The Hormonal Regulation of Pyloric Sphincter Function
|
|
|
- Vincent Walters
- 6 years ago
- Views:
Transcription
1 The Hormonal Regulation of Pyloric Sphincter Function ROBERT S. FISHER, WILLIAM LIPSHUTZ, and SIDNEY COHEN From the GastroIntestinal Section, Department of Medicine, Hospital of the University of Pennsylvania and Veterans Administration Hospital Philadelphia, Pennsylvania A B S T R A C T The purpose of this study was to evaluate the hormonal control of pyloric sphincter function. Studies were performed on both pyloric circular muscle, in vitro, and the human pylorus, in vivo. Full doseresponse curves to gastrin I, cholecystokinin, and secretin were constructed for the pyloric muscle of the opossum studied at its length of optimal tension development, Lo. Both cholecystokinin and secretin were potent agonists on the muscle but gastrin I gave no increase in muscle tension. The combination of cholecystokinin and secretin was additive at submaximal concentrations but potentiation of the maximal responses was not observed. Gastrin I produced a surmountable, competitivelike antagonism to the effect of cholecystokinin on the pyloric muscle. The octapeptide of cholecystokinin was a more potent agonist than the whole molecule of cholecystokinin on the pyloric muscle. In man, the pyloric pressure rose significantly during intravenous infusion of either cholecystokinin or secretin. The combination of maximal doses of both hormones did not show signficant potentiation. Gastrin I did not significantly increase pyloric pressure but did antagonize the pyloric response to duodenal acidification. These studies suggest that: (a) Both secretin and cholecystokinin augment pyloric sphincter pressure while gastrin I is an antagonist inhibiting their effects. (b) The hormonal responses of pyloric sphincter circular muscle, in vitro, can be correlated with human sphincter function, in vivo. INTRODUCTION Intraluminal manometry employing opentipped infused catheters has recently been utilized to show that the human pylorus is characterized by a zone of elevated pressure which relaxes with antral peristalsis, contracts in response to intraduodenal stimulation, and prevents the Dr. Fisher's present address is Temple University Health Sciences Center, Philadelphia, Pennsylvania Received for publication 14 August 1972 and in revised form 2 Januarl, retrograde movement of duodenal contents into the stomach (1). Similar properties suggesting a sphincteric function have been recorded at the canine pylorus (2, 3). The mechanism by which the human pylorus contracts in response to the intraduodenal instillation of amino acids, olive oil, and HC1 is not known. It has been presumed to be hormonal because the substances which were instilled into the duodenum are known to release both secretin and cholecystokinin (411). In addition, a precedent for hormonal regulation of other gastrointestinal sphincters has been established (1216). The purpose of this study is: (a) to utilize pyloric circular muscle, in vitro, to characterize the interaction of the gastrointestinal hormones on the pyloric sphincter; and (b) to relate these observations on pyloric muscle, in vitro, to studies carried out in man, in vivo. METHODS In vitro experimientts. Studies were performed on 12 adult opossums of both sexes, weighing kg. The methods outlined here have been described previously in detail (17, 18). All animals were killed by intravenous pentobarbital. The entire stomach and duodenum were mobilized and freed from surrounding tissues. The distal duodenum was ligated and the upper gastrointestinal tract from esophagus to distal duodenum was excised and washed in KrebsRinger solution (composition in millimoles per liter: Na+, 138.6; K+, 4.6; Ca++, 2.5; Mg++, 2.1; Cl, 126.2; HC03, 21.9; P04, 1.2; glucose, 49.6) at 3738 C. The tissue was then transferred to an organ bath of Krebs Ringer solution, bubbled with 95% 02 and 5%o C02, and maintained at 3738'C. The pylorus was identified as a distinct, prominent anatomic ring at the junction of the duodenum and antrum. The pyloric ring was separated from the antrum proximally and from the duodenum distally. The mucosa from each region was removed to the level of the submucosa. The antral circular muscle strips were taken 2 cm proximal to the pylorus. Each anatomic region was identified histologically by its lining epithelium. The antrum and pylorus were lined by columnar epithelium with gastric glands. No Brunner's glands or intestinal villi were present in pyloric sections. Circular smooth muscle strips, 0.5 cm wide and 1.0 cm long, were cut from each anatomic region. Each muscle was mounted in a 20 ml bath of KrebsRiqger solution, The Journal of Clinical investigation Volume.52 May
2 d mmm Pwinriae 80 Duodenum 'I) ~~~Antrum E 60 k 40 "z 20 / CCK (Molar Concentration) FIGURE 1 Cholecystokinin (CCK) doseresponse curves at Lo for pyloric antral and duodenal circular muscle. Active tension is expressed as a percent of maximum response of pyloric muscle. Each point represents mean+se for 12 separate experiments. Pyloric muscle responded at a lower threshold dose and attained a greater maximum active tension than muscle from adjacent areas. bubbled with 95% 02 and 5% C02, and kept at 37380C. All strips were arranged to record the isometric tension of the circular smooth muscle. One end of the muscle was attached to an inflexible wire which was hooked to an external force transducer (Grass FT03C, Grass Instrument Co., Quincy, Mass.). The other end was attached to a metal rod which could be raised and lowered by adjustment of a screw micrometer. The isometric force from each transducer was graphed on a Beckman curvilinear inkwriting recorder (Beckman Instruments, Inc., Fullerton, Calif.). After a 30 min equilibration period, lengthtension curves were constructed as previously described (17, 18). Each muscle at graded increases in length, was stimulated with 10' M acetylcholine. The length of maximum active tension development, Lo,1 was thus obtained for each muscle studied. All muscles were then set at their respective Lo for the remainder of the, experiment. Doseresponse curves were constructed on each muscle strip, at Lo. Each hormone and paired combination was given in randomized order. The response of each muscle was used as its own control. Cholecystokinin, mol wt 3950, secretin, mol wt 3055 (GIH Research Unit, Karolinska Institute, Stockholm, Sweden), and gastrin I, mol wt (amino acid residue 217; Imperial Chemical Industries, Ltd., Alderley Park, Cheshire, England), were solubilized with KrebsRinger solution, and 1 ml volumes were added to a 20 ml bath to obtain the final molar concentration as noted. The molar concentration of secretin was calculated using 3.4 U of secretin being equal to 1,tg (19). The molar concentration of cholecystokinin was calculated using the stated quantity of 0.03 mg of active peptide per 75 U vial. Octapeptide of cholecystokinin, mol wt 1142 (The Squibb Institute for Medical Research, Princeton, N. J.), was evaluated in a similar manner. The doseresponse curves 'Abbreviation used in this paper: Lo, length of maximum active tensiondevelopment R. S. Fisher, W. Lipshutz, and S. Cohen were constructed using individual doses given in random order. No cumulative doses were given. The response to each dose of a hormone was recorded over a 10 min period. The maximum tension recorded at each dose was used to construct the doseresponse curves. The peak spontaneous contractions of each muscle during control periods were quantified and subtracted from the peak tension recorded at each dose. Stimulation of these muscles produced an increase in the amplitude of the spontaneous contractions with low concentrations of the hormone. At higher concentrations, the entire baseline pressure often became elevated. The muscle response to each hormone began promptly and was dissipated within 5 min. In the experiments where hormone combinations were utilized, studies were performed both by adding one hormone before the other and by simultaneous addition of the two hormones into the bath. At least 15 min were allowed between each dose combination. All muscles were lightly blotted and weighed at the termination of the experiment. All active tensions have been corrected for muscle weight. In vivo studies. 10 normal volunteer subjects, 8 males and 2 females, age 2154 yr (mean, 29) were studied. The subjects had no history of abdominal surgery and no symptoms of gastrointestinal disease. Informed consent was obtained from all volunteers. The tube assembly and recording apparatus utilized in these studies has been previously described (1). Five polyvinyl catheters were joined together into a single unit with an outside diameter of 5.4 mm. Intraluminal pressures were measured using three waterfilled catheters, each infused with distilled water at 1.1 ml/min, with their side orifices 1.2 mm in diameter, spaced at 1 cm intervals. The pyloric sphincter pressure was taken as the mean respiratory value obtained from the maximum zone of tonic pressure elevation. Phasic contractions were not quantified. The pressures recorded from the three recording orifices were meaned for each pullthrough. All pressures were expressed using intra
