Chapter III. Regulation of viral late genes by a baculovirus-encoded mirna
|
|
|
- Sophie Wilkerson
- 5 years ago
- Views:
Transcription
1 Chapter III Regulation of viral late genes by a baculovirus-encoded mirna
2 3.1 Abstract In the last decade, mirnas have emerged as fine tuners of gene expression in various biological processes including host-pathogen interaction. Apart from the role of host encoded mirnas in host-virus interaction, recent studies have also indicated the key role of virus-encoded mirnas in the regulation of host defense responses. In the present study, I have reported a BmNPV-encoded mirna, bmnpv-mir-3 that regulates the expression of its own essential late genes including the basic DNA binding protein (P6.9), vital for the late stage of viral infection in the host, B. mori. I have shown through cell culture and in vivo experiments, the role of bmnpv-mir-3 in the infection cycle of BmNPV. Results show that bmnpv-mir-3 is likely transcribed by the host RNA polymerase, expressed during early stage of infection, and negatively regulates the expression of P6.9 and other late genes. Expression of bmnpv-mir-3 targets were upregulated upon blocking of bmnpv-mir-3 by LNA, whereas overexpression of bmnpv-mir-3 resulted in decreased expression of these targets. A remarkable enhancement and reduction in the viral loads were observed upon blocking and overexpression of bmnpv-mir-3, respectively. Furthermore, I have also assessed the host immune response using one of the lepidopteran-specific antimicrobial proteins, Gloverin 1, upon blocking and overexpression of bmnpvmir-3. All these results together; clearly imply that bmnpv-mir-3 mediated controlled regulation of BmNPV late genes in the early stage of infection help BmNPV to escape the early immune responses from the host.
3 3.2 Introduction Intimate relationship of viruses with their host cells provides unique insights into cellular function. As overcoming of host defense is essential for virus survival, it is not surprising that most, if not all, viruses encode effectors for immune evasion. mirnas are widely used as modulators of host gene regulation by DNA viruses. The small size of mirna makes them unnoticeable for the host immune machinery (Skalsky and Cullen 2010). Though viral mirnas have shown various mechanisms of over-powering the host system, initially, it was assumed that the purpose of the virus to produce mirna is mainly to counterattack the induced expression of the host genes, but many recent studies have suggested that the viral mirna-mediated regulation of viral genes, thus is vital for virus in maintaining persistent infection in the host (Skalsky and Cullen 2010; Grundhoff and Sullivan 2011). Viral-encoded mirnas based on their functions and sequence homology with host mirnas, can be broadly divided into three groups, 1) viral mirnas targeting the host genes by mimicking the host mirnas (Gottwein et al. 2007; Skalsky et al. 2007; Boss et al. 2011; Lin et al. 2011; Zhao et al. 2011; Kincaid et al. 2012), 2) novel viral mirnas, which do not have significant sequence similarity with host mirnas, regulating host genes (Samols et al. 2007; Stern-Ginossar et al. 2007; Choy et al. 2008; Xia et al. 2008; Nachmani et al. 2009; Gottwein and Cullen 2010; Lei et al. 2010), and 3) viral mirnas, regulating their own genes (Cantalupo et al. 2005; Sullivan et al. 2005; Grey et al. 2007; Hussain et al. 2008; Murphy et al. 2008; Seo et al. 2009; Sullivan et al. 2009; Skalsky and Cullen 2010; Bauman et al. 2011; Chen et al.
4 2011; Lin et al. 2011). Regulation of virus genes by viral-derived mirnas suggests a critical role for the viral mirnas in establishing virus infection, through evading the immune response by lowering viral protein level and consequently the antigenicity (Skalsky and Cullen 2010; Grundhoff and Sullivan 2011). After their discovery in 2004, understanding viral-encoded mirnas functions has been subject of intensive research. Identification of viral targets of viral-encoded mirnas came as one would expect, if one strand is producing mirna and opposite strand would give mrna upon transcription, then this mrna is obviously the natural target of this mirna. In mammalian systems, the studies of autoregulation of viral genes are mainly confined to controlled expression of the early expressing genes to increase latency inside the host cell (Cantalupo et al. 2005; Sullivan et al. 2005; Murphy et al. 2008; Seo et al. 2009; Sullivan et al. 2009; Bauman et al. 2011; Chen et al. 2011) but in insects, these studies are still in preliminary stage (Hussain et al. 2008; Asgari 2011). Here, for the first time I demonstrate that an insect virus, BmNPV, encodes a mirna (bmnpv-mir-3) to regulate the expression of its own late genes, during early stage of infection to avoid hindrance by the host immune barriers. Baculoviruses have a very distinctive profile of gene expression ensuring their infection and genome replication in specific hosts. Orchestrated regulation of baculoviral genes is very crucial for their temporal and spatial expression during the infection. Studies carried out till date have shown how baculoviruses genes are expressed and what promoter they utilize, in various
5 classes of baculoviruses (Ono et al. 2012), but possibility of mirna-mediated gene regulation has not yet been demonstrated. Annually, cocoon crop of the domesticated silkmoth gets greatly affected by BmNPV infection at the larval stage (Watanabe 1986; Watanabe 2002; Rahman and Gopinathan 2004). BmNPV is a dsdna genome containing B. mori specific baculovirus, which efficiently modulates the host gene expression and utilize the host machinery to produce enormous numbers of viral progeny (GF. 2008). In this study, using computational analysis I predicted the target sites of BmNPV-encoded mirna, bmnpv-mir-3, on many viral late expressing genes, including DNA binding protein, P6.9 (Table 3.2). In baculoviruses, P6.9 is a conserved DNA binding protein (van Oers and Vlak 2007), which is essential for the infectious virion formation inside the host insect (Maeda et al. 1991; Wang et al. 2010a). Phosphorylation and dephosphorylation of arginine-serine residues, present in the P6.9, play an important role in release and packaging of BmNPV DNA into nucleocapsid, respectively (Wang et al. 2010a; Liu et al. 2012). P6.9 similar to protamine-like proteins in other viruses (Mirza and Weber 1982; Borca et al. 1996) helps viral DNA to get condensed and incorporated into the small nucleocapsid during packaging of viral DNA inside the nucleus of the host cell. However, expression of other viral target genes of the bmnpv-mir-3 is required in the late stage of virus infection, mostly for transcription of viral late genes, viral assembly and liquefaction of the host insect (GF. 2008).
6 By performing the experiments in cell culture as well as in vivo, I have shown that bmnpv-mir-3 negatively regulates the expression of BmNPV P6.9 and other late genes. Effect of bmnpv-mir-3-medited regulation of viral late genes was also studied on BmNPV proliferation. Additionally, I tried to study transcriptional regulation of bmnpv-mir-3. Further, infectivity of viral OBs upon depletion of P6.9 and blocking of bmnpv-mir-3 was also determined. The results of this study suggest that bmnpv-mir-3-mediated negative regulation of viral late genes, hence maintaining lower viral load, is one of the important mechanisms of BmNPV for escaping the host defense in the early stage of infection.
7 3.3 Materials and Methods In silico prediction of bmnpv-mir-3 viral targets As described in the chapter-i, in silico analysis was done for target prediction of bmnpv-mir-3 on BmNPV genes, by considering various parameters; seed region pairing, minimum free energy and 3 -end pairing (for details of the viral targets of BmNPV-encoded mirnas please refer appendix-ii). I found a perfect complementary match of bmnpv-mir-3 on the 3 UTR of BmNPV P6.9 gene (GenBank accession no: M63416), which was reconfirmed by employing RNAhybrid program (Rehmsmeier et al. 2004). Additionally, the binding site of bmnpv-mir-3 on other BmNPV late genes were also analyzed using RNAhybrid program by taking the minimum free energy of the mirna-mrna duplex less than or equal to - 18kcal/mol (details of the predicted targets are provided in Table 3.2) Luciferase assay To generate the luciferase construct for bmnpv-mir-3 target validation by luciferase assay, P6.9 3 UTR region consisting the predicted binding site of bmnpv-mir-3 was cloned downstream of firefly luciferase in pmirglo vector (Promega ). PmirGLO vector was double digested with SacI and XhoI and ligated with P6.9 3 UTR fragment, which was also digested with the same pair of restriction enzymes that harbors bmnpv-mir-3 binding site with some flanking nucleotides from both the ends (primer sequences are given in the Table 3.1). For negative control, Ran 3 UTR
8 was cloned in pmirglo. Sequences and orientation of cloned fragments were confirmed by sequencing. HeLa cells were maintained in DMEM (Thermo scientific ) containing 2 mm L-glutamine and 10% FBS (Invitrogen ) at 37 C in 5% CO2. Around 5X10^4 cells were plated in 96-well plate, a day prior to transfection. The cells were first transfected with 250ng of luciferase construct and then after 7 hours, 200 nm of bmnpv-mir-3 duplex. Luciferase construct and mirna duplex transfections were performed using TransIT and TransIT-siQUEST transfection reagents respectively from Mirus Bio. 48 hours post bmnpv-mir-3 transfection, cells were assayed for luciferase activity using Dual Luciferase Reporter Assay System (Promega ) according to manufacturer s instructions. Luciferase activity was normalized against luciferase vector without insert (pmirglo) as well as containing an unrelated insert (pmirglo-ran). Assays were performed independently three times in triplicate for each sample B. mori cells and transfections B. mori ovary derived BmN cells were maintained in TC-100 insect medium (Sigma ) supplemented with 10% FBS at 25 C. Around 10^5 cells were seeded in 12-well plate overnight prior to transfection. Overexpression of bmnpv-mir-3 was achieved by transfecting the cells with one picomole perfect duplex of bmnpv-mir-3 (sense 5 GAAAGCCAAACGAGGGCAGGCG 3 and antisense 5 CCUGCCCUCGUUUGGCUUUCCG). As negative control, sirna against GFP (Sense 5 GCUACCUGUUCCAUGGCCATT 3 and antisense 5
9 TGGCCATGGAACAGGTAGCTT 3 ) was used. All transfections were done using TransIT-siQUEST (Mirus Bio ). After 4 days, total RNA was isolated from these cells by TRIzol method (Invitrogen ) BmNPV infection of B. mori cells For analyzing the bmnpv-mir-3 effect on its predicted viral targets, BmNPV infection was performed in BmN cells which were subsequently transfected with bmnpv-mir-3 duplex. Initially, around 10^6 BmN cells were cultured in T25-flask and then transfected with 5µg of BmNPV DNA using TransIT (Mirus Bio ). After 4 days, when the medium turned turbid due to the release of viral bodies, culture medium was collected and used for further infection experiments. 100µl of the virus containing supernatant was used for the infecting BmN cells, which were seeded 24 hours prior. These cells were incubated at 25 C for 5 hours and subsequently, fresh medium was added after removing the virus inoculum containing medium In vivo injections of mirna duplex and its antagomir in larvae BmNPV infected (20,000 OBs/larva) B. mori larvae (5 th instar, 2 nd day) were injected with 100 pmoles of specific inhibitor of bmnpv-mir-3 (LNA-3: 5 CGCCTGCCCTCGTTTGGCTTTC 3 ) in the fourth abdominal leg. For negative controls, larvae were injected with nonspecific inhibitor (random-lna: 5 - CATGAGCTGACCGGAACAGCT-3 ). LNAs used in this study were a mixture of
10 modified DNA/RNA oligonucleotides, which contain phosphotiorate backbone with selected nucleotides methylated (Exiqon ). Overexpression of bmnpv-mir-3 was achieved by injecting 100 pmoles of perfect duplexes of bmnpv-mir-3 at the same time point as done for LNAs. In this case, sigfp administered larvae were used as negative control. LNA and bmnpv-mir-3 administered larvae were scarified after two and three days of administration, respectively, and fat body tissues were extracted and used for RNA isolation. LNA and bmnpv-mir-3 injection experiments were repeated thrice independently on different sets of larvae RNAi To perform RNAi experiments, dsrna was synthesized using Megascript in vitro transcription kit (Ambion ) according to the manufacturer s instructions. Briefly, all selected viral late genes including P6.9 were amplified using specific primers by RT-PCR (Table 3.1) and subsequently cloned into pcrii-topo vector (Invitrogen ). Templates for in vitro transcription were generated by amplifying insert containing pcrii-topo vectors using M13 primers. 1µg DNA template was used to transcribe sense and antisense RNA strands using T7 and SP6 RNA polymerases (Ambion ) respectively. Equimolar concentration of both the RNA strands was annealed and their size was determined by running them on 2% agarose gel with their respective DNA templates. As a negative control for RNAi, dsrna for GFP was prepared as described previously for other viral genes. 5th instar two days old larvae were administered with 20µg dsrna of P6.9, Fusolin, Chitinase k1, Very late factor-1
11 and GFP, at their fourth abdominal leg. After 4 days of dsrna injection, fat body and hemolymph tissues of the injected larvae were extracted and total RNA was isolated RNA isolation and RT-qPCR Total RNA was isolated from BmNPV infected and uninfected fat body tissues of B. mori larvae, using TRIzol reagent (Invitrogen ) according to the manufacturer s instructions. Subsequently, DNA contamination was removed by treating with DNase I (Invitrogen ). 2µg RNA and 5µM oligo-dt primer were used to synthesize cdna using SuperScript III First-Strand Synthesis System (Invitrogen ) in 20µl reaction mixture. 1µl of reverse transcription product was used to perform qpcramplification using specific primers to BmNPV late genes, as previously described (Chapter-II-Materials and Methods 2.3.2) (Singh et al. 2012), on RT-7500 system (Applied Biosystems ) Northern blotting 100µg and 20µg of RNA derived from BmNPV infected B. mori larvae and cells, respectively, were resolved on a 15% denaturing polyacrylamide gel and subsequently transferred to nylon-hybond+ membrane (Amersham) using Semi-dry transfer cell (Trans-blot SD Bio-Rad). Hybridization was carried out using bmnpvmir-3 specific radiolabelled DNA probes as previously described (Chapter-II- Materials and Methods 2.3.9). Sequences of the probes are provided in Table S rrna probes were used to confirm the equal loading of RNA.
