Departments of Molecular Endocrinology, Metabolic Disorders, and Comparative Medicine, Merck Research Laboratories, Rahway, New Jersey, USA 2
|
|
|
- Joan Phelps
- 5 years ago
- Views:
Transcription
1 Role of AMP-activated protein kinase in mechanism of metformin action Gaochao Zhou, 1 Robert Myers, 1 Ying Li, 1 Yuli Chen, 1 Xiaolan Shen, 1 Judy Fenyk-Melody, 1 Margaret Wu, 1 John Ventre, 1 Thomas Doebber, 1 Nobuharu Fujii, 2 Nicolas Musi, 2 Michael F. Hirshman, 2 Laurie J. Goodyear, 2 and David E. Moller 1 Rapid Publication See related Commentary on pages Departments of Molecular Endocrinology, Metabolic Disorders, and Comparative Medicine, Merck Research Laboratories, Rahway, New Jersey, USA 2 Joslin Diabetes Center and Harvard Medical School, Boston, Massachusetts, USA Address correspondence to: Gaochao Zhou, Merck Research Laboratories, Rahway, New Jersey 07065, USA. Phone: (732) ; Fax: (732) ; gaochao_zhou@merck.com. Received for publication June 13, 2001, and accepted in revised form August 28, Metformin is a widely used drug for treatment of type 2 diabetes with no defined cellular mechanism of action. Its glucose-lowering effect results from decreased hepatic glucose production and increased glucose utilization. Metformin s beneficial effects on circulating lipids have been linked to reduced fatty liver. AMP-activated protein kinase (AMPK) is a major cellular regulator of lipid and glucose metabolism. Here we report that metformin activates AMPK in hepatocytes; as a result, acetyl-coa carboxylase (ACC) activity is reduced, fatty acid oxidation is induced, and expression of lipogenic enzymes is suppressed. Activation of AMPK by metformin or an adenosine analogue suppresses expression of SREBP-1, a key lipogenic transcription factor. In metformin-treated rats, hepatic expression of SREBP-1 (and other lipogenic) mrnas and protein is reduced; activity of the AMPK target, ACC, is also reduced. Using a novel AMPK inhibitor, we find that AMPK activation is required for metformin s inhibitory effect on glucose production by hepatocytes. In isolated rat skeletal muscles, metformin stimulates glucose uptake coincident with AMPK activation. Activation of AMPK provides a unified explanation for the pleiotropic beneficial effects of this drug; these results also suggest that alternative means of modulating AMPK should be useful for the treatment of metabolic disorders. This article was published online in advance of the print edition. The date of publication is available from the JCI website, J. Clin. Invest. 108: (2001). DOI: /JCI Introduction Metformin is widely used for the therapy of type 2 diabetes mellitus (DM2) (1). Metformin ameliorates hyperglycemia without stimulating insulin secretion, promoting weight gain, or causing hypoglycemia (2, 3). In addition, metformin has beneficial effects on circulating lipids linked to increased cardiovascular risk (2 4). Although used as a drug since 1957, the mechanism(s) by which metformin lowers glucose and lipids remains an enigma. Two effects, decreased hepatic glucose production (2, 5, 6) and increased skeletal myocyte glucose uptake (7, 8), have been implicated as major contributors to glucose-lowering efficacy. Metformin also decreases hepatic lipids in obese mice (9). Metformin is, however, a low-potency compound that is used at high doses, resulting in only modest net efficacy; in addition, significant side effects can occur (2). Thus, an understanding of the molecular basis for metformin s effects on glucose and lipid homeostasis is a critical focus of research directed toward improved therapeutic approaches to DM2 and related disorders. AMP-activated protein kinase (AMPK) provides a candidate target capable of mediating the beneficial metabolic effects of metformin. AMPK is a multisubunit enzyme that is recognized as a major regulator of lipid biosynthetic pathways due to its role in the phosphorylation and inactivation of key enzymes such as acetyl-coa carboxylase (ACC) (10). More recent data strongly suggest that AMPK has a wider role in metabolic regulation (10, 11): this includes fatty acid oxidation, muscle glucose uptake (12 14), expression of camp-stimulated gluconeogenic genes such as PEPCK and G6Pase (15), and glucose-stimulated genes associated with hepatic lipogenesis, including fatty acid synthase (FAS), Spot-14 (S14), and L-type pyruvate kinase (16). Chronic activation of AMPK may also induce the expression of muscle hexokinase and glucose transporters (Glut4), mimicking the effects of extensive exercise training (17). Thus, it has been predicted that AMPK activation would be a good approach to treat DM2 (11). In this report we tested the hypothesis that activation of AMPK mediates the beneficial metabolic effects of metformin. Methods Measurements of AMPK, ACC, and fatty acid oxidation in primary hepatocytes. Hepatocytes were isolated from male Sprague Dawley (SD) rats by collagenase digestion (18). For the AMPK assay, cells were seeded in six-well plates at cells/well in DMEM containing 100 U/ml penicillin, 100 µg/ml streptomycin, 10% FBS, 100 nm insulin, 100 nm dexamethasone, and 5 µg/ml transferrin for 4 hours. Cells were then cultured in serum-free DMEM for 16 hours followed by treatment for 1 hour or 7 hours with control medium, 5-amino-imidazole carboxamide ribo- The Journal of Clinical Investigation October 2001 Volume 108 Number
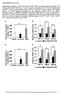
2 side (AICAR), or metformin at concentrations indicated. For a 39-hour treatment, cells for both control and metformin (10 or 20 µm) groups were cultured in DMEM plus 5% FBS and 100 nm insulin, and the fresh control and metformin-containing medium were replaced every 12 hours (last medium change was 3 hours before harvest). After treatment, the cells were directly lysed in digitonin-containing and phosphatase inhibitor containing buffer A (19), followed by precipitation with ammonium sulfate at 35% saturation. AMPK activity was determined by measurement of phosphorylation of a synthetic peptide substrate, SAMS (HMR- SAMSGLHLVKRR) (20). For ACC assay, the 35% ammonium sulfate precipitate from digitonin-lysed hepatocytes (4 µg each) was used for determination of ACC activity via 14 CO 2 fixation in the presence of 20 mm citrate as done previously (19). For fatty acid oxidation, the oxidation of 14 C-oleate to acid-soluble products was performed as done previously (21), but in medium M199 in the absence of albumin. AMPK partial purification and in vitro kinase assay. Liver AMPK was partially purified from male SD rats (22) to the blue-sepharose step. The 100-µl reaction mixture contained 100 µm AMP, 100 µm ATP (0.5 µci 33 P-ATP per reaction), and 50 µm SAMS in a buffer (40 mm HEPES, ph 7.0, 80 mm NaCl, 0.8 mm EDTA, 5 mm MgCl 2, 0.025% BSA, and 0.8 mm DTT). The reaction was initiated with addition of the enzyme. After 30-minute incubation at 30 C, the reaction was stopped by addition of 80 µl 1% H 3 PO 4. Aliquots (100 µl) were transferred to 96-well MultiScreen plates (MAPHNOB50; Millipore Corp., Bedford, Massachusetts, USA). The plate was washed three times with 1% H 3 PO 4 followed by detection in a Topcount. The in vitro AMPK inhibition data obtained with compound C (6- [4-(2-Piperidin-1-yl-ethoxy)-phenyl)]-3- pyridin-4-yl-pyyrazolo[1,5-a] pyrimi- Figure 1 Metformin mediates AMPK activation in primary hepatocytes. (a) Metformin (black bars) and AICAR (A; 500 µm) activate AMPK in rat primary hepatocytes. The treatments were 1 hour, 7 hours, and 39 hours, respectively. (b) Metformin (500 µm) and AICAR (500 µm) activated both AMPKα1 and AMPKα2 complexes demonstrated by immunoprecipitation-ampk assay. DPM, disintegrations per minute. (c) Metformin (1 mm) and AICAR (500 µm) stimulated AMPK Thr172 phosphorylation. (d) Metformin does not activate partially purified rat liver AMPK in vitro. (e) Metformin and AICAR (500 µm) inactivate ACC in rat primary hepatocytes. (f) Metformin (500 µm, 4 hours) and AICAR (500 µm, 4 hours) stimulate hepatocyte fatty acid oxidation. C, vehicle control. Mean (n = 3 wells per treatment for 1 hour and 7 hours; for 39-hour treatment, n = wells per treatment) ± SEM values are shown. *P < 0.05, **P < 0.01, ***P < vs. control medium (paired t test) The Journal of Clinical Investigation October 2001 Volume 108 Number 8
3 Figure 2 Treatment of hepatocytes with metformin or AICAR suppresses lipogenic genes in rat primary hepatocytes. Taqman-based real-time RT-PCR was used to quantitate mrnas for FAS, S14, and HMGR. Mean (n = 3 per treatment) ± SEM values are shown. *P < 0.05, **P < 0.01 vs. control medium (paired t test). C, control; A, positive control (AICAR, 500 µm). These experiments were repeated a minimum of three times with similar results. dine were fit to the following equation for competitive inhibition by nonlinear regression using a least-squares Marquardt algorithm in a computer program written by N. Thornberry of Merck Research Laboratories: V i /V o = (K m + S)/[S + K m (1 + I/K i )], where V i is the inhibited velocity, V o is the initial velocity, S is the substrate (ATP) concentration, K m is the Michaelis constant for ATP, I is the inhibitor (compound C) concentration, and K i is the dissociation constant for compound C. Quantitation of mrna. Rat hepatocytes were seeded and starved in M199 medium as described (16). Cells were then treated for 6 hours with the same serum-free and hormone-containing medium containing 25 mm glucose in the presence or absence of AICAR or increasing concentrations of metformin as indicated. Total RNAs were extracted from cultured hepatocytes using the guanidine thiocyanate method (TRIZOL; Life Technologies Inc., Gaithersburg, Maryland, USA). The mrna was quantitated using a TaqMan One Step Gold RT-PCR kit (Applied Biosystems, Branchburg, New Jersey, USA). Primers were as follows: for S14, S14p (6FAM-TGGTGATGATC- CCCAGCCTTCTGAG), S14F (TGTGGT- GCGGAACATGGA), and S14R (CTCCG- GACCCACTCAGCTC); for FAS, FASp (6FAM-TCCGCC AGAGCCCTTTGTTAA- TTGG), FASF (AACTGAACGGCATTACT- CGGTC), and FASR (GTGTCCCATGTTG- GATTTGGT); for sterol regulatory element-binding protein 1 (SREBP1), SREBP1p (6FAM-TCCACCATCGGCAC- CCACTGCT), SREBP1F (AGGACCCAAG- GTGACACCTG), SREBP1R (GCCG- GACGGG-TACATCTTT). Hepatocyte glucose production. Hepatocytes from 24-hour starved rats were incubated at cell/ml in bicarbonate-buffered saline medium containing 10 mm L-lactate, 1 mm pyruvate, 0.3 µm glucagon, with a gas phase of O 2 /CO 2 (19:1). Glucose was measured using glucose oxidase kit from Sigma Chemical Co. (St. Louis, Missouri, USA). Measurements of muscle AMPK activity and glucose uptake. Isolated rat epitrochlearis muscles were incubated for 3 hours with metformin (2 mm) or control medium followed by measurement of AMPKα1 or AMPKα2 activities as described (23). For glucose uptake, insulin (300 nm) was present where indicated for the last 30 minutes of the 3-hour incubation. Then, 3-0-methylglucose uptake was measured using a 10-minute incubation in the absence or presence of metformin and/or insulin as described previously (23). Animal experiments. Oral gavage was used to administer 1 ml of metformin (100 mg/ml) or water alone to male SD rats ( g, n = 7 8). Rats were treated once (see Table 1and Figure 5b) or twice (see Figure 6) a day for 5 days. Rats were starved for 20 hours and then re-fed for 2 hours before the final dose; 4 hours after final dose, the animals were anesthetized and livers rapidly removed by freeze clamping followed by blood withdrawal. RNA was prepared from the freeze-clamped liver by Ultraspec RNA isolation reagent (Biotecx Laboratories Inc., Houston, Texas, USA). Nuclear extracts were prepared from a pool of seven rat livers (24). Glucose levels were determined using the standard glucose oxidase assay kit; β-hydroxybutyrate concentrations were assayed by measuring the reduction of NAD to NADH with a standard assay kit (Sigma Chemical Co.). FFA levels were measured with the assay kit from Boehringer Mannheim GmbH (Mannheim, Germany). Triglyceride levels were assayed with a kit from Roche Diagnostics (Indianapolis, Indiana, USA). Insulin concentrations were measured with the enzyme-immunoassay kit from ALPCO Ltd. (Windham, New Hampshire, USA). All in vivo experiments were approved by the Institutional Animal Care and Use Committee. Immunoblot analysis. For phospho- AMPK (Thr172) detection, the 35% ammonium sulfate precipitate from treated rat hepatocytes was used for Western blot analysis using polyclonal Ab s to phospho AMPKα (Thr172; Cell Signaling Technology, Beverly, Massachusetts, USA). For SREBP-1, 100 µg nuclear extracts from metformin-treated rat livers were similarly analyzed, but using monoclonal anti SREBP-1 Ab (prepared from hybridoma CRL-2121; American Type Culture Collection [ATCC], Rockville, Maryland, USA). The Journal of Clinical Investigation October 2001 Volume 108 Number
