must not be altered in any manner as the content is subject to FDA regulations. This presentation
Top 5 Things to Know about Pacemakers and ICD s. Jeffrey S. Osborn, M.D., C.C.D.S. March 4, 2017.
 Top 5 Things to Know about Pacemakers and ICD s Jeffrey S. Osborn, M.D., C.C.D.S. March 4, 2017. Top 5 Things 1. Remote Monitoring leads to better care and outcomes. 2. MRI s CAN be done on device patients.
Top 5 Things to Know about Pacemakers and ICD s Jeffrey S. Osborn, M.D., C.C.D.S. March 4, 2017. Top 5 Things 1. Remote Monitoring leads to better care and outcomes. 2. MRI s CAN be done on device patients.
Active Fixation Models: 7740, 7741 and 7742 Passive Fixation Models: 7731, 7732, 7735 and 7736
 The INGEVITY MRI pacing leads are 6F (2.0 mm) steroid-eluting, endocardial pace / sense leads designed for permanent implantation for either atrial or ventricular applications. INGEVITY MRI is the only
The INGEVITY MRI pacing leads are 6F (2.0 mm) steroid-eluting, endocardial pace / sense leads designed for permanent implantation for either atrial or ventricular applications. INGEVITY MRI is the only
Call Medtronic at 1 (800) to verify the patient s current implanted system
 MRI SURESCAN SYSTEMS Patient Scanning Process Transvenous Implantable Cardiac Systems PATIENT PRESCREENING SureScan Pacing, Defibrillation, and CRT (CRT-D and CRT-P) Systems Verification Verify that patient
MRI SURESCAN SYSTEMS Patient Scanning Process Transvenous Implantable Cardiac Systems PATIENT PRESCREENING SureScan Pacing, Defibrillation, and CRT (CRT-D and CRT-P) Systems Verification Verify that patient
Recent Advances in Pacing and Defibrillation Harish Doppalapudi, MD
 Recent Advances in Pacing and Defibrillation Harish Doppalapudi, MD Harish Doppalapudi, MD Assistant Professor of Medicine Director, Clinical Cardiac Electrophysiology Training Program University of Alabama
Recent Advances in Pacing and Defibrillation Harish Doppalapudi, MD Harish Doppalapudi, MD Assistant Professor of Medicine Director, Clinical Cardiac Electrophysiology Training Program University of Alabama
Leadless pacemakers a panacea for bradyarrhythmias?
 Leadless pacemakers a panacea for bradyarrhythmias? Peysh A Patel Take Home Messages Why may leadless systems be required? Where the cessation of vital action is very complete, and continues long, we ought
Leadless pacemakers a panacea for bradyarrhythmias? Peysh A Patel Take Home Messages Why may leadless systems be required? Where the cessation of vital action is very complete, and continues long, we ought
Subcutaneous ICD Emerging Role of Sudden Cardiac Death Prevention
 Subcutaneous ICD Emerging Role of Sudden Cardiac Death Prevention Dr Ngai-Yin Chan, MBBS, FRCP(Lond), FRCP(Edin), FRCP(Glasg), FACC, FHRS, Consultant Physician, Department of Medicine & Geriatrics, Princess
Subcutaneous ICD Emerging Role of Sudden Cardiac Death Prevention Dr Ngai-Yin Chan, MBBS, FRCP(Lond), FRCP(Edin), FRCP(Glasg), FACC, FHRS, Consultant Physician, Department of Medicine & Geriatrics, Princess
Subcutaneous Implantable Cardioverter Defibrillator (S-ICD)
 Subcutaneous Implantable Cardioverter Defibrillator (S-ICD) D. D. MANOLATOS, MD, PhD, FESC Electrophysiology and Device Lab General Hospital Evangelismos, Athens The Problem: 300,000 people die each year
Subcutaneous Implantable Cardioverter Defibrillator (S-ICD) D. D. MANOLATOS, MD, PhD, FESC Electrophysiology and Device Lab General Hospital Evangelismos, Athens The Problem: 300,000 people die each year
Tachycardia Devices Indications and Basic Trouble Shooting
 Tachycardia Devices Indications and Basic Trouble Shooting Peter A. Brady, MD., FRCP Cardiology Review Course London, March 6 th, 2014 2011 MFMER 3134946-1 Tachycardia Devices ICD Indications Primary and
Tachycardia Devices Indications and Basic Trouble Shooting Peter A. Brady, MD., FRCP Cardiology Review Course London, March 6 th, 2014 2011 MFMER 3134946-1 Tachycardia Devices ICD Indications Primary and
M Series with Rectilinear Biphasic Waveform Defibrillator Option Indications for Use
 DEFIBRILLATOR OPTION General Information Introduction M Series products are available with an advanced electrical design that provides a unique rectilinear biphasic waveform for defibrillation and cardioversion.
DEFIBRILLATOR OPTION General Information Introduction M Series products are available with an advanced electrical design that provides a unique rectilinear biphasic waveform for defibrillation and cardioversion.
