Specific and rapid induction of the proapoptotic protein Hrk after growth factor withdrawal in hematopoietic progenitor cells
|
|
|
- Randolph Perkins
- 5 years ago
- Views:
Transcription
1 Plenary paper Specific and rapid induction of the proapoptotic protein Hrk after growth factor withdrawal in hematopoietic progenitor cells Cristina Sanz, Adalberto Benito, Naohiro Inohara, Daryoush Ekhterae, Gabriel Nunez, and Jose Luis Fernandez-Luna Hrk is a newly described proapoptotic member of the Bcl-2 family that is mainly expressed in hematopoietic tissues and cultured neurons. In this study we have examined the expression and activity of Hrk in hematopoietic progenitors. To address these issues, we used 3 growth factor-dependent murine hematopoietic cell lines, HCD-57, FDCP-Mix, and FL5.12. The expression of Hrk was undetectable in cells cultured with growth factors, but it was rapidly up-regulated on growth factor withdrawal. In contrast, the expression of Bcl-x L decreased and that of Introduction proapoptotic Bax, Bad, and Bak was unchanged or down-regulated after removal of growth factors. This pattern of expression correlated with the induction of apoptosis. Hrk was also up-regulated in human cell lines and in bone marrowderived CD34 cells cultured in the absence of growth factors. In addition, the levels of Hrk were up-regulated after treatment with the chemotherapeutic drug etoposide. Expression of prosurvival Bcl-x L or Bcl-2 proteins blocked the induction of Hrk. Hrk was induced in FDCP-Mix cells treated with ionomicin in the presence of IL-3, suggesting that cytosolic calcium may regulate the expression of this proapoptotic protein. Furthermore, ectopic expression of Hrk induced cell death of hematopoietic progenitors in the presence of IL-3. Thus, Hrk is specifically and rapidly induced in hematopoietic progenitors after growth factor deprivation or treatment with chemotherapeutic drugs, and this may be sufficient to induce apoptosis in these cells. (Blood. 2000;95: ) 2000 by The American Society of Hematology There is increasing evidence that most cells in multicellular organisms require constant stimulation by extracellular signals to survive. Committed hematopoietic progenitor cells require defined growth factors for survival, differentiation, and proliferation. 1 Withdrawal of these growth factors leads to apoptosis, and this cell death mechanism has been proposed to play a critical role in the control of cell numbers in both hematopoietic precursors and their fully differentiated cell populations. 2-4 Apoptosis is implemented by a death machinery that is evolutionarily conserved and activated in the dying cell. In mammals, the executory arm of apoptosis involves a family of death proteases, called caspases, that are activated in a proteolytic cascade to execute the cell death program. 5 The activation of upstream caspases represents a critical checkpoint in the decision to survive or to die. 6,7 Upstream caspases are controlled by several molecules, including proteins of the Bcl-2 family. Members of this family possess at least 1 of 4 conserved motifs known as Bcl-2 homology domains (BH1 to BH4), and can exert both prosurvival and proapoptotic activity. 8 In hematopoietic progenitor cells, several mechanisms have been proposed to explain how growth factors promote survival through Bcl-2 family members. Several hematopoietins, including interleukin-3 (IL-3) and erythropoietin, have been shown to maintain the expression of prosurvival Bcl-2 family members, including Bcl-2, Bcl-x L, A1, and Mcl-1, at the transcriptional level. 9,10 By contrast, RNA levels of proapoptotic Bcl-2 members, including Bax, Bad, and Bak, appear to be regulated independently of IL-3 in hematopoietic progenitor cells. Additionally, IL-3 and perhaps other growth factors can promote survival through phosphorylation and inactivation of proapoptotic BAD via activation of the Akt kinase. 11 Some proapoptotic Bcl-2 family members, including Bik/Nbk, Blk, Bad, and Bid, contain only the BH3 motif that acts as a dimerization domain for prosurvival proteins, including Bcl-2 and Bcl-x L The proapoptotic activity of these BH3-only proteins is mediated by the BH3 region, as deletion or mutation of this region abrogates their binding to Bcl-2 and Bcl-x L and their killing activity. 16,17 Thus, these BH3-only Bcl-2 family proteins have been suggested to represent the physiologic antagonists of prosurvival Bcl-2 family members. 8 Little is known about the regulation of the newly described BH3 subfamily of proapoptotic Bcl-2 family members. These BH3 proteins are evolutionarily conserved, in that they are structurally related to EGL-1, a nematode BH3-containing protein that interacts with and inhibits CED Human Hrk and its mouse homologue (DP5) are members of this BH3 subfamily of proapoptotic proteins. Human Hrk was isolated as a Bcl-x L and Bcl-2-interacting protein that was found to be preferentially expressed in spleen and bone marrow. 19 Mouse Hrk was identified during a screening for genes up-regulated after nerve growth factor withdrawal in primary sympathetic neurons. 20 We report here that the expression of Hrk, but not that of related proapoptotic Bcl-2 family members, including Bad, Bax, and Bak, is rapidly induced by growth factor deprivation at the messenger RNA (mrna) and protein levels in hematopoietic progenitors. This up-regulation of Hrk was inhibited by prosurvival Bcl-2 and Bcl-x L proteins. We From the Servicio de Inmunologia, Hospital Universitario Marques de Valdecilla, INSALUD, Santander, Spain, and Department of Pathology, University of Michigan Medical School, Ann Arbor, MI. Submitted July 13, 1999; accepted January 3, Supported by Comision Interministerial de Ciencia y Tecnologia Grant No. SAF-96/ 0274 to J.L.F-L., and grant P01 CA75136 from the National Institutes of Health to G.N. C.S. is a recipient of a predoctoral fellowship from the Fundacion Marques de Valdecilla ; A.B. is a recipient of a NATO postdoctoral fellowship. Reprints: Jose Luis Fernandez-Luna, Servicio de Inmunologia, Hospital Universitario Marques de Valdecilla, INSALUD, Santander, Spain; inmflj@humv.es. The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked advertisement in accordance with 18 U.S.C. section by The American Society of Hematology 2742 BLOOD, 1 MAY 2000 VOLUME 95, NUMBER 9
2 BLOOD, 1 MAY 2000 VOLUME 95, NUMBER 9 ACTIVITY OF HRK IN HEMATOPOIETIC PROGENITOR CELLS 2743 also report that enforced expression of Hrk in the presence of growth factor induces cell death in hematopoietic progenitor cells. Materials and methods Cell culture The erythropoietin-dependent HCD-57 cell line and its derivative HCD-57- Bcl-x L were maintained in Iscove s modified Dulbecco s medium (IMDM) (GIBCO-BRL, Grand Island, NY), supplemented with 0.1 U/mL of recombinant erythropoietin (Boehringer Mannheim, Indianapolis, IN) as previously described. 9 FL5.12, FL5.12-Bcl-2, 21 FDCP-Mix, and UT7 cell lines were grown in RPMI 1640 medium (Seromed Biochrom KG, Berlin, Germany), supplemented with 10% fetal calf serum (FCS) (Flow Laboratories, Irvine, CA), and 10% of Wehi 3B culture supernatant as an IL-3 source for the murine cells, or 5 ng/ml granulocyte-macrophage colonystimulating factor (GM-CSF) (Sigma, St Louis, MO) for UT7 cells. Mo7e cell line was cultured in IMDM supplemented with 20% FCS and 5 ng/ml IL-3 (Sigma). When indicated, cell lines were treated with 10 µg/ml etoposide or 1 µmol/l ionomycin (both from Sigma) for different time intervals and then analyzed for expression of Hrk. Bone marrow was obtained from healthy donors after appropriate informed consent. The CD34 positive bone marrow population was selected using an immunomagnetic system as previously described 22 and then cultured in IMDM containing 20% FCS and recombinant stem cell factor, IL-3, and IL-6 (Immunex, Seattle, WA) at a final concentration of 100 ng/ml. Transfection and cell death analysis FL5.12 and FDCP-Mix cells ( ) were transfected by electroporation (200 V, 950 µf) with a construct containing the human Hrk cdna in the sense or inverted orientation, cloned into the unique EcoRI site of the pires-egfp vector (Clontech Lab, Palo Alto, CA) or with a control pires-neo plasmid. After 24 hours of transfection, cells were analyzed for expression of the green fluoresce protein and the uptake of propidium iodide by flow cytometry using a FACScan analyzer (Becton Dickinson, San Jose, CA). For propidium iodide uptake, cells were incubated in a solution containing 0.1% sodium citrate and 0.1 mg/ml propidium iodide, for 10 minutes and then analyzed. In some experiments, FL5.12 cells were transfected with 1 µg of pcdna3-hrk 19 or control pcdna3 plasmid. After 48 hours, cells were seeded into 96-well plates at 2000 cells per well in the presence of 1 mg/ml G418 and the number of wells containing viable cells were scored on day 12 after transfection. Antibodies against Hrk and Western blot analysis To examine the expression of Hrk, we synthesized a 35-amino acid peptide corresponding to residues that encompass the BH3 region of human Hrk. 19 A rabbit was immunized and, after antigen boosting, the immune serum that recognized a protein of the expected molecular mass for human and mouse Hrk was used for Western blot analysis. The expression of Bcl-x L, Bax, Bad, Bak, and Hrk proteins was determined by Western blotting as previously described. 9 Blots were incubated with rabbit antibodies against Bcl-x (Transduction Lab, Lexington, KY), Bax (Santa Cruz Biotechnology, Santa Cruz, CA), Bak (Upstate Biotechnology, Lake Placid, NY), and Hrk, or mouse anti-bad antibodies (Transduction Lab), and then incubated with goat antirabbit or goat antimouse antibodies conjugated to alkaline phosphatase (Tropix, Bedford, MA). Bound antibody was detected by a chemiluminescence system (Tropix). Messenger RNA expression analysis Total RNA was prepared using TRIZOL reagent (GIBCO-BRL). To assess mrna expression, a semiquantitative reverse transcriptase-polymerase chain reaction (RT-PCR) method was used as previously described. 9 The generated cdna was amplified by using primers for murine Bcl-x, -actin, 9 and Hrk (5 TAGGCGACGAGCTGCA3 and 5 CTCCAAGGACA- CAGGGTT3 ). The amplification profile was as follows: 94 C for 30 seconds, 55 C for 30 seconds, and 72 C for 30 seconds. After 25 to 30 amplification cycles, the expected PCR products were size fractionated onto a 2% agarose gel and stained with ethidium bromide. Assays for apoptotic cells For DNA fragmentation analysis, cells (1 to ) were washed with phosphate-buffered saline and pelleted by centrifugation. Genomic DNA was isolated from cell pellets as described previously. 