4 Figure 3 Discovery and use of a novel small-molecule AMPK inhibitor to establish that the effects of metformin on ACC and glucose production are AMPK dependent. (a) Inhibition of partially purified AMPK by compound C (inset shows the chemical structure) is reversible and competitive with respect to ATP. The in vitro kinase assay was performed in the presence of 5 µm ATP, 0 µm AMP (filled circles), 100 µm ATP, 0 µm AMP (filled squares), and 100 µm ATP plus 100 µm AMP (open circles). (b) Compound C inhibits the effects of AICAR and metformin on ACC in rat hepatocytes. Mean (n = 3 per treatment) ± SEM values are shown. *P < 0.05 vs. control medium (paired t test). (c) Compound C attenuates the ability of metformin to suppress glucagon-stimulated glucose production by hepatocytes from 24-hour starved rats. At time 0, compound C (40 µm) was added, and at time 30 minutes, metformin (2 mm) was added. Squares, control; diamonds, metformin; circles, metformin plus compound C. Each point represents the mean ± SEM of four replicate assays. *P < 0.05, **P < 0.01 vs. metformin alone at the same time point (paired t test). The experiment was repeated three times with similar results. Immunoprecipitation-AMPK assay. Ten micrograms of 35% ammonium sulfate precipitate (containing AMPK) from AICAR- or metformin-treated rat hepatocytes was immunoprecipitated using polyclonal Ab s raised against AMPKα1 (NH 2 -DFYLATSPPDSFLD- DHHLTR-OH) or AMPKα2 (NH 2 - MDDSAMHIPPGLKPH-OH), followed by AMPK assay. Results Metformin promotes AMPK activation in primary rat hepatocytes. Although steadystate plasma levels of metformin in humans are reported to be around 10 µm (25, 26) to as high as 40 µm (27), studies in rats showed that levels in liver are severalfold higher than in plasma (28). Thus, liver levels of greater than 180 µm can be achieved in rats after a 50 mg/kg dose (29); this dose is lower than doses typically required for efficacy in diabetic rats (30, 31). In addition to the fact that high concentrations may be achieved in target tissues, membrane permeability of metformin is a time-dependent and slow process (32). Therefore, we anticipated that high concentrations and/or longer-term experiments might be required in order to see effects of metformin on AMPK in cells or tissues. In primary cultured hepatocytes from both rats and humans, metformin activated AMPK in a concentration- and time-dependent manner (Figure 1a and data not shown). AICAR was used as positive control. AICAR is a cell-permeable adenosine analogue that can be phosphorylated to ZMP, an AMP analogue and known AMPK activator (10). After a 1-hour treatment, 500 µm metformin was required to significantly activate AMPK, whereas after a 7-hour treatment, 50 µm was sufficient to significantly activate the enzyme. Maximal AMPK stimulation, comparable to the effect of 500 µm AICAR, was achieved with metformin concentrations of 2,000 µm (1 hour) or 500 µm (7 hours). To ensure that lower concentrations could mediate a similar effect, rat hepatocytes were incubated with 10 µm or 20 µm metformin for 39 hours. Both 10 µm and 20 µm metformin produced significant AMPK activation (1.3-fold, P = , and 1.6- fold, P = , respectively; Figure 1a). Given the time-dependent effects of metformin, we relied on relatively high concentrations in several shorterterm experiments described below in order to augment the ability to see relevant effects. In Figure 1b, we used an immunoprecipitation-ampk assay to show that metformin as well as AICAR activated AMPK in both AMPKα1 and AMPKα2 complexes efficiently. AMP activates AMPK by promoting its phosphorylation at Thr 172 and by direct activation via an allosteric AMP site. Since enzyme activity shown in Figure 1, a and b, was measured after precipitation from hepatocyte lysates and in the presence of AMP, it is likely that metformin promotes AMPK phosphorylation. In keeping with this notion, we observed that treatment of hepatocytes with either metformin or AICAR resulted in slower electrophoretic mobility of AMPKα1 and AMPKα2, which is consistent with increased Ser/Thr phosphorylation (data not shown). More specifically, both metformin and AICAR treatment induced AMPK Thr172 phosphorylation demonstrated by using anti phospho-ampk Ab (Figure 1c). Recent observations suggest that metformin can impair oxidative phosphorylation by inhibiting mitochondrial phosphorylation complex 1 (32, 33). Although some have reported that very high metformin concentrations (10 mm) can suppress total cellular ATP levels (25, 33), Owen et al. (32) pointed out that more subtle changes in the free ATP/ADP ratio might occur with concentrations of metformin that do not suppress total ATP, but do inhibit gluconeogenesis. We also found that measured total ATP concentrations from metformin-treated 1170 The Journal of Clinical Investigation October 2001 Volume 108 Number 8
5 (500 2,000 µm) rat hepatocytes were not affected (data not shown). Thus, it is still plausible that metformin could produce a subtle decline in the free ATP/ADP and ATP/AMP ratios or a change in local concentrations of nucleotides, serving as a stimulus for AMPK phosphorylation and activation. Alternatively, metformin could directly activate AMPK kinase (AMPKK) or promote AMPK phosphorylation by binding to AMPK and making it a better substrate for AMPKK or worse substrate for phosphatase PP2C (10). The possibility that metformin could be a direct allosteric AMPK activator was excluded by showing that, when incubated with partially purified rat liver AMPK, metformin (from 4 µm to 12 mm), unlike AMP, did not activate the enzyme (Figure 1d). Metformin induces ACC inactivation in primary hepatocytes. As a result of AMPK activation, hepatocyte ACC activity was markedly suppressed by metformin treatment, paralleling concentrations and time points associated with AMPK activation (Figure 1e). ACC catalyzes the biosynthesis of malonyl-coa from acetyl-coa. Malonyl-CoA is an initial substrate for de novo fatty acid biosynthesis, and it serves as a potent inhibitor of carnitine palmitoyl transferase I (CPT-I), a rate-limiting step for fatty acid oxidation (34). Thus, decreased malonyl- CoA concentrations, as a result of ACC inactivation, could lead to decreased lipid synthesis and an increased rate of fatty acid oxidation. Indeed, like AICAR, metformin stimulated fatty acid oxidation in isolated rat hepatocytes (Figure 1f). Metformin and AICAR suppress lipogenic gene expression. AMPK is also involved in gene regulation (10). As noted above, activation of AMPK can suppress the expression of several glucoseactivated lipogenesis-associated genes. Similar to the effect of AICAR, progressive inhibition of glucose-induced FAS and S14 expression was measured in hepatocytes in response to increasing concentrations of metformin (Figure 2). As a control, expression of HMG-CoA reductase (HMGR), a gene not affected by AICAR, was not affected by metformin. Identification and characterization of AMPK inhibitor. To firmly establish the role of AMPK in mediating metformin s effects, a high-throughput in vitro assay was used to identify small-molecule AMPK inhibitors. Screening of a compound library containing more than 10,000 molecules lead to the discovery and characterization of compound C (inset in Figure 3a). Experiments conducted using variable ATP concentrations revealed that compound C is a potent reversible inhibitor that is competitive with ATP, with K i = 109 ± 16 nm in the absence of AMP (Figure 3a). In in vitro assays, compound C did not exhibit significant inhibition of several structurally related kinases including ZAPK, SYK, PKCθ, PKA, and JAK3. Thus, to our knowledge, this is the first description of a potent and selective small-molecule AMPK inhibitor that may be useful as a means to further assess the physiologic effects of this pathway. Incubation of cultured hepatocytes with compound C inhibited ACC inactivation by either AICAR or metformin (Figure 3b). Compound C also attenuated AICAR and metformin s effect to increase fatty acid oxidation or suppress lipogenic genes in hepatocytes (not shown). Metformin-mediated hepatocyte glucose production requires AMPK activation. It is believed that metformin-mediated inhibition of hepatic glucose production (HGP) plays a major role in its glucose-lowering efficacy (2, 3, 6). Here, we determined that both metformin and AICAR were able to inhibit cumulative glucose production in primary cultured rat hepatocytes stimulated with glucagon (Figure 3c and data not shown). Importantly, coincubation of hepatocytes with compound C was able to attenuate the effects of metformin to decrease glucose production in these cells (Figure 3c), demonstrating that AMPK activation was required for metformin s inhibitory effect on hepatocyte glucose production. Metformin activates muscle AMPK and promotes glucose uptake. Since AMPK activation is implicated as a mechanism for stimulation of glucose Figure 4 Metformin (Met; 2 mm, 3 hours) stimulates AMPK activity in skeletal muscle in association with induction of glucose uptake. (a) AMPK activity; (b) glucose uptake. Mean (n = 5 6 isolated rat epitrochlearis muscles per treatment) ± SEM values is shown. The Journal of Clinical Investigation October 2001 Volume 108 Number
6 Figure 5 Metformin and AICAR downregulate hepatic SREBP-1. (a) Metformin (500 µm) and AICAR (500 µm) have similar effects to suppress SREBP-1 mrna expression in rat hepatocytes. Mean ± SEM (n = 3 replicate assays) are shown. Insulin significantly increased SREBP-1 mrna (P = 0.03 vs. control medium by Student s t test). Both AICAR and metformin significantly decreased SREBP-1 mrna with P values of and 0.01, respectively, versus insulin. (b) Metformin prevents re-fed stimulated accumulation of mature SREBP-1 in nuclear extracts from treated rats. Western blot analysis using a monoclonal anti SREBP-1 Ab (prepared from hybridoma CRL-2121) was performed. Similar results were obtained from a separate in vivo experiment. uptake in skeletal muscle (12 14), we assessed the effect of metformin on glucose uptake and AMPK activity in intact rat epitrochlearis muscles. Incubation of isolated muscles with metformin resulted in an increase in the activity of both catalytic subunits of AMPK (Figure 4a). This was coincident with a significant increase in glucose uptake that was also observed to be additive with the effect of insulin stimulation (Figure 4b). In previous studies, AMPKα2 was stimulated to a greater extent than AMPKα1 by AICAR and other stress (23). Further studies will be required to assess this difference. AMPK activation suppresses SREBP-1. SREBP-1c is an important insulinstimulated transcription factor that is implicated in the pathogenesis of insulin resistance, dyslipidemia, and DM2 (35, 36). Target genes that are induced by SREBP-1 include those that encode lipogenic enzymes, such as FAS and S14. Under conditions where SREBP-1 was induced by insulin, incubation of rat hepatocytes with metformin or AICAR strongly suppressed SREBP-1 mrna expression (Figure 5a). Thus, a new pathway by which AMPK can mediate effects on lipogenic gene expression was also identified via these experiments. Assessment of metformin s in vivo effects. To