B and T lymphocyte attenuator regulates CD8 + T cell intrinsic homeostasis and memory cell generation
|
|
|
- Emil Fox
- 6 years ago
- Views:
Transcription
1 27 Nature Publishing Group B and T lymphocyte attenuator regulates CD8 + T cell intrinsic homeostasis and memory cell generation Carsten Krieg 1, Onur Boyman 1,2, Yang-Xin Fu 3 & Jonathan Kaye 1 B and T lymphocyte attenuator (BTLA) is a negative regulator of T cell activation, but its function in vivo is not well characterized. Here we show that mice deficient in full-length BTLA or its ligand, herpesvirus entry mediator, had increased number of memory CD8 + T cells. The memory CD8 + T cell phenotype resulted from a T cell intrinsic perturbation of the CD8 + T cell pool. Naive BTLA-deficient CD8 + T cells were more efficient than wild-type cells at generating memory in a competitive antigen-specific system. This effect was independent of the initial expansion of the responding antigen-specific T cell population. In addition, BTLA negatively regulated antigen-independent homeostatic expansion of CD4 + and CD8 + T cells. These results emphasize two central functions of BTLA in limiting T cell activity in vivo. To induce optimal protective immunity, the adaptive immune system maintains a fairly constant number of naive T cells from which longlived memory T cells arise. During an immune response, the number of antigen-specific T cells greatly increases to carry out effector functions before contracting and leaving behind protective memory T cells. After a lymphopenic state, such as that found in newborns 1, during infection 2 or as a result of chemotherapy 3 or irradiation 4,5, naive T cells also expand. This process, called homeostatic proliferation, restores lymphocyte cellularity in the absence of an induced effector response. Although homeostatic proliferation is similar to T cell antigen driven proliferation in its requirement for interactions between the T cell antigen receptor (TCR) and major histocompatibility complex (MHC) proteins 5, the two processes differ in other respects. First, homeostatic proliferation of naive T cells after lymphopenia is driven by large amounts of interleukin 7 (IL-7) and is polyclonal, whereas antigen-driven proliferation results in expansion of T subsets with particular specificities 6,7. Second, antigen-specific T cell responses are dependent on the interaction of costimulatory molecules including CD28-B7, whereas homeostatic proliferation relies on distinct costimulatory pathways such as CD24 (refs. 8 1). Little is known, however, about the influence of coinhibitory molecules on antigen-specific T cell responses and there are no data on negative regulation of homeostatic proliferation. The group of coinhibitory regulators of T cell activation include the attenuators of TCR signaling CTLA-4 and PD-1, in addition to the B and T lymphocyte attenuator BTLA (also known as CD272; ref. 11). The murine Btla gene has been independently identified in screens for genes upregulated in thymocytes undergoing positive selection and T helper type 1 (T H 1) cells 12,13. The BTLA protein is a member of the immunoglobulin superfamily of cell surface molecules and binds herpesvirus entry mediator (HVEM) 14,15, a member of the tumornecrosis factor receptor family 14. Although BTLA was originally considered to be a member of the CD28 family of proteins, its immunoglobulin-like domain and manner of ligand binding is distinct, consistent with its binding to a non-member of the immunoglobulin superfamily 16. Resting murine lymphocytes constitutively express BTLA in varying amounts: B cells show the highest cell surface expression, followed by T cells 12,13 ; BTLA is also expressed by some antigen-presenting cells (APCs) 13,17. A similar expression pattern has been reported for human BTLA 18. The BTLA ligand HVEM has been characterized both in terms of its costimulatory function on binding to the cell surface protein LIGHT, which is expressed on activated T lymphocytes, and as a receptor for lymphotoxin-a (refs ). Subsequent studies suggest, however, that HVEM has a negative regulatory role 22.The expression pattern of BTLA and HVEM is reciprocal on murine T cells and APCs: HVEM is expressed on resting T cells and immature dendritic cells (DCs) and is downregulated after T cell activation 23 ;by contrast, murine BTLA is upregulated by activated T cells and expressed by mature DCs 12,13,17. BTLA, HVEM, LIGHT, lymphotoxin-a and the LIGHT receptor LTbR constitute a complex regulatory network. LIGHT is induced on T cell activation 21, but how its induction might influence BTLA-mediated signals remains to be determined. Unlike LIGHT, which promotes signaling through HVEM by trimeric clustering, BTLA associates with HVEM in a monomeric interaction 16 and thus may not induce signaling to the HVEM-bearing cell. HVEM can simultaneously bind BTLA and LIGHT, although whether such a trimolecular complex of membrane bound proteins forms in vivo is not clear 15,16,24. 1 Department of Immunology, The Scripps Research Institute, La Jolla, California 9237, USA. 2 Division of Immunology and Allergy, University Hospital of Lausanne, CH-111 Lausanne, Switzerland. 3 Department of Pathology, University of Chicago, Chicago, Illinois 6637, USA. Correspondence should be addressed to J.K. (jkaye@scripps.edu). Received 1 August 26; accepted 6 November 26; published online 7 January 27; doi:1.138/ni VOLUME 8 NUMBER 2 FEBRUARY 27 NATURE IMMUNOLOGY
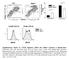
2 27 Nature Publishing Group a b 24 CD8 c + c.p.m. ( 1 3 ) 35 3 Total Anti-CD3 (µg/ml) Accumulating data indicate that BTLA is a negative regulator of lymphocytes. Engagement of BTLA by agonist antibody or a soluble form of HVEM in vitro profoundly inhibits TCR-mediated T cell proliferation and cytokine production 15,18,25. The cytoplasmic tail of BTLA contains tyrosine-based motifs that are sites of phosphorylation capable of recruiting the tyrosine phosphatases SHP-1 and SHP-2 (refs. 12,13), although some data question whether SHP recruitment mediates the negative regulatory activity of BTLA 26. Despite the relatively broad expression of this negative regulator, mice deficient in BTLA show no gross perturbations in lymphocyte development, cell activation or the composition of lymphoid organs 12,13. T cells from BTLA-deficient mice are, however, hyperresponsive to TCR-induced proliferation in vitro 12,13. In addition, BTLA-deficient mice have greater severity and duration of experimental autoimmune encephalomyelitis 12, prolonged airway inflammation 27 and increased rejection of minor mismatched allografts 28, further supporting a negative regulatory role for BTLA in vivo. Here we report that the hyperproliferative response of BTLAdeficient T cells in vitro is not an intrinsic property of the T cell but is instead due to increased numbers of memory-phenotype CD8 + T cells. We found a similar phenotype in mice lacking HVEM. Bone marrow chimeras that allow development of a mixture of wild-type and BTLA-deficient T cells revealed that the increased generation of memory-phenotype CD8 + T cells was a T cell intrinsic property. Moreover, BTLA-deficient CD8 + T cells were not different from wild-type T cells in response to antigen-specific DC priming but were more efficient generators of memory. When compared with cotransferred wild-type T cells in the same host, BTLA-deficient CD4 + and CD8 + T cells also showed enhanced homeostatic proliferation. Together, these data show that BTLA-mediated negative regulation of memory CD8 + T cell formation and T cell homeostasis has a role in vivo. RESULTS BTLA-deficient CD8 + T cells are hyper-responsive in vitro Purified T cells from BTLA-deficient mouse strains show enhanced proliferation in response to TCR stimulation in vitro as compared with wild-type T cells 12,13. These data have been interpreted to indicate that BTLA is a negative regulator of T cell activation; however, BTLA is not thought to act autonomously on a T cell, but rather to mediate its negative regulatory function by engagement of HVEM. Because HVEM is also expressed on resting T cells 21,29, T cell T cell interactions, at least under culture conditions, might inhibit T cell responses and this inhibition might be relieved by loss of BTLA. Alternatively, the composition of T cell populations in normal and BTLA-deficient c.p.m. ( 1 3 ) Anti-CD3 (µg/ml) Figure 1 Naive and activated-memory BTLA-deficient T cells are not intrinsically hyperproliferative. Purified wild-type () and BTLA-deficient () T cell subsets were cultured for 72 h on plates coated with anti-cd3 at indicated concentrations. Data are expressed as the mean ± s.d. incorporation of [ 3 H]thymidine in triplicate cultures. Results are shown for total T cells (a), CD4 + and CD8 + T cells (b), and lo (naive) and hi (activated memory) subsets of CD4 + and CD8b + Tcells(c). Data are representative of three independent experiments. c.p.m. ( 1 3 ) CD4 + lo 12 CD4 + hi CD8β + lo CD8 + hi Anti-CD3 (µg/ml) mice might differ. To distinguish between these two possibilities, we compared the proliferative response of isolated subpopulations of CD4 + and CD8 + T cells. We targeted exon 2 of the Btla gene to create BTLA-deficient mice 13. These mice do not express BTLA that can bind HVEM but do express mrna encoding a major splice variant of the protein that lacks most of the external domain 13. As previously reported 12,13,total T cells from BTLA-deficient mice proliferated more vigorously than wild-type cells in response to plate-bound monoclonal antibody (mab) to CD3 (anti-cd3; Fig. 1a). This hyperproliferation was also observed for isolated CD8 + T cells (Fig. 1b). Purified CD4 + T cells, by contrast, did not show enhanced proliferation in the absence of BTLA, which was unexpected because BTLA expression was greater on CD4 + T cells than on CD8 + T cells 13. Ex vivo, T cells contain both naive ( lo ) and activated memory-phenotype cells ( hi ), of which the latter subpopulation is particularly sensitive to TCR stimulation in the absence of costimulation 3. As expected, hi CD4 + and hi CD8 + T cells were more responsive to stimulation with anti-cd3 than were an equal number of their lo counterparts (Fig. 1c). The absence of BTLA had no effect on the proliferative response of isolated naive lo CD4 + T cells or hi CD4 + or hi CD8 + T cells, whereas the proliferation of naive CD8 + T cells from BTLAdeficient mice was unexpectedly modestly reduced (Fig. 1c). Collectively, these results show that purified BTLA-deficient CD8 + T cells are hyperproliferative in vitro, accounting for the enhanced response of total T cells from these mutant mice. Loss of BTLA, however, does not enhance proliferation of isolated lo CD8 + or hi CD8 + T cells, suggesting that the CD8 + T cell subpopulation of BTLA-deficient mice is perturbed. Increase in memory CD8 + T cells in BTLA-deficient mice As a result of lifetime exposure to various environmental antigens and possibly self-antigens, normal (wild-type) C57BL/6 mice accumulate hi memory-phenotype CD4 + and CD8 + T cells with increasing age 31. Spleen and lymph nodes of BTLA-deficient mice had similar percentages of hi CD4 + T cells as compared with age-matched wild-type mice (Fig. 2a). By contrast, the percentage of hi CD8 + T cells in spleen and lymph node was approximately twofold greater in BTLA-deficient than in wild-type mice (Fig. 2a). The absolute number of hi CD8 + T cells in BTLA-deficient spleen and blood was also roughly twofold higher (Fig. 2b,c). Because hi T cells account for a disproportionate amount of the proliferative response of total T cells (Fig. 1c), an increase in this subpopulation explains the hyperproliferative phenotype observed in total BTLA-deficient CD8 + NATURE IMMUNOLOGY VOLUME 8 NUMBER 2 FEBRUARY