23 DNA samples were electrophoresed on a 2% agarose gel and stained with 0.1% ethidium bromide. The early apoptotic cells were detected with annexin V, labeled with fluorescein isothiocyanate (PharMingen, San Diego, CA) by flow cytometry. Results Hrk is up-regulated in hematopoietic progenitor cell lines cultured in the absence of specific growth factors To examine the regulation of Hrk, we used 3 hematopoietic progenitor cell lines that required defined growth factors for survival and proliferation. FDCP-Mix and FL5.12 are myeloid and lymphoid cell lines, respectively, that are IL-3 dependent, whereas HCD-57 is an erythroid cell line that requires erythropoietin for proliferation and survival. When these 3 hematopoietic cell lines were cultured in the absence of growth factor, the protein levels of Hrk were clearly up-regulated by 24 hours and greatly increased by 48 hours after growth factor withdrawal (Figure 1A). In contrast, expression of Bcl-x L gradually decreased within 48 hours of culture in the absence of growth factor. In addition, the expression of proapoptotic members of the Bcl-2 family, Bax, Bak, and Bad, either remained unchanged or diminished, but none were upregulated within 48 hours after growth factor withdrawal. To verify whether the increase in Hrk protein expression correlated with the level of mrna, we determined the levels of Hrk by semiquantitative RT-PCR analysis (Figure 1B). In agreement with the protein results, Hrk was up-regulated and Bcl-x L mrna levels were down-regulated within 48 hours of growth factor deprivation (Figure 1B). This expression pattern was accompanied by a loss of cell viability caused by the activation of an apoptotic process. The genomic DNA isolated from FDCP-Mix, HCD-57, and FL5.12 cells cultured in the absence of growth factor was degraded into oligonucleosomal fragments that are characteristic of apoptosis (Figure 1C). Similar results have been obtained also with human hematopoietic progenitors. Induction of the Hrk protein but not of Bax was detected in human megakaryoblastic UT-7 and Mo7e cell lines cultured in the absence of GM-CSF and IL-3, respectively, for 24 and 48 hours, and in bone marrow-derived CD34 cell population cultured without IL-3, IL-6, and stem cell factor for 36 hours (Figure 2). Previous data showed that the treatment of rat cortical neurons with inhibitors of calcium channels blocked the expression of Hrk mrna and prevented apoptosis induced by amyloid- protein. 24 To analyze whether calcium flux could also affect the expression of Hrk in hematopoietic progenitors, we incubated FDCP-Mix cells with 1 µmol/l ionomycin in the presence of IL-3. As shown in Figure 3A, the protein levels of Hrk but not of Bax increased by 24 hours after addition of ionomycin, and the expression was similar to that of cells cultured in the absence of IL-3. Furthermore, this up-regulation of Hrk correlated with the activation of an apoptotic
3 2744 SANZ et al BLOOD, 1 MAY 2000 VOLUME 95, NUMBER 9 A B Figure 2. Expression of Hrk in human cell lines and CD34 progenitor cells. Two megakaryoblastic cell lines were cultured in the absence of IL-3 (Mo7e) or GM-CSF (UT7). Bone marrow-derived CD34 cells were cultured with ( GFs) and without ( GFs) growth factors (IL-3, IL-6, and stem cell factor) for 36 hours. At the indicated time intervals, cells were analyzed for the expression of Hrk and Bax by Western blot. C mrna increased at a similar rate to that found in cycloheximidefree cultures within 2 hours after IL-3 withdrawal (Figure 4). In addition, the protein levels of Hrk reached after 24 hours of growth factor deprivation were clearly reduced by 6 hours after re-addition of IL-3 and returned to baseline by 12 to 24 hours (Figure 4B). By contrast, the steady-state protein levels of Bax were not modified during the same interval (Figure 4B). This result was consistent with the half-life of the Hrk protein, which was estimated to be around 6 hours as detected by pulse-chase labeling analysis (data not shown). Figure 1. Analysis of Bcl-2 family members and apoptosis in murine hematopoietic progenitor cell lines after growth factor withdrawal. (A) Western blot analysis of Hrk, Bcl-x L, Bax, Bad, and Bak in cells cultured in the presence (0 hours) or absence of IL-3 (FDCP-Mix, FL5.12) or erythropoietin (HCD-57) for 24 and 48 hours. (B) Semiquantitative RT-PCR analysis of Hrk and Bcl-x L mrna at 0, 24, and 48 hours after growth factor deprivation. PCR products were electrophoresed onto a 2% agarose gel. -actin mrna was used as an amplification control. (C) DNA fragmentation analysis in cells cultured with or without growth factor. Cells were incubated for the indicated time points and genomic DNA fragmentation was monitored by electrophoresis onto a 2% agarose gel and staining with ethidium bromide. process as assessed by genomic DNA fragmentation analysis (Figure 3B). Induction of Hrk is rapid and unaffected by pretreatment with cycloheximide We focused next on the expression of Hrk during the first 2 hours after IL-3 withdrawal, to study whether the induction of Hrk was an early response to an apoptotic stimulus. The levels of Hrk mrna in FDCP-Mix cells began to increase within 15 minutes and continued to accumulate by 2 hours after IL-3 deprivation (Figure 4A). The kinetics of Hrk induction resembled that of immediate early genes such as c-myc 25 or oncostatin M. 26 Indeed, as expected for an immediate early gene, when we culture growth factor-starved cells with cycloheximide at a dose (20 µg/ml) that inhibited more than 90% of protein synthesis (data not shown), the expression of Hrk Figure 3. Analysis of Hrk and apoptosis in FDCP-Mix cells treated with ionomycin. Cells were cultured for 24 hours with ionomycin in the presence of IL-3 and then analyzed. As controls, cells cultured in the presence or absence of IL-3 were also analyzed. (A) Expression of Hrk and Bax by Western blot. (B) DNA fragmentation analysis by electrophoresis onto a 2% agarose gel and staining with ethidium bromide.
4 BLOOD, 1 MAY 2000 VOLUME 95, NUMBER 9 ACTIVITY OF HRK IN HEMATOPOIETIC PROGENITOR CELLS 2745 A Figure 4. Expression of Hrk in FDCP-Mix cells treated with a protein synthesis inhibitor. (A) IL-3-deprived cells were cultured with or without cycloheximide (CHX), and at the indicated time intervals, the mrna levels of Hrk were analyzed by semiquantitative RT-PCR. As controls, cells maintained with IL-3 in the absence (first lane) or in the presence (second lane) of CHX were also analyzed and showed no induction of Hrk. PCR products were electrophoresed onto a 2% agarose gel. -actin mrna was used as an amplification control. (B) Cells were deprived of IL-3 for 24 hours and then stimulated for different time intervals with IL-3. The expression of Hrk and Bax proteins was determined by Western blotting with use of specific antibodies. Expression of Hrk is induced in hematopoietic progenitor cells after treatment with etoposide To determine whether the induction of Hrk was restricted to signals associated with growth factor withdrawal or, alternatively, it might be a response to an apoptotic signal, we analyzed the expression of Hrk in FDCP-Mix cells treated with the topoisomerase II inhibitor etoposide, an inducer of apoptosis. 27 As shown in Figure 5, when cells were cultured with etoposide in the presence of IL-3, a time-dependent increase in the number of apoptotic cells was observed, as assessed by flow cytometry analysis with annexin V (Figure 5A) and DNA fragmentation analysis (data not shown). By 12 hours of treatment, 53.3% of cells were annexin V positive, and by 24 hours after etoposide, most of the cells (76.2%) were apoptotic. Within 24 hours of treatment with etoposide, a clear correlation was observed between the increase in the number of apoptotic cells and the up-regulated expression of Hrk as assessed by Western blot analysis (Figure 5B). By contrast, etoposide treatment did not affect the expression of Bax. A similar pattern of expression was observed in FL5.12 and HCD-57 cells treated with etoposide (data not shown), indicating that in addition to growth factor withdrawal, the expression of Hrk can be up-regulated by chemotherapeutic drugs. Bcl-2 and Bcl-x L prevent the induction of Hrk Overexpression of Bcl-2 and Bcl-x L has been shown to inhibit or delay the apoptotic cell death in a number of cellular systems, 23,28-30 so that we studied whether ectopic expression of Bcl-x L or Bcl-2 could impede the up-regulation of Hrk in hematopoietic progenitor cell lines. HCD-57 cells transfected with Bcl-x L, 9 and FL5.12 expressing constitutive levels of Bcl-2 21 were cultured in the absence of erythropoietin and IL-3, respectively, and the levels of Hrk were analyzed by Western blot. Figure 6 shows a representative experiment. Both parental FL5.12, and HCD-57 displayed an increase in the expression of Hrk after growth factor withdrawal, in agreement with results shown in Figure 1. Furthermore, by 24 hours of culture without IL-3 and 48 hours in the absence of erythropoietin, most of the cells were apoptotic, as assessed by flow B Figure 5. Analysis of Hrk and apoptosis in etoposide-treated FDCP-Mix cells. (A) Cells were treated for different times with etoposide in the presence of IL-3 and then analyzed by flow cytometry with fluorescein isothiocyanate-labeled annexin V. Numbers above the selected regions indicate the percentage of apoptotic cells. (B) Western blot analysis of Hrk and Bax expression in FDCP-Mix cells treated with etoposide for 12 and 24 hours in the presence of IL-3. cytometry with annexin V and DNA fragmentation analysis (data not shown). Significantly, FL5.12-Bcl-2 cells and HCD-57-Bcl-x L did not show any change in the expression of Hrk at the protein (Figure 6) and mrna levels (data not shown) after 24 or 48 hours Figure 6. Analysis of the Hrk expression in cells transfected with Bcl-2 or Bcl-x L. FL5.12 cells transfected with human Bcl-2 cdna and HCD-57 cells transfected with human Bcl-x L cdna were cultured in the absence of IL-3 and Epo, respectively, for the indicated times and then analyzed for the expression of Hrk and Bax proteins by Western blot. As control, FL5.12 and HCD-57 cells subjected to the same culture conditions were also analyzed (lane C).