12 3.3.9 Semiquantitative PCR Transcript levels of bmnpv-mir-3 viral targets and BmNPV load were determined by semiquantitative PCR using cdna and DNA respectively. 1µl of cdna was used to perform PCR for examining the transcript levels of viral target genes, while 100ng DNA was used to estimate the viral load by PCR. BmNPV DNA was isolated from different BmNPV infected B. mori fat body tissues using DNeasy Blood & Tissue Kit (Qiagen), according to the manufacturers instructions. Reverse transcription was done as previously described in Materials and Methods section of Chapter II (2.3.2). 20µl PCR reaction included 1µl of each 5µM primer, 1µl of cdna or DNA, 10µl of EmeraldAmp GT PCR Master Mix (Takara) and water to make up the reaction volume. PCR was performed using following conditions; initial denaturation at 95 C for 2 minutes, 30 cycles at 95 C for 15 seconds, 60 C or 62 C for 30 seconds, 72 C for 1 minute and a final elongation at 72 C for 7 minutes. The resulted PCR products were resolved on 2% agarose gel, stained with EtBr and visualized under UV light Viral load determination by scoring OBs Hemolymph was collected from BmNPV infected dsrna and LNA administered larvae. Viral OBs were recovered by centrifuging hemolymph at 2000rpm for 2 min. OB pellets were washed in 1X PBS and dissolved in the same. Scoring of OBs was done using an automated cell counter, Countess (Invitrogen ). The count was also verified by hemocytometer Statistical analysis
13 Test of significance was performed to measure the significance difference between the groups. Unpaired t-test was done using online GraphPad software ( Mean with a standard deviation from the three independent experiments was used as input. P-values, indicating significant difference were listed in the respective figure legends. Table 3.1 Sequences of primers and probes Gene Primers for RT-PCR, qpcr and RNAi BmNPV p6.9 Forward 5 GCGCCGTTCTTCAGCCGGTA 3 Reverse 5 GGGCAGGCGACGAAATCGCT 3' BmNPV p40 Forward 5 TCGCGTCGCTTTCCGCACAT 3 Reverse 5 GGCGAATTGACCGCGTTGGC 3 BmNPV p95 Forward 5 ACAACGACGCCATCTTTGCTCA 3 Reverse 5 CCGGTCAGCGTGTCGCTCAA 3 BmNPV fusolin Forward 5 TGGCGTGCACACAAATCAAATTGC 3 BmNPV very late factor 1 (vlf1) BmNPV Chitinase k1 Reverse 5 AAATGGGCTGCGCGGAGAGG 3 Forward 5 AAAATTCAATCCACTCCCCGGTTCG 3 Reverse 5 TCCAGGTGAACATCGTCTACCGAGC 3 Forward 5 CGGCTGGACCGGCGTAACAA 3 Reverse 5 CCCAAGCAAACAAGCCGCCC 3 BmNPV bp15 Forward 5 GGGCCAAGCTGCTGTACCGT 3 Reverse 5 TGAGGCTGCAAGCCCAAAGCA 3 BmNPV lef-8 Forward 5 TGTCACCACGTTGGCCATTCTGT 3 Reverse 5 ACTACGTCCGTCATCGTAAAGCGA 3 BmNPV lef-9 Forward 5 CTCAACACGGCCTCGTTCTA 3 Reverse 5 CCAGTCGGTTGATGACTCGT 3 BmNPV ie-1 Forward 5 GTCCGTTGTCCGTGTGCGCT 3 B. mori Gloverin-1 B. mori 18S rrna PmirGLO vector Reverse 5 CGGCGCCGTTGGGATTTGTG 3 Forward 5 AGGCTTACGGCACCAGAGTA 3 Reverse 5 TAAACCGACATCAGGCCTTC 3 Forward 5 CGATCCGCCGACGTTACTACA 3 Reverse 5 GTCCGGGCCTGGTGAGATTT 3 Primers for luciferase construct preparation Forward: 5 TGACCGGCAAGTTGGACGCC 3 Reverse: 5 GGCCGCCCCAAGGGGTTATG 3
14 BmNPV p6.9 Forward 5 ACACGCTAC GAGCTCGACATTCCACACATCCGACA 3 Reverse 5 CAACCATACCTCGAG TACGCGCAAAAAGAAAATCA 3 B. mori Ran Forward 5 TGATGAGCTCGCCTGCCCTTCTGCCACCAG 3 Reverse 5 ACTGCTCGAGACGCTACACTGAACACATTTGCATGA 3 DNA probes used in Northern blotting bmnpv-mir-3 5 CGCCTGCCCTCGTTTGGCTTTC 3 5S rrna 5 GTTGCTTGACTTCGGTGATCGGACGAGAACCGGTGTATTCAACAT GGTATGGACG Results and discussion Prediction of bmnpv-mir-3 viral targets In my previous report (Singh et al. 2010), I found that bmnpv-mir-3 has a perfect binding site on the 3 UTR of BmNPV P6.9 mrna, which transcribe from opposite strand of bmnpv-mir-3 in the viral genome (Figure 3.1). P6.9 is a basic DNAbinding protein known to be a vital component of viral nucleocapsid and is expressed late in the infection (Maeda et al. 1991; van Oers and Vlak 2007). P6.9 is a small protein consisting of 65 amino acids. In majority of the baculoviruses P6.9 protein contains only 3 arginine repeats, while BmNPV has 4 arginine repeats, which are responsible for the neutralization of viral DNA and helps virus to pack its DNA in a very stringent manner inside the capsid (Maeda et al. 1991). It has also been shown in AcMNPV that the phosphorylation of serine-threonine residues present in P6.9 causes release of viral DNA from its capsid (Wang et al. 2010a; Liu et al. 2012).
15 Relative luciferase activity (%) bmnpv-mir-3 P6.9 mrna 3' GCGGACGGGAGCAAACCGAAAG 5' 5' CGCCTGCCCTCGTTTGGCTTTC 3' UTR * bmnpv-mir-3 translationally represses BmNPV P6.9
16 In order to validate the interaction between bmnpv-mir-3 and P6.9, luciferase reporter vector was constructed by inserting the predicted binding site of bmnpvmir-3 on P6.9 with some flaking sequences downstream of the firefly luciferase gene. Renilla luciferase was used as an endogenous control. Vectors were transfected into HeLa cells, and after 24 hours, bmnpv-mir-3 duplexes were subsequently added. More than 50% reduction was observed in luciferase activity for P6.9 (Figure 3.2), in presence of bmnpv-mir-3. To confirm that the inhibitory effect on luciferase activity was mainly because of bmnpv-mir-3 binding on its target site on P6.9, I used an unrelated BmNPV mirna (bmnpv-mir-1) and a another reporter vector harbouring sequence without bmnpv-mir-3 binding sites (pmirglo-ran) as negative controls, and compared luciferase activity. There was no alteration in luciferase activity compared to controls in the presence of the unrelated mirna (Figure 3.2). Similarly, no significant effect was observed in luciferase levels of the reporter vector that does not contain bmnpv-mir-3 binding site, upon introduction of bmnpv-mir-3 duplexes. Results of luciferase assays suggest that bmnpv-mir-3 inhibits expression of P bmnpv-mir-3 expresses heavily in the early stage of virus infection BmNPV generally takes hours to complete the infection cycle in B. mori cells but in larvae it is difficult to find out the accurate duration of the virus infection cycle due to lateral infection. In order to examine the bmnpv-mir-3 expression with BmNPV infection in B. mori larvae, the fat body tissues were collected from
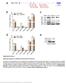
17 BmNPV infected larvae at different time points, starting from 12 hours post infection (hpi) to 144 hpi and subsequently Total RNA was isolated. bmnpv-mir-3 expression was determined in these RNA samples by Northern blot. Results show that bmnpv-mir-3 is expressed more in the early stage of virus infection and decreases as infection progresses (Figure 3.3A). I also analyzed its expression in virus infected midgut and silk gland tissues. A higher expression of bmnpv-mir-3 in the midgut tissues was observed till 48 hours of BmNPV infection. In the silk gland, I could not observe any expression of bmnpv-mir-3, which may be attributed to nonsusceptibility of silkglands by BmNPV infection (Barrett et al. 1998; Dourado et al. 2011; Katsuma et al. 2012). Similar expression pattern of bmnpv-mir-3 was observed in the BmNPV-infected BmN cells (Figure 3.3B). These results suggest that bmnpv-mir-3 is expressed heavily in the early stage of BmNPV infection and expression diminishes as infection progresses. A Hours post infection Infected fatbody Infected midgut Infected silkgland bmnpv-mir-3 5S rrna B bmnpv-mir-3 Hours post infection S rrna
18 3.4.4 bmnpv-mir-3 also targets other viral late genes Early expression of bmnpv-mir-3 during the viral infection, and bmnpv-mir-3- mediated negative regulation of P6.9, encouraged me to investigate the binding site of bmnpv-mir-3 on other viral late genes. I searched bmnpv-mir-3 binding sites by taking less stringent parameters of target prediction program, RNAhybrid. Analysis returned with seven additional target genes of bmnpv-mir-3, namely: p40, p95, very late factor (vlf1), fusolin, chitinase k1, bp15 and lef-8 (Table 3.2). These putative targets of bmnpv-mir-3 have diverse functions in the late stage of virus infection including transcription (lef-8), DNA recombination (vlf1), viral nucleocapsid packaging (p40 and p95), and liquefaction of virus-infected insect (fusolin and chitinase k1) (GF. 2008; Ono et al. 2012). Table 3.2 Details of the predicted bmnpv-mir-3 viral targets Accession No. Target gene Function Binding site on mrna *MFE (Kcal/mol) M63416 P6.9 (Ac100) Viral DNA packaging and (3 UTR) releasing of viral DNA D14468 P40 (Ac 101) Structural protein of occluded virion (3 UTR-CDS) U83330 P95 (Ac 116) Late transcription factor (CDS) U55072 BP15 (Ac87) Viral capsid protein (CDS) AB Lef-8 (Ac50) Late transcription (Subunit of (CDS) viral RNA polymerase) U55071 Fusolin (Ac64) Peroral infectivity (3 UTR) -18.5
19 AF Very late factor 1 (vlf1) (Ac77) AY Chitinase k1 (Ac126) Capsid assembly and late transcription (CDS), (CDS) -23.9, Liquefaction of insect (CDS) *MFE: minimum free energy
20 Relative expression of P Inhibition of bmnpv-mir-3 results in upregulation of bmnpv-mir-3 targets To demonstrate the interaction between bmnpv-mir-3 and its predicted viral targets in vivo, bmnpvmir-3 endogenous expression was blocked by using its antagomir, LNA-3. Random-LNA was taken as negative control. At 24 hpi, 100 pmoles of LNA-3 and random-lna were administered in 2 days old 5 th instar larvae, and the fat body tissues were collected after 48 hours from these larvae. Expression analysis of all the viral targets was done by RT-PCR. I observed a substantial increase in the transcript level of all the viral targets in LNA-3 treated samples as compared to random-lna treated samples, except for bp15 and lef-8 (Figure 3.4A). Expression of P6.9 was further analyzed by RT-qPCR and noticed more than 25 folds increase in the transcript level of P6.9 upon inhibition of bmnpv-mir-3 by LNA-3, but the expression remained unchanged in the random-lna treated samples as compared to control (Figure 3.4B). A B P6.9 P * P95 30 Vlf Fusolin Chitinase k BP15 Lef-8 0 IFB IFB + LNA-3 IFB + Random- LNA 18S rrna
21 In order to examine the temporal expression of bmnpv-mir-3 viral targets upon blocking of bmnpv-mir-3, I administered LNA-3 to the B. mori larvae to block the expression of bmnpv-mir-3. Random-LNA was administered to the larvae as negative control. Fat body tissues were collected from the LNA administrated larvae at 24, 48, 72 and 96 hpi. Expression of all the viral targets except lef-8 and bp15, which remained unchanged upon blocking of bmnpv-mir-3, was analyzed by RT-PCR using RNA extracted from the experimental larvae. A gradual increase in the transcript levels of all the viral targets was observed upon blocking of bmnpv-mir-3 by LNA-3 as compared to control and the random-lna administered samples (Figure 3.5). Precisely, P6.9, P95 and chitinase k1 were found upregulated as early as 24 hours post bmnpv-mir-3 blocking by LNA-3, and their expression gradually increased along with the infection progress. However, expression of other bmnpv-mir-3 viral targets was increased abruptly at 72 hours after bmnpv-mir-3 inhibition (Figure 3.5). An enhancement in the bmnpv-mir-3 viral target expression upon blocking of bmnpv-mir-3 clearly implies that bmnpv-mir-3 indeed negatively regulates BmNPV late genes.
