Use of Prophylactic Growth Factors and Antimicrobials in Elderly Patients with Cancer: A
|
|
|
- Ethan Garrett
- 6 years ago
- Views:
Transcription
1 Supportive Care in Cancer Use of Prophylactic Growth Factors and Antimicrobials in Elderly Patients with Cancer: A Systematic Review of the Medicare Database Romina Sosa, Shuling Li, Julia T. Molony, Jiannong Liu, Scott Stryker, Allan J. Collins Corresponding Author Allan J Collins, MD, FACP Chronic Disease Research Group, Minneapolis Medical Research Foundation ACollins@cdrg.org
2 Supplementary Table S1. Administrative codes used to identify chemotherapy Type of Codes Codes ICD-9-CM procedure codes for administration CPT codes for administration excluding HCPCS codes for administration C8953-C8955, G0355-G0362 excluding G0356, G3001, Q0083-Q0085 ICD-9-CM diagnosis code V58.1, V66.2, V67.2 Revenue codes 0331, 0332, 0335 DRG codes 410 and 492 on inpatient claims with discharge date before October 1, 2007; and on inpatient claims with discharge date on or after October 1, 2007 HCPCS codes for chemotherapeutic A9522, A9523, A9534, A9542-A9545, C1081, C1083, agents * C1166, C1167, C1178, C9017, C9110, C9117, C9118, C9127, C9205, C9213-C9215, C9253, C9257, C9259, C9260, C9265, C9414, C9415, C9417-C9429, C9431- C9433, C9437, C9439, C9440, J0594, J7527,, J8510, J8520, J8521, J8530, J8560-J8562, J8565, J8600, J8610, J8700, J8705, J9000-J9999 (excluding J9155, J9202, J9212, J9215-J9219, J9225, J9226, J9395), Q2017, Q2024, Q2025, Q2049, S0116, S0172, S0178, S0182 CPT, Current Procedural Terminology; DRG, diagnosis-related group; HCPCS, Healthcare Common Procedure Coding System; ICD-9-CM, International Classification of Diseases, Ninth Revision, Clinical Modification. * Includes Aldesleukin, Alemtuzumab, Arsenic trioxide, Asparaginase (erwinaze), Azacitidine, BCG (intravesical) per instillation, Bendamustine, Bevacizumab (Avastin), Bleomycin, Bortezomib, Brentuximab vedotin, Busulfan, Cabazitaxel, Capecitabine, Carboplatin, Carmustine, Cetuximab, Chlorambucil (Leukeran), Cisplatin, Cladribine, Clofarabine, Cyclophosphamide, Cytarabine liposome, Dacarbazine, Dactinomycin, Daunorubicin, Denileukin diftitox, Diethylstilbestrol diphosphate, Docetaxel, Doxorubicin, Elliotts b solution, Epirubicin, Eribulin mesylate, Etoposide, Everolimus, Floxuridine, Fludarabine, Fluorouracil, Gefitinib, Gemcitabine, Gemtuzumab ozogamicin, Ibritumomab tiuxetan (Zevalin), Idarubicin,
3 Ifosfamide, Interferon alfa-2a and alfa-2b, Ipilimumab, Irinotecan, Ixabepilone, Lomustine, Mechlorethamine, Melphalan, Mesna, Methotrexate, Mitomycin, Mitoxantrone, Nelarabine, Ofatumumab, Oxaliplatin, Paclitaxel, Panitumumab, Pegaspargase, Pemetrexed, Pentostatin, Plicamycin, Porfimer sodium, Pralatrexate, Procarbazine hydrochloride, Rituximab, Romidepsin, Streptozocin, Temozolomide, Temsirolimus, Teniposide, Thiotepa, Topotecan, Tositumomab (Bexxar), Trastuzumab, Valrubicin, Vinblastine, Vincristine, and Vinorelbine.
4 Supplementary Table S2. International Classification of Diseases, Ninth Revision, Clinical Modification (ICD-9-CM) diagnosis codes used to define comorbid conditions Comorbid Conditions ICD-9-CM Diagnosis Codes Chronic kidney disease 016.0, 095.4, 189.0, 189.9, 223.0, , 250.4, 271.4, 274.1, , 403.X1, 404.X2, 404.X3, 440.1, 442.1, 447.3, 572.4, , 591, 642.1, 646.2, , , 753.2, and Atherosclerotic heart disease , V45.18, and V45.82 Cerebrovascular accidents/transient ischemic attacks Chronic obstructive pulmonary disease 491.xx-494.xx, 496.xx, 510.xx Congestive heart failure , 422.xx, 425.xx, 428.xx, 402.x1, 404.x1, 404.x3, V42.1 Diabetes mellitus 250.xx, 357.2, 362.0x, Dysrhythmia 426.xx-427.xx, V45.0, and V53.3 Gastrointestinal disorders , 530.7, 531.xx-534.xx, , , 578 Liver disease 570.xx, 571.xx, 572.1, 572.4, , V42.7, Other cardiovascular disease 420.xx-421.xx, 423.xx-424.xx, 429.xx, , V42.2, and V43.3 Peripheral vascular disease ; 447; ; 557 Anemia HIV infection 042, Thyroid disease 240.x-246.x Peptic ulcer disease 531.x-539.x Rheumatoid disease (rheumatoid arthritis) 714.x Osteoarthritis 715.x Other cancer ; ; (excluding for breast cancer, for lung cancer, and 200 and 202 for non-hodgkin lymphoma)
5 Supplementary Table S3. List of oral antimicrobial agents Antimicrobial Class Antibacterials Penicillins Cephalosporins Macrolides Tetracyclines Fluoroquinolones Aminoglycosides Sulfonamides Other Antifungals Antivirals Drug Name Amoxicillin, Dicloxacillin sodium, Amoxicillin and Clavulanate, Ampicillin Cefadroxil, Cephalexin, Cefaclor, Cefprozil, Cefuroxime Axetil, Cefdinir, Cefditoren, Cefixime, Cefpodoxime, Ceftibuten Erythromycin, Azithromycin, Clarithromycin, Fidaxomicin Demeclocycline, Doxycycline, Minocycline, Tetracycline Ciprofloxacin, Levofloxacin, Moxifloxacin, Norfloxacin, Ofloxacin, Gemifloxacin Neomycin, Paromomycin Sulfadiazine, Sulfamethoxazole-Trimethoprim Vancomycin, Telithromycin, Clindamycin, Linezolid Terbinafine, Flucytosine, Griseofulvin, Ketoconazole, Nystatin, Fluconazole, Itraconazole, Posaconazole, Voriconzaole Adefovir dipivoxil, Amantadine HCl, Acyclovir, Famciclovir, Valacyclovir, Boceprevir, Entecavir, Oseltamivir, Ribavirin, Rimantadine HCl, Telaprevir, Valganciclovir
6 Supplementary Table S4. Distribution of chemotherapy regimens or top five most frequently used agents, by cancer type and febrile neutropenia risk category of chemotherapy regimen Cancer Type, Risk Category, or Agents n % Breast cancer (n = 9110) High risk (n = 923) Dose dense AC + sequential T (doxorubicin, cyclophosphamide, paclitaxel) TAC (docetaxel, doxorubicin, cyclophosphamide) Docetaxel + trastuzumab AT (doxorubicin, paclitaxel) AT (doxorubicin, docetaxel) ^ ^ Docetaxel every 14 days * * Intermediate risk (n = 908) Paclitaxel every 21 days CMF classic (cyclophosphamide, methotrexate, fluorouracil) Paclitaxel, Trastuzumab Docetaxel every 21 days FEC (fluorouracil/epirubicin/cyclophosphamide) Other risk (n = 7279) Top five agents by number of patients received during the course Cyclophosphamide/docetaxel Bevacizumab Capacitabine Trastuzumab/docetaxel/carboplatin Trastuzumab Lung cancer (n = 12,032) High risk (n = 37) Topotecan Intermediate risk (n = 5130) Carboplatin/paclitaxel Etoposide/carboplatin Cisplatin/etoposide Cisplatin/vinorelbine Docetaxel Cisplatin/docetaxel Cisplatin/paclitaxel Cisplatin/irinotecan Other risk (n = 6865) Top five agents by number of patients received during the course Docetaxel/carboplatin Pemetrexed Carboplatin/pemetrexed Gemcitabine/carboplatin Paclitaxel/carboplatin/bevacizumab
7 Non-Hodgkin lymphoma (n = 6306) High risk (n = 1035) CHOP (cyclophosphamide, doxorubicin, vincristine, prednisone) + rituximab HyperCVAD (cyclophosphamide, vincristine, doxorubicin, dexamethasone) + rituximab RICE (rituximab, ifosfamide, carboplatin, etoposide) * * MINE (mesna, ifosfamide, novantrone, etoposide) * * CHOP-14 (cyclophosphamide, doxorubicin, vincristine, prednisone) * * ESHAP (etoposide, methylprednisolone, cisplatin, cytarabine) * * Intermediate risk (n = 24) FM (fludarabine, mitoxantrone) GDP (gemcitabine, dexamethasone, cisplatin) * * EPOCH (etoposide, prednisone, vincristine, cyclophosphamide, doxorubicin) * * GDP (gemcitabine, dexamethasone, cisplatin)+rituximab * * Other risk (n = 5247) Top five agents by number of patients received during the course n % Rituximab Cyclophosphamide/rituximab/vincristine Rituximab/bendamustine Bevacizumab Rituximab/fludarabine * Values for cells with ten or fewer patients are suppressed. ^ Value is suppressed to avoid deriving another cell with ten or fewer patients.