5 2746 SANZ et al BLOOD, 1 MAY 2000 VOLUME 95, NUMBER 9 of culture in the absence of IL-3 or erythropoietin, respectively. Moreover, these cells did not show any evidence of apoptosis within the same period after growth factor withdrawal (data not shown). These data indicate that Bcl-2 and Bcl-x L can repress the induction of Hrk and suggest that up-regulation of Hrk is triggered by an intracellular apoptotic signal that is inhibited by these prosurvival Bcl-2 family members. Enforced expression of Hrk induces cell death in the presence of growth factor We transfected a plasmid producing human Hrk into IL-3- dependent FL5.12 cells but analysis of multiple clones selected after culture in G418 showed no detectable Hrk expression (data not shown). To test whether Hrk inhibits the expansion of these cells into colonies, we compared colony formation in FL5.12 cells transfected with a plasmid expressing Hrk or with a control plasmid. Transfection of the control plasmid into FL5.12 cells generated colonies in the presence of G418, whereas only were obtained by transfection of the Hrk plasmid (Figure 7), indicating that expression of Hrk impedes the formation of colonies in the presence of IL-3. To determine whether Hrk blocked the expansion of these cells by inducing cell death, we constructed a plasmid to coexpress Hrk and enhanced green fluorescence protein (EGFP). This construct contains an internal ribosomal entry site, allowing translation of Hrk and EGFP from a single bicistronic mrna (Figure 8A), which permits identifying Hrk-expressing cells on the basis of EGFP expression after transient transfection. When we examined FL5.12 cells 24 hours after transfection with Hrk in the sense orientation, 98% of the EGFP-positive cells were dead as determined by their incorporation of propidium iodide (Figure 8B). In contrast, the great majority (more than 85%) of the cells transfected with Hrk in the inverted orientation that coexpressed EGFP remained alive as they excluded propidiun iodide (Figure 8B). Similar results were obtained with FDCPMix cells (data not shown). These results indicate that expression of Hrk causes the death of hematopoietic progenitor cells in the presence of growth factor. Figure 7. Expansion of FL5.12 cells after transfection with Hrk. Cells were transfected with pcdna3 or pcdna3-hrk expression plasmids and then cultured into 96-well plates at 2000 cells per well in the presence of G418 for 12 days. Data are presented as the number of colonies (wells containing viable cells) per transfected cells (mean of triplicate cultures SD). Figure 8. Cell death analysis of FL5.12 cells transiently transfected with pires-egfp-hrk. (A) Schematic structure of the bicistronic vector containing the internal ribosomal entry site (IRES) and the Hrk and EGFP cdnas. (B) Flow cytometry analysis of cells after 24 hours of transfection with pires-egfp containing Hrk in the sense or inverted orientation (io). Quadrants in the dot plots were set according to the green fluorescence of cells transfected with a negative control vector (pires-neo). Histograms represent the percentage of cells stained with propidium iodide in the green fluorescence positive population (R1) and the green fluorescence negative population (R2). All histograms and dot plots are from a representative experiment (n 3). Discussion A number of cytokines and hematopoietic growth factors have been shown to promote viability of subpopulations of primitive progenitor cells, suggesting that the regulation of apoptosis plays a key role in hematopoiesis. 1 Apoptosis is tightly regulated by members of the caspase and Bcl-2 families. Genetic and biochemical analyses have demonstrated that the activation of caspases is influenced by the Bcl-2 family members, which either suppress or induce apoptosis. 8 In this scenario, a reasonable expectation would be that the activity and/or expression of Bcl-2 family members is regulated by signaling pathways that suppress or induce apoptosis. Here we showed that Hrk, a newly described proapoptotic member of the Bcl-2 family, is specifically and rapidly induced in hematopoietic progenitor cell lines and bone marrow-derived CD34 cells after growth factor deprivation, which is accompanied by activation of an apoptotic process. Consistent with this is the observation that the mrna of mouse Hrk (DP5) was induced in sympathetic neurons cultured in the absence of nerve growth factor (NGF). 20 However, the expression of Hrk in hematopoietic progenitor cell lines can be induced not only by growth factor withdrawal but also by treatment with etoposide, a chemotherapeutic drug that induces apoptosis. On the basis of these data, we hypothesize that the binding of growth factors (ie, IL-3, erythropoietin, GM-CSF) to their cognate receptors in hematopoietic progenitors activates a repressor pathway that silences the expression of proapoptotic Hrk. Alternatively, this repressor pathway may not be triggered by the growth factor but may be active during the normal development of hematopoietic progenitor cells. On growth factor withdrawal or treatment with certain chemotherapeutic drugs, the repressor mechanism may be released (ie, by posttranslational modification or binding to a specific factor), allowing the expression of Hrk. A similar mechanism may operate in neurons that up-regulate mouse Hrk and undergo apoptosis after NGF withdrawal or treatment with
6 BLOOD, 1 MAY 2000 VOLUME 95, NUMBER 9 ACTIVITY OF HRK IN HEMATOPOIETIC PROGENITOR CELLS 2747 amyloid- protein. 20,24 The induction of mouse Hrk in cultured neurons treated with amyloid- protein is blocked by inhibitors of voltage-dependent calcium channels and calcium release from the endoplasmic reticulum, suggesting that calcium fluxes could be involved in the regulation of Hrk expression. 24 An increase in cytosolic calcium has been implicated in signal transduction pathways that mediate apoptosis. 31,32 We have shown here that the expression of Hrk is induced in FDCP-Mix cells after incubation with ionomycin, a calcium ionophore that releases calcium from a variety of intracellular stores, including the endoplasmic reticulum and mitochondria. 33 Thus, Hrk might be regulated in these cells by an intracellular calcium-sensitive pathway that is activated on growth factor withdrawal or treatment with chemotherapeutic drugs. Furthermore, it has been described that Bcl-2 either directly or indirectly regulates the flux of calcium across the endoplasmic reticulum membrane, thereby abrogating calcium signaling of apoptosis. 34 Consistent with the model of calcium-regulated expression of Hrk, we have shown that overexpression of Bcl-2 or Bcl-x L inhibits the induction of Hrk and prevents apoptosis after growth factor withdrawal. Deletion of Bcl-x in mice leads to apoptosis of hematopoietic progenitors and embryonic lethality. 35 Thus, it appears that Bcl-x L is an important mediator of hematopoietic cell survival in vivo. Hematopoietic progenitor cells down-regulate the expression of Bcl-x L on growth factor deprivation, in addition to up-regulating Hrk. Furthermore, we demonstrate that enforced expression of Hrk into hematopoietic progenitor cell lines is sufficient to induce cell death in the presence of IL-3 within 24 hours. Consistent with this is the observation that transfection of Hrk into embryonic kidney 293T cells resulted in a loss of cell viability at 36 hours posttransfection. 19 This suggests that induction of Hrk, which is normally silenced or expressed at low levels, might be a mechanism that participates in the activation of a cell death program linked to growth factor withdrawal or treatment with chemotherapeutic drugs, in hematopoietic progenitors. Because enforced coexpression of Hrk and the antiapoptotic protein Bcl-2 or Bcl-x L inhibits the death promoting activity of Hrk, 19 it appears that the balance between the steady-state levels of Bcl-x L or Bcl-2 and those of Hrk likely contribute to either suppress or induce apoptosis. Future studies will need to address the molecular mechanisms that regulate the expression of Hrk and, more importantly, the role for Hrk in the regulation of normal hematopoiesis. To this end, mice deficient in Hrk will be very useful to study whether this proapoptotic protein is needed for the homeostasis of the hematopoietic compartment. References 1. Sachs L. The control of hematopoiesis and leukemia: from basic biology to the clinic. Proc Natl Acad Sci U S A. 1996;93: Williams GT, Smith CA, Spooncer E, Dexter TM, Taylor DR. Hemopoietic colony stimulating factors promote cell survival by suppressing apoptosis. Nature. 1990;324: Yu H, Bauer B, Lipke GK, Phillips RL, Van Zant G. Apoptosis and hematopoiesis in murine fetal liver. Blood. 1993;81: Koury MJ, Bondurant MC. Control of red cell production: the roles of programmed cell death (apoptosis) and erythropoietin. Transfusion. 1990;30: Thornberry NA, Lazebnik Y. Caspases: enemies within. Science. 1998;281: Harvey NL, Butt AJ, Kumar S. Functional activation of Nedd2/ICH-1 (caspase-2) is an early process in apoptosis. J Biol Chem. 1997;272:13, Bossy-Wetzel E, Newmeyer DD, Green DR. Mitochondrial cytochrome c release in apoptosis occurs upstream of DEVD-specific caspase activation and independently of mitochondrial transmembrane depolarization. EMBO J. 1998; 17: Adams JM, Cory S. The Bcl-2 protein family: arbiters of cell survival. Science. 1998;281: Silva M, Grillot D, Benito A, Richard C, Nunez G, Fernandez-Luna JL. Erythropoietin can promote erythroid progenitor survival by repressing apoptosis through Bcl-x L and Bcl-2. Blood. 1996;88: Packham G, White EL, Eischen CM, et al. Selective regulation of Bcl-xL by a Jak kinase-dependent pathway is bypassed in murine hematopoietic malignancies. Genes Dev. 1998;12: del Peso L, Gonzalez-Garcia M, Page C, Herrera R, Nunez G. Interleukin-3-induced phosphorylation of BAD through the protein kinase Akt. Science. 1997;278: Boyd JM, Gallo G, Elangoban B, et al. Bik, a novel death-inducing protein shares a distinct sequence motif with Bcl-2 family proteins and interacts with viral and cellular survival-promoting proteins. Oncogene. 1995;11: Wang K, Yin X-M, Chao D, Milliman C, Korsmeyer SJ. BID: a novel BH3 domain-only death agonist. Genes Dev. 1996;10: Yang E, Zha J, Jockel J, Boise LH, Thompson CB, Korsmeyer SJ. Bad, a heterodimeric partner for Bcl-x L and Bcl-2, displaces Bax and promotes cell death. Cell. 1995;80: Hegde R, Srinivasula SM, Ahmad M, Fernandes- Alnemri T, Alnemri ES. Blk, a BH3-containing mouse protein that interacts with Bcl-2 and Bcl-x L, is a potent death agonist. J Biol Chem. 1998;273: Korsmeyer SJ. BCL-2 gene family and the regulation of programmed cell death. Cancer Res. 1999;59:1693s. 17. McDonnell JM, Fushman D, Milliman CL, Korsmeyer SJ, Cowburn D. Solution structure of the proapoptotic molecule BID: a structural basis for apoptotic agonists and antagonists. Cell. 1999; 96: Conradt B, Horvitz HR. The C. elegans protein EGL-1 is required for programmed cell death and interacts with the Bcl-2-like protein CED-9. Cell. 1998;93: Inohara N, Ding L, Chen S, Nunez G. Harakiri, a novel regulator of cell death, encodes a protein that activates apoptosis and interacts selectively with survival-promoting proteins Bcl-2 and Bcl-x L. EMBO J. 1997;16: Imaizumi K, Tsuda M, Imai Y, Wanaka A, Takagi T, Tohyama M. Molecular cloning of a novel polypeptide, DP5, induced during programmed neuronal death. J Biol Chem. 1997;272:18, Nunez G, London L, Hockenbery D, Alexander M, McKearn JP, Korsmeyer SJ. Deregulated Bcl-2 gene expression selectively prolongs survival of growth factor-deprived hemopoietic cell lines. J Immunol. 1990;144: Sanz C, Benito A, Silva M, et al. The expression of Bcl-x is downregulated during differentiation of human hematopoietic progenitor cells along the granulocyte but not the monocyte/macrophage lineage. Blood. 1997;89: Benito A, Grillot D, Nunez G, Fernandez-Luna JL. Regulation and function of Bcl-2 during differentiation-induced cell death in HL-60 promyelocytic cells. Am J Pathol. 1995;146: Imaizumi K, Morihara T, Mori Y, et al. The cell death-promoting gene DP5, which interacts with the Bcl-2 family, is induced during neuronal apoptosis following exposure to amyloid b protein. J Biol Chem. 1999;274: Spangler R, Bailey SC, Sytkowski AJ. Erythropoietin increases c-myc mrna by a protein kinase C-dependent pathway. J Biol Chem. 1991;266: Yoshimura A, Ichihara M, Kinjyo I, et al. Mouse oncostatin M: an immediate early gene induced by multiple cytokines through the JAK-STAT5 pathway. EMBO J. 1996;15: Ascaso R, Marvel J, Collins MK, Lopez-Rivas A. Interleukin-3 and Bcl-2 cooperatively inhibit etoposide-induced apoptosis in a murine pre-b cell line. Eur J Immunol. 1994;24: Benito A, Silva M, Grillot D, Nunez G, Fernandez- Luna JL. Apoptosis induced by erythroid differentiation of human erythroleukemia cell lines is inhibited by Bcl-x L. Blood. 1996;87: Boise LH, Gonzalez-Garcia M, Postema CE, et al. Bcl-x, a Bcl-2-related gene that functions as a dominant regulator of apoptotic cell death. Cell. 1993;74: Schwarze MM, Hawley RG. Prevention of myeloma cell apoptosis by ectopic bcl-2 expression or interleukin 6-mediated upregulation of bcl-x L. Cancer Res. 1995;55: Caron-Leslie LA, Cidlowski JA. Similar actions of glucocorticoids and calcium on the regulation of apoptosis in S49 cells. Mol Endocrinol. 1991;5: McConkey DJ, Nicotera P, Hartzell P, Bellomo G, Wyllie AH, Orrenius S. Glucocorticoids activate a suicide process in thymocytes through an elevation of cytosolic Ca 2 concentration. Arch Biochem Biophys. 1989;269: Albert PR, Tashjian AH. Ionomycin acts as an ionophore to release TRH-regulated Ca 2 stores from GH4C1 cells. Am J Physiol. 1986;251:C Lam M, Dubyak G, Chen L, Nunez G, Miesfeld RL, Distelhorst CW. Evidence that Bcl-2 represses apoptosis by regulating endoplasmic reticulum-associated calcium fluxes. Proc Natl Acad Sci U S A. 1994;91: Motoyama N, Wang F, Roth KA, et al. Massive cell death of immature hematopoietic cells and neurons in Bcl-x-deficient mice. Science. 1995; 267:1506.