22 Hours post infection IFB IFB + LNA-3 IFB + Random-LNA P6.9 P40 P95 Vlf-1 Fusolin Chitinase k1 18S rrna Time course expression analysis of bmnpv-mir-3 viral targets upon BmNPV infection In order to verify the expression of predicted bmnpv-mir-3 viral targets upon BmNPV infection and make sure all these target genes are late expressing genes, I isolated RNA from BmNPV infected fat body tissues of B. mori at different time points (24-96 hpi). Expression analysis of these target genes was done using RTqPCR. I observed that the predicted viral targets began to express at 48 hpi and their expression exponentially increases as infection progresses (Figure 3.6). In fact, RT-qPCR results showed that the expression of these late genes goes more than 10,000 folds at 96 hpi as compared to 24 hpi (Figure 3.6), which suggests these genes are indeed expressed in the late stage of BmNPV infection.
23 Relative transcript levels hpi 48 hpi 72 hpi 96 hpi Overexpression of bmnpv-mir-3 in BmN cells suppresses expression of its viral targets BmNPV expresses bmnpv-mir-3 in the early stage of infection, and later its expression diminishes, while its viral targets are upregulated. In order to directly check the negative regulation of viral late genes by bmnpv-mir-3 in the late stage of infection, I overexpressed bmnpv-mir-3 by transfecting its perfect duplexes into BmNPV-infected BmN cells. SiRNA against GFP (sigfp) was used
24 as a negative control for all the transfections. After 3 days of mirna transfection, total RNA was isolated from the transfected cells and used for RT-PCR analysis. I observed a substantial reduction in the expression of all the bmnpv-mir-3 viral targets in the bmnpv-mir-3 transfected cells compared to sigfp transfected cells (Figure 3.7). Taken together all the above results, I conclude that bmnpv-mir-3 negatively regulates expression of the virus late genes. P6.9 P40 P95 Vlf-1 Fusolin Chitinase k1 18S rrna
25 Relative BmNPV DNA load bmnpv-mir-3 overexpression effectively decreases the viral load in B. mori cells and larvae Overexpression of bmnpv-mir-3 suppresses virus late gene expression, and to correlate this effect directly to the virus infection, I looked for the virus titer in the bmnpv-mir-3 transfected cells. For determining the viral titer, quantification of the viral DNA was done by semiquantitative-pcr using BmNPV early gene, ie-1. I found a marked decrease in the viral titer of the bmnpv-mir-3 transfected cells compared to that of sigfp treated cells (Figure 3.8A). A similar analysis was also done in B. mori larvae. bmnpv-mir-3 duplexes were administered into the larvae, and DNA was isolated from fat body tissues, harvested at 12, 24, 48, 72 and 96 hpi. A measurable reduction in the viral load was observed upon overexpression of bmnpv-mir-3 in larvae, as determined by qpcr using ie-1 specific primers (Figure 3.8B), suggests bmnpv-mir-3-mediated regulation has a negative effect on BmNPV proliferation. A B 0 12 hpi 24 hpi 48 hpi 72 hpi 96 hpi BmNPV ie-1 gene S rrna * * * *
26
27 3.4.9 BmNPV load increases upon inhibition of bmnpv-mir-3 To examine the role of bmnpv-mir-3 in BmNPV life cycle and its multiplication, I measured the BmNPV titer upon blocking of bmnpv-mir-3 in the fat body tissue of BmNPV infected larvae. For blocking bmnpv-mir-3, BmNPV-infected larvae were injected with LNA-3 and random- LNA was used as negative control. Ie-1-specific primers were used for the estimation of viral DNA levels, as determined by semiquantitative and quantitative-pcr. About 25 folds increase was observed in the viral load of the LNA-3 administered larvae as compared to the larvae injected with random-lna (Figure 3.9A and 3.9B). Propagation of BmNPV infection was examined by scoring viral OBs from the LNA administered larvae, and a significant increase was noticed in the viral load of these larvae compared to that of control (Figure 3.9C). These results indicate the plausible role of bmnpv-mir-3 in maintaining the virus infection inside the host in a controlled manner by regulating its own genes.
28 Relative BmNPV DNA load Viral titre PIB/ml (x10 6 ) A BmNPV ie-1 gene 18S rrna B * C * IFB IFB + LNA-3 0 BmNPV BmNPV + LNA-3 BmNPV + Random-LNA
29 bmnpv-mir-3-mediated negative regulation of viral late genes in early stage of infection helps virus to avoid immune response of the host Insects posses a very potent innate immune system to prevent various pathogenic infections but, unlike vertebrate they lack memory based adaptive immune system. Innate immunity in lepidopterons is well studied against bacterial pathogens, but characterization of immune pathway(s) or antiviral marker(s) against baculovirus infections is still lacking. Recent reports have shown that Gloverins, the lepidoptera-specific antimicrobial proteins (AMPs), are often found upregulated upon baculovirus infection in various lepidopterans including B. mori (Bao et al. 2009; Bao et al. 2010; Wang et al. 2010b; Moreno-Habel et al. 2012). In order to verify that virus load induces host immune response and its controlled proliferation can avoid the recognition by the host, I set out to study the immune response using expression of Gloverin-1 as a marker of induced immune response upon BmNPV infection in B. mori larvae. To reconfirm the Gloverin-1 induction upon BmNPV infection, I first examined expression of Gloverin-1 upon BmNPV infection in fat body RNA and observed remarkably higher expression of Gloverin-1 in virus infected larvae compared to uninfected ones as analyzed by RT-PCR (Figure 3.10A). To study the correlation of BmNPV load with B. mori immune response, expression of Gloverin-1 was examined in LNA-3 and bmnpv-mir-3 administered larvae, in which viral load was found affected positively and negatively, respectively. Larvae administered with Control-LNA and sigfp were used as a negative control for LNA-3 and bmnpv-mir-3 experiments, respectively. Expression of Gloverin-1
30 increases as virus load enhances upon addition of LNA-3 and inverse effect was seen when bmnvp-mir-3 was overexpressed (Figure 3.10B & C). I observed a significant increase in the expression of Gloverin1 from hpi in the LNA-3 administered larvae, although at the later stage of infection from 72 to 96hpi, Gloverin 1 expression remained unchanged Figure 3.10 B). This is possibly because in the later stage of infection all the late genes express heavily and hence blocking of bmnpv-mir-3 by LNA-3 does not cause much change in these genes expression as well as the viral load. A remarkable decrease in the Gloverin 1 expression was observed upon bmnpv-mirr-3 overexpression in larvae. However, I noticed a sharp increase in expression of Gloverin-1 at 96 hpi in bmnvp-mir-3 administered larvae, which may be attributed by degradation of bmnpv-mir-3 duplex at 96 hpi (Figure 3.10C). By considering expression of Gloverin-1 as the marker of B. mori immune response upon BmNPV infection, these results suggest that extent of BmNPV load has correlation with silkworm immune response, which increases and decreases as BmNPV load enhances and reduces in LNA-3 and bmnpv-mir-3 administered larvae, respectively.
31 Relative transcript levels of Gloverin-1 Relative transcript levels of Gloverin-1 A B Gloverin sigfp bmnpv-mir-3 18S rrna 1 0 * * * 24 hpi 48 hpi 72 hpi 96 hpi C * * 24 hpi 48 hpi 72 hpi 96 hpi Random-LNA LNA-3
32 RNAi-based knockdown of bmnpv-mir-3 viral target genes reduces BmNPV load Most of the BmNPV genes are studied for their essentiality to virus viability (Ono et al. 2012). In order to further confirm the role of bmnpv-mir-3 viral target genes in BmNPV proliferation, I performed dsrna-mediated knockdown of P6.9, P95, Fusolin and Vlf-1 in B. mori larvae. BmNPV infected larvae were injected with 20 μg of dsrna for viral late genes and GFP, which served as a negative control. Knockdown of these viral genes was confirmed by RT-PCR in the fat body tissues after 4 days of dsrna administration (Figure 3.11A, B, C & D). To analyze the knockdown effect of these viral genes on BmNPV proliferation, I scored the viral OBs in the hemolymph of knockdown larvae. A significant reduction in the number of BmNPV OBs was observed in all the selected viral late genes knockdown samples compared to GFP dsrna-injected control larvae (Figure 3.11E & F). I also noticed a reduction of BmNPV DNA levels in P6. 9 depleted larvae as examined by qpcr using ie-1 primers (Figure 3.11G). These results further provide support to essentiality of bmnpvmir-3 viral target genes for BmNPV proliferation.