assess whether selected effects of metformin described above also occurred in vivo, SD rats were studied (Table 1). Rats were orally dosed with metformin or vehicle (H 2 O) for 5 days. Rats were starved for 20 hours and then re-fed for 2 hours before the final dose. Four hours after the final dose, tissue and blood samples were obtained for analysis (see Methods). During starvation, there should be very little lipid synthesis. Upon refeeding, hepatic lipid synthesis should be dramatically induced. Metformin s effects were examined under re-fed conditions. Along with modest decreases in plasma insulin and triglycerides, a small, but significant increase in β-hydroxybutyrate was present, suggesting that hepatic fatty acid oxidation was induced in metformin-treated rats. Furthermore, metformin treatment produced significant decreases in hepatic expression of mrnas for SREBP-1, FAS, and S14 that were consistent with effects documented in cells (Table 1). The mature SREBP-1 protein in rat liver nuclear extracts was examined using an anti-srebp1 Ab (Figure 5b). As anticipated (35), SREBP-1 matureform protein was not detected in hepatic nuclear extracts from starved animals. In re-fed animals, mature SREBP-1 protein had accumulated consistent with an increase in lipid synthesis under this condition. Treat- Table 1 In vivo effects of metformin on selected parameters known to be downstream of AMPK activation Serum Vehicle Metformin Glucose (mg/dl) ± ± 7.19 TG (mg/dl) ± ± 3.49 A FFA (mm) 0.17 ± ± 0.04 Insulin (ng/ml) 1.48 ± ± 0.09 A β-hydroxybutyrate (mg/dl) 0.94 ± ± 0.15 A RNA Fold Fold SREBP-1 1 ± ± 0.03 A FAS 1 ± ± 0.02 A S14 1 ± ± 0.03 A Normal SD rats were treated as described in Methods. In rats studied in the fed state, serum samples were analyzed for glucose, triglyceride (TG), FFA, insulin, and β-hydroxybutyrate. Hepatic mrna levels were quantitated. Results are expressed as mean values from seven rats in each group. A P < 0.05 vs. corresponding values from vehicle-treated rats The Journal of Clinical Investigation October 2001 Volume 108 Number 8
7 ment with metformin prevented this accumulation. Additional results obtained using hepatic nuclear extracts from re-fed rats after treatment with AICAR (500 mg/kg/day) also showed that the presence of SREBP-1 mature-form protein was ablated (data not shown). Measurement of AMPK activation in liver ex vivo is difficult because brief hypoxia is known to produce marked activation of the enzyme (ref. 37 and data not shown). Thus we used liver tissue derived from metformin-treated rats to determine that ACC activity was decreased significantly at several tested citrate concentrations (Figure 6). The greatest ACC activity reduction was at a citrate concentration of 1 mm (from 54.6 ± 11.8 to 35.6 ± 7.7 nmol/mg/min; P < 0.01). These results are consistent with metformin having produced in vivo AMPK activation and ACC inactivation. Figure 6 Metformin inhibits hepatic ACC activity in vivo. ACC activity was analyzed in the presence or absence of the indicated citrate concentrations using liver samples from fed rats treated for 5 days with control vehicle (filled squares, n = 7) or treated with 200 mg/kg (twice daily oral gavage) metformin (open squares, n = 8). **P < AMPK mediates a decrease in SREBP-1 mrna and protein expression. Known target genes for SREBP-1, which include FAS and S14, are also downregulated in liver, further contributing to metformin s effects to modulate circulating lipids and to reduce hepatic lipid synthesis and fatty liver. It should be noted that increased SREBP-1 is postulated as a central mediator of insulin resistance in DM2 and related metabolic disorders (35, 36) and that increased liver lipid content is implicated in hepatic insulin resistance (38). Metformin-mediated effects on hepatic glucose production contribute to its glucose-lowering efficacy. We demonstrated (using an AMPK inhibitor, Figure 3) that AMPK activation is required for inhibition of hepatocyte glucose production by metformin under the test conditions we employed. Additional studies will be required to further elucidate precise mechanism(s) by which metformin-stimulated AMPK activation could result in inhibition of hepatic glucose production. AMPK activation is implicated as a mechanism for the induction of skeletal muscle glucose uptake; this effect is also additive with insulin (12). Thus, the observed association of increased glucose uptake and AMPK activation in isolated skeletal muscles suggests that metformin s effect to augment muscle insulin action in vivo may be attributed to AMPK as well. A major mechanism (AMPK activation) by which metformin produces beneficial metabolic effects has been characterized, along with discovery of the mechanism (SREBP-1 suppression), by which AMPK inhibits the expression of lipogenic genes. Attempts to generate novel therapies for metabolic disease via AMPK activation may be worthy of pursuit. Acknowledgments We thank Shiying Chen for characterizing anti-ampk Ab s, Marcie Donnelly for technical support related to animal studies, Mark Fraley for compound synthesis, and Georgianna Harris and Denis McGarry for helpful discussions and intellectual support. Discussion To summarize, results presented here are consistent with a model (Figure 7) in which increased phosphorylation and activation of AMPK by metformin leads to the effects on glucose and lipid metabolism as follows. Phosphorylation and inactivation of ACC, as a result of AMPK activation, serves to inhibit the proximal and rate-limiting step of lipogenesis. Reduced synthesis of the ACC product, malonyl-coa, is also predicted to relieve inhibition of CPT-1, resulting in increased fatty acid oxidation. These effects are likely to contribute to metformin s in vivo ability to lower triglycerides and VLDL. Figure 7 Model for the mechanism by which metformin mediates effects on lipid and glucose metabolism. FA, fatty acid. The Journal of Clinical Investigation October 2001 Volume 108 Number
8 IMS HEALTH s National Prescription Audit Plus TM Stumvoll, M., Nurjhan, N., Perriello, G., Dailey, G. and Gerich, J.E Metabolic effects of metformin in non-insulin-dependent diabetes mellitus. N. Engl. J. Med. 333: Wiernsperger, N.F., and Bailey, C.J The antihyperglycaemic effect of metformin: therapeutic and cellular mechanisms. Drugs. 58: Wu, M.S., et al Effect of metformin on carbohydrate and lipoprotein metabolism in NIDDM patients. Diabetes Care. 13: Schafer, G Biguanides. A review of history, pharmacodynamics and therapy. Diabete Metab. 9: Hundal, R.S., et al Mechanism by which metformin reduces glucose production in type 2 diabetes. Diabetes. 49: Hundal, H.S., Ramlal, T., Reyes, R., Leiter, L.A., and Klip, A Cellular mechanism of metformin action involves glucose transporter translocation from an intracellular pool to the plasma membrane in L6 muscle cells. Endocrinology. 131: Galuska, D., Nolte, L.A., Zierath, J.R., and Wallberg-Henriksson, H Effect of metformin on insulin-stimulated glucose transport in isolated skeletal muscle obtained from patients with NIDDM. Diabetologia. 37: Lin, H.Z., et al Metformin reverses fatty liver disease in obese, leptin-deficient mice. Nat. Med. 6: Hardie, D.G., and Carling, D The AMPactivated protein kinase: fuel gauge of the mammalian cell? Eur. J. Biochem. 246: Winder, W.W., and Hardie, D.G AMP-activated protein kinase, a metabolic master switch: possible roles in type 2 diabetes. Am. J. Physiol. 277:E1 E Hayashi, T., Hirshman, M.F., Kurth, E.J., Winder, W.W., and Goodyear, L.J Evidence for 5 AMP-activated protein kinase mediation of the effect of muscle contraction on glucose transport. Diabetes. 47: Merrill, G.F., Kurth, E.J., Hardie, D.G., and Winder, W.W AICA riboside increases AMP-activated protein kinase, fatty acid oxidation, and glucose uptake in rat muscle. Am. J. Physiol. 273:E1107 E Goodyear, L.J AMP-activated protein kinase: a critical signaling intermediary for exercise-stimulated glucose transport? Exerc. Sport Sci. Rev. 28: Lochhead, P.A., Salt, I.P., Walker, K.S., Hardie, D.G., and Sutherland, C aminoimidazole-4-carboxamide riboside mimics the effects of insulin on the expression of the 2 key gluconeogenic genes PEPCK and glucose-6-phosphatase. Diabetes. 49: Foretz, M., Carling, D., Guichard, C., Ferre, P., and Foufelle, F AMP-activated protein kinase inhibits the glucose-activated expression of fatty acid synthase gene in rat hepatocytes. J. Biol. Chem. 273: Holmes, B.F., Kurth-Kraczek, E.J., and Winder, W.W Chronic activation of 5 -AMP-activated protein kinase increases GLUT-4, hexokinase, and glycogen in muscle. J. Appl. Physiol. 87: Pollard, J.W., and Walker, J.M., editors Basic cell culture protocols. Methods in Molecular Biology series, Volume 75. Humana Press Inc. Totowa, New Jersey, USA Witters, L.A., and Kemp, B.E Insulin activation of acetyl-coa carboxylase accompanied by inhibition of the 5 -AMP-activated protein kinase. J. Biol. Chem. 267: Davies, S.P., Carling, D., and Hardie, D.G Tissue distribution of the AMP-activated protein kinase, and lack of activation by cyclic-ampdependent protein kinase, studied using a specific and sensitive peptide assay. Eur. J. Biochem. 186: Mannaerts, G.P., Debeer, L.J., Thomas, J., and DeSchepper, P.J Mitochondrial and peroxisomal fatty acid oxidation in liver homogenates and isolated hepatocytes from control and clofibrate-treated rats. J. Biol. Chem. 254: Carling, D., Clarke, P.R., Zammit,V.A., and Hardie, D.G Purification and characterization of the AMP-activated protein kinase. Copurification of acetyl-coa carboxylase kinase and 3-hydroxy-3-methylglutaryl-CoA reductase kinase activities. Eur. J. Biochem. 186: Hayashi, T., et al Metabolic stress and altered glucose transport: activation of AMP-activated protein kinase as a unifying coupling mechanism. Diabetes. 49: Sheng, Z., Otani H., Brown, M.S., and Goldstein, J.L Independent regulation of sterol regulatory element-binding proteins 1 and 2 in hamster liver. Proc. Natl. Acad. Sci. USA. 92: Cusi, K., and DeFronzo, R.A Metformin: a review of its metabolic effects. Diabetes Reviews. 6: Wiernsperger, N.F Membrane physiology as a basis for the cellular effects of metformin in insulin resistance and diabetes. Diabetes. Metab. 25: Sum, C.F., et al The effect of intravenous metformin on glucose metabolism during hyperglycaemia in type 2 diabetes. Diabet. Med. 9: Wilcock, C., Wyre, N.D., and Bailey, C.J Subcellular distribution of metformin in rat liver. J. Pharm. Pharmacol. 43: Wilcock, C., and Bailey, C.J Accumulation of metformin by tissues of the normal and diabetic mouse. Xenobiotica. 24: Hermann, L.S Metformin: a review of its pharmacological properties and therapeutic use. Diabete Metab. 5: Sterne, J., Duval, D., and Junien, J.L Metformin therapy. Research Clinical Forums 1: Owen, M.R., Doran, E., and Halestrap, A.P Evidence that metformin exerts its anti-diabetic effects through inhibition of complex 1 of the mitochondrial respiratory chain. Biochem. J. 348: El-Mir, M.Y., et al Dimethylbiguanide inhibits cell respiration via an indirect effect targeted on the respiratory chain complex I. J. Biol. Chem. 275: McGarry, J.D., and Brown, N.F The mitochondrial carnitine palmitoyltransferase system. From concept to molecular analysis. Eur. J. Biochem. 244: Shimomura, I., et al Decreased IRS-2 and increased SREBP-1c lead to mixed insulin resistance and sensitivity in livers of lipodystrophic and ob/ob mice. Mol. Cell. 6: Kakuma, T., et al Leptin, troglitazone, and the expression of sterol regulatory element binding proteins in liver and pancreatic islets. Proc. Natl. Acad. Sci. 97: Davies, S.P., Carling, D., Munday, M.R., and Hardie, D.G Diurnal rhythm of phosphorylation of rat liver acetyl-coa carboxylase by the AMP-activated protein kinase, demonstrated using freeze-clamping. Effects of high fat diets. Eur. J. Biochem. 203: McGarry, J.D What if Minkowski had been ageusic? An alternative angle on diabetes. Science. 258: The Journal of Clinical Investigation October 2001 Volume 108 Number 8
AMPK. Tomáš Kučera.