3 27 Nature Publishing Group a CD3 + CD8 + CD3 + CD4 + Spleen LN Spleen LN CD62L d CD4 + CD8 + CD4 + CD8 + e CD4 + CD8 + f HVEM CD HVEM b CD43 Cell number ( 1 6 ) g CD8 Spleen CD3 + CD8 + hi HVEM- Spleen c Percent lo hi lo BrdU Blood CD8β + hi *** * *** ** HVEM- LN hi Figure 2 Increased CD8 + memory-phenotype T cells in and HVEM-.2 mice. (a g) Analyses of spleen and pooled lymph node (LN) cells from 3-monthold wild-type, and HVEM- mice by flow cytometry. (a) Expression of hi within the CD3 + CD4 + and CD3 + CD8 + T cell populations. Numbers indicate percentage of cells in the gated areas of histograms and dot plots. (b) Numbersof CD3 + CD8 + hi cells in spleen of individual age-matched wild-type,.2 and HVEM- mice. (c) Percentage of CD8b + hi cells in blood of mice as in 47 lo 53 b. Horizontal bars indicate the mean. P values refer to comparisons of wild-type, and HVEM- mice. (d) Expression of CD62L and or CD122 and, as in a. (e) Expression of CD43 and within the CD3 + CD4 + and CD45.2 CD9.1 BrdU CD3 + CD8 + T cell populations as in a. (f) In vivo proliferation of lo and hi CD8 + T cells in wild-type and mice. BrdU was given in drinking water for 4 d before isolation of lymph node and spleen cells, and determination of BrdU incorporation by flow cytometry. Numbers represent the percentage of BrdU + CD8 + T cells. Data are representative of four mice per strain. (g) Competitive homeostatic proliferation of lo or hi CD8 + T cells from wild-type and mice. A 1:1 ratio of allelically marked wild-type (CD9.2 + CD ) to (CD9.1 + CD ) lo CD8 + or hi CD8 + Tcellswas transferred into intact hosts (CD ). Mice were exposed to BrdU in drinking water from days 3 7 after cell transfer. BrdU incorporation was determined 1 week after transfer. Dot plots and histograms are representative of four individual recipients. T cells. Because BTLA expression increases on T cell activation, at least in mouse 13,25, we also tested whether memory-phenotype T cells express high amounts of BTLA as compared with naive cells. However, hi CD4 + and hi CD8 + T cells showed only marginally increased BTLA expression as compared with naive T cell populations (Supplementary Fig. 1 online). hi T cells represent a heterogeneous population of recently activated and resting memory-phenotype T cells 32,33.Toassessthe distribution of these subsets, we stained splenocytes from wild-type and BTLA-deficient mice for CD62L, CD122 and the effector T cell marker CD43. The percentage of CD4 + T cell subpopulations was normal in BTLA-deficient mice as compared with wild-type mice. However, BTLA-deficient hi CD8 + T cells showed a twofold increase in the percentage of CD62L hi,cd122 hi and CD43 lo T cells (Fig. 2d,e). Most BTLA-deficient hi CD8 + T cells were also CD127 hi (that is, IL-7Ra hi ; data not shown). Central memory CD8 + T cells have been reported to be hi,cd62l hi,cd122 hi,cd43 lo and IL-7Ra hi (ref. 34). Thus, most splenic hi CD8 + T cells from BTLA-deficient mice resembled central memory T cells rather than activated or effector T cells. We considered that, if the increase in memory-phenotype CD8 + T cells in BTLA-deficient mice is caused by a failure to deliver a negative regulatory signal through BTLA, then mice that are deficient in HVEM, the BTLA ligand, might have a similar phenotype. Indeed, HVEM-deficient mice showed increased percentages and numbers of hi CD8 + T cells, similar to BTLA-deficient mice (Fig. 2b e). Most hi CD8 + splenic T cells in HVEM-deficient mice were CD62L hi, CD122 hi and CD43 lo, matching the phenotype of hi CD8 + T cells from BTLA-deficient mice. The phenotype of BTLA-deficient mice was more severe than that of HVEM-deficient mice, and there were greater numbers of hi CD8 + T cells in the former strain. Comparison of hi CD8 + T cell percentages in BTLA-deficient and HVEM-deficient blood showed a highly significant difference (P o.1; Fig. 2c). This 164 VOLUME 8 NUMBER 2 FEBRUARY 27 NATURE IMMUNOLOGY
4 27 Nature Publishing Group a b c / ratio 3. P =.3 P = P <.1 Total CD4 + CD8 + Total CD4 + CD8 + Thymus Spleen CD CD45.2 trend was also evident among splenic T cells, although greater variability between individual mice decreased the statistical significance for this sample size (P ¼.9; Fig. 2b). We also considered whether the increase in hi CD8 + T cells in BTLA-deficient mice was caused by enhanced proliferation in this cell subset. However, the rate of homeostatic proliferation, as measured by incorporation of 5-bromodeoxyuridine (BrdU), was comparable between BTLAdeficient and wild-type CD8 + T cells, both for naive and memoryphenotype cells (Fig. 2f). A complicating feature of this homeostatic proliferation experiment is the potential conversion of lo cells to hi cells. To exclude this possibility, we transferred a 1:1 ratio of wild-type and BTLA-deficient highly purified CD8 + lo or CD8 + hi T cells into allelically marked hosts, which were then given BrdU in their drinking water. There was no difference between the BrdU profiles of transferred wild-type and BTLA-deficient CD8 + T cells (Fig. 2g), indicating further that there is no enhanced turnover of naive or memory BTLA-deficient CD8 + T cells. These results are consistent with the idea that a loss of BTLA HVEM interactions produces increased numbers of memoryphenotype CD8 + T cells. The fact that this phenotype is less pronounced in HVEM-deficient mice might result from the additional role of HVEM as a receptor for costimulatory signals mediated by LIGHT 2,23. CD CD8 CD4 + CD Cell number ( 1 6 ) Cell number ( 1 6 ) owing to greater numbers of CD4 + T cells in the mutant mice (Supplementary Fig. 2 online). Further analysis of expression on CD4 + and CD8 + T cells in chimeric mice showed an approximately twofold increase in both lo and hi BTLA-deficient CD4 + T cells as compared with their wild-type counterparts (Fig. 3b,c). Thus, the BTLA-deficient CD4 + T cells maintained a percentage of hi cells comparable to that of wild-type cells (Fig. 3b). A similar percentage was not, however, observed for the CD8 + T cell subpopulation. Whereas the mean BTLA-deficient to wild-type ratio of lo CD8 + T cells was 1.6, the corresponding ratio of hi CD8 + T cells was 3.3 (Fig. 3c), resulting in an approximately twofold increase in the percentage of BTLA-deficient hi CD8 + T cells (Fig. 3b) and a phenotype recapitulating that of intact BTLA-deficient mice (Fig. 2a e). Because the BTLA-deficient and wild-type cells developed in the same mouse, the increase in memory-phenotype BTLA-deficient T cells must be a T cell intrinsic property. When naive ( lo ) T cells develop to memory ( hi )Tcells, effector gene regulation is reprogrammed 35.Asaresult, hi cells produce interferon-g (IFN-g) more rapidly than naive cells 36.Totest whether CD8 + hi BTLA-deficient T cells shared this property, we stimulated splenocytes from mixed bone marrow chimeras for 5 h CD4 + hi 2.2 CD4 + lo 2.1 CD8 + hi 3.3 CD8 + lo Figure 3 T cell intrinsic increase in CD8 + memory-phenotype T cells in mice. (a) Ratio of to wild-type total T cells, CD4 + T cells and CD8 + T cells in thymus and spleen of individual mice 3 months after bone marrow reconstitution; horizontal bars indicate the mean. (b) Donor wild-type (CD9.2 + CD45.2 ) and donor (CD9.2 + CD ) splenic T cells were analyzed for expression of CD4, CD8 and. (c) Summary of multiple mice analyzed as in b. Shown are the numbers of hi and lo CD4 + and CD8 + splenic donor T cells for wild-type and cells. The fold increase comparing the mean cell numbers in each group is also indicated. 1.6 Loss of BTLA exerts a T cell intrinsic effect on CD8 + Tcells To determine whether the increase in memory-phenotype CD8 + T cells in BTLA-deficient mice was an intrinsic property of the T cell, we generated mixed bone marrow chimeras. Lethally irradiated wild-type hosts were reconstituted with a 1:1 ratio of allelically marked wild-type and BTLA-deficient bone marrow cells. Three months after reconstitution, the ratios of donor wild-type to donor BTLA-deficient T cells in thymus and spleen were analyzed. The mean BTLA-deficient to wild-type ratio for these chimeric mice was.9 for total, 1.2 for CD4 + and 1. for CD8 + thymocytes, respectively (Fig. 3a), indicating that BTLA-deficient bone marrow cells had no overall developmental advantage or disadvantage. By contrast, the mean BTLA-deficient to wild-type ratio in the spleen increased to 2.2 for total, 2.1 for CD4 +, and 2. for CD8 + T cells (Fig. 3a), suggesting that a post-thymic effect favored the mutant cells. An increase in the CD4 + to CD8 + ratio among BTLA-deficient cells was also observed (Fig. 3b). In addition, a similar increase was observed in unmanipulated BTLA-deficient and HVEM-deficient mice, primarily CD CD IFN-γ Figure 4 In mixed bone marrow chimeras, rapid IFN-g production correlates with the percentage of the hi cell population. Splenocytes from bone marrow chimeras 1 months after reconstitution were analyzed for expression of in wild-type (CD45.2 ) and (CD ) CD8 + cells. Cells were also stimulated with (black line) or without (shaded area) PMA and ionomycin for 5 h to analyze IFN-g production. Numbers indicate the percentage of cells in the gates. A second independent experiment yielded similar results. NATURE IMMUNOLOGY VOLUME 8 NUMBER 2 FEBRUARY
5 27 Nature Publishing Group a LN Spleen CD CD9.1 b / ratio LN SP with phorbol 12-myristate 13-acetate (PMA) and ionomycin (Fig. 4). In this short-term stimulatory assay, there was good concordance between the percentage of hi T cells and that of IFN-g producers in both wild-type and BTLA-deficient T cells. Thus, BTLAdeficient hi CD8 + T cells also acquire functional characteristics of memory cells. Enhanced homeostatic expansion of BTLA-deficient T cells The competitive advantage of both CD4 + and CD8 + BTLA-deficient T cells in spleen but not thymus in the above experiments suggested that loss of BTLA might perturb T cell homeostasis. To address this, we purified naive lo T cells from allelically marked wild-type and BTLA-deficient mice, and adoptively transferred a 1:1 ratio into sublethally irradiated wild-type hosts. Under these lymphopenic conditions, naive T cells undergo homeostatic proliferation owing to an increase in cytokines, primarily IL-7 (ref. 37). One month after adoptive transfer, both CD4 + and CD8 + BTLA-deficient T cells were overrepresented in spleen and lymph nodes as compared with wildtype T cells (Fig. 5a,b). In lymph nodes, the BTLA-deficient to wildtype mean ratios were 3.6 and 3.2 for CD4 + and CD8 + T cells, respectively, whereas the corresponding mean ratios in the spleen were 25.5 and 2.9. The marked accumulation of homeostatically expanded c CD8β Percentage of precursors CD9.1 CFSE Divisions Figure 5 T cells outcompete wild-type T cells during homeostatic expansion. (a) The percentage of donor wild-type (CD CD9.1 + )and donor (CD CD9.1 ) T cells was determined in spleen and pooled lymph nodes (LN) 1 month after a 1:1 mixture of allelically marked naive (CD3 + lo ) wild-type and T cells was injected into sublethally irradiated (7.5 Gy) wild-type hosts. Numbers in dot plots indicate the percentage of gated cells. (b) Summary of the ratio of to wildtype donor T cells in lymph nodes and spleen from four mice analyzed as in a. (c) A 1:1-ratio of purified CFSE-labeled wild-type and CD8 + OT-I (V a 2 + ) T cells was transferred into sublethally irradiated wild-type hosts. Left, splenocytes were analyzed for the percentage and CFSE profiles of wild-type (CD9.1 + ) and BTLA-deficient (CD9.1 )CD8b + V a 2 + T cells 1 week after adoptive transfer. Right, on the basis of the CFSE profile, the distribution of precursor cell divisions was calculated. Data are representative of two independent experiments. BTLA-deficient T cells in spleen versus lymph nodes is presumably due to their activation state. Homeostatic proliferation in lymphopenic mice involves TCRmediated recognition of self-peptide MHC complexes in addition to a response to increased cytokine concentration. We therefore tested the possibility that the increased expansion of BTLA-deficient T cells resulted from a change in T cell repertoire in these mice (for example, thymic selection of cells with higher affinity for self-peptide MHC complexes). We analyzed the homeostatic expansion of T cells of a single TCR specificity, in particular the OT-I TCR. A 1:1 ratio of carboxyfluorescein diacetate succinimidyl diester (CFSE)-labeled CD8 + OT-I TCR transgenic (called CD8 + OT-I cells here) wild-type to BTLA-deficient T cells were transferred into sublethally irradiated hosts. One week after transfer, the percentage of BTLA-deficient CD8 + OT-I cells had increased twofold in comparison to wild-type CD8 + OT-I cells (Fig. 5c). Analysis of the CFSE profiles showed that essentially all BTLA-deficient and wild-type cells underwent homeostatic expansion, but BTLA-deficient OT-I cells underwent more divisions on average, thereby accounting for their greater cell numbers (Fig. 5c). Expression of BTLA on wild-type CD8 + OT-I cells was not increased during homeostatic expansion (Supplementary Fig. 3 online). Collectively, these data show that BTLA dampens homeostatic Figure 6 More efficient antigen-specific CD8 + memory formation by T cells in the absence of an enhanced primary response. (a) Flow cytometry on spleen cells from wild-type hosts that received a 1:1-ratio of purified OT-I CD8 + wild-type and T cells and were immunized 1 day later with SIINFEKL peptide pulsed mature bone marrow derived DCs. Shown is quantification of wild-type (open boxes) and (closed boxes) donor T cells in wildtype host spleens on days, 3, 4, 7, 14 and 3 after DC immunization. Each data point represents the mean ± average deviation of two mice. Two additional independent experiments gave similar results. (b) Experimental protocol of in vivo killing assay. CD8 + OT-I wild-type (CD9.1 + ) or (CD9.2 + ) T cells were transferred into allelically marked hosts and immunized with peptide-pulsed DC. After 3.5 d, mice were injected with an equal ratio of CFSE lo unpulsed or CFSE hi SIINFEKL peptide pulsed splenocytes. Spleens were collected 4 h or 16 h after transfer a OT-I T cells per spleen ( 1 6 ) c Transfer only Peptide response Time (d) and analyzed for the preferential loss of peptide-pulsed CFSE hi splenic target cells. (c) Representative CFSE profiles from two identically treated mice. Numbers represent the percentage of CFSE lo or CFSE hi splenocytes as indicated. Percentage lysis was calculated. Unpulsed CFSE hi splenocytes were also used as controls (transfer only). (d) Efficiency of memory T cell formation in wild-type and CD8 + donor T cells on day 14 after immunization. The mean total cell number of each genotype on day 4 after immunization was normalized to 1%. The 95%CI for differences in means was 2.1-fold to 4.7-fold. 4 h 3 b CD8 + OT-I T cells [CD9.2] CD8 + OT-I T cells [CD9.1] Host [CD9.1] Peptide-pulsed 4 h or 16 h DC immunization d 3.5 Host [CD9.2] % lysis 49% lysis 97% lysis 98% lysis CFSE h 96 2 CFSE lo no peptide splenic targets d Percent of day 4 cell numbers CFSE hi peptide-pulsed splenic targets P =.4 n = VOLUME 8 NUMBER 2 FEBRUARY 27 NATURE IMMUNOLOGY