Programmed Cell Death (apoptosis)
 Programmed Cell Death (apoptosis) Stereotypic death process includes: membrane blebbing nuclear fragmentation chromatin condensation and DNA framentation loss of mitochondrial integrity and release of
Programmed Cell Death (apoptosis) Stereotypic death process includes: membrane blebbing nuclear fragmentation chromatin condensation and DNA framentation loss of mitochondrial integrity and release of
ABSTRACT Background Deregulating the expression of Bcl-x L
 EXPRESSION OF IN ERYTHROID PRECURSORS FROM PATIENTS WITH POLYCYTHEMIA VERA MAITE SILVA, PH.D., CARLOS RICHARD, M.D., ADALBERTO BENITO, PH.D., CRISTINA SANZ, PH.D., IGNACIO OLALLA, M.D., AND JOSÉ LUIS FERNÁNDEZ-LUNA,
EXPRESSION OF IN ERYTHROID PRECURSORS FROM PATIENTS WITH POLYCYTHEMIA VERA MAITE SILVA, PH.D., CARLOS RICHARD, M.D., ADALBERTO BENITO, PH.D., CRISTINA SANZ, PH.D., IGNACIO OLALLA, M.D., AND JOSÉ LUIS FERNÁNDEZ-LUNA,
The discovery of Bcl-2
 The discovery of Bcl-2 Bcl-2: B-cell lymphoma 2 The pro-survival subfamily of Bcl-2 protein family Cloning of Bcl-2 as the oncogene which is deregulated at t(14;18) lymphomas Pioneer works by Tsujimoto
The discovery of Bcl-2 Bcl-2: B-cell lymphoma 2 The pro-survival subfamily of Bcl-2 protein family Cloning of Bcl-2 as the oncogene which is deregulated at t(14;18) lymphomas Pioneer works by Tsujimoto
Figure S1. Generation of inducible PTEN deficient mice and the BMMCs (A) B6.129 Pten loxp/loxp mice were mated with B6.
 Figure S1. Generation of inducible PTEN deficient mice and the BMMCs (A) B6.129 Pten loxp/loxp mice were mated with B6.129-Gt(ROSA)26Sor tm1(cre/ert2)tyj /J mice. To induce deletion of the Pten locus,
Figure S1. Generation of inducible PTEN deficient mice and the BMMCs (A) B6.129 Pten loxp/loxp mice were mated with B6.129-Gt(ROSA)26Sor tm1(cre/ert2)tyj /J mice. To induce deletion of the Pten locus,
Introduction to pathology lecture 5/ Cell injury apoptosis. Dr H Awad 2017/18
 Introduction to pathology lecture 5/ Cell injury apoptosis Dr H Awad 2017/18 Apoptosis = programmed cell death = cell suicide= individual cell death Apoptosis cell death induced by a tightly regulated
Introduction to pathology lecture 5/ Cell injury apoptosis Dr H Awad 2017/18 Apoptosis = programmed cell death = cell suicide= individual cell death Apoptosis cell death induced by a tightly regulated
Soft Agar Assay. For each cell pool, 100,000 cells were resuspended in 0.35% (w/v)
 SUPPLEMENTARY MATERIAL AND METHODS Soft Agar Assay. For each cell pool, 100,000 cells were resuspended in 0.35% (w/v) top agar (LONZA, SeaKem LE Agarose cat.5004) and plated onto 0.5% (w/v) basal agar.
SUPPLEMENTARY MATERIAL AND METHODS Soft Agar Assay. For each cell pool, 100,000 cells were resuspended in 0.35% (w/v) top agar (LONZA, SeaKem LE Agarose cat.5004) and plated onto 0.5% (w/v) basal agar.
RAS Genes. The ras superfamily of genes encodes small GTP binding proteins that are responsible for the regulation of many cellular processes.
 ۱ RAS Genes The ras superfamily of genes encodes small GTP binding proteins that are responsible for the regulation of many cellular processes. Oncogenic ras genes in human cells include H ras, N ras,
۱ RAS Genes The ras superfamily of genes encodes small GTP binding proteins that are responsible for the regulation of many cellular processes. Oncogenic ras genes in human cells include H ras, N ras,
Annals of Oncology Advance Access published January 10, 2005
 Annals of Oncology Advance Access published January 10, 2005 Original article Annals of Oncology doi:10.1093/annonc/mdi077 Expression of survivin and bax/bcl-2 in peroxisome proliferator activated receptor-g
Annals of Oncology Advance Access published January 10, 2005 Original article Annals of Oncology doi:10.1093/annonc/mdi077 Expression of survivin and bax/bcl-2 in peroxisome proliferator activated receptor-g
Supplementary data Supplementary Figure 1 Supplementary Figure 2
 Supplementary data Supplementary Figure 1 SPHK1 sirna increases RANKL-induced osteoclastogenesis in RAW264.7 cell culture. (A) RAW264.7 cells were transfected with oligocassettes containing SPHK1 sirna
Supplementary data Supplementary Figure 1 SPHK1 sirna increases RANKL-induced osteoclastogenesis in RAW264.7 cell culture. (A) RAW264.7 cells were transfected with oligocassettes containing SPHK1 sirna
SUPPLEMENTARY INFORMATION
 Figure S1 Induction of non-apoptotic death of SV40-transformed and primary DKO MEFs, and DKO thymocytes. (A-F) STS-induced non-apoptotic death of DKO MEF. (A, B) Reduced viability of DKO MEFs after exposure
Figure S1 Induction of non-apoptotic death of SV40-transformed and primary DKO MEFs, and DKO thymocytes. (A-F) STS-induced non-apoptotic death of DKO MEF. (A, B) Reduced viability of DKO MEFs after exposure
BL-8040: BEST-IN-CLASS CXCR4 ANTAGONIST FOR TREATMENT OF ONCOLOGICAL MALIGNANCIES. Overview and Mechanism of Action Dr.
 BL-8040: BEST-IN-CLASS CXCR4 ANTAGONIST FOR TREATMENT OF ONCOLOGICAL MALIGNANCIES Overview and Mechanism of Action Dr. Leah Klapper, CSO 88 BL-8040: Novel CXCR4 Antagonist For Hematological Cancers Indications:
BL-8040: BEST-IN-CLASS CXCR4 ANTAGONIST FOR TREATMENT OF ONCOLOGICAL MALIGNANCIES Overview and Mechanism of Action Dr. Leah Klapper, CSO 88 BL-8040: Novel CXCR4 Antagonist For Hematological Cancers Indications:
Apoptosis Chapter 9. Neelu Yadav PhD
 Apoptosis Chapter 9 Neelu Yadav PhD Neelu.Yadav@Roswellpark.org 1 Apoptosis: Lecture outline Apoptosis a programmed cell death pathway in normal homeostasis Core Apoptosis cascade is conserved Compare
Apoptosis Chapter 9 Neelu Yadav PhD Neelu.Yadav@Roswellpark.org 1 Apoptosis: Lecture outline Apoptosis a programmed cell death pathway in normal homeostasis Core Apoptosis cascade is conserved Compare
Signaling Apoptosis. Scott André Oakes, M.D. Dept. of Pathology Univ. of Calif-San Francisco. Cyt c Release BAX/BAK. Apoptosome Formation
 Signaling Apoptosis Cyt c Release BAX/BAK Apoptosome Formation Caspase Activation Scott André Oakes, M.D. Dept. of Pathology Univ. of Calif-San Francisco Why do we need cell death? Sculpt Organs Control
Signaling Apoptosis Cyt c Release BAX/BAK Apoptosome Formation Caspase Activation Scott André Oakes, M.D. Dept. of Pathology Univ. of Calif-San Francisco Why do we need cell death? Sculpt Organs Control
Problem Set 8 Key 1 of 8
 7.06 2003 Problem Set 8 Key 1 of 8 7.06 2003 Problem Set 8 Key 1. As a bright MD/PhD, you are interested in questions about the control of cell number in the body. Recently, you've seen three patients
7.06 2003 Problem Set 8 Key 1 of 8 7.06 2003 Problem Set 8 Key 1. As a bright MD/PhD, you are interested in questions about the control of cell number in the body. Recently, you've seen three patients
Molecular biology :- Cancer genetics lecture 11
 Molecular biology :- Cancer genetics lecture 11 -We have talked about 2 group of genes that is involved in cellular transformation : proto-oncogenes and tumour suppressor genes, and it isn t enough to
Molecular biology :- Cancer genetics lecture 11 -We have talked about 2 group of genes that is involved in cellular transformation : proto-oncogenes and tumour suppressor genes, and it isn t enough to
p47 negatively regulates IKK activation by inducing the lysosomal degradation of polyubiquitinated NEMO
 Supplementary Information p47 negatively regulates IKK activation by inducing the lysosomal degradation of polyubiquitinated NEMO Yuri Shibata, Masaaki Oyama, Hiroko Kozuka-Hata, Xiao Han, Yuetsu Tanaka,
Supplementary Information p47 negatively regulates IKK activation by inducing the lysosomal degradation of polyubiquitinated NEMO Yuri Shibata, Masaaki Oyama, Hiroko Kozuka-Hata, Xiao Han, Yuetsu Tanaka,
p53 and Apoptosis: Master Guardian and Executioner Part 2
 p53 and Apoptosis: Master Guardian and Executioner Part 2 p14arf in human cells is a antagonist of Mdm2. The expression of ARF causes a rapid increase in p53 levels, so what would you suggest?.. The enemy
p53 and Apoptosis: Master Guardian and Executioner Part 2 p14arf in human cells is a antagonist of Mdm2. The expression of ARF causes a rapid increase in p53 levels, so what would you suggest?.. The enemy
RNA extraction, RT-PCR and real-time PCR. Total RNA were extracted using
 Supplementary Information Materials and Methods RNA extraction, RT-PCR and real-time PCR. Total RNA were extracted using Trizol reagent (Invitrogen,Carlsbad, CA) according to the manufacturer's instructions.
Supplementary Information Materials and Methods RNA extraction, RT-PCR and real-time PCR. Total RNA were extracted using Trizol reagent (Invitrogen,Carlsbad, CA) according to the manufacturer's instructions.