 AMPK (AMP- ACTIVATED PROTEIN KINASE ) Tomáš Kučera tomas.kucera@lfmotol.cuni.cz Department of Medical Chemistry and Clinical Biochemistry 2nd Faculty of Medicine, Charles University in Prague and Motol
AMPK (AMP- ACTIVATED PROTEIN KINASE ) Tomáš Kučera tomas.kucera@lfmotol.cuni.cz Department of Medical Chemistry and Clinical Biochemistry 2nd Faculty of Medicine, Charles University in Prague and Motol
Metformin inhibits hepatic gluconeogenesis in mice independently of the LKB1/AMPK pathway via a decrease in hepatic energy state
 Related Commentary, page 2267 Research article Metformin inhibits hepatic gluconeogenesis in mice independently of the LKB1/AMPK pathway via a decrease in hepatic energy state Marc Foretz, 1,2 Sophie Hébrard,
Related Commentary, page 2267 Research article Metformin inhibits hepatic gluconeogenesis in mice independently of the LKB1/AMPK pathway via a decrease in hepatic energy state Marc Foretz, 1,2 Sophie Hébrard,
BALANCING THE SCALES USING A NOVEL CELLULAR ENERGY SENSOR
 The West London Medical Journal 2010 Vol 2 No 4 pp 29-35 BALANCING THE SCALES USING A NOVEL CELLULAR ENERGY SENSOR Sairah Akbar The topic of obesity is rarely out of the public eye with an increasingly
The West London Medical Journal 2010 Vol 2 No 4 pp 29-35 BALANCING THE SCALES USING A NOVEL CELLULAR ENERGY SENSOR Sairah Akbar The topic of obesity is rarely out of the public eye with an increasingly
Metabolic integration and Regulation
 Metabolic integration and Regulation 109700: Graduate Biochemistry Trimester 2/2016 Assistant Prof. Dr. Panida Khunkaewla kpanida@sut.ac.th School of Chemistry Suranaree University of Technology 1 Overview
Metabolic integration and Regulation 109700: Graduate Biochemistry Trimester 2/2016 Assistant Prof. Dr. Panida Khunkaewla kpanida@sut.ac.th School of Chemistry Suranaree University of Technology 1 Overview
2.5. AMPK activity
 Supplement Fig. A 3 B phos-ampk 2.5 * Control AICAR AMPK AMPK activity (Absorbance at 45 nm) 2.5.5 Control AICAR Supplement Fig. Effects of AICAR on AMPK activation in macrophages. J774. macrophages were
Supplement Fig. A 3 B phos-ampk 2.5 * Control AICAR AMPK AMPK activity (Absorbance at 45 nm) 2.5.5 Control AICAR Supplement Fig. Effects of AICAR on AMPK activation in macrophages. J774. macrophages were
Metabolism of cardiac muscle. Dr. Mamoun Ahram Cardiovascular system, 2013
 Metabolism of cardiac muscle Dr. Mamoun Ahram Cardiovascular system, 2013 References This lecture Mark s Basic Medical Biochemistry, 4 th ed., p. 890-891 Hand-out Why is this topic important? Heart failure
Metabolism of cardiac muscle Dr. Mamoun Ahram Cardiovascular system, 2013 References This lecture Mark s Basic Medical Biochemistry, 4 th ed., p. 890-891 Hand-out Why is this topic important? Heart failure
AMPK. Tomáš Kuc era. Ústav lékar ské chemie a klinické biochemie 2. lékar ská fakulta, Univerzita Karlova v Praze
 AMPK (AMP- ACTIVATED PROTEIN KINASE ) Tomáš Kuc era Ústav lékar ské chemie a klinické biochemie 2. lékar ská fakulta, Univerzita Karlova v Praze 2013 AMPK AMP-ACTIVATED PROTEIN KINASE present in all eukaryotic
AMPK (AMP- ACTIVATED PROTEIN KINASE ) Tomáš Kuc era Ústav lékar ské chemie a klinické biochemie 2. lékar ská fakulta, Univerzita Karlova v Praze 2013 AMPK AMP-ACTIVATED PROTEIN KINASE present in all eukaryotic
Supplemental Table 1 Primer sequences (mouse) used for real-time qrt-pcr studies
 Supplemental Table 1 Primer sequences (mouse) used for real-time qrt-pcr studies Gene symbol Forward primer Reverse primer ACC1 5'-TGAGGAGGACCGCATTTATC 5'-GCATGGAATGGCAGTAAGGT ACLY 5'-GACACCATCTGTGATCTTG
Supplemental Table 1 Primer sequences (mouse) used for real-time qrt-pcr studies Gene symbol Forward primer Reverse primer ACC1 5'-TGAGGAGGACCGCATTTATC 5'-GCATGGAATGGCAGTAAGGT ACLY 5'-GACACCATCTGTGATCTTG
THE GLUCOSE-FATTY ACID-KETONE BODY CYCLE Role of ketone bodies as respiratory substrates and metabolic signals
 Br. J. Anaesth. (1981), 53, 131 THE GLUCOSE-FATTY ACID-KETONE BODY CYCLE Role of ketone bodies as respiratory substrates and metabolic signals J. C. STANLEY In this paper, the glucose-fatty acid cycle
Br. J. Anaesth. (1981), 53, 131 THE GLUCOSE-FATTY ACID-KETONE BODY CYCLE Role of ketone bodies as respiratory substrates and metabolic signals J. C. STANLEY In this paper, the glucose-fatty acid cycle
number Done by Corrected by Doctor Faisal Al-Khatibe
 number 24 Done by Mohammed tarabieh Corrected by Doctor Faisal Al-Khatibe 1 P a g e *Please look over the previous sheet about fatty acid synthesis **Oxidation(degradation) of fatty acids, occurs in the
number 24 Done by Mohammed tarabieh Corrected by Doctor Faisal Al-Khatibe 1 P a g e *Please look over the previous sheet about fatty acid synthesis **Oxidation(degradation) of fatty acids, occurs in the
Supplementary Figure 1.
 Supplementary Figure 1. FGF21 does not exert direct effects on hepatic glucose production. The liver explants from C57BL/6J mice (A, B) or primary rat hepatocytes (C, D) were incubated with rmfgf21 (2
Supplementary Figure 1. FGF21 does not exert direct effects on hepatic glucose production. The liver explants from C57BL/6J mice (A, B) or primary rat hepatocytes (C, D) were incubated with rmfgf21 (2
Chapter 10. Introduction to Nutrition and Metabolism, 3 rd edition David A Bender Taylor & Francis Ltd, London 2002
 Chapter 10 Introduction to Nutrition and Metabolism, 3 rd edition David A Bender Taylor & Francis Ltd, London 2002 Chapter 10: Integration and Control of Metabolism Press the space bar or click the mouse
Chapter 10 Introduction to Nutrition and Metabolism, 3 rd edition David A Bender Taylor & Francis Ltd, London 2002 Chapter 10: Integration and Control of Metabolism Press the space bar or click the mouse
Oxidation of Long Chain Fatty Acids
 Oxidation of Long Chain Fatty Acids Dr NC Bird Oxidation of long chain fatty acids is the primary source of energy supply in man and animals. Hibernating animals utilise fat stores to maintain body heat,
Oxidation of Long Chain Fatty Acids Dr NC Bird Oxidation of long chain fatty acids is the primary source of energy supply in man and animals. Hibernating animals utilise fat stores to maintain body heat,
5.0 HORMONAL CONTROL OF CARBOHYDRATE METABOLISM
 5.0 HORMONAL CONTROL OF CARBOHYDRATE METABOLISM Introduction: Variety of hormones and other molecules regulate the carbohydrates metabolism. Some of these have already been cited in previous sections.
5.0 HORMONAL CONTROL OF CARBOHYDRATE METABOLISM Introduction: Variety of hormones and other molecules regulate the carbohydrates metabolism. Some of these have already been cited in previous sections.