6 27 Nature Publishing Group a CD43 CD122 CD127 CD62L b Percentage of day 4 response Host CD Day 4 Donor CD P =.6 P =.1 P =.3 P =.1 P =.4 hi Day 14 T cell proliferation regardless of repertoire and that, on the basis of the competitive chimera approach, this is also a T cell intrinsic process. BTLA-deficient CD8 + cells generate memory more efficiently The increase in BTLA-deficient memory-phenotype CD8 + T cells in intact mutant mice and in bone marrow chimeras could result from increased proliferation or survival of pre-formed memory T cells. Because no alteration in BrdU incorporation was detected in these cells (Fig. 2f,g), we tested whether BTLA is instead a negative regulator of de novo formation of CD8 + memory T cells (Figs. 6,7). The absence of inflammation during immunization with DCs leads to relatively rapid generation of CD8 + T cell memory 38.Weusedthis system to assess the influence of BTLA during a defined antigenspecific response. Once again we used a competitive adoptive transfer approach to eliminate individual mouse variability and host environmental effects. A 1:1 ratio of allelically marked CD8 + OT-I wild-type to CD8 + OT-I BTLA-deficient T cells were transferred into wild-type hosts. One day after transfer, mice were immunized at day with peptide-pulsed bone marrow derived mature DCs (called DC-peptide here). The percentage of wild-type and BTLA-deficient CD8 + OT-I T cells was then analyzed at days (before DC-peptide immunization), 3, 4, 14 and 3. Unexpectedly, the BTLA-deficient T cells showed no competitive advantage during the initial expansion phase of the response. For the CD8 + OT-I BTLA-deficient T cells, peak expansion on day 4 after DCpeptide immunization was comparable to or even slightly lower than that of the wild-type CD8 + OT-I T cells (Fig. 6a). The BTLA-deficient and wild-type CD8 + OT-I T cells also underwent contraction with CD43 lo CD122 + CD127 hi CD62L hi CD8 + T cell memory marker Figure 7 Enhanced acquisition of a typical memory-phenotype by CD8 + Tcells.(a) Flow cytometry on CD8 + T cells 4 d or 2 weeks after DC-peptide immunization for expression of, CD43, CD122, CD127 and CD62L. Histograms show the percentage of CD8 + T cells of host (CD ), donor wild-type (CD45.2 +, CD9.1 + ) and donor (CD45.2 +, CD9.1 ) cells expressing the indicated marker. (b) Normalized percentage of donor wild-type and donor CD8 + T cells expressing the indicated memory T cell marker on day 14 after immunization. The total wild-type and T cell number per spleen on day 4 after immunization is normalized to 1%. Data are from six mice distributed across three experiments. The 95%CI for differences between mean frequency of surviving CD43 lo OT-I T cells was 3-fold to 7.5-fold. similar kinetics (Fig. 6a). To determine whether BTLA-deficient CD8 + OT-I T cells generate fully functional effector T cells, we analyzed function (Fig. 6b,c) and phenotype (Fig. 7a) of responding T cells on days 3.5 and 4 of the response, respectively. The BTLA-deficient and wild-type CD8 + OT-I T cells showed similar effector cell phenotypes, including upregulation of and CD43 and downregulation of CD127 (Fig. 7a). The in vivo cytolytic activity of wild-type or BTLAdeficient CD8 + OT-I cells during the effector phase was also determined by using individual allelically marked hosts and a 1:1 mixture of differentially CFSE-labeled syngeneic targets that were (CFSE hi )or were not (CFSE lo ) pulsed with ovalbumin (OVA) peptide (Fig. 6b). Both BTLA-deficient and wild-type responding T cells specifically eliminated the CFSE hi peptide-pulsed targets by 16 h (98% and 97% specific lysis, respectively), and the mutant T cells showed greater activity than the wild-type cells at 4 h (49% and 29% specific lysis, respectively; Fig. 6c). Thus, loss of BTLA did not prevent T cell effector function. Despite a similar effector phase, however, BTLA-deficient CD8 + OT-I T cells outnumbered wild-type CD8 + OT-I T cells on days 7 and 14 of the response, and this difference remained up to 3 d after DC-peptide immunization (Fig. 6a). To quantify this effect, we analyzed the efficiency of T cell survival after contraction, defined as CD8 + OT-I T cells present on day 14 as a percentage of the peak response on day 4 (Fig. 6d). By this measure, BTLA-deficient donor cells were found to be 3.4-fold more efficient at generating memoryphenotype cells by day 14, despite modestly reduced expansion of these same cells during the peak of the response. Enhanced efficiency of BTLA-deficient CD8 + OT-I memory T cell generation after DC-peptide immunization was also associated with enhanced transition to a phenotype typical of CD8 + memory. As expected, both BTLA-deficient and wild-type CD8 + OT-I T cells on day 14 after DC-peptide immunization were hi, and most were CD122 hi (Fig. 7a). However, roughly twice as many of the BTLA-deficient CD8 + OT-I T cells had downregulated CD43 as compared with the wild-type CD8 + OT-I cells (Fig. 7a). The percentages of CD127 hi and CD62L hi cells were also increased in BTLA-deficient CD8 + OT-I T cells as compared with wildtype CD8 + OT-I T cells from the same donor mouse (Fig. 7a). Together, the data indicate that a greater proportion of donor BTLAdeficient CD8 + OT-I T cells that survived day 14 had a memory phenotype ( hi, CD43 lo, CD122 hi, CD127 hi, CD62L hi ) as compared with donor wild-type CD8 + OT-I T cells in these chimeric mice. Studies using these markers individually to assess the efficiency of memory CD8 + OT-I T cell generation in vivo showed significant differences between wild-type and BTLA-deficient T cells, and greater memory cell production in the latter population (4.7-fold increase by using reduction of CD43 as a marker; Fig. 7b). Because these were mixed adoptive transfers of OT-I T cells into wild-type hosts, the effect of BTLA loss on CD8 + T cell memory formation could be explained only by a T cell intrinsic process. NATURE IMMUNOLOGY VOLUME 8 NUMBER 2 FEBRUARY
7 27 Nature Publishing Group a CD8 c 2 DC-peptide LM-OVA Tetramer CD CD9.2 b CD8 + OT-I T cells per spleen To determine whether CD8 + OT-I T cells that survived 3 d maintained responsiveness to antigen, cells obtained from immunized mice were activated by specific peptide in vitro and stained with SIINFEKL-K b tetramer. Both BTLA-deficient and wild-type CD8 + OT-I T cells expanded under these conditions, but BTLA-deficient T cells were present at a 3.5-fold higher percentage than wild-type, mirroring their overrepresentation at day 14 (Fig. 8a). A characteristic of memory CD8 + T cells is their ability to undergo substantial secondary expansion and to provide protective immunity against subsequent infection 34,39. To determine whether BTLA-deficient OT-I memory T cells were functional in this regard, mice immunized with DC-peptide were rechallenged with DC-peptide or a lethal dose (5 1 5 c.f.u. per mouse) of Listeria monocytogenes engineered to express OVA (LM-OVA) 4. On day 3 after primary immunization, BTLA-deficient memory T cells outnumbered their wild-type counterparts by twofold (Fig. 8b). After DC-peptide rechallenge, the wild-type and BTLAdeficient memory T cells expanded tenfold and sevenfold, respectively (Fig. 8b). The modest reduction in responsiveness of the BTLAdeficient memory T cells, as compared with wild-type memory T cells, was similar to that seen for the primary response of naive BTLA-deficient CD8 + OT-I T cells (Fig. 6a). By contrast, after secondary challenge with LM-OVA, wild-type and BTLAdeficient memory T cells expanded 91-fold and 191-fold, respectively, increasing the BTLA-deficient to wild-type ratio to 4.3 (Fig. 8b). Thus, both BTLA-deficient and wild-type memory CD8 + T cells proliferate in vivo during a secondary response, but the former are at a specific advantage under conditions of infection as compared with immunization. To determine whether expansion of memory T cells is associated with acquisition of effector function, responding cells were activated for 5 h in culture with LM-OVA peptide and stained for intracellular IFN-g. As expected on the basis of the extent of in vivo expansion (Fig. 8b), a higher percentage of CD8 + T cells from mice infected with LM-OVA produced IFN-g in response to peptide as compared with mice challenged with DC-peptide (Fig. 8c). Nevertheless, IFN-g responders included both BTLA-deficient and wildtype CD8 + T cells, and the former were in greater abundance (Fig. 8c). Thus, BTLA-deficient memory CD8 + T cells are responsive to secondary challenge (/) (1.5) ** (2.1) * 1 4 Day 3 2 Day 3+4 DC-peptide (4.3) *** Day 3+5 LM-OVA Unstimulated P + I Peptide IFN-γ IFN-γ CD9.1 Figure 8 Secondary responses of CD8 + memory T cells. (a) Flow cytometry on splenocytes obtained from mice 1 month after DC immunization re-stimulated in vitro with SIINFEKL-pulsed irradiated splenocytes for 7 d. Shown is the percentage of wild-type and within SIINFEKL tetramer positive CD8 + Tcells.(b) Total number of wild-type and CD8 + OT-I T cells per spleen 3 d after DC immunization, an additional 4 d after DC booster immunization or an additional 5 d after LM-OVA infection. Data are the mean ± s.d. from four mice per group. Numbers in parentheses indicate the ratio of to wild-type T cells in each group. (c) Distribution of (CD9.1 ) and wild-type (CD9.1 + ) T cells among CD8 + IFN-g + T cells after DC booster immunization (day 4) or LM-OVA challenge (day 5) after re-stimulation for 5 h with peptide. IFN-g staining of cells cultured for 5 h in medium alone (unstimulated) or PMA and ionomycin (P + I) is shown for comparison. 21, secondary immunization or infection. Data are representative of four mice per group. DISCUSSION These data identify a previously unknown role for BTLA in regulating T cell homeostasis and CD8 + T cell memory formation. In both contexts, loss of BTLA leads to an increase in response, providing further evidence that BTLA functions as a negative regulator in vivo. Previously, the hyperproliferative response of BTLA-deficient T cells to immobilized anti-cd3 was thought to reflect loss of a coinhibitory signal on the T cell 12,13. We have shown, however, that this observation must be reinterpreted because purified lo and hi CD4 + and CD8 + subsets of BTLA-deficient T cells are not hyperproliferative under these same culture conditions. Because the only source of BTLA ligand (HVEM) in assays involving highly purified T cells is the T cells themselves, the fact that loss of BTLA does not alter T cell proliferation in culture suggests that regulation of T cell activation through BTLA is likely to involve binding to HVEM on non-t cells, presumably APCs. Rather than loss of a coinhibitory signal, the hyperproliferative phenotype of BTLA-deficient T cells is caused by an increase in the percentage of memory-phenotype ( hi, CD122 hi, CD43 lo and CD62L hi )CD8 + T cells. In particular, the CD62L hi phenotype of this population is consistent with that of central memory T cells 34.This T cell subset is less dependent on costimulation and is highly proliferative after stimulation with immobilized anti-cd3 in vitro, which probably accounts for the hyperproliferative response of BTLAdeficient CD8 + T cells. This observation brings caution to interpretations of this often used measure of TCR-mediated activation of T cells, particularly in the context of induced mutations that could affect the distribution of T cell subsets. Using mixed bone marrow chimeric mice containing wild-type and BTLA-deficient T cells, we have further shown that the loss of BTLA on the T cell itself is responsible for increasing the abundance of memory-phenotype CD8 + cells. Thus, within the same mouse, wild-type and BTLA-deficient T cells show independent regulation of this CD8 + T cell subset. This finding argues against a role for BTLA on APCs, or against changes in cytokine production by these mutant T cells operating in a paracrine manner, as the underlying cause of the increase in memory-phenotype CD8 + T cells. Of note, a lack of BTLA has no effect on the steady-state abundance of hi CD4 + T cells, despite the sensitivity of CD4 + T cells to BTLA-mediated inhibition 25. We found that HVEM-deficient mice also have increased percentages of memory-phenotype CD8 + T cells, consistent with the idea that engagement of BTLA by HVEM causes this perturbation in the T cell population. The increase in memory-phenotype CD8 + T cells is, however, more pronounced in BTLA-deficient than in HVEMdeficient mice. As a shared receptor for coinhibitory molecule BTLA and costimulatory molecule LIGHT, HVEM could have competing negative and positive influences, respectively, on T cells. Thus, the 168 VOLUME 8 NUMBER 2 FEBRUARY 27 NATURE IMMUNOLOGY
