Biological Chemistry: Introduction to Carbohydrates and Carbohydrate Metabolism. Sugars
|
|
|
- Scot Harris
- 6 years ago
- Views:
Transcription
1 Biological Chemistry: Introduction to Carbohydrates and Carbohydrate Metabolism Sugars 1
2 Carbohydrates: Optical Activity A carbon with 4 different R groups is said to be CHIRAL These compounds, i.e., with chiral carbons, rotate the plane of polarized light. 2
3 Polarized Light Rotation 3
4 Chiral Compounds: Examples 4
5 5
6 6
7 It s like putting your right hand in a left hand glove D-ala does not fit the enzyme L-ala DOES All AA s this way in mammals 7
8 Carbohydrates Organic ally Saccharide = Greek for sugar ose = suffix for a sugar 8
9 Pentoses 9
10 Hexoses The pentoses (previous slide) and the hexoses (this slide) are ALDOSES ( aldehyde sugars ) They are also monosaccharides ( one sugars ) 10
11 Ketoses -- monosaccharides 11
12 Fisher to Haworth Transition While pentoses and hexoses exist as ring structures almost exclusively (there ARE exceptions) in biological systems, O Chemists start from stick structures Biochemists close the sticks to form rings 12
13 Glucose Haworth formation REMEMBER: organic molecules are NOT linear when the C is in sp 3 hybridization. 13
14 In General: Hemiacetal and Acetal 14
15 In General: Hemiketals and Ketals NOTE: It s VERY difficult to tell acetals and ketals apart, therefore, we usually only talk about ACETALS. 15
16 Fructose Haworth Formation 16
17 Carbohydrates are generally seen as sources of quick energy. They consist of carbon, hydrogen and oxygen. In the old days, they were named carbohydrates as the ratio of hydrogen to oxygen was thought to be 2:1. We now know differently, although the name has stuck throughout time. There are three categories of carbohydrates in which we have interest: monosaccharides, disaccharides and polysaccharides. 17
18 Two of the simplest carbohydrates are glucose and fructose, the sugar in our blood and fruit sugar. 18
19 As you can see, both have unique geometric shapes in their base structures and each has a unique orientation of the -OH groups upon the base structure. Remember that at each corner, there is a carbon atom and that carbon takes 4 bonds. The hydrogen atoms that are by themselves are not shown but are understood to be present. Biochemists developed a short-hand for quickly sketching carbohydrates called the Hayworth projections. The graphics on the right of each of the above sugars show the Hayworth projections, where the -OH's are replaced with lines going in the correspondingly correct directions. Each of the above carbohydrates has a base structure that contains one oxygen atom in its ring. Glucose and fructose are both hexoses (6- carbon sugars); glucose is a pyranose and fructose is a furanose. The sugars are so called because their base ring structures are based upon furan and pyran. 19
20 Monosaccharides -- Furanoses These two compounds (furan and pyran) provide the framework for numerous monosaccharides (single sugars). Ribose is found in ribonucleic acid (RNA); xylose is wood sugar and is non-fermentable; arabinose is gum sugar -- it may sometimes be found in urine. A form of ribose is found in deoxyribonucleic acid (DNA): deoxy-ribose. Ribose must lose the - OH on the 2' carbon to become deoxy-ribose. Note that ribose, xylose and arabinose are pentoses (5 carbon sugars). In addition, the proper names of the above furanoses are: D-ribofuranose, D-xylofuranose and D-arabinofuranose. 20
21 Pyranoses Glucose, we've briefly discussed. Galactose is of significance in that it forms half of the disaccharide (double sugar) lactose (milk sugar). Mannose is a plant sugar. Note that these sugars differ in their geometric structure simply by the orientation of the -OH groups. Indeed, the orientation of the -OH groups -- only differing by the position of 1 -OH group -- changes the sweetness of the sugar. To remember these sugars, I've got three mnemonics for you. They are based off the first 4 carbons (1,2,3,4) and #5 carbon has no -OH group on it and #6 carbon is always up and to the left for our purposes. To remember glucose, the mnemonic is DDUD: down, down, up, down. This describes the orientation of the -OH groups on the first four carbons. Galactose is DDUU, while mannose is DUUD. The proper names for the above pyranoses are: D-glucopyranose, D-galactopyranose and D- mannopyranose. 21
22 Terminology Epimer: a class of isomers that are non-superposable, non-mirror images of one another. Anomer: is a special type of epimer. It is an isomer of a saccharide in the cyclic form (Hayworth projection) that differs only in its configuration at the hemiacetal carbon in aldoses or hemiketal carbon in ketoses, which is also called the anomeric carbon. If the the hydroxyl group on the anomeric carbon is in the down position of glucose, then the sugar is an alpha ( ) anomer. If, however, that hydroxyl group is in the up position, then the sugar is a beta ( ) anomer. For example, α-dglucopyranose and β-d-glucopyranose, the two cyclic forms of glucose, are anomers. 22
23 Anomeric Carbon We can go another step in naming monosaccharides based upon the position of the -OH on carbon #1. This carbon is called the anomeric carbon. An anomer is a special type of epimer (a class of isomers that are non-superposable, nonmirror images of one another). It is an isomer of a saccharide in the cyclic form (Hayworth projection) that differs only in its configuration at the hemiacetal carbon in aldoses or hemiketal carbon in ketoses, which is also called the anomeric carbon. When the -OH group is down on carbon #1, that is said to be in the -configuration. When the -OH group is up on carbon #1, it is said to be in the - configuration. Humans metabolize monosaccharides in the - configuration. The graphic illustrates glucose in both configurations. 23
24 The Disaccharides We are interested in only three of them: Sucrose is table sugar. Sucrose consists of one molecule of glucose and one molecule of fructose bonded together. Sucrose 24
25 Maltose Maltose is also known as malt sugar; Maltose consists of two glucose molecules bonded together; Lactose Lactose is milk sugar; Lactose consists of one molecule of galactose and one molecule of glucose (pretty clever considering that young animals living on mother's milk use the glucose for quick energy and send the galactose to their livers where it will stored for future energy needs as glycogen) are bonded together. 25
26 Glycoside Bonds The bonds that hold these sugars together are called glycoside bonds. An oxygen atom between the first and 4th carbons of each respective glucose molecule (see above) connects the two glucose molecules linked together in maltose. Since the linkage is from an -OH group on the left glucose molecule that is in the -configuration, this is called an 1 to 4 link, or 1 4 link. Since the linkage between the galactose molecule and the glucose molecule starts in the -configuration and is also between the 1st and 4th carbons via an oxygen atom, this is called a 1 4 link, or 1 4 link. The linkage between the glucose and fructose molecules in sucrose occurs through the 1st and 2nd carbons of glucose and fructose, respectively. This is an 1 to 2 link, or 1 2 link. Refer to the above graphics for clarification of the shapes of these bonds. Remember, also, that there are NO carbons in the actual glycoside bond: ONLY an oxygen atom links the monosaccharides together. 26
27 The Polysaccharides We are interested in three polysaccharides: starch, glycogen and cellulose. Starch consists of two forms of complex carbohydrates: amylose and amylopectin Amylose Is the less abundant form and forms a helix Iodine "crawls" into the helix and forms inclusion compounds which turns a dark blue Starch is found in PLANTS Amylose is hydrolyzed by amylase in our mouths Amylopectin The more abundant form in starch Forms BOTH 1 4 and 1 6 links (shown in glycogen below) Similar to glycogen due to the branching caused by the 1 6 links Has lesser helix amount, hence less iodine binding; the color obtained is a red-violet color Found in PLANTS 27
28 Glycogen is found in ANIMALS, specifically in the skeletal muscle and liver of animals. It is also found in fetal hearts and fetal lungs. Fetal hearts run off glycogen while adult hearts run off lipids. The glycogen in fetal lungs is necessary to form surfactant to make oxygen passage into the body from the lungs easier when the fetus is in room air rather than the womb. Glycogen branches due to the same sort of linkages found in amylopectin ( 1 6 links), Glycogen Having both 1 to 4 and 1 to 6 links lets the glycogen molecule become very dense and be very efficient for storage. Approximately 1/3 of the weight of the human liver is glycogen. The branches that are formed are then "de-formed" by an enzyme called the "debranching enzyme" when glycogen is needed for energy. Although glycogen has some helix, it is more like amylopectin: it forms less inclusion compounds with iodine. The color obtained is amber red and may be stabilized with the addition of the dihydrate of calcium chloride. Each glycogen molecule contains approximately 100,000 molecules of glucose per molecule of 28 glycogen.
29 Cellulose Cellulose, our last carbohydrate, is a bit different. In order to hold the glucose molecules together, they are linked by 1 4 linkages. Humans can not metabolize these links, while ruminants can. Ruminants digest cellulose because they have bacteria in their stomachs that contain the enzyme cellulase that hydrolyzes these links. Cellulose is the most abundant carbohydrate on earth; humans utilize it as dietary fiber. 29
30 Reducing Sugars ANY CHO which can reduce an alkaline solution of Cu(II) ALL monosaccharides Lactose and Maltose NOT sucrose!!!!!! IN GENERAL: if a free OH is on the ANOMERIC CARBON, the CHO is a reducing sugar. 30
31 Benedict s/fehling s Tests 31
32 Cont d Benedict s test is basis for Clinitest: urine test for urinary sugar excretion in diabetes Green: NO glucose in urine Brick Red: > 2 g glucose/100 ml urine 32
33 Barfoed Test In acid solution, Cu(II) is a WEAKER oxidizing agent Used to differentiate BETWEEN mono- and di-saccharides If the solutions are heated for > 10 minutes, false positive results will be obtained. 33
34 Tollen s Test Alkaline solution of Ag(I) with ammonia Why ALKALINE so important? 34
35 Tollen s, Cont d Alkaline Conditions favor ENEDIOL formation of BOTH glucose and fructose HOW?????? 35
36 Enediol Formation 36
37 Base Catalyzes Enediol Formation Classically, aldehydes, but NOT ketones rxt with Tollen s Reagent Due to TAUTOMERIZATION, fructose CAN react with Tollen s Reagent. 37
38 Tautomerization fast reorientation of isomers with movement of an H and a double bond. Fructose (keto) and Enediol (enol) are tautomers Hence, ketoses react with Benedict s, Fehling s and Tollen s reagents as well as do aldoses, anyways. Reiterate: Ketose Enediol Aldose 38
39 Bromine Water Bromine water oxidizes aldoses but NOT ketoses. 39
40 Fiber in the Diet The main part of the wheat kernel is the starchy endosperm, which comprises more than 80 percent of the kernel weight and is the part of the kernel that eventually will be milled to wheat flour. Wheat bran is the papery brown coating of a whole grain of wheat. Removed during milling, wheat bran is sold packaged and in bulk. A quarter cup provides 6 g fiber. Wheat germ is the embryo of the wheat kernel. It is removed during the milling of white flour but is left intact in wholewheat varieties. Wheat germ is sold raw or lightly toasted
41 Dietary Fiber Fiber 1) increases satiety rate, 2) reduces nutrient bioavailability, 3) reduces energy density, 4) alters hormonal response and 5) alters thermogenesis in obesity. The mechanisms seem to be that fiber 1) prolongs chewing and swallowing movements; 2) increases fecal fat content; 3) inhibits absorption of carbohydrate in high fiber foods; 4) increases transit time and 5) alters the action of insulin, gut glucagon and other intestinal hormones.
