During the maturation of T lymphocytes within the thymus,
|
|
|
- Flora Bishop
- 5 years ago
- Views:
Transcription
1 Extracellular Signal regulated Kinase (ERK) Activation by the Pre-T Cell Receptor in Developing Thymocytes In Vivo By Alison M. Michie,* Sébastien Trop,* David L. Wiest, and Juan Carlos Zúñiga-Pflücker* From the *Department of Immunology, University of Toronto, Toronto, Ontario M5S 1A8, Canada; the Department of Medicine, Division of Experimental Medicine, McGill University, Montréal, Québec H3A 1A3, Canada; and the Division of Basic Sciences, Immunobiology Working Group, Fox Chase Cancer Center, Philadelphia, Pennsylvania Summary The first checkpoint in T cell development occurs between the CD4 CD8 and CD4 CD8 stages and is associated with formation of the pre-t cell receptor (TCR). The signaling mechanisms that drive this progression remain largely unknown. Here, we show that extracellular signal regulated kinases (ERKs)-1/2 are activated upon engagement of the pre-tcr. Using a novel experimental system, we demonstrate that expression of the pre-tcr by developing thymocytes induces ERK-1/2 activation within the thymus. In addition, the activation of this pre-tcr signaling cascade is mediated through Lck. These findings directly link pre-tcr complex formation with specific downstream signaling components in vivo. Key words: thymus thymocyte differentiation cellular signaling fetal thymic organ culture pre-tcr During the maturation of T lymphocytes within the thymus, precursor lymphocytes undergo two key selection steps that ensure that T cells express a functional TCR CD3 complex on their cell surfaces that can recognize peptide antigens in the context of self-mhc (1). The earliest thymocytes, which migrate to the thymus from the adult bone marrow or fetal liver, lack expression of CD4 and CD8 and are thus termed CD4 CD8 double-negative (DN) 1 cells (2). The DN stage of development can be further subdivided by the ordered surface expression of CD117 (c-kit) and CD25 as follows: CD117 CD25, CD117 CD25, CD117 lo/ CD25, and CD117 CD25 (2, 3). The first developmental checkpoint in T cell differentiation, which occurs at the CD117 lo/ CD25 stage, assures that only thymocytes with a productively rearranged TCR chain proceed to the next developmental stage. This critical regulatory checkpoint is known as -selection (4 6). The TCR chain covalently associates with the nonpolymorphic pre-t (pt ) chain, which together with CD3 molecules forms the pre-tcr complex (7, 8). Expression of the pre-tcr triggers a differentiation program that includes inhibition of further V(D)J recombination at the 1 Abbreviations used in this paper: DLR, DNA loading ratio; DN, doublenegative; DP, double-positive; ERK, extracellular signal regulated kinase; FTOC, fetal thymic organ culture; LAT, linker for activation of T cells; MAPK, mitogen-activated protein kinase; RAG, recombination activating gene; RLUs, relative light units; SLP-76, SH2 domain containing leukocyte protein, 76 kd; SP, single-positive. TCR- locus (allelic exclusion), rescue from apoptosis, exponential cellular proliferation, downregulation of CD25 expression, CD4 and CD8 surface expression, and initiation of TCR- gene rearrangement (9 14). Notably, differentiation toward the CD4 CD8 double-positive (DP) stage of development occurs through an immature singlepositive (SP) stage (5, 6, 15, 16). The second regulatory checkpoint in T cell development occurs at the DP stage when thymocytes undergo positive and negative selection, resulting in the generation of mature CD4 CD8 and CD4 CD8 SP thymocytes (1). The signaling pathways that mediate -selection events are not well defined. However, the study of T cell development in mice lacking key signaling molecules has provided important clues regarding the possible signal transduction pathways essential for -selection. The pre-tcr complex seems to function together with CD3 molecules to transduce differentiation and proliferation signals (16 23). Downstream signaling events appear to be dependent on the nonreceptor tyrosine kinases Lck/Fyn and ZAP-70/Syk (15, 24 28), as well as the adapter proteins SLP-76 (SH2 domain containing leukocyte protein, 76 kd) and LAT (linker for activation of T cells) (29 31). Indeed, genetic deficiencies for pt, TCR-, CD3, CD3, Lck, SLP-76, and LAT all arrest differentiation at the CD117 lo/ CD25 stage, at the onset of -selection (16, 20, 24, 29 33). Thymocytes are also arrested at the CD117 CD25 stage of development in recombination activating gene 1 or 2 deficient (RAG / ) and SCID mice due to an inability 1647 J. Exp. Med. The Rockefeller University Press /99/12/1647/09 $5.00 Volume 190, Number 11, December 6,
2 to initiate or complete rearrangement of their antigen receptors, respectively (34 38). In particular, the introduction of productively rearranged TCR- transgenes into RAG / and SCID mice allows further differentiation of DN cells, indicating that a functional TCR chain is sufficient for progression to the DP stage in these mice (39, 40). However, RAG / mice containing constitutively active Lck (F505), Ras (Ha-Ras V12 ), and serine/threonine kinase c-raf-1 (Raf-CAAX) transgenes allow for cellular proliferation and generation of DP cells by experimentally bypassing pre-tcr dependent -selection processes (41 43). These data support the involvement of Lck, Ras, and Raf in -selection and suggest a signaling mechanism similar to that observed during T cell activation. Signaling through the mature TCR is thought to involve the following events after TCR engagement: Lck-mediated phosphorylation of the CD3 chain; recruitment of ZAP-70/Syk to the receptor complex; ZAP-70/Syk-mediated phosphorylation of downstream substrates, including LAT, SLP-76, and phospholipase C 1; linkage of the TCR CD3 complex to pathways leading to Ca 2 mobilization and Ras activation through phosphorylated LAT and SLP-76; and activation of the Ras/Raf/mitogen-activated protein kinase (MAPK)-1 (MEK1)/extracellular signal regulated kinase (ERK; p44/42 MAPK) cascade (44). Notably, results from transgenic mice carrying dominant negative forms of Ras (Ha-Ras N17 ; reference 45), Raf (46), and MEK1 (47) did not affect -selection events. Thus, studies supporting the involvement of the Ras/Raf/MEK/ERK cascade during -selection are conflicting. Furthermore, a direct link between pre-tcr expression by developing thymocytes and the activation of downstream signaling pathways has not been demonstrated. In this report, we show that ERK-1/2 is phosphorylated and activated upon engagement of the pre-tcr expressed on a SCID mouse derived pre-t cell line, indicating that the ERK signaling cascade is initiated by the pre-tcr. To study specific biochemical signaling events that ensue upon formation of the pre-tcr complex, we developed a novel experimental system that allows for the introduction of reporter plasmids into thymic lobes. This system enables signals generated by the pre-tcr to be assayed in real time within a biologically relevant setting, i.e., during thymocyte differentiation within an intact thymus. Using this system, we demonstrate that the ERK signaling cascade is activated in developing thymocytes as a physiological result of pre-tcr complex formation in vivo. Materials and Methods Animals. RAG-2 / mice were bred and maintained in our animal facility (34). Timed-pregnant RAG-2 / mice were used on day 14 of gestation. Cell Lines. The SL cell line is a pre-t cell line derived from a spontaneous SCID mouse derived thymoma that stably expresses functionally rearranged TCR chain at the cell surface with endogenous pt to form a pre-tcr (48, 49). The cell line was maintained in complete media (HG-DME media containing 10% FBS, 1% penicillin/streptomycin, 1% glutamine, 1% sodium pyruvate, 1% Hepes, 0.1% 2-ME, and 0.1% gentamicin [GIBCO BRL] supplemented with 0.5 mg/ml geneticin [GIBCO BRL]). Cells were kept in a humidified atmosphere of 5% CO 2 at 37 C. SDS-PAGE and Western Blot Analysis. SL cells ( ) were incubated at 4 C with 10 g/ml biotinylated anti TCR- mab (H57-597) for 30 min in DMEM supplemented with 0.1% BSA. Cells were then washed, resuspended in 100 l DMEM/0.1% BSA containing 25 g/ml avidin (Sigma Chemical Co.), and incubated at 37 C. Stimulation was stopped by the addition of 3 ml ice cold PBS followed by lysis in buffer containing 50 mm Tris, ph 7.4 (Boehringer Mannheim), 0.5% Triton X-100 (Boehringer Mannheim), 150 mm NaCl (ICN Biomedicals Inc.), 1 mm EDTA (BDH Chemicals Ltd.), 1 mm sodium orthovanadate (ICN Biomedicals Inc.), and Complete Inhibitor Cocktail (Boehringer Mannheim). Control cells (unstimulated) were treated similarly; however, avidin was not added before lysis. Lysates were resolved on 10% SDS-PAGE under reducing conditions and transferred onto polyvinylidene difluoride membranes (Immobilon-P; Amersham Pharmacia Biotech). Phosphorylated ERK-1 (p44 MAPK) and ERK-2 (p42 MAPK) and total ERK-2 proteins were detected using phospho-p44/22 MAPK (Thr202/Tyr204) mab (New England Biolabs Inc.) and total p42 MAPK antibody (New England Biolabs Inc.), followed by horseradish peroxidase conjugated goat anti rabbit IgG (Santa Cruz Biotechnology). Blots were revealed using Supersignal West Pico chemiluminescent substrate (Pierce Chemical Co.). Transfection of SL Cells. All electroporations were carried out using a BTX Electro Cell Manipulator 600 apparatus. SL cells were electroporated at 450 F, 186, 300 V, attaining a time constant of 30 ms cells were transfected with desired plasmid DNA (up to 40 g), as indicated in the figure legends. Each sample was transfected with PathDetect reporter plasmids (Stratagene, Inc.; as indicated in figure legends) and 1 g plasmid encoding -galactosidase (pcmv- -gal). The addition of a fixed amount of -galactosidase plasmid allowed for the control of transfection efficiency during the experiment. The -galactosidase activity was used to index the luciferase signal detected, as we were able to assay for luciferase and -galactosidase activity within the same sample (50). In brief, cells were washed in electroporation media (RPMI 1640 containing 20% FCS) and resuspended at cells/ml (250 l per transfection). DNA and cells were combined in 4-mm sterile cuvettes (Bio-Rad Labs.) and incubated on ice for 10 min. The cells were electroporated with the conditions noted above and incubated on ice for a further 10 min. Transfected cells were put into fresh complete media and incubated for 24 h at 37 C, with or without the addition of exogenous stimuli, as indicated in the figure legends. The cells were then lysed and analyzed for luciferase and -galactosidase activity. Gene Gun Transfection and Fetal Thymic Organ Culture. Fetal thymic lobes from timed-pregnant RAG-2 / mice at day 14 of gestation were placed on nucleopore filters resting on Gelfoam rafts (Upjohn) soaked in fetal thymic organ culture (FTOC) media (complete media supplemented with a further 5% FCS; five to six lobes per filter) (51). The thymic lobes were incubated at 37 C for 6 h in a humidified incubator with 5% CO 2 to allow them to adhere to the filters. After this time, the filters were briefly removed from the Gelfoam rafts, and thymi were immediately transfected by gene gun bombardment with a Helios gene gun set at 200 psi (Bio-Rad Labs.), with the desired DNA bound to gold particles (1 g gold) (as indicated in the figure legends; one cartridge per set of thymic lobes). The amount of DNA loaded per microgram of microcarriers 1648 Pre-TCR derived Signals Activate ERK In Vivo
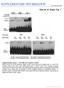
