Recall and propagation of allospecific memory T cells independent of secondary lymphoid organs
|
|
|
- Peregrine Sharp
- 6 years ago
- Views:
Transcription
1 Recall and propagation of allospecific memory T cells independent of secondary lymphoid organs Geetha Chalasani*, Zhenhua Dai*, Bogumila T. Konieczny, Fady K. Baddoura, and Fadi G. Lakkis* *Section of Nephrology (Department of Medicine) and Section of Immunobiology, Yale University School of Medicine, P.O. Box , 333 Cedar Street, New Haven, CT 06520; Emory Vaccine Center and Department of Microbiology and Immunology, Emory University School of Medicine, Atlanta, GA 30322; and Pathology and Laboratory Medicine, Veterans Affairs Medical Center, and State University of New York, Buffalo, NY Edited by Philippa Marrack, National Jewish Medical and Research Center, Denver, CO, and approved March 15, 2002 (received for review November 7, 2001) The allospecifc T cell population responding to a transplanted organ consists of both naïve and memory lymphocytes. Although it is established that naive T cells are activated by antigen within the organized structures of secondary lymphoid organs (the spleen, lymph nodes, and mucosal lympoid tissues), it is not clear whether memory T cell activation and propagation depend on homing to these organs. To answer this question, we investigated whether allospecific naïve or memory T cells can mediate acute cardiac allograft rejection in mutant mice that lack all of their secondary lymphoid tissues. The results of our experiments demonstrated that antigen-experienced memory T cells have two advantages over naïve T cells: (i) memory T cells mount a vigorous immune response that leads to allograft rejection independent of secondary lymphoid organs; and (ii) memory T cells generate more memory T cells without homing to secondary lymphoid organs. These unique properties of memory T cells were further confirmed by showing that memory-like T cells that arise from the homeostatic proliferation of naive T cells in the absence of antigenic stimulation are suboptimal at rejecting allografts and do not generate memory T cells in mice devoid of secondary lymphoid tissues. A cardinal feature of the adaptive immune response is its ability to generate long-lived populations of memory T lymphocytes (1). Memory T cells are specific to the antigen encountered during the primary immune response and react rapidly and vigorously on re-encounter with the same antigen. Memory T cells that recognize microbial antigens provide the organism with long-lasting protection against potentially fatal infections. On the other hand, memory T cells that recognize donor alloantigens could jeopardize the survival of life-saving organ transplants (2). Therefore, understanding the fundamental mechanisms that govern memory T cell activation is important for vaccine development as well as the design of effective strategies for preventing allograft rejection. The adaptive immune response is initiated within secondary lymphoid organs where naïve T cells encounter foreign antigen, are activated, and differentiate into effector cells (3 5). The majority of T cells participating in a primary immune response rapidly undergo apoptosis after the foreign antigen is eliminated, and only a small proportion of these cells survive to become memory T cells. Unlike naïve T cells whose homing is restricted to secondary lymphoid organs, memory T cells migrate to nonlymphoid tissues in the periphery (6 8). Recent studies have shown that memory T cells isolated from the nonlymphoid tissues of immunized mice have immediate ex vivo effector functions (6, 7). However, it remains unclear whether memory T cells can mount a productive immune response that leads to antigen clearance and the generation of more memory T cells in vivo independent of secondary lymphoid organs. Here we addressed these questions by examining whether allospecific memory T cells can mediate the rejection of cardiac allografts and generate more memory T cells in mutant mice that lack secondary lymphoid organs. We demonstrate that, unlike naïve T cells, antigen-experienced memory T cells mount a productive alloimmune response and beget more memory T cells independent of secondary lymphoid organs. We also show that these unique properties of memory T cells are not shared by memorylike T cells that arise from the homeostatic proliferation of naive T lymphocytes in the absence of antigenic stimulation. Methods Mouse Strains. B6 mice homozygous for the mutation that leads to alymphoplasia (aly aly) (CLEA Japan, Tokyo) (9), recombination activating gene-2 knockout (Rag2 / ) B6 mice (The Jackson Laboratory), and 2C T cell receptor-transgenic (TCRtg) B6 mice were housed in specific pathogen-free environment. Wild-type (wt) BALB c and B6 mice were purchased from The Jackson Laboratory. Mouse Surgery and Tissue Analysis. Heart donors were 6- to 8-week-old BALB c (H-2 d ), and recipients were 6- to 8-week-old splenectomized B6 aly aly (H-2 b ) mice. Splenectomy was performed 2 weeks before heart transplantation as follows: A 1.5-cm skin incision was made under anesthesia midway between the last rib and the hip, the peritoneal membrane was opened, and the entire spleen was removed intact after ligating the splenic vein and artery at the hilum. The peritoneal membrane and the skin were then closed separately. This procedure ensures that the spleen is removed in total and that no splenic fragments are left behind as confirmed by examining the mice at the time of death. Fully vascularized heterotopic heart transplantation was performed as described (5, 10). Transplanted mice were monitored daily, and rejection was defined as cessation of palpable cardiac contractions. Grafts were harvested at the time of rejection or at 100 days after transplantation in mice that did not reject their allografts. Graft tissue was fixed in B5 solution, embedded in paraffin, sectioned, and stained with hematoxylin eosin or antimouse CD3 (PharMingen) followed by peroxidase-conjugated secondary antibody. T Cell Preparation, Phenotyping, and Adoptive Transfer. An adoptive transfer model in which a small proportion of allospecific CD8 TCR-tg (2C) lymphocytes are parked in syngeneic wt hosts was used to prepare naïve and memory T cell populations. 2C lymphocytes recognize the L d BALB c alloantigen and therefore serve as a marker of allospecific T cells (11). Spleen and lymph node cells from naïve 2C mice (B6 background) were This paper was submitted directly (Track II) to the PNAS office. Abbreviations: aly, alymphoplasia gene; Rag2, recombination activating gene-2; TCR-tg, T cell receptor-transgenic; wt, wild type; CTL, cytotoxic T lymphocyte. G.C. and Z.D. contributed equally to this work. To whom reprint requests should be addressed. fadi.lakkis@yale.edu. The publication costs of this article were defrayed in part by page charge payment. This article must therefore be hereby marked advertisement in accordance with 18 U.S.C solely to indicate this fact. IMMUNOLOGY cgi doi pnas PNAS April 30, 2002 vol. 99 no
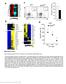
2 pooled and enriched for T cells by nonadherence to nylon wool. A population of enriched T cells containing transgenic (CD8 1B2 ) lymphocytes (1B2 is the clonotypic antibody that recognizes the transgenic TCR) was then injected i.v. into syngeneic wt B6 mice on day 0. The mice were immunized with BALB c splenocytes in PBS i.p. on days 2 and 10 to generate allospecific memory T cells or were left unimmunized to obtain naive T cells. Ten weeks later, spleen and lymph node cells were pooled and enriched for 2C T cells by magnetic cell separation (MACS) (Miltenyi Biotec, Auburn, CA). MACS was performed by depleting MHCII cells followed by positive selection of 1B2 cells. The CD8 1B2 (2C) cells present in the enriched naïve and memory populations were then quantitated before adoptive transfer by flow analysis using an anti-mouse CD8 antibody conjugated to phycoerythrin (PharMingen) and 1B2 IgG1 mab followed by anti-mouse IgG1 antibody conjugated to FITC (Zymed) (12). The naïve and memory CD8 1B2 cell populations were also phenotyped by flow analysis using anti-cd44-biotin streptavidin-percp, anti-cd62l-apc, anti- CD25-APC, anti-1b11 followed by biotin-igg2a streptavidin- PerCP, and appropriate isotype antibody controls (all antibodies were purchased from PharMingen). Finally, a population of enriched naïve or memory T cells containing a constant number of CD8 1B2 (2C) T cells ( ) was adoptively transferred to splenectomized aly aly recipients of BALB c hearts 2 days after transplantation. An adoptive transfer model in which 2C lymphocytes are parked in syngeneic, lymphocyte-deficient Rag2 / hosts was used to prepare allospecific memory-like T cell populations that arise in the absence of antigenic stimulation (naïve Rag2 / ) (13 16). A total of CD8 1B2 T cells mixed with wt B6 T cells were injected i.v. into syngeneic Rag2 / mice on day 0. Ten weeks later, spleen and lymph node cells were pooled and enriched for 2C T cells as described in the previous paragraph. As positive control, another group of Rag2 / recipients was immunized with BALB c splenocytes i.p. on days 2 and 10 after adoptive transfer to generate memory T cells (memory Rag2 / ). CD8 1B2 cells present in the enriched naïve Rag2 / and memory Rag2 / populations were then quantitated and phenotyped before adoptive transfer as described in the previous paragraph. Finally, a population of enriched memory Rag2 / or naive Rag2 / T cells containing a constant number of allospecific 2C T cells ( ) was adoptively transferred to splenectomized aly aly recipients of BALB c hearts 2 days after transplantation. As an additional control, a group of transplant recipients received a mixture of freshly isolated naive 2C ( ) and naive wt B6 T cells (total number T cells). Phenotyping