3 *S * Cholecystokinin _e Secretin ee@gastrin t t oI' 10i9 10T Hormone ( Molar Concentration) FIGURE 2 Cholecystokinin, gastrin I, and secretin doseresponse curves at Lo for pyloric circular muscle. Active tension is expressed as a percent of maximum response to cholecystokinin. Each point represents mean+se for 12 separate experiments. Both cholecystokinin and secretin increased the active tension. Pyloric muscle was inhibited by gastrin, but this response was not statistically significant (P > 0.05). abdominal (intragastric) pressure as a zero reference. A fourth catheter was employed to measure forearm skin to mucosa electrical potential difference (PD) using KCI bridges. The PD recording orifice was at the same level as the middle pressure detector. The duodenum was perfused through a fifth catheter whose openings were 4 cm beyond the distal pressure orifice. In subjects who had fasted overnight, the recording unit was passed into the second or third portion of the duodenum under direct fluoroscopic visualization. All measurements were made with the patient in the right lateral decubitus position. After stable readings were observed, the recording assembly was slowly withdrawn at 0.5cm intervals from duodenum to stomach, recording at each interval for 1 min. Basal records were obtained in all subjects. After the basal pullthrough, the assembly was again passed into the duodenum and the pyloric response to the intravenous administration of various gastrointestinal hormones was investigated. At least 1 h was allowed between testing of different hormones. Individual pullthrough measurements were recorded during administration of gastrin I at 1,ug/ kgh and 2,ug/kgh, secretin at 1.0 U/kgh (clinical units), cholecystokinin at 2.0 U/kgh (Ivy dog units) (GIH Research Unit, Stockholm, Sweden) and the combination of secretin and cholecystokinin at the above doses, in each subject. Each pullthrough was performed 10 min after hormonal administration had begun. Individual pullthrough measurements were also performed during the duodenal infusion of 0.1 N HCl at 7.6 cc/min (48 meq/h) alone, and with simultaneous intravenous administration of gastrin I at 1 jug/kgh and 2 Ag/kgh. Each subject received all intravenous test substances in random order. Each endogenous and exogenous pyloric response was compared to a basal level obtained between stimuli. The Student's t test was utilized in the statistical analysis of the data (20). RESULTS In vitro studics. All data was computed on the basis of the peak response to each hormone or hormone combination. Quantification of the peak response at the length of optimal tension development, Lo normalized the responses of muscle with dissimilar lengthtension properties (17). At Lo, each muscle was capable of generating its peak active tension to a given stimulus. Thus, Lo provided a constant reference for the study of muscle from different portions of the gastrointestinal tract and for the stud) of muscle from the same portion on different days. The maximum absolute responses of pyloric circular muscle to each hormone administered are listed in Table I. All subsequent doseresponse curves are constructed using the maximum absolute response to the single hormone that yielded the greatest active tension TABLE I Comparison of Maximum Absolute Responses of Pyloric Circular Muscle Peak active tension to secretin, g 4.6i1.0 Peak active tension to cholecystokinini, g Peak active tension to gastrin I, g 0.7i0.2 Peak active tension to octapeptide of cholecystokinin, g Each value represents the mean ±SE of 12 separate determinations. Hormonal Regulation of Pyloric Sphincter Function 1291
4 0) it 100r 8OF O* Octopeptide _. CCK ko Q H V Hormone ( Molar Concentration) FIGURE 3 Octapeptide of cholecystokinin and cholecystokinin doseresponse curves at Lo, for pyloric circular muscle. Active tension is expressed as a percent of maximum response to the octapeptide of cholecystokinin. Synthetic octapeptide gave a significantly greater response at molar concentrations of cholecystokinin, 8.7X1013 and 8.7X1012 (P <0.001). within the respective figure. All statistical comparisons are based upon absolute values for data obtained on the same muscle strips. In Fig. 1 the cholecystokinin log doseresponse curves are shown for circular muscle obtained from the duodenum, pylorus, and antrum. The responses of the muscle from each region are expressed as a percent of the maximum response of the pylorus, the muscle that developed the greatest active tension. The threshold and the peak responses for the pyloric circular muscle occurred at lower doses than for either the adjacent antrum or duodenum. The pyloric muscle showed a prominent diminution in response to cholecystokinin at doses beyond the peak value of 7.5 X 10'l M. Next the dose 100 t 80 (5 60, 40 Control *_O Gastrin xzi0m) 20 % 20 T X Te*10 I /~~~~~~~~~~ Ii 4 I I /i jo21 O'9 lo17 io15 IO 13 loii 109 ICD7 CCK (Molar Concentration) FIGURE 4 Doseresponse curves, at Lo, for cholecystokinin (CCK) alone and in the presence of 5X10"1 M gastrin I, on pyloric muscle. Active tension is expressed as a percent of maximum response to cholecystokinin when given alone. Each point represents mean+se for 12 separate experiments. In the presence of gastrin I, the threshold dose of cholecystokinin was increased and the maximum response to cholecystokinin was achieved. The slope of the steep linear portion of the cholecystokinin curve (y = 19.2x , r = 0.98) did not differ from the comparable slope (y = 20.8x , r = 0.99) in the presence of gastrin (P > 0.05) R. S. Fisher, W. Lipshutz, and S. Cohen
5 120J 100 S Control I * Secretin (3x 1013M) 80 E 60 Secretin Z 40 e 20 _ 0o21 io19 l i013 10"II CCK ( Molar Concentration ) FIGURE 5 Doseresponse curves, at Lo, for cholecystokinin (CCK) alone, and in the presence of 3X10'3 M secretin, on pyloric muscle. Active tension is expressed as a percent of maximal response to cholecystokinin alone. Each point represents mean+se for 12 separate experiments. The horizontal line represents the maximal tension to secretin alone. In the presence of secretin, the maximal response to cholecystokinin occurred at a lower concentration but the maximal response was not altered (P > 0.05). response curve of pyloric muscle to cholecystokinin was compared to the doseresponse curves of the other hormones. In Fig. 2 the log doseresponse curves of pyloric muscle are compared for secretin, gastrin I, and cholecystokinin. The response of the pyloric muscle is expressed as a percent of the maximal active tension to cholecystokinin. Both cholecvstokinin and secretin contracted pyloric muscle while gastrin I gave no response. Although cholecystokinin gave a response at the lowest threshold dose and developed the greatest active tension, secretin developed its maximum response at a lower molar concentration than cholecystokinin. To evaluate the pyloric muscle response to a synthetic purified form of cholecystokinin, a doseresponse curve was constructed for the synthetic terminal octapeptide of cholecystokinin. In Fig. 3 the doseresponse curves for both cholecystokinin and the octapeptide are compared. The data is expressed as a percent of the maximal active tension to the octapeptide. The octapeptide gave a significantly greater response at a lower concentration than the whole molecule of cholecystokinin (P < ). The interaction of different hormone combinations was studied next. In Fig. 4 the log doseresponse curves for pvloric circular muscle are shown for cholecvstokinin, alone, and for the combination of cholecystokinin with "gastrin I. In the presence of gastrin 1 (5 X 1011 AI), the entire doseresponse curve to cholecystokinin was shifted to higher concentrations. The threshold dose was greater and in the presence of gastrin I, the maximum response to cholecystokinin was still attained at higher concentrations. No significant difference was found between the slopes of the steep linear phases of each doseresponse curve. In Fig. 5 the doseresponse curves are shown for cholecystokinin alone, and cholecystokinin in the presence of secretin (3 X 10'3 M). This is the concentration of secretin that produced the maximum active tension on pyloric muscle. The data is expressed as a percent of the maximal active tension to cholecystokinin, alone. The horizontal line indicates the maximal tension to secretin, alone. In the presence of secretin, the maximal response to cholecystokinin was not significantly increased. However, the maximal response occurred at a lower concentration. Similar findings in the magnitude of the maximal response to cholecystokinin were observed when secretin was added 5 min before the cholecvstokinin; the additive effects of secretin were not seen at lower cholecystokinin concentrations. lin vizo studies. The hormonal responses observed in muscle strips in vitro, were next evaluated in human subjects under basal conditions and during the intravenouis infusion of each gastrointestinal hormone or hormone combination. The recording assembly was slowly withdrawn at 0.5cm intervals from duodenum to Hormonal Regulation of Pyloric Sphincter Function 129.3
6 , 16 t12 z T 0 Basal Secretin CCK Secretin.0Iu/kgh 1.0 U/kg h 2.OU/kgh + CCK 2.0 U/kgh FIGURE 6 Human pyloric response to the intravenous administration of secretin and cholecystokinin (CCK), alone and in combination. Data is expressed in millimeters Hg above intraabdominal pressure. Each bar represents mean ±SE of 10 experiments. The human pyloric responses to secretin, cholecystokinin and the combination were significant (P < 0.01). The response to the hormone combination was not greater than the individual hormone maximal responses (P > 0.05). stomach in each subject. The pylorus was identified as previously described, by the change in skin to mucosa PD at the gastroduodenal junction (1, 21, 22). The pyloric pressure responses to the intravenous administration of secretin and cholecystokinin alone and in combination in 10 subjects are shown in Fig. 6. All pressures are expressed using intragastric pressure as a zero reference. The basal pyloric pressure was 3.8±0.3 mm Hg (mean + SE). In response to secretin (1 U/kgh) pyloric pressure increased significantly above basal level to 10.3±+1.6 mm Hg (P < 0.01). Cholecystokinin (2 U/kgh) increased pyloric pressure to a maximal value of 10.4±1.9 mm Hg (P < 0.01). Secretin infusion at doses of 0.25, 0.50, and 2.0 U/kgh and cholecystokinin infusion at doses of 0.5, 1.0, and 3.0 U/kgh gave lower responses than to those doses cited above. The combination of secretin and cholecystokinin at maximal doses produced the greatest level of pyloric pressure, mm Hg. However, this response to the hormone combination did not significantly differ from the response to either hormone administered separately. The length of the pyloric high pressure zone was approximately 1.5 cm and did not change with hormonal stimulation. The pylorus was closely associated with an abrupt mean PD change of minus 33.4±2.6 mv as the tube was withdrawn from duodenum to stomach. This, too, was not altered by gastrointestinal hormones. The effect of gastrin I on pyloric pressure was next evaluated as shown in Fig. 7. During the intravenous infusion of gastrin I (1 Ag/kgh and 2 Lg/kgh) pyloric 1294 R. S. Fisher, W. Lipshutz, and S. Cohen T T pressure did nof significantly change from basal level. To determine whether gastrin I could affect the pyloric response to endogenous hormonal stimulation, the pyloric pressure was quantified during duodenal acidification (0.1 N HCl, 7.6 ce/min, 48 meq/h) alone, and during acidification with simultaneous gastrin I administration. In response to duodenal acidification alone pyloric pressure increased from its basal level of 3.8± 0.3 mm Hg to 16.1±3.6 mm Hg (P < 0.001). During gastrin I administration at 1,ug/kgh and simultaneous duodenal acidification, pyloric pressure increased to 10.8±2.6 mm Hg. At 2 iug/kgh of gastrin I, the pyloric pressure during duodenal acidification was only 4.7±1.8 mm Hg. Only the diminution in response to 2 Ag/kgh of gastrin was statistically significant