Metformin and phenformin are derivatives of
 The Antidiabetic Drug Metformin Activates the AMP-Activated Protein Kinase Cascade via an Adenine Nucleotide-Independent Mechanism Simon A. Hawley, 1 Anne E. Gadalla, 1 Grith Skytte Olsen, 2 and D. Grahame
The Antidiabetic Drug Metformin Activates the AMP-Activated Protein Kinase Cascade via an Adenine Nucleotide-Independent Mechanism Simon A. Hawley, 1 Anne E. Gadalla, 1 Grith Skytte Olsen, 2 and D. Grahame
Role of fatty acids in the development of insulin resistance and type 2 diabetes mellitus
 Emerging Science Role of fatty acids in the development of insulin resistance and type 2 diabetes mellitus George Wolf Insulin resistance is defined as the reduced responsiveness to normal circulating
Emerging Science Role of fatty acids in the development of insulin resistance and type 2 diabetes mellitus George Wolf Insulin resistance is defined as the reduced responsiveness to normal circulating
UNIVERSITY OF PNG SCHOOL OF MEDICINE AND HEALTH SCIENCES DIVISION OF BASIC MEDICAL SCIENCES DISCIPLINE OF BIOCHEMISTRY AND MOLECULAR BIOLOGY
 1 UNIVERSITY OF PNG SCHOOL OF MEDICINE AND HEALTH SCIENCES DIVISION OF BASIC MEDICAL SCIENCES DISCIPLINE OF BIOCHEMISTRY AND MOLECULAR BIOLOGY GLUCOSE HOMEOSTASIS An Overview WHAT IS HOMEOSTASIS? Homeostasis
1 UNIVERSITY OF PNG SCHOOL OF MEDICINE AND HEALTH SCIENCES DIVISION OF BASIC MEDICAL SCIENCES DISCIPLINE OF BIOCHEMISTRY AND MOLECULAR BIOLOGY GLUCOSE HOMEOSTASIS An Overview WHAT IS HOMEOSTASIS? Homeostasis
Companion to Biosynthesis of Ketones & Cholesterols, Regulation of Lipid Metabolism Lecture Notes
 Companion to Biosynthesis of Ketones & Cholesterols, Regulation of Lipid Metabolism Lecture Notes The major site of acetoacetate and 3-hydorxybutyrate production is in the liver. 3-hydorxybutyrate is the
Companion to Biosynthesis of Ketones & Cholesterols, Regulation of Lipid Metabolism Lecture Notes The major site of acetoacetate and 3-hydorxybutyrate production is in the liver. 3-hydorxybutyrate is the
Final Review Sessions. 3/16 (FRI) 126 Wellman (4-6 6 pm) 3/19 (MON) 1309 Surge 3 (4-6 6 pm) Office Hours
 Final Review Sessions 3/16 (FRI) 126 Wellman (4-6 6 pm) 3/19 (MON) 1309 Surge 3 (4-6 6 pm) Office ours 3/14 (WED) 9:30 11:30 am (Rebecca) 3/16 (FRI) 9-11 am (Abel) Final ESSENTIALS Posted Lecture 20 ormonal
Final Review Sessions 3/16 (FRI) 126 Wellman (4-6 6 pm) 3/19 (MON) 1309 Surge 3 (4-6 6 pm) Office ours 3/14 (WED) 9:30 11:30 am (Rebecca) 3/16 (FRI) 9-11 am (Abel) Final ESSENTIALS Posted Lecture 20 ormonal
Integration Of Metabolism
 Integration Of Metabolism Metabolism Consist of Highly Interconnected Pathways The basic strategy of catabolic metabolism is to form ATP, NADPH, and building blocks for biosyntheses. 1. ATP is the universal
Integration Of Metabolism Metabolism Consist of Highly Interconnected Pathways The basic strategy of catabolic metabolism is to form ATP, NADPH, and building blocks for biosyntheses. 1. ATP is the universal
Roles of Lipids. principal form of stored energy major constituents of cell membranes vitamins messengers intra and extracellular
 Roles of Lipids principal form of stored energy major constituents of cell membranes vitamins messengers intra and extracellular = Oxidation of fatty acids Central energy-yielding pathway in animals. O
Roles of Lipids principal form of stored energy major constituents of cell membranes vitamins messengers intra and extracellular = Oxidation of fatty acids Central energy-yielding pathway in animals. O
Integrative Metabolism: Significance
 Integrative Metabolism: Significance Energy Containing Nutrients Carbohydrates Fats Proteins Catabolism Energy Depleted End Products H 2 O NH 3 ADP + Pi NAD + NADP + FAD + Pi NADH+H + NADPH+H + FADH2 Cell
Integrative Metabolism: Significance Energy Containing Nutrients Carbohydrates Fats Proteins Catabolism Energy Depleted End Products H 2 O NH 3 ADP + Pi NAD + NADP + FAD + Pi NADH+H + NADPH+H + FADH2 Cell
Successful completion of Phase I clinical trial of AMPK activator O304
 Successful completion of Phase I clinical trial of AMPK activator O304 O304 is safe and very well tolerated in young healthy subjects, in middle aged obese subjects, and in type 2 diabetics in combination
Successful completion of Phase I clinical trial of AMPK activator O304 O304 is safe and very well tolerated in young healthy subjects, in middle aged obese subjects, and in type 2 diabetics in combination
Medical Biochemistry and Molecular Biology department
 Medical Biochemistry and Molecular Biology department Cardiac Fuels [Sources of energy for the Cardiac muscle] Intended learning outcomes of the lecture: By the end of this lecture you would be able to:-
Medical Biochemistry and Molecular Biology department Cardiac Fuels [Sources of energy for the Cardiac muscle] Intended learning outcomes of the lecture: By the end of this lecture you would be able to:-
LIPID METABOLISM
 LIPID METABOLISM LIPOGENESIS LIPOGENESIS LIPOGENESIS FATTY ACID SYNTHESIS DE NOVO FFA in the blood come from :- (a) Dietary fat (b) Dietary carbohydrate/protein in excess of need FA TAG Site of synthesis:-
LIPID METABOLISM LIPOGENESIS LIPOGENESIS LIPOGENESIS FATTY ACID SYNTHESIS DE NOVO FFA in the blood come from :- (a) Dietary fat (b) Dietary carbohydrate/protein in excess of need FA TAG Site of synthesis:-
Biochemistry: A Short Course
 Tymoczko Berg Stryer Biochemistry: A Short Course Second Edition CHAPTER 28 Fatty Acid Synthesis 2013 W. H. Freeman and Company Chapter 28 Outline 1. The first stage of fatty acid synthesis is transfer
Tymoczko Berg Stryer Biochemistry: A Short Course Second Edition CHAPTER 28 Fatty Acid Synthesis 2013 W. H. Freeman and Company Chapter 28 Outline 1. The first stage of fatty acid synthesis is transfer
Integration & Hormone Regulation
 Integration Branchpoints in metabolism where metabolites can go several directions 1. Glucose 6-phosphate Energy needed (low energy charge): glycolysis Low blood sugar: high [glucagon], low [insulin] glycogen
Integration Branchpoints in metabolism where metabolites can go several directions 1. Glucose 6-phosphate Energy needed (low energy charge): glycolysis Low blood sugar: high [glucagon], low [insulin] glycogen
Integration of Metabolism 1. made by: Noor M. ALnairat. Sheet No. 18
 Integration of Metabolism 1 made by: Noor M. ALnairat Sheet No. 18 Data :24/11/2016 SLIDE 2: Metabolism Consist of Highly Interconnected Pathways The basic strategy of catabolic metabolism is to form ATP,
Integration of Metabolism 1 made by: Noor M. ALnairat Sheet No. 18 Data :24/11/2016 SLIDE 2: Metabolism Consist of Highly Interconnected Pathways The basic strategy of catabolic metabolism is to form ATP,
INSULIN RESISTANCE: MOLECULAR MECHANISM
 INSULIN RESISTANCE: MOLECULAR MECHANISM Ashish K. Saha ABSTRACT Insulin resistance in skeletal muscle is present in humans with type 2 diabetes (non-insulin dependent diabetes mellitus) and obesity and
INSULIN RESISTANCE: MOLECULAR MECHANISM Ashish K. Saha ABSTRACT Insulin resistance in skeletal muscle is present in humans with type 2 diabetes (non-insulin dependent diabetes mellitus) and obesity and
Regulation of Metabolism
 Regulation of Metabolism Pratt and Cornely Chapter 19 Regulation by Compartmentalization Form of reciprocal regulation Degradation vs biosynthesis Requires transporters 1 Specialization of organs Fuel
Regulation of Metabolism Pratt and Cornely Chapter 19 Regulation by Compartmentalization Form of reciprocal regulation Degradation vs biosynthesis Requires transporters 1 Specialization of organs Fuel
Energy metabolism - the overview
 Energy metabolism - the overview Josef Fontana EC - 40 Overview of the lecture Important terms of the energy metabolism The overview of the energy metabolism The main pathways of the energy metabolism
Energy metabolism - the overview Josef Fontana EC - 40 Overview of the lecture Important terms of the energy metabolism The overview of the energy metabolism The main pathways of the energy metabolism
AMPK as a metabolic switch in rat muscle, liver and adipose tissue after exercise
 Acta Physiol Scand 23, 78, 43 442 AMPK as a metabolic switch in rat muscle, liver and adipose tissue after exercise N. B. Ruderman, H. Park, V. K. Kaushik, D. Dean, S. Constant, M. Prentki 2 and A. K.
Acta Physiol Scand 23, 78, 43 442 AMPK as a metabolic switch in rat muscle, liver and adipose tissue after exercise N. B. Ruderman, H. Park, V. K. Kaushik, D. Dean, S. Constant, M. Prentki 2 and A. K.
What systems are involved in homeostatic regulation (give an example)?