and Quantitation of 2C T Cells Recovered from Transplanted Mice. Blood was collected from a splenectomized aly aly transplant recipient at the time of rejection or at 100 days after transplantation in mice that did not reject their grafts. After RBC lysis, CD8 1B2 T cells present in the blood were quantitated and phenotyped by flow analysis as described above. The number of retrieved 2C T cells was extrapolated according to the following formula: % CD8 1B2 cells number of cells per ml blood 2 ml (estimated volume of blood per mouse). In each mouse studied, the number and phenotype of recovered 2C T cells were compared with the number and phenotype of the 2C T cells that were injected 2 days after transplantation. Cytotoxic T Lymphocyte (CTL) Activity. Spleen and lymph node T cells were harvested from wt B6 mice harboring either naïve or memory T cells (same as donor mice used in the adoptive experiments described above) were pooled, enriched for CD8 T cells by magnetic cell separation (MACS), and immediately assayed for ex vivo CTL activity against BALB c target splenocytes. Another group of wt B6 mice harboring memory T cells was rechallenged with BALB c splenocytes i.p. and s.c. 2 days before ex vivo CTL assay. Allospecific CTL activity was measured by incubating the CD8-enriched T cells with either Con A-activated (H-2 d )BALB c target cells or third-party cells, LK35.2 (H-2 k ) (American Type Culture Collection) for 3 h. Target cells were labeled with calcein-am (Molecular Probes), and calcein release was measured in a LS50B luminescence spectrometer (Perkin Elmer) (17). Experiments in which spontaneous calcein release was more than 25% of maximal release were excluded. Antigen-specific cytotoxic activity was calculated according to the following formula: % specific lysis 100 [(sample release spontaneous release) (maximum release spontaneous release)]. The ex vivo CTL activity of RBC-lysed peripheral blood cells from splenectomized aly aly recipients of BALB c cardiac allografts was measured by the same method. Results Memory T Cells Mediate Allograft Rejection Independent of Secondary Lymphoid Organs. To investigate the role of secondary lymphoid organs in the in vivo recall of memory T cells, we tested whether alloreactive memory T cells mediate allograft rejection in mice devoid of secondary lymphoid tissues [splenectomized alymphoplastic (aly aly) mice] (5, 9). These mice do not mount a primary alloimmune response and accept cardiac allografts permanently (5). Allospecific memory T cells were generated in vivo by immunizing B6 mice harboring 2C (CD8 TCR-tg) T cells with allogeneic BALB c splenocytes as described in Methods. Ten weeks after immunization, spleen and lymph node cells were pooled, enriched for T cells, and adoptively transferred to splenectomized aly aly mice transplanted with BALB c hearts. To compare memory T cell function to that of naive T cells, a second group of transplanted, splenectomized aly aly mice received T cells from unimmunized B6 mice harboring naive 2C lymphocytes for 10 weeks. The number of T cells was adjusted such that the transferred population contained an equal number of allospecific 2C lymphocytes ( CD8 1B2 cells) in each case. As shown in Fig. 1A, 2C lymphocytes in unimmunized B6 mice retained a naive phenotype (CD44 low CD62L high CD25 low 1B11 low ) whereas those in immunized mice exhibited a shift toward the memory phenotype (CD44 high CD62L low CD25 low 1B11 low ). Moreover, CD8 T cells from immunized mice lacked immediate, allospecific, ex vivo CTL activity but rapidly acquired such activity upon in vivo rechallenge with BALB c splenocytes (Fig. 1B), suggesting that the harvested population contained predominantly central memory T cells. No cytolytic activity was observed against third-party target cells. After transfer to transplanted, splenectomized aly aly hosts, the naive T cell population failed to induce cardiac allograft rejection (Fig. 1C). This finding is consistent with the dependence of naïve T cells on secondary lymphoid organs to mount a primary alloimmune response (5). In contrast, the T cell population that contains memory T cells induced prompt acute rejection of cardiac allografts upon transfer to splenectomized aly aly hosts (Fig. 1B). Histopathologic examination confirmed that allograft loss in mice that received memory T cells was caused by high-grade acute rejection characterized by heavy infiltration of the myocardium with CD3 cells (Fig. 1D). On the other hand, cardiac allografts accepted by mice that received naive T cells did not exhibit histopathologic findings of rejection and were free of CD3 cell infiltrates (Fig. 1D). These results establish that memory, but not naive, T cells mount a productive immune response in vivo that leads to allograft rejection independent of secondary lymphoid organs. Memory-Like T Cells Have Intermediate in Vivo Function in the Absence of Secondary Lymphoid Organs. Naive T cells transferred into a lymphopenic host undergo homeostatic proliferation and cgi doi pnas Chalasani et al.
3 IMMUNOLOGY Fig. 1. Memory T cells mediate allograft rejection independent of secondary lymphoid organs. Naïve and memory T cells were generated in wt hosts and adoptively transferred to splenectomized aly aly heart transplant recipients as described in Methods. Identical enrichment methods were applied to naïve and memory T cells. (A) Phenotype of naive and memory allospecific T cells determined before adoptive transfer by four-color flow analysis after gating on the CD8 1B2 (2C TCR-tg) cell population. Histograms shown are representative of three separate experiments. (B) CTL activity of naïve and memory T cells against target BALB c splenocytes before adoptive transfer. CD8-enriched T cells from memory (immunized) mice were assayed for ex vivo CTL activity either immediately or 2 days after in vivo recall with BALB c splenocytes. One representative assay is shown. (C) Cardiac allograft survival in splenectomized aly aly mice that received either naive (n 3) or memory (n 3) T cell populations. (D) Histological (hematoxylin eosin, H&E) and immunohistochemical (CD3) analysis of cardiac allograft tissue harvested 100 days after transplantation in the naive group and on the day of rejection in the memory group. acquire memory-like markers in the absence of antigen-specific stimulation (13 16). However, it is not known whether these cells function in vivo as antigen-experienced, true memory T lymphocytes or as naïve T cells. To address this question, we transferred a mixture of naive B6 and 2C T cells that were parked for 10 weeks in lymphocyte-deficient Rag2 / mice (naive Rag2 / ) to splenectomized aly aly mice transplanted with BALB c hearts. Control mice received an equal number of either un- Chalasani et al. PNAS April 30, 2002 vol. 99 no
4 Fig. 2. Memory-like T cells have intermediate in vivo function in the absence of secondary lymphoid organs. Memory-like (naive Rag2 / ) and antigenexperienced memory (memory Rag2 / ) T cells were generated in Rag2 / hosts and adoptively transferred to splenectomized aly aly heart transplant recipients. The naïve group received T cells harvested from naïve wt and 2C mice as described in Methods.(A) Phenotype of naive, naive Rag2 /, and memory Rag2 / allospecific T cells determined before adoptive transfer by four-color flow analysis after gating on the CD8 1B2 (2C TCR-tg) cell population. Histograms shown are representative of 4 5 experiments. (B) Cardiac allograft survival in splenectomized aly aly mice that received naïve (F, n 4), naive Rag2 / (, n 4), or memory Rag2 / (E, n 5) T cell populations. (C) Histological (hematoxylin eosin, H&E) and immunohistochemical (CD3) analysis of cardiac allograft tissue harvested 100 days after transplantation in the naive and naive Rag2 / group and on the day of rejection in the memory Rag2 / group. (Magnification 200.) parked naive B6 and 2C T cells (naive) or memory B6 and 2C T cells generated in immunized Rag2 / hosts (memory Rag2 / ). The majority of 2C T lymphocytes in the naive Rag2 / population were CD44 high (Fig. 2A), but unlike antigen-experienced memory Rag2 / T cells, the naive Rag2 / population did not consistently induce allograft rejection in splenectomized aly aly recipients (Fig. 2B). As shown in Fig. 2B, acute allograft rejection occurred in only one of four mice that received naive Rag2 / T cells. Naive Rag2 / T cells also behaved differently from the unparked naive population in that they infiltrated cardiac allografts that were still functioning at 100 days after transplantation whereas the naive T cells did not (Fig. 2C). Therefore, memory-like T cells arising from the homeostatic proliferation of naive T cells in the absence of antigenic stimulation have intermediate in vivo function in mice that lack secondary lymphoid organs and thus are distinct from either naive or true memory T cells cgi doi pnas Chalasani et al.