Sestrin2 and BNIP3 (Bcl-2/adenovirus E1B 19kDa-interacting. protein3) regulate autophagy and mitophagy in renal tubular cells in. acute kidney injury
 Sestrin2 and BNIP3 (Bcl-2/adenovirus E1B 19kDa-interacting protein3) regulate autophagy and mitophagy in renal tubular cells in acute kidney injury by Masayuki Ishihara 1, Madoka Urushido 2, Kazu Hamada
Sestrin2 and BNIP3 (Bcl-2/adenovirus E1B 19kDa-interacting protein3) regulate autophagy and mitophagy in renal tubular cells in acute kidney injury by Masayuki Ishihara 1, Madoka Urushido 2, Kazu Hamada
Follicular Lymphoma. ced3 APOPTOSIS. *In the nematode Caenorhabditis elegans 131 of the organism's 1031 cells die during development.
 Harvard-MIT Division of Health Sciences and Technology HST.176: Cellular and Molecular Immunology Course Director: Dr. Shiv Pillai Follicular Lymphoma 1. Characterized by t(14:18) translocation 2. Ig heavy
Harvard-MIT Division of Health Sciences and Technology HST.176: Cellular and Molecular Immunology Course Director: Dr. Shiv Pillai Follicular Lymphoma 1. Characterized by t(14:18) translocation 2. Ig heavy
Bcl-X L interacts with Apaf-1 and inhibits Apaf-1-dependent caspase-9 activation (apoptosis cell death)
 Proc. Natl. Acad. Sci. USA Vol. 95, pp. 4386 4391, April 1998 Cell Biology Bcl-X L interacts with Apaf-1 and inhibits Apaf-1-dependent caspase-9 activation (apoptosis cell death) YUANMING HU, MARY A. BENEDICT,
Proc. Natl. Acad. Sci. USA Vol. 95, pp. 4386 4391, April 1998 Cell Biology Bcl-X L interacts with Apaf-1 and inhibits Apaf-1-dependent caspase-9 activation (apoptosis cell death) YUANMING HU, MARY A. BENEDICT,
Supplementary Table; Supplementary Figures and legends S1-S21; Supplementary Materials and Methods
 Silva et al. PTEN posttranslational inactivation and hyperactivation of the PI3K/Akt pathway sustain primary T cell leukemia viability Supplementary Table; Supplementary Figures and legends S1-S21; Supplementary
Silva et al. PTEN posttranslational inactivation and hyperactivation of the PI3K/Akt pathway sustain primary T cell leukemia viability Supplementary Table; Supplementary Figures and legends S1-S21; Supplementary
Table S1. New colony formation 7 days after stimulation with doxo and VCR in JURKAT cells
 Table S1. New colony formation 7 days after stimulation with and in JURKAT cells drug co + number of colonies 7±14 4±7 48±11 JURKAT cells were stimulated and analyzed as in Table 1. Drug concentrations
Table S1. New colony formation 7 days after stimulation with and in JURKAT cells drug co + number of colonies 7±14 4±7 48±11 JURKAT cells were stimulated and analyzed as in Table 1. Drug concentrations
Determination of the temporal pattern and importance of BALF1 expression in Epstein-Barr viral infection
 Determination of the temporal pattern and importance of BALF1 expression in Epstein-Barr viral infection Melissa Mihelidakis May 6, 2004 7.340 Research Proposal Introduction Apoptosis, or programmed cell
Determination of the temporal pattern and importance of BALF1 expression in Epstein-Barr viral infection Melissa Mihelidakis May 6, 2004 7.340 Research Proposal Introduction Apoptosis, or programmed cell
Impact factor: Reporter:4A1H0019 Chen Zi Hao 4A1H0023 Huang Wan ting 4A1H0039 Sue Yi Zhu 4A1H0070 Lin Guan cheng 4A1H0077 Chen Bo xuan
 Curcumin Protects Neonatal Rat Cardiomyocytes against High Glucose-Induced Apoptosis via PI3K/Akt Signalling Pathway Wei Yu,1,2 Wenliang Zha,1 Zhiqiang Ke,1 Qing Min,2 Cairong Li,1 Huirong Sun,3 and Chao
Curcumin Protects Neonatal Rat Cardiomyocytes against High Glucose-Induced Apoptosis via PI3K/Akt Signalling Pathway Wei Yu,1,2 Wenliang Zha,1 Zhiqiang Ke,1 Qing Min,2 Cairong Li,1 Huirong Sun,3 and Chao
IMMP8-1. Different Mechanisms of Androg and IPAD on Apoptosis Induction in Cervical Cancer Cells
 IMMP8-1 Different Mechanisms of Androg and IPAD on Apoptosis Induction in Cervical Cancer Cells Assanan Dokmaikaew* Tipaya Ekalaksananan** Dr.Chamsai Pientong** ABSTRACT Androg and IPAD are recently known
IMMP8-1 Different Mechanisms of Androg and IPAD on Apoptosis Induction in Cervical Cancer Cells Assanan Dokmaikaew* Tipaya Ekalaksananan** Dr.Chamsai Pientong** ABSTRACT Androg and IPAD are recently known
Supplementary Information
 Supplementary Information Supplementary Figure 1. CD4 + T cell activation and lack of apoptosis after crosslinking with anti-cd3 + anti-cd28 + anti-cd160. (a) Flow cytometry of anti-cd160 (5D.10A11) binding
Supplementary Information Supplementary Figure 1. CD4 + T cell activation and lack of apoptosis after crosslinking with anti-cd3 + anti-cd28 + anti-cd160. (a) Flow cytometry of anti-cd160 (5D.10A11) binding
EML Erythroid and Neutrophil Differentiation Protocols Cristina Pina 1*, Cristina Fugazza 2 and Tariq Enver 3
 EML Erythroid and Neutrophil Differentiation Protocols Cristina Pina 1*, Cristina Fugazza 2 and Tariq Enver 3 1 Department of Haematology, University of Cambridge, Cambridge, UK; 2 Dipartimento de Biotecnologie
EML Erythroid and Neutrophil Differentiation Protocols Cristina Pina 1*, Cristina Fugazza 2 and Tariq Enver 3 1 Department of Haematology, University of Cambridge, Cambridge, UK; 2 Dipartimento de Biotecnologie
Apoptosis Oncogenes. Srbová Martina
 Apoptosis Oncogenes Srbová Martina Cell Cycle Control point Cyclin B Cdk1 Cyclin D Cdk4 Cdk6 Cyclin A Cdk2 Cyclin E Cdk2 Cyclin-dependent kinase (Cdk) have to bind a cyclin to become active Regulation
Apoptosis Oncogenes Srbová Martina Cell Cycle Control point Cyclin B Cdk1 Cyclin D Cdk4 Cdk6 Cyclin A Cdk2 Cyclin E Cdk2 Cyclin-dependent kinase (Cdk) have to bind a cyclin to become active Regulation
HCC1937 is the HCC1937-pcDNA3 cell line, which was derived from a breast cancer with a mutation
 SUPPLEMENTARY INFORMATION Materials and Methods Human cell lines and culture conditions HCC1937 is the HCC1937-pcDNA3 cell line, which was derived from a breast cancer with a mutation in exon 20 of BRCA1
SUPPLEMENTARY INFORMATION Materials and Methods Human cell lines and culture conditions HCC1937 is the HCC1937-pcDNA3 cell line, which was derived from a breast cancer with a mutation in exon 20 of BRCA1
#19 Apoptosis Chapter 9. Neelu Yadav PhD
 #19 Apoptosis Chapter 9 Neelu Yadav PhD Neelu.Yadav@Roswellpark.org Why cells decide to die? - Stress, harmful, not needed - Completed its life span Death stimulation or Stress Cell Survival Death Functions
#19 Apoptosis Chapter 9 Neelu Yadav PhD Neelu.Yadav@Roswellpark.org Why cells decide to die? - Stress, harmful, not needed - Completed its life span Death stimulation or Stress Cell Survival Death Functions
An epithelial-to-mesenchymal transition-inducing potential of. granulocyte macrophage colony-stimulating factor in colon. cancer
 An epithelial-to-mesenchymal transition-inducing potential of granulocyte macrophage colony-stimulating factor in colon cancer Yaqiong Chen, Zhi Zhao, Yu Chen, Zhonglin Lv, Xin Ding, Renxi Wang, He Xiao,
An epithelial-to-mesenchymal transition-inducing potential of granulocyte macrophage colony-stimulating factor in colon cancer Yaqiong Chen, Zhi Zhao, Yu Chen, Zhonglin Lv, Xin Ding, Renxi Wang, He Xiao,
Supporting Information Table of Contents
 Supporting Information Table of Contents Supporting Information Figure 1 Page 2 Supporting Information Figure 2 Page 4 Supporting Information Figure 3 Page 5 Supporting Information Figure 4 Page 6 Supporting
Supporting Information Table of Contents Supporting Information Figure 1 Page 2 Supporting Information Figure 2 Page 4 Supporting Information Figure 3 Page 5 Supporting Information Figure 4 Page 6 Supporting
Recombinant Protein Expression Retroviral system
 Recombinant Protein Expression Retroviral system Viruses Contains genome DNA or RNA Genome encased in a protein coat or capsid. Some viruses have membrane covering protein coat enveloped virus Ø Essential
Recombinant Protein Expression Retroviral system Viruses Contains genome DNA or RNA Genome encased in a protein coat or capsid. Some viruses have membrane covering protein coat enveloped virus Ø Essential
Electron micrograph of phosphotungstanic acid-stained exosomes derived from murine
 1 SUPPLEMENTARY INFORMATION SUPPLEMENTARY FIGURES Supplementary Figure 1. Physical properties of murine DC-derived exosomes. a, Electron micrograph of phosphotungstanic acid-stained exosomes derived from
1 SUPPLEMENTARY INFORMATION SUPPLEMENTARY FIGURES Supplementary Figure 1. Physical properties of murine DC-derived exosomes. a, Electron micrograph of phosphotungstanic acid-stained exosomes derived from
Figure S1. Western blot analysis of clathrin RNA interference in human DCs Human immature DCs were transfected with 100 nm Clathrin SMARTpool or
 Figure S1. Western blot analysis of clathrin RNA interference in human DCs Human immature DCs were transfected with 100 nm Clathrin SMARTpool or control nontargeting sirnas. At 90 hr after transfection,
Figure S1. Western blot analysis of clathrin RNA interference in human DCs Human immature DCs were transfected with 100 nm Clathrin SMARTpool or control nontargeting sirnas. At 90 hr after transfection,
Enzyme-coupled Receptors. Cell-surface receptors 1. Ion-channel-coupled receptors 2. G-protein-coupled receptors 3. Enzyme-coupled receptors
 Enzyme-coupled Receptors Cell-surface receptors 1. Ion-channel-coupled receptors 2. G-protein-coupled receptors 3. Enzyme-coupled receptors Cell-surface receptors allow a flow of ions across the plasma
Enzyme-coupled Receptors Cell-surface receptors 1. Ion-channel-coupled receptors 2. G-protein-coupled receptors 3. Enzyme-coupled receptors Cell-surface receptors allow a flow of ions across the plasma
MATERIALS AND METHODS. Neutralizing antibodies specific to mouse Dll1, Dll4, J1 and J2 were prepared as described. 1,2 All
 MATERIALS AND METHODS Antibodies (Abs), flow cytometry analysis and cell lines Neutralizing antibodies specific to mouse Dll1, Dll4, J1 and J2 were prepared as described. 1,2 All other antibodies used