8 27 Nature Publishing Group promotion of memory-phenotype CD8 + T cells in HVEM-deficient mice by loss of BTLA-mediated signaling may be partially counterbalanced by loss of LIGHT-mediated costimulation. In such a scheme, the BTLA LIGHT HVEM interaction could be a single binding event 15 or, alternatively, separable regulatory events. LIGHT is expressed more highly by activated CD8 + T cells than by CD4 + T cells 23. It has been shown that LIGHT deficiency impairs the in vivo proliferative response of V b 8 + CD8 + but not V b 8 + CD4 + T cells to staphylococcal enterotoxin B, suggesting that LIGHT has a costimulatory function 41. By contrast, primary and memory CD8 + T cell responses to influenza virus are not impaired in LIGHT-deficient mice 42. Thus, the particular balance between LIGHT-mediated costimulation and BTLA-mediated negative regulation may vary depending on quantitative and/or qualitative aspects of the immune response. We also examined how loss of BTLA would perturb the generation of antigen-specific memory by using the well-characterized OT-I system and the ability of DC immunization to induce memory T cells rapidly. In a competitive adoptive transfer system, BTLA-deficient T cells were more efficient than wild-type cells in generating antigen-specific memory CD8 + T cells, mirroring the phenotypes obtained in unimmunized mice. Moreover, antigen-specific BTLA-deficient memory T cells were responsive to secondary challenge in terms of both proliferation and production of the effector cytokine IFN-g. Loss of negative regulation by BTLA did not enhance the initial expansion of antigen-specific CD8 + OT-I T cells in response to DCpeptide immunization, nor did it alter the kinetics of the contraction phase. In some circumstances, the burst size of the initial priming event can determine the extent of memory T cell formation 43. However, independent of the initial burst size, BTLA seems to regulate a relatively late event in memory generation. Whether BTLA might also play a part in T cell priming under conditions that elicit inflammation, however, remains to be tested. This is particularly interesting because loss of BTLA resulted in a modest inhibitory effect on the secondary response to DC-peptide but a significant enhancement of the secondary response to LM-OVA. Thus, inflammatory signals might increase the role of BTLA in braking T cell expansion. How loss of BTLA engagement might enhance CD8 + memory T cell formation is not clear. Because we observed normal BrdU incorporation into hi and lo BTLA-deficient CD8 + T cells during homeostatic proliferation and because, once established, the BTLAdeficient to wild-type ratio of antigen-specific T cells was maintained over time, it seems that BTLA influences de novo formation but not homeostatic maintenance of the memory CD8 + T cell compartment. Thus, BTLA could have a role in limiting cell survival during the initial contraction phase of the response and/or negatively regulate differentiation of the T cells to a memory cell fate. The contraction phase of a response is tightly regulated and only a few primary effector T cells survive to form the memory T cell population 43,44. It has been suggested that, as infection wanes, cytokines that support clonal expansion and T cell survival also decline, triggering apoptosis of activated T cells owing to growth factor withdrawal. Indeed, greater amounts of IL-2, IL-4, IL-7 and IL-15 in vivo can increase the number of antigen-specific CD8 + T cells, and IL-15 enhances protective immunity Administration of IL-2 during the contraction phase also delays the death of superantigen-specific CD8 + effector T cells and enhances the generation of antigen-specific memory after DC immunization IL-2 signaling during the primary response is necessary for the formation of fully responsive CD8 + memory T cells 52.This effect, however, operates in a paracrine manner because in mixed chimeras wild-type cells were found to correct defective secondary responses of T cells with an IL-2 secretion defect. Engagement of BTLA in vitro induces a profound block in T cell proliferation and IL-2 secretion 25. Thus, engagement of BTLA during a response could affect subsequent memory T cell formation by limiting IL-2 secretion. Such a mechanism, however, would necessitate autocrine regulation by IL-2, because our results with mixed chimeric mice show a T cell intrinsic effect of loss of BTLA. Alternatively, BTLA could modify responses to IL-2 or other cytokines. Engagement of BTLA in vitro does inhibit expression of CD25, although not in the presence of costimulation 25. We found two other perturbations of the T cell compartment in BTLA-deficient mice. First, these mice have an increased CD4 to CD8 ratio caused by a T cell intrinsic effect. Second, in competitive assays, expression of BTLA dampens both reconstitution of the periphery in irradiated bone marrow chimeras and lymphopeniainduced T cell homeostatic expansion potentially two manifestations of a similar phenomenon. Unlike effects on T cell memory, loss of BTLA enhances peripheral reconstitution and homeostatic expansion of both CD4 + and CD8 + T cells. For CD4 + T cells, the specific accumulation of hi cells is not affected by loss of BTLA, resulting in maintenance of a normal equilibrium between hi and lo T cells. Homeostatic expansion is dependent on TCR specificity 5 and thus can be affected by selection events during T cell development 53. The enhanced proliferation of BTLA-deficient T cells in vivo is not due to changes in T cell repertoire, however, because similar results were obtained with monoclonal BTLA-deficient CD8 + OT-I T cells. Could there be a mechanistic link between the negative regulation of peripheral reconstitution, homeostatic expansion and CD8 + T cell memory formation by BTLA? T cells maintain relatively stable numbers through homeostatic proliferation and homeostatic expansion 37. Slow homeostatic proliferation occurs under normal resting conditions. By contrast, fast homeostatic T cell expansion takes place in response to lymphopenic episodes. As a result of lymphopenia, IL-7 is considerably increased, causing naive T cells to become activated by peptide-mhc complexes. In the course of their proliferation, naive T cells upregulate and resemble memory T cells generated experimentally by a defined antigen 54. These cells not only possess a memory phenotype but also resemble memory T cells in protective immune function 54, and are similarly MHC-independent and require IL-15 and/or IL-7 signals for their survival Thus, a common thread that might underlie all of the enhanced responses of BTLAdeficient T cells reported here is the response to cytokines. We have not found abnormal expression of CD122 or CD127 on BTLAdeficient memory-phenotype CD8 + T cells. Nevertheless, we cannot rule out the possibility that other aspects of cytokine responsiveness, including regulation of cell survival during the contraction phase of a response, might be enhanced in BTLA-deficient T cells, leading to increased T cell reconstitution, memory formation and homeostatic expansion. It is also possible that increases in LIGHT costimulation in BTLA-deficient T cells could affect several aspects of T cell activation. However, such a mechanism would not be expected to influence CD4 + T cell expansion, as we observed here. Finally, our results point to the potential of BTLA as a target to manipulate immune responses. Although the expression of BTLA does not distinguish T H 1 and T H 2 cells in humans, the protein is expressed by naive CD4 + and CD8 + human T cells 18. One of the central goals of cancer therapy or vaccination is to support the formation of a longlived CD8 + memory T cell pool. On the basis of our data, blockade of BTLA might enhance CD8 + memory T cell formation under appropriate immunization conditions and promote T cell reconstitution after T cell depletion and bone marrow transplantation. NATURE IMMUNOLOGY VOLUME 8 NUMBER 2 FEBRUARY
9 27 Nature Publishing Group METHODS Mice. We used C57BL/6J (B6, referred to as wild type), BTLA-deficient (Btla / ) 13,HVEM-deficient(Tnfrsf14 / ) 59 and OT-I TCR transgenic 6 mice in these studies. Initial experiments were carried out in Btla / mice backcrossed for six generations to wild-type mice and results were confirmed in Btla / mice backcrossed to wild-type mice for ten generations. CD9.1 (B6.PL) and CD45.1 (B6.SJL) congenic mice were purchased from The Jackson Laboratory. All mice were bred at The Scripps Research Institute, maintained under specific pathogen-free conditions, and used at 2 6 months of age. Experiments were conducted in accordance with National Institutes of Health guidelines for the care and use of animals and with an approved animal protocol from The Scripps Research Institute Animal Care and Use Committee. Antibodies and flow cytometry. Cell suspensions of spleen or pooled lymph nodes (inguinal, axillary, cervical and mesenteric) were prepared by standard protocols and stained for analysis by flow cytometry using PBS containing 1% FCS and 2 mm EDTA. BTLA-specific mabs PK3 and PJ196 (IgG1k) 13 and HVEM-specific mab 22 have been described. We also used the following antibodies: fluorescein isothiocyanate (FITC)- or phycoerythrin (PE)- conjugated anti-cd3 (clone 145-2C11), FITC-conjugated anti-cd4 (clone GK1.5), FITC-conjugated anti-cd62l (clone MEL-14), PE-conjugated anti- CD127 (clone A7R34), allophycocyanin- or FITC-conjugated anti-cd9.1 (clone HIS51; all from ebioscience); peridinin chlorophyll protein cyanin 5.5 (PerCP-Cy5.5)-conjugated anti-cd3e (clone 145-2C11), allophycocyanincyanin7 or PerCP-Cy5.5 conjugated anti-cd8a (clone ), PE-conjugated anti-cd8b (clone H ), allophycocyanin-conjugated or biotinylated anti- (clone IM7), PE- or allophycocyanin-conjugated anti-cd45.2 (clone 14; all from BD Biosciences); Alexa Fluor 45 conjugated anti-cd4 (clone RM4-5; Caltag Laboratories); PE-conjugated anti-cd122 (clone TM-b1), PE-conjugated anti-cd43 (clone 1B11), FITC-conjugated anti-cd9.2 (clone ), and PE-conjugated anti-v a 2 (clone B2.1; all from Pharmingen). Secondary staining reagents included PE-conjugated anti-rat IgM (clone G53-238; Pharmingen) or Fab anti-mouse IgG (ebioscience) and PerCP- Cy5.5 conjugated streptavidin (Pharmingen). Cell surface staining was done as described 25. For intracellular cytokine staining, PE- or allophycocyaninconjugated anti IFN-g (clone XMG1.2; ebioscience) and a Cytofix/Cytoperm kit (BD Biosciences) were used in accordance with the manufacturer s recommendations. In some instances, cells were pre-incubated with anti-cd16/cd32 to block Fc receptors. At least 1, viable cells were live-gated on a digital LSRII flow cytometer using Cell Quest Diva software (BD Biosciences) and analyzed by FlowJo software (TriStar). T cell activation. A total of T cells/ml was cultured in 1-ml volumes in 96-well flat-bottom plates coated with anti-cd3e (clone 145-2C11, ebioscience) at the indicated concentrations. T cells were cultured for 72 h and pulsed with 1 mci per well of [ 3 H]thymidine (PerkinElmer) for the last 16 h before [ 3 H]thymidine incorporation was determined. For the in vitro expansion of OT-I specific T cells, SIINFEKL-pulsed irradiated splenocytes were cultured with splenocytes from DC-immunized mice. Cells were collected on day 7, stained with PE-conjugated MHC-I SIINFEKL tetramer (Beckman Coulter), CD8b and CD9.2, and analyzed by flow cytometry. For cytokine responses, splenocytes were incubated for 5 h in the presence of GolgiPlug (BD Biosciences) and either 5 ng/ml of PMA and 5 ng/ml of ionomycin (both Sigma-Aldrich) or 1 6 MSIINFEKLpeptide (Anaspec), or were untreated. Cells were washed, stained for surface markers, fixed and permeabilized before intracellular staining. Measurement of cell turnover in vivo. Proliferation of cells in vivo was measured by dilution of the dye CFSE or by incorporation of BrdU (.8 mg/ ml) given in the drinking water. CFSE labeling was done with a Vybrandt CFDA-SE Cell Tracer kit (Molecular Probes). Intracellular staining for BrdU was done by using an FITC BrdU kit (BD Biosciences) in accordance with the manufacturer s instructions. Generation of bone marrow derived DCs. Bone marrow cells flushed from femurs and tibias of wild-type mice were cultured at cells/ml in complete RPMI medium supplemented with 2 ng/ml of murine granulocytemacrophage colony-stimulating factor (mgm-csf; Biosource). Every 2 d of culture, 5% of the medium was replaced with fresh medium containing 4 ng/ ml of mgm-csf. On day 4 of culture, non-adherent and loosely adherent cells were resuspended in fresh medium and transferred to fresh plates. On day 7 of culture, lipopolysaccharide (1 ng/ml; Sigma-Aldrich) was added for 1 d to induce maturation. Mature bone marrow derived DCs were purified by density gradient centrifugation (Ficoll Paque Plus, Amersham Biosciences). Maturity and phenotype of enriched bone marrow derived DCs were confirmed by flow cytometric expression of CD11c, CD86, CD8 and MHC-II. The resultant cell population was Z8% positive for CD11c +, CD86 + and MHC-II hi cells. Mature bone marrow derived DCs were pulsed with 1 6 MSIINFEKLpeptide for 1 h at 37 1C and washed twice. Each mouse was injected intravenously with mature peptide-pulsed DCs. Cell sorting and adoptive transfer of T cells. T cell subsets were obtained by negative T cell enrichment (StemCell Technologies), stained appropriately, and sorted for CD3 +,CD4 + or CD8 +,and lo or hi cells by using a Becton Dickinson Aria. Purity of the samples was routinely tested after sorting and was 496%. The indicated numbers of sorted cells were injected into mice exposed to 7.5 Gy of whole-body irradiation 1 d before cell transfer. Host lymph node and spleen cells were collected at various times after transfer and analyzed by flow cytometry. Generation of bone marrow chimeras. Bone marrow cells were obtained from CD45.1-marked wild-type or CD45.2-marked Btla / mice. Mature B, T and MHC-II-expressing cells were depleted by the use of mabs to heat-stable antigen (J11d), anti-cd9.2 (clone J1J), anti-cd4 (clone RL172), anti-cd8 (clone 3.168) and MHC-II ( s) plus complement. Contamination of purified bone marrow cells with mature B, T or MHC-II expressing cells was less than 1%. A 1:1 ratio of B6.Ly (CD )tobtla / (CD45.1 ) bone marrow cells was injected intravenously into CD9.1 (B6.PL) mice lethally irradiated with 1 Gy. T cells from these chimeras were analyzed after 3 1 months. In vivo killing assay. Wild-type target spleen cells were labeled with a high (CFSE hi ) or low (CFSE lo ) concentration of CFSE. Where indicated, the CFSE hi population was pulsed with 1 6 M SIINFEKL peptide for 1 h at 37 1C. The CFSE lo population was cultured without peptide under the same conditions. Target cells were then washed twice and mixed at a 1:1 ratio, and target cells were transferred by tail vein injection into mice. At 4 h or 16 h after transfer, mice were killed and red blood cell lysed splenocytes were analyzed for CFSE profiles. The percentage of specific lysis was determined by the following formulae: ratio ¼ (percentage of CFSE lo /percentage of CFSE hi ); percentage of specific lysis ¼ [1 (ratio transfer only/ratio peptide response) 1]. Bacterial infection. The OVA-expressing recombinant Listeria monocytogenes bacterial strain (LM-OVA) was provided by H. Shen 4. Mice were challenged with c.f.u. of LM-OVA by tail vein injection 3 d after DC immunization. Statistical analysis. Results are expressed as the mean ± s.d. Differences between groups were examined for statistical significance by the unpaired Student s t-test. In some instances, the 95% confidence intervals (95%CI) for differences between unpaired means of unequal variance are also presented as a fold increase over the wild-type mean. The percentage of naive OT-I CD8 + T cell precursors that homeostatically expanded was calculated as (number of CFSE + cells in each division)/2 n divided by the input number of cells, where n is the number of divisions. Note: Supplementary information is available on the Nature Immunology website. ACKNOWLEDGMENTS We thank P. Aliahmad and M. Fung for critically reviewing this manuscript, S. Freigang and J.T. Tan for discussion; N. Sanathara and O. Goularte for technical assistance; and the Department of Animal Resources and the Flow Cytometry Core Facility at The Scripps Research Institute for support. Supported by the National Institutes of Health (AI to J.K.), and the Swiss National Science Foundation and the Novartis Foundation (to O.B.). This is manuscript from the Scripps Research Institute. 17 VOLUME 8 NUMBER 2 FEBRUARY 27 NATURE IMMUNOLOGY