5 CD8 Memory T Cells Generate More Memory T Cells in the Absence of Secondary Lymphoid Organs. It has been proposed that central memory T cells that reside in secondary lymphoid organs are responsible for replenishing the memory pool whereas effector memory T cells that circulate through peripheral tissues exert effector functions upon reencounter with antigen but do not generate more memory T cells (6, 18). We addressed this hypothesis by testing whether secondary lymphoid organs are necessary for propagating T cell memory. Specifically, we asked whether the transferred memory T cell population that leads to cardiac allograft rejection in mice devoid of secondary lymphoid organs (as shown in Fig. 1) gave rise to more memory T cells. To answer this question, we quantitated and phenotyped 2C T cells present in the blood of splenectomized aly aly hosts at the time of graft harvest and compared them to the number and phenotype of the 2C T cells that were injected 2 days after transplantation. Antigen-experienced memory 2C T cells generated in B6 mice (memory) or in Rag2 / mice (memory Rag2 / ) increased significantly in number (5- to 40-fold increase) after allograft rejection and retained their memory phenotype (Fig. 3 A and B). In contrast, the memory-like 2C T cell population (naive Rag2 / ) generated in the absence of antigenic stimulation increased by only 2-fold and did not retain the CD44 high phenotype irrespective of whether allograft rejection occurred or not. Naive 2C T cells, on the other hand, retained their naive phenotype and appeared to have decreased in number 100 days after transplantation. Moreover, the T cell population retrieved from the blood of the memory group exhibited immediate ex vivo CTL activity against BALB c target splenocytes, suggesting that it contained functional memory T cells, predominantly of the effector subset (Fig. 3C). These findings indicate that antigen-experienced, true memory T cells beget more memory T cells in the absence of secondary lymphoid tissue. Discussion Allograft rejection is a T cell-dependent process mediated by either CD4 or CD8 T cells (19). Using cardiac allograft rejection as a readout for in vivo memory T cell function, we demonstrated here that an unfractionated population of antigen-experienced memory T cells mounts a productive immune response and generates more CD8 memory T cells independent of secondary lymphoid organs. These characteristics of antigen-experienced, true memory T cells were not shared by either naïve T cells or memory-like T cells generated in the absence of antigenic stimulation. Although our experimental approach does not distinguish between CD4 and CD8 memory recall, it approximates physiologic conditions wherein CD4 and CD8 T cells function in unison during alloimmune responses. Our observation that the number of memory CD8 TCR-tg lymphocytes (2C) increases after allograft rejection in splenectomized aly aly hosts indicates that the propagation of CD8 memory T cells is also independent of secondary lymphoid organs. Whether CD4 memory T cells follow the same rules as their CD8 counterparts remains to be tested in a system where alloreactive CD4 T cells can be tracked and quantitated. Recent studies have suggested the existence of two subsets of memory T cells: one that circulates through secondary lymphoid organs (central memory) and another that circulates through nonlymphoid tissues (effector memory) (6, 7, 18). It has also been proposed that the extralymphoid effector memory T cells are poised for immediate response to foreign antigen in the periphery whereas central memory T cells are specialized to proliferate within secondary lymphoid organs to generate more memory T cells (20). The memory T cell population studied in our experiments was isolated from the spleens and lymph nodes of immunized mice and lacked immediate ex vivo cytolytic activity (but rapidly acquired such activity upon antigenic recall), suggesting that it contained predominantly central CD8 memory Fig. 3. CD8 memory T cells generate more memory T cells in the absence of secondary lymphoid organs. Heart transplantation and adoptive transfer of T cells to splenectomized aly aly mice were performed as in Figs. 1 and 2. CD8 1B2 (2C TCR-tg) cells present in the blood of splenectomized aly aly hosts at the time of cardiac allograft rejection (naive Rag2 /, memory Rag2 /, and memory) or at 100 days after transplantation in mice that accepted their allografts [naive and naive Rag2 / (a)] were quantitated (A), phenotyped by flow analysis (B), and assayed for immediate ex vivo CTL activity (C). The bar graph compares the number of CD8 1B2 T cells injected to the number of CD8 1B2 cells retrieved in seven individual mice. The number of CD8 1B2 cells retrieved was extrapolated based on the number of CD8 1B2 T cells present per ml of blood. T cells. Interestingly, this memory population mounted a productive immune response and generated more effector memory T cells in mice that lack secondary lymphoid organs. Our results therefore suggest that a CD8 memory population consisting IMMUNOLOGY Chalasani et al. PNAS April 30, 2002 vol. 99 no
6 predominantly of central memory T cells is not only capable of mounting a productive immune response outside secondary lymphoid organs but also expands the effector memory pool without homing to secondary lymphoid tissues. It remains possible that the expansion of the central memory pool, on the other hand, depends on antigen re-encounter within secondary lymhoid tissues (6, 21). Alymphoplasia in aly aly mice is caused by a recessive mutation in the gene encoding Nf- b-inducing kinase (NIK) (22). Although adoptively transferred memory T cells used in our experiments did not contain the NIK mutation, it is possible that the absence of functional NIK in host cells could have influenced the migration or function of the transferred T cells caused by dysregulated cytokine or chemokine production in extralymphoid tissues. We believe that this possibility is unlikely as the only significant immune defects in aly aly mice that cannot be attributed to the absence of secondary lymphoid tissues are restricted primarily to B cells (23), and the expression of chemokine and chemokine receptors involved in lymphocyte homing appears to be normal in these mice (24). The data presented here underscore that immunologic memory is not merely the consequence of increased frequency of antigen-specific naïve T cells but is caused by the presence of a qualitatively distinct population of memory T cells. These memory T cells differ from their naïve counterparts in that they respond to foreign antigen outside the context of secondary lymphoid organs. This property, which is shared by memory B cells (25), endows the host with the ability to rapidly eliminate dangerous microbes shortly after their entry into peripheral tissues. On the other hand, this same property of memory lymphocytes poses a formidable obstacle to the long-term survival of organ transplants as current immunosuppressive agents are designed to target the homing and activation of naïve lymphocytes. We thank Drs. Rafi Ahmed (Emory University) and Kaja Murali- Krishna (University of Washington, Seattle) for their helpful comments. This work was supported by National Institutes of Health Grants AI41643, AI44644, and AI Sprent, J. & Surh, C. D. (2001) Curr. Opin. Immunol. 13, Heeger, P. S., Greenspan, N. S., Kuhlenschmidt, S., Dejelo, C., Hricik, D. E., Schulak, J. A. & Tary-Lehmann, M. (1999) J. Immunol. 163, Karrer, U., Althage, A., Odermatt, B., Roberts, C. W. M., Korsmeyer, S. J., Miyawaki, S., Hengartner, H. & Zinkernagel, R. M. (1997) J. Exp. Med. 185, Ochsenbein, A. F., Klenerman, P., Karrer, U., Ludewig, B., Pericin, M., Hengartner, H. & Zinkernagel, R. M. (1999) Proc. Natl. Acad. Sci. USA 96, Lakkis, F. G., Arakelov, A., Konieczny, B. T. & Inoue, Y. (2000) Nat. Med. 6, Reinhardt, R. L., Khoruts, A., Merica, R., Zell, T. & Jenkins, M. K. (2001) Nature (London) 410, Masopust, D., Vezys, V., Marzo, A. L. & Lefrancois, L. (2001) Science 291, Weninger, W., Crowley, M. A., Manjunath, N. & von Andrian, U. H. (2001) J. Exp. Med. 194, Miyawaki, S., Nakamura, Y., Suzuka, H., Koba, M., Yasumizu, R., Ikehara, S. & Shibata, Y. (1994) Eur. J. Immunol. 24, Niimi, M. (2001) J. Heart Lung Transplant. 20, Sha, W. C., Nelson, C. A., Newberry, R. D., Kranz, D. M., Russel, J. H. & Loh, D. Y. (1988) Nature (London) 335, Dai, Z., Arakelov, A., Wagener, M., Konieczny, B. T. & Lakkis, F. G. (1999) J. Immunol. 163, Kieper, W. C. & Jameson, S. C. (1999) Proc. Natl. Acad. Sci. USA 96, Murali-Krishna, K. & Ahmed, R. (2000) J. Immunol. 165, Cho, B. K., Rao, V. P., Ge, Q., Eisen, H. N. & Chen, J. (2000) J. Exp. Med. 192, Goldrath, A. W., Bogatzki, L. Y. & Bevan, M. J. (2000) J. Exp. Med. 192, Dai, Z., Konieczny, B. T. & Lakkis, F. G. (2000) J. Immunol. 165, Sallusto, F., Lenig, D., Forster, R., Lipp, M. & Lanzavecchia, A. (1999) Nature (London) 401, Auchincloss, H. J., Sykes, M. & Sachs, D. H. (1999) in Fundamental Immunology, ed. Paul, W. E. (Lippincott Raven, Philadelphia), pp Mackay, C. R. & von Andrian, U. H. (2001) Science 291, Sallusto, F. & Lanzavecchia, F. (2001) J. Clin. Invest. 108, Shinkura, R., Kitada, K., Matsuda, F., Tashiro, K., Ikuta, K., Suzuki, M., Kogishi, K., Serikawa, T. & Honjo, T. (1999) Nat. Genet. 22, Karrer, U., Althage, A., Odermatt, B., Hengartner, H. & Zinkernagel, R. M. (2000) Eur. J. Immunol. 30, Fagarasan, S., Shinkura, R., Kamata, T., Nogaki, F., Ikuta, K., Tashiro, K. & Honjo, T. (2000) J. Exp. Med. 191, Ochsenbein, A. F., Pinschewer, D. D., Sierro, S., Horvath, E., Hengartner, H. & Zinkernagel, R. M. (2000) Proc. Natl. Acad. Sci. USA 97, cgi doi pnas Chalasani et al.
CD4 + CD25 + regulatory T cells suppress allograft rejection mediated by memory CD8 + T cells via a CD30-dependent mechanism
 CD4 + CD25 + regulatory T cells suppress allograft rejection mediated by memory CD8 + T cells via a CD30-dependent mechanism Zhenhua Dai, 1 Qi Li, 1 Yinong Wang, 2 Ge Gao, 3 Lonnette S. Diggs, 1 George
CD4 + CD25 + regulatory T cells suppress allograft rejection mediated by memory CD8 + T cells via a CD30-dependent mechanism Zhenhua Dai, 1 Qi Li, 1 Yinong Wang, 2 Ge Gao, 3 Lonnette S. Diggs, 1 George
SUPPLEMENTARY INFORMATION
 Complete but curtailed T-cell response to very-low-affinity antigen Dietmar Zehn, Sarah Y. Lee & Michael J. Bevan Supp. Fig. 1: TCR chain usage among endogenous K b /Ova reactive T cells. C57BL/6 mice
Complete but curtailed T-cell response to very-low-affinity antigen Dietmar Zehn, Sarah Y. Lee & Michael J. Bevan Supp. Fig. 1: TCR chain usage among endogenous K b /Ova reactive T cells. C57BL/6 mice
NIH Public Access Author Manuscript Nature. Author manuscript; available in PMC 2009 July 28.