42 Fiber The common idea is that fiber gives bulk/roughage to aid in defecation. Burkitt noticed in 1983 in Africa that people eating high fiber diets had reduced incidences of GI disease. Epidemiological evidence shows the converse in developed countries (remember the paradox with pregnancy and lactation, too). Current research classifies fiber into 3 groups based upon their structure (chemistry) and properties.
43 Cellulose Cellulose comes from and is a constituent of the main cell wall. It is poly- -glucopyranosyl- -glucopyranoside. It is insoluble fiber and holds water. It acts as a laxative and reduces colonic intraluminal pressure. It also binds minerals.
44 Cellulose Fiber Sources Selected sources of cellulose fibers include grains (bran, whole wheat, whole rye), fruits (apples, pears) and vegetables (beans, peas, cabbage family, root vegetables and fresh tomatoes).
45 The Non-Cellulose Polysaccharides Summarized, Below Dietary Fiber Class Plant Part of Origin Function Hemicellulose Secretions; cell wall material Mostly insoluble; holds water, increases stool bulk; reduces colonic pressure; binds bile acids Pectins Intracellular cement material Soluble, binds cholesterol and bile acids Gums Special cell secretions Soluble; binds cholesterol and bile acids; slows gastric emptying; provide fermentable material for colonic bacteria with production of volatile fatty acids and gas Mucilages Cell secretions Soluble; slows gastric emptying time; fermentable substrate for colonic bacteria; binds bile acids Algal substances Algae, seaweed Soluble; slows gastric emptying time; fermentable substrate; binds bile acids
46 Non-Cellulose Polysaccharides Sources Hemicelluloses may be obtained from bran, cereals and whole grains. Pectins may be obtained from fruits (apples, citrus, fruits, berries -- especially strawberries) and vegetables (green beans, carrots). Gums may be found in grains (oatmeal) and vegetables (dried beans and other legumes). Gums, mucilages and algal substances are used as food product thickeners and stabilizers.
47 Lignins Non-carbohydrate fiber includes lignin, which comes from the woody part of plants. It is insoluble, it is an anti-oxidant and it binds bile acids and metals.
48 Lignin Sources Lignins may be found in grains (whole wheat, whole rye), fruits (strawberries, peaches, pears, plums) and vegetables (mature vegetables).
49 There are 4 Physiological Effects of Dietary Fiber 1) water absorption, The absorption of water leads to a bulkier stool and gives a laxative effect. This influences the transit time of the food mass through the bowel. 2) binding effect, The rate of absorption of nutrients is effected by the amount of time in the bowel. Non-cellulose fibers bind bile salts and cholesterol, preventing their absorption. Some of these binding effects are undesirable: excessive amounts of dietary fiber bind iron, zinc and/or calcium. These binding effects also effect lipid levels. 3) relation to colonic bacteria Some non-cellulose dietary fibers (gums) provide fermentation substances for colon bacteria. This produces short chain fatty acids and gas. 4) satiety Satiety is enhanced by fiber, since there is extra bulk. These foods also take more time to eat. This property regulates the amount of food eaten, it increases transit time in the bowel and it contributes to the management of obesity and diabetes.
50 Fiber and Cancer For a number of years it was thought that fiber in the diet reduced the incidence of colon cancer by altering the kinds and amounts of bile acids and their metabolites in the bowel. It was thought that these chemicals altered the structure of the bowel, its cell turnover rate and the function of the resulting cell. An extensive study in late 1998 to mid-1999, though, has shown that this is not the case. It seems that fiber does NOT protect against bowel cancer. Interestingly enough, it appears that calcium may play a significant role in reducing the incidence of colon cancer.
51 Fiber and GI Disorders In other GI disorders (diverticular disease, constipation, hiatal hernia and hemorrhoids), fiber 1) reduces pressure from within the intestinal lumen and 2) increases the diameter of the intestinal lumen, thus allowing the intestinal tract to contract more, propelling its contents more rapidly and inhibiting segmentations. Fiber seems to do these by 1) increasing transit time, 2) increasing water absorption, resulting in a larger, softer stool and 3) increasing pressure and weakness along the walls of the GI tract.
52 Diabetes and Fiber In the case of diabetes mellitus, it seems that there are at least 5 effects of fiber in the diet: 1) it reduces fasting blood sugar levels; 2) it reduces glycosuria; 3) it reduces insulin requirements; 4) it increases insulin sensitivity and 5) it inhibits postprandial hyperglycemia. The modes of action are to, overall, slow carbohydrate absorption across the bowel. Fiber appears to do this by one of 5 mechanisms: 1) it delays gastric emptying time; 2) it forms gels with pectin or guar gum in the small bowel, thus impeding carbohydrate absorption; 3) by "protecting" carbohydrates from enzymatic activity with a fibrous coat; 4) by allowing "protected" carbohydrates to escape into the large bowel where they are digested by bacteria and 5) they alter gut hormone levels (glucagon) to enhance glucose metabolism in the liver.
53 Fiber and Coronary Artery Disease In coronary artery disease (aka coronary heart disease), fiber 1) inhibits recirculation of bile acids and 2) reduces triglyceride and cholesterol levels (epidemiological evidence). The putative mechanisms that this takes include 1) fiber alters bacterial metabolism of bile acids; 2) fiber alters bacterial flora, resulting in a change in metabolic activity; 3) fiber forms gels that bind bile acids; 4) fiber alters the function of pancreatic and intestinal enzymes; 5) fiber reduces insulin levels; 6) fiber binds cholesterol, preventing absorption and 7) fiber slows fat absorption by forming gel matrices in the intestine.
54 Carbohydrate Metabolism The Nickel Tour 54
55 Embden-Meyerhof-Parnas Overview Carbohydrate metabolism seems to get most of the time and attention throughout all of metabolism This pathway begins with a sixcarbon sugar, glucose, and ends with 2 threecarbon intermediates (pyruvate) as glucose is oxidized. 55
56 Embden-Meyerhof-Parnas for glucose to be adequately catabolized, it has to be "trapped" in cells 1. Hexokinase 2. Phosphoglucoisomerase 3. PFK 4. Aldolase 5. Triose phosphate isomerase 6. G-3-PDH 7. Phosphoglycerate kinase 8. Phosphoglyceromutase 9. Enolase 10. Pyruvate kinase 56
57 2,3-bPG 2,3-bPG reduces the affinity of Hb for O 2 is a primary compensatory factor in going to higher altitudes -- Is a side-rxn of EMP E.g., if live in San Francisco, to adapt (short term) to life at Lake Tahoe, body increases [2,3-bPG] so more oxygen is released to the cells in the body 57
58 Anaerobic/Fermentative Metabolism The stoichiometry aerobic AND anaerobic -- (review your Chem 121) is that per molecule of glucose (1 six-carbon sugar), TWO molecules of three-carbon sugars are formed, i.e., one times six is six, as are two times three. 58
59 EMP Stimulators and Inhibitors Inhibitors Stimulators ( Activators ) PFK: ADP and AMP Low energy turns on EMP PFK: Citrate and ATP G-3-PDH: AsO 3-4 Enolase: fluoride ion Pyruvate kinase: ATP High energy turns off EMP arsenate inhibits because it looks like phosphate 59
60 ATP Summary Used and Gained -- AEROBIC ATP Used Hexokinase: -1 PFK: -1 ATP Gained G-3-PDH: +6 ( ) Phosphoglycerate kinase: +2 Total USED = 2 ATP Pyruvate kinase: +2 Total GAINED: 10 ATP Overall: 8 ATP produced 60
61 ATP Summary Used and Gained ANAEROBIC (FERMENTATIVE, too) ATP Used Hexokinase: -1 ATP Gained G-3-PDH: +6 ( ) PFK: -1 LDH (alcohol DH) : -6 ( ) Total ATP USED: -8 ATP Phosphoglycerate kinase: +2 Pyruvate kinase: +2 Total GAINED: 10 ATP In anaerobic glycolysis (fermentation), # ATP go down from 8 to 2 because the NADH and NAD cycle 61
62 62
63 Voges-Proskauer Metabolism and Application 63
64 Principles Behind the VP Test -naphthol added first as it enhances the red color sensitivity using second may give false negative; KOH and oxygen in the air oxidize the secondary alcohol to the ketone; -naphthol is also known as Barritt s Reagent A KOH is also known as Barritt s Reagent B 64
65 Application Voges-Proskauer test is used in the identification of bacteria by identifying the presence of acetoin (3-keto-2-butanol) in the bacterial media: Voges-Proskauer Positive Bacteria Include: E. aerogenes S. marcescens K. pneumoniae Voges-Proskauer Negative Bacteria Include: E. coli Shigella Citrobacter C. freundii Yersinia Voges-Proskauer Inconclusive Bacteria Include: Proteus 65
66 Butylene glycol (2,3-butanediol) pathway Initiation of the butylene glycol pathway, as it s also known, derives from EMP: the formation of pyruvate. Two pyruvates are condensed with a concomitant loss of one CO 2 and the reduction of one of the ketones to an alcohol to form acetolactate. The enzyme that catalyzes this reaction is acetolactate synthetase. 66
67 Acetolactate is commonly known as an intermediate in the synthesis of BCAA s. If BCAA s are not needed by the prokaryote, acetolactate decarboxylase removes the carboxyl group to form [presumably] 3-ketobutanol. 67
68 Again, presumably,an isomerase rearranges the 1-ol to a 2-ol to form acetoin. It may very well be that acetoin is a branch point to regulate the bacterium or bacteria 68
69 Acetoin (top pathway) is reduced to 2,3-butanediol by, presumably, NADH. The reaction is catalyzed by acetoin reductase. 69
70 Acetoin (bottom pathway) is oxidized and thiolated to form acetyl-coa. The enzyme that catalyses this reaction is acetoin dehydrogenase. An additional product formed is acetaldehyde. Its purpose is not known. The acetyl CoA feeds into the TCA. 70
71 It seems likely that the 2,3- butanediol (butylene glycol) is probably synthesized during times when the bacteria has sources of energy and needs to regulate its population growth/decline. Top pathway. It seems likely that the acetoin reduction to release acetaldehyde and acetyl-coa are for when the bacterium lacks energy sources in its "diet" and uses a portion of the acetoin catabolically. Bottom pathway. It also seems likely that the acetaldehyde that is generated is oxidized and thiolated to form more acoa 71