REVIEW ARTICLE. Leadless Cardiac Pacemaker Therapy. An Overview for the Hospitalist Richard Weachter 1
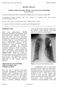 Leadless Cardiac Pacemaker Therapy. An Overview for the Hospitalist Richard Weachter 1 1 Division of Cardiovascular Medicine, Department of Medicine, University of Missouri, Columbia, MO Correspondence:
Leadless Cardiac Pacemaker Therapy. An Overview for the Hospitalist Richard Weachter 1 1 Division of Cardiovascular Medicine, Department of Medicine, University of Missouri, Columbia, MO Correspondence:
We are IntechOpen, the world s leading publisher of Open Access books Built by scientists, for scientists. International authors and editors
 We are IntechOpen, the world s leading publisher of Open Access books Built by scientists, for scientists 4,000 116,000 120M Open access books available International authors and editors Downloads Our
We are IntechOpen, the world s leading publisher of Open Access books Built by scientists, for scientists 4,000 116,000 120M Open access books available International authors and editors Downloads Our
Dual-Chamber Implantable Cardioverter-Defibrillator
 February 1998 9 Dual-Chamber Implantable Cardioverter-Defibrillator A.SH. REVISHVILI A.N. Bakoulev Research Center for Cardiovascular Surgery, Russian Academy of Medical Sciences, Moscow, Russia Summary
February 1998 9 Dual-Chamber Implantable Cardioverter-Defibrillator A.SH. REVISHVILI A.N. Bakoulev Research Center for Cardiovascular Surgery, Russian Academy of Medical Sciences, Moscow, Russia Summary
UnitedHealthcare Medicare Advantage Cardiology Prior Authorization Program
 Electrophysiology Implant Classification Table The table below contains the codes that apply to our UnitedHealthcare Medicare Advantage cardiology prior Description Includes Generator Placement Includes
Electrophysiology Implant Classification Table The table below contains the codes that apply to our UnitedHealthcare Medicare Advantage cardiology prior Description Includes Generator Placement Includes
SUPPLEMENTAL MATERIAL
 SUPPLEMENTAL MATERIAL Table S1. Patient Selection Codes, CIED Generator Procedures Code Type Code Description ICD9 Proc 00.51 Implantation of cardiac resynchronization defibrillator, total system [CRT-D]
SUPPLEMENTAL MATERIAL Table S1. Patient Selection Codes, CIED Generator Procedures Code Type Code Description ICD9 Proc 00.51 Implantation of cardiac resynchronization defibrillator, total system [CRT-D]
Implantable Cardioverter-Defibril. Defibrillators. Ratko Magjarević
 Implantable Cardioverter-Defibril Defibrillators Ratko Magjarević University of Zagreb Faculty of Electrical Engineering and Computing Croatia ratko.magjarevic@fer.hr Ventricular Fibrillation Ventricular
Implantable Cardioverter-Defibril Defibrillators Ratko Magjarević University of Zagreb Faculty of Electrical Engineering and Computing Croatia ratko.magjarevic@fer.hr Ventricular Fibrillation Ventricular
SVT Discriminators. Definition of SVT Discrimination. Identify which patient populations might benefit from these features
 Definition of SVT Discrimination Identify which patient populations might benefit from these features Understand the 4 types of SVT discriminators used by St Jude Medical Be aware of programmable parameters
Definition of SVT Discrimination Identify which patient populations might benefit from these features Understand the 4 types of SVT discriminators used by St Jude Medical Be aware of programmable parameters
The EFFORTLESS SICD Registry. The EFFORTLESS SICD Registry. SICD Implant 11/14/2016
 11/14/2016 VuMedi Webinar: Safety and Efficacy of the Subcutaneous ICD Primary Results of The EFFORTLESS SICD Registry November 21, 2016 Philip Chang, MD, FACC, FHRS Assistant Professor of Medicine and
11/14/2016 VuMedi Webinar: Safety and Efficacy of the Subcutaneous ICD Primary Results of The EFFORTLESS SICD Registry November 21, 2016 Philip Chang, MD, FACC, FHRS Assistant Professor of Medicine and
Diagnostic capabilities of the implantable therapeutic systems
 Cardiac pacing 2012 and beyound Monday August 27, 2012 Diagnostic capabilities of the implantable therapeutic systems Pekka Raatikainen Heart Center Co. Tampere University Hospital and University of Tampere
Cardiac pacing 2012 and beyound Monday August 27, 2012 Diagnostic capabilities of the implantable therapeutic systems Pekka Raatikainen Heart Center Co. Tampere University Hospital and University of Tampere
Tech Corner. ATP in the Fast VT zone
 Tech Corner ATP in the Fast VT zone NOTE: PLEASE NOTE THAT THE FOLLOWING INFORMATION IS A GENERAL DESCRIPTION OF THE FUNCTION. DETAILS AND PARTICULAR CASES ARE NOT DESCRIBED IN THE ARTICLE. FOR ADDITIONAL
Tech Corner ATP in the Fast VT zone NOTE: PLEASE NOTE THAT THE FOLLOWING INFORMATION IS A GENERAL DESCRIPTION OF THE FUNCTION. DETAILS AND PARTICULAR CASES ARE NOT DESCRIBED IN THE ARTICLE. FOR ADDITIONAL
The Nuts and Bolts of ICD Therapy
 Electrical Management of Cardiac Rhythm Disorders For Cardiology Fellows December 5-8 Austin, Texas The Nuts and Bolts of ICD Therapy 1 2 Action Potential Localized Differences in Conduction Conduction
Electrical Management of Cardiac Rhythm Disorders For Cardiology Fellows December 5-8 Austin, Texas The Nuts and Bolts of ICD Therapy 1 2 Action Potential Localized Differences in Conduction Conduction
Cardiac Implanted Electronic Devices Pacemakers, Defibrillators, Cardiac Resynchronization Devices, Loop Recorders, etc.