Primary malignant neoplasms, not lymphatic or hematopoietic. Secondary malignant neoplasms (i.e.metastatic) Malignant neoplasm, unknown site
 Supplementary Table 1. ICD-9-CM codes used to define cancer ICD-9 Diagnosis code 140.xx-172.xx 174.xx-195.xx 196.xx 198.xx 199.xx 200.xx-208.xx Description Primary malignant neoplasms, not lymphatic or
Supplementary Table 1. ICD-9-CM codes used to define cancer ICD-9 Diagnosis code 140.xx-172.xx 174.xx-195.xx 196.xx 198.xx 199.xx 200.xx-208.xx Description Primary malignant neoplasms, not lymphatic or
MEDICAL NECESSITY GUIDELINE
 PAGE: 1 of 10 IMPORTANT REMINDER This Clinical Policy has been developed by appropriately experienced and licensed health care professionals based on a thorough review and consideration of generally accepted
PAGE: 1 of 10 IMPORTANT REMINDER This Clinical Policy has been developed by appropriately experienced and licensed health care professionals based on a thorough review and consideration of generally accepted
 Exhibit B United States Patent Application 20020012663 Kind Code A1 Waksal, Harlan W. January 31, 2002 Treatment of refractory human tumors with epidermal growth factor receptor antagonists Abstract A
Exhibit B United States Patent Application 20020012663 Kind Code A1 Waksal, Harlan W. January 31, 2002 Treatment of refractory human tumors with epidermal growth factor receptor antagonists Abstract A
Guidelines on Chemotherapy-induced Nausea and Vomiting in Pediatric Cancer Patients
 Guidelines on Chemotherapy-induced Nausea Vomiting in Pediatric Cancer Patients COG Supportive Care Endorsed Guidelines Click here to see all the COG Supportive Care Endorsed Guidelines. DISCLAIMER For
Guidelines on Chemotherapy-induced Nausea Vomiting in Pediatric Cancer Patients COG Supportive Care Endorsed Guidelines Click here to see all the COG Supportive Care Endorsed Guidelines. DISCLAIMER For
ELECTRONIC HEALTH RECORD (EHR) ENHANCEMENTS FOR MARCH 15, 2016 SUMMARY
 ELECTRONIC HEALTH RECORD (EHR) ENHANCEMENTS FOR MARCH 15, 2016 SUMMARY Problem Opening PACS Images on ipads or ibooks has Been Fixed Changes have been made in PROD to enable user credentials to be passed
ELECTRONIC HEALTH RECORD (EHR) ENHANCEMENTS FOR MARCH 15, 2016 SUMMARY Problem Opening PACS Images on ipads or ibooks has Been Fixed Changes have been made in PROD to enable user credentials to be passed
Objective: To provide a standard procedure for the recycling of unused medication and the disposal of medicines across all BCPFT Hospital sites.
 WARDS/DEPARTMENTS By: 0 01/09/019 1 of 3 Objective: To provide a standard procedure for the recycling of unused medication and the disposal of medicines across all BCPFT Hospital sites. Scope: All Black
WARDS/DEPARTMENTS By: 0 01/09/019 1 of 3 Objective: To provide a standard procedure for the recycling of unused medication and the disposal of medicines across all BCPFT Hospital sites. Scope: All Black
Committee Approval Date: December 12, 2014 Next Review Date: July 2015
 Medication Policy Manual Policy No: dru378 Topic: Akynzeo, netupitant/palonosetron Date of Origin: December 12, 2014 Committee Approval Date: December 12, 2014 Next Review Date: July 2015 Effective Date:
Medication Policy Manual Policy No: dru378 Topic: Akynzeo, netupitant/palonosetron Date of Origin: December 12, 2014 Committee Approval Date: December 12, 2014 Next Review Date: July 2015 Effective Date:
West of Scotland Cancer Network Guideline for Managing Chemotherapy Induced Nausea and Vomiting
 West of Scotland Cancer Network Guideline for Managing Chemotherapy Induced Nausea and Vomiting Definitions Acute nausea and vomiting Delayed nausea and vomiting Anticipatory nausea and vomiting Initial
West of Scotland Cancer Network Guideline for Managing Chemotherapy Induced Nausea and Vomiting Definitions Acute nausea and vomiting Delayed nausea and vomiting Anticipatory nausea and vomiting Initial
SCI. SickKids-Caribbean Initiative Enhancing Capacity for Care in Paediatric Cancer and Blood Disorders
 1.0 Introduction The (SCI) is a not-for-profit collaboration between the Hospital for Sick Children (SickKids), Toronto, Canada, and seven Caribbean health care institutions across six countries that strive
1.0 Introduction The (SCI) is a not-for-profit collaboration between the Hospital for Sick Children (SickKids), Toronto, Canada, and seven Caribbean health care institutions across six countries that strive
Part B payment for drugs in Medicare 0
 Part B payment for drugs in Medicare 0 Introduction: The recent pilot proposal 1 on Part B drug payment from the Center for Medicare and Medicaid Innovation of CMS has met strong resistance 2 from the
Part B payment for drugs in Medicare 0 Introduction: The recent pilot proposal 1 on Part B drug payment from the Center for Medicare and Medicaid Innovation of CMS has met strong resistance 2 from the
Vivida Health Specialty Pharmacy Drugs (Injectable) Prior-Authorization Requirements Effective 1/1/19
 Vivida Health Specialty Pharmacy Drugs (Injectable) Prior-Authorization Requirements Effective 1/1/19 All Non-Par Provider Requests Requires Authorization Regardless of Service J0178 J0180 J0202 J0205
Vivida Health Specialty Pharmacy Drugs (Injectable) Prior-Authorization Requirements Effective 1/1/19 All Non-Par Provider Requests Requires Authorization Regardless of Service J0178 J0180 J0202 J0205
Co-morbidities in Elderly Breast and Lung Cancer Patients who receive Chemotherapy of NCCN defined Intermediate Risk of Febrile Neutropenia
 Co-morbidities in Elderly Breast and Lung Cancer Patients who receive Chemotherapy of NCCN defined Intermediate Risk of Febrile Neutropenia Authors: John H. Page, Shuling Li, Julia Molony, Romina Sosa,
Co-morbidities in Elderly Breast and Lung Cancer Patients who receive Chemotherapy of NCCN defined Intermediate Risk of Febrile Neutropenia Authors: John H. Page, Shuling Li, Julia Molony, Romina Sosa,
Subject: Palonosetron Hydrochloride (Aloxi )
 09-J0000-87 Original Effective Date: 02/15/09 Reviewed: 07/09/14 Revised: 03/15/18 Subject: Palonosetron Hydrochloride (Aloxi ) THIS MEDICAL COVERAGE GUIDELINE IS NOT AN AUTHORIZATION, CERTIFICATION, EXPLANATION
09-J0000-87 Original Effective Date: 02/15/09 Reviewed: 07/09/14 Revised: 03/15/18 Subject: Palonosetron Hydrochloride (Aloxi ) THIS MEDICAL COVERAGE GUIDELINE IS NOT AN AUTHORIZATION, CERTIFICATION, EXPLANATION
Managements of Chemotherpay Induded Nausea and Vomiting