when compared to the control response to duodenal acidification (P < 0.001). DISCUSSION Recently, it has been observed that individual gastrointestinal sphincters may have different responses to the gastrointestinal hormones, both in vitro and in vivo. The lower esophageal sphincter contracts in response to gastrin (1214) whereas both the ileocecal (15) and choledochal sphincters (16) are inhibited. Contrary to previous studies, the pylorus has now been shown to have properties similar to other gastrointestinal sphincters (1). In the resting state it is characterized by a zone of high pressure. This high pressure zone relaxes preceding antral peristalsis. Furthermore, in man, pyloric pressure increases in response to intraduodenal stimuli such as 0.1 N HCl, olive oil, and amino acids which are known to release secertin and cholecystokinin. In dogs pyloric contraction has also been reported in response to the administration of the octapeptide of cholecystokinin (3). The purpose of this study was to investigate the responses of pyloric circular muscle to the gastrointestinal hormones given singularly and in combination, in vitro, and to correlate these observations with the behavior of the human pyloric sphincter, in vivo. Our results indicated that pyloric circular muscle responds specifically to each gastrointestinal hormone tested. Secretin and cholecystokinin are agonists contracting the muscle, in vitro, and increasing pyloric pressure, in vivo. Gastrin I is an antagonist inhibiting the effects of the other hormones. Initial studies indicated that pyloric circular muscle responded at lower concentrations of cholecystokinin and developed a greater maximal active tension than muscle from either the adjacent antrum or duodenum. Secretin also contracted the pyloric muscle but the maximal active tension was less than that attained with cholecystokinin. The interaction of secretin and cholecystokinin showed an additive effect at submaximal concentrations but at the maximal concentration of both hormones, the response did not significantly exceed the response to cholecystokinin, alone. Thus, potentiation was not demonstrated by this criterion (23, 24). The
7 more potent response to the octapeptide of cholecystokinin as compared to the whole molecule was consistent with other effects of this residue (25) and suggested that contaminants present in the GIH Research Unit preparation of cholecystokinin were not solely responsible for the observed response (2628). These observations on muscle preparations correlated with the findings in man. The pyloric pressure increased in response to either cholecystokinin or secretin and the maximal response to each hormone was similar. The response to their combined administration did not differ significantly from the maximal response to each hormone given separately. Again, potentiation of the maximal response was not demonstrated. The increase in pyloric pressure during duodenal acidification was significantly greater than the response to hormonal administration. This observation may be explained by either the presence of impurities in the hormone preparation (2628) or the existence of other hormonal and neural mechanisms which may be involved in the acidification response. Further studies are required to resolve this point. The effect of gastrin I on pyloric muscle differed from that of either cholecystokinin or secretin. Gastrin I had no independent effect on pyloric circular muscle, but yet could antagonize the effect of cholecystokinin. In the presence of gastrin I, the cholecystokinin doseresponse curve was shifted to higher doses. The threshold dose was increased and the steep phase of linear response remained parallel to that of the doseresponse curve to cholecystokinin, alone. The maximum response was attained at higher doses of cholecystokinin. These features characterize competitive inhibition (29). We have shown previously that gastrin I antagonized the effect of secretin on pyloric muscle, but here the kinetics were noncompetitive (18, 29). These features of gastrin I antagonism for secretin and cholecystokinin on pyloric muscle are consistent with the gastrointestinal hormone receptor hypothesis proposed by Grossman (30). Observations on human pyloric function, in vivo, indicated that while gastrin I did not increase pyloric pressure significantly at any dose, it acted as an antagonist to the endogenous hormonal stimulation of the pylorus produced by perfusion of the duodenum with 0.1 N HCI. This observation was consistent with the antagonism of gastrin I for secretin and cholecystokinin on the pyloric muscle since duodenal acidification releases both hormones (4, 5). The relevance of these observations to the function of the pylorus must be interpreted cautiously. We have previously suggested that an important function of the pylorus is the prevention of duodenogastric reflux of bile because duodenal acidification in normals markedly reduced the amount of reflux of duodenal contents into the stomach (1). The most likely mechanism by which 12Iit 14 j 10 4 Basal Gastrin HCI HCI HCI 1IAg/kgh + + Gastrin Gastrin 1,4g/kgh 2,4g/kgh FIGURE 7 Human pyloric response to duodenal acidification alone and during the intravenous administration of graded doses of gastrin I. Data is expressed in millimeters Hg above intraabdominal pressure. Each bar represents the mean+se of 10 experiments. Gastrin I, administered alone, did not significantly alter pyloric pressure (P > 0.05). Duodenal acidification, alone significantly increased pyloric pressure to 16.1±3.6 mm Hg (P < 0.001). During gastrin I administration at 1,ug/kgh and 2,ug/kgh there was a graded decrease in the response to duodenal acidification. the pylorus serves this protective function would be the release of the duodenal hormones, secretin and cholecystokinin, in response to acidification. This would be analogous to the role of gastrin in maintaining lower esophageal sphincter competence (1214). However, other hormonal or neural pathways can not be excluded. Another possible function of the pylorus may be the control of gastric emptying. Secretin and cholecystokinin are thought to inhibit gastric emptying. Intraduodenal amino acids, known to release only cholecystokinin (7), and intraduodenal HCl known to release secretin (4), both delay gastric emptying (31). Exogenous secretin (32) and cholecystokinin (33) also delay gastric emptying. The role of the pylorus in gastric emptying is difficult to resolve because secretin and cholecystokinin not only increase pyloric tone but they also inhibit antral motility (19, 34). Therefore, the effect of these hormones upon emptying may be through either increased pyloric resistance or decreased antral propulsive force. Gastrin's effect on gastric emptying is even more complex. Since gastrin is known to increase antral motor activity (35) and inhibit pyloric contraction, we would predict an acceleration of gastric emptying. Quite the contrary, a diminution of gastric emptying has been reported (36, 37). However, this effect could be explained by a decrease in antral propulsive force rather than by an alteration of pyloric tone. Certainly, the physiologic role of the pylorus in the prevention of duodenogastric reflux and in the regulation of gastric emptying requires further investigation. Hormonal Regulation of Pyloric Sphincter Function 1295
8 ACKNOWLEDGM ENTS The authors wish to thank Miss Aileen Lahti and Mrs. Fe Green for their expert technical assistance. This work was supported by Training Grant TR15A from the Veterans Administration, Clinical Research Center Grant SMOIRR40 from the Division of Research Facilities and Resources, and Research Grant 1 RO1 AM from the National Institutes of Health. Dr. Cohen was supported, in part, by Clinical investigator funds from the Veterans Administration and by Research Career Development Award 1 K4 AM 70, from the National Institutes of Health. REFERENCES 1. Fisher, R. S., and S. Cohen The physiologic characteristics of the human pyloric sphincter. Gastroenterology. 64: Brink, B. M., J. F. Schlegel, and C. F. Code The pressure profile of the gastroduodenal junctional zone in dogs. Gut. 6: Isenberg, J. I., and A. Csendes Effect of octapeptide of cholecystokinin on canine pylorus pressure. Am. J. Physiol. 222: Nakajima, S., and D. F. Magee Influence of duodenal acidification on acid and pepsin secretion of the stomach in dogs. Am. J. Physiol. 218: Barbezat, G. O., and M. I. Grossman Cholecystokinin released by duodenal acidification. Gastroenterology. 60: 761. (Abstr.) 6. Johnson, L. R., and M. I. Grossman Effects of fat, secretin and cholecystokinin on histaminestimulated gastric secretion. Am. J. Physiol. 216: Go, V. L. W., A. F. Hofmann, and W. A. J. Summerskill Pancreozymin bioassay in man based on pancreatic enzyme secretion: potency of specific amino acids and other digestive products. J. Clini. Invest. 49: Young, J. D., L. Lazarus, and D. J. Chisholm Radioimmunoassay of pancreozymincholecystokinin in human serum. J. Nucl. Med. 10: Young, J. D., L. Lazarus, and D. J. Chisholm Secretin and pancreozymincholecystokinin after glucose. Lancet. 2: Chisholm, D. J., J. D. Young, and L. Lazarus The gastrointestinal stimulus to insulin release. I. Secretin. J. Clini. Invest. 48: Kraegen, E. W., D. J. Chisholm, J. D. Young, and L. Lazarus The gastrointestinal stimulus to insulin release. II. A dual action of secretin. J. Clin. Invest. 49: Castell, D. O., and L. D. Harris Hormonal control of gastroesophageal sphincter strength. N. Engl. J. Med. 282: Giles, G. R., M. D. Mason, C. Humphries, and C. G. Clark Action of gastrin on the lower oesophageal sphincter in man. Gut. 10: Cohen, S., and W. Lipshutz Hormonal regulation of human lower esophageal sphincter competence: interaction of gastrin and secretin. J. Clin. Invest. 50: Castell, D. O., S. Cohen, and L. D. Harris Response of human ileocecal sphincter to gastrin. Am. J. Physiol. 219: Lin, T. M Hepatic, cholecystokinetic and choledochal action of cholecystokinin, secretin, caerulein and gastrinlike peptides. Proc. Int. Cong. Physiol. Sci. 9: Abstr Lipshutz, W. H., and S. Cohen Physiologic determinants of lower esophageal sphincter function. Gastroenterology. 61: Lipshutz, W. H., and S. Cohen Interaction of gastrin I and secretin on gastrointestinal circular muscle. Am. J. Physiol. 222: Vagne, M., G. F. Stening, F. P. Brooks, and M. I. Grossman Synthetic secretin: comparison with natural secretin for potency and spectrum of physiological actions. Gastroenterology. 55: Snedecor, G. W., and W. G. Cochran Statistical Methods. Iowa State University Press, Ames. 21. Andersson, S., and M. I. Grossman Profile of ph, pressure, and potential difference at gastroduodenal junction in man. Gastroenterology. 