SUPPLEMENTARY INFORMATION
 Complete but curtailed T-cell response to very-low-affinity antigen Dietmar Zehn, Sarah Y. Lee & Michael J. Bevan Supp. Fig. 1: TCR chain usage among endogenous K b /Ova reactive T cells. C57BL/6 mice
Complete but curtailed T-cell response to very-low-affinity antigen Dietmar Zehn, Sarah Y. Lee & Michael J. Bevan Supp. Fig. 1: TCR chain usage among endogenous K b /Ova reactive T cells. C57BL/6 mice
Supplementary Figure 1. Characterization of basophils after reconstitution of SCID mice
 Supplementary figure legends Supplementary Figure 1. Characterization of after reconstitution of SCID mice with CD4 + CD62L + T cells. (A-C) SCID mice (n = 6 / group) were reconstituted with 2 x 1 6 CD4
Supplementary figure legends Supplementary Figure 1. Characterization of after reconstitution of SCID mice with CD4 + CD62L + T cells. (A-C) SCID mice (n = 6 / group) were reconstituted with 2 x 1 6 CD4
MATERIALS AND METHODS. Neutralizing antibodies specific to mouse Dll1, Dll4, J1 and J2 were prepared as described. 1,2 All
 MATERIALS AND METHODS Antibodies (Abs), flow cytometry analysis and cell lines Neutralizing antibodies specific to mouse Dll1, Dll4, J1 and J2 were prepared as described. 1,2 All other antibodies used
MATERIALS AND METHODS Antibodies (Abs), flow cytometry analysis and cell lines Neutralizing antibodies specific to mouse Dll1, Dll4, J1 and J2 were prepared as described. 1,2 All other antibodies used
Blocking antibodies and peptides. Rat anti-mouse PD-1 (29F.1A12, rat IgG2a, k), PD-
 Supplementary Methods Blocking antibodies and peptides. Rat anti-mouse PD-1 (29F.1A12, rat IgG2a, k), PD- L1 (10F.9G2, rat IgG2b, k), and PD-L2 (3.2, mouse IgG1) have been described (24). Anti-CTLA-4 (clone
Supplementary Methods Blocking antibodies and peptides. Rat anti-mouse PD-1 (29F.1A12, rat IgG2a, k), PD- L1 (10F.9G2, rat IgG2b, k), and PD-L2 (3.2, mouse IgG1) have been described (24). Anti-CTLA-4 (clone
NK cell flow cytometric assay In vivo DC viability and migration assay
 NK cell flow cytometric assay 6 NK cells were purified, by negative selection with the NK Cell Isolation Kit (Miltenyi iotec), from spleen and lymph nodes of 6 RAG1KO mice, injected the day before with
NK cell flow cytometric assay 6 NK cells were purified, by negative selection with the NK Cell Isolation Kit (Miltenyi iotec), from spleen and lymph nodes of 6 RAG1KO mice, injected the day before with
T-cell activation T cells migrate to secondary lymphoid tissues where they interact with antigen, antigen-presenting cells, and other lymphocytes:
 Interactions between innate immunity & adaptive immunity What happens to T cells after they leave the thymus? Naïve T cells exit the thymus and enter the bloodstream. If they remain in the bloodstream,
Interactions between innate immunity & adaptive immunity What happens to T cells after they leave the thymus? Naïve T cells exit the thymus and enter the bloodstream. If they remain in the bloodstream,
T-cell activation T cells migrate to secondary lymphoid tissues where they interact with antigen, antigen-presenting cells, and other lymphocytes:
 Interactions between innate immunity & adaptive immunity What happens to T cells after they leave the thymus? Naïve T cells exit the thymus and enter the bloodstream. If they remain in the bloodstream,
Interactions between innate immunity & adaptive immunity What happens to T cells after they leave the thymus? Naïve T cells exit the thymus and enter the bloodstream. If they remain in the bloodstream,
Determinants of Immunogenicity and Tolerance. Abul K. Abbas, MD Department of Pathology University of California San Francisco
 Determinants of Immunogenicity and Tolerance Abul K. Abbas, MD Department of Pathology University of California San Francisco EIP Symposium Feb 2016 Why do some people respond to therapeutic proteins?
Determinants of Immunogenicity and Tolerance Abul K. Abbas, MD Department of Pathology University of California San Francisco EIP Symposium Feb 2016 Why do some people respond to therapeutic proteins?
Cover Page. The handle holds various files of this Leiden University dissertation.
 Cover Page The handle http://hdl.handle.net/1887/23854 holds various files of this Leiden University dissertation. Author: Marel, Sander van der Title: Gene and cell therapy based treatment strategies
Cover Page The handle http://hdl.handle.net/1887/23854 holds various files of this Leiden University dissertation. Author: Marel, Sander van der Title: Gene and cell therapy based treatment strategies
Supplementary Figure 1. Efficient DC depletion in CD11c.DOG transgenic mice
 Supplementary Figure 1. Efficient DC depletion in CD11c.DOG transgenic mice (a) CD11c.DOG transgenic mice (tg) were treated with 8 ng/g body weight (b.w.) diphtheria toxin (DT) i.p. on day -1 and every
Supplementary Figure 1. Efficient DC depletion in CD11c.DOG transgenic mice (a) CD11c.DOG transgenic mice (tg) were treated with 8 ng/g body weight (b.w.) diphtheria toxin (DT) i.p. on day -1 and every
Supplemental Table I.
 Supplemental Table I Male / Mean ± SEM n Mean ± SEM n Body weight, g 29.2±0.4 17 29.7±0.5 17 Total cholesterol, mg/dl 534.0±30.8 17 561.6±26.1 17 HDL-cholesterol, mg/dl 9.6±0.8 17 10.1±0.7 17 Triglycerides,
Supplemental Table I Male / Mean ± SEM n Mean ± SEM n Body weight, g 29.2±0.4 17 29.7±0.5 17 Total cholesterol, mg/dl 534.0±30.8 17 561.6±26.1 17 HDL-cholesterol, mg/dl 9.6±0.8 17 10.1±0.7 17 Triglycerides,
SUPPLEMENTARY INFORMATION
 Supplemental Figure 1. Furin is efficiently deleted in CD4 + and CD8 + T cells. a, Western blot for furin and actin proteins in CD4cre-fur f/f and fur f/f Th1 cells. Wild-type and furin-deficient CD4 +
Supplemental Figure 1. Furin is efficiently deleted in CD4 + and CD8 + T cells. a, Western blot for furin and actin proteins in CD4cre-fur f/f and fur f/f Th1 cells. Wild-type and furin-deficient CD4 +
Effector mechanisms of cell-mediated immunity: Properties of effector, memory and regulatory T cells
 ICI Basic Immunology course Effector mechanisms of cell-mediated immunity: Properties of effector, memory and regulatory T cells Abul K. Abbas, MD UCSF Stages in the development of T cell responses: induction
ICI Basic Immunology course Effector mechanisms of cell-mediated immunity: Properties of effector, memory and regulatory T cells Abul K. Abbas, MD UCSF Stages in the development of T cell responses: induction
Optimizing Intracellular Flow Cytometry:
 Optimizing Intracellular Flow Cytometry: Simultaneous Detection of Cytokines and Transcription Factors An encore presentation by Jurg Rohrer, PhD, BD Biosciences 10.26.10 Outline Introduction Cytokines
Optimizing Intracellular Flow Cytometry: Simultaneous Detection of Cytokines and Transcription Factors An encore presentation by Jurg Rohrer, PhD, BD Biosciences 10.26.10 Outline Introduction Cytokines
Adaptive immune responses: T cell-mediated immunity
 MICR2209 Adaptive immune responses: T cell-mediated immunity Dr Allison Imrie allison.imrie@uwa.edu.au 1 Synopsis: In this lecture we will discuss the T-cell mediated immune response, how it is activated,
MICR2209 Adaptive immune responses: T cell-mediated immunity Dr Allison Imrie allison.imrie@uwa.edu.au 1 Synopsis: In this lecture we will discuss the T-cell mediated immune response, how it is activated,
Interferon γ regulates idiopathic pneumonia syndrome, a. Th17 + CD4 + T-cell-mediated GvH disease
 Interferon γ regulates idiopathic pneumonia syndrome, a Th17 + CD4 + T-cell-mediated GvH disease Nora Mauermann, Julia Burian, Christophe von Garnier, Stefan Dirnhofer, Davide Germano, Christine Schuett,
Interferon γ regulates idiopathic pneumonia syndrome, a Th17 + CD4 + T-cell-mediated GvH disease Nora Mauermann, Julia Burian, Christophe von Garnier, Stefan Dirnhofer, Davide Germano, Christine Schuett,
ACTIVATION AND EFFECTOR FUNCTIONS OF CELL-MEDIATED IMMUNITY AND NK CELLS. Choompone Sakonwasun, MD (Hons), FRCPT
 ACTIVATION AND EFFECTOR FUNCTIONS OF CELL-MEDIATED IMMUNITY AND NK CELLS Choompone Sakonwasun, MD (Hons), FRCPT Types of Adaptive Immunity Types of T Cell-mediated Immune Reactions CTLs = cytotoxic T lymphocytes
ACTIVATION AND EFFECTOR FUNCTIONS OF CELL-MEDIATED IMMUNITY AND NK CELLS Choompone Sakonwasun, MD (Hons), FRCPT Types of Adaptive Immunity Types of T Cell-mediated Immune Reactions CTLs = cytotoxic T lymphocytes
Supplementary Figures
 Supplementary Figures Supplementary Fig. 1. Surface thiol groups and reduction of activated T cells. (a) Activated CD8 + T-cells have high expression levels of free thiol groups on cell surface proteins.
Supplementary Figures Supplementary Fig. 1. Surface thiol groups and reduction of activated T cells. (a) Activated CD8 + T-cells have high expression levels of free thiol groups on cell surface proteins.
Immune Regulation and Tolerance
 Immune Regulation and Tolerance Immunoregulation: A balance between activation and suppression of effector cells to achieve an efficient immune response without damaging the host. Activation (immunity)
Immune Regulation and Tolerance Immunoregulation: A balance between activation and suppression of effector cells to achieve an efficient immune response without damaging the host. Activation (immunity)
T cell maturation. T-cell Maturation. What allows T cell maturation?
 T-cell Maturation What allows T cell maturation? Direct contact with thymic epithelial cells Influence of thymic hormones Growth factors (cytokines, CSF) T cell maturation T cell progenitor DN DP SP 2ry
T-cell Maturation What allows T cell maturation? Direct contact with thymic epithelial cells Influence of thymic hormones Growth factors (cytokines, CSF) T cell maturation T cell progenitor DN DP SP 2ry
Detailed step-by-step operating procedures for NK cell and CTL degranulation assays
 Supplemental methods Detailed step-by-step operating procedures for NK cell and CTL degranulation assays Materials PBMC isolated from patients, relatives and healthy donors as control K562 cells (ATCC,
Supplemental methods Detailed step-by-step operating procedures for NK cell and CTL degranulation assays Materials PBMC isolated from patients, relatives and healthy donors as control K562 cells (ATCC,
Antigen Presentation and T Lymphocyte Activation. Abul K. Abbas UCSF. FOCiS
 1 Antigen Presentation and T Lymphocyte Activation Abul K. Abbas UCSF FOCiS 2 Lecture outline Dendritic cells and antigen presentation The role of the MHC T cell activation Costimulation, the B7:CD28 family
1 Antigen Presentation and T Lymphocyte Activation Abul K. Abbas UCSF FOCiS 2 Lecture outline Dendritic cells and antigen presentation The role of the MHC T cell activation Costimulation, the B7:CD28 family
B6/COLODR/SPL/11C/83/LAP/#2.006 B6/COLODR/SPL/11C/86/LAP/#2.016 CD11C B6/COLODR/SPL/11C/80/LAP/#2.011 CD11C
 CD3-specific antibody-induced immune tolerance and suppression of autoimmune encephalomyelitis involves TGF-β production through phagocytes digesting apoptotic T cells Sylvain Perruche 1,3, Pin Zhang 1,
CD3-specific antibody-induced immune tolerance and suppression of autoimmune encephalomyelitis involves TGF-β production through phagocytes digesting apoptotic T cells Sylvain Perruche 1,3, Pin Zhang 1,
TITLE: MODULATION OF T CELL TOLERANCE IN A MURINE MODEL FOR IMMUNOTHERAPY OF PROSTATIC ADENOCARCINOMA
 AD Award Number: DAMD17-01-1-0085 TITLE: MODULATION OF T CELL TOLERANCE IN A MURINE MODEL FOR IMMUNOTHERAPY OF PROSTATIC ADENOCARCINOMA PRINCIPAL INVESTIGATOR: ARTHUR A HURWITZ, Ph.d. CONTRACTING ORGANIZATION:
AD Award Number: DAMD17-01-1-0085 TITLE: MODULATION OF T CELL TOLERANCE IN A MURINE MODEL FOR IMMUNOTHERAPY OF PROSTATIC ADENOCARCINOMA PRINCIPAL INVESTIGATOR: ARTHUR A HURWITZ, Ph.d. CONTRACTING ORGANIZATION:
Therapeutic PD L1 and LAG 3 blockade rapidly clears established blood stage Plasmodium infection
 Supplementary Information Therapeutic PD L1 and LAG 3 blockade rapidly clears established blood stage Plasmodium infection Noah S. Butler, Jacqueline Moebius, Lecia L. Pewe, Boubacar Traore, Ogobara K.
