Downloaded From: on 07 Mar 2019 Terms of Use:
|
|
|
- Marjorie Copeland
- 5 years ago
- Views:
Transcription
1 The First and The Second Mitotic Phases of Spermatogonial Stage in Xenopus laevis: Secondary Spermatogonia Which Have Differentiated after Completion of The First Mitotic Phase Acquire an Ability of Mitosis to Meiosis Conversion Authors: Kazufumi Takamune, Toshihiro Kawasaki, and Shuji Ukon Source: Zoological Science, 18(4) : Published By: Zoological Society of Japan URL: BioOne Complete (complete.bioone.org) is a full-text database of 200 subscribed and open-access titles in the biological, ecological, and environmental sciences published by nonprofit societies, associations, museums, institutions, and presses. Your use of this PDF, the BioOne Complete website, and all posted and associated content indicates your acceptance of BioOne s Terms of Use, available at Usage of BioOne Complete content is strictly limited to personal, educational, and non-commercial use. Commercial inquiries or rights and permissions requests should be directed to the individual publisher as copyright holder. BioOne sees sustainable scholarly publishing as an inherently collaborative enterprise connecting authors, nonprofit publishers, academic institutions, research libraries, and research funders in the common goal of maximizing access to critical research.
2 ZOOLOGICAL SCIENCE 18: (2001) 2001 Zoological Society of Japan The First and The Second Mitotic Phases of Spermatogonial Stage in Xenopus laevis: Secondary Spermatogonia Which Have Differentiated after Completion of The First Mitotic Phase Acquire an Ability of Mitosis to Meiosis Conversion Kazufumi Takamune*, Toshihiro Kawasaki and Shuji Ukon Department of Biological Science, Faculty of Science, Kumamoto University, Kurokami , Kumamoto , Japan ABSTRACT In Xenopus laevis, the spermatogenic cells derived from a primary spermatogonium divide synchronously and form a cyst surrounded by Sertoli cells. Therefore, it is easy to know how many times the spermatogenic cells divide from the primary spermatogonium by counting the cells in a cyst. In the present report, the distinctive feature of mitotic divisions during the spermatogonial stage in Xenopus laevis is described as having two phases. The first phase took place during five mitotic divisions. During this phase, the nucleus of primary spermatogonium, which was identified by its large size and lobulated shape with weak stainability, progressively decreased in size and became round with heterochromatin patches. In the second phase, the spermatogonia, that had completed the fifth mitotic division and morphologically differentiated into the secondary spermatogonia, could undergo up to three additional mitotic divisions without any morphological change. From the observation of the cysts that consisted of approximately 2 5, 2 6, 2 7, or 2 8 primary spermatocytes, it was further suggested that the secondary spermatogonia at the second phase could also enter a meiotic phase. All primary spermatocytes were believed to have the ability to undergo the meiotic phase-specific gene expression and two meiotic divisions because the round spermatids in the cyst consisting of approximately 2 7, 2 8, 2 9, or 2 10 cells expressed SP4 mrna which was transcribed in the spermatogenic cells at mid-pachytene stage and thereafter. These observations suggested that after the fifth mitotic division, the spermatogenic cells differentiated into the secondary spermatogonia and acquired an ability for mitosis to meiosis conversion. INTRODUCTION Conversion from mitosis to meiosis is a characteristic feature in gametogenesis because a diploid chromosome set is reduced by meiosis to a haploid set which culminates in the successful development of fertilized eggs without chromosome multiplication from generation to generation. During the spermatogenesis, spermatogonia that have undergone several mitotic divisions enter the meiotic prophase and differentiate into the primary spermatocytes before two meiotic divisions take place. The cell proliferation and the initiation of meiosis is known to be regulated by several kinds of hormones (Steinberger, 1971; Russel et al., 1987; Skinner, 1991). Successful induction of cell proliferation and conversion from mitosis to meiosis was carried out with the cultivation of testes * Corresponding author: Tel ; FAX takamune@gpo.kumamoto-u.ac.jp fragments in a chemically defined medium containing follicle stimulating hormone (FSH: Ji et al., 1992; Abe and Ji, 1994) or insulin-like growth factors (IGFs: Nakayama et al., 1994) in newts, human chorionic gonadotropin (HCG: Miura et al., 1991b) or 11-ketotestosterone (Miura et al., 1991c) in eels, and a mixture of several kinds of hormones such as FSH and insulin in Xenopus laevis (Risley et al., 1987). In contrast with the thorough search for the substance(s) inducing the conversion from mitosis to meiosis, an analyses on the timing when the spermatogonia acquire an ability for conversion from mitosis to meiosis has never been carried out. Recently, Yazawa et al. (2000) observed the features of mitotic divisions during the spermatogonial stage and showed that there were two kinds of mitotic phases. The nuclear diameter of spermatogonia progressively decreased after the division during early phase and then remained constant throughout late phase. Since the spermatogonia at the late phase (secondary spermatogonia) did not change their morphology before and after the mitotic divisions until they en-
3 578 K. Takamune et al. tered the meiotic phase, it may be natural for us to expect that these cells have acquired an ability for conversion from mitosis to meiosis. The present observations were attempted to understand the correct features of the mitotic divisions prior to the conversion from mitotic to meiotic phase in Xenopus laevis and showed that the spermatogonial stage in Xenopus spermatogenesis could be divided into two phases similar to that in newt spermatogenesis based on the transition (in the first phase) and the constancy (in the second phase) of spermatogonial morphology before and after the mitotic division. We also found primary spermatocytes which were believed to have differentiated from the spermatogonia immediately after the completion of mitotic divisions at the first phase based on the results of the cell number in a cyst and suggested that all secondary spermatogonia, which were morphologically distinguished from the spermatogonia at the first phase, possessed the ability of the mitosis to meiosis conversion. MATERIALS AND METHODS Animal Sexually mature male South African clawed frogs, Xenopus laevis (DAUDIN), were purchased from a dealer in Aomori (JAPAN). Light Microscopic Observations The testes were fixed with Bouin s fixative for more than 12 hr, dehydrated through a graded ethanol series, embedded in paraffin, and sectioned serially at 7 µm in thickness. The sections were spread onto slides (Matsunami Glass Ind. LTD., Japan) with warm H 2 O at 50 C and fixed completely on the slides by drying overnight at 37 C. After removal of the paraffin from the sections with xylene, specimens were hydrated and stained with Delafield hematoxylin and eosin, according to the conventional methods. When the number of spermatogenic cells in a cyst was estimated to be few, i.e., less than 16 cells, we counted them directly taking care not to count the same cells in adjacent sections twice. For the cyst consisting of a large number of secondary spermatogonia, primary spermatocytes, or round spermatids, we adopted Abercrombie s method (Abercrombie, 1946) to estimate the number of cells. We counted the number of the nuclei (n) in a cyst in all sections that contained the cyst, measured the diameter of five to ten of the largest nuclei (d: average), and calculated the estimated number of cells (N) by the following equation to avoid repeated counting of the same nuclei in adjacent sections: N = n x 7/(7 + d) wherein 7 is the thickness of the section (µm). in situ Hybridization Analyses SP4 cdna in puc 18 vector (pxsp531; Hiyoshi et al., 1991) was digested with Eco RI and inserted into the Eco RI site of pbluescript SK II+ (Stratagene). This construct was used for synthesizing sense and antisense RNA probes labeled with digoxygenin (DIG) 11-UTP (Boehringer Mannheim), using T7 RNA polymerase (Boehringer Mannheim) for sense probe and T3 RNA polymerase (Boehringer Mannheim) for antisense probe. Labeled RNA probes were dissolved in the hybridization buffer (1x Denhardt s solution, 600 mm NaCl, 50% formamide, 200 µg/ml E. coli trna, 10% dextran sulfate, 0.25% SDS, 1 mm EDTA, 10 mm Tris-HCl; ph 7.5) to a final concentration of 1.0 µg/ml. Testis was fixed with the mixture of 1/4 vol. of acetic acid and 3/4 vol. of 4% paraformaldehyde in phosphatebuffered saline (PBS: 137 mm NaCl, 2.7 mm KCl, 8.1 mm Na 2 HPO 4, 1.47 mm KH 2 PO 4 ; ph 7.4) on ice for 4 hr, dehydrated through a graded ethanol series, embedded in paraffin, and sectioned serially at 7 µm in thickness. The 1st to 14th or 16th to 29th sections were spread onto slides (Matsunami Glass Ind. LTD., Japan) and stained with Delafield hematoxylin and eosin as described above. The 15th and 30th sections were spread on warm water at 50 C and picked up onto silanized slides (Dako Japan). After drying the section at 40 C for 4 hr and removing the paraffin from the section by treatment with xylene, the specimens were successively treated with 10 µg/ml Proteinase K (Boehringer Mannheim) at 37 C for 7 min, 4% paraformaldehyde in PBS at room temperature (RT) for 10 min, 0.1 N HCl at RT for 10 min, and 0.1 M triethanolamine (ph 8.0) / 0.25% acetic anhydride at RT for 10 min. After hybridization with sense or antisense probe at 50 C for 16 hr, each specimen was treated with 2 µg/ml RNase A (Sigma) in 500 mm NaCl/ 1 mm EDTA/ 10 mm Tris-HCl (ph 7.5) at 37 C for 30 min and washed successively in 2 SSC (0.3 M NaCl / 0.03 M sodium citrate) at 50 C for 20 min, and twice in 0.2 SSC at 50 C for 20 min. For immunological detection of the DIG, the specimens were incubated in DIG II buffer (1.5% blocking reagent (Boehringer Mannheim) in DIG I buffer (150 mm NaCl, 100 mm maleic acid; ph 7.5)) at RT for 1 hr, followed by treatment with 1:500 diluted alkaline phosphatase-conjugated anti-dig antibody (Boehringer Mannheim) in DIG II buffer overnight at 12 C. After washing the specimens in DIG I buffer for 30 min and then in DIG III buffer (100 mm NaCl, 50 mm MgCl 2, 100 mm Tris-HCl; ph 9.5), localization of the DIG in the specimens was detected by treatment with 1:50 diluted NBT/BCIP Stock Solution (Boehringer Mannheim) in DIG III buffer. RESULTS During spermatogenesis in Xenopus laevis, primary spermatogonium undergoes mitotic divisions and develops to secondary spermatogonium followed by entering the meiotic phase and differentiating into primary spermatocyte. By the criteria given by Kalt (1976), the primary spermatogonium was distinguished easily in section as a cell which contained lobulated nucleus with weak stainability (Fig. 1, A) from secondary spermatogonium which had a round nucleus with heterochromatin patches (Fig. 1, F and G) and typical primary spermatocyte (zygotene to diplotene stage) which was characterized by the presence of a large round nucleus (Fig. 1, H). These observations revealed that the differentiation of primary spermatogonium to primary spermatocyte is accompanied with drastical morphological alterations in size, shape, and stainability of nucleus. In contrast with the detailed observations on the morphological changes from secondary spermatogonium to primary spermatocyte (Takamune et al., 1995), it is unclear how the primary spermatogonium differentiates into the secondary spermatogonium with morphological alterations. The spermatogenic cells derived from one primary spermatogonium develop synchronously and form a cyst with Sertoli cells around them (Lofts, 1974). Therefore, we can easily know how many times the spermatogenic cells divide mitotically by counting the cell number in a cyst. When we observed cysts consisting of two spermatogenic cells, which were produced by one mitotic division of the primary spermatogonium, the nuclei of the cells became smaller than that of the primary spermatogonium but otherwise were similar to the primary spermatogonium (Fig. 1, B). When the number of spermatogenic cells in a cyst increased to four to sixteen as a result of two to four mitotic divisions, the nuclei of the sper-
4 Timing of Mitosis to Meiosis Conversion 579 Fig. 1. Photomicrographs of sections of Xenopus testis showing various stages from primary spermatogonium to primary spermatocytes. Arrows, arrowheads, or double arrowheads show the cysts consisting of a primary spermatogonium (A: arrowheads), 2 (2 1 ) spermatogonia (B:arrowhead), 4 (2 2 ) spermatogonia (C: arrowhead), 8 (2 3 ) spermatogonia (D: arrowhead), 16 (2 4 ) spermatogonia (E: arrowhead), 32 (2 5 ) secondary spermatogonia (F: arrowhead), 56 (approximately 2 6 ) secondary spermatogonia (F: double arrowhead), 112 (approximately 2 7 ) secondary spermatogonia (G: arrowhead), 232 (approximately 2 8 ) secondary spermatogonia (G: double arrowhead), 32 (2 5 ) primary spermatocytes (H: arrowhead), 108 (approximately 2 7 ) primary spermatocytes (H: double arrowhead), and 202 (approximately 2 8 ) primary spermatocytes (H: arrow). Scale bar represents 20 µm.