33 Viral titer OBs/ml (x10 6 ) Viral titer OBs X10 6 /ml Relative BmNPV DNA load Relative expression of p6.9 Relative expression of P95 Relative expression of Vlf1 Relative expression of Fusolin A B C D * * * * E dsgfp 0 dsp6.9 dsgfp dsp95 F * * 0 dsgfp dsvlf1 G dsgfp dsgfp dsfusolin dsp dsgfp dsp * * dsgfp dsp95 dsfusolin dsvlf * Depletion of P6.9 negatively affects the viral OBs infectivity P6.9 is an essential gene of BmNPV, required for the tight packaging of viral DNA to produce proper viral bodies. In AcMNPV, it has been shown that P6.9 mutated virus is less virulent than wild type
34 Viral titer OBs/ml (x10 6 ) (Wang et al. 2010a). In order to determine the function of P6.9 in BmNPV, I checked the infectivity of viral OBs, which was derived from P6.9 knockdown larvae, by infecting them into a fresh set of larvae through feeding. A significant decrease in the viral OBs count was observed in dsp6.9 derived OB samples compared to dsgfp derived control samples (Figure 3.12). Reduction in infectivity of BmNPV OBs derived from P6.9 knockdown larvae indicates conserved function of P6.9, as reported in AcMNPV (Wang et al. 2010a) * dsgfp dsp6.9
35 Viral titer OBs/ml (x10 6 ) Blocking of bmnpv-mir-3 has no effect on secondary infection As discussed earlier, a higher viral load was observed upon inhibition of bmnpv-mir-3 in the larvae. In order to see this blocking effect on viral OBs-mediated infection, I infected fresh set of larvae with those OBs derived from LNA-3 and random-lna administered larvae, which served as negative control. Equal dose of OBs (20,000 OBs/larva) was fed to larvae and scoring of OBs was done in the hemolymph of these larvae at 72hpi. There was no observable change in the viral load of LNA-3 and random-lna derived OBs compared with that of LNA uninjected larvae (Figure 3.13) BmNPV BmNPV + LNA-3 BmNPV + Random-LNA The secondary infection analysis results suggest that knockdown of P6.9 affects the proper viral OBs formation, whereas its rescue from bmnpv-mir-3 affects only on the primary infection
36 (Figure 3.9C). All these results infer that upregulation of viral late genes, especially P6.9, does not affect secondary infection of BmNPV, while their downregulation reduces the infectivity of the virus Knockdown of viral RNA polymerase subunit, LEF-9 has no effect on bmnpv-mir-3 transcription In baculoviruses, the early gene expression is mainly governed by different host RNA polymerases [42], but how they transcribe mirnas is yet to be investigated. To address this in BmNPV, I performed knockdown of the viral RNA polymerase by generating a dsrna against one of its subunit, LEF-9 (Guarino et al. 1998), which has shown to be involved in transcription of viral late genes (Lu and Miller 1994), in the larvae. As a result of RNAi, a significant reduction in the levels of LEF-9 transcript was observed (Figure 3.14A). I also noticed downregulation of all the bmnpv-mir-3 viral targets upon knockdown of LEF-9, while expression of ie-1, an immediate early gene of BmNPV, remained unchanged (Figure 3.14B). Surprisingly, bmnpv-mir-3 levels were observed higher in LEF-9 knockdown larvae compared to the control, sigfp treated larvae, indicating that bmnpv-mir-3 is most likely transcribed by the host RNA polymerase (Figure 3.14C). Higher expression of bmnpv-mir-3 may be understood by comparing the needs of the host and viral system at any given time, so when virus RNA polymerase is not consuming the resources (here depleted by dsrna) its outcome can be seen as an enhanced expression of the host governed genes or mirnas. These results suggest that knockdown of LEF-9 negatively affects transcription of viral targets of bmnpv-mir-3, while has no effect on bmnpv-mir-3 transcription.
37 A B P6.9 LEF-9 18S rrna P40 C P95 bmnpv-mir-3 5S rrna Vlf-1 Fusolin Chitinase k1 Ie-1 18S rrna Viruses use different strategies to combat the host arsenal by producing a variety of effector molecules (Marques and Carthew 2007). Generally, structural and regulatory viral proteins helps virus to manipulate the host system for its own advantage, interestingly recent advancements in the small RNA field and their
38 functional characterization in virus, demonstrate that viral mirnas perform similar role (Skalsky and Cullen 2010; Grundhoff and Sullivan 2011; Cullen 2013). Of late, studies have revealed the function of mirna in maintaining the viral latency (Skalsky and Cullen 2010; Grundhoff and Sullivan 2011; Wu et al. 2011). These studies to an extent also explain why virus prefers mirna over protein in the latency stage. Conceptually, mirnas can contribute to combat the host immune evasion directly by suppressing the components of the host defense system and indirectly, by limiting the expression levels of viral proteins. I have elucidated the role of one of the BmNPV-encoded mirnas, bmnpvmir-1, in avoiding the host defense (Singh et al. 2012). bmnpv-mir-1 suppresses the host small RNA pathway by regulating Ran, a co-factor of Exportin-5 (Singh et al. 2012). Here, I have shown that BmNPV employs its mirna, bmnpv-mir-3, for regulating its own genes to effectively replicate inside the host, B. mori (Figure 3.15). I found an increase in the BmNPV load, when its own mirna, bmnpv-mir-3 expression was inhibited by LNA (Figure 3.9A, B & C), and vice versa when same mirna was overexpressed (Figure 8A & B). RNAi analysis of bmnpv-mir-3 viral targets also resulted in a decrease of the viral load (Figure 3.11E, F & G), suggesting that these genes are required for virus proliferation. Higher expression of bmnpv-mir-3 viral targets in the last stage of infection, when bmnpv-mir-3 expression decreases, correlates with mirna-mediated regulation of these viral late genes. In diverse classes of viruses, several studies have reported regulation of viral genes by viral-encoded mirnas, which can be grouped into two classes; first, which
39 have natural antisense targets, for example, T-antigen regulation by mir-s1 in SV40 (Sullivan et al. 2005), DNA polymerase (BALF5) regulation by mir-bart2 in Epstein Barr virus (EBV) (Pfeffer et al. 2005), and ICP0 regulation by mir-h2 in Herpes simplex viruses (HSV-1 and HSV-2) (Tang et al. 2008; Umbach et al. 2008; Tang et al. 2009; Umbach et al. 2010). The second class includes targets derived from other than antisense transcripts, and generally they are partially complementary to respective viral mirna, for example, ICP4 regulation by mir-h6 in HSV-1 (Umbach et al. 2008), LMP1 regulation by mir-bart1-5p, mirbart16, and mir-bart17-5p in EBV (Lo et al. 2007), DNA polymerase I regulation by HvAV-miR-1 in Heliothis virescens ascovirus (HvAV) (Hussain et al. 2008). In this study, I have found that bmnpv-mir-3 not only targets its natural antisense target, P6.9, but also other viral late genes. bmnpv-mir-3 expressed early in the infection as observed in Northern blot results and have target sites on the several viral late genes. This is very unusual, where a viral-encoded mirna have acquired binding site for multiple viral genes.
40 Late stage BmNPV gene expression Early stage Early stage BmNPV bmnpv-mir-3 negatively regulates expression of viral late genes in the early stage of infection Transcription Pri-miRNA(s) Drosha Pre-miRNA Exp5 Signal Recognized by host defense machinery Pre-miRNA bmnpv-mir-3 P6.9 P40 Fusolin Signal blocked Escape the recognition by host defense machinery Dicer bmnpv-mir-3 (Expresses in early stage of infection) In late stage, bmnpv-mir-3 expression is reduced or its binding site is protected X Early expressing genes Late expressing genes Signal Recognition by Host defense machinery but the host can not counterattack due to heavy viral load mirnas are an important revelation of the last decade, which have revolutionized our thinking towards so-called junk content of the genome. These tiny RNAs have been shown to play a crucial role in the diverse biological pathways. mirnas can act as oncogenes or tumor suppressors and have potential to change the fate of any cell (Esquela-Kerscher and Slack 2006; Xiao and Rajewsky 2009). In host-pathogen interactions, deregulation of either the host or the viral-encoded mirnas can severely affect their survival (Esquela-Kerscher and Slack 2006;
41 Asgari 2011; Grundhoff and Sullivan 2011). In the present study, I have shown that a baculovirus-encoded mirna regulates the early expression of its various late genes, which may otherwise be a waste of viral as well as the host resources. Late genes are mostly involved in the viral assembly, either at the structural level or as supporting factors to accomplish the virus infection. These viral factors generally remain along with the virus particles at the time of fresh viral infection, and also during the release of the virus from the host. The host defense machinery would possibily treat these factors as the signal of virus attack and hence, virus has to conceal these factors initially to escape the host defense. Although, in the later stage of virus infection these genes are expressed, but by then the host cannot counterattack by recognizing these factors due to heavy load of virus. Also, virus needs these late genes to form viral particles for horizontal infection.
42 3.5 Conclusion The present study has demonstrated viral mirna mediated regulation of viral late genes. bmnpv-mir-3 expressed in the early stage of virus infection, probably through viral RNA polymerase, and negatively regulates many important late expressing viral genes. These viral genes are mainly responsible for accomplishing virus late stage infection and producing new viral occlusion bodies. Expression analysis of Gloverin-1 in bmnpv-mir-3 blocked and overexpressed larvae suggests that higher BmNPV load induces the immune response of the B. mori. This orchestrated regulation of late genes in the early stage of virus infection helps the virus to maintain the minimal viral load in order to escape the initial recognition and first line of defense by the host. This study reveals mechanism of viral gene regulation by a viral-encoded mirnas in baculovirus, as reported in mammalianviruses.
MicroRNA sponges: competitive inhibitors of small RNAs in mammalian cells
 MicroRNA sponges: competitive inhibitors of small RNAs in mammalian cells Margaret S Ebert, Joel R Neilson & Phillip A Sharp Supplementary figures and text: Supplementary Figure 1. Effect of sponges on
MicroRNA sponges: competitive inhibitors of small RNAs in mammalian cells Margaret S Ebert, Joel R Neilson & Phillip A Sharp Supplementary figures and text: Supplementary Figure 1. Effect of sponges on
Supplementary information
 Supplementary information Human Cytomegalovirus MicroRNA mir-us4-1 Inhibits CD8 + T Cell Response by Targeting ERAP1 Sungchul Kim, Sanghyun Lee, Jinwook Shin, Youngkyun Kim, Irini Evnouchidou, Donghyun
Supplementary information Human Cytomegalovirus MicroRNA mir-us4-1 Inhibits CD8 + T Cell Response by Targeting ERAP1 Sungchul Kim, Sanghyun Lee, Jinwook Shin, Youngkyun Kim, Irini Evnouchidou, Donghyun
Supplemental Materials and Methods Plasmids and viruses Quantitative Reverse Transcription PCR Generation of molecular standard for quantitative PCR
 Supplemental Materials and Methods Plasmids and viruses To generate pseudotyped viruses, the previously described recombinant plasmids pnl4-3-δnef-gfp or pnl4-3-δ6-drgfp and a vector expressing HIV-1 X4
Supplemental Materials and Methods Plasmids and viruses To generate pseudotyped viruses, the previously described recombinant plasmids pnl4-3-δnef-gfp or pnl4-3-δ6-drgfp and a vector expressing HIV-1 X4
Soft Agar Assay. For each cell pool, 100,000 cells were resuspended in 0.35% (w/v)
 SUPPLEMENTARY MATERIAL AND METHODS Soft Agar Assay. For each cell pool, 100,000 cells were resuspended in 0.35% (w/v) top agar (LONZA, SeaKem LE Agarose cat.5004) and plated onto 0.5% (w/v) basal agar.
SUPPLEMENTARY MATERIAL AND METHODS Soft Agar Assay. For each cell pool, 100,000 cells were resuspended in 0.35% (w/v) top agar (LONZA, SeaKem LE Agarose cat.5004) and plated onto 0.5% (w/v) basal agar.
Circular RNAs (circrnas) act a stable mirna sponges
 Circular RNAs (circrnas) act a stable mirna sponges cernas compete for mirnas Ancestal mrna (+3 UTR) Pseudogene RNA (+3 UTR homolgy region) The model holds true for all RNAs that share a mirna binding
Circular RNAs (circrnas) act a stable mirna sponges cernas compete for mirnas Ancestal mrna (+3 UTR) Pseudogene RNA (+3 UTR homolgy region) The model holds true for all RNAs that share a mirna binding
Bi 8 Lecture 17. interference. Ellen Rothenberg 1 March 2016
 Bi 8 Lecture 17 REGulation by RNA interference Ellen Rothenberg 1 March 2016 Protein is not the only regulatory molecule affecting gene expression: RNA itself can be negative regulator RNA does not need
Bi 8 Lecture 17 REGulation by RNA interference Ellen Rothenberg 1 March 2016 Protein is not the only regulatory molecule affecting gene expression: RNA itself can be negative regulator RNA does not need
CHAPTER 4 RESULTS. showed that all three replicates had similar growth trends (Figure 4.1) (p<0.05; p=0.0000)
 CHAPTER 4 RESULTS 4.1 Growth Characterization of C. vulgaris 4.1.1 Optical Density Growth study of Chlorella vulgaris based on optical density at 620 nm (OD 620 ) showed that all three replicates had similar
CHAPTER 4 RESULTS 4.1 Growth Characterization of C. vulgaris 4.1.1 Optical Density Growth study of Chlorella vulgaris based on optical density at 620 nm (OD 620 ) showed that all three replicates had similar
Supplementary Figure 1. SC35M polymerase activity in the presence of Bat or SC35M NP encoded from the phw2000 rescue plasmid.