 1 UNIVERSITY OF PNG SCHOOL OF MEDICINE AND HEALTH SCIENCES DIVISION OF BASIC MEDICAL SCIENCES DISCIPLINE OF BIOCHEMISTRY AND MOLECULAR BIOLOGY GLUCOSE HOMEOSTASIS (Diabetes Mellitus Part 1): An Overview
1 UNIVERSITY OF PNG SCHOOL OF MEDICINE AND HEALTH SCIENCES DIVISION OF BASIC MEDICAL SCIENCES DISCIPLINE OF BIOCHEMISTRY AND MOLECULAR BIOLOGY GLUCOSE HOMEOSTASIS (Diabetes Mellitus Part 1): An Overview
In glycolysis, glucose is converted to pyruvate. If the pyruvate is reduced to lactate, the pathway does not require O 2 and is called anaerobic
 Glycolysis 1 In glycolysis, glucose is converted to pyruvate. If the pyruvate is reduced to lactate, the pathway does not require O 2 and is called anaerobic glycolysis. If this pyruvate is converted instead
Glycolysis 1 In glycolysis, glucose is converted to pyruvate. If the pyruvate is reduced to lactate, the pathway does not require O 2 and is called anaerobic glycolysis. If this pyruvate is converted instead
23.1 Lipid Metabolism in Animals. Chapter 23. Micelles Lipid Metabolism in. Animals. Overview of Digestion Lipid Metabolism in
 Denniston Topping Caret Copyright! The McGraw-Hill Companies, Inc. Permission required for reproduction or display. Chapter 23 Fatty Acid Metabolism Triglycerides (Tgl) are emulsified into fat droplets
Denniston Topping Caret Copyright! The McGraw-Hill Companies, Inc. Permission required for reproduction or display. Chapter 23 Fatty Acid Metabolism Triglycerides (Tgl) are emulsified into fat droplets
Over-expression of MKP-3 and knockdown of MKP-3 and FOXO1 in primary rat. day, cells were transduced with adenoviruses expressing GFP, MKP-3 or shgfp,
 SUPPLEMENTAL METHODS Over-expression of MKP-3 and knockdown of MKP-3 and FOXO1 in primary rat hepatocytes Primary rat hepatocytes were seeded as described in experimental procedures. The next day, cells
SUPPLEMENTAL METHODS Over-expression of MKP-3 and knockdown of MKP-3 and FOXO1 in primary rat hepatocytes Primary rat hepatocytes were seeded as described in experimental procedures. The next day, cells
Lipid Metabolism. Remember fats?? Triacylglycerols - major form of energy storage in animals
 Remember fats?? Triacylglycerols - major form of energy storage in animals Your energy reserves: ~0.5% carbs (glycogen + glucose) ~15% protein (muscle, last resort) ~85% fat Why use fat for energy? 1 gram
Remember fats?? Triacylglycerols - major form of energy storage in animals Your energy reserves: ~0.5% carbs (glycogen + glucose) ~15% protein (muscle, last resort) ~85% fat Why use fat for energy? 1 gram
ANSC 619 PHYSIOLOGICAL CHEMISTRY OF LIVESTOCK SPECIES. Carbohydrate Metabolism
 ANSC 619 PHYSIOLOGICAL CHEMISTRY OF LIVESTOCK SPECIES I. Glycolysis A. Pathway Regulation of glycolysis Hexokinase: Activated by glucose. Inhibited by G6P. 6-Phosphofructokinase: Inhibited by ATP, especially
ANSC 619 PHYSIOLOGICAL CHEMISTRY OF LIVESTOCK SPECIES I. Glycolysis A. Pathway Regulation of glycolysis Hexokinase: Activated by glucose. Inhibited by G6P. 6-Phosphofructokinase: Inhibited by ATP, especially
Lipid Metabolism. Catabolism Overview
 Lipid Metabolism Pratt & Cornely, Chapter 17 Catabolism Overview Lipids as a fuel source from diet Beta oxidation Mechanism ATP production Ketone bodies as fuel 1 High energy More reduced Little water
Lipid Metabolism Pratt & Cornely, Chapter 17 Catabolism Overview Lipids as a fuel source from diet Beta oxidation Mechanism ATP production Ketone bodies as fuel 1 High energy More reduced Little water
Chronic activation of 5 -AMP-activated protein kinase increases GLUT-4, hexokinase, and glycogen in muscle
 highlighted topics Chronic activation of 5 -AMP-activated protein kinase increases GLUT-4, hexokinase, and glycogen in muscle B. F. HOLMES, E. J. KURTH-KRACZEK, AND W. W. WINDER Department of Zoology,
highlighted topics Chronic activation of 5 -AMP-activated protein kinase increases GLUT-4, hexokinase, and glycogen in muscle B. F. HOLMES, E. J. KURTH-KRACZEK, AND W. W. WINDER Department of Zoology,
CARBOHYDRATE METABOLISM 1
 CARBOHYDRATE METABOLISM 1 web 2017 József Mandl Strategy of metabolism 1 Strategy of metabolism to extract energy ( hydrogen ) from the environment to store the energy excess to store hydrogen CH 3 O 2
CARBOHYDRATE METABOLISM 1 web 2017 József Mandl Strategy of metabolism 1 Strategy of metabolism to extract energy ( hydrogen ) from the environment to store the energy excess to store hydrogen CH 3 O 2
AMPK Phosphorylation Assay Kit
 AMPK Phosphorylation Assay Kit Catalog Number KA3789 100 assays Version: 02 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
AMPK Phosphorylation Assay Kit Catalog Number KA3789 100 assays Version: 02 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle
Multiple choice: Circle the best answer on this exam. There are 12 multiple choice questions, each question is worth 3 points.
 CHEM 4420 Exam 4 Spring 2015 Dr. Stone Page 1 of 6 Name Use complete sentences when requested. There are 120 possible points on this exam. Therefore there are 20 bonus points. Multiple choice: Circle the
CHEM 4420 Exam 4 Spring 2015 Dr. Stone Page 1 of 6 Name Use complete sentences when requested. There are 120 possible points on this exam. Therefore there are 20 bonus points. Multiple choice: Circle the
Modulating Glucose Uptake in Skeletal Myotubes: Insulin Induction with Bioluminescent Glucose Uptake Analysis
 icell Skeletal Myoblasts Application Protocol Modulating Glucose Uptake in Skeletal Myotubes: Insulin Induction with Bioluminescent Glucose Uptake Analysis Introduction The skeletal muscle is one of the
icell Skeletal Myoblasts Application Protocol Modulating Glucose Uptake in Skeletal Myotubes: Insulin Induction with Bioluminescent Glucose Uptake Analysis Introduction The skeletal muscle is one of the
anabolic pathways- Catabolic Amphibolic
 METABOLISM Introduction The fate of dietary components after digestion and absorption constitute metabolism regulated by metabolic pathway 3 types: anabolic pathways- Synthesis of compound e.g. synthesis
METABOLISM Introduction The fate of dietary components after digestion and absorption constitute metabolism regulated by metabolic pathway 3 types: anabolic pathways- Synthesis of compound e.g. synthesis
Intermediary metabolism. Eva Samcová
 Intermediary metabolism Eva Samcová Metabolic roles of tissues Four major tissues play a dominant role in fuel metabolism : liver, adipose, muscle, and brain. These tissues do not function in isolation.
Intermediary metabolism Eva Samcová Metabolic roles of tissues Four major tissues play a dominant role in fuel metabolism : liver, adipose, muscle, and brain. These tissues do not function in isolation.
General Laboratory methods Plasma analysis: Gene Expression Analysis: Immunoblot analysis: Immunohistochemistry:
 General Laboratory methods Plasma analysis: Plasma insulin (Mercodia, Sweden), leptin (duoset, R&D Systems Europe, Abingdon, United Kingdom), IL-6, TNFα and adiponectin levels (Quantikine kits, R&D Systems
General Laboratory methods Plasma analysis: Plasma insulin (Mercodia, Sweden), leptin (duoset, R&D Systems Europe, Abingdon, United Kingdom), IL-6, TNFα and adiponectin levels (Quantikine kits, R&D Systems
(de novo synthesis of glucose)
 Gluconeogenesis (de novo synthesis of glucose) Gluconeogenesis Gluconeogenesis is the biosynthesis of new glucose. The main purpose of gluconeogenesis is to maintain the constant blood Glc concentration.
Gluconeogenesis (de novo synthesis of glucose) Gluconeogenesis Gluconeogenesis is the biosynthesis of new glucose. The main purpose of gluconeogenesis is to maintain the constant blood Glc concentration.
Requires Signaling though Akt2 Independent of the. Transcription Factors FoxA2, FoxO1, and SREBP1c
 Cell Metabolism, Volume 14 Supplemental Information Postprandial Hepatic Lipid Metabolism Requires Signaling though Akt2 Independent of the Transcription Factors FoxA2, FoxO1, and SREBP1c Min Wan, Karla
Cell Metabolism, Volume 14 Supplemental Information Postprandial Hepatic Lipid Metabolism Requires Signaling though Akt2 Independent of the Transcription Factors FoxA2, FoxO1, and SREBP1c Min Wan, Karla
Glucose. Glucose. Insulin Action. Introduction to Hormonal Regulation of Fuel Metabolism
 Glucose Introduction to Hormonal Regulation of Fuel Metabolism Fasting level 3.5-5 mmol (1 mmol = 18 mg/dl) Postprandial 6-10 mmol Amount of glucose in circulation is dependent on: Absorption from the
Glucose Introduction to Hormonal Regulation of Fuel Metabolism Fasting level 3.5-5 mmol (1 mmol = 18 mg/dl) Postprandial 6-10 mmol Amount of glucose in circulation is dependent on: Absorption from the
Phosphorylation of rat muscle acetyl-coa carboxylase by AMP-activated protein kinase and protein kinase A
 Phosphorylation of rat muscle acetyl-coa carboxylase by AMP-activated protein kinase and protein kinase A W. W. WINDER, 1 H. A. WILSON, 1 D. G. HARDIE, 2 B. B. RASMUSSEN, 1 C. A. HUTBER, 1 G. B. CALL,
Phosphorylation of rat muscle acetyl-coa carboxylase by AMP-activated protein kinase and protein kinase A W. W. WINDER, 1 H. A. WILSON, 1 D. G. HARDIE, 2 B. B. RASMUSSEN, 1 C. A. HUTBER, 1 G. B. CALL,
Dr. Mohnen s notes on GLUCONEOGENESIS
 Dr. Mohnen s notes on GLUCONEOGENESIS Note: Even though we did not get through all of these slides during lecture, I advise you to look them all through because they will be helpful to you as you learn
Dr. Mohnen s notes on GLUCONEOGENESIS Note: Even though we did not get through all of these slides during lecture, I advise you to look them all through because they will be helpful to you as you learn
PXL770, a novel direct AMPK activator, improves metabolic disorders in a diet-induced mice model of obesity and diabetes
 PXL770, a novel direct AMPK activator, improves metabolic disorders in a diet-induced mice model of obesity and diabetes Sébastien Bolze 1 ; Sophie Hallakou-Bozec 1 ; Michael Roden 2, 3,4 ; Julien Roux
PXL770, a novel direct AMPK activator, improves metabolic disorders in a diet-induced mice model of obesity and diabetes Sébastien Bolze 1 ; Sophie Hallakou-Bozec 1 ; Michael Roden 2, 3,4 ; Julien Roux