 REVIEW ARTICLE Managements of Chemotherpay Induded Nausea and Vomiting Department of Surgery, The Catholic University of Korea Sung Geun Kim 23 24 Sung Geun Kim Korean Journal of Clinical Oncology Summer
REVIEW ARTICLE Managements of Chemotherpay Induded Nausea and Vomiting Department of Surgery, The Catholic University of Korea Sung Geun Kim 23 24 Sung Geun Kim Korean Journal of Clinical Oncology Summer
Part B payment for drugs in Medicare: Phase 1 of CMS s proposed pilot and its impact on oncology care
 Part B payment for drugs in Medicare: Phase 1 of CMS s proposed pilot and its impact on oncology care Raina H. Jain, Stephen M. Schleicher, Coral L. Atoria, Peter B. Bach Executive Summary The recent pilot
Part B payment for drugs in Medicare: Phase 1 of CMS s proposed pilot and its impact on oncology care Raina H. Jain, Stephen M. Schleicher, Coral L. Atoria, Peter B. Bach Executive Summary The recent pilot
GUIDELINES FOR ANTIEMETIC USE IN ONCOLOGY SUMMARY CLASSIFICATION
 GUIDELINES FOR ANTIEMETIC USE IN ONCOLOGY SUMMARY More than half of all cancer patients experience nausea or vomiting during the course of their treatment. If nausea or vomiting becomes severe enough,
GUIDELINES FOR ANTIEMETIC USE IN ONCOLOGY SUMMARY More than half of all cancer patients experience nausea or vomiting during the course of their treatment. If nausea or vomiting becomes severe enough,
Part B payment for drugs in Medicare 0
 Part B payment for drugs in Medicare 0 Introduction: The recent pilot proposal 1 on Part B drug payment from the Center for Medicare and Medicaid Innovation of CMS has met strong resistance 2 from the
Part B payment for drugs in Medicare 0 Introduction: The recent pilot proposal 1 on Part B drug payment from the Center for Medicare and Medicaid Innovation of CMS has met strong resistance 2 from the
Guidelines for the Use of Anti-Emetics with Chemotherapy
 Guidelines for the Use of Anti-Emetics with The purpose of this document is to provide guidance on the rational use of anti-emetics for prevention and treatment of chemotherapy-induced nausea and vomiting
Guidelines for the Use of Anti-Emetics with The purpose of this document is to provide guidance on the rational use of anti-emetics for prevention and treatment of chemotherapy-induced nausea and vomiting
Emetogenicity level 1. Emetogenicity level 2
 Emetogenicity level 1 15 mins Pre-Chemo Maxalon 10mg po During chemo and Post Chemo 3 days Maxalon10mg po 8 hourly Increase Maxalon 20mg po 8 hourly Change to Cyclizine 50mg po 8 hourly 3 days If nausea
Emetogenicity level 1 15 mins Pre-Chemo Maxalon 10mg po During chemo and Post Chemo 3 days Maxalon10mg po 8 hourly Increase Maxalon 20mg po 8 hourly Change to Cyclizine 50mg po 8 hourly 3 days If nausea
Protocol Number Tumour Group Protocol Name on NCCP website 22/02/ Lung Afatinib Monotherapy 244 Gastrointestinal Regorafenib Monotherapy
 Last Updated 22-Feb-18 Date of last update Protocol Number Tumour Group Protocol Name on NCCP website 22/02/2018 221 Afatinib Monotherapy 244 Gastrointestinal Regorafenib Monotherapy 249 Gynaecology Intrathecal
Last Updated 22-Feb-18 Date of last update Protocol Number Tumour Group Protocol Name on NCCP website 22/02/2018 221 Afatinib Monotherapy 244 Gastrointestinal Regorafenib Monotherapy 249 Gynaecology Intrathecal
Subject: Fosnetupitant-Palonosetron (Akynzeo) IV
 09-J3000-01 Original Effective Date: 06/15/18 Reviewed: 05/09/18 Revised: 01/01/19 Subject: Fosnetupitant-Palonosetron (Akynzeo) IV THIS MEDICAL COVERAGE GUIDELINE IS NOT AN AUTHORIZATION, CERTIFICATION,
09-J3000-01 Original Effective Date: 06/15/18 Reviewed: 05/09/18 Revised: 01/01/19 Subject: Fosnetupitant-Palonosetron (Akynzeo) IV THIS MEDICAL COVERAGE GUIDELINE IS NOT AN AUTHORIZATION, CERTIFICATION,
Protocol Number Intrathecal Methotrexate for CNS 01/02/2018 Prophylaxis in GTN Gynaecology 249
 Last updated Feb 9, 2018 Revision due Protocol Name on NCCP website Tumour Group Protocol Number Intrathecal Methotrexate for CNS 01/02/2018 Prophylaxis in GTN Gynaecology 249 Two Day Etoposide CISplatin
Last updated Feb 9, 2018 Revision due Protocol Name on NCCP website Tumour Group Protocol Number Intrathecal Methotrexate for CNS 01/02/2018 Prophylaxis in GTN Gynaecology 249 Two Day Etoposide CISplatin
DOXOrubicin, Cyclophosphamide (AC 60/600) 21 day followed by weekly PACLitaxel (80) Therapy (AC-T) 261 CARBOplatin (AUC4-6) Monotherapy-21 days
 Last updated Oct 17, 2018 Tumour Group Protocol Number Protocol Name on NCCP website Breast 200 Trastuzumab (IV) Monotherapy 21 days 201 Trastuzumab (IV) Monotherapy 7 days 202 DOCEtaxel Monotherapy 100mg/m2
Last updated Oct 17, 2018 Tumour Group Protocol Number Protocol Name on NCCP website Breast 200 Trastuzumab (IV) Monotherapy 21 days 201 Trastuzumab (IV) Monotherapy 7 days 202 DOCEtaxel Monotherapy 100mg/m2
Haematology, Oncology and Palliative Care Directorate.
 Anticancer Treatment for Administration on the Somerset Mobile Chemotherapy Unit The table below details the suitability of different types of anticancer treatment for administration on the Somerset Mobile
Anticancer Treatment for Administration on the Somerset Mobile Chemotherapy Unit The table below details the suitability of different types of anticancer treatment for administration on the Somerset Mobile
Description The following are synthetic cannabinoids requiring prior authorization: dronabinol (Marinol, Syndros ), nabilone (Cesamet )
 Clinical Policy: Nabilone (Cesamet), Dronabinol (Marinol, Syndros) Reference Number: CP.CPA.242 Effective Date: 11.16.16 Last Review Date: 11.17 Line of Business: Commercial Revision Log See Important
Clinical Policy: Nabilone (Cesamet), Dronabinol (Marinol, Syndros) Reference Number: CP.CPA.242 Effective Date: 11.16.16 Last Review Date: 11.17 Line of Business: Commercial Revision Log See Important
Guideline Update on Antiemetics
 Guideline Update on Antiemetics Clinical Practice Guideline Special Announcements Please check www.asco.org/guidelines/antiemetics for current FDA alert(s) and safety announcement(s) on antiemetics 2 Introduction
Guideline Update on Antiemetics Clinical Practice Guideline Special Announcements Please check www.asco.org/guidelines/antiemetics for current FDA alert(s) and safety announcement(s) on antiemetics 2 Introduction
Guideline for Classification of the Acute Emetogenic Potential of Antineoplastic Medication in Pediatric Cancer Patients
 Guideline for Classification of the Acute Emetogenic Potential of Antineoplastic Medication in Pediatric Cancer Patients POGO Antineoplastic Induced Nausea and Vomiting Guideline Development Panel: L.