72 Fructose Metabolism Every now and again, a diabetic patient's parent, sibling or other relative reads about how diabetes is a disease of glucose metabolism. On occasion, they read a little more and discover that fructose is a carbohydrate, but not glucose. They then come in to see you as the health care person who knows something about diabetes and ask you, "Since fructose isn't glucose, can I substitute all of my relative's carbohydrate needs with fructose?" Your answer is, of course, no. So, why is it that your answer is no? The catabolism of fructose and how it intertwines with triglyceride (TGS) synthesis follows. 72
73 Fructose Metabolism 1. Hexokinase (adipose tissue) 2. Fructokinase (liver) 3. F-1-P aldolase 4. Triose kinase 5. Triose phosphate isomerase (TPI) 6. DHAP DH 7. Glycerol phosphate DH 8. Phosphatase 9. Glycerol kinase 73
74 Hereditary Fructose Intolerance This disorder is inherited as an autosomal recessive disorder. There is a fructose-1-phosphate (F-1-P) aldolase deficiency. This disease causes hypoglycemia with increased accumulation of F-1-P in tissues. The patient fails to thrive and has nausea and vomiting (N/V), jaundice, an enlarged liver, which may develop into liver failure, proteins and amino acids in the urine and tyr in the urine, as well. Diagnostic testing is to "pre-load" the patient with fructose and observe for hypoglycemia and hypophosphatemia. Therapy is to discontinue cane sugar from the diet. Patients must double check over the counter (OTC) medications for sucrose additions as tablet binders. If the diet is discontinued, the patient has an increased risk of growth failure; on the diet, the patient will grow relatively normally. 74
75 Galactose Metabolism Galactose is an important constituent of lactose, Galactose may be used in the synthesis of glycoproteins Galactose may be used in the synthesis of glycogen 75
76 Galactose Metabolism 1. Galactokinase 2. Galactose-1- phosphate uridyl transferase 3. Lactose synthetase (BOTH catalytic unit and specificity protein) 4. Lactose synthetase (catalytic unit only) 5. UDP-galactose-4- epimerase 6. UDP-glucose pyrophosphorylase 7. Glycogen synthetase 76
77 Glycogen Metabolism Regulated by camp Phosphodiesterase (PDE) inhibited by: Theophylline Caffeine Theobromine These compounds are called xanthines. When PDE is inactivated, camp levels build up, making it easier for patients to breathe. 77
78 In General 78
79 In General Glycogenolysis (destruction of glycogen) Glycogenesis (production of glycogen) As the body doesn't like to be confused during times of stress, camp inhibits glycogenesis and activates glycogenolysis. One example of this occurs when epinephrine binds with the appropriate receptor on the cell membrane of a target cell. 79
80 In Detail 80
81 Hexose Monophosphate Shunt (Pentose Phosphate Pathway) 1. G-6-PDH 2. Lactonase 3. 6-P-gluconate DH 4. Phosphopentose isomerase 5. Phosphopentose epimerase 6. Transketolase -- (2 C transfer) 7. Transaldolase -- (3 C transfer) 8. Transketolase -- (2 C transfer) 81
82 Hexose Monophosphate Shunt (Pentose Phosphate Pathway): Significance 2 NADPH reductive power Ribose nucleoside/nucleotide synthesis Hexose Monophosphate Shunt (Pentose Phosphate Pathway): Clinical Significance People with G-6-PDH deficiency don t make enough NADPH to reduce G-S-S-G and causes health problems 82
83 Hexose Monophosphate Shunt (Pentose Phosphate Pathway): Clinical Significance 83
84 Gluconeogenesis Gluconeogenesis is NOT the absolute reverse of glycolysis. Some enzymes are the same -- 4 are NOT When the body produces new glucose, it utilizes various substrates as necessary. These include the carbon skeletons of amino acids and anaerobic end-products of catabolism. The carbon skeletons of Gly, Ala, Thr, Ser and Cys feed into gluconeogenesis via pyruvate, as does lactate. The carbon skeletons of Asn and Asp feed in to OAA. 84
85 Gluconeogenesis 85
86 Gluconeogenesis Remember, though, that the purpose of phosphorylation of glucose in the first place is to trap it in the cell. Since that is a trapping mechanism, there has to be a way to remove the phosphate so that the newly formed glucose can get "dumped" into the blood. ONLY the liver cells contain G-6-phosphatase (4) that cleaves off the phosphate so that the glucose may be routed through the body for use as needed by its cells. Which tissue does NOT require insulin for glucose uptake? 86
87 These pathways are all fine and dandy, but of what significance are anaerobic glycolysis and gluconeogenesis? The significance is the Cori cycle, named after Dr. and Dr. Cori who discovered it. As muscle tissue anaerobically catabolizes glucose for whatever energy needs, lactate is produced. Lactate is small enough to freely diffuse across the muscle cell membranes into the blood. Once in the blood, it travels to the liver where it diffuses into liver cells. The lactate is used in gluconeogenesis to synthesize more glucose in the liver, which is then sent back to the muscles for utilization until aerobic catabolism catches up or until the muscle needs no more glucose. The Cori cycle buys time and changes the metabolic burden to the liver. 87
88 Pyruvate Dehydrogenase Complex -- AEROBIC Pyruvate is first decarboxylated by PDH in the presence of thiamine pyrophosphate (TPP) to form carbon dioxide (2 moles, remember) and an acetol-tpp intermediate. The acetol is transferred from TPP to lipoic acid by DHLTA and, then thiolated with the addition of HSCoA (Coenzyme A) and release of reduced lipoic acid and acetyl CoA (acoa). 88
89 Krebs Cycle -- TCA As long as the system (cell and/or tissues and/or body) remains in an aerobic state and fuel is present, the TCA will continue to provide energy to the cells. 89
90 Krebs Cycle -- TCA 1. Citrate synthetase 2. Aconitase 3. Isocitrate DH 4. KGDH 5. Succinyl CoA synthetase 6. Succinate DH 7. Fumarase 8. Malate DH 90
91 TCA: ATP Summary All MADE PDH: +6 ( ) icdh: +6 ( ) KGDH: +6 ( ) scoa synthetase: +2 SDH: +4 ( ) MDH: +6 ( ) TOTAL: +30 Glycolysis: +8 WAY TOTAL: = 38 ATP under aerobic conditions 91
92 TCA Regulators Energy is the PRIME regulator!! Enzyme Regulators Positive Effectors (stimulators) ADP on icdh Negative Effectors (inhibitors) ATP, acoa, NADH on PDH ATP on CS scoa and NADH on -kgdh 92
93 Carbon Tracing in Glycolysis Radioactive carbon Oxygen Regular carbon 93
94 Carbon Tracing in the TCA Regular Carbon Radioactive Carbon Why 100% labeling at succinate? SYMMETRY! 94
95 ATP Synthesis by Tissue: Clarification NAD/NADH will NOT cross mito membranes how get in there? Aspartate-Malate Shuttle! In HEART and LIVER = 38 ATP from aerobic glycolysis and TCA Note: OAA won t cross mito membrane, either 95
96 ATP Synthesis by Tissue: Clarification Any other way? YES!!!! Glycerol phosphate shuttle in SKELETAL MUSCLE Has an effect on ATP production because NADH = 3 ATP and FADH 2 = 2 ATP Hence, 36 ATP produced in aerobic glycolysis and TCA in MUSCLE 96
97 In order to fully understand why one mole of NADH is worth 3 ATP molecules and why one mole of FADH 2 is worth 2 ATP molecules, we need to discuss how each is oxidized by electron transport (ET) to form ATP through oxidative phosphorylation (OP; the combination of the two is called ET-OP) NADH to NAD and FADH 2 to FAD = Oxidative; ADP phosphorylated to ATP = Phosphorylation
98 ET-OP Inhibitors Oligomycin inhibits the proton pump
99 Co-Q Ubiquinone 10 99
100 Proton Pump Functioning 1. H + moving out of matrix at I, III, IV 2. ph gradient established F o opens and H + run through to drive ATP ase 3. [H + ] in IMS decreasing to point of maximal flow into matrix 4. H + being recycled and re-starting cycle F o fully closed 100
101 Application of Proton Pump in ETOP -- WAT White Adipose Tissue Not as many mito 101
102 Application of Proton Pump in ETOP -- BAT Brown Adipose Tissue Lots of Mito Thermogenesis T 4 and T 3 regulate Fatty Acids act as uncouplers NE regulates FA release in BAT 102
103 EtOH Metabolism This sequence makes mito an incredibly REDUCED environment! With >>> NADH, this inhibits ALL NAD-requiring enzymes THEREFORE: EtOH typically used in fat synthesis rather than in oxidation 103
104 Energy Sources from Intermediary Metabolism The first system is the phosphagen system. In this system, the source of the energy is ATP. Remember that during muscular contraction, ATP is hydrolyzed to ADP, P i and energy. When this happens, there is only enough energy for 5-6 seconds. So how do our cells get additional energy? Our cells get it via a compound called phosphocreatine (PCr). The concentration of PCr is about 2-3 times greater than the concentration of ATP. When PCr is available, it is hydrolyzed to Cr and P i and energy. The P i is used to re-phosphorylate ADP to make more ATP. This gives us about 15 seconds of maximal contractions and is used for short bursts. 104
105 PCr Shuttle Quick View To give you an idea of what is involved in PCr utilization and synthesis, let's examine the PCr shuttle in cardiac and skeletal muscle. This shuttle increases incredibly the movement/transport of high-energy phosphate (ATP) from the matrix of the mitochondrion to the cytosol of the cell. 105
106 In the first step (1), an ADP-ATP translocase re-phosphorylates ADP to form ATP in the mitochondrial matrix. This occurs via electron transport/oxidative phosphorylation (the "ETOP" in the graphic). When the ATP is "dumped" into the intermembrane space, it is reacted with creatine via a mitochondrial creatine kinase (6) to form PCr. The PCr is then transported via a creatine-creatine phosphate (C:PCr) transport pore (2) into the cytosol to "dump" into a cytosolic PCr store. 10 6
107 The cytosolic PCr store is derived from an EMP-linked (glycolysis-linked) creatine kinase (CK) (3) and another CK that maintains the equilibrium between C and PCr (or ATP and ADP, if you prefer)(4). The utilization of the PCr from the cytosolic store occurs via an ATP dependent CK that cranks out hugely elevated levels of ATP (5). The ATP is used by processes requiring high energy, e.g., contracting muscle
108 Of significance is that there are three isozymes (enzymes with the same function that are found in different tissues and are slightly different, structurally) of CK: CK-BB CK-MB CK-MM = CK-1 = CK-2 = CK-3 Brain, uterus, prostate, lung Heart (and skeletal muscle) Skeletal muscle (and cardiac, too) 108
109 Creatine, Urea, TCA and Malate-Aspartate Shuttle Interconnections 1. Creatine: is a nitrogenous organic acid that occurs naturally in vertebrates and helps to supply energy to muscle and nerve cells. In humans and animals, approximately half of stored creatine originates from food (mainly from fresh meat). Ninety-five percent of creatine is later stored in the skeletal muscles. 2. Creatinine: is a break-down product of creatine phosphate in muscle, and is usually produced at a fairly constant rate by the body (depending on muscle mass). Creatinine is chiefly filtered by the kidney, though a small amount is actively secreted. There is little-to-no tubular reabsorption of creatinine. If the filtering of the kidney is deficient, blood levels rise. Men tend to have higher levels of creatinine because they have more skeletal muscle than women. Vegetarians tend to have lower creatinine levels, because vegetables contain no creatine. 109