3 is referred to as the DNA loading ratio (DLR; see figure legends). The filters carrying the transfected thymic lobes were then placed back on the Gelfoam rafts and incubated at 37 C for the indicated time (see figure legends). For short-term biochemical readout assays, the thymic lobes were incubated for a further h, and then a single-cell suspension was prepared and the cells were lysed and assayed for luciferase and -galactosidase activities as described below. For long-term developmental progression studies, the transfected thymic lobes were incubated for 7 d, with the FTOC media being changed once during the incubation. After this time, a single-cell suspension of the lobes was prepared, and the thymocytes were analyzed by flow cytometry as described below. Flow Cytometry. FITC-, PE, and APC-conjugated anti mouse CD4, CD25, and CD8 were used for flow cytometric analysis (purchased from PharMingen). Staining of cells was carried out as described previously (51). In brief, thymic cell suspensions were washed in FACS buffer (HBSS containing 1% BSA and 0.1% sodium azide) and incubated with antibodies (1:300 dilution) for 30 min on ice. The cells were then washed in FACS buffer before analysis. Stained cells were analyzed with a FACS- Calibur flow cytometer (Becton Dickinson). Analysis was performed using CELLQuest software (Becton Dickinson); the data were live gated by size and lack of propidium iodide uptake. Luciferase and -galactosidase Assay. Cells transfected with the PathDetect reporter plasmids (Stratagene Inc.) were assayed for luciferase and -galactosidase activities using the Dual-Light reporter gene assay system (Tropix; Perkin Elmer-Applied Biosystems) as previously described (50). In brief, the cells were lysed in lysis buffer (40 mm tricine, ph 7.8, 50 mm NaCl, 2 mm EDTA, 1 mm MgSO 4, 5 mm dithiothreitol, and 1% Triton X-100). Supernatant (25 l) was combined with an equal volume of luciferase reaction buffer (30 mm tricine, ph 7.8, 3 mm ATP, 15 mm MgSO 4, 1 mm coenzyme A, and 10 mm dithiothreitol), and after addition of 1 mm luciferin, the sample was immediately assayed for luciferase activity with a Lumat LB 9507 Luminometer (Fisher Scientific Co.). To assay for -galactosidase activity, Galacton-Plus (substrate for -galactosidase; Tropix) was added to each tube after the luciferase assay was completed. The tubes were incubated for min at room temperature, and then Accelerator-II (Tropix) was added to each tube and the samples were immediately assayed for -galactosidase activity, measured as light emission with the Lumat LB 9507 Luminometer. Results represent the average luciferase activity indexed for -galactosidase activity. Results Pre-TCR Complex Engagement Induces ERK-1/2 Phosphorylation. To aid in deciphering the signaling pathways that lead to the differentiative and proliferative events of -selection, the phosphorylation status of ERK-1/2 was investigated by performing Western blot analyses using an antiphospho-erk-1/2 mab. A SCID mouse derived pre-t cell line, SL , preincubated with biotinylated anti TCR- (H57-597) mab, was stimulated for 1 30 min upon cross-linking with avidin. Engagement of the pre-tcr complex resulted in a rapid and transient phosphorylation of ERK-1/2 (Fig. 1 A). Phosphorylation was observed as early as 1 min but declined almost to background levels 5 min after stimulation; subsequently, a secondary, weaker level of phosphorylation was evident between 10 and 30 min (Fig. 1 A). To control for protein loading, the total amount Figure 1. ERK-1/2 is phosphorylated upon cross-linking of the pre- TCR in the SL pre-t cell line. Lysates from SL cells stimulated with 10 g/ml biotinylated anti TCR- mab H and cross-linked with avidin (lanes 1, 2, 5, 10, and 30) or unstimulated cells (lane 0) were prepared and resolved on a 10% SDS-PAGE under reducing conditions and subsequently immunoblotted with specific antibodies against (A) phospho-erk-1/2 and (B) ERK-2. of ERK-2 was assessed by Western blot with an anti ERK-2 antibody (Fig. 1 B). To study whether the decline in phosphorylation of ERK-1/2 at 5 min was specific to pre-tcr activation, we performed a similar experiment with variants of the SL12 cell line expressing comparable levels of TCR chain with either a TCR chain (mature TCR) or a pt chain (pre- TCR), as determined by flow cytometry (data not shown). The TCR chain on both cell lines was cross-linked for 1 30 min, and the phosphorylation status of ERK-1/2 was determined. In both cases, there was an attenuation of ERK-1/2 phosphorylation levels at 5 min, indicating that this phosphorylation pattern is characteristic of the SL-12 cell line (data not shown). These results indicate that engagement of the pre-tcr on SL cells leads to the phosphorylation of ERK-1/2, demonstrating the activation of the Ras/Raf/MEK/ERK signaling cascade. Reporter Plasmid Detection of ERK-1/2 Activation after Pre- TCR Engagement. We sought to determine if the phosphorylation of ERK-1/2 observed upon cross-linking of the pre-tcr (Fig. 1 A) leads to their activation. To this end, we employed a reporter plasmid system (described in Materials and Methods) that allows detection of ERK-1/2 activation within the cell. This system utilizes two plasmids: a fusion activator plasmid (pfa-elk), which encodes for the transactivation domain of the ERK substrate, Elk-1 (residues ), fused with the DNA-binding domain of GAL4; and a luciferase reporter plasmid (pfr-luc), which encodes for the luciferase gene under the control of five GAL4 binding elements. Hence, in transfected cells, phosphorylation of the Elk-1 fusion protein can be read in the form of luciferase activity. Accordingly, we transfected SL cells with pfr-luc, either alone or together with pfa-elk. The transfected cells were then stimulated with immobilized anti-tcr- mab or with PMA and ionomycin. Cells transfected with pfr-luc alone displayed background luciferase activity (31 18 relative light units [RLUs]). However, luciferase activity increased almost 850-fold above background levels in cells transfected with pfr-luc and pfa-elk (26,221 1,119 RLUs; Fig. 2). This elevation in luciferase activity may reflect low-level 1649 Michie et al.
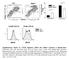
4 Figure 2. Activation of the ERK signaling cascade upon stimulation of SL cells with anti TCR- mab (H57-597; A) or PMA/ionomycin (B). SL cells were transfected with reporter plasmids as indicated in the figure: pfr-luc (10 g), pfa-elk (20 g), and pfc-mek1 (20 g). All cells were transfected with a control -galactosidase encoding plasmid (1 g). The transfected cells were incubated overnight at 37 C and then stimulated for 8 h with (A) immobilized anti TCR- or (B) phorbol ester, PMA, and the calcium ionophore, ionomycin (10 ng/ml of each). Cells were then lysed, and the lysates were assayed for luciferase and -galactosidase activity. The experiment shown is representative of results carried out three times. Each experiment was carried out in triplicate, and the luciferase activities shown are indexed against -galactosidase activity measured within each same sample. constitutive ERK activity within SL cells (Fig. 1 A), as it was blocked by the addition of the MEK1 inhibitor, PD98059 (data not shown). Importantly, anti TCR- mab stimulation of pfr-luc/pfa-elk transfected SL cells resulted in a fivefold increase in luciferase activity compared with unstimulated cells (Fig. 2 A). This elevation in luciferase activity was abrogated by the addition of PD98059 (129% inhibition). These data indicate that engagement of the pre-tcr complex results in the activation of ERK and subsequent phosphorylation of the ERK-1/2 substrate, Elk-1 (Fig. 2 A). Activation of pfr-luc/pfa-elk transfected cells by the addition of PMA/ionomycin induced a 20-fold stimulation in luciferase activity over unstimulated cells (Fig. 2 B); again, addition of PD98059 curtailed this activity (data not shown). As a control to demonstrate that ERK-1/2 activation can be efficiently detected with these reporter plasmids, pfr-luc/pfa-elk transfected cells were also transfected with a plasmid encoding a constitutively active MEK1, pfc-mek1. This serine/threonine kinase is responsible for the phosphorylation and activation of ERK-1/2. These cells showed maximal luciferase activity irrespective of PMA/ ionomycin treatment (Fig. 2 B), indicating that this reporter system provides a highly sensitive method for detecting ERK activation. Taken together, these results confirm that the ERK signaling cascade is activated upon engagement of the pre- TCR, as shown by the phosphorylation of ERK-1/2 (Fig. 1 A) and the phosphorylation/activation of the ERK-1/2 substrate, Elk-1 (Fig. 2 A), in the SL cell line. Generation of DP Thymocytes in RAG-2 / FTOCs Transfected with TCR- and Active p56 lck Genes. An important application of the above reporter plasmid system would be to detect specific pre-tcr mediated signals as they occur within an intact thymus. Therefore, an accelerated DNA/ particle bombardment delivery system (gene gun) was adapted to transfect thymocytes in FTOC (52). This approach allows transfected cells to respond to normal microenvironmental stimuli encountered during differentiation within the thymic milieu (52, 53). We have previously used this method to study transcriptional regulation events in the developing fetal thymus (52). The preservation of structural and functional integrity of gene gun transfected thymi was tested by allowing FTOCs to complete a program of -selection. Thus, RAG-2 / mouse derived FTOCs were gene gun transfected with control DNA and cultured for 7 d, either alone (Fig. 3 A, Control) or in the presence of anti-cd3 mab (Fig. 3 A, anti-cd3). Transfected FTOCs were then analyzed by flow cytometry for evidence of differentiation to the DP stage (17 19). This analysis revealed that the thymocytes from anti-cd3 treated RAG-2 / gene gun transfected FTOCs progressed to the DP stage, as indicated by upregulation of CD8/CD4 (Fig. 3 A, anti-cd3). This differentiative event involved the loss of CD25 cell surface expression and proceeded through a CD8 immature SP stage (Fig. 3 A, anti- CD3). These results indicated that thymocytes and stromal elements within gene gun transfected thymic lobes remain viable. To determine the long-term developmental capacity of transfected thymocytes, RAG-2 / thymic lobes were gene gun transfected with plasmids encoding either constitutively active Lck (Lck[F505]) or a productively rearranged TCR chain. It has been previously shown in RAG / 1650 Pre-TCR derived Signals Activate ERK In Vivo
5 Figure 3. Generation of CD4 CD8 DP cells in RAG-2 / FTOCs gene gun transfected with plasmid encoding a constitutively active Lck or a functional TCR chain. Intact fetal thymic lobes removed from timedpregnant RAG-2 / mice (day 14 of gestation) were used as targets for DNA-covered gold particles as described in Materials and Methods. Fetal thymi were gene gun transfected with (A) control DNA and incubated in the absence (Control) or presence (anti-cd3) of 10 g/ml anti-cd3 mab (145-2C11) or (B) plasmid encoding a constitutively active Lck (DLR, 750 ng) or plasmid encoding a functionally rearranged TCR chain (DLR, 250 ng). The transfected FTOCs were cultured for 7 d. After this time, a single-cell suspension was prepared and thymocytes were analyzed for surface expression of CD25, CD8, and CD4 by flow cytometry. mice that introduction of transgenes encoding a kinaseactive Lck or a functionally rearranged TCR chain induced thymocyte proliferation and differentiation toward the DP stage of development (39, 41). Indeed, these events were also observed in gene gun transfected RAG-2 / FTOCs (Fig. 3 B). Flow cytometric analysis of Lck(F505)- and TCR- transfected FTOCs after 7 d revealed that thymocytes differentiated from the DN stage to the DP stage, as evidenced by upregulation of CD4 and CD8 surface expression and downregulation of CD25 surface expression (Fig. 3 B). The apparent difference in the levels of CD8 surface expression in active Lck-transfected FTOCs was due to the fact that the experiment shown was analyzed separately and a different set of fluorochrome-labeled antibodies was used. These phenotypic changes are consistent with differentiation events that occur after completion of -selection (Fig. 3 B; reference 5, 10 13). Our finding that gene gun transfection of RAG-2 / FTOCs with a functional TCR chain resulted in the normal developmental progression of DN thymocytes indicated that a functional pre-tcr complex was generated (39). These data imply that after gene gun transfection, the formation of the pre-tcr results in signaling events that occur during normal -selection in vivo. ERK Signaling Cascade Is Activated upon Formation of the Pre-TCR in the Thymus. Because the cellular signaling processes that underlie -selection are not well defined, we combined gene gun mediated transfection of FTOCs with the reporter plasmid system. In this way, the intracellular signaling cascades that occur after pre-tcr formation can be monitored in a biological time frame and within a relevant in vivo setting. We hypothesized that gene gun mediated transfection of RAG-2 / mouse derived FTOCs with a plasmid encoding a functionally rearranged TCR chain, together with reporter plasmids, would allow us to capture de novo -selection signaling events that occur after the generation of the pre-tcr. Our results show that compared with FTOCs transfected with pfr-luc alone (Fig. 4), there was a slight elevation in luciferase activity in FTOCs transfected with pfr-luc/ pfa-elk (6 1.8 vs RLUs; Fig. 4). This level of luciferase activity may represent endogenous ERK-1/2 activity present in transfected FTOCs. To directly test our hypothesis, RAG-2 / FTOCs were gene gun transfected with reporter plasmids (pfr-luc/ pfa-elk) together with a plasmid encoding a functional TCR chain. Our analysis revealed an almost sixfold increase in luciferase activity in FTOCs transfected with a TCR- plasmid as compared with FTOCs transfected with the reporter plasmids alone ( RLUs vs ; Fig. 4, TCR- ). A substantial luciferase activity was also observed in FTOCs transfected with a constitutively active MEK1 plasmid together with the reporter plasmids (9,919 1,933 RLUs; Fig. 4, MEK1). The level of stimulation observed in MEK1-transfected FTOCs demonstrates the sensitive nature of this assay. Several reports have suggested that the Ras/Raf/MEK/ ERK signaling cascade is involved at the -selection stage (13, 42, 43, 54). However, our data provides the first direct evidence for coupling of the pre-tcr complex to the ERK signaling cascade in vivo. Activation of the ERK Cascade Requires Lck. Lck is thought to play an essential role as a mediator of -selection events (15, 17, 24, 41, 55). Therefore, we investigated whether Lck could activate the ERK signaling cascade in FTOCs. To this end, FTOCs were gene gun transfected with a kinase-active Lck together with the reporter plasmids. Fig. 5 shows that a sixfold increase in luciferase activity was observed in FTOCs transfected with constitutively active Lck 1651 Michie et al.