 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 Supplementary Figure 1. SC35M polymerase activity in the presence of Bat or SC35M NP encoded from the phw2000 rescue plasmid. HEK293T
1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 Supplementary Figure 1. SC35M polymerase activity in the presence of Bat or SC35M NP encoded from the phw2000 rescue plasmid. HEK293T
HEK293FT cells were transiently transfected with reporters, N3-ICD construct and
 Supplementary Information Luciferase reporter assay HEK293FT cells were transiently transfected with reporters, N3-ICD construct and increased amounts of wild type or kinase inactive EGFR. Transfections
Supplementary Information Luciferase reporter assay HEK293FT cells were transiently transfected with reporters, N3-ICD construct and increased amounts of wild type or kinase inactive EGFR. Transfections
Phosphate buffered saline (PBS) for washing the cells TE buffer (nuclease-free) ph 7.5 for use with the PrimePCR Reverse Transcription Control Assay
 Catalog # Description 172-5080 SingleShot Cell Lysis Kit, 100 x 50 µl reactions 172-5081 SingleShot Cell Lysis Kit, 500 x 50 µl reactions For research purposes only. Introduction The SingleShot Cell Lysis
Catalog # Description 172-5080 SingleShot Cell Lysis Kit, 100 x 50 µl reactions 172-5081 SingleShot Cell Lysis Kit, 500 x 50 µl reactions For research purposes only. Introduction The SingleShot Cell Lysis
~Lentivirus production~
 ~Lentivirus production~ May 30, 2008 RNAi core R&D group member Lentivirus Production Session Lentivirus!!! Is it health threatening to lab technician? What s so good about this RNAi library? How to produce
~Lentivirus production~ May 30, 2008 RNAi core R&D group member Lentivirus Production Session Lentivirus!!! Is it health threatening to lab technician? What s so good about this RNAi library? How to produce
7.012 Quiz 3 Answers
 MIT Biology Department 7.012: Introductory Biology - Fall 2004 Instructors: Professor Eric Lander, Professor Robert A. Weinberg, Dr. Claudette Gardel Friday 11/12/04 7.012 Quiz 3 Answers A > 85 B 72-84
MIT Biology Department 7.012: Introductory Biology - Fall 2004 Instructors: Professor Eric Lander, Professor Robert A. Weinberg, Dr. Claudette Gardel Friday 11/12/04 7.012 Quiz 3 Answers A > 85 B 72-84
Doctoral Degree Program in Marine Biotechnology, College of Marine Sciences, Doctoral Degree Program in Marine Biotechnology, Academia Sinica, Taipei,
 Cyclooxygenase 2 facilitates dengue virus replication and serves as a potential target for developing antiviral agents Chun-Kuang Lin 1,2, Chin-Kai Tseng 3,4, Yu-Hsuan Wu 3,4, Chih-Chuang Liaw 1,5, Chun-
Cyclooxygenase 2 facilitates dengue virus replication and serves as a potential target for developing antiviral agents Chun-Kuang Lin 1,2, Chin-Kai Tseng 3,4, Yu-Hsuan Wu 3,4, Chih-Chuang Liaw 1,5, Chun-
Supplementary Figure S1. Venn diagram analysis of mrna microarray data and mirna target analysis. (a) Western blot analysis of T lymphoblasts (CLS)
 Supplementary Figure S1. Venn diagram analysis of mrna microarray data and mirna target analysis. (a) Western blot analysis of T lymphoblasts (CLS) and their exosomes (EXO) in resting (REST) and activated
Supplementary Figure S1. Venn diagram analysis of mrna microarray data and mirna target analysis. (a) Western blot analysis of T lymphoblasts (CLS) and their exosomes (EXO) in resting (REST) and activated
Phenomena first observed in petunia
 Vectors for RNAi Phenomena first observed in petunia Attempted to overexpress chalone synthase (anthrocyanin pigment gene) in petunia. (trying to darken flower color) Caused the loss of pigment. Bill Douherty
Vectors for RNAi Phenomena first observed in petunia Attempted to overexpress chalone synthase (anthrocyanin pigment gene) in petunia. (trying to darken flower color) Caused the loss of pigment. Bill Douherty
FOXO Reporter Kit PI3K/AKT Pathway Cat. #60643
 Data Sheet FOXO Reporter Kit PI3K/AKT Pathway Cat. #60643 Background The PI3K/AKT signaling pathway is essential for cell growth and survival. Disruption of this pathway or its regulation has been linked
Data Sheet FOXO Reporter Kit PI3K/AKT Pathway Cat. #60643 Background The PI3K/AKT signaling pathway is essential for cell growth and survival. Disruption of this pathway or its regulation has been linked
Intrinsic cellular defenses against virus infection
 Intrinsic cellular defenses against virus infection Detection of virus infection Host cell response to virus infection Interferons: structure and synthesis Induction of antiviral activity Viral defenses
Intrinsic cellular defenses against virus infection Detection of virus infection Host cell response to virus infection Interferons: structure and synthesis Induction of antiviral activity Viral defenses
MicroRNA and Male Infertility: A Potential for Diagnosis
 Review Article MicroRNA and Male Infertility: A Potential for Diagnosis * Abstract MicroRNAs (mirnas) are small non-coding single stranded RNA molecules that are physiologically produced in eukaryotic
Review Article MicroRNA and Male Infertility: A Potential for Diagnosis * Abstract MicroRNAs (mirnas) are small non-coding single stranded RNA molecules that are physiologically produced in eukaryotic
Hepatitis B Antiviral Drug Development Multi-Marker Screening Assay
 Hepatitis B Antiviral Drug Development Multi-Marker Screening Assay Background ImQuest BioSciences has developed and qualified a single-plate method to expedite the screening of antiviral agents against
Hepatitis B Antiviral Drug Development Multi-Marker Screening Assay Background ImQuest BioSciences has developed and qualified a single-plate method to expedite the screening of antiviral agents against
Determination of the temporal pattern and importance of BALF1 expression in Epstein-Barr viral infection
 Determination of the temporal pattern and importance of BALF1 expression in Epstein-Barr viral infection Melissa Mihelidakis May 6, 2004 7.340 Research Proposal Introduction Apoptosis, or programmed cell
Determination of the temporal pattern and importance of BALF1 expression in Epstein-Barr viral infection Melissa Mihelidakis May 6, 2004 7.340 Research Proposal Introduction Apoptosis, or programmed cell
LESSON 4.4 WORKBOOK. How viruses make us sick: Viral Replication
 DEFINITIONS OF TERMS Eukaryotic: Non-bacterial cell type (bacteria are prokaryotes).. LESSON 4.4 WORKBOOK How viruses make us sick: Viral Replication This lesson extends the principles we learned in Unit
DEFINITIONS OF TERMS Eukaryotic: Non-bacterial cell type (bacteria are prokaryotes).. LESSON 4.4 WORKBOOK How viruses make us sick: Viral Replication This lesson extends the principles we learned in Unit
Nature Structural & Molecular Biology: doi: /nsmb Supplementary Figure 1. Differential expression of mirnas from the pri-mir-17-92a locus.
 Supplementary Figure 1 Differential expression of mirnas from the pri-mir-17-92a locus. (a) The mir-17-92a expression unit in the third intron of the host mir-17hg transcript. (b,c) Impact of knockdown
Supplementary Figure 1 Differential expression of mirnas from the pri-mir-17-92a locus. (a) The mir-17-92a expression unit in the third intron of the host mir-17hg transcript. (b,c) Impact of knockdown
Supplementary Fig. 1. Delivery of mirnas via Red Fluorescent Protein.
 prfp-vector RFP Exon1 Intron RFP Exon2 prfp-mir-124 mir-93/124 RFP Exon1 Intron RFP Exon2 Untransfected prfp-vector prfp-mir-93 prfp-mir-124 Supplementary Fig. 1. Delivery of mirnas via Red Fluorescent
prfp-vector RFP Exon1 Intron RFP Exon2 prfp-mir-124 mir-93/124 RFP Exon1 Intron RFP Exon2 Untransfected prfp-vector prfp-mir-93 prfp-mir-124 Supplementary Fig. 1. Delivery of mirnas via Red Fluorescent
Supplementary Figure 1
 Supplementary Figure 1 Asymmetrical function of 5p and 3p arms of mir-181 and mir-30 families and mir-142 and mir-154. (a) Control experiments using mirna sensor vector and empty pri-mirna overexpression
Supplementary Figure 1 Asymmetrical function of 5p and 3p arms of mir-181 and mir-30 families and mir-142 and mir-154. (a) Control experiments using mirna sensor vector and empty pri-mirna overexpression
Analysis of small RNAs from Drosophila Schneider cells using the Small RNA assay on the Agilent 2100 bioanalyzer. Application Note
 Analysis of small RNAs from Drosophila Schneider cells using the Small RNA assay on the Agilent 2100 bioanalyzer Application Note Odile Sismeiro, Jean-Yves Coppée, Christophe Antoniewski, and Hélène Thomassin
Analysis of small RNAs from Drosophila Schneider cells using the Small RNA assay on the Agilent 2100 bioanalyzer Application Note Odile Sismeiro, Jean-Yves Coppée, Christophe Antoniewski, and Hélène Thomassin
Supporting Information
 Supporting Information Palmisano et al. 10.1073/pnas.1202174109 Fig. S1. Expression of different transgenes, driven by either viral or human promoters, is up-regulated by amino acid starvation. (A) Quantification
Supporting Information Palmisano et al. 10.1073/pnas.1202174109 Fig. S1. Expression of different transgenes, driven by either viral or human promoters, is up-regulated by amino acid starvation. (A) Quantification
An Epstein-Barr virus-encoded microrna targets PUMA to promote host cell survival
 An Epstein-Barr virus-encoded microrna targets to promote host cell survival The Journal of Experimental Medicine 205(11): 2551-2560, 2008. 1 Elizabeth Yee-Wai Choy, Kam-Leung Siu, Kin-Hang Kok, Raymond
An Epstein-Barr virus-encoded microrna targets to promote host cell survival The Journal of Experimental Medicine 205(11): 2551-2560, 2008. 1 Elizabeth Yee-Wai Choy, Kam-Leung Siu, Kin-Hang Kok, Raymond
York criteria, 6 RA patients and 10 age- and gender-matched healthy controls (HCs).
 MATERIALS AND METHODS Study population Blood samples were obtained from 15 patients with AS fulfilling the modified New York criteria, 6 RA patients and 10 age- and gender-matched healthy controls (HCs).