ENERGY FROM INGESTED NUTREINTS MAY BE USED IMMEDIATELY OR STORED
 QUIZ/TEST REVIEW NOTES SECTION 1 SHORT TERM METABOLISM [METABOLISM] Learning Objectives: Identify primary energy stores of the body Differentiate the metabolic processes of the fed and fasted states Explain
QUIZ/TEST REVIEW NOTES SECTION 1 SHORT TERM METABOLISM [METABOLISM] Learning Objectives: Identify primary energy stores of the body Differentiate the metabolic processes of the fed and fasted states Explain
18. PANCREATIC FUNCTION AND METABOLISM. Pancreatic secretions ISLETS OF LANGERHANS. Insulin
 18. PANCREATIC FUNCTION AND METABOLISM ISLETS OF LANGERHANS Some pancreatic functions have already been discussed in the digestion section. In this one, the emphasis will be placed on the endocrine function
18. PANCREATIC FUNCTION AND METABOLISM ISLETS OF LANGERHANS Some pancreatic functions have already been discussed in the digestion section. In this one, the emphasis will be placed on the endocrine function
Fig. S1. Dose-response effects of acute administration of the β3 adrenoceptor agonists CL316243, BRL37344, ICI215,001, ZD7114, ZD2079 and CGP12177 at
 Fig. S1. Dose-response effects of acute administration of the β3 adrenoceptor agonists CL316243, BRL37344, ICI215,001, ZD7114, ZD2079 and CGP12177 at doses of 0.1, 0.5 and 1 mg/kg on cumulative food intake
Fig. S1. Dose-response effects of acute administration of the β3 adrenoceptor agonists CL316243, BRL37344, ICI215,001, ZD7114, ZD2079 and CGP12177 at doses of 0.1, 0.5 and 1 mg/kg on cumulative food intake
BIOL212 Biochemistry of Disease. Metabolic Disorders - Obesity
 BIOL212 Biochemistry of Disease Metabolic Disorders - Obesity Obesity Approx. 23% of adults are obese in the U.K. The number of obese children has tripled in 20 years. 10% of six year olds are obese, rising
BIOL212 Biochemistry of Disease Metabolic Disorders - Obesity Obesity Approx. 23% of adults are obese in the U.K. The number of obese children has tripled in 20 years. 10% of six year olds are obese, rising
Manipulation of the Nutrient Sensors (AMPK/TOR) with Anaplerotic Diet Therapy (Triheptanoin) An Alternative to Diet Restriction
 Manipulation of the Nutrient Sensors (AMPK/TOR) with Anaplerotic Diet Therapy (Triheptanoin) An Alternative to Diet Restriction CharlesR.Roe,MD Institute of Metabolic Disease Baylor University Medical
Manipulation of the Nutrient Sensors (AMPK/TOR) with Anaplerotic Diet Therapy (Triheptanoin) An Alternative to Diet Restriction CharlesR.Roe,MD Institute of Metabolic Disease Baylor University Medical
Integration Of Metabolism
 Integration Of Metabolism Metabolism Consist of Highly Interconnected Pathways The basic strategy of catabolic metabolism is to form ATP, NADPH, and building blocks for biosyntheses. 1. ATP is the universal
Integration Of Metabolism Metabolism Consist of Highly Interconnected Pathways The basic strategy of catabolic metabolism is to form ATP, NADPH, and building blocks for biosyntheses. 1. ATP is the universal
A Hepatocyte Growth Factor Receptor (Met) Insulin Receptor hybrid governs hepatic glucose metabolism SUPPLEMENTARY FIGURES, LEGENDS AND METHODS
 A Hepatocyte Growth Factor Receptor (Met) Insulin Receptor hybrid governs hepatic glucose metabolism Arlee Fafalios, Jihong Ma, Xinping Tan, John Stoops, Jianhua Luo, Marie C. DeFrances and Reza Zarnegar
A Hepatocyte Growth Factor Receptor (Met) Insulin Receptor hybrid governs hepatic glucose metabolism Arlee Fafalios, Jihong Ma, Xinping Tan, John Stoops, Jianhua Luo, Marie C. DeFrances and Reza Zarnegar
Biochemistry 7/11/ Bio-Energetics & ATP. 5.1) ADP, ATP and Cellular Respiration OVERVIEW OF ENERGY AND METABOLISM
 Biochemistry 5. Bio-Energetics & ATP 5.1) ADP, ATP and Cellular Respiration Prof. Dr. Klaus Heese OVERVIEW OF ENERGY AND METABOLISM 1. The food we eat, (carbohydrates/ glucose /sugar, lipids/fat, proteins),
Biochemistry 5. Bio-Energetics & ATP 5.1) ADP, ATP and Cellular Respiration Prof. Dr. Klaus Heese OVERVIEW OF ENERGY AND METABOLISM 1. The food we eat, (carbohydrates/ glucose /sugar, lipids/fat, proteins),
Chapter 10. Regulatory Strategy
 Chapter 10 Regulatory Strategy Regulation of enzymatic activity: 1. Allosteric Control. Allosteric proteins have a regulatory site(s) and multiple functional sites Activity of proteins is regulated by
Chapter 10 Regulatory Strategy Regulation of enzymatic activity: 1. Allosteric Control. Allosteric proteins have a regulatory site(s) and multiple functional sites Activity of proteins is regulated by
Biol 219 Lec 7 Fall 2016
 Cellular Respiration: Harvesting Energy to form ATP Cellular Respiration and Metabolism Glucose ATP Pyruvate Lactate Acetyl CoA NAD + Introducing The Players primary substrate for cellular respiration
Cellular Respiration: Harvesting Energy to form ATP Cellular Respiration and Metabolism Glucose ATP Pyruvate Lactate Acetyl CoA NAD + Introducing The Players primary substrate for cellular respiration
ANSC 689 PHYSIOLOGICAL CHEMISTRY OF LIVESTOCK SPECIDS. Enzyme Kinetics and Control Reactions
 Handout Enzyme Kinetics and Control Reactions ANSC 689 PHYSIOLOGICAL CHEMISTRY OF LIVESTOCK SPECIDS Enzyme Kinetics and Control Reactions I. Kinetics A. Reaction rates 1. First order (reaction rate is
Handout Enzyme Kinetics and Control Reactions ANSC 689 PHYSIOLOGICAL CHEMISTRY OF LIVESTOCK SPECIDS Enzyme Kinetics and Control Reactions I. Kinetics A. Reaction rates 1. First order (reaction rate is
Chemistry 1120 Exam 4 Study Guide
 Chemistry 1120 Exam 4 Study Guide Chapter 12 12.1 Identify and differentiate between macronutrients (lipids, amino acids and saccharides) and micronutrients (vitamins and minerals). Master Tutor Section
Chemistry 1120 Exam 4 Study Guide Chapter 12 12.1 Identify and differentiate between macronutrients (lipids, amino acids and saccharides) and micronutrients (vitamins and minerals). Master Tutor Section
LIPID METABOLISM. Sri Widia A Jusman Department of Biochemistry & Molecular Biology FMUI
 LIPID METABOLISM Sri Widia A Jusman Department of Biochemistry & Molecular Biology FMUI Lipid metabolism is concerned mainly with fatty acids cholesterol Source of fatty acids from dietary fat de novo
LIPID METABOLISM Sri Widia A Jusman Department of Biochemistry & Molecular Biology FMUI Lipid metabolism is concerned mainly with fatty acids cholesterol Source of fatty acids from dietary fat de novo
OVERVIEW OF ENERGY AND METABOLISM
 Biochemistry 5. Bio-Energetics & ATP 5.1) ADP, ATP and Cellular Respiration OVERVIEW OF ENERGY AND METABOLISM 1. The food we eat, (carbohydrates/ glucose /sugar, lipids/fat, proteins), are our only source
Biochemistry 5. Bio-Energetics & ATP 5.1) ADP, ATP and Cellular Respiration OVERVIEW OF ENERGY AND METABOLISM 1. The food we eat, (carbohydrates/ glucose /sugar, lipids/fat, proteins), are our only source
Biosynthesis of Fatty Acids. By Dr.QUTAIBA A. QASIM
 Biosynthesis of Fatty Acids By Dr.QUTAIBA A. QASIM Fatty Acids Definition Fatty acids are comprised of hydrocarbon chains terminating with carboxylic acid groups. Fatty acids and their associated derivatives
Biosynthesis of Fatty Acids By Dr.QUTAIBA A. QASIM Fatty Acids Definition Fatty acids are comprised of hydrocarbon chains terminating with carboxylic acid groups. Fatty acids and their associated derivatives
Application Note. Introduction
 Simultaneously Measuring Oxidation of Exogenous and Endogenous Fatty Acids Using the XF Palmitate-BSA FAO Substrate with the Agilent Seahorse XF Cell Mito Stress Test Application Note Introduction The
Simultaneously Measuring Oxidation of Exogenous and Endogenous Fatty Acids Using the XF Palmitate-BSA FAO Substrate with the Agilent Seahorse XF Cell Mito Stress Test Application Note Introduction The
Validation & Assay Performance Summary
 Validation & Assay Performance Summary LanthaScreen IGF-1R GripTite Cells Cat. no. K1834 Modification Detected: Phosphorylation of Multiple Tyr Residues on IGF-1R LanthaScreen Cellular Assay Validation
Validation & Assay Performance Summary LanthaScreen IGF-1R GripTite Cells Cat. no. K1834 Modification Detected: Phosphorylation of Multiple Tyr Residues on IGF-1R LanthaScreen Cellular Assay Validation
BIOL212- Biochemistry of Disease. Metabolic Disorders: Diabetes
 BIOL212- Biochemistry of Disease Metabolic Disorders: Diabetes Diabetes mellitus is, after heart disease and cancer, the third leading cause of death in the west. Insulin is either not secreted in sufficient
BIOL212- Biochemistry of Disease Metabolic Disorders: Diabetes Diabetes mellitus is, after heart disease and cancer, the third leading cause of death in the west. Insulin is either not secreted in sufficient
Integration of Metabolism
 Integration of Metabolism Metabolism is a continuous process. Thousands of reactions occur simultaneously in order to maintain homeostasis. It ensures a supply of fuel, to tissues at all times, in fed
Integration of Metabolism Metabolism is a continuous process. Thousands of reactions occur simultaneously in order to maintain homeostasis. It ensures a supply of fuel, to tissues at all times, in fed
Glycolysis Part 2. BCH 340 lecture 4
 Glycolysis Part 2 BCH 340 lecture 4 Regulation of Glycolysis There are three steps in glycolysis that have enzymes which regulate the flux of glycolysis These enzymes catalyzes irreversible reactions of
Glycolysis Part 2 BCH 340 lecture 4 Regulation of Glycolysis There are three steps in glycolysis that have enzymes which regulate the flux of glycolysis These enzymes catalyzes irreversible reactions of
Glucose is the only source of energy in red blood cells. Under starvation conditions ketone bodies become a source of energy for the brain
 Glycolysis 4 / The Text :- Some Points About Glucose Glucose is very soluble source of quick and ready energy. It is a relatively stable and easily transported. In mammals, the brain uses only glucose
Glycolysis 4 / The Text :- Some Points About Glucose Glucose is very soluble source of quick and ready energy. It is a relatively stable and easily transported. In mammals, the brain uses only glucose
3-Thia Fatty Acids A New Generation of Functional Lipids?