6 Figure 4. Activation of the ERK signaling cascade in fetal thymic lobes gene gun transfected with a plasmid encoding a functional TCR chain. Intact fetal thymic lobes removed from timed-pregnant RAG / mice (day 14 of gestation) were used as targets for DNA-covered gold particles as described in Materials and Methods. Fetal thymi were gene gun transfected with pfr- Luc (DLR, 250 ng) and CMV- -gal (DLR, 250 ng) and either pfa-elk (DLR, 250 ng) alone or together with TCR- (DLR, 250 ng) or constitutively active MEK-1 (DLR, 250 ng), as indicated. The transfected lobes were then cultured for h. Cells were then lysed, and the lysates were assayed for luciferase and -galactosidase activity. The data shown is an average of at least five independent experiments. The individual luciferase activities were indexed against a constant -galactosidase activity. ( RLUs; Fig. 5). This level of luciferase activity is comparable to the stimulation observed in TCR- transfected FTOCs (Fig. 5, TCR- ). To address whether the observed ERK activation after the pre-tcr complex formation was dependent on Lck activity, we took advantage of a plasmid encoding a dominant negative Lck (Lck[R273]). This mutant form of Lck has been shown to cause a dramatic block in T cell development at the DN stage (15). Fig. 5 shows that transfection of RAG-2 / FTOCs with the reporter plasmids together with plasmids encoding a TCR chain and a kinase-negative Lck led to an attenuation of luciferase activity, as compared with FTOCs transfected with TCR- alone (84 56 vs RLUs; Fig. 5). These results indicate that expression of the pre-tcr complex by developing thymocytes activates the ERK signaling cascade and that this activation is directly dependent on Lck during thymocyte differentiation within the thymus. cues and undergo rapid programmed cell death (56, 57). Indeed, transfection of thymocytes from RAG / mice with reporter plasmids revealed that cells in suspension fail to respond when cotransfected with a plasmid encoding a TCR chain (data not shown). Thus, thymocytes in cell suspension appear to be unresponsive to pre-tcr mediated signals, possibly due to the lack of stromal elements to assist in this signaling. These results are consistent with previous data showing that anti-cd3 mab mediated differentiation of RAG / thymocytes to the DP stage in FTOC does not occur when these thymocytes are treated in cell suspension (58). Discussion In this study, we demonstrate that formation and/or engagement of the pre-tcr complex results in the activation of the ERK signaling cascade. We have shown this in two distinct systems: (a) in a SCID mouse derived pre-t cell line, SL (48), in which engagement of the pre-tcr leads to phosphorylation and activation of ERK-1/2; and (b) within the thymus, where formation of the pre-tcr in developing thymocytes induces ERK activation and phosphorylation of the downstream substrate, Elk-1. Furthermore, our findings confirm the central role of the nonreceptor tyrosine kinase Lck in -selection by demonstrating that ERK activation is strictly dependent on Lck function in vivo. The ability to introduce reporter plasmids into developing thymocytes in vivo provides an important diagnostic tool for the detection of signaling events during T cell development in the thymus (Figs. 3 and 4). The significance of this method is highlighted by the fact that, in the absence of the thymic microenvironment (i.e., cell suspension), thymocytes fail to respond to normal developmental Figure 5. Activation of ERK signaling cascade by the pre-tcr is dependent on Lck function. Intact fetal thymic lobes removed from timedpregnant RAG / mice (day 14 of gestation) were used as targets for DNA-covered gold particles as described in Materials and Methods. Fetal thymi were gene gun transfected with pfr-luc (DLR, 250 ng) and CMV- -gal (DLR, 250 ng) and either pfa-elk (DLR, 250 ng) alone or together with constitutively active Lck (Lck[F505]; DLR, 750 ng), TCR- (DLR, 250 ng), or TCR- (DLR, 250 ng) plus dominant negative Lck (Lck[R273]; DLR, 750 ng), as indicated. The transfected lobes were then cultured for h. Cells were then lysed, and the lysates were assayed for luciferase and -galactosidase activity. The data shown is an average of at least four independent experiments. The individual luciferase activities were indexed against a constant -galactosidase activity Pre-TCR derived Signals Activate ERK In Vivo
7 The notion that the Ras/Raf/MEK/ERK signaling cascade plays a central role during -selection is supported by data obtained from mice bearing targeted genetic deficiencies of key signaling molecules such as Lck/Fyn, ZAP-70/ Syk, LAT, or SLP-76 (24 26, 28 31). These mice displayed a block in T cell development at the DN stage, suggesting that these molecules are essential for differentiation to the DP stage of development (24 26, 28 31). However, several contradicting reports using transgenic mice carrying either constitutively active or dominant negative members of these signaling molecules have left the involvement of this pathway unresolved. Studies introducing constitutively active Lck, Ras, and Raf into RAG / mice also indicate that the Ras/Raf pathway is crucial at this stage of development (41 43). In contrast, studies using transgenic mice carrying dominant negative forms of Ras (Ha-Ras N17 ) (45), Raf (46), and MEK-1 (47) failed to support a role for these signaling molecules during -selection. In this regard, it is important to note that mice carrying a dominant negative Lck transgene displayed varying degrees of DN to DP thymocyte development arrest directly correlating with the levels of transgene expression (15). Thus, the conflicting results obtained using dominant negative molecules may reflect their ability to be sufficiently expressed at the appropriate stage of development. In addition, studies using retroviral infection of thymocytes with dominant negative MEK-1 showed a perturbation in the transition of thymocytes from the DN to DP stage, suggesting that the inhibitory effectiveness of dominant negative molecules appears to be dependent on the experimental system used (54). Although several studies have indirectly supported the importance of the Ras/Raf/MEK/ERK-mediated signals downstream of the pre-tcr, in our system, the fact that the detection of ERK activation is predicated by the expression of the pre-tcr within individually transfected developing thymocytes directly links the pre-tcr to the activation of these kinases. Thus, this study provides the first evidence for the pre-tcr being responsible for the generation of specific downstream signals within the thymus (Fig. 4) and shows that Lck acts proximally to the nascent pre-tcr complex, leading to the subsequent activation of ERK (Fig. 5). The transcription factor Elk-1 has been shown to be phosphorylated by ERK-1/2, although more recently it has been demonstrated that JNK (c-jun NH 2 -terminal kinase) also has the ability to phosphorylate and activate Elk-1 (59 61). However, mouse models with gene-targeted disruptions of key signaling components of the JNK signaling pathway (JNK2 and stress-activated protein kinase kinase 1) did not reveal any abnormalities in -selection events (62 64). In this regard, our data with the SCID mouse derived pre-t cell line SL show that engagement of the pre-tcr results in ERK phosphorylation and activation of the ERK substrate, Elk-1 (Figs. 1 and 2), and that this can be blocked by addition of the MEK1 inhibitor, PD Together, these data indicate that, in this setting, Elk-1 is specifically activated by ERK. Pre-TCR signals can be experimentally bypassed by introducing constitutively active mutants of certain signaling components, inducing -selection mediated events. Interestingly, the introduction of an activated form of Ras (Ha-Ras V12 ) or Raf-1 (Raf-CAAX), although allowing the differentiation of RAG / thymocytes to the DP stage, failed to establish allelic exclusion at the TCR- locus when introduced into wild-type mice (43, 65). This indicates that although certain -selection outcomes are mediated by the Ras/Raf pathway, distinct pre-tcr derived signals upstream of Raf are responsible for the control of allelic exclusion. Therefore, it is interesting to note that RAG / FTOCs retrovirally infected with constitutively active MEK1 do not differentiate to the DP stage of development (54). This finding suggests that Raf may activate additional signaling pathways enabling developing thymocytes to undergo full differentiation to the DP stage. Therefore, the intrathymic signal detection system developed here will allow for detailed studies of distal signaling events activated by the pre-tcr, facilitating the elucidation of potential signaling branchpoints. We would like to thank Drs. Pamela Ohashi, Michael Julius, and James Carlyle for their critical review of the manuscript, P. Ohashi and Sanjeev Mariathasan for the gift of antibodies and advice with Western blotting, André Veillette for his kind gift of the plasmids encoding constitutively active and dominant negative Lck, and Michael Lenardo for his gift of the plasmid encoding a functionally rearranged TCR chain. A.M. Michie is supported by a postdoctoral fellowship from the Cancer Research Institute. S. Trop is supported by a Doctoral Research Award from the Medical Research Council of Canada (MRC). J.C. Zúñiga- Pflücker is supported by a scholarship from the MRC. This work was funded by grants from the Leukemia Research Fund of Canada and the MRC. Address correspondence to J.C. Zúñiga-Pflücker, Dept. of Immunology, University of Toronto, Medical Sciences Bldg., Toronto, Ontario M5S 1A8, Canada. Phone: ; Fax: ; jc.zuniga.pflucker@utoronto.ca Submitted: 18 August 1999 Revised: 23 September 1999 Accepted: 29 September Michie et al.
8 References 1. Sebzda, E., S. Mariathasan, T. Ohteki, R. Jones, M.F. Bachmann, and P.S. Ohashi Selection of the T cell repertoire. Annu. Rev. Immunol. 17: Shortman, K., and L. Wu Early T lymphocyte progenitors. Annu. Rev. Immunol. 14: Zúñiga-Pflücker, J.C., and M.J. Lenardo Regulation of thymocyte development from immature progenitors. Curr. Opin. Immunol. 8: Dudley, E.C., H.T. Petrie, L.M. Shah, M.J. Owen, and A.C. Hayday T cell receptor chain gene rearrangement and selection during thymocyte development in adult mice. Immunity. 1: von Boehmer, H., I. Aifantis, J. Feinberg, O. Lechner, C. Saint-Ruf, U. Walter, J. Buer, and O. Azogui Pleiotropic changes controlled by the pre-t-cell receptor. Curr. Opin. Immunol. 11: Levelt, C.N., and K. Eichmann Receptors and signals in early thymic selection. Immunity. 3: Saint-Ruf, C., K. Ungewiss, M. Groettrup, L. Bruno, H.J. Fehling, and H. von Boehmer Analysis and expression of a cloned pre-t cell receptor gene. Science. 266: Groettrup, M., and H. von Boehmer Role for a pre-t cell receptor in T-cell development. Immunol. Today. 14: Falk, I., J. Biro, H. Kohler, and K. Eichmann Proliferation kinetics associated with T cell receptor- chain selection of fetal murine thymocytes. J. Exp. Med. 184: Hoffman, E.S., L. Passoni, T. Crompton, T.M.J. Leu, D.G. Schatz, A. Koff, M.J. Owen, and A.C. Hayday Productive T-cell receptor -chain gene rearrangement: coincident regulation of cell cycle and clonality during development in vivo. Genes Dev. 10: Koyasu, S., L.K. Clayton, A. Lerner, H. Heiken, A. Parkes, and E.L. Reinherz Pre-TCR signalling components trigger transcriptional activation of a rearranged TCR gene locus and silencing of the pre-tcr locus: implications for intrathymic differentiation. Int. Immunol. 9: Fehling, H.J., and H. von Boehmer Early T cell development in the thymus of normal and genetically altered mice. Curr. Opin. Immunol. 9: Anderson, S.J., and R.M. Perlmutter A signalling pathway governing early thymocyte maturation. Immunol. Today. 