MATERIALS AND METHODS Study population Blood samples were obtained from 15 patients with AS fulfilling the modified New York criteria, 6 RA patients and 10 age- and gender-matched healthy controls (HCs).
p47 negatively regulates IKK activation by inducing the lysosomal degradation of polyubiquitinated NEMO
 Supplementary Information p47 negatively regulates IKK activation by inducing the lysosomal degradation of polyubiquitinated NEMO Yuri Shibata, Masaaki Oyama, Hiroko Kozuka-Hata, Xiao Han, Yuetsu Tanaka,
Supplementary Information p47 negatively regulates IKK activation by inducing the lysosomal degradation of polyubiquitinated NEMO Yuri Shibata, Masaaki Oyama, Hiroko Kozuka-Hata, Xiao Han, Yuetsu Tanaka,
Construction of a hepatocellular carcinoma cell line that stably expresses stathmin with a Ser25 phosphorylation site mutation
 Construction of a hepatocellular carcinoma cell line that stably expresses stathmin with a Ser25 phosphorylation site mutation J. Du 1, Z.H. Tao 2, J. Li 2, Y.K. Liu 3 and L. Gan 2 1 Department of Chemistry,
Construction of a hepatocellular carcinoma cell line that stably expresses stathmin with a Ser25 phosphorylation site mutation J. Du 1, Z.H. Tao 2, J. Li 2, Y.K. Liu 3 and L. Gan 2 1 Department of Chemistry,
Human Genome: Mapping, Sequencing Techniques, Diseases
 Human Genome: Mapping, Sequencing Techniques, Diseases Lecture 4 BINF 7580 Fall 2005 1 Let us review what we talked about at the previous lecture. Please,... 2 The central dogma states that the transfer
Human Genome: Mapping, Sequencing Techniques, Diseases Lecture 4 BINF 7580 Fall 2005 1 Let us review what we talked about at the previous lecture. Please,... 2 The central dogma states that the transfer
Lecture 2: Virology. I. Background
 Lecture 2: Virology I. Background A. Properties 1. Simple biological systems a. Aggregates of nucleic acids and protein 2. Non-living a. Cannot reproduce or carry out metabolic activities outside of a
Lecture 2: Virology I. Background A. Properties 1. Simple biological systems a. Aggregates of nucleic acids and protein 2. Non-living a. Cannot reproduce or carry out metabolic activities outside of a
Data Sheet. Notch Pathway Reporter Kit Catalog # 60509
 Data Sheet Notch Pathway Reporter Kit Catalog # 60509 6042 Cornerstone Court W, Ste B Background The Notch signaling pathway controls cell fate decisions in vertebrate and invertebrate tissues. NOTCH signaling
Data Sheet Notch Pathway Reporter Kit Catalog # 60509 6042 Cornerstone Court W, Ste B Background The Notch signaling pathway controls cell fate decisions in vertebrate and invertebrate tissues. NOTCH signaling
Herpesviruses. Virion. Genome. Genes and proteins. Viruses and hosts. Diseases. Distinctive characteristics
 Herpesviruses Virion Genome Genes and proteins Viruses and hosts Diseases Distinctive characteristics Virion Enveloped icosahedral capsid (T=16), diameter 125 nm Diameter of enveloped virion 200 nm Capsid
Herpesviruses Virion Genome Genes and proteins Viruses and hosts Diseases Distinctive characteristics Virion Enveloped icosahedral capsid (T=16), diameter 125 nm Diameter of enveloped virion 200 nm Capsid
Polyomaviridae. Spring
 Polyomaviridae Spring 2002 331 Antibody Prevalence for BK & JC Viruses Spring 2002 332 Polyoma Viruses General characteristics Papovaviridae: PA - papilloma; PO - polyoma; VA - vacuolating agent a. 45nm
Polyomaviridae Spring 2002 331 Antibody Prevalence for BK & JC Viruses Spring 2002 332 Polyoma Viruses General characteristics Papovaviridae: PA - papilloma; PO - polyoma; VA - vacuolating agent a. 45nm
mirna Dr. S Hosseini-Asl
 mirna Dr. S Hosseini-Asl 1 2 MicroRNAs (mirnas) are small noncoding RNAs which enhance the cleavage or translational repression of specific mrna with recognition site(s) in the 3 - untranslated region
mirna Dr. S Hosseini-Asl 1 2 MicroRNAs (mirnas) are small noncoding RNAs which enhance the cleavage or translational repression of specific mrna with recognition site(s) in the 3 - untranslated region
Overview: Chapter 19 Viruses: A Borrowed Life
 Overview: Chapter 19 Viruses: A Borrowed Life Viruses called bacteriophages can infect and set in motion a genetic takeover of bacteria, such as Escherichia coli Viruses lead a kind of borrowed life between
Overview: Chapter 19 Viruses: A Borrowed Life Viruses called bacteriophages can infect and set in motion a genetic takeover of bacteria, such as Escherichia coli Viruses lead a kind of borrowed life between
Transcription and RNA processing
 Transcription and RNA processing Lecture 7 Biology W3310/4310 Virology Spring 2016 It is possible that Nature invented DNA for the purpose of achieving regulation at the transcriptional rather than at
Transcription and RNA processing Lecture 7 Biology W3310/4310 Virology Spring 2016 It is possible that Nature invented DNA for the purpose of achieving regulation at the transcriptional rather than at
VIRAL TITER COUNTS. The best methods of measuring infectious lentiviral titer
 VIRAL TITER COUNTS The best methods of measuring infectious lentiviral titer FLUORESCENCE CYCLES qpcr of Viral RNA SUMMARY Viral vectors are now routinely used for gene transduction in a wide variety of
VIRAL TITER COUNTS The best methods of measuring infectious lentiviral titer FLUORESCENCE CYCLES qpcr of Viral RNA SUMMARY Viral vectors are now routinely used for gene transduction in a wide variety of
Ch. 18 Regulation of Gene Expression
 Ch. 18 Regulation of Gene Expression 1 Human genome has around 23,688 genes (Scientific American 2/2006) Essential Questions: How is transcription regulated? How are genes expressed? 2 Bacteria regulate
Ch. 18 Regulation of Gene Expression 1 Human genome has around 23,688 genes (Scientific American 2/2006) Essential Questions: How is transcription regulated? How are genes expressed? 2 Bacteria regulate
Chapter 2. Investigation into mir-346 Regulation of the nachr α5 Subunit
 15 Chapter 2 Investigation into mir-346 Regulation of the nachr α5 Subunit MicroRNA s (mirnas) are small (< 25 base pairs), single stranded, non-coding RNAs that regulate gene expression at the post transcriptional
15 Chapter 2 Investigation into mir-346 Regulation of the nachr α5 Subunit MicroRNA s (mirnas) are small (< 25 base pairs), single stranded, non-coding RNAs that regulate gene expression at the post transcriptional
he micrornas of Caenorhabditis elegans (Lim et al. Genes & Development 2003)
 MicroRNAs: Genomics, Biogenesis, Mechanism, and Function (D. Bartel Cell 2004) he micrornas of Caenorhabditis elegans (Lim et al. Genes & Development 2003) Vertebrate MicroRNA Genes (Lim et al. Science
MicroRNAs: Genomics, Biogenesis, Mechanism, and Function (D. Bartel Cell 2004) he micrornas of Caenorhabditis elegans (Lim et al. Genes & Development 2003) Vertebrate MicroRNA Genes (Lim et al. Science
Chapter 19: Viruses. 1. Viral Structure & Reproduction. 2. Bacteriophages. 3. Animal Viruses. 4. Viroids & Prions
 Chapter 19: Viruses 1. Viral Structure & Reproduction 2. Bacteriophages 3. Animal Viruses 4. Viroids & Prions 1. Viral Structure & Reproduction Chapter Reading pp. 393-396 What exactly is a Virus? Viruses
Chapter 19: Viruses 1. Viral Structure & Reproduction 2. Bacteriophages 3. Animal Viruses 4. Viroids & Prions 1. Viral Structure & Reproduction Chapter Reading pp. 393-396 What exactly is a Virus? Viruses
In vitro DNase I foot printing. In vitro DNase I footprinting was performed as described
 Supplemental Methods In vitro DNase I foot printing. In vitro DNase I footprinting was performed as described previously 1 2 using 32P-labeled 211 bp fragment from 3 HS1. Footprinting reaction mixes contained
Supplemental Methods In vitro DNase I foot printing. In vitro DNase I footprinting was performed as described previously 1 2 using 32P-labeled 211 bp fragment from 3 HS1. Footprinting reaction mixes contained
Transcription and RNA processing
 Transcription and RNA processing Lecture 7 Biology 3310/4310 Virology Spring 2018 It is possible that Nature invented DNA for the purpose of achieving regulation at the transcriptional rather than at the
Transcription and RNA processing Lecture 7 Biology 3310/4310 Virology Spring 2018 It is possible that Nature invented DNA for the purpose of achieving regulation at the transcriptional rather than at the
Human Rotavirus A. genesig Standard Kit. Non structural protein 5 (NSP5) 150 tests. Primerdesign Ltd. For general laboratory and research use only
 TM Primerdesign Ltd Human Rotavirus A Non structural protein 5 (NSP5) genesig Standard Kit 150 tests For general laboratory and research use only 1 Introduction to Human Rotavirus A Rotavirus is a genus
TM Primerdesign Ltd Human Rotavirus A Non structural protein 5 (NSP5) genesig Standard Kit 150 tests For general laboratory and research use only 1 Introduction to Human Rotavirus A Rotavirus is a genus
Identification of Mutation(s) in. Associated with Neutralization Resistance. Miah Blomquist
 Identification of Mutation(s) in the HIV 1 gp41 Subunit Associated with Neutralization Resistance Miah Blomquist What is HIV 1? HIV-1 is an epidemic that affects over 34 million people worldwide. HIV-1
Identification of Mutation(s) in the HIV 1 gp41 Subunit Associated with Neutralization Resistance Miah Blomquist What is HIV 1? HIV-1 is an epidemic that affects over 34 million people worldwide. HIV-1
Summary and conclusions
 Summary and conclusions 133 Part 1: Toxicity effects of Cry toxins upon hemocoelic delivery to A. janata larvae The first investigation was aimed to find out whether other modes of delivery of Cry toxins
Summary and conclusions 133 Part 1: Toxicity effects of Cry toxins upon hemocoelic delivery to A. janata larvae The first investigation was aimed to find out whether other modes of delivery of Cry toxins
Part-4. Cell cycle regulatory protein 5 (Cdk5) A novel target of ERK in Carb induced cell death
 Part-4 Cell cycle regulatory protein 5 (Cdk5) A novel target of ERK in Carb induced cell death 95 1. Introduction The process of replicating DNA and dividing cells can be described as a series of coordinated
Part-4 Cell cycle regulatory protein 5 (Cdk5) A novel target of ERK in Carb induced cell death 95 1. Introduction The process of replicating DNA and dividing cells can be described as a series of coordinated
Online Data Supplement. Anti-aging Gene Klotho Enhances Glucose-induced Insulin Secretion by Upregulating Plasma Membrane Retention of TRPV2
 Online Data Supplement Anti-aging Gene Klotho Enhances Glucose-induced Insulin Secretion by Upregulating Plasma Membrane Retention of TRPV2 Yi Lin and Zhongjie Sun Department of physiology, college of
Online Data Supplement Anti-aging Gene Klotho Enhances Glucose-induced Insulin Secretion by Upregulating Plasma Membrane Retention of TRPV2 Yi Lin and Zhongjie Sun Department of physiology, college of
Recombinant Protein Expression Retroviral system
 Recombinant Protein Expression Retroviral system Viruses Contains genome DNA or RNA Genome encased in a protein coat or capsid. Some viruses have membrane covering protein coat enveloped virus Ø Essential
Recombinant Protein Expression Retroviral system Viruses Contains genome DNA or RNA Genome encased in a protein coat or capsid. Some viruses have membrane covering protein coat enveloped virus Ø Essential
Human Immunodeficiency Virus
 Human Immunodeficiency Virus Virion Genome Genes and proteins Viruses and hosts Diseases Distinctive characteristics Viruses and hosts Lentivirus from Latin lentis (slow), for slow progression of disease
Human Immunodeficiency Virus Virion Genome Genes and proteins Viruses and hosts Diseases Distinctive characteristics Viruses and hosts Lentivirus from Latin lentis (slow), for slow progression of disease
Chapter13 Characterizing and Classifying Viruses, Viroids, and Prions
 Chapter13 Characterizing and Classifying Viruses, Viroids, and Prions 11/20/2017 MDufilho 1 Characteristics of Viruses Viruses Minuscule, acellular, infectious agent having either DNA or RNA Cause infections
Chapter13 Characterizing and Classifying Viruses, Viroids, and Prions 11/20/2017 MDufilho 1 Characteristics of Viruses Viruses Minuscule, acellular, infectious agent having either DNA or RNA Cause infections
VIRUSES. Biology Applications Control. David R. Harper. Garland Science Taylor & Francis Group NEW YORK AND LONDON
 VIRUSES Biology Applications Control David R. Harper GS Garland Science Taylor & Francis Group NEW YORK AND LONDON vii Chapter 1 Virus Structure and 2.2 VIRUS MORPHOLOGY 26 Infection 1 2.3 VIRAL CLASSIFICATION
VIRUSES Biology Applications Control David R. Harper GS Garland Science Taylor & Francis Group NEW YORK AND LONDON vii Chapter 1 Virus Structure and 2.2 VIRUS MORPHOLOGY 26 Infection 1 2.3 VIRAL CLASSIFICATION
Viruses Tomasz Kordula, Ph.D.