110 Creatine/CreatinINe 1. Mitochondrial Arginine-glycine: amidinotransferase (E.C ) 2. ga Transporter (from mito to cytosol) 3. Cytosolic S-adenosylmethionine :guanidinoacetate-n-methyltransferase (SAM:gA NMT) 4. Cytosolic Creatine kinase 5. Cytosolic non-enzymatic cyclization 110
111 Inside black lines is mito matrix Outside black lines is cytosol N-acetylglutamate is an obligatory on switch for CPS-I. Note relationships between Urea cycle, TCA, Creatine Synthesis and Asp-Malate Shuttle. 111
112 ATP Fine Tune
113 NADH: through ETS ADP-ATP Translocase coupled with Dihydrogen phospate/proton Co-Transporter Complex 1 requires 4 protons to pass on electron to Complex III Complex III requires 2 protons to pass on electron to Complex IV Complex IV requires 4 protons to reduce molecular oxygen to water 10 protons exported per NADH to make an ATP
114 FADH 2 : through ETS ADP-ATP Translocase coupled with Dihydrogen phospate/proton Co-Transporter Complex III requires 2 protons to pass on electron to Complex IV Complex IV requires 4 protons to reduce molecular oxygen to water 6 protons exported per FADH 2 to make an ATP
115 Complex V and Co-Transporter 3 protons required to turn on F o 1 proton co-transported with P i 4 protons imported to synthesize 1 ATP molecule Currently Accepted Stoichiometry # ATP 1NADH 10 protons NADH 4 protons ATP 2.5 ATP NADH # ATP 1FADH 2 6 protons FADH 2 4 protons ATP 1.5 ATP FADH 2 Suggested Source:
116 ATP Fine Tune Part 2 Type I Muscle Fibers Red Fibers Slow Twitch For Endurance Small Diameter Aerobic Lots of Mitochondria Malate-Aspartate Shuttle Liver, Heart, Kidney Back Muscles, too Type II Muscle Fibers White Fibers Fast Twitch For Explosive Bursts of Power Large Diameter Anaerobic Fewer Mitochondria -Glycerol Phosphate Shunt Skeletal Muscle, Brain Digits and Extraocular Muscles Store Glycogen
117 Special Topic Carbonic Acid-Bicarbonate Ion: Physiological Interactions 117
118 Carbon Dioxide: CO 2 CO 2 is fairly soluble in water (more soluble in cold water like in cold soda; less soluble in warm water like in "flat" soda). A saturated solution at 1 atm and 25 C is approximately 0.033M. At equilibrium only 0.17% of dissolved CO 2 is in the form of carbonic acid (H 2 CO 3 ). An aqueous solution of CO 2 is typically acidic: CO 2 (g) + H 2 O(l) H + + HCO 3 - (Remember the trick with phenolphthalein and blowing into it during titrations in CHEM 121 Lab?) 118
119 Carbon Dioxide: CO 2 CO 2 plays a major role in maintaining the ph of blood (and sea water). CO 2 is not normally transported as such, rather as HCO 3-. This occurs via an enzymatic reaction catalyzed by carbonic anhydrase: CO 2 + H 2 O H 2 CO 3 HCO H + IMPORTANT in acid/base balance
120 CO 2 origin from metabolic activity of the cells TCA!!! Normal breathing and cellular metabolism cause a steady state of CO 2 anabolism and catabolism Alter breathing, then alter CO 2 content Approximately 250 ml O 2 produces approximately 200 ml CO 2 Divide latter by former and get an RQ of about 0.8 RQ = Respiratory Quotient, a measure of metabolism About as efficient as a gasoline engine 120
121 Buffers Consist of a weak acid and its conjugate base. In this case: H 2 CO 3 and HCO 3 - The acid-base pair serves to maintain an essentially constant ph. The acid-base pair can NOT regulate excessive amounts of acid/alkaline substances secondary to a pathology of respiration or of metabolism. 121
122 Buffers, Cont d Buffers work to: Neutralize excess acid to elevate (raise; elevate) the ph Neutralize excess base to reduce (lower) the ph End result: keep ph within normal ( in human arterial blood) limits under normal circumstances This is simply a physiological TITRATION!! It just doesn t use phenolphthalein. 122
123 Under normal conditions, the bicarbonate to proton ratio is about 20 to 1 and the hydrogen ion concentration may be calculated by multiplying 24 times the ratio of pco 2 to the bicarbonate ion concentration: [ H ] pco 24 * [ HCO 2 3 ] That means, then, that the ph is proportional to the ratio of the pco 2 (the respiratory contributor to ph balance) to bicarbonate (the metabolic contributor to ph balance) ion concentration. 123
124 Bicarbonate, Protons and Their Relationship Condition: Acidic Normal Alkaline Bicarbonate to proton ratio: Shifts to: 8 to to 1 44 to 1 Left making the blood acidic or normal balance Right making the blood alkaline 124
125 Red Cells and Acid-Base Balance 125
126 Cl - and CO 2 are intertwined. Note direction of movement of CO 2 and Cl - at Lung (9) AND at RBC (5). Transport protein (antiport) moves Cl - and HCO
127 Haldane Effect With Hb(O 2 ) 4, at some pco 2 CO 2 content of blood With Hb(O 2 ) 4 at some pco 2 CO 2 content of blood 127
128 pco 2 Levels Above 70 mm Hg May decrease respirations May cause stupor, coma (CO 2 narcosis) May cause hypoxemia SLOWLY decrease the pco 2 so as to not cause posthypercapnic metabolic alkalosis 128
129 129
130 130
Carbohydrate Metabolism. The Nickel Tour
 Carbohydrate Metabolism The Nickel Tour 1 Embden-Meyerhof-Parnas Overview Carbohydrate metabolism seems to get most of the time and attention throughout all of metabolism This pathway begins with a sixcarbon
Carbohydrate Metabolism The Nickel Tour 1 Embden-Meyerhof-Parnas Overview Carbohydrate metabolism seems to get most of the time and attention throughout all of metabolism This pathway begins with a sixcarbon
Carbohydrates. Haworth Projections
 Carbohydrates There are, for all intents and purposes, 2 classes of carbohydrates: pyranoses and furanoses ( ose is the suffix for a sugar). These two classes are so called because their carbon skeletons
Carbohydrates There are, for all intents and purposes, 2 classes of carbohydrates: pyranoses and furanoses ( ose is the suffix for a sugar). These two classes are so called because their carbon skeletons
Introduction to Intermediary Metabolism. The Nickel Tour
 Introduction to Intermediary Metabolism The Nickel Tour Carbohydrate Metabolism Part I As a general rule, whenever you hear "kinase", think ATP and magnesium ions. Hexokinase is a generic enzyme, capable
Introduction to Intermediary Metabolism The Nickel Tour Carbohydrate Metabolism Part I As a general rule, whenever you hear "kinase", think ATP and magnesium ions. Hexokinase is a generic enzyme, capable
Chemistry 1120 Exam 4 Study Guide
 Chemistry 1120 Exam 4 Study Guide Chapter 12 12.1 Identify and differentiate between macronutrients (lipids, amino acids and saccharides) and micronutrients (vitamins and minerals). Master Tutor Section
Chemistry 1120 Exam 4 Study Guide Chapter 12 12.1 Identify and differentiate between macronutrients (lipids, amino acids and saccharides) and micronutrients (vitamins and minerals). Master Tutor Section
Carbohydrates. Chapter 12
 Carbohydrates Chapter 12 Educational Goals 1. Given a Fischer projection of a monosaccharide, classify it as either aldoses or ketoses. 2. Given a Fischer projection of a monosaccharide, classify it by
Carbohydrates Chapter 12 Educational Goals 1. Given a Fischer projection of a monosaccharide, classify it as either aldoses or ketoses. 2. Given a Fischer projection of a monosaccharide, classify it by
Dr. Nafith Abu Tarboush. Rana N. Talj
 2 Dr. Nafith Abu Tarboush June 19 th 2013 Rana N. Talj Review: Fischer suggested a projection in which the horizontal bonds are projecting towards the viewer and the vertical ones project away from the
2 Dr. Nafith Abu Tarboush June 19 th 2013 Rana N. Talj Review: Fischer suggested a projection in which the horizontal bonds are projecting towards the viewer and the vertical ones project away from the
Sheet #10 Dr. Mamoun Ahram Sec 1,2,3 15/07/2014. Carbohydrates 2
 Carbohydrates 2 A study Guide: Kindly,refer to the slide number,look at the structures and read the sheet notes well,most of the slides content besides all what the doctor said are mentioned here,good
Carbohydrates 2 A study Guide: Kindly,refer to the slide number,look at the structures and read the sheet notes well,most of the slides content besides all what the doctor said are mentioned here,good
BCH 4054 Chapter 19 Lecture Notes
 BCH 4054 Chapter 19 Lecture Notes 1 Chapter 19 Glycolysis 2 aka = also known as verview of Glycolysis aka The Embden-Meyerhoff Pathway First pathway discovered Common to almost all living cells ccurs in
BCH 4054 Chapter 19 Lecture Notes 1 Chapter 19 Glycolysis 2 aka = also known as verview of Glycolysis aka The Embden-Meyerhoff Pathway First pathway discovered Common to almost all living cells ccurs in
MahaAbuAjamieh. BahaaNajjar. MamoonAhram
 7 MahaAbuAjamieh BahaaNajjar MamoonAhram Carbohydrates (saccharides) can be classified into these main categories: 1. Monosaccharides, they are simplesugars (the simplest units), such as glucose, galactose
7 MahaAbuAjamieh BahaaNajjar MamoonAhram Carbohydrates (saccharides) can be classified into these main categories: 1. Monosaccharides, they are simplesugars (the simplest units), such as glucose, galactose
CARBOHYDRATE METABOLISM
 Note (Study Glycolysis, fermentation and their regulation, Gluconeogenesis and glycogenolysis, Metabolism of galactose, TCA cycle and Amphibolic role of the cycle, and Glyoxalic acid cycle, HMP shunt in
Note (Study Glycolysis, fermentation and their regulation, Gluconeogenesis and glycogenolysis, Metabolism of galactose, TCA cycle and Amphibolic role of the cycle, and Glyoxalic acid cycle, HMP shunt in
Marah Bitar. Faisal Nimri ... Nafeth Abu Tarboosh
 8 Marah Bitar Faisal Nimri... Nafeth Abu Tarboosh Summary of the 8 steps of citric acid cycle Step 1. Acetyl CoA joins with a four-carbon molecule, oxaloacetate, releasing the CoA group and forming a six-carbon
8 Marah Bitar Faisal Nimri... Nafeth Abu Tarboosh Summary of the 8 steps of citric acid cycle Step 1. Acetyl CoA joins with a four-carbon molecule, oxaloacetate, releasing the CoA group and forming a six-carbon
Chapter 20 Carbohydrates Chapter 20
 Chapter 20 Carbohydrates Chapter 20 1 Carbohydrates Carbohydrate: A polyhydroxyaldehyde or polyhydroxyketone, or a substance that gives these compounds on hydrolysis. Monosaccharide: A carbohydrate that