MATERIALS AND METHODS Antibodies (Abs), flow cytometry analysis and cell lines Neutralizing antibodies specific to mouse Dll1, Dll4, J1 and J2 were prepared as described. 1,2 All other antibodies used
SUPPLEMENTARY INFORMATION
 doi:10.1038/nature12215 Supplementary Figure 1. The effects of full and dissociated GR agonists in supporting BFU-E self-renewal divisions. BFU-Es were cultured in self-renewal medium with indicated GR
doi:10.1038/nature12215 Supplementary Figure 1. The effects of full and dissociated GR agonists in supporting BFU-E self-renewal divisions. BFU-Es were cultured in self-renewal medium with indicated GR
Muse Assays for Cell Analysis
 Muse Assays for Cell Analysis Multiple Assay Outputs for Cell Analysis Cell Health Cell Signalling Immunology Muse Count & Viability Kit Muse Cell Cycle Kit Muse Annexin V & Dead Cell Kit Muse Caspase
Muse Assays for Cell Analysis Multiple Assay Outputs for Cell Analysis Cell Health Cell Signalling Immunology Muse Count & Viability Kit Muse Cell Cycle Kit Muse Annexin V & Dead Cell Kit Muse Caspase
Pro-apoptotic signalling through Toll-like receptor 3 involves TRIF-dependent
 Pro-apoptotic signalling through Toll-like receptor 3 involves TRIF-dependent activation of caspase-8 and is under the control of inhibitor of apoptosis proteins in melanoma cells Arnim Weber, Zofia Kirejczyk,
Pro-apoptotic signalling through Toll-like receptor 3 involves TRIF-dependent activation of caspase-8 and is under the control of inhibitor of apoptosis proteins in melanoma cells Arnim Weber, Zofia Kirejczyk,
The functional investigation of the interaction between TATA-associated factor 3 (TAF3) and p53 protein
 THESIS BOOK The functional investigation of the interaction between TATA-associated factor 3 (TAF3) and p53 protein Orsolya Buzás-Bereczki Supervisors: Dr. Éva Bálint Dr. Imre Miklós Boros University of
THESIS BOOK The functional investigation of the interaction between TATA-associated factor 3 (TAF3) and p53 protein Orsolya Buzás-Bereczki Supervisors: Dr. Éva Bálint Dr. Imre Miklós Boros University of
Introduction: 年 Fas signal-mediated apoptosis. PI3K/Akt
 Fas-ligand (CD95-L; Fas-L) Fas (CD95) Fas (apoptosis) 年 了 不 度 Fas Fas-L 力 不 Fas/Fas-L T IL-10Fas/Fas-L 不 年 Fas signal-mediated apoptosis 度降 不 不 力 U-118, HeLa, A549, Huh-7 MCF-7, HepG2. PI3K/Akt FasPI3K/Akt
Fas-ligand (CD95-L; Fas-L) Fas (CD95) Fas (apoptosis) 年 了 不 度 Fas Fas-L 力 不 Fas/Fas-L T IL-10Fas/Fas-L 不 年 Fas signal-mediated apoptosis 度降 不 不 力 U-118, HeLa, A549, Huh-7 MCF-7, HepG2. PI3K/Akt FasPI3K/Akt
Supplementary figure legends
 Supplementary figure legends Supplementary Figure 1. Exposure of CRT occurs independently from the apoptosisassociated loss of the mitochondrial membrane potential (MMP). (A) HeLa cells treated with MTX
Supplementary figure legends Supplementary Figure 1. Exposure of CRT occurs independently from the apoptosisassociated loss of the mitochondrial membrane potential (MMP). (A) HeLa cells treated with MTX
Lecture 14 - The cell cycle and cell death
 02.17.10 Lecture 14 - The cell cycle and cell death The cell cycle: cells duplicate their contents and divide The cell cycle may be divided into 4 phases The cell cycle triggers essential processes (DNA
02.17.10 Lecture 14 - The cell cycle and cell death The cell cycle: cells duplicate their contents and divide The cell cycle may be divided into 4 phases The cell cycle triggers essential processes (DNA
TFEB-mediated increase in peripheral lysosomes regulates. Store Operated Calcium Entry
 TFEB-mediated increase in peripheral lysosomes regulates Store Operated Calcium Entry Luigi Sbano, Massimo Bonora, Saverio Marchi, Federica Baldassari, Diego L. Medina, Andrea Ballabio, Carlotta Giorgi
TFEB-mediated increase in peripheral lysosomes regulates Store Operated Calcium Entry Luigi Sbano, Massimo Bonora, Saverio Marchi, Federica Baldassari, Diego L. Medina, Andrea Ballabio, Carlotta Giorgi
Genome of Hepatitis B Virus. VIRAL ONCOGENE Dr. Yahwardiah Siregar, PhD Dr. Sry Suryani Widjaja, Mkes Biochemistry Department
 Genome of Hepatitis B Virus VIRAL ONCOGENE Dr. Yahwardiah Siregar, PhD Dr. Sry Suryani Widjaja, Mkes Biochemistry Department Proto Oncogen and Oncogen Oncogen Proteins that possess the ability to cause
Genome of Hepatitis B Virus VIRAL ONCOGENE Dr. Yahwardiah Siregar, PhD Dr. Sry Suryani Widjaja, Mkes Biochemistry Department Proto Oncogen and Oncogen Oncogen Proteins that possess the ability to cause
Cells communicate with each other via signaling ligands which interact with receptors located on the surface or inside the target cell.
 BENG 100 Frontiers of Biomedical Engineering Professor Mark Saltzman Chapter 6 SUMMARY In this chapter, cell signaling was presented within the context of three physiological systems that utilize communication
BENG 100 Frontiers of Biomedical Engineering Professor Mark Saltzman Chapter 6 SUMMARY In this chapter, cell signaling was presented within the context of three physiological systems that utilize communication
Part-4. Cell cycle regulatory protein 5 (Cdk5) A novel target of ERK in Carb induced cell death
 Part-4 Cell cycle regulatory protein 5 (Cdk5) A novel target of ERK in Carb induced cell death 95 1. Introduction The process of replicating DNA and dividing cells can be described as a series of coordinated
Part-4 Cell cycle regulatory protein 5 (Cdk5) A novel target of ERK in Carb induced cell death 95 1. Introduction The process of replicating DNA and dividing cells can be described as a series of coordinated
C-Phycocyanin (C-PC) is a n«sjfc&c- waefc-jduble phycobiliprotein. pigment isolated from Spirulina platensis. This water- soluble protein pigment is
 ' ^Summary C-Phycocyanin (C-PC) is a n«sjfc&c- waefc-jduble phycobiliprotein pigment isolated from Spirulina platensis. This water- soluble protein pigment is of greater importance because of its various
' ^Summary C-Phycocyanin (C-PC) is a n«sjfc&c- waefc-jduble phycobiliprotein pigment isolated from Spirulina platensis. This water- soluble protein pigment is of greater importance because of its various
Supplementary Figure 1 CD4 + T cells from PKC-θ null mice are defective in NF-κB activation during T cell receptor signaling. CD4 + T cells were
 Supplementary Figure 1 CD4 + T cells from PKC-θ null mice are defective in NF-κB activation during T cell receptor signaling. CD4 + T cells were isolated from wild type (PKC-θ- WT) or PKC-θ null (PKC-θ-KO)
Supplementary Figure 1 CD4 + T cells from PKC-θ null mice are defective in NF-κB activation during T cell receptor signaling. CD4 + T cells were isolated from wild type (PKC-θ- WT) or PKC-θ null (PKC-θ-KO)
Materials and Methods , The two-hybrid principle.
 The enzymatic activity of an unknown protein which cleaves the phosphodiester bond between the tyrosine residue of a viral protein and the 5 terminus of the picornavirus RNA Introduction Every day there
The enzymatic activity of an unknown protein which cleaves the phosphodiester bond between the tyrosine residue of a viral protein and the 5 terminus of the picornavirus RNA Introduction Every day there
Supplemental Experimental Procedures
 Cell Stem Cell, Volume 2 Supplemental Data A Temporal Switch from Notch to Wnt Signaling in Muscle Stem Cells Is Necessary for Normal Adult Myogenesis Andrew S. Brack, Irina M. Conboy, Michael J. Conboy,
Cell Stem Cell, Volume 2 Supplemental Data A Temporal Switch from Notch to Wnt Signaling in Muscle Stem Cells Is Necessary for Normal Adult Myogenesis Andrew S. Brack, Irina M. Conboy, Michael J. Conboy,
Research progress on the use of estrogen receptor agonist for treatment of spinal cord injury
 Research progress on the use of estrogen receptor agonist for treatment of spinal cord injury Swapan K. Ray, PhD Professor, Department of Pathology, Microbiology, and Immunology USC School of Medicine,
Research progress on the use of estrogen receptor agonist for treatment of spinal cord injury Swapan K. Ray, PhD Professor, Department of Pathology, Microbiology, and Immunology USC School of Medicine,
Supplemental material for Hernandez et al. Dicoumarol downregulates human PTTG1/Securin mrna expression. through inhibition of Hsp90
 Supplemental material for Hernandez et al. Dicoumarol downregulates human PTTG1/Securin mrna expression through inhibition of Hsp90 Dicoumarol-Sepharose co-precipitation. Hsp90 inhibitors can co-precipitate
Supplemental material for Hernandez et al. Dicoumarol downregulates human PTTG1/Securin mrna expression through inhibition of Hsp90 Dicoumarol-Sepharose co-precipitation. Hsp90 inhibitors can co-precipitate
Supplemental Data. TGF-β-mediated mir-181a expression promotes breast cancer metastasis by targeting Bim.
 Supplemental Data TGF-β-mediated mir-181a expression promotes breast cancer metastasis by targeting Bim. Molly A. Taylor 1, Khalid Sossey-Alaoui 2, Cheryl L. Thompson 3, David Danielpour 4, and William
Supplemental Data TGF-β-mediated mir-181a expression promotes breast cancer metastasis by targeting Bim. Molly A. Taylor 1, Khalid Sossey-Alaoui 2, Cheryl L. Thompson 3, David Danielpour 4, and William
Supplementary Figure 1: Hsp60 / IEC mice are embryonically lethal (A) Light microscopic pictures show mouse embryos at developmental stage E12.
 Supplementary Figure 1: Hsp60 / IEC mice are embryonically lethal (A) Light microscopic pictures show mouse embryos at developmental stage E12.5 and E13.5 prepared from uteri of dams and subsequently genotyped.
Supplementary Figure 1: Hsp60 / IEC mice are embryonically lethal (A) Light microscopic pictures show mouse embryos at developmental stage E12.5 and E13.5 prepared from uteri of dams and subsequently genotyped.
Neurotrophic factor GDNF and camp suppress glucocorticoid-inducible PNMT expression in a mouse pheochromocytoma model.