5 580 K. Takamune et al. matogenic cells became smaller and round in shape with heterochromatin patches (Fig. 1, C E). The spermatogenic cells in a cyst consisting of 30.4±2.9 (S.E.) cells, which had undergone five mitotic divisions, showed the morphology as typical secondary spermatogonia (Fig. 1, F, arrowhead). The cysts consisting of the cells which showed the morphology of the typical secondary spermatogonia were classified into the other three groups in terms of the number of cells in a cyst; 55.2 ± 5.5 (S.E.) (Fig. 1, F, double arrowhead), 103.7±12.3 (S.E.) (Fig. 1, G, arrowhead), and 213.9±25.9 (S.E.) (Fig. 1, G, double arrowhead) cells, which were approximately 2 6, 2 7, and 2 8, respectively. These results indicate that after the completion of morphological alterations from primary spermatogonium to secondary spermatogonium by five mitotic divisions, the secondary spermatogonia underwent an additional three mitotic divisions without morphological changes. Fig. 2 shows the relationship between the number of cells in a cyst and the size of the nucleus. It clearly indicates the existence of two types of mitotic divisions at the spermatogonial stage; the first to the fifth mitotic divisions were accompanied with changes in nuclear size while the sixth to the eighth mitotic divisions coincided with neither change in nuclear size nor in shape nor in stainability (Fig. 2, closed triangles). In a similar way to the cysts consisting of the secondary spermatogonia, the cysts consisting of primary spermatocytes were classified into four groups in terms of the number of cells in a cyst; 27.6±3.2 (S.E.), 54.2±6.2 (S.E.), 104.1±10.2 (S.E.), and 203.7±17.2 (S.E.) cells, which were approximately 2 5, 2 6, 2 7, and 2 8, respectively (Fig. 1, H; Fig. 2, open circles). These results indicate that the secondary spermatogonia which had undergone at least five mitotic divisions, could enter the meiotic phase and differentiate into the primary spermatocytes, even though they did not undergo the full (eight) mitotic divisions. In order to clarify whether the primary spermatocytes which have differentiated after a few (five to seven) mitotic divisions can proceed to develop normally, we investigated the capability of those cells in the meiotic phase-specific gene expression and the progression of two meiotic divisions. SP4 gene is known to be expressed in the primary spermatocytes at the mid-pachytene stage and its transcripts remain until completion of the spermiogenesis (Mita et al., 1991). If all primary spermatocytes can proceed to develop normally, irrespectively of the number of mitotic divisions before the differentiation to these cells, the cysts consisting of 2 7 to 2 10 round spermatids that contain SP4 mrna should be found. Therefore, we performed in situ hybridization using antisense RNA for SP4 as a probe and counted the number of the cells in a cyst consisting of the round spermatids which contained the SP4 mrna (Fig. 3). Consistent with a previous report (Mita et al. 1991), all round spermatids as well as groups of spermatocytes and other post meiotic cells, including elongating spermatids and the residual body of mature sperm exhibited a Fig. 2. Relationship between the number of cells in a cysts (abscissa) consisting of cells identical to spermatogonia (closed triangles) and primary spermatocytes (open circles) and the diameter of nucleus of those cells (ordinate). Results from four individuals are shown separately. Fig. 3. Photomicrographs of sections of Xenopus testis, stained with hematoxylin and eosin (A, B, and C) and hybridized in situ with digoxigenin-labeled SP4 antisense RNA (a, b, and c). Arrows or arrowheads show the cysts consisting of 122 (approximately 2 7 ; A: arrowhead), 253 (approximately 2 8 ; A: double arrowhead), 472 (approximately 2 9 ; B: arrowhead), and 1000 (approximately 2 10 ; C: arrowhead) round spermatids. Scale bar represents 20 µm.
6 significant labeling over the background level in the cytoplasmic area, but there was no signal above the background in the areas occupied by spermatogonia, primary spermatocytes at leptotene-zygotene stage, and somatic cells, including Sertoli cells and other sustentacular cells. After confirmation of the expression of SP4 gene in the round spermatids, we enumerated the number of cells in the cysts consisting of those round spermatids using the serial sections before and behind the section which was used for in situ hybridization. As Fig. 3 shows, there were cysts consisting of 122 cells (Fig. 3, A and a, arrowhead), 253 cells (Fig. 3, A and a, double arrowhead), 472 cells (Fig. 3, B and b, arrowhead), and 1000 cells (Fig. 3, C and c, arrowhead), which were approximately 2 7, 2 8, 2 9, and 2 10, respectively. These results clearly indicate that the primary spermatocytes which have undergone a few (especially five) mitotic divisions prior to entering the meiotic prophase have normal ability to cause the stage-specific gene expression and proceed to two meiotic divisions to differentiate into round spermatids. Timing of Mitosis to Meiosis Conversion 581 DISCUSSION Fig. 4. Schematic illustration of spermatogenesis in Xenopus laevis. PG; primary spermatogonium. SG; secondary spermatogonia. PC; primary spermatocytes. RT; round spermatids. MS; mature sperm. Asterisk shows the timing of acquisition of an ability for conversion from mitosis to meiosis. Figure 4 shows the schematic illustration of spermatogenesis in Xenopus laevis, which is based on the results of this study. The primary spermatogonium, which was one of two daughter cells produced by the division of a stem cell, divided mitotically five times with the decrease in nucleus size and differentiated into secondary spermatogonia. Some of the secondary spermatogonia entered the meiotic phase and differentiated into the primary spermatocytes and others repeated the mitotic division up to three more times prior to entering the meiotic phase. The primary spermatocytes underwent two meiotic divisions, followed by spermiogenesis to form the mature sperm. As it is clear from this figure, there were two types of mitotic phases during the spermatogonial stage; the first (the first to the fifth mitotic divisions) and the second (the sixth to the eighth mitotic divisions) phases. During the first phase, the morphology of the spermatogonial nuclei drastically changed after each division, while the mitotic division at the second phase was not accompanied with a morphological alteration of the secondary spermatogonia. It should be noticed that the secondary spermatogonia at the second phase could either enter the meiotic phase or undergo further mitotic divisions. These observations suggested that after the fifth mitotic division, the secondary spermatogonia acquired the ability to enter the meiotic phase and that conversion of these cells from mitosis to meiosis was regulated by an external signal(s) but not by cells themselves. With regard to this acquisition of ability for mitotic to meiotic phase conversion, Miura et al. (1997) have also suggested the possibility of the existence of a regulatory point prior to entering the meiotic phase, based on the observations of impaired spermatogenesis in Japanese eels. In general, the type A spermatogonium (corresponds to the primary spermatogonium in Xenopus) of eel undergoes ten mitotic divisions prior to entering the meiotic phase when the spermatogenesis is induced by HCG injection into male eel (Miura et al., 1991a). In some cases, however, the spermatogenesis stopped after four or five mitotic divisions and the late type B spermatogonia (correspond to the secondary spermatogonia in Xenopus) in these eels could not differentiate into primary spermatocytes (Miura et al., 1997). From the results of differential mrna display to search for mrna that was expressed prior to entering the meiotic phase, Miura et al. (1999) concluded that PCNA mrna was expressed only in the late type B spermatogonia which had undergone five to six mitotic divisions. These results strongly pointed out the existence of a regulatory point at which spermatogenic cells had to go through before entering the meiotic phase and proceeding to a later stage of spermatogenesis. As the fifth mitotic division in Xenopus spermatogenesis seems to correspond to this regulatory point seen in eels, the stagespecific expression of PCNA mrna during Xenopus spermatogenesis was anticipated. However, we detected the PCNA mrna in all spermatogonia except for the primary spermatogonium from in situ hybridization, in contrast with the results seen in eels (data not shown). PCNA is a DNA polymerase auxiliary protein that functions during DNA replication and repair (Waga and Stillman, 1994). Therefore, it is no wonder that spermatogonia undergoing DNA replication followed by mitotic division express the PCNA mrna. More recently, we