Guideline for Classification of the Acute Emetogenic Potential of Antineoplastic Medication in Pediatric Cancer Patients POGO Antineoplastic Induced Nausea and Vomiting Guideline Development Panel: L.
Fee-for-Service Pharmacy Provider Notice #215 ** January 2016 PDL Changes ** Existing Drug Classes
 Fee-for-Service Pharmacy Provider Notice #215 ** January 2016 PDL Changes ** December 19, 2016 Please be advised that the Department for Medicaid Services (DMS) is making changes to the Kentucky Medicaid
Fee-for-Service Pharmacy Provider Notice #215 ** January 2016 PDL Changes ** December 19, 2016 Please be advised that the Department for Medicaid Services (DMS) is making changes to the Kentucky Medicaid
Hodgkin and Non-Hodgkin Lymphoma (LYM) Post-Infusion Data
 Hodgkin and Non-Hodgkin Lymphoma (LYM) Post-Infusion Data Registry Use Only Sequence Number: Date Received: CIBMTR Center Number: CIBMTR Research ID: Event date: / / Visit 100 day 6 months 1 year 2 years
Hodgkin and Non-Hodgkin Lymphoma (LYM) Post-Infusion Data Registry Use Only Sequence Number: Date Received: CIBMTR Center Number: CIBMTR Research ID: Event date: / / Visit 100 day 6 months 1 year 2 years
General Authorization Criteria for ALL Agents and Indications:
 Neulasta (peg-filgrastim; G-CSF) Neupogen (filgrastim; G-CSF) Neulasta Onpro (peg-filgrastim; G-CSF) Leukine (sargramostim; GM-CSF) General Authorization Criteria for ALL Agents and Indications: Prescribed
Neulasta (peg-filgrastim; G-CSF) Neupogen (filgrastim; G-CSF) Neulasta Onpro (peg-filgrastim; G-CSF) Leukine (sargramostim; GM-CSF) General Authorization Criteria for ALL Agents and Indications: Prescribed
Adverse effects of anticancer drugs (Antimetabolites agents, Alkylating agents, Antimicrotubule agents, Miscellaneous agents, Immune therapies and
 35 Adverse effects of anticancer drugs (Antimetabolites agents, Alkylating agents, Antimicrotubule agents, Miscellaneous agents, Immune therapies and Biologically directed therapies ) 1 1- Nausea and vomiting
35 Adverse effects of anticancer drugs (Antimetabolites agents, Alkylating agents, Antimicrotubule agents, Miscellaneous agents, Immune therapies and Biologically directed therapies ) 1 1- Nausea and vomiting
Subject: NK-1 receptor antagonist injectable therapy (Emend, Cinvanti, Varubi )
 09-J2000-60 Original Effective Date: 06/15/16 Reviewed: 04/11/18 Revised: 01/01/19 Subject: NK-1 receptor antagonist injectable therapy (Emend, Cinvanti, Varubi ) THIS MEDICAL COVERAGE GUIDELINE IS NOT
09-J2000-60 Original Effective Date: 06/15/16 Reviewed: 04/11/18 Revised: 01/01/19 Subject: NK-1 receptor antagonist injectable therapy (Emend, Cinvanti, Varubi ) THIS MEDICAL COVERAGE GUIDELINE IS NOT
MASCC Guidelines for Antiemetic control: An update
 MASCC / ISOO 17 th International Symposium Supportive Care in Cancer June 30 July 2, 2005 / Geneva, Switzerland MASCC Guidelines for Antiemetic control: An update Sussanne Börjeson, RN, PhD Linköping University,
MASCC / ISOO 17 th International Symposium Supportive Care in Cancer June 30 July 2, 2005 / Geneva, Switzerland MASCC Guidelines for Antiemetic control: An update Sussanne Börjeson, RN, PhD Linköping University,
APPHON/ROPPHA Guideline for the Prevention and Management of Chemotherapy Induced Nausea and Vomiting in Children with Cancer
 APPHON/ROPPHA Guideline for the Prevention and Management of Chemotherapy Induced Nausea and Vomiting in Children with Cancer 5850/5980 University Avenue, PO Box 9700, Halifax, N.S. B3K 6R8 PEDIATRIC HEMATOLOGY/ONCOLOGY
APPHON/ROPPHA Guideline for the Prevention and Management of Chemotherapy Induced Nausea and Vomiting in Children with Cancer 5850/5980 University Avenue, PO Box 9700, Halifax, N.S. B3K 6R8 PEDIATRIC HEMATOLOGY/ONCOLOGY
Current and Emerging Therapeutic Options in the Management of Chemotherapy-Induced Nausea and Vomiting (CINV) Objectives
 Current and Emerging Therapeutic Options in the Management of Chemotherapy-Induced Nausea and Vomiting (CINV) Susan Urba, M.D. University of Michigan Comprehensive Cancer Center Objectives Mechanisms of
Current and Emerging Therapeutic Options in the Management of Chemotherapy-Induced Nausea and Vomiting (CINV) Susan Urba, M.D. University of Michigan Comprehensive Cancer Center Objectives Mechanisms of
Pharmacy Prior Authorization Colony Stimulating Factor (CSF)/Myeloid Growth Factor (MGF) Clinical Guideline
 Neulasta (peg-filgrastim; G-CSF) Neupogen (filgrastim; G-CSF) Neulasta Onpro (peg-filgrastim; G-CSF) Leukine (sargramostim; GM-CSF) General Authorization Criteria for ALL Agents and Indications: Prescribed
Neulasta (peg-filgrastim; G-CSF) Neupogen (filgrastim; G-CSF) Neulasta Onpro (peg-filgrastim; G-CSF) Leukine (sargramostim; GM-CSF) General Authorization Criteria for ALL Agents and Indications: Prescribed
Adult Intravenous Systemic Anticancer Therapy (SACT) Section A. SUMMARY of SCHEME QIPP Reference
 CA2 Nationally standardised Dose banding for Adult Intravenous Anticancer Therapy (SACT) Scheme Name CA2: Nationally Standardised Dose Banding for Adult Intravenous Systemic Anticancer Therapy (SACT) Section
CA2 Nationally standardised Dose banding for Adult Intravenous Anticancer Therapy (SACT) Scheme Name CA2: Nationally Standardised Dose Banding for Adult Intravenous Systemic Anticancer Therapy (SACT) Section
DRUG PROPERTIES YOU NEED TO KNOW
 Dr. Janet Fitzakerley Summer 2013 Med 6541 Hematopoiesis and Host Defences jfitzake@d.umn.edu www.d.umn.edu/~jfitzake Page 1 of 11 DRUG PROPERTIES YOU NEED TO KNOW 1. Mechanism of action a. chemical class
Dr. Janet Fitzakerley Summer 2013 Med 6541 Hematopoiesis and Host Defences jfitzake@d.umn.edu www.d.umn.edu/~jfitzake Page 1 of 11 DRUG PROPERTIES YOU NEED TO KNOW 1. Mechanism of action a. chemical class
VI.2 Elements for a Public Summary VI.2.1 Overview of Disease Epidemiology Acute Nausea and Vomiting (N&V) Etiologies:
 VI.2 Elements for a Public Summary VI.2.1 Overview of Disease Epidemiology Acute Nausea and Vomiting (N&V) Incidence: The incidence of acute and delayed N&V was investigated in highly and moderately emetogenic
VI.2 Elements for a Public Summary VI.2.1 Overview of Disease Epidemiology Acute Nausea and Vomiting (N&V) Incidence: The incidence of acute and delayed N&V was investigated in highly and moderately emetogenic
COST CONSIDERATIONS Union for International Cancer Control 2014 Review of Cancer Medicines on the WHO List of Essential Medicines!!!!!!!!!