 Viruses Tomasz Kordula, Ph.D. Resources: Alberts et al., Molecular Biology of the Cell, pp. 295, 1330, 1431 1433; Lehninger CD Movie A0002201. Learning Objectives: 1. Understand parasitic life cycle of
Viruses Tomasz Kordula, Ph.D. Resources: Alberts et al., Molecular Biology of the Cell, pp. 295, 1330, 1431 1433; Lehninger CD Movie A0002201. Learning Objectives: 1. Understand parasitic life cycle of
Regulation of Gene Expression in Eukaryotes
 Ch. 19 Regulation of Gene Expression in Eukaryotes BIOL 222 Differential Gene Expression in Eukaryotes Signal Cells in a multicellular eukaryotic organism genetically identical differential gene expression
Ch. 19 Regulation of Gene Expression in Eukaryotes BIOL 222 Differential Gene Expression in Eukaryotes Signal Cells in a multicellular eukaryotic organism genetically identical differential gene expression
Supplemental Figure S1. Expression of Cirbp mrna in mouse tissues and NIH3T3 cells.
 SUPPLEMENTAL FIGURE AND TABLE LEGENDS Supplemental Figure S1. Expression of Cirbp mrna in mouse tissues and NIH3T3 cells. A) Cirbp mrna expression levels in various mouse tissues collected around the clock
SUPPLEMENTAL FIGURE AND TABLE LEGENDS Supplemental Figure S1. Expression of Cirbp mrna in mouse tissues and NIH3T3 cells. A) Cirbp mrna expression levels in various mouse tissues collected around the clock
October 26, Lecture Readings. Vesicular Trafficking, Secretory Pathway, HIV Assembly and Exit from Cell
 October 26, 2006 Vesicular Trafficking, Secretory Pathway, HIV Assembly and Exit from Cell 1. Secretory pathway a. Formation of coated vesicles b. SNAREs and vesicle targeting 2. Membrane fusion a. SNAREs
October 26, 2006 Vesicular Trafficking, Secretory Pathway, HIV Assembly and Exit from Cell 1. Secretory pathway a. Formation of coated vesicles b. SNAREs and vesicle targeting 2. Membrane fusion a. SNAREs
supplementary information
 Figure S1 Nucleotide binding status of RagA mutants. Wild type and mutant forms of MycRagA was transfected into HEK293 cells and the transfected cells were labeled with 32 Pphosphate. MycRagA was immunoprecipitated
Figure S1 Nucleotide binding status of RagA mutants. Wild type and mutant forms of MycRagA was transfected into HEK293 cells and the transfected cells were labeled with 32 Pphosphate. MycRagA was immunoprecipitated
Published online 17 July 2010 Nucleic Acids Research, 2010, Vol. 38, No doi: /nar/gkq637
 Published online 17 July 2010 Nucleic Acids Research, 2010, Vol. 38, No. 21 7673 7688 doi:10.1093/nar/gkq637 GAM/ZFp/ZNF512B is central to a gene sensor circuitry involving cell-cycle regulators, TGFb
Published online 17 July 2010 Nucleic Acids Research, 2010, Vol. 38, No. 21 7673 7688 doi:10.1093/nar/gkq637 GAM/ZFp/ZNF512B is central to a gene sensor circuitry involving cell-cycle regulators, TGFb
The functional investigation of the interaction between TATA-associated factor 3 (TAF3) and p53 protein
 THESIS BOOK The functional investigation of the interaction between TATA-associated factor 3 (TAF3) and p53 protein Orsolya Buzás-Bereczki Supervisors: Dr. Éva Bálint Dr. Imre Miklós Boros University of
THESIS BOOK The functional investigation of the interaction between TATA-associated factor 3 (TAF3) and p53 protein Orsolya Buzás-Bereczki Supervisors: Dr. Éva Bálint Dr. Imre Miklós Boros University of
Epstein-Barr virus driven promoter hypermethylated genes in gastric cancer
 RESEARCH FUND FOR THE CONTROL OF INFECTIOUS DISEASES Epstein-Barr virus driven promoter hypermethylated genes in gastric cancer J Yu *, KF To, QY Liang K e y M e s s a g e s 1. Somatostatin receptor 1
RESEARCH FUND FOR THE CONTROL OF INFECTIOUS DISEASES Epstein-Barr virus driven promoter hypermethylated genes in gastric cancer J Yu *, KF To, QY Liang K e y M e s s a g e s 1. Somatostatin receptor 1
Lentiviral Delivery of Combinatorial mirna Expression Constructs Provides Efficient Target Gene Repression.
 Supplementary Figure 1 Lentiviral Delivery of Combinatorial mirna Expression Constructs Provides Efficient Target Gene Repression. a, Design for lentiviral combinatorial mirna expression and sensor constructs.
Supplementary Figure 1 Lentiviral Delivery of Combinatorial mirna Expression Constructs Provides Efficient Target Gene Repression. a, Design for lentiviral combinatorial mirna expression and sensor constructs.
7.012 Problem Set 6 Solutions
 Name Section 7.012 Problem Set 6 Solutions Question 1 The viral family Orthomyxoviridae contains the influenza A, B and C viruses. These viruses have a (-)ss RNA genome surrounded by a capsid composed
Name Section 7.012 Problem Set 6 Solutions Question 1 The viral family Orthomyxoviridae contains the influenza A, B and C viruses. These viruses have a (-)ss RNA genome surrounded by a capsid composed
WHO Prequalification of In Vitro Diagnostics PUBLIC REPORT. Product: Alere q HIV-1/2 Detect WHO reference number: PQDx
 WHO Prequalification of In Vitro Diagnostics PUBLIC REPORT Product: Alere q HIV-1/2 Detect WHO reference number: PQDx 0226-032-00 Alere q HIV-1/2 Detect with product codes 270110050, 270110010 and 270300001,
WHO Prequalification of In Vitro Diagnostics PUBLIC REPORT Product: Alere q HIV-1/2 Detect WHO reference number: PQDx 0226-032-00 Alere q HIV-1/2 Detect with product codes 270110050, 270110010 and 270300001,
Profiles of gene expression & diagnosis/prognosis of cancer. MCs in Advanced Genetics Ainoa Planas Riverola
 Profiles of gene expression & diagnosis/prognosis of cancer MCs in Advanced Genetics Ainoa Planas Riverola Gene expression profiles Gene expression profiling Used in molecular biology, it measures the
Profiles of gene expression & diagnosis/prognosis of cancer MCs in Advanced Genetics Ainoa Planas Riverola Gene expression profiles Gene expression profiling Used in molecular biology, it measures the
Supplementary data Supplementary Figure 1 Supplementary Figure 2
 Supplementary data Supplementary Figure 1 SPHK1 sirna increases RANKL-induced osteoclastogenesis in RAW264.7 cell culture. (A) RAW264.7 cells were transfected with oligocassettes containing SPHK1 sirna
Supplementary data Supplementary Figure 1 SPHK1 sirna increases RANKL-induced osteoclastogenesis in RAW264.7 cell culture. (A) RAW264.7 cells were transfected with oligocassettes containing SPHK1 sirna
Supplementary information. MARCH8 inhibits HIV-1 infection by reducing virion incorporation of envelope glycoproteins
 Supplementary information inhibits HIV-1 infection by reducing virion incorporation of envelope glycoproteins Takuya Tada, Yanzhao Zhang, Takayoshi Koyama, Minoru Tobiume, Yasuko Tsunetsugu-Yokota, Shoji
Supplementary information inhibits HIV-1 infection by reducing virion incorporation of envelope glycoproteins Takuya Tada, Yanzhao Zhang, Takayoshi Koyama, Minoru Tobiume, Yasuko Tsunetsugu-Yokota, Shoji
Supplementary Information
 Supplementary Information HBV maintains electrostatic homeostasis by modulating negative charges from phosphoserine and encapsidated nucleic acids Authors: Pei-Yi Su 1,2,3, Ching-Jen Yang 2, Tien-Hua Chu
Supplementary Information HBV maintains electrostatic homeostasis by modulating negative charges from phosphoserine and encapsidated nucleic acids Authors: Pei-Yi Su 1,2,3, Ching-Jen Yang 2, Tien-Hua Chu
Egr-1 regulates RTA transcription through a cooperative involvement of transcriptional regulators
 /, 2017, Vol. 8, (No. 53), pp: 91425-91444 Egr-1 regulates RTA transcription through a cooperative involvement of transcriptional regulators Roni Sarkar 1 and Subhash C. Verma 1 1 Department of Microbiology
/, 2017, Vol. 8, (No. 53), pp: 91425-91444 Egr-1 regulates RTA transcription through a cooperative involvement of transcriptional regulators Roni Sarkar 1 and Subhash C. Verma 1 1 Department of Microbiology
Chapter 18. Viral Genetics. AP Biology
 Chapter 18. Viral Genetics 2003-2004 1 A sense of size Comparing eukaryote bacterium virus 2 What is a virus? Is it alive? DNA or RNA enclosed in a protein coat Viruses are not cells Extremely tiny electron
Chapter 18. Viral Genetics 2003-2004 1 A sense of size Comparing eukaryote bacterium virus 2 What is a virus? Is it alive? DNA or RNA enclosed in a protein coat Viruses are not cells Extremely tiny electron
Last time we talked about the few steps in viral replication cycle and the un-coating stage:
 Zeina Al-Momani Last time we talked about the few steps in viral replication cycle and the un-coating stage: Un-coating: is a general term for the events which occur after penetration, we talked about
Zeina Al-Momani Last time we talked about the few steps in viral replication cycle and the un-coating stage: Un-coating: is a general term for the events which occur after penetration, we talked about
Cell isolation. Spleen and lymph nodes (axillary, inguinal) were removed from mice
 Supplementary Methods: Cell isolation. Spleen and lymph nodes (axillary, inguinal) were removed from mice and gently meshed in DMEM containing 10% FBS to prepare for single cell suspensions. CD4 + CD25
Supplementary Methods: Cell isolation. Spleen and lymph nodes (axillary, inguinal) were removed from mice and gently meshed in DMEM containing 10% FBS to prepare for single cell suspensions. CD4 + CD25
Choosing Between Lentivirus and Adeno-associated Virus For DNA Delivery
 Choosing Between Lentivirus and Adeno-associated Virus For DNA Delivery Presenter: April 12, 2017 Ed Davis, Ph.D. Senior Application Scientist GeneCopoeia, Inc. Outline Introduction to GeneCopoeia Lentiviral
Choosing Between Lentivirus and Adeno-associated Virus For DNA Delivery Presenter: April 12, 2017 Ed Davis, Ph.D. Senior Application Scientist GeneCopoeia, Inc. Outline Introduction to GeneCopoeia Lentiviral
Section 6. Junaid Malek, M.D.
 Section 6 Junaid Malek, M.D. The Golgi and gp160 gp160 transported from ER to the Golgi in coated vesicles These coated vesicles fuse to the cis portion of the Golgi and deposit their cargo in the cisternae
Section 6 Junaid Malek, M.D. The Golgi and gp160 gp160 transported from ER to the Golgi in coated vesicles These coated vesicles fuse to the cis portion of the Golgi and deposit their cargo in the cisternae
numbe r Done by Corrected by Doctor
 numbe r 5 Done by Mustafa Khader Corrected by Mahdi Sharawi Doctor Ashraf Khasawneh Viral Replication Mechanisms: (Protein Synthesis) 1. Monocistronic Method: All human cells practice the monocistronic
numbe r 5 Done by Mustafa Khader Corrected by Mahdi Sharawi Doctor Ashraf Khasawneh Viral Replication Mechanisms: (Protein Synthesis) 1. Monocistronic Method: All human cells practice the monocistronic
Supplementary Data Table of Contents:
 Supplementary Data Table of Contents: - Supplementary Methods - Supplementary Figures S1(A-B) - Supplementary Figures S2 (A-B) - Supplementary Figures S3 - Supplementary Figures S4(A-B) - Supplementary
Supplementary Data Table of Contents: - Supplementary Methods - Supplementary Figures S1(A-B) - Supplementary Figures S2 (A-B) - Supplementary Figures S3 - Supplementary Figures S4(A-B) - Supplementary
Blocking the expression of the hepatitis B virus S gene in hepatocellular carcinoma cell lines with an anti-gene locked nucleic acid in vitro
 Blocking the expression of the hepatitis B virus S gene in hepatocellular carcinoma cell lines with an anti-gene locked nucleic acid in vitro Y.-B. Deng, H.-J. Qin, Y.-H. Luo, Z.-R. Liang and J.-J. Zou
Blocking the expression of the hepatitis B virus S gene in hepatocellular carcinoma cell lines with an anti-gene locked nucleic acid in vitro Y.-B. Deng, H.-J. Qin, Y.-H. Luo, Z.-R. Liang and J.-J. Zou
NOVEL FUNCTION OF mirnas IN REGULATING GENE EXPRESSION. Ana M. Martinez
 NOVEL FUNCTION OF mirnas IN REGULATING GENE EXPRESSION Ana M. Martinez Switching from Repression to Activation: MicroRNAs can Up-Regulate Translation. Shoba Vasudevan, Yingchun Tong, Joan A. Steitz AU-rich
NOVEL FUNCTION OF mirnas IN REGULATING GENE EXPRESSION Ana M. Martinez Switching from Repression to Activation: MicroRNAs can Up-Regulate Translation. Shoba Vasudevan, Yingchun Tong, Joan A. Steitz AU-rich
Chapter 19: Viruses. 1. Viral Structure & Reproduction. What exactly is a Virus? 11/7/ Viral Structure & Reproduction. 2.