 NIH Public Access Author Manuscript Published in final edited form as: Nature. 2009 January 29; 457(7229): 557 561. doi:10.1038/nature07665. Adaptive Immune Features of Natural Killer Cells Joseph C. Sun,
NIH Public Access Author Manuscript Published in final edited form as: Nature. 2009 January 29; 457(7229): 557 561. doi:10.1038/nature07665. Adaptive Immune Features of Natural Killer Cells Joseph C. Sun,
SUPPLEMENTARY FIGURE 1
 SUPPLEMENTARY FIGURE 1 A LN Cell count (1 ) 1 3 1 CD+ 1 1 CDL lo CD hi 1 CD+FoxP3+ 1 1 1 7 3 3 3 % of cells 9 7 7 % of cells CD+ 3 1 % of cells CDL lo CD hi 1 1 % of CD+ cells CD+FoxP3+ 3 1 % of CD+ T
SUPPLEMENTARY FIGURE 1 A LN Cell count (1 ) 1 3 1 CD+ 1 1 CDL lo CD hi 1 CD+FoxP3+ 1 1 1 7 3 3 3 % of cells 9 7 7 % of cells CD+ 3 1 % of cells CDL lo CD hi 1 1 % of CD+ cells CD+FoxP3+ 3 1 % of CD+ T
MATERIALS AND METHODS. Neutralizing antibodies specific to mouse Dll1, Dll4, J1 and J2 were prepared as described. 1,2 All
 MATERIALS AND METHODS Antibodies (Abs), flow cytometry analysis and cell lines Neutralizing antibodies specific to mouse Dll1, Dll4, J1 and J2 were prepared as described. 1,2 All other antibodies used
MATERIALS AND METHODS Antibodies (Abs), flow cytometry analysis and cell lines Neutralizing antibodies specific to mouse Dll1, Dll4, J1 and J2 were prepared as described. 1,2 All other antibodies used
Antigen Presentation and T Lymphocyte Activation. Abul K. Abbas UCSF. FOCiS
 1 Antigen Presentation and T Lymphocyte Activation Abul K. Abbas UCSF FOCiS 2 Lecture outline Dendritic cells and antigen presentation The role of the MHC T cell activation Costimulation, the B7:CD28 family
1 Antigen Presentation and T Lymphocyte Activation Abul K. Abbas UCSF FOCiS 2 Lecture outline Dendritic cells and antigen presentation The role of the MHC T cell activation Costimulation, the B7:CD28 family
NK cell flow cytometric assay In vivo DC viability and migration assay
 NK cell flow cytometric assay 6 NK cells were purified, by negative selection with the NK Cell Isolation Kit (Miltenyi iotec), from spleen and lymph nodes of 6 RAG1KO mice, injected the day before with
NK cell flow cytometric assay 6 NK cells were purified, by negative selection with the NK Cell Isolation Kit (Miltenyi iotec), from spleen and lymph nodes of 6 RAG1KO mice, injected the day before with
Immunology Lecture 4. Clinical Relevance of the Immune System
 Immunology Lecture 4 The Well Patient: How innate and adaptive immune responses maintain health - 13, pg 169-181, 191-195. Immune Deficiency - 15 Autoimmunity - 16 Transplantation - 17, pg 260-270 Tumor
Immunology Lecture 4 The Well Patient: How innate and adaptive immune responses maintain health - 13, pg 169-181, 191-195. Immune Deficiency - 15 Autoimmunity - 16 Transplantation - 17, pg 260-270 Tumor
Effector mechanisms of cell-mediated immunity: Properties of effector, memory and regulatory T cells
 ICI Basic Immunology course Effector mechanisms of cell-mediated immunity: Properties of effector, memory and regulatory T cells Abul K. Abbas, MD UCSF Stages in the development of T cell responses: induction
ICI Basic Immunology course Effector mechanisms of cell-mediated immunity: Properties of effector, memory and regulatory T cells Abul K. Abbas, MD UCSF Stages in the development of T cell responses: induction
Immunity and Cancer. Doriana Fruci. Lab di Immuno-Oncologia
 Immunity and Cancer Doriana Fruci Lab di Immuno-Oncologia Immune System is a network of cells, tissues and organs that work together to defend the body against attacks of foreign invaders (pathogens, cancer
Immunity and Cancer Doriana Fruci Lab di Immuno-Oncologia Immune System is a network of cells, tissues and organs that work together to defend the body against attacks of foreign invaders (pathogens, cancer
Naive and memory CD8 T cell responses after antigen stimulation in vivo
 University of Iowa Iowa Research Online Theses and Dissertations Summer 2011 Naive and memory CD8 T cell responses after antigen stimulation in vivo Matthew David Martin University of Iowa Copyright 2011
University of Iowa Iowa Research Online Theses and Dissertations Summer 2011 Naive and memory CD8 T cell responses after antigen stimulation in vivo Matthew David Martin University of Iowa Copyright 2011
Donor Lymphoid Organs Are a Major Site of Alloreactive T-Cell Priming Following Intestinal Transplantation
 American Journal of Transplantation 2006; 6: 2563 2571 Blackwell Munksgaard C 2006 The Authors Journal compilation C 2006 The American Society of Transplantation and the American Society of Transplant
American Journal of Transplantation 2006; 6: 2563 2571 Blackwell Munksgaard C 2006 The Authors Journal compilation C 2006 The American Society of Transplantation and the American Society of Transplant
SUPPLEMENTARY INFORMATION
 doi:1.138/nature1554 a TNF-α + in CD4 + cells [%] 1 GF SPF 6 b IL-1 + in CD4 + cells [%] 5 4 3 2 1 Supplementary Figure 1. Effect of microbiota on cytokine profiles of T cells in GALT. Frequencies of TNF-α
doi:1.138/nature1554 a TNF-α + in CD4 + cells [%] 1 GF SPF 6 b IL-1 + in CD4 + cells [%] 5 4 3 2 1 Supplementary Figure 1. Effect of microbiota on cytokine profiles of T cells in GALT. Frequencies of TNF-α
T Cell Activation, Costimulation and Regulation
 1 T Cell Activation, Costimulation and Regulation Abul K. Abbas, MD University of California San Francisco 2 Lecture outline T cell antigen recognition and activation Costimulation, the B7:CD28 family
1 T Cell Activation, Costimulation and Regulation Abul K. Abbas, MD University of California San Francisco 2 Lecture outline T cell antigen recognition and activation Costimulation, the B7:CD28 family
Determinants of Immunogenicity and Tolerance. Abul K. Abbas, MD Department of Pathology University of California San Francisco
 Determinants of Immunogenicity and Tolerance Abul K. Abbas, MD Department of Pathology University of California San Francisco EIP Symposium Feb 2016 Why do some people respond to therapeutic proteins?
Determinants of Immunogenicity and Tolerance Abul K. Abbas, MD Department of Pathology University of California San Francisco EIP Symposium Feb 2016 Why do some people respond to therapeutic proteins?
IMMUNOLOGICAL MEMORY. CD4 T Follicular Helper Cells. Memory CD8 T Cell Differentiation
 IMMUNOLOGICAL MEMORY CD4 T Follicular Helper Cells Memory CD8 T Cell Differentiation CD4 T Cell Differentiation Bcl-6 T-bet GATA-3 ROR t Foxp3 CD4 T follicular helper (Tfh) cells FUNCTION Provide essential
IMMUNOLOGICAL MEMORY CD4 T Follicular Helper Cells Memory CD8 T Cell Differentiation CD4 T Cell Differentiation Bcl-6 T-bet GATA-3 ROR t Foxp3 CD4 T follicular helper (Tfh) cells FUNCTION Provide essential
T Lymphocyte Activation and Costimulation. FOCiS. Lecture outline
 1 T Lymphocyte Activation and Costimulation Abul K. Abbas, MD UCSF FOCiS 2 Lecture outline T cell activation Costimulation, the B7:CD28 family Inhibitory receptors of T cells Targeting costimulators for
1 T Lymphocyte Activation and Costimulation Abul K. Abbas, MD UCSF FOCiS 2 Lecture outline T cell activation Costimulation, the B7:CD28 family Inhibitory receptors of T cells Targeting costimulators for
Lineage relationship and protective immunity of memory CD8 T cell subsets
 23 Nature Publishing Group http://www.nature.com/natureimmunology Lineage relationship and protective immunity of memory CD8 T cell subsets E. John Wherry 1 *,Volker Teichgräber 1 *,Todd C. Becker 1,David
23 Nature Publishing Group http://www.nature.com/natureimmunology Lineage relationship and protective immunity of memory CD8 T cell subsets E. John Wherry 1 *,Volker Teichgräber 1 *,Todd C. Becker 1,David
Supplementary Materials for
 www.sciencemag.org/content/348/6241/aaa825/suppl/dc1 Supplementary Materials for A mucosal vaccine against Chlamydia trachomatis generates two waves of protective memory T cells Georg Stary,* Andrew Olive,
www.sciencemag.org/content/348/6241/aaa825/suppl/dc1 Supplementary Materials for A mucosal vaccine against Chlamydia trachomatis generates two waves of protective memory T cells Georg Stary,* Andrew Olive,
MECHANISMS OF CELLULAR REJECTION IN ORGAN TRANSPLANTATION AN OVERVIEW
 MECHANISMS OF CELLULAR REJECTION IN ORGAN TRANSPLANTATION AN OVERVIEW YVON LEBRANCHU Service Néphrologie et Immunologie Clinique CHU TOURS ANTIGEN PRESENTING CELL CD4 + T CELL CYTOKINE PRODUCTION CLONAL
MECHANISMS OF CELLULAR REJECTION IN ORGAN TRANSPLANTATION AN OVERVIEW YVON LEBRANCHU Service Néphrologie et Immunologie Clinique CHU TOURS ANTIGEN PRESENTING CELL CD4 + T CELL CYTOKINE PRODUCTION CLONAL
Harnessing tolerance mechanisms with monoclonal antibodies Prof. Herman Waldmann
 Harnessing tolerance mechanisms Herman Waldmann Sir William Dunn School of Pathology, Oxford University, UK 1 1. Transplantation 2 Lymphocyte depletion strategies to minimise drug immunosuppression in
Harnessing tolerance mechanisms Herman Waldmann Sir William Dunn School of Pathology, Oxford University, UK 1 1. Transplantation 2 Lymphocyte depletion strategies to minimise drug immunosuppression in
TITLE: MODULATION OF T CELL TOLERANCE IN A MURINE MODEL FOR IMMUNOTHERAPY OF PROSTATIC ADENOCARCINOMA
 AD Award Number: DAMD17-01-1-0085 TITLE: MODULATION OF T CELL TOLERANCE IN A MURINE MODEL FOR IMMUNOTHERAPY OF PROSTATIC ADENOCARCINOMA PRINCIPAL INVESTIGATOR: ARTHUR A HURWITZ, Ph.d. CONTRACTING ORGANIZATION:
AD Award Number: DAMD17-01-1-0085 TITLE: MODULATION OF T CELL TOLERANCE IN A MURINE MODEL FOR IMMUNOTHERAPY OF PROSTATIC ADENOCARCINOMA PRINCIPAL INVESTIGATOR: ARTHUR A HURWITZ, Ph.d. CONTRACTING ORGANIZATION:
The Role of Ectopic Lymphoid Tissue in Allograft Rejection
 Yale University EliScholar A Digital Platform for Scholarly Publishing at Yale Yale Medicine Thesis Digital Library School of Medicine 11-15-2006 The Role of Ectopic Lymphoid Tissue in Allograft Rejection
Yale University EliScholar A Digital Platform for Scholarly Publishing at Yale Yale Medicine Thesis Digital Library School of Medicine 11-15-2006 The Role of Ectopic Lymphoid Tissue in Allograft Rejection