49: Blackwood, W. D Pylorus identification. Gastroenterology 57: Henriksen, F. W Concept of potentiation. Gastroenterology. 57: Meyer, J. H., L. J. Spinzola, and M. I. Grossman Endogenous cholecystokinin potentiates exogenous secretin on pancreas of dog. Am. J. Physiol. 221: Rubin, B., A. M. Engel, A. M. Drungis, M. Dzelzalns, E. 0. Grigas, M. H. Waugh, and Yiacas Cholecystokininlike activities in guinea pigs and in dogs of the Cterminal octapeptide (SQ 19, 844) of cholecystokinin. J. Pharm. Sci. 58: Brown, J. C., R. A. Pederson, E. Jorpes, and V. Mutt Preparation of highly active enterogastrone. J. Physiol. (Lond.). 209: Lucien, H. W., Z. Itoh, and A. V. Sholly Inhibitory effects of purified enterogastrone, secretin, and cholecystokinin on histamine stimulated gastric acid secretion. Gastroenterology. 59: Magee, D. F., and B. Dutt Effect of pure and commercial cholecystokinin and an inhibitory polypeptide on gastric secretion. Am. J. Physiol. 222: Ariens, E. J., editor Molecular Pharmacology. Academic Press, Inc., New York Grossman, M. I Hypothesis: gastrin, cholecystokinin and' secretin act on one receptor. Lancet. 1: Thomas, J. E Mechanics and regulation of gastric emptying. Physiol. Rev. 37: Vagne, M., and C. Andre The effect of secretin on gastric emptying in man. Gastroenterology. 60: Chey, W. Y., S. Hitenant, J. Hendricks, and S. H. Lorber Effect of intestinal hormones on human gastric function. Gastroenterology. 54: (Abstr.) 34. Johnson, L. P., J. C. Brown, and D. F. Magee Effects of secretin and cholecystokininpancreozynim extracts on gastric motility in man. Gut. 7: Misiewicz, J. J., D. J. Holdstock, and S. L. Waller Motor responses of the human alimentary tract to nearmaximal infusions of pentagastrin. Gut. 8: Hunt, J. N., and N. Ramsbottom Effect of gastrin II on gastric emptying and secretion during a test meal. Br. Med. J. 4: Dozois, R. R., and K. A. Kelly Effect of a gastrin pentapeptide on canine gastric emptying of liquids. Am. J. Physiol. 221: R. S. Fisher, W. Lipshutz, and S. Cohen
EFFECT OF SECRETIN AND CHOLECYSTOKININ ON GASTRIC EMPTYING AND GASTRIC SECRETION IN MAN
 GA8TRONTROLOGY Copyright 197 by The Williams & Wilkins Co. Vol. 58, No.6 Fdnted in U.S.A. FFCT OF SCRTIN AND CHOLCYSTOKININ ON GASTRIC MPTYING AND GASTRIC SCRTION IN MAN W. Y. CHY, M.D., S. HITANANT, M.D.,
GA8TRONTROLOGY Copyright 197 by The Williams & Wilkins Co. Vol. 58, No.6 Fdnted in U.S.A. FFCT OF SCRTIN AND CHOLCYSTOKININ ON GASTRIC MPTYING AND GASTRIC SCRTION IN MAN W. Y. CHY, M.D., S. HITANANT, M.D.,
The abnormal lower oesophageal sphincter in pernicious anaemia'
 The abnormal lower oesophageal sphincter in pernicious anaemia' Gut, 1973, 14, 767-772 RAYMOND L. FARRELL, OTTO T. NEBEL, ARTHUR T. McGUIRE,2 AND DONALD 0. CASTELL3 From the Gastrointestinal Branch, Internal
The abnormal lower oesophageal sphincter in pernicious anaemia' Gut, 1973, 14, 767-772 RAYMOND L. FARRELL, OTTO T. NEBEL, ARTHUR T. McGUIRE,2 AND DONALD 0. CASTELL3 From the Gastrointestinal Branch, Internal
On the relationship between gastric ph and pressure
 Gut, 1979, 20, 59-63 On the relationship between gastric ph and pressure in the normal human lower oesophageal sphincter M. D. KAYE1 From the Gastroenterology Unit, Department of Medicine, University of
Gut, 1979, 20, 59-63 On the relationship between gastric ph and pressure in the normal human lower oesophageal sphincter M. D. KAYE1 From the Gastroenterology Unit, Department of Medicine, University of
PROGRESS IN GASTROENTEROLOGY
 GASTROENTEROLOGY Copyright 1971 by 'The Williams & Wilkins Co. Vol. 60, No.1 Printed in U. S. A. PROGRESS IN GASTROENTEROLOGY INTESTINAL HORMONES AS INHIBITORS OF GASTRIC SECRETION LEONARD R. JOHNSON,
GASTROENTEROLOGY Copyright 1971 by 'The Williams & Wilkins Co. Vol. 60, No.1 Printed in U. S. A. PROGRESS IN GASTROENTEROLOGY INTESTINAL HORMONES AS INHIBITORS OF GASTRIC SECRETION LEONARD R. JOHNSON,
Section Coordinator: Jerome W. Breslin, PhD, Assistant Professor of Physiology, MEB 7208, ,
 IDP Biological Systems Gastrointestinal System Section Coordinator: Jerome W. Breslin, PhD, Assistant Professor of Physiology, MEB 7208, 504-568-2669, jbresl@lsuhsc.edu Overall Learning Objectives 1. Characterize
IDP Biological Systems Gastrointestinal System Section Coordinator: Jerome W. Breslin, PhD, Assistant Professor of Physiology, MEB 7208, 504-568-2669, jbresl@lsuhsc.edu Overall Learning Objectives 1. Characterize
INHIBITION OF GASTRIC EMPTYING IS A PHYSIOLOGICAL ACTION OF CHOLECYSTOKININ
 GASTROENTEROLOGY68:1211-1217, 1975 Copyright 1975 by The Williams & Wilkins Co. Vol. 68, No. 5, Part 1 Printed in U.S.A. NHBTON OF GASTRC EMPTYNG S A PHYSOLOGCAL ACTON OF CHOLECYSTOKNN HALE T. DEBAS, M.D.,
GASTROENTEROLOGY68:1211-1217, 1975 Copyright 1975 by The Williams & Wilkins Co. Vol. 68, No. 5, Part 1 Printed in U.S.A. NHBTON OF GASTRC EMPTYNG S A PHYSOLOGCAL ACTON OF CHOLECYSTOKNN HALE T. DEBAS, M.D.,
University of Buea. Faculty of Health Sciences. Programme in Medicine
 Faculty of Health Sciences University of Buea Wednesday, 28 th January 2009 Time: 8 00-10 00 Programme in Medicine MED 303 (Gastrointestinal Physiology) EXAMS (2008-2009) Identify the letter of the choice
Faculty of Health Sciences University of Buea Wednesday, 28 th January 2009 Time: 8 00-10 00 Programme in Medicine MED 303 (Gastrointestinal Physiology) EXAMS (2008-2009) Identify the letter of the choice
Effect of acid infusion into various levels of the intestine on gastric and pancreatic secretion in the cat
 Gut, 1969, 10, 749-753 Effect of acid infusion into various levels of the intestine on gastric and pancreatic secretion in the cat S. J. KONTUREK, J. DUBIEL, AND B. GABRY9 From the Department of Medicine,
Gut, 1969, 10, 749-753 Effect of acid infusion into various levels of the intestine on gastric and pancreatic secretion in the cat S. J. KONTUREK, J. DUBIEL, AND B. GABRY9 From the Department of Medicine,
Diversion of bile and pancreatic juices from the duodenum to the jejunum has
 GASTROENTEROLOGY Copyright 1969 by The Williams & Wilkins Co. Vol. 56, No.4 Printed in U.S.A. EFFECT OF EXCLUSION, ACIDIFICATION, AND EXCISION OF THE DUODENUM ON GASTRIC ACID SECRETION AND THE PRODUCTION
GASTROENTEROLOGY Copyright 1969 by The Williams & Wilkins Co. Vol. 56, No.4 Printed in U.S.A. EFFECT OF EXCLUSION, ACIDIFICATION, AND EXCISION OF THE DUODENUM ON GASTRIC ACID SECRETION AND THE PRODUCTION
to food and histamine
 Gut, 97,, 53-57 Maximal acid response of Pavlov pouches to food and histamine A. MARVIN BROOKS AND MORTON I. GROSSMAN From the Veterans Administration Center and UCLA School of Medicine, Departments of
Gut, 97,, 53-57 Maximal acid response of Pavlov pouches to food and histamine A. MARVIN BROOKS AND MORTON I. GROSSMAN From the Veterans Administration Center and UCLA School of Medicine, Departments of
Role of Gastrin Supersensitivity in the Pathogenesis of Lower Esophageal Sphincter Hypertension in Achalasia
 Role of Gastrin Supersensitivity in the Pathogenesis of Lower Esophageal Sphincter Hypertension in Achalasia SIDNEY COHEN, WILLIAM LIPsHurz, and WILLIAM HUGHES From the Gastrointestinal Section, Department
Role of Gastrin Supersensitivity in the Pathogenesis of Lower Esophageal Sphincter Hypertension in Achalasia SIDNEY COHEN, WILLIAM LIPsHurz, and WILLIAM HUGHES From the Gastrointestinal Section, Department
EFFECT OF VAGOTOMY ON PANCREATIC SECRETION STIMULATED BY ENDOGENOUS AND EXOGENOUS SECRETIN
 GASTROENTEROLOGY Copyright,. 1971 by The Williams & Wilkins Co. Vol. 60, No. 3 P>-inted in U. S. A. EFFECT OF VAGOTOMY ON PANCREATIC SECRETION STIMULATED BY ENDOGENOUS AND EXOGENOUS SECRETIN HARRIS J.
GASTROENTEROLOGY Copyright,. 1971 by The Williams & Wilkins Co. Vol. 60, No. 3 P>-inted in U. S. A. EFFECT OF VAGOTOMY ON PANCREATIC SECRETION STIMULATED BY ENDOGENOUS AND EXOGENOUS SECRETIN HARRIS J.
OPERATIVE TREATMENT OF ULCER DISEASE
 Página 1 de 8 Copyright 2001 Lippincott Williams & Wilkins Greenfield, Lazar J., Mulholland, Michael W., Oldham, Keith T., Zelenock, Gerald B., Lillemoe, Keith D. Surgery: Scientific Principles & Practice,
Página 1 de 8 Copyright 2001 Lippincott Williams & Wilkins Greenfield, Lazar J., Mulholland, Michael W., Oldham, Keith T., Zelenock, Gerald B., Lillemoe, Keith D. Surgery: Scientific Principles & Practice,
SIMULTANEOUS MEASUREMENT OF THE PANCREATIC AND BILIARY RESPONSE TO CCK AND SECRETIN
 GASTROENTEROLOGY 70:403-407, 1976 Copyright 1976 by The Williams & Wilkins Co. Vol. 70, No. 3 Printed in U.S.A. SIMULTANEOUS MEASUREMENT OF THE PANCREATIC AND BILIARY RESPONSE TO CCK AND SECRETIN Primate
GASTROENTEROLOGY 70:403-407, 1976 Copyright 1976 by The Williams & Wilkins Co. Vol. 70, No. 3 Printed in U.S.A. SIMULTANEOUS MEASUREMENT OF THE PANCREATIC AND BILIARY RESPONSE TO CCK AND SECRETIN Primate
SPHINCTER OF ODDI DYSFUNCTION (SOD)
 SPHINCTER OF ODDI DYSFUNCTION (SOD) Sphincter of Oddi dysfunction refers to structural or functional disorders involving the biliary sphincter that may result in impedance of bile and pancreatic juice
SPHINCTER OF ODDI DYSFUNCTION (SOD) Sphincter of Oddi dysfunction refers to structural or functional disorders involving the biliary sphincter that may result in impedance of bile and pancreatic juice
Asma Karameh. -Shatha Al-Jaberi محمد خطاطبة -
 -2 Asma Karameh -Shatha Al-Jaberi محمد خطاطبة - 1 P a g e Gastrointestinal motilities Chewing: once you introduce the first bolus to the mouth you started what we call chewing reflex appears by muscle
-2 Asma Karameh -Shatha Al-Jaberi محمد خطاطبة - 1 P a g e Gastrointestinal motilities Chewing: once you introduce the first bolus to the mouth you started what we call chewing reflex appears by muscle
Digestive System Module 4: The Stomach *
 OpenStax-CNX module: m49286 1 Digestive System Module 4: The * Donna Browne Based on The by OpenStax This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License 4.0
OpenStax-CNX module: m49286 1 Digestive System Module 4: The * Donna Browne Based on The by OpenStax This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License 4.0
The effect of metoclopramide on gastroduodenal
 Gut, 1971, 12, 158-163 The effect of metoclopramide on gastroduodenal and gallbladder contractions A. G. JOHNSON From the Department of Surgery, Charing Cross Hospital Medical School, London SUMMARY The
Gut, 1971, 12, 158-163 The effect of metoclopramide on gastroduodenal and gallbladder contractions A. G. JOHNSON From the Department of Surgery, Charing Cross Hospital Medical School, London SUMMARY The
Digestive System. Digestive System. Digestion is the process of reducing food to small molecules that can be absorbed into the body.