16: Wilson, A., W. Held, and H.R. MacDonald Two waves of recombinase gene expression in developing thymocytes. J. Exp. Med. 179: Levin, S.D., S.J. Anderson, K.A. Forbush, and R.M. Perlmutter A dominant-negative transgene defines a role for p56 lck in thymopoiesis. EMBO (Eur. Mol. Biol. Organ.) J. 12: Malissen, M., A. Gillet, L. Ardouin, G. Bouvier, J. Trucy, P. Ferrier, E. Vivier, and B. Malissen Altered T cell development in mice with a targeted mutation of the CD3- gene. EMBO (Eur. Mol. Biol. Organ.) J. 14: Levelt, C.N., P. Mombaerts, B. Wang, H. Kohler, S. Tonegawa, K. Eichmann, and C. Terhorst Regulation of thymocyte development through CD3: functional dissociation between p56 lck and CD3 in early thymic selection. Immunity. 3: Levelt, C.N., P. Mombaerts, A. Iglesias, S. Tonegawa, and K. Eichmann Restoration of early thymocyte differentiation in T cell receptor -chain-deficient mutant mice by transmembrane signalling through CD3. Proc. Natl. Acad. Sci. USA. 90: Levelt, C.N., A. Ehrfeld, and K. Eichmann Regulation of thymocyte development through CD3. I. Timepoint of ligation of CD3 determines clonal deletion or induction of developmental program. J. Exp. Med. 177: Haks, M.C., P. Krimpenfort, J. Borst, and A.M. Kruisbeek The TCR chain is essential for development of both the TCR and TCR lineages. EMBO (Eur. Mol. Biol. Organ.) J. 17: Malissen, M., A. Gillet, B. Rocha, J. Trucy, E. Vivier, C. Boyer, F. Kontgen, N. Brun, G. Mazza, E. Spanopoulou, et al T cell development in mice lacking the CD3 / gene. EMBO (Eur. Mol. Biol. Organ.) J. 12: Ardouin, L., J. Ismaili, B. Malissen, and M. Malissen The CD3- and CD3- modules are each essential for allelic exclusion at the T cell receptor locus but are both dispensable for the initiation of V to (D)J recombination at the T cell receptor-, -, and - loci. J. Exp. Med. 187: Malissen, B., L. Ardouin, S. Lin, A. Gillet, and M. Malissen Function of the CD3 subunits of the pre-tcr and TCR complexes during T cell development. Adv. Immunol. 72: Molina, T.J., K. Kishihara, D.P. Siderovski, W. van Ewijk, A.W. Navendran, E. Timms, A. Wakeham, C.J. Paige, K.U. Hartmann, A. Veillette, et al Profound block in thymocyte development in mice lacking p56 lck. Nature. 357: Groves, T., P. Smiley, M.P. Cooke, K. Forbush, R.M. Perlmutter, and C.J. Guidos Fyn can partially substitute for lck in T lymphocyte development. Immunity. 5: van Oers, N.S.C., B. Lowin-Kropf, D. Finlay, K. Connolly, and A. Weiss T cell development is abolished in mice lacking both lck and fyn protein tyrosine kinases. Immunity. 5: Sugawara, T., V. Di Bartolo, T. Miyazaki, H. Nakauchi, O. Acuto, and Y. Takahama An improved retroviral gene transfer technique demonstrates inhibition of CD4 CD8 thymocyte development by kinase-inactive ZAP-70. J. Immunol. 161: Cheng, A.M., I. Negishi, S.J. Anderson, A.C. Chan, J. Bolen, D.Y. Loh, and T. Pawson The syk and ZAP-70 SH2- containing tyrosine kinases are implicated in pre-t cell receptor signalling. Proc. Natl. Acad. Sci. USA. 94: Clements, J.L., B. Yang, S.E. Ross-Barta, R.F. Eliason, R.A. Williamson, and G.A. Koretzky Requirement for the leukocyte-specific adapter protein SLP-76 for normal T cell development. Science. 281: Piviouk, V., E. Tsitsikov, P. Swinton, G. Rathbun, F.W. Alt, and R.S. Geha Impaired viability and profound block in thymocyte development in mice lacking the adapter protein SLP-76. Cell. 94: Zhang, W., C.L. Sommers, D.N. Burshtyn, C.C. Stebbins, J.B. DeJarnette, R.P. Trible, A. Grinberg, H.C. Tsay, H.M. Jacobs, C.M. Kessler, et al Essential role of LAT in T cell development. Immunity. 10: Fehling, H.J., A. Krotkova, C. Saint-Ruf, and H. von Boehmer Crucial role of the pre-t-cell receptor gene in development of but not T cells. Nature. 375: Mombaerts, P., A.R. Clarke, M.A. Rudnicki, J. Iacomini, S. Itohara, J.J. Lafaille, L. Wang, Y. Ichikawa, R. Jaenisch, M.L. Hooper, et al Mutations in the T-cell antigen receptor genes and block thymocyte development at different 1654 Pre-TCR derived Signals Activate ERK In Vivo
9 stages. Nature. 360: Shinkai, Y., G. Rathbun, K.-P. Lam, E.M. Oltz, V. Stewart, M. Mendelsohn, J. Charron, M. Datta, F. Young, A.M. Stall, et al RAG-2-deficient mice lack mature lymphocytes owing to inability to initiate V(D)J rearrangement. Cell. 68: Mombaerts, P., J. Iacomini, R.S. Johnson, K. Herrup, S. Tonegawa, and V.E. Papaioannou RAG-1-deficient mice have no mature B and T lymphocytes. Cell. 68: Pénit, C., B. Lucas, and F. Vasseur Cell expansion and growth arrest phases during the transition from precursor (CD4 8 ) to immature (CD4 8 ) thymocytes in normal and genetically modified mice. J. Immunol. 154: Rothenberg, E.V., D. Chen, and R.A. Diamond Functional and phenotypic analysis of thymocytes in SCID mice. Evidence for functional response transitions before and after the SCID arrest. J. Immunol. 151: Diamond, R.A., S.B. Ward, K. Owada-Makabe, H. Wang, and E.V. Rothenberg Different developmental arrest points in RAG-2 / and SCID thymocytes on two genetic backgrounds. Developmental choices and cell death mechanisms before TCR gene rearrangement. J. Immunol. 158: Shinkai, Y., S. Koyasu, K.-I. Nakayama, K.M. Murphy, D.Y. Loh, E.L. Reinherz, and F.W. Alt Restoration of T cell development in RAG-2-deficient mice by functional TCR transgenes. Science. 259: Kishi, H., P. Borgulya, B. Scott, K. Karjalainen, A. Traunecker, J. Kaufman, and H. von Boehmer Surface expression of the beta T cell receptor (TCR) chain in the absence of other TCR or CD3 proteins on immature T cells. EMBO (Eur. Mol. Biol. Organ.) J. 10: Mombaerts, P., S.J. Anderson, R.M. Perlmutter, T.W. Mak, and S. Tonegawa An activated lck transgene promotes thymocyte development in RAG-1 mutant mice. Immunity. 1: Swat, W., Y. Shinkai, H.L. Cheng, L. Davidson, and F.W. Alt Activated ras signals differentiation and expansion of CD4 CD8 thymocytes. Proc. Natl. Acad. Sci. USA. 93: Iritani, B.M., J. Alberola-Ila, K.A. Forbush, and R.M. Perlmutter Distinct signals mediate maturation and allelic exclusion in lymphocyte progenitors. Immunity. 10: van Leeuwen, J.E.M., and L.E. Samelson T cell antigen-receptor signal transduction. Curr. Opin. Immunol. 11: Swan, K.A., J. Alberola-Ila, J.A. Gross, M.W. Appleby, K.A. Forbush, J.F. Thomas, and R.M. Perlmutter Involvement of p21ras distinguishes positive and negative selection in thymocytes. EMBO (Eur. Mol. Biol. Organ.) J. 14: O Shea, C.C., T. Crompton, I.R. Rosewell, A.C. Hayday, and M.J. Owen Raf regulates positive selection. Eur. J. Immunol. 26: Alberola-Ila, J., K.A. Forbush, R. Seger, E.G. Krebs, and R.M. Perlmutter Selective requirement for MAP kinase activation in thymocyte differentiation. Nature. 373: Berger, M.A., V. Dave, M.R. Rhodes, G.C. Bosma, M.J. Bosma, D.J. Kappes, and D.L. Wiest Subunit composition of pre-t cell receptor complexes expressed by primary thymocytes: CD3- is physically associated but not functionally required. J. Exp. Med. 186: Trop, S., A.-M. Steff, F. Denis, D.L. Wiest, and P. Hugo The connecting peptide domain of pt dictates weak association of the pre-t cell receptor with the TCR- subunit. Eur. J. Immunol. 29: Michie, A.M., and J.C. Zúñiga-Pflücker Transfection and transcription of genes in developing thymocytes. In Methods in Molecular Biology: Protocols in T Cell Development and Activation. Vol. 52. K.P. Kearse, editor. Humana Press Inc., Totawa, NJ. 34: Michie, A.M., J.R. Carlyle, and J.C. Zúñiga-Pflücker Early intrathymic precursor cells acquire a CD4(low) phenotype. J. Immunol. 160: Zúñiga-Pflücker, J.C., H.L. Schwartz, and M.J. Lenardo Gene transcription in differentiating immature T cell receptor neg thymocytes resembles antigen-activated mature T cells. J. Exp. Med. 178: Jenkinson, E.J., and J.J.T. Owen T-cell differentiation in thymus organ cultures. Semin. Immunol. 2: Crompton, T., K.C. Gilmour, and M.J. Owen The MAP kinase pathway controls differentiation from doublenegative to double-positive thymocyte. Cell. 86: Fehling, H.J., B.M. Iritani, A. Krotkova, K.A. Forbush, C. Laplace, R.M. Perlmutter, and H. von Boehmer Restoration of thymopoiesis in pt / mice by anti-cd3 antibody treatment or with transgenes encoding activated lck or tailless pt. Immunity. 6: Fisher, G.H., M.J. Lenardo, and J.C. Zúñiga-Pflücker Synergy between T cell receptor and Fas (CD95/APO-1) signaling in mouse thymocyte death. Cell. Immunol. 169: Anderson, G., and E.J. Jenkinson Use of explant technology in the study of in vitro immune responses. J. Immunol. Methods. 216: Levelt, C.N., and K. Eichmann Parallel development of the T cell and its receptor: immature forms of the CD3 complex control early thymocyte maturation. The Immunologist. 1/5: Cavigelli, M., F. Dolfi, F.X. Claret, and M. Karin Induction of c-fos expression through JNK-mediated TCF/ Elk-1 phosphorylation. EMBO (Eur. Mol. Biol. Organ.) J. 14: Vanhoutte, P., J.V. Barnier, B. Guibert, C. Pages, M.J. Besson, R.A. Hipskind, and J. Caboche Glutamate induces phosphorylation of Elk-1 and CREB, along with c-fos activation, via an extracellular signal-regulated kinase-dependent pathway in brain slices. Mol. Cell. Biol. 19: Whitmarsh, A.J., S.H. Yang, M.S. Su, A.D. Sharrocks, and R.J. Davis Role of p38 and JNK mitogen-activated protein kinases in the activation of ternary complex factors. Mol. Cell. Biol. 17: Alberola-Ila, J., S.D. Levin, G. Barton, K. Forbush, L.I. Zon, and R.M. Perlmutter Analysis of the role of MKK-4/ Sek-1 in T cell development and apoptosis. Int. Immunol. 10: Sabapathy, K., Y. Hu, T. Kallunki, M. Schreiber, J.P. David, W. Jochum, E.F. Wagner, and M. Karin JNK2 is required for efficient T-cell activation and apoptosis but not for normal lymphocyte development. Curr. Biol. 9: Swat, W., K. Fujikawa, S. Ganiatsas, D. Yang, R.J. Xavier, N.L. Harris, L. Davidson, R. Ferrini, R.J. Davis, M.A. Labow, et al SEK1/MKK4 is required for maintenance of a normal peripheral lymphoid compartment but not for lymphocyte development. Immunity. 8: Gärtner, F., F.W. Alt, R. Monroe, M. Chu, B.P. Sleckman, L. Davidson, and W. Swat Immature thymocytes employ distinct signaling pathways for allelic exclusion versus differentiation and expansion. Immunity. 10: Michie et al.
During development of T cells in the thymus most
 Role of Different T Cell Receptors in the Development of Pre T Cells By Jan Buer,* Iannis Aifantis,* James P. DiSanto, Hans Joerg Fehling, and Harald von Boehmer* From the *Institut Necker, Institut National
Role of Different T Cell Receptors in the Development of Pre T Cells By Jan Buer,* Iannis Aifantis,* James P. DiSanto, Hans Joerg Fehling, and Harald von Boehmer* From the *Institut Necker, Institut National
This information is current as of August 19, Gillian C. Grady, Susan M. Mason, Jillian Stephen, Juan Carlos Zúñiga-Pflücker and Alison M.
 This information is current as of August 19, 2018. References Subscription Permissions Email Alerts Cyclic Adenosine 5 -Monophosphate Response Element Binding Protein Plays a Central Role in Mediating
This information is current as of August 19, 2018. References Subscription Permissions Email Alerts Cyclic Adenosine 5 -Monophosphate Response Element Binding Protein Plays a Central Role in Mediating
Unexpectedly late expression of intracellular CD3ε and TCR γδ proteins during adult thymus development
 International Immunology, Vol. 11, No. 10, pp. 1641 1650 1999 The Japanese Society for Immunology Unexpectedly late expression of intracellular CD3ε and TCR γδ proteins during adult thymus development
International Immunology, Vol. 11, No. 10, pp. 1641 1650 1999 The Japanese Society for Immunology Unexpectedly late expression of intracellular CD3ε and TCR γδ proteins during adult thymus development
Multiple nonreceptor protein tyrosine kinases (PTKs)
 Brief Definitive Report Itk and Fyn Make Independent Contributions to T Cell Activation By X. Charlene Liao,* Dan R. Littman, and Arthur Weiss* From the *Department of Microbiology and Immunology, and
Brief Definitive Report Itk and Fyn Make Independent Contributions to T Cell Activation By X. Charlene Liao,* Dan R. Littman, and Arthur Weiss* From the *Department of Microbiology and Immunology, and
T cell maturation. T-cell Maturation. What allows T cell maturation?