 UICCEMLCostingScenarios BackoftheEnvelope Calculations PreparedforWorkingGroupSession:19621November2014,Geneva MethodsSummary We have chosen a conservative approach, calculating cost per vial. We have
UICCEMLCostingScenarios BackoftheEnvelope Calculations PreparedforWorkingGroupSession:19621November2014,Geneva MethodsSummary We have chosen a conservative approach, calculating cost per vial. We have
Pharmacy Prior Authorization Colony Stimulating Factors Clinical Guideline. Neulasta Onpro (peg-filgrastim; G-CSF) Leukine (sargramostim; GM-CSF)
 Neulasta (peg-filgrastim; G-CSF) Neupogen (filgrastim; G-CSF) Neulasta Onpro (peg-filgrastim; G-CSF) Leukine (sargramostim; GM-CSF) General Authorization Criteria for ALL Agents and Indications: Prescribed
Neulasta (peg-filgrastim; G-CSF) Neupogen (filgrastim; G-CSF) Neulasta Onpro (peg-filgrastim; G-CSF) Leukine (sargramostim; GM-CSF) General Authorization Criteria for ALL Agents and Indications: Prescribed
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: Reference Number: CP.CPA.223 Effective Date: 11.16.16 Last Review Date: 11.17 Line of Business: Commercial Revision Log See Important Reminder at the end of this policy for important regulatory
Clinical Policy: Reference Number: CP.CPA.223 Effective Date: 11.16.16 Last Review Date: 11.17 Line of Business: Commercial Revision Log See Important Reminder at the end of this policy for important regulatory
Adjuvant/Curative/Neo-adjuvant High Grade and Burkitt s Lymphoma Regimens. High Grade Lymphoma
 Adjuvant/Curative/Neo-adjuvant High Grade and Burkitt s Lymphoma Regimens The following table lists the evidence-informed regimens (both IV and non-iv) for high grade and Burkitt s lymphoma used in the
Adjuvant/Curative/Neo-adjuvant High Grade and Burkitt s Lymphoma Regimens The following table lists the evidence-informed regimens (both IV and non-iv) for high grade and Burkitt s lymphoma used in the
The AngCN Antiemetic Guidelines
 The AngCN Antiemetic Guidelines Guidelines for the Management of Nausea and Vomiting in Adult Patients Receiving Chemotherapy and/or Radiotherapy AngCN Document Reference: AngCN-CCG-C31 CONTENTS 1.0 Introduction
The AngCN Antiemetic Guidelines Guidelines for the Management of Nausea and Vomiting in Adult Patients Receiving Chemotherapy and/or Radiotherapy AngCN Document Reference: AngCN-CCG-C31 CONTENTS 1.0 Introduction
1. Purpose Documentation Description.1 4. References 8 5. Cross-References.8 6. Development of the guideline.8
 CLINICAL GUIDELINE For use in (clinical areas): For use by (staff groups): For use for (patients): Document owner: Status: Oncology/Haematology Unit (excluding Paediatrics) Oncologists, Haematologists,
CLINICAL GUIDELINE For use in (clinical areas): For use by (staff groups): For use for (patients): Document owner: Status: Oncology/Haematology Unit (excluding Paediatrics) Oncologists, Haematologists,
Prevention and Management of chemo-and radiotherapy-induced nausea and vomiting
 Prevention and Management of chemo-and radiotherapy-induced nausea and vomiting Focusing on the updated MASCC/ESMO guidelines Karin Jordan Department of Hematology and Oncology, University of Heidelberg
Prevention and Management of chemo-and radiotherapy-induced nausea and vomiting Focusing on the updated MASCC/ESMO guidelines Karin Jordan Department of Hematology and Oncology, University of Heidelberg
ICON Formulary - October 2018 Legend - ICON Protocols Essential (previously Standard), Core, Enhanced Core, Enhanced Enhanced
 ICON Formulary - October 2018 Legend - ICON Protocols Essential (previously Standard), Core, Enhanced Core, Enhanced Enhanced Class Medicine Name Nappi Strength Form Size Route Abiraterone Acetate ZYTIGA
ICON Formulary - October 2018 Legend - ICON Protocols Essential (previously Standard), Core, Enhanced Core, Enhanced Enhanced Class Medicine Name Nappi Strength Form Size Route Abiraterone Acetate ZYTIGA
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Zofran, Zuplenz) Reference Number: CP.CPA.173 Effective Date: 11.16.16 Last Review Date: 11.17 Line of Business: Medicaid Medi-Cal Revision Log See Important Reminder at the end of this
Clinical Policy: (Zofran, Zuplenz) Reference Number: CP.CPA.173 Effective Date: 11.16.16 Last Review Date: 11.17 Line of Business: Medicaid Medi-Cal Revision Log See Important Reminder at the end of this
To help doctors give their patients the best possible care, the American
 Patient Information Resources from ASCO What to Know ASCO s Guideline on Preventing Vomiting Caused by Cancer Treatment SEPTEMBER 2011 KEY MESSAGES The risk of nausea and vomiting depends on the specific
Patient Information Resources from ASCO What to Know ASCO s Guideline on Preventing Vomiting Caused by Cancer Treatment SEPTEMBER 2011 KEY MESSAGES The risk of nausea and vomiting depends on the specific
ANTIEMETICS UTILIZATION MANAGEMENT CRITERIA
 ANTIEMETICS [Akynzeo, Anzemet, Cesamet, Emend, Sancuso, Varubi, Zuplenz ] DRUG CLASS: UTILIZATION MANAGEMENT CRITERIA 5-HT 3 Receptor Antagonists 5-HT 3 Receptor Antagonist and Substance P/Neurokinin (NK
ANTIEMETICS [Akynzeo, Anzemet, Cesamet, Emend, Sancuso, Varubi, Zuplenz ] DRUG CLASS: UTILIZATION MANAGEMENT CRITERIA 5-HT 3 Receptor Antagonists 5-HT 3 Receptor Antagonist and Substance P/Neurokinin (NK
DRUG EXTRAVASATION. Vesicants. Irritants
 DRUG EXTRAVASATION Vesicants Irritants Vesicants Antineoplastic drugs Amsacrine Dactinomycin Daunorubicin Docetaxel (rare) Doxorubicin Epirubicin Idarubicin Mechlorethamine Mitomycin Oxaliplatin (rare)
DRUG EXTRAVASATION Vesicants Irritants Vesicants Antineoplastic drugs Amsacrine Dactinomycin Daunorubicin Docetaxel (rare) Doxorubicin Epirubicin Idarubicin Mechlorethamine Mitomycin Oxaliplatin (rare)
2019 PHP PRIMARY CARE INCENTIVE
 Primary Care Physicians (PCP) of Physicians Health Network (PHN) may be eligible for an incentive payment in accordance with this PHP PCP Incentive (hereinafter referred to as the PCP Incentive ). As described
Primary Care Physicians (PCP) of Physicians Health Network (PHN) may be eligible for an incentive payment in accordance with this PHP PCP Incentive (hereinafter referred to as the PCP Incentive ). As described
Antiemetic Guidelines. Guidelines for the Management of Nausea and Vomiting in Adult Patients Receiving Chemotherapy and/or Radiotherapy
 Antiemetic Guidelines Guidelines for the Management of Nausea and Vomiting in Adult Patients Receiving Chemotherapy and/or Radiotherapy For approvals and version control see Document Management Record
Antiemetic Guidelines Guidelines for the Management of Nausea and Vomiting in Adult Patients Receiving Chemotherapy and/or Radiotherapy For approvals and version control see Document Management Record
Northern Cancer Alliance
 Northern Cancer Alliance Anti-emetic Guidelines for Chemotherapy Induced Nausea and Vomiting (CINV) Adult Oncology & Haematology Document Control Document Title: Antiemetic Guidelines for CINV NESCN v2.2
Northern Cancer Alliance Anti-emetic Guidelines for Chemotherapy Induced Nausea and Vomiting (CINV) Adult Oncology & Haematology Document Control Document Title: Antiemetic Guidelines for CINV NESCN v2.2