Chapter 20 Carbohydrates Chapter 20 1 Carbohydrates Carbohydrate: A polyhydroxyaldehyde or polyhydroxyketone, or a substance that gives these compounds on hydrolysis. Monosaccharide: A carbohydrate that
Glucose is the only source of energy in red blood cells. Under starvation conditions ketone bodies become a source of energy for the brain
 Glycolysis 4 / The Text :- Some Points About Glucose Glucose is very soluble source of quick and ready energy. It is a relatively stable and easily transported. In mammals, the brain uses only glucose
Glycolysis 4 / The Text :- Some Points About Glucose Glucose is very soluble source of quick and ready energy. It is a relatively stable and easily transported. In mammals, the brain uses only glucose
Integrative Metabolism: Significance
 Integrative Metabolism: Significance Energy Containing Nutrients Carbohydrates Fats Proteins Catabolism Energy Depleted End Products H 2 O NH 3 ADP + Pi NAD + NADP + FAD + Pi NADH+H + NADPH+H + FADH2 Cell
Integrative Metabolism: Significance Energy Containing Nutrients Carbohydrates Fats Proteins Catabolism Energy Depleted End Products H 2 O NH 3 ADP + Pi NAD + NADP + FAD + Pi NADH+H + NADPH+H + FADH2 Cell
Vocabulary. Chapter 19: The Citric Acid Cycle
 Vocabulary Amphibolic: able to be a part of both anabolism and catabolism Anaplerotic: referring to a reaction that ensures an adequate supply of an important metabolite Citrate Synthase: the enzyme that
Vocabulary Amphibolic: able to be a part of both anabolism and catabolism Anaplerotic: referring to a reaction that ensures an adequate supply of an important metabolite Citrate Synthase: the enzyme that
Chapter 18. Carbohydrates with an Introduction to Biochemistry. Carbohydrates with an Introduction to Biochemistry page 1
 Chapter 18 Carbohydrates with an Introduction to Biochemistry Carbohydrates with an Introduction to Biochemistry page 1 Introduction to Proteins, Carbohydrates, Lipids, and Bioenergetics Metabolism and
Chapter 18 Carbohydrates with an Introduction to Biochemistry Carbohydrates with an Introduction to Biochemistry page 1 Introduction to Proteins, Carbohydrates, Lipids, and Bioenergetics Metabolism and
Farah Al-Khaled. Razi Kittaneh. Mohammad Omari
 7 Farah Al-Khaled Razi Kittaneh Mohammad Omari Dr. Mamoun Ahram In this lecture we are going to talk about modified sugars. Remember: The Fischer projection can be turned into a ring structure (which is
7 Farah Al-Khaled Razi Kittaneh Mohammad Omari Dr. Mamoun Ahram In this lecture we are going to talk about modified sugars. Remember: The Fischer projection can be turned into a ring structure (which is
METABOLISM Biosynthetic Pathways
 METABOLISM Biosynthetic Pathways Metabolism Metabolism involves : Catabolic reactions that break down large, complex molecules to provide energy and smaller molecules. Anabolic reactions that use ATP energy
METABOLISM Biosynthetic Pathways Metabolism Metabolism involves : Catabolic reactions that break down large, complex molecules to provide energy and smaller molecules. Anabolic reactions that use ATP energy
GLYCOLYSIS Generation of ATP from Metabolic Fuels
 GLYCOLYSIS Generation of ATP from Metabolic Fuels - Catabolic process degradative pathway - Energy stored in sugars (carbohydrates) released to perform biological work - Transforms GLUCOSE to PYRUVATE
GLYCOLYSIS Generation of ATP from Metabolic Fuels - Catabolic process degradative pathway - Energy stored in sugars (carbohydrates) released to perform biological work - Transforms GLUCOSE to PYRUVATE
MULTIPLE CHOICE QUESTIONS
 MULTIPLE CHOICE QUESTIONS 1. Which of the following statements concerning anabolic reactions is FALSE? A. They are generally endergonic. B. They usually require ATP. C. They are part of metabolism. D.
MULTIPLE CHOICE QUESTIONS 1. Which of the following statements concerning anabolic reactions is FALSE? A. They are generally endergonic. B. They usually require ATP. C. They are part of metabolism. D.
Chapter-8 Saccharide Chemistry
 Chapter-8 Saccharide Chemistry Page 217-228 Carbohydrates (Saccharides) are most abundant biological molecule, riginally produced through C 2 fixation during photosynthesis I (C 2 ) n or - C - I where
Chapter-8 Saccharide Chemistry Page 217-228 Carbohydrates (Saccharides) are most abundant biological molecule, riginally produced through C 2 fixation during photosynthesis I (C 2 ) n or - C - I where
Major Pathways in Carbohydrate Metabolism
 Major Pathways in Carbohydrate Metabolism 70 Stage 1: Digestion of Carbohydrates In Stage 1, the digestion of carbohydrates Begins in the mouth where salivary amylase breaks down polysaccharides to smaller
Major Pathways in Carbohydrate Metabolism 70 Stage 1: Digestion of Carbohydrates In Stage 1, the digestion of carbohydrates Begins in the mouth where salivary amylase breaks down polysaccharides to smaller
Carbohydrates. Monosaccharides
 Carbohydrates Carbohydrates (also called saccharides) are molecular compounds made from just three elements: carbon, hydrogen and oxygen. Monosaccharides (e.g. glucose) and disaccharides (e.g. sucrose)
Carbohydrates Carbohydrates (also called saccharides) are molecular compounds made from just three elements: carbon, hydrogen and oxygen. Monosaccharides (e.g. glucose) and disaccharides (e.g. sucrose)
Chapter 4: Sugars, Starches and Fibers. Copyright 2012 John Wiley & Sons, Inc. All rights reserved.
 Chapter 4: Sugars, Starches and Fibers Copyright Student learning outcomes: At the end of this chapter, you should be able to: Compare and contrast whole grains and refined sugars Compare and contrast
Chapter 4: Sugars, Starches and Fibers Copyright Student learning outcomes: At the end of this chapter, you should be able to: Compare and contrast whole grains and refined sugars Compare and contrast
Topic 4 - #2 Carbohydrates Topic 2
 Topic 4 - #2 Carbohydrates Topic 2 Biologically Important Monosaccharide Derivatives There are a large number of monosaccharide derivatives. A variety of chemical and enzymatic reactions produce these
Topic 4 - #2 Carbohydrates Topic 2 Biologically Important Monosaccharide Derivatives There are a large number of monosaccharide derivatives. A variety of chemical and enzymatic reactions produce these
Carbohydrates. What are they? What do cells do with carbs? Where do carbs come from? O) n. Formula = (CH 2
 Carbohydrates What are they? Formula = (C 2 O) n where n > 3 Also called sugar Major biomolecule in body What do cells do with carbs? Oxidize them for energy Store them to oxidize later for energy Use
Carbohydrates What are they? Formula = (C 2 O) n where n > 3 Also called sugar Major biomolecule in body What do cells do with carbs? Oxidize them for energy Store them to oxidize later for energy Use
Metabolism. Metabolic pathways. BIO 5099: Molecular Biology for Computer Scientists (et al) Lecture 11: Metabolic Pathways
 BIO 5099: Molecular Biology for Computer Scientists (et al) Lecture 11: Metabolic Pathways http://compbio.uchsc.edu/hunter/bio5099 Larry.Hunter@uchsc.edu Metabolism Metabolism is the chemical change of
BIO 5099: Molecular Biology for Computer Scientists (et al) Lecture 11: Metabolic Pathways http://compbio.uchsc.edu/hunter/bio5099 Larry.Hunter@uchsc.edu Metabolism Metabolism is the chemical change of
Structural Polysaccharides
 Carbohydrates & ATP Carbohydrates include both sugars and polymers of sugars. The simplest carbohydrates are the monosaccharides, or simple sugars; these are the monomers from which more complex carbohydrates
Carbohydrates & ATP Carbohydrates include both sugars and polymers of sugars. The simplest carbohydrates are the monosaccharides, or simple sugars; these are the monomers from which more complex carbohydrates
BCH 445 Biochemistry of nutrition Dr. Mohamed Saad Daoud
 BCH 445 Biochemistry of nutrition Dr. Mohamed Saad Daoud 1 Carbohydrates Carbohydrates: Compounds composed of carbon, oxygen, and hydrogen arranged as monosaccharides or multiples of monosaccharides. Most,
BCH 445 Biochemistry of nutrition Dr. Mohamed Saad Daoud 1 Carbohydrates Carbohydrates: Compounds composed of carbon, oxygen, and hydrogen arranged as monosaccharides or multiples of monosaccharides. Most,
Ch13. Sugars. What biology does with monosaccharides disaccharides and polysaccharides. version 1.0
 Ch13 Sugars What biology does with monosaccharides disaccharides and polysaccharides. version 1.0 Nick DeMello, PhD. 2007-2015 Ch13 Sugars Haworth Structures Saccharides can form rings. That creates a
Ch13 Sugars What biology does with monosaccharides disaccharides and polysaccharides. version 1.0 Nick DeMello, PhD. 2007-2015 Ch13 Sugars Haworth Structures Saccharides can form rings. That creates a
Energy Production In A Cell (Chapter 25 Metabolism)
 Energy Production In A Cell (Chapter 25 Metabolism) Large food molecules contain a lot of potential energy in the form of chemical bonds but it requires a lot of work to liberate the energy. Cells need
Energy Production In A Cell (Chapter 25 Metabolism) Large food molecules contain a lot of potential energy in the form of chemical bonds but it requires a lot of work to liberate the energy. Cells need
Chapter 24 Lecture Outline
 Chapter 24 Lecture Outline Carbohydrate Lipid and Protein! Metabolism! In the catabolism of carbohydrates, glycolysis converts glucose into pyruvate, which is then metabolized into acetyl CoA. Prepared
Chapter 24 Lecture Outline Carbohydrate Lipid and Protein! Metabolism! In the catabolism of carbohydrates, glycolysis converts glucose into pyruvate, which is then metabolized into acetyl CoA. Prepared
Introduction to Carbohydrate metabolism
 Introduction to Carbohydrate metabolism Some metabolic pathways of carbohydrates 1- Glycolysis 2- Krebs cycle 3- Glycogenesis 4- Glycogenolysis 5- Glyconeogenesis - Pentose Phosphate Pathway (PPP) - Curi
Introduction to Carbohydrate metabolism Some metabolic pathways of carbohydrates 1- Glycolysis 2- Krebs cycle 3- Glycogenesis 4- Glycogenolysis 5- Glyconeogenesis - Pentose Phosphate Pathway (PPP) - Curi
CITRIC ACID CYCLE ERT106 BIOCHEMISTRY SEM /19 BY: MOHAMAD FAHRURRAZI TOMPANG
 CITRIC ACID CYCLE ERT106 BIOCHEMISTRY SEM 1 2018/19 BY: MOHAMAD FAHRURRAZI TOMPANG Chapter Outline (19-1) The central role of the citric acid cycle in metabolism (19-2) The overall pathway of the citric
CITRIC ACID CYCLE ERT106 BIOCHEMISTRY SEM 1 2018/19 BY: MOHAMAD FAHRURRAZI TOMPANG Chapter Outline (19-1) The central role of the citric acid cycle in metabolism (19-2) The overall pathway of the citric
2. What is molecular oxygen directly converted into? a. Carbon Dioxide b. Water c. Glucose d. None of the Above