 T-cell Maturation What allows T cell maturation? Direct contact with thymic epithelial cells Influence of thymic hormones Growth factors (cytokines, CSF) T cell maturation T cell progenitor DN DP SP 2ry
T-cell Maturation What allows T cell maturation? Direct contact with thymic epithelial cells Influence of thymic hormones Growth factors (cytokines, CSF) T cell maturation T cell progenitor DN DP SP 2ry
Specific requirement for CD3 in T cell development
 Proc. Natl. Acad. Sci. USA Vol. 95, pp. 14909 14914, December 1998 Immunology Specific requirement for CD3 in T cell development JAN B. DEJARNETTE*, CONNIE L. SOMMERS*, KUN HUANG*, KENNETH J. WOODSIDE*,
Proc. Natl. Acad. Sci. USA Vol. 95, pp. 14909 14914, December 1998 Immunology Specific requirement for CD3 in T cell development JAN B. DEJARNETTE*, CONNIE L. SOMMERS*, KUN HUANG*, KENNETH J. WOODSIDE*,
Microbiology 204. Background Slides on T Cell Development For Preparation for Flipped Classroom setting. Art Weiss.
 Microbiology 204 Background Slides on T Cell Development For Preparation for Flipped Classroom setting Art Weiss October 23, 2015 Thymic Lobule Structure From: Immunobiology, Janeway, et al., 5th edition
Microbiology 204 Background Slides on T Cell Development For Preparation for Flipped Classroom setting Art Weiss October 23, 2015 Thymic Lobule Structure From: Immunobiology, Janeway, et al., 5th edition
Development of B and T lymphocytes
 Development of B and T lymphocytes What will we discuss today? B-cell development T-cell development B- cell development overview Stem cell In periphery Pro-B cell Pre-B cell Immature B cell Mature B cell
Development of B and T lymphocytes What will we discuss today? B-cell development T-cell development B- cell development overview Stem cell In periphery Pro-B cell Pre-B cell Immature B cell Mature B cell
T cell development October 28, Dan Stetson
 T cell development October 28, 2016 Dan Stetson stetson@uw.edu 441 Lecture #13 Slide 1 of 29 Three lectures on T cells (Chapters 8, 9) Part 1 (Today): T cell development in the thymus Chapter 8, pages
T cell development October 28, 2016 Dan Stetson stetson@uw.edu 441 Lecture #13 Slide 1 of 29 Three lectures on T cells (Chapters 8, 9) Part 1 (Today): T cell development in the thymus Chapter 8, pages
The Differential Staging of Murine Thymic Lymphoma Cell Lines, Scid.adh, R1.1 and EL-4
 The Differential Staging of Murine Thymic Lymphoma Cell Lines, Scid.adh, R1.1 and EL-4 Jong Seok Chae 1, Hae-jung Kim 1, Weon Seo Park 1, Youngmee Bae 1 and Kyeong Cheon Jung 2 1 Department of Pathology,
The Differential Staging of Murine Thymic Lymphoma Cell Lines, Scid.adh, R1.1 and EL-4 Jong Seok Chae 1, Hae-jung Kim 1, Weon Seo Park 1, Youngmee Bae 1 and Kyeong Cheon Jung 2 1 Department of Pathology,
During their development, thymocytes are subjected to
 Role of the Multiple T Cell Receptor (TCR)- Chain Signaling Motifs in Selection of the T Cell Repertoire By Elizabeth W. Shores,* Tom Tran,* Alexander Grinberg, Connie L. Sommers, Howard Shen, and Paul
Role of the Multiple T Cell Receptor (TCR)- Chain Signaling Motifs in Selection of the T Cell Repertoire By Elizabeth W. Shores,* Tom Tran,* Alexander Grinberg, Connie L. Sommers, Howard Shen, and Paul
T Cell Development. Xuefang Cao, MD, PhD. November 3, 2015
 T Cell Development Xuefang Cao, MD, PhD November 3, 2015 Thymocytes in the cortex of the thymus Early thymocytes development Positive and negative selection Lineage commitment Exit from the thymus and
T Cell Development Xuefang Cao, MD, PhD November 3, 2015 Thymocytes in the cortex of the thymus Early thymocytes development Positive and negative selection Lineage commitment Exit from the thymus and
Tcell development progresses in the thymus via an ordered
 Published Online: 5 April, 1999 Supp Info: http://doi.org/10.1084/jem.189.7.1163 Downloaded from jem.rupress.org on April 28, 2018 Brief Definitive Report ZAP-70 Protein Promotes Tyrosine Phosphorylation
Published Online: 5 April, 1999 Supp Info: http://doi.org/10.1084/jem.189.7.1163 Downloaded from jem.rupress.org on April 28, 2018 Brief Definitive Report ZAP-70 Protein Promotes Tyrosine Phosphorylation
Lecture 7: Signaling Through Lymphocyte Receptors
 Lecture 7: Signaling Through Lymphocyte Receptors Questions to Consider After recognition of its cognate MHC:peptide, how does the T cell receptor activate immune response genes? What are the structural
Lecture 7: Signaling Through Lymphocyte Receptors Questions to Consider After recognition of its cognate MHC:peptide, how does the T cell receptor activate immune response genes? What are the structural
Recommended reading: Abbas et al. 5th edition, chapters 7 and 10; Janeway and Travers, 5th edition, chapter 7.
 Harvard-MIT Division of Health Sciences and Technology HST.176: Cellular and Molecular Immunology Course Director: Dr. Shiv Pillai 10/05/05; 11 AM Shiv Pillai T Lymphocyte Development Recommended reading:
Harvard-MIT Division of Health Sciences and Technology HST.176: Cellular and Molecular Immunology Course Director: Dr. Shiv Pillai 10/05/05; 11 AM Shiv Pillai T Lymphocyte Development Recommended reading:
Supplementary Table; Supplementary Figures and legends S1-S21; Supplementary Materials and Methods
 Silva et al. PTEN posttranslational inactivation and hyperactivation of the PI3K/Akt pathway sustain primary T cell leukemia viability Supplementary Table; Supplementary Figures and legends S1-S21; Supplementary
Silva et al. PTEN posttranslational inactivation and hyperactivation of the PI3K/Akt pathway sustain primary T cell leukemia viability Supplementary Table; Supplementary Figures and legends S1-S21; Supplementary
Data Sheet TIGIT / NFAT Reporter - Jurkat Cell Line Catalog #60538
 Data Sheet TIGIT / NFAT Reporter - Jurkat Cell Line Catalog #60538 Background: TIGIT is a co-inhibitory receptor that is highly expressed in Natural Killer (NK) cells, activated CD4+, CD8+ and regulatory
Data Sheet TIGIT / NFAT Reporter - Jurkat Cell Line Catalog #60538 Background: TIGIT is a co-inhibitory receptor that is highly expressed in Natural Killer (NK) cells, activated CD4+, CD8+ and regulatory
MEK1 Assay Kit 1 Catalog # Lot # 16875
 MEK1 Assay Kit 1 Kit Components Assay Dilution Buffer (ADB), Catalog # 20-108. Three vials, each containing 1.0ml of assay dilution buffer (20mM MOPS, ph 7.2, 25mM ß-glycerol phosphate, 5mM EGTA, 1mM sodium
MEK1 Assay Kit 1 Kit Components Assay Dilution Buffer (ADB), Catalog # 20-108. Three vials, each containing 1.0ml of assay dilution buffer (20mM MOPS, ph 7.2, 25mM ß-glycerol phosphate, 5mM EGTA, 1mM sodium
During embryogenesis, T lineage committed lymphoid
 Regulation of Thymocyte Development through CD3. II. Expression of T Cell Receptor 0 CD3e and Maturation to the CD4+8+ Stage Are Highly Correlated in Individual Thymocytes By Christiaan N. L,evelt, Rita
Regulation of Thymocyte Development through CD3. II. Expression of T Cell Receptor 0 CD3e and Maturation to the CD4+8+ Stage Are Highly Correlated in Individual Thymocytes By Christiaan N. L,evelt, Rita
Chapter 11. B cell generation, Activation, and Differentiation. Pro-B cells. - B cells mature in the bone marrow.
 Chapter B cell generation, Activation, and Differentiation - B cells mature in the bone marrow. - B cells proceed through a number of distinct maturational stages: ) Pro-B cell ) Pre-B cell ) Immature
Chapter B cell generation, Activation, and Differentiation - B cells mature in the bone marrow. - B cells proceed through a number of distinct maturational stages: ) Pro-B cell ) Pre-B cell ) Immature
Nature Immunology: doi: /ni.3631
 Supplementary Figure 1 SPT analyses of Zap70 at the T cell plasma membrane. (a) Total internal reflection fluorescent (TIRF) excitation at 64-68 degrees limits single molecule detection to 100-150 nm above
Supplementary Figure 1 SPT analyses of Zap70 at the T cell plasma membrane. (a) Total internal reflection fluorescent (TIRF) excitation at 64-68 degrees limits single molecule detection to 100-150 nm above
Chapter 11. B cell generation, Activation, and Differentiation. Pro-B cells. - B cells mature in the bone marrow.
 Chapter B cell generation, Activation, and Differentiation - B cells mature in the bone marrow. - B cells proceed through a number of distinct maturational stages: ) Pro-B cell ) Pre-B cell ) Immature
Chapter B cell generation, Activation, and Differentiation - B cells mature in the bone marrow. - B cells proceed through a number of distinct maturational stages: ) Pro-B cell ) Pre-B cell ) Immature
Supplementary Materials for
 www.sciencesignaling.org/cgi/content/full/3/114/ra23/dc1 Supplementary Materials for Regulation of Zap70 Expression During Thymocyte Development Enables Temporal Separation of CD4 and CD8 Repertoire Selection
www.sciencesignaling.org/cgi/content/full/3/114/ra23/dc1 Supplementary Materials for Regulation of Zap70 Expression During Thymocyte Development Enables Temporal Separation of CD4 and CD8 Repertoire Selection
Electrical Stimulation Control Nerve Regeneration via the p38 Mitogen-activated Protein Kinase and CREB
 Electrical Stimulation Control Nerve Regeneration via the p38 Mitogen-activated Protein Kinase and CREB Kenji Kawamura, Yoshio Kano. Kibi International University, Takahashi-city, Japan. Disclosures: K.
Electrical Stimulation Control Nerve Regeneration via the p38 Mitogen-activated Protein Kinase and CREB Kenji Kawamura, Yoshio Kano. Kibi International University, Takahashi-city, Japan. Disclosures: K.