Supplementary Information Therapeutic PD L1 and LAG 3 blockade rapidly clears established blood stage Plasmodium infection Noah S. Butler, Jacqueline Moebius, Lecia L. Pewe, Boubacar Traore, Ogobara K.
Supporting Information
 Supporting Information lpek et al. 1.173/pnas.1121217 SI Materials and Methods Mice. cell knockout, inos / (Taconic arms), Rag1 /, INγR /, and IL-12p4 / mice (The Jackson Laboratory) were maintained and/or
Supporting Information lpek et al. 1.173/pnas.1121217 SI Materials and Methods Mice. cell knockout, inos / (Taconic arms), Rag1 /, INγR /, and IL-12p4 / mice (The Jackson Laboratory) were maintained and/or
Cell isolation. Spleen and lymph nodes (axillary, inguinal) were removed from mice
 Supplementary Methods: Cell isolation. Spleen and lymph nodes (axillary, inguinal) were removed from mice and gently meshed in DMEM containing 10% FBS to prepare for single cell suspensions. CD4 + CD25
Supplementary Methods: Cell isolation. Spleen and lymph nodes (axillary, inguinal) were removed from mice and gently meshed in DMEM containing 10% FBS to prepare for single cell suspensions. CD4 + CD25
Test Bank for Basic Immunology Functions and Disorders of the Immune System 4th Edition by Abbas
 Test Bank for Basic Immunology Functions and Disorders of the Immune System 4th Edition by Abbas Chapter 04: Antigen Recognition in the Adaptive Immune System Test Bank MULTIPLE CHOICE 1. Most T lymphocytes
Test Bank for Basic Immunology Functions and Disorders of the Immune System 4th Edition by Abbas Chapter 04: Antigen Recognition in the Adaptive Immune System Test Bank MULTIPLE CHOICE 1. Most T lymphocytes
Supplementary Figure S1. PTPN2 levels are not altered in proliferating CD8+ T cells. Lymph node (LN) CD8+ T cells from C57BL/6 mice were stained with
 Supplementary Figure S1. PTPN2 levels are not altered in proliferating CD8+ T cells. Lymph node (LN) CD8+ T cells from C57BL/6 mice were stained with CFSE and stimulated with plate-bound α-cd3ε (10µg/ml)
Supplementary Figure S1. PTPN2 levels are not altered in proliferating CD8+ T cells. Lymph node (LN) CD8+ T cells from C57BL/6 mice were stained with CFSE and stimulated with plate-bound α-cd3ε (10µg/ml)
W/T Itgam -/- F4/80 CD115. F4/80 hi CD115 + F4/80 + CD115 +
 F4/8 % in the peritoneal lavage 6 4 2 p=.15 n.s p=.76 CD115 F4/8 hi CD115 + F4/8 + CD115 + F4/8 hi CD115 + F4/8 + CD115 + MHCII MHCII Supplementary Figure S1. CD11b deficiency affects the cellular responses
F4/8 % in the peritoneal lavage 6 4 2 p=.15 n.s p=.76 CD115 F4/8 hi CD115 + F4/8 + CD115 + F4/8 hi CD115 + F4/8 + CD115 + MHCII MHCII Supplementary Figure S1. CD11b deficiency affects the cellular responses
General Overview of Immunology. Kimberly S. Schluns, Ph.D. Associate Professor Department of Immunology UT MD Anderson Cancer Center
 General Overview of Immunology Kimberly S. Schluns, Ph.D. Associate Professor Department of Immunology UT MD Anderson Cancer Center Objectives Describe differences between innate and adaptive immune responses
General Overview of Immunology Kimberly S. Schluns, Ph.D. Associate Professor Department of Immunology UT MD Anderson Cancer Center Objectives Describe differences between innate and adaptive immune responses
Supplemental Information. T Cells Enhance Autoimmunity by Restraining Regulatory T Cell Responses via an Interleukin-23-Dependent Mechanism
 Immunity, Volume 33 Supplemental Information T Cells Enhance Autoimmunity by Restraining Regulatory T Cell Responses via an Interleukin-23-Dependent Mechanism Franziska Petermann, Veit Rothhammer, Malte
Immunity, Volume 33 Supplemental Information T Cells Enhance Autoimmunity by Restraining Regulatory T Cell Responses via an Interleukin-23-Dependent Mechanism Franziska Petermann, Veit Rothhammer, Malte
Phenotypical and Functional Analysis of Peripheral T Cells in Foxn1 Transgenic Mice: Effects of Aging
 Loyola University Chicago Loyola ecommons Master's Theses Theses and Dissertations 2010 Phenotypical and Functional Analysis of Peripheral T Cells in Foxn1 Transgenic Mice: Effects of Aging Paulette Krishack
Loyola University Chicago Loyola ecommons Master's Theses Theses and Dissertations 2010 Phenotypical and Functional Analysis of Peripheral T Cells in Foxn1 Transgenic Mice: Effects of Aging Paulette Krishack
Naive and memory CD8 T cell responses after antigen stimulation in vivo
 University of Iowa Iowa Research Online Theses and Dissertations Summer 2011 Naive and memory CD8 T cell responses after antigen stimulation in vivo Matthew David Martin University of Iowa Copyright 2011
University of Iowa Iowa Research Online Theses and Dissertations Summer 2011 Naive and memory CD8 T cell responses after antigen stimulation in vivo Matthew David Martin University of Iowa Copyright 2011
Supporting Online Material for
 www.sciencemag.org/cgi/content/full/1175194/dc1 Supporting Online Material for A Vital Role for Interleukin-21 in the Control of a Chronic Viral Infection John S. Yi, Ming Du, Allan J. Zajac* *To whom
www.sciencemag.org/cgi/content/full/1175194/dc1 Supporting Online Material for A Vital Role for Interleukin-21 in the Control of a Chronic Viral Infection John S. Yi, Ming Du, Allan J. Zajac* *To whom
Supplementary Figure S1. Flow cytometric analysis of the expression of Thy1 in NH cells. Flow cytometric analysis of the expression of T1/ST2 and
 Supplementary Figure S1. Flow cytometric analysis of the expression of Thy1 in NH cells. Flow cytometric analysis of the expression of T1/ST2 and Thy1 in NH cells derived from the lungs of naïve mice.
Supplementary Figure S1. Flow cytometric analysis of the expression of Thy1 in NH cells. Flow cytometric analysis of the expression of T1/ST2 and Thy1 in NH cells derived from the lungs of naïve mice.
and follicular helper T cells is Egr2-dependent. (a) Diagrammatic representation of the
 Supplementary Figure 1. LAG3 + Treg-mediated regulation of germinal center B cells and follicular helper T cells is Egr2-dependent. (a) Diagrammatic representation of the experimental protocol for the
Supplementary Figure 1. LAG3 + Treg-mediated regulation of germinal center B cells and follicular helper T cells is Egr2-dependent. (a) Diagrammatic representation of the experimental protocol for the
Micro 204. Cytotoxic T Lymphocytes (CTL) Lewis Lanier
 Micro 204 Cytotoxic T Lymphocytes (CTL) Lewis Lanier Lewis.Lanier@ucsf.edu Lymphocyte-mediated Cytotoxicity CD8 + αβ-tcr + T cells CD4 + αβ-tcr + T cells γδ-tcr + T cells Natural Killer cells CD8 + αβ-tcr
Micro 204 Cytotoxic T Lymphocytes (CTL) Lewis Lanier Lewis.Lanier@ucsf.edu Lymphocyte-mediated Cytotoxicity CD8 + αβ-tcr + T cells CD4 + αβ-tcr + T cells γδ-tcr + T cells Natural Killer cells CD8 + αβ-tcr
Supplemental Materials
 Supplemental Materials Programmed death one homolog maintains the pool size of regulatory T cells by promoting their differentiation and stability Qi Wang 1, Jianwei He 1, Dallas B. Flies 2, Liqun Luo
Supplemental Materials Programmed death one homolog maintains the pool size of regulatory T cells by promoting their differentiation and stability Qi Wang 1, Jianwei He 1, Dallas B. Flies 2, Liqun Luo
T Cell Development. Xuefang Cao, MD, PhD. November 3, 2015
 T Cell Development Xuefang Cao, MD, PhD November 3, 2015 Thymocytes in the cortex of the thymus Early thymocytes development Positive and negative selection Lineage commitment Exit from the thymus and
T Cell Development Xuefang Cao, MD, PhD November 3, 2015 Thymocytes in the cortex of the thymus Early thymocytes development Positive and negative selection Lineage commitment Exit from the thymus and
Supplementary Table; Supplementary Figures and legends S1-S21; Supplementary Materials and Methods
 Silva et al. PTEN posttranslational inactivation and hyperactivation of the PI3K/Akt pathway sustain primary T cell leukemia viability Supplementary Table; Supplementary Figures and legends S1-S21; Supplementary
Silva et al. PTEN posttranslational inactivation and hyperactivation of the PI3K/Akt pathway sustain primary T cell leukemia viability Supplementary Table; Supplementary Figures and legends S1-S21; Supplementary
Supplementary Figures
 Inhibition of Pulmonary Anti Bacterial Defense by IFN γ During Recovery from Influenza Infection By Keer Sun and Dennis W. Metzger Supplementary Figures d a Ly6G Percentage survival f 1 75 5 1 25 1 5 1
Inhibition of Pulmonary Anti Bacterial Defense by IFN γ During Recovery from Influenza Infection By Keer Sun and Dennis W. Metzger Supplementary Figures d a Ly6G Percentage survival f 1 75 5 1 25 1 5 1
Supplementary Information:
 Supplementary Information: Follicular regulatory T cells with Bcl6 expression suppress germinal center reactions by Yeonseok Chung, Shinya Tanaka, Fuliang Chu, Roza Nurieva, Gustavo J. Martinez, Seema
Supplementary Information: Follicular regulatory T cells with Bcl6 expression suppress germinal center reactions by Yeonseok Chung, Shinya Tanaka, Fuliang Chu, Roza Nurieva, Gustavo J. Martinez, Seema
activation with anti-cd3/cd28 beads and 3d following transduction. Supplemental Figure 2 shows
 Supplemental Data Supplemental Figure 1 compares CXCR4 expression in untreated CD8 + T cells, following activation with anti-cd3/cd28 beads and 3d following transduction. Supplemental Figure 2 shows the
Supplemental Data Supplemental Figure 1 compares CXCR4 expression in untreated CD8 + T cells, following activation with anti-cd3/cd28 beads and 3d following transduction. Supplemental Figure 2 shows the
SUPPORTING INFORMATIONS
 SUPPORTING INFORMATIONS Mice MT/ret RetCD3ε KO α-cd25 treated MT/ret Age 1 month 3 mnths 6 months 1 month 3 months 6 months 1 month 3 months 6 months 2/87 Survival 87/87 incidence of 17/87 1 ary tumor
SUPPORTING INFORMATIONS Mice MT/ret RetCD3ε KO α-cd25 treated MT/ret Age 1 month 3 mnths 6 months 1 month 3 months 6 months 1 month 3 months 6 months 2/87 Survival 87/87 incidence of 17/87 1 ary tumor
Optimizing Intracellular Flow Cytometry
 Optimizing Intracellular Flow Cytometry Detection of Cytokines, Transcription Factors, and Phosphoprotein by Flow Cytometry Presented by Erika O Donnell, PhD, BD Biosciences 23-14876-00 Outline Basic principles
Optimizing Intracellular Flow Cytometry Detection of Cytokines, Transcription Factors, and Phosphoprotein by Flow Cytometry Presented by Erika O Donnell, PhD, BD Biosciences 23-14876-00 Outline Basic principles
Supplemental Methods. CD107a assay
 Supplemental Methods CD107a assay For each T cell culture that was tested, two tubes were prepared. One tube contained BCMA-K562 cells, and the other tube contained NGFR-K562 cells. Both tubes contained
Supplemental Methods CD107a assay For each T cell culture that was tested, two tubes were prepared. One tube contained BCMA-K562 cells, and the other tube contained NGFR-K562 cells. Both tubes contained
Scott Abrams, Ph.D. Professor of Oncology, x4375 Kuby Immunology SEVENTH EDITION
 Scott Abrams, Ph.D. Professor of Oncology, x4375 scott.abrams@roswellpark.org Kuby Immunology SEVENTH EDITION CHAPTER 11 T-Cell Activation, Differentiation, and Memory Copyright 2013 by W. H. Freeman and
Scott Abrams, Ph.D. Professor of Oncology, x4375 scott.abrams@roswellpark.org Kuby Immunology SEVENTH EDITION CHAPTER 11 T-Cell Activation, Differentiation, and Memory Copyright 2013 by W. H. Freeman and
Optimizing Intracellular Flow Cytometry:
 Optimizing Intracellular Flow Cytometry: Simultaneous Detection of Cytokines and Transcription Factors Presented by Jurg Rohrer, PhD, BD Biosciences 23-10780-00 Outline Introduction Cytokines Transcription
Optimizing Intracellular Flow Cytometry: Simultaneous Detection of Cytokines and Transcription Factors Presented by Jurg Rohrer, PhD, BD Biosciences 23-10780-00 Outline Introduction Cytokines Transcription
Examples of questions for Cellular Immunology/Cellular Biology and Immunology
 Examples of questions for Cellular Immunology/Cellular Biology and Immunology Each student gets a set of 6 questions, so that each set contains different types of questions and that the set of questions
Examples of questions for Cellular Immunology/Cellular Biology and Immunology Each student gets a set of 6 questions, so that each set contains different types of questions and that the set of questions
Canberra, Australia). CD11c-DTR-OVA-GFP (B6.CD11c-OVA), B6.luc + and. Cancer Research Center, Germany). B6 or BALB/c.FoxP3-DTR-GFP mice were
 Supplemental Materials and Methods Mice Female C57BL/6 (B6, I-E null, H-2 b ), BALB/c (H-2 d ) + ), FVB/N (H-2 q, I-E null, CD45.1 + ), and B6D2F1 (H-2 b/d ) mice were purchased from the Animal Resources
Supplemental Materials and Methods Mice Female C57BL/6 (B6, I-E null, H-2 b ), BALB/c (H-2 d ) + ), FVB/N (H-2 q, I-E null, CD45.1 + ), and B6D2F1 (H-2 b/d ) mice were purchased from the Animal Resources
T Cell Differentiation
 T Cell Differentiation Ned Braunstein, MD MHC control of Immune Responsiveness: Concept Whether or not an individual makes an immune response to a particular antigen depends on what MHC alleles an individual
T Cell Differentiation Ned Braunstein, MD MHC control of Immune Responsiveness: Concept Whether or not an individual makes an immune response to a particular antigen depends on what MHC alleles an individual
The autoimmune disease-associated PTPN22 variant promotes calpain-mediated Lyp/Pep
 SUPPLEMENTARY INFORMATION The autoimmune disease-associated PTPN22 variant promotes calpain-mediated Lyp/Pep degradation associated with lymphocyte and dendritic cell hyperresponsiveness Jinyi Zhang, Naima
SUPPLEMENTARY INFORMATION The autoimmune disease-associated PTPN22 variant promotes calpain-mediated Lyp/Pep degradation associated with lymphocyte and dendritic cell hyperresponsiveness Jinyi Zhang, Naima
Supplemental Figure 1. Signature gene expression in in vitro differentiated Th0, Th1, Th2, Th17 and Treg cells. (A) Naïve CD4 + T cells were cultured
 Supplemental Figure 1. Signature gene expression in in vitro differentiated Th0, Th1, Th2, Th17 and Treg cells. (A) Naïve CD4 + T cells were cultured under Th0, Th1, Th2, Th17, and Treg conditions. mrna
Supplemental Figure 1. Signature gene expression in in vitro differentiated Th0, Th1, Th2, Th17 and Treg cells. (A) Naïve CD4 + T cells were cultured under Th0, Th1, Th2, Th17, and Treg conditions. mrna
T Cell Activation, Costimulation and Regulation
 1 T Cell Activation, Costimulation and Regulation Abul K. Abbas, MD University of California San Francisco 2 Lecture outline T cell antigen recognition and activation Costimulation, the B7:CD28 family
1 T Cell Activation, Costimulation and Regulation Abul K. Abbas, MD University of California San Francisco 2 Lecture outline T cell antigen recognition and activation Costimulation, the B7:CD28 family
SUPPLEMENTARY INFORMATION
 doi: 1.138/nature775 4 O.D. (595-655) 3 1 -ζ no antibody isotype ctrl Plated Soluble 1F6 397 7H11 Supplementary Figure 1 Soluble and plated anti- Abs induce -! signalling. B3Z cells stably expressing!