SUPPLEMENTARY INFORMATION
 Supplemental Figure 1. Furin is efficiently deleted in CD4 + and CD8 + T cells. a, Western blot for furin and actin proteins in CD4cre-fur f/f and fur f/f Th1 cells. Wild-type and furin-deficient CD4 +
Supplemental Figure 1. Furin is efficiently deleted in CD4 + and CD8 + T cells. a, Western blot for furin and actin proteins in CD4cre-fur f/f and fur f/f Th1 cells. Wild-type and furin-deficient CD4 +
I. Lines of Defense Pathogen: Table 1: Types of Immune Mechanisms. Table 2: Innate Immunity: First Lines of Defense
 I. Lines of Defense Pathogen: Table 1: Types of Immune Mechanisms Table 2: Innate Immunity: First Lines of Defense Innate Immunity involves nonspecific physical & chemical barriers that are adapted for
I. Lines of Defense Pathogen: Table 1: Types of Immune Mechanisms Table 2: Innate Immunity: First Lines of Defense Innate Immunity involves nonspecific physical & chemical barriers that are adapted for
Shiv Pillai Ragon Institute, Massachusetts General Hospital Harvard Medical School
 CTLs, Natural Killers and NKTs 1 Shiv Pillai Ragon Institute, Massachusetts General Hospital Harvard Medical School CTL inducing tumor apoptosis 3 Lecture outline CD8 + Cytotoxic T lymphocytes (CTL) Activation/differentiation
CTLs, Natural Killers and NKTs 1 Shiv Pillai Ragon Institute, Massachusetts General Hospital Harvard Medical School CTL inducing tumor apoptosis 3 Lecture outline CD8 + Cytotoxic T lymphocytes (CTL) Activation/differentiation
Eosinophils are required. for the maintenance of plasma cells in the bone marrow
 Eosinophils are required for the maintenance of plasma cells in the bone marrow Van Trung Chu, Anja Fröhlich, Gudrun Steinhauser, Tobias Scheel, Toralf Roch, Simon Fillatreau, James J. Lee, Max Löhning
Eosinophils are required for the maintenance of plasma cells in the bone marrow Van Trung Chu, Anja Fröhlich, Gudrun Steinhauser, Tobias Scheel, Toralf Roch, Simon Fillatreau, James J. Lee, Max Löhning
T-cell activation T cells migrate to secondary lymphoid tissues where they interact with antigen, antigen-presenting cells, and other lymphocytes:
 Interactions between innate immunity & adaptive immunity What happens to T cells after they leave the thymus? Naïve T cells exit the thymus and enter the bloodstream. If they remain in the bloodstream,
Interactions between innate immunity & adaptive immunity What happens to T cells after they leave the thymus? Naïve T cells exit the thymus and enter the bloodstream. If they remain in the bloodstream,
T-cell activation T cells migrate to secondary lymphoid tissues where they interact with antigen, antigen-presenting cells, and other lymphocytes:
 Interactions between innate immunity & adaptive immunity What happens to T cells after they leave the thymus? Naïve T cells exit the thymus and enter the bloodstream. If they remain in the bloodstream,
Interactions between innate immunity & adaptive immunity What happens to T cells after they leave the thymus? Naïve T cells exit the thymus and enter the bloodstream. If they remain in the bloodstream,
Supplementary Figure 1. Characterization of basophils after reconstitution of SCID mice
 Supplementary figure legends Supplementary Figure 1. Characterization of after reconstitution of SCID mice with CD4 + CD62L + T cells. (A-C) SCID mice (n = 6 / group) were reconstituted with 2 x 1 6 CD4
Supplementary figure legends Supplementary Figure 1. Characterization of after reconstitution of SCID mice with CD4 + CD62L + T cells. (A-C) SCID mice (n = 6 / group) were reconstituted with 2 x 1 6 CD4
alveolar macrophages (AMs) after 24 hours of in vitro culture in complete medium
 Online Supplement for: NF-κB ACTIVATION IN ALVEOLAR MACROPHAGES REQUIRES IκB KINASE-β, BUT NOT NF-κB INDUCING KINASE Supershift and Competition Assays for NF-κB Competition and supershift assays were performed
Online Supplement for: NF-κB ACTIVATION IN ALVEOLAR MACROPHAGES REQUIRES IκB KINASE-β, BUT NOT NF-κB INDUCING KINASE Supershift and Competition Assays for NF-κB Competition and supershift assays were performed
Adaptive immune responses: T cell-mediated immunity
 MICR2209 Adaptive immune responses: T cell-mediated immunity Dr Allison Imrie allison.imrie@uwa.edu.au 1 Synopsis: In this lecture we will discuss the T-cell mediated immune response, how it is activated,
MICR2209 Adaptive immune responses: T cell-mediated immunity Dr Allison Imrie allison.imrie@uwa.edu.au 1 Synopsis: In this lecture we will discuss the T-cell mediated immune response, how it is activated,
Dendritic cells in cancer immunotherapy Aimin Jiang
 Dendritic cells in cancer immunotherapy Aimin Jiang Feb. 11, 2014 Dendritic cells at the interface of innate and adaptive immune responses Dendritic cells: initiators of adaptive immune responses Dendritic
Dendritic cells in cancer immunotherapy Aimin Jiang Feb. 11, 2014 Dendritic cells at the interface of innate and adaptive immune responses Dendritic cells: initiators of adaptive immune responses Dendritic
Medical Virology Immunology. Dr. Sameer Naji, MB, BCh, PhD (UK) Head of Basic Medical Sciences Dept. Faculty of Medicine The Hashemite University
 Medical Virology Immunology Dr. Sameer Naji, MB, BCh, PhD (UK) Head of Basic Medical Sciences Dept. Faculty of Medicine The Hashemite University Human blood cells Phases of immune responses Microbe Naïve
Medical Virology Immunology Dr. Sameer Naji, MB, BCh, PhD (UK) Head of Basic Medical Sciences Dept. Faculty of Medicine The Hashemite University Human blood cells Phases of immune responses Microbe Naïve
The Immune System: Innate and Adaptive Body Defenses Outline PART 1: INNATE DEFENSES 21.1 Surface barriers act as the first line of defense to keep
 The Immune System: Innate and Adaptive Body Defenses Outline PART 1: INNATE DEFENSES 21.1 Surface barriers act as the first line of defense to keep invaders out of the body (pp. 772 773; Fig. 21.1; Table
The Immune System: Innate and Adaptive Body Defenses Outline PART 1: INNATE DEFENSES 21.1 Surface barriers act as the first line of defense to keep invaders out of the body (pp. 772 773; Fig. 21.1; Table
A different view on immunity Dempsey, Claire; Jones, Nicholas
 A different view on immunity Dempsey, Claire; Jones, Nicholas DOI: 10.1097/TP.0000000000000738 License: None: All rights reserved Document Version Peer reviewed version Citation for published version (Harvard):
A different view on immunity Dempsey, Claire; Jones, Nicholas DOI: 10.1097/TP.0000000000000738 License: None: All rights reserved Document Version Peer reviewed version Citation for published version (Harvard):
Supplementary Information:
 Supplementary Information: Follicular regulatory T cells with Bcl6 expression suppress germinal center reactions by Yeonseok Chung, Shinya Tanaka, Fuliang Chu, Roza Nurieva, Gustavo J. Martinez, Seema
Supplementary Information: Follicular regulatory T cells with Bcl6 expression suppress germinal center reactions by Yeonseok Chung, Shinya Tanaka, Fuliang Chu, Roza Nurieva, Gustavo J. Martinez, Seema
Blocking antibodies and peptides. Rat anti-mouse PD-1 (29F.1A12, rat IgG2a, k), PD-
 Supplementary Methods Blocking antibodies and peptides. Rat anti-mouse PD-1 (29F.1A12, rat IgG2a, k), PD- L1 (10F.9G2, rat IgG2b, k), and PD-L2 (3.2, mouse IgG1) have been described (24). Anti-CTLA-4 (clone
Supplementary Methods Blocking antibodies and peptides. Rat anti-mouse PD-1 (29F.1A12, rat IgG2a, k), PD- L1 (10F.9G2, rat IgG2b, k), and PD-L2 (3.2, mouse IgG1) have been described (24). Anti-CTLA-4 (clone
Third line of Defense
 Chapter 15 Specific Immunity and Immunization Topics -3 rd of Defense - B cells - T cells - Specific Immunities Third line of Defense Specific immunity is a complex interaction of immune cells (leukocytes)
Chapter 15 Specific Immunity and Immunization Topics -3 rd of Defense - B cells - T cells - Specific Immunities Third line of Defense Specific immunity is a complex interaction of immune cells (leukocytes)
Origin of CD8 + Effector and Memory T Cell Subsets
 Cellular & Molecular Immunology 399 Review Origin of CD8 + Effector and Memory T Cell Subsets Christian Stemberger 1, Michael Neuenhahn 1, Veit R. Buchholz 1 1, 2, 3 and Dirk H. Busch It is well accepted
Cellular & Molecular Immunology 399 Review Origin of CD8 + Effector and Memory T Cell Subsets Christian Stemberger 1, Michael Neuenhahn 1, Veit R. Buchholz 1 1, 2, 3 and Dirk H. Busch It is well accepted
Distinct subsets of lymphocytes depend on IL-15 (1, 2). Mice
 ITK and IL-15 support two distinct subsets of CD8 T cells Sigrid Dubois, Thomas A. Waldmann*, and Jürgen R. Müller* Metabolism Branch, Center for Cancer Research, National Cancer Institute, National Institutes
ITK and IL-15 support two distinct subsets of CD8 T cells Sigrid Dubois, Thomas A. Waldmann*, and Jürgen R. Müller* Metabolism Branch, Center for Cancer Research, National Cancer Institute, National Institutes
The Journal of Experimental Medicine
 Are Major Histocompatibility Complex Molecules Involved in the Survival of Naive CD4 T Cells? Isabelle Grandjean, 1 Livine Duban, 1 Elizabeth A. Bonney, 2,3 Erwan Corcuff, 4 James P. Di Santo, 4 Polly
Are Major Histocompatibility Complex Molecules Involved in the Survival of Naive CD4 T Cells? Isabelle Grandjean, 1 Livine Duban, 1 Elizabeth A. Bonney, 2,3 Erwan Corcuff, 4 James P. Di Santo, 4 Polly
Role for CD40 and CD40L Expression in Generating CD8 T Cell Response to Minor Histcompatibility Antigen, H60
 Role for CD40 and CD40L Expression in Generating CD8 T Cell Response to Minor Histcompatibility Antigen, H60 Kyoung Min Jung and Eun Young Choi Graduate Program of Immunology, Seoul National University
Role for CD40 and CD40L Expression in Generating CD8 T Cell Response to Minor Histcompatibility Antigen, H60 Kyoung Min Jung and Eun Young Choi Graduate Program of Immunology, Seoul National University
CONTRACTING ORGANIZATION: Johns Hopkins University School of Medicine Baltimore, MD 21205