 Cardiac Implanted Electronic Devices Pacemakers, Defibrillators, Cardiac Resynchronization Devices, Loop Recorders, etc. The Miracle of Living February 21, 2018 Matthew Ostrom MD,FACC,FHRS Division of
Cardiac Implanted Electronic Devices Pacemakers, Defibrillators, Cardiac Resynchronization Devices, Loop Recorders, etc. The Miracle of Living February 21, 2018 Matthew Ostrom MD,FACC,FHRS Division of
ΤΟ ΗΚΓ ΣΤΟΝ ΒΗΜΑΤΟΔΟΤΟΥΜΕΝΟ ΑΣΘΕΝΗ
 ΤΟ ΗΚΓ ΣΤΟΝ ΒΗΜΑΤΟΔΟΤΟΥΜΕΝΟ ΑΣΘΕΝΗ ΤΖΩΡΤΖ ΔΑΔΟΥΣ ΕΠΙΚΟΥΡΟΣ ΚΑΘΗΓΗΤΗΣ Α.Π.Θ. ΜΑΡΙΑ ΚΑΡΑΛΙΟΛΙΟΥ ΕΙΔΙΚΕΥΟΜΕΝΗ ΙΑΤΡΟΣ ΚΑΡΔΙΟΛΟΓΟΣ Β ΚΑΡΔΙΟΛΟΓΙΚΗ ΚΛΙΝΙΚΗ Α.Π.Θ. ΙΠΠΟΚΡΑΤΕΙΟ Γ.Ν.Θ. 5 Why a Pacemaker is Implanted
ΤΟ ΗΚΓ ΣΤΟΝ ΒΗΜΑΤΟΔΟΤΟΥΜΕΝΟ ΑΣΘΕΝΗ ΤΖΩΡΤΖ ΔΑΔΟΥΣ ΕΠΙΚΟΥΡΟΣ ΚΑΘΗΓΗΤΗΣ Α.Π.Θ. ΜΑΡΙΑ ΚΑΡΑΛΙΟΛΙΟΥ ΕΙΔΙΚΕΥΟΜΕΝΗ ΙΑΤΡΟΣ ΚΑΡΔΙΟΛΟΓΟΣ Β ΚΑΡΔΙΟΛΟΓΙΚΗ ΚΛΙΝΙΚΗ Α.Π.Θ. ΙΠΠΟΚΡΑΤΕΙΟ Γ.Ν.Θ. 5 Why a Pacemaker is Implanted
Transvenous Pacemaker Procedures
 Cardiology: Pacemaker and Defibrillator Coding Presented By: Moderate Sedation 2017 99151 : under age 5, initial 15 minutes by MD performing intervention 99152: age 5 or older, initial 15 minutes by MD
Cardiology: Pacemaker and Defibrillator Coding Presented By: Moderate Sedation 2017 99151 : under age 5, initial 15 minutes by MD performing intervention 99152: age 5 or older, initial 15 minutes by MD
ESC Stockholm Arrhythmias & pacing
 ESC Stockholm 2010 Take Home Messages for Practitioners Arrhythmias & pacing Prof. Panos E. Vardas Professor of Cardiology Heraklion University Hospital Crete, Greece Disclosures Small teaching fees from
ESC Stockholm 2010 Take Home Messages for Practitioners Arrhythmias & pacing Prof. Panos E. Vardas Professor of Cardiology Heraklion University Hospital Crete, Greece Disclosures Small teaching fees from
Pediatric Pacemaker Implantation Endocardial or Epicardial
 Pediatric Pacemaker Implantation Endocardial or Epicardial HAITHAM BADRAN, MD, FEHRA CONSULTANT OF INTERVENTIONAL CARDIOLOGY CONSULTANT OF CARDIAC PACING AND ELECTROPHYSIOLOGY LECTURER OF CARDIOLOGY AIN
Pediatric Pacemaker Implantation Endocardial or Epicardial HAITHAM BADRAN, MD, FEHRA CONSULTANT OF INTERVENTIONAL CARDIOLOGY CONSULTANT OF CARDIAC PACING AND ELECTROPHYSIOLOGY LECTURER OF CARDIOLOGY AIN
PERIOPERATIVE MANAGEMENT: CARDIAC PACEMAKERS AND DEFIBRILLATORS
 PERIOPERATIVE MANAGEMENT: CARDIAC PACEMAKERS AND DEFIBRILLATORS DR SUSAN CORCORAN CARDIOLOGIST ONCE UPON A TIME.. Single chamber pacemakers Programmed at 70/min VVI 70 UNIPOLAR SYSTEMS A Unipolar Pacing
PERIOPERATIVE MANAGEMENT: CARDIAC PACEMAKERS AND DEFIBRILLATORS DR SUSAN CORCORAN CARDIOLOGIST ONCE UPON A TIME.. Single chamber pacemakers Programmed at 70/min VVI 70 UNIPOLAR SYSTEMS A Unipolar Pacing
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE
 NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE Implantable cardioverter defibrillators for the treatment of arrhythmias and cardiac resynchronisation therapy for the treatment of heart failure (review
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE Implantable cardioverter defibrillators for the treatment of arrhythmias and cardiac resynchronisation therapy for the treatment of heart failure (review
EXAMPLE PROTOCOL FOR PATIENT SCANNING PROCESS
 EXAMPLE PROTOCOL FOR PATIENT SCANNING PROCESS MRI SureScan Pacing, ICD, and CRT (CRT-D and CRT-P) Transvenous Implantable Cardiac Systems Timothy S. E. Albert, M.D. President CVMR Solutions, Inc. Carmel
EXAMPLE PROTOCOL FOR PATIENT SCANNING PROCESS MRI SureScan Pacing, ICD, and CRT (CRT-D and CRT-P) Transvenous Implantable Cardiac Systems Timothy S. E. Albert, M.D. President CVMR Solutions, Inc. Carmel
Pacing Without Wires: Leadless Cardiac Pacing
 REVIEWS AND CONTEMPORARY UPDATES Ochsner Journal 16:238 242, 2016 Ó Academic Division of Ochsner Clinic Foundation Pacing Without Wires: Leadless Cardiac Pacing Michael L. Bernard, MD, PhD Department of