 Chapter 19: Viruses 1. Viral Structure & Reproduction 2. Bacteriophages 3. Animal Viruses 4. Viroids & Prions 1. Viral Structure & Reproduction Chapter Reading pp. 393-396 What exactly is a Virus? Viruses
Chapter 19: Viruses 1. Viral Structure & Reproduction 2. Bacteriophages 3. Animal Viruses 4. Viroids & Prions 1. Viral Structure & Reproduction Chapter Reading pp. 393-396 What exactly is a Virus? Viruses
For in vitro Veterinary Diagnostics only. Kylt Rotavirus A. Real-Time RT-PCR Detection.
 For in vitro Veterinary Diagnostics only. Kylt Rotavirus A Real-Time RT-PCR Detection www.kylt.eu DIRECTION FOR USE Kylt Rotavirus A Real-Time RT-PCR Detection A. General Kylt Rotavirus A products are
For in vitro Veterinary Diagnostics only. Kylt Rotavirus A Real-Time RT-PCR Detection www.kylt.eu DIRECTION FOR USE Kylt Rotavirus A Real-Time RT-PCR Detection A. General Kylt Rotavirus A products are
VIRUSES. 1. Describe the structure of a virus by completing the following chart.
 AP BIOLOGY MOLECULAR GENETICS ACTIVITY #3 NAME DATE HOUR VIRUSES 1. Describe the structure of a virus by completing the following chart. Viral Part Description of Part 2. Some viruses have an envelope
AP BIOLOGY MOLECULAR GENETICS ACTIVITY #3 NAME DATE HOUR VIRUSES 1. Describe the structure of a virus by completing the following chart. Viral Part Description of Part 2. Some viruses have an envelope
The Blueprint of Life: DNA to Protein. What is genetics? DNA Structure 4/27/2011. Chapter 7
 The Blueprint of Life: NA to Protein Chapter 7 What is genetics? The science of heredity; includes the study of genes, how they carry information, how they are replicated, how they are expressed NA Structure
The Blueprint of Life: NA to Protein Chapter 7 What is genetics? The science of heredity; includes the study of genes, how they carry information, how they are replicated, how they are expressed NA Structure
The Blueprint of Life: DNA to Protein
 The Blueprint of Life: NA to Protein Chapter 7 What is genetics? The science of heredity; includes the y; study of genes, how they carry information, how they are replicated, how they are expressed 1 NA
The Blueprint of Life: NA to Protein Chapter 7 What is genetics? The science of heredity; includes the y; study of genes, how they carry information, how they are replicated, how they are expressed 1 NA
Supporting Online Material Material and Methods References Supplemental Figures S1, S2, and S3
 Supporting Online Material Material and Methods References Supplemental Figures S1, S2, and S3 Sarbassov et al. 1 Material and Methods Materials Reagents were obtained from the following sources: protein
Supporting Online Material Material and Methods References Supplemental Figures S1, S2, and S3 Sarbassov et al. 1 Material and Methods Materials Reagents were obtained from the following sources: protein
AP Biology Reading Guide. Concept 19.1 A virus consists of a nucleic acid surrounded by a protein coat
 AP Biology Reading Guide Name Chapter 19: Viruses Overview Experimental work with viruses has provided important evidence that genes are made of nucleic acids. Viruses were also important in working out
AP Biology Reading Guide Name Chapter 19: Viruses Overview Experimental work with viruses has provided important evidence that genes are made of nucleic acids. Viruses were also important in working out
Overview: Conducting the Genetic Orchestra Prokaryotes and eukaryotes alter gene expression in response to their changing environment
 Overview: Conducting the Genetic Orchestra Prokaryotes and eukaryotes alter gene expression in response to their changing environment In multicellular eukaryotes, gene expression regulates development
Overview: Conducting the Genetic Orchestra Prokaryotes and eukaryotes alter gene expression in response to their changing environment In multicellular eukaryotes, gene expression regulates development
Graveley Lab shrna knockdown followed by RNA-seq Biosample Preparation and Characterization Document
 Graveley Lab shrna knockdown followed by RNA-seq Biosample Preparation and Characterization Document Wet Lab: Sara Olson and Lijun Zhan Computational Lab: Xintao Wei and Michael Duff PI: Brenton Graveley
Graveley Lab shrna knockdown followed by RNA-seq Biosample Preparation and Characterization Document Wet Lab: Sara Olson and Lijun Zhan Computational Lab: Xintao Wei and Michael Duff PI: Brenton Graveley
Received 3 September 2002/Accepted 15 January 2003
 JOURNAL OF VIROLOGY, Apr. 2003, p. 4646 4657 Vol. 77, No. 8 0022-538X/03/$08.00 0 DOI: 10.1128/JVI.77.8.4646 4657.2003 Copyright 2003, American Society for Microbiology. All Rights Reserved. Ability of
JOURNAL OF VIROLOGY, Apr. 2003, p. 4646 4657 Vol. 77, No. 8 0022-538X/03/$08.00 0 DOI: 10.1128/JVI.77.8.4646 4657.2003 Copyright 2003, American Society for Microbiology. All Rights Reserved. Ability of
Norgen s HIV proviral DNA PCR Kit was developed and validated to be used with the following PCR instruments: Qiagen Rotor-Gene Q BioRad icycler
 3430 Schmon Parkway Thorold, ON, Canada L2V 4Y6 Phone: (905) 227-8848 Fax: (905) 227-1061 Email: techsupport@norgenbiotek.com HIV Proviral DNA PCR Kit Product # 33840 Product Insert Background Information
3430 Schmon Parkway Thorold, ON, Canada L2V 4Y6 Phone: (905) 227-8848 Fax: (905) 227-1061 Email: techsupport@norgenbiotek.com HIV Proviral DNA PCR Kit Product # 33840 Product Insert Background Information
SC-L-H shared(37) Specific (1)
 A. Brain (total 2) Tissue-specific (2) Brain-heart shared (8) Specific (2) CNS-heart shared(68) Specific () Heart (total 4) Tissue-specific () B-L-H shared (37) specific () B. Brain (total 2) Tissue-specific
A. Brain (total 2) Tissue-specific (2) Brain-heart shared (8) Specific (2) CNS-heart shared(68) Specific () Heart (total 4) Tissue-specific () B-L-H shared (37) specific () B. Brain (total 2) Tissue-specific
Single Cell Quantitative Polymer Chain Reaction (sc-qpcr)
 Single Cell Quantitative Polymer Chain Reaction (sc-qpcr) Analyzing gene expression profiles from a bulk population of cells provides an average profile which may obscure important biological differences
Single Cell Quantitative Polymer Chain Reaction (sc-qpcr) Analyzing gene expression profiles from a bulk population of cells provides an average profile which may obscure important biological differences
number Done by Corrected by Doctor Ashraf
 number 4 Done by Nedaa Bani Ata Corrected by Rama Nada Doctor Ashraf Genome replication and gene expression Remember the steps of viral replication from the last lecture: Attachment, Adsorption, Penetration,
number 4 Done by Nedaa Bani Ata Corrected by Rama Nada Doctor Ashraf Genome replication and gene expression Remember the steps of viral replication from the last lecture: Attachment, Adsorption, Penetration,
EGFR shrna A: CCGGCGCAAGTGTAAGAAGTGCGAACTCGAGTTCGCACTTCTTACACTTGCG TTTTTG. EGFR shrna B: CCGGAGAATGTGGAATACCTAAGGCTCGAGCCTTAGGTATTCCACATTCTCTT TTTG
 Supplementary Methods Sequence of oligonucleotides used for shrna targeting EGFR EGFR shrna were obtained from the Harvard RNAi consortium. The following oligonucleotides (forward primer) were used to
Supplementary Methods Sequence of oligonucleotides used for shrna targeting EGFR EGFR shrna were obtained from the Harvard RNAi consortium. The following oligonucleotides (forward primer) were used to
Oxford Expression Technologies Ltd
 Oxford Expression Technologies Ltd Founded in 2007 as a spin out from Oxford Brookes University and Natural Environment Research Council Technology based on the insect baculovirus expression vectors (BEVs)
Oxford Expression Technologies Ltd Founded in 2007 as a spin out from Oxford Brookes University and Natural Environment Research Council Technology based on the insect baculovirus expression vectors (BEVs)
Cellular MicroRNA and P Bodies Modulate Host-HIV-1 Interactions. 指導教授 : 張麗冠博士 演講者 : 黃柄翰 Date: 2009/10/19
 Cellular MicroRNA and P Bodies Modulate Host-HIV-1 Interactions 指導教授 : 張麗冠博士 演講者 : 黃柄翰 Date: 2009/10/19 1 MicroRNA biogenesis 1. Pri mirna: primary mirna 2. Drosha: RNaseIII 3. DCR 1: Dicer 1 RNaseIII
Cellular MicroRNA and P Bodies Modulate Host-HIV-1 Interactions 指導教授 : 張麗冠博士 演講者 : 黃柄翰 Date: 2009/10/19 1 MicroRNA biogenesis 1. Pri mirna: primary mirna 2. Drosha: RNaseIII 3. DCR 1: Dicer 1 RNaseIII
Viral reproductive cycle
 Lecture 29: Viruses Lecture outline 11/11/05 Types of viruses Bacteriophage Lytic and lysogenic life cycles viruses viruses Influenza Prions Mad cow disease 0.5 µm Figure 18.4 Viral structure of capsid
Lecture 29: Viruses Lecture outline 11/11/05 Types of viruses Bacteriophage Lytic and lysogenic life cycles viruses viruses Influenza Prions Mad cow disease 0.5 µm Figure 18.4 Viral structure of capsid
Viruses. Properties. Some viruses contain other ingredients (e.g., lipids, carbohydrates), but these are derived from their host cells.
 Viruses Properties They are obligate intracellular parasites. Probably there are no cells in nature that escape infection by one or more kinds of viruses. (Viruses that infect bacteria are called bacteriophages.)
Viruses Properties They are obligate intracellular parasites. Probably there are no cells in nature that escape infection by one or more kinds of viruses. (Viruses that infect bacteria are called bacteriophages.)
7.014 Problem Set 7 Solutions
 MIT Department of Biology 7.014 Introductory Biology, Spring 2005 7.014 Problem Set 7 Solutions Question 1 Part A Antigen binding site Antigen binding site Variable region Light chain Light chain Variable
MIT Department of Biology 7.014 Introductory Biology, Spring 2005 7.014 Problem Set 7 Solutions Question 1 Part A Antigen binding site Antigen binding site Variable region Light chain Light chain Variable
Supplementary Figure 1. HGL-DTG levels in uninduced and herbivory induced leaves. Total HGL-
 Supplementary Figure 1. HGL-DTG levels in uninduced and herbivory induced leaves. Total HGL- DTG concentrations [F 1,4 = 36.88, P 0.0037; significant differences (threshold: P 0.05) between means (± SE)
Supplementary Figure 1. HGL-DTG levels in uninduced and herbivory induced leaves. Total HGL- DTG concentrations [F 1,4 = 36.88, P 0.0037; significant differences (threshold: P 0.05) between means (± SE)