1 The Cancer Programs Regulation (AR 242/98) is amended by this Regulation.
 Alberta Regulation 18/2005 Cancer Programs Act AMENDMENT REGULATION Filed: February 22, 2005 For information only: Made by the Minister of Health and Wellness (M.O. 9/2005) on February 17, 2005 pursuant
Alberta Regulation 18/2005 Cancer Programs Act AMENDMENT REGULATION Filed: February 22, 2005 For information only: Made by the Minister of Health and Wellness (M.O. 9/2005) on February 17, 2005 pursuant
AD regimen, 576 adrenal steroid inhibitor. aminoglutethimide, 23 25
 A 640 Index Index abiraterone acetate, 5 7 Abraxane. See albuminbound paclitaxel ABVD regimen, 57, 540 ABV regimen, 519 520 AC docetaxel regimen, 476, 478 acetaminophen busulfan and, 71 AC regimen, 475,
A 640 Index Index abiraterone acetate, 5 7 Abraxane. See albuminbound paclitaxel ABVD regimen, 57, 540 ABV regimen, 519 520 AC docetaxel regimen, 476, 478 acetaminophen busulfan and, 71 AC regimen, 475,
Acute Lymphocytic Leukemia
 Acute Lymphocytic Leukemia Splenectomy (Removal of Spleen) 6-Mercaptopurine (Purinethol, 6-MP) Alemtuzumab (Campath ) Arsenic Trioxide (Trisenox ) Bendamustine (Treanda ) Bexarotene (Targretin ) Bleomycin
Acute Lymphocytic Leukemia Splenectomy (Removal of Spleen) 6-Mercaptopurine (Purinethol, 6-MP) Alemtuzumab (Campath ) Arsenic Trioxide (Trisenox ) Bendamustine (Treanda ) Bexarotene (Targretin ) Bleomycin
OVERVIEW OF COMMENTS RECEIVED ON LIST OF PAEDIATRIC NEEDS ONCOLOGY I (CYTOTOXIC THERAPY)
 European Medicines Agency Evaluation of Medicines for Human Use London, September 2006 Doc. Ref. OVERVIEW OF COMMENTS RECEIVED ON LIST OF PAEDIATRIC NEEDS ONCOLOGY I (CYTOTOXIC THERAPY) Table 1: Organisations
European Medicines Agency Evaluation of Medicines for Human Use London, September 2006 Doc. Ref. OVERVIEW OF COMMENTS RECEIVED ON LIST OF PAEDIATRIC NEEDS ONCOLOGY I (CYTOTOXIC THERAPY) Table 1: Organisations
Disclosure. Objectives. Objectives. Oncologic Emergency. Classification of Emergencies 1 3/2/2015
 Disclosure Oncologic Emergencies and Acute Supportive Care of the Critically Ill Oncology Patient By: Kimberly Regis, Pharm.D. Broward Health Medical Center Pharmacy Resident 2014-2015 I do not have a
Disclosure Oncologic Emergencies and Acute Supportive Care of the Critically Ill Oncology Patient By: Kimberly Regis, Pharm.D. Broward Health Medical Center Pharmacy Resident 2014-2015 I do not have a
HCPCS Code/ generic (Brand) Name J7506. J8520 capecitabine (Xeloda) 1. J8521 capecitabine. J8530 cyclophosphamide. (Cytoxan) 1
 drug and administration compendia Current Price () J7506 Prednisone, oral, per 5 mg 12/1/07 $0.07 $0.19 N/A prednisone 2 J8520 capecitabine (Xeloda) 1 Capecitabine, oral, 150 mg 8/1/07 $5.79 $4.59 N/A
drug and administration compendia Current Price () J7506 Prednisone, oral, per 5 mg 12/1/07 $0.07 $0.19 N/A prednisone 2 J8520 capecitabine (Xeloda) 1 Capecitabine, oral, 150 mg 8/1/07 $5.79 $4.59 N/A
Chemotherapy induced emesis: Are we doing are best? David Warr University of Toronto
 Chemotherapy induced emesis: Are we doing are best? David Warr University of Toronto david.warr@uhn.on.ca Conflict of interest Merck: speakers bureau and consultant Eisai: consultant Outline What is the
Chemotherapy induced emesis: Are we doing are best? David Warr University of Toronto david.warr@uhn.on.ca Conflict of interest Merck: speakers bureau and consultant Eisai: consultant Outline What is the
Introduction to Antineoplastic Prescribing
 Introduction to Antineoplastic Prescribing Robert Bradbury, R.Ph., BCPS Clinical Coordinator H. Lee Moffitt Cancer Center Objectives Meet the following goals concerning antineoplastic prescribing: Understand
Introduction to Antineoplastic Prescribing Robert Bradbury, R.Ph., BCPS Clinical Coordinator H. Lee Moffitt Cancer Center Objectives Meet the following goals concerning antineoplastic prescribing: Understand
Ovarian Cancer. compendia TREATMENT OF
 drug & compendia TREATMENT OF Ovarian Cancer With each publication, ManagedCare Oncology s drug & Compendia highlights a single medication or a group of medications that could be utilized in the management
drug & compendia TREATMENT OF Ovarian Cancer With each publication, ManagedCare Oncology s drug & Compendia highlights a single medication or a group of medications that could be utilized in the management
Clinical Tools and Resources for Self-Study and Patient Education
 Clinical Tools and Resources for Self-Study and Patient Education CHEMOTHERAPY-INDUCED NAUSEA AND VOMITING CLINICIAN'S RESOURCE GUIDE The clinical tools and resources contained herein are provided as educational
Clinical Tools and Resources for Self-Study and Patient Education CHEMOTHERAPY-INDUCED NAUSEA AND VOMITING CLINICIAN'S RESOURCE GUIDE The clinical tools and resources contained herein are provided as educational
Job title: Consultant Pharmacist/Advanced Practice Pharmacist
 Title : Guidelines for the Use of Antiemetics Purpose: To provide trust-wide guidance on the safe and effective use of antiemetics for the prevention and treatment of chemotherapy and radiotherapy induced
Title : Guidelines for the Use of Antiemetics Purpose: To provide trust-wide guidance on the safe and effective use of antiemetics for the prevention and treatment of chemotherapy and radiotherapy induced
Supplementary Figures
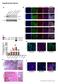 Supplementary Figures Supplementary Figure 1. Characterization of the generated hipsc lines a) Western blot analysis indicating the efficient downregulation of p53 upon knockdown in three hipsc lines (
Supplementary Figures Supplementary Figure 1. Characterization of the generated hipsc lines a) Western blot analysis indicating the efficient downregulation of p53 upon knockdown in three hipsc lines (
Co-morbidities in Elderly Breast Cancer Patients treated with Chemotherapy
 Co-morbidities in Elderly Breast Cancer Patients treated with Chemotherapy Authors: Romina Sosa, Julia Molony, Shuling Li, Richard Barron, Phuong Khanh Morrow, Scott Stryker, Jiannong Liu, Allan J. Collins,
Co-morbidities in Elderly Breast Cancer Patients treated with Chemotherapy Authors: Romina Sosa, Julia Molony, Shuling Li, Richard Barron, Phuong Khanh Morrow, Scott Stryker, Jiannong Liu, Allan J. Collins,
Chemotherapy Induced Nausea and Vomiting (CINV) Anti-emetic Guidelines
 North of England Cancer Network Chemotherapy Induced Nausea and Vomiting (CINV) Anti-emetic Guidelines Adult Oncology & Haematology Quality and safety for every patient every time For more information
North of England Cancer Network Chemotherapy Induced Nausea and Vomiting (CINV) Anti-emetic Guidelines Adult Oncology & Haematology Quality and safety for every patient every time For more information
Patient-Centered Management of Chemotherapy-Induced Nausea and Vomiting
 Clear and accurate communication between patients and health care teams is the key to successfully controlling chemotherapy-induced nausea and vomiting. Roseate Spoonbill_2005. Photograph courtesy of Henry