REVIEWS AND CONTEMPORARY UPDATES Ochsner Journal 16:238 242, 2016 Ó Academic Division of Ochsner Clinic Foundation Pacing Without Wires: Leadless Cardiac Pacing Michael L. Bernard, MD, PhD Department of
Riata Lead Summit Jan 20, SJM Perspective Dr. Mark Carlson
 Riata Lead Summit Jan 20, 2012 SJM Perspective Dr. Mark Carlson 1 1 Presentation Outline Background, Characterization of Externalized Conductors Modeling and Testing Diagnostics and Therapeutics Recommendations
Riata Lead Summit Jan 20, 2012 SJM Perspective Dr. Mark Carlson 1 1 Presentation Outline Background, Characterization of Externalized Conductors Modeling and Testing Diagnostics and Therapeutics Recommendations
EBR Systems, Inc. 686 W. Maude Ave., Suite 102 Sunnyvale, CA USA
 Over 200,000 patients worldwide are estimated to receive a CRT device each year. However, limitations prevent some patients from benefiting. CHALLENGING PROCEDURE 5% implanted patients fail to have coronary
Over 200,000 patients worldwide are estimated to receive a CRT device each year. However, limitations prevent some patients from benefiting. CHALLENGING PROCEDURE 5% implanted patients fail to have coronary
CARDIAC DEVICE MR-CONDITIONAL PRODUCT SUMMARY CHART
 CARDIAC DEVICE MR-CONDITIONAL PRODUCT SUMMARY CHART December 2015 This chart encompasses all Medtronic cardiac devices FDA-Approved as MR Conditional and included in the MRI SureScan portfolio. If a device
CARDIAC DEVICE MR-CONDITIONAL PRODUCT SUMMARY CHART December 2015 This chart encompasses all Medtronic cardiac devices FDA-Approved as MR Conditional and included in the MRI SureScan portfolio. If a device
CONSULTA CRT-P Model C4TR01
 CONSULTA CRT-P Model C4TR01 Physical characteristics Volume a 15 cm 3 Mass H x W x D Surface area of titanium device can Surface area of each titanium nitride LECG electrode Radiopaque ID Materials in
CONSULTA CRT-P Model C4TR01 Physical characteristics Volume a 15 cm 3 Mass H x W x D Surface area of titanium device can Surface area of each titanium nitride LECG electrode Radiopaque ID Materials in
Selecting the Best ICD for your Patient-SICD v. TV. Martin C. Burke DO, FACOI CorVita Science Foundation & Academic Medical Center, Amsterdam
 Selecting the Best ICD for your Patient-SICD v. TV Martin C. Burke DO, FACOI CorVita Science Foundation & Academic Medical Center, Amsterdam COI DISCLOSURES I have received lecture and proctoring honoraria
Selecting the Best ICD for your Patient-SICD v. TV Martin C. Burke DO, FACOI CorVita Science Foundation & Academic Medical Center, Amsterdam COI DISCLOSURES I have received lecture and proctoring honoraria
Procurement Support to Ministry of Health, Ukraine. Invitation to Bid for:
 Procurement Support to Ministry of Health, Ukraine Invitation to Bid for: PROGRAMME XIX: MEDICAL PRODUCTS FOR HEALTHCARE INSTITUTIONS FOR TREATMENT OF PATIENTS WITH CARDIOVASCULAR AND CEREBROVASCULAR DISEASES
Procurement Support to Ministry of Health, Ukraine Invitation to Bid for: PROGRAMME XIX: MEDICAL PRODUCTS FOR HEALTHCARE INSTITUTIONS FOR TREATMENT OF PATIENTS WITH CARDIOVASCULAR AND CEREBROVASCULAR DISEASES
Q & A: How to Safely Scan Patients with a SureScan MRI Pacemaker
 Q & A: How to Safely Scan Patients with a SureScan MRI Pacemaker Patient Scheduling and Screening Q: Is there any special (other than regular MRI consent) consent form that should be signed by the patient?
Q & A: How to Safely Scan Patients with a SureScan MRI Pacemaker Patient Scheduling and Screening Q: Is there any special (other than regular MRI consent) consent form that should be signed by the patient?
Cardiac implantable electronic devices (CIEDs) in children include pacemakers and implantable cardioverter defibrillators (ICDs).