 Digestive System Digestion is the process of reducing food to small molecules that can be absorbed into the body. 2 Types of Digestion Mechanical digestion physical breakdown of food into small particles
Digestive System Digestion is the process of reducing food to small molecules that can be absorbed into the body. 2 Types of Digestion Mechanical digestion physical breakdown of food into small particles
Intestinal phase of gastric secretion in patients with
 Gut, 1978, 19, 321-326 Intestinal phase of gastric secretion in patients with duodenal ulcer S. J. KONTUREK1, N. KWIECIEN, W. OBTULOWICZ, E. SITO, AND J. OLEKSY From the Institute ofphysiology, Medical
Gut, 1978, 19, 321-326 Intestinal phase of gastric secretion in patients with duodenal ulcer S. J. KONTUREK1, N. KWIECIEN, W. OBTULOWICZ, E. SITO, AND J. OLEKSY From the Institute ofphysiology, Medical
INTRODUCTION TO GASTROINTESTINAL FUNCTIONS
 1 INTRODUCTION TO GASTROINTESTINAL FUNCTIONS 2 Learning outcomes List two main components that make up the digestive system Describe the 6 essential functions of the GIT List factors (neurological, hormonal
1 INTRODUCTION TO GASTROINTESTINAL FUNCTIONS 2 Learning outcomes List two main components that make up the digestive system Describe the 6 essential functions of the GIT List factors (neurological, hormonal
MECHANISM BY WHICH FAT IN THE UPPER SMALL INTESTINE INHIBITS GASTRIC ACID
 GASTROENTEROLOGY Copyright 1969 by The Williams & Wilkins Co. Vol. 56, No.3 Printea in U.S.A. MECHANISM BY WHICH FAT IN THE UPPER SMALL INTESTINE INHIBITS GASTRIC ACID H. T. DEBAS, M.D., B. S. BEDI, M.B.,
GASTROENTEROLOGY Copyright 1969 by The Williams & Wilkins Co. Vol. 56, No.3 Printea in U.S.A. MECHANISM BY WHICH FAT IN THE UPPER SMALL INTESTINE INHIBITS GASTRIC ACID H. T. DEBAS, M.D., B. S. BEDI, M.B.,
Chapter 20 The Digestive System Exam Study Questions
 Chapter 20 The Digestive System Exam Study Questions 20.1 Overview of GI Processes 1. Describe the functions of digestive system. 2. List and define the four GI Processes: 20.2 Functional Anatomy of the
Chapter 20 The Digestive System Exam Study Questions 20.1 Overview of GI Processes 1. Describe the functions of digestive system. 2. List and define the four GI Processes: 20.2 Functional Anatomy of the
Digestive System 7/15/2015. Outline Digestive System. Digestive System
 Digestive System Biology 105 Lecture 18 Chapter 15 Outline Digestive System I. Functions II. Layers of the GI tract III. Major parts: mouth, pharynx, esophagus, stomach, small intestine, large intestine,
Digestive System Biology 105 Lecture 18 Chapter 15 Outline Digestive System I. Functions II. Layers of the GI tract III. Major parts: mouth, pharynx, esophagus, stomach, small intestine, large intestine,
Digestive Lecture Test Questions Set 4
 Digestive Lecture Test Questions Set 4 1. Which of the following is not associated directly with the small intestine: a. villi b. circular folds c. microvilli d. haustrae e. secretin 2. The largest (longest)
Digestive Lecture Test Questions Set 4 1. Which of the following is not associated directly with the small intestine: a. villi b. circular folds c. microvilli d. haustrae e. secretin 2. The largest (longest)
All organisms must obtain and process essential nutrients (food) *** Exception: Venus Fly Traps undergo photosynthesis but needs source of nitrogen
 All organisms must obtain and process essential nutrients (food) AUTOTROPHS self feeder makes their own food eg. Plants do not require a digestive tract *** Exception: Venus Fly Traps undergo photosynthesis
All organisms must obtain and process essential nutrients (food) AUTOTROPHS self feeder makes their own food eg. Plants do not require a digestive tract *** Exception: Venus Fly Traps undergo photosynthesis
The Digestive System. Chapter 25
 The Digestive System Chapter 25 Introduction Structure of the digestive system A tube that extends from mouth to anus Accessory organs are attached Functions include Ingestion Movement Digestion Absorption
The Digestive System Chapter 25 Introduction Structure of the digestive system A tube that extends from mouth to anus Accessory organs are attached Functions include Ingestion Movement Digestion Absorption
(b) Stomach s function 1. Dilution of food materials 2. Acidification of food (absorption of dietary Fe in small intestine) 3. Partial chemical digest
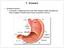 (1) General features a) Stomach is widened portion of gut-tube: between tubular and spherical; Note arranged of smooth muscle tissue in muscularis externa. 1 (b) Stomach s function 1. Dilution of food
(1) General features a) Stomach is widened portion of gut-tube: between tubular and spherical; Note arranged of smooth muscle tissue in muscularis externa. 1 (b) Stomach s function 1. Dilution of food
Overview of Gastroenterology
 Harvard-MIT Division of Health Sciences and Technology HST.121: Gastroenterology, Fall 2005 Instructors: Dr. Jonathan Glickman Overview of Gastroenterology Gastroenterology Made Really Simple! Food Waste
Harvard-MIT Division of Health Sciences and Technology HST.121: Gastroenterology, Fall 2005 Instructors: Dr. Jonathan Glickman Overview of Gastroenterology Gastroenterology Made Really Simple! Food Waste
THE NORMAL HUMAN ESOPHAGEAL MUCOSA: A HISTOLOGICAL REAPPRAISAL
 GASTROENTEROLOGY 68:40-44, 1975 Copyright 1975 by The Williams & Wilkins Co. Vol. 68, No.1 Printed in U.S.A. THE NORMAL HUMAN ESOPHAGEAL MUCOSA: A HISTOLOGICAL REAPPRAISAL WILFRED M. WEINSTEIN, M.D., EARL
GASTROENTEROLOGY 68:40-44, 1975 Copyright 1975 by The Williams & Wilkins Co. Vol. 68, No.1 Printed in U.S.A. THE NORMAL HUMAN ESOPHAGEAL MUCOSA: A HISTOLOGICAL REAPPRAISAL WILFRED M. WEINSTEIN, M.D., EARL
Two main groups Alimentary canal continuous coiled hollow tube Accessory digestive organs
 Digestion Breakdown of ingested food Absorption of nutrients into the blood Metabolism Production of cellular energy (ATP) Constructive and degradative cellular activities Two main groups Alimentary canal
Digestion Breakdown of ingested food Absorption of nutrients into the blood Metabolism Production of cellular energy (ATP) Constructive and degradative cellular activities Two main groups Alimentary canal
Gastrointestinal Physiology
 Gastrointestinal Physiology Digestion and Absorption Definition Digestion: i Food dis dissolved d and broken down. Physical digestion: Propulsion and mixing of food by muscle in the alimentary tract. Chemical
Gastrointestinal Physiology Digestion and Absorption Definition Digestion: i Food dis dissolved d and broken down. Physical digestion: Propulsion and mixing of food by muscle in the alimentary tract. Chemical
Chapter 20 The Digestive System Exam Study Questions
 Chapter 20 The Digestive System Exam Study Questions 20.1 Overview of GI Processes 1. Describe the functions of digestive system. 2. List and define the four GI Processes: 20.2 Functional Anatomy of the
Chapter 20 The Digestive System Exam Study Questions 20.1 Overview of GI Processes 1. Describe the functions of digestive system. 2. List and define the four GI Processes: 20.2 Functional Anatomy of the
Our gut reactions to food or, gut reactions - to food
 Key concepts in Digestion. Our gut reactions to food or, gut reactions to food Prof. Barry Campbell Cellular & Molecular Physiology email: bjcampbl@liv.ac.uk http://pcwww.liv.ac.uk/~bjcampbl Swallowing
Key concepts in Digestion. Our gut reactions to food or, gut reactions to food Prof. Barry Campbell Cellular & Molecular Physiology email: bjcampbl@liv.ac.uk http://pcwww.liv.ac.uk/~bjcampbl Swallowing
Why would fatty foods aggravate the patient s RUQ pain? What effect does cholecystokinin (CCK) have on gastric emptying?
 CASE 28 A 43-year-old woman presents to the emergency department with the acute onset of abdominal pain. Her pain is located to the right upper quadrant (RUQ) and radiates to the right shoulder. She reports
CASE 28 A 43-year-old woman presents to the emergency department with the acute onset of abdominal pain. Her pain is located to the right upper quadrant (RUQ) and radiates to the right shoulder. She reports
HISTAMINE RECEPTORS IN ESOPHAGEAL SMOOTH MUSCLE OF THE OPOSSUM
 GASTROENTEROLOGY 70:1071-1075, 1976 Copyright 1976 by The Williams & Wilkins Co. Vol. 70, No.6 Printed in U.S.A. HISTAMINE RECEPTORS IN ESOPHAGEAL SMOOTH MUSCLE OF THE OPOSSUM DAVID J. DE CARLE, M.B.,
GASTROENTEROLOGY 70:1071-1075, 1976 Copyright 1976 by The Williams & Wilkins Co. Vol. 70, No.6 Printed in U.S.A. HISTAMINE RECEPTORS IN ESOPHAGEAL SMOOTH MUSCLE OF THE OPOSSUM DAVID J. DE CARLE, M.B.,
COMPARATIVE EFFECTS OF METOCLOPRAMIDE AND BETHANECHOL ON LOWER ESOPHAGEAL SPHINCTER PRESSURE IN REFLUX PATIENTS
 GASTROENTEROLOGY 68: 111-1118, 1975 Copyright 1975 by The Williams & Wilkins Co. Vol. 68, No. 5, Part 1 Printed in U.S.A. COMPARATIVE EFFECTS OF METOCLOPRAMIDE AND BETHANECHOL ON LOWER ESOPHAGEAL SPHINCTER
GASTROENTEROLOGY 68: 111-1118, 1975 Copyright 1975 by The Williams & Wilkins Co. Vol. 68, No. 5, Part 1 Printed in U.S.A. COMPARATIVE EFFECTS OF METOCLOPRAMIDE AND BETHANECHOL ON LOWER ESOPHAGEAL SPHINCTER
in gastric emptying' of the stomach was recorded by an electronic transducer (T1) attached by an air-filled tube to the second limb of
 Gut, 1963, 4, 174 Function of the pylorus and pyloric antrum in gastric emptying' A. K. ARMITAGE AND A. C. B. DEAN From the Department of Surgery, King's College Hospital Medical School, London EDITORIAL
Gut, 1963, 4, 174 Function of the pylorus and pyloric antrum in gastric emptying' A. K. ARMITAGE AND A. C. B. DEAN From the Department of Surgery, King's College Hospital Medical School, London EDITORIAL
Nutrition. Autotrophs. plants, some protists & bacteria producers
 Nutrition Autotrophs plants, some protists & bacteria producers Nutrition Heterotrophs animals, fungi, some protists & bacteria consumers Animal Nutrition Most obtain food by ingestion take in their food
Nutrition Autotrophs plants, some protists & bacteria producers Nutrition Heterotrophs animals, fungi, some protists & bacteria consumers Animal Nutrition Most obtain food by ingestion take in their food
(A) Diarrhea. (B) Stomach cramps. (C) Dehydration due to excess fluid loss. (D) A, B, and C are correct. (E) Only answer B is correct.