Clear and accurate communication between patients and health care teams is the key to successfully controlling chemotherapy-induced nausea and vomiting. Roseate Spoonbill_2005. Photograph courtesy of Henry
Trust Guideline for Prevention and Control of Chemotherapy and Radiotherapy Induced Nausea and Vomiting in Adults
 A Clinical Guideline For Use in: Organisation-wide By: For: Key words: Name of document author: Job title of document author: Name of document author s Line Manager: Job title of author s Line Manager:
A Clinical Guideline For Use in: Organisation-wide By: For: Key words: Name of document author: Job title of document author: Name of document author s Line Manager: Job title of author s Line Manager:
Systemic Anticancer Therapy Drug Interactions Table
 Systemic Anticancer Therapy Drug Interactions Table 1 This guide is intended to cover the most common interactions involving Systemic Anticancer therapies, e.g. cytotoxic agents, monoclonal antibodies
Systemic Anticancer Therapy Drug Interactions Table 1 This guide is intended to cover the most common interactions involving Systemic Anticancer therapies, e.g. cytotoxic agents, monoclonal antibodies
Welcome. Coding uidelines Coding Guidelines Coding. Coding Guidelines Coding Guidelines. Contact Us. Edition #1 March 2018
 Coding Guidelines Coding Guidelines uidelines Coding Guidelines Coding Coding Coding Guidelines Guidelines Coding uidelines Coding Guidelines Coding Coding Chemotherapy Guidelines and Radiation Coding
Coding Guidelines Coding Guidelines uidelines Coding Guidelines Coding Coding Coding Guidelines Guidelines Coding uidelines Coding Guidelines Coding Coding Chemotherapy Guidelines and Radiation Coding
Guidelines for the Management of Extravasation (Version 5 May 2012)
 Guidelines for the Management of Extravasation (Version 5 May 2012) Quality and safety for every patient every time Document Control Prepared By Chemo Nurse Group Chemo Nurse Group/ South Tees FT NECDAG
Guidelines for the Management of Extravasation (Version 5 May 2012) Quality and safety for every patient every time Document Control Prepared By Chemo Nurse Group Chemo Nurse Group/ South Tees FT NECDAG
DRUG PROPERTIES YOU NEED TO KNOW
 jfitzake@d.umn.edu www.d.umn.edu/~jfitzake Page 1 of 8 DRUG PROPERTIES YOU NEED TO KNOW 1. Mechanism of action a. chemical class b. resistance 2. Pharmacokinetics 3. Therapeutic uses 4. Major side effects/toxicities
jfitzake@d.umn.edu www.d.umn.edu/~jfitzake Page 1 of 8 DRUG PROPERTIES YOU NEED TO KNOW 1. Mechanism of action a. chemical class b. resistance 2. Pharmacokinetics 3. Therapeutic uses 4. Major side effects/toxicities
intolerance to, contraindication to, or therapeutic failure on a minimum 3 month trial of Inflectra*
 What s New Medical Pharmaceutical Policy March 2018 Updates MBP 5.0 Remicade (infliximab), Inflectra (infliximab-dyyb), Renflexis (infliximab-abda)- Updated policy Remicade (infliximab), Inflectra (infliximab-dyyb)
What s New Medical Pharmaceutical Policy March 2018 Updates MBP 5.0 Remicade (infliximab), Inflectra (infliximab-dyyb), Renflexis (infliximab-abda)- Updated policy Remicade (infliximab), Inflectra (infliximab-dyyb)
Hazardous Medication List
 Hazardous Medication List REDUCING OCCUPATIONAL EXPOSURE TO HAZARDOUS MEDICATION FOR ALL STAFF Table of Contents Hazardous Medication List Key Points... 1 Hazardous Medication List... 2 Special Handling
Hazardous Medication List REDUCING OCCUPATIONAL EXPOSURE TO HAZARDOUS MEDICATION FOR ALL STAFF Table of Contents Hazardous Medication List Key Points... 1 Hazardous Medication List... 2 Special Handling
Antiemetics: Guidelines, Interactions and more.
 Antiemetics: Guidelines, Interactions and more. Chemotherapy-induced side effects -the patient s view Loss of hair The thought of coming for chemo Affects family/partner Affects work/home duties Jude Lees
Antiemetics: Guidelines, Interactions and more. Chemotherapy-induced side effects -the patient s view Loss of hair The thought of coming for chemo Affects family/partner Affects work/home duties Jude Lees
Title Developed By. Implementation Contact person(s) Review Date Group Responsible. NICaN Policy: Management of Chemotherapy Extravasation; Version 3
 Title Developed By Management of Chemotherapy Extravasation NICaN Regional Pharmacy Group Version History Version 3: 2009 (See appendix 6) Version 2: January 2006 Version 1: June 2005 Consultation Period
Title Developed By Management of Chemotherapy Extravasation NICaN Regional Pharmacy Group Version History Version 3: 2009 (See appendix 6) Version 2: January 2006 Version 1: June 2005 Consultation Period
Working Formulary January 2013 Oncology Chemotherapy Regimens
 Working Formulary January 2013 Oncology Chemotherapy Regimens In the currently changing commissioning landscape, this document is intended to represent the up to date list of non clinical trial chemotherapy
Working Formulary January 2013 Oncology Chemotherapy Regimens In the currently changing commissioning landscape, this document is intended to represent the up to date list of non clinical trial chemotherapy
Guidelines for the Management of Extravasation
 Guidelines for the Management of Extravasation (Version 5.5-04 Nov 2016) Quality and safety for every patient every time Document Control Prepared By Chemo Nurse Group Chemo Nurse Group/ South Tees FT
Guidelines for the Management of Extravasation (Version 5.5-04 Nov 2016) Quality and safety for every patient every time Document Control Prepared By Chemo Nurse Group Chemo Nurse Group/ South Tees FT
H&HD ANTINEOPLASTIC DRUG CARD ASSEMBLY INSTRUCTIONS
 H&HD ANTINEOPLASTIC DRUG CARD ASSEMBLY INSTRUCTIONS Each of you should have 37 new cards: 7 orange cards for antimetabolites 11 white cards for miscellaneous drugs (2 DNA synthesis inhibitors, 1 enzyme,
H&HD ANTINEOPLASTIC DRUG CARD ASSEMBLY INSTRUCTIONS Each of you should have 37 new cards: 7 orange cards for antimetabolites 11 white cards for miscellaneous drugs (2 DNA synthesis inhibitors, 1 enzyme,
Standard Regimens for Haematology
 Regimens for Haematology ChlVPP Chlorambucil 6mg/m 2 PO D1 to 14 Vinblastine 6mg/m 2 (max 10mg) IV on D1 & 8 Procarbazine 100mg/m 2 PO on D1 to 14 Prednisolone 40mg PO D1 to 14 ABVD Doxorubicin 25mg/m
Regimens for Haematology ChlVPP Chlorambucil 6mg/m 2 PO D1 to 14 Vinblastine 6mg/m 2 (max 10mg) IV on D1 & 8 Procarbazine 100mg/m 2 PO on D1 to 14 Prednisolone 40mg PO D1 to 14 ABVD Doxorubicin 25mg/m
Richard J. Gralla, MD Medical Director Quality of Life Research Associates New York, NY
 Oncology Consultations Improving the Management of Chemotherapy-Induced Nausea and Vomiting (CINV) A CE-Certified Activity Featuring Consultations With Supported by an educational grant from Eisai. Dannemiller
Oncology Consultations Improving the Management of Chemotherapy-Induced Nausea and Vomiting (CINV) A CE-Certified Activity Featuring Consultations With Supported by an educational grant from Eisai. Dannemiller
DRUGS YOU NEED TO KNOW