Supplementary data Supplementary Figure 1 Supplementary Figure 2
 Supplementary data Supplementary Figure 1 SPHK1 sirna increases RANKL-induced osteoclastogenesis in RAW264.7 cell culture. (A) RAW264.7 cells were transfected with oligocassettes containing SPHK1 sirna
Supplementary data Supplementary Figure 1 SPHK1 sirna increases RANKL-induced osteoclastogenesis in RAW264.7 cell culture. (A) RAW264.7 cells were transfected with oligocassettes containing SPHK1 sirna
Functional analysis of LAT in TCR-mediated signaling pathways using a LAT-deficient Jurkat cell line
 International Immunology, Vol. 11, No. 6, pp. 943 950 Functional analysis of LAT in TCR-mediated signaling pathways using a LAT-deficient Jurkat cell line Weiguo Zhang, Brenda J. Irvin 1,2, Ronald P. Trible,
International Immunology, Vol. 11, No. 6, pp. 943 950 Functional analysis of LAT in TCR-mediated signaling pathways using a LAT-deficient Jurkat cell line Weiguo Zhang, Brenda J. Irvin 1,2, Ronald P. Trible,
SUPPLEMENTARY INFORMATION
 Supplementary Figures Supplementary Figure S1. Binding of full-length OGT and deletion mutants to PIP strips (Echelon Biosciences). Supplementary Figure S2. Binding of the OGT (919-1036) fragments with
Supplementary Figures Supplementary Figure S1. Binding of full-length OGT and deletion mutants to PIP strips (Echelon Biosciences). Supplementary Figure S2. Binding of the OGT (919-1036) fragments with
Detailed step-by-step operating procedures for NK cell and CTL degranulation assays
 Supplemental methods Detailed step-by-step operating procedures for NK cell and CTL degranulation assays Materials PBMC isolated from patients, relatives and healthy donors as control K562 cells (ATCC,
Supplemental methods Detailed step-by-step operating procedures for NK cell and CTL degranulation assays Materials PBMC isolated from patients, relatives and healthy donors as control K562 cells (ATCC,
T Cell Differentiation
 T Cell Differentiation Ned Braunstein, MD MHC control of Immune Responsiveness: Concept Whether or not an individual makes an immune response to a particular antigen depends on what MHC alleles an individual
T Cell Differentiation Ned Braunstein, MD MHC control of Immune Responsiveness: Concept Whether or not an individual makes an immune response to a particular antigen depends on what MHC alleles an individual
SUPPLEMENTAL INFORMATION
 SUPPLEMENTAL INFORMATION EXPERIMENTAL PROCEDURES Tryptic digestion protection experiments - PCSK9 with Ab-3D5 (1:1 molar ratio) in 50 mm Tris, ph 8.0, 150 mm NaCl was incubated overnight at 4 o C. The
SUPPLEMENTAL INFORMATION EXPERIMENTAL PROCEDURES Tryptic digestion protection experiments - PCSK9 with Ab-3D5 (1:1 molar ratio) in 50 mm Tris, ph 8.0, 150 mm NaCl was incubated overnight at 4 o C. The
SUPPLEMENTARY INFORMATION
 Complete but curtailed T-cell response to very-low-affinity antigen Dietmar Zehn, Sarah Y. Lee & Michael J. Bevan Supp. Fig. 1: TCR chain usage among endogenous K b /Ova reactive T cells. C57BL/6 mice
Complete but curtailed T-cell response to very-low-affinity antigen Dietmar Zehn, Sarah Y. Lee & Michael J. Bevan Supp. Fig. 1: TCR chain usage among endogenous K b /Ova reactive T cells. C57BL/6 mice
MTC-TT and TPC-1 cell lines were cultured in RPMI medium (Gibco, Breda, The Netherlands)
 Supplemental data Materials and Methods Cell culture MTC-TT and TPC-1 cell lines were cultured in RPMI medium (Gibco, Breda, The Netherlands) supplemented with 15% or 10% (for TPC-1) fetal bovine serum
Supplemental data Materials and Methods Cell culture MTC-TT and TPC-1 cell lines were cultured in RPMI medium (Gibco, Breda, The Netherlands) supplemented with 15% or 10% (for TPC-1) fetal bovine serum
T Cell Development II: Positive and Negative Selection
 T Cell Development II: Positive and Negative Selection 8 88 The two phases of thymic development: - production of T cell receptors for antigen, by rearrangement of the TCR genes CD4 - selection of T cells
T Cell Development II: Positive and Negative Selection 8 88 The two phases of thymic development: - production of T cell receptors for antigen, by rearrangement of the TCR genes CD4 - selection of T cells
Supplementary Information
 Supplementary Information Supplementary Figure 1. CD4 + T cell activation and lack of apoptosis after crosslinking with anti-cd3 + anti-cd28 + anti-cd160. (a) Flow cytometry of anti-cd160 (5D.10A11) binding
Supplementary Information Supplementary Figure 1. CD4 + T cell activation and lack of apoptosis after crosslinking with anti-cd3 + anti-cd28 + anti-cd160. (a) Flow cytometry of anti-cd160 (5D.10A11) binding
Serum Amyloid A3 Gene Expression in Adipocytes is an Indicator. of the Interaction with Macrophages
 Serum Amyloid A3 Gene Expression in Adipocytes is an Indicator of the Interaction with Macrophages Yohei Sanada, Takafumi Yamamoto, Rika Satake, Akiko Yamashita, Sumire Kanai, Norihisa Kato, Fons AJ van
Serum Amyloid A3 Gene Expression in Adipocytes is an Indicator of the Interaction with Macrophages Yohei Sanada, Takafumi Yamamoto, Rika Satake, Akiko Yamashita, Sumire Kanai, Norihisa Kato, Fons AJ van
Data Sheet. NFAT Reporter (Luc) Jurkat Cell line Catalog #: 60621
 Data Sheet NFAT Reporter (Luc) Jurkat Cell line Catalog #: 60621 Background The nuclear factor of activator T cells (NFAT) family of transcription factors plays an important role in immune response. T
Data Sheet NFAT Reporter (Luc) Jurkat Cell line Catalog #: 60621 Background The nuclear factor of activator T cells (NFAT) family of transcription factors plays an important role in immune response. T
As we know, T cell precursors (pro-t cells) originate in the bone marrow and migrate to the thymus via the blood.
 Immunology Dr. John J. Haddad Chapter 10 T Cell Maturation, Activation and Differentiation T Cell Maturation As we know, T cell precursors (pro-t cells) originate in the bone marrow and migrate to the
Immunology Dr. John J. Haddad Chapter 10 T Cell Maturation, Activation and Differentiation T Cell Maturation As we know, T cell precursors (pro-t cells) originate in the bone marrow and migrate to the
Data Sheet IL-2-Luciferase Reporter (Luc) - Jurkat Cell Line Catalog # 60481
 642 Cornerstone Court W, Ste B Tel: 1.858.829.382 Data Sheet IL-2-Luciferase Reporter (Luc) - Jurkat Cell Line Catalog # 6481 Description Human IL-2 reporter construct is stably integrated into the genome
642 Cornerstone Court W, Ste B Tel: 1.858.829.382 Data Sheet IL-2-Luciferase Reporter (Luc) - Jurkat Cell Line Catalog # 6481 Description Human IL-2 reporter construct is stably integrated into the genome
Immune Regulation and Tolerance
 Immune Regulation and Tolerance Immunoregulation: A balance between activation and suppression of effector cells to achieve an efficient immune response without damaging the host. Activation (immunity)
Immune Regulation and Tolerance Immunoregulation: A balance between activation and suppression of effector cells to achieve an efficient immune response without damaging the host. Activation (immunity)
Introduction. Introduction. Lymphocyte development (maturation)
 Introduction Abbas Chapter 8: Lymphocyte Development and the Rearrangement and Expression of Antigen Receptor Genes Christina Ciaccio, MD Children s Mercy Hospital January 5, 2009 Lymphocyte development
Introduction Abbas Chapter 8: Lymphocyte Development and the Rearrangement and Expression of Antigen Receptor Genes Christina Ciaccio, MD Children s Mercy Hospital January 5, 2009 Lymphocyte development
SUPPLEMENTARY INFORMATION
 doi:10.1038/nature10495 WWW.NATURE.COM/NATURE 1 2 WWW.NATURE.COM/NATURE WWW.NATURE.COM/NATURE 3 4 WWW.NATURE.COM/NATURE WWW.NATURE.COM/NATURE 5 6 WWW.NATURE.COM/NATURE WWW.NATURE.COM/NATURE 7 8 WWW.NATURE.COM/NATURE
doi:10.1038/nature10495 WWW.NATURE.COM/NATURE 1 2 WWW.NATURE.COM/NATURE WWW.NATURE.COM/NATURE 3 4 WWW.NATURE.COM/NATURE WWW.NATURE.COM/NATURE 5 6 WWW.NATURE.COM/NATURE WWW.NATURE.COM/NATURE 7 8 WWW.NATURE.COM/NATURE
Attribution: University of Michigan Medical School, Department of Microbiology and Immunology
 Attribution: University of Michigan Medical School, Department of Microbiology and Immunology License: Unless otherwise noted, this material is made available under the terms of the Creative Commons Attribution
Attribution: University of Michigan Medical School, Department of Microbiology and Immunology License: Unless otherwise noted, this material is made available under the terms of the Creative Commons Attribution
Supplementary Figure 1 Role of Raf-1 in TLR2-Dectin-1-mediated cytokine expression
 Supplementary Figure 1 Supplementary Figure 1 Role of Raf-1 in TLR2-Dectin-1-mediated cytokine expression. Quantitative real-time PCR of indicated mrnas in DCs stimulated with TLR2-Dectin-1 agonist zymosan
Supplementary Figure 1 Supplementary Figure 1 Role of Raf-1 in TLR2-Dectin-1-mediated cytokine expression. Quantitative real-time PCR of indicated mrnas in DCs stimulated with TLR2-Dectin-1 agonist zymosan
5/1/13. The proportion of thymus that produces T cells decreases with age. The cellular organization of the thymus
 T cell precursors migrate from the bone marrow via the blood to the thymus to mature 1 2 The cellular organization of the thymus The proportion of thymus that produces T cells decreases with age 3 4 1
T cell precursors migrate from the bone marrow via the blood to the thymus to mature 1 2 The cellular organization of the thymus The proportion of thymus that produces T cells decreases with age 3 4 1
Tyrosine Phosphorylation of Pyk2 Is Selectively Regulated by Fyn During TCR Signaling
 Tyrosine Phosphorylation of Pyk2 Is Selectively Regulated by Fyn During TCR Signaling By Dapeng Qian,* Sima Lev, Nicolai S.C. van Oers,* Ivan Dikic, Joseph Schlessinger, and Arthur Weiss* From the *Howard
Tyrosine Phosphorylation of Pyk2 Is Selectively Regulated by Fyn During TCR Signaling By Dapeng Qian,* Sima Lev, Nicolai S.C. van Oers,* Ivan Dikic, Joseph Schlessinger, and Arthur Weiss* From the *Howard
Intracellular MHC class II molecules promote TLR-triggered innate. immune responses by maintaining Btk activation
 Intracellular MHC class II molecules promote TLR-triggered innate immune responses by maintaining Btk activation Xingguang Liu, Zhenzhen Zhan, Dong Li, Li Xu, Feng Ma, Peng Zhang, Hangping Yao and Xuetao
Intracellular MHC class II molecules promote TLR-triggered innate immune responses by maintaining Btk activation Xingguang Liu, Zhenzhen Zhan, Dong Li, Li Xu, Feng Ma, Peng Zhang, Hangping Yao and Xuetao
Pre-made Reporter Lentivirus for MAPK/ERK Signal Pathway
 Pre-made Reporter for MAPK/ERK Signal Pathway Cat# Product Name Amounts LVP957-P or: LVP957-P-PBS SRE-GFP (Puro) LVP958-P or: LVP958-P-PBS SRE-RFP (Puro) LVP959-P or: LVP959-P-PBS SRE-Luc (Puro) LVP960-P
Pre-made Reporter for MAPK/ERK Signal Pathway Cat# Product Name Amounts LVP957-P or: LVP957-P-PBS SRE-GFP (Puro) LVP958-P or: LVP958-P-PBS SRE-RFP (Puro) LVP959-P or: LVP959-P-PBS SRE-Luc (Puro) LVP960-P
Part-4. Cell cycle regulatory protein 5 (Cdk5) A novel target of ERK in Carb induced cell death
 Part-4 Cell cycle regulatory protein 5 (Cdk5) A novel target of ERK in Carb induced cell death 95 1. Introduction The process of replicating DNA and dividing cells can be described as a series of coordinated
Part-4 Cell cycle regulatory protein 5 (Cdk5) A novel target of ERK in Carb induced cell death 95 1. Introduction The process of replicating DNA and dividing cells can be described as a series of coordinated
Ex vivo Human Antigen-specific T Cell Proliferation and Degranulation Willemijn Hobo 1, Wieger Norde 1 and Harry Dolstra 2*
 Ex vivo Human Antigen-specific T Cell Proliferation and Degranulation Willemijn Hobo 1, Wieger Norde 1 and Harry Dolstra 2* 1 Department of Laboratory Medicine - Laboratory of Hematology, Radboud University
Ex vivo Human Antigen-specific T Cell Proliferation and Degranulation Willemijn Hobo 1, Wieger Norde 1 and Harry Dolstra 2* 1 Department of Laboratory Medicine - Laboratory of Hematology, Radboud University
T-cell activation T cells migrate to secondary lymphoid tissues where they interact with antigen, antigen-presenting cells, and other lymphocytes:
 Interactions between innate immunity & adaptive immunity What happens to T cells after they leave the thymus? Naïve T cells exit the thymus and enter the bloodstream. If they remain in the bloodstream,
Interactions between innate immunity & adaptive immunity What happens to T cells after they leave the thymus? Naïve T cells exit the thymus and enter the bloodstream. If they remain in the bloodstream,
T-cell activation T cells migrate to secondary lymphoid tissues where they interact with antigen, antigen-presenting cells, and other lymphocytes:
 Interactions between innate immunity & adaptive immunity What happens to T cells after they leave the thymus? Naïve T cells exit the thymus and enter the bloodstream. If they remain in the bloodstream,
Interactions between innate immunity & adaptive immunity What happens to T cells after they leave the thymus? Naïve T cells exit the thymus and enter the bloodstream. If they remain in the bloodstream,
T cell and Cell-mediated immunity
 T cell and Cell-mediated immunity ( 第十章 第十二章第十二章 ) Lu Linrong ( 鲁林荣 ) PhD Laboratory of Immune Regulation Institute of Immunology Zhejiang University, School of Medicine Medical Research Building B815-819
T cell and Cell-mediated immunity ( 第十章 第十二章第十二章 ) Lu Linrong ( 鲁林荣 ) PhD Laboratory of Immune Regulation Institute of Immunology Zhejiang University, School of Medicine Medical Research Building B815-819
Supplemental Experimental Procedures
 Cell Stem Cell, Volume 2 Supplemental Data A Temporal Switch from Notch to Wnt Signaling in Muscle Stem Cells Is Necessary for Normal Adult Myogenesis Andrew S. Brack, Irina M. Conboy, Michael J. Conboy,
Cell Stem Cell, Volume 2 Supplemental Data A Temporal Switch from Notch to Wnt Signaling in Muscle Stem Cells Is Necessary for Normal Adult Myogenesis Andrew S. Brack, Irina M. Conboy, Michael J. Conboy,
Supplementary Data 1. Alanine substitutions and position variants of APNCYGNIPL. Applied in
 Supplementary Data 1. Alanine substitutions and position variants of APNCYGNIPL. Applied in Supplementary Fig. 2 Substitution Sequence Position variant Sequence original APNCYGNIPL original APNCYGNIPL
Supplementary Data 1. Alanine substitutions and position variants of APNCYGNIPL. Applied in Supplementary Fig. 2 Substitution Sequence Position variant Sequence original APNCYGNIPL original APNCYGNIPL
The development of T cells in the thymus
 T cells rearrange their receptors in the thymus whereas B cells do so in the bone marrow. The development of T cells in the thymus The lobular/cellular organization of the thymus Immature cells are called
T cells rearrange their receptors in the thymus whereas B cells do so in the bone marrow. The development of T cells in the thymus The lobular/cellular organization of the thymus Immature cells are called
W/T Itgam -/- F4/80 CD115. F4/80 hi CD115 + F4/80 + CD115 +
 F4/8 % in the peritoneal lavage 6 4 2 p=.15 n.s p=.76 CD115 F4/8 hi CD115 + F4/8 + CD115 + F4/8 hi CD115 + F4/8 + CD115 + MHCII MHCII Supplementary Figure S1. CD11b deficiency affects the cellular responses
F4/8 % in the peritoneal lavage 6 4 2 p=.15 n.s p=.76 CD115 F4/8 hi CD115 + F4/8 + CD115 + F4/8 hi CD115 + F4/8 + CD115 + MHCII MHCII Supplementary Figure S1. CD11b deficiency affects the cellular responses
Autonomous Maturation of / T Lineage Cells in the Absence of COOH-terminal Src Kinase (Csk)
 Autonomous Maturation of / T Lineage Cells in the Absence of COOH-terminal Src Kinase (Csk) By Christian Schmedt and Alexander Tarakhovsky From the Laboratory for Lymphocyte Signaling, The Rockefeller
Autonomous Maturation of / T Lineage Cells in the Absence of COOH-terminal Src Kinase (Csk) By Christian Schmedt and Alexander Tarakhovsky From the Laboratory for Lymphocyte Signaling, The Rockefeller
T H 1, T H 2 and T H 17 polarization of naïve CD4 + mouse T cells
 A complete workflow for cell preparation, isolation, polarization and analysis T H 1, T H 2 and T H 17 polarization of naïve CD4 + mouse T cells Introduction Workflow CD4 + T helper (T H) cells play a
A complete workflow for cell preparation, isolation, polarization and analysis T H 1, T H 2 and T H 17 polarization of naïve CD4 + mouse T cells Introduction Workflow CD4 + T helper (T H) cells play a
Figure S1. Generation of inducible PTEN deficient mice and the BMMCs (A) B6.129 Pten loxp/loxp mice were mated with B6.