 Human Anatomy - Problem Drill 21: The Digestive System Question No. 1 of 10 1. A 26-year-old male is treated in the emergency department for severe gastrointestinal disturbance. Which of the following
Human Anatomy - Problem Drill 21: The Digestive System Question No. 1 of 10 1. A 26-year-old male is treated in the emergency department for severe gastrointestinal disturbance. Which of the following
Secretion by Pentagastrin in Duodenal Ulcer
 Increased Sensitivity to Stimulation of Acid Secretion by Pentagastrin in Duodenal Ulcer JON I. ISENBERG, MORTON I. GROSSMAN, VERNON MAXWELL, and JOHN H. WALSH From Medical Service and Research Service,
Increased Sensitivity to Stimulation of Acid Secretion by Pentagastrin in Duodenal Ulcer JON I. ISENBERG, MORTON I. GROSSMAN, VERNON MAXWELL, and JOHN H. WALSH From Medical Service and Research Service,
Digestive System. - Food is ingested
 11 V. Digestive Processes in the Mouth - Food is ingested - Mechanical digestion begins (chewing) - Salivary amylase begins chemical breakdown of starch - Propulsion is initiated by Deglutition (Swallowing)
11 V. Digestive Processes in the Mouth - Food is ingested - Mechanical digestion begins (chewing) - Salivary amylase begins chemical breakdown of starch - Propulsion is initiated by Deglutition (Swallowing)
Bio& 242 Unit 1 / Lecture 4
 Bio& 242 Unit 1 / Lecture 4 system: Gastric hormones GASTRIN: Secretion: By enteroendocrine (G) in gastric pits of the mucosa. Stimulus: Stomach distention and acid ph of chyme causes Gastrin. Action:
Bio& 242 Unit 1 / Lecture 4 system: Gastric hormones GASTRIN: Secretion: By enteroendocrine (G) in gastric pits of the mucosa. Stimulus: Stomach distention and acid ph of chyme causes Gastrin. Action:
Gastrointestinal Anatomy and Physiology. Bio 219 Napa Valley College Dr. Adam Ross
 Gastrointestinal Anatomy and Physiology Bio 219 Napa Valley College Dr. Adam Ross Functions of digestive system Digestion Breakdown of food (chemically) using enzymes, acid, and water Absorption Nutrients,
Gastrointestinal Anatomy and Physiology Bio 219 Napa Valley College Dr. Adam Ross Functions of digestive system Digestion Breakdown of food (chemically) using enzymes, acid, and water Absorption Nutrients,
What part of the gastrointestinal (GI) tract is composed of striated muscle and smooth muscle?
 CASE 29 A 34-year-old man presents to his primary care physician with the complaint of increased difficulty swallowing both solid and liquid foods. He notices that he sometimes has more difficulty when
CASE 29 A 34-year-old man presents to his primary care physician with the complaint of increased difficulty swallowing both solid and liquid foods. He notices that he sometimes has more difficulty when
Metoclopramide in gastrooesophageal reflux
 Metoclopramide in gastrooesophageal reflux C. STANCIU AND JOHN R. BENNETT From the Gastrointestinal Unit, Hull Royal Infirmary Gut, 1973, 14, 275-279 SUMMARY In 3 patients with gastrooesophageal reflux,
Metoclopramide in gastrooesophageal reflux C. STANCIU AND JOHN R. BENNETT From the Gastrointestinal Unit, Hull Royal Infirmary Gut, 1973, 14, 275-279 SUMMARY In 3 patients with gastrooesophageal reflux,
Chapter 14: The Digestive System
 Chapter 14: The Digestive System Digestive system consists of Muscular tube (digestive tract) alimentary canal Accessory organs teeth, tongue, glandular organs 6 essential activities 1. 2. 3. 4. 5. 6.
Chapter 14: The Digestive System Digestive system consists of Muscular tube (digestive tract) alimentary canal Accessory organs teeth, tongue, glandular organs 6 essential activities 1. 2. 3. 4. 5. 6.
Lab 5 Digestion and Hormones of Digestion. 7/16/2015 MDufilho 1
 Lab 5 Digestion and Hormones of Digestion 1 Figure 23.1 Alimentary canal and related accessory digestive organs. Mouth (oral cavity) Tongue* Parotid gland Sublingual gland Submandibular gland Salivary
Lab 5 Digestion and Hormones of Digestion 1 Figure 23.1 Alimentary canal and related accessory digestive organs. Mouth (oral cavity) Tongue* Parotid gland Sublingual gland Submandibular gland Salivary
Introduction. The oxytocin/ / vasopressin receptor antagonist, atosiban, delays the gastric emptying of a semisolid meal compared to saline in human
 The oxytocin/ / vasopressin receptor antagonist, atosiban, delays the gastric emptying of a semisolid meal compared to saline in human Bodil Ohlsson, et. al., Sweden BMC Gastroenterology, 2006 Presented
The oxytocin/ / vasopressin receptor antagonist, atosiban, delays the gastric emptying of a semisolid meal compared to saline in human Bodil Ohlsson, et. al., Sweden BMC Gastroenterology, 2006 Presented
INFLUENCE OF INTRAJEJUNAL GLUCOSE ON PANCREATIC EXOCRINE FUNCTION IN MAN
 GASTROENTEROLOGY Copyright @ 1971 by The Williams & Wilkins Co. Vol. 60, No.5 Printed in U.S.A. INFLUENCE OF INTRAJEJUNAL GLUCOSE ON PANCREATIC EXOCRINE FUNCTION IN MAN WALTER P, DYCK, M.D. Department
GASTROENTEROLOGY Copyright @ 1971 by The Williams & Wilkins Co. Vol. 60, No.5 Printed in U.S.A. INFLUENCE OF INTRAJEJUNAL GLUCOSE ON PANCREATIC EXOCRINE FUNCTION IN MAN WALTER P, DYCK, M.D. Department
Chapter 26 The Digestive System
 Chapter 26 The Digestive System Digestive System Gastroenterology is the study of the stomach and intestine. Digestion Catabolism Absorption Anabolism The actions of the digestive system are controlled
Chapter 26 The Digestive System Digestive System Gastroenterology is the study of the stomach and intestine. Digestion Catabolism Absorption Anabolism The actions of the digestive system are controlled
For more information about how to cite these materials visit
 Author: John Williams, M.D., Ph.D., 2009 License: Unless otherwise noted, this material is made available under the terms of the Creative Commons Attribution Non-commercial Share Alike 3.0 License: http://creativecommons.org/licenses/by-nc-sa/3.0/
Author: John Williams, M.D., Ph.D., 2009 License: Unless otherwise noted, this material is made available under the terms of the Creative Commons Attribution Non-commercial Share Alike 3.0 License: http://creativecommons.org/licenses/by-nc-sa/3.0/
Official Publication of the American Gastroe1lterological Association
 GASTROENTEROLOGY@ Official Publication of the American Gastroe1lterological Association COPYRIGHT 1977 THE WILLIAMS & WILKINS Co. Vol. 72 March 1977 Number 3 ALIMENTARY TRACT EVIDENCE THAT ABNORMAL MYOELECTRICAL
GASTROENTEROLOGY@ Official Publication of the American Gastroe1lterological Association COPYRIGHT 1977 THE WILLIAMS & WILKINS Co. Vol. 72 March 1977 Number 3 ALIMENTARY TRACT EVIDENCE THAT ABNORMAL MYOELECTRICAL
The Small Intestine. The pyloric sphincter at the bottom of the stomach opens, squirting small amounts of food into your small intestine.
 The Small Intestine The pyloric sphincter at the bottom of the stomach opens, squirting small amounts of food into your small intestine. approximately six metres (the longest section of your digestive
The Small Intestine The pyloric sphincter at the bottom of the stomach opens, squirting small amounts of food into your small intestine. approximately six metres (the longest section of your digestive
KRISHNA TEJA PHARMACY COLLEGE HUMAN ANATOMY AND PHYSIOLOGY. DIGESTIVE SYSTEM Dr.B.Jyothi
 KRISHNA TEJA PHARMACY COLLEGE HUMAN ANATOMY AND PHYSIOLOGY DIGESTIVE SYSTEM Dr.B.Jyothi Prof, Dept. Of Pharmacology KTPC The Digestive System Food undergoes six major processes: 1. Ingestion : process
KRISHNA TEJA PHARMACY COLLEGE HUMAN ANATOMY AND PHYSIOLOGY DIGESTIVE SYSTEM Dr.B.Jyothi Prof, Dept. Of Pharmacology KTPC The Digestive System Food undergoes six major processes: 1. Ingestion : process
Histologically the pyloric glands and the glands of Brunner in the. Ivy(7) and his co-workers seem to believe, at the present time, in the
 6I2.323[6I2.33 6I2.OI5.2I THE SMALL INTESTINE AND GASTRIC SECRETION. (With special reference to Brunner's Glands.) BY MARGARET M. MURRAY. (From the Department of Physiology, Bedford College.) EVIDENCE
6I2.323[6I2.33 6I2.OI5.2I THE SMALL INTESTINE AND GASTRIC SECRETION. (With special reference to Brunner's Glands.) BY MARGARET M. MURRAY. (From the Department of Physiology, Bedford College.) EVIDENCE
A COMPARATIVE STUDY OF THE EFFECTS OF VERAPAMIL AND CIMETIDINE ON ISOLATED RAT STOMACH STRIP
 A COMPARATIVE STUDY OF THE EFFECTS OF VERAPAMIL AND CIMETIDINE ON ISOLATED RAT STOMACH STRIP ABSTRACT Pages with reference to book, From 263 To 266 Talat Ahmed Nishat ( Department of Pharmacology, Rawalpindi
A COMPARATIVE STUDY OF THE EFFECTS OF VERAPAMIL AND CIMETIDINE ON ISOLATED RAT STOMACH STRIP ABSTRACT Pages with reference to book, From 263 To 266 Talat Ahmed Nishat ( Department of Pharmacology, Rawalpindi
DIGESTIVE SYSTEM CLASS NOTES. tube along with several
 DIGESTIVE SYSTEM CLASS NOTES Digestion Breakdown of food and the of nutrients in the bloodstream. Metabolism Production of for and cellular activities. The digestive system is composed of the canal which
DIGESTIVE SYSTEM CLASS NOTES Digestion Breakdown of food and the of nutrients in the bloodstream. Metabolism Production of for and cellular activities. The digestive system is composed of the canal which
PHYSIOLOGY OF THE DIGESTIVE SYSTEM
 Student Name CHAPTER 26 PHYSIOLOGY OF THE DIGESTIVE SYSTEM D igestion is the process of breaking down complex nutrients into simpler units suitable for absorption. It involves two major processes: mechanical
Student Name CHAPTER 26 PHYSIOLOGY OF THE DIGESTIVE SYSTEM D igestion is the process of breaking down complex nutrients into simpler units suitable for absorption. It involves two major processes: mechanical
2. A digestive organ that is not part of the alimentary canal is the: a. stomach b. liver c. small intestine d. large intestine e.