7 582 K. Takamune et al. identified a novel gene which was expressed only in germ cells and cleavage stage embryos in Xenopus laevis and showed by in situ hybridization that the amount of this transcript clearly increased in secondary spermatogonia (Ikema et al., unpublished data). The up-regulation of this gene seemed to be closely related with the acquisition of the ability for mitotic to meiotic phase conversion. It should be focused to clarify the regulatory mechanisms of this gene expression for understanding the mitotic to meiotic phase conversion mechanisms. After the fifth mitotic division, the secondary spermatogonia could either enter the meiotic phase or undergo further mitotic divisions. This observation clearly indicates that the timing of the mitotic to meiotic phase conversion is not regulated by the number of mitotic divisions and leads us to suspect the necessity of an external signal(s) for conversion of mitotic phase to meiotic phase. Our presumption may be supported by the following experimental results: Human recombinant activin A and B could not induce the mitotic to meiotic phase conversion in eels although it could successfully induce the spermatogonial proliferation in the same manner with 11-ketotestosterone (Miura et al., 1995), which could induce both proliferation of spermatogonia and mitotic to meiotic phase conversion through the activation of Sertoli cells (Miura et al., 1991c). Experimental induction both of spermatogonial proliferation and mitotic to meiotic phase conversion were also successful when newt testes fragments rich in secondary spermatogonia were cultured in a medium containing FSH (Ji et al., 1992; Abe and Ji, 1994) or IGFs (Nakayama et al., 1999). Since the FSH receptor exists only in the Sertoli cells, FSH is believed to act through the activation of Sertoli cells (Ji et al., 1995; Maekawa et al., 1995). On the other hand, since IGF receptors exist in germ cells as well as Sertoli cells, IGFs are thought to be a candidate for the signals inducing mitotic to meiotic phase conversion by directly interacting with germ cells. Spermatogenesis is also maintained in Xenopus testes fragments cultured in serum free media supplemented with several kinds of hormones and vitamins, such as FSH, insulin and retinol (Risley et al., 1987). Our preliminary observations with a culture system showed the possibility that the differentiation of secondary spermatogonia to primary spermatocytes in Xenopus could be induced by treatment of testes fragments solely with FSH (Kawasaki et al., unpublished data). As the effect of FSH in Xenopus spermatogenesis was the same as that in newt spermatogenesis, we assumed that the initiation mechanisms of meiosis in Xenopus were similar to that in newt. Recently, Yazawa et al. (2000) revealed that the spermatogonial stage in the newt could be divided into the following two phases similar to that in Xenopus; early (the first to the fourth generations) and late (the fifth to the eighth generations) phases. In contrast with Xenopus secondary spermatogonia which are able to convert mitotic phase to meiotic phase after the merely five mitotic divisions, the newt spermatogonia usually do not enter the meiotic phase until the completion of the eight mitotic divisions. In general, the spermatogonia enter the meiotic phase after the species-specific fixed number of mitotic divisions (Roosen-Runge, 1977; Miura et al., 1991a; de Rooji and Grootegoed, 1998; Ando et al., 2000) although there are a few species in which number of mitotic divisions prior to entering meiosis is not fixed (nine or ten in the medaka, Sibata and Hamaguchi, 1988; five or six in the zebrafish, Ewing, 1972; Ando et al., 2000). We also reported previously that the differentiation of the secondary spermatogonia into the primary spermatocytes occurred after eight mitotic divisions in Xenopus (Takamune et al., 1995). In this study, however, we observed the primary spermatocytes which had divided mitotically fewer times (five to seven) before entering the meiotic phase. In contrast with the previous study in which we fixed the testes obtained from males which were injected with HCG and kept for a week before use for reinitiating the spermatogenesis following the release of the mature sperm from the lumen of the seminiferous tubules, we used the testes from the sexually mature males which were not injected with HCG. Therefore, lumen in the seminiferous tubules was filled with mature sperm. With regard to this point, almost all cysts consisting of primary spermatocytes in the testis from juvenile at five weeks after metamorphosis, in which the space of lumen had not fully formed yet, contained approximately 2 5 spermatogenic cells (data not shown). The number of mitotic divisions of the secondary spermatogonia after the fifth division may be a result of spatial restriction in the lumen where mature sperm are stored until ejaculation. In summary, the present study suggested that the conversion from mitosis to meiosis was regulated at two points; acquisition of an ability for the conversion and initiation of the meiotic phase. In Xenopus, the secondary spermatogonia possessed the ability for both proceeding to further mitotic divisions and entering the meiotic phase. We are expecting to elucidate the mechanisms for the mitotic to meiotic phase conversion through investigating the molecular basis for the differentiation into the secondary spermatogonia. REFERENCES Abe S-I, Ji Z-S (1994) Initiation and stimulation of spermatogenesis in vitro by mammalian follicle-stimulating hormone in the Japanese newt, Cynops pyrrhogaster. Int J Dev Biol 38: Abercrombie M (1946) Estimation of nuclear population from microtome sections. Anat Rec 94: Ando N, Miura T, Nader MR, Miura C, Yamauchi K (2000) A method for estimating the number of mitotic divisions in fish testes. Fish Sci 66: de Rooji DG, Grootegoed JA (1998) Spermatogonial stem cells. Curr Opin Cell Biol 10: Ewing H (1972) Spermatogenesis in zebrafish, Brachydanio rerio (Hamilton-Buchanan). Anat Rec 172: 308 Hiyoshi H, Uno S, Yokota T, Katagiri C, Nishida H, Takai M, Agata K, Eguchi G, Abe S-I (1991) Isolation of cdna for a Xenopus spermspecific basic nuclear protein (SP4) and evidence for expression of SP4 mrna in primary spermatocytes. Exp Cell Res 194: Ji Z-S, Kubokawa K, Abe S-I (1995) Promotion of differentiation of newt primary spermatocytes into spermatids by mammalian FSH via Sertoli cells. J Exp Zool 272:
8 Timing of Mitosis to Meiosis Conversion 583 Ji Z-S, Kubokawa K, Ishii S, Abe S-I (1992) Differentiation of secondary spermatogonia to primary spermatocytes by mammalian follicle-stimulating hormone in organ culture of testes fragments from the newt, Cynops pyrrhogaster. Develop Growth Differ 34: Kalt MR (1976) Morphology and kinetics of spermatogenesis in Xenopus laevis. J Exp Zool 195: Lofts B (1974) Reproduction. In Physiology of the Amphibia Vol 2 Ed by B Lofts, Academic Press, New York, pp Maekawa K, Ji Z-S, Abe S-I (1995) Proliferation of newt spermatogonia by mammalian FSH via Sertoli cells in vitro. J Exp Zool 272: Mita K, Takamune K, Katagiri C (1991) Genes for sperm-specific basic nuclear proteins in Bufo and Xenopus are expressed at different stages in spermatogenesis. Develop Growth Differ 33: Miura C, Miura T, Kudo N, Yamashita M, Yamauchi K (1999) cdna cloning of a stage-specific gene expressed during HCG-injected spermatogenesis in the Japanese eel. Develop Growth Differ 41: Miura T, Kawamura S, Miura C, Yamauchi K (1997) Impaired spermatogenesis in the Japanese eel, Anguilla japonica: Evidence for the existence of factors that regulate entry of germ cells into meiosis. Develop Growth Differ 39: Miura T, Miura C, Yamauchi K, Nagahama Y (1995) Human recombinant activin induces proliferation of spermatogonia in vitro in the Japanese eel Anguilla japonica. Fish Sci 61: Miura T, Yamauchi K, Nagahama Y, Takahashi H (1991a) Induction of spermatogenesis in male Japanese eel, Anguilla japonica, by a single injection of human chorionic gonadotropin. Zool Sci 8: Miura T, Yamauchi K, Takahashi H, Nagahama Y (1991b) Human chorionic gonadotropin induces all stages of spermatogenesis in vitro in the male Japanese eel (Anguilla japonica). Dev Biol 146: Miura T, Yamauchi K, Takahashi H, Nagahama Y (1991c) Hormonal induction of all stages of spermatogenesis in vitro in the male Japanese eel (Anguilla japonica). Proc Natl Acad Sci USA 88: Nakayama Y, Yamamoto T, Abe S-I (1999) IGF-I, IGF-II and insulin promote differentiation of spermatogonia to primary spermatocytes in organ culture of newt testes. Int J Dev Biol 43: Risley MS, Miller A, Bumcrot DA (1987) In vitro maintenance of spermatogenesis in Xenopus laevis testis explants cultured in serum-free media. Biol Rep 36: Roosen-Runge EC (1977) The Process of Spermatogenesis in Animals. Cambridge University Press, Cambridge, London Russel LD, Alger LE, Nequim LG (1987) Hormonal control of pubertal spermatogenesis. Endocrinology 120: Shibata N, Hamaguchi S (1988) Evidence for the sexual bipotentiality of spermatogonia in the fish, Oryzias latipes. J Exp Zool 245: Skinner MK (1991) Cell-cell interactions in the testis. Endocr Rev 12: Steinberger E (1971) Hormonal control of mammalian spermatogenesis. Physiol Rev 51: 1 22 Takamune K, Teshima K, Maeda M, Abe S-I (1995) Characteristic features of preleptotene spermatocytes in Xenopus laevis: Increase in the nuclear volume and first appearance of flattened vesicles in these cells. J Exp Zool 273: Waga S, Stillman B (1994) Anatomy of a DNA replication fork revealed by reconstitution of SV40 DNA replication in vitro. Nature 369: Yazawa T, Yamamoto T, Nakayama Y, Hamada S, Abe S-I (2000) Conversion from mitosis to meiosis: Morphology and expression of proliferating cell nuclear antigen (PCNA) and Dmc 1 during newt spermatogenesis. Develop Growth Differ 42: (Received February 6, 2001 / Accepted March 12, 2001)
Spermatogenesis. What is it and what does it look like? How do hormones regulate spermatogenesis?