 Biochem 1 Mock Exam 3 Chapter 11: 1. What is glucose completely oxidized into? a. Carbon Dioxide and Water b. Carbon Dioxide and Oxygen c. Oxygen and Water d. Water and Glycogen 2. What is molecular oxygen
Biochem 1 Mock Exam 3 Chapter 11: 1. What is glucose completely oxidized into? a. Carbon Dioxide and Water b. Carbon Dioxide and Oxygen c. Oxygen and Water d. Water and Glycogen 2. What is molecular oxygen
Chemical Energy. Valencia College
 9 Pathways that Harvest Chemical Energy Valencia College 9 Pathways that Harvest Chemical Energy Chapter objectives: How Does Glucose Oxidation Release Chemical Energy? What Are the Aerobic Pathways of
9 Pathways that Harvest Chemical Energy Valencia College 9 Pathways that Harvest Chemical Energy Chapter objectives: How Does Glucose Oxidation Release Chemical Energy? What Are the Aerobic Pathways of
Chapter 23 Carbohydrates and Nucleic Acids. Carbohydrates
 Chapter 23 Carbohydrates and Nucleic Acids Carbohydrates Synthesized by plants using sunlight to convert CO 2 and H 2 O to glucose and O 2. Polymers include starch and cellulose. Starch is storage unit
Chapter 23 Carbohydrates and Nucleic Acids Carbohydrates Synthesized by plants using sunlight to convert CO 2 and H 2 O to glucose and O 2. Polymers include starch and cellulose. Starch is storage unit
Cellular Respiration
 Cellular Respiration 1. To perform cell work, cells require energy. a. A cell does three main kinds of work: i. Mechanical work, such as the beating of cilia, contraction of muscle cells, and movement
Cellular Respiration 1. To perform cell work, cells require energy. a. A cell does three main kinds of work: i. Mechanical work, such as the beating of cilia, contraction of muscle cells, and movement
Cellular Respiration: Harvesting Chemical Energy
 Chapter 9 Cellular Respiration: Harvesting Chemical Energy You should be able to: 1. Explain how redox reactions are involved in energy exchanges. Name and describe the three stages of cellular respiration;
Chapter 9 Cellular Respiration: Harvesting Chemical Energy You should be able to: 1. Explain how redox reactions are involved in energy exchanges. Name and describe the three stages of cellular respiration;
Disaccharides. Compound dehydration synthesis puts sugars together Hydrolysis (hydro-water, lysisbreakdown)
 Carbohydrate Carbo-hydrate -carbon, water Cn(H2O) n Monosaccharides Hexose hex = 6 [carbons], "-ose" means sugar Glucose monosaccaccharide usually assume a ring structure Disaccharides Compound dehydration
Carbohydrate Carbo-hydrate -carbon, water Cn(H2O) n Monosaccharides Hexose hex = 6 [carbons], "-ose" means sugar Glucose monosaccaccharide usually assume a ring structure Disaccharides Compound dehydration
Chemistry 1120 Exam 2 Study Guide
 Chemistry 1120 Exam 2 Study Guide Chapter 6 6.1 Know amines are derivatives of ammonia, which is not an amine. Classify amines as primary, secondary or tertiary. Master Tutor Section 6.1 Review Section
Chemistry 1120 Exam 2 Study Guide Chapter 6 6.1 Know amines are derivatives of ammonia, which is not an amine. Classify amines as primary, secondary or tertiary. Master Tutor Section 6.1 Review Section
What s the point? The point is to make ATP! ATP
 2006-2007 What s the point? The point is to make ATP! ATP Glycolysis 2 ATP Kreb s cycle 2 ATP Life takes a lot of energy to run, need to extract more energy than 4 ATP! There s got to be a better way!
2006-2007 What s the point? The point is to make ATP! ATP Glycolysis 2 ATP Kreb s cycle 2 ATP Life takes a lot of energy to run, need to extract more energy than 4 ATP! There s got to be a better way!
Transfer of food energy to chemical energy. Includes anabolic and catabolic reactions. The cell is the metabolic processing center
 Metabolism There are a lot of diagrams here. DO NOT, I repeat, DO NOT get overly anxious or excited about them. We will go through them again slowly!! Read the slides, read the book, DO NOT TAKE NOTES.
Metabolism There are a lot of diagrams here. DO NOT, I repeat, DO NOT get overly anxious or excited about them. We will go through them again slowly!! Read the slides, read the book, DO NOT TAKE NOTES.
NBCE Mock Board Questions Biochemistry
 1. Fluid mosaic describes. A. Tertiary structure of proteins B. Ribosomal subunits C. DNA structure D. Plasma membrane structure NBCE Mock Board Questions Biochemistry 2. Where in the cell does beta oxidation
1. Fluid mosaic describes. A. Tertiary structure of proteins B. Ribosomal subunits C. DNA structure D. Plasma membrane structure NBCE Mock Board Questions Biochemistry 2. Where in the cell does beta oxidation
number Done by Corrected by Doctor Nayef Karadsheh
 number 11 Done by حسام أبو عوض Corrected by Moayyad Al-Shafei Doctor Nayef Karadsheh 1 P a g e General Regulatory Aspects in Metabolism: We can divide all pathways in metabolism to catabolicand anabolic.
number 11 Done by حسام أبو عوض Corrected by Moayyad Al-Shafei Doctor Nayef Karadsheh 1 P a g e General Regulatory Aspects in Metabolism: We can divide all pathways in metabolism to catabolicand anabolic.
III. Metabolism - Gluconeogenesis
 Department of Chemistry and Biochemistry University of Lethbridge III. Metabolism - Gluconeogenesis Carl & Gertrude Cori Slide 1 Carbohydrate Synthesis Lactate, pyruvate and glycerol are the important
Department of Chemistry and Biochemistry University of Lethbridge III. Metabolism - Gluconeogenesis Carl & Gertrude Cori Slide 1 Carbohydrate Synthesis Lactate, pyruvate and glycerol are the important
24.1 Introduction to Carbohydrates
 24.1 Introduction to Carbohydrates Carbohydrates (sugars) are abundant in nature: They are high energy biomolecules. They provide structural rigidity for organisms (plants, crustaceans, etc.). The polymer
24.1 Introduction to Carbohydrates Carbohydrates (sugars) are abundant in nature: They are high energy biomolecules. They provide structural rigidity for organisms (plants, crustaceans, etc.). The polymer
CHY2026: General Biochemistry UNIT 7& 8: CARBOHYDRATE METABOLISM
 CHY2026: General Biochemistry UNIT 7& 8: CARBOHYDRATE METABOLISM Metabolism Bioenergetics is the transfer and utilization of energy in biological systems The direction and extent to which a chemical reaction
CHY2026: General Biochemistry UNIT 7& 8: CARBOHYDRATE METABOLISM Metabolism Bioenergetics is the transfer and utilization of energy in biological systems The direction and extent to which a chemical reaction
Number of Carbohydrate Units
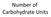 Number of Carbohydrate Units Monosaccharides = single unit Disaccharides = two units Oligiosaccharide = 3 10 units Polysaccharide = 11+ units Bonus: Can you name the most common Mono (4), Di(3), and Poly(4)
Number of Carbohydrate Units Monosaccharides = single unit Disaccharides = two units Oligiosaccharide = 3 10 units Polysaccharide = 11+ units Bonus: Can you name the most common Mono (4), Di(3), and Poly(4)
Cellular Pathways That Harvest Chemical Energy. Cellular Pathways That Harvest Chemical Energy. Cellular Pathways In General
 Cellular Pathways That Harvest Chemical Energy A. Obtaining Energy and Electrons from Glucose Lecture Series 12 Cellular Pathways That Harvest Chemical Energy B. An Overview: Releasing Energy from Glucose
Cellular Pathways That Harvest Chemical Energy A. Obtaining Energy and Electrons from Glucose Lecture Series 12 Cellular Pathways That Harvest Chemical Energy B. An Overview: Releasing Energy from Glucose
III. 6. Test. Respiració cel lular
 III. 6. Test. Respiració cel lular Chapter Questions 1) What is the term for metabolic pathways that release stored energy by breaking down complex molecules? A) anabolic pathways B) catabolic pathways
III. 6. Test. Respiració cel lular Chapter Questions 1) What is the term for metabolic pathways that release stored energy by breaking down complex molecules? A) anabolic pathways B) catabolic pathways
Fundamentals of Organic Chemistry. CHAPTER 6: Carbohydrates
 Fundamentals of Organic Chemistry CHEM 109 For Students of Health Colleges Credit hrs.: (2+1) King Saud University College of Science, Chemistry Department CHEM 109 CHAPTER 6: Carbohydrates Carbohydrates
Fundamentals of Organic Chemistry CHEM 109 For Students of Health Colleges Credit hrs.: (2+1) King Saud University College of Science, Chemistry Department CHEM 109 CHAPTER 6: Carbohydrates Carbohydrates
Integration of Metabolism
 Integration of Metabolism Metabolism is a continuous process. Thousands of reactions occur simultaneously in order to maintain homeostasis. It ensures a supply of fuel, to tissues at all times, in fed
Integration of Metabolism Metabolism is a continuous process. Thousands of reactions occur simultaneously in order to maintain homeostasis. It ensures a supply of fuel, to tissues at all times, in fed
CARBOHYDRATES. By: SHAMSUL AZAHARI ZAINAL BADARI Department of Resource Management And Consumer Studies Faculty of Human Ecology UPM
 CARBOHYDRATES By: SHAMSUL AZAHARI ZAINAL BADARI Department of Resource Management And Consumer Studies Faculty of Human Ecology UPM OBJECTIVES OF THE LECTURE By the end of this lecture, student can: Define
CARBOHYDRATES By: SHAMSUL AZAHARI ZAINAL BADARI Department of Resource Management And Consumer Studies Faculty of Human Ecology UPM OBJECTIVES OF THE LECTURE By the end of this lecture, student can: Define
Carbohydrates Dr. Ameerah M. Zarzoor
 Carbohydrates Dr. Ameerah M. Zarzoor What Are Carbohydrates? Carbohydrates are the most abundant biomolecules on Earth Produced by plants during photosynthesis Carbohydrates are polyhydroxyl aldehydes
Carbohydrates Dr. Ameerah M. Zarzoor What Are Carbohydrates? Carbohydrates are the most abundant biomolecules on Earth Produced by plants during photosynthesis Carbohydrates are polyhydroxyl aldehydes
Yield of energy from glucose
 Paper : Module : 05 Yield of Energy from Glucose Principal Investigator, Paper Coordinator and Content Writer Prof. Ramesh Kothari, Professor Dept. of Biosciences, Saurashtra University, Rajkot - 360005
Paper : Module : 05 Yield of Energy from Glucose Principal Investigator, Paper Coordinator and Content Writer Prof. Ramesh Kothari, Professor Dept. of Biosciences, Saurashtra University, Rajkot - 360005
Chemistry 107 Exam 3 Study Guide
 Chapter 7 Chemistry 107 Exam 3 Study Guide 7.1 Recognize the aldehyde, ketone and hydroxyl (-OH) functional groups found in carbohydrates. Differentiate between mono-, di-, and polysaccharides. Master
Chapter 7 Chemistry 107 Exam 3 Study Guide 7.1 Recognize the aldehyde, ketone and hydroxyl (-OH) functional groups found in carbohydrates. Differentiate between mono-, di-, and polysaccharides. Master
This is an example outline of 3 lectures in BSC (Thanks to Dr. Ellington for sharing this information.)