 161 Neurotrophic factor GDNF and camp suppress glucocorticoid-inducible PNMT expression in a mouse pheochromocytoma model. Marian J. Evinger a, James F. Powers b and Arthur S. Tischler b a. Department
161 Neurotrophic factor GDNF and camp suppress glucocorticoid-inducible PNMT expression in a mouse pheochromocytoma model. Marian J. Evinger a, James F. Powers b and Arthur S. Tischler b a. Department
Page 32 AP Biology: 2013 Exam Review CONCEPT 6 REGULATION
 Page 32 AP Biology: 2013 Exam Review CONCEPT 6 REGULATION 1. Feedback a. Negative feedback mechanisms maintain dynamic homeostasis for a particular condition (variable) by regulating physiological processes,
Page 32 AP Biology: 2013 Exam Review CONCEPT 6 REGULATION 1. Feedback a. Negative feedback mechanisms maintain dynamic homeostasis for a particular condition (variable) by regulating physiological processes,
Receptor mediated Signal Transduction
 Receptor mediated Signal Transduction G-protein-linked receptors adenylyl cyclase camp PKA Organization of receptor protein-tyrosine kinases From G.M. Cooper, The Cell. A molecular approach, 2004, third
Receptor mediated Signal Transduction G-protein-linked receptors adenylyl cyclase camp PKA Organization of receptor protein-tyrosine kinases From G.M. Cooper, The Cell. A molecular approach, 2004, third
Serum Amyloid A3 Gene Expression in Adipocytes is an Indicator. of the Interaction with Macrophages
 Serum Amyloid A3 Gene Expression in Adipocytes is an Indicator of the Interaction with Macrophages Yohei Sanada, Takafumi Yamamoto, Rika Satake, Akiko Yamashita, Sumire Kanai, Norihisa Kato, Fons AJ van
Serum Amyloid A3 Gene Expression in Adipocytes is an Indicator of the Interaction with Macrophages Yohei Sanada, Takafumi Yamamoto, Rika Satake, Akiko Yamashita, Sumire Kanai, Norihisa Kato, Fons AJ van
THE INFLUENCE OF SODIUM FLUORIDE ON THE CLONOGENECITY OF HUMAN HEMATOPOIETIC PROGENITOR CELLS: PRELIMINARY REPORT
 168 Fluoride Vol. 33 No. 4 168-173 2 Research Report THE INFLUENCE OF SODIUM FLUORIDE ON THE CLONOGENECITY OF HUMAN HEMATOPOIETIC PROGENITOR CELLS: PRELIMINARY REPORT Boguslaw Machaliński, a Maria Zejmo,
168 Fluoride Vol. 33 No. 4 168-173 2 Research Report THE INFLUENCE OF SODIUM FLUORIDE ON THE CLONOGENECITY OF HUMAN HEMATOPOIETIC PROGENITOR CELLS: PRELIMINARY REPORT Boguslaw Machaliński, a Maria Zejmo,
Introduction NEOPLASIA. Qun Liu and Yair Gazitt
 NEOPLASIA Potentiation of dexamethasone-, paclitaxel-, and Ad-p53 induced apoptosis by Bcl-2 antisense oligodeoxynucleotides in drug-resistant multiple myeloma cells Qun Liu and Yair Gazitt Overexpression
NEOPLASIA Potentiation of dexamethasone-, paclitaxel-, and Ad-p53 induced apoptosis by Bcl-2 antisense oligodeoxynucleotides in drug-resistant multiple myeloma cells Qun Liu and Yair Gazitt Overexpression
Nature Genetics: doi: /ng Supplementary Figure 1
 Supplementary Figure 1 MSI2 interactors are associated with the riboproteome and are functionally relevant. (a) Coomassie blue staining of FLAG-MSI2 immunoprecipitated complexes. (b) GO analysis of MSI2-interacting
Supplementary Figure 1 MSI2 interactors are associated with the riboproteome and are functionally relevant. (a) Coomassie blue staining of FLAG-MSI2 immunoprecipitated complexes. (b) GO analysis of MSI2-interacting
Samali A Figure S1.
 Deegan S, Saveljeva S, Logue SE, Pakos-Zebrucka K, Gupta S, Vandenabeele P, Bertrand MJ,Samali A. (2014) Deficiency in the mitochondrial apoptotic pathway reveals the toxic potential of autophagy under
Deegan S, Saveljeva S, Logue SE, Pakos-Zebrucka K, Gupta S, Vandenabeele P, Bertrand MJ,Samali A. (2014) Deficiency in the mitochondrial apoptotic pathway reveals the toxic potential of autophagy under
Supplemental Figure 1. Signature gene expression in in vitro differentiated Th0, Th1, Th2, Th17 and Treg cells. (A) Naïve CD4 + T cells were cultured
 Supplemental Figure 1. Signature gene expression in in vitro differentiated Th0, Th1, Th2, Th17 and Treg cells. (A) Naïve CD4 + T cells were cultured under Th0, Th1, Th2, Th17, and Treg conditions. mrna
Supplemental Figure 1. Signature gene expression in in vitro differentiated Th0, Th1, Th2, Th17 and Treg cells. (A) Naïve CD4 + T cells were cultured under Th0, Th1, Th2, Th17, and Treg conditions. mrna
Construction of a hepatocellular carcinoma cell line that stably expresses stathmin with a Ser25 phosphorylation site mutation
 Construction of a hepatocellular carcinoma cell line that stably expresses stathmin with a Ser25 phosphorylation site mutation J. Du 1, Z.H. Tao 2, J. Li 2, Y.K. Liu 3 and L. Gan 2 1 Department of Chemistry,
Construction of a hepatocellular carcinoma cell line that stably expresses stathmin with a Ser25 phosphorylation site mutation J. Du 1, Z.H. Tao 2, J. Li 2, Y.K. Liu 3 and L. Gan 2 1 Department of Chemistry,
Page 39 of 44. 8h LTA & AT h PepG & AT h LTA
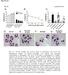 Page 39 of 44 Fig. S1 A: B: C: D: 8h LTA 8h LTA & AT7519 E: F: 8h PepG G: 8h PepG & AT7519 Fig. S1. AT7519 overrides the survival effects of lipoteichoic acid (LTA) and peptidoglycan (PepG). (A) Human
Page 39 of 44 Fig. S1 A: B: C: D: 8h LTA 8h LTA & AT7519 E: F: 8h PepG G: 8h PepG & AT7519 Fig. S1. AT7519 overrides the survival effects of lipoteichoic acid (LTA) and peptidoglycan (PepG). (A) Human
Supplemental Information
 Supplemental Information Tobacco-specific Carcinogen Induces DNA Methyltransferases 1 Accumulation through AKT/GSK3β/βTrCP/hnRNP-U in Mice and Lung Cancer patients Ruo-Kai Lin, 1 Yi-Shuan Hsieh, 2 Pinpin
Supplemental Information Tobacco-specific Carcinogen Induces DNA Methyltransferases 1 Accumulation through AKT/GSK3β/βTrCP/hnRNP-U in Mice and Lung Cancer patients Ruo-Kai Lin, 1 Yi-Shuan Hsieh, 2 Pinpin
T Cell Development. Xuefang Cao, MD, PhD. November 3, 2015
 T Cell Development Xuefang Cao, MD, PhD November 3, 2015 Thymocytes in the cortex of the thymus Early thymocytes development Positive and negative selection Lineage commitment Exit from the thymus and
T Cell Development Xuefang Cao, MD, PhD November 3, 2015 Thymocytes in the cortex of the thymus Early thymocytes development Positive and negative selection Lineage commitment Exit from the thymus and
Supplementary Figure 1 Role of Raf-1 in TLR2-Dectin-1-mediated cytokine expression
 Supplementary Figure 1 Supplementary Figure 1 Role of Raf-1 in TLR2-Dectin-1-mediated cytokine expression. Quantitative real-time PCR of indicated mrnas in DCs stimulated with TLR2-Dectin-1 agonist zymosan
Supplementary Figure 1 Supplementary Figure 1 Role of Raf-1 in TLR2-Dectin-1-mediated cytokine expression. Quantitative real-time PCR of indicated mrnas in DCs stimulated with TLR2-Dectin-1 agonist zymosan
Supplementary Fig. 1. GPRC5A post-transcriptionally down-regulates EGFR expression. (a) Plot of the changes in steady state mrna levels versus
 Supplementary Fig. 1. GPRC5A post-transcriptionally down-regulates EGFR expression. (a) Plot of the changes in steady state mrna levels versus changes in corresponding proteins between wild type and Gprc5a-/-
Supplementary Fig. 1. GPRC5A post-transcriptionally down-regulates EGFR expression. (a) Plot of the changes in steady state mrna levels versus changes in corresponding proteins between wild type and Gprc5a-/-
REGULATION OF ENZYME ACTIVITY. Medical Biochemistry, Lecture 25
 REGULATION OF ENZYME ACTIVITY Medical Biochemistry, Lecture 25 Lecture 25, Outline General properties of enzyme regulation Regulation of enzyme concentrations Allosteric enzymes and feedback inhibition
REGULATION OF ENZYME ACTIVITY Medical Biochemistry, Lecture 25 Lecture 25, Outline General properties of enzyme regulation Regulation of enzyme concentrations Allosteric enzymes and feedback inhibition
Molecular Hematopathology Leukemias I. January 14, 2005
 Molecular Hematopathology Leukemias I January 14, 2005 Chronic Myelogenous Leukemia Diagnosis requires presence of Philadelphia chromosome t(9;22)(q34;q11) translocation BCR-ABL is the result BCR on chr
Molecular Hematopathology Leukemias I January 14, 2005 Chronic Myelogenous Leukemia Diagnosis requires presence of Philadelphia chromosome t(9;22)(q34;q11) translocation BCR-ABL is the result BCR on chr
RCPA Research Award Final Progress Review
 RCPA Research Award 2010-2011 Final Progress Review Name: Dr Craig Wallington-Beddoe Degree/Institution/Year: PhD, The University of Sydney, Year 2 Research Project Title: New Therapeutic Strategies for
RCPA Research Award 2010-2011 Final Progress Review Name: Dr Craig Wallington-Beddoe Degree/Institution/Year: PhD, The University of Sydney, Year 2 Research Project Title: New Therapeutic Strategies for
T-cell activation T cells migrate to secondary lymphoid tissues where they interact with antigen, antigen-presenting cells, and other lymphocytes:
 Interactions between innate immunity & adaptive immunity What happens to T cells after they leave the thymus? Naïve T cells exit the thymus and enter the bloodstream. If they remain in the bloodstream,
Interactions between innate immunity & adaptive immunity What happens to T cells after they leave the thymus? Naïve T cells exit the thymus and enter the bloodstream. If they remain in the bloodstream,
T-cell activation T cells migrate to secondary lymphoid tissues where they interact with antigen, antigen-presenting cells, and other lymphocytes:
 Interactions between innate immunity & adaptive immunity What happens to T cells after they leave the thymus? Naïve T cells exit the thymus and enter the bloodstream. If they remain in the bloodstream,
Interactions between innate immunity & adaptive immunity What happens to T cells after they leave the thymus? Naïve T cells exit the thymus and enter the bloodstream. If they remain in the bloodstream,
Under the Radar Screen: How Bugs Trick Our Immune Defenses
 Under the Radar Screen: How Bugs Trick Our Immune Defenses Session 8: Apoptosis Marie-Eve Paquet and Gijsbert Grotenbreg Whitehead Institute for Biomedical Research Myxoma virus Poxvirus Infects rabbits
Under the Radar Screen: How Bugs Trick Our Immune Defenses Session 8: Apoptosis Marie-Eve Paquet and Gijsbert Grotenbreg Whitehead Institute for Biomedical Research Myxoma virus Poxvirus Infects rabbits
Bioluminescence Resonance Energy Transfer (BRET)-based studies of receptor dynamics in living cells with Berthold s Mithras
 Bioluminescence Resonance Energy Transfer (BRET)-based studies of receptor dynamics in living cells with Berthold s Mithras Tarik Issad, Ralf Jockers and Stefano Marullo 1 Because they play a pivotal role
Bioluminescence Resonance Energy Transfer (BRET)-based studies of receptor dynamics in living cells with Berthold s Mithras Tarik Issad, Ralf Jockers and Stefano Marullo 1 Because they play a pivotal role
McAb and rhil-2 activated bone marrow on the killing and purging of leukemia cells
 Effects of McAb and rhil-2 activated bone marrow on the killing and purging of leukemia cells X.C. Wei, D.D. Yang, X.R. Han, Y.A. Zhao, Y.C. Li, L.J. Zhang and J.J. Wang Institute of hematological research,
Effects of McAb and rhil-2 activated bone marrow on the killing and purging of leukemia cells X.C. Wei, D.D. Yang, X.R. Han, Y.A. Zhao, Y.C. Li, L.J. Zhang and J.J. Wang Institute of hematological research,
Cellular Physiology and Biochemistry
 Original Paper 2015 The Author(s). 2015 Published The Author(s) by S. Karger AG, Basel Published online: November 27, 2015 www.karger.com/cpb Published by S. Karger AG, Basel 2194 1421-9778/15/0376-2194$39.50/0
Original Paper 2015 The Author(s). 2015 Published The Author(s) by S. Karger AG, Basel Published online: November 27, 2015 www.karger.com/cpb Published by S. Karger AG, Basel 2194 1421-9778/15/0376-2194$39.50/0
7.06 Cell Biology Exam #3 April 23, 2002
 RECITATION TA: NAME: 7.06 Cell Biology Exam #3 April 23, 2002 This is an open book exam and you are allowed access to books, notes, and calculators. Please limit your answers to the spaces allotted after
RECITATION TA: NAME: 7.06 Cell Biology Exam #3 April 23, 2002 This is an open book exam and you are allowed access to books, notes, and calculators. Please limit your answers to the spaces allotted after
Figure S1. Analysis of genomic and cdna sequences of the targeted regions in WT-KI and
 Figure S1. Analysis of genomic and sequences of the targeted regions in and indicated mutant KI cells, with WT and corresponding mutant sequences underlined. (A) cells; (B) K21E-KI cells; (C) D33A-KI cells;
Figure S1. Analysis of genomic and sequences of the targeted regions in and indicated mutant KI cells, with WT and corresponding mutant sequences underlined. (A) cells; (B) K21E-KI cells; (C) D33A-KI cells;
BIO360 Fall 2013 Quiz 1
 BIO360 Fall 2013 Quiz 1 1. Examine the diagram below. There are two homologous copies of chromosome one and the allele of YFG carried on the light gray chromosome has undergone a loss-of-function mutation.