 Spermatogenesis What is it and what does it look like? How do hormones regulate spermatogenesis? FSH, androgens, growth factors Animal Physiology (Hill, Wise, Anderson): Ch. 15 435-438 1 Spermatogenesis:
Spermatogenesis What is it and what does it look like? How do hormones regulate spermatogenesis? FSH, androgens, growth factors Animal Physiology (Hill, Wise, Anderson): Ch. 15 435-438 1 Spermatogenesis:
To General Embryology Dr: Azza Zaki
 Introduction To General Embryology The Human Development is a continuous process that begins when an ovum from a female is fertilized by a sperm from a male. Cell division, growth and differentiation transform
Introduction To General Embryology The Human Development is a continuous process that begins when an ovum from a female is fertilized by a sperm from a male. Cell division, growth and differentiation transform
Adapted from Preg. & Part., Senger
 MALE ENDOCRINOLOGY AND SPERMATOGENESIS (Chapter 10) AVS 222 (Instructor: Dr. Amin Ahmadzadeh) I. MALE ENDOCRINOLOGY (Figure10-1 to 10-3) A. Glands and their respective hormones 1) Hypothalamic hormone:
MALE ENDOCRINOLOGY AND SPERMATOGENESIS (Chapter 10) AVS 222 (Instructor: Dr. Amin Ahmadzadeh) I. MALE ENDOCRINOLOGY (Figure10-1 to 10-3) A. Glands and their respective hormones 1) Hypothalamic hormone:
Identification of the spermatogenic stages in living seminiferous tubules of man
 Identification of the spermatogenic stages in living seminiferous tubules of man V. Nikkanen, K.-O. S\l=o"\derstr\l=o"\m and M. Parvinen Department of Obstetrics and Gynecology, Turku University Central
Identification of the spermatogenic stages in living seminiferous tubules of man V. Nikkanen, K.-O. S\l=o"\derstr\l=o"\m and M. Parvinen Department of Obstetrics and Gynecology, Turku University Central
THE EFFECTS OF REPEATED INJECTIONS OF CHORIONIC GONADOTROPIN ON THE TESTES OF THE LEOPARD FROG (RANA PIPIENS SCHREBER)
 THE EFFECTS OF REPEATED INJECTIONS OF CHORIONIC GONADOTROPIN ON THE TESTES OF THE LEOPARD FROG (RANA PIPIENS SCHREBER) ROBERT P. McCOURT Department of Zoology and Entomology, The Ohio State University,
THE EFFECTS OF REPEATED INJECTIONS OF CHORIONIC GONADOTROPIN ON THE TESTES OF THE LEOPARD FROG (RANA PIPIENS SCHREBER) ROBERT P. McCOURT Department of Zoology and Entomology, The Ohio State University,
5 15/3/2012. Malik Al-Momani
 5 15/3/2012 Malik Al-Momani بسم هللا الرحمن الرحيم Spermatogenesis Note : Please refer to slides so see photos. Quick Revision : - Testis is divided by septum into testicular lobules, inside the lobules
5 15/3/2012 Malik Al-Momani بسم هللا الرحمن الرحيم Spermatogenesis Note : Please refer to slides so see photos. Quick Revision : - Testis is divided by septum into testicular lobules, inside the lobules
Histology of Male Reproductive system (1)
 Histology of Male Reproductive system (1) Prof. Dr. Malak A. Al-yawer Learning Objectives At the end of this lecture, the medical student will be able to: State the organization of the testis Define seminiferous
Histology of Male Reproductive system (1) Prof. Dr. Malak A. Al-yawer Learning Objectives At the end of this lecture, the medical student will be able to: State the organization of the testis Define seminiferous
Cell Divisions. The autosomes represent the whole body. * Male Sex Chromosomes: XY * Female Sex Chromosomes: XX
 Cell Divisions Each Cell (including gonads) has 46 chromosomes (23 pairs of chromosomes: 22 pairs of autosomes, 1 pair of sex chromosomes) which are located in the nucleus). The autosomes represent the
Cell Divisions Each Cell (including gonads) has 46 chromosomes (23 pairs of chromosomes: 22 pairs of autosomes, 1 pair of sex chromosomes) which are located in the nucleus). The autosomes represent the
Gametogenesis. Omne vivum ex ovo All living things come from eggs.
 Omne vivum ex ovo All living things come from eggs. William Harvery, 1651 Gametogenesis This lecture is the preface, so to speak, to embryology; that is, it introduces the development of the specialized
Omne vivum ex ovo All living things come from eggs. William Harvery, 1651 Gametogenesis This lecture is the preface, so to speak, to embryology; that is, it introduces the development of the specialized
Sex differentiation of germ cells in the teleost, Oryzias latipes, during normal embryonic development
 J. Embryol. exp. Morph. Vol. 2, 2, pp. -, 2 Printed in Great Britain Sex differentiation of germ cells in the teleost, Oryzias latipes, during normal embryonic development ByNORIYUKI SATOH AND NOBUO EGAMI
J. Embryol. exp. Morph. Vol. 2, 2, pp. -, 2 Printed in Great Britain Sex differentiation of germ cells in the teleost, Oryzias latipes, during normal embryonic development ByNORIYUKI SATOH AND NOBUO EGAMI
IN normal male fowls, four developmental stages of spermatogenetic activity
 Development of the Testis Tubule in the Fowl By GAMAL A. R. KAMAR (From the Animal Production Department, Faculty of Agriculture, Cairo University, Giza, Egypt) With three plates (figs. 1-3) SUMMARY Three
Development of the Testis Tubule in the Fowl By GAMAL A. R. KAMAR (From the Animal Production Department, Faculty of Agriculture, Cairo University, Giza, Egypt) With three plates (figs. 1-3) SUMMARY Three
Timing of commitment to autogamy in Paramecium bursaria
 Jpn. J. Protozool. Vol. 43, No. 2. (2010) 95 Short Communication Timing of commitment to autogamy in Paramecium bursaria Akira YANAGI Department of Biotechnology, Senshu University of Ishinomaki, Ishinomaki
Jpn. J. Protozool. Vol. 43, No. 2. (2010) 95 Short Communication Timing of commitment to autogamy in Paramecium bursaria Akira YANAGI Department of Biotechnology, Senshu University of Ishinomaki, Ishinomaki
Animal Development. Lecture 3. Germ Cells and Sex
 Animal Development Lecture 3 Germ Cells and Sex 1 The ovary of sow. The ovary of mare. The ovary of cow. The ovary of ewe. 2 3 The ovary. A generalized vertebrate ovary. (Wilt and Hake, Ch 2, 2004) 4 The
Animal Development Lecture 3 Germ Cells and Sex 1 The ovary of sow. The ovary of mare. The ovary of cow. The ovary of ewe. 2 3 The ovary. A generalized vertebrate ovary. (Wilt and Hake, Ch 2, 2004) 4 The
Gametogenesis. To complete this worksheet, select: Module: Continuity Activity: Animations Title: Gametogenesis. Introduction
 Gametogenesis To complete this worksheet, select: Module: Continuity Activity: Animations Title: Gametogenesis Introduction 1. a. Define gametogenesis. b. What cells are gametes? c. What are the two cell
Gametogenesis To complete this worksheet, select: Module: Continuity Activity: Animations Title: Gametogenesis Introduction 1. a. Define gametogenesis. b. What cells are gametes? c. What are the two cell
Lesson 1. Quiz (short) Cell cycle Chromosomes Mitosis phases
 Lesson 1 Quiz (short) Cell cycle Chromosomes Mitosis phases 2 Cell division is needed for Growth (Mitosis) Repair (Mitosis) Reproduction (Meiosis) 3 Mitosis consists of 4 phases (division of the nuclear
Lesson 1 Quiz (short) Cell cycle Chromosomes Mitosis phases 2 Cell division is needed for Growth (Mitosis) Repair (Mitosis) Reproduction (Meiosis) 3 Mitosis consists of 4 phases (division of the nuclear
The Cell Life Cycle. S DNA replication, INTERPHASE. G 2 Protein. G 1 Normal THE CELL CYCLE. Indefinite period. synthesis. of histones.
 Mitosis & Meiosis The Cell Life Cycle INTERPHASE G 1 Normal cell functions plus cell growth, duplication of organelles, protein synthesis S DNA replication, synthesis of histones THE CELL CYCLE M G 2 Protein
Mitosis & Meiosis The Cell Life Cycle INTERPHASE G 1 Normal cell functions plus cell growth, duplication of organelles, protein synthesis S DNA replication, synthesis of histones THE CELL CYCLE M G 2 Protein
Male Reproduction Organs. 1. Testes 2. Epididymis 3. Vas deferens 4. Urethra 5. Penis 6. Prostate 7. Seminal vesicles 8. Bulbourethral glands
 Outline Terminology Human Reproduction Biol 105 Lecture Packet 21 Chapter 17 I. Male Reproduction A. Reproductive organs B. Sperm development II. Female Reproduction A. Reproductive organs B. Egg development
Outline Terminology Human Reproduction Biol 105 Lecture Packet 21 Chapter 17 I. Male Reproduction A. Reproductive organs B. Sperm development II. Female Reproduction A. Reproductive organs B. Egg development
Meiosis & Sexual Reproduction. AP Biology
 Meiosis & Sexual Reproduction 2007-2008 Cell division / Asexual reproduction Mitosis produce cells with same information identical daughter cells exact copies clones same amount of DNA same number of chromosomes
Meiosis & Sexual Reproduction 2007-2008 Cell division / Asexual reproduction Mitosis produce cells with same information identical daughter cells exact copies clones same amount of DNA same number of chromosomes
Morphogenesis of the residual body of the mouse testis
 93 Morphogenesis of the residual body of the mouse testis By CASIMIR F. FIRLIT and JOSEPH R. DAVIS (From the Department of Pharmacology and Therapeutics, Stritch School of Medicine, and Graduate School,
93 Morphogenesis of the residual body of the mouse testis By CASIMIR F. FIRLIT and JOSEPH R. DAVIS (From the Department of Pharmacology and Therapeutics, Stritch School of Medicine, and Graduate School,
The spermatogenesis CHARACTERISTICS OF THE SPERMATOZOON 26/04/2017. Reproductive Biotechnologies Andrology I. Prof. Alberto Contri
 Reproductive Biotechnologies Andrology I The spermatogenesis Prof. Alberto Contri CHARACTERISTICS OF THE SPERMATOZOON 1) Aploid cell with high condensed DNA 2) Forward motility - flagellum 3) Enzymes for
Reproductive Biotechnologies Andrology I The spermatogenesis Prof. Alberto Contri CHARACTERISTICS OF THE SPERMATOZOON 1) Aploid cell with high condensed DNA 2) Forward motility - flagellum 3) Enzymes for
SUPPLEMENTARY MATERIAL. Sample preparation for light microscopy
 SUPPLEMENTARY MATERIAL Sample preparation for light microscopy To characterize the granulocytes and melanomacrophage centers, cross sections were prepared for light microscopy, as described in Material
SUPPLEMENTARY MATERIAL Sample preparation for light microscopy To characterize the granulocytes and melanomacrophage centers, cross sections were prepared for light microscopy, as described in Material
Production of Fertile Sperm. Animal Science 434. Hormonal Regulation of the Testis. hormonal regulation of the testis
 roduction of Fertile Sperm hormonal regulation of the testis nimal Science 434 Lecture 12: Spermatogenesis mitotic division of spermatogonia meiotic divisions of spermatocytes morphologic transformation
roduction of Fertile Sperm hormonal regulation of the testis nimal Science 434 Lecture 12: Spermatogenesis mitotic division of spermatogonia meiotic divisions of spermatocytes morphologic transformation
SPERMATOGENESIS IN VITRO
 SPERMATOGENESIS IN VITRO INDUCTION OF PROLIFERATION, MEIOSIS AND DIFFERENTIATION Mário Sousa Lab Cell Biology Institute of Biomedical Sciences (ICBAS) University of Porto msousa@icbas.up.pt Spermatogonia
SPERMATOGENESIS IN VITRO INDUCTION OF PROLIFERATION, MEIOSIS AND DIFFERENTIATION Mário Sousa Lab Cell Biology Institute of Biomedical Sciences (ICBAS) University of Porto msousa@icbas.up.pt Spermatogonia
Spermatogenesis in Man
 Spermatogenesis in Man I. Nuclear Morphology During Spermatogenesis in Man BRUNETTO CHIARELLI, PH.D., ARTHUR FALEK, PH.D., KAREN J. BACK, B.S., and C. THOMAS COWART, M.D. THE SEQUENCE of transformations
Spermatogenesis in Man I. Nuclear Morphology During Spermatogenesis in Man BRUNETTO CHIARELLI, PH.D., ARTHUR FALEK, PH.D., KAREN J. BACK, B.S., and C. THOMAS COWART, M.D. THE SEQUENCE of transformations
BIOH122 Session 26 Gametogenesis. Introduction. 1. a. Define gametogenesis. b. What cells are gametes?