 Figure S1. Generation of inducible PTEN deficient mice and the BMMCs (A) B6.129 Pten loxp/loxp mice were mated with B6.129-Gt(ROSA)26Sor tm1(cre/ert2)tyj /J mice. To induce deletion of the Pten locus,
Figure S1. Generation of inducible PTEN deficient mice and the BMMCs (A) B6.129 Pten loxp/loxp mice were mated with B6.129-Gt(ROSA)26Sor tm1(cre/ert2)tyj /J mice. To induce deletion of the Pten locus,
Instructions for Use. APO-AB Annexin V-Biotin Apoptosis Detection Kit 100 tests
 3URGXFW,QIRUPDWLRQ Sigma TACS Annexin V Apoptosis Detection Kits Instructions for Use APO-AB Annexin V-Biotin Apoptosis Detection Kit 100 tests For Research Use Only. Not for use in diagnostic procedures.
3URGXFW,QIRUPDWLRQ Sigma TACS Annexin V Apoptosis Detection Kits Instructions for Use APO-AB Annexin V-Biotin Apoptosis Detection Kit 100 tests For Research Use Only. Not for use in diagnostic procedures.
SUPPLEMENTARY INFORMATION
 doi: 1.138/nature775 4 O.D. (595-655) 3 1 -ζ no antibody isotype ctrl Plated Soluble 1F6 397 7H11 Supplementary Figure 1 Soluble and plated anti- Abs induce -! signalling. B3Z cells stably expressing!
doi: 1.138/nature775 4 O.D. (595-655) 3 1 -ζ no antibody isotype ctrl Plated Soluble 1F6 397 7H11 Supplementary Figure 1 Soluble and plated anti- Abs induce -! signalling. B3Z cells stably expressing!
Signaling Alterations in Activation-Induced Nonresponsive CD8 T Cells
 This information is current as of November 11, 2018. Signaling Alterations in Activation-Induced Nonresponsive CD8 T Cells Ee Loon Tham and Matthew F. Mescher J Immunol 2001; 167:2040-2048; ; doi: 10.4049/jimmunol.167.4.2040
This information is current as of November 11, 2018. Signaling Alterations in Activation-Induced Nonresponsive CD8 T Cells Ee Loon Tham and Matthew F. Mescher J Immunol 2001; 167:2040-2048; ; doi: 10.4049/jimmunol.167.4.2040
A Hepatocyte Growth Factor Receptor (Met) Insulin Receptor hybrid governs hepatic glucose metabolism SUPPLEMENTARY FIGURES, LEGENDS AND METHODS
 A Hepatocyte Growth Factor Receptor (Met) Insulin Receptor hybrid governs hepatic glucose metabolism Arlee Fafalios, Jihong Ma, Xinping Tan, John Stoops, Jianhua Luo, Marie C. DeFrances and Reza Zarnegar
A Hepatocyte Growth Factor Receptor (Met) Insulin Receptor hybrid governs hepatic glucose metabolism Arlee Fafalios, Jihong Ma, Xinping Tan, John Stoops, Jianhua Luo, Marie C. DeFrances and Reza Zarnegar
Cell Signaling part 2
 15 Cell Signaling part 2 Functions of Cell Surface Receptors Other cell surface receptors are directly linked to intracellular enzymes. The largest family of these is the receptor protein tyrosine kinases,
15 Cell Signaling part 2 Functions of Cell Surface Receptors Other cell surface receptors are directly linked to intracellular enzymes. The largest family of these is the receptor protein tyrosine kinases,
Signaling Through Immune System Receptors (Ch. 7)
 Signaling Through Immune System Receptors (Ch. 7) 1. General principles of signal transduction and propagation. 2. Antigen receptor signaling and lymphocyte activation. 3. Other receptors and signaling
Signaling Through Immune System Receptors (Ch. 7) 1. General principles of signal transduction and propagation. 2. Antigen receptor signaling and lymphocyte activation. 3. Other receptors and signaling
Early Thymocyte Development Is Regulated by Modulation of E2A Protein Activity
 Published Online: 10 September, 2001 Supp Info: http://doi.org/10.1084/jem.194.6.733 Downloaded from jem.rupress.org on January 15, 2019 Early Thymocyte Development Is Regulated by Modulation of E2A Protein
Published Online: 10 September, 2001 Supp Info: http://doi.org/10.1084/jem.194.6.733 Downloaded from jem.rupress.org on January 15, 2019 Early Thymocyte Development Is Regulated by Modulation of E2A Protein
Technical Resources. BD Immunocytometry Systems. FastImmune Intracellular Cytokine Staining Procedures
 FastImmune Intracellular Cytokine Staining Procedures BD has developed protocols for the detection of intracellular cytokines in activated lymphocytes and in activated monocytes. The procedures have been
FastImmune Intracellular Cytokine Staining Procedures BD has developed protocols for the detection of intracellular cytokines in activated lymphocytes and in activated monocytes. The procedures have been
Product Datasheet. HLA ABC Antibody (W6/32) NB Unit Size: 0.25 mg. Store at -20C. Avoid freeze-thaw cycles. Reviews: 1 Publications: 22
 Product Datasheet HLA ABC Antibody (W6/32) NB100-64775 Unit Size: 0.25 mg Store at -20C. Avoid freeze-thaw cycles. Reviews: 1 Publications: 22 Protocols, Publications, Related Products, Reviews, Research
Product Datasheet HLA ABC Antibody (W6/32) NB100-64775 Unit Size: 0.25 mg Store at -20C. Avoid freeze-thaw cycles. Reviews: 1 Publications: 22 Protocols, Publications, Related Products, Reviews, Research
Ig light chain rearrangement: Rescue pathway
 B Cell Development Ig light chain rearrangement: Rescue pathway There is only a 1:3 chance of the join between the V and J region being in frame Vk Jk Ck Non-productive Rearrangement Light chain has a
B Cell Development Ig light chain rearrangement: Rescue pathway There is only a 1:3 chance of the join between the V and J region being in frame Vk Jk Ck Non-productive Rearrangement Light chain has a
Kinase Activation and CD8 T Cell Development
 This information is current as of February 23, 2013. The Cytoplasmic Domain of CD8β Regulates Lck Kinase Activation and CD8 T Cell Development Hanna Yoko Irie, Mimi S. Mong, Andrea Itano, M. E. Casey Crooks,
This information is current as of February 23, 2013. The Cytoplasmic Domain of CD8β Regulates Lck Kinase Activation and CD8 T Cell Development Hanna Yoko Irie, Mimi S. Mong, Andrea Itano, M. E. Casey Crooks,
A novel bfgf antagonist peptide inhibits breast cancer cell growth
 210 A novel bfgf antagonist peptide inhibits breast cancer cell growth QUCHOU LI 1, SUSU GAO 1, YONGLIN YU 1, WENHUI WANG 1, XILEI CHEN 1, RUIXUE WANG 1, TAO LI 1, CONG WANG 1, XIAOKUN LI 1,2 and XIAOPING
210 A novel bfgf antagonist peptide inhibits breast cancer cell growth QUCHOU LI 1, SUSU GAO 1, YONGLIN YU 1, WENHUI WANG 1, XILEI CHEN 1, RUIXUE WANG 1, TAO LI 1, CONG WANG 1, XIAOKUN LI 1,2 and XIAOPING
Cell isolation. Spleen and lymph nodes (axillary, inguinal) were removed from mice
 Supplementary Methods: Cell isolation. Spleen and lymph nodes (axillary, inguinal) were removed from mice and gently meshed in DMEM containing 10% FBS to prepare for single cell suspensions. CD4 + CD25
Supplementary Methods: Cell isolation. Spleen and lymph nodes (axillary, inguinal) were removed from mice and gently meshed in DMEM containing 10% FBS to prepare for single cell suspensions. CD4 + CD25
Product Datasheet. EMMPRIN/CD147 Antibody (MEM-M6/1) NB Unit Size: 0.1 mg. Store at 4C. Do not freeze. Publications: 2
 Product Datasheet EMMPRIN/CD147 Antibody (MEM-M6/1) NB500-430 Unit Size: 0.1 mg Store at 4C. Do not freeze. Publications: 2 Protocols, Publications, Related Products, Reviews, Research Tools and Images
Product Datasheet EMMPRIN/CD147 Antibody (MEM-M6/1) NB500-430 Unit Size: 0.1 mg Store at 4C. Do not freeze. Publications: 2 Protocols, Publications, Related Products, Reviews, Research Tools and Images
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION FOR Liver X Receptor α mediates hepatic triglyceride accumulation through upregulation of G0/G1 Switch Gene 2 (G0S2) expression I: SUPPLEMENTARY METHODS II: SUPPLEMENTARY FIGURES
SUPPLEMENTARY INFORMATION FOR Liver X Receptor α mediates hepatic triglyceride accumulation through upregulation of G0/G1 Switch Gene 2 (G0S2) expression I: SUPPLEMENTARY METHODS II: SUPPLEMENTARY FIGURES
Tlymphocytes are produced in the thymus, where T cell
 Published Online: 6 October, 1997 Supp Info: http://doi.org/10.1084/jem.186.7.1099 Downloaded from jem.rupress.org on July 9, 2018 Peripheral Selection of T Cell Repertoires: The Role of Continuous Thymus
Published Online: 6 October, 1997 Supp Info: http://doi.org/10.1084/jem.186.7.1099 Downloaded from jem.rupress.org on July 9, 2018 Peripheral Selection of T Cell Repertoires: The Role of Continuous Thymus
Computational Biology I LSM5191
 Computational Biology I LSM5191 Aylwin Ng, D.Phil Lecture 6 Notes: Control Systems in Gene Expression Pulling it all together: coordinated control of transcriptional regulatory molecules Simple Control:
Computational Biology I LSM5191 Aylwin Ng, D.Phil Lecture 6 Notes: Control Systems in Gene Expression Pulling it all together: coordinated control of transcriptional regulatory molecules Simple Control:
Supplementary Figure 1 Cytokine receptors on developing thymocytes that can potentially signal Runx3d expression.