 AD Award Number: DAMD7---7 TITLE: Development of Artificial Antigen Presenting Cells for Prostate Cancer Immunotherapy PRINCIPAL INVESTIGATOR: Jonathan P. Schneck, M.D., Ph.D. Mathias Oelke, Ph.D. CONTRACTING
AD Award Number: DAMD7---7 TITLE: Development of Artificial Antigen Presenting Cells for Prostate Cancer Immunotherapy PRINCIPAL INVESTIGATOR: Jonathan P. Schneck, M.D., Ph.D. Mathias Oelke, Ph.D. CONTRACTING
Supplemental Figure 1
 Supplemental Figure 1 1a 1c PD-1 MFI fold change 6 5 4 3 2 1 IL-1α IL-2 IL-4 IL-6 IL-1 IL-12 IL-13 IL-15 IL-17 IL-18 IL-21 IL-23 IFN-α Mut Human PD-1 promoter SBE-D 5 -GTCTG- -1.2kb SBE-P -CAGAC- -1.kb
Supplemental Figure 1 1a 1c PD-1 MFI fold change 6 5 4 3 2 1 IL-1α IL-2 IL-4 IL-6 IL-1 IL-12 IL-13 IL-15 IL-17 IL-18 IL-21 IL-23 IFN-α Mut Human PD-1 promoter SBE-D 5 -GTCTG- -1.2kb SBE-P -CAGAC- -1.kb
FIT Board Review Corner March 2016
 FIT Board Review Corner March 2016 Welcome to the FIT Board Review Corner, prepared by Sarah Spriet, DO, and Tammy Peng, MD, senior and junior representatives of ACAAI's Fellows-In-Training (FITs) to the
FIT Board Review Corner March 2016 Welcome to the FIT Board Review Corner, prepared by Sarah Spriet, DO, and Tammy Peng, MD, senior and junior representatives of ACAAI's Fellows-In-Training (FITs) to the
Tertiary Lymphoid Tissues Generate Effector and Memory T Cells That Lead to Allograft Rejection
 American Journal of Transplantation 2007; 7: 1071 1079 Blackwell Munksgaard C 2007 The Authors Journal compilation C 2007 The American Society of Transplantation and the American Society of Transplant
American Journal of Transplantation 2007; 7: 1071 1079 Blackwell Munksgaard C 2007 The Authors Journal compilation C 2007 The American Society of Transplantation and the American Society of Transplant
Micro 204. Cytotoxic T Lymphocytes (CTL) Lewis Lanier
 Micro 204 Cytotoxic T Lymphocytes (CTL) Lewis Lanier Lewis.Lanier@ucsf.edu Lymphocyte-mediated Cytotoxicity CD8 + αβ-tcr + T cells CD4 + αβ-tcr + T cells γδ-tcr + T cells Natural Killer cells CD8 + αβ-tcr
Micro 204 Cytotoxic T Lymphocytes (CTL) Lewis Lanier Lewis.Lanier@ucsf.edu Lymphocyte-mediated Cytotoxicity CD8 + αβ-tcr + T cells CD4 + αβ-tcr + T cells γδ-tcr + T cells Natural Killer cells CD8 + αβ-tcr
COURSE: Medical Microbiology, PAMB 650/720 - Fall 2008 Lecture 16
 COURSE: Medical Microbiology, PAMB 650/720 - Fall 2008 Lecture 16 Tumor Immunology M. Nagarkatti Teaching Objectives: Introduction to Cancer Immunology Know the antigens expressed by cancer cells Understand
COURSE: Medical Microbiology, PAMB 650/720 - Fall 2008 Lecture 16 Tumor Immunology M. Nagarkatti Teaching Objectives: Introduction to Cancer Immunology Know the antigens expressed by cancer cells Understand
NKTR-255: Accessing The Immunotherapeutic Potential Of IL-15 for NK Cell Therapies
 NKTR-255: Accessing The Immunotherapeutic Potential Of IL-15 for NK Cell Therapies Saul Kivimäe Senior Scientist, Research Biology Nektar Therapeutics NK Cell-Based Cancer Immunotherapy, September 26-27,
NKTR-255: Accessing The Immunotherapeutic Potential Of IL-15 for NK Cell Therapies Saul Kivimäe Senior Scientist, Research Biology Nektar Therapeutics NK Cell-Based Cancer Immunotherapy, September 26-27,
Nature Medicine: doi: /nm.3922
 Title: Glucocorticoid-induced tumor necrosis factor receptor-related protein co-stimulation facilitates tumor regression by inducing IL-9-producing helper T cells Authors: Il-Kyu Kim, Byung-Seok Kim, Choong-Hyun
Title: Glucocorticoid-induced tumor necrosis factor receptor-related protein co-stimulation facilitates tumor regression by inducing IL-9-producing helper T cells Authors: Il-Kyu Kim, Byung-Seok Kim, Choong-Hyun
Supplemental Information. Gut Microbiota Promotes Hematopoiesis to Control Bacterial Infection. Cell Host & Microbe, Volume 15
 Cell Host & Microbe, Volume 15 Supplemental Information Gut Microbiota Promotes Hematopoiesis to Control Bacterial Infection Arya Khosravi, Alberto Yáñez, Jeremy G. Price, Andrew Chow, Miriam Merad, Helen
Cell Host & Microbe, Volume 15 Supplemental Information Gut Microbiota Promotes Hematopoiesis to Control Bacterial Infection Arya Khosravi, Alberto Yáñez, Jeremy G. Price, Andrew Chow, Miriam Merad, Helen
RAISON D ETRE OF THE IMMUNE SYSTEM:
 RAISON D ETRE OF THE IMMUNE SYSTEM: To Distinguish Self from Non-Self Thereby Protecting Us From Our Hostile Environment. Innate Immunity Acquired Immunity Innate immunity: (Antigen nonspecific) defense
RAISON D ETRE OF THE IMMUNE SYSTEM: To Distinguish Self from Non-Self Thereby Protecting Us From Our Hostile Environment. Innate Immunity Acquired Immunity Innate immunity: (Antigen nonspecific) defense
FINAL REPORT For Japan-Korea Joint Research Project AREA
 (Form4-2) FINAL REPORT For Japan-Korea Joint Research Project AREA 1. Mathematics & Physics 2. Chemistry & Material Science 3. Biology 4. Informatics & Mechatronics 5. Geo-Science & Space Science O 6.
(Form4-2) FINAL REPORT For Japan-Korea Joint Research Project AREA 1. Mathematics & Physics 2. Chemistry & Material Science 3. Biology 4. Informatics & Mechatronics 5. Geo-Science & Space Science O 6.
Supplementary Figure Legends. group) and analyzed for Siglec-G expression utilizing a monoclonal antibody to Siglec-G (clone SH2.1).
 Supplementary Figure Legends Supplemental Figure : Naïve T cells express Siglec-G. Splenocytes were isolated from WT B or Siglec-G -/- animals that have not been transplanted (n= per group) and analyzed
Supplementary Figure Legends Supplemental Figure : Naïve T cells express Siglec-G. Splenocytes were isolated from WT B or Siglec-G -/- animals that have not been transplanted (n= per group) and analyzed
Supplementary Data. Treg phenotype
 Supplementary Data Additional Experiment An additional experiment was performed using cryopreserved peripheral blood mononuclear cells (PBMC) derived from five renal cell carcinoma (RCC) patients [see
Supplementary Data Additional Experiment An additional experiment was performed using cryopreserved peripheral blood mononuclear cells (PBMC) derived from five renal cell carcinoma (RCC) patients [see
Immunology and Immunotherapy 101 for the Non-Immunologist
 Immunology and Immunotherapy 101 for the Non-Immunologist Stephen P. Schoenberger, Ph.D La Jolla Institute for Allergy and Immunology & UCSD Moores Cancer Center Disclosures Human Longevity Inc: Salary
Immunology and Immunotherapy 101 for the Non-Immunologist Stephen P. Schoenberger, Ph.D La Jolla Institute for Allergy and Immunology & UCSD Moores Cancer Center Disclosures Human Longevity Inc: Salary
Supplemental Table I.
 Supplemental Table I Male / Mean ± SEM n Mean ± SEM n Body weight, g 29.2±0.4 17 29.7±0.5 17 Total cholesterol, mg/dl 534.0±30.8 17 561.6±26.1 17 HDL-cholesterol, mg/dl 9.6±0.8 17 10.1±0.7 17 Triglycerides,
Supplemental Table I Male / Mean ± SEM n Mean ± SEM n Body weight, g 29.2±0.4 17 29.7±0.5 17 Total cholesterol, mg/dl 534.0±30.8 17 561.6±26.1 17 HDL-cholesterol, mg/dl 9.6±0.8 17 10.1±0.7 17 Triglycerides,
IMMUNOLOGICAL MEMORY. May 28, 2017
 IMMUNOLOGICAL MEMORY May 28, 2017 Francesca Di Rosa Institute of Molecular Biology and Pathology National Research Council francesca.dirosa@uniroma1.it Pathogen infection and adaptive immune response Primary
IMMUNOLOGICAL MEMORY May 28, 2017 Francesca Di Rosa Institute of Molecular Biology and Pathology National Research Council francesca.dirosa@uniroma1.it Pathogen infection and adaptive immune response Primary
Nature Immunology: doi: /ni Supplementary Figure 1. Gene expression profile of CD4 + T cells and CTL responses in Bcl6-deficient mice.
 Supplementary Figure 1 Gene expression profile of CD4 + T cells and CTL responses in Bcl6-deficient mice. (a) Gene expression profile in the resting CD4 + T cells were analyzed by an Affymetrix microarray
Supplementary Figure 1 Gene expression profile of CD4 + T cells and CTL responses in Bcl6-deficient mice. (a) Gene expression profile in the resting CD4 + T cells were analyzed by an Affymetrix microarray
IMMUNOBIOLOGY OF TRANSPLANTATION. Wasim Dar
 IMMUNOBIOLOGY OF TRANSPLANTATION Wasim Dar Immunobiology of Transplantation Overview Transplantation: A complex immunologic process Contributions Innate Immunity Adaptive immunity T Cells B Cells HLA Consequences
IMMUNOBIOLOGY OF TRANSPLANTATION Wasim Dar Immunobiology of Transplantation Overview Transplantation: A complex immunologic process Contributions Innate Immunity Adaptive immunity T Cells B Cells HLA Consequences
Canberra, Australia). CD11c-DTR-OVA-GFP (B6.CD11c-OVA), B6.luc + and. Cancer Research Center, Germany). B6 or BALB/c.FoxP3-DTR-GFP mice were
 Supplemental Materials and Methods Mice Female C57BL/6 (B6, I-E null, H-2 b ), BALB/c (H-2 d ) + ), FVB/N (H-2 q, I-E null, CD45.1 + ), and B6D2F1 (H-2 b/d ) mice were purchased from the Animal Resources
Supplemental Materials and Methods Mice Female C57BL/6 (B6, I-E null, H-2 b ), BALB/c (H-2 d ) + ), FVB/N (H-2 q, I-E null, CD45.1 + ), and B6D2F1 (H-2 b/d ) mice were purchased from the Animal Resources
Reviewers' Comments: Reviewer #1 (Remarks to the Author)
 Reviewers' Comments: Reviewer #1 (Remarks to the Author) The manuscript by Zhuang and colleagues characterizes in depth the phenotype, origin and role of mononuclear phagocytes infiltrating murine cardiac
Reviewers' Comments: Reviewer #1 (Remarks to the Author) The manuscript by Zhuang and colleagues characterizes in depth the phenotype, origin and role of mononuclear phagocytes infiltrating murine cardiac
Naive, memory and regulatory T lymphocytes populations analysis
 Naive, memory and regulatory T lymphocytes populations analysis Jaen Olivier, PhD ojaen@beckmancoulter.com Cellular Analysis application specialist Beckman Coulter France Introduction Flow cytometric analysis
Naive, memory and regulatory T lymphocytes populations analysis Jaen Olivier, PhD ojaen@beckmancoulter.com Cellular Analysis application specialist Beckman Coulter France Introduction Flow cytometric analysis
Citation Annals of transplantation (2014), 1. use them commercially.