 BIOH122 Session 26 Gametogenesis Introduction 1. a. Define gametogenesis. b. What cells are gametes? c. What are the two cell division processes that occur during the cell cycle? d. Define the cell cycle.
BIOH122 Session 26 Gametogenesis Introduction 1. a. Define gametogenesis. b. What cells are gametes? c. What are the two cell division processes that occur during the cell cycle? d. Define the cell cycle.
Comparative Studies between in vivo and in vitro Spermatogenesis of Japanese Eel (Anguilla japonica)
 Comparative Studies between in vivo and in vitro Spermatogenesis of Japanese Eel (Anguilla japonica) Author(s): Takeshi Miura, Nobutaka Ando, Chiemi Miura, and Kohei Yamauchi Source: Zoological Science,
Comparative Studies between in vivo and in vitro Spermatogenesis of Japanese Eel (Anguilla japonica) Author(s): Takeshi Miura, Nobutaka Ando, Chiemi Miura, and Kohei Yamauchi Source: Zoological Science,
Whole Mount Drosophila Embryo In Situ Hybridization with RNA probes 2/5/2001 Leslie Vosshall
 Whole Mount Drosophila Embryo In Situ Hybridization with RNA probes 2/5/2001 Leslie Vosshall DAY ONE All incubations are done at room temperature unless otherwise noted. All solutions and all containers
Whole Mount Drosophila Embryo In Situ Hybridization with RNA probes 2/5/2001 Leslie Vosshall DAY ONE All incubations are done at room temperature unless otherwise noted. All solutions and all containers
Embryology 3. Spermatogenesis:
 Embryology 3 Spermatogenesis: The 2 testis in males are each divided into lobes and lobules by connective tissue septa forming 250 lobule and in each lobule there are 1 to 4 seminefrous tubule ( so almost
Embryology 3 Spermatogenesis: The 2 testis in males are each divided into lobes and lobules by connective tissue septa forming 250 lobule and in each lobule there are 1 to 4 seminefrous tubule ( so almost
The Reproductive System
 PowerPoint Lecture Slide Presentation by Patty Bostwick-Taylor, Florence-Darlington Technical College The Reproductive System 16PART A The Reproductive System Gonads primary sex organs Testes in males
PowerPoint Lecture Slide Presentation by Patty Bostwick-Taylor, Florence-Darlington Technical College The Reproductive System 16PART A The Reproductive System Gonads primary sex organs Testes in males
Male Reproductive System
 Male Reproductive System Constitution of male reproductive system Genital gland ----testis Genital ducts epididymis / ductus deferens / urinary duct Accessory sex glands Penis prostate gland Seminal vesicle
Male Reproductive System Constitution of male reproductive system Genital gland ----testis Genital ducts epididymis / ductus deferens / urinary duct Accessory sex glands Penis prostate gland Seminal vesicle
Male Reproductive System
 Male Reproductive System organs that function in: gamete and hormone production not all in abdominal cavity paired testicles = controlled by LH & FSH duct systems accessory glands Testis: Gross Histology
Male Reproductive System organs that function in: gamete and hormone production not all in abdominal cavity paired testicles = controlled by LH & FSH duct systems accessory glands Testis: Gross Histology
MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question.
 MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) Which of the following hormones controls the release of anterior pituitary gonadotropins? A) LH
MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) Which of the following hormones controls the release of anterior pituitary gonadotropins? A) LH
The Reproductive System
 16 PART A The Reproductive System PowerPoint Lecture Slide Presentation by Jerry L. Cook, Sam Houston University ESSENTIALS OF HUMAN ANATOMY & PHYSIOLOGY EIGHTH EDITION ELAINE N. MARIEB The Reproductive
16 PART A The Reproductive System PowerPoint Lecture Slide Presentation by Jerry L. Cook, Sam Houston University ESSENTIALS OF HUMAN ANATOMY & PHYSIOLOGY EIGHTH EDITION ELAINE N. MARIEB The Reproductive
HANDOUT # 1 GAMETOGENESIS
 Gametogenesis 1 HANDOUT # 1 GAMETOGENESIS Anatomy Department R.A. FADEL Gametogenesis 2 بسم هللا الرحمن الرحيم ت هأ م وى }46 } ا نل و و ا زل و ج و ن و و و و و ن ه و أ ل ه ث وى }45{ إ ول ن أ و ة م ن ان
Gametogenesis 1 HANDOUT # 1 GAMETOGENESIS Anatomy Department R.A. FADEL Gametogenesis 2 بسم هللا الرحمن الرحيم ت هأ م وى }46 } ا نل و و ا زل و ج و ن و و و و و ن ه و أ ل ه ث وى }45{ إ ول ن أ و ة م ن ان
Acute myocardial infarction (MI) on initial presentation was diagnosed if there was 20 minutes
 SUPPLEMENTAL MATERIAL Supplemental Methods Diagnosis for acute myocardial infarction Acute myocardial infarction (MI) on initial presentation was diagnosed if there was 20 minutes or more of chest pain
SUPPLEMENTAL MATERIAL Supplemental Methods Diagnosis for acute myocardial infarction Acute myocardial infarction (MI) on initial presentation was diagnosed if there was 20 minutes or more of chest pain
Dynamics of mono, di and tri-methylated histone H3 lysine 4 during male meiotic prophase I. Nuclei were co-stained for H3.1/H3.2. Progressing stages
 Dynamics of mono, di and tri-methylated histone H3 lysine 4 during male meiotic prophase I. Nuclei were co-stained for H3.1/H3.2. Progressing stages of spermatogenesis are shown from left to right. Arrows/dotted
Dynamics of mono, di and tri-methylated histone H3 lysine 4 during male meiotic prophase I. Nuclei were co-stained for H3.1/H3.2. Progressing stages of spermatogenesis are shown from left to right. Arrows/dotted
Primary sex organs (gonads): testes and ovaries. Accessory reproductive organs: ducts, glands, and external genitalia
 Male Reproductive System Primary sex organs (gonads): testes and ovaries Produce sex cells (gametes) Secrete steroid sex hormones Androgens (males) Estrogens and progesterone (females) Accessory reproductive
Male Reproductive System Primary sex organs (gonads): testes and ovaries Produce sex cells (gametes) Secrete steroid sex hormones Androgens (males) Estrogens and progesterone (females) Accessory reproductive
Chapter 22 The Reproductive System (I)
 Chapter 22 The Reproductive System (I) An Overview of Reproductive Physiology o The Male Reproductive System o The Female Reproductive System 22.1 Reproductive System Overview Reproductive system = all
Chapter 22 The Reproductive System (I) An Overview of Reproductive Physiology o The Male Reproductive System o The Female Reproductive System 22.1 Reproductive System Overview Reproductive system = all
Cell Cycle and Cell Division
 122 Cell Cycle and Cell Division 1. Meiosis I is reductional division. Meiosis II is equational division due to [1988] (a) pairing of homologous chromosomes (b) crossing over (c) separation of chromatids
122 Cell Cycle and Cell Division 1. Meiosis I is reductional division. Meiosis II is equational division due to [1988] (a) pairing of homologous chromosomes (b) crossing over (c) separation of chromatids
 a Control IgG Intestine c Testis b Thymus 1 3 2 S S 2 1 3 4 4 Figure S1 The wild-type mouse (C57BL/6J) organs (intestine, thymus and testis) were frozen in liquid nitrogen and sectioned at 5 µm on a cryostat.
a Control IgG Intestine c Testis b Thymus 1 3 2 S S 2 1 3 4 4 Figure S1 The wild-type mouse (C57BL/6J) organs (intestine, thymus and testis) were frozen in liquid nitrogen and sectioned at 5 µm on a cryostat.