 Management of Children with Cardiac Devices Guideline originally developed by Leann Miles, APRN; Lindsey Pumphrey, RN; Srikant Das, MD, and the ANGELS Team. Last reviewed by Lindsey Pumphrey, RN, Srikant
Management of Children with Cardiac Devices Guideline originally developed by Leann Miles, APRN; Lindsey Pumphrey, RN; Srikant Das, MD, and the ANGELS Team. Last reviewed by Lindsey Pumphrey, RN, Srikant
COGNIS. Specifications. COGNIS: Model N119 ZIP TELEMETRY CARDIAC RESYNCHRONIZATION THERAPY
 COGNIS Specifications COGNIS: Model N119 The COGNIS cardiac resynchronization therapy defibrillator (CRT-D) provides ventricular tachyarrhythmia therapy and cardiac resynchronization therapy (CRT) for
COGNIS Specifications COGNIS: Model N119 The COGNIS cardiac resynchronization therapy defibrillator (CRT-D) provides ventricular tachyarrhythmia therapy and cardiac resynchronization therapy (CRT) for
EnTrust D154VRC Single Chamber ICD 35J delivered 8 seconds BOL, 11.8 seconds ERI 10.7 years**** 35cc, 68g Programmable Active Can
 EnTrust D154ATG Dual Chamber ICD 35J delivered 8 seconds BOL, 11 seconds ERI 7.7 years** 35cc***, 68g Programmable Active Can EnTrust D154VRC Single Chamber ICD 35J delivered 8 seconds BOL, 11.8 seconds
EnTrust D154ATG Dual Chamber ICD 35J delivered 8 seconds BOL, 11 seconds ERI 7.7 years** 35cc***, 68g Programmable Active Can EnTrust D154VRC Single Chamber ICD 35J delivered 8 seconds BOL, 11.8 seconds
MRI SURESCAN SYSTEMS. Patient Scanning Process PATIENT PRESCREENING. SureScan Pacing, Defibrillation, and CRT-D Systems Verification
 MRI SURESCAN SYSTEMS Patient Scanning Process PATIENT PRESCREENING SureScan Pacing, Defibrillation, and CRT-D Systems Verification Verify that patient has a complete SureScan pacing, defibrillation, or
MRI SURESCAN SYSTEMS Patient Scanning Process PATIENT PRESCREENING SureScan Pacing, Defibrillation, and CRT-D Systems Verification Verify that patient has a complete SureScan pacing, defibrillation, or
Riata lead extraction- a single centre experience
 Riata lead extraction- a single centre experience Rebecca L Noad, Keith W Morrice, Vivek N Kodoth, Carol M Wilson, Michael JD Roberts. Royal Victoria Hospital, Belfast, United Kingdom Background of previous
Riata lead extraction- a single centre experience Rebecca L Noad, Keith W Morrice, Vivek N Kodoth, Carol M Wilson, Michael JD Roberts. Royal Victoria Hospital, Belfast, United Kingdom Background of previous
Practice Questions.
 IBHRE Prep Practice Questions Question 1 The relative refractory yperiod of the ventricular myocardium corresponds to which of the following phases of the action potential? A. (0) B. (1) C. (2) D. (3)
IBHRE Prep Practice Questions Question 1 The relative refractory yperiod of the ventricular myocardium corresponds to which of the following phases of the action potential? A. (0) B. (1) C. (2) D. (3)
Interactive Simulator for Evaluating the Detection Algorithms of Implantable Defibrillators
 22 March 2002 Interactive Simulator for Evaluating the Detection Algorithms of Implantable Defibrillators F. HINTRINGER, O. PACHINGER Division of Cardiology, Department for Internal Medicine, University
22 March 2002 Interactive Simulator for Evaluating the Detection Algorithms of Implantable Defibrillators F. HINTRINGER, O. PACHINGER Division of Cardiology, Department for Internal Medicine, University
SJM MRI ACTIVATOR HANDHELD DEVICE WORKFLOW Model: EX4000. SJM-EDTR Item approved for U.S. use only.
 SJM MRI ACTIVATOR HANDHELD DEVICE WORKFLOW Model: EX4000 1 APPROVED MRI SCAN CONDITIONS 1.5 TESLA FULL BODY 2 SETUP SJM MRI ACTIVATOR HANDHELD DEVICE The SJM MRI Activator handheld device must be enabled
SJM MRI ACTIVATOR HANDHELD DEVICE WORKFLOW Model: EX4000 1 APPROVED MRI SCAN CONDITIONS 1.5 TESLA FULL BODY 2 SETUP SJM MRI ACTIVATOR HANDHELD DEVICE The SJM MRI Activator handheld device must be enabled
How to treat Cardiac Resynchronization Therapy complications? C. Leclercq Departement of Cardiology Centre Cardio-Pneumologique Rennes, France
 How to treat Cardiac Resynchronization Therapy complications? C. Leclercq Departement of Cardiology Centre Cardio-Pneumologique Rennes, France Presenter Disclosure Information Christophe Leclercq, MD,
How to treat Cardiac Resynchronization Therapy complications? C. Leclercq Departement of Cardiology Centre Cardio-Pneumologique Rennes, France Presenter Disclosure Information Christophe Leclercq, MD,
UnitedHealthcare, UnitedHealthcare of the River Valley and Neighborhood Health Partnership Cardiology Notification and Prior Authorization Program:
 Notification and Prior Authorization Program: Electrophysiology Implant Classification Table The following chart contains the codes that require notification or prior authorization as part of UnitedHealthcare
Notification and Prior Authorization Program: Electrophysiology Implant Classification Table The following chart contains the codes that require notification or prior authorization as part of UnitedHealthcare
Tachyarrhythmia Suspicion and Detection
 Tech Corner Tachyarrhythmia Suspicion and Detection NOTE: PLEASE NOTE THAT THE FOLLOWING INFORMATION IS A GENERAL DESCRIPTION OF THE FUNCTION. DETAILS AND PARTICULAR CASES ARE NOT DESCRIBED IN THE ARTICLE.