 Chapter 14 The Digestive System and Body Metabolism Review Questions Multiple Choice: 1. Which of the following terms are synonyms? a. Gastrointestinal tract b. Digestive system c. Digestive tract d. Alimentary
Chapter 14 The Digestive System and Body Metabolism Review Questions Multiple Choice: 1. Which of the following terms are synonyms? a. Gastrointestinal tract b. Digestive system c. Digestive tract d. Alimentary
Serum gastrin and gastric acid responses to meals at various ph levels in man
 Gut, 1974, 15, 526-530 Serum gastrin and gastric acid responses to meals at various ph levels in man S. J. KONTURK,1 J. BIRNAT, AND J. OLKSY From the Institute ofphysiology, Medical Academy, Krak6w, Poland,
Gut, 1974, 15, 526-530 Serum gastrin and gastric acid responses to meals at various ph levels in man S. J. KONTURK,1 J. BIRNAT, AND J. OLKSY From the Institute ofphysiology, Medical Academy, Krak6w, Poland,
BIO 139 ANATOMY AND PHYSIOLOGY II
 BIO 139 ANATOMY AND PHYSIOLOGY II THE DIGESTIVE SYSTEM LAB ANALOGY PAGES 248-265 MARY CATHERINE FLATH, Ph.D. DIGESTIVE ORGANS ALIMENTARY CANAL MOUTH PHARYNX ESOPHAGUS STOMACH SMALL INTESTINE LARGE INTESTINE
BIO 139 ANATOMY AND PHYSIOLOGY II THE DIGESTIVE SYSTEM LAB ANALOGY PAGES 248-265 MARY CATHERINE FLATH, Ph.D. DIGESTIVE ORGANS ALIMENTARY CANAL MOUTH PHARYNX ESOPHAGUS STOMACH SMALL INTESTINE LARGE INTESTINE
Digestive System Notes. Biology - Mrs. Kaye
 Digestive System Notes Biology - Mrs. Kaye Digestion There are two kinds of digestion: Mechanical digestion - the physical breakdown of food into smaller pieces to increase surface area for more enzyme
Digestive System Notes Biology - Mrs. Kaye Digestion There are two kinds of digestion: Mechanical digestion - the physical breakdown of food into smaller pieces to increase surface area for more enzyme
Sphincters heartburn diaphragm The Stomach gastric glands pepsin, chyme The Small Intestine 1-Digestion Is Completed in the Small Intestine duodenum
 Sphincters are muscles that encircle tubes and act as valves. The tubes close when the sphincters contract and they open when the sphincters relax. When food or saliva is swallowed, the sphincter relaxes
Sphincters are muscles that encircle tubes and act as valves. The tubes close when the sphincters contract and they open when the sphincters relax. When food or saliva is swallowed, the sphincter relaxes
BIO 139 ANATOMY AND PHYSIOLOGY II. THE DIGESTIVE SYSTEM LAB ANALOGY PAGES MARY CATHERINE FLATH, Ph.D.
 BIO 139 ANATOMY AND PHYSIOLOGY II THE DIGESTIVE SYSTEM LAB ANALOGY PAGES 248-265 MARY CATHERINE FLATH, Ph.D. DIGESTIVE ORGANS ALIMENTARY CANAL MOUTH PHARYNX ESOPHAGUS STOMACH SMALL INTESTINE LARGE INTESTINE
BIO 139 ANATOMY AND PHYSIOLOGY II THE DIGESTIVE SYSTEM LAB ANALOGY PAGES 248-265 MARY CATHERINE FLATH, Ph.D. DIGESTIVE ORGANS ALIMENTARY CANAL MOUTH PHARYNX ESOPHAGUS STOMACH SMALL INTESTINE LARGE INTESTINE
1-It is to prevent back flow of fecal content from colon into small intestine.
 Function of the ileocecal valve: 1-It is to prevent back flow of fecal content from colon into small intestine. 2-The wall of the ileum for several centimeters preceding valve has a thickened muscular
Function of the ileocecal valve: 1-It is to prevent back flow of fecal content from colon into small intestine. 2-The wall of the ileum for several centimeters preceding valve has a thickened muscular
Influence of Successive Vagal Stimulations on
 Influence of Successive Vagal Stimulations on Contractions in Esophageal Smooth Muscle of Opossum JASWANT S. GIDDA and RAJ K. GOYAL, The Charles A. Dana Research Institute and the Harvard-Thorndike Laboratory
Influence of Successive Vagal Stimulations on Contractions in Esophageal Smooth Muscle of Opossum JASWANT S. GIDDA and RAJ K. GOYAL, The Charles A. Dana Research Institute and the Harvard-Thorndike Laboratory
Urinary system. Kidney anatomy Renal cortex Renal. Nephrons
 Urinary system Aids homeostasis by removing cellular wastes and foreign compounds, and maintains salt and water balance of plasma Kidney anatomy Renal cortex Renal pelvis Renal medulla Cortex Ureter Medulla
Urinary system Aids homeostasis by removing cellular wastes and foreign compounds, and maintains salt and water balance of plasma Kidney anatomy Renal cortex Renal pelvis Renal medulla Cortex Ureter Medulla
Topic 6: Human Physiology
 Topic 6: Human Physiology 6.1 Digestion and Absorption D.1 Human Nutrition D.2 Digestion Essential Understandings: The structure of the digestive system allows it to move, digest, and absorb food. A balanced
Topic 6: Human Physiology 6.1 Digestion and Absorption D.1 Human Nutrition D.2 Digestion Essential Understandings: The structure of the digestive system allows it to move, digest, and absorb food. A balanced
Includes mouth, pharynx, esophagus, stomach, small intestine, large intestine, rectum, anus. Salivary glands, liver, gallbladder, pancreas
 Chapter 14 The Digestive System and Nutrition Digestive System Brings Nutrients Into the Body The digestive system includes Gastrointestinal (GI) tract (hollow tube) Lumen: space within this tube Includes
Chapter 14 The Digestive System and Nutrition Digestive System Brings Nutrients Into the Body The digestive system includes Gastrointestinal (GI) tract (hollow tube) Lumen: space within this tube Includes
Human Biology. Digestive System
 Human Biology Digestive System Digestion - Defined Prepares food for use by all body cells The physical and/or chemical breakdown of food Did you know: the average person eats more than 500kg of food per
Human Biology Digestive System Digestion - Defined Prepares food for use by all body cells The physical and/or chemical breakdown of food Did you know: the average person eats more than 500kg of food per
Propulsion and mixing of food in the alimentary tract Chapter 63
 Propulsion and mixing of food in the alimentary tract Chapter 63 Types of GI movements: Propulsive movement-peristalsis Propulsion: controlled movement of ingested foods, liquids, GI secretions, and sloughed
Propulsion and mixing of food in the alimentary tract Chapter 63 Types of GI movements: Propulsive movement-peristalsis Propulsion: controlled movement of ingested foods, liquids, GI secretions, and sloughed
An Inhibitory Innervation at the Gastroduodenal Junction
 An Inhibitory Innervation at the Gastroduodenal Junction Sinn Anuras,, Allan R. Cooke, James Christensen J Clin Invest. 1974;54(3):529-535. https://doi.org/10.1172/jci107789. Research Article Transverse
An Inhibitory Innervation at the Gastroduodenal Junction Sinn Anuras,, Allan R. Cooke, James Christensen J Clin Invest. 1974;54(3):529-535. https://doi.org/10.1172/jci107789. Research Article Transverse
The Digestive System Laboratory
 The Digestive System Laboratory 1 The Digestive Tract The alimentary canal is a continuous tube stretching from the mouth to the anus. Liver Gallbladder Small intestine Anus Parotid, sublingual, and submaxillary
The Digestive System Laboratory 1 The Digestive Tract The alimentary canal is a continuous tube stretching from the mouth to the anus. Liver Gallbladder Small intestine Anus Parotid, sublingual, and submaxillary
General principles of gastrointestinal motility
 General principles of gastrointestinal motility OBJECTIVES Physiological anatomy General Principles Circulation of blood through the GIT organs Control of all GIT functions by local, nervous, and hormonal
General principles of gastrointestinal motility OBJECTIVES Physiological anatomy General Principles Circulation of blood through the GIT organs Control of all GIT functions by local, nervous, and hormonal
Figure Nutrition: omnivore, herbivore, carnivore
 Figure 41.1 Nutrition: omnivore, herbivore, carnivore Essential Nutrients: Amino acids Fatty acids Vitamins Minerals Figure 41.2 Complete vs incomplete Omnivore vs herbivore (vegetarian) Table 41.1 Table
Figure 41.1 Nutrition: omnivore, herbivore, carnivore Essential Nutrients: Amino acids Fatty acids Vitamins Minerals Figure 41.2 Complete vs incomplete Omnivore vs herbivore (vegetarian) Table 41.1 Table
THE DIGESTIVE SYSTEM
 THE DIGESTIVE SYSTEM TYPES OF DIGESTIVE SYSTEMS Ingested food may be stored or first subjected to physical fragmentation Chemical digestion occurs next Hydrolysis reactions liberate the subunit molecules
THE DIGESTIVE SYSTEM TYPES OF DIGESTIVE SYSTEMS Ingested food may be stored or first subjected to physical fragmentation Chemical digestion occurs next Hydrolysis reactions liberate the subunit molecules
Soft palate elevates, closing off the nasopharynx. Hard palate Tongue Bolus Epiglottis. Glottis Larynx moves up and forward.