The Reproductive System
 Essentials of Human Anatomy & Physiology Elaine N. Marieb Seventh Edition Chapter 16 The Reproductive System Slides 16.1 16.20 Lecture Slides in PowerPoint by Jerry L. Cook The Reproductive System Gonads
Essentials of Human Anatomy & Physiology Elaine N. Marieb Seventh Edition Chapter 16 The Reproductive System Slides 16.1 16.20 Lecture Slides in PowerPoint by Jerry L. Cook The Reproductive System Gonads
LABORATORY EXERCISES FOR MALE REPRODUCTIVE SYSTEM
 LABORATORY EXERCISES FOR MALE REPRODUCTIVE SYSTEM Slide #101 (1096). Testis, rat. sustentacular ( Sertoli ) cells Nuclei of Sustentacular cells Leydig cells Spermatogonia Spermatocytes Spermatids pale
LABORATORY EXERCISES FOR MALE REPRODUCTIVE SYSTEM Slide #101 (1096). Testis, rat. sustentacular ( Sertoli ) cells Nuclei of Sustentacular cells Leydig cells Spermatogonia Spermatocytes Spermatids pale
SUPPLEMENTAL INFORMATION FOR. PAX7 expression defines germline stem cells in the adult testis
 SUPPLEMENTAL INFORMATION FOR PAX7 expression defines germline stem cells in the adult testis Gina M. Aloisio, Yuji Nakada, Hatice D. Saatcioglu, Christopher G. Peña, Michael D. Baker, Edward D. Tarnawa,
SUPPLEMENTAL INFORMATION FOR PAX7 expression defines germline stem cells in the adult testis Gina M. Aloisio, Yuji Nakada, Hatice D. Saatcioglu, Christopher G. Peña, Michael D. Baker, Edward D. Tarnawa,
Sperm production. Sperm production. Meiosis. Mitosis. The cells of Leydig in testes secrete
 Sperm production Ductus deferens Epididymis The cells of Leydig in testes secrete Seminiferous testosterone (T) tubules T secreted at puberty produces 2 o sex characteristics, spermatogenesis, & maintain
Sperm production Ductus deferens Epididymis The cells of Leydig in testes secrete Seminiferous testosterone (T) tubules T secreted at puberty produces 2 o sex characteristics, spermatogenesis, & maintain
Sperm production. Sperm production. Controlling sperm production. Meiosis. Mitosis. The cells of Leydig in testes secrete
 Ductus deferens Sperm production Epididymis The cells of Leydig in testes secrete Seminiferous testosterone (T) tubules T secreted at puberty produces 2 o sex characteristics, spermatogenesis, & maintain
Ductus deferens Sperm production Epididymis The cells of Leydig in testes secrete Seminiferous testosterone (T) tubules T secreted at puberty produces 2 o sex characteristics, spermatogenesis, & maintain
Chapter 2. Mitosis and Meiosis
 Chapter 2. Mitosis and Meiosis Chromosome Theory of Heredity What structures within cells correspond to genes? The development of genetics took a major step forward by accepting the notion that the genes
Chapter 2. Mitosis and Meiosis Chromosome Theory of Heredity What structures within cells correspond to genes? The development of genetics took a major step forward by accepting the notion that the genes
Downloaded From: on 10 Mar 2019 Terms of Use:
 Switching from Asexual to Sexual Reproduction in the Planarian Dugesia ryukyuensis: Change of the Fissiparous Capacity along with the Sexualizing Process Authors: Kazuya Kobayashi, and Motonori Hoshi Source:
Switching from Asexual to Sexual Reproduction in the Planarian Dugesia ryukyuensis: Change of the Fissiparous Capacity along with the Sexualizing Process Authors: Kazuya Kobayashi, and Motonori Hoshi Source:
Biology 4361 Developmental Biology. October 11, Multiple choice (one point each)
 Biology 4361 Developmental Biology Exam 1 October 11, 2005 Name: ID#: Multiple choice (one point each) 1. Sertoli cells a. surround spermatocytes b. are the structural components of the seminiferous tubules
Biology 4361 Developmental Biology Exam 1 October 11, 2005 Name: ID#: Multiple choice (one point each) 1. Sertoli cells a. surround spermatocytes b. are the structural components of the seminiferous tubules
A COMPARATIVE STUDY OF GERM CELL KINETICS IN THE TESTES OF CHILDREN WITH UNILATERAL CRYPTORCHIDISM: A PRELIMINARY REPORT*
 FERTILITY AND STERILITY Copyright 1970 by the Williams & Wilkins Co. Vol. 21, No. 11, November 1970 Printed in U.S.A. A COMPARATIVE STUDY OF GERM CELL KINETICS IN THE TESTES OF CHILDREN WITH UNILATERAL
FERTILITY AND STERILITY Copyright 1970 by the Williams & Wilkins Co. Vol. 21, No. 11, November 1970 Printed in U.S.A. A COMPARATIVE STUDY OF GERM CELL KINETICS IN THE TESTES OF CHILDREN WITH UNILATERAL
The Use of Rabbits in Male Reproductive Toxicology
 Environmental Health Perspectives Vol. 77, pp. 5-9, 1988 The Use of Rabbits in Male Reproductive Toxicology by Daniel Morton* The rabbit is the smallest and least expensive laboratory animal in which serial
Environmental Health Perspectives Vol. 77, pp. 5-9, 1988 The Use of Rabbits in Male Reproductive Toxicology by Daniel Morton* The rabbit is the smallest and least expensive laboratory animal in which serial
Basic histology 5/4/2015
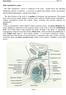 Male reproductive system The male reproductive system is composed of the testes, genital ducts (the adjoining epididymis, and the vas deferens, a accessory sex glands (the seminal vesicles, the prostrate
Male reproductive system The male reproductive system is composed of the testes, genital ducts (the adjoining epididymis, and the vas deferens, a accessory sex glands (the seminal vesicles, the prostrate
a. the tail disappears b. they become spermatids c. they undergo capacitation d. they have been stored in the uterus for several days
 (2 points each) Multiple Choice. Read each question thoroughly before answering. From the choices available, choose the answer that is the most correct. Place all answers on the accompanying answer sheet.
(2 points each) Multiple Choice. Read each question thoroughly before answering. From the choices available, choose the answer that is the most correct. Place all answers on the accompanying answer sheet.
Instructions for Use. APO-AB Annexin V-Biotin Apoptosis Detection Kit 100 tests
 3URGXFW,QIRUPDWLRQ Sigma TACS Annexin V Apoptosis Detection Kits Instructions for Use APO-AB Annexin V-Biotin Apoptosis Detection Kit 100 tests For Research Use Only. Not for use in diagnostic procedures.
3URGXFW,QIRUPDWLRQ Sigma TACS Annexin V Apoptosis Detection Kits Instructions for Use APO-AB Annexin V-Biotin Apoptosis Detection Kit 100 tests For Research Use Only. Not for use in diagnostic procedures.
Cycle of the Seminiferous Epithelium of the Guinea Pig
 Cycle of the Seminiferous Epithelium of the Guinea Pig A Method for Identification of the Stages Yves Clermont, Ph.D. IN THE GUINEA PIG, the cells of the seminiferous epithelium are arranged in definite
Cycle of the Seminiferous Epithelium of the Guinea Pig A Method for Identification of the Stages Yves Clermont, Ph.D. IN THE GUINEA PIG, the cells of the seminiferous epithelium are arranged in definite
Hormones of brain-testicular axis
 (Hormone Function) Hormones of brain-testicular axis anterior pituitary drives changes during puberty controlled by GnRH from hypothalamus begins to secrete FSH, LH LH targets interstitial endocrinocytes
(Hormone Function) Hormones of brain-testicular axis anterior pituitary drives changes during puberty controlled by GnRH from hypothalamus begins to secrete FSH, LH LH targets interstitial endocrinocytes
Nature Structural & Molecular Biology: doi: /nsmb Supplementary Figure 1. Generation and validation of mtef4-knockout mice.
 Supplementary Figure 1 Generation and validation of mtef4-knockout mice. (a) Alignment of EF4 (E. coli) with mouse, yeast and human EF4. (b) Domain structures of mouse mtef4 compared to those of EF4 (E.
Supplementary Figure 1 Generation and validation of mtef4-knockout mice. (a) Alignment of EF4 (E. coli) with mouse, yeast and human EF4. (b) Domain structures of mouse mtef4 compared to those of EF4 (E.
Cellular Reproduction, Part 2: Meiosis Lecture 10 Fall 2008
 Mitosis & 1 Cellular Reproduction, Part 2: Lecture 10 Fall 2008 Mitosis Form of cell division that leads to identical daughter cells with the full complement of DNA Occurs in somatic cells Cells of body
Mitosis & 1 Cellular Reproduction, Part 2: Lecture 10 Fall 2008 Mitosis Form of cell division that leads to identical daughter cells with the full complement of DNA Occurs in somatic cells Cells of body
Datum der Verteidigung :
 Datum der Verteidigung : 18. 07. 2003 Gutachter : Prof. Dr. Ludwig-Müller, Institut für Botanik, TU Dresden Prof. Dr. Gutzeit, Institut für Zoologie, TU Dresden Prof. Dr. Nagel, Institut für Hydrobiologie,
Datum der Verteidigung : 18. 07. 2003 Gutachter : Prof. Dr. Ludwig-Müller, Institut für Botanik, TU Dresden Prof. Dr. Gutzeit, Institut für Zoologie, TU Dresden Prof. Dr. Nagel, Institut für Hydrobiologie,
Testicular stem cells
 Testicular stem cells Dirk G. de Rooij Department of Endocrinology Faculty of Biology, Utrecht University 1. Knowledge on the development of the spermatogenic stem cell lineage 2. Principals of the nature
Testicular stem cells Dirk G. de Rooij Department of Endocrinology Faculty of Biology, Utrecht University 1. Knowledge on the development of the spermatogenic stem cell lineage 2. Principals of the nature
A Photographic Representation of Mitosis and Meiosis in the Male of Rattus norvegicus
 422 Cytologia 23 A Photographic Representation of Mitosis and Meiosis in the Male of Rattus norvegicus S. Ohno, W. D. Kaplan and R. Kinosita Department of Cytology and Genetics, Medical Research Institute,
422 Cytologia 23 A Photographic Representation of Mitosis and Meiosis in the Male of Rattus norvegicus S. Ohno, W. D. Kaplan and R. Kinosita Department of Cytology and Genetics, Medical Research Institute,
CHAPTER 8 CELL REPRODUCTION
 CHAPTER 8 CELL REPRODUCTION CHROMOSOME STRUCTURE Structures in the nucleus that carry genetic information Composed of DNA coiled around proteins called histones Consists of 2 identical parts called sister
CHAPTER 8 CELL REPRODUCTION CHROMOSOME STRUCTURE Structures in the nucleus that carry genetic information Composed of DNA coiled around proteins called histones Consists of 2 identical parts called sister
(A) PCR primers (arrows) designed to distinguish wild type (P1+P2), targeted (P1+P2) and excised (P1+P3)14-
 1 Supplemental Figure Legends Figure S1. Mammary tumors of ErbB2 KI mice with 14-3-3σ ablation have elevated ErbB2 transcript levels and cell proliferation (A) PCR primers (arrows) designed to distinguish
1 Supplemental Figure Legends Figure S1. Mammary tumors of ErbB2 KI mice with 14-3-3σ ablation have elevated ErbB2 transcript levels and cell proliferation (A) PCR primers (arrows) designed to distinguish
Chapter 28: REPRODUCTIVE SYSTEM: MALE
 Chapter 28: REPRODUCTIVE SYSTEM: MALE I. FUNCTIONAL ANATOMY (Fig. 28.1) A. Testes: glands which produce male gametes, as well as glands producing testosterone 2. Seminiferous tubules (Fig.28.3; 28.5) a.