Tech Corner Tachyarrhythmia Suspicion and Detection NOTE: PLEASE NOTE THAT THE FOLLOWING INFORMATION IS A GENERAL DESCRIPTION OF THE FUNCTION. DETAILS AND PARTICULAR CASES ARE NOT DESCRIBED IN THE ARTICLE.
Device Update Implantable Cardioverter Defibrillator (ICD) 박상원
 2012 년춘계학술대회 Device Update Implantable Cardioverter Defibrillator (ICD) 박상원 Arrhythmia Center, KUMC www.korea-heartrhythm.com Korea University Medical Center Seoul, Korea The Development of ICD by a team
2012 년춘계학술대회 Device Update Implantable Cardioverter Defibrillator (ICD) 박상원 Arrhythmia Center, KUMC www.korea-heartrhythm.com Korea University Medical Center Seoul, Korea The Development of ICD by a team
Leadless Cardiac Pacemaker
 Leadless Cardiac Pacemaker Policy Number: Original Effective Date: MM.02.042 XXXMay 1, 2019 Lines of Business: Current Effective Date: HMO; PPO; QUEST Integration; FED87 XXXMay 1, 2019 Section: Medicine
Leadless Cardiac Pacemaker Policy Number: Original Effective Date: MM.02.042 XXXMay 1, 2019 Lines of Business: Current Effective Date: HMO; PPO; QUEST Integration; FED87 XXXMay 1, 2019 Section: Medicine
Performance of a Miniaturized Transcatheter Pacing System: First-in-human experience
 Performance of a Miniaturized Transcatheter Pacing System: First-in-human experience Philippe Ritter, MD Gabor Z Duray, MD, PhD, FESC; Clemens Steinwender, MD, FESC; Kyoko Soejima, MD; Razali Omar, MD;
Performance of a Miniaturized Transcatheter Pacing System: First-in-human experience Philippe Ritter, MD Gabor Z Duray, MD, PhD, FESC; Clemens Steinwender, MD, FESC; Kyoko Soejima, MD; Razali Omar, MD;
You have a what, inside you?
 Costal Emergency Medicine Conference You have a what, inside you? Less than mainstream medical devices encountered in the ED. Eric Ossmann, MD, FACEP Associate Professor Duke University Medical Center
Costal Emergency Medicine Conference You have a what, inside you? Less than mainstream medical devices encountered in the ED. Eric Ossmann, MD, FACEP Associate Professor Duke University Medical Center
You have a what, inside you?
 Costal Emergency Medicine Conference You have a what, inside you? Less than mainstream medical devices encountered in the ED. Eric Ossmann, MD, FACEP Associate Professor Duke University Medical Center
Costal Emergency Medicine Conference You have a what, inside you? Less than mainstream medical devices encountered in the ED. Eric Ossmann, MD, FACEP Associate Professor Duke University Medical Center
The protection you need - without touching your heart
 The S ICD System The protection you need - without touching your heart listen to your heart Patient information You have options An implantable defibrillator, commonly known as an ICD, is a device designed
The S ICD System The protection you need - without touching your heart listen to your heart Patient information You have options An implantable defibrillator, commonly known as an ICD, is a device designed
SUBCUTANEOUS IMPLANTABLE CARDIOVERTER DEFIBRILLATORS
 SUBCUTANEOUS IMPLANTABLE CARDIOVERTER DEFIBRILLATORS Protocol: CAR044 Effective Date: December 1, 2017 Table of Contents Page COMMERCIAL & MEDICAID COVERAGE RATIONALE... 1 MEDICARE COVERAGE RATIONALE...
SUBCUTANEOUS IMPLANTABLE CARDIOVERTER DEFIBRILLATORS Protocol: CAR044 Effective Date: December 1, 2017 Table of Contents Page COMMERCIAL & MEDICAID COVERAGE RATIONALE... 1 MEDICARE COVERAGE RATIONALE...