BIO360 Fall 2013 Quiz 1 1. Examine the diagram below. There are two homologous copies of chromosome one and the allele of YFG carried on the light gray chromosome has undergone a loss-of-function mutation.
Supplementary Materials and Methods
 Supplementary Materials and Methods Immunoblotting Immunoblot analysis was performed as described previously (1). Due to high-molecular weight of MUC4 (~ 950 kda) and MUC1 (~ 250 kda) proteins, electrophoresis
Supplementary Materials and Methods Immunoblotting Immunoblot analysis was performed as described previously (1). Due to high-molecular weight of MUC4 (~ 950 kda) and MUC1 (~ 250 kda) proteins, electrophoresis
Supporting Information. FADD regulates NF-кB activation and promotes ubiquitination of cflip L to induce. apoptosis
 1 2 Supporting Information 3 4 5 FADD regulates NF-кB activation and promotes ubiquitination of cflip L to induce apoptosis 6 7 Kishu Ranjan and Chandramani Pathak* 8 9 Department of Cell Biology, School
1 2 Supporting Information 3 4 5 FADD regulates NF-кB activation and promotes ubiquitination of cflip L to induce apoptosis 6 7 Kishu Ranjan and Chandramani Pathak* 8 9 Department of Cell Biology, School
AHI-1 interacts with BCR-ABL and modulates BCR-ABL transforming activity and imatinib response of CML stem/progenitor cells
 AHI-1 interacts with BCR-ABL and modulates BCR-ABL transforming activity and imatinib response of CML stem/progenitor cells Liang L. Zhou, 1 Yun Zhao, 1 Ashley Ringrose, 1 Donna DeGeer, 1 Erin Kennah,
AHI-1 interacts with BCR-ABL and modulates BCR-ABL transforming activity and imatinib response of CML stem/progenitor cells Liang L. Zhou, 1 Yun Zhao, 1 Ashley Ringrose, 1 Donna DeGeer, 1 Erin Kennah,
Microglia, Inflammation, and FTD
 FTD Minicourse April, 2009 Microglia, Inflammation, and FTD Li Gan, Ph.D Gladstone Institute of Neurological Disease University of California, San Francisco Outline Why study inflammation in neurodegeneration?
FTD Minicourse April, 2009 Microglia, Inflammation, and FTD Li Gan, Ph.D Gladstone Institute of Neurological Disease University of California, San Francisco Outline Why study inflammation in neurodegeneration?
Identification and characterization of genes responsive to apoptosis: Application of DNA chip technology and mrna differential display
 Histol Histopathol (2000) 15: 1271-1 284 http://www.ehu.es/histol-histopathol Histology and H istopat hology Cellular and Molecular Biology Invited Revie W Identification and characterization of genes
Histol Histopathol (2000) 15: 1271-1 284 http://www.ehu.es/histol-histopathol Histology and H istopat hology Cellular and Molecular Biology Invited Revie W Identification and characterization of genes
CYTOKINE RECEPTORS AND SIGNAL TRANSDUCTION
 CYTOKINE RECEPTORS AND SIGNAL TRANSDUCTION What is Cytokine? Secreted popypeptide (protein) involved in cell-to-cell signaling. Acts in paracrine or autocrine fashion through specific cellular receptors.
CYTOKINE RECEPTORS AND SIGNAL TRANSDUCTION What is Cytokine? Secreted popypeptide (protein) involved in cell-to-cell signaling. Acts in paracrine or autocrine fashion through specific cellular receptors.
Phospho-AKT Sampler Kit
 Phospho-AKT Sampler Kit E 0 5 1 0 0 3 Kits Includes Cat. Quantity Application Reactivity Source Akt (Ab-473) Antibody E021054-1 50μg/50μl IHC, WB Human, Mouse, Rat Rabbit Akt (Phospho-Ser473) Antibody
Phospho-AKT Sampler Kit E 0 5 1 0 0 3 Kits Includes Cat. Quantity Application Reactivity Source Akt (Ab-473) Antibody E021054-1 50μg/50μl IHC, WB Human, Mouse, Rat Rabbit Akt (Phospho-Ser473) Antibody
Cell Signaling part 2
 15 Cell Signaling part 2 Functions of Cell Surface Receptors Other cell surface receptors are directly linked to intracellular enzymes. The largest family of these is the receptor protein tyrosine kinases,
15 Cell Signaling part 2 Functions of Cell Surface Receptors Other cell surface receptors are directly linked to intracellular enzymes. The largest family of these is the receptor protein tyrosine kinases,
Supplementary Figure 1.TRIM33 binds β-catenin in the nucleus. a & b, Co-IP of endogenous TRIM33 with β-catenin in HT-29 cells (a) and HEK 293T cells
 Supplementary Figure 1.TRIM33 binds β-catenin in the nucleus. a & b, Co-IP of endogenous TRIM33 with β-catenin in HT-29 cells (a) and HEK 293T cells (b). TRIM33 was immunoprecipitated, and the amount of
Supplementary Figure 1.TRIM33 binds β-catenin in the nucleus. a & b, Co-IP of endogenous TRIM33 with β-catenin in HT-29 cells (a) and HEK 293T cells (b). TRIM33 was immunoprecipitated, and the amount of
Effect of Survivin-siRNA on Drug Sensitivity of Osteosarcoma Cell Line MG-63
 68 Chin J Cancer Res 22(1):68-72, 2010 www.springerlink.com Original Article Effect of Survivin-siRNA on Drug Sensitivity of Osteosarcoma Cell Line MG-63 Jing-Wei Wang 1, Yi Liu 2, Hai-mei Tian 2, Wei
68 Chin J Cancer Res 22(1):68-72, 2010 www.springerlink.com Original Article Effect of Survivin-siRNA on Drug Sensitivity of Osteosarcoma Cell Line MG-63 Jing-Wei Wang 1, Yi Liu 2, Hai-mei Tian 2, Wei
T cell maturation. T-cell Maturation. What allows T cell maturation?
 T-cell Maturation What allows T cell maturation? Direct contact with thymic epithelial cells Influence of thymic hormones Growth factors (cytokines, CSF) T cell maturation T cell progenitor DN DP SP 2ry
T-cell Maturation What allows T cell maturation? Direct contact with thymic epithelial cells Influence of thymic hormones Growth factors (cytokines, CSF) T cell maturation T cell progenitor DN DP SP 2ry
Oncolytic Adenovirus Complexes Coated with Lipids and Calcium Phosphate for Cancer Gene Therapy
 Oncolytic Adenovirus Complexes Coated with Lipids and Calcium Phosphate for Cancer Gene Therapy Jianhua Chen, Pei Gao, Sujing Yuan, Rongxin Li, Aimin Ni, Liang Chu, Li Ding, Ying Sun, Xin-Yuan Liu, Yourong
Oncolytic Adenovirus Complexes Coated with Lipids and Calcium Phosphate for Cancer Gene Therapy Jianhua Chen, Pei Gao, Sujing Yuan, Rongxin Li, Aimin Ni, Liang Chu, Li Ding, Ying Sun, Xin-Yuan Liu, Yourong
Effects of metallothionein-3 and metallothionein-1e gene transfection on proliferation, cell cycle, and apoptosis of esophageal cancer cells
 Effects of metallothionein-3 and metallothionein-1e gene transfection on proliferation, cell cycle, and apoptosis of esophageal cancer cells Z.Q. Tian 1, Y.Z. Xu 1, Y.F. Zhang 1, G.F. Ma 1, M. He 1 and
Effects of metallothionein-3 and metallothionein-1e gene transfection on proliferation, cell cycle, and apoptosis of esophageal cancer cells Z.Q. Tian 1, Y.Z. Xu 1, Y.F. Zhang 1, G.F. Ma 1, M. He 1 and
Supplementary Materials
 Supplementary Materials Figure S1. MTT Cell viability assay. To measure the cytotoxic potential of the oxidative treatment, the MTT [3-(4,5-dimethylthiazol- 2-yl)-2,5-diphenyl tetrazolium bromide] assay
Supplementary Materials Figure S1. MTT Cell viability assay. To measure the cytotoxic potential of the oxidative treatment, the MTT [3-(4,5-dimethylthiazol- 2-yl)-2,5-diphenyl tetrazolium bromide] assay
Cell Communication. Local and Long Distance Signaling
 Cell Communication Cell to cell communication is essential for multicellular organisms Some universal mechanisms of cellular regulation providing more evidence for the evolutionary relatedness of all life
Cell Communication Cell to cell communication is essential for multicellular organisms Some universal mechanisms of cellular regulation providing more evidence for the evolutionary relatedness of all life