Chapter 28: REPRODUCTIVE SYSTEM: MALE I. FUNCTIONAL ANATOMY (Fig. 28.1) A. Testes: glands which produce male gametes, as well as glands producing testosterone 2. Seminiferous tubules (Fig.28.3; 28.5) a.
SISTEMA REPRODUCTOR (LA IDEA FIJA) Copyright 2004 Pearson Education, Inc., publishing as Benjamin Cummings
 SISTEMA REPRODUCTOR (LA IDEA FIJA) How male and female reproductive systems differentiate The reproductive organs and how they work How gametes are produced and fertilized Pregnancy, stages of development,
SISTEMA REPRODUCTOR (LA IDEA FIJA) How male and female reproductive systems differentiate The reproductive organs and how they work How gametes are produced and fertilized Pregnancy, stages of development,
Mohammad Sha ban. Basheq Jehad. Hamzah Nakhleh
 11 Mohammad Sha ban Basheq Jehad Hamzah Nakhleh Physiology of the reproductive system In physiology, we are concerned with the mechanisms in which the system functions, and how the system responds to different
11 Mohammad Sha ban Basheq Jehad Hamzah Nakhleh Physiology of the reproductive system In physiology, we are concerned with the mechanisms in which the system functions, and how the system responds to different
Supplemental Figure 1. (A) The localization of Cre DNA recombinase in the testis of Cyp19a1-Cre mice was detected by immunohistchemical analyses
 Supplemental Figure 1. (A) The localization of Cre DNA recombinase in the testis of Cyp19a1-Cre mice was detected by immunohistchemical analyses using an anti-cre antibody; testes at 1 week (left panel),
Supplemental Figure 1. (A) The localization of Cre DNA recombinase in the testis of Cyp19a1-Cre mice was detected by immunohistchemical analyses using an anti-cre antibody; testes at 1 week (left panel),
Unit 4 - Reproduction
 Living Environment Practice Exam- Parts A and B-1 1. Which cell process occurs only in organisms that reproduce sexually? A) mutation B) replication C) meiosis D) mitosis 2. Which sequence represents the
Living Environment Practice Exam- Parts A and B-1 1. Which cell process occurs only in organisms that reproduce sexually? A) mutation B) replication C) meiosis D) mitosis 2. Which sequence represents the
Male reproduction. Cross section of Human Testis ผศ.ดร.พญ.ส ว ฒณ ค ปต ว ฒ ภาคว ชาสร รว ทยา คณะแพทยศาสตร ศ ร ราชพยาบาล 1. Aims
 Aims Male reproduction Male reproductive structure Spermatogenesis ส ว ฒณ ค ปต ว ฒ ห อง 216 โทร: 7578 Hypothalamo-pituitary-testicular axis Male sex hormone action Male reproductive structure Male reproductive
Aims Male reproduction Male reproductive structure Spermatogenesis ส ว ฒณ ค ปต ว ฒ ห อง 216 โทร: 7578 Hypothalamo-pituitary-testicular axis Male sex hormone action Male reproductive structure Male reproductive
Sex Determination and Gonadal Sex Differentiation in Fish
 Sex Determination and Gonadal Sex Differentiation in Fish Yoshitaka Nagahama Okazaki National Research Institutes, Japan This first slide shows the processes of gonadal sex differentiation and gametogenesis
Sex Determination and Gonadal Sex Differentiation in Fish Yoshitaka Nagahama Okazaki National Research Institutes, Japan This first slide shows the processes of gonadal sex differentiation and gametogenesis
Localization of Topoisomerase 2 and During the Rat Spematogenesis Cycle
 The Open Andrology Journal, 2011, 3, 25-29 25 Open Access Localization of Topoisomerase 2 and During the Rat Spematogenesis Cycle Lihua Liu 1, Ben Emery 1, John R. Gannon 1 and Douglas T. Carrell*,1,2,3
The Open Andrology Journal, 2011, 3, 25-29 25 Open Access Localization of Topoisomerase 2 and During the Rat Spematogenesis Cycle Lihua Liu 1, Ben Emery 1, John R. Gannon 1 and Douglas T. Carrell*,1,2,3
Supplementary Materials and Methods
 Supplementary Materials and Methods Whole Mount X-Gal Staining Whole tissues were collected, rinsed with PBS and fixed with 4% PFA. Tissues were then rinsed in rinse buffer (100 mm Sodium Phosphate ph
Supplementary Materials and Methods Whole Mount X-Gal Staining Whole tissues were collected, rinsed with PBS and fixed with 4% PFA. Tissues were then rinsed in rinse buffer (100 mm Sodium Phosphate ph
The Living Environment
 Name: Class: 1. Asexual reproduction primarily involves the process of 1) pollination 3) spermatogenesis 2) mitosis 4) ovulation The Living Environment Ms. Fazio Base your answers to questions 4 and 5
Name: Class: 1. Asexual reproduction primarily involves the process of 1) pollination 3) spermatogenesis 2) mitosis 4) ovulation The Living Environment Ms. Fazio Base your answers to questions 4 and 5
BIOL 2402 Reproductive Systems
 Collin College Dr. Chris Doumen BIOL 2402 Reproductive Systems 1 Reproductive System Most systems between males and females in the human body are similar in structure. The exception of course are the organs
Collin College Dr. Chris Doumen BIOL 2402 Reproductive Systems 1 Reproductive System Most systems between males and females in the human body are similar in structure. The exception of course are the organs
describe the parts and function of semen and the glands that contribute to it
 You need to be able to: describe spermatogenesis (How is sperm made?) describe the anatomy of a sperm describe the parts and function of semen and the glands that contribute to it How is sperm made? Spermatogenesis
You need to be able to: describe spermatogenesis (How is sperm made?) describe the anatomy of a sperm describe the parts and function of semen and the glands that contribute to it How is sperm made? Spermatogenesis
Cell Division. The Process of Cell Division Section Section 10.2: The Process of Cell Division 12/8/2010
 The Process of Cell Division Section 10.2 Biology B Section 10.2: The Process of Cell Division The student will investigate and understand common mechanisms of inheritance and protein synthesis. Key concepts
The Process of Cell Division Section 10.2 Biology B Section 10.2: The Process of Cell Division The student will investigate and understand common mechanisms of inheritance and protein synthesis. Key concepts
Testes (male gonads) -Produce sperm -Produce sex hormones -Found in a sac called the scrotum -Suspended outside of the body cavity for temperature
 REPRODUCTION Testes (male gonads) -Produce sperm -Produce sex hormones -Found in a sac called the scrotum -Suspended outside of the body cavity for temperature reduction -Testes wall made of fibrous connective
REPRODUCTION Testes (male gonads) -Produce sperm -Produce sex hormones -Found in a sac called the scrotum -Suspended outside of the body cavity for temperature reduction -Testes wall made of fibrous connective
Chromosomes and Cell Cycle
 Chromosomes and Cell Cycle Cell Basics There are trillions of cells in your body Cells are microscopic Cells have DNA inside a structure called the nucleus The nucleus is enclosed by a structure called
Chromosomes and Cell Cycle Cell Basics There are trillions of cells in your body Cells are microscopic Cells have DNA inside a structure called the nucleus The nucleus is enclosed by a structure called
Cell Division Questions. Mitosis and Meiosis
 Cell Division Questions Mitosis and Meiosis 1 10 Do not write outside the box 5 Figure 3 shows a pair of chromosomes at the start of meiosis. The letters represent alleles. Figure 3 E E e e F F f f 5 (a)
Cell Division Questions Mitosis and Meiosis 1 10 Do not write outside the box 5 Figure 3 shows a pair of chromosomes at the start of meiosis. The letters represent alleles. Figure 3 E E e e F F f f 5 (a)
Outline. Male Reproductive System Testes and Sperm Hormonal Regulation
 Outline Male Reproductive System Testes and Sperm Hormonal Regulation Female Reproductive System Genital Tract Hormonal Levels Uterine Cycle Fertilization and Pregnancy Control of Reproduction Infertility
Outline Male Reproductive System Testes and Sperm Hormonal Regulation Female Reproductive System Genital Tract Hormonal Levels Uterine Cycle Fertilization and Pregnancy Control of Reproduction Infertility
Change in frequency of radiation induced micronuclei during interphase of four-cell mouse embryos in vitro
 Radiat Environ Biophys (1986) 25:195-199 Radiation and Environmental Biophysics Springer-Verlag 1986 Change in frequency of radiation induced micronuclei during interphase of four-cell mouse embryos in
Radiat Environ Biophys (1986) 25:195-199 Radiation and Environmental Biophysics Springer-Verlag 1986 Change in frequency of radiation induced micronuclei during interphase of four-cell mouse embryos in
Biology of gender Sex chromosomes determine gonadal sex (testis-determining factor)
 Indifferent ducts of embryo Biology of gender Sex chromosomes determine gonadal sex (testis-determining factor) Y chromosome present Y chromosome absent Phenotypic sex is depends on development of external
Indifferent ducts of embryo Biology of gender Sex chromosomes determine gonadal sex (testis-determining factor) Y chromosome present Y chromosome absent Phenotypic sex is depends on development of external