 The Cephalic Phase Chemical and mechanical digestion begins in the mouth Saliva is an exocrine secretion Salivary secretion is under autonomic control Softens and lubricates food Chemical digestion: salivary
The Cephalic Phase Chemical and mechanical digestion begins in the mouth Saliva is an exocrine secretion Salivary secretion is under autonomic control Softens and lubricates food Chemical digestion: salivary
ACETYLSALICYLIC ACID AND IONIC FLUXES ACROSS THE GASTRIC MUCOSA OF MAN
 GASTROENTEROLOGY Copyright 1968 by The Williams & Wilkins Co. Vol. 54, No.4, Part 1 of 2 Parts Printed in U.S.A. ACETYLSALICYLIC ACID AND IONIC FLUXES ACROSS THE GASTRIC MUCOSA OF MAN BERGEIN F. OVERHOLT,
GASTROENTEROLOGY Copyright 1968 by The Williams & Wilkins Co. Vol. 54, No.4, Part 1 of 2 Parts Printed in U.S.A. ACETYLSALICYLIC ACID AND IONIC FLUXES ACROSS THE GASTRIC MUCOSA OF MAN BERGEIN F. OVERHOLT,
- Digestion occurs during periods of low activity - Produces more energy than it uses. - Mucosa
 Introduction Digestive System Chapter 29 Provides processes to break down molecules into a state easily used by cells - A disassembly line: Starts at the mouth and ends at the anus Digestive functions
Introduction Digestive System Chapter 29 Provides processes to break down molecules into a state easily used by cells - A disassembly line: Starts at the mouth and ends at the anus Digestive functions
s. J. RUNE, M.D., AND F. W. HENRIKSEN, M.D.
 GASTROENTEROLOGY Copyright 1969 by The Williams & Wilkins Co. Vol. 56, No.4 Printed in U.S.A. CARBON DOXDE TENSONS N TlE PROXMAL PART OF THE CANNE GASTRONTESTNAL TRACT s. J. RUNE, M.D., AND F. W. HENRKSEN,
GASTROENTEROLOGY Copyright 1969 by The Williams & Wilkins Co. Vol. 56, No.4 Printed in U.S.A. CARBON DOXDE TENSONS N TlE PROXMAL PART OF THE CANNE GASTRONTESTNAL TRACT s. J. RUNE, M.D., AND F. W. HENRKSEN,
Grossman, 1975). Chemical ingredients of food, particularly the peptides and amino
 J. Physiol. (1979), 295, pp. 229-239 229 With 4 text-ftgure8 Printed in Great Britain ROLE OF GASTRIC ANTRUM IN GASTRIC AND INTESTINAL PHASES OF GASTRIC SECRETION IN DOGS BY A. FOKINA, S. J. KONTUREK,
J. Physiol. (1979), 295, pp. 229-239 229 With 4 text-ftgure8 Printed in Great Britain ROLE OF GASTRIC ANTRUM IN GASTRIC AND INTESTINAL PHASES OF GASTRIC SECRETION IN DOGS BY A. FOKINA, S. J. KONTUREK,
Key words: acetylcholine, capsaicin, presynaptic cholinergic neurons, postsynaptic cholinergic neurong truncal vagotomy
 Key words: acetylcholine, capsaicin, presynaptic cholinergic neurons, postsynaptic cholinergic neurong truncal vagotomy Fig. 2 Effects of hexamethonium (Co) on CCK8 infusion (2.5-80ng/kglmin)-induced gallbladder
Key words: acetylcholine, capsaicin, presynaptic cholinergic neurons, postsynaptic cholinergic neurong truncal vagotomy Fig. 2 Effects of hexamethonium (Co) on CCK8 infusion (2.5-80ng/kglmin)-induced gallbladder
5. Which component of the duodenal contents entering the stomach causes the most severe changes to gastric mucosa:
 Gastro-intestinal disorders 1. Which are the most common causes of chronic gastritis? 1. Toxic substances 2. Chronic stress 3. Alimentary factors 4. Endogenous noxious stimuli 5. Genetic factors 2. Chronic
Gastro-intestinal disorders 1. Which are the most common causes of chronic gastritis? 1. Toxic substances 2. Chronic stress 3. Alimentary factors 4. Endogenous noxious stimuli 5. Genetic factors 2. Chronic
ANATOMY & PHYSIOLOGY ONLINE COURSE - SESSION 13 THE DIGESTIVE SYSTEM
 ANATOMY & PHYSIOLOGY ONLINE COURSE - SESSION 13 THE DIGESTIVE SYSTEM The digestive system also known as the alimentary canal or gastrointestinal tract consists of a series of hollow organs joined in a
ANATOMY & PHYSIOLOGY ONLINE COURSE - SESSION 13 THE DIGESTIVE SYSTEM The digestive system also known as the alimentary canal or gastrointestinal tract consists of a series of hollow organs joined in a
Progress report Radioimmunoassay of intestinal
 Gut, 1974, 15, 502-510 Progress report Radioimmunoassay of intestinal hormones Alimentary diseases are common yet often their aetiology is unknown. This ignorance reflects a basic lack of understanding
Gut, 1974, 15, 502-510 Progress report Radioimmunoassay of intestinal hormones Alimentary diseases are common yet often their aetiology is unknown. This ignorance reflects a basic lack of understanding
Gastrointestinal Hormones: Introduction and Index
 Gastrointestinal Hormones: Introduction and Index Digesting, absorbing and assimilating a meal requires precise coordination of a huge number of physiologic processes. Control over gastrointestinal function
Gastrointestinal Hormones: Introduction and Index Digesting, absorbing and assimilating a meal requires precise coordination of a huge number of physiologic processes. Control over gastrointestinal function
Digestive Care Advisor Training #1. Digestion 101 & H.O.P.E.
 Digestive Care Advisor Training #1 & H.O.P.E. The Digestive System in Brief The Process of Digestion The human digestive system is a complex series of organs and glands that process food and excrete waste.
Digestive Care Advisor Training #1 & H.O.P.E. The Digestive System in Brief The Process of Digestion The human digestive system is a complex series of organs and glands that process food and excrete waste.
Symposium on diagnosis of pancreatic disease
 Gut, 1975, 16, 648-663 Symposium on diagnosis of pancreatic disease Pancreatic function tests: T. SCRATCHERD The physiological background From the Department ofphysiology, The University of Sheffield,
Gut, 1975, 16, 648-663 Symposium on diagnosis of pancreatic disease Pancreatic function tests: T. SCRATCHERD The physiological background From the Department ofphysiology, The University of Sheffield,
BIO 139 ANATOMY AND PHYSIOLOGY II THE DIGESTIVE SYSTEM
 BIO 139 ANATOMY AND PHYSIOLOGY II THE DIGESTIVE SYSTEM MARY CATHERINE FLATH, Ph.D. DIGESTIVE SYSTEM ORGANS MOUTH PHARYNX ESOPHAGUS STOMACH SMALL INTESTINE LARGE INTESTINE SALIVARY GLANDS PANCREAS LIVER
BIO 139 ANATOMY AND PHYSIOLOGY II THE DIGESTIVE SYSTEM MARY CATHERINE FLATH, Ph.D. DIGESTIVE SYSTEM ORGANS MOUTH PHARYNX ESOPHAGUS STOMACH SMALL INTESTINE LARGE INTESTINE SALIVARY GLANDS PANCREAS LIVER
Overview of digestion or, gut reactions - to food
 Key concepts in Digestion. Indigestion module Overview of digestion or, gut reactions - to food Prof. Barry Campbell Gastroenterology Cellular & Molecular Physiology e-mail: bjcampbl@liv.ac.uk http://pcwww.liv.ac.uk/~bjcampbl
Key concepts in Digestion. Indigestion module Overview of digestion or, gut reactions - to food Prof. Barry Campbell Gastroenterology Cellular & Molecular Physiology e-mail: bjcampbl@liv.ac.uk http://pcwww.liv.ac.uk/~bjcampbl
The Digestive System. Prepares food for use by all body cells.
 The Digestive System Prepares food for use by all body cells. Digestion The chemical breakdown of complex biological molecules into their component parts. Lipids to fatty acids Proteins to individual amino
The Digestive System Prepares food for use by all body cells. Digestion The chemical breakdown of complex biological molecules into their component parts. Lipids to fatty acids Proteins to individual amino
Human Digestive System
 3.3.4 Human Digestive System Need to know The functions of the main parts of the alimentary canal and the associated glands Function of teeth. The human dental formula Role of mechanical digestion and
3.3.4 Human Digestive System Need to know The functions of the main parts of the alimentary canal and the associated glands Function of teeth. The human dental formula Role of mechanical digestion and
- Digestion occurs during periods of low activity - Produces more energy than it uses. 3 Copyright 2016 by Elsevier Inc. All rights reserved.
 Introduction Digestive System Chapter 29 Provides processes to break down molecules into a state easily used by cells - A disassembly line: Starts at the mouth and ends at the anus Digestive functions
Introduction Digestive System Chapter 29 Provides processes to break down molecules into a state easily used by cells - A disassembly line: Starts at the mouth and ends at the anus Digestive functions
3, 4), although its concentration in mixed gastric
 THE VALUE OF THE ACID TEST MEAL: A STUDY OF NORMAL PERSONS AND OF PERSONS WITH DUODENAL ULCER By C. STUART WELCH AND MANDRED W. COMFORT (From The Mayo Foundation and the Division of Medicine, The Mayo
THE VALUE OF THE ACID TEST MEAL: A STUDY OF NORMAL PERSONS AND OF PERSONS WITH DUODENAL ULCER By C. STUART WELCH AND MANDRED W. COMFORT (From The Mayo Foundation and the Division of Medicine, The Mayo
Chapter 21 NUTRITION AND DIGESTION
 Chapter 21 NUTRITION AND DIGESTION Stages of Food Processing 1. Ingestion: The act of eating. Usually involves placing food in mouth or oral cavity. 2. Digestion: Macromolecules in food (fats, proteins,
Chapter 21 NUTRITION AND DIGESTION Stages of Food Processing 1. Ingestion: The act of eating. Usually involves placing food in mouth or oral cavity. 2. Digestion: Macromolecules in food (fats, proteins,
EFFECT OF VAGOTOMY AND PYLOROPLASTY: THE ORAL GLUCOSE TOLERANCE TEST
 GASTROENTEROLOGY 64: 217-222, 1973 Copyright 1973 by The Williams & Wilkins Co. Vol. 64, No.2 Printed in U.S.A. EFFECT OF VAGOTOMY AND PYLOROPLASTY: THE ORAL GLUCOSE TOLERANCE TEST W, H, HALL, M,D" L.
GASTROENTEROLOGY 64: 217-222, 1973 Copyright 1973 by The Williams & Wilkins Co. Vol. 64, No.2 Printed in U.S.A. EFFECT OF VAGOTOMY AND PYLOROPLASTY: THE ORAL GLUCOSE TOLERANCE TEST W, H, HALL, M,D" L.
The Digestive System
 The Digestive System Identify the Structure and Function. Mesentery of the Large Intestine The mesentery functions to connect the visceral organs to the abdominal wall. Identify the Structure. Nasal Cavity
The Digestive System Identify the Structure and Function. Mesentery of the Large Intestine The mesentery functions to connect the visceral organs to the abdominal wall. Identify the Structure. Nasal Cavity