PERFORMANCE MEASURE TECHNICAL SPECIFICATIONS. HRS-3: Implantable Cardioverter-Defibrillator (ICD) Complications Rate
 PERFORMANCE MEASURE TECHNICAL SPECIFICATIONS HRS-3: Implantable Cardioverter-Defibrillator (ICD) Complications Rate Measure Title Description Measure Type Data Source Level of Analysis Numerator HRS-3:
PERFORMANCE MEASURE TECHNICAL SPECIFICATIONS HRS-3: Implantable Cardioverter-Defibrillator (ICD) Complications Rate Measure Title Description Measure Type Data Source Level of Analysis Numerator HRS-3:
Implantable cardioverter defibrillator, Inappropriate shock, Lead failure
 Inappropriate Discharges of Intravenous Implantable Cardioverter Defibrillators Owing to Lead Failure Takashi WASHIZUKA, 1 MD, Masaomi CHINUSHI, 1 MD, Ryu KAZAMA, 1 MD, Takashi HIRONO, 1 MD, Hiroshi WATANABE,
Inappropriate Discharges of Intravenous Implantable Cardioverter Defibrillators Owing to Lead Failure Takashi WASHIZUKA, 1 MD, Masaomi CHINUSHI, 1 MD, Ryu KAZAMA, 1 MD, Takashi HIRONO, 1 MD, Hiroshi WATANABE,
Biomedical Instrumentation
 Biomedical Instrumentation Prof. Dr. Nizamettin AYDIN naydin@yildiz.edu.tr naydin@ieee.org http://www.yildiz.edu.tr/~naydin Therapeutic and Prosthetic Devices 1 Figure 13.1 Block diagram of an asynchronous
Biomedical Instrumentation Prof. Dr. Nizamettin AYDIN naydin@yildiz.edu.tr naydin@ieee.org http://www.yildiz.edu.tr/~naydin Therapeutic and Prosthetic Devices 1 Figure 13.1 Block diagram of an asynchronous
EHRA Accreditation Exam - Sample MCQs Cardiac Pacing and ICDs
 EHRA Accreditation Exam - Sample MCQs Cardiac Pacing and ICDs Dear EHRA Member, Dear Colleague, As you know, the EHRA Accreditation Process is becoming increasingly recognised as an important step for
EHRA Accreditation Exam - Sample MCQs Cardiac Pacing and ICDs Dear EHRA Member, Dear Colleague, As you know, the EHRA Accreditation Process is becoming increasingly recognised as an important step for
MEDICAL POLICY Cardioverter Defibrillators
 POLICY........ PG-0224 EFFECTIVE......06/01/09 LAST REVIEW... 01/27/17 MEDICAL POLICY Cardioverter Defibrillators GUIDELINES This policy does not certify benefits or authorization of benefits, which is
POLICY........ PG-0224 EFFECTIVE......06/01/09 LAST REVIEW... 01/27/17 MEDICAL POLICY Cardioverter Defibrillators GUIDELINES This policy does not certify benefits or authorization of benefits, which is
New scientific documents from EHRA Management of patients with defibrillator shocks
 New scientific documents from EHRA Management of patients with defibrillator shocks Frieder Braunschweig MD PhD FESC Karolinska University Hospital Stockholm, Sweden Evolution of ICD therapy Worldwide
New scientific documents from EHRA Management of patients with defibrillator shocks Frieder Braunschweig MD PhD FESC Karolinska University Hospital Stockholm, Sweden Evolution of ICD therapy Worldwide
CRF procedure PROCEDURE FLOW. PATIENT ASSESSMENT Symptoms and indication. Pacemaker & ICD registration. Procedures. Procedure ICD.
 Pacemaker & ICD registration Procedures CRF procedure PROCEDURE FLOW Procedure ICD ICD PM ICM lead only PATIENT ASSESSMENT Symptoms and indication Symptoms and events (multiple possibilities) Asymptomatic
Pacemaker & ICD registration Procedures CRF procedure PROCEDURE FLOW Procedure ICD ICD PM ICM lead only PATIENT ASSESSMENT Symptoms and indication Symptoms and events (multiple possibilities) Asymptomatic
ICD Diagnostics: Making the most of it
 ICD Basics and Beyond ICD Diagnostics: Making the most of it Dulce Obias-Manno RN, BSN, MHSA, FHRS, CCDS, CEPS Cardiac Arrhythmia Center, Device Clinic Washington Hospital Center, Washington DC Objectives
ICD Basics and Beyond ICD Diagnostics: Making the most of it Dulce Obias-Manno RN, BSN, MHSA, FHRS, CCDS, CEPS Cardiac Arrhythmia Center, Device Clinic Washington Hospital Center, Washington DC Objectives
Defibrillation threshold testing should no longer be performed: contra
 Defibrillation threshold testing should no longer be performed: contra Andreas Goette St. Vincenz-Hospital Paderborn Dept. of Cardiology and Intensive Care Medicine Germany No conflict of interest to disclose
Defibrillation threshold testing should no longer be performed: contra Andreas Goette St. Vincenz-Hospital Paderborn Dept. of Cardiology and Intensive Care Medicine Germany No conflict of interest to disclose
ICD leads survival and troubles in the last 10 years
 XVI Symposium on Progress in Clinical Pacing, Rome, Italy - Dec 2 to 7, 2014 ICD leads survival and troubles in the last 10 years Antonio Raviele, MD, FESC, FHRS President ALFA Alliance to Fight Atrial
XVI Symposium on Progress in Clinical Pacing, Rome, Italy - Dec 2 to 7, 2014 ICD leads survival and troubles in the last 10 years Antonio Raviele, MD, FESC, FHRS President ALFA Alliance to Fight Atrial
Cardiac Rhythm Management Coder 2017
 Cardiac Rhythm Management Coder 2017 An easy-to-use tool for coding and reimbursement compliance Prepared and Published By: MedLearn Publishing, A Division of Panacea Healthcare Solutions, Inc. 287 East
Cardiac Rhythm Management Coder 2017 An easy-to-use tool for coding and reimbursement compliance Prepared and Published By: MedLearn Publishing, A Division of Panacea Healthcare Solutions, Inc. 287 East
MRI imaging for patients with cardiac implantable electronic devices (CIEDs)
 MRI imaging for patients with cardiac implantable electronic devices (CIEDs) 13 th annual International Winter Arrhythmia School Collingwood, Ontario, Canada February 6, 2016 Andrew C.T. Ha, MD, MSc, FRCPC
MRI imaging for patients with cardiac implantable electronic devices (CIEDs) 13 th annual International Winter Arrhythmia School Collingwood, Ontario, Canada February 6, 2016 Andrew C.T. Ha, MD, MSc, FRCPC
09/05/2018. True/False: All pacemakers have defibrillation capabilities.