 Conference on Food Structure and Food Quality 3-Thia Fatty Acids A New Generation of Functional Lipids? Rolf K. Berge rolf.berge@med.uib.no Fatty acids- Essential cellular metabolites Concentrations must
Conference on Food Structure and Food Quality 3-Thia Fatty Acids A New Generation of Functional Lipids? Rolf K. Berge rolf.berge@med.uib.no Fatty acids- Essential cellular metabolites Concentrations must
Commentary. New mechanisms by which statins lower plasma cholesterol. Henri Brunengraber
 Commentary New mechanisms by which statins lower plasma cholesterol Henri Brunengraber Department of Nutrition Case Western Reserve University Cleveland, OH 44109 Phone: 216 368 6429 Fax: 216 368 6560
Commentary New mechanisms by which statins lower plasma cholesterol Henri Brunengraber Department of Nutrition Case Western Reserve University Cleveland, OH 44109 Phone: 216 368 6429 Fax: 216 368 6560
Exercise and insulin stimulate glucose transport
 Ca 2 and AMPK Both Mediate Stimulation of Glucose Transport by Muscle Contractions David C. Wright, Kathleen A. Hucker, John O. Holloszy, and Dong Ho Han It is now generally accepted that activation of
Ca 2 and AMPK Both Mediate Stimulation of Glucose Transport by Muscle Contractions David C. Wright, Kathleen A. Hucker, John O. Holloszy, and Dong Ho Han It is now generally accepted that activation of
Lecture 29: Membrane Transport and metabolism
 Chem*3560 Lecture 29: Membrane Transport and metabolism Insulin controls glucose uptake Adipose tissue and muscles contain a passive glucose transporter GluT4 which takes up glucose from blood. (This is
Chem*3560 Lecture 29: Membrane Transport and metabolism Insulin controls glucose uptake Adipose tissue and muscles contain a passive glucose transporter GluT4 which takes up glucose from blood. (This is
Cellular Respiration Other Metabolites & Control of Respiration. AP Biology
 Cellular Respiration Other Metabolites & Control of Respiration Cellular respiration: Beyond glucose: Other carbohydrates: Glycolysis accepts a wide range of carbohydrates fuels. polysaccharides glucose
Cellular Respiration Other Metabolites & Control of Respiration Cellular respiration: Beyond glucose: Other carbohydrates: Glycolysis accepts a wide range of carbohydrates fuels. polysaccharides glucose
CHY2026: General Biochemistry. Lipid Metabolism
 CHY2026: General Biochemistry Lipid Metabolism Lipid Digestion Lipid Metabolism Fats (triglycerides) are high metabolic energy molecules Fats yield 9.3 kcal of energy (carbohydrates and proteins 4.1 kcal)
CHY2026: General Biochemistry Lipid Metabolism Lipid Digestion Lipid Metabolism Fats (triglycerides) are high metabolic energy molecules Fats yield 9.3 kcal of energy (carbohydrates and proteins 4.1 kcal)
Modifications of Pyruvate Handling in Health and Disease Prof. Mary Sugden
 Modifications of Handling Modifications of Handling Centre for Diabetes and Metabolic Medicine Institute of Cell and Molecular Science Barts and the London School of Medicine and Dentistry 1 Potential
Modifications of Handling Modifications of Handling Centre for Diabetes and Metabolic Medicine Institute of Cell and Molecular Science Barts and the London School of Medicine and Dentistry 1 Potential
Pathogenesis of Diabetes Mellitus
 Pathogenesis of Diabetes Mellitus Young-Bum Kim, Ph.D. Associate Professor of Medicine Harvard Medical School Definition of Diabetes Mellitus a group of metabolic diseases characterized by hyperglycemia
Pathogenesis of Diabetes Mellitus Young-Bum Kim, Ph.D. Associate Professor of Medicine Harvard Medical School Definition of Diabetes Mellitus a group of metabolic diseases characterized by hyperglycemia
Exercise is an important component of the treatment
 Rapid Publication AMP-Activated Protein Kinase (AMPK) Is Activated in Muscle of Subjects With Type 2 Diabetes During Exercise Nicolas Musi, 1 Nobuharu Fujii, 1 Michael F. Hirshman, 1 Ingvar Ekberg, 2 Sven
Rapid Publication AMP-Activated Protein Kinase (AMPK) Is Activated in Muscle of Subjects With Type 2 Diabetes During Exercise Nicolas Musi, 1 Nobuharu Fujii, 1 Michael F. Hirshman, 1 Ingvar Ekberg, 2 Sven
Supplementary data Supplementary Figure 1 Supplementary Figure 2
 Supplementary data Supplementary Figure 1 SPHK1 sirna increases RANKL-induced osteoclastogenesis in RAW264.7 cell culture. (A) RAW264.7 cells were transfected with oligocassettes containing SPHK1 sirna
Supplementary data Supplementary Figure 1 SPHK1 sirna increases RANKL-induced osteoclastogenesis in RAW264.7 cell culture. (A) RAW264.7 cells were transfected with oligocassettes containing SPHK1 sirna
Lecture 36. Key Concepts. Overview of lipid metabolism. Reactions of fatty acid oxidation. Energy yield from fatty acid oxidation
 Lecture 36 Lipid Metabolism 1 Fatty Acid Oxidation Ketone Bodies Key Concepts Overview of lipid metabolism Reactions of fatty acid oxidation Energy yield from fatty acid oxidation Formation of ketone bodies
Lecture 36 Lipid Metabolism 1 Fatty Acid Oxidation Ketone Bodies Key Concepts Overview of lipid metabolism Reactions of fatty acid oxidation Energy yield from fatty acid oxidation Formation of ketone bodies
number Done by Corrected by Doctor Nayef Karadsheh
 number 13 Done by Asma Karameh Corrected by Saad hayek Doctor Nayef Karadsheh Gluconeogenesis This lecture covers gluconeogenesis with aspects of: 1) Introduction to glucose distribution through tissues.
number 13 Done by Asma Karameh Corrected by Saad hayek Doctor Nayef Karadsheh Gluconeogenesis This lecture covers gluconeogenesis with aspects of: 1) Introduction to glucose distribution through tissues.
Metabolism of acylglycerols and sphingolipids. Martina Srbová
 Metabolism of acylglycerols and sphingolipids Martina Srbová Types of glycerolipids and sphingolipids 1. Triacylglycerols function as energy reserves adipose tissue (storage of triacylglycerol), lipoproteins
Metabolism of acylglycerols and sphingolipids Martina Srbová Types of glycerolipids and sphingolipids 1. Triacylglycerols function as energy reserves adipose tissue (storage of triacylglycerol), lipoproteins
Biosynthesis of Fatty Acids
 Biosynthesis of Fatty Acids Fatty acid biosynthesis takes place in the cytosol rather than the mitochondria and requires a different activation mechanism and different enzymes and coenzymes than fatty
Biosynthesis of Fatty Acids Fatty acid biosynthesis takes place in the cytosol rather than the mitochondria and requires a different activation mechanism and different enzymes and coenzymes than fatty
Cellular Respiration
 Cellular Respiration 1. To perform cell work, cells require energy. a. A cell does three main kinds of work: i. Mechanical work, such as the beating of cilia, contraction of muscle cells, and movement
Cellular Respiration 1. To perform cell work, cells require energy. a. A cell does three main kinds of work: i. Mechanical work, such as the beating of cilia, contraction of muscle cells, and movement
Hormones and Target Tissues
 Hormones and Target Tissues The hypothalamus is the coordination center of the endocrine system Hypothalamus is a small region of the forebrain in animals with skulls It receives and integrates nerve signals
Hormones and Target Tissues The hypothalamus is the coordination center of the endocrine system Hypothalamus is a small region of the forebrain in animals with skulls It receives and integrates nerve signals
1st half of glycolysis (5 reactions) Glucose priming get glucose ready to split phosphorylate glucose rearrangement split destabilized glucose
 Warm- Up Objective: Describe the role of in coupling the cell's anabolic and catabolic processes. Warm-up: What cellular processes produces the carbon dioxide that you exhale? 1st half of glycolysis (5
Warm- Up Objective: Describe the role of in coupling the cell's anabolic and catabolic processes. Warm-up: What cellular processes produces the carbon dioxide that you exhale? 1st half of glycolysis (5
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION FOR Liver X Receptor α mediates hepatic triglyceride accumulation through upregulation of G0/G1 Switch Gene 2 (G0S2) expression I: SUPPLEMENTARY METHODS II: SUPPLEMENTARY FIGURES
SUPPLEMENTARY INFORMATION FOR Liver X Receptor α mediates hepatic triglyceride accumulation through upregulation of G0/G1 Switch Gene 2 (G0S2) expression I: SUPPLEMENTARY METHODS II: SUPPLEMENTARY FIGURES
The Randle cycle revisited: a new head for an old hat
 Review Am J Physiol Endocrinol Metab 297: E578 E591, 2009. First published June 16, 2009; doi:10.1152/ajpendo.00093.2009. The Randle cycle revisited: a new head for an old hat Louis Hue 1 and Heinrich
Review Am J Physiol Endocrinol Metab 297: E578 E591, 2009. First published June 16, 2009; doi:10.1152/ajpendo.00093.2009. The Randle cycle revisited: a new head for an old hat Louis Hue 1 and Heinrich
SUPPLEMENTARY DATA. Supplementary Table 1. Primers used in qpcr
 Supplementary Table 1. Primers used in qpcr Gene forward primer (5'-3') reverse primer (5'-3') β-actin AGAGGGAAATCGTGCGTGAC CAATAGTGATGACCTGGCCGT Hif-p4h-2 CTGGGCAACTACAGGATAAAC GCGTCCCAGTCTTTATTTAGATA
Supplementary Table 1. Primers used in qpcr Gene forward primer (5'-3') reverse primer (5'-3') β-actin AGAGGGAAATCGTGCGTGAC CAATAGTGATGACCTGGCCGT Hif-p4h-2 CTGGGCAACTACAGGATAAAC GCGTCCCAGTCTTTATTTAGATA
OVERVIEW M ET AB OL IS M OF FR EE FA TT Y AC ID S
 LIPOLYSIS LIPOLYSIS OVERVIEW CATABOLISM OF FREE FATTY ACIDS Nonesterified fatty acids Source:- (a) breakdown of TAG in adipose tissue (b) action of Lipoprotein lipase on plasma TAG Combined with Albumin
LIPOLYSIS LIPOLYSIS OVERVIEW CATABOLISM OF FREE FATTY ACIDS Nonesterified fatty acids Source:- (a) breakdown of TAG in adipose tissue (b) action of Lipoprotein lipase on plasma TAG Combined with Albumin
Lipid metabolism. Degradation and biosynthesis of fatty acids Ketone bodies
 Lipid metabolism Degradation and biosynthesis of fatty acids Ketone bodies Fatty acids (FA) primary fuel molecules in the fat category main use is for long-term energy storage high level of energy storage:
Lipid metabolism Degradation and biosynthesis of fatty acids Ketone bodies Fatty acids (FA) primary fuel molecules in the fat category main use is for long-term energy storage high level of energy storage:
Sustained hyperleptinemia induced in normal rats by adenovirus
 PPAR is necessary for the lipopenic action of hyperleptinemia on white adipose and liver tissue Y. Lee*, X. Yu*, F. Gonzales, D. J. Mangelsdorf, May-Yun Wang*, C. Richardson **, L. A. Witters **, and R.
PPAR is necessary for the lipopenic action of hyperleptinemia on white adipose and liver tissue Y. Lee*, X. Yu*, F. Gonzales, D. J. Mangelsdorf, May-Yun Wang*, C. Richardson **, L. A. Witters **, and R.
Role of the Pyruvate
 Role of the Pyruvate Dehydrogenase Complex in the Regulation of Blood Glucose Robert A. Harris Indiana University School of Medicine Indianapolis, Indiana Kyungpook National University School of Medicine
Role of the Pyruvate Dehydrogenase Complex in the Regulation of Blood Glucose Robert A. Harris Indiana University School of Medicine Indianapolis, Indiana Kyungpook National University School of Medicine
METABOLISM Biosynthetic Pathways
 METABOLISM Biosynthetic Pathways Metabolism Metabolism involves : Catabolic reactions that break down large, complex molecules to provide energy and smaller molecules. Anabolic reactions that use ATP energy
METABOLISM Biosynthetic Pathways Metabolism Metabolism involves : Catabolic reactions that break down large, complex molecules to provide energy and smaller molecules. Anabolic reactions that use ATP energy
Under most conditions, glucose transport is the
 Rapid Publication Metabolic Stress and Altered Glucose Tr a n s p o r t Activation of AMP-Activated Protein Kinase as a Unifying Coupling Mechanism Tatsuya Hayashi, Michael F. Hirshman, Nobuharu Fujii,
Rapid Publication Metabolic Stress and Altered Glucose Tr a n s p o r t Activation of AMP-Activated Protein Kinase as a Unifying Coupling Mechanism Tatsuya Hayashi, Michael F. Hirshman, Nobuharu Fujii,