doi: 1.138/nature775 4 O.D. (595-655) 3 1 -ζ no antibody isotype ctrl Plated Soluble 1F6 397 7H11 Supplementary Figure 1 Soluble and plated anti- Abs induce -! signalling. B3Z cells stably expressing!
The Adaptive Immune Responses
 The Adaptive Immune Responses The two arms of the immune responses are; 1) the cell mediated, and 2) the humoral responses. In this chapter we will discuss the two responses in detail and we will start
The Adaptive Immune Responses The two arms of the immune responses are; 1) the cell mediated, and 2) the humoral responses. In this chapter we will discuss the two responses in detail and we will start
Supplementary Figure Legends. group) and analyzed for Siglec-G expression utilizing a monoclonal antibody to Siglec-G (clone SH2.1).
 Supplementary Figure Legends Supplemental Figure : Naïve T cells express Siglec-G. Splenocytes were isolated from WT B or Siglec-G -/- animals that have not been transplanted (n= per group) and analyzed
Supplementary Figure Legends Supplemental Figure : Naïve T cells express Siglec-G. Splenocytes were isolated from WT B or Siglec-G -/- animals that have not been transplanted (n= per group) and analyzed
Nature Immunology: doi: /ni Supplementary Figure 1. Gene expression profile of CD4 + T cells and CTL responses in Bcl6-deficient mice.
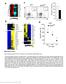 Supplementary Figure 1 Gene expression profile of CD4 + T cells and CTL responses in Bcl6-deficient mice. (a) Gene expression profile in the resting CD4 + T cells were analyzed by an Affymetrix microarray
Supplementary Figure 1 Gene expression profile of CD4 + T cells and CTL responses in Bcl6-deficient mice. (a) Gene expression profile in the resting CD4 + T cells were analyzed by an Affymetrix microarray
5/1/13. The proportion of thymus that produces T cells decreases with age. The cellular organization of the thymus
 T cell precursors migrate from the bone marrow via the blood to the thymus to mature 1 2 The cellular organization of the thymus The proportion of thymus that produces T cells decreases with age 3 4 1
T cell precursors migrate from the bone marrow via the blood to the thymus to mature 1 2 The cellular organization of the thymus The proportion of thymus that produces T cells decreases with age 3 4 1
Ex vivo Human Antigen-specific T Cell Proliferation and Degranulation Willemijn Hobo 1, Wieger Norde 1 and Harry Dolstra 2*
 Ex vivo Human Antigen-specific T Cell Proliferation and Degranulation Willemijn Hobo 1, Wieger Norde 1 and Harry Dolstra 2* 1 Department of Laboratory Medicine - Laboratory of Hematology, Radboud University
Ex vivo Human Antigen-specific T Cell Proliferation and Degranulation Willemijn Hobo 1, Wieger Norde 1 and Harry Dolstra 2* 1 Department of Laboratory Medicine - Laboratory of Hematology, Radboud University
The encephalitogenicity of TH17 cells is dependent on IL-1- and IL-23- induced production of the cytokine GM-CSF
 CORRECTION NOTICE Nat.Immunol. 12, 568 575 (2011) The encephalitogenicity of TH17 cells is dependent on IL-1- and IL-23- induced production of the cytokine GM-CSF Mohamed El-Behi, Bogoljub Ciric, Hong
CORRECTION NOTICE Nat.Immunol. 12, 568 575 (2011) The encephalitogenicity of TH17 cells is dependent on IL-1- and IL-23- induced production of the cytokine GM-CSF Mohamed El-Behi, Bogoljub Ciric, Hong
FOCiS. Lecture outline. The immunological equilibrium: balancing lymphocyte activation and control. Immunological tolerance and immune regulation -- 1
 1 Immunological tolerance and immune regulation -- 1 Abul K. Abbas UCSF FOCiS 2 Lecture outline Principles of immune regulation Self-tolerance; mechanisms of central and peripheral tolerance Inhibitory
1 Immunological tolerance and immune regulation -- 1 Abul K. Abbas UCSF FOCiS 2 Lecture outline Principles of immune regulation Self-tolerance; mechanisms of central and peripheral tolerance Inhibitory
L-selectin Is Essential for Delivery of Activated CD8 + T Cells to Virus-Infected Organs for Protective Immunity
 Cell Reports Supplemental Information L-selectin Is Essential for Delivery of Activated CD8 + T Cells to Virus-Infected Organs for Protective Immunity Rebar N. Mohammed, H. Angharad Watson, Miriam Vigar,
Cell Reports Supplemental Information L-selectin Is Essential for Delivery of Activated CD8 + T Cells to Virus-Infected Organs for Protective Immunity Rebar N. Mohammed, H. Angharad Watson, Miriam Vigar,
Defensive mechanisms include :
 Acquired Immunity Defensive mechanisms include : 1) Innate immunity (Natural or Non specific) 2) Acquired immunity (Adaptive or Specific) Cell-mediated immunity Humoral immunity Two mechanisms 1) Humoral
Acquired Immunity Defensive mechanisms include : 1) Innate immunity (Natural or Non specific) 2) Acquired immunity (Adaptive or Specific) Cell-mediated immunity Humoral immunity Two mechanisms 1) Humoral
SEVENTH EDITION CHAPTER
 Judy Owen Jenni Punt Sharon Stranford Kuby Immunology SEVENTH EDITION CHAPTER 16 Tolerance, Autoimmunity, and Transplantation Copyright 2013 by W. H. Freeman and Company Immune tolerance: history * Some
Judy Owen Jenni Punt Sharon Stranford Kuby Immunology SEVENTH EDITION CHAPTER 16 Tolerance, Autoimmunity, and Transplantation Copyright 2013 by W. H. Freeman and Company Immune tolerance: history * Some
T cell protein tyrosine phosphatase attenuates T cell signaling to maintain tolerance in mice
 SUPPLEMENTAL METHODS T cell protein tyrosine phosphatase attenuates T cell signaling to maintain tolerance in mice Florian Wiede 1, Benjamin J. Shields 1, Sock Hui Chew 1, Konstantinos Kyparissoudis 2,
SUPPLEMENTAL METHODS T cell protein tyrosine phosphatase attenuates T cell signaling to maintain tolerance in mice Florian Wiede 1, Benjamin J. Shields 1, Sock Hui Chew 1, Konstantinos Kyparissoudis 2,
Interleukin-7 Differentially Regulates The Activation, Proliferation, And Homing Of T-cells: Implications For Immunotherapy
 University of Central Florida Electronic Theses and Dissertations Doctoral Dissertation (Open Access) Interleukin-7 Differentially Regulates The Activation, Proliferation, And Homing Of T-cells: Implications
University of Central Florida Electronic Theses and Dissertations Doctoral Dissertation (Open Access) Interleukin-7 Differentially Regulates The Activation, Proliferation, And Homing Of T-cells: Implications
Commercially available HLA Class II tetramers (Beckman Coulter) conjugated to
 Class II tetramer staining Commercially available HLA Class II tetramers (Beckman Coulter) conjugated to PE were combined with dominant HIV epitopes (DRB1*0101-DRFYKTLRAEQASQEV, DRB1*0301- PEKEVLVWKFDSRLAFHH,
Class II tetramer staining Commercially available HLA Class II tetramers (Beckman Coulter) conjugated to PE were combined with dominant HIV epitopes (DRB1*0101-DRFYKTLRAEQASQEV, DRB1*0301- PEKEVLVWKFDSRLAFHH,
T Cell Development II: Positive and Negative Selection
 T Cell Development II: Positive and Negative Selection 8 88 The two phases of thymic development: - production of T cell receptors for antigen, by rearrangement of the TCR genes CD4 - selection of T cells
T Cell Development II: Positive and Negative Selection 8 88 The two phases of thymic development: - production of T cell receptors for antigen, by rearrangement of the TCR genes CD4 - selection of T cells
Central tolerance. Mechanisms of Immune Tolerance. Regulation of the T cell response
 Immunoregulation: A balance between activation and suppression that achieves an efficient immune response without damaging the host. Mechanisms of Immune Tolerance ACTIVATION (immunity) SUPPRESSION (tolerance)
Immunoregulation: A balance between activation and suppression that achieves an efficient immune response without damaging the host. Mechanisms of Immune Tolerance ACTIVATION (immunity) SUPPRESSION (tolerance)
Mechanisms of Immune Tolerance
 Immunoregulation: A balance between activation and suppression that achieves an efficient immune response without damaging the host. ACTIVATION (immunity) SUPPRESSION (tolerance) Autoimmunity Immunodeficiency
Immunoregulation: A balance between activation and suppression that achieves an efficient immune response without damaging the host. ACTIVATION (immunity) SUPPRESSION (tolerance) Autoimmunity Immunodeficiency
Supplemental Figure 1. Cell-bound Cetuximab reduces EGFR staining intensity. Blood
 Antibody-mediated depletion of CD19-CAR T cells Supplemental 1 Supplemental Materials Supplemental Figure 1. Supplemental Figure 1. Cell-bound Cetuximab reduces EGFR staining intensity. Blood cells were
Antibody-mediated depletion of CD19-CAR T cells Supplemental 1 Supplemental Materials Supplemental Figure 1. Supplemental Figure 1. Cell-bound Cetuximab reduces EGFR staining intensity. Blood cells were
A crucial role for HVEM and BTLA in preventing intestinal inflammation
 Washington University School of Medicine Digital Commons@Becker Open Access Publications 2008 A crucial role for HVEM and BTLA in preventing intestinal inflammation Marcos W. Steinberg La Jolla Institute
Washington University School of Medicine Digital Commons@Becker Open Access Publications 2008 A crucial role for HVEM and BTLA in preventing intestinal inflammation Marcos W. Steinberg La Jolla Institute
A Slfn2 mutation causes lymphoid and myeloid immunodeficiency due to loss of immune cell quiescence
 Supplementary Information A Slfn mutation causes lymphoid and myeloid immunodeficiency due to loss of immune cell quiescence Michael Berger, Philippe Kres, Karine Crozat, Xiaohong Li, Ben A. Croker, Owen
Supplementary Information A Slfn mutation causes lymphoid and myeloid immunodeficiency due to loss of immune cell quiescence Michael Berger, Philippe Kres, Karine Crozat, Xiaohong Li, Ben A. Croker, Owen
Supplemental Figure 1
 Supplemental Figure 1 1a 1c PD-1 MFI fold change 6 5 4 3 2 1 IL-1α IL-2 IL-4 IL-6 IL-1 IL-12 IL-13 IL-15 IL-17 IL-18 IL-21 IL-23 IFN-α Mut Human PD-1 promoter SBE-D 5 -GTCTG- -1.2kb SBE-P -CAGAC- -1.kb
Supplemental Figure 1 1a 1c PD-1 MFI fold change 6 5 4 3 2 1 IL-1α IL-2 IL-4 IL-6 IL-1 IL-12 IL-13 IL-15 IL-17 IL-18 IL-21 IL-23 IFN-α Mut Human PD-1 promoter SBE-D 5 -GTCTG- -1.2kb SBE-P -CAGAC- -1.kb
Rapid antigen-specific T cell enrichment (Rapid ARTE)
 Direct ex vivo characterization of human antigen-specific CD154+CD4+ T cell Rapid antigen-specific T cell enrichment (Rapid ARTE) Introduction Workflow Antigen (ag)-specific T cells play a central role
Direct ex vivo characterization of human antigen-specific CD154+CD4+ T cell Rapid antigen-specific T cell enrichment (Rapid ARTE) Introduction Workflow Antigen (ag)-specific T cells play a central role
Principles of Adaptive Immunity
 Principles of Adaptive Immunity Chapter 3 Parham Hans de Haard 17 th of May 2010 Agenda Recognition molecules of adaptive immune system Features adaptive immune system Immunoglobulins and T-cell receptors