 Induction of alloantigen-specific C Title cells by alloantigen immunization a a pilot study in murine transplanta cardiac allografts. Author(s) Hori, Tomohide; Kuribayashi, Kagema Wang, Linan; Torii, Mie;
Induction of alloantigen-specific C Title cells by alloantigen immunization a a pilot study in murine transplanta cardiac allografts. Author(s) Hori, Tomohide; Kuribayashi, Kagema Wang, Linan; Torii, Mie;
Cell isolation. Spleen and lymph nodes (axillary, inguinal) were removed from mice
 Supplementary Methods: Cell isolation. Spleen and lymph nodes (axillary, inguinal) were removed from mice and gently meshed in DMEM containing 10% FBS to prepare for single cell suspensions. CD4 + CD25
Supplementary Methods: Cell isolation. Spleen and lymph nodes (axillary, inguinal) were removed from mice and gently meshed in DMEM containing 10% FBS to prepare for single cell suspensions. CD4 + CD25
Lecture 9: T-cell Mediated Immunity
 Lecture 9: T-cell Mediated Immunity Questions to Consider How do T cells know where to go? Questions to Consider How do T cells know where to go? How does antigen get targeted to a T cell expressing the
Lecture 9: T-cell Mediated Immunity Questions to Consider How do T cells know where to go? Questions to Consider How do T cells know where to go? How does antigen get targeted to a T cell expressing the
Phenotypical and Functional Analysis of Peripheral T Cells in Foxn1 Transgenic Mice: Effects of Aging
 Loyola University Chicago Loyola ecommons Master's Theses Theses and Dissertations 2010 Phenotypical and Functional Analysis of Peripheral T Cells in Foxn1 Transgenic Mice: Effects of Aging Paulette Krishack
Loyola University Chicago Loyola ecommons Master's Theses Theses and Dissertations 2010 Phenotypical and Functional Analysis of Peripheral T Cells in Foxn1 Transgenic Mice: Effects of Aging Paulette Krishack
This information is current as of March 2, Roberto Carrio, Oliver F. Bathe and Thomas R. Malek.
 This information is current as of March 2, 2014. References Subscriptions Permissions Email Alerts Initial Antigen Encounter Programs CD8 + T Cells Competent to Develop into Memory Cells That Are Activated
This information is current as of March 2, 2014. References Subscriptions Permissions Email Alerts Initial Antigen Encounter Programs CD8 + T Cells Competent to Develop into Memory Cells That Are Activated
Supporting Information
 Supporting Information lpek et al. 1.173/pnas.1121217 SI Materials and Methods Mice. cell knockout, inos / (Taconic arms), Rag1 /, INγR /, and IL-12p4 / mice (The Jackson Laboratory) were maintained and/or
Supporting Information lpek et al. 1.173/pnas.1121217 SI Materials and Methods Mice. cell knockout, inos / (Taconic arms), Rag1 /, INγR /, and IL-12p4 / mice (The Jackson Laboratory) were maintained and/or
PBS Class #2 Introduction to the Immune System part II Suggested reading: Abbas, pgs , 27-30
 PBS 803 - Class #2 Introduction to the Immune System part II Suggested reading: Abbas, pgs. 15-25, 27-30 Learning Objectives Compare and contrast the maturation of B and T lymphocytes Compare and contrast
PBS 803 - Class #2 Introduction to the Immune System part II Suggested reading: Abbas, pgs. 15-25, 27-30 Learning Objectives Compare and contrast the maturation of B and T lymphocytes Compare and contrast
D CD8 T cell number (x10 6 )
 IFNγ Supplemental Figure 1. CD T cell number (x1 6 ) 18 15 1 9 6 3 CD CD T cells CD6L C CD5 CD T cells CD6L D CD8 T cell number (x1 6 ) 1 8 6 E CD CD8 T cells CD6L F Log(1)CFU/g Feces 1 8 6 p
IFNγ Supplemental Figure 1. CD T cell number (x1 6 ) 18 15 1 9 6 3 CD CD T cells CD6L C CD5 CD T cells CD6L D CD8 T cell number (x1 6 ) 1 8 6 E CD CD8 T cells CD6L F Log(1)CFU/g Feces 1 8 6 p
GRAFT ANTIGEN-SPECIFIC CD4+ T CELLS REQUIRE CD154 EXPRESSION TO CLONALLY EXPAND AND DIFFERENTIATE. TO THE TH1 PHENOTYPE AND INITIATE mtor DEPENDANT
 GRAFT ANTIGEN-SPECIFIC CD4+ T CELLS REQUIRE CD154 EXPRESSION TO CLONALLY EXPAND AND DIFFERENTIATE TO THE TH1 PHENOTYPE AND INITIATE mtor DEPENDANT INTIMAL HYPERPLASIA IN CARDIAC ALLOGRAFTS A DISSERTATION
GRAFT ANTIGEN-SPECIFIC CD4+ T CELLS REQUIRE CD154 EXPRESSION TO CLONALLY EXPAND AND DIFFERENTIATE TO THE TH1 PHENOTYPE AND INITIATE mtor DEPENDANT INTIMAL HYPERPLASIA IN CARDIAC ALLOGRAFTS A DISSERTATION
B6/COLODR/SPL/11C/83/LAP/#2.006 B6/COLODR/SPL/11C/86/LAP/#2.016 CD11C B6/COLODR/SPL/11C/80/LAP/#2.011 CD11C
 CD3-specific antibody-induced immune tolerance and suppression of autoimmune encephalomyelitis involves TGF-β production through phagocytes digesting apoptotic T cells Sylvain Perruche 1,3, Pin Zhang 1,
CD3-specific antibody-induced immune tolerance and suppression of autoimmune encephalomyelitis involves TGF-β production through phagocytes digesting apoptotic T cells Sylvain Perruche 1,3, Pin Zhang 1,
Tumors arise from accumulated genetic mutations. Tumor Immunology (Cancer)
 Tumor Immunology (Cancer) Tumors arise from accumulated genetic mutations Robert Beatty MCB150 Mutations Usually have >6 mutations in both activation/growth factors and tumor suppressor genes. Types of
Tumor Immunology (Cancer) Tumors arise from accumulated genetic mutations Robert Beatty MCB150 Mutations Usually have >6 mutations in both activation/growth factors and tumor suppressor genes. Types of
CD8 T cells leave the thymus 4 5 days after undergoing positive
 Exploring regulatory mechanisms of T cell contraction Martin Prlic and Michael J. Bevan 1 Department of Immunology and Howard Hughes Medical Institute, University of Washington, Seattle, WA 98195 Contributed
Exploring regulatory mechanisms of T cell contraction Martin Prlic and Michael J. Bevan 1 Department of Immunology and Howard Hughes Medical Institute, University of Washington, Seattle, WA 98195 Contributed
FOCiS. Lecture outline. The immunological equilibrium: balancing lymphocyte activation and control. Immunological tolerance and immune regulation -- 1
 1 Immunological tolerance and immune regulation -- 1 Abul K. Abbas UCSF FOCiS 2 Lecture outline Principles of immune regulation Self-tolerance; mechanisms of central and peripheral tolerance Inhibitory
1 Immunological tolerance and immune regulation -- 1 Abul K. Abbas UCSF FOCiS 2 Lecture outline Principles of immune regulation Self-tolerance; mechanisms of central and peripheral tolerance Inhibitory
After severe T cell depletion, induced by ionizing radiation,
 Homeostatic T cell proliferation in a T cell-dendritic cell coculture system Qing Ge, Deborah Palliser, Herman N. Eisen, and Jianzhu Chen* Center for Cancer Research and Department of Biology, Massachusetts
Homeostatic T cell proliferation in a T cell-dendritic cell coculture system Qing Ge, Deborah Palliser, Herman N. Eisen, and Jianzhu Chen* Center for Cancer Research and Department of Biology, Massachusetts
Therapeutic PD L1 and LAG 3 blockade rapidly clears established blood stage Plasmodium infection
 Supplementary Information Therapeutic PD L1 and LAG 3 blockade rapidly clears established blood stage Plasmodium infection Noah S. Butler, Jacqueline Moebius, Lecia L. Pewe, Boubacar Traore, Ogobara K.
Supplementary Information Therapeutic PD L1 and LAG 3 blockade rapidly clears established blood stage Plasmodium infection Noah S. Butler, Jacqueline Moebius, Lecia L. Pewe, Boubacar Traore, Ogobara K.