 Paul G. Tarasi M.D. Staff Anesthesiologist Allegheny Health Network Adjunct Clinical Assistant Professor of Anesthesiology Temple University School of Medicine Describe different types of implanted cardiac
Paul G. Tarasi M.D. Staff Anesthesiologist Allegheny Health Network Adjunct Clinical Assistant Professor of Anesthesiology Temple University School of Medicine Describe different types of implanted cardiac
Automatic Identification of Implantable Cardioverter-Defibrillator Lead Problems Using Intracardiac Electrograms
 Automatic Identification of Implantable Cardioverter-Defibrillator Lead Problems Using Intracardiac Electrograms BD Gunderson, AS Patel, CA Bounds Medtronic, Inc., Minneapolis, USA Abstract Implantable
Automatic Identification of Implantable Cardioverter-Defibrillator Lead Problems Using Intracardiac Electrograms BD Gunderson, AS Patel, CA Bounds Medtronic, Inc., Minneapolis, USA Abstract Implantable
Biphasic Clinical Summaries
 Biphasic Clinical Summaries Defibrillation of Ventricular Fibrillation and Ventricular Tachycardia Background Physio-Control conducted a multi-centered, prospective, randomized and blinded clinical trial
Biphasic Clinical Summaries Defibrillation of Ventricular Fibrillation and Ventricular Tachycardia Background Physio-Control conducted a multi-centered, prospective, randomized and blinded clinical trial
Clinical Data Summary: Avoid FFS Study
 Atrial Pacing Lead with 1.1 mm Tip-to-Ring Spacing Clinical Data Summary: Avoid FFS Study A Multi-center, Randomized, Prospective Clinical Study Designed to Evaluate the 1699T Lead Clinical Data Summary:
Atrial Pacing Lead with 1.1 mm Tip-to-Ring Spacing Clinical Data Summary: Avoid FFS Study A Multi-center, Randomized, Prospective Clinical Study Designed to Evaluate the 1699T Lead Clinical Data Summary:
HRC Carole Joyce. Bradford Royal Infirmary. Senior Chief Cardiac Physiologist. Pacing & Invasive Services.
 HRC 2010 Carole Joyce Carole.joyce@bradfordhospitals.nhs.uk Pacing & Invasive Services Carole Joyce Introduction Basic Protocol Troubleshooting Case Studies Questions & Answer Carole Joyce ICD Troubleshooting
HRC 2010 Carole Joyce Carole.joyce@bradfordhospitals.nhs.uk Pacing & Invasive Services Carole Joyce Introduction Basic Protocol Troubleshooting Case Studies Questions & Answer Carole Joyce ICD Troubleshooting
Cigna - Prior Authorization Procedure List Cardiology
 Cigna - Prior Authorization Procedure List Cardiology Category CPT Code CPT Code Description 33206 Insertion of new or replacement of permanent pacemaker with transvenous electrode(s); atrial 33207 Insertion
Cigna - Prior Authorization Procedure List Cardiology Category CPT Code CPT Code Description 33206 Insertion of new or replacement of permanent pacemaker with transvenous electrode(s); atrial 33207 Insertion
INTERNAL CARDIOVERSION. Lancashire & South Cumbria Cardiac Network
 INTERNAL CARDIOVERSION Lancashire & South Cumbria Cardiac Network DC Cardioversion AF & AFL safe 1,2 efficacious 3,4 (60-94%) 5 SR - Increases exercise tolerance 6 Maintainence SR unlikely in patients
INTERNAL CARDIOVERSION Lancashire & South Cumbria Cardiac Network DC Cardioversion AF & AFL safe 1,2 efficacious 3,4 (60-94%) 5 SR - Increases exercise tolerance 6 Maintainence SR unlikely in patients
Cardiac Devices Past, Present, and Future. Outline
 Cardiac Devices Past, Present, and Future Charles Henrikson, MD Director, Electrophysiology Service 11 September 2016 Outline Introduction Native conduction system Indications for PPMs Indications for
Cardiac Devices Past, Present, and Future Charles Henrikson, MD Director, Electrophysiology Service 11 September 2016 Outline Introduction Native conduction system Indications for PPMs Indications for
Cardiac Resynchronization Therapy. Michelle Khoo, MD
 Cardiac Resynchronization Therapy Michelle Khoo, MD 10.7.08 HuiKuri HV NEJM 2001 Sudden Death (SD) in Subset Populations HuiKuri HV NEJM 2001 Sudden Death (SD) in Subset Populations SD in Competitive Athletes
Cardiac Resynchronization Therapy Michelle Khoo, MD 10.7.08 HuiKuri HV NEJM 2001 Sudden Death (SD) in Subset Populations HuiKuri HV NEJM 2001 Sudden Death (SD) in Subset Populations SD in Competitive Athletes
Cardiac Rhythm Management Coder 2018
 Cardiac Rhythm Management Coder 2018 An easy-to-use tool for coding and reimbursement compliance Prepared and Published By: MedLearn Publishing, A Division of MedLearn Media, Inc. 445 Minnesota Street,
Cardiac Rhythm Management Coder 2018 An easy-to-use tool for coding and reimbursement compliance Prepared and Published By: MedLearn Publishing, A Division of MedLearn Media, Inc. 445 Minnesota Street,