 This is an example outline of 3 lectures in BSC 2010. (Thanks to Dr. Ellington for sharing this information.) Topic 10: CELLULAR RESPIRATION (lectures 14-16) OBJECTIVES: 1. Know the basic reactions that
This is an example outline of 3 lectures in BSC 2010. (Thanks to Dr. Ellington for sharing this information.) Topic 10: CELLULAR RESPIRATION (lectures 14-16) OBJECTIVES: 1. Know the basic reactions that
Metabolism. Chapter 5. Catabolism Drives Anabolism 8/29/11. Complete Catabolism of Glucose
 8/29/11 Metabolism Chapter 5 All of the reactions in the body that require energy transfer. Can be divided into: Cell Respiration and Metabolism Anabolism: requires the input of energy to synthesize large
8/29/11 Metabolism Chapter 5 All of the reactions in the body that require energy transfer. Can be divided into: Cell Respiration and Metabolism Anabolism: requires the input of energy to synthesize large
Carbohydrate Metabolism
 Chapter 34 Carbohydrate Metabolism Carbohydrate metabolism is important for both plants and animals. Introduction to General, Organic, and Biochemistry, 10e John Wiley & Sons, Inc Morris Hein, Scott Pattison,
Chapter 34 Carbohydrate Metabolism Carbohydrate metabolism is important for both plants and animals. Introduction to General, Organic, and Biochemistry, 10e John Wiley & Sons, Inc Morris Hein, Scott Pattison,
number Done by Corrected by Doctor Nayef Karadsheh
 number 13 Done by Asma Karameh Corrected by Saad hayek Doctor Nayef Karadsheh Gluconeogenesis This lecture covers gluconeogenesis with aspects of: 1) Introduction to glucose distribution through tissues.
number 13 Done by Asma Karameh Corrected by Saad hayek Doctor Nayef Karadsheh Gluconeogenesis This lecture covers gluconeogenesis with aspects of: 1) Introduction to glucose distribution through tissues.
1st half of glycolysis (5 reactions) Glucose priming get glucose ready to split phosphorylate glucose rearrangement split destabilized glucose
 Warm- Up Objective: Describe the role of in coupling the cell's anabolic and catabolic processes. Warm-up: What cellular processes produces the carbon dioxide that you exhale? 1st half of glycolysis (5
Warm- Up Objective: Describe the role of in coupling the cell's anabolic and catabolic processes. Warm-up: What cellular processes produces the carbon dioxide that you exhale? 1st half of glycolysis (5
I tried to put as many questions as possible, but unfortunately only answers were found without the questions.
 I tried to put as many questions as possible, but unfortunately only answers were found without the questions. These are some questions from doctor2015 med exam : 1. One of them isn t acute phase protein
I tried to put as many questions as possible, but unfortunately only answers were found without the questions. These are some questions from doctor2015 med exam : 1. One of them isn t acute phase protein
I tried to put as many questions as possible, but unfortunately only answers were found without the questions.
 I tried to put as many questions as possible, but unfortunately only answers were found without the questions. These are some questions from doctor2015 med exam : 1. One of them isn t acute phase protein
I tried to put as many questions as possible, but unfortunately only answers were found without the questions. These are some questions from doctor2015 med exam : 1. One of them isn t acute phase protein
Chapter 16: Carbohydrates
 Vocabulary Aldose: a sugar that contains an aldehyde group as part of its structure Amylopectin: a form of starch; a branched chain polymer of glucose Amylose: a form of starch; a linear polymer of glucose
Vocabulary Aldose: a sugar that contains an aldehyde group as part of its structure Amylopectin: a form of starch; a branched chain polymer of glucose Amylose: a form of starch; a linear polymer of glucose
Biol 219 Lec 7 Fall 2016
 Cellular Respiration: Harvesting Energy to form ATP Cellular Respiration and Metabolism Glucose ATP Pyruvate Lactate Acetyl CoA NAD + Introducing The Players primary substrate for cellular respiration
Cellular Respiration: Harvesting Energy to form ATP Cellular Respiration and Metabolism Glucose ATP Pyruvate Lactate Acetyl CoA NAD + Introducing The Players primary substrate for cellular respiration
Cellular Respiration Harvesting Chemical Energy ATP
 Cellular Respiration Harvesting Chemical Energy ATP 2006-2007 What s the point? The point is to make ATP! ATP 2006-2007 Harvesting stored energy Energy is stored in organic molecules carbohydrates, fats,
Cellular Respiration Harvesting Chemical Energy ATP 2006-2007 What s the point? The point is to make ATP! ATP 2006-2007 Harvesting stored energy Energy is stored in organic molecules carbohydrates, fats,
CHAPTER 27 CARBOHYDRATES SOLUTIONS TO REVIEW QUESTIONS
 27 09/17/2013 11:12:35 Page 397 APTER 27 ARBYDRATES SLUTINS T REVIEW QUESTINS 1. In general, the carbohydrate carbon oxidation state determines the carbon s metabolic energy content. The more oxidized
27 09/17/2013 11:12:35 Page 397 APTER 27 ARBYDRATES SLUTINS T REVIEW QUESTINS 1. In general, the carbohydrate carbon oxidation state determines the carbon s metabolic energy content. The more oxidized
BCH 4053 Spring 2001 Chapter 7 Lecture Notes
 BC 4053 Spring 2001 Chapter 7 Lecture Notes 1 Chapter 7 Carbohydrates 2 Carbohydrates: Nomenclature ydrates of carbon General formula (C 2 ) n (simple sugars) or C x ( 2 0) y Monosaccharides (simple sugars)
BC 4053 Spring 2001 Chapter 7 Lecture Notes 1 Chapter 7 Carbohydrates 2 Carbohydrates: Nomenclature ydrates of carbon General formula (C 2 ) n (simple sugars) or C x ( 2 0) y Monosaccharides (simple sugars)
Glycolysis Part 2. BCH 340 lecture 4
 Glycolysis Part 2 BCH 340 lecture 4 Regulation of Glycolysis There are three steps in glycolysis that have enzymes which regulate the flux of glycolysis These enzymes catalyzes irreversible reactions of
Glycolysis Part 2 BCH 340 lecture 4 Regulation of Glycolysis There are three steps in glycolysis that have enzymes which regulate the flux of glycolysis These enzymes catalyzes irreversible reactions of
Biochemistry 7/11/ Bio-Energetics & ATP. 5.1) ADP, ATP and Cellular Respiration OVERVIEW OF ENERGY AND METABOLISM
 Biochemistry 5. Bio-Energetics & ATP 5.1) ADP, ATP and Cellular Respiration Prof. Dr. Klaus Heese OVERVIEW OF ENERGY AND METABOLISM 1. The food we eat, (carbohydrates/ glucose /sugar, lipids/fat, proteins),
Biochemistry 5. Bio-Energetics & ATP 5.1) ADP, ATP and Cellular Respiration Prof. Dr. Klaus Heese OVERVIEW OF ENERGY AND METABOLISM 1. The food we eat, (carbohydrates/ glucose /sugar, lipids/fat, proteins),
How Cells Harvest Chemical Energy
 How Cells Harvest Chemical Energy Global Athlete Outreach Program US CytoThesis Systems Medicine Center www.cytothesis.us US OncoTherapy Systems BioMedicine Group CytoThesis Bioengineering Research Group
How Cells Harvest Chemical Energy Global Athlete Outreach Program US CytoThesis Systems Medicine Center www.cytothesis.us US OncoTherapy Systems BioMedicine Group CytoThesis Bioengineering Research Group
4. Which step shows a split of one molecule into two smaller molecules? a. 2. d. 5
 1. Which of the following statements about NAD + is false? a. NAD + is reduced to NADH during both glycolysis and the citric acid cycle. b. NAD + has more chemical energy than NADH. c. NAD + is reduced
1. Which of the following statements about NAD + is false? a. NAD + is reduced to NADH during both glycolysis and the citric acid cycle. b. NAD + has more chemical energy than NADH. c. NAD + is reduced
Chapter 13 Carbohydrate Metabolism
 Chapter 13 Carbohydrate Metabolism Chapter bjectives: Learn about Blood glucose. Learn about the glycolysis reaction pathways and the regulation of glycolysis. Learn about the fates of pyruvate under various
Chapter 13 Carbohydrate Metabolism Chapter bjectives: Learn about Blood glucose. Learn about the glycolysis reaction pathways and the regulation of glycolysis. Learn about the fates of pyruvate under various
Chapter 9. Cellular Respiration and Fermentation
 Chapter 9 Cellular Respiration and Fermentation Energy flows into an ecosystem as sunlight and leaves as heat Photosynthesis generates O 2 and organic molecules, which are used in cellular respiration
Chapter 9 Cellular Respiration and Fermentation Energy flows into an ecosystem as sunlight and leaves as heat Photosynthesis generates O 2 and organic molecules, which are used in cellular respiration
Integration Of Metabolism
 Integration Of Metabolism Metabolism Consist of Highly Interconnected Pathways The basic strategy of catabolic metabolism is to form ATP, NADPH, and building blocks for biosyntheses. 1. ATP is the universal
Integration Of Metabolism Metabolism Consist of Highly Interconnected Pathways The basic strategy of catabolic metabolism is to form ATP, NADPH, and building blocks for biosyntheses. 1. ATP is the universal
SIMPLE BASIC METABOLISM
 SIMPLE BASIC METABOLISM When we eat food such as a tuna fish sandwich, the polysaccharides, lipids, and proteins are digested to smaller molecules that are absorbed into the cells of our body. As these
SIMPLE BASIC METABOLISM When we eat food such as a tuna fish sandwich, the polysaccharides, lipids, and proteins are digested to smaller molecules that are absorbed into the cells of our body. As these
Lecture 2 Carbohydrates
 Lecture 2 Carbohydrates Sources of CHOs Wholegrains major dietary intake Vegetables, legumes ad fruit contain dietary fibre Milk products provide lactose essential for infants Glycogen is a storage carbohydrate,
Lecture 2 Carbohydrates Sources of CHOs Wholegrains major dietary intake Vegetables, legumes ad fruit contain dietary fibre Milk products provide lactose essential for infants Glycogen is a storage carbohydrate,
III. Metabolism Glucose Catabolism Part II
 Department of Chemistry and Biochemistry University of Lethbridge III. Metabolism Glucose Catabolism Part II Slide 1 Metabolic Fates of NADH and Pyruvate Cartoon: Fate of pyruvate, the product of glycolysis.