Biology of gender Sex chromosomes determine gonadal sex (testis-determining factor)
 Indifferent ducts of embryo Y chromosome present Y chromosome absent Male Female penis ovary uterus vagina testis Biology of gender Sex chromosomes determine gonadal sex (testis-determining factor) Phenotypic
Indifferent ducts of embryo Y chromosome present Y chromosome absent Male Female penis ovary uterus vagina testis Biology of gender Sex chromosomes determine gonadal sex (testis-determining factor) Phenotypic
The Cell Cycle CHAPTER 12
 The Cell Cycle CHAPTER 12 The Key Roles of Cell Division cell division = reproduction of cells All cells come from pre-exisiting cells Omnis cellula e cellula Unicellular organisms division of 1 cell reproduces
The Cell Cycle CHAPTER 12 The Key Roles of Cell Division cell division = reproduction of cells All cells come from pre-exisiting cells Omnis cellula e cellula Unicellular organisms division of 1 cell reproduces
Pathology of Male Reproductive System 1
 Pathology of Male Reproductive System 1 Professor dr Ali Hassan Altimimi Professor of Pathology& Histology MSc, PHD, MD(UK) MALE REPRODUCTIVE SYSTEM The internal male genitalia consist of the testes with
Pathology of Male Reproductive System 1 Professor dr Ali Hassan Altimimi Professor of Pathology& Histology MSc, PHD, MD(UK) MALE REPRODUCTIVE SYSTEM The internal male genitalia consist of the testes with
Effects of Ablation of the Submaxillary Gland in Guinea Pigs IV. Cause of deterioration of the tubules in the testes
 1961 475 Effects of Ablation of the Submaxillary Gland in Guinea Pigs IV. Cause of deterioration of the tubules in the testes Kazuo Suzuki Received August 1, 1960 Shakujii Institute, Tokyo Medical College,
1961 475 Effects of Ablation of the Submaxillary Gland in Guinea Pigs IV. Cause of deterioration of the tubules in the testes Kazuo Suzuki Received August 1, 1960 Shakujii Institute, Tokyo Medical College,
The Cell Cycle. Chapter 12. Biology. Edited by Shawn Lester. Eighth Edition Neil Campbell and Jane Reece. PowerPoint Lecture Presentations for
 Chapter 12 The Cell Cycle Edited by Shawn Lester PowerPoint Lecture Presentations for Biology Eighth Edition Neil Campbell and Jane Reece Lectures by Chris Romero, updated by Erin Barley with contributions
Chapter 12 The Cell Cycle Edited by Shawn Lester PowerPoint Lecture Presentations for Biology Eighth Edition Neil Campbell and Jane Reece Lectures by Chris Romero, updated by Erin Barley with contributions
Analysis of Spermatogenesis Using an Eel Model
 Aqua-BioScience Monographs, Vol. 4, No. 4, pp. 105 129 (2011) www.terrapub.co.jp/onlinemonographs/absm/ Analysis of Spermatogenesis Using an Eel Model Chiemi Miura and Takeshi Miura* Research Group for
Aqua-BioScience Monographs, Vol. 4, No. 4, pp. 105 129 (2011) www.terrapub.co.jp/onlinemonographs/absm/ Analysis of Spermatogenesis Using an Eel Model Chiemi Miura and Takeshi Miura* Research Group for
Induction of spermatogenic synchrony by retinoic acid in neonatal mice
 EDITOR'S Letter to CORNER the Editor Spermatogenesis 3:1, e23180; January/February/March 2013 2013; 2013 Landes Bioscience EDITOR'S CORNER Induction of spermatogenic synchrony by retinoic acid in neonatal
EDITOR'S Letter to CORNER the Editor Spermatogenesis 3:1, e23180; January/February/March 2013 2013; 2013 Landes Bioscience EDITOR'S CORNER Induction of spermatogenic synchrony by retinoic acid in neonatal
The first success of glass eel production in the world: basic biology on fish reproduction advances new applied technology in aquaculture
 Fish Physiol Biochem (2005) 31:193 199 DOI 10.1007/s10695-006-0024-3 RESEARCH ARTICLE The first success of glass eel production in the world: basic biology on fish reproduction advances new applied technology
Fish Physiol Biochem (2005) 31:193 199 DOI 10.1007/s10695-006-0024-3 RESEARCH ARTICLE The first success of glass eel production in the world: basic biology on fish reproduction advances new applied technology
Sami Ventelä, 1,2 Hiroshi Ohta, 3 Martti Parvinen, 2 and Yoshitake Nishimune 3
 BIOLOGY OF REPRODUCTION 66, 1422 1429 (2002) Development of the Stages of the Cycle in Mouse Seminiferous Epithelium after Transplantation of Green Fluorescent Protein-Labeled Spermatogonial Stem Cells
BIOLOGY OF REPRODUCTION 66, 1422 1429 (2002) Development of the Stages of the Cycle in Mouse Seminiferous Epithelium after Transplantation of Green Fluorescent Protein-Labeled Spermatogonial Stem Cells
Dako IT S ABOUT TIME. Interpretation Guide. Agilent Pathology Solutions. ALK, ROS1 and RET IQFISH probes (Dako Omnis) MET IQFISH probe (Dako Omnis)
 INTERPRETATION Dako Agilent Pathology Solutions IQFISH Interpretation Guide ALK, ROS1 and RET IQFISH probes (Dako Omnis) MET IQFISH probe (Dako Omnis) IT S ABOUT TIME For In Vitro Diagnostic Use ALK, ROS1,
INTERPRETATION Dako Agilent Pathology Solutions IQFISH Interpretation Guide ALK, ROS1 and RET IQFISH probes (Dako Omnis) MET IQFISH probe (Dako Omnis) IT S ABOUT TIME For In Vitro Diagnostic Use ALK, ROS1,
General Biology. Overview: The Key Roles of Cell Division. Unicellular organisms
 General Biology Course No: BNG2003 Credits: 3.00 8. The Cell Cycle Prof. Dr. Klaus Heese Overview: The Key Roles of Cell Division The continuity of life is based upon the reproduction of cells, or cell
General Biology Course No: BNG2003 Credits: 3.00 8. The Cell Cycle Prof. Dr. Klaus Heese Overview: The Key Roles of Cell Division The continuity of life is based upon the reproduction of cells, or cell
Chapter 4 The Chromosome Theory of Inheritance
 Chapter 4 The Chromosome Theory of Inheritance 4-1 Sections to study 4.1 Chromosomes: The carriers of genes 4.2 Mitosis: Cell division that preserves chromosome number 4.3 Meiosis: Cell division that halve
Chapter 4 The Chromosome Theory of Inheritance 4-1 Sections to study 4.1 Chromosomes: The carriers of genes 4.2 Mitosis: Cell division that preserves chromosome number 4.3 Meiosis: Cell division that halve
Spermatogonial proliferation and apoptosis in hypospermatogenesis associated with nonobstructive azoospermia
 FERTILITY AND STERILITY VOL. 76, NO. 5, NOVEMBER 2001 Copyright 2001 American Society for Reproductive Medicine Published by Elsevier Science Inc. Printed on acid-free paper in U.S.A. Spermatogonial proliferation
FERTILITY AND STERILITY VOL. 76, NO. 5, NOVEMBER 2001 Copyright 2001 American Society for Reproductive Medicine Published by Elsevier Science Inc. Printed on acid-free paper in U.S.A. Spermatogonial proliferation
Chapter 14 Cellular Reproduction
 Chapter 14 Cellular Reproduction Biology 3201 Introduction One of the important life functions of living things is their ability to reproduce. Reproduction depends on the cell. Cells reproduce in order
Chapter 14 Cellular Reproduction Biology 3201 Introduction One of the important life functions of living things is their ability to reproduce. Reproduction depends on the cell. Cells reproduce in order
Reproductive Toxicology
 Reproductive Toxicology 32 (2011) 395 406 Contents lists available at SciVerse ScienceDirect Reproductive Toxicology jo u r n al hom epa ge: ww w.elsevier.com/locate/reprotox Effects of multiple doses
Reproductive Toxicology 32 (2011) 395 406 Contents lists available at SciVerse ScienceDirect Reproductive Toxicology jo u r n al hom epa ge: ww w.elsevier.com/locate/reprotox Effects of multiple doses
Chapter 36 Active Reading Guide Reproduction and Development
 Name: AP Biology Mr. Croft Chapter 36 Active Reading Guide Reproduction and Development Section 1 1. Distinguish between sexual reproduction and asexual reproduction. 2. Which form of reproduction: a.
Name: AP Biology Mr. Croft Chapter 36 Active Reading Guide Reproduction and Development Section 1 1. Distinguish between sexual reproduction and asexual reproduction. 2. Which form of reproduction: a.
GROWTH AND OBSERVATIONS OF CHINESE HAMSTER SEMINIFEROUS EPITHELIUM IN VITRO
 J. Cell Sci. 6, 19S-205 (1970) Printed in Great Britain GROWTH AND OBSERVATIONS OF CHINESE HAMSTER SEMINIFEROUS EPITHELIUM IN VITRO D. J. ELLINGSON AND K. T. S. YAO U.S. Department of Health, Education
J. Cell Sci. 6, 19S-205 (1970) Printed in Great Britain GROWTH AND OBSERVATIONS OF CHINESE HAMSTER SEMINIFEROUS EPITHELIUM IN VITRO D. J. ELLINGSON AND K. T. S. YAO U.S. Department of Health, Education
Germ Cell Degeneration During Postprophase of Meiosis and Serum Concentrations of Gonadotropins in Young Adult and Older Adult Men
 BIOLOGY OF REPRODUCTION 31, 779-784 (1984) Germ Cell Degeneration During Postprophase of Meiosis and Serum Concentrations of Gonadotropins in Young Adult and Older Adult Men LARRY JOHNSON,2 CHARLES S.
BIOLOGY OF REPRODUCTION 31, 779-784 (1984) Germ Cell Degeneration During Postprophase of Meiosis and Serum Concentrations of Gonadotropins in Young Adult and Older Adult Men LARRY JOHNSON,2 CHARLES S.

![[ 3<M ] STUDIES ON TAPEWORM PHYSIOLOGY [ 3<M ] STUDIES ON TAPEWORM PHYSIOLOGY](/thumbs/78/78631839.jpg) [ 3
[ 3