 Supplementary Figure 1 Cytokine receptors on developing thymocytes that can potentially signal Runx3d expression. (a) Characterization of c-independent SP8 cells. Stainings for maturation markers (top)
Supplementary Figure 1 Cytokine receptors on developing thymocytes that can potentially signal Runx3d expression. (a) Characterization of c-independent SP8 cells. Stainings for maturation markers (top)
Supplementary Data Table of Contents:
 Supplementary Data Table of Contents: - Supplementary Methods - Supplementary Figures S1(A-B) - Supplementary Figures S2 (A-B) - Supplementary Figures S3 - Supplementary Figures S4(A-B) - Supplementary
Supplementary Data Table of Contents: - Supplementary Methods - Supplementary Figures S1(A-B) - Supplementary Figures S2 (A-B) - Supplementary Figures S3 - Supplementary Figures S4(A-B) - Supplementary
RAS Genes. The ras superfamily of genes encodes small GTP binding proteins that are responsible for the regulation of many cellular processes.
 ۱ RAS Genes The ras superfamily of genes encodes small GTP binding proteins that are responsible for the regulation of many cellular processes. Oncogenic ras genes in human cells include H ras, N ras,
۱ RAS Genes The ras superfamily of genes encodes small GTP binding proteins that are responsible for the regulation of many cellular processes. Oncogenic ras genes in human cells include H ras, N ras,
In vitro bactericidal assay Fig. S8 Gentamicin protection assay Phagocytosis assay
 In vitro bactericidal assay Mouse bone marrow was isolated from the femur and the tibia. Cells were suspended in phosphate buffered saline containing.5% BSA and 2 mm EDTA and filtered through a cell strainer.
In vitro bactericidal assay Mouse bone marrow was isolated from the femur and the tibia. Cells were suspended in phosphate buffered saline containing.5% BSA and 2 mm EDTA and filtered through a cell strainer.
Supplementary Information
 Supplementary Information Supplementary Figure 1! a! b! Nfatc1!! Nfatc1"! P1! P2! pa1! pa2! ex1! ex2! exons 3-9! ex1! ex11!!" #" Nfatc1A!!" Nfatc1B! #"!" Nfatc1C! #" DN1! DN2! DN1!!A! #A!!B! #B!!C! #C!!A!
Supplementary Information Supplementary Figure 1! a! b! Nfatc1!! Nfatc1"! P1! P2! pa1! pa2! ex1! ex2! exons 3-9! ex1! ex11!!" #" Nfatc1A!!" Nfatc1B! #"!" Nfatc1C! #" DN1! DN2! DN1!!A! #A!!B! #B!!C! #C!!A!
HCC1937 is the HCC1937-pcDNA3 cell line, which was derived from a breast cancer with a mutation
 SUPPLEMENTARY INFORMATION Materials and Methods Human cell lines and culture conditions HCC1937 is the HCC1937-pcDNA3 cell line, which was derived from a breast cancer with a mutation in exon 20 of BRCA1
SUPPLEMENTARY INFORMATION Materials and Methods Human cell lines and culture conditions HCC1937 is the HCC1937-pcDNA3 cell line, which was derived from a breast cancer with a mutation in exon 20 of BRCA1
Introduc)on to Immunology. T Cell Development
 Introduc)on to Immunology T Cell Development Adolfo Ferrando af2196@columbia.edu Thymic stroma cells provide the microenvironment for T cell development. T cells develop from progenitors that are derived
Introduc)on to Immunology T Cell Development Adolfo Ferrando af2196@columbia.edu Thymic stroma cells provide the microenvironment for T cell development. T cells develop from progenitors that are derived
Sipper BK Experimental Animal Co. (Shanghai, China) and bred in a specific. pathogen-free environment. The animal study protocol was approved by the
 Supplementary information, Data S1 Materials and Methods Mice, Ad vectors and reagents Female C57BL/6 mice, 8-10 weeks of age, were purchased from Joint Ventures Sipper BK Experimental Animal Co. (Shanghai,
Supplementary information, Data S1 Materials and Methods Mice, Ad vectors and reagents Female C57BL/6 mice, 8-10 weeks of age, were purchased from Joint Ventures Sipper BK Experimental Animal Co. (Shanghai,
G-Protein Signaling. Introduction to intracellular signaling. Dr. SARRAY Sameh, Ph.D
 G-Protein Signaling Introduction to intracellular signaling Dr. SARRAY Sameh, Ph.D Cell signaling Cells communicate via extracellular signaling molecules (Hormones, growth factors and neurotransmitters
G-Protein Signaling Introduction to intracellular signaling Dr. SARRAY Sameh, Ph.D Cell signaling Cells communicate via extracellular signaling molecules (Hormones, growth factors and neurotransmitters
The Annexin V Apoptosis Assay
 The Annexin V Apoptosis Assay Development of the Annexin V Apoptosis Assay: 1990 Andree at al. found that a protein, Vascular Anticoagulant α, bound to phospholipid bilayers in a calcium dependent manner.
The Annexin V Apoptosis Assay Development of the Annexin V Apoptosis Assay: 1990 Andree at al. found that a protein, Vascular Anticoagulant α, bound to phospholipid bilayers in a calcium dependent manner.
Ikaros Null Mice Display Defects in T Cell Selection and CD4 versus CD8 Lineage Decisions
 This information is current as of February 23, 2013. Ikaros Null Mice Display Defects in T Cell Selection and CD4 versus CD8 Lineage Decisions Julie A. Urban and Susan Winandy J Immunol 2004; 173:4470-4478;
This information is current as of February 23, 2013. Ikaros Null Mice Display Defects in T Cell Selection and CD4 versus CD8 Lineage Decisions Julie A. Urban and Susan Winandy J Immunol 2004; 173:4470-4478;
Rapid antigen-specific T cell enrichment (Rapid ARTE)
 Direct ex vivo characterization of human antigen-specific CD154+CD4+ T cell Rapid antigen-specific T cell enrichment (Rapid ARTE) Introduction Workflow Antigen (ag)-specific T cells play a central role
Direct ex vivo characterization of human antigen-specific CD154+CD4+ T cell Rapid antigen-specific T cell enrichment (Rapid ARTE) Introduction Workflow Antigen (ag)-specific T cells play a central role
SUPPLEMENTARY INFORMATION. Involvement of IL-21 in the epidermal hyperplasia of psoriasis
 SUPPLEMENTARY INFORMATION Involvement of IL-21 in the epidermal hyperplasia of psoriasis Roberta Caruso 1, Elisabetta Botti 2, Massimiliano Sarra 1, Maria Esposito 2, Carmine Stolfi 1, Laura Diluvio 2,
SUPPLEMENTARY INFORMATION Involvement of IL-21 in the epidermal hyperplasia of psoriasis Roberta Caruso 1, Elisabetta Botti 2, Massimiliano Sarra 1, Maria Esposito 2, Carmine Stolfi 1, Laura Diluvio 2,
Data Sheet PD-1 / NFAT Reporter - Jurkat Cell Line Catalog #: 60535
 Data Sheet PD-1 / NFAT Reporter - Jurkat Cell Line Catalog #: 60535 Product Description Recombinant Jurkat T cell expressing firefly luciferase gene under the control of NFAT response elements with constitutive
Data Sheet PD-1 / NFAT Reporter - Jurkat Cell Line Catalog #: 60535 Product Description Recombinant Jurkat T cell expressing firefly luciferase gene under the control of NFAT response elements with constitutive
Supporting Information Table of Contents
 Supporting Information Table of Contents Supporting Information Figure 1 Page 2 Supporting Information Figure 2 Page 4 Supporting Information Figure 3 Page 5 Supporting Information Figure 4 Page 6 Supporting
Supporting Information Table of Contents Supporting Information Figure 1 Page 2 Supporting Information Figure 2 Page 4 Supporting Information Figure 3 Page 5 Supporting Information Figure 4 Page 6 Supporting
Supplementary Figure 1. Normal T lymphocyte populations in Dapk -/- mice. (a) Normal thymic development in Dapk -/- mice. Thymocytes from WT and Dapk
 Supplementary Figure 1. Normal T lymphocyte populations in Dapk -/- mice. (a) Normal thymic development in Dapk -/- mice. Thymocytes from WT and Dapk -/- mice were stained for expression of CD4 and CD8.
Supplementary Figure 1. Normal T lymphocyte populations in Dapk -/- mice. (a) Normal thymic development in Dapk -/- mice. Thymocytes from WT and Dapk -/- mice were stained for expression of CD4 and CD8.
Immunology - Lecture 2 Adaptive Immune System 1
 Immunology - Lecture 2 Adaptive Immune System 1 Book chapters: Molecules of the Adaptive Immunity 6 Adaptive Cells and Organs 7 Generation of Immune Diversity Lymphocyte Antigen Receptors - 8 CD markers
Immunology - Lecture 2 Adaptive Immune System 1 Book chapters: Molecules of the Adaptive Immunity 6 Adaptive Cells and Organs 7 Generation of Immune Diversity Lymphocyte Antigen Receptors - 8 CD markers
SEVENTH EDITION CHAPTER
 Judy Owen Jenni Punt Sharon Stranford Kuby Immunology SEVENTH EDITION CHAPTER 16 Tolerance, Autoimmunity, and Transplantation Copyright 2013 by W. H. Freeman and Company Immune tolerance: history * Some
Judy Owen Jenni Punt Sharon Stranford Kuby Immunology SEVENTH EDITION CHAPTER 16 Tolerance, Autoimmunity, and Transplantation Copyright 2013 by W. H. Freeman and Company Immune tolerance: history * Some
ECM1 controls T H 2 cell egress from lymph nodes through re-expression of S1P 1
 ZH, Li et al, page 1 ECM1 controls T H 2 cell egress from lymph nodes through re-expression of S1P 1 Zhenhu Li 1,4,Yuan Zhang 1,4, Zhiduo Liu 1, Xiaodong Wu 1, Yuhan Zheng 1, Zhiyun Tao 1, Kairui Mao 1,
ZH, Li et al, page 1 ECM1 controls T H 2 cell egress from lymph nodes through re-expression of S1P 1 Zhenhu Li 1,4,Yuan Zhang 1,4, Zhiduo Liu 1, Xiaodong Wu 1, Yuhan Zheng 1, Zhiyun Tao 1, Kairui Mao 1,
S-NITROSOGLUTATHIONE MODULATES CXCR4 AND ICOS EXPRESSION
 CELLULAR & MOLECULAR BIOLOGY LETTERS Volume 11, (2006) pp 30 36 http://www.cmbl.org.pl Received: 01 August 2005 Revised form accepted: 17 November 2005 DOI: 10.2478/s11658-006-0003-9 2006 by the University
CELLULAR & MOLECULAR BIOLOGY LETTERS Volume 11, (2006) pp 30 36 http://www.cmbl.org.pl Received: 01 August 2005 Revised form accepted: 17 November 2005 DOI: 10.2478/s11658-006-0003-9 2006 by the University
Protocol for Gene Transfection & Western Blotting
 The schedule and the manual of basic techniques for cell culture Advanced Protocol for Gene Transfection & Western Blotting Schedule Day 1 26/07/2008 Transfection Day 3 28/07/2008 Cell lysis Immunoprecipitation
The schedule and the manual of basic techniques for cell culture Advanced Protocol for Gene Transfection & Western Blotting Schedule Day 1 26/07/2008 Transfection Day 3 28/07/2008 Cell lysis Immunoprecipitation
Received 30 January 2006/Returned for modification 28 February 2006/Accepted 24 August 2006
 MOLECULAR AND CELLULAR BIOLOGY, Nov. 2006, p. 8655 8665 Vol. 26, No. 22 0270-7306/06/$08.00 0 doi:10.1128/mcb.00168-06 Copyright 2006, American Society for Microbiology. All Rights Reserved. Lck Regulates
MOLECULAR AND CELLULAR BIOLOGY, Nov. 2006, p. 8655 8665 Vol. 26, No. 22 0270-7306/06/$08.00 0 doi:10.1128/mcb.00168-06 Copyright 2006, American Society for Microbiology. All Rights Reserved. Lck Regulates
Supplemental Data Macrophage Migration Inhibitory Factor MIF Interferes with the Rb-E2F Pathway
 Supplemental Data Macrophage Migration Inhibitory Factor MIF Interferes with the Rb-E2F Pathway S1 Oleksi Petrenko and Ute M. Moll Figure S1. MIF-Deficient Cells Have Reduced Transforming Ability (A) Soft
Supplemental Data Macrophage Migration Inhibitory Factor MIF Interferes with the Rb-E2F Pathway S1 Oleksi Petrenko and Ute M. Moll Figure S1. MIF-Deficient Cells Have Reduced Transforming Ability (A) Soft
The Adaptive Immune Response. T-cells
 The Adaptive Immune Response T-cells T Lymphocytes T lymphocytes develop from precursors in the thymus. Mature T cells are found in the blood, where they constitute 60% to 70% of lymphocytes, and in T-cell
The Adaptive Immune Response T-cells T Lymphocytes T lymphocytes develop from precursors in the thymus. Mature T cells are found in the blood, where they constitute 60% to 70% of lymphocytes, and in T-cell