Department of Chemistry and Biochemistry University of Lethbridge III. Metabolism Glucose Catabolism Part II Slide 1 Metabolic Fates of NADH and Pyruvate Cartoon: Fate of pyruvate, the product of glycolysis.
Chapter 7 How Cells Release Chemical Energy
 Chapter 7 How Cells Release Chemical Energy 7.1 Mighty Mitochondria More than forty disorders related to defective mitochondria are known (such as Friedreich s ataxia); many of those afflicted die young
Chapter 7 How Cells Release Chemical Energy 7.1 Mighty Mitochondria More than forty disorders related to defective mitochondria are known (such as Friedreich s ataxia); many of those afflicted die young
ATP. Principles of Energy Harvest. Chapter 9~ The point is to make ATP! Cellular Respiration: Harvesting Chemical Energy. What s the point?
 Chapter 9~ Cellular Respiration: Harvesting Chemical Energy What s the point? The point is to make! 2006-2007 Principles of Energy Harvest Catabolic pathway Fermentation Cellular Respiration C6H126 + 62
Chapter 9~ Cellular Respiration: Harvesting Chemical Energy What s the point? The point is to make! 2006-2007 Principles of Energy Harvest Catabolic pathway Fermentation Cellular Respiration C6H126 + 62
METABOLISM Sri Widia A Jusman Department of Biochemistry & Molecular Biology FMUI
 METABOLISM Sri Widia A Jusman Department of Biochemistry & Molecular Biology FMUI 1 METABOLISM Process of how cells acquire, transform, store and use energy Study of the chemistry, regulation and energetics
METABOLISM Sri Widia A Jusman Department of Biochemistry & Molecular Biology FMUI 1 METABOLISM Process of how cells acquire, transform, store and use energy Study of the chemistry, regulation and energetics
Questions- Carbohydrates. A. The following structure is D-sorbose. (Questions 1 7) CH 2 OH C = O H C OH HO C H H C OH
 Questions- Carbohydrates A. The following structure is D-sorbose. (Questions 1 7) CH 2 C = O H C HO C H H C CH 2 1. 2. 3. 4. 5. Which characteristic is different when comparing the open-chain forms of
Questions- Carbohydrates A. The following structure is D-sorbose. (Questions 1 7) CH 2 C = O H C HO C H H C CH 2 1. 2. 3. 4. 5. Which characteristic is different when comparing the open-chain forms of
Cellular Respiration and Fermentation
 CAMPBELL BIOLOGY IN FOCUS URRY CAIN WASSERMAN MINORSKY REECE 7 Cellular Respiration and Fermentation Lecture Presentations by Kathleen Fitzpatrick and Nicole Tunbridge, Simon Fraser University SECOND EDITION
CAMPBELL BIOLOGY IN FOCUS URRY CAIN WASSERMAN MINORSKY REECE 7 Cellular Respiration and Fermentation Lecture Presentations by Kathleen Fitzpatrick and Nicole Tunbridge, Simon Fraser University SECOND EDITION
Cellular Respiration and Fermentation
 CAMPBELL BIOLOGY IN FOCUS URRY CAIN WASSERMAN MINORSKY REECE 7 Cellular Respiration and Fermentation Lecture Presentations by Kathleen Fitzpatrick and Nicole Tunbridge, Simon Fraser University SECOND EDITION
CAMPBELL BIOLOGY IN FOCUS URRY CAIN WASSERMAN MINORSKY REECE 7 Cellular Respiration and Fermentation Lecture Presentations by Kathleen Fitzpatrick and Nicole Tunbridge, Simon Fraser University SECOND EDITION
How Did Energy-Releasing Pathways Evolve? (cont d.)
 How Did Energy-Releasing Pathways Evolve? (cont d.) 7.1 How Do Cells Access the Chemical Energy in Sugars? In order to use the energy stored in sugars, cells must first transfer it to ATP The energy transfer
How Did Energy-Releasing Pathways Evolve? (cont d.) 7.1 How Do Cells Access the Chemical Energy in Sugars? In order to use the energy stored in sugars, cells must first transfer it to ATP The energy transfer
OVERVIEW OF ENERGY AND METABOLISM
 Biochemistry 5. Bio-Energetics & ATP 5.1) ADP, ATP and Cellular Respiration OVERVIEW OF ENERGY AND METABOLISM 1. The food we eat, (carbohydrates/ glucose /sugar, lipids/fat, proteins), are our only source
Biochemistry 5. Bio-Energetics & ATP 5.1) ADP, ATP and Cellular Respiration OVERVIEW OF ENERGY AND METABOLISM 1. The food we eat, (carbohydrates/ glucose /sugar, lipids/fat, proteins), are our only source
Chapter 11. Learning objectives: Structure and function of monosaccharides, polysaccharide, glycoproteins lectins.
 Chapter 11 Learning objectives: Structure and function of monosaccharides, polysaccharide, glycoproteins lectins. Carbohydrates Fuels Structural components Coating of cells Part of extracellular matrix
Chapter 11 Learning objectives: Structure and function of monosaccharides, polysaccharide, glycoproteins lectins. Carbohydrates Fuels Structural components Coating of cells Part of extracellular matrix
7 Cellular Respiration and Fermentation
 CAMPBELL BIOLOGY IN FOCUS Urry Cain Wasserman Minorsky Jackson Reece 7 Cellular Respiration and Fermentation Lecture Presentations by Kathleen Fitzpatrick and Nicole Tunbridge Overview: Life Is Work Living
CAMPBELL BIOLOGY IN FOCUS Urry Cain Wasserman Minorsky Jackson Reece 7 Cellular Respiration and Fermentation Lecture Presentations by Kathleen Fitzpatrick and Nicole Tunbridge Overview: Life Is Work Living
Introduction to Macromolecules. If you were to look at the nutrition label of whole milk, what main items stick out?
 Introduction to Macromolecules Macromolecules are a set of molecules that are found in living organisms. Macromolecules essentially mean big molecules as the word macro means large. The functions of these
Introduction to Macromolecules Macromolecules are a set of molecules that are found in living organisms. Macromolecules essentially mean big molecules as the word macro means large. The functions of these
Ch. 9 Cell Respiration. Title: Oct 15 3:24 PM (1 of 53)
 Ch. 9 Cell Respiration Title: Oct 15 3:24 PM (1 of 53) Essential question: How do cells use stored chemical energy in organic molecules and to generate ATP? Title: Oct 15 3:28 PM (2 of 53) Title: Oct 19
Ch. 9 Cell Respiration Title: Oct 15 3:24 PM (1 of 53) Essential question: How do cells use stored chemical energy in organic molecules and to generate ATP? Title: Oct 15 3:28 PM (2 of 53) Title: Oct 19
How Cells Release Chemical Energy. Chapter 8
 How Cells Release Chemical Energy Chapter 8 Impacts, Issues: When Mitochondria Spin Their Wheels More than forty disorders related to defective mitochondria are known (such as Friedreich s ataxia); many
How Cells Release Chemical Energy Chapter 8 Impacts, Issues: When Mitochondria Spin Their Wheels More than forty disorders related to defective mitochondria are known (such as Friedreich s ataxia); many
Chapter 22 Carbohydrates
 Chapter 22 Carbohydrates Introduction Classification of Carbohydrates Carbohydrates have the general formula C x (H 2 O) y Carbohydrates are defined as polyhydroxy aldehydes or ketones or substances that
Chapter 22 Carbohydrates Introduction Classification of Carbohydrates Carbohydrates have the general formula C x (H 2 O) y Carbohydrates are defined as polyhydroxy aldehydes or ketones or substances that
Carbohydrates. Learning Objective
 , one of the four major classes of biomolecules, are aldehyde or ketone compounds with multiple hydroxyl groups. They function as energy stores, metabolic intermediates and important fuels for the body.
, one of the four major classes of biomolecules, are aldehyde or ketone compounds with multiple hydroxyl groups. They function as energy stores, metabolic intermediates and important fuels for the body.
CH 7: Cell Respiration and Fermentation Overview. Concept 7.1: Catabolic pathways yield energy by oxidizing organic fuels
 CH 7: Cell Respiration and Fermentation Overview Living cells require energy from outside sources Some animals obtain energy by eating plants, and some animals feed on other organisms Energy flows into
CH 7: Cell Respiration and Fermentation Overview Living cells require energy from outside sources Some animals obtain energy by eating plants, and some animals feed on other organisms Energy flows into
Chapter 9: Cellular Respiration Overview: Life Is Work. Living cells. Require transfusions of energy from outside sources to perform their many tasks
 Chapter 9: Cellular Respiration Overview: Life Is Work Living cells Require transfusions of energy from outside sources to perform their many tasks Biology, 7 th Edition Neil Campbell and Jane Reece The
Chapter 9: Cellular Respiration Overview: Life Is Work Living cells Require transfusions of energy from outside sources to perform their many tasks Biology, 7 th Edition Neil Campbell and Jane Reece The
CLASS 11th. Biomolecules
 CLASS 11th 01. Carbohydrates These are the compound of carbon, hydrogen and oxygen having hydrogen and oxygen in the same ratio as that of water, i.e. 2 : 1. They are among the most widely distributed
CLASS 11th 01. Carbohydrates These are the compound of carbon, hydrogen and oxygen having hydrogen and oxygen in the same ratio as that of water, i.e. 2 : 1. They are among the most widely distributed
Photosynthesis in chloroplasts. Cellular respiration in mitochondria ATP. ATP powers most cellular work
 Light energy ECOSYSTEM CO + H O Photosynthesis in chloroplasts Cellular respiration in mitochondria Organic molecules + O powers most cellular work Heat energy 1 becomes oxidized (loses electron) becomes
Light energy ECOSYSTEM CO + H O Photosynthesis in chloroplasts Cellular respiration in mitochondria Organic molecules + O powers most cellular work Heat energy 1 becomes oxidized (loses electron) becomes
2/25/2015. Chapter 6. Carbohydrates. Outline. 6.1 Classes of Carbohydrates. 6.1 Classes of Carbohydrates. 6.1 Classes of Carbohydrates
 Lecture Presentation Chapter 6 Carbohydrates Julie Klare Fortis College Smyrna, GA Outline 6.7 Carbohydrates and Blood The simplest carbohydrates are monosaccharides (mono is Greek for one, sakkhari is
Lecture Presentation Chapter 6 Carbohydrates Julie Klare Fortis College Smyrna, GA Outline 6.7 Carbohydrates and Blood The simplest carbohydrates are monosaccharides (mono is Greek for one, sakkhari is
Physiological Chemistry II Exam IV Dr. Melissa Kelley April 13, 2004
 Name Write your name on the back of the exam Physiological Chemistry II Exam IV Dr. Melissa Kelley April 13, 2004 This examination consists of forty-four questions, each having 2 points. The remaining
Name Write your name on the back of the exam Physiological Chemistry II Exam IV Dr. Melissa Kelley April 13, 2004 This examination consists of forty-four questions, each having 2 points. The remaining