 jfitzake@d.umn.edu Page 1 of 7 DRUGS YOU NEED TO KNOW ALKYLATING AGENTS (blue cards) BUSULFAN CARMUSTINE (BCNU) CYCLOPHOSPHAMIDE DACARBAZINE LOMUSTINE (CCNU) MECHLORETHAMINE MELPHALAN THIOTEPA NATURAL
jfitzake@d.umn.edu Page 1 of 7 DRUGS YOU NEED TO KNOW ALKYLATING AGENTS (blue cards) BUSULFAN CARMUSTINE (BCNU) CYCLOPHOSPHAMIDE DACARBAZINE LOMUSTINE (CCNU) MECHLORETHAMINE MELPHALAN THIOTEPA NATURAL
Medication Review. Cancer Chemotherapy Drugs. Pharmacy Technician Training Systems Passassured, LLC
 Medication Review Cancer Chemotherapy Drugs Pharmacy Technician Training Systems Passassured, LLC Medication Review, Cancer Chemotherapy Drugs PassAssured's Pharmacy Technician Training Program Medication
Medication Review Cancer Chemotherapy Drugs Pharmacy Technician Training Systems Passassured, LLC Medication Review, Cancer Chemotherapy Drugs PassAssured's Pharmacy Technician Training Program Medication
Supportive care session 1:
 Board review in oncology pharmacy 2013 Managing Disease or Treatment Related Complication Supportive care session 1: Chemotherapy induced-nausea and vomiting Suthan Chanthawong, B. Pharm, RPh. Objectives
Board review in oncology pharmacy 2013 Managing Disease or Treatment Related Complication Supportive care session 1: Chemotherapy induced-nausea and vomiting Suthan Chanthawong, B. Pharm, RPh. Objectives
Antiemesis. NCCN Clinical Practice Guidelines in Oncology. Antiemesis. Version Continue
 Table of Contents Clinical in Oncology Version 2.2010 Continue www.nccn.org Version 2.2010, 04/07/2010 2010 National Comprehensive Cancer Network, Inc. All rights reserved. These guidelines and this illustration
Table of Contents Clinical in Oncology Version 2.2010 Continue www.nccn.org Version 2.2010, 04/07/2010 2010 National Comprehensive Cancer Network, Inc. All rights reserved. These guidelines and this illustration
The following are J Code requirements
 The following are J Code requirements J Codes 20610 Arthrocentesis, aspiration and/or injection; major joint or bursa (eg, shoulder, hip, knee joint, subacromial bursa) A9579 Injection, gadolinium based
The following are J Code requirements J Codes 20610 Arthrocentesis, aspiration and/or injection; major joint or bursa (eg, shoulder, hip, knee joint, subacromial bursa) A9579 Injection, gadolinium based
CPT Service Description Effective Date
 Medical Oncology Program Review Code List 2 nd Quarter 2018 This list is subject to change once per quarter. Changes will be posted to the BCBSNC website at www.bcbsnc.com by the 10th day of January, April,
Medical Oncology Program Review Code List 2 nd Quarter 2018 This list is subject to change once per quarter. Changes will be posted to the BCBSNC website at www.bcbsnc.com by the 10th day of January, April,
PHARMACY PEARLS CHANIN WRIGHT, PHARMD
 PHARMACY PEARLS CHANIN WRIGHT, PHARMD OBJECTIVES From the pearls heard, discuss with someone near you What is something you can apply to your setting Traumatic brain injury in children Goals Correct and
PHARMACY PEARLS CHANIN WRIGHT, PHARMD OBJECTIVES From the pearls heard, discuss with someone near you What is something you can apply to your setting Traumatic brain injury in children Goals Correct and
Corporate Medical Policy
 Antiemetic Injection Therapy Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: antiemetic_injection_therapy 5/2015 3/2017 3/2018 3/2017 Description of Procedure
Antiemetic Injection Therapy Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: antiemetic_injection_therapy 5/2015 3/2017 3/2018 3/2017 Description of Procedure
2. Date of most recent breast cancer diagnosis / / 3. Pathlogy: Padget s Phyllodes Tubular Medullary Mucinous
 Breast Cancer Patients: Begin on page 1, then skip to page 3 Lymphoma Patients: Begin on page 2 1. Newly diagnosed with Breast carcinoma Stage: (includes inflammatory breast and newly diagnosed recurrent
Breast Cancer Patients: Begin on page 1, then skip to page 3 Lymphoma Patients: Begin on page 2 1. Newly diagnosed with Breast carcinoma Stage: (includes inflammatory breast and newly diagnosed recurrent
Contents Trials. Dose guidelines. Calvert formula Issue Date: January Author: No: Copy. Pagee 1 of 21. Issue.
 Chapter 1: General information Contents Chapter 1: General information... 1 General observations... 2 Protocol Additions 2013... 2 Cancer Drugs Fund... 2 Off Protocol Treatment Policy... 2 Trials... 2
Chapter 1: General information Contents Chapter 1: General information... 1 General observations... 2 Protocol Additions 2013... 2 Cancer Drugs Fund... 2 Off Protocol Treatment Policy... 2 Trials... 2
Chemotherapy-induced Nausea and Vomiting: The Pharmacist s Role in Integrating Clinical Guidelines into Patient Care
 Chemotherapy-induced Nausea and Vomiting: The Pharmacist s Role in Integrating Clinical Guidelines into Patient Care Presented as a Live Webinar Wednesday, April 13, 2016 12:00 p.m. 1:00 p.m. ET Tuesday,
Chemotherapy-induced Nausea and Vomiting: The Pharmacist s Role in Integrating Clinical Guidelines into Patient Care Presented as a Live Webinar Wednesday, April 13, 2016 12:00 p.m. 1:00 p.m. ET Tuesday,
Chemotherapy 101 for Radiation Oncology Workers
 Chemotherapy 101 for Radiation Oncology Workers James Sinclair, M.D. CCARE/Medical Director Scripps Cancer Center March 9, 2013 Category 1) Alkylating agents Alkylating agents directly damage DNA to prevent
Chemotherapy 101 for Radiation Oncology Workers James Sinclair, M.D. CCARE/Medical Director Scripps Cancer Center March 9, 2013 Category 1) Alkylating agents Alkylating agents directly damage DNA to prevent
REVIEW Guideline for the Classification of the Acute Emetogenic Potential of Antineoplastic Medication in Pediatric Cancer Patients
 Pediatr Blood Cancer 2011;57:191 198 REVIEW Guideline for the Classification of the Acute Emetogenic Potential of Antineoplastic Medication in Pediatric Cancer Patients L. Lee Dupuis, MScPhm, ACPR, FCSHP,
Pediatr Blood Cancer 2011;57:191 198 REVIEW Guideline for the Classification of the Acute Emetogenic Potential of Antineoplastic Medication in Pediatric Cancer Patients L. Lee Dupuis, MScPhm, ACPR, FCSHP,
Lisa M. Hess Diane Michael Daniel S. Mytelka Julie Beyrer Astra M. Liepa Steven Nicol
 Gastric Cancer (2016) 19:607 615 DOI 10.1007/s10120-015-0486-z ORIGINAL ARTICLE Chemotherapy treatment patterns, costs, and outcomes of patients with gastric cancer in the United States: a retrospective
Gastric Cancer (2016) 19:607 615 DOI 10.1007/s10120-015-0486-z ORIGINAL ARTICLE Chemotherapy treatment patterns, costs, and outcomes of patients with gastric cancer in the United States: a retrospective
Gazyva (obinutuzumab)
 STRENGTH DOSAGE FORM ROUTE GPID 1000mg/40mL Vial Intravenous 35532 MANUFACTURER Genentech, Inc. INDICATION(S) Gazyva (obinutuzumab) is a CD20- directed cytolytic antibody and is indicated, in combination
STRENGTH DOSAGE FORM ROUTE GPID 1000mg/40mL Vial Intravenous 35532 MANUFACTURER Genentech, Inc. INDICATION(S) Gazyva (obinutuzumab) is a CD20- directed cytolytic antibody and is indicated, in combination