Supplementary Figures
 Supplementary Figures Supplementary Figure 1. NKT ligand-loaded tumour antigen-presenting B cell- and monocyte-based vaccine induces NKT, NK and CD8 T cell responses. (A) The cytokine profiles of liver
Supplementary Figures Supplementary Figure 1. NKT ligand-loaded tumour antigen-presenting B cell- and monocyte-based vaccine induces NKT, NK and CD8 T cell responses. (A) The cytokine profiles of liver
General Overview of Immunology. Kimberly S. Schluns, Ph.D. Associate Professor Department of Immunology UT MD Anderson Cancer Center
 General Overview of Immunology Kimberly S. Schluns, Ph.D. Associate Professor Department of Immunology UT MD Anderson Cancer Center Objectives Describe differences between innate and adaptive immune responses
General Overview of Immunology Kimberly S. Schluns, Ph.D. Associate Professor Department of Immunology UT MD Anderson Cancer Center Objectives Describe differences between innate and adaptive immune responses
Attribution: University of Michigan Medical School, Department of Microbiology and Immunology
 Attribution: University of Michigan Medical School, Department of Microbiology and Immunology License: Unless otherwise noted, this material is made available under the terms of the Creative Commons Attribution
Attribution: University of Michigan Medical School, Department of Microbiology and Immunology License: Unless otherwise noted, this material is made available under the terms of the Creative Commons Attribution
NKTR-255: Accessing IL-15 Therapeutic Potential through Robust and Sustained Engagement of Innate and Adaptive Immunity
 NKTR-255: Accessing IL-15 Therapeutic Potential through Robust and Sustained Engagement of Innate and Adaptive Immunity Peiwen Kuo Scientist, Research Biology Nektar Therapeutics August 31 st, 2018 Emerging
NKTR-255: Accessing IL-15 Therapeutic Potential through Robust and Sustained Engagement of Innate and Adaptive Immunity Peiwen Kuo Scientist, Research Biology Nektar Therapeutics August 31 st, 2018 Emerging
Memory CD4 + T cells do not induce graft-versus-host disease
 Memory CD4 + T cells do not induce graft-versus-host disease See the related Commentary beginning on page 25. Britt E. Anderson, 1 Jennifer McNiff, 2 Jun Yan, 3 Hester Doyle, 3 Mark Mamula, 3 Mark J. Shlomchik,
Memory CD4 + T cells do not induce graft-versus-host disease See the related Commentary beginning on page 25. Britt E. Anderson, 1 Jennifer McNiff, 2 Jun Yan, 3 Hester Doyle, 3 Mark Mamula, 3 Mark J. Shlomchik,
Supporting Information
 Supporting Information Idoyaga et al. 10.1073/pnas.0812247106 SSC a) Single cell suspension 99 Aqua b) Live cells 96 -W c) Singlets 92 -A CD19+ER119 d) CD19 ER119 cells 97 CD3 e) CD3 cells 27 f) DX5 cells
Supporting Information Idoyaga et al. 10.1073/pnas.0812247106 SSC a) Single cell suspension 99 Aqua b) Live cells 96 -W c) Singlets 92 -A CD19+ER119 d) CD19 ER119 cells 97 CD3 e) CD3 cells 27 f) DX5 cells
Functional differences between memory and naive CD8 T cells
 Proc. Natl. Acad. Sci. USA Vol. 96, pp. 2976 2981, March 1999 Immunology Functional differences between memory and naive CD8 T cells BRYAN K. CHO, CHANGYU WANG,* SATOSHI SUGAWA, HERMAN N. EISEN, AND JIANZHU
Proc. Natl. Acad. Sci. USA Vol. 96, pp. 2976 2981, March 1999 Immunology Functional differences between memory and naive CD8 T cells BRYAN K. CHO, CHANGYU WANG,* SATOSHI SUGAWA, HERMAN N. EISEN, AND JIANZHU
Active STAT5 promotes long lived cytotoxic CD8 T cells that induce regression of autochthonous mouse melanoma.
 Active STAT5 promotes long lived cytotoxic CD8 T cells that induce regression of autochthonous mouse melanoma. SITC 211 Grégory Verdeil No relationships to disclose T cell adoptive transfer for cancer
Active STAT5 promotes long lived cytotoxic CD8 T cells that induce regression of autochthonous mouse melanoma. SITC 211 Grégory Verdeil No relationships to disclose T cell adoptive transfer for cancer
Memory T cells in organ transplantation: progress and challenges
 Memory T cells in organ transplantation: progress and challenges Jaclyn R. Espinosa, Emory University Kannan P. Samy, Duke University Allan Kirk, Emory University Journal Title: Nature Reviews Nephrology
Memory T cells in organ transplantation: progress and challenges Jaclyn R. Espinosa, Emory University Kannan P. Samy, Duke University Allan Kirk, Emory University Journal Title: Nature Reviews Nephrology
Detailed step-by-step operating procedures for NK cell and CTL degranulation assays
 Supplemental methods Detailed step-by-step operating procedures for NK cell and CTL degranulation assays Materials PBMC isolated from patients, relatives and healthy donors as control K562 cells (ATCC,
Supplemental methods Detailed step-by-step operating procedures for NK cell and CTL degranulation assays Materials PBMC isolated from patients, relatives and healthy donors as control K562 cells (ATCC,
Viral Persistence Alters CD8 T-Cell Immunodominance and Tissue Distribution and Results in Distinct Stages of Functional Impairment
 JOURNAL OF VIROLOGY, Apr. 2003, p. 4911 4927 Vol. 77, No. 8 0022-538X/03/$08.00 0 DOI: 10.1128/JVI.77.8.4911 4927.2003 Copyright 2003, American Society for Microbiology. All Rights Reserved. Viral Persistence
JOURNAL OF VIROLOGY, Apr. 2003, p. 4911 4927 Vol. 77, No. 8 0022-538X/03/$08.00 0 DOI: 10.1128/JVI.77.8.4911 4927.2003 Copyright 2003, American Society for Microbiology. All Rights Reserved. Viral Persistence
B cell activation and antibody production. Abul K. Abbas UCSF
 1 B cell activation and antibody production Abul K. Abbas UCSF 2 Lecture outline B cell activation; the role of helper T cells in antibody production Therapeutic targeting of B cells 3 Principles of humoral
1 B cell activation and antibody production Abul K. Abbas UCSF 2 Lecture outline B cell activation; the role of helper T cells in antibody production Therapeutic targeting of B cells 3 Principles of humoral
Supplementary Figure 1. mrna expression of chitinase and chitinase-like protein in splenic immune cells. Each splenic immune cell population was
 Supplementary Figure 1. mrna expression of chitinase and chitinase-like protein in splenic immune cells. Each splenic immune cell population was sorted by FACS. Surface markers for sorting were CD11c +
Supplementary Figure 1. mrna expression of chitinase and chitinase-like protein in splenic immune cells. Each splenic immune cell population was sorted by FACS. Surface markers for sorting were CD11c +
Supplementary Figures
 Supplementary Figures Supplementary Fig. 1. Surface thiol groups and reduction of activated T cells. (a) Activated CD8 + T-cells have high expression levels of free thiol groups on cell surface proteins.
Supplementary Figures Supplementary Fig. 1. Surface thiol groups and reduction of activated T cells. (a) Activated CD8 + T-cells have high expression levels of free thiol groups on cell surface proteins.
Chapter 10 (pages ): Differentiation and Functions of CD4+ Effector T Cells Prepared by Kristen Dazy, MD, Scripps Clinic Medical Group
 FIT Board Review Corner September 2015 Welcome to the FIT Board Review Corner, prepared by Andrew Nickels, MD, and Sarah Spriet, DO, senior and junior representatives of ACAAI's Fellows-In-Training (FITs)
FIT Board Review Corner September 2015 Welcome to the FIT Board Review Corner, prepared by Andrew Nickels, MD, and Sarah Spriet, DO, senior and junior representatives of ACAAI's Fellows-In-Training (FITs)
Immunological memory.
 Chapter 11: Dynamics of Adaptive Immunity the answers will lie in the cytokines produced by the environment and by the T cells themselves, and in the affinity of the T-cell receptors for their antigens.
Chapter 11: Dynamics of Adaptive Immunity the answers will lie in the cytokines produced by the environment and by the T cells themselves, and in the affinity of the T-cell receptors for their antigens.
Regulation of anti-tumor immunity through migration of immune cell subsets within the tumor microenvironment Thomas F. Gajewski, M.D., Ph.D.
 Regulation of anti-tumor immunity through migration of immune cell subsets within the tumor microenvironment Thomas F. Gajewski, M.D., Ph.D. Professor, Departments of Pathology and Medicine Program Leader,
Regulation of anti-tumor immunity through migration of immune cell subsets within the tumor microenvironment Thomas F. Gajewski, M.D., Ph.D. Professor, Departments of Pathology and Medicine Program Leader,
Third line of Defense. Topic 8 Specific Immunity (adaptive) (18) 3 rd Line = Prophylaxis via Immunization!
 Topic 8 Specific Immunity (adaptive) (18) Topics - 3 rd Line of Defense - B cells - T cells - Specific Immunities 1 3 rd Line = Prophylaxis via Immunization! (a) A painting of Edward Jenner depicts a cow
Topic 8 Specific Immunity (adaptive) (18) Topics - 3 rd Line of Defense - B cells - T cells - Specific Immunities 1 3 rd Line = Prophylaxis via Immunization! (a) A painting of Edward Jenner depicts a cow
Effector T Cells and
 1 Effector T Cells and Cytokines Andrew Lichtman, MD PhD Brigham and Women's Hospital Harvard Medical School 2 Lecture outline Cytokines Subsets of CD4+ T cells: definitions, functions, development New
1 Effector T Cells and Cytokines Andrew Lichtman, MD PhD Brigham and Women's Hospital Harvard Medical School 2 Lecture outline Cytokines Subsets of CD4+ T cells: definitions, functions, development New
Natural Killer Cells: Development, Diversity, and Applications to Human Disease Dr. Michael A. Caligiuri
 Natural Killer Cells: Development, Diversity, November 26, 2008 The Ohio State University Comprehensive Cancer Center The James Cancer Hospital and Solove Research Institute Columbus, Ohio, USA 1 Human
Natural Killer Cells: Development, Diversity, November 26, 2008 The Ohio State University Comprehensive Cancer Center The James Cancer Hospital and Solove Research Institute Columbus, Ohio, USA 1 Human
General Biology. A summary of innate and acquired immunity. 11. The Immune System. Repetition. The Lymphatic System. Course No: BNG2003 Credits: 3.
 A summary of innate and acquired immunity General iology INNATE IMMUNITY Rapid responses to a broad range of microbes Course No: NG00 Credits:.00 External defenses Invading microbes (pathogens). The Immune
A summary of innate and acquired immunity General iology INNATE IMMUNITY Rapid responses to a broad range of microbes Course No: NG00 Credits:.00 External defenses Invading microbes (pathogens). The Immune