Principles of Adaptive Immunity Chapter 3 Parham Hans de Haard 17 th of May 2010 Agenda Recognition molecules of adaptive immune system Features adaptive immune system Immunoglobulins and T-cell receptors
The Adaptive Immune Response. B-cells
 The Adaptive Immune Response B-cells The innate immune system provides immediate protection. The adaptive response takes time to develop and is antigen specific. Activation of B and T lymphocytes Naive
The Adaptive Immune Response B-cells The innate immune system provides immediate protection. The adaptive response takes time to develop and is antigen specific. Activation of B and T lymphocytes Naive
CD40-Activated B Cells Can Efficiently Prime Antigen- Specific Naïve CD8 + T Cells to Generate Effector but Not Memory T cells
 CD40-Activated B Cells Can Efficiently Prime Antigen- Specific Naïve CD8 + T Cells to Generate Effector but Not Memory T cells Mélissa Mathieu 1,2, Natacha Cotta-Grand 1,2, Jean-François Daudelin 1, Salix
CD40-Activated B Cells Can Efficiently Prime Antigen- Specific Naïve CD8 + T Cells to Generate Effector but Not Memory T cells Mélissa Mathieu 1,2, Natacha Cotta-Grand 1,2, Jean-François Daudelin 1, Salix
SUPPLEMENTARY FIGURE 1
 SUPPLEMENTARY FIGURE 1 A LN Cell count (1 ) 1 3 1 CD+ 1 1 CDL lo CD hi 1 CD+FoxP3+ 1 1 1 7 3 3 3 % of cells 9 7 7 % of cells CD+ 3 1 % of cells CDL lo CD hi 1 1 % of CD+ cells CD+FoxP3+ 3 1 % of CD+ T
SUPPLEMENTARY FIGURE 1 A LN Cell count (1 ) 1 3 1 CD+ 1 1 CDL lo CD hi 1 CD+FoxP3+ 1 1 1 7 3 3 3 % of cells 9 7 7 % of cells CD+ 3 1 % of cells CDL lo CD hi 1 1 % of CD+ cells CD+FoxP3+ 3 1 % of CD+ T
NKTR-255: Accessing The Immunotherapeutic Potential Of IL-15 for NK Cell Therapies
 NKTR-255: Accessing The Immunotherapeutic Potential Of IL-15 for NK Cell Therapies Saul Kivimäe Senior Scientist, Research Biology Nektar Therapeutics NK Cell-Based Cancer Immunotherapy, September 26-27,
NKTR-255: Accessing The Immunotherapeutic Potential Of IL-15 for NK Cell Therapies Saul Kivimäe Senior Scientist, Research Biology Nektar Therapeutics NK Cell-Based Cancer Immunotherapy, September 26-27,
B220 CD4 CD8. Figure 1. Confocal Image of Sensitized HLN. Representative image of a sensitized HLN
 B220 CD4 CD8 Natarajan et al., unpublished data Figure 1. Confocal Image of Sensitized HLN. Representative image of a sensitized HLN showing B cell follicles and T cell areas. 20 µm thick. Image of magnification
B220 CD4 CD8 Natarajan et al., unpublished data Figure 1. Confocal Image of Sensitized HLN. Representative image of a sensitized HLN showing B cell follicles and T cell areas. 20 µm thick. Image of magnification
Combined Rho-kinase inhibition and immunogenic cell death triggers and propagates immunity against cancer
 Supplementary Information Combined Rho-kinase inhibition and immunogenic cell death triggers and propagates immunity against cancer Gi-Hoon Nam, Eun-Jung Lee, Yoon Kyoung Kim, Yeonsun Hong, Yoonjeong Choi,
Supplementary Information Combined Rho-kinase inhibition and immunogenic cell death triggers and propagates immunity against cancer Gi-Hoon Nam, Eun-Jung Lee, Yoon Kyoung Kim, Yeonsun Hong, Yoonjeong Choi,
Direct ex vivo characterization of human antigen-specific CD154 + CD4 + T cells Rapid antigen-reactive T cell enrichment (Rapid ARTE)
 Direct ex vivo characterization of human antigen-specific CD154 + CD4 + T cells Rapid antigen-reactive T cell enrichment (Rapid ARTE) Introduction Workflow Antigen (ag)-specific T cells play a central
Direct ex vivo characterization of human antigen-specific CD154 + CD4 + T cells Rapid antigen-reactive T cell enrichment (Rapid ARTE) Introduction Workflow Antigen (ag)-specific T cells play a central
NK1.1 þ CD8 þ T cells escape TGF-b control and contribute to early microbial pathogen response
 Received 24 Apr 214 Accepted 5 Sep 214 Published 6 Oct 214 DOI: 1.138/ncomms615 NK1.1 þ CD8 þ T cells escape TGF-b control and contribute to early microbial pathogen response Anne L. Ruiz 1,2,3,4, Saidi
Received 24 Apr 214 Accepted 5 Sep 214 Published 6 Oct 214 DOI: 1.138/ncomms615 NK1.1 þ CD8 þ T cells escape TGF-b control and contribute to early microbial pathogen response Anne L. Ruiz 1,2,3,4, Saidi
SUPPLEMENTARY INFORMATION
 doi:1.138/nature1554 a TNF-α + in CD4 + cells [%] 1 GF SPF 6 b IL-1 + in CD4 + cells [%] 5 4 3 2 1 Supplementary Figure 1. Effect of microbiota on cytokine profiles of T cells in GALT. Frequencies of TNF-α
doi:1.138/nature1554 a TNF-α + in CD4 + cells [%] 1 GF SPF 6 b IL-1 + in CD4 + cells [%] 5 4 3 2 1 Supplementary Figure 1. Effect of microbiota on cytokine profiles of T cells in GALT. Frequencies of TNF-α
Immunology Basics Relevant to Cancer Immunotherapy: T Cell Activation, Costimulation, and Effector T Cells
 Immunology Basics Relevant to Cancer Immunotherapy: T Cell Activation, Costimulation, and Effector T Cells Andrew H. Lichtman, M.D. Ph.D. Department of Pathology Brigham and Women s Hospital and Harvard
Immunology Basics Relevant to Cancer Immunotherapy: T Cell Activation, Costimulation, and Effector T Cells Andrew H. Lichtman, M.D. Ph.D. Department of Pathology Brigham and Women s Hospital and Harvard
Supplementary Materials for
 www.sciencesignaling.org/cgi/content/full/3/114/ra23/dc1 Supplementary Materials for Regulation of Zap70 Expression During Thymocyte Development Enables Temporal Separation of CD4 and CD8 Repertoire Selection
www.sciencesignaling.org/cgi/content/full/3/114/ra23/dc1 Supplementary Materials for Regulation of Zap70 Expression During Thymocyte Development Enables Temporal Separation of CD4 and CD8 Repertoire Selection
Supplementary Data 1. Alanine substitutions and position variants of APNCYGNIPL. Applied in
 Supplementary Data 1. Alanine substitutions and position variants of APNCYGNIPL. Applied in Supplementary Fig. 2 Substitution Sequence Position variant Sequence original APNCYGNIPL original APNCYGNIPL
Supplementary Data 1. Alanine substitutions and position variants of APNCYGNIPL. Applied in Supplementary Fig. 2 Substitution Sequence Position variant Sequence original APNCYGNIPL original APNCYGNIPL
Lecture outline. Immunological tolerance and immune regulation. Central and peripheral tolerance. Inhibitory receptors of T cells. Regulatory T cells
 1 Immunological tolerance and immune regulation Abul K. Abbas UCSF 2 Lecture outline Central and peripheral tolerance Inhibitory receptors of T cells Regulatory T cells 1 The immunological equilibrium:
1 Immunological tolerance and immune regulation Abul K. Abbas UCSF 2 Lecture outline Central and peripheral tolerance Inhibitory receptors of T cells Regulatory T cells 1 The immunological equilibrium:
SUPPLEMENTARY FIGURES
 SUPPLEMENTARY FIGURES Supplementary Figure 1: Chemokine receptor expression profiles of CCR6 + and CCR6 - CD4 + IL-17A +/ex and Treg cells. Quantitative PCR analysis of chemokine receptor transcript abundance
SUPPLEMENTARY FIGURES Supplementary Figure 1: Chemokine receptor expression profiles of CCR6 + and CCR6 - CD4 + IL-17A +/ex and Treg cells. Quantitative PCR analysis of chemokine receptor transcript abundance
Supporting Information Table of Contents
 Supporting Information Table of Contents Supporting Information Figure 1 Page 2 Supporting Information Figure 2 Page 4 Supporting Information Figure 3 Page 5 Supporting Information Figure 4 Page 6 Supporting
Supporting Information Table of Contents Supporting Information Figure 1 Page 2 Supporting Information Figure 2 Page 4 Supporting Information Figure 3 Page 5 Supporting Information Figure 4 Page 6 Supporting
Immunological memory.
 Chapter 11: Dynamics of Adaptive Immunity the answers will lie in the cytokines produced by the environment and by the T cells themselves, and in the affinity of the T-cell receptors for their antigens.
Chapter 11: Dynamics of Adaptive Immunity the answers will lie in the cytokines produced by the environment and by the T cells themselves, and in the affinity of the T-cell receptors for their antigens.
T Lymphocyte Activation and Costimulation. FOCiS. Lecture outline
 1 T Lymphocyte Activation and Costimulation Abul K. Abbas, MD UCSF FOCiS 2 Lecture outline T cell activation Costimulation, the B7:CD28 family Inhibitory receptors of T cells Targeting costimulators for
1 T Lymphocyte Activation and Costimulation Abul K. Abbas, MD UCSF FOCiS 2 Lecture outline T cell activation Costimulation, the B7:CD28 family Inhibitory receptors of T cells Targeting costimulators for
Absence of mouse 2B4 promotes NK cell mediated killing of activated CD8 + T cells, leading to prolonged viral persistence and altered pathogenesis
 Research article Absence of mouse 2B4 promotes NK cell mediated killing of activated CD8 + T cells, leading to prolonged viral persistence and altered pathogenesis Stephen N. Waggoner, 1 Ruth T. Taniguchi,
Research article Absence of mouse 2B4 promotes NK cell mediated killing of activated CD8 + T cells, leading to prolonged viral persistence and altered pathogenesis Stephen N. Waggoner, 1 Ruth T. Taniguchi,
CD44
 MR1-5-OP-RU CD24 CD24 CD44 MAIT cells 2.78 11.2 WT RORγt- GFP reporter 1 5 1 4 1 3 2.28 1 5 1 4 1 3 4.8 1.6 8.1 1 5 1 4 1 3 1 5 1 4 1 3 3.7 3.21 8.5 61.7 1 2 1 3 1 4 1 5 TCRβ 2 1 1 3 1 4 1 5 CD44 1 2 GFP
MR1-5-OP-RU CD24 CD24 CD44 MAIT cells 2.78 11.2 WT RORγt- GFP reporter 1 5 1 4 1 3 2.28 1 5 1 4 1 3 4.8 1.6 8.1 1 5 1 4 1 3 1 5 1 4 1 3 3.7 3.21 8.5 61.7 1 2 1 3 1 4 1 5 TCRβ 2 1 1 3 1 4 1 5 CD44 1 2 GFP
Supplemental Materials for. Effects of sphingosine-1-phosphate receptor 1 phosphorylation in response to. FTY720 during neuroinflammation
 Supplemental Materials for Effects of sphingosine-1-phosphate receptor 1 phosphorylation in response to FTY7 during neuroinflammation This file includes: Supplemental Table 1. EAE clinical parameters of
Supplemental Materials for Effects of sphingosine-1-phosphate receptor 1 phosphorylation in response to FTY7 during neuroinflammation This file includes: Supplemental Table 1. EAE clinical parameters of
Supporting Information
 Supporting Information Desnues et al. 10.1073/pnas.1314121111 SI Materials and Methods Mice. Toll-like receptor (TLR)8 / and TLR9 / mice were generated as described previously (1, 2). TLR9 / mice were
Supporting Information Desnues et al. 10.1073/pnas.1314121111 SI Materials and Methods Mice. Toll-like receptor (TLR)8 / and TLR9 / mice were generated as described previously (1, 2). TLR9 / mice were
Darwinian selection and Newtonian physics wrapped up in systems biology
 Darwinian selection and Newtonian physics wrapped up in systems biology Concept published in 1957* by Macfarland Burnet (1960 Nobel Laureate for the theory of induced immune tolerance, leading to solid
Darwinian selection and Newtonian physics wrapped up in systems biology Concept published in 1957* by Macfarland Burnet (1960 Nobel Laureate for the theory of induced immune tolerance, leading to solid
